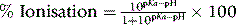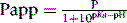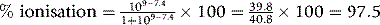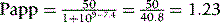Chapter 3 Amines
Introduction
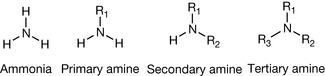
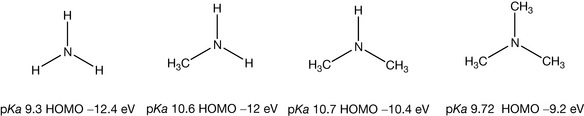
The amine group can be considered as the single most important group for conferring pharmacological activity. This is because it is the principle group within organic molecules that bears a positive charge. When it is present in a drug molecule it can exert biological effects through electrostatic interactions with negatively charged groups within proteins and also compete with positively charged cations such as sodium, potassium and calcium ions in biological processes. Amines are derived from ammonia and have the structures shown in Figure 3.1 for mono-, di- and trisubstituted nitrogen.
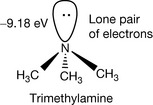
The structures may look planar but, in fact, the structures of amines are more or less tetrahedral, like tetrasubstituted carbon, as shown for trimethylamine in Figure 3.2. In the case of nitrogen, the position that would be occupied by a fourth bond in tetrasubstituted carbon is occupied by a lone pair of electrons. Since nitrogen is less electronegative than oxygen the lone pair within an amine structure is more available than the lone pairs on an oxygen atom to interact with a proton. An approximate measure of the availability of lone pairs of electrons is given by the energy of the highest occupied molecular orbital (HOMO). The deeper the ‘energy well’ that the molecular orbital occupies the less available the electrons are for bond formation with a proton. Thus water has a HOMO of −12.5 eV while ammonia has a HOMO of −10.5 eV, and thus its electrons are more available and it protonates more readily than water. The energies of HOMOs are arrived at by complex calculation and do not always match the experimentally observed energy levels for bonds, but they can reveal trends. The availability of the lone pair of electrons on the nitrogen atom is increased by substitution with alkyl groups; thus the HOMO of trimethylamine has an energy of −9.18 eV, i.e. it does not lie in an energy well as deep as that of ammonia and is a stronger base.
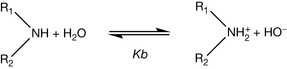
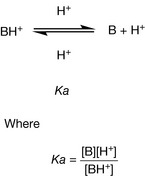
A variety of factors contribute to the lone pair on the nitrogen being more or less available. The pKa value for a base is derived from the equilibrium between the forms of the base shown in Figure 3.3 for a secondary amine in water where Kb is the equilibrium constant. In water, the equilibrium for nitrogen-containing bases lies well to the left and, for instance, in ammonia solution (Kb =1.8 × 10−5) in water only 0.0018% of the ammonia is protonated. Organic bases become increasingly water insoluble as the size of the R groups attached to them increases. However, in the case of a strong organic base the equilibrium shown in Figure 3.3 lies well to the right at physiological pH (7.4) and for most bases the equilibrium is almost entirely to the right at the pH of the stomach (1.0). The equilibrium constant Kb for the equilibrium shown in Figure 3.3 gives a measure of basic strength but more often the measure of basic strength given in tables is the pKa value, which describes the equilibrium shown in Figure 3.4 where B is a base.
For a strong base the equilibrium lies to the left at physiological pH since it does not readily give up its proton, thus behaving as a weak acid. The pKa value is −log Ka and the range of pKa values in water extends from 0 to 14. For a base, the higher its pKa value the stronger it is. At the pH of the stomach (ca. 1) the majority of amines are completely ionised but at physiological pH (pH 7.4) some basic drugs may contain an appreciable amount of un-ionised amine. The degree of ionisation of an amine drug has a bearing on its degree of protein binding and its degree of partitioning into tissues and hence the pharmacokinetics of a drug. The Henderson-Hasselbalch equation can be rearranged to calculate two important physicochemical properties for an ionisable drug. The percentage ionisation of a basic drug at a given pH may be determined from the following equation:
The variation in partition coefficient with pH for a base may be given from the following equation:
where P is the partition coefficient of the un-ionised drug and Papp is its partition coefficient at a particular pH. From equation 2 it can be seen that the partition coefficient of a drug is halved when the pH = its pKa value.
Factors affecting the pKa value of a base
Alkyl groups
In simple aliphatic amines the number of alkyl groups attached to the nitrogen has an effect on the basicity of the amine. Thus the order of basicity for amines is approximately: secondary > primary > ammonia > tertiary. The increased basicity of primary and secondary amines over ammonia is due to the inductive release of electron density from the alkyl groups which increases the electron density on the nitrogen atom. This can be seen for the methylamines in Figure 3.5.
