66 Diagnostic procedures
Key points
• A central aim in choosing antimicrobial therapy is to match the most appropriate agent to the specific microbial aetiology. In practice, this is constrained by urgency of the situation, the diagnostic information available and policy decisions about antimicrobial use that are made on a local basis consistent with national policies.
• The initial decision on whether or not to give an antimicrobial and on the agent chosen is generally made before microbiological information is available but, where possible, after microbiological samples have been taken.
• In the UK, local and specific guidance is generally available for chemotherapy of common infective syndromes. The strategy reflects the likely causal agents and their local susceptibility patterns, the need to control antimicrobial resistance, and cost. This guidance is reflected in the local antibiotic policy.
• The laboratory will assist in modifying or initiating therapy by determining in vitro susceptibilities and by monitoring antimicrobial drug concentrations in specific circumstances. These analyses may lead to modification of therapy.
• Certain organisms such as streptococci and anaerobes have predictable susceptibilities, whereas others, such as enterobacteria, must be monitored continuously.
• Prophylactic antimicrobials are recommended in highly defined settings and following precise regimens. Ad hoc usage is to be avoided.
• Antimicrobial combinations may be given to increase efficacy, extend the spectrum of cover or prevent the development of resistance during therapy.
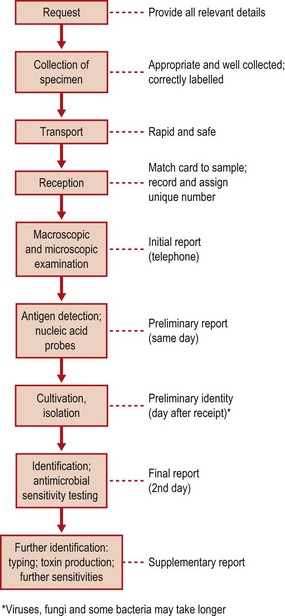
The role of the laboratory in assisting clinicians in the diagnosis of infection is illustrated in the specimen flow diagram shown in Figure 66.1. The choice of specimen depends on following the principles outlined in Chapter 65. The microbiology laboratory requires enough information on the request card accompanying the specimen to use the optimal methods necessary for identification of potential pathogens in particular infective syndromes. At the most basic level, it is obvious that, when a swab is received in the laboratory, it is necessary to know if it comes from the throat or the vagina! But additional information is also essential: is the patient being investigated for pharyngitis or diphtheria; for vaginal discharge or septic abortion? Furthermore, the specimen must be obtained with care and transported to the laboratory without delay in an appropriate manner. The value of the result is in direct proportion to the attention given to these details, as well as to the skill and efficiency of the laboratory. For further details of laboratory methods, including specimen containers and culture media used, the reader is encouraged to consult the recommended reading section.
Collection of specimens
Samples for microbiological examination need to be carefully collected, if possible without contamination with commensals or from external sources. Some points to remember with specimens from individual sources are shown in Table 66.1. It is essential to use sterile containers which are leak-proof and able to withstand transportation through the post if necessary. It is more convenient for both the clinician and microbiologist if the laboratory provides request cards, containers and an efficient transport system. There is a need for staff to be aware of safety regulations and for all parties to understand who has responsibility for each step of the process and how to minimize handling by untrained people. Special precautions required for ‘high-risk’ specimens need to be defined by the laboratory and hospital management. Storage of clinical material must be separate from food and drugs, and this may necessitate provision of additional refrigerator space and transport facilities.
