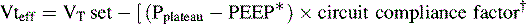Respiratory monitoring and support
Respiratory Monitoring Devices
Respiratory monitoring, particularly in combination with clinical assessment skills, is invaluable for detecting and documenting cardiopulmonary insufficiency and for guiding intervention. Ideally, such monitoring assists in the detection of cardiopulmonary insufficiency before the development of cardiopulmonary failure or arrest.
As biomedical technology continues to advance, so does the ability to monitor subtle physiologic changes in respiratory function. For these instruments to be used effectively, the clinician must be familiar with their basic principles of operation and must be able to couple use of these instruments with careful clinical observation.
Impedance Pneumography
Impedance pneumography detects chest wall movement by recording changes in resistance (impedance) across an electrical field that result from variations in thoracic volume. Chest wall impedance is measured by placing an electrode on each side of the patient's chest. Many bedside cardiac monitors include filters enabling simultaneous ECG and respiratory monitoring.45,162
When the child's respiratory rate is monitored, a high and low rate alarm must be set. In addition to high and low respiratory rate alarms, most monitors have an apnea alarm, with thresholds that can be set at 10-, 15-, or 20-second intervals. If there is a high incidence of false-positive alarms, the bedside nurse should evaluate and adjust electrode placement until placement provides maximum sensitivity to both ECG and respiratory patterns; this should reduce the number of false apnea alarms.
The most significant limitation of impedance pneumography is that all chest movement is sensed, whether or not the movement is producing effective ventilation.162 If airway obstruction develops, struggling respiratory movements will continue to be detected by the monitor, even if ventilation (air movement) is ineffective. If a child's respiratory function is poor, the nurse should not rely on this monitor to determine respiratory rate, and it should never be used to reflect effectiveness of ventilation.
Spirometry
Measurement of lung volume is accomplished by the use of spirometers. Spirometry has become more popular in the pediatric population as advancements in microprocessor technology have allowed for more accurate measurement of small lung volumes and enabled a greater variety of available bedside tests.23
Spirometry may be performed in the intubated patient if a cuffed tube is used or if there is minimal air leak around an uncuffed tube. If the patient is not intubated, a mouthpiece may be used if the child can maintain a tight mouth seal around the mouthpiece. However, patient inability to cooperate remains a significant limitation to spirometry in children less than 8 years of age. In this younger age group, patients may be unable or unwilling to make a tight seal around the mouthpiece or provide maximal effort during spirometry testing. These behaviors may result in misleading volume measurements.
A simple manometer or pressure gauge that can read both positive and negative pressures of − 150 to + 100 or + 150 mm Hg is required for these measurements. School-age children should be able to generate at least − 30 mm Hg pressure during inspiration and at least + 30 mm Hg during expiration.
In the pediatric critical care setting, spirometry may be used to measure spontaneous exhaled tidal volumes of intubated children or the vital capacity of children with restrictive lung disease. Vital capacity is defined as the maximum amount of gas that can be expired after full inspiration (deep breath) and is easily measured with a respirometer. Forced vital capacity may be measured to assess pulmonary reserve.
Other useful bedside spirometry tests include minute ventilation (tidal volume with each breath multiplied by the respiratory rate), peak expiratory flow rate, and maximum inspiratory pressure, also known as negative inspiratory force (NIF).
To measure the peak expiratory flow rate, the child is instructed to give his or her best maximum expiratory effort, exerted after a deep inhalation. A peak expiratory flow rate (PEF) is the maximum flow rate measured during forceful exhalation following a maximum inhalation. The PEF decreases when there is resistance in central airways, and it is a helpful indicator of airway constriction in patients with asthma.
The negative inspiratory force is the maximum negative pressure generated by the respiratory muscles at the patient's peak inspiratory force. It can be measured in children who are school age or older and capable of following directions. To measure the negative inspiratory pressure, the child is instructed to give his or her best maximum inspiratory effort after an exhalation to a near residual volume. Inspiratory force must be at least − 20 to −25 cm H2O (15 to 18 mm Hg) to generate a sufficient cough and clear secretions.
These volumes are presented in more detail in Chapter 9, Essential Anatomy and Physiology—Lung Volumes.
Noninvasive Transcutaneous Blood Gas Monitoring
Transcutaneous measurement of oxygen (PtcO2) and carbon dioxide (PtcCO2) tension provide the clinician with a tool for immediate and continuous assessment of tissue respiration—delivery of oxygen and removal of carbon dioxide. Transcutaneous monitoring is a noninvasive means of assessing the tissue oxygen and carbon dioxide tension, yielding more information about oxygen transport and carbon dioxide elimination than the patient's arterial oxygen tension (PaO2), pulse oximetry or cardiac index alone.
It is important to note that transcutaneous monitoring provides information regarding the gas tension of the tissue, and not the arterial blood gas tension. When the patient is hemodynamically stable, the arterial blood gas tensions and transcutaneous (tissue) measurements correlate well. However, frequently the PtcO2 is lower than the PaO2 and the PtcCO2 is slightly higher than the PaCO2.170,171 This is caused by metabolism at the tissue level consuming oxygen and producing CO2.
Instrumentation
Transcutaneous Oxygen Monitoring
The PtcO2 is measured by a heated electrode (Clark electrode) that is placed on the skin surface. The heat increases the capillary blood flow to the area, thus “arterializing” blood flow under the electrode. The sensor then measures oxygen tension at the skin surface itself; this oxygen tension should reflect the underlying tissue PO2.170,171,183 The heat ranges of the electrodes vary (most commonly between 40° C and 45° C), but the typical temperature generated is approximately 44° C.
Transcutaneous Carbon Dioxide Monitoring
Transcutaneous carbon dioxide (PtcCO2) measurements can be obtained using a pH electrode (Stow-Severinghaus electrode), infrared electrode, a mass spectrometer, or gas chromatography. To discuss each of these types of electrodes in detail is beyond the scope of this chapter. For more information regarding these electrodes, the reader is referred to Martin Tobin's Principles and Practice of Intensive Care Monitoring.171
Measurement of skin-surface or transcutaneous carbon dioxide tension (monitoring) may be a useful adjunct to the nursing care of children with acute or chronic respiratory disease. Several studies have verified high correlations between the PtcCO2 and the PaCO2 in children, with a predictable gradient between the two.21,60,76,190 Increased gradients may be caused by three of the following conditions: (1) tissue CO2 production is increased by the heat from the electrode; (2) heating the capillary blood beneath the sensor elevates the CO2 (anaerobic temperature coefficient); and (3) a countercurrent CO2 exchange mechanism in the dermal loop maintains a higher PCO2 at the tip of the loop (where the sensor lies).170,171
Consistently good correlations between PaCO2 and PtcCO2 make transcutaneous carbon dioxide monitoring a valuable tool in the pediatric critical care setting. Correlation studies have demonstrated that although the PtcCO2 will be 9 to 23 mm Hg higher than the PaCO2,98 the relationship between the two remains relatively constant. With application to an individual patient, the nurse should note the difference between PaCO2 and PtcCO2 to enable detection of trends in the patient's PaCO2. With this difference established, the number of necessary arterial blood samples is reduced, and continuous monitoring of trends in CO2 elimination are possible during procedures and changes in therapy.
The CO2 electrode is reliable even in the presence of hypotension and decreased cardiac output (i.e., shock regardless of etiology).21,22,60,76,98 Limitations of PtcCO2 monitoring include delayed measurement, inaccurate measurement, thermal injury, need for repeated calibration and site change, cost, and altered skin perfusion.
Clinical Applications
A high correlation between PaO2 and PtcO2 has been verified by many studies,34,48,101,172,175,185 particularly when the range of PaO2 is 30 to 100 torr. In fact, brief periods of hypoxemia that are reflected by a fall in PtcO2 may not be detected by intermittent PaO2 sampling. These episodes are frequently associated with nursing interventions, such as turning of the patient, vital sign measurement, dressing changes, suctioning, and chest physiotherapy.
Thick skin reduces the accuracy of the PtcO2 because fewer deep capillaries are present beneath the sensor site. In addition, thicker skin offers more resistance to oxygen diffusion than thinner skin, and it has higher oxygen consumption; thus the PtcO2 over thick skin will be lower than the PaO2. For this reason, the transcutaneous electrodes should not be applied over areas of thickened skin, such as calluses or the soles of the feet.
The PtcO2 electrode also should be placed on the trunk rather than over extremities because extremity perfusion will be influenced more readily by temperature and cardiac output. The patient should never be positioned on top of a sensor because this may decrease local blood flow.
The relationship among PtcO2, PaO2, and cardiac output have been documented in a somewhat predictive pattern: the PtcO2 correlates linearly with the PaO2 when the cardiac output is greater than 65% of normal.172,173,174,175 Tremper175 reports a high correlation between the PaO2 and PtcO2 levels when the cardiac index is greater than 1.54 L/minute per m2 BSA. If cardiac output is compromised significantly (< 65% of normal), the PtcO2 will be less than 80% of the PaO2. This poor correlation reflects a compromise in tissue perfusion and often is observed during episodes of low cardiac output or shock even before the PaO2 falls.174,175
Nursing Considerations
Erythematous marks may develop at the electrode site, resulting from heat produced by the electrodes. Although these marks may disturb the family and staff, actual blisters (second-degree burns) seldom develop if the electrodes are changed as recommended by the manufacturer.
The schedule for rotation of electrode sites on the skin surface should be strictly maintained and documented. The erythematous marks caused by an electrode may last for hours or days following electrode removal, but rarely leave scars. Many studies recommend a maximum of a 3- to 4-hour interval for each electrode location,183 but the nurse should check the manufacturer's recommendation for each electrode used.
Accurate transcutaneous monitoring requires meticulous electrode and machine calibration. The nurse should be especially aware of the following:
1. Unit calibration and skin warming time vary from 7 to 25 minutes each time the electrode is moved. The nurse should consult the device operator's manual for manufacturer's recommendations applicable to each specific unit.
2. The correlation between PaO2 and PtcO2 should be determined if changes in the patient's clinical condition are observed.
