DISEASES OF THE PARANASAL SINUSES
SINUSITIS
Definition and Etiology
Sinusitis refers to inflammation of the paranasal sinuses, from primary microbial infection or secondary bacterial infection associated with dental disease or other sinus disease. Lined by respiratory mucosa, the paranasal sinuses are at risk for developing diseases affecting the respiratory tract.1173 Sinus empyema, accumulation of pus within a sinus cavity, may result from bacterial or viral infection.
Incidence of sinusitis is relatively low, likely less than 0.5% of disease in equine practice.1174 In 256 horses with sinonasal disease, primary sinusitis (24%) and dental disease—associated sinusitis (22%) occurred with similar frequency and were the most common cause of sinusitis, whereas sinus cysts (13%), sinonasal neoplasia (8%), ethmoid hematoma (8%), sinonasal trauma (6%), mycosis (5%), and rostral maxillary tooth infection (4%) were the other more common causes.1175 The frontal and maxillary sinuses are more commonly involved, with the corresponding conchal sinuses and the sphenopalatine sinus affected less often. Median age of horses affected with sinonasal disease is 7 to 11 years.1175
Acute or chronic upper respiratory tract infections of viral or bacterial origin can result in primary sinusitis; Streptococcus species infection is the most common. Maxillary sinusitis caused by disease of the third (caudal roots) through sixth cheek teeth (modified Triadian numbers 109 to 111, 209 to 211) often results from alveolar periostitis, patent infundibula, or fractured or split teeth.1173 Dental defects permit access of food material or bacteria to the tooth root and sinus cavity, with extension to the frontal sinus likely through the frontomaxillary opening. Reported neoplasms include osteoma, osteosarcoma, adenocarcinoma, lymphosarcoma, squamous cell carcinoma, and fibroma.1176-1179
Clinical Signs and Differential Diagnosis
Signs vary depending on cause, location, and extent of sinus involvement, with unilateral nasal discharge (serous, mucoid, mucopurulent, purulent) being the most frequent sign.1175,1180 Physical examination of a horse with sinusitis should include observation for epiphora, facial asymmetry, altered nasal airflow, abnormal breath odor, mandibular lymphadenopathy, and sinocutaneous fistula.
Nasal discharge is typically unilateral because the nasomaxillary opening is located rostral to the caudal edge of the nasal septum. Intermittent or continuous, nasal discharge need not be related to a previous upper respiratory infection. Malodorous discharge is commonly associated with dental sinusitis and sinonasal mycosis, whereas mucopurulent discharge is more commonly associated with sinus cysts.1175 Sanguineous discharge is common with ethmoid hematoma and sinonasal trauma; however, guttural pouch mycosis, pulmonary hemorrhage, nasal turbinate necrosis, and neoplastic or granulomatous lesions should also be considered. Other differential diagnoses of nasal discharge should include guttural pouch empyema or mycosis; acute pharyngitis (strangles, rhinopneumonitis, and influenza); neoplasia or necrosis of the turbinates; ethmoid hematoma; and pulmonary disease.
After nasal discharge, submandibular lymphadenopathy is the most common sign, particularly when microbial infection is a component of sinusitis. Facial swelling occurs with frequency similar to that of lymphadenopathy, typically when sinus drainage is obstructed or there is an expansile mass within the sinus, or as an acute sign after facial trauma. Occasionally exophthalmos can occur with marked sinus distortion.
Patency of the nasomaxillary opening generally precludes facial distortion. Loss of patency occurs when inspissated exudate accumulates or mucosal lining tissue reaction obstructs the opening. Expansion of the sinus may result in reduced nasal airflow, particularly ipsilaterally, caused by distortion of the architecture of the nasal passages, and in such instances abnormal respiratory noise rather than nasal discharge may be the primary presenting sign.1178
Epiphora occurs if there is nasolacrimal duct involvement from trauma, or compression or destruction of the duct from underlying sinus disease. Approximately one third to one half of horses with dental sinusitis, sinus cyst, and sinonasal trauma have epiphora as a presenting sign.1175
Percussion of the affected sinus may reveal dullness or pain, although normal resonance does not preclude the possibility of sinusitis. If there is bone thinning over gas above a fluid line (as can occur with some maxillary sinus cysts), percussion may elicit increased resonance.
Careful examination of the oral cavity for signs of dental or periodontal disease should be performed when any of these signs are present. Particular attention should be paid to examination of the occlusal surface of the teeth with a very fine dental pick (e.g., 22-gauge needle); however, it should be recognized that periapical abscess formation can occur without defects in the occlusal surface. Accuracy of diagnosis of dental disease is substantially enhanced by use of intraoral endoscopy and CT.
Clinical Pathology
The hemogram in animals with sinusitis generally remains within the normal range, although acute sinusitis of infectious origin may be associated with neutrophilia. With chronic sinusitis there may be concurrent hyperfibrinogenemia. Sinus fluid obtained by percutaneous centesis should be examined cytologically (including a Gram stain) and submitted for microbial culture and susceptibility testing to differentiate among bacterial, fungal, and neoplastic disease. Flecks of feed material indicate sinusitis secondary to dental abnormalities.
Laboratory Aids and Definitive Diagnostic Tests
A presumptive diagnosis of sinusitis can be made from the physical examination and associated clinical signs.1175,1181 Procedures most helpful in establishing a diagnosis of sinus disease in 85 horses were radiography (92%), endoscopy (38%), percutaneous centesis (21%), and examination of the oral cavity (20%).1177
Endoscopic examination of the nasal cavity may permit identification of exudate draining from the nasomaxillary opening or in advanced cases may reveal distortion of the nasal cavity secondary to sinus enlargement. Middle meatus examination is an important component of endoscopic examination. Diagnosis beyond recognition of the potential source of the nasal discharge is limited unless there is an obvious mass or abnormal tissue is observed. Sinoscopy can be accomplished by using an arthroscope1182 or flexible endoscope1183 inserted through small trephine holes in either the maxillary or the frontal sinus. Examination of the rostral compartment of the maxillary sinus requires a separate portal unless the bony septum between the rostral and caudal compartments has been destroyed. Observation may be limited by fluid or tissue especially in the rostral compartment of the maxillary sinus but can potentially be enhanced after sinus lavage and aspiration.
