Chapter 37 Infection control procedures in dentistry
Implementation of standard infection control in dentistry (previously termed universal precautions) entails prevention of infection transmission within the dental clinic environment, and assumes that ALL patients are carriers of infectious diseases. Such a policy protects both patients and staff, reduces staff concerns and prevents discrimination against patients. In this chapter, the major features reflecting the best current practice of standard infection control are outlined, but the reader is strongly advised to keep up to date with the literature because of the rapidity of changes that occur in this area.
Practice management and staff development
All staff who join a practice should undergo a formal education programme that includes the theory and practice of infection control in dentistry. In addition, a written infection control protocol specific for the practice should be available for inspection by patients and other interested parties.
An in-service training programme, updating techniques and material, should be provided for the staff. This may take the form of regular attendance at local scientific meetings and access to current information such as journals and the internet.
Infection control: specific practical features
There are a number of elements in a comprehensive infection control protocol:
Patient evaluation
A thorough medical history should be taken from each patient and updated at each recall visit. It is not only good clinical practice but may also reveal disease that is important in relation to cross infection and relevant to the dental procedure to be undertaken. If a questionnaire is used for this purpose, it should always be supported by direct discussions with the patient. The medical history should not be used to categorize patients as high- or low-risk, as was the procedure prior to the introduction of standard infection control. In taking a history, the practitioner should identify the infectious disease of concern, and relevant questions should be asked in an environment conducive to the disclosure of sensitive personal information. It is also important that:
Personal protection
This subject is dealt with under the following headings:
Personal hygiene
The personal hygiene of all members of staff who are either directly or indirectly in contact with patients should be scrupulous. A rigidly followed code of hygiene will greatly reduce cross infection in the dental clinic. In general, when working with patients, dental personnel should observe the following precautions:
Hand care
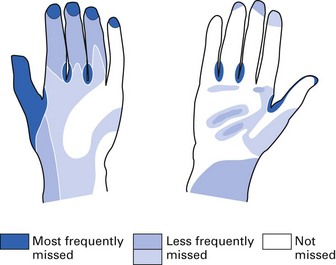
Fingers are the most common vehicles of infection transmission. This fact is poorly recognized by all.
The whole dental team should pay attention to meticulous hand care:
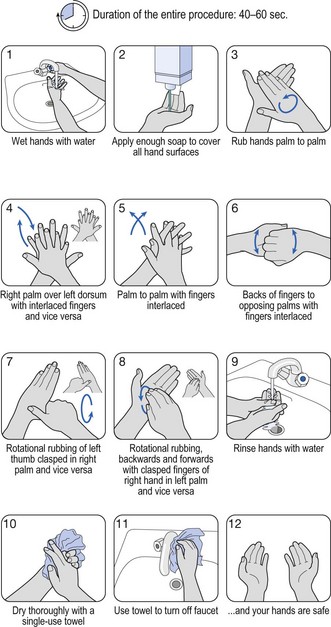
Fig. 37.1 A schematic presentation of a suggested hand-washing technique.
(Courtesy of the World Health Organization.)
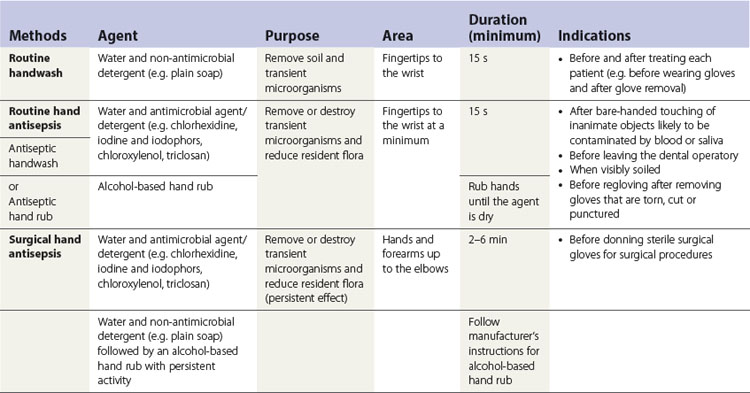
A ready reckoner for hand hygiene and antisepsis is provided in Table 37.1.
Clinic clothing
A freshly laundered uniform or overgarment should be worn by all clinical personnel. Garments should be changed at least daily, and more frequently if they become visibly contaminated. Renewable overgarments should be washed at an appropriate temperature in a well-maintained washing machine. Grossly contaminated clothing should be dealt with separately.
Wear overgarments only in the clinic premises, not in corridors, canteens or lifts. An additional waterproof vinyl apron could be worn to protect the overgarment when working in the instrument-cleaning area or the laboratory (e.g. denture-trimming).
Barrier protection
Personal hygiene measures reduce the level of possible pathogens on our bodies and clothes, although they do not completely eliminate them. In order to minimize further the spread of organisms from staff to patients (and vice versa), the following protective barriers should be used:
Gloves
All dentists and close support personnel should routinely wear disposable latex or vinyl gloves. The main aim of wearing gloves in routine dentistry is not to achieve consistent surgical sterility but to establish reasonable standards of hygiene in order to safeguard both the dental personnel and the patient.
The efficacy of gloves greatly diminishes if they are perforated. As gloves may perforate during surgical procedures, it is advisable to change gloves at least hourly during long operative procedures on the same patient. Gloves should be checked for visible defects immediately after wearing them, and immediately changed when breaches occur; never wash and reuse gloves. Rarely, allergic reactions to gloves may develop in staff or patients. Skin creams, a spray-on microfilm on the skin or a cotton glove liner may help these individuals.
There are three main types of gloves used in dentistry: their different uses should be clear:
Care should be taken to prevent contact between gloves and incompatible material (e.g. some impression materials) or naked flames.
