CHAPTER 18 Geriatric dentistry
Introduction
Geriatric equine medicine is that part of medicine that relates to the prevention and treatment of diseases in aged horses. The age at which an equid requires geriatric care varies depending on the breed of the horse (or other equid), management practices and type of work. Horses have an increased prevalence of dental disorders, and in particular of periodontal disease, after 15 years of age.1–3 A recent study has shown that many donkeys start to develop serious dental related disorders at 16–20 years of age.4 Cheek teeth diastemata, wear abnormalities, overgrowths, displacements, loss of teeth, and other dental disorders, such as periodontal disease, wave mouth, step mouth, and smooth mouth have an increasing prevalence in donkeys older than 20 years of age.4 This emphasizes the need for additional preventative dental treatment to equids aged 15 years and older, although such additional care is needed at an earlier age if the animal has pre-existing dental disorders.
Clinical signs associated with dental disease vary with the severity of dental disease, and up to 24 % of horses that do not show any dental related clinical signs, have one or more dental abnormalities.5 In milder cases, clinical signs may be limited to bitting problems or abnormal head carriage during ridden exercise. With more advanced dental disease, equids may display quidding, with boluses of partially chewed forage found on the stable floor. With painful oral lesions, equids may exhibit slow deliberate chewing and make ‘slurping’ noises instead of the normal ‘crunching’ noises when masticating. Accumulation of forage boluses between the cheeks and cheek teeth may cause temporary cheek swellings. The presence of long forage fibers and un-masticated, undigested whole grain particles in the feces is also an indication of dental related problems. Eventually, in cases of severe dental disease, equids may have weight loss associated with decreased food intake and inefficient digestion.
Intercurrent geriatric diseases
When presented with a geriatric equid requiring dental treatment, it is important to acquire a detailed medical history and perform a complete physical examination. Older horses may have concurrent disease of other body systems, especially gastrointestinal, musculoskeletal, and respiratory tract problems.6 Laboratory analyses, such as complete blood counts and biochemistry functions, should be performed on initial examination to rule out other concurrent medical or metabolic conditions. The presence of large or non-healing cheek or tongue ulcers should alert practitioners to a possible underlying disease, such as pituitary dysfunction.7 Pituitary dysfunction should be considered in geriatric equids exhibiting any related clinical signs as 85 % of horses with pituitary dysfunction have been shown to be >15 years of age.8 A dental survey in live donkeys demonstrated that older donkeys are more likely to have dental disease, a low body condition score, and have a need for supplemental concentrate feeding.9 Furthermore, dental disease has been shown to be significantly associated with colic in donkeys.9,10
Anatomic overall tooth changes
Equid teeth are tapered from the occlusal to apical aspect, and as the teeth progressively wear down and erupt, the erupted (clinical) crown and occlusal surface become gradually smaller on cross-section. The occlusal surface of incisors changes in shape in older equids, initially appearing oval after eruption, then triangular and eventually becoming oval again with extreme age, as described in detail in Chapter 7. The rostral orientation of the caudal cheek teeth and caudal orientation of the rostral cheek teeth compress each cheek teeth row tightly to work as a single functional unit, even despite the initial narrowing of the reserve crown.11–14 However, eventually the tapering allows for the development of diastemata between the cheek teeth (’senile diastemata’) with food impaction and development of periodontal disease.13,15 Radiography of such a case is shown in Fig. 13.33.
The loss of maxillary cheek tooth infundibular enamel results in wearing out of the center of the tooth, leaving it with a thin, elevated peripheral enamel ridge (‘cupped out tooth’). The exposure of all three dental tissues (enamel, dentin and cementum) is essential for efficient mastication in equids, as the differential wear rate results in prominent enamel ridges and acts as a self-sharpening mechanism. As the teeth are worn to their more apical aspects, the peripheral enamel infolding in mandibular and maxillary cheek teeth becomes less pronounced, before finally wearing out at the junction with the cemental roots (Fig. 18.1). This is characterized by a smooth occlusal surface composed predominantly of cementum and dentin termed ‘smooth mouth’.13 As these teeth no longer have enamel ridges, they are ineffective at mastication, and as they have no wear resistance from enamel, they are quickly worn away (Fig. 18.2).
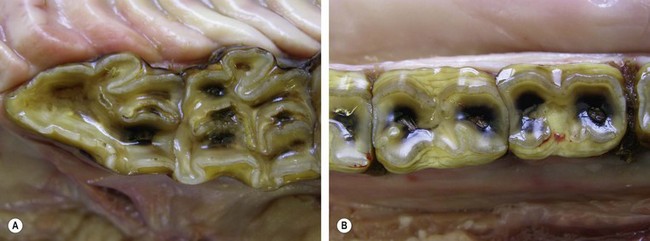
Fig. 18.1 Loss of the rostral infundibulum and ‘cupping’ of a 106 (A) and loss of peripheral enamel infolding in mandibular cheek teeth (B) in a 31-year-old donkey. Note the occlusal pulpal exposures in all the cheek teeth.