Table 66.1 Some important points to remember in the collection of specimens for microbiological examination
| Type of specimen | Comments |
|---|---|
| Respiratory secretions | |
| Nasal swab (anterior) | Only for carriage of staphylococci and streptococci |
| Nasopharyngeal swab | For pertussis and meningococci |
| External ear swab | Wide range of microbes, including fungi |
| Myringotomy and sinus samples | As for abscesses – including anaerobes |
| Throat (pharyngeal) swab | Specify if only for streptococci; mention if diphtheria possible; use special transport media for virology |
| Saliva | Used for antibody detection (gingival swab); otherwise discard |
| Laryngeal swab | Specify if for mycobacteria |
| Expectorated sputum | Often poorly collected; specify mycobacteria, legionellae, pneumocystis |
| Transtracheal aspirate, bronchoscopy specimens, lung biopsy | Specify likely diagnosis; ask for specific tests |
| Pleural fluid | Treat as pus; always look for mycobacteria |
| Gastrointestinal specimens | |
| Vomitus | Only for virology |
| Gastric washings | For mycobacteria (particularly in children) |
| Gastric biopsy | For Helicobacter pylori |
| Duodenal/jejunal aspirates | Protozoa (Giardia lamblia, microsporidia, etc.) |
| Liver aspirates | As for pus (anaerobes); consider amoebae |
| Spleen puncture | For Leishmania spp. |
| Rectal biopsy | Schistosomiasis |
| Rectal swab | Only for gonococci and chlamydia |
| Colonic biopsy | Histopathological diagnosis of amoebiasis, pseudomembranous colitis (Clostridium difficile) |
| Colonic scrapings | Protozoa; amoebic trophozoites (deliver to laboratory immediately) |
| Faeces | Specify possible diagnosis; ask for clostridial toxins, parasite examination if suspected |
| Peri-anal swab | For eggs of threadworm |
| Urine | |
| Mid-stream (MSU) | Suitable for most patients |
| Clean catch | Infants and elderly – increased contamination |
| Suprapubic aspirate | Infants and neonates |
| Ureteric/bladder washout | To localize infection |
| Prostatic massage | Collect samples before, during and at end of micturition |
| Terminal urine | Schistosome ova, chlamydia DNA amplification |
| Complete early morning or 24-h urine | Mycobacteria (tubercle) |
| Central nervous system | |
| Cerebrospinal fluid by spinal tap | For meningitis collect sample for protein and glucose – test blood sugar simultaneously, specify virology, fungi or syphilis serology |
| Ventricular tap | Specify if through an indwelling shunt or catheter |
| Brain abscess | As for pus (include anaerobes) |
| Skin and soft tissue | |
| Skin scraping/nail clipping | Dermatophyte fungi |
| Skin swab | Rarely valuable without pus |
| Skin snips | Onchocerciasis – seek advice |
| Vesicle fluid | Suitable for electron microscopy for viruses |
| Wound swab | Obtain pus if possible; record site |
| Pus, tissues, aspirates | Describe site and any relevant operative details |
| Genital | |
| Urethral swab | Pus for gonococci, scrape for chlamydia |
| Vaginal swab (adult) | Candida, trichomonas, bacterial vaginosis or chlamydia DNA |
| Vaginal swab (prepubertal) | State age; caution required if abuse possible |
| Cervical swab | Separate media for chlamydia |
| Ulcer scrape | Immediate dark-ground microscopy; separate media for virology or chancroid |
| Uterine secretions | Specify puerperium or post-abortion |
| Pelvic aspirates | As for pus |
| Laparoscopy specimens | Include chlamydia specimen |
| Eye | |
| Conjunctival swab | Separate virology; scrape for chlamydia |
| Aspirates | As for pus |
| Blood | |
| Culture | Strict aseptic technique; take large sample in special media before antibiotics |
| Bone marrow | Valuable for leishmania, mycobacteria, brucella |
| Film | Malaria (thick and thin), filaria, borrelia, trypanosomes |
| Whole blood | Filaria (day or night samples as appropriate) |
| Serum antigen | Rapid diagnosis of many microbial diseases (e.g. hepatitis B) |
| Serum antibody | Retrospective diagnosis of common viral diseases, syphilis and other selected infections; need rising titre or specific IgM |
Food, water and environment
The examination of nonclinical specimens is beyond the scope of this book and readers are referred to appropriate reading. However, an outbreak of gastrointestinal disease inevitably leads to the question of identifying the source and comparing strains isolated. When disease is due to preformed toxins, as with staphylococcal food poisoning, faecal examination is unhelpful, and the diagnosis can only be made by testing the food. Frequently, the offending item has been discarded and the examination of food and water related to specific patients is often unrewarding. Routine sampling of water sources and potentially contaminated food such as poultry at various critical points of production is essential in maintaining good public health. As part of the investigation of an outbreak it may be necessary to collect samples under controlled conditions which can be used as evidence in any prosecution. Laboratories need to be able to receive and process such specimens in an approved way.
