CHAPTER 15 Soft tissues
Clinical aspects
The use of FNB and cytodiagnosis in the primary evaluation of a mass lesion in the soft tissues has become an accepted diagnostic modality using the cytodiagnosis as the morphological basis for a discussion of therapeutic options. A pretreatment cytological diagnosis offers several advantages. Patient anxiety can be relieved by providing an instant diagnostic suggestion followed by a discussion of therapeutic options based on the cytodiagnosis in combination with clinical and, at times, radiological data. FNB has a minimal risk of complications and the risk of tumor spread is negligible. FNB also allows easier collection of material from different parts of large tumors than open and core needle biopsy, important in case of tumor heterogeneity. Furthermore, FNB is less traumatic to the tissues than open or core needle biopsy.
Due to the relative rarity of primary tumors of soft tissue and to the extraordinary range of different types of tumors that can arise in these sites, a case can be made for centralising the diagnostic work-up of soft tissue tumors to a multidisciplinary clinic in an orthopedic oncology center. A specialised clinic facilitates the accumulation of invaluable experience based on the consistent correlation of clinical, radiological, cytological and histopathological findings at the time of diagnosis, ancillary laboratory tests, the results of treatment and the long-term outcome. It is hardly possible for the individual pathologist to gain a comparable level of experience over his/her professional lifetime.
However, to be efficient, highly specialised clinics of this kind need to draw material from a large population and consequently are only established in large academic centers. It is not possible to refer all patients with a soft tissue tumor or mass to a multidisciplinary center, particularly since most of these lesions are benign (benign lipomatous tumors). There is still a need for an easily accessible diagnostic service to be available at the community level in order to facilitate the referral of patients with suspected soft tissue tumors to an orthopedic oncology clinic. Guidelines for referral have been proposed. The recommendations of the Orthopedic Tumour Centre at the Lund University Hospital, based on epidemiologic data and on the difficulties experienced in the management of sarcomas, especially deep-seated sarcomas which were initially biopsied or surgically treated outside specialist centers, may serve as an example. The recommendations, suggested in 1997 and still valid, are that all patients with a deep-seated soft tissue tumor irrespective of size, or with subcutaneous tumors larger than 5 cm should be referred untouched. An FNB may be performed but the smears should be reexamined at the center, if possible before the patients first visit.1
The place of FNAC in the investigative sequence
The diagnosis and classification of soft tissue tumors is one of the most difficult areas in surgical pathology. The relative absence of recognisable tissue architectural patterns in cytological preparation makes diagnosis by FNB even more difficult. However, open biopsy or inadequate surgical excision of a primary sarcoma may compromise fascial planes and necessitate more extensive (mutilating) surgery than if the tumor had been left intact.
The current use of FNB as the primary and, in a majority of cases, the definitive diagnosis of soft tissue tumors before treatment is based on the following points:
A broad categorisation of a soft tissue tumor as of primary soft tissue origin, benign, low-or high-grade malignant, is possible in the majority of cases if the multidisciplinary approach suggested above is taken. Although many pathologists are reluctant to attempt cytological subclassification of primary soft tissue tumors and tumor-like lesions, a basic knowledge of the cytology of the most common tumors is important. A wide variety of benign tumors/lesions as well as sarcomas have been characterised cytologically by comparative studies of aspirates from series of cases of the same tumor type and by correlative studies of FNB smears and histological sections. The tumors/lesions are listed in Table 15.1.
Table 15.1 Soft tissue tumors/lesions which have been cytologically characterised in smears
| Benign |
| Malignant |
Accuracy of diagnosis
The results of FNA cytology of soft tissue tumors are good overall if patients with tumors suspected of malignancy are referred to centers specialising in this field. Åkerman et al.2 reported their 20-year-experience of FNAC of soft tissue tumors as a preoperative tool in 1994. In their series of 517 cases, the aspirated material was insufficient for diagnosis in 6% and an erroneous cytological diagnosis was rendered in 5% of adequate smears, 85% of the benign tumors were reported as benign and 89% of the sarcomas were classified as malignant soft tissue tumors. Other centers have reported similar results. Many reports highlight the difficulty to correctly subtype sarcomas histogenetically and to differentiate between low-grade sarcoma and benign soft tissue tumors.3-5
Technical considerations
The biopsy technique is as for any palpable lesion. Local anesthesia is rarely needed. Multiple passes are usually necessary to obtain sufficient and representative material. Alcohol-fixed H&E- or Pap-stained smears and air-dried smears stained with MGG or Diff-Quik should be used in parallel. Wet fixation gives excellent nuclear detail, particularly of cells in microscopic solid tissue fragments, as is often the case with several types of sarcoma. Air-dried smears (MGG; Diff-Quik), however, provide more information about cytoplasm and stromal components.
The site of the biopsy should, if possible, be chosen in consultation with the surgeon responsible for the treatment. A single point of entry and tattooing the insertation point on the skin is helpful if the needle track is to be included in the surgical excision. Ultrasonography (US), computed tomography (CT) or magnetic resonance imaging (MRI) can be helpful in finding viable tissue and avoiding cystic and necrotic areas in extensively necrotic or cystic tumors. US guidance is also of great value in the biopsy of small, deep-seated (intra- or intermuscular) tumors. If sufficient cellular material can be sampled, immunocytochemical studies and chromosomal or molecular genetic analysis may give valuable diagnostic information.
The parallel use of FNB and core needle biopsy (CNB) in selected cases, both sampling methods performed by the cytopathologist, combines the advantages of both techniques. In our experience, a core needle with an outer diameter of 1.2 mm is sufficient and well tolerated by patients. FNB allows wider sampling of large tumors and an immediate assessment based on air-dried smears (especially Diff-Quik-stained), while the CNB samples facilitates evaluation of microarchitecture and in many cases provide more material for ancillary studies.6 The risk of tumor cell dissemination in the needle track is not greater for CNB than for FNB; it is negligible for both.
Cytological findings
Primary soft tissue tumors in general
Common characteristics
Fine needle biopsy samples from high-grade malignant sarcomas are usually abundant, whereas the yield from benign tumors and low-grade malignant sarcomas depends on the tumor type. Collagen-rich intercellular stroma, areas of hyalinisation, fibrosis and prominent vascularity influence the yield. Infrequently, single cells dominate the smears whether from benign tumors or sarcomas. Aggregates, bundles of cohesive cells or ‘microbiopsies’ are commonly present in smears, especially from sarcomas of various types. The cells may not form any discernible tissue architectural pattern or they may be oriented in fascicles or streams. Spindle cells which seem to radiate out from a central basement membrane core suggest a vasoformative tumor, solitary fibrous tumor, hemangiopericytoma or hemangiopericytoma-like synovial sarcoma. Bundles of parallel spindle cells are common in smooth muscle tumors, occasionally in tumors of Schwann cell origin, and occur at times in fibroblastic tumors. Palisading of nuclei can be seen in Schwann cell tumors and smooth muscle tumors and rosette-like figures (at times with a center of finely fibrillar material) in neuroblastoma and in Ewing’s family tumors.
Myxoid ground substance may have a more or less fibrillar structure and stains brightly violet/red/purple in MGG (epithelial mucus is homogenous blue/violet). It is less conspicuous and may be almost invisible in wet-fixed H&E- and Pap-stained smears. Collagen is also obvious in MGG as strands of red/violet dense material between cells or isolated in the background. Capillary vessels are prominent in tissue fragments of myxoid liposarcoma and fragments of vessels are often observed in the myxoid ground substance of myxofibrosarcoma. Inflammatory cells (nodular fasciitis) and mast cells (synovial sarcoma) should be looked for. Necrosis is suggestive of high-grade malignant sarcoma.
The cytoplasm of mesenchymal tumor cells is generally not as fragile as that of many epithelial tumor cells and lymphoid cells with the exception of the small round cell malignancies such as alveolar rhabdomyosarcoma, neuroblastoma and the Ewing’s family tumors, granular cell tumor and alveolar soft tissue sarcoma. Cell borders are usually indistinct and the cytoplasm may be drawn out into elongated processes or thin strands which anastomose with those of adjacent cells. Aggregates of cells have a syncytial cytoplasm. Nuclei tend to be eccentric in rounded cells. Spindle-shaped nuclei are common in smears from soft tissue tumors in general and are often present also in highly pleomorphic tumors. In synovial sarcoma and cellular myxoid liposarcoma (round cell liposarcoma) nuclei may be quite plump or rounded. Predominantly rounded nuclei are characteristic of granular cell tumor, alveolar soft tissue sarcoma, alveolar rhabdomyosarcoma and Ewing’s family tumors. In general nuclear pleomorphism is proportional to the grade of malignancy.7 Important exceptions occur, however. Anisokaryosis, marked variation in nuclear shape and multinucleation are often prominent in the various types of benign pseudosarcomatous soft tissue lesions as nodular fasciitis, proliferative fasciitis and myositis and pseudomalignant myositis ossificans. In such lesions, nuclear chromatin is bland and evenly distributed, but nucleoli may be large and prominent. Nuclear pleomorphism and chromatin clumping may be minimal or absent in some low-grade malignant sarcomas and even in some high-grade malignant sarcomas such as synovial sarcoma. The malignant potential in such tumors is mainly suggested by high cellularity, nuclear crowding, mitotic figures and occasionally by the presence of necrosis. Bizarre nuclei with lobulation, budding and nuclear satellites are common in high-grade tumors. Mitoses are mainly found in high-grade malignant sarcomas; nodular fasciitis is an important exception. Abnormal mitotic figures indicate high-grade sarcoma.
Comprehensive descriptions of the cytological patterns of primary soft tissue tumors can be found in two recent publications by Åkerman and Domanski and Gonzáles-Cámpora.8,9
Soft tissue tumors are usually classified histogenetically and in this section we use the classification proposed in the WHO fascicle Pathology and Genetics of Tumours of Soft tissue and Bone 200210 and Weiss SW, Goldblum JR, Soft tissue tumours, ed V.11
Fibroblastic/myofibroblastic tumors and fibrohistiocytic tumors
Benign tumors
Criteria for diagnosis
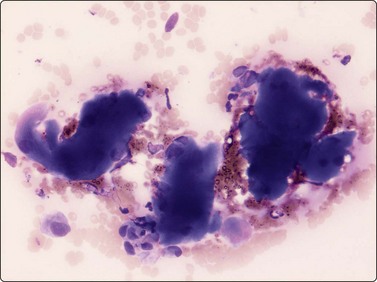
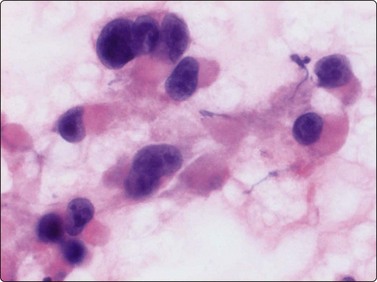
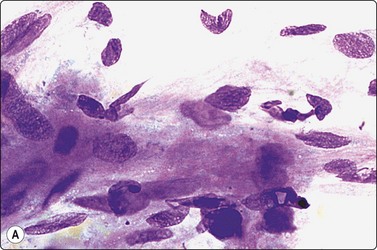
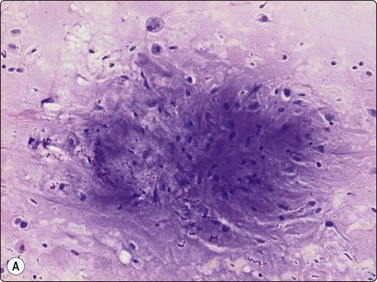
The pseudosarcomatous lesions are an important target for needling in this histogenetic group of tumors/lesions. Nodular fasciitis is among the commonest and the most frequently needled. Dahl and Åkerman reported 13 cases with cytology 1981;12 at present, our material comprises more than 70 cases, all with remarkably similar cytomorphology. In more recent investigations of the cytological features of nodular fasciitis, the results are similar to those of Åkerman and Dahl.13,14 The most important feature is the pleomorphism of the proliferating fibroblasts/myofibroblasts. Nuclei are predominantly spindly, but a proportion of cells have plump, ovoid or kidney-shaped nuclei. Bi-and/or multinucleated forms are always present and, if looked for carefully, ganglion cell-like binucleate cells with triangular shape and eccentrically placed nuclei are found (Fig. 15.1). A high cell content, nuclear pleomorphism, prominent nucleoli and the presence of mitoses may suggest malignancy, but the pale, bland nuclear chromatin is a clear indication of the benign nature of the lesion (Fig. 15.2). The correct diagnosis depends on the clinical presentation and the anatomical site (a rapidly, often tender subcutaneous nodule most frequently appearing in the upper extremity, trunk, head and neck) combined with such cytologic features as a myxoid background, actively proliferating fibroblasts/myofibroblasts and the presence of inflammatory cells.
