The Lymphatic System
The lymphatic system develops embryologically from the venous system. Two major theories exist of the embryologic origin of the lymphatic system: the centrifugal, or venous budding, theory and the centripetal theory. The centrifugal theory states that the lymphatic endothelium develops from the venous endothelium; the centripetal theory states that both systems (venous and lymphatic) develop from undifferentiated (stem type) mesenchymal cells. Advances in lymphangiogenesis research may clarify which theory is correct; this information will have a great impact on genetic research and the eventual molecular treatment of lymphangiodysplasias.92
The interstitial fluid that remains after the extracellular fluid is resorbed via the veins is taken up by the initial lymphatic vessels, into larger collecting vessels, into lymphatic trunks, and back into the right side of the heart via the lymphatic ducts that empty into the subclavian veins in the neck. This is a regional system that moves fluid from the periphery to the central circulation. It is designed to help maintain fluid balance in the tissues, fight infection, and assist in the removal of cellular debris and waste products from the extracellular spaces. In many ways, it functions like the “sanitation” system of a major city. It is largely ignored and goes unnoticed until it is disrupted and the “garbage” (in the form of lymphedema) piles up. This drainage system is separate from the general circulatory system but is the conduit for returning tissue fluids to the circulatory system.42,87
The cardiovascular system is a one-cycle system of vessels with a pump (the heart) to move the fluid (blood) through arteries, capillaries, and cells and then back to the heart via the veins. Fluid that leaves the arterial side of the capillary bed in a process called ultrafiltration nourishes the tissues and cells.
Of the fluid volume that perfuses the tissues, 90% reenters the circulation via the venous capillary network (reabsorption) because of differences in concentration of fluid and protein in the tissues and in the venous end of the capillary network. The remaining 10% of extracellular tissue fluid and plasma proteins in the interstitial spaces must be returned to the heart via the lymphatic system.
Most protein molecule’s diameters are too large to pass through openings in the endothelium of the venous capillaries. A small amount of protein, if broken down into smaller molecules by macrophages, can pass through the open junctions in the venous endothelium. However, the majority of extracellular protein must be transported via the lymphatic system. Although 10% of the total fluid volume seems small, it can amount to 2 L/day.
The lymphatic system is a pressure-driven system based on the principles of osmotic diuresis. If the normal lymphatic transport mechanisms are disrupted (e.g., by scar tissue or reduced muscle pumping), significant accumulations of water and protein can remain in the tissue spaces, resulting in latent, acute, or chronic lymphedema. This protein is a result of cellular metabolism and is not related to protein ingested from food.
The dynamics of fluid exchange in the tissues are controlled by the microcirculation unit consisting of the arterial and venous capillaries, the tissue channels, the proteolytic cells (macrophages) in the tissues, and the initial lymphatics (see the description of initial lymphatics in the next section).
ANATOMY AND PHYSIOLOGY
The lymphatic system is composed of superficial and deep lymph vessels and nodes. Other lymphatic organs and tissues include the thymus, bone marrow, spleen, tonsils, and Peyer’s patches of the small intestine. These perform important immune functions discussed in Chapter 7. Superficial vessels rely on an interaction of oncotic and hydrostatic pressures, muscle contraction, arterial pulsation, and gentle movement of the skin to move lymph fluid, whereas the deeper vessels, which generally parallel the venous system, contain smooth muscle and valves and help prevent backflow.19
The lymphatic vessel network is an intricate one-way vessel system that serves to drain the 10% excess tissue fluid volume and plasma proteins that remain in the interstitium after normal capillary perfusion/filtration has taken place and return it to the central circulation via the large veins in the neck. All lymph fluid eventually passes through lymph nodes before emptying into the right lymphatic duct and the thoracic duct. The fluid is then returned to the bloodstream via the left and right subclavian veins.42,87
The anatomy of the lymphatic vessel system can be compared in some ways to the vein system on the leaves and stems of trees. The smallest vessels, or veins, are at the periphery of the tissues (leaves), and the diameter of these vessels gradually increases in the stem of the leaf as the system progresses into larger and larger vessels (corresponding to deeper tissues) and continues to progress to larger stems and branches of the tree until the trunk is reached.
The smallest of lymphatic vessels (diameter 20 to 40 μm), called lymphatic capillaries or precollectors, begin as blind-ended sacs of endothelium just under the epidermis.19,32,42 These are referred to as the initial lymphatics and are in close proximity with the venous and arterial capillaries (Fig. 13-1). Terminology has changed over the years; the reader should be aware that initial lymphatic and lymphatic capillary refer to the same structure.
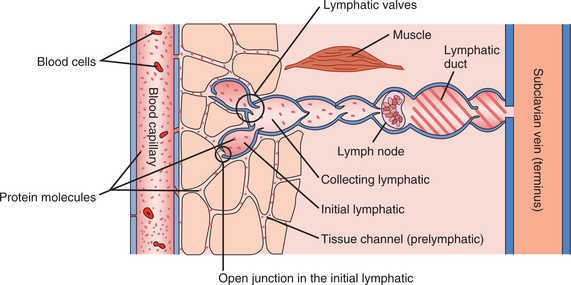
Figure 13-1 Anatomy of the lymphatic vessel system (schematic). This diagram shows the passage of protein (dots) in normal tissue from the blood capillary, through the tissue channels, into and through the lymphatic system, back to the venous system, and eventually emptying into the subclavian vein. Note that the protein molecules are not on the venous side of the diagram because for the most part, these molecules are too large to pass through the openings in the venous endothelium. Also note that this is a schematic diagram and not drawn to scale; the lymphatic duct depicted emptying into the venous system (subclavian vein) is much deeper (under the fascia) than this two-dimensional illustration can portray. Various malfunctions are illustrated in Fig. 13-15. (From Casley-Smith JR, Casley-Smith JR: Modern treatment for lymphoedema, ed 5, Adelaide, Australia, 1997, Lymphoedema Association of Australia.) Lymphoedema Association of Australia
The vessel walls of these initial lymphatics are one cell thick, formed by overlapping endothelial cells with many loose junctions between cells opening and closing (Fig. 13-2). This action allows for movement of water and proteins into the vessel and prevents the escape of protein into the interstitium during the compression of the initial lymphatics.
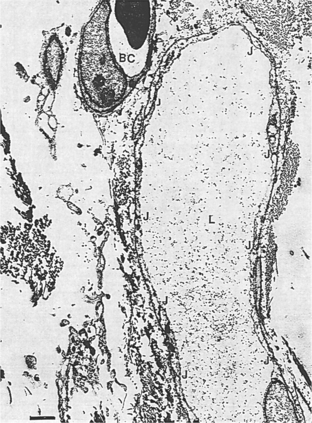
Figure 13-2 An initial lymphatic (L) in a quiescent (at rest or inactive) tissue. Many closed (narrow or tight) junctions (J) are evident. A blood capillary (BC) is shown for comparison of size, endothelial opacity, and other characteristics. The bar at the lower left (1 μm) is provided to give the viewer size perspective. (From Casley-Smith JR, Casley-Smith JR: Modern treatment for lymphoedema, ed 5, Adelaide, Australia, 1997, Lymphoedema Association of Australia. Modified from Casely-Smith JR: Br J Exp Path 46:35-49, 1965.) Lymphoedema Association of Australia
These cells are also in direct contact with the microfilaments of the surrounding connective tissues. They are connected to the tissue matrix by anchoring filaments that act as “guide wires” to pull the cell junctions open when the tissue pressure rises as a result of increased extracellular fluid volume19 (Fig. 13-3). These vessels are arranged in a meshlike plexus; for every square millimeter of tissue, 7 mm of lymphatics are available to drain it.18
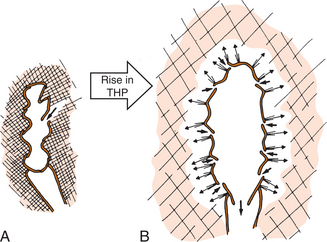
Figure 13-3 Effects of elevated tissue hydrostatic pressure (THP) on initial lymphatic functioning. A, Normal lymphatic vessel at a fairly low tissue hydrostatic pressure and normal lymphatic drainage. B, Tissue response to a tremendous increase in tissue hydrostatic pressure (represented by the large arrow). The swelling in the interstitial tissues pulls on the anchoring filaments, pulling and holding open the initial lymphatic endothelial junctions (thin arrows pointing outward), allowing fluid to pour into the initial lymphatic in an attempt to reduce edema. In this way, in place of a few widely open junctions, there are many slightly open ones, through which fluid is forced (thicker arrows directed inward) down a hydrostatic pressure gradient. (From Casley-Smith JR, Casley-Smith JR: Modern treatment for lymphoedema, ed 5, Adelaide, Australia, 1997, Lymphoedema Association of Australia.) Lymphoedema Association of Australia
The initial lymphatics function as force-pumps powered by variations in total tissue pressure caused by movement, muscular contraction, respiration, and variations in external pressure caused by massage, gravity, change in position, and other similar factors. Without changes in total tissue pressure, these force-pumps cannot function, and fluid will accumulate in the interstitium, leading to edema.
Microcirculation Unit
A brief discussion of Starling’s theory of fluid dynamics is helpful to understand the microcirculation unit. In 1897, Starling proposed the mechanism governing fluid flow out of the blood capillaries into the tissues and back into the capillaries again. There are four pressures that are important in Starling’s Law: (1) plasma hydrostatic pressure, the pressure inside the capillaries that decreases as fluid passes from the arterial to the venous side of the capillary loop; (2) tissue hydrostatic pressure, the pressure of fluid in the tissue channels (usually negative or less than atmospheric pressure) that tends to pull fluid from the capillaries into the tissues*; (3) plasma colloidal osmotic pressure, caused by plasma proteins, this pressure causes a siphon effect and fluid is pulled into the capillaries; and (4) tissue colloidal osmotic pressure, the pressure caused by plasma proteins in the tissues that causes a net movement of fluid into the tissues. All of these pressure systems determine how much fluid moves and where it moves within the body.
The laws of basic fluid dynamics dictate that fluid flows from an area of high pressure to an area of lower pressure until equilibrium is reached. Starling’s law simplified means that fluid at the arterial end of the capillary will tend to flow into the tissue spaces because plasma (blood) hydrostatic pressure is higher at the arterial end compared with the tissue hydrostatic pressure (THP) of the tissues.
If all else is “normal,” when the fluid reaches the venous side of the capillary, the plasma hydrostatic pressure will be lower than the plasma colloid (protein) osmotic pressure, and the fluid will be forced back into the venous side of the capillary, which accounts for about 90% of the fluid on the venous end of the capillary. Ten percent of net-ultrafiltrate must return to the central circulation via the lymphatics. If all is normal there, the initial lymphatics will take up that fluid, move it to the collecting lymphatics and larger lymphatic trunks, through regional lymph nodes, and eventually into the right lymphatic duct or the thoracic duct, and back into the vena cava (see Fig. 13-8).
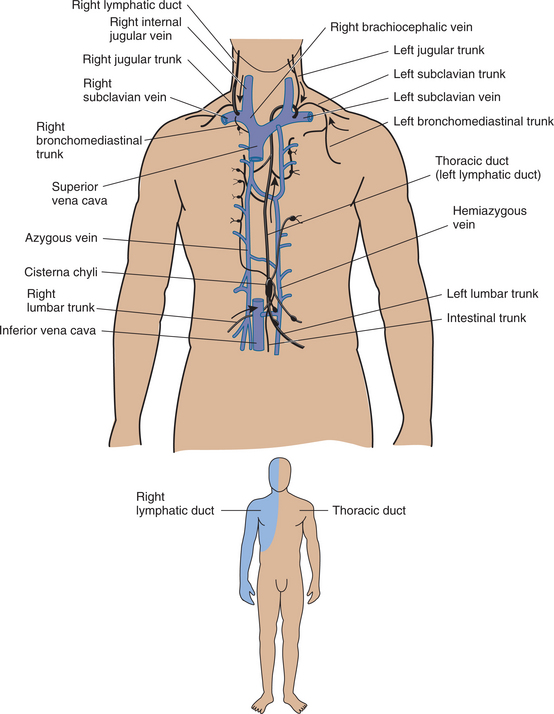
Figure 13-8 Lymphatic ducts. The thoracic duct (black), leading from the cisterna chyli to discharge into the left subclavian vein, in the neck. (The blood vessels are shaded blue.) The right lymphatic duct is also shown (see figure on bottom). This carries far less lymph than the thoracic duct, draining mainly the right arm and head, the heart and lungs, and the anterior chest wall. These two main trunks sometimes are linked by large collateral lymphatics. (From Casley-Smith JR, Casley-Smith JR: Modern treatment for lymphoedema, ed 5, Adelaide, Australia, 1997, Lymphoedema Association of Australia. Inset, Jarvis C: Physical examination and health assessment, ed 3, Philadelphia, 2000, WB Saunders.)
In the presence of lymphatic dysfunction, some part or all of the 10% of fluid volume and the proteins will remain trapped in the tissue spaces and cause lymphedema. With lymphedema, the challenge for the therapist is to effectively move that fluid back into functioning lymphatics and then into the central circulation. This model is a very gross simplification but can be helpful in understanding the basics of fluid dynamics in the capillary loop. Many other factors affect the tissues in addition to those mentioned previously.19,90
Deeper in the dermis are precollectors (Fig. 13-4), which flow into collecting lymphatics located in the subcutaneous tissue (Fig. 13-5). The true collecting vessels have valves to prevent backflow and some muscle tissue in their walls to further enhance their pumping action.19,90 Extrinsic muscle contraction or lymphatic drainage massage also increases this pumping action.
