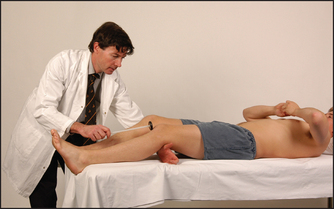
The neurological examination: the peripheral nervous system
Limbs and trunk
HISTORY
The patient may present with symptoms that are purely or predominately sensory or motor (see Questions box 34.1), or related to disorders of movement such as tremor. Sensory symptoms include pain, numbness and paraesthesias (tingling or pins and needles). It is important to find out if there is involvement of more than one modality, something the patient may not have noticed. The distribution, time of onset and duration may give clues to the aetiology of the symptoms or at least as to where the sensory examination should be concentrated.
A family history of a similar problem may help provide the diagnosis in conditions such as muscular dystrophy. A previous injury may be responsible, for example, for a peripheral nerve problem but not remembered until asked about specifically.
EXAMINATION ANATOMY
Muscle weakness has four major causes:
1. Pyramidal or upper motor neurone weakness, which is caused by a lesion in the brain proximal to the ‘pyramids’ in the brainstem. This is where the nerve fibres decussate or cross to the other side before travelling down the spinal cord (see Figure 34.1).
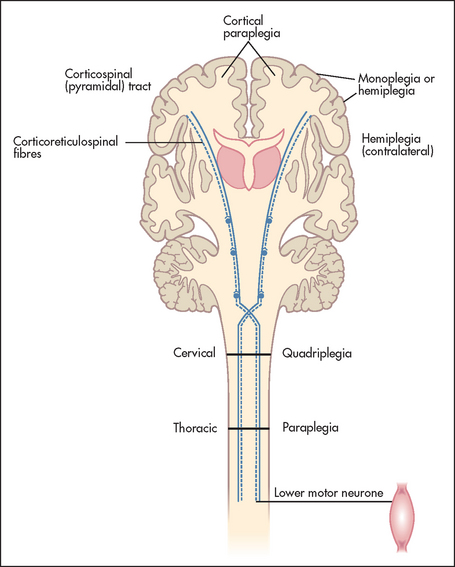
Figure 34.1 Motor neurone lesions (Adapted from Lance JG, McLeod JW. A physiological approach to clinical neurology, 3rd edn. London: Butterworths, 1981.) Butterworths
2. Lower motor neurone weakness, which is caused by a nerve lesion within the spinal cord or peripheral nerve.
3. Abnormalities of the neuromuscular junction (myasthenia gravis).
GENERAL EXAMINATION APPROACH
It is most important to have a set order of examination of the limbs for neurological signs so that nothing important is omitted. The following scheme is a standard approach.
GENERAL INSPECTION
Remember to look for asymmetry.
1. Stand back and look at the patient for an abnormal posture—for example, one due to hemiplegia caused by a stroke. In this case, the upper limb is flexed and there is adduction and pronation of the arm, while the lower limb is extended.
2. Look for muscle wasting, which indicates a denervated muscle, a primary muscle disease or disuse atrophy. Compare one side with the other for wasting and try to work out which muscle groups are involved (proximal, distal or generalised, symmetrical or asymmetrical).
3. Inspect for abnormal movements, such as tremor of the wrist or arm.
4. Inspect the skin—for example, for evidence of neurofibromatosis, cutaneous angiomata in a segmental distribution (associated with syringomyelia) or herpes zoster. Look for scars from old injuries or surgical treatment. Note the presence of a urinary catheter.
UPPER LIMBS
Motor systema
General
Shake hands with the patient and introduce yourself. A patient who cannot relax his or her hand grip has myotonia (an inability to relax the muscles after voluntary contraction). The most common cause of this is the muscle disease dystrophia myotonica (page 470). Once your hand has been extracted from the patient’s, and after pausing briefly for the vitally important general inspection, ask the patient to undress so that the arms and shoulder girdles are completely exposed.
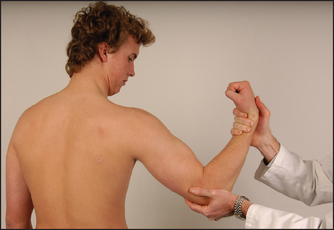
Sit the patient over the edge of the bed if this is possible. Next ask the patient to hold out both hands, palms upwards, with the arms extended and the eyes closed (see Figure 34.2). Watch the arms for evidence of drifting (movement of one or both arms from the initial neutral position). There are only three causes of arm drift:

Figure 34.2 Testing for arm drift: ‘Shut your eyes and hold your arms out straight. Now turn your palms upwards.’ (Courtesy of Glenn McCulloch)
1. Upper motor neurone (pyramidal) weakness. The drift of the affected limb(s) here is due to muscle weakness and tends to be in a downward direction. The drifting typically starts distally with the fingers and spreads proximally. There may be slow pronation of the wrist and flexion of the fingers and elbow.
2. Cerebellar disease. The drift here is usually upwards. It also includes slow pronation of the wrist and elbow.
3. Loss of proprioception. The drift here (pseudoathetosis) is really a searching movement and usually affects only the fingers. It is due to loss of joint position sense and can be in any direction.
Fasciculations
Ask the patient to relax the arms and rest them on his or her lap. Inspect the large muscle groups for fasciculations (see List 34.1). These are irregular contractions of small areas of muscle that have no rhythmical pattern. Fasciculation may be coarse or fine and is present at rest, but not during voluntary movement.b If present with weakness and wasting, fasciculation indicates degeneration of the lower motor neurone. It is usually benign if unassociated with other signs of a motor lesion.
Tone
Tone is tested at both the wrists and the elbows. Rotation of the wrists with supination and pronation of the elbow joints (supporting the patient’s elbow with one hand and holding the hand with the other) is performed passively (the examiner does the work), and the patient should be told to relax to allow you to move the joints freely.
When you raise and then drop the patient’s arm, it will fall suddenly if tone is reduced. With experience it is possible to decide if tone is normal or increased (hypertonic, as in an upper motor neurone or extrapyramidal lesion). Hypotonia is a difficult clinical sign to elicit and probably not helpful in the assessment of a lower motor neurone lesion. Most elderly people find it difficult not to try to help you and to relax their muscles completely. This leads to an increase in tone in all directions of movement, which increases with the speed of movement and with encouragement to relax. This is called gegenhaltec or paratonia. When it is severe it may be a result of frontal lobe or diffuse cerebrovascular disease. If the joints are moved unpredictably and at different rates or if the patient is distracted (e.g. by being asked to count backwards from 100), it may be reduced. Young people who are able to relax their muscle completely have little or no tone and hypotonia cannot be diagnosed in these people.
The cogwheel rigidity of Parkinson’s disease is another abnormality of tone in the upper limbs. It is best assessed by having the patient move the other arm up and down as you move the hand and forearm, testing tone at the wrist and elbow.
Myotonia as described above is also an abnormality of tone that is worse after active movement. In these patients, tone is usually normal at rest but after sudden movements there may be a great increase in tone and the patient is unable to relax the muscle. Tapping over the body of a myotonic muscle causes a dimple of contraction, which only slowly disappears (percussion myotonia). This is best tested by tapping the thenar eminence or by asking the patient to make a tight fist and then open the hand quickly. The opening of the fist is very slow when the muscles are myotonic.
Power
Muscle strength is assessed by gauging your ability to overcome the patient’s full voluntary muscle resistance. To decide whether the power is normal, the patient’s age, sex and build should be taken into account. Power is graded based on the maximum observed (no matter how briefly), according to the following modified British Medical Research Council scheme (although this lacks sensitivity at the higher grades because work against gravity may make up only a small component of a muscle’s function [e.g. the finger flexors]):
0 Complete paralysis (no movement).
1 Flicker of contraction possible.
2 Movement is possible when gravity is excluded.
3 Movement is possible against gravity but not if any further resistance is added.
4–. Slight movement against resistance.
