The cardiovascular system
The heart
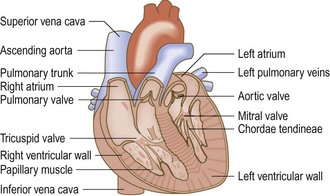
The heart comprises two muscular pumps working in series, covered in a serous sac (pericardium) which allows free movement with each heart beat and respiration. The heart delivers blood to both pulmonary and systemic circulations (Fig. 6.1). The right heart (right atrium and ventricle) pumps deoxygenated blood returning from the systemic veins into the pulmonary circulation at relatively low pressures. The left heart (left atrium and ventricle) receives blood from the lungs and pumps it round the body to the tissues at higher pressures (Fig. 6.2). The heart muscle (myocardium) is thicker in the ventricles than in the atria and in the left ventricle than the right ventricle, to generate higher pressures.
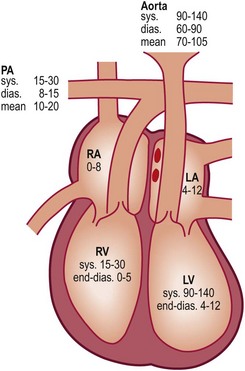
Fig. 6.2 Normal resting pressures (mmHg) in the heart and great vessels.
PA, pulmonary artery; RA, right atrium; LA, left atrium; RV, right ventricle; LV, left ventricle; sys., systolic; dias., diastolic.
Heart valves
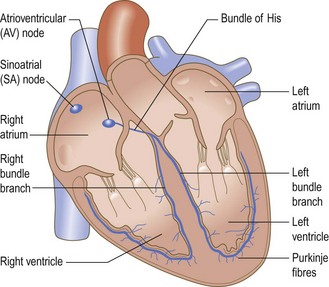
Atrioventricular valves (tricuspid on the right side, mitral on the left) separate the atria from the ventricles. They are attached to papillary muscles in the ventricular myocardium by chordae tendineae (Fig. 6.1) which prevent them from prolapsing into the atria when the ventricle contracts. The pulmonary valve on the right side of the heart and the aortic valve on the left separate the ventricles from the pulmonary and systemic arterial systems respectively. Each has three half-moon-shaped cusps called semilunar valves. Cardiac contraction is coordinated by specialised groups of cells (Fig. 6.3). The cells in the sinoatrial node normally act as the cardiac pacemaker. Subsequent spread of impulses through the heart ensures that atrial contraction is complete before ventricular contraction (systole) begins. At the end of systole the atrioventricular valves open, allowing blood to flow from the atria to refill the ventricles (diastole).
Symptoms and definitions
See Box 6.1.
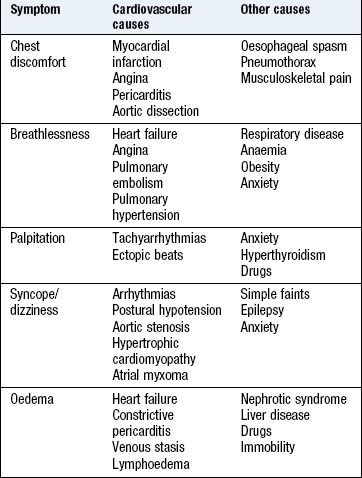
Chest pain and discomfort
Chest pain and discomfort are crucial symptoms because of their association with major pathology such as coronary artery disease and aortic dissection. Use a systematic history to distinguish serious from benign causes. Patients often describe discomfort rather than pain, and the severity of discomfort does not necessarily reflect the severity of the underlying problem. Coronary artery disease may produce no symptoms in its early phases and in elderly or diabetic patients.
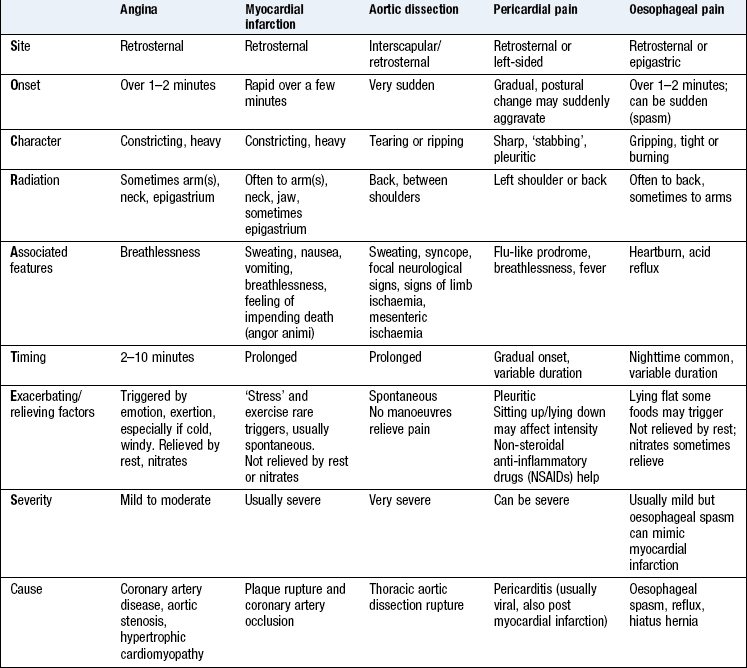
Angina pectoris: Angina pectoris is the most common cardiac pain. It is usually due to myocardial ischaemia from obstructed flow in an epicardial coronary vessel, but can occur in conditions such as aortic stenosis or hypertrophic cardiomyopathy when there is increased myocardial oxygen demand due to increased left ventricular afterload (Box 6.2). Characteristically angina is an ache or dull discomfort, felt diffusely in the centre of the anterior chest, lasting <10 minutes. Patients describe a tight or pressing ‘band-like’ sensation, similar to a heavy weight, which can be confused with indigestion. It may radiate down one or both arms and into the throat, jaw and teeth (Fig. 6.4). It is not affected by inspiration, twisting or turning. The severity of the discomfort is a poor guide to prognosis. It may be precipitated by anything that increases the force of cardiac contraction, heart rate or blood pressure (BP) and increases myocardial oxygen demand. Triggers include:
• cold or windy weather (causes peripheral vasoconstriction)
• walking uphill or carrying a heavy load (increases cardiac output and BP)
• exercise following a heavy meal (postprandial angina) causing redistribution of myocardial blood flow.
Angina is relieved by rest and glyceryl trinitrate (GTN), and is more likely to occur early during exercise. Some patients describe ‘walk-through’ angina, as peripheral vasodilatation during exercise decreases myocardial workload. Use an objective assessment of the impact of angina on the patient’s activity (Box 6.3):
• Unstable angina is limiting angina of abrupt onset, or of increasing severity, duration or frequency. It may occur with minimal exertion or at rest. It is a medical emergency, as it may herald myocardial infarction.
• Crescendo angina occurs at increasing frequency at lower workloads, but not at rest.
• Nocturnal or decubitus angina occurs at night or on lying flat. It is caused either by increased venous return or reducing efficacy of antiangina drugs, which are often taken in the morning and may wear off overnight. It indicates severe coronary artery disease.
It may be difficult to distinguish between angina pectoris and non-cardiac causes of chest pain, such as oesophageal pain (Box 6.2).
Myocardial infarction causes symptoms that are similar to, but more severe and prolonged than, those of angina pectoris. Other features include restlessness, breathlessness and a feeling of impending death (angor animi). Autonomic stimulation produces sweating, pallor, nausea, vomiting and diarrhoea, particularly in inferior wall infarction. Pain is absent in up to 30% of patients with myocardial infarction, especially the elderly and those with diabetes mellitus.
Pericardial pain is a sharp anterior central chest pain exacerbated by inspiration and movement, particularly leaning forward. It may be confused with angina but both may coexist. It is caused by inflammation of the pericardium secondary to myocardial infarction, viral infection, or after surgery, catheter ablation, angioplasty or radiotherapy.
Aortic dissection is a tear in the intima of the aorta that allows blood to penetrate the media under high pressure, cleaving the aortic wall. It is usually associated with abrupt onset of very severe, tearing chest pain which can radiate to the back (typically interscapular) and may be associated with profound autonomic stimulation. If the tear involves the coronary, cranial or upper limb arteries, it may cause myocardial infarction, syncope, stroke and upper limb pulse asymmetry. If the tear extends into the thoracoabdominal aorta it can affect the intercostal, visceral, lumbar, renal and iliac arteries, leading to paraplegia, mesenteric infarction, renal failure and lower limb ischaemia (often with an absent femoral pulse on the affected side if the dissection extends into the iliac artery). Predisposing factors include smoking, hypertension and connective tissue disorders such as Marfan’s syndrome (Fig. 3.28).
Dyspnoea (breathlessness)
This is an awareness of increased drive to breathe and is normal on exercise. It is pathological if it occurs at a significantly lower threshold than expected. Breathlessness is a non-specific symptom and may be caused by cardiac, respiratory, neuromuscular and metabolic conditions, or by toxins or anxiety (Ch. 7).
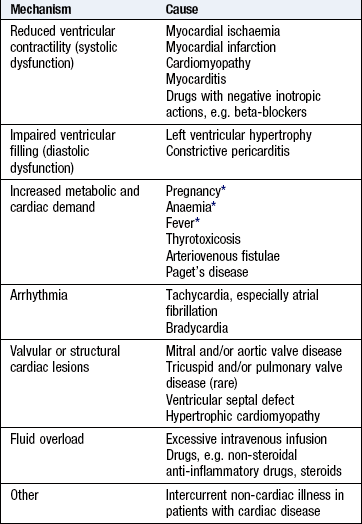
Dyspnoea may be caused by myocardial ischaemia and is known as ‘angina equivalent’. It may occur instead of, or with, chest discomfort, especially in elderly and diabetic patients. It has identical precipitants to angina and may be relieved by GTN. Dyspnoea in heart failure may be associated with fatigue. Pulmonary oedema (accumulation of fluid in the alveoli) occurs with left heart failure because increased left atrial end-diastolic pressure leads to elevated pressure in the pulmonary veins and capillaries (Box 6.4). Patients with acute pulmonary oedema usually prefer to be upright. Those with pulmonary embolism are often more comfortable lying flat and may faint (syncope) if made to sit upright. Use the New York Heart Association grading system to assess the degree of symptomatic limitation caused by the exertional breathlessness of heart failure (Box 6.5). Other cardiovascular causes of acute breathlessness include pulmonary embolism and arrhythmias.
Orthopnoea is dyspnoea on lying flat and is a sign of advanced heart failure. Lying flat increases venous return and in patients with left ventricular impairment may precipitate pulmonary oedema. The severity can be graded by the number of pillows used at night, e.g. ‘three-pillow orthopnoea’.
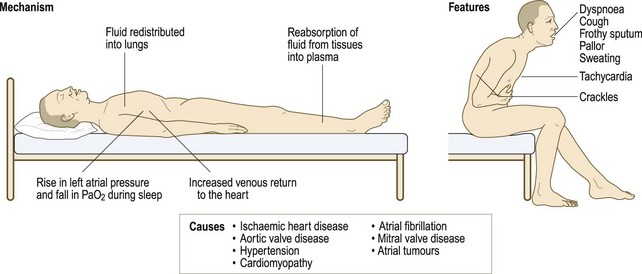
Paroxysmal nocturnal dyspnoea is sudden breathlessness waking the patient from sleep (Fig. 6.5). It is caused by accumulation of alveolar fluid. Patients may choke or gasp for air, sit on the edge of the bed and open windows in an attempt to relieve their distress. It may be confused with asthma, which can also cause night-time dyspnoea, chest tightness, cough and wheeze, but patients with heart failure may also produce frothy, blood-stained sputum.
Platypnoea is breathlessness on sitting upright. It is much rarer than orthopnoea and is usually associated with deoxygenation (platypnoea–orthodeoxia syndrome). It requires both anatomical and functional abnormalities. The anatomical component is usually an intracardiac communication, e.g. atrial septal defect. Platypnoea then develops when a right-to-left shunt occurs because of the functional component. This may be cardiac, e.g. pericardial effusion; pulmonary, e.g. pneumonectomy; abdominal, e.g. liver cirrhosis; or vascular, e.g. aortic aneurysm.
