Chapter 7 Alterations in Alimentary and Hepatic Function
DIARRHEA
Diarrhea, is defined as an increase in the frequency, fluidity, or volume of bowel movements. Diarrhea may be a sign of a primary bowel disease or a nonspecific response to sepsis, toxemia, or disease of another organ system.
Under normal circumstances a large volume of essentially isotonic fluid enters the proximal bowel daily. Most of this fluid is resorbed, and only a small percentage is passed with the feces. The fluid comes from dietary intake and from endogenous secretions of the upper digestive tract. The total daily volume exchanged exceeds that of the animal’s total extracellular fluid volume. Normally absorption just exceeds secretion; therefore very small changes in rate of absorption or secretion can result in diarrhea. In the horse most water resorption occurs in the cecum and large colon, and diarrhea in the horse (other than neonates) usually involves some abnormality in the lumen or wall of the large bowel.
Normal fecal color is tan, brown, or greenish, depending on diet. The adult horse normally produces 11 to 13 kg of fecal material per day (20 to 28 g/kg of body weight per day) while on a diet of grass hay and 3 lb of oats.1 Fecal output was as high as 20 kg/day in horses fed a mixture of alfalfa and orchard grass ad libitum.2 Horses with chronic watery diarrhea can produce up to 214 g/kg of body weight per day,1 which is over 90 L of diarrhea in a 450-kg horse. Cattle normally produce 15 to 28 kg of feces per day with a water content of about 75% to 85% on a diet of grass hay.3 Fluidity of cattle feces can increase markedly in animals on lush green feed. Sheep and goat feces contain only 50% to 60% water.3 Fluid feces in the cow and unformed feces in the horse, goat, and sheep are very nonspecific signs that often accompany sepsis or illness other than a primary gastrointestinal disease.
Animals with chronic diarrhea rarely develop severe dehydration because they compensate for increased fecal water losses by increasing water consumption by an equivalent amount. Normal water consumption in the horse on a hay diet in a mild ambient temperature environment is about 24 to 30 L/day, whereas cattle consume 30 to 60 L/day based on 10 kg of dry matter feed intake. Water and feed intake do not increase linearly with increasing body size, but by multiplying body weight in kilograms to the 0.75th power (BW0.75kg) by a base factor (about 200 mL for water). Thus a 500-kg horse requires 21 L of water. The effect of temperature on water intake is dramatic and is not linear; as ambient temperatures rise close to 37° C (98° F), water intake per kilogram of dry matter increases much more rapidly than at lower temperatures. Exercise and loss through sweat, particularly in the horse, can dramatically increase salt and water requirements.
Mechanisms of Diarrhea (Box 7-1)
The following six major mechanisms produce diarrhea:
 Abnormal intestinal motility resulting in decreased transit time, as with systemic inflammatory response syndrome (SIRS)
Abnormal intestinal motility resulting in decreased transit time, as with systemic inflammatory response syndrome (SIRS) Increased blood-to-lumen pressure as in heart failure or acute or chronic inflammatory bowel diseases
Increased blood-to-lumen pressure as in heart failure or acute or chronic inflammatory bowel diseasesThe common net result is an increase in fecal water.
Decreased surface area, is mainly a result of villus blunting (atrophy) and/or microvillus damage in the small intestine, which lead to malabsorption. Both occur to some degree with most enteric diseases, and regeneration of surface area from crypt cells with healing is accompanied by a gradual decrease in volume of diarrhea. Diseases in which this is a major mechanism include neonatal diseases such as rotavirus and coronavirus enteric disease,4 cryptosporidiosis, acute inflammatory disease such as salmonellosis, and chronic diseases such as Johne’s disease and other granulomatous bowel diseases. The finding of villus atrophy is so nonspecific that it is not diagnostic in itself. It can also occur in advanced cases of secondary copper deficiency (molybdenosis) with diarrhea. Loss of villus epithelial cells can result in maldigestion because these cells produce important enzymes such as lactase. Many neonates with enteritis develop temporary lactose intolerance as a result, especially with rotaviral and clostridial infections.
Inflammation, can be accompanied by increased mucus production and increases in size of membrane pores, through which tissue fluids and serum proteins leak into the lumen. This is associated with increased capillary and lymphatic hydraulic pressures. Whether acute (Salmonella), or chronic (Johne’s), inflammatory bowel diseases are protein-losing enteropathies. Low plasma protein concentrations, particularly low albumin, are often found (unless hypovolemia and hemoconcentration are present). Bowel inflammation often results in transudation and exudation of serum proteins, blood, and/or mucus, resulting in dysentery (bloody diarrhea). In addition to salmonellosis, dysentery may also commonly be seen with enterotoxemia caused by Clostridium perfringens, type A, B, or C, Clostridium difficile, Lawsonia intracellularis,5 attaching effacing Escherichia coli, Campylobacter jejuni, coccidiosis, malignant catarrhal fever (MCF), arsenic toxicity, and oak toxicity. Inflammation results in malabsorption, maldigestion, osmotic effects, and, in acute disease, changes in intestinal motility. Because most water absorption in the horse occurs in the cecum and colon, inflammatory typhlitis and colitis are the major causes of diarrhea in the horse (Boxes 7-2 and 7-3). The neonatal foal commonly develops small intestinal enteritis.
Box 7-2 Causes of Diarrhea in Horses (Except Neonates; see Chapter 19 for Neonates)
Irritation of the bowel with a foreign body such as sand may result in either low-grade recurrent colic or diarrhea. Weight loss may also be evident with a large amount of sand. Sand accumulation in the large bowel of the horse may be suspected when there is evidence of a significant amount of sand in the feces or when sand is auscultable with a stethoscope over the ventral abdomen. Irritation probably causes diarrhea through creation of an inflammatory response and altered motility.
Osmotic diarrhea, results from any disease causing maldigestion and/or malabsorption. Any osmotically active solute can produce diarrhea in normal animals if given in quantities sufficient to surpass the intestinal capacity for digestion or absorption. Disaccharides are natural examples. Osmotic cathartics such as dioctyl sodium sulfosuccinate (DSS) hold water in the intestine and act as fecal softeners. Magnesium phosphates and sulfates and other divalent and trivalent cations and anions are poorly absorbed and thus are effective laxatives and cathartics.