Gloves should be removed as soon as patient contact is over. The hands should then be washed and rinsed thoroughly, and hand cream should be applied to prevent excessive drying of the skin. In addition, dental personnel should wash their hands with an antimicrobial handwash before leaving the clinic. Dental personnel with exudative lesions or weeping dermatitis should refrain from all direct patient care and from handling equipment until the condition resolves.
A new pair of gloves should be worn for each patient. Gloves should never be reused, as this will result in defects that will diminish their value as an effective barrier, and adequate removal of previous patients’ pathogens cannot be guaranteed. Treat gloves as surgical waste and dispose of them accordingly.
Contact dermatitis and latex hypersensitivity
All health care workers should be educated on the signs, symptoms and diagnoses of skin reactions associated with frequent hand hygiene and glove use. Patients should be screened for latex allergy through a health history questionnaire and referred for medical consultation when latex allergy is suspected. Emergency treatment kits with latex-free products should be available at all times.
Eye shields
Eye shields should be worn by dentists and close support personnel during all procedures to protect the conjunctivae from spatter and debris generated by high-speed handpieces, scaling (manual or ultrasonic), and polishing and cleaning of instruments:
Face masks
Wearing a face mask, such as a surgical mask, is a necessary hygienic measure, particularly during high-speed instrumentation, as it prevents inhalation of contaminated aerosols that might lead to both upper and lower respiratory tract infections. The filtration efficacy of such aerosols depends upon:
Always ensure that masks are well adapted so that the nose and mouth are completely covered. Masks with metal inserts are preferable as they can be tailored to fit the individual’s profile.
Masks should not be touched with gloves during treatment or worn outside the treatment zone; they should be worn beneath face shields as the latter provide only minimal protection from aerosols.
Rubber dam isolation
As far as possible, a rubber dam should be used in operative procedures to minimize saliva and blood-contaminated aerosol production. Use of a rubber dam during operative procedures:
A note on pre-procedural mouthrinse
Chlorhexidine gluconate (0.1–0.2%), essential oils or povidone–iodine mouthwash prior to a surgical procedure is recommended by some, to reduce the intraoral microbial load leading to systemic bacteraemias as well as the number of airborne pathogens. There is no scientific evidence to indicate that pre-procedural mouth rinsing prevents or reduces clinical infections among care providers or patients. However, studies have demonstrated that a pre-procedural rinse with an antimicrobial product can reduce the level of oral microorganisms in aerosols and spatter generated during routine dental procedures with rotary instruments (e.g. dental handpieces or ultrasonic scalers).
Handling sharps and related injuries
Numerous objects with sharp edges are used in dentistry (e.g. needles, blades, burs, endodontic files, orthodontic wires and matrix bands). A list of all the types of sharps used in the practice should be kept, identifying those that are disposable and those that may be reused and hence need to be processed. Sharps containers of approved type should be used in each working area and kept as close as possible to the point of use. They should not be overfilled and must be properly closed to prevent tampering, and they must be disposed of as clinical waste, ideally by incineration.
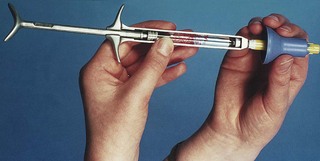
Extreme care should be taken when re-capping needles; a single-handed ‘bayonet technique’ or a resheathing device (Fig. 37.3) should be used for this purpose. The dental team should be conversant with all sharps handling procedures, which should be an integral part of ongoing staff education.
Sharps injury protocol
All sharps injuries should be recorded in a designated register and followed up. A standard protocol for sharps injury should be displayed clearly and at least one staff member assigned the responsibility for providing post-injury counselling, in the first instance. However, detailed counselling should be provided by a specialist in this field, to allay any residual concerns. Guidelines for the management of sharps injuries are shown in Table 37.2.
Table 37.2 Principles guiding the management of sharps injuries
|
Action by occupational health authority
• Record in detail circumstances of the sharps injury (i.e. demographic information of the exposed worker, details of the exposure)
|
HIV, human immunodeficiency virus.
Immunization procedures
Practitioners should have a written policy on the vaccination (including administration of boosters) of all staff and maintain an up-to-date immunization record of themselves and their staff, which should be kept confidential. Staff who refuse vaccination and follow-up tests should be counselled regarding the implications of this course of action, and a signed acknowledgement to the effect should be kept on file. A list of vaccines that are available to dental health care workers is shown in Chapter 10 (Table 10.2). In the UK, vaccination against hepatitis B virus, tuberculosis and rubella (for women) has been recommended for clinical dental staff, in addition to routine immunization against tetanus, poliomyelitis and diphtheria. In the USA, immunization against all the conditions listed, except tuberculosis and influenza, is recommended. A brief outline of vaccines available to dental personnel is given below.
Bacille Calmette–Guérin vaccine
Organism
Active against Mycobacterium tuberculosis. The vaccine contains live Mycobacterium bovis (termed bacille Calmette–Guérin or BCG) attenuated by propagation in a bile-potato medium. Killed vaccines do not produce the cell-mediated immune response essential for protection against tuberculosis.
Poliomyelitis vaccine
Organism
Live poliovirus types 1, 2 and 3 – Sabin vaccine (used in the UK) or killed poliovirus – Salk vaccine (used in developing countries and Scandinavia).
Measles–mumps–rubella vaccine
Indications
All children in the second year of life, to prevent complications of common childhood fevers, such as respiratory tract infection and encephalitis associated with measles, meningitis associated with mumps and congenital infections associated with rubella. The last is especially relevant for women of child-bearing age working in dentistry.
Triple vaccine: diphtheria–tetanus–pertussis
Organism
Three-in-one vaccine for prevention against diphtheria caused by Corynebacterium diphtheriae, whooping cough caused by Bordetella pertussis and tetanus caused by Clostridium tetani. Contains killed B. pertussis and diphtheria and tetanus toxoid.
Tetanus toxoid
Indications
Active immunization of the entire population. Although the disease is rare, tetanus can develop after very trivial wounds.
