Chapter 4 Effects of positioning and mobilization
INTRODUCTION
The purpose of this chapter is to provide a framework for clinical decision-making in the management of patients with cardiopulmonary dysfunction, with special emphasis on positioning and mobilization. ‘Cardiopulmonary dysfunction’ refers to impairment of one or more steps in the oxygen transport pathway. First, the oxygen transport pathway and the factors that contribute to impairment of oxygen transport are described. Second, three clinically significant effects of positioning and mobilization are distinguished:
 to prevent the negative effects of restricted mobility, particularly those that adversely affect oxygen transport.
to prevent the negative effects of restricted mobility, particularly those that adversely affect oxygen transport.In addition, the physiological and scientific rationale for use of positioning and mobilization for each of the above effects is described. Conceptualizing cardiopulmonary dysfunction as impairment of the steps in the oxygen transport pathway and exploiting positioning and mobilization as primary interventions in remediating this impairment will maximize physiotherapy efficacy. Emphasis is placed on impairment of oxygen transport given that such impairment in large part determines disability and handicap (Verbrugge & Lette 1993), as defined by the World Health Organization (1980), secondary to cardiopulmonary dysfunction.
The following terms (Ross & Dean 1989) have been adopted in this chapter:
CONCEPTUAL FRAMEWORK FOR CLINICAL DECISION-MAKING
The oxygen transport pathway
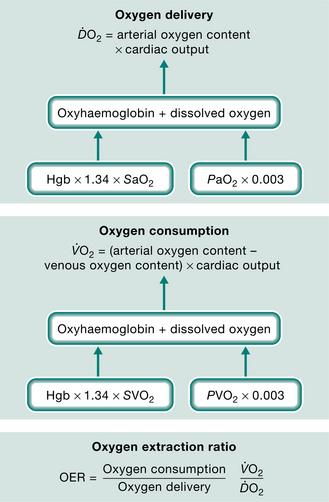
Optimal cardiopulmonary function and gas exchange reflect the optimal matching of oxygen demand and supply (Dantzker 1983, Weber et al 1983). Oxygen delivery and oxygen consumption based on demand are essential components of the oxygen transport system. Figure 4.1 shows the components of oxygen delivery (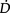 O2, namely arterial oxygen content and cardiac output (CO), and the components of oxygen consumption (
O2, namely arterial oxygen content and cardiac output (CO), and the components of oxygen consumption (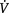 O2), namely the arteriovenous oxygen content difference and CO. In health,
O2), namely the arteriovenous oxygen content difference and CO. In health, 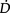 O2is approximately four times greater than
O2is approximately four times greater than 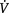 O2at rest so there is considerable oxygen reserve that is drawn upon during times of increased metabolic demand such as exercise, stress, illness and repair. Because of the large reserve,
O2at rest so there is considerable oxygen reserve that is drawn upon during times of increased metabolic demand such as exercise, stress, illness and repair. Because of the large reserve, 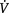 O2is thought to be normally supply-independent. This reserve capacity, however, becomes compromised secondary to acute and chronic pathological conditions. In patients who are critically ill and
O2is thought to be normally supply-independent. This reserve capacity, however, becomes compromised secondary to acute and chronic pathological conditions. In patients who are critically ill and 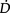 O2 is severely compromised,
O2 is severely compromised, 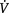 O2 may be supply-dependent until
O2 may be supply-dependent until  O2 reaches a critical threshold, i.e. the level at which metabolic demands are met (Phang & Russell 1993). Below this critical threshold, patients are increasingly dependent on anaerobic metabolism reflected by increased minute ventilation, respiratory exchange ratio and serum lactate levels.
O2 reaches a critical threshold, i.e. the level at which metabolic demands are met (Phang & Russell 1993). Below this critical threshold, patients are increasingly dependent on anaerobic metabolism reflected by increased minute ventilation, respiratory exchange ratio and serum lactate levels.
The efficiency with which oxygen is transported from the atmosphere along the steps of the oxygen transport pathway to the tissues determines the efficiency of oxygen transport overall (Fig. 4.2). The steps in the oxygen transport pathway include ventilation of the alveoli, diffusion of oxygen across the alveolar capillary membrane, perfusion of the lungs, biochemical reaction of oxygen with the blood, affinity of oxygen with haemoglobin, cardiac output, integrity of the peripheral circulation and oxygen extraction at the tissue level (Wasserman et al 1987). At rest, the demand for oxygen reflects basal metabolic requirements. Metabolic demand changes normally in response to gravitational (positional), exercise and psychological stressors. When one or more steps in the oxygen transport pathway are impaired secondary to cardiopulmonary dysfunction, oxygen demand at rest and in response to stressors can be increased significantly. Impairment of one step in the pathway may be compensated by other steps, thereby maintaining normal gas exchange and arterial oxygenation. With severe impairment involving several steps, arterial oxygenation may be reduced, the work of the heart and lungs increased, tissue oxygenation impaired and, in the most extreme situation, multiorgan system failure may ensue.
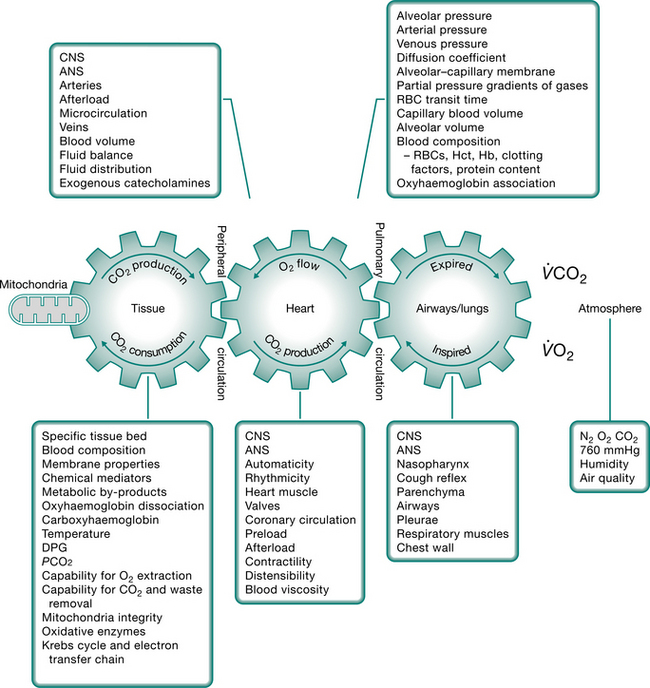
Figure 4.2 A scheme of the components of ventilatory–cardiovascular–metabolic coupling underlying oxygen transport. CNS, central nervous system; ANS, autonomic nervous system; DPG, diphosphoglycerate; RBC, red blood cell; Hct, haematocrit; Hb, haemoglobin.
(Modified from Wasserman et al 1987)
While the oxygen transport pathway ensures that an adequate supply of oxygen meets the demands of the working tissues, the carbon dioxide pathway ensures that carbon dioxide, a primary by-product of metabolism, is eliminated. This pathway is basically the reverse of the oxygen transport pathway in that carbon dioxide is transported from the tissues, via the circulation, to the lungs for elimination. Carbon dioxide is a highly diffusible gas and is readily eliminated from the body. However, carbon dioxide retention is a hallmark of diseases in which the ventilatory muscle pump is operating inefficiently or the normal elastic recoil of the lung parenchyma is lost.
Factors contributing to cardiopulmonary dysfunction
Cardiopulmonary dysfunction, in which oxygen transport is threatened or impaired, results from four principal factors:
Box 4.1 Factors contributing to cardiopulmonary dysfunction, i.e. factors that compromise or threaten oxygen transport
(Adapted from Dean 1993, Dean and Ross 1992a and Ross and Dean 1992)
An analysis of the factors that contribute to cardiopulmonary dysfunction provides the basis for assessment and prescribing the parameters of positioning and mobilization, to enhance oxygen transport for a given patient. The treatment is directed at the specific underlying contributing factors. In some cases, e.g. low haemoglobin, the underlying impairment of oxygen transport cannot be affected directly by physical intervention. However, mobilization and exercise can improve aerobic capacity in patients with anaemia, a factor not directly modifiable by non-invasive physiotherapy interventions, by increasing the efficiency of other steps in the oxygen transport pathway (Williams 1995). Further, even though some factors are not directly modifiable by non-invasive physiotherapy interventions, they influence treatment outcome, and thus need to be considered when planning, modifying and progressing treatment. Fluid imbalance for example is the result of many factors, including reduced muscle activity (Koomans & Boer 1997). Physiotherapy that incorporates body position and exercise challenges may have a profound effect on fluid balance by maintaining fluid balance between the intra- and extravascular compartments. Research is needed to address this compelling proposition.
Ageing and weight deserve special consideration. Patients who are older have progressively lower lung function, and oxygen transport reserve capacity overall (Rossi et al 1996). These changes predispose this population, who tend to constitute the majority of patients in the intensive care unit (ICU), to complications. In addition, obesity often complicates the clinical picture of patients in the ICU: the heavier the patient, the greater the risk. Body positioning and mobilization are particularly important is offsetting the deleterious effects of being recumbent with the added physical burden of adipose tissue surrounding the chest wall (Beck 1998).
Multisystem organ dysfunction and failure may lead to or result from cardiopulmonary dysfunction; thus they are associated with significant mortality and mor bidity. In these conditions, multiple factors impair multiple steps in the oxygen transport pathway, so identifying which steps are affected and amenable to physiotherapy interventions is central to optimal treatment outcome (Dean 1994b, 2006a).
Consultation with the ICU team, including a nutritionist, is critical to enhancing physiotherapy outcomes (Dean & Perme 2006). Increased metabolic demands of patients in the ICU, coupled with the demands of interventions, necessitate appropriate nutritional support to prevent or address malnutrition. Such screening has been shown to lead to better outcomes from ICU in terms of reduced complications, reduced hospital stays and reduced costs (Reid & Allard-Gould 2004).
