CHAPTER 11 Pancreas, biliary tract and intra-abdominal organs
Clinical aspects
In the developed world, the incidence of pancreatic cancer has risen threefold since the 1920s. Prognosis is very poor, this tumor accounting for almost a quarter million deaths annually worldwide. In the USA, cancer of the pancreas is now the fourth leading cause of cancer deaths in male and females. The majority of patients have irresectable disease or metastases at the time of diagnosis. Therapeutic options are limited.1 A diagnostic test such as fine needle biopsy (FNB), which combines accuracy with minimal intervention, is of unchallenged value in these patients.
Ultrasonography (US) and computed tomography (CT) made mass lesions of the pancreas, biliary tree and elsewhere in the abdomen readily accessible to FNB. Teams in Sweden and Denmark were the first to use FNB, guided by angiography or US, to investigate pancreatic masses.2,3 Endoscopic evaluation of pancreatic and biliary disease has long been part of the work-up of patients with obstructive symptoms or mass lesions, with additional retrograde imaging studies and/or the collection of pancreatobiliary fluid or brushing specimens. However, the recent addition of a fine needle biopsy channel and linear ultrasonographic functions to the endoscope, have made this the method of choice in the early assessment of patients with pancreaticobiliary symptomatology. There are several good reviews,4-6 and many recent series attest to the value, efficacy and low complication rate of endoscopically performed, ultrasonographically directed (EUS) procedures.7-14 An alternative method of obtaining a tissue diagnosis is by transduodenal FNB at laparotomy, performed with curative intent, or for surgical bypass of obstructive jaundice.
As confidence in cytological diagnosis has become well established, there is an onus on the cytologist to provide specific information on tumor type as a basis for therapeutic decisions. This has led to use of supplementary techniques such as cell blocks, EM, immunocytochemistry, and analysis of pancreatic cyst fluid.4-6,15,16 Core needle and wedge biopsies are generally discouraged in the pancreas owing to the propensity for fistula formation, spill of lytic enzymes, fat necrosis of peripancreatic tissues, and tumor track spread (see Complications below).
The place of FNAC in the investigative sequence
Fine needle biopsy of a pancreatic mass is performed after imaging has defined site, dimensions, anatomic relationships, depth and solid or cystic nature. This information is important not only for the biopsy procedure and its safety but also for the subsequent interpretation of the smears. The biopsy should be performed by the radiologist and the pathologist in close cooperation. EUS procedures are done by the endoscopist, usually a gastroenterologist. Again, the presence of a cytologist in the endoscopy suite to effect rapid evaluation is highly advisable. Cytological examination may unexpectedly reveal a benign neoplasm, an islet cell tumor, a metastatic malignancy or a lymphoma instead of the presumed pancreatic adenocarcinoma, with its inherently poorer prognosis. If pancreatic cancer is diagnosed microscopically, staging will determine further management. Unresectable or metastatic lesions may require stent insertion for relief of obstructive jaundice. Stent placement may be performed after FNB, at the same endoscopic procedure. If deemed operable, cancer cases can undergo subsequent surgery with the diagnosis and staging already accomplished.17
FNB does not contribute significantly to early diagnosis of pancreatic cancer. Neither is it suited, generally speaking, to the diagnosis of pancreatitis. Inflammatory pancreatic masses, conversely, constitute the chief clinical differential diagnosis of pancreatic neoplasia and must be recognisable to the cytopathologist.18,19 In pancreatic pseudocyst, FNB can be both diagnostic and therapeutic.15,20,21 Decompression of an acutely developed cyst may relieve symptoms and facilitate surgical treatment. Some patients are subjected to laparotomy without prior FNB in anticipation of resectability or for biliary bypass palliation to alleviate symptomatic jaundice. Under these circumstances, the FNB can be undertaken easily and with high precision intraoperatively.22-24A FNB at operation is done through the wall of the opened duodenum for pancreatic head masses. It has the advantage over the traditional wedge biopsy for frozen section of being virtually free of complications such as hemorrhage and fistula formation. It can be repeated to sample many different parts of a large mass, thereby increasing the probability of obtaining representative material. A thin needle can pass through the stomach or duodenum without risk. Rapid on-site staining and interpretation of intraoperative smears is quicker than cutting and staining frozen sections. With experience, interpretation of technically satisfactory smears is often easier than that of frozen section because of better preservation of cell detail.
Mass lesions of the gastrointestinal tract are, as a rule, investigated by radiological imaging and by endoscopy. FNB can be done percutaneously or endoscopically, an approach well-suited to submucosal neoplasms, and to lymph nodes adjacent to the bowel.6,25,26 Endoscopically visible lesions in the upper or lower gastrointestinal tract are better sampled by exfoliative means, such as brushing, or by tissue biopsy. The cytopathology of exfoliative gastrointestinal and pancreatobiliary brushings is beyond the scope of this book.
Accuracy of diagnosis
Diagnostic accuracy is related primarily to the adequacy and representativeness of the biopsy, which in turn depends on the site, size and nature of the lesion, and on the expertise of the operator. The relative merits of available radiological techniques in guiding the biopsy are discussed in Chapter 3. The requirement for technical expertise is greatest for EUS-FNA, the success of which is almost entirely operator dependent. Although technical progress continues to improve the quality of radiological tumor imaging, there is still a minimum size of approximately 1.0 cm below which a lesion cannot be clearly demonstrated by traditional US or by CT, and precise needling is not possible. EUS appears to have the capability of detecting lesions of 0.3–1.0 cm. Intraoperatively, under the control of direct palpation, lesions measuring only a few millimeters in diameter can be biopsied successfully. Very large tumors present the difficulty of identifying viable areas from which to obtain well-preserved cells, in a background of extensive necrosis or hemorrhage.
Diagnostic specificity for malignant pancreatic lesions is 100% in nearly all published series. However, occasional false-positive diagnoses have been reported in cases of chronic pancreatitis or in the presence of pancreatic intraepithelial neoplasia (PanIN).19,27 Diagnostic sensitivity is more variable, particularly in series of US- or CT-directed percutaneous biopsies with reported sensitivity of between 50% and 90%.19,28-33 Intraoperative FNB achieves sensitivities in excess of 90%.22-2434 Recent series of pancreatic EUS-FNA report sensitivies of between 90% and 100%.9,11,12,14,35-39 Factors influencing the sensitivity include pre-analytical issues such as: the size of the lesion, whether the lesion is solid or cystic, the experience of the operator, the number of needle passes, and the availability of on-site assessment by a cytologist or cytopathologist,4 as well as analytical issues. It has been reported that repeat EUS-FNA in indeterminate cases further improves the sensitivity.39,40
Many of the cited studies focus on solid lesions of the pancreas and it is acknowledged that the sensitivity and specificity in diagnosis of cystic lesions is lower.4 Biochemical analysis of fluid from cystic lesions improves the sensitivity and specificity of these lesions over cytologic assessment alone (see Intraductal papillary – mucinous and mucinous cystic neoplasms below).15,41-44
Complications
Significant complications are rare if thin needles of 0.08 mm (21 gauge) or less are used. Such needles pass through stomach or bowel without causing peritonitis, although caution is recommended in cases of bowel obstruction or distension. In a comprehensive review of the literature, and a questionnaire study, Smith documented six deaths after pancreatic FNB.45 Of these, five were due to pancreatitis and one followed sepsis post aspiration of a pancreatic pseudocyst. In an earlier review of 184 procedures, Mueller and colleagues reported a 3% incidence of (nonfatal) severe pancreatitis.46 All of the cases developing pancreatitis in either series had inflammatory mass lesions mimicking carcinoma. Hence, FNB is not recommended for the investigation of classic clinical pancreatitis but should be reserved for cases with a radiologically localized lesion. Major hemorrhage is very rare. The coagulation status should be noted but a full investigation is unnecessary unless the spleen or the liver is involved. A recent study of 1034 pancreatic EUS-FNA identified 10 hemorrhages (0.96%), none of which was fatal, two cases of acute severe pancreatitis (0.19%) and one duodenal perforation (0.09%) which lead to a post-surgical death.47 Two cases of pancreatic ascites due to fistula formation have received literature attention.48,49 Both fistulas closed with regression of the ascites.
Tumor seeding in the needle track has been reported in a small number of cases following FNB of pancreatic carcinoma. In a literature review by Smith, the risk of needle track seeding was 4.45 per 100 000 transabdominal FNB procedures in four questionnaire studies of 156 652 patients.45 Five of 11 instances of needle track spread after FNB of the pancreas used a needle greater than 21 gauge. It is thus apparent that cutaneous needle track seeding is an extremely rare event. More frequently than is clinically realized, however, may be the tracking of cells to the peritoneal surfaces as a result of FNB. Some authors have reported a significant increase in incidence of exfoliated malignant cells within operative peritoneal washings after FNB,50 while others have failed to confirm this.51 More recently, it has been shown that EUS-FNA results in positive peritoneal cytology significantly less often than percutaneous FNB.52 The significance of this positive peritoneal cytology remains uncertain, with some authors finding that it was an indicator of unresectability, advanced disease, early metastasis and short survival,51 others identified a trend to decreased survival,53 while others did not show any survival disadvantage.54 The latter group concluded that cell spillage into the peritoneum at FNB did not render the procedure unsafe, particularly if neoadjuvant chemo-radiation was part of the management protocol. Clinicians wishing to minimise this potential hazard may prefer the transduodenal route of FNB offered by EUS, or by intraoperative sampling.
Technical considerations
Transperitoneal deep FNB is done as a hospital procedure and performed in the radiology or endoscopy departments where all the facilities for tumor imaging are available. The presence of a cytologist or cytopathologist in the endoscopy suite to effect rapid evaluation is highly advisable. Smears should be prepared and stained immediately so that the adequacy of the specimen can be checked and the aspiration repeated if necessary. A cell block should be made whenever possible to allow histochemical and immunocytochemical staining if necessary. These ancillary tests can also be performed on smears, either air-dried or wet-fixed, or liquid based preparations. If a lymphoid lesion is suspected, collection of a cellular specimen into RPMI or saline for flow cytometry is mandatory. Culture is rarely required in pancreatic FNB. Collection of fluid from cystic lesions for biochemical analysis should always be considered.
