The visual system
Anatomy
Eyelids, conjunctiva and lacrimal system
The eyelids protect the eye from injury and excessive light, distribute tears and contribute to facial expression. Levator palpebrae superioris muscle (oculomotor (III) nerve), and Müller’s muscle (sympathetic autonomic system) open the lid. Active lid closure is mediated by the orbicularis oculi muscle (facial (VII) nerve).
Secretions from three glands contribute to a healthy tear film: mucin from goblet cells in the conjunctiva, aqueous tears from the lacrimal gland in the superotemporal orbit and accessory lacrimal glands in the conjunctiva and oil from meibomian glands in the eyelids.
The conjunctiva is a thin mucous membrane lining the eyelids and reflected at the superior and inferior fornices on to the surface of the eye. Basal tear production carries nutrients and immune system proteins to the cornea. Tears wash away foreign bodies and express emotion in crying. The lacrimal ducts drain into the superior fornix. Tears drain through the lacrimal canaliculi via the puncta at the medial edge of each eyelid, then into the lacrimal sac in the anterior part of the medial wall of the orbit, and from there into the nasolacrimal duct which opens into the nasal inferior meatus.
The eye
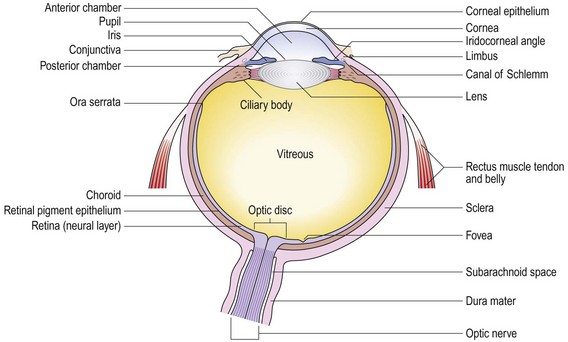
The eye focuses an image on to the neurosensory retina. The globe comprises three layers:
• an outer fibrous layer, five-sixths of which is the sclera and one-sixth the cornea. The regular orientation of collagen fibres renders the cornea transparent and it has two-thirds of the focusing power of the eye. The cornea is continuous posteriorly with the sclera, the tough, opaque white outer wall of the eyeball
• a middle vascular pigmented layer, the uveal tract, comprising the choroid posteriorly and the iris/ciliary body complex anteriorly
• an inner neurosensory layer, the retina (Fig. 12.1).
Key structures within the globe
The anterior chamber lies between the cornea and the iris and is filled with aqueous humour produced by the ciliary body in the posterior chamber. This flows through the pupil before draining through the trabecular meshwork of the angle of the anterior chamber.
The lens is a transparent, biconvex structure behind the iris and in front of the vitreous. It is suspended by the suspensory ligaments of the ciliary body.
The vitreous is a transparent gel behind the lens. Posteriorly, it is firmly attached to the margins of the optic disc, and anteriorly, to the retina at the ora serrata.
Sensation from the cornea, conjunctiva and intraocular structures is conveyed by the ophthalmic branch (V1) of the trigeminal nerve (Ch. 11).
The optic (II) cranial nerve
The visual pathway consists of the retina, optic chiasm, optic tracts, lateral geniculate bodies, optic radiations and visual cortex.
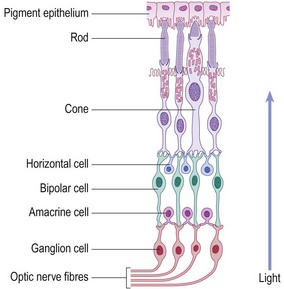
The retina consists of an outer pigmented layer, and an inner neurosensory layer which is continuous with the optic nerve. The retinal pigment epithelium lies adjacent to the highly vascular choroid. Around 90% of ocular blood supply passes through the choroid, supplying the posterior two-thirds of the retina, the optic nerve and the fovea. The retinal vessels supply the relatively inert inner one-third of the retina. The neurosensory retina consists of photoreceptors, ganglion cells and interconnecting bipolar cells.
• Rod photoreceptors are responsible for night vision and detection of peripheral movement.
• Cone photoreceptors are responsible for colour and central vision.
• Photoreceptors synapse with the vertically oriented bipolar cells of the retina, which in turn synapse with the ganglion cells of the optic nerve (Fig. 12.2).
The optic nerve is purely sensory and cannot regenerate. The optic nerve fibres myelinate on leaving the eye through the optic disc and enter the cranium through the optic canal. The two optic nerves join at the optic chiasm, where the nasal fibres, responsible for the temporal visual field, decussate. Leaving the chiasm, the visual pathway is renamed the ‘optic tract’. The optic tracts terminate in the lateral geniculate bodies of the thalamus. However, some fibres leave the tract to form the afferent limb of the pupillary reflex.
Optic radiations pass through the cerebral hemisphere in the posterior part of the internal capsule and the parietal and temporal lobes to terminate in the occipital cortex (Fig. 12.3).
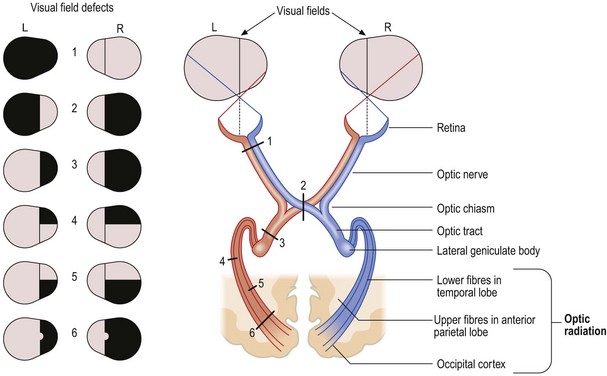
Fig. 12.3 Visual field defects.
(1) Total loss of vision in one eye because of a lesion of the optic nerve. (2) Bitemporal hemianopia due to compression of the optic chiasm. (3) Right homonymous hemianopia from a lesion of the optic tract. (4) Upper right quadrantanopia from a lesion of the lower fibres of the optic radiation in the temporal lobe. (5) Lower quadrantanopia from a lesion of the upper fibres of the optic radiation in the anterior part of the parietal lobe. (6) Right homonymous hemianopia with sparing of the macula due to lesion of the optic radiation in the occipital lobe.
