The Immune System
Immunology is the study of the physiologic mechanisms that allow the body to recognize materials as foreign and to neutralize or eliminate them. When the immune system is working properly, it protects the organism from infection and disease; when it is not, the failure of the immune system can result in localized or systemic infection or disease. In fact, the significance of a healthy immune system is apparent in states or diseases characterized by immunodeficiency, such as occurs in human immunodeficiency virus (HIV) infection or in people on immunosuppressive medication.
Without an effective immune system, an individual is at risk for the development of overwhelming infection, malignant disease, or both. Not all immune system responses are helpful, as in the case of organ or tissue transplant rejection.
Additionally, excessive or inappropriate activity of the immune system can result in hypersensitivity states, immune complex disease, or autoimmune disease. For a complete understanding of the immune system as it relates to injury, inflammation, and healing, the reader is encouraged to read this chapter along with Chapter 6.
TYPES OF IMMUNITY
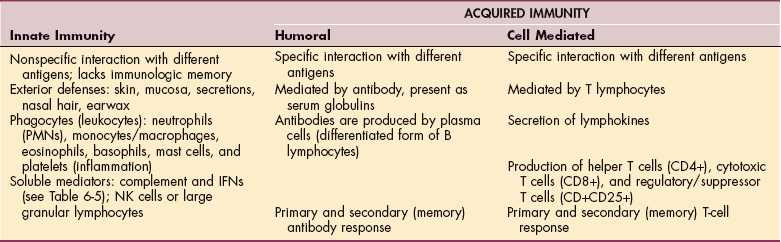
Two types of immunity are recognized: innate (natural or native immunity) and acquired immunity (adaptive or specific immunity). Innate immunity acts as the body’s first line of defense to prevent the entry of pathogens.
Two nonspecific, nonadaptive lines of defense are involved in innate immunity. Nonspecific refers to the fact that this part of the immune system does not distinguish between different types of invaders (e.g., bacteria, fungus, virus) and is nonadaptive; that is, it does not remember the encounter with specific invaders for future encounters. Each time that potential pathogen is introduced, the innate immune system reacts in the same predictable manner.
The first line of defense is the skin and its mucosal barriers, and the second is a nonspecific inflammatory response to all forms of cellular injury or death. Innate responses occur to the same extent no matter how many times the infectious agent is encountered (Fig. 7-1).
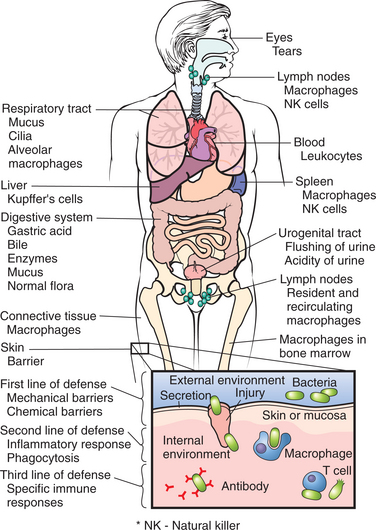
Figure 7-1 Natural protective mechanisms of the human body. (Reprinted from Damjanov I: Pathology for the health professions, ed 3, Philadelphia, 2006, Saunders.)
Acquired immunity is characterized by specificity and memory. The primary role of the immune system as a more specific line of defense (acquired immunity) is to recognize and destroy foreign substances such as bacteria, viruses, fungi, and parasites and to prevent the proliferation of mutant cells such as those involved in malignant transformation.
This type of immunity results when a pathogen gains entry to the body and the body produces a specific response to the invader. Acquired immunity has a memory so that when the same organism is encountered again, the body can respond even more rapidly to it and with a stronger reaction. The two components to acquired immunity (humoral immunity and cell-mediated immunity) are discussed in greater detail later in this section.
Acquired Immunity: Active or Passive Immunity
Acquired immune responses can occur as a result of active or passive immunity. Active immunity includes natural immunity and artificial immunity, which is intended or deliberate (Table 7-1).
Table 7-1

*Active immunity occurs when a person produces his or her own antibodies to the infecting organism; passive immunity occurs when the antibody is formed in another host and transferred to an individual.
Active acquired immunity refers to protection acquired by introduction (either naturally from environmental exposure or artificially by vaccination) of an antigen (microscopic component of pathogen that causes an immune response) into a responsive host.
The concept of vaccination is based on the fact that deliberate exposure to a harmless version of a pathogen generates memory cells but not the pathologic sequelae of the infectious agent itself. In this way, the immune system is primed to mount a secondary immune response with strong and immediate protection should the pathogenic version of the microorganism be encountered in the future.60 This type of immunity is expected to last a lifetime, but there are occasional exceptions.
Researchers are developing a new generation of vaccines to fight a variety of diseases. One of the most promising is the deoxyribonucleic acid (DNA) vaccine that allows DNA from a pathogen to be injected into the body, where cells accept the added DNA instructions and make antigens that the body can recognize and fight. Genetic manipulation allows researchers to overcome the greatest deterrent to vaccination—the ability of common pathogens such as influenza and pneumococcal bacteria to mutate too rapidly for a vaccine to match the latest version.
Some of the most promising new techniques are being investigated against malaria, cancer, ear infections, acquired immune deficiency syndrome (AIDS), sexually transmitted diseases (STDs), asthma, influenza, strep throat, diabetes, and hepatitis C. Improved administration of the vaccine with the use of mucosal sprays, skin patches, time-released pills, and genetically engineered foods to replace needle injections also is under development.
Passive acquired immunity occurs when antibodies or sensitized lymphocytes produced by one person are transferred to another. Preformed antibodies made in a laboratory or made by someone else are another form of passive immunity.
For example, the transplacental transfer of antibodies from mother to fetus, the transfer of antibodies to an infant through breast milk, or the administration of immune serum globulin (γ-globulin) provides immediate protection but does not result in the formation of memory cells and therefore provides only temporary immunity. This type of immunity (passively acquired) lasts only until the antibodies are degraded, which may be only a few weeks to months.
THE IMMUNE RESPONSE
See Table 7-2.
Antigens
Any foreign substance in the body that does not have the characteristic cell surface markers of that individual and is capable of eliciting an immune response is referred to as an antigen (from antibody generator). Bacteria, viruses, parasites, foreign tissue cells, and even large protein molecules that are recognized as antigens are called antigenic. On encountering an antigen, the immune system recognizes it as nonself, and the appropriate immune response is mounted against the antigen. A single bacterium contains hundreds of antigenic sites and therefore has multiple sites capable of stimulating an immune response.
The subunits of an antigen that elicit an immune response are called epitopes. These molecules protrude from the surface of an antigen and actually combine with an antibody (Fig. 7-2). Each antigen may display hundreds of epitopes. The more epitopes that are present, the greater is the antigenicity of a substance and the greater the immune response. Antibodies produced in response to an antigen are protein molecules structured in such a way that they only interact with the antigen that induced their synthesis, much as a key is made to fit a lock.
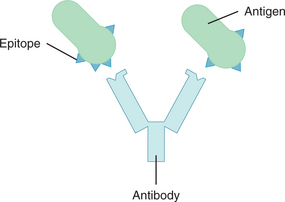
Figure 7-2 An antigen is recognized on the basis of shape. Epitopes protrude from the surface of an antigen and combine with the appropriate receptor of an antibody, much as a key fits a lock. For small antigens, the binding site on the antibody may be a pocket or cleft, but in most cases it more closely resembles an undulating surface.232 (Reprinted from Black JM, Hawks JH, Keene AM: Medical-surgical nursing: clinical management for positive outcomes, ed 7, Philadelphia, 2005, Saunders.)
The Major Histocompatibility Complex
Since the basis of immunity depends on the immune cells’ ability to distinguish self from nonself, all cells of the body contain specific cell surface markers or molecules that are as unique to that person as a fingerprint.
The immune system recognizes these cell markers and tolerates them as self; in other words, it produces self-tolerance. These cell markers are present on the surface of all body cells and are known as the major histocompatibility complex proteins. They were originally discovered on leukocytes and are commonly called human leukocytic antigens (HLAs). The six specific HLAs within the major histocompatibility complex markers are HLA-A, HLA-B, and HLA-C, referred to as class I antigens, and HLA-DP, HLA-DQ, and HLA-DR, referred to as class II antigens.136
Class I antigens are found on nucleated cells and platelets; class II antigens are found on monocytes, macrophages, B cells, activated T cells, vascular endothelial cells, Langerhans’ (skin) cells, and dendritic (nerve) cells. There is a third class of antigens (class III) including certain complement proteins (C2, C4, and factor B).
Cell markers are essential for immune function. They not only determine which antigens an individual responds to and how strongly, but they also allow immune system cells to recognize and communicate with one another.
HLAs are inherited and can predispose or increase an individual’s susceptibility to certain diseases (usually autoimmune) (see Table 40-20). Such diseases encompass many that affect the joints, endocrine glands, and skin, including rheumatoid arthritis, Graves’ disease, psoriasis, and many others. Not all people with a certain HLA pattern develop the disease, but they have a greater probability for its development than does the general population.
Innate Immunity
The innate immune system consists of all the immune defenses that lack immunologic memory. A characteristic of innate responses is that they remain unchanged no matter how often the antigen is encountered. Innate immune responses use phagocytic cells (neutrophils, monocytes, and macrophages); cells that release inflammatory mediators (basophils, mast cells, and eosinophils); and natural killer (NK) cells. The molecular components of innate responses include complement, acute-phase proteins, and cytokines such as the interferons.60
The innate immune response does not recognize every possible antigen but rather focuses on a few structures present in large groups of microorganisms. These structures are referred to as pathogen-recognition receptors.168
A key cellular component of innate immunity and one of the most intensely studied components in the last decade is the interdigitating dendritic cell. Cells of this type (e.g., Langerhans’ cells in skin) constantly but quietly endocytose extracellular antigens. Pattern recognition receptors on these dendritic cells are a part of the innate immune response to differentiate between potentially harmful microorganisms and self-constituents and include toll-like receptors that sense a broad range of microbial products.60
These types of receptors are specific for structures found exclusively in microbial pathogens (pathogen-associated molecular pattern). This differs from the adaptive immune system, which has a tremendous capacity to recognize almost any antigenic structure, but because antigen receptors are generated at random, they bind to antigens regardless of their origin—bacterial, environmental, or self.168
Exterior Defenses
As a covering for the entire body (with the exception of any openings), the skin offers the first and best line of protection (see Fig. 7-1), which is clearly demonstrated in cases of significant burns when infection becomes a major problem. The body openings also offer their own unique protection such as lysozyme in tears that can kill bacteria, waxy secretions in the ear canal to prevent bacteria from advancing inside, nasal hair, stomach acid, and unfavorable rapid pH change at the gastro-duodenal junction for ingested organisms, protective low pH vaginal secretions, acidic urine, and so on.
When organisms enter the body by penetrating the epithelial surface of the respiratory, gastrointestinal (GI), or genitourinary tract, biochemical defenses offer additional protection in the form of soluble mediators such as complement and cytokines (particularly interferons), phagocytes that engulf and destroy foreign particles, and NK cells that attack and destroy virus-infected cells and tumor cells.
Phagocytes.: Phagocytes are involved in nonspecific or innate immunity. These cells readily eat (ingest) microorganisms such as bacteria or fungi and kill them as a means of protecting the body against infection.
The two principal phagocytes are neutrophils and monocytes and are part of white blood cells (WBCs), or leukocytes. The five types of leukocytes are neutrophils, eosinophils, basophils, monocytes, and lymphocytes. Because of their granular appearance, neutrophils, eosinophils, and basophils are collectively referred to as granulocytes. Granulocytes are short lived (2 to 3 days) compared with monocytes and macrophages, which may live for months or years.
Phagocytes emigrate out of the blood and into the tissues in which an infection has developed, and each of these cell types has a specific phagocytic function in the immune system (see the section on Disorders of Leukocytes in Chapter 14). Neutrophils, eosinophils, basophils, and monocytes are classified as phagocytic leukocytes that function in nonspecific or innate immunity. A severe decrease in the blood level of these cells is the principal cause of susceptibility to infection in people treated with intensive radiotherapy or chemotherapy. These treatments suppress blood cell production in the marrow, resulting in deficiencies of these phagocytic cells.
Neutrophils, also referred to as polymorphonuclear cells (PMNs), derive from bone marrow and increase dramatically in number in response to infection and inflammation. Neutrophils can directly kill invading organisms but may also damage host tissues. In the process of phagocytosis, bacteria or debris is engulfed and then digested by enzymes contained within the neutrophils (see Fig. 6-18; see also discussion of phagocytosis in Chapter 6 and Fig. 6-17). Neutrophils die after phagocytosis; the accumulation of dead neutrophils and phagocytosed bacteria contributes to the formation of pus.
Monocytes circulate in the blood, but when they migrate to tissues they mature into macrophages, which means “large eaters.” The engulfment of a pathogen by a macrophage is an essential first step leading to a specific immune response. After neutrophils kill the invading organism and the process of phagocytosis has begun, macrophages appear to clear up the debris produced by the neutrophils and to kill any damaged but not dead bacteria or bacteria that are too large for neutrophils. Neutrophils and macrophages both have receptors for antibodies and complement so that the coating of microorganisms with antibodies, complement, or both enhances phagocytosis.3
After phagocytes digest the pathogens, antigenic material appears on their surface to identify them more specifically as foreign invaders. In this process, phagocytes (primarily macrophages) serve as antigen-presenting cells to introduce the pathogen to lymphocytes. The macrophage or antigen-presenting cell processes the pathogen and presents a small part of it, the epitope (see Fig. 7-2), to a specific cell of the immune system known as the helper or inducer lymphocyte, or T4 lymphocyte (also referred to as CD4 lymphocyte).
Microscopically, T lymphocytes appear identical, but they can be distinguished by means of distinctive molecules called cluster designations (CDs) located on their cell surface. For example, all mature T cells carry markers known as T2, T3, T5, and T7 (or CD2, CD3, CD5, and CD7). T4 (CD4) are the helper T cells, and T8 (CD8) are cytotoxic T cells. Another group of T lymphocytes are identified as NK cells for their ability to kill certain tumor cells and virus-infected cells without prior sensitization or activation.
To prompt the T4 lymphocyte to recognize the processed pathogen, the macrophage releases interleukin-1 (IL-1), a chemical messenger with many roles. In this way the macrophage processes the antigen and then signals the lymphocytes to stimulate the specific immune response.
Interleukins are one type of cytokine, a protein released by macrophages to trigger the immune response (see Cytokines later in this section). Some of the multiple functions of IL-1 include increasing the temperature set point in the hypothalamus; increasing serotonin in the brainstem and duodenum, causing sleep and nausea, respectively; stimulating the production of prostaglandins, leading to a decrease in the pain threshold, resulting in myalgias and arthralgias; increasing the synthesis of collagenases, resulting in the destruction of cartilage; and most importantly, kicking the T4 cells into action.