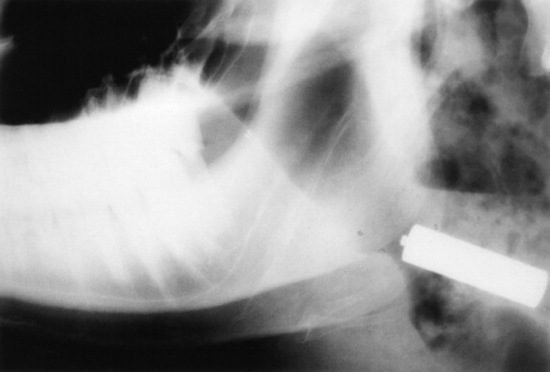
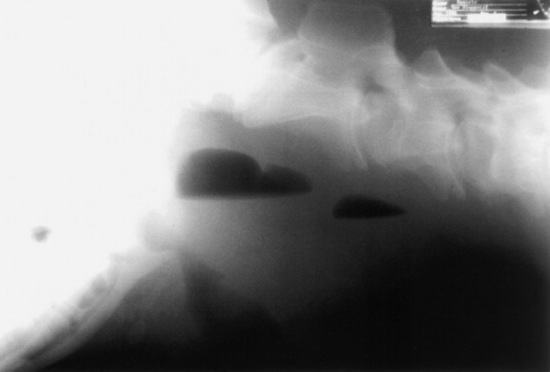
Standard radiographic projections include the standing lateral, dorsoventral, and right and left oblique views.1184,1185 Radiographic findings include fluid lines within the sinus, space-occupying soft-tissue densities, areas of decreased bone density, fractures, or dental abnormalities. Dental root disease is identified radiographically by a loss of continuity of the lamina dura and lysis of the tooth root or surrounding bone, combined with new bone formation and cement deposition.1184 Anatomy of normal equine skulls as demonstrated by CT and MRI has been described.1186-1188 Diagnostic accuracy, especially determination of the extent of involvement of structures within the skull, can be enhanced by CT.1189,1190 CT imaging enhances diagnostic accuracy of tooth involvement.1190 Findings associated with dental caries include hypoattenuation of cementum, destruction of enamel, and filling of the infundibular cavity with gas, whereas with dental decay there is gas accumulation in the root area or fragmentation of the root, and sinus mucosal thickening. Additional changes with sinusitis typically involve the maxilla with endosteal sclerosis, thickening, periosteal reaction, and deformation, especially involving the facial crest.1190 Scintigraphic examination may improve specificity in identification of dental involvement in sinusitis.1191,1192
Percutaneous sinus centesis may provide a definitive diagnosis and allow an avenue for subsequent therapy. Cytologic evaluation, with concurrent microbial culture and antibiotic susceptibility testing, may elucidate the cause of the sinusitis.1177 Isolation of a single organism such as Streptococcus species generally indicates a primary sinusitis, whereas polymicrobial infection is more compatible with sinusitis of dental origin. Visual examination of the oral cavity, especially intraoral endoscopy, and careful probing of the occlusal surfaces with a dental pick may identify dental abnormalities.
Necropsy Findings
Affected sinuses contain fluid or tissue of variable color and consistency. Fluid character ranges from clear and odorless with cystic sinus disease to white, yellow, or green purulent fluid with a variable, but often putrid, odor in sinusitis resulting from other causes. Sinusitis of dental origin has a characteristically pungent and unpleasant odor. Granulomatous lesions have been reported to appear as large lobular gelatinous masses filling the sinus cavity. The gross appearance of neoplastic lesions within the sinus cavity depends on the type of neoplasm. Neoplasia may cause surrounding soft-tissue and bony destruction, whereas large, benign space-occupying lesions may result in distortion of the nasal turbinates and nasal septum, as well as external facial bone distortion.
Treatment and Prognosis
Not infrequently, horses with a chronic mucopurulent nasal discharge from sinusitis have a history of response to antimicrobial therapy, followed by recurrence of the discharge after antibiotic therapy ceases. Definitive diagnosis of sinusitis can be accomplished using the techniques described earlier. Sinoscopy permits examination of the paranasal sinuses and in some instances facilitates treatment.1184,1193
Suggested treatment for primary sinusitis or empyema involves daily lavage of the sinus through a percutaneous centesis site with 1 L of saline, to which a broad-spectrum antibiotic or antiseptic has been added. Once the results of culture and susceptibility testing are available, the appropriate antibiotic should be administered locally in the flush solution, as well as systemically, for 14 days. Resolution or reduction in the volume of nasal discharge is an indication of successful therapy. If little progress is made after 10 to 14 days or if drainage recurs, sinusotomy (trephination or bone flap technique, standing or recumbent anesthetized) may be required to resolve the condition.1194 The prognosis is generally favorable if primary sinusitis is not chronic and if the mucous membrane is not markedly thickened.1195 Chronic sinus disease (longer than 6 months) carries a poor prognosis, and, for resolution to occur, surgical removal of the thickened, infected mucous membrane is required; creation of sinonasal drainage by sinus fenestration is also recommended. Isolation of Pseudomonas species from a sinus aspirate generally indicates an unfavorable prognosis.1180
Sinusitis that results from secondary factors is generally not responsive to medical management. Such conditions include diseased teeth, granulomas, or neoplasia; surgical removal of the inciting cause is required, and adjunctive treatment may be required. The prognosis for sinusitis associated with dental abnormalities is usually favorable once the diseased tooth has been removed.1195,1196 If the periodontal ligament is intact, endodontic therapy can be used to save the tooth. This is accomplished by surgical apicoectomy and retrograde occlusion of the root canal after debridement of the pulp. In geriatric horses with dental-associated sinusitis, when economic constraints limit surgical options, sinusotomy and periodic sinus lavage and antibiotic therapy have been used successfully to manage nasal discharge.
The prognosis for resolution of granulomatous lesions is generally guarded and depends on surgical access and extent of the lesion. Neoplastic lesions are often well established and have metastasized, either locally or regionally, by the time they become clinically apparent; the prognosis for resolution is generally guarded to poor.1176 In a series of 16 horses with sinus neoplasia, 11 were euthanized because of the extent of the lesion, four lesions recurred after surgical removal, and one horse with squamous cell carcinoma was successfully treated and had no recurrence at a 2-year follow-up evaluation.1177
Prevention and Control
Prevention of sinusitis in horses is difficult because of the variety of causes. Isolation of horses from those with upper respiratory bacterial or viral diseases may be of benefit in preventing primary sinusitis. Regular dental care and a proper diet may help circumvent sinusitis caused by dental abnormalities, although many cases most likely result from a variety of causes not yet defined or over which the owner or veterinarian has no control.
