CHAPTER 2 The techniques of FNA cytology
Basic techniques
Svante R. Orell
The success of fine needle aspiration cytology (FNAC) depends to a high degree on perfecting the technique of sampling and preparation of samples. Palpation skills learnt through practice and experience, judiciously complemented by radiological image guidance when appropriate, are essential to obtain representative samples. The choice of needles, the use or not of aspiration, and the manipulation of the needle within the target relative to the type of tissue decide the adequacy of samples. Finally, correct smearing, fixation and staining of samples is critical to assure optimal preservation and presentation of cells and non-cellular components on which a confident diagnosis can be based.
Consequently, the cytopathologist should be in a position to control and give advice on sampling and preparation techniques, directly or indirectly, to achieve a high proportion of satisfactory and diagnostic biopsies. Close cooperation with the referring clinician and radiologist is essential.
Indications for fine needle biopsy (FNB) of various organs and tissues are explained in detail in the following chapters. Meanwhile, some general principles apply. To be suitable for FNB, the disease process must be localized and clearly defined by clinical examination or radiological imaging. FNB may be tried in some diffuse processes with the understanding that a negative result has little value. The principles of screening by exfoliative cytology are obviously completely different.
Although severe complications are rare1,2 the possible benefits of a cytological diagnosis should be weighed against risks and patient discomfort. Risk factors such as age, coagulation disorders, respiratory failure, etc. should be taken into account. FNB of superficial lesions can safely be carried out as an office procedure. Biopsies of most deep sites (transpleural, transperitoneal, etc.) are better performed in hospital so that patients can be sedated if necessary and kept under observation for a few hours after the procedure. The pathologist should be consulted before the procedure to give advice on feasibility, the likely informative value of the test, the need for and choice of ancillary techniques, etc.
Preparation for biopsy
Equipment
Needles
Standard disposable 27–22-gauge (0.4–0.7 mm), 30–50 mm long needles are suitable for superficial, palpable lesions. We use 25-gauge needles for most lesions, but increasingly 27 gauge for cell-rich and vascular tissues such as lymph nodes and thyroids, in children and in sensitive sites such as orbit, eyelids, genitals and intracutaneous lesions. Although the yield is a little less, samples are usually adequate and smear quality tends to be better due to less admixture with blood. The yield from fibrotic lesions in the breast and soft tissues is less predictable. Needles of 23–22 gauge are most often used, but thinner needles can sometimes be more efficient. Larger-bore needles may be required to obtain sufficient material for ancillary tests.
Twenty-two-gauge, 90-mm disposable lumbar puncture needles with trocar, or 22-gauge 150 or 250 mm Chiba needles are used for deep biopsies. They are sufficiently rigid and the trocar prevents contamination during the passage through surrounding tissues. Special long 23-gauge needles are supplied with the Franzén instrumentarium for biopsy of pelvic organs (Unimed, Lausanne, Switzerland).
If the purpose of the biopsy is to obtain a core of tissue for paraffin embedding and sectioning, a cutting core needle is used. A range of small-gauge cutting core needles is commercially available. Core needle biopsy (CNB) fragments allow the study of tissue architecture and provide more tissue for ancillary tests. However, CNB is more traumatic and more costly, has a slightly greater risk of complications, and must be processed in a laboratory, precluding an immediate, on-site assessment of adequacy or a preliminary diagnosis.3,4
Syringes and syringe holder
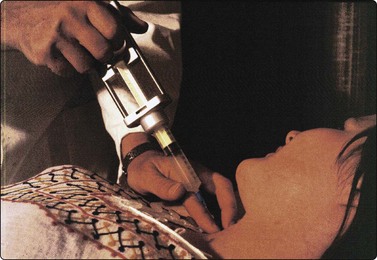
Standard disposable plastic syringes mounted in a syringe holder/pistol grip are suitable for conventional aspiration biopsy. The Cameco Syringe Pistol (Cameco AB, Taby, Sweden) is made to fit 10-cc plastic syringes. The holder leaves one hand free to fix and to feel the target, which allows better precision in placing the needle (Fig. 2.1).
Containers and slides
Small sterile containers with tight lids containing physiological saline or a transport medium such as Hank’s balanced salt solution should be at hand if a cell suspension or a cell block is needed, or to rinse needles and syringes. Special culture media may be required in certain instances.
Glass slides must be clean, dry and free of grease. Slides with frosted ends are convenient for immediate labeling. Coated or charged slides for better adhesion are recommended if smears are to be used for immunostaining.
Fixatives and stains
Smears are air-dried or wet-fixed. Routine wet fixation is either in 70–90% ethanol or using a commercial spray fixative. Carnoy’s fixative has the advantage of lysing red blood cells. Glutaraldehyde and 10% buffered formalin should be available if tissue fragments for EM or for paraffin embedding are obtained. Note that formalin must be kept in an airtight container since formalin fumes may adversely affect air-dried smears if stored together.
A set of Diff-Quik stains in suitable containers, and a lightweight portable microscope should be available at all times to allow immediate checking of smears for adequacy and an immediate preliminary diagnosis if required. Some pathologists may prefer rapid Papanicolaou staining.
Other
Skin disinfectant (pre-injection swabs), sterile dressings, local anesthetic, tongue depressors, a small electric torch and sterile scalpel blades (scrape smears from skin or mucus membranes) should also be available on the biopsy tray, tool box or trolley. Latex gloves and face masks may be required for safety reasons. A small hair dryer is a useful tool to dry smears rapidly for MGG/Diff-Quik staining.
Patient preparation
The procedure should be clearly explained to the patient to assure his/her consent and cooperation.5 A formal written consent may be required, at least for deep biopsies. The procedure is usually carried out with the patient supine on an examination couch with easy access from either side. A couch with stirrups is preferable for transrectal and transvaginal biopsy, and an examination chair with adjustable headrest for biopsy of lesions in the head and neck.
Simple skin disinfectant using prepacked swabs for injections is adequate for biopsy of superficial lesions. Preparations as for minor surgical procedures (surgical skin disinfectant, fenestrated sterile cloth, sterile gloves) are recommended for transpleural, transperitoneal and bone biopsies.
Anesthesia
Pre-biopsy sedation is rarely necessary. Atropine is recommended in preparation for transpleural biopsy to prevent the unlikely risk of vasovagal reflex. The biopsy may be coordinated with other procedures that require general anesthesia.
Local anesthesia is not often warranted in superficial biopsies. However, we recommend the injection of a local anesthetic in transpleural, transperitoneal and transperiosteal biopsies to prevent uncontrolled movements or jerks by the patient during the procedure and to make multiple passes more acceptable. A spray anesthetic can be used in biopsies of targets in the mouth, pharynx or other mucosal sites. An anesthetic ointment applied at least half an hour before the procedure is useful in children.
The biopsy procedure
Insertion of the needle
With frequent practice and growing experience, the operator acquires the ability to feel the consistency of the tissue through the needle. This helps considerably to position the needle accurately without technical aids. The ‘fingertip sensitivity’ is much greater when the needle is held directly without the interposition of a syringe and holder, as in the non-aspiration technique. A near vertical pathway tends to be less painful and allows better appreciation of depth; a tangential path is preferable in superficial skin lesions and for lesions in the chest wall.
The use of radiological imaging techniques to guide deep biopsies is described in Chapter 3. Ultrasonographic (US) guidance may be of value even in palpable lesions. It defines the lesion exactly, gives the optimal depth for biopsy, guides the needle to a solid portion of a complex lesion, and shows the relationship to other anatomical structures such as major vessels, pleura, etc.
FNB with aspiration (Figs. 2.1 and 2.2)
The aspiration technique is illustrated diagrammatically in Figure 2.2. The negative pressure does not tear cells from the tissue but merely holds the tissue against the sharp cutting edge of the needle, which scrapes or cuts softer tissue components along the track as the needle advances through the tissue.6 Highly cellular tissue components are softer and more friable than the supporting stroma and are selectively sampled. Fibrous stromal components are poorly represented, whereas myxoid stroma is more easily sampled. To increase the yield, the needle should be moved back and forth within the lesion with the negative pressure maintained, more vigorously in fibrous tissues with low cell content. Several passes may be necessary to sample a sufficient number of cells. In highly cellular and vascular tissues such as spleen, lymph nodes, liver and thyroid, a few rapid passes usually suffice. Additional passes mainly increase the amount of blood aspirated, causing dilution of the cellular component. Admixture with blood tends to be less if the needle is moved along the same track rather than in multiple directions. One should never wait to see material enter the hub of the needle, except when evacuating a cyst or an abscess. The ideal aspirate has a creamy consistency due to high cell content in a small amount of fluid and remains inside the needle.
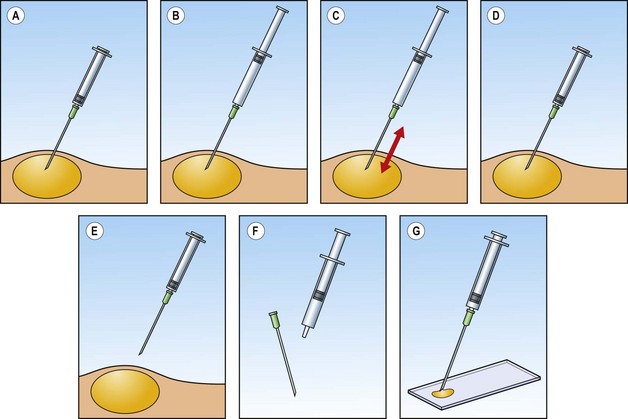
Diagrammatic stepwise illustration of biopsy procedure: (A) needle positioned within the target tissue; (B) plunger pulled to apply negative pressure; (C) needle moved back and forth inside target; (D) negative pressure released while needle remains in target tissue; (E) needle withdrawn; (F) needle detached and air drawn into syringe; (G) sample blown onto microscopy slide.
The negative pressure must be released before the needle is withdrawn. Even so, part of the aspirate is often drawn up into the hub of the needle (see below). A maintained negative pressure may draw the aspirate into the syringe, which must then be rinsed with fluid to recover the specimen. It can also cause contamination by material aspirated along the track during withdrawal of the needle. Aspiration of US gel in guided FNB of breast lesions (Chapter 7) is a good example.
