5 The lower urinary tract
By the end of the chapter you should be able to:
• State two qualities of urothelium that make it suitable to line the bladder and ureters
• Outline the function of the detrusor and sphincter muscles, and their innervations
• Describe the embryological origin and significance of double and bifid ureters
• Summarize common neurological causes of difficulties with micturition
• Explain the difference between stress and urge incontinence
• List the causes of urinary obstruction and explain the significance of hydronephrosis
• List the five types of urinary calculus and their causes
• State the maximum diameter of calculus generally considered likely to pass spontaneously
• Give the risk factors and symptoms of acute cystitis and list the common UTI pathogens
• Outline the mechanism by which schistosomiasis increases the risk of bladder cancer
• Understand the causes of inflammation of the prostate
• Define benign prostatic hyperplasia and explain the histological changes
Organization of the lower urinary tract
Overview
Urine formed in the kidneys collects in the renal pelvis and then passes down the lower urinary tract (ureters, bladder and urethra) before exiting the body. The bladder stores the urine, which is ejected intermittently from the body under voluntary control.
Ureters
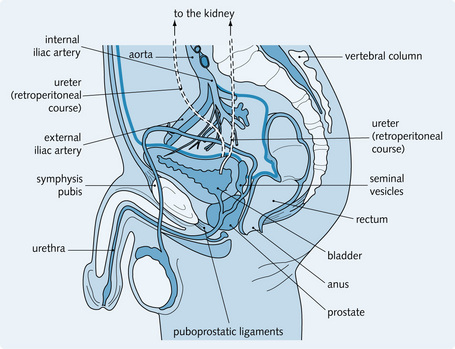
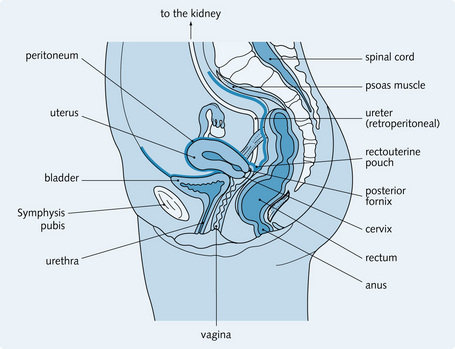
The ureters are hollow muscular tubes 25–30 cm long, which begin as funnel-shaped tubes at the renal pelvis. They run retroperitoneally over the posterior abdominal wall in front of the external iliac artery down to the pelvic brim (similar course in the female). Figs 5.1 and 5.2 show the course of the ureters through the pelvis in a man and woman, respectively. As urine collects in the renal pelvis, the pelvis dilates. Action potentials in the pacemaker cells of the renal pelvis are set up, stimulating peristaltic contractions in the ureters that propel the urine.
The ureters are divided into regions related to their anatomical course: renal pelvis, abdominal, pelvic and intramural regions (Fig. 5.3). This influences the source of blood supply to each region of the ureter – renal, lumbar segmental, gonadal, common iliac, internal iliac and superior vesical arteries, with corresponding venous drainage.
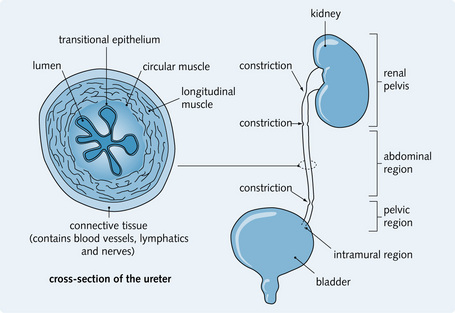
Fig. 5.3 Regions of the ureter and cross-sectional view. The normal points of reduced diameter are also shown, at which stones commonly lodge.
The ureters are innervated by both sympathetic and parasympathetic nerves. As shown in Figure 5.3 there are constrictions in the ureters. Stones can get stuck at these constrictions and produce acute colicky pain, which is referred to the skin of T11–L2. Therefore, pain starts in the loin and radiates to the scrotum and penis (men) or to the labium majora (women).
Histologically, the ureter has three layers of smooth muscle (see Fig. 5.3):
The lumen is lined by urothelium (also known as transitional epithelium), which is folded in the relaxed state, allowing the ureter to dilate during the passage of urine. The plasma membranes of urothelium are thicker than other cell membranes, preventing interstitial fluid from entering the concentrated urine. Urothelium is impermeable to urine. The cells have highly interdigitating cell junctions, allowing the epithelium to stretch without damaging the surfaces of the cells. It also lines the bladder and prostatic urethra.
Urinary bladder
The ureters enter the base of the bladder, which is partially covered by peritoneum.
