Mosaic Skin Conditions
• A mosaic organism is composed of ≥2 genetically distinct cell populations derived from a homogeneous zygote.
– Genomic mosaicism results from alteration in the DNA sequence (affecting genes or chromosomes).
– Functional (epigenetic) mosaicism results from changes in gene expression (but not the DNA sequence) that are passed on during cellular replication; an important example is lyonization in female embryos, where random inactivation of one of the two X chromosomes occurs in each cell during early development.
• Clinical findings in mosaic skin conditions depend not only on the underlying genetic alteration but also on the timing of its origin (with earlier onset generally leading to more widespread involvement) and the cells or tissues affected (cutaneous ± extracutaneous).
– For example, mosaicism for an activating HRAS mutation can produce the combination of a nevus sebaceus (keratinocytes affected), speckled lentiginous nevus (melanocytes affected), and occasionally CNS abnormalities (nerve cells affected) in patients with phakomatosis pigmentokeratotica, a form of ‘twin spotting’.
• The accessibility of the skin allows visualization of mosaic patterns.
– Blaschko's lines are streaks and swirls that represent pathways of epidermal cell (e.g. keratinocyte or melanocyte) migration during embryonic development (Fig. 51.1).
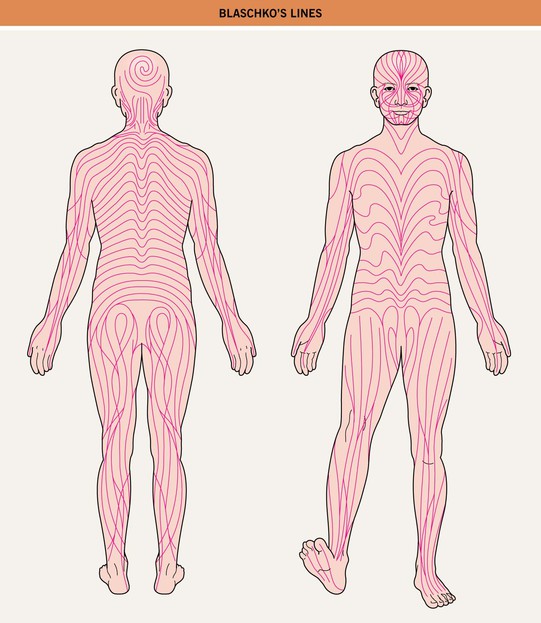
Fig. 51.1 Blaschko's lines. Note the S-shape of lines on the abdomen, the V-shape on the central back, perpendicular lines on the face, and swirls on the posterior scalp.
– Block-like, segmental, and dermatomal patterns can also reflect cutaneous mosaicism, typically involving melanocytes, mesodermal cells, and nerve cells, respectively.
• Types of cutaneous lesions that can follow Blaschko's lines or have a block-like/segmental pattern are outlined in Tables 51.1 and 51.2, respectively (Fig. 51.2).
Table 51.1
Skin findings that can occur along Blaschko's lines.
| Inflammation | |
| Examples | Clinical Clues |
| Lichen striatus | • Pink to hypopigmented flat-topped papules; common in children (see Chapter 9) |
| Linear lichen planus (Fig. 51.2A) | • Violaceous flat-topped papules with Wickham's striae; often in adults |
| ‘Blaschkitis’ | • Pruritic erythematous papulovesicles in multiple streaks on the trunk; usually develops in adults and often recurs |
| Inflammatory linear verrucous epidermal nevus (ILVEN) (Fig. 51.2B) | • Scaly, erythematous psoriasiform plaques with prominent pruritus and lack of response to therapy; onset usually by childhood |
| Linear psoriasis (Fig. 51.2C) | • Psoriasiform plaques, often responding to therapy; ± psoriasis elsewhere |
Other conditions resembling lichen planus: linear GVHD, lupus erythematous > dermatomyositis, drug eruptions Other conditions with a keratotic and/or vesiculobullous component: • Variable onset: linear Darier (Fig. 51.2D) and Hailey–Hailey (Fig. 51.2E) diseases • Early onset: linear porokeratosis (see Fig. 89.7E), PEODDN, IP* stages 1–2 (see Figs. 28.9 and 51.7A), epidermolytic epidermal nevus*, Conradi–Hünermann–Happle syndrome* (see Fig. 46.13) | |
| Verrucous lesions – e.g. epidermal/sebaceous nevi (Table 51.3; Fig. 51.3), IP stage 2 (Fig. 51.7A,B) | |
| Spines/comedones – e.g. nevus comedonicus (Table 51.3), PEODDN (favors palms/soles), linear lichen planopilaris (later onset; see Fig. 9.4H) | |
| Hypopigmentation (see Table 54.3) | |
| Hyperpigmentation (see Table 55.4) | |
| Hairlessness – e.g. X-linked hypohidrotic ectodermal dysplasia (female ‘carriers’; Fig. 51.6), Goltz syndrome, IP stage 4 (Fig. 51.7C) | |
| Atrophy – e.g. linear lichen sclerosus (epidermal wrinkling), linear atrophoderma of Moulin (hyperpigmented and depressed), Goltz syndrome (Fig. 51.8A,B), IP stage 4, Conradi–Hünermann–Happle syndrome (follicular atrophoderma) | |
| Papulonodular lesions** – e.g. adnexal neoplasms (e.g. trichoepitheliomas), BCCs, basaloid follicular hamartomas | |
* Inflammatory manifestations occur primarily during infancy.