Fine needle sampling without aspiration (Figs 2.3 and 2.4)
As mentioned, the negative pressure plays a relatively minor role compared to the scraping or cutting effect of the advancing oblique needle tip. Fine needle biopsy without aspiration was introduced by Zajdela in 1987.7 This technique is based on the observation that the capillary pressure in a fine needle is sufficient to keep the detached cells inside its lumen. A 27–23-gauge standard needle is held directly with the fingers, inserted into the target tissue, moved back and forth in several directions for a few seconds depending on the cellularity and the vascularity of the tissue, and is then withdrawn. Using this technique, the operator gets an excellent feel of the consistency of the tissues. This is a valuable piece of diagnostic information and improves precision when sampling small lesions. Admixture with blood is generally less than with aspiration. The technique is particularly well suited for biopsy of the thyroid and other vascular tissues. The cell yield may be smaller than with aspiration but not significantly so.8,9 We use sampling without aspiration routinely in superficial biopsies except in cystic lesions and in fibrotic paucicellular tumors in the breast and soft tissues. Aspiration with 22-gauge needles is used in most deep biopsies in order to obtain a maximum volume of cells with a minimum number of passes, in view of the frequent demand for ancillary tests. However, non-aspiration biopsy sometimes produces better samples in highly vascular lesions, for example in renal tumors.
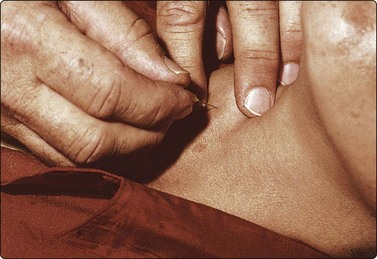
Fig. 2.3 FNB without aspiration (thyroid)
Needle held with finger tips, other hand feels and fixes the target.
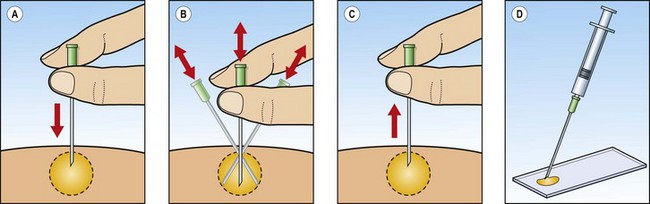
Fig. 2.4 FNB without aspiration
Diagrammatic stepwise illustration of biopsy procedure: (A) needle inserted into target tissue; (B) needle moved back and forth, varying the angle; (C) needle withdrawn; (D) needle attached to syringe and sample blown onto microscopy slide.
After biopsy of superficial lesions, pressure should be applied over the biopsy site to minimize bruising or post-biopsy hematoma. Patients should be kept under observation for a couple of hours after biopsy of deep sited lesions.
Failure to obtain a representative sample
Possible causes of failure to obtain a representative sample are illustrated diagrammatically in Figure 2.5. If the needle narrowly misses the target (Fig. 2.5B), only the adjacent tissues are sampled. An inflammatory reaction around the tumor may lead to an erroneous diagnosis. If the sample is derived from a central focus of necrosis, hemorrhage or cystic change (Fig. 2.5C), no diagnostic elements may be included. A dominant benign lesion like a cyst or lipoma can hide a small adjacent malignancy (Fig. 2.5D), for example in the breast or thyroid. Repeat biopsy of any remaining palpable abnormality after evacuation of a cyst is important. Finally, adequate samples may be difficult to obtain from desmoplastic tumor tissue in which cells are firmly held in a dense collagenous framework (Fig. 2.5E).
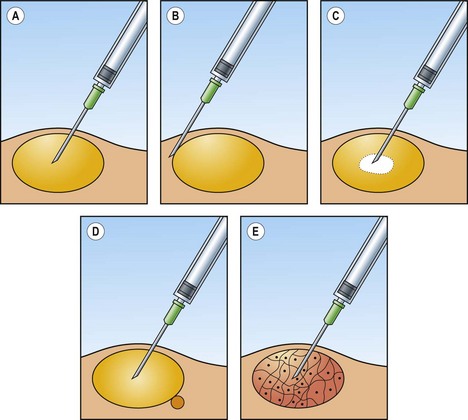
Fig. 2.5 Causes of unsatisfactory yield
(A) Needle well positioned within target tissue; (B) needle has missed the target tangentially; (C) needle in central cystic/necrotic/hemorrhagic area devoid of diagnostic cells; (D) needle sampling a dominant benign mass but missing a small adjacent malignant lesion; (E) fibrotic/desmoplastic target tissue giving a scant cell yield.
Processing the sample
The sample contained in the needle is expelled on to a clean and dry microscopy slide using air in a syringe. Care must be taken to avoid splashing. Not infrequently, the best part of the sample is found in the hub of the needle and is not easily expelled. In this case, the sample can be aspirated from the hub using another needle (Fig. 2.6).
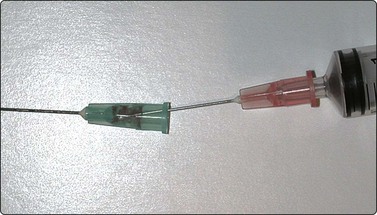
Fig. 2.6 Collecting a sample trapped in the hub of the biopsy needle
The sample is aspirated with another needle mounted on a syringe.
Direct smearing (Figs 2.7-2.12)
Smear quality is highly dependent on the smear being thin and evenly spread, ideally as a monolayer of cells. Perfect smearing is not easily learned. This is one of the reasons why FNB generally has a higher success rate when the biopsy procedure is attended by laboratory staff. Nuclear detail is poorly shown and confusing artifacts are common if smears are thick, uneven and dry slowly (Figs 2.9-2.11). Sometimes, even well prepared smears, particularly of lymphoid cells, may show a peculiar raisin-like distortion of the nuclei probably caused by moisture on the slide (Fig. 2.12). Exposure of air-dried smears to formalin vapor, which can occur during transport of material to the laboratory, can affect nuclear staining and cause loss of morphologic detail.10
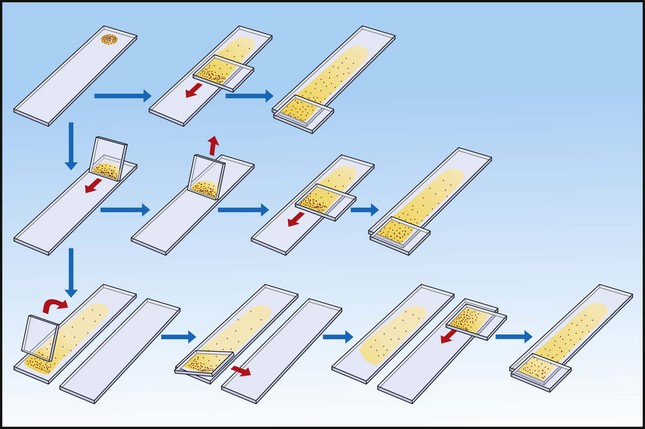
Diagrammatic illustration of smearing of ‘dry’ and ‘wet’ samples. Top line: a ‘dry’ sample is smeared with the flat of a slide exerting a well-balanced pressure; middle line: two-step smearing of a ‘wet’ sample on one slide; bottom line: two-step smearing of a ‘wet’ sample with plenty of fluid moving the concentrated cells to a second slide. For details see text.
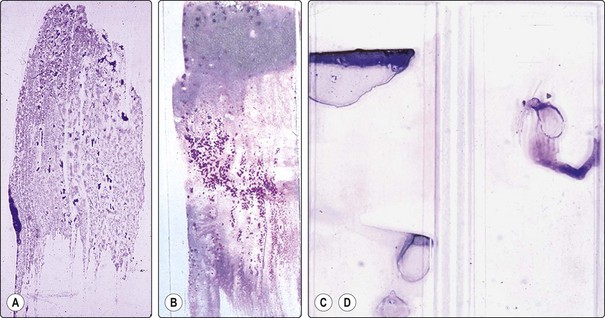
Fig. 2.8 Macro appearance of air-dried smears
(A) Optimal smear of ‘dry’ sample (carcinoma of prostate); cell clusters seen as blue dots, evenly spread; (B) Smear of ‘wet’ sample by two-step smearing; mainly blood at top end, cell clusters concentrated and evenly spread in the thin mid portion; (C, D) Examples of poorly prepared smears of bloody material, partly dried or clotted before smearing.
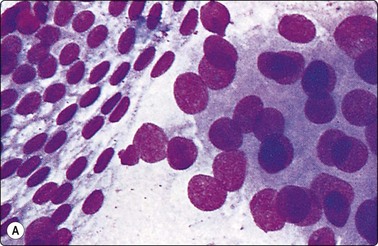
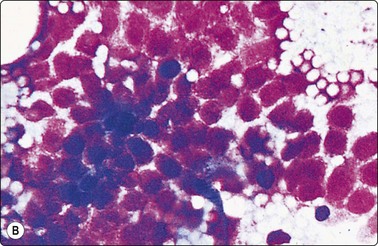
Fig. 2.9 Optimal and suboptimal air-dried smears
(A) Optimal spread and fixation of ‘dry’ sample (carcinoma prostate); (B) Artifacts caused by slow drying of thick bloody smear (prostatic hyperplasia) render diagnosis impossible in spite of good cell yield (MGG, HP).
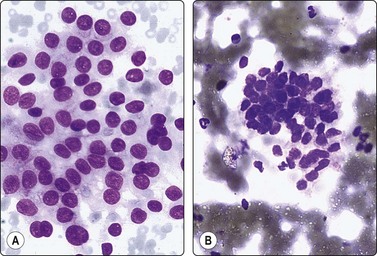
Fig. 2.10 Optimal and suboptimal air-dried smears
(A) Thinly spread, well-fixed smear of papillary carcinoma of thyroid. Typical nuclear morphology well demonstrated; (B) Cells in a thick, bloody part of the same smear. Nuclei show shrinking artifacts: small and dark, structural detail not visible (MGG, IP).
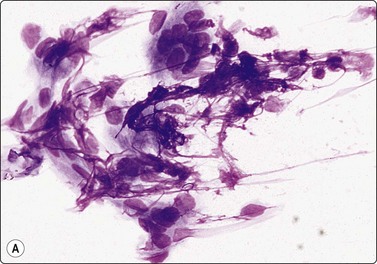
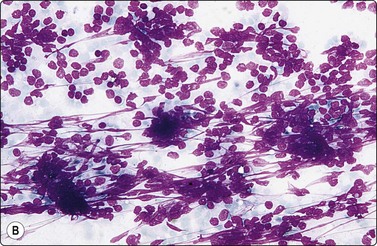
Fig. 2.11 Artifacts caused by smearing
(A) Smudging artifacts at tail of smear caused by heavy pressure; fibroadenoma of breast (MGG, HP); (B) Clumping of cells and shrinkage artifacts due to slow drying/fixation of thick and bloody smear; lymphoid tissue (MGG, IP); Interpretation difficult or impossible in both in spite of good cell yield.