Palpitation
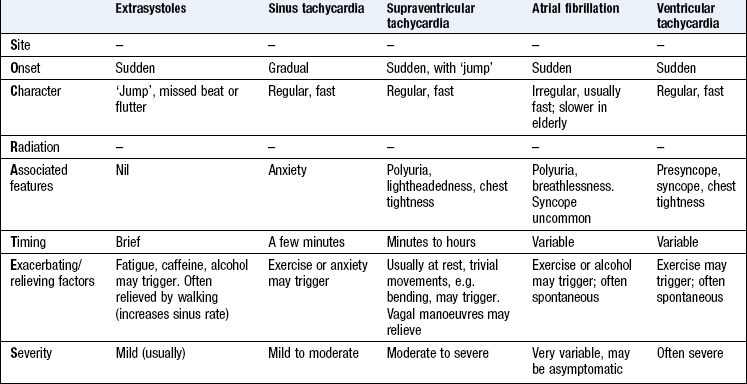
Palpitation is an unexpected awareness of the heart beating in the chest. It may be rapid, forceful or irregular, and described as thumping, pounding, fluttering, jumping, racing or skipping. The patient may be able to mimic the rhythm by tapping it out.
Palpitation may occur in sinus rhythm with anxiety, with in4termittent irregularity of the heart beat, e.g. extrasystoles, or with an abnormal rhythm (arrhythmia). Not all patients with arrhythmia experience palpitation, e.g. atrial fibrillation often occurs in the elderly but rarely causes palpitation. The history helps distinguish different types of palpitation (Box 6.6).
Healthy people are occasionally aware of their heart beating with normal (sinus) rhythm, e.g. after exercise, or when waiting for an interview or examination. The sensation is more common in bed at night when external visual and auditory inputs are minimal and visceral sensations are more prominent. Slim people may notice it when lying on their left side.
Palpitation can be induced by excessive caffeine or nicotine intake. Prescription or ‘over-the-counter’ drugs can cause palpitation, e.g. decongestants, antihistamines, as can stimulant recreational drugs, e.g. amphetamines, ecstasy and cocaine. Carotid artery disease may cause an intermittent whooshing noise (bruit) heard in the affected side of the head during an arrhythmia.
Ectopic beats (extrasystoles) are a benign cause of palpitation at rest and are abolished by exercise. Patients may describe ‘missed beats’, sometimes followed by a particularly strong heart beat. The ectopic beat produces a small stroke volume and an impalpable impulse due to incomplete left ventricular filling. The subsequent compensatory pause leads to ventricular overfilling and a forceful contraction with the next beat.
Supraventricular tachycardia produces sudden paroxysms of rapid, regular palpitation which can sometimes be terminated with breathing manoeuvres or carotid sinus pressure. Supraventricular tachycardia often affects young patients with no other underlying cardiac disease.
Ventricular tachycardia can produce similar symptoms but is more often associated with presyncope or syncope, and tends to affect patients with cardiomyopathy or previous myocardial infarction.
Urgently investigate palpitation with any high-risk features, including:
• Recent (<3 months) myocardial infarction, percutaneous coronary intervention or cardiac surgery
• Associated syncope or severe chest pain
• Family history of syncope or sudden death
• Wolff–Parkinson–White syndrome, or inherited channelopathy, e.g. long QT syndrome
• Significant structural heart disease, e.g. hypertrophic cardiomyopathy, aortic stenosis.
Syncope
Syncope is a loss of consciousness due to cerebral hypoperfusion. Dizziness may be due to vertigo or lightheadedness (p. 245). Vertigo is rarely caused by heart disease. Lightheadedness, syncope or a feeling of impending loss of consciousness (presyncope) may be cardiovascular in origin. The main causes are:
Patients with vascular disease affecting the carotid and/or vertebral arteries may present with non-focal cerebral symptoms due to hypoperfusion. Common precipitating factors are head turning, getting up quickly from sitting or lying and starting or increasing antihypertensive drugs.
Postural hypotension is a fall of >20 mmHg in systolic BP on standing. It can be caused by hypovolaemia, antihypertensive drug therapy, especially diuretics and vasodilators (Box 6.7), and autonomic neuropathy. Postural hypotension is common in the elderly, affecting up to 30% of individuals aged >65 years.
Neurocardiogenic syncope is a group of conditions caused by abnormal autonomic reflexes. A simple faint occurs in healthy people forced to stand for a long time in a warm environment or subject to painful or emotional stimuli, e.g. the sight of blood. It results from sudden slow heart rate (bradycardia) and/or vasodilatation. There may be a prior history of fainting with a prodrome of lightheadedness, tinnitus, nausea, sweating and facial pallor and a darkening of vision from the periphery as the retinal blood supply (the most oxygen-sensitive part of the nervous system) is reduced,. The person then slides to the floor, losing consciousness. When laid flat to aid cerebral circulation, the individual wakes up, often flushing from vasodilatation and nauseated or even vomiting due to vagal overactivity. If held upright by misguided bystanders, continued cerebral hypoperfusion delays recovery and may lead to a seizure and a mistaken diagnosis of epilepsy.
Frequent fainting caused by minor stimuli may be due to malignant vasovagal syndrome or hypersensitive carotid sinus syndrome (HCSS). In patients with HCSS, gentle pressure over the carotid sinus may reproduce the symptoms by triggering bradycardia.
Arrhythmias can cause syncope or presyncope. The most common cause is bradyarrhythmia, due to sinoatrial disease or to atrioventricular block, i.e. Stokes–Adams attacks. Drugs, including digoxin, beta-blockers and rate-limiting calcium channel blockers, e.g. verapamil, diltiazem are a common cause of bradyarrhythmia. Supraventricular tachyarrhythmias, e.g. atrial fibrillation, rarely cause syncope. Ventricular tachycardia often causes syncope or presyncope, especially in patients with impaired left ventricular function.
Mechanical obstruction to cardiac output, including severe aortic stenosis and hypertrophic cardiomyopathy, can obstruct left ventricular outflow causing syncope or presyncope, especially on exertion when cardiac output cannot meet the increased metabolic demand.
Pulmonary embolism can obstruct outflow from the right ventricle, and is a frequently overlooked cause of recurrent syncope. Cardiac tumours, e.g. atrial myxoma, and thrombosis or failure of prosthetic heart valves are rare causes of syncope.
Oedema
Excess fluid in the interstitial space causes oedema (tissue swelling). It is usually gravity-dependent and so especially seen around the ankles, or over the sacrum in patients lying in bed. The most common causes of lower limb swelling are chronic venous disease and lymphoedema. Other causes include heart failure and vasodilator medications (Box 6.8 and Box 3.16). In general, if the jugular venous pressure (JVP) is not elevated, then oedema is not cardiogenic.
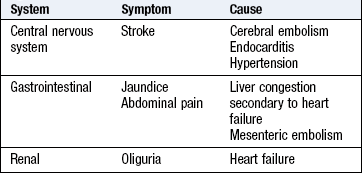
Infective endocarditis is microbial infection of the heart valves (natural or prosthetic), the lining of the cardiac chambers or blood vessels or a congenital abnormality, e.g. septal defect. The causative organism is usually bacterial, but may be fungal, rickettsial or Chlamydia. The presentation may be acute or subacute. Many features of infective endocarditis are thought to result from circulating immune complexes or emboli, e.g. petechial rash, haematuria, splinter haemorrhages, cerebral emboli. Features such as fever, splenomegaly and clubbing may occur.
Other symptoms
Non-cardiac symptoms occur in heart disease (Box 6.9). Infective endocarditis may present with non-specific symptoms, including weight loss, tiredness, fever and night sweats, and atrial fibrillation with symptoms and signs of cerebral or systemic embolisation (commonly legs, arms or viscera).
The history
Heart disease commonly occurs without abnormal physical findings so the history is critical in making a diagnosis. Examination may confirm a cardiac diagnosis, e.g. murmur or signs of heart failure but, even in serious disease, physical signs may be completely absent.
• Patients with severe carotid artery stenosis may have no neck bruit due to low volume flow.
• Large abdominal aortic aneurysms (AAAs) can be impalpable in the obese.
• Patients with extensive deep vein thrombosis (DVT) often appear to have normal legs.
Presenting complaint
Establish the frequency, duration and severity of symptoms, exacerbating and relieving factors. Urgently attend to breathlessness, recent chest or lower limb pain. As many cardiovascular diseases are slowly progressive, the evolution of symptoms guides the timing of investigations and treatment, e.g. surgery for carotid artery disease is most effective soon after a cerebral event; and heart valve surgery is indicated for significantly limiting symptoms.
Functional impairment: How do symptoms affect your patient’s functional capacity? Establish the intensity of exercise required to induce symptoms.
• Are symptoms provoked by gentle walking or strenuous exercise like climbing hills or stairs?
• Can patients keep up when walking with their peers?
• Do patients feel frustrated or restricted by their symptomatic limitation?
• How are domestic (cooking, cleaning, shopping), social (hobbies, sport) and occupational activities limited?
Lightheadedness and syncope may impair confidence, raise fear of physical injury and have safety implications when driving.
Calf leg pain on walking (intermittent claudication) from lower limb arterial disease is the most common symptom of peripheral vascular disease (p. 127):
Past history: Ask about rheumatic fever or heart murmurs during childhood and associated conditions, including:
• Thyrotoxicosis (atrial fibrillation)
• Marfan’s syndrome (aortic regurgitation or aortic dissection).
In suspected infective endocarditis ask about potential causes of bacteraemia e.g. skin infection; recent dental work; intravenous drug use and penetrating trauma.
Consider possible links between other diseases and cardiovascular illness. Examples include patients with renal failure or cancer and pericardial effusion; cytotoxic drugs and heart failure; radiotherapy and radiation arteritis. Patients with chronic lung disease may develop right-sided heart failure (cor pulmonale; p. 115) or atrial fibrillation. Connective tissue diseases, e.g. rheumatoid arthritis, are associated with Raynaud’s phenomenon (Fig. 6.38) and pericarditis.
Drug history: Drugs may cause or aggravate symptoms such as breathlessness, chest pain, oedema, palpitation or syncope (Box 6.7). Starting thyroxine for hypothyroidism may precipitate or exacerbate angina. ‘Recreational’ drugs such as cocaine and amphetamines can cause arrhythmias, chest pain, occlusive and aneurysmal peripheral arterial disease (PAD) and even myocardial infarction. Ask about ‘over-the-counter’ purchases such as NSAIDs, alternative and herbal medicines, as these may have cardiovascular actions. Beta-blockers and antihypertensives may impair the peripheral circulation and aggravate symptoms of intermittent claudication.
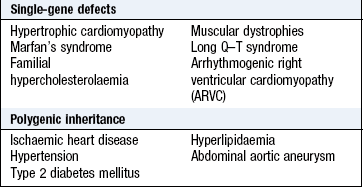
Family history: Many cardiac disorders have a genetic component (Box 6.10). Ask about premature coronary artery disease in first-degree relatives (<60 years in a female or <55 years in a male) or sudden unexplained death at a young age, raising the possibility of a cardiomyopathy or inherited arrhythmia disorder. Patients with venous thrombosis may have inherited thrombophilia, e.g. factor V Leiden mutation. Familial hypercholesterolaemia is associated with premature arterial disease.
Social history: Smoking is the strongest risk factor for coronary artery and PAD. Take a detailed smoking history (p. 16).
Alcohol can induce atrial fibrillation and, in excess, is associated with obesity, hypertension and dilated cardiomyopathy. Excess alcohol intake with poor nutrition also predisposes to PAD. Intravenous drug use can damage peripheral arteries and veins, causing infected false aneurysms, e.g. of the common femoral artery in the groin, which can act as a source for infective endocarditis.
Occupational history: Heart disease may impair physical activity and affect employment. This may be a source of anxiety and an indication for treatment. The diagnosis of heart disease and/or PAD has significant consequences in certain occupations, e.g. commercial drivers and pilots (Box 6.11). Workers exposed to occupational vibration through the use of air-powered tools may develop hand–arm vibration syndrome, which presents with vasospastic symptoms, e.g. Raynaud’s phenomenon, and neurosensory (numbness, tingling) symptoms.
