54 Picornaviruses
A 9-day-old infant with a fever and appearing septic progressed to multisystem organ syndrome with a combination of hepatitis, meningoencephalitis, myocarditis, and pneumonia. The cerebrospinal fluid (CSF) had normal glucose levels and lacked neutrophil infiltrate. The infant was started on acyclovir therapy for a suspected congenital herpes simplex virus (HSV) infection. Genome analysis (polymerase chain reaction [PCR] and reverse transcriptase [RT]-PCR) of the CSF did not detect HSV but did detect an enterovirus, which was subsequently identified as echovirus 11 and not Coxsackie B virus. Several days earlier, the mother had a slight fever and a cold.
1. How did the baby become infected?
2. How does the viral structure facilitate virus spread in the body and transmission to others?
3. What type of immunity is protective for this virus, and why was the baby not protected?
Answers
1. The baby could have been infected upon contact with fecal material from the mother but, just as likely, by contact with nasal secretions or an aerosol.
2. The virus is a naked capsid virus, and the capsid is impervious to acids, detergents, heat, and dryness. It can withstand the harsh conditions of the gastrointestinal tract and even insufficient sewage treatment. As a result, the virus is transmitted by the fecal oral route, but it can also infect the upper respiratory tract and cause common coldlike symptoms and can be transmitted by contact or aerosols.
3. Echovirus 11 kills the infected cell that it infects and then spreads to other cells. The most important immune response for protection is antibody, which will neutralize the released virus to prevent spread of the virus. Antibody in the serum also prevents the spread of the virus to the target tissue, which would be the meninges and brain.
Picornaviridae is one of the largest families of viruses and includes some of the most important human and animal viruses (Box 54-1). As the name indicates, these viruses are small (pico) ribonucleic acid (RNA) viruses that have a naked capsid structure. The family has more than 230 members divided into nine genera, including Enterovirus, Rhinovirus, Hepatovirus (Chapter 63), Cardiovirus, and Aphthovirus. The enteroviruses are distinguished from the rhinoviruses by the stability of the capsid at pH 3, the optimum temperature for growth, the mode of transmission, and their diseases (Box 54-2).
Box 54-2
Unique Properties of Human Picornaviruses
Virion is a naked, small (25 to 30 nm) icosahedral capsid enclosing a single-stranded positive RNA genome.
Enteroviruses are resistant to pH 3 to pH 9, detergents, mild sewage treatment, and heat.
Rhinoviruses are labile at acidic pH; optimum growth temperature is 33° C.
Genome is a messenger ribonucleic acid (mRNA).
Naked genome is sufficient for infection.
Virus replicates in cytoplasm.
Viral RNA is translated into polyprotein, which is then cleaved into enzymatic and structural proteins.
At least 90 serotypes of human enteroviruses exist; they are members of the polioviruses, group A or B Coxsackie viruses, or echoviruses. Several different disease syndromes may be caused by a specific serotype of enterovirus. Likewise, several different serotypes may cause the same disease, depending on the target tissue affected. Hepatitis A virus was included in this group but has been reclassified as a Hepatovirus and is discussed separately in Chapter 63.
The capsids of enteroviruses are very resistant to harsh environmental conditions (sewage systems) and the conditions in the gastrointestinal tract, which facilitates their transmission by the fecal-oral route. Although they may initiate infection in the gastrointestinal tract, the enteroviruses rarely cause enteric disease. In fact, most infections are usually asymptomatic. The best-known and most-studied picornavirus is poliovirus, of which there are three serotypes.
Coxsackieviruses are named after the town of Coxsackie, New York, where they were first isolated. They are divided into two groups, A and B, on the basis of certain biologic and antigenic differences and are further subdivided into numeric serotypes on the basis of additional antigenic differences.
The name echovirus is derived from enteric cytopathic human orphan because the disease associated with these agents was not initially known. Since 1967, newly isolated enteroviruses have been distinguished numerically.
The human rhinoviruses consist of at least 100 serotypes and are the major cause of the common cold. They are sensitive to acidic pH and replicate poorly at temperatures above 33° C. These properties usually limit rhinoviruses to causing upper respiratory tract infections.
Structure
The plus-strand RNA of the picornaviruses is surrounded by an icosahedral capsid that is approximately 30 nm in diameter. The icosahedral capsid has 12 pentameric vertices, each of which is composed of five protomeric units of proteins. The protomers are made of four virion polypeptides (VP1 to VP4). VP2 and VP4 are generated by the cleavage of a precursor, VP0. VP4 in the virion solidifies the structure, but it is not generated until the genome is incorporated into the capsid. This protein is released on binding of the virus to the cellular receptor. The capsids are stable in the presence of heat and detergent; with the exception of the rhinoviruses, they are also stable in acid. The capsid structure is so regular that paracrystals of virions often form in infected cells (Figures 54-1 and 54-2).
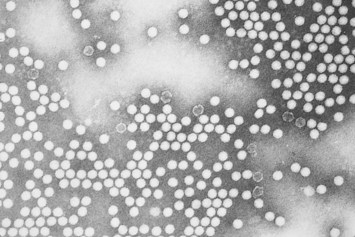
Figure 54-1 Electron micrograph of poliovirus.