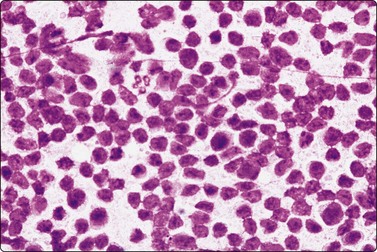
Peculiar fixation artifacts in a smear of lymphoma. Raisinoid nuclei with a ‘prickly’ contour, nuclear chromatin appears irregular (MGG, HP).
The optimal sample, obtainable from cell-rich tissues, has a creamy consistency due to high cellularity with little or no blood or fluid (’dry’ sample). A ‘dry’ sample is best smeared with the flat of a second slide exerting a light pressure as it is moved along the specimen slide (Fig. 2.7, top). The pressure must be carefully adjusted to achieve a thin, even spread without causing disruption of tissue fragments with loss of micro-architecture, or smudging artifacts as in Figure 2.11A. Optimal smearing is a fine balance between too thick and too thin. Smears of ‘dry’ aspirates dry quickly, resulting in a milky, finely granular film on the slide.
A ‘wet’ aspirate consists mainly of blood or fluid containing smaller numbers of cells. The cells can be concentrated and separated from the fluid using a two-step smearing technique as illustrated in Figure 2.7. The smearing slide is held against the specimen slide at a blunt angle near one end of the slide, allowing the fluid to accumulate in the angle. The smearing slide is then rapidly moved along the specimen slide, half way or all the way depending on the amount of fluid. Most of the fluid is left behind while the cells tend to follow the smearing slide. The concentrated cells are then smeared with the flat of the slide as for a ‘dry’ aspirate, either on the same specimen slide, or swiped to another slide.
If a larger volume of blood or fluid is obtained, it can be spread on a slide or watch glass using the needle, or by tilting the slide. With a suitable background, tiny tissue fragments become visible and can be picked up with a needle or a slide, moved to another slide and smeared. Or fragments can be placed in a drop of blood, thrombin added, to form a clot for processing as a cell block. It is critical that bloody samples are processed quickly before coagulation occurs as clotting blood makes it nearly impossible to produce optimal smears.
Examples of the macroscopical appearances of smears are shown in Figure 2.8. Figure 2.8A is an optimal smear of carcinoma showing numerous cell aggregates seen as granules spread evenly over the slide. Figure 2.8B is a two-step smear of carcinoma showing a film of blood at the top and concentrated cells at the middle, seen as a granular material. Figure 2.8C and 2.8D are examples of unsatisfactory smears from external sources.
Indirect smearing
Thin watery samples are processed by centrifugation in a cytocentrifuge. Millipore or Nucleopore filtration is an alternative but has been less satisfactory in our hands. Some laboratories prefer to rinse needles and syringes routinely with saline or with a fixative, which is then centrifuged or filtered onto slides.11,12 More recently, the ThinPrep technique developed for gynecological cytology has been increasingly applied also to FNB specimens.13-16 These techniques offer alternative solutions to the frequent problem of suboptimal samples received from distant sources, when the laboratory has no control over the biopsy procedure. However, the ThinPrep technique has its specific problems, and established diagnostic criteria may have to be redefined for FNB samples.17,18 In our opinion, direct smears expertly prepared by an experienced cytopathologist remain the optimal basis for FNAC diagnosis available today, and our first priority is to perfect this technique. ThinPrep preparations are a valuable supplement, particularly for immunocytochemical staining (see below).
Monolayered smears with optimal cell preservation are particularly important in the diagnosis of malignant lymphoma. For lymph node aspirates, we recommend that a cell suspension be prepared in addition to direct smears. Hank’s balanced salt solution with the addition of 10–20% fetal calf serum is ideal for this purpose. The suspension is spun on the cytocentrifuge at low r.p.m. Dilution may be necessary to achieve optimal dispersion of cells on the slide and to avoid clumping (Fig. 2.13). A number of slides can usually be made from one aspirate to allow immunocytochemical studies. Further details on techniques suitable for lymph node samples are given in Chapter 5.
Tissue fragments and cell blocks
Sometimes, a thin core or fragments of tissue may be obtained with a standard 22-gauge needle (Fig. 2.14). Tissue fragments are fixed in 5–10% buffered isotonic formalin and processed as for routine histology.
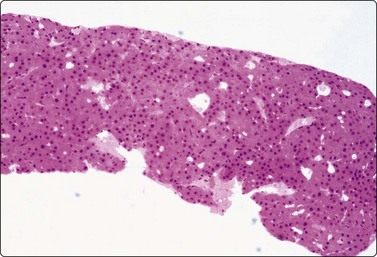
Fig. 2.14 Tissue core by 22-gauge lumbar puncture needle
Tissue section of a 22-gauge lumbar puncture needle core from a well-differentiated hepatocellular carcinoma (H&E, LP).
Some laboratories recommend the routine preparation of cell blocks for paraffin embedding of FNB samples. Cell blocks may give a better idea of tissue architecture and allow multiple sections for panels of immune markers with controls.19-21 However, they are relatively time consuming and costly compared to routine smears.22 We use cell blocks selectively, mainly if a need for immunocytochemistry is anticipated. Cell blocks are helpful if samples are heavily admixed with blood. Surprisingly good tissue fragments are often found in a cell block even when smears show only blood.
More recently, we have developed a simplified technique for cell blocks that we call ‘cell buttons’, shown diagrammatically in Figure 2.15. It is applicable to cell-rich tissues such as lymph nodes and cellular neoplasms. A drop of thick, creamy material obtainable from such tissues using a 27–25-gauge needle without aspiration is gently expelled onto a glass slide as usual, but is not spread or smeared. After a few seconds to allow the drop to adhere to the slide, the slide is carefully immersed in 90% ethanol. The sample remains stuck to the slide as a drop (‘button’). Alcohol-fixation, unlike formalin, holds the sample together. After fixation, the ‘button’ is gently detached with a scalpel blade and processed like a small biopsy. The amount of tissue obtained in this way can be substantial, cell preservation and fixation is excellent, and the material is well suited to immunocytochemical studies (Fig. 2.16). An advantage over a conventional cell block is that the cell material is concentrated, whereas multiple sections may be necessary to find scanty tissue fragments in a cell block.
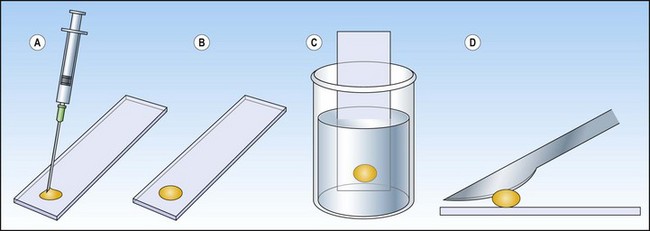
Fig. 2.15 Preparation of a ‘cell button’
(A) The FNB sample is blown onto a clean and dry microscopy slide; (B) The sample/drop is left untouched on the slide a few seconds to adhere without drying; (C) The slide with the sample is immersed in 95% ethanol and left to fix; (D) The solidified fixed ‘cell button’ is carefully removed from the slide with a scalpel and processed routinely like any small biopsy.
Fixation and staining
Two fundamentally different methods of fixation and staining are used in FNAC: air-drying followed by a Romanowsky-type stain such as MGG, Jenner-Giemsa, Wright’s stain or Diff-Quik (Harleco, Philadelphia); and alcohol-fixation followed by Papanicolaou (Pap) or hematoxylin and eosin (H&E) staining. Both methods have their advantages and deficiencies. The effect produced on cells by air-drying and wet-fixation is easily understood if one compares the three-dimensional shape of a fried egg with that of a boiled egg (Fig. 2.17). Air-drying causes the cell, both cytoplasm and nucleus, to flatten on the slide just like an egg flattens in the frying pan. It therefore appears larger than a cell fixed in ethanol, which maintains its three-dimensional rounded shape. Nuclear enlargement and variation in nuclear size are exaggerated in air-dried smears. This enhances the difference between normal and abnormal cells (see Fig. 2.9A).
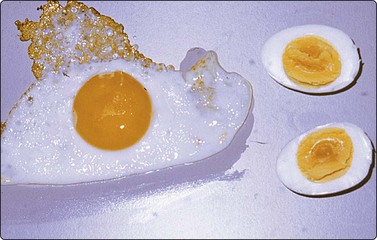
Two eggs of similar weight, one fried, the other boiled and bisected. Compare sizes of the whole egg and of the yolk. Air-drying and alcohol-fixation of cells produce a similar result on cells and nuclei.
Optimal fixation of air-dried smears depends on rapid drying. This can be enhanced by using a hair dryer with moderate heat. Slow drying of thick bloody smears tends to produce artifacts, in particular shrinkage of cells and nuclei, which may render diagnosis impossible (see Fig. 2.10). The main problem with wet-fixed smears is that highly cellular smears dry so quickly that drying artifacts can be difficult to avoid (Fig. 2.18).
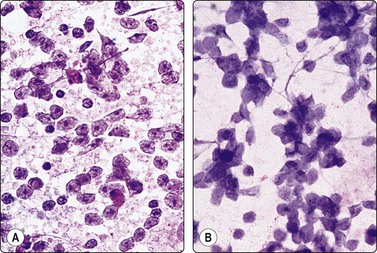
Fig. 2.18 Drying artifacts, alcohol-fixed smear
FNB of large-cell non-Hodgkin’s lymphoma: (A) Well-fixed smear, nuclear detail well demonstrated; (B) Severe drying artifacts, nuclei blurred, interpretation impossible (H&E, HP)
Pathologists trained in gynecological cytology usually prefer alcohol-fixation and Pap staining also for FNB smears while those trained in hematology choose air-dried MGG-stained smears. If sufficient material is available, the two methods should be used in parallel since some features of cells, cell products and stroma are better demonstrated by one than by the other. The differences between air-dried MGG smears and alcohol-fixed Pap smears and the features highlighted by each method are listed in Tables 2.1 and 2.2. The two methods obviously provide complementary diagnostic information. If only air-dried smears are available, some can later be rehydrated and stained with Pap or H&E.23 However, additional material processed as a cell block may be required to allow special stains, multiple immune markers or other ancillary tests.
