Mycoplasma Bovis
Definition and Etiology
Mycoplasma bovis is a member of the genus Mycoplasma of the class Mollicutes, and as such it is among the smallest free living organisms. The first known mycoplasma, identified in 1898, was the causative agent of contagious bovine pleuropneumonia, Mycoplasma mycoides subsp. mycoides, small colony variant.353 Mycoplasmas have since been recognized to cause disease in humans and a wide variety of animal and plant hosts. Mycoplasmas are small (approximately 200 nm) and pleomorphic, with a genome of only approximately 500 to 1000 kD.354 They are bounded by a single membrane, and because they lack a cell wall they are naturally resistant to antimicrobials such as penicillins and cephalosporins that work by impairing cell wall synthesis. Mycoplasma bovis was first identified in 1961 in the United States, where it was isolated from a case of mastitis.355 It has since spread worldwide. The bacterium was originally considered a subspecies of Mycoplasma agalactiae, and the two agents can be difficult to distinguish.356 However, M agalactiae is a pathogen of sheep and goats and is rarely isolated in the United States, whereas Mycoplasma bovis is regularly isolated in cases of pneumonia, respiratory disease, arthritis, tenosynovitis, and other disorders of cattle. Mycoplasma bovis can cause disease in sheep, goats, and other species, but this is a rare occurrence.
Clinical Signs
Respiratory infection with Mycoplasma bovis causes fever, tachypnea, inappetence, and sometimes respiratory distress.357 Coughing and nasal discharge are reported in some outbreaks.358,359 Respiratory disease caused by Mycoplasma bovis can occur in outbreaks; in young dairy calves, a subset of calves affected often develops otitis, characterized by unilateral or bilateral drooping of ears with purulent aural discharge, possibly with facial paralysis caused by cranial nerve VII involvement, and vestibular signs such as head tilt, nystagmus, and ataxia.358-361 In weaned beef calves and cattle entering feedlots, a subset of affected animals may develop arthritis and tenosynovitis361-363; this syndrome is sometimes referred to as chronic pneumonia and polyarthritis syndrome (CPPS).363,364 Young dairy calves can also develop arthritis or tenosynovitis.359 A typical complaint by the producer experiencing a respiratory disease outbreak involving Mycoplasma bovis is that cattle do not respond to therapy as expected, and a significant proportion of affected animals remain chronically ill and unthrifty for weeks after the onset of disease.361,362,365 In addition to respiratory disease, Mycoplasma bovis can also cause mastitis, arthritis and tenosynovitis, conjunctivitis, otitis, sinusitis, and myocarditis and/or pericarditis. The bacteria can also be isolated from aborted fetuses and from semen, linking the agent to reproductive failure.
The ability of Mycoplasma bovis to act as a primary respiratory pathogen has been debated. Classically the agent has been understood to be an opportunist, establishing itself after primary infection with viral pathogens or other bacteria. However, experimental challenge of gnotobiotic calves with Mycoplasma bovis alone induced respiratory disease with clinical signs of fever, tachypnea, and inappetence in four of seven calves and grossly evident lung lesions in all seven.357 These findings indicate that Mycoplasma bovis can cause disease while acting alone, but, as for other bacterial respiratory pathogens described earlier, it is likely that natural disease often follows other primary insult. A recent survey of causes of death in cattle in Ontario feedlots identified caseonecrotic pneumonia caused by Mycoplasma bovis as a cause of more fatalities within the first 60 days of the feeding period than acute fibrinosuppurative pneumonia typical of disease caused by M. haemolytica or H. somnus.188 This study indicates that Mycoplasma bovis is an important contributor to mortality in some feedlots, but more research is needed to determine whether the factors that predispose cattle to fatal disease resulting from Mycoplasma bovis are different from the factors known to predispose cattle to fibrinous bronchopneumonia caused by M. haemolytica.
Pathogenesis
Although Mycoplasma bovis has been recognized to cause respiratory disease in cattle for some time, very little is known about the mechanisms by which the agent causes disease. In disease caused by other mycoplasmas, attachment has been shown to be a key factor in pathogenesis. In vitro studies of Mycoplasma bovis suggest that pathogenicity is likewise associated with the ability to attach, as isolates from cases of clinical disease were better able to attach to embryonic bovine lung cells than were isolates from asymptomatic animals or high passage laboratory isolates.366 Antibodies against several of the variable surface proteins (Vsps) expressed by Mycoplasma bovis were able to partially but not completely block attachment, indicating that the Vsps play a role in attachment.366 Invasiveness is another pathogenic mechanism of Mycoplasma bovis; Mycoplasma bovis was found to be capable of migration between ciliated respiratory epithelial cells, whereas Mycoplasma dispar, another mycoplasma commonly isolated from cases of bovine pneumonia, remained attached to the surface of ciliated epithelial cells.367 The ability of Mycoplasma bovis to invade allows the organism to cause disease in organs outside the respiratory tract, such as joints and tendon sheaths. Mycoplasma bovis produces a toxin that increases vascular permeability.368 Certain strains of the agent have also been shown to be cyotoxic to mammalian cells369; it is not known whether this effect is related to the vascular toxin reported by Geary and colleagues.
Available data indicate that Mycoplasma bovis may cause disease in part through evading or impairing host immune function. Although not yet well characterized, a feature of Mycoplasma bovis that is likely important in enabling the bacteria to escape the host immune response is the expression of Vsps. The ability to vary surface protein expression has been shown to be an important pathogenic mechanism in certain mycoplasmas.354Mycoplasma bovis expresses at least three Vsps—VspA, VspB, and VspC—and isolates have been found that express some or all of these proteins.370 A study of 50 Mycoplasma bovis isolates, including many field isolates, showed extensive variability in Vsps at both the genetic and antigenic level.
Evidence indicates that Mycoplasma bovis can directly impair the activity of neutrophils. In one study Mycoplasma bovis was found to adhere to bovine neutrophils, but adherence did not elicit an expected activation response. Moreover, adherent Mycoplasma bovis inhibited normal neutrophil microbicidal activity.371 The bacteria is also able to kill lymphocytes by inducing them to undergo apoptosis (programmed cell death).372 Induction of proinflammatory cytokine production by the host may also contribute to disease caused by Mycoplasma bovis; an isolate of Mycoplasma bovis induced production of the proinflammatory cytokine TNF-α by bovine alveolar macrophages to a degree similar to that induced by M. mycoides subsp. mycoides (the cause of contagious bovine pleuropneumonia, a serious disease exotic to the United States) but in contrast to nonpathogenic mycoplasmas tested.373
Other research suggests that Mycoplasma bovis may induce an immune response that is not optimally protective. Vanden Bush and Rosenbusch showed that in calves experimentally infected with Mycoplasma bovis, serum titers of Mycoplasma bovis-specific IgG1 increased significantly after infection, whereas titers of antigen-specific IgG2 did not increase as markedly.374 Because IgG2 is considered superior in opsonizing ability as compared with IgG1, the authors speculated that preferential induction of IgG1 by Mycoplasma bovis infection may be related to the apparent inability of the immune response to rapidly clear the organism, as evidenced by the common association of chronic pneumonia with Mycoplasma bovis infection.
Epidemiology
Mycoplasma bovis can be isolated from the respiratory tracts of normal cattle188,375,376 and from cattle with respiratory disease.208,365,377-380 Surveys of nasopharyngeal swabs taken from dairy calves with no clinical signs of respiratory disease found Mycoplasma bovis in 0% to 34% of the animals sampled.376,380,381 A recent survey of multiple source weaned beef calves sampled soon after arrival at nine different backgrounding or stocker operations identified nasal shedding of Mycoplasma bovis in 0% to 6% of animals at each operation.382Mycoplasma bovis can also be found in bovine lungs without evidence of disease at postmortem examination.188 The fact that Mycoplasma bovis can be isolated from animals with no clinical or pathologic signs of pneumonia has led some to question whether the agent is a true respiratory pathogen. However, a consistent association of Mycoplasma bovis with a clinical syndrome of chronic nonresponsive pneumonia with or without otitis, arthritis, or tenosynovitis,* and a pathologic syndrome of bronchopneumonia with multifocal caseous necrosis visible histologically and/or grossly,362,364 has led to general acceptance that Mycoplasma bovis can contribute to significant morbidity and mortality in some situations.
Although more research is needed, currently available data support the concept that Mycoplasma bovis can spread from a few animals carrying the bacteria to others until a large proportion of a group is infected; transmission in these cases is most likely via respiratory infection by direct contact or short-distance aerosol.361,362,375 Feeding milk infected with Mycoplasma bovis appears to be another important source of infection for dairy calves.359-361 The fact that Mycoplasma bovis infection can be widespread in some populations was illustrated by a case-control study of cattle in the first 4 weeks after feedlot entry. Researchers used a protected BAL catheter to collect samples from the lower airways with minimal contamination from the nasal passages. Soon after arrival, these investigators found Mycoplasma bovis in the BALF of 52% of controls that were not treated for respiratory disease in the first 28 days, versus 61% of cases that were treated for respiratory disease within 28 days.299 Two weeks later Mycoplasma bovis was found in BALF from over 80% of the controls and in 100% of animals who ultimately were treated for respiratory diseases by 28 days, indicating that colonization of the respiratory tract of all cattle sampled was widespread within a short period after feedlot entry.375 Genetic characterization by arbitrarily primed PCR (AP-PCR) has been used to characterize M. bovis isolates associated with herd outbreaks of disease, with a single genetic lineage associated with disease in a closed herd, and multiple genetic lineages associated with disease in a calf ranch where animals were frequently brought on site from multiple sources.384
Mycoplasma bovis has been isolated from both dairy calves and feedlot cattle with respiratory disease in multiple studies,* with particularly high prevalence occurring in animals with chronic pneumonia nonresponsive to antimicrobial therapy.208,209,365,377 In a recent survey of cattle subjected to necropsy within 60 days of arrival at 72 feedlots, lung lesions were categorized as either fibrinosuppurative (typical lesion caused by M. haemolytica or H. somni) or caseonecrotic, which were characterized grossly by the presence of multiple foci of dry caseous material and histologically by areas of eosinophilic necrotic cellular debris (caseous necrosis) surrounded by inflammatory cells and fibrous tissue. Mycoplasma bovis was isolated in 53 of 54 cases (98%) with caseonecrotic lesions and in 52 of 58 of cases (89%) with fibrinosuppurative lesions; it was also isolated from 6 of 13 animals (46%) with normal lungs in this study.188 In 49 chronically sick cattle from a single feedlot that were subjected to necropsy in 1 month, Mycoplasma bovis was identified in the lungs of 82% of the cases, and in the joints of 45% of the cases.209 BVDV was also present in 39% of the cases from which Mycoplasma bovis was isolated. A later retrospective study by the same authors found BVDV in the lungs of 44% of cattle subjected to necropsy with a final diagnosis of pneumonia caused by Mycoplasma bovis; these data lead the authors to suggest that immunosuppression resulting from BVDV may predispose a subset of animals to chronic pneumonia and/or arthritis caused by Mycoplasma bovis. In contrast, Gagea and colleagues found that although BVDV infection was more common in feedlot cattle with bacterial pneumonia than in those with other diseases, it was not more common in cattle with caseonecrotic lesions typical of Mycoplasma bovis infection.379
Necropsy Findings
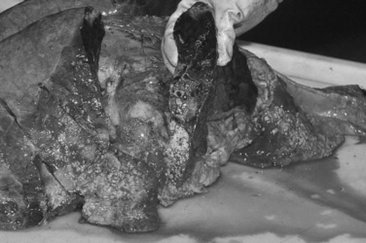
Grossly, lungs of cattle with pneumonia caused by Mycoplasma bovis have dark red, firm consolidated lobules of the cranioventral lung. Raised white to yellow, firm nodules that range from 0.5 to several centimeters in diameter are often but not invariably seen clustering in the cranioventral lung (Figs. 31-60 and 31-61).357,364,379,385 These nodules may appear to be abscesses, but in most cases they are actually foci of coagulation necrosis.357,379 Differential diagnoses for gross lesions of abscessing pneumonia in cattle include infection with Arcanobacterium (Actinomyces) pyogenes and F. necrophorum (both of which are more likely to cause a foul odor than Mycoplasma bovis) and Mycobacterium bovis (tuberculosis).364 Other gross lesions include enlargement of interlobular septa by edema and fibrin; fibrinous or fibrous pleural adhesions are unusual but could be present if the animal is or has been co-infected with M. haemolytica or H. somnus. Animals with pneumonia typical of Mycoplasma bovis may also have arthritis in one or more joints characterized by abundant yellow fluid with fibrin and sometimes purulent material, and tenosynovitis characterized by extensive caseous exudate and pyogranuloma formation in the tendon sheaths.362,364 Fibrin, purulent material, and caseous material may be present in the tympanic bullae.
Histologically there is purulent pneumonia and bronchiolitis, with extensive infiltration of neutrophils in airways, and peribronchiolar cuffing with lymphocytes and mononuclear cells. Foci of eosinophilic coagulation necrosis are surrounded by a rim of dark pyknotic inflammatory cells and, farther out, a region of primarily macrophages and some plasma cells.357,364 Areas of coagulation necrosis may extensive, and ghostlike outlines of cells, alveolar walls, and alveoli may be seen.357,364 Immunohistochemical staining for the organism often shows large numbers surrounding the periphery of areas of coagulation necrosis, as well as in association with bronchiolar and alveolar epithelial cells.364,385 Histologic evaluation of joints, tendon sheaths, and the tympanic bullae and petrous temporal bone will reveal changes consistent with the gross lesions. Close inspection may also reveal foci of mononuclear cell accumulation with positive staining for Mycoplasma bovis by IHC in the liver and kidney385 or in the pericardium.347
Diagnosis
Confirmation of disease caused by Mycoplasma bovis is best made by identification of the agent by culture or IHC in conjunction with gross and histopathologic lesions typical of disease attributed to the agent. Thus, in order to reliably confirm a role for Mycoplasma bovis in an outbreak of bovine respiratory disease, postmortem of representative affected cases is necessary. It is important to request that the diagnostic laboratory specifically identify Mycoplasma bovis; in many cases diagnostic laboratories characterize agents only to the level of the genus by identification of typical organisms in culture. Because other mycoplasma such as Mycoplasma bovirhinis, M. dispar, and Mycoplasma alkalescens can also be isolated from the respiratory tract of cattle with pneumonia, a report of “Mycoplasma sp.” is not equivalent to a diagnosis of disease caused by Mycoplasma bovis. Species-specific identification of Mycoplasma bovis can be made by antibody-based tests such as IHC or immunofluorescence (available at the Iowa State University Veterinary Diagnostic Laboratory, and possibly others) or PCR (available at many laboratories including the University of Georgia Athens Veterinary Diagnostic Laboratory).
Although available data suggest that Mycoplasma bovis is less commonly isolated from healthy cattle than other mycoplasmas are, because the organism can be found in nasal swabs and lung tissue of apparently normal animals, an isolation of the bacteria in the absence of evidence of disease is of uncertain significance. Nasal shedding has not been confirmed to be a reliable antemortem measure of the presence of pneumonia caused by Mycoplasma bovis; the agreement between the results of nasal swab culture and the presence of lung disease caused by Mycoplasma bovis was good in one study378 and only moderate to poor in two other studies.299,386 Serologic assays have been used to characterize seroconversion in epidemiologic studies, but the tests are not widely available, and their results have not been shown to reliably indicate active infection.369 A recent report evaluated the agreement between the measurement of serum antibodies using a blocking ELISA and shedding of Mycoplasma bovis in the milk in a small number of cows; agreement was significant but only moderate (κ = 0.44).387 If serology is attempted, paired serology with evidence of rising titers in association with outbreaks of respiratory and/or joint disease is likely to be more useful than measurement of titers on a single occasion.
Treatment and Prevention
Multiple authors report that cattle with pneumonia or arthritis caused by Mycoplasma bovis respond poorly to antimicrobial therapy.209,362,363,369 Early studies indicated that tilmicosin had good effect against experimentally induced388 disease caused by Mycoplasma bovis in cattle; however, recent studies of in vitro susceptibility profiles of panels of Mycoplasma bovis isolates reveal evidence of widespread evidence of resistance to tilmicosin, as well as erythromycin, ampicillin, and ceftiofur.389,390 Susceptibility to oxytetracycline, chlortetracycline, and spectinomycin has been variable, although most isolates have been susceptible to fluoroquinolones such as enrofloxacin or danofloxacin.390-392 However, no fluoroquinolone marketed in the United States is specifically labeled for treatment of cattle for disease caused by Mycoplasma bovis, and extralabel use of fluoroquinolones is illegal. A relatively new antimicrobial, tulathromycin, has shown good efficacy against experimentally induced Mycoplasma bovis infection; it is interesting to note that the response did not correlate with in vitro MIC data for the challenge isolate, which was high and predicted that the antimicrobial would not likely be effective.393 Tulathromycin (Draxxin, Pfizer Animal Health) has recently been approved for treatment in the United States of respiratory disease caused by Mycoplasma bovis.
Although clinical trials and data from well-controlled experimental studies under conditions typical of U.S. cattle operations are lacking, reports suggest that in vitro MIC data may not always predict antimicrobial efficacy in the field.369,394 If in vitro susceptibility tests do indeed fail to predict clinical efficacy, the reason for this failure is not certain. Because Mycoplasma bovis can be seen in association with foci of coagulation necrosis in lungs of affected cattle,364,379,385 it may be that antimicrobials are not maximally effective in this environment, or it may be that insufficient duration of treatment is carried out in at least some cases. Controlled trials indicating the duration of antimicrobial therapy needed for effective treatment of pneumonia caused by Mycoplasma bovis are lacking, but anecdotal reports suggest that early treatment is critical for success,359,395 and that treatment should be continued for at least 7 to 10 days.359 The high concentrations of tulathromycin found in bovine lung for 7 to 15 days after SC injection393 make this a logical choice for prolonged treatment; a form of oxytetracycline that provides therapeutic blood levels for 7 days (Tetradure, Merial) may also be a good choice for treatment of some cases of disease caused by Mycoplasma bovis. Failure of antimicrobials that are predicted to be efficacious against Mycoplasma bovis may also be related to inability of the immune response to effectively clear the organism by mechanisms described previously. Some veterinarians report that outbreaks of Mycoplasma bovis cannot be stopped with any treatment, but that outbreaks are eventually terminated when chronically affected animals die or are shipped to slaughter.
Research indicates that vaccination can afford some protection against experimental challenge with Mycoplasma bovis.396,397 Autogenous vaccines have been used for years, but no reports with large numbers of animals and appropriate controls are available to provide information about efficacy. Recently two Mycoplasma bovis vaccines have been licensed for sale in the United States. To my knowledge no studies have yet been reported in a peer-reviewed forum to provide unbiased evidence of efficacy for either product. Therefore, although small experimental studies suggest that vaccination might be an effective means of minimizing disease caused by Mycoplasma bovis, research in the clinical setting with currently available commercial vaccines is sorely needed to indicate whether vaccination can be recommended as an efficacious and cost-effective means of preventing morbidity associated with Mycoplasma bovis.
It is not yet clear why some animals develop chronic disease typical of Mycoplasma bovis and others do not when it can be found in the respiratory tracts of animals in both populations. Certainly the primary insults known to put ruminants at increased risk for infection with other bacterial respiratory pathogens, such as primary viral infection, mixing of animals from multiple sources, inadequate host immunity, and environmental stressors, may all play a role, but the relative importance of these and other factors in predisposing animals to disease caused by Mycoplasma bovis are not yet known. One group presented data suggesting that disease resulting from Mycoplasma bovis has increased in severity since the early 1980s,379 so it may be that factors such as increasing virulence of currently circulating isolates or changes in certain management practices are involved. Because of the chronic nature of disease often associated with Mycoplasma bovis, the syndromes described warrant control from both a financial and an animal welfare perspective. Anecdotal reports indicate that some producers with at-risk cattle have more problems with chronic nonresponsive disease caused by Mycoplasma bovis than other producers with similarly at-risk cattle,363 so it is hoped that future research will clarify the factors that put animals at specific risk for chronic disease associated with Mycoplasma bovis infection.