(Courtesy Centers for Disease Control and Prevention, Atlanta.)
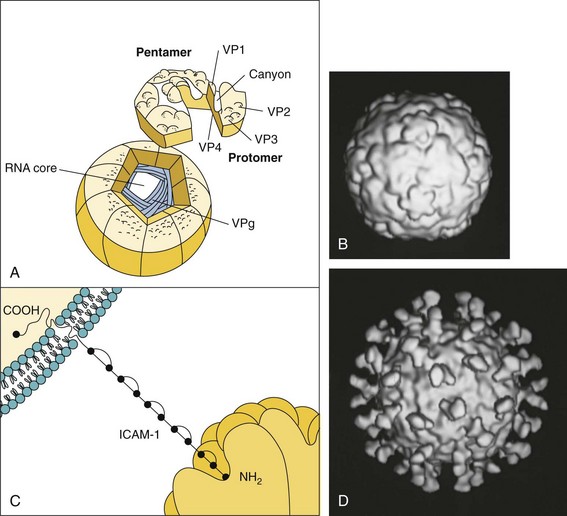
Figure 54-2 A, Structure of the human rhinovirus and its interaction with intercellular adhesion molecule-1 (ICAM-1) on the target cell. B, Cryoelectron microscopy computer-generated reconstruction of the human rhinovirus 16. C, Binding of the ICAM-1 molecule within the canyon of the virion triggers the opening of capsid for release of the genome into the cell. D, Cryoelectron microscopy reconstruction of the interaction of a soluble form of ICAM-1 with human rhinovirus 16. Note: There is one ICAM-1 per capsomere. RNA, ribonucleic acid; VP1, 2, 3, 4, viral protein 1, 2, 3, 4; VPg, viral protein genome-linked.
(B and D, Courtesy Tim Baker, Purdue University, West Lafayette, Ind.)
The genome of the picornaviruses resembles a messenger RNA (mRNA) (Figure 54-3). It is a single strand of plus-sense RNA of approximately 7200 to 8450 bases. It has a polyA (polyadenosine) sequence at the 3′ end and a small protein, VPg (viral protein genome-linked; 22 to 24 amino acids), attached to the 5′ end. The polyA sequence enhances the infectivity of the RNA, and the VPg may be important in packaging the genome into the capsid and initiating viral RNA synthesis. The naked picornavirus genome is sufficient for infection if microinjected into a cell.
Figure 54-3 Structure of the picornavirus genome. The genome (7200 to 8400 bases) is translated as a polyprotein, which is cleaved by viral-encoded proteases into individual proteins. •••, Internal ribosomal entry site for initiation of protein synthesis; gr, guanidine resistance marker (a genetic locus involved in the initiation of ribonucleic acid [RNA] synthesis); poly A, polyadenosine; VP1, 2, 3, 4, viral protein 1, 2, 3, 4; VPg, viral protein genome-linked.
The genome encodes a polyprotein that is proteolytically cleaved to produce the enzymatic and structural proteins of the virus. In addition to the capsid proteins and VPg, the picornaviruses encode at least two proteases and an RNA-dependent RNA polymerase.
Replication
The specificity of the picornavirus interaction for cellular receptors is the major determinant of the target tissue tropism and disease (see Chapter 44, Figure 44-13). The VP1 proteins at the vertices of the virion contain a canyon structure to which the receptor binds. The site of binding is protected from antibody neutralization. Pleconaril and related antiviral compounds contain a 3-methylisoxazole group that binds to the floor of this canyon and alters its conformation to prevent the uncoating of the virus.
The picornaviruses can be categorized according to their cell surface receptor specificity. The receptors for polioviruses, some Coxsackieviruses, and rhinoviruses are members of the immunoglobulin superfamily of proteins. At least 80% of the rhinoviruses and several serotypes of coxsackievirus bind to the intercellular adhesion molecule-1 (ICAM-1) expressed on epithelial cells, fibroblasts, and endothelial cells. Several Coxsackieviruses, echoviruses, and other enteroviruses bind to decay accelerating factor (CD55), and Coxsackie B virus shares a receptor with adenovirus. Poliovirus binds to a different molecule (PVR/CD155) that is similar to the receptor for HSV. The poliovirus receptor is present on many different human cells, but not all of these cells will replicate the virus.
On binding to the receptor, the VP4 is released and the virion weakened. The genome is then injected directly across the membrane through a channel created by the VP1 protein at one of the vertices of the virion. The genome binds directly to ribosomes, despite the lack of a 5′-cap structure. The ribosomes recognize a unique internal RNA loop (internal ribosome entry site [IRES]) in the genome that is also present in some cellular mRNAs. A polyprotein containing all the viral protein sequences is synthesized within 10 to 15 minutes of infection. This polyprotein is cleaved by viral proteases encoded in it. The viral RNA-dependent RNA polymerase generates a negative-strand RNA template from which the new mRNA/genome can be synthesized. The amount of viral mRNA increases rapidly in the cell, with the number of viral RNA molecules reaching as many as 400,000 per cell.