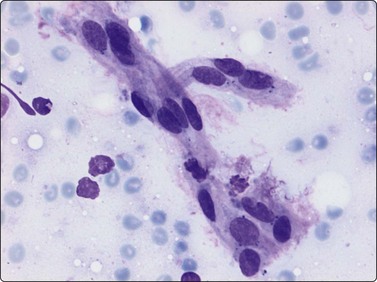
Proliferating fibroblasts embedded in a myxoid background; note binucleate cell with abundant cytoplasm and eccentric nuclei (ganglion cell-like) (MGG, HP).
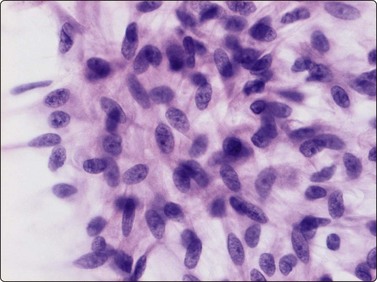
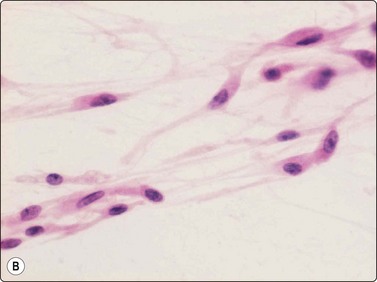
Poorly cohesive, proliferating fibroblasts; plump, oval, irregular nuclei, small nucleoli, bland chromatin; myxoid matrix not discernible in H&E-stained wet-fixed material (H&E, HP).
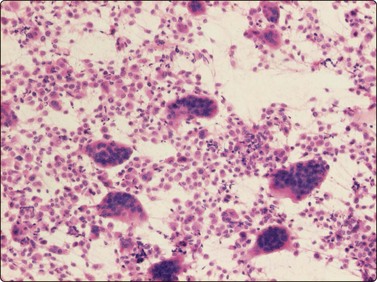
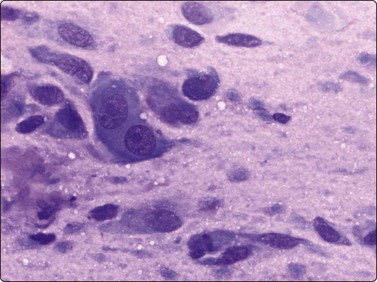
Proliferative myositis and fasciitis are less frequent pseudosarcomatous processes but are important with regard to the rapid growth (common to all these lesions) and their occurrence in children. Their cytomorphology is similar to that of nodular fasciitis although the myxoid matrix is less prominent and the ganglion cell-like cells are often numerous with very prominent nucleoli (Fig. 15.3). In proliferative myositis regenerating multinucleated muscle fibers are commonly present (Fig. 15.4). An important clinical sign is that these lesions, especially nodular fasciitis, can disappear spontaneously or diminish substantially in size within 3–4 weeks after needling.15,16 Another lesion which cytologically demonstrates prominent reactive cellular changes is pseudomalignant myositis ossificans (PMO). PMO is a rapidly growing lesion (intramuscular or subcutaneous), smears showing a mixture of proliferating fibroblasts/myofibroblasts, osteoblasts with prominent reactive changes and multinucleated giant cells of osteoclastic type (Fig. 15.5). In PMO an ossification in a zonal pattern is a typical find within 3–4 weeks and, according to our experience, spontaneous resolution is common. The cytomorphology in our series of five cases17 is similar to that described by Dodd et al.13
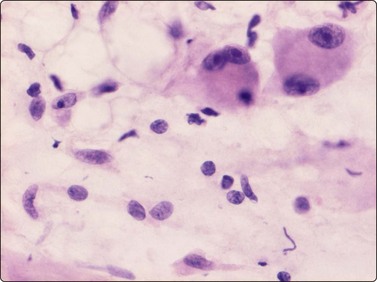
Fig. 15.3 Proliferative fasciitis
Ganglion cell-like cells are often numerous with very prominent nucleoli (H&E, HP).
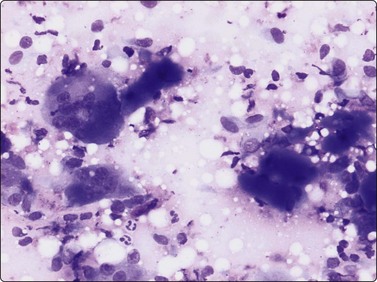
A mixture of proliferating fibroblasts/myofibroblasts, osteoblasts and multinucleated giant cells of osteoclastic type (MGG, LP).
Fibromatoses such as desmoid fibromatosis and palmar and plantar fibromatosis may partly resemble the pseudosarcomatous lesions, especially nodular fasciitis, in FNB smears. However, the nuclei are more consistently spindle and the marked nuclear pleomorphism in those lesions is not seen. Collagen fragments are common and myxoid ground substance unusual, as are inflammatory cells. Strands and clusters of spindle cells showing moderate anisokaryosis, and more or less acellular fragments of collagen are characteristic of desmoid fibromatosis, abdominal as well as extra-abdominal, in FNB smears (Fig. 15.6). When the tumor infiltrates striated muscle, muscle fragments and regenerating multinucleated muscle fibers are commonly seen (Fig. 15.7). The most important differential diagnoses are the rare low-grade malignant fibrosarcoma and monophasic fibrous synovial sarcoma. Smears from palmar and plantar fibromatoses (Dupuytren) can be surprisingly cellular, but the cells are uniformly spindled with bland spindled nuclei (Fig. 15.8). Recent summaries of the cytologic characteristic features of desmoid fibromatosis have been published 20038 and 2006.18
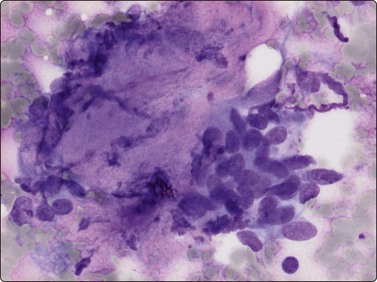
Fig. 15.6 Desmoid fibromatosis
The two typical components are shown here: a cluster of loosely cohesive ovoid or spindle fibroblasts and a fragment of collagenous stroma (MGG, HP).
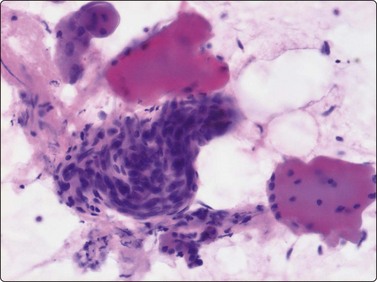
Fig. 15.7 Desmoid fibromatosis
Muscle fragments and regenerating multinucleated muscle fibers are commonly seen (H&E, HP).
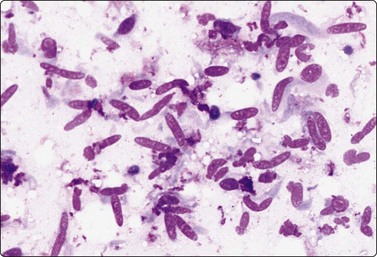
Fig. 15.8 Palmar fibromatosis (Dupuytren)
Relatively cellular smear of poorly cohesive spindle cells; bland spindle nuclei; no microarchitectural pattern; no obvious stromal material (MGG, HP).
Other benign fibroblastic/myofibroblastic and fibrohistiocytic tumors are solitary fibrous tumor (SFT) and hemangiopericytoma. According to recent investigations of these tumors, they are considered to be closely related, most probably representing two variants of the same entity.10 SFT most commonly occurrs in the pleura. SFT of the soft tissues is most often seen in adults as a deep-seated mass. In tissue sections the tumors are variably cellular and collagenous bands and hyalinisation are common findings. The fibroblastic spindle cells have no specific features and there is generally insignificant cellular atypia. A hemangiopericytoma-like vascular pattern is often present. The cytomorphology of SFT has been described in a few cases.19,20 Bland spindle cells arranged in tight cell clusters associated with ropy collagen, as well as dispersed cells often with stripped nuclei, seem to be the typical appearance in FNB smears (Fig. 15.9A). The tumor cells stain positively for CD34 (Fig. 15.9B) and occasionally for CD99 and bcl-2.
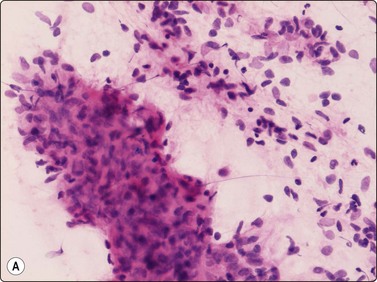
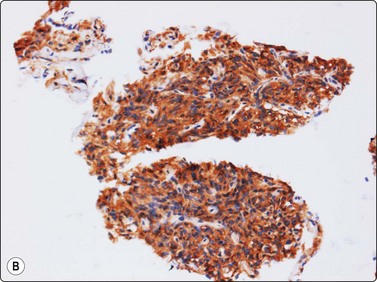
Fig. 15.9 Solitary fibrous tumour
(A) Bland, plump spindle fibroblastic cells arranged in tight cell clusters as well as dispersed cells associated with strands of collagenous stroma (H&E, IP); (B) Spindle cells staining positively for CD34 (cell block, immunoperoxidase, LP).
Hemangiopericytoma, earlier thought to be an example of a pericytic neoplasm, is in current classifications considered as a fibroblastic tumor. Hemangiopericytoma is most common in deep soft tissue. At present, it is considered as a diagnosis of exclusion since some other tumors, notably monophasic synovial sarcoma and mesenchymal chondrosarcoma, may exhibit the same vascular pattern of branching vessels (staghorn pattern) as hemangiopericytoma.10 The cytologic findings of hemangiopericytoma include branching vessel fragments; the tumor cells have bland ovoid or rounded nuclei and small cytoplasmic processes. Like SFT, hemangiopericytoma most often stains for CD34 and CD99. It is often difficult, sometimes impossible, to give a type-specific diagnosis of this tumor in FNB smears.
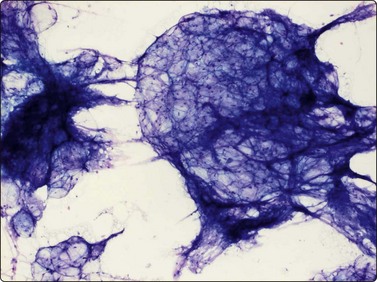
Elastofibroma is a slowly growing tumor, typically sited on the back near the scapula. The characteristic feature of elastofibroma is signs of faulty elastin fibrillogenesis. It is a hypocellular lesion composed of benign fibroblasts/myofibroblasts in a collagenous matrix with degenerated elastic fibers (Fig. 15.10). The typical cytologic findings in elastofibroma have been described in a small series of five cases.21
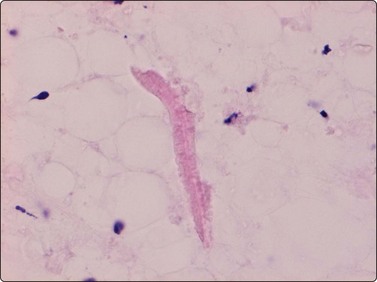
Two fibroblastic/myofibroblastic tumors of infancy, infantile fibromatosis colli (torticollis) and fibrous hamartoma of infancy, are occasionally needled. The typical smear of infantile fibromatosis colli, clinically often mistaken for malignancy, shows a mixture of spindle fibroblasts and regenerating mono- or multinucleated muscle fibers These regenerating fibers may be pleomorphic with prominent nucleoli, not to be mistaken for malignant cells (Fig. 15.11).22,23
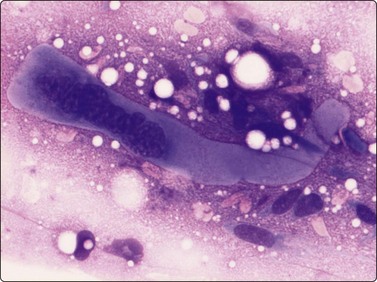
Fig. 15.11 Infantile fibromatosis colli
Single fibroblasts and a regenerating multinucleated muscle fiber (MGG, HP).
The rare fibrous hamartoma of infancy is a subcutaneous tumor-like mass in the upper arms, shoulder and axillary region. It is composed of a mixture of mature fat, strings of fibrous tissue and primitive mesenchymal cells. The cytologic findings in the few cases published24 are fragments of normal fat mixed with clusters or runs of bland spindle cells. We have had the opportunity to study the smears of two cases (Fig. 15.12).