Anaemaia associated with critical illness (Shander 2004) is a concern for the physiotherapist whose goal is to optimize  O2. Haemoglobin is fundamental to optimal
O2. Haemoglobin is fundamental to optimal  O2, however, optimal management of anaemia is not performed universally. Special attention should be paid to haemoglobin status and its remediation should be a focus of discussion by the multidisciplinary team.
O2, however, optimal management of anaemia is not performed universally. Special attention should be paid to haemoglobin status and its remediation should be a focus of discussion by the multidisciplinary team.
Assessment and monitoring
The physiotherapist needs to be particularly skilled at assessment and monitoring in the ICU both for safety reasons and when monitoring treatment response. Response to treatment can vary from minute to minute. The intervention is prescribed and progressed, and modified as needed during a treatment. Assessment has been addressed in Chapter 1 and includes a range of measures and indices of each step in the oxygen transport pathway (Dean 1999). Further, the physiotherapist needs to recognize that oxygen transport deficits and threats are mediated through impairments of virtually all organ systems not only the lungs and heart (Dean 1997). Consistent with the World Health Organization’s Definition of Health (World Health Organization 2006) and the International Classification of Function (World Health Organization 2002), consideration is also given to assessing activity and health-related quality of life measures even for patients in the ICU setting. Central to prescription and progression of treatment is the patient’s response. For safe and optimal outcomes, response-driven treatment progression is paramount, rather than blind adherence to a protocol-driven intervention. This approach helps to provide seamless care from the ICU back to the community with reduced risk of re-admission to the hospital, in the interest of lifelong health.
THERAPEUTIC EFFECTS OF POSITIONING AND MOBILIZATION
To improve oxygen transport in acute cardiopulmonary dysfunction
Positioning and mobilization have profound acute effects on cardiovascular and cardiopulmonary function and hence on the up-regulation of oxygen transport capacity (Table 4.1). These effects translate into improved gas exchange overall: reduction in the fraction of inspired oxygen, pharmacological and ventilatory support (Burns & Jones 1975, Dean 2006b, 2006c, Svanberg 1957). Such effects need to be exploited in the management of acute cardiopulmonary dysfunction with the use of positioning and mobilization as primarytreatment interventions to enhance oxygen transport and as between-treatment interventions (Dean & Ross 1992b). The physiotherapist’s role is to prescribe these interventions judiciously, to optimize gas exchange and oxygen transport overall. This role is distinct from routinepositioning and mobilization often performed jointly by the physiotherapy and nursing staff. The aim of routine positioning and mobilization is primarily to reduce the adverse effects of restricted mobility, including pulmonary complications, bedsores and contractures. Critical-illness neuropathies and myopathies can lead to irreversible consequences and need to be prevented or at least detected early and managed.
Table 4.1 Acute effects of upright positioning and mobilization on oxygen transport
| Systemic response | Stimulus | |
|---|---|---|
| Positioning (supine to upright) | Mobilization | |
| Cardiopulmonary | ||
| Cardiovascular | ||
AP, anteroposterior; ↑, increases; ↓, decreases; A-aO2, alveolar-arterial oxygen gradient; 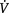 A/
A/ , alveolar-ventilation perfusion; PaO2, partial pressure of oxygen in arterial blood
, alveolar-ventilation perfusion; PaO2, partial pressure of oxygen in arterial blood
(Adapted from Dean and Ross (1992a) and Imle and Klemic (1989))
To simulate the normal ‘physiologic’ body position, the primary goal of physiotherapy is to get the patient upright and moving. Mobilization and exercise are the most physiologic and potent interventions to optimize oxygen transport and aerobic capacity and so need to be exploited with every patient. Body positioning, however, is discussed in this chapter first because a patient cannot be in a position uninfluenced by gravity. Furthermore, a patient’s oxygen transport status reflects the body position assumed regardless of whether the position is part of a treatment regimen, a routine positioning regimen or assumed randomly by the patient.
Non-invasive care in the form of physiotherapy has a particularly important place in the contemporary ICU unit. Advances in medical management have contributed to the avoidance of ICU admissions in patients who would have been admitted to the ICU a couple of decades ago. Similar advances have also contributed to patient survival; many of those who survive today may not have survived a couple of decades ago. Furthermore, advances in ventilatory support including non-invasive mechanical ventilation have helped to titrate ventilatory support more effectively to each patient, and in turn, reduce the need for intubation and invasive ventilation, and the iatrogenic effects of invasive ventilatory support. These advances have increased the demand on physiotherapy to optimize 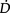 O2, and minimize undue
O2, and minimize undue 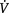 O2. Priming the oxygen transport system with progressive gravitational and exercise stress is the primary responsibility of the physiotherapist in the ICU. This requires a high level of expertise in monitoring and judiciously challenging the oxygen transport system through prescriptively challenging the patient, while minimizing risk to haemodynamic and ventilatory status. As patient survival has improved, the severity of the illness of patients in the ICU has increased, and patients can be kept alive longer. Thus, the physiotherapist needs to assess patients early, in order to establish the appropriate timing for intervention. Weaning or freeing patients from mechanical ventilation is a priority from the beginning. Weaning failure is not uncommon given the severity of the patients’ status, and warrants careful consideration by the team as a whole in helping to prevent failure in both those patients who are non-invasively and invasively ventilated, and contributing to weaning success (Moretti et al 2000, Squadrone et al 2004). However, the physiotherapist needs to do everything possible to optimize
O2. Priming the oxygen transport system with progressive gravitational and exercise stress is the primary responsibility of the physiotherapist in the ICU. This requires a high level of expertise in monitoring and judiciously challenging the oxygen transport system through prescriptively challenging the patient, while minimizing risk to haemodynamic and ventilatory status. As patient survival has improved, the severity of the illness of patients in the ICU has increased, and patients can be kept alive longer. Thus, the physiotherapist needs to assess patients early, in order to establish the appropriate timing for intervention. Weaning or freeing patients from mechanical ventilation is a priority from the beginning. Weaning failure is not uncommon given the severity of the patients’ status, and warrants careful consideration by the team as a whole in helping to prevent failure in both those patients who are non-invasively and invasively ventilated, and contributing to weaning success (Moretti et al 2000, Squadrone et al 2004). However, the physiotherapist needs to do everything possible to optimize 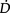 O2through intervention, and minimize the need for mechanical ventilation, particularly invasive ventilation because it is associated with poorer clinical outcomes (Bernieh et al 2004).
O2through intervention, and minimize the need for mechanical ventilation, particularly invasive ventilation because it is associated with poorer clinical outcomes (Bernieh et al 2004).
Mechanical ventilation, both non-invasive and invasive, needs to be thoroughly understood by the physiotherapist in terms of advantages and disadvantages, and these may differ depending on the patient’s status and needs (Brochard 2003). The physiotherapist should be particularly interested in promoting non-invasive ventilation as it facilitates body positioning and mobilization. These interventions have evidence-based support for optimizing oxygen delivery when prescribed appropriately. When a patient is intubated and receiving invasive mechanical ventilation, this in no way precludes the prescription of these interventions. Patients decondition rapidly when gravitational and exercise stressors are removed. It is particularly important to offset these effects in patients with a history of prolonged debility before admission, and elderly patients (Sevransky & Haponik 2003).
Extubation success is a primary concern of the physiotherapist. Failure to extubate is associated with prolonged hospital stay and, in turn, healthcare cost (Seymour et al 2004). Body positioning and mobilization maintain or increase general body conditioning, which may be argued is fundamental to successful weaning from mechanical ventilation. This may be particularly important for patients with neuromuscular conditions who are weak and at high risk of aspiration (Vianello et al 2000). While the objective of optimizing conditioning may initially appear a premature priority given the status of patients who are severely ill in the ICU, cough strength that can be correlated to general body strength, or the converse, a weak cough related to general debility, can be predictive of extubation outcome (Khamiees et al 2001). Being able to tolerate a spontaneous breathing trial is key to eventual weaning. Furthermore, maintaining spontaneous breathing may help minimize sedation and improve cardiopulmonary function in some patients, for example patients with acute lung injury and receiving airway pressure-released ventilation (Putensen et al 2001). Weaning needs to be tailored to each patient and should not be unnecessarily prolonged (Nevins & Epstein 2001).
Figure 4.3 illustrates positioning upright and mobilization of a patient in the ICU. The progression is based on ongoing assessment and monitoring, with a gravitational challenge followed by progressive movement. The rationale and the details of ongoing monitoring follow.
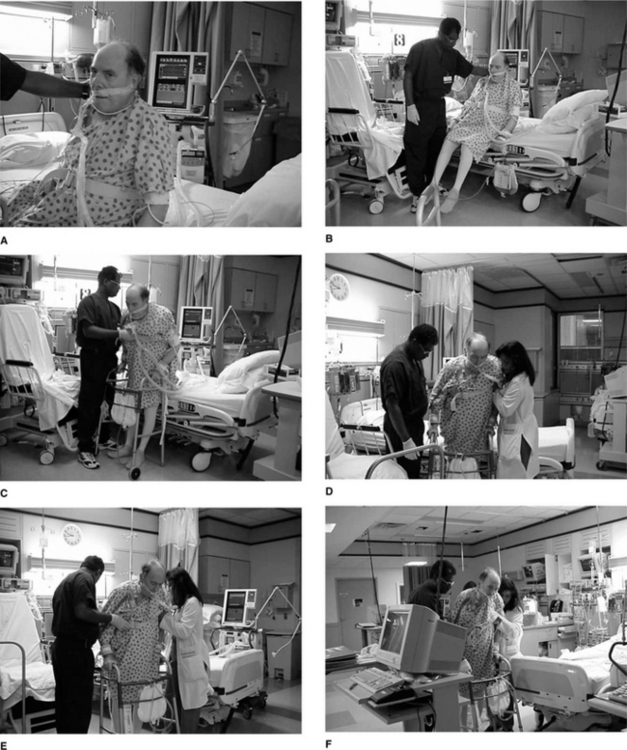
Figure 4.3 (A–F) Example of mobilizing a patient to a self-supported upright sitting position. Mobilizing a critically ill patient needs to be a priority whenever possible. Short frequent sessions to the erect position (sitting or standing if possible) with continual monitoring of the patient’s response should be the goal. As the patient progresses, sessions increase in intensity and duration and reduce in frequency.