Most picornaviruses inhibit cellular RNA and protein synthesis during infection. For example, cleavage of the 200,000-Da cap-binding protein (EIF4-G) of the ribosome by a poliovirus protease prevents most cellular mRNA from binding to the ribosome. Inhibition of transcription factors decrease cellular mRNA synthesis, and permeability changes induced by picornaviruses reduce the ability of cellular mRNA to bind to the ribosome. In addition, viral mRNA can out-compete cellular mRNA for the factors required in protein synthesis. These activities contribute to the cytopathologic effect of the virus on the target cell.
As the viral genome is being replicated and translated, the structural proteins VP0, VP1, and VP3 are cleaved from the polyprotein by a viral-encoded protease and assembled into subunits. Five subunits associate into pentamers, and 12 pentamers associate to form the procapsid. After insertion of the genome, VP0 is cleaved into VP2 and VP4 to complete the capsid. As many as 100,000 virions per cell may be produced and released on cell lysis. The entire replication cycle may be as short as 3 to 4 hours.
Enteroviruses
Pathogenesis and Immunity
Contrary to their name, enteroviruses do not usually cause enteric disease, but they do replicate within and are transmitted by the fecal-oral route. The diseases produced by the enteroviruses are determined mainly by differences in tissue tropism and the cytolytic capacity of the virus (Figure 54-4; Box 54-3). The upper respiratory tract, the oropharynx, and the intestinal tract are the portals of entry for enteroviruses. The virions are impervious to stomach acid, proteases, and bile. Viral replication is initiated in the mucosa and lymphoid tissue of the tonsils and pharynx, and the virus later infects M cells and lymphocytes of the Peyer patches and enterocytes in the intestinal mucosa. Primary viremia spreads the virus to receptor-bearing target tissues, including the reticuloendothelial cells of the lymph nodes, spleen, and liver, to initiate a second phase of viral replication, resulting in a secondary viremia and symptoms.
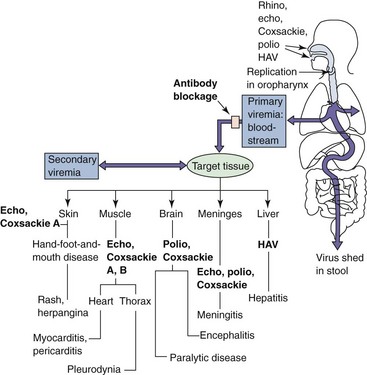
Figure 54-4 Pathogenesis of enterovirus infection. The target tissue infected by the enterovirus determines the predominant disease caused by the virus. Coxsackie, Coxsackievirus; echo, echovirus; HAV, hepatitis A virus; polio, poliovirus; rhino, rhinovirus.
Box 54-3
Disease Mechanisms of Picornaviruses
Enteroviruses enter via the oropharynx, intestinal mucosa, or upper respiratory tract and infect the underlying lymphatic tissue; rhinoviruses are restricted to the upper respiratory tract.
In the absence of serum antibody, enterovirus spreads by viremia to cells of a receptor-bearing target tissue.
Different picornaviruses bind to different receptors, many of which are members of the immunoglobulin superfamily (i.e., intercellular adhesion molecule-1).
The infected target tissue determines the subsequent disease.
Viral, rather than immune, pathologic effects are usually responsible for causing disease.
The secretory antibody response is transitory but can prevent the initiation of infection.
Serum antibody blocks viremic spread to target tissue, preventing disease.
Enterovirus is shed in feces for long periods.
Infection is often asymptomatic or causes mild, flulike, or upper respiratory tract disease.
Most enteroviruses are cytolytic, replicating rapidly and causing direct damage to the target cell. The hepatitis A virus is the exception because it is not very cytolytic. The kinetics of the immune response to hepatitis A correlate with the appearance of symptoms, indicating immunopathogenesis.
In the case of poliovirus, the virus gains access to the brain by infecting skeletal muscle and traveling up the innervating nerves to the brain, similar to the rabies virus (see Chapter 58). The virus is cytolytic for the motor neurons of the anterior horn and brainstem. The location and number of nerve cells destroyed by the virus govern the extent of paralysis and whether and when other neurons can reenervate the muscle and restore activity. The combined loss of neurons to polio and to old age may result in paralysis later in life, termed postpolio syndrome.
Viral shedding from the oropharynx can be detected for a short time before symptoms begin, whereas viral production and shedding from the intestine may last for 30 days or longer, even in the presence of a humoral immune response.
Antibody is the major protective immune response to the enteroviruses. Secretory antibody can prevent the initial establishment of infection in the oropharynx and gastrointestinal tract, and serum antibody prevents viremic spread to the target tissue and therefore disease. The time course for antibody development after infection with a live vaccine is presented in Figure 54-10 (see later).
Cell-mediated immunity is not usually involved in protection but may play a role in resolution and pathogenesis. Hepatitis A virus is the exception, in that T cells are important for the resolution of the disease and are a major determinate of the pathogenesis.
Epidemiology
The enteroviruses are exclusively human pathogens (Box 54-4). As the name implies, these viruses primarily spread via the fecal-oral route. Asymptomatic shedding can occur for up to a month, putting virus into the environment. Poor sanitation and crowded living conditions foster transmission of the viruses (Figure 54-5). Sewage contamination of water supplies can result in enterovirus epidemics. Outbreaks of enterovirus disease are seen in schools and day-care settings, and summer is the major season for such disease. The Coxsackieviruses and echoviruses may also be spread in aerosol droplets and cause respiratory tract infections.
