61 Fungi
Superficial, subcutaneous and systemic mycoses
Key points
• Most infections are caused by fungi that grow as saprobes in the environment. Superficial, subcutaneous and systemic patterns of infection are recognized.
• Fungi take the form of yeasts, which grow by budding and moulds, which grow as filamentous extensions called hyphae, forming a mycelium, or dimorphic fungi, which can grow as yeast or mould forms.
• Pathogenic fungi can establish an infection in all exposed individuals; others are opportunist pathogens that cause disease only in a compromised host.
• Many fungal diseases have a global distribution, but some are endemic to specific geographical regions.
• Subcutaneous fungal infections are acquired by traumatic implantation; systemic infections are usually acquired by inhalation.
• Only dermatophytosis, a common superficial infection of the skin, nails and scalp hair, is truly contagious.
• Some yeasts are human commensals and cause endogenous infection when there is some imbalance in the host.
• The most frequently encountered fungal agents in the UK are Candida albicans, dermatophytes, Aspergillus spp., Cryptococcus neoformans and Pneumocystis jirovecii.
• Diagnosis of fungal infections is based on a combination of clinical observation and laboratory investigation, which may include direct microscopy, histology, culture, PCR and serology. Early recognition of systemic infections in immunocompromised persons is a major challenge.
Fungi constitute a large, diverse group of heterotrophic organisms, most of which are found as saprobes in the soil and on decomposing organic matter. They are eukaryotic, with a range of internal membrane systems, membrane-bound organelles, and a well-defined cell wall composed largely of polysaccharides (glucan, mannan) and chitin. They show considerable variation in size and form, but can be divided into three main groups:
1. Moulds (multicellular filamentous fungi), which are composed of branching filaments, termed hyphae, that grow by apical extension to form an intertwined mass, termed a mycelium. In most fungi the hyphae have regular cross-walls (septa) but in lower fungi these are usually absent. Moulds reproduce by means of spores produced, often in large numbers, by an asexual process (involving mitosis only) or as a result of sexual reproduction (involving meiosis, preceded by fusion of the nuclei of two haploid cells). Many fungi can produce more than one type of spore, depending on the growth conditions. The precise method of spore production and the type(s) of spore produced are unique to each individual fungal species. In some higher fungi the sexual spores are produced in macroscopic structures such as mushrooms and toadstools. In laboratory cultures, moulds produce mainly asexual spores.
2. Yeasts, which are predominantly unicellular and oval or round in shape. Most propagate by an asexual process called budding in which the cell develops a protuberance, which enlarges and eventually separates from the parent cell. Some yeasts produce chains of elongated cells (pseudohyphae) that resemble the hyphae of moulds; some species also produce true hyphae. A small number of yeasts reproduce by fission. Yeasts are neither a natural nor a formal taxonomic group, but are a growth form shown by a wide range of unrelated fungi.
3. Dimorphic fungi, which are capable of changing their growth to either a mycelial or yeast phase, depending on the growth conditions.
The fungal kingdom is currently divided into seven phyla which include the Ascomycota, Basidiomycota and Glomeromycota. Historically, fungal classification has largely been based on the method of sexual spore production. In some fungi, however, asexual spore production has proved so successful as a means of rapid dispersal to new habitats that sexual reproduction has disappeared, or, at least, has not been discovered. Even in the absence of the sexual stage it is now often possible to assign these fungi to the phyla Ascomycota or Basidiomycota on the basis of DNA sequence analysis. There is no longer any separate formal grouping for those fungi that appear to be strictly asexual. Nonetheless, mycologists continue to employ the asexual reproductive characteristics of moulds, at least for routine identification purposes. Yeasts are identified primarily according to their ability to ferment sugars and to assimilate carbon and nitrogen compounds.
Fungal diseases of man
Fungal pathogens
There are at least 100 000 named species of fungi. However, fewer than 500 have been recognized to cause disease (mycosis) in man or animals. Most are moulds, but there are a number of pathogenic yeasts and some are dimorphic. Dimorphic fungi usually change from a multicellular mould form in the natural environment to a budding single-celled yeast form when causing infection. In the laboratory, the tissue form can be induced by culture at 37°C on rich media such as blood agar, whereas the mould form develops when incubated at a lower temperature (25–30°C) on a less rich medium such as Sabouraud dextrose agar.
Some fungi, such as the systemic pathogens Histoplasma capsulatum and Blastomyces dermatitidis, can establish an infection in all exposed individuals. Others, such as Candida and Aspergillus species, are opportunist pathogens that ordinarily cause disease only in a compromised host. In some mycoses the form and severity of the infection depend on the degree of exposure to the fungus, the site and method of entry into the body, and the level of immunocompetence of the host.
Some fungi may cause serious, occasionally fatal, toxic effects in man, either following ingestion of poisonous toadstools or consumption of mouldy food that contains toxic secondary metabolites (mycotoxins). Allergic disease of the airways may result from inhalation of fungal spores.
Epidemiology
Most human fungal infections are caused by fungi that grow as saprobes in the environment. Infection is acquired by inhalation, ingestion or traumatic implantation. Some yeasts are human commensals and cause endogenous infections when there is some imbalance in the host. Only dermatophyte infections are truly contagious.
Many fungal diseases have a worldwide distribution, but some are endemic to specific geographical regions, usually because the causal agents are saprobes restricted in their distribution by soil and climatic conditions.
Types of infection
Superficial mycoses
Diseases of the skin, hair, nail and mucous membranes are the most common of all fungal infections and have a worldwide distribution.
• Dermatophytosis (ringworm) is a complex of diseases affecting the outermost keratinized tissues of hair, nail and the stratum corneum of the skin; it is caused by a group of closely related mould fungi called dermatophytes, which can digest keratin. Dermatophyte infections occur in both man and animals.
• Yeast infections affect the skin, nail and mucous membranes of the mouth and vagina, and are usually caused by commensal Candida species, notably Candida albicans. Infection is generally endogenous in origin but genital infection can be transmitted sexually. The yeast Malassezia furfur, a skin commensal, can cause an infection of the skin called pityriasis versicolor.
Subcutaneous mycoses
Mycoses of the dermis, subcutaneous tissues and adjacent bones that show slow localized spread occur mainly in the tropics and subtropics; they usually result from the traumatic inoculation of saprobic fungi from soil or vegetation into the subcutaneous tissue. The principal subcutaneous mycoses are mycetoma, chromoblastomycosis and sporotrichosis.
Systemic mycoses
Deep-seated fungal infections generally result from the inhalation of air-borne spores produced by the causal moulds, present as saprobes in the environment. Initially there is a pulmonary infection, but the organism may become disseminated to other organs. The organisms that cause systemic mycoses can be divided into two distinct groups: the true pathogens and the opportunistic pathogens. The first of these groups is comprised mostly of dimorphic fungi and infections occur mainly in the Americas. The principal diseases are:
• blastomycosis (caused by Blastomyces dermatitidis)
• coccidioidomycosis (Coccidioides immitis and C. posadasii)
Systemic mycoses caused by opportunistic pathogens such as Aspergillus, Candida and Cryptococcus species have a more widespread distribution. These infections are being seen with increasing frequency in patients compromised by disease or drug treatment. In transplant patients, for example, these fungi are among the most frequent causes of death due to infection.
Incidence
The incidence of all the mycoses is related directly to factors that affect the degree of exposure to the causal fungi, such as living conditions, occupation and leisure activities.
• Dermatophytosis of the foot (athlete’s foot), with associated infections of nails and groin, occurs most commonly in swimmers, sportspersons and industrial workers who use communal bathing facilities.
• Animal dermatophytosis is an occupational hazard for farmers, veterinarians and others closely associated with animals.
• Agricultural workers in warm climates who wear little protective clothing frequently contract subcutaneous infections following minor injuries from vegetation.
• In developed countries the incidence of infections due to opportunistic fungal pathogens has increased following major advances in healthcare that have led to increases in the size of the population at risk.
• In many developing countries the acquired immune deficiency syndrome (AIDS) pandemic has been associated with a marked increase in the rate of opportunistic fungal infections. High mortality rates have been reported from countries where adequate treatment is often unavailable.
• The incidence of several of the systemic mycoses that are endemic in the Americas has increased as a result of urban development and changing land use in the endemic areas. Increased international travel and tourism has also led to a rise in the number of cases of disease among individuals who normally reside in countries far from the endemic areas.
Diagnosis
Diagnosis of fungal infections is based on a combination of clinical observation and laboratory investigation.
Clinical investigation
Superficial and subcutaneous mycoses often produce characteristic lesions, but they may also closely resemble and be confused with other diseases. In addition, the appearance of lesions may be modified beyond recognition by previous therapy, for example with topical steroids.
The first indication that a patient may have a systemic mycosis is often their failure to respond to antibacterial antibiotics. As early diagnosis considerably increases the chances of successful treatment, it is important that the possibility of fungal involvement be considered from the outset, particularly in those known to be at risk of developing a fungal infection. Computed tomography is widely used to help diagnose Aspergillus infections and other invasive mycoses.
Laboratory diagnosis
Laboratory diagnosis depends on:
• recognition of the pathogen in tissue by microscopy
• isolation of the causal fungus in culture
• the use of serological tests
• detection of fungal DNA by the polymerase chain reaction (PCR).
It is important that the correct type of specimen, together with adequate clinical data, is sent to the laboratory so that the appropriate investigations can be carried out. Information on factors such as travel or residence abroad, animal contacts and the occupation of the patient enable the laboratory staff to direct their investigations towards a particular fungus or group of fungi when appropriate.
Types of specimen
Skin scales, nail clippings and scrapings of the scalp that include hair stubs and skin scales are the most suitable specimens for the diagnosis of ringworm; these are collected into folded paper squares for transport to the laboratory. Swabs should be taken from suspected Candida infections from the mucous membranes and preferably sent to the laboratory in ‘clear’ transport medium. For subcutaneous infections the most suitable specimens are scrapings and crusts, aspirated pus and biopsies. In suspected systemic infection, specimens should be taken from appropriate sites.
Direct microscopy
Most specimens can be examined satisfactorily in wet mounts after partial digestion of the tissue with 10–20% potassium hydroxide. Addition of Calcofluor white and subsequent examination by fluorescence microscopy enhances the detection of most fungi as the fluorescent hydroxide–Calcofluor binds to the fungal cell walls. Gram films may also be used for the diagnosis of yeast infections of mucous membranes. Giemsa staining of smears is advised for detection of the yeast cells of H. capsulatum because of their small size.