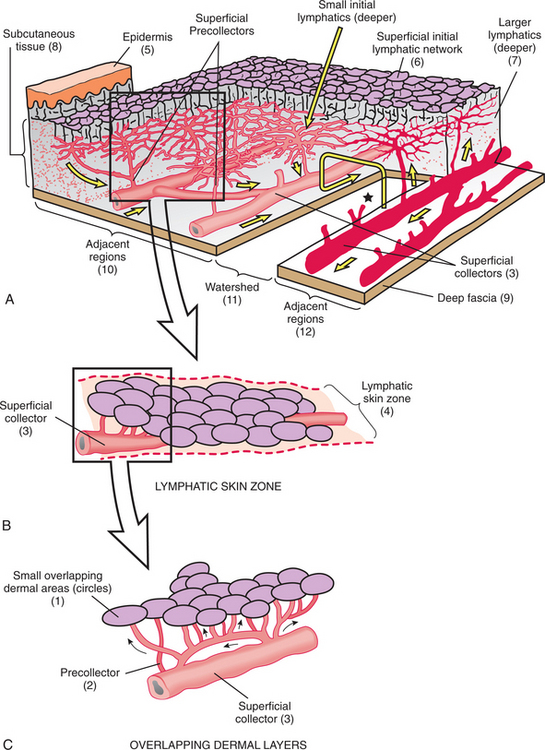
Figure 13-4 Overview of the lymphatic drainage system. A, Overview of the lymphatic drainage paths from a skin region. The epidermis (5) is superficial to a superficial initial lymphatic network (6), which sends blindly ending vessels into the dermis and which is linked to the deep dermal plexus of larger initial lymphatics (7), in the subcutaneous tissue (8), by many connections. The superficial collecting lymphatics (3), which discharge into the larger ones (not shown), lie next to the deep fascia (9). A watershed (11) lies between two adjacent regions (10 and 12), which drain in opposite directions (medium arrows). One of these is obstructed (red vessels). The deep and superficial initial lymphatic plexuses overlap across this watershed. These groups of cross-connections provide collateral drainage and are enlarged by manual massage. The large, U-shaped arrow (*) shows this path. B, Lymphatic skin zone (4) that extends along the length of a superficial collector (3). Certain areas of the skin drain into a specific superficial collector, which accounts for the clinical observation of lymphedema in portions of an extremity (e.g., pockets of extra swelling or asymmetric edema). When a specific superficial collector is blocked (or if the deep collector into which it drains is blocked), the result is edema at that site. C, Shows (1) small overlapping dermal areas (circles), which drain into networks of initial lymphatics (not shown), which drain into small collecting lymphatics called precollectors (2) and then to larger superficial collectors (3). (From Casley-Smith JR, Casley-Smith JR: Modern treatment for lymphoedema, ed 5, Adelaide, Australia, 1997, Lymphoedema Association of Australia. Modified from Földi M, Kubik S: Lehrbuch der lymphologie fur mediziner und physiotherapeuter mit anhang: praktische linweise fur die physiotherape, Stuttgart, Germany 1989, Gustav Fischer Verlag.) Lymphoedema Association of Australia
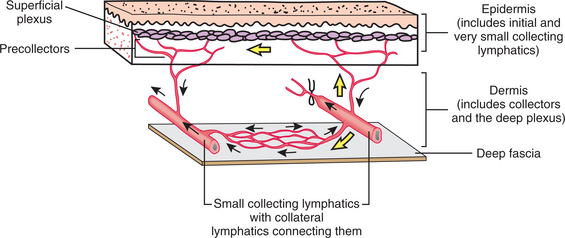
Figure 13-5 Collecting lymphatics. Lymphatics traverse through the epidermis, dermis, and deep fascia via lymphatics that increase in size as they go deeper into the tissues. Two layers of lymphatic plexuses are in the skin: the epidermis and dermis (layer just below the epidermis, formerly called the corium), which contains blood and lymphatic vessels, nerves, and nerve endings, glands, and hair follicles. Lymphatic vessels in the dermal layer can divert fluid from a blocked area and drain it into normally functioning area(s). In this illustration, one of the two larger collectors (right) is blocked; note the watershed between the blocked and the open collecting lymphatic. The lymph that would normally be transported along this blocked collector instead passes up into the superficial plexus and down into the deeper plexus formed by collaterals in the watershed area located just above the deep fascia. In these, the lymph passes to the nonblocked collector (left) and then drains into larger lymph vessels (not shown). When edema exists, the valve flaps in the collaterals are dilated and do not meet, thereby allowing lymph to move in either direction across these vessels (i.e., across the watershed). (From Casley-Smith JR, Casley-Smith JR: Modern treatment for lymphoedema, ed 5, Adelaide, Australia, 1997, Lymphoedema Association of Australia. Modified from Földi M, Kubik S: Lehrbuch der lymphologie fur mediziner und physiotherapeuter mit anhang: praktische linweise fur die physiotherape, Stuttgart, Germany 1989, Gustav Fischer Verlag.) Lymphoedema Association of Australia
Collecting lymphatics do not form a plexus, but there can be connections between them. Their diameter gradually increases in size to form the lymph trunks, which lie near the deep fascia. Each segment of collecting lymphatic vessels between valves is called a lymphangion (Fig. 13-6). The muscle in the collecting lymphatic walls contracts rhythmically. Smooth muscle cells around the endothelial cell layer face the lumen of the vessel. These are innervated by the autonomic nervous system and contract on the average of 5 to 10 times per minute.93 This “lymphangiomotoricity” combines with the contraction of the lymphangion itself, which is triggered by distention of the vessel wall. The greater the stretch, the greater the force of the contraction. If many lymphangions contract at once and outflow is obstructed (e.g., by scarred or radiated lymph nodal areas), pressure inside the vessel can reach as high as 100 mm Hg.
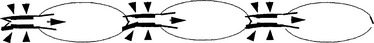
Figure 13-6 The lymphangion. Many lymphangions may contract at once, but sometimes only one lymphangion is triggered. The pressure exerted by each lymphangion is usually a few mm Hg but can be over 100 mm Hg if outflow is obstructed and many units are contracting at once. Contraction is triggered by distention (i.e., greater filling creates greater force) but can be modified by humoral (including medications) and nervous factors. Pumping is greatly aided by varying total tissue pressure (TTP) (e.g., from adjacent muscles, respiration, or manual lymphatic drainage [MLD]), as previously mentioned in the text. (From Casley-Smith JR, Casley-Smith JR: Modern treatment for lymphoedema, ed 5, Adelaide, Australia, 1997, Lymphoedema Association of Australia.) Lymphoedema Association of Australia
High intravascular pressure fatigues the muscle wall, leading to ineffective smooth muscle contraction and ultimately to vessel failure. The walls dilate, preventing closure of valve flaps, and a backflow of lymph distal to the site of obstruction occurs, causing lymphedema. This is one plausible explanation for the fact that many individuals with a “limb at risk” develop lymphedema months or even years after their original surgery. For a time, the remaining lymphatics function marginally without evidence of clinical lymphedema, but these units become overtaxed, eventually the walls fatigue, and latent lymphedema progresses to acute and then to chronic lymphedema.
The lymph trunks in the extremities join into the larger lymph vessels of the trunk, which join to form the thoracic duct and the right lymphatic duct that pump the lymph into the central circulation at the left and right subclavian veins in the root of the neck. The lymphatic vessels are embedded in fatty tissue and accompany the chains of lymph nodes along the blood vessels.32 This explains why injury to blood vessels in an area implies injury to lymphatic vessels in that area, too, regardless of whether it is “unexpected” or “controlled” trauma, as in surgery.
As lymph flows from the periphery to the root of the limbs to the center of the body, it passes through many lymph nodes, which act as filters to cleanse the lymph of waste products and cellular debris. Vessels distal to nodes are called afferent lymph vessels. Vessels leaving lymph nodes for more proximal points are called efferent lymph vessels.
Lymph nodes also produce lymphocytes and macrophages, which are critical for immune function; destroy foreign bacteria, harmful viruses, and cancer cells; and filter waste products. Lymph nodes offer 100 times the normal resistance to flow of lymph within the lymphatic vessels themselves, which explains why they are often the sites of obstruction in lymphatic dysfunction.19,40,42,90
Lymphatic Territories and Watersheds
The anatomy of the lymphatic system is a regional one. The body is divided into a series of lymph drainage territories called lymphotomes, which are bordered and separated by so-called watershed areas. The watershed areas are characterized by sparse collateral flow to adjacent lymphotomes,11,41 but connections exist between lymphotomes in the superficial and deep plexuses and via collateral lymphatics between deep collectors in adjacent lymphotomes located just above the deep fascia. Under normal conditions, the lymph drains in different directions on either side of these watersheds (Fig. 13-7).
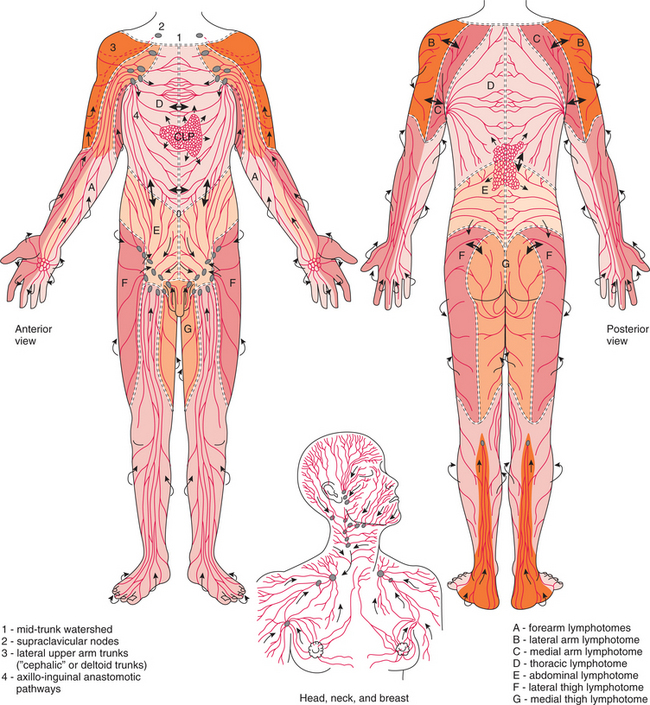
Figure 13-7 Regional lymphatic system. The dermal and subcutaneous lymph territories (lymphotomes are indicated by different shadings) of the lymphatic system are separated by watersheds marked by (= = = =). Arrows indicate the direction of the lymph flow. Normal drainage is away from the watershed, but collaterals cross the watershed (thick double arrows). When the main drainage paths from each of these regions are blocked, lymph (thick single arrows) has to be carried across the watersheds via collaterals and the plexuses. The cutaneous lymphatic plexus (CLP) is shown in the center of the chest only. It is filled from the tissues and covers the entire body; this is not shown to avoid confusion. These initial lymphatics fill superficial collectors, which drain into deep ones and then into the lymphatic trunks (small arrows). The lymphotome of the external genitals and perineum is shown but unlabeled. (From Casley-Smith JR, Casley-Smith JR: Modern treatment for lymphoedema, ed 5, Adelaide, Australia, 1997, Lymphoedema Association of Australia. Modified from Földi M, Kubik S: Lehrbuch der lymphologie fur mediziner und physiotherapeuter mit anhang: praktische linweise fur die physiotherape, Stuttgart, Germany 1989, Gustav Fischer Verlag.) Lymphoedema Association of Australia
The trunk can be divided into four quadrants: the left and right thoracic lymphotomes and the left and right abdominal lymphotomes. The left and right thoracic lymphotomes drain into the ipsilateral axilla, as do the left and right upper extremities. Some individuals possess an auxiliary drainage pathway from the lateral aspect of the upper arm called the deltoid-pectoral or cephalic chain. This pathway drains directly into the ipsilateral subclavian nodal area, bypassing the axilla entirely. If present, an individual may be less likely to develop upper extremity lymphedema secondary to axillary disruption (surgical or by radiation), as this pathway may provide sufficient lymph transport capacity for the upper extremity. This pathway can be disrupted by supraclavicular radiation therapy sometimes used to treat recurrent cancer of the chest wall.54
The left and right abdominal lymphotomes drain into the left and right superficial inguinal nodes, respectively. Each leg and corresponding half of the lumbar, gluteal, and genital region drains to the ipsilateral superficial inguinal nodes. From there, fluid drains into the deep inguinal, pelvic, and abdominal nodes, into the cisterna chyli, the thoracic duct, and to the left subclavian vein19 (Fig. 13-8). Most of the lower leg drains via the femoral trunks, which run on the anterior thigh to the inguinal nodes, also draining the medial and lateral thigh lymphotomes. There is a small posterior lower leg lymphotome draining to the popliteal nodes by way of the dorsolateral trunks.
A midline watershed divides the head, neck, and face areas. The right side drains to the right cervical nodes and then to the right supraclavicular nodes; the left side drains to the left cervical nodes and then to the left supraclavicular nodes. The posterior aspect of the head and neck drains into the vertebral lymphatics that drain into the supraclavicular nodes on the ipsilateral side.19,41
INFLAMMATION AND INFECTION IN THE LYMPHATIC SYSTEM
Disorders of the lymphatic system may result from lymphangitis (inflammation of a lymphatic vessel); lymphadenitis (inflammation of one or more lymph nodes); lymphedema (an increased amount of lymph fluid in the soft tissues); or lymphadenopathy (enlargement of the lymph nodes). Lymph nodes act as defense barriers and are secondarily involved in virtually all systemic infections and in many neoplastic disorders arising elsewhere in the body.
