54 Reoviruses
Gastroenteritis
Key points
• The Reoviridae comprise four genera which infect humans; they are characterized by the possession of 10–12 segments of double-stranded RNA encased within an unenveloped capsid of icosahedral symmetry.
• When viewed under the electron microscope, the characteristic wheel-shaped virus particles measure 70–75 nm in diameter.
• Group A rotaviruses are the major cause of acute gastroenteritis in infants and young children worldwide.
• A unique feature of rotavirus replication is the transient acquisition of an envelope during the process of budding into the endoplasmic reticulum.
• Rotaviruses produce NSP4, the first described viral enterotoxin.
• Diagnosis of rotavirus gastroenteritis is usually made by the detection of viral antigen (VP6) using enzyme-linked immunosorbent and immunochromatographic assays.
• A distinct winter seasonality of rotavirus infection is evident in temperate countries; infection is year-round in tropical countries.
• Rehydration and restoration of electrolyte balance are the primary aims of treatment of acute rotavirus gastroenteritis.
• Two live oral rotavirus vaccines are entering childhood immunization schedules and promise to substantially reduce the global rotavirus disease burden.
The family Reoviridae comprises a diverse collection of viruses that infect man, many mammals, other vertebrates (including birds), plants and insects. The genus Rotavirus is the most important cause of severe infantile gastroenteritis throughout the world, and is a recognized aetiological agent of diarrhoea in the young of many animal species. Viruses in the genera Orbivirus and Coltivirus infect various species of insects and can be transmitted to vertebrates including man, and can thus be described as arboviruses (see Ch. 51).
Classification
Four of the nine genera of the Reoviridae family infect humans:
• Orthoreovirus – reovirus types 1, 2 and 3
• Orbivirus and Coltivirus – various serogroups
• Rotavirus – of seven species, formerly called serogroups (A–G), Rotavirus A–Rotavirus C have been detected in humans. Multiple genotypes exist within Rotavirus A.
The virion has icosahedral symmetry and is triple-layered (the double-shelled capsid surrounds a core); it does not possess an envelope and measures 70–75 nm in diameter under negative-stain electron microscopy. The RNA genome consists of 10 to 12 segments of double-stranded RNA.
Replication
The virus replicates in the cytoplasm of infected cells. Attachment is via a specific protein component of the outer capsid (e.g. σ1 of orthoreoviruses or viral protein (VP) 4 of rotaviruses) to a cellular receptor; orthoreoviruses use sialic acid for binding and junctional adhesion molecule-A as a post-binding receptor, whereas rotaviruses use both sialic acid and α2β1 integrin for binding and α2β1 and other integrins, and heat shock protein 70 as post-binding receptors. The viral protein–receptor interaction leads to a conformational change in the viral capsid, which enables orthoreoviruses to enter the cell by receptor-mediated endocytosis. The multistep attachment and entry process for rotaviruses is complex and involves a remarkable conformational change in the structure of VP4 induced by proteolytic cleavage, exposing additional attachment sites. When crossing the cell membrane, the outer capsid is removed, but the viral particle is never completely uncoated, leaving the viral genomic RNA in the core of the double-layered particle (subviral particle). RNA is transcribed by the virion-associated, RNA-dependent RNA polymerase complex to produce messenger RNA (mRNA) molecules from each genomic RNA segment. The mRNA molecules, which are capped but not polyadenylated, leave the subviral particle through channels penetrating the outer and inner capsids, and are then translated to generate the various viral proteins. They also act as templates for RNA replication, which is completed by the RNA polymerase complex, and packaged within new core structures. The whole replication events occur within electron-dense structures (viroplasms) in the cytoplasm, which are localized adjacent to the nucleus and the endoplasmic reticulum (ER). Nascent double-layered particles, after binding to an intracellular viral receptor (NSP4), bud from viroplasms into the ER in a unique process during which double-layered particles transiently acquire an envelope. At the same time the outer capsid proteins, VP7 and VP4, are incorporated into newly synthesized particles, which become mature, triple-layered particles by losing their envelope. These are then released from the apical surface of enterocytes after vesicular transport from the ER by a pathway bypassing the Golgi apparatus.