Hepatitis B vaccine
Organism
The surface antigen of the hepatitis B virus, HBsAg (see Chapter 29), manufactured in yeasts by genetic recombination and absorbed on to aluminium salt. Successful vaccination also offers protection against delta hepatitis (hepatitis D).
Indications
All health care workers who are at special risk, including dentists, dental hygienists, dental surgery assistants, medical laboratory workers and those handling blood products. In countries in South-East Asia where the disease is endemic, blanket vaccination programmes of all infants have been introduced in the hope of eradicating the disease.
Administration
Three doses (two doses at an interval of 1 month, followed by a third 6 months later) intramuscularly in the deltoid.
Protection
About 95% response rate. If antibody levels are suboptimal, then a fourth (booster) dose may be given. Individuals having the initial course of vaccination should undergo pre- and post-immunization tests, and those who fail to seroconvert should be followed up as appropriate.
There is controversy over the necessity of booster doses. Some authorities in the UK advocate boosters after 3–5 years, depending on the degree of initial antibody production, whereas others, especially in the USA, contend that booster doses are unnecessary because of the anamnestic response of the immune system.
Passive immunization with hepatitis B immunoglobulin
Passive immunization with hepatitis B immunoglobulin (HBIG) should be instituted within 48 h if an unprotected health care worker sustains an accident with blood or saliva containing hepatitis B antigens. This should be followed by a complete course of the hepatitis B vaccine, the first dose of which may be administered immediately or within 7 days of the accident. If the person declines the vaccine, then a second dose of HBIG should be administered 1 month after the first dose.
Influenza vaccine
Organism
Usually contains two of the influenza A virus strains that are currently circulating, together with the influenza B strain. It is important to recognize that, because of the phenomenon of antigenic ‘drift’ and ‘shift’ seen in influenza viruses, the vaccine composition needs to be reviewed and altered each year, which is a formidable task. The vaccine contains partially purified, disrupted virus particles or the surface antigens (haemagglutinin and neuraminidase).
Occupationally acquired infections
Health care workers routinely run the risk of acquiring infections by virtue of their profession – so-called occupationally acquired infections. Particular concerns for health care workers are blood-borne viral infections, including hepatitis B and C, and human immunodeficiency virus (HIV) infection. Hepatitis B infection used to be about 10 times more common among dental health care workers than the public, but with the advent of the extremely effective hepatitis B vaccine, this danger is minimal. The average risks of transmission of these diseases after percutaneous exposure to blood are:
Thus, hepatitis B is most infectious and the least infectious in this context is HIV.
Other than viral infections, bacterial infections such as tuberculosis and legionella infections may be acquired by dental care workers, although the evidence for these is rather circumstantial.
A note on sterilization, disinfection and antisepsis
The reader should clearly bear in mind the following basic definitions of sterilization, disinfection and antisepsis as these terms are frequently used in clinical dentistry.
In general, sterilization involves extensive treatment of equipment and materials, and is costly and labour-intensive. It is dependent on:
All instruments and appliances used in dentistry should ideally be sterilized, although some items of equipment and certain surfaces (e.g. bracket tables attached to the dental chair) do pose problems. In such circumstances, the best alternative is to disinfect the items or surfaces concerned.
Decontamination (synonym: reprocessing)
Decontamination is the process by which reusable items are rendered safe for further use and for staff to handle. Decontamination is required to minimize the risk of cross infection between patients and between patients and staff. The term decontamination (as opposed to sterilization and disinfection) has gained popularity particularly in European regions and is less widely used in North America. Decontamination is a complex and an exacting process and entails:
Decontamination of instruments
Receiving, cleaning and decontamination
The removal of contaminated instruments and equipment from the treatment area should follow a set routine, avoiding cross-contamination between the soiled and sterilized instruments. Once an effective method of instrument or equipment flow has been worked out, this method should be strictly adhered to.
Reusable instruments, supplies and equipment should be received, sorted, cleaned and decontaminated in one section of the processing area. Cleaning should precede all disinfection and sterilization processes and should involve removal of debris as well as organic and inorganic contamination.
Removal of debris and contamination is achieved either by:
If visible debris, whether inorganic or organic matter, is not removed, it will interfere with microbial inactivation and can compromise the disinfection or sterilization process. After cleaning, instruments should be rinsed with water to remove chemical or detergent residue.
Considerations in selecting cleaning methods and equipment include:
Note that the use of automated cleaning equipment such as an ultrasonic cleaner or washer disinfector does not require presoaking or scrubbing of instruments. These instruments therefore:
Presterilization cleaning
Whenever possible, cleaning should be performed using an automated and validated process in preference to manual cleaning. Manual cleaning should only be considered where manufacturer’s instructions specify that the device is not compatible with automated processes. Heavy duty household utility gloves must be used when cleaning instruments; eye protection and face masks are also desirable. Instruments should be cleaned as soon as possible after use. If immediate cleaning is not feasible, placing instruments in a puncture-resistant container and soaking them with detergent, a disinfectant/detergent, or an enzymatic cleaner will prevent drying of patient material and make cleaning easier and less time-consuming. Use of a liquid chemical sterilant/high-level disinfectant (e.g. glutaraldehyde) as a holding solution is not recommended.
Sharps should be handled with extreme care during scrubbing to prevent injury to the hands. Uncapped needles should never be left on the instrument tray, and after use, these and other sharps should be placed directly in puncture-resistant containers. Work-practice controls should include use of a strainer-type basket to hold instruments and forceps to remove the items.
