CHAPTER 5 Lymph nodes
Clinical aspects
Lymphadenopathy is a commonly encountered clinical problem which has a multitude of causes. The commonest cause of peripheral lymphadenopathy is a non-specific reactive hyperplasia in which the underlying etiology is infrequently found (probably an asymptomatic inflammatory process). In general practice, less than 1% of patients with peripheral lymphadenopathy have a malignant process.1 In comparison, retroperitoneal or intra-abdominal lymphadenopathy is usually malignant. In contrast, in young patients, intrathoracic lymphadenopathy is often associated with infectious mononucleosis, sarcoidosis and tuberculosis. The likelihood of malignant disease as a cause of peripheral lymphadenopathy increases over the age of 40 years, nodes over 2 cm in size, firm or matted nodes and non-tender/non-painful nodes.2 Ultrasound is also used in the assessment of lymphadenopathy, from the point of view of a reactive versus a pathological process.3 The bounds of the ‘triple test’ are being expanded beyond breast pathology.
Although surgical excision of a palpable peripheral node is relatively simple, vicinity to other anatomical structures in the neck sometimes causes problems. The procedure does require anesthesia, strict sterility and theater time, and it may leave a scar. To avoid surgery, patients are usually watched for some time before a decision of open biopsy is taken, unless the clinical suspicion of malignancy is strong. Fine needle biopsy (FNB) offers the alternative of an immediate, preliminary, although not always specific diagnosis with little trauma and cost, thus providing ample information for further management.4-8
The plethora of monoclonal antibodies available has proven invaluable in lymph node cytology.9 In particular, it assists in identifying the source of metastatic tumor to lymph nodes, and in distinguishing between various small and large cell neoplasms and malignant lymphoma. The role of cytology in the diagnosis and typing of lymphoma has become better defined in recent years, principally due to immunophenotyping supplementation of routine cytologic preparations. Flow cytometry (FCM) has revolutionized lymphoma diagnosis and typing by FNB and plays a pivotal role.10,11 Immunophenotyping can also be accomplished by immune studies on cytocentrifuge preparations (cytospins) or cell blocks of lymph node aspirates, or core needle biopsy (CNB) specimens.9,12-20 In many cases, definitive diagnosis and typing of lymphoma is possible if cytomorphology is combined with analysis of immunophenotype, and/or cytogenetics and molecular studies.21-32
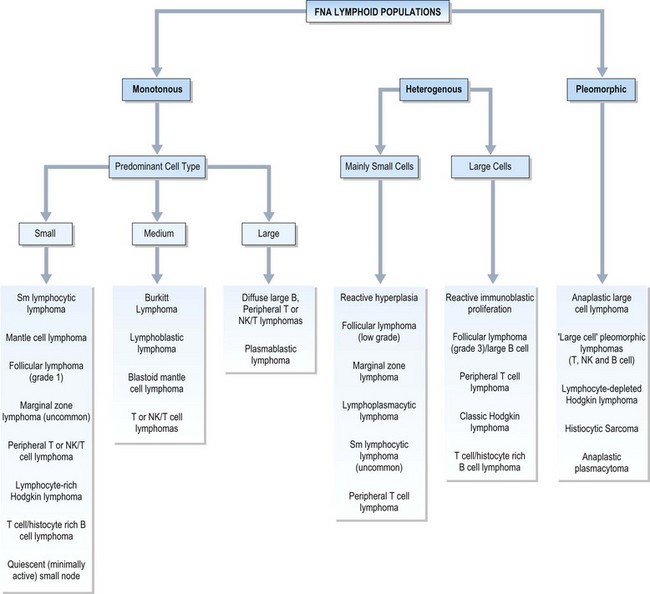
The place of FNA in the investigative sequence
As a rule, cytological examination of FNB smears can determine whether lymphadenopathy is due to reactive hyperplasia, infection, metastatic malignancy or malignant lymphoma. In order to make the most rational use of fine needle aspiration cytology (FNAC), clinicians and pathologists alike must understand and accept that the aims and purpose of FNB of peripheral lymph nodes at the ‘primary’ or ‘community’ level are different from those at the ‘secondary’ or ‘specialist’ level. At the primary level, FNAC is used as a triage to distinguish between cases of lymphadenopathy with a high or a low level of suspicion of significant disease by the simplest, least invasive and least costly method. This preliminary assessment is based on routine cytologic smears only. Depending on the result, an immediate decision can be made whether to simply observe the node, to recommend a course of antibiotics, or to refer the patient to a specialist for further investigations. Reactive lymphadenopathy is likely to resolve spontaneously in due course. Since the great majority of lymphadenopathy cases seen at the primary level are reactive, this approach has obvious practical, economical and psychological advantages.
In case of reactive hyperplasia, surgical excision is not indicated, unless the subsequent course is atypical or there is significant discrepancy with the clinical or imaging findings. Follow-up is still recommended, since there is a small but significant risk that lymphoma or other malignancy could be missed or undercalled by FNB. A specific diagnosis can be made in some cases in this category, for example of an infectious process, but most often the etiology remains obscure. If appropriate, and depending on the clinical presentation, part of the biopsy material may be used for microbiological investigation, which can be supplemented by serology.
At the specialist/secondary level, the role of FNB is to provide material for further cytomorphologic analysis and for ancillary studies. The aim is to arrive at a definitive diagnosis and lymphoma typing making full use of the armamentarium of ancillary laboratory techniques. This also applies to most abnormal lymph nodes in deep sites. In some centers, this strategy has been very successful and has virtually replaced surgical removal of nodes. This requires readily available expertise in FCM, cytogenetics, immunostaining, molecular analysis (e.g. PCR), haematopathology and cytopathology. Others, including the author, feel that a tissue sample obtained by CNB or surgical biopsy is often still needed. Lymphoma diagnosis and typing relies not only on cytology but also on altered tissue and immmuno-architecture,33 which requires a tissue sample, although a small fragment obtained by CNB or occasionally a cell block may suffice. Tissue samples can also help to overcome deviations from classical immunophenotypes on FCM. They also enable spatial recognition, assisting in the diagnosis of various reactive lymphadenopathies. Close correlation of cytological, immunological and clinical features is essential when using FCM to make a diagnosis of lymphoma and in its classification. At times, additional correlation with the cytogenetic and molecular studies will be necessary.
Lymph nodes clinically suspected of metastatic malignancy constitute one of the commonest indications for FNB. In patients with known, histologically proven malignancy, who subsequently present with lymphadenopathy, a cytological diagnosis may obviate further surgery, merely to confirm metastasis. In patients without a previous malignant diagnosis, not only can metastatic malignancy be confirmed by FNB, but clues to the nature and site of the primary can also be given in most cases. Cytocentrifuge preparations or cell blocks, or multiple smears when used for histochemical and immunohistochemical staining, are of great value in this situation. Only when FNB and supplementary investigations do not provide clues to the origin of the metastasis, and when this information is of importance to clinical management, is a diagnostic surgical excision or CNB of the node indicated. The distinction between anaplastic carcinoma and lymphoma is obviously of clinical importance. In addition, a specific diagnosis by FNB of disseminated carcinoma of the prostate, breast, ovary and thyroid, germ cell tumors and neuroendocrine carcinoma should be pursued since treatment is available.
The value of FNB in the investigation of suspected lymphoma can be summarized as follows:
Accuracy of diagnosis
The accuracy of FNAC of lymph nodes in the diagnosis of metastatic malignancy is influenced by many factors such as the size and site of the node, fibrosis, necrosis, previous irradiation and the number of punctures made. Small mobile nodes high up in the axilla are difficult to sample, while adequate material can easily be obtained from nodes only a few millimeters in diameter in a cervical or supraclavicular position.37 Deep nodes are accessible to FNB by means of radiological imaging and guidance.38,39 Fibrosis sometimes makes it difficult to obtain sufficient material for diagnosis from reactive inguinal nodes. It can also be a problem in nodular sclerosis Hodgkin lymphoma40 and in some mediastinal and retroperitoneal non-Hodgkin lymphomas (NHL).9
Diagnostic sensitivity is occasionally limited by the fact that small metastatic deposits, metastases confined to the subcapsular sinus and single-cell metastases can be missed. However, early micrometastases rarely produce significant lymph node enlargement and if a lymph node is palpable it is likely to contain enough tumor tissue to be detectable by FNB. The diagnostic sensitivity of metastatic and recurring malignancy reported in the literature is usually above 95%.23,41-53 Failure to obtain a representative sample is responsible for most false-negative diagnoses. Interpretation of a representative sample can be problematic, more often with lymphoma than with metastatic malignancy. For example, without immunophenotyping low-grade follicular lymphoma can easily be mistaken for reactive follicular hyperplasia.54 Thus, although a negative cytological report makes malignancy unlikely, it is not singularly diagnostic,37 and if the lymphadenopathy does not show signs of regression within a month or two, FNB should be repeated or a node should be excised for histology.
Diagnostic specificity for malignancy, on the other hand, is high. False-positive diagnoses are rare23,42,48,52-57 if particular caution is observed in the interpretation of smears from nodes in fields of previous irradiation and in the presence of necrosis. The existence of benign epithelial inclusions in lymph nodes (see p. 89) should be kept in mind. Again, the main problem is in relation to lymphoma, particularly the distinction between reactive follicular hyperplasia and follicular lymphoma. Most false-positive diagnoses are cases of reactive lymphadenopathy reported as suspicious of lymphoma.
Diagnostic accuracy not only depends on the aspirate being representative, but also very much on the quality of the cytological preparations. This is particularly the case in the diagnosis of some reactive lymphadenopathies and in the diagnosis and classification of lymphoma, which depends on the study of fine cytological detail in high magnification, and on an estimate of proportions of various cell types in the smear.54,55,58-60 It is essential that the aspirates are handled and smears prepared by staff with experience in cytology to achieve satisfactory results.
There are conflicting opinions regarding the accuracy of cytological diagnosis and typing of malignant lymphoma.19-26,30,55,60-65 It is difficult to extract exact figures for several reasons. Most series include relatively small numbers of cases, with histologic correlation and/or consistent follow-up. Early series are based on cytomorphology (in FNB smears) alone, later series combine cytomorphology and immunophenotyping by flow cytometry and/or immunostaining of smears. The case mix (types of lymphoma; primary diagnosis or recurrent disease) is variable and often is not specified. With these reservations, it appears that the accuracy of diagnosing NHL by cytomorphology alone is in the range 60–80%, significantly lower for low-grade than for high-grade lymphomas.26,37,45,48,51,66
Diagnostic sensitivity has generally been found to be lower for lymphoma than for metastatic malignancy.23,48,67,68 For a diagnosis of lymphoma to be of clinically practical value, it must identify good and bad prognosis subgroups and therefore must specify the subtype. In an extensive review of the literature, two-thirds of the 30 studies reviewed, in which FNB was supplemented by immunophenotyping, diagnostic sensitivity was over 80% and specificity over 90%. However, only three of eight studies achieved a correct and precise subtyping in the primary diagnosis of NHL in more than 80% of cases.22 Difficulties in correlating cytology and histology of lymphoma have been enhanced by the parallel use and frequent modifications of different systems of classification over the years. The advent of the World Health Organization (WHO) lymphoma classification based on clinical, cytologic, immunophenotypic and genetic features has improved both the detection rate and classification of NHL, due to diminished importance placed on architectural features in subtyping NHL.22,26,30,32 However, the accuracy of cytologic diagnosis is still limited in some forms of NHL, notably lymphomas with predominantly small cells, mainly marginal zone lymphoma, and in peripheral T-cell lymphoma, T-cell/histiocyte rich B-cell lymphoma and nodular lymphocyte predominant Hodgkin lymphoma. This is also the case with the diagnosis of composite lymphoma and in the grading of follicular lymphoma.
The wide variation in the reported accuracy of diagnosis and typing of NHL probably reflects the environment in which the studies were carried out. A high accuracy has been achieved in a few highly specialized centers equipped with the full armamentarium and expertise related to ancillary laboratory techniques, but many hospitals have limited resources, and most pathologists still consider histology necessary in the primary diagnosis of lymphoma. This enables determination of any altered tissue and immunoarchitecture, and also reveals the cellular compartments and their composition in the tissue section, which is not reflected per se in the aspirated cell sample.
As a rule, FNB samples from malignant lymphoma are very cellular and can be used for ancillary studies.9,31,54,69-72 However, more material to allow a wider range of immune markers, assessment of tissue and immunoarchitecture as well as storage of material for ancillary studies and research can be obtained by supplementary CNB of the node, principally in intra-abdominal or intrathoracic nodal disease. With deep-seated masses, CNB is a useful adjunct to FNB. The FNB provides superior cytomorphology and is an excellent source for flow cytometry cell suspensions. This combined approach can increase diagnostic accuracy, assist in classification and reduce the number of insufficient samples.73,74
The accuracy of cytological diagnosis of Hodgkin lymphoma (HL) is quite variable, frequently high, and in some studies greater than 85%.22,23,75-77 The main problems in cytodiagnosis are in relation to the nodular lymphocyte predominant and lymphocyte-rich variants as well as separation from HL mimicks. The accuracy of subtyping HL in FNB smears is relatively poor.22-24 Performing immunocytochemistry on cell block preparations, cytospins or NCB may help to a degree but a formal node biopsy is still necessary to provide reliable subtyping in most cases.
Complications
Significant complications do not occur. Post-aspiration hematoma or necrosis is rare.78,79 To date, septic complications or tumor implantation in the needle track have not been reported following FNB of lymph nodes.
Technical considerations
Both reactive nodes and nodes involved by metastatic malignancy or lymphoma are highly cellular and moderately vascular tissues. Sufficient material is therefore easily obtained using 23–27-gauge needles, except sometimes in the presence of fibrosis or necrosis. FNB without aspiration has been used routinely for several years in some institutions. It has the advantage over the traditional aspiration of giving the operator a more direct and sensitive feeling of the consistency of tissues through the needle. This is helpful when small or deep nodes are biopsied. Non-aspiration also results in less admixture with blood. An abundance of blood adversely affects cell fixation and tends to cause shrinkage and distortion of cells. If aspiration is used, multiple rapid biopsies from different points of entry are preferable to multiple passes in different directions, in order to obtain a representative and adequate sample without too much blood.
Local anesthetic is not used and simple skin disinfection as for an injection is adequate. Two or more samples may be necessary to secure enough material for both routine smears and for special investigations and to reduce sampling error in focal disease. The use of gloves and extreme care in handling used needles are important safety precautions. The techniques involved in the biopsy of deep nodes using radiological guidance are described in Chapter 3. If the standard technique does not yield sufficient material, for example due to fibrosis (nodular sclerosis Hodgkin lymphoma and some sclerosing NHLs), a cutting core needle can be tried.34,35 In general, large-caliber core needles of 14–18 gauge are recommended in the definitive diagnosis and typing of NHL, as they allow a more accurate assessment of tissue and immunoarchitecture as mentioned above. Artifactual change is less than with a 20-gauge needle.
It is not easy to make perfect direct smears from samples of lymphoid tissue and this takes considerable practice. An air-dried smear has to dry quickly for optimal fixation and has to be made thin and even. The smearing pressure must be well balanced to obtain a thin smear and at the same time avoiding crush artifacts. If the aspirate is bloody or thinned by a large amount of lymph fluid, cells need to be concentrated and separated as much as possible from the fluid. This can be achieved by using the two-step technique illustrated in Figure 2.7. A wet-fixed smear must be fixed immediately to minimize drying artifacts. Only those parts of the smears where the cells are evenly dispersed, well fixed and not distorted by the trauma of smearing should be chosen for diagnostic evaluation.
While air-dried MGG or Diff-Quik-stained smears are essential for the evaluation of cytoplasm and background elements, alcohol fixation and staining with H&E or Pap is helpful in assessing nucleoli and chromatin pattern. Whenever possible, both air-dried and wet-fixed smears should be made of each FNB sample as they may provide complementary information. Extra smears to allow cytochemical stains are often of great value. Staining for microorganisms (Ziehl-Neelsen, PAS, Gomori’s silver stain), mucin (PAS/diastase, Alcian blue), melanin (Masson-Fontana), and immunocytochemical studies are those most commonly used.
Heavy reliance is placed on ancillary techniques in FNB of nodes. A cell suspension made by gently dispersing the sample in Hank’s balanced salt solution with 10–20% fetal calf serum or by rinsing the needle with the fluid is a suitable preparation for this purpose. The suspension is spun in a cytocentrifuge at 300–700 rpm for 3–5 minutes. Processing should be done as soon as possible after biopsy since cell fragility increases rapidly with time. Cytospin preparations and cell blocks/cell buttons (see Chapter 2) allow assessment by a panel of immune markers by immunocytochemical staining as illustrated in the section on lymphoma. An elementary NHL screening panel could consist of CD20, CD3, CD5, kappa and lambda light chains, and this can be extended as appropriate. Background staining due to serum proteins and fragmented cells can be reduced by resuspending the cells in Hank’s fluid after initial centrifugation. Direct smears may be unsuitable due to the dispersal of cytoplasm and proteins in the background (see Chapter 2). However, immunostaining of FNB smear specimens can be utilized with the use of formal saline fixation. It eliminates background staining while allowing preservation of cellular antigenicity and morphology.80 Immunocytochemistry is helpful in tracing the origin of metastatic malignancy, in the differentiation of lymphoma from reactive processes and from anaplastic carcinoma or melanoma, and in the classification of lymphoma.13-1618 FCM analysis of aspirated cells is vital in immunophenotyping lymphoid populations, determining clonality and aberrant antigen expression. FNB material can be rinsed in a buffered balanced salt solution or RPMI solution to provide material for analysis. In addition, aspirated material can be used in conventional cytogenetics or fluorescence in situ hybridization (FISH) to assess for chromosomal translocations as well as in molecular studies/PCR to assess gene rearrangements.
If there is any suspicion of an infective process, the needle can be rinsed with sterile saline after the smears have been prepared, but preferably the biopsy should be repeated to provide sufficient material for culture for microorganisms (see p. 18 and Chapter 18).
Liquid-based preparations have been used minimally for nodal FNB samples. If used, they must be interpreted with caution as there are many cytomorphologic alterations.81
Cytological findings
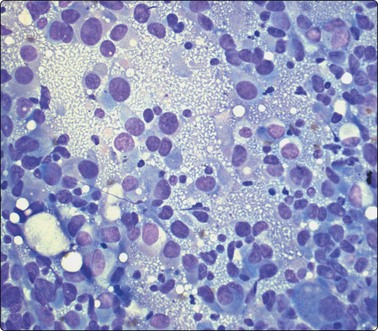
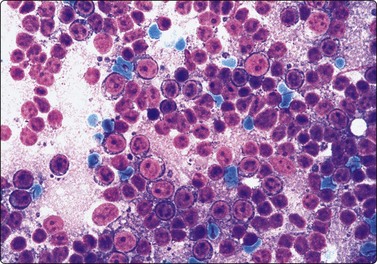
Fine needle biopsy samples of lymphoid tissue, nodal or extranodal, benign or malignant, as a rule have a very high cell content. This is obvious to the naked eye as the aspirate is smeared. It looks like a film of slimy material which turns gray on drying. The cytoplasm of lymphoid cells is fragile. Many cells are represented by naked nuclei or have only a small rim of cytoplasm. A variable number of rounded cytoplasmic fragments measuring up to 8 microns in diameter are scattered in the background (Fig. 5.1). The cytoplasmic fragments were named ‘lymphoglandular bodies’ by Söderström. The term ‘lymphoid globules’ is prefered since they are present in smears from any lymphoid infiltration, not only from lymph nodes. The fragments stain an uniform pale blue, identical to the cytoplasm of intact cells, with Giemsa stain. They differ from necrotic debris by their regular round shape and their uniform staining. Necrotic debris and nuclear fragments (karyorrhexis and apoptotic bodies) are characteristic of smears of small cell undifferentiated carcinoma (Fig. 5.2). The recognition of ‘lymphoid globules’ is of diagnostic value in the distinction of lymphoma from anaplastic carcinoma and some other tumors.
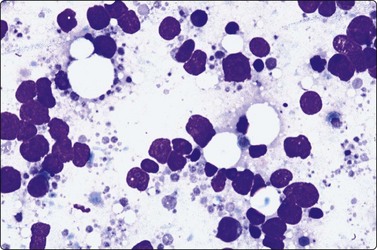
Numerous spherical fragments of blue cytoplasm of variable sizes dispersed between the lymphoid cells; some nuclear fragments. Large cell lymphoma of tonsil (MGG, HP).
