ABORTION
MARES
Information on the major causes of abortion in horses is summarized in Table 43-7.
NON-NONINFECTIOUS CAUSES
Twin Pregnancy
The incidence of abortion caused by twin pregnancy has decreased greatly because of widespread use of ultrasound in diagnosing early pregnancy in mares. Twin pregnancies are routinely detected as early as 12 to 14 days, at which time the twins can be manually reduced to a singleton pregnancy (discussed later). However, despite the overall decrease in the number of twin-related abortions, twinning still remains an important noninfectious cause of abortion in the mare.210 The mare is unable to successfully support two fetuses to term and only in very rare cases (<2%) is able to carry both fetuses to term and deliver without dystocia.211 It is much more common for the mare to have a natural reduction to a singleton pregnancy during the embryonic stage or to abort the pregnancy after the fetal stage.212,213 Some breeds more commonly than others are diagnosed with twins during the embryonic stage (e.g., thoroughbreds have 20% to 30% double ovulations and 10% to 15% diagnosed twin pregnancies).
Clinical Signs and Diagnosis
Mares should be evaluated with transrectal ultrasound 12 to 14 days after documented ovulation during a breeding cycle. The uterus should be carefully scanned from the tip of one horn to the other and along the uterine body to the cervix. Equine embryos at this stage are highly mobile within the uterus and may be located anywhere within the uterus. Increased uterine edema and fluid within the uterine lumen are often negative signs of pregnancy, but pregnancy should not be ruled out until the entire uterine tract has been scanned for the presence of embryos. After one embryo is found, the remainder of the uterus still must be visualized to ensure the absence of a twin. The embryo proper per se is not visible at this stage of pregnancy, but the embryonic vesicle is easily visualized. Care must be taken not to confuse endometrial cysts with embryos, which sometimes can look deceptively similar. It is very helpful to have scanned the mare’s uterus with ultrasound before breeding to document the size and location of endometrial cysts.
Treatment and Prognosis
If twins are confirmed before 16 days of pregnancy, the best way to treat the mare is to manually crush one of the vesicles. This is done by visualization of the twins using transrectal ultrasound and spatially separating them from each other within the uterus. One twin is moved away from the other to the tip of a uterine horn where it is crushed manually. The remaining embryo should be evaluated 1 to 2 days later to determine its status. Survival of the remaining embryo is greater than 90% when twin reduction is performed in this manner before day 20.214
It is the established practice of some veterinarians to treat mares with a nonsteroidal antiinflammatory agent (e.g., flunixin) and/or altrenogest after a twin-crush procedure. The reasoning behind this practice is that manual stimulation of the uterus may cause a release of oxytocin, signaling a prostaglandin cascade that will result in luteolysis and complete pregnancy loss. There have been no studies performed to support this theory; to the contrary, other studies have shown that the release of prostaglandin after the routine twin-crush procedure does not cause a drop in progesterone and that supplementation with antiinflammatory drugs or progesterone makes no difference in the outcome.214,215
Other techniques described for reduction of twins to a single embryo include transvaginal, ultrasound-guided fetal aspiration216; transcutaneous, ultrasound-guided fetal intracardiac injection of procaine penicillin or potassium chloride217; and craniocervical dislocation.218 The success of these techniques is lower than that of manual crush, and they are performed later in gestation, after the window for manual crushing has been missed. These are all specialty techniques and are beyond the scope of this chapter.
Other Noninfectious Causes of Equine Abortion
In a survey of causes of equine abortion in the United Kingdom from 1988 to 1997, nearly 39% were caused by umbilical cord pathology.219 Most of the cases (35%) resulted from torsion of the umbilical cord. Twisting of the umbilical cord is common in equine fetuses and often is not pathologic. A diagnosis of abortion caused by umbilical torsion should be made only if localized swelling and discoloration accompany the twisting. Congenital abnormalities may cause abortion. The most common congenital abnormality in equine fetuses is contracted tendons, accounting for 5% of abortions in a study from the United States.210 Fetuses that implant in the uterine body instead of the base of one of the horns develop placental villous atrophy and usually abort, accounting for 2% of abortions in the same study.
INFECTIOUS CAUSES
Placentitis
Placentitis is the leading cause of equine late-term pregnancy loss in the United States.210 Bacterial organisms most commonly cultured from aborted fetuses include Streptococcus, species, E. coli, Pseudomonas, species, Klebsiella, species, Staphylococcus, species, and Leptospira, species. Nocardioform actinomycetes are an important cause of placentitis in central Kentucky and have recently been reported in Florida.220 The route of infection and the pathophysiology of Nocardia, abortions are poorly understood. Aspergillus fumigatus, and Mucor, species are the most commonly diagnosed causes of mycotic abortion in mares. Fungi cause 5% to 30% of infectious equine abortions.5
The route of placental infection is most commonly ascending via the cervix, which results in loss of chorionic villi around the cervical star. In addition to loss of chorionic villi, additional allantochorionic lesions may include nodular cystic allantoic masses, edema, necrotic areas of chorion, and necrotic mucoid exudate coating the chorion. Hematogenous placentitis shows a more generalized, diffuse loss of villi.210 The distribution of the avillous chorionic lesion classically noted in mares with ascending or hematogenous placentitis does not fit the pattern noted with nocardioform placentitis. Mares with nocardioform placentitis classically show a loss of chorionic microvilli in a focal area around the base of the uterine horns. It has been postulated that the pathogen is present or introduced in the mare at the time of breeding and settles in the ventral aspect of the uterus, where it causes the infection. This hypothesis is not proven, though it is very likely that the nocardioform organisms are from the environment. Various nocardioform organisms have been reported in the soil in countries across the globe, although the pathogenicity of most of these organisms is not determined. The pathogenesis of nocardioform placentitis is still unresolved. Nocardioform organisms isolated from equine placentas include Amycolatopsis, species and Crossiella equi.,220-223 Nocardioform placentitis often results in abortion or premature delivery.210
Clinical Signs and Diagnosis
Mares that abort because of placentitis often show clinical signs of pending abortion before the actual pregnancy termination. Premature udder development and vaginal discharge are common signs of pending abortion caused by placentitis. Transrectal ultrasonography of the allantochorion in an area close to the cervix is useful to detect early signs of placentitis and impending abortion (Fig. 43-8).224 Ultrasound evaluations may reveal hyperechoic fetal fluids, placental separation, increased or decreased fetal heart rates (normal range 75 ± 7 bpm225), and thickening of the combined thickness of the uterus and placenta (CTUP). Consistently low or high fetal heart rates are associated with fetal stress. Foals experiencing fetal distress often become bradycardic initially and then become tachycardic in the terminal phase of life.226,227 Serial examinations should be performed to verify fetal well-being or distress. Mares considered “at risk” for pregnancy loss are often examined on a daily basis. Fetuses experiencing distress are often evaluated several times a day. Normal measurements of CTUP have been established (Fig. 43-9).228,229 Mares with placentitis may show increased CTUP, edema of the allantochorion, and separation from the endometrium. A 5-MHz linear transducer should be used for transrectal ultrasonography. The transducer should be positioned 1 to 2 inches cranial to the cervical-placental junction and then moved laterally until a major uterine vessel (possibly the middle branch of the uterine artery) is visible at the ventral aspect of the uterine body.228 The CTUP should then be measured between the vessel and the allantoic fluid (see Fig. 43-5). It is important to obtain all CTUP measurements from the ventral aspect of the uterine body, because physiologic edema of the dorsal aspect of the allantochorion has been noted in normal pregnant mares during the last month of gestation.228 In addition, care should be exercised to be certain that the amniotic membrane is not adjacent to the allantochorion, because this may result in a falsely increased CTUP. To examine the placenta from a transabdominal approach, a 3.5-MHz sector transducer is used most frequently. A 5-MHz linear transducer can be used for transabdominal evaluation; however, depth penetration can limit evaluation of the areas such as the placenta. All four quadrants of the placenta should be examined; right cranial, right caudal, left cranial, and left caudal. Measurements of the CTUP can be made with this technique if the fetus is not in close apposition with the uterine wall. Mares with normal pregnancies should have a minimum combined thickness of the uterus and the placenta (CTUP) of 7.1 ± 1.6 mm, and a maximal CTUP of 11.5 ± 2.4 mm.230 Pregnancies with an increased CTUP have been associated with the delivery of abnormal foals.230
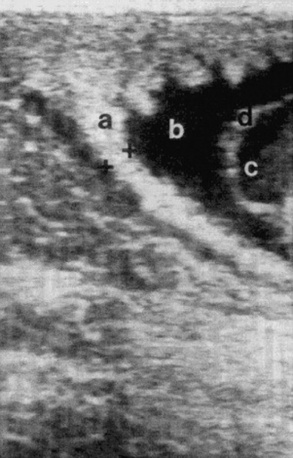
Fig. 43-8 Transrectal imaging of the combined thickness of the uterus and the placenta (CTUP). Measurements of the CTUP (distance between + and +) were recorded from the ventral part of the uterine body, close to the cervix. a, Placenta adjacent to the cervix; b, allantoic fluid; c, amniotic fluid; d, the amnion.
Modified from Renaudin CD, Troedsson MH, Gillis CL, et al: Ultrasonographic evaluation of the equine placenta by transrectal and transabdominal approach in the normal pregnant mare, Theriogenology, 47:559, 1997.
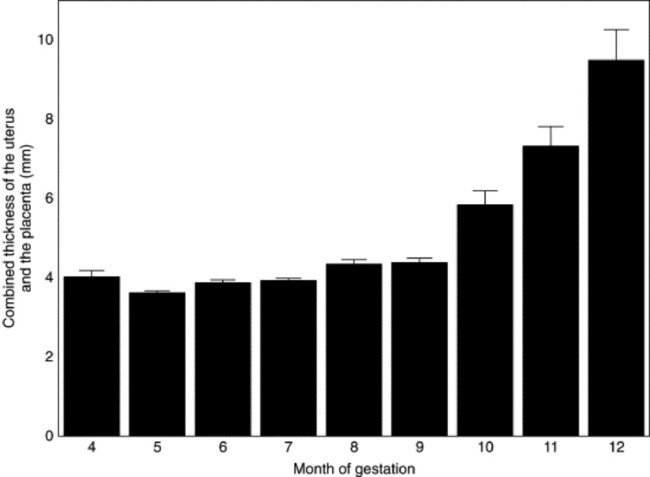
Fig. 43-9 Monthly recordings of transrectal ultrasonographic measurements of the combined thickness of the uterus and the placenta (CTUP) in normal mares from 4 months of gestation and throughout the pregnancy. Month 4 is 91 to 120 days; month 5 is 121 to 150 days; month 6 is 151 to 180 days; month 7 is 181 to 210 days; month 8 is 211 to 240 days; month 9 is 241 to 270 days; month 10 is 271 to 300 days; month 11 is 301 to 330 days; month 12 is 331 to 360 days.
Modified from Renaudin CD, Troedsson MH, Gillis CL, et al: Ultrasonographic evaluation of the equine placenta by transrectal and transabdominal approach in the normal pregnant mare, Theriogenology, 47:559, 1997.
The gross lesions of the fetus are not specific. An increased amount of fluid in the thoracic and abdominal cavities and an enlarged liver are frequently observed in aborted fetuses. Placental lesions are most severe on the chorionic surface at an area from opposite the cervix (“cervical star”) to the body of the placenta. The affected area is edematous, thickened, and discolored or brown with a mucoid or fibronecrotic exudate on the surface. The placenta is characteristically thickened and leathery in cases of mycotic placentitis, with lesions well demarcated from the rest of the chorionic surface. Microorganisms can be isolated from the placenta and several fetal organs, most consistently from the stomach.