Table 2.1 Comparison of airdried and wet-fixed smears – general properties
| Airdried MGG | Wet-fixed Pap | |
|---|---|---|
| Dependence on smearing technique | Strong | Moderate |
| The ‘dry’ smear | Good fixation | Drying artefacts common |
| The ‘wet’ smear | Arifacts common | Good fixation |
| Tissue fragments | Cells poorly seen due to heavily stained ground substance | Individual cells usually clearly seen |
| Cell and nuclear size | Exaggerated, differences enhanced | Comparable to tissue sections |
| Cytoplasmic detail | Well demonstrated | Poorly demonstrated |
| Nuclear detail | Pattern different from the familiar Pap stain | Excellently demonstrated |
| Nucleoli | Not always discernible | Well demonstrated |
| Stromal components | Well shown and often differentially stained | Poorly demonstrated |
| Partially necrotic tissue | Poor definition of cell details | Good definition of single intact cells |
Table 2.2 Comparison of airdried and wet-fixed smears – tissues-specific properties
| Tissue | Feature emphasized by MGG | Feature emphasized by Pap |
|---|---|---|
| Epithelial tissues | ||
| Lymphoid tissue | ||
| Mesenchymal tissues | ||
| Neuroendocrine tissues | ||
| Inflammatory tissue |
Many pathologists feel that nuclear chromatin structure is not well shown in air-dried Giemsa smears. However, hematologists examining blood films and bone marrow smears traditionally rely mainly on this technique diagnosing megaloblastic anemia, leukemia, metastatic malignancy, etc. Nuclear chromatin pattern is well shown also in high-quality Romanowsky-stained smears, but appearances are different from Pap smears and have to be learned.
Diff-Quik is a rapid Romanowsky-type stain (2–3 minutes), handy to use in theater or in the radiology department, to check immediately if the sample is satisfactory and if additional tests are needed. The technique was originally designed for blood films, and the staining time must be adjusted to the thickness of FNB smears. In most cases we increase the time in solution 3 to about twice that recommended for blood films. In this way, the staining does not differ significantly from MGG. Rapid modifications of H&E, Pap and other stains have also been developed.24-26
Air-dried smears should be sterilized by fixation in methanol soon after drying to prevent cross-infection, if a significant infective process is suspected.
Special stains
Special stains commonly used in histopathology are also applicable to cytological smears without major modifications.27 Examples are PAS/diastase or Alcian blue for mucins, Prussian blue for iron, Masson-Fontana for melanin, Grimelius for argyrophilic granules and Congo red for amyloid. Microorganisms are identified by Gram, PAS, Ziehl-Neelsen or Gomori’s silver stain. Stains for the demonstration of Pneumocystis, Nocardia and Actinomyces in smears have been developed. 28,29 Glycogen is stained by PAS and fat by oil-red-O in air-dried smears. Fouchet’s reagent counterstained with Sirius red demonstrates bile pigment beautifully (Fig. 2.19).
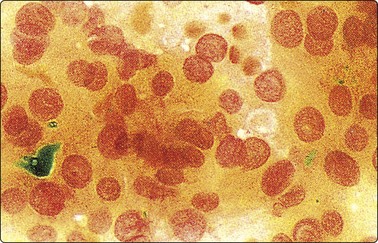
Fig. 2.19 Staining for bile pigment
Fouchet’s reagent counterstained with Sirius red applied to smear from hepatocellular carcinoma (HP).
The reader is referred to handbooks on histological techniques for descriptions of the staining methods mentioned. Minor adjustments to suit the variable quality of cytological material may be necessary.
Phase contrast microscopy
Phase contrast microscopy of unstained smears has been used in cytological diagnosis. We do not see it as a substitute for routine stains but it is a useful tool to check the adequacy of smears to be used for immunocytochemical staining or for EM, so that time and reagents are not wasted on unsatisfactory samples.30
Other investigations
Microbiology (see also Chapter 18)
Fine needle biopsy samples can be used for microbiological culture in cases of suspected infectious disease. Frank pus is best transported to the microbiology department in the aspirating needle and syringe, as rapidly as possible. Very small amounts of pus are washed into a sterile container with a few milliliters of sterile normal saline to prevent drying and desiccation of organisms. This may not provide for anaerobic or unusual organisms; however, dividing very small amounts of material into several types of media is singularly unprofitable. The use of needle washings, after most of the aspirate has been expelled onto slides for microscopy, also seldom yields results. The single most important determinant of whether an organism can be cultured seems to be the amount of aspirated material. Repeated biopsies, using all the contents of the needle for culture, are most valuable.31
In FNB of deep infectious lesions, optimal results are achieved if a microbiologist can be present at the biopsy to process material, particularly in the case of immunosuppressed patients where unusual infections are likely.
Electron microscopy32-38
Although immunocytochemistry has become the most important ancillary method for tumor subtyping, we still find EM necessary for some cases. EM is particularly useful in unusual lung or mediastinal lesions. In Silverman’s experience, FNB samples are the most frequent non-renal samples sent for EM.
We decide on cases to be further studied after an initial evaluation of material in the radiology theater. The most commonly used method of fixation is to eject the aspirate into a small test tube containing glutaraldehyde. Many methods of processing tissue are suggested, mainly with the aim of separating tumor tissue from contaminating red cells. We use Lazaro’s method of cell concentration. The small pellet produced by centrifugation is carefully removed and processed.39 For highly cellular aspirates, the material can be ejected as a semisolid droplet onto a carefully cleaned slide, which is then immersed in glutaraldehyde (compare cell ‘buttons’ as described above). The droplet can be processed on the slide or popped off for further handling. A report can be given in 24–48 hours if necessary.
There is evidence for representative sampling and superior fixation of FNB material compared with surgically excised material (Figs 2.20-2.22).24 In a review of 150 of our cases from various sites, 100 contained adequate well-preserved material for assessment. In 60% of these cases EM only confirmed the LM diagnosis, but in 40% the findings were diagnostic per se. The common applications of EM in FNAC are summarized in Table 2.3. In our experience, most value is obtained in recognizing neuroendocrine tumors and in the specific diagnosis of melanoma, mesothelioma and some carcinomas, including metastases, where immunocytochemistry often cannot provide such positive diagnostic features. An expanding literature about techniques and applications is available.
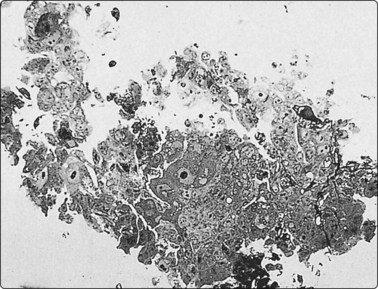
Fig. 2.20 Resin-embedded tissue fragment for EM
Fragment of sarcoma obtained with a standard 22-gauge needle, fixed in glutaraldehyde and processed on the slide, 1 µm section (Toluidin blue, HP).
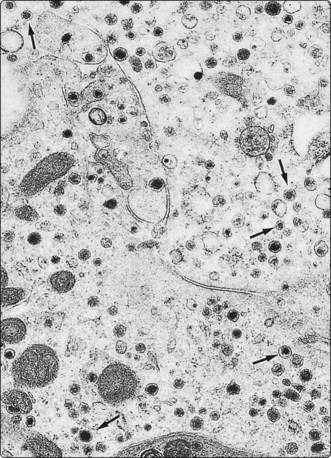
Fig. 2.21 Electron microscopy – carcinoid tumor
FNB of metastatic neoplasm in the liver. Numerous intracytoplasmic rounded neurosecretory granules averaging 155 nm in diameter. In some, a submembranous lucent halo is seen (arrows) (EM × 13 650).
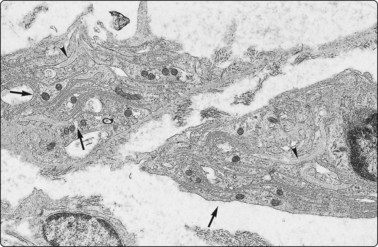
Fig. 2.22 Electron microscopy – schwannoma
Screw needle biopsy (Rotex II) of an atypical soft tissue tumor in the neck of a 20-year-old man. The complex intertwining cytoplasmic processes (arrows) and the associated external (basal) lamina (arrowheads) indicate schwannian differentiation. The abundance of the lucent proteoglycan-containing extracellular matrix is in keeping with Antoni B tissue (EM × 9940).
Table 2.3 Electron microscopy and fine needle aspiration
| Value | Tumor type |
|---|---|
| Unequivocal identification of cell type or differentiation (where available immune markers may not be specific enough for definitive diagnosis) | |
| Supports cytologic diagnosis or aids in subclassification (where immune marker studies are unlikely to be helpful) | |
| Answers general questions of cell type (where immune markers have not been diagnostic or where material is insufficient for a panel of markers) |
Immunocytochemistry37,40-43
The increasing commercial availability of monoclonal antisera to a variety of proteins and other cell products, which are more or less specific to different cell lines, is probably the most important recent development in diagnostic cytology. The demonstration and identification of such cell products in smears and cell blocks is of immense value as it offers a means of objectively recognizing the line of differentiation shown by the cells. Immunostaining may allow a confident specific diagnosis even on relatively scanty material (e.g. medullary carcinoma of thyroid, Fig. 2.23). Immune markers are extremely useful in the differentiation between anaplastic carcinoma, neuroendocrine tumors, malignant lymphoma and amelanotic melanoma (cytokeratin, EMA, chromogranin, NSE, LCA, S-100, etc.), in the search for a primary in metastatic malignancies (e.g. differential staining for a number of cytokeratins), and in the histogenetic typing of mesenchymal tumors (see Chapter 15). Markers for B and T cells, immunoglobulins and light chains are indispensable in the typing of lymphoma (see Chapter 5). Monoclonal antibodies to certain tumor antigens have been found to be useful in the distinction between malignant and benign epithelial cells.44 A list of the most common immune markers used in FNAC is presented in Table 2.4. Immunoprofiles are also included with other diagnostic criteria for most entities in the following chapters.
