CHAPTER 7 Breast
Clinical aspects
Although most countries in Europe continue to perform fine needle aspiration biopsy (FNB) as their first choice in the investigation of breast lesions in both screening and symptomatic populations, the use of core needle biopsy (CNB) is increasing.1 Centers that continue to use FNB have utilized a multidiscipinary approach to the diagnosis of breast lesions.2 A significant advantage of FNB is the low cost and the ability to render a diagnosis to the clinician and patient at the time of the procedure thus allowing treatment decisions to be made immediately. The presence of a cytopathologist with expertise in FNB provides superior diagnostic results and low inadequate rates.3 However, the inability of FNB to definitely diagnose invasion and the preference of pathologists who are not trained in cytopathology to interpret histologic samples rather than cytologic samples have contributed to the increase in the use of core biopsy. The initial investigation of breast lesions by core biopsy started with the 14-gauge spring-loaded cutting core needle guided by ultrasound or by stereotaxis. Now, many institutions use a vacuum-assisted larger-bore (8 to 11-gauge) cutting core needle (VACB), for the investigation of microcalcification, mass lesions and architectural distortion. For a detailed description of the techniques see Wong et al.4 Recent evidence suggests that the vacuum assisted core biopsy (11 gauge to 8 gauge) has better sensitivity and specificity than the 14-gauge core biopsy or FNB for architectural distortion or microcalcification and that the 14-gauge core biopsy provides better sensitivity and specificity than FNB for other lesions.5 Besides the ability to give an unequivocal diagnosis of invasion, other advantages of core biopsy include a higher proportion of definitive malignant diagnoses, more type-specific diagnoses, and a more specific diagnosis of benign lesions.6-10 The technique can be a substitute for open biopsy for the diagnosis of benign lesions and in the preoperative planning of surgery in malignant lesions. In some multidisciplinary assessment centers in Australia, CNB is the only diagnostic method now used, whereas in others FNB is used alone or in combination with CNB. Some countries have developed clear guidelines for the use of FNB and CNB, with core biopsy used in cases with discrepant cytologic and radiologic findings, inconclusive FNB or in cases of microcalcification.1
We consider breast FNB and CNB to be complementary techniques, and both should be available in any modern multidisciplinary clinic. Both are sampling methods and have their advantages, limitations and specific indications.11. For example, radiologically low-grade microcalcifications may be better investigated by VACB than by FNB. On the other hand, most high-grade DCIS are malignant by FNB, and CNB is not significantly more reliable than FNB to exclude focal invasion in these lesions. CNB is of great value both in nonpalpable and palpable lesions where initial FNB is suspicious but not diagnostic of malignancy, as is often the case in low-grade cancers, particularly in tubular carcinoma and invasive lobular carcinoma.12 Some clinical problems, such as opacities occurring postoperatively, are better diagnosed by CNB, as FNB cannot definitely diagnose scar tissue.
Cost factors must also be taken into account, given the large volume of work generated by breast cancer screening.13 The instrumentation and disposables used for CNB and particularly for VACB are much more expensive than for FNB. This must be weighed against the cost of surgical open biopsy.8 Approximately two-thirds of screen-detected cancers can be confidently diagnosed by FNB. This means a considerable cost saving compared to CNB if CNB is used for all patients, even if a number of patients may have to undergo both tests. Ultrasound (US)-guided FNB is rapid and usually causes little discomfort to the patient. In the ‘assessment center’ setting, where the pathologist is on site, FNB smears can be checked immediately for adequacy. An on-the-spot diagnosis can be given to decide on management and to advise the patient without further delay.
The challenge now is to integrate the use of the alternative techniques for the greatest benefit to the patient and for maximal cost effectiveness.
Palpable lesions
A palpable breast lump is a common clinical problem that presents to surgeons, gynecologists and general practitioners. While excisional biopsy was the accepted practice in the past, current practices utilize radiological imaging in combination with needle biopsy, reducing the need for unnecessary surgical excision of benign breast lesions.
A preoperative diagnosis offers several advantages:
The investigation of palpable breast lumps in successful breast programs utilizes a multidisciplinary approach that centers around the ‘triple test’, analyzing clinical and radiologic findings in conjunction with the pathologic features to diagnose the lesion and determine the best treatment plan for the patient.1 There has been a dramatic increase in general practitioner (GP) referral of patients with breast lumps for FNB as a first-line investigation in the work-up of these lesions. Given the lower clinical expertise of the GP compared to a surgeon, this places a unique responsibility on the cytopathologist. The cytopathologist must use his or her clinical acumen in addition to the cytological findings in recommending the need for further investigations such as radiological assessment, follow-up, referral to a specialist surgeon, or local excision. While some surgeons may continue to perform CNB of palpable lesions in their office, most lesions today are biopsied under radiologic guidance.
The trend towards more conservative surgery and individualized treatment has increased the importance of close correlation of clinical, radiological, and pathologic findings. Material obtained through preoperative biopsy (through cell block material from FNB or by CNB) is increasingly being used for hormone receptor analysis and for the evaluation of other prognostic parameters by various ancillary techniques. The ctology of palpable nodules in irradiated breast tissue has become a more frequently encountered problem as a result of conservative treatment of cancer by lumpectomy followed by irradiation. FNB is also useful in the evaluation of skin or chest wall lesions in patients post mastectomy.
The place of FNB and CNB in the investigative sequence
While FNB of a palpable breast lump should generally be preceded by mammographic and/or ultrasonographic examination, as the radiological findings help to select the most appropriate area to be biopsied, FNB may be performed as the first-line investigation, especially in symptomatic and screening populations.14,15. Caution should be exercised that if FNB is done first, post-biopsy hemorrhage may interfere with the interpretation of the films. The referring doctor normally receives a written report of the cytological findings the following day, with a phone call placed to the referring physician immediately, if necessary. Further steps can then be discussed with the patient and this will relieve anxiety and avoid unnecessary delay. If the decision to perform CNB of a palpable abnormality is made, the majority will be performed with radiographic guidance, either stereotaxically or, more commonly, with ultrasound guidance.
The main purpose of FNB or CNB of breast lumps is to confirm cancer preoperatively and to avoid unnecessary surgery in specific benign conditions.
The role of FNB in the assessment of a breast lump includes:
The role of CNB in the assessment of breast lumps is similar to FNB with the exception that it is not routinely used in the diagnosis of simple cysts.
FNB and CNB in the follow-up of breast cancer
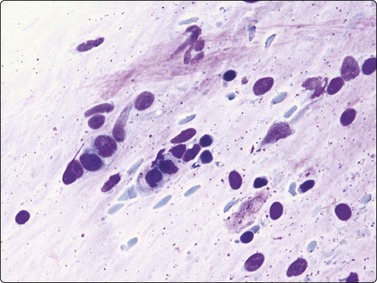
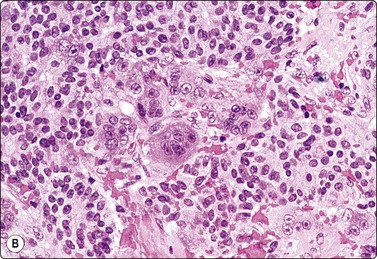
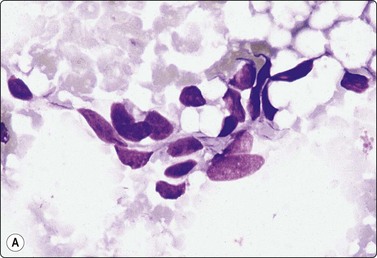
The FNB diagnosis of local recurrence of breast cancer is generally straightforward, involving a distinction between cancer and suture granuloma, fat necrosis and scarring.16-19 However, reactive atypia in reparative granulation tissue, in fat necrosis or in seroma cavities can cause diagnostic problems, and radiation-induced atypia in benign glandular epithelium can be misinterpreted as recurrent malignancy (Fig. 7.1). CNB can also be used; however, fat necrosis may be misinterpreted as invasive carcinoma on core, leading to a false-positive diagnosis of malignancy.20 Sampling error by core biopsy should always be considered in cases where the suspicion of recurrence is high.
Accuracy of diagnosis in FNB and CNB
As noted above, a significant advantage of CNB is the ability to diagnose invasiveness and thus allow the patient to undergo a single operation including sentinel node biopsy in cases diagnosed as invasive. However, in our experience the presence of malignant cells on FNB in a palpable mass yields invasive carcinoma at excision in approximately 98% and thus the addition of a CNB only adds additional information in a few cases.21 It must also be recognized that a CNB diagnosis of ductal carcinoma in situ will be upgraded to a diagnosis of invasive carcinoma at surgical excision in approximately 20% of cases.22 Core biopsy is favored in lesions that appear fibrotic or collagenous and in cases suspected to be invasive lobular carcinoma, as these lesions can be paucicellular on FNB.23
The reported sensitivities, specificities and positive and negative predictive values for FNB vary depending on how insufficient samples are considered (as positive, negative or excluded) and how atypical samples are categorized (positive or negative).24 When insufficent samples and atypical and benign findings are presumed to be negative, sensitivities range from 43.8% to 95%, specificities from 89.8% to 100%, positive predicitive values from 76.2% to 100% and negative predicitive values from 46.3% to 98.8%. If insufficient samples are excluded, sensitivities and specificities improve to a range of 58.3% to 100% and 55% to 100%, respectively, with a slight change in the negative predictive value to between 46.6% and 98.6%. The variability in reported sensitivities and specificities depends on the expertise and skill of the aspirator and of the interpreter. The aim should be a sensitivity of no less than 95% and this can be achieved with increasing experience. Sensitivity is lower for low-grade carcinomas (invasive and in situ), for lobular carcinoma, and for very small and very large cancers.25,26 The positive predictive value of a malignant diagnosis is approximately 99%, and, although rare, occasional false-positive diagnoses of malignancy are recorded in most series.27-30
The results of studies comparing FNB to CNB are difficult to evaluate due to numerous variables that influence accuracy. Skill and experience are important in performing the biopsy and in the preparation and reading of the smear. Although the technique is simple, training and continuous practice are essential to acquire and to maintain skill. Significant improvement in accuracy occurs with experience. Centers with experienced physicians performing and interpreting FNBs have shown a high diagnostic accuracy. In general, the best results are achieved if the pathologist who reads the smears either performs the biopsies or works directly with the surgeon and radiologist in a multidisciplinary team.31 The reason for this is that the microscopic examination, immediate or with some delay, provides the pathologist a feedback on the adequacy and quality of the smears causing repeat sampling and the ability to obtain additional diagnostic material, if necessary. For core biopsy, training and skill in the technique and the number and size of the cores will influence accuracy.32,33 Some studies have shown CNB to be more accurate than FNB in the diagnosis of nonpalpable breast lesions and in the diagnosis of LCIS and infiltrating lobular carcinoma.34-36 Other studies have shown CNB and FNB to be similar in sensitivity, positive predictive value and inadequate rate, with only statistical differences in specificity.37 Studies have shown FNB to be more accurate than CNB in distingushing benign from malignant papillary lesions38 and in diagnosing malignancy in palpable breast cancers.39 In one study, tumor size influenced accuracy, with tumors less than 2 cm and greater than 5 cm showing similar accuracies by CNB and FNB. However, with tumors between 2 cm and 5 cm in size, CNB showed a higher sensitivity and a lower inadequate rate as compared to FNB.40 Some studies have shown the inadequate rate to be much higher with FNB as compared to CNB;23 however, the presence of a cytopathologist at the procedure noticeably lowers inadequacy rates.3 For core biopsies, specificities range from 99% to 100%, sensitivities from 94% to 99%, negative predictive values from 94% to 99% and positive predictive values from 94% to 100%.24
A positive cytological diagnosis is possible in 75–80% of palpable cancers. Definitive treatment can often be based on the cytological diagnosis without the need for histological confirmation in centers with a large volume of cases and specialty trained cytopathologists unless there is disagreement between cytology and clinical and/or mammographic assessment. Approximately 98% of palpable masses with unequivocally malignant FNB cytology are invasive cancers, the remaining few are high-grade DCIS.21 Studies that compared FNB to CNB for palpable breast carcinoma showed a higher sensitivity (97.5% vs. 90%) for FNB regardless of the size of the tumor, its subtype or its degree of differentiation.39 In situ carcinoma constitutes a special problem.41 While high-grade/large cell DCIS (classic comedocarcinoma) is easily recognized as malignant in smears, low-grade DCIS may be impossible to distinguish from atypical but benign epithelial proliferations. Smears from low-grade DCIS are usually reported as atypical or suspicious with a recommendation for CNB or open biopsy. Furthermore, cytological examination on its own can not definitely diagnose invasion. Mammographic and US findings of a tissue density in most cases reflect invasive growth to be confirmed by CNB, while characteristic microcalcifications in the absence of a mass lesion suggests DCIS. However, focal invasion in a predominantly in situ carcinoma can only be assessed by histologic examination of the excised lesion. If present, a second-stage extended surgery usually follows. In some cases, additional information about the presence of invasion and the exact tumor type may be required to decide surgical management. This may be provided by CNB or by intraoperative frozen section. The major limitation of core biopsy is sampling error. As noted above, the finding of in situ ductal carcinoma on CNB does not rule out the chance of finding invasive disease at surgical excision. The presence of other benign and high-risk lesions such as radial scars, papillary lesions, atypical ductal hyperplasia, atypical lobular hyperplasia and lobular carcinoma in situ on CNB have been shown to underestimate the presence of carcinoma at surgical excision.
Cytologic categories for the reporting of breast FNB are described in Table 7.1. Indecisive cytology reports such as ‘suspicious of malignancy’ or ‘cytological atypia not diagnostic of malignancy’ are neither false-negative nor false-positive diagnoses and should be understood as expressing the need for CNB or open biopsy. A positive report should never be issued if smears are quantitatively or qualitatively unsatisfactory or if there is the slightest doubt of malignancy. Such a policy inevitably results in a number of indecisive reports inversely proportional to experience. Rare false-positive FNB diagnoses occur even in centers of excellence.29 In our experience, the greatest risk of making a false-positive diagnosis is in cases of fibroadenoma with prominent epithelial atypia, papillary lesions with or without infarction, radial scars,30 apocrine adenosis42 tubular adenomas43 and sclerosing adenosis.42 False-positive diagnosis by CNB is unusual but can be seen in fat necrosis,20 complex sclerosing lesions, sclerosing adenosis and adenomyoepitheliomas.44 The rare occurrence of false-positive diagnosis is an extremely important issue given the risk of overtreatment of a benign condition with adverse effects to the patient, and medicolegal consequences. A summary of diagnostic pitfalls is presented at the end of this chapter.
Table 7.1 Cytology reporting categories
| Cytological category | Description/definition |
|---|---|
| Malignant | Unequivocal diagnosis; can be used for definitive management if consistent with radiological findings and treatment protocols |
| Suspicious | Suggests malignancy; insufficient evidence for definitive management; histological confirmation needed |
| Atypical/indeterminate | Diagnosis uncertain; further investigation needed |
| Benign – specific diagnosis | Cyst; fibroadenoma; intramammary lymph node |
| Benign – non-specific | Smears of cells of non-neoplastic breast tissue. The number of cells required varies between operators. Not possible to confirm that the smear is representative, the responsibility stays entirely with the radiologist |
| Unsatisfactory sample | Fat and fibrous tissue onlyNo cellular material, or bloody samples with poorly preserved cells |
The false-negative rate for FNB is generally less than 5%, but significantly higher than the false-positive rate. It is mainly due to sampling error. Most false-negative FNB are due to low-grade carcinomas, such as lobular and tubular carcinomas and carcinomas with abundant sclerosis. Most false negatives can be attributed to a lack of cytologic atypia in well-differentiated lesions rather than misinterpretation.45 The false-negative rate for CNB is approximately 2%, mainly due to sampling error or missed targeting of a lesion.46 Ultrasound guidance has been shown to decrease the false-negative rate.47
The complete sensitivity of image-guided FNB is about 90%, compared with about 95% for direct FNB of palpable lesions, and the absolute sensitivity is 60–70% compared with 75–80% for direct FNB. The false-negative rate is 5–10%, higher than for palpable lesions.48-50
Most high-grade DCIS lesions are diagnosed as malignant at FNB. Most low-grade DCIS lesions on FNB show abnormal cell patterns requiring excision, but relatively few are given an unequivocal diagnosis of malignancy. DCIS lesions associated with dispersed ‘powdery’ microcalcifications are those most likely to be inadequately sampled. Our experience, and that of others, suggests that this type of lesion (mammographically 3B microcalcifications) is better examined by VACB since representative and diagnostic material is not easily obtained either by FNB or by conventional CNB. In a series of 124 cases of suspicious microcalcifications reported by Dahlstrom et al., no calcification was found in the CNB samples in one-quarter of the cases.51 The reason for this may be that microcalcifications often occur in soft, mainly fatty tissue offering little substance to the sampling needle, whereas the target is fixed by the vacuum applied by the mammotome. Symmans et al., however, found that stereotaxic FNB of benign microcalcifications had a high negative predictive value, higher than CNB.48
Approximately two-thirds of screen-detected cancers are given a definitive cancer diagnosis by FNB as part of triple diagnosis. The other one-third requires further investigation by CNB or open biopsy to give the go-ahead for more extensive definitive surgery. The reason may be discordance with radiological findings, technical difficulties to obtain satisfactory smears, doubts about invasion, or a relatively bland cytology as in low-grade cancers, especially tubular carcinoma and lobular carcinoma of classic type. The rate of atypical or inconclusive cytology is moderately higher from impalpable than from palpable lesions, and the proportion of cancers given an atypical/inconclusive report by image-guided FNB is higher, mainly due to the larger numbers of DCIS and small low-grade lesions.52 The percentage of cancers in excised lesions with indeterminate cytological diagnoses is lower for image-guided samples than for direct FNB.
It has been found that with experience the accuracy of ultrasound-guided FNB of opacities is comparable to that of stereotactic guidance. The complete sensitivity in the diagnosis of cancer is 90–95%.49,53 Ultrasound guidance increases accuracy for lesions under 2 cm. Since US guidance is faster and technically less demanding, it has become by far the most commonly used modality for biopsy guidance, except for microcalcifications without a tissue density, for lesions close to the chest wall, and for lesions that are not clearly seen by US. Alkuwari et al. reported a sensitivity of 65% in the ultrasound-guided FNB of metastatic breast carcinoma in nonpalpable axillary lymph nodes in breast cancer patients.
Can a cytology report54 of a benign breast lesion ever be regarded as diagnostic? If smears are satisfactory and adequate and cytological criteria are met, well-circumscribed lesions such as simple cysts, lipomas, most fibroadenomas, intramammary lymph nodes and fat necrosis can be diagnosed with confidence. Poorly circumscribed lesions – the common hormonal mastopathy-fibrocystic disease-fibroadenosis-mammary dysplasia – cannot be confidently diagnosed by FNB to the exclusion of malignancy. This is particularly true in the presence of epithelial hyperplasia, papillomatosis and adenosis. However, if multiple aspirates with a satisfactory yield of cells are obtained from different parts of the lesion and if the result is consistent with the clinical and mammographic evaluation, conservative management based on clinical follow-up is justified.
For core biopsy, a 1-year follow-up is recommended for definitive benign cases such as fibroadenoma where the imaging and pathologic findings are concordant. For other benign concordant diagnosis, 6-month follow-up is recommended.55
While false-negative diagnoses can occur, a missed cancer is rare if the triple test is used as the gold standard. Triple diagnosis, the combination of clinical examination, mammography and pathologic examination, and the multidiscplinary approach increase the quality of FNB and CB and decrease its diagnostic limitations. The use of all three modalities in parallel has led to further improvement in preoperative diagnosis.56 If all three investigations are in agreement that a lesion is either benign or malignan diagnostic accuracy is over 99%.57 This is based on the condition that the diagnosis by each modality was reached completely independently of the others. For example, radiological findings must not influence the initial cytological evaluation.
The unsatisfactory sample
When should an FNB sample of a breast lesion be regarded as unsatisfactory? What is the definition of a satisfactory sample? These questions have been discussed in many editorials and articles, and there are no simple answers.58,59 It depends very much on specific circumstances, for example if samples were taken by an experienced pathologist, or if smears were prepared by clinical staff and submitted to the laboratory. If the lesion sampled is a paucicellular fibrous mastopathy, a sclerosed fibroadenoma, a desmoplastic carcinoma, or hypertrophic adipose tissue, smears are naturally very low in cells. A hypocellular smear must be evaluated on the basis of clinical findings and consideration to the consistency of the tissue felt through the biopsy needle must be given. Poorly prepared smears with crush or drying artifacts or with cells trapped in clotted blood should be rejected as unsatisfactory. Attempts have been made to set quantitative criteria for the minimum cell yield that can be accepted as satisfactory.28,30 However, rigid criteria are unrealistic for the reasons mentioned above, and monitoring the laboratory’s own results on an ongoing basis is the best way of ensuring accuracy. Skill affects adequacy, and targeted training to suboptimal aspirators can help improve the quality of their FNB specimens.60
For lesions sampled by ultrasound or stereotactic FNB at least three and sometimes up to six passes are used, depending on whether the needle position is satisfactory, and depending on the quality of the smears. If multiple FNB passes from a radiologically indeterminate or suspicious lesion consistently produce a poor yield, CNB is more likely to provide a diagnosis. The significance of a scanty smear in the context of triple diagnosis can only be jointly decided by the assessment team. Overall, about 20% of samples will have scanty cell content. The figure is higher for microcalcifications. However, ‘unsatisfactory’ samples are mainly seen in benign lesions, and sampling of malignant microcalcifications is much more satisfactory.
Unsatisfactory specimens for CNB most often are due to missed sampling of a lesion but may be caused by fibrotic or sclerotic lesions that are difficult to sample. A CNB specimen obtained for mammographic calcification that does not contain microcalcification on the slide after X-ray of the tissue block should be considered unsatisfactory. Obtaining only skin or fatty tissue (except in suspected lipomas) should also be considered unsatisfactory.
Standardized reporting of FNB and CNB samples and quality assurance
Standard approaches to reporting FNB samples from the breast are recommended. A national conference sponsored by the National Institute of Health in the USA compiled the views of opinion leaders on information required on request forms, technical aspects of sampling, cytologic categories and information to be included in the cytopathology report.58,61 Five categories of FNB reports are recommended: benign, atypical/indeterminate, suspicious/probably malignant, malignant and unsatisfactory. In each category there should be an attempt to place the findings into a specific pathologic entity such as that used in a surgical pathology diagnosis. A slightly modified version of this system is used in Europe. The benign category is subdivided into ‘benign specific’ and ‘benign NOS’. The rational for this is that if a benign specific diagnosis such as cyst, fibroadenoma, fat necrosis, etc. can be made and if this is in accordance with radiological findings, the sample can be regarded as representative of the lesion. Benign NOS, on the other hand, means simply non-neoplastic breast tissue was obtained and radiologic and clinical follow-up is mandated to consider the sample representative of the lesion. Several surveys have studied the diagnostic criteria used in routine cytological practice or the accuracy achieved in interlaboratory comparisons in nonscreening and screening settings.25,28,62,63 Guidelines for best practices in diagnostic interventional breast procedures and standards have recently been published by the European Society of Breast Imaging.5 A similar review of prevailing recommendations and contemporary practices in breast FNA has also recently been published.64 Core biopsies should be reported similarly to surgical excision specimens. Audits of CNB diagnoses and comparison to final surgical excision diagnoses should be performed as part of quality assurance and improvement.