** In addition to conditions with inflammatory or verrucous papulonodules noted above.
GVHD, graft-versus-host disease; IP, incontinentia pigmenti; PEODDN, porokeratotic eccrine ostial and dermal duct nevus.
Table 51.2
Skin findings that can have a block-like or segmental pattern that reflects mosaicism.
Hypopigmentation – e.g. nevus depigmentosus, segmental vitiligo (see Chapter 54)
Hyperpigmention ± hypertrichosis – e.g. CALM, Becker's nevus/smooth muscle hamartoma (see Table 50.3 and Chapter 55)
Hypertrichosis – e.g. X-linked congenital generalized hypertrichosis (female ‘carriers’)
Vascular lesions – e.g. port wine stain,† CMTC, unilateral nevoid telangiectasia, venous malformation,* plaque-type glomuvenous malformation,** segmental infantile hemangioma (see Chapter 85)
Papulonodular lesions – e.g. segmental leiomyomas** or neurofibromas**
† Associated with mosaic activating mutations in the GNAQ gene encoding the Q-class G protein α-subunit; mutations in this gene have also been associated with dermal melanocytosis, which occurs as ‘twin spots’ with port wine stains in phakomatosis pigmentovascularis.
CALM, café-au-lait macule; CMTC, cutis marmorata telangiectatica congenita.
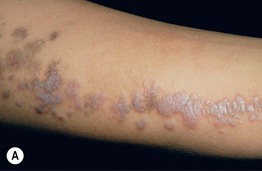
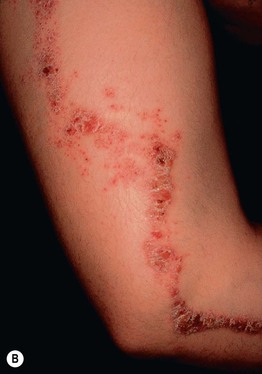
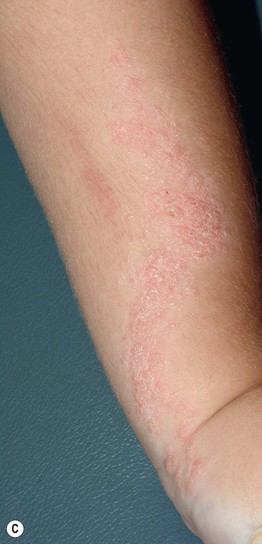
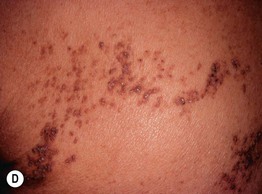
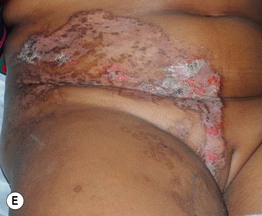
Fig. 51.2 Inflammatory lesions along Blaschko's lines. A Linear lichen planus presenting as a band of coalescing violaceous papules and plaques with Wickham's striae on an extremity. Note the postinflammatory hyperpigmentation proximally. B Inflammatory linear verrucous epidermal nevus (ILVEN) featuring persistent, extremely pruritic, scaly psoriasiform plaques. C Linear psoriasis with erythema and scale that responded to treatment with a high-potency topical CS. D Linear Darier disease presenting with keratotic papules in an adult, representing type 1 mosaicism (see Fig. 51.5A). E Linear Hailey–Hailey disease presenting with recurrent blistering and erosions in a young girl, representing type 2 mosaicism with earlier and more severe involvement in the affected region due to a ‘second hit’ mutation (see Fig. 51.5B). A, Courtesy, Joyce Rico, MD; C, E, Courtesy, Julie V. Schaffer, MD.