Osmotic diarrhea can be associated with ingestion of osmotically active poorly absorbed solutes, overloading of the intestine with carbohydrates or lipids beyond the amount that can be digested and absorbed, sudden dietary changes resulting in marked shifts in gut flora and resulting bacterial action on ingested substrate (e.g., grain overload), or bowel disease in which surface area is diminished or digestion interfered with in some manner. Lactase deficiency, secondary to rotavirus or C. difficile, infections, may result in osmotic diarrhea in foals.6 This results in increased concentration of undigested and/or unabsorbed nutrients entering the lower bowel, increased bacterial fermentation, and an increase in the concentration of osmotically active particles. Unfavorable electrochemical gradients prevent resorption of water. Mucosal digestive enzyme levels are often decreased with any disease involving the small intestine, resulting in maldigestion. When osmotic diarrhea is suspected in mature animals, dietary modification to basic roughage should be tried as part of the nonspecific therapy. Sodium and potassium are normally present in roughly equal amounts in feces and (with a little ammonium) make up the vast majority of cations in the feces. Concentrations of sodium and potassium in feces and osmotically active nonelectrolytes influence fecal water. In general, osmotic diarrheas diminish when the animal is fasted. When the offending substance is reintroduced, diarrhea occurs.
Secretory diarrheas, are most important in neonates4 (enteropathogenic E. coli,), but many strains of Salmonella, associated with colitis in large animals may produce enterotoxins that stimulate secretion. Enterotoxins act by stimulating cyclic adenosine monophosphate (AMP) or other intracellular messengers to promote secretion of chloride, sodium, and other electrolytes into the gut lumen. Water is carried with these electrolytes and osmotically retained. The hallmark of secretory diarrheas is the large volume of feces produced.
Examples of secretory diarrheas are enterotoxigenic E. coli, and many strains of Salmonella, and C. perfringens. Salmonella, and other invasive organisms produce inflammation that may induce prostaglandin-mediated secretion as well. Secretion may occur with viral diarrhea by a different mechanism, as damaged mature (absorbing) villus cells are replaced by immature (secreting) crypt cells.7 A good example is rotavirus.
Decreased intestinal transit time, associated with increased peristalsis and/or decreased segmentation appears to occur in many bowel diseases because of bowel irritation. Peritonitis, is a major cause of bowel inflammation and should always be explored as a contributing cause of diarrhea, especially when fecal output volume is scant. Abnormal motor patterns have been demonstrated to occur with many infectious diarrheas and may be a bowel response to irritation and/or increased intraluminal volume. Elimination of gut contents thus appears to be a normal gut defense mechanism against infection and probably should not be pharmacologically alleviated in acute infectious diarrheas. Primary motility disorders of animals are not well recognized; diarrhea associated with nervous or excited animals may be the best example of this type. In general, fecal volume associated with motility disorders is not great.
Increased hydraulic pressures, from the blood to the lumen also decrease net absorption of fluid. These can result from decreased oncotic pressure (hypoalbuminemia), increased capillary hydrostatic pressure (heart failure or portal hypertension as with liver disease), or decreased lymphatic drainage associated with inflamed or blocked lymph vessels or nodes (lymphosarcoma). These mechanisms are most commonly associated with chronic diarrhea, but acute inflammation can also result in diarrhea associated with this mechanism.
Two or more of these mechanisms are probably at work in most diarrheal diseases. Therapy of diarrhea is therefore nonspecific, except when the actual causative agent can be identified. Diagnosis of a specific causative agent is most important when diarrhea is caused by an infectious agent, so that appropriate therapeutic steps can be taken before chronicity develops, spread of disease can be prevented, and an accurate prognosis can be made.
In mature horses, small intestinal diseases such as granulomatous bowel disease or duodenitis or proximal jejunitis (anterior enteritis) may not be associated with diarrhea, and diseases of the stomach almost never cause diarrhea. Most significant diarrheal disease in adult horses involves the large colon because this is the principal site of water absorption. The exception to this is the neonatal foal, in which primarily small intestinal diseases such as rotavirus infection and cryptosporidiosis may cause severe diarrhea.
The frequency of defecation is usually increased when diarrhea is present, and defecation is most frequent when the colon or rectum is irritated. When these areas are involved, tenesmus, (straining) may result. Tenesmus can also occur with hepatic failure in ruminants and in horses and ruminants with rectal tears or strictures, vaginitis, retained placenta, dystocia, intussusception, urolithiasis, rabies, and diseases involving the nervous system when there is retention of feces or urine. Severe rectal irritation can lead to straining and rectal prolapse.
In ruminants, abnormalities such as grain overload (toxic indigestion) resulting in ruminal osmotic changes can produce diarrhea, as can changes in abomasal pH such as occur with type II ostertagiasis. Diarrhea in ruminants is frequently caused by forestomach problems (Boxes 7-4 and 7-5). The colon and remainder of the distal bowel are involved in diseases such as salmonellosis. Gram-negative infections and resulting endotoxemia, found in conditions such as coliform mastitis and septic metritis, are relatively common causes of nonspecific diarrhea. Foals with septicemia also commonly develop nonspecific diarrhea associated with SIRS. Diarrhea may be (1) a manifestation of a primary disease (bovine viral diarrhea [BVD]; Johne’s disease; C. difficile,), (2) one of the signs of a generalized disease (MCF, uremia), or (3) secondary to toxemia (coliform mastitis, septic metritis, septicemia).
Box 7-4 Causes of Diarrhea in Ruminants (Except Neonates; see Chapter 20 for Neonates)
Nonspecific Fluid Therapy for Diarrhea
Dehydration, electrolyte losses, and acid-base abnormalities can occur rapidly when diarrhea is present. Symptomatic treatment to correct these problems is an important component of nonspecific therapy in animals with diarrhea. Very often the cause of diarrhea remains undetermined, yet symptomatic correction of hypovolemia, dehydration, and acid-base and electrolyte abnormalities can result in a return to normal function, particularly if the diarrhea is acute and severe. Fluids and electrolytes can be given orally or parenterally. Oral fluids can be given rapidly and inexpensively. Oral fluids should be isotonic or hypotonic. The degree of dehydration should be estimated as a percentage of body weight. Mild dehydration is usually considered less than 5%; moderate, 5% to 8%; and severe, over 8%. Thus a severely dehydrated 450-kg patient with an estimated 10% dehydration (weakness, cold extremities, sunken eyes, decreased urine output, decreased elastic skin rebound, weak pulse, rapid heart rate) requires 45 L of fluids.