Positioning
Physiological and scientific rationale.
The distributions of ventilation (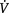 A), perfusion (
A), perfusion ( ) and ventilation and perfusion matching in the lungs are primarily influenced by gravity and therefore body position (Clauss et al 1968, West 1962, 1977). The goal is to reduce closing volume and optimize functional residual capacity (Manning et al 1999). The intrapleural pressure becomes less negative down the upright lung. Thus, the apices have a greater initial volume and reduced compliance than the bases. Because the bases are more compliant in this position, they exhibit greater volume changes during ventilation. In addition to these gravity-dependent inter-regional differences in lung volume, ventilation is influenced by intra-regional differences, which are dependent on regional mechanical differences in the compliance of the lung parenchyma and the resistance to airflow in the airways. Perfusion increases down the upright lung such that the
) and ventilation and perfusion matching in the lungs are primarily influenced by gravity and therefore body position (Clauss et al 1968, West 1962, 1977). The goal is to reduce closing volume and optimize functional residual capacity (Manning et al 1999). The intrapleural pressure becomes less negative down the upright lung. Thus, the apices have a greater initial volume and reduced compliance than the bases. Because the bases are more compliant in this position, they exhibit greater volume changes during ventilation. In addition to these gravity-dependent inter-regional differences in lung volume, ventilation is influenced by intra-regional differences, which are dependent on regional mechanical differences in the compliance of the lung parenchyma and the resistance to airflow in the airways. Perfusion increases down the upright lung such that the 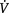 A/
A/ ratio in the apices is disproportionately high compared with that in the bases. Ventilation and perfusion matching is optimal in the mid-lung region. Manipulating body position alters both inter-regional and intra-regional determinants of ventilation and perfusion and their matching. When choosing specific positions to enhance arterial oxygenation for a given patient, the underlying pathophysiology impairing cardiopulmonary function, the effects of bed rest/recumbency and restricted mobility, extrinsic factors related to the patient’s care and intrinsic factors related to the patient need to be considered.
ratio in the apices is disproportionately high compared with that in the bases. Ventilation and perfusion matching is optimal in the mid-lung region. Manipulating body position alters both inter-regional and intra-regional determinants of ventilation and perfusion and their matching. When choosing specific positions to enhance arterial oxygenation for a given patient, the underlying pathophysiology impairing cardiopulmonary function, the effects of bed rest/recumbency and restricted mobility, extrinsic factors related to the patient’s care and intrinsic factors related to the patient need to be considered.
Although the negative effects of the supine position have been well documented for several decades (Dean & Ross 1992b, Dripps & Waters 1941), supine or recumbent positions are frequently assumed by patients in hospital. These positions are non-physiologic and are associated with significant reductions in lung volumes and flow rates and increased work of breathing (Craig et al 1971, Hsu & Hickey 1976). The decrease in functional residual capacity (FRC) contributes to closure of the dependent airways and reduced arterial oxygenation (Ray et al 1974). This effect has long been known to be accentuated in older persons (Leblanc et al 1970), patients with cardiopulmonary disease (Fowler 1949), patients with abdominal pathology, smokers and individuals who are obese.
The haemodynamic consequences of the supine position are also remarkable. The gravity-dependent increase in central blood volume may precipitate vascular congestion, reduced compliance and pulmonary oedema (Blomqvist & Stone 1983, Sjostrand 1951). The commensurate increase in stroke volume increases the work of the heart (Levine & Lown 1952). Within 6 hours, a compensatory diuresis can lead to a loss of circulating blood volume and orthostatic intolerance: i.e. haemodynamic intolerance to the upright position. Bed rest deconditioning has been attributed to this reduction in blood volume and the impairment of the volume-regulating mechanisms rather than physical deconditioning per se (Hahn-Winslow 1985). Thus, the upright position is essential to maximize lung volumes and flow rates and this position is the only means of optimizing fluid shifts such that the circulating blood volume and the volume-regulating mechanisms are maintained. The upright position coupled with movement is necessary to promote normal fluid regulation and balance (Lamb et al 1964).
The upright position is a potent stimulus to the sympathetic nervous system. This is an important clinical effect, which offsets impaired blood volume and pressure-regulating mechanisms secondary to recumbency (Hahn-Winslow 1985). Stimulation of the sympathetic nervous system has been reported to augment the effects of potent sympathomimetic pharmacological agents such that the dosages of these drugs can be reduced (Warren et al 1983). The reduction or elimination of sympathomimetic drugs is an important outcome of non-invasive physiotherapy interventions.
Side-to-side positioning is frequently used in the clinical setting. If applied in response to assessment rather than routinely (Chuley et al 1982), the benefits derived from such positioning can be enhanced. Adult patients with unilateral lung disease may derive greater benefit when the affected lung is uppermost (Remolina et al 1981). Markedly improved gas exchange without deleterious haemodynamic effects has been reported for patients with severe hypoxaemia secondary to pneumonia (Dreyfuss et al 1992). Arterial oxygen tension is increased secondary to improved ventilation of the unaffected lung when this lung is dependent, in the adult patient. Patients with uniformly distributed bilateral lung disease may derive greater benefit when the right lung is lowermost (Zack et al 1974). In this case, arterial oxygen tension is increased secondary to improved ventilation of the right lung, which may reflect the increased size of the right lung compared with the left and that, in this position, the heart and adjacent lung tissue are subjected to less compression. Improved gas exchange through non-invasive interventions can reduce or eliminate the need for supplemental oxygen, both of which are primary treatment outcomes. Although various studies have shown beneficial effects of side lying, body positioning should be based on multiple considerations including the distribution of disease if optimal results are to be obtained.
The prone position has long been known to have considerable physiological justification in patients with cardiopulmonary compromise (Douglas et al 1977), even those who are critically ill with acute respiratory failure (Bittner et al 1996, Chatte et al 1997, Mure et al 1997), those with acute respiratory distress syndrome (Vollman 2004), and patients with trauma-induced adult respiratory distress syndrome (Fridrich et al 1996). The beneficial effects of the prone position on arterial oxygenation may reflect improved lung compliance secondary to stabilization of the anterior chest wall, tidal ventilation, diaphragmatic excursion, functional residual capacity (FRC) and reduced airway closure (Dean 1985, Pelosi et al 1998). In a dog model of acute lung injury, however, improved PaO2in prone has been attributed to a reduced shunt fraction (Albert et al 1987). A variant of the prone position, prone abdomen-free, has shown additional benefits over prone abdomen-restricted. In the prone abdomen-free position, the patient is positioned such that the movement of the abdomen is unencumbered by the bed. This can be achieved either by raising the patient’s body in relation to the bed so that the abdomen falls free or by using a bed with a hole cut out at the level of the abdomen. Despite compelling evidence to support the prone position, it may be poorly tolerated in some patients or may be contraindicated in patients who are haemodynamically unstable. In these situations, intermediate positions approximating prone may produce many of the beneficial effects and minimize any potential hazard.
The use of the prone position, like other body positions, needs to be prescribed based on the patient’s presentation as well as underlying pathophysiology. The response to prone positioning may differ, for example in early adult respiratory distress syndrome the effect of prone positioning may depend on whether the primary insult is of a pulmonary or non-pulmonary nature (Lim et al 2001). The benefits of the prone position in respiratory distress syndrome have been reported to be additive to invasive medical interventions (Jolliet et al 1997). Prone positioners that passively change the patient’s body position to prone have been reported to have comparable benefit (Vollman & Bander 1996).
Extreme body position changes can be beneficial in patients who are unable to participate actively in mobilization. Although the literature to date has been mostly related to children in the ICU, the results with respect to improving oxygenation are compelling (Casadro-Flores et al 2002). Such positioning includes dramatic shifts from supine to prone, and prone to supine, as opposed to supine to side lying. Abdomen-free prone positioning has been shown to have greater benefit than abdomen-restricted prone positioning in infants. The half prone position in adults, which displaces the viscera forward, allowing greater caudal displacement of the diaphragm, may elicit comparable benefits in adults.
Positioning for drainage of pulmonary secretions may be indicated in some patients (Kirilloff et al 1985). Historically, these positions have been based on the anatomical arrangement of the bronchopulmonary segments to facilitate drainage of a particular segment (Chapter 5). The bronchiole to the segment of interest is positioned perpendicular to facilitate drainage with the use of gravity. The efficacy of postural drainage compared with deep breathing and coughing induced with mobilization/exercise and repositioning of the patient has not been established. The fact that mobilization impacts on more steps in the oxygen transport pathway including the airways, to effect secretion clearance, supports the exploitation of mobilization coupled with deep breathing manoeuvres and coughing as a primarytreatment intervention.
