Chapter 20 Diseases associated with bacteria – V
DISEASES ASSOCIATED WITH FUSOBACTERIUM AND BACTEROIDES SPP. 1061
DISEASES ASSOCIATED WITH PSEUDOMONAS AND BURKHOLDERIA SPP. 1081
DISEASES ASSOCIATED WITH CAMPYLOBACTER AND LAWSONIA SPP. 1085
DISEASES ASSOCIATED WITH LEPTOSPIRA SPP. 1094
DISEASES ASSOCIATED WITH MYCOPLASMA SPP. 1123
Diseases associated with Fusobacterium and Bacteroides spp.
Infection with Fusobacterium spp. especially F. necrophorum is common in all species of farm livestock. F. necrophorum is a non-spore-forming obligate anaerobe of the family Bacteroidaceae and a normal inhabitant of the alimentary tract (particularly the rumen), the respiratory tract, and the genital tract. There are four biovars, three of which – biovars A, AB, and B – are important in livestock disease. The fourth, biovar C, is not a pathogen. Two of these have been given subspecies status: F. necrophorum subsp. necrophorum (biovar/biotype A) and F. necrophorum subsp. funduliforme (biovar/biotype B). The subspecies vary in their production of leukotoxin, in the quantity and composition of endotoxin and probably in virulence, but the full significance of this to individual diseases in livestock is not yet fully determined as both are isolated from the diseases associated with this infection.1,2 The leukotoxin carries epitopes that can induce protective immunity to experimental infections.3
The specific diseases dealt with here as being associated with primary infection with F. necrophorum are foot rot of cattle, oral necrobacillosis, foot rot of pigs, and foot abscess of sheep. In foot rot of sheep the causative organism (Dichelobacter nodosus) occurs in association with F. necrophorum. In foot rot of pigs a species of Fusobacterium and spirochetal organisms are commonly found together.
In many infections the organism is present as a secondary invader rather than as a primary cause of disease. Some of the common conditions in which F. necrophorum is found as a secondary invader are navel ill and hepatic necrobacillosis in sheep and cattle, pneumonia of calves and in the secondary infections of covering epithelium. These include necrotic enteritis associated with Salmonella spp. in pigs, necrotic rhinitis and atrophic rhinitis of pigs, most diseases in which vesicular eruption and erosive lesions of the buccal mucosa and coronary skin of cattle and sheep occur, and in vulvitis, vaginitis, and metritis.
Fusobacterium spp. have been associated with stomatitis, enteritis, and granulocytopenia in calves, summer mastitis in cattle, hematogenous metaphyseal osteomyelitis in a 6-month-old calf, and endocarditis of swine examined at slaughter.4
The factors which contribute to the pathogenicity of F. necrophorum include a potent endotoxin, a polysaccharide capsule, an exotoxin (a leukocidin), and a hemolysin. The biochemical and functional properties of a leukocidin produced by different strains of F. necrophorum have been described.1,5,6
Extensive studies have been directed towards the immunity of the organism and the possibility of vaccination against the several diseases associated with F. necrophorum. However, the main virulence factors of F. necrophorum are only weakly immunogenic and the experiments give only limited encouragement for an effective necrobacillosis vaccine1,7 although trials with a vaccine against liver abscess in cattle show positive results.8
Other organisms are commonly present in infections with F. necrophorum and it is thought that this is a pathogenic synergy where facultative pathogens utilize oxygen and lower the redox potential to create an anaerobic environment for growth of F. necrophorum. In turn the leukotoxin of F. necrophorum protects other organisms from phagocytosis.1
An enzyme-linked immunosorbent assay (ELISA) for the detection of F. necrophorum antibodies in the serum of cattle and sheep has been developed9 as has a PCR that can differentiate F. necrophorum subsp. necrophorum from F. necrophorum subsp. funduliforme.10
Langworth BF. Fusobacterium necrophorum. Its characteristics and role as an animal pathogen. Bacteriol Rev. 1977;41:373.
Tan ZL, Nagaraga TG, Chegappa MM. Fusobacterium necrophorum infections: Virulence factors, pathogenic mechanisms and control measures. Vet Res Commun. 1996;20:113-140.
Narayanan SK, Nagaraja TJ, Chengappa MM, Stewart GC. Leukotoxins of Gram-negative bacteria. Vet Microbiol. 2002;84:337-356.
1 Tan ZL, et al. Vet Res Commun. 1996;20:113.
2 Brown R, et al. J Med Microbiol. 1997;46:873.
3 Narayan SK, et al. Vet Microbiol. 2003;93:335.
4 Narushima T, et al. J Jap Vet Med Assoc. 1986;56:221.
5 Smith GR, et al. J Hyg Camb. 1985;95:59.
6 Narayanan SK, et al. Vet Microbiol. 2002;84:337.
7 Emery DL, Vaughan JA. Vet Microbiol. 1986;12:255.
8 Jones G, et al. Bov Practit. 2004;38:36.
NECROBACILLOSIS OF THE LIVER (LIVER ABSCESS)
Etiology
Fusobacterium necrophorum subsp. necrophorum (biovar/biotype A) is the most common isolate and is usually present in pure culture. F. necrophorum subsp. funduliforme (biovar/biotype B) is less common and usually isolated as a mixed infection with Arcanobacterium pyogenes
ETIOLOGY
Fusobacterium (Sphaerophorus) necrophorum is commonly found in pure culture in hepatic abscesses in ruminants. F. necrophorum subsp. necrophorum (biovar/biotype A) is the more common type isolated and is usually present in pure culture. F. necrophorum subsp. funduliforme (biovar/biotype B) is less common and usually isolated with other bacterial species, commonly Arcanobacterium (Actinomyces, Corynebacterium) pyogenes Bacteroides spp., Streptococcus spp., and Staphylococcus spp. A. pyogenes is the most common species isolated; it acts synergistically to promote growth of F. necrophorum by utilizing oxygen to create an anaerobic environment and provide iron for growth through its hemolytic activity. The leukotoxin of F. necrophorum is believed to protect A. pyogenes against phagocytosis.1,2
EPIDEMIOLOGY
Occurrence
The disease occurs in all ages and types of cattle and sheep but achieves the greatest economic significance in grain-fed cattle where it occurs secondary to rumenitis. In feedlots in the United States the prevalence varies widely between feedlots but ranges from 12–32% in most;3 equivalent rates occur in ‘barley beef’ cattle in the United Kingdom. Sporadic cases or occasionally outbreaks of liver abscess occur in the neonate from umbilical infection.
Pathogen risk factors
F. necrophorum is a common inhabitant of the intestine and rumen in normal cattle and a common inhabitant of the environment of farm animals. It uses lactate as the major sugar substrate and its numbers in the rumen increase with a change from roughage to high grain diets.4 F. necrophorum is not capable of prolonged survival outside the animal body; 1 month is the probable maximum period under favorable conditions. Infection to the liver requires a predisposing injury at a primary site of infection. The disease can be reproduced experimentally by intraportal inoculation of F. necrophorum.5
Risk factors in grain-fed cattle
Rumenitis, resulting from rumenal acidosis, is the primary site of infection in grain-fed cattle. The risk for liver abscess is increased by factors that predispose rumenitis such as low roughage and high-energy diets and the incidence increases as roughage in the diet decreases.
Management
Introducing hungry cattle to high-energy diets, rapidly increasing dietary energy and poor feedbunk management with irregular periods and amounts of feeding is associated with higher rates of liver abscess.
Diet
The type of grain and the use of processed, including gelatinized, grain can influence risk of abscess as can the physical nature of the diet if it allows feed sorting by the animal during feeding. Cattle hair in the rumen may exacerbate the ruminal lesion and promote the invasion of F. necrophorum and may account for a higher incidence of liver abscess in the spring, when cattle are grooming coats that are shedding.1
Breed
Holsteins are at greater risk than beef breeds because they are fed longer and have higher feed intakes and the prevalence in steers is marginally higher than in heifers, probably also related to higher feed intake.2
Risk factors in other farm animals
In lambs, infection usually occurs through the navel at birth or through ruminal ulcers; the infection originates from infected bedding grounds or barn bedding. Liver abscess can be a sequella to other disease such as traumatic reticulitis and peritonitis.
Economic importance
In feedlot cattle there is considerable financial loss due to condemnation of use of livers in abattoirs. Cattle that are more severely abscessed (with very large abscesses or multiple small abscesses) have a significant depression of weight gain and decrease in feeding efficiency.3
PATHOGENESIS
Vascular drainage from the primary lesion, omphalitis or rumenitis, leads to localization in the liver. The pathogenesis of rumen acidosis in grain-fed cattle is described under that heading.
It is postulated6 that the ruminal wall in rumenitis is colonized by the bacteria in the rumen including both biotype A and biotype B F. necrophorum. Most of the ruminal wall lesions heal without penetration, especially if they contain only the less virulent biotype B. The more virulent biotype A strains persist longer and possibly penetrate the portal system with the help of leukotoxin. The lower virulence biotype B requires helper organisms to penetrate the defense mechanisms and leads to a mixed infection. There are differences in the biological activities of F. necrophorum isolated from liver abscesses and those of the general population in the rumen, and many of the ruminal inhabitants are probably not capable of invasion to cause disease.7
The experimental inoculation of viable cultures of F. necrophorum into the hepatic portal veins of cattle results in the development of diffusely distributed microabscesses within 30 min up to 2 h.6 Gross abscesses develop in 3–36 h. Neutrophils are the predominant phagocyte in lesions of 8 h or less, and macrophages are the predominant phagocyte in lesions of 12 h duration or more. The leukotoxin is postulated to be responsible for allowing the bacteria to withstand the phagocyte cell response and enable the infection to persist. If there is sufficient hepatic involvement, a toxemia develops from the bacterial infection, and causes a chronic or acute illness.
In most infections, the lesions are too small to produce clinical signs. Hematogenous spread from hepatic lesions, including rupture into the caudal vena cava, may result in multiple lesions in many organs, severe pulmonary disease with hemoptysis, and rapidly fatal termination.
CLINICAL FINDINGS
In the majority of cases of hepatic abscessation in feeder cattle there are no clinical signs of illness. Abscesses that are very large may result in an acute or chronic illness.
In acute cases in dairy cattle there is fever, anorexia, depression, fall in milk production, and weakness. Abdominal pain is evidenced on percussion over the posterior ribs on the right side and affected cattle show arching of the back, and reluctance to move or lie down. The liver may be so enlarged that it is readily palpable behind the costal arch. The abdominal pain may be sufficiently severe to cause grunting with each breath. In chronic cases, there are no localizing signs but anorexia, emaciation and intermittent diarrhea and constipation occur.
Animals infected through the navel show signs at about 7 d of age and omphalophlebitis is usually present.
CLINICAL PATHOLOGY
A high leukocytosis with a marked neutrophilia may be present with large or multiple abscesses. Clinical chemistry and liver function tests have been found to be poor indicators and of little diagnostic value in predicting the presence of liver abscesses8 but hepatic dysfunction can be detected by these means in the acute stage of hepatic injury. Ultrasound and centesis may aid in diagnosis.8,9
NECROPSY FINDINGS
Usually, multiple hepatic abscesses are present. The hepatic lesions may be deep in the parenchyma or under the capsule, especially on the diaphragmatic surface. Extension to the diaphragm or perirenal tissues is not unusual.
In bovine rumenitis cases, the anterior, ventral sac is most commonly affected. There are local or diffuse mucosal lesions with thickening of the wall, superficial necrosis and the subsequent development of ulcers. In lambs there may be lesions at the cardial end of the esophagus. The histologic appearance of acute to subacute necrobacillosis lesions consists of a zone of necrosis bordered on one edge by mats of filamentous rods and on the other by a band of karyorrhectic leukocytes.
Acute cases in cattle resemble cases of traumatic reticuloperitonitis and differentiation can only be made on localization of the pain, ultrasonography, and by exploratory rumenotomy. The latter is essential if traumatic hepatitis is a possible diagnosis.
TREATMENT
F. necrophorum is sensitive to B-lactam antibiotics, tetracyclines, the macrolide and lincosamide antimicrobials but is resistant to aminoglycosides and ionophores with no difference in sensitivity between the two subspecies except for clindamycin and lincomycin.10
Liver abscess in feedlot cattle is not clinical and not routinely treated as a clinical disease. In clinical disease associated with liver abscess, prolonged treatment with high doses of antimicrobial or sulfonamides is required if therapeutic concentrations are to be achieved at the site of infection. Relapse is common because of incomplete control of the localized infection.
CONTROL
Control procedures in feedlot cattle include prevention of rumenitis by feed management and the use of prophylactic antimicrobials.
Feed management
Feed management aims to prevent the occurrence of rumen acidosis and rumenitis and requires controlled dietary energy step up, attention to the grain type and content of the diet and correct feedbunk management.11 The control of rumen acidosis is discussed elsewhere under that title.