4 Moderate movement against resistance.
5 Normal power.d
If power is reduced, decide whether this is symmetrical or asymmetrical, whether it involves only particular muscle groups or whether it is proximal, distal or general. Sometimes painful joint or muscle disease may interfere with the assessment (see Chapter 23). Asymmetrical muscle weakness is most often the result of a peripheral nerve, brachial plexus or root lesion or an upper motor neurone lesion. As each movement is tested, the important muscles involved should be observed or palpated.
Shoulder
• Abduction—mostly deltoid and supraspinatus—(C5, C6): the patient should abduct the arms with the elbows flexed and resist your attempt to push them down (see Figure 34.3).
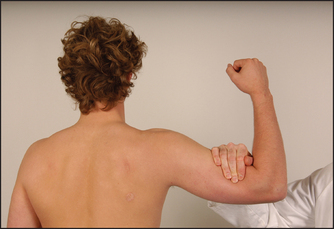
Figure 34.3 Testing power—shoulder abduction: ‘Stop me pushing your arm down’ (Courtesy of Glenn McCulloch)
• Adduction—mostly pectoralis major and latissimus dorsi—(C6, C7, C8): the patient should adduct the arms with the elbows flexed and not allow you to separate them.
Elbow
• Flexion—biceps and brachialis—(C5, C6): the patient should bend the elbow and pull so as not to let you straighten it out (see Figure 34.4).
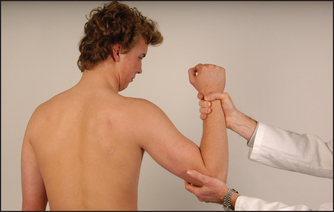
Figure 34.4 Testing power—elbow flexion: ‘Stop me straightening your elbow’ (Courtesy of Glenn McCulloch)
• Extension—triceps brachii—(C7, C8): the patient should bend the elbow and push so as not to let you bend it (see Figure 34.5).
Wrist
• Flexion—flexor carpi ulnaris and radialis—(C6, C7): the patient should bend the wrist and not allow you to straighten it.
• Extension—extensor carpi group—(C7, C8): the patient should extend the wrist and not allow you to bend it (see Figure 34.6).
Fingers
• Extension—extensor digitorum communis, extensor indicis and extensor digiti minimi (C7, C8): the patient should straighten the fingers and not allow the examiner to push them down (push with the side of your hand across the patient’s metacarpophalangeal joints).
• Flexion—flexor digitorum profundus and sublimis—(C7, C8): the patient squeezes two of your fingers (see Figure 34.7).

Figure 34.7 Testing power—finger flexion: ‘Squeeze my fingers hard’ (don’t offer more than two fingers) (Courtesy of Glenn McCulloch)
• Abduction—dorsal interossei—(C8, T1): the patient should spread out the fingers and not allow you to push them together (see Figure 34.8).
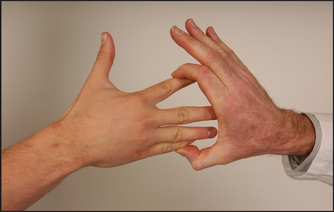
Figure 34.8 Testing power—finger abduction: ‘Stop me pushing your fingers together’ (Courtesy of Glenn McCulloch)
• Adduction—volar interossei—(C8, T1): the patient holds the fingers together and tries to prevent you from separating them further.
Reflexes
The sudden stretching of a muscle usually evokes brisk contraction of that muscle or muscle group. This reflex is usually mediated via a neural pathway synapsing in the spinal cord. It is subject to regulation via pathways from the brain. As the reflex is a response to stretching of a muscle, it is correctly called a muscle stretch reflex rather than a tendon reflex. The tendon merely transmits stretch to the muscle.
Tendon hammers are available in a number of designs. Sir William Gowerse used the ulnar side of his hand or part of his stethoscope. In Australia and the United Kingdom, the Queen Square hammerf is in common use (see Figure 34.9). The Taylor hammer is popular in the United States; it is shaped like a tomahawk and has a broad rubber edge for most tendons and a more pointed side for the cutaneous reflexes.
Reflexes are graded from absent to greatly increased (see Table 34.1).
TABLE 34.1
Classification of muscle stretch reflexes
| 0 | Absent |
| + | Present but reduced |
| ++ | Normal |
| +++ | Increased, possibly normal |
| ++++ | Greatly increased, often associated with clonus |
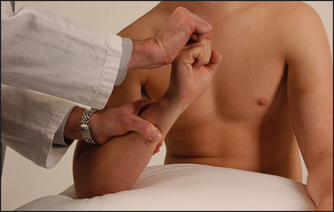
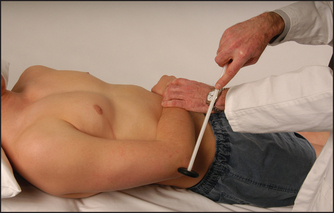
Make sure the patient is resting comfortably with the elbows flexed and hands lying pronated on the lap and not overlapping one another. To test the biceps jerk (C5, C6), place one fore-finger on the biceps tendon and tap this with the tendon hammer (see Figure 34.10). The hammer should be held near its end and the head allowed to fall with gravity onto the positioned forefinger. You will soon learn not to hit too hard. Normally, if the reflex arc is intact, there is a brisk contraction of the biceps muscle with flexion of the forearm at the elbow, followed by prompt relaxation. Practice will help you decide whether the response is within the normal range. When a reflex is greatly exaggerated, it can be elicited away from the usual zone.
If a reflex appears to be absent, always test following a reinforcement manoeuvre. For example, ask the patient to clench the teeth tightly just before you let the hammer fall. Supraspinal and fusiform mechanisms have been identified to explain reinforcement, but it works partly as a distraction, especially if the reflex is absent, because an anxious patient has contracted opposing muscle groups. Merely talking to the patient may provide enough distraction for the reflex to be elicited. Sometimes normal reflexes can be elicited only after reinforcement, but they should still be symmetrical.
An increased jerk occurs with an upper motor neurone lesion (page 462). A decreased or absent reflex occurs with a breach in any part of the reflex motor arc—the muscle itself (e.g. myopathy), the motor nerve (e.g. neuropathy), the anterior spinal cord root (e.g. spondylosis), the anterior horn cell (e.g. poliomyelitis) or the sensory arc (sensory root or sensory nerve).
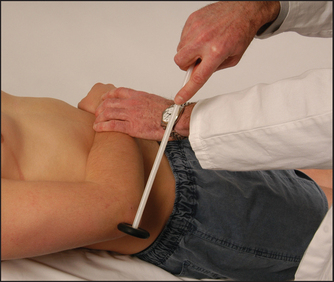
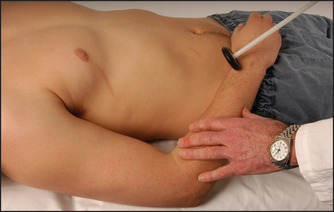
To test the triceps jerk (C7, C8), support the elbow with one hand and tap over the triceps tendon (see Figure 34.11). Normally, triceps contraction results in forearm extension.
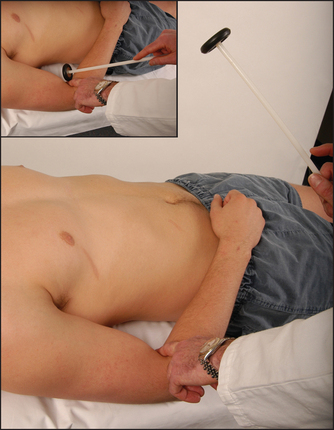
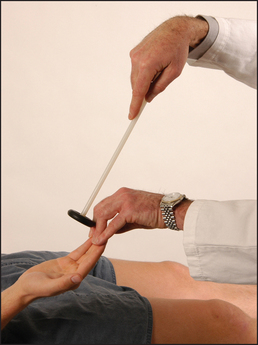
To test the brachioradialis (supinator) jerk (C5, C6), strike the lower end of the radius just above the wrist (see Figure 34.12). To avoid hurting the patient by striking the radial nerve directly, place your own first two fingers over this spot and then strike your fingers, as with the biceps jerk. Normally, contraction of the brachioradialis causes flexion of the elbow.
