Chapter 26 Diseases associated with protozoa
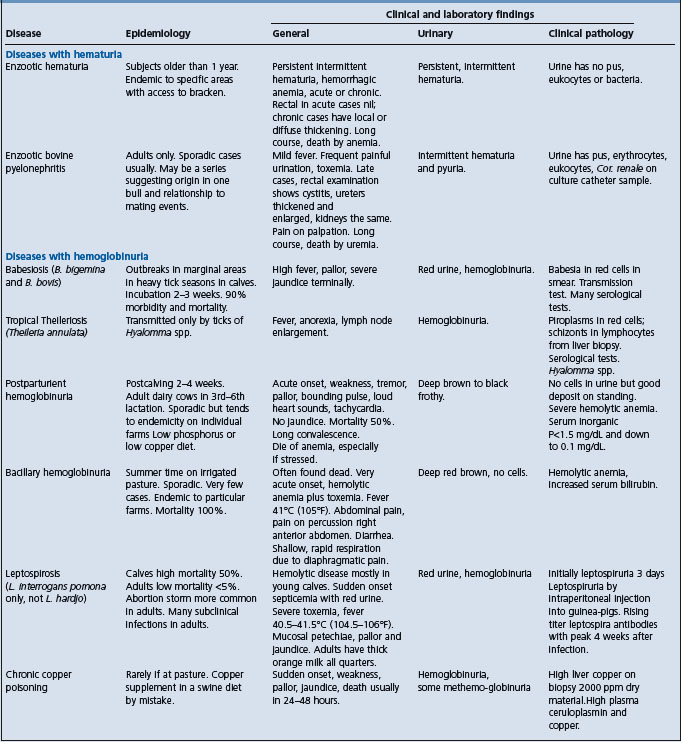
BABESIOSIS (TEXAS FEVER, REDWATER FEVER, CATTLE TICK FEVER, EQUINE PIROPLASMOSIS)
Babesia spp. are a diverse group of tick-borne, obligate, intra-erythrocytic Apicomplexan parasites infecting a wide variety of organisms. Infection of a vertebrate host is initiated by inoculation of sporozoite stage parasites into the bloodstream during the taking of a blood meal. Most babesial sporozoites directly invade circulating erythrocytes without a tissue stage of development. A few, notably, Babesia equi and Babesia microti, first invade lymphocytes where they form motile merozoites, which then invade erythrocytes, Once erythrocyte invasion occurs, a seemingly perpetual cycle of asexual reproduction is established, despite the rapid development of a strong immune response.
Epidemiology Disease of tropical and subtropical countries. Occurs in cattle, sheep and goats, horses, cervids, and pigs. Transmission by blood-sucking ticks. Young calves have innate resistance. Endemic stability occurs in herds with sufficient inoculation rate to immunize a high percentage of animals.
Zoonotic implications Babesia bigemina and B. microti occurs in humans where tick is found. Human donor blood may be infected.
Clinical signs Anemia, hemoglobinuria, jaundice, fever, high case fatality rate.
Clinical pathology Parasites in stained blood smear, positive serology. PCR for detection of parasite in blood.
Necropsy lesions Thin, watery blood, pallor, jaundice.
Diagnostic confirmation Parasites in blood smear; vector present in environment.
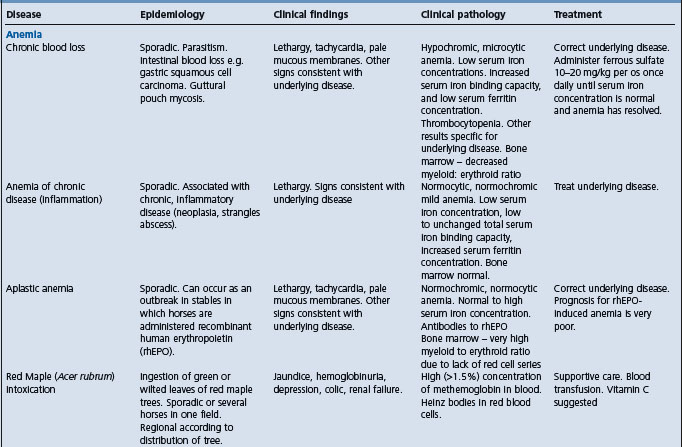
A syndrome of acute hemolytic anemia should suggest the following alternative diagnoses:
S-methyl-L-cysteine-sulfoxide (SMCO) poisoning
Foals with alloimmune hemolytic anemia
Cardiac form of African horse sickness.
Treatment Diminazene and imidocarb.
Control Tick control, vaccination with live vaccine, chemoprophylaxis with imidocarb.
ETIOLOGY
The nomenclature of these intra-erythrocytic parasites is still subject to change; the current list is:
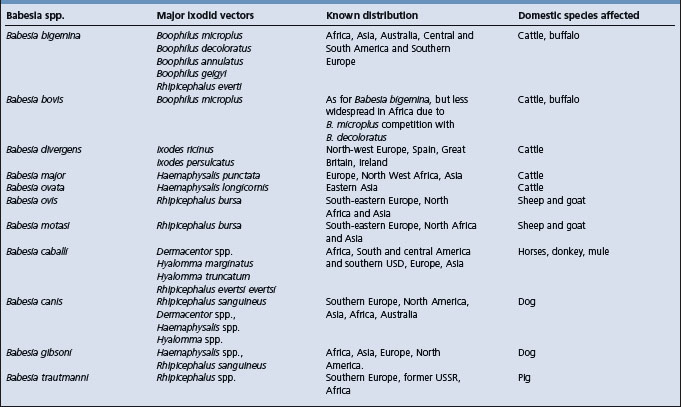
• Cattle: There are four species of bovine Babesia now recognized: Babesia bovis (includes B. argentina, B. berbera, B. colchica), B. bigemina, B. divergens (B. cauxsica, B. occidentalis, B. karelica) and B. major1
• Sheep and goats: B. motasi, B. ovis
• Pigs: B. trautmanni, B. perroncitoi
• Horses: B. equi, B. caballi. B. equi might be more accurately classified as a member of the Theileriidae family.
EPIDEMIOLOGY
Geographical occurrence
The distribution of the causative protozoa is governed by the geographical and seasonal distribution of the insect vectors that transmit them (Table 26.1)
Table 26.1 Major Babesia species infective to domestic animals, their tick vectors and geographical distribution22
Host occurrence
Bovine babesiosis
Bovine babesiosis associated with B. bigemina and B. bovis is an important disease of tropical and subtropical regions in the world, including the Americas. Both species are transmitted transovarially by Boophilus ticks, but only tick larvae transmit B. bovis, whereas nymphs and adults transmit B. bigemina.
In general terms, B. bigemina and B. bovis are infections which occur in countries in the tropics and subtropics between 40°N and 32°S. B. major and B. divergens occur in temperate regions.1,2 Thus, B. bigemina occurs in South America, the West Indies, Australia, and Africa; B. argentina in the tropics including South and Central America, Australia, Asia, and southern Europe. B. divergens occurs in north-west Europe, Italy,3 Spain, Eire, and is the principal cause of babesiosis in the United Kingdom. B. bovis occurs in Europe, South America, and Africa; B. berbera in Mediterranean Europe and North Africa; B. major in the United Kingdom and Europe. The first report of B. bovis in Spain occurred in 2000.4
Babesia divergens bovine babesiosis transmitted by Ixodes ricinus is widespread and reported often in France.5 The clinical incidence is low at 0.4% for the entire cattle population. The endemic situation is unstable and clinical cases occur more frequently with certain farming systems. Serology using immunofluorescence antibody test (IFAT), 7% of the cattle population is seropositive.6 Using the PCR, prevalence rate of carriers is 20%.
Bovine babesiosis is widespread in South Africa, and the distribution of both B. bovis and B. bigemina is determined by the distribution of their vectors. The seroprevalence of B. bigemina in non-vaccinated cattle is due to the high vector tick population and the endemically stable situation which can be achieved by adopting a tick-control method which allows a reasonable number of ticks on cattle rather than relying entirely on intensive tick control and vaccination.7
Sheep and goats
In sheep and goats, babesiosis is associated with B. ovis, B. motasi, and occurs in southeastern Europe, North Africa, and South America.8 In Iran, B. ovis, and B. motasi, occur in sheep and goats.9,10 The prevalence of B. ovis and B. motasi in sheep and goats were 23.5%. 0.5%, and 14%, 0.5%, respectively. In Iran, the clinical signs of babesiosis occurred in 8% of infected sheep and 6.8% of infected goats. Splenectomized sheep can be used as a model for B. divergens chronic infection.11
Sheep babesiosis is of considerable economic importance in the areas infested with Rhipicephalus bursa which is widely distributed in the Palaearctic region between 31–45° parallels North including the Mediterranean basin,8 the Balkans, the southern former USSR, Iraq, and Iran.
Porcine babesiosis
Associated with B. trautmanni and B. perroncitoi, porcine babesiosis occurs in southeastern Europe and Africa.
Equine babesiosis (piroplasmosis)
Babesiosis in the equine species is also known as equine piroplasmosis. In horses, donkeys, mules, and zebras the disease is associated with B. equi and B. caballi. It occurs in much of southern Europe, Asia, and the Americas. Equine piroplasmosis due to B. equi and B. caballi are widespread in China12,13 and cause for serious concern in northeast China. Australia is free of equine piroplasmosis but did allow the temporary importation of seropositive horses into the country for the Sydney Olympic games of 2000.14 While in Australia, seropositive horses were kept at certain restricted sites.
Seroepidemiologic studies of horse breeding farms in Brazil indicate the prevalence of Babesia caballi at 79%, Babesia equi 49% in mares; 36% of foals became infected with B. equi within 12 months but 100% with B. caballi within 10 months.15 Maternal antibodies against B. equi and B. caballi in foals were 44 and 68%, respectively. Titers persisted for 1–5 months for B. equi and 1–4 months for B. caballi.
In South Africa, equine piroplasmosis is a tick borne disease of horses, mules, donkeys and zebras, associated with Babesia caballi and Theileria equi. Equine piroplasmosis is widespread in South Africa. A serological survey indicated that of all serum samples collected from all parts of the country, nearly 80% were positive for T. equi, and 50% were positive for B. caballi which was cultured from horses.16
Wildlife babesiosis
Babesia odocoilei infects the cervid family including the white-tailed deer (Odocoileus virginianus) and the American elk and American woodland caribou (Rangifer tarandus caribou).17 Desert bighorn sheep (Ovis canadiensis nelsoni) and red deer (Cervus elaphus elaphus) are also susceptible to infection but do not exhibit clinical signs of disease.17 B. odocoilei is transmitted by ticks, Ixodes scapularis and Ixodes dammini.
Fatal babesiosis in domestic reindeer associated with Babesia tarandirangiferis was first described in northern Russia in 1909. B. divergens has caused babesiosis in reindeer in Scotland.18 Two morphologically dissimilar Babesia spp. have been cultured from reindeer in California.19
Origin of infection and transmission
Viable protozoa are present only in the bloodstream of animals in the active stages of the infection. Ticks are the natural vectors of babesiosis; the causative parasites persist and pass through part of their life cycle in the invertebrate host. Both B. bovis (Argentina) and B. bigemina pass part of their life cycle in the tick Boophilus microplus (recently reclassified as Rhipicephalus sp., but the name Boophilus will be used). Boophilus (Margaropus) annulatus and B. microplus are the major vectors of babesiosis, but other Boophilus, especially B. decoloratus in South Africa, Rhipicephalus, and Haemaphysalis spp. also act as vectors. Boophilus microplus is the main vector babesiosis associated with B. bovis and B. bigemina in cattle production systems in Central and South America.20 Ixodes ricinus is the common carrier of B. divergens in the United Kingdom. Rhipicephalus bursa8 and Haemaphysalis punctata spp. are the vectors in sheep; Dermacentor, Rhipicephalus, and Hyalomma spp. in horses; and Rhipicephalus and Boophilus spp. in pigs.
Information on the natural or experimental tick vectors of equine babesiosis is limited. Dermacentor nitens is the only tick species that transmits B. caballi in horses in the New World. In Brazil, Ambylomma cajennense and Anocenter Dermacentor) nitens are the most common and widespread ticks infesting horses, A. nitens is an important natural vector of B. caballi. Boophilus microplus, is the dominant tick in some areas where B. equi infection is endemic. Using conventional diagnostic methods, B. microplus collected from horses were negative for both B. equi and B. caballi. Using a nested PCR, B. equi and B. caballi DNA were detected in the blood samples of horses and in the ticks. The detection of specific B. equi and B. caballi DNA in the eggs and larvae of B. microplus suggests the possibility of both transovarial and transstadial parasite transmission.21
In Iran, five ixodid species of ticks have been collected from sheep and goats.9 The Rhipicephalus sanguineus and Hyalomma marginatum are the most common species in sheep and goats. Other tick vectors include Dermacentor daghestanicus in goats and Hyalomma anatolicum, Hyalomma asiaticum in sheep.10
When adult animals become infected they act as carriers for variable periods, up to 2 years. If they are constantly reinfected, as they are in an endemic environment, they act as carriers for life.
A knowledge of the life history of the tick is most important in applied control. Those ticks that parasitize only one host are easier to eradicate and cause less spread of the disease than those parasitizing two or three hosts. Control of ticks capable of surviving on both domestic and wild animals presents a major problem.
Life cycle and development of Babesia
The development of B. bovis and B. bigemina follow similar patterns in adult Boophilus spp. Babesia spp. do not parasitize any vertebrate host cell other than erythrocytes. Each sporozoite (merozoite) penetrates the cell membrane of an erythrocyte with the aid of a specialized apical complex. Once inside, it transforms into a trophozoite from which two merozoites develop by a process of merogony (binary fission) (see Fig. 26.1).
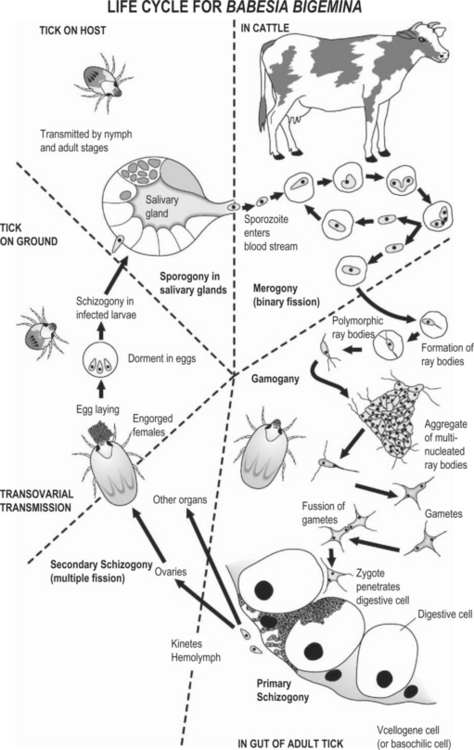
Fig. 26.1 The development life cycle of Babesia bigemina in cattle and the ixodid tick vector Boophilus microplus
(adapted from Mehlhorn, Shein 1984; Mackenstedt et al. 1995; Gough et al. 1998).
In the passage of host blood to the midgut of the tick vector, the development of two populations of ray bodies from the gamonts (gametocytes) occurs.22 The ray bodies undergo further multiplication within the erythrocytes which continues after they have emerged. Large aggregations of multinucleated ray bodies form, but once division is complete, single-nucleated ray bodies that are now haploid and assumed to be gametes emerge from the aggregates and then fuse together in pairs (syngamy) to form a spherical cell (zygote). The zygote selectively infects the digestive cell of the tick gut where they multiply and then the basophilic cells where further multiplication occurs with development to kinetes that escape into the tick hemolymph. In the gut cells, schizogony occurs with the formation of polyploid kinetes (large merozoites). These motile club-shaped kinetes then escape into the hemolymph and infect a variety of cell types and tissues, including the oocytes where successive cycles of secondary schizogony occurs. Thus, transovarial transmission occurs with further development occurring in the larval stage. Kinetes enter the salivary glands and are transformed into multinucleated stages (sporogony) and these then break up to form sporozoites. In all species, sporozoite development usually only begins when the infected tick attaches to the vertebrate host. In B. bigemina, some development occurs in the feeding larvae, but infective sporozoite take about 9 days to appear and therefore only occur in the nymphal and adult stages of the tick. Transmission can occur throughout the rest of the nymphal stage and by adult females and males. For B. bovis, the formation of infective sporozoites usually occurs within 2 to 3 days of larval tick attachment.
Contaminated needles and surgical instruments can transmit the infection physically. The ease with which infection can be transmitted in this way depends largely on the degree of parasitemia occurring with each species. Thus, the chances of physical transmission are slight with B. bovis and high with B. equi and B. bigemina.
