Treatment of Localized Renal Cell Carcinoma
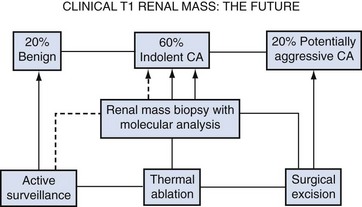
Localized renal masses, clinical stages T1 and T2, have increased in incidence owing to more widespread use of cross-sectional imaging and now represent a relatively common clinical scenario (Lipworth et al, 2006; Jemal et al, 2009). Our perspectives about clinical T1 renal masses have changed substantially in the past 2 decades. Previously, all were presumed to be malignant and managed aggressively, most often with radical nephrectomy. We now recognize great heterogeneity in the tumor biology of these lesions and multiple management strategies are now available, including radical nephrectomy (RN), partial nephrectomy (PN), thermal ablation (TA), and active surveillance (AS) (Kunkle et al; 2008; Campbell et al, 2009) (Fig. 49–18). Concepts that were once controversial, such as elective PN, are now accepted as standards of care (Kunkle et al, 2008; Campbell et al, 2009). A greater understanding of the tumor biology and appreciation of the deleterious functional consequences of RN has stimulated a reassessment of this field (Nguyen et al, 2008a; Russo and Huang, 2008; Campbell et al, 2009).
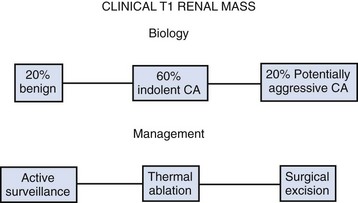
Figure 49–18 Clinical T1 renal masses are heterogeneous, with 20% benign and only about 20% exhibiting potentially aggressive features. Management options have expanded greatly, ranging from radical nephrectomy, the previous standard, to active surveillance. CA, cryoablation.
Overall, about 20% of solid, enhancing, clinical T1 renal masses are benign, most often oncocytomas or atypical AMLs, although the incidence of benign pathology can vary greatly in different subpopulations (Frank et al, 2003b; Remzi et al, 2006; Snyder et al, 2006; Pahernik et al, 2007; Kutikov et al, 2008a; Russo and Huang, 2008; Campbell et al, 2009). Young to middle-aged women, in particular, are more likely to have benign pathology, as high as 40% in some series (Eggener et al, 2004; Lane et al, 2007a). One potential explanation is that some benign renal masses, such as cystic nephroma and atypical AML, may be influenced by the hormonal milieu and are thus more common in this patient population. In contrast, the proportion of benign tumors appears to increase gradually in males as they age (Lane et al, 2007a). An even more important determinant of benign pathology is tumor size, with multiple studies confirming this (Lane et al, 2007a; Campbell et al, 2009). Frank and colleagues (2003b) have demonstrated a direct relationship between tumor size and the incidence of malignancy. In their series 30% of tumors less than 2 cm were benign, compared with 22% of tumors between 2 and 3 cm and with 20% between 3 and 4 cm. Only 9.5% of clinical T1b tumors were benign. Algorithms to predict benign versus malignant pathology for patients with clinical T1 renal masses are available and have demonstrated modest accuracy (Lane et al, 2007a; Campbell et al, 2009).
Tumor size has also correlated with biologic aggressiveness for clinical T1 renal masses, as reflected by high tumor grade, locally invasive phenotype, or adverse histologic subtype. In the study by Frank and colleagues (2003b), such adverse findings were uncommon in tumors less than 4 cm diameter. In this subset only 1.7% demonstrated invasion of the perinephric fat, 0.7% had venous involvement, 0.6% had lymph node involvement, and only 15% were high grade. Such features were more commonly observed in clinical T1b tumors in this series. Other studies suggest a cut point at 3 cm, with tumors larger than this much more likely to exhibit potentially aggressive histopathologic features (Remzi et al, 2006; Pahernik et al, 2007). Surveillance studies confirm a slow growth rate and low risk of metastasis for many small renal tumors (Bosniak et al, 1995; Kassouf et al, 2004; Kato et al, 2004; Chawla et al, 2006; Kouba et al, 2007; Kunkle et al, 2007a, 2008; Lamb et al, 2004; Sowery and Siemens, 2004; Volpe et al, 2004; Wehle et al, 2004; Abouassaly et al, 2008; Crispen et al, 2009). Other clinical factors such as patient age and sex, symptomatic presentation, and smoking history have also been studied, although none of these factors can provide substantial predictive value with respect to tumor aggressiveness (Lane et al, 2007a). Current algorithms incorporating clinical and radiographic factors to predict tumor aggressiveness are very limited in their accuracy, with concordance indices less than 0.60, and not much better than a coin flip (Lane et al, 2007a). Conventional renal mass biopsy can substantially improve on this, having demonstrated reasonable accuracy for assessment of tumor grade and histology and should be considered in patients who are candidates for a wide range of management strategies (Herts and Baker, 1995; Campbell et al, 1997a; Brierly et al, 2000; Dechet et al, 2003; Volpe et al, 2004, 2007; Lebret et al, 2007; Somani et al, 2007; Kummerlin et al, 2008a; Lane et al, 2008b; Schmidbauer et al, 2008). Younger, healthy patients who are unwilling to accept the uncertainty associated with renal mass biopsy and older, frail patients who will be managed conservatively independent of biopsy results should be managed without a biopsy.
Radical Nephrectomy
Notwithstanding advances in our understanding of the genetics and biology of RCC, surgery remains the mainstay for curative treatment of this disease. The objective of surgical therapy is to excise all tumor with an adequate surgical margin. Simple nephrectomy was practiced for many decades but was supplanted by RN when Robson and colleagues (1969) established this procedure as the “gold standard” curative operation for localized RCC with their report of 66% and 64% overall survival for stage I and II tumors, respectively. These results demonstrated improved survival rates compared with those of patients treated by pericapsular nephrectomy. RN is still a preferred option for many patients with localized RCC, such as those with very large tumors (most clinical T2 tumors) or the relatively limited subgroup of patients with clinical T1 tumors that are not amenable to nephron-sparing approaches (Nguyen et al, 2008a). RN has more recently fallen out of favor for small renal tumors due to concerns about CKD, and should only be performed when necessary (Nakada, 2005; Russo and Huang, 2008; Nguyen et al, 2008a; Campbell et al, 2009).
The main concern with RN is that it predisposes to CKD, which in turn is associated with morbid cardiovascular events and increased mortality rates. Several studies have shown an increased risk of CKD on longitudinal follow-up after RN, including a landmark study from Memorial Sloan-Kettering Cancer Center that looked at 662 patients with a small solitary tumor, a normal opposite kidney, and a “normal” serum creatinine level—essentially patients who would be considered for elective PN (Huang et al, 2006; Russo and Huang, 2008). The first major finding was that 26% of this patient population had preexisting grade 3 CKD (estimated glomerular filtration rate [eGFR] < 60 mL/min/1.73 m2), demonstrating that this patient population is substantially different than the kidney transplant donor population that is often considered analogous. In reality, the donor population is not comparable because it is carefully screened to exclude CKD and related comorbidities. The second major finding was that the incidence of grade 3 CKD (eGFR < 60 mL/min/1.73 m2) was much more common after RN than PN, 65% versus 20%, respectively (P < .001). More severe CKD (eGFR < 45 mL/min/1.73 m2) was also much more common after RN than PN, 36% versus 5%, respectively (P < .001).
Several studies illustrate the negative implications of CKD, including a population-based study that followed more than a million patients for 2.8 years and reported increased rates of cardiovascular events and death as the degree of CKD worsened even after controlling for hypertension, diabetes, and other potential confounding factors (Go et al, 2004). The relative risks of cardiovascular events were 1.4, 2.0, 2.8, and 3.4 for eGFR (mL/min/1.73 m2) of 45 to 60, 30 to 45, 15 to 30, and less than 15, respectively. Relative death rates were 1.2, 1.8, 3.2, and 5.9 for these same subgroups, respectively. These data highlight the need to optimize renal function and underscore nephron-sparing procedures as an overriding principle in the management of clinical T1 renal masses; RN should only be performed when necessary (Miller et al, 2008; Russo and Huang, 2008; Thompson et al, 2008; Campbell et al, 2009; Huang et al, 2009; Lane et al, 2009).
The prototypical concept of RN encompasses the basic principles of early ligation of the renal artery and vein, removal of the kidney with primary dissection external to the Gerota fascia, excision of the ipsilateral adrenal gland, and performance of a complete regional lymphadenectomy from the crus of the diaphragm to the aortic bifurcation (O’Malley et al, 2009a). Controversy has arisen regarding the need for many of these practices on a routine basis (Lam et al, 2004b; O’Malley et al, 2009a). Performance of a perifascial nephrectomy is of undoubted importance during RN for preventing postoperative local tumor recurrence because approximately 25% of clinical T2 RCCs manifest perinephric fat involvement (Lam et al, 2007; Thompson et al, 2007a). Preliminary renal arterial ligation remains an accepted practice; however, in large tumors with abundant collateral vascular supply, it is not always possible to achieve complete preliminary control of the arterial circulation (O’Malley et al, 2009a). It has been well demonstrated that removal of the ipsilateral adrenal gland is not routinely necessary in the absence of radiographic adrenal enlargement unless the malignant lesion extensively involves the kidney, is locally advanced, or is located in the upper portion of the kidney immediately adjacent to the adrenal gland (Sagalowsky et al, 1994; Leibovich et al, 1995; Kletscher et al, 1996; Kozak et al, 1996; Sandock et al, 1997; Tsui et al, 2000a; Paul et al, 2001a; Lam et al, 2004b; Siemer et al, 2004; O’Malley et al, 2009a; Lane et al, 2009).
The need for a complete regional lymphadenectomy in all patients undergoing RN also remains controversial, and a randomized study of lymphadenectomy versus controls at the time of renal surgery failed to show a distinct advantage (Blom et al, 1999; Schafhauser et al, 1999; Terrone et al, 2003; Blute et al, 2004a; Phillips and Taneja, 2004; Patard, 2005; Blom et al, 2008; Leibovich and Blute, 2008; O’Malley et al, 2009b). There are several factors that may mitigate against a benefit of routine lymphadenectomy (Leibovich and Blute, 2008; O’Malley et al, 2009a). First, RCC metastasizes through the bloodstream and the lymphatic system with equal frequency. Consequently, most patients with positive lymph nodes eventually develop blood-borne metastases despite undergoing lymphadenectomy, and many patients with RCC will develop disseminated metastases without ever having lymphatic involvement. In addition, the lymphatic drainage of the kidney is variable and even an extensive retroperitoneal dissection may not remove all possible sites of metastasis. Many believe that only a relatively small percentage of patients (<2% to 3%) are likely to benefit from routine lymphadenectomy, namely, the subset of patients with micrometastatic disease (Golimbu et al, 1986; Giuliani et al, 1990; Herrlinger et al, 1991; Leibovich and Blute, 2008; O’Malley et al, 2009a). In all likelihood, the involved lymph nodes in many of these patients would be removed by conventional RN, which incorporates the renal hilar and immediately adjacent paracaval or para-aortic lymph nodes.
At present, the need for routine performance of an extended lymphadenectomy in all cases of RN is not well defined and most urologists perform this selectively (Blom et al, 1999; Blute et al, 2004a; Daneshmand et al, 2005; Phillips and Taneja, 2004; Leibovich and Blute, 2008). Blute and colleagues (2004a) have reported risk factors for lymphatic involvement with RCC, including high tumor grade, sarcomatoid component, histologic tumor necrosis, tumor size larger than 10 cm, and tumor stage pT3 or pT4. The incidence of lymph node involvement in their series was 10% if two or more of these factors were present and only 0.6% if fewer than two were present. Most of these factors can be assessed preoperatively or with intraoperative frozen-section analysis, and this could be used to guide decision making with respect to lymphadenectomy. With this approach only the hilar and regional lymph nodes would be removed for low-risk patients (Leibovich and Blute, 2008).
Open surgical techniques for RN are described in detail in Chapter 54. The surgical approach for RN is determined by the size and location of the tumor as well as by the body habitus of the patient (Diblasio et al, 2006; Russo, 2006). The operation is usually performed through a transperitoneal incision to allow abdominal exploration for metastatic disease and early access to the renal vessels with minimal manipulation of the tumor. The authors prefer an extended subcostal for most patients undergoing open RN. The thoracoabdominal approach is employed only occasionally, when RN is performed in patients with very large and potentially invasive tumors involving the upper portion of the kidney. An extraperitoneal flank incision may be appropriate in elderly patients or patients of poor surgical risk, but exposure can be limiting, particularly in patients with large tumors or those with contentious hilar anatomy (Diblasio et al, 2006; Russo, 2006). In reality, most of these patients are now managed with a laparoscopic approach in this era.
Laparoscopic RN has emerged as a less morbid alternative to open surgery in the management of low- to moderate-volume (10 to 12 cm or smaller), localized RCCs with no local invasion, limited or no renal vein involvement, and manageable lymphadenopathy. Laparoscopic RN is associated with diminished postoperative discomfort and shortened recovery, and costs compare favorably with the open approach (Meraney and Gill, 2002; Hemal et al, 2007b). Available outcome data suggest that cancer-specific survival after laparoscopic RN is comparable to that after open RN (Cadeddu et al, 1998; Dunn et al, 2000; Meraney et al, 2000; Chan et al, 2001; Gill et al, 2001; Portis et al, 2002; Saika et al, 2003; Wille et al, 2004; Permpongkosol et al, 2005b; Yoshino et al, 2005; Breda et al, 2007b; Burgess et al, 2007; Hemal et al, 2007a; Columbo et al, 2008). In an early multi-institutional report of 157 patients undergoing laparoscopic RN for clinical stage I (T1-2) RCC, the 5-year actuarial disease-free rate for all patients was 91% (Cadeddu et al, 1998). Only 5 patients developed retroperitoneal or metastatic tumor recurrence, and there were no port site recurrences. Current laparoscopic techniques allow replication of the important tenets of RN, and outcome data reflect this at several centers (Cadeddu et al, 1998; Dunn et al, 2000; Meraney et al, 2000; Chan et al, 2001; Gill et al, 2001; Kim FJ et al, 2003; Portis et al, 2002; Saika et al, 2003; Simon et al, 2004; Wille et al, 2004; Permpongkosol et al, 2005b ; Burgess et al, 2007; Hemal et al, 2007b; Columbo et al, 2008). A variety of approaches including transperitoneal, retroperitoneal, and hand-assisted laparoscopic RN have been popularized and are described elsewhere in this text (see Chapter 55) (Cadeddu et al, 1998; Gill et al, 2001; Matin et al, 2002; Nelson and Wolf, 2002; Makhoul et al, 2004; Desai et al, 2005; Nadler et al, 2006; Chung et al, 2007; Kawauchi et al, 2007; Miyake et al, 2007). The current data suggest that elderly and morbidly obese patients, those with a history of previous abdominal surgery, and those with large tumor size may also be considered for laparoscopic renal surgery, although selection of patients must be judicious and surgical expertise and experience should also be taken into account (Fazeli-Matin et al, 1999; Parsons et al, 2002; Matin et al, 2003; Cobb et al, 2004; Fugita et al, 2004; Steinberg et al, 2004; Viterbo et al, 2005; Feder et al, 2008; Gabr et al, 2008).
Laparoscopic approaches are now accepted as standard in the field for RN in appropriately selected patients, providing equivalent oncologic outcomes to the open counterpart with the advantages of more rapid recovery (Desai et al, 2003b; Meraney et al, 2003; Steinberg et al, 2003; Abreu et al, 2004; Fenn and Gill, 2004; Finelli et al, 2004b, 2005; Johnston et al, 2005; Permpongkosol et al, 2005; Hollingsworth et al, 2006; Burgess et al, 2007; Hemal et al, 2007b; Columbo et al, 2008). One concern is that laparoscopic RN has become particularly appealing to patients and physicians due to these considerations, and this has likely been a major driver in the overutilization of RN over the past several years (Fig. 49–19). Even in the current era, most patients with small renal masses are still managed with RN, despite convincing outcomes data in favor of nephron-sparing approaches such as PN (Hollenbeck et al, 2006; Miller et al, 2006; Porter and Lin, 2007).
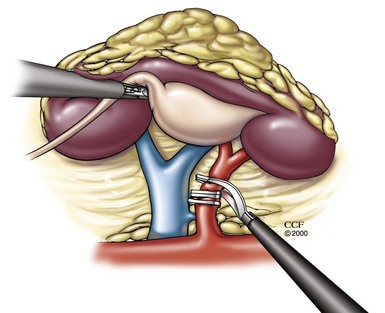
Figure 49–19 Laparoscopic radical nephrectomy (a retroperitoneal approach is illustrated) provides excellent oncologic outcomes and rapid recovery but predisposes patients to chronic kidney disease and attendant cardiovascular risks and increased mortality rates. Nephron-sparing approaches should be prioritized whenever possible.
(Reprinted with permission, Cleveland Clinic Center for Medical Art & Photography © 2007-2009. All Rights Reserved.)
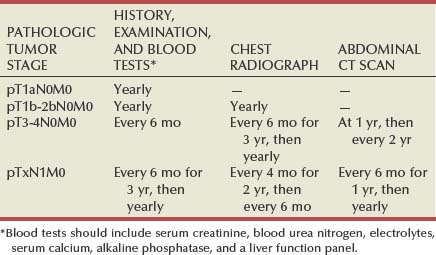
Several studies on outcomes after RN for localized RCC have now demonstrated that the risk for postoperative recurrent malignant disease is stage dependent, and surveillance protocols should reflect this (Sandock et al, 1995; Levy et al, 1998; Eggener et al, 2001; Janzen et al, 2003; Stephenson et al, 2004a; Theodorescu et al, 2004; Sorbellini et al, 2005; Skolarikos et al, 2007). For instance, in a study from M. D. Anderson Cancer Center (Levy et al, 1998), 68 of 286 patients (23.8%) developed metastatic RCC after RN and the incidence was 7.1% for stage T1N0M0, 26.5% for T2N0M0, and 39.4% for T3N0M0. The risk for developing recurrent malignant disease was greatest during the first 3 postoperative years. Stephenson and colleagues (2004a) also reported substantially increased risk of recurrence in patients with stage pT3a and pT3b compared with pT1 and pT2, and recurrences were diagnosed earlier in the former, with a median of 12 versus 26 months. These data indicate that surveillance for recurrent malignant disease after RN for RCC can be tailored according to the initial pathologic tumor stage. The recommended surveillance scheme is depicted in Table 49–20. Postoperative bone scintiscans and head CT scans are necessary only in the presence of related symptoms. This surveillance scheme is cost effective and enables early detection of most cases of recurrent RCC after RN for localized disease. Other important prognostic factors for RCC, such as tumor size, grade, and histologic subtype, can also influence the risk and pattern of recurrence and are now integrated into predictive algorithms that will be useful for more individualized surveillance in the near future (Kattan et al, 2001; Pantuck et al, 2001a; Zisman et al, 2002c; Frank et al, 2003a, 2005e; Lam et al, 2005b; Sorbellini et al, 2005; Antonelli et al, 2007). Surveillance for CKD is also of paramount importance along with timely referral for nephrologic consultation (Lane et al, 2009). Close nephrologic surveillance and skilled management can delay the progression of CKD and mitigate against the negative effects of associated conditions, including cardiovascular disease, poor bone health, anemia, and sexual dysfunction (Lane et al, 2009).
Partial Nephrectomy
Nephron-sparing surgery for the treatment of a renal tumor was first described by Czerny in 1890 (reviewed in Herr, 2005). However, high morbidity limited its application. In 1950, Vermooten suggested that peripheral encapsulated renal neoplasms could be excised locally while leaving a margin of normal parenchyma around the tumor. Interest in PN for RCC has been stimulated by advances in renal imaging, experience with renal vascular surgery for other conditions, improved methods of preventing ischemic renal damage, growing numbers of incidentally discovered low-stage RCCs, greater appreciation of the deleterious effects of CKD, and good long-term survival in patients undergoing this form of treatment (Uzzo and Novick, 2001; Russo et al, 2008; Campbell et al, 2009; Lane et al, 2009; Novick, 2009). Nephron-sparing surgery entails complete local resection of the renal tumor while leaving the largest possible amount of normal functioning parenchyma in the involved kidney (Fig. 49–20).
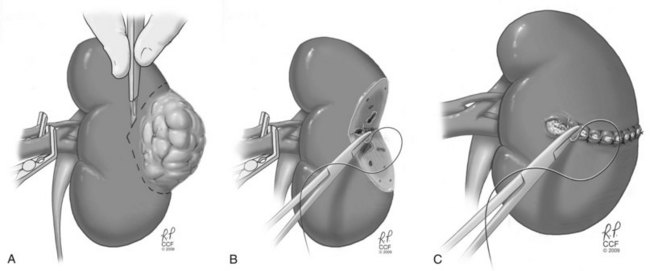
Figure 49–20 Essential steps in partial nephrectomy. A, Temporary occlusion of the vascular pedicle and excision of the tumor with a rim of normal parenchyma. B, Closure of the collecting system and ligation of transected vessels. C, Capsular reconstruction.
(Reprinted with permission, Cleveland Clinic Center for Medical Art & Photography © 2007-2009. All Rights Reserved.)
Accepted indications for PN traditionally included situations in which RN would render the patient anephric or at high risk for ultimate need of dialysis (Licht et al, 1994; Ghavamian and Zincke, 2001; Uzzo and Novick, 2001; Nguyen et al, 2008a; Russo et al, 2008; Campbell et al, 2009; Lane et al, 2009). This encompassed patients with bilateral RCC or RCC involving a solitary functioning kidney. A solitary functioning kidney may be the result of unilateral renal agenesis, prior removal of the contralateral kidney, or irreversible impairment of contralateral renal function by a benign disorder. Another traditional relative indication for PN was represented by patients with unilateral carcinoma and a functioning opposite kidney affected by a condition that might threaten its future function, such as renal artery stenosis (Campbell et al, 1993; Hafez et al, 2000), hydronephrosis, chronic pyelonephritis, ureteral reflux, calculus disease, or systemic diseases such as diabetes and nephrosclerosis (Uzzo and Novick, 2001; Gilbert et al, 2003; Nguyen et al, 2008a; Russo et al, 2008; Campbell et al, 2009; Lane et al, 2009; Novick, 2009).
In patients with bilateral synchronous RCC, the general approach has been to preserve as much functioning renal tissue as possible. This entails performing bilateral PNs when feasible, usually as staged procedures if the tumors are relatively large. When a locally extensive tumor on one side precludes nephron-sparing surgery, an RN is performed on the more involved side along with a contralateral PN (Booth et al, 2008; Nguyen et al, 2008a; Rothman et al, 2008). Margin width appears to be immaterial as long as the final margins are negative; this is particularly relevant when the tumor is located within the hilum and preservation of renal function is at a premium (Castilla et al, 2002; Sutherland et al, 2002; Li et al, 2008; Yossepowitch et al, 2008; Campbell et al, 2009).