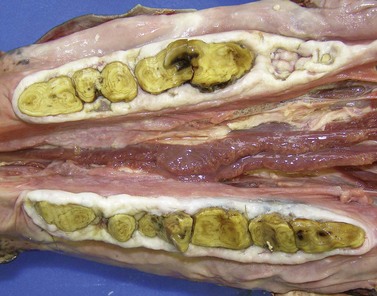
Fig. 18.2 End stage of ‘smooth mouth’ in a 28-year-old donkey with predominantly cemental root remnants of the cheek teeth remaining. Also note the loss of cheek teeth 410 and 411.
In older equids, the decreased occlusal surface area and reduced length of enamel ridges of cheek teeth result in loss of efficiency in grinding food. This is further complicated by the reduced ability to digest protein and fiber that is exhibited in geriatric horses.16 It is, therefore, essential that geriatric equids have their diet evaluated and adapted according to their dental and general health, and body condition. It has been shown that geriatric horses on a pelleted/extruded diet with 14 % protein content fared better than those fed an 8.5 % protein, textured, sweet feed mix.17 Therefore, the feeding of a good-quality, pre-digested (addition of enzyme) or extruded feedstuff with a protein content of 12–14 % would be beneficial to older equids that do not have hepatic or renal disease.18 If the maintenance of an adequate body condition is a problem, the addition of a vegetable oil or rice bran to the diet will increase the calorific content of the diet, without increasing the level of concentrate feeding.18 If the efficiency of mastication is greatly reduced as a result of ‘smooth mouth’ or other dental disorders, the feeding of moistened pellets or cubes to a liquid consistency will improve the amount of feed ingested. Commercially prepared dehydrated chopped hay with 15 % crude protein, which is available in the USA, is a suitable diet for many geriatric equids. If availability or economic consideration make this unsuitable, then fiber length may be reduced by processing hay through a wood chipper, leaf mulcher or a lawn mower using a bag attachment.
Sedation and restraint
The presence of concurrent diseases, such as cardiac disease, arthritis, and muscle wasting, needs to be taken into consideration when sedating and restraining geriatric equids for dental treatment. In general, lower doses of anesthetic and sedative agents are required in older horses, as they have increased sensitivity and decreased clearance of commonly used agents.19 The type and likely duration of dental treatment need to be determined prior to commencement of any treatments to assist in choosing the appropriate restraint methods. If a painful procedure is to be performed, the use of local nerve blocks will decrease the amount of systemic sedation and analgesia that is required.
Low dosages of alpha-2-adrenoceptor agonists, such as xylazine, romifidine hydrochloride or detomidine hydrochloride, in combination with low dosage of butorphanol tartrate for pain control, can be sufficient for most dental procedures in geriatric equids. Although butorphanol has five times the analgesic activity of morphine, the analgesia persists for only 15–20 minutes. Geriatric equids may be very fractious to extraction of worn, loose teeth, but butorphanol administered just prior to extraction usually provides sufficient analgesia. A continuous IV drip of detomidine or detomidine/butorphanol20 may be used instead of repeated dosing of these drugs. Use of a head support is recommended during dental procedures, but it should be used with stocks or after backing the horse into a corner to minimize slumping of the forelimbs and over-extension of the head, with possible atlanto-occipital subluxation.
Another option if significant dental treatment is required, especially in a fractious patient, is the use of short general anesthesia.21 The drawback to general anesthesia is the development of long bone fractures during recovery if insufficient assistance is available to help the horse rise. Protracted malnutrition may produce osteoporosis and so increase the risk of this sequel in older patients.
Heavily sedated geriatric horses, when left alone and unstimulated following dental procedures, are more susceptible to falling off their feet than younger horses. Judicious use of tolazoline HCl (100–200 mg in a slow IV injection) is indicated to partially reverse the effects of alpha-2-adrenoceptor agonists. If the sedation is insufficiently reversed after 15 to 20 minutes, the tolazoline HCl administration may be repeated using half to one-third of the initial dosage.
Incisor disorders
The incisor arcades in older equids often have disorders, such as excessive wear, corresponding overgrowths in the opposite teeth, malocclusions, missing teeth, and displaced teeth. Excessive wear of an entire incisor arcarde, most commonly the maxillary incisors, is usually as a result of a behavioral problem, such as crib-biting or wind-sucking. With loss of teeth or excessive incisor wear, repeated regular floating of the opposing teeth is required to maintain a level occlusal surface. Excessive reduction of severely overgrown incisors may expose pulp horns, and it is advisable to reduce overgrown incisors, in stages some months apart, to stimulate normal secondary dentin deposition to protect the occlusal aspect of the pulps. The tapering of incisors towards their apical aspect results in the development of incisor diastemata with the accumulation of food, in some older horses.
Geriatric equid incisors often have abnormal occlusal surfaces, such as a ventral convex curvature (’smile’), a dorsal convex curvature (‘frown’) or a diagonal (to left or right side) surface (’slant mouth or slope mouth’). These changes are often secondary to disorders of the cheek teeth and a resultant abnormal masticatory action. Once cheek teeth disorders have been corrected, these incisor table abnormalities can be corrected, in stages if severe. ‘Smile’ mouth appears to be very common in donkeys and was present in 96 % of aged donkeys with dental disease and 99 % of donkeys without dental disease4 (Fig. 18.3). Therefore, smile mouth may be regarded as a normal appearance in aged donkeys and should not be corrected unless inhibiting normal masticatory action.
