BRUCELLOSIS ASSOCIATED WITH BRUCELLA OVIS
Epidemiology Disease of sheep. Organism carried by sexually mature rams with spread by direct contact or passive venereal infection
Clinical findings Clinical or subclinical disease. Infertility in rams due to epididymitis. Epididymal abnormality can be detected by palpation in some affected rams. Abortion in ewes and neonatal mortality in lambs are also occasionally caused by the infection
Clinical pathology Semen examination. Serology of most value including complement fixation, gel diffusion, and ELISA
Diagnostic confirmation Physical palpation of the contents of the scrotum combined with cultural examination of semen (or aborted material), and serological examination
Treatment Oxytetracycline in valuable rams
Control Total segregation of normal and young rams. Initial culling of rams with palpable scrotal abnormality and subsequent repeated serological testing and culling of seropositive rams. Alternative is vaccination with live B. melitensis strain Rev. 1
ETIOLOGY
Brucella ovis has significant DNA homology with other members of the genus Brucella1,2 and also shares many antigenic and other characteristics but it is permanently rough.
EPIDEMIOLOGY
Geographical occurrence
Brucellosis of sheep associated with B. ovis has been reported in most of the major sheep-producing regions of the world and is present in Australia, New Zealand, North and South America, Central Asia, Russia, South Africa, and Europe but is not a major cause of ram wastage in Great Britain. When the disease is first diagnosed in a country, and before control procedures are established, the flock prevalence of infection can be as high as 75% and as many as 60% of rams may be infected.3,4 The prevalence of infection is generally much lower in countries and in flocks that have established control programs.
Host occurrence
In nature only sheep are affected, although the ram is more susceptible than the ewe. Infection can be established experimentally in laboratory animals only with difficulty.3 White-tailed deer and goats can be infected experimentally, leading to the development of an epididymitis, but there is no evidence of natural infection in goats, even in those that cohabit with infected sheep.
The Merino breed and Merino-derived crossbreeds show a much lower incidence of the disease than do British breeds. The disease is most important in large flocks where there is multi-sire breeding.
Source of infection
The infected ram is the source of infection and perpetuates the disease in a flock. The majority of infected rams excrete the organism in semen5 and in most rams the active excretion in semen probably persists indefinitely.
Ewes are more resistant to infection but the organism can be isolated from them in infected flocks.6 After being bred by an infected ram, the majority will not carry infection for more than one or two heat cycles.7 Infection may result in early embryonic death and occasionally abortion or the birth of weak and poorly viable lambs.3 In ewes where the infection does persist to produce abortion, the organism is present in the placenta, vaginal discharges, and milk.
Transmission
Transmission between rams occurs via passive venereal infection and by direct ram-to-ram transfer.3,7
Passive venereal infection occurs from ewes that have been bred by an infected ram in the same heat cycle. Under natural conditions, this may be the major from of transmission from ram to ram that occurs during the breeding season. Infection can also be transmitted between rams in the nonbreeding season that are housed together or grouped together on pasture. This occurs as they sniff and lick each other’s prepuce and by homosexual activity. Submissive rams may lick the prepuce of dominant rams as a trait in the dominance hierarchy. Spread of infection in a group of virgin rams is recorded.8 Lambs born from infected ewes and drinking infected milk do not become persistently infected.
The organism can survive on pasture for several months but transmission by fomites appears is believed to have no practical significance. However, transmission from infected rams to infection-free red deer stags grazed on the same pasture can occur and it is not known if this results from direct contact between the animals or indirectly via environmental and pasture contamination.9
Host risk factors
All postpubertal rams are susceptible to infection, but disease occurs more commonly in adult rams and disease prevalence increases with age, probably because of greater exposure to infection.4 Differences between flocks in the prevalence of disease suggest that environmental factors and stress may modulate susceptibility7 but the risk factors are poorly defined. When the number of affected rams in a flock is greater than about 10%, the fertility of the flock is appreciably decreased.