The best way to determine electrolyte needs is to take a plasma or serum sample before initiating fluid therapy. Electrolyte requirements can be estimated; with diarrhea, mixed water and electrolyte losses occur, so that sodium-containing fluids are usually required to replace lost sodium and improve blood volume (see also Fluid and Electrolyte Balance, Chapter 22). Unless the acid-base status can be measured, the safest sodium-containing fluids are balanced polyionic fluids such as Ringer’s or lactated Ringer’s solution. Normal saline is a satisfactory alternative in most cases, but the relatively high concentration of chloride ions in saline can aggravate a preexisting metabolic acidosis unless hypochloremia is also present. In calves with severe metabolic acidosis, use of fluids containing sodium bicarbonate is indicated. In horses, it is best not to include gram quantities of sodium bicarbonate in fluids unless there is good evidence that a severe metabolic, strong ion acidosis exists (such as hyperchloremia). Flow rates for administering isotonic intravenous fluids should be kept as slow as possible to avoid fluid overload, pulmonary edema, and excessive diuresis. When the patient is shocky, flow rates close to 20 mL/kg/hr or faster may be required for administration of isotonic crystalloid fluids, but in general rates below 10 mL/kg/hr are desirable, especially once hypovolemic shock has been addressed. Hypertonic saline (7% NaCl) may be given rapidly intravenously at a dose of 4 to 5 mL/kg. When a colloid fluid is required to combat low plasma protein levels, hetastarch can be used. It is available as 6% hetastarch in lactated Ringer’s or normal saline. See Neonatal Diarrhea, Chapter 20, Fluid and Electrolyte Balance, Chapter 22, and Fluid Therapy, Chapter 44, for more details on fluids and acid-base balance.
Approach to Diagnosis of Diarrhea in the Horse (for Neonates, see Chapter 19)
Approach to Diagnosis of Diarrhea in Ruminants (for Neonates, see Chapter 20)
COLIC
Colic, is defined as the manifestation of visceral abdominal pain. Colic may be the result of nongastrointestinal pain, such as urinary tract obstruction (Boxes 7-6 and 7-7). This section discusses colic of gastrointestinal origin. Pain may be acute, chronic, or recurrent. Gas distention or recognizable organic problems such as displacements are most frequently associated with acute colic. Five basic causes in large animals are as follows:
The horse exhibits colic frequently, because it appears to have a low threshold for pain and is frequently beset by minor digestive disturbances that result in distended bowel. Signs of colic in the horse include restlessness, lying down and getting up, groaning, grunting, rolling, sweating, kicking at the abdomen, or suddenly dropping to the ground in pain. Anorexia and depression often accompany these signs. Horses may develop tachycardia with weak pulse quality and prolonged capillary refill time, cold extremities, and bright red (vasodilatory phase) followed by dark (vasoconstrictive phase or low cardiac output state) mucous membranes if the problem is severe and affects cardiovascular integrity.
Evidence of shock is usually present only when the disease condition is severe and involves infarctive disease (volvulus, torsion, thromboembolism) or advanced visceral distention (extreme flatulence, impaction, or dilation). Foreign bodies such as sand or enteroliths in the large colon may result in low-grade recurrent colic., When an enterolith is passed into the transverse colon where it obstructs the bowel, signs of complete obstruction, and acute colic ensue. These signs are manifestations of visceral pain, mediated by the sympathetic nervous system. External palpation and pushing on the abdomen of adult horses does not usually elicit pain unless the affected inflamed or dilated viscus is contacted.
In contrast, parietal pain associated with peritonitis, usually is responsive to external palpation. Whereas the animal with visceral pain shows active signs of colic, the animal with parietal pain is usually reluctant to move and has a splinted abdomen. Acute peritonitis occurs within minutes after rupture of the stomach or perforation of an ulcer as a result of immediate irritation of serosal surfaces by the acid contents, whereas with rupture of the colon or rectum it may take 12 hours or more before peritonitis is clinically apparent.
Ruminants exhibit colic less frequently than horses, probably because of a higher threshold for pain and because dietary shifts principally affect the rumen; thus intestinal gas pains occur less frequently. Colic with visceral pain (including urinary tract) is manifested by grinding the teeth (odontoprisis or bruxism), grunting or groaning, treading with the hind feet, kicking at the abdomen, restlessness, repeatedly lying down and getting up, anorexia, and depression. With parietal pain caused by peritonitis, ruminants demonstrate abdominal pain by arching the back, splinting, and exhibiting pain on deep palpation of the area. These signs are seen most commonly with traumatic reticuloperitonitis and abomasal ulcers; rarely are these diseases associated with the colic signs described previously.
Diagnosis and Management of Colic in the Horse
Recurrent mild-to-moderate colic may, be an indication of a more serious problem such as bowel entrapment or displacement thromboembolism, internal abscess, enterolith, sand or other foreign body, tumor, gastric ulcer, hypobiotic cyathostomiasis, Strongylus vulgaris, larval migration, heavy burden of ascarids, abdominal adhesions, strictured bowel, or urinary tract disease.
If no feces are passed, a more serious condition is usually indicated, particularly if intestinal sounds are absent.
Abdominal fluid is normally present in small volume (a few drops to 50 mL); is clear and colorless to yellow; and has a total protein below 2.5 g/dL, specific gravity less than 1.015, little or no fibrinogen (<100 mg/dL), and fewer than 5000 white blood cells (WBCs) per microliter. Elevations in these parameters or increased numbers of neutrophils in peritoneal fluid often indicate inflammation on serosal surfaces. Comparisons of abdominal fluid glucose with serum glucose (greater than 50 mg/dL difference is abnormal) and evaluation of abdominal fluid pH, lactate (as compared with blood lactate), and lactate dehydrogenase (LDH) concentrations can aid in the diagnosis of septic peritonitis.28 Grossly, fluid changes to cloudy yellow, then to blood-tinged with fibrin clots, and finally to black in color as bowel necrosis and hemolysis of extravasated red blood cells (RBCs) occur. Elevated levels of peritoneal fluid protein are consistently found in anterior enteritis (duodenitis and proximal jejunitis) and provide a useful differential aid, although this finding is also common in cantharidin poisoning. Elevated protein levels are present in peritonitis but accompanied in these cases by elevated numbers of neutrophils. If peritoneal fluid is grossly contaminated with feed material, rupture of a viscus should be considered. Another peritoneal tap in a different location should be undertaken if the possibility of bowel penetration during paracentesis cannot be ruled out. It is not necessary to have abnormal peritoneal fluid before consideration of surgical correction of colic is contemplated. Recently, abdominal fluid lactate has been shown to be predictive of intestinal ischemia secondary to strangulating obstruction and may aid in early detection of intestinal strangulation, rupture, and septic peritonitis.29
Many chemical analyses (e.g., lactate) of blood or peritoneal fluid have been recommended as an aid in prognosis and gauging severity of tissue damage. Plasma fibrinogen, as well as peripheral leukocyte counts, can aid in the differentiation of enterocolitis or anterior enteritis from strangulating lesions. Serum electrolyte concentrations and acid-base status are important from a therapeutic standpoint. Serum biochemistries, including liver enzymes, creatinine, and BUN, are important in evaluating for liver and renal compromise. Hyperglobulinemia may be indicative of chronic disease.
