CHAPTER 29 Pain
What is pain?
‘an unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage’
(International Association for the Study of Pain (IASP) 2007)
Pain is highly complex with many interactive dimensions, including physiological, sensory, affective, cognitive, behavioural and psychosocial. The evidence base related to pain is often confusing for the novice therapist because of the different terminology used in different areas of speciality and a lack of clarification between types of pain, the physiological processes involved and the signs and symptoms presented. This section will attempt to summarize the issues surrounding pain, related to the neurologically impaired patient. For more in depth reading, the therapist is referred to the International Association for the Study of Pain (IASP) website (www.iasp-pain.org).
Types of pain
There are three main types of pain experienced by neurologically impaired patients, with patients often presenting with more than one type.
Nociceptive
This type of pain is physiological and arises as a consequence of the activation of nociceptors (pain receptors) following a chemical, thermal or mechanical event. The activation of primary nociceptive afferents by actual or potentially tissue-damaging stimuli is then processed within the nociceptive system (Treede et al. 2008). This type of pain may involve the musculoskeletal system but can also be from a visceral origin. It is important to note that not all nociceptive activation is perceived by the individual as ‘pain’. The perception of an unpleasant experience (pain) is highly subjective. Dystonic pain is associated with abnormal sustained muscle contraction (S3.18), which mediates the activation of nociceptive afferents in the muscles.
Psychogenic
This is pain that is caused, increased, or prolonged by cognitive, emotional, or behavioural factors. The IASP (2007) describe psychogenic pain as:
‘reported pain in the absence of tissue damage or any likely pathophysiological cause. There is usually no way to distinguish their experience from that due to tissue damage if we take the subjective report. If they regard their experience as pain and if they report it in the same ways as pain caused by tissue damage, it should be accepted as pain’.
Neuropathic
This type of pain arises by activity generated within the nociceptive system without adequate stimulation of its peripheral sensory endings (nociceptors) and is caused by a primary lesion or dysfunction in the nervous system (IASP 2007). Some authors have further narrowed the dysfunction to a lesion of the somatosensory system (Treede et al. 2008). This includes the afferent neuron, the ascending and descending pathways, medulla, thalamus, or cerebral cortex. Therefore, neuropathic pain is sub-divided in relation to the anatomical site of the lesion (Dworkin et al. 2003).
Physiology of pain
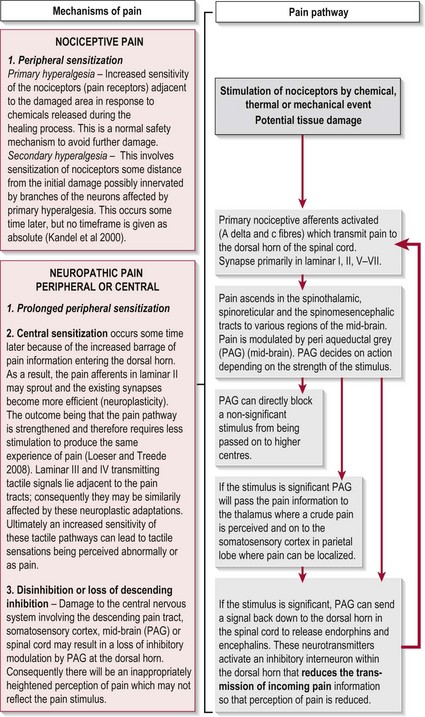
The experience of pain has a protective role which warns us of imminent or actual tissue damage and elicits responses, via signals within the nervous system, which keep such damage to a minimum. This brings about temporary pain hypersensitivity in the inflamed and adjacent tissue (peripheral sensitization). This process assists healing as contact and movement will be avoided. However, persistent pain offers no benefits and can be extremely debilitating. This maladaptive pain often occurs following damage to the peripheral nerve, the spinal cord or the CNS and is termed neuropathic pain (Woolfe and Mannion 1999). Figure 29.1 shows a simple flowchart outlining the normal pain pathway and the processes involved in pathological pain.
Symptoms associated with pain
Nociceptive pain and peripheral neuropathic pain
The symptoms associated with these types of pain are comprehensively covered in many basic physiology texts. More detailed guidance can be sought from a range of musculoskeletal texts in relation to assessment and treatment.
Central neuropathic pain (CNP)
Unfortunately there are no uniform predictable signs with regard to the clinical presentation of CNP and the descriptions often vary greatly between patients. As a result it is often diagnosed through exclusion of any other source.
