CHAPTER 8 Lung, chest wall and pleura
Clinical aspects
Lung
Although sporadic reports of the diagnosis of lung carcinoma by fine needle aspiration cytology (FNAC) appeared as early as 1886,1 the impetus for widespread use of the technique only arose with the development of image intensifiers and television viewing, allowing localization of small parenchymal lesions.2 Recognition of the high accuracy rate of fine needle biopsy (FNB)2 and simpler methods to treat pneumothorax combined to bring this diagnostic tool within the reach of most hospital radiologists and pathologists. Infections and some diffuse benign processes may be proven by FNAC, but the main indication remains the diagnosis of localized intrathoracic lesions suspected of being malignant, particularly when less invasive investigations prove to be negative.3
The role of FNB has shifted from the diagnosis of malignancy in inoperable patients and the confirmation of metastases, to its use as a first-line diagnostic procedure on which management decisions are based,4,5 with a shift away from non-invasive methods such as sputum cytology towards FNAC.6,7 All intrathoracic sites, including mediastinum and deep hilar lung lesions are safely sampled using fine (less than 20 gauge) needles with CT8 and ultrasound guidance.9 Lung function and radiological studies can help predict the risk of pneumothorax.10-12 Transbronchial or transtracheal FNB13-19 permits sampling of submucosal masses such as carcinoid tumors20 and mediastinal lesions, enhances the role of FNAC in the staging of lung cancers13,15,16,21 and may be used for the diagnosis of pulmonary infection22 and cysts.23 The more recent addition of ultrasound guided transbronchial and transoesophageal FNB has improved accuracy of targeting perihilar submucosal and mediastinal masses.24 The presence of the pathologist at the time of the procedure leads to a reduction in the number of needle passes, may decrease the pneumothorax rate,25 and increase overall sensitivity and accuracy of tumor typing.15,26 FNAC in immunocompromised individuals has gained greater acceptance, especially for the diagnosis of infectious diseases.27,28 An important role has also been found in paediatric practice8 (see Chapter 17). With greater expectations by clinicians regarding tumor typing, demonstration of high rates of cytohistological correlation for the common types of tumors has been achieved.29-32
The diagnostic utility of FNB for difficult diagnostic lesions (e.g. neuroendocrine and pleural tumors)33-44 is now widely accepted, particularly when allied with cell block preparations,31 and the use of ancillary studies.45-47 The range of cytological findings in less common tumors such as sarcomas,48,49 lymphoid lesions50,51 and metastatic tumors,52 as well as in second primary tumors,52,53 along with the most likely mimics of malignancy, have been well described.54-57 The more recent requirements for molecular assessment of gene mutations relevant to targeted therapy in some lung cancers can be readily achieved by FNB with adequate cell block preparations.58
The place of FNAC in the investigative sequence
Fine needle aspiration cytology complements other diagnostic methods.3 It is often the first choice in lesions which are located in the mediastinum, pulmonary apex, medial upper lobe or peripheral lung. Fiberoptic bronchoscopy (FOB) with brushings and biopsy is usually effective in diagnosing centrally placed pulmonary lesions, while FNB has particular value in those cases in which FOB is non-diagnostic.3
Endoscopic bronchial ultrasound (EBUS) localisation with FNB is now playing an increasing role in the sampling of central lesions, staging of lung carcinoma and the diagnosis of sarcoid and lymphoma.59-63
There is value in rendering a preoperative diagnosis, as thoracotomy is generally contraindicated for small cell carcinoma, most lymphomas, most metastatic tumors and most infections. A specific diagnosis of benign lesions such as tuberculosis or pulmonary hamartoma helps avoid unnecessary surgical intervention.64 Additionally, a diagnosis of malignancy in patients at high operative risk allows surgeons to operate without fear that surgery may be unnecessary. FNAC is a cost-effective diagnostic method that can lead to shorter hospitalization, and a reduction in the number of diagnostic thoracotomies.65
FNB may be the only way of providing a diagnosis in inoperable patients. An unequivocal diagnosis of carcinoma and distinction between squamous cell carcinoma, small cell carcinoma and adenocarcinoma provides the clinician with sufficient information to select appropriate therapy. Raab et al. reported that FNB coupled with immunohistochemistry is both sensitive and cost effective for subclassifying malignancies in patients with a prior history of extrapulmonary cancers, when compared to bronchoscopy and thoracoscopy.66 FNB can occasionally be useful intraoperatively in place of frozen section when the use of incisional or large needle biopsy is contraindicated.67
Accuracy of diagnosis
The sensitivity of FNB in the diagnosis of malignant pulmonary neoplasms varies according to both the size and the site of the mass,68 as well as the experience of the radiologist69 and the pathologist. In experienced hands it now reaches over 90% in both non-academic and academic centers.70,71 A non-specific negative result on FNB does not exclude malignancy.3,72 Therefore, repeat aspiration, careful clinical follow-up or additional diagnostic procedures may be necessary for definitive diagnosis. The rate of false-positive diagnoses is usually less than 0.5%.71 The few false-positive diagnoses of malignancy are often due to reactive bronchiolar epithelial proliferation,73 squamous metaplasia,74 reactive mesothelial cell proliferation,29 or associated with lesions such as tuberculosis, chondroid hamartomas,75 granulomatous processes,73 other infections,76 or pulmonary infarcts. Occasionally, in samples obtained by EBUS, it can be difficult to determine whether the sample is derived from a central lung lesion or nodal deposit, a potential cause of false-positive diagnosis of nodal metastases. Tao advises caution in assuming that a lack of clinical or histological correlation is a result of false-positive cytological diagnosis.77 He introduced the term ‘false false positives’ for cases where longer clinical follow-up period or closer histological examination confirms the cytological diagnosis. Well-differentiated adenocarcinomas with minimal cytological atypia and small tumors within larger areas of radiological opacity are prone to this error.
Experienced cytopathologists can reliably identify the histological type in up to 90% of primary lung tumors29,31,32,70,78,79 and approximately 80% of their predictions of tumor type are correct;2,30,70 these rates are similar to those achieved with bronchial biopsy.31,80,81 High levels of cytohistological correlation relate to the combined use of smears and cell blocks, which allow better assessment of tissue architecture.31 Rapid assessment of aspirated material in the radiology theater enhances accuracy of diagnosis by allowing appropriate triage of material for ancillary techniques. Most cases of small cell carcinoma,30,35,79,82,83 well-differentiated adenocarcinoma,84-87 and squamous cell carcinoma88 are usually identifiable. However, poorly differentiated tumors of glandular or squamous origin and large cell carcinoma are more difficult to separate.88 More studies of the range of cytological findings in neuroendocrine carcinomas, particularly large cell neuroendocrine tumors, are needed.36-38 These may be difficult to distinguish from small cell carcinomas or other large cell carcinomas, although in the view of some authorities this distinction may be artificial or arbitrary at times.89 Pulmonary hamartomas64,75,90-94 can be accurately diagnosed in the majority of cases. Other benign neoplasms are difficult to type because their cohesiveness prevents aspiration of adequate diagnostic material. Thin core needle biopsy may add to diagnostic accuracy.95-97 Radiologists are increasingly prepared to perform core biopsies on intraparenchymal lesions96,97 and this can be useful in some settings, especially where there are unusual features by FNAC. It is worth noting that a core biopsy will not necessarily provide a diagnosis where FNB has failed, particularly if the FNB sample is extensively necrotic. Cytological evidence of infection including tuberculosis may be gained in up to 80% of cases.98-103 FNB can be used with a high degree of accuracy to diagnose bacterial, fungal or pneumocystis infection in immunosuppressed patients.104
Complications
Fatalities have occurred with 20-gauge or larger needles or in patients who are terminally ill or those with severe underlying pulmonary disease or infection.105 The most common cause of death is hemorrhage, although there are rare cases of air embolism, tension pneumothorax and cardiac tamponade.106-110 To our knowledge, no deaths have occurred in our experience of over 5000 cases.
The rate of pneumothorax varies from 6% to 57% and 1.5–20% require intercostal catheterization.105,111 Emphysema, deeply situated lesions, multiple punctures, older patients and inexperienced operators all contribute to a higher rate of pneumothorax.10,11,106 Examination of aspirated material in the radiology theater and the use of coaxial needles allows the total number of needle passes to be reduced, thereby reducing the rate of pneumothorax.25 A small hemorrhage into the surrounding lung occurs in up to 10% of cases without clinical complications. Examples of neoplastic implantation along a thoracic needle track have been reported,112-115 and one case of skin implantation with fungal organisms.116 Bacteremia may occasionally be induced.117
Contraindications
Patients who do not have a cough reflex, who are unconscious or who cough intractably should not be biopsied. Arteriovenous malformation or aneurysm is usually excluded before attempting FNB, but the risk of bleeding from large hilar vessels is minimal when fine needles are used. There is an increased risk of bleeding in patients with pulmonary hypertension or bleeding disorders and in those undergoing anticoagulant therapy. These patients should be biopsied only in exceptional circumstances, for example in immunosuppressed patients where a positive diagnosis of an infection may lead to curative therapy.104,118 Most authors do not recommend FNB of suspected hydatid cysts due to the possibility of an anaphylactic reaction to leaking cyst fluid or implantation of germinal epithelium. However, when aspiration of unsuspected cysts occurs, complications are very few.119,120 Concurrent bilateral biopsies are not recommended.
Technical considerations
Obtaining material
Most radiologists use needles of less than 20 gauge, with a stylet to minimize contamination with tissue from the needle track. Needles of 22–23 gauge provide satisfactory material for cell blocks. Finer needles lessen tissue damage, although their flexibility may reduce the chance of successful puncture in deep lesions. Automated core biopsies are safe for pleural-based or chest wall lesions,121 and their use in parenchymal lesions is increasing.96,97
The puncture site is marked on the skin and local anesthetic introduced down to the pleura. A guide needle makes aiming of the aspiration needles easier, reduces the risk of bending of flexible needles and prevents contamination of the aspirate with chest wall tissues.122 Non-aspiration techniques often yield adequate material and may be particularly useful in hemorrhagic lesions (e.g. metastatic renal cell carcinoma).123,124
CT guidance is generally used although ultrasound may be useful for apical, pleural or chest wall tumors.125-129 The use of coaxial needles means that repeated re-insertion of the needle is not necessary. Some lesions have distinctive characteristics when aspirated, e.g. pulmonary hamartomas have a rubbery resistance and a tendancy to ‘move away’ from the needle; aspiration of neural tumors may cause intense pain. Adequacy of the aspirated material should be assessed in the radiology theater after rapid staining. The presence of the pathologist at the aspiration optimizes appropriate triage of material. Preliminary assessment in the radiology suite may also identify an unusual lesion where a core sample may be helpful. If the material is heavily bloodstained, collection onto a slide or watch glass (see Chapter 2) enables small clumps of tumor to be recognized as bright translucent fragments against the red background. To reduce blood contamination, these aggregates are selected and smeared onto a separate slide. Carnoys’ fixative or acid alcohol are useful for lysing blood. Material is taken for ancillary studies such as flow cytometry, ultrastructural study or submitted for culture depending on the preliminary assessment of the aspirate. Our approach is to generate an adequate fixed smear which is assessed by rapid H&E stain; subsequent material is then triaged according to the appearances of the first aspirate. Other laboratories have found air-dried MGG-stained material to be useful for initial assessment. More smears may need to be assessed if the first pass does not contain diagnostic material or is not consistent with the clinical and radiological findings. However, the days of producing numerous highly cellular smears with no cell block should be over! It should also be remembered that many lung carcinomas will not be resected; the material obtained by FNB may be the only tissue available for subsequent molecular studies, which can determine suitability for a particular treatment, further emphasizing the need to obtain an adequate cell block in all cases.
Use of ancillary techniques45,46
We rely heavily on ancillary techniques in FNB of the lung and pleura. Ancillary testing is useful in over 50% of intrathoracic aspirations.45 O’Reilly et al. used immunocytochemistry in 14.5% of cases, EM in 8%, microbiologic studies in 12%, mucin staining in 21% and cell blocks in 22%. They found the greatest value was in classifying poorly differentiated neoplasms, confirming the diagnosis of well-differentiated adenocarcinoma, identifying neuroendocrine differentiation and establishing primary sites for suspected metastases.45 Saleh and Masood found ancillary tests contributed to diagnosis in over 60% of cases.46 Immunohistochemistry was most often used, but electron microscopy was often helpful. Immunohistochemistry has become the mainstay of cytological diagnosis, especially since the availability of thyroid transcription factor (TTF-1),130-132 and differential cytokeratin staining.131,133-135 Currently, EM is seldom used. Flow cytometry has revolutionized the diagnosis of lymphoid lesions.136-138 PCR-based molecular analysis for gene rearrangements for lymphoma diagnosis and FISH studies for lymphoma (see Chapter 5) and soft tissue tumor diagnosis (see Chapter 15) can be applied to FNB material. PCR studies to identify the presence of gene mutations, e.g. EGFR mutations which are relevant to the selection of particular therapies, can be performed on cell blocks.58 One can anticipate other techniques such as proteomics and microarray technology being useful.139,140
Cell blocks
Architectural features such as gland formation may be more readily appreciated in cell block H&E sections; the main role of cell blocks is to provide material for ancillary studies.
Cytochemistry
Mucin stains (which can be performed on smears or cell block material) aid in subtyping large cell carcinomas and in distinguishing between some primary and metastatic tumors, for example in excluding renal cell or adrenal carcinoma.
Immunocytochemistry
The full range of immunohistochemical markers can be used in cell block preparations. Broad-spectrum antibodies to keratins, S-100 and leukocyte common antigen (CD45) can separate most poorly differentiated carcinomas from melanomas and lymphomas. Calretinin has been the most useful positive marker of mesothelioma in our hands, but also stains some poorly differentiated lung carcinomas and squamous carcinomas; it is most useful in pleural effusion cytology. Neuroendocrine markers such as synaptophysin, chromogranin and CD56 are useful in diagnosing various neuroendocrine tumors. Markers for prostate-specific antigen and thyroglobulin are essential in selected cases. Estrogen receptor (ER) staining helps in identifying metastases of breast carcinoma. Caution is required with particular clones of ER.141 SP1 clone has a significantly higher rate for positive estrogen receptor expression in pulmonary adenocarcinomas when compared with either 1D5 or 6F11 clones. Caution is required in the use of SP1 antibody alone in distinguishing metastatic breast carcinoma from primary pulmonary adenocarcinoma. Differential cytokeratin staining for CK7 and CK20 can help separate primary adenocarcinomas of the lung (CK7 positive, CK20 negative) from metastatic colonic carcinoma (CK7 negative, CK20 positive).134,135 CDX2 is an additional positive marker of colonic malignancy. Rarely, primary lung adenocarcinomas of so-called enteric-type are CK20 positive and may also stain for CDX2; they usually retain their CK7 positivity.142 Squamous cell carcinomas and small cell carcinomas are usually negative for both CK7 and CK20, while squamous cell carcinomas are usually positive for CK5/6 and p63.143 TTF-1 is relatively specific for lung and thyroid neoplasms130,131 and is probably the single most useful marker for confirming primary lung adenocarcinoma, although perhaps inevitably there are now reports of cases of various metastatic carcinomas being TTF-1 positive, e.g. ovary, cervix, endometrium, colorectal.144-146 Antibodies to surfactant protein may also be helpful, but negative results in metastatic tumors have not been validated to the same extent as TTF-1. TTF-1 is said to be positive in 90% of small cell carcinomas,147 although we have seen several cases of TTF1-negative small cell carcinoma.
Electron microscopy (EM)
The role of EM (see Chapter 2) in the diagnosis of pulmonary neoplasms is declining, but may still be of value in recognizing neuroendocrine tumors, in the diagnosis of occasional cases of melanoma and mesothelioma and some carcinomas, including metastases, when immunocytochemistry has not provided a diagnosis.45-47,148,149 Demonstration of surfactant granules supports the diagnosis of primary bronchogenic carcinoma. Recent studies have further emphasized the heterogeneity of cell type in lung adenocarcinomas. Tumors may show features of nonciliated bronchiolar cell, alveolar cell, mucus-secreting cells or even mixed features of squamous or neuroendocrine type.150
Molecular testing151-159
There is a trend toward targeted therapy in oncological practice, usually based on the presence of particular gene mutations. At present, the main molecular testing in lung carcinoma is the assessment of EGFR gene mutations; these mutations are usually found in adenocarcinomas and are limited to exons 18–21, with more than 80% of mutations being short deletions in exon 19 or single point mutations in exon 21.151,152 This testing is usually performed only when there is consideration for treatment with small molecule inhibitors of EGFR. In addition, mutations in KRAS are associated with lack of benefit from EGFR inhibitors. Mutations in the EGFR and KRAS genes are almost never seen together in the same tumor.153 EGFR and KRAS status can be assessed by DNA extraction from cell block samples in which there is adequate tumor representation. PCR and sequencing studies are then performed.58 When there is insufficient material in a cell block sample, material can be scraped from slides.154 Bronchioloalveolar carcinoma (BAC), now defined as an in situ adenocarcinoma, is composed of two distinct subgroups – mucinous and non-mucinous. The more frequent non-mucinous BAC which predominates in smokers and often presents as a ground-glass opacity is thought to evolve from the terminal respiratory unit cells and frequently harbors EGFR polysomy/mutations which are considered to be a critical event in the pathogenesis of non-mucinous BAC tumors. The less frequent mucinous BAC, thought to be derived from metaplasia of bronchiolar epithelia, presents more frequently as a pneumonic-type infiltrate, rarely demonstrates EGFR mutations, and more frequently harbors and is driven by a KRAS mutation.153,155
There are various other molecular markers, e.g. ERCC1, RRM1, that appear to be of increasing clinical relevance, in particular to predict tumor response to chemotherapeutic agents.156,157 The recent description of an ALK–ELM translocation which can be detected by FISH and immunohistochemistry, and which is present in a small subset of adenocarcinomas, which will respond dramatically to a particular drug therapy,158,159 highlights the need to ensure that adequate samples are obtained for cell blocks in order to perform additional studies as required.
Clinical requirements in modern practice143,160-162
There are increasing expectations from oncologists to distinguish between adenocarcinoma and squamous cell carcinoma in order to optimize treatment regimens, e.g. bevacizumab is associated with a high risk of bleeding in squamous cell carcinoma and is approved only for adenocarcinoma.160 This has led to pressure to reduce, if not eliminate, the diagnosis of ‘non-small cell carcinoma’. An immunohistochemical panel including CK5/6, p63, CK7 and TTF-1 in poorly differentiated carcinomas, may help to suggest either possible squamous (CK5/6, p63 positive) or glandular (CK7/TTF-1 positive) differentiation.143 Pardo et al.161 reduced their diagnosis of large cell carcinoma by 90% in a tissue microarray study, with the use of immunohistochemical panels and Khayyata et al.143 achieved refinement of diagnosis in 65% of cases of NSCLC to either SCC or adenocarcinoma. While the distinction between adenocarcinoma and squamous cell carcinoma can usually be achieved morphologically in well-differentiated tumors, the addition of immunohistochemical panels may increase diagnostic accuracy in poorly differentiated carcinomas.162 However, limited sampling, anaplastic tumors, tumor heterogeneity and overlapping immunohistochemical profiles can limit the ability to subclassify some lung carcinomas, and oncologists need to be aware of these issues. It is also worth noting that in some studies there may not have been any ‘gold standard’ to confirm the immunohistochemical diagnosis. Even in a well-differentiated adenocarcinoma it is still incumbent on the pathologist to ensure that there is adequate cell block material for molecular studies (see above). As new discoveries come to light there may be the need to return to archival material to perform new tests. Once a smear with adequate material has been obtained, further aspirates should be submitted for cell block preparation to ensure sufficient material is available for any additional studies that may be required.
Chest wall and pleura
Chest wall is included here to emphasize that the exact site of origin of lesions in this region may be difficult to ascertain and that it is conceptually useful to consider this site as a ‘body compartment’. Lesions arising in rib, chest wall soft tissues, pleura or even breast may extend to underlying lung parenchyma and may be confused with primary lung tumors.163 Contaminating bone marrow from rib may give rise to diagnostic dilemmas. In some cases, myeloma can be mistaken for an undifferentiated lung carcinoma. There is sometimes difficulty in distinguishing between mesothelioma, anaplastic carcinoma, melanoma, sarcomas of the chest wall and metastases. In our own practice, a chondrosarcoma arising in the rib caused difficulty in diagnosis because the site of origin was not appreciated. A malignant mesothelioma growing along a chest drain site was considered clinically to be a primary breast lesion until FNB was performed. Primary soft tissue lesions of this region are described in Chapter 15.
Cytological findings
The satisfactory smear
A specific benign diagnosis by FNB is generally restricted to the identification of infection, chondroid hamartomas, cysts and a few other disease processes. Non-specific negative findings do not exclude malignancy,3 and can only be used for patient management in conjunction with clinical and radiological findings. A precise statistical assessment of accuracy rates for a radiology department and a laboratory should be known. In a sense, the only satisfactory sample is one which contains material permitting a specific diagnosis.
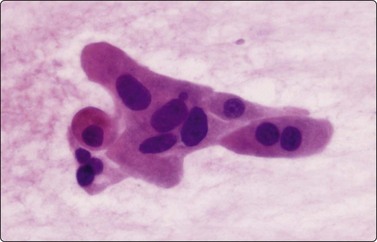
Normal structures (Figs 8.1-8.3)
Monolayered sheet of cells showing ‘spongiotic’ separation of individual cells within the sheet (Pap, LP).
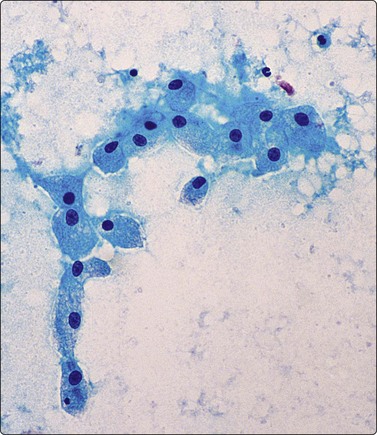
Loose aggregates and dispersed cells with rounded nuclei, small nucleoli, and abundant finely vacuolated cytoplasm (Pap, HP).
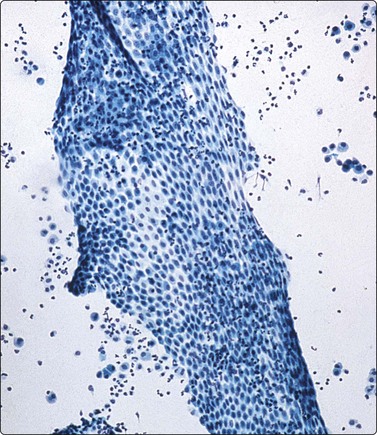
Bronchial epithelium may be abundant in percutaneous FNB if the needle traverses a medium-sized bronchus or tracks along a bronchial lumen. Transbronchial FNB may also yield abundant bronchial epithelium. Bronchial epithelium appears as small palisaded clusters with a ciliated border. In large aggregates the cells may present as flat sheets with a pavement-like aspect, but ciliated cells can usually be observed at the edges of these sheets.
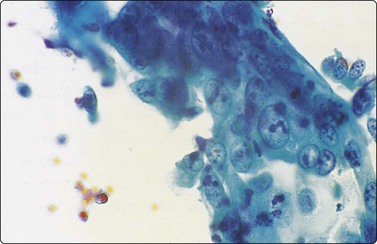
Bronchiolar epithelium or nonciliated epithelium is seen commonly as sheets of various size (Fig. 8.1). They usually have irregular edges and the component cells display variable cell separation. The nuclei are generally small and there is a low nuclear:cytoplasmic (N:C) ratio. Sometimes the nuclear outlines are slightly irregular and small intranuclear cytoplasmic inclusions are observed. Occasionally, atypia of bronchiolar epithelium may be quite pronounced, for example in reactive or inflammatory processes; however, the number of atypical cells is usually small. A diagnosis of malignancy should generally not be made on the basis of small numbers of cells.
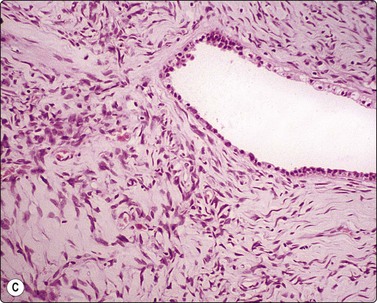
Mesothelium can be easily distinguished from bronchiolar epithelium. It is seen as various-sized, flat, monolayered sheets; there is usually more cell separation than in bronchiolar epithelium, which sometimes gives a sponge-like appearance. The cells appear to be joined by intercellular bridges similar to those of squamous epithelium in histological sections. (Fig. 8.2). Reactive mesothelium can appear atypical and be misinterpreted as neoplastic.
Normal lung yields a population of macrophages widely dispersed over the slide (Fig. 8.3); these contain small particles of brown or black particulate matter, some of which is inhaled dust, especially in smokers. Many hemosiderin-laden macrophages usually imply tissue or blood breakdown near the lesion and may add to a suspicion of pulmonary infarction should the clinical background be appropriate.
If stylets are not used, contamination of smears by chest wall tissues such as fat, skeletal muscle, cartilage and bone marrow may occur. Occasionally, intrapulmonary lymph nodes may be aspirated.