In the case of tertiary amines there is a balance of two effects contributing to their basicity. Although the alkyl groups contribute to the electron density in tertiary amines the presence of three alkyl groups in the molecules reduces the ability of the protonated tertiary amine to hydrogen bond to water and thus increase its stability. In non-polar solvents, where hydrogen bonding does not occur, and thus does not contribute to the thermodynamics of protonation, tertiary amines are the strongest bases. The basicity also increases with increasing size of the alkyl group and this can override the unfavourable effects of trisubstitution on hydrogen bonding; thus tripropylamine (Pr3N) is a strong base with a pKa value of 10.74 in water.
![]() Self Test 3.1
Self Test 3.1
Calculate the percentage ionisation and Papp for the following amine drugs at pH 1.0 and pH 7.4.
Steric factors and basic strength in drug molecules
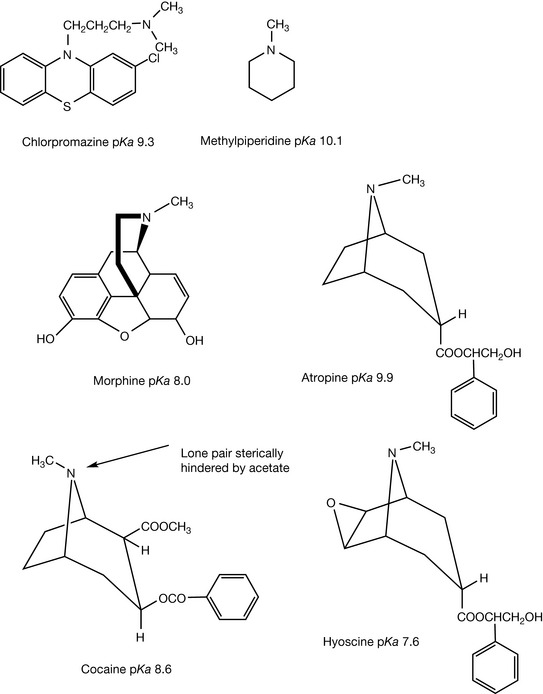
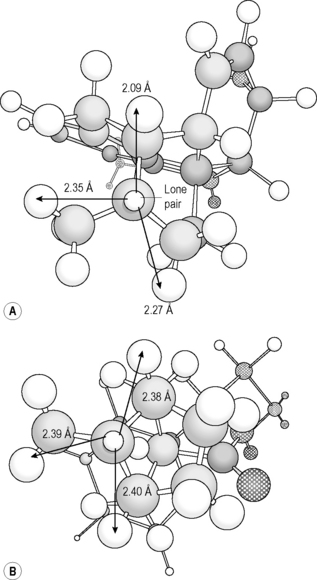
In practice, the reported pKa values for tertiary amine groups within drugs vary widely. The main explanation for variation in basicity is that the degree of steric hindrance of the basic nitrogen is very variable in tertiary amines. Often, the effects of a polar group close to the basic centre has been invoked as the explanation; however, electron withdrawing groups only exert strong effects over one bond length, with weak effects over two bond lengths. In Figure 3.6 a selection of N-methyl tertiary amines is shown. The basic strength of these amines is determined by availability of the lone pair of electrons on the nitrogen for protonation. The lone pair in N-methylpiperidine is more favourably positioned than that in chlorpromazine for protonation since there is less conformational flexibility in the alkyl groups attached to the nitrogen in the latter. The effects of conformation can be seen most clearly when morphine and atropine are compared (Fig. 3.7). Both structures are composed of interlocking rings and hence are conformationally restricted. In morphine, the geometry of the six-membered ring is quite distorted. This can be seen if one observes the distances between the proton on the nitrogen atom and the nearest hydrogens which in an undistorted ring would be the same. In the case of morphine the distances between the proton and the nearest hydrogens are 2.09, 2.27 and 2.35 Å. The ring distortion results in a flattening of the ring which means that it is difficult for the protonated nitrogen to adopt its preferred tetrahedral conformation (the angles between the proton and adjacent alkyl groups are 103.5°, 103.7° and 103.9°). In atropine, the ring is more symmetrical and less flattened (measurements from nitrogen to the nearest hydrogens are 2.38, 2.39 and 2.4 Å). Thus the nitrogen is held closer to its preferred tetrahedral geometry, ready for protonation (angles between proton and adjacent alkyl groups 105.6°, 106.1° and 106.2°). This accounts for the large difference in pKa: morphine 8.0 and atropine 9.9. This effect holds throughout the series of structural analogues. Thus codeine, dextromethorphan, diamorphine and nalorphine all have similar pKa values to morphine and homatropine,while benzotropine and hyoscyamine have similar pKa values to atropine. Cocaine is structurally related to atropine but its pKa value is lower. This has been attributed to the electron withdrawing effect of the ester group, which is two bonds removed from the nitrogen, but it is more likely that it is due to the proximity of the acetate ester group that sterically crowds the basic nitrogen. The acetate is forced into a close contact with the amine by the benzoic acid substituent that is next to it. A similar effect can be seen in hyoscine which has a pKa value of 7.6. In this case, the epoxide ring pushes the methyl towards the six-membered ring causing the ring to appear more boatlike in conformation (see Ch. 2), thus sterically crowding the nitrogen.
