Chapter 5 Physiotherapy techniques
Physiotherapy techniques are discussed throughout the book, but some are described in more detail in this chapter.
ACUPUNCTURE
What can be treated with acupuncture?
Acupuncture is recognized by the World Health Organization (WHO) in the treatment of respiratory conditions (Hopwood 1993) and acupuncture techniques can also be used to treat associated symptoms: acute and chronic pain, panic attacks and stress, which are so often a contributing or causative factor. Acupuncture has been found to be safe and potentially effective for bronchial asthma and chronic obstructive pulmonary disease (COPD) (Jobst 1995). It has been reported to help con-trol disabling breathlessness (Linde et al 1986), asthma (Blackwell 2004) and improve lung function and quality of life in COPD (Neumeister et al 1999). Acupuncture used pre- and postoperatively may reduce postoperative pain, the risk of infection and improve recovery rate.
The techniques used and the evidence base
Needling is the biggest cause of adverse effects in acupuncture (Halvorsen et al 1995, Rampes & James 1995). Many respiratory patients have contraindications to needling and to the use of certain body acupuncture points (acupoints). Therefore a combination of non-invasive body and ear acupuncture (auriculotherapy) is used to overcome these limitations. Each can be used on its own, but they are more effective if used together or with another treatment modality. Acupuncture treatment is based on evidence from different levels of research, from systematic reviews (Ezzo 2000) to clinical opinion.
Non-invasive options include acupressure, laser, magnetic and electrical stimulation (Baxter 1994, Kenyon 1983, 1988, Lawrence et al 1998) and have been found to be as effective as needling in the body (Vickers 1986). In auriculotherapy, electrical stimulation was more effective than needling for pain relief (Oleson 1998). Acupressure, where no equipment is required, is an invaluable tool for both therapist and patient, and it can be used as a home treatment to enhance and maintain the effect of treatment. Acupressure was found to reduce dyspnoea and be a useful adjunct for patients undergoing a pulmonary rehabilitation programme (Maa et al 1997). Laser acupuncture was of benefit in the treatment of sinusitis, asthma and pnuemonia (Ridge 2005). Unpublished case studies have also shown that non-invasive methods can be as effective as needling and in some cases, more effective.
How do acupuncture techniques work?
Stimulation of an acupoint activates A-delta, A-beta and C afferents, which inhibit the sensory and affective components of pain via gating and descending inhibitory pathways (Lund et al 2006). Endorphins and serotonin are released which, as well as having an analgesic effect, have been shown to effect cell growth and healing. (Kishi et al 1996, Pakala & Benedict 1998). Stimulation of certain acupoints activates cortical areas of the brain that have controlling effects on symptoms being treated (Zhang 1986). The traditional concept is one of energy flowing within meridians. The energy becomes imbalanced by disease and acupoints are chosen to create a re-balance. Active points that require treatment are detectable by electrical skin resistance measurement and by palpation for tenderness. Acupoints and meridians have a high concentration of gap junctions (Mashanskii et al 1983), facilitating intercellular communication and thereby mediating the effects of acupuncture.
Auriculotherapy (ear acupuncture)
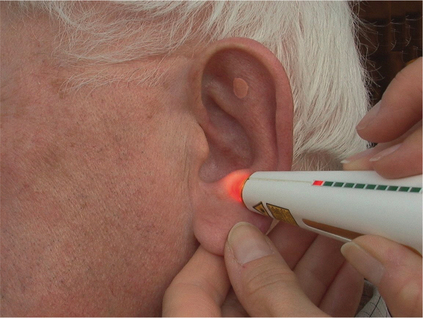
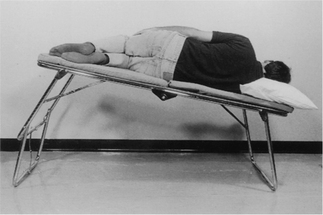
Auriculotherapy (Fig. 5.1) is safer than body acupuncture, quicker and access is easier. It is therefore useful in situations such as critical care. Patients contraindicated to body acupuncture, for example those with heart conditions, can be safely treated via the ear. Certain drugs will reduce the effectiveness of body acupuncture, for example corticosteroids acting as a partial opiate antagonist, but these same drugs do not reduce the effectiveness of auriculotherapy. Every anatomical structure and physiological function of the body is represented by an acupoint on the ear (Nogier 1981, Oleson 1998, Practical Ear Needling Therapy 1980). These points become active when disease is present and are detected by palpation for tenderness, ideal when treating pain; and electrical resistance measurement, ideal for non-painful conditions. Stimulation of these points has an effect on the corresponding body area or function. An electromagnetic balancing is achieved, internal opiates are released and there can be an immediate reflex effect on, for example, pain. Tiny gold-plated magnets can be placed over selected points between treatments, creating constant pressure and a magnetic field to prolong the effectiveness of treatment. The patient can apply additional pressure to obtain further relief of symptoms, thus reducing the frequency and number of visits required. Auriculotherapy is one of the safest and most effective ways of dealing with conditions more complicated than pain. In addition to treatment, it can also be used for prevention and diagnosis (Lichstein et al 1974).
Some useful body points
The following acupoints may be useful for certain respiratory symptoms, but these techniques are contraindicated in pregnant women and people with pacemakers or heart conditions. The points are found by palpating for a tender spot and acupressure is given using gentle circular massage over the point for about a minute.
| CV 17 | On the sternum roughly midway between the nipples. Useful for shortness of breath and in conjunction with breathing exercises, works well by applying straight pressure to the point on exhalation. |
| PC 6 | In the centre of the anterior aspect of the forearm, three fingers’ breadth from the anterior wrist crease. Effective for hyperventilation, stress reduction and relief of nausea and vomiting (Stainton & Neff 1994). |
| LI 4 | Adduct the thumb, find the high point of the interosseus muscle and press towards the second metacarpal. One of the most useful points in the body. Useful for pain relief and stress reduction. |
Acupuncture used appropriately as part of a team approach, whether by needle or non-invasive means, can act as a very effective complementary therapy to help to improve quality of life and reduce potential side effects from drug therapy. It works wells alongside the conventional treatment of respiratory diseases and concomitant symptoms, to enhance the outcome of treatment.
AIRWAY CLEARANCE TECHNIQUES
Airway clearance techniques are used to facilitate mucociliary clearance. Under normal circumstances the mucociliary clearance mechanism is extremely effective and efficient but in the presence of many respiratory diseases and following anaesthesia and surgical procedures, airway clearance techniques may be required to enhance mucociliary clearance. There is as yet no evidence to support the use of any one airway clearance technique over any other (Accurso et al 2004, Prasad & Main 1998, Pryor 2005, Pryor et al 2006). Practice tends to be influenced by culture and patient preference. The airway clearance techniques available to patients may be determined by the skills of the therapist (Hardy et al 1994) and the financing of healthcare provision. When a device, for example positive expiratory pressure (PEP) or oscillating PEP (Acapella®, Flutter® or RC-Cornet®), is included in an airway clearance regimen, meticulous cleaning and thorough drying of the equipment after use is essential to prevent the possibility of infection from the device. Other techniques, for example intermittent positive pressure breathing and glossopharyngeal breathing, may also assist in the clearance of secretions and have additional effects.
If an airway clearance technique is required in the long term, it is important to negotiate rather than prescribe a physiotherapy home programme to increase adherence to treatment (Carr et al 1996). Programmes must be realistic and appropriate general physical activities should be encouraged. Revision of techniques at intervals is important to assess the effectiveness of the regimen and to correct and update techniques as necessary. Currie et al (1986) recognized the importance of reassessment in maintaining adherence to treatment.
Traditionally airway clearance techniques have been associated with head-down tipped positioning, but this is now used only if individual patient benefit is identified on assessment. Several systematic reviews have been undertaken looking at airway clearance in people with cystic fibrosis (Elkins et al 2006a, Main et al 2005, van der Schans et al 2000). Chest physiotherapy (postural drainage and percussion) was compared with no chest physiotherapy (van der Schans et al 2000). It was concluded that airway clearance techniques may have short-term effects in the context of increasing mucus transport, but no evidence was found on which to draw conclusions concerning the long-term effects. Elkins et al (2006a) studied PEP and Main et al (2005) compared ‘conventional’ chest physiotherapy (postural drainage and percussion) with other contemporary airway clearance techniques (active cycle of breathing techniques, autogenic drainage, PEP, oscillating PEP and high-frequency chest wall oscillation). There appeared to be no advantage of ‘conventional’ chest physiotherapy over the other airway clearance techniques, in the primary outcome measure of lung function. In the review by Elkins et al (2006a), there was an indication that individuals prefer regimens that can be self-administered.
Holland & Button (2006) reviewed the place of airway clearance techniques in chronic respiratory disease and proposed a physiological rationale for the use of airway clearance techniques in chronic obstructive pulmonary disease taking into consideration the presence of bronchiectasis, amount of sputum expectorated, degree of airflow obstruction and presence of decreased lung elastic recoil.
Active cycle of breathing techniques
The active cycle of breathing techniques (ACBT) has been shown to be effective in the clearance of bronchial secretions (Pryor et al 1979, Wilson et al 1995) and to improve lung function (Webber et al 1986) without increasing hypoxaemia (Pryor et al 1990) or airflow obstruction (Pryor & Webber 1979, Pryor et al 1994, Thompson & Thompson 1968).
The ACBT is a flexible regimen that can be adapted for any patient in whom there is a problem of excess bronchial secretions and can be used with or without an assistant. It is a cycle of breathing control, thoracic expansion exercises and the forced expiration technique (FET) (Fig. 5.2). The original studies of ‘the forced expiration technique’ (Hofmeyr et al 1986, Pryor et al 1979, Webber et al 1986) used this cycle of techniques, but people began to use a regimen of huffing alone or other variations on the FET (Falk et al 1984, Reisman et al 1988) and the literature became confusing. In order to emphasize the use of thoracic expansion exercises and the periods of breathing control, in addition to the FET, the whole regimen was renamed the active cycle of breathing techniques (ACBT) (Webber 1990). The regimen did not change in practice and the early studies on the FET were controlled clinical trials on the ACBT.
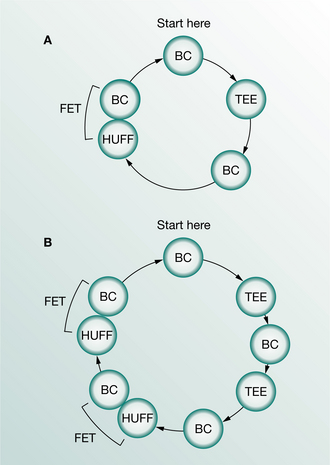
Figure 5.2 Diagrammatic representation of two examples of the active cycle of breathing techniques, demonstrating the flexibility of the technique: BC, breathing control; TEE, thoracic expansion exercises; FET, forced expiration technique.
Breathing control
Breathing control is the resting period between the more active parts of the cycle. It is tidal breathing, at the patient’s own rate and depth. The person is encouraged to relax the upper chest and shoulders and to use the lower chest, diaphragmatic, pattern of breathing as much as they are able. It allows the lungs and chest wall to revert to their resting position. This period should be continued until the person is ready to use either the thoracic expansion exercises or the huffing of the forced expiration technique.
Thoracic expansion exercises
Thoracic expansion exercises are deep breathing exercises emphasizing inspiration. Inspiration is active and is usually combined with a 3-second end- inspiratory hold before passive relaxed expiration. The postoperative manoeuvre of a 3-second hold at full inspiration has been shown to decrease collapse of lung tissue (Ward et al 1966). This ‘hold’ may also be of value in patients with medical chest conditions to allow for asynchronous ventilation as air flows more quickly into healthy, unobstructed areas than into diseased and obstructed regions. Pendelluft flow is that which takes place between parallel respiratory units with different time constants (Mead et al 1970).
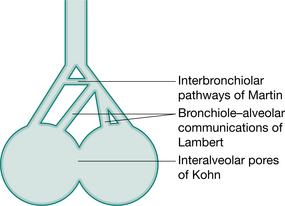
In the normal lung, the resistance to airflow via the collateral ventilatory system is high and there is little movement of gas through these channels. With increasing lung volume and in the presence of lung pathology, resistance decreases allowing air to flow via the collateral channels – the interalveolar pores of Kohn, bronchiole–alveolar communications of Lambert and interbronchiolar pathways of Martin (Menkes & Traystman 1977) (Fig. 5.3) and to come in behind secretions. These collateral channels are not present in infants and young children and it is unknown when, in the developmental process, they become patent.
The effectiveness of thoracic expansion exercises, in re-expanding lung tissue and in mobilizing and clearing excess bronchial secretions, can also be explained by the phenomenon of interdependence (Mead et al 1970). This is the effect of expanding forces exerted between adjacent alveoli. At high lung volumes the expanding forces between alveoli are greater than at tidal volume and may assist in re-expansion of lung tissue (Fig. 5.4). About three expansion exercises are usually appropriate before pausing for a few seconds for a period of breathing control. Too many deep breaths may produce the effects of hyperventilation, are tiring and reduce the number of huffs undertaken within the time period. The thoracic expansion exercises may be undertaken consecutively or patients may like to take a normal-sized breath in between.
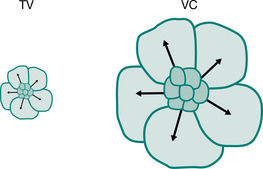
Figure 5.4 Interdependence. The effect of expanding alveoli at tidal volume (TV) and vital capacity (VC).
Thoracic expansion exercises can be facilitated with proprioceptive stimulation by placing a hand, either the patient’s or the physiotherapist’s, over the part of the chest wall where movement of the chest is to be encouraged (Fig. 5.5). There may be an initial increase in ven- tilation to this part of the lung (Tucker et al 1999) and there should be an increase in chest wall movement.
Sometimes an additional increase in lung volume can be achieved by using a ‘sniff’ manoeuvre at the end of a deep inspiration. This manoeuvre may not be appropriate in patients who are hyperinflated, but for surgical patients who need further motivation to increase their lung volume, it may be a useful technique.
Thoracic expansion exercises may be combined with chest shaking, vibrations and/or chest clapping, if there is an indication that the inclusion of these techniques may assist further the clearance of secretions.
Forced expiration technique (FET)
The forced expiration technique is a combination of one or two forced expirations (huffs) and periods of breathing control. Huffing to low lung volumes will move the more peripherally situated secretions and when secretions have reached the larger more proximal upper airways, a huff or cough from a high lung volume can be used to clear them.
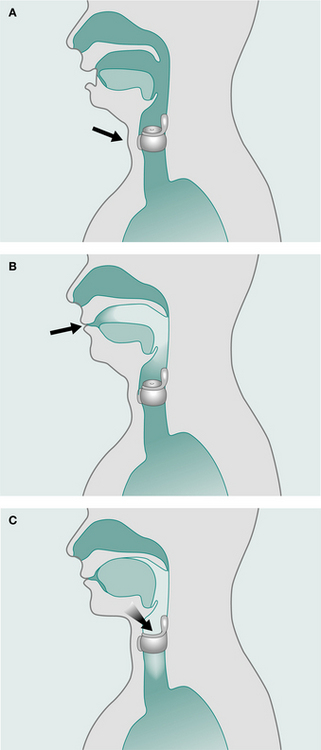
With any forced expiratory manoeuvre there is dynamic compression and collapse of the airways downstream (towards the mouth) of the equal pressure point (West 2004). This is an important part of the clearance mechanism of either a huff or cough. At lung volumes above functional residual capacity, the equal pressure points are located in lobar or segmental bronchi (Macklem 1974). As lung volume decreases, during a forced expiratory manoeuvre, the equal pressure points move distally into the smaller more peripheral airways. Figure 5.6 is a static representation of the dynamic state of a forced expiratory manoeuvre (huff or cough). The pleural pressure at this instance is +20 cmH2O, the elastic recoil pressure of the lungs is +5 cmH2O and consequently the peripheral pressure is +25 cmH2O. There is a downward gradient from +25 cmH2O at the alveolus to zero at the mouth and, somewhere along the airway, the pressure within the airway will equal the pleural pressure. Proximal to this, dynamic collapse and compression of the airway takes place. The dynamic collapse and compression during a forced expiratory manoeuvre is effective from points, choke points (Dawson & Elliott 1977), downstream of the equal pressure point.