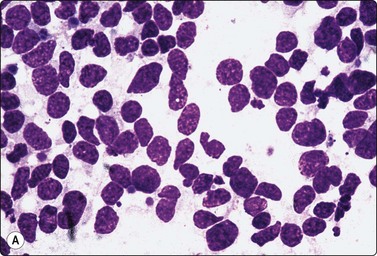
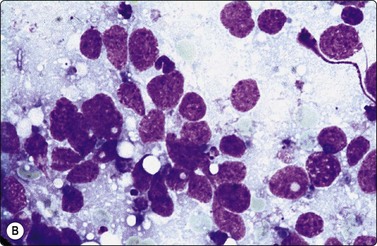
Fig. 5.2 Small cell anaplastic carcinoma
Dispersed malignant cells with dense chromatin, irregular nuclear contour, nuclear molding, inconspicuous nucleoli and cytoplasm; note irregular cytoplasmic (B) and nuclear fragments in the background representing tumor necrosis (A and B) (MGG, HP).
Most of the lymphoid cells are dispersed as single cells but some may form clumps or aggregates, especially in smears of bloody samples. Cell detail is obscured in dense clusters, which are of no diagnostic value as they can be found in both reactive and malignant nodes. However, a tendency for neoplastic follicular center cells to form aggregates resembling neuroendocrine carcinoma is commonly seen in follicular lymphoma,82,83 and cells of anaplastic large cell lymphoma can also form carcinoma-like aggregates.84-86 Tissue fragments consisting of a vascular core of endothelial cells with adherent lymphoid cells and histiocytes are sometimes present in smears from reactive nodes (Fig. 5.3).
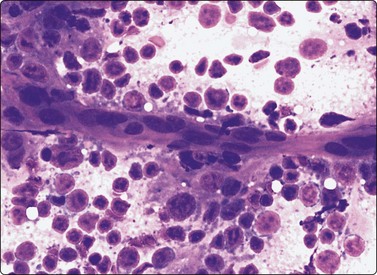
Fig. 5.3 Fragment of lymphoid tissue
Lymphoid cells and histiocytes adhering to a strand of endothelial cells representing a small blood vessel (MGG, HP).
The reactive node (Figs 5.4-5.16)
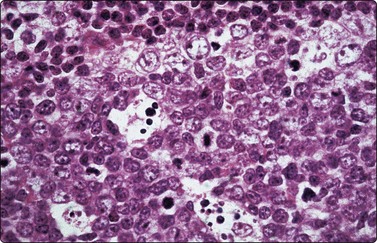
Fig. 5.4 Reactive lymphadenopathy, follicular
Tissue section showing detail of germinal center with centroblasts, centrocytes and tingible body macrophages. Outer rim of small lymphocytes. The corresponding cytological pattern is shown in Figure 5.5 (H&E, HP).
(Reproduced with permission from van Heerde et al.9)
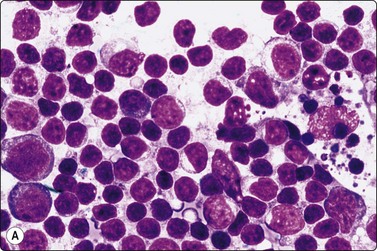
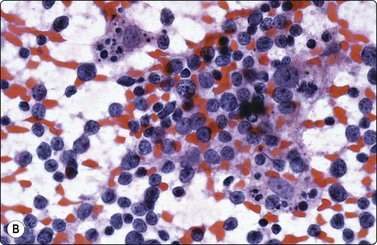
Fig. 5.5 Reactive lymphadenopathy, follicular
Smear derived from reactive follicle. Centroblasts, centrocytes, small lymphocytes and tingible body macrophages with nuclear fragments (A, MGG; B, Pap; HP).
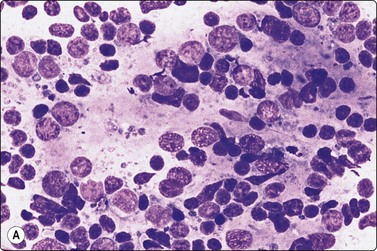
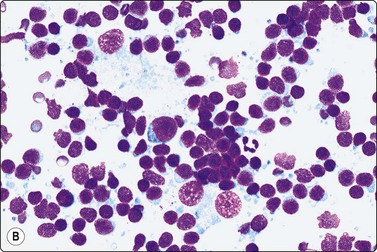
Fig. 5.6 Reactive lymphadenopathy
(A) Smear derived from germinal center; loose tissue fragment of dendritic reticulum cells with ovoid nuclei and granular chromatin, centroblasts, centrocytes and some lymphocytes; syncytial background of pale cytoplasm; (B) Mixed population of lymphoid cells; two nuclei of dendritic reticulum cells with granular chromatin and small distinct nucleoli lower mid (MGG; HP).
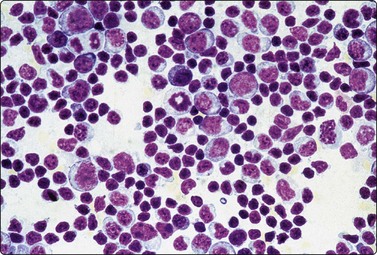
Fig. 5.7 Reactive lymphadenopathy
Paracortical hyperplasia, viral induced. Spectrum of basophilic cells, ranging from mature plasma cells to immunoblasts with a background of many small lymphocytes, favors a reactive lesion (MGG, HP).
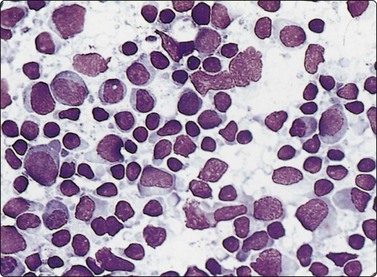
Fig. 5.8 Infectious mononucleosis
Many transforming lymphocytes, plasmacytoid cells and immunoblasts (MGG, HP).
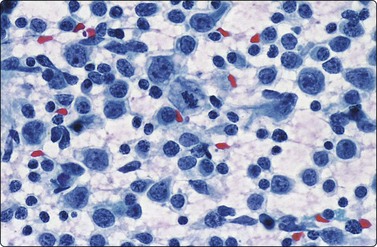
Fig. 5.9 Infectious mononucleosis
A similar pattern to Figure 5.8 of a high proportion of transformed lymphocytes and a mitotic figure (Pap, HP).
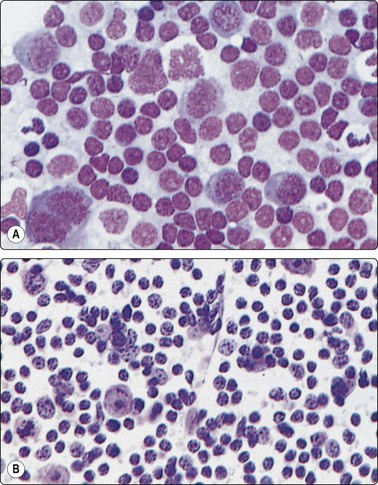
Fig. 5.10 Immunoblastic reaction
Several immunoblasts including a binucleate form; case of Dilantin lymphadenopathy (A, MGG, HP; B, H & E, HP).
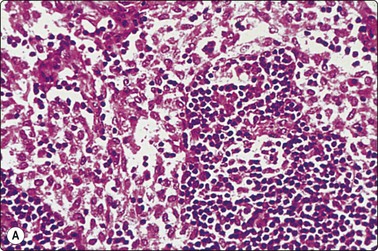
(A) Tissue section. Marginal sinus filled with large pink-stained histiocytes (H&E, IP); (B) Smear showing histiocytes with abundant pale blue vacuolated cytoplasm and phagocytosis of a lymphocyte near the centre right (MGG, IP).
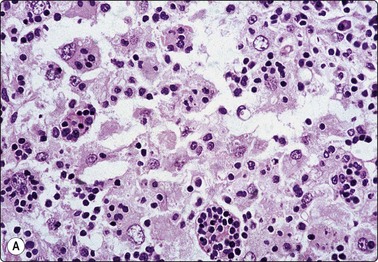
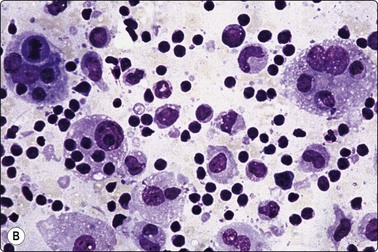
Fig. 5.12 Sinus histiocytosis with massive lymphadenopathy (Rosai-Dorfman disease)
(A) Tissue section showing very large histiocytes harboring many lymphocytes (and some plasma cells) in their cytoplasm, (H&E, IP); (B) Smear showing large histiocytes with intracytoplasmic lymphocytes and plasma cells (MGG, IP).
(Reproduced with permission from van Heerde et al.9)
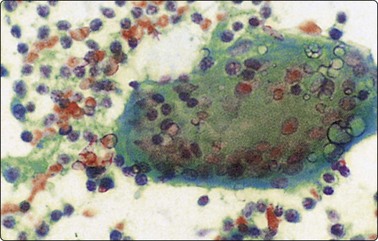
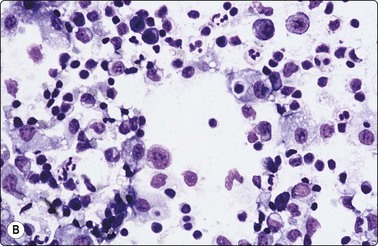
Histiocytes and large multinucleated foreign body-type giant cell with cytoplasmic vacuoles (Pap, IP).
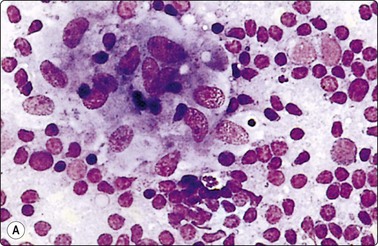
(A) Granuloma-like cluster of histiocytes with epithelioid features; (B) Activated lymphoid cells possibly including some monocytoid B-cell forms. (MGG, HP).
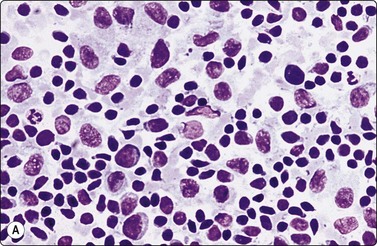
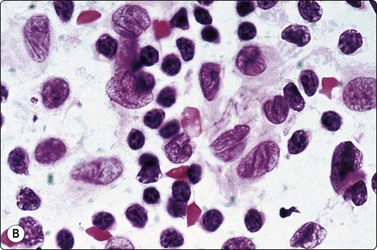
Fig. 5.15 Dermatopathic lymphadenopathy
(A) Numerous pale histiocytes/interdigitating cells in a background of predominantly small lymphocytes. Intracytoplasmic pigment was sparse in this case (MGG, IP). (B) Interdigitating cells with conspicuous nuclear folding due to long channel-like invaginations of the nuclear membrane (H&E, HP).
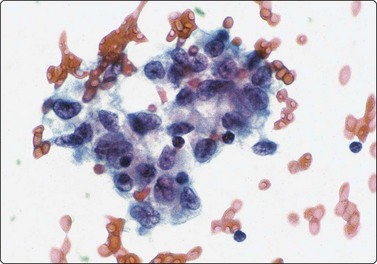
Fig. 5.16 Langerhans histiocytosis
Loose cluster of large histiocytic cells with large, vesicular, irregularly folded, lobated or convoluted nuclei; no eosinophils seen in this example (Pap, HP).
Criteria for diagnosis
The reactive pattern is variable depending on the degree of stimulation, the number and size of germinal centers and on whether the sample derives mainly from a germinal center or from interfollicular or paracortical tissue. Germinal center material is represented by poorly defined tissue fragments composed of centroblasts, centrocytes, ‘tingible body’ macrophages (Figs 5.4, 5.5), and a number of lymphocytes which adhere to the syncytial cytoplasm (pale gray/violet in MGG) of dendritic reticulum cells (Fig. 5.6A). Dendritic reticulum cells have oval or round nuclei with a smooth nuclear membrane, a coarsely granular, uniformly distributed chromatin and small distinct nucleoli (Fig. 5.6B). The cytoplasm is dispersed in the background. A smear, which derives mainly from interfollicular tissue, consists predominantly of lymphocytes with a variable but much smaller number of scattered immunoblasts, plasma cells, non-specific histiocytes and endothelial cells (Fig. 5.7). Multiple biopsies diminish the bias caused by selective sampling.
The main features which distinguish a reactive process from lymphoma are:
Axillary nodes undergoing fat involution are sometimes sampled as they may become quite large, although of soft consistency. Smears of such nodes show fat droplets, fragments of adipose tissue, a number of small lymphocytes and a few blasts. The predominance of small lymphocytes may cause a suspicion of CLL (see section on small cell lymphoma). Fibrotic but otherwise normal inguinal nodes also give a scanty yield.
Morphologically similar to reactive lymphoid hyperplasia and rarely distinguishable are HIV-associated lymphadenitis, progressive transformation of germinal centers and Castleman’s disease. When germinal centers are very large, as in some cases of reactive follicular hyperplasia, the proportion of large cells (centroblasts, dendritic reticulum cells) and the number of mitoses, in a FNB sample, may be impressive enough to suggest malignant lymphoma. However, the full range of lymphocyte transformation is still preserved, including small lymphocytes, and the various cell types occur in logical proportions. Small or slightly enlarged lymphocytes are still numerically predominant. A variable number of plasma cells can usually be found. The presence of macrophages with tingible bodies favors reactive hyperplasia but does not rule out lymphoma. Especially in high-grade lymphomas with a high turnover of cells, a considerable number of ‘starry sky’ macrophages may be present (see Figs 5.48 and 5.51A). A cytological pattern of reactive hyperplasia with a large number of plasma cells but no other distinguishing features can be seen, for example, in cases of secondary syphilis, rheumatoid arthritis, autoimmune syndromes, IgG4-related lymphadenopathy, HIV infection and the plasma cell variant of Castleman’s disease.
The differential diagnosis between prominent follicular hyperplasia and follicular lymphoma grade 1–2 can be very difficult in FNB smears even for experienced cytopathologists. The smear patterns of follicular hyperplasia have been described. In follicular lymphoma, the predominant cell type may appear small, but the nucleus is of intermediate size and has an irregular shape and a more granular chromatin similar to a centrocyte. Immunoblasts, plasma cells and tingible body macrophages are usually absent or few in numbers. The difficulty in distinguishing between the two conditions is largely due to the fact that dendritic reticulum cells associated with centroblasts and centrocytes are seen in both conditions, and that interfollicular areas in follicular lymphoma may contain large numbers of lymphocytes. Immunological demonstration usually by FCM of poly- or monoclonality may be necessary to solve the problem.
A prominent immunoblastic and plasmacellular reaction is found in several conditions. In viral lymphadenitis, particularly in infectious mononucleosis,89,90 immunoblasts, plasmacytoid cells, mature plasma cells and atypical lymphocytes can be numerous but the range of cells is still in logical proportions (Figs 5.7, 5.8 and 5.9). The atypical lymphocytes have an abundant basophilic cytoplasm, an enlarged, often eccentric, nucleus and a paler nuclear chromatin than a normal lymphocyte. Immunoblastic cells can cause differential diagnostic problems. The main differential diagnoses are large cell lymphoma (with numerous immunoblasts) and HL. Atypical binucleate immunoblasts closely resembling Reed-Sternberg cells are rarely seen (see Fig. 5.69).91 Occasionally, immunoblasts are mistaken for mononuclear Hodgkin cells; however, they are smaller with smaller nucleoli and greater basophilic cytoplasm than Hodgkin cells. Mononucleosis is usually suggested by the clinical presentation and can be confirmed by serological tests.
Abnormal immunoblastic reactions with prominent immunoblasts and sometimes Reed-Sternberg-like cells can at times be difficult to distinguish from large cell lymphoma. This pattern may be seen, for example, in postvaccinial lymphadenitis and Dilantin hypersensitivity lymphadenitis (Fig. 5.10). The clinical history may provide a clue in such cases (e.g. patients taking Dilantin usually develop adenopathy in the first 6 months of therapy). A definitive diagnosis would require either further ancillary investigation or a formal histological examination. Immunologic studies can help to confirm the polyclonal nature of the immunoblasts.
Histiocytes, which have an abundant, pale or sometimes eosinophilic cytoplasm, may be prominent in smears of lymph node aspirates. The histiocytes occur singly or in small groups. The cytoplasm is often vacuolated or granular and may contain phagocytosed debris or pigment. An increased number of histiocytes without specific features can be seen in smears from non-specific reactive nodes, and are suggestive of sinus histiocytosis (Fig. 5.11). The cytological findings in sinus histiocytosis with massive lymphadenopathy (Rosai-Dorfman disease) have been described by van Heerde and others.9,92-94 In this condition, smears contain many large histiocytes showing lymphophagocytosis (Fig. 5.12). Prominent histiocytes and multinucleated giant cells as a reaction to foreign material can occasionally be seen in axillary nodes of women with silicone breast prostheses95 (Fig. 5.13) or inguinal lymphadenopathy associated with artificial hip joint replacement.96
Scattered small clusters of a few histiocytes with ovoid, pale nuclei and abundant cytoplasm resembling epithelioid cells in a smear consistent overall with follicular hyperplasia are suggestive of toxoplasmosis (Fig. 5.14A). Well-formed histiocytic granulomata resembling sarcoid granulomata are unusual but occur occasionally. Lymphoid cells with relatively large, ovoid, pale nuclei may also be seen (Fig. 5.14B). These cells probably correspond to the pale monocytoid B cells observed in histological sections. Occasionally, the presence of many of these large cells can raise a suspicion of malignancy. The cytological pattern is not diagnostic by itself and needs confirmation by serological tests. Microcysts and toxoplasma organisms are rarely found in smears of toxoplasma lymphadenitis (see Chapter 18).97-100
Numerous noncohesive, pale, histiocyte-like cells (interdigitating cells) with typical folded nuclei (Fig. 5.15) are present in dermatopathic lymphadenopathy. Some macrophages containing pigment, either hemosiderin or melanin, are usually found. These have smaller and more consistently oval, nonfolded nuclei different from interdigitating cells, and have a better-defined cytoplasm. There may be a variable number of eosinophils. The background is predominantly of lymphocytes, which may appear slightly ‘atypical’ with small pale, central nucleoli (stimulated T cells) and blast forms are less common.
The histiocytes of Langerhans cell histiocytosis (histiocytosis X) have characteristically large nuclei of irregular shape. They may be folded, convoluted, lobulated and grooved (Fig. 5.16 and Fig. 16.10). Mitotic activity may be seen and sometimes necrosis. Such cells seen in a lymph node aspirate, especially in the absence of eosinophils, may raise a suspicion of metastatic malignancy such as melanoma. However, the nuclear chromatin of Langerhans histiocytes is bland and finely granular. If suspected, the diagnosis may be confirmed by immunocytochemistry (CD1a, S-100, langerin)69,101 and/or by electronmicroscopy (Birbeck granules).9,101-104 Histiocytic sarcoma is described on page 111 (see Fig. 5.76).105
A prominent component of eosinophils may be seen in reactive and neoplastic processes. The reactive conditions are hypersensitivity lymphadenitis, parasitic infestations with granulomas, Churg-Strauss disease and Kimura’s disease. The neoplasms associated with eosinophilia are HL, peripheral T-cell lymphoma, Langerhans cell histiocytosis, mast cell disease, eosinophilic myeloid disorders and some metastatic carcinomas (mainly nasopharyngeal carcinoma).
A prominent component of neutrophils may also be seen in reactive and neoplastic processes. The reactive conditions are acute lymphadenitis, abscess and suppurative granulomas. The neoplasms associated with neutrophilia are HL, anaplastic large cell lymphoma, histiocytic sarcoma and metastatic squamous carcinoma.
Multinucleate giant cells are usually of histocytic origin and frequently associated with granulomas; however, Warthin-Finkeldey multinucleate giant cells (polykaryocytes) are T cells. They can occasionally be seen in reactive lymphadenitis associated with measles, HIV and Kimura’s disease. Additionally, they may be seen with various lymphomas.106
Descriptions of the cytology of angiofollicular lymphoid hyperplasia can also be found in the literature,9,107-109 as well as of Kimura’s disease.110,111 Follicular dendritic sarcoma may arise in association with Castleman’s disease or de novo.112,113 Rare descriptions of interdigitating dendritic cell sarcoma are also in the literature.114
Granulomatous lymphadenitis (Figs 5.17-5.20)
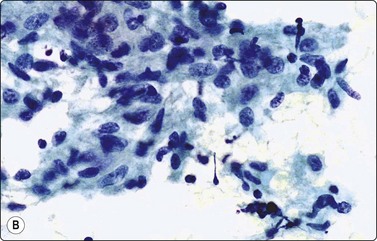
Fig. 5.17 Granulomatous lymphadenitis (sarcoidosis)
Clusters of loosely cohesive epithelioid histiocytes with characteristically pale, elongated sole-shaped nuclei; few lymphocytes; no necrosis; no giant cells seen in this example (A, MGG, IP; B, Pap, HP).