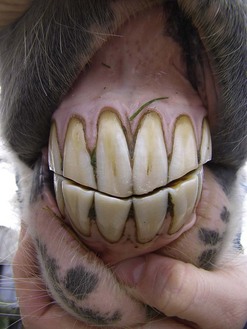
Fig. 18.3 The typical ‘smile’ mouth appearance of the incisor occlusal surface observed in most aged donkeys.
Functional incisor/molar occlusion in the presence of abnormal incisor occlusal surfaces may be determined by measuring the excursion to molar contact (EMC) distances. EMC is measured by putting the incisors arcades into centric occlusion and then pushing the mandible laterally until the upper and lower cheek teeth arcades touch and (due to their angulated occlusal surfaces) the incisors just begin to separate. The distance is measured from the center of the maxillary incisor arcade (interproximal space of 101 and 201) to the center of the mandibular incisors (interproximal space of 301 and 401). The EMC measurements are made on each side. When in centric occlusion, if the center points of the incisor arcades do not align with each other, then the offset distance is either added or subtracted from the center-to-center point measurement (Fig. 18.4).
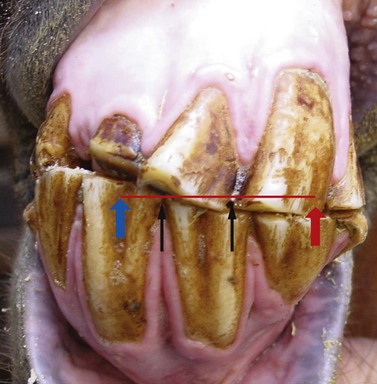
Fig. 18.4 In this 20-year-old Saddle bred horse, the mandibular center point is offset to the horse’s right by 10 mm (distance between the black arrows). EMC to the right is 8 mm (18 mm minus the 10 mm offset; blue to adjacent black arrow) and to the left is 24 mm (14 mm plus the 10 mm offset; red to adjacent black arrow). Measurement is made from the center of the maxillary incisors to the center of the mandibular incisors.
The average EMC distance for a 450 Kg horse is 12.3 mm.22 This distance, coupled with the average maximum excursion distance of 45 mm ±5 mm,23 demonstrates that a normal equid’s masticatory cycle is about 22–25 % incisor occlusion and about 75–78 % cheek teeth occlusion. Horses with heads shorter than average (Quarter horse length), have an average EMC distance less than 12.3 mm, while the EMC in longer headed horses is greater than 12.3. The mean EMC in miniature horses is about 4 mm, while the mean value in a draught horse is about 16 mm. This difference in EMC in different sized heads is because the pivot point or vertex for lateral excursion is the temporomandibular joint (TMJ). Lines drawn from the TMJ to each of the center points diverge more the further they are from the TMJ and diverge less the nearer they are to the TMJ.
Reducing EMC distances within a range of 12–16 mm should be done carefully, with reduction of tall teeth performed in 1-mm thick stages on tall incisors, before re-assessing EMC. A 1-mm reduction in incisor height can decrease the EMC by 4 mm or more. Experience indicates that total reduction should be limited to 3 mm in one session or no more than a 10 mm change in the EMC. Horses with incisor malocclusions having reasonably normal or shorter than normal EMC distances do not need incisor correction; they have functional incisor malocclusions (Fig. 18.5).
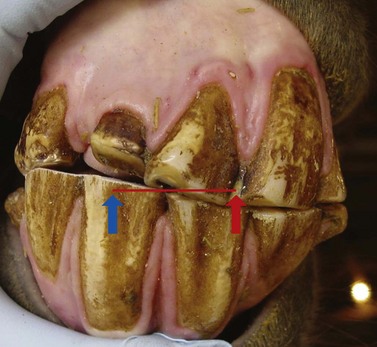
Fig. 18.5 Horse in Fig. 18.4 after correction. 103 (not visible) was reduced by 3 mm; this tooth was displaced caudally and was not interfering with cheek teeth occlusion but was making contact with 404. Teeth 402 and 403 were reduced 2.5 mm. EMC to left is now 14 mm and EMC to right has not changed. The incisors are not level but are now functional, and the horse can chew with more left cheek teeth occlusion.
Correction of a ‘smile’ should begin on the mandibular incisor arcade, removing 1-mm thick layers until the arcade is level, or the reduction (3 mm) or EMC (10 mm) limits are reached (Fig. 18.6). The lateral maxillary incisors are also reduced, if needed. ‘Frown’ correction reduction is begun on the maxillary arcade then the mandibular arcade, if needed. Correction should always commence on the 03s (corner incisors). If excursion or reduction limits are reached before reduction of the 01s (central incisors) is needed, incisor contact will be maintained on the 01s and possibly part or all of the 02s (intermediates), depending on the severity of the malocclusion.