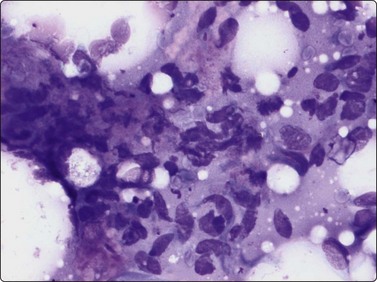
Fig. 15.12 Fibrous hamartoma of infancy
Normal fat cells mixed with clusters or runs of bland spindle cells (MGG, HP).
Tenosynovial giant cell tumor is a relatively common benign fibrohistiocytic tumor related to the tendon sheaths of the fingers and hands. Although the clinical presentation is quite characteristic, confirmation by FNB is often requested. Smears are relatively cellular and consist of mainly dispersed plump fibrohistiocytic cells with oval pale nuclei and bland chromatin. There is a variable number of scattered osteoclast-like multinucleated giant cells, which may be numerous (Fig. 15.13).25
Malignant tumors
Criteria for diagnosis
Malignant fibrous histiocytoma (MFH) was recognised in 1963 as a specific entity of probable fibrohistiocytic origin and was considered the commonest primary sarcoma of soft tissue. A number of subtypes were described, among which pleomorphic malignant fibrous histiocytoma was the most common. MFH is at present challenged as a specific histotype and the consensus is that MFH shows no evidence of histiocytic differentiation. On reexamination with extended immunohistochemistry and electron microscopy, many sarcomas initially diagnosed as pleomorphic MFH have been reclassified as dedifferentiated leiomyosarcoma, pleomorphic liposarcoma, pleomorphic rhabdomyosarcoma, soft tissue osteosarcoma or as high-grade malignant tumors of non-mesenchymal origin.26,27 Thus in current textbooks such as the WHO fascicle on soft tissue and bone tumors, the diagnosis pleomorphic MFH is reserved for a small group of pleomorphic sarcomas, which after thorough investigation show no specific histotype. The typical cytology of pleomorphic sarcoma of MFH type is described in two series.28,29 Most often, atypical spindle cells dominate the smears, but large atypical cells with more or less abundant vacuolated or foamy cytoplasm are usually also present and may be numerous, as well as multinucleated tumor giant cells (Fig. 15.14). Smears from both deep and subcutaneous tumors may contain small amounts of myxoid matrix. The diagnosis of giant cell malignant fibrous histiocytoma, formerly considered as a subtype of MFH, is currently reserved for undifferentiated pleomorphic sarcoma with numerous osteoclast-like multinucleated giant cells.10
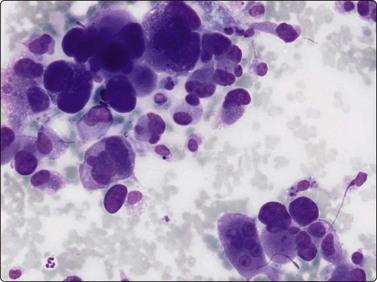
Fig. 15.14 Pleomorphic sarcoma of the MFH type
Moderately cohesive pleomorphic cells; nuclei pleomorphic with malignant chromatin, mainly oval, a few spindled forms; abundant pale syncytial cytoplasm (MGG, HP).
Myxofibrosarcoma (MFS), formerly regarded as a myxoid subtype of malignant fibrous histiocytoma, is at present considered as a specific entity, a malignant fibroblastic tumor with a variable, most often abundant amount of myxoid stroma. MFS is a common subcutaneous sarcoma in elderly patients, although it is also found in skeletal muscle and in the retroperitoneum. MFS generally contains abundant myxoid ground substance in FNB smears (Fig. 15.15). The low-grade malignant tumors are dominated by spindle-shaped cells with slight to moderate nuclear atypia. They may resemble intramuscular cellular myxoma or paucicellular myxoid liposarcoma, but typical lipoblasts are not present and the typical characteristic vascular component of thin-walled branching vessels in the aspirated tumor fragments, as seen in myxoid liposarcoma, are not present. MFS typically displays fragments of coarse, often curved vessels in the myxoid back ground (Fig. 15.16), a feature not seen in FNB smears from intramuscular myxoma (see Fig. 15.54). In high-grade malignant MFS, the cellular pleomorphism and nuclear atypia is as marked as in pleomorphic sarcoma of MFH type. The cellular features of myxofibrosarcoma have been amply described.30-32 Low grade fibromyxoid sarcoma (LGFMS) is a variant of fibrosarcoma, first described in 1987. Two series have been published 1993 and 1995.33,34 This type of sarcoma displays alternating areas of myxoid and collagenous stroma. The tumor cells are spindle-shaped with bland nuclei. As stromal vessels of the same type as in myxofibrosarcoma may be present in the myxoid areas, these low-grade malignant sarcomas may be difficult to distinguish from each other. LGFMS and cellular intramuscular myxoma may be deceptively like. The cytology of LGFMS has been thoroughly described in a report of eight cases (Figs 15.17 and 15.18).35
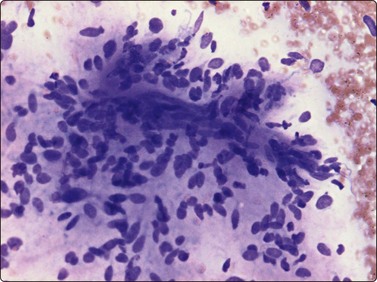
Fig. 15.15 Myxofibrosarcoma, low grade
Nuclear atypia and pleomorphism are slight to moderate note fragment of vessels embedded in myxoid matrix (MGG, IP).
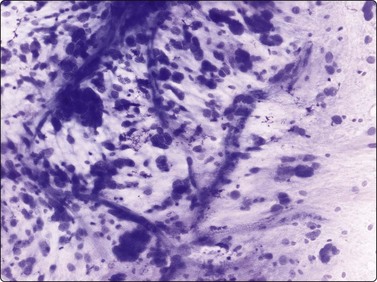
Fig. 15.16 Myxofibrosarcoma, intermediate grade
Poorly cohesive plump spindle cells and a myxoid background; nuclear atypia is more prominent but fragments of vessels are still visible in the background matrix (MGG, IP).
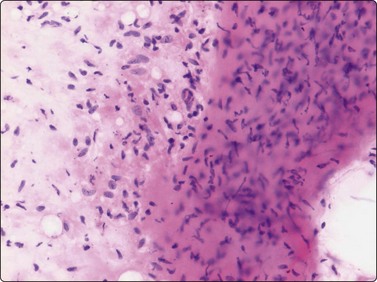
Fig. 15.17 Low-grade fibromyxoid sarcoma
Three-dimensional cohesive clusters of spindle cells embedded in myxoid and collagenous matrix with admixture of scattered bare nuclei and single cells with poorly defined cytoplasm (H&E, LP).
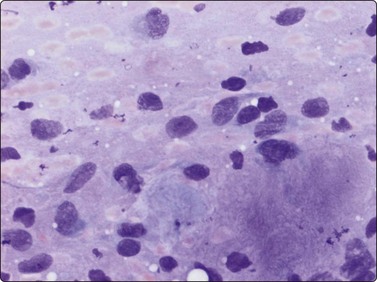
Fig. 15.18 Low-grade fibromyxoid sarcoma
Spindle shaped/fusiform or ovoid nuclei show slightly coarse chromatin and inconspicuous nucleoli; Note myxoid background (MGG, IP).
The anatomical site, size and clinical presentation must be known since atypical fibroxanthoma of the skin (AFX) may be cytologically indistinguishable from pleomorphic sarcoma of MFH type (see Chapter 15). Other differential diagnoses are mainly pleomorphic lipo- and leiomyosarcoma, and anaplastic malignant peripheral nerve sheath tumor (MPNST). Anaplastic sarcomatoid carcinoma (e.g. of renal or bronchogenic origin), or anaplastic squamous cell carcinoma and pleomorphic melanoma may show the same blend of pleomorphic spindled and plump cells. Another differential diagnosis is anaplastic large cell lymphoma. The identification of highly atypical lipoblasts is necessary for the diagnosis of pleomorphic liposarcoma, and of cells of smooth muscle origin (blunt-ended, truncated nuclei) in leiomyosarcoma. Immunocytochemical staining for keratins, S-100 protein, desmin, caldesmon, melanoma associated antigens, CD30 (Ki-1 antibody) and pan B-T cells antigen is of value in the differential diagnosis.
Benign tumors
Lipoma is the commonest of all benign soft tissue tumors. Lipoma cannot be distinguished from normal adipose tissue cytologically – smears of both consist mainly of fragments of mature adipose tissue, a few single fat cells and fat droplets. The fat cells are large and have abundant empty cytoplasm and a small eccentric dark nucleus. A few strands of branching capillary vessels may be seen in the tissue fragments. It is important to make sure that the sample is representative of the suspected tumor. A needle with trocar is recommended for deep-seated tumors to avoid contamination with normal subcutaneous fat. In infiltrating intramuscular lipoma, the fragments of adipose tissue are intimately associated with muscle fibers. Skeletal muscle fibers appear in smears as large strap-like structures which are strongly eosinophilic (navy-blue in MGG) and have rows of small ovoid and elongated, pale nuclei. Cross-striation is often evident. The best indication of an infiltrating intramuscular lipoma is the presence of regenerating muscle fibers, since subcutaneous lipoma may be contaminated with skeletal muscle fibers if needling is too deep and traverses the tumor. The vascularity of an angiolipoma can often be appreciated in FNB smears (Fig. 15.19). Clinically, angiolipomas are often small, multiple and tender on palpation.
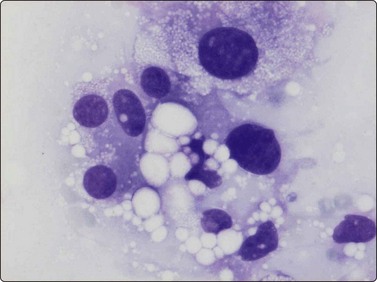
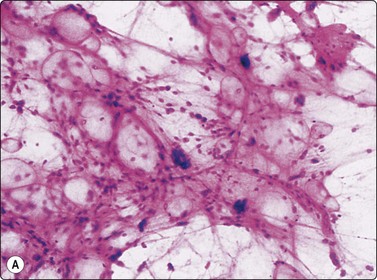
Smears of spindle cell lipoma may have a background of fibromyxoid ground substance and a variable number of spindle cells within tissue fragments or between them. (Fig. 15.20A). The spindle cells may dominate the smears and may be misinterpreted as a fibroblastic proliferation. Important diagnostic features in FNB smears from spindle lipoma are the presence of hyaline, eosinophilic collagen fibers (Fig. 15.20B) and of mast cells. The spindle cells stain strongly for CD34 (Fig. 15.21). The cytological appearance of spindle cell lipoma in smears have been described in a series of 12 cases.36 Pleomorphic lipoma, considered to be related to spindle cell lipoma (similar typical site and chromosomal aberration) typically displays so-called floret cells in smears (Figs 15.22). In a typical clinical setting (a subcutaneous tumor in the neck or back in a middle-aged male), a confident diagnosis of both these variants of lipoma is possible. Hibernoma is recognisable in FNB smears provided that typical hibernoma cells are found. These have abundant, finely granular or vacuolated cytoplasm and centrally located small, rounded uniform nuclei (Fig. 15.23).37 The typical findings in FNB smears of lipoblastoma (subcutaneous) and lipoblastomatosis (deep-seated) are a vascular network and clusters of lipoblast-like cells in a myxoid background (Fig. 15.24). Another variant of benign lipoma, chondroid lipoma, has been described. In this variant foci of mature adipocytes are seen together with chondrocyte- or lipoblast-like cells in a chondromyxoid-like matrix.38 The cytomorphology of chondroid lipoma has so far been described only in single case report.39,40 The main features are lipoblast-like cells with irregular nuclei, often lobulated or coffee-bean shaped, mixed with mature fat cells within fragments of a chondroid-like matrix (Fig. 15.25). Chondroid lipoma has been erroneously diagnosed as low-grade malignant liposarcoma (paucicellular myxoid liposarcoma) in FNB samples. Extra-adrenal myelolipoma is a tumor-like mass composed of mature fat and bone marrow cells. The typical site is the adrenals but the lesion can also occur in the retroperitoneum and in the pelvic region. Extra-adrenal myelolipoma should be considered in the differential diagnosis of tumors or tumor-like lesions in those sites. FNB smears typically contain fragments or clusters of normal fat cells mixed with bone marrow cells of all three lineages (Fig.15.26).
