30 HAEMOPTYSIS: WHAT TO LOOK FOR
| Haemoptysis: | Coughing up blood or blood stained sputum. |
|---|---|
| Massive haemoptysis: | Definitions vary1,2. Typically — coughing up more than 200 ml blood in 24 hours (approximately one cup). |
| Mild haemoptysis: | Some blood in the sputum…often spotting or a few ml only. |
| Pseudohaemoptysis: | Spitting up blood that has not originated from the lower respiratory tract. |
Some patients referred from primary care with suspected haemoptysis have had a pseudohaemoptysis. A patient will often have difficulty in distinguishing between blood resulting from epistaxis, gingivitis, gastrointestinal haemorrhage…and blood originating from the lower respiratory tract.
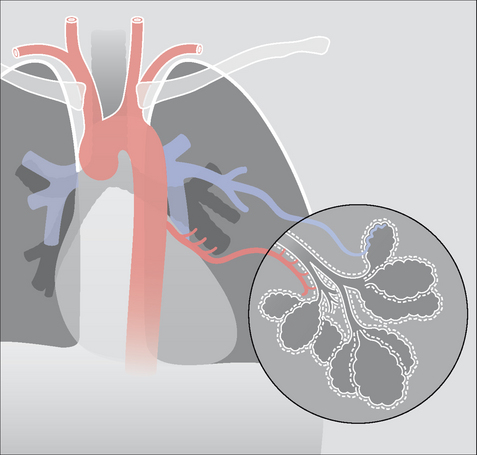
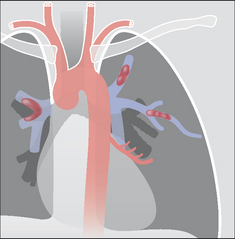
RELEVANT ARTERIAL ANATOMY3-5
An understanding of the dual blood supply to the lungs explains the differing origins of mild and massive haemoptysis (Table 30.1).
Table 30.1 Importance of the arterial supply to the lung.
| Artery | Capillary contact | Clinical relevance |
|---|---|---|
| Pulmonary | ||
| Bronchial |
The lung has two distinctive and separate blood supplies (Fig. 30.1):
 High pressure bronchial arteries arising from the aorta. Their sites of origin can vary—usually from the proximal descending aorta. Typically, two bronchial arteries supply the left lung and a single bronchial artery supplies the right lung. These arteries feed the bronchial walls up to and including the terminal bronchioles. They also supply the connective tissue of the lung as well as the visceral pleural membrane.
High pressure bronchial arteries arising from the aorta. Their sites of origin can vary—usually from the proximal descending aorta. Typically, two bronchial arteries supply the left lung and a single bronchial artery supplies the right lung. These arteries feed the bronchial walls up to and including the terminal bronchioles. They also supply the connective tissue of the lung as well as the visceral pleural membrane.CAUSES OF HAEMOPTYSIS1-3,5-11
The patient who coughs up blood has one overwhelming fear—that he has cancer. Knowledge of the numerous causes of haemoptysis—and the frequency of their occurrence—is essential (Table 30.2).
Table 30.2 Causes of haemoptysis1–3,5–11.
| Cause | Notes | |
|---|---|---|
| The haemoptysis is usually mild and the expectorate is often described as pink and frothy | ||
| Rare. Presents in childhood | ||
| Lung contusion or penetrating injury | ||
| Goodpasture’s syndrome | ||
| Monthly. A very rare cause in young women | ||
| Figures vary. As many as 20% of all patients presenting with haemoptysis6 | ||
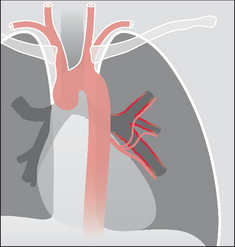
Figure 30.2 Haemoptysis due to acute bronchitis. The bronchial arteries arise from the aorta and nourish the walls of the inflamed bronchi. Inflammation around and involving the bronchial arterial capillaries can cause a mild haemoptysis.
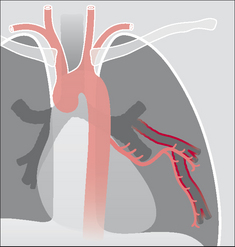
Figure 30.3 Haemoptysis due to bronchiectasis. The chronic inflammation can cause localised proliferation of the bronchial arteries; these vessels may become friable and cause a mild haemoptysis. Sometimes the bronchial arteries are actually eroded by the inflammatory process and a massive haemoptysis results.
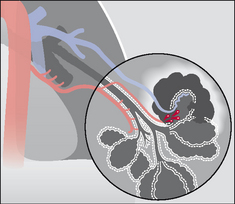
Figure 30.4 Haemoptysis due to pulmonary tuberculosis. The high pressure bronchial arteries can be eroded by the tuberculous inflammation. In this example a cavity has involved a bronchial artery, and a potentially massive haemoptysis could result. Erosion of low pressure pulmonary arteries or capillaries may also occur in chronic tuberculosis.