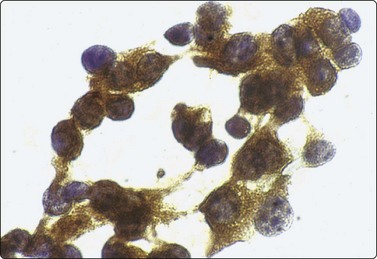
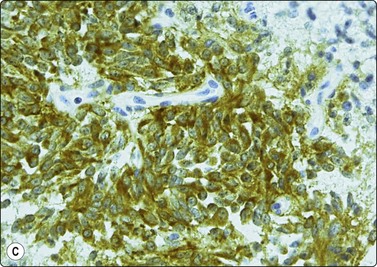
Fig. 2.23 Immunoperoxidase staining
Strongly positive cytoplasmic staining for calcitonin in a direct FNB smear from medullary carcinoma of thyroid (HP).
Table 2.4 Immunoperoxidase (IPOX) studies and fine needle aspiration
| Tumor type or differential diagnosis | Useful tumor markers |
|---|---|
| Small cell malignancies | |
| Lymphoma | Leukocyte common antigen (LCA)/CD45 |
| Small cell carcinoma | Cytokeratin (AE1/AE3, CAM 5.2, CK 20); neuroendocrine markers |
| Small round cell tumors | Cytokeratin; CD 99; Wilms’ tumor gene product (WT-1); neurofilaments; myogenin |
| Large cell malignancies | |
| Lymphoma | LCA/CD 45, CD 30 |
| Anaplastic carcinoma | Cytokeratins, carcinoembryonic antigen (CEA) |
| Melanoma | S-100; HMB-45, Melan A |
| Germ cell tumors | Human chorionic gonadotrophin (HCG); alpha fetoprotein (AFP); placental alkaline phosphatase (PLAP), CD 30, CD 117 |
| Pleomorphic spindle cell tumors | |
| Sarcoma | Cytokeratin; vimentin; desmin; muscle actins; CD 34; S-100 |
| Sarcomatoid carcinoma | Cytokeratin and vimentin coexpression; epithelial membrane antigen (EMA) |
| Melanoma | S-100; HMB-45; Melan A |
| Tubulo-glandular malignancies | |
| Mesothelioma | Calretinin, CK 5/6, EMA |
| Adenocarcinoma | CEA; CD 15; Ber EP 4; B 72.3; CK 7, CK 20 |
| Lymphoma typing | CD 20; CD 3; CD 5; CD 43; CD 15; CD 30; CD 10, Alk-1, cyclin D1; kappa and lambda light chains |
| Specific tumors | |
| Prostatic carcinoma | Prostate-specific antigen (PSA); prostatic acid phosphatase (PAP) |
| Thyroid carcinoma | Thyroid transcription factor-1 (TTF-1); thyroglobulin; calcitonin |
| Breast carcinoma | Gross cystic disease fluid protein-15 (GCDFP-15); Estrogen receptor/progesteron receptor |
| Lung carcinoma | TTF-1 |
| Neuroendocrine tumors (carcinoid; neuroendocrine carcinomas) | Synaptophysin; bombesin; chromogranin; ACTH; serotonin |
| Metastatic malignancies | CK7, CK20, Cam5.2, AE1/AE3, CEA, TTF-1, CDX2, CA-19,9, CA120 |
Sections of cell blocks fixed in 10% formalin and transferred to TBS for immunoperoxidase staining, or tissue fragments obtained by core needle biopsy, in general provide the best material for immunocytochemistry. Cell blocks and core biopsies offer the great advantage of a large number of sections, sufficient for a panel of markers and for the indispensable negative controls. On the other hand, some antigens are not well preserved in formalin-fixed material, but are demonstrable in air-dried smears. In stroma-rich tumors, FNB selectively samples the abnormal cells detached from the stroma, which sometimes facilitates interpretation.
If tissue fragments are not available, immunostaining can be performed on direct smears, or on smears derived from a cell suspension using the cytocentrifuge (Fig. 2.24). Alcohol-fixed smears are usually preferable to air-dried smears. ThinPrep slides of FNAC material have been found highly suitable for immunocytochemical staining.45,46 This technique has the advantage of eliminating background interference by blood and debris (see liquid-based cytology below). The limited number of tests possible when only smears are available can be increased by circling areas 3 mm apart on the same slide with a diamond pencil and wiping the smear between. This allows 2–3 different tests per slide. De-stained previously Pap-stained smears can be used, although poor attachment of the cells to the slide can be a problem.47 Sections of alcohol-fixed and paraffin-embedded cell ‘buttons’ are a good alternative to tissue cores and cell blocks (Fig. 2.25 and see Fig. 2.16).
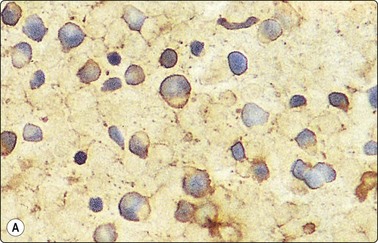
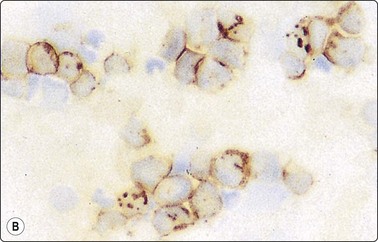
Fig. 2.24 Immunoperoxidase staining
Smears of non-Hodgkin’s lymphoma. (A) Direct smear; interpretation difficult due to background staining caused by fragmentation of cytoplasm; (B) Cytocentrifuge preparation; positive staining distinctly related to individual cells (HP).
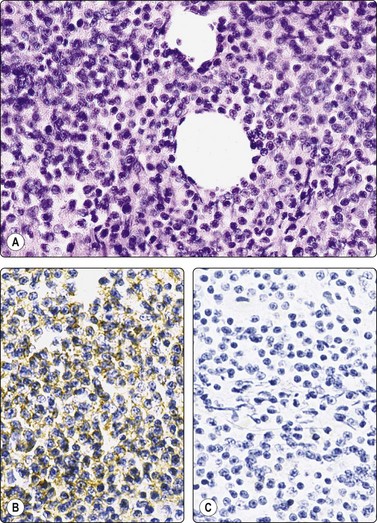
Fig. 2.25 Immunoperoxidase staining
‘Cell button’ of FNB sample of non-Hodgkin’s B-cell lymphoma; (A) Tissue section, H&E; (B) Positive staining with a pan-B marker; (C) negative staining with a pan-T marker (IP).
The avidin–biotin complex method is the most commonly used with both monoclonal and polyclonal primary antibodies. Diaminobenzidine is used as the marker dye. Immunoalkaline phosphatase staining appears to offer several advantages in cytological preparations. Commercially produced kits have made immunohistochemistry a relatively simple method available to any cytology laboratory, and an ever-increasing number of antisera are being marketed in this form. Appropriate controls are crucial to achieving diagnostic accuracy.48 A review of immunostaining techniques adapted to cytological preparations can be found in Diagnostic Cytopathology.44
Interpretation of immunohistochemical staining is often more difficult in smears than in histological sections because the cytoplasm of neoplastic cells detached from the stroma is often fragile and dispersed in the background. Blood, serum or secretory products are often superimposed on the cells. All this makes it difficult to ascribe any positive staining to specific, identifiable cells. Ways to overcome the difficulties are discussed in the paper by Suthipintawong and Leong referred to above.44 When examining the smears, it is best to focus on tissue fragments, in which the cells are protected leaving cell membranes and cytoplasm intact. Background staining is a lesser problem in cytospin preparations if the cells are washed, in ThinPrep smears, and in paraffin sections from a cell ‘button’. The results of immunocytochemistry must be interpreted with caution and in the context of conventional cytomorphology and clinical data.49
Standardized/simplified approaches to FNB for radiologists
The challenges facing laboratories include how to standardize the approach to sampling by radiologists in case pathology staff is not available. This includes guidelines for setting aside material for ancillary techniques, and the transport of cytological material to laboratories from radiology practices.
Miscellaneous techniques
Philippe Vielh
Depending on the type of question asked, the (cyto)pathologist may have to triage specimens according to the type of technique needed to solve a specific problem. From that point of view, on-site examination of a given specimen using rapid stainings will help tremensdously. Table 2.5 summarizes the possibilities and limitations of using different techniques according to the type of cellular specimen.
Liquid-based cytology
New cytopreparatory techniques include the so-called liquid-based cytology technology. Various systems are commercially available. They are mostly used for cervical cancer screening but are also adapted for FNAC samples. The basic principle is to collect cell samples into a liquid fixative solution and then create a monolayer of cells ready for microscopic observation after staining. The presence of the liquid helps collecting cells remaining in the needle, whereas immediate fixation due to the presence of methanol or ethanol in the fixative optimizes cell preservation and usually reduces dramatically the bloody background. This potentially may: (1) help in the visual and automatic detection of cells of interest, (2) improve the quality and reproducibility of immunocytochemical methods (but antibodies used should be selected considering the fact that fixation is made using coagulant compounds), and (3) preserve nucleic acids, specially DNA. However, flow cytometric methods, cytogenetic analysis, as well as more sophisticated molecular techniques such as gene expression studies based on quantification of RNA still need fresh material.