Cytological findings
The pancreas and biliary tract
Normal structures (Figs 11.1-11.3)
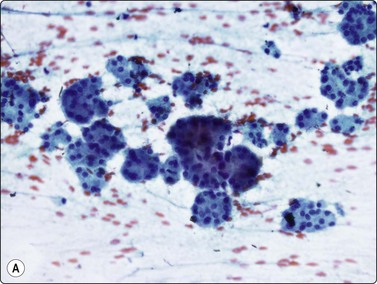
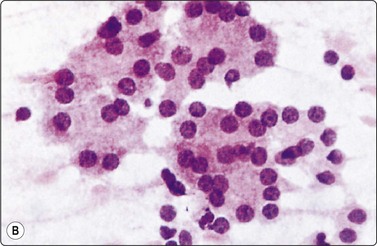
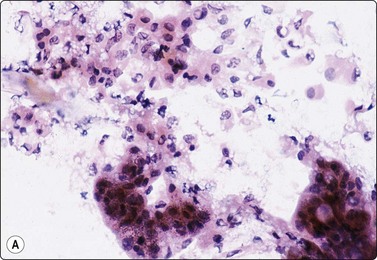
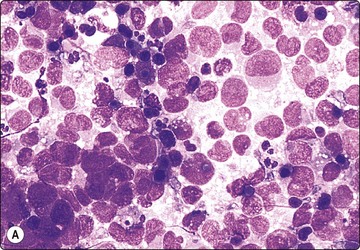
Fig. 11.1 Pancreatic acinar cells
(A) Cohesive clusters of exocrine epithelial cells forming rounded acini (Pap, IP); (B) Acinar cells showing granular cytoplasm and eccentric small round uniform nuclei (H&E, HP).
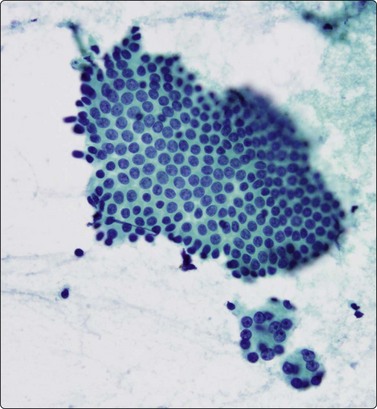
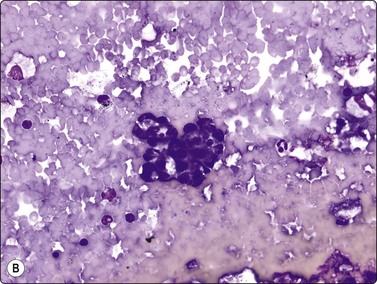
Fig. 11.2 Pancreatic ductal cells
Monolayered sheet of uniform ductal epithelial cells; note the adjacent clusters of benign acinar cells (Pap, HP).
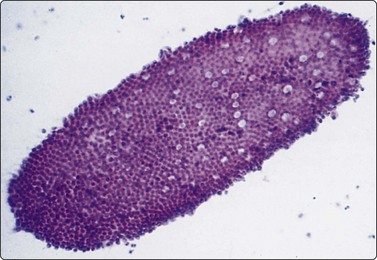
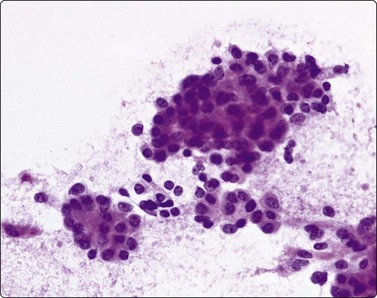
Fig. 11.3 Duodenal surface epithelium
Monolayered sheet of duodenal epithelial cells with interspersed goblet cells (Pap, LP).
Fine needle biopsy smears from normal pancreatic tissue can occasionally be surprisingly cellular. Most of the cells are acinar unless the needle traverses a major duct. Acinar cells have indistinct cell borders; their acinar arrangement suggests an individual triangular shape to the cells, the nuclei being disposed in a circle at the periphery of small, round clusters (Fig. 11.1). Larger fragments are composed of multiple acini held together by sparse fibrovascular stroma, often surrounding an intact duct. The nuclei are uniformly small, round and of similar size and shape to those of small lymphocytes. Many cells are represented by single, naked nuclei. The nuclear chromatin is densely granular and evenly distributed. There may be a single, prominent nucleolus or multiple small nucleoli. The cytoplasm is dense and granular. The ductal epithelial cells form monolayered sheets (Fig. 11.2). Cell borders are usually visible, the cytoplasm is pale and the nuclei are regularly spaced within the sheets. The nuclei are larger than those of the acinar epithelial cells and are more ovoid. They are paler and have finely granular chromatin and small, usually single, nucleoli. Palisading may be seen along the edge of sheets. Ductal epithelial cells from major bile ducts look similar and cannot be clearly distinguished from those of major pancreatic ducts in smears. Columnar cells of duodenal origin, however, although also in monolayers, are distinguished by the presence of goblet cells within the sheets (Fig. 11.3).
Pancreatitis (Fig. 11.4)
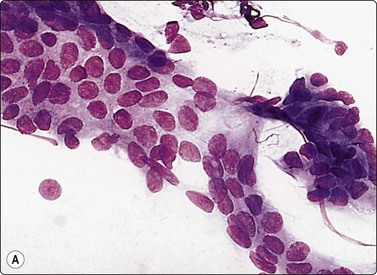
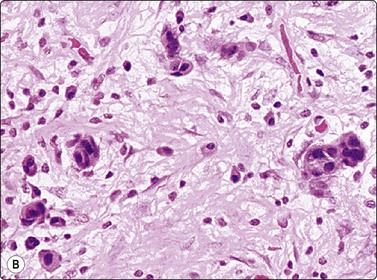
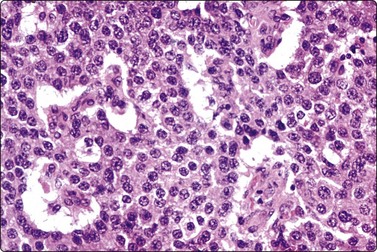
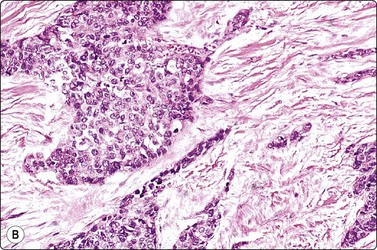
Fig. 11.4 Chronic pancreatitis, epithelial atypia
(A) Aggregate of glandular cells showing acinar pattern and prominent nuclear atypia (MGG, HP); (B) Corresponding tissue section; fibrous tissue with small islands of residual epithelium showing prominent atypia (H&E, HP).
Acute diffuse hemorrhagic pancreatitis is unlikely to be subjected to FNB. Percutaneous FNB is not a suitable method to confirm clinically suspected chronic pancreatitis, partly because the inflammatory cells found in smears are often too sparse to be diagnostic, partly because of the risk of the biopsy causing exacerbation of pre-existing pancreatitis.18,19 However, it may be utilized if fibrosis with resultant nodularity leads to a radiological presentation which is suspicious of a neoplastic mass, and, if on surgical exploration, the gland is felt to be increased in size and/or consistency, whether focally or diffusely. Since duct obstruction, edema and inflammation peripheral to a carcinoma can simulate true pancreatitis, multiple biopsies should be taken both proximal to the mass and from different parts of the abnormal area to exclude malignancy. This can be done endoscopically or intraoperatively without significantly increasing the risk of local complications.
In severe chronic pancreatitis much of the exocrine parenchyma may be destroyed and be replaced by fibrous tissue. In such cases, remaining ductal, acinar and endocrine epithelium can show prominent reactive/regenerative atypia which can be difficult to distinguish from well-differentiated adenocarcinoma (Fig. 11.4). There may be crowding of nuclei and microglandular arrangement of cells with nuclear enlargement and variation in nuclear size and shape. The atypia is usually variable in degree between cells.
Infectious pancreatitis is uncommon in immunocompetent hosts, and occurs more frequently in circumstances of immune suppression. It may be caused by a variety of pathogens, including viruses, parasites, bacteria and fungi.55 Organisms gain access to the pancreas either by the hematogenous route in systemic infections, or from the intestinal tract through the pancreaticobiliary system.
Cysts
Cysts in the pancreas can be congenital or post-pancreatitic pseudocysts. Some tumors are inherently and primarily cystic; in addition, any neoplasm, benign or malignant, may undergo cystic degeneration. Evaluation of cysts by radiologic or endoscopic features alone can sometimes be problematic.56 Cytologic evaluation of aspirated cyst fluid is widely used and often fully diagnostic of the nature of the cyst, although sampling and interpretative errors are acknowledged.57,58 Sensitivity of cytodiagnosis of pancreatic cystic lesions was considerably lower than that of solid lesions (64% vs 98%) in one study.57 Biochemical analysis of fluid from cystic lesions improves the sensitivity and specificity over cytologic assessment alone (see Intraductal papillary mucinous and mucinous cystic neoplasms below).
Pseudocyst (Fig. 11.5)
These non-neoplastic cysts are the most commonly encountered pancreatic cysts. Being fluid collections occurring in post-pancreatitic states, they are not true cysts and as such are lined by granulation tissue, not by epithelium. Aspiration yields copious turbid watery fluid which contains debris, occasional inflammatory cells including histiocytes, possible fibroblasts and bile, and perhaps rare fragments of epithelial tissue from the surrounding gland. Raised enzyme levels, e.g. amylase and lipase, and low or absent tumor marker levels are found in the aspirated cyst fluid. The fluid is nonmucinous, but may demonstrate background fibrinous strands. Should the aspirate appear purulent, it should be submitted for culture to confirm secondary infection.
Serous cystadenoma (microcystic/glycogen-rich adenoma) (Fig. 11.6)
These are uncommon benign lesions, often incidental findings in older individuals. Their presentation must be familiar to cytopathologists, as failure to identify the constituent bland monolayered sheets might result in an erroneous ‘nonrepresentative’ report. Histopathologically, these tumors demonstrate classic microcysts, lined by bland, uniform glycogen-rich mucin-negative cells (Fig. 11.6).59 Several reports have appeared in the literature on FNB cytodiagnosis.58,60,61 The aspirate is clear and watery, some are acellular, while others contain a sparse exfoliate of monomorphic round cells with finely vacuolated, glycogen-positive, mucin-negative cytoplasm, and centrally disposed nuclei containing fine chromatin. Occasionally, larger monolayered sheets may be obtained (Fig. 11.6). The cell groups may resemble mesothelial cells or bland acinar cells. Cell features are thus non-specific, but are assessed in conjunction with the watery nature of the aspirate and the very characteristic radiological picture of small microcysts within a well-demarcated round mass lesion arising anywhere in the pancreas. A PAS stain will highlight the glycogen-rich cytoplasm.
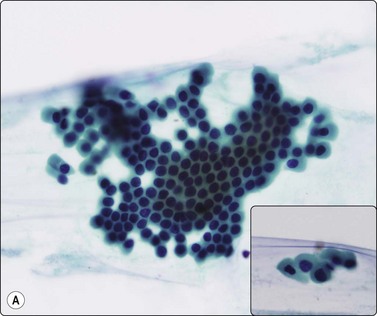
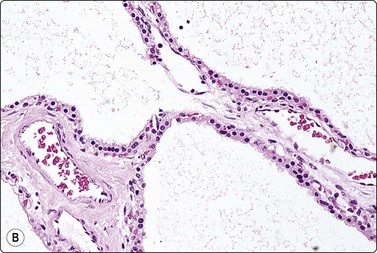
(A) Watery aspirate with a few monolayered sheets of epithelial cells with small round uniform nuclei and moderate pale cytoplasm; see inset (Pap, HP); (B) Corresponding tissue section (H&E, IP).