CLINICAL INVESTIGATION/MANAGEMENT
(1) MASSIVE HAEMOPTYSIS: RARE BUT AN EMERGENCY
Massive haemoptysis is arbitrarily defined as coughing up more than a cup of blood (200 ml) in 24 hours. This is a medical emergency and necessitates immediate hospital admission and in-patient investigation and treatment.
 The causes of massive haemoptysis are shown in Table 30.3. High pressure (i.e. aortic pressure) vessels are eroded. Severe haemorrhage results.
The causes of massive haemoptysis are shown in Table 30.3. High pressure (i.e. aortic pressure) vessels are eroded. Severe haemorrhage results. Usually, the pathology involves the bronchial artery circulation. Asphyxiation by blood is a serious risk.
Usually, the pathology involves the bronchial artery circulation. Asphyxiation by blood is a serious risk.Table 30.3 Causes of massive haemoptysis1.
| Common | Uncommon |
|---|---|
(2) MILD HAEMOPTYSIS: MUCH MORE COMMON
Mild haemoptysis is defined as some blood in the sputum…generally a little spotting or a few millilitres only.
 The most frequent causes are acute bronchitis, pneumonia, bronchial carcinoma, bronchiectasis, and pulmonary embolism.
The most frequent causes are acute bronchitis, pneumonia, bronchial carcinoma, bronchiectasis, and pulmonary embolism. The CXR (frontal and lateral projections obtained as a pair) can have an important impact on the further management of the individual patient.
The CXR (frontal and lateral projections obtained as a pair) can have an important impact on the further management of the individual patient.The vast majority of patients presenting with a mild haemoptysis and who have normal frontal—and lateral—CXRs have benign disease. A management algorithm can be constructed based on the CXR findings (Table 30.4).
Table 30.4 Investigation/management of mild haemoptysis.
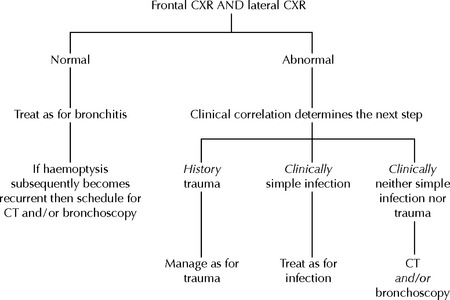 |
THE CXR: ABNORMAL AND NORMAL
CXR ABNORMAL (1) PROBABLE CAUSE SUGGESTED
Examples (Figs 30.6-30.9) include pneumonia, pulmonary oedema (heart failure), bronchial carcinoma, metastatic lung disease, bronchiectasis and traumatic pulmonary contusion.
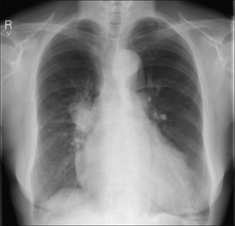
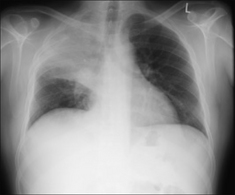
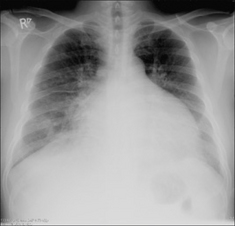
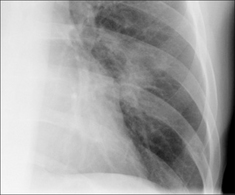
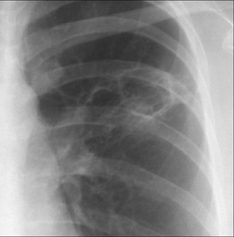
Figure 30.7 Haemoptysis due to a bronchial carcinoma. A central tumour at the right hilum. In most instances of haemoptysis resulting from a lung carcinoma the bleeding is mild and caused by the tumour eroding small vessels.
CXR ABNORMAL (2) POSSIBLE CAUSE SUGGESTED
Examples (Figs 30.10-30.13):
 Solitary pulmonary nodule (SPN) suggests bronchial carcinoma, or metastasis, or a rarity such as Wegener’s granulomatosis.
Solitary pulmonary nodule (SPN) suggests bronchial carcinoma, or metastasis, or a rarity such as Wegener’s granulomatosis.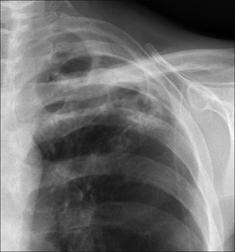
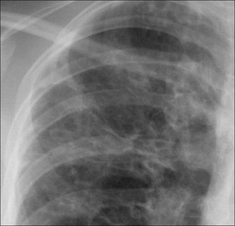
Figure 30.10 Haemoptysis due to post-primary tuberculosis at the left apex. The chronic inflammation has caused erosion of friable vessels. The bronchial arterial or the pulmonary arterial circulation may be affected.