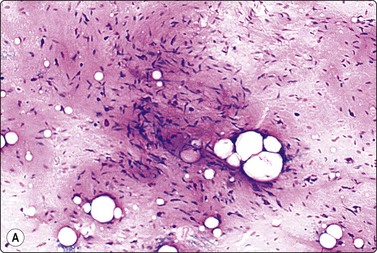
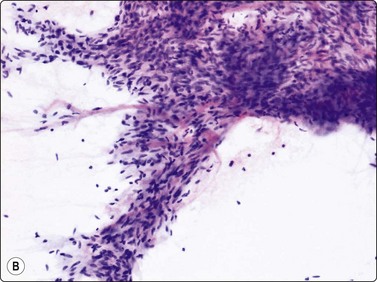
Fig. 15.20 Spindle cell lipoma
(A) A myxoid matrix may be prominent in smears and may give an impression of a truly myxoid tumor (H&E, LP); (B) The presence of hyaline, eosinophilic collagen fibers is an important diagnostic clue (ThinPrep, H&E, LP).
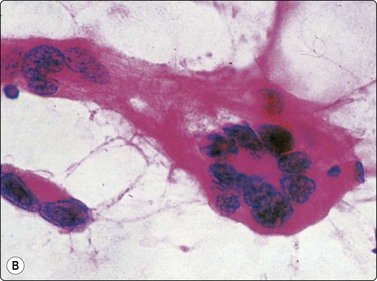
(A) Mature adipose tissue; scattered large, dark multinucleated cells (H&E, IP); (B) Same case as A; large multinucleated cell (floret cell) (H&E, HP oil).
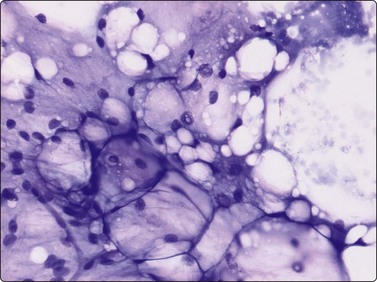
Tissue fragments of fat cells, uni- and multivacuolated lipoblast-like cells and a myxoid matrix (MGG, IP).
Malignant tumors
Well-differentiated liposarcoma and atypical lipoma are histologically closely related and cannot be distinguished in FNB smears. The anatomical site and clinical presentation decide the terminology. In 2001, Kempson introduced a new term, atypical lipomatous tumor, to include atypical lipoma and well-differentiated liposarcoma.41 Tissue fragments in smears from these tumors are composed of mature fat cells mixed with atypical lipoblast-like cells and atypical spindled or rounded cells with hyperchromatic nuclei (Fig. 15.27). The cytological findings must be evaluated in relation to the anatomical site. Especially in the retroperitoneum, recurrent well-differentiated liposarcoma may dedifferentiate, i.e. areas of high-grade malignant sarcoma may develop within the well-differentiated liposarcoma. The diagnosis of dedifferentiated liposarcoma in FNB smears rests on the presence of a high-grade sarcoma and well-differentiated liposarcoma in the same needling.
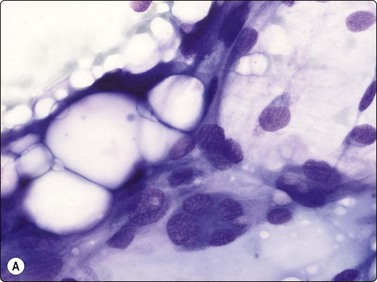
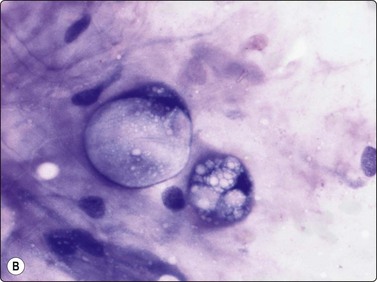
Fig. 15.27 Well-differentiated liposarcoma (atypical lipomatous tumour)
(A) Fragments of mature adipose tissue and scattered large atypical cells with hyperchromatic nuclei (MGG, HP); (B) The atypical lipoblasts embedded in fibrous stroma (MGG, HP).
Myxoid liposarcoma, the commonest of the liposarcomas is currently divided in two variants: paucicellular and hypercellular myxoid liposarcoma. Paucicellular myxoid liposarcoma is identical with the classic myxoid liposarcoma, while the hypercellular variant corresponds to the former round cell liposarcoma. The reason to rename round cell liposarcoma is that this subtype was found to express the same chromosomal aberration, t(12;16)(q13;p11), as classic myxoid liposarcoma.10 In paucicellular myxoid liposarcoma, the peculiar network of anastomosing capillary vessels is well recognisable in FNB smears and is one of three characteristic features. The other two are abundant myxoid background matrix and slightly atypical lipoblasts. Smears consist mainly of small tissue fragments with branching capillary vessels embedded in the myxoid matrix (Fig. 15.28A). The tumor cells, mainly seen in the fragments, have spindle or ovoid, relatively uniform nuclei and a thin cytoplasm. Lipoblasts are always found – uni- or multivacuolated with scalloped nuclei (Fig. 15.28B). The lipoblasts are best visualised in MGG; the cytoplasmic vacuoles are less prominent in H&E. Differentiation from low-grade malignant myxofibrosarcoma (MFS) depends on the demonstration of the capillary network within the tumor fragments and of lipoblasts. The vessel fragments seen in smears of MFS are usually thicker and are present in the myxoid ground substance. Smears of hypercellular myxoid liposarcoma (former round cell liposarcoma) show either numerous dissociated cells or tissue fragments containing closely packed tumor cells. The fragments are considerably more cellular than those of the paucicellular type; myxoid ground substance is less conspicuous and the capillary network less prominent. The tumor cells have irregular, rounded nuclei and a malignant chromatin pattern. The cytoplasm is fragile and many nuclei are stripped. Mitotic figures are occasionally seen and atypical lipoblasts always found.
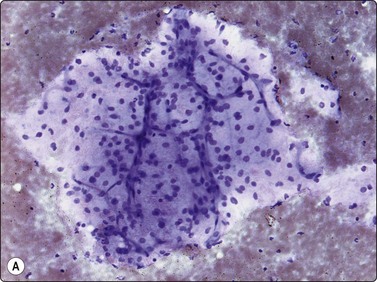
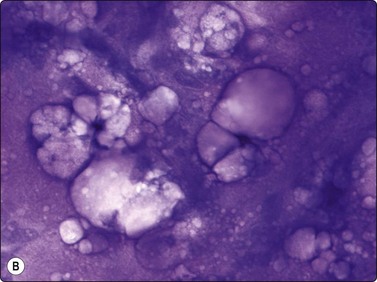
(A) Tissue fragment of cells embedded in myxoid background material; typical anastomosing vessels (MGG, IP); (B) Scattered multivacuolated lipoblasts (MGG, HP).
Pleomorphic liposarcoma resembles the pleomorphic sarcoma of MFH type cytologically. The diagnosis rests on the presence of highly atypical, sometimes multinucleated lipoblasts. Lipoblasts in liposarcoma smears may be univacuolated and resemble signet ring cells or they may be multivacuolated, containing small or large vacuoles or a mixture of both. The scalloped nucleus is an important cytologic feature (Fig. 15.29). In FNB smears, superimposed fat vacuoles from adjacent fat tissue may produce artifacts in other cell types, causing a resemblance to lipoblasts, and macrophages in adipose tissue showing inflammatory or reactive changes may have a vacuolated cytoplasm (lipophages). An infrequent diagnostic pitfall is soft tissue metastasis of renal cell carcinoma. It is important to remember that the majority of liposarcomas are deep seated and only infrequently subcutaneous. The cytologic feature of liposarcoma in FNB smears have been investigated in several series.32,42-45
Criteria for diagnosis
Benign tumors
Angioleiomyoma is most often a small, dermal or subcutaneous tumor, very tender at palpation. Angioleiomyoma may be the subject for FNB. According to our files, angioleiomyoma is very difficult to diagnose as such in smears. In a study comprising 10 cases, no case was correctly typed, albeit most were diagnosed as benign tumors.46 The FNB smears contained uniform or slightly pleomorphic spindle cells with bland chromatin and insignificant nucleoli. The nuclei were occasionally blunt-ended or cigar-shaped and the cells were predominantly dissociated, mixed with small groups of cells (Fig. 15.30).
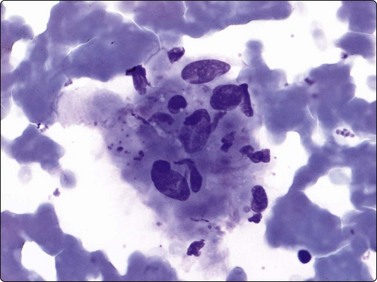
Small groups of uniform or slightly pleomorphic spindle cells with poorly defined cytoplasm borders and nuclei with bland chromatin (MGG, HP).
The histological diagnosis of malignancy in well-differentiated tumors of smooth muscle origin is to a large extent dependent on mitotic counts, invasive growth and size, which generally cannot be evaluated in FNB smears. The question of malignancy and the distinction from leiomyoma of deep soft tissue may therefore be difficult to decide in smears from low-grade malignant leiomyosarcoma. Our experience of leiomyoma of deep soft tissue is restricted to a few cases. The cytologic findings were remarkably similar. The cells were arranged in clusters and also seen as single cells. Stripped nuclei were common among single cells. Typical tumor cell nuclei were cigar-shaped (Fig. 15.31), sometimes truncated, sometimes containing vacuoles. The chromatin was finely granular and nucleoli were inconspicuous. Many nuclei were fibroblast-like.
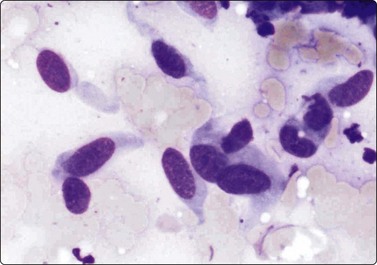
Fig. 15.31 Deep-seated leiomyoma
Typical benign smooth muscle cells with blunt-ended, bland-looking nuclei (MGG, HP).
Malignant tumors
Smears from moderately differentiated leiomyosarcoma (grade II in a three-grade scale or grade III in a four-grade scale) are cellular and are composed of clusters and fascicles of cohesive cells. The clusters have a syncytial appearance, lacking visible cell borders, and show anisokaryosis (Fig. 15.32A). Nuclei of single cells or stripped nuclei can be found displaying an unequivocally malignant chromatin pattern (Fig. 15.32B). Moderately differentiated leiomyosarcoma can be difficult to distinguish from benign as well as malignant neurogenic tumors. Ancient neurilemmoma is a particularly important diagnostic pitfall.47 Neurogenic tumors, however, tend to have more slender, elongated nuclei with pointed ends.
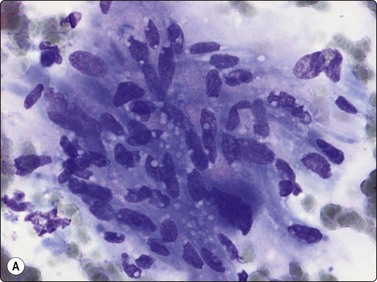
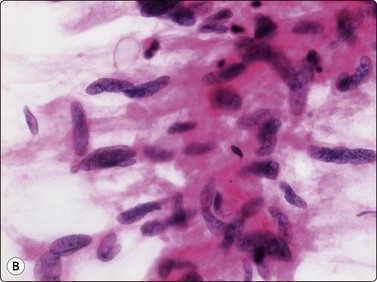
Fig. 15.32 Moderately differentiated leiomyosarcoma
(A) Tissue fragment of cohesive cells with indistinct cytoplasm and spindly, occasionally cigar-shaped nuclei (MGG, IP); (B) Moderately differentiated leiomyosarcoma. Single cells or stripped nuclei can be found displaying an unequivocally malignant chromatin pattern (H&E, IP).
Some high-grade malignant leiomyosarcomas display a round cell pattern, and these tumors can be mistaken for epithelial tumors (Fig. 15.33). High-grade malignant pleomorphic leiomyosarcoma is difficult to distinguish from pleomorphic sarcomas of MFH type (Fig. 15.34A) or from high-grade malignant peripheral nerve sheath tumors. The presence of blunt-ended, truncated or vacuolated nuclei in highly atypical spindle cells is an important diagnostic sign (Fig. 15.34B). More or less numerous multinucleated osteoclast-like giant cells can be found in smears from pleomorphic leiomyosarcoma (Fig. 15.35). The cytologic features of leiomyosarcoma is recorded in two large series.48,49 Important antibodies to prove smooth muscle origin are desmin and caldesmon (Fig. 15.36).
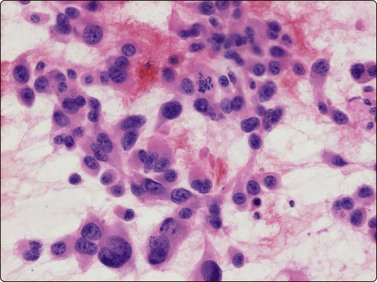
Fig. 15.33 High-grade malignant leiomyosarcoma
A round cell pattern can be found and these tumors can be mistaken for epithelial tumors (H&E, IP).
