Chapter 24 Infections of the cardiovascular system
In health, the cardiovascular system is sterile, but a few organisms may enter the blood stream (even in health) during routine procedures such as tooth-brushing, especially in the presence of periodontitis. However, these bacteria have only a transient existence as the efficient defences of the blood quickly destroy them.
Bacteraemia, septicaemia and sepsis syndrome
Definitions
Septicaemia and sepsis syndrome
Aetiology
Some common predisposing factors and agents that cause septicaemia are shown in Table 24.1.
Table 24.1 Some common predisposing factors and agents of septicaemia
| Predisposing factor | Agent |
|---|---|
| Abdominal sepsis | Enterobacteria |
| Bacteroides fragilis | |
| Enterococcus faecalis | |
| Infected wounds, burns | Staphylococcus aureus |
| Streptococcus pyogenes | |
| Enterobacteria | |
| Osteomyelitis | Staphylococcus aureus |
| Pneumonia | Streptococcus pneumoniae |
| Intravascular devices | Staphylococcus aureus |
| Staphylococcus epidermidis | |
| Enterobacteria | |
| Food poisoning | Salmonella spp. |
| Campylobacter spp. | |
| Meningitis | Streptococcus pneumoniae |
| Neisseria meningitidis | |
| Haemophilus influenzae | |
| Immunosuppressed patients | Enterobacteria |
| Staphylococcus aureus, etc. |
Pathogenesis and clinical features
Once the blood stream is invaded by microbes, the host responds by activating its defence mechanisms, leading to the production of a cascade of inflammatory cytokines (e.g. interleukin-1, tumour necrosis factor; see Chapter 10). The cytokine release is orchestrated by endotoxins of Gram-negative bacteria, peptidoglycan of Gram-positive bacteria and exotoxins from both these groups. Generally, these cytokines are beneficial in eliminating the organisms, but excessive production may lead to organ dysfunction and circulatory septic shock – the sepsis syndrome.
Some of these patients are said to develop the systemic inflammatory response syndrome (SIRS) depending on their clinical signs; these include hypotension, fever, rigors, oliguria and renal failure. Sometimes the infection may trigger a pathological activation of the coagulation system (disseminated intravascular coagulation (DIC)) and due to the resultant consumption of platelets and clotting factors, severe bleeding disorders.
Diagnosis
Blood should be cultured for a diagnosis of septicaemia. As the number of organisms circulating in the blood may vary from time to time, depending on the disease condition, more than one blood culture may be required; whenever possible, this should be carried out before antibiotic therapy is instituted. Several positive cultures are required to ensure that the culture result is not due to contamination from the venepuncture site. Cultures from sites suspected to be causing the infection are useful (e.g. pus from an abscess) to establish and localize the infective focus.
Infections of the heart
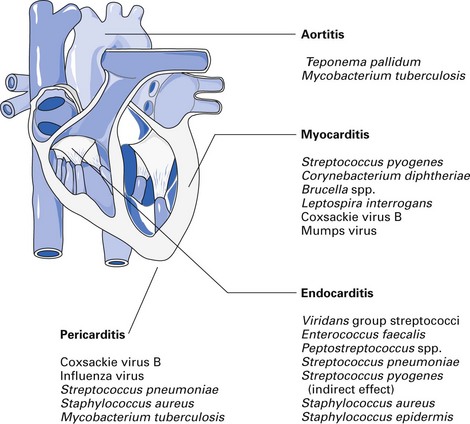
Important pathogens that cause pericarditis, myocarditis and endocarditis are shown in Figure 24.1. Of these, infective endocarditis is the most important disease of relevance to dentistry.
Infective endocarditis
Definition
Inflammation of the endocardium of the heart valves, and sometimes the endocardium around congenital defects, resulting from an infection.
Microbial aetiology
Bacteria are predominantly involved, although other organisms, such as fungi, rickettsiae and chlamydiae, may occasionally cause endocarditis (Table 24.2). More than 80% of infective endocarditis is caused by streptococci and staphylococci. The position held by the viridans group of organisms in the league table indicates the major role played by the oral commensals in causing this life-threatening disease. It is noteworthy that nearly all patients with viridans endocarditis have a previous heart lesion, and about a quarter give a history of a recent dental procedure as a precipitating factor.
