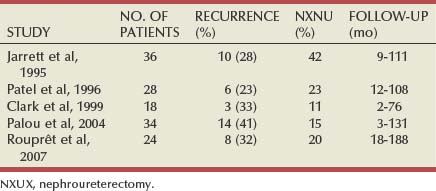
chapter 53 Urothelial Tumors of the Upper Urinary Tract and Ureter
Basic and Clinical Biology
An upper urinary tract tumor can be defined as any neoplastic growth that affects the lining of the urinary tract from the calyces to the distal ureter. These tumors display behaviors that are similar to those of tumors arising in bladder, but they also have many characteristics that separate them from those tumors. For example, unlike bladder urothelial cancers, upper urinary tract cancers are relatively less common and are associated with several familial syndromes. They are more difficult to visualize directly and to reach with topical therapies; and, given the anatomic difference between the upper and lower urinary tracts in terms of muscle thickness and other factors, they may have different prognoses.
Epidemiology and Etiology
Incidence and Mortality Rates
The etiology, natural history, pathology, detection, and staging of urothelial tumors are presented in Chapters 80, 81, and 82. Only the features that differentiate upper tract tumors from bladder tumors and that are pertinent to treatment are revisited here.
Upper urinary tract urothelial tumors involving the renal pelvis or ureter are relatively uncommon, accounting for 5% to 7% of all renal tumors and about 5% of all urothelial tumors (Fraley, 1978; Melamed and Reuter, 1993; Jemal et al, 2004). Because renal pelvic tumors generally are not reported separately, worldwide statistics vary substantially between nations and are not accurate. However, the highest incidence appears to occur in Balkan countries, where urothelial cancers represent 40% of all renal cancers.
The frequency of urothelial tumors of the upper tract is increasing, even though the tumors represent only a small percentage of all urothelial neoplasms (McCarron et al, 1982; Richie, 1988; Williams, 1991; Herr, 1998; Messing and Catalona, 1998; Munoz and Ellison, 2000; David et al, 2009). The peak incidence of upper tract tumors is 10 per 100,000 per year, occurring in the age range of 75 to 79 years. Fortunately, synchronous bilateral urothelial upper urinary tract tumors are very rare (Holmang and Johansson, 2004). In one series from Sweden the percentage of upper tract tumors that were bilateral was 1.6%, preceded in 80% of cases by a bladder cancer diagnosis. There may be a decreasing incidence of bilaterality secondary to the prohibition of phenacetin-containing analgesics in the 1960s. An evaluation of data from 1973 to 1996 with use of the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) database showed 9057 cases, 5379 of the renal pelvis and 3678 of the ureter. In comparison of age-adjusted annual incidence rates, an increase in ureteral neoplasms from 0.69 to 0.73 per 100,000 person-years was found but no change in the incidence of renal pelvic tumors was apparent. In addition, the rate of in-situ neoplasm increased from 7.2% to 23.1%. The incidence of ureteral and renal pelvis tumors in the United States from 1985 to 1994 was 0.73 and 1.0 each per 100,000 person-years, respectively, based on the SEER database (Munoz and Ellison, 2000). The 5-year disease-specific survival was 75% overall and 95%, 88.9%, 62.5%, and 16.5% for in-situ, localized, regional, and distant disease, respectively. Several factors account for this increase in survival for patients with upper tract tumors. Survival of patients with the more prevalent transitional cell carcinoma (TCC) of the bladder is improved owing to more effective diagnosis and treatment. This produces a length-time bias of longer time at risk for development of upper tract tumors. The National Cancer Data Base (NCDB) for the United States for the years 1993 to 2005 identified a total of 334,480 bladder cancers, 15,105 renal pelvis cancers, and 10,128 ureteral cancers (David et al, 2009). There was a significant increase in high-grade tumors in each of the sites during those years. The percentage of early stage tumors increased for both the renal pelvis and the ureter. However, overall, there was no change in survival during those years. Upper tract urothelial tumors are rarely diagnosed at autopsy but rather present clinically during the patient’s lifetime (Ressequie et al, 1978). It also appears that the true incidence of upper tract tumors is increasing as the population ages. Patients with upper tract cancer are generally older than patients with bladder tumors (Melamed and Reuter, 1993). Upper tract tumors rarely present before the age of 40 years, and the mean age at presentation is 65 years (Anderstrom et al, 1989). In conclusion there appears to be a slight increase in the U.S. national incidence of ureteral cancers during the past 2 decades. Fortunately, this has been associated with a slight improvement in the overall and disease-specific survival of patients with upper tract malignant neoplasms.
Variations by Sex and Race
Men are about twice as likely to develop upper urinary tract tumors as are women (Greenlee et al, 2000). In addition, whites are about twice as likely as African-Americans to develop upper tract tumors (Greenlee et al, 2000). On the other hand, SEER data suggest that disease-specific annual mortality is greater in black men than in white men (7.4% vs. 4.9%) and greater in women than in men (6.1% vs. 4.4%) (Munoz and Ellison, 2000). As is the case with bladder cancer, women who develop upper urinary tract cancer are 25% more likely than men to die of their disease (Greenlee et al, 2000). Accurate data regarding racial differences in mortality are not available.
Upper Urinary Tract Tumors after Known Bladder Cancer
On the basis of SEER data from the period 1973 to 1996, Rabbani and colleagues (2001) reported that upper urinary tract cancers developed in 657 of 91,245 cases of bladder cancer with adequate follow-up (median 4.1 years). The relative risk for upper urinary tract tumors for white men and women was 64.2% and 75.4% at or before 2 years, 44.3% and 40.5% at 2 to 5 years, 50.8% and 42.1% at 5 to 10 years, and 43.2% and 22.2% at more than 10 years, respectively. These authors concluded that the incidence of upper tract cancers is stable on long-term follow-up and that upper tract surveillance must remain rigorous for an extended period. The incidence of upper tract recurrence has been shown to be higher in patients with carcinoma in situ than in patients with noninvasive papillary TCCs and in patients treated with cystectomy for carcinoma in situ rather than for invasive cancer (Solsona et al, 1997; Premoli et al, 2006; Slaton et al, 1999). Upper urinary tract recurrence is also more likely to occur with high-grade bladder cancer (hazard ratio [HR] 2.16), in T1 versus Ta disease (HR 1.16), and in patients with trigonal or periureteral presentation (HR 1.76) (Wright et al, 2009). On pathologic evaluation, recurrence is most likely to be superficial (Ta, T1, Tis) and to occur in the distal ureter only (47%). However, this finding has not been reported in all series. For example, in patients with Ta, T1, and Tis bladder cancers treated with bacille Calmette-Guérin (BCG), Herr and colleagues (1996) reported a 21% upper tract recurrence rate after a median interval of 7.3 years; the majority of tumors were invasive, and 38.8% of patients with recurrence died of their upper urinary tract disease.
Etiology and Risk Factors
Balkan Nephropathy
Balkan nephropathy is characterized by a degenerative interstitial nephropathy occurring in Balkan countries. Afflicted families display a much higher incidence of upper urinary tract TCC, in some areas 100 to 200 times greater than in nonaffected individuals (Petkovic, 1975). Curiously, bladder cancer incidence is not affected. Tumors are generally of low grade and are more frequently multiple and bilateral than are upper tract TCCs due to other causes (Radovanovic et al, 1985). Balkan nephropathy is familial but not obviously inherited, suggesting an environmental etiology that has yet to be identified. Recent studies suggest that dietary exposure to aristolochic acid may be responsible (Grollman et al, 2007). Interestingly, family members who leave home early in life may not be affected (Radovanovic et al, 1985). Overall, the incidence of this syndrome may be declining (Stefanovic et al, 2008). However, poorer outcomes are still seen in women (HR 2.2), with tumor size larger than 3 cm (HR 2.8), and with stage T3 or T4 disease (HR 3.1) (Dragicevic et al, 2007).
Smoking
Cigarette smoking appears to be the most important of the modifiable risk factors for upper urinary tract cancer, producing an incidence three times that seen in nonsmokers. It appears that this risk is dose related, with a rate as high as 7.2 times normal for long-term (>45 years) smokers (McLaughlin et al, 1992). Former smokers also have a twofold increased risk compared with age-matched persons with no smoking history. This risk declines only partially after smoking ceases. In addition, the risk from smoking seems more often to lead to ureteral rather than to renal pelvic tumors.
Coffee Consumption
A relative risk of 1.8 times normal has been described in individuals who consumed more than seven cups of coffee per day (Ross et al, 1989). However, after controlling for cigarette smoking, this risk decreased to 1.3.
Analgesics
Analgesic abuse is a well-documented risk factor associated with the development of upper urinary tract cancers (Johansson et al, 1974; Morrison, 1984; McCredie et al, 1986). In one study, 22% of patients with renal pelvic tumors and 11% of patients with ureteral tumors reported a history of analgesic abuse with a latency period of approximately 2 years (Steffens and Nagel, 1988). Renal papillary necrosis and phenacetin consumption also appear to be independent but synergistic risk factors. Each alone resulted in relative risk factors of 6.9 and 3.6, respectively, but together increased risk 20 times (McCredie et al, 1986). Although phenacetin is the most well-described causative agent in analgesic nephropathy, most patients have reported taking combination preparations that included caffeine, codeine, acetaminophen, and aspirin or other salicylates (De Broe and Elseviers, 1998). Histologic findings associated with analgesic abuse include thickening of the basement membrane (pathognomonic) and papillary scarring. Thickening of the basement membrane has been demonstrated in 15% of patients with upper urinary tract tumors and should alert the physician to the presence of analgesic abuse and the subsequent risk of contralateral involvement (Palvio et al, 1987). The degree of papillary scarring also appears to be closely related to tumor grade, although not with the development of squamous metaplasia or squamous cancer (Stewart et al, 1999).
Arsenic
Excess inorganic arsenic in drinking water from artesian wells is a major health hazard in certain parts of the world and is associated with an increased risk of upper tract urothelial tumors in addition to other diseases (Tan et al, 2008). Chronic exposure to arsenic in southwestern Taiwan has long been associated with a form of peripheral vascular disease known as blackfoot disease that causes dry gangrene of the extremities. Throughout all regions of Taiwan an increased rate of upper tract urothelial tumors has been noted. These tumors behave in a similar fashion to other upper urinary tract tumors of similar grade and stage. However, there is a distinct female predominance of the upper tract tumors seen in Taiwan in contrast to the male predominance seen in all other areas of the world. Some have postulated that the women may be exposed to arsenic fumes during cooking by steam heat over boiling water. If this is correct it implies an inhaled risk as well as the risk of ingestion from drinking water with high arsenic content.
Occupation
A significantly increased risk for upper urinary tract tumors has been reported for persons employed in chemical, petroleum, and plastic industries (relative risk of 4); patients with exposure to coal or coke (relative risk of 4); and patients with exposure to asphalt or tar (relative risk of 5.5) (Jensen et al, 1988). Aniline dyes, β-naphthylamine, and benzidine have been implicated as causative agents, and tumors can occur at long intervals (up to 15 years or more) after exposure.
Chronic Inflammation, Infection, or Exposure to Chemotherapeutic Agents
The development of squamous cell cancer (and less commonly adenocarcinoma) has been shown to be related to chronic bacterial infection associated with urinary stones and obstruction (Godec and Murrah, 1985; Spires et al, 1993). In addition, exposure to cyclophosphamide, an alkylating agent, also appears to confer an increased risk (Brenner and Schellhammer, 1987).
Heredity
Several familial syndromes have been associated with the development of upper tract TCC (Frischer et al, 1985; Orphali et al, 1986; Lynch et al, 1990). Lynch syndrome II, for example, is characterized by the early development of colonic tumors (without polyposis) and extracolonic neoplasms, including upper tract urothelial tumors. Unlike with nonhereditary cancers, these patients are typically younger (mean, 55 years) and are more likely to be female (Lynch et al, 1990).
Natural History
Location and Distribution of Tumors
Ureteral tumors occur more commonly in the lower than in the upper ureter. Overall, about 70% of ureteral tumors occur in the distal ureter, 25% in the midureter, and 5% in the proximal ureter (Anderstrom et al, 1989; Messing and Catalona, 1998). This phenomenon may be a reflection of downstream implantation. One area of consensus is that removal of the entire ureter is mandatory when upper urinary tract cancers are removed by nephroureterectomy. Bilateral involvement (either synchronous or metachronous) occurs in 1.6% to 6.0% of sporadic upper tract TCCs (Babaian et al, 1980; Murphy et al, 1981; Kang et al, 2003).
Patients with upper urinary tract tumors are at risk for development of bladder cancer, with an estimated incidence that varies in multiple reports from 15% to 75% within 5 years of the development of the upper tract cancer (Kakizoe et al, 1980; Huben et al, 1988; Anderstrom et al, 1989; Hisataki et al, 2000; Miyake et al, 2000; Kang et al, 2003). This high incidence of metachronous bladder involvement suggests that routine bladder surveillance should be performed. Why are upper tract cancers followed by bladder cancers more often than bladder cancers are followed by upper tract cancers? Theories include downstream seeding, longer exposure time to carcinogens in the bladder, and greater number of urothelial cells in the bladder that are subject to random carcinogenic events. Studies have suggested that in high-grade cancers (with associated TP53 gene mutations), which also tend to be more rapidly recurrent, specific gene mutations noted in upper tract cancers are also demonstrated in subsequent bladder cancers (Harris and Neal, 1992; Lunec et al, 1992; Habuchi et al, 1993). In contrast, microsatellite studies in low-grade upper tract cancers, which tend to recur less rapidly in the bladder, have suggested genetic discordance between these upper tract tumors and subsequent bladder cancers in 46% of cases (Takahashi et al, 2000).
Upper tract cancers have traditionally been reported to develop in 2% to 4% of patients with bladder cancer, with a mean interval to recurrence of 17 to 170 months (Oldbring et al, 1989; Solsona et al, 1997; Rabbani et al, 2001). Risk factors that have been reported to predict a higher likelihood of the development of upper tract cancers after bladder cancer treatment include stage, grade, multiplicity of tumors in the bladder, presence of ureteral reflux, presence of recurrent carcinoma in situ in the bladder after BCG treatment, multifocal carcinoma in situ in the bladder at the time of cystectomy, and presence of bladder cancers arising close to a ureteral orifice (Zincke et al, 1984; Herr et al, 1992; Hudson and Herr, 1995). Two long-term follow-up series have reported that upper tract recurrence after bladder cancer diagnosis may be much higher than previously thought, occurring in approximately 25% of cases. This phenomenon may be the result of selection of patients, with more high-grade and dysplastic tumors reported in these series (Herr et al, 1996; Solsona et al, 1997).
Delayed recurrence is more common in the ureter than in the renal pelvis and appears to occur earlier (at 40 vs. 67 months). In patients treated with BCG for carcinoma in situ of the bladder, upper urinary tract cancer is even more common (about 30% of cases) and appears to occur distally (in the distal, juxtavesical, and intramural portions of the ureter), especially in those patients subjected to cystectomy whose disease is refractory to BCG. Therefore, in cases of high-risk bladder cancer (high-grade T1 disease or carcinoma in situ), imaging of the upper urinary tract should probably be performed annually as part of routine follow-up (Herr et al, 1996).
Upper tract urothelial cancers are often associated with a poor prognosis. Up to 19% of patients with upper tract TCC have been reported to present initially with metastatic disease (Akaza et al, 1970). However, one recent study based on a four-institution review suggested that although upper tract urothelial cancers were more often invasive and more often poorly differentiated than bladder cancers, in pathologically matched cohorts recurrence with less aggressive disease, progression to more advanced disease, and death occurred with equal frequency among patients with upper tract and lower tract (bladder) urothelial cancers (Catto et al, 2007). Several studies have suggested that renal pelvic tumors have a better overall prognosis and 5-year disease-specific and recurrence-free survival than do ureteral tumors (Park et al, 2004). However, overall prognosis of upper tract tumors seems to be principally related to tumor stage and to a lesser degree to tumor grade. In one series, 5-year survival was 100% for Ta and Tis, 91.7% for T1, 72.6% for T2, and 40.5% for T3 tumors. Multivariate analysis in this series showed that tumor stage (P = .0001) and age of the patient (P = .042) were the only statistically significant predictors of survival (Hall et al, 1998a).
Progression to Muscle Invasion and Metastases
The thin muscle layer of the renal pelvis and ureter probably allows earlier penetration of invasive upper tract tumors through the thinned muscle layer than is seen in bladder cancers (Cummings, 1980; Richie, 1988). In a recent report, 164 patients with upper tract tumors were compared with 2197 patients with bladder cancer. High-grade and deeply invasive TCC occurred in 28.2% of bladder cancers compared with 39.5% of upper tract tumors (Stewart et al, 2005). The renal parenchyma itself may be a barrier to the spread of stage T3 cancers, whereas periureteral tumor extension is a risk factor for early tumor dissemination (Batata and Grabstald, 1976; Guinan et al, 1992a). This anatomic phenomenon may be at least part of the explanation as to why renal pelvic tumors appear to have a better prognosis than ureteral tumors. In one large series of 611 patients treated at 97 hospitals, the 5-year survival rates of patients with stage T3 tumors of the renal pelvis and ureter were 54% and 24%, respectively (Guinan et al, 1992a).
Patterns of Spread
TCCs of the upper urinary tract may spread in several different ways, including direct invasion into the renal parenchyma or surrounding structures, lymphatic or vascular invasion, and epithelial spread by seeding or direct extension. It is clear that high-grade tumors demonstrate a greater propensity to invade and that renal parenchymal invasion is the most significant predictor of the development of metastases (95%), followed by vascular invasion (83%) and lymphatic invasion (77%) (Davis et al, 1987).
Epithelial
To describe the clonal nature of urothelial tumors of the bladder and upper tract, two theories of the nature of this phenomenon have been proposed. The monoclonality theory describes the multiple tumors as the descendants of a single genetically transformed cell that populates the urothelium. In contrast, the field theory assumes a diffuse “cancerization” that is the result of exposure to a carcinogen and results in the independent development of nonrelated tumors at different sites. Although the majority of the evidence supports the monoclonality theory, most of this evidence has resulted from the study of advanced invasive cancers. It seems that a small but significant proportion of multifocal cancers are, in fact, derived from different clones (Hafner et al, 2002).
Epithelial spreading may occur in both antegrade (most common) and retrograde manners. Antegrade seeding is thought to be the most likely explanation for the high incidence of recurrence in patients in whom a ureteral stump is left in situ after nephrectomy and incomplete ureterectomy (Johnson and Babaian, 1979).
Lymphatic
Lymphatic spread from the upper urinary tract extends to the para-aortic, paracaval, and ipsilateral common iliac and pelvic lymph nodes (Batata and Grabstald, 1976). This extension, of course, depends on the location of the primary tumor and is directly related to the depth of invasion of the primary tumor. Whether lymphadenectomy should be performed routinely and the extent of lymphadenectomy remain controversial (Nakazono and Muraki, 1993; Komatsu et al, 1997). In a series of nephroureterectomy and lymph node dissection, Secin and associates (2005) reported positive lymph nodes in 20% of cases (mean number of lymph nodes sampled, 7.3; range, 1 to 17). Death from disease was 25% for patients with N0 tumors compared with 66.7% in the N1-2 group (P < .001). In this series the presence of suspicious lymph nodes on preoperative imaging studies was the only preoperative predictor of lymph node metastasis on multivariate analysis (P = .02).
Hematogenous
The most common sites of hematogenous metastases from upper tract tumors are the liver, lung, and bone (Batata et al, 1975). Although it is very rare, direct extension into the renal veins and vena cava may occur in renal pelvic tumors (Jitsukawa et al, 1985; Geiger et al, 1986).
Pathology
The majority of upper tract tumors are urothelial cancers. Of these, the majority are transitional cell in origin; squamous cell cancers and adenocarcinomas represent a small minority (Bennington et al, 1975; Vincente et al, 1995; Flanigan and Kim, 2004).
Normal Upper Tract Urothelium
The urothelial lining of the upper urinary tract closely approximates that of the bladder except for the markedly reduced thickness of the muscle layer and the abutting of the urothelium to the renal parenchyma proximally. The epithelial layer is continuous from the level of the calyces to the distal ureter. It has been postulated that the urothelial layer may even “extend” into the collecting ducts, raising the possibility that collecting duct renal cancers may be closely related to urothelial cancers and perhaps better treated by agents used for urothelial cancers (Orsola et al, 2005). This observation needs further confirmation.
Renal Pelvis and Calyces
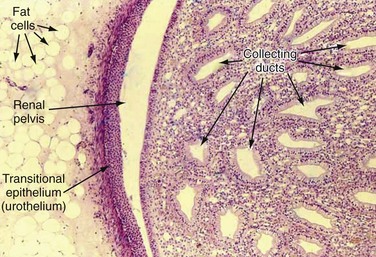
The walls of the calyces and the pelvis contain fibrous connective tissue and two layers of smooth muscle and are lined on their inner surfaces by transitional epithelium (Dixon and Gosling, 1982) (Figs. 53-1 and 53-2). Thin muscle layers originate in the minor calyces and form a spiral, helical arrangement (Fig. 53–3).

Figure 53–1 Low-magnification view of a section through the kidney. The renal medulla ends in the pointed renal papilla. Urine empties into the Y-shaped space made up of the renal calyces (the arms of the Y) and the pelvis (the base of the Y).
Ureter
The ureter demonstrates two continuous thin muscle layers with a loosely spiraled internal layer and a more tightly spiraled external layer. In the lower third of the ureter, a third outer longitudinal layer is present. All three layers merge with the three layers (inner longitudinal, middle circular, and outer longitudinal) of the bladder wall, which run longitudinally, transversely, and obliquely. Beneath the outer muscle coat is the serosa, made up of loose connective tissue and containing blood vessels and lymphatics (Hanna et al, 1976; Notley, 1978) (Figs. 53-4 and 53-5).
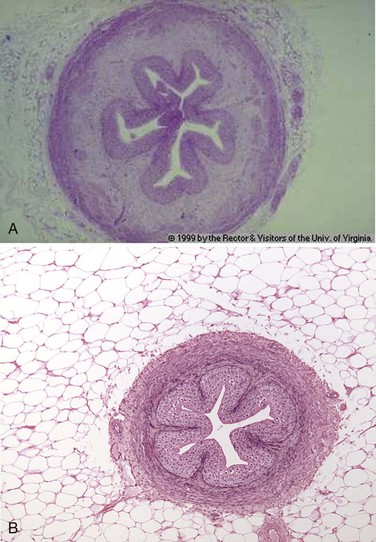
Figure 53–4 A and B, Cross section of ureter. The ureter has an irregular lumen, which is lined by transitional epithelium. Under the epithelium is a connective tissue layer and, beneath that, three layers of smooth muscle: inner longitudinal, middle circular, and outer longitudinal.
(A, © 1999, Rector & Visitors of the University of Virginia.)
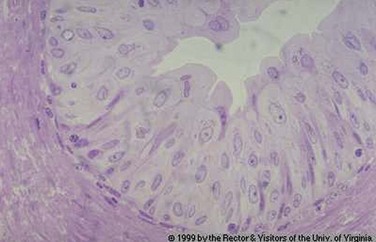
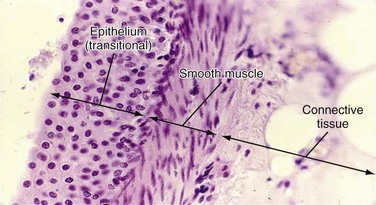
Figure 53–5 Transitional epithelium and loose connective tissue that compose the mucosa of the urinary tract. Transitional epithelium superficially resembles nonkeratinized stratified squamous epithelium, but note that the epithelial cells nearest to the apical (outer) surface are not flattened but cuboidal. Transitional epithelium is a stratified epithelium characterized by the fact that the most apical cells are the roundest and largest in diameter. It is designed to be able to increase its surface area as the lumen is dilated by urine.
(© 1999, Rector & Visitors of the University of Virginia.)
Abnormal Urothelium
Metaplasia and Dysplasia
Several studies have suggested that upper tract urothelial cancers progress through histologic changes from hyperplasia to dysplasia to frank carcinoma in situ in a significant proportion of patients (Heney et al, 1981; McCarron et al, 1982). Carcinoma in situ may be patchy and may extend proximally to the collecting ducts of the kidney (Mahadevia et al, 1983). More severe urothelial dysplastic changes are associated with a greater risk for tumor recurrence in the distal ureter and bladder and a reduced prognosis.
Benign Lesions: Inverted Papillomas and von Brunn Nests
Inverted papillomas, although generally considered benign lesions, often have been shown to be associated with either synchronous or metachronous upper tract urothelial tumors (Renfer et al, 1988; Stower et al, 1990; Chan et al, 1996; Cheville et al, 2000). One series demonstrated an 18% incidence of malignancy associated with inverted papilloma of the ureter (Grainger et al, 1990). Other studies have suggested that there are two types of urinary inverted papilloma. The lesions of type 1 behave in a benign fashion, whereas those of type 2 may have a malignant potential. Because there is currently no way to distinguish between these two types, it has been advised that follow-up for all cases of inverted papilloma be continued for at least 2 years after initial diagnosis (Asano et al, 2003). Similarly, these findings suggest close surveillance of the upper tracts for malignancy is warranted when inverted papilloma is diagnosed.
Transitional Cell Carcinoma
TCC makes up more than 90% of upper urinary tract tumors, may present as papillary or sessile lesions, and may be unifocal or multifocal. On histologic examination these lesions are similar to TCCs of the bladder, but the relative thinness of the muscle layer of the renal pelvis and ureter makes invasion through the muscle coat an earlier event. Carcinoma in situ, as in the bladder, can be particularly difficult to identify and can vary in appearance from a whitish plaque to epithelial hyperplasia or a velvety red patch due to increased submucosal vascularity (Melamed and Reuter, 1993). Progression to muscle invasion or invasion into the renal parenchyma or adventitial tissues may occur and is more likely, given the relative thinness of the muscle coat of the upper tracts.
Non–Transitional Cell Carcinoma
Non-TCCs of the upper tracts represent a wide spectrum of lesions, from benign to highly malignant. The most common of these are squamous cell cancers and adenocarcinomas.
Squamous Cell Cancers
Squamous cell cancers make up 0.7% to 7.0% of upper urinary tract cancers (Babaian and Johnson, 1980; Blacker et al, 1985). They are frequently associated with a condition of chronic inflammation or infection or with analgesic abuse (Stewart et al, 1999). These tumors occur six times more frequently in the renal pelvis than in the ureter and are generally moderately to poorly differentiated and more likely to be invasive at the time of presentation.
Adenocarcinomas
Adenocarcinomas account for less than 1% of all renal pelvic tumors and are typically associated with long-term obstruction, inflammation, or urinary calculi (Stein et al, 1988; Spires et al, 1993). These tumors typically present at advanced stage and display a poor prognosis.
Micropapillary Variant
A micropapillary variant of TCC in the bladder is associated with aggressive behavior and poor outcome. This histologic subtype is very rare in the upper urinary tract. Holmang and associates (2006) described 26 patients with this entity in the upper urinary tract. Twenty-two patients had stage T3 disease at presentation, and carcinoma in situ or lymphovascular invasion was noted in 64% and 81% of cases, respectively. Five-year survival was only 26.9%, and overall the disease-specific mortality was 77%.
Other Miscellaneous Tumors
Fibroepithelial polyps (benign) (Musselman and Kay, 1986; Blank et al, 1987) and neurofibromas (benign) (Varela-Duran, 1987) are uncommon lesions that are typically treated by simple excision.
Multiple types of sarcomas have also been reported to involve the upper urinary tracts, including leiomyosarcomas (Madgar et al, 1988), plasmacytomas (Igel et al, 1991), and angiosarcomas (Coup, 1988). Because of the rare nature of these tumors they are typically treated by excision with adjuvant therapy that is based on the experience with tumors of similar histology occurring elsewhere in the body.
Prognostic Factors
Stage
Stage is currently the most important predictor of survival in patients with upper tract urothelial tumors (Png et al, 2008). The most commonly used staging system is the TNM system (see later section on staging). Upper tract cancers can spread by direct invasion, mucosal seeding, and hematologic and lymphatic routes. Prognosis decreases as stage increases; the most significant decrease in survival is observed in T3 tumors that have penetrated into the perirenal or periureteral fat (Grabstald et al, 1971).