Association with clinical illness
The first human isolates of the Reoviridae were recovered from both respiratory secretions and faeces, but could not be associated with disease – hence the name reovirus (respiratory enteric orphan). However, orthoreoviruses cause systemic disease (meningitis and encephalitis) in mice, and this virus–host system has been used extensively to study viral pathogenesis. Although most of the human population is exposed to and develops antibodies against orthoreoviruses from an early age, there is still no clear link between these viruses and illness.
The orbiviruses include several serogroups that infect man and are transmitted by a variety of insects (ticks, midges and mosquitoes). In the blood the virus is associated with erythrocytes, and thus is hidden from immune responses. Clinically, patients experience a febrile illness, with rashes in 10% and leucopenia in two-thirds of cases. Infections of the central nervous system leading to meningitis or encephalitis are seen in 3–7% of laboratory-confirmed cases.
The coltiviruses are also transmitted by insects (ticks, mosquitoes), and rodents are considered to be the main animal reservoir. Infection is spread to man by insect bite. A febrile illness develops with gastrointestinal symptoms (20%), rash (10%), and meningitis or encephalitis (3–7%) (see Ch. 51).
Rotaviruses are the main cause of acute gastroenteritis in infants and young children, as well as in the young of many animal species.
Rotaviruses
Infection with rotavirus can result in a wide spectrum of clinical outcomes, ranging from asymptomatic infection to severe, life-threatening gastroenteritis. More severe disease outcomes are typically encountered among children in developing countries. Rotavirus also causes diarrhoea in the young of a wide variety of avian and mammalian species including cattle, sheep, goat, horses, pigs, dogs, cats and mice, rabbits, monkeys and many others.
Description and classification
Morphology
The virion measures 75 nm in diameter and has characteristic sharp-edged, double-shelled capsids, which in electron micrographs look like spokes grouped around the hub of a wheel (the Latin word, rota, means wheel); this appearance is diagnostic (Fig. 54.1). Cryo-electron microscopy has shown that the triple-layered particle is penetrated by 132 large channels and that the virion has 60 spikes on its surface that consist of trimers of VP4 (Fig. 54.2) (see below).
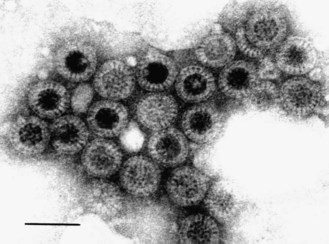
Fig. 54.1 Negatively stained electron micrograph of rotavirus particles in a faecal specimen. Potassium phosphotungstic acid stain. Bar = 100 nm.
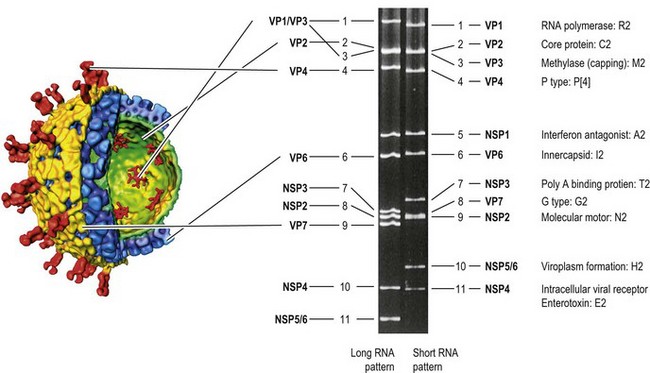
Fig. 54.2 Schematic diagram showing the relationships between the structure of a rotavirus virion (derived from cryo-electron microscopy images and computer image processing) and the genomic double-stranded RNA segments separated by polyacrylamide gel electrophoresis. Two RNA patterns, long and short, are shown. The cut-away representation of the reconstructed virion shows the triple-layered icosahedral architecture and the locations of the various structural proteins. Note that coding assignment is slightly different between long and short RNA pattern strains. RNA segment 10 of the short RNA strain is the result of rearrangement of RNA segment 11 of the long RNA pattern. The outer (VP7 and VP4), middle (VP6) and inner (VP2) layers are shown in orange, blue and green, respectively. The flower-like structures inside the VP2 layer represent VP1–VP3 complexes, which function as RNA polymerase and capping enzymes. The genotype of each RNA segment of the short RNA strain (strain DS-1) is shown in blue. The corresponding genotypes of the long RNA strain (strain Wa) are G1-P[8]-I1-R1-C1-M1-A1-N1-T1-E1-H1.