Aspirations near the diaphragm can contain liver or splenic tissue and cause concern unless the possibility of inadvertent puncture of abdominal organs is appreciated. Splenic tissue has also been identified in FNB samples from ‘thoracic splenosis’ following traumatic rupture of the spleen; these are usually pleural based but can occur within the lung.164,165
Megakaryocytes are occasionally seen in aspirates of lung166 or more often from rib marrow.
Inflammatory/non-neoplastic processes167
Acute inflammatory material
Common findings
These findings are not specific unless bacteria are evident, but they can suggest an acute infection. It is worth repeating the aspiration, and sending all the material obtained for aerobic and anaerobic culture. Associated vegetable material may suggest aspiration as a cause.168
When the center of a necrotic or cavitating carcinoma is aspirated, particularly those of squamous type, acute inflammatory material may result (the so-called carcinomatous lung abscess).169 Macroscopically, this resembles pus and microscopically only few squamous cells may be present. Small squamous cells with pyknotic nuclei may be difficult to distinguish from degenerate macrophages.
Polymorphonuclear leukocytes are not uncommonly seen in tuberculosis and the background debris may also be rather ‘watery’, similar to that in some acute inflammatory processes. We stain all inflammatory smears with the Ziehl-Neelsen (ZN) stain and routinely send material for microbiological culture or PCR studies for fungi and mycobacteria, as well as bacteria.
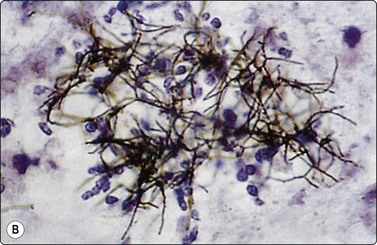
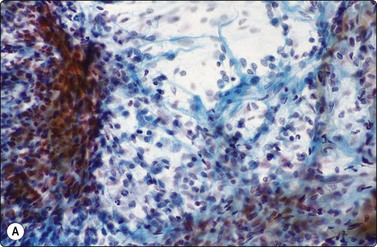
A high polymorph content is usually present in the inflammatory reaction to fungi. A search for a granulomatous component and fungal organisms is mandatory if acute inflammatory material is obtained. With infection by filamentous organisms, e.g. Nocardia, the organisms may be identified in the center of distinctive, cohesive neutrophil clusters or rosettes after silver impregnation; this clustering should be a clue to search for such an infection (Fig. 8.4). Similar appearances may also be be seen in streptococcal infection.
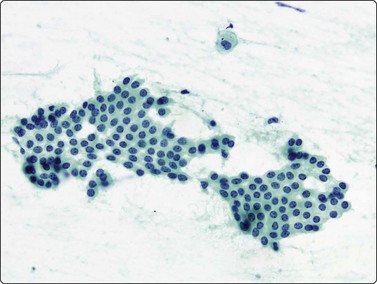
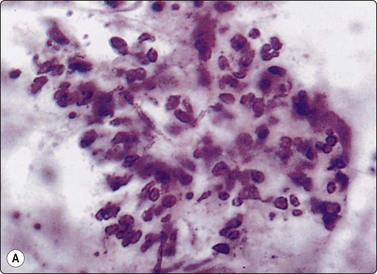
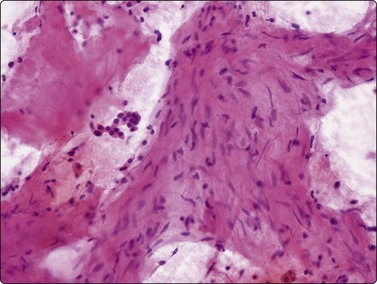
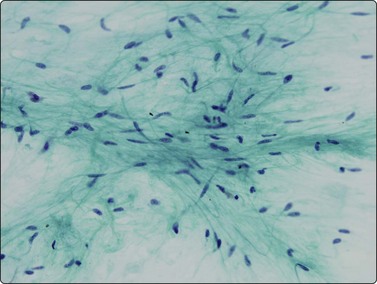
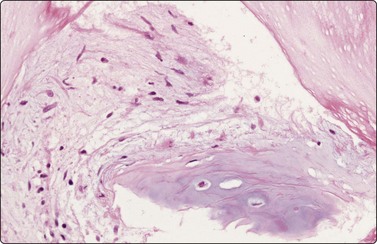
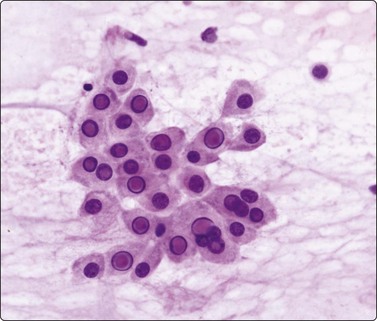
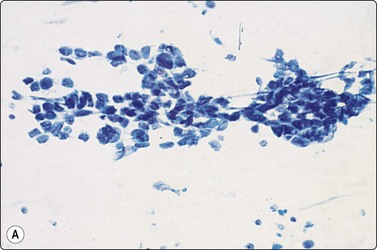
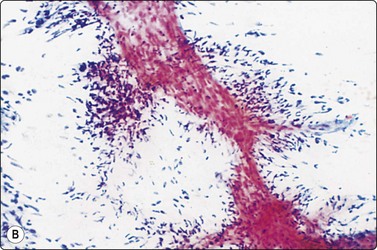
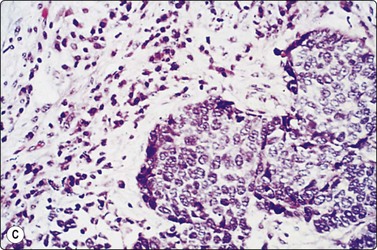
Granulomatous inflammation (Figs 8.5-8.7)98-103,170-176
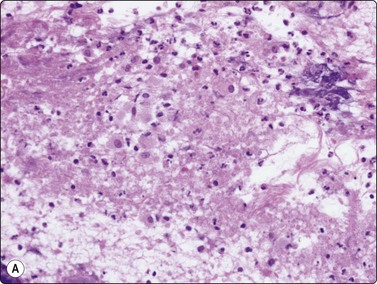
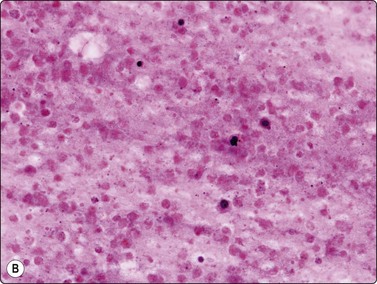
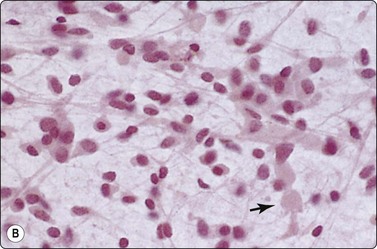
(A) Amorphous and granular debris with neutrophils (H&E, MP); (B) Single-cell pattern of necrosis (H & E, HP).
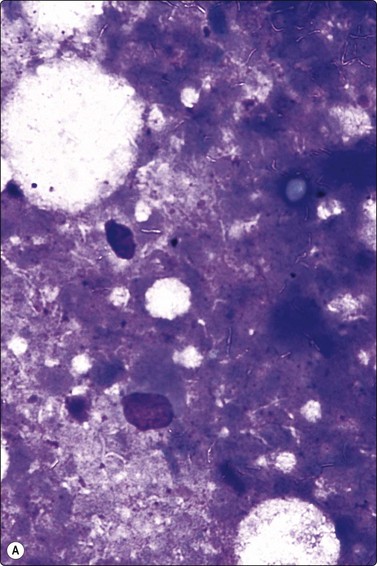
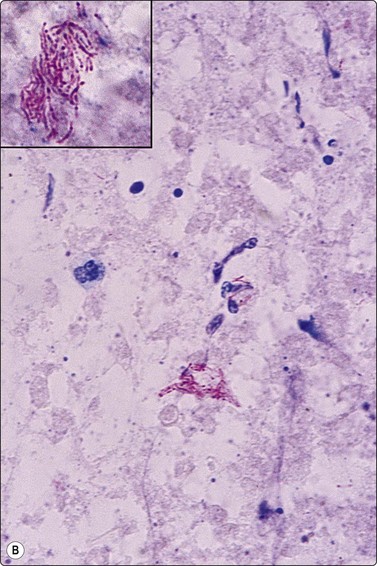
Fig. 8.7 Mycobacterial infection
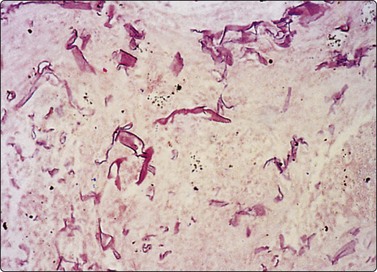
(A) Negative images of bacilli in a background of granular necrotic debris (MGG, HP); (B) Numerous acid-fast bacilli, including clumps of beaded forms (Ziehl-Neelsen, HP; Inset HP oil).
Criteria for diagnosis
One can make a confident diagnosis of a ‘granulomatous process’ on aspirated material; however, a more important aspect of the aspiration is to obtain an adequate sample for staining and culture, particularly to distinguish between ‘typical’ and ‘atypical’ mycobacteria101 and for typing fungal organisms. It is best to have a large sample for this purpose and we have not found much value in washings of the needle after a cytology smear has been made. Instead, we advocate sending the whole aspirate for culture. Culture for tuberculosis is more successful in cases with necrotic and inflammatory debris.99,102 PCR can provide a rapid diagnosis, aids in subtyping and can identify drug resistant strains.170,171
Specific diagnosis of most noninfectious granulomatous diseases is not possible, although there are reports of the cytological findings in sarcoidosis,172-174 Wegener’s granulomatosis175,176 and rheumatoid nodule.177
Epithelioid histiocytes are fairly cohesive and form granulomas which are often aspirated intact (Fig. 8.5). Epithelioid cells have an elongated or bean-shaped nucleus and abundant cytoplasm which is rather pale and indistinct both in Pap- and H & E-stained specimens (Fig. 8.5). The cytoplasmic density is higher in MGG-stained material. Multinucleated histiocytes can be seen but are usually sparse. They are mainly free of intracytoplasmic pigment or birefringent material, unlike the multinucleated histiocytes seen in non-specific reactions in pulmonary tissue. Caseous necrosis has a variable appearance. There may be an amorphous to granular background with little cell outline visible, but sometimes outlines of necrotic cells may be prominent (Fig. 8.6) and often the appearances are merely of nondescript debris, histiocytes and neutrophils. Dahlgren cites granular calcific material as a common accompaniment.98 Lymphocytes may be plentiful in granulomatous inflammation.
Bailey et al.99 diagnosed 28 of 34 cases of TB by either auramine rhodamine fluorescence or positive culture; acid-fast bacilli (AFB) were seen in only 38% of cases. In Rajwanshi’s102 and Das’s103 series AFB were identified in approximately half of the cases. Gong et al. found PCR to be about 80% sensitive compared to 40% for ZN staining in FNB material.171
Recent advances in mycobacterial genomics and human cellular immunology have resulted in two new blood tests that detect tuberculosis infection by measuring in vitro T-cell interferon-gamma release in response to two unique antigens that are highly specific for Mycobacterium tuberculosis, but absent from bacille Calmette-Guérin (BCG) vaccine and most non-tuberculous mycobacteria. These tests appear to be very useful in increasing specificity and sensitivity in the diagnosis of tuberculosis.178
Epithelioid histiocytes may resemble fibroblasts, smooth muscle cells or endothelial cells; they are most characteristically found in clusters and can rarely be identified if they lie singly.
Giant cells which contain pigment are more in keeping with a non-specific reaction than a true granulomatous process.
A granulomatous reaction may develop at the edge of a carcinoma or other neoplasm, particularly squamous cell carcinoma forming keratin; this reaction may be foreign body giant cell in type and yield many multinucleated cells. Histiocytic giant cell reactions also occur in fungal infections, e.g. cryptococcosis101,179-181 or histoplasmosis,101 so the cytoplasm of any giant cells present should be closely examined. It should be remembered that tuberculosis may coexist with carcinoma.
Necrotic tumor may be homogeneous or granular and closely resemble caseous material. Conversely, we have seen cases of tuberculosis where ghost outlines of single cells were distributed across the smear and closely mimicked necrotic tumor (Fig. 8.6).
Acid-fast bacilli are more often seen in cytological material characterized by a mixture of neutrophils, histiocytes, mucoid or necrotic material than in those lesions with a prominent epithelioid cell component,99,102 though culture is positive in a similar percentage of cases with and without epithelioid cells.103 Whenever necrotic debris is seen, we restain smears with ZN stains. Maygarden described mycobacteria as negative images in a stained background in MGG material (Fig. 8.7).182 Silverman reported negative images in both Diff-Quik and Papanicolaou-stained material in a BAL sample from a patient receiving clofazimine treatment for an atypical mycobacterial infection. The reddish, refractile and polarizable drug-derived crystals can impart a pseudo-Gaucher-like appearance to the cells, simulating the negative images of an atypical mycobacterial infection.183
Other specific infections
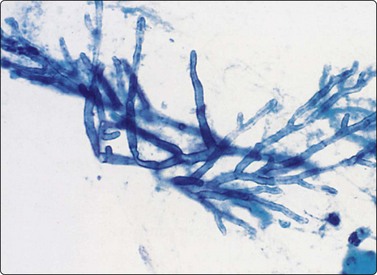
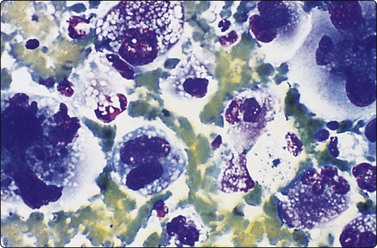
In Aspergillus (Fig. 8.8) and phycomycete (Fig. 8.9) infections in the lung, fungal hyphae are easily visible in alcohol-fixed material.184,185 The characteristic appearance of acute-angled branching, parallel cell walls and septa in Aspergillus (Fig. 8.8) is shared by several other fungi and culture is necessary for accurate typing.186 Pseudoallescheria boydii, identified as a ‘clinically significant mycosis’,187 and Fusarium sp. may be morphologically indistinguishable from Aspergillus, and even Candida resembles this organism on occasion, by forming germ-tube structures with ‘septation’.184 Oxalate crystals are produced by several species of Aspergillus and, if identified in cytological material, should lead to a search for organisms.188,189
Cryptococci179-181190 (Fig. 8.10) have a varied morphology.181 Later sclerotic foci have a more granulomatous appearance and fewer organisms. In several of our cases of cryptococcosis, organisms were only seen within multinucleated cells. A case of a pulmonary inflammatory myofibroblastic tumor caused by Cryptococcus infection, presumed to be the gattii species, has been diagnosed by FNAC.191 Mucicarmine staining of the capsule is a specific and useful criterion for diagnosis and will help distinguish the atypical forms from other yeasts; Fontana-Masson staining of cell walls will also help distinguish poorly encapsulated forms from Histoplasma, Torulopsis or Candida.181 Blastomycosis characteristically has more broad-based budding forms.
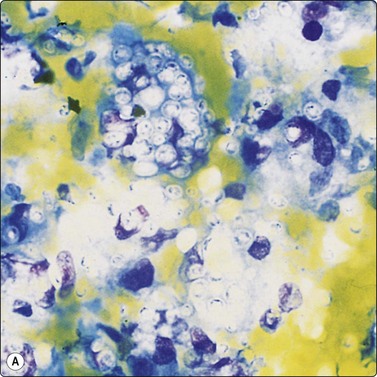
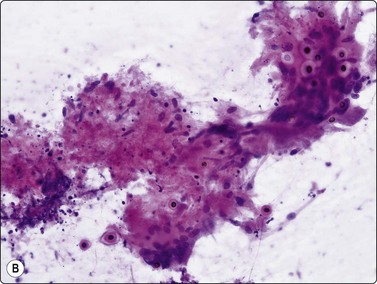
(A) Encapsulated yeast organisms (MGG, HP); (B) Necrotizing granulomatous material containing numerous encapsulated organisms. (H&E, MP).
Histoplasmosis may cause difficulties because the organism is difficult to see in ordinary stains and shows only as small refractile shadows. Methenamine silver preparations are required for diagnosis. Penicillium marneffei may mimic Histoplasma, as may the small form of Cryptococcus.
In some fungal infections, particularly histoplasmosis, organisms are only seen in methenamine silver preparations, although the Pap stain is a superb technique for identifying most other organisms including the filamentous higher bacteria.192 Destained H&E or Pap-stained smears are as suitable as unstained material for special stains. Culture is necessary for the exact classification of the organism.
Multiple and unusual infections may be encountered in AIDS or post-transplantation.27,28,193,194 Pneumocystis is uncommonly diagnosed by FNB because of the efficacy of broncho-alveolar lavage in the diagnosis of diffuse pneumonic disease, but some cases may present with nodular or cavitary lesions in the lung. CMV infection,195 coccidioidomycosis,76,196,197 paragonamiasis,198 amebiasis, hydatid disease119,120,199 and microfilariasis200 have all been described in FNB samples.
In coccidioidomycosis,197 smears show abundant granular eosinophilic debris with a paucity of acute or chronic inflammation; granulomatous inflammation is seldom seen. Coccidioides immitis spherules range in size from 20 to 200 µm; fractured or crushed forms are often seen and some are calcified. Endospores are seen only within intact spherules.196,197 In some cases, the associated reactive cellular changes have been misinterpreted as neoplastic.76
According to McCorkell,120 clear colorless fluid is an indicator of hydatid cyst and should lead to a search for scolices, hooklets or laminated membrane (see Chapter 10). Diagnosis of dirofilariasis201 requires cell block preparations (Fig. 8.11).
Reparative and reactive change
Squamous metaplasia, epithelial repair reactions, reactive bronchiolar or mesothelial proliferations and reactive histiocytic or fibroblastic cells can all produce significant cellular atypia (Fig. 8.12).56,73,76 Diagnosis of malignancy in a background of inflammatory change should be made with extreme caution. In FNAC, a malignant diagnosis should also generally be based on highly cellular smears. In this way, the risk of a false-positive diagnosis in reparative or reactive processes can be minimized.56,167,202
Pneumoconioses
Birefringent silica and collagenous tissue (Fig. 8.13) or asbestos bodies may help confirm silicosis or exposure to asbestos.203 However, concomitant malignancy, tuberculosis or other infections may be the cause of the localized opacity in these patients.204
Pulmonary infarct
Silverman describes sheets of metaplastic squamous cells showing regenerative changes and histiocytes containing clumped refractile hemosiderin in a clinically unsuspected infarct; FNAC findings led to the diagnosis being suggested. 205
Other non-neoplastic lesions
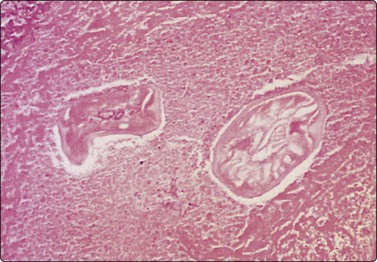
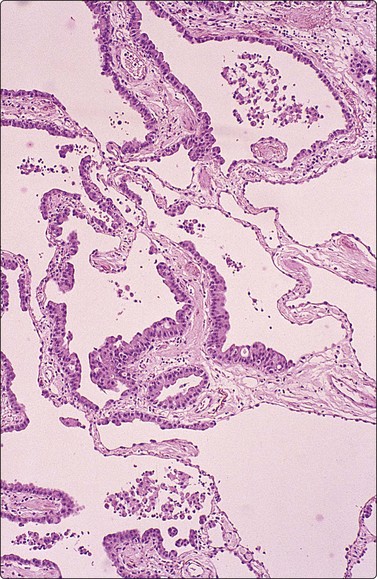
Material aspirated from congenital bronchogenic cysts usually consists of mucin and bronchial-type epithelium unless infection supervenes. So-called ‘bronchoceles’, which represent recent-onset postinflammatory dilated mucin-filled bronchial structures, may yield similar findings, and correlation with clinical and sequential imaging findings is necessary for precise diagnosis. (Fig. 8.14).
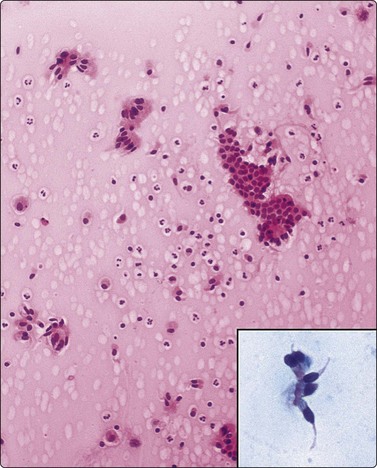
Rounded mass in mid-lung field. Mucoid material aspirated at FNB. Smears show mucin, scattered macrophages and sheets of bronchial epithelial cells. Inset: Ciliated cells. Cytological findings consistent with either bronchogenic cyst or bronchocele. Recent onset of lesion more consistent with bronchcoele (H&E, HP; Inset, Pap, HP oil).
Findings in nodular amyloidosis (Fig. 8.15),206-208 Wegener’s granulomatosis,175,176 extramedullary hemopoiesis209 and malacoplakia210 are described. We have seen one case of amyloidosis in consultation, in which amyloid was visible in both smears and cell block preparations. Miller et al. describe FNAC findings of abundant parenchymal material including thickened alveolar walls as being of help in diagnosing ‘rolled’ or ‘rounded’ atelectasis in company with consistent CT findings. This entity produces a radiologic appearance of a mass lesion involving pleura and lung, often in the inferior lobes in the posterior basal lateral vertebral area, and is sometimes associated with asbestos-induced pleural disease with underlying lung scarring.57,211
Common primary carcinomas
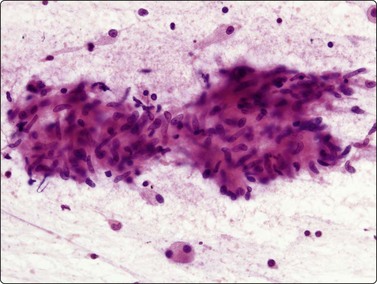
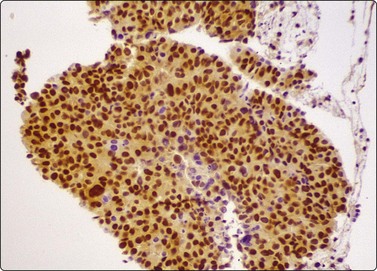
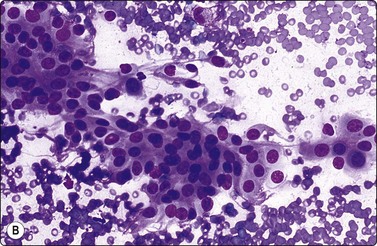
Squamous cell carcinoma (Figs 8.16 and 8.17)
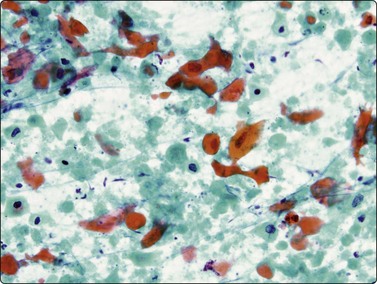
Fig. 8.16 Squamous cell carcinoma
Dispersed keratinizing malignant cells and necrotic debris (Pap, HP).
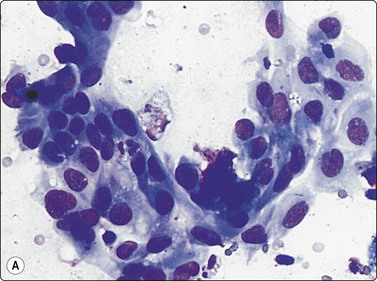
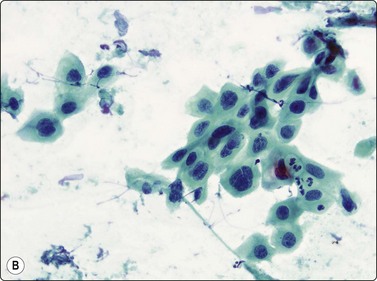
Fig. 8.17 Squamous cell carcinoma
Irregular clumps of relatively cohesive nonkeratinizing cells; variation in chromatin density (A, MGG, HP; B, Pap, MP)
Criteria for diagnosis
Well-differentiated squamous cell carcinoma with keratinizing cells and bizarre cell shapes is not difficult to recognise. The malignant cells are usually dispersed, especially when they are very well differentiated (Fig. 8.16); necrosis is a common accompaniment. Single keratinizing cells are the most reliable indicators of squamous differentiation; when keratinization is not obvious, a perinuclear halo within the cytoplasm and condensation of peripheral cytoplasm are helpful guides to squamous differentiation (Fig. 8.17). Non-keratinizing tumors are usually more cohesive and present as multilayered fragments. Their nuclei are usually spindle shaped or elongated with dense, irregularly distributed chromatin. There is often conspicuous variation in the degree of chromasia of nearby nuclei. Nucleoli vary in size and number, in contrast to adenocarcinoma where nucleolar morphology is more monotonous. Dense cytoplasm and well-defined cell borders are indicators of squamous differentiation, but some adenocarcinomas may have strikingly well-defined borders and very dense cytoplasm.
Misinterpreting the cytoplasmic eosinophilia of necrosis for keratinisation is a common source of error (see Fig. 8.30).30 Necrosis can also impart an angular hyperchromatic appearance to nuclei, similar to squamous carcinomas. This is less of a problem when Pap staining is used because orangiophilia distinguishes between necrosis and keratin better than H&E or MGG, although cells in heavily bloodstained smears may take on an eosinophilic hue simulating keratinization.
