62 Protozoa
Malaria; toxoplasmosis; cryptosporidiosis; amoebiasis; trypanosomiasis; leishmaniasis; giardiasis; trichomoniasis
Key points
• Protozoa are unicellular eukaryotic organisms.
• Most protozoal infections require laboratory confirmation for diagnosis; toxoplasmosis is diagnosed serologically.
• Malaria kills over 750 000 people each year.
• Acute malaria is a medical emergency demanding immediate diagnosis and treatment; quinine or artemisinin derivatives, usually in combination with other antimalarial drugs, are effective.
• African trypanosomiasis (sleeping sickness) is treated with toxic arsenical drugs; the West African form responds to eflornithine.
• South American trypanosomiasis (Chagas’ disease) is a chronic condition that is difficult to treat.
• Leishmaniasis takes various forms; the most dangerous is visceral leishmaniasis (kala azar). Antimonial compounds, antifungal drugs or miltefosine are used for treatment.
• Amoebiasis, giardiasis and trichomoniasis occur worldwide; they usually respond to 5-nitroimidazole drugs such as metronidazole.
Infection with pathogenic protozoa exacts an enormous toll of human suffering, notably, but not exclusively, in the tropics. Numerically the most important of the life-threatening protozoan diseases is malaria, which is responsible for at least 750 000 deaths a year, mostly in young children in Africa.
Pathogenic protozoan parasites are conveniently dealt with by placing them in four groups: sporozoa, amoebae, flagellates and a miscellaneous group of other protozoa that may cause human disease (Table 62.1).
Table 62.1 Principal protozoan pathogens of man
| Group | Species | Disease |
|---|---|---|
| Sporozoa | Plasmodium falciparum | Malignant tertian malaria |
| P. vivax | Benign tertian malaria | |
| P. ovale | Benign tertian malaria | |
| P. malariae | Quartan malaria | |
| Toxoplasma gondii | Toxoplasmosis | |
| Isospora belli | Diarrhoea | |
| Cryptosporidium parvum | Diarrhoea | |
| Cyclospora cayetanensis | Diarrhoea | |
| Amoebae | Entamoeba histolytica | Amoebic dysentery; liver abscess |
| Naegleria fowleria | Meningo-encephalitis | |
| Acanthamoeba spp.a | Keratitis | |
| Balamuthia mandrillarisa | Encephalitis | |
| Blastocystis hominisb | Pathogenicity doubtful | |
| Flagellates | Giardia lamblia | Diarrhoea, malabsorption |
| Trichomonas vaginalis | Vaginitis, urethritis | |
| Trypanosoma brucei gambiense | Sleeping sickness | |
| T. brucei rhodesiense | Sleeping sickness | |
| T. cruzi | Chagas’ disease | |
| Leishmania spp. | See Table 62.4 | |
| Others | Babesia microtia | Babesiosis |
| B. divergensa | Babesiosis | |
| Balantidium colia | Balantidial dysentery | |
| Encephalitozoon cuniculia | Microsporidiosis | |
| Enterocytozoon bieneusia | Microsporidiosis | |
| Nosema connoria | Microsporidiosis |
a Organisms rarely encountered in human disease.
Sporozoa
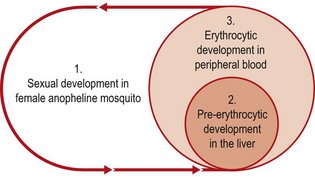
This group includes the malaria parasites and related coccidia, which exhibit a complex life cycle involving alternating cycles of asexual division (schizogony) and sexual development (sporogony). In malaria parasites, the sexual cycle takes place in the female anopheline mosquito (Fig. 62.1).
Malaria parasites
Description
Four species are commonly encountered in human disease: Plasmodium falciparum, which is responsible for most fatalities; P. vivax and P. ovale, both of which cause benign tertian malaria (febrile episodes typically occurring at 48-h intervals); and P. malariae, which causes quartan malaria (febrile episodes typically occurring at 72-h intervals). The appearances of trophozoites of the four species as seen in Romanowsky-stained films of peripheral blood are illustrated in Figures 62.2–62.7. P. knowlesi, a parasite of long-tailed macaque monkeys may also affect man. Human infection in south-east Asia, where the parasite is found, may be more common than has been supposed as the blood forms can be confused with P. falciparum or P. malariae.
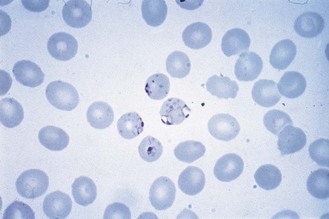
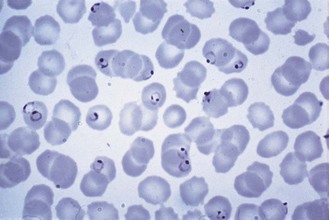
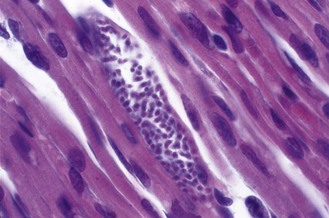
Fig. 62.4 Trophozoites of P. falciparum. Note peripheral location of the parasites (appliqué or accolé forms) and light stippling of the red cells (Maurer’s spots).
