DEVELOPMENT OF TEETH
Initiation of Tooth Development
Development of the Dental Papilla
Development of Supporting Structures
After reading this chapter the student will be able to:
• describe the origin of the tooth formative cells and the role of induction in tooth formation
• describe the stages of tooth formation and the mineralization of enamel and dentin
• describe the development of the tissues that surround the developing teeth
OVERVIEW
In the human, 20 primary and 32 permanent teeth develop from the interaction of the oral epithelial cells and the underlying mesenchymal cells. Each developing tooth grows as an anatomically distinct unit, but the basic developmental process is similar for all teeth.
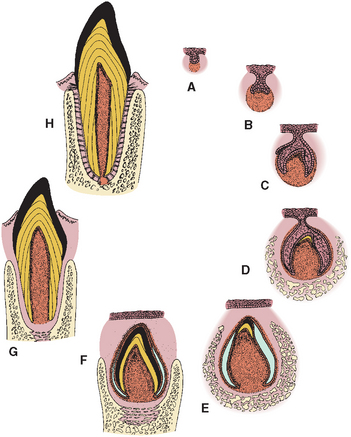
Each tooth develops through successive bud, cap, and bell stages (Fig. 5-1, A to C). During these early stages, the tooth germs grow and expand, and the cells that are to form the hard tissues of the teeth differentiate. Differentiation takes place in the bell stage, setting the stage for enamel and dentin formation (Fig. 5-1, D and E). As the crowns are formed and mineralized, the roots of the teeth begin to form. After the roots calcify, the supporting tissues of the teeth—the cementum, periodontal ligament, and alveolar bone—begin to develop (Fig. 5-1, F and G). This formation occurs whether the tooth is an incisor with a single root, a premolar with several roots, or a molar with multiple roots. Subsequently, the completed tooth crown erupts into the oral cavity (Fig. 5-1, G). Root formation and cementogenesis continue until a functional tooth and its supporting structures are fully developed (Fig. 5-1, G and H).
INITIATION OF TOOTH DEVELOPMENT
Teeth develop from two types of cells: oral epithelial cells form the enamel organ and mesenchymal cells form the dental papilla. Enamel develops from the enamel organ, and dentin forms from the dental papilla. The interaction of these epithelial and mesenchymal cells is vital to the initiation and formation of the teeth. In addition to these cells, the neural crest cells contribute to tooth development. The neural crest cells arise from the neural tissue at an early stage of development and migrate into the jaws, intermingling with mesenchymal cells. They function by integrating with the dental papillae and epithelial cells of the early enamel organ, which aids in the development of the teeth. The cells also function in the development of the salivary glands, bone, cartilage, nerves, and muscles of the face. Chapter 1 discusses neural crest cells and explains the cell’s migration (see Fig. 1-17).
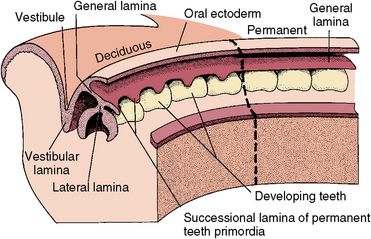
The first sign of tooth formation is the development of dental lamina rising from the oral epithelium. Dental lamina develops into a sheet of epithelial cells that pushes into the underlying mesenchyme around the perimeter of both the maxillary and mandibular jaws (Fig. 5-2). At the leading edge of the lamina, 20 areas of enlargement appear, which form tooth buds for the 20 primary teeth (see Fig. 5-2). At this early stage, the tooth buds have already determined their crown morphology of an incisor or molar. This is the result of gene expression. After primary teeth develop from the buds, the leading edge of the lamina continues to grow to develop the permanent teeth, which succeed the 20 primary teeth. This part of the lamina is thus called the successional lamina (Fig. 5-3). The lamina continues posteriorly into the elongating jaw and from it come the posterior teeth, which form behind the primary teeth. In this manner, 20 of the permanent teeth replace the 20 primary teeth, and 12 posterior permanent molars develop behind the primary dentition (see Fig. 5-3). The last teeth to develop are the third molars, which develop about 15 years after birth. Because the molars do not succeed the primary teeth, they do not form from the successional lamina but from the general lamina. The initiating dental lamina that forms both the successional and general lamina begins to function in the sixth prenatal week and continues to function until the fifteenth year, producing all 52 teeth. In general, the teeth develop anteroposteriorly, which relates to the growing jaws. The posterior molars do not develop until space is available for them in the posterior jaw area. The second dentition does not develop until after the primary teeth are formed and functioning. Gradually the permanent teeth form under the primary crowns and later posteriorly to the primary molars.
STAGES OF TOOTH DEVELOPMENT
Although tooth formation is a continuous process, it is characterized by a series of easily distinguishable stages known as the bud, cap, and bell stages. Each stage is defined according to the shape of the epithelial enamel organ, which is a part of the developing tooth. The initial stage, the bud stage, is a rounded, localized growth of epithelial cells surrounded by proliferating mesenchymal cells (Fig. 5-4). Gradually, as the rounded epithelial bud enlarges, it gains a concave surface, which begins the cap stage (Fig. 5-5). The epithelial cells now become the enamel organ and remain attached to the lamina. The mesenchyme forms the dental papilla, which becomes the dental pulp. The tissue surrounding these two structures is the dental follicle.
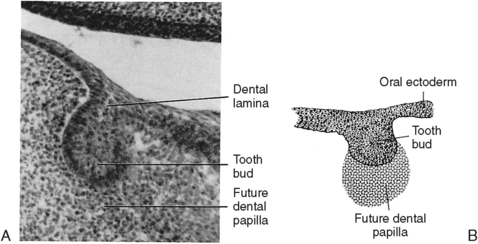
Fig. 5-4 Initiation of tooth development. A, Histology of the bud stage. B, Diagram of the bud stage.