Box 54-4
Epidemiology of Enterovirus Infections
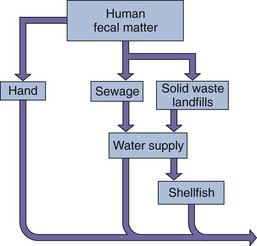
Figure 54-5 Transmission of enteroviruses. The capsid structure is resistant to mild sewage treatment, salt water, detergents, and temperature changes, allowing these viruses to be transmitted by fecal-oral routes, fomites, and on hands.
With the success of the polio vaccines, the wild-type poliovirus has been eliminated from the Western Hemisphere (Figure 54-6) and most, but not all, of the world. Paralytic polio is still prevalent in Nigeria, Afghanistan, and Pakistan. Polio may spread from these regions to areas where the vaccine is not available and to communities where vaccination is contrary to religious beliefs or other teachings. A small but significant number of vaccine-related cases of polio result from mutation of one of the three strains in the live vaccine virus, which reestablishes neurovirulence. This development has prompted a change to promote the use of the inactivated polio vaccine. Polioviruses are spread most often during the summer and autumn.
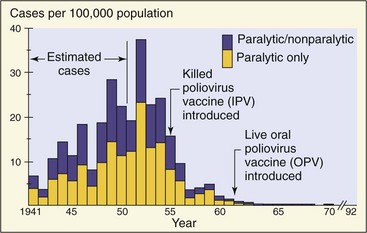
Figure 54-6 Incidence of polio in the United States. Killed (inactivated) polio vaccine (IPV) was introduced in 1955, and live (oral) polio vaccine (OPV) was introduced in 1961 and 1962. Wild-type polio has been eradicated in the United States.
(Courtesy Centers for Disease Control and Prevention: Immunization against disease: 1972, Washington, DC, 1973, U.S. Government Printing Office.)
Paralytic polio was once considered a middle class disease because good hygiene would delay exposure of a person to the virus until late childhood, the adolescent years, or adulthood, when infection would produce the most severe symptoms. Infection during early childhood generally results in asymptomatic or very mild disease.
Similar to poliovirus infection, Coxsackie A virus disease is generally more severe in adults than in children. However, Coxsackie B virus and some of the echoviruses (especially echovirus 11) can be particularly harmful to infants.
Clinical Syndromes
The clinical syndromes produced by the enteroviruses are determined by several factors, including (1) viral serotype; (2) infecting dose; (3) tissue tropism; (4) portal of entry; (5) patient’s age, gender, and state of health; and (6) pregnancy (Table 54-1). The incubation period for enterovirus disease varies from 1 to 35 days, depending on the virus, the target tissue, and the person’s age. Viruses that affect oral and respiratory sites have the shortest incubation periods.
Poliovirus Infections
There are three poliovirus types, with 85% of the cases of paralytic polio caused by type 1. Reversion of the attenuated vaccine virus types 2 and 3 to virulence can cause vaccine-associated disease. Wild-type polio infections are rare because of the success of the polio vaccines (see Figure 54-6). As noted earlier, however, vaccine-associated cases of polio do occur, and some populations remain unvaccinated, putting them at risk for infection. Poliovirus may cause one of the following four outcomes in unvaccinated people, depending on the progression of the infection (Figure 54-7):
1. Asymptomatic illness results if the viral infection is limited to the oropharynx and the gut. At least 90% of poliovirus infections are asymptomatic.
2. Abortive poliomyelitis, the minor illness, is a nonspecific febrile illness occurring in approximately 5% of infected people. Fever, headache, malaise, sore throat, and vomiting occur in such persons within 3 to 4 days of exposure.
3. Nonparalytic poliomyelitis or aseptic meningitis occurs in 1% to 2% of patients with poliovirus infections. In this disease, the virus progresses into the central nervous system and the meninges, causing back pain and muscle spasms in addition to the symptoms of the minor illness.
4. Paralytic polio, the major illness, occurs in 0.1% to 2.0% of persons with poliovirus infections and is the most severe outcome. It appears 3 to 4 days after the minor illness has subsided, thereby producing a biphasic illness. In this disease, the virus spreads from the blood to the anterior horn cells of the spinal cord and to the motor cortex of the brain. The severity of paralysis is determined by the extent of the neuronal infection and by which neurons are affected. Spinal paralysis may involve one or more limbs, whereas bulbar (cranial) paralysis may involve a combination of cranial nerves and even the medullary respiratory center.
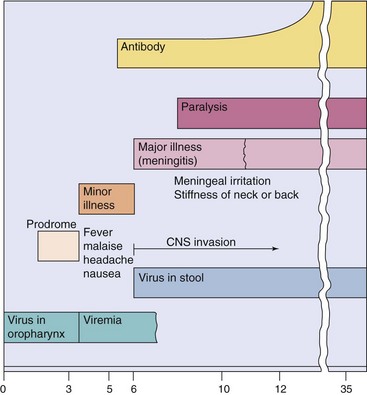
Figure 54-7 Progression of poliovirus infection. Infection may be asymptomatic or may progress to minor or major disease. CNS, Central nervous system.