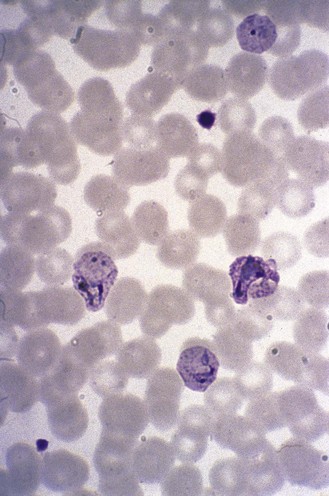
Fig. 62.5 Amoeboid trophozoites of P. vivax. Note the marked enlargement of the parasitized red cells and the intense stippling (Schüffner’s dots).
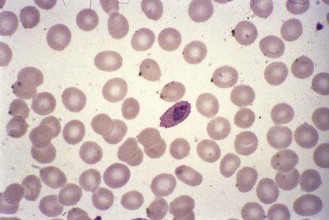
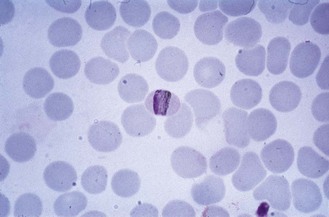
Fig. 62.6 Trophozoite of P. ovale. Note the fimbriate, oval-shaped red cell and marked stippling (James’ stippling).
Life cycle
When an infected mosquito bites, sporozoites present in the salivary glands enter the bloodstream and are carried to the liver, where they invade liver parenchyma cells. They undergo a process of multiple nuclear division, followed by cytoplasmic division (schizogony); when this is complete, the liver cell ruptures, releasing several thousand individual parasites (merozoites) into the bloodstream. The merozoites penetrate red blood cells and adopt a typical ‘signet-ring’ morphology (see Fig. 62.3).
In the case of P. vivax and P. ovale, some parasites in the liver remain dormant (hypnozoites) and the cycle of pre-erythrocytic schizogony is completed only after a long delay. Such parasites are responsible for the relapses of tertian malaria that may occur, usually within 2 years of the initial infection.
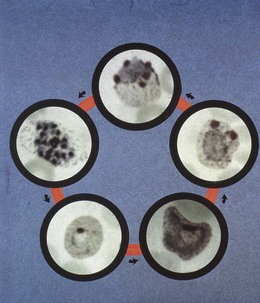
In the bloodstream, the young ring forms (trophozoites) develop and start to undergo nuclear division (erythrocytic schizogony). Depending on the species, about 8 to 24 nuclei are produced before cytoplasmic division occurs, and the red cell ruptures to release the individual merozoites, which then infect fresh red blood cells (see Fig. 62.2).
Instead of entering the cycle of erythrocytic schizogony, some merozoites develop within red cells into male or female gametocytes. These do not develop further in the human host, but when the insect vector ingests the blood, the nuclear material and cytoplasm of the male gametocytes differentiate to produce several individual gametes, which give it the appearance of a flagellate body (exflagellating male gametocyte). The gametes become detached and penetrate the female gametocyte, which elongates into a zygotic form, the ookinete. This penetrates the mid-gut wall of the mosquito and settles on the body cavity side as an oocyst, within which numerous sporozoites are formed. When mature, the oocyst ruptures, releasing the sporozoites into the body cavity, from where some find their way to the salivary glands.
P. falciparum differs from the other forms of malaria parasite in that developing erythrocytic schizonts form aggregates in the capillaries of the brain and other internal organs, so that normally only relatively young ring forms or gametocytes (which are typically crescent shaped) are found in peripheral blood.
The cycle of erythrocytic schizogony takes 48 h, except in the case of P. malariae, in which the cycle occupies 72 h. Since febrile episodes occur shortly after red cell rupture, this explains the characteristic periodic fevers. However, with P. falciparum, the cycles of different broods of parasite do not become synchronized as they do in other forms of malaria, and typical tertian fevers are not usual in falciparum malaria.
Laboratory diagnosis
Acute falciparum malaria is a medical emergency that demands immediate diagnosis and treatment. To establish the diagnosis, a drop of peripheral blood is spread on a glass slide. The smear should not be too thick; a useful criterion is that print should be just visible through it. The smear is allowed to dry thoroughly and stained by Field’s method. This is an aqueous Romanowsky stain, which stains the parasites very rapidly and haemolyses the red cells, so that the parasites are easy to detect despite the thickness of the film.
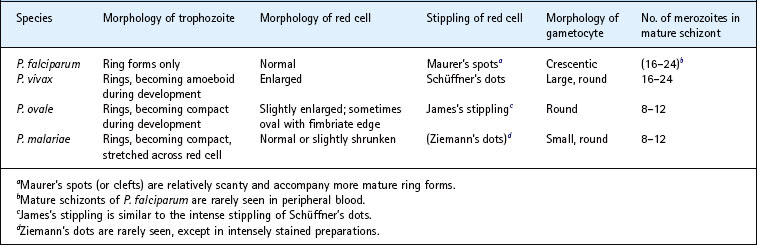
With experience, the species of malaria can usually be determined from a thick blood film, but some of the characteristic features used to establish the identity of the parasites (Table 62.2), such as the typical stippling of the red cell that accompanies infection with P. vivax or P. ovale, are better observed in a conventional thin blood film stained by one of the many modifications of Giemsa’s or Leishman’s stain. The water used to dilute the stain should be at pH 7.2 (not 6.8 as used for haematological purposes).