Patients with RCC involving a functionally or anatomically solitary kidney must be advised about the potential need for temporary or permanent dialysis postoperatively, which, according to the authors’ experience, occurs in 3.5% and 0.5% of cases, respectively (Fergany et al, 2006). In this series, 18 of 400 patients (4.5%) with a solitary kidney managed with PN eventually progressed to end-stage renal failure at a mean of 3.6 years after surgery. Many of these patients also had preexisting CKD before surgery, and in some instances only a small remnant kidney could be preserved owing to tumor size and anatomic considerations. Similarly, Ghavamian and colleagues (2002) reported a 12.7% incidence of acute renal failure when operating on a solitary kidney, with 15.9% developing proteinuria and 12.7% experiencing severe CKD on a long-term basis. A functioning renal remnant of at least 20% of one kidney is necessary to avoid end-stage renal failure, although this presumes good functional status of the remaining parenchyma (Uzzo and Novick, 2001). Overall preservation of renal function is thus achieved in the great majority of patients with PN, even in patients with traditional imperative indications (Uzzo and Novick, 2001; Nguyen et al, 2008a; Novick, 2009). Local recurrence after PN for imperative indications has ranged from 3% to 5%, because many of these cases are particularly challenging owing to hilar tumor location, the need to minimize the amount of excised functional parenchyma, tumor multifocality, or other complexities (Uzzo and Novick, 2001; Nguyen et al, 2008a).
PN is now standard of care for the management of clinical T1 renal masses in the presence of a normal contralateral kidney, presuming that the mass is amenable to this approach (Nguyen et al, 2008a; Russo et al, 2008; Russo and Huang, 2008; Huang et al, 2009; Campbell et al, 2009; Lane et al, 2009; Novick, 2009). This indication was previously considered “elective,” but recent data demonstrating the importance of optimizing renal function after surgery for clinical T1 renal masses has persuasively altered this perspective. A robust literature now demonstrates equivalent oncologic outcomes with PN when compared with RN in appropriately selected patients, and the renal functional outcomes tilt the balance in favor of nephron-sparing approaches whenever feasible (Lau et al, 2000; Lee et al, 2000; McKiernan et al, 2002b; Becker et al, 2006b; Mitchell et al, 2006; van Poppel et al, 2007; Rabets and Novick, 2006; Russo et al, 2008; Thompson et al, 2008; Campbell et al, 2009; Huang et al, 2009; Lane et al, 2009). Prior experience with “elective” PN for T1a RCC demonstrated local recurrence rates of 1% to 2%, and overall cancer-free survival well over 90% (Uzzo and Novick, 2001; Campbell et al, 2009). The morbidity of PN has also decreased substantially in recent years in experienced hands (Thompson et al, 2005d; Diblasio et al, 2006; Joudi et al, 2007; Patard et al, 2007). Extension of this approach to clinical T1b tumors has yielded similar encouraging results (Becker et al, 2006a; Carini et al, 2006; Dash et al, 2006; Campbell and Novick, 2006; Crispen et al, 2008a; Nguyen et al, 2008a; Pahernik et al, 2008; Campbell et al, 2009; Peycelon et al, 2009). The majority of local recurrences observed after PN are most likely a manifestation of undetected microscopic multifocal RCC in the renal remnant—most are found distant from the previous tumor bed. Concern about local recurrence after PN is counterbalanced by a 1% to 2% incidence of contralateral RCC on longitudinal surveillance, in which case RN would have left the patient with tumor in a solitary kidney (Nguyen et al, 2008a; Russo et al, 2008). Interestingly, Clark and colleagues (2001) have shown that quality of life and psychological adaptation may be better after PN compared with RN, and costs appear to be comparable whether RN is performed open or laparoscopically (Uzzo et al, 1999c; McKiernan et al, 2002a; Shekarriz et al, 2002; Poulakis et al, 2003; Stephenson et al, 2004b).
Evaluation of patients with RCC for PN should include preoperative testing to exclude locally extensive or metastatic disease and additional specific renal imaging to delineate the relationship of the tumor to the intrarenal vascular supply and collecting system. For centrally located tumors, this information has traditionally been obtained with a combination of studies including CT, renal arteriography, and, occasionally, renal venography. Three-dimensional volume-rendered CT (or MRI) is now established as a noninvasive imaging modality that can accurately depict the renal parenchymal and vascular anatomy in a format familiar to urologic surgeons (Coll et al, 1999; Uzzo et al, 2000a; Berman et al, 2001; Vanderbrink et al, 2004; Nguyen et al, 2008a; Simmons et al, 2007; Novick, 2009). This study integrates essential information from arteriography, venography, excretory urography, and conventional two-dimensional CT into a single imaging modality and obviates the need for more invasive renal imaging (Derweesh et al, 2004; Simmons et al, 2008). The surgical techniques for performing nephron-sparing surgery in patients with RCC are reviewed in Chapters 54 and 55.
One of the largest reported studies of nephron-sparing surgery is from the Cleveland Clinic and reviewed the results of PN for the treatment of localized, sporadic RCC in 485 patients (Hafez et al, 1999). The mean postoperative follow-up was 4 years, and overall and cancer-specific 5-year survival rates for patients in this series were 81% and 92%, respectively. Recurrent RCC developed postoperatively in 44 patients (9%), including 16 (3.2%) with local recurrence in the remnant kidney and 28 (5.8%) with metastatic disease. In another study from the Cleveland Clinic, long-term results of nephron-sparing surgery were reviewed in 107 patients with localized sporadic RCC treated before 1988 who were followed for a minimum of 10 years or until death (Fergany et al, 2000). Cancer-specific survival was 88% at 5 years and 73% at 10 years. Long-term renal function was preserved in 100 patients (93%). These results are particularly impressive given the patient population in that era, almost all of whom underwent PN for imperative indications. Ten-year follow-up was also provided by Herr (1999), who reported that 97% of patients remained cancer free after PN in the setting of a normal contralateral kidney, a more select population of patients. These data confirm that nephron-sparing surgery provides effective long-term therapy for patients with localized RCC and can preserve renal function in the overwhelming majority (Saad et al, 2005; Nguyen et al, 2008a; Novick, 2009).
A more recent trend has been to perform PN by minimally invasive approaches, which is a considerable challenge given concern about margin status when visualization can be compromised by suboptimal hemostasis and in the absence of tactile input. Nevertheless, several series have now reported encouraging results, and this field is evolving rapidly (Gill et al, 2002, 2003b, 2006, 2007, 2008; Hu et al, 2002; Guillonneau et al, 2003; Simon et al, 2003; Allaf et al, 2004; Finelli et al, 2004a; Kane et al, 2004; Mabjeesh et al, 2004; Frank et al, 2006; Porpiglia et al, 2008; Turna et al, 2008; Nadu et al, 2009). Many groups have now developed techniques that essentially duplicate the open surgical technique, including occlusion of the renal vasculature, excision of the tumor with a rim of normal parenchyma, and intracorporeal suturing to close the collecting system and repair the capsular defect (Desai et al, 2003a; Gill et al, 2007; Gill et al, 2008). Hemostasis remains the primary challenge given the great blood flow associated with the kidneys, and a variety of adjuvant approaches have been explored to accomplish this, including the use of tissue sealants, hemostatic dissecting devices, and robotic technology (Gettman et al, 2001b; Yoshimura et al, 2001; Richter et al, 2003; Hasan et al, 2004a, 2004b; Herrel et al, 2004; Urena et al, 2004; Finley et al, 2005; Gill et al, 2008; Porpiglia et al, 2008; Turna et al, 2008). Margin status and intermediate oncologic outcomes associated with laparoscopic PN appear to be equivalent to open PN in experienced hands, presuming sensible patient selection (Lane and Gill, 2007).
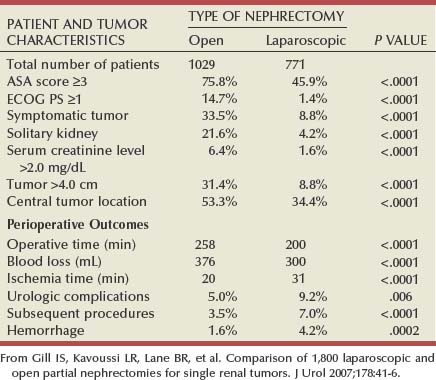
A recent study from three centers of excellence provides important information about outcomes with open and laparoscopic PN, albeit somewhat in the developmental phase of the latter (Gill et al, 2007) (Table 49–21). This study evaluated 1029 open PN (OPN) and 771 laparoscopic PN (LPN), with analysis focused on patient and tumor characteristics in addition to perioperative outcomes. Strong selection bias is evident, as expected for any new technique, because the LPN group was found to have favorable characteristics in terms of better general health, smaller tumor size, and more asymptomatic presentation and less likely to have central tumor location, preexisting CKD, or a solitary kidney. LPN was associated with shorter operative times and less blood loss, although warm ischemic times were longer and the risk of urologic complications was higher. Warm ischemic time is particularly important in this era because it is now established as the major modifiable determinant of postoperative renal function (Thompson et al, 2007c; Lane et al, 2008a). In addition, urologic complications, such as postoperative hemorrhage and urinary leak, and the need for subsequent procedures, were substantially increased in the LPN group on a comparative basis (Gill et al, 2007). Recent advances in the technique of LPN, such as the early unclamping maneuver in which the vascular pedicle clamp is removed immediately after running a deep parenchyma suture across the defect, hold great promise for improving on these outcomes (Baumert et al, 2007; Bollens et al, 2007; Breda et al, 2007b; Simmons et al, 2007). Nguyen and Gill (2008) have reported substantially reduced warm ischemic times with this approach, and postoperative bleeding rates have also fallen, presumably because potential bleeders can be identified intraoperatively and prospectively addressed prior to closing the capsule. Please refer to Chapter 55 for further technical details regarding LPN.
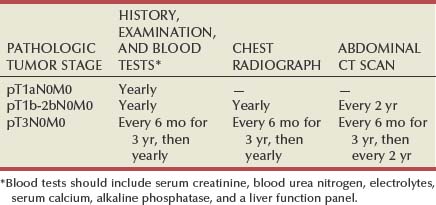
Clinical guidelines for long-term surveillance after PN for sporadic localized RCC have been developed (Eggener et al, 2001; Frank et al, 2003a; Janzen et al, 2003; Theodorescu et al, 2004; Lam et al, 2005b; Sorbellini et al, 2005; Antonelli et al, 2007; Skolarikos et al, 2007; Klatte et al, 2008b), originally based on a detailed analysis of tumor recurrence patterns in 327 patients treated at the Cleveland Clinic (Hafez et al, 1997). In this series, recurrent RCC developed after nephron-sparing surgery in 38 patients (11.6%), including 13 patients (4%) who developed local recurrence and 25 patients (7.6%) who developed metastatic disease (Hafez et al, 1997). The incidence of postoperative local recurrence and metastatic disease varied according to the initial pathologic tumor stage as follows: 0% and 4.4% for T1N0M0 tumors, 2% and 5.3% for T2N0M0 tumors, 8.2% and 11.5% for T3aN0M0 tumors, and 10.6% and 14.9% for T3bN0M0 tumors. The peak postoperative intervals for development of local recurrence were 6 to 24 months (in T3 tumors) and more than 48 months (in T2 tumors). T1 tumor stage in this series represented tumor size of less than 2.5 cm, based on a previous TNM classification system. These data indicate that surveillance for recurrent malignant disease after PN for sporadic RCC can be tailored according to the initial pathologic tumor stage and tumor size or an algorithm based on these and other clinicopathologic factors predictive of recurrence (Frank et al, 2003a; Klatte et al, 2008b). A relatively straight-forward surveillance scheme recommended by the authors is depicted in Table 49–22. This protocol minimizes costs, radiographic exposure, and patient inconvenience while still allowing for detection of most clinically relevant recurrences.
Patients who undergo nephron-sparing surgery for RCC may be left with a relatively small amount of renal tissue and are at risk for development of long-term renal functional impairment from hyperfiltration renal injury (Modlin and Novick, 2001; Abdi et al, 2003; Lane et al, 2009; Novick, 2009). In a study of 14 patients observed for up to 17 years after PN in a solitary kidney, patients with more than 50% reduction in overall renal mass were found to be at increased risk for development of proteinuria, focal segmental glomerulosclerosis, and progressive renal failure (Novick et al, 1990). The development of proteinuria correlated directly with the length of follow-up and inversely with the amount of remaining renal tissue. Renal biopsy revealed focal segmental glomerulosclerosis in several patients with severe proteinuria (Fig. 49–21). These findings mirror those observed in experimental animal models of partial renal ablation (Brenner, 1983). Because proteinuria is the initial manifestation of this phenomenon, a 24-hour urinary protein measurement should be obtained yearly in patients with a solitary remnant kidney to screen for hyperfiltration nephropathy.
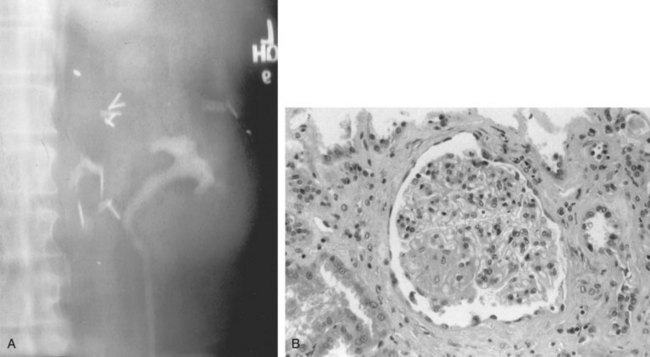
Figure 49–21 A, Ten years after partial nephrectomy for large tumor in solitary left kidney, intravenous pyelogram shows function of small renal remnant. The patient had developed nephrotic syndrome at this time. B, Renal biopsy specimen shows focal segmental glomerulosclerosis indicative of hyperfiltration nephropathy.
Efforts to prevent or to ameliorate the damaging effects of renal hyperfiltration have primarily focused on dietary and pharmacologic intervention (Modlin and Novick, 2001; Lane et al, 2009). Animal studies have suggested that angiotensin-converting enzyme inhibitor (ACEI) agents and dietary restriction of protein may mitigate this type of glomerulopathy (Meyer et al, 1985). Preliminary clinical data appear to support this concept. Novick and Schreiber (1995) studied five patients who had developed proteinuria with stable renal function after PN in a solitary kidney. Four of these patients had documented focal segmental glomerulosclerosis on renal biopsy during follow-up, although all five patients had normal renal morphologic features at the time of surgery. Treatment with ACEI therapy and a low-protein diet decreased the level of proteinuria in four patients. Data from other studies have also suggested that ACEI therapy can significantly diminish proteinuria in patients with established renal disease (Goldfarb, 1995). This information suggests that ACEI therapy and a low-protein diet may improve the long-term renal functional outcome for patients with a remnant kidney after PN. The optimal time for initiating the regimen is not clear, and it may be best to implement this therapy as early as possible to obviate the maladaptive responses that can lead to progressive sclerosis and renal failure in this setting.
There is experimental evidence that nonhemodynamic processes may also contribute to progression of sclerosis in the remnant kidney (Goldfarb, 1995). This raises other therapeutic possibilities, such as thromboxane inhibitors, anticoagulants, lipid-lowering agents, and other pharmacologic agents. Future clinical trials, it is hoped, will clarify the potential value of these treatment approaches.
Thermal Ablative Therapies
Thermal ablative (TA) therapies, including renal cryosurgery and radiofrequency ablation (RFA), have emerged as alternative nephron-sparing treatments for patients with localized RCC (Marshall, 1999; Murphy and Gill, 2001; Lowry and Nakada, 2003; Hinshaw and Lee, 2004; Mabjeesh et al, 2004; Ost and Kavoussi, 2007; Sterrett et al, 2008; Matin and Ahrar, 2009). Both can be administered percutaneously or through laparoscopic exposure and thus offer the potential for reduced morbidity and more rapid recovery (Johnson et al, 2004; Sterrett et al, 2008). However, long-term efficacy is not as established as with surgical excision and preliminary data suggest that the local recurrence rates may be somewhat higher than that reported for traditional surgical approaches (Campbell et al, 2009; Kunkle et al, 2008; Matin and Ahrar, 2009). Another concern has been the lack of accurate histologic and pathologic staging associated with these modalities, because the treated lesion is left in situ.
The ideal candidates for TA procedures may be patients with advanced age or significant comorbidities who prefer a proactive approach but are not optimal candidates for conventional surgery, patients with local recurrence after previous nephron-sparing surgery, and patients with hereditary renal cancer who present with multifocal lesions for which multiple PNs might be cumbersome if not impossible (Kunkle et al, 2008). Patient preference must also be considered, and some patients not fitting these criteria may also select TA, a decision that can be supported as long as balanced counseling about the current status of these modalities has been provided (Ost and Kavoussi, 2007; Sterrett et al, 2008; Matin and Ahrar, 2009). Finally, tumor size is also an important factor in patient selection because the current technology does not allow for reliable treatment of lesions larger than 4.0 cm in diameter (Zagoria et al, 2007; Matin and Ahrar, 2009).
Experience with renal cryosurgery predates that of RFA and has been more extensive (Sterrett et al, 2008; Matin and Ahrar, 2009). The kidney may be an anatomically favored organ for cryosurgery, particularly compared with the prostate, another target organ for cryoablation. Unlike the prostate, which is in intimate, fixed contact with the rectum and the sphincteric mechanism, the kidney can be laparoscopically mobilized, thereby minimizing the risk of injury to adjacent vital structures. Furthermore, while the prostate often harbors multifocal, infiltrative carcinoma, the kidney usually presents with unifocal, well-circumscribed malignancy. Because the renal tumor and “ice ball” that forms during treatment can be readily visualized with ultrasonography, the ice ball can be confirmed to completely and circumferentially obliterate the tumor margins, including a surrounding margin of healthy tissue, with increased precision and confidence. An extensive body of experimental work is available to guide and support clinical cryoablation (Hinshaw and Lee, 2004; Sterrett et al, 2008).
Established prerequisites for successful cryosurgery include rapid freezing, gradual thawing, and a repetition of the freeze-thaw cycle. The mechanism underlying tissue cryodestruction is thought to involve immediate cellular damage and delayed microcirculatory failure (Gill and Novick, 1999; Baust and Gage, 2005; Ost and Kavoussi, 2007; Stein and Kaouk, 2007; Sterrett et al, 2008; Matin and Ahrar, 2009). Ice formation occurs initially in the extracellular space, causing the extracellular fluid to become hyperosmotic. To equilibrate chemical osmolality, water permeates from the intracellular compartment into the extracellular compartment. Osmolality of the intracellular fluid is thus increased, leading to intracellular solute concentration and dehydration, which cause desiccation trauma—the first step of chemical cellular injury. Continued rapid supercooling leads to the second step of cellular damage—intracellular ice formation. Intracellular ice irreversibly disrupts cell organelles and the cell membrane, a lethal event. Delayed microcirculatory failure occurs during the slow thaw phase of the freeze-thaw cycle, leading to circulation arrest and cellular anoxia. Progressive disruption of the microcirculation occurs along a cascade of events: vasoconstriction, endothelial cell destruction causing vessel walls to become porous, interstitial edema, platelet aggregation, microthrombi, and, ultimately, vascular congestion and obliteration. Cells that survive the initial cryogenic assault are destroyed by this secondary insult of ischemia. Repetition of the rapid freeze–slow thaw cycle potentiates the damage. The cryoablation area is thus rendered ischemic, leading ultimately to a circumscribed necrosis (Hinshaw and Lee, 2004; Ost and Kavoussi, 2007; Sterrett et al, 2008; Matin and Ahrar, 2009).
Further work defined treatment parameters required to bring this treatment into the clinical domain. Chosy and colleagues (1996) demonstrated that complete necrosis of in-vivo porcine renal parenchyma could be consistently achieved only at temperatures of −19.4° C or lower; necrosis was incomplete at temperatures above this threshold. Campbell and coworkers (1998) confirmed that the target lethal temperature of −20° C was achieved at a distance of 3.1 mm inside the leading edge of the ice ball as visualized by real-time ultrasonography. Thus, to ensure complete cell kill, the ice ball must extend well beyond the visible margins of the targeted tumor. In practice, we routinely extend the ice ball approximately 1 cm beyond the edge of the tumor, as determined by both laparoscopic and real-time ultrasonographic imaging (Gill et al, 1998). The availability of sophisticated and reliable ultrasonography and the introduction of finer cryoprobes that allow more accurate and less traumatic probe placement have contributed to even greater interest in visceral cryosurgery (Onik et al, 1993; Pantuck et al, 2002b; Ost and Kavoussi, 2007; Stein and Kaouk, 2007; Sterrett et al, 2008; Matin and Ahrar, 2009).
Clinical experience and follow-up of patients after renal cryoablative therapy suggests successful local control in 90% to 95% of patients, although many studies provide limited, and often incomplete, follow-up (Gervais et al, 2005; Gill et al, 2005; Davol et al, 2006; Matin et al, 2006; Ost and Kavoussi, 2007; Stein and Kaouk, 2007; Atwell et al, 2008; Sterrett et al, 2008; Matin and Ahrar, 2009). Diagnosis of local recurrence after thermal ablative treatments can be challenging because evolving fibrosis within the tumor bed can be difficult to differentiate from residual cancer. In general, central or nodular enhancement within the tumor bed on extended follow-up has been considered diagnostic of local recurrence, and the general clinical experience with cryoablation has thus far supported this (Bolte et al, 2006; Weight et al, 2008). However, the number of studies that have incorporated routine post-therapy biopsies to provide histologic confirmation are in the minority (Gill et al, 2000a; Weight et al, 2008; Matin and Ahrar, 2009). Weight and colleagues (2008) demonstrated that none of the 60 patients with absence of enhancement after cryoablation had residual cancer on follow-up biopsy while peripheral and central enhancement were associated with cancer in 8% and 63% of patients, respectively.
Reported rates of local recurrence in some of the ablation series may represent underestimates because about 20% of small renal masses are benign rather than RCC, and a pretreatment biopsy, although strongly advocated in the field, has not been routinely performed in many series (Silver et al, 1997; Young et al, 2002; Heilbrun et al, 2007; Sterrett et al, 2008). In addition, patients may have a recurrence at other sites in the kidney, and this should also be considered local recurrence although the mechanism is different (novel tumor occurrence due to multifocality rather than treatment failure). Hasan and colleagues (2004a) reported two such ipsilateral occurrences when their patients were observed for a minimum of 48 months, so the true incidence of local recurrence in this series was 10%, not the reported 5%, higher than would be expected for conventional PN for an analogous population of patients with small renal tumors. Indeed, recurrence in the tumor bed after conventional PN is uncommon; most local recurrences are distant from the original tumor site. This cumulative experience suggests that local control after cryoablative therapy approaches, but may not reach, that attained by conventional surgical excision (Kunkle et al, 2008; Matin and Ahrar, 2009).
Other concerns with cryoablation, and TA in general, relate to surgical salvage and long-term outcomes. Most local recurrences can be salvaged with repeat ablation, although some patients with progressive disease eventually require conventional surgery. Nguyen and colleagues (2008b) have shown that PN and minimally invasive approaches are often not possible in this setting due to the extensive fibrotic reaction induced by the previous treatment. This reaction appears to be more significant after cryoablation than after RFA or PN, although the precise reasons for this are unclear at present. Long-term outcomes after TA are limited, and the few that are available suggest suboptimal outcomes in a finite proportion of patients (Kunkle et al, 2008; Campbell et al, 2009). In the most robust and mature single institution experience with cryoablation, 5- and 10-year disease-free survival rates were 78% and 51%, respectively, much less than would be expected for PN for an analogous population of patients with small renal tumors (Aron et al, 2007). Complications associated with cryoablation can include renal fracture, hemorrhage, adjacent organ injury, ileus, and wound infection, although major morbidity is decidedly uncommon (Lane et al, 2005; Ost and Kavoussi, 2007; Finley et al, 2008; Sterrett et al, 2008; Matin and Ahrar, 2009).
