CHAPTER 16 Bone
Clinical aspects
Needle aspiration has been used in the investigation of bone lesions ever since the technique was introduced. Coley and Ellis included several examples of primary and metastatic bone malignancies in their original paper.1 Fine needle biopsy (FNB) and cytodiagnosis of bone lesions are now well-established methods in many centers. It is not possible to penetrate intact cortical bone or sclerotic lesions using a truly thin needle (22–23 gauge) but partly destroyed or ‘moth-eaten’ cortical bone can often be penetrated. New devices, such as the Bone Biopty instrument, a coaxial biopsy system with an eccentric drill,2 have made it possible to drill through intact cortical bone and to insert the fine needle into the lesion through the drilled canal. FNB has the advantage over open biopsy of being less disruptive to bone, permitting multiple sampling without complications and leaving no scar. There is no risk of infection if simple sterility is observed. FNB is a simple outpatient procedure, cost effective and rapid. Its primary purpose is not only to obtain a morphologic diagnosis of benignity or malignancy, to investigate suspected bone secondaries, or sometimes to aspirate material for a bacteriologic diagnosis from osteolytic lesions, radiologically suspected for osteomyelitis, but also, if possible, to replace open or coarse needle biopsy in the diagnosis of primary bone tumors before treatment. Ancillary diagnostic techniques such as cytochemistry, immunocytochemistry, flow cytometric phenotyping, DNA ploidy analysis, molecular genetic analysis and, in some special situations, electron microscopy are important supplements to cytodiagnosis of specific tumor entities before definitive treatment.
Spinal lesions with threatening cord compression are a medical emergency and FNB can rapidly resolve the differential diagnosis between osteomyelitis, metastatic malignancy and a lymphoproliferative malignancy. The diagnosis of Langerhans cell histiocytosis (eosinophilic granuloma) and of plasmacytoma is, in most cases, relatively easy and the distinction between non-Hodgkin’s lymphoma and other small cell malignancies (primary or secondary) is possible. FNB is well suited to the investigation of the cause of pathological fractures, but the proliferating osteoblasts in a fracture callus must be recognised as such. Our experience, and that of others, in the type-specific diagnosis of primary bone lesions is rather substantial, covering all of the malignant bone tumors except adamantinoma, and most of the benign tumors. Definitive diagnosis is possible since the cytomorphology of primary bone tumors, benign as well as malignant, has now been described in detail in correlative cytological/histological studies3 and in single monographs and reviews4,5
The correct interpretation of smears from a bone lesion requires access to the full clinical and radiological assessment, just as in histological diagnosis of a formal bone biopsy. The cytopathologist should refrain from giving a definitive diagnosis when the results of the radiologic investigation are not known.
Accuracy of diagnosis
Adequate material was obtained in 86–97% in four series, comprising in total 1453 cases.3,6-9 The diagnostic accuracy in the diagnosis of metastases is very high, in the order of 90%. Its value in the diagnosis of primary bone lesions varied between 90% and 97% in the above cited series.
Complications
Complication are few. We have more than 30 years’ experience of FNB of bone lesions in our musculoskeletal tumor centre in Lund University Hospital and have never seen any severe complication. Brief pain at needling is not uncommon but significant hemorrhage is unusual, as is pneumothorax after biopsy of rib or spine lesion. FNB of spinal lesions carries the additional, albeit rare, complication of neurological damage.10 FNB of bone lesions in patients with a low platelet count or a bleeding disorder must be discussed with the clinician in charge before proceeding to biopsy, and the importance to clinical management of a cytological diagnosis must be decided.
Technical considerations
Fine needle biopsy guided by fluoroscopy or computed tomography (CT) in nonpalpable lytic lesion is a rapid and safe method to obtain a morphologic diagnosis of lesions anywhere in the skeleton, particularly in the spine.5,11,12 Some pain is usually felt at aspiration. Local anesthesia prevents periosteal pain and is recommended in conjunction with fluoroscopy or CT guidance, as several passes with the needle may be necessary to obtain a representative specimen. Bone aspirates are often heavily bloodstained and methods similar to those used in processing hematological bone marrow aspirates are useful to concentrate the cell material. The use of a watch glass to select tissue fragments and of thrombin-clot preparations for paraffin sections and histology are of particular value (see Chapter 2). Both air-dried MGG smears and wet-fixed smears for H&E or Pap should be made. MGG smears are particularly valuable in assessing bone marrow elements and chondroid, myxoid or osteoid material; wet-fixed smears are superior for nuclear detail, particularly in tissue fragments (‘microbiopsies’).
Cytological findings
The cytopathologist must be familiar with the appearance of cellular marrow. Taken individually, the nuclear features of immature marrow cells may be disturbingly similar to malignant cells, but the mixture of all cell types allows easy identification. The irregular multilobed nucleus of the megakaryocytes in particular can cause alarm if the site of origin is unsuspected. MGG-stained smears are much superior to wet-fixed H&E or Pap smears in identifying hematopoietic or lymphoid cells. Bone marrow cells are frequently found, particularly in aspirates from the ribs, vertebrae and sacrum.
