Chapter 20 Drugs affecting the endocrine system
Introduction
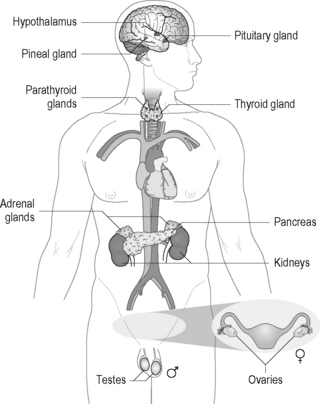
The endocrine system is a series of glands within the body including the hypothalamus, the pituitary, the thyroid, the parathyroids, the adrenal glands, the pineal gland, the pancreas and the reproductive glands (e.g. ovaries and testes) that controls many functions of the operation of the body. The endocrine system works alongside the nervous system to control many of the body ’s basic functions such as growth and development, homeostasis, energy levels, etc. The location of the glands within the body is depicted in Figure 20.1.
These glands are responsible for the production of hormones, which control many functions of the human system as indicated above. Hormones may be classified into three groups based loosely on their chemical structure:
Under normal circumstances the body will control the levels of hormones by positive and negative feedback systems, although negative feedback is most common in the endocrine system and this can be exemplified by the thyroid hormones. The regulation of the secretion of the thyroid hormones (thyroxine, triiodothyronine) is controlled by the hypothalamic–pituitary axis. When there is a need for synthesis of thyroxine or triiodothyronine the hypothalamus secretes thyroid releasing hormone (TRH), which results in the pituitary gland secreting thyroid stimulating hormone (TSH), causing the pituitary gland to synthesise the thyroid hormones. These hormones are then transported in the bloodstream to their required sites of action. The hormones may be free or bound to transport proteins (e.g. thyroxine-binding globulin), and it is only the free form which is active. When the levels of these hormones approach the required levels once again, the negative feedback system switches on and causes the secretion of TRH to be switched off. Another feedback mechanism involving the endocrine system is associated with insulin production. Following the ingestion of food there will be an increase in the blood sugar levels. Within the endocrine system the pancreas responds to this by producing the hormone insulin. The insulin aids in the uptake of glucose into cells, thereby reducing the blood sugar levels. Once the blood sugar levels drop, the pancreas stops producing insulin.
Diseases associated with the endocrine system
There are a number of disease states that are associated with the endocrine system. For example, hormones may be released in amounts that are too great or too small for the body to work normally. This situation is often referred to as hormone imbalance; however, other problems are associated with the endocrine system. Typical diseases associated with the endocrine system include hyperthyroidism and hypothyroidism, diabetes and osteoporosis. Additionally, in this chapter we will consider drugs that act on the endocrine system or drugs that are associated with problems related to this system. Details of drugs that are employed in the control of various aspects relating to the endocrine system will also be detailed. For instance, the utilisation of synthetic sex hormones for birth control will be dealt with.
Diabetes
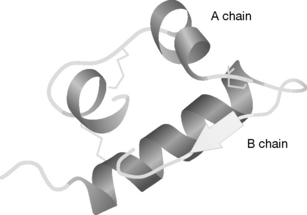
As indicated above, the endocrine system includes the pancreas, which is responsible for the production of insulin. Insulin is a peptide hormone, with the human form comprising 51 amino acids within two chains (A and B), one of 30 amino acids and the other 21 amino acids. It has a molecular weight of 5808. The 3-D structure is shown in Figure 20.2. The B chain contains a larger region of α-helix.
Insulin is synthesised in the beta cells of the islets of Langerhans in the pancreas with the alpha cells producing glucagon, a second peptide hormone. The production of these two hormones controls the levels of blood sugar in the body. Whenever the level of sugar in the bloodstream is detected as being too high the pancreas releases insulin, which aids in the uptake of sugar by muscles, other cells and the liver, where it is stored as glycogen (a polymer containing large numbers of glucose units). When the pancreas detects that the blood sugar level is too low, it releases glucagon, which causes the breakdown of glycogen and the restoration of the normal blood sugar levels.
Diabetes or diabetes mellitus results in the body having too much sugar in the blood. It occurs in two main forms, type 1 or type 2 diabetes. Type 1 diabetes, previously referred to as early-onset diabetes, juvenile diabetes or insulin-dependent diabetes mellitus, is caused by the body being unable to produce insulin, whilst type 2 diabetes (adult-onset diabetes and non-insulin-dependent diabetes) results from the body not being able to produce enough insulin or not being able to utilise insulin properly.
The treatment of type 1 diabetes is the subcutaneous injection of insulin, as insulin cannot be administered orally because it would be broken down in the stomach due to the low pH. Initially, animal insulin was used in the treatment of diabetes, since bovine and porcine insulin are structurally similar to human insulin. Nowadays, most of the insulin used in the treatment of diabetes is human insulin produced via recombinant DNA (see Ch. 27). There are a number of insulin formulations available, e.g. short-, intermediate- or long-acting and biphasic (a mixture short- and intermediate-acting insulin), and these are described in more detail in Chapter 27. There is a range of therapy protocols indicated, based on the individual condition of the patient.
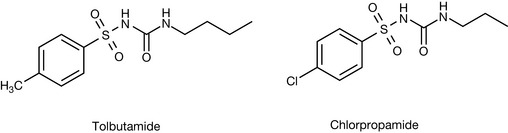
Treatment of type 2 diabetes is through a range of drugs with different modes of action. Two of the major classes are the sulphonylureas and the biguanides and these are used along with a number of individual compounds with a range of activities. The sulphonylureas are the oldest form of oral hypoglycaemic agents and work by stimulating the secretion of insulin in the pancreas. There are a large number of sulphonylureas that have been employed for this purpose and are nowadays classified as first and second generation (Fig. 20.3). The only first-generation sulphonylureas indicated in the BNF nowadays are chlorpropamide and tolbutamide (these are also discussed in Ch. 4).
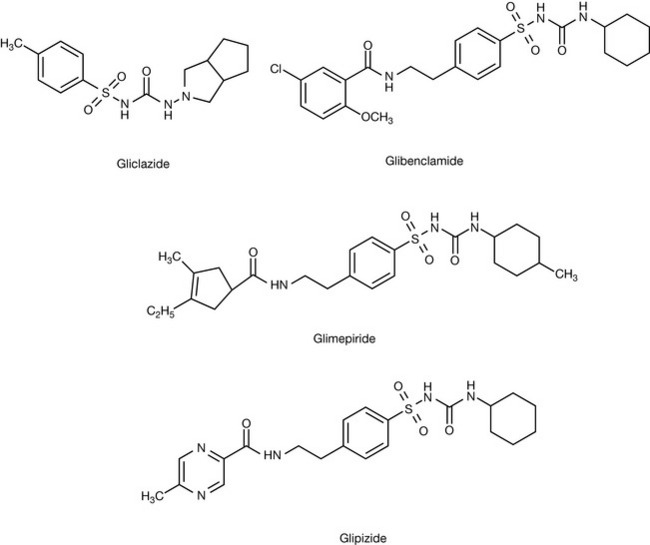
The second-generation sulphonylureas are shown in Figure 20.4 and these compounds are considered to be safer in that they have fewer side effects and are thought to be more effective than the older drugs.