Prophylactic antibiotics
The addition of antimicrobials to the feed can significantly reduce the incidence of liver abscesses and is a routine practice in most feedlots. The site of action may be in rumen or the liver or possibly in both, but is probably the rumen as tylosin and virginiamycin, which are both effective in prevention, are not absorbed into the circulation.8 Tylosin has been shown to inhibit the increase in ruminal F. necrophorum numbers that occur in association with feeding high-grain diets.2
In the United States, bacitracin, chlortetracycline, oxytetracycline, tylosin, and virginiamycin are approved for feed inclusion for the control of liver abscess in feedlot cattle. Tylosin appears highly effective2 and a summary of trials feeding tylosin at 11 g/t of feed or 90 mg/animal/d showed a 73% reduction in the occurrence of liver abscesses.2 Antimicrobial feed additives also increase average daily gain and feed conversion efficiency, but the inclusion level for these effects and prevention of liver abscess is not necessarily the same.12
Vaccination
Vaccination with leukotoxoid vaccines has shown some protection against intraportal challenge13 and has reduced the abscess rates in a study of naturally occurring disease in a feedlot.14 A trial of the efficacy of a high antigenicmass-combined Arcanobacterium pyogenes-Fusobacterium necrophorum bacterin-toxoid in preventing naturally occurring liver abscess in feedlot cattle showed a significant effect of vaccination with a reduction of the prevalence and severity of abscesses that was equivalent to that achieved by the incorporation of tylosin in the feed.15 The reduction in prevalence in two trials comparing vaccinated and non-vaccinated cattle was 48.4% (31% of controls and 16% of vaccinates with liver abscess) and 37.5% (48% of con-trols and 30% of vaccinates with liver abscess).15
Scanlan CM, Hathcock TL. Bovine rumenitis-liver abscess complex: A bacteriological review. Cornell Vet. 1983;73:288-297.
Nagaraja TG, Laudert SB, Parrott JC. Liver abscesses in feedlot cattle. Part 1. Causes, pathogenesis, pathology and diagnosis. Comp Cont Educ Pract Vet Suppl. 1996;18:S230-S256.
Nagaraja TG, Laudert SB, Parrott JC. Liver abscesses in feedlot cattle. Part 11. Incidence, economic importance and prevention. Comp Cont Educ Pract Vet Suppl. 1996;18:S264-S273.
Nagaraja TG, Chengappa MM. Liver abscesses in feedlot cattle: A review. J Anim Sci. 1998;76:287-298.
1 Nagaraja TG, et al. Comp Con Educ Pract Vet Suppl. 1996;18:S230.
2 Nagaraja TG, Chengappa MM. J Anim Sci. 1998;76:287.
3 Brink DR, et al. J Anim Sci. 1990;68:1201.
4 Tan ZL, et al. Appl Environ Microbiol. 1994;60:1387.
5 Saginala S, et al. Vet Res Commun. 1996;29:493.
6 Nakajima YH, et al. Jpn J Vet Sci. 1986;48:509.
7 Okwumabua O, et al. Appl Environ Microbiol. 1996;62:469.
8 Liberg P, Jonsson G. Acta Vet Scand. 1993;34:21.
9 Braun U, et al. Vet Rec. 1995;137:209.
10 Lechtenberg KF, et al. Am J Vet Res. 1998;59:44.
11 Popp JD, et al. Vet Clin Nutrit. 1997;4:63.
12 Rogers JA, et al. J Anim Sci. 1995;73:9.
13 Saginala S, et al. J Anim Sci. 1997;75:1160.
BOVINE INTERDIGITAL NECROBACILLOSIS (FOUL IN THE FOOT, FOOT ROT, INTERDIGITAL PHLEGMON)
Epidemiology
All ages susceptible. Infected feet are source of infection. Transmission highest where conditions are wet underfoot and in wet humid seasons
ETIOLOGY
Foot rot is usually described as being associated with F. necrophorum. Other bacteria such as Prevotella melaninogenica (formerly Bacteroides melaninogenicus) are also present and may be important in cause. Experimentally, the subcutaneous inoculation of only F. necrophorum into the interdigital skin of cattle will result in typical lesions of interdigital necrobacillosis.1 The majority of isolates of F. necrophorum obtained from the feet of cattle and sheep belong to biotypes A and AB; they produce a soluble exotoxin, a leukocidin, and are pathogenic experimentally in cattle and mice.2 The isolates obtained from lesions which are not classified as interdigital necrobacillosis and from clinically normal feet are predominantly biotype B (F. necrophorum subsp. funduliforme), and cause few experimental lesions and produce little or no leukocidin.
Strains of Bacteroides nodosus, that are associated with the non-progressive form of ovine foot rot, can also be isolated from the feet of cattle and cause mild interdigital dermatitis. It is possible they may predispose to the much more severe dermatitis that characterizes bovine interdigital necrobacillosis.
EPIDEMIOLOGY
Occurrence
The disease is common in most countries and accounts for 5–15% of cases of lameness in dairy cattle.3,4
Usually the disease is sporadic but under favorable conditions, as many as 25% of a group may be affected at one time. An epidemiological study of foot rot in pastured cattle in Denmark over a 12-year period revealed that incidence rates annually ranged from 0.1–4.8% but in most years it was below 1%.5 The incidence rates were higher in some breeds than others, higher in some geographical areas than others (usually where the fields were smaller and soil higher in pH), and 4–8 weeks after periods of high rainfall.
Transmission
Discharges from the feet of infected animals are the probable source of infection. Duration of the infectivity of pasture or bedding is unknown. Infection gains entrance through abrasions or damage to the skin in the interdigital cleft. Introduction of the infection to a farm by transient cattle is often observed but again the disease may not develop on some farms in spite of the introduction of the infection. Contaminated footbaths can be a source of infection.
Environmental risk factors
In many6 but not all3 regions, the incidence is much higher during wet, humid weather or when conditions are wet underfoot. Stony ground, lanes filled with sharp gravel and pasturing on coarse stubble also predispose to the condition. A high incidence can occur with beef cattle at high stocking densities on irrigated pastures. The observation that the disease is common on some farms and does not occur at all on others suggests that there may be factors which limit the persistence of infectivity in certain soils or environments.
Abrasions to the skin of the feet are more likely to occur when the skin is swollen and soft due to continual wetting. The increased incidence in wet summer and autumn months may be so explained in part, although wet conditions may also favor persistence of the infection in pasture. In housed cattle the incidence is higher in loose-housed cattle than tied cattle.6 Unhygienic cubicle passageways and poorly maintained straw beds may predispose to infection.
Host risk factors
Cattle of all ages, including young calves, may be affected but the disease is much more common in adults. The highest incidence occurs in cows in the first month of lactation.6 A field observation is that Bos indicus cattle are much more resistant to infectious foot rot than Bos taurus breeds and variations in prevalence have been observed among dairy breeds.6 The occurrence of the foot disease digital dermatitis on a farm or infection with bovine virus diarrhea has been suspected as predisposing to more severe foot lesions.7
Economic importance
Foot rot is of greatest economic importance in dairy cattle, in which it reaches the highest level of incidence, because of the intensive conditions under which they are kept. In beef cattle at range the incidence is usually low but many cases may occur in purebred herds and in feedlot cattle. Lame cows will lie down for longer and eat less, have difficulty rising and are at greater risk for teat trampling and mastitis. Loss of production occurs and an occasional animal may suffer a serious involvement of the joint and other deep structures of the foot necessitating amputation of a digit. The disease is not fatal but some cases may have to be slaughtered because of joint involvement.
PATHOGENESIS
The pathogenesis is not completely understood, but with the experimental SC inoculation of the virulent biotype of F. necrophorum into the interdigital skin of cattle the typical lesion of foot rot develops in approximately 5 d.1 This suggests that any injury or constant wetting of the skin of the cleft which interferes with its integrity will allow the organism to invade the tissues. There is acute swelling and necrosis of the skin and SC tissues which may spread to adjacent tendon sheaths, joint capsules and bone if treatment is delayed or ineffective.
CLINICAL FINDINGS
Severe foot lameness appears suddenly, usually in one limb only and may be accompanied by a moderate systemic reaction with a fever of 39–40°C (103–104°F). There is temporary depression of milk yield in cows and affected bulls may show temporary infertility. The animal puts little weight on the leg although the limb is carried only when severe joint involvement occurs. Swelling of the coronet and spreading of the claws are obvious.
The typical lesion occurs in the skin at the top of the interdigital cleft and takes the form of a fissure with swollen, protruding edges which may extend along the length of the cleft or be confined to the anterior part or that part between the heel bulbs. Pus is never present in large amounts but the edges of the fissure are covered with necrotic material and the lesion has a characteristic odor. Occasionally in early cases no external lesion may be visible but there is lameness and swelling of the coronet. Such cases are usually designated ‘blind fouls’ and respond well to parenteral treatment.
A more severe form of the disease which is peracute in onset and refractory to conventional therapy has been termed ‘super foul’ or ‘super foot rot’.8 With this type there is sudden onset of acute lameness, severe interdigital swelling and rapid progression to necrosis and deep erosion of the interdigital space with swelling of soft tissue above the coronary band. The hindfeet or all four feet may be affected.7 It may be predisposed by the foot disease digital dermatitis. Septic arthritis is a common sequella if this type is not treated early in its course.
Spontaneous recovery is not uncommon but if the disease is left untreated, the lameness usually persists for several weeks with adverse effects on milk production and condition. The incidence of complications is also higher if treatment is delayed and some animals may have to be destroyed because of local involvement of joints and tendon sheaths. In such cases the lameness is severe, the leg is usually carried and the animal strongly resents handling of the foot. Swelling is usually more obvious and extends up the back of the leg. There is poor response to medical treatment and surgical measures are necessary to permit drainage. Radiological examination may be of value in determining the exact degree of involvement of bony tissue.
Long continued irritation may result in the development of a wart-like mass of fibrous tissue, the interdigital fibroma, in the anterior part of the cleft and chronic mild lameness. Interdigital fibroma occurs commonly without the intervention of foot rot, the important cause being inherited defects in foot conformation in heavy animals.
CLINICAL PATHOLOGY
Bacteriological examination is not usually necessary for diagnosis but direct smears of the lesion will usually reveal large numbers of a mixture of Fusobacterium and Bacteroides spp. Routine differentiation between virulent and non-virulent bovine isolates of F. necrophorum can be done by assessment of the cultural characteristics of the colonies grown on blood agar.2
The characteristic site, nature, and smell of the lesion, the pattern of the disease in the group and the season and climate are usually sufficient to indicate the presence of true foot rot.
NECROPSY FINDINGS
Necropsy examinations are rarely carried out in cases of foot rot. Dermatitis is followed by necrosis of the skin and SC tissues. In complicated cases there may be suppuration in joints and tendon sheaths.
Interdigital dermatitis/stable foot rot
This infection occurs commonly in cattle which are housed for long periods. Although the condition occurs most commonly when the cattle are kept under unsanitary conditions, it is also seen in well-managed herds. The causative agent has not been established but Bacteroides nodosus can be isolated.
The initial lesion is an outpouring of sebaceous exudate at the skin–horn junction, particularly at the bulbs of the heel. There is a penetrating foul odor and the lesion is painful to touch, but there is little swelling and no systemic reaction. More than one foot is commonly affected. In longstanding cases there is separation of the horn at the heel bulb and this is followed by secondary bacterial infection of the sensitive structures of the foot. Often there is a purulent dermatitis of the interdigital space. Stable foot rot does not respond satisfactorily to the standard parenteral treatments used in foot rot but local treatments as set out as follows are effective.
Verrucose dermatitis
Verrucose dermatitis is a proliferative inflammatory lesion of the skin of the plantar surface of the foot extending from the bulb of the heels to the fetlock joint. The condition is seen particularly in feedlot cattle that are overcrowded in wet muddy conditions and may occur in outbreaks. All four feet may be affected, there is considerable pain and lameness and, on smear of the lesion, F. necrophorum is present in large numbers. The treatment of verrucose dermatitis consists of washing the affected skin with a disinfectant soap, followed by daily applications of 5% copper sulfate solution. When many animals are affected, a daily walk through and soaking in a foot bath containing the copper sulfate solution is very effective.
Traumatic injury
Traumatic injury to bones and joints, puncture by foreign bodies, bruising of the heels and gross overgrowth of the hoof can usually be distinguished by careful examination of the foot. Laminitis is the major cause of lameness in most herds but with this condition there are no skin lesions present.
TREATMENT
Parenteral administration of antibiotics or sulfonamides and local treatment of the foot lesion are necessary for best results. Immediate treatment as soon as possible after the onset of swelling and lameness will give excellent recovery in 2–4 d. In the experimental disease, when treatment was delayed for a few days after the onset of signs, severe lesions developed and recovery was extended. Under field conditions, the disease may be present in cattle at pasture for several days before being recognized, making it necessary to confine them for daily treatment until recovery is apparent.