Large amounts of painful gas auscultable in the right flank can be tapped and drawn off, although the risk for peritonitis should be considered. The cecum is trocarized, using a 14- to 16-gauge 6-inch needle inserted through aseptically prepared skin.
The principles of colic management include (1) control of pain, (2) relief of distention, (3) relief of obstruction, and (4) reversal of shock.
Surgery may be contraindicated in the following situations:,
Diagnosis and Management of Colic in Ruminants
It should be determined whether the onset of colic was acute or whether the colic is chronically recurrent. Few diseases cause recurrent colic in ruminants. Urolithiasis is probably the most common cause of colic in male or neutered male goats and sheep and occurs under varied dietary and environmental conditions. Urolithiasis in cattle occurs most frequently in bulls or steers eating high-grain diets, but silicate stones can occur in very young animals and animals on pasture diet.
Ultrasound examination of the abdomen is also useful (see Chapter 32).
Radiology and ultrasound can be useful in sheep and goats when urolithiasis is a consideration. Ultrasound can detect a distended bladder, and stones may sometimes be detected. Radiology (lateral view) may detect a stone in the urethra or stones in the bladder. A contrast urethrogram may also be diagnostic.
MELENA
Melena (dark, tarry feces) is caused by blood in the lumen of the stomach or proximal intestinal tract, resulting in black (digested) blood appearing in the feces (Boxes 7-9 and 7-10). Usually blood is a result of a bleeding ulcer in the stomach or abomasum but may also result from ingestion, oral or pharyngeal bleeding, or coughing up blood that is then swallowed. In ruminants the presence of dark red feces from an intussusception is the main differential to be considered. Blood must stay in the intestinal tract for hours before the hemoglobin is altered and turns black. Small amounts of hemoglobin can be detected by using one of the tests for occult blood. In general, fairly large volumes of blood (1 to 2 L) are required to produce a positive fecal occult blood test in the horse.* A 24- to 48-hour time period is needed for orally administered blood to reach the rectum in the horse. In ruminants, smaller volumes of blood are needed to produce a positive fecal occult test, and a faster transit time is expected.
In approaching a diagnosis, rule out pulmonary, oral, or pharyngeal bleeding. Bleeding of gastrointestinal origin can be determined to be caused by mucosal disease or full-thickness bowel disease (such as an intussusception or neoplasia) by examining peritoneal fluid for abnormalities. Abnormalities in peritoneal fluid are usually present in the case of serosal involvement. Bleeding abomasal ulcers are probably the leading cause of melena in ruminants. They can be silent except for the dark feces and weakness if severe anemia develops. In older horses, gastric squamous cell carcinoma is a frequent cause of gastric hemorrhage. Significant bleeding is much less common in foals and calves with gastric ulcers, and melena is rare in foals and calves with gastric ulcers.
Consideration should be given to whether or not the melena is the result of clotting abnormalities associated with such diseases as DIC or warfarin poisoning. In cattle with colic and dark red-to-black feces, intussusception should be considered likely.
When severe anemia develops, there is evidence of blood loss because the decrease in PCV and RBCs is accompanied by a decrease in plasma proteins. Nonspecific therapy for melena consists principally of blood transfusions in life-threatening cases. Sudden, massive gastric or abomasal bleeding may result in anemia and collapse before melena has appeared.
In the foal, gastric ulcers may be treated with histamine-2 (H2) blockers such as ranitidine or cimetidine. These drugs are probably less effective in ruminants. Their benefit in ruminants with abomasal ulcers is not well understood at present. New drugs such as the hydrogen pump blocker omeprazole are useful and potent gastric pH effectors. Therapy with protectants such as sucralfate (which coats the ulcer) is a viable and clinically useful therapy in the horse. In ruminants, orally administered protectants and antacids are so diluted by the time they reach the abomasum that they are probably of limited benefit.
BLOOD, FIBRIN, AND/OR MUCUS IN FECES (DYSENTERY)
Bloody diarrhea is termed dysentery., The presence of fresh blood or clots in the feces is termed hematochezia, and is the result of bleeding into the distal intestinal tract. Occasionally blood from the female reproductive tract may appear in or on the feces. Fibrin indicates severe inflammatory bowel disease. Fibrin appears as casts, chunks of yellow-gray material, or mucosa-like sheets. Mucus in feces increases with inflammatory bowel diseases such as salmonellosis. It is often seen when fecal volume is small in animals that are anorectic, in which case the feces are often coated with mucus. This mucous coating can become very obvious in the horse and is not a sign of bowel disease in this case.
Frank blood in feces without diarrhea and other evidence of gastrointestinal dysfunction or systemic illness may be a result of a bleeding disorder, a traumatic foreign body, rectal examination trauma, sadistic rectal trauma, or rectal trauma in a mare from a stallion penetrating the rectum (Boxes 7-11 and 7-12). Many of the diseases listed as causes of melena may also result in gastrointestinal hemorrhage and are therefore listed in both places. If the bleeding is in the distal gastrointestinal tract, fresh blood may be seen in the feces. With diseases midway down the tract, such as intussusception, fecal material is dark red and may appear black until a sample is examined closely and spread on a white surface.
ABDOMINAL DISTENTION AND CONSTIPATION
Abdominal distention may be caused by feed, fluid, gas, feces, or a neoplasm (Boxes 7-13 and 7-14). Pregnancy or extreme obesity may also result in an enlarged abdomen. The physical examination should determine which of these is the most likely cause. Often in ruminants the distention can be seen as primarily left sided, right sided, or bilateral. For example, bloat in ruminants results in a characteristic high left-sided gas distention. It may be primary or associated with vagal indigestion, tetanus, or hypocalcemia. With vagal indigestion the rumen becomes enlarged and fluid-filled, often giving a pear shape to the abdomen as it is viewed from the rear, or a pear shape on the right and an apple shape on the left (“papple” shape) if some degree of bloat is also present. Hypocalcemia and hypokalemia contribute to ileus and may result in constipation and abdominal enlargement. In sheep, abomasal impaction and enlargement associated with abomasal emptying defects can result in an enlarged abdomen with decreased food intake. When a mass (most commonly an abscess, a tumor, or a fat necrosis [cattle only]) obstructs fecal passage, abdominal enlargement can become severe. With obstructive disease, some degree of colic is almost always present. Ruptured bladder results in a large fluid-filled abdomen, but constipation is not an obvious sign.
Box 7-14 Causes of Abdominal Distention and Constipation in Ruminants
The most common causes of decreased fecal output in ruminants and horses are decreased feed intake and dehydration. In such cases the animal will appear gaunt or have a relatively empty abdomen or rumen. Horse feces in cases of prolonged transit are often covered with a layer of tenacious, thick, yellow mucus. When a functional obstruction (ileus, vagal indigestion) or physical obstruction (impaction, foreign body, displaced intestine, fat necrosis) occurs as a cause of constipation, the abdomen is more likely to appear normally full or to become distended. Rectal examination is of great help in determining whether a mass or an obstruction exists because loops of distended small bowel can sometimes be palpated in the latter case.