Complications
Complications for both FNB and CNB are uncommon. Major hematomas are unusual. Vasovagal reactions can occur. Pneumothorax is a rare but important complication, occurring more commonly in thin patients when the medial breast or axilla is sampled.65,66 A tangential approach to needling should be used. Patients may complain of sudden severe pain and a chest X-ray may reveal a pneumothorax. However, some patients do not experience pain until several hours after the procedure. No life-threatening cases have been reported, but there is the potential for medicolegal problems if the patient has not been made aware of the possibility before giving consent. Subpleural hematoma has also been described. Tumor implantation in a fine needle track is a very rare event but a few cases have been recorded in which local recurrence was ascribed to tumor seeding.67 Hematogeneous dissemination of breast tumor cells after FNB has been noted by RT-PCR.68 Long-term follow-up of large series of cases has not shown any adverse influence of FNB on tumor spread and prognosis.69
FNB does cause some disruption of tissue, even with good technique. A range of changes including hemorrhage, infarction and epithelial implantation resembling invasion have been described.70 Similar problems occur with CNB where epithelial displacement after core biopsy may imitate stromal or vascular invasion.71 Epithelial displacement can also lead to the misdiagnosis of DCIS as an invasive carcinoma.72 The risk of needle track seeding of mucinous carcinoma and perhaps some other malignancies should be kept in mind and the biopsy be planned so that the needle track is included in the subsequent surgical excision. There has been some concern about the risk of cutaneous seeding by CNB, but long-term experience is still insufficient to conclude if the risk is real.73 Removal of the entire lesion by CNB can be problematic if a post-biopsy clip was not placed. Accurate measurement of tumor size, and thus stage, also becomes difficult if most of a small lesion is removed by the CNB.
In patients with breast prostheses, accidental puncture of a breast prosthesis (silicone) can be avoided by careful positioning of the lesion, ultrasound guidance, and using the nonaspiration technique.74 Ultrasound-guided sampling with rapid compression of the aspiration site is a better alternative than stereotactic biopsy in patients with bleeding disorders. The venous compression, dependence of the breast and inability to compress the site during stereotactic biopsy encourage bruising.
Contraindications
There are no contraindications to FNB or CNB of breast lumps. Anticoagulation therapy is not a contraindication, but should be noted.
Technical considerations
Aspirates are best obtained with needles of 23–27 gauge. FNB without aspiration is preferable for benign and malignant breast lesions with a high cell content. The difference in tissue consistency between a ductal carcinoma of the usual type, a scirrhous cancer, a medullary carcinoma, a fibroadenoma, adipose tissue and a cyst wall is obvious when the needle is held directly with the fingertips. The feel of the consistency through the needle is a very valuable piece of information, and it helps to secure a representative specimen. We use aspiration only if the sample by the needle alone is too scanty, and in cystic lesions. Cellularity is lower in stereo-guided than in ultrasound-guided samples or in direct samples of palpable lesions, particularly for benign lesions. Several factors are involved. With stereotactic guidance the operator is less able to fix rubbery benign breast tissue and there is a lesser degree of movement of the needle through the lesion. The radiologist may need to withdraw the needle 1 cm above the lesion to allow a ‘run-up’ – a more vigorous movement through the lesion. There is also a tendency for smears to be more bloodstained, which results in some loss of bare oval nuclei in the background and sometimes in distortion of the cells.
We feel routinely rinsing the needle and syringe used in FNB with fixative, which is then filtered through a Millipore or a Nucleopore filter, is unnecessary if an adequate direct smear can be obtained. ThinPrep processed smears have been shown to be excellent for hormone receptor assessment and other prognostic markers but the use in routine diagnosis should not occur without adequate training.75 The choice of fixation and staining depends on personal experience. If possible, both alcohol-fixed and air-dried smears should be studied. Cyst fluid is processed in a similar manner to effusions.
Over time, needles used for CNB have increased in size from 18 gauge, now to 8–9-gauge needles. CNB requires local anesthesia and sterile conditions. Although core biopsy carries a higher incidence of local complications as compared to FNB the rate is still quite low. In desmoplastic carcinoma and other fibrous paucicellular lesions it may be impossible to obtain a sufficient number of cells by FNB, and a CNB may be the only alternative to a formal surgical biopsy.
Imprint smears can easily be made from CNBs by stamping or rolling the core on a dry, clean slide, then fixed and stained the same as a FNB smear. This allows an immediate preliminary assessment of the adequacy of the biopsy and may help to reduce the number of samples.76 Some surgeons find an immediate assessment to be of value when advising the patient.
For assessment of her-2-neu on core biopsy, recommendations for fixation time in formalin has not been fully addressed but is suggested to be at least 1 hour.
FNB for measurement of biological factors for diagnosis or prognostication
Subjective nuclear grading in FNB smears correlates well with nuclear grade in tissue sections and shows some correlation with prognosis.77-79 Since much of the prognostic information of histologic grading depends on the measurement of mitotic activity, the good correlation between mitotic counts in tissue sections and staining for proliferation markers in smears is promising.80 Multiple variables, such as nuclear morphometry, ploidy and cell kinetics, can be measured in cell samples or cell blocks, by microspectrophotometry, flow cytometry and video image analysis.81,82 Tumor-cell proliferation fraction measured by MIB-1 count in tissue samples has been shown to be a useful prognostic parameter in breast cancer.83 Immunohistochemical quantitation of ER and PR at the cellular level is an important development in breast pathology, and measurement in smears or cell blocks is as accurate as in tissue.84,85 Newer molecular techniques for detecting p53 gene mutations by PCR, gene alterations by fluorescence in situ hybridization or c-erb B2 oncoprotein by immunocytochemistry or fluorescence in situ hybridization can be applied to FNB material.82,86
Emerging molecular techniques such as genomic and proteomic profiling has been successfully used in cytologic material to identify patients at high risk for the development of carcinoma, for prediction of patient outcome and response to treatment and for tumor characterization.2 Studies have shown the successful use of FNB samples for cDNA microarray87 and protein microarray analyses.88
While prognostic marker studies can be performed on FNB, preference should be to perform ancillary studies on core biopsy or excision specimens.64 Prognostic and predictive factors that can be reliably assessed on core biopsy include tumor type, tumor grade, tumor size, extent of in situ component, presence of microinvasion, presence of lymphovascular invasion, and the presence of tumor necrosis.89 Biomarker assessment has included the study of molecular markers that have predictive and prognostic value including her-2, ER and PR. Other markers studied include p53, bcl-2, MIB-1, EGFR, and human milk fat globule membrane.89 High-throughput gene expression profiling have been performed on core biopsy specimens.90
Cytological findings
Non-neoplastic glandular breast tissue
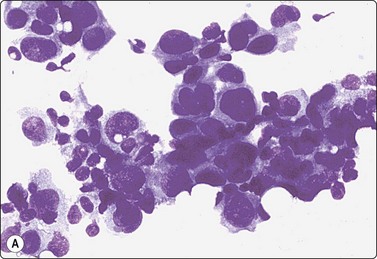
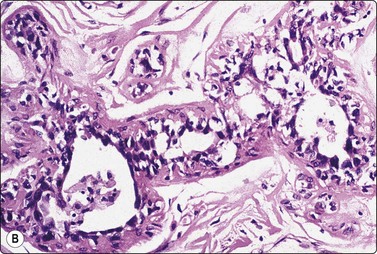
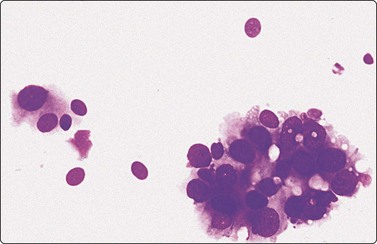
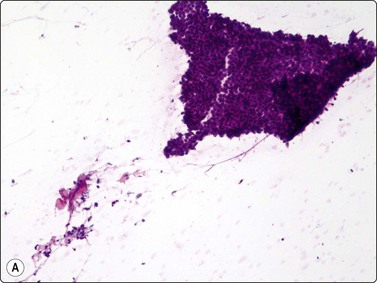
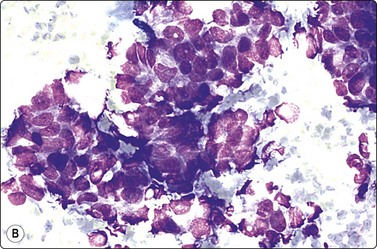
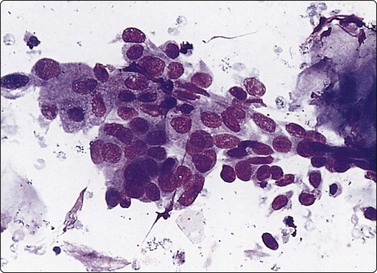
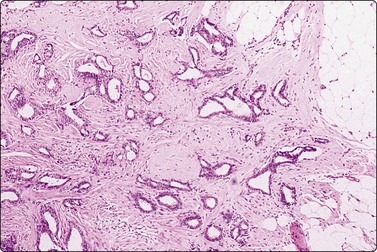
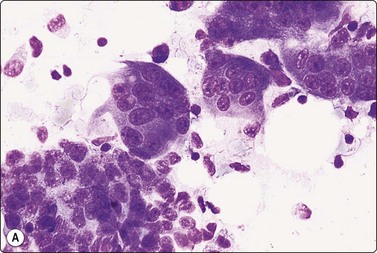
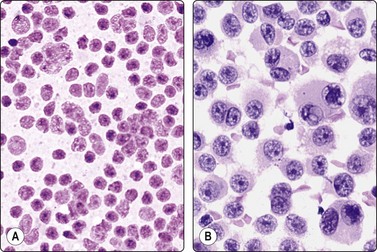
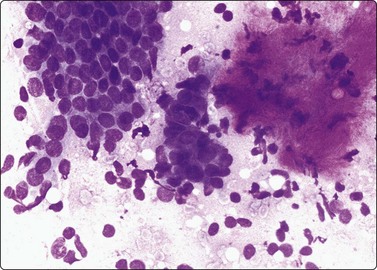
A comparison between the basic benign pattern (non-neoplastic glandular tissue) and the most common malignant pattern (low-grade carcinoma of no special type) in FNB of breast lesions is given in Table 7.2 (Figs 7.2-7.4).
Table 7.2 Comparison of the benign pattern and low-grade carcinoma in FNB smears
| Non-neoplastic breast tissue (Figs 7.2A, 7.3A and 7.4A) | Low-grade carcinoma NOS (Figs 7.2B, 7.3B and 7.4B) |
|---|---|
| 1. Overall low cell yield | 1. Variable but higher cell yield |
| 2. Sheets and aggregates of cohesive, small, uniform cells | 2. Irregular clusters of less cohesive, small, mildly irregular cells |
| 3. Small rounded nuclei, bland chromatin, some overlapping | 3. Slightly larger and darker nuclei, relatively bland chromatin |
| 4. Myoepithelial cell nuclei among epithelial cells | 4. Myoepithelial cell nuclei not seen |
| 5. Variable numbers of single, bare, bipolar nuclei scattered in the background | 5. Single cells, most with some cytoplasm, identical to those forming clusters; no bare bipolar nuclei |
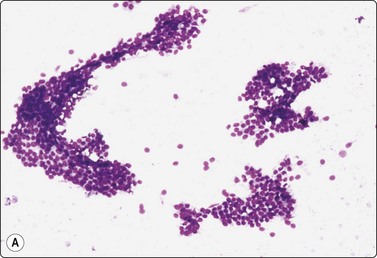
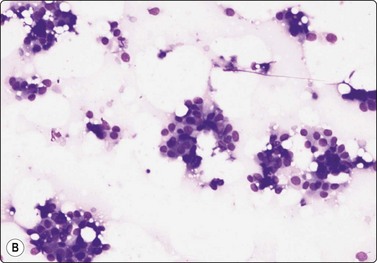
Fig. 7.2 Non-neoplastic glandular breast tissue and low-grade duct carcinoma
Low-power view; (A) Bimodal population of epithelial sheets and single bipolar nuclei of non-neoplastic glandular breast tissue; (B) Single population of epithelial cells in low-grade carcinoma (MGG, LP).
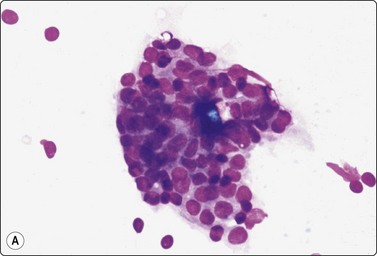
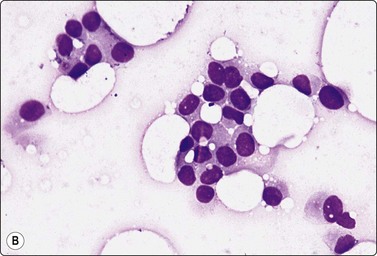
Fig. 7.3 Non-neoplastic glandular breast tissue and low-grade duct carcinoma
High-power view, air-dried smears; (A) Non-neoplastic glandular breast tissue; (B) Low-grade duct carcinoma. Note single bipolar nuclei in A, and absence of bipolar nuclei, relatively mild nuclear atypia and some loss of cohesion of malignant cells in B (MGG, HP).
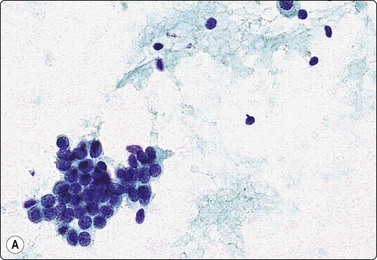
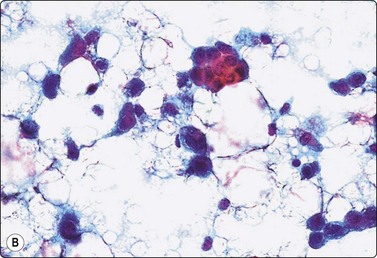
Fig. 7.4 Non-neoplastic glandular breast tissue and low-grade duct carcinoma
High-power view, Pap-stained smears; (A) Bimodal population in smear from non-neoplastic breast; (B) Single and clustered cells in low-grade carcinoma; some single cells probably stromal (Pap, HP).
The basic benign pattern is common to normal glandular breast tissue. Variations occur with the menstrual cycle and with the age of the patient, depending upon the variable proportions between epithelial cells and fibrous stroma. The yield of the needle biopsy is usually scanty and multiple biopsies should always be made to increase the likelihood that the material is representative.
The bimodal pattern of cohesive groups of epithelial cells and scattered single, bare, oval/bipolar nuclei is diagnostic of benign, non-neoplastic breast tissue. Ductular epithelial cells – this term is used here to designate cells from the intralobular epithelial structures of the resting breast, which differ distinctly from the acinar epithelial cells seen in pregnancy and lactation (see below) – are cohesive and are seen as small epithelial groups, which represent terminal ductules. The cohesiveness of non-neoplastic epithelium is in contrast to the dyscohesion of malignant cells unless very well differentiated (Figs 7.3 and 7.5). The nuclei are irregularly distributed within the groups and may appear crowded and overlapping (multilayered). They are uniform, small, round or oval, dark, with a granular chromatin. Nucleoli are indistinct or are very small. Cytoplasm is scanty, visible, but without distinct cell borders; it is pale and may show a blue granulation (MGG). Epithelial fragments from larger ducts are sometimes present. They form monolayered sheets of regularly arranged, slightly larger cells with uniform nuclei. The single, bare nuclei scattered in the background are of the same size or a little smaller than those of the epithelial cells. They have a bipolar/oval shape and a very smooth nuclear outline. The chromatin is dense and homogeneous and nucleoli are not seen (Figs 7.3A and 7.4A). The bipolar/oval nuclei sometimes wash off in heavily bloodstained and in wet-fixed smears. Nuclei of similar appearance can also be seen scattered between the cells of the epithelial fragments, distinguishable from these by their smaller size, bipolar shape, and darker staining (Fig. 7.6). They no doubt represent myoepithelial cells, whereas the single nuclei scattered in the background may be either myoepithelial or derived from the specialized, intralobular connective tissue. The number of bipolar/oval nuclei in smears corresponds closely to the cellularity of the lobular stromal component in sections. This is particularly evident in smears from fibroadenoma. However, a recent study of p63 immunoexpression in these cells concluded that the majority was of myoepithelial origin.91 Small fragments of collagen may be seen, particularly if larger-caliber needles have been used, but are usually inconspicuous, whereas fragments of adipose tissue are frequently present.
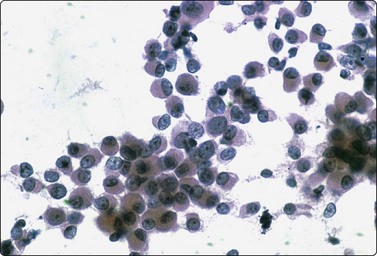
Fig. 7.5 Intermediate-grade duct carcinoma
Malignant epithelial cells with intact cytoplasm showing loss of cohesion characteristic of malignancy (Pap, HP).
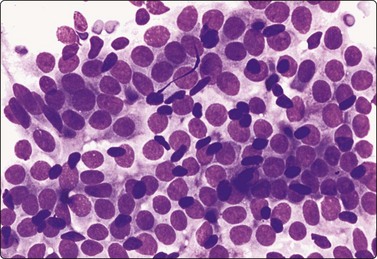
Fig. 7.6 Non-neoplastic glandular breast tissue
Sheet of cohesive ductular epithelial cells and scattered small, dark nuclei of myoepithelial cells (MGG, HP).
Hormone-related changes in non-neoplastic breast tissue
The breast gland is a hormone-responsive organ. Hormonal stimulation causes a hyperplastic response of the epithelial component. This may result in an increased cell content of FNB samples. It may also cause some nuclear enlargement and anisokaryosis, and cell cohesion may be somewhat diminished. The hormone-related ‘atypia’ that can be seen in pregnancy and in patients on hormone replacement therapy can occasionally cause some concern.92,93 However, except in late pregnancy and particularly in lactating breast tissue, the biphasic pattern of epithelial fragments and scattered single naked bipolar/oval nuclei characteristic of non-neoplastic breast tissue is maintained (Fig. 7.7).
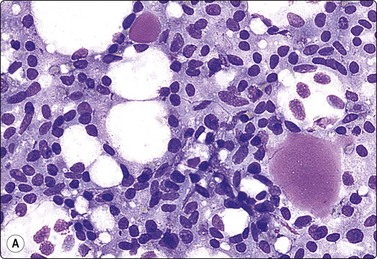
Breast tissue in pregnancy and lactation (Fig. 7.8)
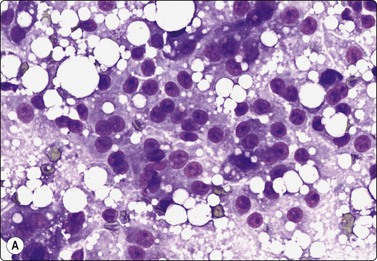
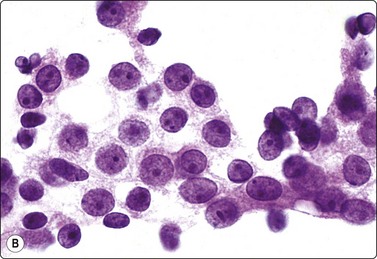
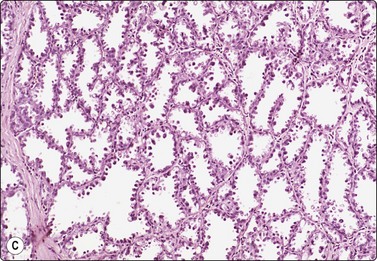
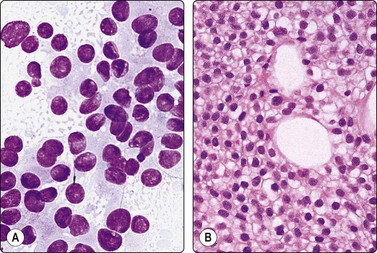
Dispersed acinar cells with abundant pale and fragile cytoplasm, rounded nuclei and prominent central nucleoli; background of lipid secretions (A, MGG, HP; B, H & E, HP oil); (C) Corresponding tissue section (H&E, IP).
FNB is very useful in reducing surgical intervention to a minimum during pregnancy and lactation. The pattern seen in FNB smears of ‘lumps’ in a pregnant or lactating breast can be problematic to inexperienced eyes and cause concern for malignancy.93,94 Smears are usually cellular. The cells are enlarged and arranged in loose groups or singly. The cells have an abundant fragile cytoplasm, vacuolated and finely granular. Nuclei are round, central, larger than the usual ductular cells, and have distinct small nucleoli (Fig. 7.8B). Some epithelial nuclei are stripped of cytoplasm. Single naked bipolar/oval nuclei are difficult to find. The background of abundant milky secretion with numerous lipid droplets seen as vacuoles is characteristic of actively secreting breast tissue and is the main clue to the diagnosis (Fig. 7.8A).
Problems and differential diagnosis
Gynecomastia of the male breast96-98
Fine needle aspiration biopsy is an extremely valuable tool in the initial evaluation of breast masses in men.96-98 In one series, gynecomastia was the most common diagnostic entity encountered in men with breast lumps.96 The cytological pattern of gynecomastia is not specific and the clinical presentation must be known to allow a diagnosis. Smears are variably cellular, ranging from scanty to markedly cellular. Epithelial fragments are often large as flat/monolayered sheets often with finger-like projections similar to fibroadenoma. Often there is a bimodal pattern of stroma and epithelial cells in addition to single bare bipolar/oval nuclei in the background. Adipose tissue may be present. Moderate nuclear variation and atypia can be allowed in the presence of a benign bimodal pattern (Fig. 7.9A). In some cases, nuclear atypia and cellularity can be quite prominent and can cause a concern for carcinoma and lead to excision (Fig. 7.9B).98 In contrast to gynecomastia, carcinoma yields more consistently highly cellular smears with discohesive single malignant cells, more pronounced atypia and lack of bare oval nuclei.