The occipital lobe analyses visual information and damage produces a homonymous hemianopia or scotoma (a discrete visual defect). The occipital lobe has a dual blood supply from the posterior and middle cerebral arteries. The middle cerebral artery supplies the posterior tip of the occipital lobe responsible for central vision from the macula. A lesion, affecting only the posterior tip of the occipital lobe, may produce central homonymous hemianopia with sparing of peripheral vision. A lesion, affecting only the anterior occipital lobe, may cause homonymous hemianopia involving peripheral vision but sparing central macular vision. Damage to secondary visual areas causes visual agnosia (inability to recognise visual stimuli) and distorted perceptions of visual images, such as macropsia (seeing things larger) or micropsia (smaller than reality) and visual hallucinations.
The oculomotor (III), trochlear (IV) and abducens (VI) cranial nerves innervate the six external ocular muscles controlling eye movement and, through parasympathetic nerves, also affect pupillary size (Fig. 12.4).
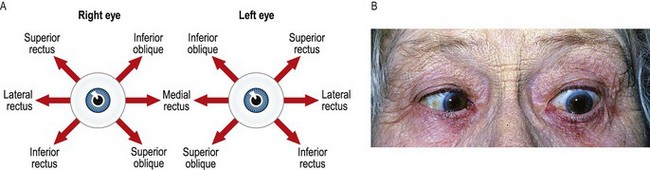
Fig. 12.4 Control of eye movement.
(A) Fields of action of pairs of extraocular muscles. This diagram will help you to work out which eye muscle is paretic. For example, a patient whose diplopia is maximum on looking down and to the right has either an impaired right inferior rectus or a weak left superior oblique. (B) Left sixth-nerve palsy causing weakness of the lateral rectus muscle. The patient is shown here attempting to look to the left.
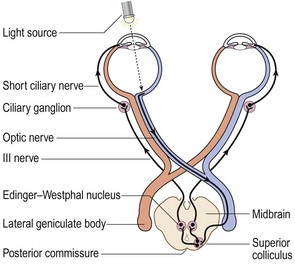
The oculomotor (III) nerve passes just below the free edge of the tentorium in relation to the posterior communicating artery and enters the dura surrounding the cavernous sinus. It enters the orbit through the superior oblique fissure, and innervates the superior, medial and inferior recti, the inferior oblique and levator palpebrae superioris muscles. These muscles open the upper lid (levator palpebrae superioris) and move the globe upwards (superior rectus, inferior oblique), downwards (inferior rectus) and medially (medial rectus). Through the parasympathetic fibres arising from the Edinger–Westphal nerves, the nerve indirectly supplies the sphincter muscles of the iris, causing constriction of the pupil, and the ciliary muscle which focuses the lens for near vision (accommodation) (Fig. 12.5).
The trochlear (IV) nerve fibres decussate before leaving the midbrain posteriorly (the left nucleus innervates the right trochlear nerve and vice versa). The nerve enters the orbit in the superior orbital fissure, supplying the superior oblique muscle, which causes downward movement of the globe when the eye is adducted.
The abducens (VI) nerve has a long course around the brainstem before it pierces the dura to enter the cavernous sinus. The nerve is in direct relation to the internal carotid artery before it passes through the superior orbital fissure to the lateral rectus muscle. Lateral rectus abducts the eye (Fig. 12.6).
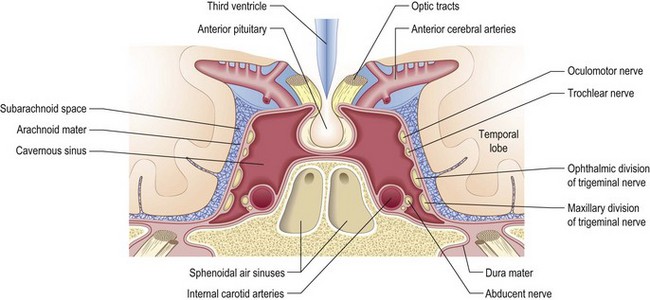
Fig. 12.6 Sagittal section illustrating neuroanatomy and lesions of the III, IV and VI cranial nerves.
The pupils admit light into the eyeball and are round, regular, equal in size and symmetrical in their responses. The autonomic nervous system and integrity of the iris determine the size of the resting pupil. The afferent limb of the pupillary reflex involves the optic nerve, chiasm (where some fibres decussate) and the optic tract, bypassing the lateral geniculate nucleus to terminate in the III nerve (Edinger–Westphal) nucleus. The efferent limb involves the inferior division of the III nerve, passing through the ciliary ganglion in the orbit to the constrictor muscle of the iris. Sympathetic stimulation causes pupillary dilatation and upper and lower eyelid retraction. With parasympathetic stimulation (the fibres travel with the III nerve), the opposite occurs.
Symptoms and definitions
Pain is common and ranges from irritation to the excruciating pain of scleritis. Note if the affected eye is red or not to make the diagnosis.
Ocular pain with a ‘white eye’
If the patient feels ‘something in the eye’, the problem involves the eye surface. The most common cause is a dry eye secondary to blepharitis (inflammation of the eyelid margins) with disruption of meibomian glandular secretions. Paradoxically, patients often complain of watery eyes due to reflex overproduction of tears from the lacrimal gland. Blepharitis may be associated with systemic skin conditions, e.g. atopic eczema, acne rosacea or seborrhoeic dermatitis. Severe dry eyes are a feature of Sjögren’s syndrome. Preceding visual disturbance associated with headache or eye pain suggests migraine. Cluster headaches may present as ocular pain. In subacute episodes of angle closure (raised intraocular pressure), patients describe seeing haloes around lights. Pain on eye movement usually indicates either optic neuritis or scleritis. The eye in optic neuritis is white; in scleritis it is red.
Ocular pain with a ‘red eye’
Circumciliary injection (redness around the corneal limbus) reflects involvement of the anterior ciliary arteries supplying the cornea, iris and ciliary body (uveitis). Diffuse redness suggests scleritis or conjunctivitis.
Entropion (an inverted eyelid) leads to painful corneal erosion (Fig. 12.7) and ectropion (everted eyelid) causes dryness in the exposed eye. Foreign bodies on the eye surface are usually associated with an at-risk activity, e.g. grinding metal without eye protection.
Severe unilateral pain with a cloudy cornea, circumciliary injection and an oval non-reactive pupil indicates acute angle closure glaucoma with high intraocular pressure. Long-sighted people with shallow anterior chambers and the elderly, in whom lens thickening makes the anterior chamber shallower, are at risk. Pupillary dilatation may precipitate acute angle closure. Corneal oedema clouds the underlying iris, which may become ischaemic and fixed in mid-dilation. The pain is severe and may be associated with systemic symptoms. Treat both eyes to prevent recurrence.