Grade
The traditional grading system used for bladder cancer is also applicable to upper urinary tract tumors. Broder’s original system, modified by Ash, grades tumors from grade 1 to grade 4: grade 1 tumors are primarily papillomas, and grade 4 tumors are highly anaplastic and poorly differentiated tumors (Melamed and Reuter, 1993). The World Health Organization’s system, proposed by Mostofi, eliminates papillomas and grades tumors from grade 1 to grade 3. Recently, tumor grading has been divided into low grade and high grade (Epstein, 1998). Papillomas and papillary urothelial neoplasms of low malignant potential are also described. Certainly, tumors of high grade are more likely to invade into the underlying connective tissue, muscle, and surrounding tissues. Tumors of high grade are also more likely to be associated with concomitant carcinoma in situ.
Location
There remains disagreement as to whether the location of an upper tract tumor affects prognosis. Several studies have suggested that renal pelvic tumors have a better prognosis than ureteral cancers (Park et al, 2004). In contrast, others have argued that when renal pelvic and ureteral tumors are matched for stage there is no significant difference in prognosis (Hall et al, 1998b).
Associated Carcinoma in Situ
As in bladder cancer, carcinoma in situ of the upper tract is associated with a poor prognosis and a likelihood of future development of invasive urothelial cancers. Carcinoma in situ of the distal ureter is most common in patients with bladder carcinoma in situ treated with BCG (30% likelihood).
Lymphovascular Invasion
Lymphovascular invasion has been suggested to be an independent prognostic factor for disease-specific survival in upper tract TCC. In a Japanese study, 173 consecutive patients undergoing surgical treatment of upper tract TCC had lymphovascular invasion determined (Kikuchi et al, 2005). Lymphovascular invasion was found in 30% of cases and was more frequent in advanced pathologic stage. Overall 5- and 10-year survival rates were 84.9% and 80.4%, respectively, in the absence of lymphovascular invasion compared with 40.2% and 21.1% with lymphovascular invasion. In three recent single-center and two multicenter series including a total of 1841 patients with upper tract tumors, the prevalence of lymphovascular invasion varied from 23.7% to 37.8%. Lymphovascular invasion correlated with increasing tumor stage and grade and with disease recurrence and disease-specific survival (Akao et al, 2008; Bolenz et al, 2008; Lin et al, 2008; Chung et al, 2009). In multivariate analysis, lymphovascular invasion was an independent and stronger predictor of recurrence and survival than were tumor stage or grade. Kikuchi and colleagues (2009) reported on a large international, 13-center collaborative series of 1453 patients who underwent radical nephroureterectomy for upper tract urothelial tumors. The overall prevalence of lymphovascular invasion was 24%. Lymphovascular invasion correlated with tumor grade stage, lymph node status, and tumor necrosis. In multivariate analysis, lymphovascular invasion was an independent predictor of disease recurrence and survival for patients with either negative lymph nodes or unknown nodal status. However, lymphovascular invasion was not an independent predictor of outcome for patients with positive lymph nodes. Further prospective, large-scale studies are needed to confirm these observations. As with bladder and other solid organ cancers, lymphovascular invasion is the presumed mechanism that leads to regional lymph node and to systemic hematogenous metastases.
Molecular Biology (Chromosome Abnormalities) and Markers
The molecular and genetic basis of upper tract TCCs appears to be similar to that of TCCs of the bladder. These events have been better described in bladder cancers and are reported in detail in Chapter 80. Briefly, the genetic events leading to the development of upper tract tumors seem to be associated with the overexpression of tumor suppressor genes, including TP53 (on chromosome 17p), the retinoblastoma gene (RB) on chromosome 13q, and several gene foci on chromosome 9 (including the genes for CDKN2C [formerly p18] and CDKN2A [formerly p16] proteins located at 9p21 and 9p32-33). It is generally thought that chromosome 9 abnormalities occur early in the development of these cancers but are not typically associated with high-grade and dysplastic changes, whereas TP53 is more often associated with increased grade and dysplasia. It would also seem, however, that by the time the tumor has been able to invade into the lamina propria both genetic events are likely to have occurred (Spruck et al, 1994). Zigeuner and associates (2004) reported that decreased TP53 immunoreactivity and TP53 overexpression in upper tract tumors were associated with advanced tumor stage and poor prognosis. However, the findings were not independent of stage and grade in multivariate analysis. Eltz and associates (2008) provided a comprehensive review of molecular markers in upper tract urothelial tumors.
TP53
The TP53 nuclear protein staining of cytology specimens obtained ureteroscopically appears to correlate well with the presence of upper tract TCC. In one study, of 36 TP53-positive specimens 28 had simultaneous evidence of upper tract TCC; 80% of the remaining patients who were evaluated serially also had confirmed TCC. All 14 TP53-negative studies occurred in patients with no sign of concurrent malignant disease on ureteroscopy (Keeley et al, 1997b).
c-MET
Recently the roles of c-MET and RON, members of the MET proto-oncogene family of tyrosine kinases, have been studied in upper urinary tract tumors (Compérat et al, 2008). c-MET overexpression correlated with vascular invasion and a worse clinical outcome, whereas that of RON did not correlate with outcome.
COX-2
Abnormal expression of cyclooxygenase-2 (COX-2) has been reported in many forms of human cancer, including bladder urothelial cancer. Kang and coworkers (2008) reported abnormal expression of COX-2 in stromal cells of upper urinary tract cancers correlated with high tumor stage and grade and poor prognosis.
CDKN1B
CDKN1B (formerly p27), a cyclin-dependent kinase inhibitor, has also been shown to predict the prognosis of upper tract tumors. In one study, low levels of CDKN1B staining were indicative of a worse disease-specific survival (Kamai et al, 2000).
Loss of Heterozygosity
Loss of heterozygosity at 9p21 has been observed in bladder cancer and now in upper tract TCC by microsatellite instability analysis. It has been shown that TCC can occur in patients with hereditary nonpolyposis colorectal cancer (HNPCC) syndrome. Patients with this syndrome show genomic lesions in DNA mismatch repair genes (Amira et al, 2003). Furthermore, an inverted growth pattern of cancer has also been associated with microsatellite instability, with a sensitivity and specificity of 0.82 in one study. This finding suggests that microsatellite instability may serve as a marker for inverted growth in upper urinary tract cancers (Hartmann et al, 2003). Ho and coworkers (2008) have reported that a urine-based assay testing for a total panel of 77 markers for microsatellite instability in 30 patients detected 83.3% of cases of an upper urinary tract tumor.
Ploidy-Flow Cytometry
Tumor ploidy has been shown to correlate with survival in upper tract tumors. In one study, tumor aneuploidy was associated with poor 5- and 10-year survival rates of 25% and 0%, respectively (Blute et al, 1988).
Overexpression of Hypoxia-Inducible Factor-1α (HIF-1α)
HIF-1α is a transcription factor that plays an important role in cellular hypoxia adaptation. In a series of patients with upper tract urothelial cancer, positive HIF-1α expression was found in two thirds of cases (was absent in normal urothelium). HIF-1α was significantly associated with high T stage, nodal stage, and grade as well as cancer-specific survival (HR 2.23, P = .004) (Ke et al, 2008).
Other Markers
Rapid urine tests for urothelial malignant neoplasms have been studied extensively for the purpose of identifying lower urinary tract tumors. Less is known about their value in upper tract cancers. Urinary levels of NMP22, a nuclear matrix protein–based marker, have been found to be elevated in patients with upper tract cancer (Carpinito et al, 1996). Although the sensitivity of this test for determining the presence of low-grade tumors is probably higher than that of cytology, the specificity is low. Recently, urine fluorescence in-situ hybridization (FISH) was reported to have a sensitivity of 87.5% and a specificity of 80% for detection of upper tract tumors in a small series (Akkad et al, 2007).
In one series, an analysis of fibrinogen-fibrin degradation products (AuraTek FDP) was compared with the bladder tumor antigen (BTA) test and urine cytology. In this study, the accuracy of the FDP test was 83% compared with 62% for BTA and 59% for cytology (Siemens et al, 2003). Telomerase activity has been shown to be present in most (>95%) upper tract urothelial cancers. It can be detected in exfoliated urinary specimens in a high percentage of patients and thus may prove to be a potentially useful marker (in addition to conventional cytology) to identify upper tract cancers (Wu et al, 2000).
Diagnosis
Symptoms and Signs
The most common presenting symptom of upper tract urothelial tumors is hematuria, either gross or microscopic. This occurs in 56% to 98% of patients (Murphy et al, 1981; Guinan et al, 1992a; Raabe et al, 1992). Flank pain is the second most common symptom, occurring in 30% of tumors. This pain is typically dull and believed to be secondary to a gradual onset of obstruction and hydronephrotic distention. In some cases, pain can be acute and mimic renal colic, typically ascribed to the passage of clots that acutely obstruct the collecting system. These common symptoms of localized disease (hematuria, dysuria) and of advanced upper tract tumors (weight loss, fatigue, anemia, bone pain) are similar in type and frequency to those of bladder cancer. However, flank pain due to obstruction by tumor or clot is more prevalent in upper tract tumors, having been reported in 10% to 40% of cases (Babaian and Johnson, 1980; McCarron et al, 1983; Richie, 1988; Williams, 1991; Melamed and Reuter, 1993). Flank pain in patients with upper tract tumors does not correlate with either locally advanced tumor stage or worse prognosis, as is the case with bladder cancer. About 15% of patients are asymptomatic at presentation and are diagnosed when an incidental lesion is found on radiologic evaluation. Patients may also present with symptoms of advanced disease, including flank or abdominal mass, weight loss, anorexia, and bone pain. Nearly all upper tract tumors are diagnosed during the patient’s life, and therefore upper tract urothelial cancer represents a rare autopsy finding (Ressequie et al, 1978).
Radiologic Evaluation
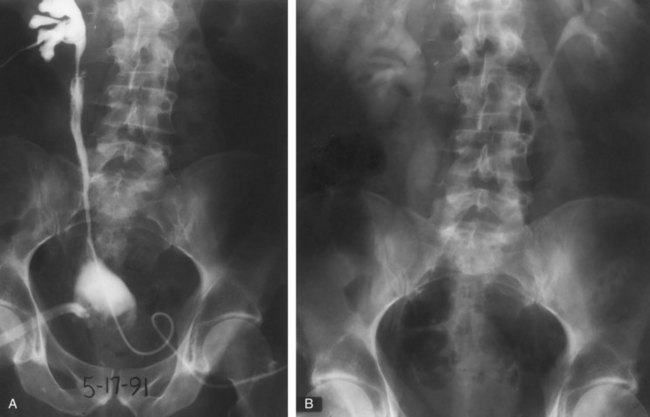
Although intravenous pyelography has been the traditional means for diagnosis of upper tract lesions, computed tomographic (CT) urography is increasingly performed today. CT is easier to perform and less labor intensive than intravenous pyelography. It also has a higher degree of accuracy in determining the presence of renal parenchymal lesions. On the other hand, small urinary filling defects (<5 mm) may be missed between the “cuts” of the traditional CT scan. More recently, CT urography has been performed to obtain a three-dimensional image of the upper tracts. This technique appears to be equal to intravenous pyelography in imaging the ureters and renal pelvis (McTavish et al, 2002). With CT urography, the sensitivity for detecting upper tract malignant disease has been reported to approach 100%, with a specificity of 60% and a negative predictive value of 100% (Caoili et al, 2002). CT urography does, however, expose the patient to higher doses of radiation.
Radiolucent filling defects, obstruction or incomplete filling of a part of the upper tract, and nonvisualization of the collecting system are the typical findings suggestive of an upper urinary tract tumor. Filling defects, which account for 50% to 75% of cases, typically require the intravenous administration of contrast material to be identified (Murphy et al, 1981; Fein and McClennan, 1986). The differential diagnosis of these defects includes blood clot, stones, overlying bowel gas, external compression, sloughed papilla, and fungus ball. Stones can be ruled out most easily by confirmation of calcification by renal ultrasonography or CT. TCCs have an average density of 46 Hounsfield units (HU) and a range of 10 to 70 HU (Lantz and Hattery, 1984). This is in contrast to an average of 100 HU seen in radiolucent uric acid stones (range, 80 to 250 HU). Thus CT can be useful in distinguishing between these two common causes of radiolucent filling defect on excretory urography or retrograde ureterography. The impact of hydronephrosis and nonvisualization for renal pelvis tumors versus ureteral tumors as indicators of a higher stage is uncertain. Nonvisualization is reported in 20% of renal pelvis tumors, only 33% of which are invasive (McCarron et al, 1983). Nonvisualization is reported in 37% to 45% of ureteral tumors and carried a 60% risk of invasion in one series (McCarron et al, 1983). In other reports there is no correlation of nonvisualization and stage (Batata and Grabstald, 1976; Anderstrom et al, 1989). Hydronephrosis with or without an associated filling defect is linked with invasion in 80% of ureteral tumors (McCarron et al, 1983; Cho et al, 2007).
Radiolucent, noncalcified lesions may require additional evaluation by retrograde urography or ureteroscopy, with or without biopsy and cytology. Overall, retrograde urography has an accuracy of 75% in diagnosis of an upper tract malignant neoplasm (Murphy et al, 1981). An incompletely filled or obstructed renal infundibulum or calyx, occurring in 10% to 30% of cases, again typically requires retrograde urography or ureteroscopy to confirm the diagnosis. Obstruction of the urinary tract is a poor prognostic sign for tumor invasion (Babaian and Johnson, 1980).
Evaluation of the contralateral kidney is important not only because of possible bilaterality of the disease but also because it allows a determination of the functionality of the contralateral kidney. This is an extremely important part of the management process. At times, a split-function renal scan may be helpful in determining the contribution of both the “diseased” and the presumed “normal” kidney to the patient’s overall renal function.
Some have suggested that ultrasonography has sensitivity equal to that of urography in evaluating patients with painless gross hematuria for upper tract malignant disease (Yip et al, 1999; Data et al, 2002). For staging purposes, CT or magnetic resonance imaging (MRI) is most useful in determining the extent of invasion, an associated mass lesion outside the collecting system, and the presence of lymph node or distant metastases (Milestone et al, 1990). CT is also more sensitive than conventional radiography in determining minimally radiopaque substances, making it useful in identifying urine excreted by poorly functioning areas of kidney (as in obstructed areas) (Kenney and Stanley, 1987). The greatest downside of CT or MRI is in the detection of small lesions that may be lost in volume averaging. In one series, CT predicted TNM stage in 60% of patients; it understaged 16% and overstaged 24% (Scolieri et al, 2000).
Cystoscopy
Because upper urinary tract tumors are often associated with bladder cancers, cystoscopy is mandatory in the evaluation to exclude coexistent bladder lesions.
Ureteroscopic Evaluation and Biopsy
The technical advances achieved in the realm of endoscopic equipment have made the flexible and rigid ureteroscope a key part of the evaluation (and treatment) of upper urinary tract tumors. Diagnostic accuracy can be improved from approximately 75% with excretory or retrograde urography alone to 85% to 90% when it is combined with ureteroscopy (Streem et al, 1986; Blute et al, 1989). Although pyelovenous and pyelolymphatic migration has been reported with ureteroscopy, this phenomenon appears to be uncommon and should not preclude its use (Lim et al, 1993).
As with bladder tumors, 55% to 75% of ureteral tumors are low grade and low stage (Cummings, 1980; Richie, 1988; Williams, 1991). Also, like bladder cancers, approximately 85% of renal pelvic tumors are papillary and the remainder sessile. Invasion of the lamina propria or muscle (stage T1 or T2) occurs in 50% of papillary and in more than 80% of sessile tumors. Overall, therefore, 50% to 60% of renal pelvic tumors are invasive into either the lamina propria or muscle. In ureteral tumors, invasion is also more common than in bladder tumors (Anderstrom et al, 1989; Williams, 1991).
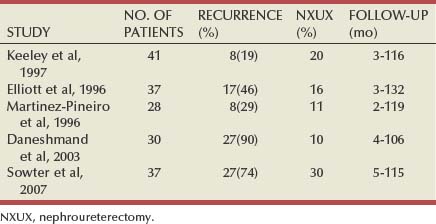
In addition to visualization of the tumor, ureteroscopy allows more accurate biopsy of suspected areas, with either biopsy forceps or brushing. Good histologic correlation (78% to 92%) between the ureteroscopic biopsy specimen and the final pathologic specimen has been established (Keeley et al, 1997c; Guarnizo et al, 2000). It appears that fresh samples obtained ureteroscopically provide the best chance of predicting eventual pathologic findings. In one study, a cell block from biopsy specimens was prepared when a visible tumor was present, and grades of ureteroscopic biopsy specimens were compared with grades and stages of surgical specimens in 42 cases. Of 30 low- or moderate-grade specimens, 29 (90%) proved to be low- or moderate-grade TCC; 11 of 12 high-grade specimens (92%) proved to be high-grade TCC, and 8 (67%) were invasive (T2 or T3) (Keeley et al, 1997c). In contrast, the urologist’s impression of the tumor grade based on ureteroscopic appearance is likely to be correct in only 70% of cases, suggesting that biopsy is also needed to further define this important aspect of staging (El-Hakim et al, 2004).
Because of the small size of ureteroscopic biopsy specimens, a precise correlation with eventual tumor stage is difficult. Therefore, in predicting the tumor stage, a combination of the radiographic studies, the visualized appearance of the tumor, and the tumor grade provides the surgeon with the best estimation of eventual tumor stage. Although, as stated earlier, grading of the tumors may be fairly accurate, staging is much more problematic. Of 40 urothelial tumors staged in one series (40% in the renal pelvis, 20% in the proximal ureter, and 40% in the distal ureter), ureteroscopic grade matched surgical grade in 78% of cases and was less than surgical grade in the remaining 22%. Lamina propria was present in 68% of biopsy specimens (62% of cup biopsies and 100% of loop biopsies), but tumors thought to be Ta were upstaged to T1 to T3 in 45% of cases at the time of complete resection of the lesion (Guarnizo et al, 2000). Therefore, accurate tumor grading on ureteroscopic biopsy is critical in estimating tumor stage. In one series, a biopsy specimen showing grade 3 tumor accurately predicted tumor stage in more than 90% of cases (Skolarikos et al, 2003).
Is ureteroscopy (with or without biopsy) necessary in all cases of suspected upper tract tumors? No. In fact, ureteroscopy should probably be reserved for situations in which the diagnosis remains in question after conventional radiographic studies and for those patients in whom the treatment plan may be modified on the basis of the ureteroscopic findings, for example, endoscopic resection. Although there is no evidence that ureteroscopy diminishes the prognosis of a patient destined to proceed to nephroureterectomy, and although the risks of tumor seeding, extravasations, and dissemination are low in experienced hands, these risks are real and should preclude ureteroscopy when it is unnecessary (Hendin et al, 1999).
Antegrade Endoscopy
In some cases of upper tract tumors, percutaneous access to the renal pelvis may be required for diagnosis or treatment. In such cases, antegrade urography and uroscopy may be useful for tumor resection, biopsy, or simple visualization. Larger-caliber scopes that can be passed into the renal pelvis in this manner may be particularly helpful in resecting or debulking larger volumes of tumor in this area (Streem et al, 1986; Blute et al, 1989). One must remember, however, that tumor cell implantation in the retroperitoneum and along the nephrostomy tube tract has been reported after these procedures (Tomera et al, 1982; Huang et al, 1995).
Role of Cytology and Other Tumor Markers
Urine cytology is a specific tool that is useful in the diagnosis of upper tract carcinomas. On the other hand, the sensitivity of cytology remains an issue. In general, the sensitivity of voided urine (or bladder wash) cytology is directly related to tumor grade. Overall accuracy estimates of the sensitivity of cytology have ranged from about 20% for grade 1 tumors to 45% and 75% for grade 2 and grade 3 tumors, respectively (Murphy and Soloway, 1982; Konety and Getzenberg, 2001).
Even if a voided cytology specimen is abnormal in a patient with an upper tract filling defect, one must be cautious in determining the site of origin of the malignant cells. Ureteral catheterization for collection of urine or washings may provide more accurate cytologic results. However, even in this setting, a substantial false-negative or false-positive result (22% to 35%) can be expected (Zincke et al, 1976). It would appear that saline washing provides a better cell yield and improves cytologic results secondary to the release by hydroscopic forces of loosely adherent cells from the urothelium. Still better accuracy can be achieved by brush biopsy through a retrograde catheter or ureteroscope. Sensitivity in the 90% range with specificity approaching 90% may be possible with these techniques (Streem et al, 1986; Blute et al, 1989). Brush biopsies have, however, also been reported to result in severe complications, including massive hemorrhage and perforation of the urinary tract with extravasation (Blute et al, 1981).
It appears that the exposure of urothelial cells to ionic, high-osmolar contrast agents as in retrograde pyelography may worsen cytologic abnormalities. Thus, it is probably prudent to obtain cytologic specimens before the use of these agents (Terris, 2004).
Early results suggest that FISH also may be useful in the diagnosis of upper tract urothelial tumors. In a study of 21 consecutive upper urinary tract urothelial cancer patients and 10 healthy controls using FISH probes for chromosomes 3, 7, 17, and the CDKN2A (9p21) gene, overall sensitivity of FISH was significantly higher than that of cytology while specificity for both was 100% (Luo et al, 2009).
Staging
The staging of upper urinary tract tumors parallels the staging of bladder tumors and is presented in Chapter 80.
TNM Staging System
The TNM classification and staging system is the most commonly used. A comparison of the American Joint Committee on Cancer (AJCC) staging system and the TNM system is presented in Table 53–1.
Table 53–1 AJCC and TNM Staging and Classification Systems for Upper Urinary Tract Tumors
| Primary Tumor (T) | |
| TX | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| Ta | Papillary noninvasive carcinoma |
| Tis | Carcinoma in situ |
| T1 | Tumor invades subepithelial connective tissue. |
| T2 | Tumor invades the muscularis. |
| T3 | Tumor invades periureteral fat (for renal pelvis only). Tumor invades beyond muscularis into perinephric fat or the renal parenchyma. |
| T4 | Tumor invades adjacent organ or through the kidney into the perinephric fat. |
| Lymph Nodes (N) | |
| NX | Regional lymph nodes cannot be assessed. |
| N0 | No regional lymph node metastases |
| N1 | Metastasis to a single lymph node, 2 cm or less in greatest dimension |
| N2 | Metastasis in a single lymph node, more than 2 cm but not more than 5 cm in greatest dimension; or multiple lymph nodes, none more than 5 cm in greatest dimension |
| N3 | Metastasis in a lymph node, more than 5 cm in greatest dimension |
| Distant Metastasis (M) | |
| MX | Distant metastasis cannot be assessed |
| M0 | No distant metastasis |
| M1 | Distant metastasis |
| AJCC Staging System | TNM Classification System |
|---|---|
| 0 | T0 |
| I | Ta, Tis, T1, N0, M0 |
| II | T2, N0, M0 |
| III | T3, N0, M0 |
| IV | T4 or any T, N+, M+ |
AJCC, American Joint Committee on Cancer.
The histologic characteristics and biology of upper tract tumors still affect treatment decisions, technologic improvements notwithstanding. The entity of benign papilloma, which responds favorably regardless of the extent of treatment, is well described in older series of upper tract tumors (Bloom et al, 1970; Batata and Grabstald, 1976). The existence of similar low-grade papillomas of low-grade malignant potential in the bladder remains controversial (Cheng et al, 1999; Cheng and Bostwick, 2000; Oyasu, 2000). It is unclear whether the differences between upper tract papillomas and bladder papillomas are biologic or semantic. Approximately 85% of renal pelvis tumors are papillary; the remainder are sessile. This distribution is similar to that of bladder tumors. However, the stage of upper tract tumors is T1 or T2 in approximately 50% of papillary and 80% of sessile lesions, respectively (Cummings, 1980; Richie, 1988; Williams, 1991). Thus, 50% to 60% of renal pelvis tumors are invasive, in contrast to most bladder tumors, which are noninvasive; 55% to 75% of ureteral tumors are low grade and low stage, but invasion is still more common than among bladder tumors (Anderstrom et al, 1989; Williams, 1991). Patients with upper urinary tract tumors present most often in the sixth or seventh decade of life and thus are generally older than patients with bladder tumors (Melamed and Reuter, 1993).
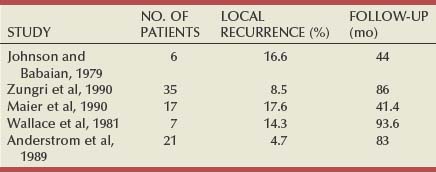
Tumors of the renal pelvis are slightly more common than ureteral tumors (Batata and Grabstald, 1976; Richie, 1988; Maulard-Durdux et al, 1996). Ureteral tumors occur in the distal, middle, and proximal segments in 70%, 25%, and 5% of cases, respectively (Babaian and Johnson, 1980; Anderstrom et al, 1989; Williams, 1991; Messing and Catalona, 1998). After conservative treatment, ipsilateral upper tract tumor recurrence is common in a proximal to distal direction and is seen in 33% to 55% of cases (Mazeman, 1976; Johnson and Babaian, 1979; Babaian and Johnson, 1980; Cummings, 1980; McCarron et al, 1983). Recurrence proximal to the original lesion is rare.
Key Point: Upper Tract Tumor Recurrence
This high rate of ipsilateral recurrence is due in part to a multifocal field change, which is even more pronounced than in bladder cancer. Areas of atypia, dysplasia, or carcinoma in situ are reported in 60% to 95% of specimens after nephroureterectomy for renal pelvis tumor (Johansson et al, 1976; Kakizoe et al, 1980; Heney et al, 1981; Nocks et al, 1982; McCarron et al, 1983; Melamed and Reuter, 1993). Molecular techniques demonstrate that downward seeding of tumor accounts for some recurrences (Harris and Neal, 1992). Tumor multifocality does not lessen survival of patients independent of stage (Messing and Catalona, 1998).
Metachronous bilateral upper tract tumors are rare. In a review of all 768 cases of upper tract tumor reported in western Sweden from 1971 to 1998, the rate of metachronous bilateral tumors was 3.1% and was associated with increased age and short survival time after the event (Holmang and Johansson, 2006).
The occurrence of bladder tumors after upper tract tumors, and vice versa, is another expression of the field change, multifocal risk that affects initial treatment decisions. Carcinoma in situ is present in the distal ureter at the time of cystectomy in 7% to 25% of cases (Melamed and Reuter, 1993; Solsona et al, 1997; Herr, 1998); 15% to 50% of all cases of upper tract tumor occur in patients with a history of bladder tumor (Batata and Grabstald, 1976; Babaian and Johnson, 1980). The incidence of upper tract tumor after bladder tumor is 2% to 4% with a mean time to occurrence of 70 months (Shinka et al, 1988; Oldbring et al, 1989; Melamed and Reuter, 1993; Herr et al, 1996). Upper tract tumors are reported in 3% to 9% of patients after cystectomy for bladder cancer in older series (Zincke and Neves, 1984; Mufti et al, 1988).