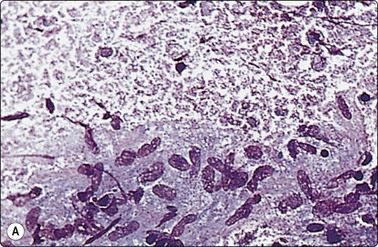
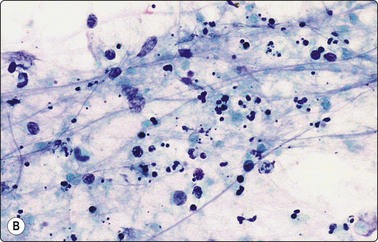
Fig. 5.18 Granulomatous lymphadenitis (tuberculosis)
(A) Granuloma-like group of epithelioid histiocytes in a background of granular caseous necrosis (MGG, HP); (B) Granular material of caseous necrosis with degenerating and fragmented nuclei. Note presence of polymorphs, a not uncommon feature, particularly in AIDS patients (Pap, HP).
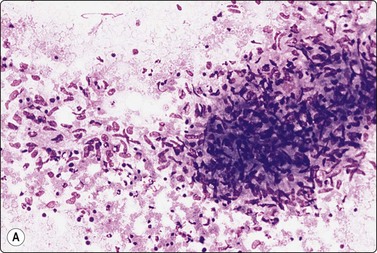
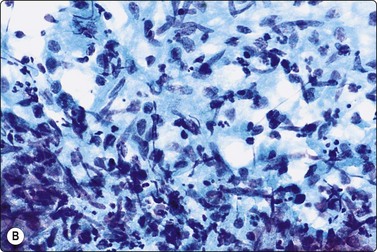
Fig. 5.19 Granulomatous lymphadenitis
(A) Lymphogranuloma venereum. Tissue fragment of epithelioid histiocytes, neutrophils and debris (MGG, LP); (B) Cat scratch disease. Epithelioid histiocytes, lymphocytes and debris with polymorphs (lower), probably corresponding to a ‘stellate abscess’ (Pap, HP).
Cluster of spindle cells resembling epithelioid granuloma but nuclei are more elongated, irregular and hyperchromatic (MGG, HP).
Epithelioid cells are quite distinctive in FNB smears. They have elongated nuclei the shape of which resembles the sole of a shoe or boomerang. The nuclear chromatin is finely granular and pale and the cytoplasm is pale without distinct cell borders (Fig. 5.17). Epithelioid cells of granulomatous lymphadenitis form clusters; large clusters resemble granulomas in tissue sections. Multinucleated Langhans giant cells may be few in numbers and are sometimes absent. Granulomatous lymphadenitis may or may not show necrosis or suppuration. Necrosis may be of fibrinoid or caseous types. Caseous material appears granular and eosinophilic in smears and usually lacks recognizable cell remnants (Fig. 5.18).
Granulomata with caseous necrosis is the hallmark of tuberculous lymphadenitis. However, necrotizing granulomata may also be seen with fungal infections. Granuloma-like clusters of epithelioid cells, in the absence of necrosis, are more suggestive of sarcoidosis, but tuberculosis and fungal infections cannot be ruled out and staining for acid-fast bacilli and fungi is imperative in all cases of granulomatous lymphadenitis. Smears from a tuberculous lymph node may sometimes show only polymorphs and necrotic debris without histiocytes, particularly in immunocompromised patients. Acid-fast bacilli should, of course, be looked for both in direct smears and in culture from the aspirate. PCR is a sensitive way to detect mycobacterial organisms.115-120
Non-necrotizing sarcoidal type granulomata may also be seen with foreign body reactions, brucellosis, Crohn’s disease, leishmaniasis and leprosy. Leprosy in lymph nodes has also been diagnosed by FNAC.121 Conspicuous neutrophils in a smear showing epithelioid granulomas and necrosis – suppurative granuloma (Fig. 5.19) – suggest atypical mycobacterial infection if the aspirate is from a cervical node in a child, cat scratch disease if from an axillary node,122-124 and lymphogranuloma venereum if it is from an inguinal node. Fungal and corynebacterial infections may produce a similar reaction, as can chronic granulomatous disease of childhood, leishmaniasis, Yersinia enterocolitica and tularemia. Occasionally, groups of epithelioid histiocytes in toxoplasmosis may be large enough to mimic true granulomatous lymphadenitis. Inclusions within histiocytes and giant cells such as birefringent particles, asteroid bodies, Schaumann bodies, etc. are not particularly helpful in making a specific diagnosis. If no etiological agent is found, one can only report the case as granulomatous lymphadenitis with or without necrosis and/or suppuration, and the etiology must be pursued by other means.
If an aspirate consists entirely of necrotic material, it may be difficult to decide whether it represents caseous necrosis or tumor necrosis (see Figs 8.6). Sampling should be repeated if no bacilli, epithelioid cells or tumor cells are found on careful examination of the smears.
Sometimes, only a few epithelioid cells are found in small groups or as single cells or the histiocytes may not quite have the typical appearance of epithelioid cells. The pattern then approaches that of non-specific, reactive lymphadenitis with prominent histiocytes. This may be the case in toxoplasma lymphadenitis and in the early stages of sarcoidosis. Endothelial cells can sometimes also be mistaken for epithelioid histiocytes. One such example is Kaposi’s sarcoma in which tissue fragments may resemble a granuloma, but the nuclei are more elongated and spindly and usually are hyperchromatic. They lack the ‘sand shoe’ indentations of epithelioid histiocytes and multinucleated giant cells are not seen (Fig. 5.20) (See also Chapter 18).125-127 In Kaposi’s sarcoma the cells are HHV-8, CD31, CD34 and D2-40 positive.
Clusters of epithelioid cells are sometimes found in cases of malignant lymphoma, particularly in HL and in peripheral T-cell (Lennert’s) lymphoma. They can also occur in metastatic seminoma and in lymph nodes regional to a carcinoma. A florid granulomatous reaction may occasionally be seen in NHL (e.g. Burkitt and lymphoplasmacytic lymphomas) and this may impair diagnosis. At times, an exuberant granulomatous reaction may also obscure an underlying metastatic carcinoma. One must therefore look carefully for abnormal lymphoid cells and for nonlymphoid cells in smears containing epithelioid histiocytes. Full knowledge of the clinical presentation is obviously essential.
Focal, extensive or total necrosis/infarction of lymph nodes occurs in some inflammatory processes (e.g. viral and bacterial infections), in metastatic malignancy, in malignant lymphoma and rarely in relation to vasculitis (e.g. Kawasaki’s disease) and to trauma. If necrosis is extensive, FNB smears may not include any well-preserved cells necessary for diagnosis.
Amorphous, granular material without identifiable cell remnants suggests caseous necrosis and smears should be searched for acid-fast bacilli and other microorganisms. In acute inflammatory necrosis, the aspirate and smears have a purulent character.
Necrotising lymphadenitis (Kikuchi-Fujimoto disease) is a condition of unknown etiology, seen mainly in young women, in which there is focal necrosis with a proliferation of histiocytes, lymphocytes and plasmacytoid dendritic cells, usually in cervical lymph nodes.128-130 The presence of large mononuclear cells in such nodes may cause a suspicion of malignant lymphoma. In FNB smears, the characteristic findings are of large numbers of pale, phagocytosing histiocytes with eccentric, crescentic nuclei, debris with nuclear fragments, absence of neutrophils and a reactive background of lymphoid cells (Fig. 5.21). The histiocytic cells are CD68, CD163 and myeloperoxidase positive.
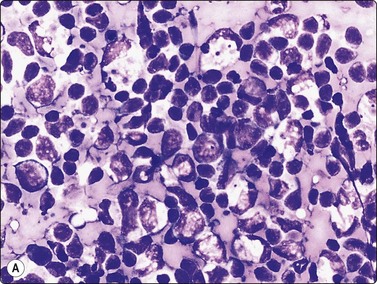
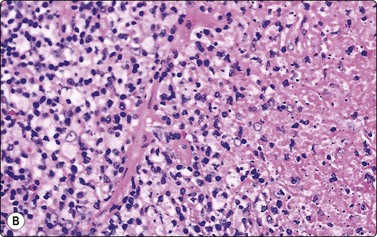
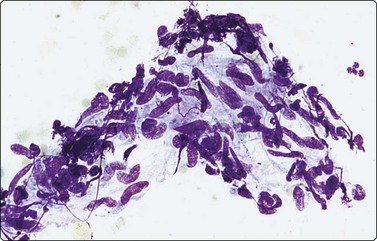
Fig. 5.21 Kikuchi-Fujimoto disease
Many large pale histiocytes with crescentic nuclei and phagocytosed debris with background of reactive lymphocytes; necrosis obvious in the tissue section (A, smear, MGG, HP; B, tissue section H&E, IP).
Necrosis in lymph nodes may also occur in systemic lupus erythematosus. In systemic lupus there may be significant numbers of plasma cells and/or hematoxylin bodies, which may help to distinguish it from Kikuchi-Fujimoto disease. If these features are absent, then serology would be necessary to make this distinction.131
Smears from areas of coagulation necrosis in lymph nodes show numerous cell shadows, some with preserved but pyknotic nuclei. Unless there is a clear history of trauma, such findings raise a strong suspicion of either metastatic carcinoma or malignant lymphoma. Nodal metastases of small cell anaplastic carcinoma of lung, melanoma and breast carcinoma are prone to necrosis and the necrotic cells with pyknotic nuclei can be indistinguishable from necrotic lymphoid cells. Extensive necrosis/infarction is common in malignant lymphoma, both non-Hodgkin and Hodgkin. Total infarction of a lymph node can sometimes precede manifest lymphoma. It has been shown that the demonstration of clonal immunoglobulin or T-cell gene rearrangement in the infarcted node can suggest a diagnosis of lymphoma.132
Metastatic malignancy is a more common etiology of peripheral lymphadenopathy than lymphoma, especially in patients over 40 years of age.
Micrometastases, either in the subcapsular sinus or as scattered single cells, are unlikely to be sampled even by repeated aspirations. Small metastatic deposits can be missed also by histological examination of an excised node. Partial lymph node involvement is the main cause of false-negative cytological reports.
Benign epithelial inclusions of salivary gland or thyroid origin have been observed in cervical nodes, inclusions of Müllerian origin in pelvic nodes. Mediastinal nodes rarely have mesothelial inclusions while neval cells may be found in axillary nodes. Such inclusions are usually very small. Although a rare occurrence, this possibility should be kept in mind when only a few epithelial cells without obvious malignant characteristics are found in lymph node aspirates in the appropriate context. Groups of glandular epithelial cells of apocrine type are commonly found in FNB smears from axillary nodes. The presence of such cells could give rise to a suspicion of metastasis from breast cancer if the benign characteristics of the cells are not appreciated. The cells most likely represent contaminants from adjacent sweat glands (see Chapter 7).
The common problem of differentiating between necrotising granulomatous lymphadenitis and necrotic tumor has already been mentioned. Squamous cell carcinoma is particularly prone to undergo liquefactive necrosis. An aspirate from such a node consists of thin, mucoid, yellow fluid. Well-preserved neoplastic squamous cells may be few in number and are often very well differentiated (see Fig. 4.2). There is therefore a risk of mistaking a cystic metastasis of well-differentiated squamous carcinoma in a cervical gland for a branchial cyst (see Chapter 4).83,133 Cystic nodes in the neck may also represent metastases of papillary carcinoma of the thyroid. This possibility should be remembered, particularly in cases of unexplained cervical lymphadenopathy in a young patient (see Chapter 6).
Follicular lymphoma (grade 1–2) may mimic metastatic small cell anaplastic carcinoma or Merkel cell carcinoma in FNB smears.134,135 This is because of the tendency for neoplastic cells of centrocytic type to form cohesive clusters, sometimes with nuclear molding. This problem will be further discussed in the section on lymphoma as will the differentiation of large cell lymphomas (centroblastic, immunoblastic) from other large cell malignancies by immunocytochemistry12,105 and pseudoepithelial clustering of lymphoid cells.
Indicators of the primary site
A stepwise approach to the investigation of nodal metastasis is suggested. This should include:
The patient’s age is an important consideration when assessing metastatic disease as certain tumors are more common in younger age groups, for example the small round blue cell group of tumors and nasopharyngeal carcinoma.
The anatomical site of the node may give some indication to the possible location of the primary tumor. For example, the axilla commonly harbors metastatic deposits from the breast, lungs or ovaries in middle-aged females. Nodes in the left supraclavicular fossa may be the site of presentation of pelvic malignancies, for example from the prostate, testis or ovaries as well as abdominal malignancies from the gastrointestinal tract.
The cytological patterns seen in routinely stained smears often give clues to the site of the primary tumor. Columnar cells with elongated nuclei arranged in palisades, stringy mucus and necrosis suggest a primary in the large bowel (see Fig 10.32), while mucin-containing signet ring cells suggest the stomach as the most likely primary site among several other possibilities. Glandular cells, moderately pleomorphic, arranged in a gland-in-gland or a cribriform pattern suggest prostatic carcinoma (Fig. 5.22). Large cells with abundant pale, granular or finely vacuolated cytoplasm and a low N : C ratio suggest a renal cell carcinoma. Very large central nucleoli are typical of less well-differentiated forms of this tumor and are also seen in large cell anaplastic carcinoma of lung and nasopharynx (see Fig. 5.55) and in hepatocellular carcinoma. Pulmonary and pancreatic adenocarcinoma can have a variety of patterns. They usually show a moderate degree of glandular differentiation, prominent nuclear pleomorphism and obvious mucin secretion. As a rule, the presence of intracytoplasmic mucin excludes renal, adrenal, hepatocellular and thyroid carcinoma. Breast cancer usually displays poor glandular differentiation while cell balls and single files of cells are more common. Nuclear pleomorphism is often relatively mild. Cells with intracytoplasmic neolumina containing ‘bull’s-eye’ inclusions are also suggestive of breast carcinoma.
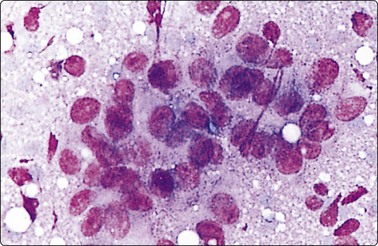
Supraclavicular node aspirate containing malignant cells from a metastatic adenocarcinoma (MGG, HP).
The nuclei of a small cell anaplastic carcinoma of lung may appear large in air-dried smears but the amount of cytoplasm is minimal. Nucleoli are very small and indistinct in typical cases. The cells are closely packed together in aggregates or as single files with prominent nuclear molding. Pyknotic nuclei and nuclear debris are commonly seen between preserved cells (see Fig. 5.2), in the absence of massive necrosis. ‘Tear-drop’ nuclear artifacts caused by smearing are characteristic (see Figs 8.25 and 8.27). Merkel cell carcinoma (neuroendocrine carcinoma of skin) is characterized by mainly solitary undifferentiated small cells mimicking NHL. However, lymphoid globules are absent while CAM 5.2 and cytokeratin 20 staining is positive with a characteristic dot-like distribution (Fig. 5.23). Neuroendocrine markers (synaptophysin, chromogranin, CD56) are often positive. Features suggesting a neuroendocrine primary are: dispersal, ‘neuroendocrine anisonucleosis’, bland chromatin that may be speckled, cells in curved rows and poorly formed follicles (see Chapters 8 and 10).
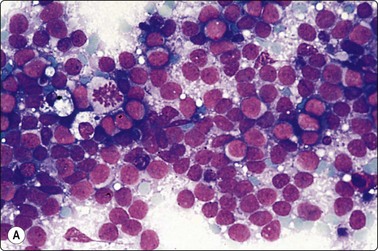
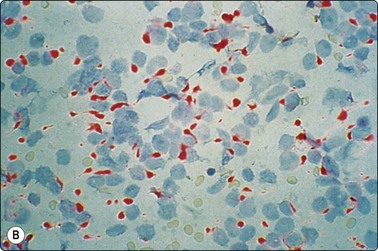
Fig. 5.23 Merkel cell carcinoma
(A) FNB lymph node with predominantly dissociated undifferentiated tumor cells, superficially resembling NHL. However, lymphoid globules absent and lymphoid markers negative (MGG, HP). (B) Immunostaining of same case showing characteristic dot-like positivity of Merkel cell carcinoma with CAM 5.2 (HP).
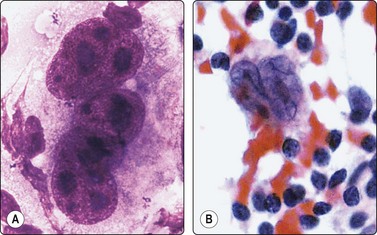
Malignant melanoma is the great mimicker. Smears usually show total dissociation of cells, well-defined cytoplasm, eccentric nuclei, prominent anisokaryosis, a uniformly dense chromatin which does not vary much from nucleus to nucleus, often large nucleoli, binucleate cells, intranuclear vacuoles and often some cells with intracytoplasmic pigment (see Chapter 14). Malignant cells with abundant cytoplasm and a dark-staining paranuclear area are a clue in amelanotic melanoma. It can occasionally mimic lymphoma in smears (see Fig. 5.60), carcinoma when cellular groups are present, sarcomas with prominent numbers of spindle cells and even pleomorphic malignancies. Testicular tumors may be clinically occult and present with metastases to pelvic, para-aortic or supraclavicular nodes. The cytological pattern of seminoma is characteristic. The tumor cells are mainly dissociated and are mixed with lymphocytes and often with epithelioid cells. They have large, rounded, vesicular nuclei and an evenly distributed nuclear chromatin. Nucleoli are prominent but not very large in wet-fixed smears. The cytoplasm is pale and both cytoplasm and nuclei are very fragile, the dispersed cytoplasm forming a ‘tigroid’ background to the nuclei (Fig. 5.24, see also Fig. 5.27).9,136 A tigroid background can, on occasions, also be seen in other glycogen-rich tumors: clear cell sarcoma, clear cell adenocarcinoma of the cervix and clear cell squamous carcinoma. Cells from a transitional cell carcinoma (TCC) may also be dispersed, resembling a large cell lymphoma (Fig. 5.25), or may form solid and sometimes papillary groups. The cells have abundant, relatively dense cytoplasm with distinct borders and pleomorphic, often eccentric, nuclei. A tendency to squamous differentiation or a spindle cell pattern (cercariform cells) may be seen, whereas papillary structures are uncommon in metastatic TCC.
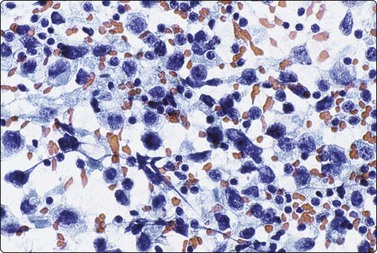
Fig. 5.24 Lymph node metastasis of testicular seminoma
Smear from enlarged left supraclavicular lymph node. Mainly dispersed population of malignant cells with fragile nuclei, prominent nucleoli, and dispersed cytoplasm, intermingled with many lymphocytes (Pap, HP).
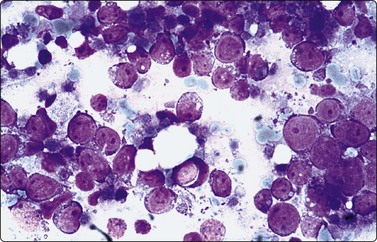
Fig. 5.25 Transitional carcinoma mimicking lymphoma
Dispersed malignant cells with large rounded nuclei, prominent nucleoli and scant blue cytoplasm. Irregular cytoplasmic fragments represent tumor necrosis, not lymphoid globules (MGG, HP).
Cytochemical stains and immune markers are often helpful. Squamous differentiation is most obvious in alcohol-fixed Pap-stained smears but can also be distinguished in MGG-stained preparations (Fig. 5.26A). Histochemical demonstration of intracytoplasmic mucin droplets confirms a diagnosis of adenocarcinoma. Positive immunocytochemical staining for prostatic acid phosphatase and/or prostate-specific antigen in an adenocarcinoma supports a prostatic origin.137 This staining should be performed in all metastatic adenocarcinomas of unknown origin in male patients in view of the palliative treatment available for disseminated prostatic carcinoma.
