The gastrointestinal system
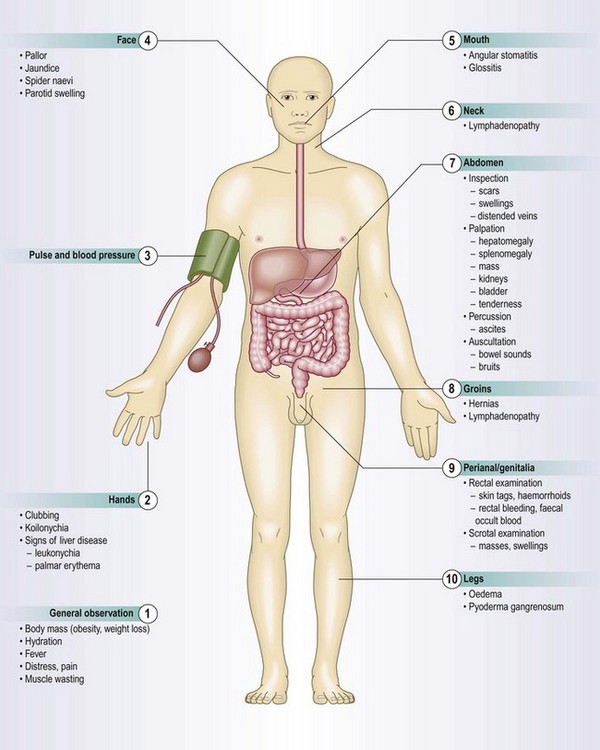
Gastrointestinal examination
Anatomy
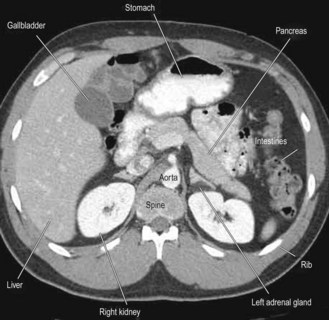
The gastrointestinal system comprises the alimentary tract plus the liver and biliary system (including the gallbladder), the pancreas and the spleen. The alimentary tract extends from the mouth to the anus and includes the oesophagus, stomach, small intestine (which is also called the small bowel, and comprises duodenum, jejunum and ileum), colon (also called the large intestine, or large bowel) and rectum (Figs 8.1–8.2 and Box 8.1).
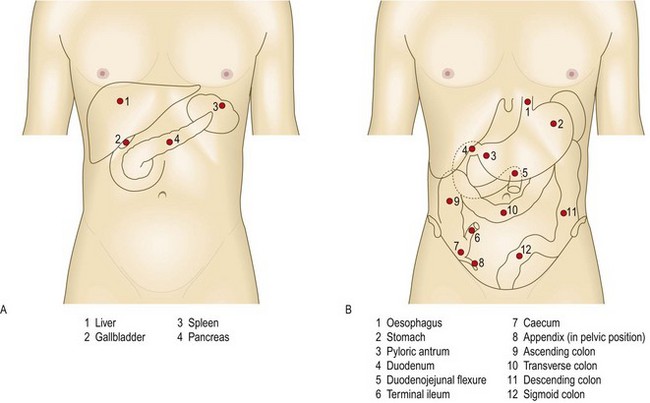
Fig. 8.1 Surface anatomy.
(A) Surface markings of non-alimentary tract abdominal viscera. (B) Surface markings of the alimentary tract.
The abdomen
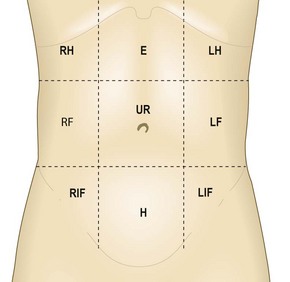
The abdomen can be divided into nine regions by the intersection of imaginary planes: two horizontal and two vertical (Fig. 8.3).
Symptoms and definitions
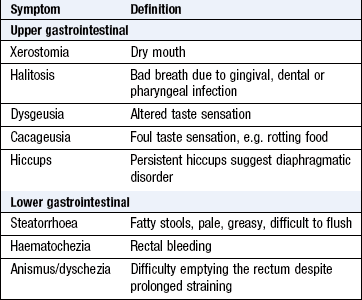
See Box 8.2.
Anorexia and weight loss
Anorexia is loss of appetite and/or a lack of interest in food. Weight loss is usually the result of reduced energy intake, not increased energy expenditure (Box 8.3). Reduced energy intake arises from dieting, loss of appetite or malabsorption and malnutrition. Energy loss occurs in uncontrolled diabetes mellitus due to marked glycosuria. Increased energy expenditure occurs in hyperthyroidism, fever or the adoption of a more energetic lifestyle. A net calorie deficit of 1000 kcal/day produces a weight loss of approximately 1 kg/week (7000 kcal ≈ 1 kg of fat). Greater weight loss during the initial stages of energy restriction arises from salt and water loss and depletion of hepatic glycogen stores, and not from fat loss. Rapid weight loss over days suggests loss of body fluid as a result of vomiting, diarrhoea or diuretic therapy (1 litre of water = 1 kg).
Weight loss, in isolation, is rarely associated with serious organic disease and loss of <3 kg in the previous 6 months is rarely significant. It does not specifically indicate gastrointestinal disease but is common in many upper gastrointestinal disorders, including malignancy and liver disease. Weight loss with amenorrhoea in an adolescent female may suggest anorexia nervosa but menstrual irregularity is common in women who lose weight from any cause.
Pain
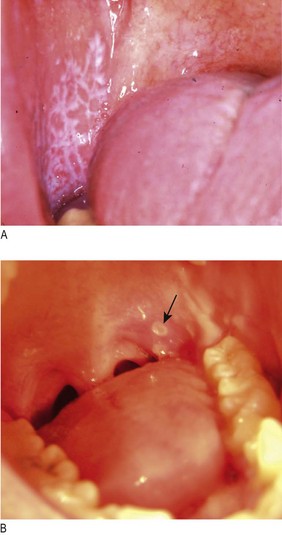
Painful mouth: There are many causes of sore lips, tongue or buccal mucosa, including iron, folate, vitamin B12 or vitamin C deficiencies, dermatological disorders, e.g. lichen planus, chemotherapy, isolated aphthous ulcers and infective stomatitis (Box 8.4 and Fig. 8.4). Inflammatory bowel disease and gluten enteropathy are associated with mouth ulcers.
Heartburn and reflux: Heartburn is a hot, burning retrosternal discomfort which radiates upwards. When heartburn is the principal symptom, gastro-oesophageal reflux disease (GORD) is the likeliest diagnosis. A sour taste in the mouth from regurgitating gastric acid is called reflux. Differentiate heartburn from cardiac chest pain by its burning quality, upward radiation, association with acid reflux and its occurrence on lying flat or bending forward. Waterbrash is the sudden appearance of fluid in the mouth due to reflex salivation as a result of GORD or, rarely, peptic ulcer disease.
Dyspepsia: Dyspepsia is pain or discomfort centred in the upper abdomen. In contrast, ‘indigestion’ is a term commonly used for ill-defined symptoms from the upper gastrointestinal tract. Dyspepsia affects up to 80% of the population at some time. In the majority, no identifiable cause is found (functional dyspepsia). Clusters of symptoms are used to classify dyspepsia:
• reflux-like dyspepsia (heartburn-predominant dyspepsia)
• ulcer-like dyspepsia (epigastric pain relieved by food or antacids)
• dysmotility-like dyspepsia (nausea, belching, bloating and premature satiety).
There is considerable overlap and it is impossible to diagnose functional dyspepsia on history alone, without investigation. Dyspepsia that is worse with an empty stomach and eased by eating is the classical symptom of peptic ulceration. The patient may indicate a single localised point in the epigastrium (pointing sign), and complain of nausea and abdominal fullness which is worse after meals with a high spice or fat content. ‘Fat intolerance’ is common in all causes of dyspepsia, including gallbladder disease.
