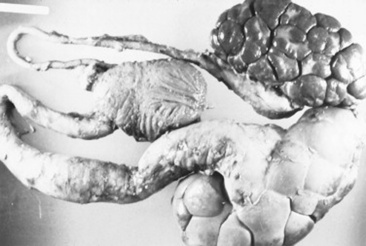
EVERSION OF THE BLADDER AND PROLAPSE OF THE BLADDER
Eversion of the bladder is an uncommon event that occurs during or shortly after parturition in cows.100-104 Forceful straining moves the bladder fundus caudally, eventually turning the bladder inside-out. The bladder is then forced out of the urethral orifice. Prolapse of the bladder is also a rare periparturient event in cattle, more frequently associated with dystocia.100,101 In this condition a full-thickness tear of the vaginal wall occurs during delivery, allowing the bladder (and possibly other viscera) to be displaced from the abdominal cavity into the vagina.
Clinical Findings and Differential Diagnosis
Cows with either eversion or prolapse of the bladder have a smooth, spherical mass within the vagina, usually protruding from the vulva. An affected cow may be alert and ambulatory, but if concurrent hypocalcemia, exhaustion, or peritonitis exists, the cow may be recumbent and depressed. Careful vaginal examination is required to differentiate these two conditions from each other and from vaginal prolapse, vaginal polyps, fat protrusion from a vaginal tear, vaginal neoplasia, fetal membranes, and uterine prolapse. Prevention of straining through epidural anesthesia is essential because expulsive efforts can cause herniation of other viscera through the urethra (bladder eversion) or through the vaginal tear (bladder prolapse).100 Epidural anesthesia also facilitates cleaning of the area because straining may result in continual fecal contamination of the perineum and vestibule.
EVERSION OF THE BLADDER
The mucosal surface of the bladder is exposed. The ureteral openings may be visible on its dorsal aspect, although these may be occluded and difficult to see if the wall of the bladder is edematous.103 Vaginal palpation reveals that the protruding tissue originates from the urethral orifice. With time, constriction of the everted bladder by the narrow urethra may cause venous congestion, edema, thrombosis, and eventual necrosis.100 Palpation and ultrasonographic examination are required for detection of herniation of other viscera through the urethral orifice and into the interior of the everted bladder. Strangulation of incarcerated bowel may occur.101,102 Careful fine-needle aspiration of the interior of the eversion has been used to differentiate eversion from prolapse of the bladder. With bladder eversion, aspiration may yield peritoneal fluid, but laceration of herniated bowel is a concern.100
PROLAPSE OF THE BLADDER
The serosal surface of the bladder is exposed. Careful palpation of the vagina reveals that the bladder protrudes from a full-thickness tear in the floor or lateral wall of the vagina. Other viscera may be present in the vagina as well. Fine-needle aspiration yields urine from the bladder lumen.
Treatment and Prognosis
EVERSION OF THE BLADDER
Manual reduction of the everted bladder may not be possible if it is edematous or if other viscera have herniated into its interior. The dorsal aspect of the urethra may be incised to widen the route through which the bladder is to be replaced.100,101 Laparotomy is required for assessment of the viability of herniated bowel, and subtotal cystectomy may be performed if extensive bladder trauma or necrosis has occurred.101 The viability of the involved structures is of primary concern for prognosis. Bladder paralysis and rupture are potential sequelae to ischemic damage that develops during eversion.100 Cystitis and pyelonephritis may develop as well, and antibiotic therapy is warranted if repair is attempted. Animals with chronic conditions may develop hydroureter, hydronephrosis, and renal failure.104
PROLAPSE OF THE BLADDER
A flexible catheter may be passed into the urethra to remove urine from the bladder, thereby confirming the diagnosis and facilitating bladder replacement. After catheterization and removal of urine, the bladder can be replaced into the abdominal cavity and the vaginal tear can be sutured. Severe contamination of the peritoneal cavity may render attempts at treatment unjustified. Antibiotic therapy is indicated for surgical candidates.
PELVIC ENTRAPMENT OF THE BLADDER
In pelvic entrapment of the bladder, the apex and fundus of the bladder are displaced caudodorsally into the pelvic cavity, resulting in impaired urine outflow. In one report the condition was diagnosed in two Holstein cows a few days after parturition had occurred, suggesting a potential role of delivery or postpartum straining in bladder displacement.105 Entrapment of the bladder in a perineal hernia106 or within a vaginal prolapse107 may also occur.