Image analysis
Image analysis deals with three different areas, namely morphometry, object counting and cytometry, which are not mutually exclusive.50
Morphometry is the quantitative description of geometric features of structures such as tissues, cells, nuclei or nucleoli.51,52 It includes (1) stereology techniques estimating the fraction of different tissue components, inner and outer surface density, as well as shape and volume by means of a test system of lower dimension (i.e. point or line grids) than the structure itself, and (2) measurement of geometric features of structures in the two-dimensional microscopical image, also called planimetry.53 Currently, the microscopical image is recorded by a video camera and displayed on a computer screen which makes it possible to trace the outlines of nuclei on the screen and then compute nuclear areas as well as nuclear shape using dedicated software able to produce quantitative data in the form of cytograms and histograms.54
Object counting mainly concerns quantitation of mitoses or measurement of the proliferation fraction of a cell population using antibodies raised against incorporated nonisotopic labels during S-phase fraction, for example, 5-bromodeoxyuridin (BrdU) or proliferation-associated molecules such as the molecule recognized by the Ki-67 or the MIB-1 antibody and the proliferating cell nuclear antigen.55 It also makes it possible to quantitate apoptotic figures by means of the TUNEL assay.56
Cytometry depends on the ability to detect a substance of interest by a specific dye and to measure the concentration of that dye by computing optical density.57 Its main application, based on the discovery of the Feulgen reaction, is DNA cytometry by means of a specific stoichiometric stain for DNA, permitting quantitative evaluation of nuclear DNA by absorbance cytometry since the amount of stain is proportional to the amount of DNA present. This can also be done using various fluorochromes, which also bind stoichiometrically, i.e. in a proportional way, such as Hoechst 4,6-dimanidino-2-phenylindole (DAPI) and propidium iodide dyes.58-60 Powerful computers also have stimulated the development of systems for automatic cell classification based on pattern recognition for diagnostic,61,62 prognostic,63-65 and predictive66 purposes. Quantitation of nuclear immunostain of estrogen and progesterone receptors,67 proliferation markers68-70 and tumor suppressor genes such as p5371 stresses the need for standardization of immunocytochemical techniques and quality control networks since immunocytochemical staining of antigens is not stoichiometric and therefore requires calibration with known external and internal controls as for DNA image cytometry.72 Finally, a new device named the laser scanning cytometer has recently been developed.73 It is supposed to bridge the gap between an image analyser machine and a flow cytometer, since it makes it possible to perform multiparameter analyses.74,75
Flow cytometry
Also based on the fundamental work showing that DNA content, measured by ultraviolet and visible light in unstained cells, doubled during the cell cycle,76 followed by the improved detection of antigens using fluorescence methods77 and the development of an apparatus capable of counting78 and sizing79 blood cells, flow cytometry has mainly been applied to the measurement of DNA content in human cancer and quantitation of cell surface antigens in hematopathology.80 Current flow cytometers provide rapid, sensitive and quantitative measurements of any cell component that can be specifically stained using appropriate fluorochromes for DNA or RNA and monoclonal antibodies raised against cytoplasmic, nuclear and membrane antigens. They permit the acquisition of monoparametric and multiparametric data characterizing heterogeneous populations in a cell suspension obtained, for example, by fine needle sampling of human cancer.81,82 Again, quality controls are mandatory83-85 for estimating the usefulness of flow cytometry measurements in a clinical setting and for comparing data published by various teams around the world. This is particularly critical when dealing with data which may influence diagnosis and which have prognostic and therapeutic implications such as flow immunophenotyping of hematologic malignancies,86,87 evaluation of proliferation by measurement of S-phase fraction88-90 and assessment of spontaneous or drug-induced human cancer apoptosis.91 However, since flow cytometry and image analysis are complementary, it is likely that the combination of both methods will contribute to better estimation of relevant pathological processes.92
The practical application of flow cytometry to the diagnosis and typing of lymphoma is discussed in Chapter 5.
Molecular cytopathology (Table 2.6)
The application of molecular probes to cytologic samples of human malignancies has refined the diagnostic and prognostic armamentarium.93-96 The use of genotypic rather than phenotypic diagnostic criteria also makes it possible to measure and combine information from both morphologic and molecular observations by means of digital image analysis.97 Current diagnostic and potential prognostic applications in pathology have improved tremendously using in situ hybridization, in situ amplification techniques and other recently developed nucleic acid-based methods of analysis.
Table 2.6 Examples of current and potential useful diagnostic and prognostic molecular markers according to tissue-derived tumours (see text for details)
| Tissue-derived tumors | Useful diagnostic molecular markers | Useful prognostic molecular markers |
|---|---|---|
| Epithelial | Gain or loss of DNA (potential) RNA-specific transcripts (thyroid) |
DNA flow cytometric-derived S-phase fraction (breast) In situ detection of HER-2/neu amplification (breast) |
| Lymphoid | DNA translocations and amplifications | Gain or loss of DNA (potential) |
| Mesenchymal | DNA translocations | Gain or loss of DNA and RNA-specific transcripts (potential) |
| Neuroectodermal | RNA-specific transcripts |
In situ hybridization is a technique for the localization of specific nucleic acid (endogenous DNA, messenger RNA, viral or bacterial) sequences within individual cells based on the complementary binding of a nucleotide probe (usually oligomers), labeled with nonisotopic (for example, fluorochromes) reporter molecules, to a specific target sequence of DNA or RNA.98 Given optimal preparation of cytologic specimens,99 applications of in situ hybridisation techniques in cytopathology are numerous, including detection of bacterial and viral infections and detection of messenger RNA of genes coding for oncoproteins, growth factors and growth factor receptors, cytokines, adhesion and multidrug resistance molecules as well as cycle proteins.98 Using probes to chromosome-specific (centromeric or telomeric) sequences, it is possible to detect aneuploidy in interphase nuclei100,101 and losses, gains or amplifications of chromosome regions with known prognostic value.102,103 Using sequence-specific probes, recurrent chromosomal rearrangements104,105 of great diagnostic value can easily be identified and documented. Finally, comparative genomic hybridization106 is a newly developed and global approach to detecting and defining the specific combination of genetic changes in individual tumors. All these molecular approaches based on in situ hybridization need the development of digital imaging analyzers for optimal quantitation.
In situ amplification techniques are based on the polymerase chain reaction (PCR) which allows recovery of large amounts of DNA from minute quantities of starting material.107 Various adaptations of the PCR have been developed for cytological preparations108 such as PCR in situ hybridization, in situ PCR, reverse transcriptase in situ PCR,109 methylation-specific PCR110 and primed in situ synthesis.111 Fixation and preparation of cells are critical steps for optimal in situ amplification with oligonucleotide primers followed by detection of the amplified product using nonisotopic reporter molecules. Appropriate controls including reference control genes, known negative samples for the target sequence, together with irrelevant primers and probes, are mandatory for specificity and quantitation of the reaction.111 This is also the case for detection of gene fusions encoding chimeric messenger RNA used as specific diagnostic genetic markers in several lymphoid and myeloid malignancies and in some solid tumors.112 In the same way, DNA or RNA amplification by means of in vitro PCR or reverse transcriptase PCR have recently been successfully performed using cytologic material laser-dissected113 or directly scraped from routinely stained, archival slides.114-117
Other nucleic acid-based methods such as microsatellite analysis,118 quantitation of telomerase activity,119 methodologies using DNA120 and cDNA121,122 chip arrays and serial analysis of gene expression 118 in small clinical samples, and the proton magnetic resonance technique123,124 are currently under evaluation.
Future prospects
Coordinating driving forces coming from the development of robust protocols using new versatile fluorochromes and automated digital optical imaging will undoubtedly help the pathologist in quantifying in situ amplification and hybridization techniques and in applying new technologies such as in situ hybridization techniques and micro-arrays for the study of DNA and RNA.125 It is also anticipated that proteomic evaluation of cytologic material may help the cytopathologist in better evaluating the diagnosis of a particular tumor and the prognosis of a given patient.126
These very promising perspectives reinforce the central role, responsibility and future implications of the (cyto)pathologist in helping the clinician to tailor and adapt the treatment of patients.127,128
1 Smith EH. Complications of percutaneous abdominal fine-needle biopsy. Review. Radiology. 1991;178:253-258.
2 Powers CN. Complications of fine needle aspiration biopsy: the reality behind the myths. In: Schmidt, WA. Cytopathology. Chicago: ASCP Press; 1996:69-91.
3 Logan-Young W, Dawson AE, Wilbur DC, et al. The cost effectiveness of fine-needle aspiration cytology and 14-gauge core needle biopsy compared with open surgical biopsy in the diagnosis of breast carcinoma. Cancer. 1998;82:1867-1873.
4 Liberman L. Percutaneous image-guided core breast biopsy. Radiol Clin N Am. 2002;40:483-500.
5 Haack LA, Meier JS, Gluth J, et al. The other side of the needle: a patient’s perspective. Diagn Cytopathol. 2006;34:303-306.
6 Thompson P. Thin needle aspiration biopsy. Letter. Acta Cytol. 1982;26:262-263.
7 Zajdela A, Zillhardt P, Voillemot N. Cytological diagnosis by fine needle sampling without aspiration. Cancer. 1987;59:1201-1205.
8 Akhtar SS, Imran-Ul-Huq, Faiz-U-Din M, et al. Efficacy of fine-needle capillary biopsy in the assessment of patients with superficial lymphadenopathy. Cancer (Cancer Cytopathol). 1997;81:277-280.
9 Kate MS, Kamal MM, Bobhate SK, et al. Evaluation of fine needle capillary sampling in superficial and deep-seated lesions. An analysis of 670 cases. Acta Cytol. 1998;42:679-684.
10 Choo C, Frost F. Blue smears: a new artifact in FNA smears due to formalin vapour. Cytoletter. 2002;20:4-6.
11 Boon ME, Lykles C. Imaginative approach to fine needle aspiration cytology. Lancet. 1980;2:1031-1032.
12 Sirkin W, Auger M, Donat E, et al. Cytospin – an alternative method for fine needle aspiration cytology of the breast: a study of 148 cases. Diagn Cytopathol. 1995;13:266-269.
13 Bédard YC, Pollett AF. Breast fine-needle aspiration. A comparison of ThinPrep and conventional smears. Am J Clin Pathol. 1999;111:523-527.
14 Nasuti JF. Utility of the ThinPrep technique in thyroid fine needle aspiration: optimal vs. practical approaches. Acta Cytol. 2006;50:3-4.
15 Saleh H, Bassily N, Hammoud J. Utility of a liquid-based, monolayer preparation in the evaluation of thyroid lesions by fine needle aspiration biopsy. Comparison with the conventional smear method. Acta Cytol. 2009;53:130-136.
16 Hoda RS. Non-gynecological cytology on liquid-based preparations. A morphologic review of facts and artifacts. Diagn Cytopathol. 2007;35:621-634.
17 Michael CW, Hunter B. Interpretation of fine-needle aspirates processed by the ThinPrep technique: cytologic artifacts and diagnostic pitfalls. Diagn Cytopathol. 2000;23:6-13.
18 Nasuti JF, Tam D, Gupta PK. Diagnostic value of liquid-based (ThinPrep) preparations in nongynecologic cases. Diagn Cytopathol. 2001;24:137-141.