Macrophages also participate in the defense against tumor cells and secrete numerous molecules called monokines that assist in the immune and inflammatory response. Stimulation of macrophages can boost the immune response.
Eosinophils are the next group of leukocytes that participate in the innate immunity process. Eosinophils are derived from bone marrow and multiply in both allergic disorders and parasitic infestations. When organisms are too large for neutrophils and macrophages, eosinophils get within close proximity of the invading organisms and release the contents of their granules to kill them.
Basophils are WBCs (leukocytes) that circulate in peripheral blood and function similarly to mast cells in allergic disorders. Basophils and mast cells are located close to blood vessels throughout the body and have similar functional characteristics; mast cells contain histamine that dilates blood vessels when released.
Mast cells are derived from stem cells and travel in the blood in such small numbers they are not recognized as blood cells. Arriving basophils and mast cells cause an increase in blood supply in the area where the bacteria or viral antigen is located. This increase in circulation also helps bring more phagocytes to the area, thus counteracting bacteria indirectly. The increased circulation is accompanied by the feeling of congestion during an allergic reaction; antihistamines work by neutralizing the histamines and reducing the excessive immune (allergic) response.
The role of erythrocytes and platelets in immune responses is sometimes overlooked, but because they have complement receptors, they play an important part in the clearance of immune complexes consisting of antigen, antibody, and components of the complement system.60
Soluble (Inflammatory) Mediators
The complement system and interferons act as soluble inflammatory mediators along with phagocytes to destroy organisms that breach the first line of defense. The complement system consists of 20 serum proteins, which are key components in the acute inflammatory response designed to enhance immune function.
When activated, these proteins interact in a cascade-like process to assist immune cells by coating microorganisms so they can be more easily phagocytosed and to participate in bacterial lysis. In some cases the invading organisms are eliminated from the body. Sometimes the inflammation produced by the complement cascade (immune response) walls off the microorganism by forming, for example, a cyst or tubercle that protects the rest of the body from infection. (See the section on The Complement System in Chapter 6; also see Table 6-5.)
The second group of soluble mediators is the cytokines, especially interferons sometimes referred to as biologic response modifiers. They act as messengers, both within the immune system and between the immune system and other systems of the body, forming an integrated network that is highly involved in the regulation of immune responses.171
In addition to acting as messengers, some cytokines have a direct role in defense, such as the interferons. Interferons are produced by virally infected cells early in infection to limit the spread of the infection by protecting surrounding (noninfected) cells (interferons also inhibit tumor growth). Once a cell becomes infected by a virus, certain genes are turned on in the cell that will produce these interferons that coat the surrounding cells and make them viral resistant.
Natural Killer Cells
NK cells are large granular lymphocytes that are neither T nor B lymphocytes. The function of NK cells is to kill viruses, other intracellular microbe-infected cells, and tumor cells. NK cells recognize targets by first binding to potential target cells followed by interaction between activating and inhibitory receptors with ligands available on the target, and then integrating signals transmitted by these receptors, which determines whether the NK cells will detach and move on or stay and respond. NK cells respond by releasing cytotoxic granules and by secreting cytokines (Fig. 7-3).146
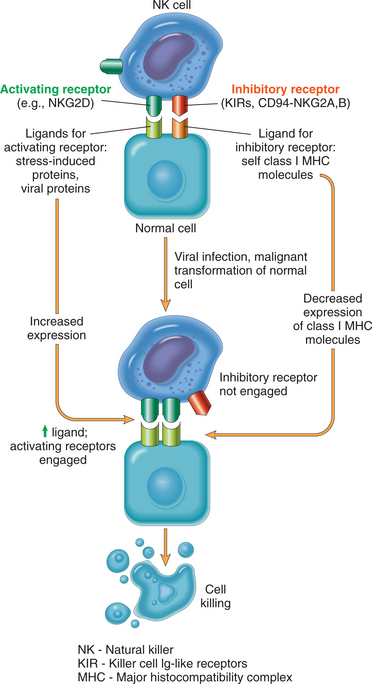
Figure 7-3 Schematic representation of NK cell receptors and cell killing. NK cells express activating and inhibitory receptors; some examples of each are indicated. Normal cells are not killed because inhibitory signals from normal major histocompatibility complex class I molecules override activating signals. In tumor cells or virus-infected cells, there is increased expression of ligands for activating receptors and reduced expression or alteration of major histocompatibility complex molecules, which interrupts the inhibitory signals, allowing activation of NK cells and lysis of target cells. (Reprinted from Kumar V: Robbins and Cotran: pathologic basis of disease, ed 7, Philadelphia, 2005, Saunders.)
Acquired Immunity
To establish an infection, the pathogen must first overcome numerous surface barriers and the innate immune responses (see Fig. 7-1). In these cases, acquired immunity is tailored to recognize each different type of organism and kill it.
The two types of acquired immune responses that occur are humoral immunity (also called immunoglobulin-related immunity) and cell-mediated immunity (also referred to as T-cell immunity). Although these two responses are often discussed separately, they are two arms of the immune system and work together; failure in one can alter the effectiveness of the other. These two types of responses overlap and interact considerably, but the distinction is useful in understanding how the immune system is activated (Fig. 7-4).
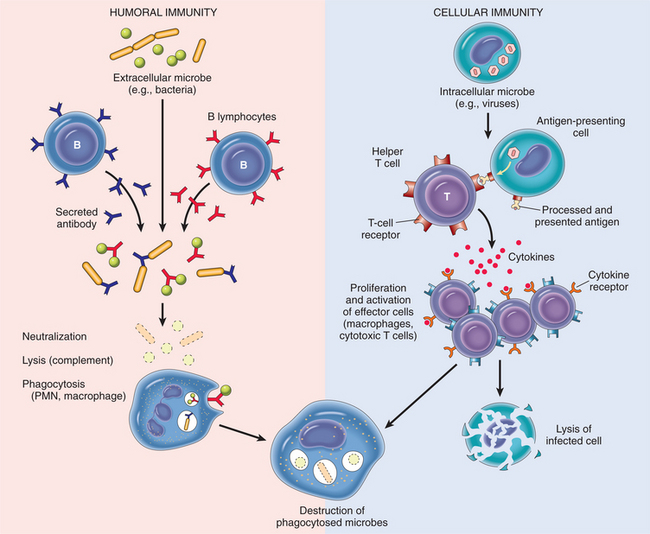
Figure 7-4 Humoral and cell-mediated immunity. (Reprinted from Kumar V: Robbins and Cotran: pathologic basis of disease, ed 7, Philadelphia, 2005, Saunders.)
The complexity of the cellular interactions that occur during acquired immune responses requires specialized microenvironments in which the relevant cells can collaborate efficiently. Because only a few lymphocytes are specific for any given antigen, T cells and B cells need to migrate throughout the body to increase the probability that they will encounter that particular antigen. In their travels, lymphocytes spend only about 30 minutes in the blood during each trip around the body.182 More specific information about T-cell function and migration is available.258
Acquired responses involve the proliferation of antigen-specific B and T cells, which occurs when the surface receptors of these cells bind to antigen and initiate the immune response involving immunoglobulins, antibodies, regulatory and suppressor T cells, and cytokines. The acquired immune system generates a highly diverse group of antigen receptors that allows the adaptive immune system to recognize virtually any antigen. However, the price for this diversity is the inability to distinguish foreign antigens from self-antigens.168
Humoral Immunity
The humoral immune response is mediated by antibodies present in different body fluids or secretions, such as saliva, blood, or vaginal secretions. Antibodies produced by B lymphocytes are very effective against organisms that are free floating in the body that can be easily reached and neutralized. B lymphocytes, or B cells, are called such because they originate in the bone marrow and then circulate throughout the extracellular fluid.
The surface of B lymphocytes is coated with immunoglobulin, and each B cell has a receptor (an antibody) that can recognize a specific foreign substance or antigen. When this happens, B cells change into protein-synthesizing cells known as plasma cells and memory B cells.
The plasma cell produces and secretes into body fluids a specific antibody to that antigen. Memory cells produced in connection with humoral immunity circulate among the blood, lymphoid system, and tissues for about 1 year or even longer. They are responsible for the more rapid and sustained (stronger) immune response that occurs with repeated exposure to the same antigen. This humoral response is particularly useful in fighting bacterial infections.
The B lymphocyte–plasma cell interaction is capable of producing five types of antibodies, or immunoglobulin (Ig), in response to specific antigens. Because of these antibodies the humoral immune response may be referred to as the antibody immune response. The five types of antibodies are IgG, IgM, IgA, IgD, and IgE; major functions of immunoglobulins are listed in Box 7-1.
IgM predominates in the primary or initial immune response and is the largest immunoglobulin; because of its size, it is found almost exclusively in the intravascular compartment. IgG is the major antibacterial and antiviral antibody and is the predominant immunoglobulin in blood; it is responsible for the protection of the newborn during the first 6 months of life and is the only immunoglobulin to cross the placenta. It is the major immunoglobulin synthesized during the secondary immune response (after IgM initially responds to foreign pathogens), conferring long-term or permanent immunity.
IgA defends external body surfaces, is the predominant immunoglobulin on mucous membrane surfaces, and is found in secretions such as saliva; breast milk (colostrum); urine; seminal fluid; tears; nasal fluids; and respiratory, GI, and genitourinary secretions.
IgD is the predominant antibody found on the surface of B lymphocytes, serves mainly as an antigen receptor, and may function in controlling lymphocyte activation or suppression. IgE is a primary factor in eliminating parasitic infections such as roundworms and is therefore significant in the immune responses of people in developing countries where adequate nutrition, hygiene, and primary medical care are lacking. IgE also functions during allergic reactions by activating the mast cells and releasing histamine in association with allergies, anaphylaxis, extrinsic asthma, and urticaria (hives). This response of IgE is a normal reaction but becomes excessive in people with allergies.
The type of antibody produced depends on genetic variability, the specific antigenic stimulus, and whether it is a first or subsequent exposure to that antigen. The humoral immune response is more rapid than the cell-mediated response and is more often a factor in resistance to acute bacterial infections. Humoral immunity can be transmitted to another person, either by inoculation or by maternal transfer via placenta or breast milk. This transfer is called passive immunity (see Table 7-1).
Cell-Mediated Immunity
Some organisms (all viruses and some bacteria) actually hide inside the cells where the antibodies cannot reach them. A second arm of the immune system (cell-mediated immunity or cellular immunity) with more specific cells (T lymphocytes) can recognize these hidden organisms, search them out, and destroy them on a cell-to-cell basis.
Lymphocytes originate from stem cells in the bone marrow and differentiate or mature into either B or T cells (Fig. 7-5; see also Fig. 21-6). The B cell (humoral immunity) is thought to mature and become immunocompetent in the bone marrow.
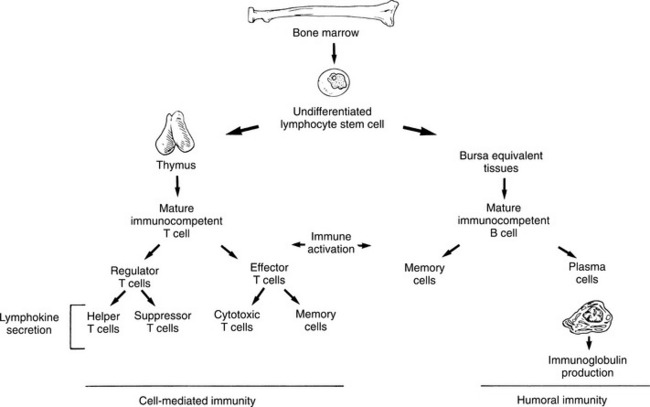
Figure 7-5 The pathway of lymphocyte maturation. Undifferentiated lymphocyte stem cells are derived from the bone marrow. B cells reach maturity within the bone marrow, but T cells must travel to the thymus to complete their development. Activation of either T or B cells by antigens leads to proliferation of immune cells that mediate either cell-mediated immunity or humoral immunity, respectively. (Reprinted from Black JM, Matassarin-Jacobs E, eds: Medical-surgical nursing: clinical management for continuity of care, ed 5, Philadelphia, 1997, W.B. Saunders, p 597.)
T lymphocytes, or T cells (cell-mediated immunity), are called such because the precursors of these cells start from the bone marrow but then mature in the thymus, located right behind the sternum, where it learns to discriminate self from nonself (see Fig. 7-8). Both T and B lymphocytes continuously circulate through blood, lymph, and lymph nodes.
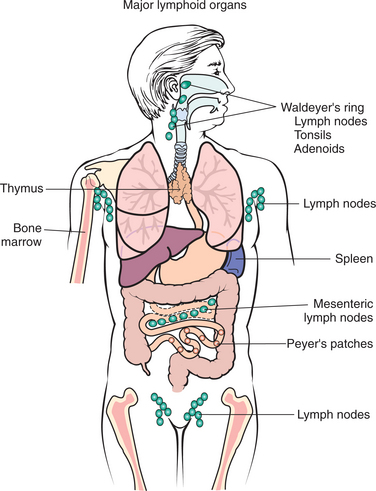
Figure 7-8 Immune system. Organs of the immune system are referred to as lymphoid tissues. The bone marrow and thymus are referred to as primary lymphoid organs since these organs are the central sites of all cells of the immune system and B-and T-cell differentiation, respectively. Immature lymphocytes migrate through the central lymphoid tissues and later reside as mature lymphocytes in the peripheral or secondary lymphoid tissues (e.g., lymph nodes, Peyer’s patches, tonsils, spleen, mucosa-associated lymphoid tissue, or MALT from the mouth to the rectum). (Reprinted from Damjanov I: Pathology for the health professions, ed 3, Philadelphia, 2006, Saunders.)
After interaction with a specific antigen, the activated lymphocyte produces numerous additional lymphocytes called sensitized T cells. This T-cell subpopulation has three primary functions. The most numerous of the T cells, helper T cells, constituting 75% of all T cells, assist the B cells to mature and produce antibody by secreting protein mediators called lymphokines. Many different lymphokines have been identified, including IL-1 and IL-2 (among others), and interferons.
Some of their functions include (1) helping B cells augment the production of antibodies, (2) activating macrophages and helping them destroy large bacteria, (3) helping other T lymphocytes (called cytotoxic T cells, or CD8 T cells) recognize and destroy virally infected cells, and (4) helping NK cells kill infected cells (Fig. 7-6).
Figure 7-6 T cells. The blue spheres seen in this scanning electron microscope view are T cells attacking a much larger cancer cell. T cells are a significant part of our defense against cancer and other types of foreign cells. (Reprinted from Thibodeau GA, Patton KT: The human body in health & disease, ed 4, St Louis, 2005, Mosby. Courtesy James T. Barrett.)