Animals with nocardioform placentitis may have premature mammary development but will not have vulvar discharge, owing to the location of the infection. Placental lesions in mares infected with nocardioform actinomycetes are located ventrally in the uterine body and at the base of the uterine horns (Fig. 43-10). Affected areas are avillous, thickened, and covered by thick brown or reddish exudate. Because of the location of the lesions away from the cervical star, transabdominal ultrasonography is needed to diagnose the condition in pregnant mares. Differential diagnoses for this clinical presentation include twins and Cellulosimicrobium cellulans, placentitis.221

Fig. 43-10 Placenta from a mare with nocardioform placentitis. The characteristic lesions are located ventrally in the uterine body and base of the uterine horns.
Prevalence of fungal abortions varies greatly with region. The most common route of fungal infection is ascending via the cervix, and fungal infections may be seen with a mixed bacterial infection. Abortions are observed from mid to late gestation. Usually no clinical signs in the mare precede the abortion. The chorion generally shows marked evidence of placentitis with edema, thick plaques, and a thick, mucoid exudate. In rare cases, amniotic thickening may be noted. The aborted fetus is often small and emaciated owing to chronic placental insufficiency. Culture of the chorion may be diagnostic but takes time. Culture of the fetus is rarely rewarding. Impression smears of the chorion, and sometimes the fetal stomach, may reveal hyphae.
The equine placenta is part of an endocrine fetal-placental interaction that synthesizes and metabolizes progestagens.231 This endocrine function of the placenta is important for maintenance of pregnancy after the endometrial cups and the secondary corpora lutea disappear at approximately day 120 to 150 of gestation. Fetal-placental progesterone is rapidly metabolized to 5α-pregnanes. Mares with placental pathology may have increased plasma concentrations of progestagens as a result of stress to the fetal placental unit.232,233 Unfortunately, 5α-pregnanes are not readily assayed in a commercial setting, so diagnosis of placental disease using 5α-pregnane concentrations is not possible. There is cross-reactivity between 5α-pregnanes and progesterone using some commercial radioimmunoassays for progesterone. In recent studies233,234 using an experimental model to induce placentitis, it was found that mares that develop a chronic form of placentitis responded with increased plasma progesterone concentrations. Conversely, mares that developed acute placentitis and abortion soon after infection experienced a rapid drop in plasma progesterone concentrations. It was suggested that measurement of repeated samples of plasma progestin concentrations in mares with placentitis might be a useful method to identify mares that may abort or deliver prematurely.233 Furthermore, sensitivity of progesterone assays can be improved when they are combined with evidence of placental thickening as detected using transrectal ultrasonography.234
RELAXIN
Relaxin is produced by the equine placenta and can be detected in peripheral blood plasma from day 80 of gestation and throughout the pregnancy. The role of relaxin during pregnancy is not fully understood, but there is some evidence that placental relaxin production is compromised in mares at risk of aborting their fetuses.235 There is currently not a commercial test available for equine relaxin.
Treatment and Prognosis
Treatment efforts should be directed at combating infection, reducing inflammation, and controlling myometrial activity. Pregnant mares with clinical signs of placentitis should be treated with systemic broad-spectrum antimicrobials and antiinflammatories. Recent studies have demonstrated that commonly used antibiotics such as penicillin G (22,000 IU/kg q6h), gentamicin (6.6 mg/kg q24h), and trimethoprim-sulfadiazine (30 mg/kg bid) cross the placenta and reach therapeutic concentrations in both placental tissues and the allantoic fluid.236 Flunixin meglumine (1 mg/kg q12h) and pentoxifylline (8.5 mg/kg bid) have been used to control proinflammatory cytokines associated with placentitis. Treatment with progestins has long been advocated to promote uterine quiescence in mares with uterine pathology. Presumably the antiprostaglandin effect of progestins contributes to reduced myometrial activity by interfering with upregulation of prostaglandin and oxytocin receptors.237 Without receptor formation, gap junction formation would be inhibited and uterine contractility prevented. Altrenogest at twice the recommended dose (0.088 mg/kg PO q24h) is commonly used.
Work from a large scale clinical trial examined the efficacy of multipronged, long-term therapy for equine placentitis.238 Investigators examined records of 477 mares over 6 years. Fifteen mares were diagnosed with placentitis. Criteria for treatment included increased thickness of the uteroplacental unit using transrectal ultrasound, placental separation, and/or vulvar discharge and udder development. The average gestational age at diagnosis was 8.6 months. Mares were treated with a combination of systemic antibiotics (trimethoprim-sulfa, ceftiofur, or penicillin and gentimicin), pentoxifylline, altrenogest, and nonsteroidal antiinflammatory agents. Mares were treated until abortion or delivery of a foal. Twelve of 15 treated mares (80%) carried their foals to term, and 11 of 15 (73%) delivered live foals. Birthweights of surviving foals from mares treated for placentitis were similar to those of foals from nonaffected mares. The benefit of this treatment strategy is supported by recent data from a controlled study on experimentally induced placentitis.238 Data from these studies suggest that long-term antibiotic, antiinflammatory, and progestin treatment may positively affect pregnancy outcome in mares with placentitis.
Although most mares are capable of conceiving and successfully carrying a foal to term in subsequent breedings, reproductive performance may be negatively affected after ascending and hematogenous placentitis. Treatments for endometritis, such as uterine lavage and intrauterine infusions of appropriate antibiotics, should also be implemented after abortion. Most mares affected by nocardioform placentitis do not require subsequent treatment and show no signs of infertility the following breeding season.
Equine Herpesvirus 1 Abortion
The most prevalent viral cause of equine pregnancy loss is EHV-1, which causes abortion, paresis, and neonatal foal death. EHV-4 causes abortion in rare cases. The primary route of transmission of EHV-I is via the respiratory tract. The virus invades the respiratory epithelium and establishes a leukocyte-associated viremia. EHV-1 establishes a chronic, possibly lifelong, latent infection. During the initial infection, placental endothelial cells are infected by the virus and transiently present targets to the immune system as the cells present viral particles. The virus eventually inactivates the major histocompatibility complex 1 (MHC-1) ability of the cells to present viral particles, thus evading the immune system.239 After respiratory infection, EHV-1 causes an episode of viremia and infects the fetus via transplacental migration of virus-bearing leukocytes. Respiratory clinical signs in infected mares may be subclinical. The time between infection and abortion varies greatly from less than 2 weeks to several months.240 Abortion occurs as a result of a rapid separation of the placenta, causing suffocation of the fetus.241 Near-term fetuses may be born alive but will die within days. Aborting mares clear the virus quickly from the reproductive tract, and subsequent fertility is often not affected by the disease. Clinical signs and fetal lesions of abortion caused by EHV-1 and those caused by EHV-4 are indistinguishable from each other.240
As with all herpesviruses, EHV-1 establishes a latent infection that may recrudesce after stressful events, such as weaning, translocation, introduction of a new animal, or other illnesses. It is unclear whether or not a reactivation event will cause an abortion, but this is considered likely.242 Recrudescent infections are certainly transmissible. Aborted, infected fetal materials are also highly contagious. Both of these sources may be responsible for abortion epizootics. Although epidemic abortions occur, losses may be confined to only a few mares in a herd.
Clinical Signs and Diagnosis
The primary lesion of EHV-1 is necrotizing vasculitis and thrombosis resulting from lytic infection of the capillary endothelium. The fetus may become infected or remain uninfected, depending on whether or not the virus crosses the uteroplacental barrier. If transplacental fetal infection occurs late in gestation, a live, infected foal may be born but will not usually survive. Stage of pregnancy during which abortions may occur varies, but the vasculitis is most pronounced from the fifth to ninth months of gestation, and 95% of abortions occur in the last trimester of pregnancy.243
Abortions occur suddenly without maternal clinical signs. The aborted fetus is fresh with minimal signs of autolysis. Increased fluid in the thoracic and abdominal cavities; congestion and edema of the lungs; an enlarged liver with small (approximately 1 mm) necrotic, yellow-white lesions; subcutaneous edema; and icterus are commonly found gross lesions in the fetus. Samples for histopathologic diagnosis should be submitted in Bouin’s solution. Histologically, the most characteristic lesion consists of areas of necrosis in lymphoid tissue, liver, adrenal cortex, and the lung, with large intranuclear eosinophilic inclusion bodies. In addition, a hyperplastic necrotizing bronchiolitis is often found. Lymphoid tissues are most commonly affected (nodes, thymus, spleen, and Peyer’s patches). Other histopathologic lesions may include mild, multifocal, necrotizing lesions in the liver and adrenal cortex and a hyperplastic, necrotizing bronchiolitis. The placenta may be grossly normal or edematous with no specific microscopic lesions.
Available diagnostic methods include serologic tests, virus isolation, and polymerase chain reaction (PCR). Serologic tests are not considered reliable in EHV-1 diagnosis, and virus isolation remains valuable as a method that allows classification and comparative evaluations. Laboratory diagnostics include fluorescent antibody (FA) staining of fetal tissue, virus isolation from aborted fetuses, virus isolation from maternal whole blood, presence of viral inclusion bodies in liver, lung, and thymus, and fetal serology. Equine fetuses have been found to be capable of producing antibodies to EHV-1 at 200 days of gestation. Maternal serology is of limited diagnostic value because mares may abort several weeks after infection. The rise in serologic titer may have disappeared by the time of the abortion.
Treatment and Prognosis
Studies offer conflicting results as to the efficacy of vaccinations in reducing viremia and abortions. Many studies do suggest a beneficial effect of vaccination, and the current recommendation is to vaccinate during the fifth, seventh, and ninth months of gestation.239,243 Both killed and modified live vaccines are available. The vaccines are not fully protective, and abortion may occur in vaccinated mares. However, consistent vaccination of pregnant mares should be expected to decrease the incidence of abortion storms and sporadic abortions in a herd.
For the effectiveness of a vaccination program to be maximized, it needs to be combined with a management strategy that minimizes exposure of mares to the virus and prevents activation of a latent viral infection. All horses, young, adult, nonpregnant, and pregnant, should be vaccinated to restrict shedding of the virus. Unnecessary stress such as transportation and overcrowding should be avoided. Pregnant mares should be kept separate from other horses on the farm. Newly arrived horses should be isolated from the resident population for 3 weeks, during which time they should be monitored daily for signs of respiratory disease.
After abortion, the fetus and fetal membranes should be transported away from the area without contaminating the surrounding environment. The stall in which the mare aborted should be disinfected with a phenolic or iodinophoric compound, and the bedding should be prevented from contaminating other areas on the farm. All pregnant mares on an infected farm should remain on the farm until they have foaled. No horse should leave the farm until 3 to 4 weeks after the last abortion.
Equine Viral Arteritis
Equine viral arteritis (EVA) is caused by equine arteritis virus (EAV). The primary target of EAV is the vasculature. Shortly after infection EAV can be found in the macrophages and later in the lymph nodes. The virus infects circulating monocytes and becomes systemic in distribution by 3 days after the primary infection, resulting in a carrier state (important in stallions). In about a week’s time after the primary infection EAV infects the blood vessel endothelium and causes enough damage by 10 days after the primary infection to cause abortion. Abortion is likely due to the effects of myometritis and vasculitis. Serum progesterone concentrations (produced exclusively by the placenta in the last half of equine pregnancy) fall to baseline levels before the abortion, because of placental hypoxia. The virus is also present in the renal tubular epithelium and is shed in the urine.244
EVA is caused by a pestivirus. Infection is often inapparent, and abortion is an occasional occurrence in infected animals.245 Although the pathophysiology is not well established, fetal death may occur by fetal anoxia secondary to compression of myometrial vessels by edema and decreased progesterone production by the placenta.246
History and Clinical Signs
Clinical signs may be absent or highly variable and may include pyrexia, depression, anorexia, leukopenia, limb edema, stiffness of gait, rhinorrhea and epiphora, conjunctivitis, rhinitis, urticarial rash, localized or diffuse edema, and abortion. Abortion typically occurs at 5 to 10 months of gestation and follows the onset of clinical signs by several days but up to 2 months. Less frequently, severe respiratory distress, ataxia, mucosal papular eruptions, submaxillary lymphadenopathy, and intermandibular and shoulder edema may be observed.244 Infection is rarely fatal in adults but is more frequently fatal in neonates. The virus may be transmitted in the semen, and infected stallions may serve as long-term carriers. Clinical pathologic findings are variable, inconsistent, and nonspecific and include hypoxia, hypercapnia, respiratory or metabolic acidosis, lymphocytosis or lymphopenia, neutrophilia or neutropenia, thrombocytopenia, and hyperfibrinogenemia. Gross and histologic lesions differ in severity with the virulence of the particular viral strain. Edema, congestion, and hemorrhage of the subcutaneous and lymphoid tissues and viscera are the most common gross lesions. Histologic lesions may be found in the vasculature, lymphoid tissues, lungs, intestines, adrenal glands, kidneys, and skin.244 Abortion may occur without any clinical signs in the mare, and the fetus may be fresh or autolyzed. Fetal lesions are uncommon.