Antimicrobials
Ceftiofur, 1–2 mg/kg (BW) IM, or procaine penicillin G, 22 000 iu/kg BW IM twice daily, or once daily for three consecutive days are effective.9 Oxytetracycline, 10 mg/kg BW IV, or long-acting tetracycline IM may also be used but injection site tolerance and withdrawal period is less favorable than ceftiofur. When a high incidence of foot rot is experienced in a herd, treatment of all animals simultaneously has been carried out.10
Sodium sulfadimidine (150–200 mg/kg BW) solution given by IV injection is highly effective. Sulfabromomethazine at the rate of 30 g/kg grain was given for two consecutive days to calves weighing 150 kg and results were excellent. Sulfonamides are not approved for use in lactating dairy cattle in many countries. ‘Super foot rot’ has been effectively treated by parenteral tylosin or a single dose of long-acting tetracycline.8
Local treatment
Local treatment necessitates restraint of the affected leg and this procedure is greatly facilitated by a restraint table or the administration of a very small dose of xylazine. The foot is scrubbed, all necrotic tissue curetted away and a local dressing applied under a pad or bandage. Any antibacterial, and preferably astringent, dressing appears to be satisfactory. A wet pack of 5% copper sulfate solution is cheap and effective. Any suitable antibacterial ointment preparation may be applied and secured with a bandage, which may be left on for several days. The main advantage of local treatment is that the foot is cleaned and kept clean. If conditions underfoot are wet, the animal should be kept stabled in a dry stall.
In cattle running at pasture, or in the case of large numbers of feedlot cattle, examination of the foot and local treatment are often omitted because of the time and inconvenience involved. However, identification of the animal with a marker is considered necessary in outbreaks to avoid unnecessary confusion in the days following, and examination of the foot is deemed necessary to insure that foreign bodies are not involved. Local treatment may not be necessary in the early stages of the disease if the animal can be prevented from gaining access to wet, muddy areas.
CONTROL
Prevention of foot injuries by filling in muddy and stony patches in barnyards and lanes will reduce the incidence of the disease. Lanes and bedding should be kept clean and dry. The incorporation of biotin in the diet, while reducing the incidence of lameness caused by white line lesions, has no effect on the incidence of interdigital necrobacillosis.11
Footbaths
Provision of a footbath containing a 5–10% solution of formaldehyde or copper sulfate, in a doorway so that cattle have to walk through it twice daily, will practically eliminate the disease on dairy farms. A mixture of 10% copper sulfate in slaked lime is often used in the same manner. Similar measures can be adopted for small groups of beef animals.
Antibacterials
Feeding chlortetracycline to feedlot cattle 500 mg/head/d for 28 d, followed by 75 mg/d throughout the finishing period has been recommended but controlled comparative trials have not been carried out. The feeding of organic iodides (200–400 mg) of ethylene diamine dihydroiodide (EDDI) in the feed daily has been used for many years as a preventive against the disease in feedlot cattle. Feeding EDDI in an ad libitum salt mixture at a level of 0.156% EDDI (0.125% iodine) is also effective in reducing the incidence of foot rot. Dosing cattle daily with zinc sulfate by including it in the feed has no prophylactic effect.
Vaccination
Commercial vaccines against bovine interdigital necrobacillosis are available but their efficacy has not been established in controlled comparative trials.12 A mineral-oil adjuvant vaccine containing whole cells or fractions of F. necrophorum provided about 60% protection from experimentally induced interdigital necrobacillosis. A similar vaccine containing Bacteroides nodosus appeared to reduce the severity of lesions but not the incidence rate compared to non-vaccinates.
1 Clark BL, et al. Aust Vet J. 1985;62:47.
2 Emery DL, et al. Aust Vet J. 1985;62:43.
3 Murray RD, et al. Vet Rec. 1996;138:586.
4 Jubb TF, Malmo J. Aust Vet J. 1991;68:21.
5 Monrad J, et al. Acta Vet Scand. 1983;24:403.
6 Alban L, et al. Prev Vet Med. 1995;24:73.
7 Daniel R, et al. Vet Rec. 1995;137:647.
8 Cook NB, Cutler KL. Vet Rec. 1995;136:19.
9 Rebhun WC, Pearson EG. J Am Vet Med Assoc. 1982;181:572.
10 Clark BL, et al. Aust Vet J. 1986;63:107.
BOVINE DIGITAL DERMATITIS, PAPILLOMATOSIS DIGITAL DERMATITIS OF CATTLE (MORTELLARO’S DISEASE) (FOOT WARTS, HAIRY FOOT WARTS, ‘HEEL WARTS’)
Papillomatous digital dermatitis (PDD) is a painful, erosive, papillomatous-like lesion of the skin of the feet of cattle. The region proximal and adjacent to the interdigital skin midway between the heel bulbs of the plantar surface of the foot is most frequently affected. Early lesions are circumscribed, with a red, granular appearance and variable degrees of proliferation of filiform papillae. Mature lesions tend to be more proliferative and may have long papillary fronds. Lameness is severe, and economic losses are due to decreased milk production and reproductive performance.1
ETIOLOGY
The etiology is uncertain. A mixed population of Gram-negative bacteria including anaerobes, microaerophilic organisms, and spirochetes have been demonstrated in or isolated from PDD lesions. Histologically, invasive spirochetes have been isolated from the tissues of cattle with typical lesions, but their significance is uncertain.2 The humoral response to spirochetes in cattle with the disease is different from that in cattle without detectable lesions.1 At least five different spirochetal phylotypes within the genus Treponema have been identified in lesions and Treponema denticola-like spirochetes were most common.3 DNA analysis of the spirochetes recovered from typical lesions revealed that the organisms were closely related to Treponema denticola.4 A combination of spirochetes and Campylobacter faecalis has also been found in typical lesions.5
A spirochete isolated from cases of severe virulent ovine foot rot (SVOFR) in Australia6 and the UK7 and Ireland8 is closely related to a treponeme isolated from human periodontitis and bovine digital dermatitis. This suggests the possibility of cross-species transmission, and that a number of spirochetes could be involved in the pathogenesis of either PDD or SVOFR.
EPIDEMIOLOGY
Occurrence and prevalence of infection
The disease was first described in Italy in 1974 as Mortellaro’s disease. It occurs primarily in dairy cattle and has been reported as a cause of lameness in cattle worldwide.9 Beef cattle are also affected. Surveys of dairy farms in California in the United States found that 25–75% of farms have had the disease and about 10% of cows have been affected with a range from 1–99% of cows in affected herds.10
The disease has occurred in a herd of dairy bulls in New Zealand.11 Lesions similar to PDD have been recognized in cattle in Australia, occurring during periods of high rainfall and being responsive to topical and parenteral therapy, but not associated with the presence of spirochetes in the lesions.12
The National Animal Health Monitoring System (NAHMS) Dairy 1996 assessed the incidence of digital dermatitis in US dairy herds.13 The study design was a population-based cross-sectional survey of US dairy herds with at least 30 cows in 20 states, representing 79% of US dairy cows. PDD was reported in the previous 12 months from 43.5% of dairy herds. Within affected herds, 18.9% of cows were affected and 81.9% of affected cows and 85.9% of affected heifers were lame. Regions of the US with the highest percent of herds affected included the Southwest, Northwest, and Northeast. Factors associated with high incidence (>5%) included region, herd size, type of land lactating cows accessed on a daily basis, flooring type where lactating cows walked, percent of cows born off the farm, use of a primary hoof trimmer who trimmed cows’ hooves on other farms. and lack of washing of hoof-trimming equipment between cows.
People providing a hoof-trimming service report that 97% of dairy farms in California have the disease but, mysteriously, some farms have none. Regional differences in the prevalence occurred but reasons for the incidence were not examined. Seasonal differences occur and may be due to a combination of weather, housing, and management. The incidence may be higher during the winter months, when the weather is cold and wet, and cows are kept in confined housing than during the summer months when cows are on pasture.
Based on examination of the feet of cattle at an abattoir, 29% of culled dairy cattle and 4% of beef cattle had gross lesions of PDD, and spirochetes were present in 61% of the lesions.14
In a commercial dairy farm in Mexicali, Mexico, 33% of cows were affected during lactation and 1% during the dry period; 68% of cows that had lesions in the previous lactation had lesions in the current lactation.15 The highest risk occurred during the first month of lactation. The yearly estimated cumulative incidence risk was 35% and the incidence density rate was 44.6 cases per 1000 cow-months. More animals were affected in summer and fall than in winter and spring. Purchased animals were 3.4 times more likely to be affected than animals born on the farm. Healthy cows conceived 93 days after calving (median), but affected cows conceived 113 days after calving.
In 214 dairy herds in two main dairy areas in Chile, 91% of herds were affected and a median of 6% of cows had PDD.16
Risk factors
Host risk factors
A cross-sectional study of prevalence and risk factors of digital dermatitis in female dairy calves between 2.5 and 12 months of age in the Netherlands revealed a mean prevalence of 67%.17 The prevalence increased with age and by 1 year of age almost all animals had lesions in at least one claw. The clinical disease is much more common in first calf heifers and young cows. First-parity cows have the highest odds of PDD, and the odds decrease, in a dose-effect manner, as parity increases.18 The odds of PDD increase with increasing days in lactation.
The plantar and palmar regions of the foot may be more conducive to the development of PDD because these anatomical sites are exposed to more moisture. Epidemiologic observations indicate that the risk of PDD is associated with environmental conditions that cause moist feet in commercial dairy herds. The greater incidence of the lesions in the hindfeet is considered to be associated with more exposure to deeper slurry during feeding times than the forelimbs. The plantar and palmar regions of the interdigital cleft are, therefore, more susceptible to being continually moist. compared with the more open dorsal locations. The anatomic location of PDD lesions also has an effect on the efficacy of topical treatment with antibiotics.19
Immune mechanisms
Treponema phagedenis-like spirochetes isolated from active PDD lesions in dairy cattle are associated with serum IgG2 antibodies and most react with lipopolysaccharide.20,21 Both the antibody and blastogenic responses were reduced in convalescent dairy cattle, suggesting the immune response to the spirochetes has short duration. The presence of IgG2 spirochete antibodies detected by ELISA does not necessarily describe an active immune protective response by affected cows but reflects prior infection and repeated exposures to treponemes.22
Cattle and sheep with PDD and severe virulent ovine foot rot, respectively, and which may be infected by the same group of treponemes, have increased seropositivity rates to both treponeme isolates, with different patterns of reactivity between farms.7
Environmental and management risk factors
Case–control studies in dairy farms indicate that the odds of having a higher proportion (>5%) of affected cows were about 20 times more likely in dairy farms with muddier corrals than in farms with drier ground surfaces in corrals.10 The disease appears to be more common in freestall confined herds where feet are constantly expose to moisture and manure conditions. The feet often become coated with a layer of dried feces which may provide the anaerobic conditions necessary for bacterial growth.
Buying replacement heifers was associated with a 4.7-fold increase in the odds of a higher occurrence of disease than in herds which did not purchase heifers. In dairy herds in Chile, cows on farms that bought heifers in the previous 10 years had a 3-fold increase in the odds of PDD compared to those on farms that never bought heifers. Cows in dairy herds that used a footbath were less likely to have PDD than those herds not using one. There also may be a positive relationship between risk and the number of heifers purchased. Animals housed in a straw yard were 3.2 times less likely to be affected compared to cattle on slatted floors. Herd size was positively associated with the presence of the disease. Feeding with a larger variety of dietary components (hay, milk, concentrates plus silage) was a protective measure.
Pathogen risk factors
Several spirochete strains have been isolated from PDD lesions in dairy cattle in Iowa and compared with strains previously isolated from dairy cattle in California.20 All strains are Treponema phagedenis-like and exhibit genetic and antigenic diversity.
Molecular typing of PDD-associated Treponema isolates have found some genetic relatedness to those of the related human-associated Treponema spp. associated with human periodontal disease.23,24 These Treponema strains have adhesion properties and produce high levels of chymotrypsin-like protease and high levels of proline iminopeptidase which are major virulence factors.
Economic importance
The economic losses associated with the lameness associated with PDD in lactating cows include loss in milk production,25 the effects on reproductive performance, and the costs of treatment including the time required to recognize the lesions and the costs of individual medication of affected cows if necessary, and the costs of construction and maintenance of a foot bath. Lameness has an important effect on milk yield. In a data set including 8000 test-day milk yields from 900 cows on five farms in the UK collected over 18 months, the total mean estimated reduction in milk yield per 305-d lactation was 360 kg.25
In a 600-cow dairy herd in Mexico, cows with PDD produced a mean of 268 lb less milk than healthy cows.26 Using Dairy Herd Improvement Association Records in the dairy herds in Florida, interdigital phlegmon was associated with a 10% decrease in milk production. Cows with PDD produced less milk than healthy cows but the difference was not significant.
Lameness has an important effect on reproductive performance.27 Dairy cows with claw lesions have a higher calving to conception interval, and a greater number of services per conception.