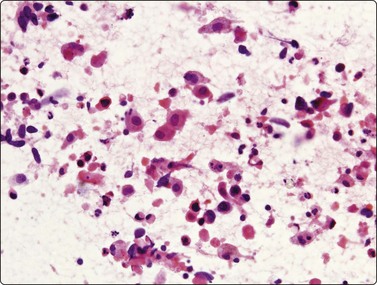
Fig. 8.30 Necrosis simulating squamous differentiation
Necrosis, eosinophilia and pyknosis in a metastatic deposit of colonic adenocarcinoma (H&E, HP).
There is an overlap between the appearances of poorly differentiated squamous cell carcinoma, large cell anaplastic carcinoma and poorly differentiated or solid adenocarcinoma in aspirated material. They all present as large, multilayered fragments corresponding to the solid growth of these carcinomas. Squamous cell carcinoma is overdiagnosed in some series because adenocarcinoma in aspirates may present as pavement-like sheets, mimicking the appearance that squamous cell carcinoma has in sections. The pavement-like appearance of squamous cell carcinoma results only from thin sectioning and does not occur in aspirated material, although it may be seen in sections of cell blocks. Palisading of cells, which may occur at the margins of lobules of nonkeratinizing squamous tumors, may give rise to a false impression of the margin of a gland in smears. There is also a tendency to overestimate the grade of tumor in aspirated material, perhaps because viable rather than maturing or exfoliating new growth is removed. Unusual variants of SCC such as the pseudovascular adenoid form may be easier to recognize as squamous in cytological rather than histological material.212
Small-celled forms of poorly differentiated squamous cell carcinoma, and those undergoing degeneration and necrosis, can be confused with small cell carcinoma. Attention to greater cohesion, larger cell size, presence of nucleoli, greater tendency to spindle at the periphery of clusters, denser cytoplasm and occasional keratinized cells should lead to the correct diagnosis. Strong positive p63 and CK 5/6 staining and absence of staining for TTF-1 would favor squamous rather than small cell carcinoma, although rare squamous carcinomas are TTF1 positive.143,213
Diagnostic malignant squamous cells may be difficult to find when there is associated acute inflammation and abundant necrosis, especially in cavitating tumors. These aspirates can simulate an abscess, leading to false-negative diagnosis. A comprehensive search for diagnostic malignant keratinized cells is recommended.
Keratinizing squamous cell carcinoma, more than other carcinomas, tends to produce a giant cell response in nearby tissues and a false impression of a granulomatous process. A variegated population of many and different types of inflammatory cells, foamy macrophages, mast cells and alveolar cells results from bronchial obstruction associated with the carcinoma. Other types of carcinoma, especially adenocarcinomas, do not usually show this reaction.
Occasionally, only keratinous debris or very well-differentiated squamous cells may be removed from a squamous cell carcinoma and cells with diagnostic malignant nuclei may be absent. Clinical and radiological features may then aid in diagnosis.
Teratoma enters the differential diagnosis, particularly when a lesion has a mediastinal component. In the aspirate from the mature teratomas we have seen, the squamous epithelium was very cohesive, in contradistinction to the dispersed cells of well-differentiated squamous cell carcinoma.
Low-grade mucoepidermoid tumors have been described in FNB samples214 as well as basaloid squamous carcinoma.215
Metastatic squamous carcinoma from head and neck or from anogenital primary sites will often be positive for high-risk HPV DNA, whereas primary lung SCC is rarely HPV positive.216 PCR studies for HPV DNA on cell block material may be helpful when metastatic disease is being considered.
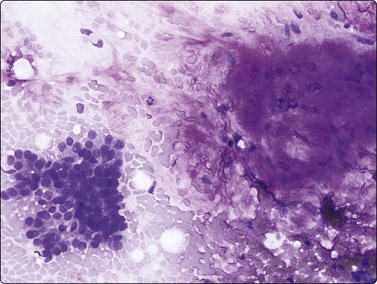
Adenocarcinoma, including BAC (Figs 8.18-8.24)
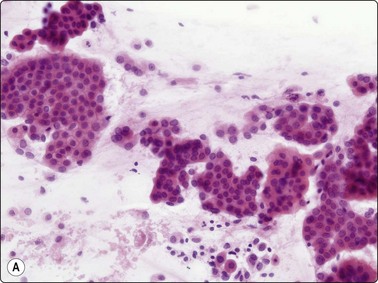
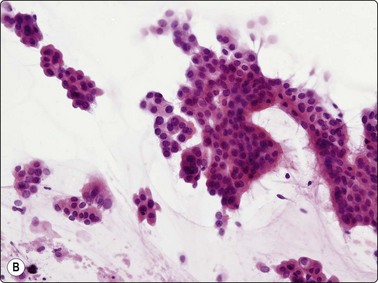
Fig. 8.18 Adenocarcinoma, well differentiated
(A) Monolayered sheets and papillaroid clusters of glandular cells (H&E, HP); (B) 3-D clusters and a sheet of glandular cells with a palisaded edge (H&E, HP). Diagnosis of malignancy based on abundance of material and complex architecture.
(A) Monolayered sheet of glandular cells showing enlarged hyperchromatic nuclei with irregular outlines and several intranuclear cytoplasmic inclusions (H&E, HP). (B) 3-D clusters of glandular cells (MGG, HP). Inset: intranuclear cytoplasmic inclusions (MGG, HP oil).
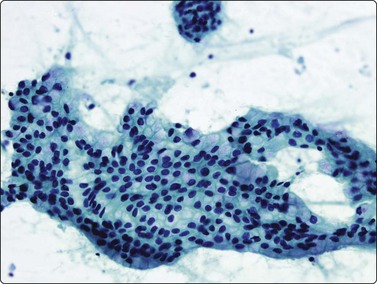
Fig. 8.21 Well-differentiated adenocarcinoma
Monolayered sheet of glandular cells with honeycombing and prominent mucin (Pap, HP).
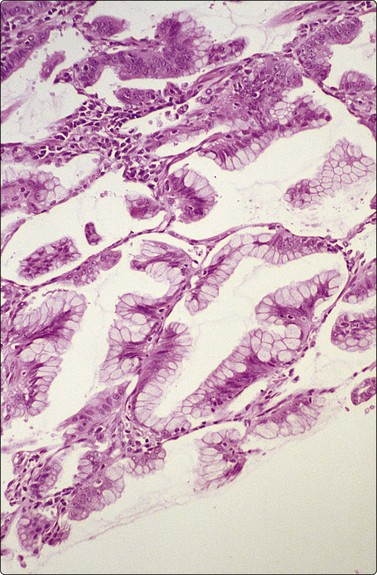
Fig. 8.22 Well-differentiated adenocarcinoma
Mucinous with lepidic growth pattern. Tissue section (H&E, MP).
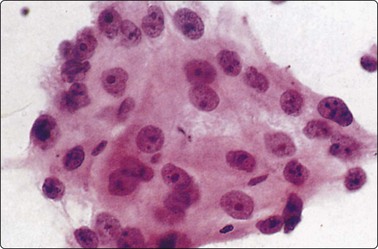
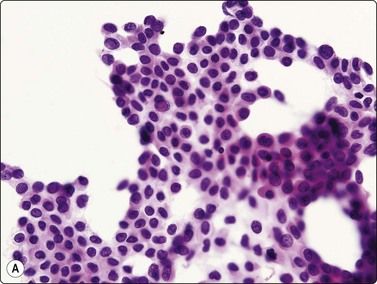
Fig. 8.23 Poorly differentiated adenocarcinoma
Delicate cytoplasm, rounded nuclei with single prominent central nucleoli (H&E, HP). Objective criteria of glandular differentiation not present in this aggregate; diagnostic criteria need to be sought elsewhere in smear or cell block.
The cellular morphology of adenocarcinoma is similar to that described in brush material. Rosettes, acinar formations or cohesive cell clusters (Figs 8.18 and 8.19) represent anatomical structures removed from the tumor by the needle. The larger the gland formations, the less likely they are to be removed intact and, when only partly removed, deposit on the slide as flat sheets in a monolayer (Fig. 8.18): a useful indicator of glandular differentiation. Artifactual spaces are often seen in large tissue fragments and may be misinterpreted as acinar structures; however, where the spaces have an ‘anatomical’ rigidity, they may indicate glandular differentiation. Mucin secretion is difficult to identify without the aid of special stains, and vacuolation of the cytoplasm may occur as a result of degeneration or the presence of glycogen. In H&E- or Pap-stained material vacuoles with a central, inspissated, eosinophilic or orangiophilic center are very suggestive of mucin secretion and correspond to the intracellular lumina described ultrastructurally in adenocarcinomas. With MGG staining, mucin may be visible as magenta or purple material within the cytoplasm, either homogeneously or as red granules within a pale vacuole. Well-formed columnar cells or groups of palisaded cells may be a guide to glandular differentiation and terminal plates/bars may also be present. In the 2004 WHO classification,217 the definition of BAC was limited to a non-invasive process and is now an uncommon diagnosis, requiring full histological assessment; there is currently debate as to whether the term should be used at all, especially since a diagnosis of BAC cannot be rendered on small biopsy samples or cytological samples. However, it is still possible to suggest that a tumor may have a BAC-like component based on cytological features. These include large, cohesive, monolayered sheets which reflect the growth of neoplastic cells in a monolayer along alveolar walls, papillary processes, cell balls and clusters, intranuclear cytoplasmic inclusions and psammoma bodies (Figs 8.18-8.22). The radiogical appearance of a purely ground-glass opacity without a solid component is also suggestive of a BAC.
In adenocarcinomas with meager mucin secretion and little or no glandular formation, the possibility of accurate diagnosis by morphological features is low. The cytoplasmic and nuclear features of large cell carcinoma and of some poorly differentiated squamous cell carcinomas may closely resemble adenocarcinoma; immunostaining for p63, CK5/6 and TTF-1 may be useful.143,213 If the tumor cells are fairly regular and the nuclei are rounded with large central nucleoli they are more likely to be glandular (Fig. 8.23); this is not, however, a completely reliable criterion.
There are no absolute cytological criteria for separating primary from secondary adenocarcinomas, although there may be features indicative of a particular organ of origin (see Chapter 5). Immunostaining for TTF-1 provides the best single marker of a primary origin (Fig. 8.24), although it should be recognised that metastic thyroid malignancies and occasionally metastases from other sites may be TTF-1 positive.144-146
It is likely that so-called atypical adenomatous hyperplasia (AAH) represents a precursor of peripheral adenocarcinomas of the lung.218 Foci of AAH are usually small (<5 mm), incidentally found in lung resections and are seldom sampled by FNB. Nevertheless, they potentially represent a source of error in FNAC diagnosis since they can be composed of highly atypical glandular cells, and may produce small opacities.219 We have encountered one case where the mass lesion seen on CT proved to be an area of non-specific fibrosis at resection and where the small numbers of highly atypical but rather poorly preserved cells seen in FNB samples were derived from adjacent areas of AAH.
It may be difficult to diagnose well-differentiated adenocarcinoma/BAC when there is a low degree of nuclear atypia. The material may mimic benign bronchiolar epithelium, especially when occurring in small sheets. The presence of abundant material should raise the index of suspicion, as should papillae or cell balls. Sometimes there may be only few diagnostic cells so that an overall impression of variation in atypia can be important in reaching a conclusion. Aspirates from non-malignant lesions of the lung frequently contain bronchiolar epithelium, sometimes showing nuclear atypia.84,167 Reactive sheets are usually small and few in number,86 and have more irregular borders than malignant sheets. Jarrett and Betsill considered high cellularity, architectural three-dimensionality, intranuclear cytoplasmic inclusions and large nucleolar size to be the most helpful features in distinguishing adenocarcinoma from reactive bronchiolar proliferations.86 Zaman et al. suggested a predominance of two- and three-dimensional tissue fragments, intranuclear cytoplasmic inclusions and paucity of multinucleated cell forms to be more indicative of malignancy.202 Silverman found a lack of hyperchromasia, prominence of cell borders, presence of terminal plates, goblet cells and nuclear molding to be more characteristic of benign reactive epithelium.84
Confusion with other primary pulmonary neoplasms may occur. Lesions such as carcinoid tumors and rare lesions such as sclerosing hemangioma/pneumocytoma220-223 yield a uniform cell population similar to that of well differentiated adenocarcinoma. Carcinoid tumors usually consist of regular cells which are smaller and more dispersed than those of adenocarcinoma and their round nuclei have a characteristic stippled ‘neuroendocrine’ chromatin pattern (see Figs 8.33 and 8.34). We have not seen examples of papillary adenoma in FNB material, but these rare tumors are also likely to provide problems in differential diagnosis. Pulmonary mucinous cystic neoplasms (PMCN) are also rare; distinction from a variety of pulmonary neoplasms, including mucinous BAC, bronchial mucous gland adenoma, mucoepidermoid carcinoma and metastatic mucinous adenocarcinoma, may be difficult.224 Moran considers that a clear morphological distinction between the benign and malignant forms of PMCN has not been convincingly made and that all lesions should be considered at least potentially malignant.225 Categorization as a cystic tumor may be important in planning surgery, as circumscribed lesions with minimal invasion are likely to have a good prognosis.
The differential diagnosis between a peripheral well-differentiated adenocarcinoma, including those showing a pseudo-mesotheliomatous growth pattern, and the epithelial form of malignant mesothelioma can usually be resolved by mucin and immunoperoxidase stains; occasionally, ultrastructural studies are required.
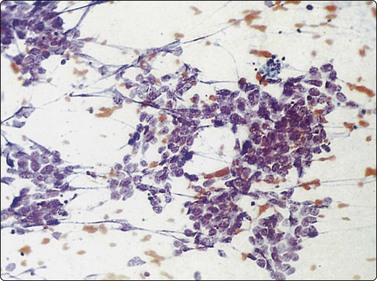
Small cell carcinoma (Figs 8.25-8.29)32,35,36,79,82,83
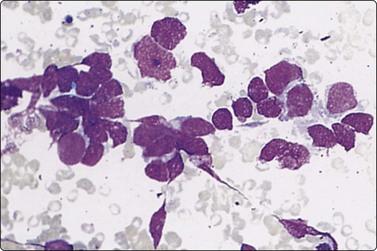
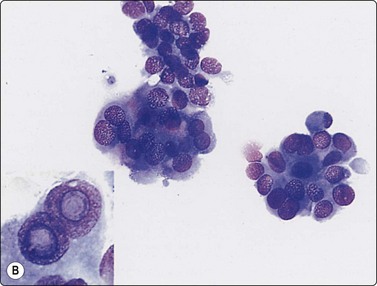
Fig. 8.26 Small cell carcinoma
Pleomorphic poorly cohesive cells with little or no cytoplasm; nuclear molding (MGG, HP).
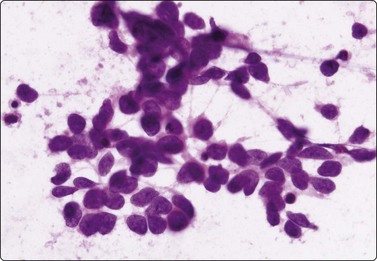
Fig. 8.27 Small cell carcinoma
Small loose cluster showing absence of cytoplasm, finely granular chromatin, inconspicuous nucleoli, nuclear molding and teardrop cells (H&E, HP).
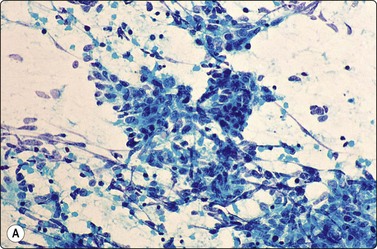
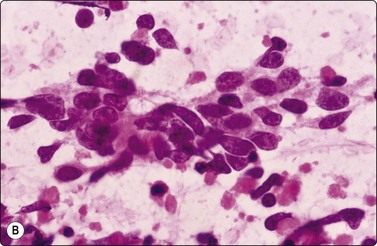
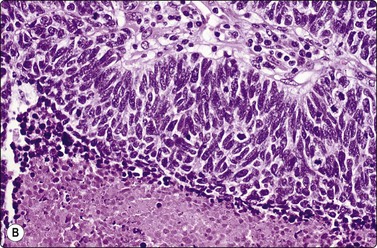
Fig. 8.28 High-grade neuroendocrine carcinoma; small cell carcinoma
(A, B) Smears showing ‘intermediate’ morphology. Loose aggregates of fragile cells with traumatization artifact and nuclear molding but some background cytoplasm (A, Pap, MP; B, H&E, HP). (C) Tissue section of resected peripheral stage 1 small cell carcinoma (H&E, HP).
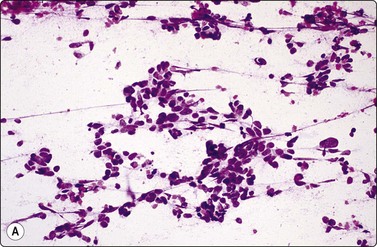
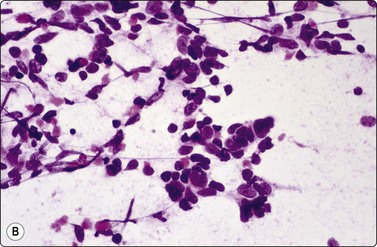
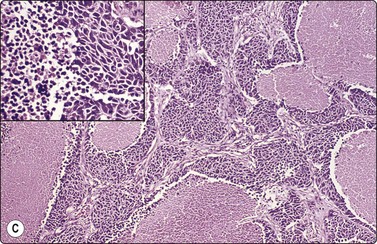
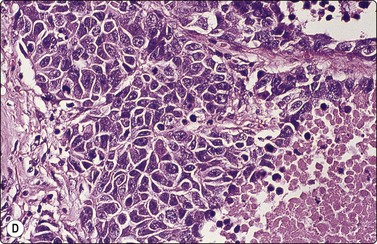
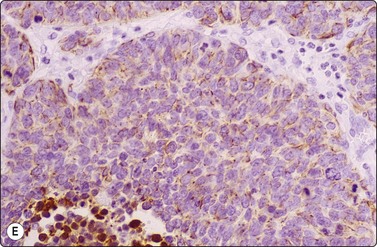
Fig. 8.29 High-grade neuroendocrine carcinoma
Variable morphology including small cell and large cell patterns. (A) Low-power smear appearances of small cell carcinoma; (B) Higher-power examination shows some large cells with prominent nucleoli (A, H&E, LP; B, H&E, MP). (C,D) Tissue sections of resected peripheral stage 1 tumor showing areas of geographic necrosis and a predominance of large cells with prominent nucleoli (C, tissue section, H&E, LP, Inset, HP; D, tissue section, H&E, HP). (E) Positive immunostaining for chromogranin in resected specimen (E, tissue section, IPOX, HP).
This group of lung carcinomas is the most aggressive of the common types, having a mean survival of less than 6 months without treatment. Small cell carcinoma is virtually unheard of in non-smokers, while for carcinoid and atypical carcinoid the smoking association is much weaker. It is important to categorize this neoplasm accurately because, in general, chemotherapy rather than surgery will be used in management. In addition, chemotherapy regimens are different from those used for inoperable non-small cell carcinomas. This group is fairly homogeneous in terms of its biology but is more heterogeneous morphologically. Attempts at morphological subclassification have been made; however, the larger ‘intermediate’ and smaller ‘oat cell’ subtypes are not reliably separable by expert pathologists and do not have significantly different behavior or response to therapy. The latest WHO classification therefore does not subcategorize small cell carcinoma although it does recognize mixtures with other types of carcinoma.217
Cytology is very successful in diagnosing small cell carcinoma in sputum and pleural fluid; in fact, sputum cytology may be more accurate than FNAC in typing this lesion. The criteria for the diagnosis of small cell carcinoma in aspirated material are similar to those in other sample types, but there are some important differences.
Cell pleomorphism is so distinctive that a diagnosis of malignancy is seldom in doubt (Figs 8.25-8.27). The most immediate impression is the absence or sparseness of cytoplasm rather than the small size of the neoplastic cell (Figs. 8.26 and 8.27). In fact, the cell nuclei may appear larger than similar cells in sputum and this may mislead one into making a diagnosis of non-small cell carcinoma. This difference in size between sputum and aspirated material is due to degenerative changes and shrinkage in sputum. It is sparseness of cytoplasm rather than size which is the most helpful initial clue in differentiating the lesion from other pulmonary carcinomas.
The combination of dispersal with clustering is also important, especially when other small cell neoplasms enter the differential diagnosis (Fig. 8.25). Lymphomas generally do not display such cell cohesion, although large fragments may be dislodged, and in some cases lymphoid cells may form clusters or packets.
Fragility of nuclei is emphasized by tear-drop cells or streaks of smeared nuclear material,226 and the close nuclear apposition and molding so commonly seen in sputum are also evident (Figs 8.26 and 8.27). Uniform coarsely granular ‘salt and pepper’ nuclear chromatin is also a well-recognized feature of this cancer in other sites, but one point of difference from sputum is the frequency of small nucleoli in aspirated material; they are less commonly seen in sputum. This may also be related to the better preservation of cells removed directly from tumor; small nucleoli are also often seen in bronchial brush material. Mitotic figures are usually easily found.
Mullins et al. described paranuclear blue inclusions as a common feature of small cell carcinomas;227 similar inclusions are seen in some non-small cell carcinomas but we have not seen them in lymphoma.
Renshaw et al.,228 in a review of QA material, found that misclassification of small cell carcinoma as non-small cell carcinoma may reflect a variety of factors, including lack of recognition that some features of non-small cell carcinoma may also be noted in well-preserved cases of small cell carcinoma; there may be increased cytoplasm and cytoplasmic globules (paranuclear blue bodies), or apparent intracytoplasmic lumina. Cases more frequently misclassified as non-small cell carcinoma tended to show better overall cellular and group preservation.
In the largest series the predictive value of a diagnosis of small cell carcinoma by FNB is over 90% and the sensitivity of tumor typing over 80%.30,82,83 However, there are few series with complete histological follow-up and some series where overall accuracy was lower. Reasonable experience with FNB diagnosis is necessary before the diagnosis can be assumed to be as reliable as by biopsy or sputum cytology.
Problems and differential diagnosis
Although ‘intermediate’ small cell carcinoma is no longer recognized as a separate category in international classifications, we find it a useful concept to highlight the occasional difficulty in distinguishing between small cell and poorly differentiated non-small cell carcinomas (Figs 8.28 and 8.29). There is overlap in nuclear size between small and large cell carcinomas and a tendency for inexperienced cytologists to include small cell carcinomas with larger than expected nuclei in the non-small cell category. In general, if the nuclear features of a problematical tumor are those of small cell carcinoma – that is, granular chromatin without prominent nucleoli – the neoplasm will usually fall into the small cell carcinoma group histologically, whereas vesicular nuclei with prominent nucleoli would generally be evidence of non-small cell tumor. However, large cell neuroendocrine carcinoma does provide special problems. Our experience is limited but is similar to Yang et al. who described various morphologic patterns in this family of tumors, including small cell-like and mixed small cell/large cell-like FNAC patterns.37 Cell size is therefore an important criterion and one to be critically evaluated. Tumors with nuclei larger than 2–3 times the diameter of a lymphocyte may be classified as LCNEC histologically, even if nuclear chromatin pattern and other cytological features are similar to those of small cell carcinoma (Fig. 8.29). Our approach is therefore to first come to a diagnosis of ‘high-grade neuroendocrine carcinoma’ and then to critically examine cell size and morphology to determine the best category – ‘small’ or ‘large’. We do, however, agree with the idea propounded by Marchevsky et al.89 that the distinction between the two categories may be somewhat artificial in view of the overlap in cell size between the two groups. This is an area which requires close cooperation with oncologists and an acceptance of the limitations of cytological diagnosis. It may be necessary to base management on clinical and staging findings in conjunction with inconclusive cytological tumor typing in some cases.
