Chapter 31 Normal oral flora, the oral ecosystem and plaque biofilms
Normal oral flora
Oral flora comprises a diverse array of organisms and includes eubacteria, archaea, fungi, mycoplasmas, protozoa and possibly a viral flora that may persist from time to time. These organisms usually live in harmony in a range of habitats including the teeth, gingival sulcus, tongue, cheek, hard and soft palate and tonsils. Collectively the oral flora have been termed oral microbiota, and more recently, the oral microbiome. Bacteria are by far the predominant group of organisms, and there are probably some 500 to 700 common oral species or phylotypes of which only 50 to 60% are cultivable. The remaining unculturable flora are currently being identified using molecular techniques. This, together with the fact that the oral cavity has a wide range of sites (habitats) with different environmental conditions, makes the study of oral microbiology complex and difficult. Interestingly, despite the enormous diversity and complexity of the oral flora, many organisms commonly isolated from neighbouring ecosystems such as the gut and skin are not found in the mouth, indicating the unique and selective ecology of the oral cavity with regard to microbial colonization.
The main bacterial genera found in the oral cavity are well characterized using mostly traditional culture-based techniques. Oral bacteria can be classified primarily as Gram-positive and Gram-negative organisms, and secondarily as either anaerobic or facultatively anaerobic according to their oxygen requirements. Some oral microbes are more closely associated with disease than others, and a proportion of these appear to be uncultivable. The following is a synopsis of the major bacterial genera isolated from the oral cavity. Students should refer to the appropriate chapters in Part 3 for detailed information on these organisms.
A note on the nomenclature of oral flora
Due to continuing advances in molecular technology, especially those based on 16S ribosomal RNA (rRNA) sequences, microbial taxonomy is always in a state of flux. This poses a challenge to both the student and the scientist alike. Despite these changes, some prefer using the traditional nomenclature, while others use the new terminology, leading to further confusion. Hence in the following text, both the old and the recent taxonomic changes of oral bacteria are highlighted.
Flora of the oral cavity
Gram-positive cocci
Genus Streptococcus
Gram-positive cocci in chains, non-motile, usually possessing surface fibrils, occasionally capsulate; facultative anaerobes; variable haemolysis but α-haemolysis most common; selective medium: mitis salivarius agar (MSA).
mutans group
salivarius group
mitis group
Anaerobic streptococci
Gram-positive rods and filaments
These organisms are common isolates from dental plaque biofilms and include actinomycetes, lactobacilli, eubacteria and propionibacteria.
Genus Actinomyces
Short, Gram-positive pleomorphic rods:
Genus Lactobacillus
Genus Eubacterium
Pleomorphic, Gram-variable rods or filaments:
Genus Propionibacterium
Other notable Gram-positive organisms
Rothia dentocariosa, a Gram-positive branching filament, is a strict aerobe, found in plaque and occasionally isolated from infective endocarditis.
Bifidobacterium dentium is a Gram-positive strict anaerobe regularly isolated from plaque biofilms; its role in disease is unclear.
Gram-negative cocci
Genus Veillonella
Gram-negative rods – facultative anaerobic and capnophilic genera
Genus Haemophilus
Genus Aggregatibacter
Gram-negative coccobacilli, microaerophilic or capnophilic (carbon dioxide-dependent).
Genus Eikenella
Gram-negative rods – obligate anaerobic genera
These comprise a large proportion of the plaque biofilms. The classification of this group of organisms is fraught with difficulties, but the advent of new tests such as lipid analysis and molecular approaches have eased the problem to some extent. Most of the oral anaerobes were previously classified under the genus Bacteroides. However, advances in taxonomic methods have shown that they belong to two major genera, now termed Porphyromonas and Prevotella, which differ in their ability to metabolize sugar. Some of these organisms produce characteristic brown-black pigments on blood agar and are referred to collectively as ‘black-pigmented anaerobes’ (see Fig. 17.1).