The experience with RFA is more limited—this technology is at an earlier state of development than cryoablation (Corwin et al, 2001; Murphy and Gill, 2001; Rendon et al, 2001; Zelkovic and Resnick, 2003; Mabjeesh et al, 2004; Hegarty et al, 2006; Aron et al, 2007; Ost and Kavoussi, 2007; Sterrett et al, 2008; Matin and Ahrar, 2009). Mechanistically, heat above 45° C leads to irreversible cellular damage, and temperatures higher than 55° C to 60° C result in immediate cell death. Application of high-frequency electrical current by RFA induces excitation of ions, frictional forces, and heat, which in turn cause denaturation of intracellular proteins and melting of cellular membranes, a lethal sequence of events. These effects are observed at tissue temperatures above 41° C but increase directly with increasing temperature and duration of treatment (Mabjeesh et al, 2004; Sterrett et al, 2008; Matin and Ahrar, 2009). Temperatures in excess of 100° C are typically obtained at the tips of the probes, and thermosensors near the tips can be used to monitor progress during active treatment. Temperature dissipates at points more distant from the probe tip, and multiple probes are typically required to achieve adequate heating of the entire region of interest (Murphy and Gill, 2001). One disadvantage of RFA is that the treatment effect is more difficult to monitor in real time—there is no true “ice ball” equivalent (Zelkovic and Resnick, 2003). Rather, treatment is typically based on empirical results from previous probe alignments, and this allows a fairly predictable target zone of up to 4.0 cm to be treated in most cases. Maximal tumor size that can be adequately treated would of necessity be smaller than this, given the need to extend the treatment zone beyond all edges of the tumor. Novel approaches to extend these boundaries are being explored (Matin and Ahrar, 2009).
Local control after RFA is difficult to determine owing to a number of complexities, although most estimate that this will be 80% to 90% on a longitudinal basis using strict definitions of local recurrence (Matin et al, 2006; Ost and Kavoussi, 2007; Kunkle et al, 2008; Levinson et al, 2008; Campbell et al, 2009; Matin and Ahrar, 2009). Loss of enhancement on cross-sectional imaging within the lesion has generally been accepted as an indicator of successful treatment after TA (Raman et al, 2008), although a report by Weight and colleagues (2008) has challenged this. This group described 6 patients with no enhancement on MRI performed 6 months after RFA who were found to have apparently viable cancer cells on biopsy of the tumor bed, indicating that 24% of lesions with no radiographic suspicion for tumor persistence may have been incompletely treated (Weight et al, 2008). The technology for RFA continues to improve, and most contemporary series report low rates of local recurrence, although some patients have required repeat treatments to achieve local control, which is an infrequent event with cryoablation and rarely required with conventional surgical treatments for localized RCC (Anderson et al, 2005; Coleman et al, 2005; Schenk et al, 2005; Ost and Kavoussi, 2007; Sterrett et al, 2008; Matin and Ahrar, 2009). Complications are uncommon but have included acute renal failure, stricture of the ureteropelvic junction, necrotizing pancreatitis, and lumbar radiculopathy, so careful and judicious selection of patients is essential (Ogan et al, 2002; Coskun et al, 2003; Elias et al, 2004; Schenk et al, 2005; Weizer et al, 2005a, 2005b; Ost and Kavoussi, 2007; Sterrett et al, 2008; Matin and Ahrar, 2009). Direct comparison with cryoablation is inevitable but perhaps unfair because RFA is earlier in its development and recent advances suggest great promise (Anderson et al, 2005; Coleman et al, 2005; Schenk et al, 2005; Ost and Kavoussi, 2007; Sterrett et al, 2008; Matin and Ahrar, 2009). Nevertheless, with the development of protocols to administer cryoablation percutaneously, many centers are offering cryoablation to patients who otherwise might have been treated with RFA in the past (Shingleton and Sewell, 2001, 2002; Bassignani et al, 2004; Silverman et al, 2005; Tuncali et al, 2006; Atwell et al, 2007, 2008; Georgiades et al, 2008; Hinshaw et al, 2008).
Other exciting new technologies, such as high-intensity focused ultrasound (HIFU) and frameless, image-guided radiosurgical treatments (CyberKnife), are also under development and may allow extracorporeal treatment of small renal tumors in the future (Kohrmann et al, 2002; Ponsky et al, 2003; Wu et al, 2003; Roberts et al, 2006; Svedman et al, 2006; Clark et al, 2007; Marberger, 2007; Ponsky et al, 2007; Klinger et al, 2008; Liang et al, 2008; Matin and Ahrar, 2009). However, at present cell kill with these modalities is not sufficiently reliable and they are best considered developmental (Matin and Ahrar, 2009).
Active Surveillance
There was once relatively little information about the growth rate of RCC because almost all renal tumors were excised shortly after detection based on prior paradigms of management (Chawla et al, 2006; Crispen and Uzzo, 2006; Jewett and Zuniga, 2008; Chen et al, 2009). The incidental discovery of many small RCCs in asymptomatic elderly patients or those of poor surgical risk has provided the opportunity to observe the growth rate of these tumors in patients who are unable or unwilling to undergo surgery (Derweesh and Novick, 2003; Chawla et al, 2006; Crispen and Uzzo, 2006; Rendon and Jewett, 2006; Kouba et al, 2007; Abou Youssif et al, 2007; Abouassaly et al, 2008; Jewett and Zuniga, 2008; Klatte et al, 2008b; Campbell et al, 2009; Chen et al, 2009). Bosniak and associates (1996) reported one of the first and largest series of active surveillance (AS) that included 72 small (<3.5 cm) renal tumors in 68 patients who were observed with serial imaging studies for intervals ranging from 2 to 10 years (mean, 3.3 years). On CT these were well-marginated, homogeneous, solid, enhancing tumors consistent with RCC. During the period of observation these tumors grew at slow and variable rates of up to 1.1 cm per year, with a median growth rate of 0.36 cm per year. In 32 patients whose tumors grew to larger than 3 cm, surgical excision was performed; all the excised tumors proved to be stage pT1a RCCs, and the majority were grade 1 tumors. Significantly, none of the patients developed metastasis during the period of surveillance.
Subsequent series from several institutions have confirmed that many small renal masses will grow relatively slowly (median growth rate 0.28 cm/yr), and there have been only 4 patients with metastasis in these series (1.2%), suggesting that this may be a reasonable management strategy in carefully selected patients who are not candidates for conventional surgery or thermal ablative approaches (Chawla et al, 2005, 2006; Crispen and Uzzo, 2006; Rendon and Jewett, 2006; Jewett and Zuniga, 2008; Campbell et al, 2009). However, a critical review of this literature is required to recognize the limitations of this series of studies (Campbell et al, 2009). First, they represent a highly select population; most AS series have included only relatively small, well-marginated, and homogeneous renal masses, a substantial proportion (20% or more) of which may have been benign (Frank et al, 2003b; Crispen and Uzzo, 2006; Lane et al, 2007a). In addition, follow-up in most series is limited to 2 to 3 years and in some cases the growth rate was calculated backward by obtaining old films for which the lesion of interest was either previously missed or dismissed, introducing a strong selection bias (Bosniak et al, 1996; Crispen and Uzzo, 2006; Jewett and Zuniga, 2008). Finally, in most of these series, there is a subpopulation of patients with rapidly growing tumors that appear to have more aggressive characteristics. For instance, in the series from Volpe and colleagues (2004), 25% of the masses doubled in volume in 12 months and 22% reached a diameter of 4 cm, triggering surgical intervention. Similarly, Sowery and colleagues (2004) reported 9 tumors with mean growth rate of 1.43 cm per year, representing a substantial proportion of their patients. Salvage of patients with metastatic RCC is unlikely, and in some patients the window of opportunity for nephron-sparing surgery may be lost. As additional retrospective series are analyzed and prospective datasets compiled, the validity of these concerns will be more rigorously evaluated. All of these concerns would argue in favor of a proactive treatment plan for most patients with masses suspicious for RCC (Crispen and Uzzo, 2006).
Nevertheless, these studies suggest that patients with small, solid, enhancing, well-marginated, homogeneous renal lesions, who are elderly or poor surgical risks, can safely be managed with observation and serial renal imaging at 6-month or 1-year intervals (Chawla et al, 2005; Viterbo et al, 2005; Crispen and Uzzo, 2006; Jewett and Zuniga, 2008; Campbell et al, 2009; Chen et al, 2009). In this population, the risk of competing noncancer causes of death and the risk of intervention will most often outweigh the risk of RCC progression (Hollingsworth et al, 2007). In fact, recent data indicate that active treatment of small renal masses in elderly patients (>75 years of age) may not confer a measurable survival benefit over AS, further supporting a conservative approach in many of these patients (Lane et al, 2009). AS is not appropriate for patients with larger (>3 to 4 cm), poorly marginated, or nonhomogeneous solid renal lesions (Remzi et al, 2006; Kunkle et al, 2007a). AS is also not advisable in younger, otherwise healthy patients with small, solid tumors that have radiographic characteristics consistent with RCC (Campbell et al, 2009). Even if these lesions are smaller than 3 cm, the current data indicate that most will grow and eventually reach a size at which metastasis becomes a possibility. Unfortunately, growth rates on observation do not allow differentiation of benign versus malignant histology (Siu et al, 2007; Crispen et al, 2008d). Therefore, in this setting, it is more appropriate to consider treating the tumor by surgical excision or thermal ablation when it is small, clearly localized, and still amenable to nephron-sparing approaches (Van Poppel et al, 2007; Campbell et al, 2009; Chen et al, 2009).
Clinical T1 Renal Mass: Algorithm for Management
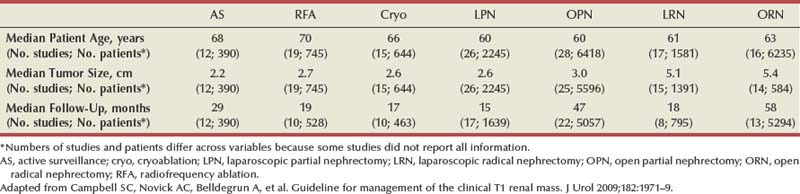
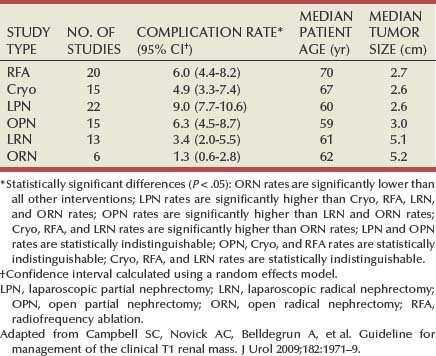
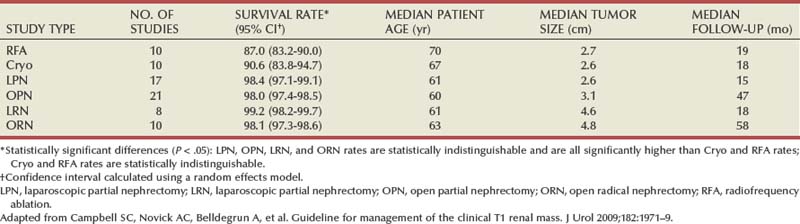
Acknowledging substantial controversy with respect to the management of small renal masses, with some current practices discordant with what the literature supports, the AUA Practice Guidelines Committee organized a panel to review this topic, with the final document published in 2009 (Campbell et al, 2009). This process included a systematic meta-analysis of the literature, and the final document was vetted through an extensive peer review process. As expected, the database for open surgical techniques was most substantial and mature (Table 49–23). In contrast, follow-up for many of the other modalities was rather limited. Review of the data demonstrates strong selection biases, with RN procedures used to treat larger tumors, and AS and TA primarily applied to an older population of patients. There were almost no comparative studies; the overwhelming majority was retrospective and primarily observational. Other limitations of the data are detailed in the final document. The analysis revealed a small number of statistically significant comparisons of consequence for which confounding factors were unlikely to account for differences (Campbell et al, 2009).
One such finding pertained to urologic complications, such as postoperative hemorrhage or urine leak, with PN procedures (laparoscopic and open) associated with the highest rates, likely reflecting the substantial technical challenges associated with these procedures (Table 49–24). This was thought to be a valid finding because PN procedures tended to be applied to younger patients and smaller tumors—patients who would be less likely to have such complications unless the complications were associated with procedural characteristics. A second significant result related to local recurrence, which was defined as any persistent or recurrent disease present in the treated kidney or ipsilateral renal fossa after initial treatment. This definition was adopted from the working group of Image-Guided Tumor Ablation (Goldberg et al, 2005; Campbell et al, 2009). TA procedures had significantly higher rates of local recurrence when compared with all other treatment modalities (Table 49–25). This was also judged to be a valid finding, because these modalities were used to treat relatively small tumors and had short follow-up durations. In reality, it has been estimated that when confounding factors such as length of follow-up are taken into consideration, the local recurrence rates for cryoablation and RFA will be substantially higher than for surgical excision (Kunkle et al, 2008). Many such recurrences can be salvaged with repeat ablation, but when this is not possible, surgical salvage can be very challenging (Kunkle et al, 2008; Nguyen et al, 2008b). Other ongoing concerns with TA were reviewed previously. Analyses of other survival end points, such as metastasis-free, cancer-specific, and overall survival indicated that all such survival rates were relatively high across treatments, reflecting the limited biologic aggressiveness of most clinical T1 renal tumors. Given strong selection biases and highly variable follow-up differences across treatments, comparisons related to these outcomes were not informative (Campbell et al, 2009).
Consensus statements regarding all of the current treatment modalities are provided in Table 49–26. Final management recommendations were framed in terms of each treatment modality’s utility in the context of four index patients defined by tumor size (T1a vs. T1b) and general health (Fig. 49–22). Index patient 1, a healthy patient with a clinical T1a renal mass, is the most commonly encountered scenario. PN is a standard for this patient, with RN an alternate standard that should be applied only when PN is not feasible. One of the main concepts that is emphasized by this guideline document relates to the status of nephron-sparing approaches as an overriding principle for the management of this patient population, presuming that adequate oncologic control can be achieved (Campbell et al, 2009). TA and AS are both options for index patient 1, although there are substantial concerns about these management strategies in this healthy patient. Given the complexity of counseling with such divergent options for management, the panel believed strongly that a urologist should be involved in this process. The panel also strongly advocates research priority for renal mass biopsy with molecular profiling to improve the estimation of tumor aggressiveness and facilitate more rational patient selection in this field (Fig. 49–23).
Table 49–26 Management of the Clinical T1 Renal Mass: AUA Guidelines Consensus Statements
| TREATMENT MODALITY | CONSENSUS STATEMENT |
|---|---|
| Radical nephrectomy (RN) | RN, particularly laparoscopic RN, is very appealing to patients and physicians but is greatly overutilized. Nephron-sparing approaches should be considered in all patients with a clinical T1 renal mass as an overriding principle, presuming adequate oncologic control can be achieved, based on compelling data demonstrating an increased risk of chronic kidney disease (CKD) associated with RN and a direct correlation between CKD and morbid cardiovascular events and mortality on a longitudinal basis. RN is still a viable option when necessary based on tumor size, location, or radiographic appearance if the surgeon judges that nephron-sparing surgery is not feasible or advisable. A laparoscopic approach to RN is now an established standard and should be considered if this procedure is required because it is associated with a more rapid recovery. |
| Active surveillance (AS) | AS is a reasonable option for the management of localized renal masses that should be discussed with all patients and should be a primary consideration for patients with decreased life expectancy or extensive comorbidities that would make them at high risk for intervention. For patients who are candidates for intervention, counseling about AS should include a balanced discussion of the small but real risk of cancer progression, lack of curative salvage therapies if metastases develop, possible loss of window of opportunity for NSS and substantial limitations of the current AS literature. Larger tumors (>3 to 4 cm) and those with aggressive appearance, such as infiltrative growth pattern, may be associated with increased risk and should be managed in a proactive manner. |
| Thermal ablation (TA) | TA (cryoablation or radiofrequency ablation), either percutaneous or laparoscopic, is an available treatment option for the patient at high surgical risk who wants active treatment and accepts the need for long-term radiographic surveillance after treatment. Tumor biopsy (core biopsy is recommended for better diagnostic accuracy) should always be performed prior to treatment to define histology and should also be considered after treatment, particularly if there is any suspicion of recurrence. Counseling about thermal ablation should include a balanced discussion of the increased risk of local recurrence when compared to surgical excision, potential need for reintervention, lack of well-proven radiographic parameters for success, particularly after RFA, potential for difficult surgical salvage if tumor progression is found and the substantial limitations of the current thermal ablation literature. Larger tumors (>3.5 cm) and those with irregular shape or infiltrative appearance may be associated with increased risk of recurrence when managed with thermal ablation. |
| Partial nephrectomy (PN) | Surgical excision by PN is a reference standard for the management of clinical T1 renal masses, whether for imperative or elective indications, given the importance of preservation of renal parenchyma and avoidance of CKD. This treatment modality is greatly underutilized. PN has well established longitudinal oncologic outcomes data comparable to RN. Adequate expertise and careful patient selection are important. A laparoscopic approach can provide more rapid convalescence but has been associated with an increased risk of major urologic complications and longer warm ischemia times when compared with traditional open partial nephrectomy (OPN). In general, OPN is preferred for complex cases such as tumor in the renal hilum, tumor in a solitary kidney, or multiple tumors. |
Adapted from Campbell SC, Novick AC, Belldegrun A, et al. Guideline for management of the clinical T1 renal mass. J Urol 2009;182:1271–9.
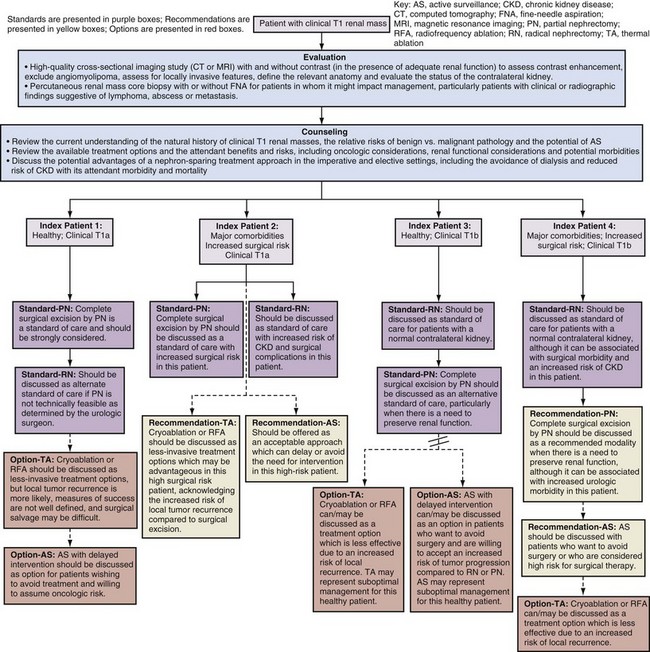
Figure 49–22 Algorithm for the evaluation, counseling, and management of the patient with a clinical T1 renal mass.
(Adapted from Novick AC et al. Guideline for management of the clinical stage 1 renal mass. 〈http://www.auanet.org/content/guidelines-and-quality-care/clinical-guidelines/main-reports/renalmass09.pdf〉; 2009 [accessed 06.01.11].)
Nephron-Sparing Surgery in von Hippel-Lindau Disease and Other Forms of Familial Renal Cell Carcinoma
RCC in von Hippel-Lindau disease differs from its sporadic counterpart in that the diagnosis is made at a young age and there are usually multiple bilateral renal tumors (Maher and Kaelin, 1997; Uzzo and Novick, 1999; Zbar et al, 1999; Grubb et al, 2005a; Nathanson and Stephenson, 2009). Although these are generally low-stage tumors, they are capable of progression to metastasis and represent a frequent cause of death in patients with von Hippel-Lindau disease. RCC in these patients is characterized histopathologically by both solid tumors and renal cysts that contain either frank carcinoma or a lining of hyperplastic clear cells representing incipient carcinoma (Fig. 49–24) (Christenson et al, 1982; Walther et al, 1995; Nathanson and Stephenson, 2008). Therefore, adequate surgical treatment of localized RCC in von Hippel-Lindau disease requires excision and/or ablation of all solid and cystic renal lesions whenever feasible (Grubb et al, 2005a; Nathanson and Stephenson, 2008). Choyke and colleagues (2001) have shown that intraoperative ultrasonography may be a valuable adjunct for this population of patients. In their series this study identified additional tumors in 25% of patients with hereditary renal cancer undergoing renal exploration.
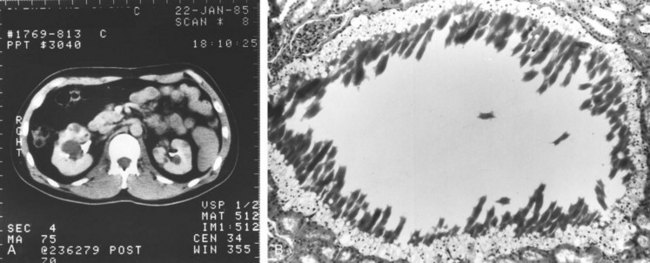
Figure 49–24 A, CT scan after administration of contrast agent shows bilateral solid and cystic renal masses in patient with von Hippel-Lindau disease. B, Histopathologic section of one of the renal cysts shows lining of clear cells representing incipient carcinoma.
The surgical options in patients with bilateral RCC and von Hippel-Lindau disease are bilateral nephrectomy and renal replacement therapy or nephron-sparing approaches such as PN or TA to avoid end-stage renal disease (Herring et al, 2001; Roupret et al, 2003; Drachenberg et al, 2004; Nathanson and Stephenson, 2008). The general philosophy has been to pursue nephron-sparing strategies whenever possible, given the multifocal nature of the disease, even for centrally located tumors (Drachenberg et al, 2004; Grubb et al, 2005a; Nathanson and Stephenson, 2008). Although early results of PN were promising, subsequent studies suggested a high incidence of postoperative tumor recurrence in the remaining portion of the kidney (Novick and Streem, 1992; Grubb et al, 2005a). It is likely that most of these local recurrences were a manifestation of residual microscopic RCC that was not removed at the time of the original PN (Walther et al, 1995; Grubb et al, 2005a).
One multicenter study delineated the long-term outcomes after surgical treatment of localized RCC in 65 patients with von Hippel-Lindau disease managed at eight medical centers in the United States (Steinbach et al, 1995). RCC was present bilaterally and unilaterally in 54 and 11 patients, respectively. RN and PN were performed in 16 and 49 patients, respectively. The mean postoperative follow-up interval was 68 months. The 5-year and 10-year cancer-specific survival rates for all patients were 95% and 77%, respectively. The corresponding rates for patients treated with PN were 100% and 81%, respectively. In the latter group, 25 patients (51%) developed postoperative local recurrence; however, only 2 of these patients had concomitant metastatic disease. Survival free of local recurrence was 71% at 5 years but only 15% at 10 years. Similarly, Roupret and colleagues (2003) reported a 27.4% incidence of local recurrence at mean follow-up of 55.9 months after PN for patients with von Hippel-Lindau disease, confirming that these patients are at much higher risk for local recurrence than are patients with sporadic RCC.