Particular insight into the contemporary risk for upper tract tumor after treatment of bladder cancer is provided in several large series (Solsona et al, 1997; Herr, 1998; Rabbani et al, 2001; Mullerad et al, 2004; Sved et al, 2004; Canales et al, 2006; Tran et al, 2008; Wright et al, 2009). Herr (1998) found that among 307 patients with bladder tumor observed for a median of 12 years there was an overall incidence of upper tract tumors of 23%. The cumulative risks for upper tract tumors were 10%, 26%, and 34% at 5 years, 5 to 10 years, and 15 years, respectively. Upper tract tumors occurred in 26% of another subgroup of 87 patients with bladder tumor observed for more than 15 years. Delayed upper tract tumors were more common in the ureter than in the renal pelvis and appeared at a median follow-up of 56 months. Median time to delayed ureteral versus renal pelvis tumors was 40 months versus 67 months, respectively. Ureteral tumors occurred in 29% of 66 patients who were successfully treated with intravesical BCG for carcinoma in situ. In a subset of 105 patients who underwent cystectomy for BCG-refractory carcinoma in situ, ureteral carcinoma in situ was present in the distal, juxtavesical, and intramural portions of the ureter in 35%, 68%, and 81% of cases, respectively. In a retrospective study from the same center, treatment outcome in patients with upper tract tumor was worse in patients with a prior history of bladder cancer (superficial or invasive) independent of upper tumor stage (Mullerad et al, 2004). Similarly, Sved and colleagues (2004) reported upper tract tumors in 2% of patients (5 of 235) observed for a mean of 42 months after radical cystectomy for bladder cancer. Upper tract tumor was diagnosed at a mean follow-up of 39.6 months, because of hematuria in four cases, and on routine intravenous urography in the remaining case. Presence of tumor in the prostatic urethra of the cystectomy specimen was the only initial tumor feature that was associated with a higher risk of subsequent upper tract tumor. This may be a predictor of a higher risk of multifocal tumor in such cases. Outcome was poor with disease-related mortality in four of the cases. In contrast, in a review of 91,245 patients with bladder cancer from the SEER database for 1973 to 1996, upper tract tumor occurred in 657 patients (0.7%) at a median follow-up of 4.1 years (Rabbani et al, 2001). These patients had lower tumor stage and improved disease-specific survival than did patients with primary upper tract tumors. Canales and coworkers (2006) found that patients with two or more stage Ta bladder cancer recurrences within 12 months were at increased risk for upper tract tumors and that surveillance of the upper tracts is indicated. In an update on the earlier study by Herr and associates (1996), Tran and associates (2008) found that the risk for upper tract recurrence after cystectomy for bladder cancer was 4% and 7% at 3 years and 5 years, respectively. Patients who had any tumor in the ureter at the time of cystectomy, including only carcinoma in situ, were at greatest risk for upper tract recurrence. Wright and colleagues (2009) reviewed 99,338 patients with bladder cancer in the SEER registry from 1988 to 2003. Upper tract tumors occurred in 0.8% of patients, at a median time of 33 months. Seventy-one percent of these were seen within 5 years, and only 6% of the total occurred after 10 years. On multivariate analysis high-grade and non–muscle-invasive bladder tumors correlated with a higher risk for subsequent upper tract tumors. The latter point likely relates to a lower mortality rate from the original bladder cancer and thus a greater competing risk for upper tract disease.
Solsona and colleagues (1997) reported that carcinoma in situ was present in the distal ureter in 25% of 138 cystectomies performed for bladder carcinoma in situ, compared with only 2.3% and 2.9% among 786 and 179 cases of stage Ta to T1 versus stage T2 disease, respectively. This may indicate that bladder carcinoma in situ carries a higher risk of multifocal disease than do other forms of bladder tumor.
The incidence of bladder tumor after treatment of upper tract tumor is 20% to 75% (Batata and Grabstald, 1976; Kakizoe et al, 1980; Nocks et al, 1982; Huben et al, 1988; Anderstrom et al, 1989; Sakamoto et al, 1991; Williams, 1991; Melamed and Reuter, 1993; Hisataki et al, 2000; Kang et al, 2003; Matsui et al, 2005). Bladder tumor after upper tract tumor occurs earlier than the reverse, at a median of 21 months versus 86 months afterward, respectively. Both bladder tumor and contralateral upper tract tumor after initial unilateral upper tract tumor are examples of the multifocal nature of urothelial tumor. In one study, subsequent bladder tumors appeared earlier than did contralateral upper tract tumors (Kang et al, 2003). Renal insufficiency was associated with a higher risk of contralateral upper tract tumor.
The occurrence of bladder tumor after treatment of upper tract tumor may be due to a field effect, distal tumor seeding, or both. Association of a higher bladder tumor incidence after upper tract tumor multifocality supports a role of distal seeding (Matsui et al, 2005). However, reports of distinct tumor clones by microsatellite analysis support a field effect (Takahashi et al, 2001). The paradoxical finding that the risk of subsequent bladder tumor is inversely related to upper tract tumor size and stage may reflect a higher and earlier risk of death from the primary tumor in these cases. In the reports by Hisataki and coworkers (2000), Matsui and associates (2005), and Terakawa and colleagues (2008), increased upper tract tumor stage at the time of nephroureterectomy correlated with a higher risk for subsequent bladder tumor. In a recent European multicenter study reported by Novara and coworkers (2008) prior bladder tumor before upper tract tumor was the only independent risk factor for bladder tumor after nephroureterectomy in multivariant analysis. Raman and associates (2007) reported that the grade, but not the stage, of the prior upper tract tumors correlated with the pathologic findings of the subsequent bladder tumors.
Three particular forms of upper tract urothelial tumors, those seen in Balkan nephropathy, those associated with analgesic abuse, as well as those seen in arsenic-endemic regions of Taiwan, each have an even higher tendency to multiple and bilateral recurrences than do sporadic tumors (Markovic, 1972; Petkovic, 1975; Mahoney et al, 1977; Johansson and Wahlquist, 1979; Melamed and Reuter, 1993; Stewart et al, 1999; Tan et al, 2008). The typically low-grade nature of the tumors and the frequent renal insufficiency seen in Balkan nephropathy underscore the importance of conservative treatment when possible. The degree of scarring of renal papillae seen in phenacetin abuse correlates in a dose-dependent manner with the risk of high tumor grade and progression. Calcification of renal papillae after analgesic abuse is associated with development of squamous carcinoma of the renal pelvis (Stewart et al, 1999).
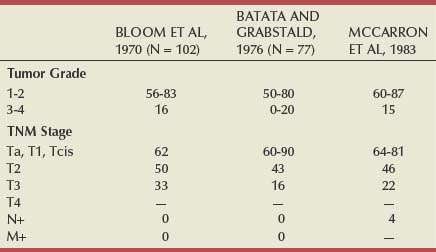
There is a strong correlation of grade and stage for upper tract tumors. The single most important determinant of outcome is tumor stage (Tables 53-2 and 53-3) (Bloom et al, 1970; Grabstald et al, 1971; Batata et al, 1975; Wagle et al, 1975; Babaian and Johnson, 1980; Cummings, 1980; McCarron et al, 1983; Huben et al, 1988; Anderstrom et al, 1989; Guinan et al, 1992b; Terrell et al, 1995; Messing and Catalona, 1998).
Table 53–2 Correlation of Tumor Stage and Grade for Upper Tract Urothelial Tumors
| LOCATION AND STAGE | HIGH GRADE (%) |
|---|---|
| Pelvis | |
| Low | 5 |
| High | 91 |
| Ureter | |
| Low | 26 |
| High | 64 |
Data from McCarron JP Jr, Mills C, Vaughn ED Jr. Tumors of the renal pelvis and ureter: current concepts and management. Semin Urol 1983;1:75–81.
Table 53–3 Literature Review of Overall Survival of Patients with Upper Tract Urothelial Tumors (Renal Pelvis or Ureter) by Stage and Grade
| 5-YEAR SURVIVAL (%) | |
|---|---|
| Tumor Grade | |
| 1-2 | 40-87 |
| 3-4 | 0-33 |
| TNM Stage | |
| Ta, T1, Tcis | 60-90 |
| T2 | 43-75 |
| T3 | 16-33 |
| T4 | 0-5 |
| N+ | 0-4 |
| M+ | 0 |
Key Points: Staging of Upper Tract Tumors
The primary tumor characteristics that suggest invasion, the high incidence of ipsilateral multifocal abnormality, and the risk of recurrence distal to the primary lesion are all offered in support of radical excision of large, proximal, and likely invasive tumors of the upper tract. Conversely, the same risks of multiple recurrence and the fact that tumor stage is the main determinant of outcome support conservative nephron-sparing surgery in many cases.
Upper tract tumors spread in the same ways as bladder tumors do, through lymphatic and hematogenous routes and by direct extension into contiguous structures. Thus, the common metastatic sites are the lungs, liver, bones, and regional lymph nodes. Preoperative evaluation for the extent of disease includes chest radiography, abdominal CT, liver function tests, and occasional bone scintigraphy. The thin muscle layer of the renal pelvis and ureter may allow earlier penetration of invasive upper tract tumors than is seen in bladder neoplasms (Cummings, 1980; Richie, 1988). However, this is not a valid reason for radical versus conservative treatment if a lesion is either noninvasive or invasive and organ confined (stage T2) (Gittes, 1980). The renal parenchyma may be a barrier, slowing distant spread of stage T3 renal pelvis tumors. In contrast, periureteral tumor extension carries a high risk of early tumor dissemination along the periureteral vascular and lymphatic supply. Improved survival of patients with stage T3 renal pelvis tumors versus ureteral tumors has been reported by several investigators (Batata and Grabstald, 1976; Guinan et al, 1992a; Park et al, 2004). Guinan and colleagues confirmed this observation among 611 patients treated at 97 hospitals and in a collection of 250 cases reported in the literature (Guinan et al, 1992a). The 5-year survival rates for patients with stage T3 tumors of the renal pelvis and ureter were 54% and 24%, respectively. In a multivariate analysis, patients with ureteral tumors had a higher local and distant failure rate than did those with renal pelvis tumors of the same stage and grade (Park et al, 2004). Renal pelvis and upper ureteral tumors spread initially to para-aortic and paracaval nodes, whereas distal ureteral tumors spread to pelvic nodes (Batata et al, 1975; Heney et al, 1981; Nocks et al, 1982; Mahadevia et al, 1983; McCarron et al, 1983;Jitsukawa et al, 1985; Geiger et al, 1986).
Treatment
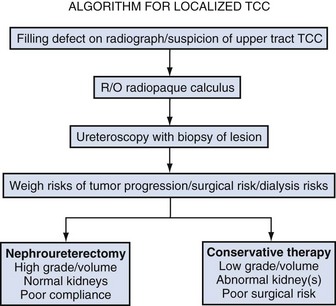
The treatment of upper tract urothelial tumors has undergone significant changes. The relatively low frequency of these lesions and the lack of prospective randomized trials do not permit absolute conclusions about treatment impact on outcomes. In the past, treatment recommendations were based, at least in part, on practical limitations in follow-up and detection of local disease recurrence. Technologic improvements in imaging and, most important, direct endoscopic visualization of all levels of the urinary tract allow earlier and more accurate initial diagnosis and treatment and improved follow-up. Treatment may be based primarily on the risk the tumor poses and on the efficacy of a specific treatment rather than on other considerations. The specific indications and techniques for each form of treatment (open vs. laparoscopic radical nephroureterectomy; open vs. retrograde endoscopic vs. percutaneous renal-sparing tumor ablation) are addressed later in this chapter. However, the following introductory considerations apply.
The least invasive and least ablative treatment necessary for safe control of the tumor is preferred. Most upper tract urothelial tumors are not large or bulky. Thus, laparoscopic surgery is ideal, at least for the renal portion of radical nephroureterectomy when the tumor warrants removal of the entire renal unit (see later). A variety of approaches with various combinations of laparoscopic and open techniques are employed for distal ureterectomy. Low-grade noninvasive upper tract tumors are managed initially by ablative renal-sparing surgery. Retrograde ureteroscopy and ureteropyeloscopy are preferred when tumor size, number, and access allow complete tumor ablation. Percutaneous antegrade tumor ablation is chosen when the anatomy and the tumor do not allow complete ablation through a retrograde approach.
Open Nephron-Sparing Surgery for Renal Pelvis Tumors: Pyelotomy and Tumor Ablation and Partial Nephrectomy
Indications
Open conservative surgery may be applied in selected cases when nephron sparing for preservation of renal function is required (Gittes, 1966, 1980; Petkovic, 1972a, 1972b; Mazeman, 1976; Johnson and Babaian, 1979; Babaian and Johnson, 1980; Cummings, 1980; Wallace et al, 1981; Tomera et al, 1982; McCarron et al, 1983; Zincke and Neves, 1984; Bazeed et al, 1986; Ziegelbaum et al, 1987; Messing and Catalona, 1998). Tumor in a solitary kidney, synchronous bilateral tumors, and predisposition to form multiple recurrences, as in endemic Balkan nephropathy, are all reasons to consider nephron sparing (Fig. 53–6). Pyeloscopy as an initial diagnostic component to open conservative surgery has been supplanted by retrograde or percutaneous antegrade renal endoscopy (Huffman et al, 1985). Direct endoscopic visualization of the lesion and biopsy with cup forceps or brush establish a definitive diagnosis and tumor grade (Gill et al, 1973). The brush biopsy has renewed value because tissue obtained by small cup biopsy forceps used through narrow-caliber flexible endoscopes is limited. Preoperative determination of the stage of renal pelvis tumors remains difficult. Large size, broad base, and nonpapillary pattern favor tumor invasiveness. These improvements in initial diagnosis and assessment of tumor stage and grade allow more specific treatment, including conservative surgery when it is indicated.
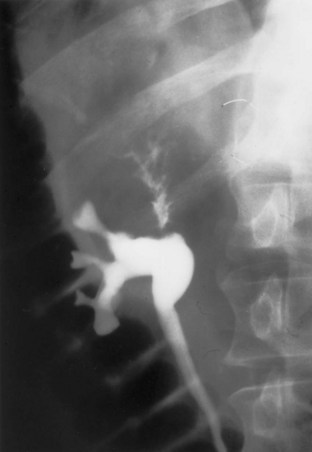
Figure 53–6 Patient with an invasive tumor of the upper calyx of a solitary kidney. The patient elected an upper pole partial nephrectomy.
The documented risk of wound implantation by tumor is low after open conservative surgery if simple precautions are followed to minimize spillage (Gittes, 1980; Tomera et al, 1982; McCarron et al, 1983). Modern percutaneous antegrade renal surgery allows resection of virtually any lesion formerly treated by open pyeloscopy, and the risk of tumor spillage is even lower (see following discussion).
Technique
Preoperative arteriography is not routinely obtained. However, it may be helpful before partial nephrectomy of rare hypervascular renal pelvis tumors. The affected segmental renal artery may be embolized or more easily identified at the time of surgery on the basis of the arteriogram.
The patient is placed in either a full flank or a torque position, as shown in Chapter 54. The full flank position provides optimal exposure to the renal pelvis for pyelotomy and excision of a large noninvasive tumor. The torque position allows both anterior and posterior exposure to the renal pedicle for partial nephrectomy. An extrapleural, extraperitoneal flank, or thoracoabdominal incision is made. Removal of a portion of the 11th or 12th rib provides maximal exposure to the kidney and aids in mobilization of the upper pole and in gaining access to the renal vessels. Removal of a rib is especially helpful in obese patients or in those with a high kidney. An incision off the tip of the 12th rib suffices in thin patients with a low kidney and is accompanied by less potential wound discomfort or morbidity. After the incision is completed, the Gerota fascia is opened posteriorly and the entire kidney is mobilized. The renal vessels and the ureter are each isolated with a vessel loop.
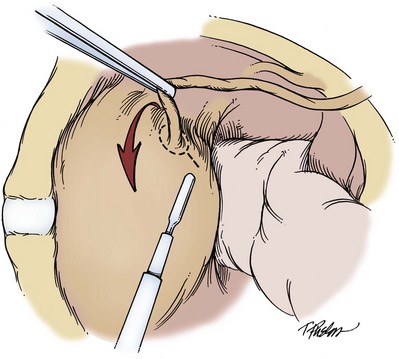
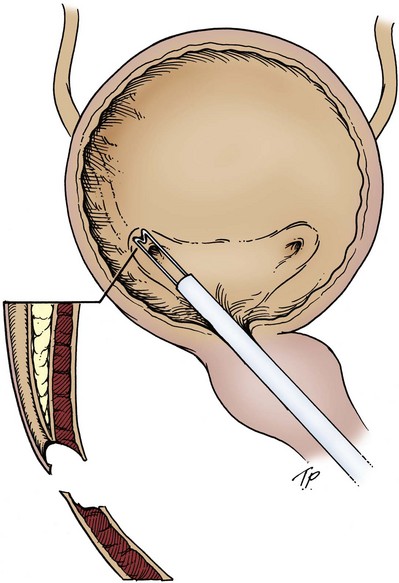
The wound is packed with dry sponges to isolate the kidney before any intrarenal incisions are made. For pyelotomy and tumor excision the renal pelvis and the major calyceal infundibula are exposed by dissecting the hilar and renal sinus fat as for an extended pyelolithotomy. A gentle curvilinear incision is made in the renal pelvis, and the tumor is excised; the base is cauterized with electrocautery, laser, or argon beam coagulator. Finally, the pelvis is closed with 4-0 chromic catgut or other absorbable suture.
The techniques of partial nephrectomy for renal pelvis tumors are essentially the same as those described in Chapter 54, with the added proviso of taking care to minimize the risk of tumor spillage. The involved segment of the intrarenal collecting system is clamped before removal of the tumor-bearing portion of the kidney is begun. The collecting system of the renal remnant is closed with absorbable 4-0 suture. Identified individual bleeding points are oversewn with 4-0 vascular Prolene or polydioxanone suture. Additional hemostasis is provided by use of electrocautery and the argon beam coagulator on the raw parenchymal surface. The edges of the renal defect and the adjoining renal capsule are approximated over Surgicel bolsters with 2-0 chromic catgut suture.
A suction drain is placed in the renal bed in all cases. A temporary urinary diversion stent is not used routinely unless difficulty is encountered in closing the collecting system.
Results
The reported overall risk of tumor recurrence in the ipsilateral renal pelvis after initial pyelotomy or partial nephrectomy varies from 7% to 60% (Mazeman, 1976; Murphy et al, 1980, 1981; Wallace et al, 1981; McCarron et al, 1983; Zincke and Neves, 1984; Ziegelbaum et al, 1987; Messing and Catalona, 1998). The risk of recurrence after conservative surgery increases with tumor stage from less than 10% for grade 1 to 28% to 60% for grades 2 and 3. The moderate to high risk of recurrence primarily reflects the inherent multifocal atypia and field change of the renal pelvis (Heney et al, 1981; Nocks et al, 1982; Mahadevia et al, 1983; McCarron et al, 1983). The possibility of incomplete initial treatment of the primary tumor cannot be totally excluded.
Estimates of overall and cancer-specific survival after conservative surgery of renal pelvis tumors are hampered by the lack of prospective, controlled, randomized trials and the small numbers of affected patients. The inherent bias introduced by selection of patients for conservative treatment based on medical comorbidities is another variable. Murphy and associates (1980) reported 5-year survival of 75% and 2-year survival of 46% after conservative surgery in patients with grade 1 and grade 2 renal pelvis tumors, respectively. McCarron and associates reported rates of cures, cancer-related deaths, and deaths due to unrelated causes of 33% each in nine patients who underwent conservative surgery (McCarron et al, 1983). Radical nephroureterectomy and dialysis still offer the best chance of cure and survival in patients with a large, invasive, high-grade, organ-confined renal pelvis tumor (T2N0M0) in a solitary kidney (Gittes, 1980; McCarron et al, 1983).
Open Radical Nephroureterectomy
Indications
Radical nephroureterectomy with excision of a bladder cuff is recommended for large, high-grade, invasive tumors of the renal pelvis and proximal ureter (Batata and Grabstald, 1976; Skinner, 1978; Babaian and Johnson, 1980; Cummings, 1980; Murphy et al, 1981; Nocks et al, 1982; McCarron et al, 1983; Richie, 1988; Williams, 1991; Messing and Catalona, 1998). Radical surgery also retains a role in treatment of medium-grade, noninvasive tumors of the renal pelvis and upper ureter when they are large, multifocal, or rapidly recurring despite maximal efforts at conservative surgery.
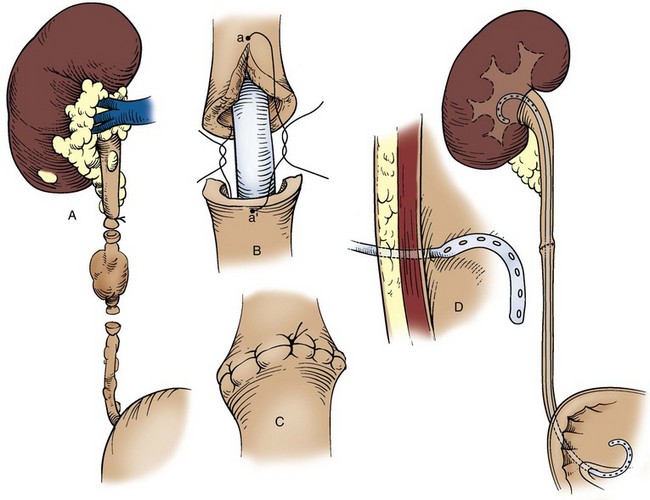
Technique
Choice of Incision
Radical nephroureterectomy may be performed through a variety of approaches. The choice of incision is largely one of the surgeon’s preference and is based on the surgeon’s experience, the patient’s body habitus and medical comorbidities, and the size of the kidney. The operation may be performed through one incision with the patient placed in a flank torque position, as shown in Chapter 54 and as described in the section on open nephron-sparing surgery (Fig. 53–7). In male patients the genitalia are included in the surgical field so that the bladder catheter may be accessed during the procedure. An extrapleural, extraperitoneal, thoracoabdominal incision is begun over the 11th or 12th rib or off the tip of the 12th rib, as described earlier. In contrast to the incision for partial nephrectomy, for which adequate exposure to the kidney is the primary concern, the lower ureter and the bladder must also be exposed for nephroureterectomy. Thus, the middle and anterior portions of the skin incision angle down toward the pelvis rather than in a subcostal direction (see Fig. 53–7). The peritoneum may be opened at any time to increase exposure as necessary. The thoracoabdominal approach offers the best exposure to the kidney but may cause a greater degree of postoperative atelectasis than the anterior subcostal approach. This may be an important consideration in patients with chronic lung disease.
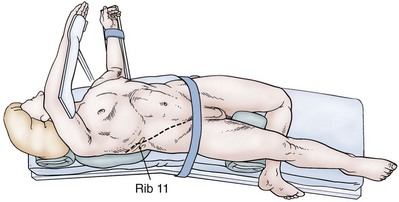
Figure 53–7 Patient in torque flank position for radical nephroureterectomy through a single thoracoabdominal incision. The shoulder girdle is rotated into a full flank position. The pelvic girdle is rolled back nearly to a supine position. All potential pressure points are carefully padded.
The operation may also be performed through a single long midline incision and intraperitoneal exposure. However, exposure to the kidney is limited from this approach, particularly for left-sided tumors.
Alternatively, the patient may be positioned supine with a small roll placed behind the rib cage on the affected side. The nephroureterectomy is performed through an upper anterior subcostal incision and a lower quadrant Gibson incision as for renal transplantation. A lower midline or Pfannenstiel incision may be substituted (Fig. 53–8). A lower midline incision avoids dividing muscles, is fast, and provides direct exposure to the prevesical space. However, a Gibson incision provides easier exposure to the distal ureter. Nephrectomy may be difficult from an anterior subcostal approach in obese patients.
Figure 53–8 Radical nephroureterectomy may be performed through a long midline incision or through a subcostal plus Gibson, lower midline, or Pfannenstiel incision. The patient is supine, and the table is mildly flexed. Placement of a small roll under the edge of the rib cage is helpful.
(From Sagalowsky AI. Renal transplantation and autotransplantation. In: Marshall F, editor. The textbook of operative urology. Philadelphia: WB Saunders; 1996:320–30.)
Radical Nephrectomy
The entire kidney, along with all the perinephric fat and the Gerota fascia, is fully mobilized (Figs. 53-9 and 53-10). The renal vessels are dissected and individually secured and divided in a standard manner. The authors prefer at least one 0 silk tie and 4-0 vascular Prolene suture ligature on an RB needle for each renal artery and renal vein. Combinations of ties plus large hemoclips may be substituted for the suture ligature. The adrenal is a not-infrequent site of metastasis for urothelial tumor. Traditionally, the ipsilateral adrenal gland has been included with the specimen. However, as in the case of renal cell carcinoma, adrenalectomy adds little to the cure for upper tract tumors. One may omit adrenalectomy if the gland appears normal on preoperative imaging studies and is grossly normal at the time of surgery and if the disease seems localized within the renal pelvis. However, adrenalectomy is prudent for maximal control if locally advanced disease is discovered.
Management of Distal Ureter and Bladder Cuff
Complete distal ureterectomy with a bladder cuff is necessary with nephroureterectomy and represents a distinct portion of the case, whether an open or laparoscopic approach to the kidney is used. General principles include intact complete ureteral excision with controlled occlusion of the ureteral orifice. The entire distal ureter, including the intramural portion and the ureteral orifice, is removed. Failure to completely remove the entire distal ureter and ureteral orifice is associated with a high rate of tumor recurrence. The ureter may be kept in continuity with the kidney to obviate the risk of tumor spillage from the lumen. However, the attached kidney is cumbersome, and this technique is not necessary as long as the distal ureter is divided between ties or clips at a location that is free of gross abnormality. Following are the options available for management of the distal ureter, whether an open or laparoscopic approach is used. If a laparoscopic approach is chosen, the incision can be placed to facilitate both intact specimen extraction and dissection of the distal ureter. One should keep in mind that TCC is a tumor that has the potential to seed nonurothelial surfaces and that maintenance of a “closed system” is important, especially with high-grade tumors.
Traditional Open Distal Ureterectomy
This operation can be done through Gibson, low midline, and Pfannenstiel incisions, depending on the patient’s anatomy and the extent of proximal ureteral dissection required. Transvesical, extravesical, and combined approaches have been described. The transvesical approach is the most reliable approach for ensuring complete ureteral excision. An anterior cystotomy may be made and intravesical and extravesical dissection performed as for ureteral neocystostomy (Fig. 53–11); 1 cm of bladder mucosa is included circumferentially around the ureteral orifice. The defect in the bladder wall at the ureteral hiatus is closed in two layers from within the bladder with interrupted 2-0 or 3-0 absorbable suture on the muscle and 4-0 suture on the mucosa. The anterior cystotomy is closed carefully in two layers with running 3-0 absorbable suture. The urethral catheter is maintained for 5 to 7 days; a suprapubic catheter is not routinely used. A flat suction drain is left in the perivesical space.
Figure 53–11 Complete distal ureterectomy with bladder cuff is performed by combined extravesical and transvesical dissection.
Complete distal ureterectomy may be performed entirely by extravesical (Fig. 53–12) dissection of the distal ureter and the intramural portion within the bladder wall all the way to the ureteral orifice. The need for a cystotomy is avoided with this approach. The ureter is tented up, and the bladder mucosa is divided between clamps. The defect in the bladder wall is closed in layers, as described previously. Care must be taken to ensure a complete dissection of the intramural portion of the ureter and to avoid contralateral injury from excessive traction.
Transurethral Resection of the Ureteral Orifice
This approach, also referred to as the pluck technique, should be reserved for proximal, low-grade tumors (Abercrombie et al, 1988; Palou et al, 1995). The patient is placed in the lithotomy position, and the ureteral orifice and intramural ureter are aggressively resected to the extravesical space (Fig. 53–13). The patient is then positioned for the nephrectomy portion of the procedure, in which dissection must extend down to the perivesical space. The advantage is avoidance of a second incision in performing an open nephroureterectomy. Walton and coworkers (2009) showed equivalent oncologic outcomes in a long-term retrospective study comparing this to traditional bladder cuff techniques. The advantages are less apparent with laparoscopic nephrectomy, in which an incision is necessary for specimen extraction, and this can be used for dissection of the distal ureter. In addition there is exposure of nonurothelial surfaces, which may predispose to local recurrence, especially with more dysplastic tumor (Jones and Moisey, 1993; Arango et al, 1997).
Intussusception (Stripping) Technique
Several variations of this procedure have been described (McDonald, 1953; Clayman et al, 1983; Roth et al, 1996; Angulo et al, 1998). Basically, a ureteral catheter is placed in the ipsilateral ureter at the beginning of the procedure. The nephrectomy portion of the procedure is carried out, and the ureter is dissected as far distally as possible. The ureter is then ligated and transected with the ureteral catheter secured to the proximal end of the distal ureter (Fig. 53–14). After nephrectomy, the patient is moved to the lithotomy position; the ureter is intussuscepted by traction into the bladder, and a resectoscope is used to excise the bladder cuff and free the ureter from the bladder. Concerns with this technique are presented by Giovansili and associates (2004), who described a failure rate of 18.7% in which there may have been disruption of the ureter and the need for an additional surgical incision.
