Chapter 16 Diseases associated with bacteria – I
INTRODUCTION TO INFECTIOUS DISEASE 765
DISEASES ASSOCIATED WITH STREPTOCOCCUS SPECIES 768
DISEASES ASSOCIATED WITH STAPHYLOCOCCUS SPECIES 783
DISEASES ASSOCIATED WITH CORYNEBACTERIUM, ACTINOBACULUM, AND ARCANOBACTERIUM SPECIES 787
DISEASES ASSOCIATED WITH LISTERIA SPECIES 805
DISEASES ASSOCIATED WITH ERYSIPELOTHRIX RHUSIOPATHIAE (INSIDIOSA) 810
DISEASES ASSOCIATED WITH BACILLUS SPECIES 815
Introduction to infectious disease
The infectious diseases are of major importance in agricultural animals. Accordingly the bacterial, viral, fungal, protozoal and parasitic diseases account for a major portion of this book. The infectious diseases are capable of affecting many animals in a short period of time and the case fatality rate in some diseases can be very high and the economic losses may be very large. Certain infectious diseases, especially the viral diseases, are endemic in some countries and pose a threat to other countries considered to be free of them. The veterinary profession has made a major contribution in developing reliable diagnostic techniques and effective control procedures for many of these diseases. Some of the infectious diseases assume major importance because they are directly transmissible to humans.
REPORTABLE DISEASES (OIE LISTINGS)
Several infectious diseases of livestock have the potential for serious and rapid spread, implications for international trade and/or risk for significant zoonotic disease. Because of the nature of these diseases and the risk to regional or national animal health, their control is beyond the capability of the individual practitioner and requires the intervention of state and possibly national veterinary personnel and organizations. In most countries, diseases of this nature are listed as reportable diseases or notifiable diseases and these must be reported (notified) to the state or national veterinary authority immediately they are diagnosed.
A world organization for animal health, called the Office International des Épizooties (OIE), has been in existence for many years and its major objectives have been the collection, analysis, and dissemination of scientific veterinary information on animal diseases of significant veterinary importance. Member countries have traditionally reported to the OIE, on a regular basis, the occurrence, or absence, of these diseases in their respective countries. The purpose has been to ensure transparency in the global animal disease and zoonotic disease situation, and to provide expertise and encourage international solidarity in the control of animal diseases.
The OIE has established a Terrestrial Animal Health Code (Terrestrial Code) where the majority of the reportable diseases are specifically detailed, including their clinical presentations, distribution amongst member countries and methods for diagnosis.1 The purpose of the Terrestrial Code is to assure the safety of international trade in terrestrial animals and their products by detailing the health measures to be used by the veterinary authorities of importing and exporting countries. The purpose is to avoid the transfer of agents pathogenic for animals or humans while also avoiding unjustified sanitary banners. It constitutes a reference within the World Trade Organization (WTO) Agreement on the Application of Sanitary and Phytosanitary Measures (SPS Agreement) as an international standard for animal health and zoonoses.2,3
Traditionally, the OIE has classified animal diseases in two lists.
• List A diseases (Box 16.1). These are transmissible diseases that have the potential for very serious and rapid spread, irrespective of national borders, that are of serious socioeconomic or public health consequence and that are of major importance in the international trade of animals and animal products. The presence of a list A disease in a country may limit or prevent its international trade of animals and animal products. In most countries the control and regulation of list A diseases are under the control of the federal veterinary authorities. Private practitioners have the responsibility to immediately report the occurrence, or suspicion of their occurrence, to state or federal regulatory authorities
• List B diseases (Box 16.2). These are transmissible diseases that are considered to be of socioeconomic and/or public health importance within countries and that are significant in the international trade of animals and animal products. The control of these diseases varies between countries and between diseases in this list, but most countries require that they be reported to state or federal authorities if they are diagnosed.
Box 16.1 List A diseases for cattle, swine, horses, sheep and goats
Transmissible diseases that have the potential for very serious and rapid spread, irrespective of national borders, that are of serious socioeconomic or public health consequence and that are of major importance in the international trade of animals and animal products
Box 16.2 List B diseases for cattle, swine, horses, sheep and goats
Transmissible diseases that are considered to be of socioeconomic and/or public health importance within countries and that are significant in the international trade of animals and animal products
Cattle
2005 single list
In 2005 the OIE reorganized the diseases in the Terrestrial Code to a single list (Box 16.3). In part, this was done in response to concerns that the administration and listings of diseases had led to unfair trade restrictions on certain countries. The new listing is accompanied by the expectation of better international cooperation in the detection and control of animal disease, and support for developing countries in the diagnosis and control of these diseases is proposed. There is also the recognition that many of the recent emerging, or reemerging, diseases of animal origin have zoonotic potential. The overriding criterion for inclusion of a disease in the OIE single list remains its potential for international spread, but other criteria include a capacity for significant spread within naïve populations and zoonotic potential.1-3 There has been agreement that the occurrence of certain listed diseases in a country does not necessarily preclude trade by that country in animal products. One of the reasons is that the disease may be absent from certain regions of the country that have geographical or ecological characteristics that would not support the disease – an example would be areas of a country that did or did not support the vectors for bluetongue. It is also recognized that management systems may allow freedom from disease and that consequently there could be trade from production systems using these management methods – an example would be the disease and security status in a high health swine unit compared to free-range pigs in the same area.2,3 There is also the recognition that many of the recent emerging, or re-emerging diseases of animal origin have zoonotic potential.
Box 16.3 2005 single list of diseases notifiable to the Office International des Épizooties
Affecting more than one animal species
Cattle
We have listed the old List A and List B categorizations of diseases as these categories are familiar to practicing veterinarians and we suspect that they will be referred to, as such, in the lifetime of this edition of this book. We further believe that there was some meaning for practicing veterinarians, in terms of disease significance and emergency response, in the old List A and List B categorization that is not evident in the new single listing, which, although it might be politically correct, has less impact for those who practice clinical veterinary medicine at the level of the farm. We have reservations concerning the new single listing and reservations about some of the diseases selected for listing. There are several examples, but three will suffice. We question how a disease such as leptospirosis, generically essentially a universal infection in farm animals, can be equated and placed in a single list with a disease such as foot and mouth disease. We question how African horse sickness, with its huge potential for spread outside Africa, can be compared with the ubiquitous, and usually clinically insignificant, equine rhinopneumonitis. We question the inclusion of a disease such as atrophic rhinitis in the swine list of diseases and the exclusion of a disease such as pleuropneumonia, but more importantly, we question why atrophic rhinitis should be equated in economic, or response, importance to African swine fever.
DIAGNOSIS OF INFECTIOUS DISEASES
The clinical and laboratory diagnosis of the infectious diseases can be difficult. However, with the appropriate laboratory support and suitable samples, most of them can be diagnosed definitively. For each disease certain samples must be submitted to the laboratory for isolation or demonstration of the specific pathogen.
Clinical and epidemiological findings will usually result in a tentative diagnosis and a rule-out list of possible diagnoses. When herd or area population epidemics occur, a detailed examination of the epidemiological characteristics of the disease is often useful in helping to make a diagnosis and in advising on the best treatment and control procedures. Particular attention should be given to the epidemiological aspects of the history, for example:
• The descriptive epidemiology including the distribution in age or other groups
• The morbidity and case fatality and population mortality rates
• Relationship to other species
• Recent changes in management
The epidemiological behavior of the epidemic includes an examination of the manner of spread of disease between individuals and groups of animals, the age of animals affected, the length of the course of the disease and the estimated incubation period. It may be necessary to establish a prospective survey or surveillance studies on sentinel herds to monitor the spread of the disease. field investigation may include the examination of nearby herds or other species, such as wildlife or humans, that may be sources of the infection.
When an infectious disease is suspected, laboratory investigations will aid in diagnosis and differential diagnosis. The veterinary literature contains a large number of reports of the development of antibody-detection or antigen-detection tests for individual animal diseases. Most of these have not been validated in a significant number of animals for sensitivity and specificity in disease detection, and most are not available commercially or available on line in diagnostic laboratories. Those that are on line vary between countries and between laboratories within countries. The practitioner should consult the appropriate diagnostic laboratories to determine the tests that are available and the laboratories’ definition of the sensitivity and specificity of the test to be used. The recommended techniques of investigation and the common differential diagnoses are given for each disease.
• Clinical, hematological, clinical chemistry and immunological examinations, as appropriate, should be done on as many clinically affected animals as possible. Similar examinations should be conducted on normal animals that have been in close contact with the affected animals. The detection of latent carriers among clinically normal animals may require special laboratory tests. Repeated visits and examinations may be necessary to determine the presence and rate of seroconversion in an affected herd as an indication of the rate and direction of spread
• Tissue samples from necropsies and from live animals (biopsies) and discharges, feces and urine should be submitted for isolation or demonstration of the suspected pathogen.
The responsibility of the veterinarian in the case of infectious diseases is to advise the owner of the risks of spread to other animals or populations and the need for treatment and/or control. In the case of notifiable diseases, the government regulatory authorities must be notified immediately. Every precaution must be taken to prevent the spread of the disease to nearby herds or other geographical areas. Veterinarians have the responsibility to advise the owner of zoonotic risk of the disease to the farm family and farm workers. Veterinarians also have a responsibility to alert their clients to the possibility that an infectious disease may be approaching geographically and to take the necessary precautions. The control of infectious diseases is dependent on a knowledge of the etiology and epidemiological characteristics of the disease. The basic principles of disease control are:
• Reduce the infection pressure
• Eliminate or minimize risk factors that can be influenced
Any one or a combination of the following may be effective for the control of a specific infectious disease:
• Insure adequate colostral immunity in the newborn
• Identify affected animals, isolate them from the normal animals and treat or dispose of them as indicated, or return them to the herd if they are considered to be safe
• Prevent the introduction of infected animals into herds previously considered free of the disease. Quarantine all animals imported into a herd for a period of 30–60 days. Serological testing may be done on the imported animals
• Determine the source of the infection and remove it if possible. Sources include infected animals, feed and water supplies, wildlife and contaminated environments
• Control by the use of mass medication of feed and water supplies may be appropriate with some diseases
• Clean and disinfect animal houses and grounds regularly – this is essential. When animals occupy a barn for prolonged periods (weeks or months) without a clean-out and disinfection, the buildup of infectious agents increases almost geometrically and the incidence of disease will increase correspondingly
• Provide an optimal environment for housed animals. This includes adequate ventilation, the prevention of overcrowding and effective removal of manure
• Establish primary breeding stock through the use of specific-pathogen-free animals obtained by hysterectomy or cesarean section and rearing them under controlled conditions; this program may be indicated for the control of diseases such as enzootic pneumonia of swine
• Vaccinate susceptible animals against endemic diseases; this should be part of a regularly scheduled herd health program that includes vaccination of the pregnant dam for the enhancement of colostral immunity in the newborn
• Avoid stress associated with long transportation, inclement weather and undernutrition
• Base effective control of intestinal parasites on measures designed to prevent or limit contact between parasite and host. The strategies are to: a) prevent the build-up of dangerous numbers of larvae on pastures; and b) anticipate the periods during which large numbers of larvae are likely to occur and remove susceptible animals from heavily contaminated pastures before these periods. These aims can be achieved using three interrelated approaches: grazing management, the use of anthelmintics and dependence on the acquisition of immunity.
1 OIE. Terrestrial Code. Available on line at: http://www.oie.int/eng/normes/en_mcode.htm, 2005. 14th ed.
Diseases associated with Streptococcus species
Mastitis associated with Streptococcus agalactiae, Streptococcus dysgalactiae, Streptococcus uberis, and Streptococcus zooepidemicus is dealt with in Chapter 15. Strangles in horses, neonatal streptococcal infections, disease associated with S. zooepidemicus in horses and streptococcal cervical abscesses of pigs are dealt with later in this section.
Other miscellaneous diseases in which streptococci appear to have etiological significance include septicemic infections of swine, sheep and calves, pneumonia in calves, meningoencephalitis and otitis media in feeder pigs, lymphangitis in foals and infectious dermatitis of piglets.
SEPTICEMIA
• Acute streptococcal septicemia of adult sows and their litters occurs sporadically. The onset is sudden and death occurs in 12–48 hours. Clinically there is weakness, prostration, fever, dyspnea, dysentery and hematuria. At necropsy, petechial and ecchymotic hemorrhages are present throughout all organs. Animals that survive for several days show extensive edema and consolidation of the lungs. The infection spreads rapidly and the mortality rate may be very high unless the drug sensitivity of the organism, usually S. zooepidemicus or Streptococcus equisimilis, is determined and appropriate treatment instituted
• Experimental inoculation of S. zooepidemicus into horses causes fever, depression, anorexia, abnormal lung sounds, joint inflammation accompanied by mild–severe lameness, emaciation and a leukocytosis1
• Streptococcus pneumoniae is the apparent cause of septicemia with sudden death in calves
• In ruminants of all ages Streptococcus suis, usually a pathogen in pigs, causes a bacteremia with localization in many organs, including lungs, joints, bones, and meninges2
• In sheep and goat flocks, septicemia due to S. zooepidemicus causes up to 90% mortality in lambs and kids.S. dysgalactiae is a common cause of polyarthritis in lambs.
