CHAPTER 23 Sensation
What is sensation?
Sensation is complex, consisting of several modalities, namely the four special senses (vision, hearing, smell and taste) and our somatosensory senses (tactile (light touch and pressure), proprioception, temperature and nociception/pain). Our sensory experience is a construct of the brain, which perceives and attaches meaning to these sensory modalities. Although all these modalities are relevant to function and our quality of life, only the somatosensory senses are covered in this section. Somatosensory information allows us to experience the environment, warns us of potential danger and is fundamental in maintaining arousal and the control of movement.
Sensory receptors
The somatosensory system reacts to diverse stimuli from different sensory receptors, thermoreceptors (temperature), mechanoreceptors (physical distortion) and chemoreceptors (chemicals). Each sensory modality has a unique sensory receptor which is activated when a specific stimulus, within its receptive field, reaches a threshold intensity. This receptive field may be altered as a result of pathology when it can contribute to the experience of pain (S3.29) and abnormal sensation. The density of receptors is relevant to the ability to locate a stimulus (awareness) and to distinguish between two stimuli (discrimination).
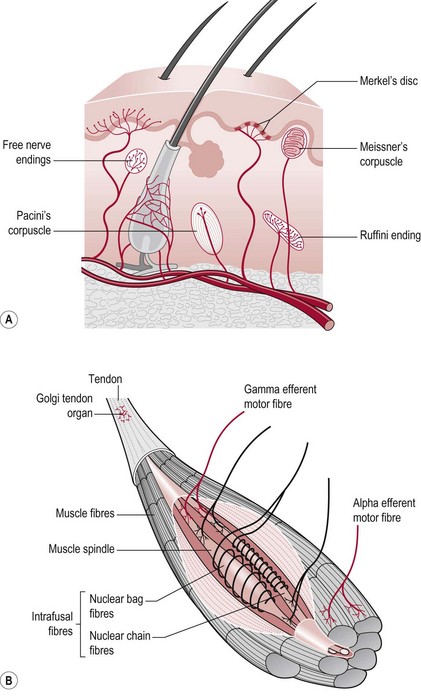
The duration of a sensory experience is related to a further characteristic of the sensory receptor termed ‘adaptation’. A rapidly adapting receptor responds to change and is therefore activated at the onset and termination of the stimulus only. A slowly adapting receptor responds continuously to a persistent stimulus. This should be borne in mind when assessing particular modalities. Figure 23.1 shows the main somatosensory receptors and Table 23.1 identifies the functional characteristics of these receptors.
Sensory pathway
This afferent information from sensory receptors in the periphery is conducted along afferent neurons to the spinal cord (S2.13) and transmitted via the ascending tracts (S2.15) to the thalamus (S2.9), brain stem (S2.10) and higher centres (S2.7). Sensory input is systematically mapped on the contralateral primary somatosensory cortex (parietal lobe), then processed and integrated with other relevant information by the somatosensory association area (parietal lobe), basal ganglia (S2.11) and cerebellum (S2.12) before being acted upon.
![]() Clinical hints and tips
Clinical hints and tips
Functionally relevant information is often transmitted in the same anatomical ascending tract (S2.15) but in separate parallel pathways. For example, when holding an object, the information related to texture (light touch) and shape (proprioception) are both conveyed in the dorsal columns. This overlap in function may be important clinically as following a lesion, the adjacent preserved tracts may be able to assist in the lost function.
Sensory modalities
Table 23.2 shows a summary of the functional anatomy related to the modalities described below.
Tactile
In terms of touch a hierarchy of sensory function was proposed by Fess (1990). The lowest level is the ability to identify a single stimulus (awareness). The hierarchy proceeds with the ability to distinguish between two stimuli (discrimination), the ability to differentiate different characteristics or strength of one stimulus (quantification) and the highest level is the ability to recognize objects by touch alone (recognition). This hierarchy suggests that if the higher levels are intact, the lower levels do not need to be assessed.
Light touch
The sensation of light touch allows us to be aware of tactile stimuli, including situations as innocuous as the clothes we wear, but also has a role in warning us of impending damage. Following a neurological lesion, a patient may become hypersensitive to touch, when even clothes may initiate a sensory experience and an inappropriate motor response. This altered sensation may also be perceived as pain, paraesthesia or dysaesthesia (S3.29). In a scenario where a patient becomes hyposensitive to light touch, the lack of awareness of limbs particularly, may result in soft tissue injury.
Pressure/deep touch
As for light touch, our sense of pressure is important in avoiding tissue damage, performing functional activities such as grip and weight bearing activities but is also likely to contribute to proprioception.