• In chimerism, different cell populations reflect a genetically heterogeneous zygote (e.g. fusion of two zygotes or fertilization of one egg by two sperm); this may manifest with a block-like, linear, or irregular pattern of pigmentary variation.
Epidermal Nevi and ‘Epidermal Nevus Syndromes’
• Epidermal nevi (see Chapter 89) present as streaks and swirls of thickened (e.g. verrucous, hyperkeratotic, or velvety) skin along Blaschko's lines, usually with hyperpigmentation and sometimes with adnexal involvement (e.g. in a nevus sebaceus or nevus comedonicus) (Fig. 51.3).
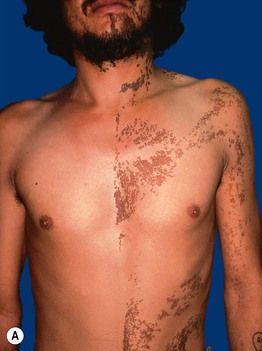
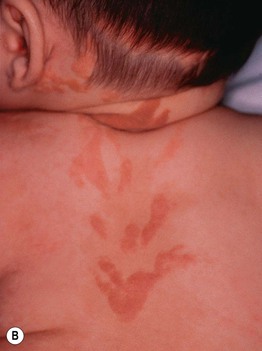
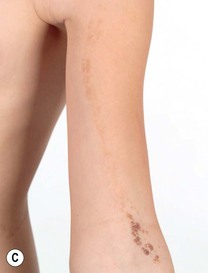
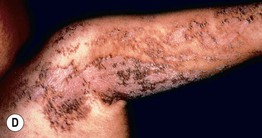
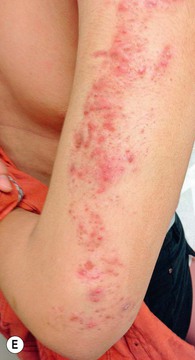
Fig. 51.3 Epidermal nevi. A Extensive lesion with especially verrucous areas on the neck. B V-shaped pattern on the mid back. C Lesion due to a FGFR3 mutation, representing a mosaic counterpart of the acanthosis nigricans in thanatophoric dysplasia, an autosomal dominant neuroskeletal syndrome. This explains the flexural accentuation of the epidermal nevus. D Epidermolytic epidermal nevus. Note the shedding of scale and associated hypopigmentation. E Nevus comedonicus with inflammatory papulonodules as well as comedones in an adolescent. The lesion initially presented as a congenital hypopigmented streak. C, Courtesy, Celia Moss, MD; E, Courtesy, Julie V. Schaffer, MD.
• The heterogeneous genetic etiologies and potential systemic associations (‘epidermal nevus syndromes’) of various types of epidermal nevi are presented in Table 51.3 (Fig. 51.4).
Table 51.3
Variants of epidermal nevi (EN) and possible associated findings.
* The underlying mosaic mutations are generally not compatible with life if present in all the cells of the body.
** Usually but not always different (e.g. more severe) mutations within the EN than in the AD disorder.
AD, autosomal dominant; CLOVES, congenital lipomatous overgrowth with vascular, epidermal, and skeletal anomalies; FGFR, fibroblast growth factor receptor; ILVEN, inflammatory linear verrucous epidermal nevus; KRT, keratin; M-CM, megalencephaly (macrocephaly)–capillary malformation syndrome; PIK3CA, phosphoinositide-3-kinase, catalytic α-polypeptide; SLN, speckled lentiginous nevus.
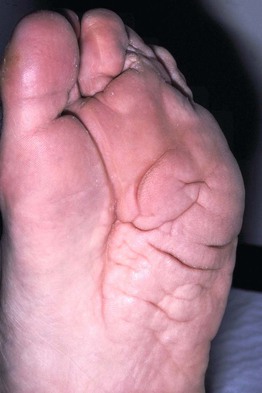
Fig. 51.4 Plantar cerebriform connective tissue nevus in a man with Proteus syndrome. Courtesy, Odie Enjolras, MD.
Mosaicism in Autosomal Dominant Skin Conditions
• When evaluating a patient with skin lesions in a mosaic pattern, it can be helpful to consider whether the findings would resemble an autosomal dominant genodermatosis if present in a more generalized distribution.
• Type 1, type 2, and revertant forms of mosaicism occurring in autosomal dominant skin disorders are summarized in Fig. 51.5.