Immunity and susceptibility to infection
The immune response of cattle to infection with B. bovis or B. bigemina involves both innate and acquired immune mechanisms22 The immune response directed against infections with Babesia involves both humoral and cellular mechanisms and is T-cell dependent. In addition, an age-related immunity to initial infection with B. bovis in cattle is well established, characterized by strong innate immunity in young calves. Mononuclear phagocytes are engaged as the primary effector cells on innate and primary immune responses and nitric oxide has been identified as at least one babesiacidal molecule produced by activated mononuclear phagocytes. When B. bovis infected erythrocytes grown in culture are exposed to nitric oxide, death of the parasites occurs rapidly within the erythrocyte.
Innate immune mechanisms
There is an age-related immunity to primary infection of cattle with B. bovis and B. bigemina. Young calves possess this strong innate immunity against B. bovis infection that lasts for approximately 6 months after birth and is abrogated with the removal of the spleen.23 Interleukin IL-12 and IL-10 are important immunoregulatory cytokines.24 The protective innate response in young calves to infection with virulent B. bovis involves the early appearance of IL-12 and interferon-((IFN-() transcripts in the spleen. This is followed by a brief period of inducible nitric oxide synthase expression.23 In contrast, IL-12 and IFN-(mRNA) expression in the spleens of adult cattle which died from infection was delayed and depressed and occurred within the context of IL-10 expression. Also, in contrast to calves, there was no detectable antibody response before death in adults.
Acquired immune mechanisms
Following B. bovis infection, antibodies directed against protective and non-protective parasite antigens and host antigens are produced.22 Hyperimmune serum from cattle infected with B. bovis many times, or a mixture of IgG1 and IgG2 prepared from hyperimmune serum of cattle can be used to immunize naïve calves passively against B. bovis infection, and the protection is strain specific. Splenectomized calves given hyperimmune serum and challenged with B. bovis recover as effectively as intact calves.
Strong immunity occurs after natural infection with most Babesia spp. There appears to be little relationship between the degree of immunity and the level of antibodies in the serum. If the infection recurs repeatedly the immunity is permanent. If the illness is treated urgently and efficiently, and the protozoa are killed before antibodies are produced, no immunity occurs. If the infection is not repeated the protozoa survive in the host for a variable time, usually about 6 months, and then disappear. A sterile immunity persists for a further 6 months and the host is susceptible again about a year after infection occurred. These periods of latent infection and resistance to reinfection are subject to significant variation and to different responses between breeds of cattle and the species of Babesia.
Despite the potential severity of the acute infection, individuals who survive generally develop immunity against disease, but not against infection, and could remain persistently infected. In the case of B. bovis, infections can persist for years, and even for the lifetime of the animal. Babesial infections have adapted well to survival in immune hosts. At least five different phenomena are known to contribute to parasite survival: rapid antigenic variation; cytoadhesion and sequestration; binding of host proteins to the infected red blood surfaces; the monoallelic expression of different members of multigene families; and establishment of transient immunosuppression.25
The inoculation rate measures the daily probability of infection. This is based on the knowledge that animals exposed to the parasite in the first 9 months of life become infected, immune and seropositive without showing any clinical signs of disease.20 Inoculation rates of 0.0005 and 0.005 are endemically unstable because a high percentage will reach the age of 9 months without having been exposed to the hemoparasite. This results in a high risk of disease (endemic instability), as primary infections in older animals are usually severe and can be fatal. A serological survey of cattle in Bolivia for B. bovis and B. bigemina to estimate the risk of outbreaks by calculating the inoculation rates of each hemoparasite. The results indicated the area surveyed is endemically unstable as h values were below 0.005.20 In such unstable conditions, calfhood vaccination against hemoparasites is recommended to ensure that the herd is immune. Alternatively, cattle producers may use tick control to break the transmission cycle; however, tick control is much more costly and risky if tick control fails.
Endemic stability is defined as the state where the relationship between host, agent, vector and environment is such that clinical disease occurs rarely or not at all.22 Endemic stability (herd immunity) in bovine babesiosis occurs when the rate of transmission (inoculation rate) of Babesia spp. by the tick vector is sufficient to immunize a majority of susceptible calves before the loss of calfhood resistance.26 In tropical areas with a high vector population, natural exposure usually occurs at an early age and cattle are therefore immune to subsequent challenges as adults. If at least 75% of calves are exposed to B. bovis infection by 6 to 9 months of age the disease incidence will be very low and a state of natural endemic stability would exist.
protection. Protective cross species immunity against infection cannot be induced with B. bovis and B. bigemina.22
Cattle develop a durable long-lasting immunity after a single infection with B. divergens, B. bovis, or B. bigemina. Immunity to both B. bovis and B. bigemina lasts at least 4 years. There is evidence in the literature suggesting the presence of antibodies is not necessarily an indication of immunity nor is absence of detectable antibodies necessarily an indication of a lack of immunity.22
Risk factors
Host factors
Bos indicus breeds of cattle are much more resistant to babesiosis than Bos taurus breeds. This phenomenon is thought to be a result of the evolutionary relationship between Bos indicus cattle, Boophilus spp. and Babesia.22 Zebu and Afrikaner cattle have a higher resistance to B. bovis than British and European breeds; Santa Gertrudis and cross-bred cattle occupy an intermediate position. Zebu-type cattle also enjoy a relative freedom from the disease because of their resistance to heavy infestations with ticks.
In Australia, B. bigemina is usually of lower pathogenicity than B. bovis and rarely lethal even when fully susceptible adult cattle are introduced to an endemic area.27 Inoculation studies with B. bigemina in Australia have shown that B. indicus and B. indicus cross cattle are more resistant than the B. taurus cattle.27
Age resistance There is a variation in susceptibility to infection according to age in cattle. The severity of clinical babesiosis increases with age. Calves and foals from naïve dams are highly susceptible to infection and clinical illness from birth to 2 months of age, at which time they develop an innate resistance that persists to about 6 months of age. Calves and foals from immune dams receive antibodies via the colostrum, and this passive immunity persists for 3–4 months after birth. The greatest infection rate is in animals in the 6 to 12-month age group; infection is uncommon in animals over 5 years of age. Animals under 1 year of age are infected predominantly with B. bigemina and those over 2 years of age by B. bovis. Calves up to 1 year of age, although fully susceptible to infection, are resistant to disease.2 The average age at which calves in endemic areas become infected is 11 weeks (2–34 weeks), but at this early age clinical signs and pathological changes are mild and short-lived. After 6 months of age the number of infected animals in enzootic areas increases.
In housed cattle, the level of antibodies in the patient are at their lowest when the cattle come out of the barn in the spring, and gradually increase as they are exposed to vector ticks.
In enzootic areas, the animals most commonly affected by clinical disease are susceptible cattle introduced for breeding purposes, for slaughter, or in transit. Cattle indigenous to these areas are rarely affected because the natural resistance of the very young, and passive immunity via colostrum from immune dams is gradually replaced by a state of active immunity. Severe clinical cases occurring in these cattle are usually caused by exposure to some stress, such as parturition, starvation or intercurrent disease. Such breakdowns in immunity are most likely to occur if there is a superimposed infection with a different parasite, especially Anaplasma marginale.
Environmental factors
There is a seasonal variation in the prevalence of clinical babesiosis, the greatest incidence occurring soon after the peak of the tick population. For example, in England babesiosis is largely a disease of spring, summer, and autumn for this reason. Of the climatic factors, air temperature is the most important because of its effect on tick activity – higher temperatures increase it; humidity and rainfall have little effect – and even with temperature the effect is limited once a threshold of 7–100°C (44–50°F) minimum temperature is exceeded. Heaviest losses occur in marginal areas where the tick population is highly variable depending on the environmental conditions. In seasons when the tick population decreases, infection may die out and immunity be lost. Then in favorable seasons when ticks multiply, the infection spreads quickly amongst what has become a susceptible population. Comparable circumstances may be created artificially by an inefficient dipping program, which reduces the tick population to a low level and is subsequently unable to keep it under control.
Pathogen factors
Many intra-erythrocytic hemoparasites survive the host immune system through rapid antigenic variation which has been demonstrated for Babesia bovis and Babesia rodhaini.28 The molecular basis for antigenic variation in babesial parasites and its possible connection with cytoadherence and sequestration have been examined.28 The existence of different strains and antigenic variation occur in both B. bovis and B. bigemina. Babesial infections in cattle by antigenic variation and by superinfection with antigenically different parasite populations. Each change in antigenic type provides a temporary respite from attack by the host immune system and prolongs the infection period. The number of antigenically distinct relapses which can occur in a herd with babesial infection could be more than 100.
Strain differences and antigen variation do not appear to be of major importance as a cause of disease or in vaccines, since cross-immunity tests between strains usually provide adequate clinical protection against each other. Babesia bigemina sporozoites expressing specific antigens which induce protective immunologic responses in cattle have been characterized.29
New sequences of Spanish isolates of B. caballi and B. equi show a relatively high degree of genetic divergence within the group of piroplasms.30 The immunoreactive polypeptides of B. equi merozoite antigen have been identified.31
Economic importance
Bovine babesiosis is the most economically important of these diseases, because of direct losses of production and because of restriction of movement of cattle for trade by quarantine laws. Many animals die or undergo a long period of convalescence entailing loss of meat and milk production. Incidental costs of immunization and treatment add to the economic burden. With early, effective treatment the mortality rate can be reduced to 5%.
The mortality rates in outbreaks of equine babesiosis are high, but the big losses in this species result from the interference with racing and pleasure horse meetings and competitions. This is especially the case at present with the movement of horses between countries to compete in increasingly international equine competitions. An additional form of loss is the death of foals infected in utero. In the 1960s with an outbreak of clinical equine babesiosis in the United States, several cases in Australia, and several seropositive identifications in the United Kingdom, it appeared to be an emerging disease which threatened to be of major importance to the horse industry, but this has not eventuated.
The morbidity and mortality rates and the losses associated with babesiosis in other animal species are difficult to determine because they exist as enzootic diseases in areas where they occur.
Zoonotic implications
Human cases of B. divergens infection have been reported in France, Britain, Ireland, Spain, Sweden, Switzerland, the former Yugoslavia, and the former USSR.2 Geographically, they coincide with B. divergens-infected cattle populations and Ixodes ricinus-infested areas, involving inhabitants of rural areas who are exposed to ticks by virtue of their occupation or their recreational activities. Most cases are reported between May and October, during main season of tick activity. B. divergens is the primary cause of human babesiosis in Europe, resulting in fatality rates of 42% among persons who have been splenectomized and 5% among those with intact spleens. B. divergens has caused human babesiosis in North America in a splenectomized person which suggests that B. divergens may be emerging in North America in areas where such infections are not endemic.32 The known vector tick, I. ricinus is not indigenous to North America.
There is evidence that human babesiosis in the US has been associated with B. microti, a parasite of white-footed mice, is transmitted by deer ticks.33 Deer-associated zoonoses have become a major public health concern in the United States because human contact with deer ticks has increased as a result of the proliferation of deer, abandonment of farmland that reverts to thick secondary vegetation, and increased use of coastal sites for human recreation. This explains the increasing frequency of reported human cases of Lyme disease, babesiosis and human granulocytic ehrlichiosis.32
Babesia represent a potential threat to the blood supply for transfusion since asymptomatic infections in humans are not uncommon and spread of the parasite via blood transfusions has been reported from various countries.34 Using the microaerophilous stationary phase (MASP) culture technique, the parasites proliferate in a settled layer of blood cells. This provides the opportunity to examine the basic biology of the organism, as well as the host-microbe interactions, immune factors triggered by the parasite, factors involved in innate resistance of young animals to infection, and antimicrobial susceptibility. Their in vitro cultivation can produce quantities of parasite nucleic acid needed for defining phylogenetic relationships of these species, developing methods for detection of the parasite in otherwise asymptomatic individuals, and producing parasite antigens and attenuated strains of Babesia that could be used for immunization.
PATHOGENESIS
Babesia spp. are a diverse group of tick-borne, obligate, intra-erythrocytic Apicomplexan parasites infecting a wide variety of organisms. Infection of a vertebrate host is initiated by inoculation of sporozoite stage parasites into the bloodstream during the taking of a blood meal. Most babesia sporozoites directly invade circulating erythrocytes without a tissue stage of development. Once erythrocyte invasion occurs, a perpetual cycle of asexual reproduction is established despite the rapid development of a strong immune response.25
Acute cases
When an animal becomes infected, multiplication of the protozoa in the peripheral vessels (B. bigemina, B. ovis), or in the visceral vessels (B. bovis), reaches a peak with the development of clinically detectable hemolysis, the principal pathogenic effect, after an incubation period of 7–20 days. The hemolysis results in profound anemia, jaundice, and hemoglobinuria. A fatal outcome due to anemic anoxia commonly follows. In longer surviving animals there are ischemic changes in skeletal and heart muscle.
In B. bovis infections there is also a profound vasodilation and hypotension, resulting from stimulation of production of vasoactive substances, and an associated increase in vascular permeability. Circulatory stasis and shock follow; disseminated intravascular coagulation (DIC) and subsequent, fatal pulmonary thrombosis are also features. Cerebral babesiosis is possible.
B. bigemina is an uncomplicated hemolytic agent and does not exert these vascular and coagulation effects.
Susceptibility to infection with Babesia spp. decreases with age, but the severity of the clinical disease increases. For example, calves up to 5–6 months of age, and infected with B. bovis, show little effect; cattle of 1–2 years of age have a moderately severe disease; and aged cows suffer a severe, often fatal, clinical disease. Intrauterine infection of 2-day-old calf with Babesia bovis has been reported.35
B. odocoilei infection in elk causes an acute hemolytic anemia which may be fatal.17
Animals which survive become carriers, a state in which a harmless, subclinical infection is maintained by a delicate immunological balance between protozoa and antibodies. This balance is readily disturbed by the stress of transport, deprivation of food, pregnancy, or intercurrent disease. Carrier animals are resistant to infection with B. bovis for up to 2 years. With constant reinfection, such as occurs in an enzootic situation, the protection is continuous, but the virulence of the blood in transmission experiments varies due to periodic disappearance of infective forms of the parasite from the peripheral blood.
The ability of cattle to infect ticks is much longer (1 year) with B. bovis than B. bigemina (4–7 weeks). Similarly, the peak incidence is at a younger age and the reinfection rate is faster with B. bigemina.
In pregnant cows there is no apparent infection of the calf in utero, but passive immunity is transferred via colostrum to the newborn calf.
Immunology
Calves less than 9 to 12 months of age are as susceptible as adult cattle to infection with B. divergens but are less likely to exhibit clinical disease. This phenomenon known as inverse age resistance is due to innate resistance in calves and is independent of the maternal immune status. Although offspring of resistant dams acquire specific antibodies (mainly IgG) via colostrum, these immunoglobulins are not necessary for protection because calves of susceptible dams without specific antibodies are equally resistant. In vitro studies with B. bovis show that erythrocytes of very young calves were unfavorable to parasitic development, possibly because of the inhibitory effect of fetal hemoglobin.2
Cattle that recover, either naturally or after chemotherapy, from acute infection with B. bigemina or B. bovis remain persistently infected and resistant to further disease upon reinfection with the same strain.36 Immunization with killed parasites or parasite extracts can afford protection against homologous and heterologous strain challenge, indicated by low parasitemias and diminished reduction in packed cell volume.
Immunity does not last indefinitely, and in the absence of exposure to further infection, the animal becomes susceptible to reinfection. Specific immune mechanisms include both cellular and humoral components. Monocytes and lymphocytes are the main agents of cell-mediated immunity. Experimentally, the exposure of cattle to avirulent and virulent strains of B. bovis causing a primary infection, results in considerable antimicrobial activity in peripheral blood monocytes and neutrophils.37 The elevated antimicrobial activity is coincident with the time that parasite numbers peaked in the circulation and occurs prior to parasite clearance. This suggests that peripheral blood monocytes and neutrophils are active mediators in the innate immune response to a primary infection with B. bovis. In cattle vaccinated against B. divergens, protection is correlated with elevated mononuclear cell proliferation.
In cattle infected with B. divergens, antibodies can be demonstrated even before infected erythrocytes appear in blood smears, indicating that they have no inhibitory effect on parasite multiplications. During secondary infections, protection seems to depend on the high specificity of some antibodies rather than the total level of anti-B. divergens antibodies, as resistant animals frequently have very low levels of specific antibodies.
The importance of the spleen in the specific immune response is illustrated by the fact that removal of the spleen following recovery may result in clinical relapse.
Specific antibodies to the parasites are produced and are used in serological diagnosis. The highest titers are obtained in the sera of cows that have had a series of infections and reinfections, but the degree of immunity resulting is not related to the antibody titer. The antibodies can be passively transferred via serum or colostrum. The immunity to each strain of B. bovis is specific. However, when an infection with a heterologous strain of the protozoa occurs, there is an increased immune response.