Periodontal disease
The formation of senile diastemata in the incisors may lead to food impaction and periodontal disease. However, as these teeth are not exposed to any grinding masticatory forces, these diastemata are very rarely associated with deep periodontal pockets. These diastemata and associated (usually) mild periodontal disease can be managed by regular cleaning of the impacted food by the owner. In more severe cases of food impaction, which may be difficult to remove on the caudal aspect of the incisors, these diastemata can be widened using a diastema burr or rotary saw. The accumulation of calculus on the canines and 03s may also cause mild, localized periodontitis, which does not appear to cause any clinical signs (Fig. 18.7). Accumulation of large amounts of calculus can result in more severe gingivitis, with hyperemia and recession of the gingival margin. Removal of the calculus at every dental examination/treatment temporarily relieves the associated periodontal disease. Frequent brushing of the canines by the owners using a normal tooth brush may slow down re-accumulation of calculus.
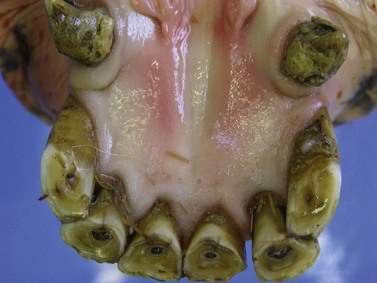
Fig. 18.7 Calculus accumulation on both mandibular canines and the 203s in a 28-year-old donkey. There is mild gingival hyperemia associated with the calculus on 304.
More recently, a more severe form of incisor periodontal disease has been recognized in geriatric equids that is associated with cemental hyoplasia, and hyperplasia and radiographic lytic changes.24–27 The exact etiopathogenesis of this disease has not been determined, but pathological studies show it to be an odontoclastic resorption of affected incisors (also of canine teeth) with subsequent marked deposition of cement on these teeth, termed equine odontoclastic tooth resorption and hypercementosis (EOTRH; discussed in Ch. 10). Affected animals may present with clinical signs associated with pain, such as masticatory and bitting problems, and halitosis (Fig. 18.8). Initially, this condition presents as mild gingival inflammation and edema with small lytic changes in the mid reserve crown incisor on radiographic examination.25 With progression of the condition, draining tracts may develop in the gingiva, and this may be accompanied by gingival recession, or marked subgingival swelling of incisors, reflecting hypercementosis of their reserve crown and apex. Radiographically, progression of the disease is characterized by lysis of the incisors in an apical direction and an increase in the thickness of the lytic areas to involve the dentin. Loss of interdental bone and widening of the periodontal ligament may also be observed on radiographs. Clinically advanced cases may have painful mobile incisors. Hypercementosis (subgingival nodular enlargements) of portions or all of the subgingival incisors may also be observed on radiographs in some advanced cases (Fig. 18.9).
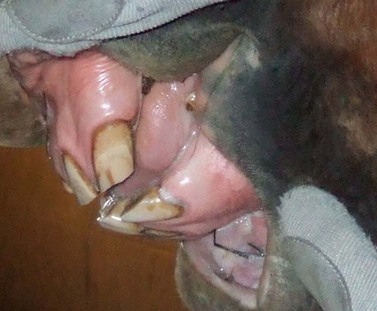
Fig. 18.8 Enlargement of thickness of reserve crown of 203 and 303 (identical to 103 and 403) due to hypercementosis associated with equine odontoclastic tooth resorption and hypercementosis. The horse was asymptomatic, and the disease was identified on oral examination. All the incisors were stable and non-painful to manipulation, but firm palpation of 303 and 403 reserve crowns was painful.
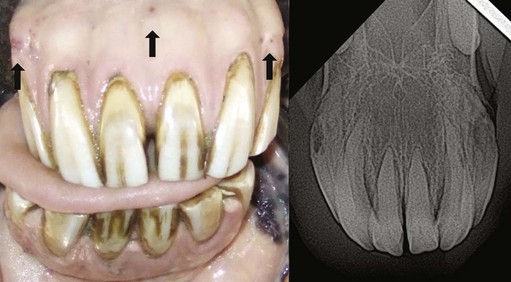
Fig. 18.9 This 20-year-old Thoroughbred was reported to refuse to bite carrots for over one year. Enlargement of all incisor reserve crowns is prominent with gingival recession, and draining tracts visible (at arrows). Radiographs showed extensive lysis and hypercementosis of 103 and 203, typical of equine odontoclastic tooth resorption and hypercementosis, with radiographic changes in all the other incisors.
Due to the predominance of resorptive lesions in this condition, it has been proposed to be more similar to feline tooth resorption than to a primary periodontal disease.24 Feline tooth resorption has been shown to start as focal lesions at the cemento-enamel junction, indicating that local factors appear to play a role in the etiopathogenesis.28 Feline tooth resorption has an initial resorptive phase where there is odontoclastic resorption of dentin and destruction of the periodontal ligament and alveolar bone.29 This is followed by cementum and bone deposition during the reparative phase. Thus far, bone deposition and dento-alveolar ankylosis, as described in feline tooth resorption, has not been described in equids. Treatments with long-term antibiotics and steroids have been unsuccessful and extraction of affected incisors has been the only treatment to alleviate the clinical signs associated with this disease.