Helper T cells (CD4 T cells) themselves can be categorized on the basis of their cytokinetic profiles into T-helper type 1 (TH1) or T-helper type 2 (TH2) cells. TH1 produce interferon (IFN)-γ and tumor necrosis factor (TNF)-β and are important in assisting CD8 T-cell activation. TH2 cells produce IL-4, IL-5, and IL-13 and drive B-cell activation and antibody generation (Fig. 7-7). HIV destroys or inactivates these helper T cells and leaves the body at risk for infectious agents such as cytomegalovirus (CMV).
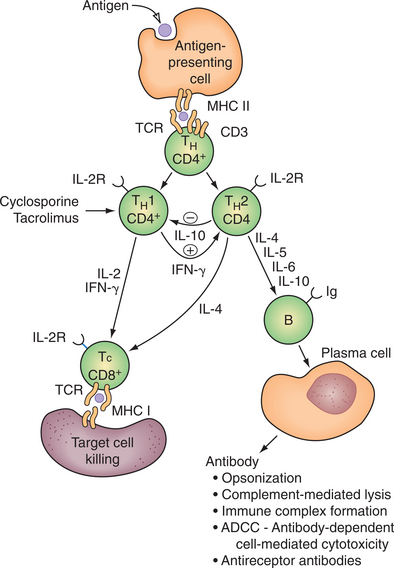
Figure 7-7 T-helper (CD4) cells can be divided into functionally distinct subsets (TH1 and TH2) based on their cytokine secretion profiles. Cytokines secreted by TH1 cells play a key role in cell-mediated immunity, whereas those produced by TH2 cells are important in B-cell stimulation and antibody production. An important feature of the TH1/TH2 cell paradigm is that cross-regulation of function between TH1 and TH2 cells occurs. Thus, for example, IFN-γ stimulates TH2 cells, whereas IL-10 inhibits TH1 cells. Cyclosporine and tacrolimus (FK-506) are potent inhibitors of IL-2 and IFN-γ production by TH1 cells. There is evidence, however, that they may spare IL-10 (cytokine synthesis inhibitory factor) production by TH2 cells. (Reprinted from Townsend CM: Sabiston textbook of surgery, ed 17, Philadelphia, 2005, Saunders.)
The immune system also consists of regulatory/suppressor T cells (CD4+CD25+) that suppress activation of the immune system and prevent pathologic self-reactivity, or autoimmune disease. Immunosuppressive cytokines tumor growth factor-β and IL-10 have been implicated in regulatory T-cell function.233
Cell-mediated immunity is responsible for the rejection of transplanted tissue, delayed hypersensitivity reactions (e.g., contact dermatitis), and some autoimmune diseases. Cell-mediated immunity is the basis for many skin tests (e.g., tuberculin test, allergy testing). Cellular immunity cannot be transferred passively to another person.
Clinical conditions that compromise the cell-mediated T-lymphocyte function include HIV infection and AIDS, with a progressive reduction in T4 lymphocytes over the duration of the illness. Other conditions known to affect T-cell number or responsiveness include stress, malignancy, general anesthesia, thermal injury, surgery, diabetes, and immunosuppressive drugs (including corticosteroids). Older adults (aged 65 years and older) show reduced numbers of circulating lymphocytes, and malnourished people show defects in most tests of T-cell function.
Summary of the Immune Response
Immune responses are initiated according to the type of antigen presented. Adaptive immune responses are generated in the lymph nodes, spleen, and mucosa-associated tissue, referred to as the secondary lymphoid tissues. For example, blood-borne antigens usually initiate responses in the spleen, whereas responses to microorganisms in tissues are generated in local lymph nodes.
Most pathogens are encountered after they are inhaled or ingested. Antigens entering the body through mucosal surfaces activate cells in the mucosa-associated lymphoid tissues, including the tonsils, adenoids, and Peyer’s patches (Fig. 7-8).61
Keeping in mind that innate immunity and acquired immunity function in tandem together (Fig. 7-9), and that within the acquired immune system humoral immunity and cellular immunity are also working simultaneously, a variety of immune responses can occur when an extracellular pathogen attempts to invade the body.
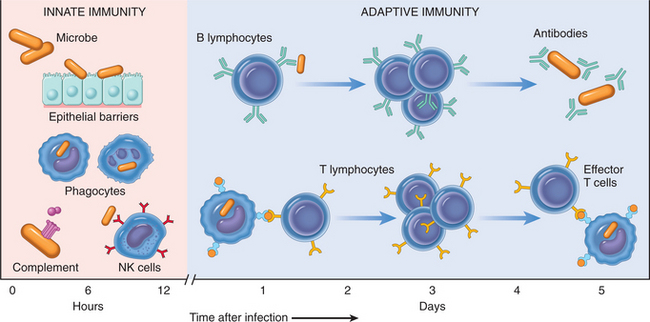
Figure 7-9 The principal mechanisms of innate immunity and adaptive immunity. (Reprinted from Kumar V: Robbins and Cotran: pathologic basis of disease, ed 7, Philadelphia, 2005, Saunders.)
If the pathogenic organism gets past the first line of defense (innate immunity) and is presented to the body, the following can happen: (1) a B lymphocyte recognizes it as a bacteria and produces antibodies that bind to it and neutralize it (humoral response); (2) a T lymphocyte recognizes it as a bacteria and produces cytokines to help the macrophages lyse and phagocytose the bacteria (cell-mediated response); (3) in the case of a virus, a cytotoxic T lymphocyte can recognize the cell and destroy it (cell-mediated response); and (4) the complement system can recognize the invading organism and destroy it (innate immunity).
In some instances, innate and acquired immunity interact with each other, such as when bacteria enter the body and the B lymphocyte recognizes it and produces specific antibodies (acquired immun- ity). Examples of this interaction include (1) antibodies (acquired immunity) binding to the bacteria, coating it, and making it available for phagocytosis by the phagocytes of the innate immune system; (2) bacteria being recognized by the B lymphocyte (acquired immunity) and coated with the antibody produced by the lymphocyte, with the complement (innate immunity) recognizing it and destroying it; (3) activation of cytotoxic T lymphocytes (acquired immunity) and NK cells (innate immunity), resulting in a direct attack on cells that have been transformed by a virus or a malignant process; and (4) the foreign invader being recognized by a T lymphocyte (from the acquired immune system), which then produces hormones (lymphokines) that help the macrophage (from the innate immune system) to destroy it.
Dysfunction of the immune system can contribute to diseases. For example, two general types of genetic alterations could lead to immunologic abnormalities: mutations that inactivate the receptors or signaling molecules involved in innate immune recognition and mutations that render them active all the time. The first type of mutation would be expected to result in various types of immunodeficiencies. The second type of mutation triggers antiinflammatory reactions and thereby contributes to a variety of conditions with an inflammatory component (e.g., asthma, allergy, arthritis, autoimmune diseases).168
AGING AND THE IMMUNE SYSTEM
Aging is accompanied by immune dysregulation as immune function declines with increasing age; this is described as the oxidative (free radical) theory of aging (see the section Cellular Aging in Chapter 6). Changes are observed in both the innate and acquired immunity defenses; the end result is reduced resistance to pathogens and increased incidence of tumors and autoimmune disorders.
Changes in Innate Immunity
Exterior defenses are affected by thinning of the skin, making older adults more prone to pressure ulcers and increasing openings for bacteria to enter the body. Decreased acidity of the GI tract, shallower breathing with decreased air exchange, less-acidic urine, and a less-elastic bladder that retains more urine are all ways exterior defenses are affected by aging, thus contributing to reduced effectiveness of the innate immune system.
Phagocytes (neutrophils and monocytes or macrophages) show decreased function with aging. Eosinophils accumulate in fewer numbers at sites of infection with age, perhaps predisposing us to parasitic infections as we age. Basophils are characterized by reduced degranulation with aging, although mast cells show no change in numbers or histamine release with aging. Platelet aggregation increases with aging, perhaps contributing to increased clot formation and decreased peripheral circulation.
Soluble mediators are characterized by increased serum levels of complement produced by the liver, perhaps as a compensatory mechanism, but this remains unproved. A decreased production of interferons by monocytes occurs with increasing age.
NK cell (large granular lymphocytes) cytotoxicity is impaired, as well as production of cytokines by activated NK cells.94
Changes in Acquired Immunity
Decline in humoral and cell-mediated immunity occurs with aging. Older adults have particular difficulty in mounting protective immune responses to newly encountered antigens, such as West Nile virus. Such responses depend on naive T and B cells, and aging is associated with a decline in their production.
Thymic involution and T-and B-lineage–specific defects in early lymphoid development in the bone marrow appear to explain this decline. However, it cannot be assumed that changes in the lymphoid compartment are entirely responsible for the poor quality of immune responses in the older adult.
The quality of the immune response may be associated with other cellular changes, such as defective cytoskeletal assembly and hyperglycosylation of proteins, telomerase shortening, replicative senescence, and failed susceptibility to apoptosis.
In human beings, CD8 T-cell expansions are often accompanied by loss of CD28, an important receptor of accessory signals during T-cell activation. Similar changes are seen in CD4 T-cell population with dysregulation of cytokine production.31
Although B-cell function appears relatively intact, there is a decline in antibody production that may be due to inappropriate or insufficient T-cell help. While this change in antibody is reflected in a decline in the level of antibody upon immunization, there may also be decreases in the affinity of the antibodies produced. Any or all of these age-associated changes in immune response may contribute to age-associated changes in response to viral infections, resulting in increased morbidity and mortality rates in older adults.176
Older adults are one of the highest risk groups for influenza infection, with at least 50% of hospitalizations and greater than 80% of all deaths from influenza occurring in individuals older than 65 years.
Each year the availability of vaccinations for influenza is prioritized according to age, general health, and co-morbidities. However, efficacy of the vaccine in the older adult population is only at 30% to 40% compared to 70% to 80% in younger individuals. Pneumonia is the fourth leading cause of death and the first among infectious diseases in the older adult population. Similarly, efficacy of the current pneumococcal vaccine only reaches 44% to 61% in individuals older than 65 years.256 The diminished cell-mediated immunity can result in the reactivation of dormant infections, such as herpes zoster and tuberculosis.
Age-related increase in autoimmune activity is both a cellular and a humoral phenomenon, and limited studies suggest that reduced humoral and cellular immunocompetence, reduced suppressor cell activity, and increased autoantibody activity are all associated with reduced survival rates.
FACTORS AFFECTING IMMUNITY
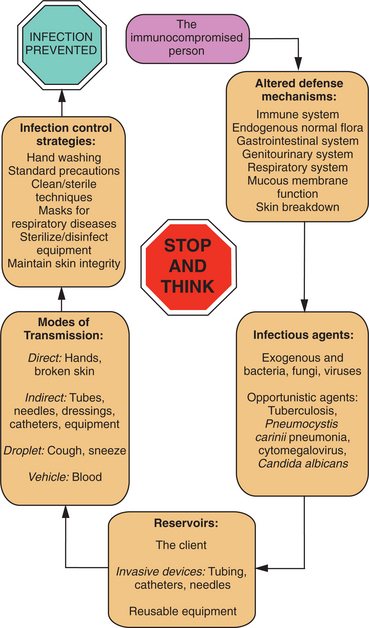
In addition to the effects of aging, other factors can affect the immune system. These factors may include nutrition; environmental pollution and exposure to chemicals that influence the host defense; prior or ongoing trauma or illnesses; medications; splenectomy (removal of the spleen); influences of the enteric, endocrine, and neurochemical systems; stress; and psychosocial-spiritual well-being and socioeconomic status.
These factors, as well as clinical conditions that contribute to an immunocompromised state, are listed in Box 7-2. Sleep deprivation has also been shown to have important effects similar to stress on the immune system by reducing cellular immunity.14,207 Some factors, such as the iatrogenically introduced interventions listed and sexual practices, do not alter the immune system directly but increase a person’s exposure to pathogens.
New information is being discovered about the sensory functions of the intestine and how neural, hormonal, and immune signals interact. Representatives of all the major categories of immune cells are found in the gut or can be rapidly recruited from the circulation in response to an inflammatory stimulus. The gut immune system has 70% to 80% of the body’s immune cells, and the protective blocking action of the secretory response in the gut is crucial to the integrity of the GI tract immune function and host defense.18 Studies suggest that the development and expression of the regional immune system of the GI tract are independent of systemic immunity, but this remains an area of debate and investigation.
Nutritional status can have a profound effect on immune function. Nutrients have fundamental and regulatory influences on the immune response of the GI tract and, therefore, on host defense. Reduction of normal bacteria in the gut after antibiotic treatment or in the presence of infection may interfere with the nutrients available for immune function in the GI tract.
Severe deficits in calories, protein intake, or vitamins such as vitamin A or vitamin E can lead to deficiencies in T-cell function and numbers. Deficient zinc intake can profoundly depress both T-and B-cell function. Zinc is required as a cofactor for at least 70 different enzymes, some of which are found in lymphocytes and are necessary for their function.
Secondary zinc deficiencies may be associated with malabsorption syndrome, chronic renal disease, chronic diarrhea, or burns or severe psoriasis (loss of zinc through the skin). Dietary changes may alter aspects of immunity, although research in this area is ongoing. Additionally, morbid obesity may alter the immune system by creating a vulnerability to certain diseases, including cancer.
Some medications (e.g., cancer chemotherapeutic agents) profoundly suppress blood cell formation in the bone marrow. Other drugs (e.g., analgesics, antithyroid medications, anticonvulsants, antihistamines, antimicrobial agents, and tranquilizers) induce immunologic responses that destroy mature granulocytes.
Many drugs also affect B-and T-cell function, especially against antigens that require the interaction of helper T cells and B cells for antibody production. These complications have been observed since the advent of potent immunosuppressive (e.g., corticosteroids) and chemotherapeutic drugs as treatment of people with autoimmune diseases, transplants, or cancer. Depression of B-and T-cell formation is manifested as a progressive increase in infections with opportunistic microorganisms (e.g., Pneumocystis carinii, CMV, and Candida albicans and other fungi).
Surgery and anesthesia can also suppress both T-and B-cell function for up to 1 month postoperatively.161 Because of the invasive nature of any surgical procedure and because defects in immunity have been described in most major illnesses, it is logical to assume that the majority of hospitalized surgical clients are immunocompromised to some degree.
Surgery to remove the spleen results in a depressed humoral response against encapsulated bacteria, especially Streptococcus pneumoniae, Haemophilus influenzae, Staphylococcus aureus, the group A streptococci, and Neisseria meningitidis (see Chapter 8).