Experimental transmission of B. ovis infection in sheep produces an acute attack of clinical illness, parasitemia, and the subsequent development of immunity, as in cattle. Experimental infection of pregnant cattle with B. bigemina results in an immune response similar to nonpregnant animals.38
CLINICAL FINDINGS
Cattle
The acute disease generally runs a course of 3 to 7 days and a fever of >40°C is usually present for several days before other signs become obvious.22 This is followed by inappetence, depression, polypnea, weakness and a reluctance to move. Hemoglobinuria is often present (known as redwater in some countries); urine is dark-red to brown in color and produces a very stable froth. Anemia and jaundice develop especially in more prolonged and severe cases. Diarrhea may occur. Muscle wasting, tremors and recumbency develop in advanced cases followed terminally by coma. Many severely affected animals die precipitately at this point, after an illness of only 24 hours. Metabolic acidosis is present in a significant percentage of cases of bovine babesiosis in Ireland.39 During the fever stage, pregnant cattle may abort and bulls may become sterile for 6 to 8 weeks. Cerebral babesiosis is manifested by incoordination followed by posterior paralysis, or by mania, convulsions and coma. The mortality rate in these cases is high in spite of treatment.
In those that survive, the febrile stage usually lasts for about a week and the total course about 3 weeks. Animals that survive recover gradually from the severe emaciation and anemia, which are inevitable sequelae.
A subacute syndrome also occurs, especially in young animals, in which the fever is mild and hemoglobinuria is absent. The syndrome in infection with B. divergens is similar to the above, except that in addition, there is spasm of the anal sphincter causing the passage of feces with great force in a long, thin stream, even in the absence of diarrhea. The sign is referred to as ‘pipe-stem’ feces.
Hemoglobinuria is present earlier and more consistently than in B. bovis infections and the fever is less of a feature. Acutely affected animals are usually not as severely affected as those with B. bovis infections. There is no cerebral involvement and recovery in non-fatal cases is usually rapid and complete. However, in some cases the disease can develop very rapidly with sudden and severe anemia, jaundice and death.22 Animals which recover from B. bigemina remain infective for ticks for 4 to 7 weeks and carriers for only a few months.
Horses
The incubation period is 8–10 days. Acute cases in adults show a sudden onset of immobility and reluctance to move; some are in lateral recumbency and do not respond to stimuli. There is complete anorexia and fever of 40°C (104°F), although the fever often subsides after 1 day and becomes intermittent. Edema of the fetlocks occurs and may also be present on the head and ventral abdomen. Fecal balls are covered with thick mucus, and colic occurs frequently. Often there is no hemoglobinuria; bronchitis occurs occasionally. The mucosae are pale pink and tinged with jaundice. In young horses, the signs are more severe – jaundice, mucosal pallor and weakness are marked, and mucosal petechiae are evident. The course is 8–10 days. Afflicted horses may die within 24–48 hours of the first signs appearing. Chronic cases may survive for months and ‘carriers’ may persist for as long as 4 years. The experimental disease produced by B. equi is mild. A high percentage of erythrocytes are parasitized by the protozoa and the horses are anemic, but there is no clinical evidence of anemia. Newborn foals develop severe jaundice and severe prostration, sometimes delayed in onset by 2 or 3 days after birth.
Wildlife
Babesiosis in elk and caribou are characterized clinically by lethargy, hemoglobinuria, icterus, fever, recumbency, and sudden death.17 Elk infected with B. odocoilei may not have any clinical signs of disease, but may become ill during periods of stress, such as the rutting season, calving, transportation, or overcrowding.
CLINICAL PATHOLOGY
Clinical cases
Hematology
Severe anemia with erythrocyte counts as low as 2 million/μL and hemoglobin levels down to 3 g/dL occur in clinical cases in cattle and horses, the anemia peaking 9–16 days after infection occurs. Significant falls in platelet counts and a depression in the fibrinogen content of the blood also occur.
Demonstration of Babesia
Direct examination of blood smears.
A diagnosis of existing babesiosis in clinically affected animals of all species depends on the demonstration of protozoa in a Giemsa-stained smear of capillary blood; venous blood may give a false-negative in B. bovis infections. There is no exact correlation between the percentage of erythrocytes containing protozoa and the severity of the clinical signs. Also in B. bigemina infections, protozoa are numerous in peripheral capillaries; B. bovis is much less readily found. This difficulty can be largely overcome by the use of thick blood smears. Microscopic examination can detect parasitemia of about 105 in thin blood films and 106 in thick blood films.
For best results, blood films should be prepared from capillary blood collected after pricking the tip of the tail or margin of the ear. Blood from the general circulation may contain 20 times fewer B bovis than capillary blood. Thick blood films are 10 times more sensitive and are more reliable for the detection of low level B. bovis infection.22
Subinoculation of blood to susceptible splenectomized calves is highly sensitive technique for direct detection of Babesia infection. In transmission tests, 50–100 mL of blood are injected into the recipient either SC or IV. In the latter case, the incubation period will be shorter. The recipients are examined daily and the blood examined for protozoa at the peak of the febrile reaction.
Carrier cattle infected with B. bovis and B. bigemina are difficult to detect because of the small number of parasites in peripheral blood. Microscopic examination of blood films is not reliable technique for detection of Babesia-carrier animals. The evaluation of persistence of B. bovis and B. bigemina infections require subinoculation of blood into splenectomized calves, and measurement of the anti-babesial antibody level.40
Babesia divergens from the blood of carrier cattle can be isolated using an in vitro culture technique in sheep erythrocytes.41 The protozoa could be isolated 9 months after the acute babesiosis phase, and can be successfully subcultured, cryopreserved and resuscitated using culture medium. This will allow for more detailed examination of the organism.
Preservation of live protozoa can be effected by cryopreservation, by culture in a medium containing infected bovine erythrocytes, and in simple culture media in special machinery for long periods and in large quantities.
PCR detection and identification of Babesia spp.
A universal PCR assay for the detection and identification of nine of the most common pathogenic bovine, equine, and rodent piroplasms, including B. divergens. Following specific amplification of the parasite DNA by nested PCR, the parasite species is identified by PCR-restriction fragment length polymorphism.2 Various applications of the PCR have detected B. bovis and B. bigemina parasitemias at levels of 10−7 to 10−9.
The most recently introduced tests are an ELISA using a recombinant B. bovis antigen, PCR and a DNA probe, which can detect specific parasitemias at very low levels of infection. The DNA probe has the added advantage of being able to detect protozoa in necropsy specimens and in tick tissues. The PCRs are most useful because of their high sensitivity, which makes them ideal for the detection of carrier animals.
A PCR assay can detect B. equi and B. caballi from the blood of horses which have recovered from the acute phase of babesiosis. The may assume a subclinical, chronic course and the animals are carriers and may act as reservoirs of infection, and parasites are present in very low numbers in the blood and generally not detectable in Giemsa-stained blood-smears.42 In these horses the complement fixation test which is the official test for the diagnosis of babesiosis in horses may be negative and the PCR test is positive. A nested PCR assay has been used to detect natural infection of Boophilus microplus and the blood of horses with B. equi and B. caballi in Brazil.21 The nested PCR is considered superior to both Wright–Giemsa-stained and primary PCR methods for the routine detection of B. equi in horses.43
Serology
Diagnosis of past or present infection is adequately demonstrated by any one of a wide range of serological tests.
Bovine
Because of the difficulty in finding protozoa in smears in animals during the subclinical stages of the disease, especially in surveillance studies for the detection of the infection in herds or areas, much attention has been directed to serological tests. These are now well-established, but none of them enjoys a completely satisfactory reputation.
Complement fixation test (CFT).
The CFT has been the most used serological test for bovine babesiosis. Other tests being assessed in field conditions include a passive agglutination test, an indirect fluorescent antibody test (IFAT), an indirect hemagglutination test, an ELISA, a microplate enzyme immunoassay (EIA), a latex agglutination test, a capillary agglutination, a slide agglutination, and a card agglutination test. All of the tests have good reputations, with the EIA being probably the most sensitive.
Immunofluorescence antibody test (IFAT).
The IFAT has been a popular test used to distinguish between Babesia spp. and to demonstrate the presence of antibodies in a population. IFAT clearly differentiates between antibodies to B. divergens and other bovine babesias but not between B. divergens and B. capreoli from red deer.
An ELISA system using a crude antigenic preparation of B. bovis has been standardized for the detection of IgM antibodies with a specificity of 94% and sensitivity of 100%.44 Specific IgM antibodies against B. bovis first appeared on the 11th day post-inoculation in animals infested with B. microplus ticks and on the 19th day post-inoculation in which animals had been inoculated with infected blood.
A competitive ELISA (cELISA) is an accurate, reliable, easily standardized, and high-throughput method for detecting hemoparasite infections.45 The gene encoding B. bovis rhoptry-associated protein 1 (RAP-1) was used to develop the assay.46 The cELISA accurately differentiated animals with B. bovis-specific antibodies from uninfected animals and from animals with antibodies against other tick-borne hemoparasites (sensitivity 98.5%, 98.7% specificity).
Equine
In the horse, the tests used include the widely used complement fixation test, and the recently introduced, still undergoing testing, ELISAs and DNA probes. Important aspects of serological testing in horses is its application in the implementation of import and export regulations, in deciding the action to be taken in releasing very valuable horses from quarantine, and in deciding whether or not to permit entry of individual horses into non-enzootic areas.
A latex agglutination test (LAT) using recombinant B. equi merozoite antigen 1 (EMA-1) has been developed for the detection of antibodies to B. equi.47 It is a simple, rapid, sensitive, specific and inexpensive alternative to IFAT or ELISA.
Because of the importance of testing individual horses, in the process of babesiosis control the culture of blood from suspect horses is now used to determine whether or not they are carriers of B. equi.
NECROPSY FINDINGS
In acute cases of babesiosis in all species, in which patients die after a brief illness and during an anemic crisis, the typical lesions are jaundice, thin watery blood, pale tissues, enlargement of the spleen which has a soft, pulpy consistency, and gross enlargement and dark brown discoloration of the liver. The gallbladder is distended with thick, granular bile, the kidneys are enlarged and dark, and the bladder contains red-brown urine. Ecchymotic hemorrhages are present under the epicardium and endocardium, and the pericardial sac contains an increased quantity of blood-stained fluid. A characteristic lesion in both cattle and horses is severe intravascular clotting.
In subacute or chronic cases of fairly long duration, the carcass is emaciated but hemoglobinuria is absent; the other changes observed in acute cases are present but less pronounced. Laboratory examination of smears taken from peripheral blood, from kidney and heart muscle and, in the case of suspected B. bovis infection, from the brain, is mandatory for clinching the diagnosis. The smears from blood and most tissues must be made within 8 hours of death, in the case of brain within 28 hours, and stained with Giemsa for the detection of B. bovis.
Direct fluorescent antibody staining of smears permits the use of slightly older tissues. Organ smears are still usable 5 days after collection provided they are kept stored at 22°C (72°F). With B. bigemina the morphology of the parasite changes quickly after the host’s death so that they resemble B. bovis. Blood serum collected after death can also be used for detection of antibodies in serological tests.
For diagnostic confirmation the presence of the insect vector must be verified before the diagnosis of babesiosis can be made, unless the animal has left an enzootic area within the preceding month. Clinically, a high morbidity and case fatality rate in cases showing jaundice with hemoglobinuria and fever are suggestive, but confirmation of the diagnosis by examination of blood smears or by transmission experiments is essential. A necropsy showing splenomegaly, jaundice, hemoglobinuria, swollen dark kidneys and liver, and myocardial ecchymoses, while highly suggestive, should also be confirmed by laboratory examination of tissues for the presence of the causative protozoa.
Differential diagnosis list
A syndrome of acute hemolytic anemia should suggest the following alternative diagnoses:
Cattle (see Table 26.2):
Horses (see Table 26.3)
TREATMENT
Primary treatment is aimed at destruction of the protozoa in the patient. Effective drugs are available for this purpose in cattle, but the initial phase of the disease is acute; if treatment is delayed for too long the animal may succumb to the anemia in spite of sterilization of the blood. When the illness is a consequence of vaccination with live vaccine, care must be taken to avoid complete sterilization of the blood before sufficient antibody is produced to provide a durable immunity. Treatment has no suppressing effect on the protozoa that are residing in the ticks parasitizing the cattle at the time.
A summary of the recommended drugs follows, but diminazene aceturate, imidocarb dipropionate, amicarbalide diisethionate, and phenamidine are most often used. Parvaquone, buparvaquone, and alovaquone are recent introductions with good reputations from clinical trials. Tetracyclines have been used extensively, but their use in acutely sick animals has been discontinued. They find some use in simultaneous administration with living babesia in a chemosterilant situation; the parasite is controlled but effective immunization is achieved.
Cattle
For many years, three babesicides, quinuronium sulfate and several generics of it, amicarbalide isoethionate, and diminazene aceturate were available in most countries for the treatment of bovine babesiosis. In the 1970s a fourth, imidodocarb dipropionate was introduced, and it rapidly became the drug of choice in those countries that licensed it, because in addition to its therapeutic utility, it also proved to be an effective prophylactic at twice the therapeutic dose.2 Currently, it is the only babesicide on the market in most of Europe. Quinuronium sulfate and amicarbilide have been withdrawn because of manufacturing safety issues, and diminazene, which is widely used in the tropics as both a babesicide and a trypanocide, was withdrawn in Europe for marketing reasons.
Imidocarb is most toxic when given IV, and IM and SC administration is generally recommended. Side effects include coughing, muscular tremors, salivation, colic, and local irritation at the site of injection following high doses. While it is regarded as being slower in action than quinuronium sulfate it is the only babesicide that consistently clears the host of parasites. In the past, the persistence of small numbers of parasites in the bloodstream was deemed necessary for the maintenance of resistance to reinfection. However, the concept of premunition is no longer accepted. Premunition is used to describe resistance that is established after the primary infection has become chronic and is only effective if the parasite persists in the host. It was thought that only cattle actually infected with Babesia were resistant to clinical disease. If all organisms were removed from an animal, resistance was thought to wane immediately. However, cattle cured of Babesia infection by chemotherapy are resistant to challenge with the homologous strain of that organism for several years. But the presence of infection does appear to be mandatory for protection against heterologous strains.
While a certain period of antigenic exposure is necessary before treatment to facilitate the establishment of immunity, cattle treated with imidocarb dipropionate ultimately have a solid sterile immunity. Long-term persistence of low-level parasitemia is now considered a disadvantage. Remaining parasites may give rise to recrudescence under adverse conditions, treated cattle may act as a source of infection, and parasites surviving at low levels of babesicide may acquire resistance.
Imidocarb, provides protection from clinical disease for 3 to 6 weeks but allows a sufficient level of infection for immunity to develop. This strategy is highly effective if the host is assured to be exposed to babesiosis during the period of protection, either through a tick bite in areas where babesiosis is endemic or by inoculation of live parasites. Acquired immunity then takes over from the passive drug protection, and the animal passes smoothly to a resistant state without an intermediate clinical stage. However, if infection rates are sporadic of if very high doses of imidocarb are used, as a complete inhibition of parasite development will hinder the mounting of an adequate immune response. The major problem associated with this approach is concern about drug residues in milk and beef, which has led to the withdrawal of imidocarb in several European countries.2
Imidocarb (Imizol)
This, and the allied drug amidocarb, are effective babesicides for cattle at the dose rate of 1 mg/kg BW. At 2 mg/kg BW it completely eliminates the parasites from the host and maintains some residual activity; non-infected cattle derive a month’s resistance to clinical infection but can be infected subclinically. It can therefore be used to protect cattle when vaccination is undesirable, e.g. pregnancy, or when exposure to infection is short lived, and as a temporary protection while awaiting vaccination. The drug can be given SC, but the hydrochloride is inclined to be irritant; the propionate is less so.
Horses and donkeys
The preferred treatment of equine babesiosis is not well-established. Many drugs have been used, but a combination of imidocarb and buparvaquone appears to be the only efficient treatment capable of eliminating B. equi infection. B. caballi is more susceptible, but it is expected to develop resistance quickly. Imidocarb must be given in a strict treatment regimen (four IM injections of 10% solution at a dose of 4 mg/kg BW at intervals of 72 hours). For B. caballi, a regimen of 2 mg/kg BW on two occasions 24 hours apart is sufficient to control an acute infection, but does not completely eliminate the babesia, and the patient may become a carrier. The hydrochloride salt of imidocarb is strongly acidic and may provoke severe local reactions in horses, therefore, the dipropionate is being used more. A note of warning is necessary about the treatment of donkeys, which are very susceptible to imidocarb (the LD50 is less than 2 mg/kg BW). Buparvaquone 4–6 mg/kg BW parenterally is also capable of controlling acute babesiosis due to B. equi, but also on its own, like imidocarb, it does not eliminate the infection, allowing the horses to remain carriers. Some initial research has been done on the use of triclosan on equine and bovine Babesia infections.48
The therapeutic efficacy of imidocarb, artesunate, arteether, buparvaquone and arteether + buparvaquone combination was evaluated against B. equi of Indian origin in splenectomized donkeys with experimentally induced infection.49 Imidocarb had deleterious effects on liver function, and the arteether + buparvaquone combination was found to be safe and may be a superior drug for treating B. equi infection.