Odynophagia: Odynophagia is pain on swallowing, often precipitated by drinking hot liquids. It can be present with or without dysphagia and may indicate active oesophageal ulceration from peptic oesophagitis or oesophageal candidiasis. It implies intact mucosal sensation, making oesophageal cancer unlikely.
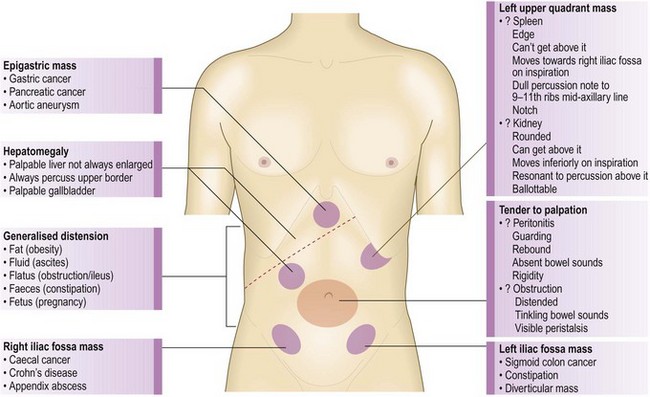
Site: Visceral abdominal pain from distension of hollow organs, mesenteric traction or excessive smooth-muscle contraction is deep and poorly localised in the midline. It is conducted via sympathetic splanchnic nerves. Somatic pain from the parietal peritoneum and abdominal wall is lateralised and localised to the area of inflammation. It is conducted via intercostal (spinal) nerves.
Pain arising from foregut structures (stomach, pancreas, liver and biliary system) is localised above the umbilicus (Fig. 8.5). Central abdominal pain arises from midgut structures, e.g. small bowel and appendix. Lower abdominal pain arises from hindgut structures, e.g. colon. Inflammation may cause localised pain, e.g. left iliac fossa pain due to diverticular disease of the sigmoid colon.
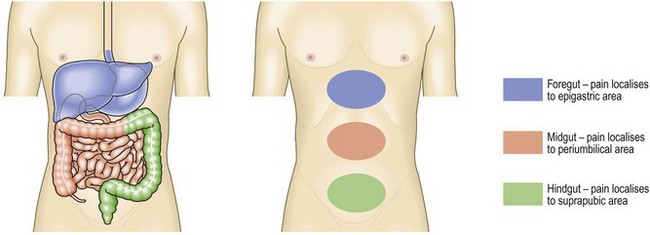
Fig. 8.5 Abdominal pain.
Perception of visceral pain is localised to the epigastric, umbilical or suprapubic region, according to the embryological origin of the affected organ.
Pain from an unpaired structure, such as the pancreas, is midline and radiates through to the back. Pain from paired structures is felt on and radiates to the affected side, e.g. renal colic. Boys with abdominal pain may have torsion of the testis (Fig. 10.51). In women, consider gynaecological causes, e.g. ruptured ovarian cyst, pelvic inflammatory disease, endometriosis or an ectopic pregnancy.
Onset: The sudden onset of severe abdominal pain, rapidly progressing to become generalised and constant, suggests a hollow viscus perforation, a ruptured abdominal aortic aneurysm or mesenteric infarction. Preceding constipation suggests colorectal cancer or diverticular disease as the cause of perforation and prior dyspepsia suggests peptic ulceration. Coexisting peripheral vascular disease, hypertension, heart failure or atrial fibrillation may suggest aortic aneurysm or mesenteric ischaemia.
Development of circulatory failure following the onset of pain suggests intra-abdominal sepsis or bleeding, e.g. ruptured aortic aneurysm or ectopic pregnancy. Torsion of the testis or ovary produces severe acute abdominal pain and nausea. Torsion of the caecum or sigmoid colon (volvulus) presents with sudden abdominal pain associated with acute intestinal obstruction.
Character: Inflammation and obstruction are the principal pathological processes producing acute abdominal pain. Inflammation usually produces constant pain exacerbated by movement or coughing. Colicky pain arises from hollow structures, e.g. small or large bowel obstruction, or the uterus during labour. It lasts for a short period of time (seconds or minutes), eases off and then returns.
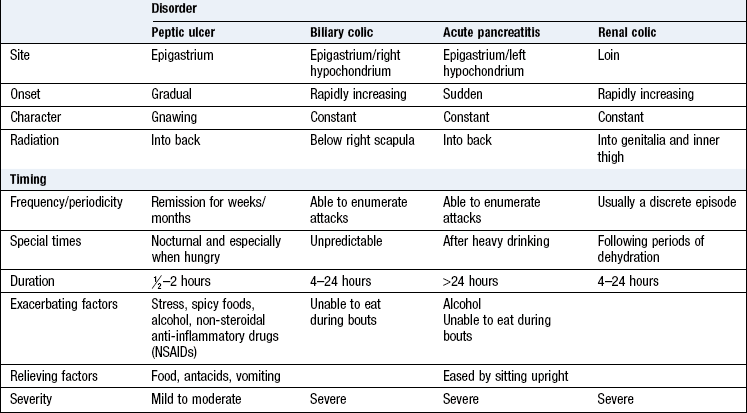
Biliary and renal ‘colic’ are misnamed, as the pain is rarely colicky; pain rapidly increases to a peak intensity and persists over several hours before gradually resolving (Box 8.5).
Biliary or renal colic is usually promptly relieved by parenteral analgesia. Dull, vague and poorly localised pain is more typical of an inflammatory process or low-grade infection, e.g. salpingitis, appendicitis or diverticulitis.
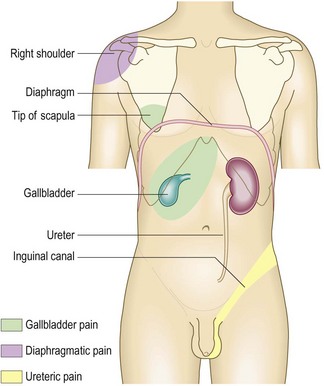
Radiation: Pain radiating from the right hypochondrium to the shoulder or interscapular region may reflect diaphragmatic irritation, e.g. in acute cholecystitis (Fig. 8.6). Pain radiating from the loin to the groin and genitalia is typical of renal colic. Central upper abdominal pain radiating through to the back, partially relieved by sitting forward, is common in pancreatitis. Central abdominal pain, which later shifts into the right iliac fossa, occurs in acute appendicitis (Fig. 8.27). The combination of severe back and abdominal pain may indicate a ruptured or dissecting abdominal aortic aneurysm.
Associated symptoms: Anorexia, nausea and vomiting are common but non-specific symptoms reflecting the nature and severity of pain; they may be absent, even in advanced intra-abdominal disease. Severe vomiting without significant pain suggests gastric outlet or proximal small-bowel obstruction. Faeculent vomiting, of small-bowel contents (not faeces), is a late feature of distal small-bowel or colonic obstruction. In peritonitis, the vomitus is usually small in volume but persistent. Severe vomiting with retching may result in laceration at the gastro-oesophageal junction (Mallory–Weiss tear) or oesophageal rupture (Boerhaave’s syndrome).
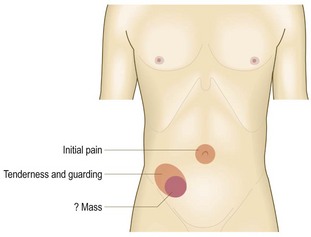
Timing: During the first hour or two after perforation, a ‘silent interval’ may occur when abdominal pain resolves transiently. The initial chemical peritonitis may subside before bacterial peritonitis becomes established. In acute appendicitis, pain is initially periumbilical (visceral pain) and moves to the right iliac fossa when localised inflammation of the parietal peritoneum becomes established (somatic pain). If the appendix ruptures, generalised peritonitis may develop. Occasionally, a localised appendix abscess develops, with a palpable mass and localised pain in the right iliac fossa.