Biochemical analysis of cyst fluid generally shows a low viscosity and low CEA and amylase levels.62 A recent study which combined imaging, biochemical and cytological data in reaching a diagnosis concluded that the preoperative diagnosis of serous cystadenomas remains a challenge.63
Problems in diagnosis
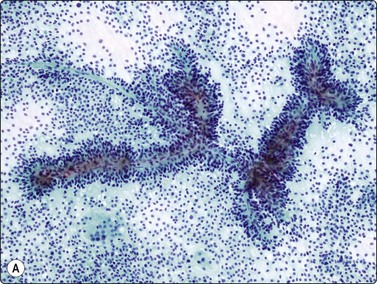
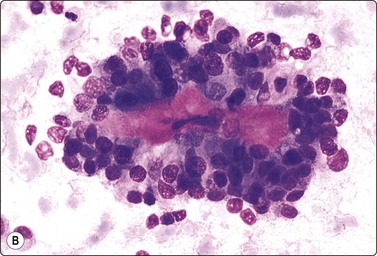
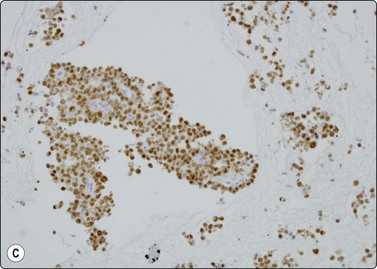
Fig. 11.7 Solid-pseudopapillary neoplasm
(A) Vessel associated pseudopapillary fragment (Pap, LP); (B) Pseudopapillary fragment. Note small tumor cells and irregular outer border with detached tumor cells (MGG, IP); (C) Cell block section showing positive staining for progesterone receptors (PR, IP).
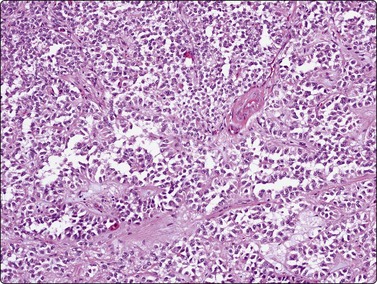
Fig. 11.8 Solid and cystic papillary neoplasm
Corresponding tissue section from Figure 11.7 (H&E, IP).
Solid-psedopapillary neoplasm (Figs 11.7 and 11.8)
Criteria for diagnosis
This relatively uncommon low malignant potential pancreatic neoplasm, with its many pseudonyms, occurs almost exclusively in young women under the age of 20 years.64 They usually behave in a benign manner but 10–15% of cases show spread or metastases. Still considered to be of uncertain histogenesis, this tumor is thought to be hormonally dependent. Occurring anywhere along the length of the pancreas, the mass is usually sizeable at presentation, averaging 8 cm. Grossly, as well as on imaging studies, it is characterized by good circumscription, multiloculation and solid and cystic areas. The histopathologic features, which are specific and diagnostic, are mirrored exactly by the cytologic pattern in FNB smears. Initial tumor growth is solid, but degeneration results in clefts, eventually enlarging to cysts, between vascularized cell fragments, which then appear ‘pseudopapillary’.
Fifty isolated case reports or limited series attesting to the ability of FNB to confidently identify this unusual but widely reported neoplasm were summarized in 2002 by Pettinato and colleagues in a definitive paper.64 Richly cellular smears demonstrate characteristic pseudopapillae and pseudorosettes, composed of small tumor cells adherent to delicate metachromatic fibrovascular stalks. Recognition of this stromal component is essential in distinguishing this tumor of relatively good prognosis from similar small-celled pancreatic neoplasms. The outline of both the pseudopapillae and the pseudorosettes appears irregular, due to exuberant exfoliation of single cells into the smear background. These vascular-based aggregates vary from large grape-like branching structures to single capillaries with leaf-like single-cell linings (Fig. 11.7). These cells are small, round to oval, plasmacytoid or cuboidal and extremely monotonous. Cytoplasm is variably preserved, vesicular to faintly granular, containing characteristic eosinophilic, hyaline intracytoplasmic globules, which are PAS positive. Nuclei are round and even, with grooves but no major irregularities or significant pleomorphism. Chromatin is described as finely granular, without clumping or clearing. Small nucleoli may be appreciated and are occasionally multiple. In the background, foam cells, multinucleated giant cells, debris and laminated psammoma bodies reflect the cystic and papillary nature of the parent tumor. Mucus is absent. Diagnostic accuracy of EUS-FNA has been reported as 75% in a series of 28 cases.65
Immunocytochemistry is extremely variable but most neoplasms studied stain positively for vimentin, α1-antitrypsin, α1-antichymotrypsin and progesterone receptor (Fig. 11.7C). Unlike PET, they are typically negative for chromogranin, cytokeratin, EMA and specific pancreatic hormones. Both tumors may express CD10, synaptophysin and CD56.64,66-69 More recently, nuclear expression of beta-catenin together with loss of normal membrane localization of E-cadherin have been proposed as useful in separating the two entities.66,67,69
Morphologic recognition is critical to extract these slow-growing, operable and potentially curable tumors of young women from the very much larger pool of usual pancreatic cancers with their abysmal prognosis.
Adenocarcinoma (Figs 11.9-11.11)
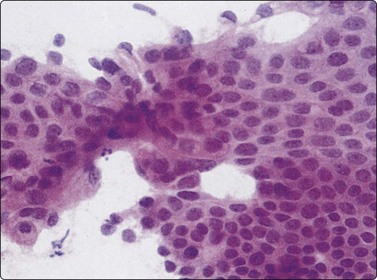
Fig. 11.9 Adenocarcinoma (pancreas)
Very well-differentiated adenocarcinoma presenting as cohesive, monolayered epithelial sheets; note nuclear crowding, overlapping and nuclear atypia present focally (H&E, HP).
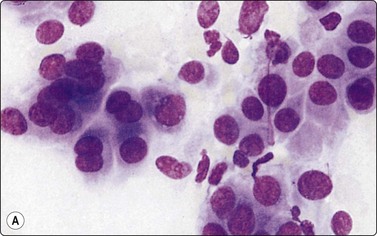
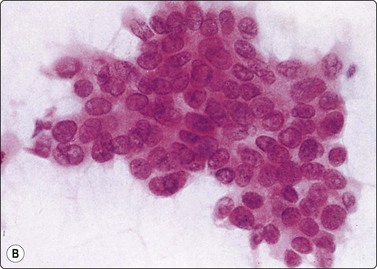
Fig. 11.10 Adenocarcinoma (pancreas)
Well-differentiated adenocarcinoma; relatively mild nuclear atypia, but nuclear crowding and some dissociation, and in B a tendency to microacinar arrangement (A, MGG, HP; B, H&E, HP).
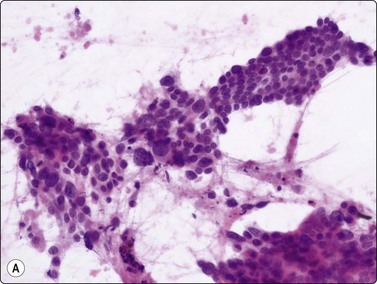
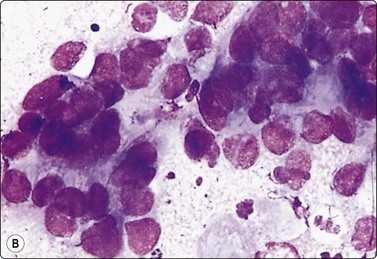
Fig. 11.11 Adenocarcinoma (pancreas) moderately differentiated
(A) Sheets showing disorganization and nuclear pleomorphism; background of necrotic debris (H&E, IP); (B) Disorganized clusters with some acinar arrangements (MGG, HP).
Almost all adenocarcinomas arising in the pancreas are of ductal origin, without unique features permitting absolute distinction from carcinomas arising in the biliary tree. The characteristic FNB pattern is of crowded ‘drunken’ monolayered sheets, with moderately tall columnar palisading cells at luminal edges. Smaller aggregates commonly show rounded glandular structures with feathered edges, or three-dimensionality. Cytoplasmic borders may be very well demarcated in better-differentiated forms; in other cases, the monolayer sheets appear syncytial. The diagnosis of malignancy ultimately depends on nuclear features, which range from very subtle alterations to overly malignant criteria with decreasing differentiation (Figs 11.9-11.11). In an assessment of individual criteria by regression analysis, Cohen and colleagues extracted as most important: anisocytosis (4 : 1), nuclear enlargement and molding, with combined sensitivity of 98% using all three criteria to distinguish malignant from benign.70 In a similar study, Robins, Katz and Evans ascribed major status to nuclear crowding, contour and chromatin irregularity, minor status to single cells, mitoses and enlarged nuclei,33 requiring two major, or one major and two minor criteria for the definitive diagnosis of malignancy. Focusing specifically on the most challenging area, the recognition of well-differentiated carcinoma, Lin and Staerkel listed the following features: nuclear enlargement > 2 rbc (99%), anisonucleosis 4 : 1 (97%), nuclear membrane irregularity (97%), and crowding/overlapping/three-dimensionality (92%).73
The use of ancillary studies has been advocated to improve sensitivity. The mucin profile of MUC1+/MUC2−/MUC5AC+ has been reported as discriminating between a benign and malignant glandular epithelial cells.74,75 Immunohistochemistry for mesothelin and prostate stem cell antigen (PSCA) was also found to be useful,76 as was a combination of P53, DPC4 (MAD4) and Kras mutation.77 Some authors have also reported molecular analysis for K-ras mutation improves the sensitivity of FNB cytology.78,79
Carcinoma variants comprise as many as 10–15% of ductal carcinomas. Of these, adenosquamous carcinoma is the most common, accounting for up to 5% of pancreatic neoplasms. The percentage of cells showing glandular and squamous differentiation is highly variable, particularly in FNB specimens. The glandular component is usually moderately or poorly differentiated; the squamous component may be keratinizing or not. The prognosis of this variant is as poor as that of usual ductal cancer.80-82
Mucinous tumors of the pancreas
These tumors are best categorized into four subgroups,83 each of which has implications for cytodiagnosis:
Of these, the last two mentioned require little further expansion, both being rare variants of ductal adenocarcinoma. Mucinous adenocarcinoma/colloid carcinoma is a solid ductal carcinoma in which greater than 50% of the tumor is of mucinous type, revealing tall columnar morphology and demonstrable intracytoplasmic and extracellular mucus. The radiologic appearance of a solid neoplasm separates it from both IPMN and MCN. Mucinous or colloid carcinomas occur mainly in the pancreatic head and have identical gender, age and prognostic implications as the usual adenocarcinoma. Signet ring carcinoma is exceptionally rare, and often difficult to distinguish histologically from chronic pancreatitis because of its diffuse infiltrative characteristics. It is cytologically recognizable by the presence of classic single-lying signet ring cells, bloated with mucin, seen in pools of mucin. This subtype, too, carries no prognostic advantage.