Laboratory Diagnosis
Diagnosis is by serologic testing in conjunction with complement-dependent virus neutralization. There are no characteristic features of EVA infection in the fetus, although autolysis and myocardial arteritis have been reported. EAV is readily isolated or detected by PCR techniques in fetal tissues and the placenta.
Epidemiology and Control
Transmission may occur venereally from infected stallions to mares. Reproductive performance of venereally infected mares is not affected, but contact transmission from venereally infected mares to late gestational mares may cause abortion. Outbreaks were reported in 1953, 1984, and 2006 in the United States. With the exception of these incidents, occasional outbreaks have occurred, with abortion occurring at low incidence.245 Mares may be isolated after infection but usually do not become carriers. A proportion of naturally infected stallions become persistently infected with EAV and shed the virus constantly in semen. Incidence of seropositive animals is higher in standardbreds than in thoroughbreds, and regulatory guidelines may govern the use of seropositive thoroughbred stallions. Carrier stallions may be the reservoir for the disease between outbreaks and should be isolated and bred only to immune mares. Mares bred in this way should be isolated for 3 weeks. A modified live vaccine is available for mares and stallions. Control involves vaccination of seronegative stallions under the guidance of regulatory authorities. There is some evidence that prepubertal infection of colts does not result in permanent carrier status.
Equine Infectious Anemia Abortion
Equine infectious anemia (EIA) is a retroviral infection transmitted by horseflies.247 After systemic infection with EIA, mares abort during the febrile stage of infection and may abort at any stage of gestation. Foals from infected but asymptomatic mares and stallions are seronegative (precolostral) and clinically normal at birth. Mechanism of abortion is unknown but may be secondary to systemic illness because EIA virus is not found in amniotic fluid.247 Coggins’ test will confirm seropositive status but is not a definitive diagnosis for abortion.
Leptospirosis Abortion
Leptospirosis causes a number of bacterial abortions in some areas and was reported to be responsible for 2.2% of abortions in a Kentucky study.210 The serovar responsible shows regional variability. Serovar Leptospira, Pomona has most commonly been associated with leptospirosis abortion in mares, but Leptospira, Grippotyphosa, Leptospira, Hardjo, Leptospira, Bratislava, and Leptospira, Icterohemorrhagiae have also been isolated from sporadic abortions.248 Infected animals shed the spirochete in their urine, which contaminates groundwater and serves as a source for further infections. Many horses are seropositive for leptospires but are subclinical.
Clinical Signs and Diagnosis
Clinical signs may include pyrexia, hemoglobinuria, jaundice, and abortion. Aborted chorioallantoic lesions are similar to those of other bacterial placentitis cases (nodular cystic allantoic masses, edema, necrotic areas of chorion, and necrotic mucoid exudate coating the chorion) but may show a diffuse pattern of distribution, indicating a hematogenous source of placental infection. Funisitis has been reported in leptospira cases of abortion, and examination, both gross and histologic, of the umbilical cord should always be a part of every abortion investigation.249 Serologic antibody conversion lasts for years, and so serologic diagnoses are not very helpful. Gross lesions are nonspecific. Diagnosis is best done by FA tests or Warthin-Starry silver stain (WS) staining of the allantochorion and umbilical cord. Demonstration of leptospirosis could aid in rapid diagnosis and have important clinical and therapeutic indications in the case of live-born, weak foals.249
Treatment and Control
Horses may shed spirochetes in urine for up to 90 days; therefore affected animals should be isolated and treated with antibiotics. Aborting mares should be isolated, and the stalls should be disinfected. Infected mares may be treated with streptomycin (10 mg/kg twice daily), penicillin (10 to 15,000 IU/kg twice daily), or oxytetracycline (5 to 10 mg/kg) for a period of 1 week. Because Leptospira, Pomona is the most common isolate in the United States, mares should be separated from other leptospiral hosts such as ruminants and pigs. Vaccines for cattle are not effective in horses.
Protozoal Abortion
Pregnant mares with equine monocytic ehrlichiosis (Potomac horse fever), caused by the protozoa Neorickettsia risticii, may abort. Abortions caused by N. risticii, have been documented in both natural and experimental cases.250,251 Mares were infected at 90 to 180 days of gestation and aborted at around 217 days of gestation.
Clinical Signs and Diagnosis
Abortions have been observed 2 to 3 months after clinical signs of ehrlichiosis.252 Abortions were associated with placentitis and RFMs. Fetal histologic lesions include enterocolitis, periportal hepatitis, myocarditis, and lymphoid hyperplasia with necrosis of the mesenteric lymph nodes and spleen. Recovery of the protozoan may be from fetal bone marrow, spleen, lymph node, colon, or liver.250,251 The diagnosis can be confirmed by identifying a small number of rickettsiae by PCR assay.
Treatment and Prevention
Treatment with oxytetracycline (6.6 mg/kg IV once daily) for 5 days in pregnant mares with clinical signs of ehrlichial colitis may prevent or reduce the incidence of abortion. Commercial vaccines against equine ehrlichiosis are available, but the protective effect of vaccines against abortion is unknown.
Neospora caninum, is a known protozoal abortifacient in cattle. This protozoan has not been definitively shown to cause abortion in the mare, but its presence has been documented in aborted foals, suggesting the need for further investigation.253
Insect-Related Abortion
In the equine breeding season of 2001, central Kentucky horses experienced pregnancy losses estimated to have affected more than 3000 mares (>60% of mares on some farms254) and to have resulted in over $330 million in losses.255 The syndrome was named mare reproductive loss syndrome, (MRLS). On a much smaller scale, MRLS was documented in mare abortions in north central Florida during the 2006 breeding season. Epidemiologic studies noted abnormal weather patterns (sudden freezing in mid April followed by unusually warm springtime temperatures) and positive correlations with the presence of black cherry trees (Prunus serotina), and abnormally large numbers of the eastern tent caterpillar (Malacosoma americanum).,256 Pregnancy loss was later linked to ingestion of the caterpillars themselves, with the toxic agent related to the larval exoskeleton.
Clinical Signs and Diagnosis
Clinical signs preceding abortion and failure to maintain pregnancy are typically not observed. Both early (40 to 150 days’ gestation) and late (near term) pregnancies are affected. Hyperechoic amniotic and allantoic fluids can be noted on ultrasonographic examination, along with a dead or dying fetus (slow heart rate, <75 beats/min), which would then be expelled within several days.257,258 Characteristic histologic lesions are noted in the placenta and umbilical cord. In many cases, endometrial cultures are positive for non—beta-hemolytic streptococci and actinobacilli. Actinobacillus, species identified in cases of MRLS are identical to commensal organisms found in the oral cavities and alimentary tracts of healthy horses.259 Larval hairs (cetae) are hypothesized to migrate through gastrointestinal system and translocate commensal organisms from the oral cavity and intestines to other sites.256
Treatment and Prevention
Because clinical signs are absent in the mare before fetal death, treatment is not possible. Prevention is aimed at limiting exposure to M. americanum, larvae. Applying insecticides, physically removing caterpillar nests from trees, removing black cherry trees, muzzling mares in pasture, and supplementing hay feeding in pastures to minimize grazing have all been used.
Salmonella, Abortion
Salmonella, Abortus equi (Salmonella abortivoequina, or “contagious abortion”) was a common cause of abortion in the early 1900s but is now rare. Non—host-specific species, including Salmonella, Typhimurium, now cause most equine salmonella abortions. Abortion caused by Salmonella, is discussed under abortions in ruminants.
Trypanosomiasis (Dourine)
The protozoan parasite Trypanosoma equiperdum, causes a venereally transmitted genital infection that may be followed by fatal systemic dissemination in horses. Systemic illness may cause abortion. It occurs in tropical and subtropical regions and has been eradicated from North America.11 Dourine is diagnosed by isolation of trypanosomes in uterine discharge. Exposed animals can be detected by serology (complement fixation). Control strategies require identification and treatment or slaughter of infected animals.
Abortion Caused by Endotoxemia
Gram-negative septicemia and endotoxemia associated with intestinal disorders that alter the integrity of the mucosal barrier (e.g., intestinal obstructions, acute enteritis, colitis, grain overload) result in the release of vasoactive metabolites including PGF. Endogenous release of PGF during an episode of experimental endotoxemia has been shown to cause luteolysis and abortion during the first 2 months of pregnancy in mares.260 The equine pregnancy is dependent on ovarian sources of progesterone for the first 80 days of gestation. After this time the fetoplacental unit takes over progesterone production, which is necessary for maintenance of the pregnancy.
Clinical Signs and Diagnosis
Abortions follow a recent episode of stress induced by endotoxemic shock or gram-negative endotoxemia. Pregnancy loss at early stages of gestation may go undetected unless fetal membranes or parts are found in the stall. Abortions during later stages of the pregnancy may be observed as vaginal discharge or the detection of an expelled fetus.
Treatment and Prevention
Daily administration of a progestagen (Altrenogest 0.044 mg/kg PO) has been shown to effectively prevent experimental endotoxin-induced abortion.260 If the animal is treated while the pregnancy is still CL dependent (approximately <day 80), analysis of serum progesterone concentrations after the acute episode of the disease helps in deciding if the supplementation needs to continue. Serum progesterone concentrations of less than 1 ng/mL indicate the loss of an active CL, and supplemental progestagen treatment should continue until the fetoplacental unit is known to be capable of maintaining the pregnancy. For practical reasons supplementation until day 100 is commonly recommended. Serum progesterone concentrations greater than 1 ng/mL are compatible with a functional CL, and the progestagen treatment can gradually be discontinued. Treatments with flunixin meglumine or other prostaglandin inhibitors have not been proven to effectively prevent endotoxin-induced fetal losses unless the agents are administered before clinical signs appear.
RUMINANTS
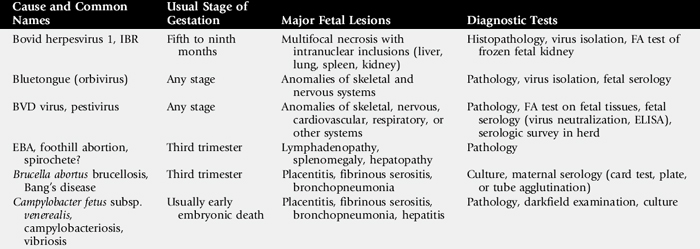
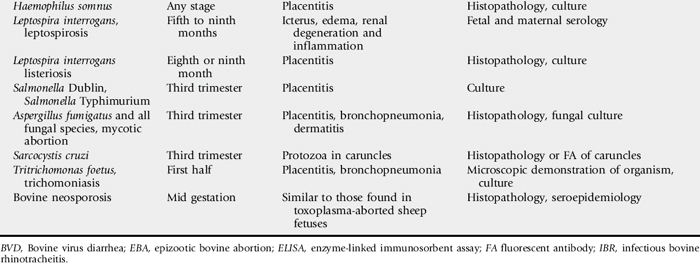
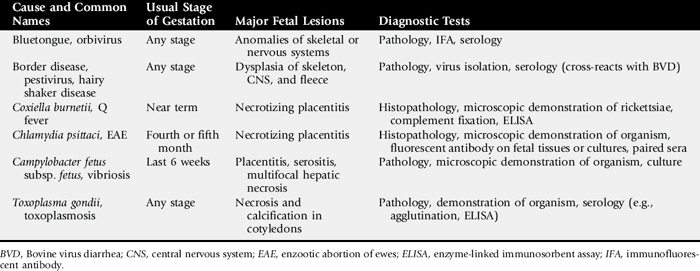
Information on the major causes of abortion in cows, sheep, and goats is summarized in Tables 43-8 and 43-9.