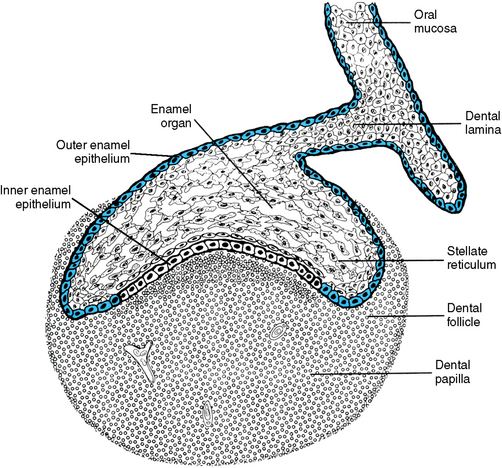
Fig. 5-5 Cap stage of tooth development. The enamel organ is outlined in blue, indicating that it is of ectodermal origin. The mesenchyme of the dental papilla surrounds the enamel organ.
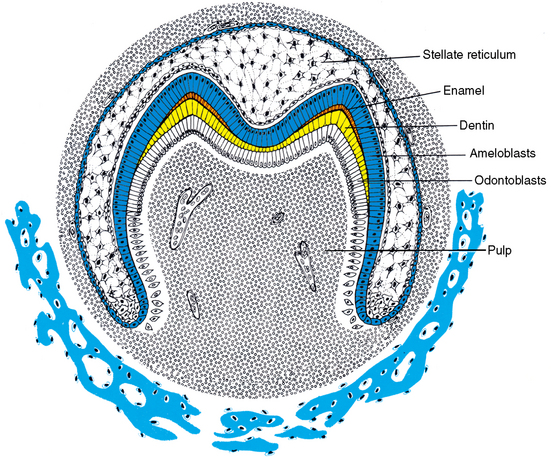
After further growth of the papilla and the enamel organ, the tooth reaches the morphodifferentiation and histodifferentiation stage, also known as the bell stage (Fig. 5-6). At this stage, the inner enamel epithelial cells are characterized by the shape of the tooth they form (see Fig. 5-6). Also, the cells of the enamel organ have differentiated into the outer enamel epithelial cells, which cover the enamel organ, and inner enamel epithelial cells, which become the ameloblasts that form the enamel of the tooth crown. Between these two cell layers are the stellate reticulum cells, which are star shaped with processes attached to each other. A fourth layer in the enamel organ is composed of stratum intermedium cells. These cells lie adjacent to the inner enamel epithelial cells. They assist the ameloblast in the formation of enamel. The function of the outer enamel epithelial cells is to organize a network of capillaries that will bring nutrition to the ameloblasts.
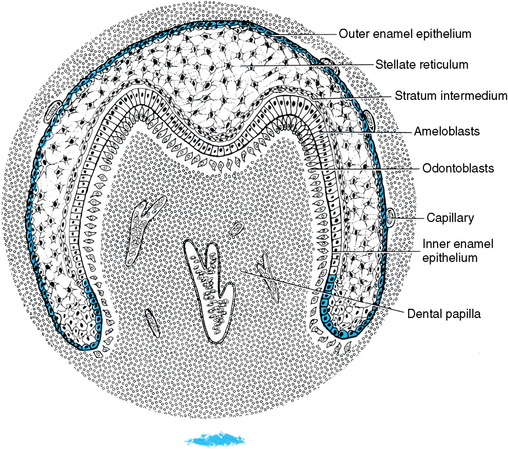
Fig. 5-6 Bell stage of tooth development. During this stage of tooth development, both odontoblasts and ameloblasts have fully differentiated in the cuspal region(s). Blood vessels develop in the dental papilla, whereas the only vascularized structure in the enamel organ is the outer enamel epithelium that contains a capillary plexus.
From the outer enamel epithelium, nutrients will percolate through the stellate reticulum to the ameloblasts. During the bell stage, cells in the periphery of the dental papilla become odontoblasts. These cells differentiate from mesenchymal cells. As the odontoblasts elongate and become columnar, they form a matrix of collagen fibers identified as predentin. After 24 hours, this increment of matrix calcifies and becomes dentin. When several increments of dentin have formed, the differentiated ameloblasts deposit an enamel matrix. Dentinogenesis always precedes amelogenesis. After the enamel organ is differentiated, the dental lamina begins to degenerate by undergoing lysis. The dental lamina disappears in the anterior part of the mouth, although it remains active in the posterior region for many years (see Figs. 5-2 and 5-3).
Cells interact through a system of effectors, modulators, and receptors called cell signaling. An example of such a system is epithelial-mesenchymal interaction in tooth development. The precursor cells, odontoblast and ameloblast, establish a positional relationship by means of effectors and receptors that are on the cell surface. The ameloblast differentiates first, causing the precursor odontoblast to locate itself adjacent. Then the odontoblast differentiates, establishing with the ameloblast a basement membrane that then forms a dentinal matrix. After this formation occurs, the ameloblast forms the enamel matrix. Thus it is not only cells, but also basal lamina and dentin matrix, that contain substances that cause cell changes and position.
DEVELOPMENT OF THE DENTAL PAPILLA
Densely packed cells characterize the dental papilla. This is evident even in the early bud stage, during which cells proliferate around the enlarging tooth buds at the leading edge of the dental lamina (Fig. 5-7). The papilla cells are believed to be significant in furthering enamel organ bud formation into the cap and bell stage. This cell density is maintained as the enamel organ grows. Cells of the dental papilla are found on close examination to be fibroblasts and appear to be in a delicate reticulum (Fig. 5-8). Blood vessels appear early in the dental papilla, initially in the central region along with nerve fibers associated with these vessels. The vessels bring nutrition to the rapidly growing organ. As the papilla grows, smaller vessels are also seen in the periphery of the area, bringing nutrition to the elongating odontoblasts (see Fig. 5-6). Cellular changes result in formation of a hard shell around the central papilla. As this occurs, the papilla becomes known as the dental pulp.