The specific node, or nodes, affected in an infectious disease depends on the location of the infection, the nature of the invading organism, and the severity of the disease. For example, infections involving the pharynx, salivary glands, and scalp often cause tender enlargement of the neck nodes, referred to as reactive cervical lymphadenopathy. Generalized lymphadenopathy, enlargement of two or three regionally separated lymph node groups, is usually a result of inflammation, neoplasm, or immunologic reactions.
These two types of lymphadenopathy are normal reactions to infection that result in large and tender lymph nodes, but the node is not necessarily infected (warm or reddened, as with lymphadenitis). The presence of lymphadenopathy is usually more significant in people older than 50 years; lymphadenopathy in people under age 30 is usually due to benign causes, but this must be medically determined.
Lymphedema
Lymphedema is a swelling of the soft tissues that results from the accumulation of protein-rich fluid in the extracellular spaces. It is caused by decreased lymphatic transport capacity and/or increased lymphatic load and is most commonly seen in the extremities but can occur in the head, neck, abdomen, and genitalia.
Classification of Lymphedema
Lymphedema is divided into two broad categories: primary (idiopathic) and secondary (acquired) lymphedema. In the past, primary lymphedema was classified as connatal, if it appeared at birth; praecox if it appeared at puberty; or tarda if it developed after age 35. The term connatal (present from birth) applies to most primary lymphedemas that are present at birth, rather than the term congenital, which implies a specific genetic abnormality. The severity of lymphedema is graded using the scale from the International Society of Lymphology (ISL): Stage 0 or latent lymphedema, Stage I, Stage II, and lymphostatic elephantiasis Stage III8,30,33 (Box 13-1).
In Stage 0 or latent lymphedema, lymph transport is impaired, but there is no clinical evidence of swelling. Stage 0 may last months or years before any obvious lymphedema occurs. Understanding the concept of latent lymphedema is critical in providing guidance to individuals at risk, as well as in recognizing the early signs and symptoms of progression from Stage 0 to Stage 1. These may include a sensation of heaviness, fatigue, ache, or pain in the limb at risk.
Stage I lymphedema is soft, pits on pressure, and reverses with elevation. In the early stages, there is a chronic inflammatory response to the excessive protein in the interstitium. The subcutaneous tissues begin to fibrose, progressing the lymphedema from Stage I to II. In fact, a lymphedematous limb may be Stage II in the foot and ankle and Stage I in the thigh.
Stage II lymphedema is nonpitting and does not reduce on elevation of the limb, and clinical fibrosis is present. Skin changes, such as eczema, warts, papillomas, and lymph fistulae, are common in severe Stage II lymph- edema. Chronic inflammation can lead to recurrent bacterial and fungal infections.
The most severe, Stage III lymphedema, is referred to as lymphostatic elephantiasis. This is characterized by severe nonpitting, fibrotic edema with atrophic skin changes such as thickened, leathery, keratotic skin, skin folds with tissue flaps, warty protrusions, papillomas, and leaking lymph fistulae. Lymphangiomas (form of lymphangiectasia) may also be present.
Incidence
The exact incidence of primary lymphedema is unknown because many people remain undiagnosed or if diagnosed, treatment or follow-up care does not occur, and the condition remains unreported.55 Approximately 15% of primary lymphedemas are present at birth (formerly called connatal). The most common form of primary lymphedema occurs from adolescence to midlife and accounts for 75% of primary lymphedema in a 4: 1 ratio of females to males (formerly called lymphedema praecox). Of all primary lymphedema, 10% to 20% appears abruptly after age 35 (formerly called lymphedema tarda).19 A small percentage of primary lymphedemas occur in association with rare genetic syndromes, such as Milroy’s (appears at birth) and Meige’s (develops anywhere from early childhood to puberty) diseases, accounting for approximately 2% of primary lymphedemas.
The incidence of secondary lymphedema also remains an approximate figure. Lymphatic filariasis affects over 120 million people in 80 countries throughout the tropics and subtropics of Asia, Africa, the Western Pacific, and parts of the Caribbean and South America. Presently, no detailed maps are available of the geographical distribution of secondary lymphedema caused by filariasis, but distribution may be governed by climate, with an estimated 420 million people exposed to this infection in Africa in the year 2000.57
The WHO estimates 700,000 people in the Americas are affected today (including 400,000 in Haiti and 100,000 in the Dominican Republic). Clinical reports on the incidence of secondary lymphedema from other causes vary, with an estimated 3 million new cases in the United States each year; up to 30% of breast cancer survivors in the United States develop lymphedema sometime in their lifetime. The incidence increases after surgery and radiation when these procedures are combined.22,29,63
According to Armer, “perhaps in part because of differences in measurement and diagnosis, the reported incidence of lymphedema varies greatly among persons treated with surgery and radiation for breast cancer. Through increased measurement accuracy, lymphedema incidence and prevalence following current therapeutic approaches for breast cancer treatment, cancer will be better understood, and more informed decisions about risk factors, treatment interventions, and recovery will be made.”3
Etiologic Factors
The exact cause of primary lymphedema is unknown and cannot be linked to any significant traumatic event (Table 13-1). Primary lymphedema is most likely the result of lymphangiodysplasias or malformations of the lymphatic vessel present at birth (Fig. 13-9) but sometimes delayed in symptomatic presentation. Although a small percentage of primary lymphedemas are linked to genetic causes (e.g., Milroy’s disease and Meige’s syndrome), most cases are not genetically linked and are more likely the result of some developmental abnormality in the fetus. A family history of lymphedema is present in only 10% to 20% of all people with primary lymphedema.25
Table 13-1

HIV, Human immunodeficiency syndrome; AIDS, acquired immunodeficiency syndrome.
*Listed in approximate descending order.
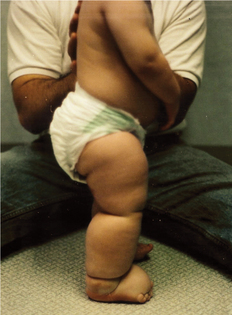
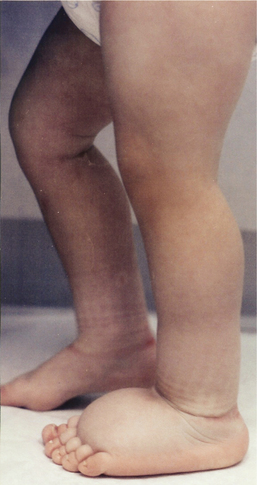
Figure 13-9 A 13-month-old with primary lymphedema of the right lower extremity, right buttock, and genital area since birth. (Courtesy Lymphedema Therapy, Woodbury, NY, 2006.)
Klippel-Trénaunay-Weber syndrome (KTWS) is a rare occurrence in embryonic development and is associated with numerous anomalies. These can include varicose veins, cavernous hemangioma of the skin, and hypertrophy of bones and soft tissues in one or several extremities. In addition, dysplasia of the lymphatic system and neurogenic and visceral vascular malformations can occur. The dysplasia of the lymphatics can result in lymphedema in the involved extremities15,90 (Fig. 13-10).
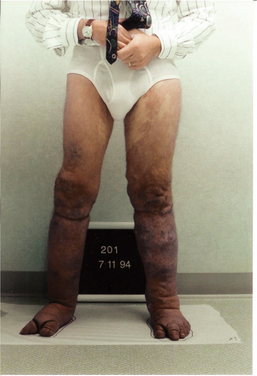
Figure 13-10 A 50-year-old man with Klippel-Trénaunay syndrome, a lymphangiodysplasia that caused lymphedema in both lower extremities. Note the skeletal abnormalities of the toes, the large hemangioma on the left thigh, and the venous varicosities in the lower legs. (Courtesy Lymphedema Therapy, Woodbury, NY, 2006.)
Malformations of the lymphatic vessels associated with primary lymphedema can be divided into three types: aplastic, hypoplastic, and hyperplastic. Aplasia occurs when the lymphatic collectors are so few that they are considered “absent.” Aplasia may also involve the absence of lymph capillaries that render the adequate collectors less functional. Aplasia is most often combined with hypoplasia; complete aplasia would result in tissues unable to support life.
Hypoplasia refers to less than the normal expected number of lymph collectors in the affected region and may also occur when collectors present are unable to function as transport vessels. Hypoplasia represents the most common cause of primary lymphedema, occurring in 75% of the cases. Hyperplasia accounts for 15% of primary cases and is characterized by grossly dilated and enlarged lymphatics that can become varicose. Hyperplasia can occur in the lymphatics of the superficial plexus of the skin or in the main lymph trunks. As a result of the overdilation of the vessels, the intralymphatic valve flaps do not seal, and a reflux of lymph occurs.
When this occurs in the mesenteric and intestinal lymphatics, a reflux of chyle to distal areas also takes place—that is, to the skin of the genitals, buttocks, and thighs, or to the knee joint (Fig. 13-11). Chyle is the protein-rich, milky fluid taken up by the intestinal lymphatics during digestion, consisting of lymph and triglyceride fat in a stable emulsion, and conveyed by the thoracic duct to empty into the venous system.
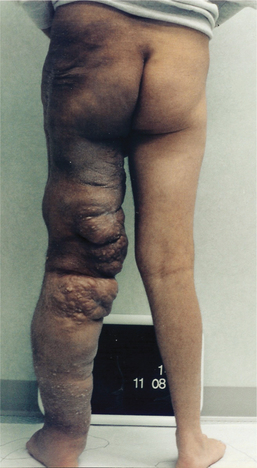
Figure 13-11 A 19-year-old male with primary lymphedema of the left lower extremity (onset age 8). Lymphedema progressed to involve the buttocks and genitalia after prolonged pneumatic compression pump usage. This young man had three microsurgical procedures (lymphovenous and lympholymphatic anastomoses) in an attempt to reduce the genital and extremity edema. In addition, he had two debulking surgeries, which also were unsuccessful in reducing the lymphedema. He developed chylous reflux and eventually had sclerotherapy to his leaking abdominal lymphatics, which was very successful in stopping the severe leakage of chyle from his scrotum, medial thigh, and buttock. The chyle-filled papules are visible on the posterior aspect of the thigh and calf. Notice the abnormal bulges and skin folds on the posterior thigh, which result when the debulked areas fill in with edema fluid. (Courtesy Lymphedema Therapy, Woodbury, NY, 2006.)
Lymphangiectasia refers to lymphatic hyperplasia in a deeper organ or localized area of a limb. Lymphangiomas and lymph cysts are forms of lymphangiectasia.
Secondary lymphedema occurs as the result of damage to otherwise normal lymphatic vessels or nodes from a known entity. The most common cause of secondary lymphedema worldwide is filariasis. Filariasis is a parasitic infection carried by mosquitoes in endemic regions, often found in tropical climates (Africa, South America, India, and Malaysia). The larvae of the worm are injected into the dermis with the mosquito bite. They pass into the initial lymphatics and larger collecting lymphatics and can grow to 20 cm in length and 1 to 2 cm in diameter as they mature into the adult worm forms. The adult male has a long tail that whips back and forth, damaging the fragile lymphatic walls.
The greatest damage, however, is done after the worm dies, often 5 to 10 years after the initial infection. At that time, foreign proteins from the worm body cause severe local inflammatory reactions leading to severe fibrosis and scarring of the tissues, totally blocking the larger lymph collectors. This total blockage results in massive swelling distal to that collection site.44,77,90
In the United States and other regions of the world where the filaria parasite does not exist or has been eradicated, the most common cause of secondary lymphedema is invasive procedures used in the diagnosis and treatment of cancer. Regional lymph node dissection for diagnostic staging and eradication of tumor sites disrupts the lymphatic system. Radiation therapy, reconstructive or other surgical procedures, or the combination of these procedures are well-known contributing factors to the development of lymphedema.
Local radiation treatment after surgery for cancer increases the incidence of secondary lymphedema three times that of surgery alone, probably a result of the increase in local tissue fibrosis that further impairs lymph flow through the remaining functioning lymphatic vessels.19 If there is significant burning and blistering of the skin during radiation treatment, the risk of lymphedema is increased because of the decreased elasticity of the skin and subcutaneous tissues.75
Other causes of secondary lymphedema include bacterial or viral infection; multiple abdominal surgeries, particularly in the obese individual; any trauma or surgery that impairs the lymphatics; or repeated pregnancies (see Table 13-1). Liposuction, done for cosmetic reasons to enhance appearance, when performed on an individual with an asymptomatic but marginal lymphatic system can trigger lymphedema in the operative limb.
Crush injuries, compound fractures, or severe lacerations or degloving injuries to the skin can significantly impair lymph flow. These types of injuries are also usually associated with damage to blood vessels. The damaged blood vessels leak fibrinogen, blocking the tissue channels and initial lymphatics and thus contributing to the development of lymphedema. Other known causes that have been reported as associated with secondary lymphedema include paralysis, lipedema, skin thinned by cortisone (sometimes referred to as cortisone skin), and acquired immunodeficiency syndrome (AIDS), particularly if Kaposi’s sarcoma is present.19
Some medications used in the treatment of breast cancer (e.g., tamoxifen or Adriamycin) have been associated with peripheral thrombophlebitis. These medications can cause blood clots resulting in deep vein thrombosis (DVT), venous insufficiency, and eventual secondary lymphedema.
