32 Bordetella
Whooping cough
Key points
• Whooping cough is caused by Bordetella pertussis, and occasionally by B. parapertussis.
• Paroxysmal cough, typically with vomit and whoop, lasts for several weeks or even months.
• Pertussis is a human disease (most severe in children) and has no animal or environmental reservoir.
• Clinical diagnosis is unreliable in mild cases.
• Bacterial culture is the optimal method of laboratory confirmation.
• Therapy for established cases is unsatisfactory.
• Immunity to B. pertussis infection is serotype-specific.
• Vaccine containing the three serotype antigens is safe, and is effective if the vaccination schedule is optimal.
• Eradication of whooping cough by childhood vaccination is an achievable goal.
The genus Bordetella constitutes one of the groups of very thin ovoid or rod-shaped Gram-negative bacilli, often described as parvobacteria. The genus contains two notable human pathogens, Bordetella pertussis and B. parapertussis, which cause one of the most frequent and serious bacterial respiratory infections of childhood in communities not protected effectively by vaccination.
Description
Bordetellae used to be classified in the genus Haemophilus. However, growth is not dependent on either of the nutritional factors X and V (see p. 325), and B. parapertussis and B. bronchiseptica do not require blood for their growth. The three species resemble one another in being small Gram-negative bacilli, in causing infection of the respiratory tract, and in sharing some surface antigens.
Bordetella pertussis
This is the most fastidious of the bordetellae. It produces toxic products that must be absorbed by a culture medium containing charcoal, or starch, or a high concentration of blood as in the original Bordet–Gengou medium. Because agglutination forms an important part of identification, a smooth growth is essential, and this is best provided by charcoal–blood agar. B. pertussis is a strict aerobe, with an optimal growth temperature of 35–36°C. Even under these conditions it usually takes 3 days for colonies to be visible to the naked eye.
Typical colonies are shiny, greyish white, convex, and with a butyrous consistency. By slide agglutination, they react strongly with homologous (pertussis) antiserum, and weakly or not at all with parapertussis antiserum, depending on the specificity of the reagent. Subculture reveals no growth on nutrient agar.
The organism produces three major agglutinogens (1, 2 and 3) which can be detected by the use of absorbed single-agglutinin sera. Factor 1 is common to all strains; the three serotypes pathogenic to man (type 1,2, type 1,3 and type 1,2,3) also possess factor 2 or factor 3 or both factors, and these type-specific agglutinogens have a vital role in immunity to infection.
Bordetella parapertussis
This organism is readily distinguished from B. pertussis by its ability to grow on nutrient agar, with the production of a brown diffusible pigment after 2 days (Table 32.1). It also grows more rapidly than B. pertussis on charcoal–blood agar, and is agglutinated more strongly by parapertussis than by pertussis antiserum.
Table 32.1 Differential properties of B. pertussis and B. parapertussis
| Property | B. pertussis | B. parapertussis |
|---|---|---|
| Duration of incubation to yield visible colonies | 3 days | 2 days |
| Growth on nutrient agar | None | Good |
| Pigment diffusing in medium | None | Brown |
| Slide agglutination with: | ||
| Pertussis antiserum | Strong | Weak |
| Parapertussis antiserum | Weak | Strong |
B. parapertussis usually causes less severe illness than B. pertussis, and is uncommon in most countries, although occasionally it has been responsible for outbreaks of whooping cough.
Bordetella bronchiseptica
Colonies of this species are visible on nutrient agar after overnight incubation; it differs from the other species by also being motile and by producing an obvious alkaline reaction in the Hugh and Leifson medium that is used to differentiate oxidative from fermentative action on sugars. It is therefore placed by some taxonomists in the genus Alcaligenes; however, it is readily distinguished from the intestinal commensal Alcaligenes faecalis by its rapid hydrolysis of urea.
Although rarely encountered in human infections, B. bronchiseptica is a common respiratory pathogen of animals, especially laboratory stocks of rodents. Because it shares antigens with other bordetellae, animals must be checked for freedom from bordetella antibody before they are used in the preparation of specific antisera. For this reason, sheep or donkeys have sometimes been used in preference to rodents.