Duffy and colleagues (2004) at the National Cancer Institute have defined a 3-cm threshold for intervention in patients with von Hippel-Lindau disease. In their series, a total of 108 patients with von Hippel-Lindau disease and solid renal tumors smaller than 3 cm were observed a mean of 58 months and none developed metastatic disease. In contrast, metastases developed in 20 of 73 patients (27.4%) with tumors larger than 3 cm and the frequency of metastases increased with increasing tumor size. A 3-cm cut point has thus been proposed to reduce the number of surgical interventions, to optimize renal function, and to minimize the risk of metastatic disease. The authors have emphasized that this recommendation also applies to patients with hereditary papillary RCC and Birt-Hogg-Dubé syndromes (Walther et al, 1999a; Herring et al, 2001; Duffey et al, 2004; Grubb et al, 2005b; Nathanson and Stephenson, 2008). Hereditary leiomyomatosis is an exception in that tumors in this syndrome are more aggressive and should be managed accordingly, even when less than 3 cm (Nathanson and Stephenson, 2008).
Taken together, these studies suggest that PN can provide effective initial treatment of patients with RCC and von Hippel-Lindau disease but should be withheld until tumor size reaches or eclipses 3.0 cm. In recent years, TA has been used more frequently in this patient population as an alternative nephron-sparing approach (Grubb et al, 2005b; Matin et al, 2008; Nathanson and Stephenson, 2008). After initial management, patients with von Hippel-Lindau disease must be observed closely because most will eventually develop locally recurrent RCC with the concomitant need for repeat renal intervention (Campbell and Novick, 1995; Grubb et al, 2005a; Ploussard et al, 2007). In this setting, repeat PN can be challenging because of postoperative fibrosis, and TA may be preferred to reclaim local control (Shingleton and Sewell, 2002; Hwang et al, 2004; Mabjeesh et al, 2004; Matin et al, 2008; Nathanson and Stephenson, 2008; Nguyen et al, 2008b). Targeted agents are now being investigated in an effort to slow disease progression in patients with this syndrome (Grubb et al, 2005a). When removal of all renal tissue is necessary to achieve control of malignant disease, renal transplantation can provide satisfactory replacement therapy for end-stage renal disease and appears to be safe despite the tumor diathesis (Goldfarb et al, 1997).
Comprehensive management of patients suspected of having familial RCC should also include genetic counseling and screening for other manifestations of the disease process (see earlier section on familial RCC and molecular genetics) (Nathanson and Stephenson, 2008). For patients with von Hippel-Lindau disease, identification of pheochromocytoma or central nervous system hemangioblastoma is particularly important before surgical intervention for RCC (Leung et al, 2008; Nathanson and Stephenson, 2008).
Treatment of Locally Advanced Renal Cell Carcinoma
Inferior Vena Caval Involvement
One of the unique features of RCC is its frequent pattern of growth intraluminally into the renal venous circulation, also known as venous tumor thrombus. In extreme cases this growth may extend into the IVC with cephalad migration as far as the right atrium or beyond (Fig. 49–25). The absence of metastases in many patients with vena caval extension is an intriguing aspect of this cancer’s behavior (Gettman and Blute, 2002; Wotkowicz et al, 2008). Forty-five to 70 percent of patients with RCC and IVC thrombus can be cured with an aggressive surgical approach including radical nephrectomy and IVC thrombectomy (see Key Points box). In general, patients with tumor that is otherwise confined to the kidney and nonadherent thrombi have the best prognosis; those with tumor extending into the perinephric fat, lymph node involvement, or direct invasion of the wall of the IVC are at much higher risk for recurrence (Hatcher et al, 1991; Montie et al, 1991; Glazer and Novick, 1996; Gettman et al, 1999; Naitoh et al, 1999; Sweeney et al, 2002b; Bissada et al, 2003; Kim et al, 2004a; Moinzadeh and Libertino, 2004; Leibovich et al, 2005b; Haferkamp et al, 2007; Zini et al, 2008).
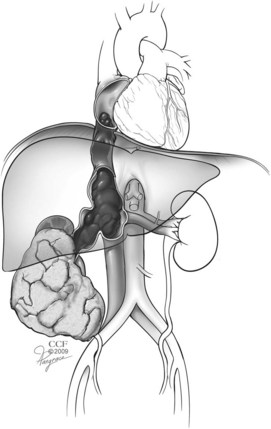
Figure 49–25 Schematic illustrating renal cell carcinoma in the right kidney with tumor thrombus extending through renal vein into the inferior vena cava and terminating in the right atrium.
(Reprinted with permission, Cleveland Clinic Center for Medical Art & Photography © 2007-2009. All Rights Reserved.)
Key Points: Treatment of Locally Advanced RCC
Overall, involvement of the venous system with RCC occurs in 4% to 10% of patients. Venous tumor thrombus should be suspected in patients with a renal tumor who also have lower extremity edema, isolated right-sided varicocele or one that does not collapse with recumbency, dilated superficial abdominal veins, proteinuria, pulmonary embolism, right atrial mass, or nonfunction of the involved kidney. Staging of the level of IVC thrombus is as follows: I, adjacent to the ostium of renal vein; II, extending up to the lower aspect of the liver; III, involving the intrahepatic portion of the IVC but below the diaphragm; and IV, extending above the diaphragm. The prognostic significance of IVC thrombus level has been controversial. Most studies suggest that the incidence of locoregional or systemic progression is higher in patients with level III-IV IVC thrombus, and this probably accounts for the reduced survival reported in this subgroup in some series (Sosa et al, 1984; Quek et al, 2001; Zisman et al, 2003; Kim et al, 2004a; Leibovich et al, 2005b). Others series have shown that any IVC involvement is worse than renal vein involvement without distinction with regard to IVC level; in these series, other factors, such as nodal or metastatic involvement and tumor grade, have more impact on overall survival (Blute et al, 2004b; Terakawa et al, 2007). However, other reports confirm that many patients with level IV IVC thrombi can be cured of disease with surgical resection, as long as the tumor is otherwise confined and other adverse features are absent (Libertino et al, 1987; Glazer and Novick, 1996; Sweeney et al, 2002b; Ciancio et al, 2007; Granberg et al, 2008). The seventh edition of the TNM staging system distinguishes tumors with thrombi above the diaphragm (stage T3c) from those with IVC thrombi below the diaphragm (stage T3b) and those with thrombi only within the renal vein or its branches (stage T3a) (Edge et al, 2010).
MRI is a noninvasive and accurate modality for demonstrating both the presence and the cephalad extent of vena caval involvement and has been the preferred diagnostic study at most centers for the past few decades (Goldfarb et al, 1990; Kallman et al, 1992; Bechtod and Zagoria, 1997; Choyke, 1997; Oto et al, 1998; Sun et al, 1999). Administration of gadolinium during the study often allows tumor thrombi to be differentiated from bland thrombus, because the latter does not demonstrate enhancement. More recent evidence suggests that multiplanar CT can provide essentially equivalent information, including delineation of the cephalad and caudal extent of the thrombus (Fig. 49–26) (Pretorius et al, 2000; Choyke et al, 2001; Bassignani, 2006; Zhang et al, 2007a; Bach and Zhang, 2008; Ng et al, 2008; Herts, 2009). The importance of high-quality preoperative imaging cannot be overemphasized and should be obtained as close as possible to the date of surgery because progression of the tumor thrombus may mandate important changes in intraoperative management (Blute et al, 2004b; Wotkowicz et al, 2008).
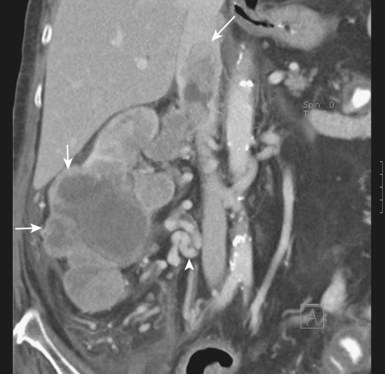
Figure 49–26 CT scan demonstrating lower pole right renal cell carcinoma (small arrows) with level III inferior vena cava (IVC) thrombus. The large arrow indicates the upper extent of the tumor thrombus within the intrahepatic portion of the IVC. The arrowhead indicates extensive retroperitoneal venous collateral vessels associated with restricted flow within the IVC.
Invasive contrast-enhanced imaging is reserved for patients in whom MRI and CT findings are equivocal or for whom MRI and CT are contraindicated. Although inferior venacavography using antegrade and/or retrograde contrast injection remains an accurate diagnostic study for assessment of IVC involvement, this invasive technique is generally unnecessary in the modern imaging era. Some have also advocated the use of renal arteriography as an adjunctive preoperative study in patients with RCC involving the IVC, with distinct vascularization of tumor thrombus observed in 35% to 40% of cases (Fig. 49–27) (Novick et al, 1990; Wotkowicz et al, 2008). When this finding is present, preoperative embolization of the renal artery can be performed in an attempt to shrink the thrombus and facilitate the surgical procedure. However, Subramanian and colleagues (2009) have demonstrated that preoperative renal artery embolization does not provide any measurable benefit in reducing blood loss or complications of nephrectomy with tumor thrombectomy. In their series of 225 patients with IVC thrombi, embolization was associated with a higher risk of perioperative death and a trend toward increasing blood transfusions in multivariable analysis. In patients with extensive supradiaphragmatic vena caval thrombi, when adjunctive cardiopulmonary bypass with deep hypothermic circulatory arrest is considered, coronary angiography can also be performed preoperatively (Novick et al, 1990). If significant obstructing coronary lesions are found, these can be repaired simultaneously during cardiopulmonary bypass. Transesophageal echocardiography is an invasive study that is unnecessary before surgery but can be an important intraoperative diagnostic modality for monitoring the supradiaphragmatic portion of the thrombus if needed (Glazer and Novick, 1997; Wotkowicz et al, 2008).
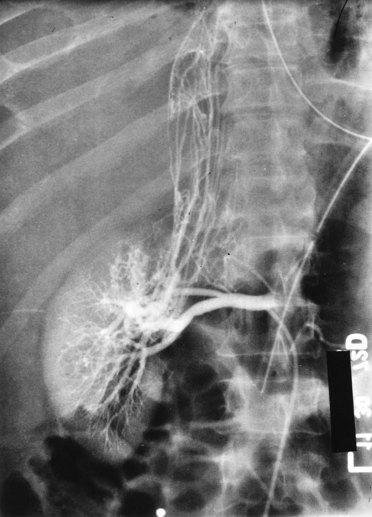
Figure 49–27 Right renal arteriogram shows arterialization of a supradiaphragmatic vena caval thrombus.
Although involvement of the IVC with RCC renders the task of complete surgical excision more complicated, this approach offers the only realistic hope for cure for most patients (Quek et al, 2001; Blute et al, 2004b). After mobilization of the kidney and ligation and transection of the arterial blood supply, vascular control of the involved portion of the IVC is then obtained. In general, level I thrombi are isolated by a Satinsky clamp and are thus readily removed (Fig. 49–28A). Level II thrombi require sequential clamping of the caudal IVC, contralateral renal vasculature, and cephalad IVC along with mobilization of the relevant segment of the IVC and occlusion of lumbar veins. The renal ostium is then opened and the thrombus is removed, all in a bloodless field (see Fig. 49–28B). When tumor thrombus invades the wall of the vena cava, aggressive resection of the involved vena cava and attainment of negative surgical margins are required to minimize the risk of recurrence (Hatcher et al, 1991; Blute et al, 2007; Wotkowicz et al, 2008). IVC grafting or reconstitution is required in some instances, but patients with a completely occluded IVC do not require this because of collateral blood flow (Sarkar et al, 1998; Blute et al, 2007). Distal bland thrombus within the IVC or iliac vessels may be left in situ, although the IVC should be ligated or clipped cephalad to this level to prevent pulmonary embolism.
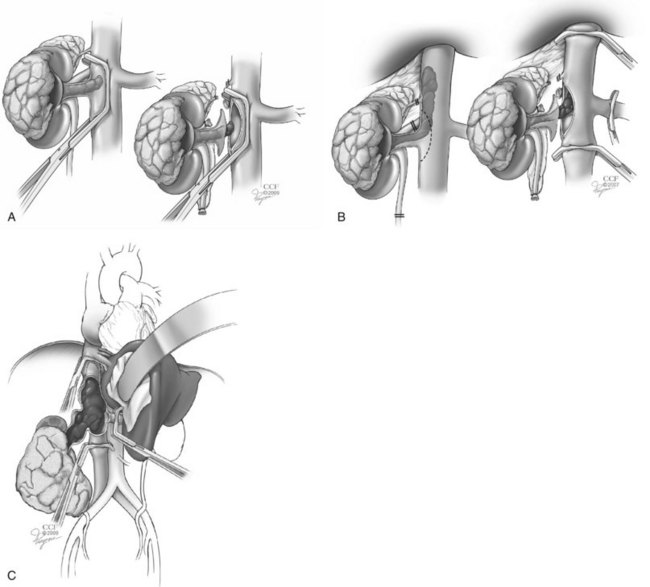
Figure 49–28 Schematic diagrams illustrating surgical techniques for management of inferior vena cava (IVC) thrombi according to level. A, Level I IVC thrombus managed with a Satinsky clamp to achieve vascular isolation. B, Level II IVC thrombus managed by sequential clamping of the lower IVC, contralateral renal vein, and cephalad IVC, along with mobilization of the IVC and occlusion of lumbar veins, allowing for vascular isolation. C, Level III IVC thrombus managed by mobilization of the liver providing exposure of the intrahepatic IVC and retraction of the thrombus to facilitate placement of the upper IVC clamp just below the level of the hepatic veins. In this manner, vascular isolation is achieved in a manner similar to that in B. If the cephalad clamp must be placed above the level of the hepatic veins, a Pringle maneuver should be performed to temporarily occlude the hepatic blood flow.
(Reprinted with permission, Cleveland Clinic Center for Medical Art & Photography © 2007-2009. All Rights Reserved.)
Vascular control for level III and level IV IVC thrombi requires more extensive dissection, venovenous bypass, or cardiopulmonary bypass and hypothermic circulatory arrest. For level III thrombi, mobilization of the liver and exposure of the intrahepatic IVC will often allow the thrombus to be mobilized caudad to the hepatic veins, and venous isolation can then proceed as for a level II thrombus (see Fig. 49–28C) (Gallucci et al, 2004; Ciancio et al, 2007). If this is not possible, the IVC should be clamped above the liver and a Pringle maneuver can be performed whereby the portal triad is temporarily occluded (Burt, 1991; Ciancio and Soloway, 2001, 2002; Vaidya et al, 2003; Jibiki et al, 2004; Ciancio et al, 2007). Venovenous bypass is commonly used in these cases but may not be required if adequate collateral flow is present (Ciancio et al, 2002). Level IV IVC thrombi have traditionally been managed with cardiopulmonary bypass and hypothermic circulatory arrest, and this is still the preferred approach in complex cases (Blute et al, 2007; Wotkowicz et al, 2008). However, many centers are now trying to avoid hypothermic circulatory arrest and the associated hypocoagulable state that ensues after coming off the pump and the increased risk of cerebrovascular accident and myocardial infarction that accompanies this procedure (Shinghal et al, 2003; Ciancio et al, 2007; Subramanian et al, 2009). In this case the thrombus is mobilized below the atrium, allowing sequential vascular control to be achieved without opening the heart. Further detail about these procedures can be found in Chapter 54.
The risk of morbidity can be substantial for thrombi extending above the diaphragm and mortality rates associated with radical nephrectomy and IVC thrombectomy have been reported to be as high as 5% to 10% in some series, depending on the patient’s comorbidities and tumor characteristics (Staehler and Brkovic, 2000; Blute et al, 2004b). Therefore patient selection and surgical planning are of paramount importance (Gettman et al, 2003), and this procedure has for the most part been reserved for patients with good performance status in whom preoperative studies show no substantial lymphadenopathy or systemic metastasis. However, there may be a palliative role for surgery in some patients with metastasis who experience severe disability from intractable edema, ascites, cardiac dysfunction, or associated local symptoms such as abdominal pain and hematuria (Slaton et al, 1997). In addition, nephrectomy and IVC thrombectomy can also be considered in patients with metastatic disease who may be expected to experience a more prolonged disease course after cytoreduction, particularly those with level I or II or readily managed level III IVC thrombi (Zisman et al, 2002b, 2003). Using predictive algorithms to identify patients with poor-risk metastatic RCC, and in consultation with colleagues in medical oncology, the significant morbidity of aggressive surgery should be avoided in those with limited life expectancy.
Locally Invasive Renal Cell Carcinoma
The sequestered location and propensity for aggressive behavior of RCC results in occasional patients presenting with large primary tumors that invade adjacent structures. Patients with pathologic stage T4 disease represent less than 2% of surgical series, but this proportion may increase somewhat with the reclassification of RCC invading directly into the adrenal gland into this category (Thompson et al, 2005a; Karellas et al, 2009). Patients with locally advanced RCC usually present with pain, generally from invasion of the posterior abdominal wall, nerve roots, or paraspinous muscles. Liver extension is uncommon, and intrahepatic metastases occur more often than local extension. The capsules of large tumors may indent and compress adjacent liver parenchyma but seldom actually grow by direct extension into the liver. Margulis and colleagues (2007a) reported that invasion of adjacent organs was confirmed pathologically in only 40% of the patients in whom it was suspected based on preoperative imaging. If tumor extension into the liver is identified intraoperatively, en-bloc partial hepatectomy is rarely curative but may occasionally be worthwhile. Duodenal and pancreatic invasion is uncommon and a poor prognostic sign. The propensity for RCC to parasitize vessels may account for extension into the colon and its mesentery. In evaluation of patients with large, invasive upper quadrant abdominal masses, a broad differential diagnosis should be considered, including adrenocortical carcinoma, infiltrative transitional cell carcinoma, sarcoma, and lymphoma, in addition to locally invasive RCC.
Because surgical therapy is the only potentially curative management for RCC, extended operations with en-bloc resection of adjacent organs are occasionally indicated (see Key Points box). Complete excision of the tumor, including resection of the involved bowel, spleen, or abdominal wall muscles, is the aim of therapy. Margulis and colleagues (2007a) report that even with aggressive surgical management, 10 of 12 patients with pathologic T4 disease experienced disease recurrence at a median of 2 months after surgery. Similarly, although negative surgical margins were achieved in 63% of patients, 34 of 38 patients (90%) in another series ultimately died of disease at a median of 12 months after surgery (Karellas et al, 2009). Important perioperative concerns for patients with locally advanced RCC include a full and proper preoperative consent, including the increased morbidity and mortality of such an approach and the likelihood of concomitant organ resection, a full bowel preparation and consideration for preoperative embolization of the renal arterial blood supply, which may reduce bleeding in cases in which it may be difficult to obtain renal arterial control as one of the initial surgical maneuvers. Vaccination against Streptococcus pneumoniae, Haemophilus influenzae type B, and Neisseria meningitides should be performed preoperatively when splenectomy is likely during nephrectomy (Shatz, 2005; Habermalz et al, 2008). If not already immunized, splenectomized patients should receive these vaccines in the postsurgical setting to prevent sepsis due to one of these encapsulated organisms (Shatz, 2005; Habermalz et al, 2009).
Incomplete excision of a large primary tumor, or debulking, is rarely indicated. In one study, only 12% of patients who underwent incomplete excision of locally extensive tumor were alive at 1 year (DeKernion et al, 1978). Karellas and colleagues (2009) reported similarly poor results in patients with positive margins at the time of nephrectomy for stage pT4 RCC. The same concern holds true during resection of recurrent RCC, in which complete excision is of paramount importance (Eggener et al, 2008; Margulis et al, 2009). Most reports suggest that less than 5% of patients with extension into adjacent viscera (stage pT4) survive 5 years after surgery. Most randomized studies evaluating the role of cytoreductive nephrectomy, including those using immunotherapy (Flanigan et al, 2004) and, more recently, targeted molecular agents, did not include a substantial number of patients with this subcategory of disease, and their conclusions should not be extrapolated to this population of patients. Resection of locally advanced disease can be a major undertaking, and its primary indications should be for curative or palliative intent.
The role of radiation therapy in the treatment of locally extensive RCC is controversial. Several early studies suggested that preoperative radiotherapy could improve survival (Richie, 1966; Cox et al, 1970). A subsequent study by van der Werf-Messing (1973), however, compared results for preoperative therapy with controls and found no survival difference at 5 years. Routine postoperative radiotherapy has not been shown to influence overall survival and can be hazardous because of proximity of small bowel, which is highly radiosensitive. When tumor is known to have been left behind in the renal fossa or adjacent structures, postoperative radiotherapy may occasionally retard progression of the tumor mass (Kao et al, 1994). This approach is rarely used today and has largely been replaced by the use of systemic agents for residual or locally recurrent disease.
Local Recurrence after Radical Nephrectomy or Nephron-Sparing Surgery
Local recurrence of RCC after radical nephrectomy, which includes recurrence in the renal fossa, ipsilateral adrenal gland, or ipsilateral retroperitoneal lymph nodes, is an uncommon event, occurring in 2% to 4% of cases (see Key Points box) (Uson, 1982; Sandock et al, 1995; Levy et al, 1998; Ljungberg et al, 1999; Itano et al, 2000; Schrodter et al, 2002; DiMarco et al, 2004; Stephenson et al, 2004a; Master et al, 2005; Margulis et al, 2009). Risk factors include locally advanced or node-positive disease and adverse histopathologic features (Esrig et al, 1992; Sandock et al, 1995; Levy et al, 1998). In contrast, local recurrence after radical nephrectomy is rare in patients with low-stage pT1-2N0M0 RCC. Only about 40% of local recurrences are isolated; the majority of patients with local recurrence also have systemic disease, and a thorough metastatic evaluation should be pursued (Uson, 1982; Phillips and Messing, 1993; Sandock et al, 1995; Itano et al, 2000; Schrodter et al, 2002; Eggener et al, 2008).
Surgical resection of isolated local recurrence of RCC after radical nephrectomy should be considered, because it can provide long-term cancer-free status for 30% to 40% of patients (Esrig et al, 1992; Tanguay et al, 1996; Itano et al, 2000; Schrodter et al, 2002; Wiesner et al, 2002; Gogus et al, 2003; Master et al, 2005; Bandi et al, 2008; Margulis et al, 2009). An algorithm incorporating risk factors that predicts survival in patients with systemic metastases (Motzer criteria) can predict survival in this group of patients; 2-year overall survival rates for low-, intermediate-, and high-risk patients are 88%, 51%, and 11%, respectively (Eggener et al, 2008). Importantly, feasibility of resection proved to be another independent favorable prognostic parameter in this patient population (Eggener et al, 2008). Complete resection of abdominal recurrences is often a formidable task because the natural tissue barriers are no longer present and invasion of contiguous organs is common. En-bloc resection of adjacent organs is often required, and the risk of morbidity can be substantial (Esrig et al, 1992; Tanguay et al, 1996; Itano et al, 2000; Wiesner et al, 2002; Gogus et al, 2003; Eggener et al, 2008; Margulis et al, 2009). Margulis and colleagues (2009) have reported on 54 patients with local recurrence after nephrectomy managed by surgical resection, 69% of whom also received adjunctive systemic therapy. Risk factors associated with an increased risk of cancer-specific death after resection included recurrent tumor size, sarcomatoid features in the recurrence specimen, positive surgical margin, abnormal alkaline phosphatase, and increased lactate dehydrogenase. Patients with 0, 1, and greater than 1 adverse risk features demonstrated cancer-specific survival times of 111, 40, and 8 months, respectively. Cancer-specific survival after resection of isolated retroperitoneal lymph node recurrence after nephrectomy is longer than after nephrectomy for node-positive disease, reflecting the aggressive biology of RCC with synchronous nodal metastases (Boorjian et al, 2008). Intraoperative radiation was not found to be of oncologic benefit (Master et al, 2005), but radiation therapy may be of value for palliation of symptomatic local recurrence in patients who are not operative candidates.