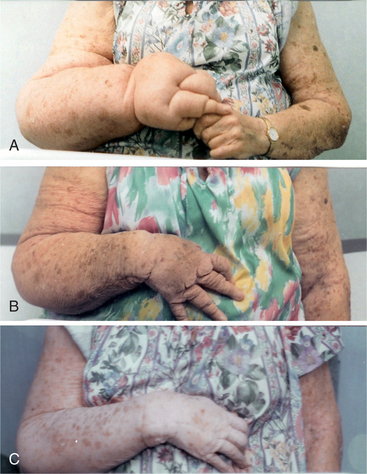
Surgery is “controlled trauma,” but it is trauma nevertheless; the more extensive the procedure, the more extensive is the trauma. Surgery in individuals with a marginal lymphatic system (where the lymph transport capacity equals the normal lymph load) can cause enough of an overload to trigger lymphedema. For example, an individual undergoing a triple coronary artery bypass graft (CABG) procedure with donor veins taken from the legs may develop chronic leg edema that is often misdiagnosed as venous insufficiency or “cardiac related.” This is particularly true in the older, obese individual who has poor functional mobility. If the diagnosis of lymphedema is delayed or never made and is not addressed, the individual may not be able to succeed in postoperative rehabilitation (Fig. 13-12).
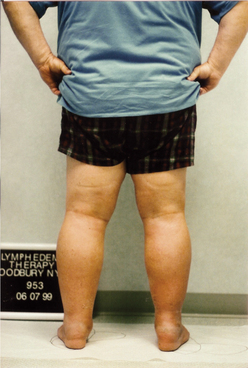
Figure 13-12 A 63-year-old man with lymphedema of both lower extremities, left greater than right. The swelling developed in the left leg 1 year after a coronary artery bypass graft (CABG) procedure in which veins were harvested from the left leg. The swelling began in the right leg, worsened in the left leg, and progressed into the abdomen after radium seed implantation for prostate cancer. This man’s marginal lymphatic system was overwhelmed by the “trauma” caused by the CABG procedure and the radium seed implantation procedure. (Courtesy Lymphedema Therapy, Woodbury, NY, 2006.)
In the past, most health care professionals knew about upper limb lymphedema secondary to axillary dissection for breast cancer, but many were not aware that an individual who had a CABG procedure; inguinal node dissection for melanoma of the foot, testicular cancer, or prostate cancer; or pelvic/abdominal node dissection for gynecologic cancer was at risk for lymphedema of the leg.
Anyone with postoperative leg edema after a fracture or total hip or knee replacement would not have been routinely evaluated for lymphedema 10 years ago. The combination of venous edema and lymphedema is often overlooked in the management of edema secondary to trauma. Many cases of chronic edema with recurrent infection and skin ulcerations are treated as pure venous edemas with poor results because the lymphatic component of the edema is not addressed.
Pathogenesis
Lymphedema by definition is a low flow edema that occurs when the lymph transport capacity is inadequate to transport the normal volume of lymph. It is a failure of the safety valve mechanisms (Fig. 13-13). This can occur when the lymph load is normal, but the lymph transport capacity is inadequate (decreased absorption of lymph in lymph nodes) or when there is an increase in the lymphatic load (e.g., fluid entering the tissues) and the transport capacity is inadequate (subtotal lymphatic blockage).
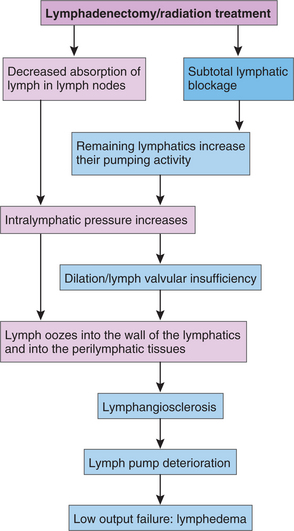
Figure 13-13 Pathogenesis of lymphedema. This flow chart follows the progression from an “at-risk” or latent phase of lymphedema to acute lymphedema after lymphadenectomy and/or radiation treatment to the nodal area. (Courtesy Dr. Michael Földi, June 1999.)
In reality, the body adjusts the load if the capacity alters in response to changes in tissue hydrostatic pressure and other changes in homeostasis (remaining lymphatics increase their pumping activity), and conversely, the capacity can be adjusted if the load alters (intralymphatic pressure increases). When the safety valve mechanisms are no longer effective or become overwhelmed, the body’s normal compensation is not enough, and lymphedema develops.
Lymphedema causes the lymphatic vessels to dilate; the valve flaps become incompetent (dilation/lymph valvular insufficiency), and the protein-rich lymphatic fluid refluxes to the tissue spaces (perilymphatic tissues). At first, a proliferation of initial lymphatic vessels occurs as the system tries to cope with the accumulation of lymphatic load. Lymph vessels can rejoin, or collateral lymphatics can develop to bypass the damaged area. In lymphedematous tissue, a state of chronic inflammation exists.67 This chronic inflammation leads to progressive tissue fibrosis, resulting in a state of relative hypoxia in the tissues, further impeding tissue oxygenation and contributing to a cycle of chronic inflammation and increased risk of infection.
In either primary or secondary lymphedema, infection or delayed wound healing (the latter can be directly related to the low oxygen state caused by edema) will add to the high-protein edema. Infection in the tissues (cellulitis) or infection in the lymph vessels (lymphangitis) can cause progressive tissue fibrosis and/or scarring in the lymph vessels (lymphangiosclerosis). Though some recanalization and collateralization of lymph vessels take place, lymphatic function remains compromised. An increase in the size of the tissue channels occurs with an increase in the distance for the oxygen to diffuse from the capillaries to the cells (Fig. 13-14). Gas exchange and metabolism of cellular waste products are impaired.
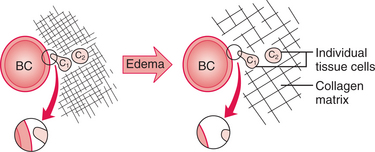
Figure 13-14 Reduction of gas (oxygen) exchange. Even a relatively minor amount of edema, which moves the fibers and individual tissue cells (C1, C2) apart by only a small amount, can cause a great increase in the resistance to diffusion of gases (and other small lipid-soluble molecules) between the cells and the blood capillaries (BC). The magnified view of the distance between the BC and the tissue cell (representing the cell’s oxygen supply) is greatly increased in the edematous state. The greater distance for oxygen to diffuse to nourish the cells will eventually lead to a hypoxic state. (From Casley-Smith JR, Casley-Smith JR: Modern treatment for lymphoedema, ed 5, Adelaide, Australia, 1997, Lymphoedema Association of Australia.)
Although the number of macrophages increases, their activity is decreased in the lymphedema fluid for reasons not clearly understood. Some theories suggest that the chronic lack of essential nutrients (e.g., oxygen) or perhaps a toxic factor produced by the stagnant proteins or the damaged tissues contribute to the deterioration of macrophage.22,26 In chronic lymphedema, the muscle wall of the collecting lymphatics hypertrophies, reducing the effective pumping power of these vessels (lymph pump deterioration).
The effect of lymphedema on the blood vessels causes a proliferation of new small blood vessels and the development of arteriovenous anastomoses. These new small vessels may leak as a result of abnormal changes in total tissue pressure in the lymphedematous region, further overloading the area. Proteins, fats, cellular waste products, and the 10% of tissue fluid volume that is not directly transported by the venous system have no alternative transport pathway from the interstitium to the venous system except via the lymphatic system. When this transport mechanism is impaired, lymphedema develops (Fig. 13-15, A). The impairment can be structural or functional.
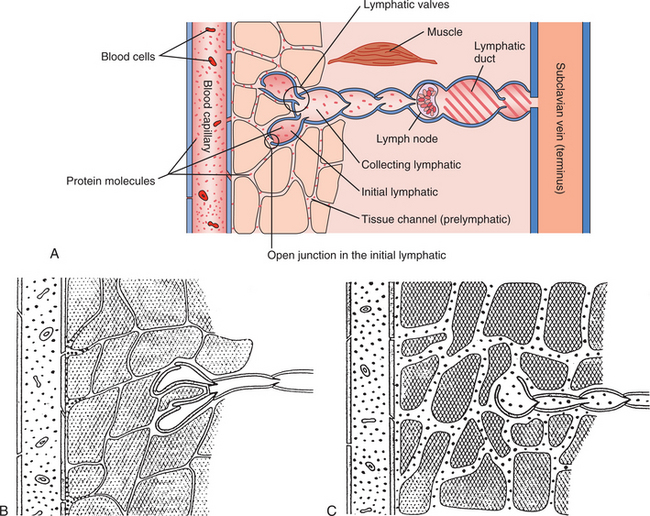
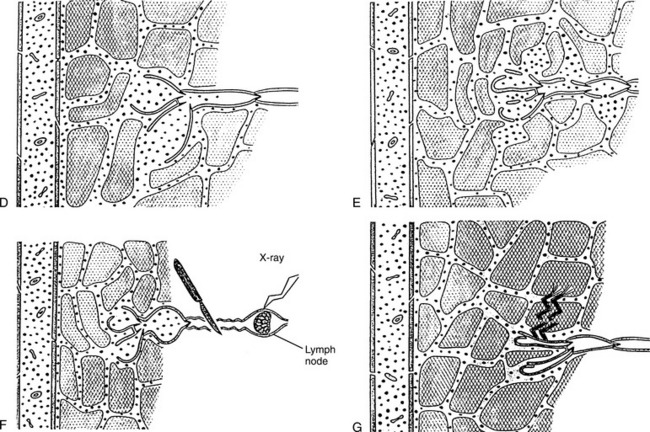
Figure 13-15 Low-flow, high-protein lymphedema caused by structural impairment. A, “Normal” tissue for comparison showing the passage of protein (dots) in normal tissue from the blood capillary, through the tissue channels, into and through the lymphatic system, and back to a vein. B, Altered interstitial tissue (e.g., too few or too narrow tissue channels). Notice that the prelymphatic channels are much narrower than in the normal tissue and the protein molecules are stacked up on the arterial side, unable to move easily through the narrow tissue channels, causing impaired lymph flow (and eventual tissue fibrosis). Inlet valves are closed because the endothelial cell junctions cannot open properly in fibrosed tissues, contributing to poor lymph drainage. C, Abnormally few initial lymphatics. This may occur developmentally or because some of the vessels become blocked (e.g., by fibrin). In this case, too few initial lymphatics are evident. Notice the dilation of the prelymphatic channels, the greater concentration of protein molecules in the tissue channels, and the malformed inlet valve. Low-flow, high-protein lymphedema caused by structural impairment. D, Malformations of the initial lymphatics preventing their inlet-valves from sealing; the prelymphatic tissue channels are dilated, a high-protein concentration exists there, and the tissue channels become dilated and stretched. This can happen in both primary and secondary lymphedema; in secondary lymphedema, it occurs after prolonged lymphostasis from a more proximal blockage. E, Injuries to the walls of the very fragile initial lymphatics. Lymph refluxes into the tissue channels, causing them to dilate. The high concentration of protein in the extracellular tissues causes a chronic inflammatory response and greater risk of infection. Note the larger spaces between the tissues. This results in a larger distance for oxygen to diffuse and leads to tissue hypoxia. F, Iatrogenic factors (e.g., surgery, radiation, tumor growth) that damage the lymphatic ducts or larger vessels impair lymph flow from the periphery. When movement of lymph from the tissue channels into the initial lymphatics is impaired, the tissue channels dilate, lymph stasis occurs, and there is a high-protein concentration in the tissues with subsequent chronic inflammation, increased risk of infection, and progressive swelling of the limb. G, Anchoring filaments tearing away from the interstitial tissue. This occurs in severe edema from other causes (e.g., rapid swelling from acute lymphedema as a result of trauma can tear the anchoring filaments from the surrounding tissues). The initial lymphatics can no longer function as conduits, as they would normally. The lymphostasis this produces worsens the existing edema. (From Casley-Smith JR, Casley-Smith JR: Modern treatment for lymphoedema, ed 5, Adelaide, Australia, 1997, Lymphoedema Association of Australia.) Lymphoedema Association of Australia
Structural Impairment.: Aging or damaged blood vessels are associated with structural impairment as fibrin physically narrows or blocks tissue channels (Fig. 13-15, B). Hypoplasia of the collecting lymphatics is also associated with the pathogenesis of structural lymphedema (Fig. 13-15, C). Primary lymphedema that manifests itself in puberty with a growth spurt and increase in tissue mass causes the body to outgrow or outstrip the capacity of the lymphatic system. The functioning vessels become overwhelmed, the walls fail, dilate, and result in valvular incompetence, causing increased peripheral intralymphatic pressures and peripheral lymphedema.
Structural lymphedema may also occur when the flaps of incompetent valves of the initial lymphatics (Fig. 13-15, D) no longer meet, allowing reflux of lymph to regions distal to the blockage. The initial lymphatics eventually dilate as well, their endothelial junctions remain open, and lymph refluxes to the tissues.
Other causes of structural lymphedema include gaps and tears in the initial lymphatic walls associated with trauma and inflammation (Fig. 13-15, E), physical obstruction of collecting lymphatics (Fig. 13-15, F) associated with fibrosis, radiation therapy, tumor growth, surgical excision of lymphatics during tumor removal, and torn anchoring filaments (Fig. 13-15, G) associated with sudden acute edema. The latter may occur secondary to massive trauma or infection and can tear the microfilaments that connect the initial lymphatics to the interstitial tissues, resulting in the collapse of the initial lymphatics because of the high total tissue pressure.
