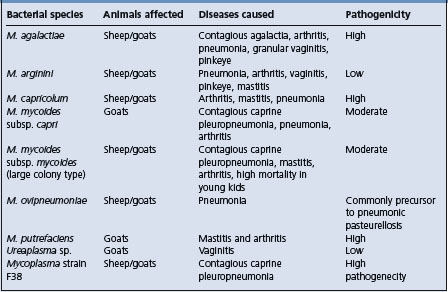
Diseases associated with Mycoplasma spp.
The genera Mycoplasma, Acholeplasma and Ureaplasma form the family Mycoplasmataceae within the class Mollicutes. (Mollicutes: A class of Gram-negative bacteria consisting of cells bounded by plasma membrane. Its organisms differ from other bacteria in that they are deficient in cell walls. It contains a single order, Mycoplasmatales. Mollicutes: mollis = soft, cutis = skin.) More than 200 mollicutes, including 102 Mycoplasma species, have been named, although many more as yet unnamed species have been isolated.1 A summary of the major Mollicutes of farm animals and the diseases associated with them is shown in Table 20.5.2
Table 20.5 Major pathogenic Mycoplasmas of ruminants, swine, and horses
| Animal host/Mycoplasma species | Disease |
|---|---|
| Bovine | |
| M. mycoides subsp. mycoides SC | Contagious bovine pleuropneumonia, CBPP |
| Mycoplasma sp. bovine group 7 | Pneumonia and arthritis |
| M. bovis | Mastitis, pneumonia (calf), polyarthritis (calf) |
| metritis, abortion, sterility | |
| M. dispar | Pneumonia (calf) |
| M. californicum | Mastitis |
| M. canadense | Mastitis |
| M. bovocculi | Conjunctivitis |
| Ureaplasma diversens | Metritis, sterility, abortion |
| Mycoplasma (Eperythrozoon) wenyonii | Anemia |
| Sheep and goat | |
| M. capricolum subsp. capripneumonia | Contagious caprine pleuropneumonia |
| M. capricolum subsp. capricolum | Mastitis, arthritis |
| M. mycoides subsp. capri | Pneumonia, arthritis septicemia (goat) |
| M. mycoides subsp. mycoides LC | Pneumonia, mastitis, arthritis, septicemia (goat) |
| M. agalactiae | Infectious agalactia |
| M. ovipneumoniae | Pneumonia (lamb) |
| M. conjunctivae | Infectious keratoconjunctivitis (IKC) (sheep) |
| Pig | |
| M. hyopneumoniae | Enzootic pneumonia |
| M. hyorhinis | Pneumonia, arthritis |
| M. hyosynoviae | Arthritis |
| Mycoplasma (Eperythrozoon) suis | Anemia |
| Horse | |
| M. felis | Pleuritis |
| M. equirhinis | |
| M. equipharyngis | |
The Mycoplasma species that affect ruminants cause some of the most economically important diseases worldwide. Contagious bovine pleuropneumonia (CBPP) an Office International des Epizootes (OIE) List A disease is associated with Mycoplasma mycoides subspecies mycoides small colony type, and contagious carprine pleuropneumonia (CCPP), and OIE List B disease is associated with Mycoplasma capricolum subspecies capripneumoniae. The disease complex contagious agalactia, an OIE List B disease, is associated with a number of mycoplasma species, including Mycoplasma agalactia, M. capricolum subspecies capricolum, M. mycoides subspecies mycoides large colony and Mycoplasma putrefaciens.