3. The electrodes must be replaced and moved to a new location on the child's trunk or extremities at regular intervals to avoid skin irritation and compromise in electrode performance from heat-induced edema or other tissue changes at the electrode site. Microelectrodes heated to 44° C may require changing only every 6 hours, whereas large cathode electrodes require repositioning every 2 to 3 hours (check manufacturer's recommendations).
4. The nurse should recognize electrical drift and/or other sources of machine error.
5. Alarm systems (for low or high PtcO2) should be established and verified at regular intervals.
6. The procedures for troubleshooting problems with the monitor should be available in the unit.
In some hospital units, nurses are required to obtain an arterial sample for blood gas analysis after every electrode change, to compare the child's arterial blood gas values with transcutaneous values. As noted, the nurse must be knowledgeable about the procedure for machine calibration and maintenance (consult operator's manual and unit protocols) and must also be able to recognize electrical drift and/or mechanical error.
Maintenance should include frequent observation of the fluid space in the sensor. Gain or loss of fluid results in an erroneous PtcCO2 measurement. Each nurse must be familiar with interpretation of measurements, procedures for troubleshooting, and setting of alarm systems.
Noninvasive Blood Gas Monitoring: Pulse Oximetry
Oxygen saturation of hemoglobin (oxyhemoglobin saturation) in arterial blood may be monitored continuously using a pulse oximeter. The pulse oximeter is the monitor of choice for noninvasive monitoring of oxygenation, and the accuracy of these monitors has been demonstrated in children over a wide range of clinical conditions.29,42,136,157,158,159 The response time of the oximeter is shorter than that of the transcutaneous oxygen monitors. The oximeter does not require calibration, and there are virtually no risks to the patient.
Mode of Operation
An instrument probe, housed in a clip or on an adhesive strip, may be placed on the finger, toe, foot, hand, or ear lobe. In the probe are two light-emitting diodes that emit red and infrared light through the tissue to a photodetector (Fig. 21-31). The red light absorption will be inversely related to the amount of saturated hemoglobin (i.e., hemoglobin that is bound to oxygen) passing through the tissue. Well saturated hemoglobin absorbs little red light and desaturated hemoglobin (i.e., hemoglobin not bound to oxygen) absorbs a large amount of red light.
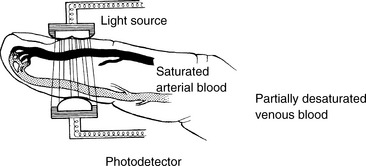
Fig. 21-31 Pulse oximeter. Two light-emitting diodes (light sources) transmit a red and an infrared light through the pulsatile tissue bed. The photodetector must be placed directly across the pulsatile tissue bed from the light source. Oxygenated (saturated) and deoxygenated (desaturated) hemoglobin absorb red and infrared light differently, and the hemoglobin saturation (percent of total hemoglobin that is oxygenated) is related inversely to the amount of red light absorbed.
Because the probe is placed over a pulsatile tissue bed, the pulse oximeter unit computes a pulse rate, and can signal the strength of the pulse. However, patient movement can produce artifact that significantly affects the accuracy of the pulse measurement. A difference between the pulse rate displayed by the pulse oximeter and the heart rate detected by the cardiac monitor should prompt an immediate evaluation of the patient and the patient's systemic perfusion.
Pulse oximeters require pulsatile blood flow to operate properly. These monitors generally provide accurate results over a wide range of clinical conditions (including hypotension, low cardiac output, and hypothermia).29,42,72,100,157,173,177,183 However, the response time in the presence of hypoxemia varies widely from instrument to instrument. Some units may fail to reflect acute, severe hypoxemia and may overestimate the hemoglobin saturation. This tendency may be observed more frequently with finger probes than with ear probes.100,158
Most oximeters provide a low signal alert, which may indicate diminished pulse intensity under the probe. Accuracy of the measured O2 saturation is not necessarily altered by low pulse intensity,100 but the low signal alert should prompt evaluation of the patient and the patient's systemic perfusion.
The pulse oximeter may be a useful adjunct to cardiovascular monitoring. It enables monitoring of the pulse in an ischemic limb and reflects changes in pulse rate in unstable patients and in patients during pacemaker support.
Pulse oximeters are not reliable in the presence of methemoglobinemia and carbon monoxide poisoning, because the effect of these hemoglobins on light absorption is not factored into the calculation of hemoglobin saturation. As a result, when these conditions are present, the pulse oximeter will display a falsely high hemoglobin saturation, reflecting only the percent saturation of the child's normal hemoglobin, rather than the saturation of the child's total hemoglobin.
Troubleshooting During Pulse Oximetry
Several operator-controlled variables may cause a poor signal (and resulting alarm), including the presence of ambient light and movement artifact. To eliminate ambient light, the monitored area can be wrapped loosely with an opaque material such as gauze. The nail bed should be clean before sensor application.36,77,189
Movement artifact is difficult to control in pediatric patients. Ear clip sensors are often the least likely to be disturbed with movement. However, if movement artifact is a significant problem the oximeter may be placed on a restrained extremity. In addition, application of a disposable sensor on the hand or foot may result in less movement artifact than that occurring with placement over a finger or toe. Occasionally, the patient may require sedation (with physician input and order).
Neonatal studies suggest that attachment of the probe to the patient before it is attached to the device may shorten the time required to obtain an initial signal.132 The nurse must be familiar with the rapidity of signal response required for the pulse oximetry devices used in the critical care unit.
Noninvasive Capnometry: End-Tidal Carbon Dioxide Monitoring (PETCO2)
Instrumentation
The exhaled carbon dioxide is the tension (in mm Hg) or partial pressure of carbon dioxide in expired (exhaled) gas. The highest exhaled carbon dioxide tension is present at the end of the expiration; this end-tidal CO2 or PETCO2 is also known as the end-expiratory CO2. The PETCO2 normally trends with the patient's arterial carbon dioxide tension.
The continuous measurement of the PETCO2 by infrared spectroscopy is called capnometry. Capnography includes a display (graph) of the waveform of the carbon dioxide tension throughout inspiration and exhalation and the monitor typically provides a digital display of the PETCO2. Analysis of the capnogram is discussed in the section that follows (see Clinical Applications of Capnography).
End-tidal CO2 monitors can now be purchased as part of a mechanical ventilation system or as separate monitors. Capnometry can also be performed via nasal cannula during spontaneous breathing.170a
The infrared CO2 analyzer consists of three components: an inferred radiation source, a gas sampling chamber, and a detector. Carbon dioxide absorbs infrared radiation of specific wavelengths, so as the infrared rays are passed through expiratory gas, a detector then registers the intensity of the radiation in the gas (and conversely, the absorption of infrared radiation) to determine the tension or partial pressure of CO2.171 This analyzer therefore enables evaluation of alveolar ventilation and CO2 elimination.
Two types of PETCO2 monitoring methods are used clinically—mainstream and sidestream analyzers. In mainstream analyzers the detector module is placed in the ventilator circuit at the proximal end of the endotracheal tube, in line with the expired gas flow; this placement allows for rapid response to changes in PETCO2. However, the mainstream module can be affected easily by condensation and mucus, and requires frequent cleaning of the sensor module.
In sidestream modules a low flow (50 to 150 mL per minute) vacuum aspirates a small sample of expired gas to the analysis module located in the monitor. This sampling method is also affected by condensation and mucus, which can occlude the sampling line. Sidestream technology has also been found to be relatively inaccurate at small tidal volumes, increased I:E ratios, and high resistance states.95,106
The presence (but not the tension) of CO2 in exhaled gas can be detected through use of a colorimetric device attached to the proximal end of an endotracheal tube (or between an endotracheal tube and a resuscitation bag or mechanical ventilation system). Colorimetric CO2 detection devices change color (typically from purple to yellow, but the color change may vary from device to device) when CO2 is present in the gas flowing through the device. Typically the color change develops within about 6 breaths, and the color will change permanently after several minutes or hours of use. These devices are considered qualitative (i.e., indicating presence or absence of CO2) rather than quantitative devices, and are not discussed further in this chapter (see Chapter 9 for additional information).
Clinical Applications of Capnography
The correct use of a PETCO2 device requires an understanding of the patient's alveolar-arterial (A-a) CO2 gradient. In addition, the PETCO2 must be evaluated in conjunction with the clinical examination.
Relationship Between Arterial and End-Tidal CO2 Tension
Normally there is no significant difference between arterial and alveolar (or end-tidal) CO2 tension, because carbon dioxide in pulmonary capillaries normally diffuses freely into the alveoli to be exhaled and measured as PETCO2. Thus, the normal difference between the PETCO2 and the arterial CO2 tension is 2 to 5 mm Hg or less.165,183 This small difference results from mixing of CO2-containing alveolar gas with exhaled gas devoid of CO2 from anatomic dead space. This high correlation has been documented even in neonates,120 particularly if they receive muscle relaxants during mechanical ventilation.
The PETCO2 will be nearly identical to the patient's arterial CO2 tension (PaCO2) only if there is a normal (very low) alveolar-arterial carbon dioxide (A-aCO2) gradient and no alveolar dead space. If a large A-aCO2 gradient or a large amount of alveolar dead space is present, the PETCO2 will not equal the PaCO2. For example, if there is impairment of CO2 diffusion from the blood into the alveoli, such as in the neonate with respiratory distress syndrome or the child with acute respiratory distress syndrome,120,183 the difference between the PETCO2 and the arterial CO2 tension increases. However, changes in the PETCO2 accurately reflect trends in the child's PaCO2, even if a significant lung disease is present.