Table 62.2 Differential characteristics of human malaria parasites as seen in Romanowsky-stained thin films of peripheral blood (see also Figs 60.2–60.7)
Various other diagnostic tests have been devised for the rapid diagnosis of malaria including simple ‘dipstick’ tests, which are sufficiently reliable to be used by inexperienced staff (perhaps even by patients themselves) or in field conditions where microscopy is not available. Tests that detect P. falciparum alone or discriminate between P. falciparum and P. vivax are available. They are not foolproof, and may remain positive for some time after successful treatment. Visualization of the parasites in Romanowsky-stained peripheral blood films remains the most reliable method.
Pathogenesis
Malaria is characterized by severe chills, high fever and sweating, often accompanied by headache, muscle pains and vomiting. Falciparum malaria, unlike the other forms, may progress (especially in primary infections) to coma, convulsions and death. This condition, cerebral malaria, is associated with the adherence of parasitized red blood cells to the endothelium of brain capillaries. However, the precise mechanism for the pathological sequelae that follow, including the marked increase in the production of tumour necrosis factor that occurs, is not known with certainty.
Falciparum malaria is notoriously varied in its presentation, so that a definitive laboratory diagnosis is most important. Various systems may be affected. Severe anaemia and renal failure are common complications, and hypoglycaemia, pulmonary oedema and gastroenteritis may be present.
Individuals that are homozygous or heterozygous for the sickle cell gene have a much-reduced susceptibility to infection with P. falciparum, and this has provided a selective advantage for the maintenance of sickle cell disease in holo-endemic areas. Similarly, individuals whose red cells lack the antigen known as the Duffy factor are protected from infection with P. vivax. In parts of tropical Africa, where most of the population are Duffy factor negative, P. vivax is rare, although the related P. ovale is found, especially in West Africa.
Treatment
For many years the standard treatment for acute malaria was chloroquine. However, resistance to that drug in P. falciparum (and, less commonly, in P. vivax) is now widespread and other agents have to be used. Derivatives of artemisinin (a natural product from the plant Artemisia annua), including artemether and sodium artesunate, are quick acting and effective. They have become the drug of choice for treatment of uncomplicated falciparum malaria in endemic areas; combination therapy with other antimalarial agents that target different functions in the parasite are recommended.
Alternatively, quinine (or quinidine), which has been available for centuries in the form of cinchona bark, remains effective. Some antibiotics, including tetracyclines and clindamycin, exhibit antimalarial activity and are used as an adjunct to quinine therapy. Mefloquine and halofantrine are active against chloroquine-resistant strains, but resistance to them is increasing in prevalence and both are associated with occasional problems of toxicity. The combination of atovaquone and proguanil offers a safer alternative.
Treatment of acute malaria with chloroquine, quinine or other antimalarials will not eliminate parasites in the liver. For this purpose the 8-aminoquinoline drug primaquine is used. This agent carries the risk of precipitating haemolysis in individuals who are deficient in the enzyme glucose-6-phosphate dehydrogenase.
Prophylaxis
Antimalarial prophylaxis is essential for non-immune travellers to malarious areas. Chloroquine and the antifolate drugs pyrimethamine (often combined with sulfadoxine or dapsone) and proguanil were formerly used widely, but the widespread occurrence of resistance to these agents has made it difficult to offer definitive advice to travellers, particularly those going to regions in which P. falciparum is prevalent. Common recommendations include:
• the combination of atovaquone and proguanil daily
Others recommend daily doxycycline or even primaquine. Whatever prophylactic guidance is given, it should be combined with advice to bring any fever to medical attention; to avoid exposure to mosquito bites by wearing long clothing in the evening when the insects are most active; to use insect repellents; and to sleep under mosquito netting impregnated with insecticide.
Because parasites in the pre-erythrocytic stage of development escape the action of most prophylactic drugs, prophylaxis should continue for at least 4 weeks after leaving a malarious area. This will effectively prevent the development of falciparum malaria, although relapses of other types may occur up to 2 years after exposure.
An effective vaccine has long been sought, but is still awaited.
Coccidia
The coccidia are related to malaria parasites and share alternating sexual and asexual phases of development. They are not, however, transmitted by insects and infection is usually acquired by ingesting mature oocysts.
Toxoplasma gondii
This is a coccidian parasite of the intestinal tract of the cat that is transmissible to many other mammals. It occurs worldwide. Serological evidence suggests that human infection is common, presumably as a transient febrile illness or a subclinical attack. Occasionally, more severe infection occurs.
• Intra-uterine toxoplasmosis is an important cause of stillbirth and congenital abnormality.
• Ocular disease is a rare but serious condition.
• Cerebral toxoplasmosis sometimes occurs in immunocompromised patients, probably through reactivation of latent infection.