ENTERITIS
Streptococcus (Enterococcus) durans has been isolated from a foal with enteritis and has caused mild enteritis and diarrhea in foals infected orally. This organism has also been isolated from outbreaks of diarrhea in neonatal pigs3 and disease has been produced in gnotobiotic piglets.4 Streptococcus entericus sp. nov. has been isolated from the intestine of a cow with enteritis but its causal significance is not known.5
PNEUMONIA
• A syndrome of pneumonia and fibrinous pleuritis and pericarditis is caused in lambs by S. zooepidemicus
• Pneumonia in calves may be associated with S. pneumoniae, and unidentified streptococci are common invaders in viral pneumonia of calves. Infections in calves with S. pneumoniae may have public health significance; the isolation of identical strains of the organism from the lungs of calves dying of the disease and from the throats of their human attendants suggests that interspecies transmission may occur. Calves may be immunized either by the use of antiserum or through vaccination of their dams with a polyvalent aluminum hydroxide-adsorbed vaccine
• S. pneumoniae is also associated with inflammatory disease of the lower respiratory tract of horses,6 sometimes in association with a virus and sometimes with S. zooepidemicus7
• Streptococcal pneumonia can be a complication of systemic corticosteroid therapy in horses.8
MENINGOENCEPHALITIS
Meningoencephalitis is a common complication of streptococcal septicemia of the newborn but S. suis is a specific cause in pigs and is dealt with under that heading. Streptococcal meningitis is restricted to the neonate in all species except piglets with S. suis and rarely lambs where sporadic meningitis in 3–5-month-old lambs occurs. Streptococcal meningitis and brain abscess may be a rare complication in horses of sinusitis and rhinitis.9
LYMPHANGITIS
An ulcerative lymphangitis, caused in many instances by S. zooepidemicus, has been observed in foals from 6 months to 2 years of age and may be confused with ulcerative lymphangitis associated with Corynebacterium pseudotuberculosis.
DERMATITIS
Infectious dermatitis (contagious pyoderma) of pigs is characterized by the formation of pustules about the face and neck, and to a lesser extent the trunk. Streptococci and staphylococci are present in the lesions and spread appears to occur through abrasions, especially in young pigs that fight and have not had their needle teeth removed. The disease may be confused with exudative epidermitis.
ARTHRITIS IN LAMBS
In the UK, S. dysgalactiae is a significant cause of outbreaks of arthritis in lambs, especially during the first 3 weeks of life. It occurs more commonly in lambs that are lambed indoors. Affected lambs are lame in one or more legs and are often found recumbent. There is minimal joint swelling in the initial stage of the disease and for this reason differential diagnoses include nutritional myopathy, swayback and spinal abscess. Infection is present in any joint but most common in the tarsal and atlanto-occipital joints. Some die with systemic disease and myocarditis. Survivors may be chronically lame.10
GENITAL TRACT INFECTIONS
Streptococcal infections of the genital tract, including S. zooepidemicus, occur commonly, especially in mares, in which the disease is thought to be spread by coitus and is accompanied by a high incidence of abortion, sterility and neonatal infection in foals.11,12
Foals from infected mares may be affected each year. Although streptococcal metritis occurs in sows, there appears to be no relationship between uterine infection and neonatal septicemia. Abortions in sows may be associated with infection with beta-hemolytic streptococci.
One of the important features of streptococcal diseases is their lack of susceptibility to control by vaccination. Vaccines against strangles of horses, S. suis in piglets and cervical adenitis of pigs are available, but results with them are equivocal. When the vaccines are properly prepared and carefully and intelligently applied the results are good. However, it is apparent that the streptococci are not good antigens and that the vaccines made with them lack the ease of application and safety that one expects with, say, the clostridial vaccines.
1 Varma KJ, et al. J Vet Pharmacol Ther. 1984;7:213.
2 Hommez J, et al. Vet Rec. 1988;123:626.
3 Choen D-S. J Vet Diagn Invest. 1996;8:123.
4 Tzipori S. J Infect Dis. 1984;150:589.
5 Vela AI, et al. Int J Syst Evol Microbiol. 2004;52:665.
6 Mackintosh ME, et al. In: Proceedings of the 5th International Conference on Infectious Diseases in Horses 1988;41–44.
7 Burrell MH, et al. Equine Vet J. 1986;18:183.
8 Mair TS. Vet Rec. 1996;138:205.
9 Smith JS, et al. J Am Vet Med Assoc. 2004;224:739.
10 Watkins G. State Vet J. 2000;10:15.
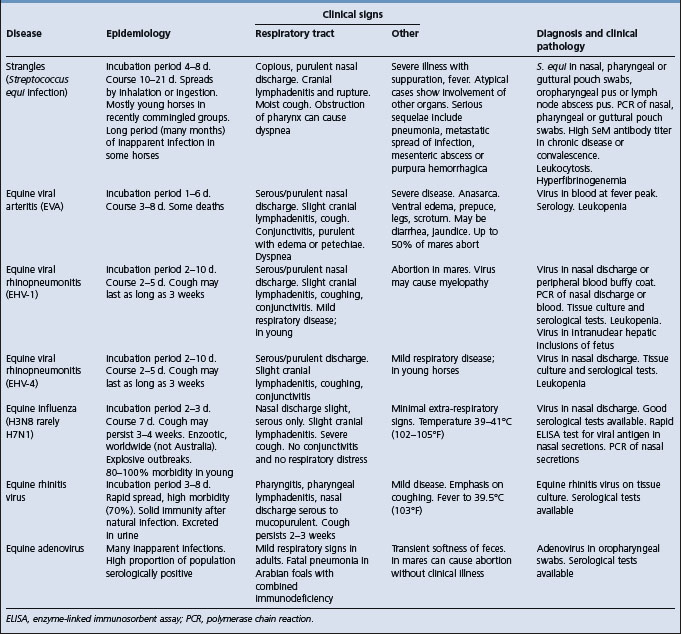
STRANGLES (EQUINE DISTEMPER)
Etiology Streptococcus equi subsp. equi
Epidemiology Highly contagious disease that affects horses of all ages but is most common in young animals. Prolonged carrier state in asymptomatic animals
Clinical signs Acute onset of fever, anorexia, depression, submandibular and pharyngeal lymphadenopathy with abscessation and rupture, and copious purulent nasal discharge. Metastatic infection in other organ systems
Clinical pathology Culture of S. equi from nasal and abscess discharges. Polymerase chain reaction (PCR) of nasal, pharyngeal or guttural pouch swabs. High serum antibody titer to SeM
Lesions Caseous lymphadenopathy with rhinitis and pharyngitis, pneumonia and metastatic infection in severe cases
Diagnostic confirmation Culture of S. equi
Treatment Systemic administration of penicillin. Local treatment of abscesses
Control Isolation and quarantine of cases and new admissions to barns and stables. Detection of carrier status by PCR and/or culture of guttural pouch washings. Vaccination may reduce the case attack rate and severity of disease
ETIOLOGY
Streptococcus equi subsp. equi (S. equi) is a Gram-positive coccobacillus that produces a beta-hemolysin, evident as a zone of clear hemolysis surrounding colonies growing on blood agar. There is evidence that S. equi is a biovar or genovar of S. zooepidemicus.1 S. equi, which is highly host-adapted to Equidae, demonstrates no serological variation, although genetic analysis demonstrates the existence of clones that vary geographically.2 There is variation in virulence related to the amount of M protein and hyaluronic capsule produced.3 An atypical milder form of the disease is associated with a capsule-deficient variant of S. equi.
EPIDEMIOLOGY
Occurrence
Strangles occurs in horses, donkeys, and mules worldwide. Outbreaks are seen relatively frequently on breeding farms and in polo and racing stables, when the infection is introduced by new arrivals that are often asymptomatic, and in horses taken to fairs and riding schools. An incidence of 35% over a 3-year period is reported for horse studs in Australia.4
Strangles can affect horses of any age, although the morbidity rate is usually greater in younger horses such as foals and weanlings.5 Age-specific attack rates of strangles of 18% for brood mares, 48% for 1-year-old horses and 38% for foals during an outbreak on a breeding farm are reported, although higher morbidity rates (100%) can occur, especially in young horses.5,6 The risk of occurrence of an outbreak of strangles increases with the size of the group of horses: farms with 100 or more horses have a 26 times greater risk of experiencing an outbreak than farms with fewer than 15 horses.4
The case-fatality rate without treatment is about 9%, but with adequate early treatment may be as low as 1–2%. Deaths are usually due to pneumonia.5,7
Source of infection and transmission
S. equi is an obligate parasite of horses and all infections are attributable to transmission from infected horses, either directly or by fomites. Nasal and abscess discharge from infected animals that contaminates pasture, tack, stalls, feed and water troughs, grooming equipment, and hands and clothes of grooms and veterinarians is often the source of infection for susceptible horses. S. equi can survive in the environment for at least 2 months, and fomite transfer is important in transmission of infection.8 Direct transmission from infected animals to susceptible animals occurs through contact.
Approximately 10–40% of horses that recover from the clinical disease have persistent infection of S. equi in the pharynx and guttural pouches for many months and are an important source of infection.9,10 Horses with clinically inapparent disease, such as some cases of guttural pouch empyema, may shed the organism for over 3 years.9 The period of infectivity is important in terms of the length of quarantine that needs to be imposed on horses that have apparently recovered from the disease. Because shedding of S. equi may be intermittent, repeated culture of nasopharyngeal swabs or use of PCR examination of guttural pouch washings may be necessary to document the carrier status of individual horses.10 Endoscopic or radiographic examination of clinically inapparent shedders may demonstrate lesions in the guttural pouches, paranasal sinuses, or pharynx.11 However, some persistent carriers of S. equi do not have detectable abnormalities of the nasopharynx. The clinically inapparent nature of the infection makes detection of carriers problematic, especially when considering introduction of horses into a previously closed herd in which strangles is not endemic.
Animal risk factors
Strangles is more common in young or naive horses, although the disease can occur in horses of any age. Animals that have previously had the disease are less likely than naive animals to develop the disease on subsequent exposure.3,6,12 A proportion (approximately 25%) of horses that recover from the disease do not develop a protective immune response and are susceptible to reinfection and a second bout of strangles.3,6 Resistance to the disease is associated with the production of serum and mucosal IgG antibodies to the streptococcal M protein.6 The presence in the nasopharynx of antibodies to streptococcal M protein is thought to be important in conferring resistance to the disease.3 Serum IgGb antibodies specific for SeM protein, which is important in the antiphagocytic activities of S. equi, are produced by most but not all horses during convalescence. Similarly, IgA and IgGb against SeM protein are detectable on nasal and pharyngeal mucosa after S. equi infection but not after intramuscular administration of vaccines containing M protein.13 Serum bactericidal activity alone is not considered to be a good indicator of resistance to the disease, especially if it is induced by administration of a vaccine.1,3 Antibodies similar to those found in the nasopharynx after infection with S. equi are present in colostrum and milk of mares that have recovered from the disease, are passed to foals via the colostrum and are secreted into the foal’s nasopharyngeal mucosa.12 These acquired antibodies are important in mediating the resistance of young foals to the disease.12
Although strong immunity occurs immediately after an attack, this immunity wanes with time and a horse may suffer further disease if the organism is virulent.
Importance
Strangles is one of the most important diseases of horses in developed countries, accounting for up to 30% of reported infectious disease episodes.1 The disease is important not only because of the deaths that it causes but more importantly because of the disruption of the management of commercial horse establishments, the time necessary to treat affected horses and the esthetic unpleasantness of the running noses and draining abscesses.
PATHOGENESIS
Virulence of S. equi is attributable to the presence of M proteins on the surface of the bacteria, a hyaluronic acid capsule and the production of a leukocidal toxin.2 M proteins are associated with S. equi adhesion to oral, nasal, and pharyngeal tissues, invasion of pharyngeal tonsils and associated lymphoid structures, and evasion of the innate host immune response.13 S. equi produces two M proteins – SeM and SzPSe. SeM is unique to S. equi and plays a dominant role in resistance of the organism to phagocytosis.14 Variations in structure of M protein are associated with decreased virulence.15 The M proteins interfere with the deposition of complement component 3b on the surface of the bacteria and bind fibrinogen, both of which reduce the susceptibility of the bacteria to phagocytosis by neutrophils.16,17 The antiphagocytic activity of S. equi reduces the efficacy of neutrophils in engulfing and destroying the bacteria.