In large commercial operations a low percentage of intermittently occurring abortions is considered acceptable; such abortions are seen as “background abortions.” In beef and dairy operations 2% to 3% and 10% are considered acceptable, respectively. Abortion levels beyond these are considered abnormal and are usually investigated. Only rarely is a cause established. When investigating bovine abortions, it is important to determine commonalities among abortion events including stage of pregnancy affected, sire used, heifers versus cows affected, dates of new arrivals to the herd, season of year, vaccination protocols, presence of dogs, and management practices among different groups. Cows and heifers on the farm presumed to be pregnant should be evaluated to detect unobserved pregnancy losses. These examinations should document apparently normal, open animals who had previously been documented as pregnant, those with mummified fetuses, and those with pyometra. If possible an entire fetus (or more than one) should be submitted for culture, serology, and histopathology. If the placenta is available, this should be submitted as well. If it is not practical to submit the entire animal and placenta, tissues including lung, stomach and contents, liver, spleen, and placenta should be collected and appropriately submitted for culture (chilled), viral isolation (frozen), and histopathology (formalin fixed).
INFECTIOUS CAUSES
Bovine Herpesvirus 1
Bovine herpesvirus 1 causes IPV and IBR. The latter disease is the major cause of bovine viral abortion. IBR manifests as upper respiratory disease and abortion. The virus causes clinical signs in both dam and fetus. Immune dams exposed to the virus do not experience abortions. In naive animals the virus is carried in leukocytes and can become localized in placental tissues. Fetuses exposed to the virus usually die within 24 hours of placental infiltration. Although other stages of pregnancy may be affected, 5 to 6 months’ gestation is the most susceptible stage. Fetal lesions include renal hemorrhagic edema; acute general necrosis in the liver, spleen, kidneys, lungs, and adrenal glands; widespread hemorrhage; petechia; epithelial destruction; and hepatic necrosis with intranuclear inclusion bodies. The amnion may be thickened without obvious signs of inflammation.
Diagnosis of IBR is by virus isolation from the placenta, identification of intranuclear inclusion bodies in fetal tissues, or serum neutralization testing. If the latter is to be used, at least two samples must be taken days apart. The first must be negative and the second positive, or the first must be positive and the second titer must show a fourfold increase. Serum testing may not be helpful because the dam may have been infected months before the abortion and the titer after the abortion may actually be falling.
Prevention of disease is by vaccination (though infection and latency may occur despite vaccination). It is recommended to vaccinate heifers at 6 months and administer a booster 3 to 4 weeks before breeding. A modified live virus is commonly used but is unsafe in pregnant cows from the third to eighth months of gestation. A killed viral vaccine is available for pregnant cow use; a modified live virus vaccine labeled for intranasal administration is also available.261
Bovine Virus Diarrhea Virus
Bovine virus diarrhea virus (BVDV) is a pestivirus that can produce early embryonic death, fetal anomalies, or abortion (see Chapter 32 for complete discussion). Isolates from bovine aborted fetuses are usually noncytopathic.262
History and Clinical Signs
Although fetal death is most common during the first trimester, abortion can occur at any stage of gestation.263,264 Pathogenicity of the disease depends on gestational time of infection, the viral strain, viral biotype (cytopathic or noncytopathic), and fetal immunocompetence.265 Often there is a history of repeat breeding and a recent episode of febrile disease in the herd before the onset of abortions.266
Laboratory Diagnosis
Fetal loss generally occurs 10 to 27 days postexposure, with expulsion of the fetus up to 50 days later.265 As a result the fetus is most often autolyzed, although in some cases it may be mummified or fresh. The aborted fetus may have a variety of dysplastic lesions, including cerebellar hypoplasia, cerebral malformations (hydrencephaly, porencephaly, microencephaly) and cataracts, brachygnathia, arthrogryposis, alopecia, thymic hypoplasia, and intrauterine growth restriction.5,263,266,267 Microscopic lesions include a mild nonsuppurative placentitis. Nonsuppurative vasculitis may be observed in the placenta, liver, or lymph nodes.266
Virus isolation from fetal tissue is seldom successful, likely because of the protracted time before fetal expulsion generally occurs after infection. Viral antigen may be detected by FA test on kidney, lung, or lymph node.266 Virus neutralization and enzyme-linked immunosorbent assay are used to detect antibodies in fetal thoracic fluid, which indicate prenatal exposure to the virus but do not necessarily incriminate BVDV as the cause of abortion. Maternal titers are seldom of diagnostic value because a rise in titer generally occurs before abortion.5
Pathophysiology
BVDV may be shed in most body secretions. The effects of exposure to BVDV vary greatly depending on the gestational time during which exposure occurred (Fig. 43-11). In seronegative cows, exposure to BVDV at the time of breeding prevents conception.268 Placental attachment at approximately 35 days’ gestation seemingly must precede fetal infection. During the first 4 months of gestation, infection usually causes fetal death and abortion.166 Fetuses that survive infection with noncytopathic strains between 18 and 125 days’ gestation will be persistently infected (PI), are typically seronegative at birth, and subsequently shed BVDV continuously.265 They may develop mucosal disease later in life from superinfection with cytopathic BVD virus.269,270 Fetuses infected at between 100 and 150 days’ gestation, considered congenitally infected (CI), are at risk for the development of dysplastic lesions including teratologic defects in the brain, skin, or bronchioles. Fetuses infected after 150 days usually recover without dysplastic lesions.263 Recent evidence, however, suggests that those fetuses born with neutralizing titers to BVDV are more likely to develop a serious illness within the first 10 months of life and less likely to conceive as heifers than those calves born without neutralizing titers to BVDV.271,272
Fig. 43-11 Potential clinical reproductive outcomes after infection with bovine virus diarrhea virus. EED, Early embryonic death.265
Epidemiology
Most cattle have serum antibodies to the virus. The abortion rate may approach 25% with new infection of a susceptible herd.5
Leptospirosis
Leptospirosis is a spectrum of diseases caused by multiple serovars of Leptospira interrogans. Leptospira, Hardjo is the major serovar associated with bovine leptospiral abortion, although isolations of Leptospira, Pomona, Leptospira, Canicola, Leptospira, Icterohemorrhagiae, Leptospira, Grippotyphosa, and Leptospira, Szwajizak have also been reported.5,266 Two serologically indistinguishable but genetically distinct types of serovar Hardjo have been identified: L. interrogans, serovar Hardjo (type hardjo-prajitno) and Leptospira borgpetersenii, serovar Hardjo (type hardjo-bovis). Serovar Hardjo type hardjo-bovis is cosmopolitan in cattle populations, whereas type hardjo-prajitno is isolated primarily from cattle in the United Kingdom.265
History and Clinical Signs
In cattle, Leptospira, Hardjo is associated with infertility, early embryonic death, abortions from 4 months’ gestation to term, and birth of weak calves. Abortion rate is usually less than 10% but may approach 50% in some areas.273Leptospira, Pomona abortion usually occurs in the last 3 months of gestation, with an abortion rate as high as 50%.5 Clinical signs of leptospirosis in the cow may include icterus, hemoglobinuria, anemia, fever, and mastitis that is characterized by a flaccid udder and thick ropy secretions from all four quarters, but usually cows abort without clinical illness.266 Dead or weak calves may be delivered at term.273 Abortions caused by serovar Hardjo tend to occur sporadically rather than in storms (as may be seen with serovars Pomona or Grippotyphosa).
Laboratory Diagnosis
The aborted fetus is usually autolyzed, icteric, and edematous. Histologically, renal tubular necrosis is accompanied by lymphocytic interstitial nephritis, pneumonia, and placentitis.5,266
Leptospires are rapidly destroyed by autolysis or freezing. Isolation from fetal liver, kidney, or brain is possible but slow and impractical. Leptospires may be isolated or demonstrated by darkfield microscopy, FA staining, or histologic techniques in fetal or placental tissues or in the urine of the aborting dam within approximately 2 weeks after abortion.5,266
Diagnosis is usually based on serology. A few infected fetuses develop microagglutination titers of 1:10 or more. It is difficult to distinguish among vaccinated, acutely infected, and recovered animals, but titers to Leptospira, Pomona greater than 1:12,800 in the dam suggest leptospiral abortion. Maternal titer usually has peaked by the time of abortion. Single titers of 1:800 or more in unvaccinated animals, seroconversion, or fourfold changes in titers in paired sera indicate leptospirosis in the herd. Titers to Leptospira, Hardjo are often less than 1:100 in affected cows and seldom exceed 1:1600.266
Pathophysiology
Hematogenously spread leptospires colonize the gravid uterus up to 142 days after infection. Abortion occurs 1 to 6 weeks after acute disease with Leptospira, Pomona infection and 1 to 3 months with Leptospira, Hardjo.5Leptospira, Hardjo remains in the oviducts of infected cows up to 22 days after calving.273
Epidemiology
Leptospira, organisms are ubiquitous and an important cause of abortion in all cattle-producing regions. Leptospira, organisms persist in the genital tract and kidneys. The organisms localized in the kidneys of infected animals are shed in the urine and serve as a source of infection for other animals. The organisms localized in the female reproductive tract are responsible for abortions. Aborted tissues are infectious to other animals and humans and should be handled with caution.274 Vaccination may be useful in endemic areas.
Treatment and Control
In abortion outbreaks, pregnant cows can be vaccinated with killed bacterin and treated with oxytetracycline (antibiotic treatment can be limited to sick cows in dairy herds).275 Aborting cows should be isolated and treated with streptomycin if they are not destined for slaughter. Aborted fetuses and placentas should be removed from the premises.276 Preventing exposure to swine, rodents, and contaminated water lessens the opportunities for infection.5,202Leptospira, Pomona usually has no permanent effect on fertility, but infection with Leptospira, Hardjo has been associated with persistent herd infection and recurring abortions.5
Herd vaccination is recommended at 6-month intervals or more frequently in areas with heavy exposure to leptospires. Vaccination programs are aimed at reducing urinary shedding of leptospires and decreasing fetal loss. However, a commercial pentavalent leptospiral vaccine (serovar Leptospira, Hardjo type hardjo-prajitno is used for the Leptospira, Hardjo component of U.S. Department of Agriculture [USDA]—licensed leptospiral vaccines) did not prevent renal colonization, urinary shedding, or fetal infection after conjunctival instillation of cows with serovar Leptospira, Hardjo type hardjo-bovis (the only type of Leptospira, Hardjo isolated from cattle in the United States).277
Epizootic Bovine Abortion (Foothill Abortion)
Epizootic bovine abortion (EBA) or foothill abortion is a syndrome of late abortions in cattle in the foothills bordering the central valley of California.278 Once thought to be caused by C. psittaci, studies have demonstrated that EBA differs from chlamydial abortion.278 Currently a spirochete-like agent isolated from abortuses and from the tick vector of EBA is under investigation as the causative agent.279-281
History and Clinical Signs
Late abortion or delivery of weak calves occurs in affected herds. Many fetuses in the sixth to seventh month of gestation may be aborted, especially from heifers without premunition immunity from natural exposure to the causative agent. Older native cows show no clinical signs of infection.
