69 Hospital infection
Key points
• Hospitals constitute a special environment where the epidemiology of infection is distinct. The chief contributing factors are the accumulation of patients with particular features, the special activities undertaken in hospitals, and the special environment created by the patients and the activities.
• Patients may constitute a special hazard because they are infectious, or they may be unusually susceptible to infection because they have particular conditions or are receiving immunosuppressive treatments.
• Special activities include surgery and extensive use of intravascular devices. Needlestick injuries are a constant hazard.
• The extensive use of antibiotics and disinfectants, and the need to reuse equipment and areas that may become contaminated, contribute to the special environment.
• Certain organisms such as methicillin-resistant Staphylococcus aureus (MRSA), Pseudomonas aeruginosa and Clostridium difficile are important agents of hospital-acquired infection owing to the factors outlined above.
• Careful and detailed attention must be paid to controlling the routes of transmission of infection, through the establishment and maintenance of an infection control policy.
The battle between man and microbe is at its most obvious in institutions where vulnerable people are crowded together. Historically, hospitals have a notorious reputation for infection. The hazards of puerperal sepsis and the horrors of septic infection in the pre-Listerian era have been well documented; admission to hospital in the mid-nineteenth century was associated with the fear of gangrene and death.
Since then, surgical and medical techniques have developed dramatically, basic standards of building and hygiene have greatly improved, and the identification and treatment of most infecting micro-organisms have become possible. Despite such changes, infection acquired in hospitals remains a major cause of morbidity and mortality, leading directly or indirectly to an enormous increase in the cost of hospital care and to the emergence of new health hazards for the community. In the past two decades enormous advances in biomedical technology and therapeutics have produced greater numbers of highly susceptible patients requiring treatment in hospitals, and this is aggravated by the occurrence of transferable resistance to antibiotics in pathogenic bacteria and the emergence of new pathogens transmitted by a variety of routes. In spite of these advances in medical care, in many countries pressures on health care facilities and shortages of trained staff make it difficult to practise adequate infection control. There has also been a mistaken view among many health professionals that the advent of the antibiotic era made such precautions unnecessary, and many studies have shown poor compliance with simple hygiene.
Classification
To measure the extent of hospital and healthcare-associated infection (HAI) and conduct surveillance the following definitions should be considered:
1. Community-acquired infections: either those contracted and developing outside hospitals which require admission of the patient (e.g. pneumococcal pneumonia) or those contracted outside hospital or those which become clinically apparent within 48 hours of admission or when the patient has been admitted to hospital for other reasons (e.g. chickenpox or zoster).
2. Infections contracted and developing within hospital (e.g. device-associated bacteraemias).
3. Infections contracted in hospital but not becoming clinically apparent until after the patient has been discharged (e.g. many postoperative wound infections).
4. Infections contracted by healthcare staff as a consequence of their work, whether or not this involves direct contact with patients (e.g. hepatitis B).
On average, around 9% of all hospitalized patients will develop an infection as a result of their stay in hospital. Urinary, respiratory and wound infections are the most common.
Factors that influence infection
Hospital infection, also known as nosocomial infection, may be exogenous or endogenous in origin. The exogenous source may be another person in the hospital (cross-infection) or a contaminated item of equipment or building service (environmental infection). A high proportion of clinically apparent hospital infections are endogenous (self-infection), the infecting organism being derived from the patient’s own skin, gastrointestinal or upper respiratory flora.
Most infections acquired in hospital are caused by micro-organisms that are commonly present as commensals in the general population. Thus, contact with micro-organisms is seldom the sole or main event predisposing to infection. Various risk factors, alone or in combination, influence the frequency and nature of hospital infection. In comparing rates of HAI it is important to be aware of the frequencies of risk factors such as age, drug treatment or preexisting diseases in the population surveyed as well as the medical or surgical procedures used.