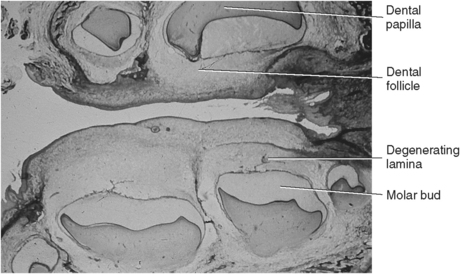
Fig. 5-7 Histology of tooth development. Sagittal view of the human maxillary and mandibular molar tooth buds.
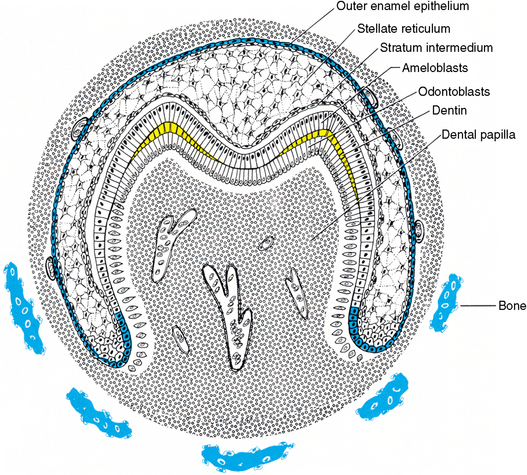
Fig. 5-8 Dentinogenesis stage of tooth development. The initial formation of dentin (yellow) at the cuspal tips and the vascularized pulp organ are characteristic of the dentinogenesis stage. The dental follicular cells are differentiating around the enamel organ and alveolar bone proper is beginning to define the dental crypt.
DENTINOGENESIS
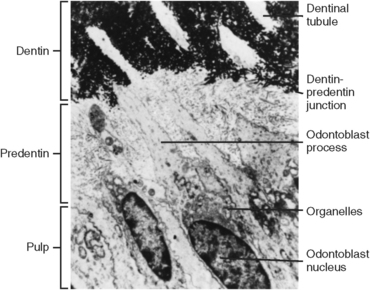
As the odontoblasts elongate, they gain the appearance of a protein-producing cell. A process develops at the proximal end of the cell, adjacent to the dentinoenamel junction. Gradually the cell moves pulpward, and the cell process, known as the odontoblast process, elongates (Fig. 5-9). The odontoblast becomes active in dentinal matrix formation similar to an osteoblast when it moves away from a spicule of bone. Increments of dentin are formed along the dentinoenamel junction. The dentinal matrix is first a meshwork of collagen fibers, but within 24 hours it becomes calcified. It is called predentin before calcification and dentin after calcification. At that time, the dental papilla becomes the dental pulp as dentin begins to surround it. The odontoblasts maintain their elongating processes in dentinal tubules (see Fig. 5-9).
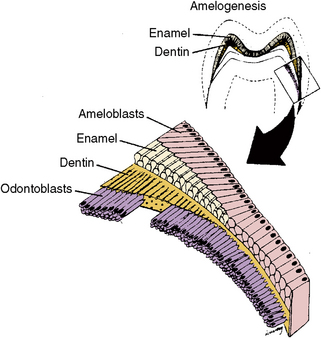
Fig. 5-9 Appositional stage of tooth development. During this stage of tooth development, both enamel and dentin are actively secreted until the crown is complete.
When the odontoblasts are functioning, their nuclei occupy a more basal position in the cell, and the organelles become more evident in the cell cytoplasm. The appearance of granular endoplasmic reticulum, Golgi’s complex, and mitochondria indicates the protein-producing nature of these cells (Fig. 5-10, C to E). The odontoblasts then secrete protein externally via vesicles at the apical part of the cell and along the cell processes (see Fig. 5-10). The collagenous dentinal matrix is laid down in increments like bone or enamel, which is indicative of a daily rhythm for hard tissue formation. The site of initial formation is at the cusp tips (see Fig. 5-8), and as further increments are formed, more odontoblasts are activated along the dentinoenamel junction (see Fig. 5-9). As the odontoblastic process elongates, a tubule is maintained in the dentin, and the matrix is formed around this tubule (Fig. 5-10, C and D).
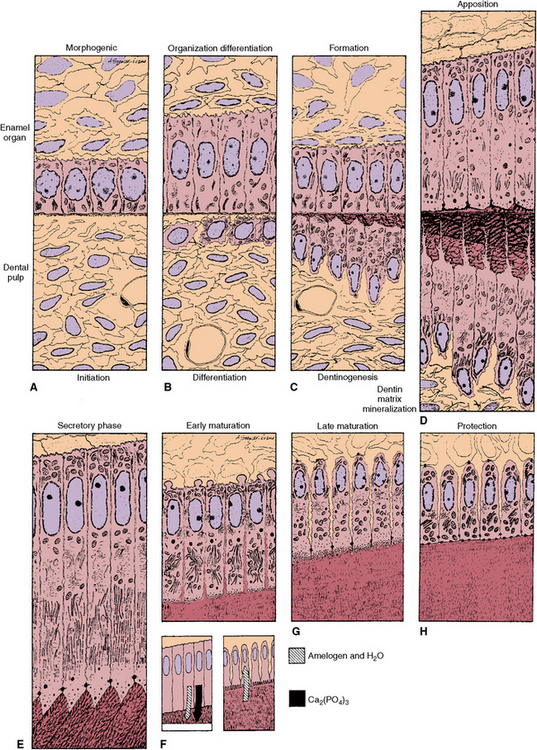
Fig. 5-10 Diagram of enamel and dentin formation. A, Initiation. B, Differentiation. C, Dentinogenesis. D, Apposition of enamel and dentin. E to H, Stages of enamel formation. E, Secretory stage of enamel formation. F, Early maturation. G, Late maturation. H, Protective stage in which the ameloblasts secrete the developmental cuticle. During maturation of enamel, an influx of mineral is accompanied by a loss of organic matter and water from the enamel matrix.