PATHOGENESIS
PDD is an acute or chronic ulcerative lesion of the skin of the bulbs of the heel or interdigital cleft. In the early stages of the lesion there is loss of superficial keratin, with a concurrent thickening of the epithelium by both hyperplasia and hypertrophy of epithelial cells. Superficial layers are eosinophilic and undergo necrotic change with the appearance of small holes. Large numbers of spirochetes are present around the holes. Loss of superficial layers of keratin stimulates epidermal proliferation and hyperplasia. In advanced cases, large numbers of spirochetes infiltrate the eroded dermis and may destroy the epidermis.28 PDD is characterized by erosion of the superficial layers of the epidermis, epithelial hyperplasia and hypertrophy, pain, swelling, and a foul odor. Lesions usually occur on the hind feet and are prone to bleeding. Early lesions are circumscribed with a red, granular (strawberry-like) appearance and variable degrees of proliferation of filiform papillae. Mature lesions are more proliferative and may have long wart-like projections, thus the term ‘hairy-wart’ disease.
CLINICAL FINDINGS
PDD typically occurs in dairy cattle as lameness episodes of variable severity. Affected cattle are lame and reluctant to move. The affected limb is often held trembling in partial flexion as if the animal is in pain. Less severely affected limbs are rested on the toes and animals may walk on their toes, which become markedly worn and may even expose the sensitive laminae. Affected cattle lose weight and may not eat normally if they have to walk some distance to obtain feed. Milk production may decline if the lesions are severe enough.
Lesions are confined to the digits and do not occur above the dewclaws. The feet of the hind limbs are most commonly affected. The plantar surface of the feet are most commonly affected but the palmar aspects may also be involved.
The majority of lesions are medium to large, measure 2–6 cm across at their largest dimension and are located on the skin at its junction with the soft perioplic horn of the heel and midway between the two claws. Most lesions are situated proximal and adjacent to the plantar/palmar interdigital space and rarely involve the interdigital skin. The surface of the lesions is moist, prone to bleeding and intensely painful to the touch. The lesions are circular to oval in shape, raised and variable in color and in degree of papillary proliferation. The washed surfaces are typically red and granular or a composite of white-yellow, gray, brown, or black papillary areas mixed with red granular areas (strawberry-like) with a very strong pungent odor. Filiform papillae commonly protrude from the surface of the lesions. Most lesions are circumscribed or delineated by a discrete line of raised hyperkeratotic skin with long wart-like projections. The lesions are restricted to the skin and do not extend into the deeper soft tissues. If untreated, PDD can persist for months associated with persistent lameness, reduced milk production, and impaired reproductive performance, and premature culling. More advanced lesions may lead to progressive separation of horn from the sensitive laminae, resulting in a typical underrun sole which may extend forward from the heel to reach half-way to the toe. Outbreaks of the disease may occur in dairy herds in which up to 75% of all cows may be affected over a period of several months.29
A screening method for the detection of lesions of dairy cattle has been described.16 At the milking parlor and once the cows are in place for milking, a water hose is used to wash the cows’ feet. Then using a powerful flashlight, the digits are carefully inspected for PDD lesions. A PDD case is defined as a cow with a circular-to-oval, well-demarcated, alopecic, moist, erosive foot lesion, surrounded by a white hyperkeratotic ridge or hypertrophic hairs. Lesions bleed easily and are very painful. When impacted by a concentrated jet of water from a hose, the animal frequently reacts by pulling the foot away and sometimes shaking it. Lesions can be classified into one of three stages based on their morphology: early, classical, and papillomatous. The screening method has a sensitivity of 0.72 and a specificity of 0.99.
CLINICAL PATHOLOGY
Detection of organism
Smears of the exudate and scrapings of the surface of the lesions are submitted for culture and for staining for spirochetes. Dark-field microscopy of the scrapings may reveal profuse motile spirochetes with vigorous rotational and flexing movements. Biopsy specimens of the lesions can be submitted for histological examination and special silver staining to identify the spirochetes.
PATHOLOGY
The majority of lesions are 2–6 cm across their largest dimension, circular to oval, raised, and variable in color.30 Washed surfaces are typically either extensively red and granular or a composite of white-yellow, gray, brown, and/or black papillary areas interspersed with red granular areas. The surface of the lesions is covered by filiform papillae, 0.5–1 mm in caliber and 1–3 mm in length. Most lesions are characteristically circumscribed or delineated by a discrete line of raised hyperkeratotic skin often bearing erect hairs 2 to 3 times longer than normal. The surfaces are also partially to completely alopecic, moist, prone to bleed and intensely painful to touch. Histologically, active lesions are characterized by zones of acute degeneration, necrosis, and inflammatory cell infiltration within the stratum corneum, usually associated with focal thinning.14 Using immunocytochemical staining and PCR of lesion biopsies, Borrelia burgdorferi, Treponema denticola, and Treponema vincentii have been identified.31
Digital dermatitis must be differentiated from:
• Interdigital dermatitis: moist, gray thickening of the skin, with focal areas of shallow ulceration and hyperkeratosis. It is less painful and rarely has a granular, tufted or papillomatous surface
• Heel erosion (slurry heel) – occurs commonly in dairy cows standing for long periods in slurry.32 The intact smooth horn of the heel develops deep, black fissures which may become totally eroded. There is no liquefaction necrosis of keratin characteristic of digital dermatitis32
• Interdigital necrobacillosis (foot rot) – a necrotizing infection of the interdigital skin. There is marked painful deep swelling of the tissues of the interdigital cleft; cracking of the skin may occur with release of a foul-smelling discharge. Response to treatment with antimicrobials is good unless the lesion is advanced
• Verrucose dermatitis – occurs in cattle kept in deep muddy yards and is characterized by marked painful proliferative dermatitis of the plantar surface of pastern from the bulbs of the heels to the fetlocks. Fusobacterium necrophorum is usually present in the lesions. Affected cattle are lame and respond to topical treatment and use of a footbath with a suitable antimicrobial
• Interdigital fibroma (corn) – develops from the fold of skin adjacent to the axial wall of the hoof in the interdigital space. The lesion consists of firm fibrous tissue and may extend the entire length of the interdigital cleft. Lameness is caused by the presence of the corn in the interdigital cleft; advanced corns must be removed surgically.
TREATMENT
Antimicrobials parenterally and topically
Antimicrobial therapy is indicated and effective and various methods of administration have been used including parenteral and topical application in individual animals, and foot baths for medication of large numbers of animals.
Parenteral antimicrobials
Procaine penicillin, at 18 000 U/kg BW IM twice daily for 3 days, or ceftiofur sodium at 2 mg/kg daily for 3 days was highly successful for the treatment of PDD in dairy cattle in California.30 However, the use of parenteral antimicrobials on an individual basis is labor intensive, costly, and not feasible when large numbers of animals are involved. In addition, drug residues in the milk are more likely when animals are treated parenterally. Recurrence after treatment may also occur. In one report, the lesions recurred in 18% of cows treated with antibiotics parenterally.30
Cleaning the surface of the lesion
It is very important to wash and clean the surface of the lesion with a disinfectant soap before the topical administration of any medication. Treatment failures are commonly associated with failure to adequately wash and clean the surface of the lesion.
Topical antimicrobials
Topical antimicrobial sprays or ointments are used on individual animals after the lesions have been cleaned. Direct spraying of the lesions with oxytetracycline at 25 mg/mL in 20% glycerine in deionized water once daily for 5 d using a garden-type spray applicator was effective.33 Only affected cows and individual lesions should be treated.34
The anatomic location of PDD lesions has an effect on the efficacy of topical treatments. The use of oxytetracycline solution (25 mg/mL in distilled water) as a topical spray on cows with PDD lesions was most effective on lesions located on the heels or dewclaws compared to those in the interdigital cleft.19 Application of oxytetracycline and bandaging the affected part has been effective but not practical when large numbers of animals are affected.
Oxytetracycline solution (100 mg/mL), acidified ionized copper solution, acidified sodium chlorite, or placebo given as a topical spray three times daily, after washing the lesions, for 3 weeks was effective in decreasing the lameness associated with the disease.33 In a Swedish dairy herd, topical oxytetracycline was more effective for the treatment of PDD in cattle with heel-horn erosion than hoof trimming alone and than glutaraldehyde.35
The use of an oxtetracycline solution topically at doses of 15 mL of a solution containing 100 mg oxtetracycline/mL sprayed twice daily for 7 days, or a one-time application of a bandage of cotton soaked with 20 mL of a solution containing 100 mg oxtetracycline/mL had a low risk of causing violative antibiotic residues in milk.36
Lincomycin at a dose of 25 mL of a solution containing 0.6 mg lincomycin/mL or valnemulin at a dose of 25 mL of a solution containing 100 mg/mL valnemulin, given as an individual topical spray for two treatments 48 hours apart resulted in significant improvement within 14 days after the first treatment.37
Nonantibiotic formulations
The efficacy of oxytetracycline has been compared with nonantibiotic solutions, a commercial preparation of soluble copper, peroxide compound and a cationic agent, 5% copper sulfate, acidified ionized copper solution, hydrogen peroxide-peroyacetic acid solution, and, tap water, for the treatment of PDD.38 The commercial formulation of soluble copper, peroxide compound, and a cationic agent appeared to be as effective as oxytetracycline. A non-antimicrobial cream containing soluble copper with peroxide and a cationic agent was compared with topical lincomycin.39 The efficacy of the treatments was not different for decreasing pain or lesion activity but lincomycin was more effective in decreasing lesion size and preventing recurrence. Cows with ≥3 lactations were more likely to have a healed lesion at 29 days, compared with first and second lactation cows.
A nonantibiotic formulation based on a reduced soluble copper solution, peroxide compound, and a cationic agent (Victory), was most effective for the treatment of PDD, once daily for 5 days, compared to other similar formulations and oxytetracycline.40
A nonantibiotic paste, Protexin Hoof-Care, containing formic acid (6.8%), acetic acid (3.74%), copper (3.29%), and zinc sulfate (0.40%), essential oils (peppermint/eucalyptus, 0.16% and a pH of 3.5 has been compared under controlled conditions with topical oxytetracycline and is considered an effective alternative to the antibiotic for the treatment of PDD.41 Only one topical application is required after cleaning the lesion. Advantages include no prescription is required, no withdrawal time is required, and it does not result in any concerns about antibiotic residue in meat or milk.
Bandaging the lesion
Whether or not the lesion should be bandaged after cleaning and medicating is controversial. Bandaging requires additional restraint to handle the leg and foot, it is labor intensive, and is an additional cost. However, field observations indicate topical treatment under a bandage is particularly effective with most cows showing remarkable improvement in 24–48 hours. Futhermore, when properly applied the bandage and the topical medication have the potential of reaching lesions in the interdigital cleft.
Footbaths
Footbaths containing antimicrobials and germicides have been used for treatment of groups of animals and for control of the disease. The most important benefit of using footbaths is that all animals are treated for PDD at the same time. Types of footbaths include walk-through and stand-in (stationary). The walk-through footbath, commonly located in milking-parlor exit lanes, is most popular in loose housing systems. Portable walk-through footbaths constructed of rubber, fiberglass, or hard plastic are also available and can be relocated as needed.42 The portable footbath is also the most convenient type for individual treatment situations that may involve bathing two, or possibly all four feet, for prolonged periods.
Most walk-through footbaths are at least 1 m wide, 2 to 3 m long, and 12 to 15 cm deep. Proper construction includes systems for efficient drainage, cleaning and refilling. The capacity of a rectangular footbath varies according to its dimensions, which can be calculated using the formula: width × length × depth 7.46 = capacity in gallons. (Multiplying the number of gallons by 3.8 will provide capacity in liters – US.) The size of the footbath needed will depend on the number of feet which will be treated with the system. Footbaths must be carefully monitored for excessive contamination with dirt and feces and solutions should be changed after every cow passes.34
The maximum number of cows which can be treated with a footbath varies according to the cleanliness of the cows, size of the bath, type and concentration of the medication used, housing system, weather conditions, and cow flow patterns. One recommendation suggests one footbath is sufficient for 150 to 200 cows and that PDD may be controlled with a single monthly passage through a footbath containing 5 to 10 g/L of oxytetracycline or 1 to 3 g/L of lincomycin in 200 L of water or erythromycin at a rate of 50 g/150 L of water.42 A common recommendation is that a foot bath can treat 150 to 200 cows per change of solution, used three times per week in outbreaks, once every week or two for maintenance.
Other observations however, suggest that as few as 30 to 50 cows through a footbath may cause major shifts in pH and solids loading, and the largest increment of change in pH occurred with the passage of the first 32 cows through the bath.
In summary, footbaths make biological sense for the treatment and control of PDD. However, most of the recommendations regarding their use, frequency of use, optimum number of cows, are based on uncontrolled field observations.