Mycoplasma Pneumonia of Goats
Definition and Etiology
Caprine pneumonias that are not contagious among adults are caused by several species of Mycoplasma. Mycoplasma capricolum has not been associated with disease in the United States since 1955, and M. mycoides subsp. capri has not been identified in the United States.398 However, large colony type M. mycoides subsp. mycoides (Mmm) can be a very serious cause of mortality among goat kids and does in North America.399,400
Clinical Signs and Differential Diagnosis
In herds with Mmm infections, goat kids usually appear clinically normal until 2 to 8 weeks of age, when the following three clinical syndromes occur399,400:
1
A peracute illness characterized by high fevers (41.1° C to 42.2° C [105.8° F to 107.9° F]) and death within 12 to 24 hours
2
A CNS syndrome with opisthotonos and death within 24 to 72 hours
3
An acute to subacute syndrome with high fevers, multiple hot swollen joints, and pneumonia
The most common manifestations are swollen joints, lameness, and recumbency. About one half of affected kids have increased lung sounds on expiration and elevated respiratory rates. During an outbreak, 80% to 90% of kids die or are euthanized because of permanent recumbency. Mmm infection in adult does is also life-threatening. Forty-six does died in 1 week on a 600-goat dairy during an acute outbreak of arthritis and polyarthritis, mastitis, and interstitial pneumonia caused by Mmm infection.401
In the United States the main differential diagnosis of Mmm infection in goats is caprine arthritis-encephalitis (CAE), which is a chronic, sporadic disease. Animals with CAE are generally alert and nonfebrile and continue to eat well. Affected kids exhibit a CNS syndrome at 8 to 16 weeks of age characterized by ataxia and posterior paresis progressing to tetraparesis in 2 weeks to 2 months. They also have a progressive interstitial pneumonia that is usually inapparent. The arthritic form of CAE usually occurs in goats 1 to 2 years of age. In addition, the joints of animals with acute Mmm contain fibrinopurulent exudate, whereas mononuclear cells are present in the joint fluid of CAE cases.
Diagnosis
The definitive diagnosis of Mmm infection in individuals requires isolation of the agent from milk, joint fluid, blood, urine, or tissue. Infected goat herds can be readily identified by culturing bulk tank milk because infected does shed up to 1010 Mmm organisms per milliliter of milk.399 Inapparent carriers can be identified by milk culturing, but false-negative results are a risk because organisms are shed intermittently. An ELISA to detect specific antibodies against Mmm has been developed but has not been evaluated for detection of carrier animals.402
Pathophysiology
Field cases of Mmm infection show evidence of widespread thrombosis, suggesting disseminated intravascular coagulation. A coagulopathy, indicated by increases in prothrombin and partial thromboplastin times and a decrease in number of platelets, has been demonstrated in experimental infections.403 Mmm has been shown to cause direct damage to cultured endothelial cells and to activate complement.404
Epidemiology
Localization of Mmm in the udder with no overt signs of mastitis is a key feature of transmission of the disease. In other does, udder infections develop through contact with the organisms during milking, and their kids become infected by ingestion of colostrum.405 The localization of Mmm, often associated with mites in the external ear canal of asymptomatic goats, may also play a role in the epidemiologic process of Mmm infections.406
Necropsy Findings
The most common necropsy finding in goat kids that die of Mmm infections is a fibrinopurulent polyarthritis.399,400 Approximately one half of field cases have pneumonia. One or more lung lobes have areas of patchy to diffuse red consolidation that is sometimes covered with a fibrinous exudate. Clear, golden yellow to serosanguineous fluid is found in the thorax in half of the cases. In some patients there are fibrinous adhesions between the lungs and thoracic wall. Affected lungs have microscopic evidence of bronchopneumonia or interstitial pneumonia. Other common lesions include pericarditis, peritonitis, and enlargement of the kidneys, liver, and spleen.
Treatment and Prognosis
Conventional antibiotic therapy for goats with Mmm infections is almost always unsuccessful.399,400 Tylosin or tetracyclines are commonly used. A low percentage of kids make a clinical recovery from the septicemic illness but often have arthritis by the time they freshen. Does that recover from mastitis become chronic carriers.
Prevention and Control
Prevention is based on maintaining herds free of Mmm infection. Purchased does should originate only from herds that have no history of mortality in kids from arthritis and pneumonia and that have negative bulk tank cultures for Mmm. Purchased individuals should be held separate from the milking herd until they have an Mmm-negative milk culture result; treatment for ear mites may also be prudent. A vaccine is not commercially available; however, an experimental formalin-killed vaccine has been shown to be protective.407
Control of Mmm outbreaks is centered on prevention of the systemic infection in kids and mastitis in milking does. Rapid prevention of new cases in kids can be expected from a program of feeding heat-treated goat colostrum (56° C [132.4° F] for 1 hour) or cow colostrum at birth, pasteurized milk up to 1 month of age, and pasteurized milk or a high-quality milk replacer from 1 month to weaning.399 All kids with swollen joints should be culled. Milking hygiene should be improved to prevent transmission of infection during milking. Udders should be dried with individual cloths or paper towels, teats should be dipped with an organic iodine base preparation, and teat cups should be backflushed. Milk samples from all does in the milking herd should be cultured to identify carrier does. Infected does should be kept in a separate string and milked last or culled, depending on production. Colostrum of dry does should be cultured as they freshen, and the does should be hand-milked separately until their milk is found to be free of Mmm. Monthly cultures of the bulk tank milk from the noninfected string should be performed to ensure that it is free of Mmm infection. The goal of control procedures is eradication of Mmm from the herd.
Other Mycoplasmas
Mycoplasmas are isolated, usually in combination with other pathogens, from 50% to 90% of beef and dairy cattle pneumonias.111,112,408,409 Mycoplasmas have been associated with peribronchial and peribronchiolar lymphoid hyperplasia, which is sometimes referred to as a “cuffing pneumonia,” a common lesion in calves that die or are euthanized because of ECP.410,411 Mycoplasmas have also been recovered from lesions of acute and chronic bronchopneumonia in which a cuffing pneumonia was not apparent.
Other than Mycoplasma bovis, the species of mycoplasmas (see Box 31-4) prevalent in North America are generally considered to be mild respiratory pathogens, mainly causing subclinical infections unless coupled with environmental stresses or infections by other pathogens.409 Tracheal bronchial aspiration performed on dairy calves at random found that calves with both Mycoplasma species and Pasteurella species present in the aspirate were at significantly greater risk of developing ECP than calves with only one organism or no organisms. A recent study of feedlot cattle found seroconversion to M. alkalescens to be significantly associated with undifferentiated fever, a clinical definition similar to undifferentiated respiratory disease.297 Effects such as immunosuppression412 and inhibition of the mucociliary transport mechanism,413 which can be mediated by mycoplasmas, suggest that they may play an important contributory role in the pathogenesis of bovine pneumonia. Mycoplasma ovipneumonia is often isolated from pneumonic lungs of sheep and goats, usually accompanied by M. haemolytica. On its own, M. ovipneumonia is capable of causing mild, subacute to chronic bronchiolitis or bronchopneumonia that probably predisposes to M. haemolytica infections.
APPROACH TO DIAGNOSIS AND TREATMENT OF RESPIRATORY DISEASE OF UNDETERMINED CAUSE (UNDIFFERENTIATED RESPIRATORY DISEASE OF RUMINANTS)
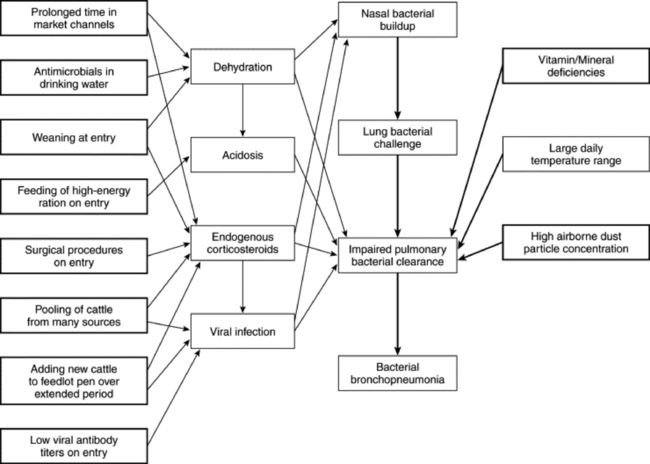
Information regarding diagnosis and treatment of individual infectious agents that can cause ruminant respiratory disease was described earlier. However, it is most common for one or more of these agents to be involved when a group of cattle, sheep, or goats has an outbreak of respiratory disease. Moreover, when a veterinarian is called to make an initial evaluation of a group of ruminants with respiratory disease, the exact causative diagnosis is not known. The term undifferentiated respiratory disease has been applied to respiratory disease of uncertain cause.419 Thinking of an outbreak as “undifferentiated respiratory disease” before a causative diagnosis is confirmed is helpful to ensure that the veterinarian considers all possible infectious agents and management factors that may be contributing to disease in the animals in question, rather than immediately focusing on one agent or factor that is guessed to be of importance. A plan to diagnose and treat the current problem and prevent future respiratory disease can then be made by administering treatments and instituting management changes appropriate for any of the agents likely to affect the class of animals involved.
Clinical Signs
Successful intervention with an outbreak of undifferentiated respiratory disease is based on identification and alteration of the risk factors associated with the outbreak. An investigation begins with collection of a thorough history of the problem and is followed by examination of affected animals and the environment. The history questions are directed to management practices that predispose to pneumonia (see later). It is important to observe personally as many management practices as possible to ensure that what is described is actually implemented. Examination of the environment includes evaluation of the nutrition program to determine whether any of the dietary factors that predispose to pneumonia are present.
Once a thorough history is collected, several animals involved in the outbreak should be examined. Because infectious respiratory disease is so common in ruminants, producers may assume that any illness not obviously occurring in another body system is a result of respiratory disease. However, to ensure rational use of antimicrobials and vaccines and to make properly focused management changes, confirmation of the clinical diagnosis of respiratory disease is necessary.
Ruminants affected by bronchopneumonia exhibit signs of respiratory tract inflammation and, sometimes, toxemia. In early stages, animals stand off by themselves and do not approach feed. They hold their heads and ears low, appear depressed, and move slowly. Respirations become rapid and shallow, there is frequent licking of the muzzle, and a moist cough is often present. Animals may have a fever of 40° C to 41° C (104° F to 105.8° F) and as the disease progresses they appear gaunt, have deep labored respirations, and may hold the head extended. Dyspnea may be both inspiratory and expiratory. Ocular and nasal discharges progress from serous to mucopurulent. Normal lung sounds are difficult to hear except in calves, goats, and sheep. Sheep normally have harsh inspiratory sounds. The heavy chest wall of larger cattle makes it difficult to hear normal airway sounds. The first auscultable lung changes are increased harshness of inspiratory sounds. By the time expiratory sounds are as loud as or louder than inspiratory sounds, severe bronchopneumonia exists. In the most severe cases, auscultation of the anterior ventral lung fields reveals crackles and wheezes and an increase in bronchial sounds, especially on inspiration. When ventral consolidation occurs, harsh tracheal breathing is still audible ventrally, but percussion reveals ventral dullness. Percussion is best accomplished on young calves and goats of any age. Recently shorn sheep can be readily percussed, but heavy wool makes percussion difficult. Animals in which a fibrinous pleuritis develops are reluctant to move because of pain, have shallow respirations, and sometimes have pleural friction rubs detectable on auscultation. Nasal discharge, dyspnea, abnormal lung sounds, cough, and high fevers are cardinal signs of bronchopneumonia. Other respiratory tract conditions that must be considered as differential diagnoses include acute bovine pulmonary edema and emphysema (ABPEE), interstitial pneumonia, pulmonary edema, pleuritis, laryngitis, tracheitis, and lungworms. Rare conditions include thoracic neoplasia and diaphragmatic hernia. Systemic conditions that result in respiratory signs include septicemia, heart failure, acid-base imbalances, and poisonings such as nitrate toxicity. An important feature that separates systemic conditions from bronchopneumonia is that, in addition to signs of pulmonary dysfunction, systemic conditions often are manifested by clinical signs of damage to other organ systems. In sheep and goats, ovine progressive pneumonia (OPP), CAE, and lung or mediastinal abscesses caused by C. pseudotuberculosis or other bacterial invaders are additional differential diagnoses.
Clinical signs associated with specific viral infections of the respiratory tract have previously been presented. In general the clinical signs observed are dependent on the stage of the disease and particularly on whether secondary bacterial pneumonia has been superimposed. In the early stages of viral pneumonia common clinical features include mild depression and anorexia, often marked elevation in body temperature, serous to mucopurulent lacrimal and nasal discharges, cough, and elevated respiratory rates. On auscultation of the lungs there may be an increase in breath sounds. In the presence of secondary bacterial pneumonia, the severity of clinical signs becomes more pronounced.
Diagnostic Workup of Undifferentiated Ruminant Respiratory Disease
If clinical examination of a group of cattle, sheep, or goats indicates that respiratory disease is present, it is not always necessary to immediately submit diagnostic tests. Occasional outbreaks of respiratory disease occur in groups of ruminants, and such occasional outbreaks can often be managed successfully with administrative symptomatic therapy alone (described later). However, if recurrent outbreaks occur, or if animals do not respond appropriately to symptomatic therapy, then diagnostic tests are warranted so that more information is available to better characterize the nature of the problem. In such situations a careful evaluation of management practices and facility design is also warranted to ensure that all practices possible are maximizing the ability of animals to resist respiratory infection. Management practices important to minimizing respiratory infection are described later (p. 638).
Clinical Pathology and Assessment of Immune Status
CBCs or serum biochemical analyses are rarely of much value in diagnosis of respiratory disease in ruminants. Some viruses, such as BVDV, may cause leukopenia, but when bacterial pneumonia is superimposed the WBC count is most often in the high-normal range to mildly elevated with a left shift. Animals with bacterial pneumonia may have an inflammatory leukogram characterized by a leukocytosis with a mature neutrophilia, possibly with a left shift; hyperfibrinogenemia is likely to occur. Animals with chronic pneumonia may have a normal WBC count even when there is significant pulmonary pathology.
Failure of passive transfer is a major risk factor for pneumonia in calves, so investigation of outbreaks in calves should include an evaluation of passive transfer status by measurement of immunoglobulins in the serum of calves 1 to 7 days of age. The zinc sulfate turbidity test, sodium sulfite test, and measurement of total serum protein with a refractometer are practical, satisfactory procedures for estimation of serum immunoglobulin concentrations.
Necropsy Findings
The value of necropsy findings to confirm the cause of death, particularly in animals that die unexpectedly, cannot be overemphasized. If producers can be encouraged to allow all animals that die to be subjected to necropsy, much valuable information may be gained before a disease outbreak causes excessive mortality. It is unfortunately common for veterinarians to be contacted only after several animals have died without any having been subjected to necropsy, and thus much valuable information that could have helped prevent further disease and death has been lost. Establishing a practice of subjecting all animals that die to at least gross necropsy can aid greatly in maintaining management practices that limit animal disease and death; this is because the actual cause of death identified at necropsy can sometimes be unexpected. For example, if an animal is assumed to have died of pneumonia, but it actually died of acute enterocolitis, then control measures may be undertaken that are inappropriate for the true problem.
The cost of having animals sent to the local diagnostic laboratory for full necropsy may dissuade some owners from allowing necropsies. However, much useful information can be gained simply by gross necropsy evaluation performed on the farm by the local veterinarian. With some practice veterinarians can develop confidence in identifying the major differentiating gross features of common diseases, and they can thus ensure that management is aimed at control of disease of the correct organ system. Moreover, there are some identifiable characteristics typical of the major infectious causes of bronchopneumonia. Gross necropsy can help the veterinarian make a more accurate list of differential diagnoses for outbreaks of bronchopneumonia, allowing the development of a more logical plan for treatment and prevention.
A respiratory necropsy should include assessment of the upper airways and trachea. Fibrinopurulent material in the larynx is evident in cattle with necrotic laryngitis (see Fig. 31-50). Infection with BHV-1 (IBR) causes generalized reddening (congestion) and small raised, red or pale plaques on the mucosa of the nasal passages and trachea; more severe cases have dark red, hemorrhagic changes, possibly with yellow-brown exudate adherent to the mucosa (fibrinopurulent tracheitis) (see Fig. 31-53).
Bronchopneumonia in recently transported cattle (shipping fever pneumonia) is most commonly a fibrinopurulent bronchopneumonia. The infection is aerogenous; it begins in the bronchioles and extends through their walls into the surrounding parenchyma. The cranioventral areas of affected lungs are swollen, dark red to gray-brown in color, firm, and heavy. Bronchial lymph nodes are swollen, wet, and dark red. The inflamed lung and parietal pleura are sometimes covered with variable amounts of yellow fibrin, and the pleural cavity may contain straw-colored fluid (see Fig. 31-56). Fibrinous pleuritis usually indicates the presence of M. haemolytica or H. somnus. These species can also cause necrosis of lung, which will be firm and brown to gray (see Fig. 31-57), or dark red wedge-shaped lesions (infarcts) that are caused by thrombosis of an artery supplying the region (see Fig. 31-58). The dorsal regions of the caudal lobes often are mottled by interspersed patches of inflammation and normal parenchyma. In up to one third of bronchopneumonia cases, forced respirations result in vesicular to bullous pockets of emphysema in the dorsal areas of the caudal lobe (see Fig. 31-54). These changes can also be seen with primary BRSV infections or with AIP. Focal or multifocal areas of firm white to yellow material that look like abscesses may actually be caseous necrosis caused by Mycoplasma bovis (see Figs. 31-60 and 31-61); the chronic phase of pneumonia resulting from M. haemolytica may also cause similar lesions. Abscesses containing caseous or liquid purulent material may also be caused by Arcanobacterium (Actinomyces) pyogenes or anaerobic bacteria. Lambs that die of bronchopneumonia caused by M. haemolytica have swollen lungs with reddish purple anterior ventral consolidation. An extensive fibrinous pleuritis with large amounts of straw-colored exudate is often present. Chronic cases have multiple abscesses and pleural adhesions. As much as 60% to 80% of the lung tissue is usually involved in fatal cases of severe bacterial pneumonia.
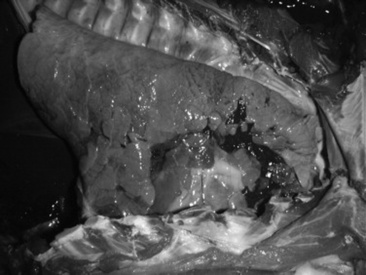
Bronchopneumonia in housed dairy calves (ECP) is less often fibrinous, but rather is characterized by the presence of firm, collapsed, dark red lobules in the cranioventral or caudal ventral lung (Fig. 31-62); in severe cases whole lobes of the lung may be affected. Firm dark red lobules without fibrin on the pleura are common in pneumonia caused by P. multocida, Mycoplasma bovis, or other mycoplasmas. Infection with BRSV and PI3 can also cause lobular consolidation of the ventral lung (see Fig. 31-55). Less commonly M. haemolytica or H. somnus organisms are isolated from such lesions without fibrinous pleuritis.
Animals that die after chronic persistent coughing, dyspnea, and weight loss exhibit lesions of chronic suppurative pneumonia. Bronchi and bronchioles are filled with purulent exudate, there are multiple mature lung abscesses, and greatly dilated bronchioles contain malodorous exudate. When bronchiectasis is severe, the lung lobes have a nodular appearance. Pulmonary abscesses and bronchiectasis are common findings in cases of chronic pneumonia and explain poor weight gains.414
Although gross necropsy alone can be very helpful in generating an accurate list of differential diagnoses, histopathologic and microbiologic findings often add critical information. Gross necropsy findings are not always definitive; for example, in one study of AIP in feedlot cattle, only 67% of the cases that were diagnosed with AIP based on clinical and gross pathologic findings were confirmed by histopathology.420 Therefore in outbreaks with relatively high mortality or in cases in which long-term and possibly expensive therapy is not yielding expected results, the cost of full necropsy of two or three typical cases is likely to be well worth the expense.
If full necropsy at a local diagnostic laboratory is to be undertaken, two or more animals showing signs that are typical of the early stages of the disease outbreak should be selected for euthanasia and necropsy. Although the producer may be reluctant to euthanize animals that may recover, and would rather send animals with chronic, nonresponsive disease, the chronic cases are unlikely to yield information relevant to the primary problem.
MICROBIOLOGIC TESTS
If samples are collected from a necropsy on the farm for testing for viral or bacterial pathogens, proper handling and transport of the specimens is critical to maximize the chance of an accurate diagnosis. The veterinarian is encouraged to contact the diagnostic laboratory if uncertain about proper methods of sample collection and transport; most laboratories now post this information on their websites, making it easy to find for those with Internet access.
Viruses
A specific viral diagnosis requires laboratory confirmation. Most laboratories direct their diagnostic efforts toward the viruses for which vaccines are available. Diagnosis of other respiratory viruses may require the assistance of specialized laboratories. Because of the time and expense that specific viral diagnosis entails, care must be taken in the collection, storage, and transport of appropriate specimens to a diagnostic facility. The veterinarian is encouraged to contact the diagnostic laboratory if uncertain about the proper methods for collecting and transporting specimens for microbiologic diagnosis.
