VIRAL PAPULAR DERMATITIS
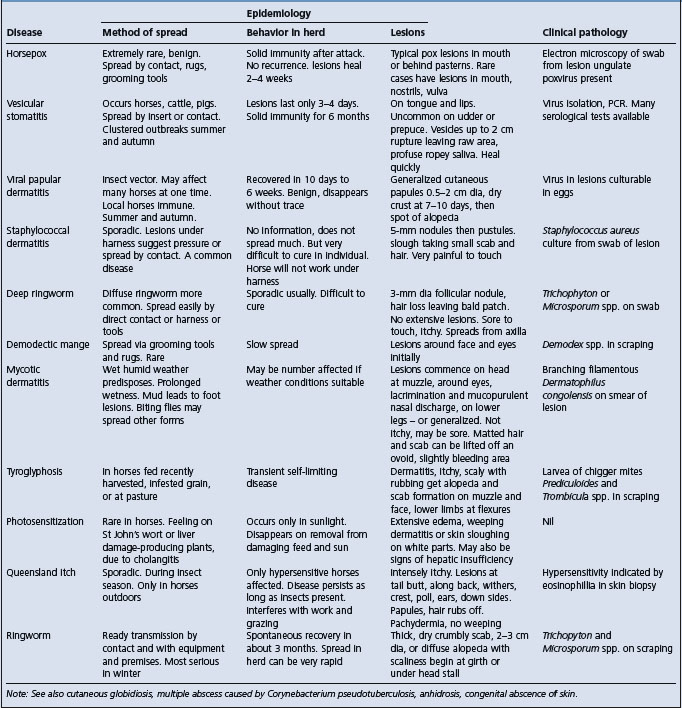
This disease of horses occurs in the United States, United Kingdom, and Australia. It is a contagious disease characterized by cutaneous lesions in the form of firm papules 0.5–2 cm in diameter. No vesicles or pustules are formed, but after 7–10 days a dry crust is detached, leaving small circumscribed areas of alopecia. The lesions are not itchy, there is no systemic disease and the distribution of the lesions, and the way in which they can develop simultaneously in large numbers in introduced horses, is suggestive of an insect-borne disease.
The course of the disease varies between 10 days and 6 weeks. An unidentified virus has been isolated from lesions and cultured on eggs. A febrile reaction, up to 40.2°C (104.5°F), precedes the appearance of skin lesions by about 24 hours. There is no histological description. Recovery is usually complete and uncomplicated. The disease is clinically similar to molluscum contagiosum in horses associated with poxvirus. This disease has similar papular lesions, which are hypopigmented and covered by tufts of raised hair but the disease has a long clinical course. Histologically, these show proliferation of keratinocytes containing large intracytoplasmic inclusions, known as molluscum bodies, which are composed of numerous pox virions.
COWPOX
Etiology Cowpox virus is a member of the genus Orthipoxvirus in the family Poxviridae
Epidemiology Endemic infection of certain rodents in Europe and east Asia. Cattle are a rare and incidental host. Spread in cattle by contact
Clinical findings Typical pox lesions on the teats and udder. Erythema, papules with a zone of hyperemia around the base, vesiculation, pustular stage and scab
Clinical pathology Electron microscopy
Diagnostic confirmation Electron microscopy and virus isolation
ETIOLOGY
Cowpox virus is a member of the genus Orthipoxvirus in the family Poxviridae. Other orthipoxviruses infecting agricultural animals include horsepox, Uasin Gishu, buffalopox and camelpox.1 All orthipoxviruses are antigenically extremely similar, but they can be identified by a combination of phenotypic and genetic tests.
EPIDEMIOLOGY
Occurrence
Cowpoxvirus could be considered to be misnamed. Infection with this virus is endemic in wild rodents such as voles (Microtus spp.) in Great Britain, Europe and western Asia, with infection in different rodent species acting as the reservoir host in different geographic areas.2,3 Domestic cats are commonly infected from hunting rodents, but cowpoxvirus infection can occur in a number of different mammalian species,2 one of which is cattle. The clinical syndrome of cowpox in cattle is now extremely rare but it occurs sporadically in Europe. Seroprevalence in British cattle is less than 1%.
Origin of infection and transmission
The origin of infection is most probably from infected farm cats or humans. Transmission from cow to cow within a herd is effected by milkers’ hands or teat cups. Spread from herd to herd is probably effected by the introduction of infected animals, by carriage on milkers’ hands, and in the absence of either of the above methods, transport by biting insects is possible. In a herd in which the disease is enzootic, only heifers and new introductions develop lesions. Milkers recently vaccinated against smallpox may serve as a source of infection for cattle, although the vaccinia virus, the smallpox vaccine virus, is a different virus.