ETHMOID HEMATOMA
Definition and Etiology
Also termed progressive ethmoidal hematoma1197 and hemorrhagic nasal polyps,1198 ethmoid hematomas are slowly expanding angiomatous masses that appear to originate principally from the mucosal lining of the ethmoid conchae. Smaller hemangiomas arising from the mucosal lining of the frontal, maxillary, and sphenopalatine sinuses have been recognized, but the relationship between these benign endothelial tumors and ethmoid hematoma is uncertain. A relationship between paranasal sinus cysts and ethmoid hematoma has been suggested,1199 but distinctly different histologic features characterize each disorder,1200 seemingly arguing against a common cause and the likelihood that these lesions are variants of each other. The cause of ethmoid hematoma is unknown, and it remains a relatively uncommon condition.1201-1205 Although reported in a 4-week-old foal and in 3-year-old horses, most affected horses are older than 8 years—generally thoroughbred, Arabian, or warmblood horses.1201-1207
Clinical Signs
A blood-tinged nasal discharge with intermittent epistaxis from one or both nostrils is the most common clinical sign.1208 Unilateral or bilateral, epistaxis varies from blood-tinged mucoid or mucopurulent discharge to blood spots or a trickle of blood. Fulminant or fatal epistaxis as can occur with guttural pouch mycosis is uncommon. If the hematoma occupies the choana(e) or nasal cavity, a mucopurulent, occasionally malodorous nasal discharge with some blood discoloration is more commonly seen. Typically these horses have a history of abnormal respiratory noise, both inspiratory and expiratory, especially during exercise. With nasal cavity involvement, airflow is usually reduced or may be absent on the affected side. Facial distortion or asymmetry is uncommon and is more likely to occur when the hematoma occupies the frontal and maxillary sinuses. Less commonly there may be an associated history of coughing, choking, ptyalism, increased respiratory effort during resting breathing, and either head shyness or head shaking.1197 If the hematoma has expanded into the paranasal sinuses, percussion yields a dull sound.
Laboratory Aids and Definitive Diagnostic Tests
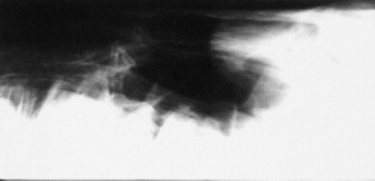
Confirmation requires endoscopy of the ethmoid conchae and skull radiography (Figs. 31-45 and 31-46); however, the origin and extent of the mass can be delineated more accurately by computed tomographic examination of the skull. Sinoscopy may be of diagnostic value in horses with ethmoid hematoma involving the paranasal sinuses without protrusion into the nasal cavity. Rarely, ethmoid hematoma infiltrates the nasal conchae; these lesions, identifiable by CT, may be missed by sinoscopy. Because ethmoid hematoma is bilateral in approximately 30% of affected horses, it is prudent to examine both the left and right ethmoidal conchae. The ethmoidal labyrinth is visible approximately 25 cm from the nares, with the endoscope positioned in the ventral nasal meatus and the viewing tip deflected dorsally. The rostral surface of the ethmoidal concha does not protrude beyond the caudal nasal cavity and has a bulbous shape and a moist pink to pale red mucosal covering.
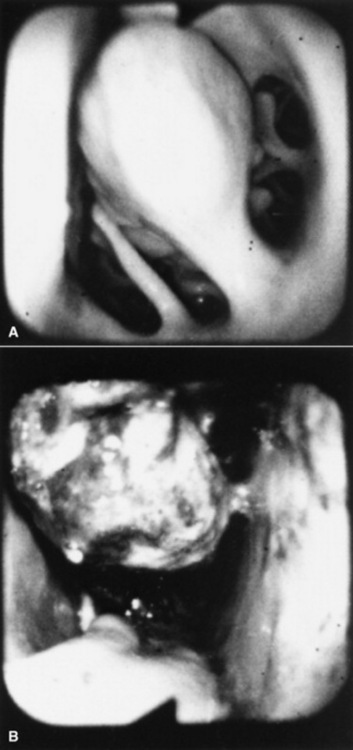
Fig. 31-45 Endoscopic view of left ethmoturbinate. A, Normal appearance. B, Ethmoturbinate obscured by an ethmoid hematoma.
Beyond the rostral surface the numerous pillars that form the ethmoidal conchae and separate the ethmoidal spaces (cellulae ethmoidales) are visible. Ethmoidal hematomas that project into the ventral meatus or through the choana into the nasopharynx often obscure the ethmoidal concha. Occasionally, unilateral ethmoid hematomas that have expanded into the nasopharynx may protrude into the contralateral ventral meatus, obscuring the view of the ethmoidal labyrinth on that side. The origin of hematomas that expand dorsally into the frontal sinus may not be visible on endoscopy, but hemorrhage that originates deep to the visible portion of the ethmoidal conchae may be evident or may be noticed from the region of the nasomaxillary opening in the middle meatus. Visible ethmoid hematomas can vary in color from deep red to red-purple or may have a yellow-brown or yellow-green-brown to bronze color. The surface is irregularly rounded, with small punctate hemorrhages or erosions, and may be partially covered in yellow-white mucopurulent material that may be admixed with blood. Often the floor of the ventral meatus and the regions of contact with the nasal cavity have pooled exudate of blood and mucopurulent matter. Manipulation of the visible surface of the hematoma with the tip of the endoscope may elicit bleeding or oozing.
Recognition of a discrete, often smooth-surfaced homogeneous radiodensity originating from the ethmoidal conchae and extending into the frontal, maxillary, or sphenopalatine sinuses or into the pharynx or nasal cavity is suggestive of an ethmoid hematoma. Radiography is beneficial in determining the extent of the hematoma and in identifying suspected ethmoid hematomas that are not visible by endoscopy; however, precise definition of the origin of any hematoma is difficult from radiographic projections. Small hematomas contained within the ethmoid labyrinth may not be visible on radiographs. Computed tomographic examination of the skull allows more accurate assessment of the origin of the ethmoid hematoma,1207,1209 allows determination of the extent of involvement of the paranasal sinuses and conchae, and facilitates surgical planning.