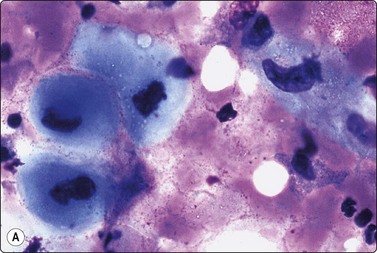
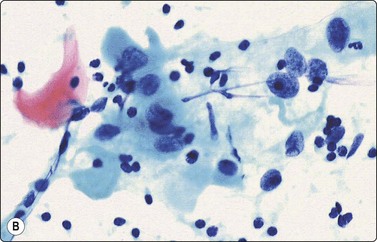
Fig. 5.26 Squamous cell carcinoma
(A) Characteristic clear blue cytoplasmic staining, indicating squamous differentiation (MGG, HP). (B) Abundant dense cytoplasm and an occasional orangeophilic keratinised cell (Pap, HP).
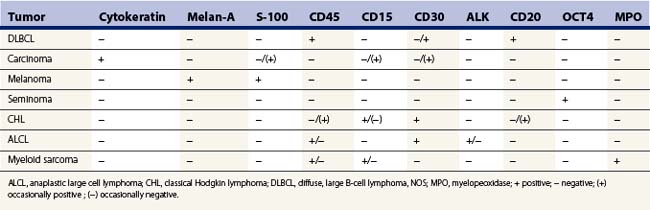
Immunocytochemical staining for S-100, Melan A and HMB-45 is of great value in diagnosing melanoma. Immune markers with some ‘relative tumor specificity’ can be very helpful, e.g. PSA (prostate), TTF-1 (lung and thyroid), GCDFP-15 (breast and sweat gland, salivary gland) and PAX-2 (renal and mullerian). Other useful tumor markers are chromogranin, CD56 and synaptophysin in neuroendocrine tumors, a variety of hormones and polypeptides in endocrine tumors, glypican-3, Hep-Par1 and alpha-fetoprotein in hepatocellular carcinoma. Similarly, OCT4 and placental alkaline phosphatase (PLAP) are useful in some germ cell tumors, with CD117 in seminoma. Also, calcitonin and CEA in thyroid medullary carcinoma, and FLI-1 in primitive neuroectodermal tumor (see Table 2.4). CDX-2 is a colorectal carcinoma marker; however, it also reacts with some gastric and pancreatobiliary adenocarcinomas. Differential cytokeratins (CK7, CK20) may be helpful in suggesting the origin of metastatic carcinoma.138,139 In some cases, electron microscopical examination of the aspirate and cytogenetic studies are extremely helpful, particularly in small round cell tumors and in some mesenchymal tumors (see Table 2.3).
Metastatic sarcomas are uncommon findings in lymph nodes.140 Sarcomas other than Kaposi’s, which have a tendency to involve regional nodes, are rhabdomyosarcoma, epithelioid sarcoma, clear cell sarcoma, angiosarcoma, Ewing’s sarcoma and synovial sarcoma (Fig. 5.27)
Non-Hodgkin lymphoma9,10,11,38,41,54,55,58,60,68-70,84,141-145
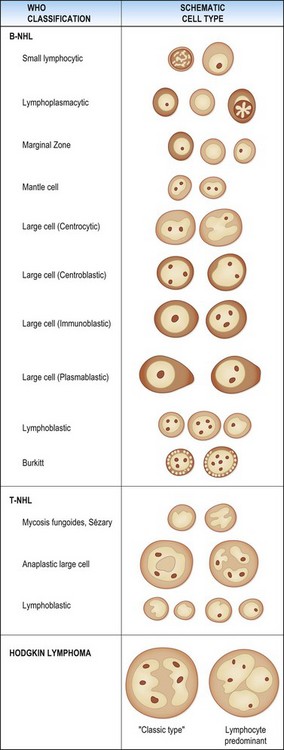
Today, most pathologists use the WHO classification of lymphomas (Table 5.1) which is based on a combination of cytomorphology, immunophenotype, genetic characteristics and clinical findings.69 Lymphoma cell types, including Hodgkin lymphoma, are diagramatically illustrated in Figure 5.28. This classification allowed a greater role for FNAC with FCM in lymphoma diagnosis and subclassification. Cytological subtyping of NHL in FNB smears can be difficult and needs extensive experience and ancillary support. It requires a complete comprehension of the WHO classification of lymphomas (and/or hematopathology experience), a working knowledge of FCM and immunoperoxidase use and interpretation as well as cytogenetic and molecular studies in order to make the appropriate correlation with clinical information so that a classification can be made. There should also be close collaboration with hematopathologists/hematologists and senior people in the molecular, FCM and genetic laboratories.
Table 5.1 Entities of WHO classification of lymphoid tumours
| B-cell lymphomas | T/NK-cell lymphoma |
FCM is the mainstay in the diagnosis and classification of NHL using FNB. FCM immunophenotyping provides quantitative evidence of cell lineage, B-cell clonality and T-cell aberrancies, and facilitates subclassification. FCM is not infallible and has pitfalls which may lead to misclassification or incorrect diagnosis. It is essential to correlate morphology with FCM data and the clinical situation.
The original 2001 WHO classification was revised in late 2008. It contains additions of new entities and subtypes (e.g. in the large cell lymphoma group), recognition of specific gray-zone areas and more degrees of acceptable uncertainty. More uncertainty has been recognized even within well-established entities, e.g. distinguishing lymphoplasmacytic lymphoma from other B-cell lymphomas with plasmacytic differentiation. These and other changes will add greater complexity to cytological subtyping of NHL. The number of possible diagnoses has more than doubled since the 2001 edition.
Over 90% of NHL are B-cell neoplasms, with diffuse large B-cell lymphoma and follicular lymphoma accounting for 65% of these. Less than 10% of NHL are T- and NK-cell neoplasms. As the B-cell lymphomas are commonly seen in daily practice, these will be highlighted, but not all of those listed in the WHO classification will be discussed. Some of the T-cell lymphomas also will be briefly discussed.
FNAC diagnosis of NHL is based largely on monomorphism of smear populations or disproportionate representation of cell types, often with features similar to their benign counterparts, which contrasts to most neoplasms diagnosed on the basis of cellular atypia. Assessment of cell size and nuclear features aid in subtyping. Small cells are less than 1.5 × the size of a normal lymphocyte. Medium sized cells are 1.5–2 × the size of a normal lymphocyte or no larger than a histiocyte nucleus. Large cells are more than 2–3 × the size of normal lymphocytes or larger than a histiocyte nucleus. The various cellular populations and cell size group entities are illustrated in Figure 5.29. The morphologic findings are then integrated with the immunophenotyping.
B-Cell neoplasms
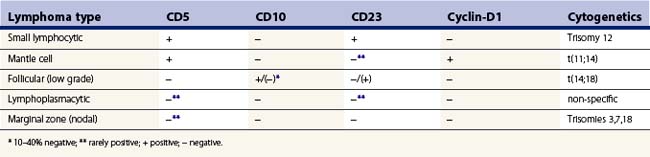
Small cell lymphoid proliferations may have morphologic similarities and at times may be difficult to differentiate from reactive nodes. FCM is essential in distinguishing reactive from neoplastic small cell lymphoid populations as it demonstrates monoclonality in the neoplastic B cells, and aids in subtyping by assessing CD5, CD10, CD23 and FMC7 expression on the neoplastic B cells. This distinction can be difficult based on morphology alone (Table 5.2). Cytogenetics can also aid in subtyping.
Small lymphocytic lymphoma/chronic lymphocytic leukaemia (SLL/CLL) (Figs 5.30-5.33)
Patients are mostly middle aged and elderly.
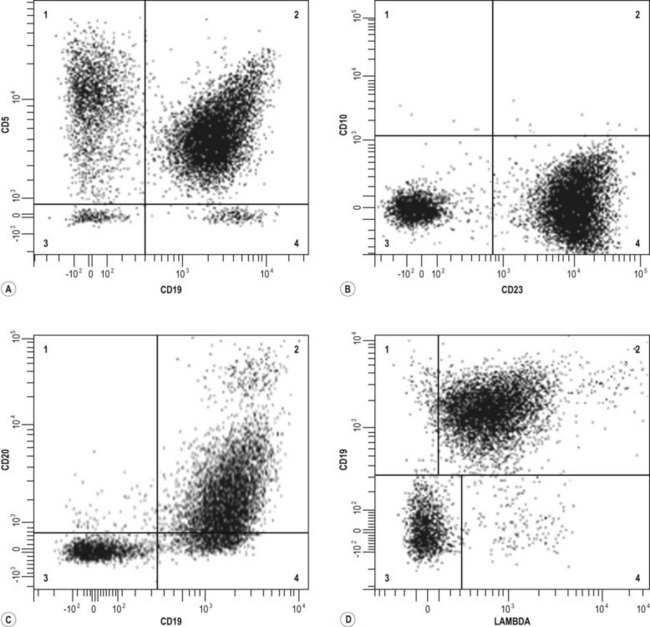
Fig. 5.30 Flow cytometric analysis of chronic lymphocytic leukemia
The upper left panel demonstrates CD5 and CD19 dual staining; note that quadrant 2 contains cells positive for both CD19 and CD5. Quadrant 1 contains the remaining CD5-positive T cells. The upper right panel demonstrates that the CLL cells are positive for CD23 (quadrant 4) and negative for CD10. The lower left panel demonstrates dual staining with CD19 and CD20. Note the typical weak CD20 staining on CLL cells. The lower right quadrant demonstrates the lambda light chain restriction, in that virtually all of the CD19-positive cells express (weak) lambda light chain.
(Courtesy A/Prof P. Macardle, Flinders Medical Center).
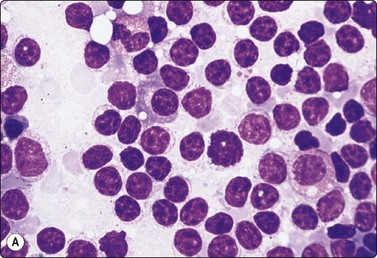
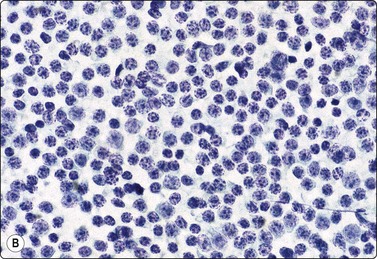
Fig. 5.31 Small lymphocytic lymphoma (B-CLL)
Monotonous population of slightly enlarged lymphocytes with coarsely granular chromatin (grumelé pattern) particularly obvious in Pap-stained smears; (A) MGG, HP; (B) Pap, HP.
Recognition of the subtle morphological differences between small lymphocytes and the small cells of lymphocytic lymphoma requires optimal cytological preparations. The typical SLL/CLL is readily recognized by the monotonous population of cells resembling small lymphocytes (Fig. 5.31). Difficulties can arise if the process contains numerous proliferation centers with many large and intermediate size cells – paraimmunoblasts and prolymphocytes (Fig. 5.32). A large B cell lymphoma may develop in 2–8% of patients with B-CLL, the so-called Richter syndrome (Fig. 5.33) or rarely HL.9,69,145
Lymphoplasmacytic lymphoma (LPL) (Fig. 5.34)
Most patients are elderly and the majority of cases have a serum monoclonal IgM paraprotein.
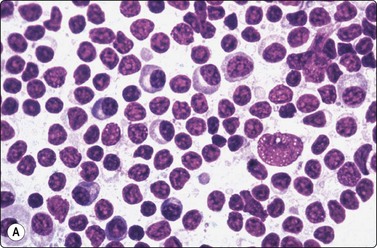
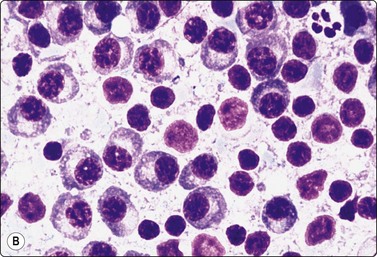
Fig. 5.34 Lymphoplasmacytic lymphoma
(A) Predominantly small lymphocytes, some with plasmacytoid cytoplasm, and a single large blastic cell (MGG, HP) (Reproduced with permission from van Heerde et al.9); (B) Lymphoplasmacytic lymphoma with obvious plasma cell component (MGG, HP).
Occasional cases have a large number of immunoblasts. This may indicate a worse prognosis. Transformation to a large cell lymphoma is rare.69 Other small cell lymphomas with plasmacytic features need to be excluded. At times marginal zone lymphoma may not be clearly separated from LPL.
Plasmacytoma (Figs 5.35, 5.36)9,69,146,147
Usually in middle aged or elderly patients. Commonly involve bones, upper airways and only rarely lymph nodes.9,71
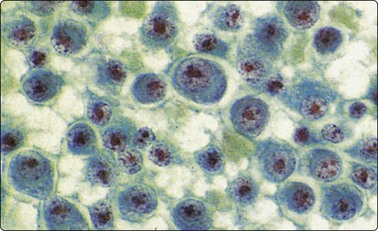
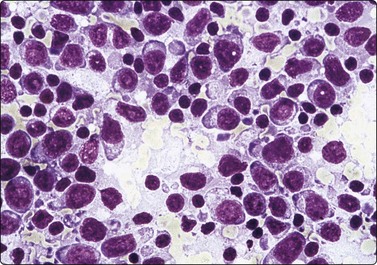
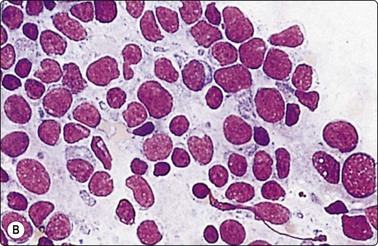
Fig. 5.36 Myeloma in lymph node
Poorly differentiated plasma cells. This patient had multiple myeloma (Pap, HP).
Be wary of unusual cellular morphology, e.g. occasional cases dominated by pleomorphic, signet ring, clear and sarcomatoid cells, which may be difficult to recognize as plasma cells. The rare variant with a small lymphocyte-like morphology may mimic a B-cell lymphoma.
Follicular lymphoma (FL) (Figs 5.37-5.39)69
Follicular lymphoma constitutes about 30% of NHL and is composed of a mixture of centrocytic and centroblastic cells. In the WHO classification, follicular lymphomas are divided into three histologic grades based on the number of large centroblasts present: grade 1 – predominantly centrocytes (0–5 centroblasts/HPF), grade 2 – a mixture of centrocytes and centroblasts, (6–15 centroblasts/HPF) and grade 3 – predominantly centroblasts (> 15 centroblasts/HPF).
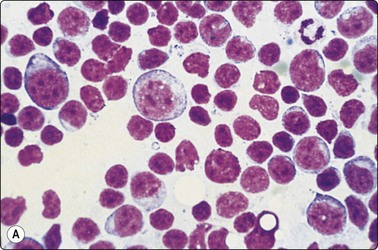
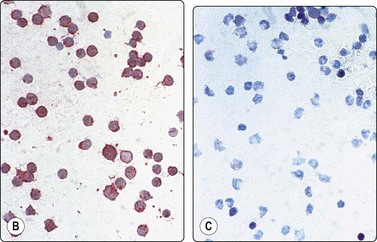
Fig. 5.37 Follicular (grade 1) lymphoma
(A) Predominance of medium-sized cells with irregular, sometimes cleaved nuclei; a few centroblasts with multiple nucleoli. Relatively few small lymphocytes; (MGG, HP). (B,C) Kappa/lambda staining in cytospin preparation showing lambda monoclonality (B) (immunostain, alkaline phosphatase method, IP).
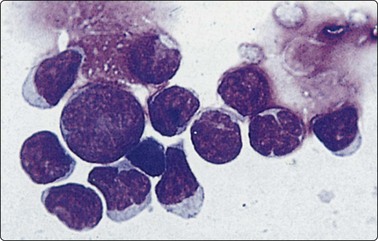
Fig. 5.38 Follicular (grade 1) lymphoma
Cytocentrifuge preparation; centrocytes showing cleaved nuclei; one centroblast (MGG, HP oil).
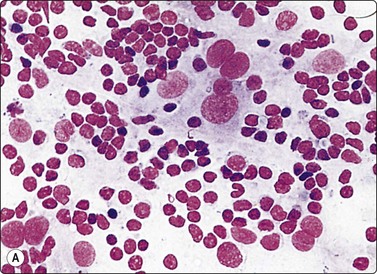
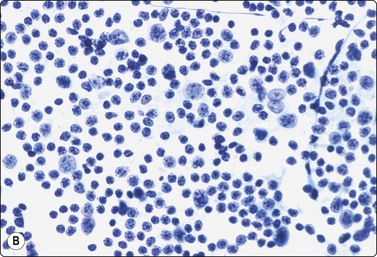
Fig. 5.39 Follicular (grade 1) lymphoma
Dendritic reticulum cells may be prominent in follicular lymphomas; predominance of centrocytes; (A, MGG, B, Pap, HP).
Criteria for diagnosis
It is not possible in FNB smears to reliably predict the architectural pattern of a follicular lymphoma (FL). However, some authors have greater success in identifying aggregates of follicle center cells.142 A follicular pattern in cell blocks or NCB can be inferred by staining of dendritic cells with CD21/35. It is also difficult to assess the relative proportions of small and large centrocytes and centroblasts, as these are influenced by sampling bias. This makes grading difficult. Also, a clear distinction between FL grade 3B and DLBCL (mainly centroblastic) is often not possible. However, this distinction may not be critical for therapeutic management.
There are no well-established criteria for grading FL in FNB smears. FL grade 3 has been variably defined,21,29,145 for example, greater than 20%, or 50% centroblasts in the smear population, or there is sheeting of large cells. Grading has also been based on centroblast count with an associated proliferative index or proliferative index alone.148,149 As a rule, this author favors FL grade 3 if half or more of the cell population, as assessed subjectively, are centroblasts.
In FL grade 1, the majority of cells are centrocytes. The characteristic cleaved nuclear morphology of centrocytes is well demonstrated in cytocentrifuge preparations of cell suspensions (Fig. 5.38). The irregular nuclear shape is often less obvious in direct smears and centrocytes are then identified by the intermediate size of the nuclei and by the nuclear chromatin, paler and more granular than that of lymphocytes. Starry-sky macrophages are usually absent. Dendritic reticulum cells are commonly seen (Fig. 5.39). FCM and/or immunohistochemistry are often necessary to distinguish follicular lymphoma from follicular hyperplasia in FNB smears. In 25–35% of FL there is transformation to a large cell lymphoma or high-grade Burkitt-resembling lymphoma. Rarely, they ‘transdifferentiate’ into various histiocytic and dendritic neoplasms.69
Mantle cell lymphoma (MCL) (Fig. 5.40)143,150,151
Usually in adults over 50 years of age
Criteria for diagnosis
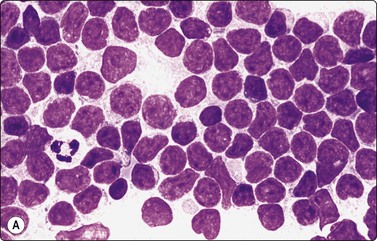
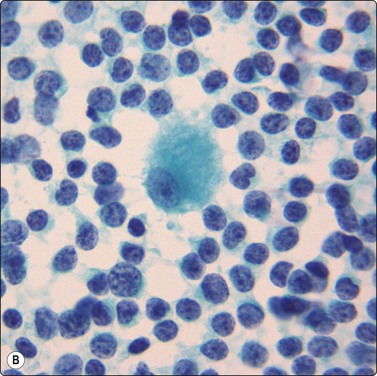
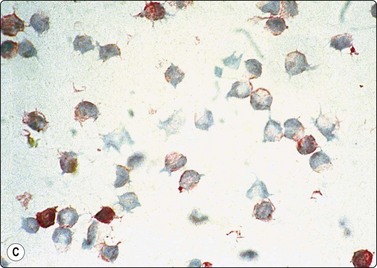
Fig. 5.40 Mantle cell lymphoma
(A) Nuclei a little larger than those of lymphocytes irregular shape, finely granular chromatin without distinct nucleoli. A few normal lymphocytes present. (B) Central histiocyte for size comparison (C) Immunostaining with CD5: strongly positive reactive T cells and moderate positivity in malignant lymphoma cells (A, MGG, HP; B,Pap HP; C immunostain CD5, IP).
Mantle cell lymphomas are generally clinically aggressive neoplasms, usually high stage at the time of presentation: generalized lymphadenopathy, splenomegaly, bone marrow and blood involvement. One must be wary of some uncommon variants (pleomorphic, small round cell, marginal zone-like) which may overlap with other lymphoma subtypes.69 The small cell variant appears to have a more indolent course. Immunostaining for Cyclin D1 on cytospin preparations can be unreliable.