Acute iritis produces a small, irregularly shaped pupil (Fig. 12.8A) and redness around the limbus. The inflamed iris becomes stuck to the underlying lens. The pain is not as severe as in angle closure glaucoma and photophobia may be prominent. Rarely, iritis presents bilaterally.
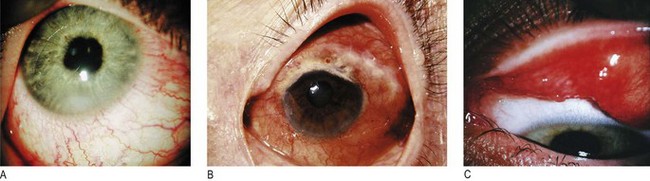
Fig. 12.8 Inflammation of the eye.
(A) Acute iritis. Irregular pupil and circumciliary injection. (B) Necrotising scleritis. Areas of pallor within diffuse areas of redness, indicating ischaemia. (C) Giant papillary conjunctivitis seen on everting upper lid. A form of allergic conjunctivitis induced by contact lens wear.
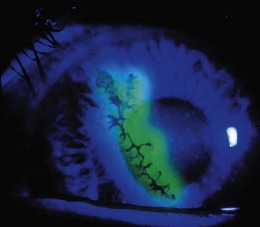
Corneal ulceration may be due to herpes simplex virus (Box 12.1), but in contact lens wearers, dry eyes or debilitated patients suspect bacterial infection. Depending on severity, redness may vary from circumciliary injection to diffuse redness associated with spontaneous lacrimation.
Pain on moving a red eye indicates scleritis (Fig. 12.8B) and may be the first manifestation of systemic vasculitis. The redness is frequently bilateral and involves the whole sclera or a sector of the sclera, unlike the circumciliary injection of iritis or angle closure glaucoma. Episcleritis is uncomfortable rather than painful and appears less dramatic than scleritis.
Conjunctivitis is an uncomfortable inflammation of the conjunctivae. There is always associated discharge or watering. The inner eyelid is inflamed and red, unlike scleritis or episcleritis (Fig. 12.8C). Bacterial conjunctivitis is associated with a yellow/green purulent discharge. Chlamydial and viral infection causes a clear discharge and preauricular lymphadenopathy. Persistent watery discharge with itchy eyes and no lymphadenopathy suggests an allergic cause.
Visual disturbance
The curvature of the optic lens alters to adapt the focal length to suit the varying distance of the entering light rays and produce a clear, focused image. Common refractive errors are:
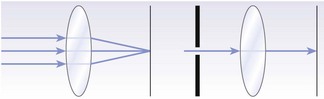
• Hypermetropia (long-sightedness): rays of light from a distant object are focused behind the retina. It is common in infants, and young people can compensate for hypermetropia by contracting the ciliary muscle, increasing the refractive power of the lens (Fig. 12.9).
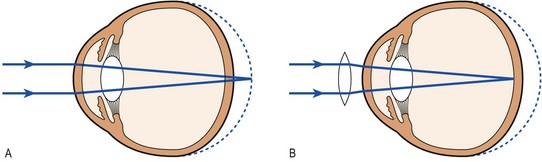
Fig. 12.9 The hypermetropic (long-sighted) eye.
(A) The eye is too short and the image on the retina is not in focus. (B) The use of a convex (plus) lens brings the image on the retina into focus.
• Myopia (short-sightedness): rays from a distant object are focused in front of the retina. Simple myopia usually starts in childhood and worsens during the growing years (Fig. 12.10).
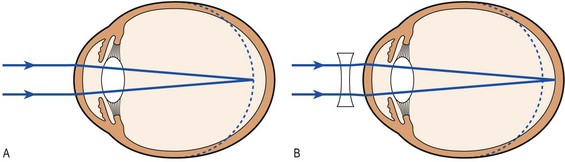
Fig. 12.10 The myopic (short-sighted) eye.
(A) The eye is too long and the image on the retina is not in focus. (B) The use of a concave (minus) lens brings the image on the retina into focus.
• Presbyopia (impaired power of accommodation for near objects) occurs as the lens ages and is less able to change its curvature. It is very common in those >45 years.
• Astigmatism is when the cornea is irregularly curved, preventing light rays being brought to a common focus on the retina.
Diplopia (double vision)
This may be uniocular or binocular. Binocular diplopia is caused by imbalance of eye movements; uniocular is due to intraocular disease.
Blurred vision
Distinguish true blurring of the whole visual field from a scotoma (a discrete defect). Generalised blurring is due to an ocular problem, ranging from uncorrected refractive error, e.g. myopia, hypermetropia, astigmatism or presbyopia, to opacities in the cornea, lens, aqueous chamber or vitreous gel.
Sudden-onset visual loss
This may be temporary or permanent, unilateral or bilateral. Patients may not notice gradual visual loss in one eye but only notice and complain when they close their unaffected eye.
Transient visual loss or disturbance is caused by the aura of migraine or unilateral amaurosis fugax (transient retinal ischaemia). The aura of classical migraine usually takes the form of coloured lines, often scintillating, and is always homonymous (present in both visual fields), although patients may mistakenly attribute it to one eye. Unlike amaurosis fugax, it is still seen with the eyes shut. In contrast, retinal migraine causes unilateral visual loss and may be difficult to differentiate from amaurosis fugax.
Amaurosis fugax produces a negative unilateral visual phenomenon which is black or grey. This is short-lasting (minutes) and appears like a shutter coming down, up or from the side, before resolving in a similar fashion. It may be confused with migraine aura or the homonymous hemianopia of transient occipital lobe ischaemia. Visual impairment following exercise or a hot bath is characteristic of demyelinating optic neuritis (Uthoff’s phenomenon). Permanent, sudden visual loss is usually due to vascular occlusion. Establish whether the symptoms are uniocular or homonymous (Box 12.2). Permanent homonymous visual loss, in the absence of hemiparesis or dysphasia, usually indicates occipital lobe infarction.
Gradual-onset visual loss
Gradual onset of visual loss is commonly caused by cataract or atrophic age-related macular degeneration. Both cause glare from bright lights. Slowly progressive visual loss, accompanied by optic atrophy, occurs when the optic nerve or chiasm is compressed by tumours within the orbit or skull base, e.g. meningioma or pituitary adenoma.
Distortion of vision
Distortion is caused by disruption of the photoreceptors at the macula, most commonly macular degeneration in the elderly. Untreated, central visual loss is rapid and irreversible. Scarring of the outer surface of the vitreous (epiretinal membrane) may pucker the normally smooth surface of the macula and follow any insult to the vitreous, including haemorrhage, inflammation or trauma.