Any time the PaCO2-to-PETCO2 gradient increases, dead space ventilation has increased. Increased dead space ventilation occurs any time pulmonary perfusion decreases relative to alveolar ventilation. Conditions causing increased dead space ventilation include pulmonary vascular disease or increased pulmonary vascular resistance, pulmonary embolus, decreased right ventricular output (e.g., shock or cardiac arrest), and excessive PEEP. A sudden decrease in the PETCO2 to zero could indicate extubation, ETT obstruction, esophageal intubation, or a disruption or leak in the system
The PETCO2 will not correlate with the PaCO2 if the child is breathing rapidly and shallowly or hyperpnea is present. Such alterations in respiratory patterns will result in sampling error and poor correlation between PETCO2 and PaCO2.95,106,171
Analysis of Capnograph
The waveform that is displayed by variations in exhaled CO2 throughout the respiratory cycle is known as a capnogram. To use the capnogram, the clinician must first be familiar with a normal PETCO2 waveform (Fig. 21-32).183
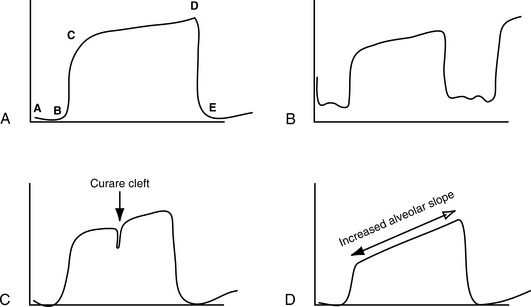
Fig. 21-32 Capnogram phases. A, Normal capnogram illustrating each phase of exhalation (see text, Analysis of Capnograph). B, Rebreathing capnogram: Inspiratory level does not return to zero. C, Curare cleft: Cleft in the alveolar plateau indicates a return of diaphragmatic activity in a patient receiving neuromuscular blockade. D, Increased alveolar plateau slope is representative of small airway obstruction (e.g., asthma, bronchiolitis).
A capnogram is divided into four phases. Phase A-B is the inspiratory cycle, when no carbon dioxide is detected. Phase B-C is the beginning of exhalation (emptying of dead space and alveolar gas), at which point carbon dioxide tension rapidly increases.183 Phase C-D reflects exhalation of predominantly alveolar gas, and is also known as the “alveolar plateau.” Point D is the “end-tidal” point at which carbon dioxide exhalation is at its maximum level (i.e., the PETCO2). Phase D-E is the beginning of inspiration (inhalation of CO2 free gas) where the waveform returns to zero.
Evaluation of Changes in PETCO2.
Analysis of the capnogram and trends in the PETCO2 may enable detection of improvement or deterioration in ventilation, increase in dead space, a fall in cardiac output and ET tube displacement. It can enable detection of an obstructed airway, and/or the return of diaphragm function in the paralyzed patient.183 If the PETCO2 and the PaCO2 fall together, the patient's ventilation has improved; if both rise, the patient's ventilation is reduced.
As noted, if the PETCO2 falls and the PaCO2 rises, dead space has been added to the system (areas are ventilated thats are not perfused). This condition can develop if the lung is overdistended with PEEP. The PETCO2 can fall without a rise in the PaCO2 when cardiac output falls, and pulmonary blood flow and delivery of CO2 to the lungs decreases. The PETCO2 will fall to near zero with spontaneous extubation. For additional information about PETCO2 monitoring in the child with pulmonary disorders, see Chapter 9.
Recently capnography has been used to monitor resuscitation quality during cardiac arrest in intubated patients. When cardiac output is very low during attempted resuscitation, little blood flow is delivered to the lungs, so little carbon dioxide is detected in exhaled gases. When blood flow improves during attempted resuscitation, blood flow to the lungs improves and the PETCO2 rises. The PETCO2 rises abruptly when there is return of spontaneous circulation (see Chapter 6).
Nursing Considerations
When capnometry is in use, the nurse must be able to calibrate the instrument and must be aware of the relationship between the patient's alveolar (or end-tidal) and arterial CO2 tensions. The nurse must be able to correlate PETCO2 values with the clinical status of the patient and must be aware of sources of instrument error.
The absolute PETCO2 at any one time is usually not as important as the trends documented by this equipment, although a sudden fall in PETCO2 in an intubated patient should prompt immediate suspicion of spontaneous extubation (see Fig. 9-21). The PETCO2 should be compared with either a venous or arterial PCO2 to ensure that the two correlate. In addition, it is critical to verify effectiveness of oxygenation and ventilation through careful clinical assesment.183
Invasive Arterial Oximetry
Arterial oximetry is an invasive method of continuously monitoring arterial oxygen saturation. This method of oximetry uses an intraarterial electrode threaded through to the tip of an arterial catheter.
Clinical trials have demonstrated the accuracy of the polarographic electrode, although a high incidence of electrode failure has been reported.55 LeSoeuf 101 reported moderately high correlations between the indwelling oximeter and the oxyhemoglobin saturation reported by a PtcO2-measuring device. However, the arterial oximetry was not compared with oxygen saturation measured by direct blood sampling and measurement using a co-oximeter.
Clinical Applications
Arterial oximetry allows direct, continuous measurement of oxygen saturation, which is preferable to intermittent measurement. The indwelling electrode does not require frequent repositioning (e.g., as is necessary with transcutaneous oxygen monitoring).
The disadvantages of arterial oximetry are related to vascular effects of a foreign body and the potential for inaccurate results. The catheter may lodge against the arterial wall, producing arterial spasm. Fibrin clots may form at the catheter tip, resulting in inaccurate readings and risk of embolism. Hemodilution (particularly a hematocrit < 30%) may result in erroneous oxygen saturation calculations. Finally, electrode failure has been reported.
Use of the arterial oximeter may be limited in pediatric patients because they have small vessel size. The risks of infection, thromboembolic events, and other potential complications of the indwelling arterial oximeter have not been documented in pediatric patients. Currently, arterial oximetry is not performed frequently in children because further research is required to verify its effectiveness.
Invasive Mixed Venous Oxygen Saturation Monitoring
Continuous monitoring of mixed venous oxygen saturation (SvO2) in pediatric patients is possible with a fiberoptic pulmonary artery catheter. The central venous oxygen saturation (SCVO2), typically obtained from the superior vena cava, approximates the mixed venous oxygen saturation (typically the SCVO2 is about 2% to 3% higher than the SvO2), and is often used as a surrogate for the mixed venous oxygen saturation. The SCVO2 can be monitored with a fiberoptic central venous catheter.
Changes in the SvO2 (and SCVO2) may reflect alterations in cardiac output, oxygen delivery, hemoglobin, or changes in oxygen consumption. In some instances, oxygen demands are changing continually (as with sepsis), and continuous SvO2 (SCVO2) monitoring may provide an early indication of decreased oxygen delivery, increased oxygen demand or decreased oxygen utilization.
Description
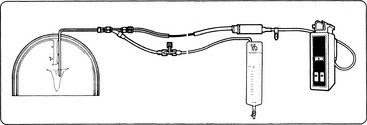
The SvO2 can be monitored with a fiberoptic 5-French, 5-lumen balloon-tipped pulmonary artery catheter that also enables measurement of pulmonary artery pressure, central venous pressure, and pulmonary wedge pressure, and for thermodilution cardiac output calculations.
Continuous monitoring of SCVO2 is performed using a fiberoptic central venous catheter. Pediatric central venous oximetry catheters are available in 4.5 and 5.5-Fr sizes and three different lengths (Edwards PediaSat, Edwards Lifesciences, Irvine, CA).110a
The hemoglobin saturation is determined by spectrophotometric reflection. The catheter port used for SvO2 monitoring is connected to an optical module, which transmits a narrow band width (wavelength) of light to the tip of the fiberoptic catheter. This light will be reflected by saturated (oxygenated) hemoglobin differently than by desaturated hemoglobin (i.e., hemoglobin not bound to oxygen).
The light emitted by the catheter is reflected by the hemoglobin and transmitted back via a separate fiberoptic filament to the module (Fig. 21-33). The microprocessor analyzes the amount of light transmitted and reflected and averages the signal over 2 to 3 seconds. A tandem recorder trends the SvO2 on a graph recording. A light intensity indicator (at the tip of the catheter) is also recorded and monitored; changes in the light intensity may indicate a change in catheter position, inadequate blood flow, or damage to the fiber optics.19,62,81
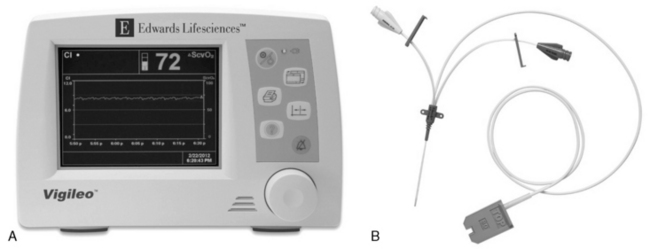
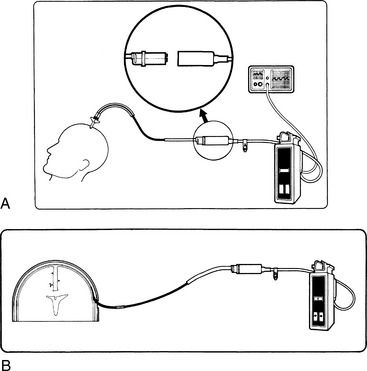
Fig. 21-33 A, Continuous central venous oxygen saturation (SCVO2) microprocessor/monitor and B, multilumen central venous catheter. The fiberoptic catheter contains filaments that transmit red light to and from the blood. The light is reflected by circulating hemoglobin and transmitted via a second optical fiber to the microprocessor, where the hemoglobin saturation is determined. Catheters are available in 3 lengths. (Vigileo monitor and PediaSat catheter photograph courtesy of Edwards Lifesciences, Irvine, California.)
Two different reflection spectrophotometry processor units are available: a three-wavelength and a two-wavelength device. The three reference wavelength processor is thought to be more accurate over a wide range of physiologic conditions, particularly changes in hemoglobin concentration. When a device with two reference wavelengths is used, the hemoglobin concentration must be entered whenever it changes55 (typically this calibration is performed at regular intervals, based on institutional protocol and manufacturer's recommendations). The bedside team must familiarize themselves with published experience of the device in use to be aware of accuracy of the device and potential drift.55,110a,187
Operation
Unit calibration is recommended before insertion of the catheter. This calibration is performed using the catheter, the processor, and a standard optical reference provided with the catheter. If the SvO2 monitor is part of a pulmonary artery catheter, the appropriate pressure transducers also must be calibrated and connected to their respective monitoring systems and catheter ports before the catheter is placed.