VIRUS ISOLATION
Virus isolation is time-consuming and expensive, but it is a sensitive method for identifying viruses. Virus isolation is performed in cell culture. A variety of specimens can be tested, including nasopharyngeal, conjunctival, and tracheal swabs, TTAs, BALFs, and a variety of respiratory tract tissues that can be obtained at postmortem examination. Fluids, tissues, and swabs may be frozen; alternatively, swabs and tissue specimens may be placed in a viral transport medium and kept refrigerated until arrival at the diagnostic laboratory, preferably within 24 hours. BRSV does not appear to survive freezing or transport well, and it is important that specimens be inoculated onto cell cultures as soon as possible. In general, better success at virus isolation is obtained when specimens are collected in the acute phase of disease. Chances of successful isolation may be improved by sampling asymptomatic animals that are in close contact with affected animals. These animals may be in an incubation phase of infection. Some viruses appear to be more difficult to isolate than others. For example, BRSV is very difficult to isolate by routine procedures, and other diagnostic procedures (discussed later) should be performed in conjunction with attempts at virus isolation. During isolation procedures, viruses are detected by production of cytopathic changes in cell monolayers. Viral identification is accomplished by a variety of procedures such as neutralization with specific antiserum, FA staining, immunoperoxidase staining, and examination by electron microscopy and immunoelectron microscopy. An immunoperoxidase monolayer assay has been developed for detection of BVDV and is in routine use for screening serum samples for detection of cattle persistently infected with BVDV.
DETECTION OF VIRAL ANTIGENS
Immunofluorescence is a rapid method for identification of specific respiratory viruses. Antemortem identification can be made from conjunctival or nasal smears and from cells obtained by tracheal lavage or BAL. Postmortem identification can be made from frozen tissue sections prepared from a variety of respiratory tract tissues.
Another technique that is used to detect viral antigen in tissues is immunoperoxidase staining, which is most often carried out using formalin-fixed tissue. This is a very useful procedure that allows histologic examination of tissues in conjunction with immunologic identification of the causative agent.
Antigen capture enzyme immunoassay (EIA) provides a rapid means for detection of respiratory viruses. These tests can be performed on fluids obtained from the respiratory tract. Commercially available antigen capture EIAs are available for diagnosis of human RSV infections in infants and young children that are also capable of detecting BRSV,421 and these are in use in some veterinary diagnostic laboratories. The same technique has been developed for the detection of BVDV and is in use for screening serum samples to detect cattle persistently infected with BVDV.
DETECTION OF VIRAL NUCLEIC ACIDS
The nucleotide sequence has been determined for the genome or partial genome for many of the ruminant respiratory viruses. Testing for viral nucleic acid by PCR (for DNA viruses) or RT-PCR (for RNA viruses) is increasingly more widely available at veterinary diagnostic laboratories. Although they are not currently in routine use, the possibility exists of using nucleotide probes for the detection of these viruses in tissue samples. One benefit of using nucleic acid detection to identify pathogens is that the pathogen does not have to be alive to be identified; this can be a particular benefit for relatively fragile viruses such as BRSV.
SEROLOGIC DIAGNOSIS
Retrospective diagnosis of viral infections can be made by determination of antibody titers in paired sera from individual animals. The first sample is collected in the acute phase of the disease, and the second is collected 2 to 4 weeks later (“convalescent sample”). In a respiratory disease outbreak multiple animals should be tested to achieve a serologic diagnosis; it is typical for seroconversion to be identified in only a subset of animals tested in any outbreak. Serologic diagnosis is made by demonstrating a fourfold increase in antibody titer in the convalescent sample as compared with the acute sample; a fourfold fall in titer also indicates recent infection. Because day-to-day variation in results of tests used for serologic diagnosis is typical, the acute and convalescent samples should be run by the laboratory on the same day. Thus the acute samples can be stored in the freezer by the local veterinarian and shipped together with the convalescent samples.
Because infections in young ruminants can occur in the presence of passively derived antibodies, seroconversion might not always occur during outbreaks of bronchopneumonia involving young animals.422 This problem may be overcome by inclusion of older individuals in contact with the younger animals in the population sampled, which are likely to have lost passively derived antibody to these viruses and will be more likely to seroconvert. Also, BRSV antibody levels appear in some instances to peak at the onset of severe disease, and a decreasing antibody level is seen on paired serologic analysis rather than a rising level. Serologic testing of normal appearing, in-contact cattle that may be in early stages of infection may be helpful in demonstrating seroconversion to BRSV. A wide variety of serologic procedures is available for antibody determinations, but most laboratories use a microtiter serum neutralization test (also known as virus neutralization test) for IBRV, BVDV, PI3, and BRSV. It is important to remember that serum neutralization tests take several days to run. Some laboratories are beginning to use more rapid procedures such as ELISA for determination of serum antibody titers. Through use of an isotype-specific ELISA, diagnosis of BRSV can be achieved with a single serum sample by measurement of IgM levels423; similar tests could be developed for other pathogens, but these are unlikely to be widely available. A hemagglutination-inhibition test can also be used for PI3 and respiratory coronavirus.
Bacteria
A wide variety of bacteria have been isolated from the respiratory tract of ruminants in association with respiratory disease. However, the most frequent and most important isolates are M. haemolytica, P. multocida, H. somni, and M. bovis. The isolation of A. pyogenes, coliforms, or anaerobic bacteria often is indicative of chronic pneumonia or aspiration pneumonia and may be associated with lung abscessation.
Before bacterial culture is attempted, the status of any recent antibacterial therapy should be determined, and, if possible, specimens from untreated cattle should be collected. It is important to remember that M. haemolytica, P. multocida, H. somni, and mycoplasmas are normal inhabitants of the nasal passages of cattle and may be cultured in the absence of respiratory disease. It is important to note that not all laboratories will identify the species of mycoplasmas cultured; thus an isolate may be identified only as “Mycoplasma sp.” If involvement with Mycoplasma bovis is suspected, the laboratory may need to be specifically asked to identify the species of any mycoplasmas they isolate. Specific identification of M. bovis is most often done by immunofluorescent assay (available at the diagnostic laboratory at Iowa State University, and possibly others), IHC (available at Iowa State University, the University of Saskatchewan, and possibly others), or PCR (available at the diagnostic laboratory at the University of Georgia in Athens, and possibly others). Some mycoplasmas are more hardy or easier to grow than others; for example, it is not unusual for Mycoplasma bovis to overgrow M. dispar when both are present in the same sample. Culture of mycoplasmas typically takes at least 1 week, and laboratories often call a sample negative only after the sample has been subcultured at least once.
Although isolates obtained from nasal swabs may reflect the organisms causing pneumonia on a group level, specificity can be increased by obtaining specimens from the lower respiratory tract.299 Specimens appropriate for bacterial culture are similar to those discussed for viruses, such as nasopharyngeal and tracheal swabs, TTAs, and BAL or lung aspirates. Swabs are acceptable for transferring samples directly to culture medium, but if transport is necessary, the swab must be placed in a transport chamber such as a Culturette (Marion Scientific, Kansas City, Mo.) to ensure adequate moisture for the sample during transport. Contamination of the sample with environmental agents can best be prevented by first searing the lung surface, then making an incision with a sterile scalpel, followed by sampling with a swab through the incision; but it may not be practical to sear the surface of the lung in the field setting. Specimens of respiratory tract tissues such as lung and bronchial lymph nodes can be placed in sterile containers such as Petri dishes or self-sealing plastic bags and transported to the laboratory on ice.
Chlamydial organisms may be demonstrated by staining smears and sections of lesions with a Gimenez stain or by immunofluorescence techniques. Isolation attempts are done by inoculation of yolk sacs of embryonated chicks. Serologic tests such as complement fixation are also available.
DETECTION OF BACTERIAL ANTIGENS OR NUCLEIC ACIDS
It is increasingly common for diagnostic laboratories to use IHC for identification of bacteria in formalin-fixed tissues. This technique offers many advantages, including correlation of the pathogenic organism with the lesion and detection of pathogens not found on bacterial culture because of overgrowth of other organisms such as A. pyogenes or P. multocida. The use of PCR for identification of bacterial agents in a variety of samples is also increasingly available. The veterinarian is encouraged to contact the diagnostic bacteriologist at the local diagnostic laboratory or check the laboratory website for information regarding which tests are available.
SEROLOGIC TESTING FOR BACTERIAL PATHOGENS
Diagnosis of infection with bacterial pathogens can be attempted as described earlier for viral pathogens. However, the use of serologic tests for identification of infection by bacterial respiratory pathogens has mostly been limited to use in research. Thus these tests are not likely to be widely available for clinical use.
Treatment of Undifferentiated Bronchopneumonia
ANTIMICROBIAL THERAPY
The basic foundations of antimicrobial therapy for bacterial bronchopneumonia are treat early enough, treat long enough, and treat with the appropriate antimicrobial agent. Because there are currently many effective products marketed for the treatment of important bacterial respiratory pathogens in cattle, treating early enough is perhaps more important than what is used for therapy. It should be remembered that a major reason for treatment failure is the presence of a lesion that is too far advanced for successful therapy. If lesions becomes too far advanced, the antimicrobial agents will have difficulty reaching walled-off areas of necrosis and suppuration; moreover, the regenerative response will not be able to return this tissue to normal lung parenchyma.
Although antibacterial agents for the treatment of bacterial bronchopneumonia may reduce losses caused by fatality and retarded growth, they do not serve as a substitute for preventive management practices. Cattle requiring treatment do not perform as well as those that have not needed treatment. However, cattle requiring only one treatment perform better than those that require two or more treatments424; this further emphasizes the need for treating early enough with an effective antimicrobial, because animals with treatment failure will have suboptimal performance.
The precise temperature used to determine whether animals need treatment depends on the balance between the costs of overtreatment (drugs and labor) and undertreatment (treatment failures and mortality.) This temperature may vary depending on the animal type. The cutoff commonly used for feedlot cattle is 104° F to 104.5° F (40° C to 40.3° C), but 103.5° F (39.7° C) may be more appropriate for calves. This recommendation is based on the long-term effects that ECP has been shown to have on growth rate, age at first calving, culling before calving, and culling after calving, indicating that ECP can have long-lasting effects if not properly treated with early antibiotic therapy. When outbreaks of respiratory disease occur, surveillance of the affected group must be increased to ensure early detection of diseased animals.
Treatment of sufficient duration can be achieved only if the response to therapy is monitored. Therapy should be continued for at least 48 hours after clinical signs of fever, dyspnea, and toxemia have abated. Many of the antimicrobial drugs labeled for use in the treatment of pneumonia in cattle provide multiple days of therapeutic drug concentrations in lung tissue after only a single injection. These products decrease the time and stress associated with daily treatment; they also make it easier for feedlots to return animals to home pens rather than keeping them in hospital pens, which seems to be associated with poorer responses by treated animals.425 Frequently, antibiotics are evaluated over a 3-day treatment period, with cases failing to demonstrate a normal body temperature after 3 days being classified as nonresponders; a different class of antimicrobial should be administered to nonresponders.
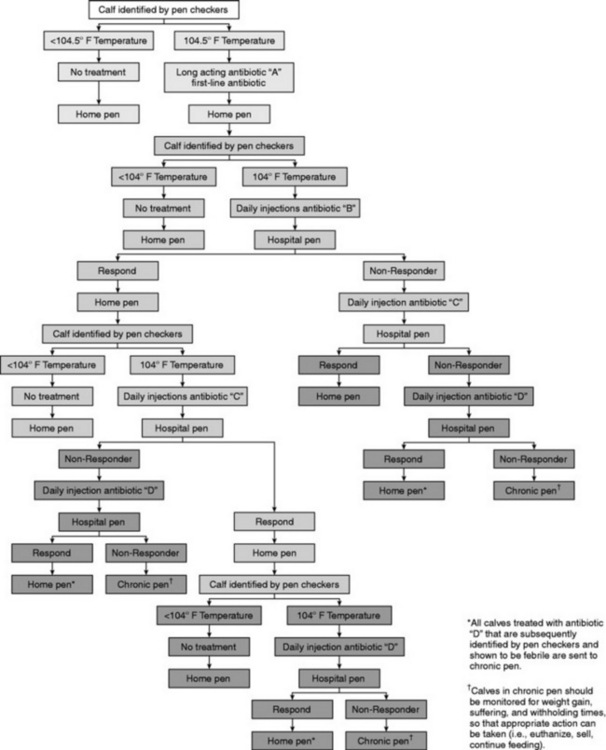
Although a 3-day course of antimicrobial therapy for bronchopneumonia was for many years standard operating practice, particularly in the treatment of feedlot cattle, the view is increasingly held that treatment for a longer duration may be more appropriate. This is particularly true for pneumonia caused by Mycoplasma bovis in dairy calves or feedlot cattle, which seem to respond better to treatment for at least 7 to 10 days.359,395 Although treatment for 7 to 10 days would be extralabel for many antibiotics, at least three products currently marketed have been shown to provide therapeutic drug levels for at least 7 days when used at dosages recommended on the label (see Table 31-10). Unfortunately there is little research evaluating the effect of treatment duration on long-term outcome in ruminants with bacterial bronchopneumonia, so it is difficult to make evidence-based recommendations regarding exactly how long antibiotics should be administered. Determination of therapeutic response by evaluation of general appearance without regard to restoration of normal body temperature has been shown to result in high relapse rates. A typical decision tree for treating feedlot cattle with antibiotics is shown in Fig. 31-63, but protocols such as these may be modified as more information about the use of long-acting antimicrobials is obtained from field research.
Selection of the appropriate antibiotic tends to be what most veterinarians focus on when treating respiratory disease because this is the aspect of therapy over which they have the greatest control. Factors such as cost, route of administration, treatment interval, drug labeling, necessity of extralabel doses, and withholding times quickly cull a number of antibiotics, leaving a short list of suitable alternatives for use as first-line antimicrobial agents. Antibiotics that are associated with severe injection site reactions such as erythromycin or those associated with complications of administration such as balling gun injuries with oral boluses are often avoided. Only antibiotics that are licensed and effective at label doses should be considered for routine use in food animals. Table 31-10 lists the approved antimicrobials for treating respiratory disease in cattle. If data regarding the MIC of bacteria isolated from animals before treatment with antimicrobials are available, the dose or duration of therapy can be rationally modified, but it is imperative that proper withdrawal times be observed when antimicrobials are used in an extralabel fashion. At the time of this writing, the Food Animal Residue Avoidance Databank (FARAD; www.farad.org or 1–888-USFARAD) is an invaluable resource for veterinarians who need to identify withdrawal times for drugs administered to food animals at extralabel dosages. The use of MIC data requires some understanding of the pharmacokinetics of the drugs in question; published information is available regarding interpretation of MIC data, or veterinarians can contact FARAD or the diagnostic bacteriologist at their local diagnostic laboratory for assistance.
Determining the antimicrobial sensitivity patterns or MIC data for causative agents such as M. haemolytica in the case of bacterial bronchopneumonia can be difficult. The best information is that gained from bacterial culture and susceptibility testing of animals subjected to necropsy before receiving any antimicrobial therapy; however, producers may be reluctant to allow necropsy of animals that have never been treated. Once an animal has been treated, any antimicrobials administered can bias the results of susceptibility testing of bacteria later cultured from the animal. Many bacteria, including M. haemolytica, can develop plasmid-mediated multiple antimicrobial resistance by bacterial conjugation so that, for example, exposure to oxytetracycline may induce resistance not only to oxytetracycline, but also to penicillin. Therefore, M. haemolytica recovered from cattle treated with antibiotics will have a different sensitivity pattern than M. haemolytica cultured from untreated cattle. This is especially important when reviewing publications reporting antimicrobial susceptibility of M. haemolytica isolates cultured at diagnostic laboratories, because these results will most often be from cases that have been treated with antimicrobial agents before death. The findings of these studies may be looked at as a worst-case scenario demonstrating antimicrobial agents for which acquired resistance is rarely or never a problem, versus those for which acquired antimicrobial resistance occurs commonly. In general, resistance to many drugs used for the treatment of bovine pneumonia has been identified when M. haemolytica isolates from cattle treated with antibiotics before death are evaluated.
Sensitivity testing using isolates from clinical cases raises questions regarding the best site for sample collection. Antibacterial sensitivities of isolates cultured from nasal swabs may not represent sensitivities of organisms causing pneumonia. This is surprising, because pneumonia usually is preceded by multiplication of bacteria such as M. haemolytica in the upper respiratory tract, and the nasopharynx serves as the source of bacteria colonizing the lungs. Nevertheless, there are discrepancies between antimicrobial sensitivities of bacteria isolated from nasal swabs and clinical outcome. Specimens for sensitivity testing should be collected from pneumonic lung, tracheal swabs, or TTAs collected from cattle before treatment whenever possible.
The choice of which drug to use can also be based on records of efficacy for animals treated in the past. Producers who keep accurate records of drugs used, with results of treatment responses, relapses, and chronic cases, may select first-line antibiotics based on historical performance of a drug. This may be the best approach to antimicrobial selection. A final method of choosing a first-line antimicrobial drug is reliance on published treatment trials. Published trials give comparisons between among responses for various antibiotics in cattle with naturally occurring bacterial bronchopneumonia. The outcomes for these trials are often expressed as both health and production values. When evaluating published treatment trials that use cases of naturally occurring bacterial bronchopneumonia, it is important to realize that the results may not be applicable to the cattle and pathogens outside the operation where the trial was carried out, but if the trial was well designed, it should provide useful comparisons. The characteristics of a well-designed field trial are discussed later in the section on vaccination (p. 641).
Mass medication with antibiotics at full therapeutic doses during an outbreak of feedlot cattle pneumonia dramatically curtails the daily number of new cases and improves feed consumption. In the acute stages of an outbreak, whatever drug is chosen should be given by injection; therapeutic levels cannot be reliably maintained by administering drugs in feed or water. Bronchopneumonia cases that occur after administration of mass medication have an increased possibility of being resistant to therapy with the antimicrobial used in mass treatment and have a greater than usual resistance to other antimicrobials. Thus mass medication should not be a standard practice but is warranted to control severe outbreaks of pneumonia. It is important to base a decision to mass medicate on measurable criteria. Some feedlot veterinarians implement mass medication when the pull rate of sick animals is 10% on any 1 day or is 25% over a 3- to 5-day period. A sudden drop in feed consumption, especially in high-risk cattle, is another situation in which mass antibiotic medication should be cost-effective. Long-acting antibiotics have extended withdrawal times to slaughter. It is of critical importance that animals that receive them are properly identified and that the withdrawal times are observed.
The same principles of therapy described previously apply to the treatment of sheep and goats.220 Unfortunately, fewer drugs are labeled for the treatment of sheep and goats as compared with products used for cattle. Drugs labeled for use in sheep and goats are listed in Table 31-10. Note that whereas tilmicosin (Micotil 300, Elanco Animal Health) is labeled for use in sheep, fatal reactions to tilmicosin have occurred in goats treated with this drug. Goats should not be treated with tilmicosin. Long-acting tetracycline at 10 mg/kg is very effective against experimental M. haemolytica infections in sheep. Administration of tetracycline subcutaneously is effective and less painful than IM injection. It is imperative to ensure that producers are given an appropriate meat and milk withdrawal time when antimicrobials are administered to sheep and goats at extralabel dosages; this is a particular concern in feedlot lambs or goats and in lactating dairy sheep or goats, from which products may enter the food chain in a relatively short time after treatment.
ANTIINFLAMMATORY THERAPY
Favorable responses to treatment with corticosteroids and antihistamines have been reported from field outbreaks of BRSV infection. However, corticosteroids should not be used indiscriminately in the treatment of respiratory disease because of the potential for immunosuppression. Corticosteroids may have a place in treatment of respiratory diseases such as necrotic laryngitis or tracheal edema syndrome of feedlot cattle.90 It is unlikely that a single administration of a glucocorticoid will have a detrimental effect on the immune system of cattle. The dose for dexamethasone in cattle is 0.05 to 0.2 mg/kg IM or IV, and the dose for isoflupredone acetate is 10 to 20 mg IM. Treatment with corticosteroids may cause recrudescence of BHV-1 infections.
NSAIDs such as acetylsalicylic acid (aspirin), flunixin meglumine, and ibuprofen have been reported to be beneficial in the treatment of respiratory disease in ruminants. Aspirin (100 mg/kg every 12 hours) is approved for use in cattle, as is flunixen meglumine (1.1 to 2.2 mg/kg IV either as a single dose or divided into two doses at 12-hour intervals). Flunixin meglumine administered intravenously at 2.2 mg/kg to calves with pneumonia induced by PI3 virus results in a marked improvement in clinical signs and reduction in lung consolidation.426 However, the use of flunixin meglumine may not be cost-effective in large numbers of animals. It is important to remember that flunixin meglumine is approved only for IV use; administration via the IM or SC route can result in violative residues in tissues if longer withdrawal times are not observed. Phenylbutazone, which has been used in the past for antiinflammatory effect in ruminants, is now illegal to use in lactating dairy cattle, and because of prolonged tissue levels found in treated cattle its use is discouraged in all food animals.
Unlike corticosteroids, NSAIDs do not impair immune function. The clinical responses of calves with either experimental or naturally occurring pneumonic pasteurellosis are markedly improved by adding flunixin meglumine to tetracycline therapy. In contrast, supplementation of antibiotic therapy with corticosteroids usually results in poorer responses, more relapses, and prolonged illness, although there is still some controversy over the use of corticosteroids for treatment of pneumonia. Because of the potential for renal toxicity with NSAIDs, dehydrated animals should be rehydrated before administration of these drugs. Care should also be taken not to overdose with NSAIDs or use them for prolonged periods, because they may also result in abomasal ulceration.