Flashes and floaters
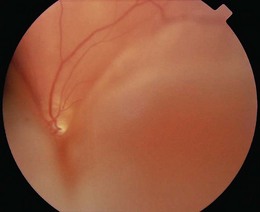
Flashes and floaters are caused by vitreous degeneration, especially in those >65 years and in myopia (short sight). As the vitreous degenerates, the gel liquefies and fluid escapes through perforations in the outer surface of the vitreous overlying the macula. The fluid peels the vitreous off the retina and the remaining contents swirl about on eye movement. The vitreous is attached to the retina in places, and as it detaches, retinal traction may produce flashes and lead to retinal tears. Retinal tears allow fluid from the vitreous cavity to enter the potential space between the retina and the retinal pigment epithelium, causing retinal detachment (Fig. 12.11). The patient notices visual loss starting peripherally and moving centrally.
The history
The time course of the presenting complaint is crucial to diagnosis. For example, vascular causes are sudden in onset, whereas slow, inexorable progression of symptoms is seen in compression of the optic pathway by a tumour. Symptoms which worsen over about 2 weeks, last 2–3 weeks and then resolve suggest demyelination. Always clarify terms used by patients; for example, diplopia and blurring are easily confused.
Past history
Information about previous ocular and systemic illnesses can help the diagnosis (Box 12.3). Old family photographs can help assess the onset of pupil abnormalities and features such as proptosis.
Family history
Ask about family history, especially in children. Genetic diseases include retinitis pigmentosa, and the incidence of multiple sclerosis is increased in those with a positive family history. Many patients with thyroid eye disease have a family history of autoimmune disease.
Social history
Cigarette smoking is the most important cause of vascular disease affecting the eye and is a major risk factor for age-related macular degeneration. A history of hayfever or allergies to animals suggests an allergic eye disorder. Recreational drug use may be associated with visual loss, particularly cocaine-induced vascular occlusions. Take a sexual history in all patients with ocular inflammation or unexplained neuro-ophthalmic symptoms as uveitis may be the first manifestation of human immunodeficiency virus (HIV) infection or neurosyphilis (Box 12.5).
The physical examination
Assess cranial nerves II, III, IV and VI and their central connections:
Inspection
Posture: Congenital and longstanding paralytic squint often causes an abnormal head posture with the head turned or tilted to minimise the diplopia.
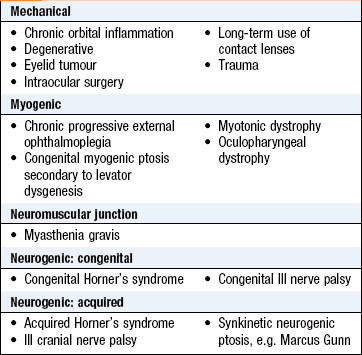
Eyelids: A narrow palpebral fissure (the gap between upper and lower eyelids) suggests ptosis (drooping eyelid) (Box 12.8) or blepharospasm (tonic spasm in the orbicularis oris muscle). Eyelid retraction or proptosis makes the sclera visible above the cornea, ectropion visible below.
• involutional: stretching of the levator muscle aponeurosis with age is the commonest cause
• III nerve palsy: causes unilateral ptosis that is often complete. The pupil is large because of loss of parasympathetic innervation of the iris. Unopposed action of IV and VI cranial nerves results in the eye looking inferolaterally. Urgently investigate an acute III nerve palsy, especially if painful, as this may indicate a posterior communicating artery aneurysm
• Horner’s syndrome (damage to sympathetic ocular innervation): paralyses Müller’s muscle and lower lid retractors, producing a partial ptosis and a pseudoenophthalmos. Miosis (small pupil) is present due to lack of sympathetic innervation to the iris
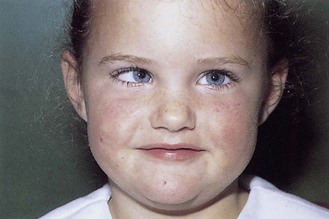
• myasthenia gravis causes bilateral variable ptosis due to fatigability of the levator muscle. The patient may be unable to ‘bury the eyelashes’ (orbicularis muscle, VII nerve innervation), which suggests a neuromuscular junction disorder, a myopathy or a meningeal disorder, as it is anatomically impossible for a single lesion to affect both III and VII ipsilateral cranial nerves (Fig. 12.15).
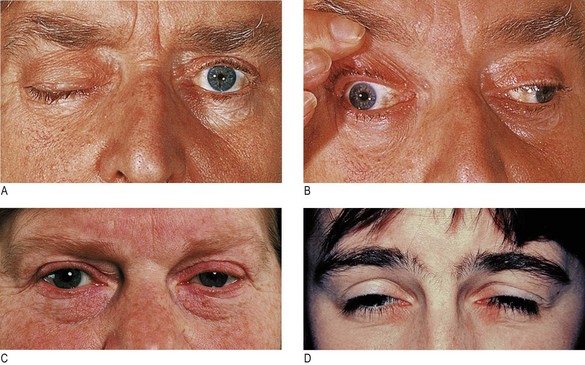
Fig. 12.15 Ptosis.
(A) Complete right ptosis in III nerve palsy. (B) The same patient looking down and to the left; the right eye has rotated medially, demonstrating that the trochlear (IV) nerve is intact. (C) Left Horner’s syndrome. (D) Myasthenia gravis. The patient is attempting to open his eyelids. Raised forehead browlines (frontalis overactivity) reflect the effort of attempting to open the eyelids.
Periorbital oedema may be associated with chemosis (conjunctival oedema).
Lacrimal apparatus: The lacrimal gland may become swollen through:
Nasolacrimal duct blockage produces watering and sticky discharge, and may cause dacrocystitis (acute inflammation of the lacrimal sac). It is common in neonates and usually resolves spontaneously.
Eyelid margin: Blepharitis (inflammation of the eyelid margin) may be associated with systemic skin involvement. Look for:
• ‘ace of clubs’ appearance of rosacea (erythema on forehead, cheeks and chin)
Common lumps on the eyelids include:
Conjunctiva: Circumciliary injection (redness around the corneal limbus) reflects involvement of the anterior ciliary arteries supplying the cornea, iris and ciliary body (uveitis). Diffuse redness suggests scleritis or conjunctivitis.
Sclera: Thin sclera is transparent, revealing the bluish choroid beneath, and occurs in scleromalacia, osteogenesis imperfecta and Ehlers–Danlos syndrome. The sclerae are yellow in jaundice (Fig. 8.8).