Change in the pattern of symptoms suggests either that the initial diagnosis was wrong, or that complications have developed. In acute small-bowel obstruction, a change from typical intestinal colic to persistent pain with abdominal tenderness suggests intestinal ischaemia, e.g. strangulated hernia, an indication for urgent surgical intervention.
Exacerbating and relieving factors: Pain exacerbated by movement or coughing suggests inflammation. Patients tend to lie still in order not to exacerbate the pain. Patients with colic typically move around or draw their knees up towards the chest during painful spasms. Abdominal pain persisting for hours or days suggests an inflammatory disorder, such as acute appendicitis, cholecystitis or diverticulitis.
Accompanying features: Nausea and vomiting may accompany any very severe pain. Abdominal pain due to irritable bowel syndrome, diverticular disease or colorectal cancer is invariably accompanied by an alteration in bowel habit. Other features such as breathlessness or palpitation suggest non-alimentary causes (Box 8.6).
Dysphagia
Dysphagia (Boxes 8.7 and 8.8) is difficulty swallowing. Always investigate it. Do not confuse dysphagia with early satiety, the inability to complete a full meal because of premature fullness, or globus, the feeling of a lump in the throat. Globus does not interfere with swallowing and is not related to eating.
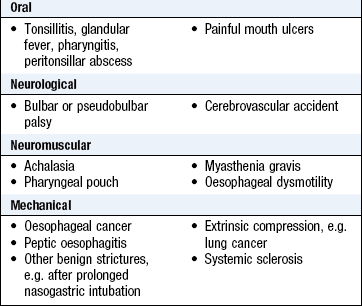
Neurological dysphagia resulting from bulbar or pseudobulbar palsy is worse for liquids than for solids, and may be accompanied by choking, spluttering and fluid regurgitating from the nose.
Neuromuscular dysphagia, or oesophageal dysmotility, presents in middle age, is worse for solids and may be helped by liquids and sitting upright. Achalasia, when the lower oesophageal sphincter fails to relax normally, leads to progressive oesophageal dilatation above the sphincter. Overflow of secretions and food into the respiratory tract may then occur, especially at night when the patient lies down, and cause aspiration pneumonia. Oesophageal dysmotility can cause oesophageal spasm and central chest pain which may be confused with cardiac pain.
‘Mechanical’ dysphagia is often due to oesophageal stricture. With weight loss, a short history and no reflux symptoms, suspect oesophageal cancer. Longstanding dysphagia without weight loss but accompanied by heartburn is more likely to be due to benign peptic stricture. Record the site at which the patient feels the food sticking but this is not a reliable guide to the site of oesophageal obstruction.
Nausea and vomiting
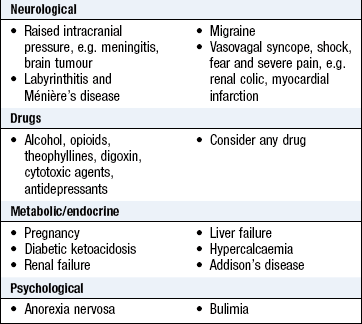
Nausea is the sensation of feeling sick. Vomiting is the expulsion of gastric contents via the mouth. Both are associated with pallor, sweating and hyperventilation. Nausea and vomiting, particularly with abdominal pain or discomfort, suggest upper gastrointestinal disorders. Remember to consider non-gastrointestinal causes of nausea and vomiting, especially adverse drug effects, pregnancy and vestibular disorders (Boxes 8.9 and 8.10).
Dyspepsia causes nausea without vomiting. Peptic ulcers seldom cause painless vomiting unless complicated by pyloric stenosis. Gastric outlet obstruction causes projectile vomiting of large volumes of gastric content that is not bile-stained (green). Obstruction distal to the pylorus produces bile-stained vomit. The more distal the level of intestinal obstruction, the more marked the accompanying symptoms of abdominal distension and intestinal colic.
Vomiting is common in gastroenteritis, cholecystitis, pancreatitis and hepatitis. Vomiting is typically preceded by nausea, but in raised intracranial pressure may occur without warning. Severe pain may cause it, e.g. renal or biliary colic, myocardial infarction, as well as systemic disease, metabolic disorders and drug therapy.
Anorexia nervosa and bulimia are eating disorders characterised by undisclosed, self-induced vomiting. In bulimia, weight is maintained or increased, unlike in anorexia nervosa, where weight loss is obvious.
Wind and flatulence
Belching, excessive or offensive flatus, abdominal distension and borborygmi (audible bowel sounds) are often called ‘wind’ or flatulence. Clarify exactly what patients mean. Belching is due to air swallowing (aerophagy) and has no medical significance. It may indicate anxiety, but sometimes occurs in an attempt to relieve abdominal pain or discomfort, and accompanies GORD.
Normally 200–2000 ml of flatus is passed each day. Flatus is a mixture of gases derived from swallowed air and from colonic bacterial fermentation of poorly absorbed carbohydrates. Excessive flatus occurs particularly in lactase deficiency and intestinal malabsorption. No flatus is passed with intestinal obstruction.
Borborygmi result from movement of fluid and gas along the bowel. Loud borborygmi, particularly if associated with colicky discomfort, suggest small-bowel obstruction or dysmotility.
Abdominal distension
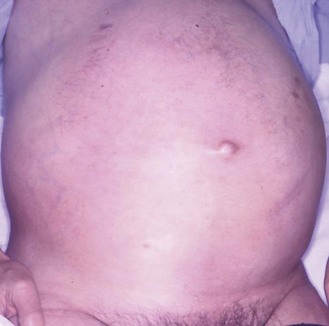
Abdominal girth slowly increasing over months or years is usually due to obesity, but in a patient with weight loss it suggests intra-abdominal disease (Box 8.11).
Ascites is an accumulation of fluid in the peritoneal cavity. Exudates from the peritoneal membrane have a higher protein content than transudates and indicate inflammatory or malignant disease (Fig. 8.7 and Box 8.12).
Functional bloating is fluctuating abdominal distension that develops during the day and resolves overnight. It is rarely due to organic disease and usually occurs in irritable bowel syndrome.
Chronic simple constipation rarely produces painful distension, unless associated with the irritable bowel syndrome. Painless abdominal distension in women may be the presenting symptom of ovarian pathology or a concealed pregnancy.
Altered bowel habit
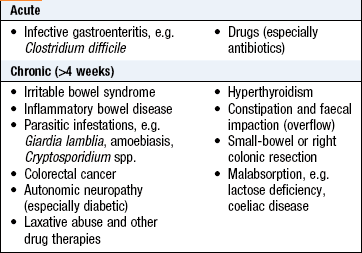
Diarrhoea: Diarrhoea is the frequent passage of loose stools. Normal bowel movement frequency ranges from three times daily to once every 3 days.
High-volume diarrhoea (>1 litre per day) occurs when stool water content is increased (the principal site of water absorption being the colon) and may be:
• secretory, due to intestinal inflammation, e.g. infection, or inflammatory bowel disease
• osmotic, due to malabsorption, adverse drug effects or motility disorders (Box 8.13).
If the patient fasts, osmotic diarrhoea stops but secretory diarrhoea persists. Steatorrhoea is diarrhoea associated with fat malabsorption. The stools are greasy, pale and bulky, and float, making them difficult to flush away.
Low-volume diarrhoea is associated with the irritable bowel syndrome (Box 8.14). The diagnosis of irritable bowel syndrome is based on a pattern of gastrointestinal symptoms (Box 8.15). Abdominal bloating, dyspepsia and often non-alimentary symptoms commonly accompany irritable bowel symptoms.
Bloody diarrhoea may be due to inflammatory bowel disease, colonic ischaemia or infective gastroenteritis.
Constipation: Constipation is the infrequent passage of hard stools (Box 8.16) and may be due to impaired colonic motility, physical obstruction, impaired rectal sensation or anorectal dysfunction causing anismus (impaired process of evacuation) (Box 8.17). Absolute constipation (no gas or bowel movements) suggests intestinal obstruction and is likely to be associated with pain, vomiting and distension. Tenesmus, the sensation of needing to defecate although the rectum is empty, suggests rectal inflammation or tumour.