If elbow extension and finger flexion are the only response when the patient’s wrist is tapped, the response is said to be inverted, known as the inverted brachioradialis (supinator) jerk. The triceps contraction causes elbow extension instead of the usual elbow flexion. This is associated with an absent biceps jerk and an exaggerated triceps jerk. It indicates a spinal cord lesion at the C5 or C6 level due, for example, to compression (e.g. disc prolapse), trauma or syringomyelia. It occurs because a lower motor neurone lesion at C5 or C6 is combined with an upper motor neurone lesion affecting the reflexes below this level.
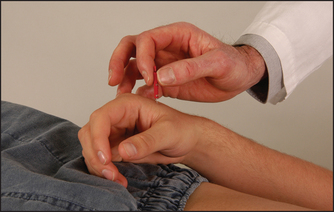
To test finger jerks (C8), the patient rests the hand palm upwards, with the fingers slightly flexed. Place your hand over the patient’s and strike the hammer over your fingers (see Figure 34.13). Normally, slight flexion of all the patient’s fingers occurs.
Coordination
The cerebellum has multiple connections (afferent and efferent) to sensory pathways, brainstem nuclei, the thalamus and the cerebral cortex. Via these connections the cerebellum plays an integral role in coordinating voluntary movement. A standard series of simple tests is used to test coordination. Always demonstrate these movements for the patient’s benefit.
Finger–nose test
Ask the patient to touch his or her nose with the index finger and then turn the finger around and touch your outstretched forefinger at nearly full extension of the shoulder and elbow (see Figure 34.14). The test should be done at first slowly and then briskly, and repeated a number of times with the patient’s eyes open and later closed. Slight resistance to the patient’s movements by you pushing on his or her forearm during the test may unmask less-severe abnormalities.
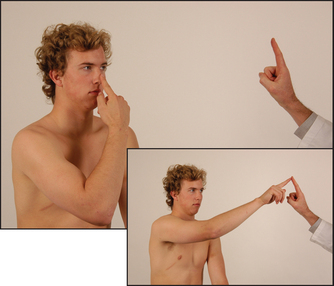
Figure 34.14 Finger–nose test: ‘Touch your nose with your forefinger and then reach out and touch my finger’ (Courtesy of Glenn McCulloch)
Look for the following abnormalities: (1) intention tremor, which is tremor increasing as the target is approached (there is no tremor at rest); and (2) past-pointing, where the patient’s finger overshoots the target towards the side of cerebellar abnormality. These abnormalities occur with cerebellar disease.
Rapidly alternating movements
Ask the patient to pronate and supinate his or her hand on the dorsum of the other hand as rapidly as possible (see Figure 34.15). This movement is slow and clumsy in cerebellar disease and is called dysdiadochokinesis.g
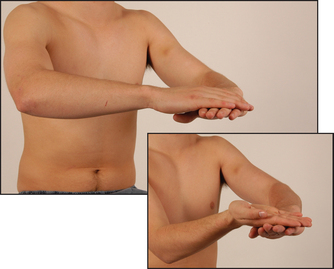
Figure 34.15 Testing for dysdiadochokinesis in the upper limbs: ‘Turn your hand over, backwards and forwards on the other one, as quickly and smoothly as you can’ (Courtesy of Glenn McCulloch)
Rapidly alternating movements may also be affected in extrapyramidal disorders (e.g. Parkinson’s disease) and in pyramidal disorders (e.g. internal capsule infarction).
Rebound
Ask the patient to lift the arms rapidly from the sides and then stop. Hypotonia due to cerebellar disease causes delay in stopping the arms. This method of demonstrating rebound is preferable to the more often used one where the patient flexes the arm at the elbow against the examiner’s resistance. When the examiner suddenly lets go, violent flexion of the arm may occur and, unless prevented, the patient can strike himself or herself in the face. Therefore, only medical students trained in self-defence should use this method.h
Muscle weakness may also cause clumsiness, but motor testing should have revealed any impairment of this sort.
The sensory system
To test sensation, which can be difficult and frustratingly time-consuming, use the following routine.1
Spinothalamic pathway (pain and temperature)
Pain and temperature fibres enter the spinal cord and cross, a few segments higher, to the opposite spinothalamic tract (see Figure 34.16). This tract ascends to the brainstem.
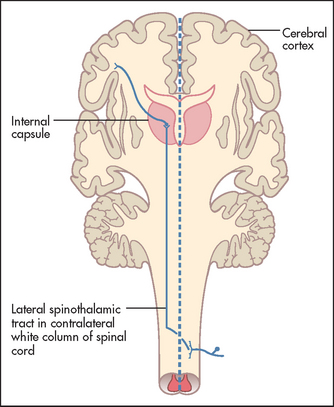
Figure 34.16 Pain and temperature pathways (Adapted from Snell RS, Westmoreland BF. Clinical neuroanatomy for medical students, 4th edn. Boston: Little Brown, 1997.) Little Brown
Pain (pin-prick) testing
Using a new pin,2 demonstrate to the patient that this induces a relatively sharp sensation by touching lightly a normal area, such as the anterior chest wall. Then ask the patient to say whether the pin-prick feels sharp or dull. Begin proximally on the upper arm and test in each dermatome—the area of skin supplied by a vertebral spinal segment (see Figure 34.17). Also compare right with left in the same dermatome. Map out the extent of any area of dullness. Always do this by going from the area of dullness to the area of normal sensation.
Temperature testing
This can be done in a similar fashion, using test tubes filled with hot (40–45°C) and cold (5–10°C) water. Cold sensation can also be tested with a metal object, such as a tuning fork. Absence of ability to feel heat is almost always associated with inability to feel cold. Temperature differences of 2–5° can usually be distinguished. These tests are performed only in special circumstances—for example, for suspected syringomyelia.i)
Posterior columns (vibration and proprioception
These fibres enter and ascend ipsilaterally in the posterior columns of the spinal cord to the nucleus gracilis and nucleus cuneatus in the medulla, where they decussate (see Figure 34.18).
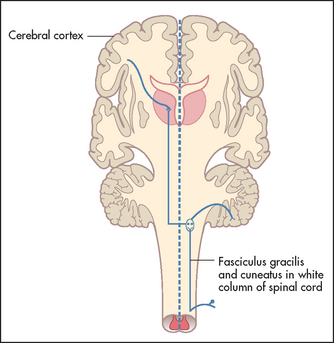
Figure 34.18 Vibration and joint position sense pathways (Adapted from Snell RS, Westmoreland BF. Clinical neuroanatomy for medical students, 4th edn. Boston: Little Brown, 1997.) Little Brown
Vibration testing
Use a 128 Hz tuning fork (not a 256 Hz fork). Ask the patient to close the eyes, and place the vibrating tuning fork on one of the distal interphalangeal joints. The patient should be able to describe a feeling of vibration. Then deaden the tuning fork with your hand; the patient should be able to say exactly when this occurs. Compare one side with the other. If vibration sense is reduced or absent, test over the ulnar head at the wrist, then the elbows (over the olecranon) and then the shoulders to determine the level of abnormality. Although the tuning fork is traditionally placed only over bony prominences, vibration sense is just as good over soft tissues.
Proprioception testing
Use the distal interphalangeal joint of the patient’s little finger. When the patient has his or her eyes open, grasp the distal phalanx from the sides and move it up and down to demonstrate these positions. Then ask the patient to close the eyes while these manoeuvres are repeated randomly. Normally, movement through even a few degrees is detectable, and should be reported correctly. If there is an abnormality, proceed to test the wrists and elbows similarly. As a rule, sense of position is lost before sense of movement, and the little finger is affected before the thumb.
Light-touch testing
Some fibres travel in the posterior columns (i.e. ipsilaterally) and the rest cross the middle line to travel in the anterior spinothalamic tract (i.e. contralaterally). For this reason, light touch is of the least discriminating value. Irritation of light-touch receptors is probably responsible for paraesthesias—for example, following ischaemia of a limb.
Test light touch by touching the skin with a wisp of cottonwool. Ask the patient to shut the eyes and say ‘yes’ when the touch is felt. Do not stroke the skin because this moves hair fibres. Test each dermatome,j comparing left and right sides.