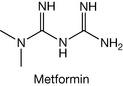
The second class of compounds used are the biguanides and, although a number of these have been used in the past, several have been withdrawn due to side effects (lactic acidosis) and only one such agent, metformin (Fig. 20.5), is presently indicated in the UK. The mode of action of the biguanides still seems to be unclear but is reported to include decreasing the absorption of glucose and inhibiting hepatic glucose output.
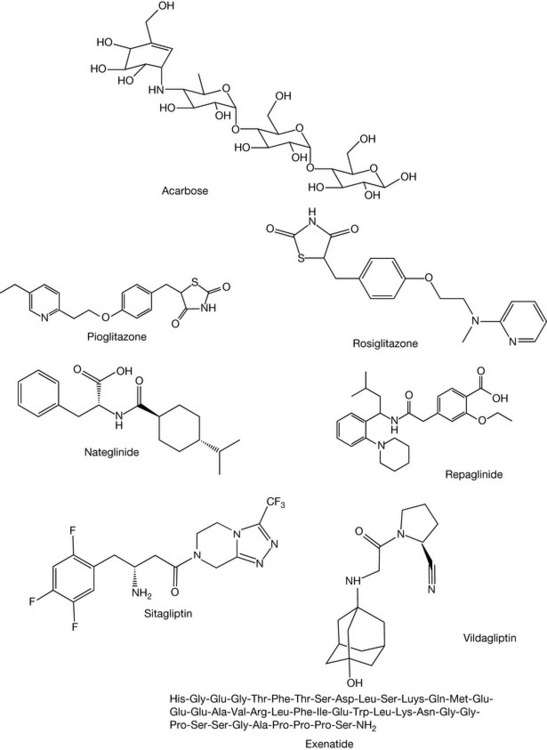
A variety of other agents come under the heading of antidiabetic drugs and the list includes acarbose, nateglinide and repaglinide, pioglitazone and rosiglitazone, sitagliptin and vildagliptin, and exenatide (Fig. 20.6).
Acarbose is a synthetic oligosaccharide that was designed to reduce the rate at which enzymes in the intestine (alpha-amylase and alpha-glucosidase) break down carbohydrates. In doing so, acarbose slows down the release of sugar into the blood stream.
Pioglitazone and rosiglitazone are both classified as thiazolidenediones (based on the heterocyclic ring system in this group of drugs). This group of drugs is often banded under the heading ‘glitazones’. This group of drugs is known to be useful in type 2 diabetes as they help in what is known as insulin resistance, in which the body produces insulin but cannot utilise it effectively to reduce blood sugar levels. These thiazolidenediones bind to peroxisome proliferator activated receptor-gamma (PPAR-γ) which is a nuclear receptor. In doing so, these drugs are involved in altering the transcription of several genes involved in glucose and lipid metabolism and energy balance. Consequently, the insulin resistance is reduced and the body can effectively deal with blood sugar.
Nateglinide and repaglinide are secretagogues in that they promote the secretion of insulin in the pancreas. These agents interfere with the beta cells in the islets of Langerhans and open the calcium channels in the cells, the increased calcium resulting in the enhanced insulin secretion.
Sitagliptin and vildagliptin belong to a new class of drugs known as dipeptidyl peptidase-4 (DPP-4) inhibitors. DPP-4 acts on two incretin hormones, namely glucagon-like peptide 1 (GLP-1) and glucose-dependent insulinotropic peptide (GIP), both of which are released in the intestine following food intake. They are able to stimulate the production of insulin, depending on the levels of glucose; however, they are both inactivated via the enzyme DPP-4. Sitagliptin and vildagliptin, as inhibitors of DPP-4, act by preventing the inactivation of GLP-1 and GIP.
Exenatide is a drug that is described as an incretin mimetic in that it has a strong structural resemblance to GLP-1. This drug is a synthetic peptide based on a hormone, exendin-4, found in the saliva of the Gila monster. Research on this peptide showed that it had properties similar to GLP-1. Now, GLP-1 cannot be administered to patients with type 2 diabetes since it is rapidly inactivated in the body by DPP-4, as indicated above. Due to the structural similarity between GLP-1 and exendin-4, it was investigated as a GLP-1 mimetic and shown to work effectively. It was also shown to have a considerably longer half-life than GLP-1 as it was not broken down by DPP-4 as quickly as the natural analogue.
Anti-obesity drugs
Obesity is defined as a body mass index greater than 30. Obesity may result via a variety of conditions but is, in some instances, closely associated with certain endocrine diseases such as diabetes. Obese people are advised to overcome their problem by means of appropriate dietary changes and an increase in physical exercise. However, when these do not work, drug treatment is necessary. Several drugs may be employed in these circumstances and fall into two types of anti-obesity drugs acting on the gastrointestinal (GI) tract and appetite suppressants.
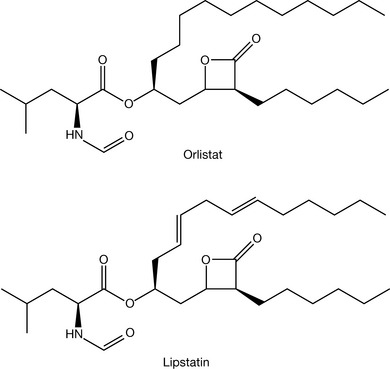
Orlistat (Fig. 20.7) is the only GI tract drug indicated in the BNF, and is a lipase inhibitor, which reduces fat intake. This drug is a synthetic analogue of lipstatin which is a naturally occurring substance produced by Streptomyces toxytricini. Orlistat is an irreversible inhibitor of pancreatic and gastric lipases and as such it prevents these lipases from breaking down triglycerides into their absorbable form (free fatty acids and monoglycerols). The triglycerides are thus eliminated without absorption and thus there is a decreased dietary intake of fat.
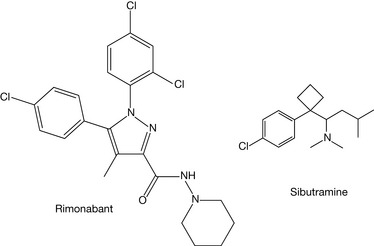
In the BNF there are only two appetite suppressants (Fig. 20.8) indicated as anti-obesity drugs, namely Rimonabant and Sibutramine. Rimonabant has been marketed as an anti-obesity drug for several years in the UK, although it never received FDA clearance. It is a cannabinoid receptor antagonist, which blocks binding to neuronal CB1 receptors. This inhibition of binding of endogenous cannabinoids prevents an increase in appetite. This drug has recently had its market authorisation in the UK suspended due to concerns regarding psychiatric problems associated with its use.
Sibutramine (hydrochloride) is structurally related to the amfetamines, which were at one stage marketed as appetite suppressant drugs. These drugs were removed from the market due to their increased use as recreational drugs and because of the serious side effects of their use, e.g. tachycardia, hypertension and addiction. Sibutramine is a serotonin and noradrenaline reuptake inhibitor, thereby promoting weight loss because of its ability to help patients feel that their hunger has been satisfied. It is also suggested that weight loss may be helped by the drug, increasing energy expenditure.
Thyroid hormones
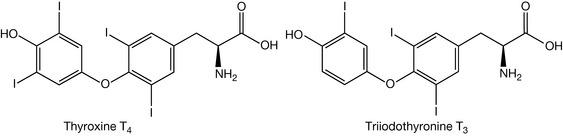
The thyroid hormones, as indicated above, are amino acid-based hormones. The thyroid gland produces two main active thyroid hormones, thyroxine and triiodothyronine. When produced, these hormones circulate in the blood with the major fraction being bound to proteins which transport them throughout the body. However, these hormones are only active in the unbound form. The structures of the two hormones are shown in Figure 20.9.