Bleeding
Haematemesis: Haematemesis is vomiting blood, which can be fresh and red, or degraded by gastric pepsin, when it is dark brown in colour and resembles coffee grounds (Box 8.18). If the source of bleeding is above the gastro-oesophageal sphincter, e.g. from oesophageal varices, fresh blood may well up in the mouth, as well as being actively vomited. With a lower oesophageal mucosal tear due to the trauma of forceful retching (Mallory–Weiss syndrome), the patient vomits forcefully several times and fresh blood only appears after the patient has vomited several times.
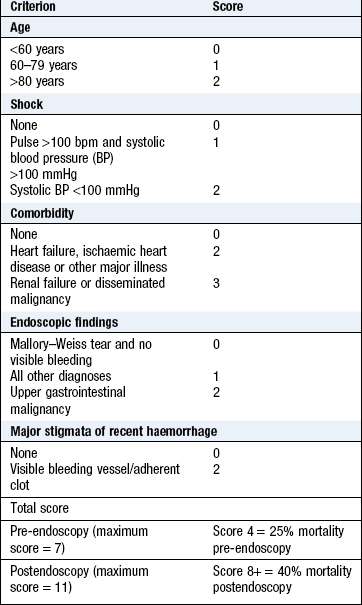
Melaena: Melaena is the passage of tarry, shiny black stools with a characteristic odour and results from upper gastrointestinal bleeding. Distinguish this from the matt black stools associated with oral iron or bismuth therapy. Excessive alcohol ingestion may cause haematemesis from erosive gastritis, Mallory–Weiss tear or bleeding oesophageal varices. Peptic ulceration is a common cause of upper gastrointestinal bleeding (Box 8.19). The Rockall and Blatchford scores are used to assess the severity (Box 8.20). A profound upper gastrointestinal bleed may lead to the passage of purple stool, or, rarely, fresh blood (see below).
Rectal bleeding: Fresh rectal bleeding indicates a disorder in the anal canal, rectum or colon (Box 8.21). Blood may be mixed with stool, coat the surface of otherwise normal stool, or be seen on the toilet paper or in the pan. Melaena signifies blood loss from the upper gastrointestinal tract. During severe upper gastrointestinal bleeding, blood may pass through the intestine unaltered, causing fresh rectal bleeding.
Jaundice
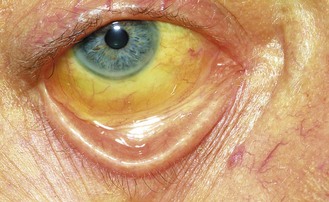
Jaundice is a yellowish discoloration of the skin, sclerae (Fig. 8.8) and mucous membranes due to hyperbilirubinaemia (Box 8.22). There is no absolute level at which jaundice is clinically detected but, in good light, most clinicians will recognise jaundice when bilirubin levels exceed 50 µmol/l.
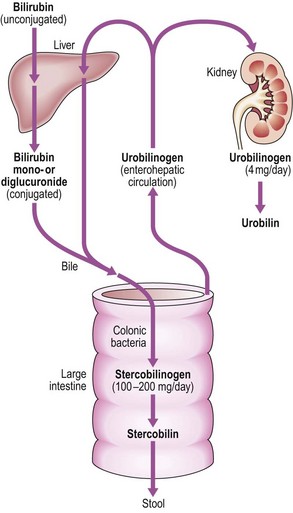
Unconjugated bilirubin is insoluble and transported in plasma bound to albumin; it is therefore not filtered by the renal glomeruli. In jaundice from unconjugated hyperbilirubinaemia, the urine is a normal colour (acholuric jaundice; Box 8.23).
Bilirubin is conjugated to form bilirubin diglucuronide in the liver, and excreted in bile, producing its characteristic green colour (Fig. 8.9). In conjugated hyperbilirubinaemia, the urine is dark brown in colour due to the presence of bilirubin diglucuronide. In the colon, conjugated bilirubin is metabolised by bacterial flora to stercobilinogen and stercobilin which are excreted in the stool, contributing to the brown colour of stool. Stercobilinogen is absorbed from the bowel and excreted in the urine as urobilinogen, a colourless, water-soluble compound.
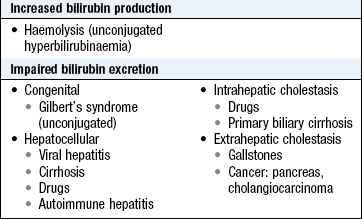
Prehepatic jaundice: In haemolytic disorders the accompanying anaemic pallor combined with jaundice may produce a pale lemon complexion. The stools and urine are normal in colour. Gilbert’s syndrome is common and causes unconjugated hyperbilirubinaemia. Serum liver enzyme concentrations are normal and jaundice is mild (plasma bilirubin <100 µmol/l) but increases during prolonged fasting or intercurrent febrile illness.
Hepatic jaundice: Hepatocellular disease causes hyperbilirubinaemia that is both unconjugated and conjugated. Conjugated bilirubin is soluble and filtered by the kidney, so the urine is dark brown. The stools are normal in colour.
Posthepatic/cholestatic jaundice: In biliary obstruction, conjugated bilirubin in the bile does not reach the intestine, so the stools are pale. Obstructive jaundice may be accompanied by pruritus (generalised itch) due to skin deposition of bile salts. Obstructive jaundice with abdominal pain is usually due to gallstones; if fever or rigors also occur (Charcot’s triad), ascending cholangitis is likely. Painless obstructive jaundice suggests malignant biliary obstruction, e.g. cholangiocarcinoma or cancer of the head of the pancreas. Obstructive jaundice can be due to intrahepatic as well as extrahepatic cholestasis, e.g. primary biliary cirrhosis, certain hepatotoxic drug reactions and profound hepatocellular injury (Boxes 8.24 and 8.25).
The history
Gastrointestinal symptoms are common and are often caused by functional dyspepsia and irritable bowel syndrome. Explore the patient’s ICE (p. 8) to understand the context in which symptoms have arisen. Alarm features raise the probability of a serious alternative or coexistent diagnosis (Box 8.26). The risk of serious disease increases with age, and patients >50 years should be investigated before a functional bowel disorder is diagnosed. Clarify exactly what patients mean by terms they use, especially constipation and diarrhoea.
Anorexia and weight loss: In addition to asking about appetite, ask: ‘Do you still enjoy your food?’ Confirm subjective assessment of weight loss from ill-fitting clothes and by reviewing documented weights from the case records.
Dysphagia and odynophagia: Ask: ‘Does food (or drink) stick when you swallow and is this painful?’ Enquire if there is difficulty swallowing liquids (neuromuscular disorder) or solids (oesophageal obstruction due to cancer, peptic stricture, achalasia).
Vomiting: Ask about relation to meals and about associated symptoms such as abdominal pain, weight loss and haematemesis (Box 8.10).
Reflux symptoms and dyspepsia: Ask about reflux-like dyspepsia (heartburn-predominant dyspepsia), ulcer-like dyspepsia (epigastric pain relieved by food or antacids) and dysmotility-like dyspepsia (nausea, belching, bloating and premature satiety).
Abdominal pain: Characterise pain using SOCRATES (Box 2.10) and use the questions listed in Box 8.8.
Altered bowel habit: Clarify what the patient means by diarrhoea or constipation. Use the Bristol stool chart to ensure agreement (Fig. 8.10). Use questions listed in Box 8.14 for those with diarrhoea and Box 8.16 for those with constipation. For those with either diarrhoea or constipation, always ask: ‘Have you noticed any red blood in the stools?’
Jaundice: Ask about alcohol intake, recent travel abroad, use of illicit or intravenous drugs and take a sexual history (Box 8.27). Always review recently prescribed drugs. Ask about itching or dark urine or pale stools (obstructive jaundice) and whether there is associated weight loss, abdominal pain or fever.