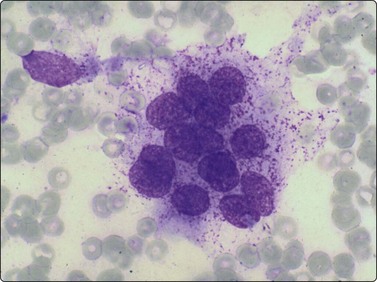
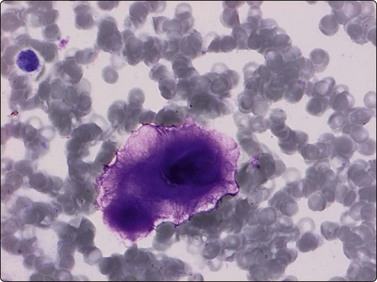
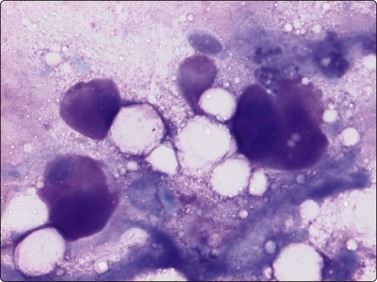
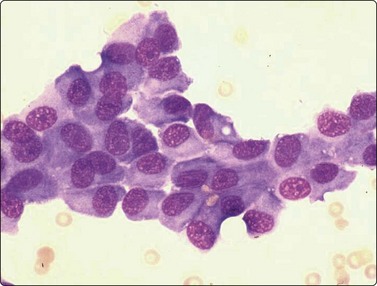
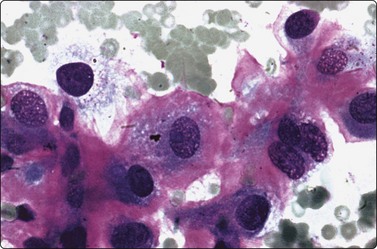
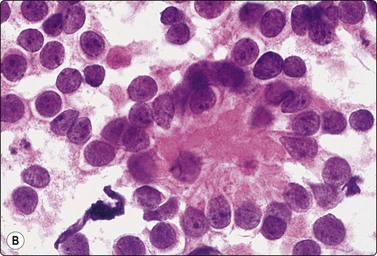
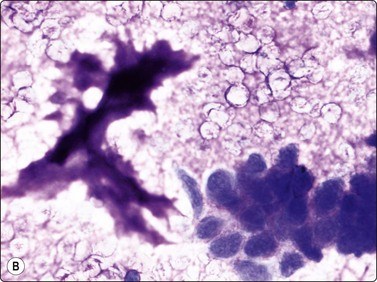
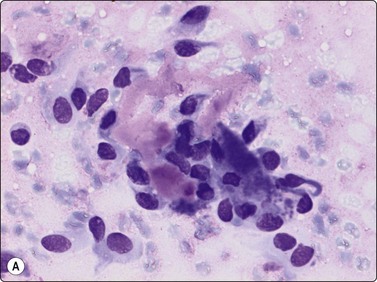
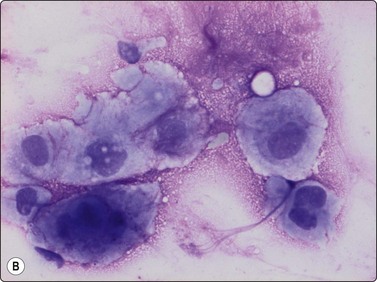
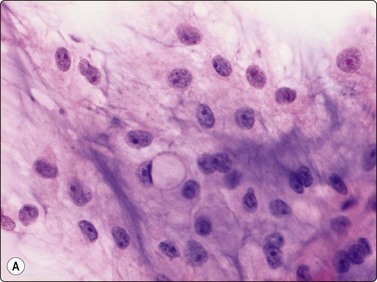
Osteoblasts are commonly seen in aspirates from all kinds of bone lesions. They present either as single cells or as small groups or runs. They have a characteristic eccentric nucleus which sometimes seems to protrude from the cytoplasm. The nucleus is round or oval and often contains a central nucleolus. The cytoplasm is dense, amphophilic or basophilic with a central clear area or ‘Hof’ separated from the nucleus (Fig. 16.1). Reactive osteoblasts in a fracture callus or in benign lesions such as reactive periosteitis may show marked anisokaryosis and prominent nucleoli (Fig. 16.2). Osteoclasts are large cells which possess at least 10–15 uniform nuclei and have abundant cytoplasm with a similar texture to osteoblasts and well-defined borders. A fine, pink cytoplasmic granularity is visible in MGG-stained smears (Fig. 16.3). Normal cartilage does not smear well as it is very cohesive. Flecks or clumps of cartilage may be removed from joint surfaces or costochondral junctions. These show bright red or magenta staining with MGG and are pale and translucent with Pap. Scalloped or fibrillar edges are a feature (Fig. 16.4). Free chondrocytes are almost never seen, but the cells are visible within lacunae in the chondroid matrix. They have small, pale, rounded nuclei and poorly stained cytoplasm, often presenting as a halo. They are best visualised in wet-fixed smears. The chondroid matrix stains heavily with MGG and obscures the fine structure of the cells (Fig. 16.5). A rare but important contamination of smears from spinal FNBs is clusters of mesothelial cells which may be sampled if the lesion is missed by the needle. Such cells may be mistaken for metastatic deposits of well-differentiated carcinoma, but usually do not have a bloody background (Fig. 16.6).
Cluster of cells with a plasma cell-like appearance; abundant cytoplasm and eccentric nuclei; ‘Hof’ separated from nucleus is visible in several cells. Smear derived from an area of moderate osteoblastic activity and new bone formation (H&E, HP).
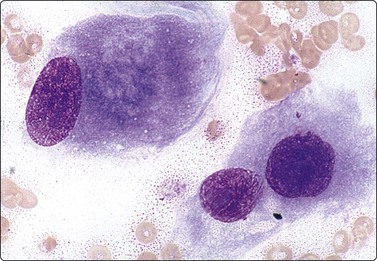
Reactive osteoblasts (florid reactive periosteitis) showing marked anisokaryosis and enlarged nucleoli. A cytoplasmic ‘Hof’ is visible (MGG, HP oil).
Osteomyelitis
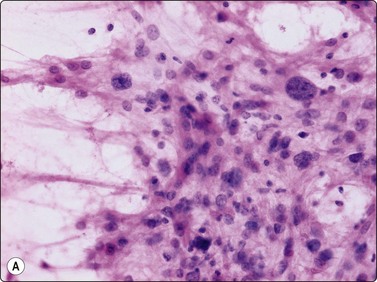
Smears from bacterial osteomyelitis are dominated by abundant neutrophils but also contain other inflammatory cells and macrophages. The aspirate may look like pus from any other type of non-specific acute inflammatory process (Fig. 16.7). Culture of the aspirated material is the most valuable part of the procedure if an infectious lesion is suspected. Clusters of epithelioid histiocytes with characteristic banana- or bean-shaped nuclei and indistinct cell borders and granular (caseous) necrotic material provoke a strong suspicion of tuberculosis (Fig. 16.8). In our experience, Langhans-type giant cells are difficult to find. Again, the need for cultural evidence is emphasised.
Metastatic carcinoma
As with sites such as lymph nodes, diagnosis is usually easy, especially if the histologic type of the primary tumor is known and sections are available for comparison with the smears. Descriptions of specific types of metastatic neoplasms are not included here; their cytological appearances have generally been discussed elsewhere.
Problems in differential diagnosis
Solitary plasmacytoma; myeloma
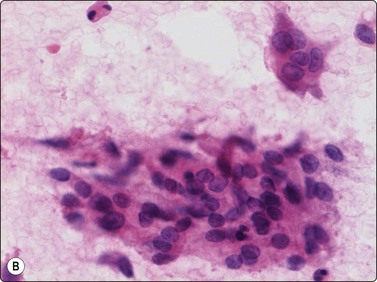
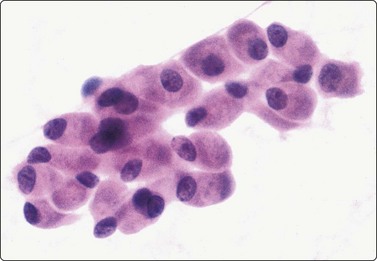
The diagnosis is obvious if there is a uniformly dispersed population of plasma cells including multinucleate and pleomorphic forms. The eccentric nucleus, with its speckled or clock-face chromatin, and the abundant amphophilic cytoplasm make recognition easy. A pale or clear cytoplasmic zone near the nucleus is another distinctive cytologic feature (Fig. 16.9).