Automated cleaning using washer disinfectors (Fig. 37.4b)
A washer disinfector is the preferred method for cleaning dental instruments as it offers the best option for the control and reproducibility of cleaning; a typical washer disinfector cycle for instruments includes the following five stages:
Preparation and packaging
In a separate section of the processing area, cleaned instruments and other supplies should be inspected; assembled into sets or trays; and wrapped, packaged, or placed into container systems as appropriate for sterilization. Instruments used in dentistry may be packaged for sterilization using:
Prior to packaging, all hinged instruments should be opened and unlocked. An internal chemical indicator should be placed in every package. In addition, an external chemical indicator (e.g. chemical indicator tape) should be used when the internal indicator cannot be seen from outside the package. For unwrapped loads, at a minimum, an internal chemical indicator should be placed in the tray or cassette with items to be sterilized. Dental practices should refer to the manufacturer’s instructions regarding use and correct placement of chemical indicators. Critical and semicritical instruments that will be stored should be wrapped or placed in containers (e.g. cassettes or organizing trays) designed to maintain sterility during storage.
Sterilization
The sterilization process
In dentistry, sterilization is usually achieved by one of three methods:
Other sterilization methods, not used in dentistry, are ethylene oxide gas and gamma-irradiation (employed by commercial suppliers of plastic goods), and filtration (used for sterilization of injectable drugs).
Moist heat sterilization (steam under pressure)
Steam is a very effective sterilizing agent as it:
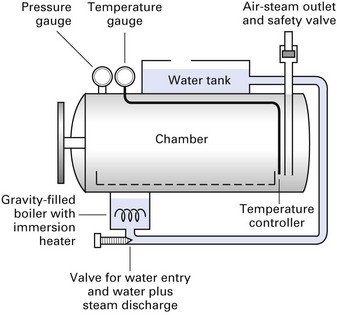
When water is heated in a closed environment, its boiling point is raised, together with the temperature of the generated steam; for example, at 104 kPa (15 psi), the steam temperature is 121°C. This phenomenon is utilized in steam sterilization by the autoclave (Fig. 37.5). Put simply, an autoclave is a glorified domestic pressure cooker with a double-walled or jacketed chamber; steam circulates under high pressure inside the chamber, in which the objects for sterilization (the load) have been placed. Once the sterilization cycle is complete, drying the load is accomplished by evacuating the steam. Drying can be accelerated by the suction of warm, filtered air into and through the chamber. It is important to expel the air in the chamber at the beginning of a sterilization cycle because:
There are two types of autoclaves:
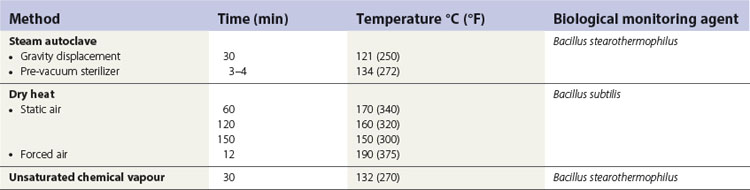
Examples of sterilization times and temperature for autoclaves are shown in Table 37.3. Of the options given, a sterilization cycle of 134°C for 3–4 min at 207 kPa is recommended for both wrapped and unwrapped dental instruments.
Autoclaves used in dentistry
Three different types of autoclaves are used in dentistry; these are:
The sterilization cycle
The sterilization cycle (either in an autoclave or a hot-air oven) can be divided into three periods (Fig. 37.6): the heating-up period, the holding period and the cooling period. For the bench-top autoclave (routinely used in dentistry), this entails:
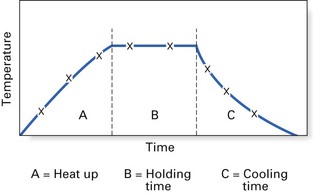
Fig. 37.6 The stages of a full sterilization cycle. (A) Heat up; (B) holding time; (C) cooling time.
Notes on the proper use of bench-top autoclaves
Sterilization with dry heat
Dry heat penetrates less well and is less effective than moist heat; consequently, higher temperatures and longer times are required for sterilization. The total time for heating up, holding and cooling may be several hours (Table 37.3). It is therefore essential that hot-air ovens should have a time lock on the door so that items cannot be added or removed during the cycle, and a fan to distribute the heat evenly. Dry-heat sterilizers used in dentistry include static-air and forced-air types:
Chemical vapour sterilization (chemiclave)
A combination of formaldehyde, alcohols, acetone, ketones and steam at 138 kPa serves as an effective sterilizing agent. (The premixed chemicals must be purchased from the manufacturer as their balance is critical.) Microbial destruction results from the dual action of the toxic chemicals and the heat. In general, chemical vapour units sterilize more slowly than autoclaves (30 min versus 15–20 min, for packaged instruments) but are faster than hot-air ovens. The usual temperature and pressure combinations are 127–132°C at 138–176 kPa for a period of 30 min, once the correct temperature has been attained (Table 37.3).
This process cannot be used for materials or objects that can be altered by the chemicals or are made of heat-sensitive material. Rusting is unusual if instruments are dried before sterilization as there is relatively low (7–8%) humidity throughout the process.
The major advantages of the chemiclave are that it is faster than dry-heat sterilization, it does not corrode instruments or burs, and dry instruments are available as soon as the cycle is over. Adequate ventilation must be provided in order to dispel the residual fumes released on opening the chamber at the end of the cycle.
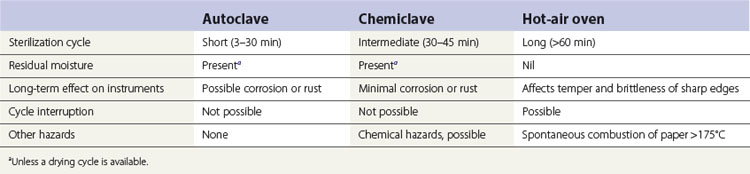
The advantages and disadvantages of sterilization using autoclave, chemiclave and hot-air oven are summarized in Table 37.4.