Radiographs and ultrasound may be valuable to help determine the cause of abdominal distention in foals, calves, and small ruminants. Increased gastrointestinal gas may result in abdominal distention. Abomasal bloat and necrotizing enterocolitis in young animals may best be confirmed with lateral abdominal radiographs.
Dehydration may also result in dry feces but not in abdominal enlargement. When constipation is present and feces are drier than normal, rehydration and correction of hypocalcemia, hypokalemia, and any existing acid-base abnormalities are important parts of correction of the constipation. Other nonspecific therapies for functional constipation include laxatives, cathartics, and cholinergic drugs. When treating constipation, which is usually a secondary problem, it is important to simultaneously attempt to diagnose the primary disease.
In ruminants, when abdominal distention involves the rumen or is caused by pregnancy or obesity, colic is absent. When abdominal distention is the result of obstruction from the pylorus distal, colic is usually present. Abdominal distention and constipation are frequently accompanied by colic in the horse, regardless of anatomic site involved (review the approach to colic).
REGURGITATION AND VOMITING
Regurgitation is the reflux of esophageal, gastric, or rumen contents into the mouth or nose. This may be caused by malfunction of the esophagus or in ruminants as part of the normal physiology for rechewing ingested plant fiber (Boxes 7-15 and 7-16). Vomiting is a coordinated, centrally (medulla) mediated event, usually preceded by nausea (inappetence), increased salivation, or retching. In vomiting the abdominal musculature contracts, the diaphragm is pushed caudally, and the cardia relaxes. The medullary vomiting center can be stimulated by visceral afferent stimuli or through the chemoreceptor trigger zone. Most toxins and drugs that cause vomiting act by directly affecting the chemoreceptor trigger zone. Other than with toxins, most cases of feed returning to the mouth in large animals are examples of regurgitation rather than true vomiting. Vomiting is unusual in both ruminants and horses.
Although regurgitation is a normal phenomenon in ruminants, it is unusual to find excessive regurgitation as a sign of disease. Physical blockage of rumenoreticular outflow by a foreign body, warts, granulomas, or diaphragmatic hernia can cause rumen distention and excessive regurgitation after eating. An esophageal foreign body can cause irritation and result in regurgitation. Animals with facial paralysis may drool feed and saliva on the affected side; this should be differentiated from animals with excessive or abnormal regurgitation. Vomiting or forced regurgitation in ruminants is rare and is seen principally with the toxins listed.
Horses have such a marked tone at the cardiac sphincter that vomiting occurs only when extreme intragastric pressures develop, usually in small intestinal obstructive diseases or proximal enteritis. Vomiting in the horse thus often occurs with gastric rupture or terminally with shock. Stomach contents are usually pH 5 or below. Because it is a terminal event, vomiting in the horse is often grounds for rendering a poor prognosis. Abdominocentesis should be performed on a horse after vomiting to rule out gastric rupture. To avoid this sequence of events, decompression using a nasogastric tube should be performed in any horse with evidence of gastric distention (see approach to colic). Regurgitation and vomiting in horses most commonly occur from the nose rather than into the mouth, because of the anatomy of the soft palate. With choke (esophageal obstruction), esophageal regurgitation from the nares consists of mixed feed and saliva.
In foals a few weeks to several months of age, milk returning from the nares is often associated with gastric ulceration, along with signs of colic, lying in dorsal recumbency, hypersalivation, and champing movements of the mouth. In advanced cases with duodenal ulcers, pyloric outflow can be obstructed by scarring, resulting in more pronounced signs. Foals 1 to 6 months of age are most susceptible to gastric ulceration.
Occasionally, neonatal foals without cleft palate have some mild degree of dysphagia with milk regurgitation from the nose for the first 24 to 48 hours of life, which spontaneously corrects. The cause of this is unknown, but it would appear to be a failure of normal swallowing events to be sufficiently strong or coordinated in the newborn. The major ruleout in these cases is cleft palate.
Approach to Diagnosis of Regurgitation and Vomiting
Evaluation of regurgitation or vomiting should include a history to determine possible exposure to toxins or poison plants, which is most likely when multiple animals are affected. Age of the animal limits some considerations; young animals are more prone to meningitis and central nervous system (CNS) trauma, and congenital problems such as esophageal diverticula and persistent right aortic arch are found only in neonates and may not manifest as choke or regurgitation until solid food intake is increased.
The physical examination can determine whether the problem is vomiting or regurgitation. In ruminants, regurgitation often occurs as a result of distention and overfilling of the rumen, resulting in an obviously distended abdomen. Painful pharyngeal lesions can also cause pharyngeal paresis, which results in gagging and regurgitating. In horses the most common causes of feed coming from the nares are spontaneous choke and pharyngeal paresis associated with guttural pouch lesions (see section on dysphagia).
Physical examination should also include passing a stomach tube to determine whether any impediment to passage of ingesta is present (Box 7-17). Endoscopy is useful to visualize esophageal defects. Many endoscopes currently in use are not long enough to reach the stomach of the adult horse. Endoscopy of the rumen is rarely diagnostic, because it is almost impossible to empty it adequately to allow for visualization of a lesion. Ultrasound of the cervical esophagus may also be useful.
In horses and small ruminants radiography, particularly barium contrast studies, can be useful in detecting esophageal abnormalities. In foals, prolonged gastric emptying time may be diagnosed from contrast studies. Normal emptying and movement of contrast media into small bowel occur in less than 2 hours; contrast media reach the large bowel by 3 hours. Radiography may also be useful in detecting diaphragmatic hernia.
The most significant complications of regurgitation and vomiting include aspiration pneumonia, dehydration, and electrolyte imbalances. The marked hypochloremic alkalosis common to most monogastrics is rare in horses and occurs in ruminants mainly with internal vomiting (one type of vagal indigestion) associated with reflux of abomasal contents back into the rumen.
Vomiting, like diarrhea, is often an attempt by the body to rid itself of a noxious or toxic substance. Antiemetics are therefore rarely indicated in vomiting of central origin and rarely effective in regurgitation in large animals.
DYSPHAGIA (INCLUDING FEED FROM NARES AND EXCESSIVE SALIVATION)
Dysphagia, is used here to refer to abnormalities of prehension, mastication, or swallowing. It is associated with diseases of the mouth, lips, pharynx, esophagus, mandible, or masseter muscles or, in the case of neurologic problems, with central or peripheral lesions resulting in malfunction in these areas. Diseases resulting in erosions, ulcers, swellings, crusts, or growths in or on the lips, mouth, or pharynx are discussed under a separate heading. Painful causes of dysphagia such as dental problems require differentiation from oral lesions such as ulcers.