Figure 53–14 With the intussusception technique, a ureteral catheter is placed at the beginning of the case. After nephrectomy the ureter is divided and the catheter is secured to the distal portion of the ureter. The patient is moved to the lithotomy position, and the ureter is intussuscepted into the bladder with retrograde traction. A resectoscope is used to excise the attached orifice.
Transvesical Ligation and Detachment Technique
This technique most closely mimics the open technique. Before the nephrectomy portion of the procedure, the patient is placed in the low lithotomy position, a ureteral catheter is placed, and two suprapubic 3-mm trocars (Fig. 53–15) are placed transvesically. The ureter is tented up, and a loop ligature is placed around the catheter and the orifice, creating a “closed” system. A Collins knife circumscribes the ureteral orifice, and further dissection is facilitated by the grasper, which provides traction until the extravesical space is reached. There has been excellent clinical success reported with this technique (Gill et al, 1999), but it can be technically difficult. Agarwal and associates (2008) described a technique that appears similar but dissects the ureter only to the extravesical space.
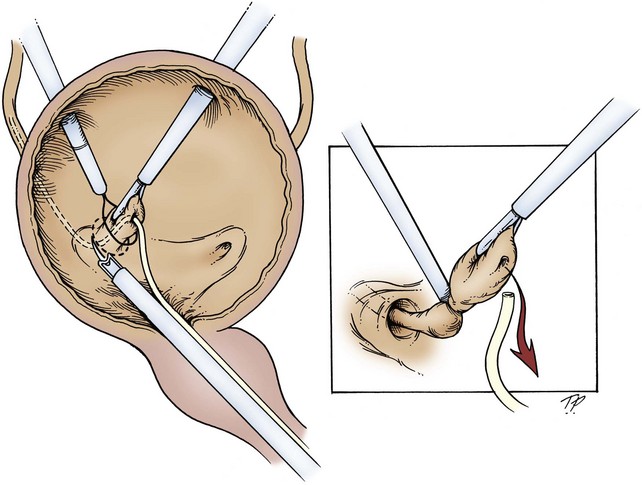
Figure 53–15 A ureteral catheter is placed, and two laparoscopic ports are placed transvesically. The ureteral orifice is tented up; a loop is placed around the orifice to occlude the opening and to place traction on the ureter. A Collins knife then facilitates the dissection to the extravesical space.
Total Laparoscopic Technique
Flexible cystoscopy is performed before the laparoscopic portion of the case, and a Bugbee electrode is used to cauterize the orifice and intramural ureter completely. Cauterization of the intramural portion of the ureter is important for identification of the distal limits of the dissection during the laparoscopic portion of the procedure. A 20- or 22-Fr Foley catheter is placed before proceeding to the laparoscopic portion. The nephrectomy and ureteral dissection are carried down to the detrusor muscle. Antegrade traction is placed on the ureter, everting the orifice out of the bladder, and an endovascular stapling device is placed as distally as possible (Fig. 53–16). A flexible cystoscope can be placed at this time if there is any question of the adequacy of the specimen or concern of disturbance of the contralateral ureteral orifice. The endovascular stapling device is then deployed, laying two rows of staples on the bladder and specimen sides. This allows a closed system after division of the ureter.
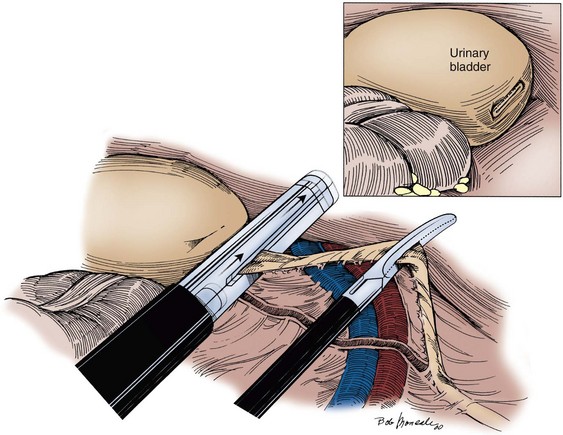
Figure 53–16 The ureter is dissected extravesically to the ureteral orifice. Lateral traction is placed on the ureter, everting the orifice, and the endovascular stapling device is placed at the distal margin, providing simultaneous ligation and division of the distal ureter at the level of the bladder. A cystoscope can be placed to ensure that the entire ureter is removed.
Management of Distal Ureter—Considerations
Much controversy surrounds the management of the distal ureteral cuff during nephroureterectomy. No study has shown a clear advantage of any single technique (Macejko et al, 2008). However, it is universally accepted that the ureter must be removed in its entirety. Failure to do so places the patient at an unacceptable risk for distal recurrence, especially with high-grade tumors. Because of the rarity of the disease and other factors it is unlikely that a study will be designed to resolve this issue. There is some concern of the use of techniques that violate the closed system, especially with high-grade/stage and lower ureteral tumors. In addition, total laparoscopic techniques appear to be associated with a higher local recurrence rate, possibly due to retained ureter in the bladder (Romero et al, 2007). When considering an approach, the surgeon should take into account tumor location, potential for seeding, patient factors, and personal experience.
Lymphadenectomy
Regional lymphadenectomy is included with radical nephroureterectomy. For renal pelvis and proximal or middle ureteral tumors the ipsilateral renal hilar nodes and the adjacent para-aortic or paracaval nodes are resected. This dissection adds little time or morbidity to the surgery. On occasion, lymphadenectomy is more hazardous in patients with severe atherosclerosis of the aorta or grossly positive and fixed nodes. Node dissection is limited or omitted in these cases.
Key Point: Lymphadenectomy with Radical Nephroureterectomy
Results
The outcome strongly correlates with tumor stage and grade as summarized in Tables 53-2 and 53-3, compiled from numerous series. The role of complete distal ureterectomy with excision of a bladder cuff in radical nephroureterectomy for upper tract tumors is well established. The risk of tumor recurrence in a remaining ureteral stump is 30% to 75% (Bloom et al, 1970; Strong et al, 1976; Johansson and Wahlquist, 1979; Babaian and Johnson, 1980; Kakizoe et al, 1980; Mullen and Kovacs, 1980; McCarron et al, 1983). This risk is likely to remain or even to increase as the use of radical nephroureterectomy becomes more selective for patients, mainly those with higher risk tumors. All patients who underwent an extravesical dissection and tenting up of the ureter in the series by Strong and associates (1976) retained a portion of the ureter and the ureteral orifice; this demonstrates that the transvesical dissection of the ureter must be thorough to avoid such an error. Complete ureterectomy with a bowel cuff should accompany nephroureterectomy of a renal unit draining into a urinary diversion. Mufti and coworkers (1988) reported a tumor recurrence rate of 37.5% when the ureteroenteric anastomosis was not removed.
The need or benefit of radical nephroureterectomy versus simple nephrectomy or conservative nephron-sparing surgery is less well defined. Radical nephroureterectomy is recommended as providing optimal chance for survival in numerous series (Batata et al, 1975; Johansson and Wahlquist, 1979; Murphy et al, 1980; McCarron et al, 1983; Zungri et al, 1990). Batata and colleagues (1975) reported that radical nephroureterectomy provided a 5-year survival rate of 23% among patients with locally advanced disease (stage T3 to T4, N1 to N2). Johansson and Wahlquist (1979) reported survival advantage at 5 years for radical versus simple nephrectomy of 84% versus 51%, respectively, and of 74% versus 37%, respectively, for patients with high-stage tumors only. McCarron and associates (1983) reported that the subgroup of patients with large, high-grade, but organ-confined disease obtained the most benefit from radical versus conservative surgery. The death rate due to tumor was 29% versus 89% for the radical and conservative surgery groups, respectively. In the series reported by Murphy and associates (1980), radical surgery conferred a survival advantage over conservative surgery at 5 years of 88% versus 75% for grade 1 tumors and of 90% versus 46% for grade 2 tumors. The retrospective, uncontrolled nature of all these series limits the conclusions that may be drawn. In summary, radical nephroureterectomy is warranted for patients with high-grade invasive disease that is organ confined (stage T1 to T2, N0) or is only locally advanced (stage T3 to T4, N0 to N2, M0). Tumor-free survival rates are high after radical nephroureterectomy for organ-confined disease and are guarded to poor for locally advanced disease. Treatment decisions in patients with compromised renal function must balance the potential beneficial effect of radical surgery on control of the tumor versus the morbidity, mortality, and quality of life risks associated with dialysis.
Regional lymphadenectomy is a recommended accompaniment of radical nephroureterectomy for upper tract tumors in multiple reports (Grabstald et al, 1971; Batata et al, 1975; Batata and Grabstald, 1976; Skinner, 1978; Johansson and Wahlquist, 1979; Babaian and Johnson, 1980; Cummings, 1980; Heney et al, 1981; McCarron et al, 1983; Richie, 1988; Williams, 1991; Messing and Catalona, 1998; Brausi et al, 2007; Kondo et al, 2007; Abe et al, 2008). However, the authors in nearly every one of these series find little therapeutic value from lymphadenectomy, and almost every patient with node-positive disease develops early distant metastases. In the series by Batata and Grabstald (1976), 5-year survival was seen in 23% of patients with completely resected stage T3 to T4, N1 to N2 disease. One of the limitations in assessing the role of lymph node dissection for radical nephroureterectomy is that, as with the treatment of renal cell carcinoma, the lymphatic drainage for renal pelvis tumors is not precise and thus the boundaries of lymphadenectomy are unclear. Investigators in recent series have readdressed these issues. Kondo and colleagues (2007) compared outcomes after radical nephroureterectomy for 45, 36, and 88 patients who underwent complete regional, incomplete regional, or no lymphadenectomy, respectively. Overall cancer-specific survival did not correlate with more extensive lymphadenectomy. However, with multivariate analysis a statistically significant benefit was seen with complete lymphadenectomy and higher lymph node counts for patients with stage pT3-4 disease. In a recent retrospective, uncontrolled series Brausi and associates (2007) compared outcomes in 82 patients with stage pT2-4 upper tract tumors who underwent lymphadenectomy with removal of more than five nodes at the time of radical nephroureterectomy or no lymphadenectomy in 44 and 42 cases, respectively. In both univariate and Cox proportional hazard analysis the T stage and performance of lymph node dissection correlated with disease recurrence and disease-specific survival. In a larger contemporary series, Abe and associates (2008) compared outcomes in 166 patients and 146 patients who did or did not undergo lymphadenectomy, respectively, at the time of renal surgery for upper urinary tract tumor. In multivariate analysis, pathologic lymph node status, as well as grade and stage, correlated with cause-specific survival. Among the group who underwent lymphadenectomy survival was similar for those with fewer than six and more than six nodes removed. Survival for patients with resected negative nodes was higher than for those who did not undergo lymphadenectomy (stage pNx). The rationale for continuing regional lymphadenectomy is that it adds little time or morbidity to the surgery, is important for prognosis, and may occasionally have therapeutic value. Earlier diagnosis in contemporary experience may identify more patients with limited nodal metastases who might benefit from radical surgery. Patients with proven nodal disease are obvious candidates for more careful follow-up and for trials exploring the role of adjuvant therapy.
Laparoscopic Radical Nephroureterectomy
The indications for laparoscopic nephroureterectomy are the same as those for open nephroureterectomy. Laparoscopic nephroureterectomy can be performed by transperitoneal, retroperitoneal (Hattori et al, 2005), and hand-assisted approaches (Landman et al, 2002; Stifleman et al, 2000). All show a significant decrease in morbidity compared with an open surgical approach for appropriately selected patients. All laparoscopic techniques involve two distinct portions of the procedure: nephrectomy and excision of the distal ureter with intact specimen extraction for accurate staging. Management of the distal ureter is described previously in the chapter. One should bear in mind several factors with laparoscopic nephroureterectomy, including the risk of tumor seeding from both the ureter and the bladder. For these reasons, removal of an intact specimen is desirable. The incision should be strategically placed for both extraction of the specimen and dissection of the distal ureter. Because an incision is necessary regardless of the approach chosen, some techniques for avoidance of a second incision for the distal ureter described previously have less utility.
Technique
Transperitoneal Laparoscopic Nephroureterectomy
Laparoscopic Removal of Kidney Down to Midureter
The patient is placed supine with the ipsilateral hip and shoulder rotated approximately 20 degrees (Fig. 53–17). The patient is secured to the table and can be easily moved from the flank position (nephrectomy portion) to the modified supine position (open portion) by rotating the operative table. The ipsilateral flank and urethra are prepared and draped, and a Foley catheter is placed before insufflation of the abdomen.
Figure 53–17 The patient is positioned on the table in a modified lateral decubitus position with the ipsilateral flank rotated up 15 degrees. The patient is secured to the table at the chest, waist, and lower extremity. This setup allows the patient to be moved to the full flank or supine position with simple rotation of the operating table.
(From Jarrett TW. Laparoscopic nephroureterectomy. In: Bishoff JT, Kavoussi LR, editors. Atlas of laparoscopic retroperitoneal surgery. Philadelphia: WB Saunders; 2000. p. 105.)
The abdomen is insufflated, and three or four trocars are placed as outlined in Figure 53–18, with the first usually being the lateral trocar. Subsequent trocars are placed under direct vision. With this configuration, the camera is kept at the umbilicus for the entire procedure. The upper midline and lateral trocars are used by the surgeon for the dissection of the kidney and the proximal half of the ureter. The lower midline and lateral trocars are used for the dissection of the distal ureter. A 3-mm trocar just below the xiphoid can be helpful in retracting the spleen and liver for left- and right-sided lesions, respectively. The exception is with obese patients, when shifting of the trocars may be necessary to provide optimal visualization (Fig. 53–19). If a hand-assist approach is chosen, the hand port site should be placed so that it can be used for the dissection of the distal ureter and open bladder cuff as indicated.
Figure 53–18 Port configuration for laparoscopic-assisted nephroureterectomy. Three ports are generally used for the kidney and upper ureteral dissection. A fourth midline port between the umbilicus and symphysis can be placed, if needed, for further ureteral dissection. The incision is then strategically placed to allow the distal ureteral dissection and specimen removal. The choice of incision largely depends on patient factors and level of dissection reached during the laparoscopic portion of the procedure. A low abdominal (midline or Pfannenstiel) incision is favored if the dissection is below the iliac vessels. A Gibson-type incision will give exposure of the more proximal ureter, if necessary.
Figure 53–19 For obese patients undergoing laparoscopic-assisted nephroureterectomy the trocars are shifted laterally to accommodate the increased distance from the kidney.
The table is rotated so the patient is in the flank position. The peritoneum is incised along the white line of Toldt from the level of the iliac vessels to the hepatic flexure on the right and to the splenic flexure on the left. The colon is moved medially by releasing the renocolic ligaments while leaving the lateral attachments of the Gerota fascia in place to prevent the kidney from “flopping” medially. The colon mesentery should be mobilized medial to the great vessels to facilitate dissection of the ureter, renal hilum, and local lymph nodes as needed.
Proximal Ureteronephrectomy
The proximal ureter is identified, just medial to the lower pole of the kidney, and dissected toward the renal pelvis. If an invasive ureteral lesion is suspected, the dissection should include a wide margin of tissue. The renal hilum is identified, and its vessels are exposed with a combination of blunt and sharp dissection. The artery is ligated and divided by use of a gastrointestinal stapling device with a vascular load or multiple clips. The renal vein is then divided in a similar fashion (Fig. 53–20). With vascular control ensured, the kidney is dissected free, either inside or outside the Gerota fascia, depending on the tumor location and stage. For upper pole lesions thought to invade the parenchyma, the ipsilateral adrenal should be removed.
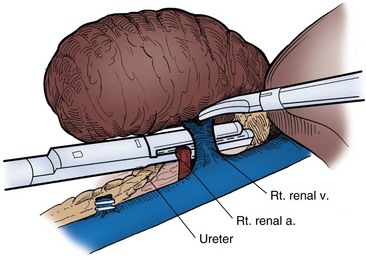
Figure 53–20 The vessels of the renal hilum are carefully dissected, and the endovascular stapling device, with a vascular load, is used to simultaneously ligate and divide the vessels in a controlled environment.
(From Jarrett TW. Laparoscopic nephroureterectomy. In: Bishoff JT, Kavoussi LR, editors. Atlas of laparoscopic retroperitoneal surgery. Philadelphia: WB Saunders; 2000. p. 112.)
The ureteral dissection is continued distally, keeping in mind that the ureteral blood supply is generally anteromedially located in the proximal third, medially located in the middle third, and laterally located in the distal third. Dissection of the lower half may require placement of the fourth trocar. In the area of primary disease, surrounding tissue should be left to provide an adequate tumor margin. The ureteral dissection is continued as far as is technically feasible. If the distal limits of the dissection are below the level of the iliac vessels, the remainder of the procedure can easily be completed through a lower abdominal incision. The specimen is placed in the pelvis, and the renal bed is inspected meticulously for bleeding. At this time, the 10-mm port sites are closed before proceeding to the open portion of the case.
Open Distal Ureterectomy with Excision of Bladder Cuff
The patient is now moved to the supine position, which can usually be done without re-preparation, and a low midline Pfannenstiel or Gibson incision is made (see Fig. 53–18). The choice of incision largely depends on the tumor location, the body habitus of the patient, and the most caudal level of ureteral dissection attained during the laparoscopic portion. The Gibson incision is preferable when the distal ureter cannot be freed laparoscopically to the level of the iliac vessels. The open distal ureterectomy is described in the section on open techniques.
Dissection of the Distal Ureter
If one is to consider a total laparoscopic procedure or to minimize the open distal portion, the ureteral dissection needs to continue to the level of the bladder. The patient is placed in the Trendelenburg position to move the bowel contents out of the pelvis. The peritoneal incision is extended from the level of the iliac vessels into the pelvis lateral to the bladder and medial to the medial umbilical ligament (Fig. 53–21). The vas deferens in male patients and the round ligament in female patients is clipped and divided. The ureter can now be traced between the bladder and the medial umbilical ligament down to its origin at the bladder. The bladder cuff may be dissected extravesically, freeing the ureter from the surrounding detrusor muscle. Flexible cystoscopy at this point may be helpful in confirming complete ureterectomy and patency of the contralateral ureteral orifice.
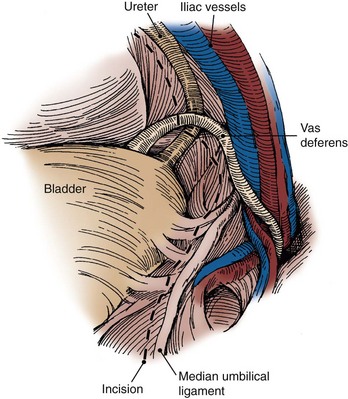
Figure 53–21 The peritoneal incision is continued below the iliac vessels medial to the median umbilical ligament and lateral to the bladder. The vas deferens is divided between clips in the male patient. In the female patient the round ligament is divided, giving full exposure of the distal ureter to the bladder.
Results
The first laparoscopic nephroureterectomy was performed in 1991 by Clayman and associates. Since that time the technical aspects and safety of laparoscopic procedures have been well established. There are multiple published series of laparoscopic nephroureterectomy with varying techniques (Chung et al, 1996; Keeley and Tolley, 1998; Salomon et al, 1999; Shalhav et al, 2000; Jarrett et al, 2001; Stifleman et al, 2001; Bariol et al, 2004; Hsueh et al, 2004; Matin and Gill, 2005; Wolf et al, 2005). Each varies with regard to approach (transperitoneal vs. retroperitoneal), management of the distal ureter by open removal, transurethral resection, and total laparoscopic management. As with other laparoscopic renal procedures there is no clear-cut benefit of any one approach with regard to morbidity, cosmesis, or return to activity. All, however, show a benefit with regard to morbidity compared with open surgery.
The efficacy of laparoscopic nephroureterectomy is being established for cancer control. With intermediate and long-term follow-up, cancer-related outcomes appear comparable to those of the open counterpart (McNeill et al, 2000). El Fettouh and colleagues (2002), in a multi-institutional study with 116 patients, showed the local and bladder recurrence rates to be 2% and 24%, respectively. The rate of distant metastasis was 9%, and positive margins were seen in 4.5% of cases. More recently, Berger and associates (2008) showed a 5-year cancer-specific survival of 80%, 70%, 68%, 60% and 0% for stage Ta/Tis, T1, T2, T3, and T4 lesions, respectively. Schatteman and associates (2007) similarly showed cancer-specific survival rates of 100%, 86%, 100%, 77%, and 0% for stage Ta, T1, Tis, T3, and T4 lesions. In both studies there was a worsening prognosis with increasing tumor stage. Long-term data are available from Muntener and associates (2007a), who studied 37 patients with follow-up of 60 to 148 months. In this study, 11 patients had disease progression and died of TCC 7 to 59 months after the operation. Tumor stage was the only factor significantly associated with disease recurrence. Waldert and coworkers (2009) compared open to laparoscopic outcomes and showed no significant difference in cancer outcomes and the advantage of lower morbidity for well-selected patients.
Local recurrence and port-site seeding are major concerns. There have been five reported instances of port-site seeding involving TCC of the upper urinary tract. Two of these cases were discovered after simple nephrectomy for presumed benign disease in which the principles of surgical oncology were inadvertently not followed (Ahmed et al, 1998; Otani et al, 1999). Three instances were for intended nephroureterectomy for high-grade disease (Ong et al, 2003; Matsui et al, 2004; Barrett, verbal communication, September 1998). Muntener and associates (2007b) reported a single case of local recurrence among 166 cases. In this instance there was obvious violation of the ipsilateral urinary tract, noted perioperatively. Although the potential for seeding exists, it does not appear any higher than that for the open surgical counterpart as long as good surgical principles are followed.
In summary there does not appear to be a significant difference between laparoscopic and open nephroureterectomy when the principles of surgical oncology are followed. Management of the bladder cuff still has shown variability and a tendency toward higher recurrences with minimally invasive approaches. Lymphadenectomy can be performed laparoscopically and should be utilized based on the clinical situation.
Open Segmental Ureterectomy
Ureteroureterostomy
Indications
Segmental ureterectomy and ureteroureterostomy are indicated for noninvasive grade 1 and grade 2 tumors of the proximal ureter or midureter that are too large for complete endoscopic ablation and for grade 3 or invasive tumors when nephron sparing for preservation of renal function is a goal.
Technique
The patient is placed in a modified flank position for lesions of the proximal ureter or in a supine position with a roll under the ipsilateral lumbar space for midureteral tumors. An angular extraperitoneal incision from the tip of the 12th rib toward the pelvis provides good exposure to the midureter. The ureter is identified, mobilized, and secured with vessel loops proximally and distally. The approximate location of the lesion is known from preoperative imaging and ureteroscopy. The tumor is palpated, and the ureter is ligated 1 to 2 cm proximal and distal to the tumor (Fig. 53–22A). The involved ureteral segment is excised and sent for frozen-section analysis to confirm the diagnosis, grade, and stage and to verify that both ends of the ureter are tumor free. A regional lymphadenectomy is performed. The ureter is repaired by mobilizing both ends to allow a tension-free anastomosis. A defect of up to 4 cm is amenable to ureteroureterostomy in most cases. The entire kidney may be mobilized downward to provide additional length to the proximal ureter when necessary. The periureteral blood supply is preserved as the ureter is mobilized. The ureteral ends are spatulated and anastomosed with interrupted absorbable 4-0 sutures (see Fig. 53–22B and C). A ureteral stent is placed as the anastomosis is in progress, and a closed-suction drain is placed in the retroperitoneal space near but not touching the repair (see Fig. 53–22D).
Distal Ureterectomy and Direct Neocystostomy or Ureteroneocystostomy with a Bladder Psoas Muscle Hitch or a Boari Flap
Indications
Complete distal ureterectomy is recommended for tumors in the distal ureter that cannot be removed completely by endoscopic means (Bloom et al, 1970; Johnson and Babaian, 1979; Babaian and Johnson, 1980; Pagano, 1984; Johnson et al, 1988; Anderstrom et al, 1989; Williams, 1991; Messing and Catalona, 1998). Larger, high-grade, organ-confined, invasive lesions comprise the majority of these tumors.
Technique
The patient is placed in the supine position. It is desirable to include the genitalia in the surgical field so that a bladder catheter may be placed and accessible during the procedure. A Gibson, Pfannenstiel, or lower midline incision provides good exposure to the distal ureter and bladder for the ureteral excision and the repair. The distal ureterectomy is performed as described in the section on radical nephroureterectomy. Extra care is warranted to avoid spillage of known tumor in the distal ureteral segment.
The choice of a nonrefluxing or refluxing anastomosis is controversial (Pagano, 1984). The nonrefluxing anastomosis may limit infections as well as seeding of the upper tract with tumors from the bladder. The refluxing anastomosis may facilitate follow-up surveillance of the upper tract with imaging studies and ureteroscopy.
Direct ureteroneocystostomy may be possible if only a short segment of the juxtavesical and intramural ureter is removed. Additional techniques are required when the entire distal ureter is removed. The first choice is a bladder psoas muscle hitch. The superior vesical attachments on the opposite side of the bladder are freed. An anterior cystotomy is made, and the bladder is mobilized upward over the iliac vessels to the psoas muscle at the level of the iliac crest (Fig. 53–23A and B). The bladder is secured to the psoas muscle and tendon with a row of interrupted 2-0 chromic sutures. Entrapment of the genitofemoral nerve is avoided. The ureteral end is spatulated and brought through an opening in the uppermost portion of the bladder. Angulation must be avoided at the ureteral hiatus. A nonrefluxing anastomosis is performed by placing the ureter in a mucosal trough or tunnel if there is sufficient ureteral length (see Fig. 53–23C). The anastomosis is completed with interrupted 4-0 chromic sutures. A ureteral stent, a urethral catheter, and a closed-suction drain in the perivesical space are placed in all cases (see Fig. 53–23D). The anterior cystotomy is closed in two layers with 3-0 absorbable suture. Insertion of a suprapubic catheter is optional.
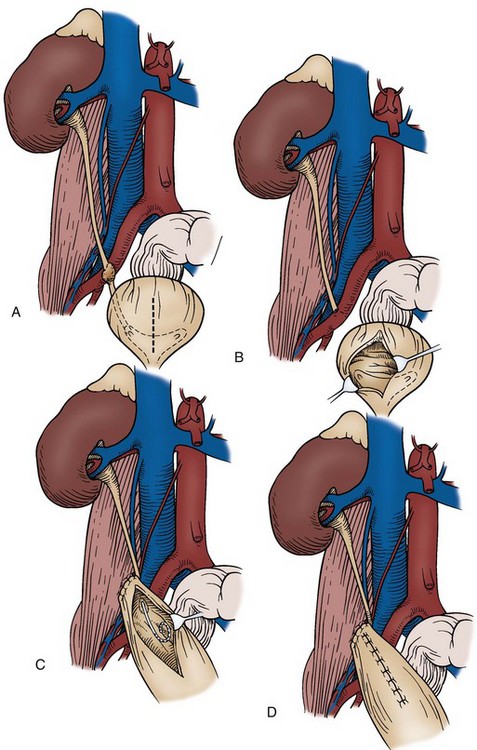
Figure 53–23 A and B, Distal ureterectomy for invasive tumor of the distal ureter. C, The bladder is advanced and secured to the psoas muscle. The ureter is anastomosed to the posterior upper aspect of the bladder hitch. D, The anterior cystotomy is closed. A closed-suction drain, ureteral stent, and bladder catheter are placed (not shown).
The bladder psoas muscle hitch provides considerable length if the bladder is of normal size and compliance. However, if an even greater gap must be bridged, a Boari flap from the anterior bladder wall combined with a psoas muscle hitch will reach the proximal ureter (Figs. 53-24 and 53-25). If the need for a Boari flap is anticipated, the incisions for the flap should be marked before making the cystotomy and psoas hitch. The spatulated end of the ureter is anastomosed to the cephalad end of the bladder flap, as described previously. The bladder tube is folded and closed in two layers with running 3-0 absorbable suture.