Electron withdrawing groups
Tertiary amines
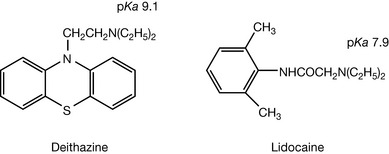
In some tertiary amines the proximity of electron withdrawing groups to the amine has an effect on basic strength. Thus diethazine has a pKa of 9.1 while lidocaine has a pKa of 7.9 (Fig. 3.8). In lidocaine the electron withdrawing amide group, separated by just one CH2 group from the amine, has a base weakening effect.
Secondary amines
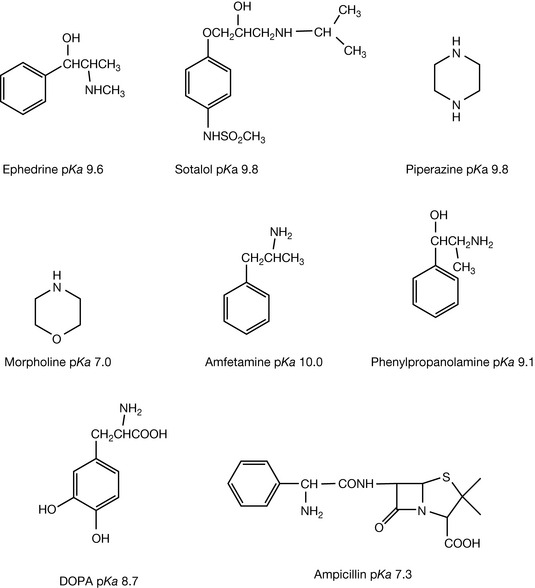
The basic strength of secondary amines is more predictable than that of tertiary amines. This is because one of the substituents is hydrogen and thus there are fewer problems with steric crowding, and hydrogen bonding to water is more favourable. This can be seen in the case of piperazine, ephedrine and sotalol (Fig. 3.9) which are quite different in structure but have similar pKa values. If interaction with neighbouring groups is strong, then the basic strength of secondary amines is affected as in the case of piperazine where the basic strength of the second amine group is reduced due to the presence of a positive charge on the first basic centre which repels the approach of a second proton. Other examples include the morpholino ring where the presence of an oxygen atom, which is only two bond lengths away from the nitrogen, because of the puckering of the ring, withdraws electrons, thus reducing the pKa of the secondary amine group to ca. 7.
Primary amines
Environmental factors have a strong influence on the basic strength primary amines. Thus amfetamine is strongly basic since there are no neighbouring electron withdrawing groups. The basic strength of phenylpropanolamine is lowered by the presence of a β-hydroxyl group which withdraws electrons from the amine group. In the case of DOPA the carboxyl group on the α-carbon withdraws electron density from the amine group, thus reducing its basic strength. In ampicillin the electron withdrawing effect of the amide and the benzyl group reduce the pKa value of the primary amine to 7.3.
Aromatic amines
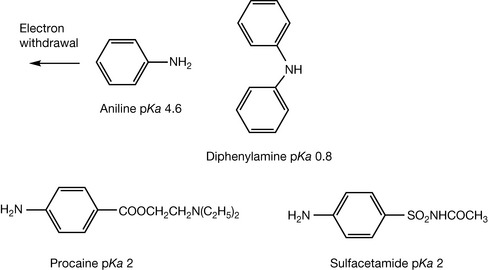
Aromatic amines are very weak due to the electron withdrawing effect of the benzene ring and aniline (Fig. 3.10) has a pKa value of 4.6. The presence of a second aromatic ring as in diphenylamine (Fig. 3.10) reduces the pKa value of the amine to 0.8. The presence of other electron withdrawing substituents in an aromatic ring either ortho or para to an aromatic amine group also reduces its pKa value. The pKa values of the aromatic amine groups found in sulphonamides such as sulfacetamide and some local anaesthetics such as procaine are ca. 2.
Heterocyclic amines
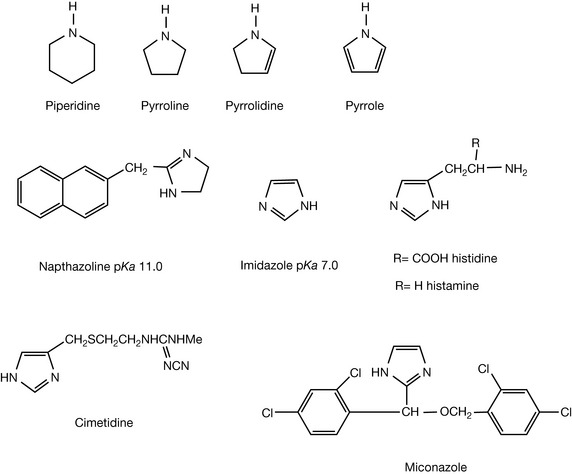
Chapter 4 discusses heterocyclic compounds in more detail; in this chapter some of the heterocyclic amines are discussed. The basic properties of amines such as piperidine and azolidine are similar to those of open-chain amines. Thus piperidine has a pKa of 11.1, that is similar to that of diethylamine that has a pKa of 11.0 (Fig. 3.11). Introduction of double bonds into the ring containing the nitrogen atom affects its basicity. Introduction of a single double bond into the pyrrolidine ring, to form pyrroline, does not have a marked effect on the basicity of the nitrogen but the introduction of two double bonds in the case of the pyrrole ring produces a nitrogen atom that has lost its basic character and is essentially neutral. Pyrrole and other π-excessive compounds will be discussed in Chapter 4.