Interpretation of sensory abnormalities
Try to fit the distribution of any sensory loss into a dermatome (due to a spinal cord or nerve root lesion), a single peripheral nerve territory, a peripheral neuropathy pattern (glove distribution, page 464) or a hemisensory loss (due to spinal cord or upper brainstem or thalamic lesion).
Sensory dermatomes of the upper limb (see Figures 34.19 and 34.20) can be recognised by memorising the following rough guide:
Figure 34.19 C8 nerve. Transverse sonograms of the right C5–C8 ventral rami demonstrating the measuring calipers of the ellipse function at the locations used to measure the cross-sectional area of each ventral ramus as it exits adjacent to the transverse processes. (Haun DW. Ultrasound. Medicine & Biology 2010; 36(0): 1422–1430.)
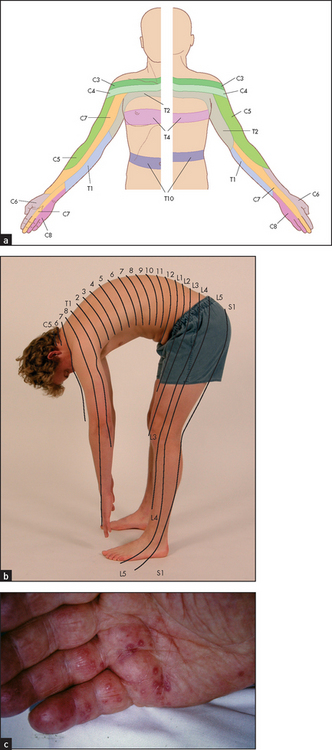
Figure 34.20 Dermatomes of the upper limb and trunk (a) The dermatomes explained. (b) The distribution of the dermatomes makes more sense if we are thought of as quadrupedal. (c) Herpes zoster of the C8 dermatome showing its distribution. ((b) Courtesy of Glenn McCulloch; (c) Courtesy of Dr A Watson, Infectious Diseases Department, The Canberra Hospital)
Examination of the peripheral nerves of the upper limb
A lesion of a peripheral nerve causes a characteristic motor and sensory loss.3 Peripheral nerve lesions may have local causes, such as trauma or compression, or may be part of a mononeuritis multiplex, where more than one nerve is affected by systemic disease.
The radial nerve (C5–C8)
This is the motor nerve supplying the triceps and brachioradialis and the extensor muscles of the hand. The characteristic deformity that results from radial nerve injury is wrist drop. To demonstrate this, if it is not already obvious, get the patient to flex the elbow, pronate the forearm and extend the wrist and fingers. If a lesion occurs above the upper third of the upper arm, the triceps muscle is also affected. Therefore, test elbow extension, which will be absent if the lesion is high.
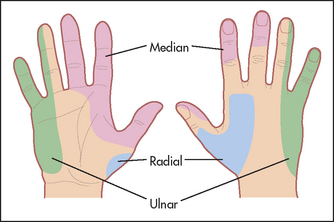
Test sensation using a pin over the area of the anatomical snuff box. Sensation here is lost with a radial nerve lesion before the bifurcation into posterior interosseous and superficial radial nerves at the elbow (see Figure 34.21).
The median nerve (C6–T1)
This nerve contains the motor supply to all the muscles on the front of the forearm except the flexor carpi ulnaris and the ulnar half of the flexor digitorum profundus. It also supplies the following short muscles of the hand—LOAF:
Lesion at the wrist (carpal tunnel)4,5
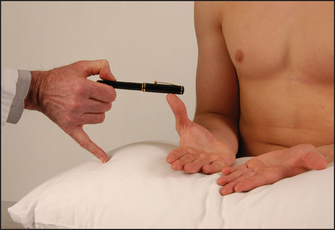
Use the pen-touching test to assess for weakness of the abductor pollicis brevis. Ask the patient to lay the hand flat, palm upwards on the table, and attempt to abduct the thumb vertically to touch your pen held above it (see Figure 34.22). This may be impossible if there is a median nerve palsy at the wrist or above. Remember, however, that most patients with the carpal tunnel syndrome have normal power and may indeed have symptoms but no signs at all.
Lesion in the cubital fossa
Ochsner’s clasping testk (for loss of flexor digitorum sublimis): ask the patient to clasp the hands firmly together (see Figure 34.23(a))—the index finger on the affected side fails to flex with a lesion in the cubital fossa or higher (see Figure 34.23(b)).
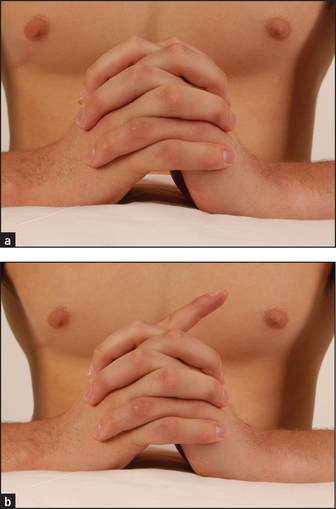
Figure 34.23 Ochsner’s clasping test (a) Normal; (b) abnormal due to loss of function of the flexor digitorum (simulated demonstration). (Courtesy of Glenn McCulloch)
For the sensory component of the median nerve, test pin-prick sensation over the hand. The constant area of loss includes the palmar aspect of the thumb, the index finger, the middle finger and the lateral half of the ring finger (see Figure 34.21). The palm is spared in median nerve lesions in the carpal tunnel.
The ulnar nerve (C8–T1)
This nerve contains the motor supply to all the small muscles of the hand (except the LOAF muscles), the flexor carpi ulnaris and the ulnar half of the flexor digitorum profundus. Look for wasting of the small muscles of the hand and for partial clawing of the little and ring fingers (a claw-like hand). Clawing is hyperextension at the metacarpophalangeal joints and flexion of the interphalangeal joints. Note that clawing is more pronounced with an ulnar nerve lesion at the wrist, as a lesion at or above the elbow also causes loss of the flexor digitorum profundus, and therefore less flexion of the interphalangeal joints. This is the ‘ulnar nerve paradox’, in that a more distal lesion causes greater deformity.
Froment’sl sign
Ask the patient to grasp a piece of paper between the thumb and lateral aspect of the forefinger with each hand. The affected thumb will flex because of loss of the adductor of the thumb.
Causes of a true claw hand are shown in List 34.2, while causes of wasting of the small muscles of the hand are shown in List 34.3; see also Figure 34.24.
For the sensory component of the ulnar nerve, test for pin-prick loss over the palmar and dorsal aspects of the little finger and the medial half of the ring finger (see Figure 34.21).
The brachial plexus
Brachial plexus lesions vary from mild to complete; motor and/or sensory fibres may be involved. Nerve roots form trunks, which divide into cords and then form peripheral nerves (see Tables 34.2 and 34.3). The anatomy is shown in Figure 34.25.
TABLE 34.2
Nerve roots and brachial plexus trunks
| Nerve roots | Trunks | Muscles supplied |
| C5 and 6 | Upper | Shoulder (especially biceps and deltoid) |
| C7 | Middle | Triceps and some forearm muscles |
| C8 and T1 | Lower | Hand and some forearm muscles |
TABLE 34.3
Brachial plexus cords, nerves and their supplied muscles
| Cords | Nerves formed | supplied |
| Lateral | Musculocutaneous, median | Biceps, pronator teres, flexor carpi radialis |
| Medial | Median and ulnar | Hand muscles |
| Posterior | Axillary and radial | Deltoid, triceps and forearm extensors |
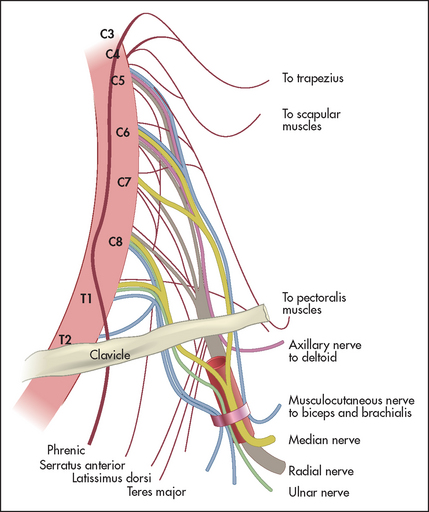
Figure 34.25 The brachial plexus (Adapted from Chusid JG. Correlative neuroanatomy and functional neurology, 19th edn. Los Altos: Lange Medical, 1985.)