Laboratory Diagnosis
A 3-month period is required for full development of pathologic changes in the fetus. Superficial cervical lymph nodes are enlarged up to 16 g, the spleen is enlarged up to 250 g, the thymus is slightly smaller than normal, and the liver may be enlarged and nodular.266,278,280 Histologically there is loss of thymic cortical lymphocytes; remaining lymphocytes are enlarged and poorly differentiated. Follicular hyperplasia, histiocytosis, vasculitis, necrosis, and pyogranulomas occur in lymph nodes and spleen. Lymphohistiocytic proliferation may also occur around vessels in the liver, lung, and meninges.266
EBA has been diagnosed mainly by pathologic examination of the fetus. Recent studies have demonstrated high levels of IgG (3 mg/mL or more) in fetal blood.266,278 A spirochete-like agent can be demonstrated in the plasma of aborted fetuses but also may occur in plasma of normal fetuses.279 Currently no serologic test for EBA exists because the cause is uncertain.
Pathophysiology
Infection is transmitted by the soft-shell tick Ornithodoros coriaceus.,280,281 The disease also can be transmitted with fresh or frozen fetal tissue.278 Transformation and proliferation of fetal lymphocytes and macrophages occur by 50 days but are not severe enough for diagnosis until 100 days after maternal exposure to the tick vector. IgG and IgM are deposited in vascular lesions, but increase in fetal serum immunoglobulin is not detectable until at least 80 days after tick exposure.278,281 Repeated superinfection may be necessary to result in fetal death.280 Because at least 90 days are required for development of fetal lesions, infection after 6 months’ gestation is not likely to result in abortion.5,266
Epidemiology
EBA is limited to the range of the tick vector in the foothills bordering the central valley of California.278 Of the annual calf loss in California, 5% to 10% is attributed to EBA.281 The prevalence of infection by the spirochete is far greater than the prevalence of abortion.280 Abortion occurs 3 to 4 months after exposure to ticks but almost always late in gestation, regardless of the time of tick exposure.282 Older native cows from enzootic areas usually do not abort, and introduced cows and heifers generally abort only once.278 The abortion rate may be 30% to 80% in susceptible animals.266,282
Treatment and Control
Chlortetracycline therapy (2 to 5 g/day in the feed) reduces the rate of abortion.281 Currently no vaccine exists for EBA, but abortions can be controlled by exposing heifers to the tick vector before breeding or by changing from spring to fall calving,5 which takes advantage of limiting exposure to the last trimester of gestation in some management systems. Therefore exposure of susceptible pregnant cattle to the tick only after the sixth month of pregnancy is a practical solution for ranchers using summer foothill pastures and fall calving. The tick lives in ground duff (e.g., leaves) and is not found on cattle that graze in irrigated pastures and most other areas outside of brushy foothills.
Brucella abortus, Abortion
B. abortus, infection (Bang’s disease) causes abortion in cattle and, less commonly, in sheep and goats. Horses may be infected with B. abortus, which has been associated with fistulous withers, but usually experience no infertility, abortion, or other clinical evidence of infection. Bovine infections are caused by eight biovars of B. abortus, three of which (biovars 1, 2, and 4) are recognized in the United States.5
History and Clinical Signs
Abortion is the chief clinical sign of bovine brucellosis and usually occurs after the fifth month of gestation. Lameness, mastitis, epididymitis, and/or orchitis may be present in infected herds.283
Laboratory Diagnosis
Autolysis frequently obscures gross lesions in the fetus, but fibrinous serositis may be apparent, and abomasal content may be discolored and flocculent. Placentitis is a consistent finding. Cotyledons are necrotic; the intercotyledonary placenta is thickened and opaque with accumulation of odorless, flocculent, yellow-brown exudate between maternal and fetal membranes. Histologically there is suppurative placentitis (and endometritis in the dam). Suppurative bronchopneumonia and lymphoreticular hyperplasia are frequent histologic findings in the fetus.
Diagnosis depends on culture of B. abortus, from fetal lung, abomasum, or placenta or from maternal uterine or mammary secretions. Organisms and Brucella, antigen can be detected in fetal tissues by avidin-biotin-peroxidase complex immunostaining.284
No serologic test is 100% accurate, but a positive card test or titer of 1:100 or more on plate or tube agglutination suggests brucellosis. False-negative serologic reactions occur in approximately 15% of infected cows, particularly just before or after parturition. False-positive reactions are a problem in cows vaccinated with strain 19. Supplemental tests such as the Rivanol test, complement fixation, and mercaptoethanol sensitivity of agglutination are used in suspect cases. Dairy herds are surveyed by the Brucella, ring test on milk.5 In goats, tube agglutination titers of 1:25 or more indicate infection.283
Pathophysiology
Initial replication of B. abortus, occurs in regional lymph nodes. Bacteremia is followed by colonization of supramammary lymph nodes, the mammary gland, and the gravid uterus. Uterine infection occurs during the second trimester.5,285 In the placenta the bacteria appear first in phagosomes of erythrophagocytic trophoblasts. Replication occurs in the rough endoplasmic reticulum of chorioallantoic trophoblasts.285 Preferential replication in chorioallantoic trophoblasts has been attributed to their erythritol content; however, the placentas of several laboratory rodents that lack detectable erythritol still support B. abortus, replication.286 The organism also occurs in fetal placental endothelial cells and capillary lumina, where it is associated with vasculitis and destruction of chorionic villi. Placental inflammation spreads along the allantochorion to involve additional cotyledons with resultant chorioallantoic ulceration, necrosis of trophoblasts, and ulcerative endometritis. Fetal death results from placental disruption and endotoxemia.5,285 The fetus is frequently retained 1 to 3 days in utero. Numerous bacteria are expelled from the genital tract at parturition, but shedding usually stops by 3 weeks after abortion.5
Epidemiology
Infection with B. abortus, occurs naturally by ingestion. Contaminated materials are infectious for humans and should be handled with caution. Infection is not easily transmitted between cattle separated by fences or roads.287 Most calves infected at birth clear the infection, but persistent congenital infection has been documented.288,289
Bovine brucellosis has been nearly eradicated in the United States by test and slaughter of seropositive cattle and vaccination. However, a reservoir remains in bison and elk, especially in the greater Yellowstone area.
Treatment and Control
Treatment of brucellosis is usually not effective. Combination therapy with long-acting oxytetracycline and streptomycin has been shown to reduce shedding in most cows and potentially eliminate infection in some,290 but because of the eradication program used in many countries, infected cows are rarely treated. In the United States, infected individuals are destroyed, and exposed herdmates are quarantined to the herd until slaughter or the herd is recertified as brucellosis-free (United States Department of Agriculture, 2003).
In the past, strain 19 vaccine was used as part of the eradication program. However, vaccinated and field strain—infected cattle could not be differentiated. A newer vaccine for B. abortus, vaccine strain RB51, was developed to overcome the serologic problems associated with the strain 19 vaccine.291 Animals vaccinated with RB51 lack antibodies to the O-polysaccharide chain and can thus be distinguished from field-infected cattle. Strain RB51 is also less abortifacient for cattle than strain 19, although vaccination of pregnant heifers may result in abortion and subsequent zoonotic exposure if obstetric assistance is necessary (Centers for Disease Control, Morbidity and Mortality Weekly Report, March 13, 1998), and so the vaccine is to be administered only to young heifers before pregnancy.261,290a,291
Campylobacter fetus, subspecies venerealis, Abortion
C. fetus, subsp. venerealis, is the main cause of bovine campylobacteriosis (vibriosis).5 The organism is an obligate parasite of the bovine genital tract and is not known to cause disease in other species.266
History and Clinical Signs
Infection with C. fetus, subsp. venerealis, mainly causes temporary infertility or early embryonic death, but sporadic abortions from the fourth to eighth months of gestation are possible.266 The usual history includes a high percentage of cows exposed for the first time returning to estrus or found nonpregnant after the breeding season, and cows calving late because they returned to estrus one or more times.
Laboratory Diagnosis
Autolysis is usually minimal, and the lungs of the term fetus may be partially inflated. Dehydration, fibrinous serositis, and necrotizing placentitis may be apparent grossly. Histologically, bronchopneumonia and hepatitis may also be evident. Diagnosis is based on demonstration or isolation of the organism. By darkfield microscopy the bacterium appears as a curved rod with darting corkscrew motility.266 Cows that abort may have serum antibody titers; however, they may not be diagnostic because they are not specific for C. fetus, subsp. venerealis., Culture from placenta or fetal abomasal contents requires at least 72 hours. The vaginal mucus agglutination test is used to survey herds for infection.5 Alternatively the penis and preputial mucosa of infected bulls may be swabbed and cultured, although culture is difficult because the organism is slow-growing and often overwhelmed by saprophytes.
Pathophysiology
Within a week of vaginal infection the organism is established in the uterus, causing mucopurulent endometritis, which persists 3 to 4 months. Intrauterine infection either prevents conception or causes embryonic death, and infected heifers typically return to estrus by 40 days. Less commonly, abortions occur at up to 8 months’ gestation.5,266
Epidemiology
C. fetus, subsp. venerealis, is ubiquitous. Venereal transmission from infected bulls to virgin heifers approaches 100%. Cows with previous exposure to infected bulls develop immunity and therefore are less likely to experience infertility than heifers. The abortion rate seldom exceeds 10%.266
Treatment and Control
Infected cows usually recover spontaneously within 5 months and resist reinfection. Recovery is hastened by intrauterine infusions of streptomycin and penicillin. Infertility may be permanent if endometritis or salpingitis is severe.5 Heifers should be vaccinated with a killed bacterin before breeding. Most vaccines are administered 1 month before breeding and require a booster vaccination 2 weeks later.*† Higher than normal doses of vaccine may be needed to clear the infection from bulls. Cows and bulls must be vaccinated annually. Exclusive use of C. fetus, subsp. venerealis,—negative semen via AI controls the disease by preventing transmission.
Haemophilus somnus, Abortion
H. somnus, has been associated with vulvitis, vaginitis, endometritis, weak calf syndrome, stillbirths, and occasional abortion in cattle.
Laboratory Diagnosis
Aborted fetuses have been free of gross lesions. Necrotizing placentitis is associated with fibrinoid necrosis of placental arteries.207 Diagnosis is based on recovery of large numbers of the organisms in relatively pure culture from placenta or fetus, histologic evidence of placentitis, and lack of other apparent causes.
Interpretation of maternal titers to H. somnus, is difficult, and it is best to take paired serum samples. Titers between 1:256 and 1:512 in nonvaccinated herds may be the result of early active or chronic infection. Titers between 1:1040 and 1:4096 indicate recent active infection. A fourfold change in titer in paired sera is the best indicator of active infection.
Pathophysiology
Although H. somnus, can be isolated from the genital tract of clinically normal cows,194 the rate of isolation is higher in cows with endometritis or cervicitis.207,292 Experimentally, H. somnus, can adhere to zona pellucida—intact embryos and cause degeneration.293 Vaginitis can be induced by inoculation with H. somnus,294 and abortion has been induced by intraamniotic, intravenous, or intrabronchial challenge with the organism.295 Cervical infusion with the organism has resulted in colonization of the chorioallantois and placentitis, but calves were born alive without culturable H. somnus.,296,297
Treatment and Control
Antibiotic treatment and vaccination anecdotally increase fertility in herds affected with H. somnus,—induced vulvovaginitis.207
Listeria monocytogenes, Abortion
Listeriosis is caused by L. monocytogenes., Listerial abortions are of importance mainly in ruminants.
History and Clinical Signs
Bovine abortions usually occur in the last 2 months of gestation.266 Infected ewes and does typically abort in the last month.5,298 Fever, depression, RFMs, or endometritis may occur,5,266,299 but often the dam shows no clinical signs of infection.
Laboratory Diagnosis
In less severely autolyzed fetuses, fibrinous polyserositis may be apparent. Most aborted fetuses have gray-white hepatic foci up to 2 mm in diameter. Similar foci may be visible in cotyledons; exudation occurs between cotyledons. Abomasal erosions have been reported in aborted lambs.266 Histologically, suppurative placentitis and endometritis are consistent findings.