Imidazoles
In the case of imidazole there are two nitrogen atoms in a five-membered ring as well as two double bonds. Imidazoles are weakly basic having pKa values in the range of 6–7. The basicity of the imidazole ring is weakened by the fact that the ring is very flat and thus the nitrogen cannot adopt its preferred tetrahedral stereochemistry when it is protonated. The imidazole ring occurs in histamine that is the natural ligand for histamine receptors. Histamine receptor antagonists such as cimetidine (Fig. 3.11) and omeprazole contain an imidazole ring. It is also found in the range of azole antifungal agents including clotrimazole, miconazole (Fig. 3.11) and ketoconazole. These agents all act as inhibitors of fungal P450 enzymes that are involved in the biosynthesis of ergosterol which is the major sterol of fungal membranes. The imidazole ring in these compounds in some way interferes with the binding of iron (II) at the active site of the enzyme; the iron is bound at the active site by imidazole ring containing histidine residues.
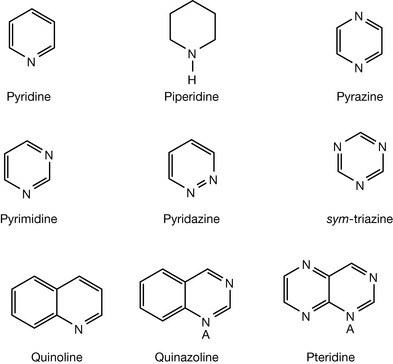
Pyridine and π-deficient heterocycles
In pyridine one of the carbon atoms in a benzene ring is replaced by a nitrogen atom that is in its sp2 hybridised form so that it lies in the plane of the ring. The nitrogen lone pair is in one of the sp2 orbitals and thus is less available for protonation because of the increased s character compared to the normal sp3 hybridisation for nitrogen. In s orbitals electrons are held closer to the nucleus of the atom and are thus in a deeper energy well. The pyridine ring is quite distorted with C–N–C angle being 117° rather than the 120° angle that applies to the C–C–C bonds in benzene. This is an indication of increased p-character (with pure p the bond angles are 90°) in the bonding orbitals while the orbital containing the non-bonding lone pair has increased s character, i.e. the non-bonding orbital and the bonding orbitals are no longer completely equivalent. Thus pyridine is a much weaker base, pKa 5.2, in comparison with piperidine, pKa 11.1. When two nitrogens are introduced into a heteroaromatic ring the s character of the lone pairs on the nitrogen atoms is further increased and the dinitrogen heteroaromatics are all very weak bases. This may seem rather counterintuitive but if the values in Table 3.1 are examined it can be seen that, in particular, the C–N–C bond angle is indicative of increased s character in the lone pair of electrons, hence decreased basicity. This bond angle more or less reflects the relative basicities of the dinitrogen heteroaromatics. Quinazoline and pteridine are stronger bases than pyrimidine since the additional aromatic ring increases the rigidity of the pyrimidine ring, resulting in bond angles closer to 120°, and thus decreased s character for the non-bonding lone pair at position A (Fig. 3.12).