Pathogenesis
Whooping cough is a non-invasive infection of the respiratory mucosa, with man as the only natural host. In a typical case, an incubation period of 1–2 weeks is followed by a ‘catarrhal’ phase with a simple cough but no distinctive features. Within about a week, this leads into the ‘paroxysmal’ phase, with increasing severity and frequency of paroxysmal cough, which may last for many weeks and be followed by an equally prolonged ‘convalescent’ phase.
In the initial preventable stage of the infection there is colonization of the ciliated epithelium of the bronchi and trachea with vast numbers of bacteria whose agglutinogens play a vital and type-specific role in attachment. B. pertussis produces a tracheal cytotoxin that paralyses the cilia and leads to paroxysms of coughing as an alternative means of removing the increased mucus. Another bacterial product, called pertussis toxin, is responsible for the characteristic lymphocytosis in uncomplicated whooping cough. A subsequent increase in the number of neutrophils, together with fever, suggests bronchopneumonia or other secondary infection, maybe with pyogenic cocci. Blockage of airways may cause areas of lung collapse, and anoxia may lead to convulsions, although with modern intensive care the disease is rarely fatal.
Clinical features
Pertussis (severe cough) has been recognized as a clinical entity for several centuries. Typically, the child suffers many bouts of paroxysmal coughing each day; during these, with no pause for air intake, the tongue is protruded fully, fluids stream from the eyes, nose and mouth, and the face becomes cyanotic; when death seems imminent, a final cough clears the secretions and, with a massive inspiratory effort, air is sucked through the narrowed glottis, producing a long high-pitched whoop – hence the term whooping cough. Such attacks often terminate with vomiting. Between them the patient does not usually appear ill.
If a characteristic attack is witnessed, a diagnosis of pertussis is usually made on clinical grounds alone. However, the illness is often mild and atypical, especially in:
• younger children who have been incompletely immunized
• very young infants partially protected by maternal antibody.
In these cases, the laboratory has a vital role in diagnosis, because similar coughing may be caused by a variety of viruses and such illness is generally mild and of short duration. Here, the term pseudo-whooping cough has been aptly applied. False diagnosis may create a popular impression of pertussis as a trivial disease; and it is recommended that, in the absence of positive bacterial culture, whooping cough should not be diagnosed for paroxysmal coughing lasting less than 3 weeks. With genuine pertussis, the illness is likely to persist for months rather than weeks. Furthermore, because pertussis vaccine cannot be expected to protect against viral infection, estimates of vaccine efficacy require an accurate diagnosis. Thus, a study in the UK found an efficacy of 93% against pertussis confirmed by bacterial culture, compared with only 82% for cases diagnosed solely on clinical criteria.
In developing countries, whooping cough is still a major cause of death; however, in developed countries, concern is focused on a very prolonged and frightening illness with possible respiratory and neurological sequelae, on the anxiety and exhaustion of parents, and on the heavy use of hospital and community medical resources.
Experimental infection in animals
Some, though not all, of the features of human disease have been produced in animals. Thus, marmosets and rabbits develop catarrh during prolonged colonization of the respiratory tract; they produce a similar range of agglutinin response to vaccination, and this immunity shows evidence of serotype specificity.
However, the mouse, which has long been used in the evaluation of pertussis vaccine potency, does not show these features. It can be infected and even killed by degraded organisms of serotype 1, which have lost the type-specific agglutinogens (2 and 3) that are necessary for human infection. It also reveals additional properties of pertussis toxin that are not seen in human beings: histamine sensitization, and islets activation (including hyperinsulinaemia and hypoglycaemia). Furthermore, pertussis toxin and other components of the organism – filamentous haemagglutinin and pertactin – are virulence factors in the mouse; but their role in human infection is uncertain. It is therefore necessary to interpret with caution experimental evidence from mice or other small rodents.
Laboratory diagnosis
Bacterial culture
Because atypical clinical cases occur frequently, laboratory confirmation of the diagnosis is often essential. Bacterial culture has the highest specificity of the tests available. In the absence of really effective therapy, accuracy in the diagnosis is more important than speed. Bacterial culture has the additional advantage that the isolate can be serotyped and genotyped, and thus provide valuable epidemiological information.