Genus Porphyromonas
Gram-negative pleomorphic rods, non-motile; six serotypes based on capsular polysaccharides (K antigen); asaccharolytic:
Genus Prevotella
Gram-negative pleomorphic rods, non-motile; moderately asaccharolytic, producing acetic, succinic and other acids from glucose:
Genus Fusobacterium
Slender, cigar-shaped Gram-negative rods with rounded ends (see Fig. 18.1):
Genus Wolinella
Genus Selenomonas
Genus Treponema
Motile Gram-negative helical cells, in three main sizes (large, medium and small):
A note on unculturable bacteria
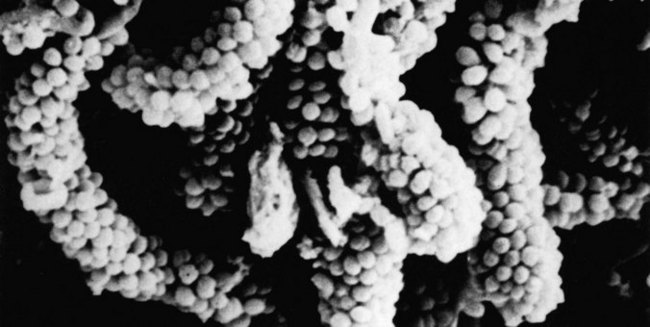
As stated above, it is now estimated that only about 50% of the oral bacteria that can be visualized by microscopy can be cultivated through traditional laboratory culture techniques. The identity and the role of these so-called unculturable bacteria is mostly an enigma. There are two major reasons that these bacteria cannot be cultured. First, their nutritional requirements are unknown, and second, they coexist in a supportive ecosystem in tandem with neighbouring organisms that sustain them nutritionally as well as physically (through an intricate architectural hierarchy) (Figs 31.1 and 31.2). Some examples of novel species and clones of bacteria detected from subgingival plaque using 16S rRNA and other techniques such as pyrosequencing are given in Table 31.1.
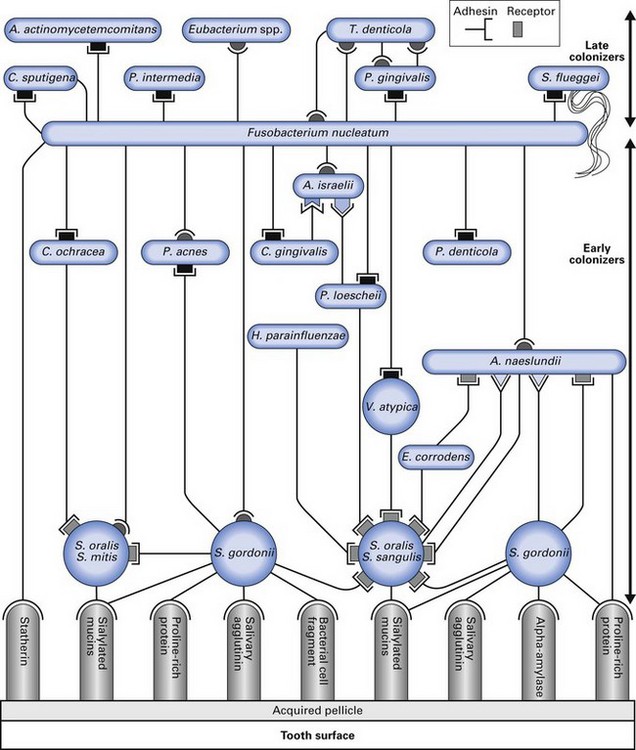
Fig. 31.2 A schematic picture illustrating the various interactions of oral microbial species that lead to plaque biofilm formation.
(Reproduced from Kolenbander, PE, Andersen, RN, Blehert, DS, Egland, PG, Foster, JS, Palmer, RJ, Jr (2002). Communication among oral bacteria. Microbiology and Molecular Biology Reviews 66:486–505, with permission.)