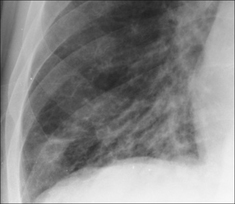
Figure 30.11 Mild haemoptysis due to bronchiectasis. The crowding of vessels and the prominent tramlines (thickened bronchial walls) at the right base raised the suggestion of bronchiectasis. Bronchiectasis was subsequently confirmed on CT.
CXR NORMAL—IMPLICATIONS AND USEFULNESS
 In most patients presenting with a mild haemoptysis and no other symptoms a normal CXR is good news. Remember that a lateral CXR should always be part of the haemoptysis CXR investigation protocol.
In most patients presenting with a mild haemoptysis and no other symptoms a normal CXR is good news. Remember that a lateral CXR should always be part of the haemoptysis CXR investigation protocol. Any symptom or sign that could be attributable to a pulmonary embolus (PE) means that this must be the default diagnosis—even if the frontal and lateral CXRs appear normal (see Chapter 23).
Any symptom or sign that could be attributable to a pulmonary embolus (PE) means that this must be the default diagnosis—even if the frontal and lateral CXRs appear normal (see Chapter 23). A massive haemoptysis—200 ml or more in 24 hours—is most commonly due to an inflammatory condition eroding bronchial artery vessels.
A massive haemoptysis—200 ml or more in 24 hours—is most commonly due to an inflammatory condition eroding bronchial artery vessels. Mortality from a massive haemoptysis can be as high as 50%5. Death results from asphyxiation because the airways are flooded with blood.
Mortality from a massive haemoptysis can be as high as 50%5. Death results from asphyxiation because the airways are flooded with blood. Despite thorough investigation the cause of haemoptysis remains unexplained in a significant number of patients. Figures vary between different series, but may be as many as 20–30% of cases6,11,12.
Despite thorough investigation the cause of haemoptysis remains unexplained in a significant number of patients. Figures vary between different series, but may be as many as 20–30% of cases6,11,12. Persistent haemoptysis and a normal CXR. Approximately 5% of these patients will have a bronchial carcinoma13.
Persistent haemoptysis and a normal CXR. Approximately 5% of these patients will have a bronchial carcinoma13.1. Johnson JL. Manifestations of hemoptysis. How to manage minor, moderate and massive bleeding. Postgrad Med. 2002;112:101-113.
2. Thompson AB, Teschler H, Rennard SI. Pathogenesis, evaluation, and therapy for massive haemoptysis. Clin Chest Med. 1992;13:69-82.
3. Fraser RG, Muller NL, Colman NC, Pare PD. Fraser and Pare’s Diagnosis of Diseases of the Chest, 4th ed. Philadelphia, PA: WB Saunders, 1999.
4. Ryan S, McNicholas M, Eustace S. Anatomy for Diagnostic Imaging, 2nd ed. Philadelphia, PA: WB Saunders, 2004.
5. Marshall TJ, Flower CD, Jackson JE. The role of radiology in the investigation and management of patients with haemoptysis. Clin Radiol. 1996;51:391-400.
6. Santiago S, Tobias J, Williams AJ. A reappraisal of the causes of hemoptysis. Arch Intern Med. 1991;151:2449-2451.
7. Goldman JM. Hemoptysis: Emergency assessment and management. Emerg Med Clin North Am. 1989;7:325-338.
8. Reisz G, Stevens D, Boutwell C, et al. The causes of hemoptysis revisited: a review of the etiologies of hemoptysis between 1986 and 1995. Mo Med. 1997;94:633-635.
9. Gudbjerg CE. Roentgenologic diagnosis of bronchiectasis. An analysis of 112 cases. Acta Radiol. 1955;43:210-226.
10. Corder R. Hemoptysis. Emerg Med Clin North Am. 2003;21:421-435.
11. Bidwell JL, Plachner RW. Hemoptysis: diagnosis and management. Am Fam Physician. 2005;72:1253-1260.
12. Andersen PE. Imaging and interventional radiological treatment of hemoptysis. Acta Radiol. 2006;47:780-792.
13. Lederle FA, Nichol KL, Perenti CM. Bronchoscopy to evaluate hemoptysis in older men with nonsuspicious chest roentgenograms. Chest. 1989;95:1043-1047.
14. Worsley DF, Alavi A, Aronchick JM, et al. Chest radiographic findings in patients with acute pulmonary embolism: observations from the PIOPED study. Radiology. 1993;189:133-136.