Clinical Findings
Bladder emptying is impaired, and the presence of the bladder in the pelvic inlet induces straining. An affected cow may show tenesmus, pollakiuria, and stranguria. On rectal examination a soft, fluctuant mass may be detected beside the vagina.105 In calves, radiography has been used to demonstrate the pelvic position of the bladder.108
Differential Diagnoses
Differential diagnoses include proctitis, vaginitis, retained placenta, bladder paralysis, cystitis, and perivaginal abscess. Needle aspiration of the bladder per vaginum has been used to make a definitive diagnosis,105 although the danger of uterine puncture should be considered.107 Ultrasonographic examination can also be useful for definitive diagnosis.107 Draining the bladder through catheterization or needle aspiration facilitates replacement of the bladder by manipulation per vaginum. However, laparotomy was necessary in one cow to reset the bladder because of the development of fibrinous adhesions between the bladder and vagina.105 In cases of bladder entrapment within a vaginal prolapse, complete correction of the prolapse usually restores the bladder to its normal position.105
ENZOOTIC HEMATURIA
Enzootic hematuria is a disease of chronic or intermittent hematuria in cattle and sheep and is associated with chronic ingestion of bracken fern (Pteridium aquilinum)109-115 (Fig. 34-21). A different fern species, Cheilanthes sieberi, may induce this disease in Australian cattle.109,111 Hemorrhagic cystitis is the initial consequence of exposure to the toxic compound(s) in the plant. With continued ingestion of bracken fern, cattle develop bladder neoplasms of epithelial, mesenchymal, or mixed origin. Bladder infection with bovine papillomavirus type 2 is involved in carcinogenesis.
Clinical Findings
In most cases, several animals are affected in a group that is grazing a particular pasture or being fed a particular type or cutting of hay. Protracted, possibly intermittent, hematuria is the first clinical sign detected in most animals.109 Blood clots may be voided on occasion. Chronic blood loss eventually results in tachycardia, tachypnea, exercise intolerance, pale mucous membranes, and a decline in productivity and body condition. Bladder wall thickening and bladder tumors may be palpated per rectum. Proliferative changes or overt neoplasia of the bladder may cause dysuria, pollakiuria, and rarely, obstruction of the bladder trigone. Occasionally, blood clots may cause urethral obstruction. Depending on the magnitude and duration of bracken fern ingestion, hematuria may last for months to years before severe debilitation or death occurs. Ultrasonographic examination of the urinary bladder of affected animals may reveal bladder thickening and an irregularly shaped bladder wall. In an Indian study, affected cattle showed a bladder wall thickness of 4 to 5 mm (normal, 1 to 2 mm).110
The syndrome of enzootic hematuria is quite different from acute bracken poisoning, which occurs after ingestion of large quantities of bracken fern (approximating the animal’s body weight), usually over 1 to 3 months.111,112 Acute bracken poisoning manifests as an acute coagulopathy or fulminant septicemic crisis associated with severe bone marrow suppression. Clinical signs include fever, profound weakness, epistaxis, hyphema, dysentery, and petechial hemorrhages of the mucosal surfaces and sclera.111 Acute bracken poisoning is further described in Chapter 54.
Clinical Pathology
Severe anemia is often seen on hematologic examination of cattle and sheep with enzootic hematuria. Evidence of a regenerative response may not be present if bone marrow suppression is severe. The platelet, segmented neutrophil, and lymphocyte counts may be reduced.111-113 Urinalysis reveals hematuria, proteinuria, and variable pyuria.109
Differential Diagnosis
Examination of serum for evidence of hemolysis and analysis of sediment from a freshly voided urine sample for intact red blood cells allow for differentiation of hematuria from the hemoglobinuria found in hemolytic diseases. Icterus, also characteristic of ruminant hemolytic disorders, is not found in cases of enzootic hematuria. Hematuria may be evident in urinary tract infection, but pyuria and bacteriuria are marked, and anemia, if present, is usually mild. Simultaneous involvement of several animals is uncommon with urinary tract infection but common in enzootic hematuria. Hematuria may be evident in cattle affected by malignant catarrhal fever (MCF). Protracted, severe hematuria is rare in cases of urolithiasis, and anemia is also not expected. Without necropsy, a diagnosis of enzootic hematuria requires documentation of access to bracken fern in animals with characteristic clinical signs and laboratory data.