(The 3D image structure of reconstructed rotavirus virion was conceived by B. V. V. Prasad, Baylor College of Medicine, Houston, Texas, USA.)
Genome and gene-coding assignments
The 11 segments of double-stranded RNA can be extracted easily from rotaviruses and separated by polyacrylamide gel electrophoresis. This has been used to establish so-called ‘electropherotypes’ of rotavirus isolates (Fig. 54.2). As well as ‘long’ and ‘short’ electropherotypes (differing in the rates of relative migration of RNA segments 10 and 11), various minor differences in the migration of corresponding segments have been recognized and utilized extensively in epidemiological studies.
Generally, each genome segment codes for only one virus-specific protein (VP). The gene-coding assignments have been established (Fig. 54.2). RNA segments 1, 2 and 3 code respectively for the inner core proteins VP1 (which functions as the viral polymerase), VP2 (the main scaffolding protein) and VP3 (the capping enzyme). RNA segment 6 codes for the middle layer protein, VP6, which is the single most abundant rotavirus protein and which interacts with the core protein VP2 and the outer capsid proteins VP7 and VP4 (see below). VP6 carries epitopes specifying groups and subgroups. To date, seven different group antigens (A–G) have been identified; for groups A–E a complete lack of serological cross-reactivity has been shown.
• Rotavirus A is responsible for the vast majority of human rotavirus infections in infants and young children, and may also cause disease in adults.
• Rotavirus B was initially discovered as the cause of outbreaks of acute diarrhoea among all age groups in China, and is now endemic in several countries including China, India, Bangladesh, Myanmar and Nepal.
• Rotavirus C causes occasional episodes of gastroenteritis in older children.
The outer capsid (third layer) is formed by two proteins: VP7, a glycoprotein (encoded by RNA 7, 8 or 9, depending on the strain, and determining the G serotype), and VP4 (encoded by RNA 4, and determining the P serotype). Each of these surface proteins carries neutralization epitopes that define the virus serotype. VP4 is cleaved post-translationally into VP5* and VP8* (an asterisk is to denote post-translational product); this proteolytic cleavage is essential for infectivity. Six non-structural proteins are coded for by RNAs 5 (NSP1), 7, 8 or 9 (NSP2 and NSP3, depending on the strain), and 10 or 11 (NSP4, NSP5 and NSP6, depending on the strain). NSP2 is proposed to act as a molecular motor to drive replicating mRNA into the nascent viral particles and so maintain nucleotide pools in viroplasms during replication. Both NSP2 and NSP5 are involved in viroplasm formation. NSP3 functions as polyadenylic acid-binding protein (rotavirus mRNA is not polyadenylated), and binds to the 3′ end of viral mRNA and to cellular eIF4G, thus enhancing the translation of viral mRNA. While NSP4 is an intracellular viral receptor playing an important role in viral morphogenesis (see above), it also functions as a viral enterotoxin (see below).
Antigenic and genetic diversity
Rotavirus A is genetically diverse in each of the 11 genome segments (called genotypes), and a nucleotide sequence-based, complete genome classification system is used; the genome of individual rotavirus strains is given the complete descriptor of Gx-P[x]-Ix-Rx-Cx-Mx-Ax-Nx-Tx-Ex-Hx (Fig. 54.2). Of these eleven genotypes, the G and P genotypes, corresponding to the G serotype and the P serotype, respectively, have been extensively investigated because of their importance in protective immunity. There are thus far 26 G genotypes and 35 P genotypes reported among human and animal rotaviruses. Reverse transcriptase–polymerase chain reaction (RT-PCR) with gene- and type-specific primers has been widely applied as a reliable typing procedure. Although the correlation between G serotypes (determined by neutralization assays) and G genotypes (determined by molecular methods) is absolute, not all P types have as yet been confirmed as serotypes, and therefore the P-serotype designation differs from the P-genotype designation (which is included in a square bracket). For example, strain Wa is designated as P1A[8]G1, where P1A represents the P-serotype; [8] represents the P-genotype; and G1 represents the G-genotype/serotype.