Scleritis causes a dark-red colour, tenderness and pain on eye movement. Systemic vasculitis causes white patches within red areas, suggesting impending necrosis.
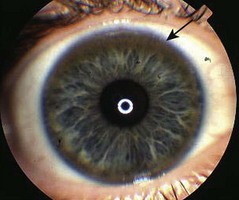
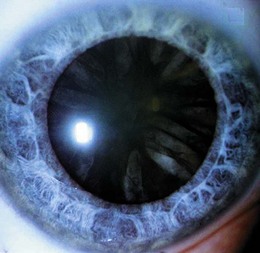
Cornea: The corneal epithelium may be affected by dryness, trauma or infection. Peripheral corneal arcus (lipid deposition) is seen with hyperlipidaemia (Fig. 6.8C). In Wilson’s disease, copper is deposited round the cornea, causing Kayser–Fleischer rings (Fig. 12.16). Calcium may be deposited in chronic ocular inflammation and chronic hypercalcaemia (band keratopathy).
Visual fields
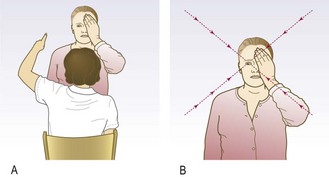
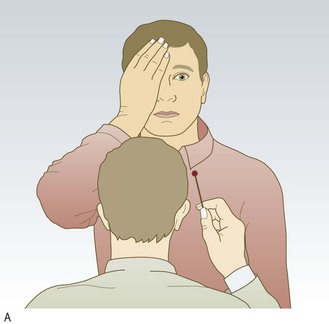
The normal visual field extends 160° horizontally and 130° vertically. Fixation is the very centre of a patient’s visual field. The physiological blind spot is located 15° temporal to the point of visual fixation and represents the optic nerve head. At the bedside, test visual fields by confrontation (Figs 12.20 and 12.21): an automated visual field analyser provides a more accurate assessment.
Abnormal findings: Intraocular causes of visual field defects include:
• Macular lesions, e.g. age-related macular degeneration, cause central scotomas, which may be incomplete and associated with distortion. Commonly, a patient with a central scotoma cannot see a face clearly but the rest of the visual field is unaffected.
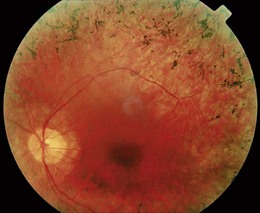
• Peripheral retinal lesions spare central vision, causing localised scotomas, e.g. retinal detachment or scarring, or constriction of peripheral field, e.g. retinitis pigmentosa.
Lesions of the optic nerve within the orbit also cause central scotomas, but, unlike those of macular origin, red desaturation (red colours appear orange or pink) occurs early and there is no visual distortion.
Unilateral optic nerve lesions cause a relative afferent pupillary defect.
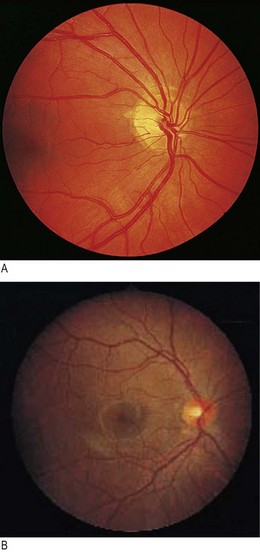
Distension of the nerve sheath around the optic nerve, e.g. papilloedema (Fig. 12.22), causes an enlarged blind spot. Optic atrophy occurs later.
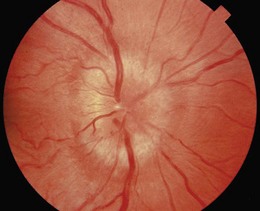
Fig. 12.22 Swollen optic disc.
Papilloedema is suggested if visual acuity is unaffected, colour vision is normal and there is an enlarged blind spot.
Lesions at the optic chiasm cause bilateral temporal visual field defects (bitemporal hemianopia; Fig. 12.3). As the optic chiasm is a continuation of the optic nerve, central red desaturation may precede peripheral visual field loss.
Optic tract lesions are uncommon; they are usually suprasellar lesions, e.g. pituitary tumours, and produce asymmetrical homonymous visual field defects.
The optic radiations and occipital cortex are more commonly affected and produce:
• symmetrical visual field defects (homonymous hemianopia)
• lack of awareness of visual field loss: this suggests parietal lobe involvement
• superior homonymous quadrantanopia: this suggests temporal lobe involvement
• visual field defects: these may affect only central vision or spare the macula because of the dual blood supply of the occipital cortex
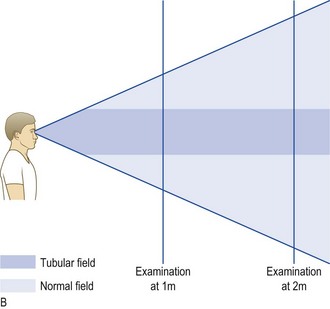
• Functional visual loss is common, particularly bilateral visual field constriction that does not expand on testing further away. This tubular constriction differentiates it from the funnel constriction of bilateral retinal disorders, such as retinitis pigmentosa (Fig. 12.23) or bilateral homonymous hemianopia (cortical visual impairment). The normal visual field is conical and, no matter how small, its diameter doubles when the patient is tested in front of a tangent screen and then moved back from 1 metre distance to 2 metres. Patients with functional tunnel vision have an unchanged or smaller visual field (Fig. 12.21B).
Ocular alignment and eye movements
The eyes are normally parallel in all positions of gaze, except near convergence. If not, a squint (strabismus) is present. Squints are associated with:
• paresis of one or more extraocular muscles (paralytic or incomitant squint)
• defective binocular vision (non-paralytic or concomitant squint).
Acquired paralytic squints cause diplopia, in which the images are maximally separated and squint is greatest in the direction of action of the paretic muscle. Concomitant squints are the same in all positions of gaze. They usually become manifest in childhood when they are not associated with diplopia (Fig. 12.24). In children, the visual acuity of the squinting eye falls, causing amblyopia (a ‘lazy’ eye).
Abnormal findings: Monocular diplopia is created by ‘ghosting’ from structural abnormality anywhere between the cornea and fovea.
Pure horizontal diplopia usually results from involvement of the VI cranial nerve. The symptoms are worse looking to the affected side.
Orbital trauma may trap a medial rectus muscle.
A demyelinating pontine lesion may be associated with ipsilateral lower motor neurone nerve palsy.
Disruption of the neuronal connection between the medial rectus and the contralateral VI nerve (median longitudinal fasciculus), e.g. in multiple sclerosis, causes underaction of the medial rectus and an internuclear ophthalmoplegia. Unlike VI nerve palsy, diplopia is worse on looking to the contralateral side.