Contact with other patients and staff
In common with any large institution or workplace, the patients and staff of a hospital share many facilities in close or crowded conditions. Outbreaks of diarrhoeal and food-borne disease may be traced to a common source via the hospital water or food supplies. The specific role of hospitals in admitting infected patients or carriers for treatment clearly serves as a potential source of infection for others. Patients with comparable susceptibility to infection tend to be concentrated in the same area, e.g. in neonatal units, burns units or urological wards, where infected and non-infected patients may be cared for by the same staff, thus creating numerous opportunities for the spread of micro-organisms by direct contact. The more susceptible patients usually require the most intensive care with far more daily contacts with staff who act as vectors in the transmission of microbes like insects spreading parasites.
Inanimate reservoirs of infection
Equipment and materials in use in hospitals often become contaminated with micro-organisms which may subsequently be transferred to susceptible body sites on patients. Gram-positive cocci, derived from skin scales of the hospital population, are found in the air, dust and on surfaces where they may survive along with fungal and bacterial spores of environmental origin. Gram-negative aerobic bacilli are common in moist situations and in fluids, where they often survive for long periods, and may even multiply in the presence of minimal nutrients. An important example of this is legionellae in hospital domestic water supplies. Awareness of the common reservoirs of environmental and contaminating hospital micro-organisms provides the basis for maintaining standards of hygiene (cleaning, disinfection, sterilization) throughout the hospital as well as good engineering and building.
Role of antibiotic treatment
At least 30% of patients receive antibiotics during their stay in hospital, and this exerts strong selective pressures on the microbial flora, especially of the gastrointestinal tract, leading to the development of antibiotic-associated diarrhoea due to Clostridium difficile (see Ch. 22), one of the commonest causes of outbreaks of hospital infection. Sensitive species or strains of micro-organisms which normally maintain a protective function on the skin and other mucosal surfaces tend to be eliminated, whereas those that are more resistant survive and become endemic in the hospital population. This may restrict the range of agents available for treatment and may lead to the transmission of plasmid-mediated antibiotic resistance into strains that show increased virulence, survival and spread within the hospital.
Micro-organisms causing hospital infection
The most important micro-organisms responsible for hospital infection are listed in Table 69.1.
Table 69.1 Commonly occurring micro-organisms in hospital infection
| Type of infection | Micro-organisms responsible |
|---|---|
| Urinary tract infections | |
| Respiratory infections | |
| Wounds and skin sepsis | |
| Gastro-intestinal infections |
Outbreaks of Staphylococcus aureus infection were often seen in surgical wards and maternity units before the advent of penicillinase-stable penicillins such as methicillin in the early 1960s. Some strains, such as the notorious phage type 80/81, demonstrated particular virulence and colonizing capabilities. Subsequently, epidemic or pandemic strains characterized by resistance to methicillin (MRSA) have been found in many hospitals worldwide, presenting a daunting challenge. Some strains are better able to colonize patients or staff than to produce systemic disease. This illustrates the adaptation and evolutionary changes possible within common hospital bacteria revealed by careful epidemiological typing and observation. The changing patterns may be related to particular events and infection control measures, so that lessons can be learnt for future control and prevention.
With the advent of more elaborate surgery and intensive care, combined with the use of broad-spectrum antibiotics and immunosuppressive drugs, Gram-negative bacteria increased in importance. Many, such as Pseudomonas aeruginosa, are opportunists capable of causing infection in compromised patients. Such organisms may be found in the patient’s own flora, or in damp environmental sites, including patient equipment and medicaments. They may exhibit natural resistance to many antibiotics and antiseptics, and have the ability to colonize traumatized skin such as burns and areas with poor tissue viability such as decubitus ulcers and bed sores.
In recent years, groups of micro-organisms which formerly played no recognized part in hospital infection have emerged. These include the coagulase-negative staphylococci and Acinetobacter baumanii present in normal skin flora. Viral or fungal infection, particularly of the immunocompromised patient, has become more important. Legionella pneumophila, disseminated from environmental sources such as air conditioning and hot water systems, causes sporadic cases or outbreaks of respiratory infection in hospitals. Awareness of the risks of blood-borne viruses, including hepatitis B and C, human immunodeficiency virus (HIV) and of many agents such as cytomegalovirus, which can be transmitted by organ or cellular transplants, has increased in patients and in staff. The possible risk of iatrogenic spread of the prion causing Creutzfeldt–Jakob disease (see Ch. 60) has persuaded the UK government to take stringent measures on decontamination of surgical instruments and the US blood transfusion service to ban British donors.