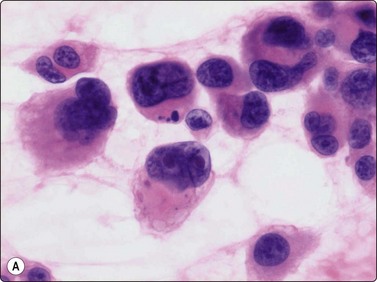
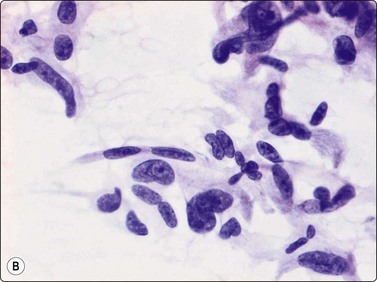
Fig. 15.34 High-grade malignant leiomyosarcoma
(A) A pleomorphic cell pattern is difficult to distinguish from pleomorphic sarcomas of MFH type (H&E, HP); (B) The presence of blunt-ended, truncated or vacuolated nuclei in highly atypical spindle cells is an important diagnostic sign (H&E, IP).
Benign tumors
Benign (adult) rhabdomyoma is rare. In the few examples from our files, the smears contained many fragments of muscle fibers which were paler, smaller and more irregular in shape than normal skeletal muscle fibers (Fig. 15.37).Nuclei were larger and had prominent nucleoli. Cross-striation was difficult to distinguish. The fragments closely resembled regenerating myocytes. Published case reports describe similar findings.50,51
Malignant tumors
The three different types of rhabdomyosarcoma can be recognised in FNB smears.52 Embryonal rhabdomyosarcoma is the commonest soft tissue sarcoma in children (see p. 432). In our experience, although rhabdomyosarcoma generally is considered as one of the small round cell malignant tumors of childhood, embryonal rhabdomyosarcoma is predominantly a pleomorphic tumor in FNB smears, composed of both small and larger cells (Fig. 15.38). Cells with dense, eosinophilic cytoplasm and an eccentric nucleus, triangular, strap-shaped or tadpole-like, may be recognised as rhabdomyoblasts and may suggest the correct diagnosis (Fig. 15.39). However, in many cases most cells are spindle-like with elongated nuclei and fusiform cytoplasm. We have examined smears from one case of the spindle cell variant of embryonal rhabdomyosarcoma.53 The vast majority of cells were spindle-shaped. Nuclei were fusiform, moderately pleomorphic, alternately blunt-ended or with pointed ends. Typical rhabdomyoblasts were few and difficult to find (Fig. 15.40).
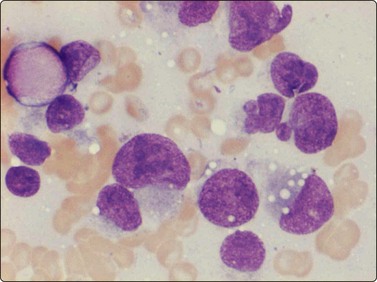
Fig. 15.38 Embryonal rhabdomyosarcoma
Predominantly a pleomorphic tumor in FNB smears, composed of both small and larger cells (MGG, HP).
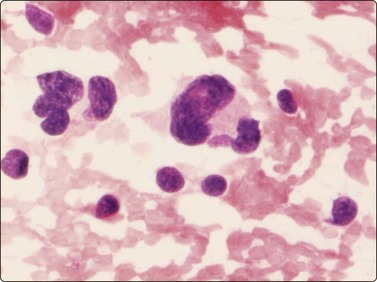
Fig. 15.39 Embryonal rhabdomyosarcoma
Cells with eosinophilic cytoplasm and an eccentric nucleus, triangular, strap-shaped or tadpole-like, may be recognised as rhabdomyoblasts and may suggest the correct diagnosis (H&E, HP).
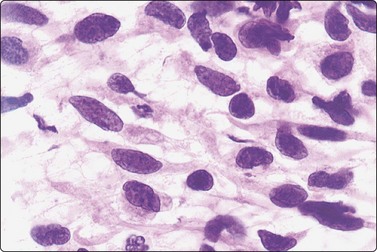
Fig. 15.40 Spindle cell embryonal rhabdomyosarcoma
The majority of cells are spindle-shaped with fusiform, moderately pleomorphic nuclei (H&E, IP).
Alveolar rhabdomyosarcoma is a typical small round cell malignancy in FNB smears (Fig. 15.41). Smears show predominantly single cells, and stripped nuclei are common. Alveolar or rosette-like structures may be seen. The cells are mainly uniform with rounded or irregular nuclei and scanty cytoplasm. In almost every case, small rounded or pear-shaped cells with eccentric nuclei and eosinophilic cytoplasm are found (Fig. 15.42). The chromatin is variably coarse and nucleoli may be prominent. The multinucleated cells with small, dark nuclei, commonly seen in histologic sections are, in our experience, not a common finding in smears (Fig. 15.43).
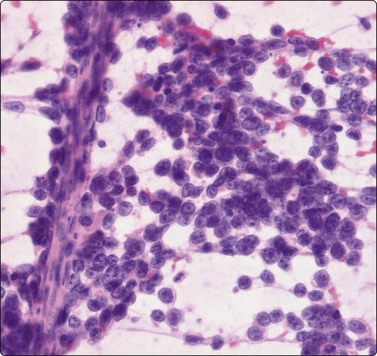
Fig. 15.41 Alveolar rhabdomyosarcoma
The cells are mainly uniform with rounded or irregular nuclei and scanty cytoplasm (H&E, IP).
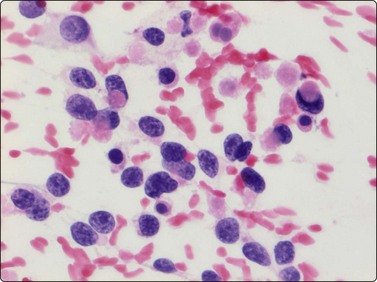
Fig. 15.42 Alveolar rhabdomyosarcoma
In almost every case, small rounded or pear-shaped cells with eccentric nuclei and eosinophilic cytoplasm (small rhabdomyoblasts) are found (H&E, HP).
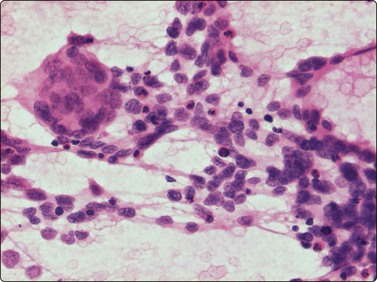
Fig. 15.43 Alveolar rhabdomyosarcoma
The multinucleated cells with small, dark nuclei, commonly seen in histologic sections, are not a common finding in FNA smears (H&E, IP).
In recent classifications, rhabdomyosarcomas are divided into favorable and unfavorable subtypes. All variants of embryonal rhabdomyosarcoma are considered as favorable tumors, while alveolar rhabdomyosarcoma and embryonal rhabdomyosarcoma with areas of alveolar rhabdomyosarcoma are unfavorable.54 It is thus clinically important to arrive at a type-specific diagnosis based on the FNB sample. In our experience and in that of others, this is possible in many cases.55,56 Immunocytochemical demonstration of desmin and/or the more specific markers for striated muscle, myogenin and MyoD-1 (Fig. 15.44), are valuable adjuncts to confirm the diagnosis as the demonstration of the chromosomal aberration t(2;13)(q35;q14), basis for a fusion transcript between the PAX3 and GKHR genes in alveolar rhabdomyosarcoma.57 Positivity for myoglobin is only present in more differentiated myoblasts.
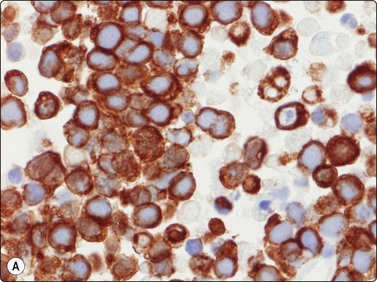
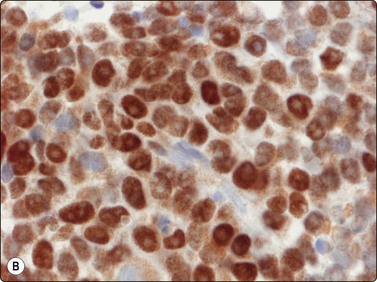
Fig. 15.44 Alveolar rhabdomyosarcoma
Important antibodies to prove muscle and striated muscle origin are desmin and MyoD-1 (Cell block, desmin and MyoD-1, immunoperoxidase, HP).
Pleomorphic rhabdomyosarcoma, for many years thought to be a non-existent entity, is in current classifications considered as a rhabdomyosarcoma subtype in adults, especially the elderly.58 The few cases in our files showed similar features; a pleomorphic sarcoma resembling pleomorphic sarcoma of MFH type but displaying scattered highly atypical rhabdomyoblast-like cells with abundant dense eosinophilic cytoplasm and eccentric nuclei (Fig. 15.45).
Benign tumors
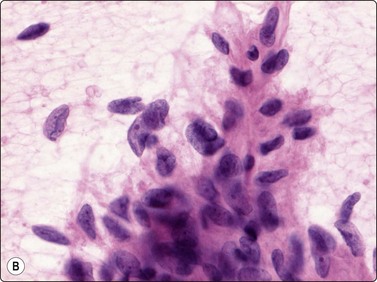
The needling of a neurilemmoma or of a neurofibroma may trigger a sharp pain along the nerve and this is a valuable diagnostic sign. However, needling of a tumor adjacent to nerve may cause similar pain if the needle traverses the tumor and touches the nerve. Neurilemmoma is a common target for FNB; the material obtained varies from case to case. If Antoni A areas are sampled, tissue fragments of cohesive cells are characteristic and more commonly seen than single cells (Fig. 15.46A). Samples from Antoni B areas often show mainly dispersed cells and a myxoid or at times cystic background, and only few and small tissue fragments (Fig. 15.46B). Whichever portion is sampled, the cellularity of the fragments is variable. The most typical feature is the fibrillar appearance of the intercellular stroma in fragments (Fig. 15.46C). Nuclear palisading may occur and is more commonly seen than true Verocay bodies (Fig. 15.47A). Nuclei tend to be long and slender with pointed ends. Often, a number of the slender nuclei are comma-shaped or are bent like a fishhook (Fig. 15.47B). There is often a moderate degree of nuclear pleomorphism, but the chromatin pattern is uniformly bland. If cells with plump, spindled nuclei and more abundant cytoplasm predominate, a diagnosis of low-grade malignant leiomyosarcoma may be considered. The pleomorphic and bizarre cells in ancient neurilemmoma with their degenerate, dark nuclei, at times with vacuoles (so called ‘kern-loche’) (Fig. 15.48A), have been mistaken for malignancy47 but the overall smear pattern is that of a benign tumor (Fig. 15.48B).
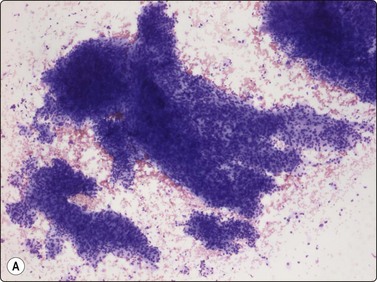
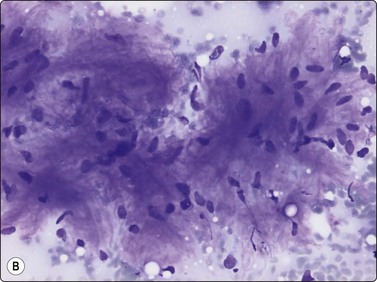
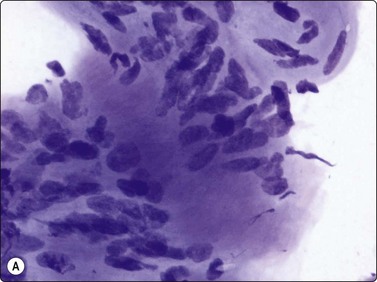
(A) Tissue fragments of cohesive cells are commonly seen in samples from Antoni A areas (MGG, LP); (B) Mainly dispersed cells and only few small tissue fragments in a myxoid or at times cystic background are commonly seen in samples from Antoni B areas (MGG, IP); (C) The most typical feature is the fibrillar appearance of the intercellular stroma in fragments (H&E, HP).