Antibiotics in footbaths
Antibiotics are commonly used in footbaths for the treatment and control of PDD.42 Antibiotics in footbaths are rapidly neutralized in the presence of excessive contamination from mud and manure. This is a significant limitation in large herds or in housing situations in which muddy conditions are present.
Tetracycline in 6–8 g/L of water has been used in a footbath for treatment of PDD. Tetracycline powder (324 g/lb) at 20 to 40 g per gallon (US) to deliver 0.5 to 1%. Lincomycin mix at 0.5 to 4 g/gallon US is also used. A mixture of lincomycin and spectinomycin in 150 g/200 L of water for treatment and 125 g/200 L of water for control was also effective. Walking cows through a footbath containing erythromycin, at a concentration of 35 mg erythromycin/L, after two consecutive milkings is effective.43 Four days after treatment, four of the measured signs (exudation, reddening, creaminess, and pain) were all significantly improved.
Most of the antibiotics used require a veterinary prescription and must be used according to specific recommendations and compliance with withdrawal periods as necessary.
Non-antibiotics in footbaths
Copper sulfate has been used widely in footbaths to control PDD but is considered ineffective.42 Copper sulfate and zinc sulfate combine with organic matter and become neutralized.
Formalin (37%) diluted to a 3–5% concentration has also been used extensively worldwide in footbaths for the control of lameness in cattle. However, it is not recommended for use in footbaths for the treatment of PDD.
A 7-day footbath of 6% formalin, 2% copper sulfate or 1% peracetic acid can be as effective in controlling PDD as erythromycin at 210 g/L for two days.44
In a dairy herd with heel-horn erosion, a foot bath containing acidic ionized copper through which the cows walked twice daily after milking for a total of 47 days was effective in controlling PDD.35
Treatment failure
The possible causes of treatment failure include inconsistent application of treatments and/or failure to periodically retreat all feet of all cows in the herd every 2 to 3 months with a topical spray, improper formulation of the medication, the neutralization of the antibiotics by manure, and the inaccessibility of the medication when the lesion is in the interdigital cleft.45 In addition, ideally lesions should be washed and cleaned thoroughly before applying the medication.
Protocol for evaluation of treatment
A uniform protocol to evaluate the results of treatment and control of PDD has been described.46 The protocol includes herd information including history and facilities, animal identification and production and clinical information about the lesions, the trial design which includes the duration of the trial, the positive and negative controls, how the lesions will be described and scored, the lameness score, the evaluation of pain, and the different treatments being evaluated.
CONTROL
Because the risk factors which predispose to the lesions are uncertain, specific environmental control strategies have not been examined using controlled field trials. Recurrence rates of PDD vary from 40 to 52% after 7 to 12 months.45 Thus it makes biological sense to have an infectious disease control system in place in the herd to provide optimum control.
• Housing, environment, and management. The high incidence of the disease in dairy cattle in drylot and freestall housing suggests that a high infection rate may be associated with high population density and contamination of bedding and the environment. Providing environmental conditions which promote clean and dry ground surfaces and bedding appears to be a logical strategy. Improving cow comfort by providing clean stalls, corrals, and alleys, dry and comfortable bedding, reducing the stocking rate, and improving ventilation to allow drying of stalls and alleys may decrease the incidence and severity of clinical cases. Hoof trimming, mobile tilt tables, and livestock trailers should be thoroughly cleaned and disinfected to prevent potential transmission of the agent of PDD.
• Biosecurity. For herds which are free of PDD, the most important control consideration is the purchase of herd replacements. According to the NAHMS survey of 1996 in the US, the odds of PDD infection were 8 times greater in herds which purchased replacements from outside sources compared with those that did not.45 Herd replacements should be purchased from herds known to be free of PDD. Quarantine procedures may be applicable but often impractical.
• Footbath. Successful control is possible by single passage of cattle through a footbath containing 5–6 g/L oxytetracycline or 150 g lincospectin 100 in 200 L water.47 For optimum results the heels of affected cows should be spray washed prior to entering the footbath. Repeating the footbath treatment in 4–6 weeks is recommended. Regular footbaths with 5% copper sulfate solution and formalin 3–5% solution once weekly, according to the incidence of the disease may be necessary in certain circumstances. Regular inspection of the feet of cattle is recommended to monitor the occurrence of the lesions.
• Vaccination. There is no evidence that a vaccine is effective for the control of PDD.45
Symposium on Digital Dermatitis. 8th International Symposium on Disorders of Ruminant Digits. Conference on Bovine Lameness, Banff, Canada. Cont Vet Education Program. Saskatchewan, Canada: University of Saskatchewan, Saskatoon, 1994, pp. 137–185.
Shearer JK, Elliott JB. Papillomatous digital dermatitis: treatment and control strategies: Part I. Compend Contin Educ Pract Vet. 1998;20:S158-S168.
Shearer JK, Hernandez J, Elliott JB. Papillomatous digital dermatitis: treatment and control strategies: Part II. Compend Contin Educ Pract Vet. 1998;20:S213-S223.
Berry SL, Read DH, Walker RL, Hird DW. Etiology, treatment and prospects for vaccination against papillomatous digital dermatitis. In: 12th International Symposium on Lameness in Ruminants. Orlando, Florida: Editor: JK Shearer; January 9–13 2002:5-11.
1 Walker RL, et al. Am J Vet Res. 1997;58:744.
2 Walker RL, et al. Vet Microbiol. 1995;47:343.
3 Choi BK, et al. Int J System Bacteriol. 1997;47:175.
4 Rijpkema SGT, et al. Vet Rec. 1997;140:257.
5 Dopfer D, et al. Vet Rec. 1997;140:620.
6 Collighan RJ, et al. Vet Microbiol. 2000;74:249.
7 Dhawi A, et al. Vet J. 2005;169:232.
8 Demirkan I, et al. J Med Microbiol. 2001;50:1061.
9 Rodriguez LA, et al. J Am Vet Med Assoc. 1996;209:1464.
10 Rodriguez LA, et al. Prev Vet Med. 1996;28:117.
11 Vermunt JJ, Hill FI. NZ Vet J. 2004;52:99.
12 Milinovich GJ, et al. Aust Vet J. 2004;82:223.
13 Wells SJ, et al. Prev Vet Med. 1999;38:11.
14 Brown CC, et al. Am J Vet Res. 2000;61:928.
15 Argaez-Rodriguez F, et al. Prev Vet Med. 1997;32:275.
16 Rodriguez-Lainz A, et al. Prev Vet Med. 1998;37:197.
17 Frankema K, et al. Prev Vet Med. 1993;17:137.
18 Rodriguez-Lainz A, et al. Prev Vet Med. 1999;42:87.
19 Hernandez J, et al. J Am Vet Med Assoc. 2000;216:1288.
20 Trott DJ, et al. J Clin Microbiol. 2003;41:2522.
21 Demirkan I, et al. Vet J. 1999;157:69.
22 Murray RD, et al. Res Vet Sci. 2002;73:223.
23 Stamm LV, et al. J Clin Microbiol. 2002;40:3463.
24 Edwards AM, et al. Microbiology. 2003;149:1083.
25 Green LE, et al. J Dairy Sci. 2002;85:2250.
26 Hernandez J, et al. J Am Vet Med Assoc. 2002;220:640.
27 Hernandez J, et al. J Am Vet Med Assoc. 2001;218:1611.
28 Blowey RW, et al. Vet Rec. 1994;135:117.
29 van Amstel SR. J South Afr Assoc. 1995;66:177.
30 Read DH, Walker RL. J Vet Diagn Invest. 1998;10:67.
31 Demirkan I, et al. Vet Microbiol. 1998;60:285.
32 Blowey RW, Done SH. Vet Rec. 1995;137:379.
33 Shearer JK, Elliott JB. In: Proc Int Symp Disorders Ruminant Digit, Banff, Canada, 1994, p. 182.
34 Britt JS, et al. J Am Vet Med Assoc. 1996;209:1134.
35 Manske T, et al. Prev Vet Med. 2002;53:215.
36 Britt JS, et al. J Am Vet Med Assoc. 1999;215:833.
37 Laven RA, Hunt H. Vet Rec. 2001;149:302.
38 Hernandez J, et al. J Am Vet Med Assoc. 1999;214:688.
39 Moore DA, et al. J Am Vet Med Assoc. 2001;219:1435.
40 Shearer JK, Herandez JJ. Dairy Sci. 2000;83:741.
41 Kofler J, et al. J Vet Med A. 2004;51:447.
42 Shearer JK, Elliott JB. Compend Contin Educ Pract Vet. 1998;20:S158.
43 Laven RA, Proven MJ. Vet Rec. 2000;147:503.
44 Laven RA, Hunt H. Vet Rec. 2002;151:144.
45 Shearer JK, et al. Compend Contin Educ Pract Vet. 1998;20:S213.
46 Britt JS, et al. Bov Pract. 1999;33:149.
47 Blowey RW. In: Proc Int Symp Disorders Ruminant Digit, Banff, Canada, 1994, p. 168.
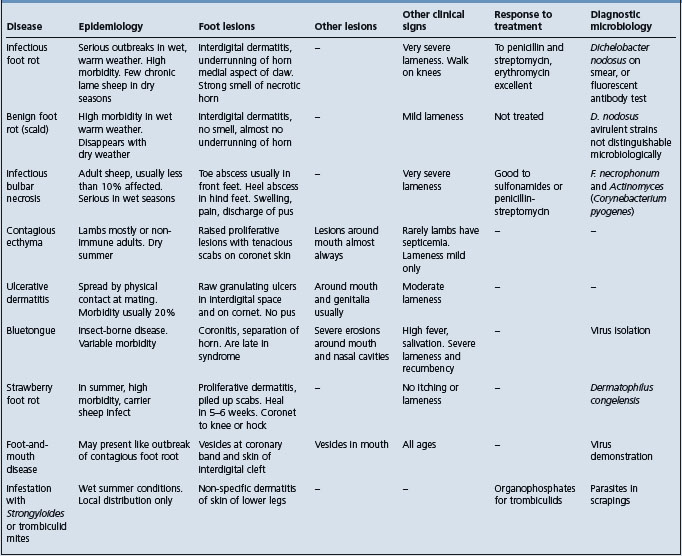
INFECTIOUS FOOT ROT IN SHEEP
Epidemiology
Source of infection is lesion discharge from other infected sheep. The survival of D. nodosus in the environment is short. Highly contagious disease with high attack rate in warm wet conditions. Lesions are present on both claws of the foot and commonly in more than one foot. Significant effect on productivity
Clinical findings
Inflammation of the skin at the skin–horn junction in the interdigital area with underrunning of the soft horn in benign (non-progressive) foot rot. Progresses to underrunning of the hard horn and inflammation of the sensitive laminae in virulent (progressive) foot rot and severe lameness
ETIOLOGY
Dichelobacter (Bacteroides) nodosus is the essential causal pathogen. It is a highly specialized organism in the small taxonomic group, the Cardiobacteriaciae. F. necrophorum aids D. nodosus in the invasion of the foot and contributes in the inflammatory reaction. Two other bacteria, a treponeme originally known as Spirochaeta (Treponema) penortha, and a motile fusiform bacillus, are commonly present in affected feet but are believed to have no primary etiological importance.
The type IV fimbriae of D. nodosus are recognized as a major virulence factor, are highly immunogenic, and provide the basis for the classification of D. nodosus strains into two major classes based on the genetic organization of the fimbrial gene region, with class I containing strains of serogroups A, B, C, E, F, G, I, and M and class II consisting of serogroups D and H. The serological diversity observed in the fimbriae is due to sequence variation in the fimbrial subunit protein and the fimbriae are the major immunoprotective antigens, although protection is serogroup-specific.1-4
Within this typing scheme there are strains that have major and minor prevalence in the disease. A strain-specific typing system based on genetic analysis of the fimA locus and combined with DNA sequencing is suggested for the characterization of D. nodosus strains.5
EPIDEMIOLOGY
Geographical occurrence
Foot rot of sheep is common in all countries where there are large numbers of sheep, except that it does not occur in arid and semi-arid areas unless the sheep have access to wet areas such as sub-irrigated swales.
Host occurrence
Sheep are the species principally affected but goats are also susceptible. Infection has been identified in farmed red deer,6 in cattle and is considered the cause of overgrown and deformed claws in wild mouflon in Europe.7 With environmental conditions of moisture and warmth, the disease in sheep has a high attack rate and a large proportion of a group of sheep can be affected within 1 to 2 weeks. Both claws of a foot and more than one foot (usually all) on the sheep will be affected. The disease is common and in high-risk areas the prevalence of infected flocks is high.