At the molecular level, several factors have been identified that can explain the genomic and antigenic variability of co-circulating rotavirus strains.
• Like other viruses that depend on virion-associated, RNA-dependent RNA polymerases for their replication, rotavirus genomes undergo frequent point mutations that accumulate over time and give rise to multiple lineages and sublineages (when this occurs in the neutralization proteins, antigenic drift will result).
• Rotaviruses, like other segmented RNA viruses, undergo extensive reassortment in doubly infected cells. This has been shown to occur both in vitro and in vivo. If RNA segments coding for serotype-specific proteins are involved, major antigenic changes can result (antigenic shift).
• Rotaviruses may be transferred to man from animal species either as whole virions or by reassortment of genome segments. The result is that in such cases, the genome of human group A rotavirus isolates is related to that of animal rotaviruses (e.g. cat, cow, pig), either in whole or in part.
• Rotaviruses that establish chronic infections in immunodeficient hosts may undergo various forms of genome rearrangement, resulting in highly atypical RNA profiles.
Pathogenesis and immunity
Rotaviruses replicate exclusively in the differentiated epithelial cells at the tips of the small intestinal villi and the crypts, which contain undifferentiated enteroctye stem cells, are spared. Progeny virus is produced after 10–12 h, and released in large numbers into the intestinal lumen ready to infect other cells. Biopsies show atrophy of the villi and mononuclear cell infiltrates in the lamina propria. The pathogenesis of rotavirus diarrhoea includes both malabsorptive and secretory components. Malabsorption may be consequent upon damage to mature absorptive enterocytes resulting in malabsorption of nutrients, electrolytes and water; virus-induced down-regulation of the expression of absorptive enzymes; and functional changes in tight junctions between enterocytes leading to paracellular leakage. Secretory mechanisms include those mediated by activation of the enteric nervous system and the effect of NSP4, the latter being via activation of cellular Cl− channels (different from the cystic fibrosis transmembrane regulator), leading to increased Cl− and consequently water secretion.
Rotavirus infection was previously thought to be limited to the intestine, but rotavirus causes viraemia for at least a short period in the acute phase of infection in immunocompetent infants as well as in experimentally infected animals. The clinical significance of this systemic spread of rotavirus is unclear, however.
Studies of the natural history of rotavirus infection show that immunity acquired following primary infection results predominantly in protection against the development of subsequent severe disease, but that protection against asymptomatic infection or mild disease is much less. Such post-infection immunity is believed to be mediated by both humoral and cell-mediated immune responses. Rotavirus-specific immunoglobulin (Ig) A antibodies on the enteric mucosal surface are thought to be the primary mediator of protective immunity.
The role of the innate immune system in rotavirus infection has captured recent attention. NSP1 functions as a viral ubiquitin ligase, interacting with and promoting the degradation of IFN regulatory factor (IRF) 3 and IRF7 through a ubiquitination-proteasome mechanism. NSP1 also inhibits activation of nuclear factor-κB. Thus, rotavirus counteracts the production of INFβ by host cells.
Clinical features
The onset of symptoms is abrupt after a short incubation period of 1–2 days. Fever, vomiting and watery diarrhoea are seen in the majority of infected children, lasting for 2–6 days. If body fluids are not replaced, dehydration ensues that may range in severity from mild to life threatening. There is little evidence that illness severity is related to virus serotype. Rotavirus infection can cause gastroenteritis in older children and adults, although severe disease is less common. Outbreaks of gastroenteritis due to rotavirus infection have been observed in the elderly, in whom severe dehydration can result. In the immunodeficient host, a persistent infection may occur with severe chronic diarrhoea associated with rotavirus excretion that can last for many months.