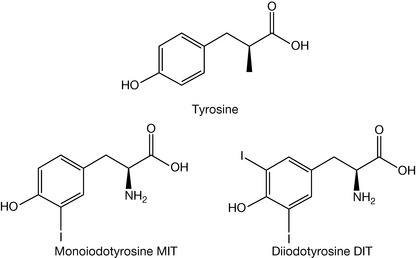
The thyroid hormones (often referred to as T3 and T4) when released into the blood stream are transported throughout the body where they control metabolic processes in almost all cells in the body. T3 is more active than T4 by a factor of approximately 10. As one can observe, these two hormones contain iodine and it is the function of the thyroid gland to utilise dietary iodine in the conversion of tyrosine into monoiodotyrosine (MIT) and diiodotyrosine (DIT) (Fig. 20.10).
The linking of one molecule of MIT with one of DIT results in the formation of T3, whilst two molecules of DIT combine in the formation of T4. T4 can also be converted into T3 by the 5′-deiodinase enzyme system. Within the body, the control system associated with the production of these hormones is directed by TSH (thyroid stimulating hormone) and TRH (thyroid releasing hormone).
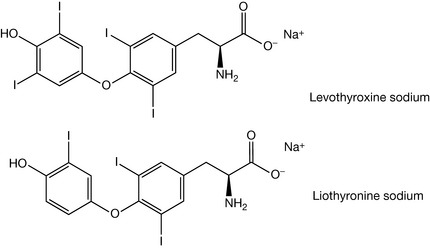
Within the thyroid system, disease states can lead to hypothyroidism and hyperthyroidism. Hypothyroidism results from abnormally low production of thyroid hormone in thyroid glands. The prevalence of this condition is reported as being between 2% and 5% of the world population, although a substantial number are in the subclinical category. Hypothyroidism is much more common in females than in males, and the frequency of the disease increases with age. The problem may arise due to an insufficient intake of iodine in the diet. It may be due to an inherited disorder (Hashimoto ’s thyroiditis) or it may be due to inflammation of the thyroid gland (lymphocytic thyroiditis). The conventional treatment for this condition is use of thyroid hormones levothyroxine sodium or liothyronine sodium (Fig. 20.11).
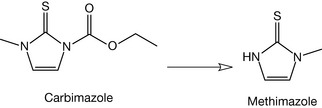
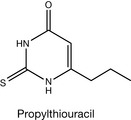
Hyperthyroidism results from an overactive thyroid gland producing excess amounts of thyroid hormones which circulate through the body. The thyroid hormones affect many cellular functions throughout the body and this excess results in increased metabolic activity leading to a variety of symptoms, the most common being increased heart rate, tremor, diarrhoea and weight loss. The treatment of hyperthyroidism involves the use of two drugs: carbimazole and propylthiouracil. Carbimazole is a prodrug that is converted into methimazole (Fig. 20.12), which is the active agent. Methimazole interferes by inhibiting the thyroid peroxidase system that is involved in the conversion of tyrosine into MIT and DIT, thereby preventing the synthesis of T3 and T4.
Propylthiouracil (Fig. 20.13) acts similar to methimazole in that it inhibits the thyroid peroxidase system. It also acts by inhibiting the enzyme 5′-deiodinase, which converts T4 into the more active T3 form.
It should also be noted that beta-blockers (e.g. propanolol) are indicated in hyperthyroidism for relief of some of the symptoms such as increased heart rate, tremor, etc.
Glaucoma
Glaucoma is an eye condition in which the optic nerve becomes damaged due, in the main, to an increase in intraocular pressure (IOP). The increase in IOP results from problems with the eye ’s drainage system. In the eye, aqueous humour is found in the part of the eye in front of the lens, and this is continually produced in the ciliary body. Under normal conditions the aqueous humour is removed from the eye by means of drainage into the bloodstream through the trabecular meshwork and canal of Schlemm. In the normal forms of glaucoma these drainage systems become restricted or blocked and thus the IOP increases. There are several types of glaucoma: primary open-angle glaucoma (POAG) and closed-angle glaucoma (COAG) account for the majority of cases, although there are other types such as normal tension glaucoma, secondary glaucoma, paediatric glaucoma and acute glaucoma.
Primary open-angle glaucoma
In primary open angle glaucoma the drainage channels in the trabecular meshwork pathway become clogged. This prevents aqueous humour outflow and an imbalance occurs because fluid continues to be produced but is unable to drain away. This results in increased intraocular pressure (IOP). The intraocular pressure exerts force on the optic nerve at the back of the eye, resulting in damage, and can lead eventually to blindness.
Closed-angle glaucoma
In closed-angle glaucoma (angle-closure glaucoma) the angle between the iris and the cornea changes and completely restricts the drainage, resulting in a rapid increase in IOP and thus optic nerve damage.
There is no cure for glaucoma, although surgical procedures can be performed to relieve IOP. Treatment of glaucoma involves the use of drugs in order to reduce IOP and there are a number of different drug classes that might be employed. These are listed in Table 20.1.
Table 20.1 Details of drug classes used in the treatment of glaucoma
| Drug class | Mode of action |
|---|---|
| Beta-blockers | Reduce the production of aqueous humour by blocking the messages which stimulate fluid production. |
| Carbonic anhydrase inhibitors |
Produce a fall in pressure by suppressing production of aqueous humour. |
| Prostaglandin analogues | Increase the aqueous outflow from the eye. |
| Sympathomimetics | Decrease intraocular pressure by increasing the outflow of fluid. May be used in conjunction with a miotic. |
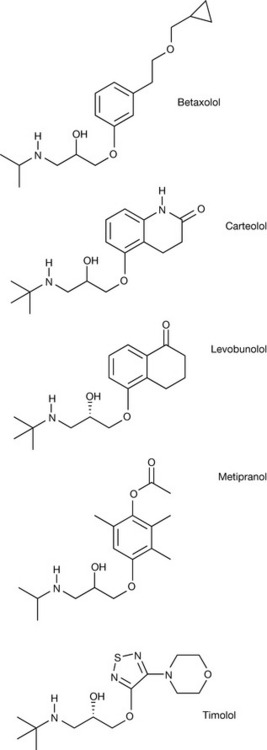
The use of drugs in the treatment of glaucoma is designed mainly to reduce the production of aqueous humour or to improve the outflow of aqueous humour once produced. Beta-blockers commonly used for the treatment of glaucoma include betaxolol, carteolol, levobunolol, metipranolol and timolol (Fig. 20.14). This group of compounds acts by causing a decrease in production of the aqueous humour in the ciliary body.
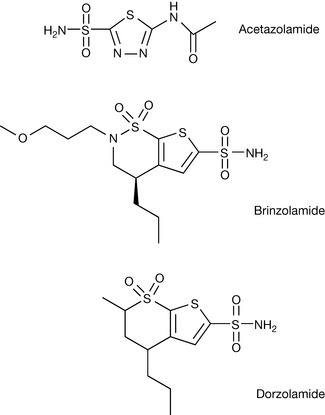
Carbonic anhydrase (CA) inhibitors acetazolamide, brinzolamide and dorzolamide (Fig. 20.15) act through inhibition of carbonic anhydrase in the eye. These compounds all have the typical primary sulphonamide group associated with CA inhibition (see Ch. 4). CA (probably CA II which is the predominant isoform in the eye) catalyses the formation of bicarbonate, which is involved in production of aqueous humour. The inhibition of this enzyme thus reduces the production of the aqueous humour and consequently results in a decrease in IOP.