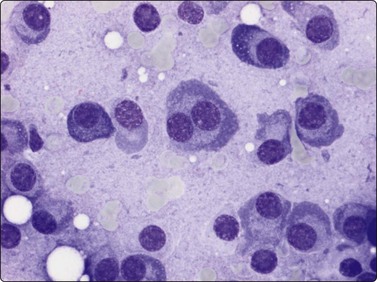
Pure population of atypical slightly pleomorphic plasma cells; the eccentric nucleus and pale or clear cytoplasmic zone near the nucleus are distinctive cytologic features (MGG, HP).
Problems in differential diagnosis
Neoplastic plasma cells are monoclonal (express monotypic immunoglobulin). This feature is possible to demonstrate in cell block or cytospin preparations. Neoplastic plasma cells, including most anaplastic forms, express CD138 and CD79A.
Malignant lymphoma
Focal bone involvement, usually in form of lytic lesions and most commonly involving the spine, pelvis and the long bones, is uncommon., Primary non-Hodgkin’s lymphoma comprises about 3–7% of the primary malignant bone tumors. Primary Hodgkin’s lymphoma in bone is extremely rare. Most primary non-Hodgkin’s lymphomas are diffuse large cell B-cell lymphomas. Lymphoplasmacytic lymphoma, anaplastic large cell lymphoma and precursor lymphoma (lymphoblastic lymphoma) have also been reported; the two last mentioned may occur in children and adolescents. Cytological criteria for diagnosis are the same as in other sites (see Chapter 5). Case series of primary non-Hodgkin’s lymphoma of bone have been presented.13,14 The important differential diagnosis between precursor lymphoma and classical Ewing’s sarcoma is discussed on page 424.
Langerhans cell histiocytosis (histiocytosis X, eosinophilic granuloma)
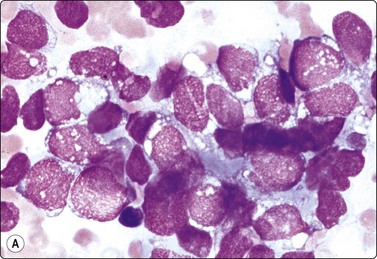
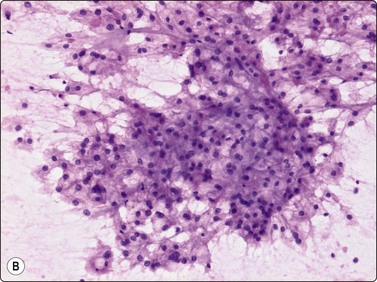
Langerhans cell histiocytosis presents as lytic and often well-defined lesions. Most cases are seen in children. The lesions may be solitary (most cases) or multiple. The long bones (femur and humerus) and the skull are the most common sites in children, and the pelvic bones and ribs in adults. The cytological pattern of Langerhans cell histiocytosis is fairly characteristic and may be diagnostic. The typical histiocytes have moderately larger and paler nuclei than those seen in common inflammatory processes. Generally reniform, the nuclei have a distinct irregular and folded outline (Fig. 16.10A). Coffee-bean nuclei has been reported to be typical of this lesion (Fig. 16.10B). The chromatin is entirely bland and nucleoli small. The cytoplasm is abundant and pale and has fairly well-defined borders. It is often vacuolated. In exceptional cases the Langerhans cells may show signs of phagocytosis. Multinucleated cells of similar type are commonly present. These can be quite large and may resemble osteoclasts in wet-fixed smears.
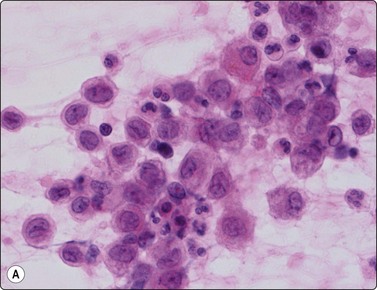
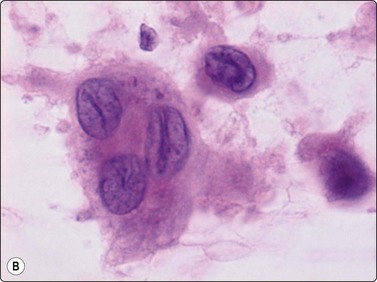
Fig. 16.10 Langerhans cell histiocytosis (eosinophilic granuloma)
(A) Lytic bone lesion, many histiocytes with irregular reniform pale nuclei, some eosinophils (H&E, IP); (B) Coffee-bean nuclei have been reported to be typical of this lesion (H&E, HP, Oil)
Large number of histiocytes can occasionally be found in aspirates of chronic osteomyelitis, but these are of the common, smaller type seen in the usual inflammatory processes and are mixed with neutrophils, lymphocytes and plasma cells.
The Langerhans cell histiocytes express S-100 protein and CD1-antigen (Fig. 16.11). According to the Histiocyte Society the definitive diagnosis of Langerhans cell histiocytosis is based on the demonstration of Birbeck granules by electron microscopy or positivity for CD1 antigen.15 The examination of conventionally stained material gives a presumptive diagnosis. The cytological features of Langerhans cell histiocytosis has been recorded in several series.16-18
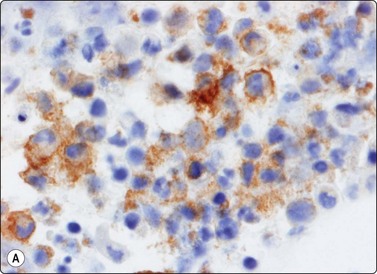
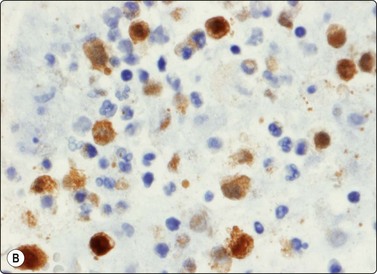
Fig. 16.11 Langerhans cell histiocytosis (eosinophilic granuloma)
The Langerhans cell histiocytes express (A) S-100 protein and (B) CD1-antigen (Cell block, immunoperoxidase).
The soft tissue deposits of generalised Langerhans cell histiocytosis (Letterer-Siwe disease) generally consist of a more pure population of abnormal histiocytes and have a less inflammatory appearance.
Giant cell tumor of bone
The giant cells have numerous (about 20–50) uniform nuclei. The spindle cells are mainly cohesive but single cells are always seen. The giant cells are typically attached to the periphery of clusters of spindle cells (Fig. 16.12A). Strands of collagen or basement membrane material, and endothelial cells are discernible in cell clusters and tissue fragments. The cells have a moderate amount of dense, amphophilic cytoplasm, which is often vacuolated, and well-defined borders. The nuclei vary only moderately in size and shape. They are ovoid, the chromatin is bland and nucleoli small (Fig. 16.12B). Malignancy (giant cell-rich osteosarcoma) should be suspected if spindled and multinucleated cells show nuclear pleomorphism and irregular chromatin, and if mitoses are plentiful. The differential diagnosis of other benign bone lesions with many osteoclastic giant cells such as aneurysmal bone cyst, osteoblastoma, brown tumor of hyperparathyroidism and reparative granuloma of jaw and small bones is difficult. Detailed knowledge of clinical and radiological findings is essential for a correct evaluation. The cytological appearance of giant cell tumor is recorded in a total of 38 cases in three series.3,19,20
Chondroma
Single cells are uncommon in smears from chondroma. The chondromyxoid ground substance is usually abundant and very conspicuous in MGG (Fig. 16.13A). It has a finely fibrillar texture. It is less obvious in wet-fixed smears where it is seen as a hyaline, pale violet material with H&E (Fig. 16.13B), and even paler with Pap. The morphology of the tumor cells is best studied in wet-fixed smears, as the cells are obscured by the intensely stained ground substance in MGG smears. The cellularity of the fragments is generally low and the tumor cells are uniform and rounded with a well-defined cytoplasm, rounded nuclei and one or two nucleoli. Binucleate cells are almost never seen. Chondromas of the small peripheral bones, however, often show marked pleomorphism and binucleated cells (Fig. 16.14).