Canine teeth disorders
The most common abnormality observed in canine teeth is, as noted, the accumulation of calculus, which may cause mild local periodontal disease. This calculus is easily removed with the use of forceps. Excessive calculus accumulation on the mandibular canines has also been associated with corresponding tongue ulcers. Rarely, canine teeth may be displaced or enlarged and interfere with the bit. Canine teeth have very long, well embedded reserve crowns, and extraction of these teeth should not be performed without prior radiography. The canine teeth are often floated, purportedly to prevent bitting problems or injuries to operators’ hands and arms during dental procedures. Extreme floating or even cutting of these canine teeth may expose the pulp cavity and result in apical infections. Enlargement of the clinical crown, exposure of the alveolar bone or reserve crown, and the presence of pain if the canine is palpated, percussed or subjected to a cold substance (piece of ice), are all indications for radiographic evaluation of the canine.
Cheek teeth disorders
Dental abnormalities of wear encountered in geriatric equids are the same abnormalities that are recognized in younger equids, but they have reached an advanced stage over a prolonged period of time. These disorders, e.g., shear mouth, wave mouth, and focally overgrown teeth, are often complicated by the lack of reserve crown and hence, instability of teeth within the alveolus, which results in displacement and eventually loss of teeth (Fig. 18.10).
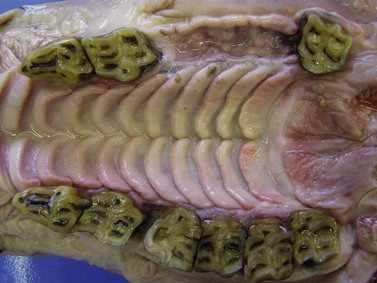
Fig. 18.10 Loss of maxillary cheek teeth 208, 209, and 210 is due to loss of reserve crown in this geriatric donkey. Also note the large, open diastema between 107 and 108 and the loss of infundibular enamel in 108, indicating the presence of a high degree of wear in these teeth and the beginning of ‘smooth mouth’.
A post-mortem survey of dental disease in geriatric donkeys (estimated median age 31 years) showed a very high prevalence (93 %) of dental disease; with diastemata (85 %), missing (56 %), displaced (43 %) and worn teeth (34 %) being the most common dental disorders present30 (Fig. 18.11). Studies in horses have also shown an increased prevalence of dental disease in older age groups.1,2,31–33 A comparison of clinical dental disorders in different age groups of donkeys (age range 2–53) showed a significant increase in the prevalence of dental disease in donkeys over 20 years of age (≥88 % prevalence) compared to donkeys younger than 20 years (≤64 %).4 More specifically, a significant increase in the prevalence of diastemata, missing teeth, overgrown teeth, worn teeth, displaced teeth, and periodontal disease with increasing age has been shown, in particular in donkeys over 20 years of age.4 As expected, the prevalence of enamel points decreased significantly with age, to a prevalence of less than 20 % in donkeys older than 20 years, as compared to >40 % prevalence in donkeys under 20 years of age.4
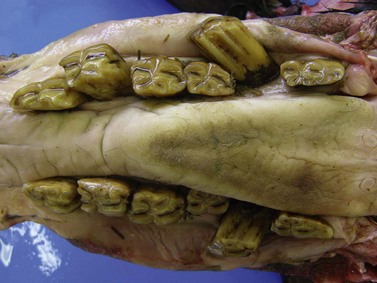
Fig. 18.11 Severe bilateral lateral displacement of the 310 and 410 is present in this 38-year-old donkey. There is also moderate lateral displacement of the 407.
Treatments of cheek teeth disorders in geriatric equids are often limited by the lack of reserve crown. Dental treatment should be aimed at preserving as much of the functional occlusal surface for mastication as possible, while ensuring oral comfort. Sharp enamel overgrowths should be floated to prevent soft tissue trauma to the cheeks (maxillary cheek teeth) or tongue (mandibular cheek teeth). Cheek teeth diastemata are often a result of tapering reserve crowns and loss of angulation of the rostral and caudal cheek teeth (’senile diastemata’), and, therefore, the cause of these diastemata cannot be eliminated (Fig. 18.12). As diastemata often involve the caudal cheek teeth,30,34 it is essential that a dental mirror and good light source are used to evaluate these. Impacted food should be flushed out, and the severity of associated periodontal disease determined. Very often, the only treatment for diastemata is to reduce the opposing cheek teeth occlusal surface by 2–3 mm to decrease the impaction of food into the diastema, particularly if there are focal transverse overgrowths secondary to the diastemata.34 Some clinicians advise filling the base of cleaned diastemata with dental impression material (as later described), but no objective studies are available on its efficacy in treating any type of periodontal disease in the horse. Following removal of cheek teeth overgrowths, mild-to-moderate periodontal disease may be arrested or its progression slowed by oral rinsing with diluted chlorhexidine gluconate (5 ml of 2 % chlorhexidine added to one liter of water). The owner should rinse the mouth out once a day for 10 days, then twice weekly. Some horses object to the taste of chlorhexidine and may become briefly anorectic. If this occurs, a flavored 0.12 % chlorhexidine (or human mouthwash) can be used as a rinse, 50 to 100 ml per rinse. The mouth should be rinsed with lukewarm water to remove food particles prior to the chlorhexidine rinsing.