Cell cohesion is not a feature of small cell cancer and, when present, some caution should be adopted; distinction from small-celled squamous cell carcinoma with minimal cytoplasm may be especially difficult. Basaloid squamous carcinoma, analogous to the tumors described in upper respiratory tract, may rarely occur as a primary lung tumor or more commonly as a metastasis.229,230 Greater cell cohesion and the presence of even minimal or focal squamous differentiation will be helpful in diagnosis. ‘Borderline’ tumors exist, where there may be difficulty deciding even histologically between a diagnosis of small cell and non-small cell carcinoma. Combined tumors may occasionally be diagnosed by cytology.231 Discrepancies between cytological and histological classification can occur because of combined or collision tumors or separate synchronous primaries where cytological sampling may not reveal all tumor elements. Ten percent of predominantly non-small cell tumors have a small cell component histologically.232 A high percentage of small cell carcinoma cases (28%) are said to show combinations with non-small cell lung cancer, with large cell carcinoma the most common.233
A few very large cells are occasionally seen in typical small cell tumors. These usually lie singly and scattered across the smear; however, when clusters of larger cells are present or when these cells are numerous, caution should be exercised. Combined tumors of mixed small and large cell type are said to have a worse prognosis and to be less sensitive to therapy.234
Our approach to problem cases has been to request more biopsy or cytological material for cell block and immunoperoxidase studies. Immunohistochemical demonstration of neuroendocrine differentiation using immunostaining for synaptophysin, chromogranin and CD56 reinforces a diagnosis of small cell carcinoma; however, some studies find up to 25% of small cell carcinomas are negative with all neuroendocrine markers.235 TTF-1 staining is usually seen in small cell carcinoma (>90% of cases) but is seldom seen in SCC. Strong positive nuclear p63 staining would favor a squamous lesion.213 A paranuclear CAM 5.2 keratin-positive dot similar to those seen in Merkel cell tumors and some other neuroendocrine tumors is a helpful criterion not seen in other poorly differentiated lung carcinomas. In ‘operable’ stage I tumors or peripheral tumors, very careful cytological assessment is necessary. Biopsy or lobectomy is advisable when diagnostic difficulty is not resolved by repeated FNB or FOB because some of these tumors will be large cell neuroendocrine carcinomas or atypical carcinoids, best treated by surgery (see Fig. 8.36).34
Low-grade carcinomas, e.g. carcinoid or adenoid cystic carcinoma, are also composed of small cells. The regularity and lack of fragility of the former should prevent misdiagnosis. MIB-1 staining also distinguishes low-grade from high-grade neuroendocrine tumors. Lin et al.236 found that over 50% of nuclei stained in high-grade tumors, compared to less than 25% of nuclei in low-grade tumors. In metastatic adenoid cystic carcinoma in the lung, the appearances are usually easily recognized;237 however, poorly differentiated adenoid cystic carcinomas can have cytological appearances resembling small cell anaplastic carcinoma. Other lesions such as spindle cell carcinoid, atypical carcinoid and large cell neuroendocrine carcinoma should be considered when the morphology or location of the tumor or clinical features are unusual, for example in small peripheral lesions, polypoid intrabronchial lesions, or in non-smokers.238
The cytological distinction of small cell carcinoma from atypical carcinoid is based on prominent nuclear molding and pleomorphism, and abundant mitotic activity and necrosis, including single-cell necrosis, in small cell carcinoma.35 However, it is safe to heed Frierson’s advice that small, peripheral or stage I tumors thought to be small cell carcinoma on cytological or small biopsy histological examination should be further evaluated, because at least some of these would be classified as atypical carcinoids, large cell neuroendocrine carcinomas or ‘peripheral small cell carcinoma of lung resembling carcinoid tumor’.34
When only small amounts of material are aspirated, artifactual smearing is more common; in these cases the loss of cytoplasm and the disruption of nuclei occurring in other poorly differentiated carcinomas may mimic small cell carcinoma. It is sometimes very difficult to distinguish small cell carcinoma from lymphomas in smears, particularly those of follicular center cell origin with pronounced cell pleomorphism and nuclear irregularity. Cell dispersal together with a rim or a tail of intact cytoplasm in individual cells and a background of round, cytoplasmic fragments staining blue with MGG (lymphoid globules/lymphoglandular bodies) are helpful features in making a diagnosis of lymphoma. Dispersed cells of small cell carcinoma are usually bare nuclei. Some low-grade lymphomas can show pseudomolding due to clustering of the nuclei. Problems can be resolved by immunocytochemistry or flow cytometry.
We have seen one case of metastatic melanoma in which FNB material was virtually indistinguishable from small cell carcinoma. Metastatic neuroendocrine carcinomas from other sites, e.g Merkel cell carcinoma, and high-grade neuroendocrine carcinoma of bladder, cervix, prostate and GIT may also need to be considered.239
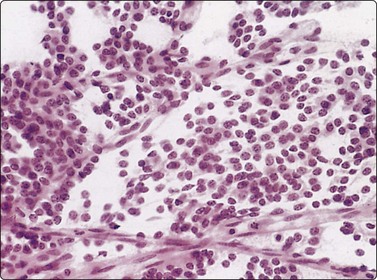
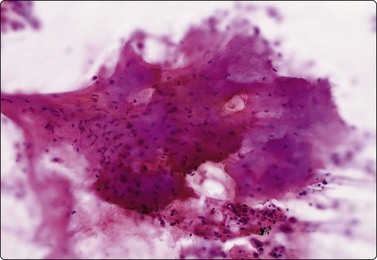
Large cell carcinoma (Figs 8.31 and 8.32)
Usual findings
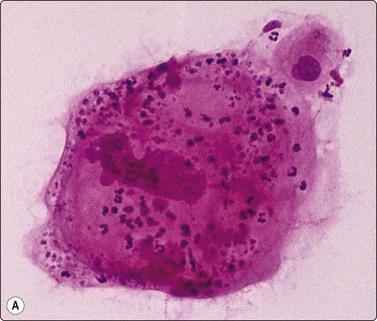
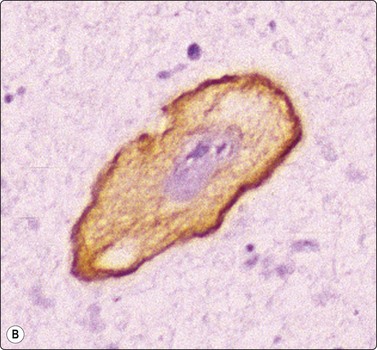
Fig. 8.32 Giant cell carcinoma
(A) Giant tumor cell with prominent neutrophil ingestion; (B) Positive immunostaining for AE1/AE3 keratins (A, H&E, HP; B, IPOX, HP).
Large cell carcinoma is a diagnosis of exclusion in that cytological features of squamous, glandular or neuroendocrine differentiation are absent in a non-small cell carcinoma.88 It may be necessary to examine large areas histologically to ensure that differentiation is lacking. This diagnosis can therefore never be established by FNAC alone. Instead, we use the category of poorly differentiated large cell or non-small cell carcinoma to designate non-small cell tumors in which further subtyping is not possible. Many of these prove to be squamous cell carcinoma or adenocarcinoma after histological examination, and the category is virtually eliminated if typing by electron microscopy is used. As previously discussed, attempts should be made to further classify ‘non-small cell’ carcinoma in cytological specimens using immunohistochemistry. Tumors showing dual differentiation such as adenosquamous carcinoma are uncommonly diagnosed cytologically.231 Carcinosarcoma (see Fig. 8.54),240 sarcomatoid/spindle cell carcinoma241 or blastoma242 are other considerations in poorly differentiated tumors with a spindle component.
The findings in large cell carcinoma are highly variable; those listed are the common features but have little specific diagnostic value, and other cytological patterns may occur. For example, in those tumors diagnosed as large cell carcinoma by subsequent histology, there is often a high N:C ratio and cell dispersal may be striking. An appearance resembling melanoma may sometimes be observed. Polymorph ingestion appears to be linked to an abundance of cytoplasm and cell pleomorphism rather than any particular form of differentiation, and virtually any tumor may demonstrate this phenomenon. FNAC findings in lymphoepithelioma-like carcinomas have been described.243
Pure giant cell carcinomas have been diagnosed cytologically (Figs. 8.31 and 8.32), although immunostaining is necessary to confirm carcinoma.244 They are said to be peripheral highly aggressive neoplasms, but occasionally, smaller resectable tumors are encountered.245
The cytological findings in poorly differentiated squamous cell carcinomas and adenocarcinomas overlap with those of large cell anaplastic carcinoma. In particular, highly eosinophilic cytoplasm simulating squamous differentiation may occur because of necrosis and is a common source of error in classification.
Metastatic neoplasms, especially other anaplastic carcinomas, melanoma, sarcoma and carcinosarcom,a may have similar cytological findings; large cell pulmonary neoplasms may have strikingly similar cytological features to melanoma.
Megakaryocytes, seen in FNB samples of lung either due to contamination from bone marrow, pulmonary capillaries,166 or extramedullary hematopoiesis,209 may give rise to suspicion of neoplasia.
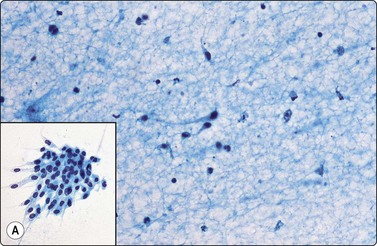
Carcinoid tumors (Figs 8.33-8.35)32-36,238,246,247
Typical carcinoid33,35,36,246,247
Usual findings
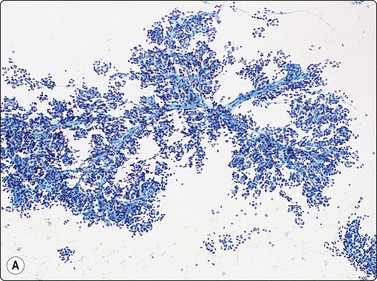
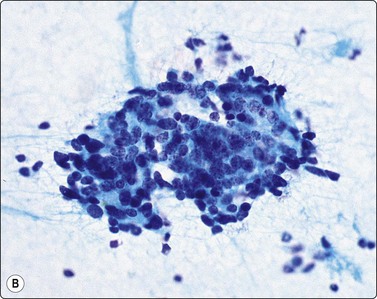
(A) Plexiform aggregate of small blood vessels with adherent tumor cells (Pap, LP); (B) Aggregate of small regular cells with stippled neuroendocrine nuclear chromatin pattern (Pap, HP).
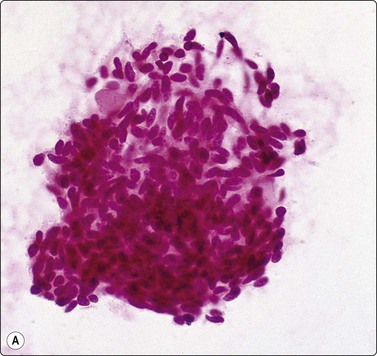
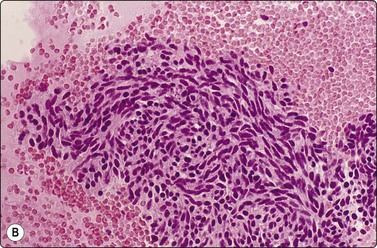
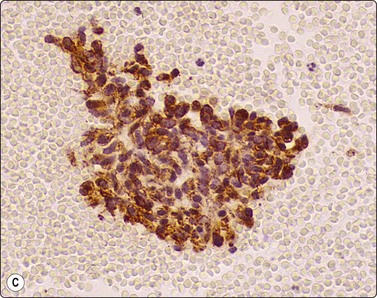
Fig. 8.35 Carcinoid tumor, spindle cell type
Tight aggregate of spindle cells with little pleomorphism. Cell block showing no mitotic activity or necrosis. Strong positive immunostaining for synaptophysin (A, H&E, HP; B, Cell block, H&E, HP; C, Cell block, IPOX, HP).
In ‘classic’ carcinoid tumors, the FNB findings are often distinctive enough to permit diagnosis, with or without ancillary tests such as immunocytochemistry. In contrast, the atypical carcinoids that we have seen were more difficult to classify before resection. Nicholson et al. found similar problems in recognizing a proportion of their neuroendocrine carcinomas, including low- and high-grade tumors, and suggested that ‘attention to the presence of loose cell aggregates in a background of singly dispersed cells; feathery patterns created by tumor cells clinging to capillaries; rosette formations; delicate, granular cytoplasm; inconspicuous nucleoli; molding in high-grade tumors; and, most importantly, speckled or dusty chromatin patterns are useful in identifying neuroendocrine differentiation in cytologic specimens’.36
In classic carcinoids, the ‘neuroendocrine’ round or oval nuclei with stippled nuclear chromatin and inconspicuous nucleoli are distinctive; the chromatin pattern may be rather similar to the ‘clock face’ chromatin of plasma cells (Fig 8.33). Although the cells are not particularly cohesive, small groups and loose clusters are quite common. Dispersed cells usually retain their cytoplasm and the nuclei are rather robust. Even if bare nuclei are prominent, traumatized cells, cell debris or streaks of nuclear material are not a feature.
Plexiform leashes of capillaries or venules or vascular cores with adherent tumor cells are a feature of bronchial carcinoids (Fig. 8.33) and are very seldom seen in small cell carcinomas.246 In Anderson’s series, this feature was present in 21 of 23 tumors including spindle and atypical forms.247 Collins et al. found this feature in only 4 of 19 cases but these were a mixture of pulmonary and metastatic intestinal tumors.33 In our material, most carcinoids of the classic and atypical type have shown this element when sampled by FNB, in contrast to bronchial brushings where the observation is seldom made.
The amount of cytoplasm varies and we have seen examples with abundant glassy or pale cytoplasm. In a few cases, we observed intracytoplasmic densities corresponding to the intermediate filament ‘buttons’ seen ultrastructurally and similar to those seen in Merkel cell tumors, salivary small cell carcinomas, some pulmonary small cell carcinomas and mediastinal carcinoids. Intracytoplasmic mucin is present in some cases.
Since atypical carcinoid is defined by mitotic activity or necrosis, even when focal, this diagnosis cannot be excluded in cytological material. This may need to be pointed out to clinicians in cytological reports.
Small cell anaplastic carcinoma is usually distinguishable by degree of pleomorphism, absence of cytoplasm, mitotic rate and cell fragility, unless material is minimal or poorly preserved. In small peripheral tumors or apparent stage I small cell tumors, a diagnosis of small cell carcinoma should be given cautiously because some of these tumors may be classified as atypical carcinoids by histopathology.34 Well-differentiated adenocarcinoma may be composed of very uniform cells; however, the cells are usually much larger, having more abundant cytoplasm and more cohesion than those of carcinoids. Large monolayered sheets are not usually a feature of carcinoids. Some carcinoids of ordinary type can simulate adenocarcinoma and show acinar formations, flat sheets or three-dimensional structures without characteristic neuroendocrine nuclear features.
The epithelial cells of hamartomas may bear some resemblance to carcinoid cells; other small cell lesions such as adenoid cystic carcinoma should also be considered.247 Bronchiolar cells and plasma cells may resemble neuroendocrine cells.33,35 Rare tumors such as sclerosing hemangioma resemble carcinoid in smears.220,223,247,248,249
Carcinoid variants include those with amyloid stroma, osseous metaplasia, melanin production, psammoma bodies33 and a papillary structure.
Metastatic carcinoid tumors of, for example, small bowel have similar features to primary tumors of the lung,33 although, to our knowledge, a plexiform vascular pattern has not yet been detailed in metastases. None of our primary lung tumors has shown the striking red granularity on MGG preparations seen in some small bowel tumors. Immunohistochemistry can aid in the distinction of primary versus metastatic carcinoid; TTF-1 expression is said to be specific for a lung primary in typical and atypical carcinoids and TTF-1-positive carcinoids are predominantly found in a peripheral location.147,250
Spindle cell carcinoid35,238,251
Peripheral location of a spindle or small cell tumor raises this possibility. We have seen cytological material from several spindle cell carcinoid tumors in which uniformity of nuclear size was a feature which, together with absence of nuclear smearing and background debris, excluded small cell anaplastic carcinoma. Cell clumps were present to a varying degree (Fig. 8.35) and in some smears cell dispersal was more evident. Adherence to proliferative vascular cores led to an appearance much like a vascular tumor in one case. Ultrastructural examination can exclude other spindle cell lesions.239 One example of a pulmonary tumorlet associated with an opacity on imaging, and providing diagnostic difficulties, has been described.252 TTF-1 is usually positive in spindle cell carcinoid.250
Atypical carcinoid35,36
These tumors occupy a position between carcinoid tumors and small cell carcinoma in the spectrum of neuroendocrine carcinomas of lung, in terms of both morphology and biological behavior. In histological material they are defined in the WHO classification as tumors with a carcinoid growth pattern but showing either greater than two mitoses per 2 mm squared, or necrosis.217,253 They may be associated with pleomorphism/nuclear atypia but this is not a necessary feature for diagnosis. There are examples which are difficult to categorise even when all the tumor is available for histological study, and individual cases may contain mixtures of typical and atypical carcinoid or even foci indistinguishable from small cell carcinoma or large cell neuroendocrine carcinoma. Atypical carcinoid cannot therefore be excluded in FNB samples which suggest carcinoid. We have reviewed cytological material from 15 classic and 11 atypical carcinoids of lung. All the classic tumors were specifically diagnosed; all the atypical cases were recognized as malignant but specific diagnosis was difficult in all. On review, some showed features in common with classic carcinoids such as a combination of cohesive aggregates and dissociation, small to moderate amounts of cytoplasm, neuroendocrine nuclei, trabeculae, palisades and rosettes and plexiform vascularity. Features favoring atypical carcinoid included necrosis and mitotic activity in 5 of 11 cases and more marked pleomorphism in 10 of 11. The absence of widespread molding, smeared fragile nuclei, abundant single-cell necrosis or abundant mitoses and cohesive acinar rosette-like or sheet-like groupings with palisading help exclude small cell carcinoma. Low levels of staining for MIB-1 would also be helpful.236 Metastatic tumors such as breast carcinoma or prostatic carcinoma may cause differential diagnostic problems.
Adenocarcinoid35,254,255
Tumors with prominent glandular differentiation but showing ultrastructural neuroendocrine features are well described and may not be recognizable as being neuroendocrine cytologically.
Large cell neuroendocrine carcinoma (Fig. 8.36)36-38,255,256
Large cell neuroendocrine carcinoma is included as a subtype within the large cell carcinoma category in the WHO classification.217 This entity is a non-small cell carcinoma without squamous or glandular differentiation, with morphological features suggesting neuroendocrine differentiation, which can be confirmed by immunocytochemistry or EM.217,255,256 Histologically, cell nesting with peripheral palisading of nuclei, large cells with prominent nucleoli, a high mitotic rate, (>10/10 high-power fields) low nuclear:cytoplasmic ratio, an organoid, trabecular or palisading pattern and large areas of geographic necrosis are features aiding recognition. A ‘neuroendocrine’ uniformly stippled nuclear chromatin pattern may be present in some tumors. Criteria for recognition by FNB samples have been suggested.36-38 In our view there is potential for ‘misdiagnosis’ because of the overlap in appearances between this tumor, small cell carcinoma, atypical carcinoid and basaloid carcinoma. At least some of these carcinomas may have been previously classified as ‘intermediate’ small cell carcinoma or atypical carcinoids. Yang et al. identified three cytological patterns in a series of 20 cases including small cell-like and mixed small cell and large cell-like tumors. Close attention to nuclear size and prominence of nucleoli are the most important criteria for distinction from small cell carcinoma.37 In the series of 20 cases reviewed by Wiatrowska, initial diagnoses included a range of non-small cell type tumors. Peripheral palisading of flattened three-dimensional clusters, finely or coarsely granular chromatin and some nuclear molding or crush artifact were features more characteristic of this tumor type than other non-neuroendocrine tumors.38 TTF-1 positivity has been described in up to 40% of large cell neuroendocrine carcinoma and may be useful in distinguishing this tumor from basaloid squamous carcinoma.257
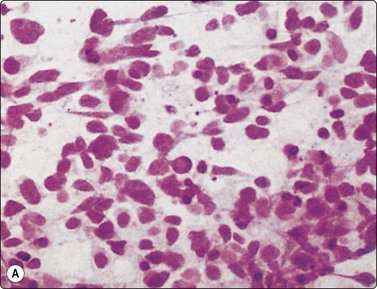
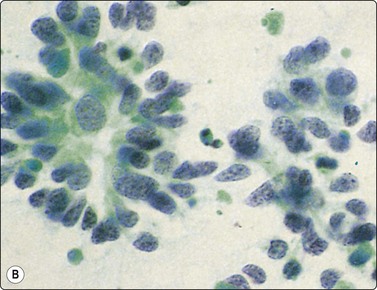
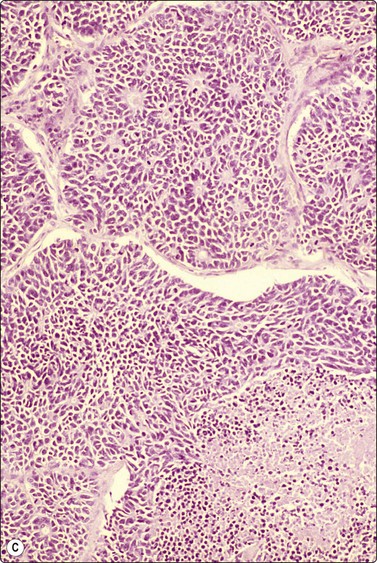
Fig. 8.36 Large cell neuroendocrine carcinoma
(A) Cell pattern resembling small cell carcinoma including single-cell necrosis, and pleomorphic fragile cells with minimal cytoplasm (H&E, HP); (B) Cohesive acinar and rosette-like structure (Pap, HP); (C) Sheets, rosette-like structures and foci of confluent necrosis (tissue section; H&E, IP).
Diffuse idiopathic neuroendocrine cell hyperplasia (DIPNECH)258,259
DIPNECH is a putative precursor lesion of carcinoid tumors of the lung.258,259 As this process may present with multiple nodules radiologically, there is the potential for interpretation as metastatic disease; cytological findings have not yet been reported in the literature.
Primary pulmonary salivary-type neoplasms
Primary pulmonary neoplasms that have similar histological features to those seen in salivary glands are rare. Careful clinical evaluation is necessary to establish the primary pulmonary nature of these tumors and to rule out the possibility of metastasis.260
Adenoid cystic carcinoma237,261-264
Distinction from other small cell malignancies is usually possible if large acellular basement membrane spheres are present (Fig. 8.37). Pitman et al. found diagnosing metastatic tumors to be easily achieved in FNB samples.237 Ozkara has described FNAC findings of primary pulmonary solid adenoid cystic carcinoma.261
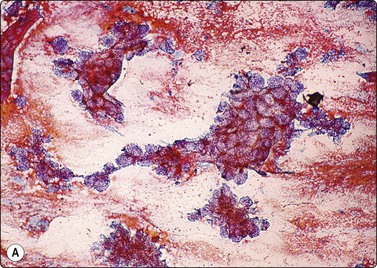
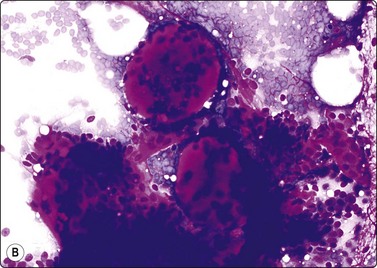
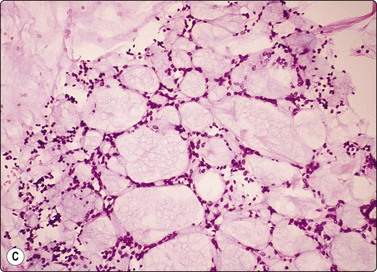
Fig. 8.37 Metastatic adenoid cystic carcinoma; primary oral cavity tumor
(A) Large aggregates of tumor cells with a cribriform appearance; (B) Prominent hyaline globules of basement membrane material (A, Pap, LP; B, MGG, HP). (C) Cell block; cribriform pattern around loose basement membrane material (C, Cell block, H&E, IP).
Mucoepidermoid carcinoma214,263,264
Nguyen263 describes several low-grade tumors with moderately pleomorphic squamous cells and occasional cytoplasmic mucin vacuoles. Brooks and Baandrup found a mixture of intermediate, squamous and mucin-secreting cells with some spindle cells.214 Segletes considered the findings to be suggestive in their three cases.264
This can present as a primary pulmonary lesion or as metastasis from a benign-appearing salivary pleomorphic adenoma; its appearances by FNAC in either setting are identical to those described in the salivary gland.
Bronchial acinic cell carcinoma267
Watanabe et al. have described transbronchial FNAC findings in a bronchial acinic cell carcinoma, which were essentially similar to acinic cell carcinomas of the head and neck region.267
Other bronchial gland tumors
Cwierczyk et al. described a bronchial oncocytoma.268 Oncocytomas, carcinoid tumors with oncocytic features and metastatic oncocytic or oxyphilic malignancies may have similar features. An oncocytic carcinoid mimicking granular cell tumor has been described in bronchial brushings.269 Pulmonary epithelial myoepithelial carcinoma is a rare tumor, usually endobronchial; cytological findings have not yet been described.270
Metastatic malignancy52
The cytopathologist’s approach to the diagnosis of metastases by FNAC is similar to that of the surgical pathologist. Detailed knowledge of the clinical history must be available, together with earlier cytological and histological preparations, for review and comparison with current material. Using these principles, the diagnosis is usually achieved with a high degree of reliability, although some caution is necessary because of the ability of lung carcinoma to mimic other tumors, including sarcomas. Even if the aspirated material cytologically resembles a previous malignancy at another site, a new primary tumor may still be difficult to exclude completely, especially in the case of adenocarcinoma. Perry57 and Freidman et al.53 drew attention to the relatively common occurrence of new primary tumors when metastases were suspected clinically. Metastatic carcinoma, particularly from colon and kidney, may show endobronchial growth mimicking primary tumor. Adenocarcinomas from various sites may also show a bronchiolo-alveolar growth pattern along alveolar septa. Specific immunohistochemical stains such as thyroid transcription factor (TTF-1), differential cytokeratins (high and low molecular weight cytokeratins and CK7 versus CK20), CEA, hormone receptors (ER and PR), mammoglobin and gross cystic disease protein (GCDFP-15) may be useful markers in differentiating primary from metastatic carcinoma. Other immunohistochemical stains which are fairly specific for certain malignancies may also be of value, e.g. prostate-specific antigen (PSA), thyroglobulin and S-100, HMB-45 and Melan A for malignant melanoma.
Metastatic colorectal adenocarcinoma (Figs 8.38-8.40)271
Usual findings
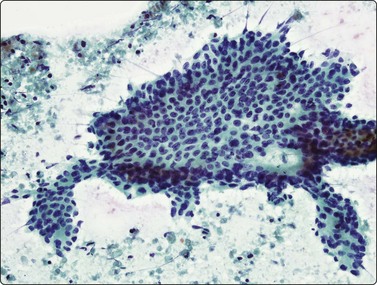
Fig. 8.38 Metastatic colonic carcinoma
Well-differentiated tumor with monolayered sheet, gland formation and a background of necrotic tumor (Pap, HP).