Several prostaglandin F2α (PGF2α) analogues have been developed for the treatment of glaucoma, including latanoprost, bimatoprost and travoprost (Fig. 20.16). These compounds are prodrugs of the active compounds that are hydrolysed to the active free acids. These compounds act by increasing uveoscleral outflow (drainage from the anterior chamber, through the ciliary body and choroids) of the aqueous humour from the eye. Although this process does occur normally it is a relatively minor pathway until promoted by these PGF2α analogues. As indicated above, these analogues are prodrugs that are hydrolysed by ocular tissue, e.g. cornea. The prodrug portions of the compounds are highlighted in Figure 20.16.
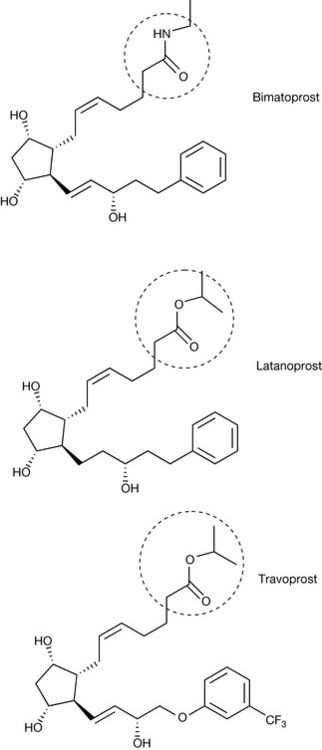
Figure 20.16 Prostaglandin (PGF2α) analogues used for the treatment of glaucoma (circled areas indicating prodrug moiety).
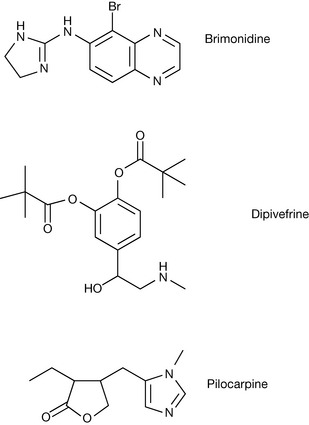
Sympathomimetics and miotics (Fig. 20.17) have been used for the treatment of glaucoma although they are less widely used today given the advent of the drug classes reported above. The sympathomimetic dipivefrine is the dipivalyl prodrug of adrenaline. As such it is highly lipophilic, resulting in better penetration than adrenaline itself. The prodrug is hydrolysed by esterases in the aqueous humour. Adrenaline thus formed acts in a non-selective fashion on α and β adrenoreceptors, which can lead to opposing effects. However, the cumulative effect is to reduce the IOP. Brimonidine is a highly selective σ2 agonist, which reduces IOP by reducing aqueous inflow and increasing uveoscleral outflow. Pilocarpine acts on a muscarinic receptor (M3) on the iris sphincter muscle, which causes muscle contraction, resulting in the opening of the trabecular meshwork. This results in an increase in the rate at which aqueous humour leaves the eye, thereby reducing IOP.
Steroid hormones
One of the three classes of hormones is the steroid hormones. These hormones are synthesised in the adrenal glands and in the testes/ovaries. They are transported throughout the body where they control a variety of different physiological functions. The steroid hormones include the glucocorticoids (glucocorticosteroids) involved in metabolism, inflammation and stress; mineralocorticoids (mineralocorticosteroids) involved in control of water/salt balance; androgens, oestrogens and progestogens which are the sex hormones that are involved in the development of male and female secondary sexual characteristics and in reproduction.
Steroid biosynthesis
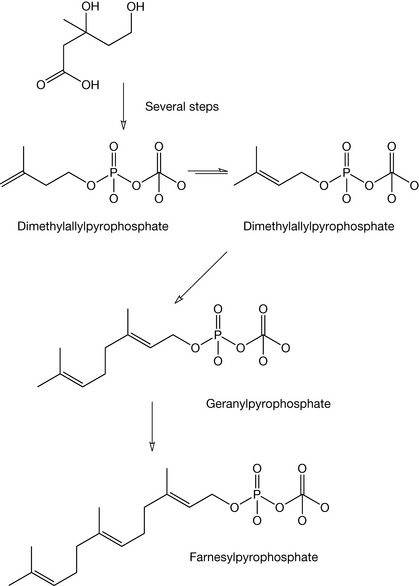
All of the steroid hormones are derived from cholesterol. Cholesterol biosynthesis is the major route through which steroids (including steroid hormones) are formed in the body. Cholesterol biosynthesis proceeds initially via the mevalonic acid (mevalonate) pathway (Fig. 20.18). Two molecules of acetyl coenzyme A [acetyl CoA] combine to generate acetoacetyl CoA. This reacts with a further molecule of acetyl CoA to generate 3-hydroxy-3-methylglutaryl CoA (HMG CoA). HMG CoA is reduced by HMG CoA reductase to yield mevalonic acid. Note this last step is most important since it is the inhibition of this enzyme system which is the basis of the cholesterol lowering effect of statins.
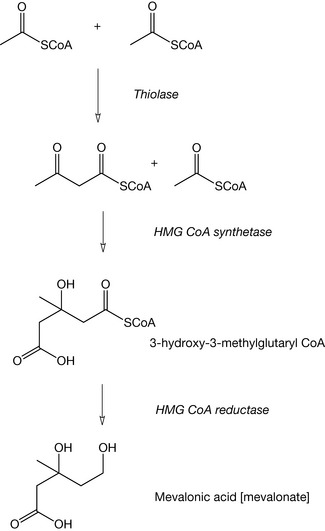
Figure 20.18 The initial steps in the biosynthesis of cholesterol leading to mevalonic acid formation.
The second step in the biosynthesis of cholesterol is the conversion of mevalonic acid into farnesylpyrophosphate (Fig. 20.19). This is initiated by phosphorylation of the mevalonic acid, followed by decarboxylation yielding isopentylpyrophosphate, which can reversibly isomerise to 3,3-dimethylallylpyrophosphate.
Condensation of this compound with one molecule of isopentylpyrophosphate results in the formation of geranylpyrophosphate, and addition of a second molecule of isopentylpyrophosphate yields farnesylpyrophosphate (Fig. 20.19).
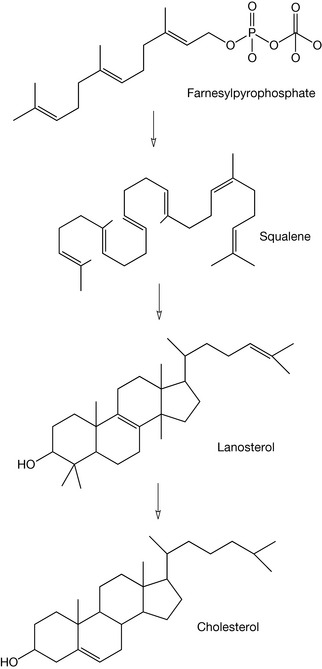
The third phase of cholesterol biosynthesis (Fig. 20.20) involves the condensation of two molecules of farnesylpyrophosphate, yielding squalene via the enzyme squalene synthetase.