Burns cause increased susceptibility to severe bacterial infections as a result of decreased external defenses (intact skin), neutrophil function, decreased complement levels, decreased cell-mediated immunity, and decreased primary humoral responses. Blood serum from clients with burns also contains nonspecific immunosuppressive factors that will suppress all immune responses, regardless of the antigen involved.
The relationship between stress, psychosocial-spiritual well-being, and socioeconomic status and susceptibility to disease through depressed immune function has become an area of intense research interest. In the past, there were anecdotal reports of increased incidence of infection, diseases, and malignancy associated with periods of both intense and relatively minor stress (see Table 2-6).
In the new and expanding world of psychoneuroimmunology, almost any stress seems capable of altering immune function. The role of stress in the development of pathology is discussed in Chapter 3. Likewise, the role of environmental pollution and exposure to chemicals on susceptibility to immune system dysfunction and development of disease is discussed in Chapter 5.
INTERACTIONS BETWEEN THE IMMUNE AND CENTRAL NERVOUS SYSTEMS
The role of the nervous and endocrine systems in homeostasis has been shown to include interaction with the immune system. The study of interactions between the immune system, the endocrine system, and the CNS has been called neuroimmunology. Newer terms include neuroimmunomodulation, psychoneuroimmunology, and neuroimmunoendocrinology (see Chapter 1).
Two pathways link the brain and the immune system: the autonomic nervous system (ANS) and neuroendocrine outflow via the pituitary. Immune responses alter neural and endocrine functions and, in turn, neural and endocrine activity modifies immunologic function.269
Many regulatory peptides and their receptors previously thought to be limited to the brain or to the immune system are now known to be expressed by both. It is now known that endocrine factors can alter immune function, immune responses can alter both endocrine and CNS responses, and communication between the CNS and the immune system is bidirectional.9,13
This means the immune system has the capacity not only to sense the presence of foreign molecules, but also to communicate this to the brain and neuroendocrine system. The interaction between the immune and neuroendocrine systems is termed bidirectional communication.
Findings that link immune and neuroendocrine function may help explain how emotional state or response to stress can modify a person’s capacity to cope with infection or cancer and influence the course of autoimmune disease. Whether emotional factors can influence the course of autoimmune disease, cancer, and infection in human beings is a subject of intense research, but studies so far have shown reduced lymphocyte sensitivity with chronic distress.11
The CNS can be involved in immune reactions arising from within the brain or in response to peripheral immune stimuli. Activated immunocompetent cells such as monocytes, lymphocytes, and macrophages can cross the blood-brain barrier and take up residence in the brain, where they secrete their full repertoire of cytokines and other inflammatory mediators such as leukotrienes and prostaglandins. All aspects of immune and complement cascades can occur in the brain because of these nerve-macrophage communications. The CNS modulates immune cells by direct synaptic-like contacts in the brain and at peripheral sites, such as the lymphoid organs (see Fig. 7-8).245
A number of cytokines called neurocytokines (e.g., IL-1, IL-2, IL-4, and IL-6; neuroleukin; and TNF-α) are formed by glia, the supporting structure of nervous tissue. The activation of cytokines in the CNS occurs in response to local tissue injury and can lead to profound changes in neural functions, ranging from mild behavioral disturbances to anorexia, drowsiness, sleep disturbances, coma, dementia, and the destruction of neurons. The activation of cytokines in neural tissue by injury or toxins has a positive benefit as well by stimulating the production of nerve growth factor.
On the basis of studies using animal models, researchers suggest that the brain can regulate immunocompetence. Much of this neuroimmunomodulation takes place through the hypothalamic-pituitary system and sympathetic nervous system, with the latter by the release of catecholamines at autonomic nerve endings and from the adrenal medulla.
The principal immunoregulatory organs (lymph nodes, thymus, spleen, and intestinal Peyer’s patches) are abundantly supplied by autonomic nerve fibers. Sensory neurons contain a variety of neurotransmitters and neuropeptides that can influence lymphocyte function.
EXERCISE IMMUNOLOGY
The effect of physical activity and exercise (aerobic, endurance, and resistance) on the immune and neuroimmune systems has been an area of research interest. A brief summary of the results is presented here, but a more detailed accounting of exercise and the immune system and future direction for studies is available.180,187
Depending on the intensity, activity or exercise can enhance or suppress immune function. In essence, the immune system is enhanced during moderate exercise. Moreover, regular, moderate physical activity can prevent the neuroendocrine and detrimental immunologic effects of stress.75
In contrast to the beneficial effects of moderate exercise on the immune system, strenuous or intense exercise or long-duration exercise such as marathon running is followed by impairment of the immune system. Intense exercise can suppress the concentration of lymphocytes, suppress NK cell activity, and leave the host open to microbial agents, especially viruses that can invade during this open window of opportunity, and may lead to infections.
Extreme and long-duration strenuous exercise appears to lead to deleterious oxidation of cellular macromolecules. The oxidation of DNA is important because the oxidative modifications of DNA bases are mutagenic and have been implicated in a variety of diseases, including aging and cancer.195
Effect on Neutrophils and Macrophages
Exercise triggers a rise in blood levels of neutrophils (PMNs) and stimulates phagocytic activity of neutrophils and macrophages. The exercise-evoked increase in the PMN count is greater if the exercise has an eccentric component, such as downhill running. If the exercise goes beyond 30 minutes, a second, or delayed, rise in PMNs occurs over the next 2 to 4 hours while the exerciser is at rest.
This delayed rise in PMNs is probably the result of cortisol, which spurs release of PMNs from the bone marrow and hinders the exit of PMNs from the bloodstream.165 After brief, gentle exercise, the PMN count soon returns to baseline, but after prolonged, strenuous exercise, this return to normal may take 24 hours or longer.70
In many instances, exercise enhances macrophage function and can increase antitumor activity in mice, but many questions still remain regarding the mechanism(s) by which short-or long-term exercise affects macrophage function.267
Effect on Natural Killer Cells
Most researchers agree that the number of NK cells and the function or activity of these cells in the blood increase during and immediately after exercise of various types, duration, and intensity.187
This phenomenon, referred to as NK enhancement, is temporary and seems to be the result of a surge in epinephrine levels and from cytokines released during exercise. NK enhancement by exercise occurs in everyone regardless of sex, age, or level of fitness training; however, once a person is accustomed to a given exercise level, the NK enhancement falls off, suggesting it is a response not to exercise per se, but to physiologic stress.
After intense exercise of long duration the concentration of NK cells and NK cytolytic activity declines below preexercise values. Maximal reduction in NK cell concentrations and lower NK cell activity occurs 2 to 4 hours after exercise.187 Although this depression in NK cell count seems too brief to have major practical importance for health, there may be a cumulative adverse effect in athletes who induce these changes several times per week. Further study is warranted before specific exercise guidelines are determined.231
Effect on Lymphocytes
Brisk exercise (even brief, heavy exertion such as maximal bicycle ergometry for 30 or 60 seconds) increases the WBC count in proportion to the effort.99,178 This exercise-induced increase in WBCs (including lymphocytes and NK cells) is largely the result of the mechanical effects of an increased cardiac output and the physiologic effects of a surge in serum epinephrine concentration. Lymphocytes may be recruited to the circulation from other tissue pools during exercise (e.g., the spleen, lymph nodes, or GI tract). The number of cells that enter the circulation is determined by the intensity of the stimulus.187
The number of lymphocytes in circulation increases during exercise but decreases below the normal levels for several hours after intense exercise. Decreased numbers of lymphocytes are associated with decreased lymphocyte responsiveness and antibody response to several antigens after intense exercise.125
The effects of intense exercise on secondary antibody response in older adults remain unknown. In one study with older mice, no adverse effect(s) of multiple bouts of intense exercise on antibody levels occurred.125 In contrast to intense exercise, moderate exercise training enhances secondary antibody response in young animals and is mediated in part by endogenous opioids.124,126 Primary antibody response is not influenced by exercise training.
Effect on Cytokines
Strenuous exercise, defined as exercising at a minimum of 80% of maximal oxygen consumption (VO2max), can suppress immune function and damage enough tissue to evoke the acute phase response in human beings.186 This complex cascade of reactions can modulate immune defense by activating complement and spurring the release of TNF, interferons, interleukins, and other cytokines.
Plasma IL-6 increases in an exponential fashion with exercise (without muscle damage) and is related to exercise intensity, duration, the mass of muscle recruited, and endurance capacity. The antiinflammatory effects of IL-6 are demonstrated by the fact that IL-6 stimulates the production of antiinflammatory cytokines IL-1ra and IL-10.189
Furthermore, IL-6 stimulates the release of soluble TNF-α receptors, but not IL-1β and TNF-α, and appears to be the primary inducer of the hepatocyte-derived acute phase proteins, many of which have antiinflammatory properties.
Therefore IL-6 induces an antiinflammatory environment by inducing the production of IL-1ra and IL-10, but it also inhibits the production of proinflammatory cytokine TNF-α. The possibility exists that, with regular exercise, antiinflammatory effects of an acute bout of exercise will protect against chronic systemic low-grade inflammation, but such a link between the acute effects of exercise and the long-term benefits has not yet been proven.
However, regular exercise protects against diseases associated with chronic low-grade systemic inflammation. This long-term effect of exercise may be ascribed to the antiinflammatory response elicited by a short-term bout of exercise, which is partly mediated by muscle-derived IL-6.189
Exercise and Apoptosis
The role of apoptosis, or programmed cell death, in exercise is the focus of much research in the area of exercise science. Apoptotic cell death differs morphologically and biochemically from necrotic cell death, although both appear to occur after exercise.
Accelerated apoptosis has been documented to occur in a variety of disease states, such as AIDS and Alzheimer’s disease, and in the aging heart. In striking contrast, failure to activate this genetically regulated cell death may result in cancer and certain viral infections. It is surmised that exercise-induced apoptosis is a normal regulatory process that removes certain damaged cells without a pronounced inflammatory response, thereby ensuring optimal body function.191
Exercise and Infection
As determined by experimental studies, the effects of exercise stress on disease lethality clearly vary with the type of and time the exercise is performed. In general, exercise or training before infection has either no effect or decreases morbidity and mortality rates.
Exercise during the incubation period of the infection appears either to have no effect or to increase the severity of infection. However, a recent study showed that moderate exercise, when performed after influenza virus infection but before flu symptoms, resulted in reduced mortality rates in mice.156
Several epidemiologic studies on exercise and upper respiratory tract infection report an increased number of related symptoms (based on self-report rather than clinical verification) in the days after strenuous exercise (e.g., a marathon race), whereas moderate training has been claimed to reduce the number of symptoms. However, in neither strenuous nor moderate exercise have these symptoms been causally linked to exercise-induced changes in immune function.187
IMMUNODEFICIENCY DISEASES
In immunodeficiency, the immune response is absent or depressed as a result of a primary or secondary disorder. Primary immunodeficiency reflects a defect involving T cells, B cells, or lymphoid tissues. Secondary immunodeficiency results from an underlying disease or factor that depresses or blocks the immune response.
Primary Immunodeficiency
The recognition of impaired immunity in children 50 years ago has resulted in a tremendous increase in knowledge of the functions of the immune system. More than 95 inherited immunodeficiency disorders have now been identified. Genetically determined immunodeficiency can cause increased susceptibility to infection, autoimmunity, and increased risk of cancer (Fig. 7-10).
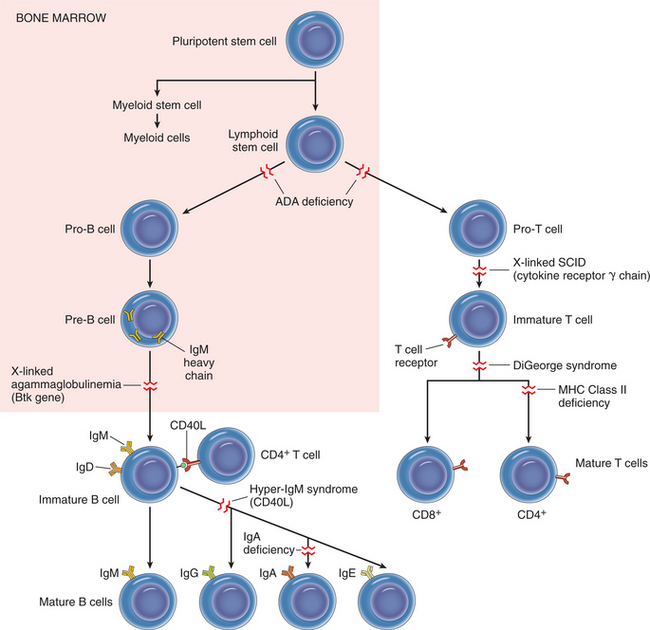
Figure 7-10 Scheme of lymphocyte development and sites of block in primary immunodeficiency diseases. The affected genes are indicated in parentheses for some of the disorders. ADA, Adenosine deaminase; CD40L, CD40 ligand; SCID, severe combined immunodeficiency. (Reprinted from Kumar V: Robbins and Cotran: pathologic basis of disease, ed 7, Philadelphia, 2005, Saunders.)
The defects may affect one or more components of the immune system, including T cells, B cells, NK cells, phagocytic cells, and complement proteins. No further discussion of these conditions is included in this book because the therapist rarely encounters these congenital conditions. A review of the pathophysiology of primary immunodeficiency is available.26
Secondary Immunodeficiency
Secondary immunodeficiency disorders such as leukemia and Hodgkin’s disease follow and result from an earlier disease or event. Multiple, diverse, and nonspecific defects in the immune defenses occur in viral and other infections and also in malnutrition, alcoholism, aging, autoimmune disease, diabetes mellitus, cancer, chronic disease, steroid therapy, cancer chemotherapy, and radiation. More specific causes such as AIDS also contribute to secondary immunodeficiency.
Iatrogenic Immunodeficiency
Immunodeficiency induced by immunosuppressive drugs, radiation therapy, or splenectomy is referred to as iatrogenic immunodeficiency. Immunosuppressive drugs fall into several categories, including cytotoxic drugs, corticosteroids, cyclosporine, and antilymphocyte serum or antithymocyte globulin.
Cytotoxic drugs kill immunocompetent cells while they are replicating, but since most cytotoxic drugs are not selective, all rapidly dividing cells are affected. Not only are lymphocytes and phagocytes eliminated, but these drugs also interfere with lymphocyte synthesis and release of immunoglobulins and lymphokines.
Other effects of this nonselectivity of cytotoxic drugs are discussed in Chapter 5 and may include bone marrow suppression with neutropenia, anemia, and cytopenia; gonadal suppression with sterility; alopecia; hemorrhagic cystitis; and vomiting, nausea, and stomatitis. The risk of lymphoproliferative malignancy is also increased.