VI nerve palsy occurs with raised intracranial pressure when the nerve is stretched as it passes upward to the cavernous sinus (Fig. 12.6).
Other causes of impaired horizontal movement include:
• pontine gaze palsies (nuclear VI nerve palsy)
• convergence spasm (impaired uniocular lateral gaze associated with bilateral miosis).
• Impaired vertical gaze can occur in:
• orbital floor fractures, which may entrap the inferior rectus and restrict upgaze.
• brainstem stroke, demyelination and children with hydrocephalus (when the inferior segment of the cornea lies below the lower lid – the ‘setting sun’ sign). IV nerve palsies cause vertical diplopia, particularly noticeable on down-gaze. The IV nerve is the only nerve to emerge from the posterior brainstem and is particularly susceptible to a blow to the occiput. Damage is often bilateral.
Thyroid eye disease is a common cause of vertical diplopia. A complaint of squint rather than diplopia suggests non-paralytic squint resulting from defective binocular vision. An amblyopic eye often diverges while monocular eye movements will be intact. The cover test is useful in children.
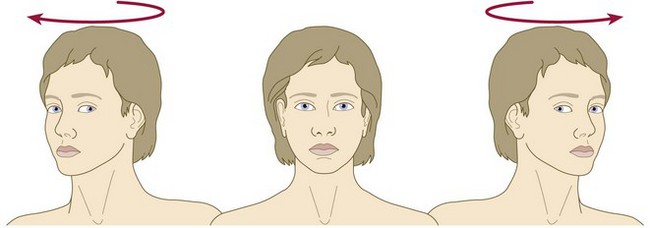
The oculocephalic test differentiates supranuclear lesions from cranial nerve lesions and is impaired in brainstem stroke, metabolic dysfunction or drug intoxication.
Nystagmus
Biphasic or jerk nystagmus is commonest where there is a slow drift in one direction, followed by a fast correction in the opposite direction. The direction of the fast phase or jerk is called the direction of nystagmus. Oscillations occurring at the same speed and over the same range about a central point are pendular nystagmus.
Normal findings: At extremes of lateral gaze, normal subjects may show a few nystagmoid jerks. When a person looks out of a train window at the passing scenery, a physiological opticokinetic nystagmus occurs. This can be tested using a vertically striped drum that is spun in front of the subject. The fast phase of the nystagmus is in the opposite direction to the drum’s spin. It can be impaired in people with visual field defects.
Abnormal findings: Peripheral vestibular nystagmus often has horizontal, vertical and rotatory components, and is usually associated with vertigo. The amplitude of the oscillation increases with gaze towards the direction of the fast phase and is suppressed by visual fixation using Fresnel lenses.
Central vestibular nystagmus is usually unidirectional, does not alter with direction of gaze or with visual fixation, and vertigo is less prominent. Common causes include multiple sclerosis and cerebrovascular disease.
Vertical nystagmus is uncommon and indicates brainstem damage. Upbeat nystagmus, with the fast phase on looking upwards, occurs with upper brainstem lesions in multiple sclerosis, infarction and Wernicke’s encephalopathy. Downbeat nystagmus may result from lesions around the craniocervical junction, e.g. Arnold–Chiari malformation or demyelination, and phenytoin or lithium intoxication. Periodic alternating nystagmus is present in the primary position but changes direction in a crescendo–decrescendo manner, often every 90 seconds with a null period of up to 10 seconds. This may be congenital, due to lesions at the craniocervical junction, or a feature of drug intoxication. Demyelination of the medial longitudinal bundle within the brainstem can cause ataxic nystagmus, where the oscillations are more marked in the abducting eye than in the adducting one. This is often associated with an internuclear ophthalmoplegia with reduced adduction.
Congenital nystagmus is usually a horizontal jerk nystagmus but can be pendular. Acquired pendular nystagmus results from cerebellar or brainstem disease, most commonly multiple sclerosis, but also from spinocerebellar degenerations and brainstem ischaemia. In functional blindness optico-kinetic nystagmus is present.
Pupils
Normal findings: Simple, or essential, anisocoria, a >0.4 mm difference between the pupil diameters, is common.
Abnormal findings: In diabetes mellitus, autonomic neuropathy may produce small pupils that respond poorly to pharmacological dilatation. They may mimic the Argyll Robertson pupils of syphilis (pinpoint, irregular pupils that constrict only on convergence).
An Adie pupil is usually mid-dilated and responds poorly to convergence. It results from ciliary ganglion malfunction within the orbit. With time it may shrink in size and be confused with an Argyll Robertson pupil. It is frequently bilateral, and when associated with absent neurological reflexes, is termed the Holmes–Adie syndrome.
Optic nerve damage results in an afferent pupillary defect (Marcus Gunn pupil). Both pupils are dually innervated at the level of the midbrain. Normally, both pupils constrict to light, regardless of which eye is illuminated. If one optic nerve is damaged, whatever lighting the dominant optic nerve is exposed to determines the size of both pupils (Fig. 12.26).
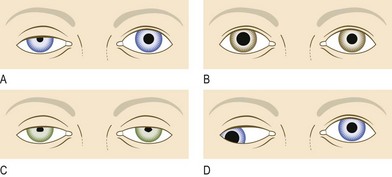
Fig. 12.26 Pupillary defects.
(A) Right Horner’s syndrome (ptosis and miosis). (B) Right Holmes–Adie pupil. (C) Argyll Robertson pupils with bilateral ptosis and small irregular pupils. (D) Right III nerve palsy (looking down and out, ptosis and a dilated pupil).
A unilateral dilated pupil in a patient with deteriorating conscious level secondary to intracranial mass lesions, e.g. tumour or enlarging haematoma, occurs when brain herniation compresses the III nerve.
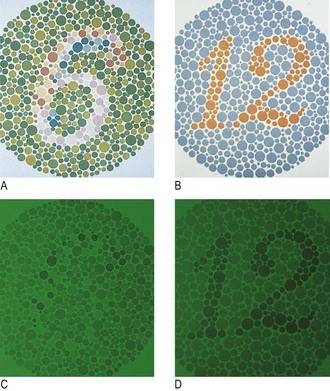
Colour vision
Abnormal findings: Red desaturation is impaired ability to identify red objects and an early indicator of optic nerve pathology.
Congenital red–green blindness is an X-linked recessive condition and affects 7% of the male population. Red–green colour vision is impaired before loss of visual acuity with optic nerve damage anywhere from the photoreceptors to the lateral geniculate nucleus of the thalamus.