CONTROL (BOVINE BABESIOSIS)
Prevention and biosecurity
Prevention of introduction of the disease into a non-enzootic area depends on effective quarantine to prevent the introduction of the vector tick, and laboratory testing to ensure freedom of the importee from infection with the pathogen. The international movement of animals has become a very important matter to the horse industry where teams of pleasure horses attend competitions in other countries, and where valuable stallions move to another country for a brief period to stand at stud. There is a tendency for some countries to be very restrictive in their quarantine procedures for horses, and international relations would be enhanced if more was known about the relationship between a positive serological test and infectivity for other horses.
Eradication
Eradication of bovine babesiosis from an area depends upon eradicating the vector tick – a problem in applied entomology. It was achieved in the United States but is unlikely to be attempted again because of the high cost to local wildlife, which are potential hosts to the ticks. Other problems encountered in the eradication process include:
• Difficulty of getting a complete muster of all cattle on every dipping day
• Multihost ticks, which can be infective but temporarily not resident on a beast on dipping day
• Spread of ticks or infested cattle due to environmental activity, e.g. floods, windstorms
A major problem is encountered when the protozoan persists through succeeding generations of the vector tick. The resistance of ticks to acaricides is also a factor relating to the infestation level of cattle.50
The effect of three tick (Boophilus microplus) control strategies (none, threshold, and strategic) on endemic stability and the likelihood of babesiosis (Babesia bovis) has been examined using a spreadsheet age-class computer simulation model based on weekly tick counts from Brazil and Uruguay.26 The Brazil bovine population was in a naturally occurring state of enzootic stability with an inoculation rate exceeding 0.005 throughout the year. Threshold dipping strategies did not increase the risk of babesiosis. Strategic dipping resulted in an extended period of enzootic instability lasting 30 weeks which required protection of the herd by vaccination. Because of the more prolonged low winter temperature conditions in Uruguay, the bovine population was in a naturally occurring state of endemic instability, characterized by a 28-week period in which the inoculation rate was below 0.005. Strategic dipping would lead to eradication of the babesial parasite from tick and bovine populations, but would not result in eradication of the tick vector. This could lead to subsequent outbreaks if Babesia carrier animals were introduced into the herd. In both populations, strategic tick control could be accompanied by concurrent babesiosis vaccination.
Limitation of prevalence
To accomplish limitation of prevalence at economically sustainable levels requires different solutions in different circumstances as set out below. It is largely dependent on tick control by the frequent application of acaricides, chemotherapy to kill the babesia in the cattle host and, to a lesser degree, by immunization of the host cattle. These measures are only partly effective, and are time-consuming and expensive. The reason for the poor performance of vaccination procedures after a great deal of research is that the mechanisms of immunity to protozoa, especially Babesia spp. is the lack of knowledge on how immunity to these parasites works.
• Susceptible cattle moving into an enzootic area need prior vaccination
• Marginal areas next to enzootic areas where tick populations vary with climatic change so that resident cattle lose their immunity after some dry years, and are then exposed to infection when wet years foster the migration of vector ticks back into the area. Recommended techniques are vaccination before outbreaks commence, if forecasting is available, plus temporary chemoprophylaxis after outbreaks have commenced
• Enzootic areas where losses are occurring due to environmental stress or, especially, concurrent infection with a second pathogen, e.g. Anaplasma marginale, or where the tick population has been decimated by overzealous dipping; chemoprophylaxis and relaxation of the dipping program are recommended. An adequate tick population is one ensuring that all cattle are infected and reinfected early and sufficiently often to maintain them in a state of constant of infection and therefore of immunity.
Vaccination
Vaccination has been done with varying degrees of success with live and dead whole parasites, crude parasite extracts, and isolated parasite antigens.2 Several findings support the development of vaccines against babesiosis. First, cattle which recover from a primary Babesia infection or that have been immunized with attenuated parasites are resistant to challenge infection. Second, immunization of cattle with native Babesia antigen extracts or culture-derived supernatants containing secreted Babesia antigens elicit protective immunity against both homologous and heterologous challenge.
The features of cattle farms on which the exposure of young cattle to tick fever organisms is sufficient to ensure that immunity is high and the risk of clinical disease is low (see endemic stability, under Epidemiology) can be compared with those farms on which exposure is insufficient (endemic instability) can be compared to examine the relationships between the management of ticks and tick fever.51 In Queensland, Australia, the majority of cattle herds do not have sufficient exposure to B. bovis, B. bigemina or A. marginale to confer endemic stability for tick fever.51 For B. bovis, the major cause of outbreaks of clinical disease in Queensland, fewer than half the herds had evidence of endemic stability. The decision to leave a few ticks on cattle in an effort to induce endemic stability did increase the likelihood of endemic stability to A. marginale. However, it was ineffective, because only 26% of herds had endemic stability against all three organisms. Thus given the low proportion of herds with endemic stability to tick fever organisms and the high likelihood of clinical disease, vaccination is recommended to protect dairy cattle from tick fever throughout the tick-infested area of Queensland.
Vaccination with living immunogens
Vaccines incorporating live, attenuated strains of B. bovis and B. bigemina have been used routinely or experimentally in Australia and a number other countries.52 The literature on designing blood-stage vaccines against Babesia bovis and Babesia bigemina has been reviewed.36 The data available on the efficacy, degree and duration of immunity provided by live vaccines against B. bovis and B. bigemina infections in Australia have been reviewed.53 Most of the available live vaccines are produced in government-supported production facilities, in Australia, Argentina, South Africa, Israel, and Uruguay. These vaccines include bovine erythrocytes infected with selected strains. The risk of contamination of blood-derived vaccine is real and makes post-production quality control essential, and unfortunately beyond the means of many countries in endemic regions. Techniques developed in Australia over many decades have formed the basis for production of live Babesia vaccines in most countries where they are used.22
Origin and purification of strains.
Since 1990, three strains of B. bovis, and one of B. bigemina (G strain) have been used to produce vaccines in Australia. After testing for virulence, immunogenecity and purity, suitable strains are preserved as master stabilates in liquid nitrogen.22
Attenuation of parasites
The most reliable method of reducing the virulence of B. bovis is the rapid passage of strains through susceptible splenectomized calves. Attenuation usually occurs after 8 to 20 calf passages.
Rapid passage in splenectomized caves is not reliable but the virulence of B. bigemina decreases during prolonged residence in latently infected animals. A single B. bigemina isolate (G strains) has been used in the Australian and South African vaccines since 1972 and the early 1980s, respectively.22
Vaccine specifications
Live vaccines have proven very effective and reasonably safe, particularly when vaccination is restricted to cattle less than 1 year of age, when they still have natural resistance to the disease. Parasites for vaccines are derived from splenectomized donor calves infected with attenuated strains or parasites grown in vitro.52 The vaccines are provided either chilled or cyropreserved. Despite the disadvantages, live vaccines provided greater than 95% protection for the life of the animals.
Frozen vaccine is superior to chilled vaccine because of long shelf-life which allows post-production testing of potency and safety before dispatch. Glycerol is used as cryoprotectant in Australia in preference to dimethyl sulphoxide because it allows post-thaw storage life of the vaccine for at least 8 hours. Frozen vaccine is the only product available in South Africa and Israel, and demand for it is growing in Australia. Frozen vaccines are transported in suitably insulated containers with liquid N2 or solid CO2 as refrigerant which limits the ability to supply vaccines to all destinations. To ensure infectivity, the prepared vaccine must be used within 8 hours of thawing, and once thawed should not be refrozen. If glycerol is used, a thawed vaccine can remain viable for only 8 hours at temperatures ranging from 4 to 30°C. A frozen bivalent B. bovis and B. bigemina vaccine and frozen monovalent B. bovis and B. bigemina vaccines using dimethyl sulphoxide as the cryoprotectant are produced in South Africa and Israel, respectively. If dimethyl-sulphoxide is used, a vaccine should be used within 30 minutes of thawing.
Most of the babesiosis vaccines produced to date have been provided in a chilled form. In Australia, 35 million doses were supplied between 1996 and 2003. It is popular because of ease of production, ease of transportation even with limited resources, ease of use, and low cost. The chilled vaccines currently used in Australia contain 1 × 107 B. bovis, 2.5 × 106 B. bigemina and 1 × 107 Anaplasma centrale organisms per 2 mL dose. Chilled vaccine has a very short shelf-life, which is currently 4 days in Australia, which requires rapid, reliable means of communication and transportation to ensure viability. Chilled vaccines can remain viable for up to a week if stored at 4°C.
To reduce the risk of neonatal hemolytic disease in calves (alloimmune hemolytic anemia) of vaccinated dams, the vaccine should not be used repeatedly; most owners vaccinate only young animals seldom more than twice. Reduction of the dose rate from 5 to 2 mL and use of a cell free diluent has eliminated the problem in Australia.22
The procedures to ensure quality assurance of the vaccines has been described.22
The development of effective living vaccines against bovine babesiosis in Australia required laboratory and field research over the period from 1959 to 1996 is a remarkable success story of veterinary medicine.54 The most significant change occurred in 1964 with the traditionally used carriers of Babesia being replaced as vaccine donors by acutely infected splenectomized calves. This ensured the infectivity of the vaccine and was fortuitously associated with a reduction in the virulence of the Babesia bovis vaccine. The vaccine reduced serious losses from babesiosis in vaccinated cattle in Australia to very low levels and gained acceptance worldwide.
The demand for live trivalent tick fever vaccine containing B. bovis, B. bigemina and Anaplasma centrale produced by the Department of Primary Industries, Queensland, has increased from less than 10000 doses in 1988 to 500000 doses in 2001.55 The challenge to obtain B. bigemina parasitized erythrocytes on a large enough scale from infected splenectomized calves to meet the demand was achieved by reducing the dose rate of infected cells without affecting immunogenicity and still leave a safety margin of at least 50-fold for infectivity. This change quadrupled the potential yield of doses per calf and allowed the Department to meet the increased demand for B. bigemina vaccine.
Use of live vaccine
Cattle born in vector-infested regions.
Any factor affecting the survival of the tick vectors will affect the risk of babesiosis occurring. An increased number of ticks will increase the threat of disease until an endemically stable situation develops. Conversely, reduced tick numbers will increase the longer-term risk of babesiosis due the reduced natural exposure of calves. Therefore, cattle owners in endemic areas in Australia are advised to supplement natural exposure by vaccinating calves at weaning age. Vaccination is also recommended if cattle are being moved within the endemic area.
Susceptible cattle imported into vector-infested country or region.
Large numbers of cattle, predominantly of Bos taurus breeds are being imported into tropical, developing countries to upgrade local livestock industries. This has resulted in significant economic losses due to tick-borne diseases, including babesiosis. Vaccination of naïve cattle moving from tick-free to endemic areas within Australia is usually very effective. This practice has played a crucial role in making the livestock industries in these countries more sustainable and competitive in meeting market demand with regard to breed type.
K strain B. bovis and G strain B. bigemina from Australia have been shown experimentally to be protective in South Africa and Sri Lanka. Vaccine containing these strains has also been used with beneficial results in countries such as Zimbabwe and Swaziland in Africa, Venezuela and Ecuador in South America, Malaysia and the Philippines in Southeast Asia, and islands of the Caribbean.
Use of a vaccine in the face of an outbreak is common practice in Australia.22 Superimposing vaccination in this way on a natural infection will not exacerbate the disease, but will pre-empt the development of virulent infections in the proportion of the herd not yet exposed to field challenge. To prevent further exposure, the group should also be treated with an acaricide capable of preventing tick attachment from the time of diagnosis to 3 weeks after vaccination. Injectable or pour-on formulations of ivermectin and moxidectin as well as fluazuron are highly effective acaricides but do not prevent transmission of Babesia.
Clinically affected cattle should be treated as soon as possible with a suitable babesiacide. In the case of a severe outbreak, it may be advisable to treat all the cattle with a prophylactic compound (imidocarb or diminazene) and to vaccinate them later when the drug residue will not affect vaccine parasite multiplication.
Hazards and precautions of live vaccine use
The likelihood of vaccine-induced reactions has been reduced with the development of attenuated strains but there is always the risk of reactions when highly susceptible, adult cattle are vaccinated. Calves 3 to 9 months of age have a high level of natural resistance and a low risk of reactions. In Argentina, vaccination is only recommended for calves while in Australia and South Africa, adult cattle can be vaccinated, provided proper precautions are taken. Concurrent infections may increase the likelihood of reactions.22 The fever associated with reactions in pregnant cows may cause abortion and in large bulls a temporary loss of fertility. In the case of valuable cows and bulls, their body temperatures should be monitored during the reaction and those with prolonged fever should be treated with a babesiacide.
Potential for spread of Babesia following vaccination.
There is no reliable evidence that current live vaccines may spread the disease from vaccinated to unvaccinated cattle.22
Since the introduction of a standardized method of production in Australia, live babesiosis vaccines have generally proved to be highly effective. In most cases, a single vaccination provided lasting, probably life-long immunity against field infections with antigenically different strains. However, some failures have occurred and are thought to be associated with loss immunogenicity by frequent passaging of the vaccine strains in splenectomized calves. This was corrected by replacing the vaccine strain. To prevent future recurrences of failure, the number of passages of the vaccine strains of B. bovis is limited by frequently reverting to a master stabilate with a low passage number. Other failures may be associated with the immune responsiveness of the host and the immunogenicity of the vaccine strain subpopulations.
A single inoculation of an attenuated vaccine containing B. bovis and B. bigemina at 6 to 9 months of age provides good, long-lasting protection both in Australia and overseas.53 At that age, the risk of vaccine reactions is minimal. The immunity following use of live B. bovis vaccine lasts for at least 4 years, and possibly less for B. bigemina.52 It is known to persist even after elimination of Babesia infections and studies on drug cured cattle suggest that the degree of acquired immunity is related to the degree of antigenic stimulation (duration of prior infection) rather than the presence of live parasites. There is no evidence of a loss of immunity with time and revaccination is unnecessary. Revaccination is advisable when there is uncertainty over the accuracy of previous procedures, to ensure all animals seroconvert or when there has been a change in the strains used in the vaccine.
An attenuated frozen vaccine containing in vitro culture-derived stains of B. bovis and B. bigemina, provided protection to 90% of vaccinated cattle against the virulent Babesia spp. field strains.56
The persistence of Babesia bovis and B. bigemina infection in Friesian cows, following vaccination with attenuated live vaccines has been shown by subinoculation of blood into splenectomized calves.40 B. bigemina persisted in some cows vaccinated 10 and 46 months, previously, and B. bovis persisted in 50% of cows vaccinated 10 and 47 months previously. Parasites of both species persisted among the serologically negative cows, whereas blood obtained from serologically positive cows failed to transmit infection. Thus in the absence of reinfection, Friesian cattle may spontaneously eliminate B. bigemina and B. bovis infection after various periods of time.
The inherent disadvantages of vaccines derived from blood of animals include the risk of reactions or contamination with pathogenic organisms, sensitization against blood groups, tick transmissibility of vaccine strains and need for a cold chain transportation.
Vaccinated cattle should be housed or kept under close observation for a month in case excessive reactions occur. A major problem in vaccination with living protozoa is the occasional apparent failure to transmit the protozoa. This may be due to the absence of the protozoa from the bloodstream of the donor at the time that the blood is drawn, or to the presence of a prophylactic drug – for example, imidocarb dipropionate – or low levels of antibody in the animal’s tissues. Revaccination is necessary in these circumstances, preferably with blood from a donor that is undergoing a severe reaction at the time.
The attenuated organisms used in unfrozen South Africa B. bovis and B. bigemina vaccines are susceptible for longer periods to the residual effect of the anti-babesial drugs diminazene and imidocarb dipropionate than the virulent field strains. The waiting periods before administration of the frozen B. bovis and B. bigemina vaccines in animals which have been treated with diminazene at 3.5 mg/kg BW, compare favorably with unfrozen vaccines at 4 and 8 weeks. The inhibitory effect of imidocarb dipropionate at 3.0 mg/kg BW on the infectivity of both frozen B. bovis and B. bigemina vaccines is longer, and requires minimum waiting periods before administration of these vaccines of 12 weeks and 24 weeks, respectively.57
Vaccination with subunit vaccines
Subunit vaccines offer an attractive alternative to virulent or attenuated parasites.58 The vaccines are based on recombinant antigens derived from cloned DNA of protozoan parasites.58 Several protective antigens associated with merozoites or merozoite-infected erythrocytes of B. bigemina and B. bovis have been identified as possible approaches.58 Rhoptry-associated proteins may become the first targets of generic recombinant vaccines.2
Non-living vaccines
Non-living vaccines would overcome many of the inherent difficulties in production, transport and use of live vaccines.22 However, they have not been sufficiently efficacious and more research is required.