Table 3.1 pKa values and bond angles for the heteroaromatic structures shown in Figure 3.12 (determined from molecular mechanical calculations)
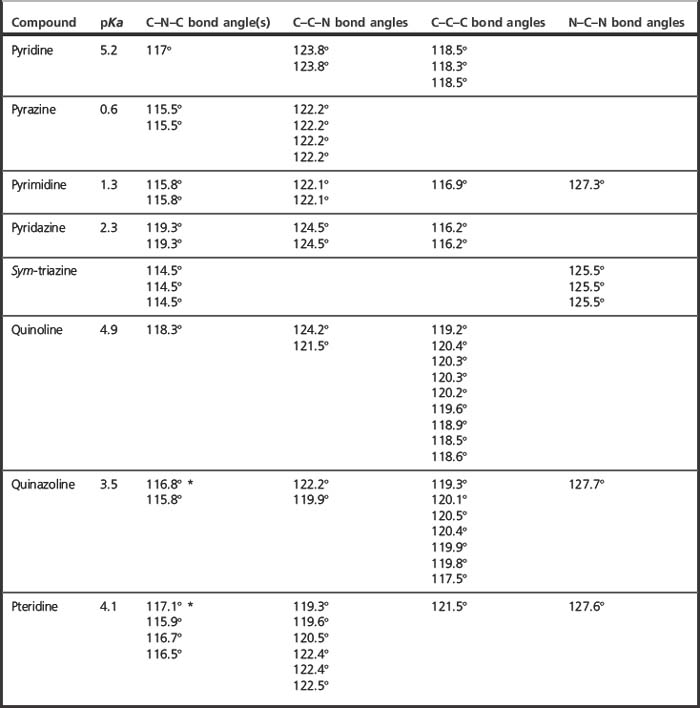
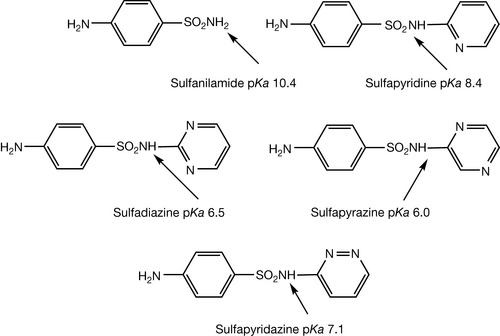
π-Deficient heteroaromatic rings occur in many drug structures. The most consistent exploitation of their properties is in the sulphonamide drugs (Fig. 3.13) where the properties of the heterocyclic ring contribute to the acidity of the sulphonamide. The acidity of the sulphonamide is influenced by the electron withdrawing properties of the heterocyclic ring. This can again be viewed simply as electron withdrawal by the heterocyclic ring or it can be viewed in terms of increased s character of some of the carbon atoms within the ring. This can be seen particularly where the bond angle is increased well above 120°, indicating movement towards the sp state where the bond angle is 180°. Thus, in comparison with sulfanilamide, sulfapyridine is a stronger acid and acid strength increases still further when two nitrogens are introduced into the heteroaromatic ring.
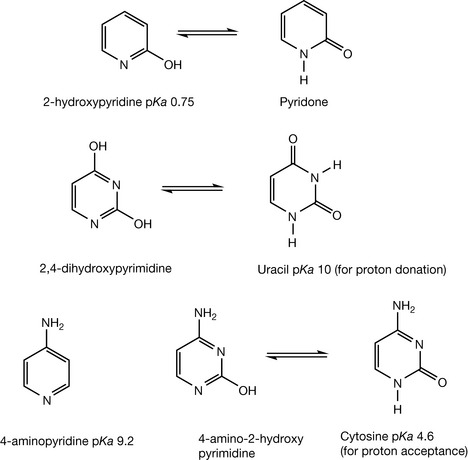
Substitution within the ring of nitrogen-containing heteroaromatic compounds can have a marked effect on their properties. Substitution with an amine or a hydroxyl causes the greatest effect on the pKa values of the heteroatoms. Thus a hydroxyl adjacent to the nitrogen in pyridine almost eliminates its basicity due to electron withdrawal by the oxygen and the hydroxy pyridine can also exist in its pyridone form (Fig. 3.14). The compound 2,4-dihydroxypyrimidine is acidic rather than basic and exists largely in its pyrimidone form, which is commonly known as uracil. Uracil is one of the bases found in RNA but, in fact, it is a weak acid rather than a base and so is its methyl derivative thymine which is a DNA ‘base’. 4-aminopyridine is a relatively strong base in comparison to aniline having a pKa of 9.2. The compound 4-amino-2-hydroxypyrimidine is a weak base with a pKa similar to that of aniline and is commonly known as cytosine, one of the DNA bases.
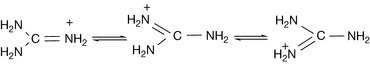
Guanidines
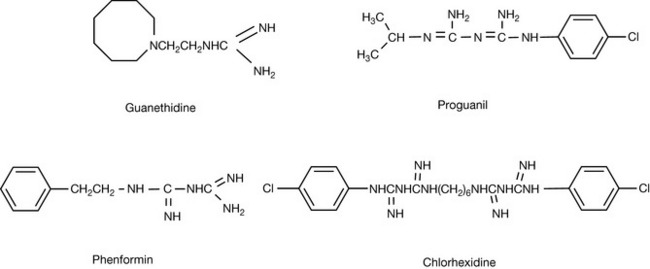
Guanidine is derived biosynthetically from urea. The ureides themselves will be discussed in Chapter 4. Guanidine is a very strong base with a pKa value of 13.65; thus it can form salts even with very weak acids. The strength of the base arises from the resonance stabilisation of the charged base (Fig. 3.15). The high degree of symmetry of the guanidinium ion and its complete ionisation with the charge distributed over quite a large ion enables it to mimic inorganic ions to some extent. Thus it competes effectively with Mg2+ for binding sites on phosphate groups in phospholipids. This ability to mimic an inorganic ion may account for the ability of guanethidine to inhibit the Ca2+ promoted fusion of noradrenaline-containing vesicles with the membrane of a neuron and thus act as an anti-hypertensive agent.