Dentinogenesis takes place in two phases. First is the collagen matrix formation, followed by the deposition of calcium phosphate (hydroxyapatite) crystals in the matrix. The initial calcification appears as crystals that are in small vesicles on the surface and within the collagen fibers (Fig. 5-11). The crystals grow, spread, and coalesce until the matrix is completely calcified. Only the newly formed band of dentinal matrix along the pulpal border is uncalcified (Fig. 5-12). Matrix formation and mineralization therefore are closely related. Mineralization proceeds by an increase in mineral density of the dentin. As each daily increment of predentin forms along the pulpal boundary, the adjacent peripheral increment of predentin formed the previous day calcifies and becomes dentin (see Figs. 5-10 and 5-12; Fig. 5-13).
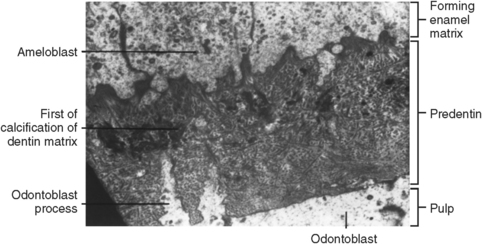
Fig. 5-11 Initiation of dentinogenesis. A transmission electron micrograph of a band of predentin, dentin, and enamel at the dentinoenamel junction. This initially secreted dentin is mantle dentin and is significantly different from the dentin that is formed by incremental deposition later in development. Calcification of the predentin will spread from nucleation sites within the matrix vesicles.
AMELOGENESIS
Ameloblasts begin enamel deposition after a few micrometers of dentin have been deposited at the dentinoenamel junction (Fig. 5-14). At the bell stage, cells of the inner enamel epithelium differentiate. They elongate and are ready to become active secretory ameloblasts. The ameloblasts then exhibit changes as they differentiate and pass through five functional stages: (1) morphogenesis, (2) organization and differentiation, (3) secretion, (4) maturation, and (5) protection (see Fig. 5-10). Golgi’s apparatus appears centrally in the ameloblasts, and the amount of rough endoplasmic reticulum (RER) increases in the apical area (see Fig. 5-10, D and E). The row of ameloblasts maintains orientation by cell-to-cell attachments (desmosomes) at both the proximal and distal ends of the cell. This maintains the cells in a row as they move peripherally from the dentinoenamel junction depositing enamel matrix (see Fig. 5-9).
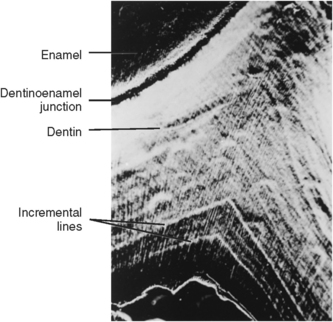
Fig. 5-14 Microradiograph of dentin illustrating incremental lines in dentin showing the deposition of dentin in increments.
Short conical processes (Tomes’ processes) develop at the apical end of the ameloblasts during the secretory stage (see Fig. 5-10, E; Fig. 5-15). Junctional complexes called the terminal bar apparatus appear at the junction of the cell bodies and Tomes’ processes and maintain contact between adjacent cells (see Fig. 5-10, E). As the ameloblast differentiates, the matrix is synthesized within the RER, which then migrates to Golgi’s apparatus, where it is condensed and packaged in membrane-bound granules. Vesicles migrate to the apical end of the cell, where their contents are exteriorized and are deposited first along the junction of the enamel and dentin (Fig. 5-16). This first enamel deposited on the surface of the dentin establishes the dentinoenamel junction. Figure 5-17 is an electron micrograph of young enamel matrix formed along the dentinoenamel junction. The Tomes’ process of the ameloblast indents the surface of the enamel (see Figs. 5-10, E, 5-15, and 5-16). This is because the center of the rod does not form at the same rate as the rod walls; this can best be seen in Figure 5-17. As the enamel matrix develops, it forms in continuous rods from the dentinoenamel junction to the surface of the enamel.
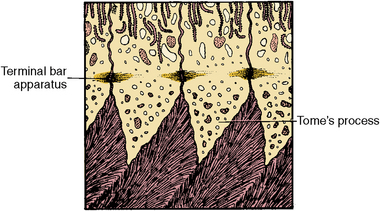
Fig. 5-15 Diagram of Tome’s process, the specialized secretory process of the ameloblast during enamel formation.
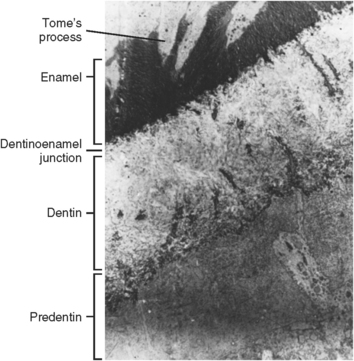
Fig. 5-16 Ultrastructure of the dentoenamel junction showing early enamel and dentin matrix formation.
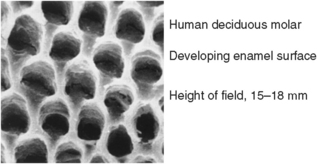
Fig. 5-17 Scanning electron micrograph showing interface between ameloblast and enamel matrix during amelogenesis. Pits are result of presence of Tome’s process.