Patients with brachial plexus lesions may complain of pain or weakness in the shoulders or arms. Pain is often prominent, especially when there has been nerve root avulsion. A neurological cause is more likely if there is dull pain that is difficult to localise, if the pain is not related to limb movement and is worse at night and if there is no associated tenderness. The patient may be unable to get comfortable. An orthopaedic or traumatic cause is more likely if the pain is much worse with movement, or there are signs of inflammation, joint deformity or local tenderness. Most plexus lesions are supraclavicular (i.e. proximal), especially when they occur after trauma. When infraclavicular (i.e. distal) lesions occur, they are usually less severe.
Examine the arms and shoulder girdle (see List 34.4). Remember that the dorsal scapular nerve (which supplies the rhomboid muscles) comes from the C5 nerve root proximal to the upper trunk, and so rhomboid function is usually spared in upper trunk lesions. Typical lesions of the brachial plexus are described in List 34.5. The cervical rib syndrome may cause a lower brachial plexus lesion (see List 34.6).Table 34.4 suggests a scheme for distinguishing between plexus and nerve root lesions.
TABLE 34.4
Distinguishing between brachial plexus lesions and nerve root compression
| Plexus | Root | |
| Previous trauma | Some types | Occasionally |
| Insidious onset | Some types | Usually |
| Neck pain | No | Yes |
| Unilateral interscapular pain | No | Yes |
| Weakness | Often severe | Mild-moderate |
| Pattern of weakness | Usually shoulder and biceps or hand | Most commonly triceps (C7 lesions, the most commonly affected root) |
Ask the patient to stand facing away from you with the arms and hands stretched out to touch and push against the wall. Winging of the scapulae is seen typically in fascio-scapular-humeral dystrophy (see Figure 34.26).
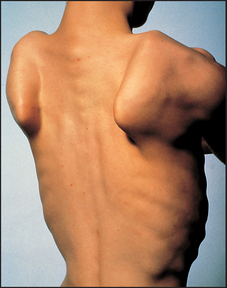
Figure 34.26 Winging of the scapulae, often a result of muscular dystrophy (From Mir MA. Atlas of clinical diagnosis, 2nd edn. Edinburgh: Saunders, 2003, with permission.)
Causes of brachial plexus lesions include:
• inflammation, autoimmune disorders (more often upper plexus)
• radiotherapy (more often upper plexus)
• cancer (more often lower plexus)—cancer causes a brachial plexus lesion by local invasion; the lower trunk is usually affected first. These plexus lesions are usually painful and progress rapidly. Weakness and sensory loss are both present.
• trauma: direct (motor vehicle crash, surgery including sternotomy, lacerations and gunshots), traction (birth injuries, motor vehicle crashes, sporting injuries such as rugby tackles—more often upper plexus), chronic compression (thoracic outlet, ‘backpack palsy’, fractures with bone displacement).
LOWER LIMBS
Begin by testing gait, if this is possible (see page 458).
Inspect the legs with the patient lying in bed with the legs and thighs entirely exposed (place a towel over the groin). Note whether there is a urinary catheter present, which may indicate that there is spinal cord compression or other spinal cord disease, particularly multiple sclerosis.
The motor system
Fasciculations and muscle wasting
Inspect for fasciculations. Look for muscle wasting. Feel the muscle bulk of the quadriceps and calves. Then run a hand along each shin, feeling for wasting of the anterior tibial muscles.
Tone
Test tone at the knees and ankles. Place one hand under a chosen knee and then abruptly pull the knee upwards, causing flexion. When the patient is relaxed this should occur without resistance. Then, supporting the thigh, flex and extend the knee at increasing velocity, feeling for resistance to muscle stretch (tone). Tone in the legs may also be tested by sitting the patient with legs hanging freely over the edge of the bed. Raise one of the patient’s legs to the horizontal and then suddenly let go. The leg will oscillate up to half a dozen times in a healthy person who is completely relaxed. If hypotonia is present, as occurs in cerebellar disease, the oscillations will be wider and more prolonged. If increased tone or spasticity is present, the movements will be irregular and jerky.
Next test for clonus of the ankle and knee. This is a sustained rhythmical contraction of the muscles when put under sudden stretch. It is due to hypertonia from an upper motor neurone lesion. It represents an increase in reflex excitability (from increased alpha motor neurone activity).
Sharply dorsiflex the foot with the knee bent and the thigh externally rotated. When ankle clonus is present, recurrent ankle plantar flexion movement occurs. This may persist for as long as you sustain dorsiflexion of the ankle. Test for patellar clonus by resting a hand on the lower part of the quadriceps with the knee extended and moving the patella down sharply. Sustained rhythmical contraction of the quadriceps occurs as long as the downward stretch is maintained.
Power
Hip
• Flexion—psoas and iliacus muscles—(L2, L3): ask the patient to lift up the straight leg and not let you push it down (having placed your hand above the knee; see Figure 34.27).
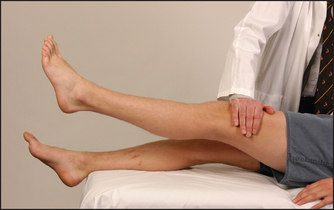
Figure 34.27 Testing power—hip flexion: ‘Lift your leg up and don’t let me push it down’ (Courtesy of Glenn McCulloch)
• Extension—gluteus maximus—(L5, S1, S2): ask the patient to keep the leg down and not let you pull it up from underneath the calf or ankle (see Figure 34.28).
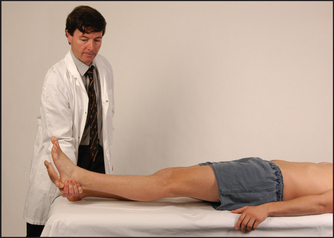
Figure 34.28 Testing power—hip extension: ‘Push your heel down and don’t let me pull it up’ (Courtesy of Glenn McCulloch)
• Abduction—gluteus medius and minimus, sartorius and tensor fasciae latae—(L4, L5, S1): ask the patient to abduct the leg and not let you push it in (see Figure 34.29).
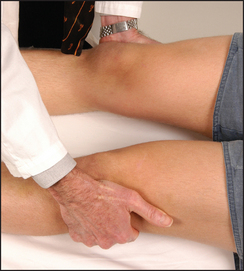
Figure 34.29 Testing power—hip abduction: ‘Don’t let me push your knees together’ (Courtesy of Glenn McCulloch)
• Adduction—adductors longus, brevis and magnus—(L2, L3, L4): ask the patient to keep the leg adducted and not let you push it out (see Figure 34.30).
Knee
• Flexion—hamstrings (biceps femoris, semimembranosus, semitendinosus)—(L5, S1): ask the patient to bend the knee and not let you straighten it (see Figure 34.31). If there is doubt about the real strength of knee flexion, it should be tested with the patient in the prone position. Here possible help from hip flexion is prevented and the muscles can be palpated during contraction.
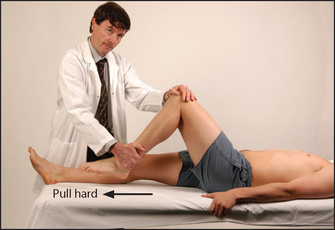
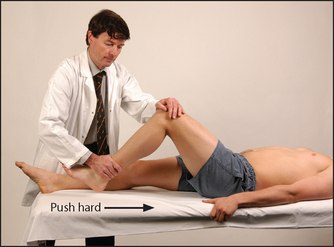
Figure 34.31 Testing power—knee flexion: ‘Bend your knee and don’t let me straighten it’; pull hard (Courtesy of Glenn McCulloch)
• Extension—quadriceps femoris (this muscle is three times as strong as its antagonists, the hamstrings)—(L3, L4): with the knee slightly bent, ask the patient to straighten the knee and not let you bend it (see Figure 34.32).