Experimental reproduction
Experimentally, rams can be infected by the intravenous, subcutaneous, intratesticular, oral, conjunctival, and preputial routes but the latter two are the most effective.3,10,11 The first observable abnormality is the presence of inflammatory cells in the semen, which appear at 2–8 weeks. B. ovis appears in the ejaculate at approximately 3 weeks but it is not invariably present in all examinations of an infected ram. Testicular and epididymal lesions can be palpated at about 9 weeks after infection but may occur earlier in some rams. A significant proportion of infected rams have no palpable lesions but still excrete the organism.
Ewes in early pregnancy can also be infected by the oral and intravenous routes but many of these infections are transient and do not result in abortion. Abortion due to placentitis has been produced experimentally. Intrauterine infection produced experimentally also causes lesions in, and death of, the fetus but the significance of this to natural cases is undetermined.
Economic importance
The economic effects of the disease are subtle but significant. The effect of the disease on ram fertility can influence the number of rams that are required in a flock: the required ram to ewe ratio is significantly reduced in B. ovis-free flocks. The percentage of lambs born early and within the first 3 weeks of the lambing period is also markedly increased. Lambing percentage may be reduced by 30% in flocks recently infected and by 15–20% in those where the infection is endemic.7 Additional costs are the loss of rams of high genetic potential and the cost of repeat serological testing. In the USA, an additional return of US$12 per ewe mated has been calculated as the advantage in a control program.12
PATHOGENESIS
There is an initial bacteremia, often with a mild systemic reaction, and the organism can be isolated from the internal organs of animals slaughtered after experimental infection. However, systemic disease is not a feature of the natural disease, and clinical disease results from localization and inflammation in the epididymides. Inflammation in this area results in sperm stasis and extravasation with a subsequent immunological reaction that is usually in the tail and unilateral, causing a spermatocele and therefore reduced fertility. Not all infected rams have palpable lesions in the epididymis: infection can also establish in the seminal vesicles and ampullae. In either case the organism is shed in the ejaculate.
In general, the evidence is that B. ovis has low pathogenicity for ewes: the primary effect of infection is a placentitis, which interferes with fetal nutrition, sometimes to the point of causing fetal death but more commonly producing lambs of low birth weight and poor viability.
CLINICAL FINDINGS
The first reaction in rams is a marked deterioration in the quality of the semen together with the presence of leukocytes and Brucella. Acute edema and inflammation of the scrotum may follow. A systemic reaction, including fever, depression, and increased respiratory rate, accompanies the local reaction.
Regression of the acute syndrome is followed, after a long latent period, by the development of palpable lesions in the epididymis and tunicae of one or both testicles.
The palpation of both testicles simultaneously from behind is the best method of examination. The epididymis is enlarged and hard, more commonly at the tail, the scrotal tunics are thickened and hardened, and the testicles are usually atrophic. The groove between the testis and epididymis may be obliterated.
The abnormalities are often detectable by palpation but many affected rams show no acute inflammatory stage and others may be actively secreting Brucella and poor-quality semen in the chronic stage in the absence of palpable abnormalities. Palpable abnormality of the scrotal contents may be present in less than 50% of serologically positive rams.3 Affected rams have normal libido.
There are usually no clinical signs in the ewe but in some flocks infection causes abortion or the birth of weak or stillborn lambs, associated with a macroscopic placentitis.
CLINICAL PATHOLOGY
Semen examination, including culture of the ejaculate, and serological tests are used in suspect individuals and in groups of rams. The complement fixation or other serological tests (agar gel immunodiffusion, ELISA) are by far the most important; many infected rams have palpably normal scrotal contents and microbiologically negative semen.
Semen examination
A combination of semen examination and palpation of the testicles for abnormality is stated to identify approximately 80% of infected rams.12 In affected animals the findings are a general reduction in semen quality, a reduced total sperm output, poor motility, and a high proportion of spermatozoa with secondary morphological abnormalities.