Histology
Invasive procedures are required to obtain specimens for histological examination. Although sometimes necessary to provide firm evidence of invasive disease, such procedures are often impracticable on patients who are already seriously ill. Haematoxylin and eosin staining is seldom of value for demonstrating fungi in tissue, and specific fungal stains such as periodic acid–Schiff and Grocott–Gomori methenamine–silver are widely used.
Culture
Most pathogenic fungi are easy to grow in culture. Sabouraud dextrose agar and 4% malt extract agar are most commonly used. These may be supplemented with chloramphenicol (50 mg/L) to minimize bacterial contamination and cycloheximide (500 mg/L) to reduce contamination with saprophytic fungi. Many fungal pathogens have an optimum growth temperature below 37°C. Consequently, cultures are incubated at 25–30°C and at 37°C. With some dimorphic pathogens, enriched media such as brain–heart infusion or blood agar are used to promote growth of the yeast phase.
Many fungi develop relatively slowly and cultures should be retained for at least 2–3 weeks (in some cases up to 6 weeks) before being discarded; yeasts usually grow within 1–5 days. Moulds are identified by their macroscopic and microscopic morphology. Yeasts are identified by sugar fermentation and their ability to assimilate carbon and nitrogen sources. Commercial kits are available for the identification of medically important yeasts.
Culture may provide unequivocal evidence of fungal infection when established pathogens are isolated or when fungi are recovered from normally sterile sites. However, when commensals such as Candida species are isolated, results must be interpreted according to the quantity of the fungus isolated, the source and clinical evidence.
Treatment
There are relatively few therapeutically useful antifungal agents compared with the large number of antibacterial agents that are available (see Chs 5 & 67). As fungi and human beings are both eukaryotes, most substances that kill or inhibit fungal pathogens are also toxic to the host. Antifungal agents vary considerably in their spectrum of activity (see Table 5.4, p. 61). Most exploit differences in the sterol composition of the fungal and mammalian cell membranes, although the echinocandins (anidulafungin, caspofungin and micafungin) interfere with β-glucan synthesis in the fungal cell wall.
Most antifungal agents are available only for topical use. Relatively few can be administered systemically.
• Amphotericin B and the echinocandins are given parenterally because of poor absorption from the gastrointestinal tract.
• Fluconazole, itraconazole, posaconazole, voriconazole and flucytosine are available for oral and/or parenteral administration.
• Terbinafine and griseofulvin are usually administered orally.
• Amphotericin B has long been the treatment of choice in life-threatening disease, despite its toxicity; liposomal and lipid complex formulations are less toxic but much more expensive.
• A combination of amphotericin B and flucytosine reduces the likelihood of the emergence of resistance to flucytosine. Combinations of azole drugs and amphotericin B are seldom used therapeutically.
• New antifungals are being evaluated for use in systemic mycoses. Posaconazole appears to be effective in histoplasmosis and mucormycosis, but it is not yet licensed for these indications.
Antifungal prophylaxis is often used to help prevent opportunistic infections in patients undergoing solid-organ, blood or marrow transplants and in those with haematological malignancies. Oral or topical antifungals are also used to prevent recurrent vaginal candidosis.
Primary or acquired resistance is not a major problem. Resistance to azole antifungals is sometimes encountered, especially after prolonged fluconazole therapy of oropharyngeal Candida infections in persons with AIDS. Some yeast species (e.g. Candida krusei, Candida glabrata) are inherently resistant to triazoles such as fluconazole. Consequently, susceptibility testing is often carried out for fluconazole and for any drug that fails to produce the expected therapeutic response.
Superficial infections
Dermatophytosis
Dermatophyte infections are common diseases of the stratum corneum of the skin, hair and nail; they are also referred to as ringworm or as tinea, a name that is qualified by the site affected, for example tinea pedis or tinea capitis for infections of the feet or scalp, respectively. These infections are caused by about 20 species of fungi that are grouped into three genera: Trichophyton, Microsporum and Epidermophyton. Some species are worldwide in distribution, whereas others are restricted to, or are more common in, particular parts of the world.
Many dermatophyte species produce two types of asexual spore: multi-celled macroconidia and single-celled microconidia. Classification into the three genera Trichophyton, Microsporum and Epidermophyton is based on the morphology of the macroconidia, although the identification of species is also based on the shape and disposition of the microconidia and the macroscopic appearance of the colonies.
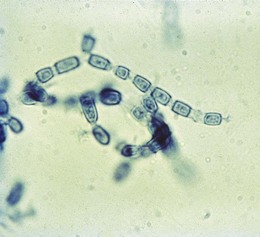
The clinical appearances of dermatophyte infections are the result of a combination of direct tissue damage caused by the fungus and of the immune response of the host. The damage to tissue is due to a combination of mechanical pressure and enzymatic activities. In tissue the dermatophytes take the form of branching hyphae, which may eventually break up into arthroconidia, particularly in infected hair (Fig. 61.1).
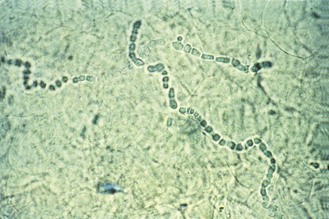
Fig. 61.1 Microscopical appearance of infected skin scrapings showing the development of arthroconidia.
(Reproduced with permission from Richardson MD, Warnock DW, Campbell CK 1995 Slide Atlas of Fungal Infection: Superficial Fungal Infections. Blackwell Science, Oxford.)
Epidemiology
The dermatophytes can be divided into three groups depending on whether their normal habitat is the soil (geophilic species), animals (zoophilic species) or man (anthropophilic species). Members of all three groups can cause human infection, but their different natural reservoirs have important epidemiological implications in relation to the acquisition, site and spread of human disease.
Although geophilic dermatophytes occasionally cause infection in both animals and man, their normal habitat is the soil. Members of the anthropophilic and zoophilic groups are thought to have evolved from these and other keratinophilic soil-inhabiting fungi, different species having adapted to different natural hosts. Individual members of the zoophilic group are often associated with a particular animal host, for instance M. canis with cats and dogs and T. verrucosum with cattle. However, these organisms can also spread to man. The anthropophilic species are the most highly specialized group of dermatophytes. They rarely infect other animals and often show a strong preference for a particular body site, only occasionally being found in other regions. For instance, M. audouinii commonly infects scalp hair, whereas E. floccosum is usually found on the skin.
Infections are spread by direct or indirect contact with an infected individual or animal. The infective particle is usually a fragment of keratin containing viable fungus. Indirect transfer may occur via the floors of swimming pools and showers or on brushes, towels and animal grooming implements. Dermatophytes can remain viable for long periods of time and the interval between deposition and transfer may be considerable. In addition to exposure to the fungus, some abnormality of the epidermis, such as slight peeling or minor trauma, is probably necessary for the establishment of infection.
In industrialized countries, tinea capitis is relatively uncommon, and is caused by dermatophytes of both human and animal origin, although infections with the anthropophilic species T. tonsurans are on the increase among urban populations in Europe and the Americas. However, the use of communal bathing facilities has resulted in a considerable increase in the incidence of tinea pedis and associated nail and groin infections. These now comprise about 75% of all dermatophyte infections diagnosed in temperate zones.
In developing countries, particularly in warm climates, scalp, body and groin infections predominate, with T. rubrum and T. violaceum among the most common causes.
Clinical features
Lesions vary considerably according to the site of the infection and the species of fungus involved. Sometimes there is only dry scaling or hyperkeratosis, but more commonly there is irritation, erythema, oedema and some vesiculation. More inflammatory lesions with weeping vesicles, pustules and ulceration are usually caused by zoophilic species.
In skin infections of the body, face and scalp, spreading annular lesions with a raised, inflammatory border are usually produced (Fig. 61.2). Lesions in body folds, such as the groin, tend to spread outwards from the flexures. In tinea pedis, infection is often confined to the toe clefts, but it can spread to the sole; sometimes, painful secondary bacterial infection occurs in the toe clefts.
In nail infection, the nail becomes discoloured, thickened, raised and friable; most nail infections are due to T. rubrum and involve the toenails (Fig. 61.3).
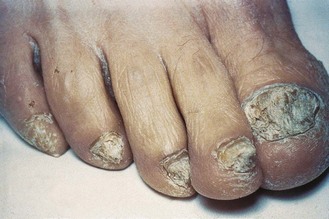
Fig. 61.3 Toenail infection due to Trichophyton rubrum.
(Reproduced with permission from Richardson MD, Warnock DW, Campbell CK 1995 Slide Atlas of Fungal Infection: Superficial Fungal Infections. Blackwell Science, Oxford.)
In scalp infections there is scaling and hair loss, the extent of which depends on the causal fungus. Some zoophilic species give rise to a highly inflammatory, raised, suppurating lesion called a kerion; kerions may also occur in the beard area of adults. It is important that tinea capitis is recognized and treated promptly because it can lead to scarring and permanent hair loss.
In scalp infection the fungus invades the hair shaft and the hyphae then break up into chains of arthroconidia. In some species (e.g. T. tonsurans, T. violaceum) the arthroconidia are retained within the hair shaft (endothrix invasion), whereas in others (e.g. Microsporum species, T. verrucosum) they are produced in a sheath surrounding the hair shaft (ectothrix invasion). The pattern of hair invasion affects the clinical appearance of the lesion.
• In endothrix infection the hair breaks off at, or just below, the mouth of the follicle to give what is described as a ‘black dot’ appearance.
• In ectothrix infection the hair usually breaks off 2–3 mm above the mouth of the follicle, leaving short stumps of hair.
• In favus, caused by T. schoenleinii, fungal growth within the hair is minimal. The hair remains intact, but intense fungal growth within and around the hair follicle produces a waxy, honeycomb-like crust on the scalp (Fig. 61.4).
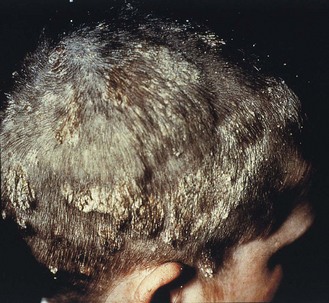
Fig. 61.4 Tinea capitis (favus) due to Trichophyton schoenleinii.
(Reproduced with permission from Richardson MD, Warnock DW, Campbell CK 1995 Slide Atlas of Fungal Infection: Superficial Fungal Infections. Blackwell Science, Oxford.)
Infections of the groin, hands and nails are nearly always secondary to infection of the feet and are usually (except in developing countries) caused by T. rubrum, T. mentagrophytes or E. floccosum. Mixed infections also occur.