Necropsy Findings
Most of the morphologic features have been described from surgical specimens; few skulls with intact ethmoid hematomas have been examined.1198 Except in regions with necrosis or secondary infection associated with contact with the sinus or nasal cavity walls, the hematoma is a smooth-surfaced saclike structure containing blood in various stages of organization. The sac lining is generally healthy respiratory mucosa originating from a pedunculated region of the mucosal covering of the ethmoturbinal or sinus wall. On section the contents are amorphous red-black to chocolate brown, and in larger masses some evidence of irregular compartmentalization by fibrous tissue exists, especially on the inner surface of the sac.
Morphologic features include an outer covering of respiratory epithelium (flattened columnar or cuboidal ciliated epithelium containing glands, and occasionally stratified squamous epithelium1200) overlying an irregular zone of submucosal fibrous tissue, containing hemosiderophages, occasional plasma cells, and lymphocytes, less commonly neutrophils, that forms a pseudocapsule around hemorrhage in varying states of organization. There is typically variable organization of the fibrous tissue components. Endothelial cells do not show evidence of neoplasia. Thin endothelium-lined sinuses are often present within the myxomatous stroma. The respiratory epithelium is sometimes focally ulcerated and infiltrated with neutrophils, and occasionally there are squamous metaplastic changes. Ethmoid adenocarcinoma with a similar gross appearance to ethmoid hematoma has been reported in one horse.1210
Treatment and Prognosis
Because these masses slowly and progressively increase in size and can cause distortion of skull architecture if the paranasal sinuses are involved, removal is recommended. Treatment method depends on the location and size. Surgical ablation has been the preferred method; however, destruction of the hematoma by intralesional injection of formaldehyde solution is associated with less morbidity, although recurrence rates are similar to those with other methods.1204-1212 Surgical access is usually achieved by sinusotomy and then hematoma ablation by curettage, cryosurgery, or use of an Nd:YAG laser1213-1215; photoablation can also be accomplished through the biopsy channel of an endoscope. After sinusotomy, the pedunculated origin of the hematoma is identified by digital palpation and then dissected, frozen, or photoablated, and the hematoma removed. If the hematoma is friable, intact removal may not be possible, and hemorrhage may make observation of the origin of the mass difficult. After removal the paranasal sinuses and nasal cavity are packed with gauze to control postoperative hemorrhage.
Surgical curettage can have the disadvantage of being associated with marked intraoperative blood loss typically from the turbinates or sinus mucosa rather than the hematoma. Temporary occlusion of both carotid arteries1214 can substantially decrease blood loss until the sinus cavity is packed with gauze. Blood loss is minimized by cryosurgical extirpation and photoablation techniques, but these approaches are not always practical when initially dealing with large hematomas. There is also minimum blood loss with transnasal photoablation; however, this technique requires multiple procedures to destroy large masses but can be performed in the standing sedated horse. Photoablation (Nd:YAG laser, 100 W in noncontact technique) is effective in controlling remnants after surgical extirpation or subsequent regrowth.1215,1216
Destruction of ethmoid hematomas by endoscopically guided intralesional injection of formalin in standing sedated horses may reduce the need for surgical ablation in many horses.1211,1217 A catheter passed through the endoscope biopsy channel is advanced through the rostral surface of the mass toward its origin, then 10% formalin (4% formaldehyde solution) is injected intralesionally. Commercially available catheters with a beveled needle tip have been used, but relatively stiff plastic tubing that will slide through the biopsy channel works will penetrate the capsule of the ethmoid hematoma. Sufficient volume (from 10 to 100 mL) is injected until fluid leaks back alongside the catheter or leaks from the mass. For visibly pedunculated masses, it is best to inject the formalin at the origin or neck of the mass. Tissue necrosis and slough occur in 5 to 10 days and may be associated with nasal discharge. Repeat injections, typically at no longer than 10- to 14-day intervals may be necessary to destroy the mass. Removal of necrotic tissue can be facilitated by use of long grasping forceps and hydropulsion. Mycotic plaques may cover the treated site during healing but typically resolve without treatment; endoscopically delivered topical natamycin has been used if mycotic plaque is extensive and associated with malodorous, purulent discharge.1218
Occasionally progression of the ethmoid hematoma may result in weakening or loss of the cribriform plate or roof of the sphenopalatine sinus.1218 During mass removal, loss of this protective bony covering may result in intraoperative or postoperative neurologic complications. Recognition of loss of the integrity of these bony plates may not occur until sinus lavage. In a rare complication death occurred after intralesional formalin injection in a horse where the cribriform plate had been eroded by the ethmoid hematoma.1219 Erosion of calvarial bone is rare, but careful computed tomographic evaluation of these regions in horses with extensive hematoma formation involving the frontal or sphenopalatine sinuses is warranted.
Irrespective of treatment method, recurrence of the hematoma occurs in 30% to 50% of cases from several months to years after the initial surgery.1198,1203-1206,1211,1212
DISEASES OF THE NASAL CAVITY
Mycotic or Bacterial Nasal Granuloma
Infectious granulomas in the nasal cavity of ruminants are not common. Documented causes include the fungal organisms Rhinosporidium seeberi and other Rhinosporidium species (which cause rhinosporidiosis), Helminthosporium species (which cause maduromycosis), Drechslera rostrata, Aspergillus species, Phycomycetes species, Stachybotrys species, and Bipolaris species. Phycomycosis is discussed in the dermatology section in Chapter 40. Nasal granuloma caused by Nocardia species bacteria has also been reported.1 There is no apparent age, breed, or seasonal predilection, and cases are sporadic. The major clinical signs are upper respiratory noise (stridor), dyspnea, and mucopurulent nasal discharge, sometimes with epistaxis. Affected animals may rub the nose, suggesting pruritus or irritation.2-3 Nasal airflow may be reduced, and open-mouth breathing may occur in advanced cases. Hot or dusty weather may accentuate the signs, giving the appearance of seasonal exacerbation, but the lesions are progressive. The granulomas may be single or multiple, unilateral or bilateral, and located anywhere in the nasal cavity. They consist of 0.5- to 5-cm yellow to yellow-green or red nodules or polyps, which may be sessile or pedunculated. Rhinosporidiosis tends to be a single unilateral polyp in the posterior nasal cavity, and maduromycosis tends to occur in the anterior cavity, but these distinctions are not consistent. Red and black spots (spores) may occur on the masses, and some may become secondarily infected with bacteria and ulcerate. Differential diagnoses include allergic rhinitis, foreign bodies, tumors, nasal actinobacillosis, nasal actinomycosis, and Oestrus ovis infection in small ruminants.