Monitoring sterilization
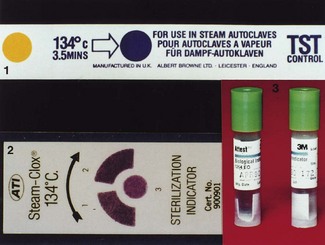
Achievement of the requisite temperature and pressure, as indicated by the gauges of the autoclave (or any other sterilizer), does not guarantee that the entire load has been sterilized. All sterilization procedures must therefore be carefully and regularly monitored so that failures are detected and sterility is assured. The indicators used for checking sterility are (Fig. 37.7):
Process indicators are materials (either liquid or paper) that change colour on exposure to the appropriate sterilization cycle, indicating that the load has been processed. Note that process indicators do not prove sterilization but merely verify that the items have been subjected to the processing conditions; thus, the main function of a process indicator is to assure the operator that the material has gone through a sterilization cycle. At least one process indicator should be cycled with every sterilization load, and the results should be documented in a sterility control file.
In contrast to process indicators, biological monitors are designed to prove sterilization. The indicators used in this system are bacterial spores (Table 37.3), which require high temperatures for extended periods to lose their viability (the corollary is that, if the spores are killed, then less-resistant microbes are killed more readily and sterility is achieved).
Biological monitoring or spore tests should be used on a weekly basis in dentistry. The monitor should be placed in the sterilizer at a point where sterilization is most difficult to achieve (e.g. inside bags or trays). After cycling, each strip should be sent for culture or cultured in the clinic according to the manufacturer’s instructions. The results of biological monitoring should be routinely recorded and kept in a sterility control file. Spore tests should also be done when commissioning a new autoclave, after servicing or repairs and as part of the training of new staff.
Quality control of small bench-top autoclaves
Small autoclaves should be operated to ensure that they are:
Daily tests of small autoclaves
The daily tests should be performed by the user and will normally consist of:
The Bowie–Dick test is used in vacuum autoclaves to check the steam penetration into the centre of the autoclave load and to signal the presence of any air pockets.
Storage and care of sterile instruments/devices
Once sterilized, the instruments or devices should be maintained in a sterile state until they are used again. The proper storage of sterile instruments is therefore as important as the sterilization process itself; improper storage would break the ‘chain of sterility’ and introduce the possibility of pathogenic recolonization risk. A barrier(s) should be maintained between the instruments and the general practice environment. The following guidelines should be followed in storing sterile instruments/devices:
Disinfection
Methods of disinfection consist of:
Disinfection by heat
Pasteurization
Pasteurization is named after Louis Pasteur’s discovery that mild heating prevents the spoilage of wine by selective killing of unwanted microbes. A similar treatment is now applied to milk to delay souring due to microbial activity. Milk is raised to a temperature of either 63–66°C for 30 min or (in the flash method) to 72°C for 15 s. This procedure renders the milk safe from contamination with M. tuberculosis, Campylobacter and other pathogens. It should be noted that pasteurization is not a sterilization process.
Physical methods: ultrasonics
Ultrasound is an effective way of disrupting microbial cell membranes and is used for removing debris before autoclaving.
Chemical methods
Choosing a chemical disinfectant should be done carefully because a disinfectant used for one purpose may not be equally effective for another. Further, the antimicrobial activity of a chemical disinfectant falls drastically in the presence of organic debris. Products that usually disinfect items or surfaces may not do so when there is heavy contamination, particularly with resistant microbes in large numbers. The residual levels of organisms following disinfection may still represent an infection risk to unusually susceptible patients.
Mode of action of chemical disinfectants
The chemicals used as disinfectants generally behave as ‘protoplasmic poisons’ in three different ways:
Conditions determining the effectiveness and choice of a disinfectant
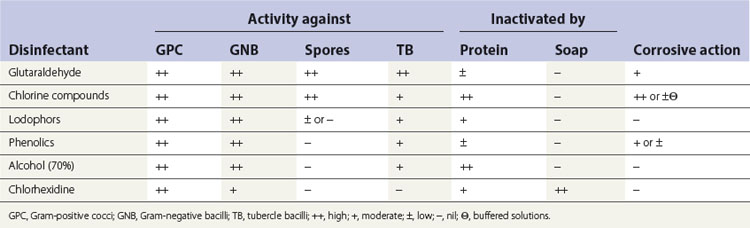
Spectrum of activity
Disinfectants vary widely in their activity; e.g. some are more active against Gram-positive than Gram-negative bacteria (Table 37.5).
Satisfactory contact
All contaminated surfaces should come into contact with the disinfectant for the specified period. Organic debris, air and greasy material may prevent this, hence the importance of thorough cleaning of the material or instrument before disinfection.
Concentration
Adequate concentration of disinfectants is essential, and they should always be accurately dispensed. It is important to use the manufacturer’s stated dilution of the disinfectant.
pH
The activity of a disinfectant is often pH-dependent (e.g. glutaraldehydes act only at alkaline pH, whereas phenols work best at acid pH).
Neutralization
A wide range of substances, including hard water, soaps and detergent, may neutralize the disinfectant.
Stability
Not all disinfectants are stable, especially when diluted, and may deteriorate with age or storage. Solutions should be freshly prepared for use and marked with an expiry date.
Speed of action
In general, disinfectants act slowly, and their activity depends on the concentration used. Hypochlorites have a rapid action but are corrosive at high concentrations. Glutaraldehyde is slow-acting but is an effective sporicidal agent.
Potency of disinfectants and their uses
Disinfectants can be generally categorized as having high, intermediate or low potency, depending on their ability to kill various groups of organisms:
A rough guide to the use of these three categories of disinfectants is given below.