Physiotherapists need to consider two aspects of body positioning when the goal is to optimize oxygen transport. One is to select and apply specific body positions based on the patient’s presentation, laboratory test results and radiographic imaging. The other is to elicit physiologic ‘stir-up’ with the use of extreme body positions (Dean 2006c, Dripps & Waters 1941). The purpose is to effect the normal gravitational stress on cardiopulmonary and cardiovascular function that is experienced in health. This is best simulated if patients are changed from one extreme position to another, for example supine to prone or upright, rather than from half to full side lying which is associated with a lesser ‘stir-up’ effect. Patients who are haemodynamically unstable require greater monitoring during extreme position changes and may not tolerate some position changes well or for too long a period. Thus, based on assessment and patient response, frequent extreme position changes may be preferable to minimal shifts in body position in order to optimize cardiovascular, pulmonary and haemodynamic function. Ongoing monitoring is essential to ensure responses are optimal and adverse responses averted.
Assessment and treatment planning.
Body positioning – the specific positions selected, the duration of time spent in each position and the frequency with which the position is assumed – is based on a consideration of the factors that contribute to cardiopulmonary dysfunction and treatment response. Understanding of the physiology of cardiopulmonary and cardiovascular function and the effects of disease highlights certain positions that are theoretically ideal. However, these positions need to be modified or may be contraindicated for a given patient, based on other considerations (see Box 4.1). For example, if extreme positional changes are contraindicated, small degrees of positional rotation, performed frequently, can have significant benefit on gas exchange and arterial oxygenation. A three-quarters prone position may produce favourable results when the full prone position is contraindicated or is not feasible. This modification may simulate the prone abdomen-free position, which has been shown to augment the effect of the traditional prone abdomen-restricted position (Douglas et al 1977). Furthermore, a three-quarters prone position may be particularly beneficial in patients with obese or swollen abdomens who may not tolerate other variations of the prone position. With attention to the patient’s condition, invasive lines and leads and appropriate monitoring, a patient can be aggressively positioned.
The time that a patient spends in a position and the frequency with which that position is assumed, over a period of time, are based on the indications for the position and treatment outcome. Objective measures of the various steps that are compromised in the oxygen transport pathway, as well as indices of oxygen transport overall, are used in making these decisions. Subjective evaluation based on clinical judgement is also important. A specific position can be justified, provided there is objective and subjective evidence of improvement. Signs and symptoms of deterioration need to be monitored so that deleterious positions can be avoided and deterioration secondary to excessive time in any one position can be detected. Prolonged duration in any single position will inevitably lead to compromise of the function of dependent lung zones and impaired gas exchange.
The ratio of treatment to between-treatment time is low. Typically, between-treatment time consists of some combination of positioning and mobilization. Positioning and mobilizing patients between treatments may be incorporated as an extension of treatment. Patients require monitoring and observation during these periods, as well as during treatment. Between-treatment time may include the use of maximally restful positions that do not compromise oxygen transport. Lastly, patients are positioned and mobilized between treatments to prevent the negative effects of restricted mobility and recumbency.
Special consideration (for example, with respect to specific positioning and the use of supports) needs to be given to positioning patients who are comatose or paralysed because their joints and muscles are relatively unprotected and prone to trauma. Positions need to be selected that avoid injury to unprotected head, neck and limbs.
Progression.
Progression of positioning involves new positions or modification of previous positions and modification of the duration spent in each position and the frequency with which each position is assumed over a period of time. These clinical decisions are based on the factors that contribute to cardiopulmonary dysfunction and objective and subjective indices of change in the patient’s cardiopulmonary status. With improvement in cardiopulmonary status, the patient spends more time in erect positions and is mobilized more frequently and independently.
Mobilization
Physiological and scientific rationale.
The negative effects of recumbency during critical illness, combined with the patient’s life-threatening pathology, are compounded by progressive deconditioning. These problems need to be anticipated by the physiotherapist. A patient for whom short-term mechanical ventilation is anticipated may be one who eventually requires long-term ventilation. Thus, a whole-body rehabilitation approach warrants being considered from the start (Martin et al 2005) to prevent as well as address recumbency and deconditioning effects and their compounded effects on the patient’s other problems.
Compared with long-term exercise, the mechanisms underlying adaptation of the oxygen transport system to acute exercise, from session to session and day to day, are less well understood. Although these mechanisms have yet to be elucidated, the responses to acute exercise are well documented. The acute response to mobilization/exercise reflects a commensurate increase in oxygen transport to provide oxygen to the working muscles and other organs. The increase is dependent on the intensity of the mobilization/exercise stimulus. The demand for oxygen and oxygen consumption (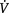 O2) increases as exercise continues, with commensurate increases in minute ventilation (
O2) increases as exercise continues, with commensurate increases in minute ventilation (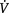 E), that is, the amount of air inhaled per minute, cardiac output and oxygen extraction at the tissue level. Relatively low intensities of mobilization can have a direct and profound effect on oxygen transport in patients with acute cardiopulmonary dysfunction (Dean 2006a, Dull & Dull 1983, Lewis 1980) and need to be instituted early after the initial pathological insult or surgery (Orlava 1959, Wenger 1982). The resulting exercise hyperpnoea, the increase in
E), that is, the amount of air inhaled per minute, cardiac output and oxygen extraction at the tissue level. Relatively low intensities of mobilization can have a direct and profound effect on oxygen transport in patients with acute cardiopulmonary dysfunction (Dean 2006a, Dull & Dull 1983, Lewis 1980) and need to be instituted early after the initial pathological insult or surgery (Orlava 1959, Wenger 1982). The resulting exercise hyperpnoea, the increase in 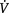 E, is effected by an increase in tidal volume and breathing frequency (Zafiropoulos et al 2004). In addition, ventilation and perfusion matching is augmented by the distension and recruitment of lung zones with low ventilation and low perfusion. Spontaneous exercise-induced deep breaths are associated with improved flow rates and mobilization of pulmonary secretions (Wolff et al 1977). In clinical populations, these effects elicit spontaneous coughing. When mobilization is performed in the upright position, the anteroposterior diameter of the chest wall assumes a normal configuration compared with the recumbent position in which the anteroposterior diameter is reduced and the transverse diameter is increased. In addition, diaphragmatic excursion is favoured, flow rates augmented and coughing is mechanically facilitated. The work of breathing may be reduced with caudal displacement of the diaphragm and the work of the heart is minimized by the displacement of fluid away from the central circulation to the legs. Thus, despite increased metabolic demands of mobilization and exercise, the goal is to ensure that this increased demand is not wasteful and the demand can be met by the supply.
E, is effected by an increase in tidal volume and breathing frequency (Zafiropoulos et al 2004). In addition, ventilation and perfusion matching is augmented by the distension and recruitment of lung zones with low ventilation and low perfusion. Spontaneous exercise-induced deep breaths are associated with improved flow rates and mobilization of pulmonary secretions (Wolff et al 1977). In clinical populations, these effects elicit spontaneous coughing. When mobilization is performed in the upright position, the anteroposterior diameter of the chest wall assumes a normal configuration compared with the recumbent position in which the anteroposterior diameter is reduced and the transverse diameter is increased. In addition, diaphragmatic excursion is favoured, flow rates augmented and coughing is mechanically facilitated. The work of breathing may be reduced with caudal displacement of the diaphragm and the work of the heart is minimized by the displacement of fluid away from the central circulation to the legs. Thus, despite increased metabolic demands of mobilization and exercise, the goal is to ensure that this increased demand is not wasteful and the demand can be met by the supply.
With respect to cardiovascular effects, acute mobilization/exercise increases cardiac output (CO) by increasing stroke volume and heart rate. This is associated with increased blood pressure and increased coronary and peripheral muscle perfusion. Despite their energetic demands, mobilization and exercise may augment outcomes even in acute respiratory failure (Wong 2000, 2004), and patients requiring left ventricular assist devices (Perme 2006). When titrated carefully, based on the patient’s response, mobilization can be performed safely with potential benefit, including avoidance of mechanical ventilation.
Passive movement of the limbs may stimulate deep breaths and heart function; however, this effect is considered minimal compared with active movement (West 2004). In addition, there is little scientific evidence to support any additional benefit from various facilitation techniques (Bethune 1975). Thus, time allocated to the use of passive manoeuvres may compete with time for positioning and mobilization, that is, interventions with demonstrated clinical efficacy. Although passive movements have a relatively small effect on cardiopulmonary function, they have several important benefits for neuromuscular and musculoskeletal function which support their use, provided they do not replace active movement.
Current medical research has been focusing on protection of the heart in patients in the ICU. Such measures include new inotropic agents, supporting the metabolic activity of the heart which has a high 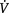 O2/
O2/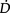 O2 ratio, conservative administration of blood products and greater use of mechanical support devices (Bosenberg & Royston 2002). Irrespective of these advances, the place of physiotherapy is to exploit the person’s capacity for
O2 ratio, conservative administration of blood products and greater use of mechanical support devices (Bosenberg & Royston 2002). Irrespective of these advances, the place of physiotherapy is to exploit the person’s capacity for 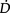 O2 through priming the oxygen transport system and optimizing aerobic conditioning regardless of how small an effect may result.
O2 through priming the oxygen transport system and optimizing aerobic conditioning regardless of how small an effect may result.
Assessment and treatment planning.
For practical and ethical considerations, the mobilization plan for the patient with acute cardiopulmonary dysfunction cannot be based on a standardized exercise test, as is the case for patients with chronic conditions. However, response to a mobilization/exercise stimulus can be assessed during a mobilization challenge test, that is, during the patient’s routine activities such as turning or moving in bed, activities of daily living or responding to routine nursing and medical procedures. Comparable to prescribing exercise for the patient with chronic cardiopulmonary dysfunction, the parameters are specifically defined so that the stimulus is optimally therapeutic. The optimal stimulus is that which stresses the oxygen transport capacity of the patient and effects the greatest adaptation without deterioration or distress.