Laboratory diagnosis
At the peak of infection, as many as 1011 virus particles per millilitre of faeces are present, and can be detected by a variety of methods. Electron microscopy will easily detect the characteristic virus particles (Fig. 54.1). In the majority of cases there are sufficient numbers of virions in faeces to allow identification of RNA profiles (Fig. 54.2). Antigen detection tests, targeted on the middle-layer protein VP6, include enzyme-linked immunosorbent assays and immunochromatographic assays. The diagnosis of rotavirus infection can also be made by genome detection (RT-PCR), although such assays are much less frequently applied in the diagnostic laboratory than are rotavirus antigen detection tests.
Epidemiology
Rotavirus infections occur worldwide, but the vast majority of deaths occur in children in developing countries (Fig. 54.3). Thus, an estimated 2 million children under the age of 5 years die from diarrhoeal disease in developing countries each year, and rotavirus accounts for about 40% of these deaths (an estimated 527 000 rotavirus-associated deaths in 2004, the last year before the introduction of rotavirus vaccine). In industrialized countries rotavirus accounts for 40–50% of hospital admissions due to diarrhoeal disease. Most symptomatic infections are seen in children between 6 months and 2 years of age in industrialized countries, although in developing countries symptomatic rotavirus infection below 6 months of age is common. By the age of 5 years, virtually all children have been infected with rotavirus. In temperate countries rotavirus infections display marked seasonality, with distinct peaks during the winter months and few infections identified outside this period (Fig. 54.4). In contrast, rotavirus infections occur year-round in most tropical countries. Transmission of rotavirus within families to siblings and parents is well recognized. The release of enormous numbers of virions during the acute phase contributes to the easy transmission of the virus. Only a few virus particles are sufficient to cause disease in the susceptible host.
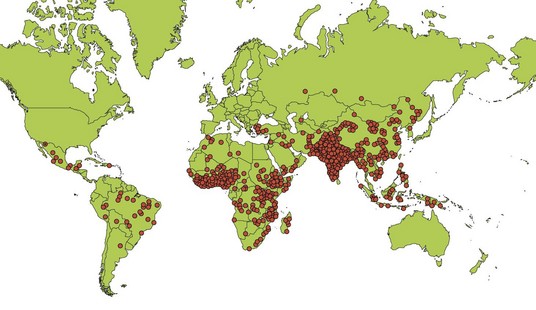
Fig. 54.3 Estimated distribution of deaths due to rotavirus disease among children < 5 years of age, by country. Each dot represents 1000 deaths.
(Adapted from Parashar UD, Burton A, Lanata C et al 2009 Global mortality associated with rotavirus disease among children in 2004. Journal of Infectious Diseases 200: S9–15.)
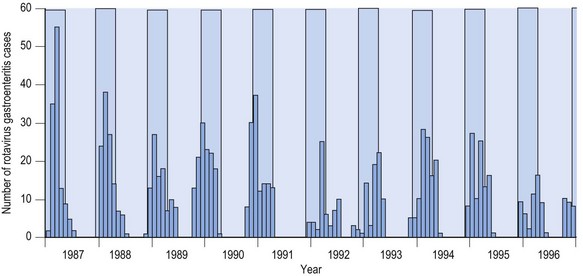
Fig. 54.4 Rotavirus gastroenteritis in children admitted to hospital in Akita, Japan, 1987–1996. The light blue shaded area represents the winter season (December to March).
Among group A rotaviruses, five genotype combinations comprise more than 85% of rotaviruses detected in humans, including P[8],G1, P[4],G2, P[8],G3, P[8],G4, and P[8],G9 (Fig. 54.5). However, strains with G12 emerged globally over the last decade, and G8 has been commonly reported in many African countries.
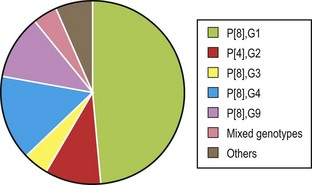
Fig. 54.5 Distribution of combined P and G types in Europe between 2006 and 2009.