Local recurrence in the remnant kidney after PN for RCC has been reported in 1.4% to 10% of patients, and the main risk factor is advanced T stage (Campbell and Novick, 1994; Lane and Gill, 2007; Krambeck et al, 2008). Most of these local recurrences are distant from the tumor bed and are thus probably a result of unrecognized tumor multicentricity or de novo occurrence rather than true treatment failure (Campbell and Novick, 1994; Lane and Gill, 2007; Krambeck et al, 2008). Recent data confirm that local recurrence after PN is uncommon, even with a positive surgical margin (Permpongkosol et al, 2006; Kwon et al, 2007; Kutikov et al, 2008b; Yossepowitch et al, 2008). Patients with isolated local recurrence after PN can be considered for repeat PN, completion nephrectomy, TA, or AS (Bratslavsky et al, 2008; Johnson et al, 2008; Magera et al, 2008; Berger et al, 2009). TA is now being used more frequently in this setting because of concern about fibrosis within the renal fossa, but conventional PN can and should be performed when anatomy and tumor characteristics are amenable (Gittes and Blute, 1982; Moll et al, 1993; Campbell and Novick, 1994; Frank et al, 2005a). Regardless, the histopathologic features of the resected tumor and the disease-free interval since initial surgery should be considered along with the presence of CKD and other comorbidities during this decision process, because preservation of renal function may have greater impact on survival than cancer extirpation in certain patients. For this reason, biopsy of the recurrent tumor may be of value when nonextirpative options are being considered.
Local recurrence after thermal ablation often represents treatment failure; many local recurrences develop within the previous tumor bed (McDougal et al, 2005; Matin et al, 2006; Levinson et al, 2008; Nguyen et al, 2008b; Berger et al, 2009). Reported incidences range between 3% and 10% for cryoablation and between 5% and 20% for radiofrequency ablation (McDougal et al, 2005; Gill et al, 2006; Matin et al, 2006; Weight et al, 2008; Levinson et al, 2008; Kunkle and Uzzo, 2008; Kunkle et al, 2008; Berger et al, 2009). Diagnosis of local recurrence in this setting is established by the demonstration of persistent central enhancement within the tumor bed or an enhancing lesion at any other site within the ipsilateral kidney (Weight et al, 2007; Berger et al, 2009). A biopsy can provide more definitive information. Importantly, the true incidence of local recurrence in this population is not well defined because radiographic factors predictive of recurrence have been challenged, particularly for RFA, most studies have not incorporated routine biopsy of the tumor bed during surveillance, and length of follow-up in many series is limited (Weight et al, 2007; Stein et al, 2007). Most of these recurrences can be managed with repeat ablation, but this is not always feasible. Beyond this, management options are similar to those for recurrence after PN, with the caveat that salvage surgery in this setting is often challenging owing to the dense inflammatory reaction induced by thermal ablation (Kowalczyk et al, 2008; Nguyen et al, 2008b).
Adjuvant Therapy for Renal Cell Carcinoma
Unfortunately, recurrence develops in a significant proportion of patients thought to be rendered disease free after surgical resection, primarily due to occult micrometastatic disease. Although postsurgical recurrence is not common in patients with low-stage, organ-confined disease, locally advanced RCC and RCC with other adverse histopathologic features carry a significant risk of recurrence. Various predictive tools can assist in the assessment of the risk in individual patients (Zisman et al, 2002c; Leibovich et al, 2003a), although on the whole, distant metastases develop in 20% to 35% and local recurrence in 2% to 5% of patients (Rabinovitch et al, 1994; Lane and Kattan, 2008). In view of these findings, a strong rationale for systemic postoperative adjuvant therapy exists in high-risk patients. However, almost all adjuvant studies in this field have been negative thus far, with a few highly controversial exceptions, and the standard in this field remains observation if the patient will not consider an adjuvant trial (see Key Points box).
Key Points: Treatment of Local Recurrence and Adjuvant Therapy for Renal Cell Carcinoma
Clinical end points used in adjuvant trials include recurrence-free survival, time to recurrence, and overall survival. Ideal trial design incorporates placebo-controlled and blinded protocols and preferably independent radiographic review to confirm that all entered patients are truly disease free at study entry and to confirm and accurately time all recurrences. Intent to treat analysis is another strong expectation in this field. Until recently, many adjuvant trials performed in patients with RCC have been underpowered to detect small differences in survival and primary end points related to tumor recurrence have been difficult to assess in view of flaws in study design. A variety of adjuvant approaches have been investigated, including hormonal manipulation, radiotherapy, immunotherapeutics, vaccines, and, most recently, targeted molecular agents. Early adjuvant trials tested postoperative medroxyprogesterone acetate (Pizzocaro et al, 1987) or perioperative radiotherapy (van der Werf-Messing, 1973; Kjaer et al, 1987) with negative results.
Despite demonstrable antitumor effects in patients with metastatic disease, IL-2 and interferon alfa did not prove to be beneficial in the adjuvant setting. Four randomized studies were performed, three with interferon alfa and one with IL-2 (Porzolt et al, 1992; Pizzocaro et al, 2001; Messing et al, 2003; Clark et al, 2003). The studies investigating interferon used various doses, preparations (L-interferon, interferon alfa-2a, interferon alfa-2b), and duration of therapy (6 to 12 months); none demonstrated a benefit when compared with controls. Clark and colleagues (2003) randomized patients to observation versus high-dose IL-2 (600,000 units/kg every 8 hours on days 1 to 5 and 15 to 19). A total of 69 patients were registered, 44 with locally advanced and/or nodal disease and 25 with completely resected metastatic disease. An interim analysis prompted closure of the trial primarily because of the toxicity of high-dose IL-2; there was no clinically meaningful benefit for the population as a whole or in either subgroup. Given the limited usage of immunotherapy in patients with metastatic disease at present, it seems unlikely that further adjuvant trials of immunotherapy will be conducted in the near future.
A variety of autologous tumor vaccine–based approaches have been used to immunize RCC patients in the postoperative setting, with largely negative results (Table 49–27). The most controversial results came from a prospective trial in which 553 patients were apparently randomized to observation or intradermal treatment with an autologous vaccine (Jocham et al, 2004). The final report included 202 patients on observation and 177 on active treatment, and this cohort of 379 patients was referred to as the intent-to-treat group by the investigators, although biases may have played a role in this determination. At 5 years, the recurrence-free survival rates were significantly improved for the vaccine-treated group (P = .0204). However, in addition to concern about the legitimacy of its intent to treat analysis, this trial was not blinded, did not include a placebo control, and did not undergo external review of data; therefore, this finding has been viewed by experts in the field with some degree of skepticism.
The largest adjuvant clinical trial for patients with high-risk localized RCC, the Oncophage trial, has recently been completed, reporting no benefit with an autologous tumor vaccine (Wood et al, 2008). Patients with clear cell histology meeting entry criteria after nephrectomy were randomized to undergo observation versus intradermal injections of autologous heat-shock protein HSPPC-96 (Vitespan/Oncophage). HSPPC-96 binds peptide fragments derived from the patient’s own tumor and can act as a chaperone for antigen presentation; a dose of 25 mg was administered weekly for 4 weeks, then once every 4 weeks until vaccine depletion (Wood et al, 2004). More than 2000 patients were evaluated, and 728 were analyzed by intention-to-treat (Wood et al, 2008). No patient experienced grade 3 or 4 treatment-related toxicity. A placebo arm was not incorporated, but an independent review was performed and determined time to progression. Unfortunately, it appears that HSPPC-96 tumor vaccine does not influence the natural history of RCC in the adjuvant setting.
Current trials using targeted molecular agents with activity in patients with metastatic RCC, such as sunitinib and sorafenib, have enrolled a large number of patients with surgically resected RCC who are at high risk for recurrence (Table 49–28) (Jonasch and Tannir, 2008). These agents are orally administered and thus their use is appealing in an adjuvant setting, although there are many uncertainties regarding optimal choice and dose and duration of therapy. In addition, the toxicities of these agents may limit their usefulness in the absence of demonstrable metastatic disease because patients taking a potentially preventive treatment tend to have a much lower threshold for toxicities. Please refer to Chapter 50 for a more detailed discussion of these tyrosine kinase inhibitors and their rationale for patients with advanced RCC.
Other Malignant Renal Tumors
Sarcomas of the Kidney
Sarcomas represent 1% to 2% of all malignant renal tumors in adults, with a peak incidence in the fifth decade of life (Srinivas et al, 1984; Russo, 1991; Spellman et al, 1995; Frank et al, 2000). Renal sarcoma is less common but more lethal than sarcoma of any other genitourinary site, including the prostate, bladder, and paratesticular region (Russo, 1991; Russo et al, 1992). Differentiation of renal sarcoma from sarcomatoid RCC is often difficult on the basis of clinical presentation, radiographic findings, and, in some cases, pathologic analysis. Identification of any features of the various subtypes of RCC excludes the diagnosis of primary renal sarcoma. The common signs and symptoms associated with renal sarcoma in adults include palpable mass, abdominal or flank pain, and hematuria and are similar to those seen with large, rapidly growing RCCs (Economou et al, 1987). Abdominal CT typically demonstrates a large soft tissue mass involving or derived from the kidney, a finding that is again similar to that associated with many sarcomatoid RCCs (Farrow et al, 1968; Shirkhoda and Lewis, 1987; Russo, 1991). Specific findings suggestive of sarcoma rather than of RCC include apparent origin from the capsule or perisinuous region, growth to large size in the absence of lymphadenopathy, presence of fat or bone suggestive of liposarcoma or osteosarcoma, and hypovascular pattern on angiography, although one notable exception is the hemangiopericytoma, which is highly vascular (Smullens et al, 1982; Shirkhoda and Lewis, 1987). Renal sarcoma should be suspected in any of these circumstances or in any patient with a very large or rapidly growing renal mass (Table 49–29).
Table 49–29 Characteristics of Other Malignant Renal Tumors
| TUMOR TYPE | CHARACTERISTICS | MANAGEMENT |
|---|---|---|
| Sarcomas | ||
| Renal lymphoma and leukemia | ||
| Metastatic tumors | ||
| Carcinoid | ||
| Small cell carcinoma of the kidney | ||
| Primitive neuroectodermal tumor | ||
| Wilms tumor |
Sarcomas of the kidney, like sarcomas of any other site, share in common a distinct tumor biology that has important implications with respect to management (reviewed in Russo, 1991). These tumors are derived from mesenchymal components and are thus free of many of the natural barriers to dissemination that confine other tumor types. They are typically surrounded by a pseudocapsule that is readily identifiable but that cannot be relied on for surgical dissection because it is often infiltrated with cancer cells, which can extend for some distance into the surrounding tissues. In many cases this cannot be recognized macroscopically, although it is often manifested in the form of local recurrences, which are common after surgical extirpation, even when a wide excision has been performed. High-grade sarcomas often metastasize, with the lungs being a primary site of spread, and prognosis is poor; many patients die of disease progression in a matter of months. Low-grade sarcomas tend to pursue a more indolent course, although local recurrences often require repeat resection to prolong survival and minimize morbidity.
In general, the most important prognostic factors for sarcomas are margin status and tumor grade. The initial resection is the key event because, for many patients, this is their best chance for a long-term cure. For renal sarcomas this often mandates radical nephrectomy along with en bloc excision of adjacent organs (Srinivas et al, 1984; Brescia et al, 2008). MRI can be useful for preoperative planning by defining tissue planes and proximity to vital structures. This is primarily a surgical disease, and wide excision is the goal with intraoperative monitoring of margin status. Chemotherapeutic agents that have demonstrated activity against metastatic sarcomas include doxycycline and ifosfamide, but even in the best of circumstances response rates are disappointing (Antman et al, 1993). The combination of radiation therapy and chemotherapy, which has proved effective in an adjuvant setting for the management of sarcomas of the extremity, has not provided much benefit for renal or retroperitoneal sarcomas (Russo, 1991). At present, the role of such adjuvant approaches for the management of renal sarcomas is not well defined, although a multimodal approach is often pursued if performance status allows, given the poor prognosis.
The largest single-institution series of renal sarcomas include only 15 to 26 cases and represent a composite experience extending for a period of several years (Farrow et al, 1968; Srinivas et al, 1984; Shirkhoda and Lewis, 1987). In all such series leiomyosarcoma has been the most common histologic subtype, and in many series liposarcoma has been the second most common. In contrast, for retroperitoneal sarcomas, the order is reversed, with liposarcoma being the most common histologic subtype (Karakousis et al, 1995). All such series report a poor prognosis; the experience of Srinivas and colleagues (1984) at Memorial Sloan-Kettering Cancer Center is representative. In this series of 16 patients with renal sarcomas, 15 underwent nephrectomy, often with en-bloc excision of adjacent organs; 5 received adjuvant radiation therapy and chemotherapy without apparent benefit; and 13 died within 6 months after surgery. Vogelzang and associates (1993) reported a more encouraging experience with 21 patients with primary renal sarcomas; there were six long-term survivors after surgery alone. A predominance of low-grade disease may account for these favorable outcomes. However, even in this series, the majority of patients died of progressive disease within a few to several months after diagnosis. Saitoh and colleagues (1982) defined the common sites of metastases of renal sarcomas: lung first and foremost but also lymph nodes and liver.
Leiomyosarcoma is the most common histologic subtype of renal sarcoma, accounting for 50% to 60% of such tumors (Fig. 49–29). The cell of origin is the smooth muscle cell of the capsule or other perinephric structures (Moudouni et al, 2001; Deyrup et al, 2004). Niceta and associates (1974) identified 66 cases of renal leiomyosarcoma in the literature and reported a female predominance; most patients presented in the fourth through sixth decades of life. Renal leiomyosarcoma, like other renal sarcomas, tends to displace rather than invade the parenchyma, and is characterized by rapid growth rate, frequent metastasis, and high local and systemic recurrence rates (Pollack et al, 1987; Deyrup et al, 2004). In the cases reviewed by Niceta, most patients were treated primarily with radical nephrectomy and died within 2 years. In the Mayo Clinic series, 14 of 15 patients with renal leiomyosarcoma died of disease progression within 4 months to 5.5 years after surgery (Farrow et al, 1968).
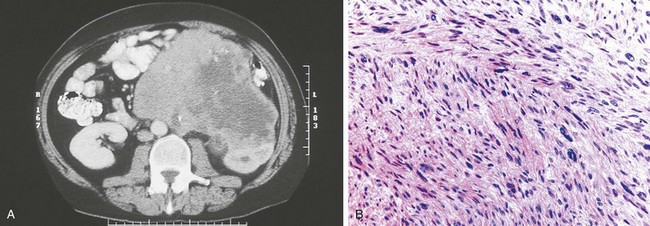
Figure 49–29 A, CT scan demonstrates a large leiomyosarcoma of the left kidney. B, Microscopic features of leiomyosarcoma include spindle cells, blunt-ended nuclei, and eosinophilic cytoplasm.
(Courtesy of Dr. Michael McGuire, Evanston, IL, and Dr. Ming Zhou, Cleveland, OH.)
Other than leiomyosarcoma, a wide variety of histologic subtypes have been described because almost every conceivable type of sarcoma has been found in the kidney. Liposarcoma is readily distinguished from RCC because of the presence of adipose tissue, but it is often confused with AMLs or with large, benign renal lipomas (Frank et al, 2000). Renal liposarcoma typically develops in the fifth and sixth decades of life and often grows to extremely large size (Economou et al, 1987). Response to radiation therapy and cisplatin-based chemotherapy in an adjuvant setting has been reported by Belldegrun and deKernion (1987) and should be considered in patients with high-grade disease or positive margins. Osteogenic sarcoma is a rare but distinctive form of renal sarcoma that contains calcium and is often rock hard (Micolonghi et al, 1984; Leventis et al, 1997). Extensive calcification in a large, hypovascular tumor should suggest the diagnosis. The appearance on plain films can mimic a staghorn calculus, but the readily evident mass effect should suggest xanthogranulomatous disease or, more rarely, osteogenic sarcoma. Again, prognosis is poor; most patients die of disease progression within a few years after diagnosis. Less common histologic subtypes include rhabdomyosarcoma, fibrosarcoma, carcinosarcoma, malignant fibrous histiocytoma, synovial sarcoma, schwannoma, angiosarcoma, and malignant hemangiopericytoma (Raghavaiah et al, 1979; Weiss et al, 1984; Mordkin et al, 1997; Tsuda et al, 1997; Cerilli et al, 1998; Merchant et al, 1998; Chieng et al, 1999; Koyama et al, 2001; Bella et al, 2002; Chen et al, 2003; Shannon et al, 2005; Khan et al, 2006; Leggio et al, 2006; Hung et al, 2008; Mirza et al, 2008). Malignant hemangiopericytomas present at a mean age of 40.3 years, which is younger than most other renal sarcomas, and are notable for their extensive vascularity (Argyropoulos et al, 2005; Chaudhary et al, 2007; Brescia et al, 2008). Preoperative angioembolization has been described and may simplify surgical excision (Smullens et al, 1982).
Renal Lymphoma and Leukemia
Renal involvement with hematologic malignant neoplasms, which include the lymphomas and leukemias, is common—found at autopsy in approximately 34% of patients dying of progressive lymphoma or leukemia. However, these processes are uncommonly seen in clinical practice because they are often silent and generally occur only as a late manifestation of systemic disease (Richmond et al, 1962; Pollack et al, 1987; McVary, 1991). The role of the urologist in the evaluation of renal lymphoma or leukemia is critically important and can include differentiation from other renal malignant neoplasms, timely provision of a pathologic diagnosis, and preservation of renal function (reviewed in McVary, 1991). Renal involvement is more common with non-Hodgkin’s lymphoma than with Hodgkin’s disease, and, as with most other forms of extranodal non-Hodgkin’s lymphoma, histologically diffuse forms predominate over nodular forms (Richmond et al, 1962; Pollack et al, 1987; Yasunaga et al, 1997; O’Riordan et al, 2001). Primary renal lymphoma is rare, with only a few well-documented case reports in the literature (Kandel et al, 1987; Pollack et al, 1987; Arranz-Arija et al, 1994; Dimopoulos et al, 1996; Yasunaga et al, 1997; Tuzel et al, 2003; Zomas et al, 2004; Ahmad et al, 2005; Garcia et al, 2007). This is not surprising given the relative paucity of lymphoid tissue in the normal renal parenchyma. Hematogenous dissemination of lymphoma to the kidney is most common and is thought to occur in 90% of cases; direct extension from retroperitoneal lymph nodes accounts for the remainder. Hartman and colleagues (1982) have correlated pathologic and radiographic patterns and have shown that the most common pattern of renal involvement consists of multiple small renal nodules that tend to develop between the individual nephrons. Eventually, these nodules become confluent, forming radiographically detectable masses. At the extreme, they can replace the entire parenchyma of the kidney, leading to renal failure.
The CT scan is the radiographic modality of choice for the diagnosis of renal lymphoma and for monitoring response to therapy (Jafri et al, 1982; Pollack et al, 1987; Sheeran and Sussman, 1998; Urban and Fishman, 2000). The common radiographic patterns associated with renal lymphoma have been defined by Heiken and associates (1991) and confirmed by a number of other investigators (Table 49–30). Renal lymphoma can present as multiple distinct renal masses; as a solitary renal mass, which can be difficult to differentiate from RCC; as diffuse renal infiltration; or as direct invasion of the kidney from enlarged retroperitoneal nodes (Cohan et al, 1990; Heiken et al, 1991; Eisenberg et al, 1994; Sheeran and Sussman, 1998; Sheth et al, 2006). A hypovascular pattern on angiography is typical for renal lymphoma (Pollack et al, 1987). Renal lymphoma should be suspected in patients with massive retroperitoneal lymphadenopathy, splenomegaly, or lymphadenopathy in other regions of the body or atypical regions within the retroperitoneum (Jafri et al, 1982; Dimopoulos et al, 1996). Relative to this, the main landing zones for RCC should be kept in mind—the interaortocaval region for right RCC and para-aortic region for left RCC—and lymphadenopathy centered outside of these areas should raise suspicion for lymphoma. Any patient with a prior history of lymphoma and a renal mass should also be evaluated for renal recurrence rather than for RCC. In general, lymphomas are more common in patients with iatrogenic immune suppression, acquired immunodeficiency syndrome (AIDS), autoimmune diseases, or graft-versus-host disease and in patients with a history of radiation therapy (McVary, 1991). These clinical associations may also increase the index of suspicion about a diagnosis of systemic lymphoma.
Table 49–30 CT Findings Associated with Lymphoma
| FINDING | INCIDENCE (%) |
|---|---|
| Multiple renal masses | 45 |
| Solitary renal mass | 15 |
| Renal invasion from enlarged retroperitoneal lymph nodes | 25 |
| Diffuse renal involvement | 10 |
| Predominantly perinephric involvement | 5 |
Data from Pollack HM, Banner MP, Amendola MA. Other malignant neoplasms of the renal parenchyma. Semin Roentgenol 1987;22:260–74; and Heiken JP, Gold RP, Schnur MJ. Computed tomography of renal lymphoma with ultrasound correlation. J Comput Assist Tomogr 1991;1:245–50.
Renal involvement related to leukemia is more common in children, paralleling the demographics of the disease, and is more commonly due to lymphocytic leukemia than the myelogenous forms (Araki, 1982; Pollack et al, 1987). Leukemia typically involves the kidney in a diffusely infiltrative pattern and most often represents a late manifestation of systemic disease. However, as for renal lymphoma, a variety of other radiographic patterns have been observed, as reviewed by Pollack and colleagues (1987).
If lymphoma or leukemic renal involvement is suspected, consideration should be given to percutaneous biopsy to obtain a pathologic diagnosis; and if exploratory surgery is necessary, intraoperative biopsy and frozen-section analysis should take priority. If lymphoma or leukemia is confirmed, the kidney should be spared and complete staging of the neoplasm should be performed to preclude the need for a secondary procedure (McVary, 1991). Truong and colleagues (2001) have demonstrated the utility of fine-needle aspiration in the diagnosis of renal lymphoma, although additional tissue may be required in many cases to classify the lymphoma accurately and to guide further therapy (Hunter et al, 2006). Extirpative surgery should be avoided if renal lymphoma and leukemia are suspected because the treatment of these processes is generally systemic chemotherapy with or without radiation therapy. The classic chemotherapy regimen for non-Hodgkin’s lymphoma is the CHOP protocol, which includes cyclophosphamide, doxorubicin, vincristine, and prednisolone (Colevas et al, 2000). Nephrectomy is seldom indicated except in patients with severe symptoms, such as uncontrollable hemorrhage. The other notable exception is the extremely rare patient with primary renal lymphoma in whom a combination of nephrectomy and systemic chemotherapy may represent optimal therapy (Okuno et al, 1995; Garcia et al, 2007). Fourteen cases of marginal zone B-cell lymphoma (MALT) localized to the kidney have been described with some apparently cured by surgery alone (Garcia et al, 2007).