Occasionally, patients with inflammatory infections develop a secondary rash known as an id reaction, which is thought to be an immunological reaction to fungal antigens. In patients with tinea pedis this takes the form of a vesicular eczema of the hands, whereas patients with tinea capitis (especially kerion) develop a follicular rash, usually on the trunk or limbs. These secondary lesions do not contain viable fungus and they disappear spontaneously when the infection subsides.
Laboratory diagnosis
Dermatophyte infections may be reliably diagnosed in the laboratory by direct microscopical examination and culture of skin, crusts, hair and nail.
Collection of samples
Skin, hair and nail samples are best collected into folded squares of black paper or card, which can be fastened with a paper clip. The use of paper allows the specimen to dry out, which helps reduce bacterial contamination and provides conditions under which specimens can be stored for 12 months or more without appreciable loss in viability of the fungus.
Nail samples should be collected by taking clippings from any discoloured, dystrophic or brittle parts of the nail and, importantly, by scraping material from underneath the nail. The sample should be taken from as far back as possible from the free edge of the nail.
Scales from skin lesions should be collected by scraping outwards with a blunt scalpel from the edges of the lesions, where most viable fungus is likely to be. Specimens from the scalp should include hair stubs, the contents of plugged follicles and skin scales. Infected hairs are usually easy to pluck from the scalp with forceps. Cut hairs are unsatisfactory because the focus of infection is usually below or near the surface of the scalp.
Wood’s lamp
This is a source of long-wave ultraviolet light that can be used to detect fluorescence in infected hair. It is especially useful for the detection of inconspicuous scalp lesions, and to select infected hairs for laboratory investigation.
Hairbrush sampling
Adequate material from minimal lesions may be obtained by brushing the scalp with a sterilized plastic hairbrush or scalp massage pad; this is then used to inoculate an appropriate culture medium by pressing the brush or pad spines into the agar.
Processing of specimens
If there is insufficient material for both microscopy and culture, the sample should be used for culture, since this is generally the more sensitive procedure (except for nails).
The specimen should first be examined macroscopically; hair samples are examined under a Wood’s lamp. Material from representative parts, and any fluorescent hairs, are divided up into 1–2-mm fragments with a sterile scalpel blade before microscopical examination and culture.
Direct microscopy
Microscopy of wet mounts of keratinous material in potassium hydroxide is simple and reliable. The preparation is allowed to stand for 15–20 min to digest and ‘clear’ the keratin. Dermatophytes are seen in skin and nail as branching hyphae, which often appear slightly greenish in colour and run across the outlines of the colourless host cells. With Calcofluor (see above) the cell outlines fluoresce white.
Culture
Small fragments of keratinous material are planted or scattered on Sabouraud dextrose or 4% malt extract agar and incubated at 28–30°C for up to 2 weeks; room temperature is adequate but the dermatophytes grow more slowly. Only T. verrucosum grows well at 37°C.
Identification is based on colonial appearance and colour, pigment production, and the micromorphology of any spores produced. Special tests exist for differentiating certain morphologically similar species. Thus, the ability of T. mentagrophytes to produce urease within 2–4 days distinguishes it from T. rubrum, and the ability to grow on rice grains distinguishes M. canis from M. audouinii.
Treatment and prevention
Topical therapy is satisfactory for most skin infections, although oral antifungals are required to treat infections of the nail and scalp, and severe or extensive skin infections.
Topical agents include azole compounds, terbinafine, amorolfine and ciclopirox olamine. Oral griseofulvin is useful for scalp, skin and fingernail infections, but gives poor results in toenail infections, even after 18 months’ therapy. Terbinafine and itraconazole have largely replaced griseofulvin for the treatment of nail infections because of their much better cure rates and shorter periods of treatment (around 85% cure for toenails after 3 months’ therapy).
Relatively little has been done to control the spread of dermatophyte infections. The prophylactic use of antifungal foot powder after bathing helps to reduce the spread of infection among swimmers. Foot-baths containing antiseptic solutions, which are commonplace in swimming pools, are of no value.
Superficial candidosis
Superficial Candida infections involving the skin, nails and mucous membranes of the mouth and vagina are very common throughout the world. Candida albicans accounts for 80–90% of cases, but other species, notably C. glabrata, C. parapsilosis, C. tropicalis, C. krusei and C. guilliermondii, may occur.
On Sabouraud dextrose agar Candida species grow predominantly in the yeast phase as round or oval cells, 3–8 mm in diameter. A mixture of yeast cells, pseudohyphae and true hyphae is found in vivo and under micro-aerophilic growth conditions on nutritionally poor media. C. glabrata does not form either hyphae or pseudohyphae.
Epidemiology
Candida species, usually C. albicans, are found in small numbers in the commensal flora (mouth, gastrointestinal tract, vagina, skin) of about 20% of the normal population. The carriage rate tends to increase with age and is higher in the vagina during pregnancy. Commensal yeasts are more prevalent among patients in hospital. Yeast overgrowth and infection occur when the normal microbial flora of the body is altered or when host resistance to infection is lowered by disease.
In most cases, infection is derived from an individual’s own endogenous reservoir. In some instances, however, transmission from person to person can occur; for example neonatal oral candidosis is more common in infants born of mothers with vaginal candidosis. The hands of healthcare workers are another potential source of neonatal infection.
Individuals colonized with Candida species possess numerous non-specific and immunological defences to prevent infection. In superficial candidosis the non-specific inhibitory factors include inhibitors in serum, such as unsaturated transferrin, and epithelial proliferation. Specific defence largely depends on the development of active T cell-mediated immunity.
Both general and local predisposing factors are important in the development of oropharyngeal candidosis. Debilitated and immunosuppressed individuals, such as persons with diabetes mellitus, stem cell transplant recipients and those with human immunodeficiency virus (HIV) infection, are more susceptible to infection. Local factors, such as xerostomia and trauma from unhygienic or ill-fitting dentures, are also important. Local tissue damage is also a critical factor in the development of cutaneous forms of candidosis. Most infections occur in moist, occluded sites and follow maceration of tissue.
Candida vaginitis is more common during pregnancy. The lower prevalence of this infection after menopause emphasizes the hormonal dependence of the infection. Most cases of vaginal candidosis in older women are associated with uncontrolled diabetes mellitus or the use of exogenous oestrogen replacement treatment.
Clinical features
Mucosal infection
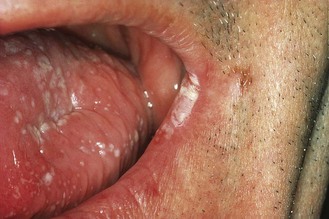
This is the most common form of superficial candidosis. Discrete white patches develop on the mucosal surface, and may eventually become confluent and form a curd-like pseudomembrane (Fig. 61.5).
In oropharyngeal candidosis white flecks appear on the buccal mucosa, tongue, and the hard and soft palate; although these are adherent, they can be removed. The surrounding mucosa is red and sore. This form of oropharyngeal candidosis occurs most frequently in infancy and old age, or in severely immunocompromised patients, including those with AIDS. Other forms of oral candidosis occur:
• lesions in the occluded area under the denture in those who wear dentures
• painful infection of the tongue in some individuals receiving antibiotic therapy
• chronic infection with extensive leucoplakia and infection of the angles of the mouth (angular cheilitis).
In vaginal candidosis, itching, soreness and a non-homogeneous white discharge accompany typical white lesions on the epithelial surfaces of the vulva, vagina and cervix. Sometimes the mucosa simply appears inflamed and friable. The perivulval skin may become sore and small satellite pustules may appear around the perineum and natal cleft. Some women suffer recurrent episodes.
Chronic, intractable oropharyngeal candidosis, which may extend to give oesophageal infection, is common in persons with HIV infection, although the use of combinations of antiretroviral drugs has reduced its incidence. The appearance of this infection can be the indicator of the transition from HIV-positive status to AIDS.
Skin and nail infection
Cutaneous candidosis is less common than dermatophytosis. The lesions usually develop in warm, moist sites such as the axillae, groin and submammary folds. In infants, Candida species are often secondary invaders in napkin dermatitis. Infection of the finger webs, nail folds and nails is associated with frequent immersion of the hands in water.
Chronic mucocutaneous candidosis
This is a rare form of candidosis that usually becomes apparent in childhood and takes the form of a persistent, sometimes granulomatous, infection of the mouth, scalp, hands, feet and nails. In some cases, disfiguring hyperkeratotic lesions develop on the scalp and face. Some of those who develop this condition have subtle defects in lymphocyte function.
Laboratory diagnosis
Specimens of skin and nail are collected in the same way as for suspected dermatophytosis. For infections of the mouth or vagina, scrapings taken with a blunt scalpel or a spatula from areas with white plaques or erythema are better than swabs if the material is to be processed immediately. However, swabs are more convenient for transport to the laboratory, and they are better for collecting vaginal discharge. Swabs should first be moistened with sterile water or saline before taking the sample and should be sent to the laboratory in ‘clear’ transport medium.
In Gram-stained smears of mucous membrane samples the fungus is seen as budding Gram-positive yeast cells; pseudohyphae are usually present except in the case of C. glabrata (Fig. 61.6). Contrary to popular belief, the presence of Candida pseudohyphae in clinical material does not confirm infection with the organism, particularly as it may have developed in the period between collection and processing of the sample.
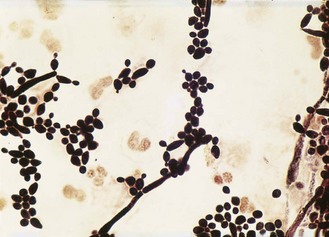
Fig. 61.6 Microscopical appearance of Candida albicans yeast cells and pseudohyphae in a Gram-stained vaginal smear.
Candida species grow well on Sabouraud medium or on blood agar at 25–37°C; typical yeast colonies appear within 1–2 days. C. albicans isolates can be identified by the germ tube test: after incubation in serum at 37°C for 1.5–2 h, C. albicans produces short hyphae known as germ tubes. Other yeasts may be identified with one of the commercial kits, or by fermentation and assimilation tests.
Quantification of growth, especially in the case of vulvovaginal samples, may help the clinician to distinguish between commensal carriage and infection.
Treatment and prevention
Most superficial infections respond well to topical therapy with an imidazole. In oral candidosis, nystatin, amphotericin B or miconazole may be effective in lozenge or gel form. Most patients with vaginal candidosis can be treated successfully with a single application of a topical imidazole, or with oral fluconazole or itraconazole. Intermittent prophylaxis with an oral azole or vaginal pessaries is of benefit in controlling recurrent vaginal candidosis.