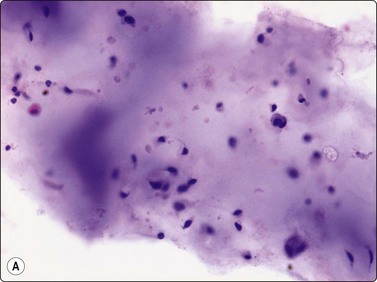
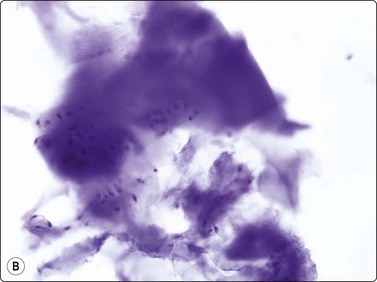
(A) Widely separated chondrocytes with small nuclei; The chondromyxoid ground substance is usually abundant and very conspicuous in MGG; (B) Chondroid background substance in wet-fixed smears appears as a hyaline, pale violet material in H&E (A, MGG, IP; B, H&E, IP).
As can be expected from histopathological experience, it can be difficult or impossible in FNB smears to distinguish between chondroma and low-grade (grade 1) malignant chondrosarcoma. The combined evaluation of clinical data, radiological findings and cytology is emphasised. The cytology of chondroma has been reported in a few publications21,22.
Chondroblastoma
Chondroblastoma is a rare tumor, and most cases are found in children and adolescents. The cytology has been investigated in four series.21-25 Our experience is limited to a few cases. The double cell population and the fragments of chondroid material are the clues to the diagnosis. According to the series of 12 cases described by Fanning et al.,23 the cells resembling chondroblasts are typical. They are mononuclear and rounded with well-demarcated cytoplasm and rounded lobulated or reniform nuclei. Nuclei are centrally located, showing slight anisokaryosis. One feature often described is longitudinal grooves in nuclei. The chondroblast-like nuclei are either single or embedded in the chondroid fragments (Fig. 16.15).
Chondromyxoid fibroma
Few cases of FNB of chondromyxoid fibroma have been reported.21,26,27 We have experience of a few tumors. Smears show a mixture of chondroid fragments, fibroblast-like spindled or stellate cells and osteoclast-like giant cells embedded in myxoid material (Fig. 16.16A). Rounded chondroblast-like cells in lacunar spaces may be seen in the chondroid fragments. The spindle-shaped cells may show some nuclear pleomorphism (Fig. 16.16B) and prominent nucleoli, and binucleated chondroblasts are not uncommon. Chondromyxoid fibroma has been misdiagnosed as chondrosarcoma.
Osteoblastoma
Only a few cases of osteoblastoma with cytology have been published.21,28 Our experience is restricted to a few cases. A mixed cell population has been recorded; the majority of cells resemble osteoblasts with eccentric nuclei and a cytoplasmic ‘Hof’. A moderate anisokaryosis and the presence of binucleated cells is not uncommon. A blue–red or pink (MGG) background matrix was seen between the cells in cell groups (Fig. 16.17A). Osteoclast-like giant cells and a few groups of spindle cells with fusiform nuclei were present in all cases recorded (Fig. 16.17B). The differential diagnosis is osteosarcoma versus aggressive (epithelioid) osteoblastoma,29 a rare variant not defined in FNB material.
Primary malignant tumors of bone
Osteosarcoma (conventional intramedullary osteosarcoma)
Criteria for diagnosis
The experience of FNB of osteosarcoma is mainly related to the conventional, high-grade intramedullary osteosarcoma (the most common type) and to the rare high-grade surface osteosarcoma. The rare periosteal osteosarcoma has not been characterised and, due to the often prominent bone formation, this osteosarcoma variant is not suitable for FNB.
The criteria listed above apply to the osteoblastic type. Smears contain both dissociated neoplastic cells and cell clusters. Osteoid is seen as faintly eosinophilic clumps in H&E, bright red or pink in MGG, amorphous or finely fibrillar material in the background (infrequent) or as thin intercellular strand within cell clusters (Fig. 16.18A). Macronucleoli may be present (Fig. 16.18B) and malignant giant cells with pleomorphic nuclei are commonly seen (Fig. 16.18C). Benign osteoclast-like giant cells may be present, especially in giant cell-rich osteosarcoma.
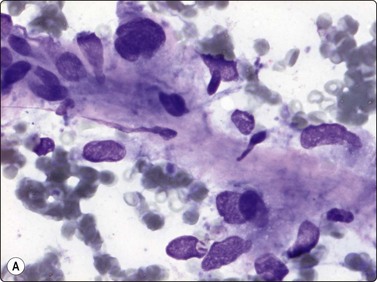
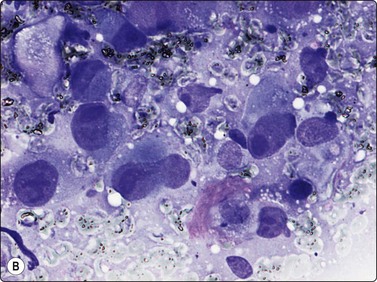
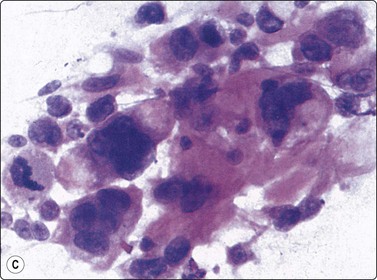
(A) Small cluster of pleomorphic sarcoma cells; strands of intercellular pink material consistent with osteoid (MGG, HP); (B) Malignant cells with macronucleoli are commonly seen in smears from high-grade osteosarcoma (MGG, HP); (C) Loose cluster of pleomorphic, often multinucleate cells; the fragments of pink amorphous material may represent osteoid; note mitotic figure (H&E, HP).
The chondroblastic osteosarcoma may be difficult to distinguish from a high-grade malignant chondrosarcoma (Fig. 16.19) and in the fibroblastic osteosarcoma; cellular pleomorphism is less marked in the spindle-shaped tumor cells (Fig. 16.20). Osteoid between tumor cells is uncommon. The tumor cell population in the rare small cell osteosarcoma resembles cells of conventional (classic) Ewing’s sarcoma. In another rare variant, parosteal osteosarcoma, the smears are dominated by fragment of hyaline cartilage and moderately atypical, spindly cells.