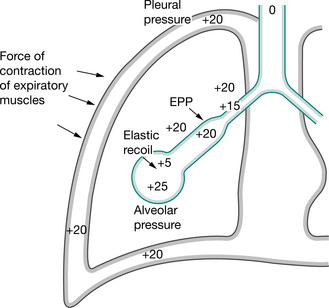
Figure 5.6 Forced expiratory manoeuvre: huff or cough.
(Pryor 1991. In Pryor JA (ed) Respiratory care, p 84. Reproduced with permission of Elsevier, Edinburgh)
A series of coughs without intervening inspirations was advocated by Mead et al (1967) to clear bronchial secretions, but clinically a single continuous huff down to the same lung volume is as effective and less exhausting. Hasani et al (1994) compared cough with the FET and concluded that both were equally effective in clearing lung secretions, but that the FET required less effort.
The mean transpulmonary pressure during voluntary coughing is greater than during a forced expiration. This results in greater compression and narrowing of the airways, which limits airflow and reduces the efficiency of bronchial clearance (Langlands 1967). In 1989 Freitag et al demonstrated an oscillatory movement, ‘hidden’ vibrations, of the airway walls in addition to the squeezing action produced by the forced expiratory manoeuvre.
The viscosity of mucus is shear-dependent (Lopez-Vidriero & Reid 1978) and the shear forces generated during a huff should reduce mucus viscosity (Selsby & Jones 1990). This, together with the high flow of a forced expiratory manoeuvre, would also be expected to aid mucus clearance and the expectoration of sputum.
When mobilizing and clearing peripheral secretions, it is an unnecessary expenditure of energy to start the huff from a high lung volume. A huff from mid-lung volume is more efficient and probably more effective. To huff from mid-lung volume a medium-sized breath should be taken in and, with the mouth and glottis open, the air is squeezed out using the chest wall and abdominal muscles. It should be long enough to loosen secretions from the more peripherally situated airways and should not be just a clearing noise in the back of the throat. However, if the huff is continued for too long it may lead to unnecessary paroxysmal coughing. Too short a huff may be ineffective (Partridge et al 1989) but when secretions have reached the upper airways, a shorter huff or a cough from a high lung volume is used to clear them.
The huff is a forced but not violent manoeuvre. To be maximally effective, the length of the huff and force of contraction of the expiratory muscles should be altered to maximize airflow and to minimize airway collapse. A peak flow mouthpiece, or similar piece of tubing, may improve the effectiveness of the huff as it should help to keep the glottis open. Some people find huffing through a tube, at a tissue or a cotton wool ball, helpful in perfecting the technique. The huff can be introduced to children as blowing games (Thompson 1978) and from about the age of 2 years they are usually able to copy others huffing (Fig. 5.7).
An essential part of the forced expiration technique is the pause for breathing control after one or two huffs, to prevent any increase in airflow obstruction. The length of the pause will vary from patient to patient. In a patient with bronchospasm or unstable airways or in one who is debilitated and fatigues easily, longer pauses (perhaps 10–20 seconds) may be appropriate. In patients with no bronchospasm the periods of breathing control may be considerably shorter (perhaps two or three breaths or 5–10 seconds).
In the tetraplegic patient, clearance of secretions from the upper airways is difficult because maximum lung volume cannot be achieved and the equal pressure points will therefore never reach the largest airways (Morgan et al 1986). Secretions can be cleared from the smaller airways, but accumulate in the larger upper airways. The use of glossopharyngeal breathing may assist clearance from the upper airways.
The techniques of breathing control, thoracic expansion exercises and the forced expiration technique should be used flexibly and adapted for, and by, each patient during each treatment session. One set of thoracic expansion exercises may be followed by the forced expiration technique (see Fig. 5.2A) but if secretions loosen slowly, it may be more appropriate to use two sets of thoracic expansion exercises (see Fig. 5.2B) interspersed with a period of breathing control. Most surgical patients will benefit from the 3-second hold with the thoracic expansion exercises and there is rarely an indication, in the surgical patient, for the use of chest clapping. Wound support may be more suitable than chest compression during huffing and coughing (Fig. 5.8).
In many patients, medical and surgical, secretions can be cleared effectively using the ACBT in the sitting position, but on occasions gravity-assisted positions may be indicated, for example a lung abscess. Cecins et al (1999) studied the effects of gravity (positions with and without a head-down tilt) in a group of patients with cystic fibrosis, bronchiectasis and immotile cilia syndrome, using the ACBT. There were no significant differences in lung function or in the weight of sputum expectorated during treatment. Most of the patients preferred the horizontal position and felt less breathless without a head-down tilt.
The ‘endpoint’ of a treatment session can be recognized, either by the physiotherapist or the patient self-treating, when an effective huff to low lung volume has become dry sounding and non-productive (for example two cycles). The sicker patient may not reach this endpoint before tiring and should stop before becoming exhausted with any airway clearance technique.
Autogenic drainage
Autogenic drainage (AD) aims to maximize airflow within the airways, to improve ventilation and the clearance of mucus (Chevaillier 2002). Chevaillier developed this concept in Belgium in the late 1960s but little was published until 1979 (Dab & Alexander 1979). Autogenic drainage utilizes gentle breathing at different lung volumes to loosen, mobilize and clear bronchial secretions.
In people with pressure-dependent airway collapse the increase in airflow, with an unforced compared with a forced expiratory manoeuvre, can be demonstrated using the flow–volume loop (Fig. 5.9). Many people with chronic respiratory disease will have a degree of pressure-dependent airway collapse, but in the absence of pressure-dependent collapse it is not possible to move outside the flow–volume loop, i.e. to increase airflow with an unforced expiratory manoeuvre.
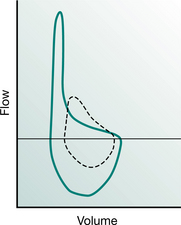
Figure 5.9 Flow–volume loop demonstrating pressure-dependent collapse (concave shape of expiratory loop). The continuous line represents a forced manoeuvre and the broken line, an unforced manoeuvre. There is an increase in expiratory airflow, from the small airways, with the forced expiratory manoeuvre.
Chevaillier originally described three phrases: ‘unstick’, ‘collect’ and ‘evacuate’ (Schöni 1989) (Fig. 5.10). The breathing technique is that of a slow breath in, keeping the upper airways (mouth and glottis) open. It is recommended that the breath in be held for 2–4 seconds. This ‘hold’ facilitates more equal filling of lung segments, by allowing for the variation in time constants within different lung regions (collateral ventilation). Expiration is performed keeping the upper airways open, as if sighing. The expiratory force is balanced so that the expiratory flow reaches the highest rate possible without causing airway compression. Air flow at a high velocity enhances shear forces. As mucus is mobilized it can be both heard and felt (by placing the hands on the chest) (Fig. 5.11). This cycle is repeated through varying lung volumes.
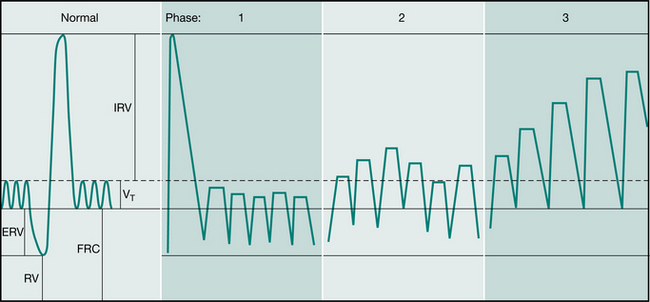
Figure 5.10 Autogenic drainage: Belgian method. Phases of autogenic drainage shown on a spirogram of a normal person. Phase 1: unstick; Phase 2: collect; Phase 3: evacuate. (VT = tidal volume, ERV = expiratory reserve volume, RV = reserve volume, FRC = functional residual capacity, IRV = inspiratory reserve volume, IRV + VT + ERV = vital capacity).
(Schöni 1989. Reproduced with permission of the Journal of the Royal Society of Medicine)
Breathing at low lung volumes is said to mobilize peripheral mucus. This is the first or ‘unstick’ phase. It is followed by a period of breathing around the individual’s tidal volume which is said to ‘collect’ mucus from the middle airways. Then, by breathing around high lung volumes, the ‘evacuate’ phase, expectoration of secretions from the central airways is promoted. When sufficient mucus has been collected in the large airways it may be cleared by coughing or huffing. Coughing before this point is discouraged (Chevaillier 2001 personal communication, Chevaillier 2002). Autogenic drainage is usually undertaken in the sitting or supine lying positions.
A short-term study compared AD with the ACBT (Miller et al 1995), but this was not the ACBT as outlined above. In a long-term study of patients with cystic fibrosis, AD was compared with postural drainage and per- cussion, with an assistant (Davidson et al 1992). AD was found to be at least as effective as the conventional treatment and the patients expressed a marked preference for AD.
For infants and young children (under the age of about 5 years), ‘passive’ AD is used in some countries. The goals of AD are achieved by the therapist using their hands to guide the thorax and respiratory movements. The treatment is passive, gentle and effective.
In Germany the practice of AD was modified (David 1991, Kieselmann 1995) and not split into the three phases, as the patients found breathing at low lung volumes uncomfortable. This technique is known as modified autogenic drainage (M AD) (Fig. 5.12). The patient breathes around tidal volume while breath holding for 2–3 seconds at the end of each inspiration. Coughing is used to clear mucus from the larynx (Kieselmann 1995).
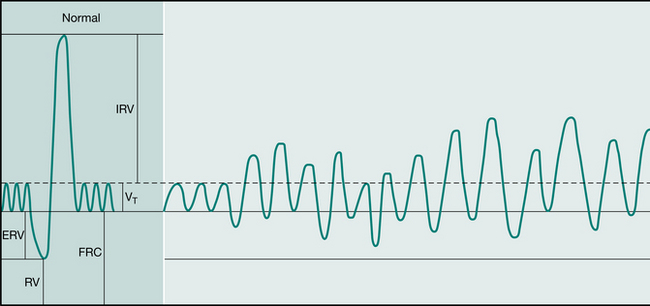
Figure 5.12 Autogenic drainage: German method. Autogenic drainage shown on a spirogram of a normal person. The method is not divided into separate phases. (VT = tidal volume, ERV = expiratory reserve volume, RV = reserve volume, FRC = functional residual capacity, IRV = inspiratory reserve volume, IRV + VT + ERV = vital capacity).
(David 1991. In: Pryor JA (ed) Respiratory care, p 69. Reproduced with permission of Elsevier, Edinburgh)
Chest clapping
Chest clapping is performed using a cupped hand with a rhythmical flexion and extension action of the wrist. The technique is often performed with two hands but, depending on the area of the chest, it may be more appropriate to use one hand. For the infant, chest clapping is performed using two or three fingers of one hand or using a soft rubber facemask (Chapter 10). Single-handed chest clapping is probably the technique of choice for self-chest clapping as it is difficult to coordinate two-handed clapping at the same time as using thoracic expansion exercises.
Chest clapping should never be uncomfortable and should be performed over a layer of clothing to avoid sensory stimulation of the skin. It should not be necessary to use extra layers of clothing or towelling, as the force of the chest clapping should be adapted to suit the individual.
Mechanical percussion has been shown to increase intrathoracic pressure (Flower et al 1979) and chest clapping may have a similar effect, but this change in intrathoracic pressure has not been correlated with an increase in the clearance of bronchial secretions. Andersen (1987, personal communication) hypothesized that the air-filled alveoli would buffer increases in intrathoracic pressure and markedly reduce the mechanical effect of chest clapping.
Some studies (Campbell et al 1975, Wollmer et al 1985) have demonstrated an increase in airflow obstruction when chest clapping is included in an airway clearance regimen, but other studies (Gallon 1991, Pryor & Webber 1979) have shown no increase in airflow obstruction with chest clapping.
In infants and small children who are not yet old enough to cooperate with voluntary breathing techniques, in patients with neuromuscular weakness or paralysis and in the intellectually impaired, chest clapping may be a useful technique to stimulate coughing. The cough is probably stimulated by the mobilization of secretions; however, other methods such as physical activity or other airway clearance modalities may be more appropriate.
Chest clapping has been shown to cause an increase in hypoxaemia (Falk et al 1984, McDonnell et al 1986) when performed for short periods only (less than 30 seconds) but when combined with three to four thoracic expansion exercises there was no decrease in oxygen saturation (Pryor et al 1990).
In a group of clinically stable patients with cystic fibrosis, no advantage was shown when self-chest clapping was used in addition to thoracic expansion exercises (Webber et al 1985), but this cannot be extrapolated to either all medical chest conditions or to acute chest problems.
If a patient feels that self-chest clapping is beneficial but the physiotherapist thinks it is tiring and may be causing hypoxaemia, the patient should be monitored using an oximeter. If oxygen desaturation of clinical significance occurs during the self-chest clapping, the patient should be encouraged to omit the clapping but to continue with the thoracic expansion exercises. Patients studied by Carr et al (1995) felt that self-chest clapping was useful when they were clinically stable, but more particularly when they were unwell. The benefits of chest clapping remain uncertain, but if chest clapping is considered to be clinically beneficial for an individual it should be continued, provided there are no adverse effects.
There is probably no indication for chest clapping in postoperative patients and in patients following chest trauma. Severe osteoporosis and frank haemoptysis are contraindications, although chest clapping is unlikely to increase bleeding when bronchial secretions are lightly streaked with blood.
Vigorous and rapid chest clapping may lead to breath holding and may induce bronchospasm in a patient with hyper-reactive airways. There is no evidence that alteration in the rate of chest clapping increases or decreases the mobilization of bronchial secretions. A rhythmical rate that is comfortable for both patient and physiotherapist is probably the most appropriate.
Chest shaking, vibrations and compression
The therapist’s hands are placed on the chest wall and, during expiration, a vibratory action in the direction of the normal movement of the ribs is transmitted through the chest using body weight. This action augments expiratory flow (McCarren & Alison 2006) and may help to mobilize secretions. It is unknown whether airway closure will be increased if the vibratory action is continued into the expiratory reserve volume, but these techniques are frequently combined with thoracic expansion exercises, which would be likely to counteract any resulting airway closure.
The vibratory action may be either a coarse movement (chest shaking) or a fine movement (chest vibrations). Physiotherapists have tended to adopt the techniques that they find the most helpful clinically. Vibrations are commonly used in infants and children, where the chest wall is more compliant than in adults (Chapter 10). Chest vibrations and shaking should never be uncomfortable and should be adapted to suit the individual patient. Some patients, doing their own chest physiotherapy, find self-chest vibrations helpful. One hand is placed on top of the other on the appropriate part of the chest wall and vibrations or shaking are carried out during expiration. With the hands in a similar position, chest compression throughout expiration may be helpful to augment the forced expiratory manoeuvre of the huff. When in side lying, selfcompression can be given over the side of the chest with the upper arm and elbow and the hand of the other arm.