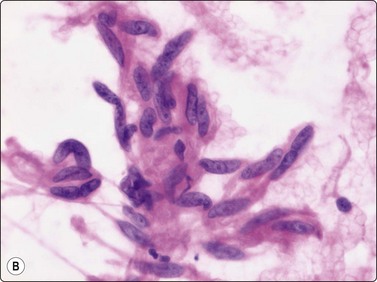
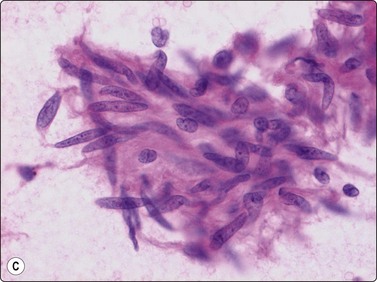
(A) Nuclear palisading may occur and is more commonly seen that true Verocay bodies (MGG, HP); (B) Nuclei tend to be long and slender with pointed ends, Some of nuclei are comma-shaped or are bent like a fishhook (H&E, HP).
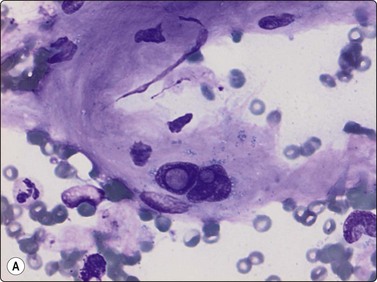
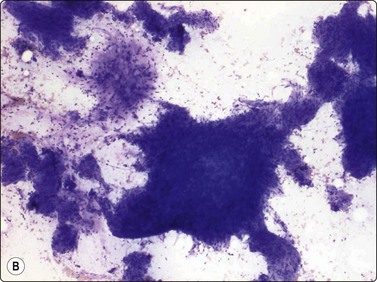
(A) The pleomorphic and bizarre cells with dark nuclei, at times with vacuoles (so called ‘kern-loche’) (MGG, HP); (B) The overall smear pattern is that of a benign tumor (MGG, HP).
In FNB smears from neurofibroma, dispersed cells are more common than tissue fragments and a myxoid background matrix often seen. In 2006, Domanski et al. reported the cytological features of neurilemmoma in a series of 116 cases.59
The granular cell tumor (Abrikosoff tumor) is currently regarded as a neurogenic tumor and not of myogenic origin, as formerly considered. Granular cell tumors are mainly found in the subcutaneous and submucosal tissues, most commonly in the head and neck region. In the breast, it can closely simulate cancer clinically and radiologically (Chapter breast Ch 7 in ed 4). It may also occur as a deep-seated tumor in the limbs. FNB smears show many neoplastic cells in syncytial clusters and singly. The nuclei may be arranged in a vaguely follicular pattern. The cytoplasm of intact cells is abundant and relatively dense, eosinophilic in H&E and dark blue in MGG with prominent characteristic granulation. The cytoplasm is fragile and may be dispersed in the background together with naked nuclei. The nuclei are predominantly small round or ovoid, with bland chromatin and small but prominent nucleoli (Fig. 15.49A). Anisokaryosis can be marked in some tumors and there may be irregularity of nuclear shape.
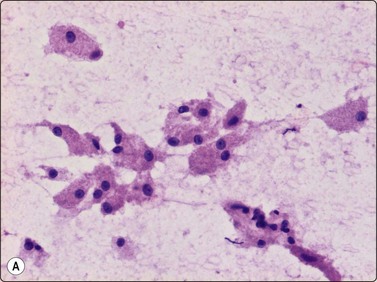
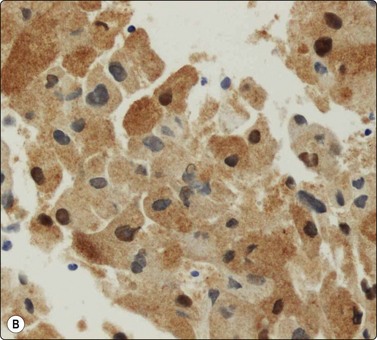
Fig. 15.49 Granular cell tumor
(A) The cytoplasm is abundant and relatively dense with prominent characteristic granulation (H&E, HP); (B) S-100 protein is generally positive in granular cell tumors (cell block, S-100, immunoperoxidase, HP).
S-100 protein is generally positive in all these benign neurogenic tumors (Fig. 15.49B). Many granular cell tumors are, furthermore, positive for NSE and inhibin.
Histologically, focal necrosis, increased mitotic activity, nuclear hyperchromatism and atypical spindle cells are the typical features of the rare malignant granular cell tumors.
The cytological appearance of benign as well as malignant granular cell tumors has been recorded.60-62
Malignant tumors
Most malignant peripheral nerve sheath tumors (MPNST) show the pattern of spindle cell sarcomas; some are highly pleomorphic and some have epithelioid features (Fig. 15.50A). Cells from a low-grade tumor may be only moderately pleomorphic. In high-grade tumors, the smears are highly cellular and the cells have elongated, spindly, atypical nuclei (Fig. 15.50B). Mitotic figures are not uncommon. Pleomorphic MPNST often displays a cytomorphology resembling that of a pleomorphic sarcoma of MFH type, including multinucleated tumor cells with bizarre nuclei. Nuclear chromatin is coarse and irregular. Cells are single and in clusters with intercellular collagen, and a background of myxoid material is occasionally present. Neurofibrosarcoma in von Recklinghausen’s disease may be difficult to diagnose as these tumors may show a mixture of benign and malignant areas (Fig. 15.51). The variable cytologic features of MPNST in FNB smears have been recorded in a number of series.63,64 Positivity for S-100 protein, an important feature in benign neurogenic tumors, is of less diagnostic value in MPNST, as many tumors are negative and positivity often focal.
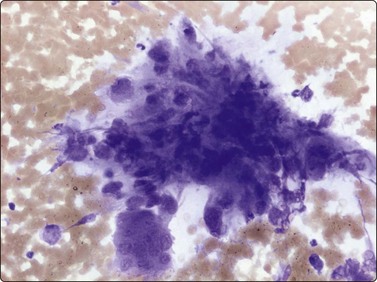
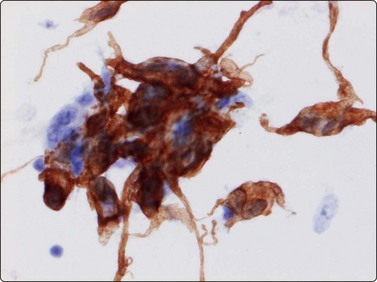
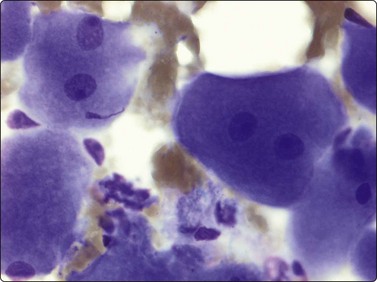
Fine needle aspiration of hemangioma of any type yields plenty of venous blood. Smears may contain a few strands of endothelial cells with pale, spindly nuclei (Fig. 15.52). Macrophages, hemosiderin pigment and fibroblasts may be present if thrombosis has occurred. Most angiosarcomas are cutaneous tumors; only a minority are deep-seated. The cytologic features of angiosarcoma are variable. The cell population may be predominantly spindled, pleomorphic or epithelioid (Fig. 15.53A, B). A correct diagnosis is often not possible without immunocytochemical confirmation (CD31, CD34 and Fli-1) that the tumor cells are of endothelial origin (Fig. 15.53C). In epithelioid variants of angiosarcoma, EMA and keratin antibodies are often positive. The variable cytologic features in angiosarcoma have been described in several recent papers.65-67 Angiosarcoma of the breast is illustrated in Chapter 7 and of the spleen in Chapter 11.
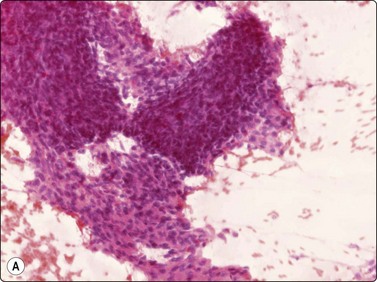
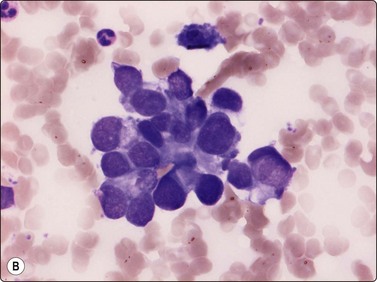
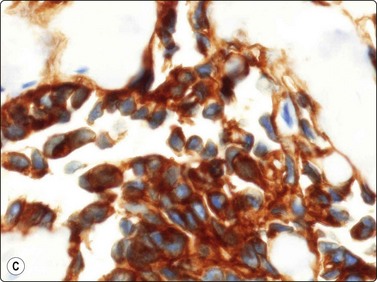
(A) In this example, the smear population is predominantly of atypical spindle cells; differentiation from other spindle cell sarcomas requires immunostaining (H&E, LP); (B) Epithelioid pattern of gland-like cluster of malignant cells with rounded nuclei (MGG, HP); (C) A correct diagnosis is often not possible without immunocytochemical confirmation that the tumor cells are of endothelial origin (cell block, CD34, immunoperoxidase, HP).
Tumors of uncertain or debated histogenesis
Benign tumors
Intramuscular myxoma has a quite characteristic appearance in FNB smears.68 Macroscopically, the aspirate consists of droplets of colorless, mucoid, stringy and semi-liquid material resembling the aspirate from a ganglion. Smears show small tissue fragments and single cells in an abundance of myxoid background material (Fig. 15.54A). The cells are predominantly spindle shaped and have elongated nuclei and most often very long, thin cytoplasmic processes (Fig. 15.54B). The chromatin is regularly dispersed and finely granular, and nucleoli, if present, are small. Some histiocyte-like cells with abundant vacuolated cytoplasm, sometimes containing a large cytoplasmic globule of bluish-violet material (MGG), are also present. Fragments of capillary vessels are rare and this is important in distinguishing intramuscular myxoma from paucicellular myxoid liposarcoma, low-grade malignant myxofibrosarcoma and low-grade fibromyxoid sarcoma. Nielsen et al. described cases of intramuscular myxoma with focally increased cellularity and hypervascularity designated as cellular myxoma.69 FNB smears from such areas of cellular myxoma may be very difficult to distinguish from low-grade fibromyxoid sarcoma and low-grade myxofibrosarcoma due to the cellularity and the increased number of vessel fragments.
Malignant tumors
The cytomorphology of epithelioid sarcoma has been characterised.70,71 The three cases in our files displayed spindled or polygonal tumor cells in loose clusters or singly, with abundant cytoplasm, rounded or ovoid nuclei and large nucleoli (Fig. 15.55). In one case, histiocytes and inflammatory cells dominated the smears. The cytology of clear cell sarcoma and of epithelioid sarcoma is rather similar and the different clinical presentation and immunocytochemical profile are important in the differential diagnosis.
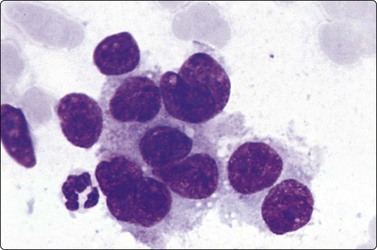
Fig. 15.55 Epithelioid sarcoma
A loose cluster of large cells with abundant cytoplasm, irregular nuclei, unevenly distributed chromatin and prominent nucleoli (MGG, HP oil).
FNB smears from clear cell sarcoma (malignant melanoma of soft tissues) have been studied in rather few cases.72,73 We have seen two examples of this relatively rare tumor. The cells are mostly dispersed but small groups of loosely cohesive cells are also present. The cells are polygonal or spindle shaped and have abundant pale cytoplasm and rounded nuclei with prominent nucleoli (Fig. 15.56). Immunocytochemistry (S-100 protein and melanoma-associated antigens) are helpful in diagnosis but soft tissue metastasis of malignant melanoma is a problem. Cytogenetic investigation is important in the differential diagnosis versus malignant melanoma, as clear cell sarcoma has been found to harbor the chromosomal aberration t(12;22)(q13-14;q 12-13), giving rise to the transcript ATF1/EWS.
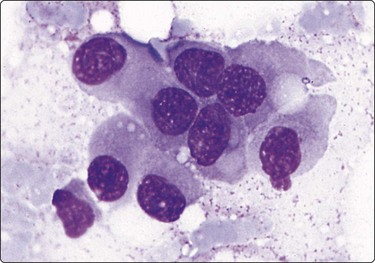
Fig. 15.56 Malignant melanoma of soft tissues (clear cell sarcoma)
A cluster of loosely cohesive cells. The cells are large, ovoid or spindle with abundant cytoplasm, rounded nuclei and prominent nucleoli (MGG, HP oil).