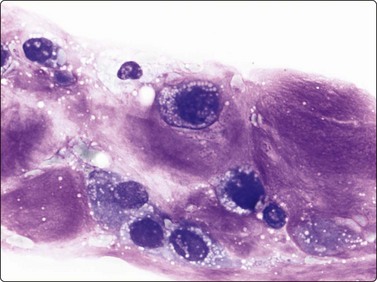
Fig. 16.19 Osteosarcoma (chondroblastic)
Malignant cells embedded in the abundant chondroid matrix may be difficult to distinguish from a high-grade malignant chondrosarcoma (MGG, HP).
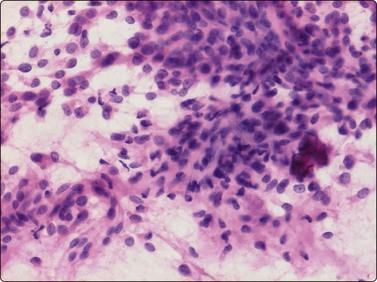
Fig. 16.20 Osteosarcoma (fibroblastic)
Smears from a patient with known Paget’s disease who developed a large mass in the left hip region with extensive bone destruction; moderately pleomorphic spindle cell sarcoma without distinctive features (H&E, IP).
The presence of cytoplasmic alkaline phosphatase (present in all variants of osteosarcoma) is an important feature in the differential diagnosis (metastatic anaplastic carcinoma, melanoma and anaplastic large cell lymphoma), as is the expression of osteonectin or/and osteocalcin in osteosarcoma cells (Fig. 16.21) The vast majority of conventional intramedullary high-grade osteosarcoma are non-diploid, and this feature is an important diagnostic sign in the distinction from lesions featuring reactive osteoblasts, e.g. fracture callus, reactive periosteitis and pseudomalignant myositis ossificans. The cytology of osteosarcoma in FNB smears has been investigated in several large series30,31,32,33.
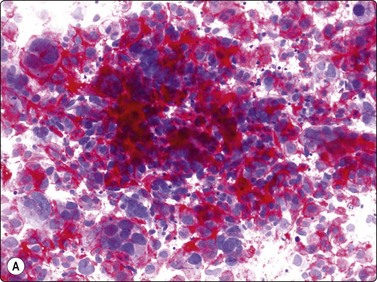
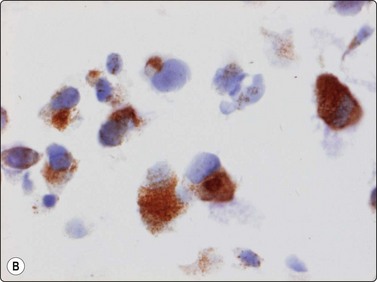
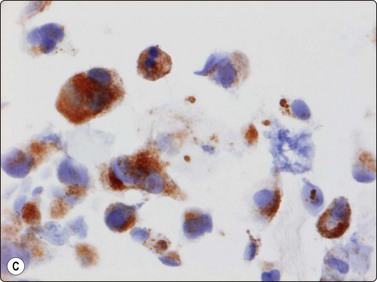
(A) Strong positive cytoplasmic staining for alkaline phosphatase and expression of (B) osteonectin or/and (C) osteocalcin in tumor cells are important features of osteosarcoma in the differential diagnosis (metastatic anaplastic carcinoma, melanoma and anaplastic large cell lymphoma) (A, alkaline phosphatase staining, HP; B, osteonectin, immunoperoxidase; C, osteocalcin, immunoperoxidase).
Chondrosarcoma
Criteria for diagnosis
The chondromyxoid ground substance is usually as abundant in low-grade tumors as in chondroma (Fig. 16.22). The neoplastic cells are best studied in wet-fixed preparations. Cellularity is variable in low-grade chondrosarcoma. This is important in the distinction from chordoma. A rich yield with plenty of tissue fragments demonstrating the variable cell content makes the diagnosis easier (Fig. 16.23). The tumor cells have a well-defined cytoplasm and rounded nuclei with one or two nucleoli. Binucleate cells are present and nuclear pleomorphism is of moderate degree (Fig. 16.24).
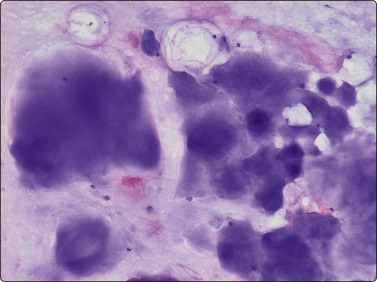
Fig. 16.22 Chondrosarcoma, well differentiated
The chondromyxoid ground substance is usually as abundant in low-grade tumors, as in chondroma (MGG, IP).
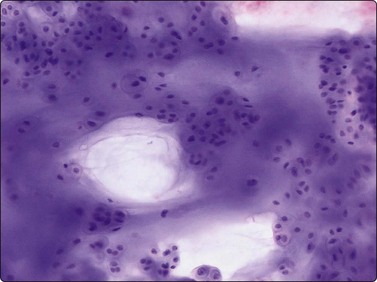
Fig. 16.23 Chondrosarcoma, well differentiated
A rich yield with plenty of tissue fragments demonstrating the variable cell content makes the diagnosis easier (MGG, IP).
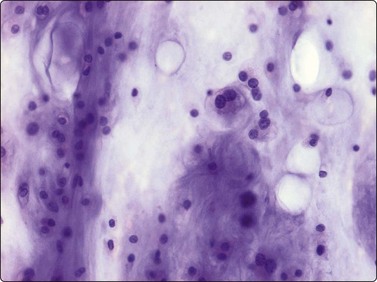
Fig. 16.24 Chondrosarcoma, well differentiated
Increased number of cells with moderately enlarged and irregular nuclei; some binucleate forms (MGG, HP).
Single cells dominate in high-grade malignant tumors, cellular and nuclear pleomorphism is prominent and mitoses are present. Often abundant myxoid background matrix is present, while fragments of hyaline cartilage are few. As stated above, it is difficult or impossible to distinguish between chondroma and some low-grade (grade 1) chondrosarcomas in FNB smears. Grade 3 chondrosarcomas may be difficult to distinguish from chondroblastic osteosarcoma and from metastatic poorly differentiated epithelial tumors if cartilaginous fragments are absent. Chordoma should be considered in the differential diagnosis in spinal or sacral tumors (see below). Dedifferentiated chondrosarcoma is yet another pitfall. This distinct variant has two components: a low-grade chondrosarcoma (or chondroma) and a high-grade sarcoma which is not a grade 3 chondrosarcoma. The clue to the cytological diagnosis is the presence of a low-grade cartilaginous tumor and a high-grade sarcoma (most often pleomorphic sarcoma of MFH type or rarely osteosarcoma or rhabdomyosarcoma) in the same sample (Fig. 16.25). Inadequate sampling may result in misinterpretation.34
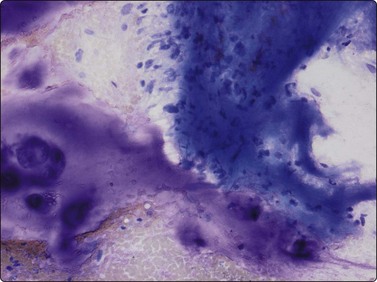
Fig. 16.25 Chondrosarcoma, dedifferentiated
The clue to the cytological diagnosis is the presence of a low-grade cartilaginous tumor and a high-grade sarcoma (MGG, IP).