The causes of dysphagia can be divided into three categories: (1) pain induced, (2) neurologic, and (3) obstructive (Boxes 7-18 and 7-19). A fourth category is mechanical interference with prehension and swallowing, but this usually manifests in a manner resembling the manifestation of painful lesions. Particularly in horses, worn, missing, capped, abscessed, overgrown, or broken teeth often result in mechanical interference with chewing, resulting in half-chewed feed being dropped from the mouth (quidding). Observation of the animal as it attempts to eat and a good physical examination, including oral inspection and passage of a stomach tube to rule out choke, are essential in determining the cause of dysphagia. Use of a fiberoptic endoscope to visualize the pharynx, guttural pouches, and esophagus may be helpful. Plain film radiographs and barium swallows may also be indicated to see functional abnormalities in the pharynx and esophagus during swallowing and to rule out fractures of the hyoid or mandible. Ultrasound should also be employed.
Pain is probably the most frequent cause of dysphagia in ruminants and horses. Oral lesions, oral foreign bodies, and poor teeth result in decreased feed intake, in increased salivation, and often in dropping feed from the mouth while attempting to chew. Dental problems are relatively common in sheep and goats. In cattle, pharyngeal injuries from balling guns and paste wormers can result in severe pharyngeal cellulitis, which is manifested by an extended head, ptyalism, foul breath, and a painful, externally palpable, pharyngeal swelling. Mandibular fractures must be ruled out by careful examination, because even nondisplaced unilateral mandibular fractures can result in weak jaw tone, reluctance to eat, and drooling.
When dysphagia is associated with loss of large amounts of saliva, metabolic acid-base and electrolyte disorders may develop. Cattle and sheep have saliva high in sodium (136 to 201 mEq/L) and bicarbonate (108 mEq/L), with potassium and chloride values in the 14 to 15 mEq/L range.30,31 As a result, losses of large amounts of saliva can result in hypovolemia and severe metabolic acidosis. In contrast, horses have relatively high levels of salivary chloride (48 to 82 mEq/L) with relatively low salivary bicarbonate (44 to 52 mEq/L). Equine salivary potassium is 14 to 18 mEq/L, and sodium 54 to 90 mEq/L.32,33 As a result, horses with esophageal fistulas that lost saliva had a transient metabolic alkalosis.32
In the horse a common cause of acute dysphagia is choke (esophageal obstruction), followed in frequency by pharyngeal paresis (neurologic) resulting from lesions in the guttural pouch that affect the pharyngeal nerves. Feed coming from the nose is the most obvious sign of both of these conditions. In choked horses, as a result of the length and position of the soft palate, feed comes mainly from the nares rather than coming back into the mouth. Choke and other obstructive diseases can be easily identified by using a nasogastric tube, whereas pharyngeal paresis may be associated with a number of neurologic or neuromuscular conditions, such as botulism or guttural pouch mycosis, which require careful differentiation. The most frequent serious problem associated with choke or pharyngeal paresis is inhalation (aspiration, foreign body) pneumonia. Mineral oil or other material that is particularly damaging if it gains entry into the lung should never be used in choke for this reason. In any animal with dysphagia, care must be taken to prevent aspiration pneumonia and to evaluate the thorax periodically.
Animals with facial paralysis often drool from the affected side and may pack feed into the cheek on the affected side. Listeriosis in ruminants is frequently associated with facial paralysis. In horses facial paralysis is usually caused by halter trauma or a blow to the head.
ORAL VESICLES, EROSIONS, ULCERS, OR GROWTHS
Oral lesions are found with many conditions (Boxes 7-20 and 7-21). In general, they result in some degree of dysphagia or reluctance to eat because of pain. The lesions include vesicles, erosions, ulcers, crusts, or growths in or on the lips, tongue, gums, palate, or pharynx. Oral lesions are often associated with champing and increased amounts of saliva being observed on the lips or running from the mouth. When the volume of saliva is increased, the condition is called ptyalism, and the animal may be observed swallowing repeatedly. Pseudoptyalism, refers to a normal volume of saliva that because it is not swallowed is visible to the observer and may be confused with dysphagia (see previous section).
Box 7-20 Conditions Accompanied by Oral Vesicles, Erosions, Ulcers, or Growths in Horses
Box 7-21 Conditions Accompanied by Oral Vesicles, Erosions, Ulcers, or Growths in Ruminants
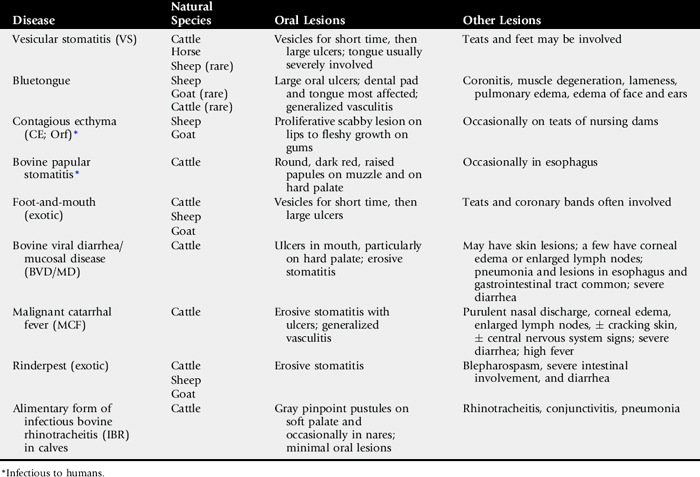
The approach to determining the cause of oral lesions is based on first determining whether the cause is likely to be an infectious disease (Table 7-1). Essentially all these infectious diseases are associated with a fever, although it is short-lived and moderate in the case of bovine papular stomatitis (BPS) and actinobacillosis. Papular stomatitis rarely causes illness and is usually an incidental finding in calves with a different clinical problem. Most of the infectious diseases are associated with additional lesions or symptoms. They can be conveniently grouped into those causing diarrhea and those not usually associated with diarrhea. Of those not associated with diarrhea in North America, BPS, actinobacillosis, and vesicular stomatitis (VS) are most common in cattle; VS in horses; bluetongue in sheep; and contagious ecthyma (CE) in sheep and goats. CE can be readily differentiated from bluetongue because it involves primarily lips and gums and is proliferative, whereas bluetongue involves the tongue and dental pad most severely, is erosive, and is associated with other signs of generalized vasculitis. Laboratory diagnosis in acute cases of VS is done by working with state and federal veterinarians. Because VS is highly contagious and similar in clinical appearance to foot-and-mouth disease, quarantine and proper diagnosis are essential. Bluetongue is diagnosed by serology (AGID), PCR, and virus isolation. CE can be diagnosed serologically, by fluorescent antibody on the impression smear or biopsy of a lesion, and by isolation. Asymptomatic seroconversion to bluetongue is common where Culicoides, vectors are active. Congenital defects can result from bluetongue infection of the fetus in sheep, goats, and cattle.