Transport
Many microbes may perish on transit from the host’s body to a laboratory incubator. Some contaminants, especially coliforms, may overgrow the pathogen and so mask its presence. These two constraints make it essential that any material for cultivation of microbes is transported as quickly as possible to the laboratory in a manner expected to protect the viability of any pathogens. Such problems may be minimized by the use of antigen or gene probe detection because of the relative stability of the chemical structures identified on dry swabs.
The ideal situation is to bring the patient to the laboratory for specimen collection or take the laboratory to the clinic. Both approaches are used for special purposes but are obviously inconvenient for many patients and inappropriate and costly for complicated techniques such as virus isolation which need specialized (and safe) facilities.
To overcome any drawbacks due to delay in reaching the microbiology department the following methods may be used:
1. Transport media. See Table 66.2.
2. Boric acid. The addition of boric acid to urine at a concentration of 1.8% (v/w) will stop bacterial multiplication but lower concentrations are ineffective and higher ones may kill the pathogen.
3. Dip slides. These provide a convenient way of inoculating urines at the clinic. They comprise small plastic spoons or strips holding a thin layer of agar which is dipped into the urine and then put in a screw-topped bottle for transport. The agar adsorbs a fixed volume of urine and, after incubation, colony counts of bacteria give a semiquantitative estimation of numbers.
4. Refrigeration. Storage at 4°C before processing will prevent multiplication of most bacteria. However, delicate microbes such as neisseriae may not survive whereas certain organisms, notably listeriae, flourish at low temperatures.
5. Freezing. Temperatures of −70°C or below, which can be achieved in liquid nitrogen or special deep freezes, will preserve many microbes, providing they are protected by a stabilizing fluid such as serum or glycerol.
Table 66.2 Types of transport medium
| Type of organism | Medium | Comments |
|---|---|---|
| Bacteria | Stuart’s semi-solid agar | Contains charcoal to inactivate toxic material |
| Anaerobic bacteria | Various systems, including gassed-out tubes and anaerobic bags | Not widely used, but essential for some strict anaerobes |
| Viruses | Buffered salts solution containing serum | Contains antibiotics to control bacteria and fungi |
| Chlamydiae | Similar to viral transport medium, but without agents that inhibit chlamydiae | Chlamydial antigen media contain detergent to lyse infected cells |
| Protozoa, helminths | Merthiolate–iodine–formalin | Kills active protozoa, but preserves cysts and ova in a form suitable for concentration and microscopy |
Reception
The importance of good documentation cannot be overstressed. No matter how well the specimen was taken, transported and processed in the laboratory, the end result depends on communication between people. The clinician making the request must give complete details on the request card and specimen to reduce errors. Staff receiving specimens in the laboratory must match them with the cards and record them into a book or computer. This is usually done by assigning a unique number to each specimen and labelling both the specimen and the request. When parts of the specimen are separated from the original bottle (e.g. after centrifugation of serum), the laboratory number becomes the only recognizable identification. Transcription errors are far more common than is supposed and are especially important for requests for human immunodeficiency virus (HIV) or syphilis serology, which may have disastrous consequences and medicolegal implications if wrong results are given.