Two rare chondrosarcoma subtypes, clear-cell chondrosarcoma and mesenchymal chondrosarcoma are occasionally the target for FNB. The typical tumor cell in clear cell chondrosarcoma, a low-grade tumor, is rather large with a clear or vacuolated abundant cytoplasm and a central or paracentral nucleus. The cells are seen singly or in small groups in a myxoid background matrix (Fig. 16.26). A few cases of clear cell chondrosarcoma have been described.21 FNB smears of mesenchymal chondrosarcoma are composed of small tumor cells with rounded or ovoid nuclei and a sparse cytoplasm. The cells are often embedded in a myxoid matrix (red–violett in MGG) (Fig. 16.27). The most important differential diagnoses are other small cell malignant tumors such as small cell osteosarcoma and conventional Ewing’s sarcoma. Single reports of the cytology of mesenchymal chondrosarcoma in FNB have been published.21
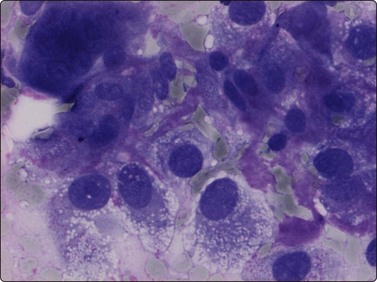
Fig. 16.26 Clear-cell chondrosarcoma
Rather large cells with a clear or vacuolated abundant cytoplasm and a central or paracentral nucleus (MGG, HP).
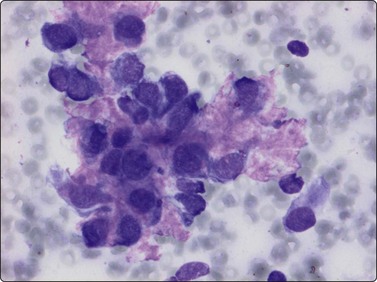
Fig. 16.27 Mesenchymal chondrosarcoma
The small tumor cells with rounded or ovoid nuclei and a sparse cytoplasm are often embedded in a red–violet matrix (MGG, HP).
DNA image ploidy analysis is a valuable adjunct in the diagnosis; a non-diploid histogram is a sign of a high-grade tumor, irrespective of yield. The cytological findings in smears of chondrosarcoma have been described in several series.3,21,35,36
Chordoma
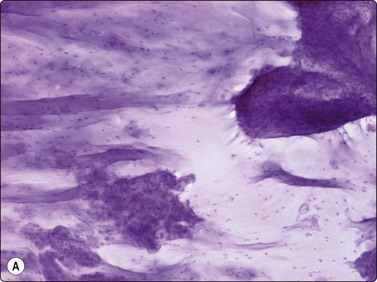
The characteristic findings are the abundant background of myxoid ground substance and the large, physaliphorous cells with abundant pale, vacuolated, bubbly cytoplasm and well-defined cell borders. The myxoid matrix, often fibrillar, intensely purple in MGG smears, pale pink in H&E, forms a network encircling individual tumor cells, cell clusters or fragments (Fig. 16.28). The physaliphorous cells have one, sometimes two, rounded nuclei of moderate size, a bland chromatin and small nucleoli. Moderate anisokaryosis is common (Fig. 16.29A). Some tumors show clusters of markedly pleomorphic cells with prominent nucleoli, and multinucleated tumor giant cells may be present (Fig. 16.29B). There are also clusters of small to medium-sized, non-characteristic or epithelial-like cells with rounded nuclei. The cytoplasm of these cells may be vacuolated; some cells have one large vacuole pushing the nucleus to the periphery and may resemble signet ring cells (Fig. 16.30A). The main differential diagnosis is chondrosarcoma. If only wet-fixed smears are available, metastatic clear cell carcinoma, especially renal cell carcinoma or mucus-producing adenocarcinoma can also cause differential diagnostic difficulties. The abundant ground substance may be inconspicuous in H&E or Pap smears and the epithelial-like tumor cells may give a false impression of an epithelial neoplasm (Fig. 16.30B). Typical physaliphorous cells are never encountered in chondrosarcoma and the network of myxoid matrix encircling individual cells is not a feature of either chondrosarcoma or metastatic carcinoma. A chordoma originating in the cervical spine does not always present as a midline tumor and, if information on the radiologic finding is not available, may be interpreted as a malignant myxoid soft tissue tumor. Chordomas typically express S-100 protein and low molecular weight cytokeratins. The cytological appearance of chordoma in FNB has been evaluated in rather large series.37-39
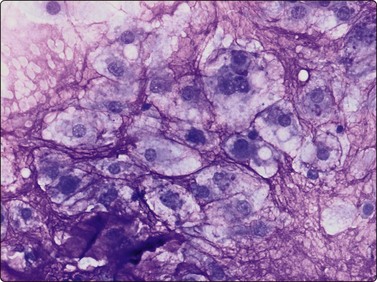
A network of intensely purple myxoid material encircles individual tumor cells with abundant pale bubbly cytoplasm (MGG, HP).
Ewing’s family tumors
Several studies by various investigators have provided evidence that conventional Ewing’s sarcoma (ES), atypical Ewing’s sarcoma, the so called Askin tumor, neuroepithelioma and primitive neuroectodermal tumor (PNET) belong to the same family of neuroectodermal tumors.40 All tumors in this family share the same cytogenetic aberration, t(11;22)(q24;q12), resulting in the EWS/FLI-1 transcript. Furthermore, they express the same phenotype, CD99 (the antibody to the MIC2 gene) and FLI-1. The different variants have a variable morphology and variable expression of neuroectodermal markers.
Criteria for diagnosis (conventional Ewing’s sarcoma)
The cytological appearance of conventional ES is distinctive. Smears are generally highly cellular and are composed of both single cells and groups of loosely cohesive cells. The cells are fragile and naked nuclei as well as a faint gray–blue (MGG) background of detached cytoplasm are commonly seen. There is a characteristic mixture of two types of cells. One has abundant pale cytoplasm with vacuoles or large clear spaces, rounded or ovoid nuclei with finely granular chromatin and 1–3 small nucleoli (‘large light cells’). The other has scanty cytoplasm and irregular nuclei with dense chromatin (’small dark cells’). The two types of cells are most clearly distinguished within groups or clusters of cells, the small dark cells are interspersed, often as small molded groups, between large light cells (Fig. 16.31A). Rosette-like structures without a fibrillar center are occasionally present (Fig. 16.31B,C). The cytoplasmic vacuoles or clear spaces correspond to large deposits of glycogen (Fig. 16.31A).