The capsule of S. equi is associated with resistance to non-immune phagocytosis and pathogenicity. Strains of S. equi that do not produce a capsule do not induce disease, although they are able to infect guttural pouches and cause seroconversion in experimental studies.18
Following exposure of the oral and nasopharyngeal mucosal surfaces to S. equi, bacteria lodge in the pharyngeal and tonsillar lymphoid tissues where they multiply rapidly. There is no evidence of colonization of mucosal surfaces and streptococci can be detected in pharyngeal tonsils within hours of exposure.19 The binding of S. equi to pharyngeal cells is caused by fibrinogen binding proteins associated with M protein.14 The resistance of S. equi to non-immune phagocytosis results in accumulation of large numbers of organisms surrounded by degenerating neutrophils. Release of streptolysin S and streptokinase may contribute to tissue damage by directly injuring cell membranes and indirectly through activation of plasminogen.19 Bacteremia may occur. Migration of neutrophils into the lymph nodes causes swelling and abscessation, with associated disruption of lymph drainage and development of edema in tissues drained by the affected nodes. Swelling of retropharyngeal lymph nodes may interfere with deglutition and respiration. Most abscesses eventually rupture and drain and the infection resolves with the development of an effective immune response. Nasal shedding of S. equi usually begins 4–7 days after infection, or 2 days after onset of fever, and persists for 2–3 weeks in most horses but up to years in exceptional horses. Cessation of shedding accompanies development of an effective serum and mucosal immune response.12
Death is usually due to pneumonia caused by aspiration of infected material, although other causes of death include asphyxiation secondary to upper airway swelling and impairment of organ function by metastatic infection. Rare deaths also occur as a result of infarctive purpura hemorrhagica in horses infected with S. equi.20
Metastatic infection of the heart valves, brain, eyes, joints, and tendon sheaths or other vital organs may occur and cause a chronic illness and eventual death. Metastatic infection may occur because of bacteremia or extension of infection along chains of lymph nodes. Purpura hemorrhagica may occur as a sequela to S. equi infection and is associated with high serum antibody titers to SeM.
CLINICAL FINDINGS
The disease manifests as an acute disease of varying severity, chronic infection of retropharyngeal lymph nodes and guttural pouches, and as chronic disease associated with metastatic infection of organs distant to the upper respiratory tract. The severity of the acute disease varies with the age and immune status of the animal, and size of the inoculum and duration of exposure to infection. The term strangles derives from the enlarged retropharyngeal lymph nodes and guttural pouches causing respiratory distress in severely affected equids.
Acute disease
The acute disease is characterized by mucopurulent nasal discharge and abscessation of submandibular and retropharyngeal lymph nodes. After an incubation period of 1–3 weeks the disease develops suddenly with complete anorexia, depression, fever (39.5–40.5°C, 103–105°F), a serous nasal discharge, which rapidly becomes copious and purulent, and a severe pharyngitis and laryngitis.21,22 Rarely there is a mild conjunctivitis.
Lymphadenopathy becomes apparent as the submandibular lymph nodes enlarge and palpation elicits a painful response. The pharyngitis may be so severe that the animal is unable to swallow and there is a soft, moist cough. The head may be extended.
The febrile reaction commonly subsides in 2–3 days but returns as the characteristic abscesses develop in the lymph nodes of the throat region. The affected nodes become hot, swollen, and painful. Swelling of the retropharyngeal lymph nodes may cause obstruction of the oro- and nasopharynx with subsequent respiratory distress and dysphagia. Death by asphyxiation may occur at this time in severe cases. Obvious swelling of the nodes may take 3–4 days to develop; the glands begin to exude serum through the overlying skin at about 10 days and rupture to discharge thick, cream-yellow pus soon afterwards. Average cases run a course of 3 weeks, severe cases may last as long as 3 months.
Retropharyngeal abscesses may rupture into the guttural pouches, resulting in guttural pouch empyema and ultimately in prolonged infection and formation of chondroids. Retropharyngeal lymph node abscessation may not be apparent on external evaluation and can often only be detected by radiographic or endoscopic examination of the pharynx. Infection of retropharyngeal lymph nodes and guttural pouches is important in persistent infection and carrier status of some horses.
If the infection is particularly severe, many other lymph nodes, including the pharyngeal, submaxillary, parotid and retrobulbar nodes, may abscess at the same time. Local abscesses may also occur at any point on the body surface, particularly on the face and limbs, and the infection may spread to local lymphatic vessels causing obstructive edema. This occurs most frequently in the lower limbs, where edema may cause severe swelling. Abscess formation in other organs probably occurs at this time.
An atypical form of the disease may occur and is characterized by widespread subclinical infection and a mild disease. Affected horses have a transient fever for 24–48 hours and a profuse nasal discharge, and are anorexic. A moderate enlargement of the mandibular lymph nodes occurs in only about one-half of the affected horses.
Strangles in burros is a slowly developing debilitating disease. At postmortem examination the characteristic lesions consist of caseation and calcification of abdominal lymph nodes.
Complications
Complications occur in about 20% of cases.5,6 The most common fatal complication is the development of suppurative necrotic bronchopneumonia, which probably occurs secondary to the aspiration of pus from ruptured abscesses in the upper airway, or metastatic infection of the lungs.23
Extension of the infection into the guttural pouches, usually as a result of rupture of retropharyngeal lymph nodes into the medial compartment, causes empyema, which may lead to the formation of accretions of inspissated pus (chondroids). Involvement of the guttural pouches is evident clinically as distension and, after resolution of other signs, unilateral or bilateral nasal discharge. Guttural pouches of affected horses should be examined endoscopically for evidence of retropharyngeal abscessation or guttural pouch empyema or chondroid formation.
Retropharyngeal lymphadenopathy may impair the function of the recurrent laryngeal nerves, with subsequent unilateral or bilateral laryngeal paresis and consequent respiratory distress.
Metastatic infection (‘bastard strangles’) results in the formation of abscesses in any organ or body site but most commonly in the lungs, mesenteric lymph nodes, liver, spleen, kidneys, and brain. Clinical signs depend on the organ affected and the severity of the infection, but intermittent fever, chronic weight loss and sudden death due to rupture of abscesses into a body cavity are common manifestations of metastatic infection. Rectal examination or percutaneous ultrasonographic examination may reveal intra-abdominal abscesses in some horses with metastatic abscesses in the abdomen. Peritoneal fluid from these horses is often abnormal.
Metastatic infections may occur in the central nervous system. Extension of infection to the meninges results in suppurative meningitis characterized clinically by excitation, hyperesthesia, rigidity of the neck, and terminal paralysis. Abscesses in the brain cause a variety of clinical signs, depending on location of the abscess, including severe depression, head pressing, abnormal gait, circling, and seizures.24 Metastatic infections of the ocular and extraocular structures, heart valves and myocardium, joints, bones, tendon sheaths, and veins may occur.
Purpura hemorrhagica can occur as a sequela to S. equi infection.20,25
Two myopathic syndromes occur with S. equi infection in horses.26 Muscle infarction, which may be extensive, is assumed to result from immune-mediated vasculitis associated with purpura hemorrhagica.20 Often the muscle lesions in these horses are associated with other lesions consistent with severe purpura hemorrhagica, including infarctions in the gastrointestinal tract, skin and lungs. Rhabdomyolysis and subsequent muscle atrophy results in signs of muscle disease, including stilted gait and elevated serum activity of creatine kinase and other muscle-derived enzymes, and is assumed to be due to cross-reactivity of anti-SeM antibodies with myosin.26
Myocarditis and glomerulonephritis have been suggested as sequelae to S. equi infection but have not been conclusively demonstrated to occur.
CLINICAL PATHOLOGY
Hematological abnormalities during the acute phase of the disease include leukocytosis with a neutrophilia reaching a peak as the lymph nodes abscess. Hyperfibrinogenemia is characteristic of both the acute and chronic disease. Hematological and biochemical abnormalities associated with metastatic infection depend on the site of the infection and its severity. Leukocytosis with a hyperproteinemia attributable to a polyclonal gammaglobulinemia is characteristic of metastatic and chronic abscessation. Hypoalbuminemia may be present. Serum biochemical profile may reveal evidence of specific organ dysfunction. There may or may not be an anemia, which may be due to the hemolytic effect of streptolysin O, immune-mediated hemolysis, or anemia of chronic disease.
A commercial test that measures the serum IgG antibody titer to SeM has been developed and has been used to determine response to vaccination, suitability for vaccination and presence of metastatic infection.19,27 The test is not useful in diagnosis of the acute disease. Serum antibody titers to SeM are very high (>1:12800) in horses with metastatic infection or purpura hemorrhagica.
PCR testing is useful to detect shedding of S. equi DNA. The test is reported to be more specific than culture for detection of S. equi shedding.10 The PCR does not differentiate between live and dead S. equi and false-negative results occur in the presence of large numbers of S. equi. PCR testing should be considered an adjunct to routine culture for detection of S. equi.
Culture of nasal, pharyngeal, guttural pouch, or abscess discharge will usually yield S. equi in horses with active disease or in carriers. Abscesses may rapidly become contaminated with S. zooepidemicus, which may impede isolation of S. equi.
NECROPSY FINDINGS
In the rare fatalities that occur, necropsy examination usually reveals suppuration in internal organs, especially the liver, spleen, lungs, pleura, and peritoneum. When the last is involved, it is usually as a result of extension from abscesses in the mesenteric lymph nodes. The microscopic changes of abscessation and suppurative lymphadenitis are uncomplicated. The widespread ecchymotic hemorrhages of purpura hemorrhagica are not specific to this infection, but S. equi should always be investigated as a potential cause of such lesions.
DIAGNOSTIC CONFIRMATION
Confirmation of strangles depends on the isolation of S. equi from nasopharyngeal swabs and discharges from abscesses. Shedding of S. equi in nasal discharges begins 1–4 days after the onset of fever, and ruptured abscesses often become contaminated with Streptococcus zooepidemicus and S. equisimilis.
TREATMENT
The specific treatment of choice for S. equi infection of horses is penicillin, either as procaine penicillin G (22000 IU/kg intramuscularly every 12 h) or potassium or sodium penicillin G (22000 IU/kg intravenously every 6 h). Tetracycline (6.6 mg/kg intravenously every 12–24 h) and sulfonamide–trimethoprim combinations (15–30 mg/kg orally or intravenously every 12 h) may be efficacious but should only be used if penicillin cannot be administered. Aminoglycosides, such as gentamicin or amikacin, and the fluoroquinolones are not effective.
See Table 16.1 for a list of differential diagnoses of infectious upper respiratory tract disease of horses. Pneumonia should be differentiated from pleuropneumonia associated with transport or other stress. Chronic weight loss due to metastatic infection should be differentiated from equine infectious anemia, parasitism, inadequate nutrition and neoplasia, especially gastric squamous cell carcinoma, alimentary lymphosarcoma and granulomatous enteritis.
There is considerable debate about the treatment of horses with strangles. Folklore and anecdotal reports suggest that antibiotic treatment of horses with strangles is contraindicated because it promotes the development of metastatic infection. There is no experimental or empirical evidence to support this contention and horses with strangles should be treated with therapeutic doses of an appropriate antibiotic, such as procaine penicillin, for a period of time sufficient to effect a cure, as appropriate.
Treatment for S. equi infection depends on the stage of the disease:28
• Horses with early clinical signs including fever, anorexia, depression, and purulent nasal discharge should be isolated and treated with therapeutic doses of penicillin for at least 5 days. The purpose of treatment is to prevent further development of the disease in the affected animal, and to minimize environmental contamination with S. equi and transmission to other horses. Treatment should start as soon as clinical signs are observed and the full course of treatment should be completed to minimize the chances of recrudescence of the infection. Treatment at this stage causes rapid resolution of fever, anorexia, nasal discharge, and lymphadenopathy in individual horses and may abort an incipient outbreak of the disease in a stable or yard. However, treated horses may not develop a protective immune response and consequently may be at risk of reinfection if exposed to S. equi after completion of the course of treatment, leading one authority to recommend that only severely affected animals be treated28
• Horses with submandibular lymph node abscessation but without other clinical abnormalities probably do not require antibiotic treatment. Such horses should be isolated and efforts made to aid maturation and rupture of affected lymph nodes
• Systemic antibiotic therapy with penicillin is indicated in horses with advanced signs of strangles, including prolonged fever, depression, anorexia or dyspnea resulting from retropharyngeal lymphadenopathy. Retropharyngeal abscessation frequently responds to antimicrobial therapy, although surgical drainage may be required in some instances29
• Horses with metastatic infection require systemic penicillin therapy in combination with specific therapy for the complication. Pulmonary and mesenteric abscesses are problematic because they are usually not amenable to surgical drainage, and prolonged antimicrobial therapy is required to effect a cure
• Guttural pouch empyema requires either surgical drainage or repeated flushing of the affected pouch through the pharyngeal openings.11 Removal of pus and inspissated material in the guttural pouches can be achieved under endoscopic guidance. Alternatively, rigid or flexible indwelling catheters can be inserted for repeated flushing of the pouches with sterile isotonic electrolyte solutions (such as 0.9% NaCl) and topical medications. Substances and solutions that are irritating or injurious to mucus membranes, such as iodine, hydrogen peroxide and similar irritant compounds, should not be infused into the guttural pouches. Combined topical and systemic administration of potassium benzyl penicillin may be beneficial.30 Chondroids can often be removed using wire snares. Horses with metastatic or guttural pouch infections should be isolated
• Treatment of purpura hemorrhagica is dealt with elsewhere
• Management of horses that have been exposed to horses with strangles is controversial. Some authorities recommend treatment of such in-contact horses with penicillin until affected horses are isolated and no longer are a source of infection. However, close examination of exposed animals, including monitoring rectal temperature, and treatment of horses at the first sign of illness is probably a more reasonable approach.
Ancillary treatment consists of administration of nonsteroidal anti-inflammatory drugs (NSAIDs) to reduce swelling and provide pain relief, application of hot poultices to encourage rupture of abscesses, provision of intravenous hydration in animals unable to drink, and wound care, including cleaning of ruptured abscesses and application of petroleum ointment to surrounding skin to prevent scalding. Horses with severe upper airway obstruction may require placement of a short-term tracheotomy.