The physiotherapist or the patient’s carer may give compression during huffing or coughing. Some patients find this helpful, but others prefer to be unsupported. Postoperative patients usually find that supporting the wound facilitates both huffing and coughing (Fig. 5.8). With fractured ribs and other chest injuries, shaking of the chest wall would be inappropriate but compressive support may assist the clearance of secretions.
In the paralysed patient the technique of rib springing may be used, where compression of the chest wall is continued throughout expiration and overpressure is applied at the end of the breath out. By releasing the hands quickly, inspiration is encouraged. This technique is inappropriate in the non-paralysed patient and may be harmful as compression against a reflexly splinted chest wall may produce rib fractures. Assisted coughing for the paralysed patient is described in Chapter 16.
In the drowsy, semicomatose patient (for example, the chronic bronchitic in respiratory failure with sputum retention), chest compression similar to, but less vigorous than, rib springing may stimulate a deeper inspiration. Chest shaking or chest vibrations are often used during the expiratory phase of a manual hyperinflation treatment to assist the clearance of secretions (Chapters 8 and 10). Care must be taken when using the techniques of chest shaking, vibrations and compression if there are signs of osteoporosis or metastatic deposits affecting the ribs or vertebral column.
A study by McCarren & Alison (2006) of people with cystic fibrosis compared the expiratory flow rates and frequencies of airflow oscillation of vibration and percussion (chest clapping) with positive expiratory pressure and the oscillating positive expiratory pressure of the Flutter® and Acapella®. They concluded that vibration produced greater expiratory flow rates and a higher peak expiratory/peak inspiratory flow ratio, although the Flutter® and Acapella® had higher oscillation frequencies. Their clinical recommendation was that if patients were unable to cough or huff effectively, vibration could be used.
High-frequency chest wall oscillation
High-frequency chest wall oscillation (HFCWO) (also known as high-frequency chest wall compression (HFCWC) or high-frequency chest compression (HFCC)) commonly provides compression of the chest wall at frequencies of 5–20 Hz. Usually this compressive force is created using an inflatable jacket adjusted to fit snugly over the thorax. This is linked to an air-pulse generator, which delivers intermittent positive pressure airflow into the jacket. As a result the jacket rapidly expands, compressing the chest wall, producing a transient (hence oscillatory) increase in airflow in the airways (Fig. 5.13).
It is hypothesized that mucus clearance is enhanced as a consequence of this airflow oscillation and vibration of the airway walls. The proposed mechanism is that increased mucus / airflow interaction leads to increased cough-like shear forces and decreased mucus viscoelasticity (King et al 1983, Tomkiewicz et al 1994). Furthermore, HFCWO creates an expiratory bias to airflow and it is hypothesized that this promotes movement of mucus downstream toward the mouth. It has also been suggested that HFCWO may enhance ciliary activity (Chang et al 1988, Freitag et al 1989, Hansen et al 1994, King et al 1984, 1990).
There is short-term evidence to support increased mucociliary clearance with HFCWO in patients with cystic fibrosis. Some studies have demonstrated a significant increase in sputum clearance after HFCWO in comparison with baseline or control, but no significant difference in efficacy between HFCWO and other airway clearance techniques including postural drainage and percussion (PD&P), positive expiratory pressure (PEP) and high-frequency oral oscillation (HFOO) (Braggion et al 1995, Scherer et al 1998). Other studies, comparing HFCWO with PD&P, have demonstrated significantly more sputum clearance with HFCWO over PD&P (Hansen & Warwick 1990, Kluft et al 1996). Benefits in airway clearance with HFCWO have also been reported in other conditions including asthma, chronic obstructive pulmonary disease, neuromuscular disease, postoperative patients and those requiring long-term ventilatory support (Allan et al 2003, Chiappetta & Beckerman 1995, Perry et al 1998, Wen et al 1996, Whitman et al 1993).
Different studies have used different protocols in the application of HFCWO (Arens et al 1994, Braggion et al 1995, Darbee et al 2005, Kluft et al 1996, Oermann et al 2001, Stites et al 2006, Varekojis et al 2003, Warwick & Hansen 1991, Warwick et al 2004). Most commonly, a total treatment time of 30 minutes covering six frequencies between 6–25Hz has been used and standard manufacturers’ guidelines suggest 10–30 minutes of total treatment time (Electromed 2006, Hill-Rom 2006). Early animal studies found 13 Hz to be an optimum treatment frequency (Gross et al 1985, King et al 1983) and some manufacturers suggest 10 minutes each at 10, 12 and 14 Hz (Electromed 2006). However, more recently it has been recommended that an individual ‘tuning’ method should be used to identify optimum treatment frequencies, which vary among individuals and according to the waveform of the machine used (Milla et al 2006).
HFCWO is widely used in the USA where it is an attractive alternative to PD&P with an assistant, with cost benefit. However, in the UK and Europe where other independently performed techniques are the mainstay of care for airway clearance, HFCWO is not used as extensively and has cost implications.
Infants and young children
For infants and young children regular physical activity is important, not just for airway clearance but for the many other benefits it provides. Physical activity is possible even in the infant, for example, playing on a gym ball (Fig. 5.14). In babies who have a chronic respiratory disorder (with increased airway secretions) an additional airway clearance technique may be necessary (e.g. modified postural drainage and percussion, positive expiratory pressure (PEP) or assisted autogenic drainage (AD)). For young children, trampolining (Fig. 5.15) and, when they are able to cooperate, bubble PEP (Fig. 5.24) are fun. At an older age, other airway clearance techniques can be added.
Insufflation/exsufflation-assisted cough
A mechanical insufflator/exsufflator uses positive pressure to deliver, to the upper airway, a maximal lung inhalation followed by an abrupt switch to negative pressure. The rapid change from positive to negative pressure is aimed at simulating the airflow changes that occur during a cough, thereby assisting sputum clearance. In 1953 various portable devices were manufactured to deliver mechanical insufflation/exsufflation (e.g. OEM Cof-flater portable cough machine, St Louis). The most commonly used mechanical insufflator/exsufflator today is the CoughAssistTM (Emerson, Massachusetts) (Fig. 5.16). Initial investigations showed mechanical insufflation/exsufflation to be effective at removing foreign bodies from anaesthetized dogs (Bickerman 1954).
Beck & Barach (1954) demonstrated clinical and radiographic improvements in 92 of 103 acutely ill patients with respiratory tract infections, with the use of mechanical insufflation/exsufflation. Seventy-two patients had bronchopulmonary lung disease and 27 had chest wall or neuromuscular disease. Improvement was more evident in patients with neuromuscular disease. The cardiovascular effects of mechanical insufflation/exsufflation were evaluated by Beck & Scarrone (1956) who found that patients demonstrated an increase in mean heart rate of 17 beats per minute and an increase in systolic blood pressure of 8 mmHg. There was also an increase in cardiac output of 2.1 litres per minute and echocardiograph changes reflective of rotation of the heart during normal coughing.
Mechanical insufflation/exsufflation has been shown to increase peak cough flow (PCF) in patients with neuromuscular disease (Bach 2003, Chatwin et al 2003, Mustfa et al 2003, Sancho et al 2004, Sivasothy et al 2001). An increase in PCF is thought to improve the efficacy of the cough and thus assist in secretion removal. Mustfa et al (2003) and Sancho et al (2004) found a significant improvement from baseline PCF with mechanical insufflation/exsufflation for both bulbar and non-bulbar motor neuron disease patients, although non-bulbar patients had the greatest change in PCF. Mechanical insufflation/exsufflation has not been shown to increase PCF in patients with chronic obstructive pulmonary disease (Sivasothy et al 2001, Winck et al 2004).
Patients who are novices to the device may not tolerate high pressure changes initially and very high pressures cause leakage around the mask. Several authors (Chatwin et al 2003, Miske et al 2004, Sivasothy et al 2001, Vianello et al 2005) have reported a good outcome with low pressures. One study (Miske et al 2004) (age range 3 months to 28.6 years) used median pressures of +30 to −30 cmH2O with a range from +15 to +40 cmH2O (insufflation) and −20 to −50 cmH2O (exsufflation). Other researchers however, advocate higher pressure spans of +40 to −40/−60 (Bach 1993, 1994; Bach et al 1993, 2000, 2003, Tzeng & Bach 2000). The CoughAssistTM (Emerson, Massachusetts) is often used in automatic mode (Bach 1993, 1994, Bach et al 1993, 2000, 2003, Tzeng & Bach 2000) enabling the device to be used in the domiciliary environment without a trained professional. In this mode the device swings between a set negative and positive pressure, and will hold in insufflation for a set period before switching to exsufflation for a set time and then providing a pause. The movement in manual mode should be one sweeping movement with no pauses between insufflation and exsufflation.
Patients learn to coordinate their cough when the device switches to exsufflation and initial instructions should indicate that a deep breath will be coming and they should cough when they feel the negative pressure. Mechanical insufflation/exsufflation can be combined with a manually assisted cough (Chapter 16). Initially the patient acclimatizes to the device in manual mode and usually with a full-face mask. The insufflation (positive) pressure should be set between 15 and 20 cmH2O and increased to give an inspiration to total lung capacity. Initially the exsufflation (negative) pressure should be the same as the insufflation pressure, but when appropriate the negative pressure should be increased to 10–20 cmH2O above the positive pressure. The best indicator of efficacy is an increase in the audible sound of the cough.
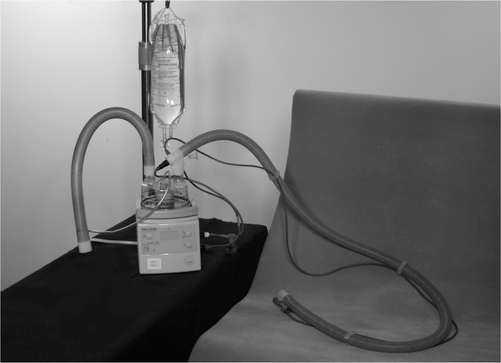
Intrapulmonary percussive ventilation
Intrapulmonary percussive ventilation (IPV) has been used as a method of airway clearance, both within hospitals and in the home. The IPV device was developed in 1979 by Forrest M Bird (Percussionaire®, Idaho). It consists of a high-pressure flow generator, a valve for flow interruption and a breathing circuit with a nebulizer that can be attached to a face mask, mouthpiece or catheter mount (Fig. 5.17). IPV is a modified method of intermittent positive pressure breathing (IPPB) which superimposes high-frequency mini bursts of air (50 to 550 cycles per minute) on the individual’s intrinsic breathing pattern to create an internal vibration (percussion) within the airways. It is hypothesized that internal or external vibration of the chest may promote clearance of mucus from the peripheral bronchial tree (Fink & Mahlmeister 2002). IPV may provide ventilatory support in patients with neuromuscular disease (Chatwin et al 2004) and in patients with chronic obstructive pulmonary disease (Nava et al 2006). IPV devices include: IMPULSATOR®-F00012, IPV1C®-F00001-C, IPV2C®-F00002-C, Percussionaire® (Idaho) and IMP II (Percussionaire, Breas, Sweden).
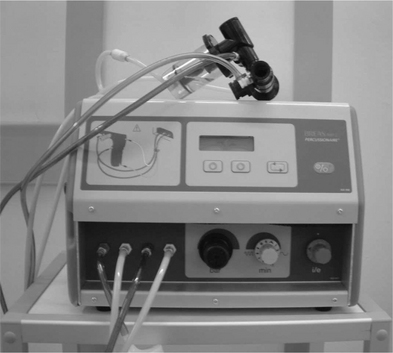
Figure 5.17 The intrapulmonary percussive ventilation device.
(IMP II Percussionaire, Breas, Sweden)
In tracheostomized patients with Duchenne muscular dystrophy (DMD), IPV has been compared with physiotherapy consisting of the forced expiration technique and manually assisted coughing (Toussaint et al 2003) using sputum weight as the primary outcome measure. The subjects were divided into a hypersecretive group and a normosecretive group. It was concluded that IPV enhanced peripheral bronchial secretion clearance in the hypersecretive DMD patients compared with conventional physiotherapy. Clini et al (2006) investigated the effects of IPV ICU patients in the intensive care unit with a tracheostomy, who were weaning from mechanical ventilation. The patient group (n = 46) received 15 days of two, 1-hour physiotherapy sessions per day with one group (n = 24) receiving IPV for 10 minutes before physiotherapy sessions. The subjects were assessed every 5 days, over a 15-day period, using the outcome measurements of arterial blood gases, PaO2/FiO2 ratio and maximal expiratory pressure. At 15 days the IPV group had a significantly improved PaO2/FiO2 ratio and a higher maximal expiratory pressure. At follow-up the IPV group also had a lower incidence of pneumonia, indicating that IPV may have a place in decreasing atelectasis in long-term weaning patients.
Vargas et al (2005) investigated patients with chronic obstructive pulmonary disease during an acute respiratory exacerbation and mild respiratory acidosis. In this study, one group received standard treatment (including oxygen, bronchodilators, steroids and antibiotics) and the other standard treatment with the addition of IPV (30 minutes twice a day). The patients who received IPV were discharged home significantly earlier 6.8 ± 1 days versus 7.9 ± 1.3 days (p <0.05). IPV also helped to prevent further deterioration, that of pH <7.35 requiring non-invasive ventilation (IPV and standard treatment 0/16 patients, standard treatment alone 6/17 patients (p < 0.05)). The authors hypothesized that the IPV group may have benefited from improved secretion removal; however, they do state that this cannot be confirmed as sputum weight was not measured. Previous studies investigated sputum mobilization, in people with cystic fibrosis, by comparing the use of IPV to other airway clearance techniques e.g. postural drainage and percussion, high-frequency chest wall oscillation and the Flutter (Newhouse et al 1998, Scherer et al 1998, Varekojis et al 2003). These studies demonstrated IPV to be equal in efficacy to the other methods of airway clearance in sputum mobilization, when the amount of sputum expectorated was assessed by dry weight. IPV can also be used in conjunction with other airway clearance techniques, for example autogenic drainage and the active cycle of breathing techniques, to assist in clearance of secretions.
IPV has a place in the clearance of secretions in both restrictive and obstructive patients. Following preliminary safety studies investigating the physiogical affects of IPV (Chatwin et al 2004, Nava et al 2006) and sputum clearance (Toussaint et al 2003), guidelines for starting settings have been suggested.
When the patient has acclimatized to IPV, the pressure and frequency should be adjusted to optimize secretion clearance. Inverting the I : E ratio, to 2 : 1, will assist ventilatory support (Toussaint et al 2003). When treating patients with neuromuscular weakness, manually assisted coughs may be required to clear secretions from the large airways (Chapter 16).
Oscillating positive expiratory pressure
These devices combine an oscillation of the air within the airways during expiration and a variable positive expiratory pressure. Three commonly used ones are described below.