Insertion into the central vein is accomplished using a standard Seldinger technique. Pressure waveforms and SvO2 readings are monitored during insertion to guide catheter placement.
If the fiberoptic is contained in a pulmonary artery catheter, placement is correct when the pulmonary catheter is correctly positioned. If the tip of the pulmonary artery catheter is advanced too far into a pulmonary artery, a falsely high SvO2 will be calculated, reflecting the proximity of the fiberoptic to blood that is oxygenated by surrounding alveoli.154
Recalibration after insertion should be performed according to manufacturer's recommendation: a mixed venous blood sample is sent for laboratory measurement of the oxygen saturation using a co-oximeter (i.e., not a calculated saturation). Simultaneous with the blood sampling, the nurse should document the mixed venous oxygen saturation displayed by the device. Once the laboratory analysis of the blood sample is complete, the device should be recalibrated based on the laboratory measurement.
Clinical Applications
The normal mixed venous oxygen saturation is usually between 65% and 75%. A rise in SvO2 reflects one of four conditions: (1) increased oxygen delivery, caused by a rise in cardiac output, or an increase in arterial oxygen content; (2) reduced oxygen consumption, as observed with hypothermia, neuromuscular blockade, and anesthesia; (3) the presence of a left-to-right intracardiac shunt; or (4) mechanical interference from the measuring unit (e.g., a wedged catheter).
The SvO2 may fall for the following reasons: (1) decreased oxygen delivery, resulting from decreased cardiac output, hypoxemia associated with pulmonary dysfunction, or anemia, or (2) increased oxygen consumption caused by shivering, seizures, hyperthermia, sepsis, or agitation. The SvO2 will track with changes in cardiac output, but there are other factors, such as oxygen demand and oxygenation of blood in the lungs, that influence the SvO2.
The SvO2-monitoring system has been used in adult patients since 1981, and its efficacy has been documented in a variety of physiologic conditions.19,55,62,81,93,122,146,154,187 The enthusiasm for its use in adult patients is based on its accuracy and reliability as well as its ability to reflect changes in cardiac output and arterial oxygenation instantly and continuously.
The use of venous fiberoptic oximetric catheters has been successful in the management of pediatric patients following surgery for congenital heart disease and for pediatric patients with septic shock. Muller and co-workers described the use of a 2-French fiberoptic probe that is introduced through a single-lumen central venous catheter in a series of three infants who underwent a stage 1 Norwood procedure.126 The fiberoptic probe was inserted approximately 2.5 cm beyond the central venous catheter tip, allowing for continuous central venous oximetry (SCVO2). The group found good correlation between the fiberoptic SCVO2 and laboratory SvO2 evaluated by co-oximeter (r = 0.912. 95% CI: 0.716-0.975).126 However, they reported that the fiberoptic probe was less accurate in conditions of very low saturation (< 40%).
More recently, de Oliveira reported that goal-directed resuscitation using the end point of a superior vena caval oxygen saturation (ScvO2) ≥ 70% significantly improved the outcome of pediatric patients with septic shock (28-day mortality 11.8% vs. 39.2%).43 Since 2006, pediatric SCVO2 catheters have been approved by the Food and Drug Administration for use in children in the United States.
Disadvantages
Changes in the calculated SvO2 (or SCVO2—in this section they are used interchangeably) are not always indicative of changes in patient condition. The high incidence of artifact during continuous SvO2 monitoring seems to be linked to the fact that SvO2 determination is dependent on reflected light. As rapid blood flow passes the catheter tip, consistent light reflection may not occur. Faulty connections, fiberoptic fracture, occlusion of the catheter tip by emboli, and wedging of the tip against the vessel wall also may produce inaccurate SvO2 readings.122,154
Whenever a fall in SvO2 is noted, the patient's oxygenation and systemic perfusion must be assessed. Clinical evaluation always should be used to confirm any changes associated with deterioration in the SvO2. The function of the monitor should be assessed only after the patient's condition is evaluated.
Factors reducing the accuracy of fiberoptic SvO2 monitoring include lack of in-vitro and in vivo calibration, lack of intensity calibration during insertion, bent or broken optics, catheter tip close to or facing the vessel wall, increased carboxyhemoglobin or methemoglobin, and extreme hypoxemia (< 40%).171
The limited variety of available sizes of the fiberoptic catheters prevents its use in very small children. Although the 2- to 4-French SvO2 fiberoptic catheter can be used in infants and for central vein oximetry, these small fiberoptic catheters do not enable other hemodynamic measurements and calculations (e.g., CO, PAWP, vascular resistances) that are possible with a pulmonary artery catheter with fiberoptic and thermistor. With the availability of the pediatric fiberoptic central venous catheter more experience is being gained with continuous monitoring of central venous oxygen saturation as a surrogate for the mixed venous oxygen saturation in critically ill children.110a More data about its reliability and effectiveness is anticipated.
Esophageal Pressure Monitoring
Esophageal pressure (Pes) monitoring provides important information regarding intrathoracic pressure during mechanical ventilation. It yields an indirect measurement of pleural pressure, providing information regarding the distending pressure of the lung and chest wall. Evaluation of Pes also allows for division of the respiratory system's resistance and compliance into pulmonary and chest wall components.
A change in Pes (ΔPes) as well as Pes swings reflect the level of patient effort during spontaneous and supported mechanical breaths; these values can be used to calculate the work of breathing imposed by the lung and ventilator circuit. The Pes may also be useful in determining optimal PEEP in patients with acute lung injury/acute respiratory distress syndrome (ALI/ARDS).
In a randomized controlled trial, Talmor and colleagues reported that a “ventilator strategy using esophageal pressures to estimate the transpulmonary pressure significantly improves oxygenation and compliance” in patients with ALI/ARDS.20,166 They demonstrated improved PaO2:FiO2 ratios, respiratory system compliance, and dead space to tidal volume ratio (Vd/Vt) when the PEEP was set to keep the transpulmonary pressure greater than 0 cm H2O guided by Pes monitoring.20,166
Instrumentation
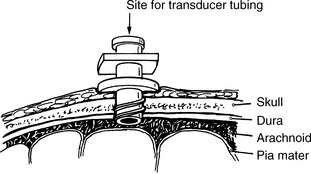
Esophageal pressure is measured using an air-containing balloon that is sealed in a catheter connected to a pressure transducer. The catheter is inserted into the thoracic esophagus. A change in pressure imposed on the balloon is conveyed via the catheter to the pressure transducer, and the pressure is displayed on the monitor or ventilator.
Catheter dimensions range from an internal diameter of 1 to 1.2 mm (for use in newborns and smaller children) to an internal diameter of 1.4 to 1.7 mm (for use in large children and adults). Adequate balloon volume is essential for accurate detection of the Pes.20,171 Typically, a balloon volume of 0.5 mL is sufficient; however, the range of gas volume should be determined from the catheter packaging; the packaging indicates the clinical situations in which more or less volume may be indicated.171
The balloon is positioned by passing an empty balloon catheter into the stomach via the nares or mouth. A volume of 0.5 mL of air is then injected into the system and the catheter is attached to the pressure transducer.171 A positive pressure swing indicates that the balloon is in the stomach. The catheter is then withdrawn until a negative pressure deflection is identified on the monitor, indicating the balloon is in the thoracic esophagus. The catheter is then pulled back an additional 5 to 10 cm, which positions the entire balloon in the esophagus.
The final position of the balloon in the mid- and lower esophagus can be verified by chest radiograph.20 Correct position of the balloon can be validated by using the “occlusion test,” which is accomplished by having the patient take a spontaneous breath against a closed airway while observing the ΔPes and change of pressure at the airway opening (ΔPAO).171 If the balloon is in correct position, the clinician will find near unity between the ΔPes and ΔPAO throughout the inspiratory cycle.171
Clinical Applications
Esophageal pressure monitoring has a wide range of clinical applications. As noted, Pes is beneficial in determining the patient's work of breathing imposed by underlying lung disease and the addition of the ventilator circuit. Evaluation of the work of breathing allows adjustment of mechanical support to decrease the patient's metabolic demands and/or identify if the patient is ready for extubation.
In addition to work of breathing assessment, Pes monitoring provides information regarding respiratory muscle function. More specifically, monitoring of the Pes in conjunction with gastric pressure monitoring enables calculation of diaphragm force-generation, relatively isolated from intercostal and other accessory muscles, and from elastic recoil of the chest wall. Some authors have suggested that isolated diaphragm function should be assessed using Pes measurement in patients with suspected diaphragm weakness or paralysis.20
Esophageal pressure measurement may be beneficial in any clinical situation characterized by decreased lung compliance.20,166,171 If poor respiratory system compliance is determined to be caused by chest wall edema, low lung compliance, or abdominal compartment syndrome, the clinician may be better able to titrate PEEP without causing lung trauma.
Oxygen Administration Systems
Supplementary oxygen is commonly used in the pediatric critical care unit. Although oxygen may be administered in a variety of ways, it must always be treated as a drug, with accurate administration and documentation of the dose and careful assessment and documentation of patient response.
The appropriate device for delivery of supplementary oxygen is determined by the patient's age, size, and inspiratory flow rate (tidal volume in mL/sec), and the fraction of inspired oxygen (FiO2) needed. The method for estimation of inspiratory flow rate is summarized in Box 21-1. Oxygen delivery devices can be divided into two classes: variable-performance oxygen delivery devices (low flow devices) and fixed-performance oxygen delivery devices (high-flow devices).