Both renal lymphoma and leukemia are commonly silent but can be associated with hematuria, flank pain, or progressive renal failure. Fever, weight loss, and fatigue, the so-called B symptoms of lymphoma, are much more common (Yasunaga et al, 1997; Zomas et al, 2004). Renal failure can be due to extensive replacement of the functioning parenchyma or bilateral ureteral obstruction associated with enlarged retroperitoneal lymph nodes (more common with lymphoma) (reviewed in McVary, 1991). In reality, renal failure in such patients is more often related to medical causes, such as hypercalcemia or urate nephropathy, that can develop during systemic treatment of advanced disease. The diagnosis of renal failure related to parenchymal replacement by lymphoma or leukemia requires exclusion of other related causes of renal failure and demonstration of improvement in renal function after administration of systemic chemotherapy or radiation therapy (Choi et al, 1997; O’Riordan et al, 2001).
Metastatic Tumors
Metastatic tumors are the most common malignant neoplasms in the kidney, outnumbering primary renal tumors by a wide margin. Autopsy studies have shown that 12% of patients dying of cancer have renal metastases, making the kidney one of the most common sites for metastatic dissemination (Pollack et al, 1987). The profuse vascularity of the kidney makes it a fertile soil for the deposition and growth of cancer cells. Almost all renal metastases develop through a hematogenous route of spread; only a small minority are caused by direct invasion of tumors derived from adjacent organs such as the pancreas, colon, and adrenal gland. The most common sources of renal metastases include lung, breast, and gastrointestinal cancers; malignant melanoma; and the hematologic malignant neoplasms, which are reviewed in the preceding section (Klinger, 1951; Choyke et al, 1987; Pollack et al, 1987; Aron et al, 2004; Martino et al, 2004; Sánchez-Ortiz et al, 2004b; Stage et al, 2005). Of the solid malignant neoplasms, lung cancer is most commonly associated with renal metastases. Olsson and colleagues (1971) found that 20% of patients dying of lung cancer had renal metastases, 60% of which were bilateral. Klinger (1951) reviewed 5000 autopsies and found 17 cases of renal metastases from lung cancer, 11 from gastric cancer, 9 from breast cancer, 7 from pancreatic cancer, 4 from esophageal cancer, 6 from other gastrointestinal primary cancers, and 1 from malignant melanoma. In a series of 27 renal metastases reviewed by Choyke and associates (1987), 7 patients had primary lung cancer, 7 had gastrointestinal malignant neoplasms, 4 had malignant melanoma, 2 had breast cancer, and 2 had endometrial carcinoma. Most renal metastases are multifocal, and almost all are associated with widespread nonrenal metastases (Choyke et al, 1987; Pollack et al, 1987). Choyke and associates (1987) reported that renal metastases from lung, breast, and colon carcinomas are notable because they are occasionally large and solitary, making them difficult to differentiate from RCC.
The typical pattern of renal metastases consists of multiple small nodules that are often clinically silent, although they can lead to hematuria or flank pain in exceptional circumstances (Walther et al, 1979; Pollack et al, 1987). CT is the primary diagnostic modality for renal metastases and typically demonstrates isodense masses that enhance only moderately (5 to 30 HU) after administration of intravenous contrast material (Pollack et al, 1987; Ferrozzi et al, 1997).
Renal metastases should be suspected in any patient with multiple renal lesions and widespread systemic metastases or a history of nonrenal primary cancer. If there is any uncertainty about the diagnosis, a CT- or ultrasound-guided percutaneous renal biopsy usually provides pathologic confirmation (Gattuso et al, 1999; Sánchez-Ortiz, 2004a). Most patients with renal metastases are managed with systemic therapy or placed on a palliative care pathway, depending on the clinical circumstances. Nephrectomy is almost never required except in extenuating circumstances, such as uncontrollable renal hemorrhage. Patients with a solitary, strongly enhancing renal lesion and a history of organ-confined, nonrenal malignant disease are more likely to have RCC, particularly if the interval between the two diagnoses is substantial. In one study involving 100 consecutive patients with a renal mass and a history of nonrenal malignancy, none of the 54 patients without other evidence of disease progression had a renal metastasis (Rybicki et al, 2003; Sánchez-Ortiz, 2004a). However, even in this circumstance, a renal mass biopsy or aspiration should be considered because it may preclude the need for surgical intervention.
Other Malignant Tumors of the Kidney
Other malignant tumors of the kidney include adult Wilms tumor and neuroendocrine tumors including renal carcinoid, small cell carcinoma, and primitive neuroectodermal tumor (PNET). All are relatively uncommon, but each has distinct tumor biology.
Carcinoid tumors arise from neuroendocrine cells, which are not normally present in the kidney (Romero et al, 2006). This is thus a rare renal malignant neoplasm with fewer than 60 cases reported in the English literature (Kawajiri et al, 2004; Mufarrij et al, 2005; Romero et al, 2006; Hansel et al, 2007; Lane et al, 2007b; Canacci et al, 2008). Primitive stem cells within the kidney or retained neural crest tissue may give rise to these tumors (Krishnan et al, 1997; Begin et al, 1998). A correlation with horseshoe kidneys has been reported, with previous studies showing an increased relative risk of 82-fold compared with normal kidneys (Begin et al, 1998; Romero et al, 2006). Carcinoid tumors stain positive for markers of neuroendocrine tissue such as neuron-specific enolase and chromogranin (Kulke and Mayer, 1999; Lane et al, 2007b). Measurement of urinary or plasma serotonin or its metabolites can be diagnostic (Kulke and Mayer, 1999). Only a minority of patients will present with the carcinoid syndrome—episodic flushing, wheezing, and diarrhea (Jensen and Doherty, 2001; Romero et al, 2006; Lane et al, 2007b). Median age at diagnosis is 49 years (Romero et al, 2006). CT findings are nonspecific, and many renal carcinoids are small and nonaggressive. However, in a recent review of renal carcinoids, metastases were found in 46% of patients at diagnosis (Romero et al, 2006). Surgical excision is the mainstay of treatment (Kawajiri et al, 2004). Nephron-sparing surgery is preferred if the diagnosis is suspected preoperatively. Prognosis is good, particularly when associated with a horseshoe kidney (Krishnan et al, 1997; Begin et al, 1998; Lowrance et al, 2006). Significant adverse prognostic factors include age older than 40 years, tumor size greater than 4 cm, high mitotic rate, purely solid gross morphology, metastasis at initial diagnosis, and tumor extending through the renal capsule (Romero et al, 2006).
Other neuroendocrine tumors including small cell carcinoma and large cell neuroendocrine carcinoma can occur in the kidney but are even less common than renal carcinoids (Gonzales-Lois et al, 2001; Majhail et al, 2003; Lane et al, 2007b). Approximately 30 cases of small cell carcinoma of the kidney have been reported for which another primary site could not be identified (Mackey et al, 1998; Gonzalez-Lois et al, 2001; Akkaya et al, 2003; Majhail et al, 2003; Mirza et al, 2007). On pathologic examination, small cell carcinoma has features of neuroendocrine and epithelial neoplasms and must be differentiated from Wilms tumor, PNET, lymphoma, and metastasis from pulmonary small cell carcinoma (Gonzalez-Lois et al, 2001; Akkaya et al, 2003). Positive staining for neuron-specific enolase, chromogranin, and synaptophysin is characteristic (Akkaya et al, 2003). Preoperative differentiation from RCC is difficult, although a relatively hypovascular pattern may be an indication. Many small cell carcinomas of the kidney are locally advanced or metastatic at presentation, and flank pain or hematuria is common (Majhail et al, 2003; Mirza et al, 2007). Multimodal therapy with nephrectomy or tumor debulking combined with platinum-based chemotherapy regimens is advocated for extrapulmonary small cell carcinoma in general and may also be useful for the renal manifestation of this malignant neoplasm (Majhail et al, 2003; Mirza et al, 2007). Long-term survivors are described but rare, and new treatment regimens are needed.
PNET is related to the Ewing sarcoma family of tumors that are more common in the pediatric population, typically manifesting in the bone or soft tissues of the extremities, trunk, and head and neck and only rarely in the viscera or kidneys (Jimenez et al, 2002; Maly et al, 2004). However, a person of any age may be affected and several cases of PNET have been reported in the kidneys of adults (Rodriquez-Galindo et al, 1997; Karnes et al, 2000; Doerfler et al, 2001; Parham et al, 2001; Thomas et al, 2002; Pomara et al, 2004; Thyavihally et al, 2008). The majority of renal PNET are seen in patients aged 10 to 39 years (Ellinger et al, 2006). These tumors are derived from primitive neural crest cells, and positive staining for CD99 in addition to vimentin, cytokeratin, and neuron-specific enolase strongly supports the diagnosis (Gonlusen et al, 2001; Parham et al, 2001; Ginsberg et al, 2002; Maly et al, 2004; Ellinger et al, 2006). On microscopic examination, renal PNET typically shows small round cells that may form characteristic Homer-Wright rosettes (Marley et al, 1997; Pomara et al, 2004; Thyavihally et al, 2008). A characteristic t(11;22)(q24;q12) translocation is highly specific for PNET and can help differentiate it from neuroblastoma or adult Wilms tumor (Marley et al, 1997; Quezado et al, 1997; Parham et al, 2001; Jimenez et al, 2002). Clinical symptoms are nonspecific, and CT often demonstrates a heterogeneous ill-defined mass with areas of necrosis (Doerfler et al, 2001). Again, equivocal enhancement with contrast medium is typical and differentiation from RCC is often difficult. Renal PNET appears to behave more aggressively than similar tumors at other sites and may represent a distinct clinical entity (Rodriquez-Galindo et al, 1997; Parham et al, 2001; Jimenez et al, 2002; Pomara et al, 2004; Thyavihally et al, 2008). A strong propensity for local recurrence and early metastasis to lymph nodes, lung, liver, and bone has been described (Rodriquez-Galindo, 1997; Quezado et al, 1997; Gonlusen et al, 2001; Ellinger et al, 2006). Multimodal treatment protocols combining tumor debulking, chemotherapy targeted to round cell or Ewing’s family of tumors, and radiotherapy to the renal bed are often employed, but prognosis is poor with overall 5-year disease-free survival of 45% to 55% (Casella et al, 2001; Ellinger et al, 2006; Thyavihally et al, 2008).
Wilms tumor is the most common abdominal malignant neoplasm in children, but 3% of Wilms tumors are seen in adults. Of these, 20% are found between the ages of 15 and 20 years and the remaining 80% are distributed between the third and seventh decades of life (Winter et al, 1996). Adult and pediatric Wilms tumors are histologically similar with a distinctive triphasic pattern consisting of varying amounts of blastema, epithelium, and stroma (Orditura et al, 1997). Pathologic staging is the same as for pediatric Wilms tumor, and most authors recommend treating adult patients with the same therapeutic protocols that have been tested and proven in children (Firoozi and Kogan, 2003). Adult Wilms tumor typically presents as a heterogeneous intrarenal mass on CT with a relatively hypovascular pattern, but differentiation from RCC can be difficult if not impossible in many cases (Winter et al, 1996; Orditura et al, 1997; Reinhard et al, 2004). Clinical presentation of adult Wilms tumor is also similar to that of RCC, and this tends to be an unsuspected pathologic diagnosis in most cases. Multimodal therapy should be considered analogous to the treatment protocols for pediatric Wilms tumor (Bozeman et al, 1995; Green, 1997; Neville and Ritchey, 2000; Terenziani et al, 2004). Prognosis is worse for adults with Wilms tumor than for children with this malignant neoplasm because adults are more likely to present with advanced disease and a sudden drop in performance status (Winter et al, 1996; Akmansu et al, 1998).
Campbell SC, Novick AC, Belldegrun A, et al. Guideline for management of the clinical T1 renal mass. J Urol. 2009;182:1271-1279.
Chawla SN, Crispen PL, Hanlon AL, et al. The natural history of observed enhancing renal masses: meta-analysis and review of the world literature. J Urol. 2006;175:425-431.
DeCastro GJ, McKiernan JM. Epidemiology, clinical staging and presentation of renal cell carcinoma. Urol Clin North Am. 2008;35:581-592.
Edge SB, Byrd DR, Compton CC, et al, editors. AJCC cancer staging manual, 7th ed. Springer, New York, 2010, 479-489.
Grubb RI3rd, Choyke PL, Pinto PA, et al. Management of von Hippel-Lindau–associated kidney cancer. Nat Clin Pract Urol. 2005;2:248-255.
Jonasch E, Tannir NM. Adjuvant and neoadjuvant therapy in renal cell carcinoma. Cancer J. 2008;14:315-319.
Kim WY, Armstrong AJ, George DJ. mTOR pathway biology in renal cell carcinoma. In: Rini BI, Campbell SC, editors. Renal cell carcinoma. Shelton (CT): People’s Medical Publishing House; 2009:225-234.
Lane BR, Campbell SC, Poggio ED, et al. Renal function assessment in the era of chronic kidney disease: renewed emphasis on renal function centered patient care. J Urol. 2009;182:4335-4444.
Lane BR, Samplaski MK, Herts BR, et al. Renal mass biopsy: a renaissance? J Urol. 2008;179:20-27.
Leibovich BC, Blute ML. Lymph node dissection in the management of renal cell carcinoma. Urol Clin North Am. 2008;35:673-678.
Lipworth L, Tarone RE, McLaughlin JK. The epidemiology of renal cell carcinoma. J Urol. 2006;176:2353-2358.
Nguyen C, Campbell SC. Staging of renal cell carcinoma. Clin Genitourin Cancer. 2006;5(3):190-197.
Nguyen CT, Campbell SC, Novick AC. Choice of operation for clinically localized renal tumor. Urol Clin North Am. 2008;35:645-655.
Ost MC, Kavoussi LR. Image guided percutaneous treatment of renal masses: cryoablation and radiofrequency ablation. AUA Update Series. 2007;26:126-135.
Rini BI, Campbell SC, Escudier B. Renal cell carcinoma. Lancet. 2009;373:1119-1132.
Roma AA, Zhou M. Pathological classification and molecular genetics of renal cell carcinoma. AUA Update Series. 2007:26.
Russo P, Huang W. The medical and oncological rationale for partial nephrectomy for the treatment of T1 renal cortical tumors. Urol Clin North Am. 2008;35:635-643.
Simmons MN, Herts B, Campbell SC. Image-based approaches to the diagnosis and treatment of renal masses. AUA Update Series. 2007;26(39):381-391.
Vira MA, Novakovic KR, Pinto Peter A, et al. Genetic basis of kidney cancer: a model for developing molecular-targeted therapies. BJU Int. 2007;99:1223-1229.
Abdi R, Dong VM, Rubel JR, et al. Correlation between glomerular size and long-term renal function in patients with substantial loss of renal mass. J Urol. 2003;170:42-44.
Abou El Fettouh HI, Cherullo EE, El-Jack M, et al. Sporadic renal cell carcinoma in young adults: presentation, treatment, and outcome. Urology. 2002;60(5):806-810.
Abou Youssif T, Kassouf W, Steinberg J, et al. Active surveillance for selected patients with renal masses: updated results with long-term follow-up. Cancer. 2007;110(5):1010-1014.
Abouassaly R, Lane BR, Novick AC. Active surveillance of renal masses in elderly patients [see comment]. Comment in J Urol. 2008;180(2):433-434. J Urol 2008;180(2):505–8; discussion 508–9
Abreu SC, Gill IS. Renal cell carcinoma: modern surgical approach. Curr Opin Urol. 2003;13(6):439-444.
Abreu SC, Kaouk JH, Steinberg AP, et al. Retroperitoneoscopic radical nephrectomy in a super-obese patient (body mass index 77 kg/m2). Urology. 2004;63(1):175-176.
Adley BP, Smith ND, Nayar R, et al. Birt-Hogg-Dubé syndrome: clinicopathologic findings and genetic alterations. Arch Pathol Lab Med. 2006;130:1865-1870.
Ahmad A, MacLennan GT, Listinsky C. Primary renal lymphoma: a rare neoplasm that may present as a primary renal mass. J Urol. 2005;173:239.
Akkaya BK, Mustafa U, Esin O, et al. Primary small cell carcinoma of the kidney. Urol Oncol. 2003;21:11-13.
Akmansu M, Yapici T, Tulay E. Adult Wilms’ tumor: a report of two cases and their treatment and prognosis. Int Urol Nephrol. 1998;30:529-533.
Alam NA, Olpin S, Leigh IM. Fumarate hydratase mutations and predisposition to cutaneous leiomyomas, uterine leiomyomas and renal cancer. Br J Dermatol. 2005;153(1):11-17.
Albarran J, Imbert L. Les tumeurs du rein. Paris: Masson et Cie; 1903.
Alexander JP, Kudoh S, Melsop KA, et al. T-cell infiltrating renal cell carcinoma display a poor proliferative response even though they can produce IL-2 and express IL2 receptors. Cancer Res. 1993;53:1380-1387.
Alimov A, Li C, Gizatullin R, et al. Somatic mutation and homozygous deletion of PTEN/MMACI gene of 10q23 in renal cell carcinoma. Anticancer Res. 1999;19:3841-3846.
Allaf ME, Bhayani SB, Rogers C, et al. Laparoscopic partial nephrectomy: evaluation of long-term oncological outcome. J Urol. 2004;172:871-873.
Altundag O, Altundag K, Gunduz E. Interleukin-6 and C-reactive protein in metastatic renal cell carcinoma. J Clin Oncol. 2005;23(5):1044.
Amin MB, Corless CL, Renshaw AA, et al. Papillary (chromophil) renal cell carcinoma: histomorphologic characteristic and evaluation of congenital pathologic prognostic parameters in 62 cases. Am J Surg Pathol. 1997;21:621-635.
Amin MB, Tamboli P, Javidan J, et al. Prognostic impact of histologic subtyping of adult renal epithelial neoplasms: an experience of 405 cases. Am J Surg Pathol. 2002;26(3):281-291.
Ammar H, Brown SH, Malani A, et al. A case of paraneoplastic cerebellar ataxia secondary to renal cell carcinoma. South Med J. 2008;101(5):556-557.
Amsellem-Ouazana D, Allory Y, Viellefond A, et al. Survival and prognostic factors of papillary renal cell carcinoma (RCC): long term follow-up of 43 patients. J Urol. 2002;167:192.
Amy-Bazille C, Allory Y, Moline V, et al. Immunohistochemical characterization of the main histological types of epithelial renal tumours using tissue micro-arrays: a study of 310 cases. J Urol. 2004;171:198-205.
Ananthakrishnan L, Herts BR, Simmons MN, Campbell SC. Imaging of the common benign disorders of the urinary tract: urolithiasis and renal infections. AUA Update Series. 2007;26:394-399. [lesson 40]
Anderson JK, Matsumoto ED, Johnson DB, et al. Radiofrequency ablation of small renal tumors: intermediate follow-up. J Urol. 2005;173:263.
Anderson JK, Shingleton WB, Cadeddu JA. Imaging associated with percutaneous and intraoperative management of renal tumors. Urol Clin North Am. 2006;33:339-352.
Antman K, Crowley J, Balcrzak SP, et al. An intergroup phase II randomized study of doxorubicin and dacarbazine with or without ifosfamide and mesna in advanced soft tissue and bone sarcomas. J Clin Oncol. 1993;11:1276-1285.
Antonelli A, Cozzoli A, Zani D, et al. The follow-up management of non-metastatic renal cell carcinoma: definition of a surveillance protocol. BJU Int. 2007;99:296-300.
Aoyama T, Fujikawa K, Yoshimura K, et al. Bilateral renal cell carcinoma in a patient with tuberous sclerosis. Int J Urol. 1996;3:150-515.
Araki T. Leukemic involvement of the kidney in children: CT features. J Comput Assist Tomogr. 1982;6:781-784.
Argani P, Antonescu CR, Couturier J, et al. PRCC-TFE3 renal carcinomas: morphologic, immunohistochemical, ultrastructural, and molecular análisis of an entity associated with the t(X;1)(p11.2;q21). Am J Surg Pathol. 2002;26(12):1553-1566.
Argani P, Antonescu CR, Illei PB, et al. Primary renal neoplasms with the ASPL-TFE3 gene fusion of alveolar soft part sarcoma: a distinctive tumor entity previously included amont renal cell carcinomas of children and adolescents. Am J Pathol. 2001;159:179-192.
Argyropoulos A, Liakatas I, Lykourinas M. Rare haemangiopericytoma: the characteristics of a rare tumor. BJU Int. 2005;95:943-947.
Aron M, Gill IS. Minimally invasive nephron-sparing surgery (MINSS) for renal tumors. II. Probe ablative therapy. Eur Urol. 2007;51(2):348-357.
Aron M, Nair M, Hemal AK. Renal metastasis from primary hepatocellular carcinoma: a case report and review of the literature review. Urol Int. 2004;73(1):89-91.
Arranz-Arija JA, Carrion JR, Garcia FR, et al. Primary renal lymphoma: report of 3 cases and review of the literature. Am J Nephrol. 1994;14:148-153.
Arroyo JC, Gabilondo F, Llorente L, et al. Immune response induced in vitro by CD16− and CD16+ monocyte-derived dendritic cells in patients with metastatic renal cell carcinoma treatment with dendritic cell vaccines. J Clin Immunol. 2004;24(1):86-96.
Asakuma J, Sumitomo M, Asano T, et al. Modulation of tumor growth and tumor induced angiogenesis after epidermal growth factor receptor inhibition by ZD1839 in renal cell carcinoma. J Urol. 2004;171:897-902.
Asanuma H, Nakai H, Takeda M, et al. Renal cell carcinoma in children: experience at a single institution in Japan. J Urol. 1999;162:1402-1405.
Ashida S, Furihata M, Tanimura M, et al. Molecular detection of von Hippel-Lindau gene mutations in urine and lymph node samples in patients with renal cell carcinoma: potential biomarkers for early diagnosis and postoperative metastatic status. J Urol. 2003;169:2089-2093.
Ather MH, Mithani S, Bhutto S, et al. Lupus type anticoagulant in a patient with renal cell carcinoma: an autoimmune paraneoplastic syndrome. J Urol. 2002;167:2129.
Atkins MB, Choueiri TK, Cho D, et al. Treatment selection for patients with metastatic renal cell carcinoma. Cancer. 2009;115:w2327-w2333.
Atkins MB, Hidalgo M, Stadler WM, et al. Randomized phase II study of multiple dose levels of CCI-779, a novel mammalian target of rapamycin kinase inhibitor, in patients with advanced refractory renal cell carcinoma. J Clin Oncol. 2004;22:909-918.
Atkins M, Regan M, McDermott D, et al. Carbonic anhydrase IX expression predicts outcome of interleukin 2 therapy for renal cancer. Clin Cancer Res. 2005;11:3714-3721.
Atwell TD, Farrell MA, Callstrom MR, et al. Percutaneous cryoablation of 40 solid renal tumors with US guidance and CT monitoring: initial experience. Radiology. 2007;243:276-283.
Atwell TD, Farrell MA, Leibovich BC. Percutaneous renal cryoablation: experience treating 115 tumors. J Urol. 2008;179(6):2136-2140. discussion 2140–1
Atzpodien J, Kirchner H, Jonas U, et al. Interleukin-2 and interferon alfa-2a-based immunochemotherapy in advanced renal cell carcinoma: a prospectively randomized trial of German Cooperative Renal Carcinoma Chemoimmunotherapy Group (DGCIN). J Clin Oncol. 22, 2004. 1188–1184
Avigan D. Dendritic cell–tumor fusion vaccines for renal cell carcinoma. Clin Can Res. 2003;19(18 Pt. 2):6347s-6352s.