Corticosteroids are used to treat immune-mediated disorders because of their potent antiinflammatory and immunosuppressive effects. Corticosteroids stabilize the vascular membrane, blocking tissue infiltration by neutrophils and monocytes, thus inhibiting inflammation. They also kidnap T cells in the bone marrow, causing lymphopenia. Corticosteroids also appear to inhibit immunoglobulin synthesis and interfere with the binding of the immunoglobulin to antigen.
Cyclosporine (immunosuppressive drug) selectively suppresses the proliferation and development of helper T cells, resulting in depressed cell-mediated immunity. This drug is used primarily to prevent rejection of organ transplants but is also being investigated for use in several other disorders. Antilymphocyte serum or antithymocyte globulin is an anti–T-cell antibody that reduces T-cell number and function, thereby suppressing cell-mediated immunity. It has been used effectively to prevent cell-mediated rejection of tissue grafts and transplants (see the section on Immunosuppressants under Adverse Drug Reactions in Chapter 5).
Radiation therapy is cytotoxic to most lymphocytes, inducing profound lymphopenia, which results in immunosuppression. Irradiation of all major lymph node areas, a procedure known as total nodal irradiation, is used to treat disorders such as Hodgkin’s lymphoma. It is being investigated for its effectiveness in severe rheumatoid arthritis and lupus nephritis and the prevention of kidney transplant rejection.
Splenectomy increases a person’s susceptibility to infection, especially with pyogenic bacteria such as Streptococcus pneumoniae. This risk of infection is even greater when the person is very young or has an underlying reticuloendothelial disorder. These people should be observed carefully for any signs of infection (see Box 8-1).
Consequences of Immunodeficiency
People who are immunocompromised from any of the immunodeficiency disorders are at increased risk of developing infection because their impaired immune system does not provide adequate protection against invading microorganisms. Normal mechanical defense mechanisms may be affected (respiratory, GI systems). Body flora that are normally harmless, such as Candida, may become pathogenic and a source of infection.
Additional risk factors for people who are already immunocompromised include poor physiologic and psychologic health status, old age, coexistence of other diseases or conditions, invasive procedures (e.g., surgery, invasive lines), and treatments (e.g., chemotherapy, radiation therapy, bone marrow transplantation).
The weakened immune system can cause the person to become susceptible to common everyday infectious agents, such as influenza viruses and S. aureus, as well as the more exotic organisms such as Histoplasma capsulatum and Toxoplasma gondii.
Acquired Immune Deficiency Syndrome
HIV can be considered an infection of the immune system, resulting in progressive and ultimately profound immune suppression. Currently one of the most widely publicized diseases, AIDS was first recognized in homosexual men in 1981 (the earliest sample of HIV-infected blood dates back to 1959,111 but computer analysis suggests an emergence date of 1930137).
The virus thought to be responsible for the transmission of AIDS was identified as HIV in July 1986. As new discoveries were made the classification scheme later included two subtypes as HIV-1 and HIV-2, with several strains of HIV-1 further identified. This text deals primarily with HIV-1 (the cause of most of the AIDS cases in the United States), hereafter referred to as HIV.
AIDS is characterized by progressive destruction of cell-mediated (T-cell) immunity and changes in humoral immunity and even elements of autoimmunity because of the central role of the CD4+ T lymphocyte in immune reactions (see the section on Monocytes and Macrophages earlier in this chapter for a discussion of CD4+ cells).
The resultant immunodeficiency leaves the affected person susceptible to opportunistic infections, including unusual cancers, tuberculosis, and other abnormalities that characterize this syndrome. For example, HIV-positive individuals are nearly two and a half times more likely than HIV-negative persons to have a recurrence of tuberculosis239 and 8 to 10 times more likely to develop Hodgkin’s disease when compared with the general population.152
Additionally, 25% to 40% of Americans with HIV are believed to be infected with the hepatitis C virus (HCV), primarily among injection drug users and those with hemophilia as a result of blood products used to treat the hemophilia.7 Mortality rates are higher and life expectancy lower in people with hemophilia who are HIV positive.193
Definition
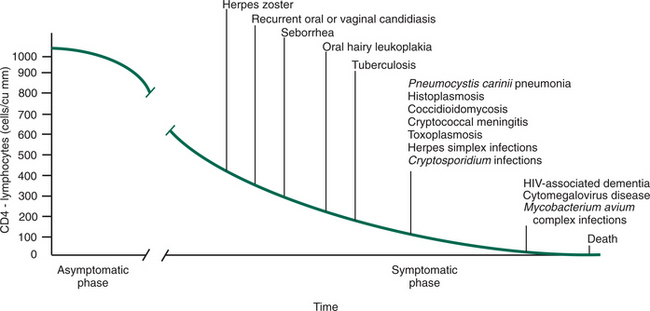
The Centers for Disease Control and Prevention (CDC) revised the definition of AIDS in 1992 to include those who have HIV-1 and a CD4 count below 200/mL (the normal CD4 lymphocyte count is 600 to 1200/mL) or 14% of the total lymphocyte count, even if the person has no other signs or symptoms of infection. The AIDS definition was further expanded in 1993 to include diseases affecting women (e.g., cervical cancer) and people with tuberculosis or depressed immune systems.
The term HIV infection includes the entire spectrum of illness from initial diagnosis to full-blown expression of AIDS. Three distinct points identify this continuum: (1) asymptomatic HIV seropositive, (2) early symptomatic HIV, and (3) HIV advanced disease (AIDS).
Not everyone who is exposed to HIV becomes infected, and not everyone who is infected develops AIDS. The explanation for this phenomenon remains unknown, but researchers have shown that infection with HIV and progression to AIDS are controlled by both host genetic factors and viral factors.
The HLA region in human beings controls immune response functions and influences susceptibility to infectious diseases, including HIV. There are HLA alleles associated with susceptibility to and protection from HIV infection, and these differ among ethnic groups.213 Alleles are situated at one or more sites on chromosomes and carry genetic information that determines a specific genetic characteristic or trait. A single amino acid change in HLA molecules has a substantial effect on the rate of progression to AIDS.87
Incidence and Prevalence
Since the first AIDS cases were reported in the United States in 1981, the number of cases and deaths among people with AIDS increased rapidly during the 1980s, followed by substantial declines in new cases and deaths in the late 1990s.
AIDS remains an epidemic of vast proportions in other countries such as in South Africa, where the number of deaths has exceeded the estimated 25 million caused by the Black Death in the fourteenth century.91,244 An increase among women has been especially steep in East Asia, Eastern Europe, and Central Asia.68
In 2007 there were 2.5 million newly infected individuals worldwide, including 700,000 children younger than 15 years. Of the 33.2 million people worldwide living with HIV/AIDS, 30.7 million are adults (with numbers almost equal between men and women), 2.5 million are children younger than 15 years, and two thirds (20 million) live in Sub-Saharan Africa. Twenty percent (6.6 million) are in Asia and the Pacific.91
New infections in the United States have declined to an estimated 40,000 per year (down from a peak of 150,000 per year in the mid-1980s). The number of people living with AIDS in the United States is the highest ever reported at more than 1 million.254 Deaths from AIDS declined 63% from approximately 52,000 to 19,000 in the late 1990s.103
In the United States the greatest impact of the epidemic is among injection drug users (IDUs), sex workers, and men who have sex with men (MSM), with an 8% increase in this last group91,223,253 and among racial/ethnic minorities.33 Increases have been observed in the number of cases attributed to heterosexual transmission among minority women and women older than 50 years. The total number of people living with AIDS has increased as deaths have declined.175,253
Half of all new cases among adults and adolescents are caused by IDU (up from 28% in 2000),35 and almost an equal amount (41%) are infected through MSM. Women account for more than one quarter of all new HIV/AIDS diagnoses. Women of color are especially affected. The rate of AIDS diagnoses for African-American women is approximately 23 times the rate for white women and four times the rate for Hispanic women34; women in general are infected through the use of shared injection drug needles and sex with infected men (Fig. 7-12).253
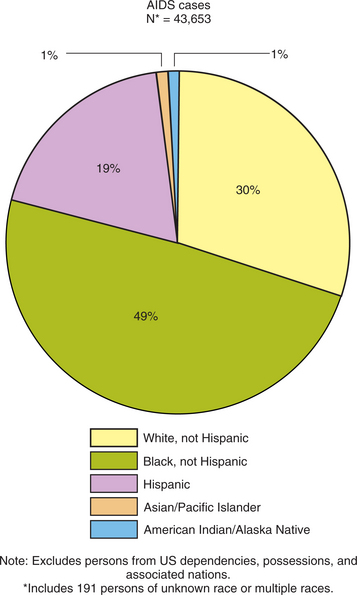
Figure 7-12 Proportion of AIDS cases and population by race/ethnicity, reported in 2004 (50 states and Washington, DC). (Reprinted from Centers for Disease Control and Prevention: HIV/AIDS surveillance by race/ethnicity, 2006, www.cdc.gov/hiv/topics/surveillance/resources/slides/epidemiology/index.htm.)
Most people diagnosed with AIDS in the United States are aged 20 to 49 years; however, the number of adolescents with HIV in the United States doubles every year. Teens account for one quarter of the cases of new STDs reported each year, and AIDS in older adults accounts for 11% of all AIDS cases.175
Etiologic Factors, Transmission, and Risk Factors
The primary cause of AIDS is the type 1 retrovirus (HIV). Transmission of HIV occurs by exchange of body fluids (notably blood and semen) and is associated with high-risk behaviors.
High-risk behaviors include unprotected anal and oral sex, including having six or more sexual partners in the past year; sexual activity with someone known to carry HIV; exchanging sex for money or drugs; or injecting drugs. HIV is not transmitted by fomites (e.g., coffee cups, drinking fountains, or telephone receivers) or casual household or social contact.
As mentioned, IDU also continues to play a key role in the HIV epidemic. In some large drug-using communities, HIV seroincidence and seroprevalence among IDUs have declined in recent years.64 This decline has been attributed to several factors, including increased use of sterile injection equipment, declines in needle sharing, shifts from injection to noninjection methods of using drugs, and cessation of drug use.2
However, IDU among young adult heroin users has increased substantially in some areas, an indication that, as with sexual behaviors, changes to less-risky behaviors may be difficult to sustain.116,253
Transmission of HIV varies by gender. In 2004, 57% of male HIV infections were related to MSM, 19% were related to IDU, and 17% of the infections resulted from heterosexual sex. In the same year in females, 70% of HIV infections were related to heterosexual sex and 29% resulted from IDU.37
A woman is twice as likely as a man to contract HIV infection during vaginal intercourse, and the presence of some STDs greatly increases the likelihood of acquiring or transmitting HIV infection. The rates of gonorrhea and syphilis are higher among women of color, especially between the ages of 15 and 24 years.34
Overall, MSM has been the most common mode of exposure among people reported with AIDS, followed by IDU and heterosexual contact. To avoid social isolation, discrimination, or verbal or physical abuse, many MSM, especially young and minority MSM, do not disclose their sexual orientation.
Young MSM who do not disclose their sexual orientation (nondisclosers) are thought to be at particularly high risk for HIV infection because of low self-esteem, depression, or lack of peer support and prevention services that are available to MSM who are more open about their sexuality (disclosers).
Additionally, one in three nondisclosers reports having recent female sex partners, suggesting that nondisclosing MSM may have an important role in HIV and STD transmission to women.223 This might be particularly true for black nondisclosing MSM, of whom approximately one in five is infected with hepatitis B virus (HBV) and one in seven is infected with HIV.229
The chief determinant of whether HIV is transmitted during heterosexual intercourse is the viral load in the infected partner’s bloodstream. Viral load refers to the number of viral RNA particles present in the blood and correlates strongly with the stage of disease.
Viral load tests measure the amount of HIV-specific RNA, which is highest at the time of seroconversion when antibodies appear in the serum and the person is considered positive for HIV. This is a useful measurement for determining the effectiveness of drug treatment and also directly correlates with the risk of perinatal transmission in pregnant women with HIV.
Transmission does not occur when serum HIV-1 RNA levels are less than 1500 copies/mL (i.e., low viral load).203 Uncircumcised men have a greater risk of contracting HIV through sexual contact than do circumcised men. The thinner epithelial lining of the glans penis may be susceptible to increased trauma during sexual activity, increasing the likelihood of viral transmission.147
Nearly all transmissions of HIV through transfusion of blood or blood products occurred before testing of the blood supply for HIV antibody and self-referral programs were initiated in 1985. People with hemophilia were especially vulnerable and susceptible to transmission.
In 1991, approximately 70% of people with hemophilia had seroconverted to being positive for HIV; however, since 1986 no further HIV transmission has occurred by that mechanism. The number of people reported with AIDS who were exposed through blood transfusions was 284 in 2000, down from a peak of 1098 in 1993.175
The number of perinatally acquired AIDS cases peaked in 1992, followed by a sharp decline from that point in time.175
Ethnicity is not directly related to increased AIDS risk, but it is associated with other determinants of health status such as poverty, illegal drug use, access to health care, and living in communities with a high prevalence of AIDS. Adolescents are one of the groups at greatest risk for HIV infection, particularly minority inner-city youth. Runaway and homeless youth are especially likely to engage in high-risk sexual activity. The use of amphetamines, ecstasy, and amyl nitrate is associated with increased frequency of unprotected anal sex, especially among homosexual and bisexual individuals younger than 23 years.67
The prevalence of HIV infection is nearly five times higher in incarcerated populations than for the general population. HIV transmission among inmates in correctional and detention facilities is associated with MSM and potentially tattooing. Sex among inmates occurs, and laws or policies prohibiting sexual contact between inmates are difficult to implement and enforce. Condom distribution is unavailable in most correctional facilities.
Although no case of HIV transmission by tattooing has been documented, the procedure carries a theoretical risk for transmission if nonsterile equipment is used. In some instances, receipt of a tattoo has been associated with HIV seroconversion.249
Pathogenesis
The rapid convergence of information from diverse areas of AIDS research makes it impossible to present the most up-to-date information. Scientists are reporting new discoveries daily about the pathogenesis of HIV disease.
The natural history of AIDS begins with infection by the HIV retrovirus detectable only by laboratory tests. This retrovirus predominantly infects human T4 (helper) lymphocytes (also known as the CD4 cell), the major regulators of the immune response, and destroys or inactivates them. Macrophages and B cells are also infected (Fig. 7-13).