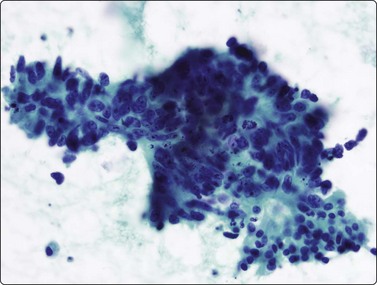
Fig. 8.39 Metastatic colonic carcinoma
Complex aggregate with palisading columnar cells, gland openings, mitoses and apoptotic debris (Pap, HP).
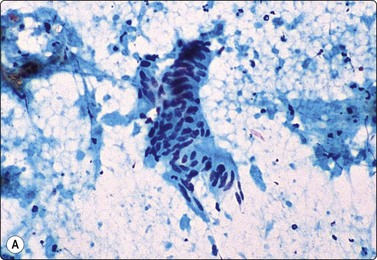
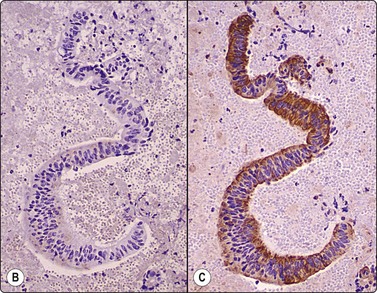
Fig. 8.40 Metastatic colonic carcinoma
(A) Strip of tumor tissue with elongated palisaded nuclei, in a background of ‘dirty’ necrosis; (B) Negative immunostaining for cytokeratin 7 and (C) Strong diffuse cytoplasmic staining for cytokeratin 20 (right) in cell-block preparations (A, Pap, MP; B & C, Cell block; IPOX, MP).
Metastatic adenocarcinoma of colorectum usually shows the above features. The ‘dirty’ necrosis is rather characteristic, with amorphous and granular eosinophilic material and a background of nuclear debris.
We have seen significant necrosis in only one case of well-differentiated, apparently primary carcinoma of the lung and the histological appearances in this case were strikingly similar to those of colonic carcinoma. Palisading of elongated nuclei of columnar cells is often a feature of colonic carcinoma, but not bronchogenic adenocarcinoma. In colonic carcinoma, glandular structures are often evident either within large aggregates (Figs 8.38-8.39) or lying separately; the demonstration of a clear apical space and a brush border in the tumor cells is another characteristic.271 A CK7/TTF-1 negative CK20/ CDX2-positive immunophenotype is generally confirmatory (Fig. 8.40). Some mucinous tumors of the lung may also possess a CK7 negative/CK20 positive phenotype although usually not with such intense or widespread CK20 staining.272,273 Pulmonary enteric adenocarcinoma can closely resemble colorectal carcinoma but is often CK7 and TTF-1 positive, in addition to being CK20 positive.142 EM demonstration of junctional complexes and cell surface features of hindgut type in colonic cancer is also helpful.47
Other metastatic tumors
Breast carcinoma exhibits variable features including a predominantly dispersed pattern, aggregates, sometimes including cell balls, and linear arrays, the latter mainly in metastatic lobular carcinoma. Positive hormone receptor immunocytochemistry is a valuable criterion (Fig. 8.41). It is worth noting that some clones of estrogen receptor protein can be positive in primary lung adenocarcinoma; SP1 clone has a significantly higher rate of positive estrogen receptor expression in pulmonary adenocarcinomas compared with either 1D5 or 6F11 clones. Caution is required in the use of SP1antibody alone in distinguishing metastatic breast carcinoma from primary pulmonary adenocarcinoma.141 Recently, cases of primary adenocarcinoma of lung with a TTF-1-negative GCDFP-15-positive phenotype have been described in small samples, further highlighting difficulties in this area.274
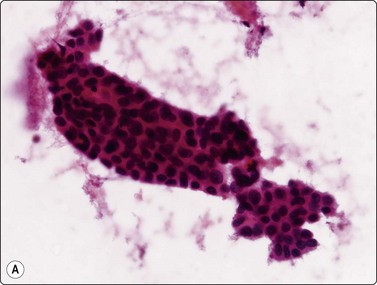
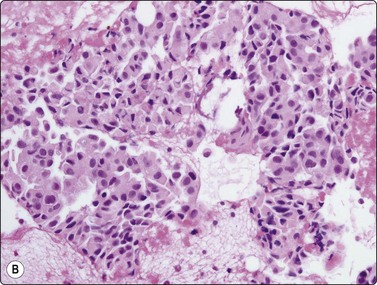
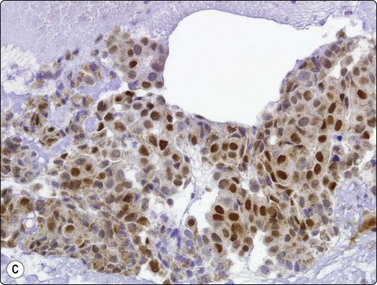
Fig. 8.41 Metastatic breast carcinoma
(A) Tubular aggregate of tumor cells (H&E, HP) (B) Cell block; microbiopsy (H&E, HP) (C) Positive nuclear staining for estrogen receptor protein (IPOX, HP).
Renal cell carcinoma presents with aggregates of cells with abundant granular or clear cytoplasm, rounded nuclei and macronucleoli.275,276 Cytoplasmic clearing may not be evident in smears and is better demonstrated in cell blocks. Immunohistochemistry for RCC antigen and PAX-2 may be of value.277 Adrenal metastases are essentially similar in appearance to renal cell carcinoma. Benign clear cell (’sugar’) tumors may also cause difficulties.278 Clear cell tumors provide a particular challenge but the site of origin can often be established using cytological features, immunohistochemistry and clinical information.279 Ancillary studies are valuable in differentiating these and in excluding hepatocellular carcinoma or large cell neuroendocrine carcinoma. Prostatic carcinoma may show rosette-like structures with a central cytoplasmic mass or monolayered sheets similar to BAC; positivity for PSA can be used to confirm the diagnosis. Transitional/urothelial carcinoma (TCC) possesses similar features to primary large cell carcinomas of lung and is difficult to identify specifically, although many cases will be CK7 and CK20 positive, in contrast to large cell carcinomas of lung or adenocarcinomas which are usually CK7 positive and CK20 negative. Johnson and Kini suggest that the most distinctive features were the presence of ‘spindled, pyramidal, and/or racquet-shaped malignant cells with eccentric nuclei and cytoplasmic features of both squamous and glandular differentiation including endoplasmic/ectoplasmic interfaces and intracytoplasmic vacuoles’, and some glandular features.280 ‘Cercariform’ cells, which are non-keratinized cells with a globular body and a cytoplasmic process with a bulbous or fishtail end, have also been suggested as a useful criterion, especially when present in large numbers, and without keratinization (Fig. 8.42).281,282 Unusual metastases such as choriocarcinoma,283 mesothelioma,284 meningioma,285 basal cell carcinoma of skin,286 acinic cell carcinoma of salivary gland,287 benign giant cell tumor of bone,288 benign metastasizing leiomyoma,289 adamantinoma,290 sacrococcygeal ependymoma291 and pleomorphic adenoma of salivary gland265,292 have been diagnosed by FNAC.
Melanoma (Fig. 8.43)291,292
An unequivocal diagnosis of metastatic melanoma by FNAC smears alone is difficult without pigment in tumor cells. In Perry’s series of 120 cases of melanomas, 60% were amelanotic in FNB samples.293 Large cell anaplastic carcinomas of lung may exhibit very similar cytological features to melanoma, as may other metastatic carcinomas and sarcomas.
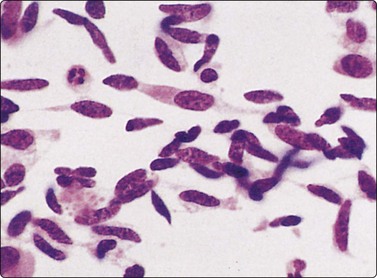
Spindle cell tumor including cells with cytoplasmic tails resembling a mesenchymal neoplasm (H&E, HP).
Melanoma cells shed their pigment, which is taken up by surrounding macrophages, often leaving few or no malignant cells containing pigment. Melanin within macrophages cannot be reliably distinguished from lipofuscin or hematoidin using conventional stains. MGG stains all brown pigments blue-black and Fontana-Masson staining is not as reliable in smears. Blaustein reports melanin pigmentation in a metastatic breast carcinoma to the lung.294 Some carcinoid tumors show intracytoplasmic melanin. S-100, HMB-45 or Melan A staining and negative staining for keratins are prerequisites to FNAC diagnosis of amelanotic melanoma. Spindle cell melanoma should be considered when any spindle cell malignancy is found and especially when there is little or no accompanying connective tissue stroma and when the cells are monotonous or easily dispersed. Some melanomas may closely mimic sarcomas (Fig. 8.43) and possess very fragile, elongated cytoplasmic tails. Other variants include small cell tumors which may resemble lymphoma, small cell carcinoma, or rhabdoid tumors.295 Metastatic melanoma may have a different immunohistochemical staining profile from the primary melanoma; most commonly, the metastasis will lose Melan A or HMB-45 but occasionally there is loss of S-100 expression.296 Although rare, it is worth noting that primary melanomas of lung are described and are thought to derive from bronchial melanocytes. FNAC findings are indistinguishable from metastatic melanoma.297
Spindle cell lesions of lung (see Chapter 15)48,49,298-320
A wide range of lesions may present as a predominant spindle cell pattern cytologically, including reactive processes, and benign and malignant neoplasms. Benign neural tumors may show alarming nuclear atypia and are a possible source of false-positive diagnoses of malignancy (see also Chapter 15). Although a few false diagnoses were recorded, in the experience of Hummel et al. about 85% of spindle cell lesions may be diagnosed specifically.298 Nevertheless, in individual cases, definitive diagnosis is often extremely difficult, for example in distinguishing between pleomorphic high-grade sarcoma, melanoma, spindle cell or sarcomatoid carcinoma and mesothelioma. Problems also exist at the low-grade end of the spectrum. Histological material, for example from core biopsies, may be useful; however, we have found immunohistochemistry on small samples, e.g. cell blocks and thin cores, to be unreliable in this setting. Cytogenetics, PCR or FISH studies to detect specific translocations, e.g. t (X;18) of synovial sarcoma, may be diagnostic, however, in the absence of such a specific finding or a known primary elsewhere, resection will usually be required for definitive diagnosis.
Several FNAC series of primary or metastatic sarcomas of lung or pleura have been documented48,49 and there are reports of metastatic malignant schwannoma,299,300 malignant fibrous histiocytoma,301-303 leiomyosarcoma,304 haemangiopericytoma,305 dermatofibrosarcoma protruberans,306 synovial sarcoma,307 including monophasic forms,308 metastatic endometrial stromal sarcoma,309 chondrosarcoma,310,311 osteosarcoma,312 epithelioid sarcoma,313 phyllodes tumor with osteogenesis,314 and alveolar soft part sarcoma,315 some presenting for the first time as metastases. Spindle-shaped cells with fragile cytoplasm, which are poorly cohesive and form rather flat cellular aggregates and display many single cells, nuclei with finely granular chromatin and small nucleoli, and bizarre multinucleated cells are all suggested as general indicators of mesenchymal malignancy.48,49 This contrasts with the three-dimensional cell aggregates and the coarsely granular nuclear chromatin of carcinoma. However, an awareness of the full spectrum of cytological appearances of sarcomas is necessary.
Inflammatory myofibroblastic tumor (Fig. 8.44)57,316-320
Inflammatory pseudotumor is a heterogeneous group of lesions occurring in various organs, and is histologically characterized by fibroblastic and myofibroblastic proliferation, with an inflammatory infiltrate. Inflammatory myofibroblastic tumor is a neoplastic counterpart which often occurs in the lung and which may show aberrant expression of ALK and its gene translocation.319 ALK positivity by immunohistochemistry has been described in approximately 50% of cases and ALK rearrangement can be detected; however, this has not been documented in FNAC samples. A range of biological behavior is described; most are cured by complete resection; however, recurrences do occur. Up to one-third of patients have fever, weight loss, anemia and elevated ESR. We have seen a few examples of this entity in FNB smears where the smears are often sparsely cellular and contain histiocytes, lymphocytes and plasma cells, sometimes with fibroblastic or ganglion-like cells identified (Fig. 8.44). Hosler et al.320 found diagnostic accuracy of cytology for IMT to be low (42%). In general, cytological findings are not specific. Histological examination of the whole lesion is necessary for specific diagnosis and to exclude a reaction to other benign or malignant processes.
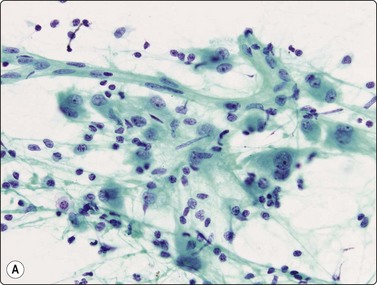
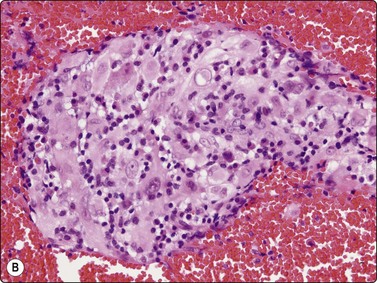
Fig. 8.44 Inflammatory myofibroblastic tumor/pseudotumor
(A) Loose collection of large mesenchymal cells with rounded nuclei, binucleation, prominent nucleoli and dense cytoplasm. Background of lymphocytes and plasma cells (Pap, HP). (B) Cell block aggregate of pleomorphic histiocyte-like cells with a background of lymphocytes and plasma cells (H&E, HP).
Pulmonary hamartoma (Figs 8.45-8.51)64,75,90-93,321
Pulmonary hamartomas represent approximately 8% of coin lesions64 and account for 77% of benign lung tumors.322 Cytogenetic studies support the view that hamartomas are clonal mesenchymal neoplasms.323 The peripheral location of this lesion and its rounded, well-demarcated border, calcification, or CT densitometric appearances may lead to a strong suspicion of the diagnosis and, if the aspiration needle is deflected by the lesion or bends when inserted into it, this impression is reinforced. Despite its firmness, a good sample of material is often obtained. Aspirated chondroid material may be difficult to express onto the slide. It has an opaque ‘glassy’ appearance, is firm to gelatinous in consistency and gives a gritty sensation on being smeared. The cytological features correspond to the components of the lesion found in histological sections.
Mature cartilage has a homogeneous waxy, purple-gray appearance in H&E-stained material (Fig. 8.45). It is sparsely cellular; chondrocytes may be seen in lacunae. In contrast, the immature cartilage or myxoid connective tissue has a fibrillar texture (Figs 8.46-8.48). It is pale-staining in H&E, magenta in MGG (Fig. 8.46) and poorly cellular, resembling mucus at first glance. There is, however, a component of spindle cells and the material is usually more cellular than mature cartilage (Fig. 8.49). This material is the most useful diagnostic indicator of hamartoma.91,93,321 S-100 positivity of the spindle cells has been suggested as an additional criterion.92,93 The bronchiolar epithelial sheets may show some differences from normal bronchiolar epithelium. There is more abundant pale cytoplasm with less obvious borders; multinucleation may be present. Nuclei are usually rounded, but may be quite large and variable in size (Fig. 8.50). Large intranuclear cytoplasmic inclusions may be evident (Fig. 8.51). Mature fat may or may not be evident. An associated lymphocytic and macrophage component may be present.
In some series, over 90% of hamartomas are recognized by FNAC64,91,92 and diagnosis by this method is now widely used to justify conservative management,64 or, in lesions which continue to grow, enucleation rather than lobectomy. Chondromas, which may be seen as part of Carney’s triad, are thought to be distinct from pulmonary hamartomas; they consist of well-differentiated cartilage but do not contain fat, spindled cells or entrapped pulmonary epithelium.
It is possible to aspirate material from the costochondral junctions, especially if a stylet or guide needle is not used and, if found in an aspirate with fat, a diagnosis of hamartoma may be incorrectly assumed.91
In one of our cases, the variation in nuclear size and large intranuclear inclusions were sufficient to lead to a false-positive diagnosis of carcinoma; this should not occur if the other components of the lesion are recognized (Figs 8.50 and 8.51), but this is a common source of error in the literature.75,321
It may be difficult to discern the myxoid material or cartilage in smears, especially in Pap stains, unless one is familiar with its appearances. The material resembles the myxoid stroma of other entities, for example fibroadenoma of the breast. It is conceivable that other lesions, such as mixed salivary tumors of the bronchial wall, myxoid fibrous neoplasms or amyloidosis,206 might produce similar cytological pictures but all are extremely rare. The combination of appearances listed above is diagnostic for practical purposes when combined with radiological correlation to support the diagnosis. A 2005 College of American Pathologists Interlaboratory Comparison Program321 examining FNB diagnosis of pulmonary hamartoma found the specificity of the interpretation of a benign process was 78%. The false-positive rate was 22%, with the most common false-positive diagnoses being carcinoid tumor, adenocarcinoma, and small cell carcinoma. The overall accuracy for making the correct specific reference diagnosis of pulmonary hamartoma was 26%. The high false-positive rate and the low overall interpretative accuracy was attributed to the failure to recognize the mesenchymal component, which was often subtle on the Papanicolaou-stained slides, and failure to recognize the benign nature of the often very cellular epithelial component. The dominance of the epithelial component was thought to have resulted in incorrectly classifying the lesions as low-grade neoplasms, most often carcinoid tumor and adenocarcinoma.
Sclerosing hemangioma/pneumocytoma220-223,248,249,324-326
These lesions are rare benign tumors occurring mainly in middle-aged adults, but are also described in young persons including children. There is a female to male ratio of 5 : 1. They usually present as incidental rounded masses on chest X-ray. The tumors are composed of epithelial elements and stromal-like cells, both of which show positive staining for TTF-1 suggesting a primitive bronchiolar epithelial cell type.326 The epithelial and stromal cells may or may not show positive staining for keratin and EMA. Sclerosing ‘pneumocytoma’ is the currently favored terminology.217 FNAC descriptions include aggregates of epithelial-like cells surrounding a proliferating network of blood vessels or spaces and a stromal-like component,221,248 and cases with a prominent spindle cell component,222 or a papillary pattern in cell block preparations.221 The epithelial component, when abundant, may lead to an appearance similar to BAC, carcinoid or other tumors. Few cases have been prospectively diagnosed by FNAC. Gal et al.323 were able to diagnose a case by identifying a two-cell population, both of which were TTF-1 positive. The patient was followed for over 7 years before surgery was necessary due to a growing tumor. Some authors describe frequent progesterone receptor and occasional estrogen receptor positivity in these tumors; however, other studies suggest that hormone receptor positivity is only rarely seen.325,326
Figure 8.52 shows our only case. Recognising stromal elements has been suggested as a criterion for avoiding a false-positive diagnosis of BAC. However, we have seen well-differentiated adenocarcinoma with stromal elements resembling those described in sclerosing pneumocytoma. We consider that the clinical and imaging findings, including marked circumscription of the lesion, are likely to provide the best initial clue to a benign diagnosis.
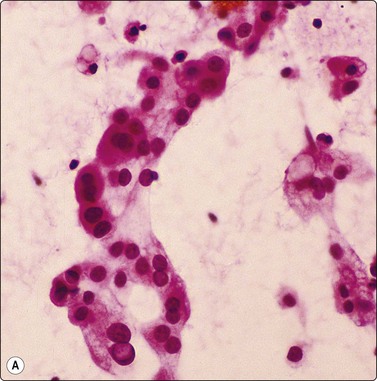
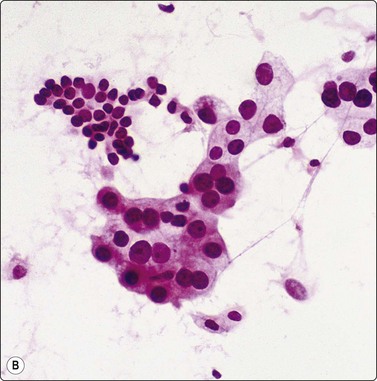
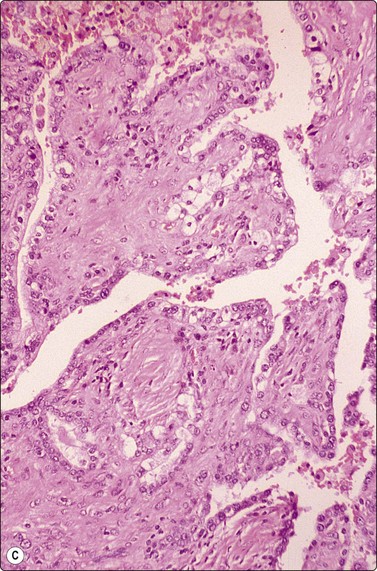
Fig. 8.52 Sclerosing haemangioma/pneumocytoma
The patient was a young Asian male with a 10-cm rounded intrapulmonary mass. (A,B) Aggregates of moderately pleomorphic glandular epithelial cells including some with prominent intranuclear cytoplasmic inclusions. Note contrast with non-neoplastic bronchiolar cells in B; (C) Tissue section showing papillary tumor edge with collagenous stromal cores lined by pleomorphic bronchiolar epithelial cells (A,B, H&E, HP; C, tissue section, MP).
Lymphoid lesions in lung and pleura (Fig. 8.53)55,56,302-305
Non-Hodgkin’s lymphomas are uncommon primary lesions and may occur de novo, or in a background of AIDS or pre-existing autoimmune disorders such as Sjögren’s syndrome. Primary Hodgkin’s lymphoma is rarely seen, but the lung is a common site for relapse. Leukemic infiltration of minor degree is commonly seen in the lung at post-mortem, but clinically significant involvement is rare.
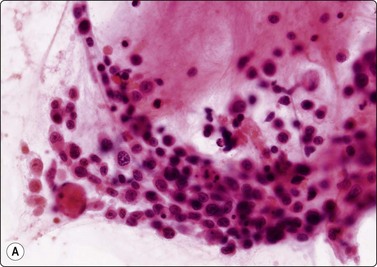
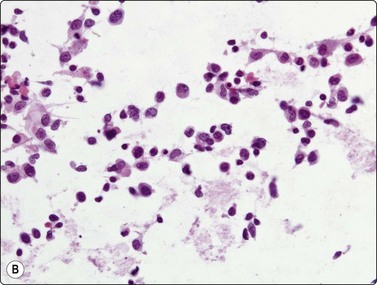
Fig 8.53 High-grade non-Hodgkin’s lymphoma
(A) Large B-cell lymphoma; (B) NK/T lymphoma (H&E, HP). In general, the diagnosis of even recurrent lymphomas in the lung will require a morphological comparison with the previous tumor and consistent immunophenotyping.
FNAC is valuable in diagnosing focal lung lesions in patients with hematological malignancies. Wong et al., in a series of 67 patients, were able to confirm metastatic lymphoma or leukemia, another lung malignancy or infection, in most samples examined.327 Confirming spread of lymphoma which has been previously diagnosed may be relatively simple. Recurrent large cell or high-grade lymphomas can usually be diagnosed by FNB, but low-grade lymphomas present more problems. Flow cytometry is an indispensable adjunct (see Chapter 5).136-138 In Bonfiglio’s series, 8 of 10 cases were diagnosed and in two the findings were used as a basis for definitive therapy.50 In Flint’s series of 13 cases of previously diagnosed Hodgkin’s disease affecting the lung secondarily, all were diagnosed by FNB.51 Gattuso et al. found FNAC to be highly specific in diagnosing post-transplant lymphoproliferative disorders.328 Figure 8.53 shows two examples of NHL seen in our laboratory. The case illustrated in 8.53A was a 17-year-old boy who presented with a 3-month history of viral-like illness, serologically proven EBV infection and multiple hepatic and pulmonary nodules. He also had an orbital lesion with perforation of one eye. The first diagnostic material was from a CT-guided FNB of a lung nodule. The cytological diagnosis was non-Hodgkin’s lymphoma of large cell type, excluding infection as a cause. Flow cytometry did not show a clonal population of B cells. A recommendation was made to obtain histological material, and a biopsy of the right eye sclera and orbital tissue gave a diagnosis of EBV-associated angiocentric large B-cell lymphoma. The patient died soon after and autopsy revealed widespread lymphoma involving lung, liver, spleen and bone marrow. The case in Fig. 8.53B was a 55-year-old male with a history of NK/T cell lymphoma involving lung, diagnosed on wedge biopsy several years previously. CT-FNA was performed of a recurrent tumor mass in lung. The smears showed a pleomorphic population of dispersed tumor cells consistent with recurrent lymphoma.
In small cell carcinoma the neoplastic cells may be dispersed and may resemble a high-grade lymphoma.
Occasional cases of lymphoma demonstrate clustering and even molding of nuclei, resembling small cell carcinoma.
The distinction between lymphoid interstitial pneumonia, reactive lymphoid hyperplasia and low-grade non-Hodgkin’s lymphoma may be difficult even in histological sections. Cytological findings are unlikely to be diagnostic because these lesions are all composed of small ‘mature’ cells. Sending material for flow cytometry or for molecular studies to detect clonal rearrangements of immunoglobulin or T-receptor genes may be helpful.