There has been little work done to evaluate the use of antihistamines as an ancillary treatment for bovine respiratory disease. Tripelennamine HCl is labeled for cattle at a dose of 1.1 mg/kg, which can be repeated in 6 to 12 hours if needed.
Tilmicosin has been suggested to have antiinflammatory effects in cattle because of the effect of the drug to induce apoptosis in leukocytes, which could theoretically decrease inflammatory responses in treated animals.427 However, the concentrations of tilmicosin used in this study were quite high relative to concentrations obtained in treated animals, and a later study of the effect of tilmicosin on the function of leukocytes taken from treated cattle showed no effect.428
ANTIVIRAL AND IMMUNOMODULATING THERAPY
Because viral respiratory infections predispose to development of secondary bacterial infections, antibiotic therapy is indicated to prevent or limit the development of bacterial pneumonia in animals with viral respiratory tract disease. Few antiviral drugs are available in human medicine, and none of these is in routine use in veterinary medicine for the treatment of viral respiratory disease in ruminants. Interferon has potential as an immunomodulating and antiviral drug in the prevention and treatment of viral respiratory disease. Human leukocyte interferon has been shown to decrease morbidity associated with shipping fever, but the therapy has not become widely used. Levamisole and isoprinosine have been used in attempts to stimulate the bovine immune system with equivocal success and cannot yet be recommended as supportive treatments.426 It is important to recognize that the immunostimulatory benefits of levamisole occur at doses in the 2- to 3-mg/kg range, compared with the anthelmintic dose of 6 mg/kg. A decreased immune response has been observed after an 8-mg/kg dose of levamisole. High doses of vitamin C (1 g/45 kg; 1 g/100 lb) have been shown to enhance the activity of bovine neutrophils and reverse dexamethasone-induced suppression of neutrophil activity.429 Isoprinosine has been evaluated as an immunomodulating drug for treatment of bovine respiratory disease and has shown some potential on the cellular level.426
SUPPORTIVE THERAPY
Supportive treatment of any kind will relieve stress, thus fostering the resistance of the patient, a very important component of the successful therapy of pneumonia cases. Sick animals should be provided shelter that protects them from rain, cold, wind, and hot sun. They should not be crowded, and the best-quality feed and clean water should be easily accessible. Mineral and vitamin deficiencies should be corrected with the use of injections or oral preparations if necessary. An IM vitamin A injection is considered useful adjunctive therapy by some veterinarians treating ruminants with bronchopneumonia.
Epidemiology of Ruminant Respiratory Disease
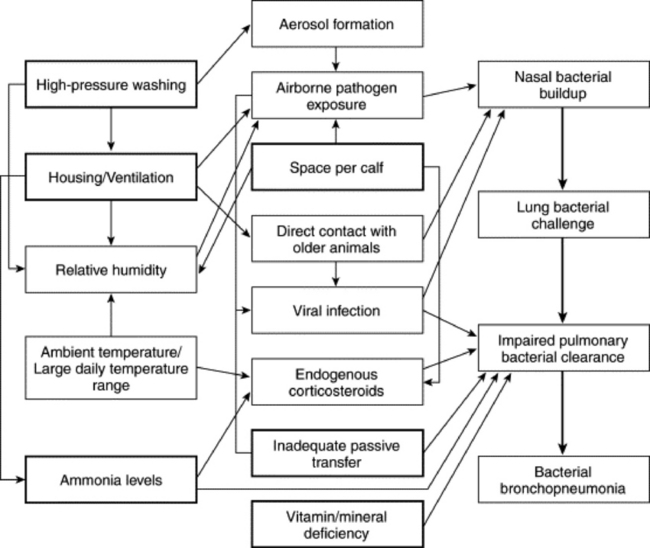
ENZOOTIC CALF PNEUMONIA
ECP has traditionally been described as affecting calves from 2 to 6 months of age; however, prospective studies examining cohorts of calves have found calves may be affected with ECP as early as 2 weeks of age.430 Slaughter surveys of dairy calves 4 to 14 days of age have found ECP to be the second most common cause of slaughter condemnation.431 Virtala and colleagues430 found that veterinarian-diagnosed ECP occurred at a younger age than did caretaker-diagnosed ECP. As a whole, these studies suggest that ECP may start much earlier than previously recognized.
Pneumonia of dairy calves occurs both as endemic disease and as outbreaks (epizootics) of respiratory disease. Chronic endemic disease is the most common manifestation of this disease, and as a result pneumonia of dairy calves is commonly called enzootic calf pneumonia. The distinction between enzootic and epizootic calf pneumonia is important in reference to etiology because different causes are more important in each form of the disease.
Waltner-Toews, Martin, and Meek432 determined from producer diagnosis that 15% of Ontario Holstein dairy calves were treated for pneumonia before weaning. Curtis, Erb, and White433 reported that Holstein calves in New York had a crude incidence risk of 7.4% for respiratory tract illness, as diagnosed by the farmer. Sivula and colleagues434 found that 7.6% of 845 Minnesota dairy calves were diagnosed by producers as having pneumonia. Van Donkersgoed and colleagues435 found that the risk of pneumonia in Saskatchewan dairy calves was 39% as diagnosed by the farmer and 29% when the pneumonia was veterinarian diagnosed. Virtala and colleagues430 found that the risk of pneumonia was 11% in New York dairy calves when diagnosed by producers and 25.6% when diagnosed by a veterinarian.
Mortality rates reported for ECP vary from 1.8%434,435 to 4.2%.430 Case fatality rates reported for calves with ECP range from 2.2% to 9.4% and vary with the sensitivity of the initial detection method (veterinarian versus producer).430
Pneumonia accounts for a significant proportion of the mortality (proportionate mortality) in dairy calves raised on dairy farms. Pneumonia accounted for 24% of deaths in New York calves430 and 30% in Minnesota calves.434 In one study examining Ontario veal calves raised in veal barns, pneumonia accounted for 52% of mortality in 4863 calves on six farms.436 Producer accuracy in diagnosing causes of mortality was examined by Sivula and colleagues.434 Producers were found to be moderately accurate but often listed the cause of death as unknown. This emphasizes the importance of laboratory confirmation of cause of death over producer diagnosis.
SHIPPING FEVER
Despite the undisputed economic importance of the disease, surprisingly little has been established regarding the behavior of pneumonic mannheimiosis or shipping fever at the population level, and even the question of whether the disease is truly contagious has yet to be answered from the epidemiologic perspective. Reviews of the literature from North American feedlot studies before 1985 found that published measures of morbidity in calves ranged from 0% to 69%, whereas measures of population mortality ranged from 0% to 15%. Incidence of disease was found to peak within 3 weeks of calves arriving at the feedlot. In view of the lack of uniformity in methods used to define cases of respiratory disease and calculate disease incidence, however, these data may not be reliable. In most of the studies the crude measures of total morbidity and total mortality were used as outcomes, and case definitions for these were often highly variable or absent. This lack of uniformity with respect to case definition and nomenclature, along with the inherently subjective nature of morbidity assessment, makes it difficult to draw legitimate comparisons among reports. One observational study specifically addressing epidemiology of fibrinous pneumonia (the classic lesion of shipping fever caused by M. haemolytica) was conducted by Ribble and colleagues.305 Because of the difficulty and expense inherent in making definitive diagnoses of the causes of illness and death among feedlot cattle, most epidemiologic studies have used crude (total) mortality as an estimate of death losses caused by shipping fever, and treatment rate as a measure of respiratory morbidity. The results of several necropsy surveys and the previously cited epidemiologic study, however, indicate that crude mortality is unreliable as a surrogate measure of fibrinous pneumonia mortality and may lead to erroneous conclusions regarding risk factors contributing to this important disease.
In the observational study of fatal fibrinous pneumonia conducted by Ribble and colleagues in 1995,305 risk factors specifically associated with shipping fever mortality in western Canadian feedlot calves were investigated. Data were collected on all 58,885 spring-born calves entering a single feedlot in southwestern Alberta between September 1 and December 31 over a 4-year period (1985 to 1988). The vast majority of calves were purchased from auction marts throughout western Canada and transported to the feedlot by truck. A complete necropsy was performed on all dead cattle within 24 hours of death, and a gross diagnosis recorded. Cases were assigned a diagnosis of fatal fibrinous pneumonia on the basis of pathologic evidence of acute fulminating lobar bronchopneumonia with fibrinous exudate, and data from each year were analyzed separately. Crude mortality ranged from 2.44% to 4.78%, whereas the mortality caused specifically by fibrinous pneumonia varied more than tenfold (0.25% to 2.73%) between years. Proportionate mortality caused by fibrinous pneumonia ranged from 10% to 57%, and this large annual variation was interpreted as evidence that crude mortality should not be used as a measure of fibrinous pneumonia mortality in epidemiologic studies. Epidemic curves constructed for each of the 4 years showed that peak mortality occurred approximately 16 days after arrival at the feedlot and that at least 50% of fibrinous pneumonia mortalities occurred within 3 weeks of arrival. Epidemic curves using the time of first treatment for all cases that eventually died of fibrinous pneumonia revealed that peak fatal disease onset occurred within 8 days of arrival and that 75% of fibrinous pneumonia mortalities were already sick within 2 weeks of arrival. The consistent onset of fatal disease in calves within days of their arrival at the feedlot indicates that the disease process may have been well underway in affected calves before their installation in a home pen, and that preventive measures should be implemented at the time of arrival, or possibly even before. In one of the study’s most important findings Ribble and colleagues305 demonstrated that when the incidence of fatal shipping fever was high (greater than 2%), the disease clustered within truckload groups of calves and also, in 1 year, within pens. This was contrary to the conclusions of other studies and lends credence to anecdotal reports from feedlot owners that shipping fever mortality is not randomly distributed in calves throughout the feedlot, but may often be abnormally high in individual truckloads or pens, indicating a contagious nature to the disease.
Although H. somni can also cause fibrinous bronchopneumonia in feedlot cattle, epidemiologic curves evaluating the day of first treatment of calves ultimately dying of disease caused by H. somni are distinct from epidemic curves of cattle dying of fibrinous pneumonia caused by M. haemolytica. The median days after arrival for the onset of fatal disease caused by H. somni is 28, as opposed to 8 days after arrival for fatal fibrinous pneumonia.345,346
BRONCHOPNEUMONIA OF ADULT CATTLE
Although anecdotal reports indicate that adult beef or dairy cows can experience outbreaks of bronchopneumonia that can have high morbidity and an important impact on milk production (in dairy herds), very little is published in the veterinary research literature regarding the problem. Outbreaks of significant respiratory disease with notable mortality caused by BRSV infection have been described in individual herds of adult dairy cattle.149,150 Respiratory disease and decreased milk production in adult dairy cows was associated with seroconversion to influenza virus and BRSV,253 but the authors could not confirm that influenza was contributing to clinical disease in the affected cows. The lack of research on respiratory disease in adult cows is likely a result of the fact that other diseases that affect reproductive performance and milk yield are of greater economic significance in this class of cattle, but the few published descriptions of respiratory disease outbreaks confirm that the problem can on occasion be of major importance to individual herds. Little research has investigated the effects of infectious respiratory disease in adult cattle on productivity.
BRONCHOPNEUMONIA OF SHEEP AND GOATS
Little information has been published regarding the epidemiology of bronchopneumonia in sheep and goats. The 2001 National Animal Health Monitoring System (NAHMS) survey of producers indicated that 7% of death in adult sheep were due to respiratory disease, whereas 12% of lamb deaths were due to respiratory disease.437 Feedlots reported that shipping fever pneumonia accounted for 13% of feedlot lamb deaths, and other respiratory disorders accounted for 29% of lamb deaths.438
Host and Environmental Risk Factors for Respiratory Disease
ENZOOTIC CALF PNEUMONIA
The documented host and environmental risk factors of dairy and veal calf bronchopneumonia involve inadequate passive transfer of immunoglobulins, nutritional deficiencies, and adverse environmental conditions (Fig. 31-64). Calves are generally affected at less than 2 months of age. Successful passive transfer is the foundation of protection against pneumonia at this age and older and is effective except in situations in which the other risk factors are so severe that even calves with high concentrations of passively acquired immunoglobulins are at high risk. Successful passive transfer requires good-quality colostrum and adequate volume (4 L in the first 12 hours for 45-kg calves). In addition, regular herd vaccination, especially in dry cows, should increase the levels of specific antibody in calves, provided passive transfer is accomplished. Vaccination of calves may produce a protective immune response in calves that prevents or limits the severity of ECP from certain infectious agents. Historically it has been recommended that vaccination be delayed until maternal immunity has waned, but recent research indicates that calves can be primed for a protective immune response by vaccination in the face of maternal antibodies in at least some cases (see further discussion on p. 640).
Nutritional problems that predispose to calf pneumonia include deficiencies of energy, protein, vitamins, or the minerals necessary for the immune response. Deficiencies of copper, selenium, zinc, manganese, iron, and vitamins A and E are of special concern. Dairy managers sometimes create energy and protein deficiencies by feeding calves a low volume of milk during the first few weeks of life to minimize the incidence of neonatal diarrhea.
The calf’s immediate environment affects the calf in a number of ways. Ambient temperature is an important factor affecting dairy calf health.439 Cold weather is especially detrimental to young calves, which have little body insulation. Increased humidity or precipitation in the calf’s environment worsens the calf’s ability to maintain thermal neutrality. Warm weather can also be undesirable, because young calves are capable of greater perspiration per pound of body weight than adults, and warm weather may predispose young calves to dehydration.439
The bacterial content of air in cattle barns can be as high as 106 organisms per cubic meter.440 Disease incidence can be affected by length of pathogen survival time as an aerosol and the concentration of the pathogen in the air space. Humidity is an important limiting factor affecting pathogen survival. The optimum zone for limiting survival time of bovine pathogens is 55% to 75% relative humidity. Adequate fresh airflow into the calf’s environment is important in limiting humidity and reducing the concentration of noxious gases and pathogens. The flow of air should be from younger, more susceptible cattle to older, less susceptible cattle to limit moving pathogens from older cattle to younger cattle. Adequate fresh airflow and proper directional movement of air are important goals of ventilation.
Calf housing with overcrowding of calves or excessive stocking densities results in increased transmission of pathogens, especially if there is mixing of age-groups. Overcrowding also puts additional stress on the building ventilation through buildup of noxious gases and pathogens. Cleaning of calf crates with high-pressure water sprayers is associated with new cases of pneumonia several days later. Bates and Anderson441 have recommended standards for ventilation, including building location, fan capacity and location, intake location and design, temperature regulation, air space needed and airflow directions, and acceptable humidity levels. Individual calf hutches that are properly located provide the calf with adequate fresh air free of pathogens and noxious gases and overcome many of the problems found with calf barns. Calves moved out of hutches can then be put into small groups (seven or eight calves) separated from older cattle through use of super hutches. Like the calf hutch the super hutch also serves to limit pathogen transmission and buildup of noxious gases in this susceptible group of calves. Alternatively, calves can be moved out of hutches and placed in pens in pole sheds provided groups are small (7 to 10 calves), air quality is good, and proper segregation from older age-groups is maintained. Standards of housing for calves are shown in Table 31-11.
Table 31-11 Standards for Adequacy of Ventilation of CalfHousing434,441
| Housing Type |
Ventilation Standards |
| Calf hutch |
One calf per hutch |
| |
Minimum of 4 ft between hutches |
| |
Hutches further than 50 ft from exhaust outlets of other buildings |
| |
Hutches 10 ft from fenced enclosure with older cattle |
| Mechanically ventilated calf barn |
200 ft3 of air space per calf
Barrier walls to separate age-groups of cattle housed in building |
| |
Fan capacity to achieve four air changes per hour in winter, 15 air changes per hour in spring and fall, and 40 air changes per hour in summer |
| |
Humidity levels between 50% and 80% |
| |
Ammonia levels less than 10 ppm |
| |
Intake velocity for fresh air intakes of 200 to 800 feet/sec |
| Naturally ventilated calf housing |
Adjustable opening on sidewalls
Open ridge for dual slope (2 in/10 ft of building width) |
| |
Eve opening for monoslope buildings (2 in/10 ft of width) |
| |
Barrier walls between age-groups (especially first postweaning age-group and older animals) |
| |
Separate waterer for first postweaning age-group |
Sivula and colleagues434 found that 80% of calf barns provided housing that failed to meet adequate standards of ventilation and housing441 regardless of whether calves were housed individually or in groups. In addition, calf housing in which calves shared the same air space as adults never met the adequate standards of ventilation and housing.434 A much higher percentage of calf housing that used calf hutches met these adequate standards of ventilation and housing, and virtually 100% would have been adequate housing if the hutches had been positioned correctly.434 Calves raised in inadequate housing have significantly poorer growth rates than do calves raised in housing that is considered adequate,434 which emphasizes the importance of adequate housing. The percentage of producers who use calf hutches continues to increase, as the benefits of their use are documented and published.
Veal calves are at greatest risk because they are reared in rooms that are filled to high stocking density with calves from multiple dairies that put minimal effort into ensuring that the calves are fed adequate amounts of high-quality colostrum. Similar problems occur on “calf ranches,” where hundreds to thousands of very young dairy calves are raised to the point at which they are old enough to be returned to dairies or sent on to feedlots. A high proportion of calves that enter calf ranches have FPT. Also, the accumulation of a large number of calves with a variety of histories and from a variety of sources causes problems similar to those encountered with older animals in feedlots. The respiratory defenses of the calf lung include aerodynamic filtration, particle removal, adhesion resistance, secretory defenses, and cellular defenses. The physical respiratory defenses (filtration, removal, adhesion resistance) can be compromised by inhaled noxious gases, temperature extremes, dehydration, and viral infections causing impairment through damage to the mucosal lining of the upper respiratory tract, or by increased viscosity of respiratory secretions. Noxious gases, such as ammonia, methane, hydrogen sulfide, and carbon dioxide, which become increased from inadequate manure handling or poor ventilation, can also impair secretory defenses via damage to the mucosal lining and impair cellular defenses by direct effect on alveolar macrophages. Viral infection may also damage the mucosal lining and impair production of secretory defenses, such as lysozymes, lactoferrin, complement, or secretory immunoglobulin. Viral infections can also have a direct effect on cellular defenses, including alveolar macrophages, and for some viruses, the neutrophils. Stress caused by overcrowding, temperature extremes, commingling, surgical procedures, or vaccination may impair cellular defenses and immunoglobulin production and enhance bacterial adherence.
Calves that have neonatal diarrhea are at greater risk of pneumonia.435 Thus risk factors that are unique to neonatal diarrhea, such as poor sanitation in the calving area, must be added to those outlined in Fig. 31-64 as possible risk factors for dairy calf bronchopneumonia. Calves born in loose housing have a higher risk of illness than those born in individual maternity pens. Treatments at birth can affect subsequent calf health. All dairy calves should have the navel treated. Injections of vitamin A and iron at birth may increase the disease resistance of dairy calves, which can be deficient in these nutrients at birth.
Other risk factors associated with ECP include large herd size, weather extremes (hot and cold), birth to a heifer, and low antibody titers to certain respiratory pathogens.430,435 Pneumonia of beef calves on the farm has the greatest incidence after weaning. The occasional outbreaks of pneumonia in suckling beef calves may be associated with adverse weather, parasitism, or nutritional deficiencies.
SHIPPING FEVER PNEUMONIA
Risk factors of feedlot calf bronchopneumonia are active in three areas: (1) at the farm of origin, (2) during transit, and (3) in the feedlot305,442,443 (Fig. 31-65).
Several on-farm management practices have a large impact on feedlot cattle pneumonia. Weaning, creep feeding, and performance of routine surgeries at least 3 weeks before shipment have been shown to reduce morbidity rate 20% to 25%.443 Vaccination on the farm against respiratory pathogens would be expected to reduce the incidence of feedlot cattle pneumonia but has not always been beneficial. Other farm factors are deficiencies of nutrients necessary for normal immune responses. Pneumonia has a higher incidence in young calves, and severe outbreaks sometimes occur in early weaned calves that enter feedlots.
Risk factors that are active during transport from the farm to the feedlot include sale through auctions, feeding of low-energy diets before shipping, and prolonged time in market channels (sales barns, transport vehicles). Excessive dehydration or “shrink” from transport has been shown to account for significant morbidity and mortality in the feedlot. Movement through multiple auctions greatly increases the risk of pneumonia.
