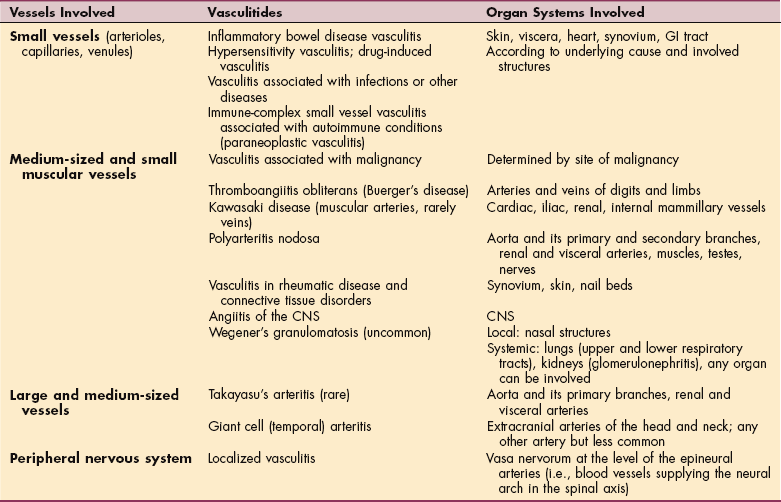
DISEASES AFFECTING THE BLOOD VESSELS
Diseases of blood vessels observed in a therapy setting can include intestinal infarction, aneurysm, PVD, vascular neoplasm, and vascular malformation; only intestinal infarction will not be discussed here.
Aneurysm
An aneurysm is an abnormal stretching (dilation) in the wall of an artery, a vein, or the heart with a diameter that is at least 50% greater than normal. When the vessel wall becomes weakened from trauma, congenital vascular disease, infection, or atherosclerosis, a permanent saclike formation develops. A false aneurysm can occur when the wall of the blood vessel is ruptured and blood escapes into surrounding tissues, forming a clot (Fig. 12-25; see also Fig. 12-27).
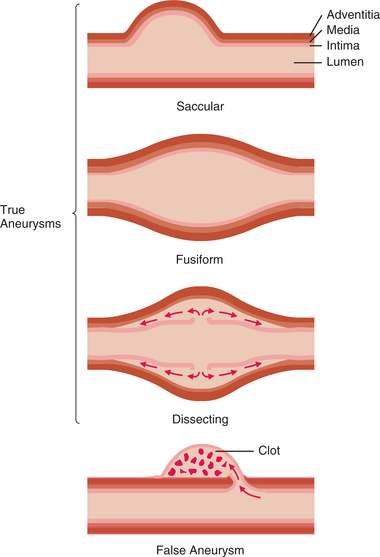
Figure 12-25 Longitudinal sections showing types of aneurysms. In a true aneurysm, layers of the vessel wall dilate in one of the following ways: saccular, a unilateral outpouching; fusiform, a diffuse dilation involving the entire circumference of the artery wall; or dissecting, a bilateral outpouching in which layers of the vessel wall separate, with creation of a cavity. In a false aneurysm, the wall ruptures, and a blood clot is retained in an outpouching of tissue.
Aneurysms are of various types (either arterial or venous) and are named according to the specific site of formation (Fig. 12-26). The most common site for an arterial aneurysm is the aorta, forming a thoracic aneurysm (which involves the ascending, transverse, or first part of the descending portion of the aorta) or an abdominal aneurysm (which generally involves the aorta between the renal arteries and iliac branches).
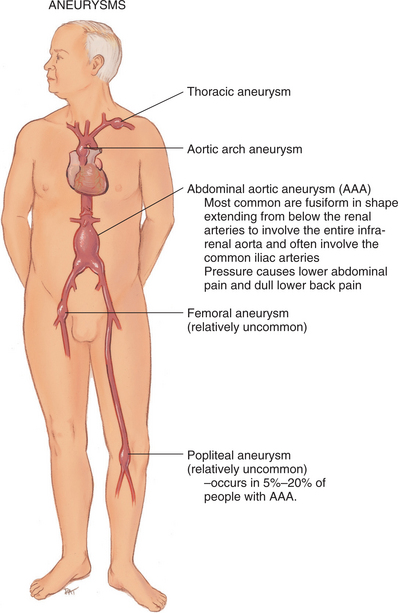
Figure 12-26 Aneurysms are named according to the specific site of formation. Abdominal aortic aneurysms are the most common type; more than 95% of abdominal aortic aneurysms are located below the renal arteries and extend to the umbilicus, causing low back pain. (From Jarvis C: Physical examination and health assessment, ed 5, Philadelphia, 2008, Saunders.)
Thoracic aortic aneurysms located above the diaphragm account for approximately 10% of all aortic aneurysms and occur most frequently in hypertensive men between the ages of 40 and 70 years. Men are more likely to have thoracic or abdominal aneurysms. Thoracic aortic aneurysms occur less often than other types but tend to be more life-threatening.
Abdominal aortic aneurysms located below the diaphragmatic border occur about four times more often than thoracic aneurysms, most likely because the aorta is not supported by skeletal muscle at this location. The incidence of abdominal aortic aneurysm is increasing, probably because of the increasing number of adults over 65 years of age.
Peripheral arterial aneurysms affect the femoral and popliteal arteries.
Incidence and Etiologic Factors
According to the Society for Vascular Surgery, approximately 200,000 people in the United States are diagnosed annually with aortic aneurysm and 15,000 of those aneu- rysms are severe enough to rupture, causing a medical emergency.312
Incidence increases with increasing age, usually beginning after age 50 years, presumably as a result of chronic inflammatory cellular changes resulting in atherosclerosis. However, someone without evidence of atherosclerosis can develop an aneurysm, especially in the presence of congenital weakness of the blood vessel walls.