The Flutter® (Flutter) is a small, portable device (Fig. 5.18). It is pipe-shaped with a single opening at the mouthpiece and a series of small outlet holes at the top of the bowl. The bowl contains a high-density stainless steel ball enclosed in a small cone. During expiration the movement of the ball along the surface of the cone creates a positive expiratory pressure (PEP) and an oscillatory vibration of the air within the airways. In addition, intermittent airflow accelerations are produced by the same movements of the ball. The device is held horizontally and tilted slightly either downwards or upwards until a maximal oscillatory effect can be felt. It is usually used either sitting or in supine lying, but may be used in other positions provided an effective oscillation can be maintained. Sputum viscoelasticity has been shown to be reduced following the use of the Flutter (App et al 1998).
The Flutter is placed in the mouth and inspiration is either through the nose or through the mouth by breathing around the Flutter mouthpiece (it is not possible to breathe in through the Flutter device). A slow breath in, only slightly deeper than normal, with a breath hold of 3–5 seconds is followed by a breath out, through the Flutter, at a slightly faster rate than normal. This is known as the ‘mucus loosening and mobilization’ stage. After four to eight of these breaths, a deep breath with a ‘hold’ at full inspiration is followed by a forced expiration through the Flutter. This may be repeated a second time and is the ‘mucus elimination’ stage. It may precipitate expectoration and should be followed by a pause for breathing control following a huff or cough. In some parts of Europe this forced expiratory manoeuvre, down to a low lung volume, is not used as a part of the regimen.
Konstan et al (1994) compared three regimens: the Flutter, voluntary coughing and a regimen of postural drainage and percussion, which included up to ten positions in the treatment session. Each session lasted 15 minutes. The Flutter regimen was the most effective as measured by the weight of sputum expectorated. Ambrosino et al (1995) and Homnick et al (1998) compared the Flutter with ‘standard’ chest physiotherapy and showed the Flutter to be as effective.
Originally the recommended technique for the Flutter was a gentle exhalation through the device. Treatment was continued for a period of 10 minutes and secretions were expectorated by spontaneous coughing. It was this regimen that was shown by Pryor et al (1994) to be less effective than the ACBT. The inclusion of a forced expiratory manoeuvre or huffing, in the regimen, would be likely to increase the effectiveness of airway clearance and this was demonstrated, in the short term, by Pike et al (1999).
When autogenic drainage was compared with the Flutter it was concluded that both regimens were equally effective, but the Flutter was easier to teach (Lindemann 1992). A clinical trial was undertaken using the Flutter in patients following thoracotomy, but no advantage was found in its inclusion (Chatham et al 1993). Two studies including the Flutter were published in 2001, both in people with cystic fibrosis. Oermann et al (2001) compared high-frequency chest wall oscillation and the Flutter, and McIlwaine et al (2001) compared the Flutter with PEP. In the 4-week study by Oermann et al, there were no significant differences between the regimens, but the authors stated that patient preference varied and patient satisfaction and preference should be considered. In the longer-term (1 year) study by McIlwaine et al there were significant rates of decline in lung function (forced vital capacity) in the Flutter group during this period. The authors hypothesized that the differences between the two regimens may be a consequence of the continuation of expiration into the expiratory reserve volume with the Flutter. This was likely to enter closing volume, with consequent airway closure. With the positive expiratory pressure regimen, end expiration remains above resting lung volume. Pryor et al (2006) did not observe deterioration in lung function with the Flutter but this study was in adults, whereas the McIlwaine study used a much younger population.
The Shaker® (NCS, Sao Paulo, Brazil) (Fig. 5.19) is similar to the Flutter. It also contains a high-density stainless steel ball enclosed in a small cone, but has a detachable mouthpiece making it easier to use in positions other than sitting.
The R-C Cornet® (Cornet) (Fig. 5.20) consists of a curved hard plastic outer tube, mouthpiece and flexible latex-free inner tube (valve-hose). The Cornet is placed in the mouth and inspiration is either through the nose or through the mouth, by breathing around the Cornet mouthpiece (it is not possible to breathe in through the Cornet). During expiration through the Cornet, a positive expiratory pressure and an oscillatory vibration of the air within the airways are generated. The flow, pressure and frequency of the oscillations can be adjusted to suit the individual and it can be used in any position (e.g. sitting, side lying, head-down tilt) as it is independent of gravitational forces. It is used in a similar way to the Flutter with initial breaths (five to eight) being only slightly deeper than normal and these breaths may be interspersed with one or two deeper and more forceful ones. A breath hold of 2–3 seconds at the end of inspiration is usually included. Huffing or coughing is used to clear secretions mobilized to the upper airways and is followed by breathing control. It is recommended that the Cornet be used for 10–15 minutes (Cegla 1999, personal communication). The Cornet has been shown to be as effective as the Flutter in airway clearance (Cegla et al 1997), to be beneficial in the management of people with chronic obstructive pulmonary disease (Cegla et al 2002) and to be equivalent, in the long term, to the ACBT, AD, Flutter and PEP in people with cystic fibrosis (Pryor et al 2006). It has also been shown to decrease the cohesiveness and viscoelasticity of sputum from patients with bronchiectasis (Feng et al 1998, Nakamura et al 1998).
The Acapella® (Acapella) (Fig. 5.21) is another form of oscillating PEP which uses a counterweighted plug and magnet to create airflow oscillations. Again, following six to eight breaths (with inspiration through the nose or around the mouthpiece of the device and expiration through the device) one or two deeper breaths, with a more active expiration, are followed by huffing and coughing. A short end-inspiratory hold is usually included. As with the Cornet, it is gravity-independent. Patterson et al (2005) compared the Acapella with the ACBT and concluded that both were effective in airway clearance. Volsko et al (2003) undertook a physical comparison between the Acapella and the Flutter and con-cluded that the devices have similar performance (pressure flow) characteristics.
Positive expiratory pressure
The delivery of positive expiratory pressure (PEP) via a face mask was described by Falk et al (1984) who found an increase in sputum yield and an improvement in transcutaneous oxygen tension when compared with postural drainage, percussion and breathing exercises. It was suggested that the increase in sputum yield was produced by the effect of PEP on peripheral airways and collateral channels. Falk & Andersen (1991) suggest that with the PEP treatment, the increase in lung volume may allow air to get behind secretions blocking small airways and assist in mobilizing them.
The original PEP apparatus, on which most of the clinical trials have been undertaken, consists of a facemask and a one-way valve to which expiratory resistances can be attached. A small-sized mask is available for infants. A flanged mouthpiece and nose clip can be used in place of the mask if the patient will not tolerate a mask but the required pressure, in mid-expiration, must be achieved for this equipment to be equivalent to that of a mask. A manometer is inserted into the system between the valve and resistance to monitor the pressure, which should be between 10 and 20 cmH2O during mid-expiration (Falk & Andersen 1991).
The patient sits leaning forward with the elbows supported on a table and holding the mask firmly over the nose and mouth (Fig. 5.22) while breathing at tidal volume, with a slightly active expiration, for 6–10 breaths. The lung volume should be kept up by avoiding complete expiration. This is followed by the forced expiration technique to clear secretions that have been mobilized. The duration and frequency of treatment are adapted to each individual, but treatment is usually performed for approximately 15 minutes, twice a day, in patients with stable chest disease and excess bronchial secretions (Falk & Andersen 1991). This should be adjusted according to the patient’s signs and symp-toms. In postoperative patients, short periods of PEP used every hour as a prophylactic treatment have been described by Ricksten et al (1986).
The study by Falk et al (1984) in patients with cystic fibrosis compared an assisted ‘conventional’ postural drainage treatment with an unassisted PEP mask regimen and found that the PEP mask regimen was more effective and the one preferred by the patients. Hofmeyr et al (1986) compared the unassisted treatment of PEP combined with the forced expiration technique, with thoracic expansion exercises combined with the forced expiration technique (the active cycle of breathing techniques). In this study the ACBT was found to be advantageous in terms of the amount of sputum expectorated. van der Schans et al (1991) studied mucus clearance with PEP using a radio-aerosol technique in patients with cystic fibrosis. They showed that PEP temporarily increased lung volume, but did not lead to an improvement in mucus transport. Mortensen et al (1991) demonstrated improved central and peripheral radioaerosol clearance with both PEP and the ACBT compared with controls. More recently Darbee et al (2004) studied the ventilation distribution and gas mixing with positive expiratory pressure breathing in adults with cystic fibrosis and demonstrated improved gas mixing associated with improvements in lung function, sputum expectoration and oxygen saturation.
Falk and Andersen (1991) have demonstrated collateral flow in experiments in postmortem human lungs (Fig. 5.23). In this experiment, a 3 mm airway was completely blocked and the pressures across the obstruction monitored during normal breathing and during breathing with 10 cmH2O PEP. In normal lungs and lungs affected by disease, acute respiratory failure and chronic airflow limitation, the obstruction pressure swings around zero during normal breathing. When PEP is applied at the airway opening, the pressure behind the obstruction becomes negative initially. This would tend to move the obstruction towards the periphery, but this is followed rapidly by a positive pressure tending to move the obstruction centrally. The duration of the positive obstruction period was far greater than the negative period and this culminated in movement towards the more central airways.
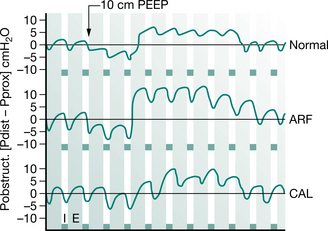
Figure 5.23 Pressure across a peripheral experimental airway obstruction (3 mm in diameter) during application of 10 cmH2O PEEP in different human pathology. Studies were performed in excised postmortem human lungs (Pobstruct: Obstruction pressure, positive if distal pressure exceeds proximal pressure; Pdistal: Pressure distal to obstruction; Pprox: Pressure proximal to obstruction; Normal: Normal lungs; ARF: Lungs affected by severe acute respiratory failure; CAL: Lungs affected by severe chronic airflow limitation; I; Inspiratory time; E: Expiratory time; PEEP: positive expiratory pressure).
(Falk and Andersen 1991. In: Pryor JA (ed) Respiratory care, p 55. Reproduced with permission of Elsevier, Edinburgh)
Lung function has been shown to improve with PEP in a long-term study (1 year) when compared with postural drainage and percussion (McIlwaine et al 1997). McIlwaine et al (2001) also compared the use of PEP and the Flutter in the long term (1 year). They concluded that PEP was more effective than Flutter in maintaining lung function, but Pryor et al (2006) did not identify any significant deterioration in lung function with the Flutter, compared with other airway clearance techniques, and this was also over a period of 1 year. Elkins et al (2006a) in a Cochrane review concluded that there was no clear evidence that PEP was a more or less effective intervention than other forms of physiotherapy.
Inspiratory resistance–positive expiratory pressure (IR-PEP) is obtained by placing a resistor into the inspiratory port of the PEP mask which provides resistance to both inspiration and expiration. The patient breathes in and out against a resistance and this has the effect of slowing down the flow rate and making the inspiratory breath more even. IR-PEP has been studied in patients undergoing abdominal surgery (Olsen et al 1999) and coronary artery bypass graft surgery (Westerdahl et al 2001) and its effects are said to be similar to PEP and bubble PEP.
Bubble PEP is an alternative way to administer PEP to young children, using a system that incorporates a column of water (to the level of PEP required: 10–12 cmH2O) and asking the child to blow through the column of water via a flexible straw or tubing (Fig. 5.24). By adding a little liquid detergent to the water, exhalations produce a stream of bubbles and the addition of a drop or two of food colouring to the water adds to the novelty of the treatment.
The equipment for bubble PEP is outlined in Fig. 5.25. When using bubble PEP, it is imperative that all the equipment is kept thoroughly clean. The water should be discarded immediately following treatment and the equipment cleaned. Fresh water should be used for each treatment session and the carton and tubing should be changed regularly. It is also important that the child is instructed only to exhale through the tubing and never to inhale (suck).
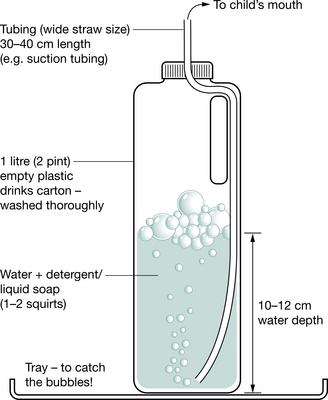
Figure 5.25 Equipment required for bubble PEP.
(Reproduced with permission from Great Ormond Street Hospital for Children, London, UK)
High-pressure PEP is a modified form of PEP mask treatment described by Oberwaldner et al (1986) for the treatment of patients with cystic fibrosis. The mask is used during tidal volume breaths and also for a full forced expiratory manoeuvre. By applying a positive expiratory pressure during forced expiration, secretions may be mobilized more easily in patients with unstable airways. Assessment for the technique involves forced vital capacity manouevres with the mask attached to a spirometer (Oberwaldner et al 1986). The resultant expiratory flow volume curves are used to determine the appropriate resistor for the PEP mask. The appropriate resistance is one that obtains a good flow plateau, achieves an expiration in excess of the patient’s normal forced vital capacity and eliminates any curvilinearity or scooping (indicating airway obstruction) (Fig. 5.26). The technique should be used only if full lung function equipment is available for regular reassessment of the appropriate expiratory resistance for each individual. Meticulous care must be taken, as an incorrect resistance may lead to deterioration in lung function and ineffective airway clearance.
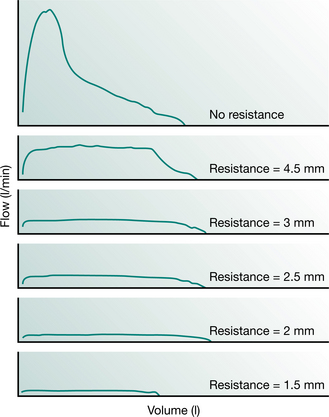
Figure 5.26 High-pressure PEP. Maximum expiratory flow–volume curves obtained without resistance (top) and with each of the PEP mask expiratory resistors. In this example, the resistor of choice for treatment is the 2 mm aperture as it demonstrates a maximal forced vital capacity, good plateau formation and no curvilinearity at the end of flow.
For treatment, the patient sits upright holding the mask firmly against the face. Six to ten rhythmical breaths, at tidal volume, are followed by an inspiration to total lung capacity and then a forced expiratory manoeuvre against the resistance to low lung volume. The pressure generated during this manouevre ranges from 50 to 120 cmH2O and usually results in the expectoration of sputum (Oberwaldner et al 1991).
Pfleger et al (1992) compared high-pressure PEP and autogenic drainage. More sputum was cleared with high- pressure PEP than with AD or with a combination of PEP and AD. Using this technique there is no evidence of any increase in incidence in pneumothorax (Zach & Oberwaldner 1992).
Physical activity
Physical activity will augment airway clearance (Sahl et al 1989), but may often not be as effective as other airway clearance techniques, particularly in people who produce moderate to large amounts of sputum. In people with cystic fibrosis, the combination of an airway clearance technique and exercise programme led to a reduction in the decline in lung function when compared with airway clearance and usual physicial activities (Schneiderman-Walker et al 2000). In addition to airway clearance there are many other benefits of physical activity (Chapters 13, 14 and 18).
Resistive inspiratory manoeuvres
The technique of resistive inspiratory manoeuvres is that of repeated maximal inspiratory vital capacity manoeuvres against a fixed resistance. Chatham et al (2004) reported an increase in sputum expectorated, using resistive inspiratory manoeuvres, when compared with the ACBT in a group of people with cystic fibrosis. In contrast, Patterson et al (2004) found that more sputum was expectorated with the ACBT than with resistive inspiratory manoeuvres in people with bronchiectasis.