Between 1990 and 2000, more than 1600 mycoplasmas and the related acholeplasmas were identified from ruminant animals by the Mycoplasma Group at the Weybridge Laboratories Agency, Weybridge.1 Over the period, Mycoplasma bovis was the most commonly identified pathogen, with an overall mean of 52% of the isolates, mostly from pneumonic calves but also from cattle with mastitis and arthritis. Mycoplasma canis was first isolated in Britain in 1995 from pneumonic calves and the number of isolates increased 18% of the total mycoplasmas isolated from cattle in 1999. Other species isolated include Mycoplasma dispar from lungs of cattle with respiratory disease, and Mycoplasma bovigenitalium from the reproductive tract of cows with vulvovaginitis and infertility. Mycoplasma bovirhinis and Acholeplasma species were found commonly but are considered as opportunists. In sheep and goats, the majority of Mycoplasma species isolated were Mycoplasma ovipneumonia from pneumonic sheep. Mycoplasma conjunctivae from sheep with keratoconjunctivitis, and the ubiquitous Mycoplasma arginini.1
Molecular techniques such as denaturing gradient electrophoresis (DGGE) of a 16S ribosomal DNA PCR product have been used to differentiate almost all mycoplasmas within a host animal group.3 The method can enable the rapid identification of many mycoplasma species for which there is no specific PCR available and which are currently being identified by culture and serological tests. This has demonstrated that Mycoplasma species are not necessarily host specific. Molecular epidemiological analysis of 53 Mycoplasma bovis isolates from pneumonic cattle collected in UK between 1996 and 2002, revealed two genetically distinct clusters (A and B).4 There was no clear relationship between the geographic origin or year of isolation of the isolates and the profiles produced. Group B isolates were relatively more heterogeneous than isolates of the A group.
Mycoplasma bovis is a major pathogen causing respiratory disease, arthritis, mastitis, and other diseases such as otitis in cattle.5 M. bovis is found worldwide and has spread into new areas, including Ireland and parts of South America, in the last decade. There is considerable antigenic variation in M. bovis associated with its variable surface proteins (Vsps) which differ from other mycoplasmas.6 M. bovis is highly invasive and is not confined to the initial area of colonization, the respiratory tract. Consequently, organisms rapidly gain access to multiple organ systems.
Mycoplasma sp. bovine group 7 can cause substantial economic losses due to mastitis, polyarthritis and abortion in dairy cattle.7,8 The disease may been spread from mammary gland infections, and neonatal calves were most likely infected by the consumption of milk contaminated with the mycoplasmas. Abortions probably occurred due to a systemic infection of mycoplasma.
Mycoplasmas are the smallest prokaryotes with autonomous replication. They are extracellular parasites with an affinity for mucous membranes, where they exist as commensals or pathogens. Pathogenic mycoplasma have a predilection for the respiratory system, urogenital tract, mammary gland and serous membranes. Most mycoplasmas are adapted to a main host in which they are commonly pathogenic. They may colonize other hosts without being pathogenic. As parasites of mucous membranes, they adhere firmly to epithelial cells and adhesion is a prerequisite for colonization and infection. Their mechanism of virulence is not well-understood and activation of the immune system of the host probably plays a major role in the pathogenesis of mycoplasmoses. In general, mycoplasmas are not highly virulent but rather induce chronic diseases. Most animal mycoplasmoses are herd problems with high morbidity and relatively low mortality and healthy carriers are an important part of the epidemiology of mycoplasmoses.
The most important pathogens are found in the mycoides cluster with six species, among them the pathogens of contagious bovine pleuropneumonia, contagious caprine pleuropneumonia, and the pathogens of pneumonia, arthritis and mastitis in goats and sheep. Another group of related mycoplasmas are M. agalactiae (contagious agalactia of goats and sheep) and M. bovis (mastitis, pneumonia and arthritis in cattle). M. hyopneumoniae is the agent of enzootic pneumonia of swine. Many other mycoplasma species, which occur commonly as commensals on mucous membranes, are sporadically associated with disease. They are often found along with bacteria or viruses.
Mycoplasmas lack a cell wall and are therefore resistant to γ-lactam antimicrobials but sensitive to numerous other antimicrobials. The most active are macrolides (erythromycin, spiramycin, and tylosin), the tetracyclines, quinolines and chloramphenicol. The in vitro activity of danofloxacin, tylosin and oxytetracycline against field isolates of seven Mycoplasma species from cattle and pigs from five European countries were determined.9 Danofloxacin, a fluorquinolone, has excellent in vitro activity against M. hyopneumoniae, M. dispar and M. bovigenitalium and similar activity to that of tylosin against M. bovis.9 However, response to therapy is often unsatisfactory. In general, the antimicrobial sensitivity of mycoplasmas and ureaplasmas is greatest to tiamulin, then tylosin and least to oxytetracycline, but individual sensitivities vary sufficiently for it to be necessary to carry out laboratory tests of sensitivity on each isolate.