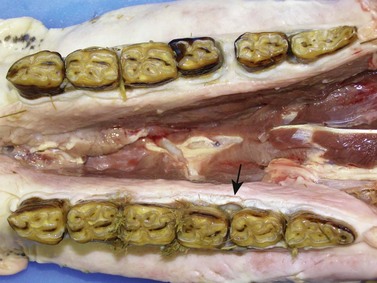
Fig. 18.12 Multiple senile diastemata of both mandibular cheek teeth rows in a donkey of 27 years. Some of these diastemata have adjacent severe periodontitis and deep periodontal pockets (arrow).
Periodontal disease is an important disease in geriatric equids and has been shown to occur in 80 % of horses over 20 years of age2 (Fig. 18.13). If the periodontal disease is severe and associated with periodontal pockets, the application of an antibiotic oral gel (Doxirobe [doxycycline] Gel, Pfizer Animal Health) with impression material may allow temporary alleviation of the inflammation and allow healing of the periodontal pocket.35,36 In contrast to small animals where the accumulation of calculus is an important cause of periodontal disease,37 cheek teeth calculus is less common in equids and is not a major cause of periodontal disease. Cheek teeth calculus accumulation has been observed in 19 % of geriatric donkeys at post mortem, predominantly on the maxillary 07, 08, and 09 cheek teeth30 (Fig. 18.14). Cheek teeth calculus often accumulates secondary to food stagnation in equids that have painful dental disease and subsequent decreased normal masticatory movements. These teeth are beside the salivary duct opening that possibly provides much of the mineral component for calculus.
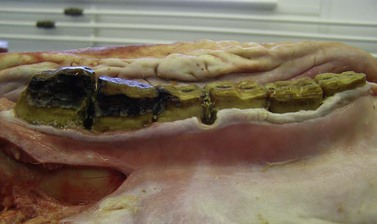
Fig. 18.13 Slight wave mouth with overgrowths of 410 and 411 and moderate peripheral caries. Diastemata are present in all the interdental spaces in this cheek teeth row, but the diastema between 408/409 has severe periodontal disease and food pocketing associated with it.
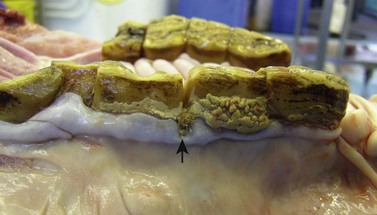
Fig. 18.14 Calculus accumulation on the buccal aspect of 207 and 208 in a geriatric (33-year-old) donkey. There is also a diastema with food impaction and periodontal pocketing between 208 and 209, and displacements of 209 and 210 (arrow).
Excessive cheek teeth overgrowths resulting in ‘step mouth’ should be reduced as much as possible (Fig. 18.15). If overgrowths are more than a few mm high, the initial reduction should be done in stages (e.g., 3 mm at a time) at 2–3-month intervals to prevent potential pulp horn exposure. As these overgrowths are secondary to missing, displaced, or ‘cupped out’ teeth in the opposing cheek teeth row, treatment needs to be repeated at regular intervals, to prevent re-development of these overgrowths to an extent that they are interfering with masticatory movements or causing soft tissue damage. These overgrowths do not have to be fully reduced (Fig. 18.16). Severely displaced teeth should be extracted if they are protruding into the cheeks or tongue and causing chronic ulceration and pain. Mild to moderately displaced teeth with focal overgrowths on the displaced and opposing teeth due to the malocclusion can be managed by reducing the overgrowths if the teeth are not digitally loose. Overgrown, slightly loose teeth may re-attach firmly if overgrowths are removed. However, if teeth are very loose or have marked periodontal disease and diastemata, extraction is the most appropriate treatment. Oral extraction of displaced teeth, particularly if still well embedded in the alveolus, may be complicated by the limited space between the cheek and tooth and the inability to satisfactorily apply molar extractors. Therefore, careful consideration must be given to the potential duration of the procedure and the required sedation to a debilitated geriatric equid.
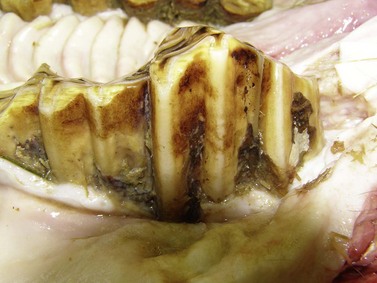
Fig. 18.15 Severely overgrown maxillary cheek tooth resulting in’step mouth’. This overgrown tooth needs to be reduced in stages to prevent potential pulpal exposure. Also note the associated periodontal disease and gingival recession.