Table 31.1 Examples of novel species and clones of bacteria detected from subgingival plaque using 16S rRNA and other techniques such as pyrosequencing
| Named species | Novel phylotype |
|---|---|
| Atopobium parvulum | Selenomonas clone |
| Cantonella morbii | Megasphaera clone |
| Slackia exigua | Eubacterium clone |
| Filifactor alocis | TM7 (clone 1025) |
| Dialister pneumosintes | Deferribacteres clone |
Note: the significance of these isolates and their role in oral disease is still speculative.
Oral protozoa
Genus Trichomonas
Flagellated protozoa, about 7.5 µm in diameter:
For mycoplasmal and fungal infections of the oral cavity, see Chapters 20 and 22, respectively.
The oral ecosystem
Ecology is the study of the relationships between living organisms and their environment. An understanding of oral ecology is essential to comprehend the pathogenesis of diseases, such as caries and periodontal disease, caused by oral bacteria.
The oral environment
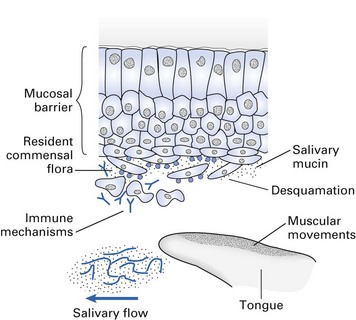
The human mouth is lined by stratified squamous epithelium. This is modified in areas according to function (e.g. the tongue) and interrupted by other structures such as teeth and salivary ducts. The gingival tissues form a cuff around each tooth, and there is a continuous exudate of crevicular fluid from the gingival crevice. A thin layer of saliva bathes the surface of the oral mucosa.
The mouth, being an extension of an external body site, has a natural microflora. This commensal (or indigenous, or resident) flora exists in harmony with the host, but disease conditions supervene when this relationship is broken. The predominant dental diseases in humans (caries and periodontal disease) are caused in this manner. In addition to the commensal flora, there are others (such as coliforms) that survive in the mouth only for short periods (transient flora). These transient flora cannot get a foothold in the oral environment due to the ecological pressure, i.e. the colonization resistance exerted by the resident flora. Indeed, the latter are considered critical in defending the key portal of entry into the digestive system, by offending pathogens.
The oral ecosystem comprises the oral flora, the different sites of the oral cavity where they grow (i.e. habitats) and the associated surroundings.
Oral habitats
Buccal mucosa and dorsum of the tongue
Special features and niches of the oral mucosa contribute to the diversity of the flora; for instance, the cheek mucosa is relatively sparsely colonized, whereas the papillary surface of the tongue is highly colonized because of the safe refuge provided by the papillae. The papillary surface of the tongue has a low redox potential (Eh), promoting the growth of anaerobic flora, and thus may serve as a reservoir for some of the Gram-negative anaerobes implicated in periodontal disease. Further, the keratinized and non-keratinized mucosae may offer refuge to variants of oral flora.
Teeth
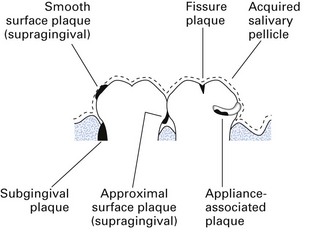
The surfaces of the teeth are the only non-shedding area of the body that harbours a microbial population. Large masses of bacteria and their products accumulate on tooth surfaces to produce dental plaque, present in both health and disease. Plaque is a classic example of a natural biofilm and is the major agent initiating caries and periodontal disease. In the latter situations, there is a shift in the composition of the plaque flora away from the species that predominate in health (see Chapters 32 and 33).
A range of habitats are associated with the tooth surface (Fig. 31.3). The nature of the bacterial community varies depending on the tooth concerned and the degree of exposure to the environment: smooth surfaces are colonized by a smaller number of species than pits and fissures; subgingival surfaces are more anaerobic than supragingival surfaces.