When ameloblasts begin secretion, the overlying cells of the stratum intermedium change in shape from spindle to pyramidal (see Fig. 5-10, B to F). As amelogenesis proceeds, both of these cell layers, ameloblasts and stratum intermedium, are held together by cell junctional complexes termed desmosomes, with synthesis of enamel occurring in both cells. Substances needed for enamel production arrive via the blood vessels and pass through the stellate reticulum to the stratum intermedium and ameloblasts. In this manner, the protein amelogenin is produced. Only a few ameloblasts at the tip of the cusps begin to function initially (see Fig. 5-13). As the process proceeds, more ameloblasts become active, and the increments of enamel matrix become more prominent.
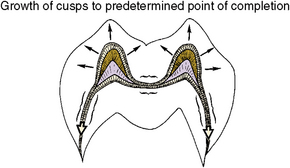
Growth of individual cusps by incremental deposition continues until tooth eruption. This occurs in posterior multicuspid teeth as the ameloblasts continue to differentiate from the inner enamel epithelium and form enamel. Cusps then coalesce in the intercuspal region of the crown (Fig. 5-18). In radiographs, cusps initially appear separated and are joined together as growth progresses. The inner enamel epithelium forms the blueprint for the shape of the developing crown.
CROWN MATURATION
As amelogenesis is completed and amelogenin is deposited, the matrix begins to mineralize (see Fig. 5-10, F to H). As soon as the small crystals of mineral are deposited, they begin to grow in length and diameter. The initial deposition of mineral amounts to approximately 25% of the total enamel. The other 70% of mineral in enamel is a result of growth of the crystals (5% of enamel is water). The time between enamel matrix deposition and its mineralization is short. Therefore the pattern of mineralization closely follows the pattern of matrix deposition. The first matrix deposited is the first enamel mineralized, occurring along the dentinoenamel junction. Matrix formation and mineralization continue peripherally to the tips of the cusps and then laterally on the sides of the crowns, following the enamel incremental deposition pattern (Fig. 5-19). Finally, the cervical region of the crown mineralizes. During this process, protein of the enamel changes or matures and is termed enamelin.
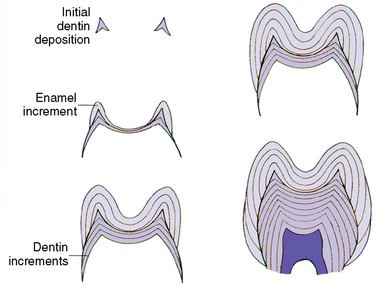
Fig. 5-19 Incremental pattern of enamel and dentin formation from initiation to completion. Development proceeds from upper left to lower right.
The mineral content of enamel is approximately 95% as it rapidly surpasses that of dentin (69%) to become the most highly calcified tissue in the human body. Because of the high mineral content of enamel, almost all water and organic material are lost from it during maturation (see Fig. 5-10, E to H).
As the ameloblast completes the matrix deposition phase, its terminal bar apparatus disappears, and the surface enamel becomes smooth (see Fig. 5-10, F and G). This phase is signaled by a change in the appearance of the cell as well as by a change in the function of the ameloblast. The apical end of this cell becomes ruffled along the enamel surface. The length of the ameloblast decreases, as does the number of organelles within it. The enamel has now reached the maturation phase, and the ameloblast becomes more active in absorption of the organic matrix and water from enamel, which allows mineralization to proceed (see Fig. 5-10, F to H).
The increased mineral content in enamel is dependent on the loss of fluid and protein. This process of exchange occurs throughout much of enamel maturation and is not limited to the final stage of mineralization. Even after the teeth erupt, mineralization of enamel continues.
Finally, after the ameloblasts have completed their contributions to the mineralization phase, they secrete an organic cuticle on the surface of the enamel, which is known as the developmental or primary cuticle. The ameloblasts then attach themselves to this organic covering of the enamel by hemidesmosomes (see Fig. 5-10, H). A hemidesmosome is half of a desmosome attachment plaque. Whereas a desmosome functions in attaching a cell to an adjacent cell, a hemidesmosome relates to the attachment of a cell to a surface membrane. The hemidesmosome attachment plaque is developed by the ameloblast, and this stage of plaque formation and attachment is known as the protective stage of ameloblast function. The ameloblasts shorten and contact the stratum intermedium and other enamel epithelium, which fuse together to form the reduced enamel epithelium. This cellular organic covering remains on the enamel surface until the tooth erupts into the oral cavity.
With mineralization of enamel complete and its thickness established, the crown of the tooth is formed (Fig. 5-20). The nearly completed crown with the reduced enamel epithelium is seen in Figure 5-21. Meanwhile, dentin formation proceeds. The next stage of development will be root formation.
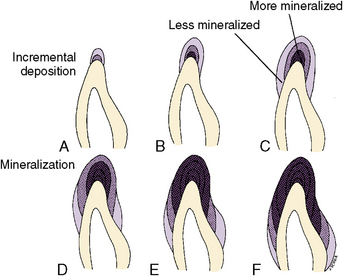
Fig. 5-20 Summary of enamel mineralization stages. A, Initial enamel is formed. B, Initial enamel is calcified as further enamel is formed. C, More increments are formed. D, Matrix deposition and mineralization proceeds. E and F, Matrix is formed on the sides and cervical areas of the crown.