Marginal zone lymphoma (MZL)
Marginal zone lymphoma accounts for approximately 10% of B-cell lymphomas. It has two major clinical presentations, extranodal (mucosal-associated lymphoid tissue) and nodal.
MZL is one of the most difficult types to recognize in smears as it can have morphologic features which overlap with a reactive process.25 It usually has a heterogeneous population and lacks specific cytologic and immunologic markers, apart from occasional cases having many monocytoid cells. In some of these latter cases, the cell population may be monotonous.69,152,153 The high content of reactive cells may impair detection of B-cell clonality.
Diffuse large B-cell lymphoma, not otherwise specified (DLBCL, NOS)69
Diffuse large B-cell lymphoma (DLBCL) is a heterogeneous group. Those that do not belong to specific subtypes or disease entities are classified as diffuse large B-cell lymphoma, not otherwise specified (NOS). These make up 25–30% of NHL. There are three common variants: centroblastic, immunoblastic and anaplastic (pleomorphic). Not infrequently, the distinction between centroblastic and immunoblastic lymphoma is not clear, on morphologic grounds, due to a mixed population of large cells of different type.
Criteria for diagnosis
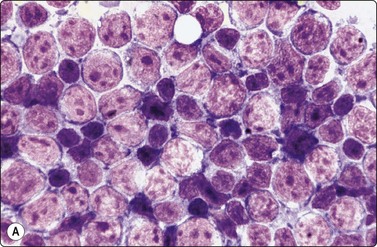
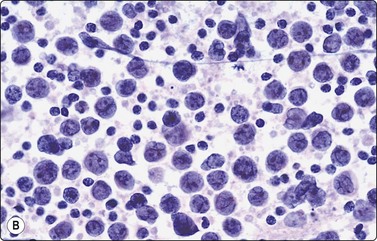
Fig. 5.41 Diffuse large B-cell lymphoma, NOS
Predominance of large centroblastic lymphoid cells with pale nuclei, scanty cytoplasm and multiple, often peripheral, nucleoli (A, MGG, B, Pap, HP).
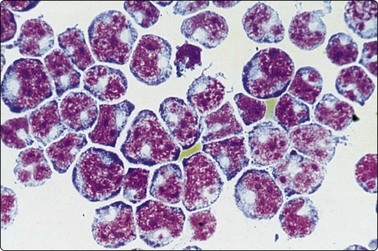
Fig. 5.42 Diffuse large B-cell lymphoma, NOS
Cytocentrifuge preparation of cerebrospinal fluid showing pronounced irregularity of nuclear contour (MGG, HP).
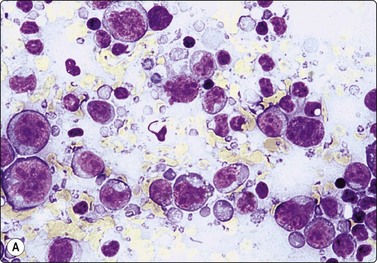
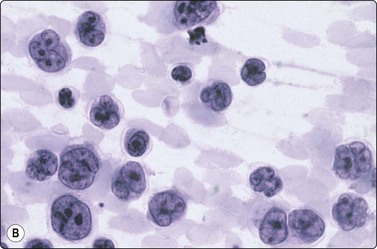
Fig. 5.43 Diffuse large B-cell lymphoma, NOS.
Polymorphous. Centroblasts, immunoblasts and large centrocytic cells, particularly distinct in Pap-stained preparations (A, MGG, HP; B, Pap, HP oil).
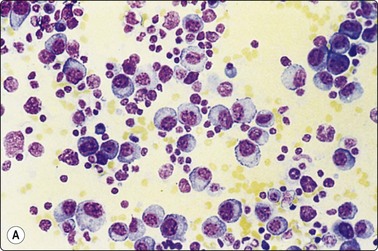
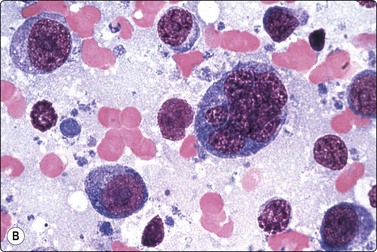
Fig. 5.44 Diffuse large cell lymphoma, NOS (immunoblastic B cells)
Predominantly large lymphoid cells with large round nuclei, large nucleoli and abundant basophilic cytoplasm (A, MGG, IP; B, MGG, HP oil).
(Reproduced with permission from van Heerde et al.9)
T-cell/histiocyte-rich large B-cell lymphoma (Fig. 5.46)
Affects mainly middle-aged men.
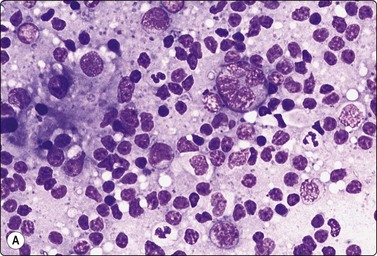
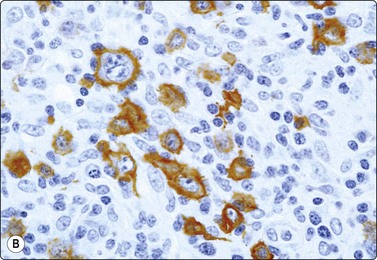
Fig. 5.46 T-cell/histiocyte rich B-cell lymphoma
(A) Scattered large abnormal lymphoid cells with a background of reactive lymphocytes (MGG, HP); (B) The neoplastic B cell highlighted by immunostaining for CD20 (immunostain CD20, HP).
Criteria for diagnosis
It is difficult to diagnose on FNB smears and to distinguish from HL and reactive adenopathy154-157 FCM is usually non-diagnostic. Usually a tissue biopsy with immune marker studies is essential to establish the diagnosis.
Primary mediastinal (thymic) large B-cell lymphoma (see chapter 9)
Plasmablastic lymphoma (Fig. 5.47)69,158
Plasmablastic lymphoma is uncommon and may be associated with immunodeficiency.
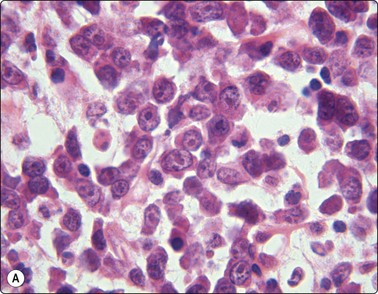
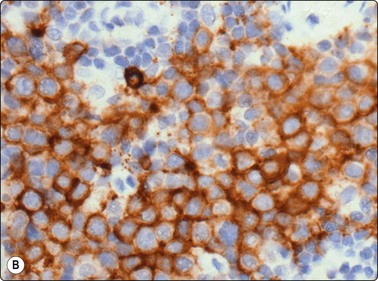
Fig. 5.47 Plasmablastic lymphoma
(A) Uniform large cells with vesicular nucleus, prominent nucleoli and moderate cytoplasm-resembling immunoblasts (cell button. H&E, HP); (B) CD138 positive (immunostain CD138, HP).
Criteria for diagnosis
It has an immunophenotype of plasma cells and needs to be distinguished from myeloma, principally on clinical and radiological findings.
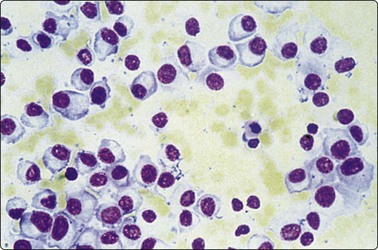
Burkitt lymphoma (Figs 5.48-5.50)159-161
Burkitt lymphoma in African children is endemic and associated with Epstein-Barr virus. The jaws often being involved. In non-African cases, most patients present with abdominal localization.
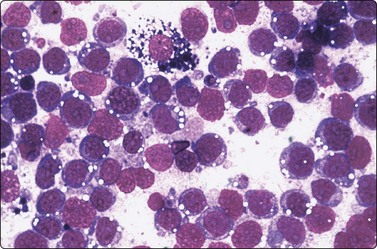
Rounded lymphoid cells; some variation in nuclear size; distinct nucleoli; granular chromatin; dense blue cytoplasm with lipid vacuoles and some starry-sky macrophages (MGG, HP).
(Reproduced with permission from van Heerde et al.9)
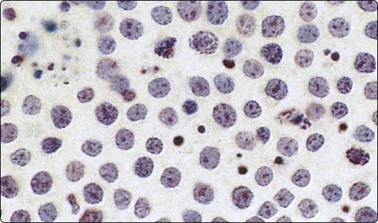
Blastic cells with round nuclei of varying size and small amount of cytoplasm; occasional starry-sky macrophages (Pap, HP).
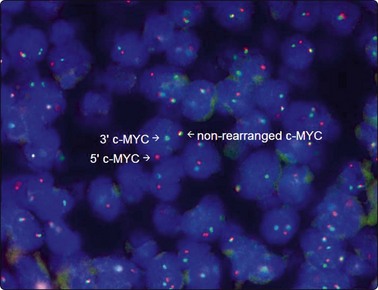
Dual-color FISH with break-apart c-MYC probes ( 5’c-MYC probe orange and 3’c-MYC probe green) showing nuclei in which one pair of probe signals (yellow) is split apart due to a c-MYC region rearrangement.
(Courtesy Sarah Moore, Institute of Medical and Veterinary Science).
Criteria for diagnosis
The diagnosis of Burkitt lymphoma requires a combination of ancillary techniques as there is no single diagnostic gold standard, e.g. the c-myc rearrangement is highly characteristic but not specific.69 In addition, there may be cases with greater nuclear pleomorphism with fewer and more prominent nucleoli.
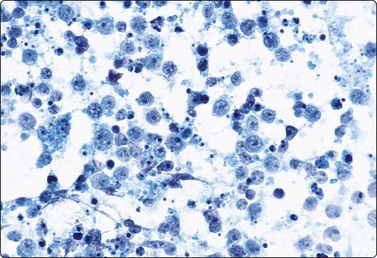
B-lymphoblastic leukemia/ lymphoma (Fig. 5.51)9,69,145,162
The majority of patients are under 18 years of age and the lymphoma often involves nodes, skin and bones. It comprises 10% of lymphoblastic lymphomas.
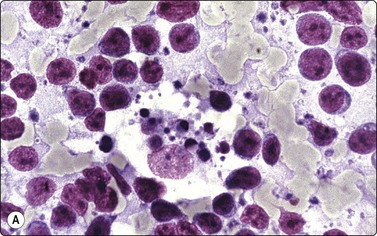
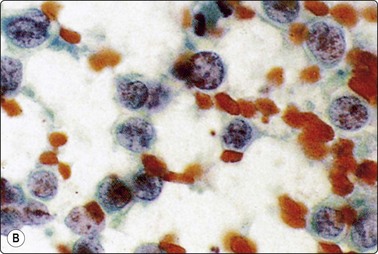
Fig. 5.51 B-lymphoblastic lymphoma
Rounded nuclei of variable, mainly intermediate size; speckled nuclear chromatin; small but distinct nucleoli. Starry sky macrophages may be present (A, MGG, HP; B, Pap, HP).
This lymphoblastic morphology may be seen with T-cell lymphoblastic lymphomas
T-lymphoblastic leukemia/lymphoma (Figs 5.52, 5.53)
The lymphoma is frequently associated with a mediastinal mass and it comprises 90% of lymphoblastic lymphomas.
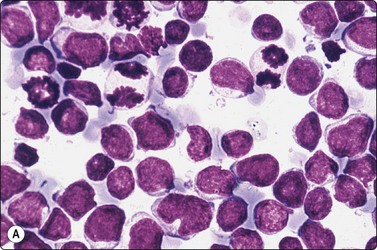
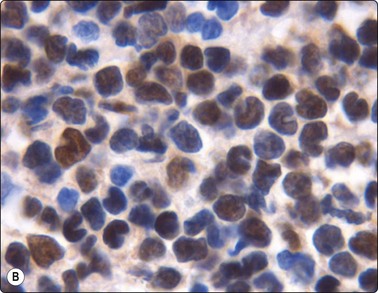
Fig. 5.52 T-lymphoblastic lymphoma
Note unipolar indentation (convolution) in some cells. Anisokaryosis, dense chromatin without distinct nucleoli, many mitoses. TdT positivity. (A, MGG, HP; B, immunostain,TdT, HP).
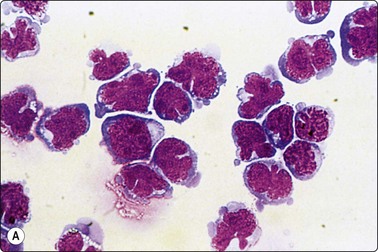
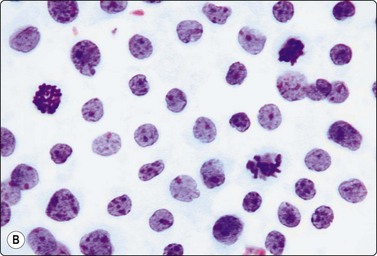
Fig. 5.53 T-lymphoblastic lymphoma
(A) Cerebrospinal fluid. In cytocentrifuge preparations the convolutions are very pronounced (MGG, HP oil); (B) Lymph node smear. Note mitotic figures (Pap, HP).
Criteria for diagnosis
The convoluted, lobulated or segmented (cerebriform) shape is most striking in cytocentrifuge preparations of cell suspensions (Fig. 5.53A). It seems to be related to the mode of preparation to some extent and appears to be exaggerated by centrifugation. Lymphoblasts with convoluted nuclei often have markers for T cells but a similar morphology can be seen with some B-cell lymphoblastic lymphomas. Also, a similar nuclear complexity can be seen in several B-cell lymphomas after treatment. The cytomorphology alone is thus insufficient to classify the lymphoma as T-cell type and needs to be supported by immune marker typing.
Mature T and NK-Cell neoplasms
These lymphomas are more difficult to diagnose on FNAC than B-cell lymphomas as they do not have a specific immunophenotype, apart from anaplastic large cell lymphoma, ALK positive. In addition, FCM does not clearly demonstrate monoclonality as seen with B-cell lymphomas. However, immunophenotyping is essential to recognize T-cell lineage and aberrant T-cell phenotype, which is strong presumptive evidence of a monoclonal T-cell population. The loss of one or more of the pan-T-cell markers (CD2, CD3, CD5, CD7) is the most useful criterion in helping to make the diagnosis of T-cell neoplasia. Also suggestive are loss of both CD4 and CD8 or coexpression of both.12,163 PCR for rearrangement of the T-cell receptor gene is often necessary to demonstrate clonality. Frequently tissue biopsy is necessary for initial diagnosis.
The cytology of peripheral T-cell lymphomas (PTCL) not otherwise specified (NOS) and NK/T lymphomas has been reported.164-168 PTCL NOS have a varied morphologic appearance, commonly heterogeneous, with atypical medium to large lymphocytes (some Reed Sternberg-like cells (see Fig. 5.71) and an exuberant benign host inflammatory reaction, producing difficulty in its separation from reactive lymphadenopathy. Occasionally, there is a monomorphous proliferation of small, medium or large atypical cells. The nuclei are often indented, grooved or knobbly.
Angioimmunoblastic T-cell lymphoma (Fig. 5.54 and see Fig. 5.70A)69,169,170
Typically seen with generalized lymphadenopathy, systemic symptoms and peripheral hypergammaglobulinaemia.
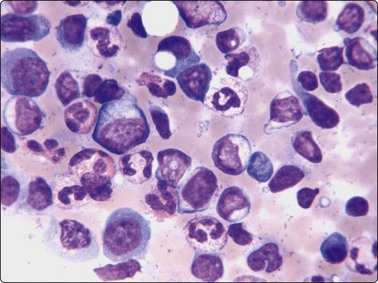
Fig. 5.54 Angioimmunoblastic lymphoma
Heterogeneous population of cells with small, medium and large lymphocytes, neutrophils, eosinophils and plasmacytoid cells (Diff-Quik, HP).
Criteria for diagnosis
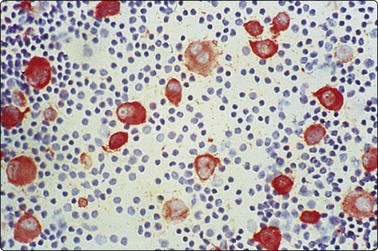
Anaplastic large cell lymphoma (ALCL), ALK positive (Fig. 5.55)69,85,86,171-173
Most frequent in childhood and young adults, presenting with lymphadenopathy.
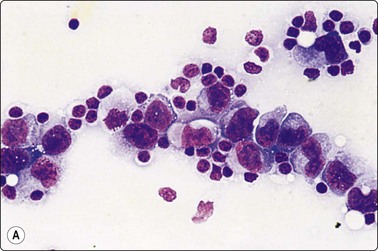
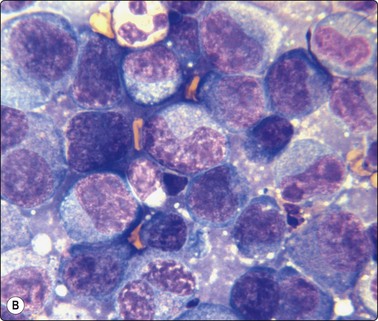
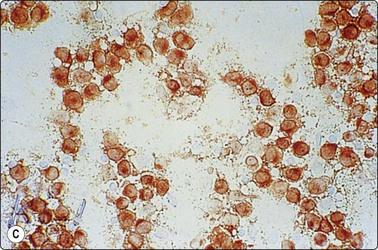
Fig. 5.55 Anaplastic large cell lymphoma
(A), Large polymorphous cells with abundant cytoplasm; pseudoepithelial arrangement; multinucleation; distinct nucleoli. (B), Occasional ‘hallmark’ cells with horseshoe/boomerang nuclear forms. (C), CD30 (Ki-1) positive (A, MGG, IP; B, Diff-Quik, oil HP, C, immunostain CD30, IP).
Criteria for diagnosis
Several cytomorphologic variants exist, e.g. small cell, Hodgkin-like, sarcomatoid and lymphohistiocytic.69 The ALK negative, anaplastic large cell lymphoma (ALCL) is seen in older adults and needs to be distinguished from HL (PAX 5 and cytotoxic markers being most helpful in making the distinction).
Problems and differential diagnosis (general)
Direct smears must be made expertly, since poor preparation makes accurate diagnosis impossible. Suboptimal smears are the commonest cause of diagnostic difficulties and misinterpretations.
Sampling bias cannot always be avoided and may lead to an erroneous diagnosis. An aspirate from a node which is only partially involved with lymphoma may not be representative. Due to several mechanisms, the number of centroblasts and centrocytes does not always reflect the real composition within the lymph node. Thus, malignancy grading on cytological grounds is not reliable in follicular lymphomas.
The difference between normal lymphocytes and the neoplastic cells of small lymphocytic lymphoma is relatively subtle and the most obvious diagnostic feature is the monotony of the cell population. However, a sample including proliferation centers does not appear monotonous since there can be many cells of intermediate and large size (prolymphocytes and paraimmunoblasts9,69 mixed with the typical cells of CLL type (see Figs 5.31 and 5.32). Such a case can be mistaken for reactive lymphadenopathy unless close attention is paid to fine cytological detail of the lymphocytes and to the result of immunocytology and FCM. Smears from small quiescent axillary and groin nodes may show a monotonous population of small lymphocytes. This can mimic small lymphocytic lymphoma, therefore this diagnosis should be avoided in small nodes unless there is immunologic proof of clonality.
Lymphoplasmacytic lymphoma (LPL) can also be mistaken for reactive lymphadenopathy in view of the sometimes heterogeneous character of the smear population. Again, a close study of cell detail usually reveals atypical lymphoid cells with plasmacytoid features. LPL is a diagnosis of exclusion, as other small cell lymphomas may have a mixture of relatively similar cells. At times, LPL cannot be separated from marginal zone lymphoma (MZL), as neither has a specific immunophenotype. MZL, with its mixed cell population, can mimic reactive hyperplasia. FCM, immunocytochemical, clinical and biochemical data are of utmost diagnostic importance.
In follicular lymphoma grade 1 there may be a high proportion of small lymphocytes to suggest a benign, reactive process. Also, the small centrocytes of some follicular lymphomas can be difficult to distinguish from lymphocytes in a reactive process, particularly if smears are not technically optimal. Nuclei must be studied carefully at high power to appreciate the slightly larger size, irregular shape and granular chromatin. Follicular lymphoma grade 1 with a predominance of small centrocytes is the type of lymphoma which is most likely to be missed cytologically and misinterpreted as reactive lymphadenopathy. FCM demonstrating B-cell clonality with CD10 positivity is important in making this distinction.