To promote adaptation of the steps in the oxygen transport pathway to the stimulation of acute mobilization in patients who are acutely ill, the stimulus is administered in a comparable manner to that in an exercise programme prescribed for patients with chronic cardiopulmonary dysfunction and who are medically stable. The components include a pre-exercise period, a warm-up period, a steady-state period, a cool-down period and a recovery period (Blair et al 2005). These components optimize the response to exercise by preparing the cardiopulmonary and cardiovascular systems for steady-state exercise and by permitting these systems to re-establish resting conditions following exercise. The cool-down period, in conjunction with the recovery period, ensures that exercise does not stop abruptly, and allows for biochemical degradation and removal of the by-products of metabolism. Mobilization consists of discrete warm-up, steady-state and cool-down periods; the components need to be identified, even in the patient with a very low functional capacity; for example, a patient who is critically ill and may be only able to sit up over the edge of the bed. In such cases, preparing to sit up constitutes a warm-up period for the patient; the stimulus of sitting unsupported for several minutes while being aroused and encouraged to talk or be interactive non-verbally, if mechanically ventilated, constitutes a steady-state period; returning to bed constitutes the cool-down period. In the recovery period, observation of the patient continues to ensure that mobilization is tolerated well and that the indices of oxygen transport return to resting levels. This information is then used as the basis for mobilization in the next treatment.
Monitoring.
As clinical exercise physiologists, physiotherapists can benefit from the use of metabolic measures in monitoring and evaluating mobilization and exercise as interventions. Although these measures are mostly used in exercise laboratories, indirect calorimetry is a tool used by nutritionists in the ICU to estimate dietary needs. The concept that the metabolic demands of patients in the ICU can vary considerably from day to day has been well established (Vermeij et al 1989). Physiotherapists can benefit greatly from knowledge of the metabolic demands of their interventions when superimposed on such daily variation; thus, these measures should be used routinely by physiotherapists who may stress the patient energetically more than any other team member (Brandi et al 1997).
Valid and reliable monitoring practices provide the basis for defining the parameters of mobilization, assessing the need for progression and defining the adaptation or training-sensitive zone. In the case of patients who are acutely ill and particularly those who are critically ill, the training-sensitive zone refers to the physiologic zone where conditioning of the oxygen transport system is stimulated. This zone will be based on many more variables than for the patient who is medically stable, and will be at a markedly reduced intensity. Sessions will be shorter in duration as well as less intense. This ensures that the oxygen transport system and aerobic conditioning are progressively challenged commensurate with the patient’s responses and within the margins of safety. Ongoing monitoring is essential given that subjecting patients to mobilization/exercise stimulation is inherently risky, particularly for patients with cardiopulmonary dysfunction. Indices of overall oxygen transport in addition to indices of the function of the individual steps in the oxygen transport pathway provide a detailed profile of the patient’s cardiopulmonary status. In critical care settings, the physiotherapist has access to a wide range of measures to assess the adequacy of gas exchange. Minimally, in the general ward setting, measures of breathing frequency, arterial blood gases, arterial saturation, heart rate, blood pressure and clinical observation provide the basis for ongoing assessment, mobilization/exercise and progression. With appropriate attention to the patient’s condition, invasive lines and leads and appropriate monitoring, a patient can be aggressively mobilized and ambulated (see Fig. 4.3).
A fundamental requirement in defining the parameters for mobilization is that the patient’s oxygen transport system is capable of increasing the oxygen supply to meet an increasing metabolic demand. If not, mobilization is absolutely contraindicated and the treatment of choice to optimize oxygen transport is body positioning. However, in the case of a patient being severely haemodynamically unstable or at risk of being so, for example hypotensive or on a high FiO2, even the stress of positioning may be excessive. Thus, although patients who are critically ill may be treated aggressively, every patient has to be considered individually and in conjunction with the team’s goals as a whole, otherwise the patient may deteriorate or be seriously endangered. Physiotherapy interventions including mobilization alter metabolic demand. This has particular implications for the patient with blood sugar abnormalities, as the stress of illness and hospital admission can compound this abnormality. Discussion of sliding-scale insulin with the team may be warranted in a patient whose blood sugars fluctuate widely (Kee et al 2006). Monitoring of glycaemic control is important in that poor control is associated with poor outcomes in patients who have diabetes and those who are not known to have diabetes (Laver & Padkin 2005, Rady et al 2005).
Progression.
Progression and modification of the mobilization stimulus usually occur more rapidly in the management of the patient with acute cardiopulmonary dysfunction compared with the progression of the exercise stimulus for the patient with chronic illness. The status of patients who are acutely ill can vary considerably within minutes or hours. Whether the mobilization stimulus is increased or decreased in intensity depends on the patient’s status and altered responses to mobilization. The mobilization stimulus is adjusted to remain optimal despite the patient’s changing metabolic needs. Capitalizing on narrow windows of opportunity for therapeutic intervention must be exploited 24 hours a day with respect to the type of mobilization stimulus, its intensity, duration and frequency, particularly in the critically ill patient. At times, the intensity and duration of the intervention may be quite minimal, yet cumulatively produce a marked improvement in oxygen transport efficiency.
Fluctuations in blood gases should correct with appropriate treatment pacing, and the provision of supplemental oxygen during treatment. However, if the PaO2/FiO2remains below 250, or the alveolar–arterial (A-a) gradient remains widened, these signs may portend deterioration including acute renal failure (Chawla et al 2005). Acute renal failure is associated with a high mortality rate (Ympa et al 2005).
The ‘immovable’ patient.
Given the well-documented negative effects of restricted mobility, the ‘immovable’ patient deserves special consideration. Although bed rest or activity restriction is ordered for patients frequently without reservation, the risks need to be weighed against the benefits (Allen et al 1999). Restricted mobility coupled with recumbency constitutes a death knell for many patients who are severely compromised. Thus, an order for bed rest needs to be evaluated and challenged to ensure that this order is physiologically justified. For many years, chair nursing has been advocated based on solid physiologic evidence, yet this knowledge has lagged behind integration into practice.
There are some instances where a patient cannot be moved, for example with an open chest, with intra-aortic balloon pumps, on neuromuscular blockade, or hypotensive. Patients receive neuromuscular blockade to facilitate mechanical ventilation. The effects of prolonged blockade are being better documented, for example neuropathies and myopathies (Gehr & Sessler 2001, Hund 1999). Periodic peripheral nerve stimulation may need to be conducted to offset these effects. However, body positioning needs to be exploited to relieve the effect of prolonged pressure on muscles and nerve, in addition to skin, that can complicate this clinical picture.
Kinetic beds and chairs.
Advances in furniture technology to facilitate positioning and mobilizing patients have lagged behind advances in clinical medicine, particularly in the critical care area. Conventional hospital beds are designed to be stationary and their widths and heights are often non-adjustable, making it difficult for the patient to get in and out of bed. Kinetic beds and chairs have become increasingly available over the past decade but they are not widely used clinically. These devices were originally designed to facilitate positioning and moving heavy and comatose patients. Some beds are designed to rotate on their long axis from side to side over several minutes. Other beds simulate a side-to-side movement with inflation and deflation of the two sides of an air-filled mattress. Although these beds have potential cardiopulmonary benefit (Powers & Daniels 2004), they do not replace active positioning and movement.
Mechanically adjustable bedside chairs and stretcher chairs constitute an important advance. These chairs adjust to a flat horizontal surface that can be matched to bed height and positioned beneath the patient lying on the bed. The device with the patient on top is then wheeled parallel to the bed where it can be adjusted back into a chair and thus the patient assumes a seated position. The degree of recline can be altered to meet the patient’s needs – for example those who are very weak but capable of bearing some weight and some transferring activities – safety considerations and for comfort. This chair also facilitates returning the patient to bed. Comparable to these chairs are beds that can be converted into a chair while the patient is lying down. These avoid the negative effects of using tilt tables where the fluid shifts caudally are extreme and more risky by comparison.
Kinetic beds have a place in the management of some patients. Patients who are immobile because of induced coma or marked haemodynamic instability may benefit. Patients with multiple injuries have also been shown to benefit substantially from the continual movement of kinetic beds (Stiletto et al 2000). In addition, kinetic beds may increase safety to physiotherapists and nurses in obviating the need for shifting and moving heavy patients. The disadvantages of kinetic beds and chairs include the expense and the potential for over-reliance on them. Without these devices, a heavy patient may require several people and several minutes to position in a chair that may be tolerated only for a few minutes. However, the cardiopulmonary benefits of the stimulation of preparing to be moved, the reflex attempts of the patient to assist and adjust to changing position, as well as actually sitting upright in a chair are not reproduced by bed positioning alone or by a kinetic bed. Each case, in terms of therapeutic benefit and safety, needs to be evaluated individually. Research is needed to determine the indications and potential benefits of kinetic beds and chairs so that they can be used judiciously in the clinical setting as an integral therapeutic intervention.
To improve oxygen transport in post-acute and chronic cardiopulmonary dysfunction
In post-acute and chronic cardiopulmonary dysfunction, a primary consequence of impaired oxygen transport is reduced functional work capacity (Belman & Wasserman 1981, Wasserman & Whipp 1975). Work capacity can be improved with long-term exercise which improves the efficiency of the steps in the oxygen transport pathway and promotes compensation within the pathway as well as by other mechanisms. To optimize the patient’s response, exercise can be carried out in judicious body positions in which oxygen transport is favoured.
Exercise is the treatment of choice for patients whose impaired oxygen transport has resulted from chronic cardiopulmonary dysfunction. Body positioning may have a place in severely ill patients by optimizing oxygen transport at rest. Barach & Beck (1954) reported that emphysematous patients were less breathless, had reduced accessory muscle activity and had a significant reduction in ventilation when positioned in a 16° head down position. Some patients exhibited greater symptomatic improvement than in the upright position with supplemental oxygen. Classic relaxation positions, for example leaning forward with the forearms supported, can also be supported physiologically. Coupling such physiologically justifiable positions with mobilization/exercise will augment the benefits of exercise.