Intraductal papillary mucinous and mucinous cystic neoplasms (Figs 11.12-11.14)
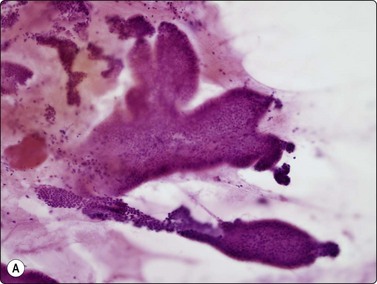
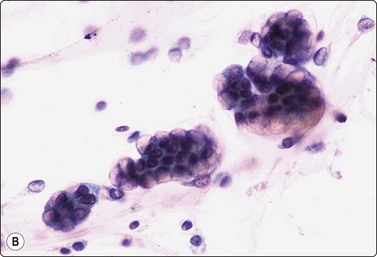
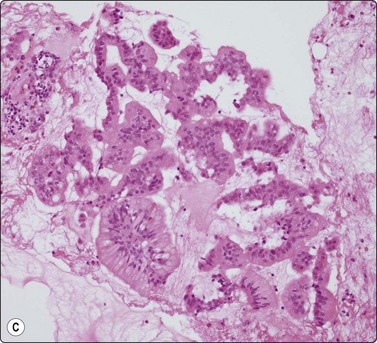
Fig. 11.12 Intraductal papillary-mucinous neoplasm (IPMN)
(A) Large multilayered papillary structures; note the smooth borders (H&E, IP); (B) Small papillary clusters of cuboidal epithelial cells with mucin vacuoles; the pale mucin in the background is not easily seen (Pap, HP); (C) Cell block showing strips and papillary formations of atypical columnar epithelium (H&E, IP).
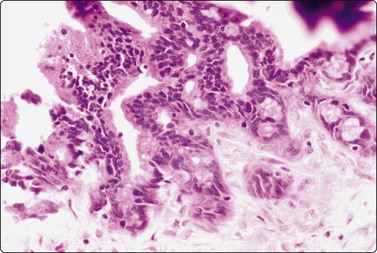
Fig. 11.13 Intraductal papillary-mucinous neoplasm (IPMN)
Tissue section corresponding to Figure 11.12 (H&E, IP).
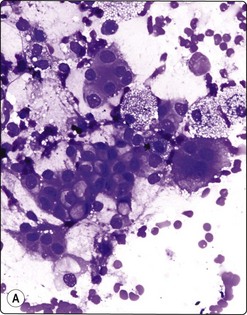
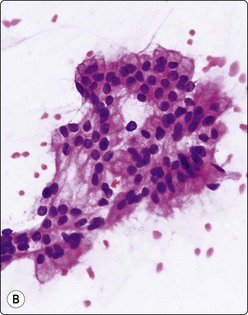
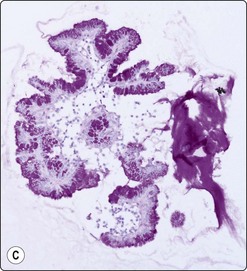
Fig. 11.14 Mucinous cystic neoplasm (MCN)
(A, B) Clusters and sheets of relatively monotonous malignant glandular cells showing intracytoplasmic mucin vacuoles (A, MGG, HP; B, H&E, HP); (C) PAS diastase stain on cell block material highlights the intra- and extracellular mucin (PAS/D, IP).
Distinction between IPMN and MCN on cytologic grounds is problematic and requires correlation with clinical, imaging and endoscopic findings as the clinico-radiologic characteristics are very different.
Intraductal papillary-mucinous neoplasms (IPMN) were first described as a distinct entity in the 1980s.83 They are now well characterized as tumors of better prognosis as they have a long intraductal course but eventually will invade into periductal pancreatic parenchyma, and then go on to nodal and distant metastases.84 Both sexes can be affected, but elderly males predominate. IPMN usually involves the main duct in the head of the pancreas but can arise in side branches. As the name implies, the lining epithelium is both papillary and mucinous. The duct system is filled with viscous mucus, leading to a very characteristic endoscopic appearance, where the ampulla of Vater is often seen to be patulous, and draining mucin. The cytology is distinctive (but not entirely specific), showing abundant background mucin, in which sheets and papillary aggregates of cuboidal to columnar, mucin-filled cells are seen (Figs 11.12 and 11.13). Several reports attest to the ability of cytology to diagnose these good-prognosis tumors preoperatively, in conjunction with radiologic and endoscopic features.84-86 In a series of 19 patients, a sensitivity of 82% and specificity of 100% was reported.87
The classic mucinous cystic neoplasm (MCN) is an indolent tumor of middle-aged females, the majority occurring in the tail of the pancreas. These tumors show a spectrum of grade and behavior including benign cystadenomas, borderline forms, and malignant cystadenocarcinomas. The latter obviously has a poorer prognosis if invasion has occurred. Radiologic studies identify a usually discrete and obviously cystic mass in the pancreatic tail. As there is no communication with the pancreatic ductal system, endoscopy does not demonstrate mucinous discharge from a patulous ampulla. As with IPMN, cytology shows a background of copious extracellular mucin, in which cohesive monolayered sheets of mucinous columnar cells are found (Fig. 11.14). These demonstrate regular to irregular honeycombing, with a spectrum of nuclear changes depending on the stage of tumor progression. Epithelial cell cytoplasm is distended with mucin vacuoles.88-91
Biochemical analysis of fluid from cystic lesions improves the sensitivity and specificity over cytologic assessment alone. Various parameters have been measured including the presence of mucin, viscosity, CEA, CA 19-9, amylase, lipase.21,44 Amylase levels are a normal.16 CEA appears to be the most helpful for diagnosing IPMN and MCN15,42,43,92 and some authors state that cytologic analysis adds little to biochemical analysis of the cyst fluid.92,93 A very high levels of CEA (> 6000 ng/mL) can predict malignancy.42,94
Analysis of the mucin expression profile (MUC1, MUC2 and MUC5AC) has been reported as useful in discriminating between mucinous versus non-mucinous lesions as well as between benign versus malignant mucinous neoplasms.31,75,86 More recently, there have been studies suggesting various molecular analyses on cyst fluid such as k-ras mutation and loss of heterozygosity, may be similarly useful.95-98
Other variants of pancreatic cancer
Pure osteoclastic giant cell tumors of the pancreas also require cytologic recognition as their behavior pattern may be more indolent than that of usual ductal carcinoma.99 Osteoclastic cells, with clustered bland nuclei disposed in dense cytoplasm, are interspersed with mononuclear cells displaying identical, uniform nuclei (Fig. 11.15).99,100 It is mandatory to distinguish this from another carcinoma variant, pleomorphic giant cell tumor (sometimes called anaplastic carcinoma) of the pancreas, which, as its name implies, also contains giant cells but with anisokaryotic daughter nuclei and macronucleoli (Fig. 11.16). Phagocytosis of inflammatory cells by tumor cells is common, and residual recognizable adenocarcinoma may be seen. This is the most lethal of all the pancreatic malignancies.101,102 Interestingly, mixed forms, containing both osteoclastic and pleomorphic giant cells, occur.103,104 Most reports indicate the prognosis to be extremely poor, approximating that of the pleomorphic variant.
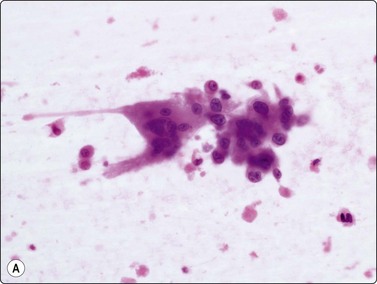
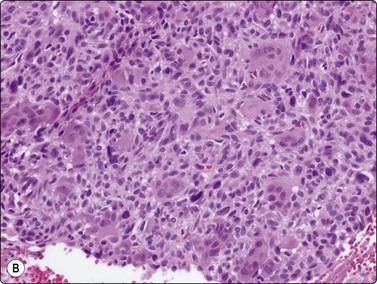
Fig. 11.15 Osteoclastic adenocarcinoma (pancreas)
(A) Two osteoclastic type giant cells mixed with a poorly cohesive cluster of large, highly atypical epithelial cells (H&E, HP); (B) Cell block section corresponding to A (H&E, IP).
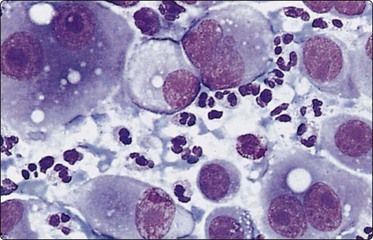
Fig. 11.16 Anaplastic pancreatic carcinoma
Highly pleomorphic mainly dispersed mononuclear and multinucleated tumor cells with macronucleoli (MGG, HP).
Deposits of metastatic cancer within the pancreas or adjacent lymph nodes occur from a wide variety of primary sites, predominantly lung, kidney and breast. The possibility of a non-pancreatic primary should be considered whenever the cytological pattern deviates from that of typical adenocarcinoma. A pancreatic adenocarcinoma of ductal type is cytologically indistinguishable from extrahepatic bile duct carcinoma. Diagnosis is facilitated when the patient has had a history of a previous primary tumor; conversely, the pancreas may be the presenting site of a tumor originating elsewhere.105,106 Comparison of current cytomorphology with previous aspirates or tissue sections will solve many cases of suspected metastasis. Alternately, immunocytochemistry may be utilized to distinguish metastases from various sites. Differential cytokeratin 7 and 20 staining, together with TTF-1 (for lung), ER and PR (for breast), CD10 (for kidney) and melanoma markers, form a useful panel.
Pancreatic lymphoma is rare, but nodal lymphomas in the peripancreatic region are not. These may cause space-occupying lesions and biliary obstruction, mimicking pancreatic neoplasia. Rapid on-site evaluation will detect a lymphoid lesion, and will prompt collection of a separate needle pass into an appropriate medium (RPMI) for flow cytometry, or cell block material for lymphoma immunophenotyping. The majority of pancreatic lymphomas are reported to be diffuse large-cell non-Hodgkin’s lymphomas; a few cases have been small non-cleaved cell lymphomas of B lineage.105
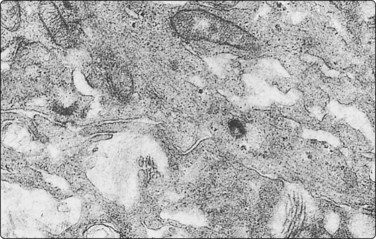
Rarest of all exocrine tumors of the pancreas is the highly aggressive acinar cell adenocarcinoma.107-112 The few cytologic case reports describe and illustrate highly cellular smears, with the cells in loosely cohesive aggregates, sometimes showing acinar formations. The cells having poorly defined granular cytoplasm, large smooth central nuclei, irregularly clumped chromatin, prominent chromatin clearing and large cherry-red nucleoli (Fig. 11.17).107,110,111 Distinction from islet cell tumor, which can be a problem, is generally made using immunocytochemical staining for exocrine enzymes and neuroendocrine markers, respectively.113 Zymogen granules are demonstrable, as are microvilli on ultrastructural examination.107,110,112
Islet cell tumours (Figs 11.18-11.20)
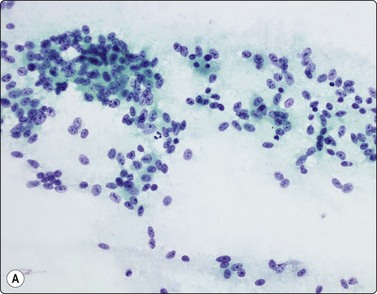
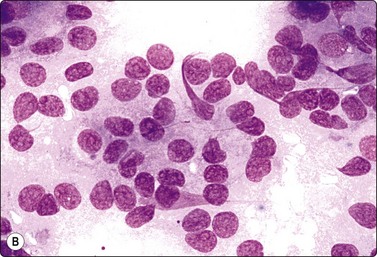
(A) Poorly cohesive sheets and dispersal; cells with oval nuclei, stippled chromatin and small nucleoli (Pap, IP); (B) Mainly dispersed cells with uniformly round nuclei, speckled chromatin, moderate anisokaryosis; delicate cytoplasm; a suggestion of pseudorosettes (MGG, HP).