Source of infection
The source of infection of D. nodosus is discharge from the active or chronic infection in the feet of affected animals. The major reservoir of infection for virulent strains of sheep is other sheep as the isolates from cattle and deer generally produce the benign form of foot rot in sheep. The organism does not survive in the environment for more than a few days, 2 weeks at the most, but it can survive virtually indefinitely in lesions on chronically infected feet. Two classifications of foot rot have made based on the site of survival and perpetuation of the organism in a flock and the importance of this to control strategies:
1. Virulent foot rot (progressive foot rot) and intermediate foot rot– strains survive between foot rot transmission periods in pockets in previously underrun ovine hoof
2. Benign foot rot (non-progressive foot rot) – strains survive in the interdigital skin and the organism can be demonstrated in interdigital skin of a high proportion of asymptomatic sheep.8,9
Methods of transmission
Infection is usually introduced into a flock by the introduction of carrier sheep. Infection into a flock can also occur from the environment when foot rot-free sheep use yards, roads, or trucks that have been used by foot rot-infected sheep in the immediate past. Transmission has occurred to sheep held for 1 hour in a yard that 4 hours previously had contained a flock of sheep in which less than 1% had foot rot.10 Spread within a flock is facilitated by the flocking nature of sheep and heavy environmental contamination around communal drinking and feeding areas.
Host risk factors
Age
Foot rot occurs in sheep of all ages. In a flock outbreak, the age-specific incidence and severity of lesions in ewes increases with age.11 Older lambs have more severe lesions; young lambs and lambs from maiden ewes have less severe lesions than lambs from higher parity ewes. Prior natural infection does not engender immunity to a subsequent challenge that have had the disease. However, sheep do vary in their resistance or susceptibility to footrot infection. This appears to be, in part, immunologically mediated with the ability of some sheep to mount a strong T-cell response and to produce agglutinating antibodies to D. nodosus fimbriae an important factor in conferring resistance to severe infection.5
Breed
Sheep of the Merino type are the most susceptible to foot rot. British breeds, particularly Romney Marsh, are less susceptible and suffer from a milder form of the disease; they respond better to vaccination by suffering fewer subsequent attacks of foot rot but have worse reactions to the vaccination than Merinos.12 In the natural disease some animals never become infected, a few become infected but recover and most become infected and persist as chronic cases. There is developing evidence that this variation is genetically determined and selection for resistance, based on exposure to the disease and rigorous culling of affected individuals, has been demonstrated in the Targhee, Romney, Merino, Corriedale, and Perrendale breeds.13,14 Genetic markers can be identified and the SY1b histoglobulin may be associated with this resistance.13
Environmental risk factors
Climate
Moistness of the pasture and environmental temperature are major determinants for the transmission of foot rot. Conditions of wetness and warmth favor persistence of the bacteria in pasture and increase susceptibility of the feet to injury and dermatitis, thus facilitating spread of the disease from carrier sheep. The wetness has to be of considerable duration; short, heavy rainfalls are not significant – persistent rain over several weeks is, as there must be continued moistness on the ground for transmission to occur. Warmth is also required and moisture in winter in colder climates exerts no effect on foot rot – the daily mean temperature must be above 10°C (50°F) for transmission to occur. There is a linear relationship between the prevalence of farms with foot rot and yearly rainfall.15
Season
The requirement for both moisture and a warm environmental temperature determines that most serious outbreaks in sheep at pasture occur in the spring or in autumn and the transmission period is usually 4–6 weeks of each of these seasons. Transmission and outbreaks occur in winter in housed sheep when conditions underfoot are wet and in summer with sheep on irrigated pastures.
Management
Any practice that concentrates sheep in small areas will favor spread of the disease when environmental conditions favor transmission. Routine foot trimming may increase risk of infection and clinical disease.16
Failure to isolate introduced sheep until their foot rot status has been determined is a risk factor.
Pasture type
Foot rot is commonly associated with lush or improved pastures, irrigated pastures, and clover-dominant pastures. Long mature grass may result in interdigital abrasions as it is dragged through the interdigital space and facilitates infection, as may penetration of interdigital skin by barley grass seeds (Hordeum murinum).10,11 Skin penetration by larvae of the nematode Strongyloides spp. may also predispose to infection.
Pathogen factors
The major D. nodosus-encoded virulence factors that have been implicated in the disease are type IV fimbriae and extracellular proteases and the fimbrial subunit gene, fimA, is essential for virulence.
There is considerable variation in the virulence of strains of D. nodosus. Some produce benign foot rot whereas others produce deep lesions that facilitate their survival and confound eradication programs.17-19 As a result they have traditionally been subdivided into benign, intermediate, and virulent strains to conform with the types of clinical foot rot they are associated with in the field.20 The virulence of each strain depends on its keratinolytic capacity; virulent strains produce more extracellular protease, and an earlier production of elastase, than benign strains.20,21 The separation of the hard horn of the claw from the germinal layer, which is a characteristic of virulent foot rot, has been associated with infection with strains that produce a heat-stable protease with a single isoenzyme pattern, whereas benign strains have thermolabile protease.18,22 Infection associated with more than one strain is reported and up to five serogroups including up to eight strains have been reported from a single foot.5,19
Economic importance
Benign foot rot is generally considered to cause little if any economic effect and its occurrence is confined to the wet season. However, even benign foot rot infections are reported to depress body weight, wool growth, and wool quality in some countries.7
In contrast, virulent foot rot certainly results in severe loss of bodily condition and this, combined with a moderate mortality rate, a reduction in wool production, the disruption of the general routine of the farm, and the expense of labor and materials to treat the disease adequately, makes foot rot one of the most costly of sheep diseases. In addition there are welfare concerns, societal pressures encountered by owners of foot rot-infected flocks and, in control areas, the community costs of statutory foot rot control programs.
In controlled studies severe underrunning foot rot has resulted in an 11% depression in body weight and an 8% reduction in clean fleece weight of affected sheep.3 The magnitude of the loss can be related to the virulence of the infecting organism and the severity of the disease.
The effects on body weight are most severe during the active transmission period of the disease and there may be compensatory growth during the recovery period. Both greasy and clean fleece weight are significantly depressed by foot rot with a linear association between the extent of the depression and the severity of the disease. Wool fiber diameter is also decreased, which could partially compensate for fleece weight decrease in the final price for wool. Some sheep with severe disease may develop a break in the wool.
Feet of sheep affected with foot rot are attractive to blowfly strike. Necrotic exudate with accompanying maggots from affected feet can be deposited on the fleece to result in a focus of body strike.
PATHOGENESIS
Maceration of the interdigital skin from prolonged wet conditions underfoot allows infection with F. necrophorum. This initial local dermatitis associated with infection with F. necrophorum at the skin and the skin–horn junction may progress no further, but the hyperkeratosis induced by this infection facilitates infection by D. nodosus if it is present. The preliminary dermatitis has been named ‘ovine interdigital dermatitis’ and is also called ‘foot scald’.
Ovine interdigital dermatitis, foot scald
This disease is commonly seasonal and occurs when moist conditions under-foot, or trauma from pastures or frost, produces maceration of the interdigital skin and allows invasion by Fusobacterium necrophorum, a ubiquitous organism in feces and soil.
Lesions are in the interdigital space where there is hyperemia, or swelling and blanching, and wetness of the interdigital skin. There is no, or only minimal, separation at the skin–horn junction. Lambs are more commonly affected, particularly in spring, but the disease can involve all ages of sheep. Most or all feet on a sheep are affected and it is present in a large proportion of the age cohort of the flock.
In Australia and New Zealand the disease is not usually associated with lameness and is often found incidentally when examining sheep for other reasons.
In Britain it is reported as a common cause of lameness23 but this might reflect a lack of cultural differentiation from the less virulent forms of infectious foot rot that can present with identical clinical findings.24,25
Control is by avoiding grazing lambs on long grass and where there are muddy conditions. The disease will regress spontaneously when the pasture dries up but can be treated with topically applied oxytetracycline or by walking through a foot bath containing 3% formalin or standing (10 minutes) in one containing 10% zinc sulfate.
Ovine interdigital dermatitis can predispose to infectious foot rot, or to foot abscess.
Benign and virulent foot rot
It is assumed that the pili of D. nodosus facilitate attachment of the organism to the epithelium of the foot. When the feet of sheep with interdigital dermatitis are colonized with a strain of D. nodosus that has little keratinolytic ability, there is underrunning of the soft horn but no further progression and this infection has been given the name of benign foot rot or non-progressive foot rot. Benign foot rot cannot be easily distinguished on clinical examination from ovine interdigital dermatitis. Colonization with keratinolytic and virulent strains leads to the clinical disease of virulent foot rot. The underrunning lesion is the result of keratolytic activity and the associated inflammation is a consequence of a combined activity of D. nodosus and F. necrophorum. A designation of intermediate foot rot has been used by some to provide a mid-classification of severity between benign and virulent foot rot and the classification of infected sheep into these categories is based on a foot lesion scoring system (see below). There is much difficulty in specifying exactly the characteristics of these clinical forms in natural outbreaks, in part because commonly more than one strain is involved, the more so because the severity of the lesions is also modulated by geographic and climatic conditions.8-1126
CLINICAL FINDINGS
Sheep
Virulent foot rot
In a flock, a sudden onset of lameness of several sheep is the usual presenting sign of foot rot as the disease is not detected before this occurs. The pain associated with infection is severe and affected sheep will limp or carry the affected leg. Usually more than one foot is affected and affected sheep may graze on their knees.
On close examination the earliest sign of virulent foot rot is swelling and moistness of the skin of the interdigital cleft and a parboiled and pitted appearance at the skin–horn junction in the cleft. This inflammation is accompanied by slight lameness which increases as necrosis underruns the horn in the cleft. The underrunning starts as a separation of the skin–horn junction at the axial surface just anterior to the bulb of the heel and proceeds down the axial surface and forwards and backwards. There is destruction of the epidermal matrix beneath the hard horn, which is subsequently separated from the underlying tissues. In severe cases both the axial and the abaxial wall and the sole are underrun and deep necrosis of tissue may lead to the shedding of the horn case. The separation may not be obvious on superficial visual examination but can be detected with a knife blade or by paring of the feet. There is a distinctive, foul-smelling exudate, which is always small in amount. Abscessation does not occur.
Both claws of the one foot will be involved and commonly more than one foot is involved. When extensive underrunning has occurred lameness is severe. A systemic reaction, manifest by anorexia and fever, may occur in severe cases. Recumbent animals become emaciated and may die of starvation. Secondary bacterial invasion and/or fly strike may result in the spread of inflammation up the legs.
Benign foot rot
Benign foot rot is manifest with interdigital lesions, a break at the skin–horn junction and separation of the soft horn, but the disease does not progress beyond this stage to severe underrunning of the hard horn of the foot. The interdigital skin becomes inflamed and covered by a thin film of moist necrotic material; the horn is pitted and blanched.
It is difficult to distinguish between an established infection with benign foot rot and the early stages of virulent foot rot. With virulent foot rot it is common to find all stages and severity of the disease in the same flock. A large number of sheep should be examined in order to differentiate benign from virulent foot rot and it may be necessary to re-examine the flock after a period of time to determine if the disease has progressed to the virulent type.
Scoring systems
Several scoring systems, based on severity and persistence, have been devised to aid in epidemiological and control programs and can be used to categorize the severity of disease, and the virulence of associated strains of D. nodosus, within a flock.27 In the Australian system, a score of 0 to 4 is allocated to each foot but the score is used to classify the flock or a cohort, as to the severity of disease. Feet scored 0 have no evidence of necrosis or inflammation or cleavage of horn. Scores of 1 and 2 are confined to sheep with interdigital lesions whereas a gradation of scores from 3 to 4 reflect progressive underrunning and separation of horn from the underlying laminar with 3 representing underrunning of the soft horn. In this score system benign foot rot cases score 0 with some allowance for scores of 3. Intermediate foot rot in a cohort is defined when 1 to 10% of the cohort have a score of 4. Virulent foot rot is defined by greater than 10% of the cohort with a score of 4.28 It has been suggested that the score system is not valid as the severity of disease can be influenced by climate but one study suggests that score systems are strain specific rather than climate specific. It found that a cohort of sheep with scores defining intermediate foot rot maintained that clinical classification when moved to a climate that would promote the development of more severe disease.28
Symptomless carriers
These carriers may be affected for periods of up to 3 years. Most such animals have a misshapen foot and a pocket of infection beneath underrun horn can be found if the foot is pared. A less common form of the chronic disease is an area of moist skin between the claws without obvious involvement of the claw.
Goats
Foot rot is associated with D. nodosus and is manifested by severe interdigital dermatitis; there may be some separation of the skin–horn junction at the axial surface but rarely is there an underrunning of the horn of the sole or the abaxial surface of the foot as in sheep.29
Cattle
Infection with D. nodosus is also associated primarily with a severe interdigital dermatitis and there may be lameness. There is fissuring and hyperkeratosis of the interdigital skin with pitting and erosion at the skin–horn junction in the cleft. There is also fissuring, pitting, and erosion on the horny bulbs of the heel. There may be underrunning at the heel but it is usually minimal.