At times, separating T-cell lymphomas with a polymorphous cell population from reactive hyperplasia can be difficult. In the evaluation, it is important to have the clinical history and presentation and to correlate it with the ancillary studies. Even then, a tissue biopsy is usually necessary to make a firm diagnosis.
In some cases of large cell lymphoma, the neoplastic cells may be relatively few in numbers, and the smears may be dominated by a background of reactive lymphocytes (Fig. 5.56). T-cell/histiocyte rich B-cell lymphoma is one important example (see Fig. 5.46).155 Results of FCM may be difficult to interpret in such cases. Occasionally, distinction from reactive lymphadenopathy with scattered large, atypical-looking immunoblasts can be difficult.
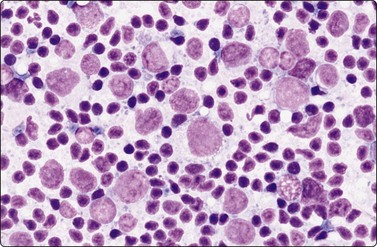
Fig. 5.56 Diffuse large cell lymphoma, NOS, with reactive lymphocytes
Scattered neoplastic cells with very large nuclei with a dominant background population of reactive lymphocytes (MGG, HP).
In FNB smears from follicular lymphoma grade 1 and 2, the large centrocytic cells may have a tendency to clump together into aggregates showing some moulding of the nuclei. Rows and palisades of closely apposed, ovoid nuclei which appear columnar through moulding may simulate small cell anaplastic carcinoma or even adenocarcinoma (Fig. 5.57). However, the proportion of isolated cells is usually larger in lymphoma and many of these have the typical appearance of lymphoid cells with a rim of basophilic cytoplasm and a nuclear chromatin pattern which is different from that of carcinoma cells. Importantly, nuclei of lymphoma cells of a similar size to those of small cell anaplastic carcinoma usually have prominent nucleoli. This is a helpful but not infallible feature: nuclei of large centrocytes and of small cell carcinoma of intermediate type may be very similar in size and may have similar nucleoli. Nuclear molding and well-formed single files of tumor cells are more obvious in small cell carcinoma and other metastatic small cell tumors. The pale-blue, uniformly rounded cytoplasmic fragments characteristic of lymphoid tissue (lymphoid globules) are different from the cytoplasmic and nuclear fragments of tumor necrosis in smears of carcinomas (compare Figs 5.1 and 5.2). Immunocytochemical staining for cytokeratin, EMA and CD45 can usually solve the problem of distinguishing between small cell carcinoma and lymphoma in difficult cases. Additionally, FCM can help as small cell carcinomas are CD 56 positive and CD 45 negative.
Fig. 5.57 Pseudoglandular clusters in lymphoma
Such clusters may simulate small cell anaplastic carcinoma or adenocarcinoma; this problem mainly occurs in follicular lymphomas and large cell anaplastic lymphomas (A and B, MGG, HP).
Burkitt and lymphoblastic lymphomas occur commonly in childhood and need to be differentiated from small round blue cell tumors of childhood via morphologic, immunologic and cytogenetic means (see Chapter 17).
Smears of undifferentiated carcinoma (Figs 5.58 and 5.59, and see Fig. 5.25) and melanoma (Fig. 5.60) may show total dissociation of the tumor cells, nuclei may be large, with fine chromatin, prominent nucleoli and abundant basophilic cytoplasm. This pattern can be indistinguishable from DLBCL (particularly immunoblastic rich type) or ALCL, and at times even HL. Also, a dissociated seminoma may similarly be difficult to distinguish from a lymphoma. Myeloid sarcomas may be mistaken for DLBCL. They have dissociated large cells with pale chromatin, prominent nucleoli and rarely cytoplasmic granules. The presence of lymphoid globules in lymphoma and lobulated nuclei is helpful. Well-formed, sharply delineated aggregates of tumor cells, not just clumping of cells, are not seen in lymphoma. Again, in difficult cases, immunological staining for cytokeratin, Melan-A, S-100, OCT4, myeloperoxidase and lymphoid associated markers is usually decisive (Table 5.3).85,86,171-174
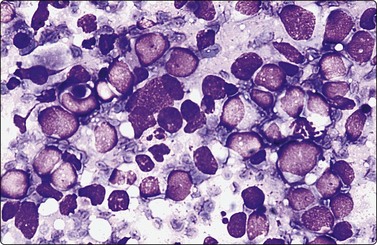
Fig. 5.58 Small cell anaplastic (neuroendocrine) carcinoma
This example of a FNB smear from a lung tumor closely mimics anaplastic large cell lymphoma: dispersed cells with large eccentric nuclei and a thin rim of blue cytoplasm (MGG, HP).
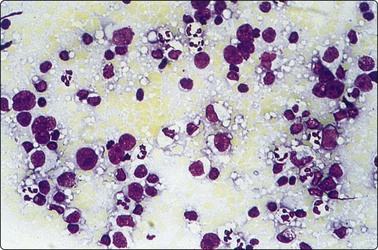
Fig. 5.59 Nasopharyngeal carcinoma
Metastatic large cell undifferentiated carcinoma with solitary tumor cells may mimic large cell lymphoma (MGG, IP).
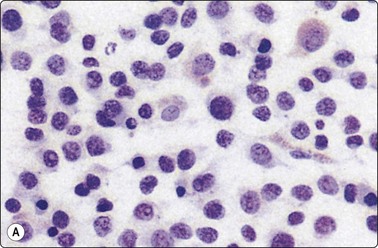
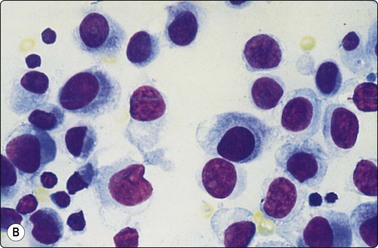
Fig. 5.60 Metastatic melanoma mimicking lymphoma
(A) Dispersed malignant cells with eccentric nuclei and pale cytoplasm; lymphoid globules absent (Pap, HP). (B) Amelanotic melanoma showing dense smooth chromatin and characteristic paranuclear dark area in tumor cells (MGG, HP).
FCM results may be unreliable or noninformative if the aspirated material is necrotic or heavily admixed with blood. It must be remembered that B-cell clonality does not always equate with malignancy and its absence does not necessarily indicate a benign process. Polyclonality on flow cytometry may be seen in T-cell lymphoma, HL, partial involvement of a node by lymphoma, absence of immunoglobulin expression in some DLBCL NOS, occasionally marginal zone lymphoma as well as in a reactive lymph node.10 Close correlation of FNAC, immunologic and clinical findings is always essential. One must also be aware of aberrant immunophenotypes in B-cell NHL (see NHL overveiw) and also false-positive B-cell clones. An unusual, infrequent immunophenotype is the B-cell lymphoma coexpressing CD5 and CD10, such as follicular lymphoma, mantle cell lymphoma and DLBCL, NOS.170 Rarely, with follicular hyperplasia in children and young adults there may be clonality.175,176 The author has also seen similar false B-cell clonality in lymph nodes with metastatic melanoma and adenocarcinoma. Infrequently, in progressive transfomation of germinal centers, there may be coexpression of CD4 and CD8, causing confusion with a T-cell neoplasm.
PCR false-negative results may occur with follicular, marginal zone and DLBCL NOS.177,178 In contrast, small lymphocytic lymphoma and mantle-cell lymphoma have a much lower rate of false-negative clonal detection.179,180 False-positive results may occur with some benign conditions, for example autoimmune diseases and some infectious processes.10
PTLD are more often extranodal in presentation, and there is a spectrum ranging from plasma cell hyperplasia/infectious mononucleosis-like reaction to diffuse lymphoma (predominantly B cell), however T/NK-cell and Hodgkin lymphomas also occur. FNB may provide a rapid diagnosis of monomorphic PTLD as it usually resembles B-cell neoplasms: DLBCL, Burkitt lymphoma and plasma cell neoplasms.69,181-183 In polymorphic PTLD there is a mixture of small and large lymphocytes with plasma cells, which may resemble a lymphoma with plasmacytic differentiation or infectious mononucleosis. In this category, the diagnosis requires flow cytometry, PCR and usually histologic assessment.181
There are two main groups in the gray-zone (unclassifiable) lymphomas. Firstly, cases from a morphological and phenotypical perspective resemble Burkitt lymphoma; however, some feature(s) prevent the diagnosis. Secondly, cases may morphologically resemble DLBCL; however, the phenotype is that of HL or vice versa. These cases cause difficulty in subtyping with histologic specimens, and equally cause problems with cytologic samples.
Hodgkin lymphoma (HL) (Figs 5.61-5.68)40,75-77,184,185
Hodgkin lymphoma accounts for approximately 30% of all lymphomas.69 It is comprised of two entities: nodular lymphocyte predominant HL and classic HL. The subtypes of classic HL are nodular sclerosis, mixed cellularity, lymphocyte-rich and lymphocyte depleted. These account for up to 95% of cases of HL.
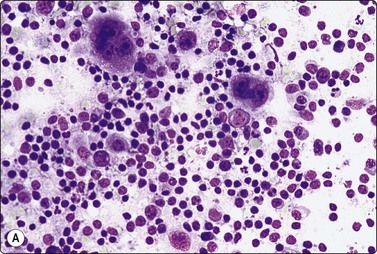
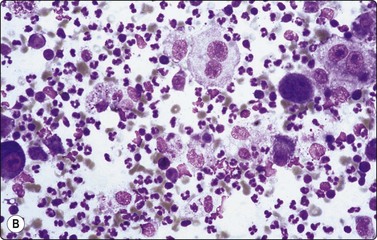
Binuclear Reed-Sternberg cells and mononuclear Hodgkin cells in a background of mainly small lymphocytes (A) and/or granulocytes (B). Varying basophilia (A and B, MGG, IP).
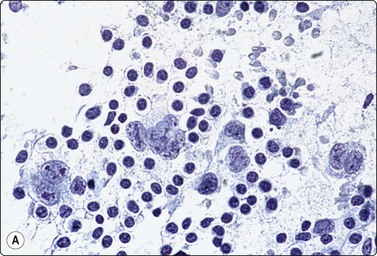
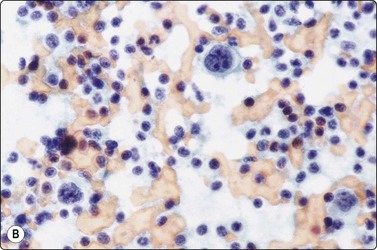
Many scattered binuclear or multilobated Reed-Sternberg cells with a background of lymphocytes (A and B Pap, HP).
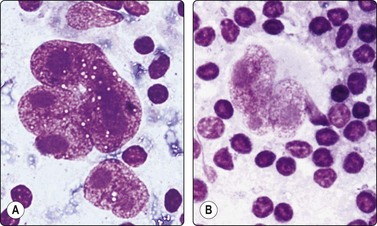
Examples of Reed-Sternberg cells from different cases; multiple or multilobated large nuclei with very large nucleoli; reticulate chromatin; pale cytoplasm (A and B, MGG, HP).
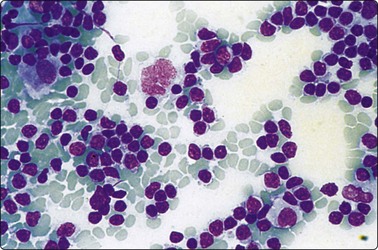
Fig. 5.67 Lymphocyte predominant Hodgkin lymphoma
Multinucleated ‘popcorn’ cell in a background of small B lymphocytes and some histiocytes (MGG, IP).
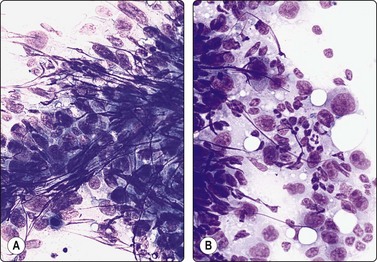
Fig. 5.68 Syncytial variant of Hodgkin lymphoma
Clusters of numerous large atypical lymphoid cells dominate smears focally; some are of Reed-Sternberg type (A and B, MGG, HP).
Classic HL, in nearly all cases, is derived from B cells. In nodular lymphocyte predominant HL, the Reed-Sternberg (‘popcorn’) cells are B cells reacting with CD20, OCT-2, BOB.1 and CD79a, and the small background lymphocytes are predominantly B cells but there are also many T cells. In classic HL the background lymphocytes are usually predominantly T cells, except in the nodular lymphocyte-rich variant where they are usually predominantly B cells. The Reed-Sternberg cells in these classic variants are often but not always negative for B-cell markers CD20 and CD79a. When positive for these markers, only a minority of these cells usually stains, and the staining intensity is varied. They are also weakly positive for PAX5.69
Criteria for diagnosis
A confident diagnosis of classic HL can only be made in the presence of typical Reed-Sternberg cells with a background of lymphocytes and reactive cells, as listed above, with its typical immunophenotype (Figs 5.61-5.65). Reed-Sternberg cells have large, lobulated nuclei which may appear symmetrically double (mirror nuclei) or complex and multiple. The nuclear chromatin is coarse and irregularly distributed in a reticular fashion with clear areas in between which give the nucleus an overall pale appearance. Nucleoli are large, often huge, eosinophilic in H&E preparations, pale grayish to dark magenta in MGG-stained smears. The cytoplasm is abundant, but pale and fragile so that the nucleus often appears to be surrounded by an empty space (see Fig. 5.64). Sometimes the characteristic nucleoli are not well demonstrated. Frequently only mononuclear cells which have a nuclear structure similar to the typical Reed-Sternberg cells (mononuclear Hodgkin cells, Fig. 5.66) along with their bare nuclear forms are present. In such cases, the definitive diagnosis must await histological examination, except in recurrent disease when classic Reed-Sternberg cells are not essential for diagnosis. The background cell population is mainly lymphocytes of reactive type, but polymorphs may be predominant in some cases (see Fig. 5.61B). The term ‘suppurative variant of HL’ has been proposed.185
Histological examination is also necessary to distinguish confidently the five subtypes of HL. The two extremes, lymphocyte predominant and lymphocyte depleted, are, of course, reflected in the smears by the number of lymphocytes relative to the number of Reed-Sternberg and mononuclear Hodgkin cells. A suggestion of the presence of the lacunar type of Reed-Sternberg cell, characteristic of nodular sclerosis Hodgkin lymphoma in histological sections, is occasionally present in FNB smears. The tough consistency of the node felt with the needle, a scanty aspirate and the presence of fibroblasts and collagen fragments in the smear are features suggestive of the nodular sclerosis type. In the mixed cellularity type, typical Reed-Sternberg cells are usually easy to find and there are many plasma cells and eosinophils as well as histiocytes in the background cell population.186
The nodular lymphocyte-predominant subtype is characterized by a monotonous population of slightly irregular small lymphocytes with relatively few scattered large pale multinucleated giant cells, corresponding to the ‘popcorn’ cells in histology, usually without distinct nucleoli (Fig. 5.67). In contrast to the other (classic) variants of HL, the bulk of the small cells and also the giant cells exhibit a B-cell phenotype.9,69 This is a difficult diagnosis to make on FNB smears as there are usually few atypical cells in a sea of reactive small lymphoid cells and the architecture cannot be assessed.
Poor biopsy yield is a problem mainly in the nodular sclerosis subtype. Not infrequently, smears show only a few lymphocytes, fibroblasts and fragments of collagen, which may suggest a chronic inflammatory process. Multiple biopsies or the use of a cutting core needle may be necessary to obtain sufficient material.
Large, multilobated nuclei resembling Reed-Sternberg cells can be seen in a variety of conditions (Figs 5.69-5.74). Atypical immunoblasts in non-neoplastic reactive lymphadenopathy, for example in mononucleosis (Fig. 5.69), toxoplasmosis (Fig. 5.70B), rheumatoid arthritis-associated and drug-induced lymphadenopathy, may have nuclei of this type. They usually differ from the typical Reed-Sternberg cells by having smaller and darker nucleoli, a denser chromatin and a basophilic cytoplasm. Multinucleated giant cells in small or large cell non-Hodgkin lymphoma, e.g. lymphoplasmacytic lymphoma, PTCL NOS (Fig. 5.71) and especially ALCL (Fig. 5.72), can also have large nucleoli similar to Reed-Sternberg cells.9,187 The distinction is particularly difficult in the presence of eosinophils, plasma cells and epithelioid cells, which is not unusual in T-cell lymphoma. Immunological studies may be necessary to solve the problem. There are some examples of large malignant cells with very large nucleoli and with many eosinophils and reactive lymphoid cells in the background, representing single malignant cells of metastatic – especially nasopharyngeal – carcinoma (Fig. 5.73), which were misdiagnosed as HL. In addition, megakaryocytes can be mistaken for Reed-Sternberg cells, e.g. in nodal myeloid metaplasia/extramedullary hematopoiesis. Also rarely with follicular dendritic cell sarcoma, Reed-Sternberg-like cells may also be seen in an inflammatory background (Fig. 5.74).
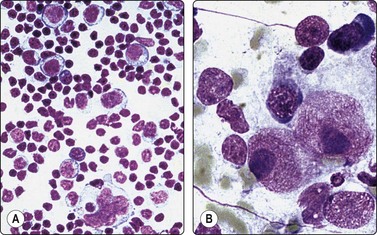
Fig. 5.69 Reed-Sternberg-like cells in infectious mononucleosis
(A) Multilobated giant cell (MGG, IP); (B) Binucleate cell with very large nucleoli (MGG, HP), from two different cases of infectious mononucleosis.
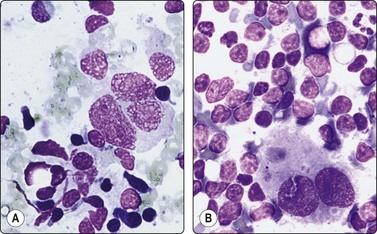
Fig. 5.70 Reed-Sternberg-like immunoblasts
(A) Large binucleate cell in case of angioimmunoblastic T-cell lymphoma; (B) Binucleate cell in toxoplasma lymphadenitis (MGG, HP).
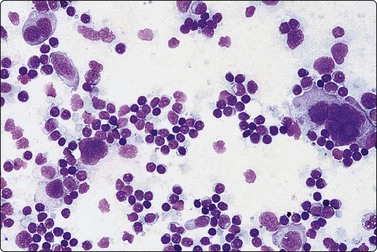
Fig. 5.71 Reed-Sternberg-like cells in peripheral T-cell lymphoma
Pleomorphic T-cell lymphoma with Reed-Sternberg-like cells (MGG, IP).
(Reproduced with permission from van Heerde et al.9)
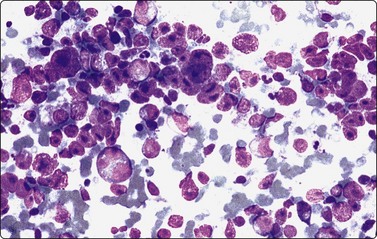
Fig. 5.72 Anaplastic large cell lymphoma
Large cell anaplastic lymphoma, CD30 positive; differential diagnosis of Hodgkin lymphoma is a common problem in ALK negative cases (MGG, LP).
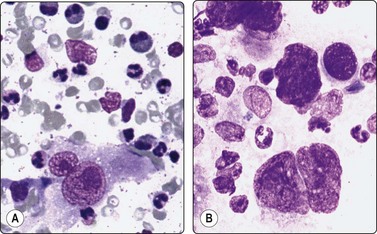
Fig. 5.73 Reed-Sternberg-like cells in metastatic carcinoma
(A) Large binucleate cells with large nucleoli in metastatic nasopharyngeal carcinoma; note eosinophils in the background; (B) Large binucleate cell in lymph node metastasis from squamous cell carcinoma of floor of mouth (MGG, HP).
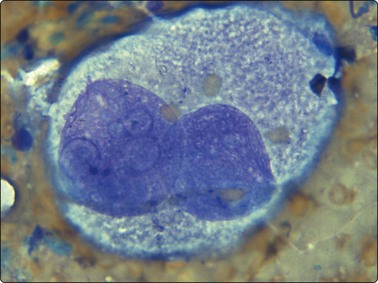
Fig. 5.74 Reed Sternberg-like cell in dendritic follicular cell sarcoma
Large binucleate cell with large nucleoli (Diff-Quik, HP oil).
The syncytial variant of HL, an uncommon form of nodular sclerosis HL, can be difficult to distinguish from NHL, carcinoma, melanoma, germ cell tumor and at times even sarcoma on FNB smears. Smears show cohesive sheets and clusters of large malignant cells with vesicular nuclei, prominent nucleoli and pale cytoplasm, and sometimes necrotic debris (see Fig. 5.68). Giant cells and multinucleated cells of Reed-Sternberg type can be recognized and may be numerous.188 An immunologic panel can help in the diagnosis (see Table 5.3).