Guanidino groups are also present in streptomycin, which was the first aminoglycoside antibiotic used in treating tuberculosis. Streptomycin and related antibiotics interfere in the translation of RNA into protein and this is probably due to their polycationic nature and ability to mimic both Mg2+ and polyamines that are involved in phosphate group stabilisation.
Biguanides
Biguanine, like guanine, is strongly basic, having a pKa of 12.8. Biguanides occur in a number of therapeutic agents.
Proguanil (Fig. 3.16) was a relatively early antimalarial drug and it is still useful as a first line of defence against the erythrocytic stage of malaria. It is metabolised in the human body to an active metabolite which mimics the pteridine ring in folic acid and acts as a selective inhibitor of folate reductase in the parasite, thus inhibiting its DNA synthesis.
Phenformin (Fig. 3.16) is used as an antidiabetic drug and lowers plasma glucose levels in diabetics but has no effect on plasma glucose levels in non-diabetics. The mechanism of action of these drugs is not fully understood; however, they are believed to affect glucose metabolism. This might be linked to an ability to mimic magnesium that is involved as a co-factor in many of the enzymes of glucose catabolism.
The bisbiguanide chlorhexidine (Fig. 3.16) is a powerful antibacterial agent and its actions are related to those of other cationic surfactants that are effective antibacterial agents. The charged groups in the molecules bind to the bacterial membrane, thus disrupting the functioning of the cell.
Salt formation
One of the fundamental reactions of amines is the formation of a salt that will generally occur by reaction with both inorganic and organic acids. Salt selection is an important step in pharmaceutical development. Most organic amines are water insoluble in the form of their free bases and for the purposes of convenience in their formulation they are usually used as a salt. The salts of amines can be purified by recrystallisation and are less susceptible to oxidative degradation than the free bases. They are also water soluble and thus readily formulated into injections. In many cases amines are used in the form of their hydrochloride salts and the use of sulphates and hydrobromides is also common. Solutions of the hydrochloride salts of amines are usually quite acidic due to salt hydrolysis, as shown in Figure 3.17, having pH values of ca. 4.0. Surprisingly, sometimes due consideration is not given to the irritating acidity of hydrochloride salts in formulations such as eyedrops.
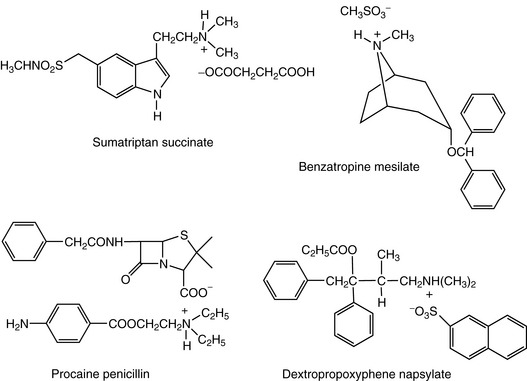
In some cases hydrochloride salts are not very water soluble due to the strength of association between the base and its counter ion, and other salts have to be used. For instance, salts may be prepared with di- or tricarboxylic acids, e.g. ergometrine tartrate, diethylcarbamazine citrate, sumatriptan succinate (Fig. 3.18) and lisuride maleate are all used because of the water insolubility of their hydrochlorides. Similarly strong monobasic organic acids such as mesilate are used to promote water solubility, as in the case of benzatropine mesilate (Fig. 3.18) and phentolamine mesilate. In some instances salts are too ready to absorb water from the atmosphere, for instance the muscle relaxant drug atracurium is formulated as its besilate salt to avoid the problems of water absorption experienced with its mesilate.
Sometimes, low water solubility is a desirable property and may be useful in the purification of a base after synthesis. It is also of therapeutic use in the formulation of some amines as depot injections, e.g. procaine penicillin (Fig. 3.18).
Salts are sometimes chosen with regard to their organoleptic properties and, for instance dextropropoxyphene napsylate (Fig. 3.18), is sometimes used because it is not as bitter as the hydrochloride.
Reactions of amines in relation to storage and formulation
Oxidation
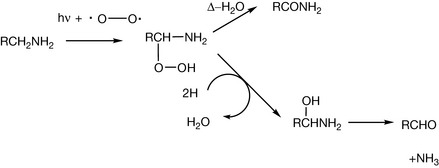
All amines are prone to oxidation in air and aromatic amines are particularly unstable in this regard. Primary amines are susceptible to oxidation on the α-carbon, the reaction being catalysed by light (Fig. 3.19).
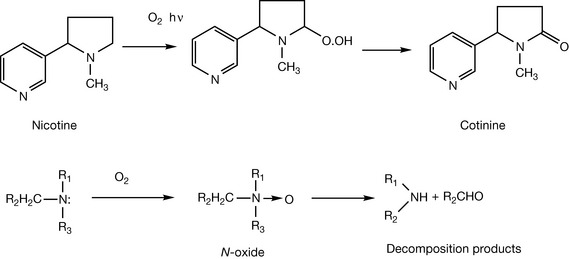
Secondary and tertiary amines behave in a similar manner (Fig. 3.20).