Table 24.2 Causative microorganisms in infective endocarditis
| Microorganisms | Cases (%) |
|---|---|
| Total streptococci | 60 |
| Viridans group | 35 |
| Enterococcus faecalis | 13 |
| Microaerophilic streptococci | 3 |
| Anaerobic streptococci | 2 |
| Others | 7 |
| Total staphylococci | 25 |
| Staphylococcus aureus | 20 |
| Staphylococcus epidermidis | 5 |
| Miscellaneous | 5 |
| Culture-negative | 10 |
(cumulative data from several sources)
Clinical features
Although two clinical forms of the disease – acute and subacute – have been identified, the line of demarcation between these forms is not often clear. The acute form is a rapidly progressive condition and is caused by bacteria such as Streptococcus pneumoniae, Staphylococcus aureus and Streptococcus pyogenes. The subacute form is more insidious and chronic, and progresses rather slowly. The agents of this form of the disease are less virulent bacteria, such as viridans streptococci, Staphylococcus epidermidis and Enterococcus faecalis.
Signs and symptoms
The classic signs are fever, malaise, loss of weight, anaemia, splinter haemorrhages, petechiae, cardiac murmur, haematuria and splenomegaly.
Diagnosis
Clinical signs supported by positive blood culture are used to make the diagnosis. Repeated culture may be necessary to isolate the causal organism owing to the low-grade bacteraemia. If possible, blood should be collected when the temperature of the patient rises, indicating fever due to bacteraemia. At least 10 ml of blood should be collected prior to antibiotic therapy and cultured under aerobic and anaerobic conditions (see Fig. 6.4). Any agent isolated from two different blood culture sets (on separate occasions) is considered significant. Identification and antibiotic sensitivity tests are then performed on the isolate.
Pathogenesis and epidemiology
Infective endocarditis normally occurs in patients with some pathological condition of the endocardium, although those with apparently normal heart valves may rarely be affected. The predisposing conditions include valve prostheses, septal defects, atheroma of the valve, congenital valve deformities and pre-existing rheumatic fever (Table 24.3). Infective endocarditis is the end result of the sequential interaction of events shown in Figure 23.2:
Table 24.3 Cardiac valvular disease predisposing to infective endocarditis
| Disease | Degree of risk |
|---|---|
| Aortic valvular disease | High |
| Prosthetic valves | |
| Mitral insufficiency | |
| Ventricular septal defect | |
| Patent ductus arteriosus | |
| Coarctation of aorta | |
| Previous infective endocarditis | |
| Mitral valve prolapse and stenosis | Intermediate |
| Pulmonary and tricuspid valve disease | |
| Degenerative (calcific) aortic valve disease | |
| Non-valvular intracardiac prosthetic implants | |
| Atrial septal defect | Low/negligible |
| Coronary artery disease | |
| Cardiac pacemakers | |
| Arteriosclerotic plaques |
Treatment
High-dosage single or combination antibiotic therapy, guided by the microbiological findings from the blood culture, is necessary. The antibiotic regimen selected should be:
The rationale behind management is:
Infective endocarditis and dentistry
The oral cavity acts as a portal of entry for organisms causing bacteraemia, and dental manipulations may set in motion the disease process leading to infective endocarditis. Bacteraemia can occur after dental procedures such as extractions, surgical or non-surgical endodontics, gingivectomy, root-planing, scaling and flossing, intraligamentary injections, and reimplantation of avulsed teeth. The frequency of bacteraemia is also related to the preoperative oral sepsis of the patient and the degree of trauma and tissue injury; a routine activity such as tooth-brushing may also cause bacteraemia, depending on the degree of oral sepsis.
The real risk of development of infective endocarditis in a ‘risk’ patient following dental procedures is difficult to ascertain, and the evidence base is rather contradictory; it has been estimated that bacteraemias vary between 10% and 90%. Clearly, a proportion of infections is associated with random transient bacteraemias that commonly follow mastication, and even tooth-brushing, in patients with chronic periodontitis.
Infective endocarditis prophylaxis
As eventual development of endocarditis may well be the most common potentially fatal complication of dental treatment, all dentists must have a good working knowledge of the problem and the appropriate preventive measures.
Accurate identification of at-risk patients
The main risk conditions are shown in Table 24.3. Dentists usually identify patients at risk from their medical history. It is also important to obtain confirmatory and expert information from the patient’s medical practitioner.
Patient awareness of risk status and dental involvement in cardiac clinics
Warning cards given to patients with cardiac disease increase their awareness of the disease. Dentists should be part of the medical team involved in the preoperative and post-operative management of patients undergoing cardiac surgery who are at risk.