CONTROL
The principles of control measures include the prevention of transmission of S. equi from infected horses (cases or carriers) to susceptible animals and enhancement of resistance to infection and disease.
Prevention of transmission
Preventing introduction of strangles to herds free of the disease is difficult, especially when there is frequent movement of horses in and out of the herd. Close attention should be paid to the health of horses introduced to herds free of the disease and, although this is an imperfect safeguard, the owner or manager of horses to be introduced to a herd should be questioned about the likelihood of exposure of the horse to strangles. Ideally, horses should be isolated for 3 weeks before introduction to a herd that is free of the disease and should have washings of the guttural pouches performed on three occasions at weekly intervals. The washings should be examined by culture and PCR for the presence of S. equi. Clearly, this requirement, while ideal, is demanding of time, space and labor, is expensive and would be difficult to implement as a routine practice.
Methods to control transmission of S. equi on affected premises are detailed in Table 16.2.19
Table 16.2 Aims and associated measures used to control transmission of Streptococcus equi in affected premises and herds
| Aim | Measure |
|---|---|
| Prevention of spread of S. equi infection to horses on other premises and to new arrivals on the affected premises | Stop all movement of horses on and off affected premises immediately and until the outbreak is controlled |
| Horses with strangles and their contacts should be maintained in well demarcated quarantine areas | |
| Clustering of cases in groups allow parts of the premises to be allocated as contaminated or clean | |
| Establish whether clinically recovered horses are carriers | At least three nasopharyngeal swabs or washings taken at weekly intervals from all recovered cases and their contacts and examined by culture and PCR |
| Horses that are consistently negative are returned to the clean area | |
| Investigate apparently healthy horses from which S. equi is recovered | Endoscopic examination of the upper airways and guttural pouches |
| Eliminate S. equi from guttural pouches | Treatment of guttural pouches, as detailed under ‘Treatment’ |
| Prevention of infection of uninfected horses by S. equi from infected horses | Personnel should have dedicated protective clothing when dealing with infected horses |
| Personnel should not deal with infected and uninfected horses. If this is not possible, then infected horses should be dealt with after uninfected horses | |
| Strict hygiene should be implemented, including provision of disinfection facilities for personnel and diligent and thorough cleaning of stables and barns. | |
| If practicable, equipment should be destroyed after use with infected horses | |
| Organic material should be removed from stables and then appropriate phenolic disinfectants or steam should be applied. This cleaning should be repeated | |
| Feces and waste from infected animals should be composted in an isolated location | |
| Uninfected horses should not be introduced to pastures used to house infected horses for 4 weeks | |
| Water troughs should be disinfected daily. Horse vans should be thoroughly cleaned and disinfected after each use |
PCR, polymerase chain reaction.
Source: modified and used with permission from Sweeney CR et al. J Vet Intern Med 2005; 19:123–134.
• Infected animals should be isolated immediately
• All potential sources of fomites – including pails, brooms, grooming brushes and blankets – should be thoroughly cleaned and disinfected and the bedding burned. Disinfection with phenolic compounds is preferred because they retain their activity in the presence of some organic matter, whereas bleach and quaternary ammonium compounds are inactivated by organic material31
• Emergency prophylactic treatment, using injections of benzathine penicillin every 48 hours in foals and yearlings that are most susceptible, has been used but most treated animals develop strangles when the treatment is discontinued.28 This method of prophylaxis is not recommended
• People who care for affected horses should, ideally, avoid contact with susceptible animals. If this is not practical, then strict isolation protocols, including the wearing of protective boots and clothes that are changed between affected and normal horses, should be implemented
• Newly introduced animals should be quarantined for 3 weeks and observed for signs of strangles. Rectal temperature should be monitored twice daily
• Horses with elevated temperatures should have nasopharyngeal or guttural pouch swabs cultured
• Ideally, recovered animals should not be mingled with susceptible animals for a period of several months. The optimum length of a quarantine period has not been determined, but it should be recalled that prolonged shedding (several months to years) occurs in horses with lesions of the guttural pouch. Animals without such lesions can shed S. equi for at least 60 days.
Enhanced resistance
The majority of horses develop solid immunity to strangles after recovery from the spontaneous disease. This immunity lasts for up to 5 years in approximately three-quarters of recovered horses.6 Maximum resistance to disease probably requires both systemic and mucosal immunity to a variety of S. equi factors including, but not limited to, M protein.19,32
The efficacy of vaccination of adult horses with S. equi bacterins or M protein extracts of S. equi administered intramuscularly is controversial. Administration of M protein vaccines elicits an increase in the concentration of serum opsonizing antibodies but does not confer a high degree of resistance to natural exposure.3 Anecdotal and case reports suggest that vaccination is not effective. However, in a controlled field trial, vaccination with an M protein commercial vaccine three times at 2-week intervals reduced the clinical attack rate by 50% in a population of young horses in which the disease was endemic.33 Horses vaccinated only once were not protected against strangles.33 This result suggests that, in the face of an outbreak, vaccination may reduce the number of horses that develop strangles but will not prevent strangles in all vaccinated horses. A common vaccination protocol involves the administration of an M protein vaccine intramuscularly for an initial course of three injections at 2-week intervals, with further administration of the vaccine every 6 months in animals at increased risk of contracting the disease.34 On breeding farms, vaccination of mares during the last 4–6 weeks of gestation and of the foals at 2–3 months of age may reduce the incidence of the disease.35
The vaccines are administered by the intramuscular route and frequently cause swelling and pain at the injection site.34 Injection site reactions are usually less severe with the M protein vaccines. Injection into the cervical muscles may cause the horse to be unable to lower its head to eat or drink for several days – injection into the pectoral muscles is preferred for this reason. There are reports of purpura hemorrhagica, the onset of which was temporally associated with administration of a S. equi vaccine. Owners should be clearly warned of the limited efficacy and potential side-effects of vaccination.
Foals that receive adequate high-quality colostrum from exposed or vaccinated mares have serum and nasopharyngeal mucosal immunoglobulins (IgGb) that provide them with resistance to S. equi infection.6 This passive immunity wanes at approximately 4 months of age. Vaccination of brood mares 1 month before foaling increases colostral IgG antibodies to M protein, and presumably serum and mucosal immunoglobulin concentrations in their foals,3 but the efficacy of this approach in preventing strangles in foals is not reported.
An intranasal vaccine of an avirulent live strain of Streptococcus equi has recently been developed and appears useful. The vaccine is composed of a live variant (strain 707–27) that does not possess a capsule and is therefore avirulent when administered intranasally. Anecdotal reports suggest that recent manipulation of the genome by deletion of genes HasA and HasB, associated with formation of the capsule, has increased the genetic stability of the vaccine strain.36 The live attenuated vaccine should only be administered intranasally to healthy horses. The efficacy of the vaccine in field situations, safety in the face of an outbreak and in pregnant mares, incidence of adverse effects and risk of reversion to virulence have not been reported. It should not be used in potentially exposed horses during an outbreak of the disease. Intramuscular injection of the vaccine results in the formation of abscesses. The vaccine should not be administered to horses concurrently with intramuscular administration of other vaccines because of the risk of contamination of needles and syringes with S. equi vaccinal strain and subsequent development of abscesses at injection sites.
Vaccination by submucosal injection of a modified live vaccine is reported to provide short-lived (90-day) immunity to disease.37 The commercial form of the vaccine is administered into the submucosal tissues of the upper lip and is recommended for use in horses at moderate to high risk of developing strangles.38 At present there is no evidence of reversion of the vaccinal strain to virulence, and horses developing strangles subsequent to vaccination have all been infected with virulent strains of S. equi, apparently before development of immunity as a result of vaccination.38
1 Chanter N. J Appl Microbiol. 1997;83:100s.
2 Al-Ghamdi GM. Am J Vet Res. 2000;61:699.
3 Timoney JF. Vet Clin North Am Equine Pract. 1993;9:365.
4 Jorm LR. Aust Vet J. 1990;67:436.
5 Sweeney CR. J Am Vet Med Assoc. 1989;194:1281.
6 Hamlen HJ, et al. J Am Vet Med Assoc. 1994;204:768.
7 Ford J, Lokai MD. Equine Pract. 1980;2:41.
8 Jorms LR. In: Proceedings of the Sixth International Conference on Equine Infectious Diseases 1991:39.
9 Newton JR, et al. Vet Rec. 1997;140:84.
10 Newton JR. Equine Vet J. 2000;32:515.
11 Newton JR, et al. Equine Vet Educ. 1997;9:98.
12 Galan JE, et al. Infect Immun. 1986;54:202.
13 Meehan M. Microbiology. 2001;147:3311.
14 Harrington DJ. Microbes Infect. 2002;4:501.
15 Chanter N. Microbiology. 2000;146:1361.
16 Boschwitz JS, Timoney JF. J Immunol. 1994;62:3515.
17 Boschwitz JS, Timoney JF. Microb Pathog. 1994;17:121.
18 Anzai T. Vet Microbiol. 1999;67:277.
19 Sweeney CR, et al. J Vet Intern Med. 2005;19:123.
20 Kaese HJ, et al. J Am Vet Med Assoc. 2005;226:1893.
21 Dagleish R. Vet Rec. 1993;132:528.
22 Fintl C. Vet Rec. 2000;147:480.
23 Sweeney CR, et al. Compend Contin Educ Pract Vet. 1987;9:845.
24 Spoormakers TJ. Equine Vet J. 2003;35:146.
25 Pusterla N. Vet Rec. 2003;153:118.
26 Valberg SJ. Proc Am Assoc Equine Pract. 1996;42:292.
27 Sheoran AS, et al. Vet Immunol Immunopathol. 1997;59:239.
28 Sweeney CR. Equine Vet Educ. 1996;8:317.
29 Golland LC, et al. Aust Vet J. 1995;72:161.
30 Verheyen K, et al. Equine Vet J. 2000;32:527.
31 Dwyer RM. Equine Dis Q. 1996;4:4.
32 Flock M, et al. Infect Immun. 2004;72:3228.
33 Hoffman AM, et al. Am J Vet Res. 1991;52:589.
34 Smith H. Vet J. 1994;71:410.
35 Wilson WD. J Am Vet Med Assoc. 1995;207:429.
36 Walker JA, Timoney JF. Vet Microbiol. 2002;89:311.
STREPTOCOCCUS ZOOEPIDEMICUS INFECTION
Streptococcus equi var. zooepidemicus (S. zooepidemicus) is one of the bacteria most commonly isolated from the upper respiratory tract of clinically normal horses.1 Almost all horses harbor a number of antigenic types of S. zooepidemicus in their tonsils, and this may be the source of opportunistic infections of other body systems, including the lungs.1 Pneumonia in an individual horse is usually associated with a single strain of S. zooepidemicus, based on variants of the M-protein-like compound SzP.1 However, there are a large number of SzP phenotypes and it does not appear to be an important determinant of invasiveness or epizootic capability.1 S. zooepidemicus is also frequently isolated from horses with pleuropneumonia, endometritis, neonatal septicemia, abortion, and mastitis, suggesting a role for this organism in the pathogenesis of these diseases.2,3 S. zooepidemicus is likely important in the development of respiratory disease in foals and adult horses.4-7 S. zooepidemicus was isolated from 88% of foals with clinical evidence of lower respiratory tract disease, and isolation of the organism was associated with an increased proportion of neutrophils in bronchoalveolar lavage fluid,4 suggesting a causal role for this organism. Similarly, the number of S. zooepidemicus isolated from tracheal aspirates of adult horses is directly proportional to the number of neutrophils in the aspirate and the probability that they have a cough.5,8,9 The association of S. zooepidemicus and inflammatory airway disease in race horses is independent of previous viral infection, suggesting a role for S. zooepidemicus as a primary pathogen.7 Presence and number of colony forming units (cfu) of S. zooepidemicus in tracheal aspirates of horses is significantly associated with the risk of the horse having inflammatory airway disease.7,10 Adult horses dying of pneumonia associated with transportation often yield S. zooepidemicus on culture of lung lesions and the disease can be reproduced experimentally.6,11 These results clearly demonstrate a role for S. zooepidemicus in the pathogenesis of respiratory disease of horses. However, it is unclear whether S. zooepidemicus is a primary cause of disease, a secondary contaminant or an invader of airways compromised by viral infection or other agents.
Clinical signs of S. zooepidemicus infection of the lower respiratory tract of foals and horses include coughing, mild fever, mucopurulent nasal discharge, and increased respiratory rate. Endoscopic examination of the trachea and bronchi reveals erythema and presence of mucopurulent exudate.4,5 Tracheal aspirates or bronchoalveolar lavage fluid of affected horses or foals have an increased (>10%) proportion of neutrophils. S. zooepidemicus is a frequent isolate from the cornea of horses with ulcerative keratitis.12
Treatment consists of the administration of antimicrobials including penicillin (procaine penicillin, 20000 IU/kg every 12 h) or the combination of a sulfonamide and trimethoprim (15–30 mg/kg orally every 12 h). Control consists of isolation to prevent spread of infectious respiratory disease and vaccination to prevent viral respiratory disease.
