36 Feline eosinophilic plaque
INTRODUCTION
Eosinophilic granuloma complex represents a reaction pattern often seen in cats mostly with an allergic or ectoparasitic aetiology.
Eosinophilic granuloma complex includes three different entities – eosinophilic granuloma, indolent ulcer and eosinophilic plaque – which, although they have distinct clinical appearances, may have common aetiologies. It is not unusual to see more than one syndrome on the same animal, either at the same time or sequentially. In a small number of cases, cytological examination and a good response to antibacterial therapy confirms bacterial involvement.
CASE HISTORY
Given that eosinophilic granuloma complex is a reaction pattern rather than a diagnosis, a detailed history is invaluable in helping to determine the underlying cause. It is important, therefore, to take a full history, which includes the signalment, age of onset, management and any previous treatments.
In this case the relevant history was:
CLINICAL EXAMINATION
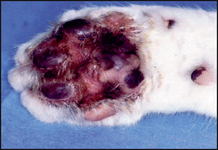
In cats with eosinophilic plaque, the lesions are usually located on the ventral abdomen and medial aspects of the thighs. They appear as circular raised, eroded or ulcerated plaques, ranging from 1 to 2 cm in diameter, but they may coalesce into a large lesion several centimetres in length. Uncommonly, lesions may appear on the feet, which usually present with crusting, exudation, erythema and swelling of the plantar or the interdigital skin (Fig. 36.1).
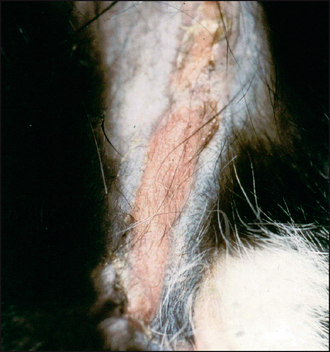
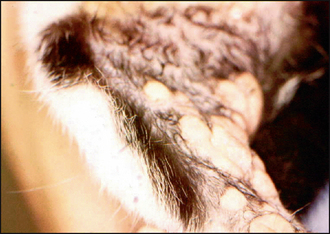
Other variants of the eosinophilic granuloma complex may be found on the same individual, e.g. ‘linear granuloma’, which is seen as a raised chord-like linear plaque, with a yellowish surface, on the caudal aspect of the thighs (Fig. 36.2). Indolent (rodent) ulcer may also be seen as an area of ulceration on the upper lip. Some individuals may also have lesions on the hard or soft palates.
Generally, affected cats do not show any signs of systemic illness, but they may have varying degrees of peripheral lymph node enlargement.
CASE WORK-UP
It cannot be stressed enough that eosinophilic granuloma is not a diagnosis but a reaction pattern, and therefore the aim, particularly in cases such as this where the disease was recurrent, is to identify and correct the underlying cause. Amongst other tests, this usually involves therapeutic and diet trials to rule out the involvement of ectoparasitic and microbial diseases, and adverse food reactions. However, the clinician should be aware when undertaking such trials that despite correcting the underlying cause (e.g. avoidance of food allergens in a food allergic cat), the eosinophilic lesions may still not resolve. This is because the lesion is a source of cytokines and other factors (see ‘Aetiopathogenesis’) that not only cause tissue damage, but continue to stimulate eosinophil production in the bone marrow, recruit inflammatory cells to the damaged tissue, increase eosinophil survival time and perpetuate the lesion. Thus, it may be necessary to achieve resolution by therapeutic intervention in addition to investigation of the underlying cause. As might be expected, this complicates assessment of the response to therapeutic and diet trials. These are complex cases and it is important to warn the client at the outset of the time and commitment involved in making a definitive diagnosis.
In the first instance, the following investigations were carried out:
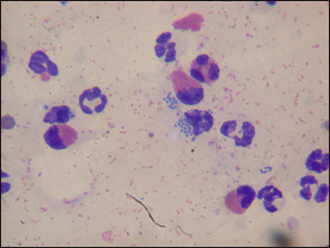
Figure 36.4 Eosinophils, neutrophils and intracellualr coccoid bacteria.