Endoscopy, biopsy, and culture of the lesions aid in the diagnosis. Histopathologic analysis reveals granulation tissue containing eosinophils, mononuclear cells, round sporangia, and sometimes hyphae or filamentous bacteria.1,2 The pathogenesis of the disease involves inoculation of eroded nasal mucosa with fungal spores or filamentous bacteria from the environment. The infectious agent causes a chronic delayed (type IV) hypersensitivity reaction, which eventually leads to the formation of a granuloma. Fungal granulomas can be more common in warm, wet climates. The granulomas can be difficult to treat, and although rarely fatal the disease is chronically debilitating, with salvage often being the most practical solution.
Recommended treatments include surgical removal of the granulomas when possible and long-term sodium iodide (NaI) therapy. NaI can be administered at a dose of 66 mg/kg IV as a 20% solution, repeated at 10- to 14-day intervals until remission or iodism occurs. Iodism is characterized by lacrimation, cough, and scaling of the skin. The use of antifungal drugs to treat this condition in ruminants has not been reported.
Allergic Rhinitis and Enzootic Nasal Granuloma
Allergic rhinitis occurs in cattle and in its chronic stages may lead to the formation of granulomas. A similar condition may occur in sheep.4 The inciting antigen is frequently a plant pollen or more likely a fungal spore.5 Once homocytotropic antibody (immunoglobulin E or possibly other classes in cattle) to the allergen has developed, subsequent exposure results in a localized, ongoing, immediate (type I) hypersensitivity reaction.5-7 If recurrent exposure to the allergen occurs, repeated tissue damage by mast cell factors results in chronic epithelial, duct, and goblet cell hyperplasia and metaplasia, as well as mucous hypersecretion and granulomatous inflammation, suggesting that a type IV hypersensitivity reaction contributes to the chronic lesion.5,6,8
Any breed may be affected, but Channel Island breeds and Friesians seem most susceptible.5 The disease occurs sporadically in the United States. A familial predisposition has been reported.9 Most affected animals are between 6 months and 2 years of age. The signs are initially seasonal, usually occurring in warm, moist conditions; they include rhinorrhea, sneezing, nasal pruritus, a sudden onset of dyspnea, and stertorous inspiration.9,10 There is a profuse bilateral nasal discharge. Intense pruritus is characteristic and associated with sneezing, head shaking, and nose rubbing.10 In severe cases facial swelling, tachypnea, hyperpnea, and ulceration of the nasal mucosa may occur.6,10 Nasal foreign objects may result from the animal’s attempts to scratch the nasal mucosa. Lacrimation, chemosis, and blepharitis may also be present. In the chronic stages (the “enzootic nasal granuloma”), the signs are more constant, with seasonal exacerbations.6,8,10 The granulomas tend to be multiple, firm, white, raised nodules 1 to 2 mm in diameter with an intact mucosa, or pale pink flat plaques scattered throughout the nasal cavity. Differential diagnoses include fungal granulomas, foreign bodies, respiratory viruses, nasal actinomycosis or actinobacillosis, tumors, O. ovis infection (small ruminants), and irritation caused by inhalation of hot or irritant gases.
Endoscopy, biopsies, cultures, antigen detection tests for viruses, bacteria, or fungi, and serologic analysis can be used to rule out these differential diagnoses. Eosinophil counts in nasal secretions correlate with the susceptibility of the animal and activity of the disease, but no absolute level is diagnostic.5 Intradermal allergen testing has been suggested to aid in diagnosis, but interpretation of results needs to be done in conjunction with historical and clinical findings.7 This condition should be differentiated from fungal or bacterial granuloma because the therapy is different.
Treatment and control entail removal of the allergen, or removal of the animal from the allergen, and therapy to block the hypersensitivity reaction. Recommended drugs include various antihistamines, meclofenamic acid, and corticosteroids at standard antiinflammatory doses (0.05 to 0.2 mg of dexamethasone per kilogram IM or IV or 1 to 2.2 mg of prednisolone per kilogram IM or IV daily). Topical corticosteroid therapy can be considered in severe, acute occurrences of the disease. The adverse effects of corticosteroids on milk production and their potential to induce abortion or parturition should be considered before their use. Antihistamine therapy has had equivocal results.7
Nasal Foreign Bodies
Cattle are more prone than small ruminants to the acquisition of nasal foreign bodies. Foreign objects may be acquired as a result of attempts to scratch the nose in cases of allergic rhinitis, or because of the cow’s aggressive eating habits. Depending on the size and duration of residence of the object, signs may include head shaking, stridor, sneezing, snorting, frequent nose licking, unilateral decreases in airflow, foul odors, and serous, mucopurulent, or hemorrhagic discharges. Differential diagnoses include fungal granulomas, allergic rhinitis, tumors, nasal actinomycosis or actinobacillosis, and O. ovis (small ruminants). Many objects can be visualized on careful examination of the nasal cavity with an adequate light source, whereas some may require endoscopy for diagnosis and removal.
Nasal Trauma and Fractures
Trauma to the facial bones, sinuses, and turbinates may result from fighting, accidents caused by improper restraint, farm machinery accidents, human maliciousness, and passage of excessively large NGTs. Severe fractures can lead to facial swelling, SC emphysema, obstruction of airflow, stertor, and epistaxis. Secondary infection causes foul odors and mucopurulent nasal discharge. Differential diagnoses for the acute external swelling of the head with stertor include snakebite, actinobacillosis, actinomycosis, and phlegmon (Fusobacterium, Clostridium species). Unless the development of severe depression fractures, formation of sequestra, or severe obstruction of airflow occurs, surgery is usually not indicated. Radiographs confirm the diagnosis and help determine the need for surgical removal of potential sequestra or elevation and fixation of large displaced segments. Prophylactic antibiotics (typically penicillin, 22,000 U/kg IM or SC q12-24h) are recommended to prevent fracture infection and sinusitis, and NSAIDs (aspirin, 100 mg/kg PO twice; flunixin meglumine, 1.1 to 2.2 mg/kg IV daily or divided twice daily) may help relieve pain, swelling, and stridor. The prognosis is usually good.