Treatment of chronic paronychia involves a combination of antifungal therapy, nail care and avoidance of prolonged exposure to water by use of protective gloves; patients should dry their hands carefully after washing. Regular application of an azole lotion or an azole given orally, sometimes in conjunction with a topical steroid and an antibacterial agent, is the most appropriate therapy but it may take several months to cure the condition; antifungal creams or ointments are less effective.
Oral therapy is essential for the treatment of intractable chronic Candida infections; treatment is given until remission is achieved but in some patients, for instance those with AIDS, relapse is common, and intermittent or prolonged therapy may be required. This may, however, lead to the development of resistance, as occasionally happens with fluconazole.
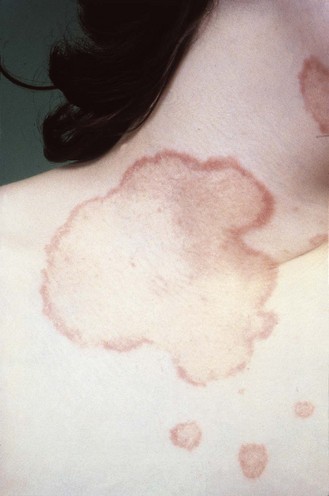
Pityriasis versicolor
This is a common, mild and often recurrent infection of the stratum corneum that produces a patchy discoloration of the skin caused by lipophilic yeasts of the genus Malassezia. These organisms require lipids for growth and special media containing Tween and lipid supplements have been developed.
On normal skin and in conditions such as dandruff and seborrhoeic dermatitis (in which its precise role is uncertain), Malassezia occurs as an oval or bottle-shaped yeast, which characteristically produces buds on a broad base. In pityriasis versicolor the organism produces predominantly round yeast cells and short hyphae.
Epidemiology
Malassezia species are common members of the normal skin flora and most infections are thought to be endogenous. The incidence of skin colonization rises from around 25% in children to almost 100% in adolescents and adults. Disease is probably related to host and environmental factors. It is very common in hot, humid tropical climates, where 30–40% of adults may be affected.
Clinical features
Small patches of well demarcated, non-inflammatory scaling are usually present on the upper trunk or neck; these may appear hypopigmented or hyperpigmented, depending on the degree of pigmentation of the surrounding skin. The lesions tend to spread and coalesce, and occasionally they spread to other sites.
Other superficial infections
Skin and nail
Certain non-dermatophyte moulds may cause infection of skin and nail. It is important that these are recognized because they are often resistant to the agents used to treat dermatophytosis and superficial candidosis.
In the UK, about 5% of fungal nail infections are caused by non-dermatophyte moulds. Scopulariopsis brevicaulis, a ubiquitous saprophyte of soil, is the most common, although other saprophytic moulds such as Fusarium, Aspergillus and Acremonium species are also occasionally implicated. There is some debate as to whether these moulds are primary pathogens of nails – infection usually follows trauma and in many cases they are found in nails along with a dermatophyte.
Non-dermatophyte mould infections do not respond to existing antifungal agents. Attempts may be made to remove the nail with topical 40% urea paste.
Otomycosis
About 10–20% of chronic ear infections are due to fungi. The disease has a worldwide distribution but is more common in warm climates. Topical antibiotics and steroids are predisposing factors. The most common causes are species of Aspergillus, in particular A. niger. The fungi are easy to see in material from swabs or scrapings and grow readily in culture.
Treatment with topical antifungals is usually successful, although relapse is common. Any concurrent bacterial infection or other underlying abnormality should also be treated.
Mycotic keratitis
Fungal infections of the cornea usually follow traumatic implantation of spores. Topical antibiotics and steroids are important predisposing factors. These infections occur most often in hot climates and are caused by common saprophytic moulds, in particular Aspergillus and Fusarium species. Culture results should be interpreted with care as these opportunist pathogens are also encountered as contaminants. Superficial swabs are of no value for laboratory investigation, and scrapings should be taken from the base or edge of the ulcer. The branched, septate hyphae may be rather sparse in potassium hydroxide mounts and some of the material should also be stained with periodic acid–Schiff or Grocott–Gomori methenamine–silver.
Management entails surgical debridement of infected tissue, discontinuation of topical corticosteroids, and topical or oral treatment with an antifungal drug. Topical treatment with natamycin is often successful, but oral treatment with an azole drug is required in patients with severe or worsening lesions. Even with intensive treatment, corneal perforation can occur.
Subcutaneous infections
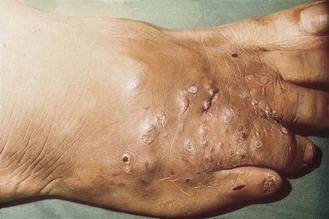
Mycetoma
Mycetoma is a chronic granulomatous infection of the skin, subcutaneous tissues, fascia and bone that most often affects the foot or the hand. It may be caused by one of a number of different actinomycetes (actinomycetoma) (see Ch. 20) or moulds (eumycetoma). The disease is most prevalent in tropical and subtropical regions of Africa, Asia, and Central and South America.
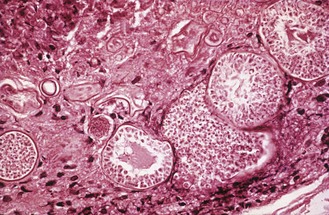
A large number of organisms have been implicated in this disease, including species of Madurella, Leptosphaeria, Acremonium, Pseudallescheria, Actinomadura, Nocardia and Streptomyces. Within host tissues the organisms develop to form compacted colonies (grains) 0.5–2 mm in diameter, the colour of which depends on the organism responsible; for example, in unstained preparations Madurella grains are black and Actinomadura pelletieri grains are red (Fig. 61.7).
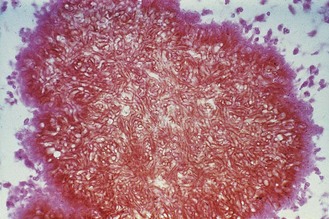
Fig. 61.7 Microscopical appearance of Madurella grisea in a stained mycetoma grain.
(Reproduced with permission from Richardson MD, Warnock DW, Campbell CK 1995 Slide Atlas of Fungal Infection: Subcutaneous and Unusual Fungal Infections. Blackwell Science, Oxford.)
Epidemiology
Infection follows traumatic inoculation of the organism into the subcutaneous tissue from soil or vegetation, usually on thorns or wood splinters. Consequently, the disease occurs most frequently in agricultural workers, in whom minor penetrating skin injuries are common.
Clinical features
Localized swollen lesions that develop multiple draining sinuses are usually found on the limbs, although infections occur on other parts of the body (Fig. 61.8). There is often a long period between the initial infection and formation of the characteristic lesions; spread from the site of origin is unusual but may occur, particularly from the foot up the long bones of the leg.
Laboratory diagnosis
The presence of grains in pus collected from draining sinuses or in biopsy material is diagnostic. The grains are visible to the naked eye and their colour may help to identify the causal agent. Grains should be crushed in potassium hydroxide and examined microscopically to differentiate between actinomycetoma and eumycetoma; material from actinomycetoma grains may be Gram stained to demonstrate the Gram-positive filaments. Samples should also be cultured, at both 25–30°C and 37°C, on brain–heart infusion agar or blood agar for actinomycetes and on Sabouraud agar (without cycloheximide) for fungi. The fungi that cause eumycetoma are all septate moulds that appear in culture within 1–4 weeks, but their identification requires expert knowledge.
Treatment
The prognosis varies according to the causal agent, so it is important that the identity is established. Actinomycetoma responds well to medical treatment: combinations of streptomycin with co-trimoxazole or dapsone are often effective, but an average of 9 months’ therapy is required. In eumycetoma, chemotherapy is ineffective and radical surgery is usually necessary. However, some antifungals have yet to be properly evaluated in this condition.
Chromoblastomycosis
This disease, also known as chromomycosis, is a chronic, localized infection of the skin and subcutaneous tissues, characterized by slow-growing verrucous lesions usually involving the limbs. The disease is encountered mainly in the tropics, in Central and South America, and Madagascar. The principal causes are Fonsecaea pedrosoi, F. compacta, Phialophora verrucosa and Cladophialophora carrionii. Like mycetoma, infection follows traumatic inoculation of the organism into the skin or subcutaneous tissue, and is seen most often among those with outdoor occupations.
Laboratory diagnosis
The characteristic clusters of brown-pigmented, thick-walled fungal cells are relatively easy to see on microscopical examination of skin scrapings, crusts and pus. Culture on Sabouraud agar at 25–30°C yields slow-growing, greenish grey to black, compact, folded colonies. Cultures should be incubated for 4–6 weeks. Specific identification of these closely related fungi is usually left to a reference laboratory.
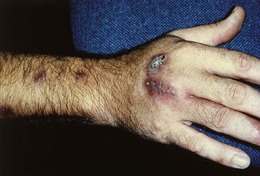
Sporotrichosis
Sporotrichosis is a chronic, pyogenic, granulomatous infection of the skin and subcutaneous tissues that may remain localized or show lymphatic spread. It is caused by Sporothrix schenckii, which is found in the soil and on plant materials, such as wood and sphagnum moss. The disease is worldwide in distribution, but occurs mainly in Central and South America, parts of the USA and Africa, and Australia; it is rare in Europe.
S. schenckii is a dimorphic fungus. In nature and in culture at 25–30°C, it develops as a mould with septate hyphae. The yeast phase is formed in tissue and in culture at 37°C, and is composed of spherical or cigar-shaped cells (1–3 × 3–10 µm).
Epidemiology
Minor trauma, such as abrasions or wounds due to wood splinters, is often sufficient to introduce the organism. Infection occurs mainly in adults, and is more common among individuals whose work or recreational activities bring them into contact with soil or plant materials, such as gardeners and florists.
Clinical features
Sporotrichosis presents most frequently as a nodular, ulcerating disease of the skin and subcutaneous tissues, with spread along local lymphatic channels (Fig. 61.9). Typically, the primary lesion is on the hand with secondary lesions extending up the arm. The primary lesion may remain localized or disseminate to involve the bones, joints, lungs and, in rare cases, the central nervous system. Disseminated disease usually occurs in debilitated or immunosuppressed individuals.