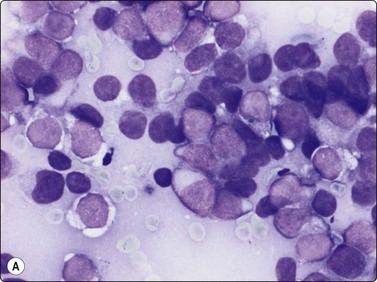
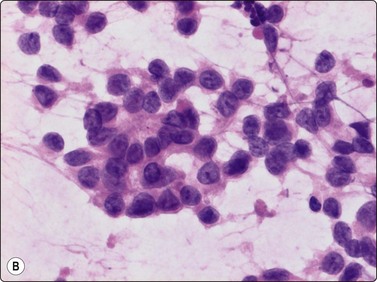
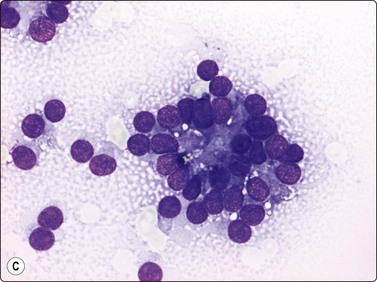
(A) A mixture of cells with larger pale-staining nuclei and cells with smaller and darker nuclei; note the cytoplasmic vacuoles and clear spaces in the pale cells (MGG, HP); (B,C) Rosette-like structure; nuclear chromatin and nucleoli more clearly seen in H&E than in MGG (A, H&E, HP; B, MGG, HP).
In atypical ES and PNET the cellular and nuclear atypia is more marked than in conventional ES. Rosette-like structures are more common and the distinction between large light and small dark cells less obvious, especially in PNET, and cells with thin cytoplasmic processes as well as rhabdomyoblast-like cells are present (Fig. 16.32).
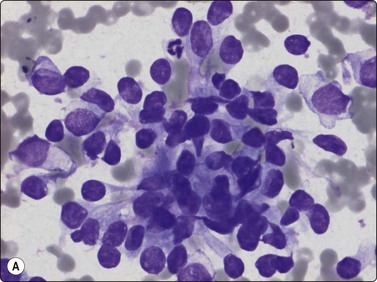
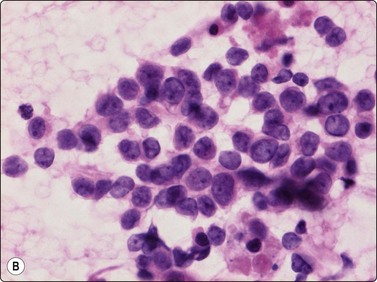
Fig. 16.32 ES/PNET family of tumors
Distinction between large light and small dark cells less obvious in PNET than in conventional Ewing’s, and cells with thin cytoplasmic processes (A) and rhabdomyoblast-like cells (B) are present (A, MGG, HP; B, H&E, HP).
Distinction from other malignant small cell tumors such as alveolar rhabdomyosarcoma, primitive neuroblastoma, precursor B- and T-cell lymphoma are important differential diagnoses and is relatively easy in typical cases of conventional ES.
In the precursor lymphomas, nuclei generally are of variable size with irregular or folded contours and the cytoplasm is narrow and pale without vacuoles, but often demonstrates a small clear spot close to the nucleus. The rhabdomyoblast-like cells in alveolar rhabdomyosarcoma are rather distinctive with eccentric nuclei and a dense eosinophilic cytoplasm on wet-fixed smears. The long, thin cytoplasmic processes, often connecting cells, in neuroblastoma are not a feature of conventional ES. However, the differential diagnosis between atypical ES and PNET and poorly differentiated Ewing’s sarcoma-like synovial sarcoma and the above mentioned tumors is much more difficult in routinely stained smears. Cytogenetic analysis is the most valuable diagnostic adjunct.41,42 Immunocytochemistry, including CD99 and FLI-1 are also of value in spite of the fact that both these antibodies are reported to be positive in other malignant small cell tumors (Fig.16.33).
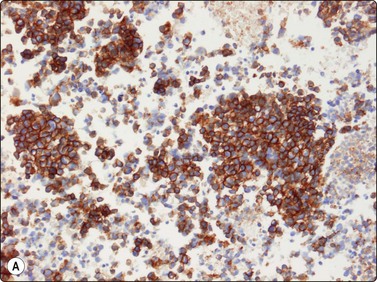
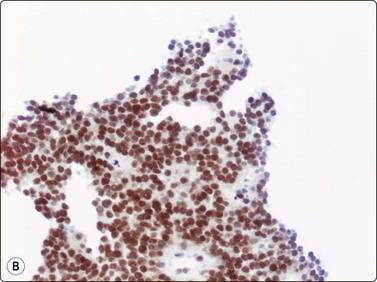
Fig. 16.33 ES/PNET family of tumors
Immunocytochemistry, mainly (A) CD99 and (B) FLI-1 are of diagnostic value (A, B, cell block, immunoperoxidase).
The cytology of Ewing’s family tumors in FNB smears has been thoroughly investigated in several series.43-47
1 Coley BL, Sharp GS, Ellis EB. Diagnosis of bone tumours by aspiration. Am J Surg. 1931;13:214-224.
2 Ahlström KH, Åström KC. CT guided bone biopsy performed by means of a coaxial biopsy system with an eccentric drill. Radiology. 1993;188:549-552.
3 Åkerman M, Domanski HA. Fine needle aspiration (FNA) of bone tumors with special emphasis on the definitive treatment of primary malignant tumors based on FNA. Curr Diagn Pathol. 1998;5:82-92.
4 Layfield LJ. Cytologic diagnosis of osseous lesions: a review with emphasis on the diagnosis of primary neoplasms of bone. Diagn Cytopathol. 2009;37:299-310.
5 Åkerman M, Domanski HA, Jonsson K. Fine needle aspiration of bone tumours. Monographs in Clinical Cytology. vol 19. Kargel, Basel, 2010.
6 Kreicbergs A, Bauer HCF, Brosjö O, et al. Cytological diagnosis of bone tumours. J Bone Joint Surg (Br). 1996;78B:258-263.
7 Bommer K, Ramzay I, Mody D. Fine-needle aspiration biopsy in the diagnosis and management of bone lesions. Cancer (Cancer Cytopathol). 1997;81:148-156.
8 Söderlund V, Skoog L, Kreicbergs A. Combined radiology and cytology in the diagnosis of bone lesions: a retrospective study of 370 cases. Acta Orthop Scand. 2004;75:492-499.
9 Wahane RN, Lele VR, Bobhate SK. Fine needle aspiration cytology of bone tumors. Acta Cytol. 2007;51:711-720.
10 McLaughlin RE, Miller WR, Miller CW. Quadriparesis after needle aspiration of the cervical spine. J Bone Joint Surg. 1976:1167-1168.