Vector control
Vector control was first used successfully to control and eventually eradicate the cattle tick Boophilus annulatus and Babesia from the United State.59,60 In 1906, an eradication program began which involved livestock owners, state officials, and the US Department of Agriculture specialists.60 The program involved three tactics. First, some pastures were rendered tick-free by excluding all host animals until the ticks had starved to death. The second and more common tactic was to retain the livestock on the infested pastures and to disinfect the animals at regular 2-week intervals by immersion in an arsenic solution which killed the engorged female ticks. Third, the interstate movement of tick-infested cattle was prohibited through quarantine. The campaign to eradicate cattle ticks from the United States is the most sustained, extensive, coordinated area-wide attack ever made against an arthropod pest. The tick was removed from over a million square km during a period of 34 years. The tick is confined to the lower Rio Grande River in Texas, where reinfestation occurs via animal movement from Mexico. This necessitates continual control of fringe populations of cattle.
In Africa, babesiosis is only part of very important complexes of ticks and tick-borne disease, and intensive government-regulated tick control programs have been used for many years. In other continents, the situation is much less complex and where babesiosis is endemic, disease control rather than eradication is more realistic. Eradication of the tick vectors is a permanent solution to the problem but is rarely considered practical, environmentally sustainable, or economically justifiable on either a national or a local basis.
Natural endemic stability
Natural endemic stability can seldom be relied upon on as a disease control strategy. First, in endemic areas, climatic effects, genetic make-up of hosts and management strategies, inevitably have a major effect on the rate of transmission and ultimately on the likelihood of endemic stability developing. Second, endemic stability is an economic concept that incorporates risk management and loss thresholds. The climatic, animal, and management parameters which allow endemic stability can change on a seasonal and annual basis. Third, the model for endemic stability was developed in Australia and the Americas where disease/vector interactions are relatively simple. The African situation is much more complex and less predictable with four main diseases, several vectors, presence of game reservoirs, and larger range of susceptibility of bovine breeds.22
Control of babesiosis in species other than cattle
For the most part, control of babesiosis in other species is carried out similarly to the procedures used in control of the disease in cattle. Most attention is focused on eradicating the vector tick, selecting infected and carrier animals by an appropriate laboratory test, and sterilizing the positive reactors by appropriate treatments. Control of ticks in pleasure horses by periodic spraying and inspection is a practical proposition when the animals are in constant use. No vaccines have been produced for use in horses. An attenuated vaccine produced by rapid passage through splenectomized lambs produces solid immunity in sheep.
Bock R, et al. Babesiosis in cattle. Parasitol. 2004;129:S247-S269.
Bock RE, DeVos AJ. Immunity following the use of Australian tick fever vaccine: a review of the evidence. Aust Vet J. 2001;79:832-839.
De Vos AJ, Bock RE. Vaccination against bovine babesiosis. Ann NY Acad Sci. 2000;916:540-545.
Brown WC, Palmer GH. Designing blood-stage vaccines against Babesia bovis and B. bigemina. Parasitol Today. 1999;15:275-280.
Bose R, et al. Current state and future trends in the diag nosis of babesiosis. Vet Parasitol. 1995;57:61-74.
Bruning A. Equine piroplasmosis. Update on diagnosis treatment and prevention. Br Vet J. 1996;1522:139-151.
Zintl A, et al. Babesia divergens, a bovine blood parasite of veterinary and zoonotic importance. Clin Microbiol. 2003;16:622-636.
Wagner GG, Holman P, Waghela S. Babesiosis and heartwater: threats without boundaries. Vet Clin North Am Food Anim Pract. 2002;18:417-430.
Yeruham I, Hadani A, Galker F. Some epizootiological and clinical aspects of ovine babesiosis caused by Babesia ovis — a review. Vet Parasitol. 1998;74:153-163.
Allred DR. Antigenic variation in babesiosis: is there more than one ‘why’? Microbes Infect. 2001;3:481-491.
Allred DR. Babesiosis: persistence in the face of diversity. Trends Parasitol. 2000;19:51-55.
Jenkins MC. Advances and prospects for subunit vaccines against protozoa of veterinary importance. Vet Parasitol. 2001;101:291-310.
Callow LL, Dalgleish RJ, DeVos AJ. Development of effective living vaccines against bovine babesiosis — the longest field trial? Int J Parasitol. 1997;27:747-767.
Schuster FL. Cultivation of Babesia and Babesia-like blood parasites: agents of an emerging zoonotic disease. Clin Microbiol Rev. 2002;15:365-373.
Pegram RG, Wilson DD, Hansen JW. Past and present national tick control program. Ann NY Acad Sci. 2000;916:546-554.
Myers JH, Savoie A. van Randen E. Eradication and pest management. Annu Rev Entomol. 1998;43:471-491.
1 Angus B. Int J Parasitol. 1996;26:1341.
2 Zintl A, et al. Clin Microbiol. 2003;16:622.
3 Cringoli G, et al. Vet Res. 2002;33:421.
4 Almeria S, et al. Vet Rec. 2001;149:716.
5 L’Hostis M, Seegers H. Vet Res. 2002;33:599.
6 Devos J, Geysen D. Vet Parasitol. 2004;125:237.
7 Regassa A, et al. Vet Parasitol. 2003;116:267.
8 Yeruham I, et al. Vet Parasitol. 1998;74:153.
9 Razmi GR, et al. Small Rum Res. 2003;50:39.
10 Razmi GR, et al. Vet Parasitol. 2002;108:109.
11 Chauvin A, et al. Vet Res. 2002;33:429.
12 Xuan X, et al. Vet Parasitol. 2002;108:179.
13 Xu Y, et al. J Vet Med Sci. 2003;65:1015.
14 Martin R. Aust Vet J. 1999;77:308.
15 Heuchert CMS, et al. Vet Parasitol. 1999;85:1.
16 Zweygrarth E, et al. Parasitol Res. 2002;88:460.
17 Gallatin LL, et al. J Am Vet Med Assoc. 2003;223:1027.
18 Langton C, et al. Parasitol Res. 2003;89:194.
19 Holman PJ, et al. Parasitol Res. 2002;88:405.
20 Carrique JJ, et al. Vet J. 2000;160:162.
21 Battsetseg B, et al. Vet Parasitol. 2002;107:351.
22 Bock R, et al. Parasitol. 2004;129:S247.
23 Goff WL, et al. Parasite Immunol. 2001;23:463.
24 Goff WL, et al. Ann NY Acad Sci. 2002;969:164.
25 Allred DR. Trends Parasitol. 2000;19:51.
26 Smith RD, et al. Ann NY Acad Sci. 2000;916:510.
27 Bock RE, et al. Aust Vet J. 1999;77:465.
28 Allred DR. Microbes Infect. 2001;3:481-491.
29 Mosqueda J, et al. Ann NY Acad Sci. 2004;1026:222.
30 Criado-Fornelio A, et al. Vet Parasitol. 2004;119:337.
31 Kumar S, et al. Vet Parasitol. 2002;107:295.
32 Holman PJ, et al. New Eng J Med. 2002;347:697.
33 Thompson C, et al. Clin Infect Dis. 2001;33:676.
34 Shuster FL. Clin Microbiol Rev. 2002;15:365.
35 Yeruham I, et al. J Vet Med B. 2003;50:60.
36 Brown WC, Palmer GH. Parasitol Today. 1999;15:275.
37 Court RA, et al. Int J Parasitol. 2001;19:29.
38 Garcia TD, et al. Ann NY Acad Sci. 2004;1026:298.
39 Sherlock M, et al. Vet Rec. 1999;152:687.
40 Pipano E, et al. Vet J. 2002;164:64.
41 Malandrin L, et al. Vet Res. 2004;35:131.
42 Bashiruddin JB, et al. Vet Parasitol. 1999;84:75.
43 Rampersad J, et al. Vet Parasitol. 2003;114:81.
44 Goncalves PM, et al. Vet Parasitol. 1999;82:11.
45 Goff WL, et al. Clin Diag Lab Immunol. 2003;10:38.
46 Boonchit S, et al. J Clin Microbiol. 2002;40:3771.
47 Xuan X, et al. Clin Diag Lab Immunol. 2001;8:645.
48 Bork S, et al. Am J Trop Med Hyg. 2003;68:334.
49 Kumar S, et al. J Vet Med Sci. 2003;65:1171.
50 Bianchi MW, et al. Vet Parasitol. 2003;112:75.
51 Serugga JN, et al. Aust Vet J. 2003;81:147.
52 De Vos AJ, Bock RE. Ann NY Acad Sci. 2000;916:540.
53 Bock RE, DeVos AJ. Aust Vet J. 2001;79:832.
54 Callow LL, et al. Int J Parasitol. 1997;27:747.
55 Standfast NF, et al. Vet Parasitol. 2003;115:213.
56 Alvarez JA, et al. Ann NY Acad Sci. 2004;1026:277.
57 Combrink MP, et al. Ann NY Acad Sci. 2002;969:169.
58 Jenkins MC. Vet Parasitol. 2001;101:291.
COCCIDIOSIS
Etiology Many different Eimeria spp., Isospora spp.
Epidemiology Young calves, lambs, piglets, kids and, rarely, foals. Infection rate is high, clinical disease much less common; high morbidity with low case fatality rate. Occurs most commonly in crowded conditions both in barns and on pasture, especially in calves and lambs moved from pasture to feedlot. Transmitted by fecal– oral route; oocysts shed from infected animals. Immunity develops after infection; clinical disease occurs rarely in adult cattle.
Signs Diarrhea, dysentery, tenesmus, appetite normal or inappetence, mild abdominal pain in lambs, nervous signs in calves with coccidiosis in cold climates, loss of body weight, anemia in some cases but not common. Epidemics occur in calves and lambs, especially feedlot animals. Diarrhea without blood in feces of piglets. Diarrhea with large amount of blood in foals.
Clinical pathology Diagnostic number of oocysts in feces.
Lesions Ileitis, cecitis, colitis.
Diagnostic confirmation Oocysts in feces; merozoites in intestinal tissues.
Calves: Rotavirus and coronavirus diarrhea; Clostridium perfringens Type C enterotoxemia; colibacillosis due to attaching and effacing Escherichia coli.
Lamb: Salmonellosis; helminthiasis; Clostridium perfringens Type C enterotoxemia.
Piglets: Transmissible gastroenteritis; colibacillosis; Strongyloides ransomi; Clostridium perfringens Type C enterotoxemia.
Foals: Salmonellosis; Foal-heat diarrhea; Rotavirus diarrhea; Clostridium perfringens type B enterotoxemia.
Treatment Supportive therapy. Coccidiostats.
Control Control population density to minimize number of oocysts ingested while immunity develops. Use of coccidiostats in feed and water supplies.
ETIOLOGY
The pathogenic coccidial species are as follows:
• Cattle: Eimeria zuernii, E. bovis (smithii) and E. ellipsoidalis. E. alabamensis,1 E. auburnensis and E. wyomingensis may also cause disease in calves
• Sheep: E. arloingi A (ovina), E. weybridgenis (E. arloingi B), E. crandallis,2 E. ahsata and E. ovinoidalis (previously known as E. ninakohlyakimovae)2 and E. gilruthi3
• Goats: E. arloingi, E. faurei and E. gilruthi, E. caprovina, E. ninakohlyakimovae1 and E. christenseni
• Pigs: Isospora suis, E. debliecki, E. scabra and E. perminuta
• Horses and donkeys: E. leuckarti.4
The eimeriid coccidia are one of the more controversial groups of protozoa, and their taxonomy and classification have been debated for more than 50 years.5 A Controversial Roundtable was held in 2001 to initiate discussion on how a new and more comprehensive classification of eimeriid coccidia can be devised.
EPIDEMIOLOGY
Occurrence and prevalence of infection
Coccidiosis occurs universally, most commonly in animals housed or confined in small areas contaminated with oocysts. The coccidia are host-specific and there is no cross-immunity between species of coccidia. Clinical disease is most common in cattle and sheep. Coccidiosis causing diarrhea in newborn piglets is a major problem in some swine herds. Coccidiosis is less common in horses, but sporadic clinical cases and deaths do occur, especially in weaning age foals.
Coccidiosis occurs most commonly in young animals, with a seasonal incidence that may be associated with the time of year young calves and lambs are brought together for weaning or moved into feedlots or fed in small areas for the winter months. The prevalence of infection and the incidence of clinical disease are also age related. In housed dairy cattle, the prevalence of infection is 46% in calves, 43% in yearlings, and 16% in adult cows.6
Calves
In North America, the disease occurs most commonly in beef calves after weaning in the fall of the year and when confined and fed in small overcrowded areas during the winter months. Infection occurs commonly when weaned calves are fed on the ground resulting in continuous fecal contamination of the feed. The prevalence of infection in calves in the northwestern and midwestern part of the United States is highest in summer, fall and spring compared to midwinter (January) and early summer (June). In Canada, winter coccidiosis occurs in beef calves 6–10 months of age most commonly following a prolonged cold period or a sudden change from a moderate winter to severely cold temperature.7 Cold weather may act as a stressor to precipitate clinical disease in animals previously infected. Acute coccidiosis and a marked increase in the numbers of oocysts discharged will occur following the treatment of infected calves with a corticosteroid on the 20th day after infection when clinical signs are apparent, or from the 12th–15th day after infection.8
Occasional outbreaks occur in nursing beef calves on pasture when they mingle near water supplies. Post-weaning coccidiosis occurs in beef calves grazing pasture in the subcostal dry tropics of northern Queensland, Australia. It may be more severe in dry years suggesting that the oocyst challenge is less important than the immunosuppressive effect of weaning and dietary stress in precipitating clinical disease. Calves are usually weaned and yarded for 3 weeks and then turned out to graze. Severe Eimeria zuernii coccidiosis causing diarrhea, dysentery, weight loss, and death occurs with up to 10% of calves being clinically affected. The disease is most severe in hot, dry and sunny conditions when, despite heavy fecal contamination, the yard conditions remain dry and dusty, and oocysts are difficult to find.
In South Africa, the prevalence is highest in calves and the most prevalent species are Eimeria zuernii, Eimeria ellipsoidalis, and Eimeria bovis.9 The oocyst levels in the feces of adult cows were low.
In Sweden, coccidiosis due to E. alabamensis along with other species is a common cause of diarrhea and unthriftiness in calves 2–4 months of age within the first few weeks after being placed on permanent pasture in the spring.11 The onset of the diarrhea coincides with the prepatent period of 6–8 days of E. alabamensis. Overwintered sporulated oocysts of several different species are the source of infection.10 The incidence of diarrhea is much higher in first-season grazing calves placed on pastures that had been used for cattle in the previous 5 years compared to those calves placed on different pastures not previously used by cattle.9 The likely primary source of infection is the calves or previous occupants of pens, with the mature cows being unimportant.12 Clinical coccidiosis can be reproduced by inoculation of calves with a pathogenic dose of E. alabamensis,13 and is most severe in calves receiving the highest number of oocysts.
In dairy calves, the disease occurs in overcrowded and dirty, wet conditions, and when fecal contamination of the feed is common. Surveys of dairy farms in Pennsylvania reveal that coccidiosis was found the third most common health-related problem observed by dairy farmers.14 Over 16% of dairy farmers were treating animals, or feeding a coccidiostat, for the control of coccidiosis.
Adult cattle
Coccidiosis is uncommon in adult cattle, but occasional cases and even epidemics do occur, especially in dairy cows that have calved 6–8 weeks earlier.3 Surveys of farms in Maryland found that 2.3% of dairy cows, 21.7% of dairy heifers, and 32.9% of beef cattle were all infected with E. bovis and all were subclinical.15 These older animals serve as source of infection for younger calves in the herd.
Lambs
Coccidiosis in lambs is a common disease in housed flocks.16 In Germany, the cumulative incidences of E. ovinoidalis and E. weybridgensis/crandallis increased rapidly resulting in almost 100% incidence in 8 week-old lambs.
Acute coccidiosis in intensively grazed lambs in Britain occurs at about 6–8 weeks of age when the oocyst output is very high in healthy, as well as in clinically affected, lambs. There is no periparturient rise in oocyst output by the ewes. The oocyst output by grazing lambs is very large compared to the output by ewes.
Coccidiosis occurs commonly in lambs following introduction into a feedlot situation where overcrowding and other stressors are operative. Lambs moved directly from range have had almost no previous exposure to coccidia, making them highly susceptible to infection and outbreaks of clinical disease. Coccidiosis is also a major problem in rearing housed lambs in the United Kingdom.17
Goats
Coccidiosis is one of the most important diseases of goats kept in large numbers under intensive management conditions.18 The prevalence of infection may be as high as 100% in some goat farms, and higher infection rates occur in young goats in large herds.19 Kids are the major source of pasture contamination, and newly weaned kids have high oocyst counts.20 Over 13 different species of coccidia have been identified in goats from different parts of the world. Surveys in Zimbabwe reveal a population mortality of 20–40% among kids on different farms; coccidiosis is thought to be a major cause of these deaths.18 Coccidial and helminth infections may occur concurrently in goats of varying ages surveyed on farms but nematode infections do not occur in kids.21 Goat kids concurrently infected with third-stage Haemonchus contortus may gain weight more slowly.22 Goats do not shed as many oocysts and helminth eggs as do sheep,23 which may be related to their mode of feeding (browsing versus grazing).