Mature oocysts excreted by infected cats contain two sporocysts, within which tachyzoites develop. On ingestion the tachyzoites pass to the bloodstream and lymphatics to invade macrophages, in which they multiply (Fig. 62.8). As the immune response develops, other cells are infected and tissue cysts containing slowly metabolizing bradyzoites are formed. Infection is acquired by ingestion of oocysts, or of tissue cysts in undercooked meat. Intra-uterine infection is acquired transplacentally.
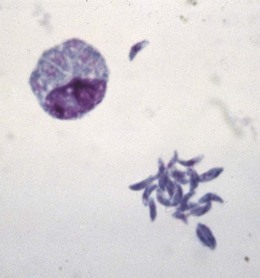
Fig. 62.8 Tachyzoites of Toxoplasma gondii in a macrophage (top left) and lying free (bottom right.)
It is difficult to find toxoplasmas in clinical material. Diagnosis of acute infection is made by demonstration of a rising titre of serum antibodies to Tox. gondii. The Sabin–Feldman dye exclusion test recognizes the ability of serum antibody to kill viable toxoplasmas. Other serological tests, including an enzyme-linked immunosorbent assay (ELISA), are available and have the advantage that they avoid the use of live toxoplasmas. The polymerase chain reaction (PCR) is useful in the diagnosis of intra-uterine and cerebral infections.
The combination of pyrimethamine and a sulphonamide is effective against active tachyzoites. Spiramycin is also effective and may be preferred during pregnancy. Clindamycin, azithromycin and atovaquone, usually in combination with pyrimethamine, offer alternatives in patients with cerebral toxoplasmosis.
Isospora belli
This coccidian parasite usually causes mild self-limiting diarrhoea, but is occasionally associated with more severe infection, particularly in patients with acquired immune deficiency syndrome (AIDS). It is more common in areas of poor sanitation. The characteristic oocysts can be seen in faecal ‘wet’ mounts, but are poorly refractile and easily missed. Co-trimoxazole is usually effective if antimicrobial treatment is necessary.
Cryptosporidium parvum
Cryptosporidia are common animal parasites. One species, Cryptosporidium parvum, commonly causes diarrhoea in humans, especially infants. Infection is usually water-borne or acquired directly from animals or infected cases. Large numbers of oocysts are often present in faeces; they are partially acid-fast and can be demonstrated by modifications of the Ziehl–Neelsen method (see p. 217).
The infection usually responds to symptomatic treatment, with fluid replacement if necessary. In severely immunocompromised patients, cryptosporidia may cause severe life-threatening diarrhoea for which nitazoxanide or macrolides such as azithromycin may offer some benefit.
Cyclospora cayetanensis
Unlike cryptosporidia, cyclospora develop intracellularly in the gut mucosa. The immature oocysts are excreted in the faeces as round bodies about 10 µm in diameter, with a characteristic ‘mulberry’ appearance. They are more variably acid-fast than are cryptosporidia.
Cyclospora cayetanensis causes diarrhoea and is associated with poor sanitation. As with other coccidian parasites, infection is more severe in the immunocompromised. Mild infection is treated symptomatically, with rehydration if necessary. Co-trimoxazole appears to be effective in serious infection.
Amoebae
Entamoeba histolytica
This is the most important amoebic parasite of man. The amoebae invade the colonic mucosa, producing characteristic ulcerative lesions and a profuse bloody diarrhoea (amoebic dysentery). Systemic infection may arise, leading to abscess formation in internal organs, notably the liver. Such disease may arise in the absence of frank dysentery.
Laboratory diagnosis
In acute amoebiasis, blood-stained mucus or colonic scrapings from ulcerated areas are examined by direct microscopy. The material should be examined within 2 h of collection. Entamoeba histolytica may be recognized by its active movement, pushing out fingerlike pseudopodia and, sometimes, progressing across the microscope field. If mucosal invasion has occurred, the amoebae usually contain ingested red blood cells, but these may be absent if infection is confined to the gut lumen. The nucleus is not usually visible in unstained ‘wet’ preparations, but in fixed smears stained with haematoxylin it is seen as a delicate ring of chromatin with a central karyosome (Fig. 62.9A).
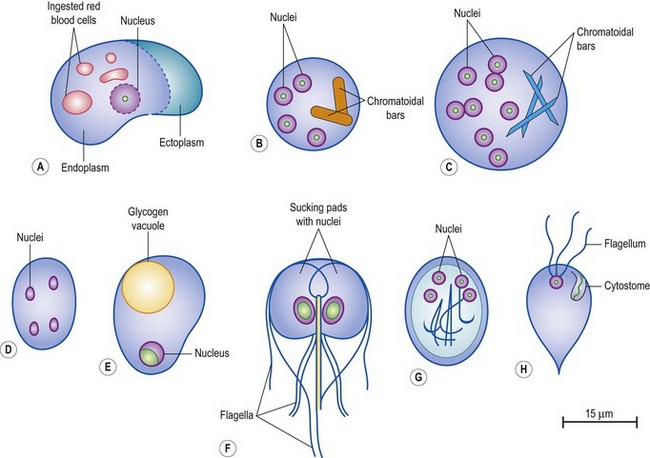
Fig. 62.9 Diagrammatic representation of some intestinal parasites: (A) Entamoeba histolytica, trophozoite with ingested red blood cells; (B) E. histolytica, mature cyst; (C) E. coli, mature cyst; (D) Endolimax nana, mature cyst; (E) Iodamoeba bütschlii, mature cyst; (F) Giardia lamblia, trophozoite; (G) G. lamblia, mature cyst; (H) Chilomastix mesnili, trophozoite.