Feedlot risk factors that influence morbidity and mortality rates include processing procedures, numbers of calves and number of different origins of calves per pen, diet, on-arrival surgeries (e.g., dehorning), and environmental conditions. Calves that arrive in the feedlot with moderate levels of antibodies against the respiratory viruses have been shown to have a decreased incidence of pneumonia, indicating that natural exposure or vaccination at the ranch, rather than the feedlot, is protective. Mixing of calves from different origins and filling of feedlot pens with calves over a prolonged period increase mortality and morbidity rates because infectious agents spread more readily in large populations. Starting calves on diets containing 75% concentrate or greater or feeding corn silage as a major dietary component during the first month is associated with higher mortality and morbidity rates, probably because of an inhibition of alveolar macrophage function caused by acidosis. Nonprotein nitrogen, such as urea, fed to stocker calves at any time or to feedlot calves on arrival is associated with increased levels of pneumonia. The addition of antibiotics to the drinking water of newly arrived feedlot cattle has been shown to be detrimental to their health, possibly because of decreased water consumption.
As mentioned in the earlier discussion of respiratory defenses of dairy calves, endogenous corticosteroid release caused by physical stressors (overcrowding, mixing of calves, surgical procedures, starvation, dehydration), environmental stressors (weather extremes), and damage to respiratory defenses (vehicle exhaust, environmental dust, noxious fumes, acidosis, dehydration, and viral infections) may also weaken or overwhelm the respiratory defenses of feedlot calves.
BRONCHOPNEUMONIA OF ADULT CATTLE
Studies confirming risk factors for bronchopneumonia of adult cattle in modern North American production systems are lacking, but it is assumed that factors are similar to those described previously for calves and for cattle entering feedlots.
BRONCHOPNEUMONIA OF SHEEP AND GOATS
Many of the risk factors that have been found to predispose to bronchopneumonias of sheep and goats are similar to those affecting cattle.220 Pneumonia is more common in younger animals, after shipping or storms, and under crowded conditions. In addition, cold stress is an important risk factor for pneumonia in young lambs and recently shorn adult sheep and goats. In contrast, heat stress may predispose to pneumonia in unshorn sheep that do not have access to shade. Pneumonia also is associated with semiconfinement or total confinement of sheep in poorly ventilated barns.
Prevention and Control of Ruminant Respiratory Disease
MANAGEMENT PRACTICES TO MINIMIZE ENZOOTIC CALF PNEUMONIA
Bronchopneumonia of dairy and veal calves is prevented by eliminating or altering as many predisposing risk factors as possible. Control is based on healthy, well-vaccinated dams giving birth in maternity facilities that limit pathogen exposure. Good colostral management is needed to ensure adequate passive transfer. The calves must have their navels disinfected and be moved to calf housing that limits pathogen exposure by overcrowding and direct contact, provides good-quality air, and protects the calf from environmental extremes.
Evaluation of the microclimate of housed calves is of critical importance in the control and prevention of pneumonia. Equipment useful for evaluating mechanically ventilated buildings includes a smoke generator to visualize airflow patterns, an anemometer to measure air velocity, a psychrometer to determine air temperature and relative humidity, and a gas detector kit to measure ammonia concentrations in the air. Agricultural engineers from animal science departments at regional universities can be often be contacted for guidance in the use of these tools. Naturally ventilated buildings should also be evaluated for adequate sidewall and ridge openings, adequate segregation of age-groups, and stocking densities. Calf hutches that are managed properly provide most, if not all, housing needs required to minimize calf pneumonia. Hutches should be evaluated for proper placement in relation to other hutches, other buildings, prevailing winds, exposure to sun, bedding and draining, and ventilation of the hutch.
After weaning, calves need to move to housing that allows small groups to be segregated from older age-groups. Sharing air space or having direct contact with older calves or adult cattle is an important risk factor for calf respiratory disease. Super hutches or pens in pole sheds separated by barrier walls can effectively provide this postweaning housing. Calves should be fed proper nutrition for protein, energy, minerals, and vitamins and should be vaccinated appropriately. Vaccination programs are often used in dairy calves beginning at 1 to 2 months of age in situations where housing or mixing of calves results in a high incidence of ECP. More information regarding the use of vaccines for preventing infectious respiratory disease is presented in the following sections.
MANAGEMENT PRACTICES TO MINIMIZE FEEDLOT PNEUMONIA
Preconditioning
Preconditioning of calves is an attempt to eliminate certain risk factors that influence the occurrence of feedlot cattle pneumonia. Preconditioned calves are weaned well in advance of shipment to feedlots. They are trained to eat some grain from feed bunks and drink water from tanks (this is especially important for calves raised on rangeland where feed bunks or water tank may not be seen). Calves are castrated and dehorned, treated for internal and external parasites, and vaccinated against respiratory pathogens, with one dose of vaccine administered before weaning and the second dose administered 2 to 4 weeks after weaning. The exact details of a preconditioning program can be tailored to the needs and characteristics of the individual herd of cattle. A variety of preconditioning protocols have been described, and most of the major companies that sell vaccines have recommended standardized preconditioning programs. Also, in many areas of the United States and Canada, state, provincial, or regional groups have become established that offer a standardized preconditioning program for local cattle producers. Typically some sort of validation that calves have received preconditioning is available through these organizations. Some of these groups also help the participating producers market the preconditioned calves in large, relatively uniform groups, which helps the calves bring better prices. In some cases, producers can retain ownership of their cattle after they enter the feedlot, and they receive data regarding health and performance of their animals throughout the feeding period and at slaughter. Information about local preconditioning programs can usually be obtained from agricultural extension agents or from faculty who specialize in beef cattle management and health at colleges of animal science or veterinary medicine.
The value of preconditioning has been debated over the decades. A summary of eight studies comparing the health of preconditioned calves with that of control calves indicated that, on average, preconditioning reduced morbidity rate by 23% and mortality rate by 50% in the feedlot.443 However, preconditioned cattle do not always escape disease once they enter feedlots, and they sometimes experience outbreaks of high morbidity and mortality not unlike those seen in high-risk calves that are not preconditioned. Moreover, even when preconditioned calves experience less disease, their performance in the feedlot is not always better than that of nonpreconditioned calves,444 or they may not always perform well enough that a profit is returned that is greater than the cost of preconditioning.445 An evaluation of data collected over 8 years by the Texas A&M Ranch to Rail program showed that preconditioned cattle returned on average $90 more per head than nonpreconditioned cattle. However, in some years the return was better for preconditioned cattle than in other years.306 These data emphasize that preconditioning is likely to pay in the long run, but it may not always pay for individual groups of cattle in a single given year. Because of year-to-year uncertainty in the economic advantage of preconditioned cattle, producers who sell preconditioned calves may not always be able to sell calves at a price that provides a profit over the cost of preconditioning. Marketing preconditioned calves through state or provincial organizations that have gained the respect of cattle buyers and that allow the marketing of large numbers of uniform cattle may be the best way for the producer to realize a profit from preconditioned calves. Retaining ownership of calves sent to feedlots may also allow the producer to realize better profits, but this requires a relationship with a feedlot that will manage the calves in ways that will continue to optimize health once the calves enter the feedlot.
Backgrounding
Another practice similar to preconditioning is the practice of backgrounding. In backgrounding, weaned beef calves from a variety of sources (often purchased from multiple sale barns) are accumulated and processed in a manner similar to that carried out for high-risk calves at feedlot entry (Box 31-5). Calves are then sorted by size and type and sold to feedlots, usually within a few days of arriving at the backgrounding operations. A variation of the backgrounding operation is the stocker operation; in stocker operations calves are processed and then “stocked” onto pastures, often with supplemental concentrate feeding, to graze and grow for a few weeks to months before shipment to feedlots. The difference between preconditioning and backgrounding is that preconditioning occurs on the farm where the calf is born, or at least before the calf is mixed with other calves from different sources, with some aspects of treatment such as the first dose of vaccines ideally being administered before weaning. Calves that enter backgrounding or stocker operations often have uncertain health histories and have often been weaned immediately before purchase. Because of this, disease outbreaks of high morbidity and mortality are not uncommon on some backgrounding operations. The backgrounder or stocker operator is thus taking on some of the risk of high-risk calves and is theoretically providing the feedlot with animals more likely to grow well with minimal disease than if they entered the feedlot straight from the auction market.
Box 31-5 Examples of Processing Protocols for High- and Low-Risk Cattle Entering Feedlots
BRSV, Bovine respiratory syncytial virus; BVD, bovine virus diarrhea; IBR, infectious bovine rhinotracheitis; PI3, bovine parainfluenza virus type 3.
PROCESSING PROTOCOL FOR HIGH-RISK, FALL-WEANED, AUCTION MARKET CALVES
1
Modified live IBR, BVD, BRSV, and PI3 vaccines
2
Eight-way clostridial bacterin
3
Ivermectin type “pour-on” antiparasitical
4
Mannheimia haemolytica ±
Pasteurella multocida vaccine
5
Haemophilus somnus vaccine (if significant problem in past)
8
Bulls castrated (if needed)
9
Tip dehorning (if needed)
10
“Temp” all incoming cattle and sort by temperature (e.g., >105° F (40.5° C)—treat with tilmicosin; all others treat with long-acting oxytetracycline)
PROCESSING PROTOCOL FOR HEALTHY YEARLINGS PREVIOUSLY FED IN BACKGROUNDING OPERATIONS
1
Modified live IBR, BVD, BRSV, and PI3 vaccines
2
Eight-way clostridial vaccine
5
Ivermectin type “pour-on” if days on feed will allow for withholding times needed
Management at Feedlot Entry
It is not surprising that preconditioning does not always influence the occurrence of pneumonia in cattle after they enter the feedlot, because it has no effect on many risk factors active during transit and in the feedlot. Therefore optimal control of feedlot pneumonia should begin with preconditioning and continue with avoidance of auction yards, minimization of transport time to the feedlot, limited mixing of calves from different sources, limited number of calves per pen, control of dust, and careful diet management in the feedlot. Correction of vitamin and/or mineral deficiencies if known or detected, using vitamin injections in entering calves, may be a very important aspect of prevention of feedlot pneumonias that are associated with immunodeficiencies of nutritional origin.
Processing procedures on arrival at the feedlot affect the incidence of pneumonia. General recommendations for handling incoming cattle can be made that are appropriate for any category of animals. In a receiving program, rest, rehydration, and ruminal restoration need to be addressed because cattle are physically and psychologically stressed by the marketing and transportation processes. It is useful for these cattle to be rested for 12 to 24 hours before processing to allow the immune system to overcome the effects of stress. Prolonged holding before processing is associated with increased illness, and holding times over 48 hours should be avoided. Holding pens should be clean and dry or have dry bedding (if pens are wet from excessive precipitation), because this allows all cattle to lie down and rest. Shelter from wind, sun, rain, and dust should also be present in the receiving pen.
Holding pens should have 150 to 200 square feet of pen space per animal and 12 to 16 inches of bunk space per animal and should be located close to the processing facility. Excess mixing of cattle in the receiving pens should also be avoided.
It is important that incoming cattle have access to clean, fresh water. Raised spigots have been suggested as a way to teach incoming cattle to drink out of automatic watering devices because cattle will be attracted to the sound of splashing water. Incoming cattle should also be offered good-quality, long grass hay on arrival. This is the most similar to what cattle are used to on range. Hay is the best foodstuff for restoring or refilling the rumen. Hay can be put in the feed bunks as well as in feeders in the pen as a way of teaching cattle to eat out of bunks. Hay feeders may also be put along the pen perimeter to decrease walking the fence line and encourage eating. The starter ration is an important source of energy and should be highly palatable. The proportion of the starter ration dry matter that is forage is not usually less than 50% to prevent problems of acidosis. Starter rations often contain a coccidiostat, because coccidiosis can occur in calves after commingling.
Processing protocols may be tailored to the category of the incoming cattle. Pharmaceutical processing options include vaccination (respiratory and nonrespiratory), vitamin injections, implanting, deworming for internal parasites and acaricides for external parasites (may be the same product for both), long-acting antibiotic therapy, drugs for aborting pregnant heifers, and probiotic administration. Management procedures for processing include ear tagging, branding, tail trimming, castration, tip dehorning, and temperature sorting. Some management procedures such as castration and dehorning could be left for a later time—for example, at reimplanting at 70 to 90 days on feed (if cattle are expected to be on feed for more than 150 days). “Temping” on arrival can be very useful, because even cattle that look bright can have very high temperatures and thus can be identified as “sick” by this procedure. Examples of processing protocols for high- and low-risk cattle are shown in Box 31-5.
Metaphylactic Antimicrobial Therapy
The term metaphylaxis refers to the administration of an antimicrobial to a group of animals that may be in the early stages of bacterial pneumonia or that are at significant risk of developing bacterial pneumonia. Thus the antimicrobial drug administered may have either therapeutic or prophylactic effect, depending on the state of each animal treated. Metaphylactic administration of antibiotics to feedlot cattle on arrival in order to prevent bacterial bronchopneumonia in feedlot cattle has been shown in many studies to decrease morbidity and mortality in groups of high risk cattle.446,447 It is useful to consider the theoretic mechanisms by which metaphylactic administration of antimicrobials may limit disease. Soon after cattle are shipped, M. haemolytica A1 can proliferate in large numbers in the upper respiratory tract. This period of replication is a crucial phase because large numbers of bacteria can be inhaled into the lung, allowing colonization, proliferation, and production of virulence factors. There appears to be a short time after arrival in the feedlot when cattle that will subsequently develop bacterial bronchopneumonia have large numbers of M. haemolytica A1 present in their upper respiratory tract. Antimicrobial therapy timed to coincide with this pathologic event and designed to provide therapeutic levels in respiratory tissues is aimed at reducing the number of M. haemolytica A1 present in the upper respiratory tract of calves, which should also limit colonization of the lung and prevent horizontal transmission of M. haemolytica from calf to calf. Long-acting antibiotics have been reported to significantly alter the number of calves from which M. haemolytica can be cultured.270,448 An additional rationale for metaphylaxis on arrival is based on the epidemic curve of fatal disease onset for bacterial bronchopneumonia, which shows that feedlot calves dying from fatal fibrinous pneumonia are already sick on arrival or become ill within days of arrival. Although metaphylaxis through feed and water was used in the past, it is currently recommended that metaphylaxis be administered by use of injectable antimicrobial agents. The ability of these drugs to reach therapeutic levels quickly in all animals gives them a clear advantage given the previously discussed rationale for bacterial bronchopneumonia metaphylaxis.
A number of trials have been published that examine various antimicrobial agents and their effectiveness for bacterial bronchopneumonia metaphylaxis. Most of these studies have examined both health and production values, and a variety of products have been shown to be efficacious in decreasing respiratory morbidity and mortality and sometimes in improving production parameters.449-451 Tilmicosin or long acting oxytetracycline, both of which provide therapeutic levels of drug for 3 days, are most commonly used for metaphylaxis in feedlot cattle.
Similar use of long-acting antimicrobials in dairy calves may also be of value. The disease process does not have the same narrow windows of therapeutic intervention that occur in calves entering the feedlot. Dairy calves properly managed in maternity facilities and raised in hutches rarely experience any respiratory disease until they are moved to postweaning housing. Injectable mass medication is often used at the time calves are moved and then again at some time later (7 to 10 days after entering the postweaning housing) as a means of controlling postweaning ECP.
MANAGEMENT PRACTICES FOR PREVENTING SHEEP AND GOAT PNEUMONIA
Prevention of pneumonia in sheep and goats is also based on altering the risk factors that predispose to pneumonia. Minimizing cold and heat stress, providing properly ventilated housing, avoiding overcrowding, and avoiding long transports in adverse weather aid in prevention. Mass medication can be used to control outbreaks of pneumonia in flocks. Injectable long-acting oxytetracycline is often used subcutaneously at 10 mg/kg. Tilmicosin can also be used for mass medication therapy for sheep, but tilmicosin should never be administered to goats, as it can result in fatal reactions. Feedlot lambs are sometimes administered metaphylactic therapy (long-acting oxytetracycline) on arrival.
Vaccination To Prevent Respiratory Disease
Factors That Affect Success of Vaccination
Many factors can affect the success of vaccination. For a vaccine to successfully prevent disease, the vaccine must induce a protective immune response against a pathogen to which the host will be exposed. Apparent vaccine failure is often blamed on the product or the manufacturer, but in many cases other factors may have led to vaccine failure. Possible causes of apparent vaccine failure that must be also be considered when vaccinated animals contract disease are related to the administration of the vaccine, the ability of the host to respond to vaccination, and to the nature of pathogen exposure. Examples are listed in Box 31-6. Although the points listed may be obvious, it is important to carefully consider and rule out these problems when a vaccine has failed to prevent respiratory disease before making a quick switch to another brand or type of vaccine.
Box 31-6 Reasons for Apparent Vaccine Failure
FACTORS RELATED TO ADMINISTRATION OF THE VACCINE
Improper storage of live vaccines
Residual chemical disinfectant in reusable syringes
Using live vaccine reconstituted days or weeks previously
Administration into wrong anatomic location
Administration of antibiotics with live bacterial vaccines
Incorrect timing of vaccine administration
Animals already incubating disease
Failure to allow time for immune response to occur
Administering booster too early, too late, or not at all
FACTORS RELATED TO ABILITY OF HOST TO RESPOND TO VACCINATION
Age (very young or very old animals)
High levels of maternal antibody
Immunocompromise resulting from concurrent disease or stress
Poor air quality or other environmental insults
FACTORS RELATED TO PATHOGEN EXPOSURE
Disease caused by pathogens not included in vaccines
Antigenic variation (strain variation) of naturally occurring pathogens
Overwhelming pathogen exposure
In addition to the factors just discussed, vaccine characteristics can also affect the success of a vaccination program. Once the decision has been made regarding choice of pathogens to be included in the vaccine, the use of a live versus killed vaccine must be considered. Much research has evaluated the immune response of animals to modified live versus killed (inactivated) vaccines, and the subject has been the focus of much discussion. To summarize:

Modified live virus (MLV) vaccines in general stimulate cell-mediated immunity, important for an effective immune response to most viral pathogens, as well as effective humoral immunity.

Live vaccines can sometimes elicit protective immunity with only a single dose of vaccine.

Live vaccines contain a smaller dose of organisms, because the organisms in the vaccine are expected to replicate at least minimally.

Live vaccines generally do not require adjuvants, which are often a major cause of adverse reactions after vaccination (however, some MLV vaccines do contain adjuvants to improve the immune response).

Live vaccines are generally cheaper.

Disadvantages of live vaccines include the possibility of causing abortion in pregnant animals, transmission to nonvaccinated animals, exacerbation of morbidity in sick or immunocompromised animals, and reversion to a more virulent form with disease possibly occurring.

Another disadvantage of live vaccines is that they can be inactivated in a short time by exposure to heat or light.

In contrast, inactivated vaccines do not generally stimulate effective cell-mediated immunity, although certain adjuvants can overcome this.

Inactivated vaccines often stimulate high levels of antibody.

Inactivated viral vaccines require at least two doses at a 14–28 day interval to induce an effective anamnestic response to subsequent challenge.

Adjuvants in killed vaccines can cause adverse reactions.

Inactivated vaccines are safe in pregnant or immunocompromised animals, and storage requirements are not as rigorous for inactivated vaccines as for MLV vaccines.
Much of the above has been determined using viral vaccines; the situation with bacterial vaccines is less well characterized. Modified live bacterial vaccines may stimulate both humoral and cell-mediated arms of the immune response better than inactivated products (bacterins). However, for extracellular bacterial pathogens such as M. haemolytica, humoral immunity is the most critical component of an effective immune response. Past evidence has suggested that live M. haemolytica vaccines are superior to inactivated products; however, recent comparisons have shown some nonliving products to provide superior resistance to experimental challenge. Live bacterial vaccines can be inactivated if animals are given antibiotics at or near vaccination.
It is important to note that all vaccines for a given disease are not necessarily equal. If a vaccine is inactivated, the means by which it is inactivated can influence antigenicity. Moreover, a wide variety of adjuvants exist, and the adjuvant included in a vaccine can greatly influence the type and duration of immune response elicited. Adjuvants are included in inactivated and some modified live products. The choice of adjuvant to include in a vaccine is the subject of much creative thinking and research effort by manufacturers; this is emphasized by the fact that it is often very difficult to obtain detailed information about the adjuvant contained in a vaccine.
Impact of Maternal Antibody on the Response to Vaccination
For many decades veterinarians have been taught that young animals cannot be effectively vaccinated when they have moderate levels of maternal antibodies obtained through passive transfer after birth. This was based on numerous research studies that showed that vaccination of young animals with maternal antibodies did not lead to the expected increase in serum antibodies to the agent in the vaccine. However, an important finding is that some vaccines appear to prime calves for an improved response to subsequent challenge, even when vaccination occurs in the presence of maternal antibodies. In such cases vaccination does not induce an increase in serum antibody titer within 2 weeks of vaccination, but when calves are vaccinated or challenged at a later date, they respond with what appears to be an anamnestic response as measured by increased serum antibodies, or protection against disease resulting from experimental or natural challenge, as compared with unvaccinated calves.351,352,452,453 In general, a response was induced most reliably when modified live vaccines were administered and when the calves received at least two doses of vaccine at an appropriate (2- to 4-week) interval. Similarly, calves exposed to live pathogens in the face of maternal antibody can develop a protective immune response, even though they do not seroconvert after exposure.454,455 It has been shown that a cell-mediated immune response can be measured even when there is no evidence of humoral response in calves infected in the face of maternal antibodies.455 Although multiple studies show that calves can be primed for an anamnestic immune response in the face of maternal antibodies, if very high titers of maternal antibody are present, priming by the vaccine can be blocked, as traditionally understood.453
This information suggests that at least in some cases a protective immune response can be initiated in calves when the first dose of vaccine is given in the face of maternal antibodies; a cell-mediated response may occur even if calves do not have an increase in serum antibody titer after vaccination. This is particularly useful in the context of pneumonia in nursing dairy or beef calves, as it supports the concept that vaccination can improve the immune response to challenge even if calves have some level of maternal antibodies at the time of vaccination. More research is needed to confirm which vaccines have this effect and to determine how this information should be used to guide vaccination recommendations for calves. It is also not known if this information can be extrapolated to sheep and goats.