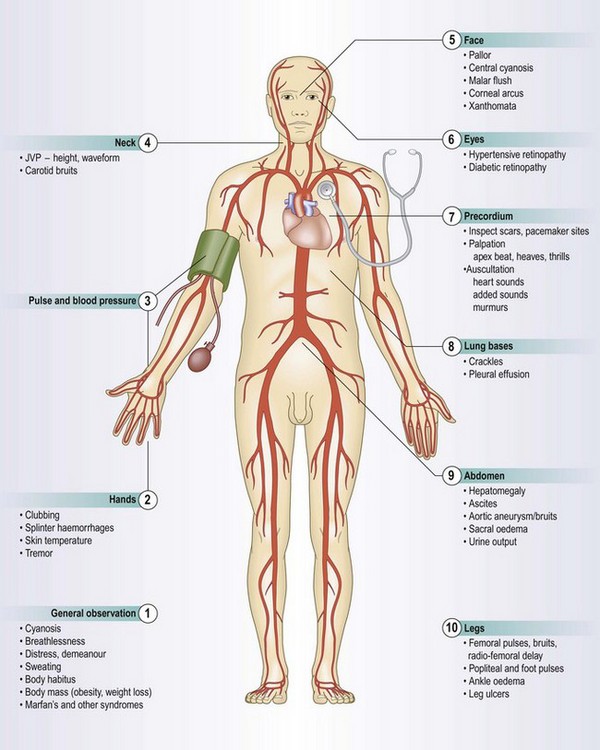
The physical examination
Tailor the sequence and extent of examination to the patient’s condition:
• Patients with cardiac or respiratory arrest or requiring immediate emergency: manage first and leave more detailed examination for later (Fig. 19.9).
General examination
• Look at the patient’s general appearance. Does he look:
• Check the temperature (p. 61).
Normal findings: The hands usually feel dry at ambient temperature. Peripheral cyanosis (p. 45) is common in healthy patients when the hands are cold. One or two isolated splinter haemorrhages are common in healthy individuals from trauma.
Abnormal findings: Fever is a feature of infective endocarditis and pericarditis and may occur after myocardial infarction. With autonomic stimulation the hands may feel warm and sweaty; with hypotension and shock they may be cold and clammy.
Splinter haemorrhages are found in infective endocarditis and some vasculitic disorders.
A petechial rash (caused by vasculitis), most often present on the legs and conjunctivae (Fig. 6.6), is a transient finding in endocarditis and can be confused with the rash of meningococcal disease (Fig. 17.2A). Janeway lesions, Osler’s nodes, nail fold infarcts and finger clubbing are uncommon features of endocarditis (Ch. 3 and Fig. 6.6).
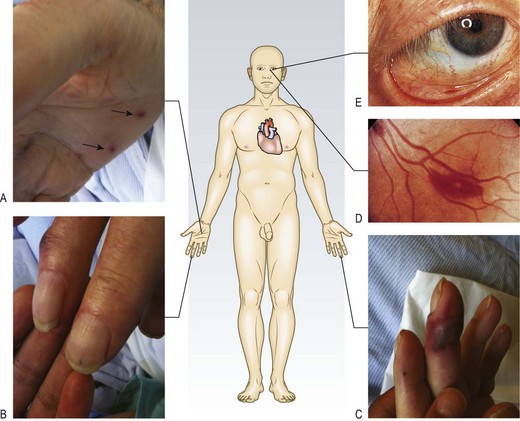
Fig. 6.6 Clinical features which may be present in infective endocarditis:
(A) Janeway lesions on hypothenar eminence (arrows), (B) Splinter haemorrhages, (C) Osler’s nodes, (D) Roth’s spot on fundoscopy, (E) Petechial haemorrhages on conjunctiva.
Urinalysis is necessary to check haematuria (endocarditis, vasculitis), glucose (diabetes) and protein (hypertension and renal disease).
Abnormal findings: Central cyanosis may be due to heart failure or, rarely, congenital heart disease, where it is associated with right-to-left shunting and finger clubbing (p. 49).
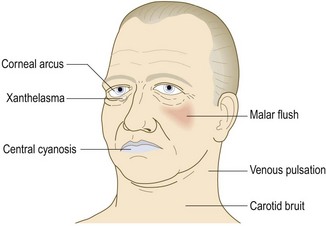
Xanthelasmata are an important predictor of cardiovascular disease (Figs 6.7 and 6.8A). If present, also check the patellar and Achilles tendons for xanthomata (Fig. 6.8B).
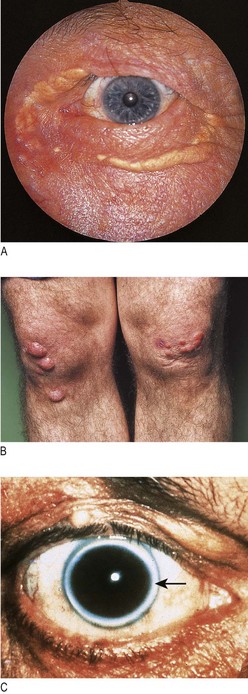
Fig. 6.8 Features of hyperlipidaemia.
(A) Xanthelasma. (B) Skin xanthomata over knees. (C) Corneal arcus (arrow).
Corneal arcus is a creamy yellow discoloration at the boundary of the iris and cornea caused by cholesterol deposition. It is more common in men and black patients (Fig. 6.8C). Both xanthelasmata and corneal arcus can, however, occur in normolipidaemic patients (Box 6.12).
Roth’s spots (Fig. 6.6) are caused by a similar mechanism to splinter haemorrhages and can also occur in anaemia or leukaemia.
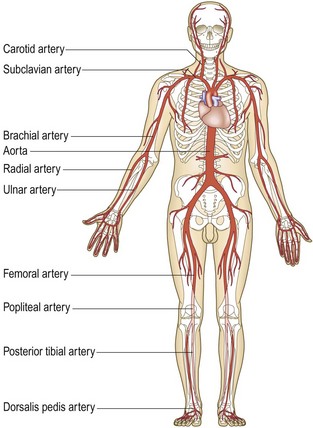
Anatomy: Ejection of blood from the left ventricle into the systemic arterial circulation (Fig. 6.9) creates a pressure wave that can be felt as a ‘pulse’ where the arteries are superficial or they pass over bone. This pressure wave is not the same as, and travels faster than, the blood flow itself. It can be possible, therefore, to feel a ‘pulse’ even if there is no flow in the artery being palpated. The pulse waveform depends upon heart rate, stroke volume, left ventricular outflow obstruction, arterial elasticity and peripheral resistance.
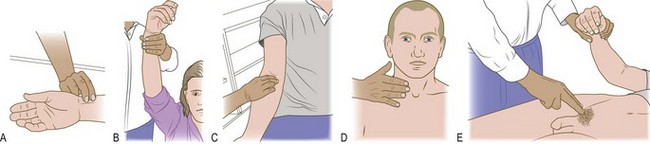
Use the larger (brachial, carotid or femoral) pulses to assess the pulse volume and character (Box 6.13). When taking a pulse, assess:
Record individual pulses as:
If you are in any doubt about whose pulse you are feeling, palpate your own pulse at the same time. If it is not synchronous with yours, it is the patient’s.
Carotid pulse: Some clinicians consider routine examination of the carotid pulse is inappropriate because it may cause distal vascular events, e.g. transient ischaemic attack, or induce reflex, vagally mediated bradycardia. In assessing a patient who may have had a cardiac arrest, however, it is the pulse of choice.
If you do examine the carotid pulse do this gently and never assess both carotids simultaneously.
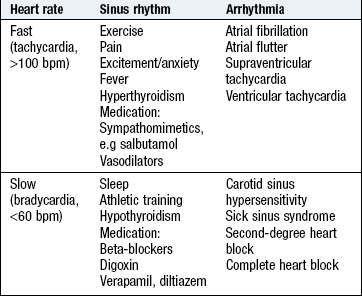
Rate: Assess the pulse rate in the clinical context. A pulse rate of 40 bpm can be normal in a fit young adult, whereas a pulse rate of 65 bpm may be abnormally low in acute heart failure. Resting heart rate is normally 60–90 bpm.
Bradycardia is a pulse rate <60 bpm; tachycardia is a rate of >100 bpm.
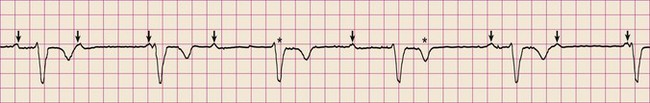
Rhythm: Sinus rhythm originates from the sinoatrial node and produces a regular rhythm (Fig. 6.12A). It varies slightly with the respiratory cycle, mediated by the vagus nerve, and is most pronounced in children, young adults or athletes (sinus arrhythmia). During inspiration, parasympathetic tone falls and the heart rate increases; on expiration, the heart rate decreases (Box 6.14).
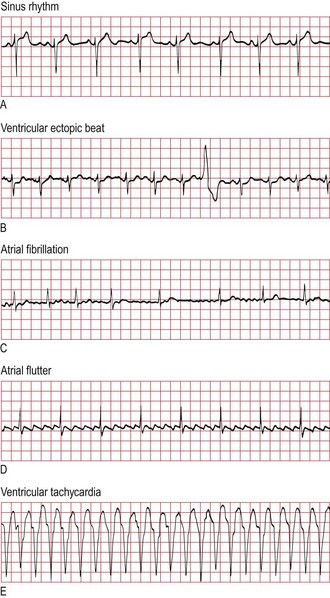
Fig. 6.12 Electrocardiogram rhythm strip.
(A) Sinus rhythm. (B) Ventricular ectopic beat. (C) Atrial fibrillation with ‘controlled’ ventricular response. (D) Atrial flutter: note the regular ‘saw-toothed’ atrial flutter waves at about 300/min. (E) Ventricular tachycardia, with ventricular rate of about 150/min.
Rate: The most common causes of bradycardia are medication, athletic conditioning, and sinoatrial or atrioventricular node dysfunction. The most common cause of tachycardia is sinus tachycardia (Box 6.15).
Rhythm: The pulse may be regular or irregular (Box 6.16). If irregular, it may be regularly irregular, due to an ectopic beat occurring at a regular interval or to second-degree atrioventricular block (Fig. 6.12B). Atrial fibrillation is the most common cause of an irregularly irregular pulse (Box 6.17 and Fig. 6.12C). The rate in atrial fibrillation depends on the number of beats conducted by the atrioventricular node. Untreated, the ventricular rate may be very fast (up to 200 bpm). The variability of the pulse rate (and therefore ventricular filling) explains why the pulse volume varies and there may be a pulse deficit, with some cycles not felt at the radial artery. Calculate the pulse deficit by counting the radial pulse rate and subtracting this from the apical heart rate assessed by auscultation (Fig. 6.12D and E).
Volume: The ventricles fill during diastole. Longer diastolic intervals are associated with increased stroke volume, which is reflected by increased pulse volume on examination. This is why pulse volume and BP vary widely during atrial fibrillation, and why the ‘compensatory pause’ following a premature ectopic beat is sometimes felt by the patient.
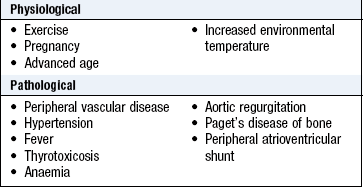
A large pulse volume is a reflection of a large pulse pressure, which can be physiological or pathological (Box 6.18). The most common cause of a large pulse pressure is arteriosclerosis, which is seen in patients with widespread vascular disease, hypertension and advanced age.
A low pulse volume may be due to reduced stroke volume and occurs in left ventricular failure, hypovolaemia or peripheral arterial disease.
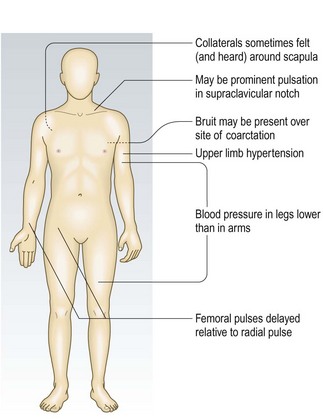
Coarctation is a congenital narrowing of the aorta, usually distal to the left subclavian artery. The clinical signs depend on the location and severity of the narrowing and the patient’s age. In children, the upper limb pulses are usually normal with reduced volume lower limb pulses, which are delayed relative to the upper limb pulses (radiofemoral delay) (Fig. 6.11). In adults, coarctation usually presents with hypertension and heart failure.