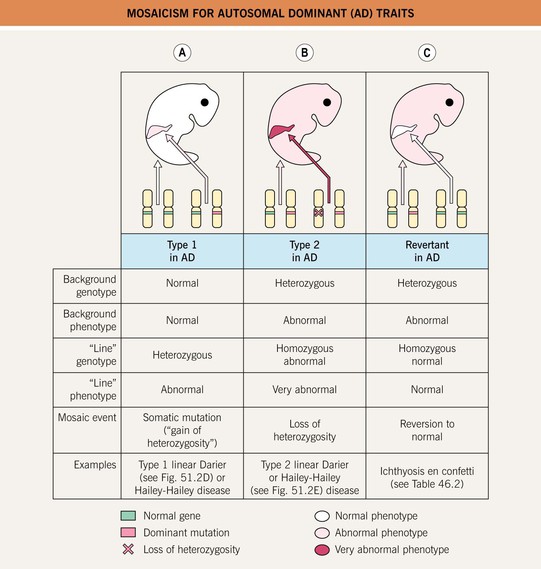
Fig. 51.5 Mosaicism for autosomal dominant (AD) traits. A Type 1 mosaicism features a linear distribution of the skin lesions that characterize an AD genodermatosis in the absence of involvement elsewhere on the body. This results from a postzygotic heterozygous mutation in the underlying gene that is only present in the affected region. B Type 2 mosaicism presents with a more severe linear lesion superimposed on the background of a generalized AD genodermatosis. This reflects a constitutional (in all the cells of the body) heterozygous mutation plus a ‘second hit’ in the other allele of the gene within the more severely affected region. C In revertant mosaicism, an area of normal-appearing skin occurs in the background of a generalized AD genodermatosis due to a reversion to normal or other correction of the underlying heterozygous mutation in that region.
Mosaicism in X-Linked Conditions
• In X-linked disorders, functional mosaicism due to lyonization occurs in female patients and carriers.
• X-linked dominant conditions are typically lethal in male embryos and are therefore seen almost exclusively in a mosaic pattern in female patients with a heterozygous mutation; male patients occasionally survive due to underlying mosaicism, e.g. in the setting of Klinefelter syndrome (47,XXY karyotype) or a postzygotic mutation.
• In conditions traditionally referred to as X-linked recessive, male patients have generalized disease and female patients or ‘carriers’ are affected to a variable (usually lesser) degree, often in a mosaic pattern; an example is hypohidrosis and hyperpigmentation following Blaschko's lines in female ‘carriers’ of X-linked hypohidrotic ectodermal dysplasia (Fig. 51.6).
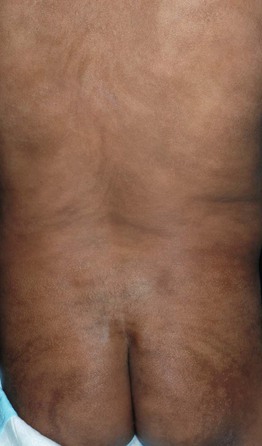
Fig. 51.6 Female ‘carrier’ of X-linked hypohidrotic ectodermal dysplasia. The skin within the hyperpigmented streaks and swirls following Blaschko's lines is extremely smooth. Starch-iodide testing on the back disclosed a patchy distribution of active sweat glands. This 2-year-old girl also had whorled areas of sparse hair on the posterior scalp and several cone-shaped teeth. Courtesy, Julie V. Schaffer, MD.
Incontinentia Pigmenti (IP)
• X-linked dominant disorder caused by mutations in the NF-κB essential modulator (NEMO) gene, which encodes a protein that protects against TNF-α-induced apoptosis; male patients with milder NEMO mutations may present with hypohidrotic ectodermal dysplasia with immune deficiency (see Table 52.5).
• IP has four stages, which may be absent or overlapping, of cutaneous findings that follow Blaschko's lines (with the exception of stage 4) (Fig. 51.7; see Fig. 28.9):
– Vesicular streaks favoring the extremities and scalp in neonates, often associated with peripheral blood leukocytosis and eosinophilia; may recur during childhood febrile illnesses.
– Verrucous linear plaques favoring the extremities in infants; acral keratotic nodules sometimes develop after puberty.
– Hyperpigmented grayish-brown streaks and swirls favoring the trunk and intertriginous areas from infancy through adolescence.
– Hypopigmented/atrophic ‘Chinese character’-like bands lacking hair and sweat glands, favoring the calves in adolescents and adults.