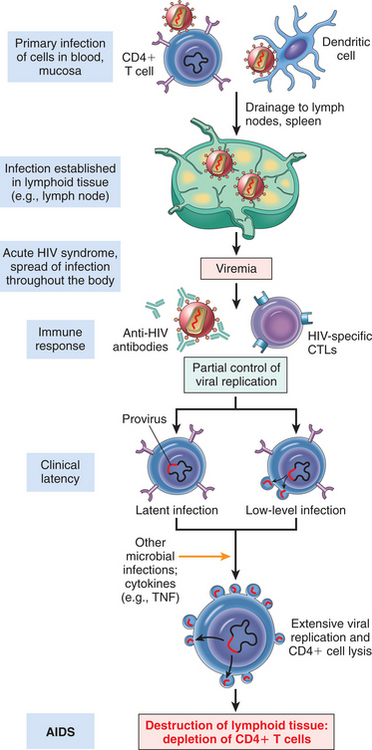
Figure 7-13 Pathogenesis of HIV-1 infection. Initially, HIV-1 infects T cells and macrophages directly or is carried to these cells by Langerhans cells. Viral replication in the regional lymph nodes leads to viremia and widespread seeding of lymphoid tissue. The viremia is controlled by the host immune response (not shown), and the patient then enters a phase of clinical latency. During this phase, viral replication in both T cells and macrophages continues unabated, but there is some immune containment of virus (not illustrated). There continues a gradual erosion of CD4+ cells by productive infection (or other mechanisms, not shown). Ultimately, CD4+ cell numbers decline, and the patient develops clinical symptoms of full-blown AIDS. Macrophages are also parasitized by the virus early; they are not lysed by HIV-1, and they may transport the virus to tissues, particularly the brain. (Reprinted from Kumar V: Robbins and Cotran: pathologic basis of disease, ed 7, Philadelphia, 2005, Saunders.)
All human cells contain DNA and RNA, known collectively as nucleic acids. The DNA is located in the cell nucleus, and the RNA is located in the cytoplasm of the cell. Viruses contain only one of the nucleic acids and are categorized as belonging to the DNA group (e.g., herpes and mononucleosis) or the RNA group (e.g., measles and mumps). HIV is classified as a lentivirus, a subclass of retroviruses that contain RNA. When the RNA virus initiates replication in the living host cell, it must convert its RNA genetic information into a DNA template to replicate.
HIV is unique in that despite the body’s immune responses after initial infection, some HIV invariably escapes. Large amounts of HIV have been discovered hiding in the immune cells lining the surface of adenoids.75 This information alters the formally held view that HIV is a slow or covert process, when in fact the virus’s growth continues in the period between infection and the onset of AIDS. It is theorized that the use of a preventive vaccine could stop hidden viral production.
Once HIV enters the body, cells containing the CD4 antigen, including macrophages and T4 cells, serve as receptors for the HIV retrovirus, allowing direct passage of the infection into target cells previously identified (e.g., GI tract, uterine cervical cells, and neuroglial cells). After invading a cell, a virus particle called a virion injects the core proteins and the two strands of viral RNA into the cell.
As a retrovirus, HIV contains reverse transcriptase, an enzyme required for successful infection by the virus. Before viral replication can occur in the host cell, this enzyme must copy all the genetic information of the virus from viral RNA to viral DNA. Once the viral genome is transcribed, it can be integrated into the host’s DNA and duplicated many times (Fig. 7-14).
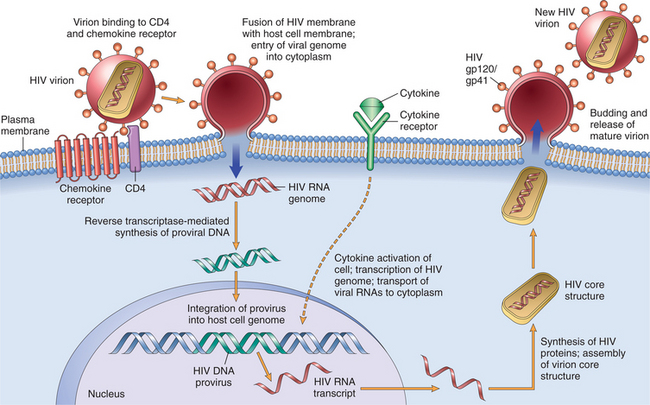
Figure 7-14 The life cycle of HIV. The steps from viral entry to production of infectious virions are illustrated. (Reprinted from Kumar V: Robbins and Cotran: pathologic basis of disease, ed 7, Philadelphia, 2005, Saunders.)
Replication of the virus can cause cell death, although the person remains asymptomatic. Seroconversion (becoming positive for HIV) usually takes place during the first 3 to 6 weeks of this replication process but can take longer. After a few months, very little virus is found in the blood; only HIV antibodies remain in the serum.
During the asymptomatic period (also called the early stage), the virus migrates from the serum into the tissues to infect CD4 cells in lymph tissue. The virus continues to kill the CD4 cells in the lymph nodes, although the CD4 count remains above 500 cells/mm3 (average CD4 cell count is 1000 cells/mm3).
The regulation of HIV expression by modules secreted by immune cells is even more complex than previously realized. The initially expanded clones that can kill HIV-infected cells are found in the bloodstream rather than in the lymph nodes, where the virus is replicating. The gene within HIV that stops production of the CD4 T lymphocytes and enhances T lymphocyte apoptosis, vpr, has now been identified.
Research continues to focus on understanding how the vpr gene prevents these disease-fighting cells from dividing. New drugs to block the gene’s actions and allow immune cells to continue multiplying and fighting HIV may be possible; alternately, researchers are studying this phenomenon in hopes of developing a live HIV vaccine.
Once all the cells are depleted, the virus once again enters the blood to infect any remaining lymphocytes and clinically apparent disease occurs. By the time this happens, the immune system has been compromised and is ineffective and unable to mount a specific immune response to these virions. The immune system dysfunction is even more exaggerated if the host has become further immunocompromised by opportunistic diseases.
HIV infection leads to profound pathology, either directly by destruction of CD4+ cells, other immune cells, or neuroglial cells, or indirectly through the secondary effects of CD4+ T-cell dysfunction and resultant immunosuppression.
The decline in CD4 cells results in progressive loss of immune system function and the development of a wide variety of clinical signs and symptoms (see Table 7-3). This describes the middle stage or symptomatic phase of AIDS when CD4 count ranges between 200 and 500 cells/mm3. A decrease in the CD4 count to less than 200 cells/mm3 and an elevated viral load occur during the late-stage or advanced disease associated with the development of opportunistic infections (Fig. 7-15).
This infectious process may result in one or more of the following: (1) immunodeficiency with opportunistic infections and unusual malignancies; (2) autoimmunity such as rheumatoid arthritis, lymphoid interstitial pneumonitis, hypergammaglobulinemia, and production of autoimmune antibodies; and (3) neurologic dysfunction, including HIV-associated dementia, HIV encephalopathy, and peripheral neuropathies.
HIV has an extremely high mutation rate even within a single individual, producing competing strains of the same virus that fight for survival against the weapons produced by the immune system. Researchers are studying how different strains of HIV use cell surface molecules, in addition to the CD4 molecule, to bind to and enter target cells.
Another consideration is how different strains of HIV have a preference for certain cells; strains that infect macrophages and T cells are the main ones found early in the disease. Later, strains appear that replicate efficiently in T cells but not in macrophages. Finally, mutations in the human genes for HIV co-receptors may help explain why some people do not become infected despite repeated exposure and why some who are infected may have different rates of disease progression.
These long-term nonprogressors are HIV-infected individuals characterized by the absence of disease, low viral loads, and stable or even increasing CD4+ T-cell counts for prolonged periods.184
Clinical Manifestations
HIV infection manifests itself in many different ways (Table 7-3) and differs between adult and pediatric populations. Great variation exists among individuals as to the amount of time that passes between acute HIV infection, the appearance of symptoms, the diagnosis of AIDS, and death.
Asymptomatic Stage (CD4 Count of 500 Cells/mm3 or More).: During the early stage, the person demonstrates laboratory evidence of seroconversion (positive for HIV) but remains asymptomatic. Some individuals develop an acute, self-limiting infectious mononucleosis-like illness or a subtle, viral-like syndrome, followed by a period of clinical latency that may last a decade or more.
During the asymptomatic period, the infected person is clinically healthy and capable of normal daily activities, normal work habits, and unrestricted level and duration of exercise. Fatigue and generalized lymphadenopathy with swollen and firm lymph glands may be reported during this stage.
Early Symptomatic Stage (CD4 Count between 200 and 500 Cells/mm3).: As the infection progresses and the immune system becomes increasingly more compromised, a variety of symptoms may develop, including persistent generalized adenopathy; nonspecific symptoms such as diarrhea, weight loss, fatigue, night sweats, and fevers; neurologic symptoms resulting from HIV encephalopathy; or an opportunistic infection (e.g., P. carinii pneumonia [PCP], CMV, toxoplasmosis) or malignancy.
CMV can cause peripheral neuropathy and HIV retinitis (with possible blindness); PCP produces pulmonary symptoms such as dyspnea on exertion, nonproductive cough, and weight loss; and toxoplasmosis is a parasitic disease that affects the CNS. In addition to the symptoms that may occur with opportunistic diseases, treatment with multiple medications can cause adverse side effects, sometimes creating a confusing clinical picture.
More than half the adults with HIV in this stage report fatigue that limits physical and recreational activities. Half the adults who report fatigue are unable to attend school or work.226
HIV Advanced Disease (AIDS; CD4 Count of 200 Cells/mm3 or Less).: The neurologic manifestations of more advanced HIV disease are numerous and can involve the central, peripheral, and autonomic nervous systems. The CNS appears to be more commonly attacked by HIV than the peripheral nervous system.
HIV or AIDS encephalopathy, also referred to as HIV-associated dementia (HAD), formerly called AIDS dementia complex, is a primary infection of the brain by HIV. Symptoms can vary and are listed in Table 7-3. In the most advanced stages of the disease, severe dementia, mutism, incontinence, and paraplegia may occur. A detailed summary of nervous system disorders associated with HIV, including treatment, is available.82,224,255
Dermatologic conditions are common and can be extensive, including malignancies; bacterial, viral, and fungal infections (Fig. 7-16); and reactions to drug treatment. Cutaneous manifestations of HIV can present as dry flaking skin, telangiectasias, and thinning of the skin (and hair).
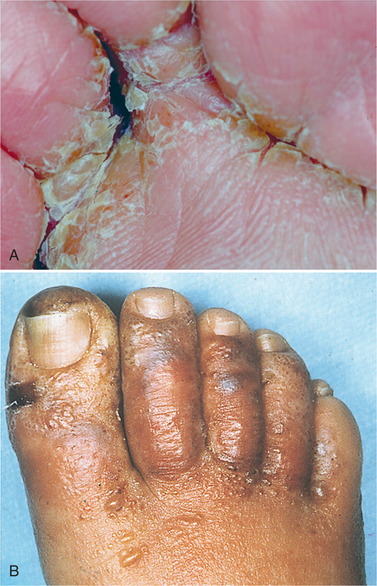
Figure 7-16 Tinea pedis. A, Fungal infections such as tinea pedis (also known as athlete’s foot) often begin between the toes and extend to the surface of the toes and foot. Red, itching skin may begin to peel or cause foot odor. B, The condition can progress, as shown here. Skin changes can become even more severe, with complete destruction of the nail beds (not shown). (A Reprinted from Seidel H: Mosby’s guide to physical examination, ed 5, St Louis, 2003, Mosby. B Reprinted from Cohen J, Powderly WG: Infectious diseases, ed 2, St Louis, 2004, Mosby.)
The prevalence of conditions such as seborrheic dermatitis, psoriasis, Reiter syndrome, acquired ichthyosis, Kaposi’s sarcoma, and scabies is on the rise. Kaposi’s sarcoma (purple nodular skin lesions) predominantly affects homosexual men (Figs. 7-17 and 7-18). HIV-associated nutritional disorders may also contribute to nail and hair changes. HIV-associated wounds may occur as a result of Kaposi’s sarcoma, herpes simplex virus, syphilis, IDU (“tracks”), candida and fungal infections, postoperative infections, and herpes zoster (see Fig. 8-7).108
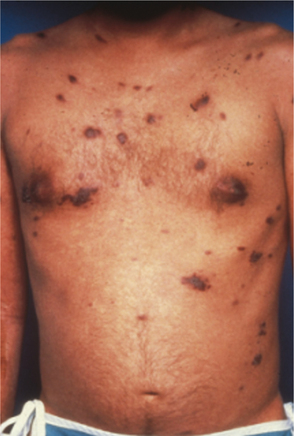
Figure 7-17 Kaposi’s sarcoma. There is a reduced incidence of Kaposi’s sarcoma in the HIV population because of better treatment, so fewer people progress this far in the disease. With better treatment have come fewer complications. Kaposi’s sarcoma appears primarily in the upper body, sometimes starting as a bulbous lesion on the end of the nose but also involving the face, chest, and lymph nodes. (Courtesy Julie Hobbs, Kingwood Medical Center, Kingwood, TX.)
Pain Syndromes.: Pain syndromes seen in HIV-infected individuals are divided into three groups: pain directly related to HIV infection or immunosuppression, pain caused by HIV diagnostic procedures and treatment, and pain unrelated to AIDS or its treatment (e.g., diabetic neuropathy, discogenic pain).
Painful sensory peripheral neuropathy is the most commonly reported pain syndrome, followed by pain associated with extensive Kaposi’s sarcoma and other dermatologic conditions, headache, abdominal pain, chest pain, and arthralgias and myalgias. Women infected with HIV experience pain more often and with greater intensity than men; women also have unique gynecologic syndromes related to opportunistic infections and cancers of the pelvis and genitourinary tract.248
Peripheral neuropathy, disease-or drug-induced myopathy, and musculoskeletal pain syndromes occur most often in advanced stages of HIV disease but can occur at any stage of HIV infection and may be the presenting manifestation. Neuropathic conditions in the AIDS population may develop as a result of neurotoxic antiretroviral medications, vitamin deficiencies resulting from poor nutrition, metabolic abnormalities, and opportunistic infections such as CMV.83
Peripheral neuropathies affect a large portion of people with AIDS and are usually distal, symmetric, and predominantly sensory, but other parts of the body may be affected such as the face or trunk. Peripheral neuropathies generally present in a stocking-glove distribution, with the feet and legs most commonly affected. Involvement of the upper extremities is less common and often occurs much later in the disease process.
The pain of peripheral neuropathy is characterized by burning, tingling, contact hypersensitivity, proprioceptive losses, and, in severe cases, secondary motor deficits. In the individual with HIV and newly acquired neuropathy with a strong major motor component, vasculitis may be the underlying cause (see the section on Vasculitis in Chapter 12).