Efficacy of Vaccines for Preventing Ruminant Respiratory Disease
Information regarding the use of vaccines for individual agents that cause ruminant respiratory disease was presented earlier in the section describing the individual infectious agents. There has been considerable debate over the years regarding whether vaccines have any impact on ruminant respiratory disease.456 The literature on the subject is extensive, and interpretation of the data is complicated by the fact that there is much variation among research studies in the type and number of animals studied, the nature of animal management, and the outcomes measured as evidence of efficacy. There are three major types of research studies by which vaccine efficacy can be measured: studies that measure production of antibodies or in vitro cell-mediated immune responses in vaccinated animals; studies that measure the resistance of vaccinated animals to experimental challenge with viral or bacterial pathogens; and studies that measure the impact of vaccination on health and productivity of animals in a conventional field setting (field trials). Of the three methods, field trials are the most meaningful, but they are also the most expensive and difficult to undertake. When they are undertaken, it is often in small numbers of animals under management practices that may not be representative of practices in other regions. Moreover, they rely on the natural occurrence of the disease in question in the animals under study. A well-designed field trial can be very expensive and time-consuming, and the results can be useless if no animals in the study contract the disease naturally.
In spite of these limitations the results of some field trials testing the efficacy of respiratory disease vaccines have been published. For some important pathogens (e.g., BVDV), there are still no large-scale field trials published that show evidence of efficacy in preventing respiratory disease. One trial showed an economic advantage to administering a modified live vaccine containing BHV-1, BVDV, PI3, and BRSV over the use of a modified live vaccine containing only BHV-1 in feedlot cattle,457 but it was not possible to determine which component of the multivalent vaccine was responsible for efficacy. Multiple field trials have been published that tested currently available vaccines against M. haemolytica311-314 and BRSV193-196; these are discussed further in the earlier sections on these specific pathogens. In general, some of these studies found that vaccination could prevent disease and sometimes improve productivity, and some of these studies could not find a beneficial effect of vaccination. It is important to remember that in nearly all of these field trials, the effect of vaccination to decrease all respiratory disease was measured. That is, in most cases, no effort was made to determine whether disease specifically caused by the agent included in the vaccine was affected by vaccination. Therefore the apparent lack of efficacy of vaccines in some studies may have been because animals developed disease caused by other respiratory pathogens.
Occasionally, in addition to evaluating the impact of vaccination on morbidity, mortality, and measures of productivity, investigators calculate the economic advantage or disadvantage of vaccination. When such calculations are made, it is important that the reader assess the assumptions made and determine whether they are appropriate. It has been noted that when the economic advantage of using a given vaccine is calculated, various scenarios should be considered that may prove the vaccine to be economically advantageous in some situations and disadvantageous in others.458
The mixed results of clinical trials published to date indicate that vaccines decrease respiratory disease, improve animal productivity, and save money for the producer in some cases, and in some cases they do not. This should not be a surprise when the multitude of factors that can converge to cause an outbreak of respiratory disease is considered. Vaccination should be seen as one component of a multifactorial approach to minimize animal disease; it should not be seen as a guarantee against all disease, all the time. Thus, the value of vaccination will depend on the risk of disease in the population in question, and the risk of disease will depend on the likelihood of pathogen exposure and the likelihood that other host and management factors can be modified to maximize the ability of the host to respond to vaccination and resist disease.
No single, simple recommendation for vaccination of ruminants for the control of respiratory disease can be made. The veterinarian must individualize each vaccination program to the situation at hand. Many factors need to be considered in formulating recommendations for vaccination, including type of production unit, age of animals, system of management, housing facilities, amount of stress imposed on animals, open or closed herd, type of ration, and level of sanitation. In addition, some infectious agents such as BHV-1, BVDV, and H. somni can cause other disease problems not associated with the respiratory tract. Thus the complete spectrum of disease caused by these agents must be considered in formulation of a vaccination program. Examples of respiratory vaccination protocols that could be used in a cow-calf operation and in a dairy are presented in Boxes 31-7 and 31-8.
BOX 31-7 One Example of a Respiratory Vaccination Protocol for Use in a Cow-Calf Herd
REPLACEMENT HEIFERS
Modified live BHV-1/PI3/BRSV/BVD with boost 2–4 weeks later, prior to breeding
COWS
Modified live BHV-1/PI3/BRSV/BVD prior to breeding
Boost with killed
or approved modified live* BHV-1/PI3/BRSV/BVD at pregnancy check
Consider
M. haemolytica/P. multocida vaccine at pregnancy check with boost one month prior to calving in herds with history of problems with pneumonia in nursing calves
CALVES
In herds with significant pneumonia in nursing calves, consider killed or
approved modified live*(preferable) BHV-1/PI3/BRSV/BVD and
M. haemolytica/P. multocida vaccine at 1–4 months of age with boost 2–4 weeks later, timed so that boost occurs 2–4 weeks prior to expected onset of calf pneumonia.
To Minimize Pneumonia in Calves Postweaning;
Killed or
approved modified-live*(preferable) BHV-1/PI3/BRSV/BVD one month prior to weaning with boost at weaning
Consider
M. haemolytica/P. multocida vaccine preweaning with boost at weaning in herds with a history of post weaning pneumonia, or if required for participants in defined preconditioning programs
BOX 31-8 One Example of a Respiratory Vaccination Protocol for Use in a Dairy
REPLACEMENT HEIFERS
Modified live BHV-1/PI3/BRSV/BVD with boost 2–4 weeks later, prior to breeding
COWS
Modified live BHV-1/PI3/BRSV/BVD prior to breeding
Consider boost with killed
or approved modified live* BHV-1/PI3/BRSV/BVD at dry off
Consider
M. haemolytica/P. multocida vaccine at 2 months prior to calving, with boost one month later, in herds with history of problems with pneumonia in nursing calves
CALVES
In herds with significant calf pneumonia prior to weaning, consider modified live
† BHV-1/PI3/BRSV/BVD and
M. haemolytica/P. multocida vaccine at 1–4 months of age with boost 2–4 weeks later, timed so that boost occurs 2–4 weeks prior to expected onset of calf pneumonia.
To Minimize Pneumonia In Calves Postweaning
Modified live BHV-1/PI3/BRSV/BVD one month prior to weaning with boost at weaning
Consider
M. haemolytica/P. multocida vaccine one month prior to weaning with boost at weaning in herds with a history of post weaning pneumonia, or if required for participants in defined preconditioning programs
Evaluating Reports of Vaccine Efficacy
Vaccines approved for sale in the United States must be proven by the manufacturer to be safe, potent, stable, and efficacious. Duration of immunity is an additional parameter that manufacturers are beginning to be required to address. Efficacy is usually determined by experimental challenge, using defined methods or methods approved on a case-by-case basis. For many bovine respiratory pathogens (e.g., BHV-1, M. haemolytica), experimental challenge protocols have been developed that can induce disease of reasonable severity. To be approved for sale, vaccines need to prevent to a significant degree clinical signs associated with such experimental challenge. However, whereas protection against experimental challenge is a useful indication of the possible efficacy of a vaccine under field conditions, few experimental challenge protocols closely model the field situation in terms of simultaneous occurrence of other stresses and concurrent infection with other pathogens. Thus, it is ideal to evaluate vaccine efficacy through evaluation of an appropriately designed field trial. The design of field trials has generally improved greatly in recent years, but in the case of many vaccines, no high-quality published field trials evaluating the efficacy of vaccination for prevention of BRD exist.
Veterinarians can arm themselves with useful information by evaluating published studies of vaccine efficacy in a critical manner. In some studies animals are vaccinated but never exposed to the pathogen. Serum and/or mucosal antibody levels may be measured, and markers of cell-mediated activity, such as lymphocyte blastogenesis, cytokine production, or cytotoxic T cell activity, may be assayed. Such studies can indicate the antigenicity of a vaccine, but one must consider whether the parameters measured are known to correlate strongly with protection against disease. More information can be gained when measurements of immune function are evaluated in light of natural or experimental challenge. In experimental challenge studies, relatively small groups of animals are vaccinated and later challenged with the pathogen contained in the vaccine. Although experimental challenge studies are rather artificial compared with the situation animals experience in the field, a high-quality experimental study has certain characteristics. Some questions to ask include the following: Was a nonvaccinated control group, identical to the vaccine group in all ways except vaccination status, included? Were control animals tested at the same time as vaccinated animals? If factors that could affect vaccination were present (e.g., maternal antibody), were affected animals divided equally between control and vaccine groups? Did experimental challenge result in disease in the control group? If not, it is impossible to say if vaccination had an effect on challenge. Were investigators who evaluated clinical or pathologic signs of disease blinded to the treatment status of the animals? This removes an important source of bias that otherwise can make data, particularly subjective data such as “depression” or “dyspnea,” suspect. Were statistical tests used to compare results of vaccine and control groups, and was the P value given to indicate likelihood that differences were due to chance alone? Other questions that can help determine the relevance of the experimental study to the field situation include the following: Was disease resulting from challenge clinically and/or pathologically similar to that seen in field cases? Was the vaccination regimen similar to that used in the field? How soon after vaccination were animals challenged? Did the time between vaccination and exposure mimic the field situation?
Field trials are characterized by allocation of animals in a natural “field” situation to either vaccine or control groups. Animals are treated accordingly and then followed for variable periods of time to determine whether disease occurs in vaccinated animals and, if so, whether vaccinated animals have disease less often, have disease that is less severe, or have improved production characteristics (e.g., average daily gain) after vaccination. When evaluating a field trial, consider the following questions to determine the value of the study: Were animals randomly allocated to control or treatment groups? This is critical; if there is no mention of randomization, it is difficult to gain useful information from the study because of the many types of bias that can affect the outcome. Were concurrent controls used, as opposed to historical controls? Historical controls are of much less value in determining vaccine efficacy, as many factors can affect disease outcome in a group of animals from year to year. How many animals were included in the study, and for how long did the study run? In general, trials with larger numbers of animals are more likely to reveal differences between vaccine and control groups. Were evaluators of disease blinded to the treatment groups to remove their bias in interpreting outcomes? Did disease occur in the control animals? One weakness of field trials is that natural disease must occur in the animals under study to determine the effect of vaccination on disease; the investigators have no control over this aspect of the study. If disease did not occur in at least the control animals, the vaccine cannot be evaluated for protection against the disease. Also, ask what outcomes were measured as evidence of protection against disease. In most cases of feedlot trials, fibrinous pneumonia morbidity and mortality are measured. If this was the case, identify how cases were identified, and determine if the definition is accurate. In many trials, total morbidity and mortality is also measured as an outcome. This may be considered a less reasonable outcome; for example, it may not be reasonable to expect BHV-1 vaccination to decrease deaths caused by ruminal acidosis. Production characteristics, such as rate of gain or feed efficiency, are also often evaluated, and net cost of vaccination, including estimated losses due to disease or loss of production, may be calculated. In these cases, evaluate how costs were estimated, and determine if estimates appear to be accurate and reasonable. Finally, veterinarians may want to consider whether the field trial was conducted under conditions similar to that seen in their practice; if so, the results may be more relevant to the needs of their clients (adapted in part from Ribble).459
ACUTE RESPIRATORY DISTRESS SYNDROMES
ARDS is any respiratory condition characterized clinically by a sudden onset of (usually severe) dyspnea (Fig. 31-66) with gross and histopathologic findings consistent with AIP. The characteristic gross findings of AIP are lungs that fail to collapse when the thorax is opened (Fig. 31-67) and that are heavy and have a firm, rubbery texture on palpation. Interlobular or bullous emphysema is usually present (see Fig. 31-54), and sometimes the cut surface of the lung has a shiny or wet appearance because of edema. In some cases affected lobules, which are dark red to purple, or sometimes grayish, are interspersed with normal-looking lobules, giving the lung a “patchwork” appearance. Although the gross findings are suggestive and often characteristic, other lung diseases can cause similar changes, so a diagnosis of AIP can be confirmed only by histopathologic evaluation. The histopathologic changes that confirm a diagnosis of AIP include alveolar hyaline membrane formation and fibrin deposition, alveolar and interstitial edema, and type 2 pneumocyte proliferation. Hemorrhage, emphysema, and interstitial inflammatory cell infiltrate can also be seen.
The term atypical interstitial pneumonia has often been applied to describe the changes characteristic of AIP, but it has been pointed out that the clinical and pathologic changes in affected cattle are not “atypical” but are rather “typical” of ALI.460,461 Use of the term “acute interstitial pneumonia” to describe the characteristic pathologic changes, and the term acute respiratory distress syndrome to describe the clinical picture in an animal with AIP of as-yet undetermined cause, is both accurate and all-encompassing. A diagnosis of AIP should be understood as a pathologic diagnosis rather than an etiologic diagnosis, because a variety of insults can lead to the same lung lesion of AIP.460,461
Acute Bovine Pulmonary Edema and Emphysema
Definition and Etiology
Acute bovine pulmonary edema and emphysema (ABPEE), classically known as fog fever, is an ARDS of adult (over 2 years old) cattle that are changed from dry, sparse forages to lush green pastures. It is caused by the conversion of L-tryptophan ingested in the lush forages to a pneumotoxic compound (3-methylindole [3-MI]), which leads to the development of pulmonary edema, alveolar epithelial hyperplasia, hyaline membranes, and emphysema.460
Clinical Signs
Adult brood cows are most commonly affected because this is the type of animal most likely to be subjected to the abrupt pasture change required to produce the condition. No breed is resistant.460 The type of pasture appears to be unimportant, as long as it is lush; ABPEE has been reported on a wide variety of grasses, alfalfa, rape, kale, and turnip tops.462 Signs usually occur within 2 weeks of the pasture change.460 In severe cases there is an acute onset of very severe dyspnea with a loud expiratory grunt, frothing at the mouth, mouth breathing, and tachypnea (35 to 75 breaths/min).462 The animals are obviously distressed (as opposed to exhibiting the typical depression that occurs with infectious diseases) and stand with the head and neck extended and elevated and the nostrils dilated.463 Temperature and heart rate may be elevated secondary to the severe dyspnea and hypoxia.463 On auscultation the breath sounds are usually surprisingly soft in view of the gross dyspnea and tachypnea; a few crackles may be heard.462 Even mild exercise increases the dyspnea and may precipitate collapse and death. As many as 30% of severely affected patients may die, usually within 2 days.462 Those that survive typically show a dramatic improvement after 3 days.462 Recovering patients and those that are less severely affected exhibit tachypnea (50 to 80 breaths/min), hyperpnea, harsh breath sounds, and crackles and wheezes, particularly in the caudal lung fields.462 SC emphysema may develop. The demeanor of the entire group tends to become more tranquil.462 In cattle that have repeated episodes of nonfatal ABPEE, a chronic respiratory condition characterized by diffuse pulmonary fibrosis and alveolitis may develop.464 For purposes of differential diagnosis, it is important to note that coughing is not prominent in the individual or the group.462 The main differential diagnoses are those diseases that cause ARDS in pastured adult cattle, usually in outbreak form. The association of ABPEE with typical management conditions (e.g., changes of pasture) and the absence of coughing, signs of sepsis, and adventitious lung sounds in early cases are also very important features. Primary considerations should include the other plant toxicities (moldy sweet potatoes, perilla mint, and possibly others) that can be differentiated only by identifying the source. An outbreak of BRSV infection or parasitic bronchitis could cause similar clinical signs and pathology, but these are characterized by more coughing, signs of depression, and more prominent adventitious sounds; fever is common in animals with acute BRSV infection.
Pathophysiology
L-Tryptophan in lush forages is converted by ruminal microorganisms to indole acetic acid and eventually to 3-MI, which is rapidly absorbed from the rumen into the blood. Metabolism of 3-MI by the cytochrome P-450 mixed function oxidase system in the nonciliated bronchiolar epithelial (Clara) cells and type 1 pneumocytes results in one or more highly reactive intermediates that bind to intracellular proteins or other macromolecules. It is thought that these intermediates produce the damage to these cells. These intermediates are detoxified by conjugation with glutathione.465 Cellular damage results in degeneration, necrosis, exfoliation of type 1 pneumocytes and Clara cells, and edema. These lesions in turn cause hyaline membrane formation, proliferation of type 2 pneumocytes, and, to a lesser extent, proliferation of Clara cells.465 The proliferation of type 2 cells is also known as adenomatosis. Emphysema is probably secondary to the severe dyspnea.465
Epidemiology
As indicated, ABPEE is consistently related to management practices in which hungry adult cattle are suddenly moved from sparse, dry grazing to lush green pastures. The British name “fog fever” arose from the association of the disease with “fog” pastures, which are the lush green regrowth pastures after hay or silage has been cut. The problem usually occurs in the fall. In the typical pattern in the western United States, cattle are moved from dry summer range onto irrigated or fertilized aftermath pastures.460 The disease usually appears as a herd outbreak, but individuals may be affected to widely varying degrees; morbidity rate commonly approaches 50%, with a case fatality rate as high as 30%.466,467 Nursing calves are apparently not at risk, and yearlings are less susceptible than adults.468
Necropsy Findings
In animals that die of ABPEE, ecchymotic to petechial hemorrhages occur in the larynx, trachea, and bronchi, and frothy fluid is present in the airways. The gross and histopathologic lung lesions are consistent with AIP. Congestion, edema, and hyaline membranes cause deep red-to-purple coloration of the cranial lung lobes and a smooth, glistening appearance to the cut surface. Interstitial emphysema with large bullae and gelatinous yellow interlobular edema is common. Histologically, eosinophilic hyaline membranes line alveoli and alveolar ducts, and there edema and proliferation of type 2 alveolar epithelial cells are present.460 In animals that are killed after 3 to 4 days, emphysema and edema are less obvious, and the lungs tend to be light brown, firm, heavy, and rubbery. There is severe diffuse alveolar epithelial hyperplasia (“adenomatosis”), and large mononuclear cells, multinucleated giant cells, and hyaline membranes are present in alveolar spaces. Edema, eosinophils, and inflammatory cells occupy the septa.460
Diagnosis
Diagnosis is made based on the history of recent exposure to lush green forage and typical clinical signs and pathology in fatal cases. Thoracic radiographs could be used to identify changes consistent with interstitial pneumonia in valuable individual animals. The results of TTA or BAL cytology have not been reported for cattle with ABPEE, but a nonseptic mixed inflammatory cell response would be expected. There are no unique hematologic or biochemical changes.463 A stress leukogram is often seen.
Treatment and Prevention
The stress of handling cattle can precipitate further losses. Some authors maintain that most cases in an outbreak occur within 4 days, that removing the herd from the pasture does not prevent additional cases, and that leaving the herd on the pasture does not result in additional cases; consequently the recommendation has been to handle severely affected cattle only if necessary to remove them to shade or to slaughter.467 Others460,468 recommend careful removal from the offending pasture. Antihistamines, corticosteroids, epinephrine, atropine, diethylcarbamazine, and diuretics are alleged to be of palliative value,460,470 but none of these has been properly tested.460 Pretreatment with antagonists to postulated mediators of inflammation, including acetylsalicylic acid, mepyramine, sodium meclofenamate, diethylcarbamazine citrate, and betamethasone, did not influence the clinical course or lesions of experimental 3-MI toxicity. Likewise, pretreatment with chloramphenicol or disodium cromoglycate failed to alter signs or lesions.469 However, in one small trial, flunixin meglumine at 1.1 mg/kg IV daily given after the onset of 3-MI–induced disease in calves was effective in lessening signs and lesions.470 Recovery often occurs without therapy in the less severe cases. In view of the dangers of handling affected cattle, the questionable efficacy of medical treatment, the probable irreversible nature of severe lesions, and the probability of spontaneous recovery in less severe cases, the best treatment may be no treatment. If treatment must be attempted, affected cattle should be handled very cautiously, and furosemide (0.5 to 1 mg/kg IM or IV once or twice daily)463 and flunixin meglumine (1.1 to 2.2 mg/kg IV daily or divided twice daily) or steroids (dexamethasone at 0.05 to 0.2 mg/kg IM or IV once daily) may be given. Most fatalities occur in the first 2 days. Severely affected animals that survive may develop chronic emphysema or heart failure secondary to cor pulmonale.467 Moderate to mild cases often show marked improvement after day 3, with recovery over about 10 days; relapses do not occur.