Paralytic poliomyelitis is characterized by an asymmetric flaccid paralysis with no sensory loss. The degree of paralysis varies, in that it may involve only a few muscle groups (e.g., one leg) or there may be complete flaccid paralysis of all four extremities. The paralysis may then progress over the first few days and may result in complete recovery, residual paralysis, or death. Most recoveries occur within 6 months, but as long as 2 years may be required for complete remission.
Bulbar poliomyelitis can be more severe, may involve the muscles of the pharynx, vocal cords, and respiration, and may result in death in 75% of patients. Iron lungs, chambers that provided external respiratory compression, were used during the 1950s to assist the breathing of patients with such polio disease. Before vaccination programs, iron lungs filled the wards of children’s hospitals.
Postpolio syndrome is a sequela of poliomyelitis that may occur much later in life (30 to 40 years later) in 20% to 80% of the original victims. Affected persons suffer a deterioration of the originally affected muscles. Poliovirus is not present, but the syndrome is believed to result from a loss of neurons in the initially affected nerves.
Coxsackievirus and Echovirus Infections
Several clinical syndromes may be caused by either a Coxsackievirus or an echovirus (e.g., aseptic meningitis), but certain illnesses are specifically associated with Coxsackieviruses. Coxsackie A viruses are associated with diseases involving vesicular lesions (e.g., herpangina), whereas Coxsackie B viruses (B for body) are most frequently associated with myocarditis and pleurodynia. The Coxsackieviruses can also cause a polio-like paralytic disease (Clinical Case 54-1). The most common result of infection is lack of symptoms or a mild upper respiratory tract or flulike disease.
Clinical Case 54-1
Polio-like Disease from Coxsackie A Virus
In a case reported by Yoshimura and Kurashige (Brain Dev 20:540–542, 1998), a 4-year-old child’s onset of abdominal pain, distended abdomen, inability to urinate, and inability to walk prompted admission to the hospital. All abdominal reflexes were gone, accompanied by bladder and rectal dysfunction. Pain and temperature sense was normal. Cerebrospinal fluid (CSF) showed an increase in cell count, with 393 cells/mm3 and with 95% neutrophils and 5% lymphocytes. CSF protein and glucose were within normal values. Serologic analysis was negative for poliovirus and echovirus (ECHO) and Coxsackievirus types (A4, A7, A9, B1, B5), viruses reported to cause polio-like paralytic disease. Antibody for Coxsackie A10 virus was detected during the acute phase (titer = 32) and after 4 weeks (titer = 128). Three weeks after admission, he was able to walk again, but mild dysfunction of the bladder and rectum remained, even 3 months after admission. Even when routine immunization for polio has eliminated natural disease in most parts of the world, polio-like disease can still be caused by other picornaviruses and revertants of the vaccine-related strains of polio.
Herpangina is caused by several types of Coxsackie A virus and is not related to a herpesvirus infection. Fever, sore throat, pain on swallowing, anorexia, and vomiting characterize this disease. The classic finding is vesicular ulcerated lesions around the soft palate and uvula (Figure 54-8). Less typically, the lesions affect the hard palate. The virus can be recovered from the lesions or from feces. The disease is self-limited and requires only symptomatic management.
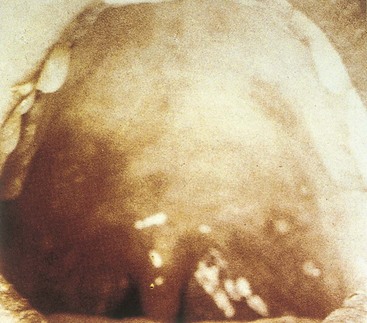
Figure 54-8 Herpangina. Characteristic discrete vesicles are seen on the anterior tonsillar pillars.
(Courtesy Dr. GDW McKendrick; from Lambert HP, et al: Infectious diseases illustrated, London, 1982, Gower.)
Hand-foot-and-mouth disease is a vesicular exanthem usually caused by Coxsackievirus A16. The name is descriptive because the main features of this infection consist of vesicular lesions on the hands, feet, mouth, and tongue (Figure 54-9). The patient is mildly febrile, and the illness subsides in a few days.
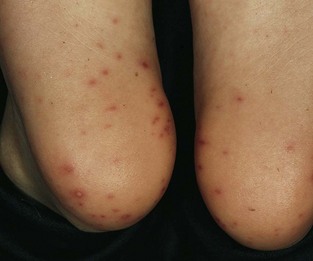
Figure 54-9 Hand-foot-and-mouth disease caused by Coxsackie A virus. Lesions initially appear in the oral cavity and then develop within 1 day on the palms and, as seen here, on soles.
(From Habif TP: Clinical dermatology: a color guide to diagnosis and therapy, ed 3, St Louis, 1996, Mosby.)
Pleurodynia (Bornholm disease), also known as the devil’s grip, is an acute illness in which patients have a sudden onset of fever and unilateral low thoracic, pleuritic chest pain that may be excruciating. Abdominal pain and even vomiting may also occur, and muscles on the involved side may be extremely tender. Pleurodynia lasts an average of 4 days but may relapse after the condition has been asymptomatic for several days. Coxsackie B virus is the causative agent.