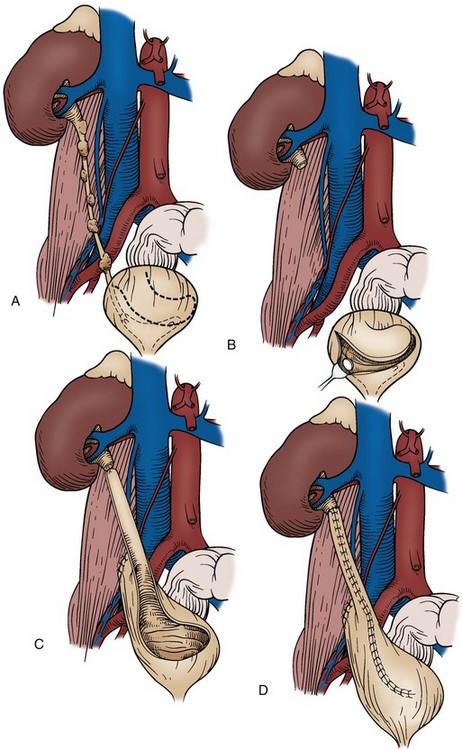
Figure 53–24 A, Subtotal ureterectomy required for nephron sparing in a patient with multiple diffuse ureteral tumors. B, A spiral flap is fashioned from the anterior bladder wall. C, The psoas hitch plus Boari flap reaches the remaining proximal ureter. D, Completed anastomosis and bladder closure. The ureteral stent, bladder catheter, and closed-suction drain are not shown.
Subtotal Ureterectomy
Indications
Some tumors may require removal of long segments of the ureter by virtue of multifocality and number. However, the tumor grade and stage may not warrant nephroureterectomy. In addition, nephron sparing may be necessary because of compromised function of the contralateral kidney.
Technique
Complete ileal ureteral substitution may be performed as described in Chapter 85. The proximal ileal segment is anastomosed to the renal pelvis, and the distal ileum is connected to the bladder. Ileal interposition may be performed to repair a long midureteral defect when neither ureteroureterostomy nor bladder mobilization is feasible. A suitable length of ileum to bridge the gap is selected. The ileum is narrowed over a 14-Fr catheter held along the mesenteric edge of the bowel to approximate the diameter of the ureter. Discarding excess bowel from the antimesenteric edge decreases problems with mucus plugging, preserves intestinal blood supply, and allows more efficient peristalsis of urine across the ileal segment. The vermiform appendix has also been used for segmental ureteral substitution (Goldwasser et al, 1994). A preoperative bowel preparation is indicated when possible use of intestine is anticipated.
Finally, total ureterectomy, renal autotransplantation to the iliac fossa, and pyelocystostomy are options when the entire ureter is involved with multiple tumors (Pettersson et al, 1984). However, modern endoscopic techniques provide less invasive means of tumor control with lower morbidity in most cases.
Results
In the past, some authors recommended radical nephroureterectomy for all patients with upper tract urothelial tumors (Skinner, 1978). Others suggested segmental ureterectomy only for patients with low-grade, noninvasive tumors of the distal ureter (Babaian and Johnson, 1980). The cumulative results from the series in Tables 53-2 and 53-3 for patients with upper tract tumors of the renal pelvis or ureter reveal that outcome strongly correlates with tumor stage and grade. Moreover, the results are similar for radical or nephron-sparing surgery. The outcomes for only ureteral tumors are shown in Tables 53-4 and 53-5. Again, overall survival strongly correlates with stage and grade regardless of the extent of surgical treatment. Overall 5-year survival is excellent for patients with grade 1 and grade 2 noninvasive ureteral tumors. Five-year survival remains approximately 50% for patients with stage T2 disease but falls dramatically for stage T3. McCarron and associates (1983) reported 5-year survival of 64% for patients with stage Ta tumors treated by either segmental ureterectomy or endoscopic tumor ablation. In the same series, 5-year tumor-free survival was 66% and 50% for stage T1 and T2 tumors, respectively, treated with segmental or distal ureterectomy. Anderstrom and colleagues (1989) reported no tumor-related deaths and only one recurrence among 21 patients treated with segmental ureterectomy for low-grade, noninvasive ureteral tumors who were observed for a median of 83 months.
Booth and associates (1980) reported 5-year survival of 72% versus 31% among 144 patients with ureteral tumors having either superficial or deep ureteral muscle invasion, respectively. This observation has not been duplicated in other series. The concept is of doubtful validity owing to the thin nature of the ureteral wall.
One of the few reports of 5-year cancer-specific survival by stage for patients with ureteral tumors comes from Grabstald and coworkers (1971). Disease-specific survival was 64% and 100% for stage Ta to T1 and stage T2 disease, respectively. All deaths were due to unrelated causes. In contrast, for patients with stage T3 disease, cancer-specific survival was only 7% and the death rate due to tumor was 87%.
The risk of ipsilateral recurrence after conservative treatment of ureteral tumors is 33% to 55% (Mazeman, 1976; Johnson and Babaian, 1979; Babaian and Johnson, 1980; McCarron et al, 1983; Williams, 1991). Most recurrences are distal to the original lesion, but proximal recurrences are also seen (Strong et al, 1976). The risk for recurrence and the need for follow-up are lifelong (Herr, 1998). Grossman (1978) reported a stage T2 local recurrence in a patient 16 years after segmental ureterectomy and ureteroureterostomy for a grade 1, stage Ta, midureteral tumor. The authors have seen a recurrence of a grade 2, stage Ta tumor in the distal ureter 18 years after segmental ureterectomy of a grade 1, stage Ta tumor of the proximal ureter (Sagalowsky, unpublished data, 1999).
The following are general recommendations for treatment of ureteral tumors in patients with good contralateral renal function. Segmental ureterectomy is offered for grade 1 to grade 2, stage Ta to stage T1 tumors of the proximal ureter or midureter that are not amenable to complete ablation by endoscopic means because of size or multiplicity. Segmental excision also is offered for grade 1 to grade 2, stage T2 tumors of the proximal ureter or midureter. Distal ureterectomy and neocystostomy are offered for low-grade, low-stage tumors that are not controlled by endoscopic means. Ureteral excision may also be suitable for some high-grade, locally invasive tumors of the distal ureter.
Laparoscopic Distal Ureterectomy and Reimplant
Various laparoscopic techniques for distal ureterectomy and reimplant have been reported (Rouprêt et al, 2007). The indications are the same as those for the open counterpart and reserved for low-risk distal tumors. The distal ureter is dissected down to the ureteral orifice and the proximal end anastomosed to the bladder using standard techniques. The early reports are encouraging, but strict adherence to oncologic principles must be followed.
Endoscopic Treatment
Basic Attributes
Hugh Hampton Young described the first endoscopic evaluation of the upper urinary tract in 1912. Subsequent advances in technology allow us to reach all parts of the urinary tract with minimal morbidity through antegrade and retrograde approaches. Diagnosis and treatment of upper tract TCC have become possible with these improvements because tumor biopsy and ablation by various energy sources are possible even through the smallest instruments. In addition, miniaturization has made follow-up surveillance of the upper tract more practical with the use of smaller ureteroscopes, which usually do not require previous stenting, or with active dilation of the distal ureter.
Tumors of the upper urinary tract can be approached in a retrograde or antegrade fashion. The approach chosen depends largely on the tumor location and size. In general, a retrograde ureteroscopic approach is used for low-volume ureteral and renal tumors. An antegrade percutaneous approach is preferred for larger tumors of the upper ureter or kidney and for those that cannot be adequately manipulated in a retrograde approach because of location (e.g., lower pole calyx) or previous urinary diversion. In cases with multifocal involvement, combined antegrade and retrograde approaches can be considered (Figs. 53-26 and 53-27).
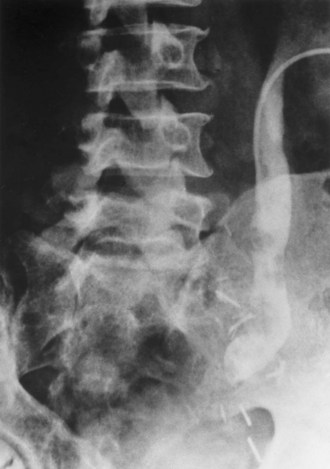
Figure 53–26 Retrograde pyelogram of a patient with a solitary kidney and multifocal low-grade tumors, which required both antegrade and retrograde approaches.
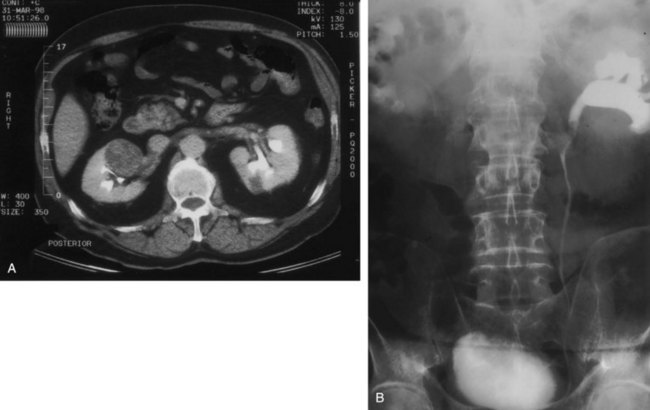
Figure 53–27 Patient with synchronous bilateral tumors. A, Right renal cell carcinoma that required radical nephrectomy. B, Left proximal ureteral tumor that required combined ureteroscopic and antegrade percutaneous ablation.
The basic principles for treatment of TCC of the upper urinary tract are similar to those for the bladder counterpart (Fig. 53–28). The tumor is sampled and ablated by electrocautery or laser energy sources. A staged procedure should be considered for high-volume disease or disease that is thought to represent high pathologic grade or stage. In such cases, when subsequent nephroureterectomy most likely will be necessary for cure, only biopsy and partial ablation are performed to minimize the risks of perforation or major complications. Endoscopic management is completed only after the pathologic examination shows that the patient is an acceptable candidate for continued minimally invasive endoscopic management. If the pathologic process is unresectable, of high grade, or invasive, the patient should proceed immediately to nephroureterectomy, provided he or she is medically fit. Patients who undergo renal-sparing therapy must be committed to a lifetime of follow-up with radiographs and endoscopy.
Ureteroscopy and Ureteropyeloscopy
The ureteroscopic approach to tumors was first described by Goodman in 1984 and is generally favored for ureteral and smaller renal tumors. With the advent of small-diameter rigid and flexible ureteroscopes, tumor location is less of a limiting factor than it used to be. The advantage of a ureteroscopic approach is lower morbidity than that of the percutaneous and open surgical counterparts, with the maintenance of a closed system. With a closed system, nonurothelial surfaces are not exposed to the possibility of tumor seeding.
The major disadvantages of a retrograde approach are related to the smaller instruments required. Smaller endoscopes have a smaller field of view and working channel. This limits the size of tumor that can be approached in a retrograde fashion. In addition, some portions of the upper urinary tract, such as the lower pole calyces, cannot be reliably reached with working instruments. Smaller instruments limit the ability to remove large tumors and to obtain deep specimens for reliable staging. Retrograde ureteroscopy is difficult in patients with prior urinary diversion.
Key Point: Ureteroscopy
Technique and Instrumentation
A wide variety of ureteroscopic instruments are available, each with its own distinct advantages and disadvantages. In general, rigid ureteroscopes are used primarily for the distal ureter and midureter. Access to the upper ureter and kidney with rigid endoscopy is unreliable, especially in the male patient. Larger, rigid ureteroscopes provide better visualization because of their larger field of view and better irrigation. Smaller rigid ureteroscopes (8 Fr) generally do not require active dilation of the ureteral orifice (Fig. 53–29A).
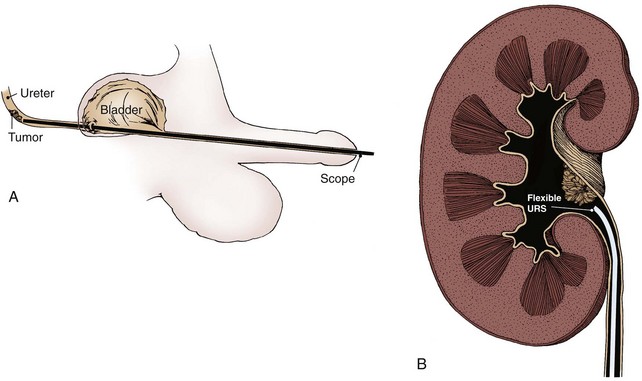
Figure 53–29 A, Rigid ureteroscopic approach. B, Flexible ureteroscopic approach. URS, ureteroscope.
Newer-generation, flexible ureteropyeloscopes are available in sizes smaller than 8 Fr to allow simple and reliable passage to most portions of the urinary tract (Abdel-Razzak and Bagley, 1993; Grasso and Bagley, 1994; Chen and Bagley, 2000; Chen et al, 2000). These are generally preferred in the upper ureter and kidney, where the rigid ureteroscope cannot be reliably passed. Flexible ureteroscopes, however, have technical limitations, such as a small working channel, that limit irrigant flow and the diameter of working instruments. Further limitations of flexible ureteroscopy include reduced access to certain areas of the kidney, such as the lower pole, where the infundibulopelvic angle may limit passage of the scope, and prior urinary diversion (Fig. 53–29B).
Endoscopic Evaluation and Collection of Urine Cytology Specimen
Cystoscopy is performed and the bladder inspected for concomitant bladder disease. The ureteral orifice is identified and inspected for lateralizing hematuria. A small-diameter (6.9 or 7.5 Fr) ureteroscope is passed directly into the ureteral orifice, and the distal ureter is inspected for any trauma from a previously placed guide wire or dilation. A guide wire is then placed through the ureteroscope and up the ureter to the level of the renal pelvis under fluoroscopic guidance. The flexible ureteroscope is used to visualize the remaining urothelium. When a lesion or suspicious area is seen, a normal saline washing of the area is performed before biopsy or intervention (Bian et al, 1995). If the ureter does not accept the smaller ureteroscope, active dilation of the ureter is necessary.
Special circumstances include prior urinary diversion and tumor confined to the intramural ureter. With cases of prior urinary diversion, identification of the ureteroenteric anastomosis is difficult and may require antegrade percutaneous passage of a guide wire down the ureter before endoscopy. The wire can be retrieved from the diversion, and the ureteroscope can be passed in a retrograde fashion. The nephrostomy tract does not need to be fully dilated in this setting. Wagner and associates (2008) described their experience with endoscopic monitoring of patients with ureteral carcinoma in situ after radical cystectomy. A second type of case is tumor in the intramural ureter. When a tumor protrudes from the ureteral orifice, complete ureteroscopic ablation of the tumor or aggressive transurethral resection of the entire most distal ureter can be done with acceptable results (Palou et al, 2000).
Biopsy and Definitive Treatment
Three general approaches can be used for tumor ablation: bulk excision with ablation of the base, resection of the tumor to its base, and diagnostic biopsy followed by ablation with electrocautery or laser energy sources. Regardless of technique used, special attention to biopsy specimens is necessary. Specimens are frequently minute and should be placed in fixative at once and specially labeled for either histologic or cytologic evaluation (Tawfiek et al, 1997).
Ureteroscopic Techniques
The tumor is debulked by use of either grasping forceps or a flat wire basket engaged adjacent to the tumor (Fig. 53–30A). Next, the tumor base is treated with either electrocautery or laser energy sources. This technique is especially useful for low-grade papillary tumor on a narrow stalk. The specimen is sent for pathologic evaluation.
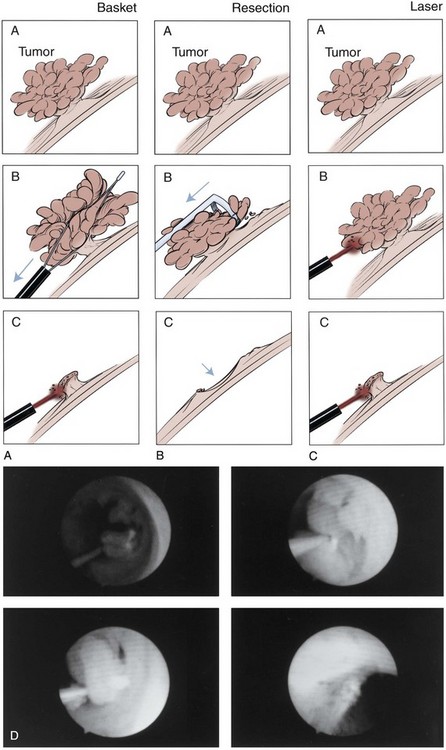
Figure 53–30 Techniques for ureteroscopic treatment of ureteral and renal tumors. A, The tumor is identified and removed piecemeal by grasping forceps to its base. B, Alternatively, a flat wire basket can be deployed alongside the tumor. The tumor is engaged and removed, with care taken not to avulse the adjacent ureter. With either of these technique, the base is treated with electrocautery or a laser energy source. C, The tumor is identified and removed by a ureteroscopic resectoscope. The technique differs from the technique for bladder tumors in that only intraluminal tumor is resected. No attempt is made to resect deep, as with a bladder tumor. The scope is not arching deep into the tissue. D, The tumor is sampled for diagnostic purposes. The bulk of the tumor is then ablated with electrosurgical or laser energy. Laser energy is generally preferred because it has more reliable delivery of energy and depth of penetration. The two most commonly used energy sources are holmium : yttrium-aluminum-garnet and neodymium : yttrium-aluminum-garnet.
A ureteroscopic resectoscope is used to remove the tumor (Fig. 53–30B). Only the intraluminal tumor is resected, and no attempt is made to resect deep (beyond the lamina propria). Extra care is necessary in the midureter and upper ureter, where the wall is thin and prone to perforation. Ureteral resectoscopes are approximately 12 Fr and require more extensive dilation of the ureteral orifice. With larger volume disease of the distal ureter, Jarrett and associates (1995a) described extensive dilation of the ureter followed by resection with a long standard resectoscope. The tumor is adequately sampled with forceps and sent to the pathology laboratory for diagnostic evaluation. The tumor bulk is then ablated to its base with laser or electrosurgical energy (Fig. 53–30C and D). Multiple biopsy specimens are often required when small, flexible 3-Fr biopsy forceps are used. Electrocautery delivered through a small Bugbee electrode (2 or 3 Fr) can be used to fulgurate tumors. However, the variable depth of penetration can make its use in the ureter dangerous, and circumferential fulguration should be avoided because of the high risk of stricture formation. More recently, laser energy with either neodymium : yttrium-aluminum-garnet (Nd : YAG) (Smith et al, 1984; Schilling et al, 1986; Schmeller and Hofstetter, 1989; Carson, 1991) or holmium : YAG (Bagley and Erhard, 1995; Razvi et al, 1995; Matsuoka et al, 2003; Suoka et al, 2003) sources has been popular. Each has characteristic advantages (Fig. 53–31) and can be delivered through small, flexible fibers (200 or 365 µm) that fit through small, flexible ureteroscopes without significant alteration of irrigant flow or scope deflection.
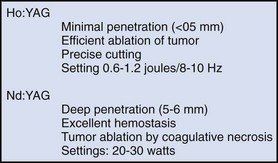
Figure 53–31 Characteristics of holmium (Ho):YAG and neodymium (Nd):YAG laser energy sources. YAG, yttrium-aluminum-garnet.
Key Point: Biopsy and Ablation
The holmium : YAG laser is well suited for use in the ureter. The tissue penetration is less than 0.5 mm, which allows tumor ablation with excellent hemostasis and minimal risk of full-thickness injury to the ureter. Its shallow penetration may, however, make its use cumbersome with larger tumors, especially in the renal pelvis. Settings most commonly used for the holmium : YAG laser are energy of 0.6 to 1 J with frequency of 10 Hz. The Nd : YAG laser has a tissue penetration of up to 5 to 6 mm, depending on laser settings and duration of treatment. In contrast to the holmium : YAG laser, which ablates tumor, the Nd : YAG laser works by coagulative necrosis with subsequent sloughing of the necrotic tumor. The safety margin is significantly lower and can limit its use in the ureter, where the ureteral wall is thin. Settings most commonly used for the Nd : YAG laser are 15 W for 2 seconds for ablation of tumor and 5 to 10 W for 2 seconds for coagulation.
A ureteral stent is placed for a variable duration to aid with the healing process. Large tumors usually require multiple treatment sessions during several months.
Results
Multiple series have shown the safety and efficacy of ureteroscopic treatment of upper tract TCC (Elliott et al, 1996; Englemeyer and Belis, 1996; Martinez-Pineiro et al, 1996; Keeley et al, 1997a, 1997b; Iborra et al, 2003). In a literature review of 205 patients (Tawfiek and Bagley, 1997), the overall recurrence rates for ureteral and renal pelvic lesions were 33% and 31.2%, respectively, and the risk of bladder recurrence was 43%. In the largest series (Keeley et al, 1997a) there was a single cancer death directly attributed to recurrent upper tract disease. As with any TCC, the most important prognostic indicator for tumor recurrence was grade. Keeley and associates (1997c) showed a recurrence rate of 26% for grade 1 tumors and 44% for grade 2 tumors, which roughly correlated with previously established recurrence rates for open conservative surgery.
More recent literature shows the long-term feasibility of the ureteroscopic approach, but concerns over the high rate of ipsilateral recurrences remain (Table 53–6). Daneshmand and colleagues (2003) reported a large number of recurrences with an overall ipsilateral recurrence rate of 90% with three to four recurrences per patient. Sowter and associates (2007) showed an ipsilateral recurrence rate of 74%. Thompson and colleagues (2008) studied 83 patients who underwent endoscopic management of tumors, 76 of which were ureteroscopic. The overall recurrence rate was 55%. Nephroureterectomy eventually was performed in 33%. This is important in considering patients with a normal contralateral kidney. Patients must be counseled in the need for lifetime follow-up and possible treatment of ipsilateral recurrence.
Complications were uncommon and usually related to the patient’s comorbidities. Complications specific to ureteroscopic therapy included ureteral perforation, which can be managed with indwelling ureteral stent, and ureteral stricture. Stricture formation rate ranged from 5% to 13% (Elliott et al, 1996; Martinez-Pineiro et al, 1996; Keeley et al, 1997a). The complication rates have dropped in more contemporary series, most likely related to smaller endoscopes, improved laser energy sources, and refinements in endoscopic techniques.
Two major concerns of the ureteroscopic approach are the accuracy of ureteroscopic biopsies and the limitations of biopsies, especially with regard to staging (Huffman, 1988). Retrospective reviews of patients who underwent ureteroscopic biopsy followed by nephroureterectomy found the accuracy of ureteroscopic diagnosis to be 89% to 94% and the pathologic grading to match the open surgical technique in 78% to 92% (Keeley et al, 1997a; Guarnizo et al, 2000). From prior studies, we know that there is an excellent correlation between grade of lesion and stage (Chasko et al, 1981; Heney et al, 1981). This holds true for the ureteroscopic approach (Keeley et al, 1997c) because 87% of patients with grade 1 or grade 2 tumors had noninvasive disease (stage Ta or T1) whereas 67% of patients with grade 3 tumors had invasive disease (stage T2 or T3). This information supports the notion that tumor grade is the most important prognostic factor; and although stage cannot be directly assessed, noninvasive disease can be expected in most cases of low-grade tumor.
A final concern is whether ureteroscopy promotes progression or spread of disease to other urothelial surfaces or metastatic sites. There have been reports of increased tumor appearance in refluxing ureters of patients with bladder tumors (de Torres Mateos et al, 1987) and in the ipsilateral urinary tract and bladder of patients after ureteroscopic treatment. However, Kulp and Bagley (1994) reported on 13 patients who underwent multiple ureteroscopic treatments followed by nephroureterectomy; they found no unusual propagation of TCC in the specimens. Concerns that ureteroscopy may promote metastatic spread were raised by Lim and associates (1993), who found tumor cells in renal lymphatics after ureteroscopy. However, Hendin and colleagues (1999) reported no increased risk of metastatic disease in a group of patients who underwent ureteroscopy before nephroureterectomy compared with a group undergoing nephroureterectomy alone.
Percutaneous Approach
The percutaneous approach was first described by Tomera and coworkers in 1982 and is generally favored for larger tumors located proximally in the renal pelvis or proximal ureter. The main advantage of the percutaneous approach is the ability to use larger instruments that can remove a large volume of tumor in any portion of the renal collecting system. Because deeper biopsy specimens are obtained, tumor staging as well as grading is usually possible. In addition, a percutaneous approach may avoid the limitations of flexible ureteroscopy, especially in complicated calyceal systems or areas difficult to access, such as the lower pole calyx or the upper urinary tract of patients with urinary diversion. With a percutaneous approach, the established nephrostomy tract can be maintained for immediate postoperative nephroscopy and administration of topical adjuvant therapy.
The main disadvantages are the increased morbidity compared with ureteroscopy and the potential for tumor seeding outside the urinary tract. Establishment of the nephrostomy tract has inherent risks, and the procedure usually requires inpatient admission. Distinct risks related to a percutaneous approach are loss of urothelial integrity and exposure of nonurothelial surfaces to tumor cells. This open system provides the possibility of tumor implantation in the nephrostomy tract.
Technique and Instrumentation
Establishment of the Nephrostomy Tract
Cystoscopy is performed, and an open-ended ureteral catheter is positioned in the pelvis. Contrast material is injected to define the calyceal anatomy, and a percutaneous nephrostomy tract is established through the desired calyx (Fig. 53–32). Tumors in peripheral calyces are best approached with direct puncture distal to the tumor (Fig. 53–33). Disease in the renal pelvis and upper ureter is best approached through an upper or middle pole access to allow scope maneuvering through the collecting system and down the ureteropelvic junction. The tract is dilated by either sequential (Amplatz) or balloon dilation to accommodate a 30-Fr sheath. Correct positioning of the nephrostomy tract is crucial to the success of the procedure and should be done by the urologist or by the radiologist after direct consultation with the operating surgeon.
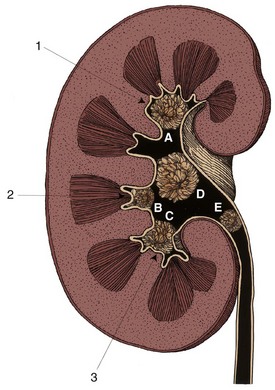
Figure 53–32 Nephrostomy tract puncture site. Position of the nephrostomy is imperative for successful percutaneous resection of transitional cell carcinoma of the renal collecting system and upper ureter and requires careful preoperative evaluation of radiographs for tumor location. Tumors in peripheral calyces (A to C) are best approached by direct puncture as far distally in the calyx as possible. Tumors in the renal pelvis (D) and upper ureter (E) are best approached by puncture to an upper (1) or middle (2) calyx, which allows the scope to be maneuvered in the renal pelvis and down the ureter. Tumors in the lower calyx are approached by lower calyx puncture (3).
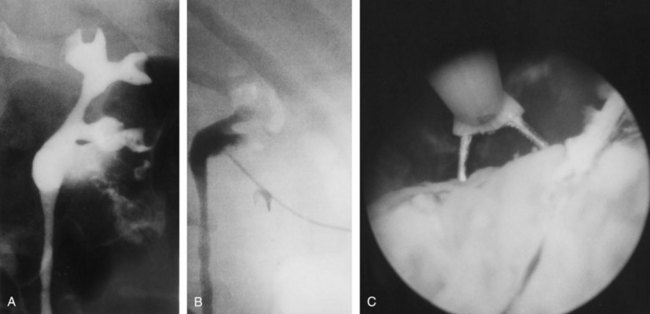
Figure 53–33 A, Retrograde pyelogram of a man with transitional cell carcinoma of the lower calyx in a solitary kidney. B, Access distal in the calyx allows a clear view of the tumor. C, Subsequent resection.
A nephroscope is inserted, and the ureteral catheter is grasped, brought out the tract, and exchanged for a stiff guide wire, thus providing both antegrade and retrograde control. Complete nephroscopy is performed with rigid and flexible endoscopes when necessary. Any suspicion of upper ureteral involvement warrants antegrade ureteroscopy.