Typical amoebic trophozoites may also be seen in aspirates of liver abscess. The pus often has a distinctive red–brown ‘anchovy sauce’ appearance. As the amoebae actively multiply in the walls of the abscess, they are most likely to be found in the last few drops of pus drained from the lesion.
In the intestinal carrier state, active amoebae are usually absent, but the encysted form, by which infection is spread, may be found. They are spherical, about 10–15 µm in diameter, and contain one to four of the nuclei typical of Entamoeba species – a circular ring with a central dot. Young uninucleate cysts may also contain a large glycogen vacuole and, in fresh specimens, cysts of all stages of development may exhibit one or more thick, blunt-ended chromatoidal bars (Fig. 62.9B).
Although demonstration of active amoebae or cysts is the best way to make a definitive diagnosis, serology is also sometimes helpful, particularly in systemic disease. Various immunodiagnostic tests have been described, but they are usually performed only in reference centres. Culture of E. histolytica is unhelpful as a diagnostic procedure.
Treatment
Metronidazole and tinidazole have superseded older drugs such as emetine and dehydroemetine for the treatment of amoebic dysentery and amoebic liver abscess.
Not all strains of E. histolytica are invasive and some (now classified as E. dispar) never cause disease. However, since cysts of pathogenic and non-pathogenic strains are indistinguishable, it is prudent to treat all asymptomatic cyst excreters, particularly if they are food handlers, at least in areas of the world in which amoebiasis is uncommon. For this purpose, diloxanide furoate is often used.
Non-pathogenic intestinal amoebae
Although there are occasional reports of diarrhoea associated with other intestinal amoebae, notably Dientamoeba fragilis (an amoeba flagellate) most are commensals and are important only because of potential confusion with E. histolytica. The differential characteristics of non-pathogenic intestinal amoebae are compared with those of E. histolytica in Table 62.3. The greatest opportunity for confusion arises with the other intestinal Entamoeba species, E. hartmanni and E. coli. E. hartmanni is morphologically identical to E. histolytica, but is smaller and the trophozoite never contains ingested red blood cells. E. coli is somewhat larger than E. histolytica, particularly in the cyst form. The trophozoites are more sluggish than those of E. histolytica, and mature cysts contain up to eight nuclei; chromatoidal bars, if present, are fine and pointed, rather like slivers of broken glass (Fig. 62.9C).
Blastocystis hominis, an organism commonly found in faeces and formerly thought to be a yeast, is probably an amoeba. Any pathogenic role is the subject of dispute, but it has been associated with diarrhoea in the absence of other known pathogens. Metronidazole is said to be useful if true infection is suspected.
Free-living amoebae
Environmental amoebae belonging to the genus Naegleria (usually N. fowleri) are occasionally implicated in meningo-encephalitis. Rare cases of granulomatous encephalitis caused by Balamuthia mandrillaris and Acanthamoeba species have also been described, often, but not exclusively, in immunocompromised patients. The outcome in all kinds of amoebic encephalitis is ordinarily fatal, although amphotericin B has been successfully used in infections with N. fowleri.
Acanthamoeba spp. more commonly cause keratitis, sometimes following use of contaminated cleaning fluids for soft contact lenses. Optimal antimicrobial chemotherapy remains to be defined, but topical treatment with propamidine, biguanides or voriconazole have been used.
Flagellates
Giardia lamblia (syn. G. intestinalis, G. duodenalis)
This intestinal parasite lives attached to the mucosal surface of the upper small intestine. Vast numbers may be present, and their presence may lead to malabsorption of fat and chronic diarrhoea. Young infants may be particularly severely affected. Infection is usually water-borne.
The trophozoite is kite-shaped, with two nucleated sucking pads and four pairs of flagella (Fig. 62.9F). Trophozoites may be found in duodenal aspirate, but examination of faeces usually reveals the cyst form by which the disease is transmitted. This is oval, about 10 × 8 µm, and contains up to four nuclei as well as the remains of the skeletal structure of the trophozoite (Fig. 62.9G).
Cysts of other, non-pathogenic, intestinal protozoa, including Chilomastix mesnili, Enteromonas hominis and Retortamonas intestinalis, may be mistaken for G. lamblia, but they are usually smaller and lack the regular oval shape and characteristic internal morphology. These non-pathogenic protozoa may also be found as trophozoites during microscopy of diarrhoeic faeces, but the most common intestinal flagellate is Trichomonas hominis, which is recognizable by its undulating membrane. There is no cyst form.
Giardiasis can be treated with 5-nitroimidazoles such as metronidazole or, on the rare occasions when this fails (and re-infection is excluded), with albendazole or mepacrine (quinacrine).
Trichomonas vaginalis
T. vaginalis is a flagellate protozoon with four anterior flagella and one lateral flagellum which is attached to the surface of the parasite to form an undulating membrane. There is no cyst form; the parasite is transmitted by sexual intercourse.
As the name suggests, T. vaginalis is predominantly a vaginal parasite, although urethritis may occur in the male consorts of infected women. The organism is responsible for a mild vaginitis, with discharge, which ordinarily responds to treatment with metronidazole or tinidazole.