Two-point discrimination
This is the ability to identify two objects in contact with the skin as two distinct points, rather than one. Lesions of the sensory cortex may result in difficulty discriminating two points within the normal distances (finger tips 2–4 mm; palm 8–15 mm; back 30–40 mm) and this increased two-point discrimination has been found to be a poor prognostic indicator for recovery in cerebrovascular accident (CVA).
Nociception
Pain (S3.29)
Pain serves to notify the nervous system about tissue damage that has occurred. Although pain can be elicited without tissue damage (neurogenic pain) (S3.29), nociceptive pain will remain the focus here. The sensory receptors sensitive to nociceptive stimuli are free nerve endings, however not all noxious stimuli is perceived as pain because the response to a stimuli is highly subjective. The free nerve endings are much less specialized than other receptors and may respond to mechanical, thermal or chemical stimuli.
Temperature
Our sense of skin temperature is protective in avoiding tissue damage and is anatomically and functionally closely related to pain.
Proprioception
Our sense of proprioception is complex and results from integration of information from several sources, visual (S3.10, S3.27), vestibular (S2.10) and somatosensory systems. In this text, proprioception is defined as consisting of three sub-modalities (Riemann and Lephart 2002a, b); the ability to sense:
Proprioception also governs the ability to recognize an object using the sense of touch through manipulation (stereognosis). This sense is mediated via the dorsal columns, however the ability to recognize any object requires integration by the higher centres with other sensory information and in relation to past experience (memory).
Historically, joint receptors have been considered to have an important role in proprioception, however recent studies have shown that this is not the case in most joints (Proske and Gandevia 2009). In fact, the major sensory receptors involved in position and movement sense are muscle spindles and stretch receptors in the skin (Ruffini endings). The skin receptors have a greater role in circumstances where the controlling muscle is distant to the joint being moved (e.g. fingers) (Collins et al. 2005) and in the case of two joint muscles (Sturnieks et al. 2007). Ruffini endings have been found to be accurate within 2° in relation to change of joint angle in the fingers and deficits contribute to altered timing and force of grip in stroke (Blennerhassett et al. 2007).
Why do I need to assess sensation?
A lesion of the somatosensory system is likely to impact greatly on the neurologically impaired patient. For example:
The incidence of sensory deficit is also high in this patient population. In Parkinson’s disease (PD), there is a growing body of evidence that suggests that non-motor symptoms such as abnormal sensation may actually precede the motor deficits by which it is presently diagnosed. Between 50% and 100% of people with PD have sensory deficits (Rahman et al. 2008) with a decrease in two-point discrimination and proprioception highlighted (Zia et al. 2000). This is attributed to an impairment of sensorimotor processing by the basal ganglia (S2.11) (Juri et al. 2009).
In CVA and multiple sclerosis (MS) the lesion may directly involve the somatosensory system or the processing centres and therefore may result in a dysfunction of any of the somatosensory modalities. The incidence is up to 60% in CVA (Schabrun and Hillier 2009) with tactile deficits most common and considered a poor prognostic indicator for recovery (Tyson et al. 2008).
How do I assess sensation?
The primary modalities that should be part of a neurological assessment include light touch, deep touch/pressure, temperature, pain and proprioception (Cassiopeia and Okun 2002). In order to save time, clinicians may opt to assess pain or temperature (anterior spinothalamic tract), light touch or deep touch (dorsal columns) and proprioception.
The patient may report that they are aware of an area of numbness or altered sensation during the subjective assessment. This can then be explored during the objective assessment. Although the following procedures are subjective in nature, they do reflect current clinical practice.
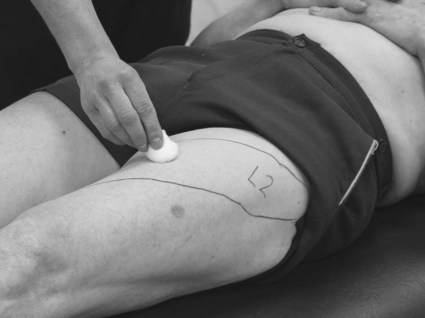
Tactile – light touch
Patient
Position the patient in a comfortable supported position suitably undressed to access as much skin as possible, while maintaining dignity.