The neck of the bladder is relatively immobile and fixed by the puboprostatic and lateral vesical ligament. The blood supply to the bladder is from the superior and inferior vesical branches of the internal iliac artery. It is drained by the vesical plexus and by the prostatic venous plexus in the male, which then drain into the internal iliac vein. Lymphatic drainage is also along the vesical blood vessels to the internal iliac nodes, then the para-aortic nodes.
Interior of the bladder
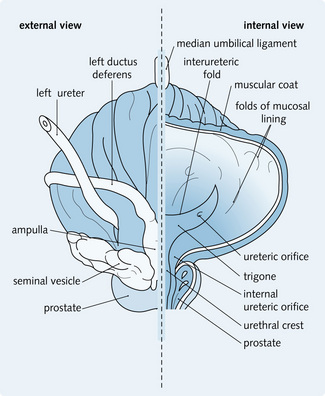
The wall is yellow with rugae (folds), lined by transitional epithelium, allowing expansion with little increase in internal pressure. The base is the trigone, which is a triangular, reddish region bounded by the ureteric openings into the bladder and the internal urethral meatus (see Fig. 5.4). This area is less mobile and less distensible than the rest of the bladder. It is more sensitive to painful stimuli.
The bladder is lined by smooth muscle, known as the detrusor muscle, which, like the ureter, is arranged in spiral, long and circular bundles. Smooth muscle bundles surround the bladder neck to form the internal urethral sphincter. Slightly further along the urethra there is a skeletal muscle sphincter – the external urethral sphincter.
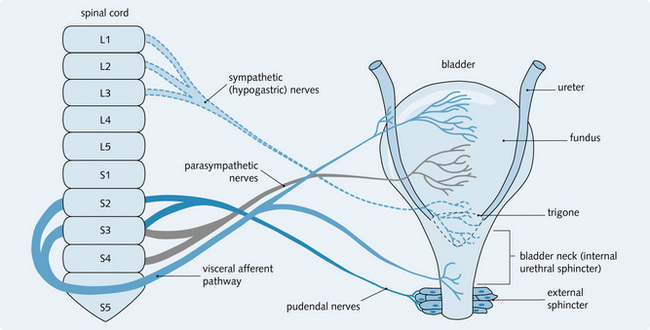
Bladder innervation (Fig. 5.5) is both:
• Sensory: gives sensation (awareness) of a full bladder and also pain from disease. The impulses are suppressed if the bladder is empty
• Motor: parasympathetic activity stimulates the detrusor muscle, so the bladder contracts. It also inhibits the external urethral sphincter, which relaxes to allow micturition. Sympathetic activity inhibits the detrusor muscle, so the bladder relaxes, and stimulates the urethral sphincter (this contracts). Both these actions prevent micturition.
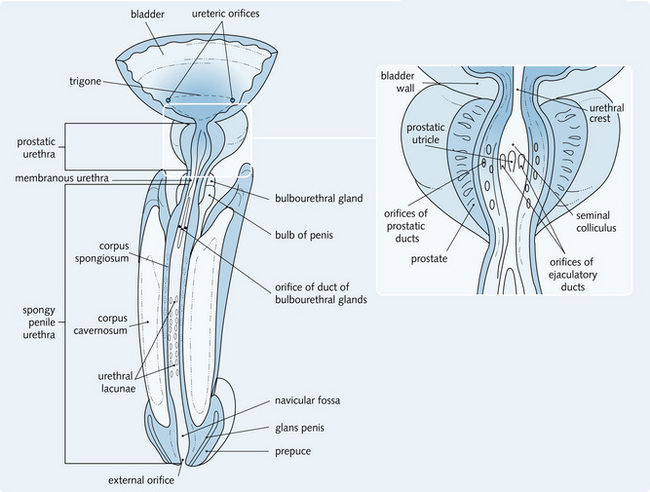
Male urethra
The male urethra (Fig. 5.6) is longer than the female urethra (male = 20 cm, female = 4 cm). It runs through the neck of the bladder, the prostate gland, the floor of pelvis and the perineal membrane to the penis and external urethral orifice at the tip of the glans penis. It has three parts:
1. Prostatic urethra: surrounded by prostate tissue, lined by transitional epithelium
2. Membranous urethra: the shortest region, with sphincter activity, lined by pseudostratified columnar epithelium
3. Spongy urethra: surrounded by penile tissue. This is lined by pseudostratified columnar epithelium except for the external opening which is lined by stratified squamous epithelium.
It is innervated by the prostatic plexus and lymphatic drainage is to the internal iliac and deep inguinal nodes.
Female urethra
This starts at the neck of the bladder and passes through the floor of the pelvis and perineal membrane to open into the vestibule just anterior to the opening of the vagina. It is 4 cm in length and is firmly attached to the anterior wall of the vagina. Lymphatics drain to the internal and external iliac lymph nodes. Proximally it is lined by transitional epithelium and the rest by stratified squamous epithelium.