T. vaginalis is readily identified by its characteristic motility in untreated ‘wet’ films of vaginal discharge and can be cultivated in appropriate culture media.
Trypanosomes
In contrast to the flagellates already described, trypanosomes have a complex life cycle involving an insect vector. The diseases that are caused in man, African trypanosomiasis (sleeping sickness) and South American trypanosomiasis (Chagas’ disease), are restricted in distribution according to the habitat of the insect host.
African trypanosomiasis
African sleeping sickness is caused by trypanosomes that are subspecies of Trypanosoma brucei, an important aetiological agent of the fatal disease nagana in cattle in tropical Africa. Tsetse flies (see p. 672) act as the insect vector. The human parasites are T. brucei gambiense, which occurs in riverine areas of west and central Africa, and T. brucei rhodesiense, a parasite of the savannah plains of east Africa, where cattle and wild antelope act as reservoirs of infection.
Pathogenesis
Following the bite of an infected tsetse fly, a localized trypanosomal chancre may appear transiently, but invasion of the bloodstream rapidly occurs. The parasites multiply in blood, and at this stage there may be non-specific symptoms with occasional febrile episodes and some lymphadenitis. Swollen lymph glands in the posterior triangle of the neck (Winterbottom’s sign) are often present in T. brucei gambiense infection. If untreated, the disease progresses inexorably to involve the central nervous system with the classical signs of sleeping sickness and, ultimately, death.
Infection with T. brucei rhodesiense tends to follow a more acute, fulminating course over a period of a few months, whereas T. brucei gambiense infection usually progresses slowly, sometimes over several years.
Laboratory diagnosis
During the parasitaemic stage, sparse trypanosomes may be found in peripheral blood in unstained ‘wet’ mounts or in smears stained by the Giemsa or Leishman methods. Examination of lymph node exudate may be helpful. Once the disease has progressed to involve the central nervous system, examination of cerebrospinal fluid reveals a lymphocytic exudate, often with morula cells (plasma cells) and sparse motile trypanosomes.
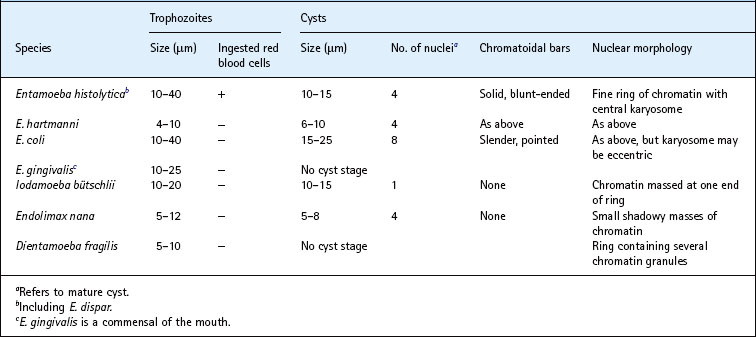
The parasites have a characteristic morphology: they are elongated, about 20–30 µm in length, with a single anterior flagellum arising via an undulating membrane from a basal body situated near a posteriorly placed kinetoplast (Figs 62.10 & 62.11).
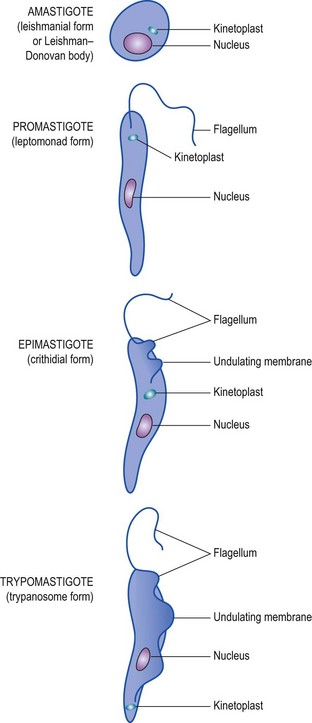
Fig. 62.11 Forms that may be adopted by Leishmania and Trypanosoma spp. In parentheses are given the terms for the forms under the old nomenclature now superseded.
In-vitro cultivation is unreliable, but animal inoculation is sometimes useful, particularly with T. brucei rhodesiense, which infects laboratory mice more readily than T. brucei gambiense. Various immunodiagnostic tests have been described, but they are not as reliable as direct microscopy in establishing a definitive diagnosis.
Treatment
In the early parasitaemic stage, the infection is amenable to treatment with suramin or pentamidine, but once the disease has progressed to sleeping sickness the trivalent arsenicals, melarsoprol or tryparsamide, are used. Less toxic alternatives are clearly required; eflornithine is effective in T. brucei gambiense infections, but not in disease caused by T. brucei rhodesiense.
South American trypanosomiasis
Chagas’ disease, caused by T. cruzi, is quite different from African trypanosomiasis. The insect vectors are various species of reduviid bugs (see p. 669). The trypanosomes are present in the bug’s faeces, which the unwitting sleeper rubs into the bite wound. They do not multiply in the bloodstream, but invade cells of the reticulo-endothelial system and muscle, where they lose their flagellum and associated undulating membrane and adopt a more rounded shape (Fig. 62.11). This morphological form is called an amastigote, and suggests a phylogenetic relationship with Leishmania species (see below). The amastigotes multiply in muscle and are liberated from ruptured cells as trypanosomal forms (trypomastigotes), which disseminate the infection and provide the parasitaemia needed to infect fresh reduviid bugs when they next feed.