Epidemiology
Enzootic hematuria has a wide geographic distribution, with cases reported in North and South America, the United Kingdom, Australia, and several European countries.109 Bracken fern is found in all areas of the United States except the Great Plains, with most livestock poisonings occurring in the Pacific Northwest and upper Midwest.112 The plant grows best in well-drained, fertile soils and is often localized in open areas of forests.114 Sheep and cattle are poisoned by grazing the plant or consuming contaminated hay.114
Enzootic hematuria is primarily seen in adult sheep and cattle. In field cases, cattle grazing infested pastures develop hematuria by 2 to 3 years of age.109,115 Feeding adult cattle 1 to 2 kg bracken fern/head/day led to hematuria within 10 to 15 months in one trial.109 Papillomas of the bladder occur as early as 1 year after bracken feeding begins, with invasive carcinomas arising 2 to 6 years later.109,115
Pathophysiology
All parts of the plant are toxic to sheep and cattle.111,112 Several compounds in bracken fern possess irritant, mutagenic, immunosuppressive, or carcinogenic activities.109,113 These include ptaquiloside (aquilide A), quercetin, and a-ecdysone.116-119 The carcinogenic principles are present in the milk of cows grazing bracken fern.120,121 Bracken fern compounds may cause recrudescence of latent bovine papillomavirus 2 (BPV-2) infections through immunosuppression. Bladder infection with BPV-2 follows, and mutagenic compounds in bracken fern interact with BPV-2 in the bladder to induce local neoplasia.109,113,116 Growth of the resultant neoplastic tissue may be enhanced by further exposure to mutagenic bracken compounds.109,113,116 Similarly, BPV-4 and mutagens from bracken fern may act in synergy to induce neoplasia of the mucosa of the upper GI tract.116,118,119
In multiple species, cyclooxygenase 2 (COX-2) is overexpressed in epithelial neoplasms, including those of the urinary bladder origin. Certain types of tumors of urinary epithelium may coexpress COX-1 and COX-2. Bladder carcinomas from cows with enzootic hematuria have been shown to express both COX-1 and COX-2 at a high level relative to normal controls, when evaluated by immunohistochemical methods.122 The efficacy of COX-2 inhibitor drugs on cancer prevention or treatment in cattle at risk for bladder tumors remains to be investigated.
Immunosuppression results from reduction in circulating neutrophil and lymphocyte counts. Neutropenia appears to be a reversible phenomenon that results from bone marrow suppression. Neutrophil counts may normalize within 1 to 2 weeks of cessation of bracken feeding. Lymphopenia persists during periods of low-level ingestion.113
Necropsy Findings
Tissue pallor from anemia is often appreciated. The bladder wall is thickened and the mucosa hemorrhagic and ulcerated. Microscopic examination of the bladder wall reveals capillary engorgement, intramural hemorrhage, and metaplasia of the bladder epithelium.111 Several types of bladder tumors and mixed-origin neoplasms may be present. Metastasis of epithelial neoplasms to the regional lymph nodes or other organs can occur.123 Pharyngeal, esophageal, or ruminal papillomas may be found as well, and carcinomas may develop in these same locations in cattle exposed to bracken fern over several years.115
Treatment and Prevention
Treatment of enzootic hematuria is limited to reduction or elimination of bracken fern in the diet. Wooded areas that support growth of bracken fern can be fenced off, and forage improvement may help to limit incorporation of the plant into hay. If such measures are not feasible, a program of early culling may help to avoid low productivity from anemia and neoplasia. Hematuria will cease if bracken feeding is discontinued before the onset of tumor formation.
URINARY TRACT INFECTION
Cystitis, ureteritis, and pyelonephritis in ruminants most often result from ascending urinary tract infection (UTI) with Corynebacterium renale or Escherichia coli.124 Less common causative organisms include various coliform species125 and other members of the C. renale group.126 Renal infection via the hematogenous route (suppurative embolic nephritis) is much less common but may result from bacteremia with such agents as Salmonella species, Actinomyces pyogenes, or in small ruminants, Corynebacterium pseudotuberculosis.127
Clinical Signs
Cystitis in cattle is typified by dysuria and pollakiuria, with or without gross hematuria and pyuria. During urination the flow rate is often decreased. An affected cow may tread or swish the tail and retain an arched stance after voiding has ceased. Blood, purulent debris, or crystalline material may occasionally be found on the hairs of the ventral commissure of the vulva. Rectal palpation may reveal a thickened, painful bladder. If UTI is limited to the bladder, an affected cow usually does not show generalized signs of infectious disease (e.g., fever, anorexia, depression).124
In contrast, cattle with acute pyelonephritis often have a history of an abrupt reduction in feed intake and milk production. Fever, depression, ruminal stasis, scleral injection, and occasional episodes of mild colic accompany the signs of cystitis previously described.124 With bilateral or left-sided pyelonephritis, renal enlargement, pain, and a loss of normal lobulation of the left kidney may be evident on rectal examination. Transabdominal ultrasonographic examination is useful for evaluation of the right kidney, because the right kidney usually cannot be reached during rectal examination unless it is greatly enlarged.124 In an adult cow, ultrasound evaluation of the right kidney can be performed using a 3.5-MHz transducer at the twelfth intercostal space. This transducer can also be used to view the left kidney at the dorsocranial aspect of the right paralumbar fossa.128 Alternatively, the left kidney can be imaged transrectally with a linear-array transducer. Dilation of renal calyces; echogenic, flocculent material within the renal pelvis; abnormal renal shape; and renal enlargement are ultrasonographic findings suggestive of pyelonephritis. Vaginal palpation is usually necessary to detect ureteritis124; the ureters may be enlarged and painful when palpated through the vaginal wall.