Past history
History of a similar problem may suggest the diagnosis: for example, bleeding peptic ulcer or inflammatory bowel disease. Primary biliary cirrhosis and autoimmune hepatitis are associated with thyroid disease and non-alcoholic fatty liver disease (NAFLD) is associated with diabetes and obesity. Ask about previous abdominal surgery and radiological and other investigations.
Drug history
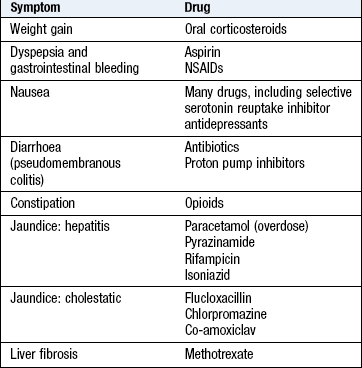
Ask about all prescribed medications, over-the-counter medicines and herbal preparations. Many drugs affect the gastrointestinal tract (Box 8.25) and are hepatotoxic.
Family history
Inflammatory bowel disease is more common in patients with a family history of either Crohn’s disease or ulcerative colitis. Colorectal cancer in a first-degree relative increases the risk of colorectal cancer and polyps. Peptic ulcer disease is familial but this may be due to environmental factors, e.g. transmission of Helicobacter pylori infection. Gilbert’s syndrome is an autosomal dominant condition; haemochromatosis and Wilson’s disease are autosomal recessive disorders. Autoimmune diseases, particularly thyroid disease, are common in relatives of those with primary biliary cirrhosis and autoimmune hepatitis. A family history of diabetes is frequently seen in the context of NAFLD.
Social history
Take a dietary history and approximately assess the intake of calories and sources of essential nutrients (Box 8.28). Painless diarrhoea may indicate high alcohol intake, lactose intolerance or gluten enteropathy. Patients with irritable bowel syndrome often report specific food intolerances, including wheat, dairy products and others.
Calculate the patient’s alcohol consumption in units (p. 17).
Smokers are at increased risk of oesophageal cancer, colorectal cancer, Crohn’s disease and peptic ulcer, while patients with ulcerative colitis are less likely to smoke. Many disorders, particularly irritable bowel syndrome and dyspepsia, are exacerbated by stress and emotional ill health. Ask about potential sources of stress, as well as about the symptoms of anxiety and depression (Box 2.49).
In patients with liver disease, ask about specific risk factors (Box 8.27). Hepatitis B and C may present with chronic liver disease or cancer decades after the primary infection, so enquire about drug use and other risk factors in the distant as well as the recent past. Foreign travel is important in relation to diarrhoeal illnesses.
The physical examination
• Record the height, weight, waist circumference and the patient’s body mass index (p. 55).
• Note whether obesity is truncal or generalised. Look for abdominal striae, which indicate rapid weight gain, previous pregnancy or, rarely, Cushing’s syndrome. Loose skin folds signify recent weight loss.
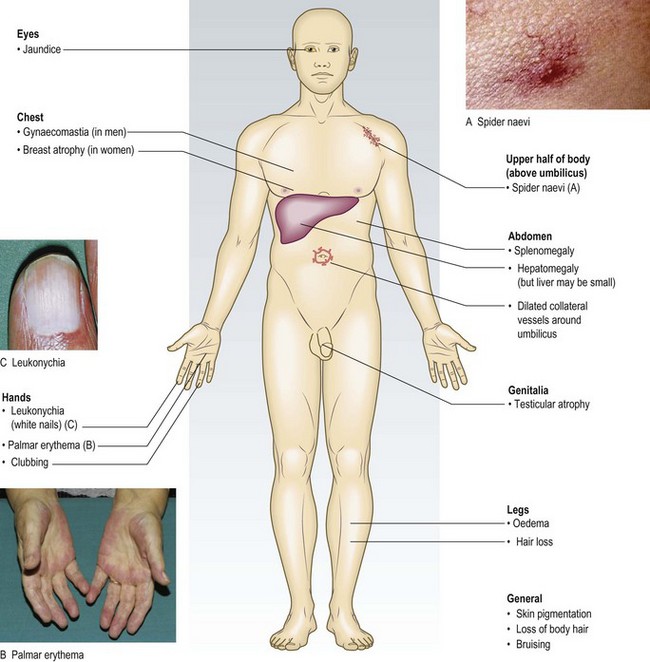
Liver disease: If jaundice is not obvious, ask the patient to look down and retract the upper eyelid to expose the sclera; look to see if it is yellow in natural light (Fig. 8.8). Do not confuse the diffuse yellow sclerae of jaundice with small yellowish fat pads (pingueculae) sometimes seen at the periphery of the sclerae.
Certain signs which suggest chronic liver disease (Fig. 8.11):
• Palmar erythema and spider naevi are due to excess oestrogen associated with reduced hepatic breakdown of sex steroids. Spider naevi are isolated telangiectases that characteristically fill from a central feeding vessel found in the distribution of the superior vena cava on the upper trunk, arms and face (Fig. 8.11). Women may have up to five spider naevi in health; palmar erythema and numerous spider naevi are normal during pregnancy. In men these signs suggest chronic liver disease (Box 8.29).
• Gynaecomastia (breast enlargement in males), with loss of body hair and testicular atrophy, may occur due to reduced breakdown of oestrogens.
• Leukonychia (white nails), caused by hypoalbuminaemia, may also occur in protein calorie malnutrition (kwashiorkor), malabsorption due to protein-losing enteropathy, e.g. coeliac disease, or heavy and prolonged proteinuria (nephrotic syndrome).
• Finger clubbing is associated with liver cirrhosis, inflammatory bowel disease and malabsorption syndromes.
Signs which suggest liver failure include the following (Box 8.30):
• Asterixis is a coarse flapping tremor which occurs with hepatic encephalopathy (p. 151).
• Fetor hepaticus, a distinctive ‘mousy’ odour on the breath, is due to the volatile compound dimethyl sulphide and is evidence of portosystemic shunting (with or without encephalopathy).
Other signs which may be associated with liver disease include:
• Dupuytren’s contracture (contracture of the palmar fascia; p. 49) is linked with alcohol-related chronic liver disease. More commonly, however, it is familial (autosomal dominant with variable penetrance) or associated with conditions causing microvascular pathology, e.g. diabetes mellitus, smoking, hyperlipidaemia, HIV infection.
• Bilateral parotid swelling due to sialoadenosis of the salivary glands may be a feature of chronic alcohol abuse or bulimia associated with recurrent vomiting.
The abdomen
Normal findings: The abdomen is normally flat or slightly scaphoid and symmetrical. At rest, respiration is principally diaphragmatic; the abdominal wall moves out and the liver, spleen and kidneys move downwards during inspiration. The umbilicus is usually inverted.
Skin: In older patients, seborrhoeic warts, ranging in colour from pink to brown or black, and haemangiomas (Campbell de Morgan spots) are common and normal, but note any striae, bruising or scratch marks.
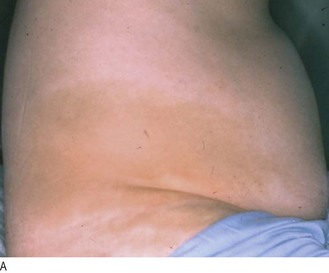
Visible veins: Abnormally prominent veins on the abdominal wall suggest portal hypertension or vena caval obstruction. In portal hypertension, recanalisation of the umbilical vein along the falciform ligament produces distended veins which drain away from the umbilicus: the ‘caput medusae’. The umbilicus may appear bluish and distended due to an umbilical varix. In contrast, an umbilical hernia is a distended and everted umbilicus which does not appear vascular and may have a palpable cough impulse. Dilated tortuous veins with blood flow superiorly are collateral veins due to obstruction of the inferior vena cava. Rarely, superior vena cava obstruction gives rise to similarly distended abdominal veins, but which all flow inferiorly.