Contagious ovine digital dermatitis (CODD)
CODD is an apparent new disease recently described in the UK.30,31
It is manifest with severe, rapidly spreading, lameness and is more common in adults than lambs. Commonly there is a history of poor response to conventional methods of footrot control. The initial lesion is a proliferative or ulcerative lesion at the coronet, with subsequent extensive under-running of the hoof horn and, in some cases, complete separation. Interdigital lesions are absent. It may affect only one claw on one foot. A spirochete, morphologically different from Spirochaeta penortha but with similarities to the spirochete associated with digital dermatitis (hairy foot wart) in cattle has been isolated.31,32
F. necrophorum and D. nodosus may also be present in some flocks but their contribution, at present, is unclear.
Response to formalin and zinc sulfate footbaths is poor but the disease is reported to respond to topical lincomycin and spectinomycin solutions.30
CLINICAL PATHOLOGY AND NECROPSY FINDINGS
In countries that do not have a large sheep industry identification of D. nodosus in most laboratories, is made by examining a smear made of exudate taken from underneath some underrun horn at the advancing edge, and stained with Gram’s stain. This is potentially inaccurate and to increase the validity of a decision, a fluorescein-stained antibody can be used. A highly sensitive polymerase chain reaction (PCR) can be used.20,33 PCR tests can be used to detect gene sequences and identify particular strains and are used in eradication schemes in Australia.17,19
Examinations of protease type, gene probe category and nucleotide sequence to differentiate virulent from benign strains are used in regions with eradication programs for virulent foot rot and in epidemiological studies.20,21 Of the protease tests, the gelatin gel protease thermostability test allows clear-cut and rapid differentiation between virulent and benign strains.34
Serum antibody following natural infection develops as early as 2 weeks after infection and the level obtained is proportional to the severity of the early clinical disease. The antibody response is not long lasting and falls to pre-infection levels within a few months after resolution of the foot lesions.35,36 An ELISA for serological detection of infected sheep has some value for diagnosis of flock infection but lacks specificity and is inaccurate in older sheep.35
Diagnosis of virulent foot rot is clinical, is based on a whole flock approach, and postmortem examination is not necessary.
Because of the rigorous control measures required sometimes by law to control or eradicate infectious foot rot of sheep, it is imperative that the diagnosis be made with great care. The greatest problem is in the identification of carrier sheep in the non-transmitting periods. A number of conditions may be confused with foot rot, especially when they occur under the same environmental conditions (Table 20.1).
Foot abscess – a major differential and can be present in several sheep in a flock at the same time. It usually affects only one foot, is not contagious and is characterized by extensive suppuration. The abscess occurs in a single claw on the foot and there is obvious local heat and pain on palpation.
Contagious ovine digital dermatitis.
Shelly toe – the name given to a condition where there is separation of the wall of the foot in Merino sheep and occasionally in other breeds on improved pasture. The abaxial wall of the hoof separates from the sole near the toe and the crevice formed becomes packed with mud and manure. The hoof in the region is dry and crumbly. The cause is not known but it is probably a form of laminitis.
Suppurative cellulitis – associated with F. necrophorum, commences as an ulcerative dermatitis of the pastern above the bulb of the heel, and extends up the leg to the knee or the hock and more deeply into SC tissues.
TREATMENT AND CONTROL
The time-honored method of treatment of foot rot has been the application of topical bactericidal agents to the foot. These are most effective early in the transmission period, prior to any extensive underrunning of the horn. When there is underrunning of the horn, the underrun horn must be removed so as to expose the infection to the topical agent. This approach has been used successfully for treatment, control, and eradication purposes. However, it requires extensive paring of affected feet, which is labor intensive, time consuming and distressing both to the operator and to the sheep. Recent years have seen the advent of treatment methods that minimize the need for extensive paring of the feet. These include the use of topical zinc sulfate, the use of parenteral antibiotics and the recognition of vaccination as an adjunct to treatment as well as a strategy for control.
Topical treatment
Most topical treatments require that all underrun horn be carefully removed so that the antibacterial agent to be applied can come into contact with infective material. This necessitates painstaking and careful examination, and paring of all feet. Incomplete paring will leave pockets of infection. With severely underrun feet, it is impossible to expose all of the underrun areas without causing hemorrhage as it may be necessary to remove all of the sole and the wall of the claw. Very sharp instruments including a knife and hoof secateurs are necessary to do the job properly and they should be disinfected after each use. The parings should be collected and burned. Sheep restraint cradles lessen the labor of paring of the feet.
With small numbers of sheep topical treatment may be applied by brush, by spray, or aerosol. Topical treatments are likely to be washed off the feet and their efficacy under wet conditions is a determinant in selecting the agent to be used. Local applications include chloramphenicol (10% tincture in methylated spirits or propylene glycol), oxytetracycline (5% tincture in methylated spirits), cetyltrimethyl ammonium bromide or cetrimide (20% alcoholic tincture), zinc sulfate (10% solution), copper sulfate (10% solution) and dichlorophen as a 10% solution in either diacetone alcohol or ethyl alcohol.
Chloramphenicol is expensive but efficient under both wet and dry conditions. Its use with food animals is prohibited in many countries. Oxytetracycline must be used as a 5% tincture for optimum results and is not as efficient as chloramphenicol under wet conditions, but gives excellent results when the weather is dry. Cetrimide is a relatively cheap product and appears to be as effective as chloramphenicol under all conditions. It is possible that in different countries with different climates and environmental conditions, the efficiency of particular treatments will vary. When only a few sheep are affected, bandaging may help maintain local concentrations of the topical preparation. In general, parenteral treatment with antibiotics, without paring of the feet, has replaced topical antimicrobial treatment of individual or small numbers of sheep.
Footbathing for treatment and control
Footbathing is a more practical approach to topical treatment, and for control during transmission periods, when dealing with large numbers of sheep. All sheep are passed through the footbath but it is usual to divide the flock into affected and unaffected groups by examination prior to the initial footbathing, and to pasture them separately following footbathing to minimize subsequent infection of uninfected sheep. After several footbaths and inspections the majority of the flock should be free of infection and the residual infected sheep are culled.
Preparations suitable for footbaths include 5% copper sulfate, 5% formalin and 10% zinc sulfate with or without a surfactant to aid wetting of tissues. Regardless of the agent used, it is recommended that the sheep be kept standing on concrete, on wooden slats, or on dry ground for a few hours after treatment.
The relative merits and disadvantages of the various preparations used are as follows.
Copper sulfate solution (5%)
Copper sulfate solution colors the wool, deteriorates with pollution, corrodes metal, and may cause excessive contamination of the environment with copper. The feet of the sheep must be pared and all infected areas exposed before treatment. Copper sulfate footbathing appears to harden the horn, which can be an advantage but also a disadvantage if further paring is required at a later date. A patented copper salt preparation (not copper sulfate) has efficacy in treatment without the disadvantages of copper sulfate.37
Formalin solution (5%)
Formalin solution does not deteriorate with pollution but causes extreme discomfort to sheep that have heavily pared feet and its use for sheep with severe lesions is discouraged on humane grounds. The feet of the sheep must be pared and all infected areas exposed before treatment. Formalin is unpleasant to work with in enclosed areas, has toxicity for humans and its use may be banned in some countries for this reason. Sheep should be passed through the footbath weekly for 4 weeks or weekly during periods of high risk for transmission.
The use of solutions containing more than 5% formalin or dipping at intervals of less than 1 week may cause irritation of the skin. Farmers can be neglectful in maintaining proper concentrations of formalin in footbaths, and frequent use of the bath combined with hot weather can result in a concentration of 30% formalin. Such concentrations cause extensive cellulitis around the coronets, and a high proportion of animals may be so badly affected that they need to be destroyed. The safest precaution is to empty the vat and prepare a new mixture.
Zinc sulfate solution (10–20%)
Zinc sulfate solution is as effective as formalin, more pleasant to use and is generally the preferred topical chemical for the treatment of foot rot.38 It has a superior ability to penetrate the hoof horn which is enhanced by the addition of the surfactant sodium lauryl sulfate to the footbath solution.39 Significant cure rates can be achieved without prior paring of the feet which removes a significant labor cost from the treatment and control of the disease.38-40
Cure rates, without paring, are higher in sheep that have moderate rather than severe lesions in their feet. Some paring of chronically affected, overgrown feet may be required to allow the treatment access to pockets of infection in the anterior aspects of the sole, and also of feet that have underrunning that has progressed to the abaxial area of the digit. Sheep are stood for 1 h in a footbath containing a 10 to 20% zinc sulfate solution with 2% sodium lauryl sulfate with sufficient depth to cover the coronet. The treatment is repeated in 5 d and after a further 21 d the sheep are individually examined to determine their status and are retreated or culled depending upon the strategy of treatment and control in the flock. Zinc sulfate footbathing can provide protection to the foot against reinfection for periods of at least 2 weeks41 and can be used effectively during periods of active spread of the disease42 Repeated daily footbathing (10 min each day for 5 days) in a zinc sulfate solution with surfactant has eradicated (as opposed to controlled) virulent foot rot in sheep associated with some strains but was ineffective against a strain that produced severe underrunning.17
Thirsty sheep that drink from the footbath may die of acute zinc poisoning.
Antibiotic treatment
Foot rot can be treated with antibiotics without the necessity of paring of the feet. Treatment is considerably more effective if done during dry periods, and when the sheep are kept on dry floors for 24 h after treatment, as in wet conditions the concentration of antibiotic at the tissue level is much reduced.
D. nodosus is susceptible in vitro to penicillin, cefamandole, clindamycin, tetracycline, chloramphenicol, erythromycin, sodium cefoxitin, tylosin tartrate, nitrofurazone, tinidazole and has the least susceptibility to sulfonamides and the aminoglycosides. However, in vitro tests may have little relevance to field application due to differences in the penetrance of antibiotics to the affected part of the foot.
Antibiotics and their dosage shown effective against virulent foot rot are:
• Penicillin/streptomycin. Single IM dose of 70 000 U/kg procaine penicillin and 70 mg/kg dihydrostreptomycin43
• Erythromycin. Single IM dose of 10 mg/kg44,45
• Long-acting oxytetracycline. Single IM dose of 20 mg/kg44,46,47
• Lincomycin/spectinomycin. Single SC dose of 5 mg/kg lincomycin and 10 mg/kg spectinomycin.48
Cure rates in sheep kept in a dry environment, preferably a shed, for 24 h following treatment are approximately 90% with all except penicillin/streptomycin, which may be slightly less efficacious.49 Cure can occur even in severely affected sheep and extensive paring prior to treatment does not improve cure rates.46,49 Cure rates fall to 60% if sheep are in a wet environment following treatment and in wet conditions cure rates improve slightly if sheep are footbathed at the time of antibiotic administration.47,49
Following treatment and holding, sheep are moved to a ‘clean’ dry pasture and inspected for cures 3–4 weeks later when clinically affected animals are culled. Reinfection will occur in foot rot spread periods and, in general, the use of antibiotics for control of foot rot should be confined to the summer period when there is no spread.26
All treatments are ‘off label’ and some are not approved for use on sheep in some countries. Choice may be determined by the withholding period required before culled sheep can be sent to market. Lincomycin/spectinomycin in sheep may precipitate severe outbreaks of salmonellosis in some flocks.49 Antibiotics are particularly valuable for the treatment of late-pregnant sheep that develop foot rot, where more drastic treatment could lead to problems such as pregnancy toxemia.
Vaccination
Vaccination against foot rot can significantly increase short-term resistance to infection and is an important component of control strategies, especially in circumstances where climate and management practices make other control strategies difficult to apply. Vaccination also shortens the clinical course in infected sheep and can be used as a treatment strategy. In neither case is vaccination 100% effective.
Pilus antigens are the major host-protective immunogens and confer protection against challenge with homologous strains. Immunity is associated with circulating antibody but high levels of pilus-specific circulating antibody are required for adequate diffusion into the epidermis and protection against the disease. In groups of sheep, resistance is associated with the acquisition of flock mean agglutination titers of greater than 5000; there is a positive correlation between antibody titer and foot rot resistance in the first few months following vaccination. Immunity can be passively transferred with gammaglobulins from immunized sheep to naive recipients and from vaccinated ewes to their lambs via colostrum to provide protection for the first 8 weeks of life.24,25
Vaccines must contain adjuvants for an adequate antibody response and vaccination with oil-adjuvant vaccines is accompanied by significant local reaction, including local swelling at the injection site and abscessation in a proportion of animals.50 This reaction is more severe in British breeds of sheep than in Merinos. In milk goats and sheep, vaccination can result in a significant drop in milk production. The potential gains from vaccination need to be weighed against this effect in a decision to use vaccination as a method of control rather than other control procedures.