11 Saad RS, Clary KM, Silverman JF, et al. Fine needle aspiration biopsy of vertebral lesions. Acta Cytol. 2004;48:39-46.
12 Akthar I, Flowers R, Siddiqi A, et al. Fine needle aspiration biopsy of vertebral and paravertebral lesions: retrospective study of 124 cases. Acta Cytol. 2006:364-371.
13 Söderlund V, Skoog L, Tani E, et al. Diagnosis of skeletal lymphoma and myeloma by radiology and fine-needle aspiration cytology. Cytopathology. 2001;12:157-167.
14 Lin F, Staerkel G, Fanning CV. Cytrodiagnosis of primary lymphoma of bone on fine-needle aspiration cytology specimens:review of 25 cases. Diagn Cytopathol. 2003;28:205-211.
15 Malone M. The histiocytoses childhood. Histopathol. 1991;19:105-119.
16 Shabb N, Fanning CV, Carrasco C. Diagnosis of eosinophilic granuloma by fine needle aspiration with concurrent institution of therapy. Diagn Cytopathol. 1993;9:3-12.
17 Pohar-Marinsek Z, Us-Krasovec M. Cytomorphology of Langerhans cell histiocytosis. Acta Cytol. 1996;40:1257-1264.
18 Kilpatrick SE. Fine needle aspiration biopsy of Langerhans cell histiocytosis: are ancillary studies necessary for a definitive diagnosis? Acta Cytol. 1998;42:820-823.
19 Sneige N, Ayala A, Carrasco H, et al. Giant cell tumour of bone. A cytologic study of 24 cases. Diagn Cytopathol. 1985;1:111-117.
20 Vetrani A, Fulciniti F, Boschi R, et al. Fine needle aspiration biopsy of giant cell tumour of bone. Acta Cytol. 1990;34:863-867.
21 Waalas l, Kindblom L-G, Gunterberg B, et al. Light and electro-microscopic examination of fine needle aspiration in the preoperative diagnosis of cartigalinous tumours. Diagn Cytopathol. 1990;6:396-408.
22 Dhawan SB, Aggarwal R, Mohan H, et al. Cytodiagnosis of enchondroma. Cytopathology. 2003;14:157-159.
23 Fanning CV, Sneige NS, Carrasco CH, et al. Fine needle aspiration’s cytology of chondroblastoma of bone. Cancer. 1990;65:1847-1863.
24 Pohar-Marinsek Z, Us-Krasovec M, Lamovee J. Chondroblastoma in fine needle aspirates. Acta Cytol. 1992;36:367-370.
25 Kilpatrick SE, Pike EJ, Geisinger KR, et al. Chondroblastoma of bone: use of fine-needle aspiration biopsy and potential diagnostic pitfalls. Diagn Cytopathol. 1997;16:65-71.
26 Layfield LJ, Ferreiro JA. Fine-needle aspiration cytology of chondromyxoid fibroma: a case report. Diagn Cytopathol. 1988;4:148-151.
27 Gupta S, Dev G, Marya S. Chondromyxoid fibroma: a fine-needle aspiration diagnosis. Diagn Cytopathol. 1993;9:63-65.
28 Rhode MG, Lucas DR, Krueger CH, et al. Fine-needle aspiration of spinal osteoblastoma in a patient with lymfangiomatosis. Diagn Cytopathol. 2006;34:295-297.
29 Bertoni F, Bacchini P, Donati D, et al. Osteoblastoma-like osteosarcoma. The Rizzoli Institute experience. Mod Pathol. 1993;6:707-716.
30 White WA, Fanning CV, Ayala AG, et al. Osteosarcoma and the role of fine-needle aspiration: a study of 51 cases. Cancer. 1988;62:1238-1246.
31 Dodd LG, Scully SR, Cothran RL, et al. Utility of fine-needle aspiration in the diagnosis of primary osteosarcoma. Diagn Cytopathol. 2002;27:350-353.
32 Domanski HA, Åkerman M. Fine-needle aspiration of primary osteosarcoma: a cytological-histological study. Diagn Cytopathol. 2005;32:269-275.
33 Klijanienko J, Caillaud J-M, Orbach D, et al. Cyto-histological correlations in primary, recurrent and metastatic bone and soft tissue osteosarcoma. Diagn Cytopathol. 2007;35:270-275.
34 Rinas AC, Ward WG, Kilpatrick SE. Potential sampling error in fine needle aspiration biopsy of dedifferentiated chondrosarcoma: a report of 4 cases. Acta Cytol. 2005;49:554-559.
35 Lerma E, Tani E, Brosjö O, et al. Diagnosis and grading of chondrosarcoma on FNB biopsy material. Diagn Cytopathol. 2003;28:13-17.
36 Dodd LG. Fine needle aspiration of chondrosarcoma. Diagn Cytopathol. 2006;34:413-418.
37 Finley JL, Silverman JF, Dabbs DJ, et al. Chordoma. Diagnosis by fine-needle aspiration with histologic, immunocytochemical, and ultrastructural confirmation. Diagn Cytopathol. 1998;2:330-337.
38 Crapanzano JP, Ali SZ, Ginsberg MS, et al. Chordoma: a cytologic study with histologic and radiologic correlation. Cancer. 2001;93:40-51.
39 Kay PA, Nasciemento AG, Unni KK, et al. Chordoma: cytomorphologic findings in 14 cases diagnosed by fine needle aspiration. Acta Cytol. 2003;47:202-208.
40 Delattre O, Zuchman J, Melot T, et al. The Ewing family of tumours: a subgroup of small-round cell tumors defined by specific chimeric transcripts. N Engl Med J. 1994;331:294-299.
41 Fröstad B. Fine needle aspiration cytology in diagnosis and management of childhood small round cell tumours. Thesis: Stockholm; 2000.
42 Udaykumar AM, Sundareshan TS, Goud TM, et al. Cytogenetic characterisation of Ewing tumors using fine needle aspiration samples, a 10-year experience and review of the literature. Cancer Genet Cytogenet. 2001;127:42-48.
43 Dahl I, Åkerman M, Angervall L. Ewing’s sarcoma of bone. A correlative cytological and histological study of 14 cases. Acta Pathol Microbiol Immunol Scand (A). 1986;94:363-369.
44 Silverman JS, Berns LA, Holbrook CT, et al. Fine needle aspiration cytology of primitive neuroectodermal tumors: a report of these cases. Acta Cytol. 1992;36:541-550.
45 Renshaw AA, Pares-Atayde AR, Fletcher JA, et al. Cytology of typical and atypical Ewing’s sarcoma/PNET. Am J Surg Pathol. 1996;106:620-624.
46 Mondal A, Misra DK. Ewing’s sarcoma of bone: a study of 71 cases by fine needle aspiration cytology. J Indian Med Ass. 1996;94:135-137.
47 Sahu K, Pai RR, Khadikar UN. Fine needle aspiration cytology of the Ewing’s sarcoma family of tumors. Acta Cytol. 2000;44:332-336.