Ankle
• Plantar flexion—gastrocnemius, plantaris, soleus—(S1, S2): ask the patient to push the foot down and not let you push it up (see Figure 34.33).
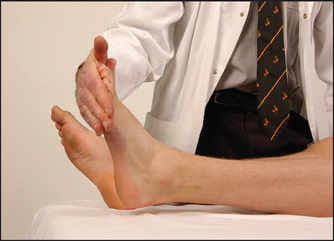
Figure 34.33 Testing power—ankle, plantar flexion: ‘Don’t let me push your foot up’ (Courtesy of Glenn McCulloch)
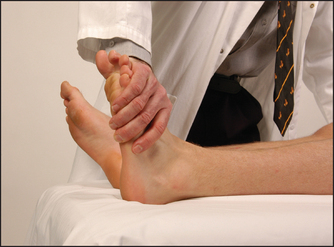
• Dorsiflexion—tibialis anterior, extensor digitorum longus and extensor hallucis longus—(L4, L5): ask the patient to bring the foot up and not let you push it down (see Figure 34.34). The power of the ankle joint can also be tested by having the patient stand up on the toes (plantar flexion) or on the heels (dorsiflexion); these movements may also be limited if coordination is impaired.
Tarsal joint
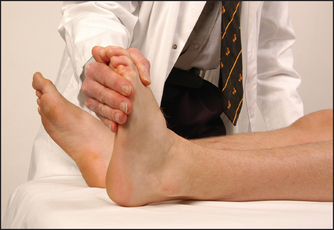
• Eversion—peroneus longus and brevis, and extensor digitorum longus—(L5, S1): ask the patient to evert the foot against resistance (see Figure 34.35).
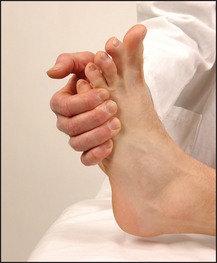
Figure 34.35 Testing power—ankle (tarsal joint) eversion: ‘Stop me turning your foot inwards’ (Courtesy of Glenn McCulloch)
• Inversion—tibialis posterior, gastrocnemius and hallucis longus—(L5, S1): with the foot in complete plantar flexion, ask the patient to invert the foot against resistance (see Figure 34.36).
Non-organic or hysterical unilateral limb weakness may be detected by Hoover’s test. Normally, when a patient attempts to resist movement, the contralateral limb works to support the effort. For example, when a patient attempts to extend the leg against resistance, the other leg pushes down into the bed. If this movement is absent, Hoover’sm sign is positive.
Quick test of lower limb power
The clinician in a hurry can test lower limb power quickly by asking the patient to:
1. stand up on his or her toes (S1) (see Figure 34.37(a))
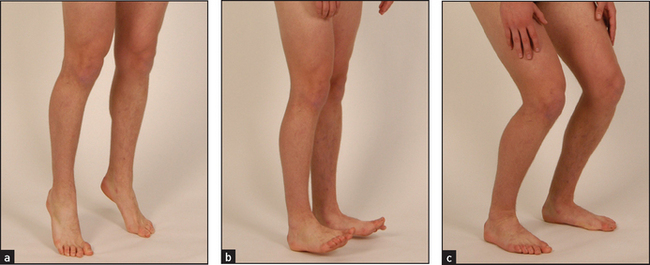
Figure 34.37 Quick test of lower limbs power (a) ‘Stand up on your toes.’ (b) ‘Now lift up on your heels.’ (c) ‘Now squat and stand up again.’ (Courtesy of Glenn McCulloch)
2. stand up on the heels (L4, L5) (see Figure 34.37(b))
3. squat and stand again (L3, L4) (see Figure 34.37(c)).
This tests ankle, knee and hip power. Inability to perform any of the tests suggests a need to test more formally.
Reflexes
Knee jerk (L3, L4)
Slide one arm under the patient’s knees so that they are slightly bent and supported. The tendon hammer is allowed to fall onto the infrapatellar tendon (see Figure 34.38). Normally, contraction of the quadriceps causes extension of the knee. Compare the two sides. If the knee jerk appears to be absent on one or both sides, it should be tested again following a reinforcement manoeuvre. Ask the patient to interlock the fingers and then pull apart hard at the moment before the hammer strikes the tendon (Jendrassik’s manoeuvre;n see Figure 34.39). This manoeuvre has been shown to restore an apparently absent ankle jerk in 70% of elderly people. A reinforcement manoeuvre such as this, or teeth-clenching or grasping an object, should be used if there is difficulty eliciting any of the muscle stretch reflexes.
Ankle jerk (S1, S2)
Have the foot in the mid-position at the ankle with the knee bent, the thigh externally rotated on the bed and the foot held in dorsiflexion by you. Allow the hammer to fall on the Achilles tendon (see Figure 34.40). The normal response is plantar flexion of the foot with contraction of the gastrocnemius muscle. Again, test with reinforcement if appropriate. This reflex can also be tested with the patient kneeling or by tapping the sole of the foot.6
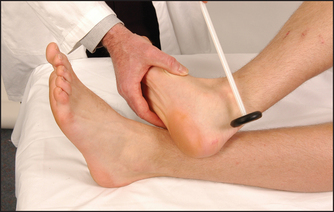
Figure 34.40 The ankle jerk (first method, see also page 365): the examiner dorsiflexes the foot slightly to stretch the tendon (Courtesy of Glenn McCulloch)
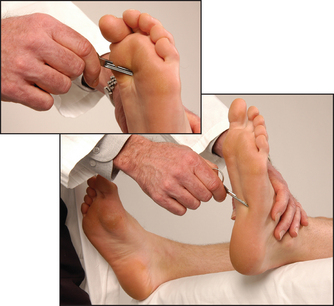
Plantar reflex (L5, S1, S2)
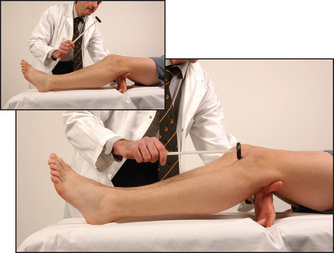
After telling the patient what is going to happen, use a blunt object (such as the key to an expensive car) to stroke up the lateral aspect of the sole and curve inwards before it reaches the toes, moving towards the middle metatarsophalangeal (MTP) joint (see Figure 34.41). The patient’s foot should be in the same position as for testing the ankle jerk. The normal response is flexion of the big toe at the MTP joint in patients over 1 year of age. The extensor (Babinskio)7 response is abnormal and is characterised by extension of the big toe at the MTP joint (the upgoing toe) and fanning of the other toes. This indicates an upper motor neurone (pyramidal) lesion, although the test’s reliability can be relatively poor. Bilateral upgoing toes may also be found after a generalised seizure, or in a patient in a coma.
Coordination
Test for cerebellar disease with three manoeuvres.
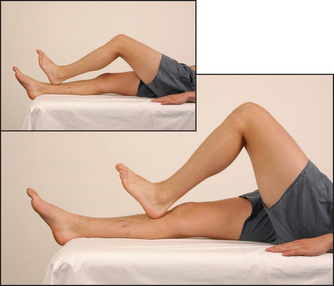
Heel–shin test
Ask the patient to run the heel of one foot up and down the opposite shin at a moderate pace and as accurately as possible (see Figure 34.42). In cerebellar disease the heel wobbles all over the place, with oscillations from side to side and overshooting. Closing the eyes makes little difference to this in cerebellar disease, but if there is posterior column loss the movements are made worse when the eyes are shut—that is, there is ‘sensory ataxia’.
The sensory system
As for the upper limb, test for pain sensation first in each dermatome, comparing the right with the left side (see Figure 34.43). Map out any abnormality and decide on the pattern of loss.
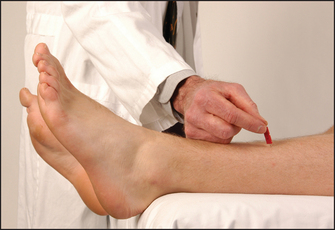
Figure 34.43 Testing pin-prick (pain) sensation in the lower limbs (do not draw blood with the pin) (Courtesy of Glenn McCulloch)
Then test vibration sense over the ankles and, if necessary, on the knees and the anterior superior iliac spines (see Figure 34.44). Next test proprioception, using the big toes (see Figure 34.45) and, if necessary, the knees and hips.