Biopsy and Definitive Therapy
After identification, the tumors are removed by one of the following four techniques (Fig. 53–34). In the first technique, which uses a cold-cup biopsy forceps through a standard nephroscope, the bulk of the tumor is grasped by forceps and removed in piecemeal fashion until the base is reached (see Fig. 53–34A). A separate biopsy of the base is performed for staging purposes, and the base is cauterized with a Bugbee electrode and cautery. Low-grade papillary lesions on a thin stalk are easily treated in this manner with minimal bleeding.
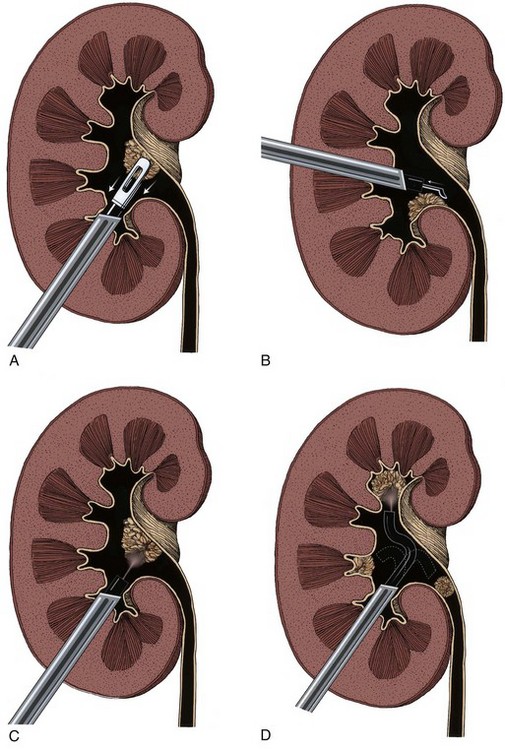
Figure 53–34 Techniques for percutaneous removal of transitional cell carcinoma of the renal collecting system. A, The tumor is identified and debulked by forceps to its base. The base is sampled and sent separately for evaluation. This technique works well for papillary tumors on a narrow stalk. Broad-based tumors may cause excessive bleeding and are best approached with resection or laser therapy. B, With use of a standard resectoscope the tumor is identified and resected to its base. Special care should be taken to avoid resection into major renal vasculature. The tumor is identified, sampled for diagnostic purposes, and treated by holmium or neodymium laser sources. This can be done through a standard nephroscope (C) or with a flexible cystoscope (D).
Alternatively, a cutting loop from a standard resectoscope is used to remove the tumor to its base (see Fig. 53–34B). Once again, the base should be resected and sent separately for staging purposes. This approach is more effective for larger, broad-based tumors for which simple debulking to a stalk is not possible.
For the third technique, which uses flexible or rigid endoscopes, the tumor is sampled and treated with holmium : YAG or Nd : YAG laser at 25 to 30 W (see Fig. 53–34C and D). Tissue may also be obtained with a small snare used for gastrointestinal polyps.
Regardless of approach, a nephrostomy tube is left in place. This access can be used for second-look follow-up nephroscopy to ensure complete tumor removal (Fig. 53–35). Nephroureterectomy is indicated if the pathologic examination shows high-grade or invasive disease.
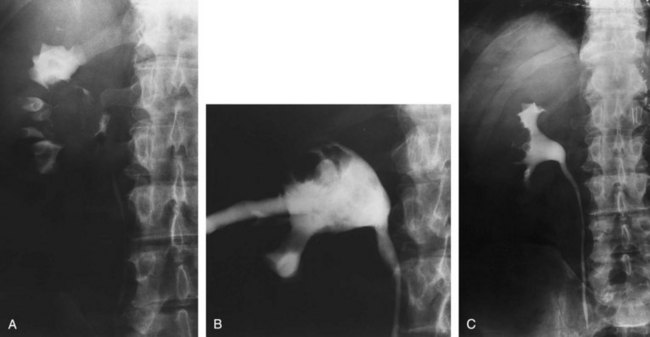
Figure 53–35 A, A 65-year-old man presented with a solitary kidney and a 5-cm renal pelvis tumor. B, Nephrostogram after patient underwent staged resection. C, Three-month follow-up retrograde pyelogram after completed resection. The patient showed grade 1 transitional cell carcinoma without invasion to submucosa.
Second-Look Nephroscopy
Follow-up nephroscopy is performed 4 to 14 days later to allow adequate healing. The tumor resection site is identified, and any residual tumor is removed. If no tumor is identified, the base should be sampled and treated by cautery or the Nd : YAG laser (15 to 20 W and 3-second exposures). The nephrostomy tube can be removed several days later if all tumors have been removed. If the patient is being considered for adjuvant topical therapy, a small, 8-Fr nephrostomy tube is left to provide access for instillations. Some authors advocate third-look nephroscopy before removal of the nephrostomy tube (Jarrett et al, 1995b).
Results
Because of the rarity of the disease there are limited retrospective series (Table 53–7) with adequate numbers and follow-up from which to draw reasonable conclusions (Jarrett et al, 1995b; Patel et al, 1996; Clark et al, 1999; Goel et al, 2003; Palou et al, 2004; Rouprêt et al, 2006). In a literature review of 84 patients, Okada and colleagues (1997) found an overall recurrence rate of 27%. Tumor grade strongly predicted outcomes; Jarrett and associates (1995b) showed the recurrence rate for grades 1, 2, and 3 lesions to be 18%, 33%, and 50%, respectively. The only cancer-related mortalities in this series were in patients with high-grade disease. Lee and colleagues (1999) reviewed their 13-year experience with percutaneous management, comparing 50 patients who underwent percutaneous management with 60 patients who underwent nephroureterectomy, and found no significant difference in overall survival. As expected, patients with low-grade disease did well regardless of modality and patients with high-grade disease did poorly regardless of treatment option.
Most would agree from the literature that percutaneous management is acceptable in patients with low-grade (grade 1) disease regardless of the status of the contralateral kidney, provided the patient is committed to lifelong endoscopic follow-up. Patients with grade 3 disease do poorly regardless of modality chosen but should probably undergo nephroureterectomy to maximize cancer therapy (provided they are medically fit). The largest area of controversy surrounds the use of percutaneous management for patients with grade 2 disease and a normal contralateral kidney. Jabbour and associates (2000) retrospectively evaluated 24 patients and found a disease-specific survival of 95% overall and 100% and 80% for stage Ta and stage T1 lesions, respectively. This study shows an acceptable result with conservative treatment of noninvasive grade 2 disease. With more invasive lesions, the potential for disease progression and metastatic disease is significant and nephroureterectomy should be considered.
Complications from percutaneous management of tumors are similar to those for benign renal processes and include bleeding, perforation of the collecting system, and secondary ureteropelvic junction obstruction. Complications increase in number and severity with higher tumor grade (Jarrett et al, 1995b). This finding is probably due to the more extensive pathologic process and treatments necessary to eradicate the tumor. Unlike ureteroscopic resection, the percutaneous method can stage tumors and, as expected, stage increases with tumor grade.
A major concern of the percutaneous approach is the potential seeding of nonurothelial surfaces with tumor cells. Although there have been multiple reported cases of nephrostomy tract infiltration with high-grade tumors (Tomera et al, 1982; Slywotzky and Maya, 1994; Huang et al, 1995; Oefelein and MacLennan, 2003; Treuthardt et al, 2004), there were no reported occurrences in the three largest series (Jarrett et al, 1995b; Patel et al, 1996; Clark et al, 1999). Tract seeding is a possibility but appears to be an uncommon event.
Isolated Upper Tract Cytologic Abnormality or Urinary Marker
On occasion, one is faced with a patient who has an isolated abnormal cytologic finding or other urinary marker from the upper urinary tract. By definition, the patient should have normal intravenous urography and retrograde pyelography, normal cystoscopy, and normal biopsy findings from the bladder and urethra. Radical nephroureterectomy was performed in the past for a unilateral cytologic abnormality of the upper tract to eliminate presumed carcinoma in situ. This practice is not recommended (Gittes, 1980; McCarron et al, 1983; Williams, 1991; Messing and Catalona, 1998). Upper tract cytology suffers from the same limitations in specificity as does bladder cytology. Furthermore, properly collected upper tract samples are of limited volume and cell count compared with bladder washings. Any source of inflammation, such as urinary infection or calculus, may produce a false-positive result. A subsequent cytologic abnormality from the contralateral side during follow-up is not rare in cases of true-positive results from early carcinoma in situ (Murphy et al, 1974; Khan et al, 1979). A variety of urinary markers are being studied to aid in detection of urothelial tumor recurrence. At this time, both urine FISH and urine nuclear matrix protein show particular promise.
Ureteropyeloscopy is indicated in such cases because the yield for direct visualization of small lesions is superior to that of retrograde pyelography. An attempt to perform biopsy and confirm the diagnosis is indicated before treatment is considered. A consistently abnormal finding on upper tract cytology may signify carcinoma in situ. Current treatment options are limited. Instillation of topical immunotherapy or chemotherapy may be performed through ureteral catheters or a small percutaneous nephrostomy. However, the contact time of the agent with the urothelium is minimal. Reports of efficacy are uncontrolled and anecdotal. Frequent-interval reevaluation with urinalysis, bladder cytology, cystoscopy every 3 months and retrograde pyelography or ureteropyeloscopy every 6 months is indicated for 1 to 2 years. However, one must avoid overtreatment and excessive evaluation and instrumentation of a patient who may have had a single false-positive result.
Adjuvant Therapy
After Organ-Sparing Therapy
Any procedure short of nephroureterectomy has a higher local recurrence due to the established risk of ipsilateral recurrence. Several approaches are available to minimize these risks. They fall into two basic categories: instillation of immunotherapeutic or chemotherapeutic agents and brachytherapy of the nephrostomy tract.
Instillation Therapy
Instillation therapy can be accomplished in several ways. Accepted techniques include antegrade instillation through a nephrostomy tube (Fig. 53–36) and retrograde instillation directly into a ureteral catheter or by reflux in a patient with an indwelling ureteral stent or iatrogenically created vesicoureteral reflux. Patel and Fuchs (1998) described a convenient technique of outpatient instillation through a ureteral catheter placed suprapubically. Regardless of the technique chosen, administration to the upper urinary tract should be done under low pressure and in the absence of active infection to minimize the risk of bacterial sepsis or systemic absorption of the agent.
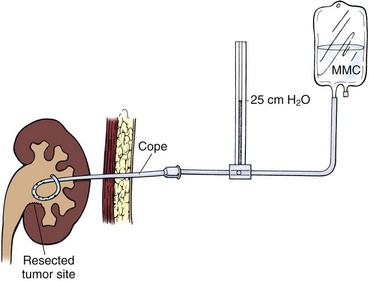
Figure 53–36 Setup for administration of topical immunotherapy or chemotherapy to the upper urinary tract through a previously placed nephrostomy tube. Therapy is instilled by gravity with a mechanism that prevents excessive intrarenal pressures. High pressures have been linked to complications of systemic absorption and bacterial sepsis. MMC, mitomycin C.
Results
The same agents used to treat urothelial carcinoma of the bladder are used to treat tumors of the upper urinary tract. Many studies have described small, retrospective, uncontrolled series of patients undergoing therapy with thiotepa (Elliott et al, 1996; Patel et al, 1996), mitomycin (Eastham and Huffman, 1993; Weston et al, 1993; Martinez-Pineiro et al, 1996; Keeley et al, 1997a; See, 2000), and BCG (Table 53–8) (Smith et al, 1987; Studer et al, 1989; Eastham and Huffman, 1993; Sharpe et al, 1993; Jarrett et al, 1995b; Hayashida, 2004; Rastinehad et al, 2009). Although the cumulative experience appears encouraging, no individual study has shown statistical improvement with relation to survival and recurrence rates. Possible reasons for this include (1) insufficient numbers to show clinical significance because of the relative rarity of the disease; (2) tumors of the upper urinary tract, which have a tumor biology different from that of their bladder counterparts; and (3) an inadequate delivery system that, unlike in the bladder, does not allow uniform delivery of the agent with adequate dwell time to enable a clinical response. Furthermore, in the report by Hayashida and colleagues (2004) with intrarenal perfusion of BCG, despite initial return of cytology results to normal, 50% of patients (5 of 10) developed disease recurrence after a mean follow-up of 50.9 months, and all of these suffered cancer-specific mortality. There is no doubt that more studies are required to settle this issue. The attempt to decrease multiple recurrences in the upper tract is reasonable in well-selected patients.
Table 53–8 Percutaneous BCG Instillation for Prevention of Recurrence of Upper Tract Urothelial Tumors

The most common complication of instillation therapy is bacterial sepsis. To minimize this problem, patients must be evaluated for active infection before each treatment and only a low-pressure delivery system should be used. Agent-specific complications of the various therapies include ramification of systemic absorption of the agent. Bellman and colleagues (1994) described upper urinary tract complications of percutaneous BCG instillation. Granulomatous involvement of the kidney in the absence of systemic signs of BCG infection was most commonly seen. Mukamel and associates (1991) saw an inordinate decrease in renal function for patients receiving BCG who had vesicoureteral reflux.
Brachytherapy
Brachytherapy to the nephrostomy tract through iridium wire or delivery system was described by Patel and coworkers (1996) and Nurse and colleagues (1989). There were no instances of tract recurrences in this series, although the authors acknowledged the rarity of the event. The only major complication attributed to the brachytherapy was cutaneous fistula formation requiring nephroureterectomy.
After Complete Excision
Radiation Therapy
The rationale for focal radiation therapy is to decrease the risk of local relapse after radical surgery for locally advanced non–organ-confined disease (stage T3 to T4, N+). Most series concluding that postoperative irradiation is beneficial are small or even anecdotal, uncontrolled, and retrospective (Holtz, 1962; Brady et al, 1968; Leiber and Lupu, 1978). In one series with 41 patients, postoperative radiation therapy decreased local recurrence but had no effect on distant relapse or survival (Brookland and Richter, 1985). Maulard-Durdux and associates (1996) retrospectively reviewed 26 patients who received 46 Gy to the wound bed after radical surgery for upper tract tumors. Tumors were grade 2 in 40% and grade 3 in 60% of cases. Tumor stage was T2, T3, and N+ in 42%, 58%, and 35% of cases, respectively. Five-year survival is shown in Table 53–9. Overall 5-year survival was 49%, with 30% remaining disease free. All patients with local relapse also had distant relapse, leading the authors to conclude that adjuvant radiation therapy is not beneficial.
Table 53–9 5-Year Survival after Postoperative Adjuvant Radiation Therapy for Upper Tract Urothelial Tumors
| 5-YEAR SURVIVAL (%) | |
|---|---|
| Tumor Grade | |
| 2 | 90 |
| 3 | 0 (P < .01) |
| TNM Stage | |
| 2 | 60 |
| 3 | 19 |
| N0 | 49 (P = .04) |
| N+ | 15 |
Data from Maulard-Durdux C, Dufour B, Hennequin C, et al. Postoperative radiation therapy in 29 patients with invasive transitional cell carcinoma of the upper urinary tract: no impact on survival? J Urol 1996;155:115–7.
The largest experience addressing this issue is that reported by Hall and associates (1998b). A retrospective review of 252 patients with upper tract tumors who were observed for a median of 64 months was performed. Radical nephroureterectomy was performed in 77% of cases. Initial tumor stage was T3 in 19% and T4 in 10% of cases; 50% and 52%, respectively, of patients with stage T3 and stage T4 tumors received 40 Gy to the wound bed postoperatively. Disease-specific and overall 5-year survival was 41% and 28%, respectively, for patients with stage T3 disease. Actuarial 5-year disease-specific survival for stage T3 with or without adjuvant radiation therapy was 45% and 40%, respectively. Median survival was 6 months for stage T4 disease. There were no long-term survivors in this group. Local relapse occurred in only 9% of the entire series and was seen only in patients with stage T3 and stage T4 disease. Among the patients who received adjuvant radiation, isolated local relapse without distant metastases occurred in only 10% and 4% of stage T3 and stage T4 cases, respectively. Thus radical nephroureterectomy alone provides a high rate of local control. Adjuvant radiation for high-stage disease does not decrease local relapse or protect against a high rate of distant failure.
Systemic Chemotherapy
The major limitation to long-term survival for patients with locally advanced upper tract tumors is distant relapse with metastatic disease. Both induction (neoadjuvant) and adjuvant systemic therapy with agents used for proven metastatic disease (see following discussion) are reasonable. However, there are no controlled trials that establish the efficacy of either induction or adjuvant chemotherapy in this setting. The small number of cases treated with adjuvant chemotherapy are anecdotal and preclude definitive conclusions of efficacy.
Follow-Up
Issues in Assessing for Recurrence
The propensity of upper tract tumors for multifocal recurrence and metastatic spread with more dysplastic lesions makes follow-up complicated. Postoperative evaluation must routinely include evaluation of the bladder, the ipsilateral (if organ-sparing therapy was chosen) and contralateral urinary tracts, and the extraurinary sites for local and metastatic spread. A follow-up regimen is thus dependent on the time from surgery, the approach chosen (organ sparing vs. radical), and the potential for metastatic spread. General recommendations for time intervals are listed in Figure 53–37.
General Procedures
All patients should be assessed at 3-month intervals the first year after they are rendered tumor free by endoscopic or open surgical approaches (Keeley et al, 1997a). This schedule is largely based on work with bladder TCC that shows that most tumor recurrences after bladder resection develop in the first year (Varkarakis et al, 1974; Loening et al, 1980). The upper urinary tract is more difficult to monitor, and delayed recognition of upper tract tumor recurrence may lead to disease progression and poor results (Mazeman, 1976). Evaluation should include history, physical examination, urinalysis, and office cystoscopy because of the high risk of bladder recurrences in patients treated both conservatively and with nephroureterectomy (Mazeman, 1976). If the patient requires endoscopic evaluation of the upper urinary tract, cystoscopy can be done in conjunction with that procedure.
Urine cytology may be helpful in assessing for upper tract recurrence, especially for high-grade tumors (Murphy et al, 1981). The utility, however, is decreased with less dysplastic tumors (Grace et al, 1967; Sarnacki et al, 1971; Zincke et al, 1976). The same tumor markers under study for bladder TCC are promising for upper urinary tract TCC (Brown, 2000). One marker that may be preferentially more involved in upper tract TCC than in bladder TCC is the DNA mismatch repair gene MSH2 (Leach et al, 2000).
Specific Procedures
Bilateral disease, either synchronous or metachronous, is seen in 1% to 4% of patients (Petkovic, 1975; Babaian and Johnson, 1980; Murphy et al, 1981), and thus imaging of the contralateral kidney is required on a regular basis. Yearly intravenous urography is usually sufficient. However, retrograde pyelography may be necessary if the patient is not a candidate for injection of contrast medium or if intravenous urography is not diagnostic. CT or ultrasonography is helpful in distinguishing stones from soft tissue densities. Further evaluation of filling defects on imaging studies usually requires ureteroscopic evaluation.
If an organ-sparing approach is chosen, the ipsilateral urinary tract must be assessed as well as the remainder of the urinary tract. The frequency and duration of the follow-up assessments depend largely on the grade and stage of the lesion, but they are usually every 6 months for several years and annually thereafter. Radiographic evaluation of the upper tracts alone is not adequate because Keeley and colleagues (1997a) showed that 75% of early tumor recurrences were visible endoscopically and not radiographically. With tumors approached in a percutaneous fashion, immediate follow-up nephroscopy can be performed through the established nephrostomy tract.
In the past, the burden of repeated endoscopic evaluation of the upper urinary tracts was a major deterrent to conservative therapy. The use of smaller, 7.5-Fr flexible ureteroscopes has greatly eased the burden of follow-up because ureteroscopes can be reliably passed up the ureter without the need for dilation of the ureteral orifice or prior stenting. Others have advocated resection of the ureteral orifice to facilitate subsequent surveillance ureteroscopy in the office setting (Kerbl and Clayman, 1993). Even though technology has somewhat facilitated follow-up, both physician and patient must be committed to nephron-sparing treatment.
Metastatic Restaging
Metastatic restaging is required in all patients at significant risk for disease progression to local or distant sites. This group includes those with high-grade or high-stage disease. Metastatic restaging is generally not necessary for low-grade disease when the risks of invasive and subsequent metastatic disease are negligible. Included in metastatic restaging is imaging of the ipsilateral renal bed for recurrence with cross-sectional imaging. Follow-up restaging includes chest radiograph, liver function tests, cross-sectional body imaging, and selective use of bone scintigraphy based on an understanding of natural disease history and metastatic pathways (Korman et al, 1996).
Treatment of Metastatic Disease
Outcomes are poor in patients with metastatic urothelial tumors of the upper urinary tract. The systemic chemotherapy regimens offered for treatment are the same as those used for TCC of the bladder. These regimens are discussed in Chapter 82. Only limited data on response rates of advanced upper tract tumors to chemotherapy are available because of the rarity of these lesions. Prospective randomized trials comparing chemotherapeutic regimens for upper tract tumors are not feasible for this reason. Patients with advanced upper tract tumors are excluded from most trials on treatment of advanced bladder cancer. Therefore, one must extrapolate likely chemotherapy response rates for upper tract disease from observations in bladder cancer trials. However, the need for effective use of perioperative chemotherapy for patients with locally advanced and metastatic upper tract tumors remains great. In a study of 184 patients accrued over three consecutive time intervals from 1986 to 2004 at M. D. Anderson Cancer Center, the median recurrence-free survival was 2.4 years and did not improve over time (Brown et al, 2006). The decline in renal function after nephroureterectomy in these mostly elderly patients may compromise the ability to administer effective postoperative chemotherapy and is yet another reason to consider neoadjuvant chemotherapy for patients with upper tract tumors and evidence of regional lymph node metastases.
The MVAC (methotrexate/vinblastine/doxorubicin [Adriamycin]/cisplatin) regimen continues to have the highest response rate (Sternberg et al, 1989). However, complete responses are rare, and the duration of response is limited, with overall survival of 12 to 24 months. Toxicity is high and dose limited in some patients. For all these reasons there is considerable ongoing investigation with newer agents, including paclitaxel, ifosfamide, carboplatin, and gemcitabine, used in various combinations and sequences (Roth et al, 1994; Bajorin et al, 1998; Redman et al, 1998; Vaughn et al, 1998; Kaufman et al, 2000; Lorusso et al, 2000). Many of these show initial overall response rates similar to the response rate to the MVAC regimen and lower toxicity. Occasional major responses are seen (Fig. 53–38). However, thus far, complete responses are rare, and neither durability of response nor survival is better than that seen with the MVAC regimen. In summary, upper tract urothelial cancer, like bladder cancer, is chemosensitive. It is to be hoped that additional improvements in treatment efficacy and safety are forthcoming, with further experience with new chemotherapeutic regimens. Alternative strategies, such as biologic response modifiers and gene therapy, also should be explored.
Figure 53–38 A patient presented with a large, biopsy-proven transitional cell carcinoma of the kidney as well as lung and axillary metastases. A, Initial appearance of the renal tumor. B, Marked shrinkage of the tumor after four courses of paclitaxel-gemcitabine chemotherapy. The lung and axillary metastases disappeared, and nephroureterectomy was performed. During the next year, the patient developed a brain metastasis.
(Courtesy of Dr. Randall Singleton.)
Abe T, Shinohara N, Harabayashi T, et al. The role of lymph-node dissection in the treatment of upper urinary tract cancer: a multi-instituional study. Br J Urol Int. 2008;102:576-579.
Batata MA, Grabstald H. Upper urinary tract urothelial tumors. Urol Clin North Am. 1976;3:79-86.
Berger MA, Haber GP, Kamoi K, et al. Laparoscopic radical nephroureterectomy for upper tract transitional cell carcinoma: oncological outcomes at 7 years. J Urol. 2008;180:849-854.
Bolenz C, Fernandez M, Trojan L, et al. Lymphovascular invasion and pathologic tumor stage are significant outcome predictors for patients with upper tract urothelial carcinoma. Urology. 2008;72:364-369.
Brausi M, Gavioli M, De Luca G, et al. Retroperitoneal lymph node dissection (RPLND) in conjunction with nephroureterectomy in the treatment of infiltrative transitional cell carcinoma (TCC) of the upper urinary tract: impact on survival. Eur Urol. 2007;52:1414-1420.
Daneshmand S, Quek ML, Huffman JL. Endoscopic management of upper urinary tract transitional cell carcinoma: long-term experience. Cancer. 2003;98:55-60.
El Fettouh HA, Rassweiler JJ, Schulze M, et al. Laparoscopic radical nephroureterectomy: results of an international multicenter study. Eur Urol. 2002;42:447-452.
Elliott DS, Blute ML, Patterson DE, et al. Long-term follow-up of endoscopically treated upper urinary tract transitional cell carcinoma. Urology. 1996;47:819-825.
Hall MC, Womack S, Sagalowsky AI, et al. Prognostic factors, recurrence, and survival in transitional cell carcinoma of the upper urinary tract: a 30-year experience in 252 patients. Urology. 1998;52:594-601.
Herr HW. Long-term results of BCG therapy: concern about upper tract tumors. Semin Urol Oncol. 1998;16:13-16.
Huben RP, Mounzer AM, Murphy GP. Tumor grade and stage as prognostic variables in upper tract urothelial tumors. Cancer. 1988;62:2016-2020.
Kang CH, Yu TJ, Hsieh HH, et al. The development of bladder tumors and contralateral upper urinary tract tumors after primary transitional cell carcinoma of the upper urinary tract. Cancer. 2003;98:1620-1626.
Kikuchi E, Margolis V, Karakiewicz P, et al. Lymphovascular invasion predicts clinical outcomes in patients with node-negative upper tract urothelial carcinoma. J Clin Oncol. 2009;27:612-618.
Konety BR, Getzenberg RH. Urine based markers of urologic malignancy. J Urol. 2001;165:600.
Matin SF, Gill IS. Recurrence and survival following laparoscopic radical nephroureterectomy with various forms of bladder cuff control. J Urol. 2005;173:395-400.
Matsui Y, Utsunomiya N, Ichioka K, et al. Risk factors for subsequent development of bladder cancer after primary transitional cell carcinoma of the upper urinary tract. Urology. 2005;54:279-283.
McTavish JD, Jinzaki M, Zou KH, et al. Multi-detector row CT urography: comparison of strategies for depicting the normal urinary collecting system. Radiology. 2002;225:783.
Muntener M, Nielson ME, Romero FR, et al. Long-term oncologic outcome after laparoscopic radical nephroureteretomy for upper tract transitional cell carcinoma. Eur Urol. 2007;51(6):1639-1644.
Novara G, De Marco V, Dalpiaz O, et al. Independent predictors of metachronous bladder transitional cell carcinoma (TCC) after nephroureterectomy for TCC of the upper urinary tract. BJU Int. 2008;101:1368-1374.
Rastinehad AR, Vanderbrink BA, Greenberg KL, et al. A 20 year experience with percutaneous resection of upper tract tract transitional cell carcinoma: is there an oncologic benefit with adjuvant bacillus Calmette Guérin. Urology. 2009;73(1):27-31.
Solsona E, Iborra I, Ricos JV, et al. Upper urinary tract involvement in patients with bladder carcinoma in situ (CIS): its impact on management. Urology. 1997;49:347-352.
Tran W, Serio AM, Raj GV, et al. Longitudinal risk of upper tract recurrence following radical cystectomy for urothelial cancer and the potential implications for long-term surveillance. J Urol. 2008;179:96-100.
Abdel-Razzak O, Bagley DH. The 6.9 F semirigid ureteroscope in clinical use. Urology. 1993;41:45-48.
Abe T, Shinohara N, Harabayashi T, et al. The role of lymph-node dissection in the treatment of upper urinary tract cancer: a multi-instituional study. Br J Urol Int. 2008;102:576-579.
Abercrombie GF, Eardley I, Payne SR, et al. Modified nephro-ureterectomy: long-term follow-up with particular reference to subsequent bladder tumours. Br J Urol. 1988;61:198-200.
Agarwal DK, Khaira MS, Clarke D, et al. Modified transurethral technique for the management of distal ureter during laparoscopic assisted nephroureterectomy. Urology. 2008;71:740-743.