AIDS is associated with neuromusculoskeletal diseases such as osteomyelitis, bacterial myositis, and infectious (reactive) arthritis. Osteonecrosis, osteopenia, and osteoporosis are increasingly observed in clients with HIV disease. Avascular necrosis (osteonecrosis) of the femoral head(s) has been reported with the use of antiretroviral therapy containing protease inhibitors.77
The increased incidence of osteonecrosis in HIV/AIDS may be due to the use of protease inhibitors or possibly as a result of an increased frequency of risk factors previously associated with osteonecrosis, such as hyperlipidemia, corticosteroid use, alcohol abuse, and hypercoagulability.225 It is possible that HIV disease itself may have an independent effect on bone loss regardless of antiretroviral therapy.166,185 Others suggest a link between lipodystrophy and bone loss.202
Musculoskeletal pain syndromes are associated with the wasting process in AIDS referred to as HIV wasting syndrome. HIV wasting is characterized by a disproportionate loss of metabolically active tissue, specifically body cell mass (i.e., tissue involved with glucose oxidation, protein synthesis, and immune system function).
These conditions occur secondary to low food intake, altered metabolism, and poor nutrient absorption with manifestations such as extreme weight loss, chronic diarrhea, unexplained weakness, fever, and malnutrition (e.g., mineral and vitamin deficiencies, especially vitamin B deficiency).
Studies have shown a clear relationship between vitamin B12 deficiency and dysfunction of the CNS and peripheral nervous system. Some of the clinical abnormalities of the nervous system seen in people with HIV are similar to those that have been described in those with vitamin B12 deficiency.
Lower genital tract infections and HIV are major causes of morbidity and mortality among women.247 As the HIV epidemic affects more women, increasing cases of pelvic inflammatory disease (PID) are being reported.114,115 Pain is a common symptom in all these conditions. PID is discussed in greater detail in Chapter 20.
Rheumatologic manifestations are transient or subtle and appear more often as HIV disease progresses. The arthritis can be severe and does not necessarily respond to conventional medications. Polymyositis involves bilaterally symmetrical proximal muscle weakness, and arthritis may precede or accompany seroconversion.
There has been a decline in reactive arthritis, psoriatic arthritis, and various forms of connective tissue disease for adults, but new syndromes such as the immune reconstitution infection following highly active antiviral therapy (HAART) may occur months later. Most cases reported resolve without intervention.30 Arthritis with spondyloarthropathy-like features is common in children with HIV infection and may be the presenting symptom.46 More detailed information regarding rheumatic disorders in HIV disease is available.228
HIV-associated myopathy presents with a progressive painless weakness in the proximal limb muscles. The weakness is symmetrical and often involves the muscles of the face and neck. This type of myopathy may occur in individuals with HIV at every stage of illness.
Muscle biopsies have shown necrosis of muscle fibers with and without inflammatory infiltrates. No evidence has been found of direct HIV infection of the muscle, and the underlying cause has not been determined. The fact that these clients improve on corticosteroids or plasmapheresis may point to an underlying autoimmune defect. Drug-induced myopathy is discussed in Chapter 6.
Lipodystrophy or lipodystrophic syndrome (LDS), a syndrome of defective fat metabolism, dyslipidemia, and insulin resistance, manifests as central fat accumulation (Fig. 7-19) with visceral fat deposition documented by computed tomographic scans. It is a common problem that occurs soon after starting HAART. Loss of fat occurs in the arms, leg, or face with concomitant fat deposits in the abdomen, breasts (men and women), and back of the neck (“buffalo hump”; see Fig. 11-7).

Figure 7-19 LDS in clients with HIV/AIDS has become more prevalent as a result of HAART treatment. LDS with cardiac complications from hyperlipidemia affects men and women, with fat deposits in the upper body and breasts contributing to body image problems, emotional trauma, and cardiac involvement. Additional fat deposits in the upper thoracic area and abdomen with loss in the extremities are seen in this client with AIDS. (Courtesy Julie Hobbs, Kingwood Medical Center, Kingwood, TX.)
AIDS-related lymphomas (ARLs), including Burkitt’s lymphoma, non-Hodgkin’s lymphoma, Hodgkin’s lymphoma, and other more uncommon types (e.g., primary effusion lymphoma) are more likely to occur in HIV-infected people when compared with the general population. ARLs are now the second most common cancer associated with HIV after Kaposi’s sarcoma and increase with time after infection. The incidence of non-Hodgkin’s lymphoma appears to be declining with the use of HAART, whereas the incidence of Hodgkin’s lymphoma may actually have increased in the HIV-infected population.152
ARL can occur at any CD4+ level, but the risk is increased with declining CD4+ lymphocyte counts.1,101 The course of disease is often aggressive, with extranodal metastases involving unusual sites such as the jaw, heart, body cavities, gallbladder, skin, and soft tissues.1
HAART has had little impact on the incidence of human papilloma virus–associated tumors (e.g., cervical cancer in women and anal cancer most commonly in MSM).21 The rising incidence of anal cancer among men with AIDS may be related to increased longevity with HAART and the consequent increased time at risk for the development of malignancy and/or the result of greater use of cytologic screening.65
Cardiopulmonary diseases continue to be an important cause of illness and death in people with HIV infection. Bacterial pneumonia, bronchitis, tuberculosis (pulmonary and extrapulmonary), and CMV are common opportunistic diseases in the HIV/AIDS population. Emphysema, asthma, and pulmonary hypertension are also observed in this population.
Cardiac involvement (heart and blood vessels) occurs as a result of a combination of the HIV infection, medical management, and secondary opportunistic infections. Both HIV infection and HAART can cause changes in lipid and glucose metabolism as well as elevation of blood pressure, promoting the development of atherosclerosis.
Cardiovascular diseases have become a major cause of mortality among HIV-infected people who respond well to antiretroviral therapy. Myocardial infarction, cardiomyopathy, pericardial effusion, and pericarditis are some other cardiovascular conditions that occur as a result of HIV and/or its treatment. There is increasing evidence to suggest that cardiovascular disease appears earlier and more often among HIV-infected adults than in the general public.113,169
MEDICAL MANAGEMENT
HIV prevention in the United States has placed the primary focus on persons who are not HIV infected to help them avoid becoming infected. Reducing sexual and drug-using risk behavior has been the main emphasis of public health prevention programs. The CDC’s overarching HIV prevention goal is to reduce the number of new HIV infections and to eliminate racial and ethnic disparities by promoting HIV counseling, testing, and referral and by encouraging HIV prevention among persons living with HIV and those at high risk for contracting the virus.254
However, further reduction of HIV transmission will require new strategies, including increased emphasis on appropriate routine screening, identification of new cases, partner notification, increased availability of sustained treatment, and prevention services for those infected. The addition of a simple, rapid HIV test may help overcome some of the traditional barriers to early diagnosis and treatment of infected persons. The CDC also recommends routine HIV testing of all pregnant women and routine screening of any infant whose mother was not screened.22,118
Recent reports suggest that behavioral changes often are not maintained and that a substantial number of HIV-infected persons continue to engage in behaviors that place others at risk for HIV infection or revert to risky sexual behavior after adopting safer sexual behavior.116 This altered behavior is referred to as “fatigue prevention.”
Two simple but effective interventions have been shown to limit the horizontal spread of HIV: condoms and counseling. Sex education and the practice of abstinence (or, less effectively, the use of condoms) and reducing or eliminating risky drug abuse behavior, including the provision of clean needles, has not been enough to stop the spread of AIDS, and no vaccine is available.
Restricting sex to partners of the same serostatus does not protect against transmission of other STDs or the possibility of HIV coinfection or superinfection (resistance to two or more classes of antiretroviral drugs) unless condoms are used correctly and consistently.116
Controlling the AIDS epidemic requires sustained prevention programs in all affected communities, particularly programs targeting MSM, sexually active individuals with multiple partners, IDUs, and minorities. Continuing the ABCs (abstinence, being faithful, condoms) prevention education is important with sexually active individuals undergoing treatment.
Even though potent antiviral treatment can eradicate HIV from the blood, these drugs cannot completely eliminate the virus from the body, especially in semen. Although antiretroviral therapy reduces shedding of HIV in semen, thereby reducing HIV transmissibility, a substantial portion of people with HIV may still be infectious and may have drug-resistant strains of the virus.10 Safe sex practices should continue to be reinforced in all people with HIV.
Unfortunately prevention fatigue has made prevention of HIV transmission less attainable as time goes by. Two drugs already used in combination, known as Truvada (tenofovir/Viread and FTC/Emtriva), to treat HIV show promise in animal studies as a possible preventive treatment.
Human tests are beginning in the United States and Africa to test the safety and effectiveness of this approach.36 The drugs keep the virus from reproducing and are already used to prevent infection in health care workers accidentally exposed to HIV.253 There is some concern that this kind of use could lead to drug resistance, while others encourage chemoprophylaxis research.97,98
Healthy People 2010 has proposed updates to three primary goals related to reducing the number of AIDS cases: (1) reduce the number of new cases of HIV/AIDS among adolescents and adults to one new case per 100,000 people (from the 19.5 cases reported in 1998 in people aged 13 years and older), (2) 25% improvement in the number of new AIDS cases among adolescent and adult MSM, and (3) 25% improvement in the number of new AIDS cases among male and female IDUs.106
A new generation of MSM has replaced those who benefited from early prevention strategies. Behavior intervention, the primary prevention tool, includes school-based programs, peer-to-peer interventions, strategies that limit needle sharing, parent-to-child communication, client-centered counseling, and personalized risk-reduction strategies (Box 7-3).
The introduction of routine counseling, voluntary testing for women with HIV, and providing zidovudine (commonly known as AZT) to infected women and their infants have dramatically reduced mother-to-child transmission rates.175,254
Research efforts on behalf of women infected with HIV via heterosexual contact are investigating genetically engineered bacteria designed to destroy HIV in the vagina. Naturally occurring bacteria present in the vagina are modified to secrete a protein that will attract HIV. Once trapped on the surface, the HIV is destroyed by other natural substances in the vagina that are toxic to the virus.44
The search is on to find an HIV-1 microbicide that can neutralize a diversity of HIV-1 strains without causing mucosal inflammation in the lining of the vagina and have a minimum of adverse side effects, all while main- taining effectiveness for prolonged periods after a single application.201
Mycobacterium tuberculosis associated with AIDS is communicable, preventable, and treatable. One in three adults with HIV/AIDS is coinfected with M. tuberculosis. Tuberculin (TB) skin testing should be available and routinely offered to individuals at HIV testing sites. Highest risk individuals for concomitant HIV and TB infections include the homeless, IDUs, and incarcerated people (see Chapter 15).82 Guidelines for the prevention of other opportunistic infections in people with HIV are also now available.39,198
Socioeconomic factors (e.g., high rates of poverty and unemployment, lack of access to health care) are associated with high rates of HIV risk behaviors among minority MSM and are barriers to accessing HIV testing, diagnosis, and treatment. Reaching minority MSM who may not identify themselves as homosexual or bisexual with prevention messages remains a challenge.
The development of an HIV vaccine is important to control the global epidemic. However, efforts to develop postexposure prophylaxis (PEP), microbicides that are safe and effective in reducing HIV transmission through sexual intercourse, and vaccines are collectively unlikely to provide full protection against HIV and offer little or no protection against other STDs such as gonorrhea and chlamydia.254
Effective behavior change programs will still be needed to address possible behavioral disinhibition (i.e., continuing or returning to high-risk behaviors when one feels protected) among persons who receive these interventions. Prevention counseling that addresses informed choice and consent; the HIV-prevention behaviors of abstinence and delay of sexual debut, being monogamous, having fewer sex partners, and using condoms correctly and consistently; and other reproductive health needs (e.g., STD treatment and family planning) must be incorporated alongside new prevention interventions.254
DIAGNOSIS.
Early diagnosis is important so that early and preventive therapies may be initiated and sex partners can be notified of their risk of HIV and the subsequent need for HIV testing. To establish uniformity in reporting AIDS cases, the CDC has established diagnostic criteria for the confirmation of AIDS.
The most commonly performed screening test is the HIV-1 antibody enzyme immunoassay test, an antibody test to indicate HIV infection indirectly by revealing HIV antibodies (indicating exposure to the virus). However, antibody testing is not always reliable because the body takes a variable amount of time to produce a detectable level of antibodies. Consequently, a person with HIV could test negative for HIV antibodies. Antibody tests are also unreliable in neonates because transferred maternal antibodies persist for 6 to 10 months. The Western blot test is a more expensive test that may be used when there is a concern about false-positive results.
The CDC AIDS surveillance definition requires direct testing, including antigen tests (p24 antigen), HIV cultures, nucleic acid probes of peripheral blood lymphocytes, and the polymerase chain reaction.
Additional laboratory confirmation of HIV infection is required; HIV RNA viral load testing as measured by the plasma HIV RNA assay and a CD4+ T-lymphocyte count of less than 200 cells/mm3 is also considered diagnostic. T-cell measurement and viral load are both important indicators of HIV status.
To use a train crash analogy, T-cell values represent the distance to the crash, whereas viral load counts represent the speed of the train, that is, how fast the person is declining.222 T-lymphocyte counts monitor the progression of HIV and should be viewed from a broad perspective rather than on a day-to-day basis; the overall trend is more important than single values.
A rapid, noninvasive test that uses saliva or a gum swab to detect HIV antibodies is available. The OraQuick Rapid HIV-1 Antibody test can be completed in as little as 10 minutes and has up to 99.6% accuracy. The advantage is in obtaining results quickly. The CDC estimates that one third of all individuals tested do not return for their test results. Rapid testing means that individuals who are HIV positive can receive immediate counseling, early treatment to preserve their health, and prompt education on preventing disease transmission.150
The disadvantage is that the test cannot detect HIV infection in people who were exposed less than 3 months before taking the test; antibody development can take up to 3 months. Positive results should be followed up with additional test to confirm the results. Concern exists that rapid tests will distract from risk reduction. Although some individuals may be reassured that they are negative for HIV, this does not necessarily translate into reduced risk without continued education.
TREATMENT.
No cure has been found for AIDS, but advances in treatment have successfully transformed AIDS into a manageable chronic condition. The national HIV Vaccine Trials Network has been established to develop and test possible vaccine compounds, but an effective vaccine is not imminent. Until a vaccine is available, the goals of intervention are to stop HIV from replicating, increase the number of CD4 cells, and delay HIV disease progression.
Medical management centers on the CD4 cell count and viral load. When CD4 cell counts drop below 500 cells/mm3, HAART is initiated. HAART, a protease inhibitor plus two nucleoside-analog reverse transcriptase inhibitors, acts on reverse transcriptase, the enzyme necessary for transcribing HIV RNA to DNA, to achieve sustained suppression of HIV replication, reducing the amount of virus in the blood to very low and even undetectable levels. These drugs do not completely eliminate the virus, and lifelong therapy is required until complete and permanent eradication of the virus is developed.
Current efforts are focused on (1) simplifying the drug regimens to improve adherence (once-daily dosing), (2) developing alternatives for those in whom the current medications have failed, (3) preventing viral rebound (return of high levels of the virus when drugs are discontinued), and (4) managing the wide range of pharmacologic side effects.