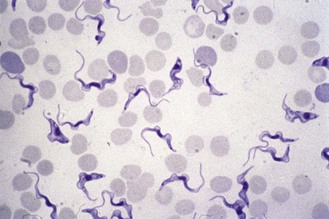
Trypomastigotes are shorter than those of the T. brucei group and have a characteristically large kinetoplast (Fig. 62.12). The appearance of amastigotes in heart muscle is shown in Figure 62.13.
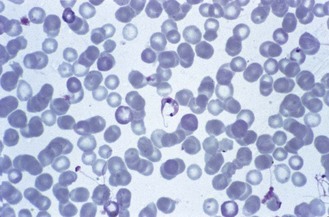
Fig. 62.12 Trypomastigote of T. cruzi in blood. Note the prominent kinetoplast and the ‘C’ shape adopted by the parasite.
Pathogenesis
Chagas’ disease is a chronic condition characterized by extensive cardiomyopathy, sometimes with gross distension of other organs (e.g. mega-oesophagus and megacolon). Death is usually from heart failure.
Laboratory diagnosis
PCR methods and various immunoassays have been described for serological diagnosis and are now preferred to older tests. These include: direct microscopy of peripheral blood, in which small numbers of trypomastigotes may be found; culture in the rich blood agar medium also used to isolate leishmania (see below); mouse inoculation; and xenodiagnosis, in which uninfected reduviid bugs are allowed to feed on the patient and after about 3–4 weeks the gut contents of the bug are examined for trypanosomes.
Leishmania species
Leishmania species are intracellular parasites of the reticulo-endothelial system. They are related to trypanosomes, but exist in only two morphological forms: amastigotes (non-flagellate forms), which occur in the infected lesion, and promastigotes (flagellate forms that lack an undulating membrane), which occur in the insect vector or in laboratory culture (Figs 62.11, 62.14 & 62.15).
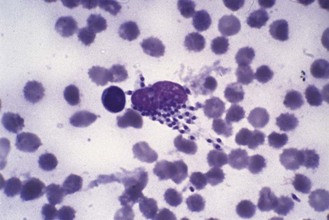
Fig. 62.14 Amastigotes of Leishmania tropica in a ruptured macrophage from a cutaneous lesion (oriental sore).
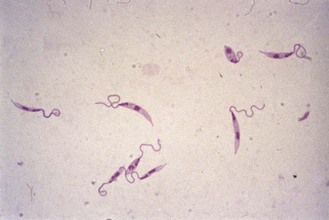
Fig. 62.15 Promastigotes of L. tropica from laboratory culture in Novy, MacNeal and Nicolle’s (NNN) medium.
The parasites are transmitted by sandflies (see p. 671) in various parts of the world, including the Middle East, India, South America, the Mediterranean littoral and parts of Africa.
Pathogenesis
Several distinct types of disease are recognized (Table 62.4), although they are caused by morphologically identical parasites. Cutaneous leishmaniasis (oriental sore) is the least troublesome, causing a boil-like swelling on the face or other exposed part of the body. The central part of the lesion may become secondarily infected with bacteria, but the leishmania organisms reside in the raised, indurated edge of the lesion. The sore usually heals spontaneously, leaving a scar, but with some species a more severe disseminated cutaneous leishmaniasis may occur. Parasites of the Leishmania mexicana complex may cause a destructive lesion of the outer ear (Chiclero’s ulcer).
Table 62.4 Leishmania species involved in human disease
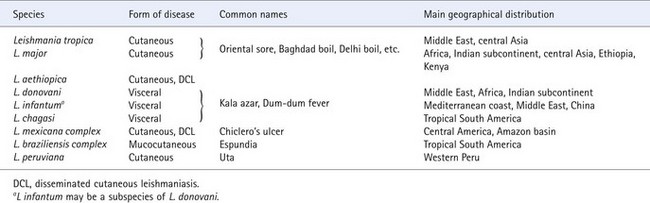
In mucocutaneous leishmaniasis (espundia), which is associated with the L. braziliensis complex, disfiguring lesions of the mouth and nose may be caused. However, the most serious form of leishmaniasis is visceral leishmaniasis (kala azar), which is a life-threatening disease involving the whole of the reticulo-endothelial system. There are estimated to be around 500 000 cases a year in the world, with the greatest burden in north east India and Bangladesh. A late complication of kala azar, post-kala azar dermal leishmaniasis, may be confused with leprosy or other skin conditions.
Laboratory diagnosis
In the cutaneous or mucocutaneous form of the disease, typical intracellular amastigotes may be recognized in Giemsa-stained smears of material obtained from tissues at the margin of the lesion (Fig. 62.14). Free amastigotes are commonly seen because of rupture of the macrophage host cell. Material should also be cultured in Novy, MacNeal and Nicolle’s (NNN) medium or a modification thereof. This is a rabbit blood agar containing antibiotic to prevent bacterial contamination and a buffered salt overlay solution in which the parasites grow as promastigotes (Fig. 62.15). Incubation is maintained for up to 3 weeks at room temperature (not 37°C).