Step 1
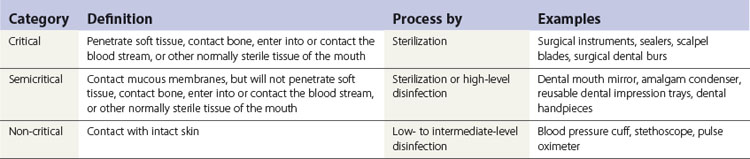
Categorize the items that require disinfection or sterilization into three groups (Table 37.6):
Disinfectant and antiseptic agents commonly used in dentistry
Alcohols
Ethyl alcohol or propyl alcohol (70%) in water is useful for skin antisepsis prior to cannulation, injection and surgical hand-scrubbing. Alcohol combined with aldehydes is used in dentistry for surface disinfection, but authorities in the USA do not recommend alcohol for this purpose as it evaporates relatively quickly and leaves no residual effect. Other disadvantages are its flammability, limited sporicidal activity and ready inactivation by organic material. Yet, alcohols are still popular because they are cheap, readily available and water-soluble.
Aldehydes
Glutaraldehyde is perhaps the most popular disinfectant used in dentistry in some regions, whereas it is banned in others. It is both a skin irritant and a sensitization agent, which results in both long-term and short-term health effects. It is mainly used for so-called ‘cold sterilization’ or the high-level disinfection of equipment (such as fibre-optic instruments) that does not withstand autoclaving procedures. All aldehydes are high-potency disinfectants.
The free aldehyde groups of glutaraldehyde react strongly with the free amino groups of proteins in a pH-dependent manner. This leads to the effective microbicidal activity, sensitization of skin and incidentally, cross-linking with proteins such as collagen when used as a component of dentine-bonding systems. Hence, as the pH decreases, the activity of glutaraldehyde declines while its stability increases. Conversely, when the pH is alkaline, the activity is higher and it becomes less stable. Hence, in practice, glutaraldehyde is commercially available as a 2% acidic solution, to which an ‘activator’ has to be added to bring the solution to the ‘in-use’ alkaline pH of 8.0. Although the activated solution has a shelf-life of up to 14 days, this should be interpreted with caution as the solution may become prematurely ineffective due to other factors.
Bisguanides
Chlorhexidine is an example of a bisguanide disinfectant; it is widely used in dentistry as an antiseptic and a plaque-controlling agent. For example, a 0.4% solution in detergent is used as a surgical scrub (Hibiscrub); 0.2% chlorhexidine gluconate in aqueous solution is used as an antiplaque agent (Corsodyl); and at a higher concentration (2%), it is used as denture disinfectant. It is a cationic bisguanide molecule, usually prepared as salts of acetate, digluconate, hydrochloride and nitrate.
As chlorhexidine has two positive charges at its polar ends, it is highly active against both Gram-positive and Gram-negative organisms. (Note: all bacteria possess negatively charged cell walls in nature.) It also kills Candida (but not M. tuberculosis). Due to ingress of the disinfectant, the cell membrane permeability is altered with resultant leakage of cell contents and precipitation of the cytoplasm leading to cell death. Its substantivity (i.e. prolonged persistence) in the oral cavity is mainly due to absorption on to hydroxyapatite and salivary mucus.
Halogen compounds
Hypochlorites and povidone–iodine are oxidizing agents and act by releasing halide ions. Although cheap and effective, they readily corrode metal and are quickly inactivated by organic matter. (Examples of proprietary preparations are Chloros, Domestos and Betadine.) Note: available chlorine is a measure commonly used to indicate the oxidizing capacity of hypochlorite agents and is expressed as the equivalent amount of elemental chlorine. Thus, the equivalence of 1% available chlorine corresponds to 10 000 ppm available chlorine.
Phenolics
Phenolic disinfectants are clear, soluble or black/white fluids (black/white fluids are not used in dentistry). They do not irritate the skin and are used for gross decontamination because they are not easily degraded by organic material. They are poorly virucidal and sporicidal. As most bacteria are killed by these agents, they are used widely in hospitals and laboratories. Examples are Clearsol and Stericol.
Chloroxylenol is also a non-irritant phenolic used universally as an antiseptic; it has poor activity against many bacteria, and its use is limited to domestic disinfection (e.g. Dettol).
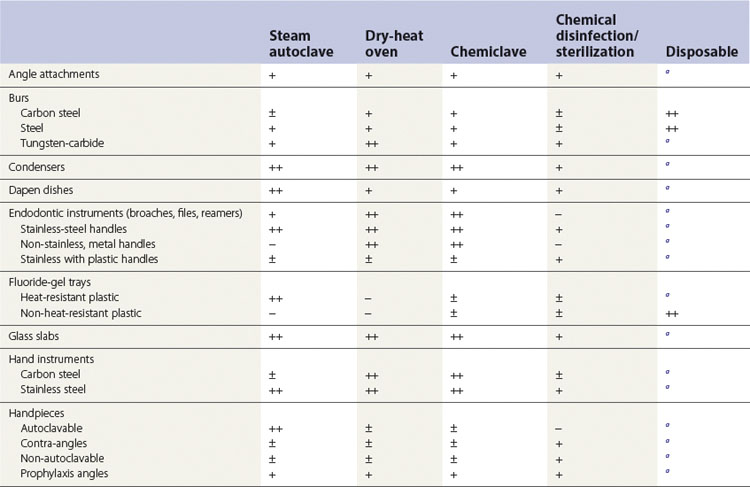
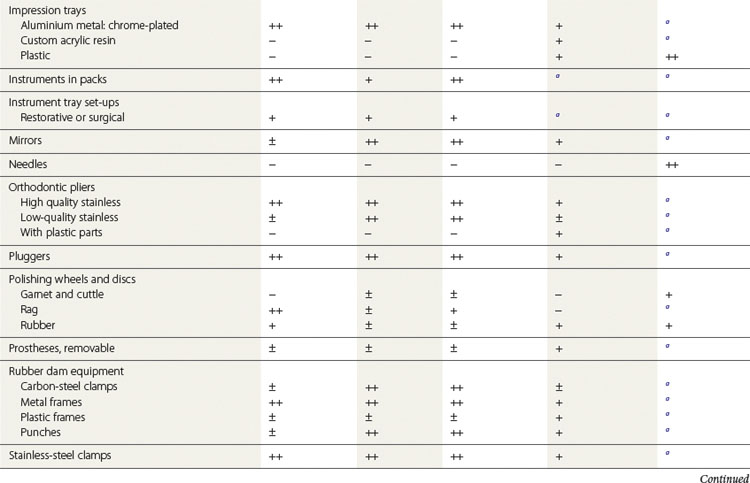
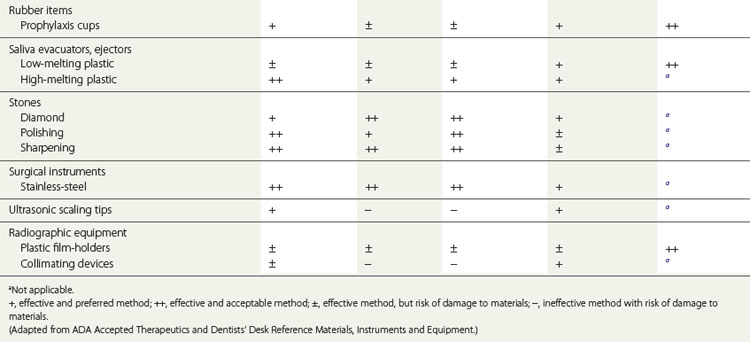
A sterilization and disinfection guide for items commonly used in dentistry is given in Table 37.7.