Factors modulating microbial growth
Different microenvironments in the mouth support their own microflora, which differ both qualitatively and quantitatively. The reasons for such variations are complex and include anatomical, salivary, crevicular fluid and microbial factors, among others.
Anatomical factors
Bacterial stagnation areas are created as a result of:
These areas are difficult to clean, either by the natural flushing action of saliva or by tooth-brushing.
Saliva
Whole (mixed) saliva bathing oral surfaces is derived from the major (parotid, submandibular and sublingual) and minor (labial, lingual, buccal and palatal) salivary glands. It is a complex mixture of inorganic ions, including sodium, potassium, calcium, chloride, bicarbonate and phosphate; the concentrations of these ions varies diurnally and in stimulated and resting saliva. The major organic constituents of saliva are proteins and glycoproteins (such as mucin), which modulate bacterial growth (Table 31.2) in the following ways:
Table 31.2 Specific and non-specific host defence factors of the mouth
| Defence factors | Main function |
|---|---|
| Non-specific | |
| Epithelial desquamation | Physical removal of microbes |
| Saliva flow | Physical removal of microbes |
| Mucin/agglutinins | Physical removal of microbes |
| Lysozyme | Cell lysis (bactericidal, fungicidal) |
| Lactoferrin | Iron sequestration (bactericidal, fungicidal) |
| Apolactoferrin | Iron sequestration (bactericidal, fungicidal) |
| Sialoperoxidase system | Hypothiocyanite production (neutral pH), hypocyanous acid production (low pH) |
| Histidine-rich peptides | Antibacterial and antifungal activity |
| Secretory leukocyte protease inhibitor (SLPI) | Blocks cell surface receptors needed for entry of HIV |
| Specific | |
| Intraepithelial lymphocytes and Langerhans cells | Cellular barrier to penetrating bacteria and/or antigens |
| Secretory IgA | Prevents microbial adhesion and metabolism |
| IgG, IgA, IgM | Prevent microbial adhesion, opsonins, complement activators |
| Complement | Activates neutrophils |
| Neutrophils/macrophages | Phagocytosis |
HIV, human immunodeficiency virus; Ig, immunoglobulin.
(See also Tables 8.1, 8.2 & 8.3.)
Gingival crevicular fluid
There is a continuous but slow flow of gingival crevicular fluid in health, and this increases during inflammation (e.g. gingivitis). The composition of crevicular fluid is similar to that of serum, and thus, the crevice is protected by these ‘surrogate’ specific and non-specific defence factors of serum. Crevicular fluid can influence the ecology of the crevice by:
Microbial factors
Microbes in the oral environment can interact with each other both in promoting and suppressing the neighbouring bacteria. Mechanisms that accomplish this include:
These mechanisms, which enable the commensal oral flora to suppress or inhibit the growth of exogenous, non-oral organisms and thereby exclude them from their habitat, are called colonization resistance.
Miscellaneous factors
Local environmental pH
Many microbes require a neutral pH for growth. The acidity of most oral surfaces is regulated by saliva (mean pH 6.7). Depending on the frequency of intake of dietary carbohydrates, the pH of plaque can fall to as low as 5.0 as a result of bacterial metabolism. Under these conditions, acidophilic bacteria can grow well (e.g. lactobacilli), while others are eliminated by competitive inhibition.
Oxidation–reduction potential
The oxidation–reduction potential of the environment (Eh) varies in different locations of the mouth. For instance, redox potential falls during plaque development from an initial Eh of over +200 mV (highly oxidized) to –141 mV (highly reduced) after 7 days. Such fluctuations favour the growth of different groups of bacteria.
Antimicrobial therapy
Systemic or topical antibiotics and antiseptics affect the oral flora; for instance, broad-spectrum antibiotics such as tetracycline can wipe out most of the endogenous flora and favour the emergence of yeast species.