DEVELOPMENT OF THE TOOTH ROOT
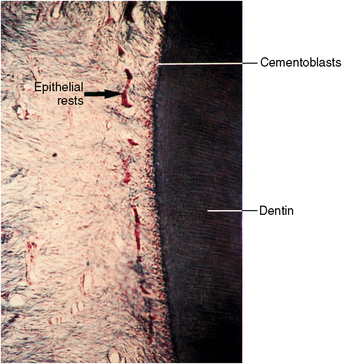
As the crown develops, cell proliferation continues at the cervical region or base of the enamel organ, where the inner and outer enamel epithelial cells join to form a root sheath (Fig. 5-22). When the crown is completed, the cells in this region of the enamel organ continue to grow, forming a double layer of cells termed the epithelial root sheath or Hertwig’s root sheath (Fig. 5-22, A). The inner cell layer of the root sheath forms from the inner enamel epithelium or ameloblasts in the crown, and enamel is produced. In the root, these cells induce odontoblasts of the dental papilla to differentiate and form dentin. The root sheath originates at the point that enamel deposits end. As the root sheath lengthens, it becomes the architect of the root. The length, curvature, thickness, and number of roots are all dependent on the inner root sheath cells. As the formation of the root dentin takes place, cells of the outer root sheath function in the deposition of intermediate cementum, a thin layer of acellular cementum that covers the ends of the dentinal tubule and seals the root surface. Then the outer root sheath cells disperse into small clusters and move away from the root surface as epithelial rests (Fig. 5-22, B). At the proliferating end, the root sheath bends at a near 45-degree angle. This area is termed the epithelial diaphragm (see Fig. 5-22). The epithelial diaphragm encircles the apical opening of the dental pulp during root development. It is the proliferation of these cells that causes root growth to occur.
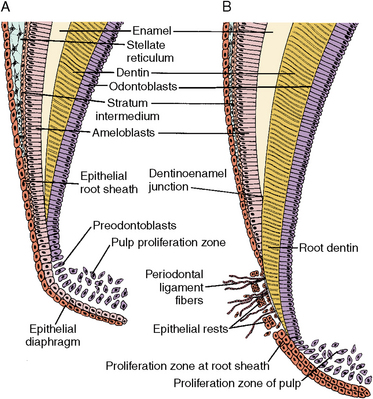
Fig. 5-22 Root formation, showing root sheath and epithelial diaphragm. A, Time of epithelial root sheath formation showing fusion of outer and inner enamel epithelium to form the epithelial root sheath, which includes the vertical epithelial root trunk and inward bending epithelial diaphragm. B, Later stage of root sheath development. Root dentin has formed below the cervical enamel on the surface of the pulp organ. Cementoblasts, periodontal ligament fibers, and epithelial rests are present in the ligament.
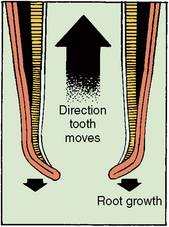
As the odontoblasts differentiate along the pulpal boundary, root dentinogenesis proceeds and the root lengthens. Dentin formation continues from the crown into the root (Fig. 5-23). The dentin tapers from the crown into the root to the apical epithelial diaphragm. In the pulp adjacent to the epithelial diaphragm, cellular proliferation occurs. This is known as the pulp proliferation zone (see Fig. 5-22). It is believed that this area produces new cells needed for root lengthening. Dentinogenesis continues until the appropriate root length is developed. The root then thickens until the apical opening is restricted to approximately 1 to 3 mm, which is sufficient to allow neural and vascular communication between the pulp and the periodontium.
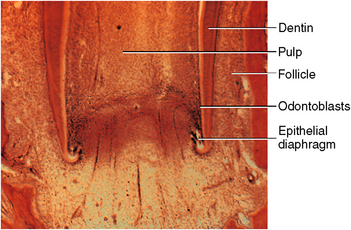
Fig. 5-23 Histology of root formation, showing root sheath and epithelial diaphragm. The highly cellular pulp proliferative zone is shown in the apical pulpal zone.
With the increase in root length, the tooth begins eruptive movements, which provide space for further lengthening of the root. The root lengthens at the same rate as the tooth eruptive movements occur (Fig. 5-24).
Single Root
The root sheath of a single-rooted tooth is a tubelike growth of epithelial cells that originates from the enamel organ, enclosing a tube of dentin and the developing pulp (see Fig. 5-23). As soon as the root sheath cells deposit the intermediate cementum, the root sheath breaks up, forming epithelial rests (see Fig. 5-22, B; Fig. 5-25). The epithelial rests persist as they move away from the root surface into the follicular area. Mesenchymal cells from the tooth follicle move between the epithelial rests to contact the root surface. Here, they differentiate into cementoblasts and begin secretion of cementoid on the surface of the intermediate cementum. Cementoid is noncalcified cementum that soon calcifies into mature cementum (Fig. 5-26). The root sheath is never seen as a continuous structure because its cell layers break down rapidly once the root dentin forms. However, the area of the epithelial diaphragm is maintained until the root is complete; then it disappears.
Multiple Roots
The roots of multirooted teeth develop in a fashion similar to those of single-rooted teeth until the furcation zone begins to form (Fig. 5-27). Division of the roots then takes place through differential growth of the root sheath. The cells of the epithelial diaphragm grow excessively in two or more areas until they contact the opposing epithelial extensions. These extensions fuse, and then the original single opening is divided into two or three openings. The epithelial diaphragm surrounding the opening to each root continues to grow at an equal rate. When a developing molar is sectioned through the center of its root, it shows the root sheath as an island of cells (Fig. 5-28).
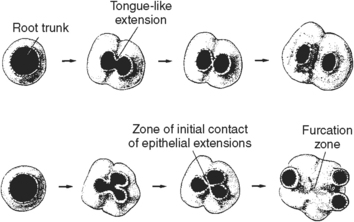
Fig. 5-27 Development of multirooted teeth. As the epithelial diaphragm grows, it may make contact and fuse to develop one-, two-, or three-rooted teeth.
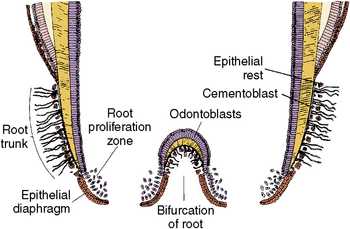
Fig. 5-28 Bifurcation root zone in multiple root formation. The root trunk is the junction area between the crown and the root bifurcation area.