A new generation of antiviral drugs emerging will inhibit HIV at several different points in the entry, fusion, and replication cycles of the HIV-1 virus. New classes of HAART include entry inhibitors, attachment inhibitors, fusion inhibitors, and maturation inhibitors, to name a few.103 For example, entry inhibitors operate early in the viral life cycle by preventing HIV-1 entry into susceptible cells. Fusion inhibitors are effective during the narrow window between virus attachment and the subsequent fusion of viral envelope with the cell membrane.148
Development of drug resistance is the inevitable consequence of incomplete suppression of virus plasma levels in individuals with HIV treated with HAART. Noncompliance with the drug regimen, which involves taking multiple drugs throughout the day, can lead to mutation of the virus and resistance to the treatment. Drug resistance is one of the most significant threats to effective therapy.
Several assays are available for testing HIV for resistance to antiretroviral agents. Genotypic and phenotypic testing has been implemented and should be carried out when the individual is not responding to drug therapy, as indicated by an increase in the viral load and a decrease in the CD4+ cell count, especially before the HAART regimen is discontinued. Genotyping detects viral mutation, whereas phenotyping detects a decrease in the sensitivity of the virus to the drugs.120
Potential toxicities from drug treatment include metabolic disorders (e.g., LDS), avascular necrosis of the femoral head(s), and lactic acidosis. There is no known treatment to stop treatment-induced osteonecrosis.
Palliative treatment may include nonsteroidal antiinflammatory drugs (NSAIDs) and pain medications. Surgical procedures to improve blood flow to the affected area or joint replacement to improve function may be considered. When present, dyslipidemia may be associated with accelerated atherosclerosis and insulin resistance, contributing to increased cardiovascular morbidity and mortality rates in the population with HIV.88,140
The lactic acidosis occurs as a result of injured cell mitochondria giving off lactic acid in response to the effects of nucleoside reverse transcriptase inhibitors. AZT myopathy, a toxic mitochondrial myopathy, is much less often seen today and is limited to people taking high doses of AZT. Both of these conditions are reversible with cessation of the drug therapy.228
The optimal time at which to initiate intervention remains controversial. When protease inhibitors were first introduced, the strategy was to “hit early, hit hard” because early intervention and the associated reduction of acute viral load might have positive effects on the subsequent course of the disease.259 However, the development of resistance and the toxic side effects that occur have brought that philosophy into question.
The complete eradication of the infection remains the focus of research today. Some people with HIV are coinfected with HCV, making treatment more complex because of the hepatotoxicity of antiretroviral medications and the important role the liver has in maintaining health.
The proper treatment of people with both HIV and HCV is still under investigation. The order in which antiretroviral drugs are prescribed is called sequencing and is designed to maximize viral suppression for as long as possible and to minimize the development of resistance and hepatotoxicity. Current guidelines advise a drug regimen that saves some categories of medications for future use if and when the person develops resistance to other medications.90
Structured treatment interruptions to improve viral control by stimulating HIV-specific T-lymphocyte immunity in primary HIV infection are also under investigation. If viral suppression is achieved, treatment can be interrupted because of adverse effects, cancer, or tuberculosis or in response to individual preference. It remains to be decided how many doses may be skipped before HIV treatment response is adversely affected.12,237
In all treatment situations, dosage adjustments are important considerations. Gender and ethnicity can affect the way a person metabolizes antiretroviral drugs and which adverse reactions occur. Women are especially prone to gender-based response to antiretroviral side effects. Several antiretrovirals are now known to interact with oral contraceptives (e.g., nevirapine [Viramune], ritonavir [Navir], nelfinavir [Viracept]). With HAART treatment, women are more likely to develop LDS and experience nausea, vomiting, fatigue, itching, abdominal pain, skin rashes, and perioral numbness, while men experience more diarrhea.107
If, as suspected, multiple variants of HIV are in a single host and the virus is capable of mutating easily, treatment approaches geared toward enhancing host resistance and restoring or building the immune system will be more successful than trying to kill the virus.
This same principle of using drugs to boost the immune system, called biotherapy (immunotherapy), immunomodulating, or immunologic therapy, is also being applied in the use of genetically engineered IL-2 to treat cancer. Previous trials using IFN-α resulted in significant toxicity and its use has been limited. Other researchers are investigating the use of gene therapy and immune reconstitution or restoration in relation to AIDS.
Other pharmacologic intervention may be used to treat various clinical manifestations of HIV disease (e.g., antibiotics, NSAIDs; corticosteroids; immunosuppressives; IFN therapy).
Nonpharmacologic intervention includes nutritional therapy, exercise, mental health support, and alternative or complementary interventions. AIDS-associated weight loss, nutritional deficiencies, loss of muscle mass (wasting syndrome), and other effects contribute to immune dysfunction, faster disease progression in some people, and a variety of complications that can be ameliorated with proper nutrition. The use of alternative and complementary intervention techniques remains an area of controversy and investigation.
PROGNOSIS.
AIDS has changed from an acute to subacute chronic illness. The development of combination therapies is extending the lives of AIDS clients, keeping many healthy enough to avoid hospitalization and/or return to their baseline after illness, extending survival, and even resulting in return to work status for some people. People with AIDS are living longer with lower CD4 levels because prophylaxis; improved supportive care; and treatment of PCP, TB, and ARL saves many who would have died sooner in the earlier days of the AIDS epidemic.
Changes in treatment rather than changes in human behavior accounts for the decline in AIDS-related deaths.194 Drug combinations with two or more antiretroviral drugs (HAART) have reduced mortality rates from AIDS by more than 80%. Because of HAART, 9 out of 10 people with AIDS can expect to live at least 10 years after infection. In the pre-HAART era adults older than 45 years at the time of diagnosis had a much poorer prognosis. Today older adults have the same life expectancy as younger people with AIDS.100
HAART is improving survival but at the price of a variety of metabolic (and other) side effects. Patterns of morbidity and mortality are changing; the leading cause of death is now kidney or liver failure (instead of opportunistic infections) as a result of medical treatment (e.g., protease inhibitors are metabolized by the liver) and HCV.29
Prognosis remains poor for anyone who has both HIV and HCV HIV (especially IDUs) or who is manifesting the wasting syndrome. IDUs have a higher burden of comorbidities of HIV disease, possibly contributing to poor response to HAART. Active drug use is most commonly associated with nonadherence to the required antiretroviral treatment regimen and prophylaxis for opportunistic infections.174
Women are less likely than men to receive prescriptions for the most effective treatments for HIV infection, potentially affecting both morbidity and mortality rates.34
On the other hand, survival has been reportedly prolonged in HIV-positive people who are coinfected with the GBV-C virus. GBV-C does not prevent infection by HIV, but it slows the replication process, thereby delaying the progression to AIDS. The GBV-C virus also increases production of various chemokines, chemicals produced by WBCs to fight infection. Further research may yield new strategies to control HIV-1 replication and progression to AIDS.89,241
Clinical presentation in the pediatric population is often more devastating due to the immaturity of the immune system. Children become sicker faster, with death within two years in many cases. The immature CNS is very vulnerable to direct infection by HIV. Developmental delays are often the first sign of HIV and contribute to morbidity in this group.
A considerable body of evidence suggests that psychosocial factors play an important role in the progression of HIV infection, its morbidity, and its mortality. Psychosocial influences relating to faster disease progression include life-event stress, sustained depression, denial or avoidance coping, concealment of gay identity, and negative outlook. Conversely, protective psychosocial factors include active coping, collaborative relationship with health care professionals, and stress management.
Biologic factors such as genetics, age of the host, viral strain and virulence, medication, and immune response are also major determinants of disease progression and also have an impact on outcome. Scientists are continuing to investigate psychoneuroimmunologic pathways by which immune and neuroendocrine mechanisms might link psychosocial factors with health and survival.
Chronic Fatigue and Immune Dysfunction Syndrome
Chronic fatigue and immune dysfunction syndrome, chronic fatigue syndrome (CFS), chronic Epstein-Barr virus, myalgic encephalomyelitis, neuromyasthenia, and the “yuppie flu” all denote a highly publicized but not new illness.
The name chronic fatigue syndrome indicates that this illness is not a single disease but the result of a combination of factors and is actually a subset of chronic fatigue, a broader category defined as unexplained fatigue of greater than or equal to 6 months’ duration. This distinction is made to facilitate epidemiologic studies of populations with prolonged fatigue and chronic fatigue.
Incidence and Risk Factors
Two U.S. community-based CFS studies found prevalences among adults of 0.23% and 0.42%; the rates were higher in women, members of minority groups, and people with lower educational attainment and occupational status.119,210
People of every age, gender, ethnicity, and socioeconomic group can have CFS. Demographic data show that in most studies 75% or more of people with CFS are female. The mean age at onset of CFS is between 29 years and 35 years. The mean illness duration ranges from 3 years to 9 years.28 Although CFS is much less common in children than in adults, children can develop the illness, particularly during the teen years.
Etiologic Factors and Pathogenesis
Many studies have investigated the etiology and pathogenesis of CFS.200 More than half of the CFS studies between 1980 and 1995 concentrated on the physical etiology of CFS, with a slight shift toward psychologic and psychiatric research in the next few years.52
Many somatic and psychosocial hypotheses on the etiology of CFS have been explored. Explanations for CFS were sought in viral infections, immune dysfunction, neuroendocrine responses, dysfunction of the CNS, muscle structure, exercise capacity, sleep patterns, genetic constitution, personality, and (neuro) psychological processes.
Although several studies found abnormalities, only a few were diagnosed in large groups of people with CFS and were independently confirmed in well-controlled studies, an exception being the subtle changes in the hypothalamopituitary-adrenal axis.
Neuroendocrine challenge tests have found a lower-than-normal cortisol response to increased corticotrophin concentrations and upregulation of the serotonergic system. Studies of neuroendocrine function in individuals with CFS have found no evidence for uniform dysfunction of the hypothalamopituitary-adrenal axis or stress hormones.53 Increasing evidence points to acquired neuroendocrine dysregulations in people with CFS.81
However, abnormalities of the neuroendocrine system and CNS alone are not sufficient to explain the symptoms of CFS. More complex interactions between regulating systems are assumed to be at work and seem to involve the CNS, the immune system, and the hormonal regulation system. The etiology and pathogenesis are generally believed to be multifactorial.45,54
Personality and lifestyle are presumed to influence vulnerability to CFS. Personality characteristics of neuroticism and introversion have been reported as risk factors for the disorder.200
Inactivity in childhood and inactivity after infectious mononucleosis have been found to increase the risk of CFS in adults.257,264 Also, acute physical or psychologic stress might trigger the onset of CFS.
Three quarters of the individuals with this disorder have reported an infection, such as a cold, flulike illness, or infectious mononucleosis, as the trigger,59,221 and high rates of chronic fatigue after Q fever and Lyme disease have been found.153 Finally, serious life events, such as the loss of a loved one or a job, and other stressful situations have been found to precipitate the disorder.104,252
Clinical Manifestations
At illness onset, the most commonly reported CFS symptoms are sore throat, fever, muscle pain, and muscle weakness. As the illness progresses, muscle pain and forgetfulness increase along with prolonged (lasting more than 6 months), often overwhelming fatigue that is exacerbated by minimal physical activity.
Neurally mediated hypotension (NMH), caused by disturbances in the autonomic regulation of blood pressure and pulse, is common in people with CFS. This condition is characterized by lowered blood pressure and heart rate accompanied by lightheadedness, visual dimming, or slow response to verbal stimuli. Many people with NMH experience lightheadedness or worsening fatigue as they stand for prolonged periods or when in warm places (e.g., hot shower, sauna, indoor pool environment).
The severity of CFS varies from person to person, with some people able to maintain fairly active lives. By definiton, however, CFS significantly limits work, school, and family activities.
While symptoms vary from person to person in number, type, and severity, all individuals with CFS are functionally impaired to some degree. CDC studies show that CFS can be as disabling as multiple sclerosis (MS), lupus, rheumatoid arthritis, heart disease, end-stage renal disease, chronic obstructive pulmonary disease, and similar chronic conditions.
CFS often follows a cyclical course, alternating between periods of illness and relative well-being. Some people experience partial or complete remission of symptoms during the course of the illness, but symptoms often reoccur.
This pattern of remission and relapse makes CFS especially hard for clients and their health care professionals to manage. People who are in remission may be tempted to overdo activities when they are feeling better, which can exacerbate symptoms and fatigue and cause a relapse. In fact, postexertional malaise is a hallmark of the illness.38
MEDICAL MANAGEMENT
There are no physical signs or diagnostic laboratory tests that help identify CFS. People who suffer the symptoms of CFS must be carefully evaluated by a physician because many treatable medical and psychiatric conditions are hard to distinguish from CFS.
Common conditions that should be ruled out through a careful medical history and appropriate testing include mononucleosis, Lyme disease, thyroid conditions, diabetes, MS, various cancers, depression, and bipolar disorder.
To aid the identification of this disease, the CDC has developed a working case definition with two criteria to distinguish CFS from other forms of fatigue (Box 7-6). The symptoms must have persisted or recurred during 6 or more consecutive months of illness and must not have predated the fatigue. Research conducted by the CDC indicates that less than 20% of the people with CFS in this country have been diagnosed.38
TREATMENT.
Since there is no known cure for CFS, treatment is aimed at symptom relief and improved function. A combination of drug and nondrug therapies is usually recommended.
No single therapy exists that helps all individuals with CFS.
Lifestyle changes, including prevention of overexertion, reduced stress, dietary restrictions, gentle stretching, and nutritional supplementation, are frequently recommended in addition to drug therapies used to treat sleep disturbances, pain, and other specific symptoms.
Carefully supervised physical therapy may also be part of treatment for CFS. However, symptoms can be exacer- bated by overly ambitious physical activity. A very moderate approach to exercise and activity management is recommended to avoid overactivity and to prevent deconditioning.38 Systematic reviews have investigated the effectiveness of several CFS treatments, and cognitive behavior therapy and graded exercise therapy are the only interventions found to be beneficial.199,209,265
PROGNOSIS.
CFS affects each individual differently. Some people with CFS remain homebound and others improve to the point that they can resume work and other activities, even though they continue to experience symptoms.
The CDC’s research has shown that those who have CFS for 2 years or less were more likely to improve. It remains unknown if early intervention is responsible for this more favorable outcome; however, the longer a person is ill before diagnosis, the more complicated the course of the illness appears to be.
Recovery rates for CFS are unclear. Improvement rates varied from 8% to 63%, with a median of 40% of clients improving during follow-up. However, full recovery from CFS may be rare, with an average of only 5% to 10% sustaining total remission.38