In the Czech Republic, where the majority of goats are dairy goats, the overall prevalence of Eimeria oocysts in feces was 92%.24 Nine Eimeria spp. were identified and E. arloingi was the most common with a prevalence of 84%. The number of oocysts was lower in adults and the highest in kids and clinical coccidiosis occurred in kids 2 to 4 weeks after weaning. The disease occurs in goats in Poland where 81% of adults and 100% kids are infected, and clinical coccidiosis occurs in 50% of the kids.25 In West African Dwarf goats in Ghana, Eimeria oocysts appeared at about 20 days after birth and oocyst output in some kids reached 2.7 million oocysts per gram of feces by about 39 days after birth, followed by declines to 23 840 per gram by 5.5 months of age.26
Pigs
Observations suggesting the presence of neonatal porcine coccidiosis include: repeated epidemics of diarrhea in piglets 5–15 days of age; no response to therapy with antimicrobials; and failure of vaccination of the pregnant sow with E. coli bacterins to control neonatal piglet diarrhea. The peak incidence occurs between 7 and 10 days of age, and most commonly during the warm summer months when high temperatures favor the sporulation of the oocysts. Isospora suis is a common parasite on pig farms; it may be found in 90% of herds and from 25 to 50% of litters.27,28 The prevalence may be higher when piglets and their sows are raised on solid cement floors compared to self-cleaning floors.27 The morbidity rates are variable and the case fatality rates may be up to 20%. Rotavirus infection may occur concurrently with Isospora suis infection in piglets 1–3 weeks of age and may be important causes of steatorrhea, or unspecified diarrhea known as milk scour, white scour, or 3-week diarrhea. Coccidiosis associated with E. scabra has also occurred in a 40 kg finishing pig affected with severe diarrhea and weight loss, and an outbreak of severe enteritis with dysentery in grower pigs from 7 to 16 weeks of age, due to a mixture of Eimeria spp. including E. porci has occurred. Coccidiosis has occurred in 20-week-old gilts moved from a high health status environment to a dirt lot, which suggests that high-health-status pigs are highly susceptible to coccidiosis.29
I. suis infection occurs widely in large pig-producing farming systems in Germany where the highest rate of infection occurs in litters at 3 to 4 weeks of age.30 Surveys of intestinal parasites in pigs from intensive farms in China have found I. suis infections along with other parasites.31
Surveys of pig farms in Germany, Switzerland, and Austria found 76% of the farms were positive for I. suis and oocysts were more commonly found on farms which reported diarrhea and uneven weight gain. Disinfection, floor types or treatment with toltrazuril did not affect the frequency of observations.32
Foals
Eimeria leuckarti is a ubiquitous protozoal parasite of horses that has been reported in North America and elsewhere. In a survey of horse farms in Kentucky, 41% of foals examined were oocyst-positive, and 85% of farms had oocyst-positive foals.4 The incidence of clinical disease is much less common in foals than other species, but clinical cases and deaths do occur, particularly in recently weaned foals.
Morbidity and case fatality
In general, for most species of farm animals, the infection rate is high, and rate of clinical disease is usually low (5–10%), although epidemics affecting up to 80% may occur. The case fatality rate is usually low, with the exception of the high case fatality rate in calves with winter coccidiosis accompanied by nervous signs.7 The case fatality rate may be high in calves or lambs with no previous exposure to coccidia after suddenly being introduced to a high level of infection. In calves, body weight gains and feed consumption are commonly reduced for many weeks after acute clinical coccidiosis, and affected calves do not regain losses in body weight compared to controls.
In lambs on pasture, subclinical infections are common but there is no documented evidence that growth rate is affected even with high levels of infection. While medication with a coccidiostat may lower the infection rate, there is no difference in performance between the medicated and non-medicated groups. In lambs raised under crowded conditions indoors, the acquisition of a natural multiple species infection had no effect on growth rate, but an artificial infection with E. ninakohlyakimovae resulted in severe clinical disease and a case fatality rate of 50%.
Piglets infected with I. suis have significantly reduced body weight at 7, 14, and 21 days of age. The reduction in 21-day-old weight is economically important because this weight is a component of the sow productivity index which is used as a management aid to help producers quantitatively determine the potential value of gilts as replacement animals.
Methods of transmission
The source of infection is the feces of clinically affected or carrier animals, and infection is acquired by ingestion of contaminated feed and water, or by licking the hair coat contaminated with infected feces. Oocysts passed in the feces require suitable environmental conditions to sporulate. Moist, temperate, or cool conditions favor sporulation, whereas high temperatures and dryness impede it. In general, oocysts sporulate at a range of 12–32°C (53.5–89.5°F) and require oxygen. They resist freezing down to about –7 to –8°C (19.5–17.5°F) for 2 months, but –30°C (–22°F) is usually lethal.33 It has been suggested that oocysts might sporulate in the winter months on the hair coats of animals contaminated with feces. This may explain the continual production of several different species of coccidia during the cold winter months when sporulation on the ground is not possible.33 Dry conditions and high temperatures also destroy sporulated oocysts within a few weeks but the oocysts may survive for up to 2 years under favorable conditions. Temperatures above 35°C, humidity below 25%, and sunlight for 4 hours are fatal for E. zuernii. Under simulated yard conditions in Queensland, Australia, where it is hot, dry, and sunny, when the yard floor materials reach up to 48°C and when air temperatures did not exceed 27°C, oocyst numbers fall by 50% after 24 hours, by 98% after 48 hours, and are undetectable after 72 hours.33
Ingestion of the sporulated oocysts results in infection, but very large numbers must be taken in before clinical disease results. Large numbers of oocysts arise by continual reinfection and building up the degree of environmental contamination. This occurs commonly when calves or lambs are crowded into small pens or confined in feedlots. Lambs can become infected within a few weeks after birth from lambing grounds heavily contaminated by the ewes. Overcrowding of pastured animals on irrigated pasture, or around surface water holes in drought conditions, may also cause heavy infestations. Feeder lambs and calves brought into feedlots from sparse grazing may carry a few oocysts, which build up into heavy infestations in the lots, especially if conditions are moist. In such situations, clinical signs of the disease usually appear about a month after the animals are confined. Young calves and lambs on pasture may shed large numbers of oocysts for long periods, which may be a factor in the development of large coccidial populations.
In cow-calf herds in Germany, the prevalence and intensity of oocyst excretion varied with time resulting in peak values around the time of parturition (periparturient rise), particularly in the case of Eimeria bovis.34 Oocysts of E. zuernii, E. auburnensis were first observed in calves 3 weeks of age and the prevalence of infection increased up to 67% by 9 weeks of age. The serum antibody levels in the cows were inversely correlated with E. bovis oocyst excretion which may reflect the degree of immunity developed against the infection in the cows. Antibodies to E. bovis were present in the sera calves after the ingestion of colostrum; the levels decreased from the first week of life until the third week. Subsequently, antibody levels increased until 9 weeks of age in direct correlation with oocyst excretion.34
Lambs may become infected soon after birth (before 4 weeks of age) from three possible sources of infective oocysts:
• Oocysts surviving in old fecal contamination of the lambing area from previous occupation
The fecal oocyst burden is high at 4 weeks of age but gradually declines, so that by 5 months of age the oocyst count is similar to that of their parent ewes.
Sows do not play a significant role in the transmission of I. suis infections transmitted from one generation of piglets to the next through contamination of the farrowing pen. Infection levels as low as 100 sporulated oocysts can result in oocysts per gram values of 100 000 and induce clinical signs of coccidiosis within 2 weeks of infection.35 Oocysts of I. suis cannot usually be found in the feces of sows on swine farms where neonatal coccidiosis occurs. In one survey, no oocysts of I. suis were found in sows on farms with a history of neonatal coccidiosis due to I. suis, but 82% of the sows were infected with Eimeria spp. On farms without a history of neonatal coccidiosis, the prevalence of I. suis in the sows was 0.6%. In a survey in Papua New Guinea, 83% of sows raised on concrete were infected with I. suis. In another study, of two swine herds where neonatal coccidiosis due to I. suis occurred, the sows either began to excrete oocysts, or oocyst excretion rose to a detectable level from 5 days before to 3 days after farrowing. The oocysts of I. suis can sporulate and become infected within 12–16 hours at temperature ranges of 32–35°C (89.5–95°F), which are common in modern farrowing units.
Risk factors
Animal risk factors
Acute coccidiosis occurs primarily in young animals, but may occur at any age when resistance is affected by intercurrent disease or inclement weather. The prevalence of infection is usually higher in calves than yearlings or adults in the same herd, but there is also evidence of variation in Eimeria species-specific age resistance.6 A concurrent experimental infection of calves with the coronavirus and E. bovis may result in clinical disease and lesions that are more severe than those caused by either infection alone.36
Nutritional status of the animal as a risk factor for clinical coccidiosis is well known. Early weaning of lambs at 21 days of age, followed by experimental infection, results in a failure of growth. Field observations have shown that early weaned lambs are more susceptible to coccidiosis than those weaned at a later date. This may be a reflection of lack of immunity in the younger lambs, but dietary stress in early weaned lambs may contribute to the disease. In sheep with naturally occurring coccidiosis, animals on moderate and high planes of nutrition may have more dual infections than those on a low plane of nutrition, whereas the latter group have triple and quadruple infections. Considerable numbers of oocysts can be excreted into the environment even by well-fed sheep of 14–16 months of age. Lambs kept on a low plane of nutrition were less affected by clinical coccidiosis than those kept on a high plane of nutrition. The planes of nutrition were also associated with differences in the prevalences of Eimeria spp.
In the United Kingdom, coccidiosis occurs commonly in housed lambs weaned at 6–8 weeks of age and reared on straw with a high stocking density, which provides an ideal environment for oocyst survival and sporulation.17 The use of coccidiostats does not affect the oocyst excretion rate, which suggests either inconsistencies in the effect of in-feed medication or the infection may be controllable in non-medicated flocks without the use of coccidiostats.
Epidemics of post-weaning coccidiosis occur in beef calves under hot and dry tropical conditions where the ingestion of oocysts is small because the conditions are unsuitable for oocyst development. Such calves have usually commenced shedding oocysts within a month of birth and can shed up to nine different species by 3–4 months. Calves developing clinical coccidiosis following weaning may be more physiologically susceptible than others.
Environmental and management risk factors
Coccidiosis occurs in all species of farm animals when environmental and managemental conditions result in oral exposure of large numbers of sporulated oocysts to non-immune animals. Overcrowding and feeding animals on the ground, or in situations in which the feed and water supplies can become heavily contaminated with feces and oocysts increases the infection pressure and promotes the fecal–oral route cycle of infection. The disease occurs commonly in small beef cattle herds that raise their own replacements and finish their own feedlot cattle in small outside pens that are crowded, and the feed becomes heavily contaminated by feces. Grazing calves for the first time on permanent pastures is associated with clinical coccidiosis due to the ingestion of overwintered oocysts.1
The occurrence of clinical coccidiosis in housed dairy cattle in The Netherlands is rare.6 This may be associated with management practices in which calves are individually housed during the first few weeks and subsequently housed in small groups in relatively large pens. General hygienic standards are high, and manure is frequently removed. These measures reduce the intake of high numbers of oocysts and are favorable because the intensity of coccidial infections is directly related to the number of oocysts in the environment and ingested by the animals. A low level of oocyst infection will induce immunity.
The production system can influence the development of subclinical and clinical coccidiosis. In Germany, two production systems of fattening lambs are common. In the extensive system, lambs are not weaned until slaughter with little or no concentrate feeding. In the intensive system, the lambs receive a high level of concentrates.37 In lambs naturally infected with Eimeria spp., the mean fattening period was significantly shorter in the intensively managed groups compared with an extensively managed group. Even if no clinical signs of coccidiosis were observed in any of the systems, based on reduced daily weight gains, the lambs were subclinically affected by coccidiosis in all systems. The highest oocyst output occurred in the extensive group which may be related to intake of straw and hay from the ground. Straw and high stocking density predispose a heavy contamination of an environment which is ideal for oocyst survival and rapid sporulation. The risk of coccidiosis increased markedly if animals have a higher intake of contaminated straw from the ground.
Pathogen risk factors
Multiple infections are most common in natural infections. A single species of coccidia may be the major pathogen, but others contribute to the disease. In some cases, clinical coccidiosis in cattle occurs only when E. bovis and E. zuernii occur together. At least 13 species of coccidia oocysts have been found in the feces of cattle in the United States, with E. bovis being the most prevalent. In sheep and goats, the prevalence of multiple species can be as high as 95% and 85%, respectively. In a single sheep flock, there may be as many as 10 species of Eimeria.38 E. ovinoidalis, E. ovina, E. ahsata, and E. parva are highly pathogenic, whereas E. faurei, E. intricata, E. crandallis, and E. weybridgensis are only moderately pathogenic. Similar results have been obtained in surveys done in feeder cattle brought from different geographical locations in the United States to a feedlot. While E. bovis and E. zuernii are the species most commonly associated with bovine coccidiosis, as many as 11 different species may be present in the cows and calves of one beef herd. I. suis is a major cause of neonatal or weanling diarrhea in pigs, while E. debliecki is not considered a pathogen. This provides evidence that coccidia have a widespread distribution wherever animals are kept.
Immune mechanisms
Immunity against intestinal coccidia consists of both cellular and humoral components. Cellular immunity is more important in resistance against reinfection than humoral immunity. Field observations suggest that coccidiosis in cattle is immunosuppressive, which may increase their susceptibility to other common infections. In experimental coccidiosis, neutrophil function may be inhibited and the feeding of decoquinate may prevent this inhibition.
The administration of dexamethasone to calves suppresses the immunological response of the animal and allows the life cycle of the coccidia to proceed uninterrupted.8 Estradiol and progesterone can enhance cell-mediated immunity and provide some protection against the often severe wasting and debilitation in calves associated with E. bovis infection.39
In Norway, coccidiosis is an important disease in young lambs on pasture. Most lambs spend the first few weeks of their life indoors and have little exposure to infective oocysts, and little or no immunity is acquired. When the lambs are turned out to pastures grazed by sheep in the previous grazing season, they rapidly become infected with overwintered oocysts, mainly as a result of their habit of eating soil. Coccidiosis develops in these non-immune lambs 2–3 weeks later. The immunity induced by the first infection seems to protect most lambs from reinfections later in the grazing season. In lambs treated with sulfadimidine at 200 mg/kg BW on days 12, 13, and 14 after turnout, there is a marked reduction in the severity of the coccidial infections.
Specific immunity to each coccidial species develops after infection, so that young animals exposed for the first time are often more susceptible to a severe infection and clinical disease than other animals. In lambs, natural infection acquired at pasture and artificial infections acquired by experimental inoculation result in immunity to challenge. A single initial infection of as few as 50 oocysts will provoke a solid immunity to reinfection with the same species, and oocyst production ceases after about 10 days. Under field conditions, lambs are probably continually ingesting oocysts from pastures that become increasingly contaminated as the season progresses. Thus, immunity to a range of species of coccidia is boosted by frequent reinfection.40
Very young lambs are relatively resistant to infection with a mixture of pathogenic species of coccidia, but susceptibility increases progressively up to at least 4 weeks of age.2 Lambs inoculated at 4–6 weeks of age, develop severe diarrhea, whereas the same inoculum given at 1 day of age causes no clinical disease. Early subclinical infection improves the resistance of lambs to later challenge. When lambs receive a relatively heavy inoculation of oocysts during their first week of life they are relatively resistant to the pathogenic effects of some coccidia, are able to respond immunologically, and are protected from subsequent challenges.2 This suggests that a challenge with coccidia, before the lamb becomes susceptible to their pathogenic effects, may help to reduce the incidence and severity of clinical coccidiosis later. Resistance to E. zuernii infection in calves occurs after chemotherapy, or experimental infection, with monensin or amprolium. Both drugs suppress the development of the experimental disease, during which time immunity develops. An effective immunity develops in piglets following natural or experimental infection with I. suis, which is the most immunogenic species of swine coccidium. Susceptible piglets are exposed to this species of infection in older swine. Piglets develop a more severe clinical disease when infected with I. suis at 1–3 days of age than when infected at 2 weeks of age.