The symptoms of CNP occur as a result of CNS damage particularly to the somatosensory system which may lead to disinhibition and central sensitization (Fig. 29.1). With the pain pathway now inappropriately active, activity dependent neural plastic changes occur at the synapses within the whole pathway and may lead to an increase in the cortical representation (sensory homunculus) of the painful part. The timescales within which these neural changes take place and in which the symptoms develop is not clear with different studies stating from 1 month to 5–6 years.
The symptoms produced as a result of disinhibition and central sensitization include:
Allodynia
This is pain perceived following a non-noxious stimulus which does not normally provoke pain (e.g. touch, heat or non-noxious cold). This is common (90%) in central neuropathic pain (Widar et al. 2002).
Dysaesthesia
This is an unpleasant abnormal sensation. For example, burning, wetness, itching, electric shock, pins and needles. This is common in CNP (80%) (Attal et al. 2008).
Behaviour of CNP symptoms
The symptoms of CNP may be spontaneous when they present from no apparent source, evoked as an over-reaction to a wide variety of stimuli, persistent (consistent/permanent) or paroxysmal (temporary).
Distribution of neuropathic pain
Central neuropathic pain in neurologically impaired patients
Although neurological conditions result from very different pathophysiology, the pathological processes involved can still be identified as causal in producing CNP. For example, demyelination in MS, compromised circulation in CVA and disruption of spinal cord transmission in SCI can all involve damage to the somatosensory system and therefore produce CNP.
Recent studies have also highlighted a further process that may contribute to CNP which involves the action of glial cells following a CNS lesion. In SCI and MS, inflammatory changes initiated in response to cell damage promote the release of inflammatory mediators and growth hormones by glial cells. If these chemical mediators affect the pain pathway, central sensitization may be enhanced (Svendsen et al. 2005). Although this has not been reported in CVA, it is likely that a similar mechanism takes place following the initial lesion and the resultant inflammatory response.
In PD the mechanism of CNP may be different. In healthy individuals the basal ganglia (S2.11) is now thought to play a significant role in the integration of sensory information and hence the modulation of pain (Juri et al. 2009). In PD the pathophysiology of pain has been linked to a disruption of the basal ganglia’s (striatum) ability to filter the vast amounts of sensory information in order to select an appropriate movement programme. This filtering is normally facilitated by the inhibitory dopaminergic neurons, however as the latter undergoes neurodegeneration in PD its effect is reduced. This may explain why pain may be relieved when in an ‘on’ period (i.e. taking L-Dopa).
Why do I need to assess pain?
While the available knowledge about pain, its physiology and pathophysiology has increased enormously over the last 20 years, it appears to have been largely ignored in respect to patients with neurological deficits. Pain in this population requires recognition as it has been shown to impact significantly on the patient’s quality of life and on the rehabilitative process (Henon 2006).
The incidence of pain in neurologically impaired patients is high, with reports of up to 74% in CVA and SCI; 50–85% in MS and between 40% and 75% in PD. A further breakdown of the different types of pain presented in these conditions can be seen in Tables 29.1-29.3
Table 29.1 Incidence of different types of pain in cerebrovascular accident
| Nociceptive pain | 5–84% |
| Shoulder pain | 30–40% |
| Chronic pain | 11–55% |
| Central neuropathic pain | 2–35% |
| Central post-stroke pain | 8–46% |
Widar et al. 2002; Jönsson et al. 2006; Lindgren et al. 2007; Kong and Woon 2004; Appelros 2006; Kumar and Soni 2009.
Table 29.2 Incidence of different types of pain in Parkinson’s disease
| Nociceptive pain | 40–70% |
| Non-dystonic | 37.8% |
| Dystonic | 6.7–40% |
| Peripheral neuropathic pain | 10% |
| Central neuropathic pain | 4.5–30% |
DeFazio and Tinazzi 2009; Beiske et al. 2009; Djaldetti et al. 2004.
Table 29.3 Incidence of different types of pain in multiple sclerosis
| Nociceptive pain | 21% (1% related to spasticity) |
| Peripheral neuropathic pain | 1% |
| Central neuropathic pain | 27.5% (87% in LL: 31% in UL) |
| Abnormal response to pain and temperature | 98% |
How do I assess pain?
Pain in the neurologically impaired patient is often complex and should be assessed thoroughly. The following offers a brief overview of the main considerations when assessing pain. Initially the therapist should try to establish the type of pain experienced by the patient, remembering that this population often have pain of more than one type.