Environmental disinfection
The dental clinic setting should always be kept free of potential pathogens by appropriate environmental infection control measures. In general, when using environmental disinfectants:
Clinical contact surfaces
Clinical contact surfaces can be directly contaminated from patient materials either by direct spray or spatter generated during dental procedures or by contact with contaminated gloved hands of the dental personnel. These surfaces can subsequently contaminate other instruments, devices, hands or gloves. Examples of such surfaces include:
Barrier protection of surfaces and equipment can prevent contamination of clinical contact surfaces, but is particularly effective for those that are difficult to clean. Barriers include clear plastic wrap, bags, sheets, tubing and plastic-backed paper or other materials impervious to moisture. Because such coverings can become contaminated, they should be removed and discarded between patients, with gloved hands. After removing the barrier, the surface needs to be cleaned and disinfected only if contamination is evident. Otherwise, after removing gloves and performing hand hygiene, clean barriers on these surfaces should be replaced before the next patient.
If barriers are not used, surfaces should be cleaned and disinfected between patients by using either a low-level or an intermediate-level disinfectant when the surface is visibly contaminated with blood or saliva.
Housekeeping surfaces
Dental unit water lines: disinfection and management
The question of the quality of water in dental unit water lines (DUWLs) attached to handpieces, ultrasonic sealers and air/water syringes has been debated widely. The source of water to the dental unit is from either the municipal supply or wells, and after entering the unit, it passes through a multichannel control box that distributes the water to hoses feeding various attachments such as the high-speed handpiece, the air/water syringe and the ultrasonic scaler. The lines have a very small bore, and hence bacteria tend to form biofilms on the internal surfaces unless they are regularly cleaned and disinfected (Chapter 31). Although it has been questioned whether these innocuous saprophytic bacteria that live in water reservoirs are truly pathogenic, legislation has provided guidelines for the upper limits of bacteria and hence the quality of the water resources that service the DUWL. Generally, the water entering the DUWL contains very few organisms: 0–100 colony-forming units (CFUs)/ml. However, water exiting the handpiece may contain up to 100 000 CFU/ml, mainly because of the organisms that are picked up from the bacterial biofilms growing within the lines.
The guidelines of the American Dental Association are that the water delivered to patients from DUWL during non-surgical dental procedures should not contain more than 200 CFU/ml of aerobic, mesophilic, heterotrophic bacteria at any point. The association also stipulates that in the future, all dental units should contain a separate water reservoir independent of the public water supply, allowing dentists to have better control over the quality of the water used in patient care.
Recommendations on care of water lines
Maintaining quality of dental unit water
This could be achieved currently using antiretraction valves, filters, flushing, chemicals or water purifiers.
Antiretraction valves (check valves)
These are now the norm in all modern dental units and prevent the re-aspiration (or suck-back or back-siphonage) of fluid contaminated with oral flora of patients into the water line. However, it is now known that the antiretraction valves are very inefficient unless they are regularly maintained and replaced periodically.
Filters
Filters may be installed, for instance, between the water line and the dental instrument. These have no effect on the biofilm in the water lines but will remove microorganisms as the water is delivered to the patient. Filters are inefficient as they must be replaced periodically, and the frequency depends on the amount of biofilm in the water lines.
Flushing (see above)
This is a simple and efficient means of reducing the bacterial load in the water line. It is recognized that regular flushing prior to patient treatment will discharge the stagnant water and reduce malodour and bad taste imparted to the water by microbial contamination. Although flushing can reduce the numbers of bacteria in expelled water, the effect is transient and has no impact on the water line biofilm.
Biocides and chemicals
These remove, inactivate or prevent the formation of biofilm. Chemicals can either be continuously infused into or be intermittently added to the dental unit water by varying technologies. Chlorine, as sodium hypochlorite or chlorine dioxide, is the most commonly employed biocide. Concerns here are the possible development of bacteria resistant to the chemicals and environmental pollution.
Water purifiers
Water purifiers treat the water coming into the dental unit (source water). These treat the source water and kill or remove microorganisms by methods such as filtration, heat or ultraviolet light. One advantage of this method is that they may delay biofilm formation on water lines or synergize other treatment methods.
Boil-water advisories
Boil-water advisory is issued by authorities when the public water supply is likely to be contaminated with pathogenic organisms or the numbers of microbes in the system are above that which is compatible with health. During such periods, the following apply:
Recommendations on care of handpieces and other devices attached to air and water lines
Dental radiology
Laboratory asepsis
Dental practitioners regularly send clinical material to the laboratory: impression material, dentures sent to the technology laboratory or pathological samples such as pus or biopsy specimens referred to pathology laboratories, for example. The dentist is obliged to deliver all such items in a manner that obviates infectious hazards, whether during transport or within the laboratory. Blood and saliva must be carefully cleaned from the impressions and denture work by washing under running water and disinfection, and, if appropriate, placed in plastic bags before transport to the laboratory. Proprietary disinfectant sprays may be useful in decontaminating the microbes retained on impression surfaces.