Laboratory diagnosis
Diagnosis is confirmed by isolation of the causative organism by culture of swabs from moist, ulcerated lesions or pus aspirated from subcutaneous nodules; biopsy specimens may be necessary in some cases. Direct microscopy is of little value as so few of the small S. schenckii yeast cells are present in diseased tissue. The mycelial phase develops within 7–10 days on Sabouraud agar or blood agar at 25–30°C; the yeast phase develops in 2 days at 37°C. Identification depends on the micromorphology of the mould phase and its conversion to the yeast phase at 37°C.
Other subcutaneous mycoses
Phaeohyphomycosis is a general term used to describe solitary subcutaneous lesions caused by any brown-pigmented mould. If left untreated these lesions slowly increase in size to form a painless abscess. Diagnosis is often made at surgery, and treatment is by excision.
Several other fungi, including Lacazia loboi, Basidiobolus ranarum and Conidiobolus coronatus, occasionally cause subcutaneous infections, usually in the tropics. Surgical excision is often curative in L. loboi infections; antifungal therapy may be of use for the other infections, but the newer drugs have not been properly evaluated.
Systemic mycoses
Coccidioidomycosis
This is primarily an infection of the lungs caused by Coccidioides immitis and C. posadasii, two closely related dimorphic fungi found in the soil of semi-arid regions of the western hemisphere. In the USA, the endemic region includes parts of California, Arizona, New Mexico and Texas. The endemic region extends southwards into the desert regions of northern Mexico, and parts of Central and South America.
In culture and in soil Coccidioides grows as a mould, producing large numbers of barrel-shaped arthroconidia (4 × 6 µm diameter), which are easily dispersed in wind currents (Fig. 61.10). In the lungs the arthroconidia form spherules (up to 120 µm in diameter) which contain numerous endospores (2–4 µm in diameter) (Fig. 61.11). Endospores are released by rupture of the spherule wall and develop to form new spherules in adjacent tissue or elsewhere in the body. In culture the mould colonies are initially moist and white but change within 5–12 days to become floccose and pale grey or brown.
Epidemiology
Infection is acquired by inhalation; the incubation period is 1–3 weeks. The major risk factor for infection is environmental exposure. Outbreaks have been associated with ground-disturbing activities, such as building construction and archaeological excavation, as well as with natural events that result in the generation of dust clouds, such as earthquakes and dust storms. The most serious disseminated forms of the disease are more common among those of black, Asian or Filipino ethnicity, and among pregnant women in the third trimester.
Clinical features
Coccidioides causes a wide spectrum of disease, ranging from a transient pulmonary infection that resolves without treatment, to chronic pulmonary infection, or to more widespread disseminated disease. About 40% of newly infected persons develop an acute symptomatic and often severe influenza-like illness. However, most otherwise healthy persons recover without treatment, their symptoms disappearing in a few weeks. In some cases primary infection may result in chronic, cavitating, pulmonary disease.
Fewer than 1% of infected individuals develop disseminated coccidioidomycosis. This is a progressive disease that usually develops within 3–12 months of the initial infection, although it can occur much later following reactivation of a quiescent infection in an immunosuppressed individual. The clinical manifestations range from a fulminant illness that is fatal within a few weeks if left untreated, to an indolent chronic disease that persists for months or years. One or more sites may be involved, but the skin, soft tissue, bones, joints and the central nervous system are most commonly affected. Meningitis is the most serious complication of coccidioidomycosis, occurring in 30–50% of patients with disseminated disease. Without therapy, it is almost always fatal.
Laboratory diagnosis
Microscopical examination of sputum, pus and biopsy material is helpful as the relatively large size and numbers of mature spherules present makes their detection and identification comparatively straightforward. Material for culture should be inoculated on to screw-capped slopes of Sabouraud agar and incubated at 25–30°C for 1–2 weeks. The fungus can be identified by its colonial morphology and the presence of numerous thick-walled arthroconidia formed in chains from alternate cells of the septate hyphae.
The arthroconidia are highly infectious and are a serious danger to laboratory staff. Consequently, Petri dishes should never be used for isolation of the organism and all procedures should be carried out in a biological safety cabinet under Category 3 containment. Preparations for microscopy should be made only after wetting the colony to reduce spore dispersal.
Serological tests play an important part in diagnosis. The immunodiffusion test is most useful for detection of early primary infection or exacerbation of existing disease; antibodies appear 1–3 weeks after infection but are seldom detectable after 2–6 months, or in patients with disseminated coccidioidomycosis. The latex agglutination test gives similar results to the immunodiffusion test, but is less specific. Complement-fixing antibodies appear 1–3 months after infection and persist for long periods in individuals with chronic or disseminated disease. In most cases the titre is proportional to the extent of infection; failure of the titre to fall during treatment of disseminated coccidioidomycosis is an ominous sign.
Treatment
The historical standard of treatment is intravenous amphotericin B, but oral fluconazole is now used to treat many patients with skin, soft tissue, bone or joint infections. Itraconazole is also effective, but less well tolerated. Because oral fluconazole is so much more benign than intrathecal amphotericin B, it is now the drug of choice for coccidioidal meningitis.
Histoplasmosis
This disease is caused by H. capsulatum, a dimorphic fungus found in soil enriched with the droppings of birds and bats. Histoplasmosis is the most common endemic mycosis in North America, but also occurs throughout Central and South America. In the USA, the disease is most prevalent in states surrounding the Mississippi and Ohio Rivers. Other endemic regions include parts of Africa, Australia, India and Malaysia. H. capsulatum var. duboisii is restricted to the continent of Africa.
H. capsulatum var. capsulatum grows in soil and in culture at 25–30°C as a mould and as an intracellular yeast in animal tissues. The small oval yeast phase cells (2–4 µm diameter) can also be produced in vitro by culture at 37°C on blood agar or other enriched media containing cysteine. In culture the mould colonies are fluffy, white or buff-brown; the mycelium is septate and two types of unicellular asexual spores are usually produced: large round, tuberculate macroconidia (8–15 µm in diameter) are most prominent and are diagnostic, but smaller, broadly elliptical, smooth-walled microconidia (2–4 µm in diameter) are also present in primary isolates (Fig. 61.12). H. capsulatum var. duboisii is morphologically identical to H. capsulatum var. capsulatum in its mycelial phase, but the yeast phase has larger cells (10–15 µm in diameter).
Epidemiology
Infection results from the inhalation of spores; the incubation period is 1–3 weeks. The major risk factor is environmental exposure; longer and more intense exposures usually result in more severe pulmonary disease. Most reported outbreaks have been associated with exposures to sites contaminated with H. capsulatum or have followed activities that disturbed accumulations of bird or bat guano, such as building demolition, soil excavation and caving.
The most serious disseminated forms of the disease are more common among individuals with underlying cell-mediated immunological deficiencies, such as persons with HIV infection, transplant recipients, and those receiving immunosuppressive treatments.
Clinical features
There is a wide spectrum of disease, ranging from a transient pulmonary infection that subsides without treatment, to chronic pulmonary infection, or to more widespread disseminated disease. Many healthy individuals develop no symptoms when exposed to H. capsulatum in an endemic setting. Higher levels of exposure result in an acute symptomatic and often severe flu-like illness, with fever, chills, non-productive cough and fatigue. The symptoms usually disappear within a few weeks, but patients are frequently left with discrete, calcified lesions in the lung.
Disseminated histoplasmosis may range from an acute illness that is fatal within a few weeks if left untreated (often seen in infants, persons with AIDS and solid organ transplant recipients) to an indolent, chronic illness that can affect a wide range of sites. Hepatic infection is common in non-immunosuppressed individuals and adrenal gland destruction is a frequent problem. Mucosal ulcers are found in more than 60% of these patients; central nervous system disease occurs in 5–20% of patients.
The clinical features of H. capsulatum var. duboisii infection differ from those of var. capsulatum infection. The illness is indolent in onset and the predominant sites affected are the skin and bones. Those with more widespread infection involving the liver, spleen and other organs have a febrile wasting illness that is fatal within weeks or months if left untreated.
Laboratory diagnosis
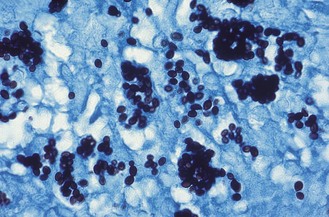
Microscopy of smears of sputum or pus should be stained by the Wright or Giemsa procedure. Blood smears may be positive for H. capsulatum, especially in persons with AIDS. Liver or lung biopsies stained with periodic acid–Schiff or Grocott–Gomori methenamine–silver may provide a rapid diagnosis of disseminated histoplasmosis in some patients. H. capsulatum is seen as small, oval yeast cells, often within macrophages or monocytes (Fig. 61.13).
Specimens should be cultured on Sabouraud agar at 25–30°C to obtain the mycelial phase. Mycelial colonies develop within 1–2 weeks but cultures should be retained for 4 weeks before discarding. The fungus is identified by its colonial morphology and the presence of the characteristic macroconidia and microconidia. Culture at 37°C for the yeast phase is not used for primary isolation, although conversion from the mould to yeast phase is useful to confirm the identity of isolates. Mould cultures of H. capsulatum are a hazard to laboratory staff and consequently screw-capped slopes rather than Petri dishes should be used for isolation.
Serological tests are useful, but cross-reactions can occur, mainly with Coccidioides. Antibody tests fail to detect antibodies in up to 50% of immunosuppressed individuals. Tests for antigen detection in the urine by ELISA are useful in disseminated histoplasmosis but are not widely available outside the USA.
Treatment
Intravenous amphotericin B is recommended for treatment of the most severe forms of disseminated histoplasmosis. Itraconazole is widely used in non-immunocompromised patients with milder forms of disseminated disease and for the continuation of treatment in those who have responded to amphotericin B.
Blastomycosis
This disease is caused by B. dermatitidis, a dimorphic soil-inhabiting fungus. The largest number of cases of blastomycosis has been reported from North America, but the disease is also endemic in Africa, and parts of Central and South America. In the USA, the organism is most commonly found in states surrounding the Mississippi and Ohio Rivers; in Canada, the disease occurs in the provinces that border the Great Lakes.
In culture at 25–30°C B. dermatitidis grows as a mould with a septate mycelium. The colony varies in texture from floccose to smooth and from white to brown in colour. Asexual conidia are produced on lateral hyphal branches of variable length; the oval or pear-shaped conidia are 2–10 µm in diameter. In tissue and in culture at 37°C the fungus grows as a large round yeast (5–15 µm in diameter) that characteristically produces broad-based buds from a single pole on the mother cell (Fig. 61.14).