Subjective assessment
Enquiry related to the following is required:
Area of pain
Identify a location of pain from the patient. This will help to start focusing in on certain structures. However, referred pain away from the origin of the pain means that this information cannot be relied upon completely. If pain reports are diffuse and widespread, the therapist may start to suspect central neuropathic or psychogenic pain.
Behaviour of the symptoms
This questioning should include the pattern of pain in terms of the 24-hour day and any factors that increase or decrease their symptoms (known as aggravating and easing factors). The acronym SIN is often used to represent other information that should be sought:
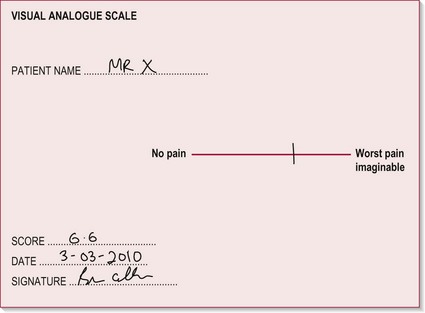
Visual analogue scale (VAS)
The VAS allows the therapist to assess the patient’s perception of their pain in terms of intensity/severity. This test can be carried out on initial assessment and during follow-up treatments to monitor the level of pain in relation to the effectiveness of interventions.
Therapist
The Neuropathic Pain Symptom Inventory (NPSI)
The Neuropathic Pain Symptom Inventory (NPSI) (Bouhassira et al. 2004) presents a broad overview of central neuropathic pain symptoms and may be useful to use as a reference source to assist the novice practitioner in identifying the signs of central neuropathic pain from the subjective assessment. Although it is often possible to diagnose neuropathic pain on the basis of history, a clinical examination is also essential (Treede et al. 2008).
Objective assessment
Observation
As the perception of pain is subjective, the therapist should also look for other signs and behaviours which may indicate pain. This process can be ongoing from the time the patient enters the department until the time they leave:
Distribution of pain and other sensations
Differential diagnosis of nociceptive pain
At this stage the therapist may have made a reasoned decision as to the type of pain (nociceptive, peripheral or central neuropathic) being experienced by the patient.
If the patient’s pain is considered to be of nociceptive origin, a comparison of the findings from AROM (S3.23) and PROM (S3.23) and special clinical tests will allow the therapist to differentiate the structures implicated. In each case, the therapist is evaluating the reproduction of the patient’s pain in order to identify the source. Further comprehensive detail guiding differential diagnosis can be found from a range of musculoskeletal based texts.
Recording
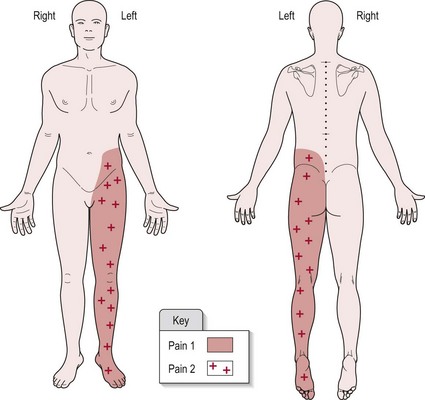
A separate section in your subjective write up should be headed ‘Pain’, with sub-headings as suggested in the above text. For the objective findings, a body chart showing the distribution of pain is a clear and easy way to present the information (Fig. 29.3). Use different symbols to highlight the various symptoms. The visual analogue scale (Fig. 29.2) should be included and the numeric result recorded in your notes.
Example
Patient diagnosed with CVA affecting the right parietal lobe (medially).
Onset of pain was 6/52 (6 weeks) post-CVA. Pain 1 is initiated by light touch and pain 2 relates to increased sensitivity to pin prick testing. Figure 29.3 shows the distribution of pain 1 and 2. There appears to be no discernible pattern to the pain behaviour. Pain is spontaneous and unaffected by movement or posture.
Analysis
Based upon the patient’s diagnosis and findings from the pain assessment the therapist should be able to use clinical reasoning skills to identify the type of pain, discriminating between nociceptive or neuropathic origin. This is vital, as understanding the pathological processes involved will facilitate more effective and focused treatment.
As central neuropathic pain is traditionally treated with medication, a referral to the medical team is indicated if the symptoms are not already being managed. Cognitive/behavioural therapy and mirror therapy are also options which the therapist may consider.
For nociceptive pain, the therapist should also aim to identify the structure responsible for the nociceptive activation in order to focus treatment appropriately. This requires a reasoning process involving knowledge of anatomy, the possible mechanisms of injury related to different structures and the physiological processes involved in healing. For further guidance, the reader is referred to Grieve (2006). Remember that the source of pain may also be visceral in nature.