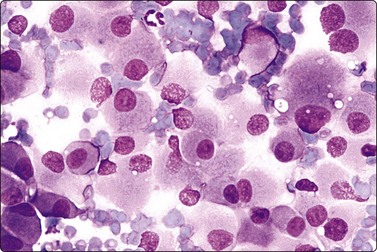
Fig. 11.20 Malignant islet cell tumor
Poorly cohesive cells of endocrine appearance; no obvious cytologic features to suggest malignancy. This tumor metastasized to the liver (MGG, HP).
These functional or nonfunctioning tumors of adults are situated mainly in the body and tail of the pancreas. The tumors are well within the scope of EUS cytodiagnosis.114,115 The neoplastic cells are mainly dissociated, but often form loose acinar or follicular clusters and curved or circular rows (Fig. 11.18). In the majority of cases, nuclei are characteristically round to oval and uniformly small. Occasionally, nuclear anisokaryosis may be prominent, but the nuclear chromatin pattern varies little between cells. The chromatin is evenly distributed, coarsely granular or ‘speckled’. The small nucleoli are not easily seen in Giemsa-stained smears. Due to its fragility, the cytoplasm is often dispersed in the background. When it is preserved, nuclei are eccentrically situated within defined cell borders. A very fine, red granularity is often discernible in MGG smears with high magnification. Clumps of amyloid may occasionally be seen, but necrosis is not observed.113,116-119 The cytological pattern of most islet cell tumors is sufficiently characteristic to be easily distinguished from that of pancreatic adenocarcinoma. Production and secretion of specific hormone products cannot be distinguished on the basis of routine cytological smears alone. If the secretory products of the neoplastic cells can be identified by immunocytochemical methods, a more specific diagnosis can be made with confidence. General neuroendocrine markers will usually be positive, such as neuron-specific enolase, synaptophysin, chromogranin-A, PGP9.5 and CD56. Specific secretory products, e.g. insulin can be marked by appropriate antibodies but this does not necessarily correlate with raised serum levels. Electron microscopy will demonstrate dense-core neurosecretory granules. A decision on whether a tumor is benign or malignant is problematic. Even histopathology is poorly predictive of aggressive behavior. In general, nuclear atypia and pleomorphism cannot be relied on as cytological criteria of malignancy (Fig. 11.20). More aggressive behavior correlates with a raised proliferation index assessed using Ki-67 immunohistochemistry (> 2%).120 This is a parameter in the WHO 2004 histology based-grading system.121,122 Only documentation of metastasis finally provides proof of malignancy.123,124
Pancreatoblastoma
These unusual, large pancreatic primary tumors are encountered in children and adolescents. They demonstrate solid and acinar architecture with interspersed squamous corpuscles and osseous and/or chondroid metaplasia. Cytologic reports are scant, but mention cellular smears, detached small cells, primitive ‘blastemal’ epithelial cells with even chromatin and delicate cytoplasm, rare squamous corpuscles and mesenchymal fragments. The most obvious differential diagnosis, particularly if mesenchyma is present and squamous differentiation absent, is the solid-cystic papillary tumor encountered in young women (see also Chapter 17).125-127
Intra-abdominal tumors
Endoscopic ultrasonograph guided FNB is increasingly being used as a modality of choice in sampling of lymph nodes, extraluminal masses in the region of the bowel and gastrointestinal wall lesions.128 Mucosal lesions encountered at endoscopy in the upper gastrointestinal tract may be sampled by endoscopically obtained brushings. Further, depending on adjunctive clinical and radiologic studies, brush devices may be passed through the ampulla of Vater into the biliary tree at endoscopy, offering an innovative and low intervention mechanism of collection of cytologic material for diagnosis of lesions involving the extrahepatic and pancreatic ductal systems.1,129-131 However, exfoliative techniques, results and morphologic features are not in the scope of this book. Should endoscopic techniques not be available, or not be successful, access to intra-abdominal mass lesions may be gained by transabdominal FNB, with US or CT guidance.
Adenocarcinoma of the gastrointestinal tract may be well or poorly differentiated and glandular, signet ring, or mucinous in type. The origin of the tumor is usually revealed by clinical and radiological findings but may be suggested by cytological features. For example, a poorly differentiated signet ring adenocarcinoma with intracytoplasmic mucin vacuoles is most likely of gastric origin; a well-differentiated adenocarcinoma of columnar cells showing a palisaded arrangement and with tumor necrosis is probably of colonic origin. An example of anal carcinoma of cloacogenic type is illustrated in Figure 11.21. However, distinction between adenocarcinoma of the pancreas, the biliary tract or the female genital tract may not be possible on the basis of routine smears. Differential cytokeratin immunostaining may be helpful.
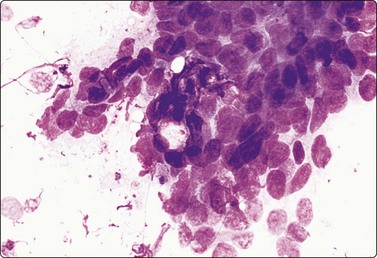
Fig. 11.21 Cloacogenic carcinoma
FNB of anal tumor; multilayered tumor fragment of malignant basaloid cells with high N : C ratio, no microarchitectural pattern (MGG, HP).
The cytological characteristics of carcinoid tumors are described in Chapter 8. The pattern is similar to that of pancreatic islet cell tumors and immunostaining for neuroendocrine markers, particularly synaptophysin and chromogranin, is positive.118
Gastrointestinal stromal tumors (GISTs), formerly classified as smooth muscle tumors, have now been extracted as a distinct entity. They are mesenchymal tumors, arising in the wall of the gastrointestinal tract, showing a spectrum of benign, borderline and malignant behavior. They are typically spindled but there is also an epithelioid variant showing the same antigen profile. Cytodiagnosis of the spindle-celled variant is relatively simple, showing fascicles of spindled cells with elongated, cigar- or comma-shaped nuclei, often set in a wispy filamentous background (Fig. 11.22A and B). The epithelioid variant presents greater difficulty, resembling epithelial tumors. The cells are round, cytoplasm distinct with a perinuclear halo, and nuclei vesicular (Fig. 11.22C). The tumors express CD117 (C-kit) and CD34 (Figs 11.23 and 11.24), but are negative for desmin, S-100 and keratin. Cytopathologists must be ‘GIST-conscious’ owing to the responsiveness of these tumors to Gleevec, which can induce tumor regression and long-term response, even in metastases.132-136
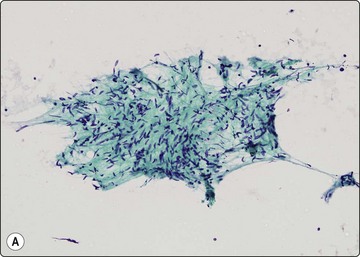
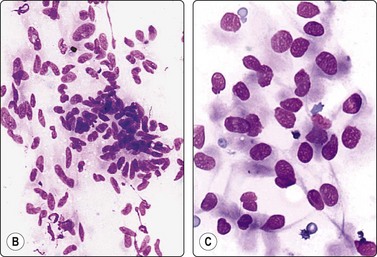
Fig. 11.22 Gastrointestinal stromal tumor (GIST)
(A, B) Spindle cell variant; typical pattern (A, Pap, LP; B, MGG, IP); (C) Epithelioid variant (MGG, HP).
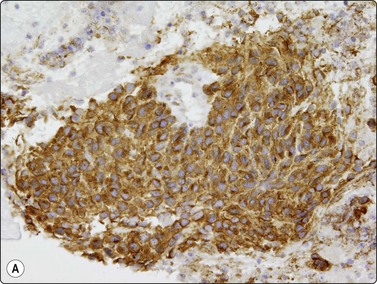
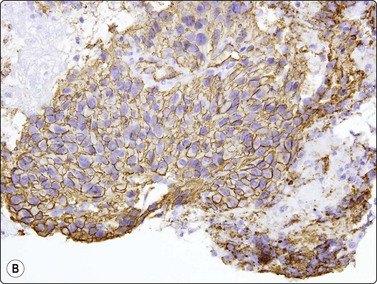
Fig. 11.23 Gastrointestinal stromal tumor (GIST)
(A, B) Cell block sections stained for C-kit (CD117) and CD34 (A, C-kit, HP; B, CD34, HP).
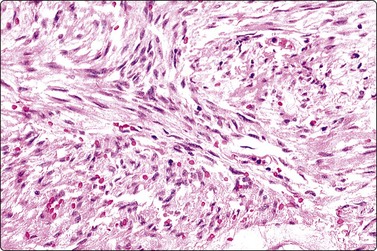
Fig. 11.24 Gastrointestinal stromal tumor (GIST)
Tissue section corresponding to Figure 11.22B (H&E, IP).
An example of a very uncommon tumor arising from small bowel wall, a gastrointestinal autonomic nerve tumor (GANT), is shown in Figures 11.25 and 11.26. This is a spindle cell tumor of neural origin. It is a biologically aggressive neoplasm with metastatic potential. Neurosecretory granules are frequent, and staining with antibodies to synaptophysin, S-100, NSE and vimentin is observed, staining for desmin is negative. Intra-abdominal desmoplastic small cell round tumor, a relatively new entity, is a rare, highly aggressive malignant neoplasm of the serosa of the abdominal cavity, seen mainly in young male patients. The findings in FNB smears have been reported.137-140 A description is given in Chapter 17, and a case is illustrated in Figure 11.27.
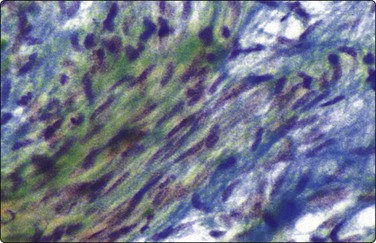
Fig. 11.25 Gastrointestinal autonomic nerve tumor
Cohesive tissue fragment of bland-looking spindle cells resembling schwannoma. Result of immunohistochemistry; see text (Pap, HP).