Other rare primary malignancies
Pulmonary blastoma242,329
These rare malignant tumors present most commonly in young adult males as peripheral lesions. They resemble fetal lung and show varying mixtures of epithelial tubules with glycogen-rich columnar cells, stromal elements composed of small compact tumor cells and cartilage or smooth or skeletal muscle. FNAC diagnosis is reported, although differentiation from carcinoma or other malignancies is difficult. A related pure epithelial tumor called fetal adenocarcinoma has been described.330 So-called pleuropulmonary blastoma331 is a different neoplasm seen in childhood, which lacks an epithelial element, is commonly associated with cystic malformations and has the potential for transformation to multiple sarcomatous types.
Pulmonary carcinosarcoma240
These lesions are rare tumors of later decades of life, sometimes presenting as pedunculated lesions of bronchi and composed of poorly differentiated epithelial and mesenchymal tissue (Fig. 8.54). There is debate over whether they represent sarcomatoid carcinomas or a distinct entity.
Chest wall and pleura
Malignant mesothelioma (Fig. 8.55)39-41,43,44,335-338
Usual findings
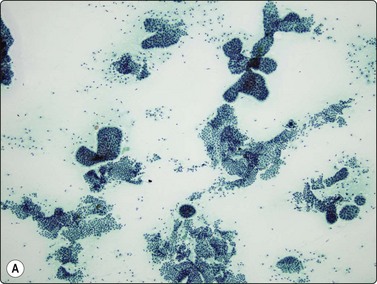
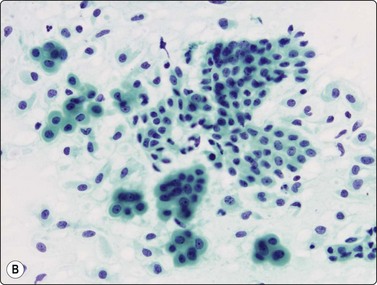
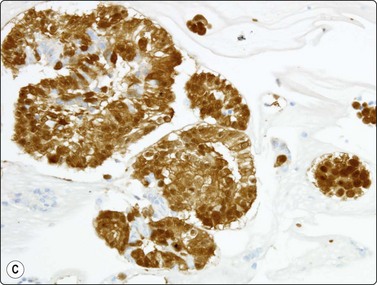
Fig. 8.55 Malignant mesothelioma
(A) Highly cellular smear with tubulo-papillary structures, monolayered sheets and some rounded 3-D aggregates (Pap, LP); (B) Sheets and 3-D aggregates with dense cytoplasm (Pap, HP); (C) Cell block; strong nuclear and cytoplasmic staining for calretinin (IPOX, HP).
When the clinical findings are consistent and there is a history of asbestos exposure, cytology can offer a clear-cut diagnosis of malignancy and supportive evidence of tumor type. FNAC diagnosis is accepted as a basis for diagnosis and management; material for cell block preparations, immunohistochemistry or ultrastructural studies is necessary for definitive diagnosis.41,336-338 Membrane-associated EMA staining of epithelial-like cells which also show strong positive nuclear and cytoplasmic staining for calretinin offers strong support of the diagnosis in the correct clinical context. We have now seen FNB material from over 100 cases and the technique is useful for lesions presenting without effusions (approximately 10–15% of pleural cases), in unusual sites such as mediastinum or in metastases.336 Nevertheless, we try to obtain core biopsy material in all cases where the diagnosis is suspected since this provides a better idea of architecture, and includes spindle cell/mesenchymal elements which are under-represented in smears. In contrast to effusions in which the tumors are mainly epithelial, the full range of growth patterns of malignant mesothelioma is seen in FNAC. The most specific cytological pattern is a combination of sheets of cells, cell groups, dispersed polygonal cells with dense cytoplasm, spindled epithelial cells and spindled fibroblastic cells. Hyaluronic acid vacuoles are commonly seen in FNB specimens; this material has a rather characteristic crumpled membrane-like appearance and stains violet in Pap and magenta in Romanowsky stains.
A distinction from well-differentiated adenocarcinoma may be difficult, particularly in peripheral lung lesions with a bronchiolo-alveolar growth pattern. Histochemistry and immunohistochemistry are essential for diagnosis. Positive nuclear staining for calretinin and cytoplasmic CK5/6 is a helpful indicator of mesothelioma in the correct context (Fig. 8.55). The presence of epithelial mucin virtually excludes mesothelioma. Positive staining for glandular markers such as carcinoembryonic antigen (CEA), B72.3, CD15, BerEp4 or TTF-1 also helps to exclude mesothelioma although aberrant staining can be seen in some cases. Vacuoles containing hyaluronic acid display magenta-staining material with MGG, resembling mucin-secreting adenocarcinoma. The prominent finger-like microvilli at the surface of mesothelioma cells, without a glycocalyceal coat, are the most distinctive ultrastructural feature of mesothelioma.
The wide variation in growth patterns and degree of differentiation in mesothelioma is well known and, especially in more poorly differentiated and sarcomatous forms, differentiation from anaplastic epithelial or mesenchymal neoplasms may be impossible with FNAC, even with recourse to electron microscopy and immunostaining. Primary pleural sarcomas are particularly difficult to distinguish from sarcomatous mesothelioma. Similar problems may be encountered in biopsy samples. Correlation with clinical and radiological findings is essential.
Small sheets of reactive mesothelial cells may show some variation in nuclear size and irregularity of nuclear outlines, but differential diagnosis is usually not a problem if one adheres to the rule of making diagnoses of malignancy only when abundant material is present. Strong positive staining for epithelial membrane antigen (EMA) has featured in most malignant mesotheliomas in cell blocks of effusions and pleural biopsy and we have not seen strong widespread mesothelial decoration in reactive conditions. This finding is an extremely useful adjunct in distinguishing between reactive change and malignant mesothelioma.338,339
Solitary fibrous tumors (benign; malignant; pleural fibroma) (Fig. 8.56)340-345
We have aspirated several solitary fibrous tumors including two histologically malignant metastasizing tumors. Smears were cellular in all cases, including the benign lesions. Many spindle-shaped fibroblastic cells were present; nuclei were moderately pleomorphic and some chromatin clumping was seen, leading to a suspicion of low-grade malignancy in a benign case. Fibers of collagen with adherent spindle cells were included in several cases. Other epithelial-like cells may have been bronchiolar epithelium entrapped within the growing edge of the lesion. In one of the histologically malignant lesions, only material from more benign-appearing areas was aspirated. We recognized only a few of these cases, but believe the combination of a localized, rounded pleural mass yielding dispersed small fibroblastic cells with dark spindled nuclei and background collagen should allow the lesion to be recognized and differentiated from mesothelioma. Cell blocks or core biopsies showing CD34, CD99 and bcl-2 positivity and negative staining for keratins, CEA, EMA and other epithelial markers will also be of value;344,345 however, resection for histological confirmation is required. Bishop et al.346 found none of 13 cases of malignant SFT were accurately diagnosed by FNAC, and only six cases were called malignant or suspicious of malignancy. Even in histologically malignant forms, the biological behavior is much less aggressive than malignant tumors of mesothelial origin, and surgical management may be curative; a distinction from mesothelioma or mesenchymal tumors of other types, such as synovial sarcoma or neural tumors, is important. Occasional intrapulmonary tumors are seen.
Peripheral primitive neuroectodermal tumor/Ewing’s sarcoma (PNET, Askin tumor)42,347-349
Peripheral primitive neuroectodermal tumors (PNET) occur mainly in older children and adolescents. They usually involve soft tissue but can extend into pleura and lung. These tumors are further described in Chapters 16 and 17.
Pleural thymoma350
Ectopic pleural thymoma can present as solitary or multiple pleural nodules mimicking metastatic disease or mesothelioma; pleural disease may be seen either in association with a thymic mass or in the absence of a thymic mass, where it presumably represents ectopic thymic tissue. The diagnosis can be established by FNB.350 This is a rare entity but is important to include in the differential diagnosis of an epithelial pleural-based mass, as management and prognosis are very different from mesothelioma or pleural metastasis. We have seen one aspirate from a pleural-based mass which was eventually diagnosed as thymoma by excision; the presence of a mediastinal mass was not appreciated at the time of FNB.
Lesions of rib and soft tissue of chest wall (see Chapters 15 and 16)351
Lesions such as multiple myeloma/plasmacytoma, metastases and occasionally primary sarcomas of bone or soft tissues may present as ‘thoracic’ tumors and be considered as primary lung lesions clinically. Myeloma can be extremely pleomorphic and mimic epithelial malignancy. Metastases are usually from breast, lung, melanoma or head and neck mucosal tumors.163 Goel et al. found a very wide range of inflammatory and neoplastic lesions in their series.351 At least in our experience, granular cell tumor should be kept in mind in the differential diagnosis of this site. We have seen three examples of subcutaneous or deep chest wall lesions, including one malignant tumor with metastases at presentation.
1 Menetrier P. Cancer primitif du poumon. Bull Soc Anat. 1886;61:643-647.
2 Dahlgren SE. Aspiration biopsy of intrathoracic tumours. Acta Pathol Microbiol Scand. 1967;70:566-576.
3 Levine MS, Weiss JM, Harrell JH, et al. Transthoracic needle aspiration biopsy following negative fiberoptic bronchoscopy in solitary pulmonary nodules. Chest. 1988;93:1152-1155.
4 Caya JG, Gilles L, Tieu TM, et al. Lung cancer treated on the basis of cytologic findings: an analysis of 112 patients. Diagn Cytopathol. 1990;6:313-316.
5 Fraire AE, McLarty JW, Greenberg SD. Changing utilization of cytopathology versus histopathology in the diagnosis of lung cancer. Diagn Cytopathol. 1991;7:359-362.
6 Blumenfeld W, Singer M, Glanz S, et al. Fine needle aspiration as the initial modality in malignant lung disease. Diagn Cytopathol. 1996;14:268-272.
7 Steffee CH, Segletes LA, Geisinger KR. Changing cytologic and histologic utilisation patterns in the diagnosis of 515 primary lung malignancies. Cancer. 1997;81:105-115.
8 Sider L, Davis TMJr. Hilar masses: evaluation with CT-guided biopsy after negative bronchoscopic examination. Radiology. 1987;164:107-109.
9 Knudsen DU, Nielsen SM, Hariri J, et al. Ultrasonographically guided fine needle aspiration biopsy of intrathoracic tumors. Acta Radiol. 1996;37:327-331.
10 Fish GD, Stanley JH, Miller KS, et al. Post-biopsy pneumothorax: estimating the risk by chest radiography and pulmonary function tests. Am J Roentgenol. 1988;150:71-74.
11 Miller KS, Fish GB, Stanley JH, et al. Prediction of pneumothorax rate in percutaneous needle aspiration of the lung. Chest. 1988;93:742-745.
12 Poe RH, Kallay MC. Transthoracic needle biopsy of lung in nonhospitalised patients. Chest. 1987;92:676-678.
13 Bhat N, Bhagat P, Pearlman E, et al. Transbronchial needle aspiration biopsy in the diagnosis of pulmonary neoplasms. Diagn Cytopathol. 1990;6:14-17.
14 DeCastro FR, Rey A, Caminero J, et al. Transbronchial fine needle aspiration in clinical practice. Cytopathol. 1995;6:22-29.
15 Roche DH, Wilsher ML, Gurley AM. Transtracheal needle aspiration. Diagn Cytopathol. 1995;12:106-112.
16 Rodriguez de Castro F, Diaz-Lopez F, Serda GJ, et al. Relevance of training in transbronchial fine needle aspiration technique. Chest. 1997;111:103-105.
17 Wang KP, Marsh BR, Summer WR, et al. Transbronchial needle aspiration for diagnosis of lung cancer. Chest. 1981;80:48-50.
18 Wang KP. Flexible transbronchial needle aspiration biopsy for histologic specimens. Chest. 1985;88:860-863.
19 Xie HB, Cornwell R, Grossman JE, et al. Bronchoscopy-guided transtracheal and transbronchial fine-needle aspiration biopsy: a 5-year institutional review of 111 cases. Diagn Cytopathol. 2002;27:276-281.
20 Givens CDJr, Marini JJ. Transbronchial needle aspiration of a bronchial carcinoid tumour. Chest. 1985;88:152-153.
21 Schenk DA, Bower JH, Bryan CL, et al. Transbronchial needle aspiration staging of bronchogenic carcinoma. Am Rev Respir Dis. 1986;134:146-148.
22 Lorch DGJr, John JFJr, Tomlinson JR, et al. Protected transbronchial needle aspiration and protected specimen brush in the diagnosis of pneumonia. Am J Respir Dis. 1987;136:565-569.
23 Schwartz AR, Fishman EK, Wang KP. Diagnosis and treatment of bronchogenic cyst using transbronchial needle aspiration. Thorax. 1986;41:326-327.
24 Sun W, Song K, Zervos M, et al. The diagnostic value of endobronchial ultrasound-guided needle biopsy in lung cancer and mediastinal adenopathy. Diagn Cytopathol. 2010;38(5):337-342.
25 Johnsrude IS, Silverman JF, Weaver MD, et al. Rapid cytology to decrease pneumothorax incidence after percutaneous biopsy. AJR. 1985;144:793-794.
26 Austin JH, Cohen MB. Value of having a cytopathologist present during percutaneous fine needle aspiration biopsy of lung: report of 55 cancer patients and metaanalysis of the literature. Am J Roentgenol. 1993;160:175-177.
27 End A, Helbich T, Wisser W, et al. The pulmonary nodule after transplantation. Chest. 1995;107:1317-1322.
28 Falguera M, Nogues A, Ruiz-Gonzalez A, et al. Transthoracic needle aspiration in the study of pulmonary infections in patients with HIV. Chest. 1994;106:697-702.
29 Johnston WW. Percutaneous fine needle aspiration biopsy of the lung. A study of 1,015 patients. Acta Cytol. 1984;28:218-224.
30 Mitchell ML, King DE, Bonfiglio TA, et al. Pulmonary fine needle aspiration cytopathology. A five year correlation study. Acta Cytol. 1984;28:72-76.
31 Pilotti S, Rilke F, Gribaudi G, et al. Transthoracic fine needle aspiration biopsy in pulmonary lesions. Updated results. Acta Cytol. 1984;28:225-232.
32 Nguyen GK, Gray JA, Wong EY, et al. Cytodiagnosis of bronchogenic carcinoma and neuroendocrine tumor of the lung by transthoracic fine-needle aspiration. Diagn Cytopathol. 2000;23:431-434.
33 Collins BT, Cramer HM. Fine needle aspiration cytology of carcinoid tumors. Acta Cytol. 1996;40:695-707.
34 Frierson HF, Covell JL, Mills SE. Fine needle aspiration cytology of atypical carcinoid of the lung. Acta Cytol. 1987;31:471-475.
35 Szyfelbein WM, Ross JS. Carcinoids, atypical carcinoids, and small-cell carcinomas of the lung: differential diagnosis of fine needle aspiration biopsy specimens. Diagn Cytopathol. 1988;4:1-8.
36 Nicholson SA, Ryan MR. A review of cytologic findings in neuroendocrine carcinomas including carcinoid tumors with histologic correlation. Cancer. 2000;90:148-161.
37 Yang YJ, Steele CT, Ou XL, et al. Diagnosis of high-grade pulmonary neuroendocrine carcinoma by fine-needle aspiration biopsy: nonsmall-cell or small-cell type? Diagn Cytopathol. 2001;25:292-300.
38 Wiatrowska BA, Krol J, Zakowski MF. Large-cell neuroendocrine carcinoma of the lung: proposed criteria for cytologic diagnosis. Diagn Cytopathol. 2001;24:58-64.
39 Sterrett GF, Whitaker D, Shikin KB, et al. Fine needle aspiration cytology of malignant mesothelioma. Acta Cytol. 1987;31:185-193.
40 Kwee W, Utama I. Malignant pleural mesothelioma and thoracic needle biopsy. Chest. 1988;93:1115-1116.
41 Tao LC. Aspiration biopsy cytology of mesothelioma. Diagn Cytopathol. 1989;5:14-21.
42 Kumar PV. Fine needle aspiration cytologic findings in malignant small cell tumor of the thoracopulmonary region (Askin tumor). Acta Cytol. 1994;38:702-706.
43 Nguyen GK, Akin MR, Villanueva RR, et al. Cytopathology of malignant mesothelioma of the pleura in fine-needle aspiration biopsy. Diagn Cytopathol. 1999;21:253-259.
44 Nguyen GK. Cytopathology of pleural mesotheliomas. Am J Clin Pathol. 2000;114(Suppl):S68-S81.
45 O’Reilly PE, Brueckner J, Silverman JF. Value of ancillary studies in fine needle aspiration cytology of the lung. Acta Cytol. 1994;38:144-150.
46 Saleh H, Masood S. Value of ancillary studies in fine needle aspiration biopsy. Diagn Cytopathol. 1995;13:310-315.
47 Davies DC, Russell AJ, Tayar R, et al. Transmission electron microscopy of percutaneous fine needle aspirates from lung: a study of 70 cases. Thorax. 1987;42:296-301.
48 Crosby JH, Hoeg K, Hager B. Transthoracic fine needle aspiration of primary and metastatic sarcomas. Diagn Cytopathol. 1985;1:221-227.
49 Kim K, Naylor B, Han IH. Fine needle aspiration cytology of sarcomas metastatic to the lung. Acta Cytol. 1986;30:688-694.
50 Bonfiglio TA, Dvoretsky PM, Piscioli F, et al. Fine needle aspiration biopsy in the evaluation of lymphoreticular tumors of the thorax. Acta Cytol. 1985;29:548-553.
51 Flint A, Kumar NB, Naylor B. Pulmonary Hodgkin’s disease. Diagnosis by fine needle aspiration. Acta Cytol. 1988;32:221-225.
52 Perry MD, Floyd PB, Johnston WW. Role of fine needle aspiration cytology in medical decision making for metastatic carcinoma of the lungs. Acta Cytol. 1984;28:624.
53 Friedman M, Shimaoka K, Fox S, et al. Second malignant tumors detected by needle aspiration cytology. Cancer. 1983;52:699-706.
54 Naryshkin S, Young NA. Respiratory cytology: a review of non-neoplastic mimics of malignancy. Diagn Cytopathol. 1993;9:89-97.
55 Cagle PT, Kovach M, Ramzy I. Causes of false results in transthoracic fine needle lung aspirates. Acta Cytol. 1993;37:16-20.
56 Silverman JF. Inflammatory and neoplastic processes of the lung: differential diagnosis and pitfalls in FNA diagnosis. Diagn Cytopathol. 1995;13:448-462.
57 Yi E, Aubry MC. Pulmonary pseudoneoplasms. Arch Pathol Lab Med. 2010;134(3):417-426.
58 Shih JY, Gow CH, Yu CJ, et al. Epidermal growth factor receptor mutations in needle biopsy/aspiration samples predict response to gefitinib therapy and survival of patients with advanced nonsmall cell lung cancer. Int J Cancer. 2006;118(4):963-969.
59 Sun W, Song K, Zervos M, et al. The diagnostic value of endobronchial ultrasound-guided needle biopsy in lung cancer and mediastinal adenopathy. Diagn Cytopathol. 2010;38(5):337-342.
60 Ernst A, Eberhardt R, Krasnik M, et al. Efficacy of endobronchial ultrasound-guided transbronchial needle aspiration of hilar lymph nodes for diagnosing and staging cancer. J Thorac Oncol. 2009;4(8):947-950.
61 Ernst A, Anantham D, Eberhardt R, et al. Diagnosis of mediastinal adenopathy-real-time endobronchial ultrasound guided needle aspiration versus mediastinoscopy. J Thorac Oncol. 2008;3(6):577-582.
62 Garwood S, Judson MA, Silvestri G, et al. Endobronchial ultrasound for the diagnosis of pulmonary sarcoidosis. Chest. 2007;132(4):1298-1304.
63 Kennedy MP, Jimenez CA, Bruzzi JF, et al. Endobronchial ultrasound-guided transbronchial needle aspiration in the diagnosis of lymphoma. Thorax. 2008;63(4):360-365.
64 de Rooij PD, Meijer S, Calame J, et al. Solitary hamartoma of the lung: is thoracotomy still mandatory? Neth J Surg. 1988;40:145-148.
65 Veale D, Gilmartin JJ, Sumerling MD, et al. Prospective evaluation of fine needle aspiration in the diagnosis of lung cancer. Thorax. 1988;43:540-544.
66 Raab SS, Slagel DD, Hughes JH, et al. Sensitivity and cost effectiveness of fine needle aspiration with immunocytochemistry. Arch Pathol Lab Med. 1997;121:695-700.
67 Capellari JO, Thompson EN, Wallenhaupt SL. Utility of intraoperative fine needle aspiration biopsy in the surgical management of patients with pulmonary masses. Acta Cytol. 1994;38:707-710.
68 Layfield LJ, Coogan A, Johnston WW, et al. Transthoracic fine needle aspiration biopsy. Sensitivity in relation to guidance technique and lesion size and location. Acta Cytol. 1996;40:687-690.
69 Philips J, Goodman B, Kelly VJ. Percutaneous transthoracic needle biopsy. Pathology. 1982;14:211-213.
70 Stanley JH, Fish GD, Andriole JG, et al. Lung lesions; cytologic diagnosis by fine needle biopsy. Radiology. 1987;162:389-391.
71 Zarbo J, Fenoglio-Preiser CM. Interinstitutional database for comparison of performance in lung fine needle aspiration cytology. Arch Pathol Lab Med. 1992;116:463-470.
72 Afify A, Davila RM. Pulmonary fine needle aspiration biopsy. Assessing the negative diagnosis. Acta Cytol. 1999;43:601-604.
73 Mourad WA, Vallieres E, Power RF, et al. Fine needle aspiration cytology of bronchocentric granulomatosis: a potential diagnostic pitfall. Diagn Cytopathol. 1996;14:263-267.
74 Kato H, Konaka C, Kawate N, et al. Percutaneous fine needle cytology for lung cancer diagnosis. Diagn Cytopathol. 1986;2:277-283.
75 Curtin CT, Proux J, Davis E. Cartilaginous hamartoma of the lung: a potential pitfall in pulmonary fine needle aspiration. Acta Cytol. 1988;32:764.
76 Chen KTK. Cytodiagnostic pitfalls in pulmonary coccidioidomycosis. Diagn Cytopathol. 1995;12:177-180.
77 Tao LC, Weisbrod G, Ritcey EL, et al. False ‘false-positive’ results in diagnostic cytology. Acta Cytol. 1984;28:450-455.
78 Raab SS, Silverman JF. The clinical utility of cytologic typing of lung tumors. Diagn Cytopathol. 1994;10:376-382.
79 Delgado PI, Jorda M, Ganjei-Azar P. Small cell carcinoma versus other lung malignancies: diagnosis by fine-needle aspiration cytology. Cancer. 2000;90:279-285.
80 Payne CR, Hadfield JW, Stovin PG, et al. Diagnostic accuracy of cytology and biopsy in primary bronchial carcinoma. J Clin Pathol. 1981;34:773-778.
81 Bocking A, Klose KC, Kyll HJ, et al. Cytologic versus histologic evaluation of needle biopsy of the lung, hilum and mediastinum. Sensitivity, specificity and tumour typing. Acta Cytol. 1995;39:463-471.
82 Sinner WN. Importance and value of a preoperative diagnosis in oat cell carcinoma by radiography and its verification by fine needle biopsy (FNB). Eur J Radiol. 1985;5:94-98.
83 Weisbrod L, Cunningham I, Tao LC, et al. Small cell anaplastic carcinoma: cytological-histological correlations from percutaneous fine needle aspiration biopsy. J Can Assoc Radiol. 1987;38:204-208.
84 Silverman JF, Finley JL, Park HK, et al. Fine needle aspiration cytology of bronchioloalveolar cell carcinoma of the lung. Acta Cytol. 1985;29:887-894.
85 Tao LC, Weisbrod GL, Pearson FG, et al. Cytologic diagnosis of bronchiolo-alveolar carcinoma by fine needle aspiration biopsy. Cancer. 1986;57:1565-1570.
86 Jarrett DD, Betsill WL. A problem oriented approach regarding the fine needle aspiration cytologic diagnosis of bronchioloalveolar carcinoma of the lung: a comparison of diagnostic criteria with benign lesions mimicking carcinoma. Acta Cytol. 1987;31:684.
87 Lozowski W, Hajdu SI. Cytology and immunocytochemistry of bronchiolo-alveolar carcinoma. Acta Cytol. 1987;31:717-725.
88 Zusman-Harach SB, Harach HR, Gibbs AR. Cytological features of non-small cell carcinomas of the lung in fine needle aspirates. J Clin Pathol. 1991;44:997-1002.
89 Marchevsky AM, Gal AA, Shah S, et al. Morphometry confirms the presence of considerable nuclear size overlap between ‘small cells’ and ‘large cells’ in high-grade pulmonary neuroendocrine neoplasms. Am J Clin Pathol. 2001;116:466-472.
90 Ramzy I. Pulmonary hamartomas: cytologic appearances of fine needle aspiration biopsy. Acta Cytol. 1976;20:15-19.
91 Dunbar F, Leiman G. The aspiration cytology of pulmonary hamartomas. Diagn Cytopathol. 1989;5:174-180.
92 Wiatrowska BA, Yazdi HM, Matzinger FR, et al. Fine needle aspiration biopsy of pulmonary hamartomas. Radiologic, cytologic and immunocytochemical study of 15 cases. Acta Cytol. 1995;39:1167-1174.