Diet
Fermentable carbohydrates are the main class of compounds that alter the oral ecology. They act as a major source of nutrients, promoting the growth of acidogenic flora. The production of extracellular polysaccharides facilitates adherence of organisms to surfaces, while the intracellular polysaccharides serve as a food resource.
Acquisition of the normal oral flora
Dental plaque biofilm
The plaque biofilm is a tenacious microbial community, which develops on the soft and hard-tissue surfaces of the mouth, comprising living, dead and dying bacteria and their extracellular products, together with host compounds mainly derived from the saliva.
Composition
The organisms in plaque biofilm are surrounded by an organic matrix, which comprises about 30% of the total volume. The matrix is derived from the products of both the host and biofilm constituents. In the gingival area, proteins from the crevicular exudate become incorporated into the plaque biofilm. This matrix acts as a food reserve and as a cement, binding organisms both to each other and to various surfaces.
The microbial composition of dental plaque biofilm can vary widely between individuals; some people are rapid plaque-formers, others slow. Further, there are large variations in plaque composition within an individual, for example:
Distribution
Plaque biofilm is found on dental surfaces and appliances especially in the absence of oral hygiene. In general, it is found in anatomical areas protected from the host defences, e.g. occlusal fissures, interproximally or around the gingival crevice. Plaque samples are described in relation to their site of origin and are categorized as supragingival:
Microbial adherence and plaque biofilm formation
Adherence of a microbe to an oral surface is a prerequisite for colonization, and it is the initial step in the path leading to subsequent infection or invasion of tissues. The complex interaction of the factors that prevent microbial colonization on oral surfaces is shown in Figure 31.4.
Plaque biofilm formation
Plaque biofilm formation is a complex process comprising a number of different stages:
Thus, biofilm formation is a complex, competitive, sequential and dynamic colonization process, and in plaque biofilms, this complexity is further compounded due to the participation of different categories of oral bacteria. Specifically, the pioneer group of organisms that selectively colonize the salivary pellicle during plaque formation are Gram-positive cocci and rods. These are followed by Gram-negative cocci and rods, and finally by filaments, fusobacteria, spirils and spirochaetes. Such an example of a natural succession of plaque flora has been elegantly demonstrated in ‘experimental gingivitis’ studies, where groups of individuals, initially subjected to meticulous oral hygiene, were then followed up during a phase of no oral hygiene, and the freshly developing plaque flora was monitored closely. Results of such a study are shown in Figure 31.5.
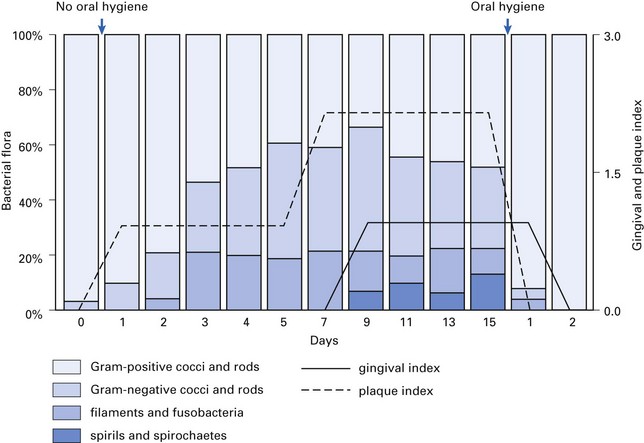
Fig. 31.5 Results from an experimental study showing the predominant groups of organisms comprising the pioneer and the climax community of plaque. Note the relationship between the plaque index and the gingival index.
One major component of a biofilm is the extracellular matrix. This comprises microbial polysaccharides and additional layers of salivary glycoprotein (or crevicular fluid components, depending on the site). The metabolic products of the early plaque colonizers can radically alter the immediate environment (e.g. create a low redox potential suitable for anaerobes), leading to new colonizers inhabiting the plaque, with a resultant gradual increase in microbial complexity, biomass and thickness. As a result of this dynamic process, the plaque biofilm mass reaches a critical size at which a balance between the deposition and loss of plaque bacteria is established; this community is termed the climax community (Fig. 31.6).