Examination
Looking at clinical material with the naked eye or hand-lens should be part of the examination of a patient at the bedside. Sadly, many doctors delegate this to the laboratory staff. Many unnecessary laboratory tests could be avoided if unsuitable specimens were rejected on the ward or in the general practitioner’s surgery. These would include: crystal-clear urine from patients with ‘cystitis’; well-formed stools from patients with ‘diarrhoea’; and mouth washings or saliva from patients with respiratory symptoms.
It was once common practice to carry out certain basic investigations in ward side-rooms, and kits offering ‘near-patient testing’ are becoming available. However, there are cogent arguments against bedside pathology, including issues of safety, time, competence and quality control.
Microscopy
Microscopy is an important part of the examination of many specimens. For bacteriology, the Gram and acid-fast (Ziehl–Neelsen or Auramine) stains are usually sufficient, but for the demonstration of fungi or parasites special stains or concentration techniques may be required. ‘Wet’ mounts, i.e. unstained preparations of fluid material, are widely used in looking at cells in urine, cerebrospinal fluid (CSF), faeces and vaginal secretions. None of these procedures takes more than 5 or 10 min and all are inexpensive in reagent costs and capital equipment. They are therefore ideal rapid methods and new diagnostic techniques have to be judged against microscopy. An initial report, such as ‘Gram-negative diplococci and pus cells seen’ from meningitic CSF, can be issued within minutes of receiving the specimen and will aid the clinician in confirming the diagnosis and starting appropriate antibiotics.
Similarly, a rapid diagnosis of falciparum malaria can be lifesaving. Indeed, suspected pyogenic meningitis and falciparum malaria are among the few conditions for which it is clearly justifiable to call upon emergency laboratory services outside normal working hours.
Electron microscopy (EM) requires more elaborate preparation than does light microscopy and it is therefore much slower. It was valuable in the diagnosis of certain viral infections, including viral diarrhoea but the availability of antigen (ELISA) and molecular probes has largely replaced EM. The nonspecific nature of electron microscopy gives it an advantage in that any type of viral agent, if present in sufficient numbers, may be recognized.
Nonculture methods
There are many situations where isolation of microbes in vitro or in tissue culture is impossible or is insufficiently sensitive to make a microbiological diagnosis. Some microbes are so fastidious or slow-growing that useful information cannot be given to clinicians. The isolation of many viruses requires laborious tissue culture methods that are too slow to influence patient management. Microscopy is usually of low sensitivity, e.g. the threshold of detection of acid-fast bacilli in sputum is about 105 microbes per millilitre. Microscopy also has low specificity; for example, Gram staining of faeces would yield millions of Gram-negative rods, but it is not possible to recognize those that are pathogenic by this means.
To overcome these deficiencies probes have been developed which combine a part that reacts with a specific microbial structure and a part that will produce a signal (colour, fluorescence or radioactivity) after the reaction. Many microbes are detectable in this way. Antibodies labelled with fluorescent molecules are widely used in diagnostic virology, e.g. for respiratory secretions to find respiratory syncytial virus. Enzyme-linked antibodies are available as enzyme-linked immunosorbent assay (ELISA) kits for chlamydia detection, and many other immunoprobes are commercially produced or under investigation. The explosion in molecular biology has led to the widespread availability of DNA and RNA probes, which have changed from complicated research techniques requiring radioisotopes to relatively simple methods that can be carried out with minimal expertise. Use of amplified nucleic acid tests (e.g. polymerase chain reaction, PCR) has greatly increased the sensitivity of probes. Provided a unique sequence of nucleotides is used, the method is highly specific. None the less, as the new technology becomes more commonplace traditional methods will not be discarded. It is more likely that it will complement them.
One of the chief advantages of probes – their specificity – is also one of their major disadvantages. For example, in the investigation of diarrhoea, it would be impossibly laborious to have to use a separate probe for each of the possible microbial causes. Furthermore, use of probes leaves little scope for detection of the unexpected and hampers the discovery of previously unsuspected aetiological agents of disease. The advent of ‘chip’ technology (microarrays) may overcome this deficiency (see Ch. 6).