Prostate
This is a gland lying below the bladder in the male and surrounding the proximal part of the urethra (prostatic urethra). It measures 4 × 3 × 2 cm and is conical in shape. It is connected to the bladder by connective tissue stroma and has three parts:
The prostate has a connective tissue capsule, which is surrounded by a thick sheath from the pelvic fascia. It is influenced by sex hormones resulting in growth during puberty. As the prostate surrounds the urethra, any enlargement can narrow the urethra and obstruct urine flow.
The prostate is supplied by the inferior vesical artery and blood drains via the prostatic plexus to the vesical plexus and internal iliac vein. Lymphatics drain to the internal iliac and sacral nodes.
The prostate contains a central zone of mucosal glands originating prenatally from the endoderm. These drain directly into the urethra. There is also a peripheral zone of mucosal glands, derived from the mesoderm, which drains into the ducts that enter the urethral sinus. Prostatic glandular epithelium can vary from inactive low cuboidal cells to active pseudostratified columnar cells, depending on the degree of androgen stimulation from the testes. The glands secrete 75% of seminal fluid, which is thin, milky and rich in citric acid and hydrolytic enzymes (e.g. fibrinolysin). This prostatic secretion liquefies coagulated semen after deposition in the female genital tract. The prostate is covered by a stroma and capsule made of dense fibroelastic connective tissue with a smooth muscle component.
Congenital abnormalities of the urinary tract
Ureteric abnormalities
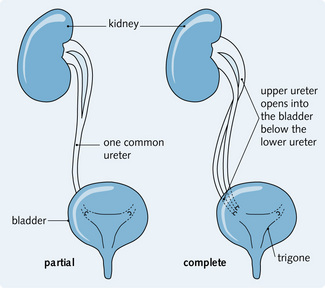
Double and bifid ureters
The ureters along with the calyces and collecting ducts are formed from an outgrowth of the mesonephric (Wolffian) duct called the ureteric bud. Early splitting of the ureteric bud or the development of two buds results in duplication, which can be:
• Partial: the two ureters meet before entering the bladder together. These are called bifid ureters
• Complete: the two ureters enter the bladder separately. The upper pole ureter enters the bladder lower and more medially than the lower ureter. These are called double ureters (see Fig. 5.7).
Renal function is rarely affected but there is a strong predisposition to infection. Urine can reflux from the bladder, especially through the upper pole ureter. Treatment involves excision of the refluxing ureter (usually the upper one).
Pelviureteric junction obstruction
This often presents in infancy, although milder forms might not present until later in adult life or may be found in asymptomatic patients at postmortem. It is more common in males and in the left ureter. It is bilateral in 20% of cases, and might present as a mass in the flank or pain after drinking. It is thought to result from abnormal smooth muscle organization at where the renal pelvis joins the upper ureter. It can be accompanied by renal agenesis of the opposite kidney; the reason for this is unknown. As a result of the back pressure from the obstruction, the pelvicalyceal system dilates. If the pressure is transmitted to the kidneys, the renal tissue atrophies.
Bladder abnormalities
Diverticula
These are sac-like outpouchings through a weak point in the bladder wall. They can be either:
• Congenital: these develop in localized areas of defective muscle within the wall or because of urinary tract obstruction in fetal development. They are usually solitary lesions, most commonly occurring close to the ureterovesical junction
• Acquired: these usually develop much later in life as a result of chronic urethral obstruction (e.g. prostatic hypertrophy). They are clinically significant and, characteristically, occur as multiple lesions.
In both cases, urine stasis increases the risk of bladder infection, leading to vesicoureteric reflux and eventual stone formation.
Exstrophy
Exstrophy of the bladder is a serious condition affecting the anterior wall of the bladder and anterior abdominal wall. It presents in infancy and is more common in males. The anterior wall of the bladder fails to develop, so the posterior wall lies exposed on the lower abdominal wall, causing squamous metaplasia of the mucosa. The mucosa is at high risk of infection. This disorder can vary in severity and can be associated with urethral and symphysis pubis defects. Even with surgical treatment there is an increased risk of adenocarcinoma of the bladder later in life, because of bladder extrusion.
Urethral abnormalities
Hypospadias
This is a spectrum of congenital abnormalities affecting 1 in 400 male infants. The urethra opens on the ventral surface of the penis, usually adjacent to the glans penis, but can open on the penile shaft or perineum. There is a ventral curvature to the penile shaft with a hooded prepuce. Surgical correction is usually carried out before the age of 2 years to allow micturition with a straight stream.
Epispadias
The urethra opens on the dorsal surface of the penis. As with hypospadias, surgical correction is usually carried out before the age of 2 years to allow micturition with a straight stream.