With this technique the subject is required to carry out a full inspiratory vital capacity manoeuvre at 80% of maximal pressure by breathing against a fixed resistance. The maximum pressure is that developed between residual volume and total lung capacity. More research is required in this field, but it is hypothesized that the resistance results in an increased inspiratory time and that resistive inspiratory manoeuvres may increase inspiratory airflow to more peripheral airways with consequent shearing forces acting on airway secretions during the inspiratory phase. This may in turn reduce the need for higher expiratory flows (Chatham et al 2004).
AIRWAY SUCTION
Airway suction for intubated adults is described in Chapter 8 and for children, including the non-intubated infant and small child, see Chapter 10. Suction is required occasionally in the non-intubated adult who has retained secretions, usually via the nasopharynx.
Nasotracheal suction is a means of stimulating a cough, but is an unpleasant procedure for the patient and should be performed only when absolutely necessary. The indication for suction is the inability to cough effectively and expectorate, when airway secretions are retained. It may be necessary, for example, when an acute exacerbation of chronic bronchitis has led to carbon dioxide narcosis and respiratory failure, in neurological disorders, postoperative complications or laryngeal dysfunction. Before airway suction is undertaken it is very important that the procedure is explained carefully to the patient.
It is important to be aware of the possibility of causing laryngeal spasm (Sykes et al 1976) or vagal nerve stimulation, which may lead to cardiac arrhythmias (Jacob 1990). Nasopharyngeal suction is contraindicated when there is stridor or severe bronchospasm, and in patients with head injuries when there is a leak of cerebrospinal fluid into the nasal passages. Retention of secretions may be a problem in patients with respiratory muscle paralysis, but there is usually no benefit in using airway suction in an attempt to stimulate an effective cough. It is the lack of volume of air that prevents clearance of secretions in these patients and techniques such as mechanical insufflation/exsufflation, assisted cough, intermittent positive pressure breathing, glossopharyngeal breathing and gravity-assisted positioning should be considered.
Airway suction causes damage to the tracheal epithelium and this can be minimized by the appropriate choice of catheter and careful technique (Brazier 1999). A flexible catheter of suitable size, usually 12 FG in adults, should be lubricated with a water-soluble jelly and gently passed through the nasal passage so that it curves down into the pharynx. Occasionally a cough may be stimulated when the catheter reaches the pharynx and suction can then be applied, the secretions aspirated and the catheter withdrawn. More often it is necessary to pass the catheter between the vocal cords and into the trachea to stimulate coughing. The catheter is less likely to enter the oesophagus if the neck is extended, and if the patient is able to cooperate it is often helpful if they can put their tongue out. The catheter should be inserted during the inspiratory phase and if it passes into the trachea, will stimulate vigorous coughing. When suction is applied the vacuum pressure should be kept as low as possible, usually in the range 60–150 mmHg (8.0–20 kPa), although this will vary depending on the viscosity of the mucus. A built-in fingertip control or Y-connector is recommended to allow a more gradual build-up of suction pressure than is possible by the release of a kinked catheter tube.
Oxygen should always be available during the suction procedure and the patient observed for signs of hypoxia. If it has been difficult to insert the catheter and the patient looks cyanosed, instead of withdrawing the catheter from the trachea, suction should be stopped and oxygen administered until the patient’s colour has improved. Suction can then be restarted.
Suction can be performed in adults who are being nursed in the sitting position, but comatosed patients should be suctioned in side lying to avoid the possibility of aspiration if vomiting occurs.
The nasopharyngeal suctioning clinical practice guidelines of the American Association for Respiratory Care (2004) provide useful evidence-based points on the technique.
Oropharyngeal suction through an airway is an alternative method, if suction is necessary. An oropharyngeal airway is a plastic tube shaped to fit the curved palate. It is inserted with its tip directed towards the roof of the mouth and is then rotated so that the tip lies over the back of the tongue.
Portable suction units are available for domiciliary use and for patients in transit. They may be powered manually, by mains electricity or by battery, for example from a car cigarette lighter adapter.
Provided suction is carried out carefully and oxygen is always available, it is a valuable technique and may avoid the need for more invasive procedures such as bronchoscopy, endotracheal intubation or minitracheotomy. However, it should not be undertaken until every attempt to achieve effective coughing has failed.
BREATHING CONTROL
Breathing techniques can be divided into normal breathing, known as ‘breathing control’, where the pattern of breathing is maximally efficient for the individual with minimal effort expended, and breathing exercises, where either inspiration is emphasized (as in thoracic expansion exercises and inspiratory muscle training) or expiration is emphasized (as in the huff of the forced expiration technique).
Breathing control is normal tidal breathing using the lower chest and encouraging relaxation of the upper chest and shoulders. This used to be known as ‘diaphragmatic breathing’, but the term is a misnomer as during normal tidal breathing there is activity not only in the diaphragm but also in the internal and external intercostal muscles, the abdominal and scalene muscles (Green & Moxham 1985). This pattern of breathing is not appropriate for everyone. Dyspnoea has been shown to increase in some people with chronic obstructive pulmonary disease using ‘diaphragmatic breathing’ owing to an increase in asynchronous and paradoxical breathing movements (Gosselink 2004, Gosselink et al 1995 and Chapter 13).
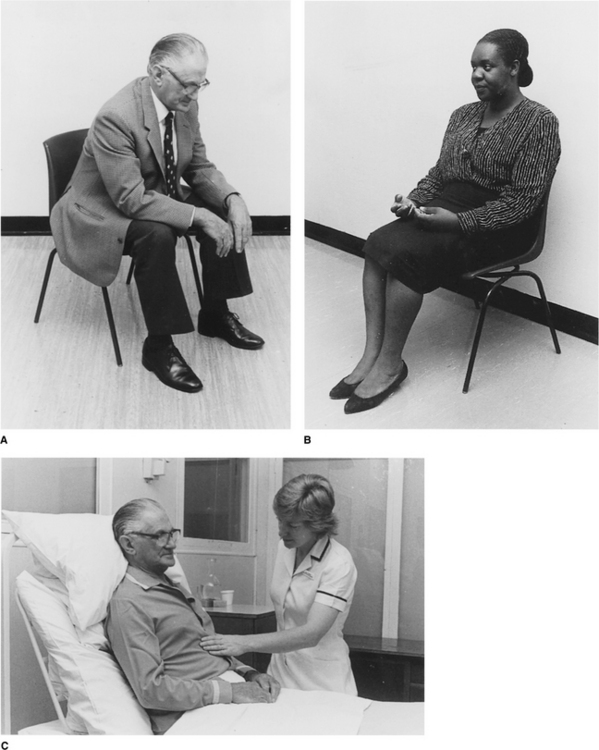
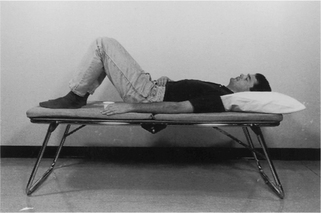
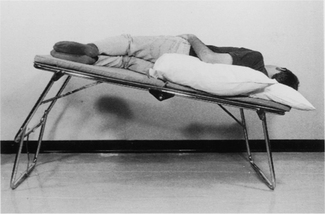
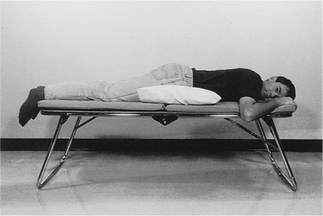
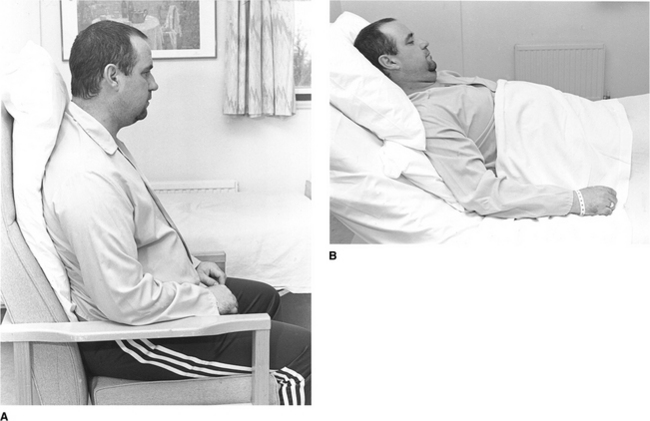
To be taught breathing control, the patient should be in a comfortable well-supported position either sitting (Fig. 5.27) or in high side lying (Fig. 5.28). The patient is encouraged to relax the upper chest, shoulders and arms. One hand, which may be either the patient’s or the physiotherapist’s or one hand of each, can be positioned lightly on the upper abdomen. As the patient breathes in, the hand should be felt to rise up and out; as the patient breathes out, the hand sinks down and in. Inspiration using the lower chest in this way is the active phase. Expiration should be relaxed and passive, and both inspiration and expiration should be barely audible. Inspiration through the nose allows the air to be warmed, humidified and filtered before it reaches the upper airways. If the nose is blocked, breathing through the mouth will reduce the resistance to the flow of air and reduce the work of breathing. If the patient is very breathless, breathing through the mouth will reduce the anatomical dead space.
Many breathless patients, for example those with emphysema, asthma, pulmonary fibrosis or lung cancer, may benefit from using breathing control in positions that encourage relaxation of the upper chest and shoulders and allow movement of the lower chest and abdomen. These positions also optimize, by leng-thening, the length tension status of the diaphragm (American Thoracic Society 1999, Dean 1985, O’Neill & McCarthy 1983, Sharp et al 1980). When the patient is sitting or standing leaning forward, the abdominal contents raise the anterior part of the diaphragm, probably facilitating its contraction during inspiration. A similar effect can be seen in the side lying and high side lying positions where the curvature of the dependent part of the diaphragm is increased. This effect, combined with relaxation of the head, neck and shoulders, promotes the pattern of breathing control.
 high side lying (see Fig. 5.28) – for maximal relaxation of the head, neck and upper chest, the neck should be slightly flexed and the top pillow should be above the shoulder, supporting only the head and neck.
high side lying (see Fig. 5.28) – for maximal relaxation of the head, neck and upper chest, the neck should be slightly flexed and the top pillow should be above the shoulder, supporting only the head and neck.These positions discourage the tendency of breathless patients to push down or grip with their hands, which causes elevation of the shoulders and overuse of the accessory muscles of breathing, and may result in a more effective length tension status of the diaphragm.
Breathing control may also be used to improve exercise capacity in breathless patients when walking up slopes, hills and stairs (Fig. 5.32). Breathless patients tend to hold their breath on exertion and rush, for example, up a flight of stairs arriving at the top extremely breathless and unable to speak. The simple technique of relaxing the arms and shoulders, reducing the walking speed a little and using the pattern of breathing in on climbing up one step and breathing out on climbing up the next step may lead to a marked reduction in breathlessness (Webber 1991). When this technique has been mastered some patients, on days when they are less breathless, may find breathing in for one step and out for two steps more comfortable. The severely breathless patient may also find the combination of breathing control with walking helpful when walking on level ground. A respiratory walking frame (see Fig. 13.4) with or without portable oxygen can be used to assist ambulation in the severely breathless patient.
Breathing control can also be used to control a bout or paroxysm of coughing.
Some people with chronic respiratory disease automatically use pursed-lip breathing. Breathing through pursed lips has the effect of generating a small positive pressure during expiration, which may to some extent reduce the collapse of unstable airways.
CONTINENCE
Management of stress incontinence
Many people with chronic respiratory disease who require airway clearance techniques have stress incontinence of urine, and sometimes faeces. Urinary incontinence, the involuntary loss of urine, is associated with adverse effects on quality of life (National Institute for Health and Clinical Excellence 2006). The prevalence of urinary incontinence in the population as a whole increases with increasing age up to 46% in women and 34% in men aged over 80 years (Scottish Intercollegiate Guidelines Network 2004). The prevalence associated with chronic lung disease is known to be higher and at an earlier age, for example in people with cystic fibrosis incontinence in women ranges from 38–68% (Cornacchia et al 2001, Orr et al 2001, White et al 2000); in men 16% (Gumery et al 2005); and in children 14–33% (Moraes et al 2002, Prasad et al 2006).
Urinary incontinence frequently reduces adherence to an airway clearance technique and often causes embarrassment. During assessment (Chapter 1) the physiotherapist should include questions to identify this problem, even in young girls with chronic respiratory disorders. Voluntary contraction of the pelvic floor muscles just before and throughout a cough or huff, known as ‘The Knack’, can be used to reduce stress-related leakage of urine (Miller et al 1998). If this technique does not lead to a clinically significant reduction in leakage, referral to a physiotherapist specializing in continence therapy should be considered.
GLOSSOPHARYNGEAL BREATHING
Glossopharyngeal breathing (GPB) is a technique useful in patients with a reduced vital capacity resulting from respiratory muscle weakness or paralysis. Although its original use was in rehabilitation of patients with poliomyelitis, it can be invaluable when taught to people with tetraplegia (Alvarez et al 1981, Bach & Alba 1990, Bianchi et al 2004, Warren 2002) and in some people with neuromuscular diseases (Bach 1995, Baydur et al 1990).
GPB was first described by Dail (1951) when patients with poliomyelitis were observed to be gulping air into their lungs. It was this gulping action that gave the technique the name ‘frog breathing’. GPB is a form of positive pressure ventilation produced by the patient’s voluntary muscles where boluses of air are forced into the lungs. It is essential to understand that GPB is not swallowing air into the stomach, but gulping air into the lungs.
Paralysed patients dependent on a mechanical ventilator are sometimes able to use GPB continuously, other than during sleep, to substitute the mechanical ventilation. GPB is very useful in patients who are able to breathe spontaneously but whose power to cough and clear secretions is inadequate. The technique also enables these patients to shout to attract attention and to help to maintain or improve lung and chest wall compliance (Bach et al 1987, Dail et al 1955). For patients dependent on a ventilator, either non-invasively or via a tracheostomy, GPB can be life saving (Bach 1995) in an emergency if the ventilator becomes disconnected or if there should be a power failure, and can increase the feeling of independence (Make et al 1998). The uses of GPB are summarized in Box 5.1.
Box 5.1 Uses of glossopharyngeal breathing
Where the lungs are normal, but the respiratory muscles are weak or paralysed:
To breathe in, a series of pumping strokes is produced by action of the lips, tongue, soft palate, pharynx and larynx. Air is held in the chest by the larynx, which acts as a valve as the mouth is opened for the next gulp. Expiration occurs by normal elastic recoil of the lungs and rib cage.
Before starting to teach GPB it may be helpful for the patient to experience the feeling of inflating the chest by using an intermittent positive pressure ventilator with a mouthpiece. After inflating the lungs, the mouthpiece is removed and the patient should try and hold all the air in the lungs with the mouth open, avoiding escape of air through the larynx or nose. A teaching video is available that may help both patient and physiotherapist when learning or teaching GPB (Webber & Higgens 1999).
Each gulp of air is made up of three stages (Box 5.2). The first and most important stage (Box 5.3 & Fig. 5.33A) in learning GPB is the enlargement of the mouth and throat cavity by depressing the tracheal and laryngeal cartilages. The tongue should remain flat with the tip touching the inside of the lower teeth. Using a torch, the physiotherapist should be able to see the uvula when this is correct. The patient may find it helpful to watch the movement in a mirror and possibly by feeling the cartilages with their fingers. An exercise that often helps to initiate movement of the cartilages is for the physiotherapist to place a finger horizontally under the chin back against the trachea and give pressure in an upwards direction resisting downward movement of the base of the tongue. To emphasize the downward cartilage movement, practice should progress to holding the throat in this open position for 3–5 seconds.