In the case of lymphedema caused by filariasis, the adult worm blocks the vessel it is in. Damage to lymph vessels occurs from the whiplike action of the constantly motile adult worms and the toxic effects of parasite secretory and excretory products. When the worm dies, the toxins released stimulate a granulomatous reaction with infiltration of plasma cells, eosinophils, and giant cells, further damaging the vessel and surrounding tissue as severe inflammation develops. Over time, repeated limb bacterial infections in previously damaged vessels may superimpose additional lymphatic damage.15,77
Functional Impairment.: Anything that causes a lack of variation in total tissue pressure (TTP) causes lymphedema. Bed rest, paralysis, or prolonged immobility can severely limit changes in total tissue pressure. For example, a survey by the Lymphoedema Association of Australia reported an increase in the incidence of lymphedema and exacerbation of preexisting lymphedema after air flight.23 It is this variation that contributes to a pressure gradient between the interstitial tissues and the intralymphatic pressure. Normally this pressure gradient stimulates the lymphangions to contract, which enhances the flow of lymph from the periphery to the center of the body. When the tissue pressure does not change or vary, the force pumps remain inactive.
Other factors contributing to functional impairment of the lymphatic system may include spasm of collecting lymphatics (e.g., lymphangiospasm caused by inflammation stimulating sympathetic nerves); paralysis of the collecting lymphatics (e.g., prolonged distention leading to fatigue and ultimately, failure); and impaired contraction of the collecting lymphatics caused by physical obstruction of fibrotic tissue surrounding lymphatic vessels. This type of functional impairment is common in severe Stage II lymphedema and lymphostatic elephantiasis. If collectors cannot pump, lymph refluxes peripherally, causing overdilation of initial lymphatics, valvular incompetence, lymphatic failure, and ultimately, lymphedema.
Clinical Manifestations
Primary or secondary lymphedema is characterized by clinical signs and symptoms caused by the effects of lymphedema on the lymphatics, body tissues, and blood vessels. Lymphedema resulting from filariasis is reversible in its early stages; secondary lymphedema can be transient if damage is minor. Secondary lymphedema can develop immediately postoperative or weeks, months, or years after surgery.
Lymphedema can develop in any part of the body or limb(s). Signs or symptoms of lymphedema include a full sensation in the affected body part; a sensation of skin tightness; decreased flexibility in the hand, wrist, or ankle; difficulty fitting into clothing in one specific area; or ring, wristwatch, or bracelet tightness. In advanced cases, fistulas to the skin, joints, or gut may develop; these are portals of entry for bacteria to invade the skin and cause recurrent infection.
Physical impairments caused by lymphedema can include increased circumferential limb girth; postural changes; tremendous discomfort (heavy, aching, or bursting sensations); neuromuscular deficits; and integumentary complications. These physical impairments can lead to functional limitations and disability along with the potential for psychosocial morbidity (e.g., social isolation, depression, or suicide).7,51 Healing time is increased, and all of this is occurring in a heavy, painful, and clumsy limb that is more prone to injury because of its abnormally large size and decreased functional mobility. Risk of injury is increased, whereas oxygenation and metabolism of waste and cellular debris are decreased. This is a most dangerous environment.
As the lymphedema progresses, atrophic skin changes can occur as a result of the low oxygen state, including loss of hair and sweat glands, formation of keratotic patches on the skin, and the development of papillomas (blisterlike outpocketings of the skin) that sometimes leak lymphatic fluid. Angiosarcoma (Stewart-Treves syndrome) is a rare malignancy that can develop in an advanced, chronic lymphedema that is left untreated.
Complications of Lymphedema.: As lymphedema progresses, the dermal layer of the skin thickens, the skin itself dries and cracks, and ulcerations often develop. These ulcers do not heal because of the tension on the tissues from the edema and the decreased oxygen state, coupled with the subcutaneous fibrosis and chronic inflammation. As the skin and tissues stretch, skin folds and tissue flaps can develop. These folds and flaps become breeding grounds for fungal and bacterial infections that further damage skin integrity, creating new portals of entry for the bacteria as the skin macerates and cracks.
Chronic fungus (tinea) is common on the foot and toes of anyone with lymphedema of the legs, as well as in the groin and under the breast. This fungus can be difficult, if not impossible, to treat topically. If this tinea is not addressed during treatment, a successful outcome is not possible19 (Fig. 13-16).
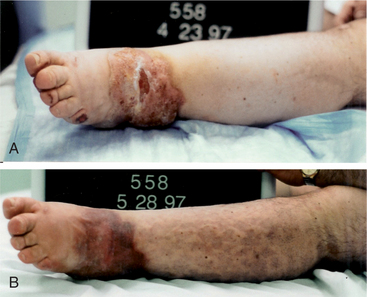
Figure 13-16 A 30-year-old male with spina bifida and lymphedema secondary to paralysis and unsuccessful plastic surgery with skin graft for a chronically itchy, 2-cm keratotic lesion on the left anterior ankle crease. A, Chronic ulceration complicated by fungal infection of the ulcer itself and the foot or toes. Note the hypertrophic fibrotic skin at the ankle and warty changes on the toes. B, After 4 weeks of CLT treatment (debridement of the ulcer was done daily in addition to the lymphatic drainage, compression bandaging, and other skin care). Exercise was not possible due to paralysis; deep abdominal breathing exercises and modified self-lymphatic drainage were taught and then practiced daily. Note that the wound healed completely and the infection resolved. (Courtesy Lymphedema Therapy, Woodbury, NY, 2006.)
The progressive increase in girth and weight of the affected areas contributes to pathologic alterations in the gait pattern and decreases in functional range of motion and strength caused by fatigue and inactivity. With increasing edema and subcutaneous fibrosis, tactile sensation and kinesthetic awareness are impaired, increasing the risk of injury to the affected areas.
If the edema progresses into the trunk quadrant adjacent to the lymphedematous limb, a further loss of trunk strength and function can occur. Some individuals, for example, must sleep in a recliner chair to prevent losing their independence, since they can no longer mobilize themselves and their heavy limbs in bed. A vicious cycle develops with decreasing mobility and strength leading to joint contractures, further increasing the risk to the already impaired skin integument.
The limbs may be painful and hypersensitive, and the adjacent trunk quadrants may ache and throb. Balance may be impaired, and the individual may no longer be able to shower or bathe independently, as a result of the fear of falling or inability to move the heavy limbs over the bath or shower ledge. Hygiene becomes a problem, further increasing the risk for fungal and bacterial infections.60,65
Primary lymphedema is more common in females and affects the lower extremities most often. However, it is seen in males and in the upper extremities, too.16 Primary lymphedema can be present in many parts of the body, and if it is present from birth, the deep internal organs can be affected. Clinical cases of primary lymphedema of the lower extremities involve the buttocks, genitals, and intestines with reflux of intestinal chyle (chylous reflux/protein-losing enteropathy) through fistulae on the genitals, buttocks, and thighs (see Fig. 13-20).
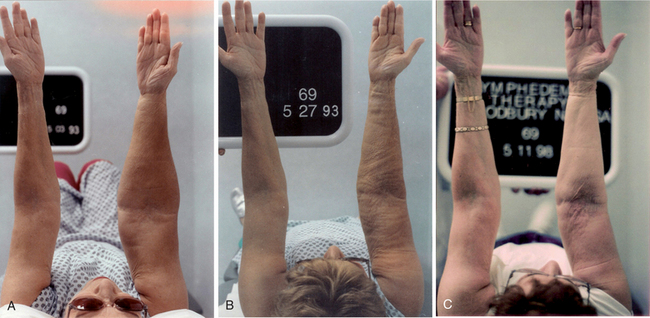
Figure 13-20 A, Before treatment, a 19-year-old male with severe Stage II primary lymphedema of the left lower extremity, progressing to the left buttock and genitals, with chylous reflux to the scrotum, buttock, and thigh. The onset of the edema was at age 8 (see Fig. 13-11). He spent most of his high school years in and out of the hospital. In the 24 months before treatment, he was hospitalized 22 times for cellulitis in the left lower extremity and placed in the intensive care unit (ICU) with septic shock three times. B, After one course of CDT of 30 treatments interrupted by a 2-week hospitalization (1 week in ICU) for cellulitis of the left leg and buttock with severe chylous leakage from the scrotum, buttock, and thigh. This individual went into septic shock and needed hyperalimentation to treat the hypoproteinemia (that resulted from the chylous leakage) via a central venous line. Despite the massive increase in swelling and open areas on the posterior left thigh and buttock resulting from the cellulitis, he achieved a 67% reduction in the lymphedema of his left lower extremity with reductions in the abdominal, suprapubic, and genital swelling as well. C, 12½ years after CDT. Note that some increase in the girth of the left lower extremity has occurred, particularly in the areas just proximal and distal to the knee, where the tissues are lax from the original debulking surgery. These areas fill in quickly without compression. Some of the girth increase is due to weight gain, now that he no longer has recurrent cellulitis and the chylous reflux is under control. This young man, having spent his high school years in and out of the hospital, was able to complete his college degree and is now a registered nurse working in an emergency department. Since his CDT treatment, he has made his lymphedema management home program a priority in his life and continues to maintain a 60% reduction in the lymphedema in his left lower extremity. (Courtesy Lymphedema Therapy, Woodbury, NY, 2006.)
This protein-losing enteropathy is a medical emergency. Severe protein loss can occur from this leakage of protein-rich chyle from the intestinal lymphatics. Individuals with primary lymphedema who complain of abdominal bloating, chronic diarrhea, or intolerance of fatty foods may also experience fluid accumulation in the abdomen and genitals from the pressure of the fluid leaking from the intestinal lymphatics into the abdomen. In some cases, these clients are treated medically or surgically with sclerotherapy to seal off these leaking lymphatics in an attempt to halt the reflux of this fluid into the abdomen.
Surgical dissection and radiation therapy involving the cervical, supraclavicular, or mandibular lymph nodes can cause secondary head and neck edema. Significant edema of the head and neck can cause severe functional impairments in speech, swallowing, and respiration, in addition to the pain and psychologic trauma from cosmetic disfigurement. When scarring and lymphatic dysfunction are severe, treatment, such as manual lymphatic drainage (MLD), cannot always restore normal cosmesis; however, it can successfully reduce some of the edema, leading to improved function. For example, in individuals with severe edema of the throat and neck after surgery and radiation for tongue cancer, lymphatic drainage can reduce the swelling enough to allow the person to discontinue use of a tracheotomy tube previously considered permanent. Some clients are able to eat solid foods again; others with severe periorbital and lower facial edema may be able to open the eyes and read and watch television again. Although these are not “cures,” they are great improvements in function that are important to individual quality of life.
MEDICAL MANAGEMENT
Remarkable progress has occurred in diagnosis and management of lymphedema in the United States since 1990. Ironically, this has occurred despite the incorrect prediction for those with breast cancer that the advent of breast-conserving lumpectomy would eliminate upper extremity lymphedema so commonly seen in individuals postmastectomy.
Many individuals who undergo lumpectomy receive axillary lymph node dissection (ALND) and radiation therapy too; these are the very factors that multiply an individual’s risk of developing lymphedema. It has been reported that a 3% incidence of lymphedema after sentinel lymph node biopsy (SLNB) alone increased to 17% when the client required an axillary dissection following the SLNB.79
Another study followed 102 women treated for breast cancer, defining lymphedema as a greater than 2 cm difference between measurement sites, and recorded lymphedema in 43.3% of women who underwent ALND alone, 22.2% of those who underwent SLNB alone, and 25% of those with combined SLNB and ALNB. Overall, the proportion of women experiencing increased arm size, numbness, or firmness/tightness in the limb on their operative side was higher in the ALNB group than the SLNB group.4
Wilke et al published data on surgical complications of SLNB in 5500 cases and reported a 6.9% incidence of “proximal upper extremity lymphedema” (change from baseline arm circumference of greater than 2 cm when compared to the contralateral arm).91 Gould et al found lower extremity lymphedema in 30% of individuals undergoing inguinal lymph node dissection in the treatment of vulvar cancer.47
Zhang et al reported only 3% lower extremity lymphedema after inguinal node dissection (during surgery to treat vulvar cancer) when they were able to preserve the saphenous vein compared to 32% lymphedema incidence in those people who underwent inguinal node dissection and saphenous vein ligation.95 Thanks to advances in surgery and chemotherapy and the emphasis on early detection, many more individuals are surviving cancer for many years, living and functioning well with “limbs at risk” for lymphedema. Refinement in measurement tools, as well as the inclusion of self-reported symp- toms, which may be present well before a 2-cm difference between measurement sites can be recorded, will hopefully lead to earlier diagnosis and management of lymphedema.5
DIAGNOSIS
An easy-to-perform clinical test for determining primary lower extremity lymphedema is Stemmer’s sign, a thickened cutaneous fold of skin over the second toe, typically present in the early and differential diagnosis of a primary ascending lymphedema without false positive findings. It appears in the late stages of the descending lymphedema80 (Fig. 13-17). Although it is possible to have a false negative Stemmer’s sign, primary lymphedema is rarely accompanied by a false positive Stemmer’s sign.
Secondary Lymphedema.
The clinical diagnosis of secondary lymphedema is fairly straightforward in a limb at risk, when there is known disruption to the regional lymphatics (e.g., after axillary dissection/radiation for breast cancer or after inguinal node dissection/radiation for melanoma of the leg). A detailed medical history, including all surgical procedures and their chronology relative to the onset or worsening of the edema, is the cornerstone of a successful lymphologic evaluation. In secondary lymphedema, most often no other diagnostic tests are needed to confirm the diagnosis.
A venous Doppler study of the edematous limb is often done to rule out a thrombus as the cause of the swelling. It is crucial to have recurrence of cancer firmly ruled out as the causative factor before initiating treatment for the lymphedema. Many oncologists will request a magnetic resonance imaging (MRI) or computed tomography (CT) scan of the chest for upper extremity swelling or the pelvis and abdomen for lower extremity swelling before initiating a referral for treatment of the lymphedema.