Culture
B. ovis is fastidious in its growth and requires special cultural techniques. The examination of the semen for the presence of leukocytes has been used to determine those sheep that should be cultured for B. ovis but this criterion is not highly sensitive.13,14 PCR for detection of B. ovis in semen has equivalent sensitivity to culture.15
Serology
The complement fixation test is the standard test in many countries, is the prescribed test for international trade and, when used in conjunction with genital palpation and semen culture, has allowed the eradication of B. ovis from flocks. However, a small proportion of infected rams are negative to the complement fixation test, which can compromise eradication programs. A number of tests, including ELISA tests,16-18 immunoblotting,19 and gel diffusion tests,18 are also used.
The sensitivity and specificity of the various serological tests depend mainly on the antigens used and the serological cutpoints,3,18,20 which may vary between countries and laboratories. One study20 reported the sensitivity of the ELISA, gel diffusion, and complement fixation tests as 97.6%, 96.4%, and 92.7% respectively, with all tests 100% specific. Studies in other countries support this ranking3,7,21,22 and the ELISA is becoming more commonly used. Other studies suggest that the ELISA has no advantage in specificity over the classic complement and gel diffusion tests.18 A combination of serological tests may improve sensitivity to 100%.20,21,23
Serological tests will not differentiate vaccinated from infected sheep or sheep infected with B. melitensis.
In Australia, there has been an unexpectedly high prevalence of false-positive reactors in some flocks because rams exposed to infection have developed a positive reaction to a serological test but have not developed the disease. These transient infections have not been found to be a problem in other regions.20,21
NECROPSY FINDINGS
In the acute stage, there is inflammatory edema in the loose scrotal fascia, exudate in the tunica vaginalis and early granulation tissue formation. In the chronic stage, the tunics of the testes become thickened and fibrous and develop adhesions. There are circumscribed indurations in the epididymis and these granulomata may also be present in the testicle. In advanced stages they undergo caseation necrosis. As the epididymis enlarges the testicle becomes atrophied. B. ovis can usually be isolated from the genital organs, especially the tail of the epididymis, and rarely from internal organs and lymph nodes.24
The abortus is characterized by thickening and edema, sometimes restricted to only a part of the placenta, with firm, elevated yellow-white plaques in the intercotyledonary areas and varying degrees of cotyledonary necrosis. Microscopically, organisms are visible within the cytoplasm of trophoblasts of the inflamed placenta. A vasculitis is often present. The organism can be isolated from the placenta and the stomach and lungs of the lamb.
Samples for confirmation of diagnosis
• Bacteriology – epididymal granuloma, seminal vesicle, inguinal lymph node/fetal lung, stomach content, placenta (CULT – has special growth requirements, CYTO – Stamp’s or Kosters’ stain on placental smear)
• Histology – formalin-fixed epididymis, testicle, inguinal lymph node/placenta, fetal lung, liver, spleen, kidney, heart, brain.
Many rams with abnormalities of intrascrotal tissues do not have brucellosis.5 Most are cases of epididymitis and need to be differentiated.
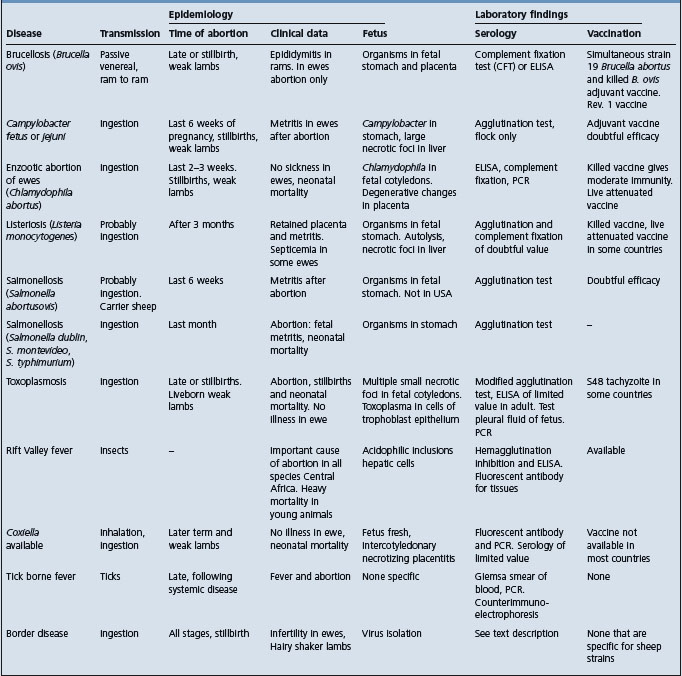
Abortion in ewes may be associated with a number of infectious diseases, summarized in Table 18.8.