1 Stewart CJ, Mills PR, Carter R, et al. Brush cytology in the assessment of pancreatico-biliary strictures: a review of 406 cases. J Clin Pathol. 2001 Jun;54(6):449-455.
2 Hancke S, Holm HH, Koch F. Ultrasonically guided percutaneous fine needle biopsy of the pancreas. Surg Gynecol Obstet. 1975 Mar;140(3):361-364.
3 Oscarson J, Stormby N, Sundgren R. Selective angiography in fine-needle aspiration cytodiagnosis of gastric and pancreatic tumours. Acta Radiol Diagn (Stockh). 1972 Nov;12(6):737-750.
4 Bellizzi AM, Stelow EB. Pancreatic cytopathology: a practical approach and review. Arch Pathol Lab Med. 2009 Mar;133(3):388-404.
5 Jhala NC, Jhala DN, Chhieng DC, et al. Endoscopic ultrasound-guided fine-needle aspiration. A cytopathologist’s perspective. Am J Clin Pathol. 2003 Sep;120(3):351-367.
6 Stelow EB, Bardales RH, Stanley MW. Pitfalls in endoscopic ultrasound-guided fine-needle aspiration and how to avoid them. Adv Anat Pathol. 2005 Mar;12(2):62-73.
7 Brandwein SL, Farrell JJ, Centeno BA, et al. Detection and tumor staging of malignancy in cystic, intraductal, and solid tumors of the pancreas by EUS. Gastrointest Endosc. 2001 Jun;53(7):722-727.
8 Faigel DO, Ginsberg GG, Bentz JS, et al. Endoscopic ultrasound-guided real-time fine-needle aspiration biopsy of the pancreas in cancer patients with pancreatic lesions. J Clin Oncol. 1997 Apr;15(4):1439-1443.
9 Fritscher-Ravens A, Izbicki JR, Sriram PV, et al. Endosonography-guided, fine-needle aspiration cytology extending the indication for organ-preserving pancreatic surgery. Am J Gastroenterol. 2000 Sep;95(9):2255-2260.
10 O’Toole D, Palazzo L, Arotcarena R, et al. Assessment of complications of EUS-guided fine-needle aspiration. Gastrointest Endosc. 2001 Apr;53(4):470-474.
11 Raut CP, Grau AM, Staerkel GA, et al. Diagnostic accuracy of endoscopic ultrasound-guided fine-needle aspiration in patients with presumed pancreatic cancer. J Gastrointest Surg. 2003 Jan;7(1):118-126. discussion 27–8
12 Shin HJ, Lahoti S, Sneige N. Endoscopic ultrasound-guided fine-needle aspiration in 179 cases: the M. D. Anderson Cancer Center experience. Cancer. 2002 Jun 25;96(3):174-180.
13 Ylagan LR, Edmundowicz S, Kasal K, et al. Endoscopic ultrasound guided fine-needle aspiration cytology of pancreatic carcinoma: a 3-year experience and review of the literature. Cancer. 2002 Dec 25;96(6):362-369.
14 Zamboni GA, D’Onofrio M, Idili A, et al. Ultrasound-guided percutaneous fine-needle aspiration of 545 focal pancreatic lesions. AJR Am J Roentgenol. 2009 Dec;193(6):1691-1695.
15 Pinto MM, Meriano FV. Diagnosis of cystic pancreatic lesions by cytologic examination and carcinoembryonic antigen and amylase assays of cyst contents. Acta Cytol. 1991 Jul–Aug;35(4):456-463.
16 Silverman JF, Geisinger KR. Ancillary studies in FNA of liver and pancreas. Diagn Cytopathol. 1995 Dec;13(5):396-410.
17 Gress FG, Hawes RH, Savides TJ, et al. Role of EUS in the preoperative staging of pancreatic cancer: a large single-center experience. Gastrointest Endosc. 1999 Dec;50(6):786-791.
18 Hollerbach S, Klamann A, Topalidis T, et al. Endoscopic ultrasonography (EUS) and fine-needle aspiration (FNA) cytology for diagnosis of chronic pancreatitis. Endoscopy. 2001 Oct;33(10):824-831.
19 Jorda M, Essenfeld H, Garcia E, et al. The value of fine-needle aspiration cytology in the diagnosis of inflammatory pancreatic masses. Diagn Cytopathol. 1992;8(1):65-67.
20 Centeno BA. Fine needle aspiration biopsy of the pancreas. Clin Lab Med. 1998 Sep;18(3):401-427. v–vi
21 Hammel P, Levy P, Voitot H, et al. Preoperative cyst fluid analysis is useful for the differential diagnosis of cystic lesions of the pancreas. Gastroenterology. 1995 Apr;108(4):1230-1235.
22 Blandamura S, Costantin G, Nitti D, et al. Intraoperative cytology of pancreatic masses. A 10-year experience. Acta Cytol. 1995 Jan–Feb;39(1):23-27.
23 Hyoty MK, Mattila JJ, Salo K, et al. Intraoperative fine needle aspiration cytologic examination of pancreatic lesions. Surg Gynecol Obstet. 1991 Sep;173(3):193-197.
24 Saez A, Catala I, Brossa R, et al. Intraoperative fine needle aspiration cytology of pancreatic lesions. A study of 90 cases. Acta Cytol. 1995 May–Jun;39(3):485-488.
24A Forsgren L, Orell SR. Aspiration cytology in carcinoma of the pancreas. Surgery. 1973;73:38-42.
25 Vilmann P, Hancke S, Henriksen FW, et al. Endosonographically-guided fine needle aspiration biopsy of malignant lesions in the upper gastrointestinal tract. Endoscopy. 1993 Oct;25(8):523-527.
26 Wiersema MJ, Wiersema LM, Khusro Q, et al. Combined endosonography and fine-needle aspiration cytology in the evaluation of gastrointestinal lesions. Gastrointest Endosc. 1994 Mar–Apr;40(2 Pt 1):199-206.
27 Jarboe EA, Layfield LJ. Cytologic features of pancreatic intraepithelial neoplasia and pancreatitis: Potential pitfalls in the diagnosis of pancreatic ductal carcinoma. Diagn Cytopathol Aug 20.
28 Brandt KR, Charboneau JW, Stephens DH, et al. CT- and US-guided biopsy of the pancreas. Radiology. 1993 Apr;187(1):99-104.
29 Di Stasi M, Lencioni R, Solmi L, et al. Ultrasound-guided fine needle biopsy of pancreatic masses: results of a multicenter study. Am J Gastroenterol. 1998 Aug;93(8):1329-1333.
30 Gupta RK. Value of image guided fine-needle aspiration cytology in the diagnosis of pancreatic malignancies. Diagn Cytopathol. 1995 Aug;13(2):120-123.
31 Paksoy N, Lilleng R, Hagmar B, et al. Diagnostic accuracy of fine needle aspiration cytology in pancreatic lesions. A review of 77 cases. Acta Cytol. 1993 Nov–Dec;37(6):889-893.
32 Pinto MM, Avila NA, Criscuolo EM. Fine needle aspiration of the pancreas. A five-year experience. Acta Cytol. 1988 Jan–Feb;32(1):39-42.
33 Robins DB, Katz RL, Evans DB, et al. Fine needle aspiration of the pancreas. In quest of accuracy. Acta Cytol. 1995 Jan–Feb;39(1):1-10.
34 Parsons LJr, Palmer CH. How accurate is fine-needle biopsy in malignant neoplasia of the pancreas? Arch Surg. 1989 Jun;124(6):681-683.
35 Afify AM, al-Khafaji BM, Kim B, et al. Endoscopic ultrasound-guided fine needle aspiration of the pancreas. Diagnostic utility and accuracy. Acta Cytol. 2003 May–Jun;47(3):341-348.
36 Eloubeidi MA, Jhala D, Chhieng DC, et al. Yield of endoscopic ultrasound-guided fine-needle aspiration biopsy in patients with suspected pancreatic carcinoma. Cancer. 2003 Oct 25;99(5):285-292.
37 Fisher L, Segarajasingam DS, Stewart C, et al. Endoscopic ultrasound guided fine needle aspiration of solid pancreatic lesions: Performance and outcomes. J Gastroenterol Hepatol. 2009 Jan;24(1):90-96.
38 Mitsuhashi T, Ghafari S, Chang CY, et al. Endoscopic ultrasound-guided fine needle aspiration of the pancreas: cytomorphological evaluation with emphasis on adequacy assessment, diagnostic criteria and contamination from the gastrointestinal tract. Cytopathology. 2006 Feb;17(1):34-41.
39 Tadic M, Kujundzic M, Stoos-Veic T, et al. Role of repeated endoscopic ultrasound-guided fine needle aspiration in small solid pancreatic masses with previous indeterminate and negative cytological findings. Dig Dis. 2008;26(4):377-382.
40 Payne M, Staerkel G, Gong Y. Indeterminate diagnosis in fine-needle aspiration of the pancreas: reasons and clinical implications. Diagn Cytopathol. 2009 Jan;37(1):21-29.
41 Brugge WR. Pancreatic fine needle aspiration: to do or not to do? JOP. 2004 Jul;5(4):282-288.
42 Linder JD, Geenen JE, Catalano MF. Cyst fluid analysis obtained by EUS-guided FNA in the evaluation of discrete cystic neoplasms of the pancreas: a prospective single-center experience. Gastrointest Endosc. 2006 Nov;64(5):697-702.
43 Ryu JK, Woo SM, Hwang JH, et al. Cyst fluid analysis for the differential diagnosis of pancreatic cysts. Diagn Cytopathol. 2004 Aug;31(2):100-105.
44 Shami VM, Sundaram V, Stelow EB, et al. The level of carcinoembryonic antigen and the presence of mucin as predictors of cystic pancreatic mucinous neoplasia. Pancreas. 2007 May;34(4):466-469.
45 Smith EH. Complications of percutaneous abdominal fine-needle biopsy. Review. Radiology. 1991 Jan;178(1):253-258.
46 Mueller PR, Miketic LM, Simeone JF, et al. Severe acute pancreatitis after percutaneous biopsy of the pancreas. AJR Am J Roentgenol. 1988 Sep;151(3):493-494.
47 Carrara S, Arcidiacono PG, Mezzi G, et al. Pancreatic endoscopic ultrasound-guided fine needle aspiration: complication rate and clinical course in a single centre. Dig Liver Dis Jul;42(7):520–523.
48 Rosenbaum DA, Frost DB. Fine-needle aspiration biopsy of the pancreas complicated by pancreatic ascites. Cancer. 1990 Jun 1;65(11):2537-2538.
49 Sims J, Carroll D, Turner JR, et al. Cardiac and metabolic activity in mild hypertensive and normotensive subjects. Psychophysiology. 1988 Mar;25(2):172-178.