Commercial vaccines have contained up to 10 strains of D. nodosus representative of the most common serogroups associated with foot rot. The extent to which these vaccines will give protection or will promote earlier cure depends upon the relationship of the vaccine pilus types to those associated with the foot rot problem. Vaccine failure is generally attributable to the occurrence of foot rot associated with strains of the organism not present in the vaccine or to which there is no cross-protection, and to individual animal variation in response.1,25,51,52 However, more recently it is recognized that there is a limitation to the number of strains that can be incorporated in current vaccines due to antigenic competition.51,53 Mixing different D. nodosus fimbriae in vaccines may lead to inadequate host responses to individual antigens and an alternative approach is to identify D. nodosus strains present in a given geographical region enabling the development of optimized and localized strain(s)-specific vaccines.5,54
Field trials have shown a wide variation in the therapeutic effect of vaccination with a reduction in foot rot incidence varying from 27–54% in sheep where there was no routine foot care to 69–91% in flocks in which vaccination was coupled with routine foot care such as trimming and footbathing.12,25,53,55 The effect is to reduce both the incidence, severity, and duration of the infection. The improvement seems to be due to accelerated healing of the lesions with some protection against reinfection. For an optimum effect, two vaccinations are required and the duration of this effect depends upon the adjuvant and the breed of sheep. Even so the duration of the protection is limited to 4–12 weeks in most studies. It can be very effective in control when coupled with a culling policy of sheep that remain clinically infected.54
Whereas vaccination is of value for control of an existing flock infection, it may not be economic to use vaccination as a strategy to prevent infection of a foot rot-free flock.25,55 The use of foot rot vaccines may be prohibited in areas where there are foot rot eradication programs.
Summary of control procedures in infected flocks
The objective of foot rot control in an infected flock is to maintain the prevalence of disease at a low level by reducing the incidence of new infections, to prevent the development of the debilitating and painful underrunning lesions of advanced foot rot, and to achieve this with strategies that are based on whole-flock control with minimal need for individual handling of individual animals. This approach is most likely to be adopted by sheep owners.56 Routine footbathing, especially during transmission periods, coupled with vaccination can achieve this and sheep that are affected with severe underrunning are treated with parenterally administered antibiotics or culled.
Genetic selection
Whereas there are breed effects on susceptibility and an apparent high heritability coefficient for resistance,13,14 genetic selection for resistance has not advanced sufficiently to be used practically in control programs. Resistance can be determined by direct challenge of a candidate ram.14 Antibody response to vaccination cannot be used as a surrogate.
ERADICATION
Eradication of virulent foot rot is a desirable but not always feasible objective but it can be accomplished in climates where the transmission periods are short. Where rainfall is heavy and the ground moist most of the year, much greater difficulty may be encountered. Area eradication of virulent foot rot is a much more daunting task but is proceeding satisfactorily in several regions in Australia. Eradication of benign foot rot is not justified economically, nor is it possible with current knowledge as benign foot rot strains can be carried in the interdigital skin of asymptomatic sheep.8,9
Eradication programs are based on the following facts:
1. D. nodosus persists in the flock in infected feet
2. Infection in the foot can be detected and the infected sheep cured or culled
3. The organism does not persist in pasture for long periods and does not transmit in dry periods.
Fields kept free of sheep for 14 d can be considered free of infection. If all infected animals are culled or cured and infection removed from the pasture, eradication is achieved. Eradication of the disease should be undertaken during a dry summer season, but active measures must be taken to reduce the incidence of infection and spread during the transmission period in the preceding spring. Vaccination and footbathing may be part of this strategy.
In the eradication phase in the non-transmission period, all feet of sheep are examined and affected or suspicious sheep are segregated. When examinations are carried out during dry weather, the feet are likely to be hard and the disease at a quiescent stage. In such circumstances minor lesions may be missed, necessitating an extremely careful trimming and examination of all feet. Clean sheep are run through a foot bath (5% formalin or 10% zinc sulfate) and put into fresh fields, while the affected are isolated and treated with antibiotics and/or footbathing with one of the preparations described earlier. Footbathing treatments must be repeated weekly. Sheep that do not respond may be treated intensively, for example two 1-hour soaks in 10% zinc sulfate,30 but preferably should be culled. In areas where flocks are small and there are insufficient fields to carry out this program completely, it has been found to be sufficient to treat all affected sheep weekly but to put all affected sheep back in the flock and the flock back onto the infective pasture, providing conditions are dry. Eradication of virulent foot rot from sheep and goats in an area of Nepal by a combination of treatment with parenteral antibiotics and a type-specific vaccine, routine vaccination with the type-specific vaccine and culling of non-responders is reported.54
Culling is an important strategy in foot rot eradication and if the number of infected sheep is small, immediate culling may be the most economic strategy. Culling can be an important strategy in the eradication of disease associated with moderately virulent strains if it is conducted in the climate periods of low transmission.58
Introduced sheep
Most breakdowns in eradication occur because of inefficient examination and treatment or the introduction of affected sheep without first insuring that they are free from the disease. Introduced sheep should be run as a separate group from the main flock until they have been proven to be foot rot-free in a climatic transmission period. Similar isolation of introduced sheep should also be practiced in flocks free of the disease. It should also be a management practice in flocks that have disease to minimize the risk of introduction of different strains of the organism.
Beveridge WIB. Footrot in sheep: a transmissible disease due to infection with Fusiformis nodosus. Bull Counc Sci Indust Res Aust. 1941;140:1-56.
Egerton JR, et al. Footrot and Foot Abscess of Ruminants. Boca Raton, FL: CRC Press, 1989.
Kimberling CV, Ellis RP. Vet Clin North Am. Food Anim Pract. 1990;6:671.
Stewart DJ, Claxton PD. Ovine footrot: Clinical diagnosis and bacteriology. Australian Standard Diagnostic Techniques for Animal Diseases. 1993:1-27.
Winter A. Lameness in sheep. 1 Diagnosis. In Practice. 2004;26:58-63.
Winter A. Lameness in sheep. 1 Treatment and control. In Practice. 2004;26:130-139.
Abbot KA, Lewis J. Current approaches to the management of ovine footrot. Vet J. 2005;169:28-41.
1 Stewart DJ, et al. Aust Vet J. 1991;68:50.
2 Katz ME, et al. J Gen Microbiol. 1991;137:2117.
3 Olson ME, et al. Can J Vet Res. 1998;62:33.
4 Kennan RM, et al. Vet Microbiol. 2003;92:169.
5 Zhou H, Pickford JGH. Vet Microbiol. 2000;71:113.
6 Skerman TM. NZ Vet J. 1983;31:102.
7 Nattermann H, et al. Monatschft Vet Med. 1991;46:471.
8 Glynn T. Aust Vet J. 1993;70:7.
9 Depiazzi LJ, et al. Aust Vet J. 1998;76:32.
10 Whittington RJ. Aust Vet J. 1995;72:132.
11 Woolaston RR. Aust Vet J. 1993;70:365.
12 Stewart DJ, et al. Aust Vet J. 1985;62:116.
13 Raadsma HW, et al. Wool Technol Sheep Breed. 1990;38:7.
14 Raadsma HW, et al. J Anim Breed Genet. 1995;112:349.
15 Locke RH, Coombes NE. Aust Vet J. 1994;71:348.
16 Wassink GJ, et al. Vet Rec. 2003;152:351.
17 Depiazzi LJ, et al. Aust Vet J. 2003;81:58.
18 Depiazzi LJ, et al. Vet Microbiol. 1991;26:151.
19 Jelinek PD, et al. Aust Vet J. 2000;78:273.
20 Lui D, Wong WK. Vet J. 1997;153:99.
21 Moore LJ, et al. Vet Microbiol. 2005;108:57.
22 Palmer MA. Vet Microbiol. 1993;36:113.
23 Wassink GJ, et al. Vet Rec. 2004;154:551.
24 Emery DL. Footrot and Foot Abscess of Ruminants. Boca Raton, FL: CRC Press, 1989;141.
25 Stewart DJ. Footrot and Foot Abscess. Boca Raton, FL: CRC Press, 1989;167.
26 Allworth B. Proceedings of the 2nd Pan Pacific Veterinary Conference, 1996; 159.
27 Links IJ, Morris S. Vet Microbiol. 1996;51:305.
28 Abbott KA, Egerton JR. Aust Vet J. 2003;81:756.
29 Whittington RJ, Nichols PS. Res Vet Sci. 1995;58:26.
30 Wassink GL, et al. Vet Rec. 2003;152:504.
31 Demirkan I, et al. Med Microbiol J. 2001;50:1061.
32 Collinhan RJ, et al. Vet Microbiol. 2000;74:249.
33 Claxton PO, O’Grady KC. In: Proceedings of a CSIRO Workshop. Footrot of Ruminants, Melbourne, 1985.
34 La Fontaine S, et al. Vet Microbiol. 1993;35:101.
35 Whittington DJ, Nichols PJ. Res Vet Sci. 1995;59:128.
36 Whittington RJ, et al. Vet Microbiol. 1997;54:255.
37 Reed GA, Alley DU. Aust Vet J. 1996;74:375.
38 Kimberling CV, Ellis RP. Vet Clin North Am. 1990;237:671.
39 Stewart DJ. Footrot of Sheep. Boca Raton, FL: CRC Press, 1989;5.
40 Malecki JC, Coffey L. Aust Vet J. 1987;64:301.
41 Bulgin MS, et al. J Am Vet Med Assoc. 1986;189:194.
42 Marshall DJ, et al. Aust Vet J. 1991;68:186.
43 Hinton DJ. Aust Vet J. 1991;68:118.
44 Webb-Ware JK, et al. Aust Vet J. 1994;71:88.
45 Rendell DK, Callinan APL. Aust Vet J. 1997;75:354.
46 Jordon D, et al. Aust Vet J. 1996;73:211.
47 Grogonothomas R, et al. Br Vet J. 1994;150:561.
48 Venning CM, et al. Aust Vet J. 1990;67:258.
49 Vizzard AL. Proceedings of the 2nd Pan Pacific Veterinary Conference, 1996; 163.
50 Walduck AK, Opdebeeck JP. Aust Vet J. 1996;74:451.
51 Hunt JD, et al. Vaccine. 1994;12:457.
52 Gradin JL, et al. Am J Vet Res. 1993;54:1069.
53 Stewart DJ, et al. Vet Microbiol. 1991;27:283.
54 Egerton JR, et al. Vet Rec. 2002;151:290.
55 Lewis RD, et al. J Anim Sci. 1989;67:1160.
56 Abbott K, et al. Vet Rec. 2003;152:510.
FOOT ABSCESS IN SHEEP
Foot abscess includes two diseases: heel abscess and toe abscess, which will be discussed in turn.1
HEEL ABSCESS/INFECTIOUS BULBAR NECROSIS
Heel abscess is a sequel to damage to the interdigital skin. This damage may result from physical damage from sharp stones or stubble, or friction in the area produced by overgrown feet, but most commonly results as an extension from ovine interdigital dermatitis into the soft tissues of the heel and is associated with Fusiformis necrophorum and Arcanobacterium (Actinomyces; Corynebacterium) pyogenes.2 In interdigital dermatitis, the organisms can invade deep into the interdigital skin. The joint capsule of the distal interphalangeal joint is extremely vulnerable to invasion on the axial interdigital area and this leads to abscessation.
Most flocks experience cases of heel abscess but the yearly incidence is usually less than 1%. Heel abscess occurs mainly during very wet seasons as does foot rot but the former is limited largely to adult sheep, especially ewes heavy in lamb, and rams. Interdigital dermatitis and heel abscess are frequently present in the flock at the same time. An increased prevalence in a flock of young rams may be due to close flocking and to increased muddying of pasture due to this high concentration of livestock.1 Usually only one foot and one claw is involved, although in severe outbreaks all four feet may become affected. Most commonly, the medial claw of the hindfoot is affected.
In the initial stages the affected digit is hot and painful. There is an acute lameness – the affected sheep holds the foot off the ground while walking. There is swelling and inflammation of the interdigital skin and pain on pressure across the heel. Pressure in this area may result in the discharge of pus from sinuses in the interdigital space. When the phalangeal joints are involved there is severe swelling at the back of the claw and the infection may extend to break out at one or more points above the coronet with a profuse discharge of pus.
Treatment of heel abscess
The treatment of foot abscess is by surgical drainage, parenteral treatment with sulfonamides or a combination of penicillin and streptomycin and the application of a local bandage. Therapy should be continued for several days. Recovery is not rapid. Because of the frequent involvement of the distal interphalangeal joints3 with heel abscess, treatment with antibiotics without surgical intervention is unlikely to be successful but some cases heal spontaneously in 6–8 weeks.
TOE ABSCESS
Toe abscess is a lamellar suppuration with purulent underrunning of the horn at the toe. It results from damage to the sole, white line, or wall of the foot and is a common sequel to overgrown feet. Most commonly, it involves a digit on the front feet. The affected digit is hot and there is pain on pressure across the sole and toe. There is severe lameness, swelling of the coronet with pain and heat apparent, and usually rupture and purulent discharge at the coronet between the toes. Penetration to deeper structures may also occur.
Treatment of toe abscess
The only treatment of toe abscess is by surgical drainage and the response is rapid. Toe granuloma can be a sequel but more commonly toe granuloma is a response to overzealous foot paring.4