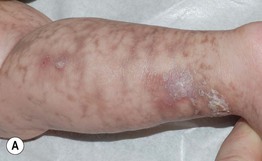
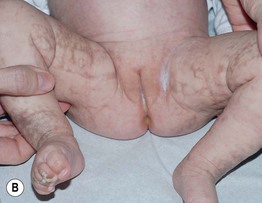
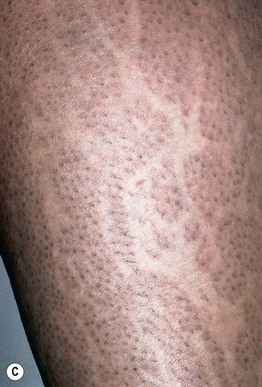
Fig. 51.7 Incontinentia pigmenti. A, B Stages 2 (verrucous) and 3 (hyperpigmented). Note the erythematous background of the keratotic papules and plaques on the leg (A) and the remaining keratotic lesions on the toes (B). The scalloped edges of the hyperpigmented streaks reflect growth of normal keratinocytes into areas of apoptosis. C Stage 4 (atrophic and hypopigmented). Note the absence of hairs within the streaks on the calf. A and B, Courtesy, Julie V. Schaffer, MD.
• Other manifestations can include alopecia (often at the vertex in a swirled pattern), missing or conical teeth, retinal vascular anomalies (requires monitoring with ophthalmologic examinations during infancy), and CNS abnormalities (e.g. seizures, developmental delay).
Goltz Syndrome (Focal Dermal Hypoplasia)
• X-linked dominant condition caused by mutations in the PORCN gene, which encodes a protein that regulates Wnt signaling.
• Skin lesions following Blaschko's lines are characterized by vermiculate dermal atrophy, outpouchings of fat, telangiectasias, and hypo- > hyperpigmentation (Fig. 51.8).
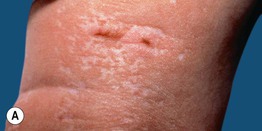
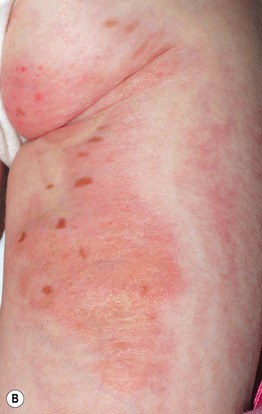
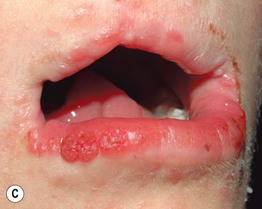
Fig. 51.8 Goltz syndrome. A Hypopigmented streaks of vermiculate atrophy and fat ‘herniation.’ B Bands of telangiectatic erythema, dermal atrophy with fat ‘herniation,’ and hyperpigmented macules on the posterior thigh. C Raspberry-like papules on the lower lip. Similar lesions can occur in the anogenital region and may be confused with warts. B, C, Courtesy, Julie V. Schaffer, MD.
• Additional features include periorificial raspberry-like papillomas, dystrophic nails, sparse hair, abnormal teeth, split hand/foot (‘lobster claw’) malformations, ocular abnormalities (e.g. microphthalmia), and the radiographic finding of osteopathia striata in long bones.
X-Linked Dominant Ichthyosiform Conditions
• Examples include congenital hemidysplasia with ichthyosiform nevus and limb defects (CHILD) and Conradi–Hünermann–Happle syndromes (see Table 46.2).
Lethal Disorders Rescued by Mosaicism
• Some mutations in autosomal genes are only observed in a mosaic state, as they would not be compatible with life if present in all the cells of the body.
• Examples include Proteus and Schimmelpenning syndromes presenting with epidermal and sebaceous nevi (see Table 51.3), Sturge–Weber syndrome (see Table 51.2 and Chapter 85), and forms of ‘pigmentary mosaicism’ including McCune–Albright syndrome (see Table 50.3 and Chapters 54 and 55).
Mosaic Manifestations of Acquired Skin Conditions
• Multifactorial skin disorders with environmental as well as genetic components occasionally occur along Blaschko's lines or in a segmental distribution, presumably reflecting mosaicism for a ‘susceptibility’ mutation; as a result, such conditions often develop in older children or adults following an environmental trigger, and they are sometimes superimposed on a pre-existing mosaic lesion (e.g. psoriasis within an epidermal nevus).
• Examples include linear lichen planus, linear GVHD, and segmental vitiligo.
For further information see Ch. 62. From Dermatology, Third Edition.