PATHOGENESIS
The coccidia of domestic animals pass through all stages of their life cycle in the alimentary mucosa and do not invade other organs, although schizonts have been found in the mesenteric lymph nodes of sheep and goats. The different species of coccidia localize in different parts of the intestine. E. zuernii and E. bovis occur primarily in the cecum, colon and the distal ileum, whereas E. ellipsoidalis and E. arloingi affect the small intestine. E. gilruthi localizes in the abomasum and occasionally the duodenum.41
Life cycle
The coccidial life cycle is self-limiting. The unsporulated oocysts are passed in the feces and develop into the infective stage in the environment. The original single cell divides, forming four sporoblasts, each of which develops into one sporocyst, and within each sporocyst two sporozoites develop. When ingested, the wall of the oocyst breaks down and the sporocysts are released. The sporocysts then enter epithelial cells. Once within the cells the sporozoites undergo asexual multiple fission (schizogony) and become first-generation shizonts, which form numerous merozoites. After the schizont matures, the merozoites are released by rupture of the epithelial cell. New epithelial cells are again invaded and second-generation schizogony occurs in the large intestine. This is followed by the release of another generation of merozoites, which invade epithelial cells and produce the sexual stages, the macrogametocyte and the microgametocyte. The second-generation schizogony and fertilization of the macrogametocyte by the microgametocyte (gametogony) are the stages of the life cycle that cause functional and structural lesions of the large intestine. As the second-generation schizonts or gamonts mature, the cells containing them slough from the basement membrane and cause hemorrhage and destruction of the cecum and colon. The oocysts are the result of fertilization of the gametocytes and are discharged at the time of rupture of the cells, which usually coincides with the onset of clinical signs of dysentery. The prepatent period varies with the species of coccidia; with E. bovis it is 5–20 days, and with E. zuernii 15–17 days. Oocyst production in calves infected with E. zuernii reaches peak numbers on the 19th and 20th day after experimental infection. The prepatent period of E. alabamensis varies from 6 to 8 days.
E zuernii and E. bovis are most pathogenic to cattle and their life cycles are similar. In calves infected with E. zuernii, first-generation schizogony occurs in the lower ileum, and second generation schizogony and gametogony occurs in the cecum and proximal colon. The gametocytes are the pathogenic stages and cause rupture of the cells they invade, with consequent exfoliation of the epithelial lining of the intestine. It is notable that the oocyst count is often low when the disease is at its peak, as the oocysts have not yet formed. Exfoliation of the mucosa causes diarrhea, and in severe cases, hemorrhage into the intestinal lumen, and the resulting hemorrhagic anemia may be fatal. If the animal survives this stage, the life cycle of the coccidia terminates without further damage and the intestinal mucosa will regenerate and return to normal. The patent period for E. bovis and E. zuernii varies from 5 to 12 days depending on the infecting dose of oocysts.
Treatment of calves with a corticosteroid can convert subclinical infection in calves into a peracute clinical form of the disease, which suggests that environmental, nutritional, and management factors may also act as stressors in producing clinical disease. The pathophysiological changes associated with experimental infection of calves with E. zuernii include a decrease in packed cell volume and reduction in plasma sodium and chloride levels. However, these are not remarkable; in naturally occurring cases the changes are not significant.
In lambs, most natural infections are composed of several different species of coccidia and there is a wide range of values in the production of oocysts from individual lambs, either in the feces from the same lamb over a period of time or in the feces from a number of lambs on any one occasion. Under practical conditions, constant reinfection occurs and waves of pathogenic stages succeed each other. The occurrence of villous atrophy in the intestinal mucosa of lambs affected by coccidiosis is probably related to the recurrence of diarrhea and loss of body weight.
However, in lambs at least, there is some doubt about the effects of coccidial infection on growth rate, feed consumption, and clinical signs in the experimental disease. There may be no obvious relationship between infective dose, the fecal oocyst production, and clinical disease. This suggests that, in lambs, the mere presence of large numbers of fecal oocysts does not constitute a diagnosis of coccidiosis and that other pathogenetic factors may be involved in conversion to clinical disease. It is also possible that the large number of oocysts may represent non-pathogenic coccidia.
Severely affected calves surviving the acute phase of the disease do not regain losses in body weight unless they are fed for an additional 3–4 weeks, suggesting that bovine coccidia can have a marked effect on performance. A subclinical coccidial infection superimposed on an established, low-grade, subclinical nematode infection in the small intestine may have a marked effect on the mineralization of the skeletal matrix in young adult ruminants, predisposing these animals to osteodystrophy.
The fact that multiple infections are so common may explain the variations in oocyst discharge from infected animals, but more importantly in groups of animals. New cases may develop every few days for a few weeks because of the variations in the length of the prepatent period between species of coccidia.
The pathogenesis of the nervous signs of coccidiosis in calves is unknown. Detailed examination of a series of cases excluded possible explanations such as:
• Alterations in serum electrolytes
• Vitamin A and thiamin deficiencies
• Uremia, Haemophilus somnus meningoencephalitis
• Severity of clinical disease
• Gross alteration in intestinal bacterial flora and hepatopathy.7
A labile neurotoxin in the serum of calves with ‘nervous coccidiosis’ has been identified using laboratory mice as test subjects, but its significance is unknown.7
The pathogenesis of bovine winter coccidiosis, which occurs during or following very cold weather in Canada and the northern United States, is not understood. In January, February, and March, the outside temperatures may reach –40°C (–40°F) with daily mean temperatures of –10 to –15°C (14–5°F) for several days consecutively. Such temperatures should be too cold for sporulation of oocysts in feces on the ground. There is speculation that sporulation could occur on the moist hair coats of cattle, or the endogenous stages of E. zuernii may be in a latent phase and reactivated by the stress of cold weather.
Isospora suis has at least three asexual, and one sexual, intra-intestinal multiplication cycles. All stages are most prominent in the distal half of the small intestine, but also occur in the proximal small intestine, cecum and spiral colon. The prepatent period extends from 8 to 16 days and the shedding of oocysts occurs in a cyclic pattern, with 2–3 peaks separated by intervals of about 5 days.35 This biphasic disease course results in diarrhea, villous atrophy and necrosis of intestinal epithelium at 4–6 and 8–10 days after infection. The feces may also be negative for oocysts between the biphasic peaks. Under temperature ranges of 32–35°C (89.5–95°F) the oocysts of I. suis can sporulate and become infective within 12–16 hours. An extra-intestinal stage of the life cycle related to the second patent period is postulated. The lesions are most pronounced in the small intestine, and consist of villous atrophy and focal ulceration from the destruction of villous epithelial cells, principally during the peak of asexual reproduction. A fibrinonecrotic pseudomembrane may develop in severe cases. A combined experimental infection of I. suis and rotavirus in gnotobiotic and conventional pigs results in a synergistic action based on a competition of rotavirus and the coccidia for mature, enzymatically active absorptive villous cells.42 The effects of neonatal I. suis infection on protein dynamics may persist after clinical recovery and could contribute to suboptimal weight gains.
The effects of experimental Eimeria bovis infection on the metabolism of water, sodium, and potassium in calves has been examined.43 Although acute sublethal bovine coccidiosis alters electrolyte and water metabolism, the overall balance of electrolytes and water is largely maintained by physiologic adaptation. This supports field observations that most cases of clinical coccidiosis do not require supportive fluid and electrolyte therapy.
Piglets experimentally inoculated with I. suis at 3 days of age develop a strong level of resistance to reinfection. Piglets develop more severe clinical signs of coccidiosis when inoculated with I. suis at 3 days of age than at 19 days of age. This suggests that maturation of non-specific components of the immune system is more important in the resistance of neonatal piglets to I. suis infection than specific immune mechanisms.44
CLINICAL FINDINGS
The incubation period after experimental dosing varies between species of coccidia and animals infected. It ranges from 16 to 30 days in cattle infected with E. zuernii and E. bovis, from 14 to 18 days in sheep, and as short as 5 days in piglets. The clinical syndromes associated with the various coccidia are similar in all animal species.
Cattle and sheep
A mild fever may occur in the early stages, but in most clinical cases the temperature is normal or subnormal. The first sign of clinical coccidiosis is the sudden onset of diarrhea with foul-smelling, fluid feces containing mucus and blood. Blood may appear as a dark, tarry staining of the feces or as streaks or clots, or the evacuation may consist entirely of large clots of fresh, red blood. The perineum and tail are commonly smudged with blood-stained feces. Severe straining is characteristic, often accompanied by the passage of feces, and rectal prolapse may occur. The degree of hemorrhagic anemia is variable depending on the amount of blood lost, and in most naturally acquired cases in calves anemia is not a feature. In experimental cases of coccidiosis in lambs, due to E. ninakohlyakimovae, although 50% of the animals died, there was no evidence of blood loss, but there was significant hemoconcentration. However, in exceptional cases, anemia is severe with pale mucosa, weakness, staggering, and dyspnea. Dehydration is common, but is not usually severe if affected animals continue to drink water.
Inappetence is common and in exceptional cases there may be anorexia. The course of the disease is usually 5–6 days, but some animals undergo a long convalescent period during which feed consumption and body weight gains are subnormal. Severely affected calves do not quickly regain the body weight losses which occurred during the clinical phase of the disease. In mild cases there is diarrhea and reduced growth rate, but not necessarily dysentery too. Subclinical cases show inferior growth rate and chronic anemia only.
Clinical coccidiosis occurs only rarely in adult cattle, and the few reports describe several animals affected over a short period of time.45 Young dairy cows may be affected commonly within 6–8 weeks after calving. Diarrhea, dysentery, tenesmus, pale mucous membrane, thickening and corrugation of the rectal wall, and rapid recovery often without treatment are common.
Coccidiosis with nervous signs
Nervous signs consisting of muscular tremors, hyperesthesia, clonic–tonic convulsions with ventroflexion of the head and neck and nystagmus, and high mortality rate (80–90%) occurs in calves with acute clinical coccidiosis.7 Outbreaks of this ‘nervous form’ have occurred in which 30–50% of all susceptible calves are affected. It has occurred most commonly during, or following, severely cold weather in midwinter in Canada and the northern United States. Affected calves may die within 24 hours after the onset of dysentery and the nervous signs, or they may live for several days, commonly in a laterally recumbent position with a mild degree of opisthotonos. In spite of intensive supportive therapy, the mortality is high. Nervous signs have not been reported in experimental clinical coccidiosis in calves, which suggests that the nervous signs may be unrelated to the dysentery or, indeed, even to coccidiosis.
Lambs
Coccidiosis in lambs is generally similar to that in calves, but with much less dysentery. In groups of lambs raised and fed under intensified conditions, inferior growth rate, diarrhea with or without blood, low-grade abdominal pain, gradual onset of weakness, inappetence, fleece damage, mild fever, recumbency, emaciation, and death with a course of 1–3 weeks were noted. The diarrhea may escape cursory examination of the animals, but clinical examination of affected lambs reveals a perineum smudged with feces, and soft feces in the rectum. Lambs moved directly from range pasture to a feedlot with little or no previous exposure to coccidia often develop acute disease with a high morbidity and case fatality rate.
Horses
In the horse, while there is some doubt about the pathogenicity of E. leuckarti, diarrhea of several days duration, and acute massive intestinal hemorrhage leading to rapid death have been described in foals and young horses.4
Piglets
In piglets, severe outbreaks of coccidiosis occur between 5 and 15 days of age.42 Anorexia and depression are common. There is profuse diarrhea and the feces are yellow, watery, and sometimes appear foamy. The diarrhea may persist for several days when dehydration and unthriftiness are obvious. Although affected piglets continue to suck, they become dehydrated and lose weight. Vomition may occur. Entire litters may be affected, and the case fatality rate may reach 20%. The disease may persist in a herd for several weeks or months, particularly where a continuous farrowing program is used.
CLINICAL PATHOLOGY
Oocyst count
A count of over 5000 oocysts/g of feces of ruminants is considered significant. Although counts below 5000/g of feces do not usually suggest clinical disease, they may indicate a potential source of severe infection if environmental conditions for spread become favorable. Oocyst counts of over 100 000/g are common in severe outbreaks, although similar counts may also be encountered in normal sheep. The output of oocysts following an acute infection falls sharply after the peak, which may result in a critically affected animal with dysentery and low oocyst count. If oocysts are not found and the disease is suspected, merozoites can be looked for in direct smears; they do not float on the conventional concentrated sugar or salt solutions used for flotation of oocysts. The several species of coccidia can be differentiated, up to a point, by the characteristics of the oocysts.
Calves
Affected animals exposed to a large number of oocysts may develop severe dysentery a few days before oocysts appear in the feces. However, this is not commonly observed when the feces from several affected animals are examined, and usually within 2–4 days after the onset of dysentery the oocysts will appear in the feces. The period during which oocysts are discharged in significant numbers (patent period) will vary between species of coccidia, the age of the animal, and the degree of immunity, which often makes it necessary to examine a number of animals in a group or herd rather than rely on the results from a single animal.
Lambs
In lambs at pasture, oocysts first appear in the feces at about 2 weeks of age. The oocyst count continues to rise in the lambs until about 8–12 weeks when the counts will be 105–106/g of feces. Thereafter, the count declines to about 500/g when the lambs are 6–12 months of age. There is also considerable variation, both between lambs and in day-to-day samples from individuals, in the numbers and species of oocysts present in the feces. Hence the need for examination of several samples over a period of several days to assess the burden.
Piglets
In piglets, the prepatent period varies from 4 to 7 days (most commonly 5 days) and oocysts are shed in the feces for 5–8 days after the onset of clinical signs. Piglets may develop the disease at 5 days of age, and oocysts may not be present in the feces until 3 days later. The use of a saturated sodium chloride with glucose as a flotation solution is recommended when examining piglet feces for I. suis.46
Necropsy examination of selected untreated clinical cases is often necessary to make the diagnosis. The disease should be suspected when piglets 5–8 days of age develop a diarrhea that responds poorly to treatment. Outbreaks of diarrhea in piglets under 5 days of age are usually associated with Escherichia coli or transmissible gastroenteritis. However, mixed infections are common and extensive laboratory investigations are often necessary to isolate the causative agents. The diagnosis often requires a combination of consideration of the history of diarrhea in piglets 5–15 days of age, gross and microscopic lesions, the presence of coccidial stages in mucosal smears and histological sections, and identification of oocysts in intestinal contents and feces. In heavy infections, piglets may die before the sexual stages of the parasite develop and the diagnosis is dependent upon finding lesions, schizonts, and merozoites of I. suis in the middle jejunum and ileum. The developmental stages can be detected by the mucosal smear technique. A rapid field diagnostic procedure consists of staining glass slide impression smears of the mucosa of the ileum and jejunum.
This technique is superior to bright field microscopy in detecting I. suis oocysts after flotation and is significantly more sensitive when direct smears are examined.47 It does not require purification of samples and requires no agents to prepare them. The calculated detection limit of autofluorescence is 10 oocysts per gram (using 100 mg of feces per smear); the flotation technique provides a lower detection rate of 334 oocysts per gram of feces.
NECROPSY FINDINGS
Carcasses often have generalized tissue pallor and there is usually fecal staining of the hindquarters. In cattle, congestion, hemorrhage, and thickening of the mucosa of the cecum, colon, rectum, and ileum are the characteristic gross changes at necropsy. The thickening may be severe enough to produce ridges in the mucosa. Small, white cyst-like bodies, formed by large schizonts, may be visible on the tips of the villi of the terminal ileum. Ulceration or sloughing of the mucosa may occur in severe cases. The lesions associated with experimental E. bovis infection in the small and large intestines are characterized by a fibrinous typhlitis and colitis. Clotted blood or bloodstained feces may be present in the lumen of the large intestine. Histologically, there is denudation of the epithelium, and merozoites may be observed in some cells. Smears of the mucosa or intestinal contents should be examined for the various developmental stages.
The necropsy findings in sheep are marked by more severe involvement of the small intestine than in cattle. Atrophy of the villi in the proximal ileum occurs in both natural and experimental infection in lambs. In experimental E. crandallis infection in lambs, there is loss of surface epithelial cells, atrophy of villi in the small intestine, and severe diffuse crypt hyperplasia in the small and large intestines.41 E. ovinoidalis infection in lambs causes massive invasion of the cecum, with destruction of crypt stem cells leading to denudation of the cecal mucosa. In naturally acquired coccidiosis of lambs, three types of macroscopic intestinal lesions may be recognized:
1. Flat oocyst patches, which are whitish spots 1–2 mm in diameter
2. Raised oocyst patches on enlarged villi
3. Polyps which are proliferative lesions protruding into the lumen.
All three lesions contain heavy concentrations of gamonts and oocysts. In sheep affected with E. gilruthi, the abomasum contains numerous nodules, 1–2 mm in diameter, similar in gross appearance to ostertagiasis. These nodules contain the large schizonts of E. gilruthi-containing merozoites.3
In piglets, the small intestines are usually flaccid, but occasionally a fibrinonecrotic enteritis may be noted. Clinical signs precede the production of oocysts, so mucosal scrapings should be examined for the presence of earlier stages of the life cycle.
Samples for confirmation of diagnosis
• Parasitology – feces (FECAL); segments of jejunum, ileum, colon (DIRECT SMEAR)
• Histology – formalin-fixed duodenum, jejunum, ileum, cecum, colon (LM).
Calves In calves, clinical coccidiosis is characterized by dysentery, tenesmus, mild systemic involvement and dehydration. The presence of large numbers of oocysts supports the diagnosis, and necropsy findings are usually characteristic. When nervous signs occur in calves appearing to have coccidiosis, differentiation from other diseases causing brain dysfunction must be made.