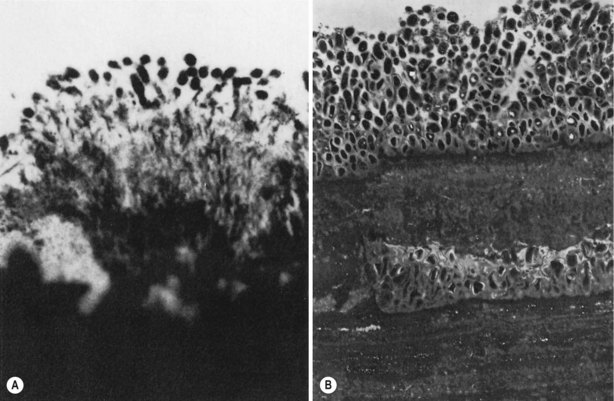
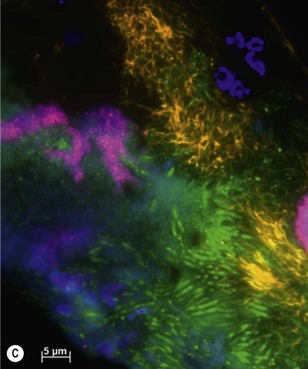
Fig. 31.6 Micrographs of (A) smooth-surface plaque showing the many relationships between different bacterial forms, including palisading and corn-cob formation and (B) mature plaque with compact bacteria and calcification at the base (approximately ×5000). (C) Mature subgingival plaque biofilms stained by Fluorescent in situ hybridization (FISH) technique showing non-specific bacteria (green), group 1 treponemes (orange) and Fusobacterium species (magenta) colonizing distinct parts of the biofilm. Some gingival host cell nuclei are stained blue with a nucleic acid stain.
(Image courtesy of Dr. Annette Motte.)
The molecular biology of biofilm formation is complex. Biofilm bacteria appear to maintain their complex structure through continuous secretion of low levels of molecules called quorum-sensing molecules (e.g. acyl homoserine lactone molecules, autoinducer-2) that coordinate gene expression. As the number of organisms in the biofilm increases, there is a simultaneous, proportionate increase in the quorum-sensing signals. These activate genes that may be related to additional extracellular polysaccharide production, or reduction of metabolism (for bacteria at the bottom of the matrix) or production of virulent factors, including drug-destroying genes.
Further notes on biofilms
The realization of the fact that up to 65% of human infections are caused by organisms encased in biofilms (i.e. sessile organisms) as opposed to planktonic or free-living forms has resulted in much research and a vast literature on the behaviour of these two rather divergent lifestyles of microbes. There is also a preponderance of biofilms in nature, for instance, as slimy coats that grow in stagnant water or water pipes (see Chapter 37 for biofilms in dental unit water lines). In clinical terms, it is recognized that biofilm organisms are more resistant to antibiotics and chemotherapeutic agents than their planktonic counterparts (see also Chapter 5). The problem of drug resistance, however, is not a major concern in dental plaque biofilms due to their ready accessibility to mechanical cleansing measures. However, drug resistance due to biofilms in other diseased states (e.g. airways infection by Pseudomonas aeruginosa in cystic fibrosis) is a major therapeutic problem.
Calculus formation
Calcium and phosphate ions derived from saliva may become deposited within deeper layers of dental plaque (as saliva is supersaturated with respect to these ions). If the plaque is allowed to grow undisturbed, then the degenerating bacteria in a climax community may act as seeding agents of mineralization. The process is accelerated by bacterial phosphatases and proteases that degrade some of the calcification inhibitors in saliva (statherin and proline-rich proteins). These processes lead to the formation of insoluble calcium phosphate crystals that coalesce to form a calcified mass of plaque, termed calculus.