Clusters of epithelioid histiocytes are sometimes seen in smears of HL – as in some NHL – which could suggest granulomatous lymphadenitis (Fig. 5.75). The lymphoid cells must therefore always be carefully scrutinized in lymph node smears containing epithelioid cells.
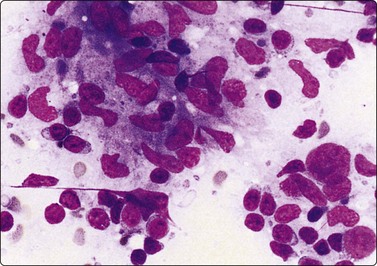
Clusters of epithelioid histiocytes resembling granulomatous lymphadenitis; note large abnormal cells lower right (MGG, HP).
Lymphocyte-rich smears may hide scanty numbers of large lymphoid neoplastic cells, resembling reactive lymphoid hyperplasia. Lymphocyte-rich classic HL may have a prominent small lymphocytic population with a few scattered Reed-Sternberg cells and variants. This may be difficult to distinguish from lymphocyte-predominant HL or T-cell/histiocyte rich B-cell lymphoma (see Fig. 5.46). Both of these may also have a paucity of large neoplastic cells in a rich reactive background. FNB is limited in making a distinction between nodular lymphocyte-predominant HL and T-cell/histiocyte rich B-cell lymphoma as the vital architecture cannot be assessed. To make the distinction between these entities, immunologic studies need to be carried out on a formal node biopsy.
Falling into the gray-zone lymphoma group are cases morphologically resembling classic HL; however, their immunophenotype is that of DLBCL ,NOS. A composite lymphoma needs to be excluded in biopsy specimens before making this classification.
FCM is of minimal value in the diagnosis of HL. An excess of CD4-positive T cells can be seen and a CD4/CD8 ratio greater than 4 should make one consider the diagnosis, even though it is not diagnostic. In nodular lymphocyte-predominant HL, not infrequently it may have a considerable proportion of CD4+CD8+(double-positive) T cells, a pointer to its diagnosis.69
Histiocytic and dendritic cell neoplasms
Histiocytic and dendritic cell neoplasms are rare and represent less than 1% of nodal tumors.69 They comprise: Langerhans cell histiocytosis (see page 84) and sarcoma, histiocytic sarcoma,9,105 follicular/interdigitating dendritic sarcoma,112-114 and dendritic cell sarcoma, NOS. There are a few case reports of most of these in the cytologic literature.
Histiocytic sarcoma (Fig. 5.76)9,105
Formerly known as true histiocytic lymphoma, it is difficult to recognize this rare type of neoplasm correctly in routine cytological smears. A very pleomorphic cell population with multilobed nuclei and multinucleated cells which may resemble Reed-Sternberg cells should make one think of this possibility. The main differential diagnosis includes large cell non-Hodgkin lymphomas (especially ALCL), HL, pleomorphic carcinoma and melanoma (see Table 5.3). At times, it is morphologically indistinguishable from DLBCL, NOS.
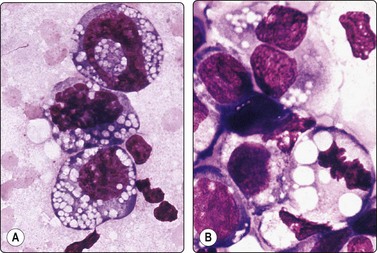
Fig. 5.76 True histiocytic lymphoma
Very polymorphous large cells, abundant basophilic cytoplasm, small vacuoles, ring-shaped nucleus in upper left corner, phagocytosis of erythrocytes in lower right corner; diagnosis confirmed by immunocytochemistry and EM (A and B, MGG, HP oil).
1 Australian Cancer Network Diagnosis and Management of Lymphoma Guidelines Working Party. Guidelines for the Diagnosis and Management of Lymphoma. Sydney: The Cancer Council Australia and Australian Cancer Network; 2005. 136
2 Ferrer R. Lymphadenopathy: differential diagnosis and evaluation. Am Fam Physician. 1998;58:1313-1320.
3 Dragoni F, Cartoni C, Pescarmona E, et al. The role of high resolution pulsed and colour Doppler ultrasound in the differential diagnosis of benign and malignant lymphadenopathy: results of multivariate analysis. Cancer. 1999;85:2485-2490.
4 Gupta AK, Nayar M, Chandra M. Reliability and limitations of fine needle aspiration cytology of lymphadenopathies: an analysis of 1,261 cases. Acta Cytol. 1991;35:777-783.
5 Perkins SL, Segal GH, Kjoldsberg CR. Work-up of lymphadenopathy in children. Sem Diagn Pathology. 1995;12:284.
6 Leong ASY, Stevens M. Fine-needle aspiration biopsy for the diagnosis of lymphoma: a perspective. Diagn Cytopathol. 1996;15:352-357.
7 Daskalopoulou D, Harhalakis N, Maouni N, et al. Institution fine needle aspiration cytology of non-Hodgkin lymphomas: a morphologic and immunophenotypic study. Acta Cytol. 1994;39:180-186.
8 Geisinger KR, Rainer RO, Field AS. Lymph nodes. In: Geisinger KR, Silverman JF, editors. Fine needle aspiration cytology of superficial organs and body sites. New York: Churchill-Livingstone; 1999:1-49.
9 van Heerde P, Meyer CJLM, Noorduyn LA, et al. An atlas and textbook of malignant lymphomas. Cytology, histopathology and immunochemistry. USA: Oxford University Press; 1996.
10 Jorgensen JL. State of the Art Symposium: flow cytometry in the diagnosis of lymphoproliferative disorders by fine-needle aspiration. Cancer (Cancer Cytopathol). 2005;105:443-451.
11 Caraway NP. State of the Art Symposuim: strategies to diagnose lymphoproliferative disorders by fine-needle aspiration using ancillary studies. Cancer (Cancer Cytopathol). 2005;105:432-442.
12 Knowles DM. Neoplastic hematopathology. Philadelphia: Lippincott, Williams and Wilkins; 2001.
13 Aratake Y, Tamura K, Kotani T, et al. Application of the avidin-biotin complex method for the light microscopic analysis of lymphocyte subsets with monoclonal antibodies on air-dried smears. Acta Cytol. 1988;32:117-122.
14 Levitt S, Cheng L, DuPuis MH, et al. Fine needle aspiration diagnosis of malignant lymphoma with confirmation by immunoperoxidase staining. Acta Cytol. 1985;29:895-902.
15 Robey SS, Cafferty LL, Beschozner WE, et al. Value of lymphocyte marker studies in diagnostic cytopathology. Acta Cytol. 1987;31:453-459.
16 Sneige N, Dekmezian RH, Katz RL, et al. Morphologic and immunocytochemical evaluation of 220 fine needle aspirates of malignant lymphoma and lymphoid hyperplasia. Acta Cytol. 1990;34:311-322.
17 Tani EM, Christensson B, Porwit A, et al. Immunocytochemical analysis and cytomorphologic diagnosis on fine needle aspirates of lymphoproliferative disease. Acta Cytol. 1988;32:209-215.
18 Robins DB, Katz RL, Swan F, et al. Immunotyping of lymphoma by fine-needle aspiration. A comparative study of cytospin preparations and flow-cytometry. Am J Clin Pathol. 1994;101:569-576.
19 Meda BA, Buss DH, Woodruff RD, et al. Diagnosis and subclassification of primary and recurrent lymphoma. The usefulness and limitations of combined fine-needle aspiration cytomorphology and flow cytometry. Am J Clin Pathol. 2000;113:688-699.
20 Liu K, Stern RC, Rogers RT, et al. Diagnosis of hematopoetic processes by fine-needle aspiration in conjunction with flow cytometry: a review of 127 cases. Diagn Cytopathol. 2001;24:1-10.
21 Young NA, Al-Saleem TI, Ehya H, et al. Utilization of fine-needle aspiration cytology and flow cytometry in the diagnosis and subclassification of primary and recurrent lymphoma. Cancer (Cancer Cytopathol). 1998;84:252-261.
22 Wakeley PE. Fine-needle aspiration cytopathology in diagnosis and classification of malignant lymphoma: accurate and reliable? Diagn Cytopathol. 2000;22:120-125.
23 Prasad RRA, Narasimkan R, Sankarou V, et al. Fine-needle aspiration cytology in the diagnosis of superficial lymphadenopathy: and analysis of 2,418 cases. Diagn Cytopathol. 1996;15:382-386.
24 Das DK. Value and limitations of fine-needle aspiration cytology in diagnosis and classification of lymphomas: a review. Diagn Cytopathol. 1999;21:240-249.
25 Dong HY, Harris NL, Preffer FJ, et al. Fine needle aspiration in the diagnosis and classification of primary and recurrent lymphoma: a retrospective analysis of the utility of cytomorphology and flow cytometry. Mod Pathol. 2001;14:472-481.
26 Zeppa P, Matrino G, Troncone G, et al. Fine-needle cytology and flow cytometry immunophenotyping and subclassification of non-Hodgkin lymphoma: a critical review of 307 cases with technical suggestions. Cancer. 2004;102:55-65.
27 Sigstad E, Dong HP, Davidson B, et al. The role of flow cytometric immunophenotyping in improving the diagnostic accuracy in referred fine-needle aspiration specimens. Diagn Cytopathol. 2004;31:159-613.
28 Nicol TL, Silberman M, Berner A, et al. The accuracy of combined cytopathologic and flow cytometric analysis of fine-needle aspirates of lymph nodes. Am Clin Pathol. 2000;114:18-28.
29 Ravinsky E, Morales C, Kutryk E, et al. Cytodiagnosis of lymphoid proliferations by fine needle aspiration biopsy. Adjunctive value of flow cytometry. Acta Cytol. 1999;43:1070-1078.
30 Mourad WA, Tulbah A, Shoukri M, et al. Primary diagnosis and REAL/WHO classification of non-Hodgkin lymphoma by fine-needle aspiration: cytomorphologic and immunophenotypic approach. Diagn Cytopathol. 2003;28:191-195.
31 Safley AM, Buckley PJ, Creager AJ, et al. The value of fluorescence in situ hybridization and polymerase chain reaction in the diagnosis of B-cell non-Hodgkin lymphoma by fine-needle aspiration. Arch Pathol Lab Med. 2004;128:1395-1403.
32 Bangerter M, Brudler O, Heinrich B, et al. Fine needle aspiration cytology and flow cytometry in the diagnosis and subclassification of non-Hodgkin lymphoma. Acta Cytol. 2007;51:390-398.
33 Gascoyne RD. Establishing the diagnosis of lymphoma: from initial biopsy to clinical staging. Oncology. 1998;12:11-16.
34 DeKerviler E, Guermazi A, Zagdanski AM, et al. Image-guided core-needle biopsy in patients with suspected or recurrent lymphoma. Cancer. 2000;89:647-652.
35 Screaton NJ, Berman LH, Grant JW. Head and neck lymphadenopathy: evaluation with US-guided cutting needle biopsy. Radiology. 2002;224:75-81.
36 Surbone A, Longo DL, DeVita VTJr, et al. Residual abdominal masses in aggressive non-Hodgkin lymphoma after combination chemotherapy: significance and management. J Clin Oncol. 1988;6:1832-1837.
37 Zajdela A, Ennuyer A, Bataini P, et al. Valeur du diagnostic cytologique des adenopathies par ponction aspiration. Confrontation cyto-histologique de 1756 cas. Bull Cancer (Paris). 1976;63:327-340.
38 Silverman SG, Lee BY, Mueller PR, et al. Impact of positive findings at image-guided biopsy of lymphoma on patient care: evaluation of clinical history, needle size, and pathologic findings on biopsy performance. Radiology. 1994;190:759-764.
39 Zornoza J, Cabanillas FF, Althoff TM, et al. Percutaneous needle biopsy in abdominal lymphoma. AJR. 1981;136:97-103.
40 Friedman M, Kim U, Shimaoka K, et al. Appraisal of aspiration cytology in management of Hodgkin’s disease. Cancer. 1980;45:1653-1663.
41 Lopes Cardozo P. The cytologic diagnosis of lymph node punctures. Acta Cytol. 1964;8:194-205.
42 Betsill WL, Hajdu SI. Percutaneous aspiration biopsy of lymph nodes. Am J Clin Pathol. 1980;73:471-479.
43 Cardillo MR. Fine needle aspiration cytology of superficial lymph nodes. Diagn Cytopathol. 1989;5:166-173.
44 Engzell U, Jakobsson PA, Sigurdson A, et al. Aspiration biopsy of metastatic carcinoma in lymph nodes of the neck: a review of 1101 consecutive cases. Acta Otolaryngol. 1971;72:138-147.
45 Kline TS, Kannan V, Kline IK. Lymphadenopathy and aspiration biopsy cytology. Review of 376 superficial nodes. Cancer. 1984;54:1076-1081.
46 Kline TS, Neal HS. Needle aspiration biopsy: a critical appraisal. Eight years and 3,267 specimens later. JAMA. 1978;239:36-39.
47 Lee RE, Valaitis J, Kalis O, et al. Lymph node examination by fine needle aspiration in patients with known or suspected malignancy. Acta Cytol. 1987;31:563-572.
48 Pilotti S, Di Palma S, Alasio L, et al. Diagnostic assessment of enlarged superficial lymph nodes by fine needle aspirate. Acta Cytol. 1993;37:853-864.
49 Hsu C, Leung BS, Lau SK, et al. Efficacy of fine needle aspiration and sampling of lymph nodes in 1484 Chinese patients. Diagn Cytopathol. 1990;6:154-159.
50 Shaha A, Webber C, Marti J. Fine needle aspiration in the diagnosis of cervical lymphadenopathy. Am J Surg. 1986;152:420-423.
51 Steel B, Schwartz MR, Ramzy I. Fine needle aspiration biopsy in the diagnosis of lymphadenopathy in 1,103 patients. Acta Cytol. 1995;39:76-81.
52 Gupta RK, Naran S, Lallu S, et al. Diagnostic value of needle aspiration cytology in the assessment of palpable inguinal lymph nodes. A study of 210 cases. Diagn Cytopathol. 2003;28:175-180.
53 Jaffer S, Zakowski M. Fine-needle aspiration biopsy of axillary lymph nodes. Diagn Cytopathol. 2002;26:69-74.
54 Cartagena N, Katz RL, Hirsch-Ginsberg C, et al. Accuracy of diagnosis of malignant lymphoma by combining fine needle aspiration cytomorphology with immunocytochemistry and in selected cases, southern blotting of aspirated cells: a tissue-controlled study of 86 patients. Diagn Cytopathol. 1992;8:456-464.
55 van Heerde P, Go DMDS, Koolman-Schellekens MA, et al. Cytodiagnosis of non-Hodgkin lymphoma. A morphological analysis of 215 biopsy proven cases. Virchows Arch (Pathol Anat). 1984;403:213-233.
56 Cafferty LL, Katz RL, Ordonez NG, et al. Fine needle aspiration diagnosis of intraabdominal and retroperitoneal lymphomas by a morphologic and immunocytochemical approach. Cancer. 1990;65:72-77.
57 Frable W. Fine needle aspiration biopsy. Philadelphia: Saunders; 1983.
58 Orell SR, Skinner JM. The typing of non-Hodgkin lymphoma using fine needle aspiration cytology. Pathology. 1982;14:389-394.
59 Pitts WC, Weiss LM. Fine needle aspiration biopsy of lymph nodes. Pathol Annu. 1988;23(pt 2):329-360.
60 Spieler P, Schmid U. How exact are the diagnosis and classification of malignant lymphomas from aspiration biopsy smears? Path Res Pract. 1978;163:232-250.
61 Carter TR, Feldman PS, Innes DJJr, et al. The role of fine needle aspiration cytology in the diagnosis of lymphoma. Acta Cytol. 1988;32:848-853.
62 Erwin BC, Brynes BK, Chan WC, et al. Percutaneous needle biopsy in the diagnosis and classification of lymphoma. Cancer. 1986;57:1074-1078.
63 Lopes Cardozo P. The significance of fine needle aspiration cytology for the diagnosis and treatment of malignant lymphomas. Folia Haematol (Leipzig). 1980;107:601-620.
64 Sandhaus LM. Fine-needle aspiration cytology in the diagnosis of lymphoma. The next step (edit). Am J Clin Pathol. 2000;113:623-627.
65 Chhieng DC, Cohen JM, Cangiarella JF. Cytology and immunophenotyping of low and intermediate grade B-cell non-Hodgkin lymphomas with a prominent small cell component. Diagn Cytopathol. 2001;24:90-97.
66 Russell J, Orell S, Skinner J, et al. Fine needle aspiration cytology in the management of lymphoma. Aust NZ J Med. 1983;13:365-368.
67 Egea AS, González MAM, Barrios AP, et al. Usefulness of light microscopy in lymph node fine needle aspiration biopsy. Acta Cytol. 2002;46:364-368.
68 Thomas JO, Adeyi D, Amanguno H. Fine-needle aspiration in the management of peripheral lymphadenopathy in a developing country. Diagn Cytopathol. 1999;21:159-162.
69 Swerdlow SH, Campo E, Harris NL, et al, editors. WHO classification of tumours of haematopoietic and lymphoid tissues. Lyon: IARC, 2008.
70 Hu E, Horning S, Flynn S, et al. Diagnosis of B cell lymphoma by analysis of immunoglobulin gene rearrangements in biopsy specimens obtained by fine needle aspiration. J Clin Oncol. 1986;4:278-283.
71 Jeffers MD, McCorriston J, Farquharson MA, et al. Analysis of clonality in cytologic material using the polymerase chain reaction (PCR). Cytopathology. 1997;8:114-121.
72 Lubinski J, Chosia M, Kotanska K, et al. Genotypic analysis of DNA isolated from fine needle aspiration biopsies. Anal Quant Cytol Histol. 1988;10:383-390.
73 Rovinsky E, Morales C. Diagnosis of lymphoma by image-guided needle biopsies: fine needle aspiration biopsy, core biopsy or both? Acta Cytol. 2005;49:51-57.
74 Gong JZ, Synder MJ, Lagoo AS, et al. Diagnostic impact of core needle biopsy on fine needle aspiration of non-Hodgkin lymphoma. Diagn Cytopathol. 2004;31:23-30.
75 Fulciniti F, Vetrani A, Zeppa P, et al. Hodgkin’s disease: diagnostic accuracy of fine needle aspiration; a report based on 62 consecutive cases. Cytopathol. 1994;5:226-233.
76 Jiménez-Hefferman JA, Vicandi B, López-Ferrer P, et al. Value of fine needle aspiration cytology in the initial diagnosis of Hodgkin disease. Analysis of 188 cases with an emphasis on diagnostic pitfalls. Acta Cytol. 2001;45:300-306.
77 Chhieng DC, Cangiarella JF, Symmans WF, et al. Fine-needle aspiration cytology of Hodgkin disease. A study of 89 cases with emphasis on false-negative cases. Cancer (Cancer Cytopathol). 2001;93:52-59.
78 Behm FG, O’Dowd CJ, Frable WJ. Fine needle aspiration effects on benign lymph node histology. Am J Clin Pathol. 1984;82:195-198.
79 Davies JD, Webb AJ. Segmental lymph-node infarction after fine needle aspiration. J Clin Pathol. 1982;35:855-857.
80 Suthipintawong C, Leong AS-Y, Vinyuvat S. Immunostaining of cell preparations. A comparative evaluation of common fixatives and protocols. Diagn Cytopathol. 1997;17:127-133.
81 Michael CW, Hunter B. Interpretation of fine-needle aspirates processed by ThinPrep techniques: cytologic artifacts and diagnostic pitfalls. Diagn Cytopathol. 2000;23:6-13.
82 Söderström N. Fine needle aspiration biopsy. Stockholm: Almqvist and Wiksell; 1966.
83 Zajicek J. Aspiration biopsy cytology. Part I. Cytology of supradiaphragmatic organs. Basel: Karger; 1974.
84 Noorduyn LA, van Heerde P, Meyer CJLM. Cytology of Ki-1 (CD-30) positive large cell lymphoma. Cytopathol. 1990;1:297-304.
85 Ng W-K, Ip P, Choy C, et al. Cytologic and immunocytochemical findings of anaplastic large cell lymphoma. Analysis of ten fine-needle aspiration specimens over 9-year period. Cancer (Cancer Cytopathol). 2003;99:33-43.
86 Zakowski MF, Feiner H, Finfer M, et al. Cytology of extranodal Ki-1 anaplastic large cell lymphoma. Diagn Cytopathol. 1996;14:155-161.