Myocardial and pericardial infections caused by Coxsackie B virus occur sporadically in older children and adults but are most threatening in newborns. Neonates with these infections have febrile illnesses and sudden and unexplained onset of heart failure. Cyanosis, tachycardia, cardiomegaly, and hepatomegaly occur. Electrocardiographic changes are found in patients with myocarditis. The mortality associated with the infection is high, and autopsy typically reveals the involvement of other organ systems, including the brain, liver, and pancreas. Acute benign pericarditis affects young adults but may be seen in older persons. The symptoms resemble those of myocardial infarction with fever.
Viral (aseptic) meningitis is an acute febrile illness accompanied by headache and signs of meningeal irritation, including nuchal rigidity. Petechiae or a rash may occur in patients with enteroviral meningitis. Recovery is usually uneventful, unless the illness is associated with encephalitis (meningoencephalitis) or occurs in children younger than 1 year. Outbreaks of picornavirus meningitis (echovirus 11) occur each year during the summer and autumn.
Fever, rash, and common coldlike symptoms may occur in patients infected with echoviruses or Coxsackieviruses. The rash is usually maculopapular but may occasionally be petechial or even vesicular. The petechial type of eruption must be differentiated from that of meningococcemia. Enteroviral disease is usually less intense for the child than meningococcemia. Coxsackieviruses A21 and A24 and echoviruses 11 and 20 can cause rhinovirus-like symptoms resembling the common cold.
Other Enterovirus Diseases
Enterovirus 70 and a variant of Coxsackievirus A24 have been associated with an extremely contagious ocular disease, acute hemorrhagic conjunctivitis. The infection causes subconjunctival hemorrhages and conjunctivitis. The disease has a 24-hour incubation period and resolves within 1 or 2 weeks. Some strains of Coxsackie B virus and echovirus can be transmitted transplacentally to the fetus. Infection of the fetus or an infant by this or another route may produce severe disseminated disease. Coxsackie B virus infections of the beta cells of the pancreas can cause insulin-dependent diabetes as a result of the destruction of the islets of Langerhans.
Laboratory Diagnosis
Clinical Chemistry
Cerebrospinal fluid (CSF) from enterovirus aseptic meningitis can be distinguished from bacterial meningitis. The CSF lacks neutrophils, and the glucose level is usually normal or slightly low. The CSF protein level is normal to slightly elevated. The CSF is rarely positive for the virus.
Culture
Polioviruses may be isolated from the patient’s pharynx during the first few days of illness, from the feces for as long as 30 days, but only rarely from CSF. The virus grows well in monkey kidney tissue culture. Coxsackieviruses and echoviruses can usually be isolated from the throat and stool during infection and often from CSF in patients with meningitis. Virus is rarely isolated in patients with myocarditis because the symptoms occur several weeks after the initial infection. The Coxsackie B viruses can be grown on primary monkey or human embryo kidney cells. Many strains of Coxsackie A virus do not grow in tissue culture but can be grown in suckling mice.
Genome and Serology Studies
The exact type of enterovirus can be determined through the use of specific antibody and antigen assays (e.g., neutralization, immunofluorescence, enzyme-linked immunosorbent assay) or reverse transcriptase polymerase chain reaction (RT-PCR) detection of viral RNA. RT-PCR of clinical samples has become a rapid and routine method to detect the presence of an enterovirus or distinguish a specific enterovirus, depending upon the primers that are used. RT-PCR has become especially important for confirming a diagnosis of echovirus 11 meningitis in an infant.
Serology can be used to confirm an enterovirus infection through detection of specific immunoglobulin M (IgM) or the finding of a fourfold increase in the antibody titer between the time of the acute illness and the period of convalescence. Because of their many serotypes, this approach may not be practical for detection of echovirus and Coxsackievirus unless a specific virus is suspected.
Treatment, Prevention, and Control
A new antiviral drug, pleconaril, is available on a limited basis. The drug inhibits the penetration of picornaviruses into the cell. It must be administered early in the course of the infection.
The prevention of paralytic poliomyelitis is one of the triumphs of modern medicine. By 1979, infections with the wild-type poliovirus disappeared from the United States, with the number of cases of polio decreasing from 21,000 per year in the prevaccine era to 18 in unvaccinated patients in 1977. Similar to smallpox, polio has been targeted for elimination. Health care delivery to underdeveloped countries is more difficult, and for this reason, wild-type viral disease still exists in Africa, the Middle East, and Asia. Misinformation, misunderstanding, and political unrest in Africa and other parts of the world have also limited acceptance of polio vaccination. New worldwide vaccination programs have been developed to reach the goal.
The two types of poliovirus vaccine are (1) inactivated polio vaccine (IPV), developed by Jonas Salk, and (2) live attenuated oral polio vaccine (OPV), developed by Albert Sabin. Both vaccines incorporate the three strains of polio, are stable, are relatively inexpensive, and induce a protective antibody response (Figure 54-10). The IPV was proven effective in 1955, but the oral vaccine took its place because it is less expensive, easy to administer, and elicits lifelong and mucosal immunity (Table 54-2).