1 Anzai T, et al. Am J Vet Res. 2000;61:162.
2 Welsh RD. Equine Pract. 1984;6:6.
3 Chand P, et al. Indian J Anim Sci. 2001;71:445.
4 Hoffman AM, et al. Am J Vet Res. 1993;54:1615.
5 Wood JLN, et al. Equine Vet J. 1993;25:314.
6 Oikawa M, et al. J Comp Pathol. 1994;111:205.
7 Wood JL, et al. J Clin Microbiol. 2005;43:120.
8 Christley RM, et al. Equine Vet J. 2001;33:256.
9 Chapman PS, et al. Vet Rec. 2000;146:91.
10 Newton JR, et al. Prev Vet Med. 2003;60:107.
NEONATAL STREPTOCOCCAL INFECTION
Etiology Various Streptococcus spp.
Epidemiology Neonatal foals, calves, lambs, piglets
Signs Acute painful swelling of joints, lameness, fever. Signs of meningitis, omphalophlebitis, ophthalmitis. Sudden death
Clinical pathology Culture organism from joint fluid
Necropsy findings Fibrinopurulent synovitis, purulent meningitis and omphalophlebitis
Diagnostic confirmation Recovery of organism from joint fluid
Differential diagnosis Other infectious causes of arthritis, meningitis and omphalophlebitis
Treatment Antimicrobials, usually penicillin
Control See ‘Principles of control and prevention of infectious diseases of newborn farm animals’ in Chapter 3
ETIOLOGY
Streptococci are an important cause of septicemia, polyarthritis, meningitis, polyserositis, endocarditis, and unexpected death in the neonates of all farm animal species. Meningitis associated with streptococcal infection is restricted to the neonate in all species except piglets, where outbreaks can occur in pigs after weaning, and lambs infected with S. suis, where meningitis can occur as a sporadic disease at 3–5 months of age. Historically, there are reports of isolates of most of the Lancefield groups of beta-hemolytic streptococci, of non-beta-hemolytic streptococci and of viridans group streptococci from neonatal disease in farm animals. Commensal skin streptococci can occasionally cause disease in presumably immunocompromised neonates.1 However, the majority of neonatal disease is associated with a limited number of streptococcal species, although there can be geographical variation in their relative prevalence within animal species.
In foals, S. equi (S. equi subsp. equi) and S. zooepidemicus (S. equi subsp. zooepidemicus) are the most common streptococcal species recovered from septicemic disease and polyarthritis and are also a cause of placentitis and abortion in mares.2-4 S. equisimilis (S. dysgalactiae subsp. equisimilis) is a less common isolate.
S. suis and S. equisimilis are the most common species incriminated in piglets. S. suis is especially important and is presented separately in the next section. Other Lancefield groups have been associated with sporadic disease.5 In calves, S. dysgalactiae and S. uberis are the common streptococcal isolates from synovial fluid of neonatal calves with arthritis.6,7
S. dysgalactiae is also reported to be the most common cause of outbreaks of arthritis in neonatal lambs in Great Britain.8,9
Streptococci can also contribute to purulent infections at local sites, such as navel ill of all species or otitis media in neonatal calves.10
EPIDEMIOLOGY
Occurrence and prevalence
The importance and relative prevalence of streptococcal infections in neonatal disease varies among countries and with surveys.
Streptococci are a common cause of postnatal infections of foals, representing 50% of such cases in some surveys10 but with a lower prevalence in others.4,11 Up to 20% of abortions in mares are due to placentitis from streptococcal infection. Streptococcal septicemia due to beta-hemolytic streptococci may occur in foals under 5 days of age that have been stressed and have failure of transfer of passive immunity.12
In calves, neonatal infections with streptococci are usually sporadic and less common than infections with Gram-negative bacteria and may be predisposed by failure of transfer of passive immunity. In lambs, S. dysgalactiae is associated with outbreaks with high morbidity and in Great Britain is reported to be the cause of over 70% of cases of polyarthritis in lambs during their first 3 weeks of life. Despite the high attack rate in these outbreaks, it is rare for more than one of twins or triplets to have disease.8,9 Streptococcal arthritis associated with S. suis infection in piglets is a common disease and is covered in a separate section.
Source of infection
The source of the infection is usually the environment, which may be contaminated by uterine discharges from infected dams or by discharges from lesions in other animals. S. dysgalactiae is reported to survive for up to a year on clean straw, as opposed to wood shavings, which do not support the persistence of the organism.8
The portal of infection in most instances appears to be the umbilicus, and continued patency of the urachus is thought to be a contributing factor in that it delays healing of the navel. In piglets there can be high rates of infection associated with infection entering through skin abrasions such as carpal necrosis resulting from abrasive floors or facial lesions following fighting.13 Contaminated knives at castration and tail docking, or contaminated ear taggers, can result in infection and disease. Other mechanical vectors include the screwworm fly (Cochliomyia americana).
The organism can be isolated from the nasopharynx of the sow, and direct infection from the sow to the piglet is suggested by some epidemiological data.
Economic importance
Affected foals and other species may die or be worthless as working animals because of permanent injury to joints. There is also loss due to condemnation at slaughter.14
Zoonotic implications
S. zooepidemicus is associated with human infections, particularly nephritis, and many human infections can be traced back to the consumption of contaminated animal food products. Some strains of S. equisimilis can also infect humans.15
PATHOGENESIS
The infection spreads from the portal of entry to produce a bacteremia that is not detectable clinically. The period of bacteremia is variable but it may last several days in piglets. A terminal acute fatal septicemia is the common outcome in animals under 1 week of age but in older animals suppurative localization in various organs is more common. Arthritis is the most common manifestation, with synovitis and invasion of medullary bone of the epiphysis with microabscessation and ischemic necrosis of bone. Other manifestations of infection include ophthalmitis in foals and calves, meningitis and endocarditis in piglets, meningitis in calves, and endocarditis and sudden death in lambs. Streptococcal endocarditis can be produced by the intravenous inoculation of group L Streptococcus. Lesions are well established within 5 days, the left heart is most commonly affected, and myocardial and renal infarction occur.
CLINICAL FINDINGS
Foals
Foals do not usually show signs until 2–3 weeks of age. The initial sign is usually a painful swelling of the navel and surrounding abdominal wall, often in the form of a flat plaque, which may be 15–20 cm in diameter. A discharge of pus may or may not be present and a patent urachus is a frequent accompaniment. A systemic reaction occurs but this is often mild, with the temperature remaining at about 39.5°C (103°F). Lameness becomes apparent and is accompanied by obvious swelling and tenderness in one or more of the joints. The hock, stifle, and knee joints are most commonly affected but in severe cases the distal joints are involved and there is occasionally extension to tendon sheaths. Lameness may be so severe that the foal lies down most of the time, sucks rarely and becomes extremely emaciated. There may be hypopyon in one or both eyes.
Recovery occurs if treatment is begun in the early stages. When joint involvement is severe, particularly if the abscesses have ruptured, the animal may have to be destroyed because of the resulting ankylosis. Death from septicemia may occur in the early stages of the disease.
Piglets
Arthritis and meningitis may occur alone or together and are most common in the 2–6-week age group. More commonly, several piglets within a litter are affected. The arthritis is identical with that described in foals above. With meningitis there is a systemic reaction comprising fever, anorexia and depression. The gait is stiff, the piglets standing on their toes, and there is swaying of the hindquarters. The ears are often retracted against the head. Blindness and gross muscular tremor develop, followed by inability to maintain balance, lateral recumbency, violent paddling and death. In many cases there is little clinical evidence of omphalophlebitis. With endocarditis the young pigs are usually found comatose or dead without premonitory signs having been observed.
Lambs
Lameness in one or more limbs of lambs up to 3 weeks of age is the common presenting sign of infection with S. dysgalactiae but approximately 25% of lambs can be initially recumbent. With this infection there is not major joint swelling in the early stages and myopathy or delayed swayback may be initial considerations. In contrast with outbreaks that occur following docking the incubation period is short, usually 2–3 days, and there is intense lameness, with swelling of one or more joints appearing in a day or two. Pus accumulates and the joint capsule often ruptures. Recovery usually occurs with little residual enlargement of the joints, although there may be occasional deaths due to toxemia.
CLINICAL PATHOLOGY
Pus from any source may be cultured to determine the organism present and its sensitivity to the drugs available. Bacteriological examination of the uterine discharges of the dam may be of value in determining the source of infection. The success rate with blood cultures is not very high but an attempt is worthwhile. The identification of the causative bacteria is important but the sensitivity of the organism may mean the difference between success and failure in treatment. The specific identity of the streptococcus should be determined.
NECROPSY FINDINGS
Suppuration at the navel and severe suppurative arthritis affecting one or more joints are usual. Abscesses may also be present in the liver, kidneys, spleen, and lungs. Friable tan masses of tissue are common on the heart valves of affected piglets and this valvular endocarditis may also be observed in other species. Peracute cases may die without suppurative lesions having had time to develop. Necropsy findings in the meningitic form in pigs include turbidity of the cerebrospinal fluid, congestion of meningeal vessels and the accumulation of white, purulent material in the subarachnoid space. Occasionally this exudate blocks the flow of cerebrospinal fluid in the ventricular system, causing internal hydrocephalus. Histologically there is infiltration of the affected tissue by large numbers of neutrophils, usually accompanied by fibrin deposition.
Samples for confirmation of diagnosis
• Bacteriology – culture swabs from joints, meninges, suppurative foci; tissue pieces of valvular lesions, lung, spleen, synovial membrane (CULT)
• Histology – formalin-fixed samples of a variety of organs, including brain, lung, spleen, liver (LM).
Omphalophlebitis and suppurative arthritis in foals may be due to infection with Escherichia coli, Actinobacillus equuli or Salmonella abortivoequina, but these infections tend to take the form of a fatal septicemia within a few days of birth whereas streptococcal infections are delayed in their onset and usually produce a polyarthritis. In pigs there may be sporadic cases of arthritis due to staphylococci but the streptococcal infection is the common one. Arthritis due to Mycoplasma hyorhinis is less suppurative but may require cultural differentiation. Glasser’s disease occurs usually in older pigs and is accompanied by pleurisy, pericarditis, and peritonitis. Erysipelas in very young pigs is usually manifested by septicemia. Nervous disease of piglets may resemble arthritis on cursory examination but there is an absence of joint enlargement and lameness. However, the meningitic form of the streptococcal infection can easily be confused with viral encephalitides. Meningitis in young calves may also be associated with Pasteurella multocida. Polyarthritis in calves, lambs, and piglets may also be associated with infection with Arcanobacterium pyogenes and Fusobacterium necrophorum. S. suis type 2 can also be the cause of meningitis in older pigs at 10–14 weeks of age.
The response of streptococcal infections to treatment with penicillin may be of value in the differentiation of the arthritides, and the microscopic and histological findings at necropsy enable exact differentiation to be made. In lambs suppurative arthritis occurs soon after birth and after docking. The other common arthritis in the newborn lamb is that associated with Erysipelothrix rhuriophiae but this usually occurs later and is manifested by lameness without pronounced joint enlargement. Calves may also develop erysipelatous arthritis.
TREATMENT
Penicillin is successful as treatment in all forms of the disease provided irreparable structural damage has not occurred. In newborn animals the dosage rate should be high (20000 IU/kg body weight (BW)) and should be repeated at least once daily for 3 days. If suppuration is already present, a longer course of antibiotics will be necessary, preferably for 7–10 days. Piglets treated early in the course of the disease will survive but may runt. Because of the common litter incidence in piglets and the occurrence of subclinical bacteremia it is wise to also treat all littermates of affected piglets. Benzathine or benethamine penicillins can be used in conjunction with shorter-acting penicillins. General aspects of treatment of the newborn are dealt with in Chapter 3.
CONTROL
The principles of control of infectious diseases of the newborn are described in Chapter 3. Because the most frequent source of infection in foals is the genital tract of the dam, some attempt should be made to treat the mare and limit the contamination of the environment. Mixed bacterins have been widely used to establish immunity in mares and foals against this infection but no proof has been presented that they are effective. On heavily infected premises the administration of long-acting penicillin at birth may be advisable. A major factor in the control of navel- and joint-ill in lambs is the use of clean fields or pens for lambing, as umbilical infection originating from the environment seems to be more important than infection from the dam in this species. Docking should also be done in clean surroundings and, if necessary, temporary yards should be erected. Instruments should be chemically sterilized between lambs. Regardless of species and where practicable, all parturition stalls and pens should be kept clean and disinfected and the navels of all newborn animals should be disinfected at birth. Where screwworms are prevalent, the unhealed navels should be treated with a reliable repellent.
1 Wichtel MEG. Vet Rec. 2003;153:22.
2 Ozgur NY. Israel J Vet Med. 2002;57:9.
3 Puran Chand, et al. Indian J Anim Sci. 2001;71:445.
4 Smith KC, et al. Equine Vet J. 2003;35:496.
5 Jones JET. J Comp Pathol. 1980;90:11.
6 Grondemange E, et al. Ir Vet J. 2002;55:237.
7 Fecteau G, et al. Can Vet J. 1997;38:95.
8 Watkins GH. State Vet J. 2000;101:15.
9 Watkins GH, Sharp MW. Vet J. 1998;156:235.
10 Platt H. Br Vet J. 1973;129:221.
11 Hong GB. J Vet Diagn Invest. 1993;5:56.
12 Dubielzig RR. J Equine Med Surg. 1978;2:28.
13 Zoric M, et al. J Vet Med B. 2004;51:278.
STREPTOCOCCUS SUIS INFECTION OF YOUNG PIGS
ETIOLOGY
There are three organisms that infect the neonatal pig quite commonly – Haemophilus parasuis, S. suis, and Actinobacillus suis, which have been dubbed the ‘suis-cides’.1 S. suis is therefore one of the early colonizers of the pig and by the end of the nursery period most pigs are infected.2 These authors suggested that virulence may be an attribute of strains that colonize the young pig poorly but infect older animals more easily in the absence of maternal antibody.