Therapist
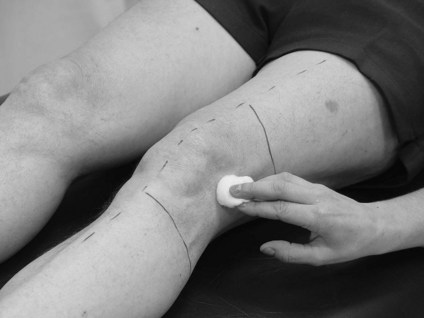
Two-point discrimination
A specialist tool provided with the Rivermead Assessment of Somatosensory Performance (Fig. 23.5) provides a standardized tool for testing, however if this is unavailable, two paper clips can be used.
Tactile – deep touch/pressure
The therapist should follow the assessment procedure as for light touch but should use the blunt end of a neurotip as the instrument for producing the stimulus of pressure (Fig. 23.6) or a pressure pen, which gives a better standardization of application (Fig. 23.6). As the Pacinian corpuscles are rapid adapting receptors the neurotip should be placed on and off rather than be applied continuously.
Pain (pin prick)
The therapist should follow the assessment procedure as for light touch but should use the sharp end of a neurotip as the instrument for producing the stimulus of pain (Fig. 23.7). Note: Be very careful not to pierce the skin! Using the tip at a 45° angle is advised.
Sharp and blunt
This technique assesses the ability of quantification related to different characteristics of pain and is carried out using the sharp and blunt end of a neurotip (Figs 23.6, 23.7).
Temperature (heat and non-noxious cold)
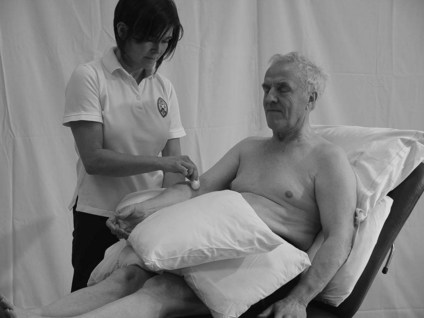
The therapist should follow the assessment procedure as for light touch but should use thermo controlled test tubes, one cold (6–10°C) and one hot (44–48°C) as the instruments for producing the stimulus of heat and non-noxious cold (Fig. 23.8). This procedure should test for temperature awareness of both heat and cold separately and also the ability to discriminate between the two (is this hot or cold?).
Proprioception
There are a few methods by which proprioception can be assessed. As traditionally these techniques are carried out with the patients eyes closed, excluding our sense of vision, a more accurate description of what is being assessed would be joint position sense and movement sense.
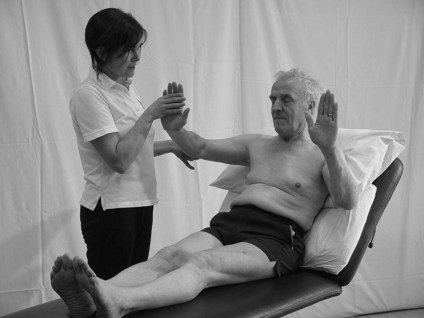
Mirroring
Therapist
![]() Clinical hints and tips
Clinical hints and tips
For patients with a primarily unilateral dysfunction such as CVA, the therapist should place the affected limb into the position to be copied. This allows the patient an attempt to copy using the unaffected limb, which has no motor dysfunction.
However, there are several shortcomings with this method:
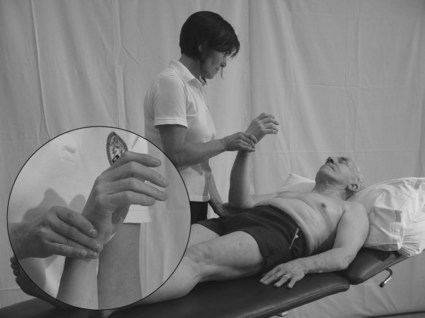
Specific joint position sense and movement sense testing
Therapist
![]() Clinical hints and tips
Clinical hints and tips
Hand hold – Whenever possible, the therapist should attempt to (a) position the patient so that the movement being assessed is gravity eliminated and (b) use a hand grip at the sides of the joint (Fig. 23.9). This will minimize the cues given to the patient through handling, making the findings more accurate.
Recording
A body chart provides a clear and time efficient way of representing most of the sensory findings:
It is usual to record only the existing deficits, which are marked on the corresponding position of the body chart. A key indicating the symbols used to identify different modalities is advisable. Awareness, discrimination and quantification can be added as appropriate, as long as the chart remains legible. See the body chart for pain (S3.29) as an example.
Recognition by graphesthesia and stereognosis can be recorded as intact, impaired or absent. Of course a text description of the above is also perfectly acceptable.
Example
Sensory testing
Light touch/pressure and temperature/pain are fully intact.