Urethral valves
Obstruction to urine flow can occur at the level of the posterior urethra in a boy due to the presence of mucosal folds or a membrane extending across the urethra (posterior urethral valve). The patient presents in early infancy with distended bladder, dribbling, vomiting and failure to thrive. As a result of obstruction to urinary flow, male fetuses can have:
• Poor renal growth with reflux and dilated upper urinary tracts
Intrauterine intervention has no proven benefit and an early delivery is performed only if there are signs of rapidly progressing renal damage. Postnatal management includes:
Micturition
Normal micturition
Micturition is the intermittent voiding of urine stored in the bladder. It is an autonomic reflex that is under voluntary control. The inside of the bladder wall is folded and can expand and accommodate fluid with little increase in pressure. However, it can accommodate only a certain volume of fluid before an increase in intravesical pressure occurs, causing an urge to micturate. Fig. 5.8 shows a normal cystometrogram in which pressure rise is compared with rise in volume in the bladder.
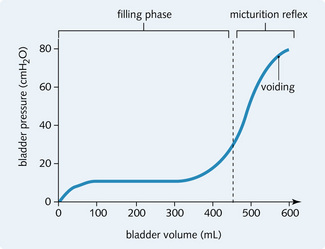
Fig. 5.8 Normal cystometrogram showing the rise in pressure associated with increasing bladder volume.
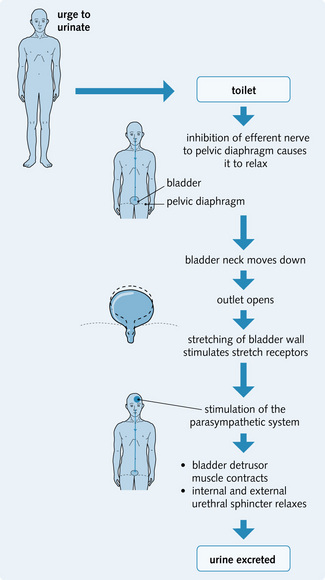
Fig. 5.5 shows the innervation of the bladder. In infants, micturition is a local spinal reflex in which the bladder empties on reaching a critical pressure. However, in adults this reflex is under voluntary control, so can be inhibited or initiated by higher centres in the brain. During micturition:
• Perineal muscles and the external urethral sphincter relax
Bladder distension with urine stimulates bladder stretch receptors, which, in turn, stimulate the afferent limb of voiding reflex and parasympathetic fibres of the bladder, resulting in the desire to urinate. Higher-centre stimulation of the pudendal nerves keeps the external sphincter closed until it is appropriate to urinate. Fig. 5.9 illustrates the voluntary control of micturition.
Neurological disorders of micturition
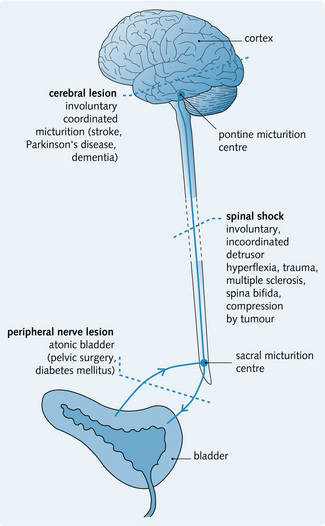
Urinary continence is affected by various neurological lesions along the micturition pathway as summarized in Fig. 5.10.
Lesion of afferent nerves from the bladder
A lesion of afferent nerves from the bladder (e.g. caused by disease of the dorsal roots such as tabes dorsalis) prevents reflex contractions of the bladder, so the bladder becomes distended, thin-walled and hypotonic.
Lesion of both afferent and efferent nerves
A lesion of both afferent and efferent nerves (e.g. because of a tumour of the cauda equina or filum terminale) results in:
This leads to a shrunken bladder with a hypertrophied bladder wall.
Spinal cord lesion
A lesion to the spinal cord above the sacral micturition centre (e.g. spinal shock sustained following trauma) results in:
• Initially: overflow incontinence, because of a flaccid and unresponsive bladder, which results in overfill and dribbling. This is spinal shock
• After shock has passed: the voiding reflex returns, but with no control from higher centres, so the patient has no voluntary control over voiding
• Occasionally: hyperactive voiding might be seen
• Eventually: bladder capacity falls and the wall hypertrophies – spastic neurogenic bladder.
Spina bifida
This is a developmental defect in which the posterior neural arches of the spine fail to develop, so part of the spinal cord and its coverings are exposed. It forms a spectrum of defects, resulting in varying degrees of bladder dysfunction.
Diabetes mellitus
Neuropathy is a common complication of diabetes. It can result in a loss of sensation, so there is no desire to micturate and the patient voids infrequently. This eventually leads to bladder distension, with overflow incontinence. The presence of residual urine increases the risk of infection.