The clinical signs of chronic pyelonephritis are relatively vague and inconsistent.124 Weight loss or poor growth rate, anorexia, and reduced milk production are common presenting complaints. Posture and behavior during urination may be normal, although affected calves have been noted to dribble urine, thereby developing phosphatic calculi adherent to the vulvar hairs and urine scald of the perineum and hindlegs. Ulcerative vulvovaginitis may be present as well.129 Polyuria without gross urine abnormalities may be found in some cases. Diarrhea and pale mucous membranes may also be seen during physical examination. Rectal and vaginal examination findings for chronic pyelonephritis are similar to those of the acute form, although the involved structures may not be painful or enlarged when palpated.
Urinalysis with chemical reagent strips appears to be a sensitive ancillary test for both acute and chronic pyelonephritis. In a study of 15 cases of bovine pyelonephritis, clinical signs suggestive of urinary tract disease were found in only three cows. However, evidence of hematuria or proteinuria was found through routine reagent strip urinalysis in all 15 cases.124 Further examination and testing of the urinary tract confirmed a diagnosis of pyelonephritis.
Differential Diagnosis
Mild colic may result from a variety of GI disorders, but urinalysis findings are normal in these conditions. Enzootic hematuria usually affects multiple animals in a particular locale, and access to bracken fern is usually demonstrable. Although dysuria and hematuria may be evident in enzootic hematuria, anemia is profound, whereas pyuria and bacteriuria are mild or nonexistent. Urolithiasis may induce colic, dysuria, and hematuria. However, urolithiasis is almost exclusively a disease of male ruminants, and UTI is more common in females. Bladder distention is a common finding in urolithiasis but rarely occurs in UTI. Other conditions that may cause dysuria include vaginitis, vulvar trauma, perivaginal abscesses, and pelvic entrapment of the bladder. Careful assessment of the neurologic system is warranted in cases of UTI to determine if the underlying cause is bladder paresis or incomplete voiding.
Clinical Pathology
Neutrophilic leukocytosis and hyperfibrinogenemia are evident on hematologic evaluation of cattle with pyelonephritis. Hyperglobulinemia may develop if the infection is established for several days. Severe, protracted proteinuria may cause hypoalbuminemia, and the resultant low plasma oncotic pressure may contribute to the development of diarrhea in occasional cases. In chronic pyelonephritis, anemia may result from reduced erythropoietin production, chronic inflammatory disease, and blood loss through the urine.124
If azotemia is found on serum chemistry analysis, the clinician must consider renal and prerenal causes before formulation of a prognosis. Pyelonephritis with azotemia and isosthenuria would indicate bilateral renal involvement, lowering the chances for successful treatment.124
Urinalysis is required for definitive diagnosis of UTI, but careful collection technique is important for valid conclusions to be made. Concurrent metritis, vaginitis, or posthitis may result in contamination of urine with blood, bacteria, and inflammatory cells, particularly if the rate of voiding is slow. A midstream or end-stream catch is likely to provide the most accurate culture results.130 Tentative identification of the organism may be obtained through Gram stain of the urine.
Urinary tract infection consistently produces hematuria, proteinuria, and bacteriuria on urinalysis. Quantitative culture of a urine sample allows for confirmation of the diagnosis and identification of the causative organism. Although not present in all cases, leukocyte casts provide definitive evidence of pyelonephritis.131
Pathophysiology
Factors involved in ascending UTI include the dose and virulence of the bacterial challenge, the presence of urogenital trauma (e.g., from calving injuries) or abnormal vulvar conformation, obstetric manipulation, bladder catheterization, and urine retention (as occurs with bladder paralysis or urethral obstruction). After cystitis is established, alterations in the contractility and thickness of the bladder wall may promote vesicoureteral reflux, spreading infection into one or both ureters.132 Hemorrhage, fibrin deposition, and epithelial necrosis may result in intermittent ureteral or renal obstruction, which may be responsible for the episodic signs of colic occasionally seen in affected cows. Once pyelonephritis is established, necrosis of papillary and tubular epithelium leads to accumulation of necrotic debris in the renal pelvis, loss of functional nephron mass, abscess formation, fibrosis, and distortion of renal shape. Renal calculi, particularly struvite uroliths, may occasionally develop in cases of pyelonephritis. Crystal deposition on necrotic debris and the high local pH caused by bacterial urease activity may contribute to calculogenesis.
Corynebacterium renale is a large, pleomorphic, club-shaped bacillus that is aerobic, ureolytic, nonmotile, and gram positive.126 Pyelonephritis caused by C. renale has been reported in a sheep132 and induced experimentally in goats.133C. renale is adapted to and maintained in the bovine and ovine urinary tract and is unlikely to be maintained in the external environment for prolonged periods.126 Subclinical carriers and diseased animals transmit the organism through direct vulvar contact or by splashing urine droplets onto the vulvas of susceptible cows. Iatrogenic transmission through contaminated obstetric instruments or urinary catheters is also possible. Venereal transmission of C. cystitidis126 and C. renale134,135 from infected bulls may also occur.