Lymphedema may be the first sign of cancer recurrence even years after cancer treatment. The prospect of recurrent disease is a frightening one, but it must be ruled out. Malignant lymphedema (i.e., directly resulting from neoplasm blocking a major nodal region or lymph vessel) is usually more severe and progresses more rapidly than nonmalignant secondary lymphedema.
It is often associated with severe pain and/or sensorimotor deficits, particularly in the upper extremity when the brachial plexus is involved. These symptoms must be differentiated from the pain and weakness of radiation plexopathy, which sometimes progresses more slowly, but causes the same type of functional deficits.
In cases of unexplained swelling, particularly in the lower extremities when no known trauma or surgery is evident, the clinical examination and history may not provide a definitive medical diagnosis. Although the MRI and CT scan will clearly show edema fluid in a limb or region, these tests do not give a description of lymphatic function. This type of information is obtained from a lymphoscintigram, a sophisticated nuclear medicine test (with cost comparable to the MRI or CT scan) that outlines the major lymphatic trunks in a region and provides a functional description of how much tracer material is moved, how far, and in what unit of time, compared with the “normal” values of a person of similar height and weight.
The test involves subcutaneously injecting a small amount of radioactive tracer in the web spaces of the first and second digits of either the hands or feet. The client then performs a set of standard movements for a specific time period and serial radiographs are taken. The amount of exposure from all the radiographs is approximately equal to one x-ray.
As the tracer is taken up by the lymphatics, it will outline the major lymph trunks and show the volume of tracer moved per unit of time. The presence of tracer reflux that goes back down a limb is called dermal backflow and gives an indication of more proximal obstruction in the deeper lymphatic collectors. When the etiologic factors of edema are unclear, this test clearly shows the functional deficits in the lymphatics. It is especially helpful in ruling out secondary lymphedema in cases of lipedema with questionable lymphedema of the legs.
TREATMENT.
The primary focus in a case of lymphedema triggered by a new or metastatic cancer is to treat the cancer first and then manage the lymphedema. Input on how to minimize exacerbation of the lymphedema during the cancer treatment is often well received, if given with the intent to provide comfort and function.
Individuals are encouraged to undergo the cancer treatment that they wish to pursue, without guilt or fear that it will worsen the lymphedema. They need to know the possibilities and risks but should not be frightened from necessary treatment for the cancer by a well-meaning health care professional.
On the other hand, the lymphedema should not be ignored. Management of the lymphedema must be coordinated with the medical team (e.g., medical oncologist, surgeon, and radiation oncologist) and the client. This communication can avoid further overloading the person with additional appointments and adding more to their daily activities than they can handle.
Medications.
No clear-cut pharmaceutical drug is available to treat lymphedema, although there is hope for a safe, effective medication that will lyse the protein accumulating in the obstructed area and speed the healing process. Clinical studies have shown that the benzopyrones (Coumarin, Venalot, Daflon, and their natural counterparts, the bioflavonoids, rutin, horse chestnut, and grapeseed extract)* have some effect on increasing proteolysis and increasing macrophage activity (remember that the macrophages become inactive in the lymphedema fluid and unable to perform their immune functions). However, these substances also raise some health risks, specifically liver toxicity. Further randomized clinical trials are needed to assess efficacy and dosing.
This increased proteolysis helps to reduce the interstitial protein concentration, signaling the body to reabsorb more extracellular fluid, thereby reducing the lymphedema. These substances have been shown to soften fibrotic tissue and increase the healing of chronic ulcerations and bacterial infections in elephantiasis lymphedema, probably a result of the activity of the macrophages, stimulating the immune response.†
Diuretics work well on sodium retention edemas but do not help lymphedema, yet they continue to be prescribed. Although it is true that taking large doses of diuretics will reduce total body fluid volume, these medications do not address the cause of the lymphedema (i.e., that the lymph load is exceeding the reduced lymph transport capacity). Furthermore, diuretics may move the “water” component of the lymphatic fluid, further concentrating the extracellular protein in the tissues increasing fibrosis. Moreover, chronic use of high-dose diuretics leaves the individual at great risk for developing electrolyte disturbances.
Experts agree that diuretics may be indicated in cases of malignant lymphedema. Individuals with comorbid conditions that require the use of diuretics, such as arterial hypertension, nephritic syndrome, or congestive heart disease, must be strongly advised to continue their medication and to consult with their primary physician with questions regarding their prescription.15,42
Some nonsteroidal antiinflammatory drugs (NSAIDs) that are cyclooxygenase-2 (COX-2) inhibitors (e.g., Celebrex) have a warning in the package insert about leg edema being a possible side effect. This has been clinically reported in several cases with lymphedema and arthritis; the individual with arthritis may experience an increase in edema when taking these drugs.10
Other commonly prescribed drugs, such as Norvasc (used to treat hypertension) and Avandia (used to treat diabetes), can cause leg edema. Lyrica (pregabalin), a fairly new drug for neuropathy (used with diabetic neuropathy and shingles), may cause heart failure and limb edema.
Many people do have neuropathy from chemotherapy (e.g., docetaxel [Taxotere]) that is permanent and take these medications to manage the symptoms of neuropathy. Therapists must be aware of this possible side effect in any of these medications and not assume that increased edema is a result of, for example, behavioral noncompliance or a problem with fit of a compression garment.
Clients may experience similar problems as new drugs for other conditions are introduced into the market. Even if the package does not warn of edema as a possible side effect, the therapist and client must observe carefully for any early signs or symptoms associated with the use of a new prescription.
Surgery.
Surgery has been used to “treat” severe lymphedema in the past with limited success. It is still done today if an individual receives no benefit from conservative treatments or does not have access to these treatments. Although numerous surgical approaches have been proposed to treat chronic lymphedema of the extremity, none has been clinically successful.55,70 Animal and cadaver studies continue investigating surgical techniques to resect tumors, reconstruct breast tissue, or perform liposuction45,46 that will prevent venous occlusion and subsequent lymphatic dysfunction.
Many microsurgical procedures attempt to anastomose a lymph vessel or node with a vein or with another functioning lymph vessel or lymph node. The morbidity and mortality from these procedures are significant. These procedures often fail soon after the surgery, leaving the individual with more superficial scarring that further blocks collateral lymph flow from the obstructed limb.
Debulking procedures (e.g., the Charles operation) seek to physically remove the excess fibrosclerotic connective tissue. (These procedures are rarely done in the United States except in extreme cases; debulking is still done in other countries.) These operations create extensive longitudinal scars on the involved extremities. Long incisions are made in the skin and subcutaneous tissues are removed down to the muscle and bone; the skin flaps are reapposed and sutured in place.
Although these procedures attempt to address the severely impaired cosmesis in the affected individuals, they do not address the cause of the impairment, which is decreased lymph transport capacity. This problem is unchanged by these procedures; over time, the limbs begin to swell again, often with disfiguring asymmetrical tissue flaps, large lymph cysts called papillomata (up to 6 to 8 cm in diameter), or warty protrusions on the skin (see Fig. 13-11).19,41
Reconstructive surgery after mastectomy is a very personal matter. Many procedures are available from the insertion of a tissue expander, followed later by the insertion of a permanent saline implant to the more extensive myocutaneous tissue flap procedures (i.e., transverse rectus abdominal muscle [TRAM] flap, free latissimus flap, or deep inferior epigastric flap [DIEP]) involving transplanting flaps of muscle and skin to the breast area to form a more “natural” breast.
The TRAM flap, which transplants the contralateral rectus abdominis muscle by tunneling it across the abdomen and up to the breast area, involves extensive scarring in the suprapubic area from hip to hip, in addition to the individual scars on the breast area. The procedure usually consists of two other minor procedures, performed separately to tattoo the areola and create a nipple. The horizontal hip-to-hip scar effectively blocks a large area of collateral lymph flow from the ipsilateral thoracic lymphotome through the abdominal lymphotome to the superficial inguinal nodes. This area of collateral lymph drainage is important for the treatment of ipsilateral upper extremity lymphedema that can develop after axillary dissection and mastectomy. In some cases, abdominal lymphedema develops after this procedure. This should not be confused with asymmetric edema, which may really be a hernia of the abdominal contents on the contralateral side where the abdominal muscle was removed. This occurs on rare occasions, but it must be medically diagnosed and surgically corrected.
The latissimus flap procedure also creates a long transverse scar on the ipsilateral posterolateral thorax, effectively blocking a large area of collateral lymph drainage from the upper extremity to the ipsilateral superficial inguinal nodes. This lymphatic pathway may be needed to treat an upper extremity lymphedema secondary to axillary dissection or radiation. These procedures may lead to truncal lymphedema, which is often not recognized, sometimes discounted as psychosomatic, and can cause considerable disability, impairment of function, and psychosocial distress.36
PROGNOSIS.
Left untreated, lymphedema is a progressive disease; current surgical and pharmacologic approaches do not bring about a “cure.” Lymphedema is a life-long, chronic condition, but one that can be managed effectively with proper intervention, client education, and regular follow-up care.
Lymphadenitis
Infections elsewhere in the body can lead to lymphadenopathy as described previously. When the lymph node becomes overwhelmed by the infection, the lymph node itself can become infected; this is called lymphadenitis. Lymphadenitis can be classified as acute or chronic; acutely inflamed lymph nodes are most common locally in the cervical region in association with infections of the teeth or tonsils or in the axillary or inguinal regions secondary to infection of the extremities.
In acute lymphadenitis, the lymph nodes are enlarged, tender, warm, and reddened. In the case of chronic lymphadenitis, long-standing infection from a variety of sources results in scarred lymph nodes with fibrous connective tissue replacement. The nodes are enlarged and firm to palpation but not warm or tender. The management of lymphadenitis is treatment of the underlying disorder.
Lymphangitis
Lymphangitis, an acute inflammation of the subcutaneous lymphatic channels, usually occurs as a result of hemolytic streptococci or staphylococci (or both) entering the lymphatic channels from an abrasion or local trauma, wound, or infection (usually cellulitis). The involvement of the lymphatics is often first observed as a red streak under the skin (referred to in layperson terms as blood poisoning), radiating from the infection site in the direction of the regional lymph nodes.
The red streak may be very obvious, or it may be very faint and easily overlooked, especially in dark-skinned people. The nodes most commonly affected are submandibular, cervical, inguinal, and axillary, in that order. Involved nodes are usually tender and enlarged (greater than 3 cm).
Systemic manifestations may include fever, chills, malaise, and anorexia. Other symptoms may present in association with the underlying infection located elsewhere in the body. Bacteremia from any cause can result in suppurative arthritis (inflammatory with pus formation), osteomyelitis, peritonitis, meningitis, or visceral abscesses.
When cellulitis results in lymphangitis, throbbing pain occurs at the site of bacterial invasion, and the client presents with a warm, edematous extremity (or possible scrotal lymphedema in males and occasionally vulvar lymphedema in females).
MEDICAL MANAGEMENT
Diagnosis.
Lymphangitis may be confused with superficial thrombophlebitis, but the erythema associated with lymphangitis is first seen as a red streak under the skin radiating toward the regional lymph nodes (usually ascending proximally), whereas the erythema associated with thrombosis is usually over the thrombosed vein with local induration and inflammation.
However, suppurative thrombophlebitis may develop if bacteria are introduced during IV therapy, especially when the needle or catheter is left in place for more than 48 hours. The physician will also differentiate cellulitis from soft tissue infections (e.g., gangrene or necrotizing fasciitis) that may require early and aggressive incision and resection of necrotic infected tissue.
Anyone with a history of vascular disease taking anticoagulant medication should have a Doppler ultrasound to rule out DVT before being treated. Laboratory tests are often not required but may include blood culture (often positive for staphylococcal or streptococcal species) and culture and sensitivity studies on the wound exudate or pus.
Treatment and Prognosis.
Prompt parenteral antibiotic therapy is mandatory because bacteremia and systemic toxicity develop rapidly once organisms reach the bloodstream via the thoracic duct. Antibiotic treatment may be accompanied by general measures such as heat, elevation, immobilization of the infected area, and analgesics for pain.
Appropriate wound care may include drainage of the pus from an infected wound when it is clear that an abscess is associated with the site of initial infection. An area of cellulitis should not be excised because the infection may be spread by attempted drainage when pus is not present. Treatment as described should be effective against invading bacteria within a few days.
MEDICAL MANAGEMENT
INFECTIONS WITH IMPAIRED AND AT-RISK LYMPHATIC SYSTEM AND LYMPHEDEMA
Diagnosis.
Confusion of cellulitis or lymphangitis with thrombophlebitis in the individual with lymphedema or at risk for lymphedema is common and has a disastrous impact on the severity of the lymphedema. An episode of cellulitis or lymphangitis often triggers the development of lymphedema in the individual who is at risk but has no clinical signs of edema. Improper, inadequate treatment of these infections can lead to chronic infection or inflammation and progression of the lymphedema.
The individual who has had regional lymph node dissection and/or radiation has an impaired immune response in the areas that drain to that regional nodal area. Consequently, infection can spread rapidly in those regions and any delay in treatment while awaiting blood cultures and vascular tests can cause progression of the infection.
The combination of the impaired nodal area and the inactivity of the macrophages in the lymphedematous region allows the bacteria to multiply rapidly, feeding on the high protein lymphedema fluid. It is not uncommon for a person to develop a high fever with shaking chills (105° F [40.5° C]) within 30 minutes of “feeling ill” or “feeling an ache or pain” in the lymphedematous region. It is not acceptable for these people to “wait and see” and call their physician in a day or two. They must be seen and evaluated by a physician immediately to rule out thrombosis versus cellulitis or lymphangitis and initiate appropriate treatment immediately.