TREATMENT
Treatment of naturally occurring cases is rarely undertaken.
In experimentally infected rams, the intramuscular administration of long-acting oxytetracycline at 20 mg/kg BW, given every 3 days for 24 days, along with the daily intramuscular administration of 20 mg/kg of dihydrostreptomycin sulfate, has resulted in bacteriological cure in 11 of 12 rams.25 Oxytetracycline alone is less effective. In another study using a similar treatment, seven of nine rams had bacteriological cure.13 Treatment is economically practicable only in valuable rams and must be instituted before irreparable damage to the epididymis has occurred. The treatment of rams that are infected but without palpable lesions results in a significant improvement in breeding soundness classification on examinations subsequent to treatment.13
CONTROL
Control is by the prevention of spread of infection between rams and the detection and culling of infected rams. In small commercial flocks, culling of all rams and replacement with B. ovis-free rams may be the most economical approach. A measure of control can be achieved using scrotal palpation to detect infected rams but this must be coupled with repeated serological examinations if eradication is the goal. Vaccination may be the most economical and practical means of controlling the disease in areas with a high incidence of infection and in regions of the world where eradication by test and slaughter is impractical.14
Eradication
In a herd where the diagnosis has been confirmed all rams are palpated and those with scrotal abnormalities are culled. The remainder are tested serologically and reactors are culled. Serological tests are repeated at monthly intervals, with culling of reactors, until all rams are serologically negative. A further test, 6 months later, is used for confirmation.23
The rate of spread of infection is high during the mating season and it is not recommended that eradication should be attempted until after the breeding season. During breeding it may be wise to run two breeding flocks, with virgin rams and rams known to be free of infection separate from older or suspicious rams. Strict separation of the two ram flocks must be maintained at all times of the year, and the clean group must not mate ewe flocks that have been mated to these older rams.
The use of the ELISA test available in the USA has allowed the eradication of the disease in flocks with as few as four tests.12
Vaccination
A number of vaccines are in use but none is fully effective. In some countries vaccination is not permitted and eradication by test and slaughter is the only method of control.
Killed B. ovis vaccines, even when adjuvanted, have poor efficacy.7,15 The use of a killed vaccine may be inadvisable in flocks where eradication is being attempted, as it may protect against clinical disease but allow a carrier state in some rams in which there is excretion of the organism in animals that become seronegative.15 An experimental vaccine prepared from enriched outer membrane proteins and rough lipopolysaccharide of B. ovis has given equivalent protection in challenge studies to that given by B. melitensis Rev. 1 vaccine.26
A combined vaccine containing killed B. ovis in an adjuvant base and B. abortus strain 19 gives a high-level, durable immunity but the vaccine has several disadvantages. Vaccinated animals become seropositive, which hinders subsequent use of serological tests for eradication. Strain 19 itself can cause epididymitis and vaccinated rams may excrete strain 19 in their semen.3 Severe outbreaks of osteomyelitis and epiphysitis have been recorded in rams following vaccination.27
Live B. melitensis strain Rev. 1 has been found to be most effective and is generally recommended.3,28,29 Strain Rev. 1 is avirulent for rams, and subcutaneous or conjunctival vaccination provides protection against experimental and field challenge. Vaccinated animals become positive to the complement fixation test and ELISA test, but the titers produced are low and can be minimized by using the conjunctival route for vaccination.29,30
B. suis strain 2 (S2) has been used in China for vaccination of all target species against brucellosis. It is given orally in the drinking water and colonizes the cranial lymph nodes. In a comparative study of S2 vaccine and Rev. 1 vaccine, S2-vaccinated rams had less protection than Rev. 1-vaccinated rams when challenged with B. ovis and an equivalent degree of protection to the nonvaccinated controls.31
B. abortus RB51 vaccine does not protect rams against experimental challenge with B. ovis.32
Outer membrane protein (Omp) antigens are currently being examined as vaccine candidates. A detergent-extracted Omp31 from B. melitensis produced in E. coli has been shown to be a protective immunogen against B. ovis in a mouse model and to induce antibody and a cellular immune response in sheep.33 Its efficacy in protecting sheep against experimental or natural infection is not reported.