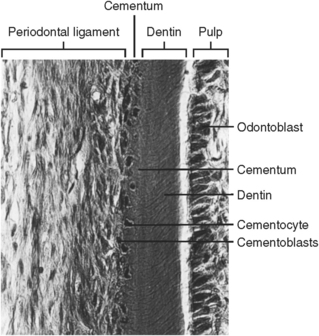
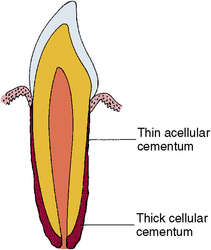
As the multiple roots form, each one develops by the same pattern as a single-rooted tooth. After the root is complete and the sheath breaks up, the epithelial cells migrate away from the root surface as they do in a single-rooted tooth. Cementum then forms on the surface of the intermediate cemental surface. The cementum usually appears cellular, although the cementum near the cementoenamel junction is less cellular than that at the apices of the root (Fig. 5-29). Because the apical cementum is thicker, it is said to require more cells to maintain vitality. The primary function of this cementum involves the attachment of the principal periodontal ligament fibers.
DEVELOPMENT OF PRIMARY AND PERMANENT TEETH
Primary and permanent teeth develop very similarly, although the time needed for development of primary teeth is much less than for the permanent teeth. Primary teeth begin development in utero, and the crown undergoes complete mineralization before birth, whereas the permanent teeth begin formation at or after birth. In Figures 6-1 and 6-2, the formation of the primary and permanent incisors is compared as is the first primary second molar and the permanent premolar. Any prenatal systemic disturbance will affect mineralization of the primary tooth crowns, whereas postnatal disturbances may affect the permanent tooth crowns.
Primary teeth function in the mouth approximately 8.5 years; this period of time may be divided into three periods: crown and root development, root maturation and root resorption, and shedding of the teeth. The first period extends for about a year, the second for about 3.75 years, and the final stage of resorption and shedding lasts for about 3.5 years. In contrast, some of the permanent teeth may be in the mouth from the fifth year until death. One must also consider the permanent molars, which may be in the mouth only from the 25th year on until they are lost or death occurs. The permanent teeth may function seven or eight times as long as the primary teeth. This time of function of permanent teeth includes 12 years of development, 3 years longer than the primary teeth.
Many separate events occur within a few millimeters during development of the dentition. For a single primary tooth and its successor, an example of two possibly simultaneous events could be eruption with root formation of the primary tooth and mineralization of the crown of the permanent tooth. Other examples of complex events during this mixed dentition stage are root resorption of the primary tooth and formation of the root of the permanent tooth. In a 6-year-old child one or more of these formative processes may be occurring in up to 28 of 32 permanent teeth, while some degree of resorption is occurring in the 20 primary teeth. Timing and coordination of myriad events allow continual function within the growing jaws.
In addition to the formative events, the primary teeth undergo root resorption and pulp degeneration.
DEVELOPMENT OF SUPPORTING STRUCTURES
The mesenchymal cells surrounding the teeth are known as the dental follicle (see Fig. 5-7). Some of these follicular cells, which lie immediately adjacent to the enamel organ, migrate during the cap and bell stages from the enamel organ peripherally into the follicle to develop the alveolar bone and the periodontal ligament (Fig. 5-30). These cells have been traced from this origin to the site where they differentiate into osteoblasts and form bone or fibroblasts, which form ligament fibers. After tooth eruption, these tissues serve to support the teeth during function.
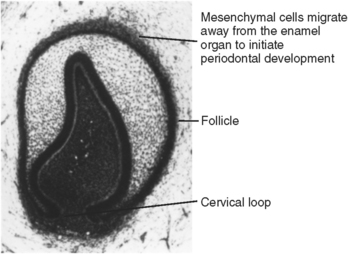
Fig. 5-30 Histology of enamel organ at time of cervical loop development. At this time, mesenchymal cells develop adjacent to the enamel organ on the external surface of the developing enamel organ and differentiate into follicular cells.
Periodontal Ligament
Cells of the dental follicle differentiate into collagen-forming cells of the ligament and form cementoblasts, which lay cementum on tooth roots. Some cells of the ligament invade the root sheath as it breaks apart. Other cells of the ligament area form delicate fibers, which appear along the forming roots near the cervical region of the crown. These are probably the stem cell fibro-blasts that form more fiber groups, which appear as the roots elongate (Fig. 5-31). As these fibers become embedded in the cementum of the root surface, the other end attaches to the forming alveolar bone. Evidence suggests that these fibers turn over rapidly and are continually renewed as the location of origin is established. Collagen fiber turnover takes place throughout the ligament, although the highest turnover is in the apical area and the lowest is in the cervical region. Maturation of the ligament occurs when the teeth reach functional occlusion. At this time, the density of fiber bundles increases notably.
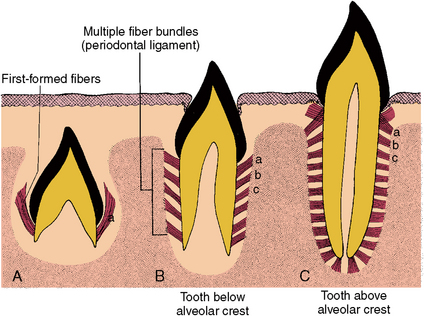
Fig. 5-31 Development of principal fibers of the periodontal ligament. A, Initial fiber development during preeruptive movements (a). B, Secondary fiber development below alveolar crest as tooth moves into prefunctional occlusion (b). C, Further fiber development and maturation of the principal fibers when the tooth reaches functional occlusion (c).