Physiological and scientific rationale.
Although the physiological responses to long-term exercise in patients with chronic cardiopulmonary disease may differ from those in healthy persons, patients can significantly improve their functional work capacity (Table 4.2). In healthy people, an improvement in aerobic capacity reflects improved efficiency of the steps in the oxygen transport pathway to adapt to the increased oxygen demands imposed by exercise stress. This adaptation is effected by both central (cardiopulmonary) and peripheral (at the tissue level) changes (Dean 2006b, Wasserman & Whipp 1975). Such aerobic conditioning is characterized by a training-induced bradycardia secondary to an increased stroke volume and increased oxygen extraction capacity of the working muscle. These adaptation or training responses result in an increased maximal oxygen uptake and maximal voluntary ventilation and reduced submaximal 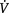 E, cardiac output, heart rate, blood pressure and perceived exertion. However, patients with chronic lung disease are often unable to exercise at the intensity required to elicit an aerobic training response. Their functional work capacity may be improved by other mechanisms, for example desensitization to breathlessness, improved motivation, improved biomechanical efficiency, increased ventilatory muscle strength and endurance or some combination (Belman & Wasserman 1981, Bernard et al 1999). Aerobic capacity can be increased through peripheral adaptations (Gosselin et al 2003), but peripheral myopathies in patients with primary lung and heart disease have been reported (Storer 2001). Patients with chronic heart disease, such as those with severe infarcts, may be able to train aerobically; however, training adaptation primarily results from peripheral rather than central factors commensurate with the level of impairment (Bydgman & Wahren 1974, Larsen et al 2001, 2002). Most patients in the ICU have oxygen transport dysfunction or threat to this life-preserving system. Patients with ventilatory failure secondary to neuromuscular and musculoskeletal conditions also constitute a significant proportion of patients in the ICU. These patients may not have primary heart or lung disease, yet their lives may be threatened due to secondary effects of neuromuscular or musculoskeletal dysfunction.
E, cardiac output, heart rate, blood pressure and perceived exertion. However, patients with chronic lung disease are often unable to exercise at the intensity required to elicit an aerobic training response. Their functional work capacity may be improved by other mechanisms, for example desensitization to breathlessness, improved motivation, improved biomechanical efficiency, increased ventilatory muscle strength and endurance or some combination (Belman & Wasserman 1981, Bernard et al 1999). Aerobic capacity can be increased through peripheral adaptations (Gosselin et al 2003), but peripheral myopathies in patients with primary lung and heart disease have been reported (Storer 2001). Patients with chronic heart disease, such as those with severe infarcts, may be able to train aerobically; however, training adaptation primarily results from peripheral rather than central factors commensurate with the level of impairment (Bydgman & Wahren 1974, Larsen et al 2001, 2002). Most patients in the ICU have oxygen transport dysfunction or threat to this life-preserving system. Patients with ventilatory failure secondary to neuromuscular and musculoskeletal conditions also constitute a significant proportion of patients in the ICU. These patients may not have primary heart or lung disease, yet their lives may be threatened due to secondary effects of neuromuscular or musculoskeletal dysfunction.
Table 4.2 Chronic effects of mobilization/exercise on oxygen transport
| Systemic response | Effect |
|---|---|
| Cardiopulmonary | |
| Cardiovascular | |
| Tissue level |
↑, increases; ↓, decreases; 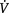 O2, oxygen consumption
O2, oxygen consumption
Planning an exercise programme.
The exercise programme is based on the principle that oxygen delivery and uptake are enhanced in response to an exercise stimulus which is precisely defined for an individual in terms of the type of exercise, its intensity, duration, frequency and the course of the training programme. These parameters are based on an exercise test in conjunction with assessment findings. Exercise tests are performed on a cycle ergometer, treadmill or with a walk test (Noonan & Dean 2000). The general procedures and protocols are standardized to maximize the validity and reliability of the results (Blair et al 2005, Dean et al 1989). The principles of, and guidelines for, exercise testing and training patients with chronic lung and heart disease have been well documented (Dean 2006d). The training-sensitive zone is defined by objective and subjective measures of oxygen transport determined from the exercise test. The components of each exercise training session include baseline, warm-up, steady-state portion, cool-down and recovery period (Blair et al 2005, Dean 1993). The cardiopulmonary and cardiovascular systems are gradually primed for sustaining a given level of exercise stress, while in addition the musculoskeletal system adapts correspondingly. Following the steady-state portion of the training session, the cool-down period permits a return to the resting physiological state. Cool-down and recovery periods are essential for the biochemical degradation and elimination of the metabolic by-products of exercise.
Progression.
Progression of the exercise programme is based on a repeated exercise test. This is indicated when the exercise prescription no longer elicits the desired physiological responses – specifically when the steady-state work rate consistently elicits responses at the low end or below the lower limit of the training-sensitive zone for the given indices of oxygen transport. This reflects maximal adaptation of the steps in the oxygen transport pathway to the given exercise stimulus. The degree of conditioning achieved is precisely matched to the demands of the exercise stimulus imposed.
To prevent the negative effects of restricted mobility
Although physiologically distinct, the effects of immobility are frequently confounded by the effects of recumbency in the hospitalized patient. Patients with restricted mobility include those under sedation, those with acute spinal cord injury, and critically ill patients who are unable to be mobilized because of haemodynamic instability due to other causes. Restricted mobility and the concomitant reduction in exercise stress affect virtually every organ system in the body, with profound effects on the cardiovascular and neuromuscular systems. Patients who are recumbent but rousable warrant vigilant monitoring. Recumbency and the elimination of the vertical gravitational stress exert effects primarily on the cardiovascular and cardiopulmonary systems (Blomqvist & Stone 1983, Dock 1944, Harrison 1944). The most serious consequences of restricted mobility and recumbency are those resulting from the effects on the cardiopulmonary and cardiovascular systems and hence on oxygen transport. Although other consequences of restricted mobility, e.g. increased risk of infection, skin breakdown and deformity, may not constitute the same immediate threat to oxygen transport and tissue oxygenation, they can have significant implications with respect to morbidity and mortality. Thus, restricted mobility and recumbency need to be minimized. Mobility and the upright position should be maximized to avert the negative consequences of restricted mobility, the risk of morbidity associated with these effects and the direct and indirect cardiopulmonary and cardiovascular effects. These negative consequences are preventable with frequent repositioning and mobilizing of the patient (Table 4.3). The prevention of these effects is a primary goal of positioning and mobilizing patients between treatments.
Table 4.3 Effects of positioning and mobilization that prevent the negative effects of restricted mobility and recumbency*
| Systemic response | Effect |
|---|---|
| Cardiopulmonary | |
| Cardiovascular | |
| Tissue level |
* Some of the preventative effects of body positioning and mobilization are comparable; however, the magnitude of these effects in response to mobilization tends to be greater than with body positioning
Education of the patient, family and ICU staff regarding early mobilization and frequent body position changes is key. Physiotherapists should work closely with other ICU team members, as well as with the patient and family. Client-focused care including extensive education has become increasingly important and given attention to health-related quality of life even in patients who are critically ill. Loved ones may provide important information about a patient’s quality of life, when that individual is unable to speak for himself.
SUMMARY AND CONCLUSION
Cardiopulmonary dysfunction refers to impairment of one or more steps in the oxygen transport pathway that can impair oxygen transport overall. Thus, a conceptual framework for clinical problem-solving in the management of patients with cardiopulmonary dysfunction, based on oxygen transport, can facilitate the identification of deficits and the directing of treatment to each specific deficit. Factors that can impair the transport of oxygen from the atmosphere to the tissues include cardiopulmonary pathology, bed rest, recumbency and restricted mobility, extrinsic factors related to the patient’s medical care, intrinsic factors related to the patient or a combination of these. Positioning and mobilization are two interventions that have potent and direct effects on several of the steps in the oxygen transport pathway. These interventions have a primaryrole in improving oxygen transport in acute and chronic cardiopulmonary dysfunction and in averting the negative effects of restricted mobility and recumbency, particularly those related to cardiopulmonary and cardiovascular function.
The principal goal of physiotherapy in the management of cardiopulmonary dysfunction is to optimize oxygen transport. A systematic approach to achieving this goal consists of:
Conceptualizing cardiopulmonary dysfunction as deficits in the steps in the oxygen transport pathway and identifying the factors responsible for each impaired step provides a systematic, evidence-based approach to clinical decision-making in cardiopulmonary physiotherapy. Positioning and mobilization can then be specifically directed at the mechanisms underlying cardiopulmonary dysfunction wherever possible. Such an approach will maximize the efficacy of positioning and mobilizing patients with cardiopulmonary dysfunction and enhance the outcome of medical management overall.
Albert RK, Leasa D, Sanderson M, Robertson HT, Hlastala MP. The prone position improves arterial oxygenation and reduced shunt in oleic-acid-induced acute lung injury. American Review of Respiratory Diseases. 1987;138:828-833.
Allen C, Glasziou P, Delman C. Bedrest: a potentially harmful treatment needing more careful evaluation. The Lancet. 1999;354:1229-1233.
Barach AL, Beck GJ. Ventilatory effect of head-down position in pulmonary emphysema. American Journal of Medicine. 1954;16:55-60.
Beck LA. Morbid obesity and spinal cord injury: a case study. Spinal Cord Injury Nurse. 1998;15:3-5.
Belman MJ, Wasserman K. Exercise training and testing in patients with chronic obstructive pulmonary disease. Basics of Respiratory Disease. 1981;10:1-6.