Box 5.3 Stage 1 (Fig. 5.33A)
When this movement has been achieved, stages 2 and 3 are added to complete the cycle (Box 5.4 & Fig. 5.33B, Box 5.5 & Fig. 5.33C). The cycle should be practised slowly at first and then gradually speeded up until the movement flows. It is important to avoid stages 2 and 3 being done simultaneously. The air has to be trapped in the throat in stage 2 before the jaw and cartilages are allowed to rise in stage 3. It is essential to maintain the lowered jaw during stage 2.
Box 5.4 Stage 2 (Fig. 5.33B)
Box 5.5 Stage 3 (Fig. 5.33C)
After opening throat (stage 1) and trapping air with lips (stage 2):
Let floor of mouth, tongue and cartilages rise to the normal position while air is pumped through the larynx
The next stage is to take a maximum breath in and, while holding this breath, to add several glossopharyngeal gulps, to augment the vital capacity. A ‘normal’ breath must not be taken before each gulp.
When correct, the patient will feel the chest filling with air and the physiotherapist can test the ‘GPB vital capacity’ by putting a mouthpiece attached to the expiratory limb of a Wright’s respirometer in the patient’s mouth before exhalation.
The respirometer can be used to measure the volume per gulp; the patient will require less effort and reach maximum capacity more quickly if a large volume gulp is achieved. The volume per gulp is directly related to the amount of downward movement of the cartilages. A study by Kelleher & Parida (1957) reported a group of patients in whom the average volume per gulp varied from 25 to 120 ml and when teaching GPB an attempt should be made to achieve at least 80 ml per gulp.
If GPB were being used continuously as a substitute for normal tidal breathing, approximately 6–8 gulps would be taken before breathing out. When used for clearance of secretions, 10–25 gulps may be required to obtain a maximal vital capacity. Volumes of 2.5–3.0 litres can be achieved and the expiratory flow produced is sufficient to mobilize secretions (Make et al 1998).
GPB would normally be taught with the patient in a comfortable sitting position, but for patients with postural hypotension following spinal cord injury, a reclined position would be more appropriate at first. When mastered, GPB should be practised in positions useful for the patient to clear bronchial secretions. After filling the chest to capacity the patient signals to the physiotherapist who compresses the chest as the air is let out. The patient may have sufficient muscle power to apply compression, or carers can be taught to give assistance.
GPB is learnt easily by some patients, but others need time and patience to acquire this skill. Although frequent self-practice can be helpful in the learning stages it is recommended to ensure correct opening of the throat (stage 1) before letting the patient proceed to stages 2 and 3. It can be tiring to learn GPB and therefore short frequent sessions are most effective. Once learnt, it is nottiring to use the technique.
There are reasons why a person may fail to achieve GPB initially. The soft palate may not be closing, so air passes out through the nose instead of into the trachea. This can often be corrected by asking the patient to take several gulps while the nose is alternately pinched closed for two gulps and released for two gulps. When the nose is held closed, the patient will feel pressure inside the mouth and throat and possibly in the ears. GPB should then be repeated without the nose being pinched and the patient should attempt to reproduce the same feeling of pressure with every gulp. Occasionally patients have very poor soft palate control and will need to wear a nose clip. Exercises to improve soft palate control may be helpful.
A few patients who find it difficult to stop air leaking through the nose achieve GPB by taking the gulps through the nose, while keeping the mouth shut. Other people prefer to do nasal GPB as it is less obtrusive and the gulps of air are humidified. It is important initially to teach the stages using the mouth technique in order to observe the tongue position. The stages for nasal GPB are shown in Box 5.6.
Another problem that may be found during the learning stages of GPB is weakness of the vocal cords. If the cords are unable to hold the air in the lungs, the vital capacity is decreased after attempting some gulps. This can be tested by asking the patient to take a very deep breath using intermittent positive pressure by mouthpiece and then, with the mouthpiece removed, he should say ‘Ah, ah, ah …’ in short staccato bursts during expiration. If all the ‘Ahs’ run into a continuous sound, the cords are failing to shut off the flow of air under pressure. To strengthen the cords, the patient can start to take a volume of air just greater than his vital capacity and practise the ‘Ah, ah’ sounds. When this is achieved, the volume of air can be gradually increased and the expiration exercises repeated.
GPB is a valuable technique to consider when treating tetraplegics with a vital capacity of less than 2 litres. Instruction can begin when the patient has reached a stable condition, but it is inappropriate in the acute phase or during an acute chest infection. When successfully learnt, it is invaluable during a period of chest infection to assist in the clearance of secretions. In patients with Duchenne muscular dystrophy and other neuromuscular diseases it would often be easiest to teach GPB as a ‘normal’ part of treatment, before deterioration in respiratory muscle function and difficulty in clearing secretions occurs. GPB has been of benefit to patients with motor neurone disease (Bereiter 2001) by making the voice more audible and providing an effective cough; and in multiple sclerosis (Aldridge 2005), allowing effective clearance of secretions.
It is possible to teach GPB to patients with an uncuffed tracheostomy tube, if there is a dressing providing an effective seal around the tube to avoid air leaks and some form of plug to the tube. A one-way valve, for example a Passy–Muir valve, fitted between the tracheostomy tube and the ventilator tubing acts as a plug preventing air escaping into the ventilator tubing, but allowing it to enter the lungs. If a ventilator-dependent patient has a one-way valve in situ and can do GPB, it gives a great sense of security should a ventilator tube become kinked or disconnected. Before starting to learn GPB, it is essential that patients who are entirely ventilator-dependent learn to use intermittent positive pressure by mouthpiece. It gives them a reliable respiratory support during the learning stages. While increasing the period of time using GPB, it is important to ensure that normal blood gases are maintained. Patients usually take 6–8 gulps per breath, 10–12 times per minute, to provide a normal minute volume. When learning to build up their endurance they may experience a sense of desperate need for air but by taking a larger number of gulps (perhaps 20 or 25) at high speed, this feeling can be relieved and the normal pattern resumed.
Weakness of the oropharyngeal muscles may make it impossible to do GPB, but it can be successful with mild weakness. The technique is contraindicated in patients with obstructive airways disease where the positive pressure could increase air trapping. It is also contraindicated in cardiac failure, where the long inspiratory period with high intrathoracic pressure could decrease venous return, causing a fall in blood pressure.
To learn GPB requires commitment from both patient and physiotherapist and, if taught as an outpatient, tuition sessions at least weekly are recommended to maintain motivation. Filming the patient on video to give visual feedback is sometimes a useful teaching tool. GPB can be a much more versatile breathing technique than using a mechanical insufflator/exsufflator. It requires no equipment and in addition to making coughing more effective, for many patients it gives confidence when away from the home ventilator allowing greater independence and improved quality of life. When taught successfully a physiotherapist gains confidence and sees the benefits it can give to patients. Bach (1992) wrote that although potentially extremely useful, GPB is rarely used because there are few healthcare professionals familiar with the technique. Many patients would benefit if physiotherapists familiarized themselves with the technique and included it as a treatment option when indicated.
GRAVITY-ASSISTED POSITIONING
Gravity-assisted positions can be used to:
Clearance of bronchial secretions.
Gravity has been shown to assist clearance of bronchial secretions (Eaton et al 2007, Ewart 1901, Hofmeyr et al 1986, Sutton et al 1983). Nelson (1934) described the use of positioning for draining secretions based on the anatomy of the bronchial tree. The recognized positions (Thoracic Society 1950) (Fig. 5.34) are shown in Figures 5.35-5.45 and described in Table 5.1.
Figure 5.34 Diagram illustrating the bronchopulmonary nomenclature approved by the Thoracic Society (1950).
(Reproduced with permission of the Editor of Thorax)
Table 5.1 Gravity-assisted drainage positions (numbers refer to Fig. 5.34 and patient position is shown in Figs 5.35-5.45)
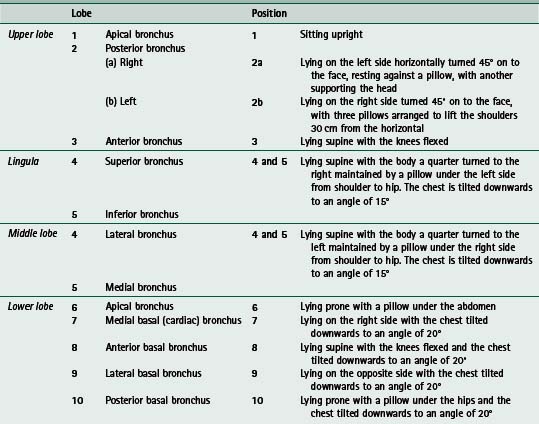
The effects of gravity on airway clearance are likely to be a consequence of both drainage and the increase in ventilation (Lannefors & Wollmer 1992) and specific positioning is likely to be of significance only, for example, in the drainage of an identified lung abscess or specific areas of atelectasis. Cecins et al (1999) demonstrated that the side lying position was as effective as the head-down tipped position for people with excess bronchial secretions as a consequence of chronic respiratory disease (non-cystic fibrosis bronchiectasis and cystic fibrosis). Eaton et al (2007) demonstrated that the inclusion of gravity-assisted positioning increased the wet weight of sputum expectorated in patients with non-cystic fibrosis bronchiectasis. In patients with cystic fibrosis the upper lobes are frequently the most severely affected, although the cause is unknown (Tomashefski et al 1986) and positions other than sitting may be indicated only occasionally.
Individual assessment will indicate whether gravity- assisted drainage positions are of clinical benefit. In some patients with very tenacious secretions, gravity is unlikely to help and a comfortable position in which effective breathing techniques can be carried out is likely to be the most beneficial. It is inappropriate to use the head-down tipped positions immediately following meals and in the following conditions: cardiac failure, severe hypertension, cerebral oedema, aortic and cerebral aneurysms, severe haemoptysis, abdominal distension, gastro-oesophageal reflux and after recent surgery or trauma to the head or neck.
Ventilation/perfusion.
In the adult, both ventilation and perfusion are preferentially distributed to the dependent parts of the lung (West 2004). In children this differs and there is a preferential distribution of ventilation to the uppermost lung and perfusion to the dependent lung (Chapter 10). In adults with unilateral lung disease, gas exchange may be improved by using the side lying position with the unaffected lung dependent (Zack et al 1974). Postoperatively the easiest method of increasing functional residual capacity (FRC) and preventing lung collapse is appropriate positioning and early ambulation (Jenkins et al 1988). Most patients can sit out of bed the day of or the day after surgery (Fig. 5.46A). If they cannot sit in a chair, they should be encouraged to sit either upright in bed or to adopt a side-lying position, but they should not be in a slumped sitting position as this will reduce lung volumes (Fig. 5.46B). See also Chapter 4, Effects of positioning and mobilization.
INCENTIVE SPIROMETRY
Incentive spirometers are mechanical devices that were originally introduced in surgical patients in an attempt to reduce postoperative pulmonary complications, by increasing inspiratory capacity. The device is activated by the patient’s inspiratory effort. When a slow deep inspiration is performed, with the lips sealed around the mouthpiece (Fig. 5.47), the ongoing inspiration is motivated by visual feedback, for example a ball rising to a preset marker. The patient aims to generate a predetermined flow or to achieve a preset volume and is encouraged to hold at full inspiration for 2–3 seconds. A short, sharp inspiration can activate the flow-generated incentive spirometry devices with little increase in tidal volume, but with a volume-dependent device an increase in tidal volume must be achieved before the preset level can be reached. The increased work of breathing required should be considered in patients at risk of inspiratory muscle fatigue and in patients with severely impaired respiratory muscle function. Spirometers with a low imposed work of breathing should be considered, if appropriate for these groups, and in some postoperative patients (Weindler & Kiefer 2001).
The pattern of breathing while using an incentive spirometer is important. Expansion of the lower chest should be emphasized rather than the use of the accessory muscles of respiration, which would encourage expansion of the upper chest. Diaphragmatic movement (Chuter et al 1990) is thought to be an important factor in the prevention of postoperative pulmonary complications. Incentive spirometry has been shown to increase abdominal movement in normal subjects, but not in subjects following abdominal surgery (Chuter et al 1989). Postoperatively, an increase in diaphragmatic movement has been observed by encouraging an increase in lung volume while using the pattern of breathing control without the resistive loading of an incentive spirometer (Chuter et al 1990). This may help to reduce postoperative pulmonary complications by increasing ventilation to the dependent parts of the lungs.
A systematic review by Overend et al (2001) examined the use of incentive spirometry for the prevention of postoperative pulmonary complications (PPCs). The authors concluded that the evidence does not support the use of incentive spirometry for decreasing the incidence of PPCs following cardiac or upper abdominal surgery. Gosselink et al (2000) undertook a randomized controlled trial to investigate the additional effect of incentive spirometry to chest physiotherapy in the prevention of PPCs following thoracic surgery for lung and oesophageal resections. The addition of incentive spirometry to physiotherapy did not further reduce either pulmonary complications or hospital stay. How-ever, a systematic review for the American College of Physicians on strategies to reduce PPCs following non-cardiothoracic surgery (Lawrence et al 2006) concluded that any type of lung expansion intervention is better than no prophylaxis.
There may be a place for the use of incentive spirometry, to increase lung volume following surgery, in children and in some adolescents to provide motivation and in patients who are immobilized, but with significant advances in anaesthesia and surgery (including the increasing use of minimally invasive surgery) ambulation may be the ‘treatment’ of choice for the majority of patients who are able to mobilize postoperatively.
INHALATION: DRUGS AND HUMIDIFICATION
Inhalation therapy has progressed significantly over the past two decades. ‘Intelligent’ nebulizers have reduced inhalation time and improved the efficacy of drug delivery. However, an understanding of the aerosol particle and its pattern of deposition within the airways remains essential when considering the delivery of drugs by the inhaled route and many ‘conventional’ systems are still in use.
A suspension of fine liquid or solid particles in air is known as an ‘aerosol’. The pattern of deposition of aerosol particles within the bronchial tree depends on particle size, method of inhalation and on the degree of airflow obstruction (Newman et al 1986). Large particles in the size range 5–10 μm deposit by impaction in the oropharynx and upper airways where the cross-sectional diameter of the airway is small and the airflow high. The total cross-section of the airway increases rapidly beyond the 10th generation of bronchi, airflow slows significantly and particles of 0.5–5 μm, known as the ‘respirable particles’, deposit in the small airways and alveoli by gravitational sedimentation. It is the particles of less than 2 μm that reach the alveoli. Gravitational sedimentation is time dependent and enhanced by breath holding. A more central patchy deposition is seen in patients with airflow obstruction (Clarke 1988).
The topical deposition of a drug by inhalation allows a smaller dose to be given than when other routes are used, the onset of action is often more rapid and with minimal systemic absorption the side effects are reduced.
Metered dose inhaler (MDI) and dry powder inhaler (DPI)
Numerous devices are available for the inhalation of drugs, ranging from the simple MDI or powder inhalers to breath-actuated inhalers and a variety of nebulizers. The physiotherapist should be aware of the range of possibilities to enable the patient to gain maximum benefit from the prescribed drugs. The choice of device will depend on the patient’s age, coordination and dexterity, severity of the respiratory condition and patient preference.
Practice with placebo inhalers may be necessary to perfect the technique. Even if a patient has been using an inhalation device for a long time, it is important to observe and review the technique, as it may not be effective.