Abdominal distension: If the abdomen is distended, is this generalised or localised? In obesity, the umbilicus is usually sunken; in ascites, it is flat or everted. Look tangentially across the abdomen and from the foot of the bed for any asymmetry associated with a localised mass, such as an enlarged liver or bladder.
Abdominal scars and stomas: Note any surgical scars or stomas and clarify what operations have been undertaken (Figs 8.12 and 8.13). A small infraumbilical incision is usually the result of previous laparoscopy. Puncture scars from the ports used for laparoscopic surgery may be visible. An incisional hernia at the site of a scar is palpable as a defect in the abdominal wall musculature and becomes more obvious as the patient raises the head off the bed or coughs.
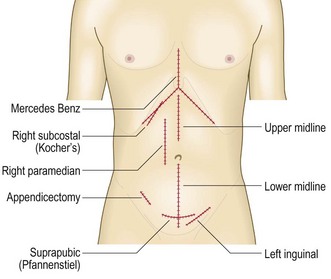
Fig. 8.12 Some abdominal incisions.
The midline and oblique incisions avoid damage to innervation of the abdominal musculature and later development of incisional herniae. These incisions however have been widely superseded by laparascopic surgery.
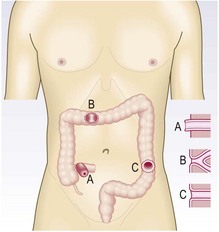
Fig. 8.13 Surgical stomas.
A, An ileostomy is usually in the right iliac fossa and is formed as a spout. B, A loop colostomy is created to defunction the distal bowel temporarily. It is usually in the transverse colon and has afferent and efferent limbs. C, A colostomy may be terminal, i.e. resected distal bowel. It is usually flush and in the left iliac fossa.
Normal findings: A pulsatile mass palpable in the upper abdomen may be normal aortic pulsation in a thin person, a gastric or pancreatic tumour transmitting underlying aortic pulsation or an aortic aneurysm.
The normal liver is identified as an area of dullness to percussion over the right anterior chest between the fifth rib and the costal margin. The normal spleen is identified as an area of dullness to percussion posterior to the left mid-axillary line beneath the ninth, 10th and 11th ribs.
Tenderness: Discomfort during palpation may vary and be accompanied by resistance to palpation. Consider the patient’s level of anxiety when assessing the severity of pain and tenderness elicited. Tenderness in several areas on minimal pressure may be due to generalised peritonitis but is more often due to anxiety. Severe superficial pain with no tenderness on deep palpation or pain that disappears if the patient is distracted also suggests anxiety. With these exceptions, tenderness usefully indicates underlying pathology.
Voluntary guarding is the voluntary contraction of the abdominal muscles when palpation provokes pain. Involuntary guarding is the reflex contraction of the abdominal muscles when there is inflammation of the parietal peritoneum. If the whole peritoneum is inflamed (generalised peritonitis) due to a perforated viscus, the abdominal wall no longer moves with respiration; breathing becomes increasingly thoracic and the anterior abdominal wall muscles are held rigid (board-like rigidity).
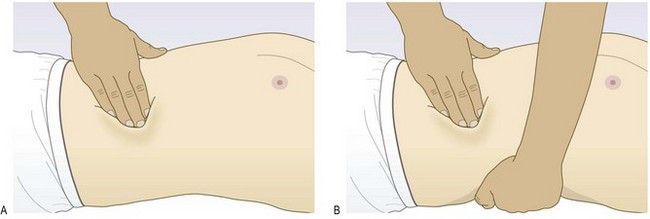
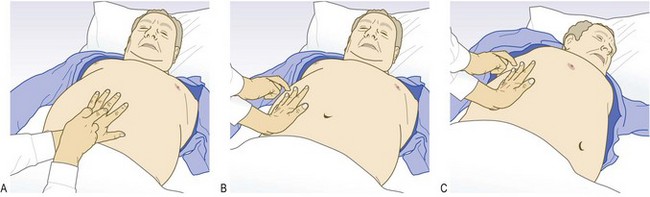
The site of tenderness is important. Tenderness in the epigastrium suggests peptic ulcer; in the right hypochondrium, cholecystitis; in the left iliac fossa, diverticulitis; in the right iliac fossa, appendicitis or Crohn’s ileitis. ‘Rebound tenderness’ is a sign of intra-abdominal disease but not necessarily of parietal peritoneal inflammation (peritonism). Ask the patient to cough or gently percuss the abdomen to elicit any pain or tenderness, rapidly removing your hand after deep palpation increases the pain (Fig. 8.14).
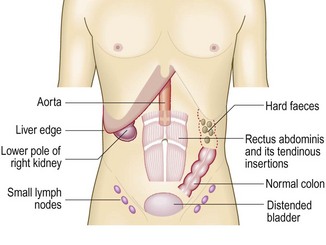
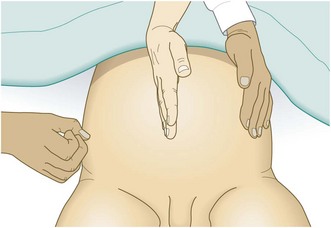
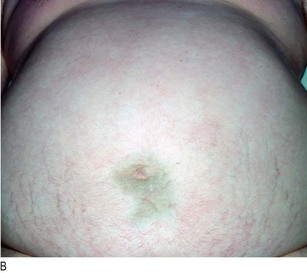
Palpable mass: A pathological mass can usually be distinguished from palpable faeces as the latter are indentable and may disappear following defecation (Fig. 8.15). A hard subcutaneous nodule palpable at the umbilicus may indicate metastatic cancer (‘Sister Mary Joseph’s nodule’).
Palpation for enlarged organs: Examine the liver, gallbladder, spleen and kidneys in turn during deep inspiration. Keep your examining hand still and wait for the organ to descend. Do not start palpation too close to the costal margin, missing the edge of the liver or spleen.
Normal findings: You may feel the liver edge below the right costal margin. Other normal findings may include:
• The aorta may be palpable as a pulsatile swelling above the umbilicus
• The lower pole of the right kidney may be palpable in the right flank
• Faecal scybala may be palpable in the sigmoid colon in the left iliac fossa
• A full bladder arising out of the pelvis may be palpable in the suprapubic region.
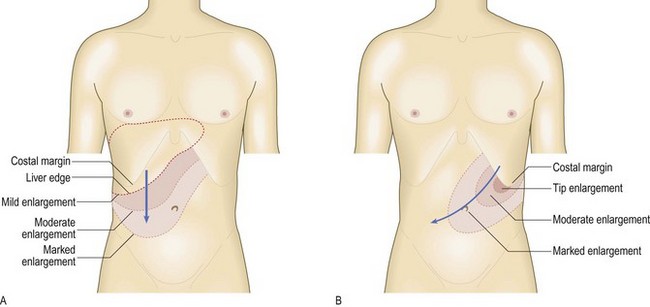
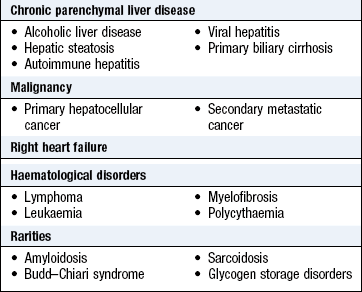
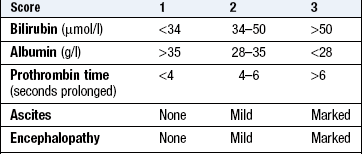
Abnormal findings: Hepatic enlargement can result from chronic parenchymal liver disease from any cause (Box 8.33). The liver is enlarged in early cirrhosis but often shrunken in advanced cirrhosis. Fatty liver (hepatic steatosis) can cause marked hepatomegaly. Hepatic enlargement due to metastatic tumour is hard and irregular. An enlarged left lobe may be felt in the epigastrium or even in the left hypochondrium. An audible bruit may be heard over the liver in hepatocellular cancer and sometimes in alcoholic hepatitis. In right heart failure, the congested liver is usually soft and tender; a pulsatile liver indicates tricuspid regurgitation. A bruit over the liver may be heard in acute alcoholic hepatitis, hepatocellular cancer and arteriovenous malformation. Liver failure produces associated symptoms and its severity can be graded (Box 8.34).