(From August J (2005): Consultations in Feline Internal Medicine, 5th Edition. Saunders, Oxford, with permission of Elsevier.)
The histological findings were:
These changes were deemed to be consistent with eosinophilic granuloma and they ruled out any neoplasia.
The cytological evidence showed evidence of bacterial infection. Even though this was a coagulase-negative organism, and pending the results of the histopathological examination, 3 weeks of amoxicillin/clavulanate was prescribed at a dosage of 15 mg/kg b.i.d. Although following this treatment there was no evidence of bacterial infection on repeat cytology, the lesions showed no evidence of resolution.
The next step in the investigation was a restricted diet trial to rule out an adverse food reaction. For the reasons already described, the cat was prescribed medication to aid resolution of lesions, before starting the trial. This consisted of ciclosporin (5 mg/kg s.i.d.; see ‘Treatment’ section). Prior to starting treatment, further blood samples were taken for FIV, FeLV and toxoplasma serology, which were all negative. Ciclosporin therapy resulted in gradual resolution of lesions over a period of 4 weeks. Treatment was continued for a further 7 weeks, initially at 5 mg/kg every second day for 4 weeks and then for the last 3 weeks at 5 mg/kg twice weekly.
After 8 weeks of ciclosporin therapy and at the same time as reducing to twice weekly dosing, the cat was started on a restricted diet trial by feeding a chicken hydrolysate diet. This diet was fed for a total of 8 weeks. After 6 weeks of the diet (and 3 weeks after ciclosporin withdrawal), the cat started to vigorously overgroom the medial thighs once more. This ruled out an adverse food reaction as a cause of the eosinophilic lesions.
The final stage of the investigation was to perform intradermal testing, which gave positive wheal and flare reactions to house dust mites (Dermatophagoides farinae and Dermatophagoides pteronyssinus) and to one storage mite (Tyrophagus putrescentiae). There is no documented withdrawal time for ciclosporin prior to intradermal testing but the 3 weeks allowed in this case was sufficient.
In summary, the steps towards a final diagnosis in this case were:
DIAGNOSIS
The final diagnosis was eosinophilic plaque. The lack of response to the diet trial and ectoparasitic disease along with the positive intradermal test supported the likelihood that the eosinophilic lesions had arisen because of underlying atopic dermatitis.
PROGNOSIS
The prognosis for initial resolution of skin lesions of the eosinophilic granuloma complex is generally good. However, the longer term prognosis for this condition depends on whether the underlying condition can be identified and corrected or controlled. Many cases require some form of lifelong therapy, in this case therapy for atopic dermatitis.
AETIOPATHOGENESIS AND IMMUNOPATHOGENESIS
In cats, the response to pruritus is often eosinophil mediated, and can result in any of the lesions of the eosinophilic granuloma complex or simply self-induced alopecia, excoriations and ulcerations. The exact mechanism by which lesions of the eosinophilic granuloma complex arise is not clear; whether the lesions are a primary eruption or the result of the cat licking the area is yet to be elucidated.
Both haematological and histological evidence suggests that, in cats, eosinophil infiltration, inflammation and consequent tissue damage are important factors in sustaining pruritus, although mast cells and other mononuclear cells also contribute to the damage.
Eosinophils are produced by the bone marrow. Only small numbers are present in the circulation, and most are found in tissues such as the gut, lung and urinary tract. One important role for eosinophils is as a defence mechanism in ectoparasitic and endoparasitic infestations. On activation they release toxic granule proteins and free radicals, which can kill microorganisms and parasites, but in allergic reactions are responsible for tissue damage. They also induce the synthesis of chemical mediators such as prostaglandins, leucotrienes and cytokines. In turn, these mediators amplify the inflammatory response by activating epithelial cells and recruiting and activating more eosinophils.