Fibrocystic change
Main features
Fibrocystic change may clinically cause an indistinct thickening or ‘lump’, or an asymmetrical density on the mammogram. Cytologically, it is a variant of the common benign pattern in which ‘cyst macrophages’, apocrine metaplastic cells and sheets of ductal epithelial cells are found in addition to the usual bimodal cell population of ductular epithelium and single bare oval nuclei. The former components may dominate the smears. Fluid from dilated ducts increases the volume of the aspirate. ‘Cyst macrophages’ have an abundant, finely vacuolated cytoplasm and may contain pigment, and small round central nuclei. At least some of the ‘macrophages’ may be degenerating epithelial cells exfoliated from the epithelial lining of dilated ducts, rather than true macrophages. Apocrine metaplastic cells have abundant, dense, finely granular eosinophilic cytoplasm, which stains gray-blue with MGG. The nuclei are round, nuclear size may vary considerably and nucleoli are often prominent. However, the nuclear outline is smooth and the chromatin pattern is uniformly granular (see Fig. 7.13A).
Fibrocystic change’ is not a specific cytological diagnosis. These changes are usually poorly circumscribed, and in the same area there may be focal epithelial hyperplasia, atypia or even malignancy. Malignancy can only be ruled out in the tiny area biopsied and the findings in the FNB samples must be evaluated in the context of the clinical and mammographic findings.
The amount of apocrine epithelium aspirated may be large and sometimes the cells show a greater degree of nuclear enlargement. Macronucleoli may also be seen. The atypia can raise a suspicion of carcinoma with apocrine differentiation (see Fig. 7.13B). However, benign apocrine epithelium in fibrocystic breast tissue is cohesive, forming monolayered sheets with few dispersed cells. Even when anisokaryosis is prominent, the N : C ratio is not increased, the nuclei are rounded, the nuclear membrane is smooth and the chromatin rarely shows any obvious abnormality. In contrast, groups of malignant apocrine cells are multilayered and disorganised. Nuclei are markedly pleomorphic and have irregular outlines and abnormal chromatin. If there is a DCIS component, as is often the case, necrotic debris is present in the background.
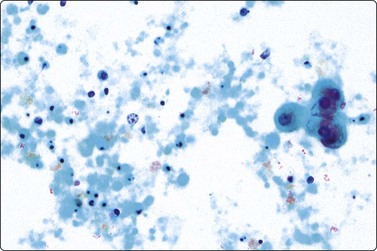
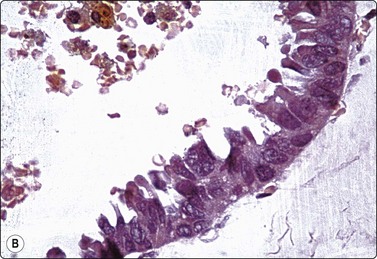
Simple cyst (Figs 7.10-7.14)
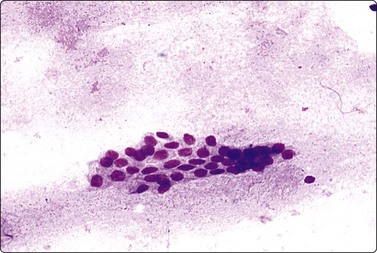
Inspissated secretion seen as a film of violet structureless material; sheet of bland ductal epithelial cells (MGG, IP).
Fig. 7.12 Non-cellular material in breast aspirates
(A) Proteinaceous structureless material aspirated from simple cyst; (B) Ultrasound gel contamination (MGG, HP).
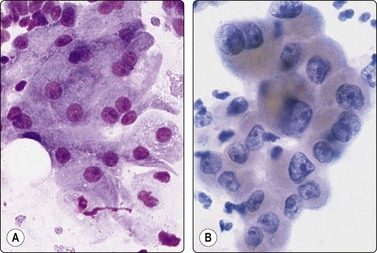
Fig. 7.13 Oxyphil/apocrine cells in cyst fluids
(A) Typical sheet of oxyphil cells with abundant cytoplasm and enlarged but bland nuclei (MGG, HP); (B) Sheet of oxyphil epithelium showing prominent nuclear enlargement, anisokaryosis, irregular chromatin and nucleoli, aspirated from simple benign cyst (Pap, HP).
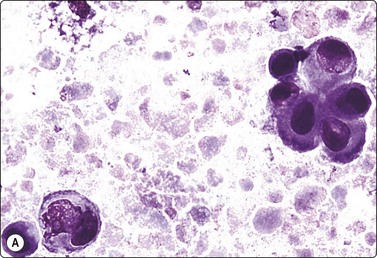
(A) Obviously malignant epithelial cells with a background of debris in aspirated fluid (MGG, HP); (B) Tissue section showing malignant cells lining the cyst wall (H&E, HP).
Breast cysts are easily diagnosed by US. However, evacuation by FNB may be a more convenient way to immediately relieve patient anxiety. Simple cysts are by far the commonest situation in which a confident benign diagnosis can be rendered by FNB, and a surgical excision avoided.
The aspirated fluid may be thin, clear or turbid, straw colored, brown or green. Smears may be practically cell free (Fig. 7.12A) or contain variable numbers of ‘cyst macrophages’ and epithelial cells, usually of apocrine metaplastic type and more or less degenerate (Fig. 7.13A). Numerous polymorphs are sometimes found in the cyst fluid even in cases with no clinical signs of inflammation.
A carcinoma may be present next to a cyst, hidden by a dominant benign cyst. Truly cystic carcinomas are uncommon but do occur, for example intracystic papillary carcinoma.99 Aspirated cyst fluid may be non-diagnostic. Correlation with radiological findings and re-aspiration of any residual lump after the fluid has been evacuated is essential. Some apparently cystic or partly cystic carcinomas are high-grade tumors with massive central hemorrhage and necrosis. The aspirated fluid is thick, murky and brown due to altered blood. A careful search of the smears may reveal aggregates of cells recognizable as malignant (Fig. 7.14A), or a mass remains after the initial aspiration. Repeat FNB usually yields diagnostic material.
It has been said that malignancy can safely be excluded if no abnormality remains after the cyst has been emptied and if the fluid is not bloody. Microscopic examination of the fluid may then not be necessary.
Apocrine metaplastic cells in cyst fluid often appear atypical, sometimes bizarre and worrying (Fig. 7.13B). They can mimic squamous differentiation and there may be unusual spindle-shaped epithelial cells. However, if all other findings are typical of a simple cyst and in the absence of altered blood or necrotic debris, or of a residual mass after the fluid has been drained, there is no indication for further investigation.
A distinction between inspissated cyst and duct ectasia as defined histologically cannot be made in smears. The diagnosis is based on the clinical findings. Duct ectasia is usually located close to the nipple, presenting as a subareolar cord-like mass of thickened tissue. It is an unlikely diagnosis in lesions located peripherally in the breast. The presence of chronic inflammatory cells and an occasional sheet of duct epithelium and a total absence of nuclear debris in the condensed secretion of duct ectasia is a clue.
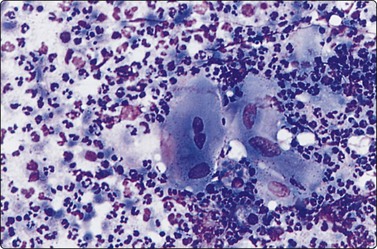
Mastitis (Fig. 7.15)
Common findings
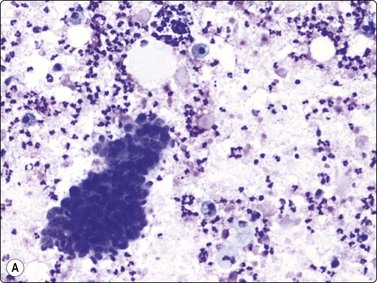
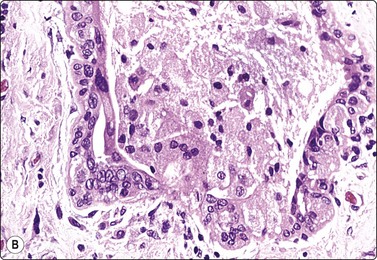
Fig. 7.15 Regenerative epithelial atypia in mastitis
(A) Atypical, reactive/regenerating epithelial cells with a background of histiocytes, inflammatory cells and debris (MGG, HP); (B) Corresponding tissue section (H&E, IP).
There are four types of mastitis recognized; acute, chronic, granulomatous and non-specific.93 The diagnosis of acute mastitis and abscess presents no problems. Pregnancy is the most common association, but occasionally cysts become infected (often by coagulase-negative staphylococci). The entity of subareolar abscess is recognized separately (Fig. 7.16). Chronic mastitis may be the result of persistence of an acute mastitis, a reaction to retained secretion in fibrocystic disease or duct ectasia, or secondary to previous surgery.
Granulomatous mastitis is an uncommon condition. It may mimic carcinoma radiographically and clinically; etiology is variable, ranging from tuberculosis, fungal, silicone, tumor related, sarcoidosis, fat necrosis, foreign body to non-specific. Aspiration reveals epithelioid histiocytes, multinucleate giant cells. Lymphocytes and plasma cells, may also be seen.
Epithelial atypia can be worrying in some cases of mastitis (Fig. 7.17). Caution is advised in interpreting cytologic findings within the clinical context. However, open biopsy may be necessary for definitive diagnosis in rare cases.
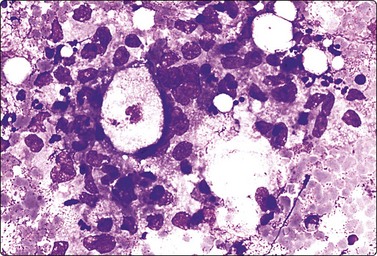
Fig. 7.17 Granulomatous mastitis
Atypical epithelial cells, histiocytes and inflammatory cells; no distinctly granulomatous pattern in this example (MGG, HP).
A foreign-body-type granulomatous reaction to silicone can occur adjacent to a breast prosthesis.100 FNB of a palpable nodule next to the implant may be performed to exclude malignancy. Great care must obviously be taken to avoid accidental puncture of the prosthesis, and US guidance is therefore required. The smear findings are of a foreign body granuloma with multinucleated giant cells, which may contain silicone particles and fibers (Fig. 7.18). If the nodule is a reaction to escaped silicone, the histiocytes and giant cells often contain vacuoles where the lipid-like material has been dissolved in processing.
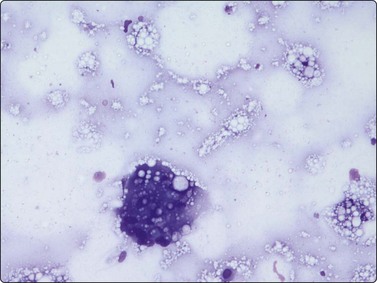
A giant cell with some foreign material from palpable nodule adjacent to breast prosthesis (MGG, HP).
Problems and differential diagnosis
Regenerative atypia of duct epithelium in an area of mastitis can look worrying and suspicious (Figs 7.15A and 7.17). In addition, the nuclei of reactive histiocytes may appear large and atypical, particularly in air-dried smears. False-positive diagnoses in cases of chronic mastitis and organizing fat necrosis have been reported. However, large numbers of both acute and chronic inflammatory cells are rarely seen in carcinoma. In medullary carcinoma with lymphocytic infiltration and in comedocarcinoma, in which lymphocytes and histiocytes are mixed with the carcinoma cells, the latter dominate and nuclear morphology is obviously malignant. The presence of necrotic cell debris should evoke a suspicion of malignancy.
Recurring subareolar abscess; lactiferous duct fistula (see Fig. 7.16)
Criteria for diagnosis:101
The prevailing view about the pathogenesis of this condition is keratinizing metaplasia of lactiferous ducts, perhaps as a pre-existing anomaly of the site, associated with recurrent bouts of inflammation, sometimes leading to a sinus track into areolar skin. Some of these episodes may be a chemical mastitis related to duct rupture, but superimposed infection does occur. The patients are often young and nulliparous. Mature or anucleate squamous cells with a background of pus aspirated from an abscess deep to the areola enable this diagnosis to be made. A specific cytologic diagnosis is of clinical significance, since these lesions generally require surgical treatment including excision of the affected duct system.
Problems in diagnosis
Keratinous material from the surface of the skin or from dirty slides may be misleading. There must be relatively abundant squamous material intimately mixed with the inflammatory cells to suggest the diagnosis. Reparative changes/atypia occur but the background of acute inflammation should prevent overdiagnosis.
Infected or ruptured epidermoid cysts produce a similar cytological picture, but occur more laterally and superficially.
Fat necrosis (Fig. 7.19)
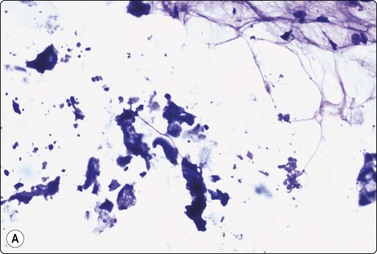
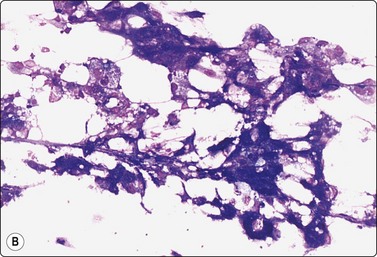
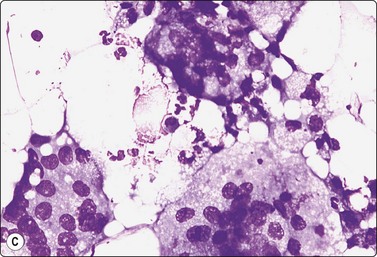
(A) Postoperative fat necrosis. Necrotic tissue, debris and macrophages; fragment of adipose tissue upper right (MGG IP); (B) Fragments consisting of altered adipocytes, macrophages and fat droplets (MGG, IP); (C) Several multinucleated histiocytes with foamy cytoplasm (MGG, HP).
Postoperative changes in response to prior surgical excision or biopsy give similar findings. Fat necrosis is always in the differential diagnosis of nodules in scars or chest wall after surgery, particularly mastectomy. Vigorous or repeated palpation of breast tissue or previous aspiration of the site can result in the same changes. The aspirate is usually scanty, sometimes of oily fluid, and consists mainly of fat with some foamy macrophages or altered, vacuolated adipocytes and multinucleated histiocytic giant cells. The untidy background of granular debris represents the actual necrosis and is the most specific diagnostic feature (Fig. 7.19A,B).
Problems in diagnosis
The dissolution of fat leads to a lipid cyst. Lipid cysts can be recognised by radiologists. The liquid is very viscous and may be yellow, clear, or have an unusual appearance of a gray-white color possibly corresponding to saponification. Solidification of the fluid in the test tube occurs after aspiration. The material does not stain with MGG although it may not dissolve completely during staining. A crystalline appearance is sometimes present in unstained slides.
The dispersed presentation of macrophages, particularly if the cytoplasm is dense or nonvacuolated and the nuclei are large with an irregular shape and prominent nucleoli, may mimic a malignant cell pattern, particularly in air-dried MGG smears. Multinucleate forms and foamy cells with similar morphology are helpful in preventing error (Fig. 7.19C).102 Conversely, some carcinoma cells may resemble macrophages. Immunostaining or excision is sometimes necessary.
‘Lipoma’ – hypertrophic fat tissue
Lesions of this type in the breast are not true lipomas but focal hypertrophy of fat tissue or ‘fatty lobules’ contained within a fibrous compartment of the breast. Focal fat hypertrophy presents as a discrete, rounded mass, which is often tender. It may be fairly firm to palpation, but at needling, once the thin fibrous capsule has been penetrated, the mass itself has a soft, ‘empty’ feel. Correlation with mammography is important to ascertain that the biopsy is representative, since the aspirated material is no different from that of normal adipose tissue of the breast or subcutis.
Intramammary and axillary lymph nodes
Lymph nodes are not uncommonly found within the breast, usually in the axillary tail but nodes can occur in any of the quadrants and more centrally.103 A lymph node may clinically simulate a fibroadenoma or a cyst and can be indeterminate on mammographic examination. Furthermore, malignant lymphoma can present as a breast lump, albeit rarely.104,105 The differential diagnosis between a reactive lymph node and lymphoma is discussed in Chapter 5, between lymphoid tissue and small cell primary breast carcinoma of neuroendocrine type on page 195.
Axillary or intramammary lymph nodes are usually correctly identified by the radiologist because of the bean shape and the central lucent hilum. Distinction between axillary node metastasis and primary carcinoma of the upper outer quadrant with a high content of lymphocytes is difficult at times, and node enlargement incidentally noted at mammography could be caused by malignant lymphoma.
US-guided FNA has been successfully utilized for the initial determination of axillary lymph node status in breast carcinoma.106 Ultrasound is more sensitive than physical examination in determining axillary lymph node status in patients with newly diagnosed breast carcinoma. US-guided FNB provides a more definitive diagnosis in sonographically indeterminate/suspicious lymph nodes compared to information provided by US alone.
Ectopic breast tissue
Breast tissue extending high up in the axilla commonly forms nodules or irregular lumps noted by the patient, especially during pregnancy and lactation, and may be referred for FNB. The ectopic glandular tissue may take part in fibrocystic change, and fibroadenoma or primary carcinoma can occasionally arise from ectopic tissue.106,107
Postradiation/chemotherapy effects17,19,108
Common findings
These features apply to glandular breast tissue undergoing radiation and/or chemotherapy. Post-treatment atypia can be severe and highly suggestive of recurrent carcinoma (see Fig. 7.1A). The key to a correct distinction cytologically between post-treatment atypia and recurrent malignancy is, above all, the awareness of this pitfall and the experience of benign reactive cellular changes from other fields of cytology. High cellularity and dissociation of the abnormal cells, and lack of bare oval nuclei are probably the most reliable criteria of recurrent malignancy in treated tissue. Most smears from benign post-treatment change are relatively low in cellularity. Although pleomorphism can be prominent, the cells are usually in cohesive groups with associated myoepithelial cell nuclei.
Fibroadenoma and phyllodes tumor
Fibroadenoma (Figs 7.20-7.28)
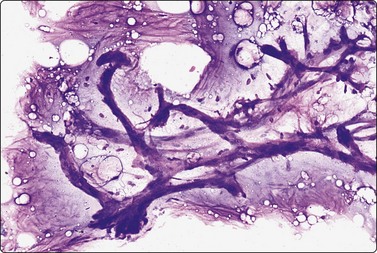
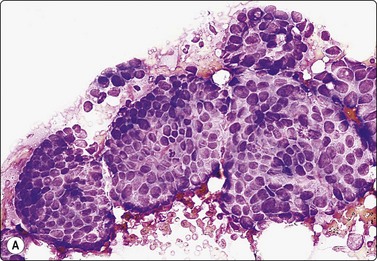
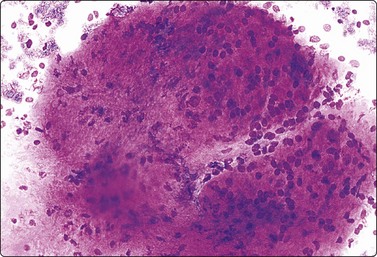
Fibroadenoma is one of the most common benign mammary neoplasms sampled by FNB. Aspiration biopsy is a highly reliable diagnostic procedure in the diagnosis of fibroadenoma when combined with clinical and imaging findings with a sensitivity reaching 97% and specificity, positive predictive value and negative predictive value reported to be 94%, 79% and 98%, respectively.109,110 Fibroadenomas are usually well-circumscribed lesions and have a characteristic rubbery consistency felt through the needle. Smears show a bimodal pattern of non-neoplastic breast tissue but are more cellular (Fig. 7.20). The epithelial fragments of regularly arranged, cohesive cells are large, elongated and branching, stag-horn-like, reflecting the appearance in tissue sections. There is variable nuclear crowding and overlapping. The nuclei are often mildly enlarged but uniform, have a bland granular chromatin pattern and often one or two small, indistinct nucleoli. Single, bare bipolar/oval nuclei are scattered in the background. They are characteristically numerous, much more numerous than in the usual glandular breast tissue, except in fibrotic and sclerosed fibroadenomas. Myoepithelial cell nuclei are also frequently seen within the epithelial aggregates. Sometimes, epithelial cells with apocrine differentiation may be prominent, and large numbers of foamy macrophages may suggest a cystic component. Fragments of fibromyxoid stroma are obtained from most but not all fibroadenomas. They stain pink to magenta with MGG, pale bluish-green with Pap, have a fibrillary structure and contain spindled fibroblastic nuclei (Figs 7.21-7.23). There may be a film of myxoid ground substance in the background. Smears from fibroadenoma with abundant collagenous stroma and scanty epithelium are less characteristic and may not differ much from the usual non-neoplastic breast tissue or fibrocystic change.
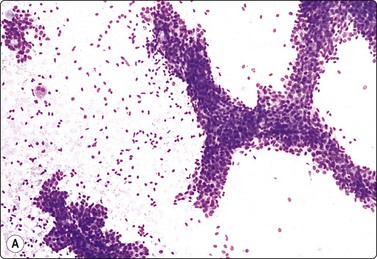
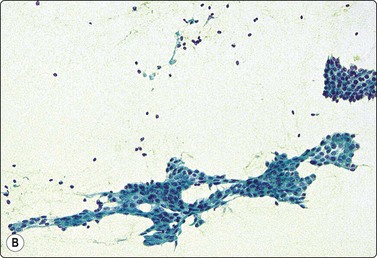
Cell-rich smear of elongated, branching fragments of ductal epithelium and numerous single bipolar nuclei in the background (A, MGG; B, Pap; LP).
Aggregates of cohesive epithelial cells, bare bipolar nuclei and a fragment of fibromyxoid stroma (MGG, HP).
Fragment of loosely fibromyxoid stroma with many spindle cells in a FNB smear. A cellular stroma per se is not diagnostic of phyllodes tumor (MGG, IP).
The triad of a cellular smear with a bimodal benign pattern, numerous single bipolar oval nuclei and fragments of stroma is virtually diagnostic of fibroadenoma. In the absence of stroma, numerous single bipolar nuclei are highly suggestive of the diagnosis.
Since a confident diagnosis of fibroadenoma usually means no excision and no further follow-up, diagnostic criteria must be strictly observed. These include the clinical and/or mammographic presentation of a well-defined rounded mass or density. There is some overlap between the smear patterns of proliferative fibrocystic change, or papilloma and fibroadenoma.109-111 Smears of papilloma may have large, branching epithelial fragments associated with fibrous stroma resembling fibroadenoma, but do not contain numerous single bipolar nuclei and the stroma is not myxoid. Fibrovascular cores and detached columnar cells are usually present. The distinction is more difficult in case of fibroadenoma with a fibrotic stroma. Fragments of myxoid stroma may also be seen in ‘fibroadenomatoid hyperplasia’. Fibroadenoma can undergo cystic degeneration, or the ductal structures can become dilated and filled with fluid. Smears from such lesions may contain numerous ‘cyst macrophages’ and apocrine metaplastic cells and may be interpreted as fibrocystic change.