Urinary incontinence
Urinary incontinence is the involuntary loss of urine. It affects more women than men and is a socially distressing condition. There are several different types.
Stress incontinence
This is involuntary leakage of small amounts of urine associated with an increase in intra-abdominal pressure (e.g. coughing, laughing, exercising). The sphincter is incapable of preventing the leakage. It is caused by:
• Pelvic floor laxity (usually the result of childbirth)
• Bladder neck sphincter impairment (more common in middle-aged, obese, multiparous women)
• Surgery affecting the urethra or prostate (e.g. TURP) causing damage or weakness to the external sphincter.
Pelvic floor exercise (e.g. Kegel) are an effective, non-invasive treatment but require cooperation and commitment from patients. Duloxetine, a serotonin and noradrenaline (norepinephrine) reuptake inhibitor, can be used for stress incontinence. There are a variety of options for surgical management, including tension-free vaginal tape (TVT) and transobturator tape (TOT) where the proximal urethra is lifted with an artificial sling, so that increases in intra-abdominal pressure compress the urethra.
Urge incontinence
This is the sudden strong urge to void followed immediately by involuntary loss of urine. It is part of overactive bladder syndrome, caused by detrusor overactivity. Causes include:
• Damage to the nervous system innervating the detrusor muscle, such as stroke, Parkinson's disease, Alzheimer's disease
• Inflammation of the lower urinary tract from infection or stones.
The management of detrusor instability involves the behavioural techniques of bladder drill and training, regulating then increasing the amount of time between voiding. Antimuscarinic medication, e.g. Oxybutynin is also used.
Overflow incontinence
This is involuntary leakage of urine when the bladder is full. It is usually due to chronic urinary retention secondary to obstruction or an atonic bladder. The causes are:
Nocturnal enuresis – bedwetting
Nocturnal enuresis is a childhood disorder. It can be primary (since birth) or secondary. Both can be due to:
After urinary tract infection has been excluded, no further investigation should be performed if the child is under 5 years – unless there are any urinary symptoms. Treatment should focus on behaviour therapy with star charts or alarms that wake the child when starting to micturate. Drug treatment with desmopressin, an ADH analogue, is a last resort.
Urinary tract obstruction and urolithiasis
Urinary tract obstruction
Obstruction in the urinary tract can occur at any level. It can be unilateral or bilateral, complete or incomplete, and of gradual or acute onset. It increases the risk of UTI, reflux and stone formation. If prolonged or unrelieved, obstruction can cause functional renal impairment and permanent renal atrophy.
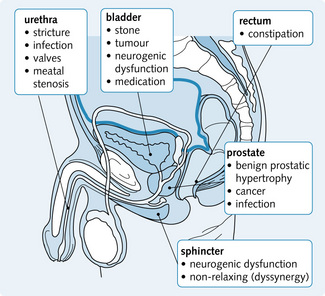
Fig. 5.11 shows the sites of obstruction in the urinary tract. Obstruction is caused by a congenital defect or, more commonly, by a structural lesion.
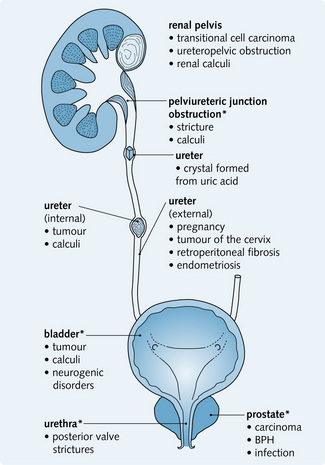
Fig. 5.11 Sites of obstruction in the urinary tract.* The most common sites of obstruction. BPH, benign prostatic hyperplasia.
Congenital abnormalities
These include the following neuromuscular defects:
Mechanical obstruction of the meatus and urethra occurs only in boys. Severe vesicoureteric reflux eventually results in upper renal tract dilatation without obstruction.
Tumours
Tumours can cause obstruction in two ways:
1. Internal: tumours within the urinary tract wall or lumen (e.g. bladder carcinoma). These occupy the urinary tract lumen, causing direct obstruction
2. External: pressure from rectal or prostate tumours or from gynaecological malignancies narrows the urinary tract lumen, causing indirect obstruction.
Pregnancy
The high levels of progesterone in pregnancy relax smooth muscle fibres in the renal pelvis and ureters and cause a dysfunctional obstruction. There might also be external compression from the pressure of the enlarging fetus on the ureters.
Hyperplastic lesions
The most common hyperplastic lesion causing urinary obstruction is benign prostatic hypertrophy (BPH).