Etiology Streptococcus suis. At least 34 capsular types (35 if you include ½) have been recognized. Type 2 is most common
Epidemiology Occurs in piglets under 12 weeks of age
Signs Septicemia, arthritis, meningitis, pericarditis, endocarditis, polyserositis, and pneumonia
Clinical pathology Culture organism
Lesions Fibrinous polyserositis, fibrinous or hemorrhagic pneumonia, purulent meningitis, myocarditis, vegetative endocarditis, fibrinous arthritis
Diagnostic confirmation Culture organism from body tissues and blood
Differential diagnosis Arthritis due to:
Treatment Antimicrobials based on culture and sensitivity
Control Provision of optimum environment (temperature and relative humidity). Avoid overcrowding in nursery pens. Age spread of pigs in pens should not exceed 2 weeks. Use all-in, all-out pig flow. Control of other common infectious diseases. Avoid nutritional deficiencies. Consider mass medication of feed with antimicrobials
S. suis type 1 and S. suis type 2 were the original capsular types of the organism, which appeared to account for most epidemics of the disease. S. suis types are subgroups of Lancefield’s group D.3 There are now at least 34 known S. suis capsular types.4 Even now, new species of streptococcus, such as S. ferus, are being isolated from pigs.5
EPIDEMIOLOGY
The epidemiology of S. suis is very complex.6 There is a great genetic heterogeneity of S. suis. The isolation of different strains within the same herd and the predominance of particular strains within some herds are evidence that infection by S. suis is a dynamic process and reinforces the idea that the epidemiology is complex.
Occurrence
Diseases associated with S. suis occur worldwide, affecting pigs less than 2 weeks of age and up to 22 weeks of age. Most cases occur within a few weeks after weaning which is associated with stressors such as moving, mixing, overcrowding, and inadequate ventilation. S. suis type 2 causes outbreaks of meningitis in young pigs 10–14 days after weaning. The disease occurs most commonly in recently weaned pigs raised in intensive systems of high population density, such as flat-deck rooms and finishing pens. In the USA, most pigs infected with S. suis are under 12 weeks of age;7 sporadic cases occur in older pigs including adults. In Canada, pigs may be affected from 3–24 weeks of age, with most cases occurring between 6 and 12 weeks of age.
The organism has also been isolated from cattle,8 sheep, and goats,4,9 a horse with meningitis,10 fallow deer,11 and cats.12
Prevalence of infection
S. suis type 2 is the most prevalent serotype. Types 3, 4, 7, 8, and 14 have been isolated from affected pigs in the UK.13-15 In a survey of pigs at slaughter in Australia, S. suis type 2 was detected in 58% of the palatine tonsils, in 66% of the pneumonic lungs and in 28% of the healthy lungs.16 Overall, the carrier rate in piggeries was 60%. The organism was also present in the blood of 3% of apparently normal pigs at slaughter. It could also be cultured from many other tissues, including those of the reproductive tract of females but not from males. The organism can be cultured from the vagina of sows, and it is possible that piglets are infected during birth. Specific-pathogen-free herds are free of the organism and hysterectomy-derived piglets are born free from infection. The rate of infection of the environment of the pigs can also be very high.17
Since 1996, at least 34 serotypes of the organism have been identified.4 Recently it has been suggested that serotypes 32 and 34 are Streptococcus orisratti.18 In Canada, all 34 serotypes have been isolated, with type 2 being most prevalent of all isolates.4 The other capsular types in decreasing order were 3, 7, ½, 8, 23, and 4. Over a period of several years, more than 60% of isolates belong to capsular types 2, ½, 3, 4, 7, and 8.19 In a survey of clinically healthy piglets 4–8 weeks of age in Quebec, the organism could be isolated from 94% of piglets and 98% of farms.20 The typable isolates of the organism are more frequently recovered from pigs between 5 and 10 weeks of age, while untypable isolates are most frequently found in animals more than 24 weeks old. In the USA, serotype 3 was most prevalent (26.1%), followed by serotypes 8 (17.4%), 2, 4, and 7 (all 15.2%) with no significant differences in the epidemiological features, clinical signs or lesions in pigs infected with multiple serotypes compared with a single serotype of S. suis.7,21 Only Scandinavian countries reported a higher incidence of type 7 over type 2.22 In Denmark, serotypes 2 accounted for 29% of the isolates, serotype 7 for 17% and 3, 4 and 8 for a further 9–10%.23 Serotype 7 was isolated more frequently than reported in other countries, causing septicemia, arthritis, and meningitis. In Finland, the most common types isolated from dead pigs were 7, 3, and 2 respectively, and they were most frequently isolated from cases of pneumonia.24 In the Netherlands, type 2 was most frequently isolated from pigs with meningitis. S. suis type 9 and S. suis type 2 have been isolated as the cause of septicemia and meningitis in weaned pigs in Australia.25 The organism has been isolated from a wild sow affected with pneumonia26 and from a young wild boar.27
Morbidity and case fatality
The incidence of clinical disease ranges from 0–15%. In a 3-year survey of a breeding herd, the combined morbidity and mortality rates due to meningitis from S. suis type 2 were 3%, 8%, and 9.1%, respectively, in each of the 3 years.
Methods of transmission
The organism is carried in the tonsils and occasionally in the nose of healthy pigs, and transmission to uninfected pigs can occur within 5 days of mixing. The introduction of breeding gilts from infected herds results in disease appearing subsequently in weanlings and growing pigs in the recipient herds. The detectable carrier rates in different groups of pigs can vary from 0–80% and are highest in weaned pigs aged 4–10 weeks. Over 80% of the sows in an individual herd may be subclinical carriers. They do not normally carry the organism in the nasal cavity but in the vagina.28 Based on the results of sampling of sows and piglets at parturition, and being able to culture multiple serotypes of the organism from the sow’s vaginal secretions and oropharyngeal samples of piglets, it is highly probable that the newborn piglet is infected during birth by the organism, which is transferred from the sow’s vagina to the dorsal surface and oral cavity of the piglet.29 However, even though most pigs are colonized by weaning age, colonization by the virulent strains of S. suis type 2 takes longer and usually does not occur before 15 days of age.30 This could constitute a risk factor for developing disease later when maternal immunity has waned.
Weaned carrier pigs transmit the infection to previously uninfected pigs after mixing following weaning. The organism can persist in the tonsils of carrier pigs for more than 1 year, and in the presence of circulating opsonic and binding antibodies, and in pigs receiving penicillin-medicated feed. Thus the organism can be endemic in some herds without causing recognizable clinical disease. House flies can carry the organism for at least 5 days and can contaminate feed for at least 4 days.
The carrier rate in some surveys of slaughtered pigs ranges from 32–50% of pigs 4–6 months of age, which may be associated with the high incidence of S. suis type 2 meningitis in humans in the Netherlands. The organism is a potential hazard for abattoir workers, particularly eviscerators who remove the larynx and lungs from carcasses, who have a significantly higher risk of exposure to S. suis than other workers.
S. suis type 2 has also been isolated from pigs affected with bronchopneumonia, usually secondary to enzootic pneumonia, from cases of pleuropneumonia, arthritis, vaginitis and aborted fetuses, and in neonatal piglets 1–2 days of age affected with fatal septicemia. It appears that the organism is found in the lungs of pigs affected with pneumonia more frequently in North America than in other countries. Although airborne infection of type 2 has been described it is said that indirect transmission is a much better way to infect piglets.31
The organism is easily transmitted via fomites. It can survive in feces for 104 days at 0°C and for 10 days at 9°C, and in dust for 54 days at 0°C and 25 days at 9°C. Experimentally, pure cultures of the organism placed on rubber and plastic surfaces, especially when protected by swine manure, are viable at up to 55°C and can survive if kept frozen for up to 10 days. The organism is readily destroyed by disinfectants. It can be spread by contaminated pig nose snares and needles used for blood sampling.32
Risk factors
Animal risk factors
The host factors that render pigs susceptible to clinical disease are uncertain. Over 30 different Gram-positive bacterial organisms may occur in the nasal cavities and tonsils of unweaned pigs of between 2 weeks and 6 weeks of age.33 It is suggested that strains of S. suis type 2 vary in pathogenicity, and that the occurrence of disease is dependent on both exposure to a pathogenic strain and undetermined secondary factors. The peak incidence of the disease from 5–10 weeks of age suggests that the stressors of weaning may render pigs susceptible to clinical disease. The presence of other infectious diseases such as porcine reproductive and respiratory syndrome (PRRS) and Actinobacillus pleuropneumoniae may be associated with a higher than average prevalence of infection with S. suis.34 PRRS certainly increases susceptibility to S. suis infection experimentally.35,36 In utero infection with PRRS makes pigs more susceptible to subsequent neonatal S. suis infections.36 Infection of specific-pathogen-free pigs with PRRS virus may be a risk factor for infection and disease associated with S. suis.37 Similarly, the pseudorabies virus may enhance clinical disease associated with S. suis.38
Environmental and management factors
The incidence of clinical disease appears to depend on environmental factors such as inadequate ventilation, high population density, and other stressors. Several environmental and managemental risk factors have been associated with a high prevalence of pigs harboring S. suis in swine herds.34 Excessive environmental temperature fluctuation in the nursery pig facilities was the most common factor. Nursery pig environmental temperatures should not fluctuate more than 1.1–1.7°C over a 24-hour period to prevent chilling of pigs. Fluctuations in temperature are caused by drafts, inadequate heaters, or poorly insulated buildings. Excessive relative humidity was also a factor; the recommended range for nursery pigs is 55–70%. The third and fourth most common factors were age spread of more than 2 weeks for pigs in the same room, and crowding. The fifth most common factor was the use of continuous flow facilities, which allows for build-up of dust and manure and increased infection pressure. An unusual case where S. suis was isolated from the lumen of the small intestine occurred when a feed was formulated with no salt and 58.5 kg instead of 3.5 kg of vitamin premix. Once the ration was corrected the problem disappeared.39
Pathogen risk factors
There are now at least 34 serotypes of S. suis.4 The organism is divided into serotypes by the antigenic specificity of its capsular polysaccharide.40 The capsule, certainly for S. suis type 2, plays an important role in pathogenesis.41 As described under Methods of transmission, colonization of piglets occurs very early in life, most pigs being colonized by weaning age; virulent strains of S. suis type 2 may not colonize until later.30 Early colonization reduces the subsequent clinical signs.30 Despite the association of bacteria with disease, they may also be recovered from the nasal cavities and tonsils of healthy pigs. High numbers of organisms were isolated from the cerebrospinal fluid of clinically normal pigs.42 A study also showed that a persistent epidemic strain of S. suis was consistently isolated from the brains of pigs over a 2-year period, all with a similar genetic pattern.43
There are differences in pathogenicity between serotypes and between strains of the same serotype. In the UK there are differences in pathogenicity between types 1 and 2, type 1 causing less severe disease in piglets while type 2 causes a more severe and acute disease in older and growing pigs. Highly virulent and completely avirulent type 2 strains exist.44 Different strains of S. suis type 2 vary in their ability to cause meningitis. Streptococci require manganese but not iron as a growth factor, which affects the activity of superoxide dismutase in cell cultures.45
The virulence markers of the organism include the structural proteins muramidase-released protein (MRP) and extracellular factor (EF).46-48 It has also been said that they are not essential for virulence.48 S. suis produces four proteases.49 Fibrinogen-binding protein played a role in the colonization of specific organisms involved in a S. suis infection.50 A secreted nuclease has also been described.51 An IgG binding protein in the 60 kDa range has been shown to bind IgG in a nonimmune way.52 Recently, 36 environmentally regulated genes have been identified.53 There are virulence differences between strains of the same serotype based on the presence or absence of muramidase-released proteins. Strains of S. suis type 2 from Europe are genotypically different from those of North America. Most Canadian field isolates of S. suis type 2 do not produce these virulence-related proteins.54 An extracellular protein with hemolytic properties, called suilysin, has been described.55 Most type 2 field strains from four different European countries produce this hemolysin.56 Between 58 and 90% of strains from the Netherlands, Denmark, France, England, and Italy produce the suilysin but only 1% of Canadian strains do.54 A total of 164 field isolates from diseased pigs in four countries were serotyped and tested for suilysin. S. suis type 2 was the most prevalent type isolated from all four countries. After type 2, type 9 was most prevalent in the Netherlands and France and type 7 in Denmark. All the English isolates were type 2. No nonvirulent suilysin-producing S. suis type 2 strains have been reported.57
The hemolysin gene was found in over 80% of the strains that were associated with meningitis, septicemia, and arthritis but with only 44% of pneumonia isolates.58
The organism bears fimbriae and the capsular materials from different serotypes have distinct morphologies.59 Certain strains possess hemagglutinating properties.60 The type 2 isolates possess a factor that allows them to adhere to porcine lung.61 Restriction endonuclease techniques are being used to examine the genomic structure of the organism, which could be useful for epidemiological monitoring of outbreaks of the disease.62
A 44 kDa protein has been isolated as a virulence marker of S. suis type 2, and the presence of antibodies against this protein appears to be necessary to obtain complete protection against the disease.63 Australian isolates of S. suis are genetically very diverse, which suggests that serotyping is not a reliable technique for identifying specific strains and not a good predictor of the genetic background of a given isolate.64
Some isolates of S. suis have developed resistance to penicillin and tetracycline, which may reflect the usage of these antimicrobials in treatment and control programs.34
S. suis type 2 can survive in feces for 104 days at 0°C (32°F), up to 10 days at 9°C (48°F) and up to 8 days at 22–25°C (71–77°F).65 It can survive in dust for up to 25 days at 9°C (48°F) but could not be isolated from dust stored at room temperature for 24 hours. The organism is rapidly inactivated by disinfectants commonly used on farms. Liquid soap inactivates S. suis type 2 in less than 1 minute at a dilution in water of 1 in 500. The organism can survive in pig carcasses at 40°C (104°F) for 6 weeks, which may be an important source of the organisms for infection in humans.