To gain maximum effect from a pressurized MDI (pMDI), the device should first be shaken to ensure that the drug is evenly distributed in the propellant gases. The inhaler is held upright and the cap is removed. The patient breathes out gently but not fully and then, with the mouth around the mouthpiece of the inhaler, the device is pressed to release the drug as soon as inspiration has begun. The breath in should be slow and deep and inspiration should be held for 10 seconds if possible, before breathing out gently through the nose (Burge 1986, Clarke 1988). Effective technique is essential as it has been shown that as little as 10% of the drug actually reaches the lungs (Clarke 1988).
Frequently, the prescribed dose will involve the inhalation of more than one ‘puff’. It is recommended that puffs be taken one after the other. If the inhalation technique described above is used, the length of time between inhalations is likely to be 15–20 seconds, which allows sufficient time to overcome the problem of cooling of the metering chamber (pMDI) as the gas evaporates. Compliance is improved when doses are taken one after the other (Burge 1986).
Spacers can be used to improve the deposition of the drug in the lungs. Large volume spacers (Fig. 5.48) have been shown to improve this by approximately 15% (Clarke 1988) and to reduce the deposition in the oropharynx, as the larger particles drop out in the spacer rather than the oropharynx. The patient is encouraged to take a slow deep breath with a hold, but if this is difficult, tidal breathing can be used. Gleeson & Price (1988) showed that a bronchodilator was equally effective when a child breathed several times at tidal volume through a spacer when compared with a deep breath and inspiratory hold.
For people with poor coordination a breath-actuated inhaler may be considered (Newman et al 1991) or a dry powder device. The dry powder inhalers may also be breath-actuated, releasing the drugs on inspiration, but require a faster inspiratory flow rate than a pMDI. The inspiratory flow required depends on the resistance within the device. It is not only important that the patient can use the device effectively but also that the patient or parent can easily recall whether a dose of the drug has been taken and many devices incorporate a monitoring system.
The British Guideline on the Management of Asthma (British Thoracic Society & Scottish Intercollegiate Guidelines Network 2004) outlines current evidence for the inhalation of β2-agonists (short- and long-acting), anticholinergics and steroids in adults and children and the recommended device. Oral candidiasis can be minimized by rinsing the mouth thoroughly following inhalation of steroids.
Nebulizers
A nebulizer may be used for the inhalation of drugs, if a more simple method is not optimally effective or if the drug cannot be administered by other means. Lannefors (2006) has reviewed the practical considerations for nebulizer therapy. A nebulizer converts a solution into aerosol particles (fine droplets), which are suspended in a stream of gas. It delivers a therapeutic dose of a prescribed drug as an aerosol in the form of respirable particles (particles <5 μm in diameter) and may be ‘continuously running’ or ‘breath actuated’. There are several types of nebulizers.
Jet nebulizers
With the jet nebulizer, a driving gas (electric air compressor or compressed air or oxygen from a hospital line or cylinder) is forced through a narrow orifice. The negative pressure created around the orifice draws the drug solution up the feed tube from the liquid reservoir and the jet of gas fragments the liquid into droplets. A screening baffle allows the smaller particles in the form of a mist to be available for inhalation by the patient and the larger particles to drop back into the reservoir to be recycled (Medic-Aid Ltd 1996).
Ultrasonic nebulizers
An aerosol can also be created by high-frequency (1–2 MHz) sound waves. An electric current applied to a piezo-electric crystal causes ultrasonic vibrations. The sound waves will travel through a liquid to the surface where they produce an aerosol. The particle size is influenced by the frequency of oscillation of the crystal. Ultrasonic nebulizers can produce a higher output than jet nebulizers and operate quietly.
Vibrating mesh technology systems
Vibrating mesh systems employ a vibrating mesh or plate with multiple apertures to generate a fine-particle, low-velocity aerosol (Dhand 2002). This development has led to shorter treatment times together with improved efficiency and efficacy of drug delivery (Fig. 5.49). Adaptive aerosol delivery systems use mesh technology in the generation of aerosol particles. They identify and adapt to the patient’s breathing pattern and deliver a preset volume of the drug during the first part of each inspiratory breath. They are programmed to deliver a total preset dose of a particular drug and give feedback on completion of administration of the dose (McGuire 2006). With an adaptive aerosol delivery system almost no drug is lost during expiration, obviating the need for the use of filters or wide-bore tube systems and minimizing ‘wasted’ medication. The residual volume is about 0.1 ml (McGuire 2006).
Nebulizer performance
The performance of a nebulizer can be measured by its respirable output. The respirable output is the mass of respirable particles (particles less than 5 μm in diameter) produced per minute, that is:
aerosol output (mg/min) × respirable fraction
The respirable fraction is the percentage of respirable particles within the aerosol output. It is recommended that a nebulizer should provide a respirable fraction of at least 50% at its recommended driving gas flow (British Standards Institution 1994) and a number of nebulizers exceed this level. The performance of an individual nebulizer has often been described by the mass median aerodynamic diameter (MMAD) of the particles. The MMAD indicates the range of size of particles leaving the nebulizer. Half of the aerosol mass from the nebulizer is of particles smaller than the MMAD and half of the aerosol mass is of particles larger than the MMAD. This is a less useful measurement of nebulizer performance, as it is not related to the mass of drug. It is important to consider an air compressor and nebulizer system as a unit and they should be matched to provide an acceptable output (Kendrick et al 1995). Factors that affect individual nebulizer performance include the following (Nebuliser Project Group 1997):
Driving gas.
Most jet nebulizers operate efficiently with a flow rate of 6–8 l/min. For patients in hospital it is often convenient to use the piped oxygen supply and in hypoxic patients without carbon dioxide retention, oxygen should be used. For patients retaining carbon dioxide who are dependent on their hypoxic drive to stimulate breathing, compressed air should be the driving gas (Gunawardena et al 1984). Occasionally it may be appropriate to increase the inspired oxygen concentration by entraining a low flow of oxygen.
Nebulizer chamber design.
With a ‘conventional’ nebulizer the output flow (from the nebulizer, towards the patient) is equal to the input flow (from the driving gas source) and nebulization is continuous. With a ‘Venturi’ nebulizer the output flow is greater than the input flow due to the presence of an open vent, but the output flow does not change with the patient’s breathing pattern. With an ‘active Venturi’ nebulizer the output flow is breath assisted and is increased during inspiration. There is therefore less wastage of the drug during expiration. When selecting a nebulizer the age of the patient must be considered. The inspiratory flow of an infant will probably be less than the output from a Venturi nebulizer. The concentration of the aerosolized drug may therefore be increased as there will be little or no air entrainment, but the total dose of the drug may be reduced (Collis et al 1990). Consideration should also be given to the optimal particle size of the drug to be delivered. Inhaled pentamidine or antibiotics need to be delivered to the more peripheral airways (requiring a high percentage of particles of less than 2 μm), whereas bronchodilator drugs probably have their effect in the more central airways.
Residual volume.
This is the volume of solution that remains in the nebulizer after nebulization has stopped. The Nebuliser Project Group of the British Thoracic Society (1997) recommends that a fill volume of 2–2.5 ml may be adequate if the residual volume is less than 1 ml, but nebulizers with a higher residual volume will probably require a fill volume of 4 ml. The patient should be encouraged to tap the side of the nebulizer to allow as much as possible to be delivered (Everard et al 1994).
Fill volume.
For effective nebulization, it is important not to exceed the manufacturer’s recommended fill volume.
Physical properties of drug solution or suspension.
Most bronchodilator solutions when nebulized have a similar volume output to normal (0.9%) sodium chloride, but solutions with a higher viscosity or a high surface tension (e.g. carbenicillin) are slow to nebulize, and other characteristics of the aerosol output may change.
Breathing pattern of the patient.
The optimal pattern of breathing using a nebulizer other than an adaptive aerosol delivery device has not yet been ascertained, but a recommended one is to intersperse one or two slow deep breaths with breathing at tidal volume. The deep breathing may increase peripheral deposition of the drug and the periods of breathing control will prevent hyperventilation. When inhaling from a nebulizer, the patient should be in a comfortable and well-supported position.
Drug delivery
A range of drugs often administered via a nebulizer include bronchodilators, corticosteroids, antibiotics, pentamidine, antifungals, local anaesthetics, rhDNase and surfactant. The nebulizer system used must be appropriate for the drug that has been prescribed. The evidence for drug device matching is outlined in the European Respiratory Society nebulizer guidelines: clinical aspects (2000). It is important to remember that the relative proportions of the airway and the anatomy and structure of the lung will alter during childhood. This will affect the deposition of inhaled drugs (Barry et al 2000).
Other points for consideration
 The dose of a prescribed nebulized bronchodilator may seem large compared with that from a pressurized aerosol, but only 10–20% of the initial dose, using a conventional jet nebulizer, is received by the patient and only 50% of this reaches the lungs. The drug that does not reach the patient is lost in the equipment and exhaled gas (Lewis & Fleming 1985).
The dose of a prescribed nebulized bronchodilator may seem large compared with that from a pressurized aerosol, but only 10–20% of the initial dose, using a conventional jet nebulizer, is received by the patient and only 50% of this reaches the lungs. The drug that does not reach the patient is lost in the equipment and exhaled gas (Lewis & Fleming 1985). A facemask is necessary for an infant or small child (Fig. 5.50), but as soon as the child is able to cooper-ate, a mouthpiece should be used to minimize deposition of the drug on the face and in the nasal passages (Wolfsdorf et al 1969). Other disadvantages of a mask are facial skin irritation from nebulized antibiotics and steroids and nebulized ipratropium bromide and salbutamol by mask have been associated with glaucoma in a group of adults with chronic airflow limitation (Shah et al 1992).
A facemask is necessary for an infant or small child (Fig. 5.50), but as soon as the child is able to cooper-ate, a mouthpiece should be used to minimize deposition of the drug on the face and in the nasal passages (Wolfsdorf et al 1969). Other disadvantages of a mask are facial skin irritation from nebulized antibiotics and steroids and nebulized ipratropium bromide and salbutamol by mask have been associated with glaucoma in a group of adults with chronic airflow limitation (Shah et al 1992). For infants and children, parents should be given written instruction, in addition to verbal instruction, to improve adherence to treatment (Barry et al 2000).
For infants and children, parents should be given written instruction, in addition to verbal instruction, to improve adherence to treatment (Barry et al 2000). For nebulized antibiotics and pentamidine a one-way valve system is recommended (Wilson et al 2000). This can be achieved either with an effective filter on the exhalation port (Fig. 5.51) or wide-bore tubing to allow the exhaled gas to be vented out through a window. This is to prevent small quantities of antibiotics remaining in the atmosphere, which could lead to patients, family members and medical personnel in the vicinity receiving a subtherapeutic dose (Smaldone et al 1991) and environmental organisms becoming resistant to the antibiotic (Sanderson 1984). A nose clip is necessary if the patient is breathing partially through the nose. A bronchoconstriction test should be undertaken at the time of the first inhalation of an antibiotic or pentamidine.
For nebulized antibiotics and pentamidine a one-way valve system is recommended (Wilson et al 2000). This can be achieved either with an effective filter on the exhalation port (Fig. 5.51) or wide-bore tubing to allow the exhaled gas to be vented out through a window. This is to prevent small quantities of antibiotics remaining in the atmosphere, which could lead to patients, family members and medical personnel in the vicinity receiving a subtherapeutic dose (Smaldone et al 1991) and environmental organisms becoming resistant to the antibiotic (Sanderson 1984). A nose clip is necessary if the patient is breathing partially through the nose. A bronchoconstriction test should be undertaken at the time of the first inhalation of an antibiotic or pentamidine. If inhaled antibiotics are prescribed for a pseudomonal infection in the upper respiratory tract, for example a patient with cystic fibrosis following lung transplantation, a nebulizer producing large particles is necessary (Webb et al 1996). A mask should be used and the patient encouraged to breathe through the nose.
If inhaled antibiotics are prescribed for a pseudomonal infection in the upper respiratory tract, for example a patient with cystic fibrosis following lung transplantation, a nebulizer producing large particles is necessary (Webb et al 1996). A mask should be used and the patient encouraged to breathe through the nose. More than one antibiotic may be prescribed. A few antibiotics are compatible when mixed, but others must be inhaled separately. Either normal saline or sterile water is used to reconstitute a powdered antibiotic or to make a prescribed solution up to the necessary volume for nebulization. Information on the advisability of mixing drugs should be obtained from a pharmacist.
More than one antibiotic may be prescribed. A few antibiotics are compatible when mixed, but others must be inhaled separately. Either normal saline or sterile water is used to reconstitute a powdered antibiotic or to make a prescribed solution up to the necessary volume for nebulization. Information on the advisability of mixing drugs should be obtained from a pharmacist. Hypertonic saline (3–7%) has been shown to assist in the clearance of secretions (Elkins et al 2006b, Eng et al 1996). It may cause an increase in airflow obstruction (Schoeffel et al 1981) and a bronchoconstriction test should be undertaken at the time of the first inhalation.
Hypertonic saline (3–7%) has been shown to assist in the clearance of secretions (Elkins et al 2006b, Eng et al 1996). It may cause an increase in airflow obstruction (Schoeffel et al 1981) and a bronchoconstriction test should be undertaken at the time of the first inhalation. Sputum induction, commonly using a hypertonic solution of saline and an ultrasonic nebulizer, is a technique that can be used to obtain sputum from people who do not produce it spontaneously. A Task Force set up by the European Respiratory Society details a standardized methodology of sputum induction and processing (European Respiratory Society Task Force 2002). Airway clearance techniques have often been used in conjunction with the inhalation, but there is no evidence to support any increase in the volume of sputum expectorated or quality of the sample obtained when these techniques are included (Elkins et al 2005).
Sputum induction, commonly using a hypertonic solution of saline and an ultrasonic nebulizer, is a technique that can be used to obtain sputum from people who do not produce it spontaneously. A Task Force set up by the European Respiratory Society details a standardized methodology of sputum induction and processing (European Respiratory Society Task Force 2002). Airway clearance techniques have often been used in conjunction with the inhalation, but there is no evidence to support any increase in the volume of sputum expectorated or quality of the sample obtained when these techniques are included (Elkins et al 2005). The mucolytic rhDNase should not be mixed with any other inhaled medication and it is recommended that where possible a separate nebulizing chamber should be used. Ultrasonic nebulizers are not recommended to deliver rhDNase (Suri et al 2002) as the drug composition may be denatured by this technology.
The mucolytic rhDNase should not be mixed with any other inhaled medication and it is recommended that where possible a separate nebulizing chamber should be used. Ultrasonic nebulizers are not recommended to deliver rhDNase (Suri et al 2002) as the drug composition may be denatured by this technology. The inhaled mucolytic acetylcysteine should be used with caution. A reduction in sputum viscosity does not necessarily produce an increase in expectoration of sputum and bronchospasm may be induced. A bronchoconstriction test should be undertaken at the time of the first inhalation. Acetylcysteine is inactivated by oxygen and, if nebulized, the driving gas should be air (Reynolds 1996).
The inhaled mucolytic acetylcysteine should be used with caution. A reduction in sputum viscosity does not necessarily produce an increase in expectoration of sputum and bronchospasm may be induced. A bronchoconstriction test should be undertaken at the time of the first inhalation. Acetylcysteine is inactivated by oxygen and, if nebulized, the driving gas should be air (Reynolds 1996).Domiciliary nebulization
If there is an indication for domiciliary nebulization, it is essential that the appropriate equipment is selected and careful instructions provided (both verbal and written). Instructions in the care and cleaning of the equipment must be given in accordance with local infection control policies.