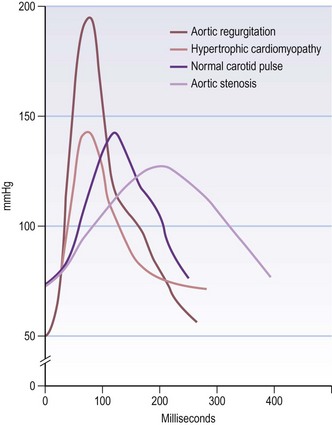
Character: A collapsing pulse is when the peak of the pulse wave arrives early and is followed by a rapid descent. This rapid fall imparts the ‘collapsing’ sensation. This is exaggerated by raising the patient’s arm above the level of the heart (Fig. 6.10B). It occurs in severe aortic regurgitation and is associated with wide pulse pressure (systolic BP – diastolic BP >80 mmHg).
A slow-rising pulse has a gradual upstroke with a reduced peak occurring late in systole, and is a feature of severe aortic stenosis.
Pulsus bisferiens is an increased pulse with a double systolic peak separated by a distinct mid-systolic dip. Causes include aortic regurgitation, and concomitant aortic stenosis and regurgitation (Fig. 6.13).
Pulsus alternans is a beat-to-beat variation in pulse volume with a normal rhythm. It is rare and occurs in advanced heart failure.
Pulsus paradoxus is an exaggeration of the normal variability of pulse volume with breathing. Pulse volume normally increases in expiration and decreases during inspiration due to intrathoracic pressure changes affecting venous return to the heart. This variability in exaggerated diastolic filling of both ventricles is impeded by increased intrapericardial pressure. This occurs in cardiac tamponade because of accumulation of pericardial fluid and in constrictive pericarditis.
Blood pressure
BP is a measure of the pressure that the circulating blood exerts against the arterial walls. Systolic BP is the maximal pressure that occurs during ventricular contraction (systole). During ventricular filling (diastole), arterial pressure is maintained at a lower level by the elasticity and compliance of the vessel wall. The lowest value (diastolic BP) occurs immediately before the next cycle.
BP is usually measured using a sphygmomanometer (Fig. 6.14). In certain situations, such as the intensive care unit, it is measured invasively using an indwelling intra-arterial catheter connected to a pressure sensor.
BP is measured in mmHg and recorded as systolic pressure/diastolic pressure, together with where, and how, the reading was taken, e.g. BP: 146/92 mmHg, right arm, supine.
BP is an important guide to cardiovascular risk and provides vital information on the haemodynamic condition of acutely ill or injured patients. BP constantly varies and rises with stress, excitement and environment. ‘White-coat hypertension’ occurs in patients only when a patient is seeing a healthcare worker. Ambulatory BP measurement, using a portable device at intervals during normal daytime activity and at night, is better at determining cardiovascular risk.
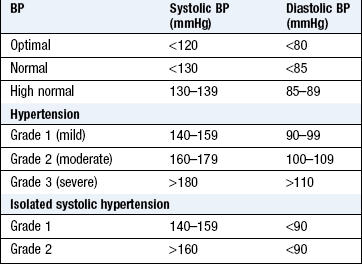
Hypertension: Abnormal elevation of BP is defined by the British Hypertension Society (Box 6.19). Normal BP is defined as <130/85 mmHg. Hypertension is extremely common, affecting 20–30% of the UK adult population, with higher rates in black Africans (Box 6.22).
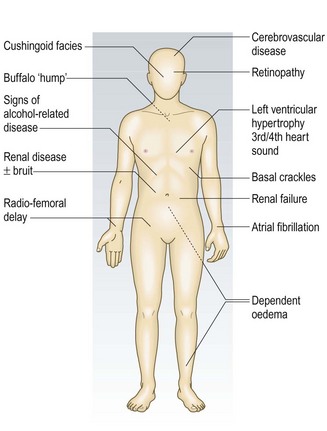
Hypertension is asymptomatic although, rarely in severe hypertension, headaches and visual disturbances occur (Fig. 6.15). It is associated with significant morbidity and mortality from vascular disease (heart failure, coronary artery disease, cerebrovascular disease and renal failure). The risk rises progressively with increasing systolic and diastolic pressure; for example, isolated grade 1 systolic hypertension has a two- to threefold increased risk of cardiac mortality. Lowering BP lowers vascular risk regardless of the starting value. In most hypertensive patients there is no identifiable cause – so-called ‘essential hypertension’. Secondary hypertension is rare, occurring in <1% of the hypertensive population (Box 6.20).
Assess the hypertensive patient for:
• renal: chronic kidney disease
• eye: hypertensive retinopathy – four grades (Fig. 6.16)
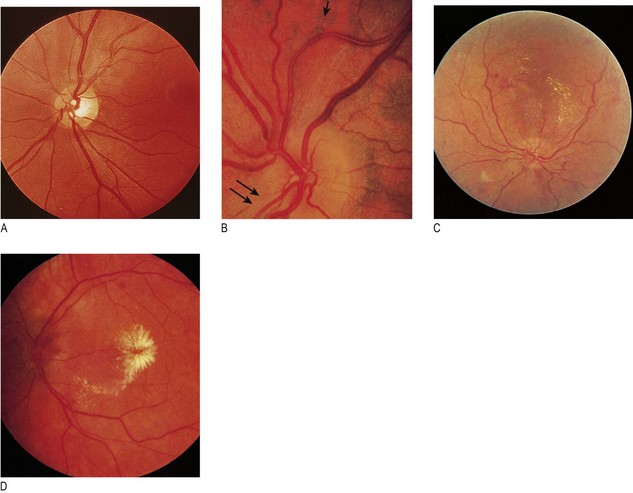
Fig. 6.16 Hypertensive retinopathy.
(A) Grade 1: early changes: increased tortuosity of a retinal vessel and increased reflectiveness (silver wiring) of a retinal artery are seen at 1 o’clock. (B) Grade 2: increased tortuosity and silver wiring (double arrow) with ‘nipping’ of the venules at arteriovenous crossings (single arrow). (C) Grade 3: similar to grade 2 plus flame-shaped retinal haemorrhages and soft ‘cotton-wool’ exudates. (D) Grade 4: swelling of the optic disc (papilloedema), retinal oedema and hard exudates around the fovea, producing a ‘macular star’.
• Overall risk of vascular disease, i.e. of stroke, myocardial infarction, heart failure.
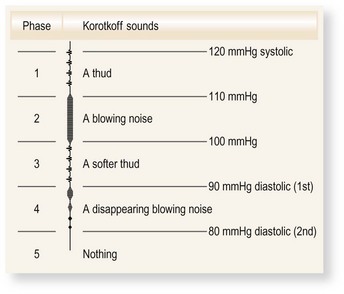
Korotkoff sounds: These sounds are produced between systole and diastole because the artery collapses completely and reopens with each heart beat, producing a snapping or knocking sound. The first appearance of sounds (phase 1) during cuff deflation indicates systole. As pressure is gradually reduced, the sounds muffle (phase 4) and then disappear (phase 5). Interobserver agreement is better for phase 5 and this is the diastolic BP. Occasionally, muffled sounds persist (phase 4) and do not disappear; in this case, record phase 4 as the diastolic pressure (Fig. 6.17).
Common problems in BP measurement:
• BP is different in each arm: a difference >10 mmHg suggests the presence of subclavian artery disease. Unequal brachial BP is a marker of increased cardiovascular morbidity and mortality (Box 6.21). Record the highest pressure and use this to guide management
• Wrong cuff size: the bladder should be approximately 80% of the length and 40% of the width of the upper arm circumference. A standard adult cuff has a bladder approximately 13 × 30 cm and suits an arm circumference 22–26 cm. In obese patients a standard adult cuff will overestimate BP, so use a large adult (bladder 16 × 38 cm) or thigh cuff (20 × 42 cm)
• Auscultatory gap: up to 20% of elderly hypertensive patients have Korotkoff sounds which appear at systolic pressure and disappear for an interval between systolic and diastolic pressure. If the first appearance of the sound is missed, the systolic pressure will be recorded at a falsely low level. Avoid this by palpating the systolic pressure first
• Patient’s arm at the wrong level: the patient’s elbow should be level with the heart. Hydrostatic pressure causes ~5 mmHg change in recorded systolic and diastolic BP for a 7 cm change in arm elevation
• Terminal digit preference: record the true reading rather than rounding values to the nearest 0 or 5
• Postural change: the pulse increases by about 11 bpm, systolic BP falls by 3–4 mmHg and diastolic BP rises by 5–6 mmHg when a healthy person stands. The BP stabilises after 1–2 minutes. Check the BP after a patient has been standing for 2 minutes; a drop of ≥20 mmHg on standing is postural hypotension
• Atrial fibrillation: makes BP assessment more difficult because of beat-to-beat variability. Deflate the cuff at 2 mmHg per beat and repeat measurement if necessary.
Jugular venous pressure and waveform
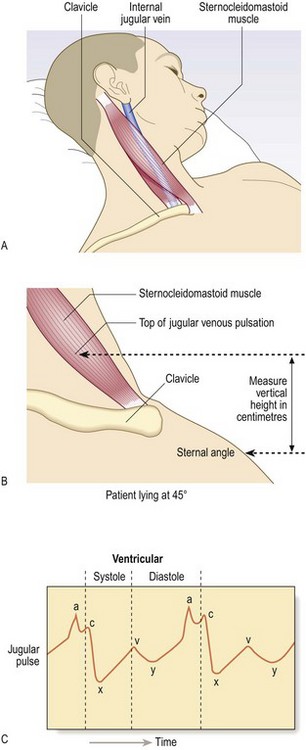
Anatomy: The internal jugular vein enters the neck behind the mastoid process. It runs deep to the sternocleidomastoid muscle before entering the thorax between the sternal and clavicular heads and should be examined with the neck muscles relaxed. A pulsation is visible when the pressure in the internal jugular vein is elevated.
The external jugular vein is more superficial, prominent and easier to see. It can be kinked or obstructed as it traverses the deep fascia of the neck but, when visible, pulsatile and not obstructed, it can be used to estimate the JVP in difficult cases.
Estimate the JVP by observing the level of pulsation in the internal jugular vein. The normal waveform has two main peaks per cycle, which helps to distinguish it from the carotid arterial pulse (Box 6.23).
The JVP level reflects right atrial pressure (normally <7 mmHg/9 cmH2O). The sternal angle is approximately 5 cm above the right atrium, so the JVP in health should be ≤4 cm above this angle when the patient lies at 45°. If right atrial pressure is low, the patient may have to lie flat for the JVP to be seen; if high, the patient may need to sit upright (Fig. 6.18).
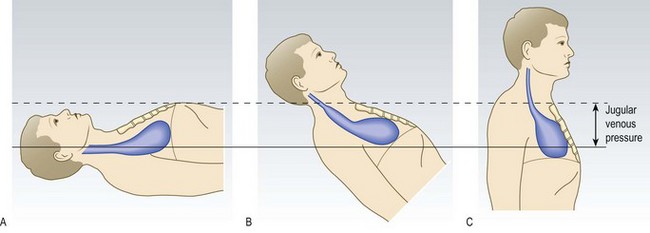
Fig. 6.18 Jugular venous pressure in a healthy subject.
(A) Supine: jugular vein distended, pulsation not visible. (B) Reclining at 45°: point of transition between distended and collapsed vein can usually be seen to pulsate just above the clavicle. (C) Upright: upper part of vein collapsed and transition point obscured.
Normal findings: Aids to differentiate the jugular venous waveform from arterial pulsation:
• Abdomino-jugular test: firmly press over the abdomen. This increases venous return to the right side of the heart temporarily and the JVP normally rises.
• Changes with respiration: the JVP normally falls with inspiration due to decreased intrathoracic pressure.
• Waveform (Fig. 6.19C): the normal JVP waveform has two distinct peaks per cardiac cycle:
• ‘a’ wave corresponds to right atrial contraction and occurs just before the first heart sound. In atrial fibrillation the ‘a’ wave is absent.
• ‘v’ wave is caused by atrial filling during ventricular systole when the tricuspid valve is closed.
• Rarely, a third peak (‘c’ wave) may be seen due to closure of the tricuspid valve.