The two most common North American infectious diseases associated with oral lesions and diarrhea in cattle are bovine viral diarrhea/mucosal disease (BVD/MD) and MCF. MCF can usually be differentiated because it most commonly occurs sporadically in single animals and has signs of generalized vasculitis such as bilateral corneal opacity, mucopurulent nasal discharge, enlarged lymph nodes, and very high fever. Dysentery is common in MCF, and some animals exhibit CNS signs or have thickened and cracking skin. Laboratory diagnosis in acute cases of BVD (see Chapter 32) involves fluorescent antibody testing of slides made from ear notch biopsies, lesion swabs, buffy coat, or tissue. Virus isolation from swabs, serum, or blood or a rise in serum titer from acute to convalescent samples is also diagnostic. Asymptomatic seroconversion is also common, and infection of the fetus may result in congenital anomalies, including cerebellar hypoplasia in cattle.
In animals without fever and other signs of systemic involvement, irritants and caustic chemicals should be considered as possible causes of oral lesions. Horses and young calves are susceptible to severe ulceration when consuming hay contaminated with yellow bristle grass, which is armed with barbed bristles.34 Horses sometimes develop gingivitis or oral ulcers associated with dry plant awns called foxtails, which become embedded into the gums around teeth. Foals and ponies are most susceptible to phenylbutazone toxicity, which can produce oral ulceration. In cattle the surfaces of masses produced by actinobacillosis and actinomycosis sometimes ulcerate. Many cattle without significant disease have one or more small ulcers of traumatic origin from plant awns on the hard palate and in the cleft (sulcus lingualis) where the base and shaft of the tongue meet.
DENTAL ABNORMALITIES
Chronic fluorosis is a cause of a variety of dental abnormalities in young animals with developing teeth (Boxes 7-22 and 7-23). Although cattle are most frequently involved, all large animals are susceptible. The teeth may appear mottled, striated, chalky, or hypoplastic or may have defective calcification. In severe cases teeth may be yellow, brown, or black and have multiple caries. Animals of any age may also develop bone lesions associated with chronic fluorosis.
Porphyria is a rare congenital condition of cattle transmitted by a simple autosomal-recessive gene. The teeth often appear pink because of the presence of porphyrins and fluoresce pink, purple, or red when exposed to ultraviolet light. Affected calves often develop photosensitization and anemia. This condition must be differentiated from superficial staining caused by ingestion of black walnut hulls or other compounds with staining properties.
Excessive or uneven wear or loss of teeth is often seen in horses and ruminants as they age. Tooth wear, particularly of incisors, is more rapid in animals on sandy range. Periodontal disease can cause premature loss of teeth (broken mouth) and tends to be most common in sheep in some geographic areas; the cause of this is unknown.35
The most common dental disease in horses has been described as periodontal disease.36 In horses fractured teeth or teeth with a small tract into the root through an open infundibulum often result in tooth root abscesses. This dental decay is a result of hypoplasia of the cementum of the enamel lakes and occurs most frequently in the second and third lower cheek teeth and third and fourth upper cheek teeth.37 These can cause sinusitis and foul-smelling unilateral nasal discharge if upper cheek teeth are involved or draining tracts to the exterior skin surface if lower cheek teeth are involved.
Most of the other causes of dental abnormalities listed here are bone abnormalities that cause secondary loss of teeth. See Chapter 32 for more details.
ICTERUS (JAUNDICE)
Icterus, and jaundice, are synonymous terms referring to the expression of a yellow coloration in the sclera and mucous membranes resulting from increased amounts of bilirubin in tissues and increased serum bilirubin levels (Boxes 7-24 and 7-25). Bilirubin especially stains elastic tissues and is thus most visible in the sclera and vulva. Icterus usually indicates decreased excretion, of bilirubin with liver or biliary tract diseases or increased production, of bilirubin with hemolytic anemia.
The accumulation of conjugated bilirubin results in more pronounced jaundice than does a similar amount of unconjugated bilirubin, with the result that the most pronounced jaundice is usually seen with hepatic or biliary obstructive disease. Laboratory examination of serum for relative amounts of unconjugated (indirect reacting) and conjugated (direct reacting) bilirubin is essential in determining the cause of the icterus. Generally, mainly unconjugated bilirubin levels are elevated with hemolytic anemia. Anorectic horses may have a plasma unconjugated bilirubin of 5 or 6 mg/dL without any evidence of hemolytic anemia or liver disease. Anorectic ruminants also experience a rise in plasma unconjugated bilirubin, often to a level between 0.5 and 2 mg/dL.
In determining the cause of icterus, laboratory tests, including PCV, RBC count, and the liver enzymes SDH and GGT, should be determined. In horses, alkaline phosphatase (AP) may also be useful, although it is not liver specific.
When active hepatocellular damage is occurring, SDH, which is liver specific, and aspartate aminotransferase (AST [SGOT]), which is not liver specific, are found in serum in elevated levels. GGT and AP are more indicative of biliary tract disease or proliferation and tend to rise more slowly but also to remain elevated for a longer period than SDH, which has a short half-life. Elevated levels of GGT or AP are often associated with chronic liver disease, cholangitis, cholelithiasis, or liver flukes. It is possible to have liver disease without icterus., Production and elimination of bilirubin are often equal in chronic liver disease, but acute liver disease or liver failure is usually associated with icterus. Although liver function tests such as BSP half-time can be run to determine the extent of liver damage, in most cases a liver biopsy must be taken for histopathologic examination to make a specific etiologic diagnosis of the cause of liver disease.
Liver abscesses rarely result in icterus because they rarely damage a sufficient percentage of liver to impair bilirubin clearance. They do cause multifocal hepatic damage and therefore are often associated with increased levels of SDH and AST when in the acute stages of formation.
Hemolytic anemia is characterized by destruction of RBCs either intravascularly or in the reticuloendothelial organs. This increased destruction results in production of bilirubin more rapidly than it can be removed by the liver, resulting in icterus. The specific cause of hemolytic anemia may sometimes be evident, as when autoagglutination is seen (autoimmune hemolytic anemia), Anaplasma, bodies are visible in stained RBCs of cattle, or E. equi, blue cytoplasmic inclusion bodies are seen in stained neutrophils.