Family members (parent, adult child, or sibling) of anyone with an aneurysm have a fourfold increased risk of aneurysm, and gene defects on chromosomes 11340 and 15172 have been identified with some of the connective tissue disorders associated with aneurysm. Recently, a mutation in a specific protein, transforming growth factor β receptor (TGFBR), has been identified as responsible for causing aneurysms.200 Aneurysms occur much more often in men than in women, and one half of affected persons are hypertensive.
Atherosclerosis or any injury to the middle or muscular layer of the arterial wall (tunica media) is responsible for most arterial aneurysms. Other less common causes of aneurysm include trauma (blunt or surgical), Marfan’s disease (congenital defects of the arterial wall) and other hereditary abnormalities of connective tissue, and inflammatory diseases and infectious agents (bacterial infection, syphilis, polyarteritis).
The emergence of HIV has been associated with a dramatic increase in the incidence of syphilis. Since syphilitic aortitis generally presents between 10 and 30 years after the primary infection, there may be an increased incidence of associated aneurysms in the coming years. Hypertension seems to enhance aneurysm formation.
Pathogenesis
Plaque formation erodes the vessel wall, predisposing the vessel to stretching of the inner and outer layers of the artery and formation of a sac. The stretching of the media produces infarct expansion, a weak and thin layer of necrotic muscle, and fibrous tissue that bulges with each systole. Abnormal proteolysis, the presence of elastolytic serum enzymes, and deficiencies of collagen and elastin have been implicated as factors contributing to the development of these aneurysms.184
With time, the aneurysm becomes more fibrotic, but it continues to bulge with each systole, thus acting as a reservoir for some of the stroke volume. In the case of thoracic aortic aneurysms, the shear force of elevated blood pressure causes a tear in the intima with rapid disruption and rupture of the aortic wall. Subsequent hemorrhage causes a lengthwise splitting of the arterial wall, creating a false vessel (Fig. 12-27), and a hematoma may form in either channel (i.e., the false or true lumen).
Clinical Manifestations
Aneurysms may be asymptomatic; when they do occur, manifestations depend largely on the size and position of the aneurysm and its rate of growth. Persistent but vague substernal, back, neck, or jaw pain may occur as enlargement of the aneurysm impinges adjacent structures.
Dissection over the aortic arch and into the descending aorta may be experienced as extreme, sharp pain felt at the base of the neck or along the back into the interscapular areas. When pressure from a large volume of blood is placed on the trachea, esophagus, laryngeal nerve, lung, or superior vena cava, symptoms of dysphagia; hoarseness; edema of the neck, arms, or jaw and distended neck veins; and dyspnea and/or cough may occur, respectively.
Other signs and symptoms may be present in the case of acute aortic dissection as a result of compression of branches of the aorta. These include acute MI, reversible ischemic neurologic deficits, stroke, paraplegia, renal failure, intestinal ischemia, and ischemia of the arms and legs. Acute chest pain may also result from a nondissecting hematoma of the aorta or erosion of a penetrating atherosclerotic ulcer.184
In the case of an untreated abdominal aortic aneurysm expansion and rupture can occur in one of several places, including the peritoneal cavity, the mesentery, the retroperitoneum, into the inferior vena cava, or into the duodenum or rectum. Rupture refers to a tearing of all three tunicae (tunica adventitia, tunica media, tunica intima) with bleeding into the thoracic or abdominal cavity. The most common site for an abdominal aortic aneurysm is just below the renal arteries, and it may involve the bifurcation of the aorta (see Fig. 12-26).
Most abdominal aortic aneurysms are asymptomatic, but intermittent or constant pain in the form of mild to severe mid-abdominal or lower back discomfort is present in some form in 25% to 30% of cases. Groin or flank pain may be experienced because of increasing pressure on other structures.
Early warning signs of an impending rupture may include abdominal heartbeat when lying down or a dull ache (intermittent or constant) in the mid-abdominal left flank or lower back. Rupture is most likely to occur in aneurysms that are 5 cm or larger, causing intense flank pain with referred pain to the back at the level of the rupture. Pain may radiate to the lower abdomen, groin, or genitalia. Back pain may be the only presenting symptom before rupture occurs.
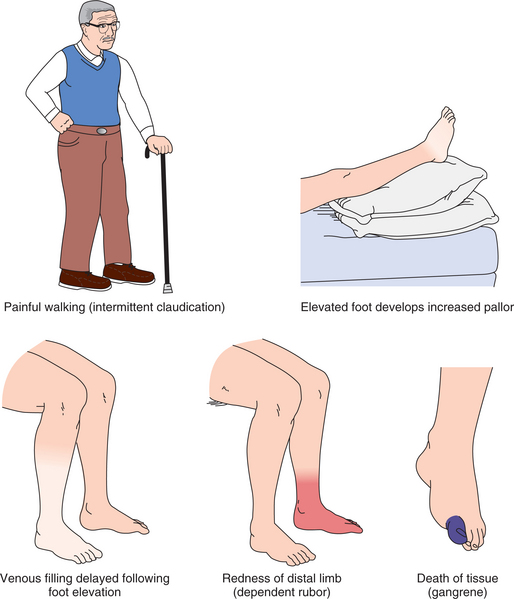
The most common site for peripheral arterial aneurysm is the popliteal space in the lower extremities. Most are caused by atherosclerosis and occur bilaterally in men. Popliteal aneurysm presents as a pulsating mass, 2 cm or more in diameter, and causes ischemic symptoms in the lower limbs (e.g., intermittent claudication, rest pain, thrombosis and embolization resulting in gangrene). Femoral aneurysm presents as a pulsating mass in the femoral area on one or both sides.