• Occlusion: the JVP waveform is obliterated by gently occluding the vein at the base of the neck with your finger.
Abnormal findings: The JVP is primarily a sign of right ventricular function. It is elevated in states of fluid overload, notably in heart failure and in conditions with right heart dilatation, e.g. acute pulmonary embolism and chronic obstructive pulmonary disease (when it is called cor pulmonale). Mechanical obstruction of the superior vena cava (most often caused by lung cancer) may cause extreme, non-pulsatile elevation of the JVP. Here the JVP no longer reflects right atrial pressure and the abdominojugular test will be negative (Box 6.24).
Kussmaul’s sign: a paradoxical rise of JVP on inspiration seen in pericardial constriction or tamponade, severe right ventricular failure and restrictive cardiomyopathy.
Prominent ‘a’ wave: caused by delayed or restricted right ventricular filling, e.g. pulmonary hypertension or tricuspid stenosis.
Cannon waves: giant ‘a’ waves occur when the right atrium contracts against a closed tricuspid valve. Irregular cannon waves are seen in complete heart block and are due to atrio-ventricular dissociation. Regular cannon waves occur during junctional rhythm and with some ventricular and supraventricular tachycardias.
‘cv’ wave: a fusion of the ‘c’ and ‘v’ waves resulting in a large systolic wave and associated with a pulsatile liver is seen in tricuspid regurgitation.
The precordium
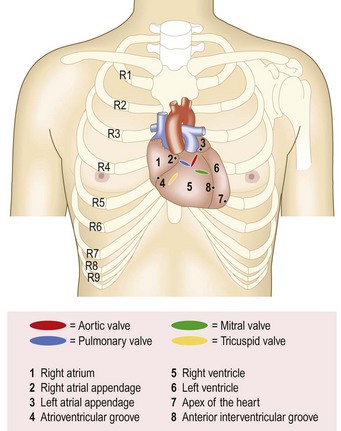
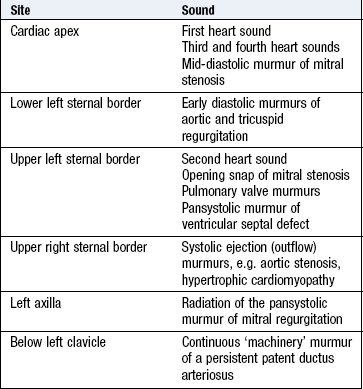
The precordium is the anterior chest surface overlying the heart and great vessels (Fig. 6.20).
Learn the surface anatomy of the heart to understand how and where the sounds and murmurs radiate and basic cardiac physiology to appreciate their timing (Figs 6.20 and 6.21). The auscultatory areas (aortic, pulmonary, apex and left sternal border) do not correspond with the location of cardiac structures, but are where transmitted sounds and murmurs are best heard (Box 6.25).
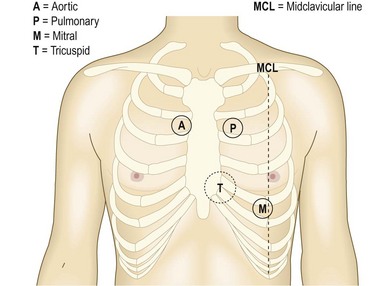
Fig. 6.21 Sites for auscultation.
Sites at which murmurs from the relevant valves are usually, but not preferentially, heard.
Chest wall abnormalities: Pectus excavatum (funnel chest), a posterior displacement of the lower sternum, and pectus carinatum (pigeon chest) may displace the heart and affect palpation and auscultation (Fig. 7.14).
A midline sternotomy scar usually indicates previous coronary artery bypass surgery or aortic valve replacement. A left submammary scar is usually the result of mitral valvotomy. Infraclavicular scars are seen after pacemaker or defibrillator implantation, and the bulge of the device may be obvious.
Apex beat: The apex beat is the most lateral and inferior position where the cardiac impulse can be felt. The cardiac impulse results from the left ventricle moving forward and striking the chest wall during systole. The apex beat is normally in the fifth left intercostal space at, or medial to, the mid-clavicular line (halfway between the suprasternal notch and the acromioclavicular joint).
A thrill is the tactile equivalent of a murmur and is a palpable vibration.
A heave is a palpable impulse that noticeably lifts your hand.
Normal findings: A normal apical impulse briefly lifts your fingers and is localised. There should be no parasternal heave or thrill.
Abnormal findings: The apex beat may be impalpable in overweight or muscular people or in patients with asthma or emphysema because the lungs are hyperinflated. It may be diffusely displaced inferiorly and laterally in left ventricular dilatation, e.g. after myocardial infarction, with aortic stenosis, severe hypertension and dilated cardiomyopathy or in chest deformity. In dextrocardia, with a prevalence of 1 : 10 000, the cardiac apex is on the right side.
Left ventricular hypertrophy, e.g. with hypertension, aortic stenosis, produces a forceful, undisplaced apical impulse. This thrusting apical ‘heave’ is quite different from the diffuse impulse of left ventricular dilatation. Pulsation over the left parasternal area (right ventricular heave) indicates right ventricular hypertrophy or dilatation, most often accompanying pulmonary hypertension. The ‘tapping’ apex beat in mitral stenosis represents a palpable first heart sound, and is not usually displaced. A double apical impulse is characteristic of hypertrophic cardiomyopathy.
The most common thrill is that of aortic stenosis which may be palpable at the apex, at the lower sternum or in the neck. The thrill caused by a ventricular septal defect is best felt at the left and right sternal edges. Diastolic thrills are very rare.
Heart sounds: Normal heart valves make a sound only when they close. The ‘lub-dub’ sounds are caused by closure of the atrioventricular (mitral and tricuspid) valves followed by the outlet (aortic and pulmonary) valves.
The bell of the stethoscope transmits all sounds well but in some patients with high-frequency murmurs any additional low-frequency sound masks the high-frequency murmur. The bell is particularly useful at the apex and left sternal edge to listen for the diastolic murmur of mitral stenosis and third and fourth heart sounds.
The diaphragm attenuates all frequencies equally, therefore making some low-frequency sounds less audible. Use the diaphragm to identify high-pitched sounds, e.g. early diastolic murmur of aortic regurgitation. Listen with it over the whole precordium for a pericardial friction rub.
• First heart sound (S1), ‘lub’, is caused by closure of the mitral and tricuspid valves at the onset of ventricular systole. It is best heard at the apex.
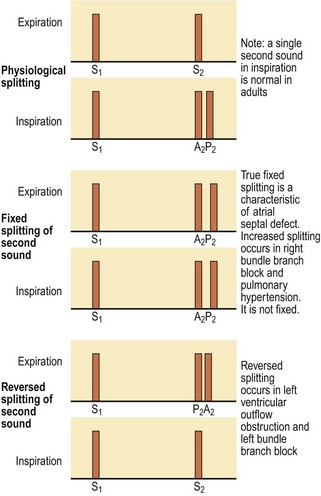
• Second heart sound (S2), ‘dup’, is caused by closure of the pulmonary and aortic valves at the end of ventricular systole and is best heard at the left sternal edge. It is louder and higher-pitched than the S1 ‘lup’, and the aortic component is normally louder than the pulmonary one. Physiological splitting of S2 occurs because left ventricular contraction slightly precedes that of the right ventricle so that the aortic valve closes before the pulmonary valve. This splitting increases at end-inspiration because increased venous filling of the right ventricle further delays pulmonary valve closure. This separation disappears on expiration (Fig. 6.24). Splitting of S2 is best heard at the left sternal edge. On auscultation, you hear ‘lub d/dub’ (inspiration) ‘lub-dub’ (expiration)
• Third heart sound (S3) is a low-pitched early diastolic sound best heard with the bell at the apex. It coincides with rapid ventricular filling immediately after opening of the atrioventricular valves and is therefore heard after the second as ‘lub-dub-dum’. It is a normal finding in children, young adults and during pregnancy.
Abnormal findings: First heart sound: In mitral stenosis the intensity of S1 is increased due to elevated left atrial pressure (Box 6.26).
Second heart sound: The aortic component of S2 is sometimes quiet or absent in calcific aortic stenosis and reduced in aortic regurgitation (Box 6.27). The aortic component is loud in systemic hypertension, and the pulmonary component increased in pulmonary hypertension.
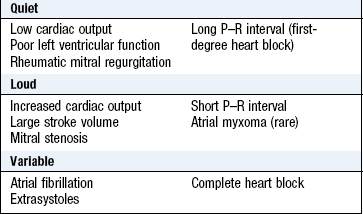
Wide splitting of S2, but with normal respiratory variation, occurs in conditions which delay right ventricular emptying, e.g. right bundle branch block. Fixed splitting, i.e. no variation with respiration of S2, is a feature of atrial septal defect (Fig. 6.25). In this condition the right ventricular stroke volume is larger than the left, and the splitting is fixed because the defect equalises the pressure between the two atria throughout the respiratory cycle.
In reversed splitting the two components of S2 occur together on inspiration and separate on expiration. This occurs when left ventricular emptying is delayed so that the aortic valve closes after the pulmonary valve. Examples include left bundle branch block and right ventricular pacing.
Third heart sound: This is usually pathological after the age of 40 years (Box 6.28). The most common causes are left ventricular failure, when it is an early sign, and mitral regurgitation, due to volume loading of the ventricle. In heart failure S3 occurs with a tachycardia, referred to as a ‘gallop’ rhythm, and S1 and S2 are quiet (lub-da-dub; Box 6.29).
Fourth heart sound: This is less common. It is soft and low-pitched, best heard with the stethoscope bell at the apex. It occurs just before S1 (da-lub-dub). It is always pathological and is caused by forceful atrial contraction against a non-compliant or stiff ventricle. An S4 is most often heard with left ventricular hypertrophy (due to hypertension, aortic stenosis or hypertrophic cardiomyopathy). It cannot occur when there is atrial fibrillation.
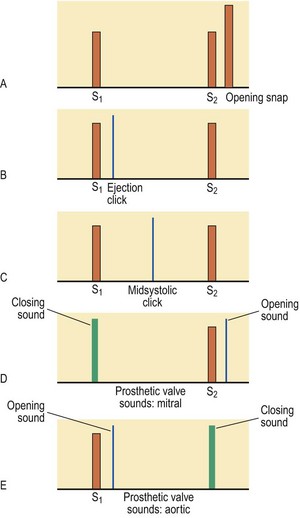
Added sounds: An opening snap is commonly heard in mitral (rarely tricuspid) stenosis. It results from sudden opening of a stenosed valve and occurs early in diastole, just after the S2 (Fig. 6.26A). It is best heard by the diaphragm at the apex.
Ejection clicks are high-pitched sounds best heard by the diaphragm. They occur early in systole just after the S1, in patients with congenital pulmonary or aortic stenosis (Fig. 6.26B). The mechanism is similar to that of an opening snap. Ejection clicks do not occur in calcific aortic stenosis because the cusps are rigid.
Mid-systolic clicks are high-pitched and best heard at the apex by the diaphragm. They occur in mitral valve prolapse (Fig. 6.26C) and may be associated with a late systolic murmur.
Mechanical heart valves can make a sound when they close or open. The closure sound is normally louder, especially with modern valves. The sounds are high-pitched, ‘metallic’ and often palpable, and may be heard even without a stethoscope. A mechanical mitral valve replacement makes a metallic S1 and a sound like a loud opening snap (Fig. 6.26D). Mechanical aortic valves have a loud, metallic S2 and an opening sound like an ejection click (Fig. 6.26E). They are normally associated with a flow murmur.
Pericardial rub (friction rub) is a coarse scratching sound, often with systolic and diastolic components. It is best heard using the diaphragm with the patient holding his breath in expiration. It may be audible over any part of the precordium but is often localised. It is most often heard in acute viral pericarditis and sometimes 24–72 hours after myocardial infarction. Pericardial rubs vary in intensity over time, and with the position of the patient.
A pleuro-pericardial rub is a similar sound that occurs in time with the cardiac cycle but is also influenced by respiration and is pleural in origin. Occasionally a ‘crunching’ noise can be heard caused by gas in the pericardium (pneumo-pericardium).