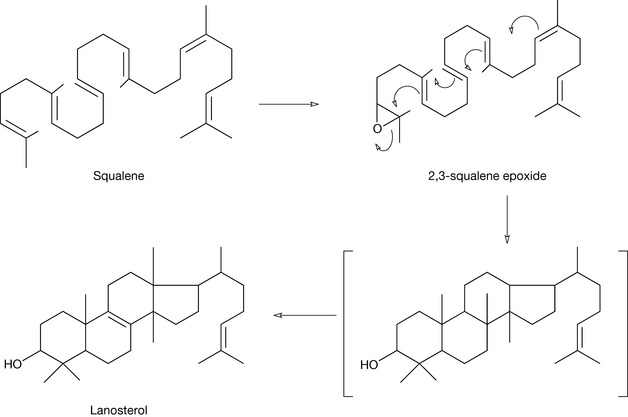
Squalene is essentially an open chain form of the steroid nucleus, which is converted into cholesterol via a number of steps involving the production of lanosterol. Figure 20.21 shows more clearly the cyclisation process in which squalene is firstly oxidised via squalene epoxidase to form 2,3-squalene epoxide. This then cyclises to produce the basic steroid nucleus, resulting in the formation of lanosterol. This is why all steroids have an oxygen atom at the 3 position.
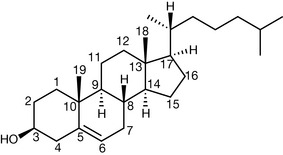
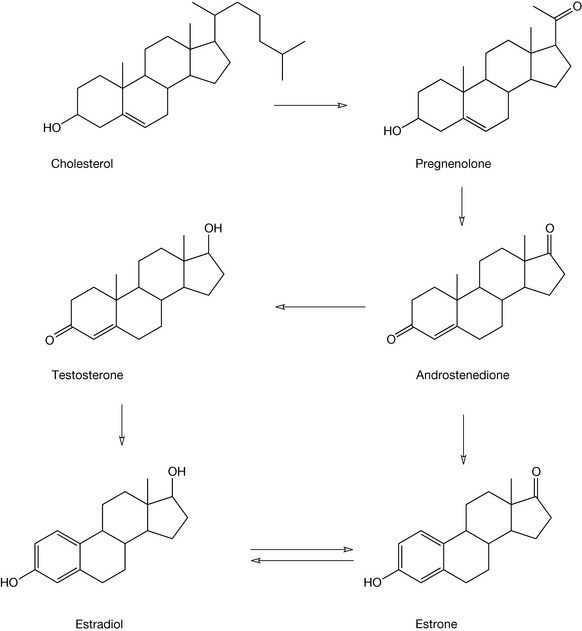
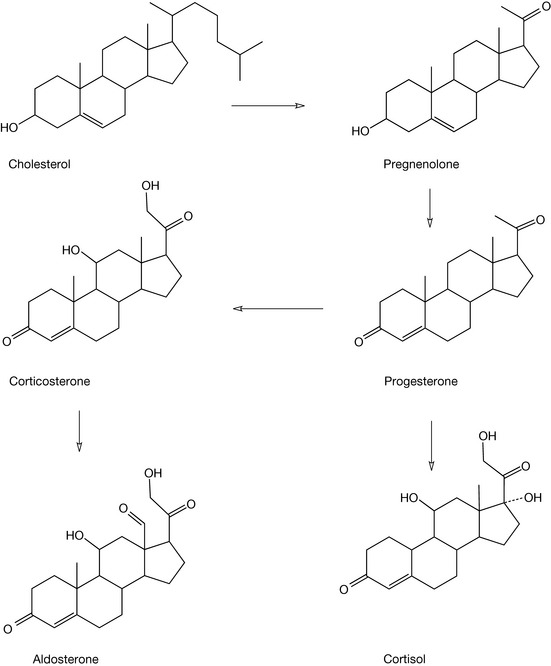
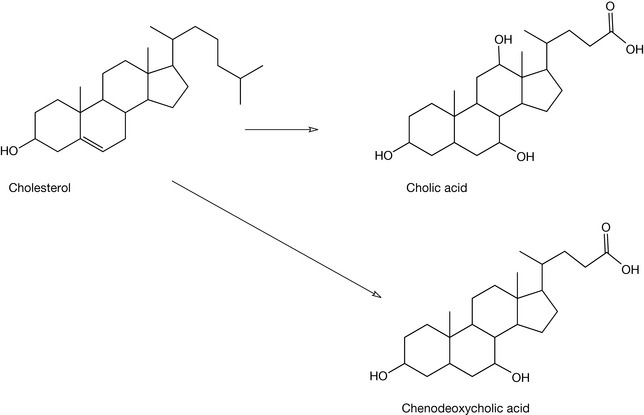
Once formed, cholesterol may be utilised in the production of a variety of steroid hormones in the body. The numbering of the steroid nucleus in cholesterol (which is valid for all steroids) is shown in Figure 20.22 and this is followed with details of the major pathways to the steroid hormones (Figs 20.23, 20.24) whilst Figure 20.25 shows the formation of bile acids from cholesterol.
The biosynthetic pathway for the conversion of cholesterol into these compounds involves a range of enzymic systems, many of which are cytochrome P450 (CYP) enzyme systems. Thus, in the conversion of cholesterol into pregnenolone, CYP11A1 (the side chain cleavage enzyme or desmolase) is responsible for loss of the six carbon chain and the C20 ketone formation. Conversion of pregnenolone into androstenedione involves CYP17 (steroid 17-alpha-hydroxylase), which brings about the formation of the ketone at the C17 position. The oxidation of the 3-hydroxyl group occurs via the non-CYP enzyme 3-beta-HSD (3-beta-hydroxysteroid dehydrogenase). During this process the rearrangement of the 5,6 double bond occurs because of the formation of the energetically favourable ene-one system. The reduction of the C17 ketone to a hydroxyl group results in the formation of testosterone via 17-beta-HSD. Formation of estrone and estradiol involves the aromatisation of the A ring via CYP19 (aromatase) enzyme.
In Figure 20.24, the formation of the corticosteroids is outlined. The formation of progesterone results from the action of 3-beta-HSD on pregnenolone. The formation of cortisol from progesterone involves a series of three hydroxylation steps with hydroxylation of C17 (CYP17), hydroxylation of C21 (CYP21) and hydroxylation of C11 (CYP11B1). The biosynthesis of aldosterone shares two of the hydroxylation steps but in the final step formation of the C18 aldehyde function occurs via CYP11B2 (aldosterone synthetase).
The sex hormones
The three groups of sex hormones in the human body are the oestrogens and progestogens, often thought of as the ‘female’ sex hormones, and the androgens, the ‘male’ sex hormone. Despite being classified in such a way, these hormones are present in both males and females, although the levels present in the two sexes are somewhat different. These hormones are responsible for the development of secondary sexual characteristics (e.g. breast development, vaginal and uterine growth in females and penis development, growth of body and facial hair in males) and are vital for reproduction in both sexes.