Prominent nuclear atypia shown by a proportion of the epithelial cells is a common phenomenon in fibroadenoma, particularly in young women and women on hormone replacement therapy. The atypia is in the form of nuclear enlargement, anisokaryosis, some irregularity in shape and nuclear chromatin, prominent nucleoli, sometimes a few mitotic figures, and loss of cohesion (Figs 7.24-7.26). The cohesiveness of the epithelial component and relatively bland apearance of epithelial cells should alert against a diagnosis of carcinoma in such cases. Fibroadenoma is the most common cause of false-suspicious and false-positive diagnoses in breast FNB.112-115 In the presence of a clearly benign component of single bipolar nuclei, fragments of bland epithelium, and stroma, a malignant diagnosis should not be made regardless of the degree of atypia shown by a proportion of the epithelial cells. The atypia is most likely hormone related, but a consistent correlation with hormone replacement therapy has not been demonstrated. Proliferative fibrocystic changes and apocrine metaplasia are also common sources of atypia. Histologically, fibroadenomas with severe cytological atypia do not differ from the usual type overall, but focally the duct epithelium may show corresponding nuclear atypia when examined with high magnification (Fig. 7.27). Nevertheless, if prominent atypia is found in FNB smears, a recommendation of excision is advised. Carcinoma can rarely arise in a fibroadenoma. We have seen a few cases of DCIS in fibroadenoma (Fig. 7.28), one with focal invasion. In clinical practice, cytologically typical benign fibroadenomas in postmenopausal patients and fibroadenomas which continue to grow in size are often excised, particularly if the lesion is large.
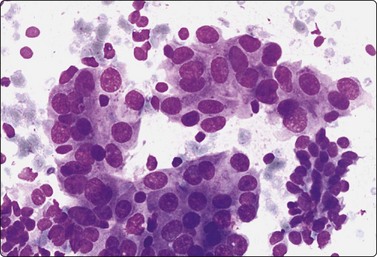
Fig. 7.24 Epithelial atypia in fibroadenoma
Aggregates of atypical epithelial cells showing prominent nuclear enlargement and some irregularity; compare the epithelial fragment of bland cells of usual type at lower right; scattered bare bipolar nuclei (MGG, HP).
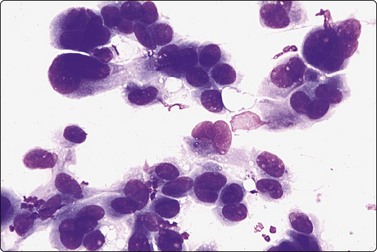
Fig. 7.25 Severe nuclear atypia in fibroadenoma
Prominent nuclear enlargement and pleomorphism, hyperchromasia and irregular chromatin. Benign elements typical of fibroadenoma were present in other parts of same smear (MGG, HP).
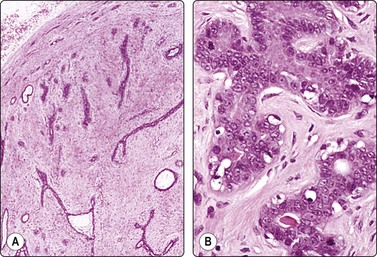
Tissue sections from same case as Figure 7.24; (A) Low power: typical appearances of fibroadenoma; (B) High power: ductal structures lined by atypical epithelium with enlarged vesicular nuclei and a few mitoses (H&E).
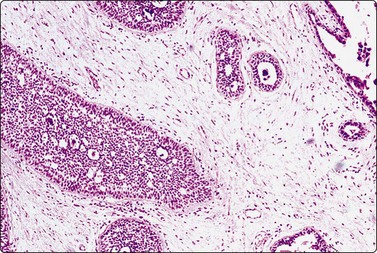
Fig. 7.28 DCIS within a fibroadenoma FNB smears from this lesion showed epithelial atypia, and excision was recommended. Tissue section (H&E, LP).
Occasionally, low-grade invasive carcinoma such as tubular carcinoma, lobular carcinoma and intraductal low-grade carcinoma may have a loose fibromyxoid stroma producing a smear pattern that mimics fibroadenoma114,115 A false-negative diagnosis of fibroadenoma can be made in such cases. The presence or absence of single bipolar nuclei typical of non-neoplastic breast tissue is of great importance in this context. Fibroadenomas can undergo cystic degeneration, or the ductal structures can become dilated and filled with fluid. Smears from such lesions may contain numerous ‘cyst macrophages’ and apocrine metaplastic cells and may be interpreted as fibrocystic change. In our experience, some of these cases are difficult to classify even after retrospective review.
In some cases of myxoid fibroadenoma, mucinous (colloid) carcinoma enters the differential diagnosis.110,116 Myxoid fibroadenoma can be distinguished from mucinous carcinoma by the background containing numerous oval bare nuclei and vascular myxoid stromal fragments, and the absence of dissociated single atypical epithelial cells floating in mucin. Smears of mucinous carcinoma lack oval bare nuclei and usually display free-floating vascular structures instead of stromal fragments. Despite these clues, some myxoid fibroadenomas may ultimately have to be surgically excised for a definitive diagnosis. In addition, in some cases atypia may be of limited value since some fibroadenomas may display more cytologic atypia then colloid carcinoma.
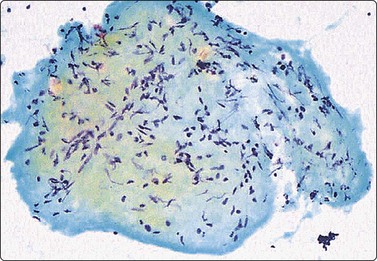
Phyllodes tumor (Figs 7.29-7.32)117-123
Phyllodes tumor (PT) is a biphasic epithelial/stromal neoplasm of the breast. In contrast to fibroadenoma, PT is a rare tumor comprising less than 0.3% of all breast tumors. It is classified as benign, low grade (borderline) and high grade (malignant) based on histologic features. Stromal cellularity and overgrowth, atypia, mitotic activity, and invasive growth pattern at tumor periphery define whether a PT is benign, low grade or high grade. Benign PTs do not metastasize, but may locally recur if incompletely excised. High-grade PTs behave like sarcomas with higher potential for recurrence and metastasis. Low-grade PTs fall in between.
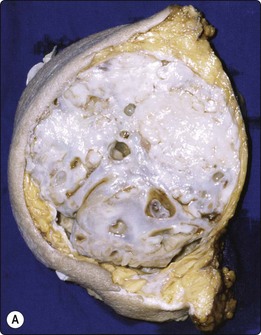
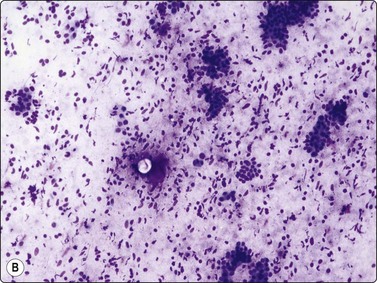
Fig. 7.29 Benign phyllodes tumor
(A) Huge, slowly growing breast mass in a 40-year-old woman; (B) FNB smears were cellular, dominated by dispersed cells with bare oval or plump spindle nuclei (MGG, HP).
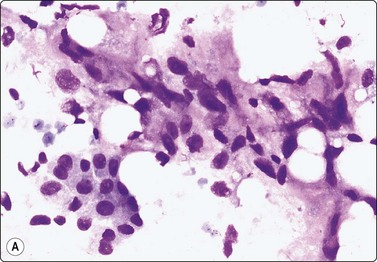
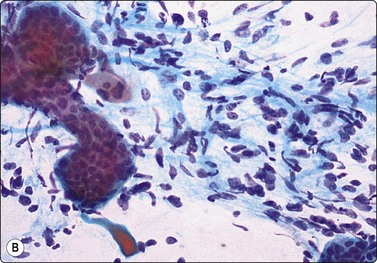
Fig. 7.30 Borderline phyllodes tumor
Smears dominated by mildly atypical, both plump and slender spindle cells, single and in loose tissue fragments with fibrous stroma; a few sheets of bland duct epithelium (A, MGG; B, Pap, HP).
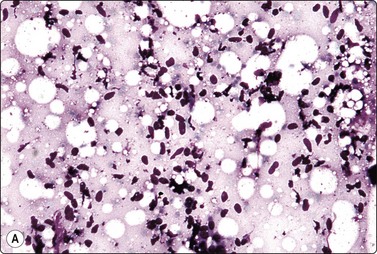
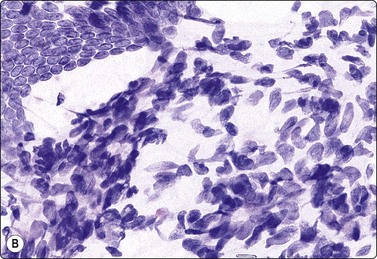
Fig. 7.31 Malignant phyllodes tumor
(A) Mainly dispersed spindle cells showing moderate nuclear atypia, no epithelial cells. Invasive growth demonstrated in tissue sections; multiple recurrences (MGG, IP); (B) Another case showing numerous spindle cells with more marked atypia and a sheet of bland epithelium (Pap, HP).
Aspiration biopsy can accurately diagnose malignant PTs in most cases. On the benign/borderline end of the spectrum, cytologic features of fibroadenoma and PT overlap, making FNA diagnosis difficult (see Fig. 7.23). Precise preoperative distinction is important for optimal patient management. Classic cytologic features in PT are similar to fibroadenoma. However, as opposed to fibroadenoma, stromal fragments are larger, increased in number (stromal overgrowth) and are hypercellular (phyllodes fragments); the single stromal cells in the background are plumper than the typical oval bare nuclei seen in fibroadenoma. These single cells are intact spindled cells with retained cytoplasm (not naked nuclei), and variable degrees of nuclear atypia with nucleoli and pleomorphism. However, some of these features may be entirely lacking in benign and low-grade PTs even after retrospective review of smears, making their differentiation from fibroadenoma virtually impossible. It is not surprising that a considerable portion of benign and low-grade PTs are initially diagnosed as fibroadenoma on cytology.117,120,121 This in part reflects sampling problems as hypo- and hypercellular areas tend to alternate within PTs. Another important diagnostic pitfall in PTs is the presence of significant epithelial proliferation including atypical ductal epithelial hyperplasia. If these areas are sampled by aspiration biopsy, this may lead to a false diagnosis of epithelial neoplasm. In our experience, we encountered this problem even with high-grade (malignant) PTs; one such retroareaolar case required core biopsy due to inability of aspiration biopsy to rule out an atypical papillary lesion. In addition, focal malignant transformation may be missed by FNA sampling.
The diagnosis of PT on CNB is equally as challenging (Fig. 7.32). It is especially difficult to differentiate cellular fibroadenoma from benign/low-grade PT. In comparison, in one study, the possibility of PT was raised in 23% on FNB and 65% on core biopsy.122 In two others, 11 of 44 (25%), and 9 of 23 (39%) of surgically resected PTs were reported as fibroadenoma or benign on core biopsy.123,124 Similar to FNB, some PTs are diagnosed as fibroadenoma on core biopsy because of tumor heterogeneity. Marked nuclear pleomorphism and mitotic activity suggest frankly malignant phyllodes tumor (Fig. 7.31A,B).
Adenomyoepithelioma (Fig. 7.33)
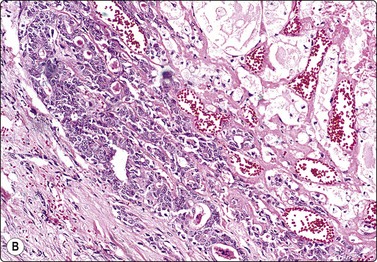
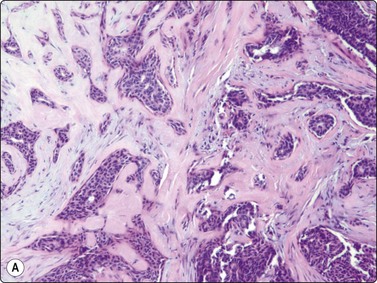
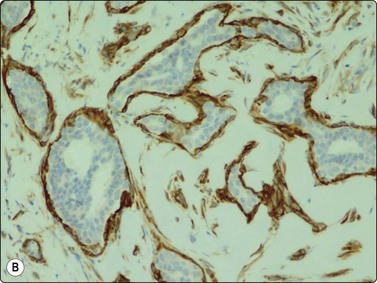
Adenomyoepithelioma, an uncommon biphasic tumor composed of epithelial and myoepithelial cells, usually presents as a single circumscribed nodule.125 Cytological features include cellular smears with epithelial aggregates, less cohesive myoepithelial cells with pale, fragile cytoplasm, and a variable amount of background stromal tissue (Fig 7.33A).126-128 Intranuclear cytoplasmic inclusions have been observed.129 It is important to recognize this entity, as the radiologic and cytologic features can mimic malignancy. However, only rarely can this diagnosis be made definitively on FNA due to overlapping features with other entities such as fibroadenoma, phyllodes tumor, myoepithelioma, and tubular carcinoma. A review of 18 cases collected from the literature showed that the cytological diagnosis of this entity is often difficult and that there is a risk of false-positive reports.130 False-positive reports of CNBs of adenomyoepithelioma have been reported (Fig. 7.33B).131 The majority of adenomyopeitheliomas are benign but local recurrences and rarely distant metastases have been described. Rarely, malignant adenomyopeitheliomas can occur. These are usually characterized by cellular pleomorphism, necrosis, high mitotic activity and invasion of the surrounding tissue.
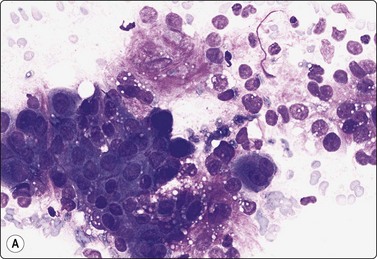
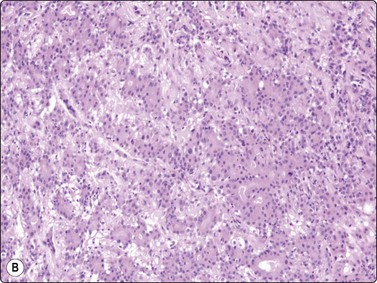
(A) A biphasic smear pattern of mildly atypical epithelial cells and dispersed myoepithelial cells with abundant pale fragile cytoplasm; scanty fibrous stroma (MGG, HP); (B) Core biopsy showing tubules in a pseudoinfiltrative pattern (H&E, IP).
Problems and differential diagnosis
Tubular adenoma, adenosis tumor, microglandular adenosis
Specific diagnosis of these entities is usually not possible by FNB. Tubular adenoma usually presents as a palpable mass in young women. Cytologic features include uniform tubules and three-dimensional epithelial balls in a background containing naked nuclei but no stromal fragments.93 Distinction from fibroadenoma may be difficult. Distinction diagnosis includes tubular carcinoma; in tubular carcinoma, tubules are more angulated and they vary in size and shape; bipolar nuclei are absent. Findings similar to tubular adenoma may be seen in adenosis tumor, which represents localised hyperplasia of lobules/ductules associated with a palpable mass. The cytologic diagnosis of adenosis tumor is difficult due to overlapping features with fibrocystic changes and fibroadenoma. Similarly, a specific cytologic diagnosis of microglandular adenosis is challenging due to overlapping features with other entities described herein. Features are similar to adenosis, with small groups of glycogen filled clear cells showing uniform nuclei and small nucleoli and clusters of fibroblastic cells. The lack of myoepithelial cells may result in an atypical/suspicious diagnosis.
The spectrum of epithelial hyperplasia
The spectrum of epithelial proliferative processes of the breast discussed herein includes usual epithelial hyperplasia, atypical ductal hyperplasia (ADH), papilloma, radial scar and complex sclerosing lesions, and sclerosing adenosis. These entities are histologically relatively well defined but there is a certain overlap that can cause inter-observer disagreement. The overlap is more important in FNB smears, and it is not often possible to seperate a particular case precisely within the spectrum.132-134 There is also an overlap with nonproliferative lesions that sometimes can give an unusually cell-rich yield.135 The difficulties are enhanced by the selective nature of FNB sampling. A spectrum of epithelial hyperplasia and atypical epithelial hyperplasia may be present within the same clinically or radiologically defined lesion, and the most abnormal component may not be represented in the sample. In our opinion, it is therefore preferable to report the cytological findings in this category of lesions as consistent with ‘proliferative fibrocystic change’ with or without atypia. The degree of atypia and any suspicion of malignancy should be specified. The definitive diagnosis is left to histology unless the lesion is considered radiologically and cytologically clearly benign with no indication for core needle or open biopsy.
It is not possible, in our opinion, to list specific cytologic criteria for each one of these processes. We can only describe and compare a number of features as they appear in the main entities – usual epithelial hyperplasia, ADH, and low-grade DCIS. Papilloma, radial scar and complex sclerosing lesions, and sclerosing adenosis will be discussed separately.
Usual epithelial hyperplasia, and ADH
Usual findings with a comparison to low-grade DCIS are listed in Table 7.3 and illustrated in Figs 7.34-7.39. Regarding usual epithelial hyperplasia (Figs 7.34 and 7.37), the correlation between cytological appearances and subsequent histology is imperfect.134 Criteria such as swirling three-dimensional masses with slit-like irregular lumens are said to be predictive of benign proliferative disease and not seen in DCIS.136,137 The question is often asked, does cellularity alone constitute a reason for excision, if the pattern is entirely benign? In general, we do not recommend excision on the basis of cellularity alone. Discordant clinical, mammographic or ultrasound findings are usually the main reason for excision. CNB or excision is also recommended if there is cytological atypia such as evidence of papillary growth, diminished cell cohesion, nuclear atypia, sparsity of bare bipolar nuclei, and presence of necrosis. Epithelial hyperplasia with or without atypia may be associated with malignancy, particularly in situ carcinoma, and close correlation with clinical and mammographic findings is essential. Malignancy can not be confidently ruled out by FNB alone.
Table 7.3 Usual epithelial hyperplasia, ADH and low-grade DCIS; cytological findings
| Epithelial hyperplasia | ADH | Low-grade DCIS |
|---|---|---|
| Cell-rich smears, large sheets of cohesive epithelial cells, few single cells | Cell-rich smears, large sheets of cohesive epithelial cells, few single cells | Cell-rich smears, large and smaller sheets of cohesive epithelial cells, few single cells |
| Cells often in a ‘streaming’ pattern; focal crowding and overlapping of nuclei, rarely ‘holes’ | Focal crowding and overlapping of nuclei; ‘holes’ suggestive of cribriform pattern in some cases | Focal crowding and overlapping of nuclei; ‘holes’ suggestive of cribriform pattern common; some papillary cell groups |
| Nuclear atypia absent or mild | Mild to moderate nuclear atypia | Mild to moderate nuclear atypia |
| Naked bipolar and myoepithelial nuclei present but may be few; clean background; calcium granules occasionally | Few naked bipolar and myoepithelial nuclei; debris and calcium occasionally present | Naked bipolar and myoepithelial nuclei absent; necrotic debris and calcium often but not invariably present |
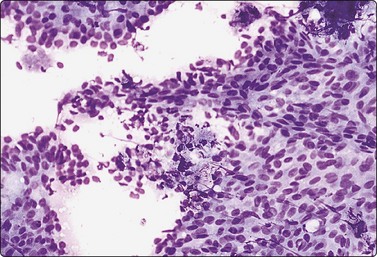
Fig. 7.34 Usual ductal epithelial hyperplasia
Large, slightly disorganised sheets of ductal cells; tendency to ‘streaming’; a few foamy cells and some bare bipolar nuclei (MGG, IP).
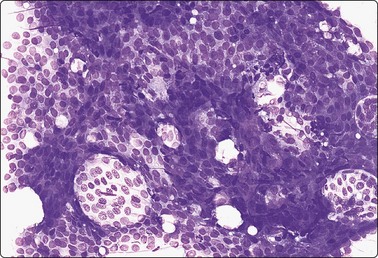
Fig. 7.35 Atypical ductal hyperplasia (ADH)
Large sheet of mildly atypical cells; ‘holes’ indicating a cribriform pattern, myoepithelial nuclei not seen (MGG, IP).
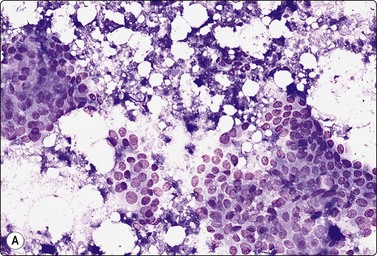
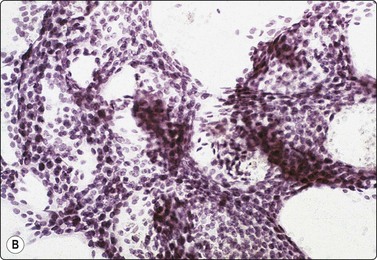
Fig. 7.36 Low-grade cribriform DCIS
(A) Sheets of mildly atypical ductal epithelial cells; necrotic debris and calcium granules (MGG, IP); (B) Large sheet of ductal epithelium with many ‘holes’ suggestive of a cribriform pattern; no myoepithelial nuclei (Pap, IP).
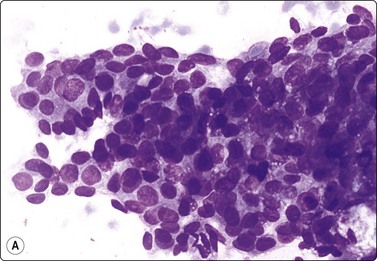
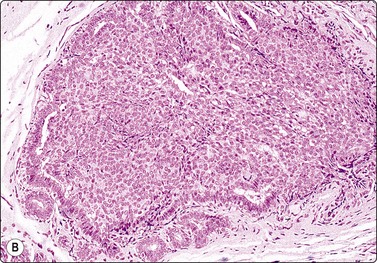
Fig. 7.37 Usual ductal epithelial hyperplasia
(A) Large aggregate of mildly atypical ductal epithelial cells, nuclear crowding and overlapping; myoepithelial nuclei not obvious (MGG, HP); (B) Corresponding tissue section (H&E, IP).