Those diseases in which mycoplasmas have been positively identified as the causative agent are described separately: contagious bovine pleuropneumonia, bovine arthritis, bovine mastitis, caprine pleuropneumonia and enzootic pneumonia of pigs. Other diseases in which mycoplasmas play a contributory part are set out with a summary in Table 20.6. The comparative characteristics of the two most important mycoplasmas of cattle are summarized in Table 20.7.
Table 20.7 Comparative properties of the two most important cattle mycoplamas
| Properties | M. mycoides subsp. | M. bovis |
|---|---|---|
| Mycoides SC | ||
| Diseases | Contagious bovine pleuropneumonia in cattle, occasionally in arthritis in calves | Calf pneumonia, mastitis, arthritis, abortion, keratoconjunctivitis |
| Distribution | Subsaharan Africa, probably in parts of Middle East, Central Asia | Worldwide |
| Hosts | Cattle, goats, (sheep) | Cattle |
| Histopathological | Fibrinous pleuropneumonima with necrosis | Interstitial pneumonia, lymphohistiocytic bronchitis, catarrhal bronchopneumonia |
| Clinical signs | Few signs, respiratory distress evident after exercise | Respiratory distress, mastitis, arthritis |
| Diagnosis | Isolation, serology, PCR, abattoir surveillance | Serology, isolation, PCR |
| Treatment | Chemotherapy not recommended because it encourages carrier status | Chemotherapy |
| Control | Vaccination, movement control, slaughter | Management, improved ventilation, reduced stocking density |
DISEASES OF THE GENITAL TRACT
Vulvovaginitis in cattle, sheep and goats may be associated with M. agalactiae var. bovis. The same infection when introduced with semen into the uterus can cause endometritis and salpingitis, resulting in a temporary infertility and failure to conceive.
Persistent infection in the genital tract of bulls has also been produced experimentally. Ureaplasmas have been isolated from the vulva of ewes with granular vulvitis and the disease was transmitted experimentally. However, the same organisms are present in the vulva of normal ewes and cows but Ureaplasma spp. are usually limited in their distribution to the vestibule and vulva of normal cows. In some areas, Ureaplasma diversum is commonly present in the lower reproductive tract of beef and dairy cattle, both cows and bulls, and is associated with granular vulvitis, which has been associated with infertility, sporadic abortions and neonatal mortality.10 This infection adversely affects reproduction when it is either acute or chronic; it is capable of producing granular vaginitis and some strains can, if introduced to the upper reproductive tract, cause transitory endometritis and salpingitis.
Ureaplasms, M. bovis and M. bovigenitalium have been found the reproductive tract of bulls and their semen.11 Using the PCR for the detection of mycoplasma in semen, M. mycoides subsp. mycoides SC has been found in semen of yearling bulls with seminal vesiculitis while negative to the complement-fixation test for CBPP.11
A combination of lincomycin– spectinomycin–tylosin has been shown to be most effective in the treatment of Ureaplasma spp. in bull semen. Chlortetracycline at 350 mg/h/d for 30 d in the prebreeding feed of virginal heifers, most of which had vulvovaginitis and from which 44% cultured positive for U. diversum, improved pregnancy rates and decreased the vaginal colonization of the organism.12
Parenteral vaccination of heifers with killed U. diversum induced antibodies to the mycoplasma but did not prevent subsequent infection or clear the ureaplasmas.13 The use of the vaginal submucosal route for vaccination resulted in characteristic granular vulvitis in both vaccinated and control animals.14
Attempts to produce abortion in cows by the injection of mycoplasmas isolated from aborted fetuses and from weak calves has had varying success. Acholeplasma spp. have been isolated from aborted equine fetuses. M. bovigenitalium has been a frequent isolate from bovine genital tracts for many years, but its role in genital disease is still uncertain. It has been isolated from frozen bull semen and poses a threat to cows inseminated with infected semen. Mycoplasmas in semen can be transmitted through in vitro fertilization and infect embryos, and supplementation of culture media with standard antibiotics and washing embryos as recommended by the International Embryo Transfer Society are not effective in making IVF embryos free from M. bovis and M. bovigenitalium.9
Mycoplasma bovis in frozen semen can survive the antibiotic combination of gentamicin, tylosin, and lincomycin and spectinomycin.15
MYCOPLASMA MASTITIS IN DAIRY HERDS
Mycoplasma bovis, Mycoplasma californicum, and Mycoplasma canadense are causes of outbreaks of mastitis in cattle, and occasionally pneumonia, otitis media, or arthritis in calves of those dairy herds. The literature on mycoplasma mastitis in dairy herds has been reviewed.16 The infection is highly contagious and is commonly introduced into a previously Mycoplasma-free herd by the purchase of infected heifers or cows. At least 11 other species of Mycoplasma have been isolated from milk of cows with mastitis and the disease produced by each is similar. Cows of all ages and at any stage of lactation are susceptible. Typically, acute mastitis occurs which is resistant to treatment. Usually all four quarters are affected, there is a marked drop in milk production, and abnormal udder secretions vary from being like watery milk with a few clots to colostrum-like material. The affected cow is usually systemically normal. Chronically affected animals have a tan-colored milk secretion with sandy or flaky sediments which may become purulent-like over several weeks. However, the majority of milk samples which are positive for mycoplasma appear normal. In affected cows, milk production commonly declines, with normal appearance of milk but a high somatic cell count.