87 Stani J. Cytologic diagnosis of reactive lymphadenopathy in fine needle aspiration biopsy specimens. Acta Cytol. 1987;31:8-13.
88 O’Dowd GJ, Frable WJ, Behm FG. Fine needle aspiration cytology of benign lymph node hyperplasias. Diagnostic significance of lymphohistiocytic aggregates. Acta Cytol. 1985;29:554-558.
89 Kardos TF, Kornstein MJ, Frable WJ. Cytology and immunocytology of infectious mononucleosis in fine needle aspirates of lymph nodes. Acta Cytol. 1988;32:722-726.
90 Stanley MW, Steeper TA, Horwitz CA, et al. Fine-needle aspiration of lymph nodes in patients with acute infectious mononucleosis. Diagn Cytopathol. 1990;6:323-329.
91 Iacobuzio-Donahue CA, Clark DP, Ali SZ. Reed-Sternberg-like cells in lymph node aspirates in the absence of Hodgkin’s disease: pathologic significance and differential diagnosis. Diagn Cytopathol. 2002;27:335-339.
92 Kumar B, Karki S, Paudgal P. Diagnosis of sinus histiocytosis with massive lymphadenopathy (Rosai-Dorfman disease) by fine needle aspiration cytology. Diagn Cytopathol. 2008;36:691-695.
93 Alegret RA, Tello AM, Ramirez T, et al. Sinus histiocytosis with massive lymphadenopathy (Rosai-Dorfman disease): diagnosis with fine-needle aspiration in a case with nodal and nasal involvement. Diagn Cytopathol. 1995;13:333-335.
94 Stastny JF, Wilkerson ML, Hamati HF, et al. Cytologic features of sinus histiocytosis with massive lymphadenopathy. A report of three cases. Acta Cytol. 1997;41:871-876.
95 Santos-Briz A, López-Ríos F, Santos-Briz A, et al. Granulomatous reaction to silicone in axillary lymph nodes. A case report with cytologic findings. Acta Cytol. 1999;43:1163-1165.
96 Munichor M, Cohen H, Volpin G, et al. Chromium-induced lymph node histiocytic proliferation after hip replacement. Acta Cytol. 2003;47:270-274.
97 Argyle JC, Schumann GB, Kjeldsberg CR, et al. Identification of a toxoplasma cyst by fine needle aspiration. Am J Clin Pathol. 1983;80:256-258.
98 Christ ML, Feltes-Kennedy M. Fine needle aspiration cytology of toxoplasmic lymphadenitis. Acta Cytol. 1982;26:425-428.
99 Jayaram N, Ramaprasad AV, Chethan M, et al. Toxoplasma lymphadenitis. Analysis of cytologic and histopathologic criteria and correlation with serologic tests. Acta Cytol. 1997;41:653-658.
100 Zaharopoulos P. Demonstration of parasites in toxoplasma lymphadenitis by fine-needle aspiration cytology. Report of two cases. Diagn Cytopathol. 2000;22:11-15.
101 van Heerde P, Egeler RM. The cytology of Langerhans cell histiocytosis (histiocytosis X). Cytopathol. 1991;2:149-158.
102 Layfield LJ, Bhuta S. Fine needle aspiration cytology of histiocytosis X: a case report. Diagn Cytopathol. 1988;4:140-143.
103 Kakkar S, Kapila K, Verma K. Langerhans cell histiocytosis in lymph nodes. Cytomorphologic diagnosis and pitfalls. Acta Cytol. 2001;45:327-332.
104 Kumar PV, Mousavi A, Karimi M, et al. Fine needle aspiration of Langerhans cell histiocytosis of the lymph nodes. A report of six cases. Acta Cytol. 2002;46:753-756.
105 Miliauskas JR. Fine needle aspiration cytology of true histiocytic lymphoma/histiocytic sarcoma. Diagn Cytopathol. 2003;29:233-235.
106 Desol G, Pradere M, Voigt JJ, et al. Warthin-Finkeldey-like cells in benign and malignant lymphoid proliferations. Histopathology. 1982;6:451-465.
107 Hidvegi DF, Sorensen K, Lawrence JB, et al. Castleman’s disease. Cytomorphologic and cytochemical features of a case. Acta Cytol. 1982;26:243-246.
108 Meyer L, Gibbons D, Ashfaq R, et al. Fine-needle aspiration findings in Castleman’s disease. Diagn Cytopathol. 1999;21:57-60.
109 Deschenes M, Michel R, Tabah R, et al. Fine needle aspiration cytology of Castelman disease: Case report with review of the literature. Diagn Cytopathol. 2008;36:904-908.
110 Jayaram G, Peh KB. Fine-needle aspiration cytology in Kimura’s disease. Diagn Cytopathol. 1995;13:295-299.
111 Deshpande AH, Nayak S, Munshi MM, et al. Kimura’s disease. Diagnosis by aspiration cytology. Acta Cytol. 2002;46:357-363.
112 Herceg RJ, Nayar R, De Frias DVS. Cytomorphologic appearance of follicular dendritic-cell tumor: a case report. Diagn Cytopathol. 1999;20:237-240.
113 Loo CKC, Henderson C, Rogan K. Intraabdominal follicular dendritic cell sarcoma. Report of a case with fine needle aspiration findings. Acta Cytol. 2001;45:999-1004.
114 Jayaram G, Mun KS, Elsayed EM, et al. Interdigitating dendritic reticulum cell sarcoma: cytologic, histologic and immunocytochemical features. Diagn Cytopathol. 2005;33:43-48.
115 Bailey TM, Akhtar M, Ali MA. Fine needle aspiration biopsy in the diagnosis of tuberculosis. Acta Cytol. 1985;29:732-736.
116 Klemi PJ, Elo JJ, Joensuu H. Fine needle aspiration biopsy of granulomatous disorders. Sarcoidosis. 1987;4:38-41.
117 Radhika S, Gupta SK, Chakrabarti A, et al. Role of culture for mycobacteria in fine needle aspiration diagnosis of tuberculous lymphadenitis. Diagn Cytopathol. 1989;5:260-262.
118 Gupta AK, Nayar M, Chandra M. Clinical appraisal of fine needle aspiration cytology in tuberculosis lymphadenitis. Acta Cytol. 1992;36:391-394.
119 Ellison E, Lapuerta P, Martin SE. Fine needle aspiration diagnosis of mycobacterial lymphadenitis. Sensitivity and predictive value in the United States. Acta Cytol. 1999;43:153-157.
120 Singh KK, Muralidhar M, Kumar A, et al. Comparison of in-house polymerase chain reaction with conventional techniques for the detection of Mycobacterium tuberculosis DNA in granulomatous lymphadenopathy. J.Clin. Pathol. 2000;53:355-361.
121 Cavett JRIII, McAfee R, Ramzy I. Hansen’s disease (leprosy). Diagnosis by aspiration biopsy of lymph nodes. Acta Cytol. 1986;30:189-193.
122 Silverman JF. Fine needle aspiration cytology of cat scratch disease. Acta Cytol. 1985;29:542-547.
123 Donnelly A, Hendricks G, Martens S, et al. Cytologic diagnosis of cat scratch disease (CSD) by fine-needle aspiration. Diagn Cytopathol. 1995;13:103-106.
124 Stastny JF, Wakeley PE, Frable WJ. Cytologic features of necrotizing granulomatous inflammation consistent with cat-scratch disease. Diagn Cytopathol. 1996;15:108-115.
125 Gamborino F, Carrilho C, Ferro J, et al. FNA diagnosis of Kaposi’s sarcoma in a developing country. Diagn Cytopathol. 2000;23:322-325.
126 Gaglioano EF. Fine needle aspiration cytology of Kaposi’s sarcoma in lymph nodes. A case report. Acta Cytol. 1987;31:25-28.
127 Hales M, Bottles K, Miller T, et al. Diagnosis of Kaposi’s sarcoma by fine needle aspiration biopsy. Am J Clin Pathol. 1987;88:20-25.
128 Viguer JM, Jiménez-Heffernan JA, Pérez P, et al. Fine-needle aspiration cytology of Kikuchi’s lymphadenitis: a report of ten cases. Diagn Cytopathol. 2001;25:220-224.
129 Tswang WY, Chan JK. Fine needle aspiration cytologic findings of Kikuchi’s lymphadenitis: an analysis of 27 cases. Am J Clin Pathol. 1994;102:454-458.
130 Tong TRS, Chan OW, Lee K. Diagnosing Kikuchi disease on fine needle aspiration biopsy. A retrospective study of 44 cases diagnosed by cytology and 8 by histopathology. Acta Cytol. 2001;45:953-957.
131 Pai MR, Adhikari P, Coimbatore RVR, et al. Fine needle aspiration cytology in systemic lupus erythematosus lymphadenopathy. A case report. Acta Cytol. 2000;44:67-69.
132 Laszewski MJ, Belding PJ, Feddersen RM, et al. Clonal immunoglobulin gene rearrangement in the infracted lymph node syndrome. Am J Clin Pathol. 1991;96:116-120.
133 Üstün M, Risberg B, Davidson B, et al. Cystic change in metastatic lymph nodes: a common diagnostic pitfall in fine-needle aspiration cytology. Diagn Cytopathol. 2002;27:387-392.
134 Shin HJ, Caraway NP. Fine-needle aspiration biopsy of metastatic small cell carcinoma from extrapulmonary sites. Diagn Cytopathol. 1998;19:177-181.
135 Nicholson SA, Ryan MR. A review of cytologic findings in neuroendocrine carcinomas including carcinoid tumors with histologic correlation. Cancer (Cancer Cytopathol). 2000;90:48-61.
136 Highman J, Oliver RT. Diagnosis of metastases from testicular germ cell tumours using fine needle aspiration cytology. J Clin Pathol. 1987;40:1324-1333.
137 Allhoff EP, Proppe KH, Chapman CM, et al. Evaluation of prostatic specific acid phosphatase and prostatic specific antigen in identification of prostatic cancer. J Urol. 1983;129:315-318.
138 Campbell F, Herrington CS. Application of cytokeratin 7 and 20 immunohistochemistry to diagnostic pathology. Current Diagn Pathol. 2001;7:113-122.
139 Chu PG, Weiss LM. Keratin expression in human tissues and neoplasms. Histopathol. 2002;40:403-439.
140 Fong Y, Coit DG, Woodruff JM, et al. Lymph node metastases from soft tissue sarcoma in adults: analysis of data from a prospective database of 1772 sarcoma patients. Ann Surg. 1993;217:72-77.
141 Bizjak-Schwarzbartl M. Cytomorphologic characteristics of non-Hodgkin lymphoma. Acta Cytol. 1988;32:216-220.
142 Frable WJ, Kardos TF. Fine needle aspiration biopsy. Applications in the diagnosis of lymphoproliferative diseases. Am J Surg Pathol. 1988;12(Suppl. 1):62-72.
143 Gagneten D, Hijazi YM, Jaffe ES, et al. Mantle cell lymphoma. A cytopathological and immunocytochemical study. Diagn Cytopathol. 1996;14:32-37.
144 Katz RL. Cytologic diagnosis of leukemia and lymphoma. Values and limitations. Clin Lab Med. 1991;11:469-499.
145 Young NA, Al-Saleem TI. Diagnosis of lymphoma by fine-needle aspiration cytology using the revised European-American classification of lymphoid neoplasms. Cancer (Cancer Cytopathol). 1999;87:325-345.
146 Tani E, Santos GC, Svedmyr E, et al. Fine-needle aspiration cytology and immunocytochemistry of soft-tissue extramedullary plasma-cell neoplasms. Diagn Cytopathol. 1999;20:120-124.
147 Bangerter M, Hildebrand A, Waidman O, et al. Fine needle aspiration cytology in extramedullary plasmacytoma. Acta Cytol. 2000;44:287-291.
148 Sun W, Caraway NP, Zhang HZ, et al. Grading follicular lymphoma or fine needle aspiration specimens: Comparison with proliferative index of DNA image analysis and Ki-67 labelling indeed. Acta Cytol. 2004;48:119-126.
149 Skoog L, Tani E. FNA cytology in the diagnosis of lymphoma. In: Orell SR, editor. Monographs in Clinical Cytology, Vol. 18. Basel: Karger; 2008:26.
150 Rassidakis GZ, Tani E, Svedmyr E, et al. Diagnosis and subclassification of follicle center and mantle cell lymphomas on fine-needle aspirates. A cytologic and immunocytochemical approach based on the revised European–American Lymphoma (REAL) classification. Cancer (Cancer Cytopathol). 1999;87:216-223.
151 Hughes JH, Caraway NP, Katz RL. Blastic variant of mantle cell lymphoma: cytomorphologic, immunocytochemical and molecular genetic features of tissue obtained by fine needle aspiration biopsy. Diagn Cytopathol. 1998;19:59-62.
152 Matsushima AY, Hamele-Bena D, Osborne BM. Fine-needle aspiration biopsy findings in marginal zone B cell lymphoma. Diagn Cytopathol. 1999;20:190-198.
153 Murphy BA, Meda BA, Buss BH, et al. Marginal zone and mantle cell lymphomas: assessment of cytomorphology in subtyping small B-cell lymphomas. Diagn Cytopathol. 2003;28:126-130.
154 De Jong D, van Gorp J, Sie-Go D, et al. T-cell rich B-cell non-Hodgkin lymphoma: a progressed form of follicle centre cell lymphoma and lymphocyte predominance Hodgkin’s disease. Histopathology. 1996;28:15-24.
155 Galindo LM, Havlioglu N, Grosso LE. Cytologic findings in a case of T-cell rich B-cell lymphoma: potential diagnostic pitfall in fine needle aspiration of lymph nodes. Diagn Cytopathol. 1996;14:253-258.
156 Tani E, Johansson B, Skoog L. T-cell rich B-cell lymphoma: fine-needle aspiration cytology and immunocytochemistry. Diagn Cytopathol. 1998;18:1-4.
157 Tong TR, Lee KC, Chow TC, et al. T-cell/histiocyte-rich diffuse large B-cell lymphoma. Report of a case diagnosed by fine needle aspiration biopsy with immunohistochemical and molecular pathologic correlation. Acta Cytol. 2002;46:893-898.
158 Lin O, Gerhard R, Zerbini M, et al. Cytologic features of plasmablastic lymphoma. Diagn Cytopathol. 2005;105:139-144.
159 Das DK, Gupta SK, Pathak IC, et al. Burkitt-type lymphoma. Diagnosis by fine needle aspiration cytology. Acta Cytol. 1987;31:1-7.
160 Stastny JF, Almeida MM, Wakely PE, et al. Fine-needle aspiration biopsy and imprint cytology of small non-cleaved cell (Burkitt’s) lymphoma. Diagn Cytopathol. 1995;12:201-207.
161 Troxell ML, Bangs CD, Cherry AM, et al. Cytologic diagnosis of Burkitt lymphoma. Cancer. 2005;105:310-318.
162 Dunphy CH, Katz RL, Fanning CV, et al. Leukemic lymphadenopathy: diagnosis by fine needle aspiration. Hematol Pathol. 1989;3:35-44.
163 Frizzera G, Wu D, Inghiremi G. The usefulness of immunophenotypic and genotypic studies in the diagnosis and classification of hematopoietic and lymphoid neoplasms. Am J Clin Pathol. 1999;111(Suppl. 1):S13-S39.
164 Al Shanqeety O, Mourad WA. Diagnosis of peripheral T-cell lymphoma by fine-needle aspiration biopsy: a cytomorphologic and immunophenotypic approach. Diagn Cytopathol. 2000;23:375-379.
165 Yao JL, Cangiarella JF, Cohen J-M, et al. Fine-needle aspiration biopsy of peripheral T-cell lymphomas. A cytologic and immunophenotypic study of 33 cases. Cancer (Cancer Cytopathol). 2001;93:151-159.
166 Oertel J, Oertel B, Lobeck KH, et al. Cytologic and immunocytologic studies of peripheral T-cell lymphoma. Acta Cytol. 1991;35:285-293.
167 Chan ABW, Chan WY, Chow JHS. Cytologic features of NK/T-cell lymphoma. Acta Cytol. 2003;47:595-601.
168 Wai-Kuen Ng, Lee CYK, Li ASM, et al. Nodal presentation of nasal-type NK/T-cell lymphoma: report of two cases with fine needle aspiration cytology findings. Acta Cytol. 2003;47:1063-1068.
169 Ng W-k, Ip P, Choy C, et al. Cytologic findings of angioimmunoblastic T-cell lymphoma. Analysis of 16 fine-needle aspirates over a 9-year period. Cancer (Cancer Cytopathol). 2002;96:166-173.
170 Attygalle A, Al-Jehani R, Diss TC, et al. Neoplastic T cells in angioimmunoblastic T-cell lymphoma express CD10. Blood. 2002;99:627-633.
171 Tani E, Löwhagen T, Nasiell K, et al. Fine needle aspiration cytology and immunocytochemistry of large cell lymphomas expressing the Ki-1 antigen. Acta Cytol. 1989;33:359-362.
172 Mourad WA, Nazer M, Tulbah A. Cytomorphologic differetiation of Hodgkin lymphoma and Ki-1+ anaplastic large cell lymphoma in fine needle aspirates. Acta Cytol. 2003;47:744-748.
173 Rapkiewicz A, Wen H, Sen F, et al. Cytomorphologic examination of anaplastic large cell lymphoma by fine needle aspiration cytology. Cancer Cytopathol. 2007;111:499-507.
174 Katz RL, Caraway NP. Lymph nodes, 5th ed. Koss LG, Melamed MR, editors. Koss Diagnostic cytology and its histopathologic bases, Vol. 2. 2006, 1205-1208.
175 Gorczyca W, Liu Z, Tsang P, et al. B-cell lymphomas with coexpression of CD5 and CD10. Am J Clin Pathol. 2003;119:218-230.
176 Kussick SJ, Kalnoski M, Braziel RM, et al. Prominent clonal B-cell populations identified by flow cytometry in histologically reactive lymphoid proliferations. Am. J Clin Pathol. 2004;121:464-472.
177 Derksen PW, Langerak AW, Kerkhof E, et al. Comparison of different polymerase chain reaction-based approaches for clonality assessment of immunoglobulin heavy-chain gene rearrangements in B-cell neoplasia. Mod Pathol. 1999;12:794-805.
178 Diss TC, Peng H, Wotherspoon AC, et al. Detection of monoclonality in low-grade B-cell lymphomas using the polymerase chain reaction is dependent on primer selection and lymphoma type. J Pathol. 1993;169:291-295.
179 Achille A, Scarpa A, Montresor M, et al. Routine application of polymerase chain reaction in the diagnosis of monoclonality of B-cell lymphoid proliferations. Diag Mol Pathol. 1995;4:14-24.
180 Segal GH, Jorgensen T, Masih AS, et al. Optimal primer selection for clonality assessment by polymerase chain reaction analysis: low-grade B-cell lymphoproliferative disorders of nonfollicular centre cell type. Hum Pathol. 1994;25:1269-1275.
181 Gattuso P, Castelli MJ, Peng Y, et al. Post transplant lymphoproliferative disorders: a fine needle aspiration biopsy study. Diagn Cytopathol. 1997;16:392-395.
182 Hecht JL, Cibas ES, Kutok JL. Fine-needle aspiration cytology of lymphoproliferative disorders in the immunosuppressed patient: The diagnostic utility of in situ hybridization for Epstein-Barr virus. Diagn Cytopathol. 2002;26:360-365.
183 Harris NL, Ferry JA, Swerdlow SH. Post transplant lymphoproliferative disorders: Summary of Society for Hematopathology Workshop. Sem Diagn Pathol. 1997;14:8-14.
184 Dmitrovsky E, Martin SE, Krudy AG, et al. Lymph node aspiration in the management of Hodgkin’s disease. J Clin Oncol. 1986;4:306-310.
185 Kardos TF, Vinson JH, Behm FG, et al. Hodgkin’s disease: diagnosis by fine needle aspiration biopsy. Analysis of cytologic criteria from a selected series. Am J Clin Pathol. 1986;86:286-291.
186 Moriarty AT, Banks ER, Bloch T. Cytologic criteria for subclassification of Hodgkin’s disease using fine needle aspiration. Diagn Cytopathol. 1989;5:122-125.
187 Strum SB, Park JK, Rappaport H. Observation of cells resembling Sternberg-Reed cells in conditions other than Hodgkin’s disease. Cancer. 1970;26:176-190.
188 Stanley MW, Powers CN. Syncytial variant of nodular sclerosing Hodgkin’s disease: fine-needle aspiration findings in two cases. Diagn Cytopathol. 1997;17:477-479.