50 Warshaw AL. Implications of peritoneal cytology for staging of early pancreatic cancer. Am J Surg. 1991 Jan;161(1):26-29. discussion 9–30
51 Leach SD, Rose JA, Lowy AM, et al. Significance of peritoneal cytology in patients with potentially resectable adenocarcinoma of the pancreatic head. Surgery. 1995 Sep;118(3):472-478.
52 Micames C, Jowell PS, White R, et al. Lower frequency of peritoneal carcinomatosis in patients with pancreatic cancer diagnosed by EUS-guided FNA vs. percutaneous FNA. Gastrointest Endosc. 2003 Nov;58(5):690-695.
53 Meszoely IM, Lee JS, Watson JC, et al. Peritoneal cytology in patients with potentially resectable adenocarcinoma of the pancreas. Am Surg. 2004 Mar;70(3):208-213. discussion 13–4
54 Johnson DE, Pendurthi TK, Balshem AM, et al. Implications of fine-needle aspiration in patients with resectable pancreatic cancer. Am Surg. 1997 Aug;63(8):675-679. discussion 9–80
55 Dassopoulos T, Ehrenpreis ED. Acute pancreatitis in human immunodeficiency virus-infected patients: a review. Am J Med. 1999 Jul;107(1):78-84.
56 Ahmad NA, Kochman ML, Lewis JD, et al. Can EUS alone differentiate between malignant and benign cystic lesions of the pancreas? Am J Gastroenterol. 2001 Dec;96(12):3295-3300.
57 Brugge WR. Role of endoscopic ultrasound in the diagnosis of cystic lesions of the pancreas. Pancreatology. 2001;1(6):637-640.
58 Centeno BA, Lewandrowski KB, Warshaw AL, et al. Cyst fluid cytologic analysis in the differential diagnosis of pancreatic cystic lesions. Am J Clin Pathol. 1994 Apr;101(4):483-487.
59 Alpert LC, Truong LD, Bossart MI, et al. Microcystic adenoma (serous cystadenoma) of the pancreas. A study of 14 cases with immunohistochemical and electron-microscopic correlation. Am J Surg Pathol. 1988 Apr;12(4):251-263.
60 Laucirica R, Schwartz MR, Ramzy I. Fine needle aspiration of pancreatic cystic epithelial neoplasms. Acta Cytol. 1992 Nov–Dec;36(6):881-886.
61 Nguyen GK, Suen KC, Villanueva RR. Needle aspiration cytology of pancreatic cystic lesions. Diagn Cytopathol. 1997 Sep;17(3):177-182.
62 van der Waaij LA, van Dullemen HM, Porte RJ. Cyst fluid analysis in the differential diagnosis of pancreatic cystic lesions: a pooled analysis. Gastrointest Endosc. 2005 Sep;62(3):383-389.
63 Belsley NA, Pitman MB, Lauwers GY, et al. Serous cystadenoma of the pancreas: limitations and pitfalls of endoscopic ultrasound-guided fine-needle aspiration biopsy. Cancer. 2008 Apr 25;114(2):102-110.
64 Pettinato G, Di Vizio D, Manivel JC, et al. Solid-pseudopapillary tumor of the pancreas: a neoplasm with distinct and highly characteristic cytological features. Diagn Cytopathol. 2002 Dec;27(6):325-334.
65 Jani N, Dewitt J, Eloubeidi M, et al. Endoscopic ultrasound-guided fine-needle aspiration for diagnosis of solid pseudopapillary tumors of the pancreas: a multicenter experience. Endoscopy. 2008 Mar;40(3):200-203.
66 Burford H, Baloch Z, Liu X, et al. E-cadherin/beta-catenin and CD10: a limited immunohistochemical panel to distinguish pancreatic endocrine neoplasm from solid pseudopapillary neoplasm of the pancreas on endoscopic ultrasound-guided fine-needle aspirates of the pancreas. Am J Clin Pathol. 2009 Dec;132(6):831-839.
67 Kim MJ, Jang SJ, Yu E. Loss of E-cadherin and cytoplasmic-nuclear expression of beta-catenin are the most useful immunoprofiles in the diagnosis of solid-pseudopapillary neoplasm of the pancreas. Hum Pathol. 2008 Feb;39(2):251-258.
68 Pelosi G, Iannucci A, Zamboni G, et al. Solid and cystic papillary neoplasm of the pancreas: a clinico-cytopathologic and immunocytochemical study of five new cases diagnosed by fine-needle aspiration cytology and a review of the literature. Diagn Cytopathol. 1995 Oct;13(3):233-246.
69 Serra S, Salahshor S, Fagih M, et al. Nuclear expression of E-cadherin in solid pseudopapillary tumors of the pancreas. JOP. 2007;8(3):296-303.
70 Cohen MB, Egerter DP, Holly EA, et al. Pancreatic adenocarcinoma: regression analysis to identify improved cytologic criteria. Diagn Cytopathol. 1991;7(4):341-345.
71 Francillon YJ, Bagby J, Abreo F, et al. Criteria for predicting malignancy in fine needle aspiration biopsies (FNAB) of the pancreas and biliary tree. Acta Cytol. 1996;40:1084.
72 Hejka AG, Baernacki EG. Cytopathology of well-differentiated columnar adenocarcinoma of the pancreas diagnosed by fine needle aspiration. Acta Cytol. 1990;34:716.
73 Lin F, Staerkel G. Cytologic criteria for well differentiated adenocarcinoma of the pancreas in fine-needle aspiration biopsy specimens. Cancer. 2003 Feb 25;99(1):44-50.
74 Giorgadze TA, Peterman H, Baloch ZW, et al. Diagnostic utility of mucin profile in fine-needle aspiration specimens of the pancreas: an immunohistochemical study with surgical pathology correlation. Cancer. 2006 Jun 25;108(3):186-197.
75 Wang Y, Gao J, Li Z, et al. Diagnostic value of mucins (MUC1, MUC2 and MUC5AC) expression profile in endoscopic ultrasound-guided fine-needle aspiration specimens of the pancreas. Int J Cancer. 2007 Dec 15;121(12):2716-2722.
76 McCarthy DM, Maitra A, Argani P, et al. Novel markers of pancreatic adenocarcinoma in fine-needle aspiration: mesothelin and prostate stem cell antigen labeling increases accuracy in cytologically borderline cases. Appl Immunohistochem Mol Morphol. 2003 Sep;11(3):238-243.
77 van Heek T, Rader AE, Offerhaus GJ, et al. K-ras, p53, and DPC4 (MAD4) alterations in fine-needle aspirates of the pancreas: a molecular panel correlates with and supplements cytologic diagnosis. Am J Clin Pathol. 2002 May;117(5):755-765.
78 Bournet B, Souque A, Senesse P, et al. Endoscopic ultrasound-guided fine-needle aspiration biopsy coupled with KRAS mutation assay to distinguish pancreatic cancer from pseudotumoral chronic pancreatitis. Endoscopy. 2009 Jun;41(6):552-557.
79 Maluf-Filho F, Kumar A, Gerhardt R, et al. Kras mutation analysis of fine needle aspirate under EUS guidance facilitates risk stratification of patients with pancreatic mass. J Clin Gastroenterol. 2007 Nov–Dec;41(10):906-910.
80 Leiman G, Markowitz S, Svensson LG. Intraoperative cytodiagnosis of pancreatic adenosquamous carcinoma: a case report. Diagn Cytopathol. 1986 Jan–Mar;2(1):72-75.
81 Rahemtullah A, Misdraji J, Pitman MB. Adenosquamous carcinoma of the pancreas: cytologic features in 14 cases. Cancer. 2003 Dec 25;99(6):372-378.
82 Wilczynski SP, Valente PT, Atkinson BF. Cytodiagnosis of adenosquamous carcinoma of the pancreas. Use of intraoperative fine needle aspiration. Acta Cytol. 1984 Nov–Dec;28(6):733-736.
83 Fukushima N, Mukai K. Pancreatic neoplasms with abundant mucus production: emphasis on intraductal papillary-mucinous tumors and mucinous cystic tumors. Adv Anat Pathol. 1999 Mar;6(2):65-77.
84 Adsay NV, Conlon KC, Zee SY, et al. Intraductal papillary-mucinous neoplasms of the pancreas: an analysis of in situ and invasive carcinomas in 28 patients. Cancer. 2002 Jan 1;94(1):62-77.
85 Emerson RE, Randolph ML, Cramer HM. Endoscopic ultrasound-guided fine-needle aspiration cytology diagnosis of intraductal papillary mucinous neoplasm of the pancreas is highly predictive of pancreatic neoplasia. Diagn Cytopathol. 2006 Jul;34(7):457-462.
86 Stelow EB, Stanley MW, Bardales RH, et al. Intraductal papillary-mucinous neoplasm of the pancreas. The findings and limitations of cytologic samples obtained by endoscopic ultrasound-guided fine-needle aspiration. Am J Clin Pathol. 2003 Sep;120(3):398-404.
87 Fernandez-Esparrach G, Pellise M, Sole M, et al. EUS FNA in intraductal papillary mucinous tumors of the pancreas. Hepatogastroenterology. 2007 Jan–Feb;54(73):260-264.
88 Compagno J, Oertel JE. Mucinous cystic neoplasms of the pancreas with overt and latent malignancy (cystadenocarcinoma and cystadenoma). A clinicopathologic study of 41 cases. Am J Clin Pathol. 1978 Jun;69(6):573-580.
89 Dodd LG, Farrell TA, Layfield LJ. Mucinous cystic tumor of the pancreas: an analysis of FNA characteristics with an emphasis on the spectrum of malignancy associated features. Diagn Cytopathol. 1995 Mar;12(2):113-119.
90 Gupta RK, Scally J, Stewart RJ. Mucinous cystadenocarcinoma of the pancreas: diagnosis by fine-needle aspiration cytology. Diagn Cytopathol. 1989;5(4):408-411.
91 Vellet D, Leiman G, Mair S, et al. Fine needle aspiration cytology of mucinous cystadenocarcinoma of the pancreas. Further observations. Acta Cytol. 1988 Jan–Feb;32(1):43-48.
92 Brugge WR, Lewandrowski K, Lee-Lewandrowski E, et al. Diagnosis of pancreatic cystic neoplasms: a report of the cooperative pancreatic cyst study. Gastroenterology. 2004 May;126(5):1330-1336.
93 Lim SJ, Alasadi R, Wayne JD, et al. Preoperative evaluation of pancreatic cystic lesions: cost-benefit analysis and proposed management algorithm. Surgery. 2005 Oct;138(4):672-679. discussion 9–80
94 Maire F, Voitot H, Aubert A, et al. Intraductal papillary mucinous neoplasms of the pancreas: performance of pancreatic fluid analysis for positive diagnosis and the prediction of malignancy. Am J Gastroenterol. 2008 Nov;103(11):2871-2877.