It is generally assumed that the virus gains access to tissues through injuries to teat skin, and extensive outbreaks of cowpox are likely to occur when the environment is conducive to teat injuries. Spread is rapid within a herd and immunity is solid, so that the disease tends to occur in sharp outbreaks of several months’ duration with subsequent immunity protecting the cattle for at least several years.
PATHOGENESIS
In cowpox, the five stages of a typical pox eruption can be observed. After an incubation period of 3–6 days, a roseolar erythema is followed by firm, raised papules light in color but with a zone of hyperemia around the base. Vesiculation, a yellow blister with a pitted center, follows. The subsequent pustular stage is followed by the development of a thick, red, tenacious scab.
In experimentally produced vaccinia virus mammillitis (produced by inoculation of smallpox vaccine), the lesions have three zones: a central brown crusty area of necrosis, surrounded by a gray-white zone of microvesicle formation, again surrounded by a red border due to congestion. The lesions are essentially hyperplastic.
CLINICAL FINDINGS
Typical lesions may be seen at any stage of development, but are mostly observed during the scab stage, the vesicle commonly having been ruptured during milking. True cowpox scabs are 1–2 cm in diameter and are thick, tenacious, and yellow-brown to red in color. In cows being milked, scab formation is uncommon, the scab being replaced by a deep ulceration.
Distribution of the lesions is usually confined to the teats and lower part of the udder. Soreness of the teats develops and milk letdown may be interfered with; the cow usually resents being milked. Secondary mastitis occurs in a few cases. Individual lesions heal within 2 weeks, but in some animals fresh crops of lesions may cause the disease to persist for a month or more. In severe cases, lesions may spread to the insides of the thighs, and rarely to the perineum, vulva and mouth.6 Sucking calves may develop lesions about the mouth. In bulls, lesions usually appear on the scrotum.
CLINICAL PATHOLOGY
The virus can be propagated in tissue culture, and differentiation is possible by electron microscopy.7
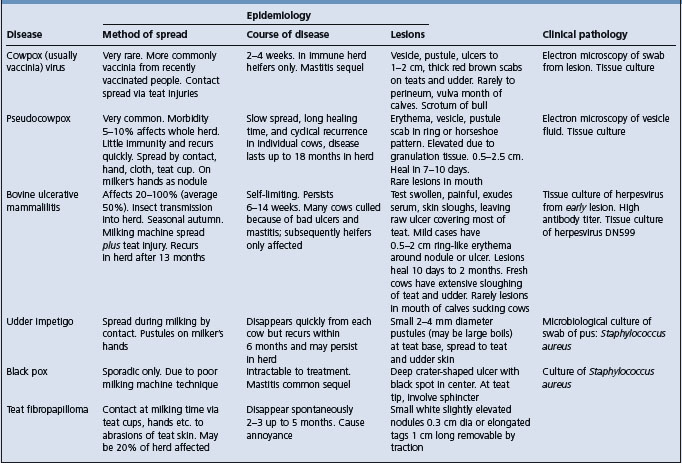
A number of skin diseases may be accompanied by lesions on the udder and can easily be confused with cowpox if the lesions are advanced in age. The differential points are listed in Table 22.2. Most outbreaks of teat skin disease that clinically resemble classical cowpox are associated with vaccinia virus from contact with a recently vaccinated person.
CONTROL
Prevention of spread is difficult, since the virus responsible for the disease is readily transmitted by direct or indirect contact. Udder cloths, milking machines and hands should be disinfected after contact with infected animals. Dipping of the teats in an alcoholic tincture of a suitable disinfectant, such as quaternary ammonium compounds, is usually satisfactory in preventing immediate spread. The prevalence and significance of the disease in cattle is too low to warrant the development of vaccines.7
1 Smith SA, Kotwal GJ. Crit Rev Microbiol. 2002;28:149.
2 Bennett M, Baxby D. J Med Microbiol. 1996;45:157.
3 Hazel SM, et al. Epidemiol Infect. 2000;124:551.
4 Baxby D, et al. Br J Dermatol. 1994;131:598.
5 Bennett M, Baxby D. Vet Ann. 1995;35:229.
6 Yerahum I, et al. Rev d’Elev Med Vet Pays Trop. 1996;49:299.