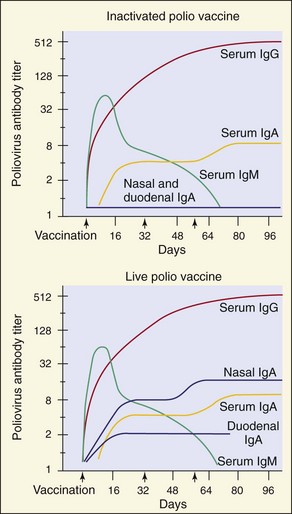
Figure 54-10 Serum and secretory antibody response to intramuscular inoculation of inactivated polio vaccine and to live attenuated oral polio vaccine. Note the presence of secretory IgA induced by the live polio vaccine.
(Modified from Ogra P, Fishaut M, Gallagher MR: Viral vaccination via the mucosal routes, Rev Infect Dis 2:352–369, 1980. Copyright 1980, University of Chicago Press.)
Table 54-2 Advantages and Disadvantages of Polio Vaccines
| Vaccine | Advantages | Disadvantages |
|---|---|---|
| Live (oral polio vaccine) | ||
| Inactivated polio vaccine |
The OPV was attenuated (i.e., rendered less virulent) by passage in human or monkey cell cultures. Attenuation yielded a virus that can replicate in the oropharynx and intestinal tract but cannot infect neuronal cells. A mixed blessing of the live vaccine strain is that it is shed in feces for weeks and may be spread to close contacts. The spread will immunize or reimmunize close contacts, thus promoting mass immunization. The major drawbacks of the live vaccine are that (1) the vaccine virus may infect an immunologically compromised person, and (2) there is a remote potential for the virus to revert to its virulent form and cause paralytic disease. The incidence of paralytic disease is estimated to be 1 per 4 million doses administered (versus 1 in 100 persons infected with the wild-type poliovirus).
In the absence of wild-type poliovirus, new recommendations call for the use of the IPV for routine vaccination. Children should receive the IPV at 2 months, 4 months, and 15 months and then at 4 to 6 years of age.
There are no vaccines for Coxsackieviruses or echoviruses. Transmission of these viruses can presumably be reduced by improvements in hygiene and living conditions. Enteroviruses are impervious to most common disinfectants and detergents but can be inactivated by formaldehyde, hypochlorite, and chlorine.
Rhinoviruses
Rhinoviruses are the most important cause of the common cold and upper respiratory tract infections. Such infections are self-limited, however, and do not cause serious disease. More than 100 serotypes of rhinovirus have been identified. At least 80% of the rhinoviruses have a common receptor that is also used by some of the Coxsackieviruses. This receptor has been identified as ICAM-1, a member of the immunoglobulin superfamily, which is expressed on epithelial, fibroblast, and B-lymphoblastoid cells.
Pathogenesis and Immunity
Unlike the enteroviruses, rhinoviruses are unable to replicate in the gastrointestinal tract (see Box 54-3). The rhinoviruses are labile to acidic pH. Also, they grow best at 33° C, a feature that contributes to their preference for the cooler environment of the nasal mucosa. Infection can be initiated by as little as one infectious viral particle. During the peak of illness, nasal secretions contain concentrations of 500 to 1000 infectious virions per milliliter. The virus enters through the nose, mouth, or eyes and initiates infection of the upper respiratory tract, including the throat. Most viral replication occurs in the nose, and the onset and the severity of the symptoms correlate with the time of viral shedding and the quantity (titer) of virus shed. Infected cells release bradykinin and histamine, which cause a “runny nose.”
Interferon, which is generated in response to the infection, may limit the progression of the infection and contribute to the symptoms. Interestingly, the release of cytokines during inflammation can promote the spread of the virus by enhancing the expression of ICAM-1 viral receptors.
Immunity to rhinoviruses is transient and unlikely to prevent subsequent infection because of the numerous serotypes of the virus. Both nasal secretory IgA and serum IgG antibody are induced by a primary rhinovirus infection and can be detected within a week of infection. The secretory IgA response dissipates quickly, and immunity begins to wane approximately 18 months after infection. Cell-mediated immunity is not likely to play an important role in controlling rhinovirus infections.
Epidemiology
Rhinoviruses cause at least half of all upper respiratory tract infections (Box 54-5). Other agents likely to cause the symptoms of the common cold are enteroviruses, coronaviruses, adenoviruses, and parainfluenza viruses. Rhinoviruses can be transmitted by two mechanisms: as aerosols and on fomites (e.g., by hands or on contaminated inanimate objects). Hands appear to be the major vector, and direct person-to-person contact is the predominant mode of spread. These nonenveloped viruses are extremely stable and can survive on such objects for many hours.
Rhinoviruses produce clinical illness in only half of the persons infected. Asymptomatic persons are also capable of spreading the virus, even though they may produce less of it.
Rhinovirus “colds” occur most often in early autumn and late spring in persons living in temperate climates. This may reflect social patterns (e.g., return to school and day care) rather than any change in the virus itself.
Rates of infection are highest in infants and children. Children younger than 2 years “share” their colds with their families. Secondary infections occur in approximately 50% of family members, especially other children.