So rarely has a positive culture been obtained from a healthy person, other than one incubating the disease, that a false-positive result can be discounted. Moreover, with good technique of swabbing and culture, the organism can be recovered up to 3 months from the onset of illness when coughing persists. This casts doubt on the widespread belief that the bacterium is eliminated in a few weeks, and has implications for the transmission of infection.
Although the disease is mainly in the lower respiratory tract, the organism can be recovered readily from the nasopharynx. ‘Cough plates’ and postnasal swabs are unsatisfactory because of overgrowth by commensal bacteria. A pernasal swab acquires fewer commensals, and these can be suppressed by penicillin (0.25 unit/mL = 0.15 mg/L) or cefalexin (30 mg/L) in the charcoal–blood agar plate; higher concentrations may suppress bordetellae. Pernasal swabs on flexible wire are available commercially; the tip is directed downwards and towards the midline, passing gently along the floor of the nose for about 5 cm (depending on the patient’s age) until stopped by the posterior wall of the nasopharynx. Practice in swabbing is necessary – initially with a co-operative adult! If old enough, patients should be warned to expect a tickling sensation but no pain; and a child’s head should be held steady. Ideally, a segment of the culture plate should be inoculated immediately after withdrawal of the swab. The use of transport medium reduces the isolation rate. A single swab may yield a negative culture, but isolation rates of up to 80% may be achieved by taking specimens on several successive days.
In the laboratory, the inoculum is spread to give separate colonies, and the plate is incubated for at least 7 days before being discarded as negative. Because of the prolonged incubation, the medium should have a depth of 6–7 mm (40 mL in a 9-cm dish) and a bowl of water may be placed in the incubator to reduce drying of the culture. Cefalexin tends to give ‘rough’ growth, which may have to be subcultured on cefalexin-free medium for reliable serological identification.
Detection of bacterial antigens or DNA
Bordetella antigens may be detected in serum and urine in tests with specific antiserum. Alternatively, bacteria in nasopharyngeal secretions are labelled with fluorescein-conjugated antiserum and examined by ultraviolet microscopy. This method has the theoretical advantage, compared with culture, of detecting dead bordetellae. However, false-negative results are likely unless the patient’s own antibody is removed from the bacteria by enzyme before application of the fluorescent reagent. Moreover, false-positive results may occur because of serological cross-reactions with organisms such as staphylococci, yeasts, haemophili and moraxellae, some of which resemble bordetellae microscopically. Bordetella antiserum should be absorbed with these organisms, but appropriate reagents are not readily available. Reports on the high specificity of this test lack conviction in the absence of reliable evidence on the true diagnosis.
There have been numerous studies of the polymerase chain reaction (PCR) in the detection of bordetella DNA in nasopharyngeal specimens, by the use of various primers. However, the method is relatively expensive and technically demanding compared with culture; and as yet there is a lack of consensus on its diagnostic reliability, with the need to detect both B. pertussis and B. parapertussis, and then to distinguish between them. Moreover, to be of epidemiological value, these methods would need modification to enable them to identify the serotype of the infecting strain of B. pertussis.
Furthermore, a test with very high sensitivity may merely detect the transient presence of a small number of bacteria attempting to colonize the mucosa before being eliminated from an immune host; such scanty bacteria would constitute a minimal risk of transmission to contacts.
Detection of bordetella antibody
Sera and nasopharyngeal secretions can usefully be examined for antibody. However, a negative result does not exclude pertussis because the serological response is often slow and weak, especially in very young children. More importantly, a positive result needs careful interpretation because antigens are shared with other organisms (see above). Even with insensitive tests, such as agglutination, pertussis antibodies are readily detected in the sera of healthy persons. More sensitive techniques, such as enzyme-linked immunosorbent assay (ELISA), are liable to increase the number of false-positive results and thereby give spurious respectability to a diagnosis that should rightly be ‘pseudo-whooping cough’. The need at present in serological diagnosis is not for greater sensitivity but for greater specificity. Even then, the detected antibody may be an ‘anamnestic’ response to previous pertussis infection or vaccination, provoked non-specifically by a current, antigenically unrelated, illness.
The ready detection of antibody in adolescents and adults, resulting from past infection or vaccination, has led some to believe that these people are an important reservoir of pertussis infection, despite the rarity of a positive culture on pernasal swabbing.