Ophthalmoscopy
Examine the eye undilated first to see the pupils and iris; then, ideally, examine the eye dilated using tropicamide drops, to visualise the lens, vitreous and retina. Only the optic nerve can be reliably assessed without pupillary dilatation. If patients have particularly thick lenses, examine their eyes with their glasses in place; however, this reduces your field of view. Advise the patient not to drive or use machinery until the effect of the mydriatic has completely worn off. This may take several hours.
Cornea: Asymptomatic corneal scars from foreign bodies, often accompanied by remnants of rust, and previous ulceration are common.
Lens: There are three common forms of cataract:
• Peripheral cortical cataract is common in diabetes mellitus (Fig. 12.30). It appears as incomplete black spokes radiating from the periphery of the lens.
• Posterior subcapsular cataract, the typical ‘steroid cataract’, appears as a black opacity coming from the centre of the lens.
• Nuclear sclerosis (‘ageing cataract’) is the normally transparent lens yellowing before it becomes brown, then black. It cannot usually be detected in the red reflex, but symptoms and an inability to focus clearly on the retina confirm its presence.
Vitreous: The patient perceives vitreous haemorrhage as black blobs that move with eye movement (intra-gel vitreous haemorrhage). Abnormal vitreous adhesion to normal retinal vessels may cause vitreous haemorrhage, and ‘flashes of light’ indicate that the retina may have torn. Haemorrhage in the space between the retina and the posterior surface of the vitreous causes a subhyaloid (preretinal) vitreous haemorrhage (Fig. 12.31B).
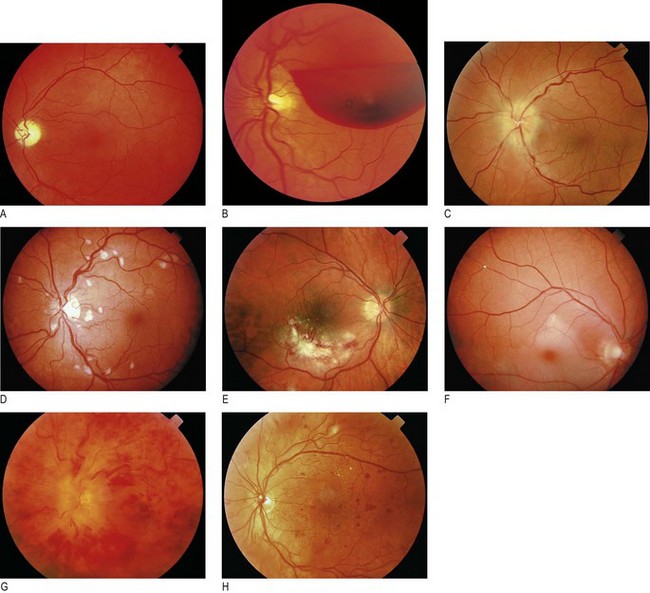
Fig. 12.31 Retinal abnormalities.
(A) Left optic atrophy. Note the lack of a pink neuroretinal rim. (B) Preretinal haemorrhage. (C) Pale white swollen disc. This is highly suggestive of giant cell arteritis, particularly if associated with visual loss. (D) Arteriolar occlusion of the horizontal nerve fibre layer. Multiple cotton-wool spots in human immunodeficiency virus (HIV) retinopathy. (E) Cytomegalovirus retinitis. Note the large superficial retinal infiltrate associated with flame haemorrhage. (F) Central retinal artery occlusion. Note the milky-white pale infarcted retina surrounding healthy pink fovea (‘cherry-red spot’). (G) Central retinal vein occlusion. Note the widespread retinal haemorrhages and swollen optic disc. (H) Diabetic retinopathy with multiple dot and blot haemorrhages, indicating widespread capillary occlusion, a precursor of new vessel formation.
Optic disc: With a swollen and white optic nerve head (anterior ischaemic optic neuropathy), consider giant cell arteritis and polyarteritis nodosa and check markers of inflammation (raised erythrocyte sedimentation rate, C-reactive protein or platelet count) (Fig. 12.31C). Pseudophakic patients with artificial intraocular lenses following cataract extraction often have falsely pale discs. Increased cup-to-disc ratio (cupped disc) is seen with chronic open-angle glaucoma (a group of diseases of the optic nerve involving loss of retinal ganglion cells, associated with raised intraocular pressure and visual field loss). Typically, the vertical margins are affected first.
The optic disc is a common site for new vessel formation. In the presence of an enlarged blind spot, blurring of the optic disc indicates distension of the optic nerve sheath. Reduced colour vision and a relative afferent pupillary response suggest an intrinsic optic nerve lesion.
Horizontal nerve fibre layer: The nerve fibre layer runs horizontally over the retinal blood vessels. Lesions within this are therefore flat and striated, and obscure retinal blood vessels.
Arteriolar occlusion (Box 12.9) causes ‘cotton-wool’ spot formation (Fig. 12.31D) and flame haemorrhages (Fig. 12.31E).
Roth’s spots are flame-shaped haemorrhages with a central cotton-wool spot. They are caused by immune complex deposition, and are seen in subacute bacterial endocarditis and serum sickness (see Fig. 6.8B).
Retinitis due to herpesvirus infection causes a large, rapidly progressive area of ‘cotton-wool’ spot formation. It can be difficult to differentiate from the cotton-wool spots of arterial occlusion and the two may coexist in HIV infection. Cotton-wool spots, however, do not enlarge over time, whereas areas of retinitis do (Box 12.10).
Retinal artery occlusion is usually embolic and causes retinal pallor because of anterior retinal layer infarction. It resembles amaurosis fugax but is permanent. The optic nerve head, the fovea and the posterior retina, including the photoreceptors, are unaffected by retinal artery occlusion, as their blood supply is from the short posterior ciliary arteries of the ophthalmic artery. This explains the cherry-red spot sign of central retinal artery occlusion, where the healthy fovea is surrounded by an oedematous retina (Fig. 12.31F). Retinal emboli may be seen at vessel bifurcations. As only the luminal contents of the vessel are normally apparent and not the wall, the embolus may appear to be paradoxically wider than the vessel it is lodged in.