Hydronephrosis
This is dilatation of the renal pelvis and calyces due to obstruction at any point in the urinary tract causing increased pressure above the blockage. It can be:
• Unilateral: caused by an upper urinary tract obstruction. This is detected late because renal function is maintained by the other kidney. Thus, the affected kidney can be severely impaired by the time obstruction is detected
• Bilateral: because of obstruction in the lower urinary tract. Renal failure develops earlier and prompt intervention is required to prevent chronic kidney disease.
Progressive atrophy of the kidney develops as the back pressure from the obstruction is transmitted to the distal parts of the nephron. The glomerular filtration rate (GFR) declines and, if the obstruction is bilateral, the patient goes into renal failure. Progressive damage to the renal structures results in flattening of the calyces with gradual thinning of the renal parenchyma, eventually leaving a cystic, thin-walled, fibrous sac with no functional capacity.
• Obstruction at the pelviureteral junction: hydronephrosis
• Obstruction of the ureter: hydroureter, eventually developing hydronephrosis
• Obstruction of the bladder neck/urethra: bladder distension with hypertrophy, eventually leading to hydroureter and thus hydronephrosis. If bilateral, renal failure develops earlier.
Presentation of urinary obstruction
This depends on the site and cause of the obstruction:
• An acute complete obstruction in the ureters (e.g. due to a stone) causes severe pain (renal colic). Only if bilateral, will the patient develop acute renal impairment
• Gradual obstruction (e.g. prostatic hypertrophy) causes bladder distension with hesitancy, terminal dribbling, poor urine flow and a sense of incomplete voiding
• A unilateral and partial obstruction causing a hydroureter or hydronephrosis might not be apparent for many years because the unaffected kidney maintains adequate renal function
• A bilateral and partial obstruction presents with nocturia and polyuria caused by tubular cell dysfunction with an inability to concentrate urine. Other chronic manifestations include renal stones, salt wasting, distal renal tubular acidosis and hypertension. If undiagnosed, the patient develops chronic renal failure
• A bilateral and complete obstruction presents as anuria or oliguria and must be treated urgently. Following removal of the obstruction, there may be a post-obstructive diuresis which can result in dehydration if not managed appropriately. Any general malaise or fever might be a sign of superimposed infection.
In all cases, the prompt and effective relief of the obstruction is essential to preserve the renal parenchyma. Depending on the site this may require a urinary catheter, urinary stent or a nephrostomy (to allow renal function to improve), followed by surgical intervention.
Urolithiasis (urinary calculi)
Urinary calculi affect 10% of the population and are more common in men and Caucasians. Stone formation is initiated by a core of mucoproteins or urates (nucleation); as more components precipitate on the core, the stone gradually increases in size (aggregation). Dehydration increases the concentration of the urine hence is a predisposing factor.
There are five main types of calculi:
• Calcium oxalate stones, the most common cause (Fig. 5.12). These are associated with hypercalcaemia and primary hyperparathyroidism and hyperoxaluria. The absence of citrate in the urine predisposes to these stones
• Mixed calcium phosphate and calcium oxalate stones. These are associated with alkaline urine, caused by renal tubular acidosis types 1 and 3
• Magnesium ammonium phosphate stones (MAP, struvite, infection stones), associated with urea-splitting bacteria, e.g. Proteus mirabilis
• Uric acid stones, associated with gout and myeloproliferative disorders
• Cystine stones occur in patients who have inherited (autosomal recessive) cystinuria.
They can form anywhere in the urinary tract. Occasionally, a calculus can grow to take up the shape of the renal pelvis and branch into calyces (staghorn calculus). The three most common sites for ureteric stones are:
Presentation
The clinical presentation depends on the site of the stone:
• Renal stones may cause a continuous dull ache in the loins
• Ureteric stones cause classic renal colic due to the increase in peristalsis in the ureters in response to the passage of a small stone. This pain typically radiates from the loin to groin. The patient appears sweaty, pale and restless, with nausea and vomiting
• Bladder stones cause strangury: the urge to pass something that will not pass
• Recurrent and untreatable UTIs, haematuria or renal failure
CT scan of the kidneys, ureters and bladder is now the gold standard for diagnosing stones (all but uric acid stones are radio-opaque). Intravenous urogram does still have advantages, as it is less expensive and gives a lower radiation dose (see Chapter 8).
Treatment
Management involves adequate analgesia and a high fluid intake. The urine should be sieved for analysis. Stones of 5 mm or less in diameter usually pass spontaneously; larger stones might require surgical intervention. Tamsulosin and nifedipine increase the likelihood of stones passing. Other treatment options include:
• Extracorporeal lithotripsy: shock waves are used to fragment the calculi into small pieces which will then pass out in the urine
• Ureteroscopic destruction or removal of stones
Prevention of further stone formation is achieved with a high fluid intake and correction of any underlying metabolic abnormality.