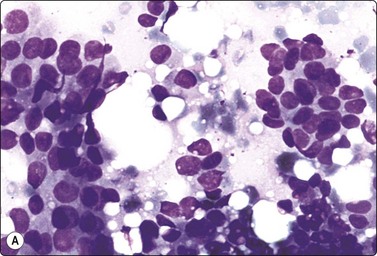
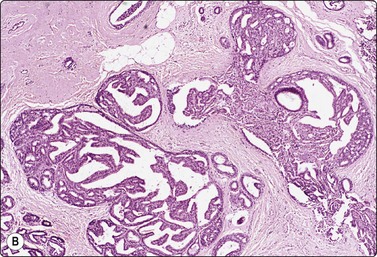
Fig. 7.38 Atypical ductal hyperplasia (ADH)
(A) Aggregates of moderately atypical ductal epithelial cells; some loss of cohesion; no myoepithelial nuclei (MGG, HP); (B) Corresponding tissue section (H&E, IP).
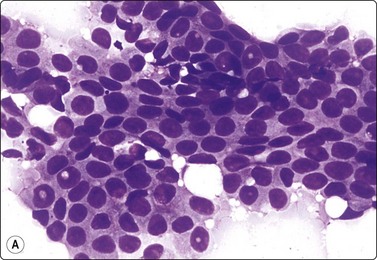
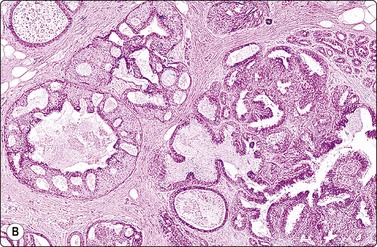
Fig. 7.39 Low-grade cribriform DCIS
(A) Sheet of mildly atypical ductal epithelial cells; some crowding of nuclei; no myoepithelial cells; no necrosis or calcium (MGG, HP); (B) Corresponding tissue section; mixed pattern of epithelial hyperplasia and cribriform DCIS (H&E, IP).
Atypical ductal hyperplasia (ADH) yields similarly cell-rich smears of mainly cohesive sheets and aggregates of epithelial cells (Figs 7.35 and 7.38). Cytological atypia is variable and equals or sometimes even exceeds that of low-grade DCIS. Necrotic debris can sometimes be found. ‘Holes’ corresponding to the cribriform pattern considered typical of low-grade DCIS may occur also in ADH (Fig. 7.35). Other cases show a lesser degree of cytological atypia and a major component of benign-appearing material. Sneige and Staerkel found both cytological and architectural features of value in identifying atypical or malignant intraductal lesions but there was an overlap with low-grade DCIS and epithelial hyperplasia without atypia.138 We feel that the overlap is too great to allow a specific diagnosis and that there is no single feature which can confidently separate ADH from low-grade DCIS. The cytologic diagnosis of low-grade DCIS is difficult, and 50% of cases in one study were called atypical on FNB.41
Smears from low-grade DCIS are also cell-rich of mainly large cohesive sheets but also smaller aggregates of ductal epithelial cells (Figs 7.36 and 7.39). In a study done by our group, all low-grade DCIS cases had moderately to highly cellular smears with cohesive, three-dimensional sheets of uniform, small cells with inconspicuous nucleoli arranged around a central lumen, forming cribriform ‘punched-out’ spaces.41 Single malignant cells were prominent in only a minority of cases. A study by Sauer et al. demonstrated cribriform architecture in more than 90% of cases and single cells in approximately 30% of cases.139 As mentioned above, rounded ‘holes’ in epithelial sheets in smears suggest a cribriform pattern, but are not specific to DCIS. In general, absence of myoepithelial cells and oval bare nuclei favor low-grade DCIS over ADH. However, myoepithelial cells were demonstrated in 51% of the nonhigh-grade DCIS lesions in one study.139 Cell balls and papillary fragments are features that overlap with those of papillary lesions (see below).
Malignancy in lesions with epithelial hyperplasia is suggested by nuclear atypia, loss of epithelial cell cohesion, necrosis, and absence of myoepithelial cells and single bare bipolar nuclei. However, nuclear atypia in low-grade DCIS is variable and relatively mild. Obvious necrosis in the form of granular debris and calcified granules is highly suggestive of DCIS (Fig.7.36A) but is not diagnostic per se. Similar material can sometimes be found in ADH, and occasionally in benign proliferative processes. A complete absence of myoepithelial cells and single bipolar nuclei is also in favor of malignancy. In some cases of DCIS, a proportion of the epithelial cells may show more obvious nuclear atypia and chromatin abnormality, increasing the level of suspicion of malignancy.
Correlation with the mammogram is essential and definitive diagnosis is in most cases deferred to histology.
The term ‘mild atypia’ should probably be avoided in reports, because it causes great confusion for clinicians. On the one hand, the pathologist is describing a departure from normal which raises the possibility of malignancy, but, on the other, it is classified as mild and presumably inconsequential. The clinician knows that if there is no further action to investigate the lesion and carcinoma is found later, there may be medicolegal problems. This term is one of the most common reasons for inappropriate open biopsies. The pathologist should generally make a judgment as to whether the degree of atypia is compatible with epithelial hyperplasia (or fibroadenoma, hormonal stimulation, etc.) and report the changes as benign, or whether there is a real possibility of a significant lesion. If the latter decision is made, there should be a recommendation for further investigation. This might be further FNB, CNB or excision, added to mammography and ultrasound examination. The pathologist should not hesitate to recommend CNB or open biopsy if epithelial atypia is more obvious, in view of the overlapping of findings not only in usual hyperplasia, ADH and low-grade DCIS described above, but also in some low-grade invasive carcinomas, notably tubular carcinoma. Overdiagnosis of cancer, on the other hand, is avoided by paying attention to the presence of more than a few clearly benign elements, both ductular epithelium and single bipolar nuclei, in the presence of which great caution should be observed.
A diagnosis of ADH on vacuum-assisted 11-gauge core biopsy warrants excision as 10% to 27% of cases will show carcinoma at surgical excision.140,141
An uncommon feature that can cause concern is ‘partial’ apocrine change, presumably a variant of apocrine metaplasia. Here, the epithelial cells have more cytoplasm than ordinary ductal cells and appear cytologically to be a halfway house to apocrine epithelium. These cells may show loss of cohesion and there may be no bare oval nuclei in the background. The most cellular and worrying cases are usually derived from areas of marked intraductal epithelial hyperplasia. Apocrine metaplasia in areas of adenosis can appear even more worrying.142 We have noted this phenomenon in association with radial scars and in a ductal adenoma (see Fig. 7.76B).
Columnar cell lesions (CCL) have recently attracted increasing interest in the context of mammographic screening. Histologically, terminal ducts are focally dilated and lined by columnar epithelial cells with cytoplasmic projections (snouts).143 There may be a variable degree of hyperplasia (columnar cell hyperplasia) with or without atypia. At one end of the spectrum, the atypia equals ADH or low-grade DCIS. Although the premalignant potential is uncertain, awareness of these lesions is important in FNAC since cytological atypia may be considerable with some risk of false-suspicious or positive diagnosis. There are only a few published studies in the cytology literature which describe features of CLLs in aspirates.144,145 While one study144 described characteristic features (three-dimensional clusters of enlarged polygonal epithelial cells intermixed with myoepithelial cells and palisading columnar cells peripherally) that allow recognition of this entity in FNA material, the other study145 concluded that columnar cell lesions cannot be reliably diagnosed on cytology due to cytomorphologic overlap with other lesions, especially with papillary neoplasms and well-differentiated adenocarcinoma. Our own experince agrees with Jensen et al.145 in that overlapping cytologic findings can also be seen in fibroadenomas and proliferative fibrocystic changes. In Jensen’s study, palisading columnar cells were present in only 50% of cases, whereas flat sheets of cells resembling apocrine metaplasia were more prominent. Atypia varied from mild to marked. In summary, it is important to recognize that CLLs display a spectrum of cytologic changes that may lead to confusion with other entities including well-differentiated adenocarcinoma.
Papillary lesions (Figs 7.40-7.45)111,146-148
Aspiration of intraductal papilloma often yields a small amount of fluid. Smears may be among the most cellular seen in FNB of the breast, with large aggregates distributed all over the slide as well as many dispersed cells. Intact papillary structures are less often seen. They are most often found in thick bloody parts of the smear, where they have not been disrupted by the smearing pressure. Complex folded sheets associated with collagenous stromal cores are the most characteristic feature (Fig. 7.40A). Frequent rows of palisaded cells with a columnar shape are commonly seen and are a useful indicator of papillary or complex cribriform lesions (Fig. 7.40B). A foamy change of epithelial cells at the margins of aggregates occurs when cells are within a fluid medium. Some of the foamy cells in the smear background may be of epithelial rather than macrophage origin. Hemosiderin pigment and debris may result from minor bleed, which commonly occur in intraductal papilloma. A blood-stained nipple discharge is a common clinical sign. Smears of the discharge show degenerating epithelial cells and hemosiderin-containing macrophages.
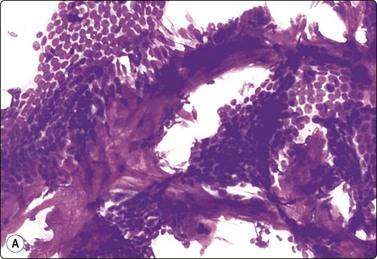
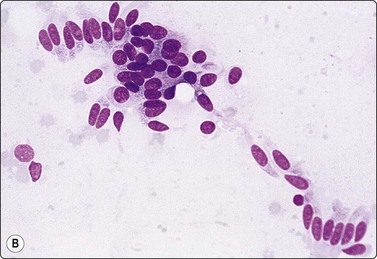
(A) Sheets of bland epithelial cells associated with strands of fibrous or fibrovascular stroma; bipolar nuclei difficult to find (MGG, IP); (B) Clusters of bland epithelial cells, many with an obvious columnar shape and lining up in palisaded rows (MGG, HP).
The cytological criteria apply to any papillary lesion in the breast and are not specific for papilloma. Cytological diagnosis of papillary lesions shows a significant error rate with overlapping features.147 Low-grade ductal carcinoma in situ of cribriform or micropapillary type and noninvasive intracystic papillary carcinoma may have all of the above features (Figs 7.41 and 7.42). Cellular atypia and fragments with long and slender papillae with ramifying edges favors papillary carcinoma. Like other forms of epithelial proliferative lesions, peripheral papillomas may be associated with DCIS. Our approach to cytological diagnosis is similar to that of frozen section of such lesions. We report this category as papillary lesions with an assessment of the degree of cytological atypia, if present, and recommend formal excision. Core needle biopsy may allow a more specific diagnosis, but may still not be entirely representative and thus complete excision of any papillary lesion that has not been entirely removed by the initial core biopsy is the optimal management for localized lesions.148 Most papillary lesions behave indolently, and outcome is usually excellent.
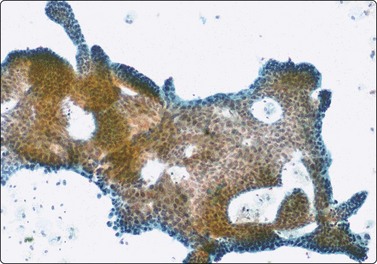
Fig. 7.41 Intracystic papillary carcinoma
Complex folded sheet with finger-like extensions/papillaroid edges; background of macrophages (Pap, LP).
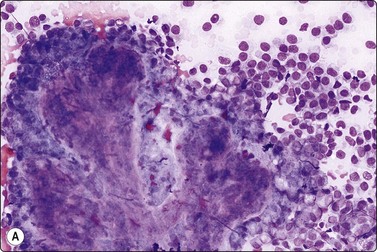
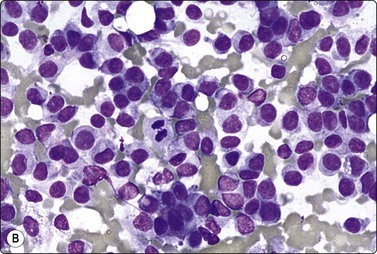
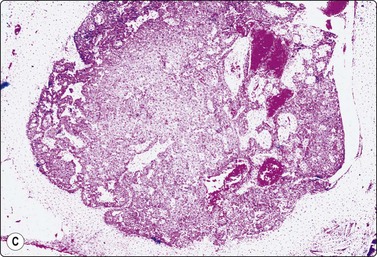
Fig. 7.42 Intracystic papillary carcinoma
(A) True papillary tissue fragment with fibrovascular stromal core from thick part of smear (MGG, IP); (B) Dispersed population of atypical epithelial cells with a mitotic figure near center; thin part of same smear as A. The presence of true papillary fragments precluded an unequivocal malignant diagnosis (MGG, HP); (C) Corresponding tissue section. There was no evidence of invasive carcinoma (H&E, LP).
Benign epithelial hyperplasia with papillomatosis may also have some of the above features and may be selected for excision.
Cytological atypia is often present to a variable degree in smears from papillary lesions. Although increasing cellularity with nuclear crowding, nuclear enlargement, irregular nuclear shape, and loss of cell cohesion increase the likelihood of malignancy (Figs 7.42 and 7.43), there is no clear distinction cytologically between papilloma with atypia, micropapillary DCIS, noninvasive intracystic papillary carcinoma and low-grade invasive papillary carcinoma. A suspicion of carcinoma can, of course, be expressed in the report if nuclear atypia is worrisome, but the definitive diagnosis is best deferred to histology.
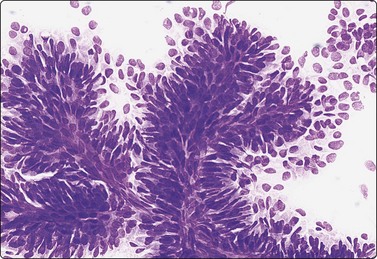
Fig. 7.43 Well-differentiated invasive papillary carcinoma
True papillary tissue fragments with fibrovascular stromal cores; crowded and stratified atypical columnar epithelial cells, moderate loss of cohesion (MGG, IP).
Cells aspirated from an atypical papillomatous lesion are sometimes poorly cohesive and mainly dispersed. Single bare bipolar nuclei may not be obvious. The similarity to the smear pattern of invasive carcinoma is enhanced by numerous single cells with intact cytoplasm, variable nuclear atypia, and sometimes mitotic activity (Fig. 7.42B). An unqualified malignant diagnosis could lead to unnecessary radical surgery of in situ or intracystic papillary carcinoma and should not be made in the presence of a papillary pattern as defined above, but be deferred to histology. Careful examination of the smears is important since papillary structures and/or palisaded columnar cells may be sparsely represented (Fig. 7.42A).
Solid, finger-like epithelial aggregates with a distinct ‘anatomical’ border of a row of cuboidal cells can be removed intact from a low-grade invasive carcinoma of particularly cohesive cells by FNB. They can be mistaken for fragments of papilloma (Fig. 7.44). However, they do not have a central stromal core, the cells are not columnar in shape and they do not tend to ‘ball up’ into multilayered fragments. The distinction has clinical importance since the management of a papillary lesion is different from that of invasive carcinoma. A CNB may be necessary to solve the problem.
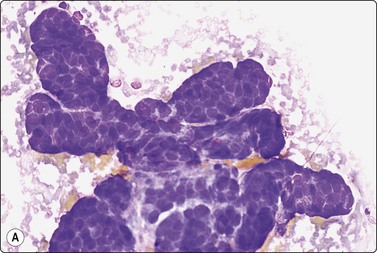
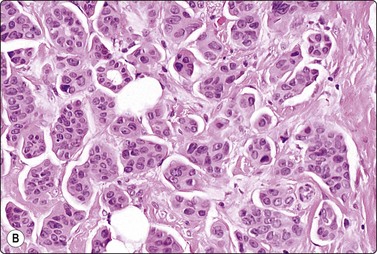
Fig. 7.44 Pseudopapillary pattern in duct carcinoma NOS
(A) Many finger-like epithelial fragments with sharp ‘anatomical’ borders but no columnar or palisading cells and no stromal cores (MGG, HP); (B) Corresponding tissue section. The cords of malignant cells are cohesive and are removed intact by the needle (H&E, IP).
Papillary-like epithelial fragments and a background of macrophages are sometimes found in fibroadenoma. The finger-like and branching structure of fibroadenoma epithelial aggregates do, however, differ from the more complex of heaped and folded aggregates of papillary lesions, and they do not have stromal cores. The distinction may be difficult in some cases. A background of numerous single bipolar nuclei and fragments of distinctly myxoid stroma strongly favors fibroadenoma.
Infarcting papilloma may produce a combination of findings closely mimicking malignancy.149 Necrotic or degenerating cells are noncohesive and there may be a completely dispersed population of epithelial cells with a background of debris (Fig. 7.45). Pyknotic change of the nuclei resembles malignant hyperchromasia. Squamous metaplasia may add to an appearance of pleomorphism and there may be other regenerative epithelial changes. The clinical background can be of help in correct assessment. These cases present with a sudden onset of bloodstained discharge and a rapid enlargement or appearance of a breast lump. Dispersed columnar cell forms indicative of a papillary lesion may be found in the smears and even if there are only necrotic columnar shapes, their presence should lead to caution. Papillary stromal cores, which may appear as hyaline rounded bodies, should also alert one to the possibility. Necrosis, infarction or hemorrhage, apart from the confluent masses of granular debris seen in DCIS, is uncommon in small low to intermediate nuclear-grade breast carcinoma and is more common in papilloma and fibroadenoma or associated with pregnancy or lactation. Previous aspiration of papilloma or fibroadenoma may also result in similar changes.
Subareolar duct papillomatosis/nipple adenoma
In subareolar duct papillomatosis (Fig. 7.46) or nipple adenoma cytologic material can be obtained either by FNB or by scraping the nipple, depending on the depth of the lesion. In scrape smears, squamous epithelium and some inflammatory cells are mixed with aggregates of mildly atypical epithelial cells of ductal type showing little tendency to dissociate. These are quite different from the single large malignant cells seen in Paget’s disease (see Fig. 7.70). Single, bare nuclei of benign type may be inconspicuous.
Complex sclerosing lesions/radial scars (Figs 7.47-7.49)
Common cytologic findings.93,150-153
These lesions are usually discovered by mammography and few are palpable. They are a complex mixture of elastosis, fibrosis and epithelial hyperplasia, often including marked intraductal hyperplasia. The hyperplastic epithelial elements are generally arranged in a radial fashion extending from a central sclerotic scar-like area, which may include some fatty tissue. A number of distorted small tubular epithelial structures are typically present in the central sclerotic area (Fig. 7.47). These are the main cause of difficulties in cytological diagnosis as they closely resemble low-grade tubular carcinoma both cytologically and histologically.
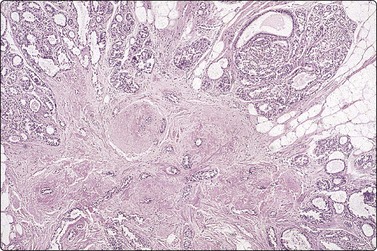
Fig. 7.47 Radial scar/complex sclerosing lesion
Tissue section of typical radial scar. Zone of epithelial hyperplasia around periphery; central fibrotic scar with small tubular structures resembling tubular carcinoma (H&E, LP).
The stellate appearance on mammography resembles that of invasive carcinoma. There are subtle radiological characteristics which allow the diagnosis to be suggested in most cases. The typical image pattern is reflected in the term ‘black star’. However, there is an overlap in appearances and about 10% of cases are given an incorrect cancer diagnosis by mammography. Most radial scars are excised for definitive histological diagnosis because of the difficulty in radiological diagnosis and the not unusual association with ADH and DCIS.
The most common smear pattern is of epithelial hyperplasia with or without atypia representing the zone of hyperplasia around the periphery of the lesion as seen in tissue sections (Figs 7.47 and 7.48). Epithelial atypia should not be ignored since low-grade DCIS is not uncommonly associated with radial scar. If the smear derives mainly from the central sclerotic area, small tubular or angular groups of mildly atypical epithelial cells may be seen associated with stromal elements, including elastotic fragments. This pattern closely resembles that of tubular carcinoma (Fig. 7.49). False-positive diagnoses by FNB have occurred in the investigation of radial scars.152 Although the findings may appear highly suspicious of malignancy, there is usually a coexistent clearly benign component of ductal epithelial fragments and single bipolar nuclei. A definite diagnosis of malignancy should not be made if there is an obvious benign component in the same smears.
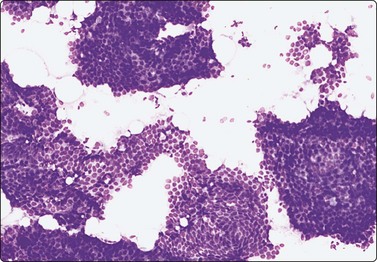
Fig. 7.48 Radial scar/complex sclerosing lesion
Cell-rich smear of many large sheets of bland epithelial cells; some single bipolar nuclei. The smear represents usual ductal epithelial hyperplasia at the periphery of the lesion (MGG, LP).
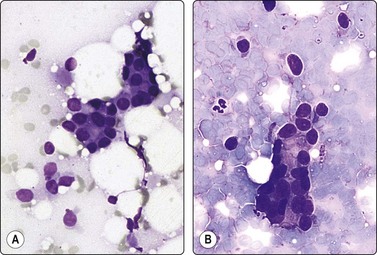
Fig. 7.49 Comparison of radial scar and tubular carcinoma
(A) Cells from center of radial scar; tubular/angular structure, mild nuclear atypia; occasional single bipolar nuclei; (B) Cells from tubular carcinoma; similar pattern (MGG. HP).
Since it is the overall tissue architecture that is diagnostic of radial scar, both FNB and CNB are of limited value. There are only a few series concerning core biopsy in radial scars. One series looking at 43 cases of radial scar on core biopsy showed an incidence of carcinoma at surgical excision of 16%.153 As mentioned above, lesions thought to be radial scars should be excised regardless of cytological findings. However, FNB or CNB may reveal unequivocal carcinoma, which obviously alters the clinical management.
Cytologic features of sclerosing adenosis (Fig. 7.50) may overlap with proliferative fibrocystic change and fibroadenoma.93 The smears of sclerosing adenosis are moderately to markedly cellular, consisting of small to large groups of benign epithelial cells.154 Small fragments of dense, hyalinized stroma are found attached to acinar sheets and groups of epithelial cells. Scattered single epithelial cells are present and may cause concern for malignancy. There may be some loss of cell cohesion and mild nuclear atypia, but single bipolar nuclei are usually present. As compared with fibrocystic change, sclerosing adenosis has more abundant cellularity, acinar sheets, single epithelial cells, and hyalinized stroma. Fibroadenomas, in comparison, display more of a branching pattern of epithelial sheets, large sheets and bipolar/oval, naked nuclei and large, hypocellular, fibromyxoid stroma.