Routes of transmission
The hospital offers many opportunities for the exchange of microbes, many of which are harmless and a normal part of the balance between man and his environment. For there to be a significant risk of infection, several factors, including the right susceptible host and the appropriate inoculum of infecting micro-organism, must be linked via an appropriate route of transmission. Understanding of the sources and spread of hospital infection enables efforts to be concentrated in more effective preventive measures.
Common routes of transmission for different micro-organisms are shown in Table 69.2.
Table 69.2 Hospital infection: sources and spread
| Route | Source | Examples of disease |
|---|---|---|
| 1. Aerial (from persons) | Mouth | Measles, tuberculosis, pneumonia |
| Droplets | Nose | Staphylococcal sepsis |
| Skin scales | Skin exudate, infected lesion | Staphylococcal and streptococcal sepsis |
| 2. Aerial (from inanimate sources) | Respiratory equipment | Gram-negative respiratory infection |
| Particles | Air-conditioning plant | Legionnaires’ disease, fungal infections |
| 3. Contact (from persons) | ||
| Direct spread | Respiratory secretions | Staphylococcal and streptococcal sepsis |
| Indirect via equipment | Faeces, urine, skin and wound exudate | Enterobacterial and viral diarrhoea, Pseudomonas aeruginosa sepsis |
| 4. Contact (environmental source) | Equipment, food, medicaments, fluids | Enterobacterial sepsis (Klebsiella/Serratia/ Enterobacter spp.) Ps. aeruginosa and other pseudomonads |
| 5. Inoculation | Sharp injury, blood products | Hepatitis B, HIV, malaria |
Airborne transmission
• By airborne transmission from the respiratory tract (talking, coughing, sneezing).
• From the skin by natural shedding of skin scales during wound dressing or bed-making.
• By aerosols from equipment such as respiratory apparatus and air-conditioning plants.
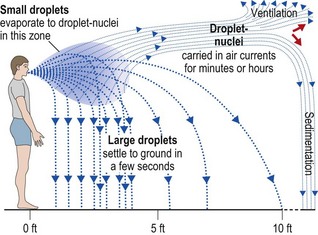
Infectious agents may be dispersed as small particles or droplets over long distances (Fig. 69.1). Staphylococci survive well on mucosal secretions, skin scales and dried pus and may be redistributed in the air after initial settlement during periods of increased activity (Fig. 69.2). Gram-negative bacilli do not generally survive desiccation in air, and this route of transmission is therefore limited to conditions of high humidity such as ventilatory equipment, showers or other fine water aerosols.
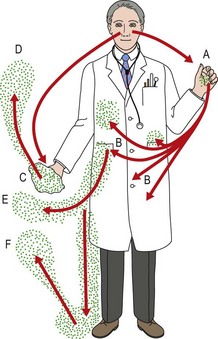
Fig. 69.2 Infection of the air with dust particles derived from nasal and oral secretions contaminating hands, handkerchief, clothing and surrounding surfaces: (A) hand soiled with secretions from lips or nose-picking; (B) clothing contaminated by hand; (C) soiled handkerchief; (D) infected dust from handkerchief; (E) dust from clothing (e.g. from near handkerchief pocket); (F) infected dust raised after settling on floor.
Contact spread
The most common routes of transmission for hospital infection are:
• By direct contact spread from person to person.
• By indirect contact spread via contaminated hands or equipment.
Human secretions as well as contaminated dust particles or fluids may be carried on thermometers, bedpans, bed-linen, cutlery or other shared items. Hands and, to a lesser extent, clothing of hospital staff serve as vectors of Gram-negative and Gram-positive infection. Procedures involving contact with mucosal surfaces, e.g. insertion of a urinary catheter, may introduce micro-organisms from the contaminated hands of the operator or from the patient’s own urethral flora into the normally sterile bladder.