Prevention is based on management and prophylactic drugs. Management strategies that prevent the exposure of susceptible cattle to potentially toxic pastures include the following:
1
Place the cattle in a drylot, feed palatable hay for several days, and turn them onto the lush pasture for 2 hours the first day. Gradually decrease the amount of hay fed and increase the time on pasture over a period of 10 to 12 days.
4672
Delay use of lush pastures until after a hard frost.
4683
Cut and windrow the pasture before turning cattle out.
4694
Use the pasture for young stock (less than 15 months old) or sheep or other livestock
468; turn adult cattle out only after the pasture has been thoroughly grazed over.
5
Use the pasture before it becomes particularly lush.
4656
Use continuous strip-grazing.
467Because such management changes are frequently not feasible, prophylactic medication is a promising alternative. Monensin or lasalocid at 200 mg/head/day PO reduces the conversion of tryptophan to 3-MI.462 Treatment with monensin should be started at least 1 day before pasture change and should be continued an additional 10 days, whereas lasalocid requires a longer pretreatment period of 6 days.471 For example, 1 kg/head/day of a protein or energy supplement containing 0.15% Rumensin 60 supplies 200 mg of monensin.460 Monensin or lasalocid is not expected to be beneficial after the onset of signs. Future possibilities include blockers of the mixed function oxidase system and enhancers of intracellular glutathione levels.
Feedlot Acute Interstitial Pneumonia
Definition and Etiology
In feedlot cattle an ARDS occurs that is commonly referred to as feedlot AIP. The exact cause of this syndrome is unknown, and it is probably multifactorial. Feedlot management practices typically do not include exposure of cattle to lush forages similar to those that cause ABPEE, and the feeding of moldy sweet potatoes has not been associated with feedlot AIP.472 A variety of causes have been proposed473,474; unfortunately, the amount of research to confirm or refute the various hypotheses ranges from small to nonexistent. The most commonly suggested possible causes or predisposing factors are (1) feed-associated pneumotoxins such as 3-MI475-477 or dietary factors related to protein metabolism478; (2) chronic bacterial pneumonia479-481; (3) gender475,476 and hormonal influences482,483; (4) chronic small airway injury480,481,484; (5) BRSV infection481,485; (6) heat or dust exposure484,486; and (7) hypersensitivity reactions.484,487,488 These possible causes could also work together in various combinations to cause feedlot AIP.477,481
Support for 3-MI as a cause of feedlot AIP comes from research that showed that levels of a stable metabolite of 3-MI, 3-methyleneindolenine (3-MEIN), were significantly higher in the blood of cattle with AIP than in control cattle.475,476 Increased levels of 3-MEIN may be a result of decreased clearance of the toxin.475 Loneragan and colleagues also found that 3-MEIN levels were higher in lung tissue of AIP cases as compared with animals without lung disease, but they were not higher than 3-MEIN levels in the lung tissue of cattle with bronchopneumonia; this suggested that 3-MI may contribute to the pathogenesis of both AIP and bronchopneumonia.476 The mechanism by which 3-MI is generated and leads to lung damage was discussed earlier in the section describing ABPEE. If 3-MI does contribute to the pathogenesis of feedlot AIP, it is not clear what about the feedlot diet predisposes cattle to produce high levels of 3-MI. Normal cattle produce some 3-MI through metabolism of dietary proteins. It may be that the protein composition (e.g., the tryptophan concentration) of feedstuffs sometimes added to feedlot rations can lead to spikes in 3-MI production. Cattle with feedlot AIP have been found to have higher ruminal pH values than expected for cattle adapted to a high concentrated diet. Ruminal pH in AIP cases ranged from 5.6 to 7.2 in one study481 and from 4.9 to 7.4 in another,489 whereas the ruminal pH of cattle adapted to a high concentrate diet is typically about 5.5 to 5.6.490 Many proteins are relatively basic; therefore the high ruminal pH could be related to abnormal protein metabolism. However, the relatively high ruminal pH could also be caused by anorexia. The concept that abnormal ruminal protein metabolism could contribute to feedlot AIP was also supported by a small study that found increased ammonia levels in the ruminal gas cap of cattle with AIP.491 An interaction between digestive health and AIP was also suggested by the finding that in feedlot pens in which at least one animal had died from digestive disease, the incidence of AIP was approximately 70% greater than in pens in which digestive disease deaths did not occur.478
The hypothesis that bacterial bronchopneumonia contributes to feedlot AIP is supported by the finding by multiple authors that cattle with AIP frequently have superimposed gross and histopathologic lesions consistent with bacterial bronchopneumonia.479-481 In a study evaluating the bacteria isolated from the lungs of cattle with feedlot AIP that had not received antimicrobial therapy, P. multocida and Mycoplasma species were significantly more likely to be isolated from the lungs of cattle with AIP than from the lungs of normal penmates.481 An earlier study found that bacterial respiratory pathogens were not more likely to be isolated from cases of AIP as compared with controls, but the samples in this study were collected at necropsy of animals that received antimicrobial treatment before death in at least some cases.492 It should be noted that no study has yet been able to confirm whether in cases with both lesions the bacterial bronchopneumonia was present before AIP occurred or the bacterial bronchopneumonia developed after the animals had AIP. If bacterial bronchopneumonia leads to the development of AIP, it is not clear how this happens. In humans with ARDS, bacterial infection is often a predisposing factor that appears to lead to ARDS through the induction of high local or systemic levels of proinflammatory cytokines.493,494 The role of proinflammatory cytokines in cattle with feedlot AIP has not been investigated.
A role for gender and/or hormonal influences in the pathogenesis of feedlot AIP is related to the frequent (although not inevitable)480 finding that the majority of affected animals are heifers.475,478,481,495 Feeding of melengestrol acetate (MGA), which is fed to heifers to control estrus, has been suggested to contribute to AIP by some authors,473,482 but others have not found evidence of any association between MGA and AIP.478,495 Although one research study indicated that MGA could exacerbate experimentally induced AIP in sheep,482 a well-controlled experimental study to determine the effect of MGA on AIP in cattle in the field is needed to clarify the involvement of MGA with AIP. It has also been suggested that growth hormone implants may contribute to development of feedlot AIP, although a survey of feedlot managers did not find data to support this.495
Infection with BRSV was linked to feedlot AIP in an early study.485 Because BRSV infection can sometimes cause AIP, the possibility that a ruminant with lesions of AIP is simply a case of BRSV infection must always be considered. However, cattle with feedlot AIP do not always have a fever,481 which is expected in animals with acute BRSV infection. Moreover, feedlot AIP can occur in outbreaks, but cases often occur sporadically, which would be less consistent with BRSV infection. Neither Sorden and colleagues nor Loneragan and colleagues found BRSV significantly more often in animals with feedlot AIP than in controls.480,492 In another study the difference in frequency of identification of BRSV in feedlot AIP cases as compared with normal penmates approached significance (P = .07).481 It is interesting to note that in this study BRSV antigen was not found in airway epithelial cells, as is characteristic of acute BRSV infection,496 but rather was found in cells, possibly macrophages, that were surrounding airways. The significance of this was not clear; perhaps the BRSV antigen found in these cases was residual antigen in phagocytic cells that was engulfed during BRSV infection in the recent past.
Although AIP is by definition “acute,” it is interesting that several authors have reported histopathologic evidence of chronic airway injury (particularly, bronchiolitis fibrosa obliterans) in the lungs of animals with AIP.480,481,484 In one study, the presence of bronchiolitis fibrosa obliterans was identified significantly more often in cattle with AIP than in penmates with no history of treatment for respiratory disease; this was in spite of the fact that AIP cases in the study also had no history of previous treatment for respiratory disease.481 It is not known how past airway injury is related to the development of AIP, although chronic airway injury could be linked to the occurrence of chronic bacterial pneumonia, which has also been associated with feedlot AIP, as described previously. It may be that the chronic airway injury is related to dust exposure, although it is not clear why animals with AIP would be more likely to have airway injury caused by dust exposure than other penmates. Dust exposure has been anecdotally related to AIP,484,486 and feedlot managers often report that efforts to control dust, such as spraying water lightly onto the surface of pens, decreases AIP occurrence when the disease is a problem. However, repeated exposure to feedlot dust or to fungal organisms from feedlot dust did not induce AIP in sheep or goats in experimental studies.497,498 One study of the effect of dust on feedlot respiratory disease found only a weak association between airborne dust particles and respiratory disease.499
High ambient temperatures are thought to contribute to feedlot AIP because several investigators have found that the majority of cases occur in the summer,478,484 but no mechanism by which hot weather might exacerbate AIP has been researched. Hypersensitivity reactions are often suggested to cause AIP.502,503 It is true that anaphylaxis can cause pulmonary edema, hemorrhage, and emphysema, with microscopic evidence of hyaline membrane formation.500 However, animals that survive longer than a few hours after an episode of anaphylaxis do not have lung changes consistent with AIP, such as type 2 pneumocytic proliferation and interstitial inflammatory cell infiltrate.501 Therefore an anaphylactic reaction may cause an occasional case of sudden death with lung changes typical of AIP, but anaphylaxis does not explain the majority of feedlot AIP cases. Other types of hypersensitivity-mediated lung disease have pathology that is unlike that seen in feedlot AIP, also making other types of hypersensitivity an unlikely cause of the majority of feedlot AIP cases.460,473
In summary, the cause of feedlot AIP is unknown, but the strongest support currently exists for some role for (1) factors related to feed, or ruminal metabolism, including 3-MI; (2) infectious respiratory disease, especially chronic bacterial pneumonia, and possibly BRSV; (3) gender and/or other hormonal influences; and (4) chronic airway injury, which may be related to infectious respiratory disease. It seems likely that multiple factors can contribute to the development of feedlot AIP, and the factors may act in some as-yet unidentified combination in at least some cases. It is also possible that some causes predominate in some feedlots or individual cases, whereas other causes predominate in other feedlots or individual cases. A study evaluating the occurrence of bacterial respiratory pathogens in cattle with feedlot AIP found bacterial respiratory pathogens in the lungs of the majority of cases in one feedlot, and in almost none of the cases in a second feedlot,481 suggesting that bacterial infection played a role in the development of AIP cases in the first feedlot but not the second.
Clinical Signs
Feedlot cattle with AIP may be found dead in the pen.504 Clinical presentation includes rapid onset of expiratory dyspnea and tachypnea, although if the respiratory effort is great, the actual respiratory rate may not be greatly elevated. Cattle typically stand with their heads extended and front legs spread apart, and exhibit open-mouth breathing (see Fig. 31-66). Frothing from the mouth may also be observed. Rectal temperatures are variable, ranging from normal to elevated.481 Physical examination may reveal cyanosis, tachycardia, and SC emphysema that extends from the cervical to dorsal thoracic area.505 Auscultation of lungs reveals dull areas throughout the lungs, along with some crackles. Differential diagnoses of bronchopneumonia, tracheal edema, tracheal obstruction, and hypersensitivity pneumonitis should be considered.
Pathogenesis
Because the cause of feedlot AIP is unknown, the pathogenesis is also uncertain. If feed-related pneumotoxins cause at least a subset of cases with AIP, the pathogenesis will be similar to that described for ABPEE, 4-ipomeanol toxicity, and perilla ketone toxicity. If bacterial infection is a cause of some cases of feedlot AIP, as is true for some human cases of ARDS, then proinflammatory cytokine production and the resultant inflammatory cascades they initiate are likely involved. More research is necessary to determine the cause and the pathogenesis of feedlot AIP.
Epidemiology
In the 1999 NAHMS survey of feedlots, AIP was identified as the second leading cause of feedlot mortality, behind bronchopneumonia (shipping fever).502 Mortality rates resulting from AIP of 0.03% to 0.15% have been reported.478,479,484,495 An important feature of AIP is the tendency of the disease to occur most often in cattle on feed more than 60 days,475,481,484,492,495 as opposed to shipping fever, in which mortality peaks by 45 days after feedlot entry.506 This means that losses due to AIP deaths are amplified by the fact that relatively more resources in feed and labor have been invested in cattle that die of AIP, as compared with cattle that die of shipping fever. Most cases of feedlot AIP occur in the summer,478,484,495 but the disease can occur in any season of the year; and most studies report that heifers are disproportionately affected.475,476,495 One survey found the odds of an animal with AIP being a heifer were 3.1 times greater than the odds of the animal being a steer.476 In feedlot pens in which an animal died from a digestive disorder, the relative risk of AIP occurring was about 1.7, indicating that pens with a digestive disorder death were about 70% more likely to also have an AIP death, as compared with pens with no digestive disorder deaths.478
A survey of feedlot managers was undertaken to determine risk factors for feedlot AIP.495,503 Although the response rate was relatively poor (12%), the responding managers oversaw just under 2.5 million cattle, which represented about 10% of the U.S. feedlot inventory the year the survey was undertaken.503 Feedlots in northern states were significantly less likely to recognize AIP as a cause of morbidity or mortality as compared with feedlots elsewhere, whereas larger feedlots and feedlots placing a higher proportion of yearlings were more likely to recognize AIP as a cause of morbidity or mortality. Feedlots that vaccinated less than 95% of placements for M. haemolytica ± P. multocida were more likely to recognize AIP as a cause of disease, and these feedlots recognized AIP as a cause of a larger proportion of deaths, as compared with feedlots that vaccinated more than 95% of cattle for these pathogens.495 If the data reported by the feedlot managers are representative of a true association between Mannheimia and Pasteurella vaccination and AIP, the reason for the link is not clear. It may simply be related to the association between placement of a high proportion of yearling cattle and AIP, as yearlings are less likely to be vaccinated for Mannheimia and Pasteurella than younger cattle. It may also be related to the finding that AIP cases have increased likelihood of having concurrent bacterial pneumonia in some feedlots, but more research is necessary before any definite conclusions can be made.
Necropsy Findings
The gross pathology of feedlot AIP is essentially the same as that described for ABPEE. If there is concurrent bacterial bronchopneumonia, there may be cranioventral consolidation, with fibrin deposition on the pleura, but in cases with no concurrent bacterial pneumonia, the pleura is free of fibrin. Grossly it is common to see a “patchwork” appearance of dark and light colored lobules interspersed, and the lobules are freely movable473,481 (see Fig. 31-67). Histologically, the most acute cases will have only hyaline membrane formation and alveolar edema, possibly with hemorrhage in the alveoli or interstitium; cases that have been going on longer will have proliferation of type 2 alveolar epithelial cells and inflammatory cell infiltrate into the interstitial space (septa). It is not unusual to find evidence of chronic airway injury, such as peribronchiolar lymphoid cuffing, peribronchiolar vascular fibrosis, and bronchiolitis fibrosa obliterans.480,481 Whether the chronic airway injury is related to the pathogenesis of AIP or whether it is an incidental finding is unknown, but in one study bronchiolitis fibrosa obliterans was significantly more common in AIP cases than in penmates with no history of lung disease.481
Diagnosis
Diagnosis of feedlot AIP can be confirmed only by histopathologic evaluation of lung from animals that die or are euthanized because of the disease. Clinical signs and even gross pathology are not definitive; only 65% to 80% of cases identified based on clinical signs were confirmed by histopathologic evaluation to have AIP in two studies.475,481
Treatment and Prevention
Treatments recommended are similar to those recommended for ABPEE. However, feeding monensin does not preclude development of feedlot AIP, as cattle that die of AIP are often consuming feed including monensin when they contract AIP.481 Treatment is supportive and typically includes administration of antiinflammatory drugs such as steroids (dexamethasone at 0.05 to 0.2 mg/kg once or twice) or flunixin meglumine (1.1 to 2.2 mg/kg IV q24h), and diuretics such as furosemide (1 mg/kg IM or IV q12-24h). Antimicrobial drugs are appropriate given the fact that cases often have superimposed bacterial pneumonia (see Table 31-10). There are no studies evaluating the response of cattle with AIP to any treatment, and such studies would be difficult because there is no perfect method of making an antemortem diagnosis of the disease. However, anecdotal reports indicate that the prognosis is guarded even with treatment. It is important to note that simply moving cattle out of the pen could lead to death from severe respiratory compromise. Because of the risks and uncertainties of treatment and the expected high case fatality rate, immediate salvage slaughter may be the most economic course to take507; if salvage slaughter is attempted, remember to observe proper drug withdrawal times.
Because the cause of feedlot AIP is unknown, it is difficult to recommend control measures. Administration of aspirin and of vitamin E have been suggested as rational preventative strategies to counteract inflammatory pathways suspected to be involved in feedlot AIP; however, two trials showed no clear effect of these therapies on levels of 3-MI in treated cattle.508,509 No cattle in these trials developed AIP, so an effect on disease could not be identified. The risk factors identified suggest that management strategies to minimize abrupt dietary changes and to control infectious respiratory disease may be helpful; anecdotal reports also suggest that efforts to control feedlot dust may be helpful.
4-Ipomeanol (Moldy Sweet Potato) Toxicity
Definition and Etiology
Moldy sweet potato toxicity is caused by the ingestion of a furanoterpenoid toxin produced by sweet potatoes (Ipomoea batatas) in response to infestation with the fungus Fusarium solani (javanicum). It should be emphasized that this disease is an intoxication and not an allergic response to the fungus.460
Clinical Signs
There is an acute onset of tachypnea, tachycardia, hyperpnea, and dyspnea, with loud expiratory grunting, frothing at the mouth, extension of the head and neck, flaring of the nostrils, and frequent deep coughing. Crackles and harsh bronchial sounds are heard on auscultation.510 Signs usually occur within 1 day of exposure, and deaths may occur 2 to 5 days later.460 Differential diagnoses are as for ABPEE (see earlier discussion), which this condition closely resembles, except for the history of exposure and the more prominent cough and adventitious lung sounds.
Pathogenesis
When F. solani (or closely related species) grows on sweet potatoes, the potato produces several 3-substituted furans, including 4-hydroxymyoporone, which is hepatotoxic. This is converted by the fungus to a series of pneumotoxins, the most abundant of which is 4-ipomeanol. When ingested by cattle in sufficient amounts, this toxin is absorbed, carried to the lungs in the blood, and converted to a highly reactive metabolite by a cytochrome P-450–dependent mixed function oxidase system.460 From this point the pathogenesis is similar to that of ABPEE—that is, the toxin binds to intracellular macromolecules in the cell, causing cellular damage, particularly in Clara cells, type I pneumocytes, and endothelium; edema, hemorrhage, cellular necrosis, hyaline membrane formation, and proliferation of cuboidal epithelium result, with secondary emphysema.
Epidemiology
The disease usually occurs in outbreak form when groups of cattle are fed damaged sweet potatoes. Morbidity and case fatality rates are high. Calves nursing affected cows are unaffected.511
Necropsy Findings
The lungs are wet, firm, and large and fail to collapse. Hemorrhages, yellow gelatinous edema fluid, and emphysema with bullae occur throughout. Lobules are dark red and firm.510,511 Microscopic lesions include edema, emphysema, hyaline membranes, hemorrhage, mixed interstitial infiltrates, alveolar epithelial hyperplasia, peribronchiolar fibrosis, and bronchiolitis obliterans.511
Diagnosis
Diagnosis is made based on history of feeding sweet potatoes and identification of clinical signs and pathology consistent with AIP. Other diagnostic tests as described for ABPEE could also be attempted in valuable individual animals.
Treatment and Prevention
Treatment has not been investigated. Because the pathophysiologic mechanisms are similar to those of ABPEE, similar recommendations are offered here: handle affected animals with extreme care; if treatment is attempted, furosemide (0.5 to 1 mg/kg IM or IV q12-24h) and flunixin meglumine (1.1 to 2.2 mg/kg IV daily or divided twice daily) or dexamethasone (0.05 to 0.2 mg/kg IV or IM q24h) may be given. The prognosis for moderate to severe cases is grave, regardless of management. Because toxicity is difficult to predict and is usually severe and irreversible when it occurs, the feeding of mold-damaged sweet potatoes should be strictly prevented.
Perilla (Perilla Frutescens) Ketone Toxicity
Definition and Etiology
Perilla ketone toxicity is an ARDS caused by ingestion of a pneumotoxin found in the leaves and seeds of Perilla frutescens, a common weed in the southeastern United States. This plant is also known as purple mint, perilla mint, wild coleus, and beefsteak plant.512 It is an erect herbaceous annual about 2 m high, with characteristic square stems, an aromatic odor, and opposite, coarsely serrated ovate leaves 5 to 10 cm long and 4 to 8 cm wide, with a purplish tint at maturity. The seed and flower stage, which occurs in August to October, appears to be most toxic.512 The flowers are small, white to purple blooms on a long raceme.512 The plant prefers semishade, such as damp, open wooded areas.