Listeria, is readily cultured from abortuses without cold enrichment. Serovars 1 and 4b are commonly isolated from bovine fetuses; serovars 4b and 5 are the usual ovine isolates.266Listeria, appears in impression smears as gram-positive pleomorphic coccobacilli.266
Pathophysiology
Listerial abortion can be induced experimentally in cattle 6 to 8 days after infection and in sheep 3 to 11 days after infection. Fetuses die from placentitis and septicemia and are often retained in utero several days before expulsion.266
Epidemiology
Listerial abortion is usually sporadic, and incidence seldom exceeds 15%.266 Infection is most common in the winter and has been associated with feeding of silage. The elevated pH of spoiled silage enhances multiplication of the organism. Aborted tissues are infectious for humans and should be handled with care.
Treatment and Control
The effect on fertility is usually transient, and aborting animals tend to resist reinfection. Tetracycline may be used in remaining pregnant animals in the herd.5 Aborting animals should be segregated, and fetuses and placentas should be removed from the premises. The feeding of spoiled silage should be avoided.
Mycoplasma, Abortion
Mycoplasmal isolations from the bovine genital tract have been mainly M. bovigenitalium, and Mycoplasma bovis. M. bovis, is probably the more important cause of abortion.299Mycoplasma mycoides, subsp. mycoides, (see Mycoplasma, Polyarthritis) and Mycoplasma agalactiae, have been associated with caprine abortions.5
History and Clinical Signs
M. bovigenitalium, is associated with granular vulvovaginitis and less commonly with endometritis, especially in heifers. Infertility is more common than abortion. M. bovis, causes mastitis and abortion.299 In goats, mycoplasmal infection is associated with septicemia, arthritis, pneumonia, mastitis, and abortion.5
Laboratory Diagnosis
Placentitis and fetal pneumonia have been associated with bovine mycoplasmal abortion.299 Isolation of Mycoplasma, from the genital tract, milk, placenta, or fetus indicates infection. However, mycoplasmosis should not be considered the cause of abortion unless placentitis or fetal inflammation is present and other more likely causes of abortion have been eliminated.
Pathophysiology
M. bovigenitalium, can be isolated from the vagina of as many as 12% of clinically normal cows, but M. bovis, is isolated from fewer than 1%. Vulvitis can be induced by inoculation with mucosal scarification with M. bovigenitalium;, therefore venereal transmission may be the natural route of infection.5,299 Experimental inoculation with M. bovis, induces abortion with placentitis and fetal pneumonia.299,300M. bovigenitalium, is rarely isolated from abortuses or normal fetuses.5
Epidemiology
Mycoplasma, species are ubiquitous, but mycoplasmal abortions are not commonly documented.
Treatment and Control
Tetracycline or tylosin is the recommended antibiotic for mycoplasmal granular vulvovaginitis in heifers.299
Salmonella, Abortion
A variety of Salmonella, serotypes have been isolated from aborted fetuses of cattle, sheep, goats, and horses. A complete discussion of salmonellosis is presented elsewhere. Infection is acquired by ingestion of contaminated feed or water. Maternal septicemia is followed by localization of salmonellae in tissues, including the pregnant uterus, where placentitis and fetal septicemia occur. Salmonellosis accompanied by endotoxemia causes early pregnancy loss without colonization of the uterus, because the infection and endotoxemia cause endogenous prostaglandin release. Endogenous PGF2α initiates luteolysis and abortion in the first trimester (<100 days for cattle), as is the case with exogenous administration of PGF products.
History and Clinical Signs
The animal may show systemic signs before abortion. Abortion may occur at any stage of gestation and is characterized by placental necrosis, edema, and hemorrhage. RFMs and fetal autolysis may occur. Abortion may be accompanied by diarrhea, fever, or vaginal discharge, particularly in the ewe, but often infection is not clinically apparent in the dam.266,301,302 Fetuses also may be lost as a result of stillbirth or perinatal septicemia.
Laboratory Diagnosis
The fetus is frequently autolyzed. Placentitis is usually present. Diagnosis is based on isolation of the organism and evidence of placentitis or inflammation of fetal tissues. FA techniques can identify the bacteria in impression smears or sections of placenta or fetal tissue.266 The dam can be tested serologically for evidence of recent active infection,301 but at present many diagnostic laboratories do not perform Salmonella, serology.
Pathophysiology
Infected adult animals are often short-term carriers and shed salmonellae in the feces or milk. True long-term asymptomatic carriers occur mainly with the host-adapted serotypes: Salmonella, Dublin in cattle, Salmonella, Abortus ovis in sheep, and S. abortivoequina, (or Salmonella, Abortus equi) in horses. Salmonellosis caused by Salmonella, Abortus equi has been eradicated in the United States. Occasionally, long-term carriers of other serotypes are seen; these are usually intestinal carriers and fecal shedders. Infection usually occurs by ingestion. There is no evidence of venereal transmission.301 Maternal septicemia is followed by localization of the organism in a variety of tissues, including the pregnant uterus. The bacteria multiply in and cause necrosis of connective tissues of the cotyledon.301 The incubation period between infection and abortion varies from approximately 1 week to 1 month.5,302 Fetal death results from placentitis and fetal septicemia.266,303 In most cases, maternal shedding of the organism in cattle ceases by 5 weeks after calving.301
In a second mechanism of Salmonella,-induced abortion, Salmonella, septicemia causes endotoxemia and release of endogenous PGF, which causes luteolysis and abortion. In this case the fetus and placenta are culture negative for salmonellae.
Epidemiology
Bovine abortion resulting from salmonellosis is caused mainly by Salmonella, Dublin and Salmonella, Typhimurium. Abortion is sporadic and most common in the summer and fall.301
Ovine abortion is associated with Salmonella, Typhimurium, S., Dublin, Salmonella, Arizona, and Salmonella, Abortus ovis. Salmonella, Abortus ovis (which infects only sheep) is enzootic in parts of England and Europe but is not reported from the United States.5,266 Young ewes in late gestation are most susceptible, and the abortion rate may approach 50%,5,302 but usually only one or two ewes in a flock abort.266
Treatment and Control
Metritis is a rare complication of salmonellosis that can be fatal.266 Usually there is no lasting effect on fertility, but animals infected with host-adapted serotypes may become carriers and should be cultured and tested serologically and culled if positive. Salmonellosis can be controlled by hygiene and by avoiding the introduction of carrier animals. Aborting animals should be isolated; the fetus, placenta, and contaminated material should be removed from the premises. Salmonella, species are infectious for humans; therefore aborted tissues should be handled with caution.
Ureaplasma, Abortion
Ureaplasma, is a small bacterium without cell walls; it differs from Mycoplasma, in its ability to hydrolyze urea. U. diversum, has been associated with granular vulvitis and abortion in cattle.
History and Clinical Signs
Granular vulvitis appears as reddish nodules in the vulvar mucosa, with mucopurulent discharge in the early stages. The discharge is usually more copious and protracted than with IPV induced by herpesvirus.5,299 Affected cows are not systemically ill.266 The organism has been recovered from embryo flushing media and can adhere to the zona pellucida, resisting removal by washing. It is believed to be responsible for an 18% reduction in pregnancy rate in embryo recipients when the transfer medium contains the organism.
Laboratory Diagnosis
Gross lesions include thickening of placental membranes with foci of hemorrhage and fibrinous exudate. Gross lesions are seldom apparent in the fetus. Microscopically the placenta is fibrotic, with heavy mononuclear cell infiltration, multifocal necrosis, fibrin deposition, and mineralization. Cuffs of lymphocytes surround fetal intrapulmonary airways.266 Diagnosis is based on isolation of the organism from genital mucosa, placenta, or fetal stomach or lung and the presence of genital or fetal inflammation.
Pathophysiology
Ureaplasma, can be isolated from the genital tract of normal cows and from normal fetuses.304 Vulvitis has been induced by inoculation of virgin heifers with U. diversum.,304 Uterine involvement is considered rare but may cause conception failure, early embryonic death, or abortion. Abortion presumably results from placentitis.5 Intraamniotic inoculation of U. diversum, caused placentitis, abortion, and fetal alveolitis in two of four experimental cows.304 One cow delivered a weak calf at term. Experimental infection in ewes did not decrease fertility.305
Epidemiology
Bovine infection is common, but documented abortions caused by Ureaplasma, are rare.299 Infertility is more common in heifers than in cows.
MISCELLANEOUS BACTERIAL ABORTIONS
In addition to the aforementioned bacteria, other bacteria occasionally produce maternal septicemia. Many of these bacteria are ubiquitous, frequently contaminate aborted fetuses and placentas, and should not be considered the cause of abortion unless (1) they are isolated from the placenta and fetus in large numbers and relatively pure culture, (2) placentitis or fetal inflammation is evident, and (3) other more likely causes of abortion have been eliminated.266
In cows, most bacterial infections of the uterus result from septicemia. Miscellaneous bacterial causes of infertility and abortion include A. pyogenes, E. coli, Bacillus, species, Pasteurella, species, Staphylococcus, species, Streptococcus, species, F. necrophorum, and Bacteroides melaninogenicus.,266
A. pyogenes, with or without accompanying anaerobes has been associated with pyometra and abortion in cattle.188,266,306 Abortions are sporadic and may occur at any stage of gestation. Clinical signs are seldom apparent in the cow. The fetus is commonly autolyzed, and placentitis is typical. Polyserositis may be evident. In fetuses aborted in the first half of gestation, 1-mm yellow foci (bacterial colonies) may be grossly apparent in the lung.266
In the ewe or doe, miscellaneous bacterial causes of abortion include Staphylococcus aureus, Streptococcus species, Pasteurella species, E. coli, Yersinia pseudotuberculosis, Francisella tularensis, Histophilus ovis, Bacillus species, A. pyogenes, and Corynebacterium species.266,307-310
A spirillum-like organism has been documented as a cause of ovine abortion, fetal mummification, stillbirth, and birth of weak lambs. Abortion generally occurs in the last 2 weeks of gestation. Placentitis is consistently present; many fetuses also have fibrinous peritonitis and focal hepatic necrosis that resembles that of campylobacteriosis.311 Abortion can be reproduced experimentally by inoculation of pregnant ewes.312 Diagnosis is based on pathologic lesions and identification of the spindloid flagellated organism by darkfield microscopy of fetal abomasal content or liver or by anaerobic culture on selective medium.311,313
Tritrichomonas foetus
Trichomoniasis is a venereal infection of cattle caused by the flagellated protozoan T. foetus.,5,189,266,314,315
History and Clinical Signs
Infertility characterized by a high percentage of cows returning to estrus or found nonpregnant after the breeding season and cows calving late plus occasional pyometras and abortions are the most common clinical signs of trichomoniasis. Pyometra in postcoital heifers or cows suggests that trichomoniasis may be the cause. Abortions generally occur in the first half of gestation at a rate of 5% to 30%.314 The placenta may be expelled or retained.5,266
Laboratory Diagnosis
Diagnosis in the female is made by identifying or culturing trichomonads from cervicovaginal mucus (approximately 76% sensitivity), uterine exudate, placental fluids, or fetal abomasal contents.5,189,266,314,315 Preputial smegma collected from bulls by using a plastic pipette run against the mucosa can also be cultured for trichomonads (sensitivity 80% to 90%). Diamond’s medium (or modified Pastridge medium) is recommended for cultures from cows, bulls, or aborted fetuses.315 Samples should be transported at ambient temperature, kept out of sunlight, not refrigerated, and delivered promptly to the diagnostic laboratory. The organisms are identified microscopically by their size (10 μm×15 μm), the presence of three anterior flagellae and an undulating membrane, and a characteristic jerky, rolling motion.266
There are no specific gross lesions in aborted fetuses. However, placentitis is a consistent microscopic lesion, and trichomonads can frequently be recognized in the placental stroma in histologic sections. Organisms may also be observed in the fetal lung in association with pyogranulomatous bronchopneumonia.316
Pathophysiology
Trichomoniasis is transmitted venereally from infected bulls to cows or vice versa. The organisms colonize the vagina, cervix, uterus, and oviducts; yet they do not generally interfere with conception. Embryonic death frequently occurs within the first 2 months of infection,5 followed by a 2- to 6-month period of immunity to reinfection.189 Clearance of infection in cows commonly occurs within 95 days; infection rarely persists as long as 6 months.314 However, infection in bulls over 4 years of age is permanent and the main source of carryover from one breeding season to the next in beef cattle.189
Treatment and Control
Systemic treatment of infected animals with imidazole compounds (ipronidazole, dimetridazole) is effective, but these compounds are prohibited in food animals in the United States.242,317,318 Infected cows should be either culled or given at least 3 months’ sexual rest. The use of AI with semen from T. foetus,—negative bulls controls the disease once infected natural service bulls have been removed from the herd. In natural breeding situations, vaccination, use of young virgin bulls, and testing and elimination of positive bulls older than 3 years of age will allow herd owners to gain control over the incidence of trichomoniasis.