95 Khalid A, Nodit L, Zahid M, et al. Endoscopic ultrasound fine needle aspirate DNA analysis to differentiate malignant and benign pancreatic masses. Am J Gastroenterol. 2006 Nov;101(11):2493-2500.
96 Khalid A, Zahid M, Finkelstein SD, et al. Pancreatic cyst fluid DNA analysis in evaluating pancreatic cysts: a report of the PANDA study. Gastrointest Endosc. 2009 May;69(6):1095-1102.
97 Sawhney MS, Devarajan S, O’Farrel P, et al. Comparison of carcinoembryonic antigen and molecular analysis in pancreatic cyst fluid. Gastrointest Endosc. 2009 May;69(6):1106-1110.
98 Schoedel KE, Finkelstein SD, Ohori NP. K-Ras and microsatellite marker analysis of fine-needle aspirates from intraductal papillary mucinous neoplasms of the pancreas. Diagn Cytopathol. 2006 Sep;34(9):605-608.
99 Manci EA, Gardner LL, Pollock WJ, et al. Osteoclastic giant cell tumor of the pancreas. Aspiration cytology, light microscopy, and ultrastructure with review of the literature. Diagn Cytopathol. 1985 Apr–Jun;1(2):105-110.
100 Chopra S, Wu ML, Imagawa DK, et al. Endoscopic ultrasound-guided fine-needle aspiration of undifferentiated carcinoma with osteoclast-like giant cells of the pancreas: a report of 2 cases with literature review. Diagn Cytopathol. 2007 Sep;35(9):601-606.
101 Moore JC, Hilden K, Bentz JS, et al. Osteoclastic and pleomorphic giant cell tumors of the pancreas diagnosed via EUS-guided FNA: unique clinical, endoscopic, and pathologic findings in a series of 5 patients. Gastrointest Endosc. 2009 Jan;69(1):162-166.
102 Silverman JF, Finley JL, Berns L, et al. Significance of giant cells in fine-needle aspiration biopsies of benign and malignant lesions of the pancreas. Diagn Cytopathol. 1989;5(4):388-391.
103 Combs SG, Hidvegi DF, Ma Y, et al. Pleomorphic carcinoma of the pancreas with osteoclast-like giant cells expressing an epithelial-associated antigen detected by monoclonal antibody 44–3A6. Diagn Cytopathol. 1988;4(4):316-322.
104 Layfield LJ, Bentz J. Giant-cell containing neoplasms of the pancreas: an aspiration cytology study. Diagn Cytopathol. 2008 Apr;36(4):238-244.
105 Benning TL, Silverman JF, Berns LA, et al. Fine needle aspiration of metastatic and hematologic malignancies clinically mimicking pancreatic carcinoma. Acta Cytol. 1992 Jul–Aug;36(4):471-476.
106 Carson HJ, Green LK, Castelli MJ, et al. Utilization of fine-needle aspiration biopsy in the diagnosis of metastatic tumors to the pancreas. Diagn Cytopathol. 1995 Feb;12(1):8-13.
107 Geisinger KR, Silverman JF. Fine-needle aspiration cytology of uncommon primary pancreatic neoplasms: a personal experience and review of the literature. In: Schmidt WA, Miller TR, Katz RL, editors. Cytopathology Annual. Baltimore: Williams & Wilkins; 1992:23-38.
108 Ishihara A, Sanda T, Takanari H, et al. Elastase-1-secreting acinar cell carcinoma of the pancreas. A cytologic, electron microscopic and histochemical study. Acta Cytol. 1989 Mar–Apr;33(2):157-163.
109 Klimstra DS, Heffess CS, Oertel JE, et al. Acinar cell carcinoma of the pancreas. A clinicopathologic study of 28 cases. Am J Surg Pathol. 1992 Sep;16(9):815-837.
110 Samuel LH, Frierson HFJr. Fine needle aspiration cytology of acinar cell carcinoma of the pancreas: a report of two cases. Acta Cytol. 1996 May–Jun;40(3):585-591.
111 Stelow EB, Bardales RH, Shami VM, et al. Cytology of pancreatic acinar cell carcinoma. Diagn Cytopathol. 2006 May;34(5):367-372.
112 Villanueva RR, Nguyen-Ho P, Nguyen GK. Needle aspiration cytology of acinar-cell carcinoma of the pancreas: report of a case with diagnostic pitfalls and unusual ultrastructural findings. Diagn Cytopathol. 1994;10(4):362-364.
113 Labate AM, Klimstra DL, Zakowski MF. Comparative cytologic features of pancreatic acinar cell carcinoma and islet cell tumor. Diagn Cytopathol. 1997 Feb;16(2):112-116.
114 Gines A, Vazquez-Sequeiros E, Soria MT, et al. Usefulness of EUS-guided fine needle aspiration (EUS-FNA) in the diagnosis of functioning neuroendocrine tumors. Gastrointest Endosc. 2002 Aug;56(2):291-296.
115 Jhala D, Eloubeidi M, Chhieng DC, et al. Fine needle aspiration biopsy of the islet cell tumor of pancreas: a comparison between computerized axial tomography and endoscopic ultrasound-guided fine needle aspiration biopsy. Ann Diagn Pathol. 2002 Apr;6(2):106-112.
116 Chang F, Vu C, Chandra A, et al. Endoscopic ultrasound-guided fine needle aspiration cytology of pancreatic neuroendocrine tumours: cytomorphological and immunocytochemical evaluation. Cytopathology. 2006 Feb;17(1):10-17.
117 Chatzipantelis P, Salla C, Konstantinou P, et al. Endoscopic ultrasound-guided fine-needle aspiration cytology of pancreatic neuroendocrine tumors: a study of 48 cases. Cancer. 2008 Aug 25;114(4):255-262.
118 Collins BT, Cramer HM. Fine-needle aspiration cytology of islet cell tumors. Diagn Cytopathol. 1996 Jul;15(1):37-45.
119 Figueiredo FA, Giovannini M, Monges G, et al. Pancreatic endocrine tumors: a large single-center experience. Pancreas. 2009 Nov;38(8):936-940.
120 Chatzipantelis P, Konstantinou P, Kaklamanos M, et al. The role of cytomorphology and proliferative activity in predicting biologic behavior of pancreatic neuroendocrine tumors: a study by endoscopic ultrasound-guided fine-needle aspiration cytology. Cancer Cytopathol. 2009 Jun 25;117(3):211-216.
121 Capella C, Heitz PU, Hofler H, et al. Revised classification of neuroendocrine tumours of the lung, pancreas and gut. Virchows Arch. 1995;425(6):547-560.
122 Heitz PU, Komminoth P, Perren A, et al. Tumours of the endocrine pancreas. In: DeLellis RA, Lloyd RV, Heitz PU, Eng C, editors. World Health Organization Classification of Tumours: Pathology and Genetics of Tumours of Endocrine Organs. Lyon, France: IARC Press, 2004.
123 al-Kaisi N, Weaver MG, Abdul-Karim FW, et al. Fine needle aspiration cytology of neuroendocrine tumors of the pancreas. A cytologic, immunocytochemical and electron microscopic study. Acta Cytol. 1992 Sep–Oct;36(5):655-660.
124 Shaw JA, Vance RP, Geisinger KR, et al. Islet cell neoplasms. A fine-needle aspiration cytology study with immunocytochemical correlations. Am J Clin Pathol. 1990 Aug;94(2):142-149.
125 Hasegawa Y, Ishida Y, Kato K, et al. Pancreatoblastoma. A case report with special emphasis on squamoid corpuscles with optically clear nuclei rich in biotin. Acta Cytol. 2003 Jul–Aug;47(4):679-684.
126 Henke AC, Kelley CM, Jensen CS, et al. Fine-needle aspiration cytology of pancreatoblastoma. Diagn Cytopathol. 2001 Aug;25(2):118-121.
127 Silverman JF, Holbrook CT, Pories WJ, et al. Fine needle aspiration cytology of pancreatoblastoma with immunocytochemical and ultrastructural studies. Acta Cytol. 1990 Sep–Oct;34(5):632-640.
128 Wiersema MJ, Vilmann P, Giovannini M, et al. Endosonography-guided fine-needle aspiration biopsy: diagnostic accuracy and complication assessment. Gastroenterology. 1997 Apr;112(4):1087-1095.
129 Govil H, Reddy V, Kluskens L, et al. Brush cytology of the biliary tract: retrospective study of 278 cases with histopathologic correlation. Diagn Cytopathol. 2002 May;26(5):273-277.
130 Vadmal MS, Byrne-Semmelmeier S, Smilari TF, et al. Biliary tract brush cytology. Acta Cytol. 2000 Jul–Aug;44(4):533-538.
131 Ylagan LR, Liu LH, Maluf HM. Endoscopic bile duct brushing of malignant pancreatic biliary strictures: retrospective study with comparison of conventional smear and ThinPrep techniques. Diagn Cytopathol. 2003 Apr;28(4):196-204.
132 Dong Q, McKee G, Pitman M, et al. Epithelioid variant of gastrointestinal stromal tumor: Diagnosis by fine-needle aspiration. Diagn Cytopathol. 2003 Aug;29(2):55-60.
133 Gu M, Ghafari S, Nguyen PT, Lin F. Cytologic diagnosis of gastrointestinal stromal tumors of the stomach by endoscopic ultrasound-guided fine-needle aspiration biopsy: cytomorphologic and immunohistochemical study of 12 cases. Diagn Cytopathol. 2001 Dec;25(6):343-350.
134 Li SQ, O’Leary TJ, Buchner SB, et al. Fine needle aspiration of gastrointestinal stromal tumors. Acta Cytol. 2001 Jan–Feb;45(1):9-17.
135 Rader AE, Avery A, Wait CL, et al. Fine-needle aspiration biopsy diagnosis of gastrointestinal stromal tumors using morphology, immunocytochemistry, and mutational analysis of c-kit. Cancer. 2001 Aug 25;93(4):269-275.
136 Wieczorek TJ, Faquin WC, Rubin BP, et al. Cytologic diagnosis of gastrointestinal stromal tumor with emphasis on the differential diagnosis with leiomyosarcoma. Cancer. 2001 Aug 25;93(4):276-287.
137 Akhtar M, Iqbal MA, Mourad W, et al. Fine-needle aspiration biopsy diagnosis of small round cell tumors of childhood: A comprehensive approach. Diagn Cytopathol. 1999 Aug;21(2):81-91.
138 Caraway NP, Fanning CV, Amato RJ, et al. Fine-needle aspiration of intra-abdominal desmoplastic small cell tumor. Diagn Cytopathol. 1993 Aug;9(4):465-470.
139 Ferlicot S, Coue O, Gilbert E, et al. Intraabdominal desmoplastic small round cell tumor: report of a case with fine needle aspiration, cytologic diagnosis and molecular confirmation. Acta Cytol. 2001 Jul–Aug;45(4):617-621.
140 Insabato L, Di Vizio D, Lambertini M, et al. Fine needle aspiration cytology of desmoplastic small round cell tumor. A case report. Acta Cytol. 1999 Jul–Aug;43(4):641-646.