Resonance below the fifth intercostal space suggests emphysema or occasionally the interposition of the transverse colon between the liver and the diaphragm (Chilaiditi’s sign).
Palpable distension of the gallbladder has a characteristic globular shape. It is rare and results from either obstruction of the cystic duct, as in a mucocoele or empyema of the gallbladder, or obstruction of the common bile duct (providing the cystic duct is patent), as in pancreatic cancer. In gallstone disease the gallbladder may be tender but impalpable because of fibrosis of the gallbladder wall.
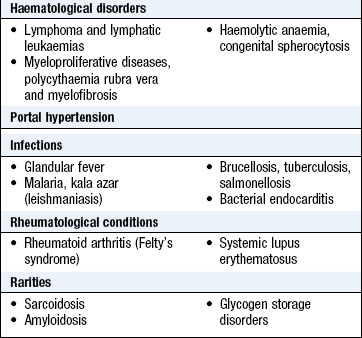
Abnormal findings: The spleen has to increase in size threefold before it becomes palpable, so a palpable spleen always indicates splenomegaly. The normal spleen lies beneath the ninth and 11th ribs in the left mid-axillary line. It enlarges from under the left costal margin down and medially towards the umbilicus (Fig. 8.17B). A characteristic notch may be palpable midway along its leading edge, differentiating it from an enlarged left kidney.
Although there are many causes of splenomegaly (Boxes 8.35–8.37), massive enlargement in the developed world is usually due to myeloproliferative disease or haematological malignancy; worldwide, malaria is a common cause.
Ascites: Ascites is the accumulation of intraperitoneal fluid. Causes include intra-abdominal malignancy, chronic liver disease, severe heart failure, nephrotic syndrome and hypoproteinaemia.
Normal findings: Bowel sounds are gurgling noises from the normal peristaltic activity of the gut. They normally occur every 5–10 seconds, but the frequency varies.
Abnormal findings: Absence of bowel sounds implies paralytic ileus or peritonitis. In intestinal obstruction, bowel sounds occur with increased frequency and volume and have a high-pitched, tinkling quality. Bruits suggest an atheromatous or aneurysmal aorta or superior mesenteric artery stenosis. A friction rub, which sounds like rubbing your dry fingers together, may be heard over the liver (perihepatitis) or spleen (perisplenitis). An audible splash more than 4 hours after the patient has eaten or drunk anything indicates delayed gastric emptying, e.g. pyloric stenosis.
Hernias
Hernias are common and typically occur at openings of the abdominal wall, e.g. the inguinal, femoral and obturator canals, the umbilicus and the oesophageal hiatus. They may occur at sites of weakness of the abdominal wall or be related to previous surgical incisions.
An external abdominal hernia is an abnormal protrusion of bowel and/or omentum from the abdominal cavity. External hernias are more obvious when the pressure within the abdomen rises, e.g. when the patient is standing, coughing or straining at stool. Internal hernias occur through defects of the mesentery or into the retroperitoneal space and are not visible.
An impulse can often be felt in a hernia during coughing (cough impulse). Identify a hernia from its anatomical site and characteristics, and attempt to differentiate between direct and indirect inguinal types (Box 8.39).
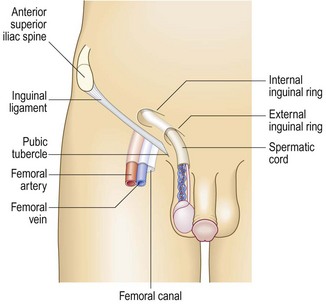
Anatomy: The inguinal canal extends from the pubic tubercle to the anterior superior iliac spine (Fig. 8.21). It has an internal ring at the mid-inguinal point and an external ring at the pubic tubercle. The mid-inguinal point is midway between the pubic symphysis and the anterior superior iliac spine, not at the midpoint of the inguinal ligament. The femoral canal lies below the inguinal ligament and lateral to the pubic tubercle.
History: Groin hernias frequently present with a dull dragging discomfort, rather than acute pain, which is often exacerbated by straining. A swelling or lump in the groin may be the only presenting feature. Ask the patient about precipitating/exacerbating factors such as straining, e.g. due to chronic constipation, chronic cough, heavy manual labour, straining at micturition. Typically symptoms are worse at the end of a day when the patient has been standing or active for a prolonged period. Often patients can manually reduce the hernia by gentle pressure over the swelling or by lying flat.
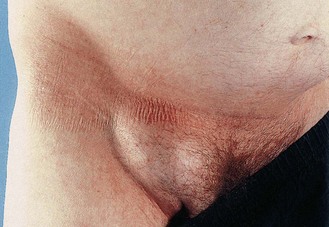
Abnormal findings: An indirect inguinal hernia bulges through the internal ring and follows the course of the inguinal canal. It may extend beyond the external ring and enter the scrotum. Indirect hernias comprise 85% of all hernias and are more common in younger men.
A direct inguinal hernia forms at a site of muscle weakness in the posterior wall of the inguinal canal and rarely extends into the scrotum. It is more common in older men and women (Fig. 8.22).
A femoral hernia projects through the femoral ring and into the femoral canal.
Inguinal hernias are palpable above and medial to the pubic tubercle. Femoral hernias are palpable below the inguinal ligament and lateral to the pubic tubercle.
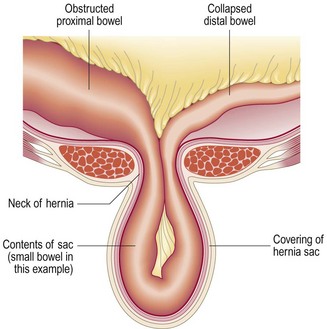
In a reducible hernia the contents can be returned to the abdominal cavity, spontaneously or by manipulation; if they cannot, the hernia is irreducible. An abdominal hernia has a covering sac of peritoneum and the neck of the hernia is a common site of compression of the contents (Fig. 8.23). If bowel is contained within the hernia, obstruction may occur. If the blood supply to the contents of the hernia (bowel or omentum) is restricted, the hernia is strangulated. It is tense and tender and has no cough impulse; there may be bowel obstruction and, later, signs of sepsis and shock. A strangulated hernia is a surgical emergency and, untreated, will lead to bowel infarction and peritonitis.
Rectal examination
Digital examination of the rectum is important (Box 8.40). Do not avoid it because you or the patient finds it disagreeable.
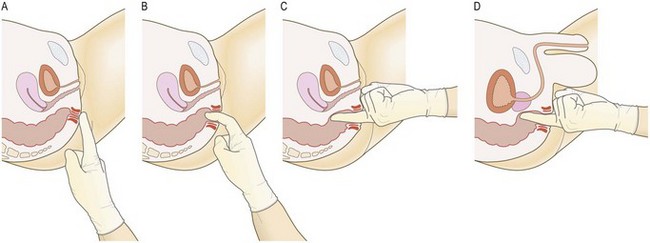
Anatomy: The normal rectum is usually empty and smooth-walled, with the coccyx and sacrum lying posteriorly. In the male, anterior to the rectum from below upwards, lie the membranous urethra, the prostate and the base of the bladder. The normal prostate is smooth and has a firm consistency, with lateral lobes and a median groove between them. In the female, the vagina and cervix lie anteriorly. The upper end of the anal canal is marked by the puborectalis muscle, which is readily palpable and contracts as a reflex action on coughing or on conscious contraction by the patient. Beyond the anal canal, the rectum passes upwards and backwards along the curve of the sacrum.
Spasm of the external anal sphincter is common in anxious patients. When associated with local pain, it is probably due to an anal fissure (a mucosal tear). If you suspect an anal fissure, give the patient a local anaesthetic suppository 10 minutes before the examination to reduce the pain and spasm and to aid examination.