Preventive dental care
Susceptible patients should be exposed to risky operative procedures as rarely as possible; this can be best achieved by careful and intensive oral hygiene instruction, dietary advice and regular dental examinations. The aim should be to reduce the amount of treatment to the absolute minimum necessary for the maintenance of a healthy natural dentition for life. The need to administer prophylactic antibiotics for dental procedures that could produce a bacteraemia capable of initiating infective endocarditis must be weighed carefully, and the respective guidelines in each jurisdiction should be strictly adhered to.
Awareness of post-operative morbidity
Even when antibiotic cover has been provided, patients at risk should be instructed to report any unexplained illness because of the insidious origin of infective endocarditis.
Cardiac patients who need antibiotic prophylaxis
There is some controversy as to the need of antibiotic prophylaxis solely to prevent infective endocarditis in people at risk, undergoing either dental or non-dental procedures. Hence, the recent British recommendations (National Institute for Health and Clinical Excellence (NICE) Guidelines), as opposed to the American, state that there is no need for antibiotic prophylaxis for any dental procedure due to the following reasons:
Despite the foregoing, American authorities still maintain that antibiotic prophylaxis for infective endocarditis is necessary for a relatively small group of patients and as this textbook is used worldwide in various jurisdictions, the current US guidelines are provided below.
According to the American Heart Association, the following groups of patients are the only cohorts that should be given antibiotic prophylaxis and include those with:
Dental procedures that need antibiotic prophylaxis
The above groups of patients must be given antibiotics for all dental procedures that involve manipulation of gingival tissue or the periapical region of teeth or perforation of the oral mucosa.
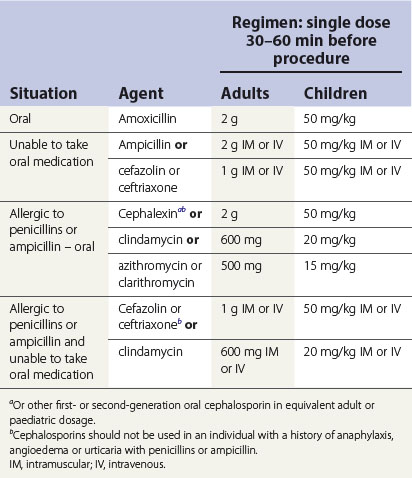
Presurgical antibiotic regimens for a dental procedure as per the American Heart Association Guidelines are given in Table 24.4. It should be borne in mind that these recommendations are regularly reviewed by the authorities, and practitioners need to keep abreast of such developments.
Antibiotic prophylaxis for miscellaneous conditions
Prosthetic cardiac valve
The dental management of patients with a prosthetic cardiac valve can be undertaken by dentists (as described above) as long as the patients require local anaesthesia and are not hypersensitive to penicillin. If the patient has received penicillin more than once within the past month, oral clindamycin should be given.
Hip joint replacements
There are few data on post-operative infection of hip prostheses to suggest that bacteria derived from the mouth are involved. There is wide consensus that patients with prosthetic joint implants, including total hip replacements, do not require antibiotic prophylaxis, because the risks of prophylaxis outweigh the benefits. Nevertheless, it is important that the possible need for prophylactic cover should be discussed with the patient’s doctor before dental treatment starts. Further, there should be liaison between orthopaedic surgeons and dentists to render patients dentally fit prior to insertion of replacements or implants.
Third molar surgery
A number of properly controlled trials have conclusively indicated that antimicrobial agents have no statistically significant effect on swelling, pain, trismus or post-operative infection in third molar surgery.
Dental implants
Surgical placement of dental implants is an elective procedure performed under relatively aseptic conditions. Hence, it does not warrant antibiotics either pre- or post-surgically, although many surgeons prefer to do so worldwide. A number of reviews have indicated that pre- or post-surgical antibiotics during implant placement is of dubious value.
Key facts
Lever A., Mackenzie I. Sepsis: definition, aetiology and diagnosis. British Medical Journal. 2007;335:879-883.
Martin M.V., Kanatas A.N., Hardy P. Antibiotic prophylaxis and third molar surgery. British Dental Journal. 2005;198:327-330.
2010 . National Institute for Health and Clinical Excellence (NICE) Guidelines: Prophylaxis against infective endocarditis. http://guidance.nice.org.uk/CG64/PublicInfo/pdf/English, 2010. (accessed 5th May 2011).
Oliver R., Roberts G.J., Hooper L. Penicillins for the prophylaxis of bacterial endocarditis in dentistry (Cochrane review). Australian Dental Journal. 2004;49:3.
Wilson W., et al. Prevention of infective endocarditis: Guidelines from the American Heart Association. Journal of the American Dental Association. 2007;138:739-760.