Mycoplasma mastitis in cattle is highly contagious and infections are transmitted at milking by means of fomites. Diagnosis is dependent on culture of milk samples from the bulk tank milk or individual cow milk samples. To identify cows with M. bovis mastitis, an indirect ELISA is available to detect antibodies to M. bovis in milk samples from cows with recently acquired M. bovis mastitis.17 For mycoplasmal mastitis, individual cows milk sampling for culture and identification of M. bovis is time consuming and expensive. Some herds sample cows monthly with the dairy herd improvement (DHI) program but a preservative is added to the milk that kills M. bovis. A nested PCR procedure allows for rapid testing of preservative treated milk and is as sensitive as traditional culture.18
In herds where the diagnosis has been made, weekly monitoring of bulk tank milk is necessary to monitor the success of control procedures. Quality Milk Production Services, Cornell University, recommends the use of 1% iodine products to reduce the number of Mycoplasma on teat skin during mastitis outbreaks. There is no treatment for mycoplasma mastitis and vaccination has been ineffective. In affected herds, milk of all lactating animals should sampled, positive animals identified and culled or segregated from the mycoplasma-free animals. Waste milk from infected cows should not be fed to calves without pasteurization.19
Experimental vaccines against mycoplasma vaccines have been unsuccessful and may even exacerbate the mastitis.5 It is best to segregate or cull carrier cows and to implement rigid sanitation procedures to prevent transmission from infected to non-infected cows.
Excellent vigilance and rapid culling of infected cattle are critical control factors affecting the spread of mycoplasma mastitis. Herds commonly become negative within the first year of the first case of mycoplasma mastitis.20
(Additional details on Mycoplasma mastitis in cattle are available in Chapter 15.)
DISEASES OF THE RESPIRATORY TRACT
Several different mycoplasmas have been isolated from pneumonic and non-pneumonic lungs of cattle, sheep and goats, but attempts to reproduce respiratory tract disease with them has resulted in inconclusive findings.
Sheep
In general, mycoplasmas cause a subclinical, mild pneumonia in gnotobiotic animals, but in combination with unidentified agents in lung homogenates administered intransally, ‘enzootic’ or chronic progressive pneumonia is produced in sheep. For example, M. dispar has a cytopathogenic effect and stops ciliary motility, and destroys ciliated epithelial cells in organ cultures. M. ovipneumoniae also has the capacity to colonize the sheep lung and produce mild pneumonic lesions, but in combination with pasteurellae in sheep, causes a proliferative exudative pneumonia. A similar disease in Icelandic sheep (kregda) has been identified as being associated with M. ovipneumoniae. Similar diseases are associated with M. ovipneumoniae and M. arginini.