The activation and degranulation of eosinophils is normally strictly regulated as inappropriate activation is harmful to the individual, so very few eosinophils are produced in the absence of infection or other immune stimulus. However, in cats, both infection and allergic skin disease activate Th2 lymphocytes, resulting in increased secretion of IL-5, which stimulates the bone marrow to increase the production of eosinophils. A second set of controls then regulates their infiltration into the tissue (eosinophil chemotaxis is controlled by eotaxin-1 (CCL11), eotaxin-2 (CCL24) and eotaxin-3 (CCL26); the eotaxin receptor (CCR3) on eosinophils is a member of the chemokine family of receptors, which can bind to other chemokines, and this binding can also activate eosinophils).
Recent studies support that the damage caused by eosinophils is associated with enzymes, toxic proteins and cytokines, and not collagenolysis as was previously thought. In fact, collagen microfibrils are unaffected. Eosinophils produce enzymes, toxins and cytokines such as:
Eosinophils also secrete lipid mediators such as leucotrienes (causing smooth muscle contraction, increased vascular permeability and increased mucus secretion) and platelet-activating factor (attracts leucocytes, amplifies production of lipid mediators, and activates neutrophils, eosinophils and platelets).
EPIDEMIOLOGY
In one study on allergic disease in cats, about a third had eosinophilic granulomatous lesions. Although there is no age, sex or breed predilections, some cats may have a genetic predisposition to develop lesions of the eosinophilic granuloma complex. The seasonality and the incidence of the disease in individuals, or within a group, will depend on the causative underlying conditions.
TREATMENT AND TREATMENT OPTIONS
Most cats at the time of presentation have lesions resulting from the damage caused by the activation and degranulation of eosinophils. Whilst the diagnosis of the underlying condition is important in preventing and controlling the disease in the long term, resolving the damage caused by the eosinophils is more important in the short term.
The initial treatment should be symptomatic, aimed at limiting and resolving the eosinophil damage and any secondary infections. For the specific underlying aetiology, the treatment or management will depend on its cause, i.e. lifelong flea control in the case of FAD, food avoidance in case of adverse food reaction or allergen-specific immunotherapy in cases of atopic dermatitis.
Symptomatic treatments aimed at limiting and resolving the damage caused by eosinophil infiltration include glucocorticoids and cyclosporin.
Glucocorticoids
Glucocorticoids are the first-line treatment for lesions of the eosinophilic granuloma complex. In general, the long-term use of glucocorticoids should be avoided but they may be the most appropriate treatment for those cats with seasonal lesions or for those owners with limited financial resources.
Injectable glucocorticoids are commonly used in practice; in particular, methylprednisolone acetate is used at the dose of 5 mg/kg i.m., 2–4 weeks apart for a maximum of three injections. Careful patient monitoring is advisable during this period, due to the incidence of side-effects to depot glucocorticoids as there is the potential for serious adverse effects such as diabetes mellitus, iatrogenic hyperadrenocorticism and congestive cardiac failure.
For long-term therapy, an alternate-day glucocorticoid regimen is safer than daily treatment because it allows the hypothalamic–adrenal–pituitary axis to recover on the ‘day off’. Thus, the shorter-acting, orally administered glucocorticoids, prednisolone or methylprednisolone, are the most appropriate drugs to use in this respect. Furthermore, in individuals where there is concern about adverse effects, they are preferred because they can be easily discontinued. Another incentive to use oral, rather than injectable, glucocorticoids is that in the authors’ experience, apparent treatment failures with injectable preparations will often respond to oral therapy.
Oral dexamethasone or triamcinolone are alternatives to prednisolone for refractory lesions, but these steroids have a high potency and an increased risk of adverse effect.
Cats have fewer glucocorticoid receptors, are more resistant to side-effects of glucocorticoids and generally require higher dosages of glucocorticoids than dogs. Apparent treatment failures are commonly the result of inadequate dosing, particularly with lesions of the eosinophilic granuloma complex.
Initial suggested dosages to achieve remission of lesions are as follows:
Once lesions are in remission, the dosage should be gradually tapered to the lowest level that maintains remission (see Chapter 17). Aim to reduce the frequency of administration to alternate days if using prednisolone or methylprednisolone and perhaps every third day for dexamethasone or triamcinolone.