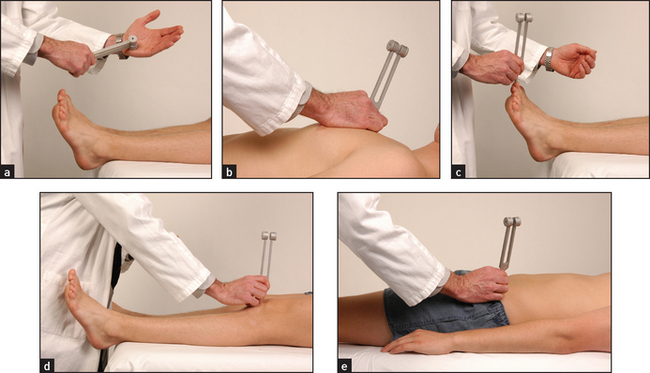
Figure 34.44 Testing vibration sense in the lower limbs (a) Strike a 128 Hz tuning fork confidently on your thenar eminence. (b) Demonstrate the vibration of the tuning fork on the patient’s sternum: ‘Can you feel this vibration?’ (c) Place the tuning fork on the great toe: ‘Can you feel the vibration there? Tell me when it stops.’ (d) If vibration sense is absent on the great toe, try testing on the patella. (e) If vibration sense is absent at the knee, try testing on the anterior superior iliac spine. (Courtesy of Glenn McCulloch)
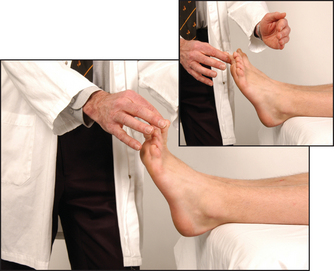
Figure 34.45 Position sense: ‘Shut your eyes and tell me whether I have moved your toe up or down’ (Courtesy of Glenn McCulloch)
Finally, test light touch (see Figure 34.46).
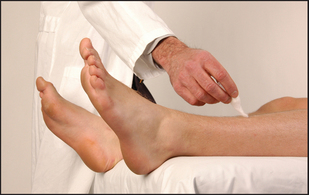
Figure 34.46 Testing light-touch sensation (do not stroke the skin with the cottonwool) (Courtesy of Glenn McCulloch)
Dermatomes
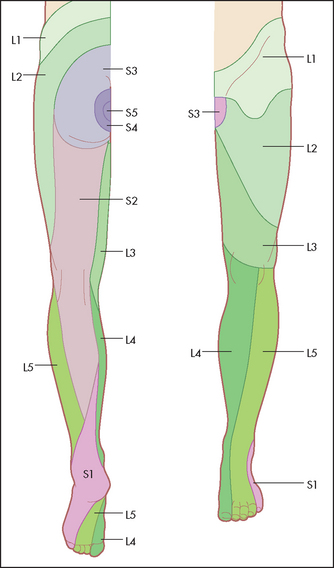
Memorise the following rough guide (see Figure 34.47):
• L2 supplies the upper anterior thigh.
• L3 supplies the area around the front of the knee.
• L4 supplies the medial aspect of the leg.
• L5 supplies the lateral aspect of the leg and the medial side of the dorsum of the foot.
• S1 supplies the heel and most of the sole.
Sensory levels
If there is peripheral sensory loss in the leg, attempt to map out the upper level with a pin, moving up at 5-centimetre intervals initially, from the leg to the abdomen, until the patient reports it to be sharp. This may involve testing over the abdominal or even the chest dermatomes. Establishing a sensory level on the trunk indicates the spinal cord level that is affected. Remember, a level of hyperaesthesia (increased sensitivity) often occurs above the sensory level and it is the upper level of this that should be determined, as it usually indicates the highest affected spinal segment. Also remember that the level of a vertebral body only corresponds to the spinal cord level in the upper cervical cord because the spinal cord is shorter than the spinal canal. The C8 spinal segment lies opposite the C7 vertebra. In the upper thoracic cord there is a difference of about two segments and in the mid-thoracic cord it is three segments. All the lumbosacral segments are opposite the T11 to L1 vertebrae.
The superficial or cutaneous reflexes
These reflexes occur in response to light touch or scratching of the skin or mucous membranes. The stimulus is more superficial than the tendon (muscle stretch) reflexes. As a rule these reflexes occur more slowly after the stimulus, are less constantly present and fatigue more easily.
Examples include the palmar or grasp reflex, the abdominal reflexes, the cremasteric reflex and the plantar responses.
The abdominal reflexes (epigastric T6–T9, mid-abdominal T9–T11, lower abdominal T11–L1)
Test these by lightly stroking the abdominal wall diagonally towards the umbilicus in each of the four quadrants of the abdomen (see Figure 34.48). Reflex contractions of the abdominal wall are absent in upper motor neurone lesions above the segmental level and also in patients who have had surgical operations interrupting the nerves. They disappear in coma and deep sleep, and during anaesthesia. They are usually difficult to elicit in obese patients and can also be absent in some healthy people (20%). Their absence in the presence of increased tendon reflexes is suggestive of corticospinal tract abnormality.
The cremasteric reflexes (L1–L2)
Stroke the inner part of the thigh in a downward direction; normally contraction of the cremasteric muscle pulls up the scrotum and testis on the side stroked. It may be absent in elderly men and in those with a hydrocele or varicocele, or after an episode of orchitis.
Saddle sensation and anal reflex
Test now for saddle sensation if a cauda equina lesion is suspected (e.g. because of urinary or faecal incontinence). The only sensory loss may be on the buttocks or around the anus (S3–S5). In this case also test the anal reflex (S2, S3, S4): normal contraction of the external sphincter in response to pin-prick of the perianal skin is abolished in patients with a lesion of the sacral segments of the cauda equina. If, however, the lowest sacral segments are spared but the higher ones are involved, this suggests that there is an intrinsic cord lesion.
Examination of the peripheral nerves of the lower limb
Lateral cutaneous nerve of the thigh
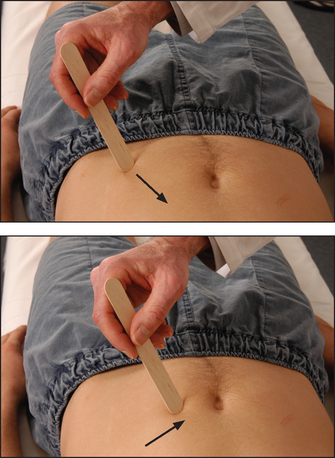
Test for sensory loss (see Figure 34.49). A lesion of this nerve usually occurs because of entrapment between the inguinal ligament and the anterior superior iliac spine. It is more common in people who are overweight and in those who spend much of their time sitting (e.g. truck drivers, public servants). It causes a sensory loss over the lateral aspect of the thigh with no motor loss detectable. If painful, it is called meralgia paraesthetica.
Femoral nerve (L2, L3, L4)
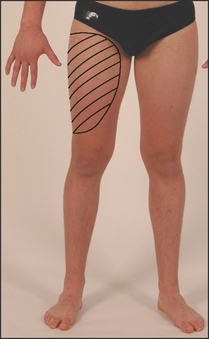
Test for weakness of knee extension (quadriceps paralysis). Hip flexion weakness is only slight, and adductor strength is preserved. The knee jerk is absent. The sensory loss involves the inner aspect of the thigh and leg (see Figure 34.50).
Sciatic nerve (L4, L5, S1, S2)
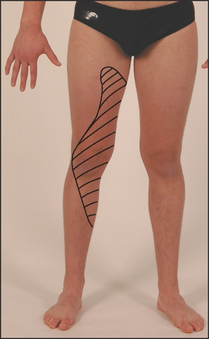
This nerve supplies all the muscles below the knee and the hamstrings. Test for loss of power below the knee resulting in a footdrop (plantar flexed foot) and for weakness of knee flexion. Test the reflexes: with a sciatic nerve lesion the knee jerk is intact but the ankle jerk and plantar response are absent. Test sensation on the posterior thigh, the lateral and posterior calf, and the foot (lost with a proximal nerve lesion; see Figure 34.51).