Box 21-1 General Principles for Estimation of Required Oxygen Inspiratory Flow Rate
1. 60 seconds/min ÷ respiratory rate = time of one respiratory cycle in seconds (s)
2. Multiply time of one respiratory cycle (in seconds) × inspiratory fraction (the portion of the respiratory cycle required for inspiration—typically 0.5, but may be 0.3 in children with asthma and long exhalation) = inspiratory time (in second[s])
3. Divide tidal volume (VT) in mL (typically 4-6 mL/kg) per breath by the inspiratory time (in seconds) = inspiratory flow in mL/s
Note: For convenience, multiply inspiratory flow in mL/s by 60 s/min to compare with gas flow rate in mL/min (and divide mL/min by 1000 to obtain L/min to compare to gas flow rate)
Examples
Example 1: 4 kg Infant
Would a 2 L/min oxygen flow through nasal cannula provide high or low flow and low FiO2 for a 4 kg infant?
The infant's normal Vt is 20 mL (using 5 mL/kg). This example uses a typical respiratory rate of 60/min with an inspiratory portion of 0.5/breath.
1. 60 s/min ÷ 60/min = respiratory cycle of 1 s
3. 20 mL/breath ÷ 0.5 s = 40 mL/s inspiratory flow (× 60 s/min = 2400 mL/min = 2.4 L/min)
A nasal cannula delivering a flow of 2 L/min gives a flow rate that would almost meet the inspired flow rate of a 4 kg infant, and thus the infant's inspired FiO2 would be fairly high.
Example 2: 20 kg preschooler
Would a 2 L/min oxygen flow through nasal cannula provide relatively high or low flow and low FiO2 for a 20 kg preschooler?
A 4 year old, 20 kg child's normal tidal volume (VT) is 100 mL.
1. 60 s/min ÷ 30/min = respiratory cycle of 2 s
2. 2 s × 0.35 = 0.7 s inspiratory time
3. 100 mL/breath ÷ 0.7 s = 142 mL/s (× 60 s/min = 8571 mL/min = 8.6 L/min)
The preschooler is breathing at a slower rate than the infant, so the preschooler's inspiratory time is slightly longer (here estimated at 0.7 s for each breath). Mean inspiratory flow rate is ~ 142 mL/s or 8571 mL/min (8.6 L/min), well higher than the delivered oxygen flow 2 L/min. The preschooler will entrain much more room air so the inspired FiO2 is low.
Courtesy of Mary Fran Hazinski, Arno Zaritsky, and Stephen S. Schexnayder.
Low flow (variable-performance) devices are unable to deliver an oxygen flow rate sufficient to supply the patient's inspiratory flow rate. As a result, when the child inspires, room air is entrained with the supplementary oxygen and the FiO2 varies with the patient's respiratory rate and tidal volume.183
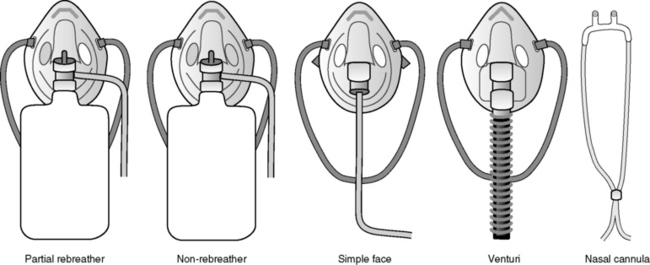
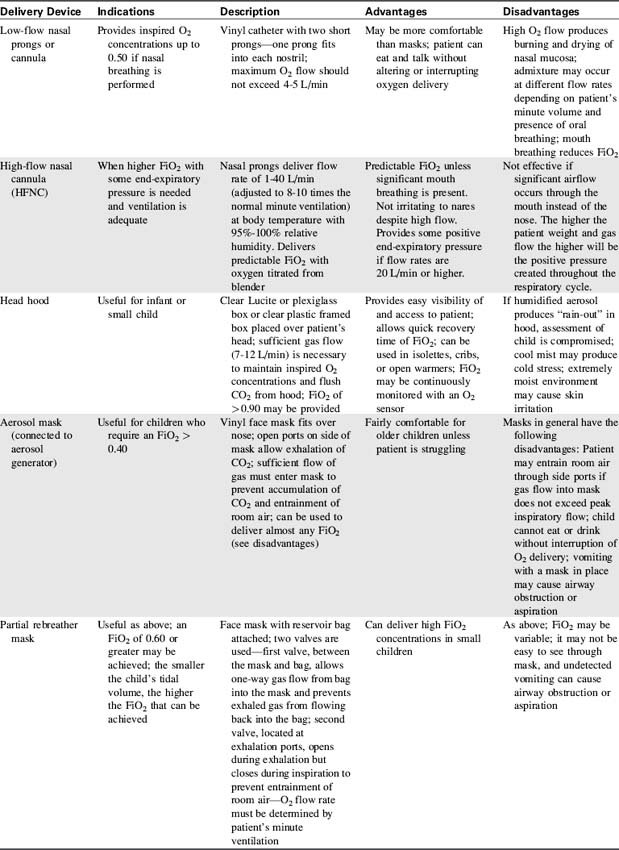
High flow (fixed-performance devices) can deliver an oxygen flow rate that meets or exceeds the patient's inspiratory flow rate. As a result, a high FiO2 can be consistently delivered. Fig. 21-34 and Table 21-9 describe types of oxygen delivery systems and their advantages and disadvantages.
The nurse caring for the child receiving supplementary oxygen must monitor and record the type of oxygen delivery device, the set liter flow (in liters per minute), the FiO2, and the child's response to therapy.
Nursing Considerations
The bedside nurse should assess the patient and the oxygen delivery system:
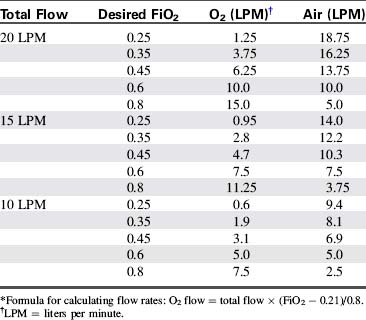
1. Analyze the fraction of inspired oxygen (FiO2) frequently—many hospitals require continuous or hourly analysis. If O2 drift is a problem, continuous analysis of the inspired oxygen concentration is usually indicated. Table 21-10 provides a formula for calculation of inspired oxygen concentrations.
2. Monitor patient oxygenation through pulse oximetry and, as needed, obtain an arterial blood gas analysis (the gold standard) to evaluate effectiveness of oxygen therapy. The nurse and the physician should determine the desired frequency of blood gas analysis. Invasive or noninvasive evaluation of oxygenation should be performed 15 to 20 minutes after any change in FiO2.
3. Observe for changes in respiratory rate, effort, and/or skin color of the patient with any changes to the oxygen delivery device or FiO2. Document these observations and notify the physician or on-call provider of any clinical changes.
4. Ensure that the inspired oxygen is humidified and warmed unless otherwise directed by the physician or on-call provider.
5. Ensure that any tubing in the O2 delivery system is changed daily to minimize the risk of tubing contamination and healthcare acquired infection.
6. Keep infants and children dry when humidified oxygen is provided by head hood or face tent. Frequent clothing and linen changes may be necessary. Monitor the child's temperature closely if heated or cooled aerosol is used.
7. Assess for potential complications of oxygen therapy.28,183
Mechanical Ventilation
Assisted ventilation is indicated for patients who are unable to maintain adequate oxygenation or eliminate carbon dioxide, or who develop refractory circulatory failure. These patients generally exhibit clinical signs of respiratory failure or shock.
Mechanical ventilation is based on the properties of normal pulmonary function. During inspiration, alveolar pressure must be significantly lower (normal breathing or negative pressure ventilation) or greater (positive pressure ventilation) than atmospheric pressure. This is accomplished in two ways:
(1) By making atmospheric pressure or pressure surrounding the chest more negative
(2) By increasing alveolar pressure via delivery of gas under positive pressure.
These inspiratory mechanisms describe the two major forms of mechanical ventilatory assistance: negative pressure ventilation and positive pressure ventilation.
Negative Pressure Ventilation
Negative pressure ventilation is a relatively uncommon mode of ventilatory support that creates a negative (with respect to airway opening pressure) extrathoracic pressure, so the child's chest expands, causing inspiration. Because negative pressure ventilation is provided without the need for insertion of an artificial airway (e.g., endotracheal tube), it is a form of noninvasive ventilation.
Description
A tank or shell surrounds the thorax; negative pressure is created around the thorax by a vacuum. The negative pressure “pulls” the thoracic cage outward, thereby increasing intrathoracic volume and reducing intrathoracic pressure. A pressure gradient is then present between the mouth and the intrathoracic space (where the pressure is now approximately − 10 to − 15 cm H2O), so air flows into the alveoli. Exhalation occurs passively when the vacuum cycles off.183
In order to generate the subatmospheric pressure around the thorax, the shell must seal around the arms, neck and lower abdomen. This required proper fit. If diapers are used, they should be secured outside (below) the shell.
Clinical Applications
Negative pressure ventilation may be used for support of children with chronic respiratory failure secondary to neuromuscular disease, such as the child with phrenic nerve injury or muscular dystrophy. The child's lung tissue must allow normal gas diffusion (i.e., pulmonary interstitial disease cannot be present), and the child must be able to maintain a patent airway (with an effective cough reflex).
An advantage of this form of ventilation is that endotracheal intubation is not required. Supplementary oxygen may be administered by nasal prongs or face mask.
Advantages and Disadvantages
There are some distinct disadvantages to negative pressure ventilation, which limits its use. The tanks are cumbersome for the patient and caretakers and they render the child virtually immobile. “Shell” devices must be fitted precisely to the child's thorax to obtain a good seal. Even if the tank fits properly, it is frequently difficult to achieve a good seal around the arms, neck, and abdomen. Air leaks diminish the effectiveness of the machine, thus reducing chest expansion and alveolar ventilation.
Exaggerated dilation of the thoracic great vessels and diminished cardiac output have been reported during negative pressure ventilation. In addition, venous pooling may develop in the legs.
In spite of the challenges during use, negative pressure ventilators may be extremely useful in the care of chronically ill ventilator-dependent patients. This form of ventilation support may be particularly useful for home ventilator therapy because the devices are relatively easy to operate and do not require an advanced (invasive) airway. With practice, the child is able to talk during negative pressure ventilation.28
Nursing Considerations
As with all other devices, the nurse must be knowledgeable about the operation of a negative pressure ventilator. At all times a resuscitation bag and mask with an O2 source should be available to provide manual ventilation in case of machine malfunction.