Adherence of C. renale to urinary tract epithelium appears to be mediated by pili136 in a pH-dependent manner.137 Adherence is enhanced under alkaline conditions and inhibited by acidic conditions. This may explain the clinical improvements reported in infected cattle fed salts that promote urinary acidification.134 Through ureolysis and ammonia production, the organism maintains urine alkalinity, thereby facilitating colonization of the epithelial surface. A serum antibody response develops after renal infection is established, but this response is rarely curative124 and does not appear to impart resistance to reinfection with C. renale.138
Ruminant UTI is also frequently caused by Escherichia coli, a ubiquitous, gram-negative coliform bacterium.124 The serotype(s) and virulence factors of E. coli involved in bovine pyelonephritis have not been identified. Clinical evidence suggests that UTI results from fecal contamination of the urogenital tract or loss of normal urinary tract defenses.
Congenital defects such as ectopic ureter occasionally result in UTI, presumably from ascending infection of an abnormally positioned ureter. Impairment of bladder emptying, as might occur with bladder adhesions, urachal remnant infection, or diseases of the spinal cord, may promote ascending UTI. Urethral trauma caused by urolithiasis, breeding injury, urogenital papillomas, or catheterization of the urethra may also be conducive to infection.
Epidemiology
An Israeli study found that the prevalence of pyelonephritis on a per-farm basis varied between 0.3% and 2.7%.139 UTI is much more common in female ruminants than in males because of the relatively short urethral length in females140 and the potential for urinary tract contamination and trauma during parturition. Seventy-three percent of pyelonephritis cases developed within the first 90 days after calving, suggesting that the postpartum period is a critical time for initiation of UTI.139 Another study identified reproductive tract abnormalities such as pneumovagina, metritis, and poor perineal conformation in 7 of 15 cows with pyelonephritis.124
In the past, C. renale has been regarded as the most common causative organism for bovine pyelonephritis125; however, in recent studies from Israel, E. coli has been the most frequent cause.129,141 Once C. renale infection exists in a herd, the number of subclinically infected cows increases over time, and the infection becomes difficult to eradicate.126 Through increased frequency of contact, overcrowded cattle may experience more rapid transmission of infection.
Necropsy Findings
Hemorrhage, ulceration, and fibrin deposition are evident on the epithelium of the bladder and urethra. With chronic infection, polypoid growths may develop in the bladder mucosa; these masses grossly resemble tumors and must be definitively identified by histopathologic examination.125 One or both ureters may be enlarged, with purulent debris occasionally occluding the ureteral lumen. Pyelonephritis cases may show gross renal enlargement in acute to subacute cases (Fig. 34-22). On sagittal sectioning of the kidney, viscous, gray, odorless exudate is found within the renal pelvis and extending into the medulla and cortex.126 A Gram stain of the exudate is useful for differentiation of C. renale from E. coli infection. Renal abscesses, with gross distortion of renal size and shape, may be seen in cases of chronic pyelonephritis.
Treatment and Prognosis
Aggressive antibiotic therapy is essential for successful treatment of UTI. Penicillin is the treatment of choice for C. renale infection; recommended dosage regimen includes procaine penicillin G (22,000 to 44,000 IU/kg IM twice daily) or ampicillin trihydrate (11 mg/kg IM twice daily).124 For valuable animals, higher serum and urinary concentrations of penicillin may be achieved with IV administration of sodium or potassium penicillin (22,000 to 44,000 IU/kg every 6 hours) or sodium ampicillin (10 to 50 mg/kg every 8 hours). Treatment should be continued for a minimum of 3 weeks. Subcutaneous injection of antimicrobial drugs may be necessary to limit muscle pain and swelling during the course of treatment. Urinalysis and urine culture should be repeated 1 week after treatment is discontinued to ensure complete resolution. After prolonged therapy with these extralabel dosages of antibiotics, residue withdrawal times for meat and milk must be extended appropriately. In addition, induction of diuresis through oral or parenteral fluid therapy may aid in removing necrotic debris and bacteria from the lumen of the urinary tract.
Urinary tract infection with E. coli or other coliforms may also be successfully treated with high doses of penicillin or ampicillin.124 Achieving high urinary concentrations of these antibiotics may render them effective against many coliforms, even those that show in vitro resistance to the expected serum concentrations of the antibiotic.124 Repeated assessment of appetite, attitude, rectal temperature, and reagent strip urinalysis is recommended for monitoring cows with coliform UTI that are receiving penicillin or ampicillin therapy. If these parameters do not improve after 96 hours of treatment, another antibiotic should be chosen.124 Gentamicin (2.2 mg/kg IM twice daily) has been used to successfully treat refractory coliform UTI in a cow, but the nephrotoxicity of the drug and the current prolonged slaughter withdrawal period are important considerations.124 Trimethoprim-sulfadiazine (15 mg/kg IV once daily)125 and ceftiofur (3 mg/kg IV twice daily)142 have also been used with success.