Murmurs: Heart murmurs are produced by turbulent flow across an abnormal valve, septal defect or outflow obstruction. ‘Innocent’ murmurs caused by increased volume or velocity of flow through a normal valve occur when stroke volume is increased, e.g. during pregnancy, in athletes with resting bradycardia or children with fever.
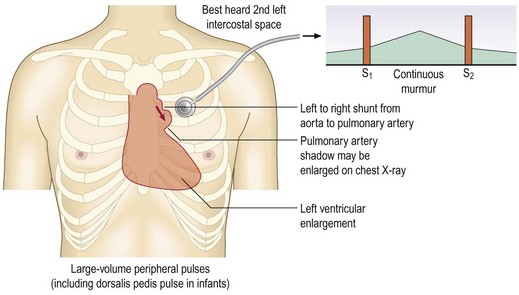
Systolic murmurs: Ejection systolic murmurs are caused by increased stroke volume (flow murmur), or stenosis of the aortic or pulmonary valve (Box 6.31). An ejection murmur is also a feature of hypertrophic cardiomyopathy and is accentuated by exercise. An atrial septal defect is characterised by a pulmonary flow murmur during systole.
The murmur of aortic stenosis is often audible all over the precordium (Fig. 6.28). It is harsh, high-pitched and musical, and radiates to the upper right sternal edge and carotid arteries. It is usually loud and there may be a thrill.
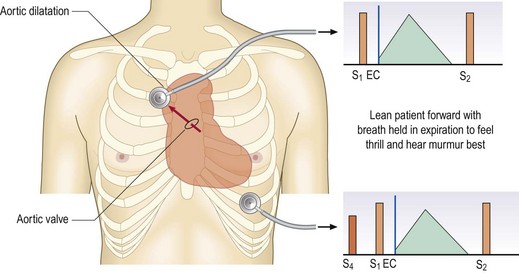
Fig. 6.28 Aortic stenosis.
There is a systolic pressure gradient across the stenosed aortic valve. The resultant high-velocity jet (arrow) impinges on the wall of the aorta, and is best heard with the diaphragm in the aortic area. Alternatively, the bell may be placed in the suprasternal notch. The ejection systolic murmur precedes an ejection click (EC). A fourth heart sound may be heard at the apex.
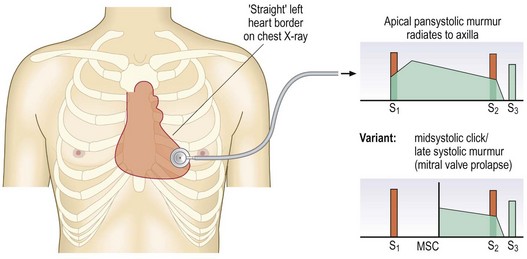
Pansystolic murmurs are usually caused by mitral regurgitation. The murmur is often loud and blowing in character, best heard at the apex and radiating to the axilla. With mitral valve prolapse, regurgitation begins in mid-systole, producing a late systolic murmur (Fig. 6.27). The murmur of tricuspid regurgitation is heard at the lower left sternal edge; if significant, it is associated with a ‘v’ wave in the JVP and a pulsatile liver.
Ventricular septal defect also causes a pansystolic murmur. Small congenital defects produce a loud murmur audible at the left sternal border, radiating to the right sternal border and often associated with a thrill. Rupture of the interventricular septum can complicate myocardial infarction, producing a harsh pansystolic murmur. Other murmurs heard after myocardial infarction include acute mitral regurgitation due to papillary muscle rupture, functional mitral regurgitation caused by left ventricular dilatation and a pericardial rub.
Early diastolic murmurs: The term ‘early diastolic murmur’ is misleading; usually the murmur lasts throughout diastole, but is loudest in early diastole. It is typically caused by aortic regurgitation (Fig. 6.29), and is best heard at the left sternal edge with the patient leaning forward holding the breath in expiration. In general the duration of the aortic regurgitation murmur is inversely proportional to lesion severity. Since the regurgitant blood volume must be ejected during the subsequent systole, significant aortic regurgitation leads to increased stroke volume and is almost always associated with a systolic flow murmur.
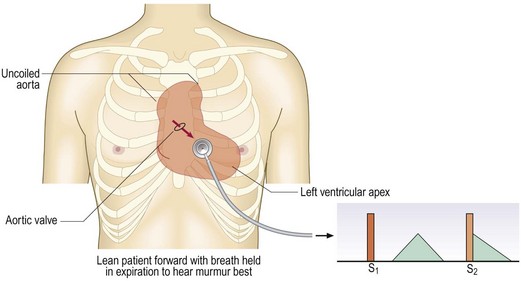
Fig. 6.29 Aortic regurgitation.
The pulse pressure is usually increased; the jet from the aortic valve is directed inferiorly towards the left ventricular outflow tract (arrow) during diastole, producing a high-pitched early diastolic murmur, best heard with the diaphragm. An associated systolic murmur is common because of the increased flow through the aortic valve in systole.
Pulmonary regurgitation is uncommon. It may be caused by pulmonary artery dilatation in pulmonary hypertension (Graham Steell murmur) or a congenital defect of the pulmonary valve.
Mid-diastolic murmurs: A mid-diastolic murmur is usually caused by mitral stenosis. This is a low-pitched, rumbling sound which may follow an opening snap (Fig. 6.30). It is best heard with the stethoscope bell at the apex with the patient rolled to the left side. The murmur is accentuated by exercise. The cadence sounds like ‘lup-ta-ta-rru’; ‘lup’ is the loud S1, ‘ta-ta’ the S2, and opening snap and ‘rru’ the mid-diastolic murmur. If the patient is in sinus rhythm, left atrial contraction increases the blood flow across the stenosed valve, leading to presystolic accentuation of the murmur. The murmur of tricuspid stenosis is similar but rare.
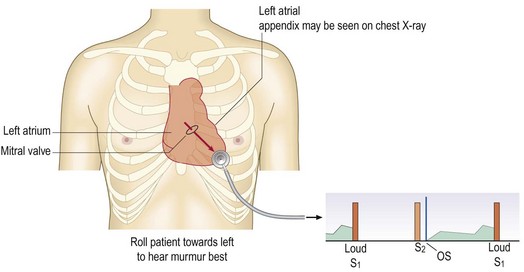
Fig. 6.30 Mitral stenosis.
There is a pressure gradient across the mitral valve; in this example it continues throughout diastole. This causes a sharp movement of the tethered anterior cusp of the mitral valve at the time when the flow commences, and an opening snap (OS) results. The jet through the stenosed valve (arrow) strikes the endocardium at the cardiac apex.
An Austin Flint murmur is a mid-diastolic murmur that accompanies aortic regurgitation. It is caused by the regurgitant jet striking the anterior leaflet of the mitral valve, restricting inflow to the left ventricle.
Continuous murmurs: Continuous murmurs are rare in adults. The most common cause is a patent ductus arteriosus. In the fetus this connects the upper descending aorta and pulmonary artery and normally closes just after birth. The murmur is best heard at the upper left sternal border and radiates over the left scapula. Its continuous character is ‘machinery-like’ (Fig. 6.31).
Putting it all together
Auscultation remains an important clinical skill despite the ready availability of echocardiography. You must be able to detect abnormal signs to prompt appropriate investigation. Auscultatory signs, e.g. S3 or S4 and pericardial friction rubs, have no direct equivalent on echocardiography but are diagnostically important. Some patients, especially those with rheumatic heart disease, have multiple heart valve defects, and the interpretation of more subtle physical signs is important. For example, a patient with mixed mitral stenosis and regurgitation will probably have dominant stenosis if the S1 is loud, but dominant regurgitation if there is an S3.
Investigations
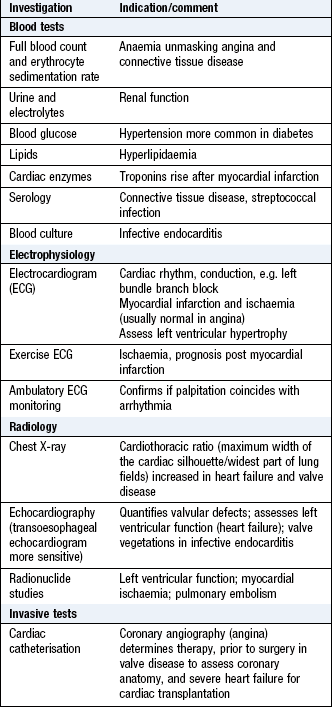
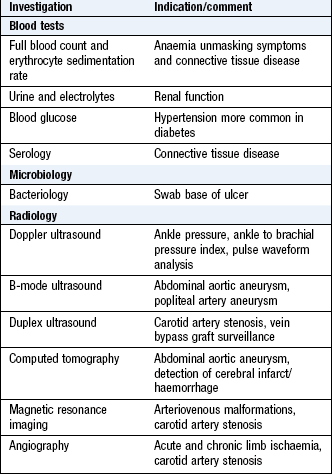
See Box 6.32.
Electrocardiography (ECG)
The standard 12-lead ECG (Fig. 6.32) uses recordings made from six precordial electrodes (V1–V6) and six different recordings from the limb electrodes (left arm, right arm and left leg). The right leg electrode is used as a reference.
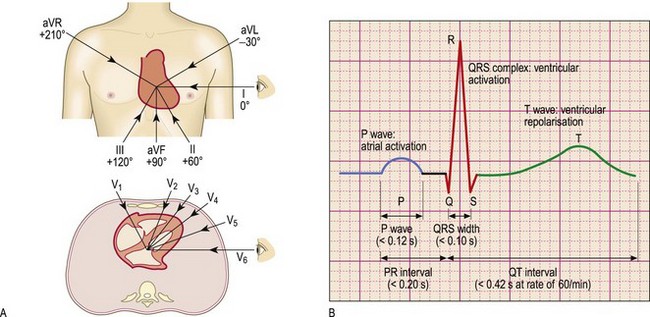
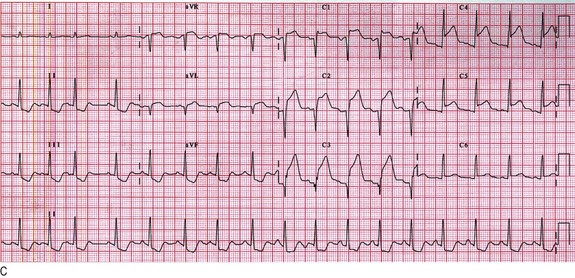
Fig. 6.32 Electrocardiography.
(A) Diagram to show the directions from which the 12 standard leads ‘look at the heart’. The transverse section is viewed from below like a computed tomography scan. (B) Normal PQRST complex. (C) Acute anterior myocardial infarction. Note ST elevation in leads V1–V6 and aVL, and ‘reciprocal’ ST depression in leads II, III and aVF.
Ambulatory ECG monitoring: This is a continuous ECG recording that lasts 24–48 hours and is read by computer. Patient-activated recorders capture occasional arrhythmias and are activated only when symptoms occur (Fig. 6.33).
Ambulatory BP monitoring: A portable device is worn by the patient at home: this device takes at least two BP measurements per hour during the person’s usual waking hours. The average value of at least 14 measurements is used to confirm a diagnosis of hypertension.
Chest X-ray: An enlarged heart, as judged by the cardiothoracic ratio (Fig. 7.22), is common in valvular heart disease and heart failure. In heart failure this is often accompanied by distension of the upper lobe pulmonary veins, diffuse shadowing within the lungs due to pulmonary oedema and Kerley B lines (horizontal engorged lymphatics at the periphery of the lower lobes) (Fig. 6.34A). A widened mediastinum may indicate a thoracic aneurysm.
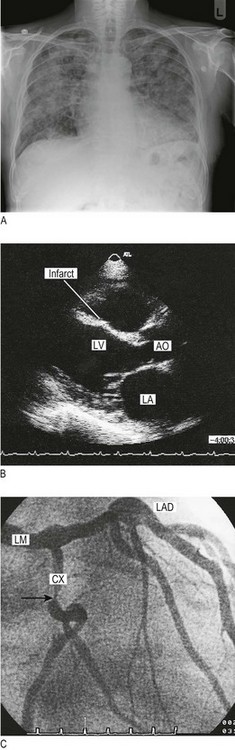
Fig. 6.34 Cardiovascular imaging.