Ahmed I, Shaikh NA, Kapadia CR. Track recurrence of renal pelvic transitional cell carcinoma after laparoscopic nephrectomy. Br J Urol. 1998;81:319.
Akao J, Matsuyama H, Yamamoto Y, et al. Clinical significance of lymphovascular invasion in upper urinary tract urothelial cancer. Br J Urol Int. 2008;102:572-575.
Akaza H, Koiso K, Niijima T. Clinical evaluation of urothelial tumors of the renal pelvis and ureter based on a new classification system. Cancer. 1970;26:583.
Akkad T, Brunner A, Pallwein L, et al. Fluorescence in situ hybridization for detecting upper urinary tract tumors—a preliminary report. Urol. 2007;70:753-757.
Amira N, River J, Soliman H, et al. Microsatellite instability in urothelial carcinoma of the upper tract. J Urol. 2003;170:1151.
Anderstrom C, Johansson SL, Pettersson S, Wahlquist L. Carcinoma of the ureter: a clinicopathologic study of 49 cases. J Urol. 1989;142:280-283.
Angulo JC, Hontoria J, Sanchez-Chapado M. One-incision nephroureterectomy endoscopically assisted by transurethral ureteral stripping. Urology. 1998;52:203.
Arango O, Bielsa O, Carles J, et al. Massive tumor implantation in the endoscopic resected area in modified nephroureterectomy. J Urol. 1997;157:1839.
Asano K, Miki J, Maeda S, et al. Clinical studies on inverted papilloma of the urinary tract: report of 48 cases and review of the literature. J Urol. 2003;170:1209.
Babaian RJ, Johnson DE. Primary carcinoma of the ureter. J Urol. 1980;123:357-359.
Babaian RJ, Johnson DE, Llamas L, et al. Metastases from transitional cell carcinoma of the urinary bladder. Urology. 1980;16:142.
Bagley D, Erhard M. Use of the holmium laser in the upper urinary tract. Tech Urol. 1995;1:25-30.
Bajorin D, McCaffrey J, Hilton S, et al. Treatment of patients with transitional cell carcinoma of the urothelial tract with ifosfamide, paclitaxel, and cisplatin: a phase II trial. J Clin Oncol. 1998;16:2722-2727.
Bariol SV, Stewart GD, McNeill SA, Tolley DA. Oncological control following laparoscopic nephroureterectomy: 7-year outcome. J Urol. 2004;172(Pt. 1):1805-1808.
Batata MA, Grabstald H. Upper urinary tract urothelial tumors. Urol Clin North Am. 1976;3:79-86.
Batata MA, Whitmore WFJr, Hilaris BS, Grabstald H. Primary carcinoma of the ureter: a prognostic study. Cancer. 1975;35:1626-1632.
Bazeed MA, Scharfe T, Becht E, et al. Local excision of urothelial cancer of the upper urinary tract. Eur Urol. 1986;12:89-95.
Bellman GC, Sweetser P, Smith AD. Complications of intra-cavitary bacillus Calmette-Guérin after percutaneous resection of upper tract transitional cell carcinoma. J Urol. 1994;51:13-15.
Bennington JL, Beckwith JB. Tumors of the kidney, renal pelvis, and ureter. In: Atlas of tumor pathology, second series, fascicle 12. Washington (DC): Armed Forces Institute of Pathology; 1975.
Berger MA, Haber GP, Kamoi K, et al. Laparoscopic radical nephroureterectomy for upper tract transitional cell carcinoma: oncological outcomes at 7 years. J Urol. 2008;180:849-854.
Bian Y, Ehya H, Bagley DH. Cytologic diagnosis of upper urinary tract neoplasms by ureteroscopic sampling. Acta Cytol. 1995;39:733-740.
Blacker EJ, Johnson DE, Abdul-Karim FW, et al. Squamous cell carcinoma of renal pelvis. Urology. 1985;25:124.
Blank C, Lissmer L, Kaneti J, et al. Fibroepithelial polyp of the renal pelvis. J Urol. 1987;137:962.
Bloom NA, Vidone RA, Lytton B. Primary carcinoma of the ureter: a report of 102 new cases. J Urol. 1970;103:590-598.
Blute ML, Segura JW, Patterson DE, et al. Impact of endourology on diagnosis and management of upper urinary tract urothelial cancer. J Urol. 1989;141:1298.
Blute ML, Tsushima K, Lieber MM, et al. Transitional cell carcinoma of the renal pelvis: nuclear deoxyribonucleic acid ploidy studied by flow cytometry. J Urol. 1988;140:844.
Blute RDJr, Gittes RR, Gittes RF. Renal brush biopsy: survey of indications, techniques and results. J Urol. 1981;126:146.
Bolenz C, Fernandez M, Trojan L, et al. Lymphovascular invasion and pathologic tumor stage are significant outcome predictors for patients with upper tract urothelial carcinoma. Urology. 2008;72:364-369.
Booth CM, Cameron KM, Pugh RCB. Urothelial carcinoma of the kidney and ureter. Br J Urol. 1980;52:430-435.
Brady LW, Gislason GJ, Faust DS, et al. Radiation therapy: a valuable adjunct in the management of carcinoma of the ureter. JAMA. 1968;206:2871-2874.
Brausi M, Gavioli M, De Luca G, et al. Retroperitoneal lymph node dissection (RPLND) in conjunction with nephroureterectomy in the treatment of infiltrative transitional cell carcinoma (TCC) of the upper urinary tract: impact on survival. Eur Urol. 2007;52:1414-1420.
Brenner DW, Schellhammer PF. Upper tract urothelial malignancy after cyclophosphamide therapy: a case report and literature review. J Urol. 1987;137:1226.
Brookland RK, Richter MP. The postoperative irradiation of transitional cell carcinoma of the renal pelvis and ureter. J Urol. 1985;133:952-955.
Brown FM. Urine cytology: is it still the gold standard for screening? Urol Clin North Am. 2000;27:25-37.
Brown GA, Busby JE, Wood CG, et al. Nephroureterectomy for treating upper urinary tract transitional cell carcinoma: time to change the treatment paradigm? BJU Int. 2006;98:1176-1180.
Canales BK, Anderson JK, Premoli J, et al. Risk factors for upper tract recurrence in patients undergoing long-term surveillance for stage Ta bladder cancer. J Urol. 2006;175(1):74-77.
Caoili EM, Cohan RH, Korobkin M, et al. Urinary tract abnormalities: initial experience with multi-detector row CT urography. Radiology. 2002;222:353.
Carpinito GA, Stadler WM, Briggman JV, et al. Urinary nuclear matrix protein as a marker for transitional cell carcinoma of the urinary tract. J Urol. 1996;156:1280.
Carson CC. Endoscopic treatment of upper and lower urinary tract lesions using lasers. Semin Urol. 1991;9:185-191.
Catto JW, Yates DR, Rehman AR, et al. Behavior of urothelial carcinoma with respect to anatomic location. J Urol. 2007;177:1715-1720.
Chan KW, Lam KY, Srivastava G. Accumulation of p53 protein in inverted transitional cell papillomas of the urinary bladder. J Clin Pathol. 1996;49:43.
Chasko SB, Gray GF, McCarron JPJr. Urothelial neoplasia of the upper urinary tract. Pathol Annu. 1981;16(pt 2):127-153.
Chen GL, Bagley DH. Ureteroscopic management of upper tract transitional cell carcinoma in patients with normal contralateral kidneys. J Urol. 2000;164:1173.
Chen GL, El-Gabry EA, Bagley DH, et al. Surveillance of upper urinary tract transitional cell carcinoma: the role of ureteroscopy, retrograde urography, cytology and urinalysis. J Urol. 2000;164:1901.
Cheng G, Bostwick DG. World Health Organization and International Society of Urologic Pathology classification and two-number grading system of bladder tumors. Cancer. 2000;88:1513-1516.
Cheng L, Neumann RM, Bostwick DG. Papillary urothelial neoplasms of low malignant potential: clinical and biological implications. Cancer. 1999;86:2102-2108.
Cheville JC, Wu K, Sebo TJ, et al. Inverted urothelial papilloma: is ploidy, MIB-1 proliferative activity, or p53 protein accumulation predictive of urothelial carcinoma? Cancer. 2000;88:632.
Cho KS, Hong SJ, Cho NH, Choi YD. Grade of hydronephrosis and tumor diameter as preoperative prognostic factors in ureteral transitional cell carcinoma. Urology. 2007;70:662-666.
Chung HJ, Chiu AW, Chen KK, et al. Retroperitoneoscopy assisted nephroureterectomy for the management of upper tract urothelial cancer. Minim Invasive Ther. 1996;5:266.
Chung S, Wang S, Lai M, et al. Lymphovascular invasion predicts poor outcome of urothelial carcinoma of renal pelvis after nephroureterectomy. BJU Int. 2009;103:1047-1051.
Clark PC, Streem SB, Geisinger MA. 13 year experience with percutaneous management of upper tract transitional cell carcinoma. J Urol. 1999;161:772-775.
Clayman RV, Garske GL, Lange PH. Total nephroureterectomy with ureteral intussusception and transurethral ureteral detachment and pull-through. Urology. 1983;21:482-486.
Clayman RV, Kavoussi LR, Figenshau RS, et al. Laparoscopic nephro-ureterectomy: initial clinical case report. J Laparoendosc Surg. 1991;1:343-349.
Compérat E, Rouprêt M, Chartier-Kastler E, et al. Prognostic value of MET, RON, and histoprognostic factors for urothelial carcinoma oin the upper urinary tract. J Urol. 2008;179:868-872.
Coup AJ. Angiosarcoma of the ureter. Br J Urol. 1988;62:275.
Cummings KB. Nephroureterectomy: rationale in the management of transitional cell carcinoma of the upper urinary tract. Urol Clin North Am. 1980;7:569-578.
Daneshmand S, Quek ML, Huffman JL. Endoscopic management of upper urinary tract transitional cell carcinoma: long-term experience. Cancer. 2003;98:55-60.
Data SN, Allen GM, Evans R, et al. Urinary tract ultrasonography in the evaluation of hematuria—a report of over 100 cases. Ann R Coll Surg Engl. 2002;84:203.
David KA, Mallin K, Milkowsky MI, et al. Surveillance of urothelial carcinoma: stage and grade migration, 1993-2005 and survival trends, 1993-2000. Cancer. 2009;115:1435-1447.
Davis BW, Hough AJ, Gardner WA. Renal pelvic carcinoma: morphological correlates of metastatic behavior. J Urol. 1987;137:857.
De Broe ME, Elseviers M. Analgesic nephropathy. N Engl J Med. 1998;338:446.
de Torres Mateos JA, Banus Gassol JM, Palou Redota J, Morote Robles J. Vesicorenal reflux and upper urinary tract transitional cell carcinoma after transurethral resection of recurrent superficial bladder carcinoma. J Urol. 1987;138:49-51.
Dixon JS, Gosling JA. The musculature of the human renal calyces, pelvis, and ureter. J Anat. 1982;135:129.
Dragicevic D, Djokic M, Pekmezovic T, et al. Survival of patients with transitional cell carcinoma of the ureter and renal pelvis in Balkan endemic nephropathy and non-endemic areas of Serbia. BJU Int. 2007;99:1357-1362.
Eastham JA, Huffman JL. Technique of mitomycin C instillation in the treatment of upper urinary tract urothelial tumors. J Urol. 1993;150(2 Pt. 1):324-325.
El Fettouh HA, Rassweiler JJ, Schulze M, et al. Laparoscopic radical nephroureterectomy: results of an international multicenter study. Eur Urol. 2002;42:447-452.
El-Hakim A, Weiss GH, Lee BR, Smith AD. Correlation of ureteroscopic appearance with histologic grade of upper tract transitional cell carcinoma. Urology. 2004;63:647.
Elliott DS, Blute ML, Patterson DE, et al. Long-term follow-up of endoscopically treated upper urinary tract transitional cell carcinoma. Urology. 1996;47:819-825.
Eltz S, Comperat E, Cussenot O, et al. Molecular and histological markers in urothelial carcinomas of the upper urinary tract. Br J Urol Int. 2008;102:532-535.
Englemeyer EL, Belis JA. Long-term ureteroscopic management of low grade transitional carcinoma of the upper urinary tract. Tech Urol. 1996;2:113.
Epstein JI, Amin MB, Reuter VR, et al. The World Health Organization/International Society of Urological Pathology consenus classification of urothelial (transitional cell) neoplasms of the urinary bladder. Bladder Consensus Conference Committee. Am J Surg Pathol. 1998;22(12):1435-1448.
Fein AB, McClennan BL. Solitary filling defects of the ureter. Semin Roentgenol. 1986;21:201.
Flanigan RC, Kim FI. Neoplastic disease of the pelvis, ureter, bladder, and urethra. In: Massry SG, Glassock RJ, editors. Massry and Glassock’s textbook of nephrology. Philadelphia: Lippincott Williams & Wilkins; 2004:1050-1058.
Fraley EE. Cancer of the renal pelvis. In: Skinner DG, deKernion JB, editors. Genitourinary cancer. Philadelphia: WB Saunders; 1978:134.
Frischer Z, Waltzer WC, Gonder MJ. Bilateral transitional cell carcinoma of the renal pelvis in the cancer family syndrome. J Urol. 1985;134:1197.
Geiger J, Fong Q, Fay R. Transitional cell carcinoma of the renal pelvis with invasion of renal vein and thrombosis of subhepatic inferior vena cava. Urology. 1986;28:52.
Gill IS, Soble JJ, Miller SD, Sung GT. A novel technique for management of the en bloc bladder cuff and distal ureter during laparoscopic nephroureterectomy. J Urol. 1999;161:430-434.
Gill WB, Lu CT, Thomsen S. Retrograde brushing: a new technique for obtaining histologic and cytologic material from ureteral, renal pelvic and renal caliceal lesions. J Urol. 1973;109:573-578.
Giovansili B, Peyromaure M, Saighi D, et al. Stripping technique for endoscopic management of distal ureter during nephroureterectomy: experience of 32 procedures. Urology. 2004;64:448-452.
Gittes RF. Management of transitional cell carcinoma of the upper tract: case for conservative local excision. Urol Clin North Am. 1980;7:559-568.
Gittes RF. Operative nephroscopy. J Urol. 1966;116:148-152.
Godec CJ, Murrah VA. Simultaneous occurrence of transitional cell carcinoma and urothelial adenocarcinoma associated with xanthogranulomatous pyelonephritis. Urology. 1985;26:412.
Goel MC, Mahendra V, Roberts JG. Percutaneous management of renal pelvic urothelial tumors: long-term followup. J Urol. 2003;169:925-929.
Goldwasser B, Leibovitch I, Avigad I. Ureteral substitution using the isolated interposed vermiform appendix in a patient with a single kidney and transitional cell carcinoma of the ureter. Urology. 1994;44:437-440.
Goodman TM. Ureteroscopy with rigid instruments in the management of distal ureteral disease. J Urol. 1984;132:250-253.
Grabstald H, Whitmore WFJr, Melamed MR. Renal pelvic tumors. JAMA. 1971;218:845-854.
Grace DA, Taylor WN, Taylor JN, Winter CC. Carcinoma of the renal pelvis: a 15-year review. J Urol. 1967;98:566-569.
Grainger R, Gikas PW, Grossman HB. Urothelial carcinoma occurring within an inverted papilloma of the ureter. J Urol. 1990;143:802.
Grasso M, Bagley A. 7.5/8.2 F actively deflectable, flexible ureteroscope: a new device for both diagnostic and therapeutic upper urinary tract endoscopy. Urology. 1994;43:435-441.
Greenlee RT, Murray T, Bolden S, Wings PA. Cancer statistics, 2000. CA Cancer J Clin. 2000;50:7.
Grollman AP, Shibutani S, Monya M, et al. Aristolochic acid and the etiology of endemic (Balkan) nephropathy. Proc Natl Acad Sci U S A. 2007;104:12129-12134.
Grossman HB. The late recurrence of grade I transitional cell carcinoma of the ureter after conservative therapy. J Urol. 1978;120:251-252.
Guarnizo E, Pavlovich CP, Seiba M, et al. Ureteroscopic biopsy of upper tract urothelial carcinoma: improved diagnostic accuracy and histopathological considerations using a multi-biopsy approach. J Urol. 2000;163:52-55.
Guinan P, Vogelzang NJ, Randazzo R, et al. Renal pelvic cancer: a review of 611 patients treated in Illinois 1975-1985. Urology. 1992;40:393-399.
Guinan P, Vogelzang NJ, Randazzo R, et al. Renal pelvic transitional cell carcinoma: the role of the kidney in tumor-node-metastasis staging. Cancer. 1992;69:1773-1775.
Habuchi T, Takahashi R, Yamada H, et al. Metachronous multifocal development of urothelial cancers by intraluminal seeding. Lancet. 1993;342:1087.
Hafner C, Knuechel R, Stoehr R, Hartmann A. Clonality of multifocal urothelial carcinomas: 10 years of molecular genetics studies. Int J Cancer. 2002;101:1.
Hall MC, Womack S, Roehrborn CG, et al. Advanced transitional cell carcinoma of the upper urinary tract: patterns of failure, survival and impact of postoperative adjuvant radiotherapy. J Urol. 1998;160:703-706.
Hall MC, Womack S, Sagalowsky AI, et al. Prognostic factors, recurrence, and survival in transitional cell carcinoma of the upper urinary tract: a 30-year experience in 252 patients. Urology. 1998;52:594-601.
Hanna MK, Jeffs RD, Sturgess JM, Barkin M. Ureteral structure and ultrastructure. J Urol. 1976;116:718.
Harris AL, Neal DE. Bladder cancer—field versus clonal origin. N Engl J Med. 1992;326:759-761.
Hartmann A, Dietmaier W, Hofstadter F, et al. Urothelial carcinoma of the upper urinary tract: inverted growth pattern is predictive of microsatellite instability. Pathology. 2003;34:222.
Hattori R, Yoshino Y, Gotoh M, et al. Long-term cancer control of retroperitoneoscopic nephroureterectomy for transitional cell carcinoma of the pelvis and ureter: Nagaya experience. J Urol. 2005;173:296.
Hayashida Y, Nomata K, Noguchi M, et al. Long-term effects of bacille Calmette-Guérin perfusion therapy for treatment of transitional cell carcinoma in situ of upper urinary tract. Urology. 2004;63:1084-1088.
Hendin BN, Streem SB, Levin HS, et al. Impact of diagnostic ureteroscopy on long-term survival in patients with upper tract transitional cell carcinoma. J Urol. 1999;161:783-785.
Heney N, Nocks B, Daly J, et al. Prognostic factors in carcinoma of the ureter. J Urol. 1981;125:632-636.
Herr HW. Long-term results of BCG therapy: concern about upper tract tumors. Semin Urol Oncol. 1998;16:13-16.
Herr HW, Cookson MS, Soloway SM. Upper tract tumors in patients with primary bladder cancer followed for 15 years. J Urol. 1996;156:1286-1287.
Herr HW, Wartinger DD, Oettgen HF. Bacillus Calmette-Guérin therapy for superficial bladder cancer: a 10-year follow-up. J Urol. 1992;147:1020.
Hisataki T, Miyao N, Masumori N, et al. Risk factors for the development of bladder cancer after upper tract urothelial cancer. Urology. 2000;55:663-667.
Ho C, Tzai T, Chen J, et al. The molecular signature for urothelial carcinoma of the upper urinary tract. J Urol. 2008;179:1155-1159.
Holmang S, Johansson SL. Synchronous bilateral ureteral and renal pelvic carcinomas: incidence, etiology, treatment and outcome. Cancer. 2004;101:741.
Holmang S, Thomsen J, Johansson SL. Micropapillary carcinoma of the renal pelvis and ureter. J. Urol. 2006;175:463-467.
Holtz F. Papillomas and primary carcinoma of the ureter: report of 20 cases. J Urol. 1962;88:380-385.
Hsueh TY, Huang YH, Chiu AW, et al. A comparison of the clinical outcome between open and hand-assisted laparoscopic nephroureterectomy for upper urinary tract transitional cell carcinoma. BJU Int. 2004;94:798-801.
Huang A, Low RK, DeVere White R. Nephrostomy tract tumor seeding following percutaneous manipulation of a ureteral carcinoma. J Urol. 1995;153:1041-1042.
Huben RP, Mounzer AM, Murphy GP. Tumor grade and stage as prognostic variables in upper tract urothelial tumors. Cancer. 1988;62:2016-2020.
Hudson M, Herr HW. Carcinoma in situ of the bladder. J Urol. 1995;153:564.
Huffman JL. Ureteroscopic management of transitional cell carcinoma of the upper urinary tract. Urol Clin North Am. 1988;15:419-424.
Huffman JL, Bagley DH, Lyon ES, et al. Endoscopic diagnosis and treatment of upper tract urothelial tumors. Cancer. 1985;55:1422-1428.
Iborra I, Solsona E, Casanova J, et al. Conservative elective treatment of upper urinary tract tumors: a multivariate analysis of prognostic factors for recurrence and progression. J Urol. 2003;169:82.
Igel TC, Engen DE, Banks PM, et al. Renal plasmacytoma: Mayo Clinic experience and review of the literature. Urology. 1991;37:385.
Jabbour ME, Desgrandchamps F, Cazin S, et al. Percutaneous management of grade II upper urinary tract transitional cell carcinoma: the long-term outcome. J Urol. 2000;163:1105-1107.
Jarrett TW, Chan DY, Cadeddu JA, Kavoussi LR. Laparoscopic nephroureterectomy for the treatment of transitional cell carcinoma of the upper urinary tract. Urology. 2001;57:448-453.
Jarrett TW, Lee CK, Pardalidis NP, Smith AD. Extensive dilation of distal ureter for endoscopic treatment of large volume ureteral disease. J Urol. 1995;153:1214-1217.
Jarrett TW, Sweetser PM, Weiss GH, Smith AD. Percutaneous management of transitional cell carcinoma of the renal collecting system: 9-year experience. J Urol. 1995;154:1629-1635.
Jemal A, Tiwari RC, Murray T, et al. Cancer statistics, 2004. CA Cancer J Clin. 2004;54:8.
Jensen OM, Knudsen JB, McLaughlin JK, et al. The Copenhagen case-control study of renal pelvis and ureter cancer: role of smoking and occupational exposures. Int J Cancer. 1988;41:557.
Jitsukawa S, Nakamura K, Nakayama M, et al. Transitional cell carcinoma of kidney extending into renal vein and inferior vena cava. Urology. 1985;25:310-312.
Johansson S, Angervall L, Bengtsson U, Wahlquist L. Uroepithelial tumors of the renal pelvis associated with abuse of phenacetin-containing analgesics. Cancer. 1974;33:743-753.
Johansson S, Angervall L, Bengtsson U, Wahlquist L. A clinicopathologic and prognostic study of epithelial tumors of the renal pelvis. Cancer. 1976;37:1376-1383.
Johansson S, Wahlquist L. A prognostic study of urothelial renal pelvic tumors: comparison between the prognosis of patients treated with intrafascial nephrectomy and perifascial nephroureterectomy. Cancer. 1979;43:2525.
Johnson DE, Babaian RJ. Conservative surgical management for noninvasive distal ureteral carcinoma. Urology. 1979;13:365-367.
Johnson DE, Swanson DA, Von Eschenbach AC. Tumors of the genitourinary tract. In: Smith DR, editor. General urology. Los Altos (CA): Lange Medical; 1988:330-434.
Jones DR, Moisey CU. A cautionary tale of the modified “pluck” nephroureterectomy. Br J Urol. 1993;71:486.
Kakizoe T, Fujita J, Murase T, et al. Transitional cell carcinoma of the bladder in patients with renal pelvic and ureteral cancer. J Urol. 1980;124:17-19.
Kamai T, Takagi K, Asami H, et al. Prognostic significance of p27 Kipl and Ki-67 expression in carcinoma of the renal pelvis and ureter. BJU Int. 2000;86:14.
Kang CH, Chiang PH, Huang SC. Correlation of COX-2 expression in stromal cells with high stage, high grade, and poor prognosis in urothelial carcinoma of upper urinary tracts. Urology. 2008;72:153-157.
Kang CH, Yu TJ, Hsieh HH, et al. The development of bladder tumors and contralateral upper urinary tract tumors after primary transitional cell carcinoma of the upper urinary tract. Cancer. 2003;98:1620-1626.
Kaufman D, Raghavan D, Carducci M, et al. Phase II trial of gemcitabine plus cisplatin in patients with metastatic urothelial cancer. J Clin Oncol. 2000;18:1921-1927.
Ke HL, Wei YC, Li CC, et al. Overexpression of hypoxia-inducible 1 alpha predicts an unfavorable outcome in urothelial carcinoma of the upper urinary tract. Int J Urol. 2008;15:200-205.
Keeley FX, Bibbo M, Bagley DM. Ureteroscopic treatment and evaluation of the upper tract TCC. J Urol. 1997;157:1560.
Keeley FX, Bibbo M, McCue PA, Bagley DH. Use of p53 in the diagnosis of upper tract transitional cell carcinoma. Urology. 1997;49:181.
Keeley FX, Kulp DA, Bibbo M, et al. Diagnostic accuracy of ureteroscopic biopsy in upper tract transitional cell carcinoma. J Urol. 1997;157:33-37.
Keeley FX, Tolley DA. Laparoscopic nephroureterectomy: making management of upper-tract transitional-cell carcinoma entirely minimally invasive. J Endourol. 1998;12:139-141.
Kenney PJ, Stanley RJ. Computed tomography of ureteral tumors. J Comput Assist Tomogr. 1987;11:102.
Kerbl K, Clayman RV. Incision of the ureterovesical junction for endoscopic surveillance of transitional cell cancer of the upper urinary tract. J Urol. 1993;150:1440-1443.
Khan S, Farrow GM, Zincke H, et al. Primary carcinoma in situ of the ureter and renal pelvis. J Urol. 1979;121:681-683.
Kikuchi E, Horiguchi Y, Nakashima J, et al. Lymphovascular invasion independently predicts increased disease specific survival in patients with transitional cell carcinoma of the upper urinary tract. J Urol. 2005;174:2120.
Kikuchi E, Margolis V, Karakiewicz P, et al. Lymphovascular invasion predicts clinical outcomes in patients with node-nagative upper tract urothelial carcinoma. J Clin Oncol. 2009;27:612-618.
Komatsu H, Tanabe N, Kubodera S, et al. The role of lymphadenectomy in the treatment of transitional cell carcinoma of the upper urinary tract. J Urol. 1997;157:1622.
Kondo T, Nakazawa H, Ito F, et al. Impact of the extent of regional lymphadenectomy on the survival of patients with urothelial carcinoma of the upper urinary tract. J Urol. 2007;178:1212-1217.
Konety BR, Getzenberg RH. Urine-based markers of urologic malignancy. J Urol. 2001;165:600.
Korman HJ, Watson R, Soloway MS. Bladder cancer: clinical aspects and management. Monogr Urol. 1996;16:83-110.
Kulp DA, Bagley DH. Does flexible uretero-pyeloscopy promote local recurrence of transitional cell carcinoma? J Endourol. 1994;8:111-113.
Landman J, Lev RY, Bhayani S, et al. Comparison of hand-assisted and standard laparoscopic radical nephroureterectomy for the management of localized transitional cell carcinoma. J Urol. 2002;167:2387.
Lantz EJ, Hattery RR. Diagnostic imaging of urothelial cancer. Urol Clin North Am. 1984;11:576.
Leach FS, Hsieh JT, Molberg K, et al. Expression of the human mismatch repair gene hMSH2: a potential marker for urothelial malignancy. Cancer. 2000;88:2333-2341.
Lee BR, Jabbour ME, Marshall FF, et al. 13-year survival comparison of percutaneous and open nephroureterectomy approaches for management of transitional cell carcinoma of renal collecting system: equivalent outcomes. J Endourol. 1999;13:289-294.
Leiber MM, Lupu AN. High-grade invasive ureteral transitional cell carcinoma with a congenital solitary kidney: long-term survival after ureterectomy and radiation therapy. J Urol. 1978;120:368-369.
Lim DJ, Shattuck MC, Cook WA. Pyelovenous lymphatic migration of transitional cell carcinoma following flexible ureterorenoscopy. J Urol. 1993;149:109-111.