In the example above, the patient presents with hyperalgesia and allodynia involving the left lower limb. As no pattern of pain behaviour is evident and the location reflects the area of somatosensory cortex damage, this is assumed to be central neuropathic pain.
References and Further Reading
Appelros P. Prevalence and predictors of pain and fatigue after stroke: a population-based study. International Journal of Rehabilitation Research. 2006;29:329-333.
Attal N, Fermanian C, Fermanian J, et al. Neuropathic pain: are there distinct subtypes depending on the aetiology or anatomical lesion? Pain. 2008;138:343-353.
Beiske AG, Loge JH, Ronningen A, et al. Pain in Parkinson’s disease: the hidden epidemic. Pain. 2009;141:173-177.
Bouhassira D, Attal N, Fermanian J, et al. Development and validation of the neuropathic pain symptom inventory. Pain. 2004;108:248-257.
Day R, Fox J, Paul-Taylor G. Neuro-musculoskeletal clinical tests: a clinician’s guide. Edinburgh: Churchill Livingstone/Elsevier; 2009.
DeFazio G, Tinazzi M. Central pain and Parkinson’s disease. Archives of Neurology. 2009;66:282-283.
Djaldetti R, Shifrin A, Rogowski Z, et al. Quantitative measurement of pain sensation in patients with Parkinson’s disease. Neurology. 2004;62:2171-2175.
Dworkin RH, Backonja M, Rowbotham MC, et al. Advances in neuropathic pain: diagnosis mechanisms and treatment recommendations. Archives of Neurology. 2003;60:1524-1534.
Ehde DM, Gibbons LE, Chwastiak L, et al. Chronic pain in a large community sample of persons with multiple sclerosis. Multiple Sclerosis. 2003;9:605-611.
Finnerup NB. A review of central neuropathic pain states. Current Opinion in Anaesthesiology. 2008;21:586-589.
Grieve DJ. Orthopaedic physical assessment, ed 4. Canada: Saunders Elsevier; 2006.
Henon H. Pain after stroke: a neglected issue. Journal of Neurology Neurosurgery and Psychiatry. 2006;77:569.
IASP, International Association for the Study of Pain. wwwiasp-painorg/AM/Templatecfm?Section=Pain_DefinitionsandTemplate=/CM/HTMLDisplaycfmandContentID=1728, 2007.
Jönsson AC, Lindgren I, Hallstrom B, et al. Prevalence and intensity of pain after stroke: a population based study focusing on patients’ perspectives. Journal of Neurology, Neurosurgery and Psychiatry. 2006;77:590-595.
Juri C, Rodriguez-Oroz M, Obeso JA. The pathophysiological basis of sensory disturbances in Parkinson’s disease. Journal of Neurologic Science. 2009;289:60-65.
Kandel ER, Schwartz JH, Jessell TM. Principles of neural science, ed 4. New York: McGraw-Hill; 2000.
Kong KH, Woon VC, Yang SY. Prevalence of chronic pain and its impact on health-related quality of life in stroke survivors. Archives of Physical Medicine and Rehabilitation. 2004;85:35-40.
Kumar G, Soni CR. Central post-stroke pain: Current evidence. Journal of Neurologic Science. 2009;284:10-17.
Lindgren I, Jönsson AC, Norrving B, et al. Shoulder pain after stroke: a prospective population-based study. Stroke. 2007;38:343-348.
Loeser JD, Treede RD. The Kyoto protocol of IASP basic pain terminology. Pain. 2008;137:473-477.
Osterberg A, Boivie J, Thuomas K-A. Central pain in multiple sclerosis: prevalence and clinical characteristics. European Journal of Pain. 2005;9:531-542.
Svendsen KB, Jensen TS, Hansen HJ, et al. Sensory function and quality of life in patients with multiple sclerosis and pain. Pain. 2005;114:473-481.
Treede RD, Jensen TS, Campbell JN, et al. Neuropathic pain: redefinition and a grading system for clinical and research purposes. Neurology. 2008;7018:1630-1635.
Widar M, Samuelsson L, Karlsson-Tivenius S, et al. Long-term pain conditions after a stroke. Journal of Rehabilitation Medicine. 2002;34:165-170.
Woolfe CJ, Mannion RJ. Neuropathic pain: aetiology, symptoms, mechanisms and management. The Lancet. 1999;353:1959-1964.