The child's heart rate, blood pressure and systemic perfusion must be closely monitored when instituting negative pressure ventilation and whenever the ventilator is adjusted. It is also important to monitor the patient closely for signs of venous dilation and signs of hypovolemia (potentially caused by venous pooling outside of the thorax).
If the child will be discharged home with this ventilator, a teaching program must be implemented for the family in the hospital. The parents must be able to provide bag-mask ventilation and troubleshoot common problems with the ventilator.
Positive Pressure Ventilation
Positive pressure ventilation is achieved by delivery of a gas (oxygen/air mixture) to the patient's proximal airway. Positive pressure ventilation changes the normal pressures during the respiratory cycle, because gas is delivered to the alveoli under positive pressure, creating positive (rather than negative) pressure during inspiration. Expiration occurs passively.
Description
Ventilators may be classified according to the mechanism that terminates inspiration. However, most new ventilators use a combination of cycling mechanisms and a variety of ventilation characteristics.
Volume-cycled ventilators are preset to deliver a specific tidal volume during inspiration. Once this volume is delivered, inspiration stops, allowing for passive exhalation. Pressure-cycled ventilators use a preset peak inspiratory pressure (PIP) at which inspiration is terminated; gas is delivered until the peak pressure is achieved without regard to the volume delivered. Time-cycled ventilators, most commonly used in the neonatal/infant population, are preset to allow a specific inspiratory time; gas will be delivered until that time is reached, without regard for the volume delivered or the peak pressure produced.88,183
Changes in the child's pulmonary resistance and compliance will affect the tidal volume that is actually delivered to the lung. For this reason, close attention to exhaled volume is required. Some centers measure the patient's effective tidal volume (Vteff) to ensure that an acceptable tidal volume is delivered. Most ventilators available today are capable of real-time measurements of Vteff. Effective tidal volume is defined as the actual volume (mL) of gas being delivered to the lung per kilogram (kg) of ideal body weight; it is measured in mL/kg. The most accurate measurements are obtained at the patient wye (the connector that joins the inspiratory and expiratory limbs of a two-limb circuit to the patient's airway); measurement at this point alleviates the need to calculate the compliance factor of the ventilator circuit.32 Depending on the level of PEEP and underlying pathology, acceptable values of Vteff range from 5 to 8 mL/kg.183
The Vteff may be calculated using pressure measurements obtained by the ventilator, with correction for tubing compliance (i.e., the circuit compliance factor). Calculation is performed as follows:
Calculation of Effective Tidal Volume Using Ventilator Values (Values Measured at the Ventilator, not from the patient wye)
* The greater the difference between the PPlateau and level of PEEP or change in pressure (ΔP), the greater the volume lost in the ventilator circuit.183
† The circuit compliance factor is available from the ventilator circuit manufacturer and varies according to the circuit diameter.
Most newer-generation pediatric ventilators have incorporated flow-sensing devices or pneumotachographs at the patient's wye to more accurately determine inhaled and exhaled volumes.32 Monitoring respiratory volumes at the patient's airway removes the inaccuracy resulting from the aforementioned volume loss related to the circuit tubing compliance.32
Most pediatric patients requiring mechanical ventilation in the critical care unit are placed on positive pressure ventilators. Selection of the appropriate ventilator should be based on the following factors:46,88,183
1. Size of the child and minute ventilation requirement. The ventilator maximum and minimum flow rates must be appropriate for the patient.
2. Lung compliance. If the patient requires high inspiratory pressures (> 40 cm H2O), a pressure-cycled ventilator may be preferable to a volume-cycled ventilator.
3. Rapidly changing lung compliance. Optimum ventilation of the patient at risk for rapid or frequent changes in compliance may be most effective using a volume-cycled ventilator (or a combination volume/time cycle).
4. Chest wall stability. If a child has an unstable chest wall (e.g., with flail chest or a median sternotomy incision), volume-cycled ventilators may be most appropriate.
Characteristics of an Ideal Pediatric Ventilator
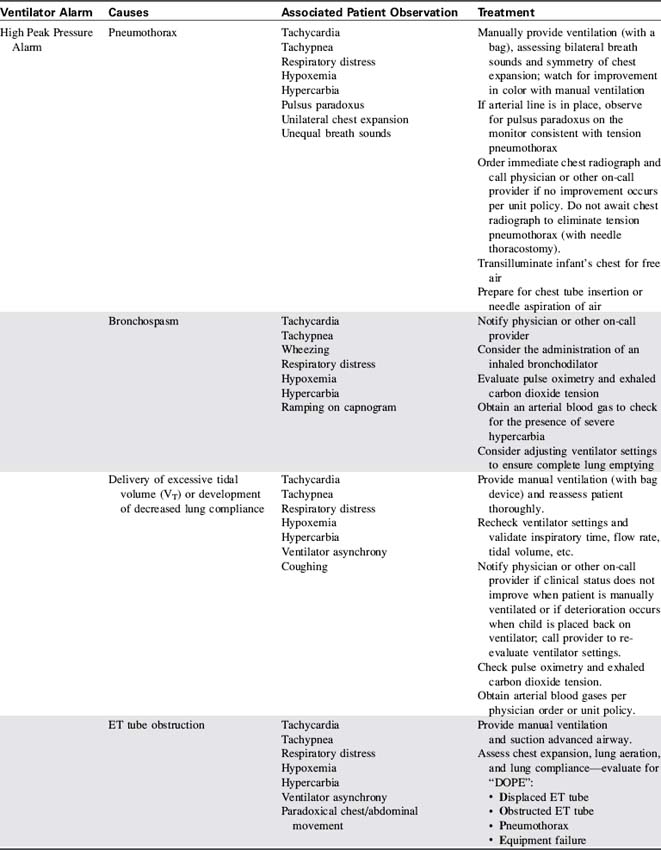
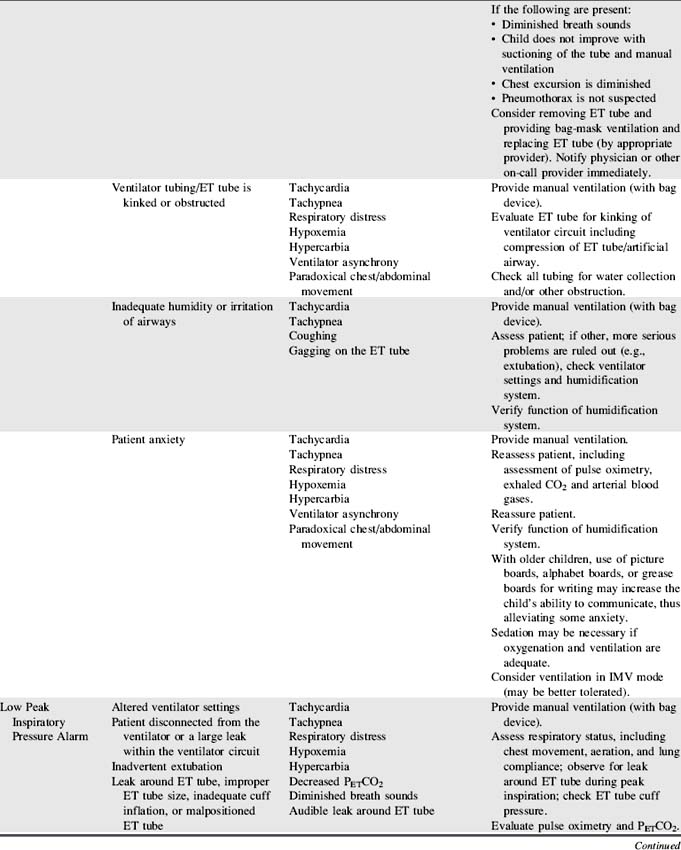
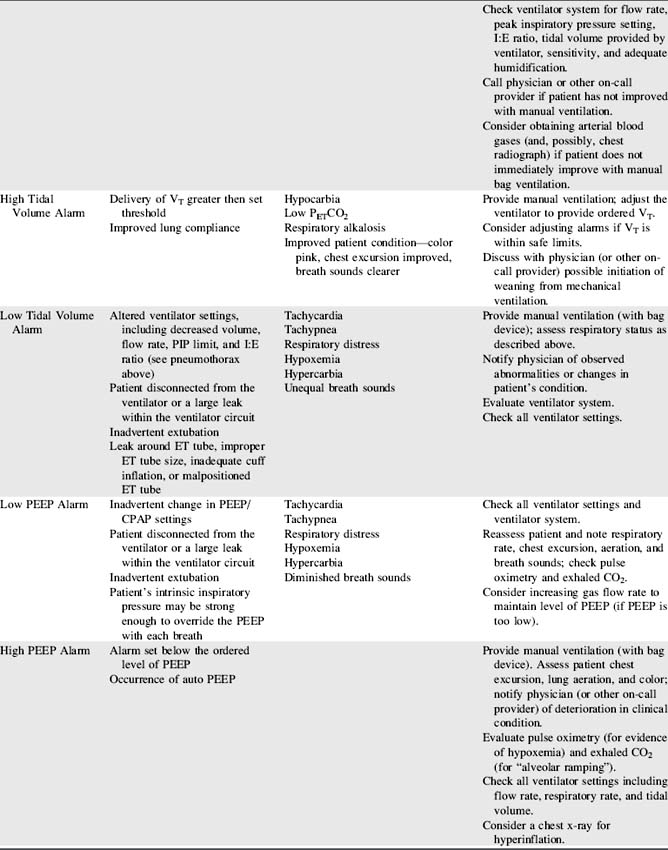
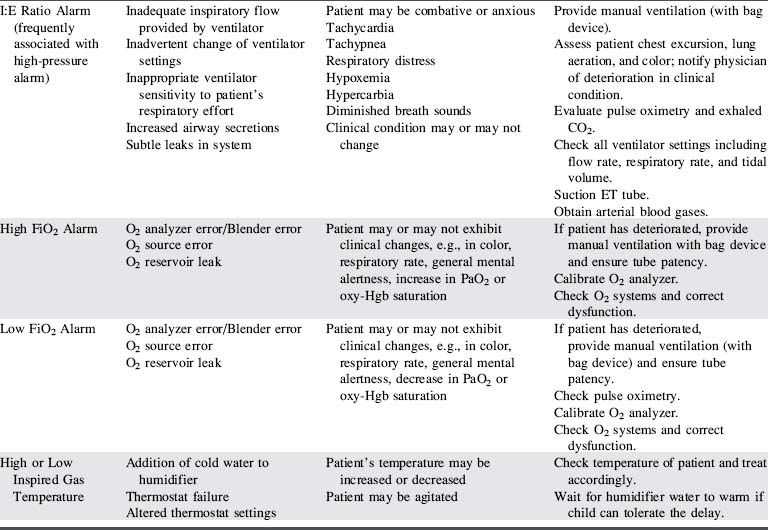
The categories in Box 21-2 represent three groups of criteria for selection of the ideal ventilator for pediatric use. See Table 21-11 for more detail regarding each alarm.