As far as the endocrine system is concerned, these sex hormones are produced in the ovaries and testes (gonads) following events in the hypothalamus and the anterior pituitary gland. In the hypothalamus, messages received via feedback loops signal the need for the synthesis of these hormones and, as a result, it secretes gonadotrophin releasing hormone (GnRH). In the anterior pituitary gland, GnRH is responsible for stimulating the production of gonadotrophins, e.g. luteinising hormone (LH) or follicle stimulating hormone (FSH). These hormones travel to the ovaries and the testes where the production of oestrogens, progestogens and androgens occurs. As indicated above, these hormones are involved in the development of the secondary sexual characteristics and, during puberty, the development into the mature adult, and then subsequently influence the sexual functioning of adults in terms of spermatogenesis in males and the reproductive cycle in females. The general biosynthesis of the steroid hormones in the body is detailed in Figures 20.23 and 20.24 and the production of the main steroid hormones estradiol, progesterone and testosterone along with estrone outlines there. A third oestrogen, estriol, is derived from either estradiol via 16α-hydrolase or estrone, again via 16α-hydrolase, and then estradiol dehydrogenase.
During the female reproductive cycle which falls into two phases, the follicle cycle and the luteal cycle, the hormones FSH, LH, estradiol and progesterone are all involved in the various changes that occur over this period. FSH, as the name implies, stimulates the development of follicles in the ovary. As this process progresses, estradiol is secreted leading to an elevation of oestrogen levels. By means of a feedback loop, the production of FSH decreases and there is a corresponding increase in LH levels and at the midpoint of the cycle there is a sharp rise in its production leading to the release of an egg from the follicle (ovulation) into the fallopian tube. The second part of the reproductive cycle is known as the luteal phase in which the empty follicle changes into a corpus luteum, which secretes progesterone under the influence of LH. If the egg is not fertilised, the rising levels of progesterone result in the inhibition of GnRH, leading subsequently to a drop in progesterone levels and finally to menstruation. If the egg is fertilised, a set of different occurrences take place with the formation of human chorionic gonadotrophin (HCG), which enables the corpus luteum to survive and as such allows progesterone production to continue, thereby supporting the pregnancy.
In males, once again there are processes that involve FSH and LH that are involved in the production of testosterone and the production of sperm.
Steroid hormones used as medicinal substances
There are numerous examples of the use of natural or synthetic steroids in a variety of medical areas that have gained importance. These include oral contraception, hormone replacement therapy, treatment for delayed puberty and muscle development, with the oral contraceptives one of the most well-known examples.
Development of oral contraceptives
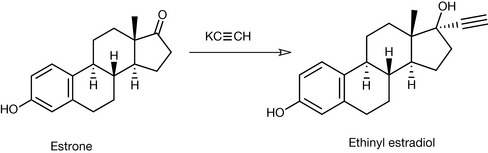
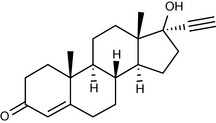
Early work in the understanding of steroid hormones and how they work resulted in investigations of their use as contraceptive agents. It was soon shown that many of the naturally occurring compounds, when given parenterally, were active, but when administered orally were only weakly active. It was found that the compounds were absorbed reasonably well but were rapidly metabolised by the liver. In order to overcome these problems of rapid metabolism, a large number of derivatives were synthesised and tested. One of the most successful methods discovered was the introduction of an acetylene group at the 17 position of the steroid ring, for example in ethinyl estradiol (Fig. 20.26). This compound was easily synthesised by reacting estrone with potassium acetylide in liquid ammonia.
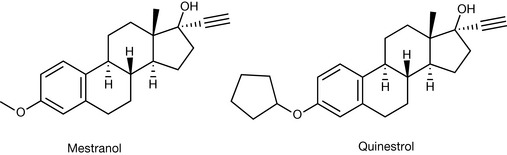
Further investigations resulted in the discovery that etherification of the 3-hydroxy group of ethinyl estradiol resulted in formation of potent orally active oestrogens such as mestranol (the 3-methyl ether) and quinestrol (the 3-cyclopentyl ether), although this is not now used in oral contraceptives (Fig. 20.27).
In a similar fashion to the oestrogens, synthetic progestogens were investigated and a large number were synthesised. One of the first to be used was ethisterone, which once again incorporated an ethinyl group at the 17 position (Figs 20.28, 20.29, 20.30).
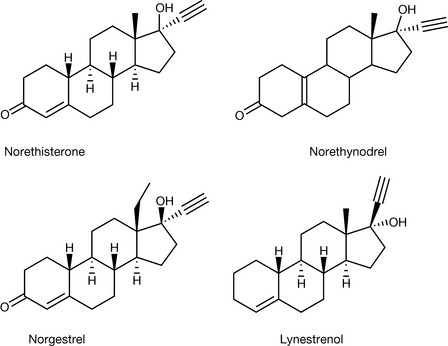
Since the early work in this area, there have been many compounds discovered that possess significant oral progestogenic activity. For example, it was discovered that removal of the C19 methyl group enhanced the activity, resulting in compounds such as norethindrone and norethinodrel. Also, compounds in which the 3-keto group is lost were also found to be useful progestogens.
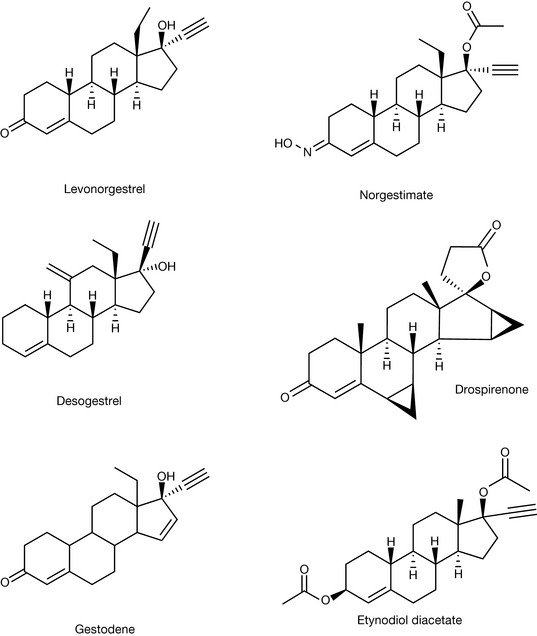
There have been many developments in formulations used for oral contraceptives since the original studies. Nowadays, there are numerous products available such as the combined hormonal therapy (oestrogen plus progestogen) or the progestogen-only contraceptive. In some instances, the combined tablets contain fixed doses of the two components and others where the contents vary. There are also tablets containing lower than normal levels of the oestrogen because of the risk factors associated with circulatory disease. Most of the oral contraceptives available in the UK at present comprise estradiol in combination with one of the following: norethisterone, levonorgestrel, norgestimate, desogestrel, drospirenone, gestodene, as well as a product which contains mestranol with ethisterone. The progestogen-only pills available contain desogestrel, etynodiol diacetate, norethisterone or levonorgestrel. An emergency contraception product is also available which contains a high dose of levonorgestrel.
Hormone replacement therapy
In addition to their use as oral contraceptives, oestrogens and progestogen, either in combination or as oestrogens only treatments, have been used as hormone replacement therapy to alleviate the problems associated with the menopause. There are concerns about such treatments because of the side effects known to be associated with these products, e.g. increased risks (in some or both) of heart attack, stroke, blood clots, certain cancers, whilst both treatments seem to have a positive effect in relation to osteoporosis.