In mature sheep, the most common cause of severe icterus is copper toxicity. In lambs, yellow lamb disease caused by C. perfringens, type A is a leading cause.
1 Merritt AM, Kohn CW, Ramberg CF, et al. Plasma clearance of (51Cr) albumin into the intestinal tract of normal and chronically diarrheal horses. Am J Vet Res. 1977;38:1769.
2 Holland JL, Kronfeld DS, Sklan D, Harris PA. Calculation of fecal kinetics in horses fed hay or hay and concentrate. J Anim Sci. 1998;76:1937.
3 Church DC. Digestive physiology and nutrition of ruminants, ed 2. Corvallis, Ore: O&B Books, 1979.
4 Moon HW. Mechanisms in the pathogenesis of diarrhea. J Am Vet Med Assoc. 1978;172:443.
5 Deprez P, Chiers K, Gebhart CJ, et al. Lawsonia intracellularis, infection in a 12-month-old colt in Belgium. Vet Rec. 2005;157(24):774.
6 Weese JS, Parsons DA, Staempfli HR. Association of Clostridium difficile, with enterocolitis and lactose intolerance in a foal. J Am Vet Med Assoc. 1999;214:229.
7 Ulshen MH. Diarrhea and steatorrhea. In: Hoeckelman RA, editor. Primary pediatric care. St Louis: Mosby; 1987:p 905.
8 Smith BP, House JK, Magdesian KG, et al. Principles of an infectious disease control program for preventing nosocomial gastrointestinal and respiratory tract diseases in large animal veterinary hospitals. J Am Vet Med Assoc. 2004;225:1186.
9 Cohen ND, Divers TJ. Acute colitis in horses. I. Assessment. Compend Cont Educ (Pract Vet). 1998;20:92.
10 Hampson DJ, Lester GD, Phillips ND, et al. Isolation of Brachyspira pilosicoli, from weanling horses with chronic diarrhoea. Vet Rec. 2006;158:661.
11 Magdesian KG, Madigan JE, Hirsh DC, et al. Clostridium difficile, and horses: a review. Rev Med Microbiol. 1997;8:S46.
12 Donaldson MT, Palmer JE. Prevalence of Clostridium perfringens, enterotoxin and Clostridium difficile, toxin A in feces of horses with diarrhea and colic. J Am Vet Med Assoc. 1999;215:358.
13 Baverud V. Clostridium difficile, diarrhea; infection control in horses. Vet Clin Equine. 2004;20:615.
14 Waters M, Raju D, Garmory HS, et al. Regulated expression of the beta 2-toxin gene (cpb2) in Clostridium perfringens, type A isolates from horses with gastrointestinal diseases. J Clin Microbiol. 2005;43:4002.
15 Brees DJ, Sondhoff AH, Kluge JP, et al. Lawsonia intracellularis,–like organism infection in a miniature foal. J Am Vet Med Assoc. 1999;215:511.
16 Soedarmanto I, Zhicai W, Setyamahanani A, Lämmler C. Pheno- and genotyping of Rhodococcus equi, isolated from faeces of healthy horses and cattle. Res Vet Sci. 1998;64:181.
17 Hathcock TL, Schumacher J, Wright JC, Stringfellow J. The prevalence of Aeromonas, species in feces of horses with diarrhea. J Vet Intern Med. 1999;13:357.
18 Herholz C, Miserez R, Nicolet J, et al. Prevalence of β-2 toxigenic Clostridium perfringens, in horses with intestinal disorders. J Clin Micro. 1999;37:358.
19 Pearson EG, Heidel JR. Colonic and rectal biopsy as a diagnostic aid in horses. Compend Cont Educ (Pract Vet). 1998;20:1354.
20 Bacciarini LN, Boelin P, Straub R, et al. Immunohistochemical localization of Clostridium perfringens, B2-toxin in the gastrointestinal tract of horses. Vet Pathol. 2003;40:376.
21 Pace LW, Turnquist SE, Casteel SW, et al. Acute arsenic toxicosis in five horses. Vet Pathol. 1997;34:160.
22 Guglick MA, Macallistar CG, Panciera R. Equine cantharidiasis. Compend Cont Ed (Pract Vet). 1996;18:77.
23 Galey FD, Holstege DM, Plumlee KH, et al. Diagnosis of oleander poisoning in livestock. J Vet Diagn Invest. 1996;8:358.
24 Bueno AC, Seahorn TL, Moore RM. Diagnosis and treatment of right dorsal colitis in horses. Compend Cont Educ (Pract Vet). 2000;22:173.
25 Cohen ND, Mealey RH, Carter GK. The recognition and medical management of right dorsal colitis in horses. Vet Med. 1995;90:687.
26 Jones PA, Tomasic M, Gentry PA. Oncotic, hemodilutional and hemostatic effects of isotonic saline and hydroxyethyl starch solutions in clinically normal ponies. Am J Vet Res. 1997;58:541.
27 McGorum BC, Dixon DG, Smith GE. Use of metronidazole in equine acute idiopathic toxaemic colitis. Vet Rec. 1998;142:635.
28 Van Hoogmoed L, Rodger LD, Spier SJ, et al. Evaluation of peritoneal fluid pH, glucose concentration, and lactate dehydrogenase activity for detection of septic peritonitis in horses. J Am Vet Med Assoc. 1999;214:1032.
29 Latson KM, Nieto JE, Beldomenico PM, et al. Evaluation of peritoneal fluid lactate as a marker of intestinal ischaemia in equine colic. Equine Vet J. 2005;37:342.
30 McDougall EI. Studies on ruminant saliva. I. The composition and output of sheep’s saliva. Biochem J. 1948;43:99.
31 Phillipson AT, Mangan JL. Bloat in cattle. XVI. Bovine saliva: the chemical composition of the parotid, submaxillary and residual secretions. N Z J Agric Res. 1959;2:990.
32 Stick JM, Robinson NE, Krehbiel JD. Acid-base and electrolyte alterations associated with salivary loss in the pony. Am J Vet Res. 1981;42:733.
33 Alexander F. A study of parotid salivation in the horse. J Physiol. 1966;184:646.
34 Bankowski RA, Wichmann RW, Stuart EE. Stomatitis of cattle and horses due to yellow bristle grass (Setaria lutescens). J Am Vet Med Assoc. 1956;129:149.
35 Andrews AH. Acquired diseases of the teeth and mouth in ruminants. In: Harvey CE, editor. Veterinary dentistry. Philadelphia: Saunders; 1985:256.
36 Baker GJ. Oral examination and diagnosis: management of oral diseases. In: Harvey CE, editor. Veterinary dentistry. Philadelphia: Saunders; 1985:217.
37 Baker GJ. Dental disorders in the horse. Compend Cont Educ (Pract Vet). 1982;4:S507.