Goats
Although contagious caprine pleuropneumonia has not been observed in Australia, a non-fatal respiratory disease of goats, characterized by coughing, fever and extensive pleurisy and pneumonia, has. A variety of mycoplasmas, including M. agalactiae and M. mycoides and M. mycoides var. capri have also been found in goats. The caprine M. mycoides var. mycoides (large colony type) is not pathogenic for cattle and has been associated with a variety of syndromes in goats including fibrinous peritonitis, pneumonia, arthritis, mastitis and abortion. It has been cultured from a goat with a subauricular abscess and mastitis.21 The most common syndrome in goats associated with mycoplasma is a chronic interstitial pneumonia with cough, unthriftiness proceeding to extreme emaciation, chronic non-painful bony enlargement of joints and chronic indurative mastitis. The pneumonia in some cases progresses to the point where cor pulmonale develops with a subsequent appearance of the signs of congestive heart failure. M. ovipneumoniae has also been credited with causing pneumonia in goats. M. adleri has been isolated from a goat with a joint abscess.22
Cattle
Mycoplasma bovis is a major cause of calf pneumonia. Mycoplasma dispar, Ureaplasma diversum, Mycoplasma bovirhinis, and Mycoplasma canis have also been isolated from the lungs of pneumonic cattle but it is uncertain if they are primary causes of disease.5 M. dispar is capable of producing a pneumonia without clinical signs in gnotobiotic calves, and in conjunction with Ureaplasma spp. it has been found commonly in ‘cuffing’ pneumonia of calves. It could, therefore, be a precursor to other infections causing enzootic pneumonia in calves or with pasteurellae producing fibrinous pneumonia of calves.
DISEASES OF THE EYES
Mycoplasma bovoculi (Acholeplasma oculi) has been associated with, without necessarily being the cause in the field of, outbreaks of infectious keratoconjunctivitis of cattle, sheep and goats. The mycoplasmas are capable of producing keratitis experimentally and M. bovis has been isolated from the ocular discharge of young cattle affected with conjunctivitis.24 The naturally occurring disease in goats is manifested by rapid spread and development with intense lacrimation, conjunctival hyperemia, corneal opacity and vascularization. A concurrent respiratory illness occurs in some goats. Response to treatment with oxytetracycline and polymyxin B is good. The disease is reproducible experimentally.
DISEASES OF THE BLOOD
Mycoplasma (Eperythrozoon) suis infection occurs worldwide in pigs causing fever and anemia in young animals and, occasionally, latent anemia in older animals. E. suis, originally classified as a Rickettsia, has now been shown to belong to the mycoplasmas based on phylogenetic analysis of its rrs (16S rRNA) genes and is now proposed to be known as Candidatus Mycoplasma haemosuis.
DISEASES OF THE JOINTS
Mycoplasma (M. hyorhinis, M. hyosynoviae) are associated with arthritis and polyarthritis in pigs and the disease is reproducible experimentally. Both organisms are carried in the respiratory tract. M. hyorhinis most commonly affects suckling and young pigs, especially after weaning or some stress, and may produce a polyserositis in addition to arthritis. The case–fatality rate is generally low, but residual fibrinous pericardial and pleural adhesions may occur and the pigs may fail to thrive.
Disease associated with M. hyosynoviae is more common in older growing pigs as arthritis. The disease is endemic in some herds and may affect up to 15% of the herd. Clinically, there is an acute episode of fever, lameness and swelling of the joint. The lesion is a serofibrinous arthritis and most cases recover, but some may be affected chronically. M. hyosynoviae was isolated from 20% of arthritic lesions of pigs from an abattoir.25 The case–fatality rate is low and does not exceed 10%. Tylosin and lincomycin are indicated and effective.
In goats and occasionally lambs, M. capricolium causes arthritis. The infection is transmissible experimentally via infected milk administered orally. There is a septicemia with subsequent pneumonia and arthritis.
M. mycoides var. mycoides (large colony type) has been isolated from goats with arthritis. It has caused acute arthritis in 3 to 8-week-old kids manifested by lameness, recumbency, diarrhea and fever. The infection may originate from infected milk from does which have had acute mastitis due to M. mycoides var. mycoides. It may also be transmitted by ear mites, Psoroptes cuniculi and goat fleas.11 A similar disease is observed in sheep. At necropsy the kids have polyarthritis, pneumonia and pleurisy. M. putrefaciens has caused major losses due to mastitis and arthritis in a large goat herd. Abortion was common in some groups.