Lin W, Hu F, Chung S, et al. The role of lymphovascular invasion in predicting the prognosis of clinically localized upper tract urothelial carcinoma (pT1-3cN0M0). J Urol. 2008;180:879-885.
Loening S, Narayana A, Yoder L, et al. Factors influencing the recurrence rate of bladder cancer. J Urol. 1980;123:29-31.
Lorusso V, Manzione L, De Vita F, et al. Gemcitabine plus cisplatin for advanced transitional cell carcinoma of the urinary tract: a phase II multicenter trial. J Urol. 2000;164:53.
Lunec J, Challen C, Wright C, et al. Amplification of c-erbB2 and mutation of p53 in concomitant transitional carcinomas of renal pelvis and urinary bladder. Lancet. 1992;339:439.
Luo B, Li W, Deng CH, et al. Utility of fluorescence in situ hybridization in the diagnosis of upper urinary tract urothelial carcinoma. Cancer Genet Cytogenet. 2009;189:93-97.
Lynch HT, Ens JA, Lynch JF. The Lynch syndrome II and urological malignancies. J Urol. 1990;143:24.
Macejko AM, Pazona JF, Loeb S, et al. Management of the distal ureter in laparoscopic nephroureterectomy: a comprehensive review of techniques. Urology. 2008;72:974-981.
Madgar I, Goldwasser B, Nativ O, et al. Long-term follow-up of patients less than 30 years old with transitional cell carcinoma of the bladder. J Urol. 1988;139:933.
Mahadevia PA, Karwa GL, Koss LG. Mapping of urothelium in carcinomas of the renal pelvis and ureter: a report of nine cases. Cancer. 1983;51:890-897.
Mahoney JF, Storey BG, Ibanez RC, et al. Analgesic abuse: renal parenchymal disease and carcinoma of the kidney or ureter. Aust N Z J Med. 1977;7:463-469.
Maier U, Mertl G, Pummer K, et al. Organ-preserving surgery in patients with urothelial tumors of the upper urinary tract. Eur Urol. 1990;18:197.
Markovic B. Endemic nephritis and urinary tract cancer in Yugoslavia, Bulgaria, and Romania. J Urol. 1972;107:212-219.
Martinez-Pineiro JA, Garcia Matres MJ, Martinez-Pineiro L. Endourological treatment of upper tract urothelial carcinomas: analysis of a series of 59 tumors. J Urol. 1996;156:377-385.
Matin SF, Gill IS. Recurrence and survival following laparoscopic radical nephroureterectomy with various forms of bladder cuff control. J Urol. 2005;173:395-400.
Matsui Y, Ohara H, Ichioka K, et al. Abdominal wall metastasis after retroperitoneoscopic assisted total nephroureterectomy for renal pelvic cancer. J Urol. 2004;171(Pt. 1):793.
Matsui Y, Utsunomiya N, Ichioka K, et al. Risk factors for subsequent development of bladder cancer after primary transitional cell carcinoma of the upper urinary tract. Urology. 2005;54:279-283.
Matsuoka K, Lida S, Tomiyasu K, et al. Transurethral endoscopic treatment of upper urinary tract tumors using a holmium:YAG laser. Lasers Surg Med. 2003;32:336-340.
Maulard-Durdux C, Dufour B, Hennequin C, et al. Postoperative radiation therapy in 29 patients with invasive transitional cell carcinoma of the upper urinary tract: No impact on survival? J Urol. 1996;155:115-117.
Mazeman E. Tumors of the upper respiratory tract calyces, renal pelvis, and ureter. Eur Urol. 1976;2:120-126.
McCarron JPJr, Chasko SB, Gray GFJ. Systematic mapping of nephroureterectomy specimens removed for urothelial cancer: pathological findings and clinical correlates. J Urol. 1982;128:243-246.
McCarron JPJr, Mills C, Vaughn EDJr. Tumors of the renal pelvis and ureter: current concepts and management. Semin Urol. 1983;1:75-81.
McCredie M, Stewart JH, Carter JJ, et al. Phenacetin and papillary necrosis: independent risk factors for renal pelvic cancer. Kidney Int. 1986;30:81.
McDonald DF. Intussusception ureterectomy: a method of removal of the ureteral stump at time of nephrectomy without an additional incision. Surg Gynecol Obstet. 1953;97:565.
McLaughlin JK, Silverman DT, Hsing AW, et al. Cigarette smoking and cancers of the renal pelvis and ureter. Cancer Res. 1992;52:254.
McNeill SA, Chrisofos M, Tolley DA. The long-term outcome after laparoscopic nephroureterectomy: a comparison with open nephroureterectomy. BJU Int. 2000;86:619.
McTavish JD, Jinzaki M, Zou KH, et al. Multi-detector row CT urography: comparison of strategies for depicting the normal urinary collecting system. Radiology. 2002;225:783.
Melamed MR, Reuter VE. Pathology and staging of urothelial tumors of the kidney and ureter. Urol Clin North Am. 1993;20:333.
Messing EM, Catalona W. Urothelial tumors of the urinary tract. In: Walsh PC, Retik AD, Vaughan ED, et al, editors. Campbell’s urology. 7th ed. Philadelphia: WB Saunders; 1998:2327-2410.
Milestone B, Freidman AC, Seidmon EJ, et al. Staging of ureteral transitional cell carcinoma by CT and MRI. Urology. 1990;36:346.
Miyake H, Hara I, Arakawa S, Kamidono S. A clinical pathological study of bladder cancer associated with upper urinary tract cancer. BJU Int. 2000;85:37.
Morrison AS. Advances in the etiology of urothelial cancer. Urol Clin North Am. 1984;11:557.
Mufti GR, Gove JRW, Riddle R. Nephroureterectomy after radical cystectomy. J Urol. 1988;139:588-589.
Mukamel E, Vilkovsky E, Hadar H, et al. The effect of intravesical bacillus Calmette-Guérin therapy on the upper urinary tract. J Urol. 1991;146:980-981.
Mullen JB, Kovacs D. Primary carcinoma of the ureteral stump: a case report and a review of the literature. J Urol. 1980;123:113-115.
Mullerad M, Russo P, Golijanin D, et al. Bladder cancer as a prognostic factor for upper tract transitional cell carcinoma. J Urol. 2004;172:2177-2181.
Munoz JJ, Ellison LM. Upper tract urothelial neoplasms: incidence and survival the last 2 decades. J Urol. 2000;164:1523-1525.
Muntener M, Nielson ME, Romero FR, et al. Long-term oncologic outcome after laparoscopic radical nephroureteretomy for upper tract transitional cell carcinoma. Eur Urol. 2007;51(6):1639-1644.
Muntener M, Schaeffer EM, Romero FR, et al. Incidence of local recurrence and port site metastasis after laparoscopic radical nephroureterectomy. Urology. 2007;70(5):864-868.
Murphy DM, Zincke H, Furlow WL. Primary grade I transitional cell carcinoma of the renal pelvis and ureter. J Urol. 1980;123:629-631.
Murphy DM, Zincke H, Furlow WL. Management of high grade transitional cell cancer of the upper urinary tract. J Urol. 1981;125:25-29.
Murphy WM, Soloway MS. Urothelial dysplasia. J Urol. 1982;127:849.
Murphy WM, von Buiduigen RP, Poley RW. Primary carcinoma in situ of renal pelvis and ureter. Cancer. 1974;34:1126-1130.
Musselman P, Kay R. The spectrum of urinary tract polyps in children. J Urol. 1986;136:476.
Nakazono M, Muraki J. Lymphadenectomy in the operative treatment of renal pelvic and ureteral cancers. Jpn J Urol. 1993;84:668.
Nocks BN, Heney NM, Daly JJ, et al. Transitional cell carcinoma of renal pelvis. Urology. 1982;14:472-477.
Notley RG. Ureteral morphology. Urology. 1978;12:8.
Novara G, De Marco V, Dalpiaz O, et al. Independent predictors of metachronous bladder transitional cell carcinoma (TCC) after nephroureterectomy for TCC of the upper urinary tract. BJU Int. 2008;101:1368-1374.
Nurse DE, Woodhouse CR, Kellett MJ, Dearnley DP. Percutaneous removal of upper tract tumors. World J Urol. 1989;7:131.
Oefelein MG, MacLennan G. Transitional cell carcinoma recurrence in the nephrostomy tract after percutaneous resection. J Urol. 2003;170(Pt. 1):521.
Okada H, Eto H, Hara I, et al. Percutaneous treatment of transitional cell carcinoma of the upper urinary tract. Int J Urol. 1997;4:130-133.
Oldbring J, Glifberg I, Mikulowski P, et al. Carcinoma of the renal pelvis and ureter following bladder carcinoma: frequency, risk factors and clinicopathological findings. J Urol. 1989;141:1311.
Ong AM, Bhayani SB, Pavlovich CP. Trocar site recurrence after laparoscopic nephroureterectomy. J Urol. 2003;170(Pt. 1):1301.
Orphali SL, Shols GW, Hagewood T, et al. Familial transitional cell carcinoma of renal pelvis and upper ureter. Urology. 1986;27:394.
Orsola A, Trias I, Raventos CX, et al. Renal collecting (Bellini) duct carcinoma displays similar characteristics to upper tract urothelial cell carcinoma. Urology. 2005;65:49.
Otani M, Irie S, Tsuji Y. Port site metastasis after laparoscopic nephrectomy: unsuspected transitional cell carcinoma within a tuberculous atrophic kidney. J Urol. 1999;162:486-487.
Oyasu R. World Health Organization and International Society of Urologic Pathology classification and two-number grading system of bladder tumors. Cancer. 2000;88:1509-1512.
Pagano F. Conservative treatment of lower ureteral tumor: modified ureteroneocystostomy for upper urinary tract endoscopic control. J Urol. 1984;132:555-557.
Palou J, Caparros J, Orsola A, et al. Transurethral resection of the intramural ureter as the first step of nephroureterectomy. J Urol. 1995;154:43-44.
Palou J, Piovesan LF, Huguet J, et al. Percutaneous nephroscopic management of upper urinary tract transitional cell carcinoma: recurrence and long-term followup. J Urol. 2004;172:66-69.
Palou J, Salvador J, Millan F, et al. Management of superficial transitional cell carcinoma in the intramural ureter: what to do? J Urol. 2000;163:744-747.
Palvio DH, Andersen JC, Falk E. Transitional cell tumor of the renal pelvis and ureter associated with capillarosclerosis indicating analgesic abuse. Cancer. 1987;59:972.
Park S, Hong B, Kim CS, Ahn H. The impact of tumor location on prognosis of transitional cell carcinoma of the upper urinary tract. J Urol. 2004;171:621-625.
Patel A, Fuchs GJ. New techniques for the administration of tropical adjuvant therapy after endoscopic ablation of upper tract transitional cell carcinoma. J Urol. 1998;159:71-75.
Patel A, Soonawalla P, Shepherd SF, et al. Long-term outcome after percutaneous treatment of transitional cell carcinoma of the renal pelvis. J Urol. 1996;155:868-874.
Petkovic SD. A plea for conservative operation for ureteral tumors. J Urol. 1972;103:220-223.
Petkovic SD. Conservation of the kidney in operations for tumors of the renal pelvis and calyces: a report of 26 cases. Br J Urol. 1972;44:1-8.
Petkovic SD. Epidemiology and treatment of renal pelvic and ureteral tumors. J Urol. 1975;114:858-865.
Pettersson S, Brynger H, Henriksson C, et al. Treatment of urothelial tumors of the upper urinary tract by nephroureterectomy, renal autotransplantation, and pyelocystostomy. Cancer. 1984;54:379-386.
Png KS, Lim EK, Chong KT, et al. Prognostic factors for upper tract transitional cell carcinoma : a retrospective review of 66 patients. Asian J Surg. 2008;31:20-24.
Premoli J, et al. Risk factors for upper tract recurrence in patients undergoing long-term surveillance for stage Ta bladder cancer. J Urol. 2006;175:74-77.
Raabe NK, Fossa SD, Bjerkehagen B. Carcinoma of the renal pelvis. Scand J Urol Nephrol. 1992;26:357.
Rabbani F, Perrotti M, Russo P, Herr HW. Upper-tract tumors after an initial diagnosis of bladder cancer: argument for long-term surveillance. J Clin Oncol. 2001;19:94-100.
Radovanovic Z, Krajinovic S, Jankovic S, et al. Family history of cancer among cases of upper urothelial tumours in the Balkan nephropathy area. J Cancer Res Clin Oncol. 1985;110:181.
Raman J, Sosa RE, Vaughan ED, Scherr DS. Pathologic features of bladder tumors after nephroureterectomy for upper tract transitional cell carcinoma. Urology. 2007;69:251-254.
Rastinehad AR, Vanderbrink BA, Greenberg KL, et al. A 20 year experience with percutaneous resection of upper tract tract transitional cell carcinoma: is there an oncologic benefit with adjuvant bacillus Calmette Guérin. Urology. 2009;73(1):27-31.
Razvi HA, Chun SS, Denstedt JD, Sales JL. Soft-tissue applications of the holmium:YAG laser in urology. J Endourol. 1995;9:387-390.
Redman B, Smith D, Flaherty L, et al. Phase II trial of paclitaxel and carboplatin in the treatment of advanced urothelial carcinoma. J Clin Oncol. 1998;16:1844-1848.
Renfer LG, Kelley J, Belville WD. Inverted papilloma of the urinary tract: histogenesis, recurrence, and associated malignancy. J Urol. 1988;140:832.
Ressequie LT, Nobrega FT, Farrow GM, et al. Epidemiology of renal and ureteral cancer in Rochester, Minnesota, 1950–1974, with special reference to clinical and pathologic features. Mayo Clin Proc. 1978;53:503.
Richie JP. Carcinoma of the renal pelvis and ureter. In: Skinner DG, Lieskovsky G, editors. Diagnosis and management of genitourinary cancer. Philadelphia: WB Saunders; 1988:323-336.
Romero FR, Schaeffer EM, Muntener M, et al. Oncologic outcomes of extravesical stapling of distal ureter in laparoscopic nephroureterectomy. J Endourol. 2007;21:1025-1027.
Ross RK, Paganini-Hill A, Landolph J, et al. Analgesics, cigarette smoking, and other risk factors for cancer of the renal pelvis and ureter. Cancer Res. 1989;49:1045.
Roth B, Dreicer R, Einhorn L, et al. Significant activity of paclitaxel in advanced transitional cell carcinoma of the urothelium: a phase II trial of the Eastern Cooperative Oncology Group. J Clin Oncol. 1994;12:2264-2270.
Roth S, van Ahlen H, Semjonow A, et al. Modified ureteral stripping as an alternative to open surgical ureterectomy. J Urol. 1996;155:1568.
Rouprêt M, Harmon JD, Sanderson KM, et al. Laparoscopic distal ureterectomy and anastomosis for management of low-risk upper urinary tract transitional cell carcinoma: preliminary results. BJU Int. 2007;99(3):623-627.
Rouprêt M, Hupertan V, Traxer D, et al. Comparison of open nephroureterectomy and ureteroscopic and percutaneous management of upper urinary tract transitional cell carcinoma. Urology. 2006;67:1181-1187.
Sakamoto N, Naito S, Kotoh S, et al. Recurrence of bladder tumors following surgery for transitional cell carcinoma of the upper urinary tract. Eur Urol. 1991;20:136-139.
Salomon L, Hoznek A, Cicco A, et al. Retroperitoneoscopic nephroureterectomy for renal pelvic tumors with a single iliac incision. J Urol. 1999;161:541-544.
Sarnacki CT, McCormack LJ, Kiser WS, et al. Urinary cytology and the clinical diagnosis of urinary tract malignancy: a clinicopathologic study of 1400 patients. J Urol. 1971;106:761-764.
Schatteman P, Chatzopoulos C, Assenmacher C. Laparoscopic nephroureterectomy for upper urinary tract transitional cell carcinoma: results of a Belgian retrospective multicentre survey. Eur Urol. 2007;51(6):1633.
Schilling A, Bowering R, Keiditsch E. Use of the neodymium:YAG laser in the treatment of ureteral tumors and urethral condylomata acuminata: clinical experience. Eur Urol. 1986;12(Suppl. 1):30.
Schmeller NT, Hofstetter AG. Laser treatment of ureteral tumors. J Urol. 1989;141:840-843.
Scolieri MJ, Paik ML, Brown SL, Resnick MI. Limitations of computed tomography in the preoperative staging of upper tract urothelial carcinoma. Urology. 2000;56:930.
Secin FP, Koppie TM, Martinez Salamanca J, et al. Clinical and pathological predictors of pathologic lymph node metastasis identified during nephroureterectomy for transitional cell carcinoma of the upper urinary tract. J Urol. 2005;173:265.
See WA. Continuous antegrade infusion of adriamycin as adjuvant therapy for upper tract urothelial malignancies. Urology. 2000;56:216.
Shalhav AL, Dunn MD, Portis AJ, et al. Laparoscopic nephroureterectomy for upper tract transitional cell cancer: the Washington University experience. J Urol. 2000;163:1100-1104.
Sharpe JR, Duffy G, Chin JL. Intrarenal bacillus Calmette-Guérin therapy for upper urinary tract carcinoma in situ. J Urol. 1993;149:457-460.
Shinka T, Uekado Y, Aoshi H, et al. Occurrence of uroepithelial tumors of the upper urinary tract after initial diagnosis of bladder cancer. J Urol. 1988;140:745-748.
Siemens DR, Morales A, Johnston B, Emerson L. A comparative analysis of rapid urine tests for the diagnosis of upper tract malignancy. Can J Urol. 2003;10:1754.
Skinner D. Technique of nephroureterectomy with regional lymph node dissection. Urol Clin North Am. 1978;5:253-260.
Skolarikos A, Griffiths TR, Powell PH, et al. Cytologic analysis of ureter washings is informative in patients with grade 2 upper tract TCC considering endoscopic treatment. Urology. 2003;61:1146.
Slaton JW, Swanson DA, Grossman HB, Dinney CP. A stage specific approach to tumor surveillance after radial cystectomy for transitional cell carcinoma of the bladder. J Urol. 1999;162:710-714.
Slywotzky C, Maya M. Needle tract seeding of transitional cell carcinoma following fine-needle aspiration of a renal mass. Abdom Imaging. 1994;19:174-176.
Smith AY, Vitale PJ, Lowe BA, Woodside JR. Treatment of superficial papillary transitional cell carcinoma of the ureter by vesicoureteral reflux of mitomycin C. J Urol. 1987;138:1231-1233.
Smith JAJr, Lee RG, Dixon JA. Tissue effects of neodymium:YAG laser photoradiation of canine ureters. J Surg Oncol. 1984;27:168-171.
Solsona E, Iborra I, Ricos JV, et al. Upper urinary tract involvement in patients with bladder carcinoma in situ (CIS): its impact on management. Urology. 1997;49:347-352.
Sowter SJ, Ilie CP, Efthimiou I, Tolley DA. Endourologic management of patients with upper-tract transitional-cell carcinoma: long-term follow-up in a single center. J Endourol. 2007;21(9):1005-1009.
Spires SE, Banks ER, Cibull ML, et al. Adenocarcinoma of renal pelvis. Arch Pathol Lab Med. 1993;117:1156.
Spruck CH, Ohneseit PF, Gonzales-Zulueta M, et al. Two molecular pathways to transitional cell carcinoma of the bladder. Cancer Res. 1994;54:784.
Stefanovic V, Radovanovic Z. Balkan endemic nephropathy and associated urothelial cancer. Nat Clin Pract Urol. 2008;5:105.
Steffens J, Nagel R. Tumours of the renal pelvis and ureter: observations in 170 patients. Br J Urol. 1988;61:277.
Stein A, Sova Y, Lurie M, et al. Adenocarcinoma of the renal pelvis: report of two cases, one with simultaneous transitional cell carcinoma of the bladder. Urol Int. 1988;43:299.
Sternberg DW, Yagoda A, Scher HI, et al. Methotrexate, vinblastine, doxorubicin, and cisplatin for advanced transitional cell carcinoma of the urothelium: efficacy and patterns of response and relapse. Cancer. 1989;64:2448-2458.
Stewart GD, Bariol SV, Grigor KM, et al. Upper tract transitional cell carcinoma is of higher grade and stage than bladder TCC. J Urol. 2005;173:268.
Stewart JH, Hobbs JB, McCredie MRE. Morphologic evidence that analgesic-induced kidney pathology contributes to the progression of tumors of the renal pelvis. Cancer. 1999;86:1576-1582.
Stifleman MD, Hyman MJ, Shichman S, Sosa RE. Hand-assisted laparoscopic nephroureterectomy versus open nephroureterectomy for the treatment of transitional-cell carcinoma of the upper urinary tract. J Endourol. 2001;15:391-395.
Stifleman MD, Sosa RE, Andrade A, et al. Hand-assisted laparoscopic nephroureterectomy for the treatment of transitional cell carcinoma of the upper urinary tract. Urology. 2000;56:741-747.
Stower MJ, MacIver AG, Gingell JC, et al. Inverted papilloma of the ureter with malignant change. Br J Urol. 1990;65:13.
Streem SB, Pontes JE, Novick AC, et al. Ureteropyeloscopy in the evaluation of upper tract filling defects. J Urol. 1986;136:388.
Strong DW, Pearse HD, Tank ES, Hodges CV. The ureteral stump after nephroureterectomy. J Urol. 1976;115:654-655.
Studer UE, Casanova G, Kraft R. Percutaneous bacillus Calmette-Guérin perfusion of the upper urinary tract for carcinoma in situ. J Urol. 1989;142:975-977.
Suoka K, Lida S, Tomiyasu K, et al. Transurethral endoscopic management of upper urinary tract tumors using a holmium:YAG laser. Lasers Surg Med. 2003;32:336.
Sved PD, Gomez P, Nieder AM, et al. Upper tract tumour after radical cystectomy for transitional cell carcinoma of the bladder: incidence and risk factors. BJU Int. 2004;94:785-789.
Takahashi T, Kakehi Y, Mitsumori K, et al. Distinct microsatellite alterations in upper urinary tract tumors and subsequent bladder tumors. J Urol. 2001;165:672-677.
Takahashi T, Mitsumori K, Kakehi Y, et al. Distinct microsatellite alterations between upper urinary tract tumors and subsequent bladder tumors. J Urol. 2000;163(Suppl.):549A.
Tan LB, Chen KT, Guo HR. Clinical and epidemiological features of patients with genitourinary tract tumour in a blackfoot disease endemic area of Taiwan. BJU Int. 2008;102:48-54.
Tawfiek ER, Bagley D. Upper-tract transitional cell carcinoma. Urology. 1997;50:321-329.
Tawfiek ER, Bibbo M, Bagley D. Ureteroscopic biopsy: technique and specimen preparation. Urology. 1997;50:117-119.
Terakawa T, Miyake H, Muramaki M, et al. Risk factors for intravesical recurrence after surgical management of transitional cell carcinoma of the upper urinary tract. Urology. 2008;71:123-127.
Terrell RB, Cheville JC, See WA, Cohen MB. Histopathologic features and p53 nuclear protein staining as predictors of survival and tumor recurrence in patients with transitional cell carcinoma of renal pelvis. J Urol. 1995;154:1342-1347.
Terris MK. The effect of contrast material on transitional cell carcinoma viability. Adv Exp Med Biol. 2004;539:121.
Thalmann GN, Markwalder R, Walter B, Studer UE. Long-term experience with bacillus Calmette-Guérin therapy of upper urinary tract transitional cell carcinoma in patients not eligible for surgery. J Urol. 2002;168:1381.
Thompson RH, Drambeck AE, Lohse CM, et al. Endoscopic management of upper tract transitional cell carcinoma in patients with normal contralateral kidneys. Urology. 2008;7:713-717.
Tomera KM, Leary FJ, Kinke H. Pyeloscopy in urothelial tumors. J Urol. 1982;127:1088-1089.
Tran W, Serio AM, Raj GV, et al. Longitudinal risk of upper tract recurrence following radical cystectomy for urothelial cancer and the potential implications for long-term surveillance. J Urol. 2008;179:96-100.
Treuthardt C, Danuser H, Studer UE. Tumor seeding following percutaneous antegrade treatment of transitional cell carcinoma in the renal pelvis. Eur Urol. 2004;46:442-443.
Varela-Duran J, Urdiales-Viedma M, Taboada-Blanco F, et al. Neurofibroma of the ureter. J Urol. 1987;138:1425-1426.
Varkarakis MJ, Gaeta J, Moore RH, Murphy GP. Superficial bladder tumor: aspects of clinical progression. Urology. 1974;4:414-420.
Vaughn D, Malkowicz S, Zoltick B, et al. Paclitaxel plus carboplatin in advanced carcinoma of the urothelium: an active and tolerable outpatient regimen. J Clin Oncol. 1998;16:255-260.
Vincente J, Pilar L, Sole-Balcells FJ, et al. Transitional cell carcinoma of the upper urinary tract: diagnosis and management. Urol Int. 1995;2:7.
Wagle DG, Moore R, Murphy GP. Secondary carcinomas of the kidney. J Urol. 1975;114:30-32.
Wagner KR, Schoenberg MP, Bianco FJJr, Jarrett TW. Prospective intermediate follow-up of carcinoma in situ involving the distal ureter at cystectomy: is there a role for ureteroscopy? J Endourol. 2008;22(6):1241-1246.
Waldert M, Remzi M, Klingler HC, et al. The oncological results of laparocopic nephroureterectomy for upper urinary tract transitional cell carcinoma. BJU Int. 2009;103(1):66-70.
Wallace DMA, Wallace DM, Whitfield HN, et al. The late results of conservative surgery for upper tract urothelial carcinomas. Br J Urol. 1981;53:537-541.
Walton TJ, Sherwood BT, Parkinson RJ, et al. Comparative outcomes following endoscopic ureteral detachment and formal bladder cuff excision in open nephroureterectomy for upper tract transitional cell carcinoma. J Urol. 2009;181:532-539.
Weston PM, Greenland JE, Wallace DM. Role of topical mitomycin C in upper urinary tract transitional cell carcinoma. Br J Urol. 1993;71:624-625.
Williams RD. Renal, perirenal, and ureteral neoplasms. In Gillenwater JY, Grayhack JT, Howards SS, Duckett JW, editors: Adult and pediatric urology, 2nd ed, St. Louis: Mosby–Year Book, 1991.
Wolf JSJr, Dash A, Hollenbeck BK, et al. Intermediate followup of hand assisted laparoscopic nephroureterectomy for urothelial carcinoma: factors associated with outcomes. J Urol. 2005;173:1102-1107.
Wright JL, Hotaling J, Porter MP. Predictors of upper tract urothelial cell carcinoma after primary bladder cancer: a population based analysis. J Urol. 2009;181:1035-1039.
Wu WJ, Liu LT, Huang CN, et al. The clinical implications of telomerase activity in upper tract cancer and washings. BJU Int. 2000;86:213.
Yip SK, Peh WC, Li JH, Lam CH. Role of ultrasonography in screening for urological malignancies in patients presenting with painless hematuria. Ann Acad Med Singapore. 1999;28:174.
Ziegelbaum M, Novick AC, Streem SB. Conservative surgery for transitional cell carcinoma of the renal pelvis. J Urol. 1987;138:1146-1149.
Zigeuner R, Tsybrovskyy O, Ratschek M, et al. Prognostic impact of p63 and p53 expression in upper urinary tract transitional cell carcinoma. Urology. 2004;63:1079-1083.
Zincke H, Aguilo JJ, Farrow GM, et al. Significance of urinary cytology in the early detection of transitional cell cancer of the upper urinary tract. J Urol. 1976;116:781-783.
Zincke H, Garbeff PJ, Beahrs JR. Upper urinary tract transitional cell cancer after radical cystectomy for bladder cancer. J Urol. 1984;131:50-52.
Zincke H, Neves RJ. Feasibility of conservative surgery for transitional cell cancer of the upper urinary tract. Urol Clin North Am. 1984;11:717-724.
Zungri E, Chechile G, Algaba F, et al. Treatment of transitional cell carcinoma of the ureter: is the controversy justified? Eur Urol. 1990;17:276-280.