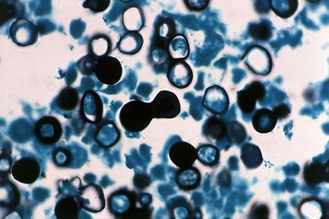
Fig. 61.14 Microscopical appearance of Blastomyces dermatitidis yeast cells in tissue, showing broad-based budding.
Epidemiology
Infection results from inhalation of spores; the incubation period is 4–6 weeks. The disease is more commonly seen in adults than in children, and often occurs in individuals with an outdoor occupation or recreational interest.
Clinical features
Acute pulmonary blastomycosis usually presents as a nonspecific influenza-like illness, similar to that seen with histoplasmosis or coccidioidomycosis. Most otherwise healthy persons recover after 2–12 weeks of symptoms, but some return months later with infection of other sites. Other patients with acute blastomycosis fail to recover and develop chronic pulmonary disease or disseminated infection.
The skin and bones are the most common sites of disseminated disease. The skin is involved in more than 70% of cases; the characteristic lesions are typically raised, with a well-demarcated edge. It is from these skin lesions that the diagnosis is most often made. Osteomyelitis occurs in about 30% of patients, with the spine, ribs and long bones being the most common sites of infection. Arthritis occurs in about 10% of patients. Meningitis is rare, except in immunocompromised individuals.
Laboratory diagnosis
Direct microscopy of pus, scrapings from skin lesions, or sputum usually shows thick-walled yeast cells 5–15 µm in diameter that characteristically produce buds on a broad base; the buds remain attached until they are almost the size of the parent cell, often forming chains of three or four cells. In biopsy material the yeasts are best seen in stained sections.
B. dermatitidis will grow in culture on Sabouraud agar (or blood agar) without cycloheximide, to which the fungus is sensitive. The mycelial phase develops slowly at 25–30°C and cultures must be retained for 6 weeks before discarding. Test-tube slopes rather than Petri dishes are used for culture. Identification is usually confirmed by subculture at 37°C to convert it to the yeast phase.
The most useful serological test is the immunodiffusion test using the A antigen of B. dermatitidis. However, a negative result does not rule out the diagnosis because the sensitivity ranges from 30% for localized infections to 90% for cases of disseminated blastomycosis.
Treatment
Intravenous amphotericin B is used to treat all forms of blastomycosis and is the drug of choice in serious life-threatening infection. Itraconazole follow-on therapy is given once the patient improves. Itraconazole is also the drug of choice in less serious infection that does not involve the central nervous system.
Paracoccidioidomycosis
This is a chronic granulomatous infection caused by the dimorphic fungus P. brasiliensis that may involve the lungs, mucosa, skin and lymphatic system. The disease may be fatal if untreated. Although P. brasiliensis has been isolated from soil, understanding of its precise environmental reservoir remains limited. The endemic region extends from Mexico to Argentina, but the disease is seen most frequently in Brazil, Colombia and Venezuela.
P. brasiliensis grows in the mycelial phase in culture at 25–30°C and in the yeast phase in tissue or at 37°C on brain–heart infusion or blood agar. The mould colonies are slow growing with a variable colonial morphology, although most are white and velvety to floccose in texture with a pale brown reverse. Spore production is usually sparse and best seen in 4–6-week-old cultures. Asexual conidia may be produced but are not characteristic, and identification depends on conversion from the mycelial to the yeast phase. The yeast phase consists of oval or globose cells 2–30 µm in diameter, with small buds attached by a narrow neck encircling the parent cell (Fig. 61.15).
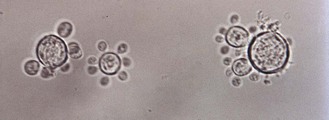
Fig. 61.15 Microscopical appearance of Paracoccidioides brasiliensis yeast cells in sputum, showing multipolar budding.
Epidemiology
Infection is usually acquired by inhalation; the incubation period is unknown. More than 90% of cases of symptomatic disease occur in men, most of whom have agricultural occupations; oestrogen-mediated inhibition of the mould to yeast transformation could help to account for this.
Clinical features
The lungs are the usual initial site of P. brasiliensis infection, but the organism then spreads through the lymphatics to the regional lymph nodes. In most cases the primary infection is asymptomatic. There is evidence of prolonged latent infection before overt disease develops, and a mild, self-limiting pulmonary form of paracoccidioidomycosis probably exists. Children and adolescents sometimes present with an acute disseminated form of infection in which superficial and/or visceral lymph node enlargement is the major manifestation. This presentation is also seen in immunocompromised patients. It has a poor prognosis.
In adults, paracoccidioidomycosis usually presents as an ulcerative granulomatous infection of the oral and nasal mucosa and adjacent skin. In 80% of cases the disease involves the lungs. In some, the liver and spleen, intestines, adrenals, bones and joints, and central nervous system are also involved. The disease is slowly progressive, and may take months or even years to become established.
Laboratory diagnosis
Microscopy of sputum or pus, crusts and biopsies from granulomatous lesions usually reveals numerous yeast cells showing the characteristic multipolar budding, which is diagnostic. In culture the mycelial and yeast phases both develop slowly and cultures must be retained for 6 weeks before discarding. The mould phase can be isolated on Sabouraud agar supplemented with yeast extract at 25–30°C, but colonies may take 2–4 weeks to appear. Serological tests are useful for diagnosis and for monitoring the response to therapy.
Aspergillosis
There are more than 200 species of Aspergillus but fewer than 20 have been implicated in human disease; the most important are A. fumigatus, A. flavus, A. terreus, A. niger and A. nidulans. In immunocompromised individuals, inhalation of spores can give rise to life-threatening invasive infection of the lungs or sinuses and dissemination to other organs often follows (invasive aspergillosis). In non-immunocompromised persons, these moulds can cause localized infection of the lungs, sinuses and other sites. Human disease can also result from non-infectious mechanisms: inhalation of spores can cause allergic symptoms in both atopic and non-atopic individuals.
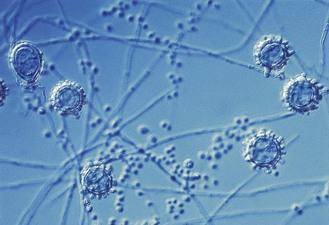
Aspergillus species are ubiquitous in the environment, growing in the soil, on plants, and on decomposing organic matter. These moulds are often found in the outdoor and indoor air, in water, on food items, and in dust. All grow in nature and in culture as moulds with septate hyphae and distinctive asexual sporing structures, termed conidiophores, that bear long chains of conidia (Fig. 61.16).
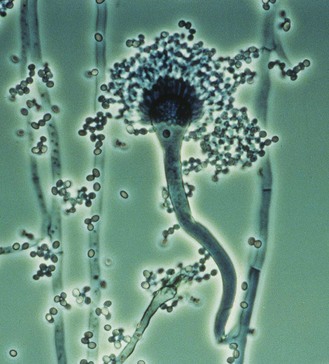
Fig. 61.16 Microscopical appearance of an Aspergillus fumigatus conidiophore.
(Reproduced with permission from Richardson MD, Warnock DW, Campbell CK 1995 Slide Atlas of Fungal Infection: Systemic Fungal Infections. Blackwell Science, Oxford.)
Epidemiology
Inhalation of Aspergillus conidia is the usual mode of infection; less frequently, infection follows the traumatic implantation of spores as in corneal infection (see p. 626), or inadvertent inoculation as in endocarditis.
Invasive aspergillosis has emerged as a major problem in several groups of immunocompromised patients, including those with acute leukaemia, stem-cell and solid organ transplant recipients, and children with chronic granulomatous disease. The likelihood of aspergillosis developing in these individuals depends on a number of host factors, the most important of which is the level of immunosuppression. The mortality rate is high, ranging from 50–100% in almost all groups of immunocompromised patients.
Clinical features
Invasive aspergillosis
This form occurs in severely immunocompromised individuals who have a serious underlying illness. A. fumigatus is the species most frequently involved. The most common initial presentation in the neutropenic patient is an unremitting fever (>38°C), without any respiratory tract symptoms, that fails to respond to broad-spectrum antibiotics.
The lung is the sole site of infection in 70% of patients, but dissemination of infection to other organs often occurs; the central nervous system is involved in 10–20% of cases. There is widespread destructive growth of Aspergillus species in lung tissue and the fungus invades blood vessels, causing thrombosis and infarction; septic emboli may spread the infection to other organs. Invasive aspergillosis has a poor prognosis; early diagnosis is essential for successful management.
Aspergilloma
In this form of aspergillosis, also referred to as fungus ball, the fungus colonizes pre-existing (often tuberculous) cavities in the lung and forms a compact ball of mycelium, eventually surrounded by a dense fibrous wall (Fig. 61.17).
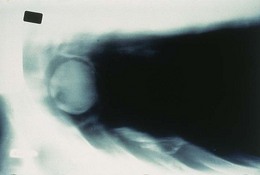
Fig. 61.17 Radiological appearance of an aspergilloma.
(Reproduced with permission from Richardson MD, Warnock DW, Campbell CK 1995 Slide Atlas of Fungal Infection: Systemic Fungal Infections. Blackwell Science, Oxford.)
Aspergillomas are usually solitary. Patients are either asymptomatic or have only a moderate cough and sputum production. Occasional haemoptysis may occur, especially when the fungus is actively growing, and haemorrhage following invasion of a blood vessel is one of the fatal complications. Surgical resection is most often used to treat this condition.
Sinusitis
Aspergillus species, particularly A. flavus and A. fumigatus, may colonize and invade the paranasal sinuses; the infection may spread through the bone to the orbit of the eye and brain. Acute invasive sinusitis is a rapidly progressive disease, most commonly seen in immunocompromised persons. There are also several forms of slowly progressive chronic Aspergillus sinusitis that occur in immunocompetent individuals.
Allergic bronchopulmonary aspergillosis
Allergy to Aspergillus species is usually seen in atopic individuals with raised immunoglobulin (Ig) E levels; about 10–20% of asthmatics react to A. fumigatus. The condition is a form of asthma with pulmonary eosinophilia that manifests as episodes of bronchial obstruction and lung consolidation; the fungus grows in the airways to produce mucous plugs of fungal mycelium that may block off segments of lung tissue and that, when coughed up, are a diagnostic feature.
Laboratory diagnosis
The value of the laboratory in diagnosis varies according to the clinical form of aspergillosis; the diagnosis of invasive disease is particularly difficult.
Direct microscopy
In potassium hydroxide preparations (preferably with Calcofluor to enhance detection) of sputum the fungus appears as non-pigmented septate hyphae, 3–5 µm in diameter, with characteristic dichotomous branching and an irregular outline; rarely the characteristic sporing heads of Aspergillus species are present.