Food-borne spread
Infection may originate in the hospital kitchen, or in special diets, infant feeds, kitchen or commercial supplies. Deteriorating hygiene standards may support the proliferation of flies, cockroaches and other insects or rodents which damage stored products and act as carriers of microbes.
Blood-borne spread
The accidental transmission of infections such as HIV, hepatitis B or C by needlestick or contaminated ‘sharp’ injuries has been well documented. In areas of high prevalence of these blood-borne viruses, malaria or syphilis stringent precautions should be taken to minimize transmission between patients and from healthcare workers by strict use of single-use items and screening blood products.
Self-infection and cross-infection
The interaction between different sources of infection may be illustrated by the example of a patient undergoing lower bowel surgery. Self-infection may occur due to transfer into the wound of staphylococci (or occasionally streptococci) carried in the patient’s nose and distributed over the skin, or of coliform bacilli and anaerobes released from the bowel during surgery. Alternatively, cross-infection may result from staphylococci or coliform bacilli derived from other patients or healthy staff carriers. The organisms may be transferred into the wound during operation through the surgeon’s punctured gloves or moistened gown, on imperfectly sterilized surgical instruments and materials, or by airborne theatre dust. Postoperatively, organisms may be transferred in the ward from contaminated bed-linen, by airborne ward dust or in consequence of a faulty wound dressing technique.
Of all the possible routes, by far the most likely in this example is self-infection from the patient’s own bowel flora and it is therefore against this route that most specific preventive measures in colorectal surgery are directed. Understanding of possible sources of infection and the methods available to block transmission to susceptible sites forms the basis of hospital infection control.
Cross-infection is more often caused by ‘hospital’ strains selected for characteristics of antimicrobial resistance and virulence. An important example at present is MRSA, which can be easily identified by the microbiology laboratory, which should have a system for alerting the infection control team who need to collect the following basic epidemiological data:
• the site and extent of infection
• the dates of admission, operative procedures, first recognition of infection
The clustering of cases according to a common surgical team or location in the ward may suggest a common source and may be the first firm indication of an outbreak of hospital infection.
Soon after admission to hospital, individuals commonly become contaminated with the ‘hospital flora’. This has been shown with Staph. aureus in studies of patients before and during hospital treatment. Those patients who need to stay longer in hospital, e.g. those requiring intensive care or the elderly, are more susceptible to infection and the risks of HAI are greater.
Prevention and control
Infection control policy
The establishment of an effective infection control organization is the responsibility of good management of any hospital. There will normally be two parts:
1. An infection control committee, meeting regularly to formulate and update policies for the whole hospital on matters having implications for infection control, and to manage outbreaks of nosocomial infection.
2. An infection control team of workers, headed by the infection control doctor (usually the microbiologist), to take day-to-day responsibility for this policy.
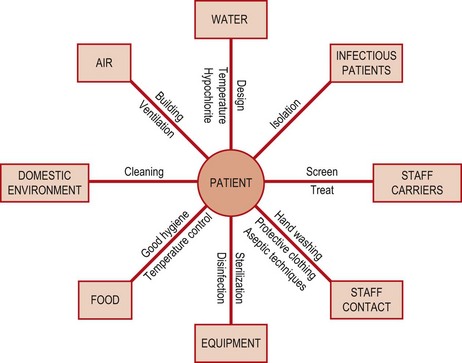
The functions of this team include surveillance and control of infection and monitoring of hygiene practices, advising the infection control committee on matters of policy relating to the prevention of infection and the education of all staff in the microbiologically safe performance of procedures. The infection control nurse is a key member of this team. Close working links between the microbiology laboratory, infection control nurse and the different clinical specialties and support services (including sterile services, laundry, pharmacy and engineering) are important to establish and maintain the infection control policy, and to ensure that it is rationally based and that the recommended procedures are practicable. It is important for all members of the committee to ensure that everyone in the organization makes infection control and hospital hygiene their business. Campaigns should be launched locally raise the profile of proven methods of control such as hand washing. Some of the control measures in which the infection control team should be involved are shown in Figure 69.3.