The more insidious onset of local infection (cellulitis) may manifest with an area of redness on the skin that looks like a rash or sunburn. The area may be warm to touch but not initially painful, and the individual may not develop any pain or fever. In fact, cellulitis is often mistaken for an allergic reaction, thought to be a reaction to an insect bite, or a local reaction to a sprain or strain, even when these triggers were not present.
Individuals with advanced Stage 2 and 3 lymphedema may develop recurrent cellulitis that is mistaken for “normal” skin changes associated with chronic lymphedema (Fig. 13-21)
Figure 13-21 This individual had suffered from pain, increased swelling, and itching of the skin on both legs for months. She had suffered from swollen ankles and feet as a teen and the swelling had worsened as she aged. She had seen a vascular surgeon who performed Doppler studies and ruled out deep vein thrombosis (DVT) but had no diagnosis for her problem. She had seen a dermatologist who diagnosed a contact dermatitis and prescribed topical cortisone cream. Luckily, her podiatrist referred her to a lymphedema specialist who promptly diagnosed primary lymphedema of both legs, complicated by cellulitis. (Courtesy Lymphedema Therapy, Woodbury, NY, 2006.)
Treatment and Prognosis.
Treatment of cellulitis or lymphangitis in the individual with lymphedema or at risk for lymphedema differs from treatment of these conditions in the individual with an intact lymphatic system. In a healthy individual, cellulitis and lymphangitis are relatively rare. Many cases of primary lymphedema are missed in young individuals who present with unexplained recurrent cellulitis in the absence of injury or trauma. Recurring cellulitis does not develop unless some underlying pathology is causing it (usually primary lymphedema).
In the healthy individual, heat is often prescribed as an adjunct to relieve pain in the inflamed area, whereas heat should never be applied to individuals with lymphedema or those at risk for developing lymphedema. This contraindication includes those individuals who have developed lymphedema after orthopedic surgeries.
Local heating will increase vasodilation and ultrafiltration of more fluid into the interstitial spaces, further overloading the decompensated lymphatic transport capacity, exacerbating the existing lymphedema, or possibly triggering lymphedema in the limb at risk, where no clinical lymphedema existed before the onset of the infection. Rest and immobilization of the involved areas are recommended measures. Cold can be applied to relieve pain.
Experienced lymphologists initiate immediate oral antibiotic therapy, usually with high doses of broad-spectrum antibiotics and periodic medical monitoring in the first 48 to 72 hours to observe the area for spread of the infection. It is imperative that the original area of redness be outlined with indelible marker to check on the progress or regression of the infection (Fig. 13-22).
Figure 13-22 Individual with primary lymphedema in the lower extremities with cellulitis in the lower leg. Note the black line drawn by the therapist to mark the extent of the infection. This is important to monitor the course of the infection. The person was prescribed an oral antibiotic and advised by the physician that if the redness progressed proximal to the black line, she must go to the emergency room and be assessed for intravenous antibiotics. Note the severity of the swelling and fibrosis of the toes. Examination of the skin between her toes revealed that she had a fungal infection that caused the skin to macerate and crack. Bacteria can then enter the broken skin and cause a secondary bacterial infection. This is a common problem in people with lymphedema of the lower extremities. (Courtesy Lymphedema Therapy, Woodbury, NY, 2006.)
If a poor response to the oral antibiotics is evident, hospitalization for IV antibiotics may be necessary. A local infection can progress to septic shock if not treated effectively. A common cause of recurrent cellulitis or lymphangitis is poor, inadequate treatment of a previous infection. A common complaint is recurrent infection in the same area of the same limb every 2 to 3 weeks. In fact, this is more likely the same infection that was never eradicated the first time. Cases like this often have a common history of administration of too short (3 to 5 days) or inadequate doses of oral antibiotics to treat the cellulitis or lymphangitis associated with lymphedema.
Lipedema
Overview and Etiologic Factors
The term lipedema was first used by Allen and Hines to describe a symmetrical “swelling” of both legs, extending from the hips to the ankles, caused by deposits of subcutaneous adipose tissue.2 The underlying etiologic factors of these fat deposits remain unknown. Although lipedema is not a disorder of the lymphatic system per se, it is often confused with bilateral lower extremity lymphedema, thus the reason for discussing it in this chapter. It occurs almost exclusively in women, may have an associated family history (20% of cases), and is usually accompanied by hormonal disorders as well.82 If present in a man, it is accompanied by massive hormonal disorder.
Fat in the lower extremities extends to the malleoli, often with flaps of tissue hanging over the foot. The feet are not affected; occasionally, lipedema is found in the arms. Typically, fatty bulges are in the medial proximal thigh and the medial distal thigh, just above the knee. Clinically, the affected individuals complain of pitting edema as the day progresses, which is relieved by prolonged elevation of the leg or legs overnight.19,73,74
Stages of Lipedema
In stage I, the skin is still soft and regular, but nodular changes can be felt on palpation (Fig. 13-23). No color changes occur in the skin, and the subcutaneous tissues have a spongy feel, like a soft rubber doll. In stage II, the subcutaneous tissue becomes more nodular and tough. Large fatty lobules begin to form on the medial distal and proximal thigh and medial and lateral ankles just above the malleoli (Fig. 13-24). Pitting edema is common, increasing as the day progresses. The individual may report hypersensitivity over the anterior tibial area. Skin color changes occur in the lower leg, indicative of secondary lymphedema, which often occurs in later stage lipedema.
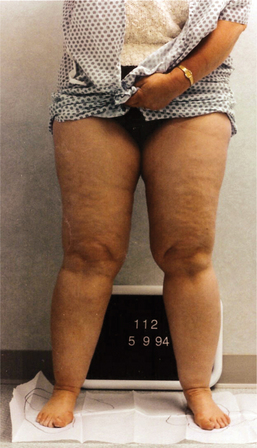
Figure 13-23 Stage I lipedema. Note that the feet are free of edema and the ankles and lower legs have pitting edema. Fatty nodules are beginning to appear on the distal thighs. (Courtesy Lymphedema Therapy, Woodbury, NY, 2006.)
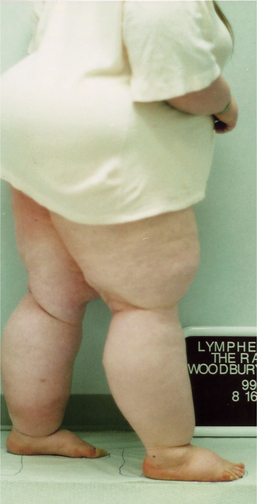
Figure 13-24 Stage II lipedema. Note that obvious pitting edema on the dorsum of the feet is evident, and the tissues are beginning to hang over the medial and lateral ankles and above the knees. The discoloration at the anterodistal left lower leg is from a resolving cellulitis, secondary to the lymphedema that developed secondary to the lipedema. (Courtesy Lymphedema Therapy, Woodbury, NY, 2006.)
Pathophysiology of Lipedema82
Many histologic and physiologic changes occur in lipedema. A decrease in the elasticity of the epidermis and subcutis also occurs. The basement membrane of vessels is thickened, and disturbances in vasomotion take place. The venoarterial reflex (VAR) is disturbed, causing decreased vascular resistance, increased skin perfusion, and increased capillary filtration. Under normal circumstances, the VAR is an important mechanism for the regulation of microcirculation and interstitial fluid exchange. It is measured as a ratio between skin perfusion in the supine versus the standing position using a laser Doppler flowmeter.
Increased venous or blood capillary pressure causes increased ultrafiltration. These changes, combined with the decreased efficiency of the calf muscle pump, result in both the dependent pitting edema seen in Stage I and the secondary lymphedema that often complicates lipedema in its later stages.42
Histologic changes seen in lipedema include a thinning of the epidermal layer, thickening of the subcutaneous tissue layer, fibrosis of arterioles, tearing of elastic fibers, dilated venules and capillaries, and hypertrophy and hyperplasia of fat cells. Clinical studies show enlargement of the prelymphatic channels81 and defects in capillary perfusion.89 Some authors have reported no alteration in lymphatic transport,14 whereas others9 have reported decreased lymph outflow in those individuals with lipedema. Földi and Földi reported an increase in fat cell growth during lymphostasis.40
MEDICAL MANAGEMENT
The diagnosis of lipedema is difficult if the clinician is unfamiliar with this condition. Often, these people are told that they are “fat” and should just lose weight to resolve the problem. For reasons still unknown, the fatty tissue accompanying this condition cannot be significantly decreased by diet. It is not uncommon for a diagnosis of primary lymphedema to be made. This results in frustration for the person who then seeks out lymphedema therapy with poor results.
Several significant clinical differences exist between lipedema and bilateral primary lymphedema. The feet are not involved in lipedema; although they are edematous with a positive Stemmer’s sign in lymphedema, Stemmer’s sign is negative in lipedema (see Fig. 13-17). The “swelling” in lipedema is symmetric, whereas in primary lymphedema, usually one limb is more involved than the other. The subcutaneous tissues feel rubbery in lipedema. In advanced Stage II lymphedema, significant subcutaneous fibrosis occurs, which feels firmer than lipedema.
Although incidences of cellulitis in stage II lipedema, usually with a component of lymphedema as well, have been reported, the frequency of cellulitis in stage II lymphedema is much higher. The time of onset of the “swelling” in lipedema is usually around puberty, and 90% of these cases have accompanying diagnoses of hormonal disturbance (thyroid, pituitary, or ovarian). This is usually not the case with primary lymphedema.
A lymphoscintigram may be helpful to differentiate between lymphedema and lipedema; however, results can be conflicting, as lymphedema often occurs to some degree in the later stages of lipedema, probably a result of impairment of lymph flow caused by the pressure of fatty tissue.
In fact, clinical cases of bilateral lower extremity lymphedema in the morbidly obese individual are seen; the onset of the lymphedema occurs after body weight exceeds 350 to 400 lbs. It is plausible to suspect that the pressure of a large apron of abdominal fat can effectively block lymph flow through the inguinal area, causing the lymphedema, but the difference between these cases and lipedema is that obesity does not cause lipedema.
Lipedema is caused by a hormonal imbalance resulting in excessive deposition of adipose tissue, most often in the lower extremities (see Figs. 13-23 and 13-24), although it can occur in the upper extremities, too.
TREATMENT AND PROGNOSIS.
No effective medical treatment for lipedema is available, and the prognosis is guarded; however, significant functional improvements are possible with good program compliance and therapy intervention. Medical management involves treating the hormonal disturbance as effectively as possible and providing nutritional guidance to avoid additional weight gain. Many of these individuals have endured years of ridicule because of their physical appearance and become recluses in their homes, further limiting their activity level.
As lipedema progresses and the hypersensitivity increases, they feel less inclined to walk or exercise because of the pain. They inevitably gain more weight as a result of the inactivity and depression, often finding food their only comfort.
References
1. Ahmed, RL, Thomas, W, Yee, D, et al. Randomized controlled trial of weight training and lymphedema in breast cancer survivors. J Clin Oncol. 2006;24(18):2765–2772.
2. Allen, EV, Hines, EA. Lipedema of the legs. Proc Staff Mayo Clinic. 1940;15:184–187.
3. Armer, J. The problem of post-breast cancer lymphedema: Impact and measurement issues. Cancer Investig. 2005;1:76–83.
4. Armer, JM, Fu, MD, Wainstock, JM, et al. Lymphedema following breast cancer treatment including SLNB. Lymphology. 2004;37:73–91.
5. Armer, JM, Radina, ME, Porock, D, et al. Predicting breast cancer-related lymphedema using self-reported symptoms. Nurs Res. 2003;52(6):370–379.
6. Armer, JM, Stewart, BK. A Comparison of four diagnostic criteria for lymphedema in a post-breast cancer population. Lymph Res Biol. 2005;3(4):208–217.
7. Augustine, E. Oncology section of the American Physical Therapy Association Position Statement physical therapy: management of lymphedema in patients with a history of cancer. Rehabil Oncol. 2000;18(1):9–12.
8. Bernas, M, Witte, CL, Witte, MH. The diagnosis and treatment of peripheral lymphedema: draft revision of the 1995 Consensus Document of the International Society of Lymphology Executive Committee for Discussion at the September 3-7, 2001, XVIII International Congress of Lymphology in Genoa, Italy. Lymphology. 2001;34(2):84–91.
9. Bilancini, S, et al. Functional lymphatic alterations in patients suffering from lipedema. Angiology. 1995;46:333–339.
10. Boris M: Personal communication, Lymphedema therapy, Woodbury, NY, 2007.
11. Boris, M, Weindorf, S, Lasinski, B. Lymphedema reduction by noninvasive complex lymphedema therapy. Oncology. 1994;8(9):95–106.
12. Boris, M, Weindorf, S, Lasinski, B. Persistence of lymphedema reduction after noninvasive complex lymphedema therapy. Oncology. 1997;11(1):99–109.
13. Boris, M, Weindorf, S, Lasinski, B. The risk of genital edema after external pump compression for lower limb lymphedema. Lymphology. 1998;31:15–20.
14. Brautigam, P, et al. Analysis of lymphatic drainage in various forms of leg edema using two compartment lymphoscintigraphy. Lymphology. 1998;31(2):43–55.
15. Browse, N, Burnand, K, Mortimer, PS. Diseases of the lymphatics. London: Arnold, 2003.
16. Brunner, U. Epidemiology and clinic of primary lymphoedema based on 500 cases. In: Bollinger A, Partsch H, Wolf JHN, eds. The initial lymphatics. Stuttgart: Stuttgart Thieme, 1985.