Neospora caninum, Abortion
Abortion in cattle caused by N. caninum, is relatively common and of economic importance especially in dairy cattle. Congenital infection with limb paresis or dysfunction at birth as a result of encephalomyelitis may also occur. Canids are the definitive hosts and shed oocysts in their feces after ingestion of infected tissues from intermediate hosts (Fig. 43-12). Both the domestic dog and the coyote have been shown to transmit the oocysts in their feces.319,320,321 Along with cattle, deer have been shown to serve as intermediate hosts.321 The role of birds and wild rodents as intermediate hosts is suspected but has not yet been definitively demonstrated.321 Aborted, infected fetal tissues ingested by canids will also result in massive shedding of oocysts in feces.322 Transmission may be vertical (dam to fetus in utero) or via point-source horizontal transmission (ingestion of feed contaminated with feces containing oocysts).323,324 Horizontal transmission from cow to cow does not occur. Bovine fetal lesions are distinctive and may include nonsuppurative encephalitis with foci of necrosis and gliosis, nonsuppurative myositis, hepatitis, and, most consistently, myocarditis.261 Most abortions occur in the early second trimester, but they may occur throughout gestation. Diagnosis is based on characteristic lesions in aborted fetuses, and seroepidemiologic study of an equal number of aborting and nonaborting herdmate cows if the proportion of seropositives is statistically higher in those that have aborted.325 Finding one aborting cow to be seropositive does not confirm N. caninum, as the cause, and cows that abort once because of N. caninum, are not protected from future abortion caused by this organism. A vaccine based on whole killed tachyzoites (Neoguard1, Intervet, Millsboro, Del.) is available for N. caninum.,
Fig. 43-12 Transmission of bovine neosporosis. Oocysts are produced by the canine definitive host, and their subsequent ingestion by a susceptible pregnant cow leads to infection of the fetus (exogenous transplacental transmission). Liveborn infected heifer calves would be expected to remain infected into adulthood, when they, in turn, may pass infection to their fetuses (endogenous transplacental transmission). Spread of N. caninum, in this second way is the principal route whereby the parasite is propagated in a herd.
Sarcocystis, Abortion
Sarcocystosis may cause abortion in cattle, sheep, and goats. Cattle are infected by Sarcocystis cruzi, sheep by Sarcocystis ovicanis, and goats by Sarcocystis capracanis.,
History and Clinical Signs
Most cattle are infected with Sarcocystis, but do not show clinical signs of infection; however, massive or repeated infections may elicit depression, anorexia, weight loss, lameness, hair loss, emaciation, or death. Abortions occur in late gestation, usually in severely affected animals.266,326,327
Laboratory Diagnosis
There are no specific gross lesions in the aborted fetus. Histologically protozoa may be observed in villi and small arteries of the cotyledon or (more likely) the caruncle but are seldom seen in the bovine, ovine, or caprine fetus. Parasites are more likely to be observed in the fetal brain than in other tissues.326 Nonsuppurative inflammation may occur in the placenta or, less commonly, in fetal tissues, particularly the brain, heart, lung, liver, or kidney.266,326,327
FA demonstration of numerous developing protozoa in the cotyledon or caruncle is considered diagnostic. Caruncles are reported to be atrophied.326 The protozoa can also be isolated by feeding aborted tissues to canids and recovering coccidian sporocysts from their feces.266
Pathophysiology
The pathogenesis of abortion in sarcocystosis is unclear. Generally, numerous spores are required to induce abortion experimentally. Fetal invasion by Sarcocystis, is rare, and abortion may result from maternal fever, anemia, or placental insufficiency.326 Pregnant does apparently have diminished immunity to Sarcocystis, because low doses of S. capracanis, result in fetal death without maternal illness if given in early pregnancy.328
Fungal Abortions
Mycotic Diseases That Cause Abortion
Fungal causes of bovine abortion include Aspergillus, Absidia, Mucor, Rhizopus, Candida, and Mortierella.,266,329,330 Mycotic abortion is uncommon in sheep and goats.266
History and Clinical Signs
Mycotic abortions usually occur in the latter half of gestation (often near term) and seldom are associated with prodromal or postabortion clinical signs in the dam.
Laboratory Diagnosis
The most consistent lesion is placentitis with necrosis and thickening of fetal membranes. In ruminants, both cotyledons and the intercotyledonary placenta are affected. Histologically, necrotizing inflammation of the chorionic villi is associated with vasculitis and thrombosis.266 Gross lesions may not be apparent in the fetus, but granulomatous bronchopneumonia is frequently observed histologically.266,330-332 In bovine aspergillosis the fetus is often near term with minimum autolysis and partially inflated lungs. Emaciation and dehydration with multifocal dry, scaly skin lesions occur in approximately 25% of affected fetuses.266 With other fungi the aborted fetus is often autolyzed, placentitis may be more severe, and skin lesions, if present, tend to be moister than those of aspergillosis.266 The placenta is characteristically thickened and leathery. The fetus may be emaciated, and granulomatous bronchopneumonia has been observed.331 The fungus can be isolated from the fetus and placenta or demonstrated on histologic sections with immunostaining.
Epidemiology
Mycotic abortion generally affects only one or two animals in a herd and is more common in the winter.266 Fungi cause 3% to 10% of bovine abortions.333,334Aspergillus, accounts for up to 80% of bovine mycotic abortions; Mucorales account for an additional 10% to 15%.5Mortierella, is a common abortifacient in Australia and New Zealand and has been associated with feeding of grass silage,330,334 but Mortierella, abortion is rare in the United States.
DOES AND EWES
INFECTIOUS CAUSES
Bluetongue Abortion
Bluetongue is an orbivirus infection that can result in embryonic death, abortion, and fetal anomalies in sheep, cattle, or other ruminants. Twenty-four serotypes of bluetongue virus have been recognized; five of these (serotypes 2, 10, 11, 13, and 17) occur in the United States (see Chapter 32 for complete discussion and Chapter 35 for nervous system anomalies).335
Border Disease (Hairy Shaker Disease)
Border disease or hairy shaker disease is an ovine pestivirus infection that causes embryonic and fetal death; stillbirths; dysplasia of the central nervous system, skeleton, and fleece; and birth of weak lambs with low viability.
Coxiella burnetii, (Q Fever)
Coxiella burnetii, a rickettsia, is the causative agent of Q fever, which can cause abortions in sheep and goats. Most infections are completely asymptomatic, and the disease is of more importance as a zoonosis than as a cause of ovine or caprine abortion.
History and Clinical Signs
Late abortions or delivery of weak lambs may occur in an affected flock over a period of 2 to 4 weeks.5,302
Laboratory Diagnosis
There are no specific gross lesions in the fetus, but the placenta is thickened with white, chalky plaques and red-brown exudate, especially in intercotyledonary areas. Histologically cotyledonary and intercotyledonary necrosis is accompanied by heavy neutrophil infiltration.5,336 Diagnosis of Q fever abortion should be based on the presence of characteristic placental lesions with large numbers of rickettsiae and a rising maternal titer. Rickettsieae can be identified in placental impression smears stained with modified Koster’s stain, Stamp’s modified Ziehl-Neelsen stain, or Gimenez stain as pleomorphic acid-fast coccoid or filamentous organisms in trophoblasts or extracellularly. Complement fixation titers greater than 1:8 in the dam are considered diagnostic. Enzyme-linked immunosorbent assay is rapid and sensitive but requires species-specific peroxidase conjugate for each host species. Laboratory results must be interpreted carefully because C. burnetii, also can be isolated from the placenta of normal animals.5 In one survey of California dairy goats, 24% were seropositive by microagglutination.337
Pathophysiology
C. burnetii, infection can be transmitted by ixodid or argasid ticks or by ingestion of infected material. The organism replicates in trophoblasts and is often of low pathogenicity in sheep and goats but can result in placentitis with late abortion and shedding of large numbers of rickettsiae.5
Epidemiology
Q fever is reported in many countries, including the United States and Canada. Abortions typically occur over a 2- to 4-week period and may affect 5% to 50% of the flock.5 The organism also is infectious for other animals, including humans. Pregnant women should not handle Q fever—infected animals or tissues.