In all vaccination programs there should be a concurrent program of culling of clinically abnormal rams. All ram replacements should be yearlings vaccinated at 4–5 months.
Afzal M, Kimberling CV. How to control Brucella ovis-induced epididymitis in rams. Vet Med. 1986;81:364-370.
Rahaley RS. Ovine brucellosis; recent advances. Aust Adv Vet Sci. 1986:90-92.
Blasco JM. Brucella ovis. In: Nielsen K, Duncan JR, editors. Animal brucellosis. Boca Raton, FL: CRC Press, 1990.
Bulgin MS. Epididymitis in rams and lambs. Vet Clin North Am Food Anim Pract. 1990;6:683-690.
Genetzky RM. Epididymitis in rams. Compend Contin Educ Pract Vet. 1995;17:447-454.
Elberg S. Rev 1 Brucella melitensis vaccine. Part III 1981–1995. Vet Bull. 1996;66:1193-1200.
Ridler AL. An overview of Brucella ovis infection in New Zealand. N Z Vet J. 2002;50:S96-S98.
1 Verger JM, et al. Int J Syst Bacteriol. 1985;35:292.
2 Michaux-Charachon S. J Bacteriol. 1997;179:3244.
3 Blasco JM. Nielsen K, Duncan JR, editors. Animal brucellosis. Boca Raton, FL: CRC Press. 1990:351.
4 Walker RL, et al. J Am Vet Med Assoc. 1986;188:393.
5 Genetzky RM. Compend Contin Educ Pract Vet. 1995;17:447.
6 Marco J, et al. Vet Rec. 1994;135:254.
7 Bulgin MS. Vet Clin North Am Food Anim Pract. 1990;6:683.
8 Bulgin MS. J Am Vet Med Assoc. 1990;196:1120.
9 Ridler AL, et al. N Z Vet J. 2000;48:57.
10 Worthington RW. N Z Vet J. 1983;32:58.
11 Rahaley RS, Dennis SM. Aust Vet J. 1984;61:353.
12 Kimberling CV, Schweitzer D. Agri-Practice. 1989;10:36.
13 Dargatz DA, et al. J Am Vet Med Assoc. 1990;196:605.
14 Marin CM, et al. Res Vet Sci. 1990;48:209.
15 Manterola L, et al. Vet Microbiol. 2003;92:65.
16 Kittleberger R, et al. Vet Microbiol. 1998;59:213.
17 Vigliocco AM, et al. Vet Microbiol. 1997;54:357.
18 ficipal A, et al. Vet Rec. 1995;137:145.
19 Kittleberger R, et al. N Z Vet J. 1997;45:75.
20 Marin CM, et al. Vet Rec. 1989;125:504.
21 West DM, Bruce RA. N Z Vet J. 1991;39:29.
22 Gall D, et al. Small Rumin Res. 2003;48:173.
23 West DM, et al. N Z Vet J. 1993;41:82.
24 Searson JE. Aust Vet J. 1986;63:30.
25 Marin CM, et al. Am J Vet Res. 1989;50:560.
26 Blasco JM, et al. Vet Immunol Immunopathol. 1993;37:257.
27 West DM, et al. N Z Vet J. 1978;26:133.
28 Erasmus JA, Bergh EC. J S Afr Vet Assoc. 1985;56:205.
29 Blasco JM. Prev Vet Med. 1997;31:275.
30 Marin CM, et al. Res Vet Sci. 1990;48:209.
31 Blasco JM. Vaccine. 1993;11:1291.