17. Cahan, ME. Trends in the evaluation of lymphedema. Lymphology. 2006;35:28–38.
18. Casley-Smith, JR. The efficiency of the lymphatic system. Lymphologie. 1978;2:24–29.
19. Casley-Smith, JR. There are many benzopyrones for lymphedema. Lymphology. 1997;30:38.
20. Casley-Smith, JR, Bjorlin, M. Some parameters affecting the removal of oedema by massage mechanical or manual. In: Casley-Smith JR, Piller NB, eds. Progress in lymphology X. Adelaide, Australia: University of Adelaide Press, 1985.
21. Casley-Smith, JR, Casley-Smith, JR. High-protein oedema and benzopyrones. Sydney and Baltimore: JB Lippincott, 1986.
22. Casley-Smith, JR, Casley-Smith, JR. Lymphedema initiated by aircraft flights. Aviat Space Environ Med. 1996;67:52–56.
23. Casley-Smith, JR, Casley-Smith, JR. Lymphedema therapy in Australia: complex physical therapy, exercises and benzopyrones on over 600 limbs. Lymphology. 1994;27:622–626. [(suppl)].
24. Casley-Smith, JR, Casley-Smith, JR. Modern treatment for lymphoedema. Adelaide, Australia: Lymphoedema Association of Australia, 1997.
25. Casley-Smith, JR, et al. Treatment for lymphedema of the arm the Casley-Smith method. A non-invasive method produces continued reduction. Cancer. 1998;83:2843–2860.
26. Casley-Smith, JR, Gaffney, RM. Excess plasma protein is a cause of chronic inflammation and lymphoedema. Quantitative electron microscopy. J Pathology. 1981;133:243–272.
27. Casley-Smith, JR, Piller, NB, Morgan, RG. Treatment of lymphedema of the arms and legs with 5,6 benzo-alpha pyrone. N Engl J Med. 1993;329:1158–1163.
28. Cheville, Al, McGarvey, CL, Petrek, JA, et al. Lymphedema management. Seminars in Radiation Oncology. 2003;13(3):290–301.
29. Chua B, Ung O, Boyges J: Toward optimal treatment of the axilla in early breast cancer. Poster presented at ASCO, New Orleans, 2000.
30. Consensus Document of the International Society of Lymphology Executive Committee. Diagnosis and treatment of peripheral lymphedema. Lymphology. 1995;28:113–117.
31. Cornish, BH, Chapman, M, Hirst, B, et al. Early diagnosis of lymphedema using multiple frequency bioimpedance. Lymphology. 2001;34:2–11.
32. Darocxy, J, Schingale, FJ, Mortimer, PS. Practical ambulant lymphology. Munich: Verlag Medical Concept, 1996.
33. Diagnosis and treatment of peripheral lymphedema. Consensus Document of the International Society of Lymphology. Lymphology. 2003;36:84–91.
34. Dickens, S, et al. Effective treatment of lymphedema of the extremities. Arch Surg. 1998;133:452–457.
35. Eliska, O, Eliskova, M. Are peripheral lymphatics damaged by high pressure manual massage? Lymphology. 1995;28:21–30.
36. Erlich, A, Vingé-Harrewijn, A, McMahon, E. Living well with lymphedema. San Francisco: Lymph Notes, 2005.
37. Földi, E. Massage and damage to lymphatics. Lymphology. 1995;28(1):3.
38. Földi, E. Treatment of lymphedema and patient rehabilitation. Anticancer Res. 1998;18:2211–2212.
39. Földi, E, Földi, M, Weissleder, H. Conservative treatment of lymphedema of the limbs. Angiology. 1985;36:171–180.
40. Földi, M, Földi, E. Lymphoedema: methods of treatment and control. A guide for patients and therapists. Stuttgart: Gustav Fischer Verlag, 1991. [(First English translation by Dr. Andrew C. Newell for the Lymphoedema Association of Victoria, Inc, Australia, 1993.)].
41. Földi, M, Kubik, S. Lehrbuch der lymphologie fur mediziner und physiotherapeuter: mit anhang, praktische hinweise fur die physiotherapie. Stuttgart and New York: Gustav Fischer Verlag, 1989.
42. Földi, M, Földi, E, Kubik, S. Textbook of lymphology for physicians and lymphedema therapists. Munich: Elsevier GmbH, 2003.
43. Franzeck, UK, Spiegel, I, Fischer, M. Combined physical therapy for lymphedema evaluated by fluorescence microlymphography and lymph capillary pressure measurements. J Vasc Res. 1997;34:306–311.
44. Freedman, DO. Filariasis. In Bennett JC, Plum F, eds.: Cecil textbook of medicine, ed 22, Philadelphia: WB Saunders, 2004.
45. Frick, A, et al. Liposuction technique and lymphatic lesions in lower legs: anatomic study to reduce risks. Plast Reconstr Surg. 1999;103(7):1868–1873.
46. Gabka, CJ, Baumeister, RG, Maiwald, G. Advancements of breast conserving therapy by onco-plastic surgery in the management of breast cancer. Anticancer Res. 1998;18(3C):2219–2224.
47. Gould, N, Kamelle, S, Tillmanns, T, et al. Predictors of complications after inguinal lymphadenectomy. Gynecol Oncol. 2001;82(2):329–332.
48. Huang, JH, Kwon, JY, Lee, KW. Changes in lymphatic function after complex physical therapy for lymphedema. Lymphology. 1999;32:15–21.
49. Johansson, K, Albertsson, M, Ingvar, C, et al. Effects of compression bandaging with or without manual lymph drainage. Treatment in patients with postoperative arm lymphedema. Lymphology. 1999;32(3):103–110.
50. Johansson, K, Tibe, K, Weibull, A, et al. Low intensity resistance exercise in breast cancer patients with arm lymphedema with or without compression sleeves. Lymphology. 2005;38:167–180.
51. Karki, A, Sopmonen, R, Malkia, E. Impairments, activity limitations and participation restrictions 6 and 12 months after breast cancer operations. J Rehabil Med. 2005;37(3):180–188.
52. Lane, K, Worsley, D, McKenzie, D. Exercise and the lymphatic system: implications for breast cancer survivors. Sports Med. 2005;35(6):461–471.
53. Lasinski, B, Boris, M. Comprehensive lymphedema management: results of a 5 year follow-up. Lymphology. 2002;35(suppl):301–304.
54. Leduc, A, Caplan, I, Leduc, O. Lymphatic drainage of the upper limb: substitution lymphatic pathways. Euro J Lymphol. 1993;4(13):11–18.
55. Lerner, R. Chronic lymphedema. In: Chang JB, ed. Textbook of angiology. New York: Springer-Verlag, 2000.
56. Levin DK: Measuring blood pressure in legs, Medscape. Available on-line at http://www.medscape.com/viewarticle/471829. Accessed 03/26/2004.
57. Lindsay, SW, Thomas, CJ. Mapping and estimating the population at risk from lymphatic filariasis in Africa. Trans R Soc Trop Med Hyg. 2000;94(1):37–45.
58. Loprinzi, CL, et al. Lack of effect of coumarin in women with lymphedema after treatment for breast cancer. N Engl J Med. 1999;340:346–350.
59. Lymphedema: understanding and managing lymphedema after cancer treatment. Atlanta: American Cancer Society, 2006.
60. McMahon, E. Overcoming the emotional challenges of lymphedema. San Francisco: Lymph Notes, 2005.
61. Miller, LT. A theory to support the use of vigorous exercise in the reduction of breast cancer lymphedema. Phys Ther. 1997;7:5.
62. Mortimer, PS, Badger, C, Clarke, I. A double-blind, randomized, parallel-group placebo-controlled trial of O-betahydroxyethyl-rutosides in chronic arm oedema resulting from breast cancer treatment. Phlebology. 1995;10:51–55.
63. Mortimer, PS, et al. The prevalence of arm lymphoedema following treatment for breast cancer. Quart J Med. 1996;89:377.
64. Moseley, AL, Piller, NB, Carati, CJ. The effect of gentle arm exercise and deep breathing on secondary arm lymphedema. Lymphology. 2005;38:136–145.
65. Mowayne, J, Heiney, SP. Psychologic and social sequelae of secondary lymphedema: a review. Cancer. 2005;104(3):457–466.
66. Newman, M, Brennan, M, Passik, S. Lymphedema complicated by pain and psychological distress: a case with complex treatment needs. J Pain Sympt Manage. 1996;12:376–379.
67. Olszewski, WL. Lymph stasis: pathophysiology, diagnosis, and treatment. London: Olszewski, WLCRC Press, 1991.
68. Oncology staff (nurses and physical therapists) across the United States: Personal communication, 2007.
69. Pecking, A, et al. Efficacy of Daflon 500 mg in the treatment of lymphedema (secondary to conventional therapy for breast cancer). Angiology. 1997;48:93–98.
70. Petrek, J, Lerner, R. Lymphedema. In: Harris JR, Lippmann ME, Morrow M, et al, eds. Diseases of the breast. Philadelphia: Lippincott Williams & Wilkins, 2000.
71. Pfalzer C: Personal communication, 2006.
72. Radina, ME, Armer, JM. Surviving breast cancer and living with lymphedema: Resiliency among women in the context of their families. J Fam Nurs. 2004;10(4):485–505.
73. Rank, BK, Wong, CSC. Lipoedema. Austral NZ J Surg. 1966;35(3):165–169.
74. Rudkin, GH, Miller, TA. Lipedema: a clinical entity distinct from lymphedema. Plast Reconstr Surg. 1994;94:841–849.
75. Ryan, TJ. The skin and its response to movement. Lymphology. 1998;31:128–129.
76. Ryan, TJ. On treatment of peripheral lymphedema. Lymphology. 2003;36:110.
77. Ryan, TJ. Lymphatic filariasis and the international society of lymphology. Lymphology. 2004;37(3):151–157.
78. Segers, P, Belgrado, JP, Leduc, A, et al. Excessive pressure in multichambered cuffs used in sequential compression therapy. Phys Ther. 2002;82(10):1000–1008.
79. Sener, S, et al. Lymphedema after sentinel lymphadenectomy for breast carcinoma. Cancer. 2001;92(4):748–752.
80. Stemmer, R. Stemmer’s sign possibilities and limits of clinical diagnosis of lymphedema. Wien Med Wochenschr. 1999;149(2-4):85–86.
81. Stoberl, CH, Partsch, H, Urbanek, A. Indirekte lymphographie beim lipodem. In: Földi E, ed. Odem perimed. Germany: Erlangen, 1986.
82. Strossenreuther, RHK. Die bahandlung des lipodems. In: Földi M, Kubik S, eds. Lehrbuch der lymphologie. Stuttgart and New York: Gustav Fischer Verlag, 1999.
83. Swartz, M. Cellulitis and subcutaneous tissue infections. In Mandell G, Bennett J, Dolin R, eds.: Principles and practice of infectious diseases, ed 6, Philadelphia: Churchill Livingstone, 2005.
84. Szuba, A, Razavi, M, Rockson, SG. Diagnosis and treatment of concomitant venous obstruction in patients with secondary lymphedema. J Vasc Interv Radiol. 2002;13(8):799–803.
85. Taylor, HM, Rose, KE, Twycross, RG. A double-blind clinical trial of hydroxy-ethylrutosides in obstructive arm lymphoedema. Phlebology. 1993;1:22–28. [(suppl)].
86. Taylor, R, Jayasinghe, VW, Koelmeyer, L, et al. Reliability and validity of arm volume measurements for assessment of lymphedema. Phys Ther. 2006;86:205–214.
87. Tunkel, R, Cohen, S. Lymphedema management. Rehabil Oncol. 2000;18(1):26–27.
88. Turner, J, Hayes, S, Raul-Hirche, H. Improving the physical status and quality of life of women treated for breast cancer: A pilot study of a structured exercise intervention. J Surg Oncol. 2004;86(3):141–146.
89. Weinert, V, Leeman, S. Das lipodem. Hautarzt. 1991;42:484–486.
90. Weissleder, H, Schuchardt, C. Lymphedema: diagnosis and therapy, ed 2. Stuttgart: Koln, Viavital Verlag GmbH, 2001.
91. Wilke, LG, McCall, LM, Posther, KE, et al. Surgical complications associated with sentinel lymph node biopsy: Results from a prospective international cooperative group trial. Ann Surg Oncol. 2005;13(4):491–500.
92. Witte M: New research perspectives: angiogenesis update 2000. Presentation at the National Lymphedema Network Conference, Orlando, 2000.
93. Wittlinger, H, Wittlinger, G. Textbook of Dr. Vodder’s manual lymph drainage, ed 4. Brussels: Haug International, 1992.
94. Wyrick, SL, Waltke, LJ, Ng, AV. Physical therapy may promote resolution of lymphatic cording in breast cancer survivors. Rehabil Oncol. 2006;24(1):29–34.
95. SH Zhang, AK Sood, JI Sorosky et al. Preservation of the saphenous vein during inguinal lymphadenectomy decreases morbidity in patients with carcinoma of the vulva, Cancer 89(7): 1520-1525
*However, in edema, it can become positive and tend to keep fluid out of the tissues. This is the one “safety factor” that controls lymphedema (i.e., the fibrous tissue actually helps to increase tissue hydrostatic pressure [THP] and control the size of the limb to some extent).
*The benzopyrones have not been approved by the Food and Drug Administration (FDA) in the United States, and the oral Coumarin was decertified in Australia because of several cases of death associated with liver problems. Other oral benzopyrones are still available in Switzerland, France, and Germany. Topical Coumarin powder and ointment is still available through compounding pharmacists in the United States. Bioflavonoid liquid and tablets have been available in U.S. health food stores for years. They are combined with various other compounds (known as bioflavonoid complex) and necessitate taking a larger dose of the complex to receive the desired amount of bioflavonoid.