Serology
In situations in which microbial isolation is impossible and probes are unavailable, evidence of infection may be obtained by finding a rise in antibody titre or the presence of specific IgM. Serology is still used for diagnosing the causes of ‘atypical’ pneumonia (mycoplasmal pneumonia, psittacosis, Legionnaires’ disease), syphilis, brucellosis and many viral infections, including HIV. It is preferable to take a blood sample early in the illness (the acute serum) and another 10–14 days after the onset (the convalescent serum); a four-fold or greater rise in antibody titre in the second specimen is diagnostic of acute infection. With some infections, such as HIV and hepatitis B and C, much longer times must elapse.
Isolation of micro-organisms
The basis of the study of medical microbiology was laid over a century ago by the isolation of microbes in pure culture outside the host animal. The methods used by the fathers of bacteriology have been adapted, simplified and, in some cases, automated for the modern diagnostic laboratory. The principles remain the same: use sterile equipment and media (with cell lines if necessary) and add clinical material. After incubation at 37°C, for a variable time, from a few hours for enterobacteria to weeks for mycobacteria and some viruses and fungi, a visible effect will be produced. This might be colonies growing on agar or a cytopathic effect (CPE) in tissue culture. The skill of microbiology is in identifying the microbes responsible for the effect.
There are limits to the methods that can be used in a routine hospital laboratory to isolate microbes. The choice of media used is dictated by the specimen and by the clinical condition of the patient. For example, in some areas of the world there is little point in looking for Corynebacterium diphtheriae in every throat swab, so that the specific media needed are not routinely used. It is therefore incumbent on the doctor to make sure that the laboratory is alerted to look for diphtheria bacilli in any suspicious case. Similarly, clinicians need to know which microbes are routinely sought from particular specimens so that they can make a special request if they suspect the unusual.
‘New’ causes of illness are often found and new methods of investigating old diseases regularly appear on the market. It is a difficult decision for the laboratory manager to assess at what point a ‘new’ pathogen becomes sufficiently important to be routinely sought, or to balance the advantages of new (and expensive) technology against cheap and well tried techniques especially in resource poor countries.
Identification
The full identification of each microbe isolated in a clinical laboratory is both uneconomic and unnecessary. Shortcuts must be made to satisfy the clinical demand of a final report which the doctor can understand and which is available in time to influence management of the patient. In practice, most laboratories use simple and incomplete methods of identification, depending on the level of useful information required. Typing of isolates is for epidemiological or other special reasons, and this is usually done in national reference centres using standardized methods.
Examples of the extent of identification are shown in Table 66.3. This shows that the same organism may not be identified even to the genus level, or it may have extensive genetic investigation depending on the reason for the request. In a cost-conscious climate you get what you pay for and what the service thinks you need!
Table 66.3 Examples of bacterial identification in medical laboratories
| Extent of identification | ||
|---|---|---|
| Reason for request | Example 1 | Example 2 |
| Test of sterility | Bacteria present | |
| Initial blood culture report | Gram-negative rod | Gram-positive cocci |
| Urine examination | ‘Coliforms’ | Staphylococci |
| Wound swab | Escherichia coli | Staphylococcus aureus |
| Outbreak epidemiology | E. coli O157 | Staph. aureus phage type 80/81 |
| Pathogenicity tests | E. coli O157 vero toxin-producing | Staph. aureus enterotoxin A-positive |
Antimicrobial sensitivity testing
One advantage of good bacterial identification is that it helps greatly in choosing antibiotics and in some situations in-vitro tests of susceptibility may be unnecessary. However, the widespread occurrence of bacterial resistance, even in genera such as Neisseria and Haemophilus in which sensitivity to β-lactam antibiotics was previously assumed, has meant that tests are usually performed on all significant clinical isolates of bacteria. The relevance of information generated in the extremely artificial conditions of the laboratory is discussed in Chapter 67.