The prognosis for UTI in ruminants depends on the duration of infection, the extent of UTI (cystitis alone vs. unilateral or bilateral ureteritis and pyelonephritis), and the remaining renal function. The chances for successful treatment are improved if treatment is initiated early in the course of infection. In recent reports the combined case fatality and cull rate for pyelonephritis in dairy cattle varied between 18%124 and 33%139 for treated cases; however, antibiotic dose and duration varied greatly between these two studies. Cows with pyelonephritis and marked azotemia (BUN >100 mg/dL) were found to be at much greater risk for culling (odds ratio = 60) than nonazotemic cows with pyelonephritis.139
Prevention and Control
Isolation of animals infected with C. renale is recommended to limit spread of the organism, and disinfection of heavily contaminated areas is advised. Aseptic technique during urogenital procedures and disinfection of obstetric and surgical equipment will limit iatrogenic transmission. In herds using natural service, venereal transmission by subclinically infected bulls may be difficult to control over the long term. An artificial insemination or mass treatment program may be required to prevent further losses from UTI.
AMYLOIDOSIS
Amyloidosis in cattle is caused by deposition of insoluble protein fibrils in the kidney, GI tract, liver, and adrenal glands. Renal amyloidosis in cattle is characterized as a sporadic, chronic wasting disease. Amyloid deposition in the kidney disrupts the normal glomerular structure, resulting in a protein-losing nephropathy.
Clinical Signs
The most common clinical signs of amyloidosis include chronic diarrhea, weight loss, and poor productivity in mature animals.143,144 Generalized or ventral edema may be present as a result of hypoproteinemia. Alterations in appetite and attitude may be present, although this may be caused by concurrent disease. Enlargement of the left kidney may be palpated during rectal examination. The enlarged kidneys generally are not painful and maintain normal lobular patterns. Urine may develops stable foam after hitting the ground or being collected and shaken in a container, a result of high urine protein concentration.
Clinical Pathology
Cattle with renal amyloidosis consistently develop marked proteinuria and hypolbuminemia.143 Serum creatinine and BUN levels may be elevated if renal damage is advanced. In cases of chronic, active inflammatory disease, hyperfibrinogenemia and hyperglobulinemia may occur.143,144 Polarized light microscopy and electron microscopy have been used to examine urine sediment for the presence of amyloid protein in urine.145
Differential Diagnosis
Amyloidosis must be differentiated from other diseases causing chronic diarrhea, hypoproteinemia, weight loss, and poor productivity. Diseases to consider include Johne’s disease, copper deficiency, salmonellosis, bovine viral diarrhea, GI parasitism, and glomerulonephritis. Other than amyloidosis, glomerulonephritis is the only other differential diagnosis routinely displaying prolonged proteinuria. Renal biopsy can be performed to differentiate glomerulonephritis from amyloidosis in the live animal.
Pathophysiology
Amyloidosis of cattle is classified as the reactive (AA) type,146,147 which is frequently associated with chronic inflammatory disease in domestic animals and humans.148 Concurrent inflammatory disease, such as traumatic reticuloperitonitis, pneumonia, mastitis, and metritis, have been found in some, but not all, cattle with amyloidosis.143,144 Serum amyloid A protein (SAA) is synthesized in the liver and is a precursor of amyloid A (AA) fibril in tissues.149 SAA concentrations increase dramatically in disorders such as trauma, neoplasia, and inflammatory disease. An elevation in SAA is apparently required for an animal to develop active amyloidosis.148 Elevations in SAA as a result of abnormal catabolism by the reticuloendothelial system may also increase AA fibril formation.148,150 AA fibrils are resistant to proteolysis, allowing for their accumulation in tissues over time.146 Accumulation of amyloid in the glomerulus alters glomerular filtration. A resultant hypoalbuminemia develops, which in turn decreases intravascular oncotic pressure. Diarrhea develops as a result of edema or amyloid deposition in the GI tract.144 The protein-losing nephropathy and diarrhea result in weight loss. Glomerular filtration rate will be reduced if the glomeruli are obliterated by amyloid deposition. Renal or pulmonary thrombosis may develop as a result of the loss of low—molecular-weight anticoagulants through the compromised kidney.151
Necropsy Findings
Renal enlargement with yellow-tan to white discoloration is frequently present. A waxy quality of the renal parenchyma may be appreciated on cut surface of the kidney.143 Generalized edema resulting from hypoalbuminemia may be present. Some animals will have renal or pulmonary thrombosis.144 Other inflammatory lesions may be found in other sites. Histologic examination of the kidney may reveal amyloid deposition in the glomerulus, interstitium, and tubule lumen. Immunohistochemical tests using antihuman AA monoclonal antibody can be used for specific demonstration of amyloid in bovine kidney specimens.151
Prognosis
Because the lesions of amyloidosis are irreversible, the prognosis for affected cattle is poor. The resilient nature of the amyloid protein results in its persistence in tissues, even if the underlying cause of inflammatory disease is treated successfully. Specific treatment for amyloidosis has not been reported in cattle.