Zoonotic implications
Splenectomized humans are particularly at risk from certain infections, including streptococci.
Infections with S. suis type 2 are the most common infections in humans and occur most commonly in people having contact with pigs or raw pork. It is possible that many human cases are misdiagnosed, such as were described in south-east Asia,66 where five of eight cases of S. suis were described as S. viridans. A truck driver has recently been described with septic shock.67 In the UK the highest incidence of meningitis due to S. suis type 2 is in butchers and abattoir workers; transmission is thought to be mainly via minor skin abrasions. The clinical manifestations include meningitis and septicemia, which may be accompanied by arthritis, endophthalmitis, and disseminated intravascular coagulation. Endocarditis68 and acute gastroenteritis have also been reported.69 In a series of 35 human cases of S. suis type 2 meningitis in the UK from 1975–1990, deafness occurred in 50% of patients, vertigo and ataxia in 30% and arthritis in 53%.62 There was a case fatality rate of 13%. The organism invades the cerebrospinal fluid within monocytes, an example of the ‘Trojan horse’ mechanism of entry. Deafness may follow in 50–60% of cases and is due to cochlear sepsis following invasion of the organism from the subarachnoid space into the perilymph of the inner ear. Subclinical infection in pigs sent to slaughter represents a potential source of infection for abattoir workers; eviscerators who remove the larynx and lungs have a significantly higher risk of exposure to the organism than other abattoir workers. Within infected herds in New Zealand, up to 100% of pigs are carriers and S. suis type 2 infection may be one of the most infectious potentially zoonotic pathogens present in New Zealand, although very rarely resulting in clinical disease.70 The annual incidence of subclinical infection and seroconversion in pig farmers in New Zealand is close to 28%.
PATHOGENESIS
Streptococci exist in extremely different phenotypes with regard to adhesion, invasion and cytotoxicity. These features depend on the state of encapsulation and environmental growth conditions.71
The tonsils are a site of persistence, multiplication and portal of entry of a variety of pathogens including S. suis.72
The organism resides in the crypts of the palatine tonsils. Invasive disease occurs in a minority of infected pigs, following entry of organisms into the systemic circulation as a bacteremia and then septicemia, leading to a meningitis.73 Most bacteria remain extracellular, with fewer than 2% of monocytes containing bacteria. There is a high level of adhesion of bacteria to phagocytic cells. S. suis adheres to brain microvascular endothelial cells and suilysin can damage these.74 It has been shown that S. suis capsular strains stimulate tumor necrosis factor alpha (TNF)- and interleukin (IL)-6 but that the suilysin and the extracellular protein do not do so on their own.75 A terminal acute fatal septicemia is the common outcome in young animals, but in older animals localization can occur in synovial cavities, endocardium, eyes, and meninges. A persistent bacteremia is an important phase in the pathogenesis of S. suis type 2 meningitis. Virulent isolates of S. suis type 2 possess capsules and are relatively resistant to phagocytosis.76 Isogenic mutants defective in capsule production were not virulent.77 S. suis is able to adhere to but not to invade epithelial cells and the adhesins are partially blocked by the capsule and are part of the cell wall.78 The highly virulent isolates possessed the suilysin, muraminidase releasing protein and extracellular protein factor phenotype.79 S. suis are able to survive and replicate within macrophages, and the bacteria enter the cerebrospinal fluid space in association with migrating monocytes, which move through choroid plexuses.80 They therefore enter the cerebrospinal fluid by a ‘Trojan horse’ mechanism similar to that used by some viral pathogens of the central nervous system.80 The predominant lesions are suppurative or fibrinopurulent inflammation in brain, heart, lungs, and serosae.81 S. suis type 9 may produce a different distribution of lesions compared to type 2.82 The disease has been reproduced experimentally in pigs and laboratory animals by intravenous, intranasal, and subarachnoid routes.83 Certain strains of S. suis can cause vascular lesions, with the development of fibrinohemorrhagic pneumonia and septal necrosis.84 Of importance in the pathogenesis of S. suis infections is the predisposing role of PRRS. This effect of PRRS has still only been experimentally demonstrated with S. suis.85
CLINICAL FINDINGS
Multiple Streptococcus spp. are implicated in lameness and central nervous system signs in piglets and sows.86 There is significant variation of carrier states and clinical signs with the individual serotypes.87
Arthritis and meningitis may occur alone or together and are most common in the 2–6-week age group. More commonly several piglets within a litter are affected. Meningitis is associated with serotypes 1, 2, ½, 3, 4, 8, 9, 14, and 16; septicemia with 2; arthritis with 7 and 14; abscesses with 2; bronchopneumonia with 2, 3, 7, 10, 15, and 27; reproductive damage with 2, 13, and 22; 14 can be associated with any clinical condition.88
The arthritis is characterized by enlarged and distended joint capsules, lameness and pain on palpation of the affected joints. Fever, depression, reluctance to move, and inactivity are common.
Meningitis is characterized by a fever, anorexia, and depression. The gait is stiff, the piglets standing on their toes, and there is swaying of the hindquarters. The ears are often retracted against the head. Blindness and gross muscular tremor develop followed by inability to maintain balance, lateral recumbency, violent paddling, and death. In many cases there is little clinical evidence of omphalophlebitis.
In epidemics of meningitis due to S. suis type 2, sudden death in one or more pigs may be the first sign. Affected pigs found alive are incoordinated and rapidly become recumbent. There is opisthotonos, paddling and convulsions and death in less than 4 hours. A fever of up to 41°C (105°F) is common. In the UK, meningitis of recently weaned pigs is the most striking feature of S. suis type 2 infection.
Otitis interna is a common sequel to many cases of S. suis meningitis.89
Arthritis is common in younger pigs.
In endocarditis and septicemia the piglets are usually found comatose or dead without premonitory signs having been observed.
Valvular endocarditis due to S. suis type 2 has also been reported in a 13-week-old fattening pig in a breeding herd that had a long history of streptococcal meningitis.69
CLINICAL PATHOLOGY
Culture or detection of organism
The organism can be cultured from joint fluid, cerebrospinal fluid, blood, brain, and lungs of pigs at necropsy. The tonsils of live pigs may be swabbed and cultured for the organism. Improved and selective media are available for the isolation and serotyping of the organism.90 An indirect fluorescent antibody test can be used to identify the organism on tonsillar swabs of live pigs. Because of multiple antimicrobial resistance among strains of the organism, drug sensitivity testing on a routine basis is recommended.91 Highly virulent strains of S. suis types 2 and 1 were detected in tonsillar specimens using PCR.92 Rapid serotype-specific PCR assays have been developed.93
Serology
The specific serotype of S. suis should be determined. A simplified laboratory method is available for the identification of S. suis strains associated with different animal hosts or located in different body regions.94 An enzyme-linked immunosorbent assay (ELISA) using monoclonal antibodies directed against virulence markers of S. suis can distinguish between virulent and avirulent strains of the organism.47 A rapid and specific double-sandwich ELISA is available for the detection and capsular typing of the organism with a specificity of 97.6% and sensitivity of 62.5%.95 However, many laboratories are not readily equipped to identify the numerous serotypes of the organism. The biochemical characteristics and serotyping techniques have been described.40
NECROPSY FINDINGS
In pigs dying from S. suis type 2 infection, the gross and microscopic findings include one or more of fibrinopurulent polyserositis, fibrinous polyarthritis, fibrinous or hemorrhagic bronchopneumonia, suppurative meningitis, hemorrhagic necrotizing myocarditis, and vegetative valvular endocarditis.60 Gross myocardial lesions cannot be distinguished from those of mulberry heart disease. In cases with meningitis, there is turbidity of the cerebrospinal fluid, congestion of meningeal vessels, and variable amounts of white exudate in the subarachnoid space. This suppuration is usually most evident along the ventral aspect of the brain. The typical histological picture is one of acute inflammation – neutrophils and fibrin dominate the response. Other changes that may be observed in S. suis infections of the central nervous system include internal hydrocephalus, foci of liquefaction necrosis, subacute (mononuclear cell-rich) meningoencephalitis or meningoencephalomyelitis with bilateral subacute optic perineuritis and Gasserian ganglioneuritis.60 In the tonsils, S. suis organisms can be seen in the subepithelial lymphoid tissue and in the crypt lumen and crypt epithelium.96 S. suis type 9 is more prone to cause bronchopneumonia than the spectrum of lesions typical of type 2.82
Samples for confirmation of diagnosis
• Bacteriology – lung, spleen; culture swabs from serosal surfaces, joints, meninges. Cerebrospinal fluid is the material for the best diagnosis (CULT). Bacterial culture is difficult if the animals have been treated and this is so even when they receive growth-promoting antibiotics.97 Immunomagnetic isolation of types 2 and ½ from swine tonsils has been described and it is better than the standard procedure98
• Histology – formalin-fixed samples of a variety of organs, including lung, brain, heart, liver (LM). Immunohistochemistry and in situ hybridization have been described for use on formalin-fixed tissue and are able to detect single cells.99
(Note the zoonotic potential of this organism when handling carcass and submitting specimens.)
In pigs there may be sporadic cases of arthritis due to staphylococci but the streptococcal infection is the common one. Arthritis due to M. hyorhinis is less suppurative but may require cultural differentiation. Glasser’s disease occurs usually in older pigs and is accompanied by pleurisy, pericarditis, and peritonitis. Erysipelas in very young pigs is usually manifested by septicemia. Nervous disease of piglets may resemble arthritis on cursory examination but there is an absence of joint enlargement and lameness. However, the meningitic form of the streptococcal infection can easily be confused with viral encephalitides. Meningitis in young pigs may also be associated with P. multocida and E. coli. Polyarthritis in calves, lambs, and piglets may also be associated with infection with A. pyogenes and F. necrophorum. S. suis type 2 can also be the cause of meningitis in older pigs of 10–14 weeks of age.
TREATMENT
Antimicrobials
In Denmark over the last 15 years there has been an increase in resistance of S. suis isolates to the two most commonly used antibiotics, tylosin and tetracyclines.100 The strains show a varying pattern of resistance dependent on which of 21 ribotype profiles they belong to.101 For example, strains causing meningitis were more resistant to sulfamethoxazole but those causing pneumonia were more resistant to tetracyclines. Tilmicosin has been used successfully to remove clinical signs of streptococcal meningitis from a herd.102
Penicillin has been the treatment of choice but penicillin-resistant isolates have emerged. Penicillin sensitivity can no longer be assumed for all strains of S. suis and the routine use of penicillin must be re-evaluated.103 In one study, more than 50% of isolates of S. suis were not susceptible to penicillin.91 Penicillin did not eliminate the organism from the tonsils of carrier pigs treated daily for several days.104 In some surveys, the antimicrobial sensitivity of S. suis indicates a high degree of sensitivity to ampicillin, cephalothin, and trimethoprim–sulfamethoxazole, and resistance to the aminoglycosides gentamicin and streptomycin.91 It is recommended that trimethoprim– sulfamethoxazole be used for the treatment of affected pigs and be given daily for 3 days. An occasional strain may be resistant to trimethoprim– sulfamethoxazole.
None of the resistant strains produced beta-lactamase. Conjugation of antibiotic resistance in S. suis has been reported, which may explain the multiple antimicrobial resistance.105 The genes responsible for resistance appear to be homologous to genes found in many other species of bacteria.106 Treatment of pigs affected with meningitis due to S. suis type 2 with either trimethoprim– sulfadiazine or penicillin reduced the case fatality rate from 55% to 21%. Cefquinone has been shown to improve cure rates (67%) compared to ampicillin (55%) and to reduce mortality from 35% with ampicillin to 24% with cefquinone.107
Passive immunization against S. suis type 2 has been described.108
CONTROL
At the present time there are no known specific methods for the prevention of the disease complex associated with S. suis type 2. The recommendations are based on empirical field observations.