(Adapted from Iturriza-Gómara M, Dallman T, Bányai K et al 2011 Rotavirus genotypes co-circulating in Europe between 2006 and 2009 as determined by EuroRotaNet, a pan-European collaborative strain surveillance network. Epidemiology and Infection 139: 895–909.)
Treatment
Although no specific anti-rotaviral treatment is available routinely, probiotic therapy (e.g. with Lactobacillus GG) has been shown in clinical trials to shorten the duration of symptoms of gastroenteritis. In selected clinical situations, anti-rotaviral immunoglobulin therapy has been used as prophylaxis against, and as treatment of, rotavirus gastroenteritis. However, the mainstay of therapy consists of oral rehydration with fluids of specified electrolyte and glucose composition (see Table 30.1, p. 318). Intravenous rehydration therapy is reserved for patients with severe dehydration, shock or reduced level of consciousness.
Control
Attention to hygienic measures such as handwashing, safe disposal of faeces and disinfection of contaminated surfaces is essential in reducing the risk of transmission. However, universal vaccination of infants is the most important preventive strategy. The rotavirus vaccines that have been developed are live-attenuated, orally administerable strains. Attenuation of virulence has been achieved either by repeated passage in cell culture or by substitution through genetic reassortment of serotype-determining gene segment(s) of a human rotavirus into the backbone of an animal rotavirus (which is both naturally attenuated for humans and attenuated through repeated cell culture passage).
The first licensed rotavirus vaccine, a Rhesus monkey rotavirus-based tetravalent human reassortant vaccine (RotaShield), was withdrawn after this live oral vaccine was associated with the development of intestinal intussusception in approximately 1 in 10 000 vaccine recipients in the USA. Two further live-attenuated oral rotavirus vaccines were developed and extensively evaluated, and have now entered childhood immunization programmes following recommendation by the World Health Organisation. These are the monovalent P1A[8]G1 human rotavirus vaccine Rotarix, and the pentavalent human–bovine reassortant rotavirus vaccine RotaTeq, which includes the world’s most common human serotypes G1, G2, G3, G4 and P[8] on a bovine rotavirus background. Both vaccines have demonstrated efficacy against severe rotavirus disease caused by globally common rotavirus strains. The administration of Rotarix (2 doses) and Rotateq (3 doses) needs to be completed by 32 weeks of age to minimize any potential risk of intusscusception. In the USA, the number of hospitalizations and emergency visits due to rotavirus infection has dramatically fallen following national introduction of rotavirus vaccination. The greatest potential for impact on child morbidity and mortality lies in the developing countries in Africa and Asia, where both vaccines have recently been evaluated. Although vaccine efficacy was lower than observed in industrialized settings, universal rotavirus vaccination in such countries would result in a large decrease in the number of severe rotavirus episodes because of the high incidence of disease.
Dormitzer PR. Rotaviruses. In: Mandell GL, Bennett JE, Dolin R. Principles and Practice of Infectious Diseases. ed 7. Philadelphia: Elsevier; 2010:2105–2115.
Estes M, Kapikian A. Rotaviruses. In: Knipe DM, Howley PM, Griffin DE, et al. Fields Virology. ed 5. Philadelphia: Kluwer Health/Lippincott, Williams and Wilkins; 2007:1917–1974.
Greenberg HB, Estes MK. Rotaviruses: from pathogenesis to vaccination. Gastroenterology. 2009;136:1939–1951.
Lopez S, Arias CF. Multistep entry of rotavirus into cells: a Versaillesque dance. Trends in Microbiology. 2004;12:271–278.
Matthijnssens J, Ciarlet M, Rahman M, et al. Recommendations for the classification of group A rotaviruses using all 11 genomic RNA segments. Archives of Virology. 2008;153:1621–1629.
Santosham M. Rotavirus vaccine – a powerful tool to combat deaths from diarrhea. New England Journal of Medicine. 2010;362:358–360.