Box 21-2 Characteristics of an Ideal Pediatric Ventilator
Specifications
• Volume, pressure or time cycled; mixed modes of ventilation
• Assist/control, control, CPAP (continuous positive airway pressure), PSV (pressure support ventilation), SIMV (synchronized intermittent mandatory ventilation)
• Tidal volume range of 20-450 mL/breath (minute ventilation of 0.4-6 L/min)
• Respiratory rate of 1-100/min (high-frequency ventilation capability is also desirable)
• Variable inspiratory flow of 0.5-40 L/min
• Variable inspiratory/expiratory flow ratios
Frequently, ventilators with the widest possible clinical application are more practical to purchase than a large number of ventilators with very specific applications. The information in Box 21-2 and Table 21-11 should be considered when selecting ventilators for use with critically ill children, as well as selection of a specific mechanical ventilator for a particular patient.
Clinical Applications
Many of the newer-generation ventilators are capable of providing mechanical support across all or most patient populations. The incorporation of microprocessors into newer-generation ventilators has enabled this wide range of use.
Manufacturer's specifications and recommendations and hospital clinical trials should facilitate the selection of the ventilator(s) that can best serve each patient (e.g., the patient size limit for use of neonatal ventilators is generally 8-15 kg body weight). The clinical condition of the patient will determine the ventilator functions needed to provide optimal ventilation.
Nursing Considerations
Throughout mechanical ventilatory support, the bedside nurse is responsible for assessing effectiveness of ventilation. The use of mechanical ventilation does not ensure that the child is ventilated effectively. The ventilator settings must be evaluated constantly in light of the child's clinical appearance.
When ventilator function is in doubt, the child should be ventilated manually with a hand-resuscitator bag. Table 21-11 offers a troubleshooting guide for use when problems arise during mechanical ventilation. It is intended to address equipment (rather than patient) problems. For more information, see Chapter 9, Positive Pressure Mechanical Ventilation and the section, Nursing Care of the Child During Mechanical Ventilation).
High-Frequency Ventilation (HFV)
An alternative mode of ventilatory support is high-frequency ventilation (HFV). High-frequency ventilation uses a mean airway pressure (map) and rapid respiratory rates (60 to 3600/minutes) to recruit atelectatic regions of the lung. The tidal volumes used are close to anatomic dead space, so this form of ventilatory support does not create risk of volutrauma that can occur with administration of higher tidal volumes. Simply put, HFV allows for higher end-expiratory pressures with lower peak inspiratory pressures.
HFV is used for infants and children with acute lung injury when conventional ventilation has failed. A metaanalysis of HFV versus conventional ventilation in premature neonatesconcluded that the use of HFV did not reduce chronic lung disease or mortality in this patient group.27 With studies including only a small number of pediatric patients, a recent Cochrane analysis concluded that HFV did not improve outcome when compared with conventional mechanical ventilation.72a
There are two basic types of high-frequency ventilators—the oscillator and jet ventilator. Each is presented briefly here. For additional information, see Chapter 9.
High-Frequency Oscillatory Ventilation (HFOV)
High-frequency oscillatory ventilation (HFOV) employs a piston moving at extremely high frequencies (about 180 to 1500 cycles/minute) to create positive and negative pressure swings. Oscillatory ventilation does not produce bulk gas delivery. It uses a continuous gas flow to eliminate CO2 and deliver oxygen to the lung's ventilatory units.
In a controlled randomized multicenter NIH trial,75 HFOV offered no advantages over conventional ventilators in the treatment of neonatal respiratory failure. In this study the incidence of bronchopulmonary dysplasia was similar to conventional ventilation, and mortality rates were equal in both groups.
A randomized, crossover trial of HFOV versus conventional mechanical ventilation in 70 critically ill children with ARDS was published in 1994 by Arnold et al12a Children treated with HFOV demonstrated improved oxygenation and decreased use of supplementary oxygen 30 days later.
High-Frequency Jet Ventilation (HFJV)
The typical HFJV uses an oxygen source and a high-pressure source to deliver gas through a small-bore injector cannula that extends into the endotracheal (ET) tube. This system allows delivery of relatively large tidal volumes at relatively low peak airway pressures. A flow interrupter adjusts the frequency and relative inspiratory time. Valve devices applied to the expiratory limb of the circuit allow the application of PEEP. A continuous infusion of saline into the path of the jet humidifies inspired air. Often a conventional ventilator is used in tandem as the gas and oxygen source for the HFJV unit.
HFJV is used most often used as a rescue therapy for patients with respiratory failure unresponsive to conventional ventilation, as evidenced by rising inspiratory pressures, persistent hypoxemia, and hypercarbia despite maximal conventional ventilatory support. In recent years high frequency jet ventilation (HFJV) has become less widely used than HFOV.
Mechanisms of Gas Exchange with High Frequency Ventilation
The mechanisms of gas exchange during high-frequency ventilation are not well understood. As previously mentioned, bulk gas flow is not a major mechanism of gas exchange during HFV. Some gas exchange probably occurs simply because of nonhomogeneous alveolar filling and pressures.183 Other explanations include gas exchange resulting from turbulent mixing of gas molecules (“augmented dispersion”), gas convection, and diffusion.
Multiple mechanisms are probably involved in gas exchange during high-frequency ventilation.183 Ultimately, the effectiveness of these mechanisms must be determined by the evaluation of the patient's response to support.
Nursing Considerations During High Frequency Ventilation
High-frequency respiratory support is very different from conventional mechanical ventilation. The nurse must be familiar with the principles of operation, assessment of effectiveness of ventilation, and the potential complications of the technique.
The assessment of the infant or child on HFV differs from conventional ventilation in the following ways:
1. The clinical progress of the patient is the ultimate indicator of the effectiveness of ventilatory support. Progress is determined through evaluation of the patient's general appearance, color, and blood gases.
2. The chest will not rise during HFV, but may instead appear to be fluttering or vibrating, often referred to as “chest wiggle.”183
3. During auscultation of breath sounds, “inspiratory” air movement is difficult to identify; the quality of the breath sounds is peculiar to the patient on HFV. Breath sounds have been described as resembling a continuous loud jack hammer and are very high pitched. Low-pitched breath sounds may, in fact, indicate poor ventilation or pneumothorax.
4. Auscultation of the heart rate is nearly impossible; some physicians instruct the nurse to briefly place the HFV on standby to assess heart tones. Without the ability to easily auscultate heart tones or blood pressure, the nurse relies on evaluation of color, perfusion, pulses, pulse oximetry, and invasive monitoring for cardiovascular assessment.
5. Assessment of quantity and consistency of secretions obtained from suctioning is critical. Changes in the quantity or consistency of secretions frequently indicates the need for adjustment of the humidification system. A change in secretion quantity or consistency also may herald the development of necrotizing tracheobronchitis. Water particles should be visible traveling down the jet tube; these particles help prevent the development of mucous plugs.86
Complications of High-Frequency Ventilation
Many of the potential complications of HFV are identical to the complications of conventional mechanical ventilation, but the development of the complication may be more difficult to detect during HFV than during positive pressure ventilation.
1. Pneumothorax: The risk of pneumothorax in patients receiving HFV is the same as with conventional ventilation. Pneumothorax may be difficult to recognize during HFV because breath sounds are difficult to evaluate. Clinical signs of pneumothorax may be acute, including severe respiratory distress, cyanosis, hypoxemia, and hypotension. Transillumination and chest radiography are used to confirm the diagnosis.
2. Tenacious secretions: Secretions tend to become very thick, and mucous plugs may develop,86 producing airway obstruction. It may be difficult to achieve adequate humidification. Suctioning should be performed with instillation of saline.86
3. Gas trapping: Gas trapping often occurs with HFV and causes carbon dioxide retention and decreased compliance. Gas trapping is most likely to occur when high tidal volumes and short expiratory times are used.183 The optimal HFV settings to minimize air trapping have not been determined.
Endotracheal Tubes
Endotracheal (ET) intubation may be necessary to establish or maintain a patent airway or to facilitate mechanical ventilatory support. Elective intubation is always preferable to intubation under emergency conditions.
Endotracheal Tube Characteristics and Sizes
Shape of the Tube
Some ET tubes are curved sharply to enable rapid intubation to the point of curvature. These tubes should not be used for more than a few hours, because it is difficult to pass a suction catheter beyond the curvature of the tube. Tubes with sharp curvature are designed for orotracheal use, so they can be very difficult to place nasotracheally.
Position Markings
A radiopaque line should be present along the length of the ET tube to allow radiographic verification of the tube's position. In addition, markings should be present at 1-cm intervals on the tube. Such markings allow the nurse to verify appropriate depth of insertion regularly so that tube displacement is detected immediately.
The depth of the tube insertion at the patient lips or nares should be recorded in the patient record and nursing care plan. The nurse should check the depth of insertion whenever the tube is retaped, when vital signs are obtained or if the patient deteriorates suddenly.