As well as being susceptible to oxidation on the α-carbon, tertiary amines readily form N-oxides. Traces of these compounds are usually formed when an aqueous solution of a tertiary amine is stored for a few days. N-oxides are fairly unstable and readily decompose to an aldehyde and a secondary amine.
Acylation
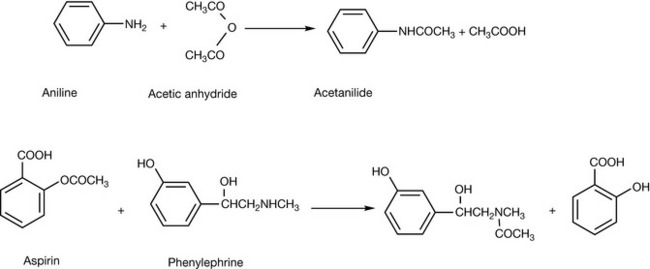
Primary and secondary amines can be chemically acylated with, for instance, an acid anhydride (Fig. 3.21) to give an amide, e.g. the acetylation of acetanilide, but this process can also occur with chemically incompatible ingredients in formulations. For example, aspirin is an acetylating reagent and can react with amines such a phenylephrine. Both of these drugs are present in some proprietary formulations for treating colds and flu. Stearic acid is another possible acylating reagent since it is used as a releasing agent in some tablet formulations.
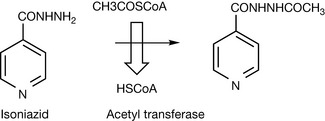
Acetylation of amines is an important route for the metabolism of primary aromatic amines such as the sulphonamide drugs or the antituberculosis drug isoniazid (Fig. 3.22). The co-factor involved in acetate transfer is acetyl co-enzyme A (acetyl CoA) and the reaction is catalysed by acetyl transferase, N-acetylation generally reduces toxicity.
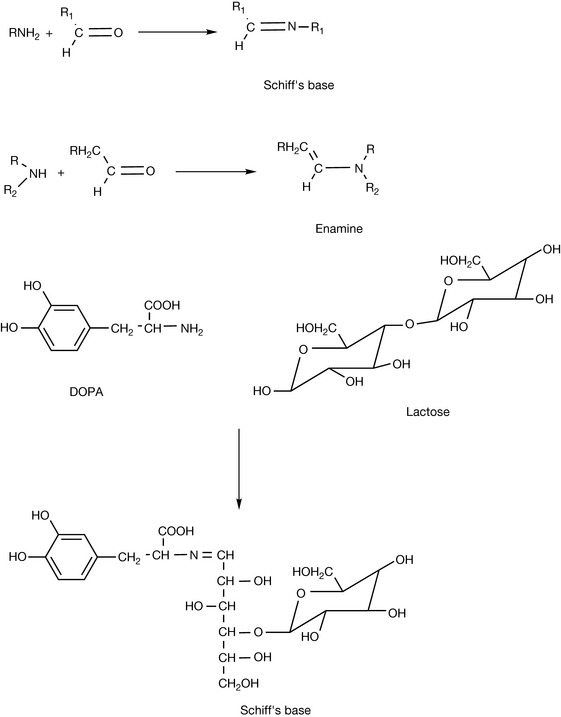
Schiff’s base formation
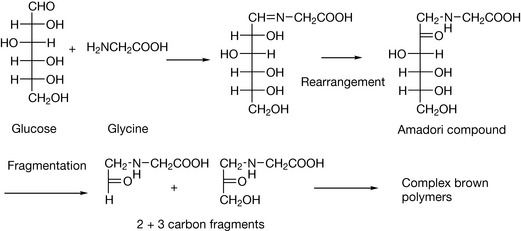
Primary and secondary amines can react readily with aldehydes and more slowly with ketones to give Schiff’s bases or enamines, respectively. This reaction is particularly important with reducing sugars such as glucose, lactose and fructose that may be incorporated in formulations with amines. For instance, if LDOPA were formulated in a tablet with lactose as a filler, the reaction shown in Figure 3.23 could occur. Such reactions, leading to discolouration of lactose-containing tablets, have been reported for the amines isoniazid and dextroamfetamine. During autoclaving of glucose or fructose, 2-hydroxymethylfurfuraldehyde is formed, which has been shown to react with sulphonamides and penicillins containing an amino group and aminoglycoside antibiotics, and this is potentially a problem in glucose-containing infusions. Glucose reacts with amino groups in proteins to form in the first instance Schiff’s bases but then more complex, brown, rearrangement products are formed (the Maillard reaction, Fig. 3.24). This type of reaction is also responsible for many of the complications of diabetes as such but also for some of the effects of ageing as well as being involved in the development of flavours during cooking of foodstuffs where nitrogen and oxygen heterocyclic compounds are generated from the type of 2 and 3 carbon intermediates shown in Figure 3.24. Cataracts in the eye arise from the reaction of glucose with the lens crystallin proteins, causing them to become opaque, and even wrinkles have been attributed to the cross-links that formed between protein chains as a result of the Maillard reaction. Reactions of proteins with reducing sugars can also be a problem in the formulation of peptide drugs. If they are used as excipients, it is important to ensure that the peptide is efficiently freeze dried so that no traces of water remain that would favour the Maillard reaction.