Decontamination and sterilization
The provision of sterile instruments, dressings and fluids is of fundamental importance in hospital practice. Items that are reused for procedures in normally sterile sites, such as surgical equipment, must be decontaminated by thorough washing before sterilization by heat. High-vacuum autoclaves have become accepted hospital practice. The development of these sterilizers for processing wrapped goods facilitated the provision of a centralized service of sterile supply to wards, complementing the existing theatre service. The availability of a wide range of prepacked single-use items (syringes, needles, catheters and drainage bags) sterilized commercially by γ-irradiation or ethylene oxide has further improved aseptic procedures and removed the need for reprocessing items that are difficult to clean and therefore impossible to sterilize. The scientific basis of sterilization is mentioned in more detail in Chapter 4.
Most fluids for topical use or intravenous administration are now prepared commercially or in regional units where standards of quality control and efficiency for bulk processes are more readily achieved than in individual hospital pharmacies.
Aseptic techniques
The provision of sterile equipment will not prevent the spread of infection if there is carelessness in its use. Wherever possible, no-touch techniques must be used, coupled with strict personal hygiene on the part of the operator. These routines are rigidly laid down in operating theatre practice and may be modified as required for other procedures such as wound dressing and insertion of intravenous catheters.
Cleaning and disinfection
The general hospital environment can be kept in good order by attention to basic cleaning, waste disposal and laundry. The use of chemical disinfectants for walls, floors and furniture is necessary only in special instances, such as spillages of body fluids from patients with blood-borne virus infections. Ward equipment such as bed-pan washer/disinfectors and dishwashers should be monitored to ensure reliable performance, and cleaning materials such as mop-heads and cloths should be heat-disinfected and stored dry after use. Pre-cleaning of contaminated instruments and equipment, preferably by means of an automatic washing process with an ultrasonicator, is an essential step before disinfection or sterilization.
Skin disinfection and antiseptics
The ease of acquisition and transfer of transient hospital contaminants, particularly staphylococci and Gram-negative bacilli on the hands of staff, is an important factor in the spread of hospital infection. Thorough handwashing after any procedure involving nursing care or close contact with the patient is essential. Alcohol-based hand antiseptics or ‘rubs’ have been introduced in wards where routine handwashing with water and detergent is not practicable. Gloves may be worn for many dirty contact procedures, such as emptying a urinary drainage bag or bedpan, although it should not be forgotten that the gloved hand may also become colonized by transient hospital flora.
Procedures for preoperative disinfection of the patient’s skin and for surgical scrubs are mandatory within the operating theatre. Dilute ‘in-use’ solutions of antiseptics may readily become colonized with Gram-negative bacteria and should be replaced regularly. Ideally, single-use preparations should be used. Restriction should be placed on the indiscriminate use of antiseptics and disinfectants by means of a disinfectant policy agreed by pharmacists, microbiologists and key users, such as theatre staff.
Decontamination and disinfectant policy
All hospitals should agree a policy to ensure all clinical staff are familiar with the agents used and procedures involved in decontamination. The policy should consider the following:
• The sources (equipment, skin and environment) for which the choice of process is required.
• The processes and products available for sterilization and disinfection. One objective of the policy is to include a limited range of options and chemicals to be used.
• The category of process required for each item in a simple table: sterilization for surgical instruments, heat disinfection for laundry and crockery, cleaning for floors and furniture.
• The specific products and method to be used for each item and staff responsible.
Effective implementation of the policy requires liaison and training of staff and regular updating as new methods and agents become available. Safety consideration for staff and patients require that a risk assessment is made of procedures and chemicals to minimize hazards and comply with local health and safety regulations. This includes the use of fume cupboards, air scavenging equipment and sealed washer-disinfectors. These requirements take decontamination of equipment out of small clinics into centralized sterile supply departments and increase the use of disposables in medical practice.