Baaten G, Voogd AC, Wagstaff J. A systematic review of the relation between interleukin-2 schedule and outcome in patients with metastatic renal cell cancer. Eur J Cancer. 2004;40:1127-1144.
Bach AM, Zhang J. Contemporary radiologic imaging of renal cortical tumors. Urol Clin North Am. 2008;35:593-604.
Balci NC, Semelka RC, Patt RH, et al. Complex renal cysts: findings on MR imaging. AJR. 1999;172:1495-1500.
Banchereau J, Palucka AK. Dendritic cells as therapeutic vaccines against cancer. Immunology. 2005;5(4):296-306.
Bandi G, Wen CC, Moon TD, Nakada SY. Single center preliminary experience with hand-assisted laparoscopic resection of isolated renal cell carcinoma fossa recurrences. Urology. 2008;71:495-499. discussion 499–500
Bangalore N, Bhargava P, Hawkins MJ, et al. Sustained response of sarcomatoid renal-cell carcinoma to MAID chemotherapy: case report and review of the literature. Ann Oncol. 2001;12(2):271-274.
Banks RE, Tirukonda P, Taylor C, et al. Genetic and epigenetic analysis of von Hippel-Lindau (VHL) gene alterations and relationship with clinical variables in sporadic renal cancer. Cancer Res. 2006;66:2000-2011.
Barbalias GA, Liatsikos EN, Tsintavis A, Nikiforidis G. Adenocarcinoma of the kidney: nephron-sparing surgical approach versus radical nephrectomy. J Surg Oncol. 1999;72:156.
Barbaric ZL. Principles of genitourinary radiology, 2nd ed. New York: Thieme Medical; 1994.
Barocas DA, Mathew S, DelPizzo JJ, et al. Renal cell carcinoma subtyping by histopathology and fluorescence in situ hybridization on a needle-biopsy specimen. BJU Int. 2006;99:290-295.
Bassignani MJ. Understanding and interpreting MRI of the genitourinary tract. Urol Clin North Am. 2006;33:301-317.
Bassignani MJ, Moore Y, Watson L, Theodorescu D. Pilot experience with real-time ultrasound guided percutaneous renal mass cryoablation. J Urol. 2004;171:1620-1623.
Bassil BA, Dosoretz DE, Prout GR. Validation of the tumor, nodes, and metastasis classification of renal cell carcinoma. J Urol. 1985;134:450-454.
Battagli C, Uzzo RG, Dulaimi E, et al. Promoter hypermethylation of tumor suppressor genes in urine from kidney cancer patients. Cancer Res. 2003;63:8695-8699.
Baumert H, Ballaro A, Shah N, et al. Reducing warm ischaemia time during laparoscopic partial nephrectomy: a prospective comparison of two rneal closure techniques [see comment]. Comment in: Eur Urol. 2007 Oct;52(4):956-958. discussion 958–60; PMID: 17445977. European Urology. 2007 Oct 52(4):1164–9
Baust JG, Gage AA. The molecular basis of cryosurgery. BJU Int. 2005;95:1187-1191.
Bazeed MA, Scharfe T, Becht E, et al. Conservative surgery for renal cell carcinoma. Eur Urol. 1986;12:238.
Beahrs OH, Henson DE, Hutter RVP, Myers MH. American Joint Committee on Cancer manual for staging of cancer, 3rd ed. Philadelphia: JB Lippincott; 1988.
Bechtold RE, Zagoria RJ. Imaging approach to staging of renal cell carcinoma. Urol Clin North Am. 1997;24:507-522.
Beck SD, Patel MI, Snyder ME, et al. Effect of papillary and chromophobe cell type on disease-free survival after nephrectomy for renal cell carcinoma. Ann Surg Oncol. 2004;11(1):71-77.
Becker F, Siemer S, Hack M, et al. Excellent long-term cancer control with elective nephron-sparing surgery for selected renal cell carcinomas measuring more than 4 cm. Eur Urol. 2006;49(6):1058-1063. discussion 1063–4
Becker F, Siemer S, Humke U, et al. Elective nephron-sparing surgery should become standard treatment for small unilateral renal cell carcinoma: long-term survival data of 216 patients. Eur Urol. 2006;49(2):308-313.
Bedke J, Buse S, Pritsch M, et al. Perinephric and renal sinus fat infilitration in pT3a renal cell carcinoma: possible prognostic differences. BJU Int. 2009;103:1349-1354.
Begin LR, Guy L, Jacobson SA, et al. Renal carcinoid and horseshoe kidney: a frequent association of two rare entities—a case report and review of the literature. J Surg Oncol. 1998;68:113-119.
Beiko DT, Nickel JC, Boag AH, et al. Benign mixed epithelial stromal tumor of the kidney of possible müllerian origin. J Urol. 2001;166:1381-1382.
Beland MD, Mayo-Smith WW, Dupuy DE, et al. Diagnostic yield of 58 consecutive imaging-guided biopsies of solid renal masses: should we biopsy all that are indeterminate? AJR Am J Roentgenol. 2007;188(3):792-797.
Bell ET. Renal disease. Philadelphia: Lea & Febiger; 1950.
Bell ET. A classification of renal tumors with observations on the frequency of the various types. J Urol. 1938;39:238-243.
Bella AJ, Winquist EW, Perlman EJ. Primary synovial sarcoma of the kidney diagnosed by molecular detection of syt-ssx fusion transcripts. J Urol. 2002;168:1092-1093.
Belldegrun A, deKernion JB. Sarcomas of the genitourinary tract. In: Eilber FR, editor. The soft tissue sarcomas. Orlando (FL): Grune & Stratton; 1987:219.
Belldegrun A, Koo AS, Bochner B, et al. Immunotherapy for advanced renal cell cancer: the role of radical nephrectomy. Eur Urol. 1990;18(Suppl. 2):42-45.
Belldegrun A, Tsui KH, deKernion JB, Smith RB. Efficacy of nephron-sparing surgery for renal cell carcinoma: analysis based on the new 1997 tumor-node-metastasis staging system. J Clin Oncol. 1999;17:28-68.
Bellon SF, Kaplan-Lefko P, Yang Y, et al. c-Met inhibitors with novel binding mode show activity against several hereditary papillary renal cell carcinoma-related mutations. J Biol Chem. 2008;283:2675-2683.
Bennett RT, Lerner SE, Taub HC, et al. Cytoreductive surgery for stage IV renal cell carcinoma. J Urol. 1995;154:32-34.
Bennington JL. Renal adenoma. World J Urol. 1987;5:66-77.
Bennington JL, Beckwith JB. Tumors of the kidney, renal pelvis, and ureter, 2nd series, fascicle 12. Atlas of tumor pathology. Armed Forces Institute of Pathology, Washington (DC), 1947.
Bennington JL, Ferguson BR, Campbell PB. Epidemiologic studies of carcinoma of the kidney: association of renal adenoma with smoking. Cancer. 1968;22:821-823.
Berger A, Kamoi K, Gill IS, Aron M. Cryoablation for renal tumors: current status. Curr Opin Urol. 2009;19:138-142.
Bergstrom A, Hsieh CC, Lindblad P, et al. Obesity and renal cell cancer—a quantitative review. Br J Cancer. 2001;85(7):984-990.
Berman A, Platt A, Israel G, et al. The role of 3-dimensional volume rendered MRI (3D MRI) in the preoperative evaluation of renal tumors and surgical planning for partial nephrectomy. J Urol. 165, 2001. 5S–652
Bernstein J, Robbins O, Kissane JM. The renal lesions of tuberous sclerosis. Semin Diagn Pathol. 1986;3:97-105.
Beroukhim R, Brunet JP, DiNapoli A, et al. Patterns of gene expression and copy-number alterations in von Hippel-Lindau disease–associated and sporadic clear cell carcinoma of the kidney. Cancer Res. 2009;69:4674-4681.
Bertini R, Roscigno M, Freschi M, et al. Renal sinus fat invasion in pT3a clear cell renal cell carcinoma affects outcomes of patients without nodal involvement or distant metastases. J Urol. 2009;181:2027-2032.
Bhatt S, MacLennan G, Dogra V, et al. Renal pseudotumors. Am J Roentgenol. 2007;188(5):1380-1387.
Bhayani SB, Rha KH, Pinto PA, et al. Laparoscopic partial nephrectomy: effect of warm ischemia on serum creatinine. J Urol. 2004;172:1264-1266.
Bindra RS, Vasselli JR, Stearman R, et al. VHL-mediated hypoxia regulation of cyclin D1 in renal carcinoma cells. Cancer Res. 2002;62(11):3014-3019.
Birch-Hirschfeld FV, Doederlein A. Zentralbl Krankh Horn Sex Org 1894;3.
Bissada NK, Yakout HH, Babanouri A, et al. Long-term experience with management of renal cell carcinoma involving the inferior vena cava. Urology. 2003;61(1):89-92.
Bissler JJ, Kingswood JC. Renal angiomyolipomata. Kidney Int. 2004;66:924-934.
Bjorge T, Tretli S, Engeland A. Relation of height and body mass index to renal cell carcinoma in two million Norwegian men and women. Am J Epidemiol. 2004;160:1168-1176.
Bjornsson J, Short MP, Kwiatowski DJ, Henske EP. Tuberous sclerosis–associated renal cell carcinoma: clinical, pathological, and genetic features. Am J Pathol. 1996;149:1201-1208.
Blath RA, Mancilla-Jimenez R, Stanley RJ. Clinical comparison between vascular and avascular renal cell carcinoma. J Urol. 1976;115:514-519.
Blay JY, Rossi JF, Eijdenes J, et al. Role of interleukin-6 in the paraneoplastic inflammatory syndrome associated with renal cell carcinoma. Int J Cancer. 1997;72:424-430.
Blom JH, van Poppel HV, Marechal JM, et al. Radical nephrectomy with and without lymph node dissection: preliminary results of the EORTC randomized phase III protocol 30881. EORTC Genitourinary Group. Eur Urol. 1999;36:570-575.
Blom JH, van Poppel HV, Marechal JM, et al. Radical nephrectomy with and without lymph node dissection: final results of the EORTC phase 3 protocol 30881. Eur Urol. 2009;55:28-34.
Blute ML, Boorjian SA, Leibovich BC, et al. Results of inferior vena caval interruption by Greenfield filter, ligation or resection during radical nephrectomy and tumor thrombectomy. J Urol. 2007;178:440-445.
Blute ML, Leibovich BC, Cheville JC, et al. A protocol for performing extended lymph node dissection using primary tumor pathological features for patients treated with radical nephrectomy for clear cell renal cell carcinoma. J Urol. 2004;172:465-469.
Blute ML, Leibovich BC, Lohse CM, et al. The Mayo Clinic experience with surgical management complications and outcome for patients with renal cell carcinoma and venous tumour thrombus. BJU Int. 2004;94:33-41.
Blute ML, Malek RS, Segura JW. Angiomyolipoma: clinical metamorphosis and concepts for management. J Urol. 1988;139:20-24.
Boczko S, Fromowitz FB, Bard RH. Papillary adenocarcinoma of the kidney: a new perspective. Urology. 1979;14:491-495.
Bodmer D, van den Hurk W, Groningen J, et al. Understanding familial and non-familial renal cell cancer. Hum Mol Genet. 2002;11:2489-2498.
Bollens R, Rosenblatt A, Espinoza BP, et al. Laparoscopic partial nephrectomy with “on-demand” clamping reduces warm ischemia time. Eur Urol. 2007;52(3):809-810.
Bolte SL, Ankem MK, Moon TD, et al. Magnetic resonance imaging findings after laparoscopic renal cryoablation. Urology. 2006;67:485-489.
Bonsib SM, Gibson D, Mhoon M, et al. Renal sinus involvement in renal cell carcinomas. Am J Surg Pathol. 2000;24(3):451-458.
Boorjian SA, Crispen PL, Lohse CM, et al. Surgical resection of isolated retroperitoneal lymph node recurrence of renal cell carcinoma following nephrectomy. J Urol. 2008;180:99-103. discussion 103
Booth J, Matin SF, Ahrar K, et al. Contemporary strategies for treating nonhereditary synchronous bilateral renal tumors and the impact of minimally invasive, nephron-sparing techniques. Urol Oncol. 2008;26(1):37-42.
Bosniak MA. The use of the Bosniak classification system for renal cysts and cystic tumors. J Urol. 1997;157:1852.
Bosniak MA, Birnhaum BA, Krinsky GA, Waisman J. Small renal parenchymal neoplasms: further observations of growth. Radiology. 1995;197:589-597.
Bosniak MA, Krinsky GA, Waisman J. Management of small incidental renal parenchymal tumors by watchful waiting in selected patients based on observations of tumor growth rates. J Urol. 1996;155:584A-1092.
Bosniak MA, Megibow AJ, Hulnick DH, et al. CT diagnosis of renal angiomyolipoma: the importance of detecting small amounts of fat. AJR Am J Roentgenol. 1998;151:497-501.
Bosquet M, Dominguez C, Balaguer J, et al. Pediatric renal adenocarcinoma: a review of our series. Urology. 2008;72(4):790-793.
Bouchelouche K, Oehr P. Positron emission tomography and positron emission tomography/computerized tomography of urological malignancies: an update review. J Urol. 2008;179(1):34-45.
Bozeman G, Bissada NK, Abboud MR, et al. Adult Wilms’ tumor: prognostic and management considerations. Urology. 1995;45:1055-1058.
Bratslavsky G, Liu JJ, Johnson AD, et al. Salvage partial nephrectomy for hereditary renal cancer: feasibility and outcomes. J Urol. 2008;179:67-70.
Brauch H, Weirich G, Hornauer MA, et al. Trichloroethylene exposure and specific somatic mutations in patients with renal cell carcinoma. J Natl Cancer Inst. 1999;91:854-861.
Brauch H, Weirich G, Brieger J, et al. VHL alterations in human clear cell renal cell carcinoma: association with advanced tumor stage and a novel hot spot mutation. Cancer Res. 2000;60:1942-1948.
Braybrooke JP, Vallis KA, Houlbrook S, et al. Evaluation of toremifene for reversal of multidrug resistance in renal cell cancer patients treated with vinblastine. Cancer Chemother Pharmacol. 2000;46(1):27-34.
Breda A, Stepanian SV, Lam JC, et al. Use of haemostatic agents and glues during laparoscopic partial nephrectomy: a multi-institutional survey from the United States and Europe of 1347 cases [see comment]. Comment in: Eur Urol. 2007;52(3):803. Eur Urol 2007a;52(3):798–803
Breda A, Stepanian SV, Liao J, et al. Positive margins in laparoscopic partial nephrectomy in 855 cases: a multi-institutional survey from the United States and Europe. J Urol. 2007;278:47-50.
Brenner BM. Hemodynamically mediated glomerular injury and the progressive nature of kidney disease. Kidney Int. 1983;23:647.
Brenner W, Färber G, Herget T, et al. Loss of tumor suppressor protein PTEN during renal carcinogenesis. Int J Cancer. 2002;99(1):53-57.
Brescia A, Pinto F, Gardi M, et al. Renal hemangiopericytoma: case report and review of the literature. Urology. 2008;71(4):e9-12.
Bretheau D, Lechevallier E, de Fromont M, et al. Prognostic value of nuclear grade of renal cell carcinoma. Cancer. 1995;76:2543-2549.
Brierly RD, Thomas PJ, Harrison, et al. Evaluation of fine-needle aspiration cytology for renal masses. BJU Int. 2000;85:14.
Broder S, Waldmann TA. The suppressor-cell network in cancer. N Engl J Med. 1978;299(23):1281-1284.
Broecker B. Non-Wilms’ renal tumors in children. Urol Clin North Am. 2000;27(3):463-469.
Brouwers AH, Buijs WC, Oosterwijk E, et al. Targeting of metastatic renal cell carcinoma with the chimeric monoclonal antibody G250 labeled with (131)I or (111)In: an intrapatient comparison. Clin Cancer Res. 2003;9(10 Pt. 2):3953S-3960S.
Brouwers AH, Dorr U, Lang O, et al. 131I-cG250 monoclonal antibody immunoscintigraphy versus [18 F]FDG-PET imaging in patients with metastatic renal cell carcinoma: a comparative study. Nucl Med Commun. 2002;23:229-236.
Brouwers AH, VanErd JEM, Frielink C, et al. Optimization of radioimmunotherapy of renal cell carcinoma: labeling of monoclonal antibody cG250 with 131I, 90Y, 177Lu, or 186Re. J Nucl Med. 2004;45:327-337.
Brown EA. Renal tumours in dialysis patients: who should we screen? Nephron Clin Pract. 2004;97:c3-c4.
Brown JA, Anderl KL, Borell TJ, et al. Simultaneous chromosome 7 and 17 gain and sex chromosome loss provide evidence that renal metanephric adenoma is related to papillary renal cell carcinoma. J Urol. 1997;158:370-374.
Bruder E, Passera O, Harms D, et al. Morphologic and molecular characterization of renal cell carcinoma in children and young adults. Am J Surg Pathol. 2004;28:1117-1132.
Brunelli M, Eble JN, Zhang S, et al. Metanephric adenoma lacks the gains of chromosomes 7 and 17 and loss of Y that are typical of papillary renal cell carcinoma and papillary adenoma. Mod Pathol. 2003;16(10):1060-1063.
Brunelli M, Eble JN, Zhang S, et al. Gains of chromosomes 7, 17, 12, 16, and 20 and loss of Y occur early in the evolution of papillary renal cell neoplasia: a fluorescent in situ hybridization study. Mod Pathol. 2003;16(10):1053-1059.
Bruning T, Pesch B, Wiesenhutter B, et al. Renal cell cancer risk and occupational exposure to trichloroethylene: results of a consecutive case-control study in Arnsberb, Germany. Am J Indust Med. 2003;43(3):274-285.
Bugert P, Gaul C, Weber K, et al. Specific genetic changes of diagnostic importance in chromophobe renal cell carcinomas. Lab Invest. 1997;76:203-208.
Bui MH, Seligson D, Han K, et al. Carbonic anhydrase IX is an independent predictor of survival in advanced renal clear cell carcinoma: implications for prognosis and therapy. Clin Cancer Res. 2003;9:802-811.
Bui MH, Visapaa H, Seligson D, et al. Prognostic value of carbonic anhydrate IX and Ki67 as predictors of survival for renal clear cell carcinoma. J Urol. 2004;171:2461-2466.
Bui MH, Zisman A, Pantuck AJ, et al. Prognostic factors and molecular markers for renal cell carcinoma. Expert Rev Anticancer Ther. 2001;1(4):565-575.
Bukowski RM. Natural history and therapy of metastatic renal cell carcinoma: the role of interleukin-2. Cancer. 1997;80(7):1198-1220.
Bukowski RM. Immunotherapy in renal cell carcinoma. Oncology. 1999;13:801-810.
Bukowski RM, Novick AC. Clinical practice guidelines: renal cell carcinoma. Cleve Clin Med. 1997;64(21(Suppl. 1)):1-48.
Bukowski RM, Rayman P, Uzzo R, et al. Signal transduction abnormalities in T lymphocytes from patients with advanced renal carcinoma: clinical relevance and effects of cytokine therapy. Clin Cancer Res. 1998;4:337-347.
Bukowski RM, Sharfman W, Murthy S, et al. Clinical results and characterization of tumor-infiltrating lymphocytes with or without recombinant interleukin 2 in human metastatic renal cell carcinoma. Cancer Res. 1991;51(16):4199-4205.
Burgess NA, Koo BC, Calvert RC, et al. Randomized trial of laparoscopic vs. open nephrectomy. J Endourol. 2007;21(6):610.
Burt M. Inferior vena caval involvement by renal cell carcinoma. Urol Clin North Am. 1991;18:437-444.
Butler BP, Novick AC, Miller DP, et al. Management of small unilateral renal cell carcinomas: radical versus nephron-sparing surgery. Urology. 1994;45:34-41.
Buzaid AC, Robertone A, Kisala C, et al. Phase II study of interferon-alpha-2a recombinant (Roferon-A) in metastatic renal cell carcinoma. J Clin Oncol. 1987;5:1083-1089.
Byun SS, Yeo WG, Lee SE, Lee E. Expression of surviving in renal cell carcinomas: association with pathologic features and clinical outcome. Urology. 2007;69:34-37.
Cadeddu JA, Moore RG, Nelson JB, et al. Laparoscopic nephrectomy for renal cell cancer: evaluation of efficacy and safety: a multicenter experience. J Urol. 1998;159:147.
Calle EE, Kaaks R. Overweight, obesity and cancer: epidemiological evidence and proposed mechanisms. Nat Rev Cancer. 2004;4(8):579-591.
Camparo P, Vasiliu V, Molinie V, et al. Renal translocation carcinomas: clinicopathologic, immunohistochemical, and gene expression profiling analysis of 31 cases with a review of the literature. Am J Surg Pathol. 2008;32(5):656-670.
Campbell SC. Advances in angiogenesis research: relevance to urological oncology. J Urol. 1997;158:1663-1667.
Campbell SC, Fichter J, Novick AC, et al. Intraoperative evaluation of renal cell carcinoma: a prospective study of the roles of ultrasonography and histopathological frozen sections. J Urol. 1996;155:1191-1195.
Campbell SC, Krishnamurthi V, Chow G, et al. Renal cryosurgery: experimental evaluation of treatment parameters. Urology. 1998;52:29-34.
Campbell SC, Novick AC. Management of local recurrence following radical nephrectomy or partial nephrectomy. Urol Clinic North Am. 1994;21(4):593-599.
Campbell SC, Novick AC. Surgical technique and morbidity of elective partial nephrectomy. Semin Urol Oncol. 1995;13:281-287.
Campbell SC, Novick AC. Expanding the indications for elective partial nephrectomy: is this advisable? Eur Urol. 2006;49:952-954.
Campbell SC, Novick AC, Belldegrun A, et al. Guideline for management of the clinical T1 renal mass. J Urol. 2009;182:1971-1979.
Campbell SC, Novick AC, Herts B, et al. Prospective evaluation of fine needle aspiration of small, solid renal masses: accuracy and morbidity. Urology. 1997;50:25.
Campbell SC, Novick AC, Streem SB, et al. Complications of nephron sparing surgery for renal tumors. J Urol. 1994;151:1177-1180.
Campbell SC, Novick AC, Streem SB, Klein EA. Management of renal cell carcinoma with coexistent renal artery disease. J Urol. 1993;150:808.
Campbell SC, Russo P, Hamed G, et al. Chromophobe cell carcinoma of the kidney: a clinicopathologic study. Proc Am Urol Assoc. 1996;155:299.
Campbell SC, Russo P, Sheinfeld A, et al. Papillary renal cell carcinoma: clinical and pathological features. J Urol. 1997;157:327.
Canacci AM, MacLennan GT. Carcinoid tumor of the kidney. J Urol. 2008;180(5):2193.
Cangiano T, Liao J, Naitoh J, et al. Sarcomatoid renal cell carcinoma: biologic behavior, prognosis and response to combined surgical resection and immunotherapy. J Clin Oncol. 1999;17:523-528.
Canobbio L, Boccardo F, Guarneri D, et al. Phase II study of navelbine in advanced renal cell carcinoma. Eur J Cancer. 1991;27:804-805.