Gross positional and movement sense (proprioception) is intact throughout, however fine positional sense is reduced in left hand and two-point discrimination is increased to 8–10 mm on the left finger pads. Stereognosis is poor with left hand.
Analysis
Sensory deficits may have wide ranging implications for the neurologically impaired patient from potential soft tissue damage because of lack of awareness, poor balance and falls and reduced fine finger function. Therefore, it is important for the therapist to understand the specific sensory deficit of the patient and the implications it may be having for movement and function. There is also some evidence to support the success of sensory retraining following CVA (Schabrun and Hillier 2009) and therefore accuracy in assessment is paramount.
From the above example, reduced finger joint position sense and two-point discrimination of the left hand may result in reduced fine finger function and grip. This will have greater implications if the patient is left handed. The poor stereognosis may be a reflection of the former, rather than a sensory integration problem but this would have to be explored further.
Outcome measures
Research
References and Further Reading
Blennerhassett JM, Matyas TA, Carey LM. Impaired discrimination of surface friction contributes to pinch grip deficit after stroke. Neurorehabilitation and Neural Repair. 2007;213:263-272.
Bromley I. Tetraplegia and paraplegia: a guide for physiotherapists, ed 6. Edinburgh: Churchill Livingstone; 2006.
Busse M, Tyson SF. How many body locations need to be tested when assessing sensation after stroke? An investigation of redundancy in the Rivermead Assessment of Somatosensory Performance. Clinical Rehabilitation. 2009;231:91-95.
Campbell M. Rehabilitation for traumatic brain injury: physical therapy practice in context. Edinburgh: Churchill Livingstone; 2000.
Cassiopeia F, Okun MS. Origins of the sensory examination in neurology. Seminars in Neurology. 2002;22:399-408.
Collins DF, Refshauge KM, Todd G, et al. Cutaneous receptors contribute to kinesthesia at the index finger elbow and knee. Journal of Neurophysiology. 2005;94:1699-1706.
Fang Y, Chen X, Li H, et al. A study on additional early physiotherapy after stroke and factors affecting functional recovery. Clinical Rehabilitation. 2003;176:608-617.
Fess EE. Assessment of the upper extremity: instrumentation criteria. Occupational Therapy Practice. 1990;1:1-11.
Forsaa EB, Larsen JP, Wentzel-Larsen T, et al. Predictors and course of health related quality of life in Parkinson’s disease. Movement Disorders. 2008;23:1420-1427.
Juri C, Rodriguez-Oroz M, Obeso JA. The pathophysiological basis of sensory disturbances in Parkinson’s disease. Journal of Neurologic Science. 2009;289:60-65.
Kandel ER, Schwartz JH, Jessell TM. Principles of neural science, ed 4. New York: McGraw-Hill; 2000.
Proske U, Gandevia SC. The kinesthetic senses. Journal of Physiology. 2009;587:4139-4146.
Rahman S, Griffin HJ, Quinn NP. Quality of life in Parkinson’s disease: the relative importance of the symptoms. Movement Disorders. 2008;23:1428-1434.
Riemann BL, Lephart SM. The sensorimotor system, part I: the physiologic basis of functional joint stability. Journal of Athletic Training. 2002;37:71-79.
Riemann BL, Lephart SM. The sensorimotor system, part II: the role of proprioception in motor control and functional joint stability. Journal of Athletic Training. 2002;37:80-84.
Schabrun SM, Hillier S. Evidence for the retraining of sensation after stroke: a systematic review. Clinical Rehabilitation. 2009;231:27-39.
Smith JL, Crawford M, Proske U, et al. Signals of motor command bias joint position sense in the presence of feedback from proprioceptors. Journal of Applied Physiology. 2009;106:950-958.
Sturnieks DL, Wright JR, Fitzpatrick RC. Detection of simultaneous movement at two human arm joints. Journal of Physiology. 2007;585:833-842.
Tyson SF, Hanley M, Chillala J, et al. Sensory loss in hospital-admitted people with stroke: characteristics associated factors and relationship with function. Neurorehabilitation and Neural Repair. 2008;222:166-172.
Widerstrom-Noga EG, Duncan R, Felipe-Cuervo E, et al. Assessment of the impact of pain and impairments associated with spinal cord injuries. Archives of Physical Medicine and Rehabilitation. 2002;83:395-404.
Winward CE, Halligan PW, Wade DT. The Rivermead Assessment of Somatosensory Performance RASP: standardization and reliability data. Clinical Rehabilitation. 2002;16:523-533.
Zia S, Cody F, O’Boyle D. Joint position sense is impaired in Parkinson’s disease. Annals of Neurology. 2000;47:218-228.