GLOMERULONEPHRITIS
Glomerulonephritis (GN) is a rare clinical disorder of ruminants that may result from deposition of antigen-antibody complexes in the glomerular basement membrane or from binding of antibody to intrinsic or foreign antigens in the glomerulus. Glomerular injury occurs subsequent to targeting of glomerular tissues by the immune system. Nonimmune mechanisms may be involved in certain forms of the disease.
Clinical Signs
Cattle with GN may have a history of weight loss, poor productivity, and chronic diarrhea.152,153 Lethargy and generalized edema may be detected on physical examination. Rectal palpation may reveal a mildly enlarged but nonpainful left kidney.153 GN may be clinically occult in cattle persistently infected with bovine viral diarrhea (BVD) virus154 and in cattle with fascioliasis.155 GN has been associated with pregnancy toxemia in ewes; affected animals tend to show clinical signs typical of pregnancy toxemia156 (see Chapter 33).
Mesangiocapillary GN has been described in Finnish Landrace lambs of specific lineage in Scotland and Canada.157 The disease is heritable, but the exact mode of inheritance remains unknown. Clinical signs of this disease begin within hours after birth to 3 months of age. Affected lambs may be dull, ataxic, and appear blind. Fine muscle tremors, colic, and convulsions may also be seen.
Differential Diagnosis
The differential diagnosis for cattle with GN is similar to that for amyloidosis (see preceding section).
Clinical Pathology
Heavy proteinuria, mild anemia, and hypoalbuminemia have been reported in cattle with GN.152,153 Granular casts, red blood cells, and leukocytes were found in the urine sediment of one affected cow.152 Azotemia, proteinuria, and ketonuria are found in ewes with GN associated with pregnancy toxemia. Mesangiocapillary GN in Finnish Landrace lambs is characterized by uremia, hypoalbuminemia, proteinuria, hypocalcemia, and hyperphosphatemia.157
Pathophysiology
In humans, GN may result from a variety of infectious, toxic, or autoimmune disorders, all of which induce eventual immunologic injury to the glomerulus.152,158 Antibodies may be directed against host or foreign antigens located in the vascular endothelium, mesangial cells, or basement membrane. In addition, circulating immune complexes may deposit in the glomerulus. The ultimate consequences of antigen-antibody interaction in the glomerulus are activation of complement and chemotaxis of leukocytes, both of which result in direct glomerular injury and increased glomerular permeability.158
Filtration of plasma albumin through the damaged glomerulus results in chronic albuminuria, eventually leading to reduced plasma oncotic pressure and generalized edema. Passage of antithrombin III through the damaged glomerulus and into the urine may result in a hypercoagulable state.159
Immunohistochemical data suggest involvement of immune-mediated mechanisms for spontaneous GN in cattle152,153 and sheep,157 GN associated with persistent BVD infection and fascioliasis in cattle,154,155 and mesangiocapillary GN of Finnish Landrace lambs.157 In the last condition a heritable deficiency of the third component of complement has been documented, but the role of this deficiency in GN remains unclear.156 Glomerulonephritis may also be an incidental histologic finding in animals with acute septic disease.157
The clinical and histopathologic characteristics of GN of pregnancy toxemia in ewes resemble those of the preeclampsia syndrome of women.156 Enlarged glomeruli with reduced blood content in glomerular capillaries are found throughout the renal cortex of affected ewes. The renal lesion in preeclampsic women may result from endothelial injury during disseminated intravascular coagulation (DIC) or an excessive glomerular vasomotor response to angiotensin.160 The lesion can be reversible in women, but the consequences of this condition in ewes have not been described.
Treatment and Prognosis
Treatment of GN in ruminants has not been described. Because most cases of GN are advanced at diagnosis, the prognosis is poor. Mesangiocapillary GN in Finnish Landrace sheep is not invariably lethal, and some affected lambs may survive until adulthood.156
HEMOLYTIC UREMIC SYNDROME
Hemolytic uremic syndrome (HUS) is classified within the group of thrombotic microangiopathy syndromes.161 HUS is a set of symptoms characterized clinically by acquired, nonimmune hemolytic anemia, thrombocytopenia, and acute renal failure (ARF).162 Histologically, HUS is characterized by renal thrombotic microangiopathy.163 HUS is the most common cause of ARF in young children and infants, with prodromal diarrhea occurring in approximately 90% of cases.164 Evidence indicates that almost all the postdiarrheal human cases of HUS are caused by enterohemorrhagic (EHEC) or verotoxigenic Escherichia coli infections, and that the majority of the cases in the United States are caused by the EHEC serotype O157:H7.164 Although three horses165,166 and a heifer167 have been reported with clinical syndromes similar to HUS, an etiologic agent was not identified in any of these four animals.