(A) Chest X-ray in heart failure. This shows cardiomegaly with patchy alveolar shadowing of pulmonary oedema and Kerley B lines (engorged lymphatics) at the periphery of both lungs. (B) Transthoracic echocardiogram in parasternal long-axis view. This shows thinning of the interventricular septum, which has an irregular shape and bright echoes indicating fibrous scarring. This is the site of an old infarct. LA, left atrium; LV, left ventricle; AO, aortic root. (C) Coronary angiography. The arrow indicates a severe discrete stenosis in the circumflex coronary artery. LM, left main; LAD, left anterior descending; CX, circumflex.
Echocardiography: Echocardiography uses high-frequency sound waves to evaluate valve abnormalities, left ventricular function and blood flow (Doppler echocardiography). Most scans are performed through the anterior chest wall (transthoracic) (Fig. 6.34B). Transoesophageal echocardiography requires sedation, but gives high resolution of posterior structures, e.g. left atrium, tricuspid valve and descending aorta.
Radionuclide studies: Technetium-99 is injected intravenously and detected using a gamma camera to assess left ventricular function. Thallium and sesta-MIBI are taken up by myocardial cells and indicate myocardial perfusion at rest and exercise.
Cardiac catheterisation: A fine catheter is introduced under local anaesthetic via a peripheral artery (usually the brachial or femoral) and advanced to the heart under X-ray guidance. Although measurements of intracardiac pressures and therefore estimates of valvular and cardiac function are possible, the primary application of this technique is coronary arterial imaging, using contrast medium. This is performed to inform revascularisation, either by coronary angioplasty or bypass grafting (Fig. 6.34C).
Computed tomography (CT) and magnetic resonance imaging (MRI): CT, with its superior temporal resolution of the coronary arteries, is particularly useful to investigate symptomatic patients at low-intermediate risk of coronary artery disease. It can also reduce the need for invasive investigation in patients, with a low probability of occlusive coronary disease, awaiting valve surgery. MRI provides superior tissue resolution and is the imaging modality of choice for investigating heart muscle disease (cardiomyopathy).
Peripheral vascular system
Clinical presentation
Approximately 20% of people aged >60 years in developed countries have PAD but only a quarter of these are symptomatic. The underlying pathology is usually atherosclerosis (hardening of the arteries) affecting large and medium-sized vessels.
Identifying patients with PAD is important for the following reasons:
• PAD, even if asymptomatic, is a powerful marker for premature vascular death
• The first manifestation of PAD may be a life- or limb-threatening complication, e.g. stroke, acute limb ischaemia or ruptured AAA
• Modifying vascular risk factors dramatically improves outcomes
• PAD may affect medical and surgical treatment for other conditions, e.g. prescription of a beta-blocker may precipitate intermittent claudication.
PAD affects the legs eight times more commonly than the arms for the following reasons:
• The arterial supply to the legs is less well developed in relation to the muscle mass
• The lower limb is more frequently affected by atherosclerosis.
There are four stages of lower limb lack of blood supply (ischaemia) (Box 6.33).
Asymptomatic ischaemia: Haemodynamically significant lower limb ischaemia is defined as an ankle to brachial pressure index (ABPI) <0.9 at rest. Most of these patients are asymptomatic, either because they choose not to walk very far, or because their exercise tolerance is limited by other comorbidity. Although asymptomatic, these patients are at high risk of ‘vascular’ complications and should be assessed and treated medically as if they have intermittent claudication.
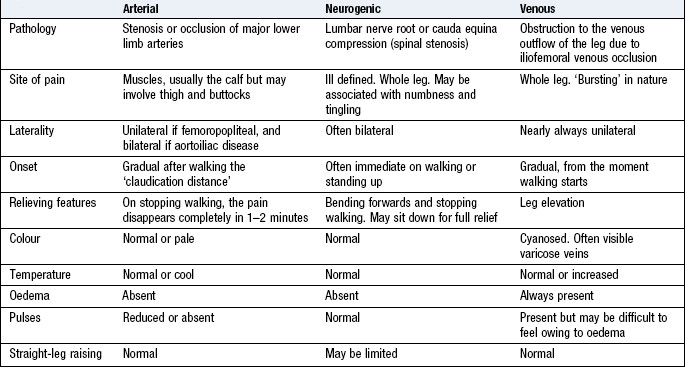
Intermittent claudication: Intermittent claudication is pain felt in the legs on walking due to arterial insufficiency and is the most common symptom of PAD. The pain typically occurs in the calf secondary to femoropopliteal disease but may be felt in the thigh and/or buttock in proximal (aorto-iliac) obstruction. Patients describe tightness or ‘cramp-like’ pain which develops after a relatively constant distance; the distance is often shorter if walking uphill, in the cold and after meals. The pain disappears completely within a few minutes of rest but recurs on walking. The ‘claudication distance’ is how far patients say they can walk before the pain stops them from walking.
There are two other types of claudication
• Neurogenic claudication is due to neurological and musculoskeletal disorders of the lumbar spine
• Venous claudication is due to venous outflow obstruction from the leg, following extensive DVT.
Neurogenic and venous claudication are much less common than arterial claudication, and can be distinguished on history and examination (Box 6.34).
Night/rest pain: The patient goes to bed, falls asleep, but is then woken 1–2 hours later with severe pain in the foot, usually in the instep. The pain is due to poor perfusion resulting from the loss of the beneficial effects of gravity on lying down and the reduction in heart rate, BP and cardiac output that occurs when sleeping. Patients often obtain relief by hanging the leg out of bed or by getting up and walking around. However, on return to bed, the pain recurs and patients often choose to sleep in a chair. This leads to dependent oedema, increased interstitial tissue pressure, a further reduction in tissue perfusion and ultimately a worsening of the pain.
Rest (night) pain indicates severe, multilevel lower limb PAD and is a ‘red flag’ symptom that mandates urgent referral to a vascular surgeon as failure to revascularise the leg usually leads to the development of critical limb ischaemia with tissue loss (gangrene, ulceration) and amputation.
In diabetic patients it may be difficult to differentiate between rest pain and diabetic neuropathy as both may be worse at night. Neuropathic pain is not usually confined to the foot, is associated with burning and tingling, is not relieved by dependency and is associated with dysaesthesia (pain or uncomfortable sensations sometimes described as burning, tingling or numbness). Many patients cannot even bear the pressure of bedclothes on their feet.
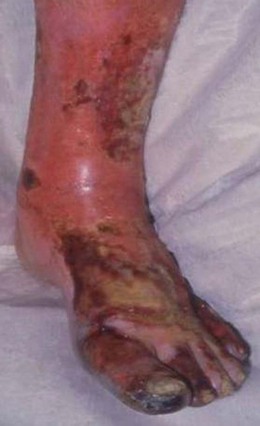
Tissue loss (ulceration and/or gangrene): In patients with severe lower limb PAD, even trivial injuries to the feet fail to heal. This allows bacteria to enter, leading to gangrene and/or ulceration. This usually progresses rapidly and, without revascularisation, often leads quickly to amputation and/or death.
Signs of lower limb PAD: Ischaemic signs include absence of hair, thin skin and brittle nails (Box 6.34). The presence of foot pulses does not completely exclude significant lower limb PAD but they are almost always diminished or absent. If the history is convincing but pulses are felt, ask the patient to walk until the claudication pain stops him and then recheck the pulses; if they have disappeared then PAD is very likely.
Patients with critical limb ischaemia (rest pain, tissue loss) typically have an ankle BP <50 mmHg and a positive Buerger’s test.
Acute limb ischaemia: The classical features of acute limb ischaemia are the ‘six Ps’ (Box 6.35). Paralysis (inability to wiggle the toes/fingers) and paraesthesia (loss of light touch sensation over the forefoot/dorsum of the hand) are the most important and indicate severe ischaemia affecting nerve function. Muscle tenderness is a grave sign indicating impending muscle infarction. A limb with these features will usually become irreversibly damaged unless the circulation is restored within a few hours.
The commonest causes of acute limb ischaemia are:
• Thromboembolism: usually from the left atrium in association with atrial fibrillation
• Thrombosis in situ: thrombotic occlusion of an already narrowed atherosclerotic arterial segment (Box 6.36).
Acute arterial occlusion produces intense spasm in the arterial tree distal to the blockage. The limb appears ‘marble white’. Over a few hours, the spasm relaxes and the skin microcirculation fills with deoxygenated blood, leading to light blue or purple mottling, which has a fine reticular pattern and blanches on pressure. As ischaemia progresses, blood coalesces in the skin, producing a coarser pattern of mottling which is dark purple, almost black, and does not blanch. Finally, large patches of fixed staining lead to blistering and liquefaction. Fixed mottling of an anaesthetic, paralysed limb, with muscle rigidity and turgor, indicates irreversible ischaemia; amputation or end-of-life care is the only option (Fig. 6.36).
Compartment syndrome occurs where there is increased pressure within the fascial compartments of the limb, most commonly the calf, which compromises perfusion and viability of muscle and nerves. The two commonest causes are lower trauma, e.g. fractured tibia, and reperfusion following treatment of acute lower limb ischaemia. Failure to recognise and treat compartment syndrome may require limb amputation. The key symptom is severe pain often unrelieved by opioids and exacerbated by active or passive movement. Peripheral pulses are usually present.
Stroke
Stroke is a focal central neurological deficit of vascular cause. Approximately 80% of strokes are ischaemic rather than haemorrhagic. Transient ischaemic attack (TIA) describes a stroke in which symptoms resolve within 24 hours. The term ‘stroke’ is reserved for those events in which symptoms last for more than 24 hours.
Carotid artery territory (anterior circulation): Up to half of all strokes and TIAs are due to embolism from an atheromatous plaque at the origin of the internal carotid artery. Clinical features vary according to the cerebral area involved but can include motor deficit, visual field defect, e.g. homonymous hemianopia (Fig. 12.3), or difficulty with speech (dysphasia, p. 250) (Box 6.37).
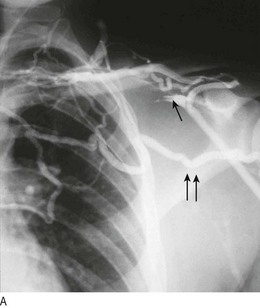
Vertebrobasilar artery territory (posterior circulation): TIAs and strokes in this territory cause giddiness, collapse with or without loss of consciousness, transient occipital blindness or complete loss of vision in both eyes (Ch. 11). Subclavian artery stenosis or occlusion proximal to the origin of the vertebral artery may cause vertebrobasilar symptoms as part of the ‘subclavian steal’ syndrome. This happens when the arm is exercised. The increased blood supply requirement in the arm is met by blood travelling up the carotid arteries and then, via the circle of Willis (Fig. 11.29), down the vertebral artery into the arm, so ‘stealing’ blood from the posterior cerebral circulation. Signs of this include asymmetry of the pulses and BP in the arms, sometimes with a bruit over the subclavian artery in the supraclavicular fossa.
Abdominal symptoms
Mesenteric angina: Because of the rich collateral circulation, usually two of the three major visceral arteries (coeliac axis, superior and inferior mesenteric arteries) must be critically stenosed or occluded before symptoms and signs of chronic mesenteric arterial insufficiency occur. Severe central abdominal pain typically develops 10–15 minutes after eating. The patient becomes scared of eating and significant weight loss is a universal finding. Diarrhoea may occur and visceral ischaemia may mimic a whole range of gastrointestinal pathologies. The patient may have had numerous investigations, even laparotomy, before the diagnosis is made and confirmed by angiography. Any patient suspected of visceral ischaemia should undergo urgent angiography.
Acute mesenteric ischaemia is a surgical emergency. The patient presents with severe abdominal pain, shock, bloody diarrhoea and profound metabolic acidosis. Rarely, renal angle pain occurs from renal infarction or ischaemia, and is associated with microscopic or macroscopic haematuria.
Abdominal aortic aneurysm: AAA is an abnormal dilatation of the aorta (Fig. 6.37) and is present in 5% of men aged >65 years. The main risk factors are smoking and hypertension; there is also a familial/genetic element to the disease. AAA is three times more common in men than women. Most patients are asymptomatic until the aneurysm ruptures, although they may present with abdominal and/or back pain or an awareness of abdominal pulsation.