Nasal Tumors and Polyps
Tumors and polyps of the nasal cavity and sinuses are rare in ruminants. Nasal tumors reported in cattle include osteomas and osteosarcomas of the sinuses, squamous cell carcinomas,11 neuroblastomas, and adenocarcinomas of the ethmoid mucosa. Ethmoid adenocarcinomas are speculated to be caused by viruses on the basis of an endemic pattern in some cases.12 They tend to occur in cattle 6 to 9 years of age and are frequently unilateral. Metastasis occurs to the lymph nodes and lungs. There is a report of a hemangiosarcoma involving the external naris of a cow.13 Signs common to all nasal tumors include mixed or inspiratory dyspnea, stridor, nasal discharge, epistaxis, foul breath odors, unilateral decreases in airflow, open-mouth breathing, and distortion of the facial bones. Differential diagnoses include fungal granulomas, atopic granulomas, foreign bodies, sinusitis, fractures, and nasal actinobacillosis and actinomycosis. Treatment has not been investigated.
The majority of nasal neoplasms in sheep and goats are adenopapillomas, adenomas, or adenocarcinomas.14 Squamous cell carcinoma15 and osteoma16 have also been reported. Nasal adenocarcinomas have also been described in goats.17
An enzootic form of nasal adenocarcinoma occurs in both sheep and goats and is associated with ovine nasal adenocarcinoma virus (ONAV)18,19 or caprine nasal adenocarcinoma virus (CNAV),20 respectively. These agents are β retroviruses that are closely related to, but distinct from, jaagsiekte sheep retrovirus (JSRV), the cause of ovine pulmonary adenocarcinoma (OPA).20,21 Although nasal adenocarcinoma has been difficult to consistently reproduce experimentally, inoculation of kids with concentrated cell-free and bacteria-free filtrate containing virus from naturally infected goats has resulted in disease.22 Neoplastic transformation is limited to secretory epithelial cells of the nasal turbinates, but CNAV appears to have a wider tissue tropism than ONAV; in one study viral provirus incorporated into host DNA was found in many tissues of infected goats but was largely confined to tumor tissue of infected sheep.20 There is no breed or sex predisposition for enzootic nasal adenocarcinoma; affected animals are most commonly young adults, but the tumor has been identified as early as 4 months of age.18 Signs include progressive inspiratory dyspnea; stridor; exercise intolerance; mouth breathing; serous, mucoid, or mucopurulent nasal discharge, which is typically profuse; tachypnea; decreased airflow; head shaking; sneezing; exophthalmos; and facial asymmetry.14,17,18,21 The lesions may be unilateral or bilateral, originating in the olfactory region of the ethmoid turbinates. The neoplasia arises either from Bowman’s glands18 or serous glands of the nasal mucosa.17 The tumor begins as a small nodule that can grossly resemble the mucoid polyps that occur in animals with chronic rhinitis. Over time the adenocarcinoma grows into a soft, gray to grayish pink, mucoid, nodular, cystic mass. The tumor is benign but locally expansive, often entering the sinuses and eroding overlying bone.17 Histologically the tumor is typically classified as a low-grade adenocarcinoma, but it may also be identified as an adenopapilloma or adenoma.17 Initially affected animals eat and drink normally and maintain body condition, but as the tumor expands the animal begins to lose condition, and death eventually occurs as a result of inanition, asphyxia, or aspiration pneumonia. Necrosis or secondary bacterial infection of the tumor can occur and may lead to the production of foul-smelling, purulent discharge and systemic signs related to bacterial infection, such as fever, depression, and hyperemic mucous membranes.
Differential diagnoses for nasal neoplasia in small ruminants include nasal fungal or bacterial granuloma, actinobacillosis, actinomycosis, O. ovis infection, and sinusitis. Endoscopy and radiology are helpful in establishing an initial diagnosis of a nasal mass. Preoperative or antemortem pinch biopsies and exfoliative cytologic analysis frequently are nondiagnostic, and findings may be misleading.14,15 Identification of tumors in multiple animals in a herd or flock supports a diagnosis of enzootic nasal adenocarcinoma. Definitive identification of infection with ONAV or CNAV can be difficult. Serologic tests are not reliable because animals do not consistently produce antibody to the viruses,23 possibly because of the presence of endogenous retroviruses24 that share epitopes with ONAV and CNAV and induce the development of immunologic tolerance. Recently PCR has been used to specifically identify and differentiate ONAV, CNAV, and JSRV in tissues of affected animals.25 Surgical management of nasal adenocarcinoma in sheep has been described.26
OESTRUS OVIS INFESTATION
Definition and Etiology
O. ovis is parasite of the nasal passages and sinuses of sheep and, less commonly, goats. Goats are relatively resistant to infection and, even when housed with sheep, tend to have a lower prevalence of infection.27 Occasionally humans and other animals are accidentally infected, with conjunctival infection the most common form of disease in humans.28,29 The pathogenic stage of the parasite is the larval stage. The first instar larvae are deposited near the nostrils of sheep by the adult female fly and migrate into the nasal and ethmoid turbinates; in the ethmoids the larvae molt to second instars, which migrate to the sinuses before molting again and becoming third instar larvae. Third instar larvae, which are yellow-white in color and have a dark dorsal stripe with rows of spines on the ventrum of each segment, return to the nasal passages and are sneezed out onto the ground, where they pupate and eventually develop into adult flies.30,31 The adult is active during warm months, and the parasite may overwinter either as a first instar larva in the host or as a pupa in the ground. Larvae can persist in the upper airways for weeks to months, and appear to be able to arrest development for a period of time if necessary to avoid climate extremes.32 Adult flies have a rudimentary mouthpiece and are not able to feed; thus the larvae must ingest adequate nutrients while in the host to support the life of the adult fly.33
Clinical Signs
The larvae cause irritation of the nasal passages and sinuses, leading to mucoid to mucopurulent and sometimes blood-tinged nasal discharge, sneezing, nose rubbing, and inspiratory stridor. Adults flies cause annoyance by flying around the heads of animals; thus both stages can lead to decreased productivity by decreasing the time spent grazing by affected animals. Occasionally the larvae may cause sinusitis or pneumonia owing to secondary bacterial infection associated with irritation caused by the larval infestation. Differential diagnoses to consider include nasal foreign bodies, allergic rhinitis, nasal adenocarcinoma, fungal rhinitis, trauma, sinusitis, actinobacillosis, or actinomycosis.