Bernard S, Whittom F, Leblanc P, et al. Aerobic and strength training in patients with chronic obstructive lung disease. American Journal of Respiratory and Critical Care Medicine. 1999;159:896-901.
Bernieh B, Al Hakim M, Boobes Y, Siemkovics E, El Jack H. Outcome and predictive factors of acute renal failure in the intensive care unit. Transplantation Proceedings. 2004;36:1784-1787.
Bethune DD. Neurophysiological facilitation of respiration in the unconscious patient. Physiotherapy Canada. 1975;27:241-245.
Bittner E, Chendrasekhar A, Pillai S. Changes in oxygenation and compliance as related to body position in acute lung injury. American Journal of Surgery. 1996;62:1038-1041.
Blair SN, Painter P, Pate RR, et al. Resource manual for guidelines for exercise testing and prescription, 5th edn., Philadelphia: Lea and Febiger, 2005.
Blomqvist CG, Stone HL. Cardiovascular adjustments to gravitational stress. In: Shepherd JT, Abboud FM, editors. Handbook of physiology. Section 2: circulation, vol 2. Bethesda: American Physiological Society; 1983:1025-1063.
Bosenberg C, Royston D. Protect the heart in the intensive care unit - but how? Current Opinions in Critical Care. 2002;8:417-420.
Brandi LS, Bertolini R, Calafa M. Indirect calorimetry in critically ill patients: clinical applications and practical advice. Nutrition. 1997;13:349-358.
Brochard L. Mechanical ventilation: invasive versus non invasive. European Respiratory Journal Supplement. 2003;47:31s-37s.
Burns JR, Jones FL. Early ambulation of patients requiring ventilatory assistance. Chest. 1975;68:608.
Bydgman S, Wahren J. Influence of body position on the anginal threshold during leg exercise. European Journal of Clinical Investigation. 1974;4:201-206.
Casadro-Flores J, Martinez de Azagra A, Ruiz-Lopez MJ, Ruiz M, Serrano A. Pediatric ARDS: effect of supine-prone postural changes on oxygenation. Intensive Care Medicine. 2002;28:1792-1796.
Chatte G, Sab J-M, Dubois J-M. Prone position in mechanically ventilated patients with severe acute respiratory failure. American Journal of Critical Care Medicine. 1997;155:473-478.
Chawla LS, Abell L, Mazhari R, et al. Identifying critically ill patients at high risk for developing acute renal failure: a pilot study. Kidney International. 2005;68:2274-2280.
Chuley M, Brown J, Summer W. Effect of postoperative immobilization after coronary artery bypass surgery. Critical Care Medicine. 1982;10:176-178.
Clauss RH, Scalabrini BY, Ray RF, Reed GE. Effects of changing body position upon improved ventilation- perfusion relationships. Circulation. 1968;37(Suppl 2):214-217.
Craig DB, Wahba WM, Don HF. ‘Closing volume’ and its relationship to gas exchange in seated and supine positions. Journal of Applied Physiology. 1971;31:717-721.
Dantzker DR. The influence of cardiovascular function on gas exchange. Clinics in Chest Medicine. 1983;4:149-159.
Dean E. Effect of body position on pulmonary function. Physical Therapy. 1985;65:613-618.
Dean E. Bedrest and deconditioning. Neurology Report. 1993;17:6-9.
Dean E. Oxygen transport: a physiologically-based conceptual framework for the practice of cardiopulmonary physiotherapy. Physiotherapy. 1994;80:347-359.
Dean E. Invited commentary on ‘Are incentive spirometry, intermittent positive pressure breathing, and deep breathing exercises effective in the prevention of postoperative pulmonary complications after upper abdominal surgery? a systematic overview and meta-analysis.’ Physical Therapy. 1994;74:10-15.
Dean E. Oxygen transport deficits in systemic disease and implications for physical therapy. Physical Therapy. 1997;77:187-202.
Dean E. Preferred practice patterns in cardiopulmonary physical therapy: a guide to physiologic measures. Cardiopulmonary Physical Therapy Journal. 1999;10:124-134.
Dean E. Optimizing outcomes: relating interventions to an individual’s needs. In Frownfelter D, Dean E, editors: Cardiovascular and pulmonary physical therapy: evidence and practice, 4th edn., St Louis: Mosby, 2006.
Dean E. Mobilization and exercise. In Frownfelter D, Dean E, editors: Cardiovascular and pulmonary physical therapy: evidence and practice, 4th edn., St Louis: Mosby, 2006.
Dean E. Body positioning. In Frownfelter D, Dean E, editors: Cardiovascular and pulmonary physical therapy: evidence and practice, 4th edn., St Louis: Mosby, 2006.
Dean E. Exercise testing and training for individuals with primary cardiopulmonary dysfunction. In Frownfelter D, Dean E, editors: Cardiovascular and pulmonary physical therapy: evidence and practice, 4th edn., St Louis: Mosby, 2006.
Dean E, Perme C. Comprehensive management of individuals in the intensive care unit. In Frownfelter D, Dean E, editors: Cardiovascular and pulmonary physical therapy: evidence and practice, 4th edn., St Louis: Mosby, 2006.
Dean E, Ross J. Oxygen transport: The basis for contemporary cardiopulmonary physical therapy and its optimization with body positioning and mobilization. Physical Therapy Practice. 1992;1:34-44.
Dean E, Ross J. Discordance between cardiopulmonary physiology and physical therapy: toward a rational basis for practice. Chest. 1992;101:1694-1698.
Dean E, Ross J, Bartz J, Purves S. Improving the validity of exercise testing: the effect of practice on performance. Archives of Physical Medicine and Rehabilitation. 1989;70:599-604.
Dock W. The evil sequelae of complete bed rest. Journal of the American Medical Association. 1944;125:1083-1085.
Douglas WW, Rehder K, Froukje BM. Improved oxygenation in patients with acute respiratory failure: the prone position. American Review of Respiratory Disease. 1977;115:559-566.
Dreyfuss D, Djedaini K, Lanore J-J, et al. A comparative study of the effects of almitrine bismesylate and lateral position during unilateral bacterial pneumonia with severe hypoxemia. American Review of Respiratory Disease. 1992;148:295-299.
Dripps RD, Waters RM. Nursing care of surgical patients. I. The ‘stir-up’. American Journal of Nursing. 1941;41:530-534.
Dull JL, Dull WL. Are maximal inspiratory breathing exercises or incentive spirometry better than early mobilization after cardiopulmonary bypass? Physical Therapy. 1983;63:655-659.
Fowler WS. Lung function studies. III. Uneven pulmonary ventilation in normal subjects and patients with pulmonary disease. Journal of Applied Physiology. 1949;2:283-299.
Fridrich P, Krafft P, Hochleuthner H. The effects of long-term prone positioning in patients with trauma-induced adult respiratory distress syndrome. Anesthesia and Analgesia. 1996;83:1206-1211.
Gehr LC, Sessler CN. Neuromuscular blockade in the intensive care unit. Seminars in Respiratory and Critical Care Medicine. 2001;22:175-188.
Gosselin N, Lambert K, Poulain M, et al. Endurance training improves skeletal muscle electrical activity in active COPD patients. Muscle Nerve. 2003;28:744-753.
Hahn-Winslow E. Cardiovascular consequences of bed rest. Heart and Lung. 1985;14:236-246.
Harrison TR. The abuse of rest as a therapeutic measure for patients with cardiovascular disease. Journal of the American Medical Association. 1944;125:1075-1078.
Hsu HO, Hickey RF. Effect of posture on functional residual capacity postoperatively. Anesthesiology. 1976;44:520-521.
Hund E. Myopathy in critically-ill patients. Critical Care Medicine. 1999;27:2544-2547.
Imle PC, Klemic N. Changes with immobility and methods of mobilization. In: Mackenzie CF, editor. Chest physiotherapy in the intensive care unit. 2nd edn. Baltimore: Williams and Wilkins; 1989:188-214.
Jolliet P, Bulpa P, Ritz M, et al. Additive beneficial effects of the prone position, nitric oxide, and almitrine bismesylate on gas exchange and oxygen transport in acute respiratory distress syndrome. Critical Care Medicine. 1997;25:786-794.
Kee CA, Tomalty JA, Cline J, Novick RJ, Stitt L. Change in practice patterns in the management of diabetic cardiac surgery patients. Canadian Journal of Cardiovascular Nursing. 2006;16:20-27.
Khamiees M, Raju P, DeGirolamo A, Amoateng-Adjepong Y, Manthous CA. Predictors of extubation outcome in patients who have successfully completed a spontaneous breathing trial. Chest. 2001;120:1262-1270.
Kirilloff LH, Owens HR, Rogers RM, Mazzocco MC. Does chest physical therapy work? Chest. 1985;88:436-444.
Koomans HA, Boer WH. Causes of edema in the intensive care unit. Kidney International Supplement. 1997;59:S105-S110.
Lamb LE, Johnson RL, Stevens PM. Cardiovascular deconditioning during chair rest. Aerospace Medicine. 1964;23:646-649.
Larsen AI, Lindal S, Aukrust P, et al. Effect of exercise training on skeletal muscle fibre characteristics in men with chronic heart failure. Correlation between muscle alterations, cytokines, and exercise capacity. International Journal of Cardiology. 2002;83:25-32.
Larsen AI, Aarsland T, Kristiansen M, Haugland A, Dickstein K. Assessing the effect of exercise training in men with heart failure; comparison of maximal, submaximal and endurance exercise protocols. European Heart Journal. 2001;22:684-692.
Laver SR, Padkin A. Does hyperglycaemia precede the clinical onset of myocardial ischemia? Resuscitation. 2005;66:237-239.