Caoili EM, Bude RO, Higgins EJ, et al. Evaluation of sonographically guided percutaneous core biopsy of renal masses. AJR Am J Roentgenol. 2002;179(2):373-378.
Cardi G, Heaney JA, Schned AR, Ernstoff MS. Expression of Fas (APO-1/CD95) in tumor-infiltrating and peripheral blood lymphocytes in patients with renal cell carcinoma. Cancer Res. 1998;58:2078-2080.
Carducci MA, Piantadosi S, Pound CR, et al. Nuclear morphology adds significant prognostic information to stage and grade for renal cell carcinoma. Urology. 1999;53:44-49.
Carini M, Minervini A, Lapini A, et al. Simple enucleation for the treatment of renal cell carcinoma between 4 and 7 cm in greatest dimension: progression and long-term survival. J Urol. 2006;175:2022-2026.
Carini M, Selli C, Barbanti G, et al. Conservative surgical treatment of renal cell carcinoma: clinical experience and reappraisal of indications. J Urol. 1988;140:725.
Carrizosa DR, Godley PA. Epidemiology and screening of renal cell carcinoma. In: Rini BI, Campbell SC, editors. Renal cell carcinoma. Shelton (CT): People’s Medical Publishing House; 2009:15-24.
Carson WJ. Tumors of the kidney: histologic study. Trans Soc Urol AMA. 1928.
Carter MD, Tha S, McLoughlin MG, Own DA. Collecting duct carcinoma of the kidney: a case report and review of the literature. J Urol. 1992;147:1096-1098.
Cartwright Pfaffenroth E, Marston Linehan W. Genetic basis for kidney cancer: opportunity for disease-specific approaches to therapy. Expert Opin Biol Ther. 2008;8(6):779-790.
Casella R, Moch H, Rochlitz C, et al. Metastatic primitive neuroectodermal tumor of the kidney in adults. Eur Urol. 2001;39:613-617.
Castellanos RD, Aron BS, Evans AT. Renal adenocarcinoma in children: incidence, therapy and prognosis. J Urol. 1974;111:534-537.
Castilla EA, Liou LS, Abrahams NA, et al. Prognostic importance of resection margin width after nephron-sparing surgery for renal cell carcinoma. Urology. 2002;60(6):993-997.
Castillo M, Petit A, Mellado B, et al. C-kit expression in sarcomatoid renal cell carcinoma: potential therapy with imatinib. J Urol. 2004;171:2176-2180.
Castillo OA, Boyle ET, Kramer SA. Multilocular cysts of kidney: a study of 29 patients and review of literature. Urology. 1991;37:156-162.
Cerilli LA, Huffman HT, Anand A. Primary renal angiosarcoma: a case report with immunohistochemical, ultrastructural and cytogenetic features and review of the literature. Arch Pathol Lab Med. 1998;122:929-935.
Cestari A, Guazzoni G, Dell’Acqua V, et al. Laparoscopic cryoablation of solid renal masses: intermediate term follow up. J Urol. 2004;172:1267-1270.
Chan DY, Cadeddu JA, Jarrett TW, et al. Laparoscopic radical nephrectomy: cancer control for renal cell carcinoma. J Urol. 2001;166:2095-2100.
Chan HSL, Daneman A, Gribbin M, Martin DJ. Renal cell carcinoma in the first two decades of life. Pediatr Radiol. 1983;13:324-328.
Chang AE, Aruga A, Cameron MJ, et al. Adoptive immunotherapy with vaccine-primed lymph node cells secondarily activated with anti-CD3 and IL-2. J Clin Oncol. 1996;15:796-807.
Chang SG, Jeon SH, Lee SJ, et al. Clinical significance of urinary vascular endothelial growth factor and microvessel density in patients with renal cell carcinoma. Urology. 2001;58(6):904-908.
Chao D, Zisman A, Pantuck AJ, et al. Collecting duct renal cell carcinoma: clinical study of a rare tumor. J Urol. 2002;167:71-74.
Chapman AE, Goldstein LJ. Multiple drug resistance: biologic basis and clinical significance in renal cell carcinoma. Semin Oncol. 1995;22:17-28.
Chaudhary A, Seenu V, Sedair G, et al. Hemangiopericytoma of renal pelvis—an unusual tumor in an adolescent. Urology. 2007;70(4):e13-e14.
Chawla SN, Crispen PL, Hanlon AL, et al. Observation of enhancing renal masses: a meta-analysis of worldwide data. J Urol. 2005;173:361.
Chawla SN, Crispen PL, Hanlon AL, et al. The natural history of observed enhancing renal masses: meta-analysis and review of the world literature. J Urol. 2006;175:425-431.
Chawla SN, Greenberg RE, Hanlon AL, et al. The natural history and growth dynamics of observed enhancing renal masses. J Urol. 171, 2004. 4S–1654
Chen CH, Lee PS, Han WJ, et al. Primary giant cell malignant fibrous histiocytoma of the kidney with stag horn calculi. J Postgrad Med. 2003;49:246-248.
Chen DYT, Wong YN, Uzzo RG. Management of small renal masses: active surveillance of sporadic renal masses. In: Rini BI, Campbell SC, editors. Renal cell carcinoma. Shelton (CT): People’s Medical Publishing House; 2009:70-80.
Chen KT. Fibroxanthosarcoma of the kidney. Urology. 1979;13:439-440.
Chen JJ, Getzenberg RH. Using nuclear matrix protein, RCCA-5, as biomarker for renal cell carcinoma. J Urol. 171, 2004. 4S–1023
Cheng WS, Farrow GM, Zincke H. The incidence of multicentricity in renal cell carcinoma. J Urol. 1991;146:1221-1223.
Cherullo EE, Ross JH, Kay R, Novick AC. Renal neoplasms in adult survivors of childhood Wilms tumor. J Urol. 2001;165:2013-2017.
Cheuk W, Lo ES, Chan AK, et al. Atypical epithelial proliferations in acquired renal cystic disease harbor cytogenetic aberrations. Hum Pathol. 2002;33(7):761-765.
Cheville JC, Blute ML, Zincke H, et al. Stage pT1 conventional (clear cell) renal cell carcinoma: pathological features associated with cancer specific survival. J Urol. 2001;166:453-456.
Cheville JC, Lohse CM, Zincke H, et al. Comparisons of outcome and prognostic features among histologic subtypes of renal cell carcinoma. Am J Surg Pathol. 2003;27(5):612-624.
Cheville JC, Lohse CM, Zincke H, et al. Sarcomatoid renal cell carcinoma: an examination of underlying histologic subtype and an analysis of associations with patient outcome. Am J Surg Pathol. 2004;28(4):435-441.
Chieng D, Cohen JM, Waisman J. Fine-needle aspiration cytology of hemangiopericytoms: a report of five cases. Cancer. 1999;87:190-195.
Chikkala NF, Lewis I, Ulchaker J, et al. Interactive effects of α-interferon A/D and interleukin-2 on murine lymphokine-activated killer activity: analysis at the effector and precursor level. Cancer Res. 1990;50:1176-1182.
Childs R, Chernoff A, Cotentin N, et al. Regression of metastatic renal-cell carcinoma after nonmyeloablative allogeneic peripheral-blood stem-cell transplantation. N Engl J Med. 2000;343:750-758.
Cho D, DiBlasio CJ, Snyder ME, et al. Contemporary surgical management of renal cortical tumors of 7 cm or less. J Urol. 171, 2004. 4S–1781
Cho D, Signoretti S, Dabora S, et al. Potential histologic and molecular predictors of response to temsirolimus in patients with advanced renal cell carcinoma. Clin Genitourin Cancer. 2007;5:379-385.
Cho NH, Shim HS, Rha SY, et al. Increased expression of matrix metalloproteinase 9 correlates with poor prognostic variables in renal cell carcinoma. Eur Urol. 2003;44(5):560-566.
Choi JH, Choi GB, Shim KN, et al. Bilateral primary renal non-Hodgkin’s lymphoma presenting with acute renal failure: successful treatment with systemic chemotherapy. Acta Haematol. 1997;97:231-235.
Choppin C. Immunotherapy for renal cell cancer in the era of targeted therapy. Expert Rev Anticancer Ther. 2008;8(6):907-919.
Chosy SG, Nicety SO, Lee FT, et al. Thermosensor-monitored renal cryosurgery in swine: predictors of tissue necrosis. J Urol. 1996;157:250.
Choueiri TK, Garcia JA, Elson P, et al. Clinical factors associated with outcome in patients with metastatic clear-cell renal cell carcinoma treated with vascular endothelial growth factor-targeted therapy. Cancer. 2007;110:543-550.
Chow GK, Myles J, Novick AC, et al. The Cleveland Clinic experience with papillary (chromophil) renal cell carcinoma: clinical outcome with histopathological correlation. Can J Urol. 2001;8(2):1223-1228.
Chow WH, Devesa SS, Warren JL, Fraumeni JRJr. Rising incidence of renal cell cancer in the United States. JAMA. 1999;281:1628-1631.
Chow WH, Gridley G, Frumeni JF, et al. Obesity, hypertension, and the risk of kidney cancer in men. N Engl J Med. 2000;343(18):1305-1311.
Choyke PL. Detection and staging of renal cancer. Magn Reson Imaging Clin North Am. 1997;5:29-47.
Choyke PL, Glenn GM, McClennan MW, et al. von Hippel-Lindau disease: genetic, clinical, and imaging features. Radiology. 1995;194:629-642.
Choyke PL, Glenn GM, Walther MM, et al. Hereditary renal cancers. Radiology. 2003;226:33-46.
Choyke PL, Pavlovich CP, Daryanani KD, et al. Intraoperative ultrasound during renal parenchymal sparing surgery for hereditary renal cancers: a 10 year experience. J Urol. 2001;165:397-400.
Choyke PL, White EM, Zeman RK, et al. Renal metastases: clinicopathologic and radiologic correlation. Radiology. 1987;162:359-363.
Christenson PJ, Craig JP, Bibro MC, O’Connell KJ. Cysts containing renal cell carcinoma in von Hippel-Lindau disease. J Urol. 1982;128:798.
Christiano AP, Yang X, Gerber GS, et al. Malignant transformation of renal angiomyolipoma. J Urol. 1999;161:1900-1901.
Chu PG, Weiss LM. Cytokeratin 14 immunoreactivity distinguishes oncocytic tumour from its renal mimics: an immunohistochemical study of 63 cases. Histopathology. 2001;39:455-462.
Chung SD, Huang KH, Lai MK, et al. Long-term follow-up of hand-assisted laparoscopic radical nephrectomy for organ-confined renal cell carcinoma. Urology. 2007;69:652.
Ciancio G, Livingstone AS, Soloway M. Surgical management of renal cell carcinoma with tumor thrombus in the renal and inferior vena cava: the University of Miami experience in using liver transplantation techniques. Eur Urol. 2007;51:988-994.
Ciancio G, Solway M. Renal cell carcinoma invading the hepatic veins. Cancer. 2001;92(7):1836-1842.
Ciancio G, Soloway M. The use of natural veno-venous bypass during surgical treatment of renal cell carcinoma with inferior vena cava thrombus. Am Surgeon. 2002;68:488-490.
Ciancio G, Vaiya A, Savoie M, et al. Management of renal cell carcinoma with level III thrombus in the inferior vena cava. J Urol. 2002;168:1374-1377.
Cibas ES, Goss GA, Kulke MH, et al. Malignant epithelioid angiomyolipoma (“sarcoma ex angiomyolipoma”) of the kidney: a case report and review of the literature. Am J Surg Pathol. 2001;25(1):121-126.
Cindolo L, Chiodini P, Gall C, et al. Validation by calibration of the UCLA integrated staging system prognostic model for nonmetastatic renal cell carcinoma after nephrectomy. Cancer. 2008;113:65-71.
Cindolo L, de la Taille A, et al. Chromophobe renal cell carcinoma: comprehensive analysis of 104 cases from multicenter European database. Urology. 2005;65:681-686.
Cindolo L, Patard JJ, Chiodini P, et al. Predictive accuracy of four prognostic models for non-metastatic renal cell carcinoma after nephrectomy: a comparative study. J Urol. 2005;173:265.
Clark JD, Atkins MB, Urba WJ, et al. Adjuvant high-dose bolus interleukin-2 for patients with high-risk renal cell carcinoma: a Cytokine Working Group randomized trial. J Clin Oncol. 2003;21(16):3133-3140.
Clark PE, Cookson MS. The von Hippel-Lindau gene: turning discovery into therapy. Cancer. 2008;113(7):1768-1778.
Clark PE, Schover LR, Uzzo RG, et al. Quality of life and psychological adaptation after surgical treatment for localized renal cell carcinoma. Urology. 2001;57(2):252-256.
Clark PE, Woodruff RD, Zagoria RJ, et al. Microwave ablation of renal parenchymal tumors before nephrectomy: phase I study. AJR Am J Roentgenol. 2007;188:1212.
Clifford SC, Czapla K, Richards FM, et al. Hepatocyte growth factor-stimulated renal tubular mitogenesis: effects on expression of c-myc, c-fos, c-met, VEGF and the VHL tumour-suppressor and related genes. Br J Cancer. 1998;77:1420-1428.
Clifford SC, Prowse AH, Affara NA, et al. Inactivation of the von Hippel-Lindau (VHL) tumour suppressor gene and allelic losses at chromosome arm 3p in primary renal cell carcinoma: evidence for a VHL-independent pathway in clear cell renal tumourgenesis. Genes Chromosomes Cancer. 1998;22:200-209.
Cobb WS, Heniford BT, Matthew BD, et al. Advanced age is not a prohibitive factor in laparoscopic nephrectomy or renal pathology. Am Surg. 2004;70(6):537-542.
Cochand-Priollet B, Molinie V, Bougaran J, et al. Renal chromophobe cell carcinoma and oncocytoma: a comparative morphologic, histochemical, and immunohistochemical study of 124 cases. Arch Pathol Lab Med. 1997;121:1081-1086.
Coffey RJ, Flickinger JC, Bissonette DJ, et al. Radiosurgery for solitary brain metastases using the cobalt-60 gamma unit: methods and results in 24 patients. Int J Radiat Oncol Biol Phys. 1991;20:1287.
Cohan RH, Dunnick R, Leder RA, Baker ME. Computed tomography of renal lymphoma. J Comput Assist Tomogr. 1990;14:933-938.
Cohen AJ, Li FP, Berg S, et al. Hereditary renal cell carcinoma associated with a chromosomal translocation. N Engl J Med. 1979;301:592-595.
Cohen D, Zhou M. Molecular genetics of familial renal cell carcinoma syndromes. Clin Lab Med. 2005;25:259-277.
Cohen HT, McGovern FJ. Medical progress: renal-cell carcinoma. N Engl J Med. 2005;353:2477-2490.
Cohn EB, Campbell SC. Screening for renal cell carcinoma. In: Bukowski RM, Novick AC, editors. Renal cell carcinoma: molecular biology, immunology, and clinical management. Totowa (NJ): Humana Press, 2000.
Coleman J, Behari A, Linehan WM, et al. Patterns of failure in patients treated by radiofrequency ablation with over one year follow up. J Urol. 2005;173:262.
Coleman JA. Familial and hereditary renal cancer syndromes. Urol Clin North Am. 2008;35:563-572.
Coleman RE. Bisphosphonates: clinical experience. Oncology. 2004;4:14-27.
Colevas AD, Kantoff PW, DeWolf WC, Canellos GP. Malignant lymphoma of the genitourinary tract. In: Vogelzang NJ, editor. Comprehensive textbook of genitourinary oncology. 2nd ed. Philadelphia: Lippincott Williams & Wilkins; 2000:1120-1132.
Coll DM, Uzzo RG, Herts BR, et al. 3-Dimensional volume rendered computerized tomography for preoperative evaluation and intraoperative treatment of patients undergoing nephron sparing surgery. J Urol. 1999;161:1097.
Colombo JRJr, Haber GP, Jelovsek JE, et al. Seven years after laparoscopic radical nephrectomy: oncologic and renal functional outcomes. Urology. 2008;71(6):1149-1154.
Contractor H, Zariwala M, Bugert P, et al. Mutation of the p53 suppressor gene occurs preferentially in the chromophobe type of renal cell tumour. J Pathol. 1997;181:136-139.
Cook A, Lorenzo AJ, Salle JL, et al. Pediatric renal cell carcinoma: single institution 25-year case series and initial experience with partial nephrectomy. J Urol. 2006;175(4):1456-1460. discussion 1460
Cooper CS, Cohen MB, Donovan JFJr. Splenectomy complicating left nephrectomy. J Urol. 1996;155:30-36.
Cooper PH, Waisman J. Tubular differentiation and basement membrane production in a renal adenoma: ultrastructural features. J Pathol. 1973;109:113-121.
Coppin C, Prozsolt F, Awa A, et al. Immunotherapy for advanced renal cell cancer. Cochrane Database Syst Rev 2005;(1):CD001425.
Corica FA, Iczkowski KA, Cheng L, et al. Cystic renal cell carcinoma is cured by resection: a study of 24 cases with long-term follow-up. J Urol. 1999;161:408-411.
Corwin TS, Lindberg G, Traxer O, et al. Laparoscopic radiofrequency thermal ablation of renal tissue with and without hilar occlusion. J Urol. 2001;166:281-284.
Corwin WC. Multiple adenomas of kidneys: report of a case. J Urol. 1940;43:249-252.
Coskun DJ, Gilchrist J, Dupuy D. Lumbosacral radiculopathy following radiofrequency ablation therapy. Muscle Nerve. 2003;28(6):754-756.
Costanzi S, Zoli A, Ferraro PM. A paraneoplastic retroperitoneal fibrosis resistant to corticosteroids treated with tamoxifen. Clin Nephrol. 2008;70(2):172-175.
Cowie A. Renal imaging in patients requiring renal replacement therapy. Semin Dialysis. 2002;15:237-249.
Cox CE, Lacy SS, Montgomery WG, Boyce WH. Renal adenocarcinoma: a 28-year review with emphasis on rationale and feasibility of preoperative radiotherapy. J Urol. 1970;105:51-57.
Crispen PL, Boorjian SA, Lohse CM, et al. Outcomes following partial nephrectomy by tumor size. J Urol. 2008;180:1912-1917.
Crispen PL, Boorjian SA, Lohse CM, et al. Predicting disease progression after nephrectomy for localized renal cell carcinoma: the utility of prognostic models and molecular biomarkers. Cancer. 2008;113:450-460.
Crispen PL, Uzzo RG. Active surveillance of clinically localized sporadic renal cell carcinoma. AUA Update Series. 2006;25:118-123. [lesson 13]
Crispen PL, Viterbo R, Boorjian SA, et al. Natural history, growth kinetics, and outcomes of untreated clinically localized renal tumors under active surveillance. Cancer. 2009;15:2844-2852.
Crispen PL, Viterbo R, Fox EB, et al. Delayed intervention of sporadic renal masses undergoing active surveillance. Cancer. 2008;112:1051.
Crispen PL, Wong YN, Greenberg RE, et al. Predicting growth of solid renal masses under active surveillance. Urol Oncol. 2008;26:555-559.
Crossey PA, Richards FM, Foster K, et al. Identification of intragenic mutations in the von Hippel-Lindau disease tumour suppressor gene and correlation with disease phenotype. Hum Mol Genet. 1994;3:1303-1308.
Crotty TB, Farrow GM, Lieber MM. Chromophobe cell renal carcinoma: clinicopathological features of 50 cases. J Urol. 1995;154:964-967.
Crotty TB, Lawrence KM, Moertel CA, et al. Cytogenic analysis of six renal oncocytomas and a chromophobe cell renal carcinoma: evidence that -Y, -1 may be a characteristic anomaly in renal oncocytomas. Cancer Genet Cytogenet. 1992;61:108-109.
Cuevas C, Raske M, Bush WH, et al. Imaging primary and secondary tumor thrombus of the inferior vena cava: multidetector computer tomography and magnetic resonance imaging. Curr Probl Diagn Radiol. 2006;35:90-101.
Culine S, Bedradda M, Terrier-Lacombe MJ, Droz JP. Treatment of sarcomatoid renal cell carcinoma: is there a role for chemotherapy? Eur Urol. 1995;27:138-141.
Culp SH, Wood CG. Should patients undergoing surgery for renal cell carcinoma have a lymph node dissection? Nat Clin Pract Urol. 2009;6:126-127.
Currie MJ, Gunningham SP, Turner K, et al. Expression of the angiopoietins and their receptor Tie2 in human renal clear cell carcinomas: regulation by the von Hippel-Lindau gene and hypoxia. J Pathol. 2002;198(4):502-510.
Cutuli BF, Methlin A, Teissier E, et al. Radiation therapy in the treatment of metastatic renal cell carcinoma. Prog Clin Biol Res. 1990;348:179.
Czene K, Hemminki K. Familial papillary renal cell tumors and subsequent cancers: a nationwide epidemiological study from Sweden. J Urol. 2003;169:1271-1275.
Czerny HE. cited by Herczel E): Ueber nierenextirpation bietr. Klin Khirurg. 1890;6:485.
Dall’Oglio MF, Antunes AA, Pompeo AC, et al. Prognostic relevance of the histological subtype of renal cell carcinoma. Int Braz J Urol. 2008;34:3-8.
Dall’Oglio MF, Lieberknecht M, Gouveia V, et al. Sarcomatoid differentiation in renal cell carcinoma: prognostic implications. Int Braz J Urol. 2005;31:10-16.
Daneshmand S, Tyler S, Quek ML, et al. Pathologic factors associated with positive lymph nodes in renal cell carcinoma. J Urol. 171, 2005. 4S–1647
Daliani DD, Papandreou CN, Thall PF, et al. A pilot study of thalidomide in patients with progressive metastatic renal cell carcinoma. Cancer. 2002;95(4):758-765.
Dancey JE. Epidermal growth factor receptor and epidermal growth factor receptor therapies in renal cell carcinoma: do we need a better mouse trap? J Clin Oncol. 2004;22:2975-2977.
D’Armiento M, Damiano R, Feleppa B, et al. Elective conservative surgery for renal carcinoma versus radical nephrectomy: a prospective study. Br J Urol. 1997;79:15.
Dash A, Vickers AJ, Schachter LR. Comparison of outcomes in elective partial vs. radical nephrectomy for clear cell renal cell carcinoma of 4 to 7 cm. BJU Int. 2006;97(5):939-945.
Davidson AJ, Choyke PL, Hartman DS, Davis CJ. Renal medullary carcinoma associated with sickle cell trait: radiologic findings. Radiology. 1995;195:83-85.
Davidson AJ, Hayes WS, Hartman DS, et al. Renal oncocytoma and carcinoma: failure of differentiation with CT imaging. Radiology. 1993;183:693-696.
Davidson AJ, Hartman DS, Choyke PL, et al. Radiologic assessment of renal masses: implications for patient care. Radiology. 1997;202:297-305.
Davis CJ, Barton JH, Sesterhenn IA, Mostofi FK. Metanephric adenoma: clinicopathological study of fifty patients. Am J Surg Pathol. 1995;19:1101-1114.
Davis CJ, Mostofi FK, Sesterhann IA. Renal medullary carcinoma: the seventh sickle cell nephropathy. Am J Surg Pathol. 1995;19:1-11.
Davis CJ, Mostofi FK, Sesterhenn IA, et al. Renal oncocytoma: clinicopathological study of 166 patients. J Urogenital Pathol. 1991;1:41-52.