Quaternary amines
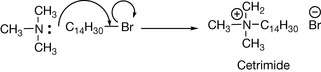
Quaternary ammonium salts are formed by direct reaction of a tertiary amine with an alkyl halide. For example, trimethylamine can be reacted with cetyl bromide to produce the disinfectant agent cetrimide (Fig. 3.25).
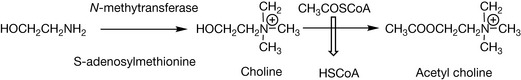
In vivo, there are a number of important biologically active quaternary amines. The neurotransmitter acetyl choline is biosynthesised as shown in Figure 3.26.
Acetyl choline is a fixed non-pH dependent cation analogous to an inorganic cation such as K+ or Na+. Like all quaternary amines, it is charged at all pH values. Its positive charge attracts to negatively charged groups on proteins and it binds to such groups in the acetyl choline receptor that is a ligand-gated ion channel. Binding of acetyl choline causes the ion channel to open and allows the entry of Na+ ions into the cell. Binding of acetyl choline requires both the charged and lipophilic portion of the molecule since its action is terminated by hydrolytic removal of the acetate portion of the molecule.
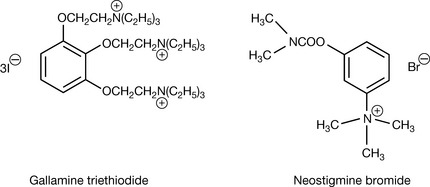
Quaternary ammonium salts with a single quaternary centre tend to be both water and fat soluble. Thus acetyl choline esterase inhibitor neostigmine bromide (Fig. 3.27) is both water soluble and moderately well absorbed from the GI tract, whereas polycationic quaternary amines such as gallamine triethiodide (Fig. 3.27) are much less lipophilic and have to be administered by injection. Apart from the role of long-chain quaternary amines as disinfectants, most quaternary amine drugs act on the cholinergic system either as acetyl choline esterase inhibitors or via direct actions on the acetyl choline receptor.
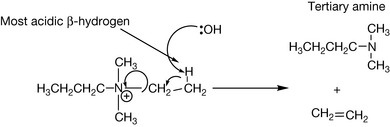
There is one important reaction of quaternary amines that has been exploited in drug design: the Hofmann elimination (Fig. 3.28). The reaction is promoted by strongly basic conditions and the more acidic a β-hydrogen atom the more readily the reaction occurs. Atracurium is based upon tubocurarine that was discovered as an arrow poison that paralyses muscles. Thus atracurium is used as a neuromuscular blocker that acts at the acetyl choline receptor and is used to relax muscles such as the abdominal muscles in preparation for surgery. The β-hydrogens in atracurium are particularly acidic and thus atracurium breaks down spontaneously at physiological pH and thus has a short duration of action and is less likely to produce cardiac side effects (see Ch. 16).
![]() Self Test 3.6
Self Test 3.6
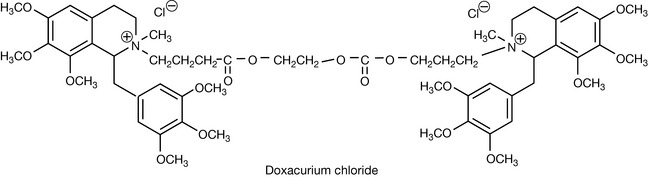
Draw the mechanism for the Hofmann elimination for the atracurium analogue doxacurium chloride.
![]() Self Test 3.3
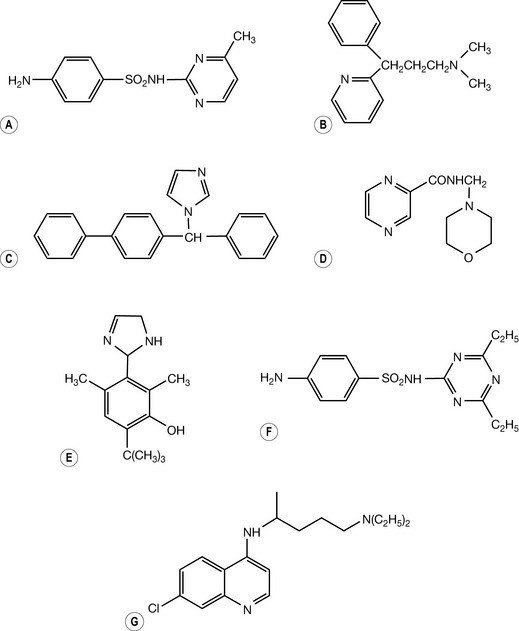
Self Test 3.3
a. A Pyrimidine, B Pyridine, C Imidazole, D Pyrazine, E Imidazoline, F sym-triazine, G Pyridine.