Clinical Signs
Animals are often found dead.512 Signs observed include a sudden onset of moderate to severe dyspnea, wheezing, frothing at the mouth, and an expiratory heave or grunt.512,513 In less severe cases the cow may pant.513 Exertion worsens the signs and may precipitate death. Mature cows are most often affected, but deaths have been reported in yearlings and calves.512 Death occurs in 3 to 7 days in experimental toxicity.512 Differential diagnoses are as for ABPEE (see earlier discussion), which this condition closely resembles and from which it can be differentiated only by history of exposure.
Pathogenesis
The volatile oils of P. frutescens contain a number of 3-substituted furans that are chemically similar to 4-ipomeanol, the moldy sweet potato toxin. One of these, perilla ketone, predominates in the later growing season (when most toxicities occur) and has been shown to be pneumotoxic when given parenterally to mice, hamsters, goats, calves, and sheep.512 The toxin is absorbed from the rumen, carried to the lungs through the blood, and probably metabolized to the toxic form by the mixed function oxidase system, as for 4-ipomeanol and 3-MI (see earlier). The pathogenesis from this point parallels that of ABPEE or moldy sweet potato toxicity.
Epidemiology
P. frutescens seems to thrive in late summer, when pastures in the southeastern United States are frequently dry and dormant.513 This also corresponds with the more toxic stage of the plant.512 Cattle normally avoid the plant when other pasture is available but may be forced to consume it during this critical period.513 However, under experimental conditions calves were noted to prefer the mint.512 The preseed stage appears to be of relatively low toxicity; the green seed-stage plant is most toxic, especially the seed parts; dried hay from seed-stage plants is less toxic than green plants but is still potentially lethal; and frosted plants appear to have relatively low toxicity.512 The exact toxic dose is unknown, but 2.3 kg of green seed-stage plant and 11.2 kg of hay were lethal for cattle in one trial.512
Necropsy Findings
The lungs are distended (often bearing the impressions of the ribs), fail to collapse, and are moist, heavy, edematous, and emphysematous. There are often bullae, pleural effusions, and froth in the airways. Histologic characteristics are edema, extensive alveolar epithelial hyperplasia, emphysema, and congestion.
Diagnosis
Diagnosis is based on history of exposure to perilla mint and characteristic clinical signs and lung pathology. Other diagnostic tests as described for ABPEE could also be attempted in valuable individual animals.
Treatment and Control
Treatment has not been investigated. On the basis of the similar pathophysiologic mechanisms, the recommendations for ABPEE should be followed (see earlier discussion). The prognosis for severe cases is grave, regardless of management. Cattle should be provided sufficient forage that they do not seek out perilla mint; once they have begun to eat it, they should be fenced away from stands of the plant, and other forage should be provided.
Other Toxic Plants
Zieria arborescens (stinkwood) leaves cause a fatal ARDS in Tasmanian and Eastern Australian cattle after ingestion of 15 to 30 kg over 2 to 4 weeks. An oil isolated from the leaves has produced the same lung lesion in rabbits. The signs are as for the other ARDS: acute tachypnea, grunting, extension of the head, mouth breathing, abdominal respiration, tachycardia, and fever secondary to the respiratory effort. Death occurs in 1 to 21 days; some animals survive. Lesions include massive pulmonary edema and emphysema.514 Treatment has not been investigated, and recommendations as for ABPEE (see earlier discussion) are suggested. As mentioned in relation to ABPEE, Brassica species (rape, kale, turnip tops) are currently regarded as one of the types of pasture that can precipitate the 3-MI–associated disease. The possibility that other specific toxins may be identified in these species has not been excluded. Morbidity and mortality rates appear to be much higher on Brassica species pastures than on other lush forages.460 The hepatotoxic effects of pyrrolizine alkaloids are well known, but they also cause lung lesions. Lung lesions develop only in animals with chronic liver lesions, and the minimum dose necessary to produce lung lesions is never less than that which is hepatotoxic; therefore signs of liver disease usually predominate. Crotalaria and Trichoderma species are the most common offenders; to a lesser extent, Senecio species are a cause. Horses, sheep, cattle, and pigs have been affected. Pulmonary lesions include edema, congestion, hemorrhage, proliferation of bronchiolar and alveolar epithelial cells with megalocytosis, and interstitial fibrosis and cellular infiltration. As with 3-MI, 4-ipomeanol, and perilla ketone, the toxicity of the pyrrolizine alkaloids depends on activation by the mixed function oxidase system; in this case, however, the toxin is probably formed in the liver and spills over into the blood to reach the lungs. Vascular endothelium is probably the primary target for injury (vs. the Clara cells and type 1 pneumocytes in the other ARDS).515
Toxic Gases
Food animals may be exposed to a variety of toxic gases in the environment. The most important are ammonia, hydrogen sulfide, carbon dioxide, and methane from excreta and respiration; these can be especially important when excreta is collected in pits or tanks. Other gases include nitrogen dioxide from silos; carbon monoxide from machinery exhausts and heaters; zinc oxide from welding of galvanized metal in barns; chlorine, formaldehyde, insecticides, and other fumes from agricultural chemicals and cleaners; and smoke from fires. In most cases concentrations usually remain below overtly toxic levels, and effects are very subtle. Such chronic low-level exposure may result in decreased disease resistance and depression of growth rates.516 Slightly higher levels of chronic exposure may cause clinically vague syndromes of lethargy, mild dyspnea, anorexia, depressed growth, excessive lacrimation and salivation, low incidence of sudden deaths over weeks or months, and stillbirths.516 Acute, severe outbreaks usually occur in tightly enclosed facilities and are related to accidents, power outages, agitation or pumping of manure pits, or other combinations of unusual circumstances. Such outbreaks are characterized by an ARDS of variable morbidity and frequently a high case fatality rate.
NITROGEN DIOXIDE
Nitrogen dioxide (NO2) is a yellow-orange to brown gas with an acrid odor that is produced by anaerobic fermentation of green plant material. It is a major component of “silo gas.” Acute exposure of farm workers to high concentrations of NO2 causes a respiratory condition known as “silo-filler’s disease,” characterized by severe acute edema and congestion and followed by bronchiolitis obliterans and progressive interstitial pulmonary fibrosis. A similar condition has been induced experimentally in cattle,517 and apparent (although unproved) spontaneous field cases have been reported.518,519 Clinical signs in experimental and apparent field cases include cough, tachycardia, tachypnea, respiratory grunting, depression, anorexia, hypogalactia, extension of the head, open-mouth breathing, fever, salivation, lacrimation, and SC emphysema. Auscultation reveals decreased breath sounds and crackles.518,519 The primary differential diagnoses should include other ARDSs of housed cattle that occur in outbreak form, especially exposure to other toxic gases (manure pit gases, zinc oxide, chlorine, carbon monoxide), and hypersensitivity pneumonitis from moldy hay. Nitrate toxicity should also be considered. Clinical pathologic evaluation is of limited benefit. Leukocyte counts remained normal in experimental cases; methemoglobin levels increased to a peak at 30 minutes after exposure and returned to normal in 12 to 24 hours.517 The pathophysiologic mechanism probably involves the dissolution of the NO2 in the water of the respiratory tract to form nitric acid. Nitrates and nitrites are also formed; these are irritating, and the nitrites cause methemoglobinemia.519 Nitrogen dioxide is also an oxidant itself and may contribute directly to the injury. The disease occurs as an outbreak, usually in housed cattle in proximity to a silo chute in a tight or poorly ventilated barn.518,519 Nitrogen dioxide is heavier than air and layers on the top of silage or spills out around the bottom of the silo. Corn silage produces more gas than hay, and a high nitrate content increases the danger. The levels are highest in the first 48 hours after filling the silo but may remain dangerous for 2 to 3 weeks.
Necropsy findings in experimental disease include hyperemia of the upper airways; hemorrhages, fibrinous membranes, and froth in the trachea; distended, noncollapsing lungs with rib imprints; a mottled appearance caused by consolidated lobules alternating with emphysematous lobules; and bullae. Microscopic lesions include alveolar epithelial hyperplasia, large foamy alveolar macrophages, hyaline membranes, hyperemia, hemorrhage, and edema.
Treatment involves the establishment of adequate ventilation; cows should be completely removed from closed buildings if necessary. Corticosteroids have apparently been beneficial in field cases,518,519 but no controlled studies have been performed. Because of the obvious differences in pathophysiologic characteristics, it would be unwise to extrapolate treatment regimens from those of ABPEE. Suggested empiric therapy might include corticosteroids (dexamethasone at 0.05 to 0.2 mg/kg IM or IV daily), furosemide (0.5 to 1.1 mg/kg IM or IV daily or twice daily), and appropriate antibiotics to prevent secondary bacterial infections.
ZINC OXIDE
Zinc oxide (ZnO) fumes have been associated with an ARDS in cattle.520 Oxyacetylene cutting or arc welding of galvanized pipe results in production of white fumes of zinc oxide containing colloidal particles 0.3 to 0.4 mm in diameter, which can reach the terminal alveoli when inhaled.520 Construction activities in closed barns containing animals may result in toxicity in animals in close proximity or in the path of ventilation. All ages may be affected.
Clinical signs in severe cases include acute onset of anorexia, frothing at the mouth, anxiety, extension of the head and neck, mouth breathing, expiratory grunting, tachypnea, tachycardia, mild fever, SC emphysema, and crackles on auscultation of the lungs. Death may occur within 12 hours. Less severely affected animals exhibit depression, mild fever, and tachypnea.520 Differential diagnoses include other ARDS of housed cattle that occur in outbreak form such as exposure to other toxic gases (nitrogen dioxide, manure pit gases) or hypersensitivity pneumonitis. The pathophysiologic process presumably involves direct damage to cells by the ZnO and its products dissolved in the fluid lining the respiratory tract. Necropsy findings include purulent conjunctivitis; SC emphysema; congestion of the airways; tracheal hemorrhages; stiff, noncollapsing lungs; and pulmonary congestion, edema, and emphysema with bullae.520 Histologic lesions include pulmonary congestion, emphysema, edema, and mixed cellular infiltrates with a prominent eosinophil component. Treatment of severe cases with epinephrine, antihistamine, atropine, and corticosteroids had no effect in one outbreak,520 whereas mild cases recover spontaneously. Suggested empiric therapy could include ventilation of the area, dexamethasone (0.05 to 0.2 mg/kg IM or IV daily), furosemide (0.5 to 1.1 mg/kg IM or IV daily or twice), and appropriate antibiotics to control secondary bacterial invaders.
CHLORINE
Chlorine is a greenish yellow gas widely used in manufacturing. Animal exposure is usually the result of industrial accidents. Exposed animals may be found dead. Signs include depression, profuse nasal discharge, lacrimation, and dyspnea; crackles may be heard on auscultation.521 The toxic effects are the result of the formation of hydrochloric and hypochlorous acids; the latter breaks down to hydrochloric acid and oxygen, both of which are toxic to tissues. Necropsy findings include congestion of the nasal mucosa, tracheitis, and pulmonary edema, hemorrhage, and emphysema. Histologic lung lesions include edema, emphysema, hemorrhage, atelectasis, hyaline membrane formation, and lymphocytic bronchitis.521 Treatment is empiric; suggestions include corticosteroids (dexamethasone at 0.05 to 0.2 mg/kg IM or IV daily), furosemide (0.5 to 1.1 mg/kg IM or IV daily to twice daily), and appropriate antibiotics to prevent secondary bacterial infection.
MANURE GASES
Manure gases include mixtures of hydrogen sulfide, ammonia, carbon dioxide, methane, and carbon monoxide.460 Accumulation of these gases can result in asphyxiation of animals in enclosed barns over manure pits.
SMOKE INHALATION
Smoke inhalation injury may occur in animals that survive barn fires. Many clinical changes may not become evident for 24 to 48 hours after the fire. It is important to assess the degree of damage as early as possible and to attempt to anticipate sequelae so that early aggressive therapy can be instituted.522 Common problems for which to check include oral burns, conjunctivitis, and laryngospasm. Hoarseness, expiratory wheezes, and carbonaceous sputum indicate potentially serious sequelae. Crackles and wheezes on auscultation may occur very early or may be delayed for hours. Cough, stridor, and tachypnea may also occur. Bright red mucous membranes may indicate CO poisoning or burns and may mask cyanosis.522 Carboxyhemoglobin determinations on iced venous blood (often available at human hospitals), serial arterial blood gas determinations, transtracheal wash, and bronchoscopy are useful in delineating the extent of damage and prognosis.522
The pathophysiology of smoke inhalation is complex and involves two main mechanisms: CO toxicity and smoke toxicity. CO toxicity results in tissue hypoxia in all organs, especially the brain; O2 consumption in the fire and the pulmonary effects of smoke may aggravate this hypoxia. Heat damage in animals is usually limited to the upper airways. Smoke toxicity is related to the inhalation of soot, superheated particles, and a variety of noxious gases (e.g., aldehydes, oxides of sulfur and nitrogen, benzene from plastics), which results in the formation of dissolved acids, alkaloids, and other direct irritants in pulmonary fluids. These mechanisms eventually (usually 2 to 24 hours after inhalation) result in alveolar damage, interstitial edema, hypoxia, and secondary bronchopneumonia.522
Treatment involves establishing a patent airway with intubation or tracheostomy if necessary. Oxygen, up to 100% for short periods, is indicated522; care must be exercised because 100% oxygen can also cause pulmonary damage. IV fluids should be given, with careful monitoring for pulmonary edema. The use of corticosteroids is controversial.522 Antibiotics are indicated to prevent secondary bronchopneumonia. Bronchodilators such as aminophylline at 6 to 10 mg/kg IV or PO three times daily may help relieve soot-induced bronchospasm.
HYPERSENSITIVITY PNEUMONITIS
Definition and Etiology
Hypersensitivity pneumonitis, also known as extrinsic allergic alveolitis (EAA) or “bovine farmer’s lung,” is an allergic respiratory disease caused by inhalation of organic dusts. Several such conditions are recognized in humans, differing only in the nature of the antigen and the circumstances under which exposure occurs. The classic example, of which the bovine disease is probably the counterpart, is “farmer’s lung.” EAA is caused by exposure to the dust from moldy hay, grain, or other plant matter containing spores and products of thermophilic actinomycetes such as S. rectivirgula (formerly M. faeni) and T. vulgaris.523,524 Other unidentified forms of hypersensitivity pneumonitis probably occur in cattle.
Clinical Signs
EAA is a disease of confined adult cattle; consequently it is more common in dairy than in beef breeds. Typically a succession of acute cases occurs during the winter housing period, so the clinician is presented with a group problem in which animals are in varying stages of the disease. The acute form is indicative of recent exposure and is characterized by a sudden onset of dullness, decreased appetite, hypogalactia, coughing, expiratory tachypnea, dyspnea, and cranial-ventral crackles on auscultation of the lungs.523-525 There is a moderate transient fever, which is frequently missed.524 The chronic form is insidious at onset and may not be detected until there is considerable fibrosis. Animals with the chronic disease may have acute exacerbations as a result of heavy antigen exposure; some may not be detected until turned out in the spring, when increased exercise causes an acute crisis. There is a history of weight loss and coughing for several winters, with remission in the grazing season. Chronic signs include hypogalactia, weight loss, productive coughing, tachypnea, obvious hyperpnea, and widespread crackles and wheezes on auscultation of the lungs, especially in the rostral-ventral areas.524 Differential diagnoses should include respiratory diseases affecting groups of housed adult cattle in winter. Infectious diseases (viral and bacterial pneumonias) can usually be differentiated from farmer’s lung by careful evaluation of factors such as clinical signs of fever and pulmonary consolidation. Therefore the main differential diagnoses are the toxic gases. It should be emphasized that hypersensitivity pneumonitis is not considered an ARDS and that no evidence exists for the involvement of hypersensitivity in the pathogenesis of the ARDS described previously. However, severe cases of EAA with prominent dyspnea and moderate, nonfatal cases of gas intoxication may appear clinically similar. If the group outbreak aspect of EAA is ignored, the clinician who examines one individual may be unable to distinguish this condition from fibrosing alveolitis.
Pathogenesis
The spores of the thermophilic actinomycetes are 0.7 to 1.3 mm in diameter and can easily reach the alveoli, where they induce both humoral (precipitating) and cellular immune responses. It is thought that repeated exposure results in the activation of a number of immunologically specific and nonspecific cellular and humoral effector mechanisms at the alveolar level, which results in tissue damage.523,524
Epidemiology
EAA is a problem in areas with wet summers and severe winters, a situation characterized by the combination of moldy hay and housing of cattle in winter. In North America, EAA occurs primarily in the Great Lakes region of the United States and the eastern provinces of Canada.525 Baling and stacking of hay with a high moisture content (over 30%) results in overheating of the stacks. Thermophilic molds become dominant and produce billions of spores, which are released when the hay is distributed for feeding. A similar situation can occur in stored grains. Affected hay is usually dry, friable, discolored, and dusty; however, it is not necessary for hay to be grossly dusty and poor in quality to release large numbers of spores.524
Necropsy Findings
In acute cases the lungs are superficially grossly normal. Closer inspection reveals small gray spots in many lobules, which represent interstitial and peribronchiolar accumulations of lymphocytes. Other lobules exhibit dark red centers of atelectasis surrounded by pale pink raised edges of trapped air that are caused by narrowing of airways with lymphocytic infiltrates. Histologic evaluation confirms the presence of these lymphocytic infiltrates and aggregations, as well as epithelial granulomas, bronchiolitis, and bronchiolitis obliterans. The gross appearance in chronic cases is similar, with the addition of focal areas of interalveolar fibrosis and epithelial hyperplasia; epithelioid granulomas are absent unless a recent acute exposure has occurred.523,524
Diagnosis
Precipitating antibodies to M. faeni are found in the serum in most cases.523,524 However, two precautions are necessary. First, the presence of precipitating antibodies only indicates exposure to the antigen; many normal animals also have antibodies. Second, it is possible that other as yet unidentified antigens may cause allergic respiratory disease in cattle. The presence or absence of titers is therefore not necessarily diagnostic of the clinical status of the animal, and the magnitude of the titer does not reflect the extent of disease.460
Treatment and Prevention
Treatment and control center around removal of the offending antigen, which is frequently difficult from an economic and management standpoint. Corticosteroids (dexamethasone at 0.05 to 0.2 mg/kg IV or IM daily) may be beneficial in acute cases. Suggestions to decrease the molding of hay or degree of exposure include making silage instead of hay, ensuring proper drying before baling, and feeding hay outside. If the condition can be arrested before significant fibrosis occurs, the prognosis is good.
Anaphylaxis
In ruminants the lung is the major target organ in immediate (type I) hypersensitivity reactions. Precipitating antigens include vaccines, drugs, blood, Hypoderma bovis or Hypoderma lineatum larvae, insect bites, and bee stings. Signs of anaphylaxis usually develop in 10 to 20 minutes and include severe acute dyspnea, with flaring of the nostrils, extension of the head and neck, open-mouth breathing, frothing of the mouth, hyperpnea, and abduction of the elbows. Pharyngeal and laryngeal edema cause stertor and inspiratory dyspnea. Urticaria may occur in some cases, such as milk allergy in Jersey cows. Shivering, salivation, lacrimation, pruritus, diarrhea, fever, edema (eyes, muzzle, anus, and vulva), collapse, nystagmus, cyanosis, and discharge of froth from the nostrils also may occur. On auscultation there are harsh breath sounds, large airway sounds, and crackles. The primary differential diagnosis is peracute interstitial pneumonia. The pathogenesis involves an initial exposure to the antigen, which results in a genetically determined production of homocytotropic antibodies. In humans and dogs, this is immunoglobulin E (IgE); in ruminants various classes may be involved.526 This antibody attaches to receptors on mast cells and basophils. On subsequent exposure to the antigen, bridging of the Fab parts of the antibody on these cells results in degranulation, with release of a variety of mediators such as histamine, bradykinin, 5-hydroxytryptamine, serotonin, slow-reacting substance-anaphylaxis (SRS-A), eosinophil chemotactic factor A, platelet activating factor, kinins, and prostaglandins.526-528 These mediators result in a cascade of vascular events, notably increased vascular permeability, which in turn results in acute severe edema. Pulmonary venous constriction, pulmonary artery hypertension, splanchnic pooling, increased mucus secretion, and bronchospasm also occur. Ruminants that die of anaphylaxis have severe pulmonary congestion and edema, laryngeal edema, and froth in the airways. Lung lesions of ARDS may be present.
The treatment of choice is epinephrine, 4 to 8 mg (4 to 8 mL of 1:1000 solution) IV or SC or 1 to 5 mg IV for an average 500-kg cow and 1 to 3 mg IV or SC for an average adult sheep or goat. Epinephrine has a short half-life, and animals should be observed closely for relapse. Ancillary therapy includes corticosteroids (dexamethasone, 0.1 mg/kg IM or IV; prednisolone, 2.2 mg/kg IM or IV daily). Other supportive therapies include shock doses of IV fluids (40 mL/kg/hr); aminophylline; diuretics such as furosemide; oxygen therapy; and tracheostomy if necessary.