Prophylactic antibiotics
Widespread and haphazard use of antibiotics hastens the emergence of antibiotic-resistant bacteria and increases both the incidence of toxic side-effects and the cost of treatment. However, rational antibiotic prophylaxis plays an important role in infection control. Specific indications include perioperative prophylaxis in gastrointestinal and gynaecological surgery directed predominantly against anaerobic infection and for patients known to have bacteriuria at the time of urological surgery or instrumentation, directed against the urine isolate. An antibiotic policy which limits the choice of broad-spectrum agents is important both for prophylaxis and treatment (see Ch. 67).
Protective clothing
Different activities within the hospital require different degrees of protection to staff and patients. In operating theatres the wearing of sterile gowns, gloves, head gear and face masks minimizes the shedding of micro-organisms. The properties of fabrics available for theatre use have improved, and now include close-weave ventile fabrics that are comfortable to wear and allow evaporation of moisture. ‘Total protection’ of the operating site may be considered for certain high-risk clean surgery such as hip replacements, during which the surgical team may wear exhaust-ventilated suits and operate under conditions of ultraclean laminar airflow.
For many ward procedures in which there may be soiling, or for simple barrier nursing of patients with communicable diseases, plastic aprons and gloves are used. Gloves, face masks and goggles are also indicated for specific procedures when blood contact is likely through splashes or aerosols, such as dental procedures. These should conform to international standards and staff should be trained in their use and disposal.
Isolation
The isolation policy should list facilities and procedures needed to prevent the spread of specific infections to other patients (source isolation) and to protect susceptible or immunocompromised patients (protective isolation). Effective isolation demands a highly disciplined approach by all staff to ensure that none of the barriers to transmission (airborne, direct and indirect contact) are breached. Multi-bedded rooms may be used, and even wards converted during hospital outbreaks, but the simplest solution wherever possible is to use single rooms.
‘Cubicle’ isolation, by which the patient is nursed alone in a room separated by a door and corridor from other patients, confers a substantial measure of protection. Preferably, each isolation room has its own toilet and washing facility. Clean, filtered air is supplied to the room, which should be at negative pressure (exhaust-ventilated) to the corridor for source isolation or at positive pressure (pressure-ventilated) to the corridor for protective isolation. If, however, there is a small airlock vestibule separating the room from the outer corridor, then exhaust ventilation of the airlock will give effective isolation for either situation. The vestibule or lobby should contain a wash basin and include space for gowning and equipment.
In some critical situations such as bone marrow transplant units, where airborne contamination with environmental fungal spores is a problem, the efficiency of air filtration may be increased and laminar airflow maintained as a barrier around the patient. Stringent isolation in a plastic tent isolator is required only for patients with highly contagious infections and such contingencies usually require transfer to a specialist hospital.
Hospital building and design
The routine maintenance of the hospital building is important, ensuring that surfaces wherever possible are smooth, impervious and easy to clean. Major rebuilding works on or near the hospital site may generate dust containing fungal and bacterial spores, with implications for specialized units serving immunocompromised patients. Close communications with the works department and hospital administration are necessary to coordinate any protective action. When a new hospital or modification of existing building is planned, the infection control team should be closely involved in discussing the plans. In many countries, guidance on new building design exists to minimize potential hospital-acquired infection. Areas requiring special attention include operating theatres, kitchens, acute wards, laboratories and air-conditioning systems. The risk of Legionnaires’ disease is reduced by regular flushing all outlets and installing water supplies that circulate below 20°C for the cold and above 60°C for the hot circuit.
Equipment
Any object or item of equipment for clinical use should be assessed to determine the appropriate method, frequency and site of decontamination. Wherever possible, heat processes are preferred, although this may be precluded for certain thermolabile items such as fibreoptic endoscopes. Dedicated washer-disinfectors in sealed units are necessary for these items. In many situations, especially in community settings, disposable instruments should be used, although monetary and environmental costs need to be balanced against availability and transport of the item.