Treatment and Control
Aborting does and ewes should be segregated, and abortuses and placentas removed from the premises to prevent oral transmission. Pregnant animals can be treated with tetracycline to reduce the chances of abortion. A carrier state may develop, but abortions do not usually occur in subsequent pregnancies in sheep; they may be more likely to recur in goats.338 Inactivated vaccines (not commercially available) lessen the chances of rickettsial abortion in sheep and reduce but do not eliminate rickettsial shedding at parturition.336
Campylobacter fetus, subspecies fetus, Abortion
C. fetus, subsp. fetus, is one agent of ovine campylobacteriosis (vibriosis) that causes abortion in sheep and sporadic abortion in cattle and goats.5,266,302
History and Clinical Signs
Infection of ewes causes abortion in the last 6 weeks of pregnancy, stillbirths, and birth of premature lambs. Infected ewes may have fever, diarrhea, depression, and vaginal discharge several days before parturition.302 In cattle the infection is not associated with infertility (unlike infection with C. fetus, subsp. venerealis,), but sporadic abortions can occur from the fourth to eighth months of gestation.266
Laboratory Diagnosis
Campylobacter, causes placentitis with cotyledonary necrosis and intercotyledonary edema. The fetus is edematous and may have fibrinous polyserositis. Foci of necrosis up to 2 cm in diameter occur in the liver of approximately 40% of aborted ovine fetuses and, although not pathognomonic, suggest campylobacteriosis.5,266 Histologic changes include suppurative necrotizing placentitis and fetal bronchopneumonia.266 Diagnosis is based on culture (which usually requires less than 48 hours)5 or on microscopic demonstration of the organism.5
Pathophysiology
C. fetus, subsp. fetus, is transmitted by ingestion. The organism localizes in the gallbladder but may invade the pregnant uterus, where it replicates in chorioallantoic trophoblasts. The incubation period in the ewe varies from 7 to 25 days.5 In the cow, dissemination of the organism to the placenta is less common.266 In either species, localization in the placenta causes placentitis and fetal bacteremia.266 Metritis, fetal retention, and maternal peritonitis may occur in the ewe.5
Epidemiology
Infection with C. fetus, subsp. fetus, is important in sheep in the United Kingdom, the United States, and New Zealand.5 Fetal infection is most common during the last 2 months of gestation. Outbreaks of abortion tend to occur in 4- to 5-year cycles.5 Infection is highly contagious in confined ewes, and the abortion rate may approach 70% but is more commonly approximately 25%.5,266,302
Treatment and Control
Abortion outbreaks can be treated with daily intramuscular injections of procaine penicillin G (22,000 IU/kg) and dihydrostreptomycin (11 to 22 mg/kg) or with oxytetracycline in the feed (75 mg/head/day).5 Metritis is rarely fatal in the ewe. Generally, affected ewes abort only once and therefore they may be retained as breeding stock.266 Ovine campylobacteriosis can be controlled by the use of a killed adjuvanted bacterin at breeding and 60 to 90 days later.5
Campylobacter jejuni, Abortion
Campylobacter jejuni, is the other agent of ovine Campylobacter (Vibrio), abortion. C. jejuni, is an enteric pathogen that causes enteritis and diarrhea in many species. Only in sheep is placental and fetal infection common. This organism has been associated occasionally with abortion in cattle and goats.5,11,266,339,340
History and Clinical Signs
Ovine infection with C. jejuni, is clinically indistinguishable from that with C. fetus, subsp. fetus.,
Laboratory Diagnosis
Aborted fetuses are autolyzed and frequently lack specific gross lesions. Cotyledons are mottled yellow to tan, but the intercotyledonary membranes are grossly normal. Histologically necrosis occurs in chorionic villi with arteriolitis and numerous leukocytes in the lamina propria. Purulent bronchopneumonia is a common histologic finding in the fetus.341
C. jejuni, can be distinguished from other Campylobacter, species by growth at 42° C (107.3° F), resistance to cephalothin, inhibition by nalidixic acid, and the presence of heat-labile glycoprotein surface antigen 1, which does not occur in C. fetus, subsp. venerealis, or C. fetus, subsp. fetus., Placenta or fetal tissues should be cultured on Campylobacter, agar with incorporated cefoperazone, vancomycin, and amphotericin B (CVA medium) at 42° C (107.3° F).340,341
Pathophysiology
Intravenous inoculation of pregnant ewes with C. jejuni, at 114 and 123 days of pregnancy consistently induced abortion 7 to 12 days later in one study.341
Epidemiology and Control
C. jejuni, is ubiquitous. Certain strains have been associated with an abortion rate as high as 80%, but usually fewer than 20% of the animals in the flock abort.341 Aborted tissue is infectious for humans and should be handled with caution. Treatment and control of ovine infection with C. jejuni, are similar to those for C. fetus, subsp. fetus.,
Brucella melitensis, Abortion
Brucella melitensis, causes abortion in goats and sheep and less commonly in cattle.5 Infection is associated with late abortion, stillbirth, or birth of weak kids or lambs.5,283 Necropsy findings include severe placentitis and fetal serositis.5,302 The disease is diagnosed by culture or demonstration of the organism in tissue or by maternal serology (complement fixation test).5 Animals are infected by ingestion, and, after bacteremia, replication occurs in chorioallantoic trophoblasts.5,285 Infection with B. melitensis, is important in Mediterranean countries and in Central and South America but is rare in the United States.283,302 Suspected cases should be reported to state and federal authorities. Human infection with B. melitensis, can be very severe.
Brucella ovis, Abortion
Brucella ovis, infects only sheep. Epididymitis in rams is the most common manifestation of infection. Ewes seldom show clinical evidence of infection; late abortions, stillbirths, and delivery of weak lambs are rare.5,266 Fetal and placental lesions resemble those induced by B. abortus., Diagnosis is based on culture or on demonstration of the organism in tissue. Serology (complement fixation) can identify infected animals. However, not all ewes showing a rise in complement fixation titer at parturition deliver infected lambs. In addition, ewes can maintain elevated titers for months or years after exposure to B. ovis., Therefore B. ovis, abortion should not be diagnosed on the basis of complement fixation testing alone.266
Infection is thought to occur by the conjunctival route.302 The bacterium has low virulence for the ewe but may replicate in chorioallantoic trophoblasts, resulting in placentitis and fetal bacteremia.285B. ovis, infection has been reported from Europe, Africa, Australia, New Zealand, and the western United States.
Chlamydia psittaci, Abortion (Enzootic Abortion of Ewes)
C. psittaci, is a major cause of abortion in sheep and goats.5,266 Ovine chlamydial abortion is called enzootic abortion of ewes. Chlamydia, may cause abortion in cattle266 but is not the cause of epizootic bovine (foothill) abortion.
History and Clinical Signs
Chlamydiosis is best characterized in sheep and goats. Abortions or stillbirths with placentitis usually occur in the fourth to fifth months of gestation. The dam seldom shows signs of illness but may have serosanguineous vaginal discharge several days before and after parturition.5 Other animals in the flock may be affected by arthritis or pneumonia.342
Laboratory Diagnosis
Placentitis is the most consistent necropsy finding in chlamydial abortion. Necrosis occurs in cotyledons; the intercotyledonary placenta is thickened with accumulation of red exudate.266,342 The fetus has no specific gross lesions.266,342 Histologically, necrotizing placentitis is accompanied by nonsuppurative vasculitis. Nonsuppurative meningoencephalitis, necrotizing hepatitis, and proliferation of mononuclear cells in spleen and lymph nodes are other histologic findings that may occur in the fetus.266,343
Diagnosis of chlamydial abortion should be based on identification of the organism and the presence of typical placental lesions. Chlamydiae appear in placental impression smears stained with Giemsa, Gimenez, or modified Ziehl-Neelsen stains as 200-nm dark red spheric bodies in the cytoplasm of trophoblasts.5,266 The organism can be positively identified by FA tests on cytologic preparations, cryostat sections of placenta or other fetal tissues, or cultures. Chlamydia, grows in chick embryos in 1 to 6 weeks, but cell culture using mouse L cells requires only 2 to 10 days.266
Abortion or delivery of chlamydial-infected fetuses induces a rise in serum titer that peaks in 2 to 3 weeks.266 Paired sera should be collected at abortion and 3 weeks later. Maternal titers greater than 1:32 generally indicate recent active infection.344 Complement fixation is the standard serologic test for the dam; indirect immunofluorescence and enzyme-linked immunosorbent assay are also used.345 Double immunodiffusion can be performed on fetal fluids.266
Pathophysiology
Chlamydiae reside in the intestinal tract and are also shed from the genital tract of infected animals before and after parturition. Ingestion is the main form of transmission.5,346 In sheep and goats, abortion occurs 4 to 8 weeks after experimental infection,342 but the fetus is not susceptible until the last third of gestation.5
High maternal antibody titers do not prevent abortion or stillbirth, but experimental work suggests that cell-mediated immunity is protective.347 Infection with small numbers of Chlamydia, organisms seldom stimulates adequate cell-mediated immunity and consequently may be more likely to cause abortion than infection with numerous organisms.5
Epidemiology
Chlamydial abortion has been reported from most major sheep- or goat-producing countries. C. psittaci, also may infect other animals, including human beings. Pregnant women should not handle infected animals or tissues. The abortion rate in sheep is usually approximately 5%266 but may be up to 30% or more in goats.5,342 Although abortions usually occur 1 to 2 months after infection in sheep, the incubation period in goats may be as short as 2 weeks.344
Treatment and Control
Aborting does or ewes should be segregated, and abortuses and placentas should be removed from the premises to avoid oral transmission. Oxytetracycline therapy (80 to 450 mg/head every day in feed or water, or long-acting oxytetracycline injected subcutaneously at a dose of 20 mg/kg twice weekly until the last month of gestation)5 reduces the number of abortions and stillbirths in sheep347 and goats, particularly if instituted in the first half of pregnancy.5 However, short-term treatment does not eradicate infection or prevent chlamydial shedding at parturition and should be reserved for abortion outbreaks.348 Most ewes develop cell-mediated immunity, which eliminates the organism from the genital tract by 3 months after lambing and protects against abortion for approximately 3 years.5,348 Does are more likely than ewes to have placental retention and metritis after abortion.346 Killed vaccines* can be used in enzootic areas 4 to 6 weeks before breeding but are not 100% effective. Experimentally avirulent live vaccines have been used with success.
Toxoplasmosis Abortion
Toxoplasma gondii, is a ubiquitous protozoan that is a major abortifacient in sheep and goats but only rarely causes abortion in cattle or horses.
History and Clinical Signs
Infection does not cause clinical illness in the adult but may result in embryonic death, fetal death and abortion, stillbirth, or birth of weak, nonviable lambs or kids.
Laboratory Diagnosis
The most characteristic gross lesion of toxoplasmosis is the presence of white, chalky foci of necrosis and calcification up to 2 mm in diameter in cotyledons.349-351 The intercotyledonary areas of the placenta are grossly normal. Specific gross lesions are not observed in the aborted fetus, but histologically most have nonsuppurative encephalomyelitis350 and many also have pneumonia, myocarditis, or hepatitis. Tachyzoites may be found in placenta or other fetal tissues but are not numerous.351,352 The tachyzoites are oval, 2 to 4 by 4 to 8 mm, with a central nucleus and appear larger in impression smears than in paraffin sections. Several serologic tests, including the modified agglutination test, indirect FA test, Sabin Feldman dye test, indirect hemagglutination test, and enzyme-linked immunosorbent assay, reliably detect toxoplasmosis in pleural or amniotic fluid or presuckling serum from nondecomposed fetuses.266,353,354 The modified agglutination test is commercially available, safer, and more sensitive than the dye test. Fetal antibodies to T. gondii, can be detected 35 days after infection.349 Absence of fetal antibody does not always preclude a diagnosis of toxoplasmosis.266 High maternal titers are not diagnostic of toxoplasmal abortion, but lack of titer eliminates toxoplasmosis as the cause for abortion.349 The peroxidase-antiperoxidase method for detecting Toxoplasma, antigen in fetal tissues or placenta is reliable even in autolyzed fetuses. Fetal heart, lung, brain, spinal cord, skeletal muscle, and placenta are the preferred specimens for the peroxidase-antiperoxidase method and should not be held in formalin more than 2 days before paraffin embedding.352T. gondii, can be isolated by intraperitoneal inoculation of placental or fetal tissue suspensions into mice.266
Pathophysiology
Placental infection occurs approximately 14 days after ingestion of oocysts.352 Infection acquired before 50 days’ gestation may result in embryonic death and resorption. Infection between 60 and 100 days’ gestation usually causes fetal death or birth of weak lambs. Infection during the last month of gestation often has no apparent effect on the fetus.349 In experimental infection of ewes between 6 and 14 weeks of pregnancy, abortions occurred 1 to 2 months after inoculation.354 In natural infections, most abortions occur 1 month before parturition.349
Epidemiology
Toxoplasmosis is a major cause of ovine abortion in many sheep-raising countries, including the United States.350,351,355 Sheep are infected by ingestion of oocysts from feed or grass contaminated with cat feces. Most ewes are infected by 4 years of age.356 Aborted tissues may be infectious for humans and should be handled with caution.
Control
Infected ewes or does seldom abort from toxoplasmosis in subsequent pregnancies.350 The prevalence of abortion can be reduced by avoiding contamination of feedstuffs with feline feces. Cats should not be allowed to eat placentas or carcasses that may contain tachyzoites or tissue cysts. In endemic areas, exposing replacement ewes to aborting ewes may provide immunity before breeding age.5
Mycoplasma, Abortion
Treatment of Mycoplasma, infection in goats is usually not recommended because treated animals may remain carriers. The infection is eliminated from the herd by testing milk and slaughtering infected animals.300
Leptosirosis
Serovar Leptospira, Pomona is the major ovine isolate,266 but Leptospira, Bratislava and Leptospira, Hardjo have also been isolated from sheep.266Leptospira, Grippotyphosa is the major caprine isolate.5 Leptospiral abortion is less common in sheep and goats than in cattle.
CAMELIDS
The incidence of abortion in llamas and alpacas is low. Infectious causes in North America include leptospirosis, toxoplasmosis, chlamydiosis, and other nonspecific uterine infections.191 Bovine virus diarrhea is considered an emerging disease in alpaca herds and may be responsible for some of the early pregnancy losses and abortions in some herds. Diagnostic workup for abortion in camelids is similar to that described in this chapter for other species.