• In allergic aspergillosis there is usually abundant fungus in the sputum and mycelial plugs may also be present.
• In aspergilloma, fungus may be difficult to find on microscopy.
Biopsy may provide a definitive diagnosis, although many clinicians are reluctant to undertake this procedure because of the associated risk in immunosuppressed patients. In tissue sections Aspergillus species are best seen after staining with periodic acid–Schiff or methenamine–silver.
Culture
Aspergillus species grow readily at 25–37°C on Sabouraud agar without cycloheximide; colonies appear after 1–2 days. Isolates can be identified by their colonial appearance and micromorphology. The ability of A. fumigatus to grow well at 45°C can be used to help identify this species or to isolate it selectively.
As aspergilli are among the most common laboratory contaminants, quantification of the amount of fungus in sputum helps to confirm the relevance of a positive culture. However, all isolates from immunocompromised patients must be taken seriously and acted upon.
Large quantities of fungus are usually recovered from the sputum of patients with allergic aspergillosis, but cultures from those with aspergilloma or invasive disease are commonly negative or yield only a few colonies. Blood cultures are negative in invasive disease.
Skin tests
Skin tests with A. fumigatus antigen are useful for the diagnosis of allergic aspergillosis. All patients give an immediate type I reaction and 70% of those with pulmonary eosinophilia also give a delayed type III Arthus reaction.
Serological tests
Immunodiffusion, CIE and ELISA are widely used for the detection of antibodies in the diagnosis of all forms of aspergillosis, particularly aspergilloma and allergic bronchopulmonary aspergillosis. Tests for Aspergillus antibodies are seldom helpful in the diagnosis of invasive infection in immunocompromised patients.
Antigen detection has also been used successfully for diagnosis of invasive aspergillosis by techniques such as ELISA and latex agglutination. However, nucleic acid amplification methods are now being developed for diagnosis of invasive aspergillosis.
Treatment
In invasive aspergillosis, the historical standard of treatment is intravenous amphotericin B (conventional or liposomal). Voriconazole proved superior to amphotericin B in a large clinical trial, and is now used to treat many patients with this disease.
Aspergilloma is treated by surgical excision because antifungal therapy is of little value, but because of the significant morbidity and mortality with this procedure it is reserved for patients with episodes of life-threatening haemoptysis. Allergic forms of aspergillosis are treated with corticosteroids.
Invasive candidosis
In addition to causing mucosal and cutaneous infections (see p. 655–57), Candida species can cause acute or chronic invasive infections in immunocompromised or debilitated individuals. This may be confined to one organ or become widespread (disseminated candidosis).
Epidemiology
In most cases invasive candidosis is endogenous in origin, but transmission of organisms from person to person can also occur. Hospital outbreaks of infection have sometimes been linked to contaminated medical devices, such as vascular catheters, and/or parenteral nutrition. There are also reports of cross-infection due to hand carriage by healthcare workers.
Invasive candidosis is a significant problem in several distinct groups of hospitalized patients:
Invasive candidosis is now more common in patients in intensive care than among neutropenic individuals. The reduced incidence of the disease among the latter group has been attributed to the widespread use of fluconazole prophylaxis. The predominant pathogen in all groups is still C. albicans, but the proportion of serious infections due to less azole-susceptible species, such as C. glabrata, has increased.
Many risk factors for invasive candidosis have been identified. These can be divided into host-related and healthcare-related factors:
Among adult patients cared for in surgical intensive care units, Candida bloodstream infection has a case fatality rate of about 40%.
Clinical features
Infection may be localized, for instance in the urinary tract, liver, heart or meninges, or may be widely disseminated and associated with a septicaemia (candidaemia). Invasive candidosis is difficult to diagnose and treat, and for some forms the prognosis is poor.
Disseminated infection is most commonly seen in seriously ill individuals who usually have one or more indwelling vascular catheters, although these are not necessarily the source of the infection. Many cases are thought to arise from translocation of organisms across the wall of the intestinal tract.
Adults with candidaemia usually present with a persistent fever that fails to respond to broad-spectrum antibiotics, but with few other symptoms or clinical signs. One sign of invasive candidosis is the presence of white lesions within the eye (Candida endophthalmitis) (Fig. 61.18). These are found in up to 45% of patients in the intensive care unit, but are seldom seen in neutropenic individuals. Other useful signs are the nodular cutaneous lesions that occur in about 10% of neutropenic individuals with disseminated Candida infection. Other manifestations include:
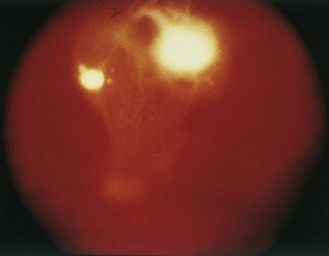
Fig. 61.18 Fundoscopic appearance of Candida endophthalmitis.
(Reproduced with permission from Richardson MD, Warnock DW, Campbell CK 1995 Slide Atlas of Fungal Infection: Systemic Fungal Infections. Blackwell Science, Oxford.)
Invasive candidosis is a common complication in infants of low birth-weight (<1000 g) requiring prolonged neonatal intensive care. Meningitis occurs more frequently than in older patients and is sometimes associated with arthritis and osteomyelitis.
Laboratory diagnosis
Candida species may be present as commensals in the absence of infection, so that isolation from clinical material, except from sites that are normally sterile, is of little significance. Similarly, antibodies to Candida species can be detected in uninfected individuals because of their exposure to commensal yeasts, although a rise in antibody titre or high titres may be of diagnostic significance. In suspected invasive candidosis, samples from as many sources as possible should be examined by direct microscopy and culture. Results should always be interpreted in the light of clinical findings.
Direct microscopy
Appropriate samples are examined microscopically in potassium hydroxide or after Gram staining. In tissue sections, the fungus is seen best in stained preparations. Hyphae are often abundant, but their presence in sputum or urine does not confirm that the yeast is present as a pathogen.
Culture
Candida species grow readily in culture at 37°C on common isolation media, such as Sabouraud dextrose agar. Blood cultures provide the most reliable evidence of invasive infection, although repeated attempts to isolate the organism may be required.
Isolation of the yeast from otherwise sterile sites provides reliable evidence for the diagnosis, but cultures obtained from urine, faeces and sputum are of less value unless done quantitatively over a period of time. Cell counts of the yeast in urine in excess of 104 per mL are usually taken to indicate urinary tract infection, except in those with an indwelling urinary catheter. As Candida species multiply rapidly in clinical material it is important that specimens are processed as soon as possible after collection.
Serological tests
Currently available tests lack specificity and sensitivity, and the results must be interpreted with care. A positive test does not necessarily indicate infection because the antigens used do not differentiate between antibodies formed during mucosal colonization and those produced during deep infection. Similarly, a negative antibody test does not necessarily rule out the possibility of invasive candidosis in immunocompromised patients who are incapable of mounting an adequate antibody response.
Antigen tests, based mainly on ELISA or latex agglutination, that detect cell wall mannan or cytoplasmic components have been developed and are used for diagnosis. Nucleic acid detection methods are increasingly being used to diagnose invasive candidosis, although their place in diagnosis is still being evaluated.
Treatment
The treatments of choice for most forms of invasive candidosis are:
• intravenous echinocandin (anidulafungin, caspofungin or micafungin)
Amphotericin B can be used in combination with flucytosine, but flucytosine is not used alone as resistance may develop during treatment. Voriconazole has also been used successfully. Removal of existing intravenous catheters is desirable if feasible, especially in non-neutropenic patients.
The choice of antifungal treatment depends on both the clinical status of the patient and the species of infecting organism.
Cryptococcosis
Cryptococcosis, caused by the encapsulated yeasts Cryptococcus neoformans and C. gattii, is most frequently recognized as a disease of the central nervous system, although the primary site of infection is the lungs. The disease occurs sporadically throughout the world but it is currently seen most often in persons with AIDS.
There are four serotypes of Cryptococcus (A–D) that represent two distinct species of the organism, namely, C. neoformans (A & D) and C. gattii (B & C). Most infections are caused by C. neoformans, which is commonly found in the droppings of wild and domesticated birds throughout the world. Pigeons carry C. neoformans in their crops, but do not appear to become infected, probably because of their high body temperature. C. gattii has been isolated from decaying wood in various species of trees including Eucalyptus. These trees are indigenous to Australia, but have been planted in numerous other countries. C. gattii infection has been reported from subtropical regions and from temperate parts of the world, including western Canada and the Pacific North West Region of the USA.
Epidemiology
Infection is acquired by inhalation; the incubation period is unknown. The likelihood that an infection with C. neoformans will develop after inhalation depends largely on host factors. Even before the advent of AIDS, infections with C. neoformans tended to occur in individuals with abnormalities of T lymphocyte function, such as are found in persons with lymphoma, and those receiving corticosteroid therapy. The major risk factor for development of infection with C. gattii appears to be environmental exposure, although there is indirect evidence that unidentified host factors contribute to the higher incidence of disease in Australian Aboriginals.
With the advent of the AIDS epidemic, cryptococcosis became the most common cause of meningitis in hospitals in which persons with HIV infection are treated. Although the incidence of the disease has declined in developed countries where combination antiretroviral treatment is available, the incidence is rising in many developing countries afflicted with large epidemics of HIV infection.
Clinical features
A mild self-limiting pulmonary infection is believed to be the most common form of cryptococcosis. In symptomatic pulmonary infection there are no clear diagnostic features. Lesions may take the form of small discrete nodules, which may heal with a residual scar or may become enlarged, encapsulated and chronic (cryptococcoma form). An acute pneumonic type of disease has also been described.
The meningeal form of cryptococcosis can occur in apparently healthy individuals, but occurs most frequently in immunocompromised persons. Chronic meningitis or meningo-encephalitis develops insidiously with headaches and low-grade fever, followed by changes in mental state, visual disturbances and eventually coma. The disease may last from a few months to several years, but the outcome is always fatal unless it is treated. Patients with AIDS and cryptococcosis generally develop a chronic meningeal form with milder symptoms.
Although predominantly a disease of the central nervous system, lesions of the skin, bones and other deep sites may also occur; in its disseminated form, the disease may resemble tuberculosis. Rarely, lesions of skin and bones may occur without any evidence of infection elsewhere.