A spare jet nebulizer and an inlet filter for the air compressor (if necessary) should be available. The nebulizer must be washed and dried thoroughly after each treatment to reduce the possibility of bacterial infection (Hutchinson et al 1996) and to keep the jets clear. The transducer of an ultrasonic nebulizer should be cleaned regularly with acetic acid (white vinegar) to maintain its efficiency. An annual check of output and general and electrical safety should be undertaken and there should be provision for servicing as required (Dodd et al 1995).
Some patients may benefit from a compressor that can be used when travelling, either by using a 12-volt adaptor in a socket in the car, ‘crocodile clips’ fitted on to a battery or, more conveniently, a compact battery pack supplied with the compressor. Those travelling to a country using a different voltage may require a transformer or a dual-voltage compressor. Some compressors incorporate a universal power pack that adapts to voltages through-out the world. An international travel plug adaptor is an accessory required for all who travel abroad. A foot pump may be useful to power a nebulizer where no electricity is available, but it requires considerable energy to operate. When travelling abroad, it is advisable to take a letter from a doctor explaining the need to travel with drugs and possibly syringes and needles.
Bronchodilator and bronchoconstriction testing
Bronchodilator testing
‘The efficacy of bronchodilator therapy should not be assessed by lung function alone but should include a variety of other measures such as improvement in symptoms, activities of daily living, exercise capacity, and rapidity of symptom relief’
Short-term responses may not reflect long-term responses and there is known variability (National Institute for Clinical Excellence 2004).
A peak flow meter (Miller 2000) is often used to assess bronchodilator response. This may be suitable in a patient with asthma, but will not detect the more subtle response of a change in forced vital capacity (FVC), which can occur in those with more irreversible airflow obstruction (Fig. 5.52). For these patients the response to a bronchodilator, detected by a change in FVC, may lead to an increase in exercise ability and improved quality of life. The unnecessary use of nebulized bronchodilators can restrict activities of daily living. A nebulizer and air compressor system should be prescribed only when a more simple delivery device, used correctly, is ineffective.
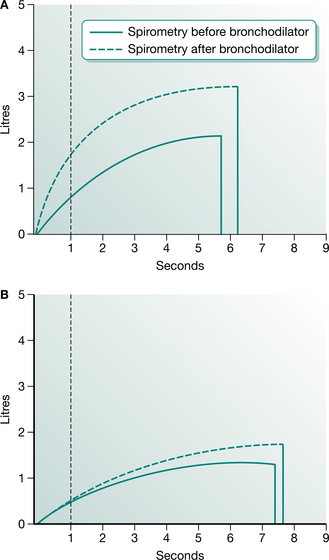
Figure 5.52 (A) Increase in FEV1 and FVC. (B) Increase in FVC only. —, Spirometry before bronchodilator; ---- spirometry after bronchodilator.
The Thoracic Society of Australia and New Zealand and The Australian Lung Foundation (McKenzie et al 2003) have outlined a bronchodilator response test:
Short-acting bronchodilators should be withheld for the previous 6 hours, long acting β-agonists for 12 hours and sustained release theophyllines for 24 hours. Baseline spirometry should be measured and repeated 15–30 minutes following administration of medication. An increase in FEV1 of >12% and 200 ml is greater than the average day-to-day variability and unlikely to occur by chance.
O’Driscoll et al (1990), in their study on home nebulizers, could not demonstrate a correlation between formal lung function testing and the domiciliary use of a nebulizer system. They recommended that patients who are referred for consideration of home nebulizer therapy be given the equipment to try under supervision for several weeks at home and that the patient’s subjective assessment should be considered. In assessing bronchodilator treatment Vora et al (1995) concluded that a quality of life measure and shuttle walking test appeared to be sensitive outcome measures. ‘Clinical thresholds’ using health-related quality of life instruments, for example the Chronic Respiratory Questionnaire, St George’s Respiratory Questionnaire and Asthma Quality of Life Questionnaire, have been identified and may be useful in assessing responsiveness to bronchodilator therapy (Jones 2002). Demonstrating a positive response to a simple inhalation device in a clinical setting and showing that nebulized therapy provides no further benefit is often sufficient to reassure a patient who is anxious that sufficient benefit may not be achieved from an inhaler compared with a compressor system.
Many patients with asthma keep a diary card at home, which will include recordings of peak expiratory flow before and after bronchodilator drugs. These will be valid only if the technique of performing a peak flow manoeuvre through the flow meter (Fig. 5.53) is correct. In terms of technique, it is important to emphasize the following points:
 A maximal inspiration is essential (in their haste to perform the test, the patient may not take a full deep breath)
A maximal inspiration is essential (in their haste to perform the test, the patient may not take a full deep breath)Bronchoconstriction testing
If a patient is to be started on inhaled antibiotics, pentamidine and/or hypertonic saline, an initial test dose should be given. Nebulized antibiotics may be isotonic, hypo- or hypertonic solutions and may cause airflow obstruction (Cunningham et al 2001, Dodd et al 1997). The first dose of a nebulized antibiotic should be monitored by recording the FEV1 and FVC before, immediately after, 15 minutes after and, if evidence of airflow obstruction persists, 30 minutes after the inhalation (Maddison et al 1994).
Individual patients respond differently and this response may vary with different drugs and over time. If airflow obstruction occurs, it can usually be controlled by the inhalation of a bronchodilator taken before physiotherapy for the clearance of secretions, preceding the inhalation of the antibiotic.
Heliox
A mixture of helium and oxygen (80 : 20 or 70 : 30) is sometimes used on a temporary basis to relieve respiratory distress in patients with upper airways obstruction, for example a tumour causing partial obstruction of the trachea. Helium is of lower density than air and may pass more easily through a narrowed airway, requiring less effort from the patient (Vater et al 1983). A side-effect of heliox is an alteration in the pitch of the voice, due to its effect on the vocal cords. This is only temporary, but should be explained to the patient before use of heliox to avoid unnecessary concern. Heliox has not been shown to be of benefit in patients with an exacerbation of chronic obstructive pulmonary disease (Rodrigo et al 2001) or in the treatment of acute asthma (Rodrigo et al 2006).
Humidification
A device to provide humidification of the airways may be considered if either the normal means of humidifying the airways or the mucociliary escalator is not functioning effectively. Inspired gas is adequately humidified in the normal situation but additional humidification may be required (Bersten 2003) during an episode of respiratory infection, in those with existing respiratory disease and in the acute situation. Humidification of inspired gases may be indicated to assist clearance of secretions when the clearance mechanism is not optimally effective or when the normal heat and moisture exchange system of the upper airways is bypassed by an endotracheal or tracheostomy tube. Patients with a long-term tracheostomy may develop metaplasia of the tracheal epithelium.
Ambient air is normally about 22°C with a relative humidity of 50%. Alveolar air usually presents a temperature of 37°C with a relative humidity of 100% (Chiumello et al 2002). During inspiration the inspired gases are progressively heated and humidified along the nose and upper airways, normally reaching the isothermic saturation boundary (ISB) of 37°C and 100% relative humidity at about 5–6 cm below the carina (Chiumello et al 2002). The volume, temperature and absolute humidity of the inspired gases can change the point of the ISB.
The epithelial lining of the airways, from the trachea to the respiratory bronchioles, contains ciliated cells that are responsible for moving mucus and particulate matter proximally to the level of the larynx. The optimal temperature for cilial activity is normal body temperature with reduced activity occurring below 20°C and above 40°C (Wanner 1977). The cilia beat within a watery fluid, the ‘periciliary’ or ‘sol’ layer. A mucus layer ‘gel’ covers the periciliary layer and interacts with the tips of the cilia.
The efficiency of mucus transport is dependent on correctly functioning cilia and the composition of the periciliary and mucus layers. If the periciliary layer becomes too shallow, as with dehydration, the cilia become enmeshed in the viscous mucus layer and cannot function effectively. If the periciliary layer is too deep, the tips of the cilia are not in contact with the mucus layer and propulsion of the mucus is inefficient.
The viscosity of mucus is increased during bacterial infection owing to an increase in the DNA content of the mucus (Wilson & Cole 1988). With hypersecretory disorders of, for example, bronchiectasis and chronic airflow limitation, there is an increase in both quantity and viscosity of mucus secretions. Bacteria directly affect cilial beating and coordination, disrupt the epithelium, stimulate mucus secretion and alter periciliary fluid composition (Cole 1995). Humidification has been shown to enhance tracheobronchial clearance when used as an adjunct to physiotherapy in a group of patients with bronchiectasis (Conway et al 1992).
Clarke (1995) has suggested that the efficiency of cough increases with a decrease in viscosity of mucus and an increase in the periciliary layer of the airway. Conway (1992) hypothesized that humidification by water or saline aerosol produces an increase in depth of the periciliary and mucus layers, thereby decreasing viscosity and enhancing the shearing of secretions by huffing or coughing.
Methods of humidification
Systemic hydration
Adequate humidification may be obtained by increasing the oral or intravenous fluid intake of a patient. Breathless patients find drinking fluids an effort, but need encouragement to avoid dehydration. Patients should be reminded to maintain an adequate fluid intake as this may help to prevent airway secretions from becoming more tenacious. During periods of infection and fever a higher fluid intake is required.
Heated water bath humidifiers
Gas is blown over a reservoir of heated sterile water and absorbs water vapour, which is then inhaled by the patient (Fig. 5.54). If the delivery tube is cold there is a temperature drop as the gas passes along the tube and condensation occurs. The humidifier should be positioned below the level of the patient’s airway to avoid flooding of the airway by condensed water. Sealed water traps (to prevent contamination) should be included in the circuit to allow regular emptying without interrupting ventilation. A heated delivery tube eliminates the problem of condensation and allows the gas to be delivered at a desired temperature of 32–36°C with a water content of 33–43 g/m3 (Hinds & Watson 1996). Sterile water must be used in these devices. If saline is used, only the water vaporizes and the sodium chloride crystallizes out.
Humidifiers can be used for the spontaneously breathing patient or can be incorporated into ventilator circuits including continuous positive airway pressure and non-invasive ventilation. If using a low output humidifier, it may be necessary to use two devices in series in order to achieve adequate humidification (Harrison et al 1993).
Heat and moisture exchangers
A heat and moisture exchanger (HME) or the ‘Swedish nose’ is a lightweight disposable device and may be used in the intubated patient either mechanically ventilated or breathing spontaneously. In patients receiving pressure support ventilation (Pelosi et al 1996) and in spontaneously breathing patients, it is important to be aware of the slight resistance that may increase the work of breathing. The HME acts in a similar way to the nasopharynx. The heat and moisture of the exhaled gas are retained either by condensation (condenser humidifier) or by absorption and returned in the inhaled gas as it passes through the device. A variety of hygroscopic materials and chemicals are used for absorption within heat and moisture exchangers.
HMEs are inefficient if there is a large air leak around an uncuffed tracheostomy tube (Tilling & Hayes 1987) and generally do not provide adequate humidification for infants. In children with an increased work of breathing, the additional resistance from a heat and moisture exchanger may further compromise breathing. If the secretions of a patient using an HME become tenacious, a more effective form of humidification will be required (Bransen et al 1993). The HME should be changed every 24 (Boots et al 2006) to 48 hours (Djedaini et al 1995) and immediately if it becomes soiled with secretions.
Large-volume jet nebulizers
A large-volume jet nebulizer which uses the same principle as a small-volume jet nebulizer (used for inhalation of medication) may be used for humidification of inspired gas and may include a heater within the circuit. However, the droplets can carry bacteria, thus increasing the risk of respiratory infection (Bersten 2003).
Ultrasonic
Many ultrasonic humidifiers (Fig. 5.55) do not have a heater, but the mist is at ambient temperature and warmer than that produced from a jet nebulizer powered from compressed piped gas. There is often an airflow control valve in addition to a control for the density of the mist. By regulating these two controls the density of mist can be adjusted to the patient’s comfort.
Sterile normal saline (0.9%) is isotonic and probably the most appropriate solution, although sterile water can be used if saline is unavailable, but has been shown to cause bronchoconstriction in patients with hyperreactive airways (Schoeffel et al 1981).
Bubble-through humidifiers
A device containing cold water, through which the inspired gas is bubbled, has not been shown to be an effective means of humidification. No objective benefits were identified when a bubble-through humidifier was attached to nasal cannulae (Campbell et al 1988). Many patients do, however, report subjective benefits from these devices. When bubble-through humidifiers are connected to nasal cannulae or an oxygen mask with narrow-bore tubing, care must be taken to ensure that water does not condense in the tubing, as this may alter the inspired oxygen concentration by partially blocking the passage of gas through the tubing. A bubble-through humidifier attached to the narrow-bore tubing of a Venturi mask is inappropriate, as the flow of oxygen is likely to be reduced by the back pressure from the humidifier device and by condensation blocking the narrow-bore tubing.
Delivery to the patient
Patients with retained secretions postoperatively or those with excess viscous secretions due to a chronic bronchopulmonary infection may benefit from a period of 10–20 minutes’ humidification before physiotherapy to assist the clearance of secretions. If the concentration of oxygen required by the patient is not critical, a mouthpiece with a hole for entrainment of additional air is simple and comfortable to use. Deep breathing, interspersed with tidal volume breathing, may encourage peripheral deposition of an aerosol, while avoiding hyperventilation.
Patients requiring an inspired oxygen concentration of 24% will probably wear a Venturi mask connected to the oxygen by narrow-bore tubing. It is impossible to give high humidification through a narrow-bore tube owing to condensation within the tubing. Effective humidification can be obtained by using a humidity adaptor, which allows the air entrained by the mask to be humidified. It is a cuff fitted over the air-entraining holes of the mask and connected by wide-bore humidity tubing to a humidifier powered by an air source. A humidity adaptor can be used to give humidification to Venturi masks delivering accurate higher concentrations of oxygen (e.g. 28% or 35%), but is unsatisfactory with a 60% Venturi mask because the air-entraining holes are too small to entrain the humidity. An ultrasonic nebulizer, set at a high flow, can be used and is quieter than a jet nebulizer system. If a patient is breathing spontaneously through a tracheostomy, a tracheostomy mask may be attached to a humidifier or nebulizer via wide-bore tubing. Alternatively a heat moisture exchanger or a ‘laryngectomy-permanent tracheostomy protector’ (tracheostomy ‘bib’) may be used.
Humidification through a head box (Chapter 10) is often used in the treatment of spontaneously breathing infants. With the narrow airways of an infant the risk of mucus plugging is higher than in adults. Humidity to a head box may be either from a heated water bath humidifier or a heated nebulizer system.
Hazards of humidification
Inhalation of cold mist or water (a hypotonic solution) may cause bronchoconstriction in patients with hyperreactive airways (Schoeffel et al 1981). Heated humidification and normal saline solution are less likely to cause this problem. It may be appropriate to take peak flow or spirometry recordings before and immediately after the first treatment.
Water reservoirs had been considered good culture mediums for bacteria, but there is little evidence to support humidification as an important factor in nosocomial respiratory tract infections (Bersten 2003). A particularly vulnerable site is the catheter mount of the ventilator circuit in the intubated patient. Regular disposal, disinfection or sterilization of all equipment is essential to prevent infection and local infection control policies must be observed.