Alveolar Process
As the teeth develop, so does the alveolar bone, which keeps pace with the lengthening roots. At first, the alveolar process forms labial and lingual plates between which a trench is formed where the tooth organs develop. As the walls lining this trench increase in height, bony septa appear between the teeth to complete the crypts (Fig. 5-32). When the teeth erupt, the alveolar process and intervening periodontal ligament mature to support the newly functioning teeth (Fig. 5-33). Bone that forms between the roots of the multirooted teeth is termed interradicular bone. In the mature form, alveolar bone is composed of alveolar bone proper and supporting bone. Alveolar bone proper lines the tooth socket, sustained by supporting bone, which is composed of both spongy and dense or compact bone. Supporting bone forms the cortical plate, which covers the mandible (Fig. 5-34).
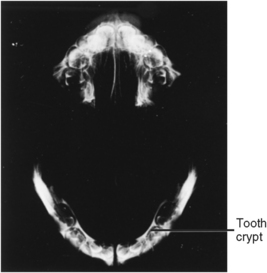
Fig. 5-32 Microradiograph of maxillary and mandibular arches showing alveolar bone and primary tooth crypts enclosing developing teeth.
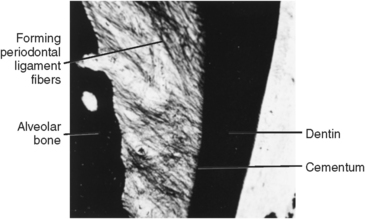
Fig. 5-33 Developing periodontal ligament fibers. Density of fibers similar to C in Figure 5-31.
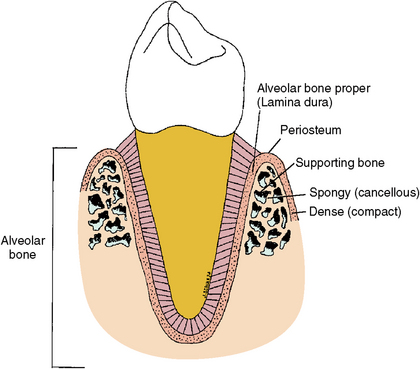
Fig. 5-34 Tooth in alveolar bone. Alveolar bone is composed of alveolar bone proper, which lines the socket, and supporting bone, which consists of spongy or cancellous bone and compact bone.
In summary, tooth development involves the interactive events of two types of tissues: epithelial and mesenchymal. These tissues develop through the soft tissue stages of bud, cap, and bell. This level is followed by the hard tissue formative stages of dentinogenesis and amelogenesis. Root formation logically follows crown development. Each developmental progression includes morphologic changes in shape and size, which are coordinated with microscopic changes in cell shape and function. Most of these relationships are seen in Figure 5-35.
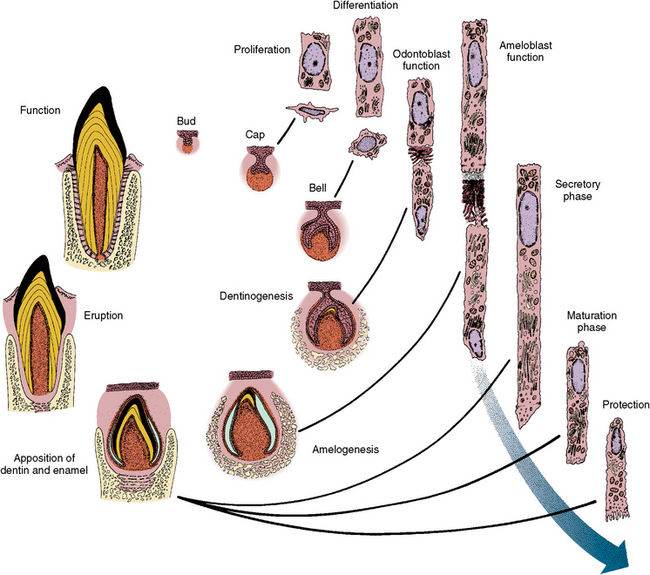
Fig. 5-35 Changes in formative cells of developing teeth shown on the right and correlated with morphologic changes of tooth organ on the left. Cell proliferation relates to the cap stage, whereas cell differentiation relates to the bell stage. Odontoblast function relates to dentinogenesis and ameloblast function to amelogenesis. The labels secretory phase, maturation phase, and protection phase relate to ameloblast function.
Acknowledgments
Dr. Nicholas P. Piesco and Dr. N.M. Elnesr contributed to the production of chapters in Avery JK: Oral development and histology, ed 3, Stuttgart, 2002, Thieme Medical. Some of the figures in that text have been used in preparation of this chapter.
SELF-EVALUATION QUESTIONS
1. What two cell types interact in tooth development?
2. Describe two characteristics of the bell stage of tooth development.
3. List and describe each stage of tooth development.
4. Describe the dental papilla. When does it become the dental pulp organ?
5. Describe the differentiation of the odontoblast and the initiation of dentin formation.
6. Why is dentinogenesis called the two-phases process?
7. What are the five phases of enamel production?
8. What structures enable the ameloblasts to move in a row rather than individually during enamel production?
9. What areas of enamel are first and last to calcify in the crown?
 CLINICAL COMMENT
CLINICAL COMMENT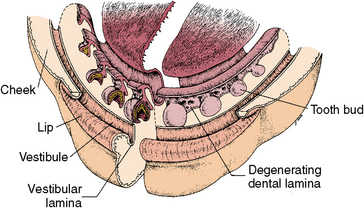
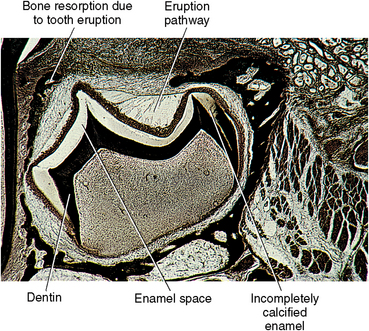
 Consider the Patient
Consider the Patient