19 Bell DA, Carr CP, Szyfelbein WM. Fine needle aspiration cytology of focal liver lesions. Results obtained with examination of both cytologic and histologic preparations. Acta Cytol. 1986;30:397-402.
20 De Boer WB, Segal A, Frost FA, et al. Can CD34 discriminate between benign and malignant hepatocytic lesions in fine-needle aspirates and thin core biopsies? Cancer (Cancer Cytopathol). 2000;90:273-278.
21 Nathan NA, Narayan E, Smith MM, et al. Cell block cytology. Improved preparation and its efficacy in diagnostic cytology. Am J Clin Pathol. 2000;114:599-606.
22 Liu K, Dodge R, Glasgow BJ, et al. Fine-needle aspiration: comparison of smear, cytospin, and cell block preparations in diagnostic and cost effectiveness. Diagn Cytopathol. 1998;19:70-74.
23 Shidham VB, Kampalath B, England J. Routine air drying of all smears prepared during fine needle aspiration and intraoperative cytologic studies. An opportunity to practice a unified protocol offering the flexibility of choosing a variety of staining methods. Acta Cytol. 2001;45:60-68.
24 Pak HY, Yokota S, Teplitz RL, et al. Rapid staining techniques employed in fine needle aspiration of the lung. Acta Cytol. 1981;25:178-184.
25 Pak HY, Yokota SB, Teplitz RL. Rapid staining techniques employed in fine needle aspirations. Acta Cytol. 1983;27:81-82.
26 Yang GCH, Alvarez II. Ultrafast Papanicolaou stain: an alternative preparation for fine needle aspiration cytology. Acta Cytol. 1995;39:55-60.
27 Sachdeva R, Kline TS. Aspiration biopsy cytology and special stains. Acta Cytol. 1981;25:678-683.
28 Pintozzi RL, Blecka LJ, Nanon S. The morphologic identification of Pneumocystis carinii. Acta Cytol. 1979;23:35-39.
29 Pollock PG, Valicenti JF, Meyers DS, et al. The use of fluorescent and special staining techniques in the aspiration of nocardiosis and actinomycosis. Acta Cytol. 1978;22:575-579.
30 Boccato P, Briani G, Bizzaro N, et al. Cytology in ‘black and white’. Acta Cytol. 1987;31:643-645.
31 Krane JF, Renshaw AA. Relative value and cost-effectiveness of culture and special stains in fine needle aspirates of the lung. Acta Cytol. 1998;42:305-311.
32 Dabbs DJ, Silverman JF. Selective use of electron microscopy in fine needle aspiration cytology. Acta Cytol. 1988;32:880-884.
33 Dardick I, Yazdi HM, Brosko C, et al. A quantitative comparison of light and electron microscopic diagnoses in specimens obtained by fine needle aspiration biopsy. Ultrastruct Pathol. 1991;15:105-129.
34 Akhtar M, Bakry M, Al-Jeaid AS, et al. Electron microscopy of fine needle aspiration biopsy specimens: a brief review. Diagn Cytopathol. 1992;8:278-282.
35 Kurtz SM. Rapid ultrastructural examination of FNAs in the diagnosis of intrathoracic tumours. Diagn Cytopathol. 1992;8:289-292.
36 Yazdi HM, Dardick I. Diagnostic immunocytochemistry and electron microscopy. Guides to clinical aspiration biopsy. New York: Igaku-Shoin; 1992.
37 Davidson DD, Goheen MP. Preparation of fine needle aspiration biopsies for electron microscopy. In: Schmidt WA, editor. Cytopathology annual. Baltimore: Williams and Wilkins; 1993:255-264.
38 Qiononez GE, Ravinsky E, Paraskevas M, et al. Contribution of transmission electron microscopy to fine-needle aspiration biopsy diagnosis: comparison of cytology and combined cytology and transmission electron microscopy with final histological diagnosis. Diagn Cytopathol. 1996;15:282-287.
39 Lazaro AV. Technical note: improved preparation of fine needle aspiration biopsies for transmission electron microscopy. Pathology. 1983;15:399-402.
40 Van Hoeven KH, Fitzpatrick BT, Bibbo M. Update of immunocytochemistry in cytopathology. In: Rosen PP, Fechner RE, editors. Pathology annual, vol.30. Stanford: Appleton and Lange; 1995. part 2
41 Osborn M, Domagala W. Immunocytochemistry. In Bibbo M, editor: Comprehensive cytopathology, 2nd ed, Philadelphia: Saunders, 1997.
42 Polak J, van Noorden S. Introduction to immunocytochemistry. Current techniques and problems, 2nd ed. Berlin: Springer-Verlag; 1997.
43 Suthipintawong C, Leong AS-Y. Immunostaining of cell preparations: a comparative evaluation of common fixatives and protocols. Diagn Cytopathol. 1996;15:167-174.
44 Campbell F, Herrington CS. Application of cytokeratin 7 and 20 immunohistochemistry to diagnostic pathology. Current Diagn Pathol. 2001;7:113-122.
45 Leung SW, Bedard YC. Immunocytochemical staining on ThinPrep processed smears. Mod Pathol. 1996;9:304-306.
46 Dabbs DJ, Abendroth CS, Grenko RT, et al. Immunocytochemistry on the ThinPrep processor. Diagn Cytopathol. 1997;17:388-392.
47 Abendroth CS, Dabbs DJ. Immunocytochemical staining of unstained versus previously stained cytologic preparations. Acta Cytol. 1995;39:379-386.
48 Kurtycz DFI, Logrono R, Leopando M, et al. Immunocytochemistry controls using cell cultures. Diagn Cytopathol. 1997;17:74-79.
49 Holmes GF, Eisele DW, Rosenthal D. PSA immunoreactivity in a parotid oncocytoma: a diagnostic pitfall in discriminating primary parotid neoplasms from metastatic prostate cancer. Diagn Cytopathol. 1998;19:221-225.
50 Meijer GA, Beliën JAM, van Diest PJ, et al. Image analysis in clinical pathology. J Clin Pathol. 1997;50:365-370.
51 Baak JPA. Manual of quantitative pathology in cancer diagnosis and prognosis. Berlin: Springer-Verlag; 1991.
52 Hamilton PW, Allen DC. Quantitative clinical pathology. Oxford: Blackwell Scientific; 1995.
53 Zajdela A, de la Riva L, Ghossein N. The relation of prognosis to the nuclear diameter of breast cancer obtained by cytologic aspiration. Acta Cytol. 1984;23:75-80.
54 True LD. Morphometric applications in anatomic pathology. Human Pathol. 1996;27:450-467.
55 Hall PA, Coates PJ. Assessment of cell proliferation in pathology – what next? Histopathology. 1995;26:105-112.
56 Maciorowski Z, Klijanienko J, Padoy, et al. Comparative image and flow cytometric TUNEL analysis of fine needle samples of breast carcinoma. Cytometry. 2001;46:150-156.
57 Caspersson TO. History of the development of cytophotometry from 1935 to the present. Anal Quant Cytol Histol. 1987;9:2-6.
58 Mikel UV. Quantitative staining techniques for image cytometry. In: Mikel UV, editor. Advanced laboratory methods in histology and pathology. Washington DC: Armed Forces Institute of Pathology; 1994:131-160.
59 Maciorowski Z, Veilleux C, Gibaud A, et al. Comparison of fixation procedures for fluorescent quantitation of DNA content using image cytometry. Cytometry. 1997;26:123-129.
60 Truong K, Vielh P, Malfoy B, et al. Fluorescence-based analysis of DNA ploidy and cell proliferation within fine needle samplings of breast tumors; a new approach using automated image cytometry. Cancer. 1998;84:309-316.
61 Cross SS, Bury JP, Stephenson TJ, et al. Image analysis of low magnification images of fine needle aspirates of the breast produces useful discrimination between benign and malignant cases. Cytopathol. 1997;8:265-273.
62 Teague MW, Wolberg WH, Street WN, et al. Indeterminate fine needle aspiration of the breast: image analysis-assisted diagnosis. Cancer (Cancer Cytopathol). 1997;81:129-135.
63 Briffod M, Le Doussal V, Spyratos F. Cytologic nuclear grading of fine-needle cytopunctures of breast carcinoma. Comparison with histologic nuclear grading and image cytometric data. Anal Quant Cytol Histol. 1997;19:114-122.
64 Cohen C. Image cytometric analysis in pathology. Human Pathol. 1996;27:482-493.
65 Wolberg WH, Street WN, Mangasarian OL. Computer-derived nuclear features compared with axillary lymph node status for breast carcinoma prognosis. Cancer (Cancer Cytopathol). 1997;81:172-179.
66 Briffod M, Spyratos F, Hacène K, et al. Evaluation of breast carcinoma chemosensitivity by flow cytometric DNA analysis and computer-assisted image analysis. Cytometry. 1992;13:250-258.
67 Auger M, Katz RL, Johnston DA, et al. Quantitation of immunocytochemical estrogen and progesterone receptor content in fine needle aspirates of breast carcinoma using the SAMBA 4000 image analysis system. Anal Quant Cytol Histol. 1993;15:274-280.
68 Bozzetti C, Nizzoli R, Camisa R, et al. Comparison between Ki-67 index and S-phase fraction on fine needle aspiration samples from breast carcinoma. Cancer (Cancer Cytopathol). 1997;81:287-292.
69 Katz RL, Wojcik EM, El-Naggar AK, et al. Proliferation markers in non-Hodgkin’s lymphoma: a comparative study between cytophotometric quantitation of Ki-67 and flow cytometric proliferation index on fine needle aspirates. Anal Quant Cytol Histol. 1993;15:179-186.
70 Oud PS, Bauwens A, Nauwelaers FA. Multiparameter absorption measurements in automated microscopy: simultaneous quantitative determination of DNA and nuclear antigen. Acta Cytol. 1997;41:188-196.
71 Friedrich K, Thieme B, Haroske G, et al. Nuclear image analysis of p53-positive and -negative cells in breast carcinoma. Anal Quant Cytol Histol. 1997;19:285-293.
72 Marchevsky A. Quality assurance issues in DNA image cytometry. Cytometry (Commun Clin Cytometry). 1996;26:101-107.
73 Kamentsky LA, Burger DE, Gershamn RJ, et al. Slide-based laser scanning cytometry. Acta Cytol. 1997;41:123-143.
74 Clatch RJ, Walloch JL. Multiparameter immunophenotypic analysis of fine needle aspiration biopsies and other hematologic specimens by laser scanning cytometry. Acta Cytol. 1997;41:109-122.