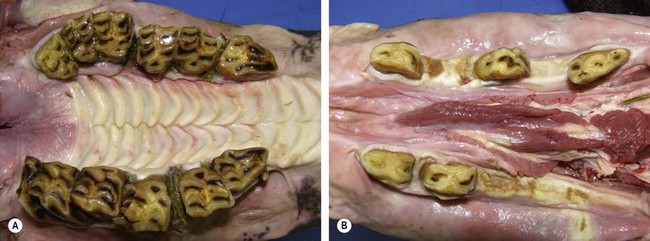
Fig. 18.16 Corresponding maxillary (A) and mandibular (B) cheek teeth rows that illustrate severely overgrown maxillary cheek teeth secondary to worn and missing mandibular cheek teeth, with mandibular soft tissue trauma. There are also multiple maxillary cheek teeth diastemata with food impaction. Note the presence of the rudimentary 205 (first premolar, wolf tooth).
Wave mouth is believed to develop as a result of the oldest teeth in the mouth (09s) wearing and becoming ‘cupped out’ prior to younger adjacent teeth, thus causing uneven wear in a cheek teeth row (Fig. 18.17).38 Treatment of wave mouth consists of floating excessively overgrown teeth to theoretically create a straight (or slightly concave) occlusal surface in the cheek teeth rows in a rostrocaudal direction, in mild cases. However, in the usually more severe cases of wave mouth, only the area of maximal overgrowth can be reduced without removal of very significant amounts of the reserve crown. The aim is to create maximal occlusal contact surface area and allow for the normal range of mandibular jaw movement for efficient mastication. The cheek teeth row must not be floated to the height of worn teeth, but rather the overgrowth on the opposing tooth should be floated. When reducing overgrown cheek teeth, it is important to remember the normal occlusal angle of mandibular (18–31°) and maxillary (12–9°) cheek teeth in a lingual-buccal and buccal-palatal direction, respectively.39

Fig. 18.17 ‘Wave mouth’ in a right mandibular cheek teeth row with a medially displaced 408 and 406, an overgrown 410, and worn 411. A large diastema between 407 and 408 with periodontal disease and gingival recession is also present, as is moderate peripheral caries of the 406 and 407, most likely secondary to food stagnation due to abnormal masticatory action.
Other abnormalities of wear may result in further malocclusions, with geriatric equids usually presented with multiple dental disorders. Prolonged overgrowths of the 106, 206, 311 and 411 may result in restricted rostrocaudal movement and contribute to more pronounced generalized cheek teeth overgrowths.
Ultimately, most geriatric equids with dental disease need to have regular (6-monthly or annually) dental examinations and treatment as appropriate on welfare grounds to ensure oral comfort and masticatory efficiency. It is highly likely that those with significant dental wear or disease will require supplemental feeding to maintain a suitable body weight, and this is particularly so in the colder months when green forage is not available. Owners need to be educated on the long-term management of geriatric equids, with particular emphasis on the formulation of an appropriate diet.
1 Baker GJ. Some aspects of equine dental disease. Equine Vet J. 1970;2:105–110.
2 Wafa NS, A study of dental disease in the horse. MVM Thesis, University College Dublin, Dublin, 1988.
3 Kirkland KD, Marretta SM, Inoue OJ, Baker GJ. Survey of equine dental disease and associated oral pathology. Proceedings of the 40th American Association of Equine Practitioners Annual Convention. 1994;40:119–120.
4 du Toit N, Burden FA, Dixon PM. Clinical dental examinations of 357 donkeys in the UK: Part 1 – Prevalence of dental disorders. Equine Vet J. 41(4), 2009. 390–394(5)
5 Uhlinger C. Survey of selected dental abnormalities in 233 horses. Proceedings of the 33rd American Association of Equine Practitioners Annual Convention. 1987;33:577–583.
6 Brosnahan MM, Paradis MR. Demographic and clinical characteristics of geriatric horses: 467 cases (1989–1999). Journal of American Veterinary Medical Association. 2003;223:93–98.
7 Hillyer MH, Taylor FRG, Mair TS, et al. Diagnosis of hyperadrenocorticism in the horse. Equine Veterinary Education. 1992;18:131–134.
8 Dybdal N. Pituitary pars intermedia dysfunction (equine Cushing’s-like disease). In: Robinson NE, ed. Current therapy in equine medicine. Philadelphia: WB Saunders; 1997:499–501.
9 du Toit N, Burden FA, Dixon PM. Clinical dental examinations of 357 donkeys in the UK: Part 2 – Epidemiological studies on possible relationship between different dental disorders, and between dental disease and systemic disorders, 2009; 41: 395–400
10 du Toit N, Gallagher J, Burden FA, Dixon PM. Post mortem survey of dental disorders in 349 donkeys from an aged population (2005–2006): Part 2 – Epidemiological studies. Equine Vet J. 2008;40:209–213.
11 Dixon PM, Copeland AN. The radiological appearance of mandibular cheek teeth in ponies of different ages. Equine Veterinary Education. 1993;5:317–323.