Pathogenesis
The larvae cause direct irritation to the nasal passages and sinuses, and this irritation may predispose animals to the development of secondary bacterial rhinitis, sinusitis, and/or pneumonia. Interstitial pneumonia has also been seen on occasion in affected sheep, presumably induced by inhalation of parasite antigens and inflammatory mediators from the upper respiratory tract.34
Epidemiology
Infection is more common in warm climates. Recent European surveys have identified O. ovis in 35% to 91% of sheep surveyed,35,36 with seroprevalences of 46% to 69%.27,36,37 A majority of flocks surveyed had at least one infected animal.27,35 No North American reports describing the prevalence of O. ovis infestation have been published in recent decades.
Diagnosis
Diagnosis is usually presumptive, based on typical clinical signs. The larvae can also be identified via radiographs of the head or by endoscopy, but these tests may not be practical for field use. Serologic diagnosis has been used for research studies of the epidemiology of O. ovis infection, but these tests are unlikely to be available at most diagnostic laboratories.
Treatment and Prevention
The larvae are susceptible to ivermectin at 200 μg/kg PO.38 Moxidectin at 0.2 mg/kg as a 0.1% oral drench was not effective against O. ovis39; these investigators found that the same dose given as a 1% injectable solution was effective, but this route is not approved in the United States. Pour-on eprinomectin at 0.5 mg/kg (applied immediately after shearing)40 and injectable doramectin at 200 μg/kg IM41 have also been shown to be effective in sheep, but these treatments are also not approved for use in sheep or goats in the United States. Because the adult fly should be killed by freezing weather, in areas with a season of freezing weather it is logical to treat after the first hard freeze, when sheep will no longer be susceptible to infection until warm weather returns. Prevention is aimed at regular strategic treatment with effective anthelmintics to prevent long-term infection with the larvae.
CONGENITAL CYSTIC NASAL TURBINATES IN CATTLE
An apparently developmental anomaly that results in signs of nasal obstruction has been reported in cattle.42 The nasal conchae lack the normal communication with the nasal cavity and become filled with a thick white fluid, which may account for the enlargement. Signs are evident at or near birth and include progressive stridor, tachypnea, decreased airflow, exercise intolerance, mouth breathing, and short, convex nasal bones. Digital, radiographic, and endoscopic examinations of the nasal cavity reveal large, smooth, bilateral cystic ventral nasal conchae, often bilobate. Differential diagnoses include foreign bodies, trauma, and tumors. Surgical removal of the conchae with bilateral dorsolateral nasal bone flaps relieves the obstruction. Transnasal removal using obstetric wire has been described.43
SINUSITIS
Definition and Etiology
Inflammation of the paranasal sinuses is most common in cattle and occurs infrequently in sheep and goats. Typically the frontal or maxillary sinuses are involved, and a variety of bacteria may be isolated. The proximate cause for infection is usually dehorning (frontal sinusitis) or infected teeth (maxillary sinusitis). Other causes include extension of actinomycosis or nasal neoplasia into the sinus, injuries to the horn, facial fractures, respiratory viruses (including malignant catarrhal fever [MCF], IBR, and parainfluenza viruses), sinus cysts,44,45 lymphosarcoma,46 and O. ovis (in sheep).47-50
Clinical Signs and Differential Diagnosis
Sinusitis associated with dehorning may be acute or may occur weeks to months later; typically only one sinus is affected. Nonspecific clinical signs include anorexia, lethargy, reluctance to move, and fever. When sinusitis occurs acutely after dehorning, the portal of entry is frequently open and discharging pus, and the animal is often febrile (39.5° C to 40.5° C). In chronic sinusitis, signs may include unilateral or bilateral nasal discharge, mild stridor, changes in airflow, and foul breath odors that are frequently unilateral; fever is not common. The animal may hold its head at an odd angle (extended up or down, tilted) and may squint the eyelids as if in pain. 49,51 With chronicity, frontal bone distortion, exophthalmos, and neurologic signs may occur.51,52 In one report of 12 cattle with frontal sinusitis, four cattle had abnormal posture, with an extended head and neck, partially closed eyes, and a tendency to head-press or to rest the head on a stationary object; the other 8 cattle were apprehensive and intolerant of head manipulation.51 Extension of infection may involve the CNS.
Occasionally sinusitis irritates the animal sufficiently that it may rub its head on the ground, driving more debris into the sinus.50 Maxillary sinus cysts have been observed in cattle.44 Typical signs included unilateral facial swelling over the affected sinus; mucopurulent, nonfetid nasal discharge; and radiographic evidence of septal deviation. One cow had stertorous respiration with diminished airflow. Differential diagnoses for sinusitis include facial fractures, nasal tumors, actinomycosis, actinobacillosis, retrobulbar abscess, and lymphosarcoma.
Diagnosis
Diagnosis can usually be made based on clinical signs. Diagnostic aids may be useful in selected cases, particularly when there is no recent history of dehorning, or in cases of maxillary sinusitis. The hemogram is quite variable and is of little assistance in diagnosis. Percussion of the sinus may reveal a dull, full sound and may elicit pain. If the bone has been greatly thinned and has gas underlying it, percussion may produce a hyperresonant sound. Fractures, soft-tissue masses, dental disease, fluid in the frontal sinus, or lysis of bony septa may be evident on radiographs.51 Sinus centesis may yield purulent material, which should be cultured and examined cytologically. A small area over the affected sinus is clipped and surgically prepared, and local anesthetic is infiltrated subcutaneously. Then a small stab incision is made through the skin and periosteum, and a Steinmann pin is used to drill a small hole. Polyethylene tubing is inserted, and attempts are made to aspirate material. A small amount of sterile isotonic fluid can be injected and aspirated to obtain a washing. The stab incision is closed unless sinusitis is confirmed.