Cuffed Versus Uncuffed Tubes
In the past, uncuffed ET tubes were generally used for infants and children less than 8 years of age, because the cricoid diameter of a child is quite narrow and was thought to provide a natural seal around the tube. However, evidence published in recent years has demonstrated that the use of cuffed tubes during in-hospital care of young children produces no higher incidence of complications than the use of uncuffed tubes.92
Cuffed tubes may reduce the incidence of aspiration, and cuffed tubes may be preferable to uncuffed tubes in some patients (i.e., those with poor lung compliance, high airway resistance, or a large glottic air leak), provided the endotracheal tube size, position and cuff inflation pressure is monitored.92 The cuff inflation pressure is maintained according to manufacturer specifications, typically at 20 to 25 cm H2O.92
Endotracheal Tube Size Selection
The diameter of the child's trachea is smallest at the level of the cricoid cartilage; therefore an ET tube may pass easily through the vocal cords yet be too large at the level of the cricoid cartilage. The ET tube size is appropriate if a small, audible air leak is present when inspiratory pressure of approximately 20- to 30-cm H2O is provided. This small leak indicates that the tube is probably small enough to avoid excessive pressure on the trachea below the level of the vocal cords.
If the tube is too large, an air leak is not detected even at positive pressure of more than 25 to 30 cm H2O. A tube that is too large can cause injury to the trachea.
If the tube is too small, an air leak is detectable at even low (< 10 to 15 cm H2O) inspiratory pressures. If the tube is too small and allows a substantial air leak, it may be impossible to provide adequate oxygenation or ventilation through the ET tube.92
The child's body length provides the best parameter for estimation of appropriate ET tube size.142 The relationship between body length and proper ET tube size has been used in the development of the Broselow resuscitation tape, which enables determination of appropriate endotracheal tube sizes, resuscitation equipment sizes, and drug dosages using the child's body length (see Figure 1-1).3,108
Several formulas enable estimation of ET tube size from age in children. The most popular formulas for estimation of uncuffed and cuffed tube sizes for children 1 to 10 years of age are:92
• Uncuffed ETT (internal diameter [I.D.] in mm) = (age/4) + 4
• Cuffed ETT (internal diameter [I.D.] in mm) = (age/4) + 3.5
These formulas provide a relatively accurate estimate of ET tube size (within 0.5 mm) in most children 1 to 10 years of age. Additional guidelines for estimation of proper ET tube size include the approximation of the size of the patient's little finger or the equivalent of the size of the child's nares.
Essential equipment for endotracheal intubation trays is listed in Table 21-12. Suggestions for intubation equipment size according to the child's age in years are listed in the table.
Table 21-12 Essential Equipment for Endotracheal Intubation
| Essential Equipment for Intubation | |
| Laryngoscope handle (2) | Suction catheter to fit endotracheal tube |
| Curved and straight laryngoscope blades | Tonsil suction (Yankauer tube) |
| Endotracheal tubes (three sizes) | Foam donut head rest |
| Stylet | Oropharyngeal airways |
| Bag and Mask | Magill forceps or Kolodny hemostats |
| Oxygen source | 1-inch tape |
| Suction set-up | Benzoin |
| Guidelines for Pediatric Endotracheal Tube Sizes | |
| Age/Size | mm Internal Diameter* |
| Premature newborn | |
| 1000 g | 2.5 |
| 1000-1500 g | 3.0 |
| 1500-2500 g | 3.5 |
| Full-term Newborns | 3.5-4.0 |
| 6-12 months | 4.0-4.5 |
| 1-2 years | 4.5 |
| 4 years | 5.0 |
| 6 years | 5.5 |
| 8 years | 6.0 |
| 10 years | 6.5 |
| Greater than 12 years | |
| Female | 6.5-8.0 |
| Male | 7.5-8.0 |
Note: Intubation should be attempted by an experienced or closely-supervised clinician with resuscitation equipment immediately available.
* An ET tube size 0.5 mm larger and one 0.5 mm size smaller than predicted size should be immediately available to accommodate unexpected anatomical deviations.
From Czervinske MP, Barnhart SL: Perinatal and pediatric respiratory care, St Louis, 2003, Saunders.
Resuscitation Bags for Hand Ventilation
A variety of manual resuscitator bags are available, each with distinctive features. In general, there are two main types of bags: the self-inflating bag and the non–self-inflating (gas-inflating or so-called “anesthesia”) bag. The self-inflating bag does not require gas flow to provide manual ventilation; the non-self-inflating bag does require gas flow for use.
Self-inflating Bags
Self-inflating bags may be used with or without an oxygen source. The natural recoil of the bag causes the bag to re-inflate after it is compressed, whether or not the bag is connected to an oxygen (or other gas) source.
If the bag is connected to an oxygen source and a reservoir bag, 100% oxygen can be delivered to the patient, because even when the bag recoils, only oxygen is drawn into the bag and administered to the patient. If oxygen is joined to the bag without a reservoir, when the bag recoils room air can be drawn into (entrained into) the bag so it mixes with the oxygen; as a result the patient receives a mix of room air and oxygen.
The 0.5-L bag is generally appropriate for ventilation of infants through preschool-age children; the 1-L bags are used for children up to 8 to 10 years of age. The larger (i.e., 1.5 L) bags may be used for adolescents and adults.183
Clinical Use
Self-inflating resuscitation bags are particularly useful for resuscitation carts because they can be used without oxygen flow, if needed. If used during resuscitation, delivery of a high concentration of oxygen is typically needed, so the bag should include a reservoir and should be attached to oxygen flow as soon as possible. Self-inflating bags require less skill to use than non-self inflating bags.
These bags are also useful for patient transport, when it is frequently impossible to predict how much air/oxygen to carry. If the oxygen source is exhausted, the self-inflating bags still enable effective ventilation of the patient with room air until additional oxygen is obtained.
When the bag is used for ventilation through an ET tube, a pressure gauge should be used, joined to the bag (with a Y connector if necessary). This enables monitoring of peak inspiratory pressures delivered.
Most bags have a pop-off valve to prevent delivery of high pressures. These valves are not precise and it may be necessary to inactivate the pop-off valve to deliver adequate bag-mask ventilation during emergencies.
The bags typically contain a valve near the adaptor where the bag can be joined to the ET tube or a mask. Unless a low-resistance valve is present, the valve opens only when the bag is squeezed (to deliver breaths to the patient) and it closes while the bag recoils and refills with air. An adjustable PEEP valve can be added to maintain positive end-expiratory pressure.
Advantages
The advantages of self-inflating bags include ease of operation (operation of valves is not needed to deliver gas flow) and ability to operate without an oxygen or gas source.
Disadvantages
There are several disadvantages to the use of self-inflating bags. A reservoir must be added to the bag to enable provision of a FiO2 greater than 0.60.
If an inspiratory pop-off valve is present in older bags (set at 40 mm Hg to prevent the delivery of high inflation pressures), the pop-off valve may prevent delivery of adequate tidal volume during bag-mask ventilation, particularly if there is low airway or lung compliance.
The operator must be familiar with appropriate manual ventilation technique. Because these bags have thicker walls than nonself-inflating bags, it is more difficult for the operator to assess the compliance of the patient's lungs during bag-mask or bag-tube ventilation. A quick, snapping motion on inspiration should be avoided because this technique creates excessive inspiratory pressure that will open the pop-off valve (if one is present) and result in loss of tidal volume (i.e., air flow delivered to the patient).
Gas flow does not occur during spontaneous patient inspiration unless a low-resistance valve is present in the bag to allow the patient to draw in gas from the bag between manually delivered breaths. In many models a valve between the mask-adapter and the bag is opened only by the force of bag compression, so gas flow (and delivery of supplementary oxygen through the bag) during spontaneous ventilation is impossible.
The volume delivered during manual ventilation may be variable and depends on the speed and force of bag compression (and recoil) and patient lung compliance. The oxygen concentration delivered to the patient can also vary widely; if a reservoir is attached to the bag an FiO2 of 1.00 (100% oxygen) may be reliably delivered.
Non–Self-inflating Bags
Non–self-inflating resuscitation bags are collapsed at rest. They inflate (and reinflate after each breath delivered) only if a continuous gas source is available. The gas flow to the bag must equal at least three to five times the patient's minute ventilation requirements to adequately fill the bag between breaths.
Clinical Use
The non-self-inflating bag (“anesthesia bag”) is used in patients with advanced airways; it is not used for mask ventilation. This bag is very useful for assisting respiratory efforts of spontaneously breathing patients, but can also be used for those with no spontaneous respiratory effort.
The operator can deliver a relatively consistent tidal volume to patients who have decreased lung compliance. If the patient is breathing spontaneously, the patient can receive gas flow from the bag between delivered breaths.
Advantages
There are many advantages of this type of resuscitation bag. A FiO2 of 1.0 can be provided without the addition of a reservoir bag. The bag and adaptor itself contain no internal valves that might dysfunction. During ventilation the provider can easily assess the patient's lung compliance because the walls of the bag are not thick and there are no valves between the bag and the patient's endotracheal tube/airway.
As noted previously, patients can breathe spontaneously and receive a continuous flow of oxygen even when the bag is not squeezed. A CPAP/PEEP valve (that provides resistance to exit of gas from the bag) and a pressure gauge easily can be added to the system.
Disadvantages
The use of this bag requires much greater skill than the use of the self-inflating bag. The use of the bag is best learned in a controlled environment. The operator must be able to monitor the patient and the patient's chest rise while ensuring that gas flow into and out of the bag is adequate.
The provider must be able to adjust gas flow into the bag so the bag will quickly reinflate between delivered breaths yet avoid delivery of high inspiratory pressure to the patient. If gas flow to the bag is insufficient, it will be impossible to deliver an adequate tidal volume and respiratory rate.
The risk of pneumothorax is significant when this bag is used by an inexperienced operator. If the gas flow to the bag is too high in light of the patient's minute ventilation, or if the PEEP valve is adjusted incorrectly, the bag may quickly become distended and ventilation may be delivered at a high pressure. Finally the bag requires a continuous flow of gas, which may be a problem during field resuscitations, such as transports.