Clinical Findings
Horses with clinical signs indicative of HUS have exhibited fever, diarrhea, hematuria, hemoglobinuria, profound azotemia, oliguria, and ventral edema.165,166 Hematologic findings included leukocytosis, anemia, and evidence of hemolysis.165,166 The blood smear of one horse revealed the presence of poikilocytes and schistocytes.166 All three horses were euthanized after unsuccessful treatment of anuria and azotemia with fluid therapy and diuretics. The reported case in a heifer was fatal postparturient HUS demonstrated by severe progressive anuric renal failure, acute hemolytic anemia, and consumptive thrombocytopenia.167
In humans, HUS presents as pallor, oligoanuria, edema, seizures (rarely), or generalized hemorrhagic diathesis. In prodromal human cases, this syndrome develops on average 1 week after onset of diarrhea.162 The classical clinical syndrome of HUS in people includes ARF, hemolysis, thrombocytopenia, and manifestations of DIC.163 For treatment of HUS in humans, evidence is insufficient to support use of specific therapies, and some treatments, such as certain antibiotics and motility-modifying agents, may be detrimental.162 Supportive therapy, including fluid/plasma therapy and dialysis, can be of paramount importance.
Pathophysiology and Necropsy Findings
In humans, HUS is the most common life-threatening complication of hemorrhagic colitis (HC) from EHEC infection. The EHEC strains produce the exotoxins Shiga toxin 1 (Stx1) and Shiga toxin 2 (Stx2), also referred to as verotoxin 1 and verotoxin 2.168 Although E. coli O157:H7 is the most widely publicized, as many as 100 different serotypes of E. coli (as well as Shigella and other Enterobacteriaceae) can carry the genes for Stx1 or Stx2 and are capable of causing disease.161
The EHEC organisms, including serotype O157:H7, are noninvasive but attach to the intestinal mucosa and produce characteristic histologic attaching and effacing lesions. The Shiga toxins (Stxs) released from the bacteria are believed to translocate across the mucosa, where they access the systemic circulation. The Stxs bind to specific glycolipid receptors on the surface of vascular endothelial cells, are internalized by endocytosis, and induce cell death through inhibition of protein synthesis.161 Activation of the coagulation cascade after exposure of subendothelial collagen may result in thrombosis of small vessels in the kidney and other organs. Oliguric or anuric ARF can result from fibrin/platelet thrombi in renal vessels and glomeruli, fibrinoid necrosis of vessel walls, congestion of glomeruli, and tubular ischemia. Because the coagulation events may be localized to certain organs, the results of laboratory tests of coagulation (e.g., PT, PTT) are not consistently abnormal.169
Cattle feces are considered to be the major source of EHEC; however, these bacteria have been isolated from the feces of many other asymptomatic species, including humans. EHEC organisms do not generally cause illness in cattle, although they do colonize the bowel. Fecal contamination of ground beef, other food sources, and water is thought to be the primary mode of transmission of the organisms. However, the small infectious dose makes person-to-person transmission a significant problem, especially in day care, nursing home, and outbreak situations.161
In the four large animals reported with HUS, the inciting cause was uncertain, and the isolation of EHEC or other Stx-producing organisms was not attempted.165-167 The heifer had a necrotizing endometritis, but during postmortem examination of the three horses, no focus of infection was identified. Renomegaly, renal infarcts, and scattered petechial and ecchymotic hemorrhages within the renal parenchyma were apparent on gross necropsy. Acute tubular necrosis and fibrin thrombi within the glomerular capillaries were evident on histologic examination.
Although the etiology was not determined, the clinical signs and pathologic lesions reported in the four large animal cases are compatible with a diagnosis of HUS. Based on these reports, the pathogenesis of HUS in large animal animals shares some features of the disease in humans.
TUBULAR NECROSIS
Tubular necrosis (TN), or tubular nephrosis, is the disease condition that results from a variety of toxic, infectious, or hemodynamic insults to the kidneys. Compounds identified as nephrotoxins for ruminants are listed in Box 34-2. Hemodynamic causes of TN include diseases that reduce renal perfusion (blood loss, endotoxic shock) or that occlude the renal vasculature (DIC, renal vein thrombosis). Bilateral bacterial infection of the kidneys may result in ARF or chronic renal failure (CRF) as a result of destruction of nephrons by bacterial toxins and the host inflammatory response. Renal infection may be established by ascending UTI or by hematogenous infection of the kidneys. Depending on the nature and duration of the primary insult, widespread dysfunction or necrosis of tubular epithelial cells may produce reversible renal injury, ARF, or CRF.