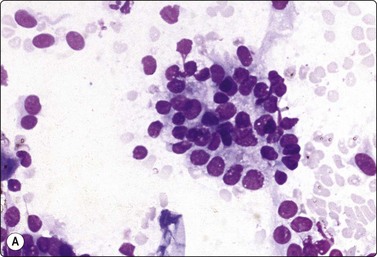
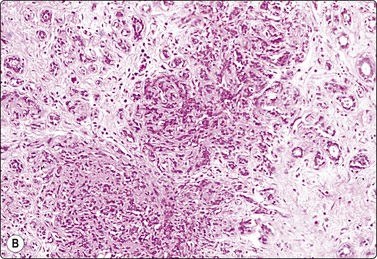
(A) Cluster of epithelial cells with a microacinar pattern; some single cells and bare bipolar nuclei (MGG, HP); (B) Corresponding tissue section (H&E, LP).
Mucocele-like lesions
Epithelial mucin is found in a group of breast lesions. The mucin can dominate the samples obtained by FNB. Mucocele-like lesions are a variant of fibrocystic disease, in which ducts are distended by mucin, leading to disruption of some ducts and extravasation of mucin into adjacent tissue. Mucocele-like lesions are most often detected by mammography due to the presence of microcalcifications in the same area. Other examples of mucinous lesions are mucinous fibroadenoma and mucinous DCIS. These lesions are further described in the section on colloid carcinoma, since they constitute an important and sometimes difficult differential diagnosis to colloid carcinoma (see p. 189).
Collagenous spherulosis
This is a stromal reaction most often encountered in areas of fibrocystic disease. It consists of spheres of hyaline basement membrane material closely resembling the hyaline globules of adenoid cystic carcinoma, associated with variable epithelial hyperplasia.155,156 It may be an incidental finding related to a palpable mass or to a mammographically detected lesion. Overlap with adenoid cystic carcinoma may be problematic (see Fig 7.83).157 Presence of bare oval nuclei, uniformity of hyaline globules capped by comma-shaped nuclei and benign appearance of epithelial cells favor collagenous spherulosis (see Fig. 7.84).
Benign mesenchymal lesions; rare lesions158
Most mesenchymal lesions occurring in the breast are not specific for this site and are described in Chapter 15 (soft tissues). Granular cell tumor can clinically and mammographically mimic breast cancer. The radiological image is of a stellate lesion graded 4 or 5, and the palpation findings are of a hard, irregular lump, not infrequently with skin dimpling. The cytological diagnosis is straightforward if smears are adequate and cells are well preserved, demonstrating the characteristic abundant granular cytoplasm (Fig. 7.51).159,160 However, the cytoplasm is fragile and may be dispersed in the background, leaving stripped nuclei that may be quite variable in size and shape, some with prominent nucleoli. This pattern could be mistaken for malignancy, particularly if the clinical and radiological diagnosis is of carcinoma. Furthermore, granular macrophage reactions may resemble granular cell tumor, and even immunocytochemistry is not always conclusive.161 Carcinoma with oxyphil differentiation has a granular cytoplasm and can cause differential diagnostic problems.
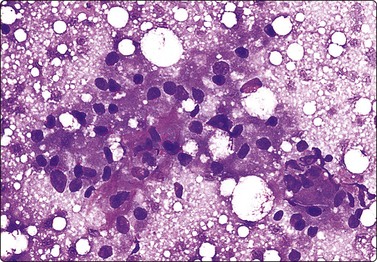
Fig. 7.51 Granular cell tumor of breast
Clusters of poorly cohesive cells with abundant, dense eosinophilic and granular but fragile cytoplasm with poorly defined cell borders; mildly irregular nuclei. This lesion was mammographically and clinically thought to be carcinoma (MGG, HP).
A spindle cell lipoma with marked atypia leading to a suspicion of malignancy has been reported.162 Our approach to unusual spindle lesions of the breast is to recommend local excision for definitive diagnosis. Cases of intramammary schwannoma diagnosed by FNB have been reported.163
Fibromatosis/nodular fasciitis can present as a stellate lesion in the breast, clinically and radiologically mimicking primary carcinoma.158 The yield of FNB can be scanty, but if smears are adequate a correct diagnosis based on the findings of fibrohistiocytic cells, singly and in tissue fragments as in soft tissue lesions of the same type, is possible (Figs 7.52 and 7.53). Reactive reparative processes such as suture granuloma, reparative granulation tissue following previous biopsy, and other scarring processes have similar features.164
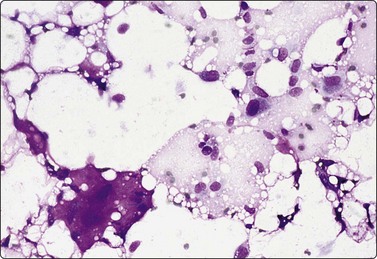
Fig. 7.52 Nodular fasciitis, breast
Scanty smear of dispersed spindle cells and a few inflammatory cells; fragments of fibrous stroma (MGG, IP).
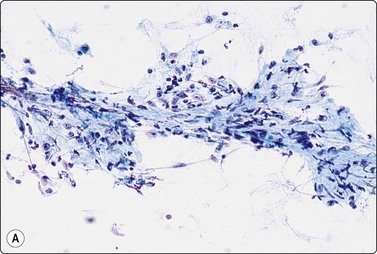
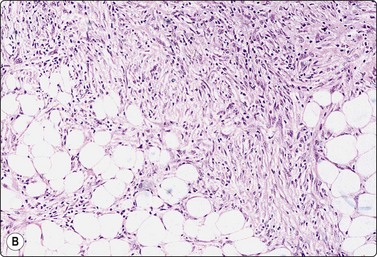
Fig. 7.53 Fibromatosis, breast
(A) Disrupted fragment of mesenchymal tissue of spindle cells and intercellular fibrous stroma; some single spindle cells (Pap, IP); (B) Corresponding tissue section (H&E, LP). This lesion was stellate on mammography and suspicious of carcinoma.
Myofibroblastoma is a rare neoplasm mainly occurring in males, characterized by a well-circumscribed mass of spindle cells with eosinophilic cytoplasm in a background of collagenous fibrous tissue. FNB findings are of a benign mesenchymal lesion composed of abundant, randomly arranged single and clustered benign spindle-shaped cells with scant cytoplasm and elongated or oval nuclei displaying a finely granular chromatin pattern and inconspicuous nucleoli.165
Primary carcinoma of the breast
Infiltrating ductal carcinoma of no special type (NOS) (Figs 7.54-7.56 and see Figs 7.2-7.4)
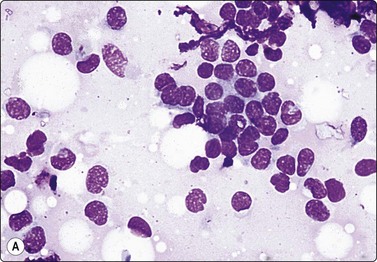
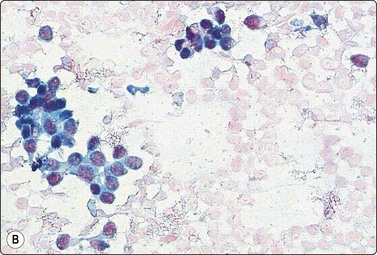
Fig. 7.54 Invasive duct carcinoma NOS, low grade
Clustered and single malignant epithelial cells, mild nuclear enlargement and atypia; absence of bipolar nuclei (A, MGG; B, Pap, HP).
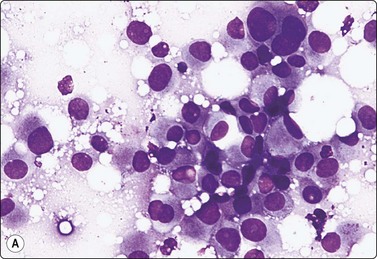
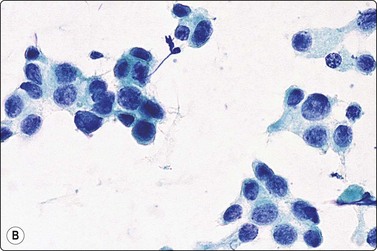
Fig. 7.55 Invasive duct carcinoma NOS, intermediate grade
Poorly cohesive malignant cells, single and in clusters; obvious nuclear enlargement and pleomorphism; irregular chromatin (A, MGG; B, Pap, HP).
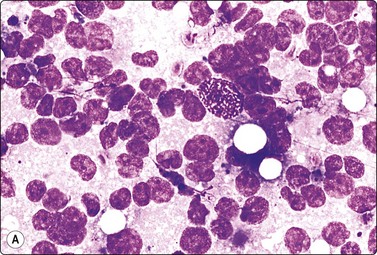
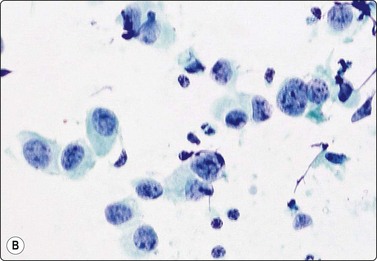
Fig. 7.56 Invasive duct carcinoma NOS, high grade
Mainly dispersed malignant cells, prominent nuclear enlargement and pleomorphism, coarse chromatin, an occasional mitotic figure; some lymphocytes and fibroblasts in the background but no bipolar nuclei (A, MGG; B, Pap, HP).
Criteria for diagnosis
Fine needle aspiration biopsy of most cancers yields a larger number of cells than normal glandular breast tissue. The yield from a scirrhous cancer may be very scanty, but the consistency of such a tumor felt through the needle is so characteristic that this is a suspicious finding on its own. A repeat FNB from the infiltrating edge of the tumor may be more rewarding. Occasionally, a core needle biopsy may prove necessary.
The overall smear pattern (cellularity, presence or absence of a bimodal cell population, cell cohesion, size and shape of cell aggregates, stromal components) is as important to the correct diagnosis as is the cytological detail. A scanty smear does not allow a proper assessment of the overall pattern. A repeat FNB, CNB or open biopsy is therefore mandatory if clinical or radiological findings are suspicious, or if cytological atypia is noted in a scanty smear. Architectural features are studied at low power. Aggregates of malignant cells are irregular, often with an angular or tubular shape. Reduced cell cohesion results in the presence of small clusters of cells, cells in single files, and single cells with intact cytoplasm, whereas single bare bipolar nuclei typical of non-neoplastic breast tissue are absent. Standard cytological criteria of malignancy – nuclear enlargement and pleomorphism, irregular nuclear membranes, irregular distribution of nuclear chromatin, prominent nucleoli, and crowding and overlapping of nuclei within aggregates of cells – are obvious in high-grade cancers but often subtle in low-grade tumors. Intracytoplasmic neolumina in atypical epithelial cells, sometimes with a ‘bull’s-eye’ inclusion, are an important criterion of malignancy, but are less commonly found in ductal carcinoma (see Fig. 7.74). In poorly differentiated carcinomas, dissociation of cells may be prominent and the smear pattern may resemble that of lymphoma but more often the malignant cells are seen both in aggregates and lying singly. Nuclei are irregularly distributed and overlapping within aggregates of tumor cells and there may be a tendency to microacinar grouping. Single carcinoma cells usually have well-defined cytoplasm but there are exceptions to this rule, particularly infiltrating lobular carcinoma (see below). Fibrous and adipose tissue fragments intimately associated with the cancer cells are suggestive of invasiveness. Stromal material is relatively scanty in most cancers. Nuclei of lymphocytes and fibroblasts derived from the tumor stroma may be present and these must not be mistaken for the bipolar, naked nuclei of benign breast tissue.
The presence of necrosis is strongly suggestive of malignancy. It is particularly characteristic of DCIS, and not often seen in invasive carcinoma. Tumor necrosis must be distinguished from condensed secretion in inspissated cysts and duct ectasia.
Small tumors, tumors obscured by surrounding fibrous tissue, or tumors adjacent to a dominant benign lesion such as a cyst or a lipoma can be missed by the needle. FNB guided by US or stereotaxis has greatly reduced the proportion of nonrepresentative biopsies but has not entirely eliminated this possibility. An overlap between the cytologic findings in fibrocystic disease and low-grade carcinoma can contribute to the problem.166
Artifacts (crush artifacts, artifactual disruption of cell aggregates, slow drying of MGG smears, drying artifacts in alcohol-fixed smears) may render interpretation difficult. Nuclear size and texture are altered by poor fixation. Smearing pressure may cause artifactual dissociation of cell aggregates mimicking the loss of cell cohesion in malignancy. Cells mixed with abundant blood or caught in blood clot tend to shrink, and cells dispersed in fat may become distorted and appear to be vacuolated. Aspiration of fat from around a tumor should be avoided, but at the same time cells are more easily obtained from the infiltrating periphery of a cancer.
Smears from low-grade carcinomas with small cells and a desmoplastic stroma (mainly infiltrating lobular but also some ductal carcinomas) may simulate the benign pattern at low magnification.167 However, the single cells are dispersed malignant cells which may have lost their cytoplasm but still differ from the bare bipolar nuclei of benign breast tissue. The nuclei have an irregular shape and there are usually some remnants of cytoplasm (see Figs 7.2-7.4 and 7.71).
Fibrosclerotic hypocellular lesions may not yield sufficient cells to allow a diagnosis by FNB. This is true of ‘scirrhous’ carcinoma as well as of sclerosed fibroadenoma. Diagnostic cell material is most likely obtained from the periphery of desmoplastic cancers, as from the peripheral parts of cancers with extensive central necrosis.
Duct carcinoma in situ (DCIS) causes special problems in cytological diagnosis. The cells of large cell DCIS of comedo type are readily recognized as malignant. The overall smear pattern of DCIS is fairly characteristic, but focal invasion can obviously never be ruled out by FNB. Small cell DCIS of papillary or cribriform type are usually reported as epithelial hyperplasia with atypia or as suspicious but not diagnostic of malignancy. Low-grade, tubular invasive carcinoma can also be difficult to recognize as malignant, particularly since naked nuclei of benign type, presumably derived from remnants of the lobular stroma, are often found in the smears.168
As described in previous sections, worrying cytological atypia can occur in several benign conditions (fibroadenoma, papilloma and epithelial hyperplasia, gynecomastia, etc.). Nuclear enlargement and anisokaryosis may be simply related to hormonal stimulation, but nuclei of irregular shape and with irregular contours (buds, indentations, sharp angles, folds, etc.) and chromatin irregularities are indicators of malignancy. To avoid overdiagnosis of malignancy, as a general rule, a definitive malignant diagnosis should not be given if smears include more than a few clearly benign elements in addition to the atypical cells.
If smears from a breast lesion show a malignant pattern not typical of the common forms of primary breast cancer, the alternatives, metastatic carcinoma, sarcoma or lymphoma, must be considered, since mastectomy may not be the appropriate treatment. Further investigation by immunostaining or core needle biopsy may be necessary to solve this problem.
The pitfalls in FNB and CNB of palpable breast lumps are summarized at the end of this chapter.
On CNB, the most significant pitfall in the diagnosis of an invasive carcinoma is confirming invasion in subtle invasive ductal and lobular lesions. Obtaining material from deeper levels can reveal subtle invasive foci, and the use of immunohistochemical stains such as cytokeratin can highlight invasive epithelial cells that have extended outside of the basement membrane.169 Another pitfall, as mentioned above, is avoiding the misdiagnosis of an invasive component when none exists. This can occur in high-grade DCIS where the stroma is desmoplastic and the ducts appear irregular in shape and mimic invasive foci. Stains for myoepithelial markers should be used to highlight the myoepithelial cells lining these irregularly shaped ducts and confirm the in situ nature of the lesion.
Ductal carcinoma in situ (DCIS) (Figs 7.57, 7.58, and see Figs 7.36, 7.39)41,138,170-173
The diagnosis of DCIS in tissue sections and CNB includes an assessment of nuclear grade (high, intermediate and low), growth pattern (solid, cribriform, micropapillary, intracystic papillary) and the presence of necrosis (confluent, comedonecrosis) and calcification. High nuclear grade lesions are biologically more aggressive and necrosis appears to impart a worse prognosis in low to intermediate nuclear grade lesions. A specific diagnosis or classification of DCIS cannot be made by FNB. High nuclear grade lesions are cytologically obviously malignant and are reported as high grade, but exclusion of invasion is clearly not possible. The low nuclear grade lesions provide greater difficulty and there is an overlap in appearances with epithelial hyperplasia with atypia. Most cases, however, are recognized as abnormal and recommended for excision.
High nuclear-grade DCIS, solid or comedo growth pattern (Fig. 7.57)
Usual findings
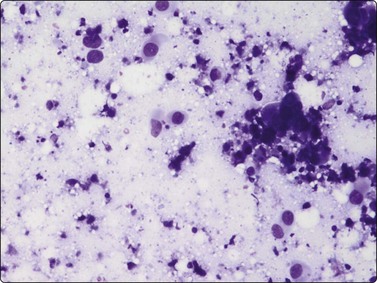
Fig. 7.57 High-grade DCIS (comedocarcinoma)
Pleomorphic malignant cells with a background of necrotic debris (MGG, IP).
The cells of DCIS of high nuclear grade (large cell, solid, comedo) are large, pleomorphic and show standard cytological criteria of malignancy. The cytoplasm may be abundant and eosinophilic, as in oxyphil cells. The diagnosis of carcinoma is easy. Abundant necrotic debris, often with calcium granules, represents the ‘comedo’ plugs seen in tissue sections. The absence of a well-defined palpable mass, the mammographic findings of characteristic calcifications without an associated tissue density and the cytologic features, taken together, strongly suggest high-grade DCIS. Invasive growth, if present, is usually suggested by the mammographic appearance, although mass-forming DCIS with periductal fibrosis is a pitfall for all diagnostic methods – radiology, clinical examination and morphology. Certain cytological features listed below are also suggestive of invasion. However, focal invasion can not be reliably excluded by mammography, FNB, or by CNB. Definitive assessment of invasive growth can only be made by excision and histological examination. On CNB, cases of high-grade DCIS can be surrounded by a desmoplastic stroma mimicking invasion. In these cases, the use of immunohistochemical markers for myoepithelial cell markers is useful to demonstrate definitive invasion.
Low-grade DCIS, cribriform, solid or micropapillary, noninvasive intracystic papillary carcinoma (Fig. 7.58, and see Figs 7.36, 7.39)41,99,138,146,172-174
Usual findings
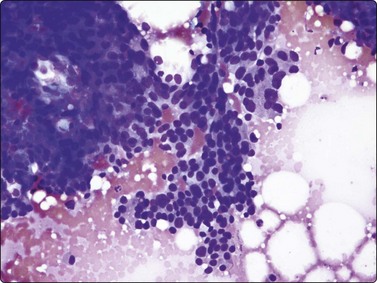
Fig. 7.58 Low-grade DCIS (cribriform)
Aggregates of mildly atypical cells in a three-dimensional pattern with ‘holes’ (left) (MGG, IP).
The findings have been described and the differential diagnosis with other lesions, mainly atypical ductal hyperplasia (ADH), has been discussed in the context of epithelial hyperplastic processes on page 175. The pattern also overlaps with that of papillary lesions. It is difficult to distinguish intracystic or intraductal papillary carcinoma from intraductal papilloma or florid papillomatosis. The presence of myoepithelial nuclei suggests benign hyperplasia or papilloma, while nuclear enlargement, nuclear pleomorphism, reduced cell cohesion and the presence of necrosis favor a malignant diagnosis. Correlation with the mammogram is essential and definitive diagnosis must usually be deferred to histology.
Intermediate-grade DCIS is difficult to separate with confidence from high- or low-grade forms, but the malignant nuclear features seen in FNB smears are closer to high-grade than to low-grade DCIS.
Predicting invasion175-180
Recent studies have suggested that the finding of tubular or angular epithelial structures, malignant cells adherent to fibrous stroma (Fig. 7.59), the present of intracytoplasmic lumina in malignant cells, fibroblast proliferation and fragments of elastoid stroma were predictive of invasion when associated with a malignant cell pattern. When two features were present the positive predictive value of invasion was 96%. Some workers have found the intimate adherence of malignant cells and fibrous stroma to be the single most useful feature predicting invasion. The presence of epithelial cells in fat tissue has not been confirmed to be a useful sign of invasive growth. Excluding focal invasion is obviously not possible due to the limited sampling. The presence of comedo-like necrosis is a useful sign of the presence of a DCIS component. Cell blocks were found to confirm invasion in 44% of breast FNBs in one study.180
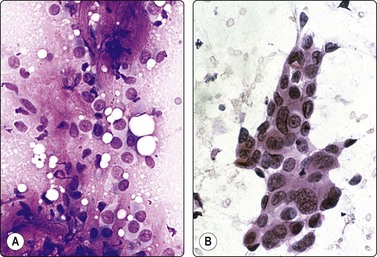
Fig. 7.59 Cytological findings suggestive of invasion
(A) Epithelial cells of low-grade carcinoma adherent to or embedded in fibrous stroma (MGG, HP); (B) A fragment of malignant epithelium with a tubular/angular configuration and spiculated edges. Smear from high-grade DCIS with focal invasion (H&E, HP).
The cytological findings described may be of some value in combination with mammographic assessment in deciding management. Clinical presentation is obviously also of significance. Chhieng et al. point out that 97.7% of palpable masses with unequivocally malignant cytology are invasive cancers.21
Cytological typing of primary breast carcinoma is of value in correlating the radiological findings with the expected histopathology. For example, medullary and mucinous carcinoma correlate well with a mammographically circumscribed lesion (rounded density), whereas invasive lobular carcinoma correlates with an asymmetrical density. Lobular carcinoma and DCIS tend to be more extensive than indicated by the mammogram. However, in the majority of cases, parameters other than tumor type, such as tumor size and grade, clinical stage, hormone receptor content and other biological factors, will determine the extent of surgery and the need for adjuvant therapy. Nevertheless, familiarity with all the various patterns of breast cancer is necessary to achieve a high level of cytological diagnostic accuracy.181 The basic diagnostic criteria for infiltrating duct carcinoma (the most common malignant pattern; p. 183) are to some extent applicable to all subtypes.
Tubular carcinoma (Figs 7.60-7.63)182-187
Usual findings
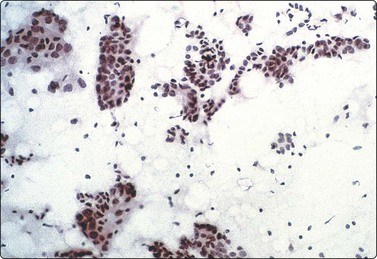
Tubular and angular epithelial fragments of mildly atypical cells; single files of cells; note some single bare nuclei in the background (MGG, LP).