The dental laboratory itself should be regarded as a clean (not contaminated) area, and appropriate protocols for disinfection of surfaces and material, as well as regular and timely renewal of disinfectant solutions, should be established. Smoking and eating should be prohibited.
Microbiological specimens sent to the laboratory should be securely bagged to avoid contamination of personnel who handle the items. The request form should be separately enclosed to prevent contamination. Biopsy specimens should be put in a sturdy container with a secure lid to prevent leakage during transport. Care should be taken when collecting specimens to avoid contamination of the external surface of the container.
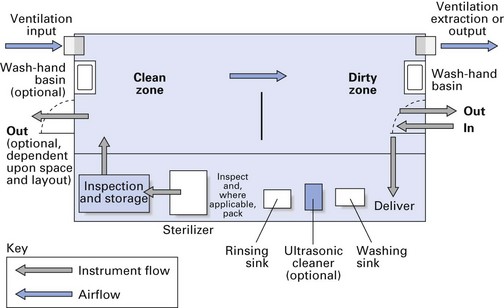
Office/surgery design and maintenance
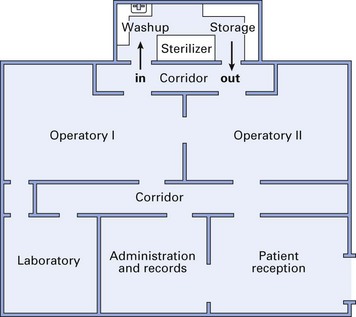
Proper office or surgery design is the cornerstone of an effective infection control programme (Fig. 37.9). Major features of such a design are:
Infection control requirements should always be borne in mind when selecting new equipment.
Instrument recirculation and office design
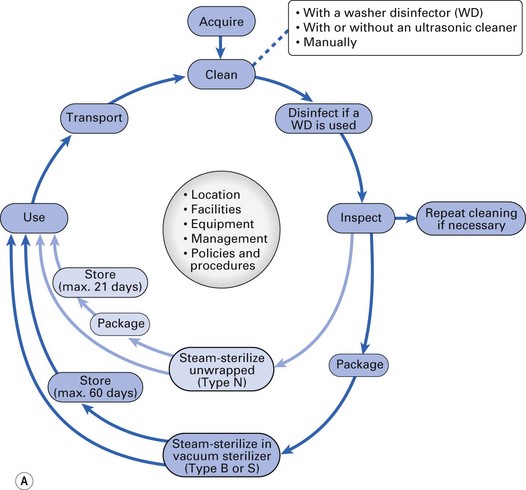
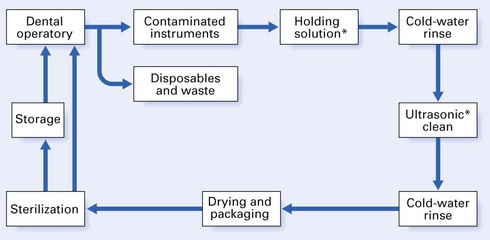
In order to conduct an efficient and routine sterility programme, it is important to organize the various arms of the infection control programme outlined above in the most effective manner. Therefore, it is essential to design the dental office and instrument recirculation areas (washing up, sterilizing and storage) to achieve this aim. The instrument recirculation area should be organized in order to:
A suitable instrument recirculation profile is shown in Figure 37.10. Other noteworthy points are:
Disposal of medical waste
Any waste material that has been in contact with human sources is contaminated with potentially pathogenic microbes or will possibly support their growth.
General recommendations
Develop a medical waste management programme. Disposal of regulated medical waste must follow local and federal regulations. Ensure that health care workers who handle and dispose of potentially infective wastes are trained in appropriate handling and disposal methods and informed of the possible health and safety hazards.
Medical waste in dental health care facilities
Use a colour-coded or labelled container that prevents leakage (e.g. biohazard bag) to contain non-sharp regulated medical waste.
All sharp items (especially needles), tissues or blood should be considered as particularly dangerous and should be handled and disposed of with special precautions. Disposable needles, scalpels or other sharp items must be placed intact into puncture-resistant containers before disposal.
If permitted by local regulations, discard blood, suctioned fluids or other liquid waste carefully into a drain connected to a sanitary sewer system. Wear appropriate protective attire while performing this task. Clinical waste should never be mixed with domestic waste, as this is a dangerous practice; it may also lead to litigation.
Key facts
Anonymous. Decontamination: Health Technical Memorandum 01-05 – Decontamination in primary care dental practices. UK: Department of Health; 2009.
Beltramy E.M., Williams I.T., Shapiro C.N., Chamberland M.E. Risk and management of blood-borne infections in health care workers. Clinical Microbiology Reviews. 2000;13:385-407.
Centers for Disease Control and Prevention. Guidelines for infection control in dental health-care settings. Morbidity and Mortality Weekly Report. 2003;52:1-66.
Franco F.F.S., Spratt D., Leao J.C., Porter S.R. Biofilm formation and control in dental unit water lines. Biofilms. 2005;2:9-17.
Russell A.D., Hugo W.B., Ayliffe G.A.J. Principles and practice of disinfection, preservation and sterilization, 2nd ed. Oxford: Blackwell; 1992.
Samaranayake L.P. Cross infection prevention in dentistry. Part II: Practical procedures. Dental Update. 1989;16:108-112.
Samaranayake L.P., Scheutz F., Cottone J. Infection control for the dental team. Copenhagen: Munksgaard; 1991.
Review questions (answers on p. 355)
Please indicate which answers are true, and which are false.