Many toothpastes now contain pyrophosphate compounds that adsorb excess calcium ions, thus reducing intraplaque mineral deposition. In general, mature calculus is composed of 80% (dry weight) mineralized material, mostly hydroxyapatite and the remainder (20%) organic compounds.
Structure
The structure of calculus is shown in Figure 31.6. Predominant flora are cocci, bacilli and filaments (especially in the outer layers), and occasionally spiral organisms. The bacteria near the enamel surface tend to have a reduced cytoplasm to cell wall ratio, suggesting that they are metabolically inactive. Supragingival calculus contains more Gram-positive organisms, while subgingival calculus tends to contain more Gram-negative species.
In some areas (especially the outer surface), cocci attach and grow on the surface of filamentous microorganisms, giving a ‘corn-cob’ arrangement. The filamentous bacteria tend to orient themselves at right angles to the enamel surface, producing a palisade effect (like books on a shelf).
The cytoplasm of some bacteria (mainly cocci) contains glycogen-like food storage granules, available as a ready source of nutrition during periods of adversity.
Calculus has a rough surface and is porous, thus serving as an ideal reservoir for bacterial toxins that are harmful to the periodontium (e.g. lipopolysaccharides (LPSs)). Hence, removal of calculus is essential to maintain good periodontal health.
The role of dental plaque in caries and periodontal disease is discussed in Chapters 32 and 33, respectively.
The role of oral flora in systemic infection
Recently, it has been recognized that plaque-related oral diseases, especially periodontitis, may alter the course and pathogenesis of a number of systemic diseases. These include:
This is a resurgence of a common belief called ‘focal infection theory’ that was popular in the late 19th and early 20th century.
Three mechanisms linking oral infections to secondary systemic disease have been proposed:
Of these, the mechanisms linking oral infection and periodontal disease have been studied the most and the following are now known:
Apart from the well-established link between endocarditis and dental bacteraemias, there is no firm evidence to indicate that the other postulated diseases above are either initiated or perpetuated by oral flora and their by-products. The evidence available is circumstantial at best, with a multitude of confounding factors. Therefore, further research is necessary to confirm or refute these observations. Nonetheless, it is beyond doubt that good oral health is important not only to prevent oral disease but also to maintain good systemic health.
Key facts
Bowden G.H.W., Hamilton I.R. Survival of oral bacteria. Critical Reviews in Oral Biology and Medicine. 1998;9:54-58.
Dewhirst F.E., Chen T., Izard J., Paster B.J., Tanner A.C.R., Yu W., Lakshmanan A., Wade W.G. The Human Oral Microbiome. Journal of Bacteriology. 2010;192:5002-5017.
Edgar W.M., O’Mullane D.M. Saliva and oral health, 2nd ed. London: British Dental Association; 1996.
Lang N.P., Mombelli A., Attstrom R. Dental plaque and calculus, Ch. 3. Clinical periodontology and implant dentistry, 3rd ed. Copenhagen: Munksgaard. 1997.
Li X., Kolltveit K.M., Tronstad L., Olsen I. Systemic disease caused by oral infection. Clinical Microbiology Reviews. 2000;13:547-558.
Listgarten M.A. The structure of dental plaque. Periodontology. 1994;5:52-65. 2000
Marsh P.D., Martin M.V. Oral microbiology, 5th ed. London: Butterworth-Heinemann; 2009.
Parahitiyawa N.B., Jin L.J., Leung W.K., Yam W.C., Samaranayake L.P. Microbiology of odontogenic bacteraemia: Beyond endocarditis. Clinical Microbiology Reviews. 2009;22:46-64.
Samaranayake L.P., Ellepola A.N.B. Studying Candida albicans adhesion. In: Y. An, R. J. Freidman, editors. Handbook of bacterial adhesion: Principles, methods and applications. New York: Humana Press; 2000:527-540.
Review questions (answers on p. 354)
Please indicate which answers are true, and which are false.