Nevertheless, bacterial agglutination may be a useful guide in the serodiagnosis of pertussis, provided that sera are absorbed with type 1 organisms and titrated for the more specific agglutinins 2 and 3, and that paired sera are taken about 3 weeks apart to detect a greater than fourfold rise in titre. This requires a serum sample of at least 0.5 mL on each occasion.
Differential blood count
Although lymphocytosis is a characteristic response to pertussis infection, many cases of true pertussis do not develop a significant increase in circulating lymphocytes; conversely, there are so many other causes of lymphocytosis that a positive result lacks diagnostic specificity.
Treatment
Antimicrobial drugs
Most antibiotics have little or no clinical effect when the infection is well established, even though the organism may be sensitive in vitro.
The drug of choice is a macrolide such as azithromycin, clarithromycin or erythromycin, which may reduce the severity of the illness if given before the paroxysmal stage. If given for at least 14 days, erythromycin has sometimes eliminated the organism and so reduced the exposure of contacts. However, positive cultures are frequently obtained after short periods of erythromycin therapy.
A macrolide may also be given to protect non-vaccinated infants, although it seems unrealistic to expect this treatment to be maintained throughout the several months that the older sibling (or adult) may remain infectious.
Appropriate antibiotics should, of course, be administered to patients who show signs of secondary bacterial infection.
Other measures
Cough suppressants and corticosteroids may control the paroxysms, but may be harmful by encouraging retention of secretions. Cyanosis and anoxia can be reduced by avoiding sudden noises, excitement or excessive medical examination, which tend to precipitate paroxysms. Mucus and vomit should be removed to prevent their inhalation.
Treatment with pertussis immunoglobulin has been tried, but with limited success, probably because such materials have never been checked for the presence of all three agglutinins.
Because of the dearth of effective therapy for whooping cough, the widespread use of pertussis vaccine is of supreme importance (see below).
Epidemiology
Source and transmission of infection
Most new cases arise from patients (usually children, occasionally adults) with typical symptoms, presumably because the paroxysmal cough provides an efficient means of droplet dissemination. Atypical cases have only a minor role in transmission; long-term asymptomatic carriage is unknown. The degree of contact is important: 80–90% of non-immune siblings exposed in the household become infected, compared with less than 50% of non-immune child contacts at school. Antibiotic therapy may reduce transmission, but is not completely effective.
Incidence and mortality
Pertussis infection occurs worldwide, affects all ages, and is a major cause of death in malnourished populations. In developed countries the mortality rate has gradually declined with a combination of improved socio-economic conditions, availability of intensive care in hospitals, and antibiotic therapy to combat secondary infection. However, the latter constitute an unnecessary use of medical resources for a disease that is eminently preventable by vaccination.
The disease is most severe and the morbidity rate highest in the first 2 years of life; most fatal cases are in infants less than 1 year old. Even very young babies are not immune: maternal antibody does pass to the fetus, but it rarely contains all three agglutinins and protection is incomplete.
Although one attack usually confers long-lasting immunity, infection with a different serotype of the organism can occur subsequently.
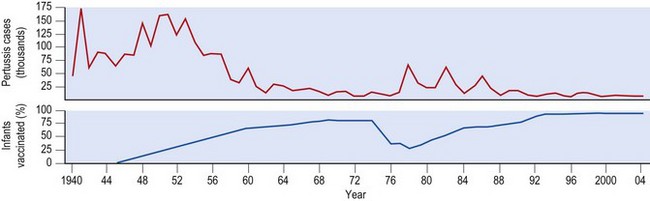
The disease occurs in epidemic waves at about 4-year intervals – the time needed to build up a new susceptible population after the ‘herd’ immunity produced by an epidemic. Figure 32.1 illustrates the pattern for England and Wales up to 2004, since when the situation has remained unchanged. The maintenance of a 4-year cycle presumably results from the interaction of various factors, such as the degree of artificial immunity produced by high vaccination rates, and the levels of natural immunity that follow either large epidemics or a high background incidence of endemic pertussis in inter-epidemic intervals.