Many different rhinovirus serotypes may be found in a given community during a specific cold season, but the predominant strains are usually the newly categorized serotypes. This pattern indicates the existence of a gradual antigenic drift (mutation) similar to that seen for the influenza virus.
Clinical Syndromes (Box 54-6)
Common cold symptoms caused by rhinoviruses cannot readily be distinguished from those caused by other viral respiratory pathogens (e.g., enteroviruses, paramyxoviruses, coronaviruses). An upper respiratory tract infection usually begins with sneezing, which is soon followed by rhinorrhea (runny nose). The rhinorrhea increases and is then accompanied by symptoms of nasal obstruction. Mild sore throat also occurs, along with headache and malaise but usually without fever. The illness peaks in 3 to 4 days, but the cough and nasal symptoms may persist for 7 to 10 days or longer.
Box 54-6
Clinical Summaries
Polio: A 12-year-old girl from Nigeria has headache, fever, nausea, and stiff neck. Symptoms improve and then recur several days later, with weakness and paralysis of her legs. She has no history of polio immunization.
Laboratory Diagnosis
The clinical syndrome of the common cold is usually so characteristic that laboratory diagnosis is unnecessary. Virus can be obtained from nasal washings. Rhinoviruses are grown in human diploid fibroblast cells (e.g., WI-38) at 33° C. Virus is identified by the typical cytopathologic effect and the demonstration of acid lability. Serotyping is rarely necessary but can be performed with the use of pools of specific neutralizing sera or by genome analysis by RT-PCR. The performance of serologic testing to document rhinovirus infection is not practical.
Treatment, Prevention, and Control
There are many over-the-counter remedies for the common cold. Nasal vasoconstrictors may provide relief, but their use may be followed by rebound congestion and a worsening of symptoms. Inhaling hot, humidified air, and even the steam from hot chicken soup, may actually help by increasing nasal drainage.
No antiviral drugs are effective. Pleconaril and similar experimental antiviral drugs (e.g., arildone, rhodanine, disoxaril) contain a 3-methylisoxazole group that inserts into the base of the receptor-binding canyon and blocks uncoating of the virus. Enviroxime inhibits the viral RNA-dependent RNA polymerase. A polypeptide receptor analogue based on the ICAM-1 protein structure has also been evaluated as an antiviral drug. The intranasal administration of interferon can block infection for a short time after a known exposure, but its long-term use (e.g., throughout the “cold season”) could cause flulike symptoms that are at least as bad as those of the rhinovirus infection.
Rhinovirus is not a good candidate for a vaccine program. The multiple serotypes, the apparent antigenic drift in rhinoviral antigens, the requirement for secretory IgA production, and the transience of the antibody response pose major problems for the development of vaccines. In addition, the benefit-to-risk ratio would be very low because rhinoviruses do not cause significant disease.
Handwashing and the disinfection of contaminated objects are the best means of preventing the spread of the virus. Virucidal facial tissues impregnated with citric acid may also limit rhinovirus spread.
A 6-year-old girl was brought to the doctor’s office at 4:30 PM because she had a sore throat, had been unusually tired, and was napping excessively. Her temperature was 39° C. She had a sore throat, enlarged tonsils, and a faint rash on her back. At 10:30 PM, the patient’s mother reported that the child had vomited three times, continued to nap excessively, and complained of a headache when awake. The doctor examined the child at 11:30 PM and noted that she was lethargic and aroused only when her head was turned, complaining that her back hurt. Her CSF contained no red blood cells, but there were 28 white blood cells/mm3—half polymorphonuclear neutrophils and half lymphocytes. The glucose and protein levels in the CSF were normal, and Gram stain of a specimen of CSF showed no bacteria.
1. What were the key signs and symptoms in this case?
2. What was the differential diagnosis?
3. What signs and symptoms suggested an enterovirus infection?
4. How would the diagnosis be confirmed?
5. What were the most likely sources and means of infection?
6. What were the target tissue and mechanism of pathogenesis?
1. The key signs and symptoms were sore throat, fever, faint rash, excessive napping, lethargy, headache, and pain upon turning head (stiff neck). The presence of lymphocytes in the CSF and normal glucose and protein levels exclude the diagnosis of a bacterial infection.
2. The differential diagnosis is aseptic meningitis that is likely caused by a virus such as an enterovirus or HSV or by an arboencephalitis virus from the Togaviridae, Flaviviridae, or Bunyaviridae families. Cryptococcus neoformans (fungus), Mycobacterium tuberculosis, and Borrelia burgdorferi are also possible. However, the presence of a rash and sore throat before signs of meningitis strengthen the likelihood of an enterovirus infection, such as Coxsackie A virus or echovirus. At an earlier time (30 years ago), polio would also be in the differential diagnosis.
3. The rash and sore throat in the prodrome period and the presence of neutrophils and lymphocytes in the CSF distinguish an enterovirus meningitis from other microbial causes.
4. An RT-PCR analysis would identify the enterovirus in the CSF and confirm the diagnosis.
5. Enteroviruses are spread by the fecal-oral route.
6. The initial target tissues for enteroviruses are the mucoepithelium, lymphoid tissue of the tonsils and pharynx, and Peyer patches of the intestinal mucosa. The virus is cytolytic.