Personnel
An occupational health service should screen staff before employment for serious communicable diseases, such as tuberculosis, and check their immunization status. Hepatitis B vaccine should be given to all health care workers if they have not been fully immunized. Those at special risk performing exposure-prone procedures, such as invasive surgery, should be screened for blood-borne viruses. All healthcare staff (including students and those working in the community) should receive occupational health advice and protection. Staff who have contracted infections such as impetigo or diarrhoea should report and be screened if necessary. Those that sustain needlestick injuries from potentially contaminated sources should have access to advice and post-exposure prophylaxis with antivirals and immunization. There should be an educational programme tailored to the needs of different cadres of staff both on induction and as part of continuing professional development.
Monitoring
Routine microbiological sampling of the environment is of little benefit, although monitoring of the physical performance of air-conditioning plants and machinery used for disinfection and sterilization is essential. In the event of an outbreak of hospital infection such as Legionnaires’ disease, more specific monitoring targeted at the known or likely causative micro-organism should be considered.
Microbiological screening of staff or patients is not undertaken routinely, but it may be needed for specific purposes: to detect carriers of MRSA and hepatitis viruses in those performing some types of surgery or where transmission to patients has occurred.
Surveillance and the role of the laboratory
The detection and characterization of hospital infection incidents or outbreaks rely on laboratory data that alert the infection control team to unusual clusters of infection, or to the sporadic appearance of organisms that may present a particular infection risk or management problem. This is sometimes referred to as the ‘alert organism’ system. Bacterial typing schemes and antibiograms (see Ch. 3) are very important in this regard. In some situations mandatory surveillance of cases of infection with particular pathogens such as MRSA or C. difficile may be useful to compare hospitals or units and reduce hospital infection by regular, timely feedback to clinicians and managers. Regular visits to the wards are also important to record data on infected patients from whom no specimens have been received and to respond to problems as they occur. Such visits also serve to provide opportunities for practical teaching, which is another important element of the infection control team’s responsibility. Advances in surgical practice have lead to short stays in hospital so that some postoperative infections are only detected after discharge. Surveillance of surgical site infection should include data collected post-discharge.
Efficacy of infection control
The evidence base in the literature for acceptable proof of efficacy for infection control measures is limited. These include sterilization, hand-washing, closed-drainage systems for urinary catheters, intravenous catheter care, perioperative antibiotic prophylaxis for contaminated wounds and techniques for the care of equipment used in respiratory therapy. Isolation techniques are assumed to be reasonable as suggested by experience or inference. Measures which are now considered to be ineffective include regular chemical disinfection of floors, walls and sinks, and routine environmental monitoring.
Effective surveillance and action by the infection control team have been shown to reduce infection rates. One important role of the team is to monitor compliance with practices known to be effective and to eliminate the many rituals or less effective practices which may even increase the incidence or cost of cross-infection. As further advances occur in medical care and limited health care resources are spread across hospital and community needs, innovations in infection control will need to be evaluated for efficacy and cost-effectiveness. With this understanding it is possible that hospital infection can be controlled and largely prevented. The dictum of Florence Nightingale, made over a century ago, that ‘the very first requirement in a hospital is that it should do the sick no harm’, remains the goal.
Fraise AP, Bradley C. Ayliffe’s Control of Hospital Infection, ed 5, London: Hodder Arnold, 2009.
Fraise AP, Lambert P, Maillard J-Y. Russell, Hugo and Ayliffe’s Principles and Practice of Disinfection, Preservation and Sterilization, ed 4, Oxford: Blackwell, 2004.
Wenzel RP, ed. Prevention and Control of Nosocomial Infection, ed 4, Baltimore: Lippincott, Williams and Wilkins, 2002.
Centers for Disease Control. Healthcare-associated Infections (HAIs). http://www.cdc.gov/hai/.
Department of Health (England). Reducing Healthcare Associated Infection. http://hcai.dh.gov.uk/.
Health and Safety Executive. http://www.hse.gov.uk/.
Hospital Infection Society. http://www.his.org.uk/.
Infection Prevention Society (formerly Infection Control Nurses Association). http://www.ips.uk.net/.
Medicines and Healthcare Products Regulatory Agency. http://www.mhra.gov.uk/.
National Resource for Infection Control (NRIC). http://www.nric.org.uk/.
Thames Valley University Evidence Based Guidelines. http://www.wolfson.tvu.ac.uk/research.