Figure 32.1 also illustrates how variations in the rates of uptake of pertussis vaccine have affected the incidence of whooping cough more than the steady improvement in the general health of the population that continued throughout the period. After the gradual introduction of pertussis vaccination during the 1950s, there was a steady reduction in the size of epidemics until the 1970s. Unfounded fear of brain damage caused a loss of faith in the vaccine, and three large epidemics occurred before the slow restoration of confidence in the vaccine began to take effect.
Prevalence of serotypes
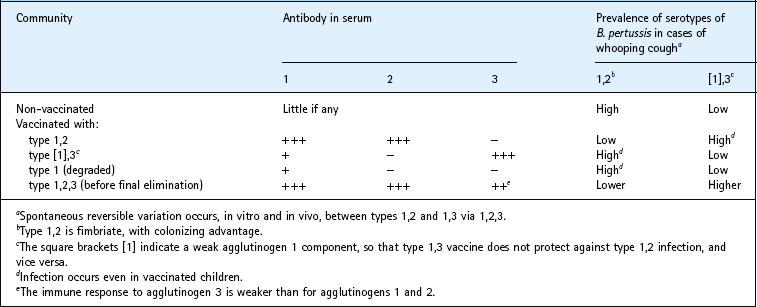
The three serotypes of B. pertussis pathogenic for man are liable to spontaneous reversible variation, in vitro and in vivo, between types 1,2 and 1,3 via 1,2,3. Fimbriae are readily demonstrable on strains possessing agglutinogen 2, aiding colonization of the respiratory mucosa; serotypes 1,2 and 1,2,3 predominate among the strains isolated from patients in non-vaccinated communities.
Table 32.2 shows the occurrence of serotypes of B. pertussis in different communities. In the 1960s, many countries used type 1,2 vaccine, and these two serotypes were suppressed. However, type 1,3 organisms, which have a weak agglutinogen 1 component, became predominant in these countries, and caused infection even in vaccinated children before the vaccine was modified in the late 1960s by addition of agglutinogen 3. Similarly, countries that used a vaccine deficient in agglutinogen 2, or in both agglutinogens 2 and 3, saw a predominance of type 1,2 infection, even in vaccinated children.
To be effective, it seems, a vaccine must contain all three agglutinogens, as recommended by the World Health Organization. However, because the agglutinin 3 response is usually the weakest (when type 1,2,3 vaccine is used), type 1,3 organisms are the last to be eliminated from a community with an effective vaccination programme.
Genotypes
It is not possible to trace the spread of infection by serotyping isolates because there are only three serotypes and they undergo spontaneous variation. In contrast, more than 40 genotypes have been demonstrated in macro-restriction profiles by pulsed-field gel electrophoresis of DNA digests. Isolates within a household have been shown to belong to the same genotype, but the technique is probably too expensive and time-consuming for routine use.
Control
Treatment and quarantine
Antibiotics and immunoglobulins currently available are not very effective for the treatment of patients or the protection of contacts. Because patients typically disseminate the organism for many weeks or months, and because children are infectious even before the most characteristic symptoms develop, control of the disease by quarantine is unrealistic.
Vaccination
Vaccination is safe and more than 90% effective and is strongly recommended. The vaccines still in widespread use are suspensions of whole bacterial cells, killed by heat or chemicals, and are administered by deep intramuscular injection. Adsorption of the bacteria on to an adjuvant, such as aluminium hydroxide, enhances the immune response (particularly important with agglutinogen 3) and also causes fewer adverse reactions (Table 32.3).
Table 32.3 Plain and adsorbed pertussis vaccine
| Vaccine | Immune response | Adverse reactions |
|---|---|---|
| Plain | Weaker | More |
| Adsorbed (onto adjuvant) | Stronger | Fewer |
Three conditions are essential for good protection:
1. Presence of all three agglutinogens in the vaccine
Since there is little passive protection from the mother and effective active immunity cannot be achieved until after the third injection of vaccine, the first dose is given as soon as a good response can be obtained. In many countries, therefore, the first injection is recommended at 3 months of age. In the UK and some other countries, concern for the vulnerability of the young infant prompted a start at 2 months, although the immune response is somewhat weaker and there have been doubts about the effectiveness of this early start. An upsurge of invasive infection with Haemophilus influenzae type b followed in the Netherlands when the policy of giving the first dose at 3 months was changed to 2 months. For these and other aspects of vaccination, see also Chapter 70.