Abnormal findings: Haemorrhoids (‘piles’, congested venous plexuses around the anal canal) are only palpable if thrombosed. In patients with chronic constipation the rectum is often loaded with faeces. Faecal masses are often palpable, should be movable and can be indented. In women, a retroverted uterus and the normal cervix are often palpable through the anterior rectal wall and a vaginal tampon may be confusing. Cancer of the lower rectum is palpable as a mucosal irregularity. Obstructing cancer of the upper rectum may produce ballooning of the empty rectal cavity below. Metastases or colonic tumours within the pelvis may be mistaken for faeces and vice versa. Lateralised tenderness suggests pelvic peritonitis. Gynaecological malignancy may cause a ‘frozen pelvis’ with a hard, rigid feel to the pelvic organs due to extensive peritoneal disease, e.g. post-radiotherapy or metastatic cervical or ovarian cancer.
Benign prostatic hyperplasia often produces palpable symmetrical enlargement, but not if the hyperplasia is confined to the median lobe. A hard, irregular or asymmetrical gland with no palpable median groove suggests prostate cancer. Tenderness accompanied by a change in the consistency of the gland may be due to prostatitis or prostatic abscess. The prostate is abnormally small in hypogonadism.
Proctoscopy
Always undertake a digital rectal examination before visual examination of the anal canal by proctoscopy. If an examination of the rectal mucosa is required, flexible sigmoidoscopy is indicated rather than proctoscopy.
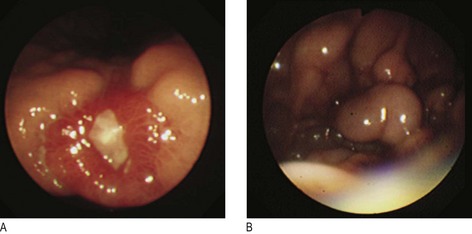
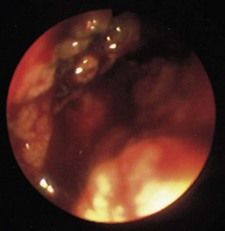
Abnormal findings: Proctoscopic examination of the anus and lower rectum can confirm or exclude the presence of haemorrhoids, anal fissures and rectal prolapse. Rectal mucosa looks like buccal mucosa, apart from the presence of prominent submucosal veins. During straining, haemorrhoids distend with blood and may prolapse. If the degree of protrusion is more than 3–4 cm, a rectal prolapse may be present.
The acute abdomen
The majority of general surgical emergencies are patients with an ‘acute abdomen’. Causes range from self-limiting conditions to severe life-threatening diseases (Box 8.42). Evaluate patients rapidly, then immediately resuscitate critically ill patients before undertaking further assessment and surgical intervention. Give parenteral opioid analgesia early to alleviate severe abdominal pain, as it will help, not hinder, clinical assessment. In a patient with undiagnosed acute abdominal pain, regularly reassess his clinical state, undertake urgent investigations and consider surgical intervention before administering repeat analgesia.
Patients may be so occupied by recent and severe symptoms that they forget important details of the history unless you ask them directly (Box 8.43). Ask family or friends for additional information if severe pain, shock or altered consciousness makes it difficult to obtain an accurate history from the patient. Note any past history which may be relevant, e.g. acute perforation in a patient with known diverticular disease. Remember that disease outside the abdomen, e.g. myocardial infarction, pneumonia, diabetic ketoacidosis or herpes zoster, may present with acute abdominal pain (Box 8.5). Abdominal signs may be masked in patients taking steroids, immunosuppressants or anti-inflammatory drugs, in alcohol intoxication or in altered states of consciousness (Boxes 8.44 and 8.45).
Investigations
See Box 8.46 and Figures 8.29-33.
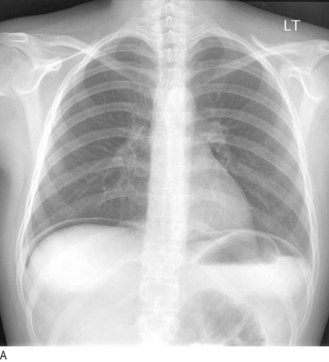
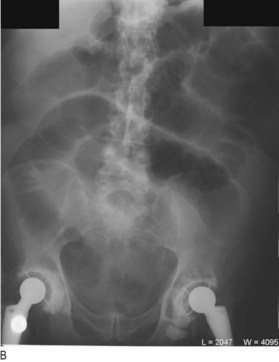
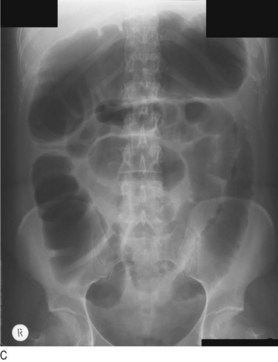
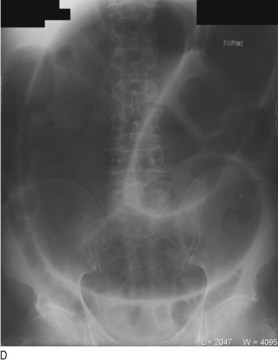
Fig. 8.29 Radiography in gastrointestinal disease.
(A) Air under the diaphragm on chest X-ray due to perforated duodenal ulcer. (B) Dilated small bowel due to acute intestinal obstruction. (C) Dilated large bowel due to toxic megacolon. (D) Dilated loop of large bowel due to sigmoid volvulus.
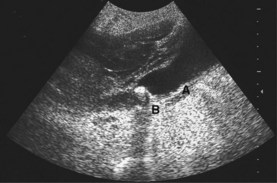
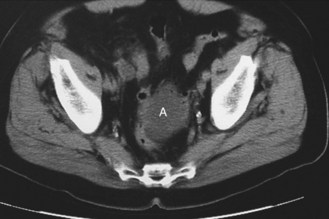
Fig. 8.30 Ultrasound scan showing thick-walled gallbladder (A) containing gallstones with posterior acoustic shadowing (B).
Stool
The stool can be tested for faecal occult blood (FOB). A positive FOB is produced by any gastrointestinal haemorrhage, e.g. bleeding peptic ulcer, colorectal cancer and inflammatory bowel disease. FOB tests are sensitive but not specific; false-positive tests occur after vigorous tooth brushing or after eating rare steak or other red meat. False-negative tests occur in patients with proven colorectal or gastric cancers, chronic upper gastrointestinal haemorrhage and inflammatory bowel disease. The test has value as a population screening tool for colorectal cancer.
Urine
Test the urine for bilirubin and urobilinogen to confirm jaundice and point to its cause (Box 8.23). Urinalysis is useful in diagnosing non-surgical causes of acute abdominal pain, e.g. diabetes mellitus (glucose, ketones), porphyria (porphobilinogen) and renal colic (haematuria).
Ascitic fluid
Obtain a sample of fluid for inspection and analysis from all patients with ascites (diagnostic ascitic tap or abdominal paracentesis). Use either iliac fossa at a point one-third of the distance from the anterior superior iliac spine to the umbilicus, avoiding any previous surgical scars. Insert a needle using strict aseptic technique and aspirate up to 20 ml.
Ascitic fluid is usually clear and straw-coloured. Uniformly blood-stained fluid suggests intra-abdominal malignancy. Turbid fluid may indicate a high cell count due to infection, or high protein content. Occasionally, ascitic fluid may be chylous, with a milky appearance due to a high lipid content, usually indicating lymphatic obstruction. Send the fluid for analysis of protein/albumin content, cell count and culture, cytology for malignant cells, and measurement of amylase and glucose. Measure the serum ascites albumin gradient, the difference in albumin content between serum and ascitic fluid. Values <11 g/L indicate an exudate, i.e. an inflammatory or malignant process; values >11 g/L indicate a transudate, most commonly due to cirrhosis and portal hypertension (Boxes 8.12 and 8.47).