Vertical bipolar layer: This contains the retinal capillaries. The commonest causes of capillary disease are diabetes mellitus and retinal vein occlusion (Fig. 12.31G). Capillary occlusion is also seen with HIV and radiation retinopathies. Microaneurysm formation may occur at the site of capillary occlusion. Capillaries are too small to visualise with the naked eye. On ophthalmoscopy microaneurysms appear as round dots separate from blood vessels; they can haemorrhage and leak, leading to:
• dot haemorrhages: thin, vertical haemorrhages that may be difficult to differentiate from microaneurysms
• blot haemorrhages: larger, full-thickness bipolar layer haemorrhages that represent larger areas of capillary occlusion (Fig. 12.31H).
Intraretinal microvascular anomalies are perfused, dilated stumps of capillaries within areas of widespread capillary occlusion. Venous beading is associated with adjacent capillary bed destruction.
Microaneurysms, dot and blot haemorrhages, intraretinal microvascular anomalies and venous beading are all surrogate markers for capillary occlusion. If sufficient capillaries occlude, then new vessels will form in the potential space between the retina and the posterior vitreous surface. They differ from normal retinal vessels because they form returning loops that are distally more dilated than their proximal origins. New vessels grow into the posterior surface of the vitreous and are found at the border of perfused and non-perfused retina (Fig. 12.32). The vitreous is most strongly attached to the optic disc, and new vessels here are more likely to haemorrhage than elsewhere.
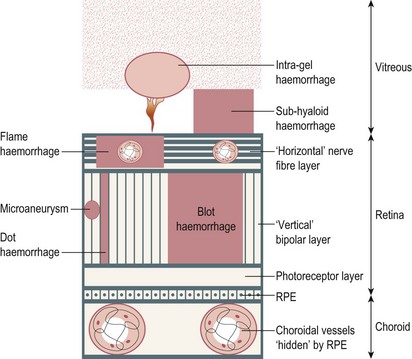
Fig. 12.32 Types of optic fundal haemorrhage, according to the retinal level.
RPE, retinal pigment epithelium.
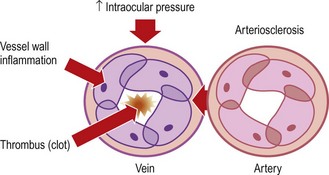
Retinal veins and arteries share a common tunica adventitia where their branches cross over. Arteriosclerosis, commonly seen with hypertension, produces arteriovenous nipping, where the thickened artery, trapped by its tunica adventitia, twists and compresses the underlying vein. Arteriosclerosis is the most common cause of retinal vein occlusion. Raised intracapillary pressure, due to retinal vein occlusion, results in capillary rupture and retinal haemorrhage. In central retinal vein occlusion, new vessel formation may occur on the iris (rubeosis iridis). Subsequent scarring of the drainage angle leads to rubeotic glaucoma, which produces extremely painful blindness. Eyes at risk often have a relative afferent pupillary defect and profound visual loss (Fig. 12.33).
Arteriosclerotic retinal vein compression usually occurs in elderly patients or those with arteriosclerotic risk factors, e.g. smoking, hypertension, hyperlipidaemia or diabetes mellitus. Raised intraocular pressure from chronic open-angle glaucoma is also a common cause.
Retinal pigment epithelium and photoreceptors: Disease of the retinal pigmented cells produces areas of depigmentation with adjacent clumps of pigment. Age-related macular degeneration is the commonest cause of change. It is preceded by drusen formation – amorphous depositions under the retinal pigment epithelium (Fig. 12.34A). Differentiate drusen from hard exudates, in people with diabetes mellitus, by the absence of adjacent microaneurysms. Atrophic age-related macular degeneration results in areas of pigment atrophy and gradual loss of central vision. Neovascular age-related macular degeneration is more severe and is associated with rapid-onset visual distortion and central visual loss.
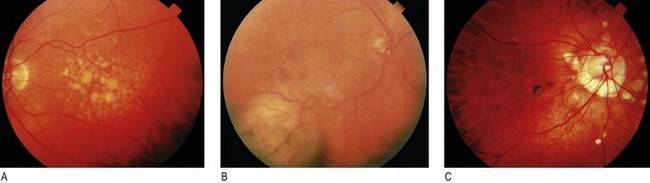
Fig. 12.34 Retinal pigment abnormalities.
(A) Drusen maculopathy; numerous large retinal drusen affecting the central retina. (B) Melanoma. Large and more significantly raised pigmented lesion deep to the retinal vessels, indicating its choroid origin. (C) Choroiditis. Multiple white lesions (multifocus choroiditis) with additional greenish choroidal neovascular membrane and adjacent retinal haemorrhage.
Choroidal naevi (hyperpigmentation of the retinal pigment epithelium) is a common asymptomatic finding. In contrast, malignant melanomas (Fig. 12.34B) are usually symptomatic and elevated, progress in size and may be associated with retinal detachment and vitreous haemorrhage.
Choroiditis (inflammation of the choroid) appears as white spots (Fig. 12.34C). When active, they have a white, poorly defined, fluffy edge with an overlying hazy vitreous, causing blurring of vision. When inactive, they have a well-defined pigmented edge. Retinal blood vessels are unaffected, and clearly visible as they cross the choroiditis. Choroiditis is associated with toxoplasmosis, sarcoidosis and tuberculosis.
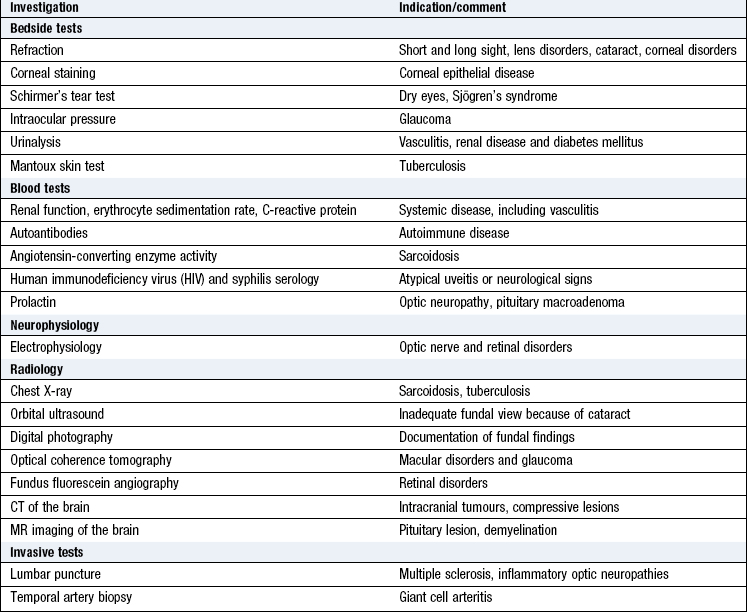
Investigations
Specialised tests may look at the visual system itself or investigate associated systemic disorders (Box 12.11).