In kala azar, spleen puncture is the most reliable method of diagnosis, but sternal marrow aspirate is safer and is usually preferred. Smears and cultures are made and examined as for cutaneous leishmaniasis. Various serological tests have been designed, but demonstration of the parasite by microscopy or culture is preferable whenever possible.
Treatment
The pentavalent antimony compounds, sodium stibogluconate and meglumine antimoniate, have been used traditionally in all forms of leishmaniasis, but they are toxic and therapy often fails. Amphotericin B is effective, but poorly tolerated, and the less toxic lipid-based formulations are preferred. Antifungal azoles and paromomycin (aminosidine) have been used with some success in cutaneous forms of disease. A phosphocholine derivative, miltefosine, offers considerable promise in kala azar.
Other pathogenic protozoa
Babesia species
Babesiae are predominantly animal parasites related to the piroplasmas that cause theileriasis in wild and domestic animals in many parts of the world. They are intracellular parasites living within red blood cells and are transmitted by ixodid ticks (see p. 673–4). In stained blood films they superficially resemble young ring forms of plasmodia.
Human infection is uncommon. European cases have mostly been in patients whose resistance was impaired by lack of a functioning spleen; the causative parasite was usually Babesia divergens. In contrast, babesiosis caused by B. microti has been reported in otherwise healthy persons in parts of the USA.
In immunocompetent individuals the disease is usually self-limiting, so that specific treatment is not required. Optimal treatment for more serious cases has not been properly defined, but the combination of quinine with clindamycin has been used successfully.
Balantidium coli
The ciliate protozoon Balantidium coli is a common parasite of the pig, and human infections have usually been traced to contact with these animals. The infective form is a large (about 50 µm in diameter) thick-walled cyst. The trophozoite inhabits the lumen of the gut and may attack the colonic mucosa in much the same way as E. histolytica, to cause balantidial dysentery. Many highly motile ciliate trophozoites are readily seen in untreated ‘wet’ films of diarrhoeic faeces. Tetracyclines and metronidazole are said to offer effective treatment.
Microsporidia
These animal and insect parasites are now thought to be fungi and are represented by several genera, including Encephalitozoon, Enterocytozoon and Nosema. They are, on rare occasions, implicated in opportunistic infections of immunocompromised patients, especially those with AIDS. Infections of the eye, meninges and other organs have been reported. Albendazole may be useful in treatment, but is not always curative.
Cook GC, Zumla A. Manson’s Tropical Diseases, ed 22, London: Elsevier: Saunders, 2008.
Croft SL. Kinetoplastida: new therapeutic strategies. Parasite. 2008;15:522–527.
Farthing MJ. Treatment options for the eradication of intestinal protozoa. Nature Clinical Practice. Gastroenterology & Hepatology. 2006;3:436–445.
Greenwood BM, Bojang K, Whitty CJM, Targett GAT. Malaria. Lancet. 2005;365:1487–1498.
Guerrant RL, Walker DH, Weller PF. Tropical Infectious Diseases, ed 3. Philadelphia: Elsevier: Saunders; 2011.
Jacquerioz FA, Croft AM: Drugs for preventing malaria in travellers, Cochrane Database Systematic Review 4:CD006491, 2009.
Nosten F, White NJ. Artemisinin-based combination treatment of falciparum malaria. American Journal of Tropical Medicine and Hygiene. 2007;77:181–192.
Peters W, Pasvol G. Atlas of Tropical Medicine and Parasitology, ed 6. London: Elsevier: Mosby; 2006.
Rosenblatt JE. Laboratory diagnosis of infections due to blood and tissue parasites. Clinical Infectious Diseases. 2009;49:1103–1108.
Stanley SL. Amoebiasis. Lancet. 2003;361:1025–1034.
World Health Organization. Basic Laboratory Methods in Medical Parasitology. Geneva: WHO; 1991.
Wellcome Trust: 2007 Malaria, ed 3, (Topics in International Health series).
Wellcome Trust: 2007 Human African Trypanosomiasis (Topics in International Health series).
Wellcome Trust: 2000 Leishmaniasis (Topics in International Health series).
The above CD-ROMs are available from Wellcome Trust, London (http://www.wellcome.ac.uk/tih/) and (for developing countries) from TALC (Teaching-aids at Low Cost; http://www.talcuk.org/)
Centers for Disease Control and Prevention. Division of Parasitic Diseases, Professional Information. http://www.cdc.gov/parasites.
Malaria Centre, London School of Hygiene and Tropical Medicine. http://malaria.lshtm.ac.uk/.
National Travel Health Network and Centre. http://www.nathnac.org/travel/index.htm.
World Health Organization. Basic Laboratory Methods in Medical Parasitology. http://whqlibdoc.who.int/publications/9241544104_%28part1%29.pdf.
World Health Organization. Malaria Rapid Diagnostic Tests. http://www.wpro.who.int/sites/rdt/home.htm.
World Health Organization. World Malaria Report. http://www.who.int/malaria/world_malaria_report_2010/worldmalariareport2010.pdf, 2010.