As there are technical problems in carrying out sensitivity tests at the same time as the primary culture, the report will be delayed for at least 24 h after isolation of the pathogen. Tests of slow-growing bacteria such as mycobacteria take much longer and tests involving fungi, viruses and protozoa are not ordinarily available. In general, all patients with acute symptoms will receive treatment before the report returns to the doctor so that the result will merely confirm that correct treatment had been chosen. Sometimes, the laboratory report will allow speculative antimicrobial therapy to be modified, e.g. by allowing one or more drugs in a precautionary mixture to be discontinued.
A frequent difficulty facing microbiologists is which, out of a rapidly increasing number of agents, should be tested and, of those tested, which should be reported. Most laboratories test a few representative compounds from among the many penicillins, cephalosporins, aminoglycosides, tetracyclines, quinolones, etc., and restrict those reported to the clinician to two or three agents selected for their appropriateness in the particular infection. In this way, institutional antibiotic policies are reinforced and the impact of the promotional activities of pharmaceutical companies is lessened.
Reports of results
Just as it is important to obtain as accurate a result as possible, so it is vital to transfer the information rapidly to the user of the laboratory in a form that is easily intelligible. Laboratory workers need to tailor their reports to suit their customers and to recognize their needs. Trained microbiologists are used to the vagaries of microbial taxonomy and the profusion of antibiotics with similar names, but physicians and surgeons are easily confused by laboratory jargon and changes in nomenclature. On the other hand, clinicians rightly expect laboratory reports to be explicit and helpful. As in so many spheres of health care, a spirit of mutual respect and cooperation is in the best interests of the patients, and this is most likely to happen if microbiologists regularly visit the wards and if clinicians are encouraged to discuss problems with laboratory staff.
In these days of computers and facsimile machines it is attractive to use the latest technology to transfer results, but the personal touch is essential, particularly for important results such as positive blood or CSF cultures. Clinicians dealing with difficult problems enjoy the reassurance that the laboratory has found the cause of a patient’s illness and advice on management is generally well received. Seeing the problem for oneself and discussing it with the responsible doctors is the ideal. The telephone is a satisfactory substitute in many cases and is far preferable to a report form printed by a computer and arriving after the crisis is over. It behoves us all to communicate better and faster.
Notification of infectious diseases
In addition to making a clinical diagnosis and treating the individual there is a need with some infectious diseases to determine the source and prevent further spread. In some countries there are public health laws specifying the method of reporting these. The list of notifiable diseases in England and Wales is shown in Box 66.1. This reporting system requires a clinical diagnosis, with or without laboratory confirmation, which is notified separately. In England and Wales each health district has a consultant responsible for communicable disease control, who is usually the ‘proper officer’ for the local government authority. Notifications are sent to the Health Protection Agency (HPA), previously the Communicable Disease Surveillance Centre (CDSC).
Box 66.1
List of notifiable diseases in England and Wales
• Acute encephalitis/meningitis
• Dysentery (amoebic or bacillary)
• Enteric fever (paratyphoid/typhoid)
Health Protection Regulations 2010.
There is also an obligation, which in some circumstances may be statutory, for laboratories to report isolates of certain pathogens to central authorities for surveillance purposes. For HIV/AIDS infection the system is voluntary and confidential, yet over 90% of cases are reported in the UK. In an outbreak, or where the pathogen is highly infectious, it is essential for the head of the laboratory to inform both the clinician looking after the patient and local public health staff. In this way a coordinated approach can be made to prevent further spread of infection. In particular, if there is any indication that the infection may have been acquired by staff working in the laboratory this must be reported to the relevant authority; in the UK that is the Health and Safety Executive.
American Society for Microbiology. Manual of Clinical Microbiology, ed 10. Washington, DC: American Society for Microbiology; 2011.
Forbes BA, Sahm DF, Weissfeld AS. Bailey and Scott’s Diagnostic Microbiology, ed 10. St Louis: Mosby; 2002.
Johnson FB. Transport of viral specimens. Clinical Microbiology Reviews. 1990;3:120–131.