Inflammation of the urinary tract
Cystitis
Inflammation of the bladder (cystitis), as part of urinary tract infection (UTI) is very common. If it involves loin pain and fever this indicates infection of the kidneys (pyelonephritis), discussed in Chapter 3. UTIs are more common in boys in infancy because of congenital abnormalities; this reverses at puberty, with more females being affected thereafter because of urethral trauma and pregnancy. Women are particularly at risk of cystitis because they have a short urethra, but further investigation is required if infections are recurrent. Any UTI in children and men should be investigated to exclude an underlying renal tract abnormality. UTIs rarely progress to renal damage in adults if the renal tract is normal. After the age of 40, UTI again becomes more common in men because of prostatic disease, causing bladder outflow obstruction. Risk factors for cystitis include:
The typical irritative symptoms of cystitis are dysuria (pain on passing urine), frequency and urgency of micturition and suprapubic pain.
A diagnosis of UTI requires over 105 organisms/mL from a midstream urine specimen. In the majority of UTIs the infecting organism comes from the patient's own faecal flora (Fig. 5.13). Treatment involves a high fluid intake, regular bladder emptying and antibiotics.
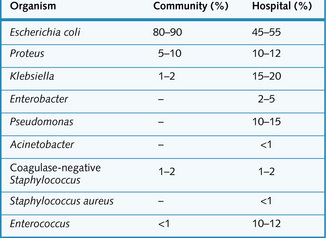
Fig. 5.13 Incidence of community- and hospital-acquired urinary tract infections (UTIs) caused by bacteria.
Non-infective cystitis can be caused by radiation, drugs (e.g. cyclophosphamide, ketamine) and instrumentation.
Chronic cystitis
This results from recurrent or persistent infection of the bladder. Chronic infection leads to fibrous thickening, so the bladder wall is less distensible. This affects the ability of the bladder to store urine and contract during micturition.
Interstitial cystitis
This type of cystitis is often associated with systemic lupus erythematosus, so is thought to be an autoimmune condition. As with all autoimmune conditions, it has a much higher incidence in women than in men. It can also result from recurrent and persistent infection that leads to fibrosis of all the layers of the bladder wall. There is often localized ulceration of the mucosa.
Malakoplakia vesicae
This is a very rare form of chronic bacterial cystitis, but it is important because it can mimic a tumour. Raised mucosal plaques of inflammation cells develop on the bladder and ureteric mucosa. These plaques are soft, yellow, 3–5 cm in diameter and are prone to ulceration.
Schistosomiasis (bilharzia)
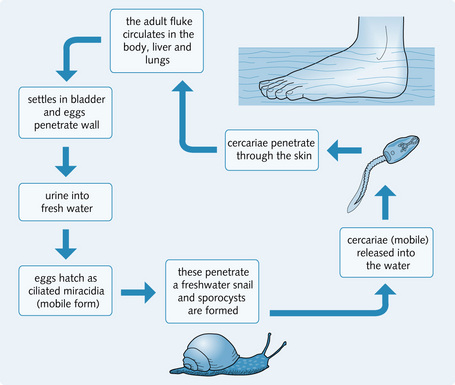
Schistosomiasis is the most common helminth infection worldwide, although it is rare in the UK. It is endemic in the Middle East, Africa, the Far East and in parts of South America. The pathogen is a blood fluke (Schistosoma haematobium). Freshwater snails are also part of its complicated life cycle. The schistosomes penetrate intact skin to enter the venous system, and migrate to the liver and bladder. They settle in the bladder to lay eggs causing chronic irritation of the transitional cells of the bladder. The eggs are excreted into local water supplies and transmitted through freshwater snails (Fig. 5.14).
People infected with cercariae can present with an itchy papular rash accompanied by myalgia, abdominal pain and headache. The most common presentation of infection with S. haematobium is recurrent haematuria. Eventually, urinary tract obstruction, bladder calcification (predisposing to squamous carcinoma) and CKD can occur.
Urethritis
Acute inflammation of the urethra occurs from infection with a sexually transmitted disease (e.g. Neisseria gonorrhoeae or Chlamydia trachomatis) (see Crash Course: Endocrine and Reproductive System for further details). It can be related to urethral diverticuli, urethral carbuncles or phimosis and can result in urethral stricture.
Disorders of the prostate
Prostatitis
There are three subgroups of inflammation of the prostate.
Acute prostatitis
The main pathogens are E. coli, Proteus and Staphylococcus species, and sexually transmitted pathogens including C. trachomatis and Neisseria gonorrhoeae. Inflammation can be focal or diffuse. Infection is usually spread from an acute infection in the urethra or bladder because of:
Occasionally, acute prostatitis is caused by a blood-borne infection.
On histological examination there is an acute inflammatory infiltrate of neutrophils and damaged cells, often resulting in abscess formation.