93 Wood B, Swarbrick N, Frost F. Diagnosis of pulmonary hamartoma by fine needle biopsy. Acta Cytol. 2008;52(4):412-417.
94 Klein JS, Salomon G, Stewart EA. Transthoracic needle biopsy with a coaxially placed 20-gauge cutting needle: results in 122 patients. Radiology. 1996;198:715-720.
95 Kim HK, Shin BK, Cho SJ, et al. Transthoracic fine needle aspiration and core biopsy of pulmonary lesions. A study of 296 patients. Acta Cytol. 2002;46:1061-1068.
96 Tsai IC, Tsai WL, Chen MC, et al. CT-guided core biopsy of lung lesions: a primer. Am J Roentgenol. 2009;193(5):1228-1235.
97 Yamagami T, Iida S, Kato T, et al. Combining fine-needle aspiration and core biopsy under CT fluoroscopy guidance: a better way to treat patients with lung nodules? Am J Roentgenol. 2003;180(3):811-815.
98 Dahlgren SE, Ekstrom P. Aspiration cytology in the diagnosis of pulmonary tuberculosis. Scand J Resp Dis. 1972;53:196-201.
99 Bailey TM, Akhtar M, Ashraf Ali M. Fine needle aspiration biopsy in the diagnosis of tuberculosis. Acta Cytol. 1985;29:732-736.
100 Robicheaux G, Moinuddin SM, Lee LH. The role of aspiration biopsy cytology in the diagnosis of pulmonary tuberculosis. Am J Clin Pathol. 1985;83:719-722.
101 Silverman JF, Marrow HG. Fine needle aspiration cytology of granulomatous diseases of the lung, including nontuberculous mycobacterium infection. Acta Cytol. 1985;29:535-541.
102 Rajwanshi A, Bhambbhani S, Das DK. Fine needle aspiration cytology diagnosis of tuberculosis. Diagn Cytopathol. 1987;3:13-16.
103 Das DK, Pant CS, Pant JN, et al. Transthoracic (percutaneous) fine needle aspiration cytology diagnosis of pulmonary tuberculosis. Tuber Lung Dis. 1995;76:84-89.
104 Batra P, Wallace JM, Ovenfors CO. Efficacy and complications of transthoracic needle biopsy of lung in patients with Pneumocystis carinii pneumonia and AIDS. J Thorac Imaging. 1987;2:79-80.
105 Sterrett G, Whitaker D, Glancy J. Fine needle aspiration of lung mediastinum and chest wall. Pathol Annu. 1982;17(Part 2):197-228.
106 Sinner WN. Complications of percutaneous transthoracic needle aspiration biopsy. Acta Radiol Diagn (Stockh). 1976;17:813-828.
107 Aberle DR, Gamsu G, Golden JA. Fatal systemic arterial air embolism following lung needle aspiration. Radiology. 1987;165:351-353.
108 Cianci P, Posin JP, Shimshak RR, et al. Air embolism complicating percutaneous thin needle biopsy of lung. Chest. 1987;92:749-751.
109 Pereira P. A fatal case of cerebral artery gas embolism following fine needle biopsy of the lung. Med J Aust. 1993;159:755-771.
110 Kucharczyk W, Weisbrod GI, Cooper JD. Cardiac tamponade as a complication of thin needle aspiration lung biopsy. Chest. 1982;82:120-121.
111 Berquist TH, Bailey PB, Cortese DA, et al. Transthoracic needle biopsy. Accuracy and complications in relation to location and type of lesion. Mayo Clin Proc. 1980;55:475-481.
112 Sinner WN, Zajicek J. Implantation metastasis after percutaneous transthoracic needle aspiration biopsy. Acta Radiol Diagn (Stockh). 1976;17:473-480.
113 McDonald CF, Baird L. Risk of needle track metastasis after fine needle lung aspiration in lung cancer – a case report. Resp Med. 1994;88:631-632.
114 Moloo Z, Finley RJ, Lefcoe MS, et al. Possible spread of bronchogenic carcinoma to the chest wall after a transthoracic fine needle aspiration biopsy. A case report. Acta Cytol. 1985;29:167-169.
115 Muller NL, Bergin CJ, Miller RR, et al. Seeding of malignant cells into the needle track after lung and pleural biopsy. J Can Assoc Radiol. 1986;37:192-194.
116 Carter RR, Wilson JP, Turner HR, et al. Cutaneous blastomycosis as a complication of transthoracic needle aspiration. Chest. 1987;91:917-918.
117 Watts WJ, Green RA. Bacteremia following transbronchial fine needle aspiration. A case report. Chest. 1984;85:295.
118 Bhat N, Miller R, Le Riche J, et al. Aspiration biopsy in pulmonary opportunistic infections. Acta Cytol. 1977;21:206-209.
119 Das DK, Bhambhani S, Pant CS. Ultrasound guided fine needle aspiration cytology: diagnosis of hydatid disease of the abdomen and thorax. Diagn Cytopathol. 1995;12:173-176.
120 McCorkell SJ. Unintended percutaneous aspiration of pulmonary echinococcal cysts. Am J Roentgenol. 1984;143:123-126.
121 Arakawa H, Nakajima Y, Kurihawa Y, et al. CT-guided transthoracic needle biopsy: a comparison between automated biopsy gun and fine needle aspiration. Clin Radiol. 1996;51:503-506.
122 Mrkve O, Skaarland E, Myking A, et al. Transthoracic fine needle aspiration guided by fluoroscopy: validity and complications with 19 operators. Respiration. 1988;53:239-245.
123 Akhtar M, Ashraf Ali M, Huq M, et al. Fine needle biopsy: comparison of cellular yield with and without aspiration. Diagn Cytopathol. 1989;5:162-165.
124 Yue X-H, Zheng S-F. Cytologic diagnosis by transthoracic fine needle sampling without aspiration. Acta Cytol. 1989;33:805-808.
125 van Sonnenberg E, Casola G, Ho M, et al. Difficult thoracic lesions: CT-guided biopsy experience in 150 cases. Radiology. 1988;167:457-461.
126 Knox AM, Fon GT, Orell S. Fine needle aspiration in the chest under CT control. Australas Radiol. 1991;35:152-156.
127 Pedersen OM, Aasen TB, Gulsvik A. Fine needle aspiration biopsy of mediastinal and peripheral pulmonary masses guided by real-time sonography. Chest. 1986;89:504-508.
128 Pang JA, Tsang V, Hom BL, et al. Ultrasound-guided tissue-core biopsy of thoracic lesions with Trucut and Surecut needles. Chest. 1987;91:823-828.
129 Chen CC, Hsu WH, Huang CM, et al. Ultrasound guided fine needle aspiration biopsy of solitary pulmonary nodules. J Clin Ultrasound. 1995;23:531-536.
130 Hecht JL, Pinkus JL, Weinstein LJ, et al. The value of thyroid transcription factor-1 in cytologic preparations as a marker for metastatic adenocarcinoma of lung origin. Am J Clin Pathol. 2001;116:483-488.
131 Chhieng DC, Cangiarella JF, Zakowski MF, et al. Use of thyroid transcription factor 1, PE-10, and cytokeratins 7 and 20 in discriminating between primary lung carcinomas and metastatic lesions in fine-needle aspiration biopsy specimens. Cancer. 2001;93:330-336.
132 Jerome Marson V, Mazieres J, Groussard O, et al. Expression of TTF-1 and cytokeratins in primary and secondary epithelial lung tumours: correlation with histological type and grade. Histopathology. 2004;45(2):125-134.
133 Blumenfeld W, Turi GK, Harrison G, et al. Utility of cytokeratin 7 and 20 subset analysis as an aid in the identification of primary site of origin of malignancy in cytologic specimens. Diagn Cytopathol. 1999;20:63-66.
134 Johansson L. Histopathologic classification of lung cancer: Relevance of cytokeratin and TTF-1 immunophenotyping. Ann Diagn Pathol. 2004;8(5):259-267.
135 Tot T. The value of cytokeratins 20 and 7 in discriminating metastatic adenocarcinomas from pleural mesotheliomas. Cancer. 2001;92(10):2727-2732.
136 Wakely PEJr. Aspiration cytopathology of malignant lymphoma: coming of age. Cancer. 1999;87:325-345.
137 Young NA, Al-Saleem T. Diagnosis of lymphoma by fine-needle aspiration cytology using the revised European-American classification of lymphoid neoplasms. Cancer. 1999;87:325-345.
138 Wakely PEJr. Fine-needle aspiration cytopathology in diagnosis and classification of malignant lymphoma: accurate and reliable? Diagn Cytopathol. 2000;22:120-125.
139 Angeletti C. Application of proteomic technologies to cytologic specimens. A review. Acta Cytol. 2003;47:535-544.
140 Rimm DL. Impact of microarray technologies on cytopathology. Overview of technologies and commentary on current and future implications for pathologists and cytopathologists. Acta Cytol. 2001;45:111-114.
141 Gomez-Fernandez C, Mejias A, Walker G, et al. Immunohistochemical expression of estrogen receptor in adenocarcinomas of the lung: the antibody factor. Appl Immunohistochem Mol Morphol. 2010;18(2):137-141.
142 Inamura K, Satoh Y, Okumura S, et al. Pulmonary adenocarcinomas with enteric differentiation: histologic and immunohistochemical characteristics compared with metastatic colorectal cancers and usual pulmonary adenocarcinomas. Am J Surg Pathol. 2005;29(5):660-665.
143 Khayyata S, Yun S, Pasha T, et al. Value of p63 and CK5/6 in distinguishing squamous cell carcinoma from adenocarcinoma in lung fine-needle aspiration specimens. Diagn Cytopathol. 2009;37(3):178-183.
144 Kubba LA, McCluggage WG, Liu J, et al. Thyroid transcription factor-1 expression in ovarian epithelial neoplasms. Mod Pathol. 2008;21(4):485-490.
145 Siami K, McCluggage WG, Ordonez NG, et al. Thyroid transcription factor-1 expression in endometrial and endocervical adenocarcinomas. Am J Surg Pathol. 2007;31(11):1759-1763.
146 Xu B, Thong N, Tan D, et al. Expression of thyroid transcription factor-1 in colorectal carcinoma. Appl Immunohistochem Mol Morphol. 2010;18(3):244-249.
147 Lin X, Saad RS, Luckasevic TM, et al. Diagnostic value of CDX-2 and TTF-1 expressions in separating metastatic neuroendocrine neoplasms of unknown origin. Appl Immunohistochem Mol Morphol. 2007;15(4):407-414.
148 Sehested M, Francis D, Hainau B. Electron microscopy of transthoracic fine needle aspiration biopsies. Acta Pathol Microbiol Immunol Scand (A). 1983;91:457-461.
149 Kurtz SM. Rapid ultrastructural examination of FNAs in the diagnosis of intrathoracic tumours. Diagn Cytopathol. 1992;8:289-292.
150 Reyes CV, Jensen JD, Graham G. Adenocarcinoma of the lung: electron microscopy of fine-needle aspiration biopsy specimens – a review of 73 cases. Diagn Cytopathol. 1999;20:257-260.
151 Lynch TJ, Bell DW, Sordella R, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med. 2004;350(21):2129-2139.
152 Gazdar AF. Activating and resistance mutations of EGFR in non-small-cell lung cancer: role in clinical response to EGFR tyrosine kinase inhibitors. Oncogene. 2009;28(Suppl 1):S24-S31.
153 Finberg KE, Sequist LV, Joshi VA, et al. Mucinous differentiation correlates with absence of EGFR mutation and presence of KRAS mutation in lung adenocarcinomas with bronchioloalveolar features. J Mol Diagn. 2007;9(3):320-326.
154 Smith GD, Chadwick BE, Willmore-Payne C, et al. Detection of epidermal growth factor receptor gene mutations in cytology specimens from patients with non-small cell lung cancer utilising high-resolution melting amplicon analysis. J Clin Pathol. 2008;61(4):487-493.
155 Garfield DH, Cadranel J, West HL. Bronchioloalveolar carcinoma: the case for two diseases. Clin Lung Cancer. 2008;9(1):24-29.
156 Zheng Z, Chen T, Li X, et al. DNA synthesis and repair genes RRM1 and ERCC1 in lung cancer. N Engl J Med. 2007;356(8):800-808.
157 Reynolds C, Obasaju C, Schell MJ, et al. Randomized phase III trial of gemcitabine-based chemotherapy with in situ RRM1 and ERCC1 protein levels for response prediction in non-small-cell lung cancer. J Clin Oncol. 2009;27(34):5808-5815.
158 Solomon B, Varella-Garcia M, Camidge DR. ALK gene rearrangements: a new therapeutic target in a molecularly defined subset of non-small cell lung cancer. J Thorac Oncol. 2009;4(12):1450-1454.
159 Shaw AT, Yeap BY, Mino-Kenudson M, et al. Clinical features and outcome of patients with non-small-cell lung cancer who harbor EML4-ALK. J Clin Oncol. 2009;27(26):4247-4253.
160 Johnson DH, Fehrenbacher L, Novotny WF, et al. Randomized phase II trial comparing bevacizumab plus carboplatin and paclitaxel with carboplatin and paclitaxel alone in previously untreated locally advanced or metastatic non-small-cell lung cancer. J Clin Oncol. 2004;22(11):2184-2191.
161 Pardo J, Martinez-Peñuela AM, Sola JJ, et al. Large cell carcinoma of the lung: an endangered species? Appl Immunohistochem Mol Morphol. 2009;17(5):383-392.
162 Idowo MO, Fuller CE, Powers CN. Non-small cell lumg carcinoma; a diagnosis beyond its prime. Pathology Case Reviews. 2009;14:199-205.
163 Gattuso P, Castelli M, Reyes AV, et al. Cutaneous and subcutaneous masses of the chest wall: a fine needle-aspiration study. Diagn Cytopathol. 1996;15:374-376.
164 Syed S, Zaharopoulos P. Thoracic splenosis diagnosed by fine-needle aspiration cytology: a case report. Diagn Cytopathol. 2001;25:321-324.
165 Sarda R, Sproat I, Kurtycz DF, et al. Pulmonary parenchymal splenosis. Diagn Cytopathol. 2001;24:352-355.
166 Chen KTK. Megakaryocytes in a fine needle aspirate of the lung. Acta Cytol. 1987;31:81-82.
167 Silverman JF. Fine needle aspiration cytology of infectious and inflammatory diseases and other nonneoplastic disorders. In: Kline T, editor. Guides to clinical aspiration biopsy series. New York: Igaku-Shoin; 1991:201.
168 Covell JL, Feldman PS. Fine needle aspiration diagnosis of aspiration pneumonitis (phytopneumonitis). Acta Cytol. 1984;38:77-80.
169 Wallace RJ, Cohen A, Awe RJ, et al. Carcinomatous lung abscess. Diagnosis by bronchoscopy and cytopathology. JAMA. 1979;242:521-522.
170 Goel MM, Ranjan V, Dhole TN, et al. Polymerase chain reaction vs. conventional diagnosis in fine needle aspirates of tuberculous lymph nodes. Acta Cytol. 2001;45:333-340.
171 Gong G, Lee H, Kang GH, et al. Nested PCR for diagnosis of tuberculous lymphadenitis and PCR-SSCP for identification of rifampicin resistance in fine-needle aspirates. Diagn Cytopathol. 2002;26:228-231.
172 Pauli G, Pelletier A, Bohner C, et al. Transbronchial needle aspiration in the diagnosis of sarcoidosis. Chest. 1984;85:482-484.
173 Vernon SE. Nodular pulmonary sarcoidosis. Diagnosis with fine needle aspiration biopsy. Acta Cytol. 1985;29:473-476.
174 Morales CF, Patefield AJ, Strollo PJ, et al. Flexible transbronchial needle aspiration in the diagnosis of sarcoidosis. Chest. 1994;106:709.
175 Fekete PS, Campbell WJJr, Bernadino ME. Transthoracic needle aspiration biopsy in Wegener’s granulomatosis. Morphologic findings in five cases. Acta Cytol. 1990;34:155-160.
176 Kaneishi NK, Howell LP, Russell LA, et al. Fine needle aspiration cytology of pulmonary Wegener’s granulomatosis with biopsy correlation. A report of three cases. Acta Cytol. 1995;39:1094-1100.
177 Filho JS, Soares MF, Wal R, et al. Fine-needle aspiration cytology of pulmonary rheumatoid nodule: case report and review of the major cytologic features. Diagn Cytopathol. 2002;26:150-153.
178 Lalvani A. Diagnosing tuberculosis infection in the 21st century: new tools to tackle an old enemy. Chest. 2007;131(6):1898-1906.
179 Whitaker D, Sterrett GF. Cryptococcus neoformans diagnosed by fine needle aspiration cytology of the lung. Acta Cytol. 1976;20:105-107.
180 Silverman JF, Johnsrude IS. Fine needle aspiration cytology of granulomatous cryptococcosis of the lung. Acta Cytol. 1985;29:157-161.
181 Williamson JD, Silverman JF, Mallak CT, et al. Atypical cytomorphologic appearance of Cryptococcus neoformans. A report of five cases. Acta Cytol. 1996;40:363-370.
182 Maygarden SJ, Flanders EL. Mycobacteria can be seen as ‘negative images’ in cytology smears from patients with acquired immunodeficiency syndrome. Mod Pathol. 1989;2:239-243.
183 Silverman JF, Holter JF, Berns LA, et al. Negative images due to clofazimine crystals simulating MAI infection in a bronchoalveolar lavage specimen. Diagn Cytopathol. 1993;9:534-539.
184 McCalmont TH, Silverman JF, Geisinger KR. Cytologic diagnosis of aspergillus in cardiac transplantation. Arch Surg. 1991;126:394-396.
185 Stanley MW, Deeike M, Knoedler J, et al. Pulmonary mycetomas in immunocompetent patients: diagnosis by fine needle aspiration. Diagn Cytopathol. 1992;8:577-579.
186 Fischler DF, Hall GS, Gordon S, et al. Aspergillus in cytology specimens: a review of 45 specimens from 36 patients. Diagn Cytopathol. 1997;16:26-30.
187 Walts AE. Pseudallescheria: an underdiagnosed fungus? Diagn Cytopathol. 2001;25:153-157.
188 Farley ML, Mabry L, Munoz LA, et al. Crystals occurring in pulmonary cytology specimens. Association with Aspergillus infection. Acta Cytol. 1985;29:737-744.
189 Lee SH, Barnes WG, Schaetzel WP. Pulmonary aspergillosis and the importance of oxalate crystal recognition in cytology specimens. Arch Pathol Lab Med. 1986;110:1176-1179.
190 Hsu C-Y. Cytologic diagnosis of pulmonary cryptococcosis in immunocompromised hosts. Acta Cytol. 1993;37:667-672.
191 Kushner YB, Brimo F, Schwartzman K. A rare case of pulmonary cryptococcal inflammatory myofibroblastic tumor diagnosed by fine needle aspiration cytology. Diagn Cytopathol. 2010;38(6):447-451.
192 Busmanis I, Harney M, Hellyar A. Nocardiosis diagnosed by lung FNA. Acta Cytol. 1995;12:56-58.
193 Hajdu SI. Cytology and pathology of acquired immune deficiency syndrome. Acta Cytol. 1986;30:599-602.
194 Strigle SM, Gal AA. A review of pulmonary cytopathology in the acquired immunodeficiency syndrome. Diagn Cytopathol. 1989;5:44-54.
195 Buchanan AJ, Gupta RK. Cytomegalovirus infection of the lung: cytomorphologic diagnosis by fine needle aspiration cytology. Diagn Cytopathol. 1986;2:341-342.
196 Freedman SI, Ang EP, Haley RS. Identification of Coccidioidomycosis of the lung by fine needle aspiration biopsy. Acta Cytol. 1986;30:420-424.
197 Raab SS, Silverman JF, Zimmerman KG. Fine needle aspiration biopsy of pulmonary coccidioidomycosis: spectrum of cytology findings in seventy-three patients. Am J Clin Pathol. 1993;99:582-587.
198 Rangdaeng S, Alpert LC, Khiyami A, et al. Pulmonary paragonamiasis. Report of a case with diagnosis by fine needle aspiration cytology. Acta Cytol. 1992;36:31-36.
199 Kapila K, Verma K. Aspiration cytology diagnosis of echinococcosis. Diagn Cytopathol. 1990;6:301-303.
200 Avasthi R, Jain AP, Swaroop K, et al. Bancroftian microfilariasis with pulmonary tuberculosis. Report of a case with diagnosis by fine needle aspiration. Acta Cytol. 1991;35:717-718.
201 Hawkins AG, Hsiu JG, Smith RMIII, et al. Pulmonary dirofilariasis diagnosed by fine needle aspiration biopsy. A case report. Acta Cytol. 1985;29:19-22.
202 Zaman SS, van Hoeven KH, Slott S, et al. Distinction between bronchioloalveolar carcinoma and hyperplastic pulmonary proliferations: a cytologic and morphometric analysis. Diagn Cytopathol. 1997;16:396-401.
203 Roggli VL, Johnston WW, Kaminsky DB. Asbestos bodies in fine needle aspirates of the lung. Acta Cytol. 1984;28:493-498.
204 Leiman G. Asbestos bodies in pulmonary fine needle aspirates: a more sinister finding than in other cytologic specimens? Acta Cytol. 1986;30:555-556.
205 Silverman JF, Weaver MD, Shaw R, et al. Fine needle aspiration cytology of pulmonary infarct. Acta Cytol. 1985;29:162-166.
206 Hsiu JG, Stitik FP, d’Amato NA, et al. Primary amyloidosis presenting as a unilateral hilar mass. Report of a case diagnosed by fine needle aspiration biopsy. Acta Cytol. 1986;30:55-58.
207 Dundore PA, Aisner SC, Templeton PA, et al. Nodular pulmonary amyloidosis: diagnosis by fine needle aspiration cytology and a review of the literature. Diagn Cytopathol. 1993;9:562-564.
208 Liaw Y-S, Kuo S-H, Yang P-C, et al. Nodular amyloidosis of the lung and the breast mimicking breast carcinoma with pulmonary metastases. Eur Resp J. 1995;5:871-873.
209 Walker AN, Feldman PS, Walker GK. Fine needle aspiration of thoracic extramedullary hematopoiesis. Acta Cytol. 1983;27:170-172.
210 Lambert C, Gansler T, Mansour KA, et al. Pulmonary malakoplakia diagnosed by fine needle aspiration. A case report. Acta Cytol. 1997;41:1833-1838.
211 Miller WTJr, Gupta PK, Grippi MA, et al. Rounded atelectasis: diagnosis by fine-needle aspiration cytology. Diagn Cytopathol. 1992;8:617-620.
212 Smith AR, Raab SS, Landreneau RJ, et al. Fine-needle aspiration cytologic features of pseudovascular adenoid squamous-cell carcinoma of the lung. Diagn Cytopathol. 1999;21:265-270.
213 Wu M, Wang B, Gil J, et al. p63 and TTF-1 immunostaining. A useful marker panel for distinguishing small cell carcinoma of lung from poorly differentiated squamous cell carcinoma of lung. Am J Clin Pathol. 2003;119:696-702.
214 Brooks B, Baandrup U. Peripheral low grade mucoepidermoid carcinoma of the lung – needle aspiration cytodiagnosis and histology. Cytopathol. 1992;3:259-265.
215 Mooney EE, Dodd LG, Vollmer RT, et al. Fine needle aspiration biopsy diagnosis of primary bronchial basaloid squamous carcinoma. Diagn Cytopathol. 1997;16:187-188.
216 Weichert W, Schewe C, Denkert C, et al. Molecular HPV typing as a diagnostic tool to discriminate primary from metastatic squamous cell carcinoma of the lung. Am J Surg Pathol. 2009;33(4):513-520.
217 Travis WD, Brambilla E, Muller-Hemerlink HK, Harris CC, editors. World Health Organization Classification of Tumours. Pathology and Genetics of Tumours of the Lung, Pleura, Thymus and Heart. Lyon: IARC Press, 2004.
218 Ullmann R, Bongiovanni M, Halbwedl I, et al. Is high-grade adenomatous hyperplasia an early bronchioloalveolar adenocarcinoma? J Pathol. 2003;201:371-376.
219 Flieder DB. Screen-detected adenocarcinoma of the lung. Practical points for surgical pathologists. Am J Clin Pathol. 2003;119(Suppl):S39-S57.
220 Wang SE, Nieberg RK. Fine needle aspiration cytology of sclerosing hemangioma of the lung, a mimicker of bronchioloalveolar carcinoma. Acta Cytol. 1986;30:51-55.
221 Wojcik EM, Sneige N, Lawrence DD, et al. Fine needle aspiration cytology of sclerosing haemangioma of the lung: case report with immunohistochemical study. Diagn Cytopathol. 1993;9:304-309.
222 Kaw YT, Nayak RN. Fine needle biopsy cytology of sclerosing haemangioma of the lung. A case report. Acta Cytol. 1993;37:933-937.
223 Gottschalk-Sabag S, Hadas Halpern I, Glick T. Sclerosing hemangioma of lung mimicking carcinoma diagnosed by fine needle aspiration (FNA) cytology. Cytopathol. 1995;6:115-120.
224 Gao ZH, Urbanski SJ. The spectrum of pulmonary mucinous cystic neoplasia: a clinicopathologic and immunohistochemical study of ten cases and review of literature. Am J Clin Pathol. 2005;124(1):62-70.
225 Moran CA. Mucin-rich tumors of the lung. Adv Anatom Pathol. 1995;5:299-305.
226 Davenport RD. Diagnostic value of crush artefact in cytologic specimens. Occurrence in small cell carcinoma of the lung. Acta Cytol. 1990;34:502-504.