12 Misk NA, Seilem SM. Radiographic studies on the development of cheek teeth in donkeys. Equine Practice. 1999;19:27–37.
13 Dixon PM, The gross, histological, and ultrastructural anatomy of equine teeth and their relationship to disease. Proceedings of the 48th American Association of Equine Practitioners Annual Convention, 2002:421–437.
14 Dixon PM, Dacre I. A review of equine dental disorders. Equine Vet Educ. 2005;169:165–187.
15 Dixon PM, Tremaine WH, Pickles K, et al. Equine dental disease part 2: a long-term study of 400 cases: disorders of development and eruption and variations in position of the cheek teeth. Equine Vet J. 1999;31:519–528.
16 Ralston SL, Squires EL, Nockels CF. Digestion in the aged horse. Equine Veterinary Science. 1989;9:203–295.
17 Ralston SL, Breuer LH. Field evaluation of a feed formulated for geriatric horses. Equine Veterinary Science. 1996;16:334–338.
18 Pugh DG. Feeding the geriatric horse. In: Proceedings of the 53th American Association of Equine Practitioners Annual Convention, Orlando, Florida, 2007, pp 193–195
19 Donaldson LL. Anesthetic considerations for the geriatric equine. In: Bertone JJ, ed. Equine geriatric medicine and surgery. St Louis: Elsevier; 2006:25–37.
20 Goodrich LR, Clark-Price S, Ludders J, How to attain effective and consistent sedation for standing procedures in the horse using constant rate infusion. Proceedings of the 50th American Association of Equine Practitioners Annual Convention, 2004:229–232.
21 Carmalt JL. Safety, restraint, and oral examination of the horse. In: AAEP Dental focus meeting, Indianapolis, Indiana, 2006
22 Rucker BA. Utilising cheek teeth angle of occlusion to determine length of incisor shortening. Proceedings of the 48th American Association of Equine Practitioners Annual Convention, 2002, pp 448–452
23 Collison M. Food processing and digestibility in horses. B Sc dissertation. Victoria: Monash University; 1994.
24 Gregory RC, Fehr J, Bryant J. Chronic incisor periodontal disease with cemental hyperplasia and hypoplasia in horses. In: AAEP Dental focus meeting, Indianapolis, Indiana, 2006
25 Baratt RM. Equine resorptive lesions. In: Conference proceedings of the 21st Annual Veterinary Dental Forum, Minneapolis, 2007, pp 123–130
26 Caldwell LA. Clinical features of chronic disease of the anterior dentition in horses. In: Conference proceedings of the 21st Annual Dental Forum, Minneapolis, 2007
27 Stazyk C, Bienert A, Kreutzer R, et al. Equine odondoclastic tooth resorption and hypercementosis. The Veterinary Journal. 2008;178:372–379.
28 DeLaurier A, Boyde A, Horton MA, Price S. A scanning electron microscopy study of idiopathic external tooth resorption in the cat. Journal of Periodontology. 2005;76:1106–1112.
29 Gorrel C. Feline odontoclastic resorptive lesions. In: World Small Animal Veterinary Association 23rd World Congress Proceedings, Bangkok, Thailand, 2003
30 du Toit N, Gallagher J, Burden FA, Dixon PM. Post mortem survey of dental disorders in 349 donkeys from an aged population (2005–2006). Part 1: prevalence of specific dental disorders. Equine Vet J. 2008;40:204–208.
31 Honma K, Yamakawa M, Yamauchi S, Hosoya S. Statistical study on the occurrence of dental caries of domestic animal: I. Horse. Japanese Journal of Veterinary Research. 1962;10:31–36.
32 Miles AEW, Grigson C. Coyler’s variations and diseases of the teeth of animals. Cambridge: Cambridge University Press; 1990. p. 672
33 Uhlinger C. Common abnormalities of the premolars and molars. Proceedings of the 37th American Association of Equine Practitioners Annual Convention. 1991;37:123–128.
34 Dixon PM, Barakzai S, Collins N, Yates J. Treatment of equine cheek teeth by mechanical widening of diastemata in 60 horses (2000–2006). Equine Vet J. 2008;40:22–28.
35 Green SK, Basile T. Recognition and treatment of equine periodontal disease. Proceedings of the 48th American Association of Equine Practitioners Annual Convention. 2002;48:463–466.
36 Klugh DO. Equine periodontal disease. Clinical Techniques in Equine Practice. 2005;4:135–147.
37 Wiggs RB, Lobprise HB. Periodontology. In: Wiggs RB, Lobprise HB. Veterinary dentistry: Principles & practice. Philadelphia: Lippincott-Raven Publishers; 1997:186–231.
38 Leue G, Zähne. Dobberstein J, Pallaske G, Stunzi H, . Handbuch der speziellen pathologischen anatomie der haustiere, 3rd edn, Berlin: Verlag Paul Parey, 1941.
39 Brown S, Arkins S, Shaw DJ, Dixon PM. The occlusal angles of cheek teeth in normal horses and horses with dental disease. Veterinary Record. 2008;162:807–810.