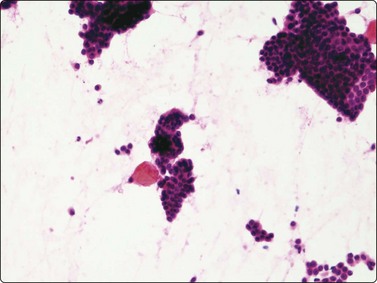
Angulated epithelial cell clusters of angulated epithelial cell clusters; variable nuclear atypia, some cells small bland, some moderately enlarged and atypical, scattered single cells (H&E, IP)
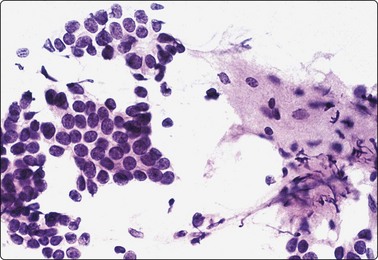
Fig. 7.63 Another case showing tubular and angular epithelial fragments; fibrous stroma, spindle cells and some debris (H & E, HP).
This tumor is a cytological challenge, commonly encountered as a small, mammographically detected lesion. The relatively uniform appearance of the nuclei, the preserved cellular cohesion and the presence of bare bipolar nuclei in up to a quarter of cases make a diagnosis of malignancy difficult. Most authors report only half of their cases as definitely malignant and there is a high false-negative rate. Fortunately, most of these lesions are stellate on mammography and suspicious by ultrasound and are selected for excision. Core biopsy is useful to confirm the diagnosis and to allow definitive treatment without previous excision.
Nuclear morphology is usually bland. Nuclear grooves186 and intracytoplasmic vacuoles are suggestive but not diagnostic features. Large cohesive aggregates of cells may represent a low-grade DCIS component. Many tubular carcinomas have a loose, cellular stroma similar to intralobular connective tissue. Smears may contain single, bare bipolar nuclei, as well as spindle cells of fibroblastic type and fragments of fibrous or elastotic stromal material. In the presence of a bimodal cell population, a definitive diagnosis of carcinoma can rarely be made cytologically.
Occasionally, tubular carcinoma may have larger, cohesive, monolayered sheets of epithelial cells which can be mistaken for fibroadenoma, since nuclear enlargement and atypia may occur in both conditions. This resemblance may be heightened by the presence of fragments of loose fibrous stroma. The sparsity of bare bipolar nuclei is against a diagnosis of fibroadenoma and should draw attention to the ‘atypical’ cell pattern.
Invasive cribriform carcinoma is another variant of low-grade breast cancer related to tubular carcinoma. In keeping with its histological growth pattern, smears from a cribriform carcinoma are usually highly cellular but the neoplastic cells are bland, cuboidal or columnar and show a tendency to an acinar, microglandular or cribriform arrangement (Fig. 7.64).
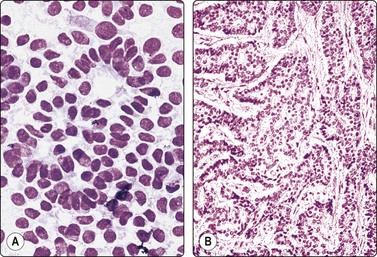
Fig. 7.64 Invasive cribriform carcinoma
(A) Cell-rich smear of poorly cohesive small monomorphic cells; some cells are columnar; suggestion of a microacinar/cribriform pattern (MGG, HP); (B) Corresponding tissue section (H&E, IP).
Columnar cell lesions have been associated with tubular carcinomas. On core biopsy, tubular carcinomas should be distinguished from radial sclerosing lesions and sclerosing adenosis. The entrapped ductules in the fibroelastic core of a radial scar may simulate invasive carcinoma, especially tubular carcinomas. The tubules in radial scars are usually elongated and flattened, which contrasts with the angulated tubular glands in tubular carcinoma. Sclerosing adenosis should also be included in the differential. Sclerosing adenosis shows expansion and distortion of lobules secondary to a disordered proliferation of acinar and ductal epithelial cells, myoepithelial cells and intralobular stroma. The lobulocentric nature of the process is helpful in distinguishing this lesion from an invasive carcinoma. The use of myoepithelial markers which are absent in tubular carcinomas can also be of use in distinguishing these lesions.
Mucinous (colloid) carcinoma (Figs 7.65 and 7.66)116,188-194
Usual findings
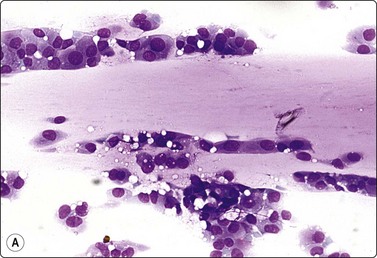
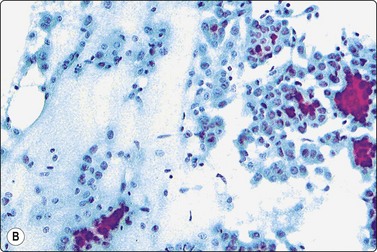
Moderately cohesive epithelial cells with abundant cytoplasm and moderate nuclear enlargement and atypia, suspended in mucus; note single files mainly in A. (A, MGG, HP; B, Pap, IP).
Mucinous carcinomas are usually round and well circumscribed, and may be clinically and mammographically mistaken for a benign lesion such as fibroadenoma or cyst. The tumor cells have abundant cytoplasm. Nuclear enlargement and pleomorphism can be relatively mild but the abundance of material, cell dispersal and absence of bare bipolar nuclei generally permit a definite diagnosis of malignancy. The mucin stains bluish violet with MGG. It is less conspicuous and rather pale in smears stained with H&E or Pap. Few mucinous carcinomas demonstrate intracytoplasmic mucin, and signet ring cells are more often seen in other variants, mainly in infiltrating lobular carcinoma.
‘Chicken wire’ blood vessels are often very prominent in smears of mucinous carcinoma (Fig. 7.66). They are suggestive but not diagnostic of this tumor, and can occur in other lesions, particularly in fibroadenoma.
We usually report a mucinous pattern in FNB samples as a ‘component’ of mucinous carcinoma since it is often only a focal change in a ductal carcinoma of the usual type.191 FNB selectively samples the softer mucinous elements.
The differential diagnosis is more complex in impalpable, mammographically detected lesions, as discussed below.
The relatively bland nuclear morphology may give a confusingly benign appearance to the smears, but high cellularity, loss of cell cohesion and the absence of single bipolar nuclei are suggestive of malignancy. Mucinous carcinoma must be included in the differential diagnosis of any lesion containing background epithelial mucin.
Mucinous material can be found in smears from several other lesions. In impalpable lesions, mucinous DCIS or ADH are included in the differential diagnosis. These present mammographically as microcalcifications or small opacities. Chicken-wire blood vessels are usually not seen in DCIS and the abnormal cells of DCIS or ADH are usually much more cohesive than in invasive carcinomas, forming large sheets.
Mucocele-like lesions (Figs 7.67 and 7.68) are also most often detected by mammography due to the presence of microcalcifications. They were described initially as benign lesions resembling salivary mucoceles, caused by extravasation of mucin from ruptured ducts with mucinous metaplasia. The lesions occur in the setting of fibrocystic disease. A spectrum of appearances in individual cases has been described, from benign areas to ADH, mucinous DCIS or mucinous carcinoma. Smears from cases of fibrocystic disease with intact ducts distended by mucin are fairly characteristic, showing monolayered sheets of bland, cohesive epithelial cells suspended in a large amount of mucus (Fig. 7.67). Calcium granules are often found. However, diagnostic difficulties can occur in mucocele-like lesions, in which there is rupture and extravasation of mucus into adjacent breast tissue. Smears may then include numerous mucinophages, which can closely resemble the atypical epithelial cells of mucinous carcinoma (Fig. 7.68).193,194
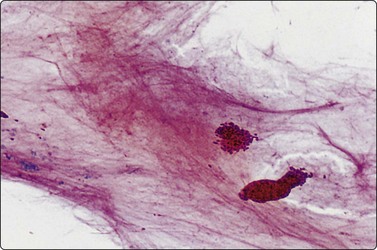
Fig. 7.67 Mucocele-like lesion
Background of abundant mucin; cohesive sheets of regular epithelial cells (H&E, LP).
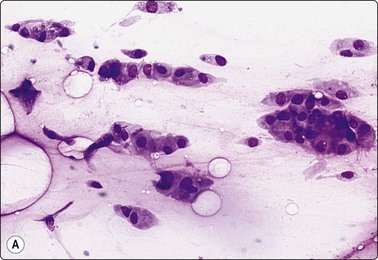
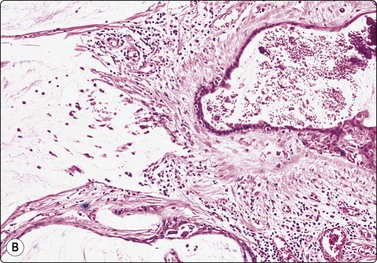
Fig. 7.68 Mucocele-like lesion
(A) Many mucinophages forming clusters and runs similar to the atypical epithelial cells of colloid carcinoma; background of mucin (MGG, HP); (B) Corresponding tissue section showing rupture of ducts distended by mucin; extravasation of mucin and a mucinophage reaction in the stroma (H&E, LP).
Sometimes the myxoid stromal ground substance of fibroadenoma and phyllodes tumor can be mistaken for epithelial mucin. However, the staining properties with MGG differ; epithelial mucin stains bluish violet, whereas myxoid ground substance stains pink/violet. The difference in color is not obvious in smears stained by Pap or H&E. There is a subtle difference in texture; epithelial mucin is homogeneous and structureless; myxoid ground substance is slightly fibrillar and often includes some fibroblastic spindle cells.
Metastatic mucinous carcinoma may be difficult to distinguish from a primary mucinous carcinoma.
FNB can cause bleeding into a mucinous carcinoma. We have seen several examples of complete infarction induced by FNB, making frozen section diagnosis difficult. Distinguishing mucinous lesions by core biopsy may be diagnostically challenging. The diagnosis of mucinous carcinoma on CNB is usually straightforward;192 however, the presence of extravasated mucin or a mucocele-like lesion on CNB warrants surgical excision to exclude a mucinous carcinoma. Extravasation of mucus and neoplastic cells caused by core needle biopsy has been observed; thus, if a CNB is done the core track should be included in the definitive surgical excision.
Ultrasound gel in smears may resemble mucin and is often accompanied by cellular artifact (see Fig. 7.12B).195
Medullary carcinoma (Fig. 7.69)196
Usual findings
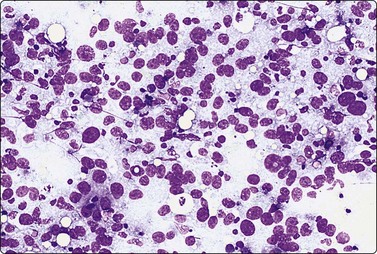
Numerous dispersed malignant cells with large, pleomorphic nuclei; many scattered lymphoid cells (MGG, IP).
Like mucinous carcinoma, medullary carcinoma tends to be mammographically rounded and well circumscribed and has a soft feel to the needle. The cytological pattern is not specific but is simply that of a high-grade carcinoma. The presence of numerous lymphocytes in the background may be due to focal lymphocytic infiltration and is not reliable evidence of true medullary carcinoma. Medullary carcinoma is an increasingly rare histological diagnosis, which takes circumscription, tumor cell cytology and lymphocytic infiltration into account. In our cytology reports, we simply comment on the high content of lymphocytes.
If the lesion is in the upper outer quadrant of the breast, consideration should be given to metastasis of high-grade carcinoma or melanoma in an axillary node. A predominance of lymphoid cells and a mixed population of lymphoid cells including germinal center material and macrophages favor this possibility. Large cell lymphoma also enters the differential diagnosis. Immunohistochemistry and flow cytometry can be useful in distinguishing these lesions.
Cells of both comedocarcinoma and medullary carcinoma have abundant cytoplasm and large, pleomorphic, obviously malignant nuclei. Necrotic debris and foamy macrophages are more typical of comedocarcinoma; loss of cell cohesion is greater in medullary carcinoma. Numerous lymphocytes are not usually seen in smears of high-grade DCIS. As compared to high-grade ductal carcinomas, medullary carcinomas more commonly have syncytial fragments, lack gland formation and have macronucleoli, but there is significant overlap.196
Paget’s disease of the nipple (Fig. 7.70)197,198
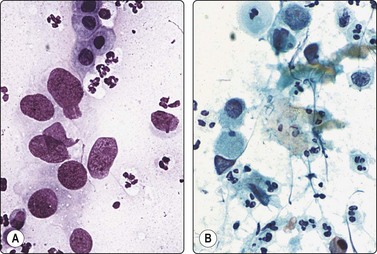
Fig. 7.70 Paget’s disease of the nipple
Nipple scrapings; mainly single, obviously malignant cells with abundant pale cytoplasm; some squamous cells; many inflammatory cells (A, MGG; B, Pap, HP).
Criteria for diagnosis
Scrape smears from the nipple are an excellent way to diagnose Paget’s disease. Any crust or exudate must first be removed carefully. The clean surface is then scraped with a scalpel blade held at a blunt angle. Both alcohol-fixed and air-dried smears are recommended. If there is any palpable mass in the breast, this should also be needled. The tumor cells are of high nuclear grade and usually dispersed or in small groups. Small whorls of cells or cell engulfment are frequently found.
Inflammation or ulceration from other causes may produce reactive changes in squamous cells. The reactive cells are usually cohesive and do not show a high degree of nuclear atypia. In nipple adenoma/subareolar duct papillomatosis (see Fig. 7.46), the epithelial cells form cohesive aggregates, they are smaller and more uniform in size and they do not show malignant nuclear characteristics.
A carcinoma arising from a major duct just below the nipple can erupt onto the nipple without infiltrating the epidermis in a pagetoid fashion. In such a case, scrape smears from the nipple contain numerous malignant cells with greater tendency to form syncytial clusters and there is not the intimate mixture of carcinoma cells and squamous epithelial cells typical of Paget’s disease.
In situ squamous carcinoma and melanoma are extremely rare in this site. Clinical appearances may be of help, or biopsy and immunohistochemistry for doubtful lesions.
Infiltrating lobular carcinoma (Figs 7.71-7.73)199-208
Usual findings
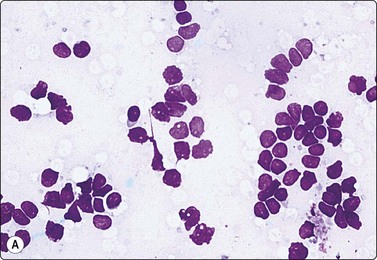
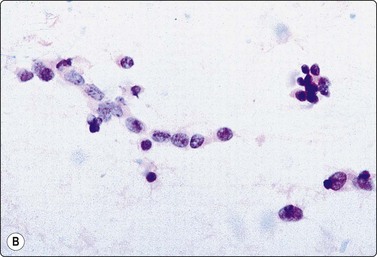
Fig. 7.71 Infiltrating lobular carcinoma, classic type
(A) Poorly cohesive cell clusters; single files; uniformly small nuclei with irregular shapes; nuclear molding; indistinct cytoplasm (MGG, HP); (B) Single file of cells with small nuclei of irregular shape (Pap, HP).
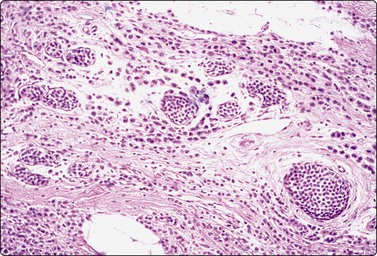
Fig. 7.72 Infiltrating lobular carcinoma, classic type
Tissue section corresponding to Figure 7.71 (H&E, LP).
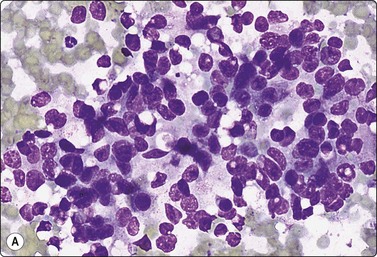
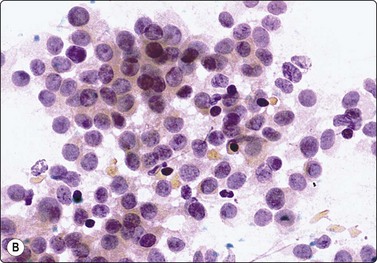
Fig. 7.73 Infiltrating lobular carcinoma, alveolar type
Cellular smears of poorly cohesive clusters of malignant cells with moderate nuclear atypia; difficult to distinguish from duct carcinoma of similar grade (A, MGG, HP; B, Pap, HP).
The criteria listed apply mainly to infiltrating lobular carcinoma of the classic type. The stroma is abundant, desmoplastic or fibrous, separating small groups and single files of neoplastic epithelial cells. The cells are not easily detached from the desmoplastic stroma and smears therefore tend to be poor in cells. The small cancer cells have a scanty and fragile cytoplasm, nuclear molding is a common finding in cell clusters and single files, and many single nuclei are stripped. In cells with intact cytoplasm, the nuclei tend to be eccentrically placed. The nuclei are only slightly larger than those of benign cells and vary little in size. However, nuclear outline is characteristically irregular, angular rather than rounded, with folds, buds and indentations, and this makes it possible to distinguish them from the single bipolar nuclei of benign breast tissue. Fragments of collagen may be seen associated with the tumor cells. The cells are often traumatised, presumably due to the force needed to detach them from the stroma. Non-neoplastic cells are more easily detached and residual benign ductal elements may be over-represented in the FNB sample.
The cells of some lobular carcinomas have the appearance of signet ring cells with intracytoplasmic neolumina and a central condensed mucin droplet (‘bull’s-eye’ inclusion). This is commonly seen in lobular carcinoma but is not specific and can be found also in duct carcinomas (see Fig. 7.74).
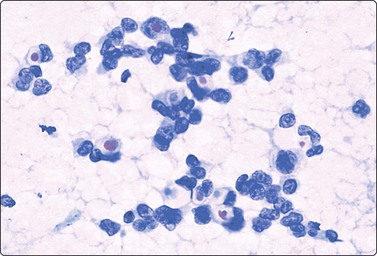
Fig. 7.74 Malignant cells with intracytoplasmic neolumina
Malignant cells with intracytoplasmic neolumina are most often seen in lobular carcinoma, but can also occur in invasive duct carcinoma NOS as in this case. Note ‘bull’s-eye’ inclusions (Pap, HP).
The cytology of the alveolar type of lobular carcinoma can show a rosette-like pattern (Fig. 7.73). Pleomorphic variants cytologically resemble poorly differentiated ductal carcinoma.
Lobular carcinoma is an important source of false-negative diagnosis in breast FNB. A smear from a lobular carcinoma, overall poor in cells with small clusters of cells and single naked nuclei, can easily be misread as a benign lesion. A scanty sample from a clinically suspicious lesion in an older woman may be due to lobular carcinoma and is always worth a second look, especially since mammography and US are less sensitive in this tumor type. At high-power examination the irregular, abnormal shape of the nuclei, including those of single cells with stripped nuclei, becomes evident. Epithelial cells with irregular, eccentric nuclei and dense cytoplasm and cells with intracytoplasmic lumina or mucin droplets are suggestive of the diagnosis.
Lobular carcinoma is characteristically poorly circumscribed and extends far into the surrounding glandular tissue. This growth pattern tends to leave residual non-neoplastic ducts within the tumor. Residual benign epithelial cells may be preferentially removed by the needle and may dominate a scanty smear to the extent that the malignant cells are overlooked.
Not surprisingly, the cells of low-grade lobular carcinoma of the alveolar type resemble those of hyperplastic acinar cells in pregnancy and lactation (compare Figs 7.8A and 7.73). Cells of both have pale vacuolated cytoplasm and rounded nuclei. Smears of alveolar lobular carcinoma are quite cellular, showing poorly cohesive multilayered clusters of cells, and lack the background of lipid secretion typical of lactating breast tissue.
The distinction between lobular and ductal carcinoma is not always possible. Some low-grade ductal carcinomas have equally small, relatively uniform neoplastic cells. Conversely, cells of lobular carcinoma of higher nuclear grade or of alveolar type are larger and have larger nuclei similar to those of ductal carcinoma NOS (compare Figs 7.5 and 7.73B).199,207 The distinction has some practical significance in the correlation with mammographic findings, as a lobular carcinoma is often more extensive than suggested by clinical and mammographic findings.
Dispersed atypical cells with intracytoplasmic lumina are a significant observation suggestive but not diagnostic of invasive lobular carcinoma. Aspirates from lobular carcinoma in situ (LCIS) and atypical lobular hyperplasia (ALH) may show these cells. Cells with intracytoplasmic neolumina are not specific for lobular neoplasia and may be found in ductal carcinomas, tubular carcinomas, colloid carcinomas, secretory carcinomas, medullary carcinomas and papillary carcinomas.208 However, even small numbers of dispersed signet ring cells or cells with intracytoplasmic lumina and a central targetoid (‘bull’s-eye’) mucin droplet are a strong indicator of a significant lesion requiring excision. This does not apply to similar cell changes in cohesive sheets, where vacuoles may be seen in benign hyperplasias, possibly within myoepithelial cells.204
Lobular carcinoma in situ/atypical lobular hyperplasia (LCIS, ALH) (Figs 7.74, 7.75)209-212
In the past, LCIS was a rare incidental finding in palpable breast lesions subjected to FNB. Few reports of its cytological features can be found in the literature. The cytologic findings in these cases include moderately cellular samples with tight or loosely cohesive clusters of atypical epithelial cells with mild nuclear atypia (Figure 7.74). The cells were small, with prominent nucleoli and occasional cells had intracytoplasmic lumina. In the majority of described cases a definitive diagnosis of LCIS could not be made but the presence of atypical cells warranted excision. In some cases, a diagnosis of infiltrating lobular carcinoma was made on aspiration biopsy209 as there is overlap in cytologic features between LCIS and infiltrating lobular carcinoma.
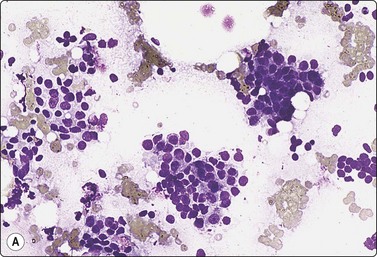
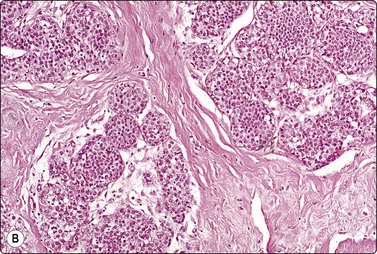
Fig. 7.75 Mass-forming lobular carcinoma in situ (LCIS)
(A) Loosely cohesive clusters of mildly atypical cells with small round nuclei and pale, fragile cytoplasm (MGG, IP); (B) Corresponding tissue section. There was no evidence of invasion in this case (H&E, IP).