75 Gorczyca W, Darzynkiewicz Z, Melamed MR. Laser scanning cytometry in pathology of solid tumors. Acta Cytol. 1997;41:98-108.
76 Caspersson TO, Schultz J. Nucleic acid metabolism of the chromosomes in relation to gene reproduction. Nature. 1938;142:294-297.
77 Coons AH, Kaplan MH. Localization of antigen in tissue cells. II. Improvements in a method for the detection of antigen by means of fluorescent antibody. J Exp Med. 1950;91:1-4.
78 Crosland-Taylor PJ. A device for counting small particles suspended in a fluid through a tube. Nature. 1953;171:37-38.
79 Coulter WH. High speed automatic blood cell counter and cell size analyser. Proc Natl Electronics Conf. 1956;12:1034-1042.
80 Giaretti W. Flow cytometry and applications in oncology. J Clin Pathol. 1997;50:275-277.
81 Robins DB, Katz RL, Swan FJr, et al. Immunotyping of lymphoma by fine needle aspiration. A comparative study of cytospin preparations and flow cytometry. Am J Clin Pathol. 1994;101:569-576.
82 Vielh P. Flow cytometry. Guides to clinical aspiration biopsy. New York: Igaku-Shoin; 1991.
83 Braylan RC, Borowitz MJ, Davis BH, et al. U.S. –Canadian consensus recommendations on the immunophenotypic analysis of hematologic neoplasia by flow cytometry. Cytometry (Commun Clin Cytometry). 1997;30:213.
84 Duque RE, Andreeff M, Braylan RC, et al. Consensus review of the clinical utility of DNA flow cytometry in neoplastic hematopathology. Cytometry. 1993;14:492-496.
85 Hiddemann W, Schumann J, Andreeff M, et al. Convention on nomenclature for DNA cytometry. Cancer Genet Cytogenet. 1984;13:181-183.
86 Jennings CD, Foon KA. Recent advances in flow cytometry: application to the diagnosis of hematologic malignancy. Blood. 1997;90:2863-2892.
87 Zeppa P, Marino G, Troncone G, et al. Fine-needle cytology and flow cytometry immunophenotyping and subclassification of non-Hodgkin lymphoma. Cancer. 2004;102:55-65.
88 Vielh P, Carton M, Padoy E, et al. S-phase fraction as an independent prognostic factor of long-term overall survival in patients with early-stage or locally advanced invasive carcinoma. Cancer. 2005;105:476-482.
89 D’Hautcourt JL, Spyratos F, Chassevent A. Quality control study by the French Cytometry Association on flow cytometric DNA content and S-phase fraction (S%). Cytometry (Commun Clin Cytometry). 1996;26:32-39.
90 Silvestrini R. Quality control for evaluation of the S-phase fraction by flow cytometry: a multicentric study. Cytometry (Commun Clin Cytometry). 1994;18:11-16.
91 Darzynkiewicz A, Juan G, Li X, et al. Cytometry in cell necrobiology: analysis of apoptosis and accidental cell death (necrosis). Cytometry. 1997;27:1-20.
92 Maciorowski Z, Klijanienko J, Padoy E, et al. Differential expression of Bax and Bcl2 in the assessment of cellular dynamics in fine-needle samples of primary breast carcinomas. Cytometry. 2000;42:264-269.
93 Krishnamurthy S. Applications of molecular techniques to fine-needle aspiration biopsy. Cancer. 2007;11:106-122.
94 Schmitt F, Loghatto-Filho A, Valent A, et al. Molecular techniques in cytopathology practice. J Clin Pathol. 2008;61:258-267.
95 Chevillard S, Pouillart P, Beldjord C, et al. Sequential assessment of multidrug resistance phenotype and measurement of S-phase fraction as predictive markers of breast cancer response to neoadjuvant chemotherapy. Cancer. 1996;77:292-300.
96 Chevillard S, Lebeau J, Pouillart P, et al. Biological and clinical significance of concurrent p53 gene alterations, MDR1 gene expression, and S-phase fraction analyses in breast cancer patients treated with primary chemotherapy or radiotherapy. Clin Cancer Res. 1997;3:2471-2478.
97 Waldman FM, Sauter G, Sudar D, et al. Molecular cytometry of cancer. Human Pathol. 1996;27:441-449.
98 McNicol AM, Farquharson MA. In: situ hybridization and its diagnostic applications in pathology. J Pathol. 1997;182:250-261.
99 Abati A, Sanford JS, Fetsch P, et al. Fluorescence in situ hybridization (FISH): a user’s guide to optimal preparation of cytologic specimens. Diagn Cytopathol. 1995;13:485-492.
100 Sauer T, Beraki K, Jebsen PW, et al. Ploidy analysis by in situ hybridization of interphase cell nuclei in fine needle aspirates from breast carcinomas: correlation with cytologic grading. Diagn Cytopathol. 1997;17:267-271.
101 Truong K, Guilly MN, Gerbault-Seureau M, et al. Quantitative FISH by image cytometry for the detection of chromosome 1 imbalances in breast cancer: a novel approach analyzing chromosome rearrangements within interphase nuclei. Lab Invest. 1998;78:1607-1613.
102 Wolman S. Applications of fluorescence in situ hybridization techniques in cytopathology. Cancer (Cancer Cytopathol). 1997;81:193-197.
103 Klijanienko J, Couturier J, Galut M, et al. Detection and quantitation by FISH and image analysis of HER-2/neu gene amplification in breast cancer fine-needle samples. Cancer. 1999;87:312-318.
104 Mitelman F, Johansson B, Mandahl N, et al. Clinical significance of cytogenetic findings in solid tumors. Cancer Genet Cytogenet. 1997;95:1-8.
105 Gong Y, Caraway N, Gu J, et al. Evaluation of interphase fluorescence in situ hybridization for the t(14;18)(q32;q21) translocation in the diagnosis of follicular lymphoma on fine-needle aspirates. Cancer. 2003;99:385-393.
106 Forozan F, Karhu R, Kononen J, et al. Genome screening by comparative genomic hybridization. Trends Genet. 1997;13:405-409.
107 O’Leary JJ, Engels K, Dada MA. The polymerase chain reaction in pathology. J Clin Pathol. 1997;50:805-810.
108 Tisserand P, Fouquet C, Marck V, et al. ThinPrep-processed fine-needle samples of breast are effective material for RNA- and DNA-based molecular diagnosis. Cancer. 2003;99:223-232.
109 Gazagne A, Claret E, Wijdenes J, et al. A fluorospot assay to detect single T lymphocytes simultaneously producing multiple cytokines. J Immunol Methods. 2003;283:91-98.
110 Pu RT, Laitala LE, Alli PM, et al. Methylation profiling of benign and malignant breast lesions and its application to cytopathology. Mod Pathol. 2003;16:1095-1101.
111 O’Leary JJ, Landers RJ, Chetty R. In: situ amplification in cytological preparations. Cytopathology. 1997;8:148-160.
112 Sheer D, Squire J. Clinical applications of genetic rearrangements in cancer. Semin Cancer Biol. 1996;7:25-32.
113 Orba Y, Tanaka S, Nishihara H, et al. Application of laser capture microdissection to cytologic specimens for the detection of immunoglobulin heavy chain rearrangement in patients with malignant lymphoma. Cancer. 2003;99:198-204.
114 Chen J-T, Lane MA, Clark DP. Inhibitors of the polymerase chain reaction in Papanicolaou stain: removal with a simple destaining procedure. Acta Cytol. 1996;40:873-877.
115 Lovchik J, Lane MA, Clark DP. Polymerase chain reaction-based detection of B-cell clonality in the fine needle aspiration biopsy of a thyroid mucosa-associated lymphoid tissue (MALT) lymphoma. Human Pathol. 1997;28:989-992.
116 Schlott T, Nagel H, Ruschenburg I, et al. Reverse transcriptase polymerase chain reaction for detecting Ewing’s sarcoma in archival fine needle aspiration biopsies. Acta Cytol. 1997;41:795-801.
117 Mittledorf CA, Leite KR, Darini E, et al. Optimal recovery of DNA for polymerase chain reaction-based assays from fine-needle aspirates. Acta Cytol. 2002;46:1117-1122.
118 Sidransky D. Nucleic acid-based methods for the detection of cancer. Science. 1997;278:1054-1058.
119 Iwao T, Hiyama E, Yokoyama T, et al. Telomerase activity for the preoperative diagnosis of pancreatic cancer. J Natl Cancer Inst. 1997;89:1621-1623.
120 Heselmeyer-Haddad K, Chaudri N, Stoltzfus P, et al. Detection of chromosomal aneuploidies and gene copy number changes in fine-needle aspirates is a specific, sensitive, and objective genetic test for the diagnosis of breast cancer. Cancer Res. 2002;62:2365-2369.
121 Staudt LM. Molecular diagnosis of the hematologic cancers. N Engl J Med. 2003;348:1777-1785.
122 Symmans WF, Ayers M, Clark EA, et al. Total RNA yield and microarray gene expression from fine-needle aspiration biopsy and core-needle biopsy samples of breast carcinoma. Cancer. 2003;97:2960-2971.
123 Mountford CE, Lean CL, MacKinnon WB, et al. The use of proton MR in cancer pathology. In: Webb GA, editor. Annual reports on NMR spectroscopy, vol. 27. London: Academic Press; 1993:173-216.
124 Russel P, Lean CL, Delbridge L, et al. Proton magnetic resonance and human thyroid neoplasia I. Discrimination between benign and malignant neoplasms. Am J Med. 1994;96:383-388.
125 Lansdorp PM, Verwoerd NP, van de Riijke FM, et al. Heterogeneity in telomere length of human chromosomes. Human Mol Genet. 1996;5:685-691.
126 Fetsch PA, Simone NL, Bryant-Greenwood PK, et al. Proteomic evaluation of archival cytologic material using SELDI affinity mass spectrometry. Am J Clin Pathol. 2002;118:870-876.
127 Bibbo M. How technology is reshaping the practice of nongynecologic cytology: frontiers of cytology symposium. Acta Cytol. 2007;51:123-152.
128 Clark DP. Seize the opportunity: underutilization of fine-needle aspiration biopsy to inform targeted cancer therapy decisions. Cancer. 2009;117:289-297.