It has been suggested that ceftiofur administered by injection for three consecutive days following S. suis challenge was the most effective regimen for minimizing disease associated with PRRS virus and S. suis infection.109
Environment and management
Good management and hygiene techniques should be emphasized. Based on observations of the effects of management practices on S. suis carrier rate in nursery pigs, excessive temperature fluctuations, high relative humidity, crowding, and an age spread exceeding 2 weeks of pigs in the same room were associated with a higher-than-average percentage of carrier pigs.
Nursery pig environmental temperature should not fluctuate more than 1.1–1.7°C over a 24-hour period.34
Excessive relative humidity must be avoided; the recommended range of relative humidity for nursery pigs is 55–70%.
The age spread between pigs in the same room should not exceed 2 weeks. Young, potentially naive piglets raised in the same air space as older animals may be exposed to high concentrations of the organism.
Adequate space to avoid crowding is also necessary. Crowding occurs when less than 0.18 m2 is provided for each 22.7 kg of pig.34 The use of an all-in, all-out production system is recommended, compared to a continuous flow system, which allows for a buildup of pathogens. The control of the most commonly encountered infectious diseases is also important. A well-fortified nutritional program may also aid in the control of S. suis infection and the carrier state in a swine herd.
Segregated early weaning programs have been used in an attempt to control the disease but appear to be unsuccessful in reducing the carrier state.110 Pigs are weaned at an early age and moved to a separate site in an effort to separate the piglets from the sows, which are the primary source of the organism. Carrier pigs readily transmit the infection to uninfected pigs and the main method of spread between herds is the movement of infected breeding stock or weaner pigs. In herds that are free of the infection, it is necessary to avoid the importation of infected pigs. Eradication of S. suis type 2 infection can be attempted by depopulation of suspected carrier sows and replacement with noninfected breeding stock.
Mass medication of feed
Mass medication of individual pigs or medication of the feed during periods of high risk may control the incidence of clinical disease. Outbreaks in sucking piglets have been controlled by a single injection of benethamine penicillin to all piglets given 5 days before the average age of onset of clinical signs. The feeding of oxytetracycline (400 g/tonne) for 14 days immediately prior to the usual onset may control the occurrence of the disease at a low level in weaned pigs. The use of a medicated feed containing trimethoprim– sulfadiazine (1:5) at a rate of 500 g/tonne for the first 6 weeks after weaning did not significantly reduce the incidence of disease. Oral prophylactic medication with either procaine penicillin G or a mixture of chlortetracycline, sulfadimidine and procaine penicillin G reduced the incidence of meningitis. Phenoxymethyl penicillin administered orally provided higher plasma concentrations of drug. The inclusion of phenoxymethyl penicillin potassium (10%) at a rate of 2 kg/tonne of feed significantly reduced the incidence of streptococcal meningitis when fed to the pigs for a total of 6 weeks from 4–10 weeks of age.111
Tiamulin in the drinking water at 180 mg/L of water for 5 days significantly reduced the effects of experimentally induced S. suis type infections.112
Vaccination
Studies are being conducted on the use of vaccines containing the immunogenic polysaccharide from S. suis type 2. Whole-cell vaccines seem to induce significant protection in pigs against a homologous challenge.113 A commercial vaccine reduced mortality from 17% to 2.6%.114 However, the protection provided by whole-cell vaccines is probably type-specific, which suggests that such vaccines should contain many serotypes if broad protection is desired. A trial minimizing variation in weaning age to achieve a uniform size with a combination of an autogenous vaccine and ceftiofur sodium has been reported.115 The protective levels of antibody did not prevent the survival of the organism in either tonsils or joints. An ELISA can be used to evaluate the antibody response in pigs vaccinated with S. suis type 2.116 Different components of the organism are being examined to identify possible fractions for the preparation of a subunit vaccine. A subunit vaccine containing both MRP and EF, formulated with an oil/water adjuvant, protected pigs against challenge with a virulent S. suis type 2.117 Vaccination of sows with 2 mL of bacterin prevented neurological signs but not lameness, bacteriuria, or mortality in their progeny from challenge at 13–21 days of age.118 Immunization of experimental mice with a live avirulent strain of S. suis type 2 provided protection, which may be extrapolated for consideration in pigs.119 A vaccine containing purified suilysin protected mice against a lethal homologous challenge and induced protection against clinical signs in pigs after homologous challenge.120 Pigs vaccinated with a vaccine containing purified suilysin were protected from challenge with the homologous strain of the organism, whereas pigs vaccinated with a vaccine containing most of the extracellular antigens, and the placebo pigs, developed clinical disease.117 Suilysin is produced by most of the field strains tested and could be an important cross-protection factor.56
1 Desrosiers R. Can J Vet Res. 1999;63:83.
2 Torremorell, T., Pijoan, C. Equine: Proceedings of the 15th International Pig Veterinary Society Congress 1998; vol 2:89.
3 Devriese LA. J Appl Bacteriol. 1991;71:478.
4 Higgins R, et al. J Vet Diagn Invest. 1995;7:405.
5 Baele M, et al. Int J Syst Evol Microbiol. 2003;53:143.
6 Vela AI, et al. J Clin Microbiol. 2003;41:2498.
7 Reams RY, et al. J Vet Diagn Invest. 1993;5:363.
8 Higgins R, et al. Can Vet J. 1990;31:529.
9 Hommez J, et al. Vet Rec. 1988;123:626.
10 Devriese LA, et al. Vet Rec. 1990;127:68.
11 Devriese LA. Vet Rec. 1993;132:283.
12 Devriese LA, Haesebrouck F. Vet Rec. 1992;130:380.
13 Heath PJ, et al. Vet Rec. 1995;136:547.
14 Heath PJ, et al. Vet Rec. 1996;139:450.
15 MacLennan M, et al. Vet Rec. 1996;139:423.
16 Mwaniki CG, et al. Aust Vet J. 1994;71:385.
17 Robertson ID, et al. Epidemiol Infect. 1991;107:119.
18 Hill JE, et al. Vet Microbiol. 2005;107:63.
19 Higgins R, Gottschalk M. Aust Vet J. 1997;38:302.
20 Brisebois LM, et al. Can J Vet Res. 1990;54:174.
21 Reams RY, et al. J Vet Diagn Invest. 1996;8:119.
22 Boetner AG, et al. Acta Pathol Microbiol Immunol Scand B. 1987;95:233.
23 Aarestrup FM, et al. Vet Microbiol. 1998;60:59.
24 Schvonen L, et al. Acta Vet Scand. 1988;29:1988.
25 Gogolewski RD, et al. Aust Vet J. 1990;67:202.
26 Griffiths IB, et al. Vet Rec. 1991;128:354.
27 Higgins R, et al. Can Vet J. 1997;38:114.
28 Cloutier G, et al. Vet Microbiol. 2003;97:135.
29 Amass SF, et al. Swine Health Prod. 1996;4:269.
30 Torremorell M, et al. Can J Vet Res. 1998;62:21.
31 Berthelot-Herault F, et al. Vet Microbiol. 2001;82:69.
32 Dee SA, Corey MM. Swine Health Prod. 1993;1:17.
33 Baele M, et al. J Appl Microbiol. 2001;91:997.
34 Dee SA, et al. J Am Vet Med Assoc. 1993;203:295.
35 Thanawongnuwech R, et al. Vet Pathol. 2000;37:143.
36 Feng WH, et al. J Virol. 2001;75:4889.
37 Galina L, et al. Vet Rec. 1994;134:60.
38 Iglesias JG, et al. Am J Vet Res. 1992;53:364.
39 Drum SD, Hoffman J. Swine Health Prod. 1998;6:217.
40 Higgins R, Gottschalk M. J Vet Diagn Invest. 1990;2:249.
41 Smith HE. Infect Immun. 1999;67:1750.
42 Amass SF. Proc Am Assoc Swine Vet. 2000;31:315.
43 Torremorell M, Pijoan C. Vet Rec. 1998;143:395.
44 Quessy S, et al. Infect Immun. 1995;63:1975.
45 Niven DF, et al. Can J Microbiol. 1999;45:1027.
46 Gauna L, et al. Am J Vet Res. 1996;60:72.
47 Vecht U, et al. Vet Microbiol. 1993;34:71.
48 Smith HE, et al. Infect Immun. 1996;64:4409.
49 Jobin M-C, Greiner D. FEMS Microbiol Lett. 2003;220:113.
50 Greeff A, et al. Infect Immun. 2002;70:1319.
51 Fontaine C, et al. Infect Immun. 2004;72:774.
52 Benkirane R, et al. FEMS Immunol Med Microbiol. 1998;20:121.
53 Smith HE. Microbiology. 2001;147:271.
54 Gottschalk M, et al. Can J Vet Res. 1998;62:75.
55 Gottschalk M, et al. Microbiology. 1995;141:189.
56 Jacobs AAC, et al. Vet Rec. 1995;137:295.
57 Gottschalk M. Proc Am Assoc Swine Vet. 2002;33:255.
58 King SJ, et al. Infect Immun. 2001;69:7572.
59 Jacques M, et al. J Bacteriol. 1990;172:2833.
60 Gottschalk M, et al. J Clin Microbiol. 1990;28:2156.
61 Gottschalk M, et al. Can J Vet Res. 1991;55:302.
62 Mwanki CG, et al. Epidemiol Infect. 1994;113:321.
63 Gottschalk M, et al. Vet Microbiol. 1992;30:59.
64 Hampson DJ, et al. J Clin Microbiol. 1993;31:2895.
65 Walsh B, et al. Rev Med Microbiol. 1992;3:65.
66 Donkasul K, et al. Southeast Asian J Trop Med Public Health. 2003;34:154.
67 Strangmann E, et al. Int J Hyg Environ Health. 2002;205:385.
68 Trottier S, et al. Rev Infect Dis. 1991;13:1251.
69 Maher D. J Infect Dis. 1991;22:303.
70 Robertson ID, Blackmore DK. Epidemiol Infect. 1989;203:157.
71 Bengal L, et al. Cell Microbiol. 2004;6:867.
72 Salles M, et al. Vet Immunol Immunopathol. 2002;87:51.
73 Gottschalk M, Segura M. Vet Microbiol. 2000;76:259.
74 Charland N, et al. Infect Immun. 2000;68:637.
75 Segura M, et al. Infect Immun. 1999;67:4646.
76 Smith HE, et al. Infect Immun. 1999;67:1750.
77 Charland N. Microbiology. 1998;144:325.
78 Lalonde M, et al. Microbiology. 2000;146:1913.
79 Staats JJ, et al. Vet Microbiol. 1999;70:201.
80 Williams AE, Blackmore WF. J Infect Dis. 1990;162:474.
81 Reams RY, et al. J Vet Diagn Invest. 1994;6:326.
82 Vasconcelos D, et al. J Vet Diagn Invest. 1994;6:335.
83 Robertson ID, Blackmore DK. Epidemiol Infect. 1990;105:469. 479
84 Reams RY, et al. J Vet Diagn Invest. 1995;7:406.
85 Thanawongnuwech R, et al. Vet Pathol. 2000;37:143.
86 Fangman TJ, Fales WH. Swine Health Prod. 1999;7:113.
87 Wisselink HJ, et al. J Clin Microbiol. 2002;40:2922.
88 Luque I, et al. Vet Rec. 1998;142:726.
89 Madsen LW, et al. Vet Pathol. 2001;38:190.
90 Kataoka Y, et al. Vet Microbiol. 1991;28:335.
91 Turgeon PL, et al. Br Vet J. 1994;150:263.
92 Wisselink HJ, et al. Vet Microbiol. 1999;67:143.
93 Smith HE, et al. J Clin Microbiol. 1999;37:3146.
94 Devriese LA, et al. Vet Microbiol. 1991;26:141.
95 Serhir B, et al. Can J Vet Res. 1993;57:19.
96 Madsen LW, et al. J Comp Pathol. 2002;126:57.
97 Done SH, et al. Pig J. 1998;41:113.
98 Gottschalk M, et al. J Clin Microbiol. 1999;28:2462.
99 Boye M, et al. J Vet Diagn Invest. 2000;12:224.
100 Aarestrup FM, et al. Vet Microbiol. 1998;63:71.
101 Rasmussen SR, et al. J Clin Microbiol. 1999;37:404.
102 Strachan WD, et al. Pig J. 2003;51:177.
103 Gottschalk LM, et al. J Vet Diagn Invest. 1991;3:170.
104 Amass SF, et al. J Vet Diagn Invest. 1996;8:64.
105 Stuart JG, et al. Vet Microbiol. 1992;30:213.
106 Wateson Y, et al. Vet Microbiol. 1994;41:41.
107 Thomas E, et al. Pig J. 2004;50:28.
108 Andresen LO, Teetmeier C. Vet Microbiol. 2001;81:331.
109 Halbur PG, et al. J Clin Microbiol. 2000;38:1156.
110 Amass SF, et al. Swine Health Prod. 1995;3:189.
111 Johnston PI, et al. Vet Rec. 1991;130:138.
112 Chengappa MM, et al. J Am Vet Med Assoc. 1990;197:1467.
113 Holt ME, et al. Res Vet Sci. 1990;48:23.
114 Torremorell M, et al. Swine Health Prod. 1997;5:139.
115 Villani D. J Swine Health Prod. 2003;11:27.
116 Blouin C, et al. Can J Vet Res. 1994;58:49.
117 Wisselink HJ, et al. Vet Rec. 2001;148:473.
118 Amass SF, et al. Swine Health Prod. 2000;8:217.