Safety of pertussis vaccine
Minor adverse reactions occur in about one-half of vaccinated children, and can be considered as part of the normal immune response. Parents should therefore be warned to expect possible erythema and local swelling, slight feverishness, and crying. Of much more concern are possible neurological sequelae, but the National Childhood Encephalopathy Study in the UK and several other studies have shown that pertussis vaccination merely triggers the manifestation of neurological disorders that would occur in any case, and there is no firm evidence that the vaccine causes serious long-term adverse side-effects.
Contra-indications to pertussis vaccination
Severe adverse reaction to a previous dose has been considered the only firm contra-indication. A current feverish illness (not merely snuffles) is cause for postponement until the child is well. Parental concern over a possible neurological contra-indication is due reason for consultation with a paediatrician. Allergy is not a contra-indication; neither is age – children who missed vaccination in infancy may receive the normal three-dose course. Vaccination is also sometimes advised for adults, such as nurses and doctors in appropriate hospitals.
Acellular pertussis vaccine
Although doubts about the efficacy and safety of whole-cell pertussis vaccine have passed, the urge to identify the essential protective components persists. Trials in different countries (including one with type 1,2 prevalence and another with type 1,3 prevalence), and with vaccines containing various components, may reveal a correlation between the protection of children and the response to individual pertussis antigens and type-specific agglutinogens.
A large-scale trial with good diagnostic criteria in Sweden showed that antibodies to pertussis toxin and filamentous haemagglutinin do not confer protection in the child, although these antigens have been incorporated in nearly all acellular vaccines because they provide mouse-protection (see above). This trial did indicate a correlation between agglutinin titres and protection of children, and it showed that whole-cell vaccine had a higher efficacy than any of the acellular vaccines used. Moreover, the whole-cell vaccine has an additional adjuvant effect on other antigens given simultaneously; in the UK an increase in Haemophilus influenzae type b infections followed the replacement of whole-cell pertussis vaccine with an acellular product owing to a reduced antibody response.
Acellular vaccines are now used in various countries, but without any assurance that they produce an adequate response to both agglutinogens 2 and 3. Moreover, they are very expensive, putting a questionable burden on limited national health resources, especially in developing countries where the need is greatest.
Eradication
Vaccination aims at a herd immunity, which breaks the cycle of transmission because the organism dies before finding a new susceptible host (see Ch. 70).
In several countries, good whole-cell vaccine is available. Eradication is possible, but even if high levels of vaccination of infants are maintained it will be some years before adequate herd immunity is achieved within the child population.
Editorial. Pertussis: adults, infants, and herds. Lancet. 1992;339:526–527.
Health Protection Agency. Laboratory confirmed cases of pertussis in 2009; and Quarterly vaccination coverage in the UK. Health Protection Report. 4, 2010. No.25
Jenkinson D. Natural course of 500 consecutive cases of whooping cough: a general practice population study. British Medical Journal. 1995;310:299–302.
Khattak MN, Matthews RC. Genetic relatedness of Bordetella species as determined by macrorestriction digests resolved by pulsed-field gel electrophoresis. International Journal of Systematic Bacteriology. 1993;43:659–664.
Lang F, ed. Encyclopedia of Molecular Mechanisms of Disease, ed 2, Heidelberg: Springer, 2008.
Preston NW. Pertussis (whooping-cough): the road to eradication is well sign-posted but erratically trodden. Infectious Diseases Review. 2000;2:5–11.
Preston NW. Why the rise in Haemophilus influenzae type b infections? Lancet. 2003;362:330–331.
Preston NW. Diagnosis and prevention of pertussis. Lancet. 2006;368:1769.
Preston NW, Matthews RC. Immunological and bacteriological distinction between parapertussis and pertussis. Lancet. 1995;345:463–464.
Preston NW, Matthews RC. Acellular pertussis vaccines: progress but déjà vu. Lancet. 1998;351:1811–1812.
Salmaso S, the Eurosurveillance editorial team. Pertussis vaccine schedules across Europe. Eurosurveillance. 2004;9:70–71.