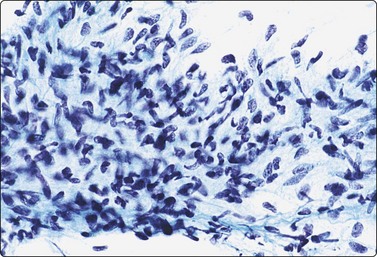
Smears from synovial sarcoma are usually highly cellular (Fig. 15.57A). The typical appearance is a mixture of tissue fragments and dispersed cells. Mitotic figures can be found in almost every case, especially in the tissue fragments. Bare nuclei are common. A hemangiopericytoma-like vascular pattern with branching capillaries (’staghorn’ pattern) is commonly seen in fragments. The cells are small to medium in size. Nuclei are rounded or ovoid with finely granular, bland chromatin and nucleoli are small and inconspicuous. Intact cells have thin uni- or bipolar cytoplasm. Vaguely acinar-like structures may be seen in the periphery of fragments (Fig. 15.57B). Mast cells are prominent in some cases. A myxoid background matrix has been reported in some cases.74 An obvious biphasic pattern is only infrequently seen in our experience. The cytology of mono- and biphasic synovial sarcoma has been reported in two large series.75,76 The cytomorphology of the poorly differentiated variant77 (involving the entire tumor or focally in other subtypes) is relatively unknown and has been described in few cases (Fig. 15.58).78-80 Poorly differentiated fibrosarcoma-like synovial sarcoma is extremely difficult to distinguish from MPNST and the rare fibrosarcoma. A reliable cytologic diagnosis of synovial sarcoma is important since neoadjuvant therapy is commonly applied before surgery. Even if synovial sarcoma is strongly suspected in an FNB smear, adjunctive diagnostic methods are almost always necessary to reach a confident diagnosis. The majority of synovial sarcomas stain positively for EMA and keratins 7 and 19 (although often focally); positive staining for CD99 and bcl-2 has also been reported. Cytogenetic analysis is a most important valuable adjunct. FISH (Fig. 15.59) and RT-PCR of aspirated material used for detection of the two most common gene fusion products (SYT/SSX1 and SYT/SSX2, respectively) is more effective than chromosomal analysis of cytologic material to diagnose the translocation t(X;18)(p11;q12).81
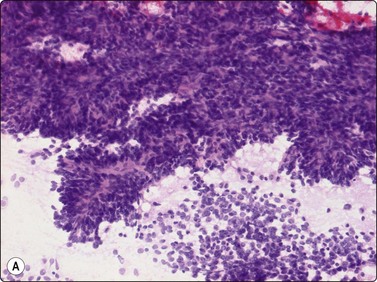
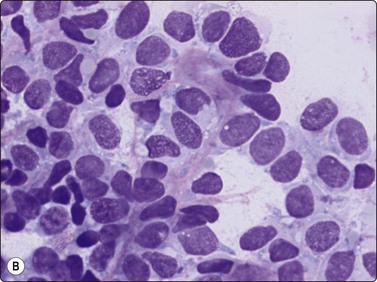
(A) Smears are usually highly cellular; the typical appearance is a mixture of tissue fragments and dispersed cells (H&E, LP); (B) Vaguely acinar-like structures may be seen in the periphery of fragments (MGG, HP).
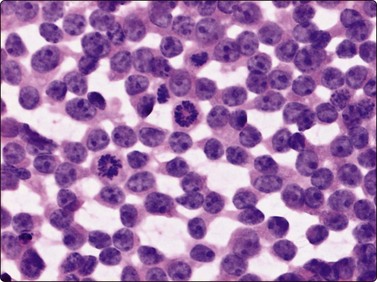
Smears from a poorly differentiated synovial sarcoma can be difficult to differ from Ewing’s sarcoma (H&E, HP).
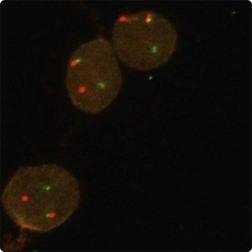
Fig. 15.59 FISH-Synovial sarcoma
Separate red and green signals indicative of a rearrangement of one copy of the SYT gene region.
Only single cases of extraskeletal chondrosarcoma and extraskeletal osteosarcoma have been studied.82 Extraskeletal myxoid chondrosarcoma (EMC), however, has been thoroughly investigated in a number of cases.83,84 Cells, forming strands or dispersed, are typically embedded in an abundant myxoid ground substance. The most usual patterns are cells arranged in clusters or branching strands (Fig. 15.60). However, the tumor cells may be arranged in cell balls embedded in the ground substance, mimicking the appearance of mucus-producing carcinoma (Fig. 15.61A). The cells are small to medium in size, there is a moderate amount of cytoplasm, and nuclei are rounded or ovoid with small but distinct nucleoli. Uni- or bipolar cytoplasmic processes are seen in some cells (Fig. 15.61B). Intranuclear inclusions were prominent in one of our cases. Fragments of chondroid substance and rounded chondroblast-like cells have been described in some cases. Recently, cases of EMC showing signs of neuroendocrine differentiation have been published. One case from our files was difficult to diagnose correctly since there was ultrastructural evidence of neuroendocrine differentiation at the electron microscopic examination of the aspirated cells.85 The FNB findings in the rare alveolar soft part sarcoma have been described in a few case reports.86,87
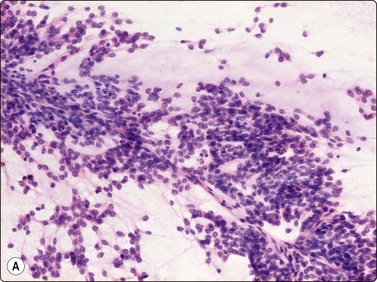
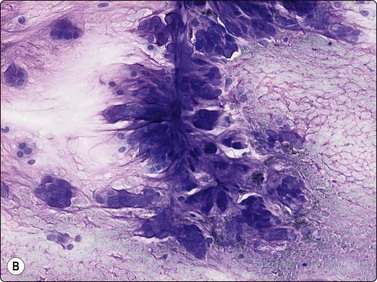
Fig. 15.60 Extraskeletal myxoid chondrosarcoma
(A, B) Cells with relatively uniform rounded nuclei embedded in a prominent myxoid matrix; a tendency for the cells to appear in small rows (A, H&E, LP; B, MGG, IP).
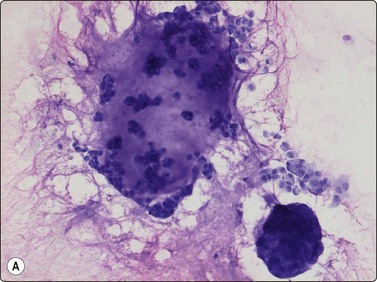
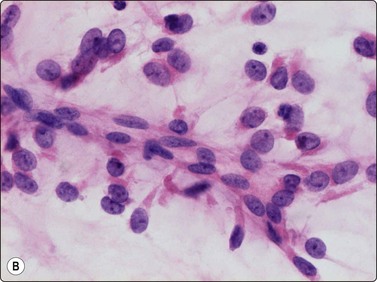
Fig. 15.61 Extraskeletal myxoid chondrosarcoma
(A) The tumor cells may be arranged in cell balls embedded in the ground substance, mimicking the appearance of mucus-producing carcinoma (MGG, IP); (B) The cells typically have rounded nuclei with small nucleoli and a variable amount of cytoplasm. Uni-or bipolar cytoplasmic processes are seen in some cells (H&E, HP).
1 Rydholm A. Centralisation of soft tissue sarcoma: The Southern Sweden Experience. Acta Orthop Scand. 1997;68(suppl. 273):4-8.
2 Åkerman M, Rydholm A. Surgery based on fine needle aspiration cytology. Acta Orthop Scand. 1994;65(suppl. 256):69-70.
3 Brosjö O, Bauer HCP, Kreisbergs A, et al. Fine needle aspiration biopsy of soft tissue tumours. Acta Orthop Scand. 1994;65(suppl. 256):108-109.
4 Dey D, Mallik MK, Gupta SK, et al. Role of fine needle aspiration cytology in the diagnosis of soft tissue tumours and tumour-like lesions. Cytopathology. 2004;15:32-37.
5 Kumar S, Chowdhury N. Accuracy, limitations and pitfall in the diagnosis o0f soft tissue tumours by fine needle aspiration cytology. Indian J Pathol Microbiol. 2007;1:42-45.
6 Domanski HA, Åkerman M, Carlén B, et al. Core-needle biopsy performed by the cytopathologist. A technique to complement fine-needle aspiration of soft tissue and bone lesions. Cancer (Cancer Cytopathol). 2005;105:229-239.
7 Jones C, Liu K, Hirschowits S, et al. Concordance of histopathologic and cytologic grading in musculoskeletal sarcoma. Can grade obtained from analysis of the fine-needle aspirates serve as a basis for therapeutic decisions? Cancer (Cancer Cytopathol). 2002;96:83-91.
8 Åkerman M, Domanski H. The cytology of soft tissue tumours. Monographs in Clinical Cytology. vol 16. Karger, Basel, 2003.
9 Gonzáles-Cámpora R. Cytoarchitectural findings in the diagnosis of primary soft tissue tumors. Acta Cytol. 2001;45:115-146.
10 Fletcher DM, Unni KK, Mertens F. World Health Organization Classification of Tumours. Pathology and genetics. Tumours of Soft Tissue and Bone. Lyon: IARC Press; 2001.
11 Weiss SW, Goldblum JR. Soft Tissue Tumors, 5th ed. St Louis: Mosby; 2008.
12 Dahl I, Åkerman M. Nodular fasciitis. A correlative cytologic and histologic study of 13 cases. Acta Cytol. 1981;25:215-222.
13 Dodd LG, Martinez S. Fine-needle aspiration cytology of pseudosarcomatous lesions of soft tissue. Diagn Cytopathol. 2001;24:28-35.
14 Kong CS, Cha I. Nodular fasciitis: diagnosis by fine needle aspiration biopsy. Acta Cytol. 2004;48:473-477.
15 Stanley M, Skoog L, Tani E, et al. Spontaneous resolution of nodular fasciitis following diagnosis by fine needle aspiration. Acta Cytol. 1991;35:616-617.
16 Willén H, Åkerman M, Rydholm A. Fine needle aspiration of nodular fasciitis. No need for surgical intervention. Proceedings of the Annual Meeting of the European Musculoskeletal Oncology Society, Amsterdam, 1994.
17 Rööser B, Herrlin K, Rydholm A, et al. Pseudomalignant myositis ossificans. Clinical, radiologic, and cytologic diagnosis. Acta Orthop Scand. 1989;60:457-460.
18 Dalén BP, Meis-Kindblom JM, Sumathi VP, et al. Fine-needle aspiration cytology and core needle biopsy in the preoperative diagnosis of desmoids tumors. Acta Orthop Scand. 2006;77:926-931.
19 Willén H, Carlén B, Rydholm A, et al. Solitary fibrous tumor of the soft tissue. Acta Orthop Scand. 1999;Suppl. 289:31-32.
20 Clayton AC, Salomao DR, Keeny GL, et al. Solitary fibrous tumor: a study of cytologic features of six cases diagnosed by fine-needle aspiration. Diagn Cytopathol. 2001;25:172-176.
21 Domanski HA, Carlén B, Sloth M, et al. Elastofibroma dorsi has distinct cytomorphologic features making diagnostic surgical biopsy unnecessary. Cytomorphologic study with clinical, radiologic and electron microscopic correlation. Diagn Cytopathol. 2003;29:327-333.
22 Pereira S, Tani E, Skoog L. Diagnosis of fibromatosis colli by fine needle aspiration. Cytopathol. 1999;10:25-29.
23 Sharma S, Mishra K, Khanna G. Fibromatosis colli in infants. A cytologic study of eight cases. Acta Cytol. 2003;47:359-362.
24 Jadushing IH. Fine needle aspiration cytology of fibrous hamartoma of infancy. Acta Cytol. 1997;41(Suppl. 4):1391-1393.
25 Layfield LJ, Moffat EJ, Dodd EG, et al. Cytologic findings in tenosynovial giant cell tumors investigated by fine-needle aspiration cytology. Diagn Cytopathol. 1997;16:317-325.
26 Meis-Kindblom J, Bjerkehagen B, Böhling T, et al. Morphologic review of 1000 soft tissue sarcomas from the Scandinavian Sarcoma Group (SSG) register. The peer-review committee experience. Acta Orthop Scand. 1999;70(Suppl. 285):18-26.