Leblanc P, Ruff F, Milic-Emili J. Effects of age and body position on airway closure in man. Journal of Applied Physiology. 1970;28:448-451.
Levine SA, Lown B. ‘Armchair’ treatment of acute coronary thrombosis. Journal of the American Medical Association. 1952;148:1365-1369.
Lewis FR. Management of atelectasis and pneumonia. Surgical Clinics of North America. 1980;60:1391-1401.
Lim CM, Kim EK, Lee JS, et al. Comparison of the response to the prone position between pulmonary and extrapulmonary acute respiratory distress syndrome. Intensive Care Medicine. 2001;27:477-485.
Manning F, Dean E, Ross J, Abboud RAT. Lung function in side lying positions compared with supine in older healthy individuals. Physical Therapy. 1999;79:456-466.
Martin V, Hincapie L, Nimbuck M, Gaugham J, Criner G. Impact of whole-body rehabilitation in patients receiving chronic mechanical ventilation. Critical Care Medicine. 2005;33:2255-2265.
Moretti M, Cilione C, Tampieri A, et al. Incidence and causes of non- invasive mechanical ventilation failure after initial success. Thorax. 2000;55:819-825.
Mure M, Martling C-R, Lindahl SGE. Dramatic effect on oxygenation in patients with severe acute lung insufficiency treated in the prone position. Critical Care Medicine. 1997;25:1539-1544.
Nevins ML, Epstein SK. Weaning from prolonged mechanical ventilation. Clinics in Chest Medicine. 2001;22:13-33.
Noonan V, Dean E. Submaximal exercise testing: clinical application and interpretation. Physical Therapy. 2000;80:782-807.
Orlava OE. Therapeutic physical culture in the complex treatment of pneumonia. Physical Therapy Review. 1959;39:153-160.
Pelosi P, Tubiolo D, Mascheroni D. Effects of the prone position on respiratory mechanics and gas exchange during acute lung injury. American Journal of Critical Care Medicine. 1998;157:387-393.
Perme C. Early mobilization of LVAD recipients who require prolonged mechanical ventilation. Texas Heart Institute Journal. 2006;33:130-134.
Phang PT, Russell JA. When does VO 2 depend on VO 2. Respiratory Care. 1993;38:618-630.
Powers J, Daniels D. Turning points: implementing kinetic therapy in the ICU. Nursing Management. 2004;35(Suppl):1-7.
Putensen C, Zech S, Wrigge H, et al. Long-term effects of spontaneous breathing during ventilatory support in patients with acute lung injury. American Journal of Respiratory Care Medicine. 2001;164:43-49.
Rady MY, Johnson DJ, Patel BM, Larson JS, Helmers RA. Influence of individual characteristics on outcome of glycemic control in intensive care unit patients with or without diabetes mellitus. Mayo Clinic Proceedings. 2005;80:1558-1567.
Ray JF, Yost L, Moallem S, et al. Immobility, hypoxemia and pulmonary arteriovenous shunting. Archives of Surgery. 1974;109:537-541.
Reid MB, Allard-Gould P. Malnutrition and the critically ill elderly patient. Critical Care Nursing Clinics of North America. 2004;16:531-536.
Remolina C, Khan AV, Santiago TV, Edelman NH. Positional hypoxemia in unilateral lung disease. New England Journal of Medicine. 1981;304:523-525.
Ross J, Dean E. Integrating physiological principles into the comprehensive management of cardiopulmonary dysfunction. Physical Therapy. 1989;69:255-259.
Ross J, Dean E. Body positioning. In: Zadai C, editor. Pulmonary management in physical therapy. New York: Churchill Livingstone, 1992.
Rossi A, Ganassini A, Tantucci C, Grassi V. Aging and the respiratory system. Aging (Milano). 1996;161(English abstract):143. 8
Sevransky JF, Haponik EF. Respiratory failure in elderly patients. Clinics in Geriatric Medicine. 2003;19:205-224.
Seymour CW, Martinez A, Christie JD, Fuchs BD. The outcome of extubation failure in a community hospital intensive care unit: a cohort study. Critical Care. 2004;8:R322-R327.
Shander A. Anemia in the critically ill. Critical Care Clinics. 2004;20:159-178.
Sjostrand T. Determination of changes in the intrathoracic blood volume in man. Acta Physiologica Scandinavica. 1951;22:116-128.
Squadrone E, Frigerio P, Fogliati C, et al. Non invasive vs invasive ventilation in COPD patients with severe acute respiratory failure deemed to require ventilatory assistance. Intensive Care Medicine. 2004;30:1303-1310.
Stiletto R, Gotzen L, Goubeaud S. Kinetic therapy for therapy and prevention of post-traumatic lung failure. Results of a prospective study of 111 polytrauma patients. Unfallchirurgie. 2000;103:1057-1064. (English abstract)
Storer TW. Exercise in chronic pulmonary disease: resistance exercise prescription. Medicine and Science in Sport and Exercise. 2001;33(7 Suppl):S680-692.
Svanberg L. Influence of position on the lung volumes, ventilation and circulation in normals. Scandinavian Journal of Laboratory Investigation. 1957;25(Suppl):7-175.
Verbrugge LM, Jette AM. The disablement process. Social Sciences and Medicine. 1993;38:1-14.
Vermeij CG, Feenstra BW, van Lanschot JJ, Bruining HA. Day-to-day variation of energy expenditure in critically ill surgical patients. Critical Care Medicine. 1989;17:623-626.
Vianello A, Bevilacqua M, Arcaro G, Gallan F, Serra F. Non-invasive ventilatory approach to treatment of acute respiratory failure in neuromuscular disorders. A comparison with endotracheal intubation. Intensive Care Medicine. 2000;26:384-390.
Vollman KM. Prone positioning in the patient who has acute respiratory distress syndrome: the art and science. Critical Care Nursing Clinics of North America. 2004;16:319-336.
Vollman KM, Bander JJ. Improved oxygenation utilizing a prone positioner in patients with acute respiratory distress syndrome. Intensive Care Medicine. 1996;22:1105-1111.
Warren JB, Turner C, Dalton N, et al. The effect of posture on the sympathoadrenal response to theophylline infusion. British Journal of Clinical Pharmacology. 1983;16:405-411.
Wasserman K, Hansen JE, Sue DY, Whipp BJ. Principles of exercise testing and interpretation. Philadelphia: Lea and Febiger, 1987.
Wasserman K, Whipp BJ. Exercise physiology in health and disease. American Review of Respiratory Disease. 1975;112:219-249.
Weber KT, Janicki JS, Shroff SG, Likoff MJ. The cardiopulmonary unit: the body’s gas transport system. Clinics in Chest Medicine. 1983;4:101-110.
Wenger NK. Early ambulation: the physiologic basis revisited. Advances in Cardiology. 1982;31:138-141.
West JB. Regional differences in gas exchange in the lung of erect man. Journal of Applied Physiology. 1962;17:893-898.
West JB. Ventilation and perfusion relationships. American Review of Respiratory Disease. 1977;116:919-943.
West JB. Respiratory physiology - the essentials, 6th edn., Baltimore: Williams and Wilkins, 2004.
Williams C. Haemoglobin - is more better? Nephrology Dialysis and Transplant. 1995;2(Suppl):48-55.
Wolff RK, Dolovich MB, Obminski G, Newhouse MT. Effects of exercise and eucapnic hyperventilation on bronchial clearance in man. Journal of Applied Physiology. 1977;43:46-50.
Wong WP. Physical therapy for a patient in acute respiratory failure. Physical Therapy. 2000;80:662-670.
Wong WP. Use of body positioning in the mechanically ventilated patient with acute respiratory failure: Application of the Sackett’s rules of evidence. Physiotherapy Theory and Practice. 2004;15:25-41.
World Health Organization. International classification of impairments, disabilities and handicaps. In: A manual for classification relating to the consequences of disease. Geneva: World Health Organization; 1980.
World Health Organization, 2002 International classification of functioning, disability and health. www.sustainable-design.ie/arch/ICIDH-2PFDec-2000.pdf Retrieved July 2007
World Health Organization, 2006 Definition of health. www.who.int/about/definition Retrieved July 2007
Ympa YP, Sakr Y, Reinhart K, Vincent JL. Has mortality from acute renal failure decreased? A systematic review of the literature. American Journal of Medicine. 2005;118:827-832.
Zack MB, Pontoppidan H, Kazemi H. The effect of lateral positions on gas exchange in pulmonary disease. American Review of Respiratory Disease. 1974;110:49-55.
Zafiropoulos B, Alison J, McCarren B. Physiological responses to the early mobilization of the intubated, ventilated abdominal surgery patient. Australian Journal of Physiotherapy. 2004;50:95-100.
American College of Sports Medicine. Guidelines for exercise testing and prescription, 7th edn., Philadelphia: Lea and Febiger, 2005.
Dean E, Frownfelter D. Clinical case study guide to accompany principles and practice of cardiopulmonary physical therapy, 3rd edn., St Louis: Mosby, 1996.
Dean E, Perme C. Intensive care unit management of individuals with primary cardiopulmonary dysfunction. In Frownfelter D, Dean E, editors: Cardiovascular and pulmonary physical therapy: evidence and practice, 4th edn., St Louis: Mosby, 2006.
Dean E, Perme C. Complications, adult respiratory distress syndrome, shock, sepsis and multiorgan system failure. In Frownfelter D, Dean E, editors: Cardiovascular and pulmonary physical therapy: evidence and practice, 4th edn., St Louis: Mosby, 2006.
McArdle WD, Katch FI, Katch VL. Exercise physiology. Energy, nutrition and human performance, 4th edn., Philadelphia: Lea and Febiger, 1996.
West JB. Ventilation, blood flow and gas exchange, 5th edn., Oxford: Blackwell Science, 1990.