Ciclosporin
Ciclosporin (5 mg/kg orally once daily) is useful for intractable corticosteroid-resistant eosinophilic granuloma complex lesions; however, it is not licensed for use in cats and it requires the owner’s written, informed consent. In addition, routine screening tests are advised (see ‘Case work-up’ section). An improvement with this drug is usually reasonably rapid, but, in the authors’ experience, a few animals will have immediate side-effects, such as soft faeces and diarrhoea, and occasionally vomiting. Other longer-term side-effects may include gingival hyperplasia, papillomatosis, and the activation of latent viral and Toxoplasma infections.
Other immunosuppressive drugs
Chlorambucil (0.1–0.2 mg/kg every 24 hours) or gold salt aurothioglucose (0.1 mg/kg i.m., weekly) may help in recalcitrant cases. If one is not familiar with the use of these drugs, the monitoring required and their potential adverse effects, referral should be considered.
Megosterol and medroxyprogestrone
In the past, megosterol acetate and medroxyprogesterone have been used to manage eosinophilic granulomas. However, the risks of using these drugs outweigh their benefits and, with the increased availability of some of the newer immunomodulating drugs, they should not be used.
Antihistamines
Antihistamines may be valuable to control pruritus in some cases, but only after the eosinophilic tissue damage is resolved (see Table 36.1). They are extremely unlikely to be of any benefit in the face of active eosinophilic granuloma lesions.
Table 36.1 Commonly used antihistamines in cats
| Antihistamine | Dosage and frequency | Side-effects (many of these are reported in humans but could occur in cats) |
|---|---|---|
| Chlorphenamine | 2–4 mg total dose b.i.d. | Drowsiness, diarrhoea, vomiting and anorexia |
| Diphenhydramine | 1–2 mg/kg b.i.d.–t.i.d. | Urine retention and sedation |
| Clemastine | 0.05–0.01 mg/kg b.i.d. | Sedation |
| Hydroxyzine | 1.0–2.0 mg/kg t.i.d. | Sedation |
| Amitriptyline | 1.0–2.0 mg/kg b.i.d. | Sedation, dry mouth, diarrhoea, vomiting, excitability, hypotension, increased appetite and weight gain |
| Cyproheptadine | 0.5–1.0 mg/kg t.i.d. | Sedation, polyphagia and weight gain |
None of these drugs hold a veterinary licence and the owner’s consent is needed for their use.
Essential fatty acids
Omega N3 and N6 fatty acids are usually prescribed concurrently with antihistamines. Because different manufacturers produce different concentrations, it is best to use their guidelines on dosing; however, higher dosages appear to increase efficacy.
Other therapies
Surgical excision, cryotherapy, laser therapy and radiotherapy have also been used with varying degrees of success; however, unless the underlying condition is successfully managed, the lesions have a tendency to recur either at the same or different sites.
Treatment in this case
In this case, the cat was treated with ciclosporin, as the lesion did not respond to increased doses and frequency of methylprednisolone acetate. The lesion had almost resolved after 4 weeks and entirely after 7 weeks (see ‘Case work-up’ section). Once the final diagnosis of atopic dermatitis had been made, long-term management with monthly injections of allergen-specific immunotherapy (see Chapter 6) was initiated.
Although there are no specific nursing aspects, there may be an involvement in educating the owner on the proper use of an Elizabethan collar in the early stages of the treatment. If oral medication is prescribed, some owners may require instruction and/or help in administering the tablets.
The role of secondary infection is often ignored in these cases. Cytology is a rapid, inexpensive and effective way of assessing whether there is an infection or not, but if the results are unclear a swab sample should be submitted for culture and sensitivity testing.
It is always important to bear in mind the role of the eosinophil. A frequent mistake is to assume that if an adverse food reaction is suspected, then a hypoallergenic diet alone will resolve the lesions, or in cases of atopic dermatitis allergen-specific therapy alone will suffice. The key to the successful management of this condition lies in limiting and resolving eosinophil damage prior to instituting long-term management.