27 Fletcher CDM, Gustafson P, Rydholm A, et al. Clinicopathologic re-evaluation of 100 malignant fibrous histiocytomas: prognostic relevance of subclassification. J Clin Onc. 2001;19:3045-3050.
28 Berardo M, Powers C, Wakely P, et al. Fine needle aspiration cytopathology of MFH. Cancer. 1997;81:228-237.
29 Klijanienko J, Caillaud JM, Lagacé R, et al. Comparative fine-needle aspiration and pathologic study of malignant fibrous histiocytoma. Cytodiagnostic features of 95 tumors in 71 patients. Diagn Cytopathol. 2002;29:320-326.
30 Merck CH, Hagmar B. Myxofibrosarcoma. A correlative cytologic and histologic study of 13 cases examined by fine needle aspiration cytology. Acta Cytol. 1980;22:137-144.
31 Kilpatrick SE, Ward WG. Myxofibrosarcoma of soft tissues: cytomorphologic analysis of a series. Diagn Cytopathol. 1999;20:6-9.
32 Kilpatrick SE, Ward WG, Bos GD. The value of fine-needle aspiration biopsy in the differential diagnosis of adult myxoid sarcoma. Cancer (Cancer Cytopathol). 2000;90:167-177.
33 Evans HL. Low-grade fibromyxoid sarcoma. A report of 12 cases. Am J Surg Pathol. 1993;17:595-600.
34 Goodlad JR, Mentzel T, Fletcher CD. Low-grade fibromyxoid sarcoma: clinicopathologic analysis of eleven new cases in support of a distinct entity. Histopathol. 1995;26:229-237.
35 Domanski HA, Mertens F, Panagopoulus I, et al. Low-grade fibromyxoid sarcoma is difficult to diagnose by fine needle aspiration cytology: a cytomorphological study of eight cases. Cytopathology. 2008;20:304-314.
36 Domanski HA, Carlén B, Jonsson K, et al. Distinct cytologic features of spindle cell lipoma. A cytologic-histologic study with clinical, radiologic, electron microscopic, and cytogenetic correlations. Cancer (Cancer Cytopathol). 2001;93:381-389.
37 Lamos MM, Kindblom L-G, Meis-Kindblom JM, et al. Fine-needle aspiration characteristics of hibernoma. Cancer (Cancer Cytopathol). 2001;93:206-210.
38 Meis JM, Enzinger FM. Chondroid lipoma. A unique tumor simultating liposarcoma and myxoid chondrosarcoma. AmJ Surg Pathol. 1993;17:1103-1112.
39 Gisselson D, Domanski HA, Höglund M, et al. Unique cytologic features and chromosome aberrations in chondroid lipoma: a case report based on fine.needle aspiration cytology, histopathology, electron microscopy, chromosome banding, and molecular cytogenetics. Am J Surg Pathol. 1999;23:1300-1304.
40 Yang YJ, Damron TA, Ambrose JL. Diagnosis of chondroid lipoma by fine-needle aspiration biopsy. Arch Pathol Lab Med. 2001;125:1224-1226.
41 Kempson RL, Fletcher CDM, Evans HL, et al. Tumors of the soft tissues. Atlas of tumor pathology. Washington DC: AFIP; 2001.
42 Nemanquani D, Mourad WA. Cytomorphologic features of fine-needle aspiration of liposarcoma. Diagn Cytopathol. 1999;20:67-69.
43 Dey P. Fine needle aspiration cytology of well-differentiated liposarcoma. A report of two cases. Acta Cytol. 2000;44:459-462.
44 Vicandi B, Limenez-Hefferman J, Lopez-Ferrer P, et al. Cytologic features of round cell liposarcoma. Cancer (Cancer Cytopathol). 2003;99:28-32.
45 Klijanienko J, Caillaud J-M, Lagecé R. Fine-needle aspiration in liposarcoma: cytologic correlative study including well-differentiated, myxoid, and pleomorphic variants. Diagn Cytopathol. 2004;30:307-312.
46 Domanski HA. Cytologic features of angioleiomyoma: cytologic-histologic study of 10 cases. Diagn Cytopathol. 2002;27:161-166.
47 Ryd W, Mugel S, Ayyash K. Ancient neurilemmoma. A pitfall in the cytologic diagnosis of soft tissue tumors. Diagn Cytopathol. 1988;2:244-247.
48 Klijanienko J, Caillaud JM, Lagacé R, et al. Fine-needle aspiration of leiomyosarcoma. A correlative cytohistopathological study of 96 tumors in 68 patients. Diagn Cytopathol. 2003;28:119-125.
49 Domanski HA, Åkerman M, Rissler P, et al. Fine needle aspiration of soft tissue leiomyosarcoma. An analysis of the most common cytologic findings and the value of ancillary techniques. Diagn Cytopathol. 2006;34:597-604.
50 Bertholf MF, Frierson HF, Feldman PS. Fine needle aspiration cytology of an adult rhabdomyoma of the neck and head. Diagn Cytopathol. 1988;4:152-155.
51 Domanski HA, Dawiskiba S. Adult rhabdomyoma in fine needle aspiration. A report of two cases. Acta Cytol. 2000;44:223-226.
52 Klijanienko J, Caillaud JM, Orbach D, et al. Cyto-histological correlations in primary, recurrent and metastatic rhabdomyosarcoma: the Institut Curie experience. Diagn Cytopathol. 2007;35:482-487.
53 Daneshbod Y, Monabati A, Kumar PV, et al. Paratesticular spindle cell rhabdomyosarcoma diagnosed by fine needle aspiration cytology: a case report. Acta ytol. 2005;49:331-334.
54 Coffin CM. The new international rhabdomyosarcoma classification, its progenitors, and considerations beyond morphology. Adv Anat Pathol. 1997;4:1-16.
55 Åkerman M, Willén H, Carlén B. Fine needle aspiratation of rhabdomyosarcoma. I. A reliable type-diagnosis possible to render? A retrospective study of 23 cases. Acta Ortop Scand. 1996;67(Suppl. 272):55.
56 Athan S, Aksu Ö, Ekinci C. Cytologic diagnosis and subtyping of rhabdomyosarcoma. Cytopathol. 1998;9:389-397.
57 Udayakumar AM, Sundareshan TS, Appaji L, et al. Rhabdomyosarcoma: cytogenetics of five cases using fine-needle aspiration samples and review of the literature. Ann Genet. 2002;45:33-37.
58 Hollowood K, Fletcher CDM. Rhabdomyosarcoma in adults. Semin Diagn Pathol. 1994;11:47-57.
59 Domanski HA, Åkerman M, Engellau J, et al. Fine-needle aspiration of neurilemmoma. A clinicocytopathologic study of 116 patients. Diagn Cytopathol. 2006;34:403-412.
60 Liu Z, Madden JF, Olatidoye BA, et al. Features of benign granular cell tumor on fine needle aspiration. Acta Cytol. 1999;43:552-557.
61 Liu Z, Mira JL, Vu H. Diagnosis of malignant granular cell tumor by fine needle aspiration cytology. A case report. Acta Cytol. 2001;45:1011-1021.
62 Wieczorek TJ, Krane JF, Domanski HA, et al. Cytologic findings in granular cell tumors with emphasis on the diagnosis of malignant granular cell tumor by fine-needle aspiration biopsy. Cancer. 2001;93:398-408.
63 Jimenez-Hefferman JA, Lopéz-Ferrer PO, Vicandi B, et al. Cytologic features of malignant peripheral nerve sheath tumor. Acta Cytol. 1999;43:175-183.
64 Klijanienko J, Caillaud J-M, Lagacé R, et al. Cytohistologic correlations of 24 malignant peripheral nerve sheath tumors (MPNST) in 17 cases. The Institut Curie experience. Diagn Cytopathol. 2002;27:103-108.
65 Wakely PE, Frable WJ, Kneisl JS. Aspiration cytopathology of epithelioid angiosarcoma. Cancer (Cancer Cytopathol). 2000;90:245-251.
66 Minimo C, Zakowski M, Lin O. Cytologic findings of malignant vascular neoplasms: a study of twenty-four cases. Diagn Cytopathol. 2002;26:349-355.
67 Klijanienko J, Caillaud KM, Lagacé R, et al. Cytohistologic correlations in angiosarcoma including classic and epithelioid variants: Institut Curie experience. Diagn Cytopathol. 2003;29:140-145.
68 Åkerman M, Rydholm A. Aspiration cytology of intramuscular myxoma. A comparative clinical, cytologic and histologic study of ten cases. Acta Cytol. 1983;27:505-510.
69 Nielsen GP, O’Conell JX, Rosenberg AE. Intramuscular myxoma: a clinipathologic study of 51 cases with emphasis on hyper cellular and hypervascular variants. Am J Surg Pathol. 1998;22:1222-1227.
70 Cardillo M, Zakowski MF, Lin O. Fine-needle aspiration of epithelioid sarcoma. Cytologic findings in nine cases. Cancer (Cancer Cytopathol). 2001;93:246-251.
71 Yildiz I, Onder S, Kutlay L, et al. Cytology of epithelioid sarcoma. Cytopathology. 2006;17:305-307.
72 Creager AJ, Pitman MB, Geisinger KR. Cytologic features of clear cell sarcoma (malignant melanoma) of soft parts: a study of fine needle aspirates and exfoliative specimens. Am J Clin Pathol. 2002;117:217-224.
73 Tong TR, Chow TC, Chan OW, et al. Clear cell sarcoma diagnosis by fine-needle aspiration: cytologic, histologic, and ultrastructural features; potential pitfalls; and literature review. Diagn Cytopathol. 2002;26:174-180.
74 Moffat EJ, Liu K, Layfield J. Demonstration of myxoid change in fine-needle aspiration of synovial sarcoma: a case report. Diagn Cytopathol. 1998;18:188-191.
75 Klijanienko J, Caillaud J-M, Lagacé R, et al. Cytohistological correlations in 56 synovial sarcomas in 56 patients: the Institut Curie’s experience. Diagn Cytopathol. 2002;27:96-102.
76 Åkerman M, Ryd W. Skytting B- Fine-needle aspiration of synovial sarcoma: criteria for diagnosis; retrospective examination of 37 cases, including ancillary diagnostics. A Scandinavian Sarcoma Group study. Diagn Cytopathol. 2003;28:232-238.
77 Folpe AL, Schmidt RA, Chapman A, et al. Poorly differentiated synovial sarcoma: immunohistochemical distinction from primitive neuroectodermal tumors and high-grade malignant MPNST. Am J Surg Pathol. 1998;22:673-682.
78 Silverman JE, Landreneau RJ, Sturgis CD, et al. Small-cell variant of synovial sarcoma: fine-needle aspiration with ancillary features and potential pitfalls. Diagn Cytopathol. 2000;23:118-123.
79 Kwon MS. Aspiration cytology of pulmonary small cell variant of poorly-differentiated synovial metastatic from the tongue: a case report. Acta Cytol. 2005;49:92-96.
80 Åkerman M, Domanski HA. The complex cytologic features of synovial sarcoma in fine needle aspirates; an analysis of four illustrative cases. Cytopathology. 2007;18:234-240.
81 Nilsson G, Ming MD, Weide J, et al. Reverse transcriptase polymerase chain reaction on fine needle aspirates for rapid detection of translocations in synovial sarcoma. Acta Cytol. 1998;42:1317-1324.
82 Calafati SA, Wright AL, Rosen SE, et al. Fine needle aspiration cytology of extraskeletal chondrosarcoma. Acta Cytol. 1984;28:81-85.
83 Willén H, Lemos M, Ryd W. Extraskeletal myxoid chondrosarcoma. A cytologic and histologic correlation. 1st Italian/Scandinavian Sarcoma Group Meeting – ISG/SSG. Acta Orthop Scand, 2000. http://home.pi.se/actaorthopscand/pages/framabst.html.p33.
84 Jakowski JD, Wakeley PE. Cytopathology of extraskeletal myxoid chondrosarcoma. Report of 8 cases. Cancer (Cancer Cytopathol). 2007;111:298-305.
85 Domanski HA, Carlén B, Mertens F. Extraskeletal myxoid chondrosarcoma with neuroendocrine differentiation: a case report with fine-needle aspiration biopsy, histopathology, electron microscopy, and cytogenetics. Ultrastructural Pathology. 2003;27:363-368.
86 Logrono R, Wojtowycs MM, Wunderlich DW, et al. Fine needle aspiration cytology and core biopsy in the diagnosis of alveolar soft part sarcoma presenting with lung metastases. A case report. Acta Cytol. 1999;43:464-470.
87 Lopez-Ferrer P, Jimenez-Hofferman JA, Vicandi B, et al. Cytologic features of alveolar soft part sarcoma: Report of three cases. Diagn Cytopathol. 2002;27:115-119.