M. capricolium and M. putrefaciens are also causes of arthritis in sheep and kids.27 The experimental inoculation of lambs with A. laidlawii, which is normally considered non-pathogenic for animals, induced arthritis and renal lesions but the organism was not cultured from the joint fluid.28
DISEASES IN HORSES
There are few records of mycoplasmosis in horses. Most descriptions relate to the isolation of mycoplasmas, usually from the respiratory and female genital tracts, and almost always without any associated lesions. M. equirhinis and M. felis are pathogens of horses.23,29,30
DISEASES IN SHEEP AND GOATS
The moderately well-identified syndromes of these species are dealt with separately. There are many other reports of mycoplasmal diseases in these species in which the mycoplasma is not identified, and a number of diseases as set out earlier in which the etiological significance of the mycoplasmas is in doubt. For example, the two main clinical syndromes of these species are pleuropneumonia and agalactia, as described in subsequent sections. However, the signs and lesions of more than one of these ‘specific’ diseases and the infections themselves can be encountered in the one animal (see Table 20.6).
Bergonier D, Berthelot X, Frey J. Mycoplasmas of ruminants: pathogenecity, diagnostics, epidemiology and molecular genetics. Volume 4. European Commission. Directorate-General for Research. COST Action 826. Agriculture and biotechnology. European cooperation in the field of scientific and technical research. 2000:1–257
Frey J. Mycoplasmas of animals. In: Razin S, Herrman S, editors. Molecular biology and pathogenicity of mycoplasmas. New York: Kluwer Academic/Plenum; 2002:73-90.
Gonzalez RN, Wilson DJ. Mycoplasmal mastitis in dairy herds. Vet Clin Food Anim Pract. 2003;19:199-221.
Nicholas RAJ, Ayling RD. Mycoplasma bovis: disease, diagnosis, and control. Res Vet Sci. 2003;74:105-112.
1 Ayling RD, et al. Vet Rec. 2004;155:413.
2 Frey J. Mycoplasmas of animals. In: Razin S, Herrman S, editors. Molecular biology and patho-genicity of mycoplasmas. New York: Kluwer Academic/Plenum; 2002:73-90.
3 McAuliffe L, et al. J Clin Microbiol. 2003;41:4844.
4 McAuliffe L, et al. J Clin Microbiol. 2004;42:4556.
5 Nicholas RAJ, Ayling RD. Res Vet Sci. 2003;74:105.
6 Minion FC. Front Biosci. 2002;7:d1410.
7 Hum S, et al. Aust Vet J. 2000;78:744.
8 Djordjevic SP, et al. Electrophoresis. 2001;22:3551.
9 Bielanski A, et al. Theriogenology. 2000;53:1213.
10 Mulira GL, et al. Can. Vet J. 1992;33:46.
11 Stradaioli G, et al. Vet Res. 1999;30:457.
12 Rae DO, et al. Theriogenology. 1993;40:497.
13 Mulira GL, Saunders JR. Am J Vet Res. 1994;58:104.
14 Mulira GL, Saunders JR. Am J Vet Res. 1994;58:109.
15 Visser IJR, et al. Theriogenology. 1999;51:689.
16 Gonzalez RN, Wilson DJ. Vet Clin Food Anim Pract. 2003;19:199.
17 Byrne WJ, et al. Vet Rec. 2000;146:368.
18 Pinnow CC, et al. J Dairy Sci. 2001;84:1640.
19 Butler JA, et al. J Dairy Sci. 2000;83:2285.
20 Fox LK, et al. J Vet Med B. 2003;50:235.
21 Blikslager AT, Anderson KL. J Am Vet Med Assoc. 1992;201:1404.
22 Guidice RAD, et al. Int J Syst Bacteriol. 1995;45:29.
23 Wood JLN, et al. J Clin Micro. 2005;43:120.
24 Kirby FD, Nicholas RAJ. Vet Rec. 1996;138:552.
25 Fries NF, et al. Acta Vet Scand. 1992;33:205.
26 Nayak NC, Bhowmik MK. Prev Vet Med. 1990;9:259.
27 Rodriquez JL, et al. Vet Rec. 1994;135:406.
28 Concha-Bermejillo A, De La, et al. J Vet Diag Invest. 1996;8:115.