Male sex hormones
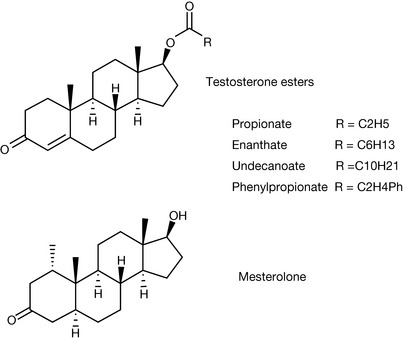
Anabolic or androgenic steroids are used for the treatment of a variety of conditions including their use in treating young males with delayed puberty, treating patients with severe weight loss due to some form of trauma or muscle wasting resulting from, for instance, HIV and in replacement therapy. In such circumstances the formulated product contains testosterone or testosterone esters such as the propionate, enantate [enenthate], undecanoate or mesterolone (Fig. 20.31).
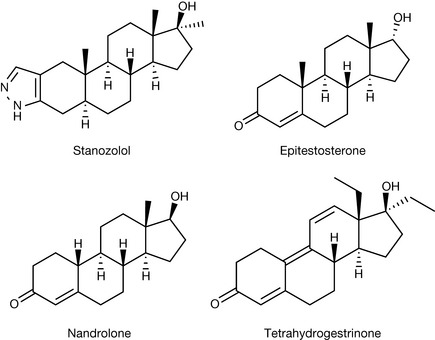
In addition to the use of androgenic anabolic steroids for medical purposes, there is considerable interest in these compounds by sportsmen and women as performance-enhancing drugs. One of the most well-known cases involved Ben Johnson, who was found to have been taking stanozolol. Diana Modahl was found to have testosterone levels that were well above the allowed limits and also her testosterone/epitestosterone ratio was also high. However, when the reserve sample was tested it was discovered that there were difficulties with the sample and the case was dropped. Dwain Chambers was found to have taken tetrahydrogestrinone, and Greg Rusedski was found to have taken nandrolone. However, he was subsequently cleared because of what was thought to be a problem with supplements supplied by the Association of Tennis Players (and taken by a large number of tennis players who also proved positive) that appears to have been contaminated with this steroid.
The cyclist Floyd Landis was also found to have high testosterone/epitestosterone levels. The structure of some of these steroids is shown in Figure 20.32. Stanozolol and nandrolone have been used for conventional medical purposes and epitestosterone is the 17-hydroxy epimer of testosterone that is present naturally in the human body. These, along with many other anabolic steroids, have been used in sport and by bodybuilders on the basis that these compounds are capable of increasing muscle mass and increasing their competitive nature. The problem is that although the use of such compounds may be relatively safe at the levels used for conventional medical treatment, their use as performance-enhancing drugs often involves much higher doses, and it has been suggested that these doses may well lead to serious side effects such as hepatotoxicity, hepatitis and the risk of tumour growth.
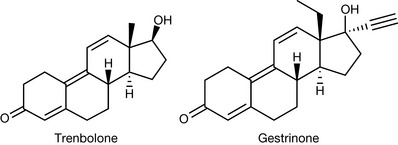
Tetrahydrogestrinone achieved fame as one of the ‘designer’ steroids produced by the American company Bay Area Laboratory Cooperative (BALCO) in California. The chemists in the company studied known agents with anabolic activity to develop new agents that would be unknown to the drug testing laboratories. In the case of tetrahydrogestrinone, the chemists worked on the production of this designer drug based on knowledge of the activities of trenbolone and gestrinone (Fig. 20.33).
The compound was synthesised by the palladium charcoal catalysed hydrogenation of the acetylenic group of gestrinone. This compound was used for a number of years by athletes and it was undetected for a number of years until a sample was sent to Don Catlin, the director of the drug testing laboratory at UCLA, who, along with his team there, developed analytical methodology for the detection and quantification of this steroid. There are likely to be more such ‘designer’ compounds emerging and thus there is a constant need for vigilance by the drug testing agencies.
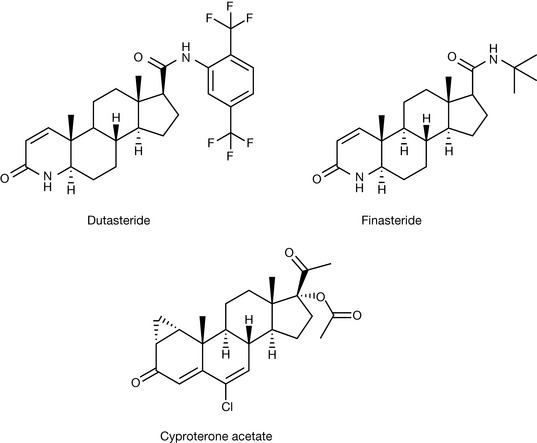
One final area where steroids are used is as anti-androgens in the treatment of benign prostatic hyperplasia, prostate cancer and male hypersexuality. Dutasteride and finasteride (Fig. 20.34) are specific inhibitors of 5α-reductase, which is involved in the metabolism of testosterone. These two compounds are indicated for the treatment of benign prostatic hyperplasia, with finasteride also being indicated for male-pattern baldness.
Cyproterone acetate is an anti-androgen, which is indicated for the treatment of prostate cancer and male hypersexuality. It is an inhibitor of 21-hydrolase.
Glucocorticosteroids and mineralocorticosteroids
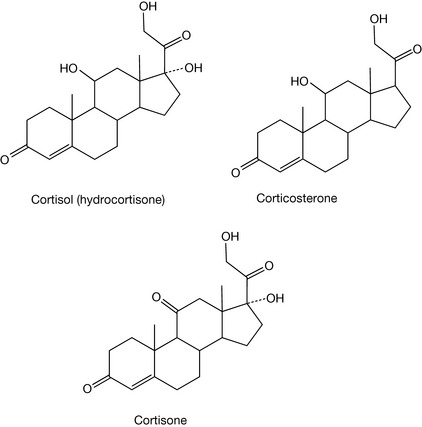
The glucocorticosteroids and mineralocorticosteroids are produced in the adrenal gland. The major glucocorticosteroids are cortisol (hydrocortisone), cortisone and corticosterosterone. The natural glucocorticosteroids are important because of their involvement in the control of metabolism of fats, carbohydrates and proteins (Fig. 20.35). Additionally, they play important roles in inflammation, the immune system and in the stress response. Production of these hormones is under the control of adrenocorticotrophic hormone (ACTH), which is secreted by the pituitary, and this influences the adrenal gland to produce cortisol, etc.