Common peroneal nerve (L4, L5, S1)
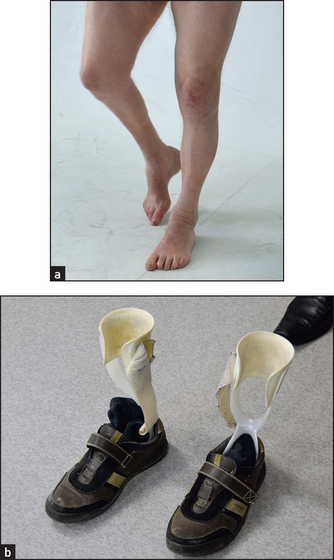
This is a major terminal branch of the sciatic nerve. It supplies the anterior and lateral compartment muscles of the leg (see Figure 34.52). On inspection one may notice a footdrop (see List 34.7 and Figure 34.53). Test for weakness of dorsiflexion and eversion. Test the reflexes, which will all be intact. Test for sensory loss. There may be only minimal sensory loss over the lateral aspect of the dorsum of the foot. Note that these findings can be confused with an L5 root lesion, but the latter includes weakness of knee flexion and loss of foot inversion as well as sensory loss in the L5 distribution.
GAIT
Make sure the patient’s legs are clearly visible. Ask the patient to walk normally for a few metres and turn around quickly and walk back and then walk heel-to-toe to exclude a midline cerebellar lesion (see Figure 34.54). Ask the patient to walk on the toes (an S1 lesion will make this difficult or impossible) and then on the heels (an L4 or L5 lesion causing footdrop will make this difficult or impossible).
Test for proximal myopathy by asking the patient to squat and then stand up, or to sit in a low chair and then stand.
To test station (Rombergp test), ask the patient to stand erect with the feet together and the eyes open (see Figure 34.55(a)) and then once the patient is stable to close the eyes (see Figure 34.55(b)). Compare the steadiness shown with the eyes open, then closed for up to 1 minute. Even in the absence of neurological disease a person may be slightly unsteady with the eyes closed, but the sign is positive if marked unsteadiness occurs to the point where the patient looks likely to fall. Normal people can maintain this position easily for 60 seconds. The Romberg test is positive when unsteadiness increases with eye closure. This is usually seen with the loss of proprioceptive sensation; unsteadiness is worse when visual information about position is removed.
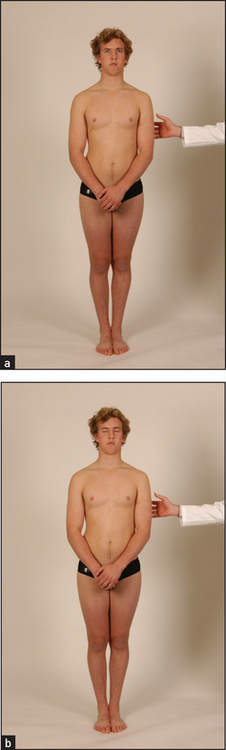
Figure 34.55 Romberg test (a) ‘Stand with your feet together.’ (b) ‘Now shut your eyes. I won’t let you fall.’ (Courtesy of Glenn McCulloch)
Marked unsteadiness with the eyes open is seen with cerebellar or vestibular dysfunction.
Gait disorders are summarised in List 34.8.
References
1. Freeman, C, Okun, MS. Origins of the sensory examination in neurology. Semin Neurol. 2002; 22:399–407.
2. Nelson, KR. Use new pins for each neurologic examination [Letter]. New Engl J Med. 1986; 314:581. [A cautionary tale.].
3. Medical Research CouncilAids to the investigation of peripheral nerve injury. London: Her Majesty’s Stationery Office, 1972. [Presents, in a clear and straightforward manner, bedside methods for testing the innervation of all important muscles. An invaluable guide.].
4. Katz, JN, Larson, MG, Sabra, A, et al. The carpal tunnel syndrome: diagnostic utility of the history and physical examination findings. Ann Intern Med. 1990; 112:321–327. [Unfortunately, individual symptoms and signs have limited diagnostic usefulness. Tinel’s sign appears to be of little value.].
5. D’Arcy, DA, McGee, S. The rational clinical examination. Does this patient have carpal tunnel syndrome. JAMA. 2000; 283:3110–3117. [Weak thumb abduction and self-reported sensory symptoms (drawn on a diagram) are useful to predict abnormal median nerve conduction testing.].
6. Schwartz, RS, et al. A comparison of two methods of eliciting the ankle jerk. Aust NZ J Med. 1990; 20:116–119. [The ankle jerk can be tested by tapping the sole of the foot.].
7. Lance, JW. The Babinski sign. J Neurol Neurosurg Psychiatry. 2002; 73(4):360–362. [A clear explanation of the history and clinical relevance of this most important of neurological signs.].
aAretaeus of Cappadocia reasoned, in 150 AD, that the nerves cross (decussate) between the brain and the periphery and that injury to the right side of the head causes abnormalities of the left side of the body.
bIf no fasciculation is seen, tapping over the bulk of the brachioradialis and biceps muscles with the finger or with a tendon hammer and watching again has been recommended, but this is controversial. Most neurologists do not do this. The reason is that fasciculations are spontaneous. Any muscle movement from a local stimulus is not spontaneous. Even if they occur, they may have nothing to do with fasciculations.
cFrom the German meaning ‘counterpressure’ or ‘standing your ground’.
dYou should not be able to overcome a normal adult patient’s power, at least in the legs.
eSir William Gowers (1845–1915), professor of clinical medicine at University College Hospital, London, and neurologist to the National Hospital for Nervous Diseases, Queen Square, London. He was also an artist who illustrated his own books and had paintings exhibited at the Royal Academy.
fThe Queen Square hammer was invented by Miss Wintle, staff nurse at Queen Square. She made hammers from brass wheels covered by a ring pessary and mounted on a bamboo handle; she sold these to medical students and resident medical officers.
gActually, dysdiadochokinesis is the inability to perform alternating movements of both wrists with the arms and forefingers extended. Diadochi is a Greek word meaning ‘succession’. The problem here is with successive movements. The Diadochi were the successors of Alexander the Great. They divided his empire.
hNick Talley has a black belt in Tae Kwon Do and Tang Soo Do.
iIn 1826 Sir Charles Bell recognised that there was a ‘sixth sense’, which was later called proprioception. Vibration sense had been recognised in the 16th century and tests for it were developed in the 19th century by Rinné and others. Rydel and Seiffer found that vibration sense and proprioception were carried in the posterior columns of the spinal cord.
jThe human dermatomes (which he called pain spots) were first mapped by Sir Henry Head (1861–1940). He was most famous for his experimental cutting of his own radial nerve. This enabled him to chart the order of return of the sensory modalities.
kAlbert Ochsner (1858–1925), American surgeon of Swiss extraction, who claimed descent from Andreas Vesalius, the great anatomist.
lJules Froment (1878–1946), Professor of Medicine, Lyons, France, described the sign in 1915.
mCharles Hoover also described an important sign of chronic obstructive pulmonary disease.
nErnst Jendrassik (1858–1921), Budapest physician.
oJosef Babinski (1857–1932), Parisian neurologist of Polish extraction, described this sign in 1896. Somatisation disorders vary in their nature according to fashion. In the 19th century, hysterical paralysis was common. Babinski, while strolling through his ward, touched the soles of the feet of two patients in adjoining beds (this would probably lead to his being struck off the medical register today). He noticed that the big toe of the woman thought to have hysterical paralysis moved down and that of the woman whose paralysis was thought genuine and due to a stroke moved up. The sign was probably first described by Ernst Remak in 1893. Babinski was a famous gourmet and assistant to Charcot.
pMoritz Heinrich von Romberg (1795–1873), Berlin professor, wrote the first modern neurology textbook. His original description of the sign was of patients with tabes dorsalis (dorsal column disease caused by syphilis).