• General symptoms: malaise, rigours and fever
• Local symptoms: difficulty in passing urine, dysuria and perineal tenderness.
Rectal examination reveals a soft, tender and enlarged prostate. Diagnosis is based on the clinical features and a positive urine culture.
Chronic prostatitis
This results from inadequately treated acute infection. This can occur because some antibiotics cannot penetrate the prostate effectively. There is often a history of recurrent prostatic and urinary tract infections. Causative pathogens are the same as for acute prostatic infection.
Patients present with dysuria and low back and perineal pain, with no preceding acute phase. Some patients are asymptomatic.
Chronic prostatitis is difficult to diagnose and treat. Diagnosis is confirmed by:
Tuberculosis is a cause of chronic infection and can affect the kidneys or epididymis. Histological examination reveals focal areas of caseation and giant cell infiltrates.
Chronic non-bacterial prostatitis
This is the most common type of prostatitis and results in enlargement of the prostate, which can obstruct the urethra. The usual pathogen is C. trachomatis so, typically, sexually active men are affected. Often there is no history of recurrent UTIs.
Presentation is similar to that of chronic prostatitis and histological examination shows fibrosis as a result of chronic inflammation.
Diagnosis is confirmed by the presence of 15 white blood cells per high-power field (this indicates inflammation) and repeated negative bacterial cultures (excludes infection).
Benign prostatic hypertrophy
Incidence
BPH is detectable to some extent in nearly all men over the age of 60. It is a non-neoplastic enlargement of the prostate gland, which can eventually lead to bladder outflow obstruction. The cause is unknown, but might be related to levels of male sex hormones (testosterone).
Presentation
Symptoms develop as the enlarging gland compresses the prostatic urethra and the periurethral glands (known as the median lobe) swell, affecting the bladder sphincter mechanism. Men present with obstructive lower urinary tract symptoms (LUTS):
These symptoms can be caused by other conditions, shown in Fig. 5.15. Examination must include:
• Abdominal examination for an enlarged palpable bladder
• Digital rectal examination for the prostate, which is firm, smooth and rubbery.
Untreated BPH can present with acute urinary retention, which is accompanied by a distended and tender bladder and a desperate urge to pass urine. Alternatively, the patient might have progressive bladder distension, leading to chronic painless retention and overflow incontinence. If undetected, BPH can lead to bilateral upper tract obstruction and renal impairment, with the patient presenting in chronic kidney disease (see Chapter 7).
Pathology
There is hyperplasia of both the lateral lobes and the median lobes (these lie behind the urethra), leading to compression of the urethra and therefore bladder outflow obstruction. Within the prostate there are solid nodules of fibromuscular material and cystic regions. Histological examination shows hyperplasia of the:
Complications
The complications of BPH develop from prolonged obstruction to urine flow. There is compensatory hypertrophy of the bladder as a result of the high pressures that develop within the bladder (Fig. 5.16).
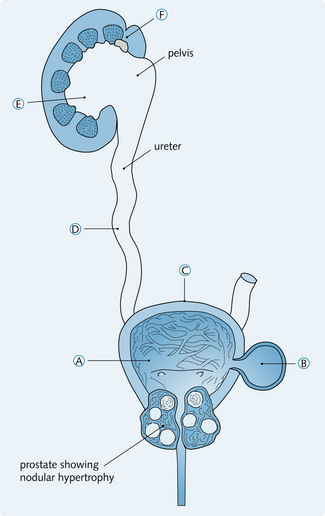
Fig. 5.16 Complications of benign prostatic hypertrophy. (A) Bands of thickened smooth muscle fibres cause trabeculation of the bladder wall. (B) Diverticula can develop on the external surface of the bladder. (C) Dilatation of the bladder once the muscle becomes hypotonic. (D) Formation of hydroureters resulting in the reflux of urine up to the renal pelvis. (E) Bilateral hydronephrosis. (F) Kidney infection, stones, calculi and renal failure.
Treatment
Medical treatment
Symptoms can be improved with α-blockers which relax smooth muscle at the bladder neck and within the prostate, thus improving urinary flow rate. Finasteride is a 5a-reductase inhibitor that prevents the conversion of testosterone to the more potent androgen dihydrotestosterone. Dihydrotestosterone promotes growth and enlargement of the prostate so inhibition of its production causes gradual reduction in prostate volume, thereby improving urinary flow rate and obstructive symptoms.
Surgical treatment
Transurethral resection of the prostate (TURP) is the most common operation for BPH. Complications include haematuria, retrograde ejaculation and impotence. A dilutional hyponatraenia can be seen following TURP as a result of absorbance of water and glycine from irrigation fluid used during the procedure. This can lead to a ‘transurethral resection syndrome’ of confusion, vomiting, hypertension and visual disturbance.