227 Mullins RK, Thompson SK, Coogan PS, et al. Paranuclear blue inclusions: an aid in the diagnosis of primary and metastatic pulmonary small cell carcinoma. Diagn Cytopathol. 1994;10:332-335.
228 Renshaw AA, Voytek TM, Haja J, et alCytology Committee, College of American Pathologists. Distinguishing small cell carcinoma from non-small cell carcinoma of the lung: correlating cytologic features and performance in the College of American Pathologists Non-Gynecologic Cytology Program. Arch Pathol Lab Med. 2005;129(5):619-623.
229 Dugan JM. Cytologic diagnosis of basal cell (basaloid) carcinoma of the lung. A report of two cases. Acta Cytol. 1995;39:539-542.
230 Vesoulis Z. Metastatic laryngeal basaloid squamous cell carcinoma simulating primary small cell carcinoma of the lung on fine needle aspiration lung biopsy. A case report. Acta Cytol. 1998;42:783-787.
231 Rollins SD, Genack LJ, Schumann GB. Primary cytodiagnosis of dually differentiated lung cancer by transthoracic fine needle aspiration. Acta Cytol. 1988;32:231-234.
232 Roggli VL, Vollmer RT, Greenberg SD, et al. Lung cancer heterogeneity: a blinded and randomized study of 100 consecutive cases. Human Pathol. 1985;16:569-579.
233 Nicholson SA, Beasley MB, Brambilla E, et al. Small cell lung carcinoma (SCLC): a clinicopathologic study of 100 cases with surgical specimens. Am J Surg Pathol. 2002;26(9):1184-1197.
234 Fushimi H, Kukui M, Morino H, et al. Detection of large cell component in small cell carcinoma by combined cytologic and histologic examination and its clinical implication. Cancer. 1992;70:599-605.
235 Guinee DGJr, Fishback NF, Koss MN, et al. The spectrum of immunohistochemical staining of small-cell lung carcinoma in specimens from transbronchial and open-lung biopsies. Am J Clin Pathol. 1994;102(4):406-414.
236 Lin O, Olgac S, Green I, et al. Immunohistochemical staining of cytologic smears with MIB-1 helps distinguish low-grade from high-grade neuroendocrine neoplasms. Am J Clin Pathol. 2003;120:209-216.
237 Pitman MB, Sherman ME, Black-Schaffer WS. The use of fine-needle aspiration in the diagnosis of metastatic pulmonary adenoid cystic carcinoma. Otolaryngol Head Neck Surg. 1991;104:441-447.
238 Craig ID, Finley RJ. Spindle cell carcinoid tumour of lung. Cytologic, histopathologic and ultrastructural features. Acta Cytol. 1982;26:495-498.
239 Kaufmann O, Dietel M. Expression of thyroid transcription factor-1 in pulmonary and extrapulmonary small cell carcinomas and other neuroendocrine carcinomas of various primary sites. Histopathology. 2000;36(5):415-420.
240 Finley JL, Silverman JF, Dabbs DJ. Fine needle aspiration cytology of pulmonary carcinosarcoma with immunocytochemical and ultrastructural observations. Diagn Cytopathol. 1988;14:239-243.
241 Schantz HD, Ramzy I, Tio FO, et al. Metastatic spindle cell carcinoma. Cytologic features and differential diagnosis. Acta Cytol. 1985;29:435-441.
242 Cosgrove MM, Chandrasoma PT, Martin SE. Diagnosis of pulmonary blastoma by fine needle aspiration biopsy: cytologic and immunocytochemical findings. Diagn Cytopathol. 1991;7:83-87.
243 Chow LT, Chow WH, Tsui WM, et al. Fine-needle aspiration cytologic diagnosis of lymphoepithelioma-like carcinoma of the lung. Report of two cases with immunohistochemical study. Am J Clin Pathol. 1995;103:35-40.
244 Craig ID, Desrosiers P, Lefcoe MS. Giant-cell carcinoma of the lung. A cytologic study. Acta Cytol. 1983;27:293-298.
245 Nonomura A, Mizukami Y, Shimizu J, et al. Small giant cell carcinoma of the lung diagnosed preoperatively by transthoracic aspiration cytology. Acta Cytol. 1995;39:129-133.
246 Mitchell ML, Parker FP. Capillaries. A cytologic feature of pulmonary carcinoid tumours. Acta Cytol. 1991;35:183-185.
247 Anderson C, Ludwig ME, O’Donnell M, et al. Fine needle aspiration cytology of pulmonary carcinoid tumours. Acta Cytol. 1990;34:505-510.
248 Chow LT-C, Chan S-K, Chow W-H, et al. Pulmonary sclerosing haemangioma. Report of a case with diagnosis by FNA. Acta Cytol. 1992;36:287-292.
249 Krishnamurthy SC, Naresh KN, Soni M, et al. Sclerosing haemangioma of the lung: a potential source of error in fine needle aspiration cytology. Acta Cytol. 1994;38:111-112.
250 Du EZ, Goldstraw P, Zacharias J, et al. TTF-1 expression is specific for lung primary in typical and atypical carcinoids: TTF-1-positive carcinoids are predominantly in peripheral location. Hum Pathol. 2004 Jul;35(7):825-831.
251 Fekete PS, Cohen C, DeRose PB. Pulmonary spindle cell carcinoid. Needle aspiration biopsy, histologic and immunohistochemical findings. Acta Cytol. 1990;34:50-56.
252 Satoh Y, Fujiyama J, Ueno M, et al. High cellular atypia in a pulmonary tumorlet. Report of a case with cytologic findings. Acta Cytol. 2000;44:242-246.
253 Travis WD, Rush W, Flieder DB, et al. Survival analysis of200 pulmonary neuroendocrine tumors with clarification of criteria for atypical carcinoid and its separation from typical carcinoid. Am J Surg Pathol. 1998;22(8):934-944.
254 Pilotti S, Rilke F, Lombardi L. Pulmonary carcinoid with glandular features. Report of two cases with positive fine needle aspiration biopsy cytology. Acta Cytol. 1983;27:511-514.
255 Nguyen GK, Shnitka TK. Aspiration biopsy cytology of adenocarcinoid tumor of the bronchial tree. Acta Cytol. 1986;31:726-730.
256 Travis WD. Lung tumours with neuroendocrine differentiation. Eur J Cancer. 2009;45(Suppl 1):251-266.
257 Sturm N, Lantuéjoul S, Laverrière MH, et al. Thyroid transcription factor 1 and cytokeratins 1, 5, 10, 14(34betaE12) expression in basaloid and large-cell neuroendocrine carcinomas of the lung. Hum Pathol. 2001;32(9):918-925.
258 Davies SJ, Gosney JR, Hansell DM, et al. Diffuse idiopathic pulmonary neuroendocrine cell hyperplasia: an under-recognised spectrum of disease. Thorax. 2007;62(3):248-252.
259 Dacic S. Pulmonary preneoplasia. Arch Pathol Lab Med. 2008;132(7):1073-1078.
260 Moran CA. Primary salivary gland-type tumors of the lung. Semin Diagn Pathol. 1995;12(2):106-122.
261 Ozkara SK, Turan G. Fine needle aspiration cytopathology of primary solid adenoid cystic carcinoma of the lung: a case report. Acta Cytol. 2009;53(6):707-710.
262 Smith RC, Amy RW. Adenoid cystic carcinoma metastatic to the lung. Report of a case diagnosed by fine needle aspiration biopsy cytology. Acta Cytol. 1985;29:535-536.
263 Nguyen GK. Cytology of bronchial gland carcinoma. Acta Cytol. 1988;32:235-239.
264 Segletes LA, Steffee CH, Geisinger KR. Cytology of primary pulmonary mucoepidermoid and adenoid cystic carcinoma. A report of four cases. Acta Cytol. 1999;43:1091-1097.
265 Zhang Y, Gomez-Fernandez CR, Jorda M, et al. Fine-needle aspiration (FNA) and pleural fluid cytology diagnosis of benign metastasizing pleomorphic adenoma of the parotid gland in the lung: a case report and review of literature. Diagn Cytopathol. 2009;37(11):828-831.
266 Ang KL, Dhannapuneni VR, Morgan WE, et al. Primary pulmonary pleomorphic adenoma. An immunohistochemical study and review of the literature. Arch Pathol Lab Med. 2003;127(5):621-622.
267 Watanabe K, Ono N, Hoshi T, et al. Fine-needle aspiration cytology of bronchial acinic cell carcinoma: a case report. Diagn Cytopathol. 2004;30(5):359-361.
268 Cwierzyk TA, Glasberg SS, Virshup MA, et al. Pulmonary oncocytoma: report of a case with cytologic, histologic and electron microscopic study. Acta Cytol. 1985;29:620-623.
269 Ogino S, al-Kaisi N, Abdul-Karim FW. Cytopathology of oncocytic carcinoid tumor of the lung mimicking granular cell tumor. A case report. Acta Cytol. 2000;44(2):247-250.
270 Nguyen CV, Suster S, Moran CA. Pulmonary epithelial-myoepithelial carcinoma: a clinicopathologic and immunohistochemical study of 5 cases. Hum Pathol. 2009;40(3):366-373.
271 Flint A, Lloyd R. Pulmonary metastases of colonic carcinoma. Acta Cytol. 1992;36:230-235.
272 Goldstein NS, Thomas M. Mucinous and nonmucinous bronchioloalveolar adenocarcinomas have distinct staining patterns with thyroid transcription factor and cytokeratin 20 antibodies. Am J Clin Pathol. 2001;116:319-325.
273 Shah RN, Badve S, Papreddy K, et al. Expression of cytokeratin20 in mucinous bronchioloalveolar carcinoma. Hum Pathol. 2002;33(9):915-920.
274 Wang LJ, Greaves WO, Sabo E, et al. GCDFP-15 positive and TTF-1 negative primary lung neoplasms: a tissue microarray study of 381 primary lung tumors. Appl Immunohistochem Mol Morphol. 2009;17(6):505-511.
275 Nguyen GK. Fine needle aspiration biopsy cytology of metastatic renal cell carcinoma. Acta Cytol. 1988;32:409-414.
276 Saleh H, Masood S, Wynn G, et al. Unsuspected metastatic renal cell carcinoma diagnosed by fine needle aspiration biopsy. A report of four cases with immunocytochemical contributions. Acta Cytol. 1994;38:554-561.
277 Gokden N, Gokden M, Phan DC, et al. The utility of PAX-2 in distinguishing metastatic clear cell renal cell carcinoma from its morphologic mimics: an immunohistochemical study with comparison to renal cell carcinoma marker. Am J Surg Pathol. 2008;32(10):1462-1467.
278 Nguyen GK. Aspiration biopsy cytology of benign clear cell (’sugar’) tumour of the lung. Acta Cytol. 1989;33:511-515.
279 Hughes JH, Jensen CS, Donnelly AD, et al. The role of fine-needle aspiration cytology in the evaluation of metastatic clear cell tumors. Cancer. 1999;87:380-389.
280 Johnson TL, Kini SR. Cytologic features of transitional cell carcinoma. Diagn Cytopathol. 1993;9:270-278.
281 Powers CN, Elbadawi A. ‘Cercariform’ cells: a clue to the cytodiagnosis of transitional cell origin of metastatic neoplasms? Diagn Cytopathol. 1995;13:15-21.
282 Hida CA, Gupta PK. Cercariform cells: are they specific for transitional cell carcinoma? Cancer. 1999;87:69-74.
283 Craig ID, Shum DT, Desrosiers P, et al. Choriocarcinoma metastatic to the lung: a cytologic study with identification of human choriogonadotropin with an immunoperoxidase technique. Acta Cytol. 1983;27:647-650.
284 Ehya H. Cytology of mesothelioma of the tunica vaginalis metastatic to the lung. Acta Cytol. 1985;29:79-84.
285 Tao LC. Pulmonary metastases from intracranial meningioma diagnosed by aspiration biopsy cytology. Acta Cytol. 1991;35:524-528.
286 Haddad MG, Silverman J. Fine needle aspiration cytology of metastatic basal cell carcinoma of the skin to the lung. Diagn Cytopathol. 1994;10:15-19.
287 McCutcheon JM, Mancer K, Dardick I. Acinic cell tumour: a metastasis in the lung diagnosed by electron microscopy of aspirated material. Cytopathology. 1992;3:373-377.
288 van Hoeven KH, Kellogg K, Bavaria JE. Pulmonary metastasis from histologically benign giant cell tumour of bone. Report of a case diagnosed by fine needle aspiration cytology. Acta Cytol. 1994;38:412-414.
289 Hafiz MA, Wang K-P, Berkman A. Fine needle aspiration diagnosis of benign metastasising leiomyoma of the lung. A case report. Acta Cytol. 1994;38:398-402.
290 Perez-Ordonez B, Bedard YC. Metastatic adamantinoma diagnosed by fine needle aspiration biopsy of the lung. Diagn Cytopathol. 1994;10:347-351.
291 Saint Martin GA, Aranha GV, Castelli MJ, et al. Fine needle aspiration diagnosis of metastatic anaplastic sacrococcygeal ependymoma to the lungs. Diagn Cytopathol. 1996;15:228-230.
292 Landolt U. Pleomorphic adenoma of the salivary glands metastatic to the lung: diagnosis by FNA cytology. Acta Cytol. 1990;34:101-102.
293 Perry MD, Gore M, Seigler HF, et al. Fine needle aspiration biopsy of metastatic melanoma. A morphologic analysis of 174 cases. Acta Cytol. 1986;30:385-397.
294 Blaustein RL. Fine needle aspiration of a metastatic breast carcinoma of the lung with melanin pigmentation. Diagn Cytopathol. 1990;6:364-365.
295 Slagel DD, Raab SS, Silverman JF. Fine needle aspiration biopsy of metastatic malignant melanoma with ‘rhabdoid’ features. Frequency, cytologic features, pitfalls and ancillary studies. Acta Cytol. 1997;41:1426-1430.
296 Zubovits J, Buzney E, Yu L, et al. HMB-45, S-100, NK1/C3, and MART-1 in metastatic melanoma. Hum Pathol. 2004;35(2):217-223.
297 Ost D, Joseph C, Sogoloff H, et al. Primary pulmonary melanoma: case report and literature review. Mayo Clin Proc. 1999;74(1):62-66.
298 Hummel P, Cangiarella JF, Cohen JM, et al. Transthoracic fine-needle aspiration biopsy of pulmonary spindle cell and mesenchymal lesions: a study of 61 cases. Cancer. 2001;93:187-198.
299 Saleh H, Beydoun R, Masood S. Cytology of malignant Schwannoma metastatic to the lung. Report of a case with diagnosis by fine needle aspiration biopsy. Acta Cytol. 1993;37:409-412.
300 Silverman J, Weaver MD, Gardner N, et al. Aspiration biopsy cytology of malignant Schwannoma metastatic to the lung. Acta Cytol. 1985;29:15-19.
301 Lozowski MS, Mishriki Y, Epstein H. Metastatic malignant fibrous histiocytoma in lung examined by fine needle aspiration: case report and literature review. Acta Cytol. 1980;24:350-354.
302 Hsiu JG, Kreuger JK, d’Amato NA, et al. Primary malignant fibrous histiocytoma of the lung. Acta Cytol. 1987;31:345-350.
303 Kawahara E, Nakanishi I, Kuroda Y, et al. Fine needle aspiration biopsy of primary malignant fibrous histiocytoma of the lung. Acta Cytol. 1988;32:226-230.
304 Krumerman MS. Leiomyosarcoma of the lung: primary cytodiagnosis in two consecutive cases. Acta Cytol. 1977;21:103-108.
305 Nickels J, Koivuniemi A. Cytology of malignant haemangiopericytoma. Acta Cytol. 1979;23:119-125.
306 Perry MD, Furlong JW, Johnston WW. Fine needle aspiration cytology of metastatic dermatofibrosarcoma protuberans. A case report. Acta Cytol. 1986;30:507-512.
307 Kilpatrick SE, Teot LA, Stanley MW, et al. Fine needle aspiration biopsy of synovial sarcoma. A cytomorphologic analysis of primary, recurrent and metastatic tumors. Am J Clin Pathol. 1996;106:769-775.
308 Costa I, Lerma E, Esteve E, et al. Aspiration cytology of lung metastasis of monophasic synovial sarcoma. Report of a case. Acta Cytol. 1997;41:1289-1292.
309 Zaharopoulos P, Wong JY, Lamke CR. Endometrial stromal sarcoma. Cytology of pulmonary metastasis including ultrastructural study. Acta Cytol. 1982;26:49-54.
310 Gattuso P, Reddy VB, Castelli MJ. Fine needle aspiration biopsy of paranasal chondrosarcoma metastatic to lung. Acta Cytol. 1990;34:102-104.
311 Stanfield B, Powers CN, Desch CE, et al. Fine needle aspiration cytology of an unusual primary lung tumour, chondrosarcoma: a case report. Diagn Cytopathol. 1991;7:423-426.
312 Dodd LG, Chai C, McAdams HP, et al. Fine needle aspiration of osteogenic sarcoma metastatic to the lung. A report of four cases. Acta Cytol. 1998;42:754-758.
313 Neimann TH, Bottles K. Cytologic diagnosis of metastatic epithelioid sarcoma, a cytologic mimic of squamous cell carcinoma. Am J Clin Pathol. 1993;100:171-173.
314 McKenzie CA, Philips J. Malignant phyllodes tumor metastatic to the lung with osteogenic differentiation diagnosed on fine needle aspiration biopsy. A case report. Acta Cytol. 2002;46:718-722.
315 Logrono R, Wojtowycz MM, Wunderlich DW, et al. Fine needle aspiration cytology and core biopsy in the diagnosis of alveolar soft part sarcoma presenting with lung metastases. A case report. Acta Cytol. 1999;43:464-470.
316 Thunnissen FB, Arends JW, Buchholtz RTF, et al. Fine needle aspiration cytology of inflammatory pseudotumour of the lung (plasma cell granuloma). Report of 4 cases. Acta Cytol. 1989;33:917-921.
317 Machicao CN, Sorensen KL, Abdul-Karim FW, et al. Transthoracic fine needle aspiration in inflammatory pseudotumor of the lung. Diagn Cytopathol. 1989;5:400-403.
318 Hannah CD, Oliver DH, Liu J. Fine needle aspiration biopsy and immunostaining findings in an aggressive inflammatory myofibroblastic tumor of the lung: a case report. Acta Cytol. 2007;51(2):239-243.
319 Gleason BC, Hornick JL. Inflammatory myofibroblastic tumours: where are we now? J Clin Pathol. 2008;61(4):428-437.
320 Hosler GA, Steinberg DM, Sheth S, et al. Inflammatory pseudotumor: a diagnostic dilemma in cytopathology. Diagn Cytopathol. 2004;31(4):267-270.
321 Hughes JH, Young NA, Wilbur DC, et alCytopathology Resource Committee, College of American Pathologists. Fine-needle aspiration of pulmonary hamartoma: a common source of false-positive diagnoses in the College of American Pathologists Interlaboratory Comparison Program in Nongynecologic Cytology. Arch Pathol Lab Med. 2005;129(1):19-22.
322 Arrigoni MG, Woolner LB, Bernatz PE, et al. Benign tumors of the lung. A ten-year surgical experience. J Thorac Cardiovasc Surg. 1970;60(4):589-599.
323 Fletcher JA, Longtine J, Wallace K, et al. Cytogenetic and histologic findings in 17 pulmonary chondroid hamartomas: evidence for a pathogenetic relationship with lipomas and leiomyomas. Genes Chromosomes Cancer. 1995;12(3):220-223.
324 Gal AA, Nassar VH, Miller JI. Cytopathologic diagnosis of pulmonary sclerosing hemangioma. Diagn Cytopathol. 2002;26:163-166.
325 Leong AS, Chan KW, Seneviratne HS. A morphological and immunohistochemical study of 25 cases of so-called sclerosing haemangioma of the lung. Histopathology. 1995;27(2):121-128.
326 Devouassoux-Shisheboran M, Hayashi T, Linnoila RI, et al. A clinicopathologic study of 100 cases of pulmonary sclerosing hemangioma with immunohistochemical studies: TTF-1 is expressed in both round and surface cells, suggesting an origin from primitive respiratory epithelium. Am J Surg Pathol. 2000;24:906-916.
327 Wong PW, Stefanec T, Brown K, et al. Role of fine-needle aspirates of focal lung lesions in patients with hematologic malignancies. Chest. 2002;121:527-532.
328 Gattuso P, Castelli MJ, Peng Y, et al. Post-transplant lymphoproliferative disorders: a fine needle aspiration study. Diagn Cytopathol. 1997;16:392-395.
329 Yokoyama S, Hayashida Y, Nagahama J, et al. Pulmonary blastoma: a case report. Acta Cytol. 1992;36:293-297.
330 Lee KG, Cho NH. Fine needle aspiration cytology of a pulmonary adenocarcinoma of fetal type: report of a case with immunohistochemical and ultrastructural studies. Diagn Cytopathol. 1991;7:408-414.
331 Gelven PL, Hopkins MA, Green CA, et al. Fine needle aspiration cytology of pleuropulmonary blastoma: case report and review of the literature. Diagn Cytopathol. 1997;16:336-340.
332 Husain M, Nguyen GK. Cytopathology of granular-cell tumor of the lung. Diagn Cytopathol. 2000;23:294-295.
333 Kintanar EB, Giordano TJ, Thompson NW, et al. Granular-cell tumor of trachea masquerading as Hurthle-cell neoplasm on fine-needle aspirate: a case report. Diagn Cytopathol. 2000;22:379-382.
334 Gomez-Aracil V, Mayayo E, Alvira R, et al. Fine needle aspiration cytology of primary pulmonary meningioma associated with minute meningothelial-like nodules. Report of a case with histologic, immunohistochemical and ultrastructural studies. Acta Cytol. 2002;46:899-903.
335 Whitaker D, Shilkin KB, Sterrett GF. Cytological appearances of malignant mesothelioma. Malignant mesothelioma. New York: Hemisphere; 1992. pp. 167–82
336 Whitaker D, Sterrett GF, Shilkin KB. Mesotheliomas. In Gray W, editor: Diagnostic cytopathology, 2nd ed, Edinburgh: Churchill Livingstone, 2003.
337 Nguyen GK, Akin MR, Villanueva RR, et al. Cytopathology of malignant mesothelioma of the pleura in fine-needle aspiration biopsy. Diagn Cytopathol. 1999;21:253-259.
338 Yu GH, Soma L, Hahn S, et al. Changing clinical course of patients with malignant mesothelioma: implications for FNA cytology and utility of immunocytochemical staining. Diagn Cytopathol. 2001;24:322-327.
339 Singh HK, Silverman JF, Berns L, et al. Significance of epithelial membrane antigen in the work-up of problematic serous effusions. Diagn Cytopathol. 1995;13:3-7.
340 Dusenbery D, Grimes MM, Frable WJ. Fine needle aspiration cytology of localised fibrous tumor of pleura. Diagn Cytopathol. 1992;8:444-450.
341 Caruso RA, LaSpada F, Gaeta M, et al. Report of an intrapulmonary solitary fibrous tumor: fine needle aspiration cytologic findings, clinicopathological and immunocytochemical features. Diagn Cytopathol. 1996;14:64-67.
342 Sironi M, Declich P, Di Bella C, et al. Solitary fibrous tumour of the pleura: a cytohistological and immunohistochemical case study. Cytopathol. 1996;7:274-278.
343 Ali SZ, Hoon V, Hoda S, et al. Solitary fibrous tumor. A cytologic-histologic study with clinical, radiological and immunohistochemical correlations. Cancer. 1997;81:116-121.
344 Apple SK, Nieberg RK, Hirschowitz SL. Fine needle aspiration biopsy of solitary fibrous tumor of the pleura. A report of two cases with a discussion of diagnostic pitfalls. Acta Cytol. 1997;41:1528-1533.
345 Clayton AC, Salomao DR, Keeney GL, et al. Solitary fibrous tumor: a study of cytologic features of six cases diagnosed by fine-needle aspiration. Diagn Cytopathol. 2001;25:172-176.
346 Bishop JA, Rekhtman N, Chun J, et al. Malignant solitary fibrous tumor: cytopathologic findings and differential diagnosis. Cancer Cytopathol. 2010;118(2):83-89.
347 Silverman JF, Berns LA, Holbrook CT, et al. Fine needle aspiration cytology of primitive neuroectodermal tumors. A report of three cases. Acta Cytol. 1992;36:543-550.
348 Folpe AL, Hill CE, Parham DM, et al. Immunohistochemical staining of FLI protein expression: A study of 132 round cell tumors with emphasis on CD-99 positive mimics of Ewing’s sarcoma / primitive neuroectodermal tumor. Am J Surg Pathol. 2000;24:1657.
349 Hummel P, Yang GC, Kumar A, et al. PNET-like features of synovial sarcoma of the lung: a pitfall in the cytologic diagnosis of soft-tissue tumors. Diagn Cytopathol. 2001;24:283-288.
350 Kim HS, Lee HJ, Cho SY, et al. Myasthenia gravis in ectopic thymoma presenting as pleural masses. Lung Cancer. 2007;57(1):115-117.
351 Goel A, Gupta SK, Dey P, et al. Cytologic spectrum of 227 fine-needle aspiration cases of chest-wall lesions. Diagn Cytopathol. 2001;24:384-388.