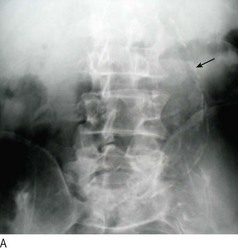
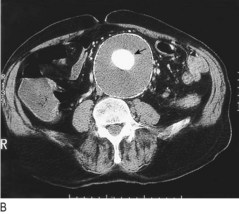
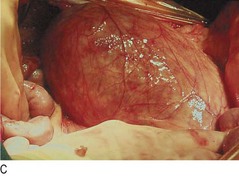
Fig. 6.37 Abdominal aortic aneurysm.
(A) Abdominal X-ray showing calcification (arrow). (B) Computed tomography of the abdomen showing an abdominal aortic aneurysm (arrow). (C) At laparotomy the aorta is seen to be grossly and irregularly dilated.
Clinical examination is unreliable in establishing the presence or size of an AAA; if in any doubt, obtain an ultrasound scan of the aorta. In the UK there is now an ultrasound-based AAA screening programme for men as they reach their 65th birthday.
A ruptured AAA can be difficult to diagnose because many patients do not have the classical features of abdominal and/or back pain, pulsatile abdominal mass and shock (hypotension). The most common misdiagnosis is renal colic (a man, >60 years, presenting with ‘renal colic’ has a ruptured AAA until proved otherwise). If there is any suspicion of a ruptured AAA speak to a vascular surgeon straight away who will probably request an immediate contrast-enhanced CT of the abdomen (if the patient is cardiovascularly stable).
Athero-embolism from an AAA can cause ‘blue toe syndrome’, characterised by purple discoloration of the toes and forefoot of both feet. There is usually a full set of pedal pulses.
Vasospastic symptoms: Raynaud’s phenomenon is digital ischaemia induced by cold and emotion and has three phases (Fig. 6.38):
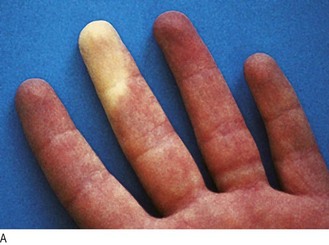
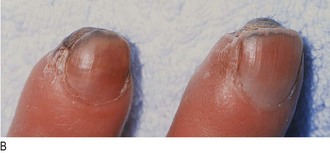
Fig. 6.38 Raynaud’s syndrome.
(A) The acute phase, showing severe blanching of the tip of one finger. (B) Primary Raynaud’s syndrome occasionally progresses to fingertip ulceration or even gangrene.
• Pallor: due to digital artery spasm and/or obstruction
• Cyanosis: due to deoxygenation of static venous blood (this phase may be absent)
Raynaud’s phenomenon may be primary (Raynaud’s disease) and due to idiopathic digital artery vasospasm, or secondary (Raynaud’s syndrome) (Box 6.38).
Patients >40 years old presenting with unilateral Raynaud’s phenomenon have underlying PAD unless proven otherwise, especially if they have risk factors (smoking, diabetes).
The history
Ask about risk factors for atheroma (smoking, hypercholesterolaemia, hypertension, diabetes mellitus) and any family history of premature arterial disease. Specifically ask about diabetes because it is associated with the early development and rapid progression of widespread atheroma.
The impact of intermittent claudication relates to the patient’s age and lifestyle. A postman/postwoman who can walk only 400 metres has a serious problem, but an elderly person who simply wants to cross the road to the shops may cope well. Rather than focusing upon absolute distances, ask specific questions like:
• Can you walk to the clinic from the bus stop or car park without stopping?
Ask about the patient’s other medical conditions. There is little point in subjecting patients with intermittent claudication to the risks of vascular surgery, only to find that they are then equally limited by osteoarthritis of the hip, angina or severe breathlessness.
Male patients with buttock (gluteal) intermittent claudication due to internal iliac disease have erectile dysfunction. Ask about sexual function as many men and their partners are extremely concerned by erectile dysfunction yet too embarrassed to mention it.
The physical examination
Follow the routine described for the heart, looking for evidence of anaemia or cyanosis, signs of heart failure, and direct or indirect evidence of PAD (Box 6.39). Then perform a detailed examination of the arterial pulses. Abnormally prominent pulsation in the neck of an elderly person is rarely of clinical significance and is normally caused by tortuous arteries rather than a carotid aneurysm or carotid body tumour. However, if in any doubt, arrange for a duplex ultrasound scan.
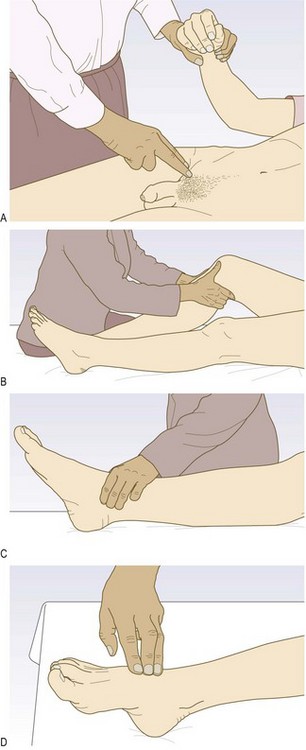
Ankle to brachial pressure index: Assessing pulse status can be unreliable in patients with obesity or oedema. Routinely measure ABPI in all patients with difficulty palpating lower limb pulses or where PAD is suspected on the basis of history.
• Use a hand-held Doppler and a sphygmomanometer
• Hold the probe over the posterior tibial artery
• Inflate a BP cuff round the ankle
• Note the pressure when Doppler signal disappears. This is the systolic pressure in that artery as it passes under the cuff
• Repeat holding the probe over dorsalis pedis, and then the perforating peroneal (Box 6.13)
• Measure the brachial BP in both arms, holding the Doppler probe over the brachial artery at the elbow of the radial artery at the wrist.
Abnormal findings: Typical values in intermittent claudication and critical limb ischaemia are <0.9 and <0.4 respectively. Absolute values may be less informative than the trend over time.
Patients with lower limb PAD, particularly those with diabetes mellitus, often have incompressible, calcified crural arteries with falsely elevated pedal pressures and ABPI. Use a Doppler ultrasound probe to detect the foot arteries while elevating the foot. The Doppler signal disappears at a height (in cm) above the bed that approximates to the perfusion pressure (in mmHg).
Choose further tests to provide the most information at the least risk to the patient and at least expense. In most situations duplex Doppler ultrasound has replaced angiography as the first-line investigation of choice (Box 6.40).
Peripheral venous system
Whereas venous return from the head and neck is passive, that from the legs must be actively pumped back up to the heart against gravity. Pressure on the sole of the foot on walking, together with contraction of muscles in the calf (the ‘calf muscle pump’), and, to a lesser extent in the thighs and buttocks, forces venous blood back up deep (90%) and superficial (10%) veins. Backward flow (reflux) is prevented by valves which divide the long column of blood from the foot to the right atrium into a series of short low-pressure segments. As a result, the ‘ambulatory venous pressure’ in the feet in health is usually <20 mmHg. The great majority of lower limb venous symptoms and signs are due to failure of the muscle pump and/or valves and the resulting ‘ambulatory venous hypertension’.
Deep veins follow the course of the main arteries; are often paired; and may be affected by primary or postthrombotic (following DVT) valvular insufficiency. DVT often leads to deep venous obstruction as well as reflux (leading to the symptoms and signs of the postthrombotic syndrome).
Superficial veins may also be affected by primary valvular failure and by reflux following superficial thrombophlebitis.
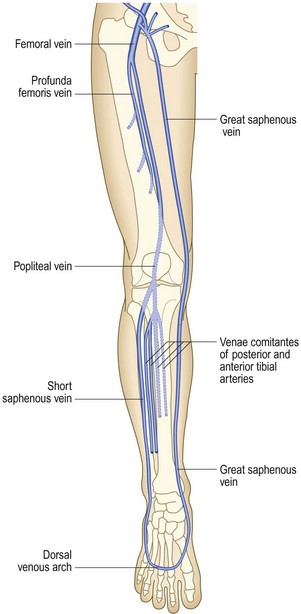
The great (long) saphenous vein passes anterior to the medial malleolus at the ankle, up the medial aspect of the calf to behind the knee, then up the medial aspect of the thigh to join the common femoral vein in the groin at the saphenofemoral junction (Fig. 6.41).
The lesser (short) saphenous vein passes behind the lateral malleolus at the ankle and up the posterior aspect of the calf. It commonly joins the popliteal vein at the saphenopopliteal junction, which usually lies 2 cm above the posterior knee crease.
There are numerous intercommunications between the long and short saphenous, and between the deep and superficial venous (via perforating or communicating veins) systems; and the venous anatomy of the leg is highly variable.
Clinical presentation
Lower limb venous disease presents in four ways:
The severity of symptoms and signs may bear little relationship to the severity of the underlying pathology and the physical signs. Life-threatening DVT may be asymptomatic, while apparently trivial varicose veins may be associated with significant complaints.
Pain
Patients with uncomplicated varicose (dilated, tortuous, superficial) veins often complain of aching leg discomfort, itching and a feeling of swelling. Symptoms are aggravated by prolonged standing and are often worse towards the end of the day. Once established, DVT causes pain and tenderness in the affected part (usually the calf). Superficial thrombophlebitis produces a red, painful area overlying the vein involved. Varicose ulceration may be surprisingly painless; if it is painful, this may be relieved by limb elevation (but exclude coexisting arterial disease) (Box 6.41). Bandaging for a leg ulcer is contraindicated unless there is documented evidence of adequate arterial circulation. Do this by feeling the pulses or by measuring the ABPI.
Swelling
Swelling (or oedema), or a ‘feeling of swelling’, may be associated with lower limb venous disease.
Discoloration
Chronic venous insufficiency is associated with lipodermatosclerosis, which results from the deposition of haemosiderin (from the breakdown of extravasated blood) in the skin. Lipodermatosclerosis varies in colour from deep blue/black to purple or bright red and usually affects the medial aspect of the lower third of the leg, although it may be lateral if superficial reflux predominates in the lesser saphenous vein.
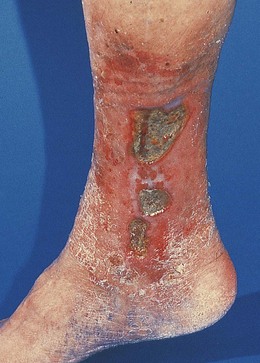
Chronic venous ulceration
In developed countries about 70–80% of leg ulcers are due primarily to venous disease. Other causes include pyoderma gangrenosum, syphilis, tuberculosis, leprosy, sickle cell disease and tropical conditions. Chronic venous ulceration usually affects the medial aspect, is shallow; is pink (granulation tissue) or yellow/green (slough); has an irregular margin; and is always associated with other skin changes of chronic venous insufficiency (varicose eczema, lipodermatosclerosis) (Fig. 6.42).
Deep vein thrombosis
The leg: The clinical features of DVT depend upon its site, extent and whether it is occlusive or not (Box 6.42). The so-called ‘classical’ features of DVT relate to well-established occlusive thrombus. Most patients who die from pulmonary embolism have non-occlusive thrombosis and the leg is normal on clinical examination. Non-occlusive DVT poses the greatest threat of pulmonary embolism as the clot lies within a flowing stream of venous blood, is more likely to propagate and has not yet induced an inflammatory response in the vein wall to anchor it in place. Risk factors for DVT are listed in Box 6.43. Perform an urgent duplex Doppler ultrasound scan of the leg in any patient with a suspected DVT.
The arm: Axillary subclavian vein thrombosis can occur as a result of repetitive trauma at the thoracic outlet due to vigorous, repetitive exercise. Upper limb DVT may also complicate indwelling subclavian/jugular venous catheters. Symptoms include arm swelling and discomfort, often exacerbated by activity, especially when holding the arm overhead.
The arm is swollen and the skin is cyanosed and mottled, especially when dependent. Look for superficial distended veins (acting as collaterals) in the upper arm, over the shoulder region and on the anterior chest wall (Fig. 6.43).
Superficial venous thrombophlebitis
This condition affects up to 10% of patients with severe varicose veins and is more common during pregnancy. Recurrent superficial venous thrombophlebitis, especially affecting different areas sequentially and non-varicose veins, may be associated with underlying malignancy. It may propagate into the deep system, leading to DVT and pulmonary embolism.