Distinction of LCIS from invasive lobular carcinoma may not be possible in some cases even when clinical and radiological findings are correlated with cytology. The existence of mass-forming LCIS needs to be kept in mind when a cytological diagnosis of invasive lobular carcinoma is made, and confirmation by CNB before proceeding to radical surgery may be considered. A definitive diagnosis of LCIS is left to histopathologic examination. A CNB diagnosis of ALH and LCIS warrants surgical excision, as carcinoma is seen in 16% and 25% of the cases, respectively.212 The main differential on core biopsy of LCIS is the small cell solid type of low-grade DCIS or cancerization of lobules by DCIS. Immunohistochemistry with E-cadherin, a transmembrane glycoprotein, is helpful as it is positive in ductal lesions but lacking in LCIS and invasive lobular carcinomas.
Some uncommon variants of carcinoma213
Breast cancer in the male96-98,214-216
Most breast cancers in male patients are infiltrating duct carcinoma NOS, identical to the common duct carcinoma in women. DCIS also occurs in the male breast. Metastatic cancer is perhaps as common as primary carcinoma. Smears of gynecomastia can occasionally show worrisome atypia (see Fig. 7.9B) and false-positive diagnoses have been reported. Correlation with clinical and mammographic findings is therefore important, as in female patients.
‘Inflammatory’ carcinoma217
The clinical presentation of ‘inflammatory’ carcinoma is a diffuse increase in the consistency of the breast without a distinct mass, and thickening and erythema of the skin due to extensive intralymphatic spread of tumor causing lymph stasis and edema. Imaging may detect a mass lesion, making sampling easier. Random sampling from central parts of the breast or intracutaneous sampling with a 25-gauge needle introduced tangentially may produce enough malignant cells to allow a confident diagnosis. The tumor cells occur in small aggregates and show malignant pleomorphism. A diagnosis can often be made on small amounts of material. Inflammatory cells are not seen. CNB, especially with the mammotome device, may prove more useful in the diagnosis of inflammatory breast carcinoma as compared to FNB.218
Carcinoma with apocrine (oxyphil) differentiation (Figs 7.76A and 7.77)219,220
Invasive carcinoma of purely oxyphil type is rare. DCIS with prominent apocrine metaplasia is more commonly seen, and in MGG preparations cells of high-grade breast carcinoma of no special type may have an apocrine-like appearance. Cancer cells of oxyphil type have large, pleomorphic nuclei, a coarse and irregular nuclear chromatin and macronucleoli. In comparison to the common duct carcinoma cells, apocrine cells have more abundant cytoplasm, which is densely eosinophilic and granular and has more distinct cell borders. Apocrine (oxyphil) metaplasia of epithelial cells in cyst fluids, fibrocystic disease, and lesions with epithelial hyperplasia including fibroadenoma and adenosis often cause concern due to marked atypia and can be the cause of a false-positive diagnosis. Great caution should be observed if atypical apocrine epithelial cells are seen together with benign elements or if they are found in a clinically or mammographically benign lesion, regardless of the degree of cytological atypia. A pure population of severely atypical oxyphil cells with evidence of necrosis and a complete absence of benign epithelium and single bipolar nuclei are features indicating carcinoma. Granular cell tumor and alveolar soft part sarcoma should be included in the differential diagnosis of apocrine carcinoma.
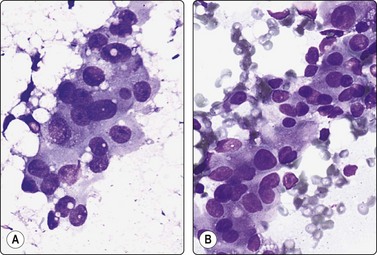
Fig. 7.76 Carcinoma with apocrine differentiation; comparison with atypical apocrine metaplasia
(A) Irregular cluster of malignant cells; abundant gray/blue, finely granular cytoplasm; pleomorphic nuclei; prominent nucleoli; (B) Similar atypical oxyphil cells from case of apocrine metaplasia in adenosis; note admixture with usual bland epithelial cells which have smaller nuclei (MGG, HP).
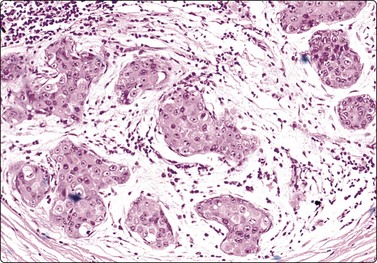
Fig. 7.77 Carcinoma with apocrine differentiation
Tissue section corresponding to Figure 7.75A. Case of high-grade DCIS with oxyphil malignant cells and focal invasion (H&E, IP).
Carcinoma of neuroendocrine type (Fig. 7.78)221
This is an uncommon variant of invasive breast carcinoma. Smears are cellular with mainly dispersed uniform plasmacytoid cells with a coarse granular nuclear chromatin pattern resembling the cells of a carcinoid or other neuroendocrine tumor. The cells may or may not stain positively with neuroendocrine markers such as chromogranin or synaptophysin and contain dense core granules by electron microscopy. The clinical behavior of these tumors does not seem to differ from that of the common duct carcinoma. However, the smear pattern of dispersed, rather uniform cells with scanty cytoplasm could be mistaken for lymphoid tissue, and the possibility of metastatic neuroendocrine carcinoma, for example of bronchogenic origin, has to be considered. Distinction from ductal carcinoma of no special type with dispersed cells and small, relatively uniform nuclei may not be possible without immunohistochemistry but seems to be of no clinical significance as the behavior is similar.
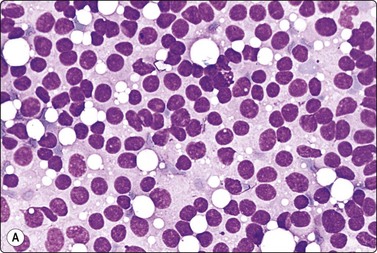
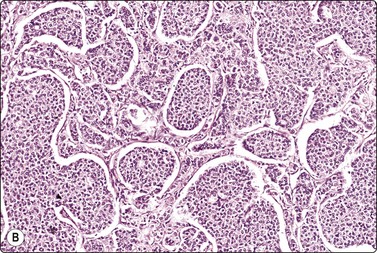
Fig. 7.78 Carcinoma of neuroendocrine type
(A) Cellular smear of totally dispersed small cells; relatively uniform nuclei; coarsely granular chromatin (MGG, HP); (B) Corresponding tissue section showing a carcinoid-like pattern; tumor cells were positive for chromogranin (H&E, IP).
Cytological findings in argyrophilic mucin producing spindle cell carcinoma and in carcinoid tumors in the breast have been reported.222-224
Breast carcinoma with osteoclast-like giant cells (Fig. 7.79)225
Invasive carcinoma with numerous multinucleated osteoclast-like giant cells is a rare type of breast carcinoma. On aspiration biopsy, the giant cells are intermingled with the malignant epithelial cells. Such tumors tend to be low grade. The giant cells are believed to be derived from fusion of stromal cells, and by electron microscopy are of histiocytic origin. Histiocytic giant cells can occasionally be found also in fibroadenoma and other benign lesions such as granulomatous mastitis.
Invasive micropapillary carcinoma (Fig. 7.80)226,227
Invasive micropapillary carcinoma is another uncommon variant of breast cancer thought to show a more aggressive behavior due to the predilection for lymphatic spread. The findings in smears closely parallel the pattern seen in tissue sections. The smears are of high cellularity with three-dimensional clusters forming rounded balls, morules and micropapillary fragments without a stromal core in addition to malignant discohesive cells. The cells show a moderate degree of nuclear atypia. Psammoma bodies, malignant apocrine cytology and focal mucin background have also been described.
Clear cell (glycogen-rich) carcinoma (Fig. 7.81)228
Smears are hypercellular with loosely cohesive syncytical groups and single malignant cells. The malignant cells have abundant, pale cytoplasm and moderate to marked nuclear pleomorphism with prominent nucleoli. A tigroid background can be noted. PAS-positive, diastase-sensitive material compatible with glycogen can be seen. The clear cell pattern is much more obvious in paraffin sections due to the high glycogen content. A similar pattern may also be seen in lipid-rich carcinoma.229
Secretory carcinoma (Fig. 7.82)230,231
Secretory carcinoma is an uncommon variant of ductal carcinoma with a favorable prognosis. Smears are moderately cellular with a background that contains abundant secretion. The cells are arranged in sheets or three-dimensional clusters with small round nuclei and abundant cytoplasm and intracytoplasmic vacuoles. The cells stain positively with a PAS stain.
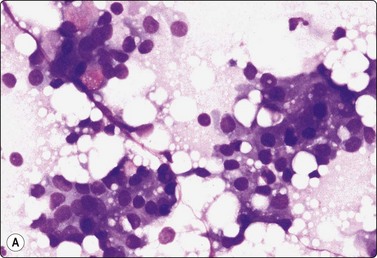
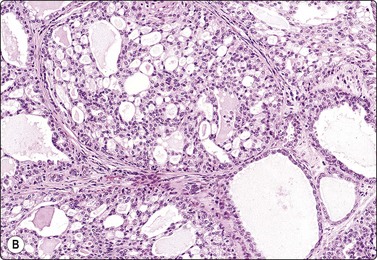
(A) Loose clusters of epithelial cells; some single cells; abundant vacuolated cytoplasm; rounded moderately atypical nuclei; some signet ring forms with pink secretory droplets (upper left); no bipolar nuclei (MGG, HP); (B) Corresponding tissue section (H&E, IP).
Case reports describing cystic hypersecretory carcinoma of the breast have been reported in the literature.232
Adenoid cystic carcinoma (Fig. 7.83)233,234
The prognosis for this rare tumor is significantly better than for most other invasive carcinomas. The cytological pattern is identical to that of adenoid cystic carcinoma in other sites, such as salivary glands, lung, etc. Hyaline stromal globules resembling those of adenoid cystic carcinoma can also be associated with collagenous spherulosis, a benign lesion confined to lobular acini or ductules that is seen incidentally with benign proliferative breast lesions (Fig. 7.84). The epithelial cells of collagenous spherulosis are small and uniformly bland whereas the cells of adenoid cystic carcinoma have a high N : C ratio and enlarged, hyperchromatic nuclei with a coarse chromatin and an irregular nuclear membrane. Bipolar nuclei are present in collagenous spherulosis, in contrast to adenoid cystic carcinoma.
Metaplastic carcinoma (Figs 7.85 and 7.86)235,236
Some breast carcinomas have a mixed pattern showing metaplasia and variable differentiation. Metaplastic carcinoma is defined as a tumor that has two distinctly different components. There are several variants of ‘metaplastic’ carcinoma. Spindle cell or sarcomatoid carcinoma closely resembles soft tissue sarcoma (Fig. 7.85). The differential diagnosis between malignant phyllodes tumor and metaplastic spindle cell carcinoma can be particularly difficult both in smears and in core needle biopsy specimens. In malignant phyllodes tumors the stromal overgrowth may be so abundant that it is impossible to distinguish from a pure spindle cell mesenchymal tumor. Sarcomatoid metaplastic carcinoma is confirmed by positive staining of the spindle cells for cytokeratin. Chondroid or osteoid stromal matrix can be found focally in some metaplastic carcinomas (Fig.7.86).
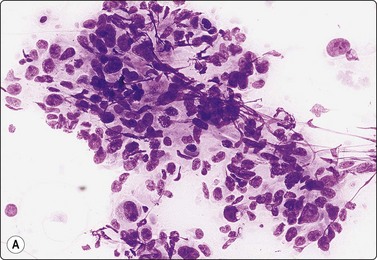
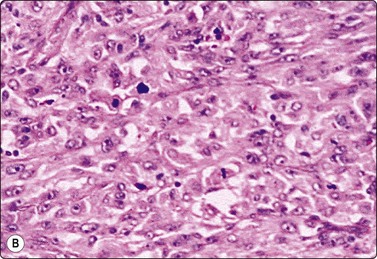
Fig. 7.85 ‘Metaplastic’ carcinoma
(A) Tissue fragment of poorly cohesive pleomorphic spindle cells suggestive of sarcoma. The cells were positive for cytokeratin and there were more obvious malignant epithelial cells in other parts of the smear (MGG, IP); (B) Corresponding tissue section (H&E, IP).
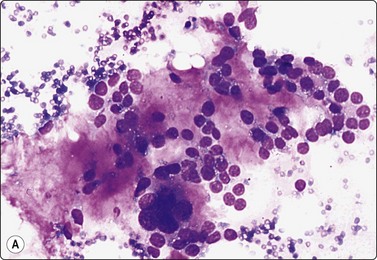
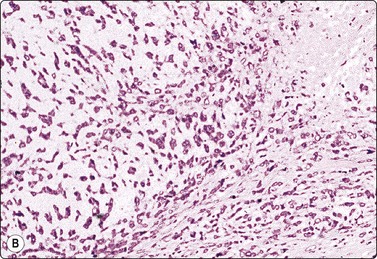
Fig. 7.86 ‘Metaplastic’ carcinoma
(A) Carcinoma with chondrosarcomatous areas; note the dense, brightly staining chondroid ground substance (MGG, HP); (B) Corresponding tissue section (H&E, IP). This tumor also contained sarcomatoid spindle cell areas and foci of anaplastic carcinoma.
Squamous differentiation is sometimes seen in poorly differentiated duct carcinoma. Low-grade adenosquamous carcinoma has been described.237,238 Pure squamous carcinoma has been diagnosed by FNB,239 including very well-differentiated forms.240 Cystic change may occur. The alternative of a secondary tumor should be excluded clinically before definitive management is decided.
Metastatic malignancy241,242
Metastatic tumor of extramammary origin can occur in the breast, presenting clinically as a solitary lump suggestive of primary breast carcinoma. If the cytological pattern does not fit any of the recognized subtypes of primary breast cancer, the possibility of metastatic malignancy has to be considered. The most common malignant tumors metastasizing to the breast are lymphomas, followed by malignant melanoma and lung carcinoma. Other sites include the ovary, the kidney, the uterine cervix and rarely from sarcomas.
Lymphoma/leukaemia (Fig.7.87A)243
Malignant lymphoma rarely presents as a primary breast tumor. The differential diagnosis of malignant lymphoma and reactive lymphoid tissue is described in Chapter 5. Large cell lymphoma may closely resemble poorly differentiated breast carcinoma with a completely dispersed cell population. In large cell lymphoma, many of the cells have a basophilic cytoplasm, an eccentric nucleus with one or multiple nucleolei, and a perinuclear halo. The presence in the background of round basophilic cytoplasmic fragments (lymphoglandular bodies) is a useful indicator of the lymphoid nature of the cells. Breast carcinoma of neuroendocrine type also needs to be considered in the differential diagnosis of lymphoma of small cell type. Leukemia, particularly myeloid, may give rise to mass lesions in the breast.
Sarcoma
Phyllodes tumors have been described on page 171. Malignant phyllodes tumor can be difficult to distinguish from soft tissue sarcoma. Smears contain fragments of highly cellular mesenchymal tissue and dispersed spindle cells showing a variable degree of nuclear atypia and pleomorphism. In some tumors the pattern is obviously malignant, like a spindle cell sarcoma (see Fig. 7.31B); others may have a relatively bland cytology, and the malignant character is suggested by high cellularity and invasive growth (tissue sections). Sheets of epithelial cells may or may not be present, depending on selective sampling. As a rule, the spindle cell component dominates the smears. The epithelial component may appear atypical but does not show malignant criteria. The histological pattern is often variable within the same tumor and a single FNB sample may not be representative, particularly if the tumor is large.
Distinction of primary sarcoma from sarcomatoid carcinoma or malignant phyllodes tumor will generally not be possible by cytomorphology alone and requires immunocytochemical study. An example of primary leiomyosarcoma, which clinically presented as a primary breast cancer, is shown in Figure 7.88. The FNB was reported as a malignant sarcomatous spindle cell tumor. Liposarcoma of the breast diagnosed by FNB has been reported.244
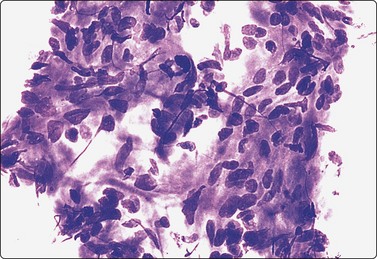
Fig. 7.88 Leiomyosarcoma of breast
Tissue fragments of cohesive, moderately atypical spindle cells in a fibrous stroma. FNB of breast mass clinically suggestive of primary breast cancer, reported as low-grade malignant spindle cell tumor inconsistent with primary breast carcinoma. No alternative primary found; leiomyosarcoma confirmed histologically (MGG, HP).
In angiosarcoma of the breast, aspiration yields plenty of blood, and tumor cells may be few in numbers and difficult to find.245,246 This is particularly the case with low-grade tumors, which consist of wide, anastomosing vascular channels lined by atypical endothelial cells showing little tendency to form solid proliferations. High-grade, more solid tumors are easily recognized as sarcomas. The tumor cells are spindle shaped, they have an attenuated basophilic cytoplasm without distinct borders and dark pleomorphic, elongated or plump spindle nuclei. Most cells occur in syncytial clusters but some are single (Fig. 7.89). There may be a suggestion of vascular structures. The presence of fat tissue fragments in smears is due to the neoplastic vascular channels widely invading adipose tissue. Pitfalls in diagnosis include misinterpreting the spindle cells as derived from benign granulation tissue or other spindle cell lesions, and overlooking a few abnormal cells in a bloodstained, apparently unsatisfactory smear.
Protocols for management are designed to maximize detection of carcinomas while maintaining a low benign to malignant ratio for excised lesions.
Some clinicians suggest that FNB of high-suspicion lesions is of no value because they will be excised whatever the result. However, there is a small but significant number of benign lesions, for example radial scar, nodular fasciitis fibromatosis and granular cell tumor, in this category. Furthermore, preoperative diagnosis gives a much higher likelihood of adequate clearance of cancer at the first operation.247
Summary of main diagnostic pitfalls in breast FNB and CNB44,169,248
Conditions in which there is a risk of false-positive diagnosis
1 Papillary lesions
The distinction between intraductal papilloma/florid papillomatosis, intracystic papillary carcinoma/papillary DCIS, and well-differentiated invasive papillary carcinoma is difficult in smears. Infarcted papilloma may closely mimic carcinoma. In general, a definitive cytological diagnosis of malignancy should not be made in papillary lesions but left to histology. Sclerosed papillomas can mimic carcinoma on core biopsy, and immuohistochemical stains for myoepithelial cell markers should be utilised to prevent a false-positive diagnosis (Fig. 7.90).
2 Epithelial hyperplasia with nuclear atypia
Highly cellular smears showing some loss of cell cohesion, and variable nuclear atypia (enlargement and anisokaryosis) can raise a suspicion of malignancy, but the presence of clearly benign elements, in particular of single bare bipolar nuclei, should prevent a cancer diagnosis.
3 Radial scar/complex sclerosing lesion
Groups of atypical epithelial cells derived from the central scar may closely resemble tubular carcinoma, but the presence of benign epithelial cells and single bare bipolar nuclei precludes a malignant diagnosis. Immunohistochemical stains for myoepithelial markers on CNB can help in distinguishing radial scars from tubular carcinomas.
4 Fibroadenoma
Epithelial atypia can be extremely worrisome in some fibroadenomas. Again, the presence of single benign bare bipolar nuclei and sheets of benign epithelium should prevent a false-positive diagnosis. The myxoid stroma characteristic of fibroadenoma is a helpful sign, but similar stroma can occasionally be found in carcinoma of no special type.
5 Regenerative epithelial atypia
In the presence of inflammatory cells, particularly of polymorphs, epithelial atypia should be interpreted with great caution. Regenerative atypia can be prominent in chronic mastitis, granulomatous inflammation and in fat necrosis.
Post-radiation atypia is a special problem which requires experience in the follow-up of postoperative irradiation for breast cancer.
6 Pregnancy and lactation
The smear pattern of dispersed cells resembles cancer at low power but the uniformly round nuclei, the bland nuclear chromatin and, above all, the lipid secretion in the background prevent misdiagnosis.
7 Atypia of ductal epithelium in cysts
Ductal epithelial cells of oxyphil (apocrine) type seen in cyst fluid can look very atypical. If the fluid does not contain altered blood or debris, and if there is no residual lump after evacuation of the fluid, there is practically no likelihood of malignancy.
Conditions in which there is a risk of making a false-negative diagnosis
1 Tumors with central necrosis or sclerosis
Smears from such lesions may be practically acellular. The distinction between scirrhous cancer and sclerosed fibroadenoma, and between necrotic cancer and duct ectasia, may require CNB or excision.
2 A small carcinoma next to a dominant benign lesion
A small cancer situated adjacent to a ‘lipoma’, a cyst or a lumpy fibroadenosis can easily be missed by FNB or CNB. Radiological guidance (mammography or US) is essential.
3 Complex proliferative lesions
Representative sampling can not be assured in poorly defined complex lesions with epithelial hyperplasia with and without atypia. Such lesions can include foci of in situ or even invasive carcinoma not sampled by FNB or CNB. Close correlation with clinical and mammographic findings is crucial.
4 Low-grade ductal carcinoma
Tubular carcinoma can be a problem, since single, bare, stromal nuclei of benign type are often present in smears from such tumors, and epithelial atypia may be minimal. In most cases, the overall architectural pattern is sufficiently atypical to suggest an open biopsy. Most tubular carcinomas are stellate lesions radiologically, with a high suspicion of carcinoma. Mammography may be more effective than cytology in selecting cases for excision. Another problematic lesion is low-grade DCIS in which fully developed criteria of malignancy are not usually present; however, most cases are diagnosed as ‘atypical’ or suspicious, with a recommendation of excision.
5 Lobular carcinoma and small cell ductal carcinoma
These lesions are difficult to detect radiologically because they infiltrate diffusely. Cells of infiltrating lobular carcinoma often have uniformly small nuclei and the cell yield is poor due to the highly desmoplastic stroma. The irregular shape of the nuclei, the tendency to form single files or clusters with nuclear molding and the absence of single, bare bipolar nuclei are diagnostic features. Low-grade duct carcinoma NOS may also have uniformly small neoplastic cells. Irregularity of nuclear outline and intracytoplasmic neolumina are helpful clues.
Other issues
1 High-grade DCIS
A combination of large cohesive clusters of malignant cells with a high nuclear grade and a background of necrotic and calcific debris allows a component of high-grade DCIS to be suggested. This finding correlates with pleomorphic or branching calcifications on mammography. FNB cannot reliably exclude invasion. Immunohistochemical stains should be used in cases of high-grade DCIS on CNB to avoid overcalling invasion.
2 Mucinous lesions
A number of lesions may be associated with mucin in smears: invasive mucinous carcinoma, mucinous DCIS or ADH, mucocele-like lesions, and mucinous fibroadenoma. The cell content associated with the mucin is the best guide to the nature of the lesion. Similar problems are seen with CNB.