Laboratory diagnosis
Cryptococcus is readily demonstrated in cerebrospinal fluid (CSF) or other material by direct microscopy, culture or serological tests for capsular antigen. The yeast load is generally higher in patients with AIDS. The cellular reaction and chemical changes in CSF usually resemble those seen in tuberculous meningitis. The yeast cells of Cryptococcus are round, 4–10 µm in diameter and surrounded by a mucopolysaccharide capsule. The width of the capsule varies and is greatest in vivo and on rich media in vitro.
In unstained wet preparations of CSF mixed with a drop of Indian ink or nigrosine, the capsule can be seen as a clear halo around the yeast cells (Fig. 61.19). Capsulate yeasts are seen in the CSF of about 60% of patients with cryptococcosis (higher in AIDS), but the capsule may be difficult to visualize in some cases. Sputum, pus or brain tissue should be examined after digestion in potassium hydroxide and here the capsulate yeasts are often delineated by the cellular debris. For examination of tissue sections it is best to use a specific fungal stain such as periodic acid–Schiff; alcian blue and mucicarmine stain the capsular material, enabling the organisms to be differentiated from H. capsulatum and B. dermatitidis.
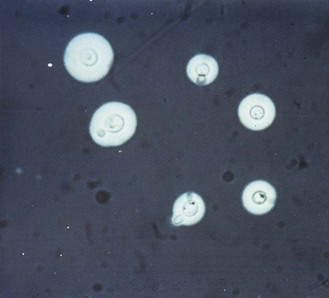
Fig. 61.19 Microscopic appearance of encapsulated Cryptococcus cells in an Indian ink preparation of CSF.
(Reproduced with permission from Richardson MD, Warnock DW, Campbell CK 1995 Slide Atlas of Fungal Infection: Systemic Fungal Infections. Blackwell Science, Oxford.)
The yeast is easily cultured from CSF although large volumes or multiple samples may be required in some cases; in patients with AIDS it is also useful to culture blood. On Sabouraud agar (without cycloheximide) cultured at 25–30°C and 37°C, colonies normally appear within 2–3 days, but cultures should not be discarded for 3 weeks. In culture, Cryptococcus appears as creamy white to yellow–brown colonies, which are mucoid in strains with well developed capsules and dry in strains that lack prominent capsules. Buds appear at any point on the cell surface but hyphae or pseudohyphae are not normally produced. Preliminary identification depends on demonstration of the capsule but this may be absent or difficult to see. Cryptococcus can be identified with commercial kits or distinguished from other yeasts by its lack of fermentative ability, its ability to produce urease, to grow at 37°C and to assimilate inositol.
The latex agglutination test for the detection of cryptococcal polysaccharide antigen in CSF or blood is highly sensitive and specific for the diagnosis of cryptococcal meningitis and disseminated forms of the disease, and gives better results than microscopy and culture. In AIDS, the test is positive in well over 90% of infected patients; titres of over 106 may be detected.
Treatment
Intravenous amphotericin B in combination with flucytosine is usually the treatment of choice for individuals with meningeal or disseminated cryptococcosis. Oral fluconazole is widely used for the continuation of treatment in those who have responded to amphotericin B. Patients with AIDS commonly relapse after the initial course of therapy and many require lifelong maintenance treatment with fluconazole.
Mucormycosis
Mucormycosis, formerly known as zygomycosis, is a relatively rare, opportunistic infection caused by saprophytic mould fungi, notably species of Rhizopus and Lichtheimia (Absidia). These moulds are ubiquitous in the soil and on decomposing organic matter. They are characterized by having broad, aseptate hyphae, with large numbers of asexual spores inside a sporangium which develops at the end of an aerial hypha.
Epidemiology
Most infections follow inhalation of spores; less frequently, infection follows traumatic inoculation into the skin and soft tissue. Major risk factors include:
• prolonged or profound neutropenia
• uncontrolled diabetes mellitus
Certain predisposing conditions seem to be more commonly associated with particular clinical forms of disease; for example, persons with diabetic ketoacidosis often develop rhinocerebral mucormycosis, whereas neutropenic individuals often develop pulmonary or disseminated disease.
Clinical features
The best known form of the disease is rhinocerebral mucormycosis. There is rapid and extensive tissue destruction, most commonly spreading from the nasal mucosa to the turbinate bone, paranasal sinuses, orbit and brain (Fig. 61.20). The condition is fatal if untreated and, although the prognosis has improved over recent years, many diagnoses are still made at necropsy.
Fig. 61.20 Necrotic palatal lesion in a case of rhinocerebral mucormycosis.
(Reproduced with permission from Richardson MD, Warnock DW, Campbell CK 1995 Slide Atlas of Fungal Infection: Systemic Fungal Infections. Blackwell Science, Oxford.)
Pulmonary and disseminated infections can occur in severely immunocompromised individuals. Primary cutaneous infections have also been reported; these are uncommon but often result in extensive necrotizing fasciitis or disseminated disease. They usually occur in patients with burns or other forms of local trauma.
Laboratory diagnosis
Recognition of the fungus in tissue by microscopy is considerably more reliable than culture, but material such as nasal discharge or sputum seldom contains much fungal material and examination of a biopsy is usually necessary for a firm diagnosis. Direct examination of curetted or biopsy material in potassium hydroxide may reveal the characteristic broad, aseptate, branched and sometimes distorted hyphae. However, they are seen much more clearly when stained with methenamine–silver; the hyphae of these fungi do not stain with periodic acid–Schiff (Fig. 61.21).
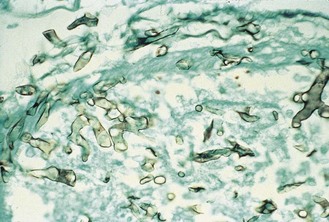
Fig. 61.21 Microscopic appearance of mucormycosis.
(Reproduced with permission from Richardson MD, Warnock DW, Campbell CK 1995 Slide Atlas of Fungal Infection: Systemic Fungal Infections. Blackwell Science, Oxford.)
The fungi are readily isolated on Sabouraud dextrose agar at 37°C, but isolation is of little diagnostic significance in the absence of strong supporting clinical evidence of infection. There are no established serological tests.
Pneumocystosis
Pneumocystis is an opportunistic pathogen with some of the features of protozoa, but comparative DNA sequence analysis showed it to be more closely related to the fungi. The organism was originally described as a cause of atypical pneumonia in malnourished infants, but came to prominence in the 1980s as a common cause of pneumonia, which was commonly fatal, in patients with AIDS. With the advent of reliable antiretroviral therapy, the incidence of the disease in HIV-positive individuals has declined.
Based on its morphology, which is similar to that of protozoa, the life cycle of Pneumocystis is divided into three stages:
1. the cyst, a spherical or crescent-shaped form (5–7 µm in diameter)
2. the sporozoite, up to eight of which develop within each cyst
When openings appear in the cyst wall, the excysted sporozoites become trophozoites. All of these forms reside within the alveoli of the lungs. As the organism is not cultivatable in vitro, its life cycle has not been fully elucidated.
The finding that Pneumocystis organisms from different mammalian hosts are genetically quite dissimilar has led to a name change from P. carinii (which infects rats) to P. jirovecii for the organisms that infect man.
Epidemiology
Infection is probably acquired by inhalation. Serological and PCR studies indicate that most human beings become subclinically infected with Pneumocystis during childhood and that this infection is usually well contained by an intact immune system. The occurrence of clinical disease is related to the extent of immunosuppression, especially impairment of cell-mediated immunity. It may be due to primary infection, re-infection or reactivation. Pneumocystosis has a global distribution.
Clinical features
The clinical presentation of Pneumocystis pneumonia is non-specific. Symptoms include:
Patients with AIDS have a more indolent course with longer duration of symptoms than patients receiving immunosuppressive drugs. Without treatment the course is progressive and usually ends in death.
In addition to pneumonia, Pneumocystis infection may disseminate to the lymph nodes, liver, spleen, bone marrow, adrenal gland, intestines and meninges. Extra-pulmonary disease occurs predominantly in patients with advanced HIV infection and in those not taking co-trimoxazole prophylaxis or receiving aerosolized pentamidine.
Laboratory diagnosis
Diagnosis usually depends on the identification of typical octonucleate cysts or trophozoites in tissues or body fluids. Organisms are detected by immunofluorescent staining of broncho-alveolar lavage fluid or induced sputum smears. Molecular diagnosis by PCR methods is being introduced in some centres.
Treatment
Co-trimoxazole and intravenous pentamidine are the most effective therapies. The former is as potent as the latter, but less toxic. Other regimens include atovaquone, trimetrexate, the combination of trimethoprim and dapsone, and the combination of clindamycin and primaquine.
Co-trimoxazole is the preferred prophylactic agent, but patients with AIDS or those undergoing solid-organ or bone marrow transplantion may suffer unacceptable side effects to the high doses used. Aerosolized pentamidine is also used for prophylaxis.
Other opportunist fungi
Penicillium marneffei causes serious disseminated disease with characteristic papular skin lesions in patients with AIDS in South-East Asia. The fungus is dimorphic, forming yeast-like cells that are often intracellular, resembling histoplasmosis, in infected tissues. Treatment is with amphotericin B, followed by itraconazole to prevent relapse.
Almost any fungus may invade a severely immunocompromised host, and infections with many common fungi, including Fusarium species, Trichosporon asahii and Pseudallescheria boydii, have been reported. Diagnosis is made by culture of the causative organism from clinical specimens and serological tests play little part. Tissue sections are often not very helpful, either because the causal fungi have no special features to enable identification or because they resemble other fungal pathogens.
Infections are usually treated speculatively, and sometimes successfully, with amphotericin B.
Anaissie EJ, McGinnis MR, Pfaller MA. Clinical Mycology, ed 2, New York: Elsevier, 2009.
Dismukes WE, Kauffman C, Pappas PG, Sobel JD. Clinical Mycology, ed 2, New York: Springer, 2010.
Hospenthal DR, Rinaldi MG. Diagnosis and Treatment of Human Mycoses. Totowa, New Jersey: Humana Press, 2008.
Richardson MD, Johnson EM. Pocket Guide to Fungal Infection, ed 2. Oxford: Wiley Blackwell; 2006.
Richardson MD, Warnock DW. Fungal Infection: Diagnosis and Management, ed 3. Oxford: Blackwell Science; 2003.
The Aspergillus Website. http://www.aspergillus.org.uk/.
Doctor Fungus. http://www.doctorfungus.org/.
University of Adelaide. Mycology Online. http://www.mycology.adelaide.edu.au/.
World of Dermatophytes. A Pictorial. http://www.provlab.ab.ca/mycol/tutorials/derm/dermhome.htm.