Treatment and Prognosis
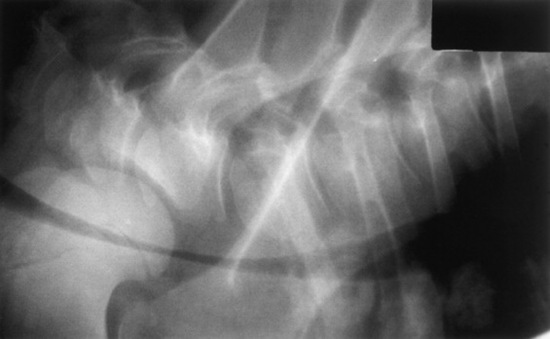
Trephine sites for sinusotomy (Fig. 31-47) are as follows:
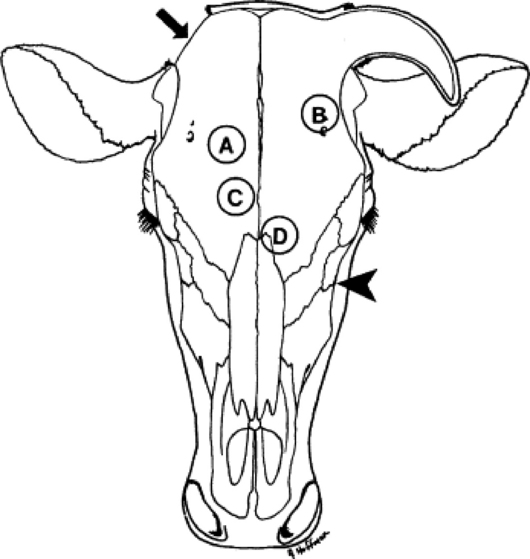
Fig. 31-47 Trephine sites for sinusotomy. A, Dorsal frontal sinus. B, Postorbital diverticulum. C, Rostral frontal sinus. D, Turbinate portion of frontal sinus. The maxillary sinus is trephined ventral to a line from the infraorbital foramen to the medial canthus (arrowhead). If draining tracts are present at the poll, an additional sinusotomy can be made in the cornual portion of the frontal sinus (arrowhead).
Cattle that have frontal sinusitis after dehorning should be treated by sinusotomy and drainage of the sinus.53 Sinusotomy sites should be based on anatomic landmarks51 and modified as needed to accommodate any frontal bone distortion or wounds related to dehorning.51 Sinusotomy should be performed 3 to 4 cm from midline, intersecting a line drawn between the caudal aspect of the orbits. If draining tracts are present at the poll, an additional sinusotomy can be made in the cornual portion of the frontal sinus.51 Sinusotomy is performed after sedation and local anesthetic infiltration of the centesis site(s). A 2-cm—diameter circular piece of skin is excised, and a 19-mm (¾-inch) trephine used to create an opening into the sinus, through which purulent fluid should be evacuated and the sinus lavaged.
Additional trephine sites that permit access to other regions of the frontal sinus include the postorbital diverticulum, which is trephined approximately 4 cm caudal to the dorsal rim of the orbit, just above the temporal crest of the frontal bone; the rostral frontal sinus, which is trephined just caudal to a line between the centers of the orbits and to either side of the midline; and the turbinate portion of the frontal sinus, which is trephined just rostral to the line described and to either side of the midline.
Access to the maxillary sinus is achieved by trephining ventral to a line from the infraorbital foramen to the medial canthus. If an infected tooth is the cause of maxillary sinusitis, a sinusotomy is created with a trephine over the affected tooth to repel it; otherwise, the hole is usually made just dorsal and caudal to the facial tuberosity. The trephine site should be higher in the sinus of younger animals because the tooth roots are longer.
If the frontomaxillary and nasomaxillary openings are still patent, one trephine site may be sufficient, with the natural opening providing ventral drainage. In more chronic cases, two trephine sites (for ingress and egress) are needed. Another alternative in chronic sinusitis is the use of a curved steel sinus probe (1 cm diameter × 55 cm long), which is forcefully driven through the septal plates of the frontal sinus into the nasal meatus for ventral drainage.54 The frontal sinus is very compartmentalized in mature sheep and goats, and effective drainage is difficult. Therapy should therefore be aggressive in these species, even in early cases, and double trephination or bone flaps for exposure and curettage should be considered.
If a tooth has been repelled, a roll of gauze or dental impression material should be used to occlude the alveolar socket to prevent feed material from entering the sinus. A strip of umbilical tape tied around the gauze roll or a wire in the dental material is passed through socket, sinus, and trephine hole and secured to the face by tying around another roll of gauze as a stent. These gauze packs are replaced each time the sinus is flushed. The sinus is lavaged daily with dilute antiseptic solutions such as 0.1% povidone iodine or chlorhexidine in saline, or 1:1000 potassium permanganate. Lavage is continued until infection is resolved. Enzymes (papain or 200,000 U of streptokinase and 50,000 U of streptodornase in at least 10 mL of normal saline solution) may help remove thick exudate.
Parenteral antibiotics and NSAIDs (aspirin, 100 mg/kg PO twice daily or flunixin meglumine, 1.1 to 2.2 mg/kg IV daily or divided twice daily) are indicated if systemic signs are present. In the absence of microbial culture and susceptibility results, penicillin (22,000 U/kg IM or SC q12-24h) is recommended as the antibiotic of choice because Arcanobacterium (Actinomyces) pyogenes is the most common organism isolated from cattle with chronic frontal sinusitis resulting from dehorning. Pasteurella multocida is the most common organism isolated from infections of the frontal sinus not associated with dehorning.51 Penicillin may be effective against some isolates of P. multocida; alternatively, oxytetracycline can be administered if P. multocida is suspected (11 mg/kg IV or SC q24h, or 20 mg of long-acting oxytetracycline per kilogram SC q72h). If bacterial culture and susceptibility results are available, antimicrobial therapy should be modified accordingly. Early cases often resolve in 10 to 14 days, with a good prognosis. Long-term therapy (weeks) is frequently needed in chronic cases; the prognosis is more guarded, and salvage is often the best option.
Prevention and Control
Dehorning ruminants as neonates, particularly if a “closed” method such as a dehorning iron is used, is the most effective way to prevent frontal sinusitis. In larger cattle surgical dehorning with primary skin closure achieved under aseptic conditions minimizes the likelihood of sinusitis.55 When this is not practical, dehorning should be avoided in rainy, windy, or dusty conditions, and fly control must be used. The dehorning of mature sheep or goats leaves massive wounds that typically take 4 to 6 weeks to close by second intention and so are susceptible to infection; special care, such as bandaging for the initial 7 to 10 days, must be taken.56 Sinusitis did not occur in goats aged 2 to 24 months dehorned with a technique in which primary skin closure was achieved.57