Sheep In sheep, the diagnosis is dependent on the clinical findings of diarrhea and dysentery, the presence of large numbers of oocysts in the feces, and the intestinal lesions at necropsy. Large numbers (100 000/g) of oocysts may occur in the feces of normal lambs, and thus the observation of large numbers of oocysts in the feces of lambs affected with diarrhea and/or dysentery may not, in itself, constitute a diagnosis of coccidiosis. In lambs that have had previous contact with coccidia, and that may be relatively immune, other causes of diarrhea such as helminthiasis, salmonellosis, Clostridium perfringens type C enterotoxemia, and helminthiasis should be considered. See Table 26.3 for epidemiological and clinical features of the diseases causing diarrhea in sheep and goats.
Piglets In piglets, diarrhea due to coccidiosis must be differentiated from enteric colibacillosis, transmissible gastroenteritis, rotavirus infection, Strongyloides ransomi, and Clostridium perfringens type C. See Table 26.3 for epidemiological and clinical features of the diseases causing diarrhea in pigs.
Foals See Table 26.3 for the epidemiological and clinical features of the diseases causing diarrhea in foals. The common causes of diarrhea of foals include those associated with: Salmonella spp., Actinobacillus equuli, and rotaviruses.
TREATMENT
Coccidiosis is a self-limiting disease, and spontaneous recovery without specific treatment occurs commonly when the multiplication stage of the coccidia has passed. Many treatments have been recommended without taking this into account and it is unlikely that any of the chemotherapeutic agents in common use for clinical coccidiosis has any effect on the late stages of the coccidia. Most of the coccidiostats have a depressant effect on the early, first-stage schizonts and are used for control.
In an outbreak, the clinically affected animals should be isolated, and given supportive oral and parenteral fluid therapy as necessary. The population density of the affected pens should be reduced. All feed and water supplies should be high enough off the ground to avoid fecal contamination. Mass medication of the feed and water supplies may be indicated in an attempt to prevent new cases and to minimize the effects of an epidemic. Cattle with coccidiosis and nervous signs should be brought indoors, kept well-bedded and warm, and given fluid therapy orally and parenterally. However, the case fatality is high in spite of intensive supportive therapy. Sulfonamide therapy parenterally may be indicated to control the development of secondary bacterial enteritis or pneumonia, which may occur in calves with coccidiosis during very cold weather. Corticosteroids are contraindicated.8
The chemotherapeutic agents recommended for treatment and control of coccidiosis in calves and lambs are summarized in Table 26.4. There is insufficient information available to make reliable recommendations for the specific treatment of acute clinical coccidiosis. None of these chemotherapeutic agents has been adequately tested in clinical trials. Sulfadimidine is used widely empirically for the treatment of acute clinical coccidiosis in calves. Amprolium is also used for treatment, and there may be a beneficial effect in terms of increased body weight gains and feed consumption compared to untreated controls recovering spontaneously.
Table 26.4 Chemotherapeutics recommended for treatment and control of coccidiosis in calves and lambs
| Chemotherapeutic agent | Treatment | Prevention |
|---|---|---|
| Sulfadimidine (sulfamethazine) | Calves and lambs: 140 mg/kg BW orally daily for 3 days individually | |
| Amprolium | Calves: individual dose at 10 mg/kg BW daily for 5 days or 65 mg/kg BW one dose | |
| Monensin | Lambs: 2 mg/kg BW daily for 20 days beginning on 13th day following experimental inoculation | |
| Lasalocid | Lambs: 25–100 mg/kg feed from weaning until market. Also, in ewe’s diet from 2 weeks before and until 60 days after lambing. |
Symmetrical triazintriones are effective against the asexual and sexual stages of experimental I. suis infection in piglets and is most effective before the onset of clinical signs.50
CONTROL
The control of coccidiosis assumes greatest importance in calves and lambs, and has been difficult to achieve with reliability.
Management of environment
Successful economical control will depend on avoiding the overcrowding of animals while they develop an immunity to the coccidial species in the environment. Only small numbers (50/day) of oocysts are required for the development of solid immunity in lambs. Lambing and calving grounds should be well drained and kept as dry as possible. Lambing pens should be kept dry, cleaned out frequently, and bedding disposed of so that oocysts do not have time to sporulate and become infective.51 All measures that minimize the amount of fecal contamination of hair coats and fleece should be practiced regularly. Feed and water troughs should be high enough to avoid heavy fecal contamination. Feeding cattle on the ground should be avoided if possible, particularly when overcrowding is a problem.
Lambs at pasture
In groups of lambs at pasture, the frequent rotation of pastures for parasite control will also assist in the controls of coccidial infection. However, when lambs are exposed to infection early in life as a result of infection from the ewe and a contaminated lambing ground, a solid immunity usually develops and only when the stocking density is extremely high will a problem develop.
Feedlot cattle and lambs
Control of coccidiosis in feeder calves and lambs brought into a crowded feedlot depends on management of population density, or use of chemotherapeutics, to control the numbers of oocysts ingested by the animals while effective immunity develops. Management procedures include establishing the optimum stocking density, which can be assessed by visual inspection. When animals are overcrowded they usually become dirty, there is excessive competition for feed supplies, and growth rate may be affected.
Piglets
The control of coccidiosis in newborn piglets infected with I. suis has been unreliable. The use of coccidiostats in the feed of the sow for several days or a few weeks prior to, and following, farrowing has been recommended and used in the field, but the results are variable. Amprolium and monensin have been evaluated for the prevention of experimental coccidiosis in piglets and are ineffective. A control program designed to decrease the number of oocysts has been recommended and consists of proper cleaning, disinfection, and steam cleaning of the farrowing housing. Amprolium (25% feed grade) at the rate of 10 kg/tonne of sows’ feed beginning 1 week before farrowing and continued until the piglets are 3 weeks of age has been recommended, but the results are unsatisfactory. A single oral dose of 1.0 mL of toltrazuril given to piglets 3–6 days of age reduced the occurrence of coccidiosis from 71% to 22%, and the number of days that oocysts were excreted in the feces reduced from 4.9 to 2.5.52
A single treatment of toltrazuril at 20 mg/kg BW at an early stage of infection (2 days post-infection) controlled a large experimental infection with I. suis in suckling pigs.53
Coccidiostats
Coccidiostats are used for the control of naturally occurring coccidiosis in calves and lambs. The ideal coccidiostat suppresses the full development of the life cycle of the coccidia, allows immunity to develop, and does not interfere with production performance. Those that have been used for treatment and control are summarized in Table 26.4.54
To be effective, coccidiostats must be given beginning early in the life cycle of the coccidia. In any group of animals, there will be several different species of coccidia at different stages of the cycle: some at the drug-susceptible stage (before 13–15 days in calves) and some beyond the drug-susceptible stage (after 16–17 days), which explains why coccidiostats appear to be effective in some epidemics and ineffective in others. In an epidemic in calves, new cases may develop for up to 12–15 days after the commencement of feeding of an effective coccidiostat to in-contact calves. However, the stage of the prepatent period is unknown at any particular point in time in the affected group, and the most that can be done is to medicate the feed and water supplies with the coccidiostat of choice, treat new cases that develop, and avoid the stressors of overcrowding and nutritional disorders.
Some comments about some of the coccidiostats are made here. Routine prophylactic medication of the feed and water supplies of feeder calves and lambs with an effective coccidiostat will usually control the disease and allow the development of effective immunity.
Antimicrobials
Sulfonamides in the feed at a level of 25–35 mg/kg BW for at least 15 days are effective for the control of coccidiosis in calves and lambs. Sulfadimidine at 55 g/tonne is also effective in goats. A combination of chlortetracycline and a sulfonamide has provided protection in calves and lambs
Ionophores
Monensin is an effective coccidiostat and growth promotant in cattle, sheep, and goats. The recommended levels are 16–33 g/tonne feed for calves and 20 g/tonne of feed for lambs. Levels of 11 g/tonne feed are not as reliable as the higher dose for calves. The recommended level for goats is 16 g/tonne of feed. A concentrated ration containing monensin at 15 g/tonne can be fed to ewes from 4 weeks before lambing until weaning, and to lambs from 4 to 20 weeks of age. Monensin can markedly reduce the oocyst output of ewes and lambs when fed before and after lambing. Withdrawal of the monensin may be followed by the development of fatal coccidiosis in some animals, presumably because the drug suppressed the development of immunity. Postweaning coccidiosis in beef calves has been controlled using monensin from intraluminal continuous release devices. The toxic level of monensin for lambs is 4 mg/kg BW.
Lasalocid is related to monensin and is also an effective coccidiostat for ruminants. For maximum benefit, lasalocid should be used daily in the feed of coccidia-susceptible lambs for as long as possible. An effective method of control is to medicate the feed of the ewes beginning about 2 weeks before lambing and continue the medication until the lambs are weaned. The lambs begin to receive lasalocid in their creep ration, and later in their rations from weaning until market. For maximum control of coccidiosis and improved feedlot performance, lasalocid should be given before and during the time that coccidia-naive lambs are first exposed to the natural occurrence of oocysts. A level as low as 25 mg/kg of feed will control coccidiosis and improve performance when fed to lambs in early life. Similar improvements in feedlot performance do not occur in heavier lambs already passing oocysts and being fed lasalocid at 25 mg/kg feed.
Lasalocid fed at a rate of 40 mg/kg of starter to dairy calves beginning at 3 days of age, and up until 12 weeks of age, is effective in reducing fecal oocysts and increasing mean daily body weight gain, dry-matter intake, and improved feed efficiency.55 Mixing lasalocid in the milk replacer of calves beginning at 2–4 days of age is an effective method of controlling coccidiosis.56 It is also effective as a coccidiostat when fed free-choice in salt at a level of 0.75% of the total salt mixture. Lasalocid at levels from 0.75 to 3 mg/kg BW are effective in preventing experimental coccidiosis in calves. The level of 1 mg/kg BW is the most effective and rapid, and is recommended when outbreaks of coccidiosis are imminent in cattle. Lasalocid and decoquinate are effective in reducing coccidia infections in young calves under conditions of apparent low exposure and good management. However, neither lasolocid nor decoquinate, or both, added to the feed of 16-week-old dairy calves naturally infected with subclinical coccidiosis for 56 days had any significant effect on weight performance.57
Monensin, lasalocid and decoquinate at the manufacturer’s recommended levels are equally effective. A combination of monensin and lasalocid at 22 and 100 mg/kg of diet, respectively, is an effective prophylactic against naturally occurring coccidiosis in early weaned lambs under feedlot conditions. The ionophores are used in the feed continuously from weaning to market, and provide control of coccidiosis and improve feedlot performance. The continuous feeding of lasalocid, decoquinate or monensin will effectively control coccidiosis; cessation of medication may result in the appearance of oocysts in the feces and diarrhea.
Decoquinate in the feed at 0.5–1.0 mg/kg BW suppressed oocyst production in experimentally induced coccidiosis of calves.58 It is most effective in preventing coccidial infections when fed continuously in dry feed at 0.5 mg/kg BW.59 When fed to dairy calves from 9 weeks to 24 weeks of age, there was an improvement in growth rate, height at withers, and heart girth measurement.10 A level of 0.5 mg/kg BW is effective in goats.
Toltrazuril at 20 mg/kg BW as a single dose, 10 days after being turned out to pasture, will almost completely prevent coccidiosis, which occurred commonly without prophylactic medication. A dose of 15 mg/kg BW is effective to control experimental infections of calves with E. bovis.60 In addition, medication of naturally infected lambs with toltrazuril on day 10 after turnout will markedly reduce the excretion of oocysts for a prolonged period, and thus lessen the contamination of the pasture with oocysts. A single treatment of toltrazuril can significantly reduce the oocyst output in naturally infected lambs for a period of approximately 3 weeks after administration. Weekly oral treatment of suckling lambs with 20 mg/kg BW of toltazuril reduced the oocyst output and improved weight gain over a 10-week period.61
Vaccines
The literature on the prospects for subunit vaccines against protozoa of veterinary importance has been reviewed.62 Although subunit vaccines offer many theoretical advantages, the lack of understanding of immune mechanisms to primary and secondary infection and the capacity of many protozoa to evade the host immunity remain obstacles to developing effective vaccines.
Tenter AM, et al. The conceptual basis for a new classification of the coccidian. Int J Parasitol. 2002;32:595-616.
Yun CH, Lillehoj HS, Lillehoj EP. Intestinal immune responses to coccidiosis. Dev Comp Immunol. 2000;24:303-324.
Jenkins MC. Advances and prospects for subunit vaccines against protozoa of veterinary importance. Vet Parasitol. 2001;101:291-310.
Step DL, Streeter RN, Kirkpatrick JG. Bovine coccidiosis: A review. Bov Pract. 2002;36:126-135.
1 Svensson C, et al. Vet Parasitol. 1994;53:33.
2 Gregory MW, Catchpole J. Vet Rec. 1989;124:458.
3 Fox MT, et al. Vet Rec. 1991;129:141.
4 Lyons ET, et al. Am J Vet Res. 1988;49:96.
5 Tenter AM, et al. Int J Parasitol. 2002;32:595.
6 Cornelissen AWCA, et al. Vet Parasitol. 1995;56:7.
7 Isler CM, et al. Can J Vet Res. 1987;51:261.
8 Niilo L. Can J Comp Med. 1970;34:325.
9 Matjila PT, Penzhorn BL. Vet Parasitol. 2002;104:93.
10 Svensson C, et al. Acta Vet Scand. 1993;34:175.
11 Svensson C, et al. Acta Vet Scand. 1995;36:9.
12 Svensson C. Acta Vet Scand. 1993;34:77.
13 Hooshmand-Rad P, et al. 1994;53:23.
14 Heinrichs AJ, et al. J Dairy Sci. 1990;73:1851.
15 Fayer R, et al. Vet Parasitol. 2000;93:103.
16 Reeg KJ, et al. Vet Parasitol. 2005;127:209.
17 Berriatua E, et al. Vet Parasitol. 1994;54:337.
18 Chhabra RC, Pandy VS. Vet Parasitol. 1991;39:199.
19 De La Fuente C, Alunda JM. Vet Parasitol. 1992;41:7.
20 Penzhorn BL, et al. Vet Parasitol. 1994;55:137.
21 Borgsteede FHM, Derksen DP. Vet Parasitol. 1996;61:321.
22 Rahman WA. Vet Rec. 1994;134:235.
23 Kanyari PWN. Vet Parasitol. 1993;51:137.
24 Koudela B, Bokova A. Vet Parasitol. 1998;76:261.
25 Balicka-Ramisz A. Vet Parasitol. 1999;81:347.
26 Agyei AD, et al. Small Rum Res. 2004;51:29.
27 Sayd SMO, Kawazoe U. Vet Parasitol. 1996;67:169.
28 Eysker M, et al. Vet Q. 1994;16:203.
29 Henry SC, Tokach LM. Swine Health Prod. 1995;3:200.
30 Meyer C, et al. Vet Parasitol. 1999;82:277.
31 Weng YB, et al. Vet Parasitol. 2005;127:333.
32 Mundt H-C, et al. J Vet Med B. 2005;52:93.
33 Parker RJ, Jones GW. Vet Parasitol. 1990;35:269.
34 Faber J-N, et al. Vet Parasitol. 2002;104:1.
35 Christensen JPB, Henriksen SA. Acta Vet Scand. 1994;35:165.
36 Hoblet KH, et al. Am J Vet Res. 1992;53:1400.
37 Gauly M, et al. Small Rum Res. 2004;55:159.
38 Da Silva NRS, Miller JE. Vet Parasitol. 1991;40:147.
39 Heath HL, et al. Am J Vet Res. 1997;58:891.
40 Catchpole J, et al. Vet Rec. 1993;132:56.
41 Gregory MW, Catchpole J. Am J Parasitol. 1990;20:849.
42 Vitovec J, et al. J Vet Med B. 1991;38:215.
43 Daugschies A, et al. Parasitol Int. 1997;46:159.
44 Koudela B, et al. Vet Parasitol. 1999;82:93.
45 Gunning RF, Wessels ME. Vet Rec. 1996;139:497.
46 Henriksen SA, Christensen JPB. Vet Rec. 1992;131:443.
47 Daugschies A, et al. Parasitol. 2001;87:409.
48 Joachim A, et al. J Vet Med B. 2004;51:140.
49 Ruttkowski B, et al. Vet Parasitol. 2001;95:17.
50 Bach U, et al. Parasitol Res. 2003;91:27.
51 Taylor M. In Practice. 1995;174:172.
52 Driesen SJ, et al. Aust Vet J. 1995;72:139.
53 Mundt HC, et al. Parasitol Res 90 (Suppl 3):s160.
54 Step DL, et al. Bov Pract. 2002;36:126.
55 Sinks GD, et al. J Am Vet Med Assoc. 1992;200:1947.
56 McMeniman NP, Elliott R. Aust Vet J. 1995;72:7.
57 Waggoner JK, et al. J Dairy Sci. 1994;77:349.
58 Heinrichs AJ, Bush GJ. J Dairy Sci. 1991;74:3223.
59 Fitzgerald PR, Mansfield ME. Am J Vet Res. 1989;50:961.
60 Mundt H-C, et al. Parasitol Res. 2003;90:S166.