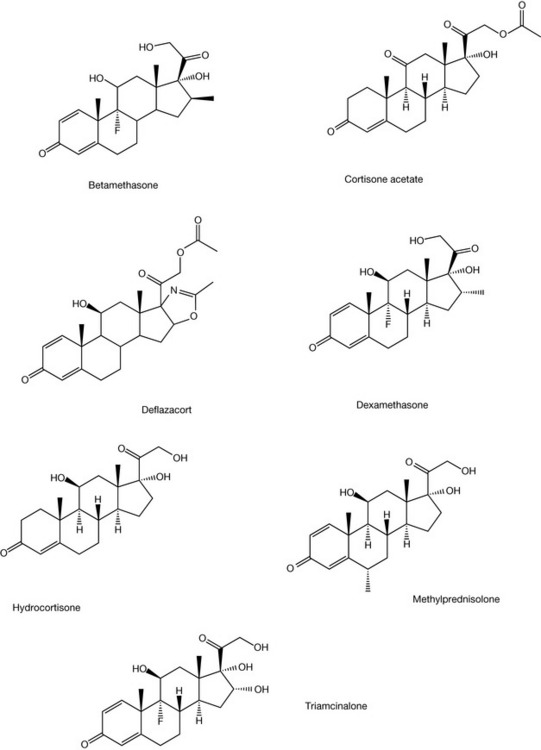
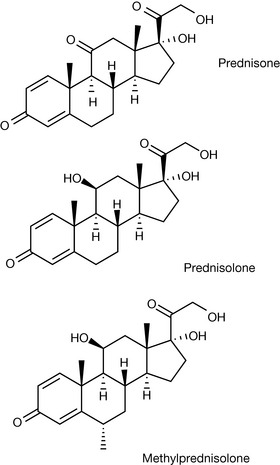
Synthetic glucocorticoids are indicated for a wide variety of conditions including rheumatoid arthritis, asthma, ankylosing spondylitis, lupus erythematosus, inflammatory bowel disease, dermatitis, allergic reaction, etc. These steroids are available in a variety of formulations allowing for oral, topical, inhalation and intravenous forms. Typical examples of synthetic glucocorticoids include betamethasone, cortisone acetate, deflazacort, dexamethasone, hydrocortisone, methylprednisolone and triamcinalone (Fig. 20.36). As can be seen, this group of compounds are structurally similar, with the modification of several positions having a major bearing on the activity. The introduction of unsaturation at C1-2 increases the glucocorticoid activity as does the ethyl group at C16. The introduction of the fluorine atom increases glucocorticoid activity but also increases mineralocorticoid activity as well. Various other modifications have also been shown to have significant effects on the activity. For example, C21 ester formation and acetonide formation via C16, and 17 hydroxyl, as in triamcinolone acetonide, aids in the topical activity of these steroids through changes in the lipophilicity, improving absorption by the skin.
The main mineralocorticosteroid is aldosterone (see Fig. 20.24), which plays an important role in the renin-angiotensin system. As a result of various possible stimuli, e.g. hypotension, decreased sodium levels, etc., the renin-angiotensin system is activated and renin is produced, and this enzyme is responsible for the conversion of angiotensinogen into angiotensin. Angiotensin is responsible for the stimulation of the adrenals to produce aldosterone. Aldosterone is then responsible for the retention of sodium in the kidneys and an increase in fluid volume within the body. Unlike most steroids, it binds to membrane receptors rather than as part of a protein receptor complex to DNA. There are a number of disease states (hypoadrenalism, Addison ’s disease) where the adrenal glands fail to produce aldosterone (and cortisone), and replacement therapy is required. In the case of aldosterone, it is not possible for it to be used directly in replacement therapy because it is too unstable, and fludrocortisone acetate (Fig. 20.37) is used in its place.
Steroids used in the treatment of asthma
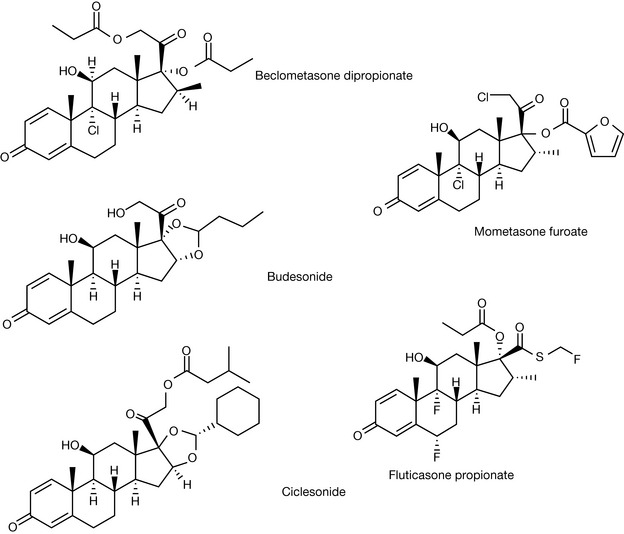
Asthma is a disease state in which the airways in the lungs become inflamed and narrow, thus causing difficulty in breathing. Reports suggest that in the UK and the USA approximately 7–10% of the population are in receipt of a prescription for asthma drugs. Asthma is a reversible condition whereby, in response to a trigger, the asthmatic attack is initiated; this trigger may be an allergen, an environmental trigger (chemical, vapour, tobacco smoke, etc.) or exercise. There are numerous drugs used in the treatment of asthma including inhaled and oral corticosteroids, β2 agonists (both long acting (LABAs) and short acting), leukotriene modifiers and bronchodilators. In some instances, corticosteroids and bronchodilators are combined in an inhaler. Glucocorticosteroids are used in the treatment of asthma because of their anti-inflammatory activity. The structures of inhaled and oral corticosteroids used in this area are detailed in Figures 20.38 and 20.39.
The corticosteroids are active anti-inflammatory agents via their involvement in the arachidonic acid cascade, which leads to the formation of prostaglandins and leucotrienes which are responsible for the inflammatory response (see Ch. 2). The arachidonic acid is formed by the breakdown of phospholipid membranes, which is brought about by the enzyme phospholipase A2. The corticosteroids are thought to be involved in the stimulation of the synthesis of proteins (lipocortins) that are able to inhibit the action of phospholipase A2, thus reducing the production of arachidonic acid.
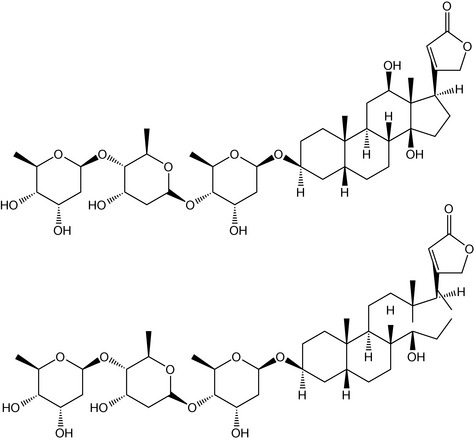
The cardiac glycosides
Cardiac glycosides, such as digoxin and digitoxin (Fig. 20.40), are indicated for the treatment of heart failure and supraventricular arrhythmias. These two compounds, which are naturally occurring compounds, are defined as glycosides since they are comprised of a sugar portion, the glycone moiety, and the aglycone portion, which in this case is a steroid (other glycosides exist, e.g. apterin which is a coumarin glycoside). These cardiac glycosides are obtained from species of digitalis, digoxin from Digitalis lanata and digitoxin from Digitalis purpurea. These compounds are cardenolides indicating that they contain an unsaturated butyrolactone ring system.
The mode of action of the cardiac glycosides is via their inhibitory action on the system Na-K-ATPase, which eventually results in increased intracellular calcium levels. These increased calcium levels result in a series of events that produce an increase in the strength of contraction of the heart (positive inotropic effect) and a reduction in the heart rate (negative chronotropic effect).
Katz M., Gans E.H. Topical corticosteroids, structure-activity and the glucocorticoid receptor: Discovery and development – A process of planned serendipity. J Pharm Sci. 2008;97(8):2936-2946.
Hormones of the reproductive system
http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/S/SexHormones.html.